
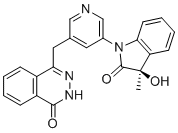

RP 12146, EX-A7910
cas 2732869-64-4
4-[[5-[(3R)-3-hydroxy-3-methyl-2-oxoindol-1-yl]pyridin-3-yl]methyl]-2H-phthalazin-1-one
| M.Wt | 398.422 | |
| Formula | C23H18N4O3 | |
RP12146 (RP12146) is a novel, selective, and potent small molecule inhibitor of PARP1/2 with IC50 of 0.6/0.5 nM, with several fold selectivity over other isoforms.
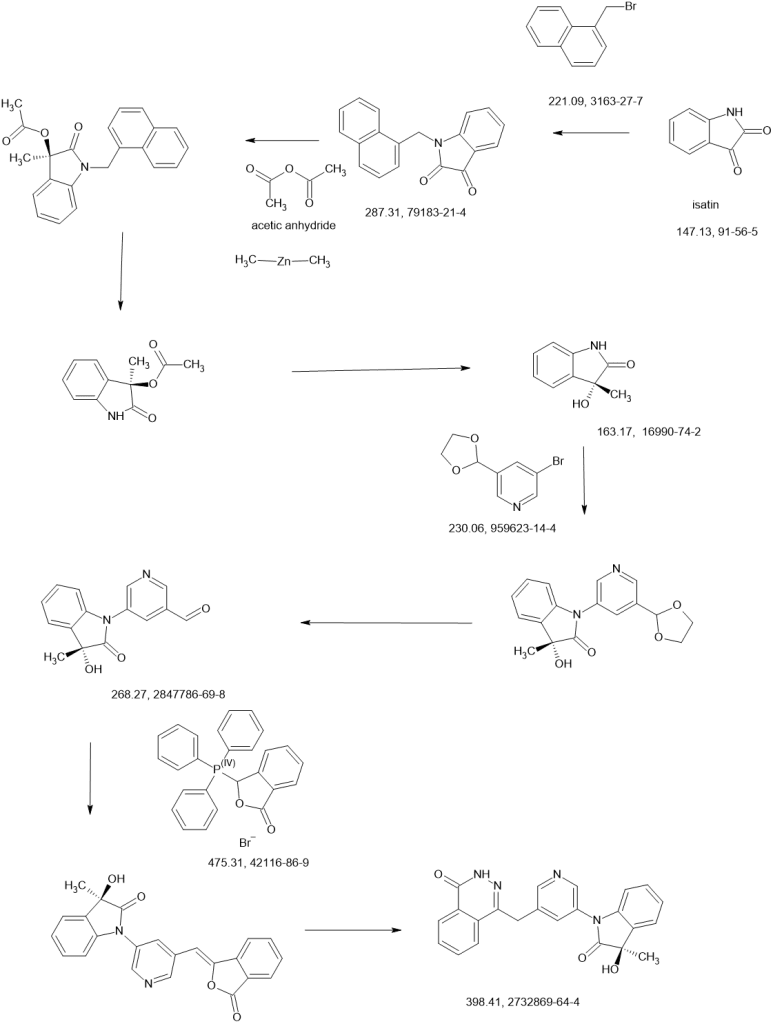
SCHEME
Ref
WO2021220120
Rhizen Pharmaceuticals AG
WO2022215034
Rhizen Pharmaceuticals AG; Incozen Therapeutics Pvt. Ltd.
RP-12146 is an oral poly (ADP-ribose) polymerase (PARP) inhibitor in phase I clinical development at Rhizen Pharmaceuticals for the treatment of adult patients with locally advanced or metastatic solid tumors.
Solid TumorExtensive-stage Small-cell Lung CancerLocally Advanced Breast CancerMetastatic Breast CancerPlatinum-sensitive Ovarian CancerPlatinum-Sensitive Fallopian Tube CarcinomaPlatinum-Sensitive Peritoneal Cancer
Poly(ADP-ribose) polymerase (PARP) defines a family of 17 enzymes that cleaves NAD+ to nicotinamide and ADP-ribose to form long and branched (ADP-ribose) polymers on glutamic acid residues of a number of target proteins, including PARP itself. The addition of negatively charged polymers profoundly alters the properties and functions of the acceptor proteins. Poly(ADP-ribosyl)ation is involved in the regulation of many cellular processes, such as DNA repair, gene transcription, cell cycle progression, cell death, chromatin functions and genomic stability. These functions have been mainly attributed to PARP-1 that is regarded as the best characterized member of the PARP family. However, the identification of novel genes encoding PARPs, together with the characterization of their structure and subcellular localization, have disclosed different roles for poly(ADP-ribosyl)ation in cells, including telomere replication and cellular transport.
Recently, poly(ADP-ribose) binding sites have been identified in many DNA damage checkpoint proteins, such as tumor suppressor p53, cyclin-dependent kinase inhibitor p21Cip1/waf1, DNA damage recognition factors (i.e., the nucleotide excision repair xeroderma pigmentosum group A complementing protein and the mismatch repair protein MSH6), base excision repair (BER) proteins (i.e. DNA ligase III, X-ray repair cross-complementing 1, and XRCC1), DNA-dependent protein kinase (DNA-PK), cell death and survival regulators (i.e.,
NF-kB, inducible nitric oxide synthase, and telomerase). These findings suggest that the different components of the PARP family might be involved in the DNA damage signal network, thus regulating protein-protein and protein-DNA interactions and, consequently, different types of cellular responses to genotoxic stress. In addition to its involvement in BER and single strand breaks (SSB) repair, PARP-1 appears to aid in the non-homologous end-joining (NHEJ) and homologous recombination (HR) pathways of double strand breaks (DSB) repair. See Lucio Tentori et al., Pharmacological Research, Vol. 45, No. 2, 2002, page 73-85.
PARP inhibition might be a useful therapeutic strategy not only for the treatment of BRCA mutations but also for the treatment of a wider range of tumors bearing a variety of deficiencies in the HR pathway. Further, the existing clinical data (e.g., Csaba Szabo et al., British Journal of Pharmacology (2018) 175: 192-222) also indicate that stroke, traumatic brain injury, circulatory shock and acute myocardial infarction are some of the indications where PARP activation has been demonstrated to contribute to tissue necrosis and inflammatory responses.
As of now, four PARP inhibitors, namely olaparib, talazoparib, niraparib, and rucaparib have been approved for human use by regulatory authorities around the world.
Patent literature related to PARP inhibitors includes International Publication Nos. WO 2000/42040, WO 2001/016136, WO 2002/036576, WO 2002/090334, WO2003/093261, WO 2003/106430, WO 2004/080976, WO 2004/087713, WO 2005/012305, WO 2005/012524, WO 2005/012305, WO 2005/012524, WO 2005/053662, W02006/033003, W02006/033007, WO 2006/033006, WO 2006/021801, WO 2006/067472, WO 2007/144637, WO 2007/144639, WO 2007/144652, WO 2008/047082, WO 2008/114114, WO 2009/050469, WO 2011/098971, WO 2015/108986, WO 2016/028689, WO 2016/165650, WO 2017/153958, WO 2017/191562, WO 2017/123156, WO 2017/140283, WO 2018/197463, WO 2018/038680 and WO 2018/108152, each of which is incorporated herein by reference in its entirety for all purposes.
There still remains an unmet need for new PARP inhibitors for the treatment of various diseases and disorders associated with cell proliferation, such as cancer.
PATENT
Illustration 1
CLIP
https://cancerres.aacrjournals.org/content/81/13_Supplement/1233
Abstract 1233: Preclinical profile of RP12146, a novel, selective, and potent small molecule inhibitor of PARP1/2
Srikant Viswanadha, Satyanarayana Eleswarapu, Kondababu Rasamsetti, Debnath Bhuniya, Gayatriswaroop Merikapudi, Sridhar Veeraraghavan and Swaroop VakkalankaProceedings: AACR Annual Meeting 2021; April 10-15, 2021 and May 17-21, 2021; Philadelphia, PA
Abstract
Background: Poly (ADP-ribose) polymerase (PARP) activity involves synthesis of Poly-ADP ribose (PAR) polymers that recruit host DNA repair proteins leading to correction of DNA damage and maintenance of cell viability. Upon combining with DNA damaging cytotoxic agents, PARP inhibitors have been reported to demonstrate chemo- and radio-potentiation albeit with incidences of myelosuppression. A need therefore exists for the development selective PARP1/2 inhibitors with a high therapeutic window to fully exploit their potential as a single agent or in combination with established therapy across various tumor types. Additionally, with the emerging concept of ‘synthetic lethality’, the applicability PARP inhibitors can be expanded to cancers beyond the well-defined BRCA defects. Herein, we describe the preclinical profile of RP12146, a novel and selective small molecule inhibitor of PARP1 and PARP2.
Methods: Enzymatic potency was evaluated using a PARP Chemiluminescent Activity Assay Kit (BPS biosciences). Cell growth was determined following incubation with RP12146 in BRCA1 mutant and wild-type cell lines across indications. Apoptosis was evaluated following incubation of cell lines with compound for 120 h, subsequent staining with Annexin-V-PE and 7-AAD, and analysis by flow cytometry. For cell cycle, cells were incubated with compound for 72 h, and stained with Propidium Iodide prior to analysis by flow cytometry. Expression of downstream PAR, PARP-trapping, phospho-γH2AX and cleaved PARP expression were determined in UWB1.289 (BRCA1 null) cells by Western blotting. Anti-tumor potential of RP12146 was tested in OVCAR-3 Xenograft model. Pharmacokinetic properties of the molecule were also evaluated. Results: RP12146 demonstrated equipotent inhibition of PARP1 (0.6 nM) and PARP2 (0.5 nM) with several fold selectivity over the other members of the PARP family. Compound caused a dose-dependent growth inhibition of both BRCA mutant and non-mutant cancer cell lines with GI50 in the range of 0.04 µM to 9.6 µM. Incubation of UWB1.289 cells with RP12146 caused a G2/M arrest with a corresponding dose-dependent increase in the percent of apoptotic cells. Expression of PAR was inhibited by 86% at 10 nM with a 2.3-fold increase in PARP-trapping observed at 100 nM in presence of RP12146. A four-fold increase in phospho-γH2AX and > 2-fold increase in cleaved PARP expression was observed at 3 µM of the compound. RP12146 exhibited anti-tumor potential with TGI of 28% as a single agent in OVCAR-3 xenograft model. Efficay was superior compared to Olaparib tested at an equivalent dose. Pharmacokinetic studies in rodents indicated high bioavailability with favorable plasma concentrations relevant for efficacy
Conclusions: Data demonstrate the therapeutic potential of RP12146 in BRCA mutant tumors. Testing in patients is planned in H1 2021.
Citation Format: Srikant Viswanadha, Satyanarayana Eleswarapu, Kondababu Rasamsetti, Debnath Bhuniya, Gayatriswaroop Merikapudi, Sridhar Veeraraghavan, Swaroop Vakkalanka. Preclinical profile of RP12146, a novel, selective, and potent small molecule inhibitor of PARP1/2 [abstract]. In: Proceedings of the American Association for Cancer Research Annual Meeting 2021; 2021 Apr 10-15 and May 17-21. Philadelphia (PA): AACR; Cancer Res 2021;81(13_Suppl):Abstract nr 1233.

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////////////////////////////
CLIP
Rhizen Pharmaceuticals AG Announces First Patient Dosing in a Phase I/Ib Study of Its Novel PARP Inhibitor (RP12146) in Patients With Advanced Solid Tumors
RHIZEN’S PARP INHIBITOR EFFORTS ARE PART OF A LARGER DDR PLATFORM THAT ALSO INCLUDES AN EARLY STAGE POLθ-DIRECTED PROGRAM; PLATFORM ENABLES PROPRIETARY IN-HOUSE COMBINATIONS
- Rhizen Pharma commences dosing in a phase I/Ib trial to evaluate its novel PARP inhibitor (RP12146) in patients with advanced cancers.
- Rhizen indicated that RP12146 has comparable preclinical activity vis-à-vis approved PARP inhibitors and shows improved preclinical safety that it expects will translate in the clinic.
- The two-part multi-center phase I/Ib study is being conducted in Europe and is designed to initially determine safety, tolerability and MTD/RP2D of RP12146 and to subsequently assess its anti-tumor activity in expansion cohorts with HRR mutation-enriched ES-SCLC, ovarian and breast cancer patients.
- RP12146 is part of a larger DDR platform at Rhizen that includes a preclinical-stage Polθ inhibitor program; the DDR platform enables novel, proprietary, in-house combinations
November 01, 2021 07:24 AM Eastern Daylight Time
BASEL, Switzerland–(BUSINESS WIRE)–Rhizen Pharmaceuticals AG (Rhizen), a Switzerland-based privately held, clinical-stage oncology & inflammation-focused biopharmaceutical company, announced today that it has commenced dosing in a multi-center, phase I/Ib trial to evaluate its novel poly (ADP-ribose) polymerase (PARP) inhibitor (RP12146) in patients with advanced solid tumors. This two-part multi-center phase I/Ib study is being conducted in Europe and has been designed to initially determine safety, tolerability, maximum tolerated dose (MTD), and/or recommended phase II dose (RP2D) of RP12146 and to subsequently assess its anti-tumor activity in expansion cohorts with HRR mutation-enriched ES-SCLC, ovarian and breast cancer patients.
“Our PARP program is foundational for our DDR platform efforts and will be the backbone for several novel proprietary combinations that we hope to bring into development going forward.”
Rhizen indicated that RP12146 has shown preclinical activity and efficacy comparable to the approved PARP inhibitor Olaparib, and shows improved safety as seen in the preclinical IND-enabling toxicology studies; an advantage that Rhizen hopes will translate in the clinical studies. Rhizen also announced that its PARP program is part of a larger DNA Damage Response (DDR) platform effort, which includes a preclinical-stage polymerase theta (Polθ) inhibitor program. Rhizen expects the platform to enable novel proprietary combinations of its PARP and Polθ assets given the mechanistic synergy and opportunity across PARP resistant/refractory settings.
“PARP inhibitors are a great success story in the DNA damage response area, but they are not without safety concerns that have limited realization of their full potential. Although our novel PARP inhibitor is competing in a crowded space, we expect its superior preclinical safety to translate into the clinic which will differentiate our program and allow us to extend its application beyond the current landscape of approved indications and combinations”, said Swaroop Vakkalanka, Founder & CEO of Rhizen Pharma. Swaroop also added that “Our PARP program is foundational for our DDR platform efforts and will be the backbone for several novel proprietary combinations that we hope to bring into development going forward.”
About Rhizen Pharmaceuticals AG.:
Rhizen Pharmaceuticals is an innovative, clinical-stage biopharmaceutical company focused on the discovery and development of novel oncology & inflammation therapeutics. Since its establishment in 2008, Rhizen has created a diverse pipeline of proprietary drug candidates targeting several cancers and immune associated cellular pathways.
Rhizen has proven expertise in the PI3K modulator space with the discovery of our first PI3Kδ & CK1ε asset Umbralisib, that has been successfully developed & commercialized in MZL & FL by our licensing partner TG Therapeutics (TGTX) in USA. Beyond this, Rhizen has a deep oncology & inflammation pipeline spanning discovery to phase II clinical development stages.
Rhizen is headquartered in Basel, Switzerland.
REF
Safety, Pharmacokinetics and Anti-tumor Activity of RP12146, a PARP Inhibitor, in Patients With Locally Advanced or Metastatic Solid Tumors….https://clinicaltrials.gov/ct2/show/NCT05002868
//////////RP 12146, oral poly (ADP-ribose) polymerase (PARP) inhibitor, phase I, clinical development, INCOZEN, Rhizen Pharmaceuticals, adult patients, locally advanced, metastatic solid tumors, PARP, CANCER, EX-A7910, EX A7910

NEW DRUG APPROVALS
ONE TIME
$10.00

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....

*Dear Anthony Melvin Crasto* Dr. *Greetings from ALMAC Enterprises India.* *Shall i add you on whatsapp*
*Thanks & Regards* *Khurram Ahmed* *Hyderabad, India.* *Whatsapp: 0091 8885286217*
On Thu, Nov 11, 2021 at 8:46 AM New Drug Approvals wrote:
> DR ANTHONY MELVIN CRASTO Ph.D posted: ” RP 12146 RP-12146 is an oral poly > (ADP-ribose) polymerase (PARP) inhibitor in phase I clinical development at > Rhizen Pharmaceuticals for the treatment of adult patients with locally > advanced or metastatic solid tumors. Solid TumorExtensive-stag” >
LikeLike