Home » FDA 2020
Category Archives: FDA 2020
Imdevimab
| (Heavy chain) QVQLVESGGG VVQPGRSLRL SCAASGFTFS NYAMYWVRQA PGKGLEWVAV ISYDGSNKYY ADSVKGRFTI SRDNSKNTLY LQMNSLRTED TAVYYCASGS DYGDYLLVYW GQGTLVTVSS ASTKGPSVFP LAPSSKSTSG GTAALGCLVK DYFPEPVTVS WNSGALTSGV HTFPAVLQSS GLYSLSSVVT VPSSSLGTQT YICNVNHKPS NTKVDKKVEP KSCDKTHTCP PCPAPELLGG PSVFLFPPKP KDTLMISRTP EVTCVVVDVS HEDPEVKFNW YVDGVEVHNA KTKPREEQYN STYRVVSVLT VLHQDWLNGK EYKCKVSNKA LPAPIEKTIS KAKGQPREPQ VYTLPPSRDE LTKNQVSLTC LVKGFYPSDI AVEWESNGQP ENNYKTTPPV LDSDGSFFLY SKLTVDKSRW QQGNVFSCSV MHEALHNHYT QKSLSLSPGK (Light chain) QSALTQPASV SGSPGQSITI SCTGTSSDVG GYNYVSWYQQ HPGKAPKLMI YDVSKRPSGV SNRFSGSKSG NTASLTISGL QSEDEADYYC NSLTSISTWV FGGGTKLTVL GQPKAAPSVT LFPPSSEELQ ANKATLVCLI SDFYPGAVTV AWKADSSPVK AGVETTTPSK QSNNKYAASS YLSLTPEQWK SHRSYSCQVT HEGSTVEKTV APTECS (Disulfide bridge: H22-H96, H147-H203, H223-L215, H229-H’229, H264-H324-H370-H428, H’22-H’96, H’147-H’203, H’223-L’215, H’264-H’324, H’370-H’428, L22-L90, L138-L197, L’22-L’90, L’138-L’197) |
イムデビマブ;
- Immunoglobulin G1, anti-(severe acute respiratory syndrome coronavirus 2 spike glycoprotein) (human monoclonal REGN10987 γ1-chain), disulfide with human monoclonal REGN10987 λ-chain, dimer
| Formula | C6396H9882N1694O2018S42 |
|---|---|
| CAS | 2415933-40-1 |
| Mol weight | 144141.7693 |
Monoclonal antibody
Treatment and prophylaxis of SARS-CoV-2 infection
ANTIVIRAL
SARS-CoV-2 spike glycoprotein
- REGN 10987
- RG 6412
Fact Sheet – US Food and Drug Administration
https://www.fda.gov › media › download
PDFBenefit of treatment with casirivimab and imdevimab has not been observed in patients hospitalized due to COVID-19. Monoclonal antibodies, such as casirivimab.
Casirivimab/imdevimab, sold under the brand name REGEN-COV,[1] is an experimental medicine developed by the American biotechnology company Regeneron Pharmaceuticals. It is an artificial “antibody cocktail” designed to produce resistance against the SARS-CoV-2 coronavirus responsible for the COVID-19 pandemic.[3][4] It consists of two monoclonal antibodies, casirivimab (REGN10933) and imdevimab (REGN10987) that must be mixed together.[1][5][6] The combination of two antibodies is intended to prevent mutational escape.[7]
Trials
In a clinical trial of people with COVID-19, casirivimab and imdevimab, administered together, were shown to reduce COVID-19-related hospitalization or emergency room visits in people at high risk for disease progression within 28 days after treatment when compared to placebo.[2] The safety and effectiveness of this investigational therapy for use in the treatment of COVID-19 continues to be evaluated.[2]
The data supporting the emergency use authorization (EUA) for casirivimab and imdevimab are based on a randomized, double-blind, placebo-controlled clinical trial in 799 non-hospitalized adults with mild to moderate COVID-19 symptoms.[2] Of these participants, 266 received a single intravenous infusion of 2,400 milligrams casirivimab and imdevimab (1,200 mg of each), 267 received 8,000 mg casirivimab and imdevimab (4,000 mg of each), and 266 received a placebo, within three days of obtaining a positive SARS-CoV-2 viral test.[2]
The prespecified primary endpoint for the trial was time-weighted average change in viral load from baseline.[2] Viral load reduction in participants treated with casirivimab and imdevimab was larger than in participants treated with placebo at day seven.[2] However, the most important evidence that casirivimab and imdevimab administered together may be effective came from the predefined secondary endpoint of medically attended visits related to COVID-19, particularly hospitalizations and emergency room visits within 28 days after treatment.[2] For participants at high risk for disease progression, hospitalizations and emergency room visits occurred in 3% of casirivimab and imdevimab-treated participants on average compared to 9% in placebo-treated participants.[2] The effects on viral load, reduction in hospitalizations and ER visits were similar in participants receiving either of the two casirivimab and imdevimab doses.[2]
As of September 2020, REGEN-COV is being evaluated as part of the RECOVERY Trial.[8]
On 12 April 2021, Roche and Regeneron announced that the Phase III clinical trial REGN-COV 2069 met both primary and secondary endpoints, reducing risk of infection by 81% for the non-infected patients, and reducing time-to-resolution of symptoms for symptomatic patients to one week vs. three weeks in the placebo group.[9]
Authorization
On 21 November 2020, the U.S. Food and Drug Administration (FDA) issued an emergency use authorization (EUA) for casirivimab and imdevimab to be administered together for the treatment of mild to moderate COVID-19 in people twelve years of age or older weighing at least 40 kilograms (88 lb) with positive results of direct SARS-CoV-2 viral testing and who are at high risk for progressing to severe COVID-19.[2][10][11] This includes those who are 65 years of age or older or who have certain chronic medical conditions.[2] Casirivimab and imdevimab must be administered together by intravenous (IV) infusion.[2]
Casirivimab and imdevimab are not authorized for people who are hospitalized due to COVID-19 or require oxygen therapy due to COVID-19.[2] A benefit of casirivimab and imdevimab treatment has not been shown in people hospitalized due to COVID-19.[2] Monoclonal antibodies, such as casirivimab and imdevimab, may be associated with worse clinical outcomes when administered to hospitalized people with COVID-19 requiring high flow oxygen or mechanical ventilation.[2]
The EUA was issued to Regeneron Pharmaceuticals Inc.[2][10][12]
On 1 February 2021, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) started a rolling review of data on the REGN‑COV2 antibody combination (casirivimab/imdevimab), which is being co-developed by Regeneron Pharmaceuticals, Inc. and F. Hoffman-La Roche, Ltd (Roche) for the treatment and prevention of COVID‑19.[13][14] In February 2021, the CHMP concluded that the combination, also known as REGN-COV2, can be used for the treatment of confirmed COVID-19 in people who do not require supplemental oxygen and who are at high risk of progressing to severe COVID-19.[15]
The Central Drugs Standards Control Organisation (CDSCO) in India, on 5 May 2021, granted an Emergency Use Authorisation to Roche (Genentech)[16] and Regeneron[17] for use of the casirivimab/imdevimab cocktail in the country. The announcement came in light of the second wave of the COVID-19 pandemic in India. Roche India maintains partnership with Cipla, thereby permitting the latter to market the drug in the country.[18]
Deployment
Although Regeneron is headquartered in Tarrytown, New York (near New York City), REGEN-COV is manufactured at the company’s primary U.S. manufacturing facility in Rensselaer, New York (near the state capital at Albany).[19] In September 2020, to free up manufacturing capacity for REGEN-COV, Regeneron began to shift production of its existing products from Rensselaer to the Irish city of Limerick.[20]
Regeneron has a deal in place with Roche (Genentech)[21]to manufacture and market REGEN-COV outside the United States.[10][22]
On 2 October 2020, Regeneron Pharmaceuticals announced that US President Donald Trump had received “a single 8 gram dose of REGN-COV2” after testing positive for SARS-CoV-2.[23][24] The drug was provided by the company in response to a “compassionate use” (temporary authorization for use) request from the president’s physicians.[23]
References
- ^ Jump up to:a b c “REGEN-COV- casirivimab and imdevimab kit”. DailyMed. Retrieved 18 March 2021.
- ^ Jump up to:a b c d e f g h i j k l m n o p q “Coronavirus (COVID-19) Update: FDA Authorizes Monoclonal Antibodies for Treatment of COVID-19”. U.S. Food and Drug Administration (FDA) (Press release). 21 November 2020. Retrieved 21 November 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Kelland K (14 September 2020). “Regeneron’s antibody drug added to UK Recovery trial of COVID treatments”. Reuters. Retrieved 14 September 2020.
- ^ “Regeneron’s COVID-19 Response Efforts”. Regeneron Pharmaceuticals. Retrieved 14 September 2020.
- ^ Morelle R (14 September 2020). “Antibody treatment to be given to Covid patients”. BBC News Online. Retrieved 14 September2020.
- ^ “Safety, Tolerability, and Efficacy of Anti-Spike (S) SARS-CoV-2 Monoclonal Antibodies for Hospitalized Adult Patients With COVID-19”. ClinicalTrials. 3 September 2020. Retrieved 14 September2020.
- ^ Baum A, Fulton BO, Wloga E, Copin R, Pascal KE, Russo V, et al. (August 2020). “Antibody cocktail to SARS-CoV-2 spike protein prevents rapid mutational escape seen with individual antibodies”. Science. 369 (6506): 1014–1018. Bibcode:2020Sci…369.1014B. doi:10.1126/science.abd0831. PMC 7299283. PMID 32540904.
- ^ “RECOVERY COVID-19 phase 3 trial to evaluate Regeneron’s REGN-COV2 investigational antibody cocktail in the UK”. Recovery Trial. Retrieved 14 September 2020.
- ^ “Phase III prevention trial showed subcutaneous administration of investigational antibody cocktail casirivimab and imdevimab reduced risk of symptomatic COVID-19 infections by 81%”. streetinsider.com. Archived from the original on 2021-04-12. Retrieved 2021-04-12.
- ^ Jump up to:a b c “Regeneron Reports Positive Interim Data with REGEN-COV Antibody Cocktail used as Passive Vaccine to Prevent COVID-19”(Press release). Regeneron Pharmaceuticals. 26 January 2021. Retrieved 19 March 2021 – via PR Newswire.
- ^ “Fact Sheet For Health Care Providers Emergency Use Authorization (EUA) Of Casirivimab And Imdevimab” (PDF). U.S. Food and Drug Administration (FDA).
- ^ “Casirivimab and Imdevimab”. Regeneron Pharmaceuticals. Retrieved 19 March 2021.
- ^ “EMA starts rolling review of REGN‑COV2 antibody combination (casirivimab / imdevimab)” (Press release). European Medicines Agency (EMA). 1 February 2021. Retrieved 1 February 2021. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ “EMA reviewing data on monoclonal antibody use for COVID-19” (Press release). European Medicines Agency (EMA). 4 February 2021. Retrieved 4 March 2021.
- ^ “EMA issues advice on use of REGN-COV2 antibody combination (casirivimab / imdevimab)” (Press release). European Medicines Agency (EMA). 26 February 2021. Retrieved 5 March 2021. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^https://www.businesswire.com/news/home/20200818005847/en/Genentech-and-Regeneron-Collaborate-to-Significantly-Increase-Global-Supply-of-REGN-COV2-Investigational-Antibody-Combination-for-COVID-19
- ^ https://timesofindia.indiatimes.com/india/india-approves-roche/regeneron-antibody-cocktail-to-treat-covid-19/articleshow/82407551.cms
- ^ “Roche receives Emergency Use Authorisation in India for its investigational Antibody Cocktail (Casirivimab and Imdevimab) used in the treatment of Covid-19 | Cipla”. http://www.cipla.com. Retrieved 2021-05-06.
- ^ Williams, Stephen (3 October 2020). “Experimental drug given to President made locally”. The Daily Gazette.
- ^ Stanton, Dan (11 September 2020). “Manufacturing shift to Ireland frees up US capacity for Regeneron’s COVID antibodies”. BioProcess International.
- ^https://www.businesswire.com/news/home/20200818005847/en/Genentech-and-Regeneron-Collaborate-to-Significantly-Increase-Global-Supply-of-REGN-COV2-Investigational-Antibody-Combination-for-COVID-19
- ^ “Roche and Regeneron link up on a coronavirus antibody cocktail”. CNBC. 19 August 2020. Retrieved 14 September 2020.
- ^ Jump up to:a b Thomas K (2 October 2020). “President Trump Received Experimental Antibody Treatment”. The New York Times. ISSN 0362-4331. Retrieved 2 October 2020.
- ^ Hackett DW (3 October 2020). “8-Gram Dose of COVID-19 Antibody Cocktail Provided to President Trump”. http://www.precisionvaccinations.com. Archived from the original on 3 October 2020.
External links
- “Casirivimab”. Drug Information Portal. U.S. National Library of Medicine.
- “Imdevimab”. Drug Information Portal. U.S. National Library of Medicine.
- “Casirivimab and Imdevimab EUA Letter of Authorization” (PDF). U.S. Food and Drug Administration (FDA).
- “Frequently Asked Questions on the Emergency Use Authorization of Casirivimab + Imdevimab” (PDF). U.S. Food and Drug Administration (FDA).
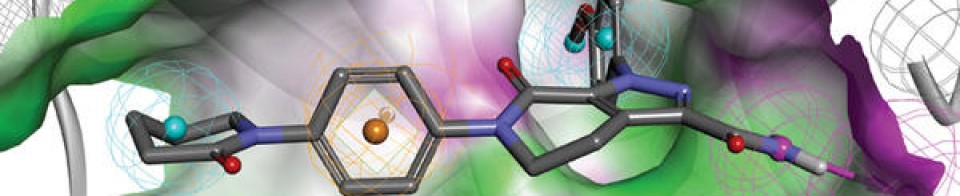
| REGN10933 (blue) and REGN10987 (orange) bound to SARS-CoV-2 spike protein (pink). From PDB: 6VSB, 6XDG. | |
| Combination of | |
|---|---|
| Casirivimab | Monoclonal antibody against spike protein of SARS-CoV-2 |
| Imdevimab | Monoclonal antibody against spike protein of SARS-CoV-2 |
| Clinical data | |
| Trade names | REGEN-COV |
| Other names | REGN-COV2 |
| AHFS/Drugs.com | Monograph |
| License data | US DailyMed: Casirivimab |
| Routes of administration | Intravenous |
| ATC code | None |
| Legal status | |
| Legal status | US: Unapproved (Emergency Use Authorization)[1][2] |
| Identifiers | |
| DrugBank | DB15691 |
| KEGG | D11938D11939 |
////////Imdevimab, ANTI VIRAL, PEPTIDE, CORONA VIRUS, COVID19, APPROVALS 2020, FDA 2020, イムデビマブ, REGN 10987, RG 6412,
NEW DRUG APPROVALS
one time
$10.00

Casirivimab
(Heavy chain)
QVQLVESGGG LVKPGGSLRL SCAASGFTFS DYYMSWIRQA PGKGLEWVSY ITYSGSTIYY
ADSVKGRFTI SRDNAKSSLY LQMNSLRAED TAVYYCARDR GTTMVPFDYW GQGTLVTVSS
ASTKGPSVFP LAPSSKSTSG GTAALGCLVK DYFPEPVTVS WNSGALTSGV HTFPAVLQSS
GLYSLSSVVT VPSSSLGTQT YICNVNHKPS NTKVDKKVEP KSCDKTHTCP PCPAPELLGG
PSVFLFPPKP KDTLMISRTP EVTCVVVDVS HEDPEVKFNW YVDGVEVHNA KTKPREEQYN
STYRVVSVLT VLHQDWLNGK EYKCKVSNKA LPAPIEKTIS KAKGQPREPQ VYTLPPSRDE
LTKNQVSLTC LVKGFYPSDI AVEWESNGQP ENNYKTTPPV LDSDGSFFLY SKLTVDKSRW
QQGNVFSCSV MHEALHNHYT QKSLSLSPGK
(Light chain)
DIQMTQSPSS LSASVGDRVT ITCQASQDIT NYLNWYQQKP GKAPKLLIYA ASNLETGVPS
RFSGSGSGTD FTFTISGLQP EDIATYYCQQ YDNLPLTFGG GTKVEIKRTV AAPSVFIFPP
SDEQLKSGTA SVVCLLNNFY PREAKVQWKV DNALQSGNSQ ESVTEQDSKD STYSLSSTLT
LSKADYEKHK VYACEVTHQG LSSPVTKSFN RGEC
(Disulfide bridge: H22-H96, H147-H203, H223-L214, H229-H’229, H232-H’232, H264-H324, H370-H428, H’22-H’96, H’147-H’203, H’223-L’214, H’264-H’324, H’370-H’428, L23-L88, L134-L194, L’23-L’88, L’134-L’194)
Casirivimab
カシリビマブ;
- Immunoglobulin G1, anti-(severe acute respiratory syndrome coronavirus 2 spike glycoprotein) (human monoclonal REGN10933 γ1-chain), disulfide with human monoclonal REGN10933 κ-chain, dimer
| Formula | C6454H9976N1704O2024S44 |
|---|---|
| CAS | 2415933-42-3 |
| Mol weight | 145233.3296 |
Monoclonal antibody
Treatment and prophylaxis of SARS-CoV-2 infection (COVID-19)
SARS-CoV-2 spike glycoprotein
- Protein Sequence
- Sequence Length: 1328, 450, 450, 214, 214
- REGN 10933
- RG 6413
https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-fda-authorizes-monoclonal-antibodies-treatment-covid-19 November 21, 2020
Today, the U.S. Food and Drug Administration issued an emergency use authorization (EUA) for casirivimab and imdevimab to be administered together for the treatment of mild to moderate COVID-19 in adults and pediatric patients (12 years of age or older weighing at least 40 kilograms [about 88 pounds]) with positive results of direct SARS-CoV-2 viral testing and who are at high risk for progressing to severe COVID-19. This includes those who are 65 years of age or older or who have certain chronic medical conditions.
In a clinical trial of patients with COVID-19, casirivimab and imdevimab, administered together, were shown to reduce COVID-19-related hospitalization or emergency room visits in patients at high risk for disease progression within 28 days after treatment when compared to placebo. The safety and effectiveness of this investigational therapy for use in the treatment of COVID-19 continues to be evaluated.
Casirivimab and imdevimab must be administered together by intravenous (IV) infusion.
Casirivimab and imdevimab are not authorized for patients who are hospitalized due to COVID-19 or require oxygen therapy due to COVID-19. A benefit of casirivimab and imdevimab treatment has not been shown in patients hospitalized due to COVID-19. Monoclonal antibodies, such as casirivimab and imdevimab, may be associated with worse clinical outcomes when administered to hospitalized patients with COVID-19 requiring high flow oxygen or mechanical ventilation.
“The FDA remains committed to advancing the nation’s public health during this unprecedented pandemic. Authorizing these monoclonal antibody therapies may help outpatients avoid hospitalization and alleviate the burden on our health care system,” said FDA Commissioner Stephen M. Hahn, M.D. “As part of our Coronavirus Treatment Acceleration Program, the FDA uses every possible pathway to make new treatments available to patients as quickly as possible while continuing to study the safety and effectiveness of these treatments.”
Monoclonal antibodies are laboratory-made proteins that mimic the immune system’s ability to fight off harmful pathogens such as viruses. Casirivimab and imdevimab are monoclonal antibodies that are specifically directed against the spike protein of SARS-CoV-2, designed to block the virus’ attachment and entry into human cells.
“The emergency authorization of these monoclonal antibodies administered together offers health care providers another tool in combating the pandemic,” said Patrizia Cavazzoni, M.D., acting director of the FDA’s Center for Drug Evaluation and Research. “We will continue to facilitate the development, evaluation and availability of COVID-19 therapies.”
The issuance of an EUA is different than an FDA approval. In determining whether to issue an EUA, the FDA evaluates the totality of available scientific evidence and carefully balances any known or potential risks with any known or potential benefits of the product for use during an emergency. Based on the FDA’s review of the totality of the scientific evidence available, the agency has determined that it is reasonable to believe that casirivimab and imdevimab administered together may be effective in treating patients with mild or moderate COVID-19. When used to treat COVID-19 for the authorized population, the known and potential benefits of these antibodies outweigh the known and potential risks. There are no adequate, approved and available alternative treatments to casirivimab and imdevimab administered together for the authorized population.
The data supporting this EUA for casirivimab and imdevimab are based on a randomized, double-blind, placebo-controlled clinical trial in 799 non-hospitalized adults with mild to moderate COVID-19 symptoms. Of these patients, 266 received a single intravenous infusion of 2,400 milligrams casirivimab and imdevimab (1,200 mg of each), 267 received 8,000 mg casirivimab and imdevimab (4,000 mg of each), and 266 received a placebo, within three days of obtaining a positive SARS-CoV-2 viral test.
The prespecified primary endpoint for the trial was time-weighted average change in viral load from baseline. Viral load reduction in patients treated with casirivimab and imdevimab was larger than in patients treated with placebo at day seven. However, the most important evidence that casirivimab and imdevimab administered together may be effective came from the predefined secondary endpoint of medically attended visits related to COVID-19, particularly hospitalizations and emergency room visits within 28 days after treatment. For patients at high risk for disease progression, hospitalizations and emergency room visits occurred in 3% of casirivimab and imdevimab-treated patients on average compared to 9% in placebo-treated patients. The effects on viral load, reduction in hospitalizations and ER visits were similar in patients receiving either of the two casirivimab and imdevimab doses.
Under the EUA, fact sheets that provide important information about using casirivimab and imdevimab administered together in treating COVID-19 as authorized must be made available to health care providers and to patients and caregivers. These fact sheets include dosing instructions, potential side effects and drug interactions. Possible side effects of casirivimab and imdevimab include: anaphylaxis and infusion-related reactions, fever, chills, hives, itching and flushing.
The EUA was issued to Regeneron Pharmaceuticals Inc.
The FDA, an agency within the U.S. Department of Health and Human Services, protects the public health by assuring the safety, effectiveness, and security of human and veterinary drugs, vaccines and other biological products for human use, and medical devices. The agency also is responsible for the safety and security of our nation’s food supply, cosmetics, dietary supplements, products that give off electronic radiation, and for regulating tobacco products.
Related Information
- Casirivimab and Imdevimab EUA Letter of Authorization
- Frequently Asked Questions on the Emergency Use Authorization for Casirivimab and Imdevimab
- Emergency Use Authorization: Therapeutics
- Coronavirus Disease (COVID-19)
Casirivimab/imdevimab, sold under the brand name REGEN-COV,[1] is an experimental medicine developed by the American biotechnology company Regeneron Pharmaceuticals. It is an artificial “antibody cocktail” designed to produce resistance against the SARS-CoV-2 coronavirus responsible for the COVID-19 pandemic.[3][4] It consists of two monoclonal antibodies, casirivimab (REGN10933) and imdevimab (REGN10987) that must be mixed together.[1][5][6] The combination of two antibodies is intended to prevent mutational escape.[7]
Trials
In a clinical trial of people with COVID-19, casirivimab and imdevimab, administered together, were shown to reduce COVID-19-related hospitalization or emergency room visits in people at high risk for disease progression within 28 days after treatment when compared to placebo.[2] The safety and effectiveness of this investigational therapy for use in the treatment of COVID-19 continues to be evaluated.[2]
The data supporting the emergency use authorization (EUA) for casirivimab and imdevimab are based on a randomized, double-blind, placebo-controlled clinical trial in 799 non-hospitalized adults with mild to moderate COVID-19 symptoms.[2] Of these participants, 266 received a single intravenous infusion of 2,400 milligrams casirivimab and imdevimab (1,200 mg of each), 267 received 8,000 mg casirivimab and imdevimab (4,000 mg of each), and 266 received a placebo, within three days of obtaining a positive SARS-CoV-2 viral test.[2]
The prespecified primary endpoint for the trial was time-weighted average change in viral load from baseline.[2] Viral load reduction in participants treated with casirivimab and imdevimab was larger than in participants treated with placebo at day seven.[2] However, the most important evidence that casirivimab and imdevimab administered together may be effective came from the predefined secondary endpoint of medically attended visits related to COVID-19, particularly hospitalizations and emergency room visits within 28 days after treatment.[2] For participants at high risk for disease progression, hospitalizations and emergency room visits occurred in 3% of casirivimab and imdevimab-treated participants on average compared to 9% in placebo-treated participants.[2] The effects on viral load, reduction in hospitalizations and ER visits were similar in participants receiving either of the two casirivimab and imdevimab doses.[2]
As of September 2020, REGEN-COV is being evaluated as part of the RECOVERY Trial.[8]
On 12 April 2021, Roche and Regeneron announced that the Phase III clinical trial REGN-COV 2069 met both primary and secondary endpoints, reducing risk of infection by 81% for the non-infected patients, and reducing time-to-resolution of symptoms for symptomatic patients to one week vs. three weeks in the placebo group.[9]
Authorization
On 21 November 2020, the U.S. Food and Drug Administration (FDA) issued an emergency use authorization (EUA) for casirivimab and imdevimab to be administered together for the treatment of mild to moderate COVID-19 in people twelve years of age or older weighing at least 40 kilograms (88 lb) with positive results of direct SARS-CoV-2 viral testing and who are at high risk for progressing to severe COVID-19.[2][10][11] This includes those who are 65 years of age or older or who have certain chronic medical conditions.[2] Casirivimab and imdevimab must be administered together by intravenous (IV) infusion.[2]
Casirivimab and imdevimab are not authorized for people who are hospitalized due to COVID-19 or require oxygen therapy due to COVID-19.[2] A benefit of casirivimab and imdevimab treatment has not been shown in people hospitalized due to COVID-19.[2] Monoclonal antibodies, such as casirivimab and imdevimab, may be associated with worse clinical outcomes when administered to hospitalized people with COVID-19 requiring high flow oxygen or mechanical ventilation.[2]
The EUA was issued to Regeneron Pharmaceuticals Inc.[2][10][12]
On 1 February 2021, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) started a rolling review of data on the REGN‑COV2 antibody combination (casirivimab/imdevimab), which is being co-developed by Regeneron Pharmaceuticals, Inc. and F. Hoffman-La Roche, Ltd (Roche) for the treatment and prevention of COVID‑19.[13][14] In February 2021, the CHMP concluded that the combination, also known as REGN-COV2, can be used for the treatment of confirmed COVID-19 in people who do not require supplemental oxygen and who are at high risk of progressing to severe COVID-19.[15]
The Central Drugs Standards Control Organisation (CDSCO) in India, on 5 May 2021, granted an Emergency Use Authorisation to Roche (Genentech)[16] and Regeneron[17] for use of the casirivimab/imdevimab cocktail in the country. The announcement came in light of the second wave of the COVID-19 pandemic in India. Roche India maintains partnership with Cipla, thereby permitting the latter to market the drug in the country.[18]
Deployment
Although Regeneron is headquartered in Tarrytown, New York (near New York City), REGEN-COV is manufactured at the company’s primary U.S. manufacturing facility in Rensselaer, New York (near the state capital at Albany).[19] In September 2020, to free up manufacturing capacity for REGEN-COV, Regeneron began to shift production of its existing products from Rensselaer to the Irish city of Limerick.[20]
Regeneron has a deal in place with Roche (Genentech)[21]to manufacture and market REGEN-COV outside the United States.[10][22]
On 2 October 2020, Regeneron Pharmaceuticals announced that US President Donald Trump had received “a single 8 gram dose of REGN-COV2” after testing positive for SARS-CoV-2.[23][24] The drug was provided by the company in response to a “compassionate use” (temporary authorization for use) request from the president’s physicians.[23]
References
- ^ Jump up to:a b c “REGEN-COV- casirivimab and imdevimab kit”. DailyMed. Retrieved 18 March 2021.
- ^ Jump up to:a b c d e f g h i j k l m n o p q “Coronavirus (COVID-19) Update: FDA Authorizes Monoclonal Antibodies for Treatment of COVID-19”. U.S. Food and Drug Administration (FDA) (Press release). 21 November 2020. Retrieved 21 November 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Kelland K (14 September 2020). “Regeneron’s antibody drug added to UK Recovery trial of COVID treatments”. Reuters. Retrieved 14 September 2020.
- ^ “Regeneron’s COVID-19 Response Efforts”. Regeneron Pharmaceuticals. Retrieved 14 September 2020.
- ^ Morelle R (14 September 2020). “Antibody treatment to be given to Covid patients”. BBC News Online. Retrieved 14 September2020.
- ^ “Safety, Tolerability, and Efficacy of Anti-Spike (S) SARS-CoV-2 Monoclonal Antibodies for Hospitalized Adult Patients With COVID-19”. ClinicalTrials. 3 September 2020. Retrieved 14 September2020.
- ^ Baum A, Fulton BO, Wloga E, Copin R, Pascal KE, Russo V, et al. (August 2020). “Antibody cocktail to SARS-CoV-2 spike protein prevents rapid mutational escape seen with individual antibodies”. Science. 369 (6506): 1014–1018. Bibcode:2020Sci…369.1014B. doi:10.1126/science.abd0831. PMC 7299283. PMID 32540904.
- ^ “RECOVERY COVID-19 phase 3 trial to evaluate Regeneron’s REGN-COV2 investigational antibody cocktail in the UK”. Recovery Trial. Retrieved 14 September 2020.
- ^ “Phase III prevention trial showed subcutaneous administration of investigational antibody cocktail casirivimab and imdevimab reduced risk of symptomatic COVID-19 infections by 81%”. streetinsider.com. Archived from the original on 2021-04-12. Retrieved 2021-04-12.
- ^ Jump up to:a b c “Regeneron Reports Positive Interim Data with REGEN-COV Antibody Cocktail used as Passive Vaccine to Prevent COVID-19”(Press release). Regeneron Pharmaceuticals. 26 January 2021. Retrieved 19 March 2021 – via PR Newswire.
- ^ “Fact Sheet For Health Care Providers Emergency Use Authorization (EUA) Of Casirivimab And Imdevimab” (PDF). U.S. Food and Drug Administration (FDA).
- ^ “Casirivimab and Imdevimab”. Regeneron Pharmaceuticals. Retrieved 19 March 2021.
- ^ “EMA starts rolling review of REGN‑COV2 antibody combination (casirivimab / imdevimab)” (Press release). European Medicines Agency (EMA). 1 February 2021. Retrieved 1 February 2021. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ “EMA reviewing data on monoclonal antibody use for COVID-19” (Press release). European Medicines Agency (EMA). 4 February 2021. Retrieved 4 March 2021.
- ^ “EMA issues advice on use of REGN-COV2 antibody combination (casirivimab / imdevimab)” (Press release). European Medicines Agency (EMA). 26 February 2021. Retrieved 5 March 2021. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^https://www.businesswire.com/news/home/20200818005847/en/Genentech-and-Regeneron-Collaborate-to-Significantly-Increase-Global-Supply-of-REGN-COV2-Investigational-Antibody-Combination-for-COVID-19
- ^ https://timesofindia.indiatimes.com/india/india-approves-roche/regeneron-antibody-cocktail-to-treat-covid-19/articleshow/82407551.cms
- ^ “Roche receives Emergency Use Authorisation in India for its investigational Antibody Cocktail (Casirivimab and Imdevimab) used in the treatment of Covid-19 | Cipla”. http://www.cipla.com. Retrieved 2021-05-06.
- ^ Williams, Stephen (3 October 2020). “Experimental drug given to President made locally”. The Daily Gazette.
- ^ Stanton, Dan (11 September 2020). “Manufacturing shift to Ireland frees up US capacity for Regeneron’s COVID antibodies”. BioProcess International.
- ^https://www.businesswire.com/news/home/20200818005847/en/Genentech-and-Regeneron-Collaborate-to-Significantly-Increase-Global-Supply-of-REGN-COV2-Investigational-Antibody-Combination-for-COVID-19
- ^ “Roche and Regeneron link up on a coronavirus antibody cocktail”. CNBC. 19 August 2020. Retrieved 14 September 2020.
- ^ Jump up to:a b Thomas K (2 October 2020). “President Trump Received Experimental Antibody Treatment”. The New York Times. ISSN 0362-4331. Retrieved 2 October 2020.
- ^ Hackett DW (3 October 2020). “8-Gram Dose of COVID-19 Antibody Cocktail Provided to President Trump”. http://www.precisionvaccinations.com. Archived from the original on 3 October 2020.
External links
- “Casirivimab”. Drug Information Portal. U.S. National Library of Medicine.
- “Imdevimab”. Drug Information Portal. U.S. National Library of Medicine.
- “Casirivimab and Imdevimab EUA Letter of Authorization” (PDF). U.S. Food and Drug Administration (FDA).
- “Frequently Asked Questions on the Emergency Use Authorization of Casirivimab + Imdevimab” (PDF). U.S. Food and Drug Administration (FDA).
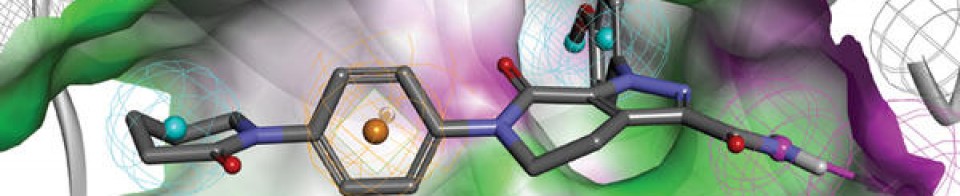
| REGN10933 (blue) and REGN10987 (orange) bound to SARS-CoV-2 spike protein (pink). From PDB: 6VSB, 6XDG. | |
| Combination of | |
|---|---|
| Casirivimab | Monoclonal antibody against spike protein of SARS-CoV-2 |
| Imdevimab | Monoclonal antibody against spike protein of SARS-CoV-2 |
| Clinical data | |
| Trade names | REGEN-COV |
| Other names | REGN-COV2 |
| AHFS/Drugs.com | Monograph |
| License data | US DailyMed: Casirivimab |
| Routes of administration | Intravenous |
| ATC code | None |
| Legal status | |
| Legal status | US: Unapproved (Emergency Use Authorization)[1][2] |
| Identifiers | |
| DrugBank | DB15691 |
| KEGG | D11938 |
//////////// Casirivimab, ANTI VIRAL, PEPTIDE, SARS-CoV-2, MONOCLONAL ANTIBODY, FDA 2020, 2020APPROVALS, CORONA VIRUS, COVID 19, カシリビマブ, REGN-COV2, REGN10933+REGN10987 combination therapy, REGN 10933, RG 6413
NEW DRUG APPROVALS
ONE TIME
$10.00

Evinacumab
(Heavy chain)
EVQLVESGGG VIQPGGSLRL SCAASGFTFD DYAMNWVRQG PGKGLEWVSA ISGDGGSTYY
ADSVKGRFTI SRDNSKNSLY LQMNSLRAED TAFFYCAKDL RNTIFGVVIP DAFDIWGQGT
MVTVSSASTK GPSVFPLAPC SRSTSESTAA LGCLVKDYFP EPVTVSWNSG ALTSGVHTFP
AVLQSSGLYS LSSVVTVPSS SLGTKTYTCN VDHKPSNTKV DKRVESKYGP PCPPCPAPEF
LGGPSVFLFP PKPKDTLMIS RTPEVTCVVV DVSQEDPEVQ FNWYVDGVEV HNAKTKPREE
QFNSTYRVVS VLTVLHQDWL NGKEYKCKVS NKGLPSSIEK TISKAKGQPR EPQVYTLPPS
QEEMTKNQVS LTCLVKGFYP SDIAVEWESN GQPENNYKTT PPVLDSDGSF FLYSRLTVDK
SRWQEGNVFS CSVMHEALHN HYTQKSLSLS LGK
(Light chain)
DIQMTQSPST LSASVGDRVT ITCRASQSIR SWLAWYQQKP GKAPKLLIYK ASSLESGVPS
RFSGSGSGTE FTLTISSLQP DDFATYYCQQ YNSYSYTFGQ GTKLEIKRTV AAPSVFIFPP
SDEQLKSGTA SVVCLLNNFY PREAKVQWKV DNALQSGNSQ ESVTEQDSKD STYSLSSTLT
LSKADYEKHK VYACEVTHQG LSSPVTKSFN RGEC
(Disulfide bridge: H22-H96, H140-L214, H153-H209, H232-H’232, H235-H’235, H267-H327, H373-H431, H’22-H’96, H’140-L’214, H’153-H’209, H’267-H’327, H’373-H’431, L23-L88, L134-L194, L’23-L’88, L’134-L’194)
Evinacumab
エビナクマブ (遺伝子組換え)
Immunoglobulin G4, anti-(human protein ANGPTL3 (angiopoietin-like 3)) (human monoclonal REGN1500 heavy chain), disulfide with human monoclonal REGN1500 light chain, dimer
| Formula | C6480H9992N1716O2042S46 |
|---|---|
| CAS | 1446419-85-7 |
| Mol weight | 146081.9345 |
Protein Sequence
Sequence Length: 1334, 453, 453, 214, 214multichain; modified (modifications unspecified)
FDA APPROVED, 2021/2/11, EVKEEZA
Antihyperlipidemic, Anti-angiopietin like 3
Monoclonal antibody
Treatment of dyslipidemia
- REGN 1500
- REGN-1500
- REGN1500
Sequence:
1EVQLVESGGG VIQPGGSLRL SCAASGFTFD DYAMNWVRQG PGKGLEWVSA51ISGDGGSTYY ADSVKGRFTI SRDNSKNSLY LQMNSLRAED TAFFYCAKDL101RNTIFGVVIP DAFDIWGQGT MVTVSSASTK GPSVFPLAPC SRSTSESTAA151LGCLVKDYFP EPVTVSWNSG ALTSGVHTFP AVLQSSGLYS LSSVVTVPSS201SLGTKTYTCN VDHKPSNTKV DKRVESKYGP PCPPCPAPEF LGGPSVFLFP251PKPKDTLMIS RTPEVTCVVV DVSQEDPEVQ FNWYVDGVEV HNAKTKPREE301QFNSTYRVVS VLTVLHQDWL NGKEYKCKVS NKGLPSSIEK TISKAKGQPR351EPQVYTLPPS QEEMTKNQVS LTCLVKGFYP SDIAVEWESN GQPENNYKTT401PPVLDSDGSF FLYSRLTVDK SRWQEGNVFS CSVMHEALHN HYTQKSLSLS451LGK
Sequence:
1EVQLVESGGG VIQPGGSLRL SCAASGFTFD DYAMNWVRQG PGKGLEWVSA51ISGDGGSTYY ADSVKGRFTI SRDNSKNSLY LQMNSLRAED TAFFYCAKDL101RNTIFGVVIP DAFDIWGQGT MVTVSSASTK GPSVFPLAPC SRSTSESTAA151LGCLVKDYFP EPVTVSWNSG ALTSGVHTFP AVLQSSGLYS LSSVVTVPSS201SLGTKTYTCN VDHKPSNTKV DKRVESKYGP PCPPCPAPEF LGGPSVFLFP251PKPKDTLMIS RTPEVTCVVV DVSQEDPEVQ FNWYVDGVEV HNAKTKPREE301QFNSTYRVVS VLTVLHQDWL NGKEYKCKVS NKGLPSSIEK TISKAKGQPR351EPQVYTLPPS QEEMTKNQVS LTCLVKGFYP SDIAVEWESN GQPENNYKTT401PPVLDSDGSF FLYSRLTVDK SRWQEGNVFS CSVMHEALHN HYTQKSLSLS451LGK
Sequence:
1DIQMTQSPST LSASVGDRVT ITCRASQSIR SWLAWYQQKP GKAPKLLIYK51ASSLESGVPS RFSGSGSGTE FTLTISSLQP DDFATYYCQQ YNSYSYTFGQ101GTKLEIKRTV AAPSVFIFPP SDEQLKSGTA SVVCLLNNFY PREAKVQWKV151DNALQSGNSQ ESVTEQDSKD STYSLSSTLT LSKADYEKHK VYACEVTHQG201LSSPVTKSFN RGEC
Sequence:
1DIQMTQSPST LSASVGDRVT ITCRASQSIR SWLAWYQQKP GKAPKLLIYK51ASSLESGVPS RFSGSGSGTE FTLTISSLQP DDFATYYCQQ YNSYSYTFGQ101GTKLEIKRTV AAPSVFIFPP SDEQLKSGTA SVVCLLNNFY PREAKVQWKV151DNALQSGNSQ ESVTEQDSKD STYSLSSTLT LSKADYEKHK VYACEVTHQG201LSSPVTKSFN RGEC
Sequence Modifications
| Type | Location | Description |
|---|---|---|
| bridge | Cys-22 – Cys-96 | disulfide bridge |
| bridge | Cys-140 – Cys-214” | disulfide bridge |
| bridge | Cys-153 – Cys-209 | disulfide bridge |
| bridge | Cys-232 – Cys-232′ | disulfide bridge |
| bridge | Cys-235 – Cys-235′ | disulfide bridge |
| bridge | Cys-267 – Cys-327 | disulfide bridge |
| bridge | Cys-373 – Cys-431 | disulfide bridge |
| bridge | Cys-22′ – Cys-96′ | disulfide bridge |
| bridge | Cys-140′ – Cys-214”’ | disulfide bridge |
| bridge | Cys-153′ – Cys-209′ | disulfide bridge |
| bridge | Cys-267′ – Cys-327′ | disulfide bridge |
| bridge | Cys-373′ – Cys-431′ | disulfide bridge |
| bridge | Cys-23” – Cys-88” | disulfide bridge |
| bridge | Cys-134” – Cys-194” | disulfide bridge |
| bridge | Cys-23”’ – Cys-88”’ | disulfide bridge |
| bridge | Cys-134”’ – Cys-194”’ | disulfide bridge |
PATENTS
WO 2017024062
US 20170305999
Evinacumab, sold under the brand name Evkeeza, is a monoclonal antibody medication for the treatment of homozygous familial hypercholesterolemia (HoFH).[1][2]
Evinacumab is a recombinant human IgG4 monoclonal antibody targeted against angiopoietin-like protein 3 (ANGPTL3) and the first drug of its kind. The ANGPTL family of proteins serve a number of physiologic functions – including involvement in the regulation of lipid metabolism – which have made them desirable therapeutic targets in recent years.2 Loss-of-function mutations in ANGPTL3 have been noted to result in hypolipidemia and subsequent reductions in cardiovascular risk, whereas increases in function appear to be associated with cardiovascular risk, and it was these observations that provided a rationale for the development of a therapy targeted against ANGPTL3.3
In February 2021, evinacumab became the first-and-only inhibitor of ANGPTL3 to receive FDA approval after it was granted approval for the adjunctive treatment of homozygous familial hypercholesterolemia (HoFH) under the brand name “Evkeeza”.8 Evinacumab is novel in its mechanism of action compared with other lipid-lowering therapies and therefore provides a unique and synergistic therapeutic option in the treatment of HoFH.
Common side effects include nasopharyngitis (cold), influenza-like illness, dizziness, rhinorrhea (runny nose), and nausea. Serious hypersensitivity (allergic) reactions have occurred in the Evkeeza clinical trials.[2]
Evinacumab binds to the angiopoietin-like protein 3 (ANGPTL3).[2] ANGPTL3 slows the function of certain enzymes that break down fats in the body.[2] Evinacumab blocks ANGPTL3, allowing faster break down of fats that lead to high cholesterol.[2] Evinacumab was approved for medical use in the United States in February 2021.[2][3]
| NAME | DOSAGE | STRENGTH | ROUTE | LABELLER | MARKETING START | MARKETING END | ||
|---|---|---|---|---|---|---|---|---|
| Evkeeza | Injection, solution, concentrate | 150 mg/1mL | Intravenous | Regeneron Pharmaceuticals, Inc. | 2021-02-11 | Not applicable | ||
| Evkeeza | Injection, solution, concentrate | 150 mg/1mL | Intravenous | Regeneron Pharmaceuticals, Inc. | 2021-02-11 | Not applicable |

History
The effectiveness and safety of evinacumab were evaluated in a double-blind, randomized, placebo-controlled, 24-week trial enrolling 65 participants with homozygous familial hypercholesterolemia (HoFH).[2] In the trial, 43 participants received 15 mg/kg of evinacumab every four weeks and 22 participants received the placebo.[2] Participants were taking other lipid-lowering therapies as well.[2]
The primary measure of effectiveness was the percent change in low-density lipoprotein (LDL-C) from the beginning of treatment to week 24.[2] At week 24, participants receiving evinacumab had an average 47% decrease in LDL-C while participants on the placebo had an average 2% increase.[2]
The U.S. Food and Drug Administration (FDA) granted the application for evinacumab orphan drug, breakthrough therapy, and priority review designations.[2] The FDA granted approval of Evkeeza to Regeneron Pharmaceuticals, Inc.[2]
References
- ^ Jump up to:a b https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/761181s000lbl.pdf
- ^ Jump up to:a b c d e f g h i j k l m n “FDA approves add-on therapy for patients with genetic form of severely”. U.S. Food and Drug Administration (FDA). 11 February 2021. Retrieved 12 February 2021.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ “FDA Approves First-in-class Evkeeza (evinacumab-dgnb) for Patients with Ultra-rare Inherited Form of High Cholesterol” (Press release). Regeneron Pharmaceuticals. 11 February 2021. Retrieved 12 February 2021 – via PR Newswire.
Further reading
- Dewey FE, Gusarova V, Dunbar RL, O’Dushlaine C, Schurmann C, Gottesman O, et al. (July 2017). “Genetic and Pharmacologic Inactivation of ANGPTL3 and Cardiovascular Disease”. N Engl J Med. 377 (3): 211–221. doi:10.1056/NEJMoa1612790. PMC 5800308. PMID 28538136.
External links
- “Evinacumab”. Drug Information Portal. U.S. National Library of Medicine.
| Monoclonal antibody | |
|---|---|
| Type | Whole antibody |
| Source | Human |
| Target | Angiopoietin-like 3 (ANGPTL3) |
| Clinical data | |
| Trade names | Evkeeza |
| Other names | REGN1500, evinacumab-dgnb |
| License data | US DailyMed: Evinacumab |
| Routes of administration | Intravenous |
| ATC code | None |
| Legal status | |
| Legal status | US: ℞-only [1][2] |
| Identifiers | |
| CAS Number | 1446419-85-7 |
| DrugBank | DB15354 |
| ChemSpider | none |
| UNII | T8B2ORP1DW |
| KEGG | D11753 |
| Chemical and physical data | |
| Formula | C6480H9992N1716O2042S46 |
| Molar mass | 146083.95 g·mol−1 |
//////////////
#Evinacumab, #Peptide, #APPROVALS 2021, #FDA 2021, #Monoclonal antibody, #dyslipidemia, #エビナクマブ (遺伝子組換え) , #REGN 1500, #REGN-1500, #REGN1500, #Anthony melvin crasto, #world drug tracker. # new drug approvals, #pharma
Fenfluramine Hydrochloride

Fenfluramine
- DEA No. 1670
- S 768
2020/12/18, FDA APPROVED, Fintepla
Fenfluramine hydrochloride
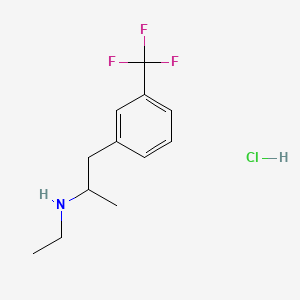
| Formula | C12H16F3N. HCl |
|---|---|
| CAS | 404-82-0458-24-2 (FREE) |
| Mol weight | 267.7183 |
Antiobesity
| Efficacy | Appetite suppressant |
|---|---|
| Disease | Dravet syndrome |
(+-)-Fenfluramine hydrochloride
Racemic fenfluramine hydrochloride
Fenfluramine hydrochloride [USAN]
1-(m-Trifluoromethylphenyl)-2-(ethylamino)propane hydrochloride
Research Code:ZX-008
MOA:Serotonin agonist
Indication:Dravet syndrome
Company:Zogenix (Originator)
Synonyms of Fenfluramine [INN:BAN]
- (+-)-Fenfluramine
- BRN 4783711
- dl-Fenfluramine
- DL-Fenfluramine
- EINECS 207-276-3
- Fenfluramina
- Fenfluramina [DCIT]
- Fenfluramine
- Fenfluraminum
- Fenfluraminum [INN-Latin]
- HSDB 3080
- Obedrex
- Pesos
- Ponderax PA
- Rotondin
- S 768
- UNII-2DS058H2CF
mp 160-161 °C, ethyl acetate US 3198834
nmr Salsbury, Jonathon S.; Magnetic Resonance in Chemistry 2005, V43(11), P910-917 C
IR BIORAD: Infrared spectral data from the Bio-Rad/Sadtler IR Data Collection was obtained from Bio-Rad Laboratories, Philadelphia, PA (US).FenfluramineCAS Registry Number: 458-24-2CAS Name:N-Ethyl-a-methyl-3-(trifluoromethyl)benzeneethanamineAdditional Names:N-ethyl-a-methyl-m-(trifluoromethyl)phenethylamine; 2-ethylamino-1-(3-trifluoromethylphenyl)propaneManufacturers’ Codes: S-768Molecular Formula: C12H16F3NMolecular Weight: 231.26Percent Composition: C 62.32%, H 6.97%, F 24.65%, N 6.06%Literature References: Prepn: L. G. Beregi et al.,FRM1658; eidem,US3198833 (1963, 1965 both to Sci. Union et Cie Soc. Franc. Recherche Méd.). Prepn of optical isomers: eidem,US3198834 (1965 to Sci. Union et Cie Soc. Franc. Recherche Med.). Pharmacology: Presse Med.71, 181 (1963). Pharmacology and toxicity of isomers and racemate: J. C. Le Douarec et al.,Arch. Int. Pharmacodyn. Ther.161, 206 (1966). Pharmacokinetics: S. Caccia et al.,Eur. J. Clin. Pharmacol.29, 221 (1985). Clinical trial of dextrofenfluramine in refractory obesity: N. Finer et al.,Curr. Ther. Res.38, 847 (1985). Comprehensive review: Pinder et al.,Drugs10, 241-323 (1975).Properties: bp12 108-112°. LD50 i.p. in mice: 144 mg/kg (US3198833).Boiling point: bp12 108-112°Toxicity data: LD50 i.p. in mice: 144 mg/kg
Derivative Type: HydrochlorideCAS Registry Number: 404-82-0Trademarks: Acino (IMA); Adipomin (Streuli); Obedrex (Beta); Pesos (Valeas); Ponderal (Servier); Ponderax (Selpharm); Ponderex (Robins); Pondimin (Robins); Rotondin (Casasco)Molecular Formula: C12H16F3N.HClMolecular Weight: 267.72Percent Composition: C 53.84%, H 6.40%, F 21.29%, N 5.23%, Cl 13.24%Properties: Crystals from ethanol + ether, mp 166°.Melting point: mp 166°
Derivative Type:d-FormCAS Registry Number: 3239-44-9Additional Names: Dexfenfluramine; dextrofenfluramineProperties: [a]D25 +9.5° (c = 8 in ethanol). LD50 orally in rats: 114.6 mg/kg (Le Douarec).Optical Rotation: [a]D25 +9.5° (c = 8 in ethanol)Toxicity data: LD50 orally in rats: 114.6 mg/kg (Le Douarec)
Derivative Type:d-Form hydrochlorideCAS Registry Number: 3239-45-0Trademarks: Adifax (Servier); Glypolix (Stroder); Isomeride (Ardix); Redux (Wyeth-Ayerst)Properties: Crystals from ethyl acetate, mp 160-161°.Melting point: mp 160-161°
Derivative Type:l-FormCAS Registry Number: 37577-24-5Properties: [a]D25 -9.6° (c = 8 in ethanol). LD50 orally in rats: 195 mg/kg (Le Douarec).Optical Rotation: [a]D25 -9.6° (c = 8 in ethanol)Toxicity data: LD50 orally in rats: 195 mg/kg (Le Douarec)
Derivative Type:l-Form hydrochlorideCAS Registry Number: 3616-78-2Properties: Crystals from ethyl acetate, mp 160-161°.Melting point: mp 160-161°
NOTE: This is a controlled substance: 21 CFR, 1308.14.Therap-Cat: Anorexic.Keywords: Anorexic.
A centrally active drug that apparently both blocks serotonin uptake and provokes transport-mediated serotonin release.
Fenfluramine Hydrochloride has been filed an IND application with the FDA in USA to initiate phase III trials by Brabant Pharma (acquired by Zogenix in 2014) for the treatment of dravets syndrome (also known as severe myoclonic epilepsy of infancy, SMEI), this compound has been granted orphan drug designation in Europe and U.S..
Fenfluramine Hydrochloride was launched in 1963 by Servier in France and in 1973 by Wyeth (now a wholly owned subsidiary of Pfizer) in US for the treatment of obesity. However, it was withdrawn from the market in 1997 due to heart disease.
Dravet syndrome is a pediatric encephalopathy that typically manifests within the first year of life following exposure to elevated temperatures. It is characterized by recurrent pharmacoresistant seizures, which increase in frequency and severity with disease progression. Concomitantly with these seizures, patients typically display delayed development and neurocognitive impairment.6,9,10,11 Fenfluramine is a serotonergic phenethylamine originally used as an appetite suppressant until concerns regarding cardiotoxicity in obese patients lead to its withdrawal from the market in 1997.6,12,13 Through its ability to modulate neurotransmission, fenfluramine has reemerged as an effective therapy against pharmacoresistant seizures, such as those involved in Dravet syndrome.3,5,8
Fenfluramine was granted initial FDA approval in 1973 prior to its withdrawal; it was granted a new FDA approval on June 25, 2020, for treatment of Dravet syndrome patients through the restricted FINTEPLA REMS program. It is currently sold under the name FINTEPLA® by Zogenix INC.16
Fenfluramine, sold under the brand name Fintepla, is a medication used for the treatment of seizures associated with Dravet syndrome in people age two and older.[2][3]
The most common adverse reactions include decreased appetite; drowsiness, sedation and lethargy; diarrhea; constipation; abnormal echocardiogram; fatigue or lack of energy; ataxia (lack of coordination), balance disorder, gait disturbance (trouble with walking); increased blood pressure; drooling, salivary hypersecretion (saliva overproduction); pyrexia (fever); upper respiratory tract infection; vomiting; decreased weight; risk of falls; and status epilepticus.[2]
Dravet syndrome is a pediatric encephalopathy that typically manifests within the first year of life following exposure to elevated temperatures. It is characterized by recurrent pharmacoresistant seizures, which increase in frequency and severity with disease progression. Concomitantly with these seizures, patients typically display delayed development and neurocognitive impairment.6,9,10,11 Fenfluramine is a serotonergic phenethylamine originally used as an appetite suppressant until concerns regarding cardiotoxicity in obese patients lead to its withdrawal from the market in 1997.6,12,13 Through its ability to modulate neurotransmission, fenfluramine has reemerged as an effective therapy against pharmacoresistant seizures, such as those involved in Dravet syndrome.3,5,8
Fenfluramine was granted initial FDA approval in 1973 prior to its withdrawal; it was granted a new FDA approval on June 25, 2020, for treatment of Dravet syndrome patients through the restricted FINTEPLA REMS program. It is currently sold under the name FINTEPLA® by Zogenix INC.16
Medical uses
Fenfluramine is indicated for the treatment of seizures associated with Dravet syndrome in people age two and older.[2][3]
Dravet syndrome is a life-threatening, rare and chronic form of epilepsy.[2] It is often characterized by severe and unrelenting seizures despite medical treatment.[2]
Adverse effects
The U.S. Food and Drug Administration (FDA) fenfluramine labeling includes a boxed warning stating the drug is associated with valvular heart disease (VHD) and pulmonary arterial hypertension (PAH).[2] Because of the risks of VHD and PAH, fenfluramine is available only through a restricted drug distribution program, under a risk evaluation and mitigation strategy (REMS).[2] The fenfluramine REMS requires health care professionals who prescribe fenfluramine and pharmacies that dispense fenfluramine to be specially certified in the fenfluramine REMS and that patients be enrolled in the REMS.[2] As part of the REMS requirements, prescribers and patients must adhere to the required cardiac monitoring with echocardiograms to receive fenfluramine.[2]
At higher therapeutic doses, headache, diarrhea, dizziness, dry mouth, erectile dysfunction, anxiety, insomnia, irritability, lethargy, and CNS stimulation have been reported with fenfluramine.[4]
There have been reports associating chronic fenfluramine treatment with emotional instability, cognitive deficits, depression, psychosis, exacerbation of pre-existing psychosis (schizophrenia), and sleep disturbances.[4][5] It has been suggested that some of these effects may be mediated by serotonergic neurotoxicity/depletion of serotonin with chronic administration and/or activation of serotonin 5-HT2A receptors.[5][6][7][8]
Heart valve disease
The distinctive valvular abnormality seen with fenfluramine is a thickening of the leaflet and chordae tendineae. One mechanism used to explain this phenomenon involves heart valve serotonin receptors, which are thought to help regulate growth. Since fenfluramine and its active metabolite norfenfluramine stimulate serotonin receptors, this may have led to the valvular abnormalities found in patients using fenfluramine. In particular norfenfluramine is a potent inhibitor of the re-uptake of 5-HT into nerve terminals.[9] Fenfluramine and its active metabolite norfenfluramine affect the 5-HT2B receptors, which are plentiful in human cardiac valves. The suggested mechanism by which fenfluramine causes damage is through over or inappropriate stimulation of these receptors leading to inappropriate valve cell division. Supporting this idea is the fact that this valve abnormality has also occurred in patients using other drugs that act on 5-HT2B receptors.[10][11]
According to a study of 5,743 former users conducted by a plaintiff’s expert cardiologist, damage to the heart valve continued long after stopping the medication.[12] Of the users tested, 20% of women, and 12% of men were affected. For all ex-users, there was a 7-fold increase of chances of needing surgery for faulty heart valves caused by the drug.[12]
Overdose
In overdose, fenfluramine can cause serotonin syndrome and rapidly result in death.[13][14]
Pharmacology
Pharmacodynamics
Fenfluramine acts primarily as a serotonin releasing agent.[15][16] It increases the level of serotonin, a neurotransmitter that regulates mood, appetite and other functions.[15][16] Fenfluramine causes the release of serotonin by disrupting vesicular storage of the neurotransmitter, and reversing serotonin transporter function.[17] The drug also acts as a norepinephrine releasing agent to a lesser extent, particularly via its active metabolite norfenfluramine.[15][16] At high concentrations, norfenfluramine, though not fenfluramine, also acts as a dopamine releasing agent, and so fenfluramine may do this at very high doses as well.[15][16] In addition to monoamine release, while fenfluramine binds only very weakly to the serotonin 5-HT2 receptors, norfenfluramine binds to and activates the serotonin 5-HT2B and 5-HT2C receptors with high affinity and the serotonin 5-HT2A receptor with moderate affinity.[18][19] The result of the increased serotonergic and noradrenergic neurotransmission is a feeling of fullness and reduced appetite.
The combination of fenfluramine with phentermine, a norepinephrine–dopamine releasing agent acting primarily on norepinephrine, results in a well-balanced serotonin–norepinephrine releasing agent with weaker effects of dopamine release.[15][16]
| Drug | NE | DA | 5-HT | Type | Ref |
|---|---|---|---|---|---|
| Fenfluramine | 739 | >10,000 | 79.3–108 | SRA | [20][15][16] |
| D-Fenfluramine | 302 | >10,000 | 51.7 | SNRA | [20][15] |
| L-Fenfluramine | >10,000 | >10,000 | 147 | SRA | [15][21] |
| Norfenfluramine | 168–170 | 1,900–1,925 | 104 | SNRA | [15][16] |
| Phentermine | 39.4 | 262 | 3,511 | NDRA | [20] |
Pharmacokinetics
The elimination half-life of fenfluramine has been reported as ranging from 13 to 30 hours.[4] The mean elimination half-lives of its enantiomers have been found to be 19 hours for dexfenfluramine and 25 hours for levfenfluramine.[13] Norfenfluramine, the major active metabolite of fenfluramine, has an elimination half-life that is about 1.5 to 2 times as long as that of fenfluramine, with mean values of 34 hours for dexnorfenfluramine and 50 hours for levnorfenfluramine.[13]
Chemistry
Fenfluramine is a substituted amphetamine and is also known as 3-trifluoromethyl-N-ethylamphetamine.[13] It is a racemic mixture of two enantiomers, dexfenfluramine and levofenfluramine.[13] Some analogues of fenfluramine include norfenfluramine, benfluorex, flucetorex, and fludorex.
History
Fenfluramine was developed in the early 1960s and was introduced in France in 1963.[13] Approximately 50 million Europeans were treated with fenfluramine for appetite suppression between 1963 and 1996.[13] Fenfluramine was approved in the United States in 1973.[13] The combination of fenfluramine and phentermine was proposed in 1984.[13] Approximately 5 million people in the United States were given fenfluramine or dexfenfluramine with or without phentermine between 1996 and 1998.[13]
In the early 1990s, French researchers reported an association of fenfluramine with primary pulmonary hypertension and dyspnea in a small sample of patients.[13] Fenfluramine was withdrawn from the U.S. market in 1997 after reports of heart valve disease[22][23] and continued findings of pulmonary hypertension, including a condition known as cardiac fibrosis.[24] It was subsequently withdrawn from other markets around the world. It was banned in India in 1998.[25]
Fenfluramine was an appetite suppressant which was used to treat obesity.[13] It was used both on its own and, in combination with phentermine, as part of the anti-obesity medication Fen-Phen.[13]
In June 2020, fenfluramine was approved for medical use in the United States with an indication to treat Dravet syndrome.[2][26]
The effectiveness of fenfluramine for the treatment of seizures associated with Dravet syndrome was demonstrated in two clinical studies in 202 subjects between ages two and eighteen.[2] The studies measured the change from baseline in the frequency of convulsive seizures.[2] In both studies, subjects treated with fenfluramine had significantly greater reductions in the frequency of convulsive seizures during the trials than subjects who received placebo (inactive treatment).[2] These reductions were seen within 3–4 weeks, and remained generally consistent over the 14- to 15-week treatment periods.[2]
The U.S. Food and Drug Administration (FDA) granted the application for fenfluramine priority review and orphan drug designations.[2][27][28] The FDA granted approval of Fintepla to Zogenix, Inc.[2]
On 15 October 2020, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) adopted a positive opinion, recommending the granting of a marketing authorization for the medicinal product Fintepla, intended for the treatment of seizures associated with Dravet syndrome.[29] Fenfluramine was approved for medical use in the European Union in December 2020.[3]
Society and culture
Recreational use
Unlike various other amphetamine derivatives, fenfluramine is reported to be dysphoric, “unpleasantly lethargic“, and non-addictive at therapeutic doses.[30] However, it has been reported to be used recreationally at high doses ranging between 80 and 400 mg, which have been described as producing euphoria, amphetamine-like effects, sedation, and hallucinogenic effects, along with anxiety, nausea, diarrhea, and sometimes panic attacks, as well as depressive symptoms once the drug had worn off.[30][31][32] At very high doses (e.g., 240 mg, or between 200–600 mg), fenfluramine induces a psychedelic state resembling that produced by lysergic acid diethylamide (LSD).[32][33] Indirect (via induction of serotonin release) and/or direct activation of the 5-HT2A receptor would be expected to be responsible for the psychedelic effects of the drug at sufficient doses.
Research
Under the development code ZX008, the pharmaceutical company Zogenix is studying fenfluramine’s potential to treat seizures.[34] Clinical trials have studied the use of fenfluramine in patients with Dravet syndrome.[35] Results of a Phase III clinical trial showed a 64% reduction in seizures.[36]Route 1
Reference:1. J. Org. Chem.1979, 44, 3580-3583.Route 2
Reference:1. EP0810195A1.
2. Chem. Ind. Times 2002, 16, 33-34.Route 3
Reference:1. ACS Symp. Ser. 2009, 1003, 165-181.
ref
BE 609630
FR 1658 M 19630218
US 3198834
DE 1593595
US 3769319
NL 7215548
Ger. (East) (1974), DD 108971
EP 3170807
SYN
US20170174613
PATENT
US 20170174613
| Step 4.2: Crystallization of Fenfluramine Hydrochloride |
| Procedure: Charge Fenfluramine.HCl (crude) (1.00 wt, 1.0 eq.) and TBME (10.0 vol, 7.4 wt) to the vessel and commence stirring. Heat the suspension to reflux (50 to 58° C.). Charge ethanol (5.0 vol, 3.9 wt) maintaining the temperature at 50 to 58° C. Addition time 20 minutes. Stir at 50 to 58° C. for 5 to 10 minutes and check for dissolution. Stir the solution at 50 to 58° C. for 5 to 10 minutes, targeting 54 to 58° C. Clarify the reaction mixture through a 0.1 μm in-line filter at 54 to 58° C., followed by a line rinse with TBME (1 vol, 0.7 wt). Cool the solution to 48 to 50° C. Charge Fenfluramine.HCl, code FP0188 (0.01 wt). Check for crystallization. Allow the suspension to cool to 15 to 20° C., target 17° C. over 5 to 5.5 hours at an approximately constant rate. Stir the mixture at 15 to 20° C., target 17° C. for 2 to 3 hours. Filter the mixture and wash the filter-cake with clarified TBME (2×3.0 vol, 2×2.2 wt) at 5 to 15° C. Dry the solid at up to 40° C. until the TBME content is <0.5% w/w TBME and the ethanol content is <0.5% w/w EtOH by 1H-NMR analysis. 4 to 8 hours. Determine the w/w assay of the isolated Fenfluramine.HCl by 1H-NMR analysis. |
| Yields and Profiles: The yield for the stage 4 Demonstration batch is summarized in Table 1E below. Input: 750.0 g uncorr. Fenfluramine.HCl crude (1.00 eq, 1.00 wt uncorr.) for input calculation. FIG. 3 shows an exemplary HPLC chromatogram of a crystallized fenfluramine hydrochloride sample (210 nm UV absorbance). |
PATENT
US 20180208543
| Fenfluramine, i.e., 3-trifluoromethyl-N-ethylamphetamine, has the following chemical structure: |
| The marketing of fenfluramine as a pharmaceutical active ingredient in the United States began in 1973 and was used in a therapy in combination with phentermine to prevent and treat obesity. Anyway, in 1997 fenfluramine was withdrawn from the market in the United States and immediately thereafter in other countries, since its use was associated with the onset of cardiac fibrosis and pulmonary hypertension. As a consequence of this event, the pharmaceutical compounds containing this active ingredient were withdrawn from the market. However, fenfluramine, even after its exit from the market, has continued to attract scientific interest, as will become apparent from the discussion presented hereinafter. |
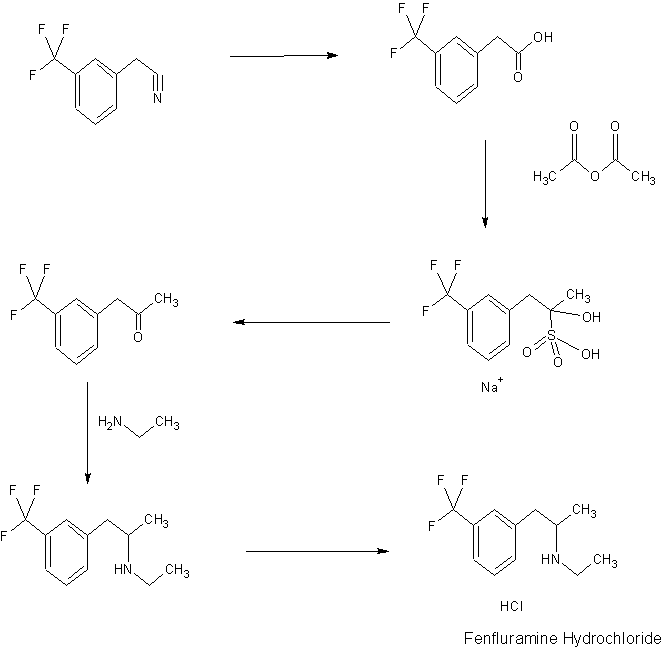
| In the literature, over the years, numerous syntheses or processes have been reported for preparing fenfluramine or its dextrorotatory enantiomer dexfenfluramine or an analog containing a highly electron-attractor group on the aromatic ring as in the fenfluramine molecule (see for example Pentafluorosulfanyl Serotonin Analogs: Synthesis, Characterization, and Biological Activity, John T. Welch and Dongsung Lim Chapter 8, pp 165-181 DOI: 10.1021/bk-2009-1003.ch008). Many of these synthesis paths are long and provide for multiple synthesis steps that can include reagents that are dangerous or scarcely environment-friendly and are therefore scarcely convenient for an industrial synthesis. Hereinafter, any reference to “fenfluramine” is understood to reference the racemic form, i.e., (RS)-N-ethyl-1-[3-(trifluoromethyephenyl]propan-2-amine. |
| To the best of the knowledge of the inventors, the first method for fenfluramine synthesis reported in the literature dates back to 1962 and is referenced in patent BE609630 and in similar patents U.S. Pat. No. 3,198,833 and FR1324220. All the synthesis methods reported in these patents provide for numerous synthesis steps. By way of example, one of the methods provides for the transformation into oxime of a ketone, 1-(3-trifluoromethyephenyl-propan-2-one, as shown here: |
| The oxime is then hydrogenated in the presence of Raney nickel catalyst so as to yield the corresponding primary amine, which is acetylated subsequently with ethanoic anhydride before being converted into fenfluramine by reduction with lithium aluminum hydride. |
| As can be seen, the final step of this chemical process provides for the use of lithium aluminum hydride and the persons skilled in the art will acknowledge that the use of this reagent should be avoided, if possible, on an industrial level, since it is extremely flammable and is the source of accidents. Furthermore, lithium is a potentially neurotoxic metal and therefore its use should be avoided where possible. Furthermore, the Raney nickel catalyst is used in the oxime reduction step and can contaminate the final active ingredient; the use of hydroxylamine also entails problems of toxicity for workers assigned to production. |
| A further disadvantage of this process is, as already mentioned earlier, the number of steps, not only because a large number of synthesis steps entails a reduction of the overall yield of active ingredient, but also because each synthesis step in principle can generate impurities and a larger number of steps can therefore entail a higher number of impurities in the final active ingredient. Many of these impurities, furthermore, due to their structural similarity to fenfluramine, are difficult to eliminate and remove from a fenfluramine preparation. One impurity for example that can be formed in the process described above and is difficult to eliminate is the following: |
| This impurity, which is a primary amine, shares physical-chemical properties that are similar to fenfluramine and therefore, like fenfluramine, it can form a hydrochloride salt by treatment with hydrochloric acid and thus contaminate the active ingredient fenfluramine hydrochloride. Furthermore, this impurity—as a free base—has a boiling point that is similar to that of fenfluramine (73° C. vs. 89° C. at 6 mmHg respectively), and therefore its elimination by distillation also can be problematic. |
| The process described above can in principle generate other impurities, which are listed in FIG. 1. |
| EP 0441160 claims a synthesis in 5 steps of dexfenfluramine, dextrorotatory enantiomer of fenfluramine. This synthesis can be adapted easily to produce fenfluramine instead of its dextrorotatory enantiomer simply by performing the first reduction step with a non-chiral reducing agent. In the first step, in fact: |
(MOL) (CDX) a ketone, 1-(3-trifluoromethyl)phenyl-propan-2-one, is first reduced to the corresponding alcohol in the presence of yeast, D-glucose, ethanol and water. Then the alcohol is converted into the tosylate in the second step:
(MOL) (CDX)
| This reaction occurs in the presence of triethylamine and tosyl chloride in methylene chloride as solvent. After purification, the tosylate is converted to fenfluramine by means of three successive steps: |
| In the first of these three steps, the tosylate is converted into an azide intermediate by reaction with sodium azide in dimethylformamide. The azide intermediate is then hydrogenated in the presence of a catalyst, palladium on carbon. Finally, the resulting primary amine is converted into fenfluramine by reaction with acetaldehyde and sodium borohydride. |
| Persons skilled in the art may see easily that this process is not desirable from an industrial standpoint due to reasons related to environmental risk, safety and costs. For example, the sodium azide used in the process is a notoriously explosive compound and its use at the industrial level is dangerous. Furthermore, palladium is an expensive material and its use in the process entails an increase in the production costs of fenfluramine. Furthermore, palladium can contaminate the finished active ingredient. |
| In another method for the synthesis of dexfenfluramine in 3-4 steps, reported by Goument et al. in Bulletin of the Chemical Society of France (1993), 130, p. 450-458, 3-bromobenzotrifluoride is subjected to a Grignard reaction with enantiopure 1,2-propylene-epoxide to yield 1-[3-(trifluoromethyl)phenyl]propan-2-ol as shown hereafter: |
| If this reaction is performed with racemic 1,2-propylene-epoxide, the synthesis can be adapted to the preparation of fenfluramine. |
| The alcohol thus obtained is first transformed into trifluoromethyl sulfonate by reaction with trifluoromethanesulfonic anhydride and then treated with ethylamine to yield fenfluramine, as shown in the diagram hereinafter: |
| In this article, the authors acknowledge that the main byproducts of the reaction are isomer alkenes having the following chemical structures: |
| The process proposed by Goument et al. is not interesting from the industrial standpoint for a series of reasons. First of all, it is known that the use of Grignard reagents, especially on an industrial scale, is problematic, because these compounds are often pyrophoric and corrosive. Furthermore, 1,2-propylene epoxide is a suspected carcinogenic compound. Finally, the formation of the three isomer alkenes as byproducts listed above is a disadvantage of the process. In the article, Goument presents methods for activation of the intermediate alcohol which are alternative to trifluoromethylsulfonate, for example by converting it to chloride (via thionyl chloride) or to mesylate (via mesyl chloride), but these process variations share the same disadvantages as the main process analyzed above. |
| In addition to the methods with multiple synthesis steps discussed so far in detail, the literature reports other methods or processes for producing fenfluramine or dexfenfluramine. In general, persons skilled in the art acknowledge that the syntheses in the literature for producing dexfenfluramine sometimes can be applied to the preparation of fenfluramine simply by replacing the initial materials and/or enantiopure reagents with the corresponding racemates while maintaining the reaction conditions. For example, patents that present long synthesis methods in multiple steps are the following: |
| DE1593595 and U.S. Pat. No. 3,769,319 |
| NL7215548 |
| EP810195 and EP882700 (dexfenfluramine) |
| EP0301925 (dexfenfluramine) |
| Other examples of preparation of fenfluramine, taken from non-patent literature, are the following: |
| Synthesis, November 1987, p. 1005-1007 |
| J. Org. Chem, 1991, 56, p. 6019 |
| Tetrahedron, 1994, 50(1), p. 171 |
| Bull. Soc. Chim. France, 1993, 130(4), p. 459-466 (dexfenfluramine) |
| Chirality, 2002, 14(4), p. 325-328 (dexfenfluramine) |
| Without analyzing in detail the individual methods described in these patents or articles, it can be stated in summary that all these methods are not attractive and interesting from the industrial standpoint because these are processes with many synthesis steps or because the initial materials described therein are not easily available and therefore have to be prepared separately, with a further expenditure of time and with further costs, or because they provide for the use of reagents that are dangerous/explosive/toxic or because they entail the use of catalysts based on heavy metals that can contaminate the final active ingredient. |
| One should consider that in the literature there are methods for the preparation of fenfluramine that did not provide for long syntheses and multiple steps but are shorter and consist of one or two steps. These processes, which therefore would be more interesting from the industrial standpoint, have other specific disadvantages, as will become apparent in detail hereinafter. For example, in the literature there is a first group of articles or patents that describe the reaction between 1-(3-trifluoromethyl)phenyl-propan-2-one and ethylamine in the presence of hydrogen gas and of a transition metal as catalyst: |
| In particular, in Huagong Shikan, 2002, 16(7), p. 33, the reaction is performed with hydrogen gas (2.9-3.38 atm), at 65-75° C., for 9 hours, in the presence of Raney nickel. Likewise, in patent DD108971 (1973), Raney nickel and hydrogen gas and methanol are used as solvent to perform this reaction. |
| In HU55343, instead, a similar reaction in one step is performed with hydrogen gas in the presence of another transition metal catalyst, such as palladium on carbon. |
| Although these three methods describe short single-step processes, they have the disadvantage of the use of hydrogen gas. As is known to persons skilled in the art, hydrogen gas is a dangerous gas due to the inherent danger of forming explosive mixtures with air and must be used by expert personnel in expensive facilities dedicated to its use and built with special precautions. Despite being used in purpose-built facilities, the use of hydrogen at the industrial level is inherently dangerous and to be avoided if possible. Another danger element that is shared by the processes described above is the fact that the reactions are performed under pressure. The third industrial disadvantage then arises from the use of heavy metal catalysts, which have a high cost and therefore increase the overall cost of the final active ingredient and may then contaminate the active ingredient fenfluramine even after filtration of the catalyst and purification of said active ingredient. |
| Analysis of the background art shows, however, that an attempt has been made to devise a process for the production or synthesis of fenfluramine that is short (one or two steps) and does not entail the use of hydrogen gas or of catalysts based on nickel or palladium or the like. In particular, for example, Synthesis 1987, 11, p. 1005, and then DECHEMA Monographien (1989), 112 (Org. Elektrochem.—Angew. Elektrothermie), 367-74, present a method for the synthesis of fenfluramine which starts from 1-(3-trifluoromethyl)phenyl-propan-2-one, which is made to react with ethylamine in great excess, in an electrochemical process, which uses a mercury cathode in a water/ethanol solution with pH 10-11. One obtains fenfluramine with 87% yield. This process has some drawbacks from an industrial standpoint: it is a process of the electrochemical type and therefore requires special equipment which is scarcely widespread, dedicated cells and reactors, and it is not possible to use the classic multipurpose reactors available in the pharmaceutical industry. Furthermore, the use of mercury at the industrial level poses severe environment safety problems, requiring constant health monitoring on workers who manage the equipment and systems for the management and destruction of wastewater that are particularly onerous; finally, mercury can be transferred from the cathode to the reaction environment and therefore to the active ingredient, and this obviously is to be considered very dangerous due to the accumulation of the metal in human beings; small traces of mercury are very toxic. |
| Another method for fenfluramine synthesis in a single step is the one presented in J. Org. Chem, 1979, 44(20), p. 3580. Here the reaction is described between an alkene derivative and ethylamine in the presence of sodium borohydride and mercury nitrate: |
| Again, this process is not interesting from an industrial standpoint since it has the same problems, if not even greater ones, related to the use of mercury (used here as a water-soluble salt) discussed previously. The complication introduced in this process with the use of mercury nitrate together with sodium borohydride highlights the level of innovation of the synthesis path found here. |
| In the past, therefore, it has not been possible to provide a process for synthesizing fenfluramine in a small number of steps by using modern reducing agents that are commonly and easily used. Indeed, while Gaodeng Xuexiao Huaxue Xuebao, 9(2), 1988, p. 134-139, describes and exemplifies the synthesis of 2-N-ethyl-1-phenyl propane by means of (1) the treatment of the precursor ketone with ethylamine followed by (2) sodium cyanoborohydride as reducing agent, Xuexiao Huaxue Xuebao provides no example for fenfluramine. Moreover, for the latter, Xuexiao Huaxue Xuebao indicates a melting point for the hydrochloride of 161° C., a data item that matches the value indicated in the literature initially (see BE609630); these facts prove thats fenfluramine synthesis with cyanoborohydride was not performed, otherwise one cannot explain why the author did not transcribe, in the document, the example of a product that at the time was very important. It should be noted in fact that 1-phenyl propan-2-one and 1-(3-trifluoromethyl)phenyl-propan-2-one can have different reactivities to reductive amination due to the presence of a highly electron-attractor-trifluoromethyl group, hence the need for an example to demonstrate its feasibility. The use of cyanoborohydride shares some disadvantages with other methods discussed in the preceding paragraphs. The excellent selectivity for reductive aminations of this reagent is highly appreciated, but its application can be less advantageous with respect to other reducing systems in the synthesis of fenfluramine, where the latter is intended for therapeutic application in human beings. The reasons for this are the possible contamination of the finished pharmaceutical active ingredient with cyanide ions, the toxicity of the reagent itself and finally the danger of its use. It is known to persons skilled in the art that sodium cyanoborohydride can release hydrocyanic acid if the pH of the reaction environment is acid enough and it is known that hydrocyanic acid is a powerful poison, since it competes with oxygen for hemoglobin coordination. As a consequence of this, particular care must be taken in its use and in the disposal of the production wastewater, which can be contaminated by cyanides. Not least, one must consider that the cost of sodium cyanoborohydride is considerable. |
| To conclude, it can be seen that more than 50 years after the publication of its first synthesis dated 1962, there are still numerous disadvantages or limitations in the synthesis paths developed in the past decades in the literature for the preparation of fenfluramine. |
| Moreover, recently there has been renewed pharmaceutical interest in the fenfluramine molecule, since the possibility of its therapeutic use in severe disorders of infancy has appeared in the medical literature. For example, mention can be made of Ceulemans et al., Epilepsia, 53(7), pages 1131 to 1139, 2012. |
| According to a certain part of medical literature, fenfluramine might therefore be interesting as a medication in a chronic therapy for the treatment of symptoms of epilepsy and other correlated severe disorders. |
| Based on recent medical developments, therefore, the need exists for a synthesis method that is better than the existing ones and can overcome in particular the disadvantages of the processes that are present in the literature. Particularly important, in view of use in chronic therapies for children such as epilepsy and other severe disorders, it would be fundamentally important to identify a path for synthesis of the active ingredient fenfluramine that does not entail the use of heavy metals and/or transition metals, which in a chronic therapy might accumulate in the body of the patients over the years, with severe consequences on health. |
| More generally, it is desirable to identify a synthesis path that uses reagents from which (or from the transformation products of which) it is then possible to easily purify fenfluramine. |
| It would be equally desirable to identify a synthesis path that comprises a small number of synthesis steps and uses reagents that are widely commercially available and easy to use. |
| At the same time, the new identified synthesis path should avoid if possible the formation of byproducts. |
DESCRIPTION OF THE FIGURES
| FIG. 1: impurities generated theoretically by means of the reagents used in the first fenfluramine synthesis according to BE609630. |
| FIG. 2: DSC of crude fenfluramine hydrochloride, obtained by reduction with sodium cyanoborohydride according to test 12 (table B) of the description that follows. |
| FIG. 3: DSC of fenfluramine hydrochloride recrystallized from 2-butanol as in example 2b (reduction with sodium borohydride). |
SUMMARY OF THE INVENTION
| The inventors of the present application have found surprisingly that the aim and objects indicated above are achieved by a new method for the synthesis of fenfluramine or of a pharmaceutically acceptable salt thereof, comprising the transformation of a ketone having the structure (I): |
(MOL) (CDX) wherein R 1 is CF 3, with ethylamine or with a salt thereof, and with a reducing agent chosen from the group consisting of alkaline cation or ammonium borohydride, alkaline cation or ammonium triacetoxyborohydride and alkaline cation or ammonium cyanoborohydride, in which the alkaline cation is always different from lithium cation and mixtures thereof, to yield fenfluramine, optionally followed by the transformation of the obtained fenfluramine into a pharmaceutically acceptable salt.
| Furthermore, the inventors of the present invention have also discovered a new preparation of fenfluramine, which can be obtained by means of the method described hereinafter, and new pharmaceutical compositions that contain it. |
Example 1
Synthesis of Fenfluramine
| A suspension of sodium hydroxide (34.62 g-0.866 mol, 3.5 eq) in 170 mL of methanol, under mechanical agitation, receives the addition, drop by drop, over the course of 30 minutes, of a solution of ethylamine hydrochloride (70.59 g-0.866 mol, 3.5 eq) in 165 mL of methanol, followed by 1-(3-trifluoromethyl)phenyl-propan-2-one (50 g-0.247 mol). The mixture is left under agitation at 20° C. for 4.5 hours, then cooling to 0° C. is performed and a solution of sodium borohydride (9.36 g-0.247 mol) in 19 mL of sodium hydroxide 1M in water is then added drop by drop, keeping the temperature below 10° C. The reaction is then left under agitation at 20° C. for another 2 hours. Once the reaction is complete, 270 mL of methanol are removed at a reduced pressure at 40° C. and then 200 mL of water are added and the mixture is extracted with heptane (200 mL). The aqueous phase is eliminated and the organic phase is washed with water (200 mL×3). The organic phase is concentrated at 50° C. at reduced pressure to yield free base fenfluramine as colorless oil. Yield: 72%; purity: 77%—as listed in test 3 of table A above. |
Example 2
Purification of Fenfluramine
| Purification of free base fenfluramine can be performed in two ways: |
| distillation of the free base |
| crystallization of the fenfluramine hydrochloride salt |
| Depending on the degree of purity that is desired, both purification processes are performed in sequence (distillation first and then crystallization), or only one of the two purification processes is performed. |
Example 2a
Distillation
| Free base fenfluramine (10 g), prepared as in Example 1, is distilled under reduced pressure with a distillation column of the Vigreux type: the distillation heads are eliminated, the fraction that is distilled at 89-90° C. at 6 mmHg, which is the active ingredient fenfluramine (8.5 g) with a high degree of purity, is collected. |
Example 2b
Conversion into Hydrochloride Salt and Crystallization
| Crude fenfluramine, prepared as in Example 1, or purified fenfluramine as in Example 2a, is dissolved in 125 mL of ethyl acetate, and cooling is performed to 0° Celsius under agitation. 272 mL of a solution of 1M HCl in ethyl acetate are added drop by drop at 0° C. The precipitate that forms is filtered and washed with ethyl acetate (125 mL×2) to yield approximately 55 g of solid fraction. The solid fraction is crystallized by 2-butanol (260 mL), keeping the solid for 22 hours at 3° C. under slow agitation before filtering it. Filtering is performed and washing is performed with cold 2-butanol. The solid fraction, fenfluramine hydrochloride, is dried in a vacuum stove, yielding 51.7 g of product. A DSC of the resulting product is shown in FIG. 3. |
PAPER
Journal of Organic Chemistry (1979), 44(20), 3580-3.J. Org. Chem. 1979, 44, 20, 3580–3583
Publication Date:September 1, 1979
https://doi.org/10.1021/jo01334a031https://pubs.acs.org/doi/abs/10.1021/jo01334a031
PATENT
https://patents.google.com/patent/US20170174613A1/en
- Fenfluramine is an amphetamine drug that was once widely prescribed as an appetite suppressant to treat obesity. Fenfluramine is devoid of the psychomotor stimulant and abuse potential of D-amphetamine and interacts with the 5-hydroxytryptamine (serotonin, 5-HT) receptors to release 5-HT from neurons. Fenfluramine has been investigated as having anticonvulsive activity in the treatment of Dravet Syndrome, or severe myoclonic epilepsy in infancy, a rare and malignant epileptic syndrome. This type of epilepsy has an early onset in previously healthy children.
- [0003]
Anorectic treatment with fenfluramine has been associated with the development of cardiac valvulopathy and pulmonary hypertension, including the condition cardiac fibrosis which led to the withdrawal of fenfluramine from world-wide markets. Interaction of fenfluramine’s major metabolite norfenfluramine with the 5-HT2B receptor is associated with heart valve hypertrophy. In the treatment of epilepsy, the known cardiovascular risks of fenfluramine are weighed against beneficial anticonvulsive activity.


- [0097]
Chemical Abstract Service (CAS) Registry Number (RN): 404-82-0 (HCl Salt), 458-24-2 (Parent Free Base) - [0098]
Chemical Name: N-ethyl-α-methyl-3-(trifluoromethyl)-benzeneethanamine hydrochloride (1:1). Other Names: Fenfluramine HCl, DL-Fenfluramine, (±)-Fenfluramine - [0099]
Structure of Hydrochloride Salt: - [0100]
Stereochemistry: Fenfluramine HCl has one chiral center and is being developed as the racemate and contains dexfenfluramine and levofenfluramine - [0101]
Molecular Formula of hydrochloride salt: C12H16F3N.HCl - [0102]
Molecular Mass/Weight: 267.72 g/mol
2. General Properties
- [0103]
Table 1 summarizes the chemical and physical properties of Fenfluramine HCl. - TABLE 1 General Properties of Fenfluramine HCl Drug Substance Property Result Appearance (color, White to off-white powder physical form) DSC (melting 170° C. (melt/sublimation) point)a TGA Onset 147° C. 0.03% at 150° C. 91% at 220° C. (evaporation) pKa (water) 10.15-10.38 Solubility (mg/mL) Resultant pH 25° C. 37° C. Solubility pH 6.69 (water) 54.13 71.22 (Aqueous) pH 1.73 buffer 25.34 53.68 pH 3.43 buffer 29.50 61.97 pH 6.41 buffer 37.42 95.60 0.9% NaCl (water) 22.98 — Solvent Solubility 25° C. (mg/mL) Solubility (Organic Ethanol 150 Solvents) Dichloromethane 30-35 Ethyl Acetate, 1-5 mg Tetrahydrofuran, Toluene, Acetonitrile UV Absorption Maxima: 210, 265 nm Solution pH (water) 6.69 Hygroscopicity @30% RH: ~0.05% (Dynamic Vapor @60% RH: ~0.07% Sorption (DVS) @90% RH: ~0.20%a) Polymorphism Fenfluramine HCl has been consistently isolated as a single crystalline Form 1 as determined by DSC and x-ray powder diffraction (XRPD) Solvation/Hydration Fenfluramine HCl is isolated as a nonhydrated, nonsolvated solid Solution Stability 8 weeks @ pH 6.7 phosphate buffer medium at 40° C. and 60° C. using concentrations of 0.5, 2.5 and 5.0 mg/ml. All conditions, no new impurities >0.1% by HPLC. Solid Stability 8 weeks @ 40° C., 60° C. and 80° C. 7 days at 150° C. All conditions, no new impurities >0.1% by HPLC.
3. Synthesis of Fenfluramine Drug Substance
- [0104]
Scheme 3.1 shows a 2-step route of synthesis used to manufacture initial clinical supplies of Fenfluramine HCl from ketone (2). The batch size is 4 kg performed in laboratory glassware (kilo lab). No chromatography is required and the process steps are amenable to scale-up. In process 1 there is one isolated intermediate Fenfluramine Free Base (1) starting from commercially supplied 1-(3-(trifluoromethyl)phenyl) acetone (Ketone 2). All steps are conducted under cGMPs starting from Ketone (2). - [0105]
Scheme 3.2 shows a 4-step route of synthesis to Fenfluramine HCl that can be used for commercial supply. Route 2 utilizes the same 2-step process used by Route 1 to convert Ketone (2) to Fenfluramine HCl with the exception that Ketone (2) is synthesized under cGMP conditions starting from 3-(Trifluoromethyl)-phenyl acetic acid (Acid 4). Bisulfate Complex (3) is an isolatable solid and can be purified before decomplexation to Ketone (2). In-situ intermediates which are oils are shown in brackets. Batch sizes of 10 Kg are performed. Commercial batch sizes of 20 kg are performed in fixed pilot plant equipment. Steps 1-2 of Scheme 3.2 to manufacture Ketone (2) have been demonstrated on a 100 g scale to provide high purity ketone (2) of >99.8% (GC & HPLC). Conversion of Ketone (2) to Fenfluramine using either Route 1 or 2 has provided similar purity profiles. - Starting materials are designated by enclosed boxes. Bracketed and non bracketed compounds respectively indicate proposed in-situ and isolated intermediates. NMI=N-Methyl Imidazole.
4.1. Narrative Description (Route 1)
- [0106]
Step 1: Reductive Amination (Preparation of Fenfluramine Free Base 1) - [0107]
A solution of ethylamine, water, methanol, and 1-(3-(trifluoromethyl)phenyl) acetone (Ketone 2) was treated with sodium triacetoxyborohydride and stirred for 16 h at 25° C. at which time HPLC analysis (IPC-1; In Process Control No. 1) showed the reaction to be complete and sodium hydroxide solution was added until pH>10. Toluene was added and the phases separated, and the aqueous phase (IPC-2) and organic phase (IPC-3) are checked for remaining Fenfluramine and Fenfluramine alcohol and the organic phase was reduced. Purified water was added and the pH adjusted to <2 using conc. HCl and the phases were separated. The aqueous phase was washed with toluene and the toluene phase (IPC-4) and the aqueous phase (IPC-5) was checked for Fenfluramine and Fenfluramine alcohol content. The aqueous phase containing product is pH adjusted to >10 using sodium hydroxide solution. The basic aqueous phase was extracted with MTBE until removal of Fenfluramine from the aqueous phase was observed by HPLC (<0.5 mg/ml) (IPC-6). The organic phase was dried over sodium sulfate and filtered. The filtrate was concentrated in vacuo to give the intermediate product Fenfluramine Free Base 1 as a pale yellow oil tested per specifications described herein which showed by NMR the material to contain 2.93% toluene giving an active yield of 88.3% with a purity of 98.23% by HPLC (0.67% Fenfluramine alcohol). - [0108]
Step 2: Salt Formation (Preparation of Fenfluramine HCl) - [0109]
To a flask was charged ethanol and acetyl chloride. The solution was stirred slowly overnight before ethyl acetate was added. The HCl in ethyl acetate solution formed was polish filtered into a clean carboy and retained for later use. To a vessel was added Fenfluramine free base 1 and MTBE. The Fenfluramine solution in MTBE was collected in two carboys before the vessel was cleaned and checked for particulate residue. The Fenfluramine solution was polish filtered into a vessel and cooled and HCl in ethyl acetate solution was added giving a final pH of 6-7. The batch was stirred for 1 h and filtered. The product was dried under vacuum at 40° C. The product (96.52% yield) was tested per IPC-7 had a purity of 99.75% by HPLC and GC headspace analysis showed MTBE (800 ppm) and EtOAc (150 ppm) to be present. The product was then tested per specifications shown herein.
4.2. Narrative Description (Route 2)
- [0110]
Step 1: Preparation of Ketone Bisulfite Adduct - [0111]
Procedure: Charge acetic anhydride, (2.8 vol, 3.0 wt, 5.0 eq.) to a vessel and commence stirring. Cool the solution to −5 to 5° C., targeting −4° C. Charge 1-methylimidazole, (0.2 vol, 0.21 wt, 0.5 eq.) to the mixture at −5 to 5° C. Caution: very exothermic. If necessary, adjust the temperature to 0 to 5° C. Charge ZX008 acid, (1.00 wt, 1.0 eq.) to the mixture at 0 to 5° C. Caution: exothermic. Stir the mixture at 0 to 5° C. until ≦2.1% area ZX008 acid by HPLC analysis, typically 7 to 9 hours. Charge 15% w/w sodium chloride solution (2.0 vol) to the mixture at 0 to 5° C., 60 to 90 minutes. Caution: very exothermic which will be slightly delayed. Warm the mixture to 18 to 23° C. over 45 to 60 minutes and continue stirring for a further 30 to 45 minutes at 18 to 23° C. Charge TBME, (5.0 vol, 3.7 wt) to the mixture and stir for 10 to 15 minutes at 18 to 23° C. Separate the aqueous layer and retain the organic layer. Back-extract the aqueous layer with TBME, (2×3.0 vol, 2×2.2 wt) at 18 to 23° C. retaining each organic layer. Adjust the pH of the combined organic layer to pH 6.5 to 9.0, targeting 7.0 by charging 20% w/w sodium hydroxide solution (5.3 to 8.3 vol) at 18 to 23° C. Caution: exothermic. Separate the aqueous layer and retain the organic layer. Wash the organic layer with 4% w/w sodium hydrogen carbonate solution (2×3.0 vol) at 18 to 23° C. Determine the residual ZX008 acid content in the organic layer by HPLC analysis, pass criterion ≦0.10% area ZX008 acid. Wash the organic layer with purified water, (2×3.0 vol) at 18 to 23° C. Concentrate the organic layer under reduced pressure to ca. 2 vol at 40 to 45° C., targeting 43° C. - [0112]
Determine the w/w assay of ZX008 ketone (WIP) in the mixture by 1H-NMR analysis for information only and calculate the contained yield of ZX008 ketone (WIP) in the mixture. Note: This step can be removed from the process since the process is robust and consistently delivers 80 to 90% th yield. The achieved yield was factored into the charges of the subsequent steps. - [0113]
Charge n-heptane, (4.0 vol, 2.7 wt) to the mixture at 40 to 45° C., targeting 43° C. Concentrate the mixture to ca. 2 vol at 40 to 45° C., targeting 43° C. Determine the TBME content in the mixture by 1H-NMR analysis, (pass criterion ≦5.0% w/w TBME vs. ZX008 ketone). Charge n-heptane, (2.4 vol, 1.6 wt) at 40 to 45° C., targeting 43° C., vessel A. To vessel B, charge sodium metabisulfite, (0.82 wt, 0.88 eq.) at 18 to 23° C. To vessel B, charge a solution of sodium hydrogen carbonate, (0.16 wt, 0.4 eq.) in purified water, code RM0120 (2.0 vol) at 18 to 23° C. followed by a line rinse with purified water, code RM0120 (0.4 vol) at 18 to 23° C. Caution: gas evolution. Heat the contents of vessel B to 40 to 45° C., targeting 43° C. Charge the contents from vessel A to vessel B followed by a line rinse with n-heptane, (0.8 vol, 0.5 wt) at 40 to 45° C., targeting 43° C. Stir the mixture for 1 to 1.5 hours at 40 to 45° C., targeting 43° C. Charge n-heptane, code RM0174 (3.2 vol, 2.2 wt) to the mixture with the temperature being allowed to cool to 18 to 45° C. at the end of the addition. Cool the mixture to 18 to 23° C. at approximately constant rate over 45 to 60 minutes. Stir the mixture at 18 to 23° C. for 1.5 to 2 hours. - [0114]
Sample the mixture to determine the residual ZX008 ketone content by 1H-NMR analysis, (pass criterion ≦10.0% mol, target 5.0% mol ZX008 ketone vs. ZX008 ketone bisulfite adduct). Filter the mixture and slurry wash the filter-cake with n-heptane, (2×2.0 vol, 2×1.4 wt) at 18 to 23° C. Dry the solid at up to 23° C. until the water content is <10.0% w/w water by KF analysis according to AKX reagent. At least 16 hours. Determine the w/w assay of the isolated ZX008 ketone bisulfite adduct by 1H-NMR analysis and calculate the contained yield of ZX008 ketone bisulfite adduct. - [0115]
Yields and Profiles: The yield for the stage 1 Demonstration batch is summarized Table below. Input: 1700.0 g uncorr., acid, 99.50% area (QC, HPLC), 2-isomer not detected, 4-isomer 0.02% area, RRT1.58 (previously not observed) 0.48% area as per the preparative method. The analytical data is summarized in Table 1A below. - TABLE 1A Table for isolated yields for step 1 Demonstration batch Corr. % area Reference Corr. Yield % w/w (HPLC, number Input Output (% th)** (1H-NMR)* QC) Comments Batch A1 1700.0 g 1500.1 g 89.1 45.0 —.— Crude ketone as TBME sol. Batch A2 1500.1 g 1716.1 77.8 76.0 98.15 Bisulfite adduct only 67.3 Overall product
- [0116]
Step 2: Preparation of Ketone - [0117]
Procedure: Charge toluene, (5.0 vol, 4.3 wt), and purified water, (5.0 vol) to the vessel and commence stirring. If necessary, adjust the temperature to 18 to 23° C. and charge ZX008 ketone bisulfite adduct, (1.00 wt corrected for % w/w assay) to the mixture at 18 to 23° C. Charge 20% w/w sodium hydroxide solution to the mixture at 18 to 23° C. adjusting the pH of the mixture to pH 8.0 to 12.0, targeting 9.0 (0.5 to 1.0 vol). - Separate the lower aqueous layer and retain the top organic layer. Wash the organic layer with purified water, (3.0 vol) at 18 to 23° C. Concentrate the organic layer under reduced pressure to ca. 2 vol at 45 to 50° C., targeting 48° C. Charge methanol, (5.0 vol, 4.0 wt) to the mixture at 45 to 50° C., targeting 48° C. Re-concentrate the mixture under reduced pressure to ca. 2 vol at 45 to 50° C., targeting 48° C. Repeat steps 7 and 8 once before continuing with step 9. Cool the mixture to 18 to 23° C. Clarify the mixture into a tared, suitably-sized drum followed by a methanol (1.0 vol, 0.8 wt) line rinse at 18 to 23° C. Determine the w/w assay of ZX008 ketone (WIP) in the mixture by 1H-NMR analysis and calculate the contained yield of ZX008 ketone (WIP) in the mixture. Determine the toluene content in the mixture by 1H-NMR analysis.
- [0118]
Yields and Profiles: The yield for the step 2 Demonstration batch is summarized in Table 1B below. Input: 1200.0 g corr. Ketone bisulfite adduct, 76.0% w/w assay (NMR, using DMB as internal standard in d6-DMSO), (1.00 eq, 1.00 wt corr. for w/w assay) for input calculation. - TABLE 1B Table for isolated yields for step 2 Demonstration batch % w/w % area Corr. Corr. Corr. Yield (1H- (HPLC, Input Output (% th) NMR)* QC) Comments 1200.0 g 858.15 g 108.3 25.5 99.31 Purified ketone
- [0119]
Step 3: Preparation of Fenfluramine HCl Crude - [0120]
Procedure: Charge the ZX008 ketone (corr. for assay, 1.00 wt, 1.00 eq. isolated as solution in MeOH in stage 2) to a vessel. Charge methanol, code RM0036 (5.0 vol, 4.0 wt) to the mixture at 18 to 23° C. Cool the solution to 0 to 5° C. Charge 70 wt % aqueous ethylamine solution (1.3 vol, 1.6 wt, 4.0 eq) to the mixture at 0 to 10° C., over 15 to 30 minutes, followed by a line rinse with methanol (1.0 vol, 0.8 wt). Warm the mixture to 15 to 20° C. and stir the mixture for a further 60 to 70 minutes at 15 to 20° C. Adjust the mixture to 15 to 18° C. if required, targeting 15° C. Charge sodium triacetoxyborohydride (2.4 wt, 2.25 eq.) to the mixture in approximately 10 portions, keeping the mixture at 15 to 20° C., targeting 17° C. Addition time 1.5 to 2 hours. Caution: Exothermic. Stir the mixture at 15 to 20° C. until complete by HPLC analysis, pass criterion ≦3.0% area ZX008 ketone, typically 2 to 3 hours. Adjust the pH of the mixture to pH>12 by charging 20% w/w aqueous sodium hydroxide solution (5.0 to 6.0 vol) to the mixture at 15 to 40° C. Addition time 10 to 30 minutes. Caution: Exothermic. If necessary, adjust the temperature to 18 to 23° C. Extract the mixture with toluene (3×3.0 vol, 3×2.6 wt) at 18 to 23° C., retaining and combining the top organic layer after each extraction. Wash the combined organic layer with purified water, (1.0 vol) at 18 to 23° C. Heat the mixture to 40 to 50° C., targeting 48° C. Concentrate the mixture under reduced pressure at constant volume maintaining ca. 5 vol by charging the organic layer at approximately the same rate as the distillation rate at 40 to 50° C., targeting 48° C. Cool the mixture to 18 to 23° C. Charge purified water (10.0 vol) to the mixture at 18 to 23° C. Adjust the pH of the mixture to 0.1<pH<1.5 at 18 to 23° C. by charging concentrated hydrochloric acid, 0.5 vol. Do not delay from this step until neutralization. - [0121]
Separate the layers at 18 to 23° C. retaining the bottom aqueous layer. Wash the aqueous layer with toluene, (3.0 vol, 2.6 wt) at 18 to 23° C. retaining the aqueous layer. Adjust the pH of the aqueous layer to pH>12 by charging 20% w/w sodium hydroxide solution at 18 to 23° C. 0.8 to 0.9 vol. Caution: Exothermic. Charge TBME, code RM0002 (2.0 vol, 1.5 wt) to the basic aqueous layer. Separate the layers at 18 to 23° C. retaining the top organic layer. Back-extract the aqueous layer with TBME (2×2.0 vol, 2×1.5 wt) at 18 to 23° C. retaining the organic layers. Wash the combined organic layer with purified water, (2×1.0 vol) at 18 to 23° C. Concentrate the combined organic layers under reduced pressure at 40 to 50° C., targeting 48° C. to ca. 3 vol. Determine the residual toluene content of the mixture by 1H-NMR analysis. Sample for determination of residual water content by KF analysis, AKX reagent. Charge TBME (8.7 vol, 6.4 wt) to the mixture at 40 to 50° C. Cool the solution to 0 to 5° C., targeting 2° C. Charge concentrated hydrochloric acid (0.54 vol, 0.46 wt) maintaining the temperature <15° C. Caution: Exothermic. Line rinse with TBME (1.0 vol, 0.7 wt). If necessary, adjust the temperature to 0 to 10° C. and stir the mixture at 0 to 10° C. for a further 2 to 3 hours. Filter the mixture and wash the filter-cake with TBME (2×4.4 vol, 2×3.3 wt) at 0 to 10° C. Dry the solid at up to 40° C. until the TBME content is <0.5% w/w TBME by 1H-NMR analysis. 4 to 8 hours. - [0122]
Yields and Profiles: The yield for the step 3 Demonstration batch is summarized in Table 1C below. Input: 856.8 g corr. Ketone, 44.2% w/w assay (NMR, using TCNB as internal standard in CDCl3), (1.00 eq, 1.00 wt corr. for w/w assay) for input calculation. FIG. 2 and Table 1D shows an exemplary HPLC chromatogram of a crude preparation of fenfluramine hydrochloride (210 nm UV absorbance). - TABLE 1C Table for isolated yields for step 3 Demonstration batch Corr. % area Reference Corr. Corr. Yield % w/w (HPLC, number Input Output (% th) (1H-NMR)* QC) Comments Batch A1 856.8 g 836.31 g 85.3 44.2 99.15 Fenfluramine free base (in situ intermediate) Batch A2 880.7 84.0 based 99.5 100.00 Fenfluramine•HCl on ketone crude (step 3 an bisulfite d 4.1) adduct (77.6 based on purified ketone)
- TABLE 1D Purity of crude fenfluramine hydrochloride by HPLC (see FIG. 2) Processed Channel Descr. DAD AU Ch 1 Sample 210, Bw 4 Peak Results USP USP USP Name RT RelRT Area Height Tailing Resolution Plate Count EP s/n % Area 1 NorFenfluramine 7.46 2 2-Fenfluramine 7.68 3 Fenfluramine 8.67 1.000 3789064 778178 1.7 70796 2549.8 99.15 4 4-Fenfluramine 8.95 5 11 34 1.308 6073 1449 1.2 23.5 215529 3 8 0.16 6 ZX008 acid 12.93 7 Fenfluramine alcohol 14.16 1.633 15266 2972 1.3 24.8 215040 8.7 0.40 8 ZX008 ketone 14.83 9 Fenfluramine acetamide 15.55 10 TOLUENE 15 75 11 15.92 1.836 4110 1122 2.7 0.11 12 16.60 1.915 6861 1630 1.5 451209 4.3 0.18 Sum 3821374 100.00
- [0123]
Step 4.2: Crystallization of Fenfluramine Hydrochloride - [0124]
Procedure: Charge Fenfluramine.HCl (crude) (1.00 wt, 1.0 eq.) and TBME (10.0 vol, 7.4 wt) to the vessel and commence stirring. Heat the suspension to reflux (50 to 58° C.). Charge ethanol (5.0 vol, 3.9 wt) maintaining the temperature at 50 to 58° C. Addition time 20 minutes. Stir at 50 to 58° C. for 5 to 10 minutes and check for dissolution. Stir the solution at 50 to 58° C. for 5 to 10 minutes, targeting 54 to 58° C. Clarify the reaction mixture through a 0.1 μm in-line filter at 54 to 58° C., followed by a line rinse with TBME (1 vol, 0.7 wt). Cool the solution to 48 to 50° C. Charge Fenfluramine HCl, code FP0188 (0.01 wt). Check for crystallization. Allow the suspension to cool to 15 to 20° C., target 17° C. over 5 to 5.5 hours at an approximately constant rate. Stir the mixture at 15 to 20° C., target 17° C. for 2 to 3 hours. Filter the mixture and wash the filter-cake with clarified TBME (2×3.0 vol, 2×2.2 wt) at 5 to 15° C. Dry the solid at up to 40° C. until the TBME content is <0.5% w/w TBME and the ethanol content is <0.5% w/w EtOH by 1H-NMR analysis. 4 to 8 hours. Determine the w/w assay of the isolated Fenfluramine.HCl by 1H-NMR analysis. - [0125]
Yields and Profiles: The yield for the stage 4 Demonstration batch is summarized in Table 1E below. Input: 750.0 g uncorr. Fenfluramine HCl crude (1.00 eq, 1.00 wt uncorr.) for input calculation. FIG. 3 shows an exemplary HPLC chromatogram of a crystallized fenfluramine hydrochloride sample (210 nm UV absorbance). - TABLE 1E Table for isolated yields for stage 4 Demonstration batch Uncorr. Uncorr. Uncorr. Yield HPLC (% area, Input Output (% th) QC) Comments 750.0 g 608.0 81.1 100.00* Fenfluramine•HCl
PATENT
https://patents.google.com/patent/EP3170807A1/en
- Fenfluramine, i.e., 3-trifluoromethyl-N-ethylamphetamine, has the following chemical structure:
- [0003]
The marketing of fenfluramine as a pharmaceutical active ingredient in the United States began in 1973 and was used in a therapy in combination with phentermine to prevent and treat obesity. However, in 1997 fenfluramine was withdrawn from the market in the United States and immediately thereafter in other countries, since its ingestion was associated with the onset of cardiac fibrosis and pulmonary hypertension. As a consequence of this event, the pharmaceutical compounds containing this active ingredient were withdrawn from the market. However, fenfluramine, even after its exit from the market, has continued to attract scientific interest, as will become apparent from the discussion presented hereinafter. - [0004]
In the literature, over the years, numerous syntheses or processes have been reported for preparing fenfluramine or its dextrorotatory enantiomer dexfenfluramine or an analog containing a highly electron-attractor group on the aromatic ring as in the fenfluramine molecule (see for example Pentafluorosulfanyl Serotonin Analogs: Synthesis, Characterization, and Biological Activity, John T. Welch and Dongsung Lim Chapter 8, pp 165-181 DOI: 10.1021/bk-2009-1003.ch008). Many of these synthesis paths are long and foresee multiple stages or synthesis steps that can include reagents that are dangerous or scarcely environment-friendly and are therefore scarcely convenient for an industrial synthesis. Hereinafter, any reference to “fenfluramine” is understood to referto the racemic form, i.e, (RS)-N-ethyl-1-[3-(trifluoromethyl)phenyl]propan-2-amine. - [0005]
To the best of the knowledge of the inventors, the first method for fenfluramine synthesis reported in the literature dates back to 1962 and is referenced in patent BE609630 and in analogous patents US3198833 and FR1324220 . All the synthesis methods reported in these patents provide for numerous synthesis steps. By way of example, one of the methods provides for the transformation into oxime of a ketone, 1-(3-trifluoromethyl)phenyl-propan-2-one, as shown here: - [0006]
The oxime is then hydrogenated in the presence of Raney nickel catalyst so as to yield the corresponding primary amine, which is acetylated subsequently with ethanoic anhydride before being converted into fenfluramine by reduction with lithium aluminum hydride. - [0007]
As can be seen, the final step of this chemical process provides for the use of lithium aluminum hydride and the persons skilled in the art will acknowledge that the use of this reagent should be avoided, if possible, on an industrial level, since it is extremely flammable and is the source of accidents. Furthermore, lithium is a potentially neurotoxic metal and therefore its use should be avoided where possible. Furthermore, the Raney nickel catalyst is used in the oxime reduction step and can contaminate the final active ingredient; the use of hydroxylamine also entails problems of toxicity for workers assigned to production. - [0008]
A further disadvantage of this process is, as already mentioned earlier, the number of steps, not only because a large number of synthesis steps entails a reduction of the overall yield of active ingredient, but also because each synthesis step in principle can generate impurities and a larger number of steps can therefore entail a higher number of impurities in the final active ingredient. Many of these impurities, furthermore, due to their structural similarity to fenfluramine, are difficult to eliminate and remove from a fenfluramine preparation. One impurity for example that can be formed in the process described above and is difficult to eliminate is the following: - [0009]
This impurity, which is a primary amine, shares physical-chemical properties that are similar to fenfluramine and therefore, like fenfluramine, it can form a hydrochloride salt by treatment with hydrochloric acid and thus contaminate the active ingredient fenfluramine hydrochloride. Furthermore, this impurity – as a free base – has a boiling point that is similar to that of fenfluramine (73°C vs. 89°C at 6 mmHg respectively), and therefore its elimination by distillation also can be problematic. - [0010]
The process described above can in principle generate other impurities, which are listed in Figure 1 . - [0011]
EP 0441160 claims a synthesis in 5 steps of dexfenfluramine, dextrorotatory enantiomer of fenfluramine. This synthesis can be adapted easily to produce fenfluramine instead of its dextrorotatory enantiomer simply by performing the first reduction step with a non-chiral reducing agent. In the first step, in fact:a ketone, 1-(3-trifluoromethyl)phenyl-propan-2-one, is first reduced to the corresponding alcohol in the presence of yeast, D-glucose, ethanol and water. Then the alcohol is converted into the tosylate in the second step: - [0012]
This reaction occurs in the presence of triethylamine and tosyl chloride in methylene chloride as solvent. After purification, the tosylate is converted to fenfluramine by means of three successive steps: - [0013]
In the first of these three steps, the tosylate is converted into an azide intermediate by reaction with sodium azide in dimethylformamide. The azide intermediate is then hydrogenated in the presence of a catalyst, palladium on carbon. Finally, the resulting primary amine is converted into fenfluramine by reaction with acetaldehyde and sodium borohydride. - [0014]
Persons skilled in the art may see easily that this process is not desirable from an industrial standpoint due to reasons related to environmental risk, safety and costs. For example, the sodium azide used in the process is a notoriously explosive compound and its use at the industrial level is dangerous. Furthermore, palladium is an expensive material and its use in the process entails an increase in the production costs of fenfluramine. Furthermore, palladium can contaminate the finished active ingredient. - [0015]
In another method for the synthesis of dexfenfluramine in 3-4 steps, reported by Goument et al. in Bulletin of the Chemical Society of France (1993), 130, p. 450-458, 3-bromobenzotrifluoride is subjected to a Grignard reaction with enantiopure 1,2-propylene-epoxide to yield 1-[3-(trifluoromethyl)phenyl]propan-2-ol as shown hereafter: - [0016]
If this reaction is performed with racemic 1,2-propylene-epoxide, the synthesis can be adapted to the preparation of fenfluramine. - [0017]
The alcohol thus obtained is first transformed into trifluoromethyl sulfonate by reaction with trifluoromethanesulfonic anhydride and then treated with ethylamine to yield fenfluramine, as shown in the diagram hereinafter: - [0018]
In this article, the authors acknowledge that the main byproducts of the reaction are isomer alkenes having the following chemical structures: - [0019]
The process proposed by Goument et al. is not interesting from the industrial standpoint for a series of reasons. First of all, it is known that the use of Grignard reagents, especially on an industrial scale, is problematic, because these compounds are often pyrophoric and corrosive. Furthermore, 1,2-propylene epoxide is a suspected carcinogenic compound. Finally, the formation of the three isomer alkenes as byproducts listed above is a disadvantage of the process. In the article, Goument presents methods for activation of the intermediate alcohol which are alternative to trifluoromethylsulfonate, for example by converting it to chloride (via thionyl chloride) or to mesylate (via mesyl chloride), but these process variations share the same disadvantages as the main process analyzed above. - [0020]
In addition to the methods with multiple synthesis steps discussed so far in detail, the literature reports other methods or processes for producing fenfluramine or dexfenfluramine. In general, persons skilled in the art acknowledge that the syntheses in the literature for producing dexfenfluramine sometimes can be applied to the preparation of fenfluramine simply by replacing the initial materials and/or enantiopure reagents with the corresponding racemates while maintaining the reaction conditions. For example, patents that present long synthesis methods in multiple steps are the following: - [0021]
Other examples of preparation of fenfluramine, taken from non-patent literature, are the following:- Synthesis, Nov.1987, p. 1005-1007
- J.Org.Chem, 1991, 56, p. 6019
- Tetrahedron, 1994, 50(1), p. 171
- Bull. Soc. Chim. France, 1993, 130(4), p. 459-466 (dexfenfluramine)
- Chirality, 2002, 14(4), p. 325-328 (dexfenfluramine)
- [0022]
Without analyzing in detail the individual methods described in these patents or articles, it can be stated in summary that all these methods are not attractive and interesting from the industrial standpoint because these are processes with many synthesis steps or because the initial materials described therein are not easily available and therefore have to be prepared separately, with a further expenditure of time and with further costs, or because they provide for the use of reagents that are dangerous/explosive/toxic or because they entail the use of catalysts based on heavy metals that can contaminate the final active ingredient. - [0023]
One should consider that in the literature there are methods for the preparation of fenfluramine that do not provide for long syntheses and multiple steps but are shorter and consist of one or two steps. These processes, which therefore would be more interesting from the industrial standpoint, have other specific disadvantages, as will become apparent in detail hereinafter. For example, in the literature there is a first group of articles or patents that describe the reaction between 1-(3-trifluoromethyl)phenyl-propan-2-one and ethylamine in the presence of hydrogen gas and of a transition metal as catalyst: - [0024]
In particular, in Huagong Shikan, 2002, 16(7), p. 33, the reaction is performed with hydrogen gas (2.9 – 3.38 atm), at 65-75°C, for 9 hours, in the presence of Raney nickel. Likewise, in patent DD108971 (1973), Raney nickel and hydrogen gas and methanol are used as solvent to perform this reaction. - [0025]
In HU55343 , instead, a similar reaction in one step is performed with hydrogen gas in the presence of another transition metal catalyst, such as palladium on carbon. - [0026]
Although these three methods describe short single-step processes, they have the disadvantage of the use of hydrogen gas. As is known to persons skilled in the art, hydrogen gas is a dangerous gas due to the inherent danger of forming explosive mixtures with air and must be used by expert personnel in expensive facilities dedicated to its use and built with special precautions. Despite being used in purpose-built facilities, the use of hydrogen at the industrial level is inherently dangerous and to be avoided if possible. Another danger element that is shared by the processes described above is the fact that the reactions are performed under pressure. The third industrial disadvantage then arises from the use of heavy metal catalysts, which have a high cost and therefore increase the overall cost of the final active ingredient and -on the other hand- may contaminate the active ingredient fenfluramine even after filtration of the catalyst and purification of said active ingredient. - [0027]
Analysis of the background art shows, however, that an attempt has been made to devise a process for the production or synthesis of fenfluramine that is short (one or two steps) and does not entail the use of hydrogen gas or of catalysts based on nickel or palladium or the like. In particular, for example, Synthesis 1987, 11, p. 1005, and then DECHEMA Monographien (1989), 112 (Org. Elektrochem.–Angew. Elektrothermie), 367-74, present a method for the synthesis of fenfluramine which starts from 1-(3-trifluoromethyl)phenyl-propan-2-one, which is made to react with ethylamine in great excess, in an electrochemical process, which uses a mercury cathode in a water/ethanol solution with pH 10-11. One obtains fenfluramine with 87% yield. This process has some drawbacks from an industrial standpoint: it is a process of the electrochemical type and therefore requires special equipment which is scarcely widespread, dedicated cells and reactors, and it is not possible to use the classic multipurpose reactors available in the pharmaceutical industry. Furthermore, the use of mercury at the industrial level poses severe environment safety problems, requiring constant health monitoring on workers who manage the equipment and systems for the management and destruction of wastewater that are particularly onerous; finally, mercury can be transferred from the cathode to the reaction environment and therefore to the active ingredient, and this obviously is to be considered very dangerous due to the accumulation of the metal in human beings; small traces of mercury are very toxic. - [0028]
Another method for fenfluramine synthesis in a single step is the one presented in J.Org.Chem, 1979, 44(20), p. 3580. Here the reaction is described between an alkene derivative and ethylamine in the presence of sodium borohydride and mercury nitrate: - [0029]
Again, this process is not interesting from an industrial standpoint since it has the same problems, if not even greater ones, related to the use of mercury (used here as a water-soluble salt) discussed previously. The complication introduced in this process with the use of mercury nitrate together with sodium borohydride highlights the level of innovation of the synthesis path found here. - [0030]
In past years, therefore, it has not been possible to provide a process for synthesizing fenfluramine in a small number of steps by using modern reducing agents that are commonly and easily used. Indeed, while Gaodeng Xuexiao Huaxue Xuebao, 9(2), 1988, p. 134-139, describes and exemplifies the synthesis of 2-N-ethyl-1-phenyl propane by means of (1) the treatment of the precursor ketone with ethylamine followed by (2) sodium cyanoborohydride as reducing agent, Xuexiao Huaxue Xuebao provides no example for fenfluramine. Moreover, for the latter, Xuexiao Huaxue Xuebao indicates a melting point for the hydrochloride of 161°C, a data item that matches the value indicated in the literature initially (see BE609630 ); these facts prove that fenfluramine synthesis with cyanoborohydride was not performed, otherwise one cannot explain why the author did not transcribe, in the document, the example of a product that at the time was very important. It should be noted in fact that 1-phenyl propan-2-one and 1-(3-trifluoromethyl)phenyl-propan-2-one can have different reactivities to reductive amination due to the presence of a highly electron-attractor -trifluoromethyl group, hence the need for an example to demonstrate its feasibility. The use of cyanoborohydride shares some disadvantages with other methods discussed in the preceding paragraphs. The excellent selectivity for reductive aminations of this reagent is highly appreciated, but its application can be less advantageous with respect to other reducing systems in the synthesis of fenfluramine, where the latter is intended for therapeutic application in human beings. The reasons for this are the possible contamination of the finished pharmaceutical active ingredient with cyanide ions, the toxicity of the reagent itself and finally the danger of its use. It is known to persons skilled in the art that sodium cyanoborohydride can release hydrocyanic acid if the pH of the reaction environment is acid enough and it is known that hydrocyanic acid is a powerful poison, since it competes with oxygen for hemoglobin coordination. As a consequence of this, particular care must be taken in its use and in the disposal of the production wastewater, which can be contaminated by cyanides. Not least, one must consider that the cost of sodium cyanoborohydride is considerable. - [0031]
To conclude, it can be seen that more than 50 years after the publication of its first synthesis dated 1962, there are still numerous disadvantages or limitations in the synthesis paths developed in the past decades in the literature for the preparation of fenfluramine. - [0032]
Moreover, recently there has been renewed pharmaceutical interest in the fenfluramine molecule, since the possibility of its therapeutic use in severe disorders of infancy has appeared in the medical literature. For example, mention can made of Ceulemans et al., Epilepsia, 53(7), pages 1131 to 1139, 2012. - [0033]
According to a certain part of medical literature, fenfluramine might therefore be interesting as a medication in a chronic therapy for the treatment of symptoms of epilepsy and other correlated severe disorders. - [0034]
Based on recent medical developments, therefore, the need exists for a synthesis method that is better than the existing ones and can overcome in particular the disadvantages of the processes that are present in the literature. Particularly important, in view of use in chronic therapies for children such as epilepsy and other severe disorders, it would be fundamentally important to identify a path for synthesis of the active ingredient fenfluramine or of isomers thereof and/or analogs thereof that does not entail the use of heavy metals and/or transition metals, which in a chronic therapy might accumulate in the body of the patients over the years, with severe consequences on health. - [0035]
More generally, it is desirable to identify a synthesis path that uses reagents from which (or from the transformation products of which) it is then possible to easily purify fenfluramine (or isomers and/or analogs thereof). - [0036]
It would be equally desirable to identify a synthesis path that comprises a small number of synthesis steps and uses reagents that are widely commercially available and easy to use. - [0037]
At the same time, the new identified synthesis path should avoid if possible the formation of byproducts.
EXAMPLES
- [0082]
The present invention is exemplified by, but not limited to, the following examples:
Example 1 – Synthesis of fenfluramine
- [0083]
A suspension of sodium hydroxide (34.62 g – 0.866 mol, 3.5 eq) in 170 mL of methanol, under mechanical agitation, receives the addition, drop by drop, over the course of 30 minutes, of a solution of ethylamine hydrochloride (70.59 g – 0.866 mol, 3.5 eq) in 165 mL of methanol, followed by 1-(3-trifluoromethyl)phenyl-propan-2-one (50 g – 0.247 mol). The mixture is left under agitation at 20°C for 4.5 hours, then cooling to 0°C is performed and a solution of sodium borohydride (9.36 g – 0.247 mol) in 19 mL of sodium hydroxide 1M in water is then added drop by drop, keeping the temperature below 10°C. The reaction is then left under agitation at 20°C for another 2 hours. Once the reaction is complete, 270 mL of methanol are removed at a reduced pressure at 40°C and then 200 mL of water are added and the mixture is extracted with heptane (200 mL). The aqueous phase is eliminated and the organic phase is washed with water (200 mL x 3). The organic phase is concentrated at 50°C at reduced pressure to yield free base fenfluramine as colorless oil. Yield: 72%; purity: 77% – as listed in test 3 of table A above.
Example 2 – Purification of fenfluramine
- [0084]
Purification of free base fenfluramine can be performed in two ways: - distillation of the free base
- crystallization of the fenfluramine hydrochloride salt
- [0085]
Depending on the degree of purity that is desired, both purification processes are performed in sequence (distillation first and then crystallization), or only one of the two purification processes is performed.
Example 2a – Distillation:
- [0086]
Free base fenfluramine (10 g), prepared as in Example 1, is distilled under reduced pressure with a distillation column of the Vigreux type: the distillation heads are eliminated, the fraction that is distilled at 89-90°C at 6 mmHg, which is the active ingredient fenfluramine (8.5 g) with a high degree of purity, is collected.
Example 2b – Conversion into hydrochloride salt and crystallization:
- [0087]
Crude fenfluramine, prepared as in Example 1, or purified fenfluramine as in Example 2a, is dissolved in 125 mL of ethyl acetate, and cooling is performed to 0°Celsius under agitation. 272 mL of a solution of 1M HCl in ethyl acetate are added drop by drop at 0°C. The precipitate that forms is filtered and washed with ethyl acetate (125 mL x 2) to yield approximately 55 g of solid fraction. The solid fraction is crystallized by 2-butanol (260 mL), keeping the solid for 22 hours at 3°C under slow agitation before filtering it. Filtering is performed and washing is performed with cold 2-butanol. The solid fraction, fenfluramine hydrochloride, is dried in a vacuum stove, yielding 51.7 g of product. A DSC of the resulting product is shown in Figure 3 .
CLIP
- Synthetic Method of Dexfenfluramine hydrochloride
- (CAS NO.: ), with its systematic name of (S)-N-Ethyl-alpha-methyl-m-(trifluoromethyl)phenethylamine hydrochloride, could be produced through many synthetic methods.Following is one of the synthesis routes:
 The action of d-camphoric acid on (rac)-fenfluramine (I) affords the camphorate of (+)-fenfluramine (II). After purification of this salt by crystallization, sodium hydroxide in methylene chloride is added, forming (+)-fenfluramine (III) after removal of camphoric acid. Finally, the action of hydrogen chloride in methyl cyclohexane on (+)-fenfluramine produces the corresponding salt: (+)-fenfluramine hydrochloride.
The action of d-camphoric acid on (rac)-fenfluramine (I) affords the camphorate of (+)-fenfluramine (II). After purification of this salt by crystallization, sodium hydroxide in methylene chloride is added, forming (+)-fenfluramine (III) after removal of camphoric acid. Finally, the action of hydrogen chloride in methyl cyclohexane on (+)-fenfluramine produces the corresponding salt: (+)-fenfluramine hydrochloride.
PAPER
https://www.designer-drug.com/pte/12.162.180.114/dcd/chemistry/fenfluramine.html
Fenfluramine 1 is the active ingredient of a obesity drug acting on the digestion of carbohydrates, the activity being restricted mainly to the S enantiomer [1, 2], which can be obtained by separation of the diastereoisomers [3] or by preferential crystallisation of derivates, which were identified of being conglomerates [4]. Only two syntheses of optical active fenfluramine have been described until now: one by stereoselective reduction of the imine derived from the ketone 2 and (R) or (S)-alpha-phenylethylamine [5], the other starting from (S)-alanine [6]. Two recent publications [7, 8] about the synthesis of (S)-fenfluramine via the intermediate alcohol (S)-3 (scheme 1) made us publish our previous results [9]. Through yeast reduction of the ketone 2, the authors obtain the alcohol (S)-3, the configuration of which they inverse in three steps. The alcohol (R)-3, via the intermediate tosylate (R)-4a and further the azide (S)-5 leads to the amine (S)-6 after reduction and finally to the (S)-fenfluramine (S)-1 after reductive amination in presence of acetaldehyde:
The (S)-fenfluramine is such obtained in 7 steps starting from the alcohol (S)-3 or in 4 steps from the alcohol (R)-3.
In this article we present a new way of preparing the two enantiomers of the alcohol 3, a new two step synthesis of (S)-fenfluramine starting from the alcohol (R)-3, a one step synthesis of (S)-fenfluramine starting from the azide (S)-5, which doesn’t pass over the intermediate primary amine (S)-6 and finally a much faster process (3 steps) of preparing (S)-fenfluramine starting from the alcohol (S)-3.
Results and discussion
Synthesis of 1-[3-(trifluoromethyl)phenyl]propan-2-ol (R)-3
The racemic alcohol is seldom mentioned. It’s one of the metabolites of fenfluramine in the human body, secreted in urine [10]. It can also be obtained by metabolic transformations of an oxime by different kinds of microorganisms [11]. It was used as an intermediate for the synthesis of a family of anorexics [12] and a family of antispasmodic and psychotherapeutic agents [13]. It was obtained by the reaction of methyloxirane 7 with the magnesium compound 8, with a yield of 50%. This same reaction was described earlier as being little regioselective [14], a fact we observed too [9].
The only synthesis of the optically active alcohol 3 is the reduction of the ketone 2 with yeast as described above. One abtains the S enantiomer, the R enantiomer is obtained by inversion.
On our part we used the condensation of the commercial [15] methyloxirane (R)-7, of which many syntheses are known [16], with the magnesium compound 8 and cuprous chloride [17, 18].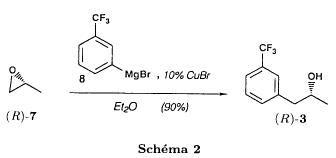
The yield is about 90% and the reaction very selective (purity GC: 93%). The optical purity of the methyloxirane 7 was determined by 1H-NMR in presence of the europium complex Eu(hfc)3 [19]. The optical purity of the alcohol (R)-3 was obtained by 1H-NMR and HPLC over silica of the Mosher derivate [20]. The comparison of these values show that the chiral centre is preserved. This procedure has the advantage of allowing us the preparation of the alcohol (S)-3 with the same reaction, because the methyloxirane (S)-7 is also commercially available and multiple syntheses are known [21].
Two step synthesis of the fenfluramine (S)-1 starting from the alcohol (R)-3
With the goal of obtaining the simplest procedure we have studied at first the transformation of the alcohol (R)-3 into fenfluramine (S)- 1 in two steps via the intermediate of the easily obtained sulfonates (R)-4: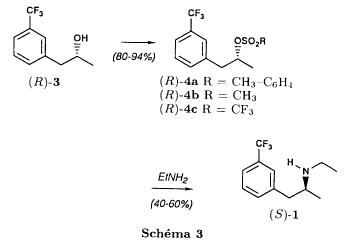
The substitution of the mesitylate (R)-4b and the tosylate (R)-4a with ethylamine was realised with medium yields always between 40 and 50% in spite of the large number of conditions tested: solvents (DMSO, DMF, ethanol, ethylamine), different dilutions (in proportions from 1 to 5) and temparatures from 50 to 160°C (with different times of contact). With the triflate (R)-4c the yield of the substitution is 60% but under non comparable conditions (-20°C in acetonitrile) because of its higher reactivity. In all cases the non aminated, and thus easily separated, byproducts are mainly the alkenes 9, 10Z and 10E (10E >> 10Z > 9).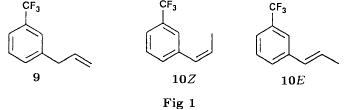
The enantiomeric purity of the amine (S)-1 is analysed by HPLC chromatography through silica of the camphanylated derivate [22] and compared to the previously analysed alcohol (R)-3: we have thus shown that the optical centre is conserved during the nucleophilic substitution. One had indeed to fear that due to the participation of the aromatic ring as neighbour group there could be partial or complete racemisation with an phenonium ion as intermediate. With the results obtained, which match with the literature [23-26], one can suppose that the trifluoromethyl group in meta position is sufficiently deactivating the aromatic ring in order to prevent participation in the substitution. We probably have thus in our case a pure nucleophilic SN2 substitution in competition with an elimination reaction. We believe that this elimination reaction is due to the simultaneous nucleophilic and basic properties of the ethylamine.
Although the yields are medium, this method has the advantage of being relatively fast because it permits to prepare fenfluramine (S)-1 starting from the alcohol (R)-2 in two steps instead of four [7, 8]. As far as we know it was never mentioned in literature.
Synthesis of fenfluramine (S)-1 from 2-azido-1-[3-(trifluoromethyl)phenyl]propane (S)-5
The substitution of the mesylate (R)-4b by sodium azide (scheme 4), an only slightly basic nucleophile compared to ethylamine, forms no elimination side products. One obtains the optically pure azide (S)-5 with a yield of 95%.
The enantiomeric purity couldn’t be directly analysed on the azide (S)-5. Only for analytical purposes did we reduce it into the amine (S)- 6. Among the numerous methods for the reductions of azides to amines mentioned in the literature [27] we chose the catalytic hydrogenation with 5% Pd on calcium carbonate at standard temperature and pressure [27f]. The HPLC analysis through silica column of the champhanyl derivate [22] of the amine (S)-6 such obtained shows that the enantiomeric centre was totally inverted during the substitution when compared to the enantiomeric purity of the alcohol (R)-3.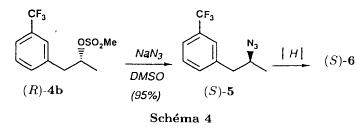
The reductive amination of the amine 6 in presence of acetaldehyde is known for a long time [28]. It was used recently in the works listed in the introduction [7, 8]. On our part, we propose another synthetic route for fenfluramine (S)-1 starting from the azide (S)-5 which does not go via the primary amine (S)-6 (Schema 5).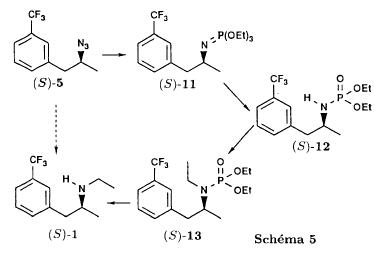
The reaction of Staudinger, reacting a stoichiometric quantity of triethylphosphite on the azide (S)-5 in THF at room temperature [29], gives quantitative yields of the phosphorimide 11 in 48 hours. It’s total conversion into the phosphoramide 13, by reacting with ethyl iodide [30] could not be realised [9]. We always obtained different mixtures of the phosphoramides 12 and 13 (referential compounds prepared from the amines 6 and 1). We also noted that the phosphorimide 11 can’t be isolated. When the solvent is evaporated, a partly transformation into the phosphoramide 12 takes place. This transformation is completed in less then 2h by simple heating to 100°C under argon after evaporation of the solvent. Because the phosphorimides are strongly basic compounds, we believe that an intramolecular arrangement of the phosphorimide, pictured in Schema 6, takes place.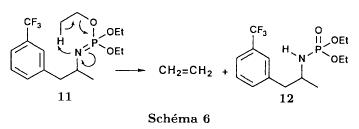
Having the phosphoramide 12, we investigated the alkylation into the phosphoramide 13 in DMF at room temperature [31, 32]: one deprotonates with sodium hydride then alkylates with diethyl sulfate. After treatment with hydrogen bromide [33], one obtains fenfluramine 1 with a yield of 85% and a purity of 97% (GC).
With the goal of simplifying the reaction scheme by avoiding the isolation of the intermediates we have again studied the transformation 5 -> 11 -> 12 in DMF (Schema 7). First, we noted that the reaction of Staudinger can be directly realised in this solvent. Thereafter we pinned down the transformation of the phosphorimide 11 into the phosphoramide 12 by reaction with water [34]. One then proceeds as described above. The transformation is thus performed without isolation of a single intermediate with a yield of 83%.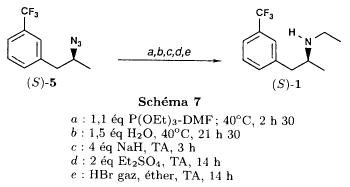
HPLC analysis on silica column of the camphanyl derivate of the amine (S)-1 [22] shows that the optical centre is conserved during the whole transformation.
Synthesis via the intermediate 2-chloro-1-[3-(trifluoromethyl)phenyl]propane 14
The yeast reduction of the ketone 2 gives the alcohol (S)-3, of which the authors have inverted the configuration to get the pharmacological active S enantiomer of fenfluramine [7, 8]. Independent research, using the epoxidation method of Sharpless [9, 35] lead us too to the alcohol (S)- 3 which we tried to convert into fenfluramine (S)-1 using a different method. The reaction scheme we kept uses the chloride 14 and proceeds via two inversions of the optical centre (scheme 8). Not owning enough alcohol (S)-3 during the studies, we tested the principle starting with the alcohol (R)-3, produced earlier, and studied the transformation into the azide (R)-5 (scheme 8), the latter being able to lead to (R)-fenfluramine using different methods, like the one outlined above: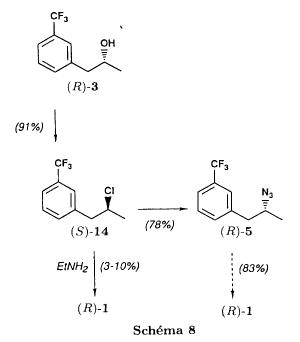
It is well known that the action of thionylchloride on an optically active alcohol gives the corresponding chlorine derivate, with inversion of the configuration in presence of bases and with retention of the configuration in the other case. We have performed the reaction with a catalytical amount of pyridine. One thus obtains the chloride (S)-14 with 91% yield and a purity of 91% (GC): it contains 9% of the elimination products 9, 10Z and 10E which are not separable by chromatography on silica.
The direct substitution of the chloride 14 with ethylamine with similar conditions to those used for the mesylate (R)-4b (EtNH2, DMSO, 110°C, 5h30 or EtNH2 (solvent and reactant), 140°C, 5h), gives mainly the elimination products. The yield of fenfluramine is below 10%.
By action of sodium azide in DMSO, on the other hand, one obtains the azide (R)-5 with a yield of 78%, the elimination products formed here or in the last step can be removed by chromatography on silica. HPLC analysis on silica of the camphanyl derivate of the amine (R)-6 [22] obtained by catalytic reduction of the azide (R)-5 has confirmed the double inversion without racemisation after comparison with the starting alcohol (R)-3. Then the fenfluramine (R)-1 is prepared without racemisation with a 83% yield starting from the azide (R)-5 like detailed above.
This procedure with two inversions allows to transform the alcohol 3 in the azide 5 with the same configuration in two steps with a global yield (non optimised) of 70% and without racemisation. It’s thus preferred over the recently published one [7, 8], which needs 5 steps for a lower global yield (55%) and in addition features an epimerisation of 10% [8]. It’s a promising way to fenfluramine (S)-1 starting from the alcohol (S)- 3.
References
- ^ “Fintepla- fenfluramine solution”. DailyMed. Retrieved 8 January 2021.
- ^ Jump up to:a b c d e f g h i j k l m n o p q “FDA Approves New Therapy for Dravet Syndrome”. U.S. Food and Drug Administration (FDA) (Press release). 25 June 2020. Retrieved 25 June 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Jump up to:a b c d “Fintepla EPAR”. European Medicines Agency. Retrieved 8 January2021.
- ^ Jump up to:a b c d Richard C. Dart (2004). Medical Toxicology. Lippincott Williams & Wilkins. pp. 874–. ISBN 978-0-7817-2845-4. Archived from the original on 2018-05-09.
- ^ Jump up to:a b James O’Donnell (Pharm. D.); Gopi Doctor Ahuja (2005). Drug Injury: Liability, Analysis, and Prevention. Lawyers & Judges Publishing Company. pp. 276–. ISBN 978-0-913875-27-8.
- ^ Integrating the Neurobiology of Schizophrenia. Academic Press. 27 February 2007. pp. 142–. ISBN 978-0-08-047508-0.
- ^ The Pharmacology of Corticotropin-releasing Factor (CRF). Effects on Sensorimotor Gating in the Rat. 2006. pp. 12–. ISBN 978-0-549-53661-1.
- ^ Christian P. Muller; Barry Jacobs (30 December 2009). Handbook of the Behavioral Neurobiology of Serotonin. Academic Press. pp. 630–. ISBN 978-0-08-087817-1.
- ^ Vickers SP, Dourish CT, Kennett GA (2001). “Evidence that hypophagia induced by d-fenfluramine and d-norfenfluramine in the rat is mediated by 5-HT2C receptors”. Neuropharmacology. 41 (2): 200–9. doi:10.1016/s0028-3908(01)00063-6. PMID 11489456. S2CID 23374227.
- ^ Roth, B. L. (2007). “Drugs and Valvular Heart Disease”. New England Journal of Medicine. 356 (1): 6–9. doi:10.1056/NEJMp068265. PMID 17202450.
- ^ Rothman, R. B.; Baumann, M. H. (2009). “Serotonergic Drugs and Valvular Heart Disease”. Expert Opinion on Drug Safety. 8 (3): 317–329. doi:10.1517/14740330902931524. PMC 2695569. PMID 19505264.
- ^ Jump up to:a b Dahl, C. F.; Allen, M. R.; Urie, P. M.; Hopkins, P. N. (2008). “Valvular Regurgitation and Surgery Associated with Fenfluramine Use: An Analysis of 5743 Individuals”. BMC Medicine. 6: 34. doi:10.1186/1741-7015-6-34. PMC 2585088. PMID 18990200.
- ^ Jump up to:a b c d e f g h i j k l m Donald G. Barceloux (3 February 2012). Medical Toxicology of Drug Abuse: Synthesized Chemicals and Psychoactive Plants. John Wiley & Sons. pp. 255–262. ISBN 978-1-118-10605-1. Archived from the original on 9 May 2018.
- ^ Mann SC, Caroff SN, Keck PE, Lazarus A (20 May 2008). Neuroleptic Malignant Syndrome and Related Conditions. American Psychiatric Pub. pp. 111–. ISBN 978-1-58562-744-8.
- ^ Jump up to:a b c d e f g h i Rothman RB, Clark RD, Partilla JS, Baumann MH (2003). “(+)-Fenfluramine and its major metabolite, (+)-norfenfluramine, are potent substrates for norepinephrine transporters”. J. Pharmacol. Exp. Ther. 305 (3): 1191–9. doi:10.1124/jpet.103.049684. PMID 12649307. S2CID 21164342.
- ^ Jump up to:a b c d e f g Setola V, Hufeisen SJ, Grande-Allen KJ, Vesely I, Glennon RA, Blough B, Rothman RB, Roth BL (2003). “3,4-methylenedioxymethamphetamine (MDMA, “Ecstasy”) induces fenfluramine-like proliferative actions on human cardiac valvular interstitial cells in vitro”. Mol. Pharmacol. 63 (6): 1223–9. doi:10.1124/mol.63.6.1223. PMID 12761331. S2CID 839426.
- ^ Nestler, E. J. (2001). Molecular Neuropharmacology: A Foundation for Clinical Neuroscience. McGraw-Hill.
- ^ Giuseppe Di Giovanni; Vincenzo Di Matteo; Ennio Esposito (2008). Serotonin-dopamine Interaction: Experimental Evidence and Therapeutic Relevance. Elsevier. pp. 393–. ISBN 978-0-444-53235-0.
- ^ Fitzgerald LW, Burn TC, Brown BS, Patterson JP, Corjay MH, Valentine PA, Sun JH, Link JR, Abbaszade I, Hollis JM, Largent BL, Hartig PR, Hollis GF, Meunier PC, Robichaud AJ, Robertson DW (2000). “Possible role of valvular serotonin 5-HT(2B) receptors in the cardiopathy associated with fenfluramine”. Mol. Pharmacol. 57 (1): 75–81. PMID 10617681.
- ^ Jump up to:a b c Rothman RB, Baumann MH, Dersch CM, Romero DV, Rice KC, Carroll FI, Partilla JS (January 2001). “Amphetamine-type central nervous system stimulants release norepinephrine more potently than they release dopamine and serotonin”. Synapse. 39 (1): 32–41. doi:10.1002/1098-2396(20010101)39:1<32::AID-SYN5>3.0.CO;2-3. PMID 11071707.
- ^ Rothman RB, Baumann MH (2002). “Therapeutic and adverse actions of serotonin transporter substrates”. Pharmacol. Ther. 95 (1): 73–88. doi:10.1016/s0163-7258(02)00234-6. PMID 12163129.
- ^ Connolly, H. M.; Crary, J. L.; McGoon, M. D.; Hensrud, D. D.; Edwards, B. S.; Edwards, W. D.; Schaff, H. V. (1997). “Valvular Heart Disease Associated with Fenfluramine-Phentermine”. New England Journal of Medicine. 337 (9): 581–588. doi:10.1056/NEJM199708283370901. PMID 9271479.
- ^ Weissman, N. J. (2001). “Appetite Suppressants and Valvular Heart Disease”. The American Journal of the Medical Sciences. 321 (4): 285–291. doi:10.1097/00000441-200104000-00008. PMID 11307869. S2CID 46466276.
- ^ FDA September 15, 1997. FDA Announces Withdrawal Fenfluramine and Dexfenfluramine (Fen-Phen) Archived 2013-07-19 at the Wayback Machine
- ^ “Drugs banned in India”. Central Drugs Standard Control Organization, Dte.GHS, Ministry of Health and Family Welfare, Government of India. Archived from the original on 2015-02-21. Retrieved 2013-09-17.
- ^ “FDA Approves FINTEPLA (fenfluramine) for the Treatment of Seizures Associated with Dravet Syndrome” (Press release). Zogenix Inc. 25 June 2020. Retrieved 25 June 2020 – via GlobeNewswire.
- ^ “Fenfluramine Orphan Drug Designations and Approvals”. U.S. Food and Drug Administration (FDA). 24 December 1999. Retrieved 25 June 2020.
- ^ “Fenfluramine Orphan Drug Designations and Approvals”. U.S. Food and Drug Administration (FDA). 24 December 1999. Retrieved 25 June 2020.
- ^ “Fintepla: Pending EC decision”. European Medicines Agency (EMA). 16 October 2020. Retrieved 16 October 2020. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ Jump up to:a b John Calvin M. Brust (2004). Neurological Aspects of Substance Abuse. Butterworth-Heinemann. pp. 117–. ISBN 978-0-7506-7313-6.
- ^ United States. Congress. Senate. Select Committee on Small Business. Subcommittee on Monopoly and Anticompetitive Activities (1976). Competitive problems in the drug industry: hearings before Subcommittee on Monopoly and Anticompetitive Activities of the Select Committee on Small Business, United States Senate, Ninetieth Congress, first session. U.S. Government Printing Office. pp. 2–.
- ^ Jump up to:a b Drug Addiction II: Amphetamine, Psychotogen, and Marihuana Dependence. Springer Science & Business Media. 27 November 2013. pp. 258–. ISBN 978-3-642-66709-1.
- ^ Drug Addiction II: Amphetamine, Psychotogen, and Marihuana Dependence. Springer Science & Business Media. 27 November 2013. pp. 249–. ISBN 978-3-642-66709-1.
- ^ “Zogenix ends enrolment in second Phase lll trial of ZX008”. Drug Development Technology. Kable. 1 February 2018. Archived from the original on 2018-02-04. Retrieved 3 February 2018.
- ^ “A Two-Part Study to Investigate the Dose-Ranging Safety and Pharmacokinetics, Followed by the Efficacy and Safety of ZX008 (Fenfluramine Hydrochloride) Oral Solution as an Adjunctive Therapy in Children ≥2 Years Old and Young Adults With Dravet Syndrome”. ClinicalTrials.gov. Archived from the original on 29 September 2017. Retrieved 9 May 2018.
- ^ “Zogenix drug cuts seizure rate in patients with severe epilepsy”. statnews.com. 29 September 2017. Archived from the original on 30 April 2018. Retrieved 9 May 2018.
Further reading
- Gustafsson BI, Tømmerås K, Nordrum I, Loennechen JP, Brunsvik A, Solligård E, Fossmark R, Bakke I, Syversen U, Waldum H (March 2005). “Long-term serotonin administration induces heart valve disease in rats”. Circulation. 111 (12): 1517–22. doi:10.1161/01.CIR.0000159356.42064.48. PMID 15781732.
- Welch, J. T.; Lim, D. S. (2007). “The Synthesis and Biological Activity of Pentafluorosulfanyl Analogs of Fluoxetine, Fenfluramine, and Norfenfluramine”. Bioorganic & Medicinal Chemistry. 15 (21): 6659–6666. doi:10.1016/j.bmc.2007.08.012. PMID 17765553.
External links
- “Fenfluramine”. Drug Information Portal. U.S. National Library of Medicine.
- “Fenfluramine hydrochloride”. Drug Information Portal. U.S. National Library of Medicine.
- Inchem.org – Fenfluramine hydrochloride
| Clinical data | |
|---|---|
| Trade names | Fintepla |
| Other names | ZX008 |
| AHFS/Drugs.com | Professional Drug Facts |
| MedlinePlus | a620045 |
| License data | US DailyMed: Fenfluramine |
| Pregnancy category | AU: B2 |
| Routes of administration | By mouth |
| ATC code | A08AA02 (WHO) N03AX26 (WHO) |
| Legal status | |
| Legal status | US: Schedule IV [1][2]EU: Rx-only [3] |
| Pharmacokinetic data | |
| Elimination half-life | 13–30 hours[4] |
| Identifiers | |
| IUPAC name[show] | |
| CAS Number | 458-24-2 |
| PubChem CID | 3337 |
| IUPHAR/BPS | 4613 |
| DrugBank | DB00574 |
| ChemSpider | 3220 |
| UNII | 2DS058H2CF |
| KEGG | D07945 C06996 |
| ChEBI | CHEBI:5000 |
| ChEMBL | ChEMBL87493 |
| CompTox Dashboard (EPA) | DTXSID4023044 |
| ECHA InfoCard | 100.006.616 |
| Chemical and physical data | |
| Formula | C12H16F3N |
| Molar mass | 231.262 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| Chirality | Racemic mixture |
| SMILES[hide]FC(F)(C1=CC(CC(C)NCC)=CC=C1)F | |
| InChI[hide]InChI=1S/C12H16F3N/c1-3-16-9(2)7-10-5-4-6-11(8-10)12(13,14)15/h4-6,8-9,16H,3,7H2,1-2H3 Key:DBGIVFWFUFKIQN-UHFFFAOYSA-N |
CLIP
http://www.inchem.org/documents/pims/pharm/pim938.htm
////////////Fenfluramine, 塩酸フェンフルラミン , dravet, AHR-3002, ZX-008, Fintepla
CCNC(C)CC1=CC(=CC=C1)C(F)(F)F.Cl
PATENT
https://patents.google.com/patent/US20170174613A1/en
- [0093]
Many general references providing commonly known chemical synthetic schemes and conditions useful for synthesizing the disclosed compounds are available (see, e.g., Smith and March, March’s Advanced Organic Chemistry: Reactions, Mechanisms, and Structure, Fifth Edition, Wiley-Interscience, 2001; or Vogel, A Textbook of Practical Organic Chemistry, Including Qualitative Organic Analysis, Fourth Edition, New York: Longman, 1978). - [0094]
Compounds as described herein can be purified by any purification protocol known in the art, including chromatography, such as HPLC, preparative thin layer chromatography, flash column chromatography and ion exchange chromatography. Any suitable stationary phase can be used, including normal and reversed phases as well as ionic resins. In certain embodiments, the disclosed compounds are purified via silica gel and/or alumina chromatography. See, e.g., Introduction to Modern Liquid Chromatography, 2nd Edition, ed. L. R. Snyder and J. J. Kirkland, John Wiley and Sons, 1979; and Thin Layer Chromatography, ed E. Stahl, Springer-Verlag, New York, 1969. - [0095]
During any of the processes for preparation of the subject compounds, it may be necessary and/or desirable to protect sensitive or reactive groups on any of the molecules concerned. This may be achieved by means of conventional protecting groups as described in standard works, such as J. F. W. McOmie, “Protective Groups in Organic Chemistry”, Plenum Press, London and New York 1973, in T. W. Greene and P. G. M. Wuts, “Protective Groups in Organic Synthesis”, Third edition, Wiley, New York 1999, in “The Peptides”; Volume 3 (editors: E. Gross and J. Meienhofer), Academic Press, London and New York 1981, in “Methoden der organischen Chemie”, Houben-Weyl, 4th edition, Vol. 15/1, Georg Thieme Verlag, Stuttgart 1974, in H.-D. Jakubke and H. Jescheit, “Aminosauren, Peptide, Protein”, Verlag Chemie, Weinheim, Deerfield Beach, and Basel 1982, and/or in Jochen Lehmann, “Chemie der Kohlenhydrate: Monosaccharide and Derivate”, Georg Thieme Verlag, Stuttgart 1974. The protecting groups may be removed at a convenient subsequent stage using methods known from the art. - [0096]
The subject compounds can be synthesized via a variety of different synthetic routes using commercially available starting materials and/or starting materials prepared by conventional synthetic methods. A variety of examples of synthetic routes that can be used to synthesize the compounds disclosed herein are described in the schemes below.
Example 11. Fenfluramine Nomenclature & Structure
- [0097]
Chemical Abstract Service (CAS) Registry Number (RN): 404-82-0 (HCl Salt), 458-24-2 (Parent Free Base) - [0098]
Chemical Name: N-ethyl-α-methyl-3-(trifluoromethyl)-benzeneethanamine hydrochloride (1:1). Other Names: Fenfluramine HCl, DL-Fenfluramine, (±)-Fenfluramine - [0099]
Structure of Hydrochloride Salt: - [0100]
Stereochemistry: Fenfluramine HCl has one chiral center and is being developed as the racemate and contains dexfenfluramine and levofenfluramine - [0101]
Molecular Formula of hydrochloride salt: C12H16F3N.HCl - [0102]
Molecular Mass/Weight: 267.72 g/mol
2. General Properties
- [0103]
Table 1 summarizes the chemical and physical properties of Fenfluramine HCl. - TABLE 1 General Properties of Fenfluramine HCl Drug Substance Property Result Appearance (color, White to off-white powder physical form) DSC (melting 170° C. (melt/sublimation) point)a TGA Onset 147° C. 0.03% at 150° C. 91% at 220° C. (evaporation) pKa (water) 10.15-10.38 Solubility (mg/mL) Resultant pH 25° C. 37° C. Solubility pH 6.69 (water) 54.13 71.22 (Aqueous) pH 1.73 buffer 25.34 53.68 pH 3.43 buffer 29.50 61.97 pH 6.41 buffer 37.42 95.60 0.9% NaCl (water) 22.98 — Solvent Solubility 25° C. (mg/mL) Solubility (Organic Ethanol 150 Solvents) Dichloromethane 30-35 Ethyl Acetate, 1-5 mg Tetrahydrofuran, Toluene, Acetonitrile UV Absorption Maxima: 210, 265 nm Solution pH (water) 6.69 Hygroscopicity @30% RH: ~0.05% (Dynamic Vapor @60% RH: ~0.07% Sorption (DVS) @90% RH: ~0.20%a) Polymorphism Fenfluramine HCl has been consistently isolated as a single crystalline Form 1 as determined by DSC and x-ray powder diffraction (XRPD) Solvation/Hydration Fenfluramine HCl is isolated as a nonhydrated, nonsolvated solid Solution Stability 8 weeks @ pH 6.7 phosphate buffer medium at 40° C. and 60° C. using concentrations of 0.5, 2.5 and 5.0 mg/ml. All conditions, no new impurities >0.1% by HPLC. Solid Stability 8 weeks @ 40° C., 60° C. and 80° C. 7 days at 150° C. All conditions, no new impurities >0.1% by HPLC.
3. Synthesis of Fenfluramine Drug Substance
- [0104]
Scheme 3.1 shows a 2-step route of synthesis used to manufacture initial clinical supplies of Fenfluramine HCl from ketone (2). The batch size is 4 kg performed in laboratory glassware (kilo lab). No chromatography is required and the process steps are amenable to scale-up. In process 1 there is one isolated intermediate Fenfluramine Free Base (1) starting from commercially supplied 1-(3-(trifluoromethyl)phenyl) acetone (Ketone 2). All steps are conducted under cGMPs starting from Ketone (2). - [0105]
Scheme 3.2 shows a 4-step route of synthesis to Fenfluramine HCl that can be used for commercial supply. Route 2 utilizes the same 2-step process used by Route 1 to convert Ketone (2) to Fenfluramine HCl with the exception that Ketone (2) is synthesized under cGMP conditions starting from 3-(Trifluoromethyl)-phenyl acetic acid (Acid 4). Bisulfate Complex (3) is an isolatable solid and can be purified before decomplexation to Ketone (2). In-situ intermediates which are oils are shown in brackets. Batch sizes of 10 Kg are performed. Commercial batch sizes of 20 kg are performed in fixed pilot plant equipment. Steps 1-2 of Scheme 3.2 to manufacture Ketone (2) have been demonstrated on a 100 g scale to provide high purity ketone (2) of >99.8% (GC & HPLC). Conversion of Ketone (2) to Fenfluramine using either Route 1 or 2 has provided similar purity profiles. - Starting materials are designated by enclosed boxes. Bracketed and non bracketed compounds respectively indicate proposed in-situ and isolated intermediates. NMI=N-Methyl Imidazole.
4.1. Narrative Description (Route 1)
- [0106]
Step 1: Reductive Amination (Preparation of Fenfluramine Free Base 1) - [0107]
A solution of ethylamine, water, methanol, and 1-(3-(trifluoromethyl)phenyl) acetone (Ketone 2) was treated with sodium triacetoxyborohydride and stirred for 16 h at 25° C. at which time HPLC analysis (IPC-1; In Process Control No. 1) showed the reaction to be complete and sodium hydroxide solution was added until pH>10. Toluene was added and the phases separated, and the aqueous phase (IPC-2) and organic phase (IPC-3) are checked for remaining Fenfluramine and Fenfluramine alcohol and the organic phase was reduced. Purified water was added and the pH adjusted to <2 using conc. HCl and the phases were separated. The aqueous phase was washed with toluene and the toluene phase (IPC-4) and the aqueous phase (IPC-5) was checked for Fenfluramine and Fenfluramine alcohol content. The aqueous phase containing product is pH adjusted to >10 using sodium hydroxide solution. The basic aqueous phase was extracted with MTBE until removal of Fenfluramine from the aqueous phase was observed by HPLC (<0.5 mg/ml) (IPC-6). The organic phase was dried over sodium sulfate and filtered. The filtrate was concentrated in vacuo to give the intermediate product Fenfluramine Free Base 1 as a pale yellow oil tested per specifications described herein which showed by NMR the material to contain 2.93% toluene giving an active yield of 88.3% with a purity of 98.23% by HPLC (0.67% Fenfluramine alcohol). - [0108]
Step 2: Salt Formation (Preparation of Fenfluramine HCl) - [0109]
To a flask was charged ethanol and acetyl chloride. The solution was stirred slowly overnight before ethyl acetate was added. The HCl in ethyl acetate solution formed was polish filtered into a clean carboy and retained for later use. To a vessel was added Fenfluramine free base 1 and MTBE. The Fenfluramine solution in MTBE was collected in two carboys before the vessel was cleaned and checked for particulate residue. The Fenfluramine solution was polish filtered into a vessel and cooled and HCl in ethyl acetate solution was added giving a final pH of 6-7. The batch was stirred for 1 h and filtered. The product was dried under vacuum at 40° C. The product (96.52% yield) was tested per IPC-7 had a purity of 99.75% by HPLC and GC headspace analysis showed MTBE (800 ppm) and EtOAc (150 ppm) to be present. The product was then tested per specifications shown herein.
4.2. Narrative Description (Route 2)
- [0110]
Step 1: Preparation of Ketone Bisulfite Adduct - [0111]
Procedure: Charge acetic anhydride, (2.8 vol, 3.0 wt, 5.0 eq.) to a vessel and commence stirring. Cool the solution to −5 to 5° C., targeting −4° C. Charge 1-methylimidazole, (0.2 vol, 0.21 wt, 0.5 eq.) to the mixture at −5 to 5° C. Caution: very exothermic. If necessary, adjust the temperature to 0 to 5° C. Charge ZX008 acid, (1.00 wt, 1.0 eq.) to the mixture at 0 to 5° C. Caution: exothermic. Stir the mixture at 0 to 5° C. until ≦2.1% area ZX008 acid by HPLC analysis, typically 7 to 9 hours. Charge 15% w/w sodium chloride solution (2.0 vol) to the mixture at 0 to 5° C., 60 to 90 minutes. Caution: very exothermic which will be slightly delayed. Warm the mixture to 18 to 23° C. over 45 to 60 minutes and continue stirring for a further 30 to 45 minutes at 18 to 23° C. Charge TBME, (5.0 vol, 3.7 wt) to the mixture and stir for 10 to 15 minutes at 18 to 23° C. Separate the aqueous layer and retain the organic layer. Back-extract the aqueous layer with TBME, (2×3.0 vol, 2×2.2 wt) at 18 to 23° C. retaining each organic layer. Adjust the pH of the combined organic layer to pH 6.5 to 9.0, targeting 7.0 by charging 20% w/w sodium hydroxide solution (5.3 to 8.3 vol) at 18 to 23° C. Caution: exothermic. Separate the aqueous layer and retain the organic layer. Wash the organic layer with 4% w/w sodium hydrogen carbonate solution (2×3.0 vol) at 18 to 23° C. Determine the residual ZX008 acid content in the organic layer by HPLC analysis, pass criterion ≦0.10% area ZX008 acid. Wash the organic layer with purified water, (2×3.0 vol) at 18 to 23° C. Concentrate the organic layer under reduced pressure to ca. 2 vol at 40 to 45° C., targeting 43° C. - [0112]
Determine the w/w assay of ZX008 ketone (WIP) in the mixture by 1H-NMR analysis for information only and calculate the contained yield of ZX008 ketone (WIP) in the mixture. Note: This step can be removed from the process since the process is robust and consistently delivers 80 to 90% th yield. The achieved yield was factored into the charges of the subsequent steps. - [0113]
Charge n-heptane, (4.0 vol, 2.7 wt) to the mixture at 40 to 45° C., targeting 43° C. Concentrate the mixture to ca. 2 vol at 40 to 45° C., targeting 43° C. Determine the TBME content in the mixture by 1H-NMR analysis, (pass criterion ≦5.0% w/w TBME vs. ZX008 ketone). Charge n-heptane, (2.4 vol, 1.6 wt) at 40 to 45° C., targeting 43° C., vessel A. To vessel B, charge sodium metabisulfite, (0.82 wt, 0.88 eq.) at 18 to 23° C. To vessel B, charge a solution of sodium hydrogen carbonate, (0.16 wt, 0.4 eq.) in purified water, code RM0120 (2.0 vol) at 18 to 23° C. followed by a line rinse with purified water, code RM0120 (0.4 vol) at 18 to 23° C. Caution: gas evolution. Heat the contents of vessel B to 40 to 45° C., targeting 43° C. Charge the contents from vessel A to vessel B followed by a line rinse with n-heptane, (0.8 vol, 0.5 wt) at 40 to 45° C., targeting 43° C. Stir the mixture for 1 to 1.5 hours at 40 to 45° C., targeting 43° C. Charge n-heptane, code RM0174 (3.2 vol, 2.2 wt) to the mixture with the temperature being allowed to cool to 18 to 45° C. at the end of the addition. Cool the mixture to 18 to 23° C. at approximately constant rate over 45 to 60 minutes. Stir the mixture at 18 to 23° C. for 1.5 to 2 hours. - [0114]
Sample the mixture to determine the residual ZX008 ketone content by 1H-NMR analysis, (pass criterion ≦10.0% mol, target 5.0% mol ZX008 ketone vs. ZX008 ketone bisulfite adduct). Filter the mixture and slurry wash the filter-cake with n-heptane, (2×2.0 vol, 2×1.4 wt) at 18 to 23° C. Dry the solid at up to 23° C. until the water content is <10.0% w/w water by KF analysis according to AKX reagent. At least 16 hours. Determine the w/w assay of the isolated ZX008 ketone bisulfite adduct by 1H-NMR analysis and calculate the contained yield of ZX008 ketone bisulfite adduct. - [0115]
Yields and Profiles: The yield for the stage 1 Demonstration batch is summarized Table below. Input: 1700.0 g uncorr., acid, 99.50% area (QC, HPLC), 2-isomer not detected, 4-isomer 0.02% area, RRT1.58 (previously not observed) 0.48% area as per the preparative method. The analytical data is summarized in Table 1A below. - TABLE 1A Table for isolated yields for step 1 Demonstration batch Corr. % area Reference Corr. Yield % w/w (HPLC, number Input Output (% th)** (1H-NMR)* QC) Comments Batch A1 1700.0 g 1500.1 g 89.1 45.0 —.— Crude ketone as TBME sol. Batch A2 1500.1 g 1716.1 77.8 76.0 98.15 Bisulfite adduct only 67.3 Overall product
- [0116]
Step 2: Preparation of Ketone - [0117]
Procedure: Charge toluene, (5.0 vol, 4.3 wt), and purified water, (5.0 vol) to the vessel and commence stirring. If necessary, adjust the temperature to 18 to 23° C. and charge ZX008 ketone bisulfite adduct, (1.00 wt corrected for % w/w assay) to the mixture at 18 to 23° C. Charge 20% w/w sodium hydroxide solution to the mixture at 18 to 23° C. adjusting the pH of the mixture to pH 8.0 to 12.0, targeting 9.0 (0.5 to 1.0 vol). - Separate the lower aqueous layer and retain the top organic layer. Wash the organic layer with purified water, (3.0 vol) at 18 to 23° C. Concentrate the organic layer under reduced pressure to ca. 2 vol at 45 to 50° C., targeting 48° C. Charge methanol, (5.0 vol, 4.0 wt) to the mixture at 45 to 50° C., targeting 48° C. Re-concentrate the mixture under reduced pressure to ca. 2 vol at 45 to 50° C., targeting 48° C. Repeat steps 7 and 8 once before continuing with step 9. Cool the mixture to 18 to 23° C. Clarify the mixture into a tared, suitably-sized drum followed by a methanol (1.0 vol, 0.8 wt) line rinse at 18 to 23° C. Determine the w/w assay of ZX008 ketone (WIP) in the mixture by 1H-NMR analysis and calculate the contained yield of ZX008 ketone (WIP) in the mixture. Determine the toluene content in the mixture by 1H-NMR analysis.
- [0118]
Yields and Profiles: The yield for the step 2 Demonstration batch is summarized in Table 1B below. Input: 1200.0 g corr. Ketone bisulfite adduct, 76.0% w/w assay (NMR, using DMB as internal standard in d6-DMSO), (1.00 eq, 1.00 wt corr. for w/w assay) for input calculation. - TABLE 1B Table for isolated yields for step 2 Demonstration batch % w/w % area Corr. Corr. Corr. Yield (1H- (HPLC, Input Output (% th) NMR)* QC) Comments 1200.0 g 858.15 g 108.3 25.5 99.31 Purified ketone
- [0119]
Step 3: Preparation of Fenfluramine HCl Crude - [0120]
Procedure: Charge the ZX008 ketone (corr. for assay, 1.00 wt, 1.00 eq. isolated as solution in MeOH in stage 2) to a vessel. Charge methanol, code RM0036 (5.0 vol, 4.0 wt) to the mixture at 18 to 23° C. Cool the solution to 0 to 5° C. Charge 70 wt % aqueous ethylamine solution (1.3 vol, 1.6 wt, 4.0 eq) to the mixture at 0 to 10° C., over 15 to 30 minutes, followed by a line rinse with methanol (1.0 vol, 0.8 wt). Warm the mixture to 15 to 20° C. and stir the mixture for a further 60 to 70 minutes at 15 to 20° C. Adjust the mixture to 15 to 18° C. if required, targeting 15° C. Charge sodium triacetoxyborohydride (2.4 wt, 2.25 eq.) to the mixture in approximately 10 portions, keeping the mixture at 15 to 20° C., targeting 17° C. Addition time 1.5 to 2 hours. Caution: Exothermic. Stir the mixture at 15 to 20° C. until complete by HPLC analysis, pass criterion ≦3.0% area ZX008 ketone, typically 2 to 3 hours. Adjust the pH of the mixture to pH>12 by charging 20% w/w aqueous sodium hydroxide solution (5.0 to 6.0 vol) to the mixture at 15 to 40° C. Addition time 10 to 30 minutes. Caution: Exothermic. If necessary, adjust the temperature to 18 to 23° C. Extract the mixture with toluene (3×3.0 vol, 3×2.6 wt) at 18 to 23° C., retaining and combining the top organic layer after each extraction. Wash the combined organic layer with purified water, (1.0 vol) at 18 to 23° C. Heat the mixture to 40 to 50° C., targeting 48° C. Concentrate the mixture under reduced pressure at constant volume maintaining ca. 5 vol by charging the organic layer at approximately the same rate as the distillation rate at 40 to 50° C., targeting 48° C. Cool the mixture to 18 to 23° C. Charge purified water (10.0 vol) to the mixture at 18 to 23° C. Adjust the pH of the mixture to 0.1<pH<1.5 at 18 to 23° C. by charging concentrated hydrochloric acid, 0.5 vol. Do not delay from this step until neutralization. - [0121]
Separate the layers at 18 to 23° C. retaining the bottom aqueous layer. Wash the aqueous layer with toluene, (3.0 vol, 2.6 wt) at 18 to 23° C. retaining the aqueous layer. Adjust the pH of the aqueous layer to pH>12 by charging 20% w/w sodium hydroxide solution at 18 to 23° C. 0.8 to 0.9 vol. Caution: Exothermic. Charge TBME, code RM0002 (2.0 vol, 1.5 wt) to the basic aqueous layer. Separate the layers at 18 to 23° C. retaining the top organic layer. Back-extract the aqueous layer with TBME (2×2.0 vol, 2×1.5 wt) at 18 to 23° C. retaining the organic layers. Wash the combined organic layer with purified water, (2×1.0 vol) at 18 to 23° C. Concentrate the combined organic layers under reduced pressure at 40 to 50° C., targeting 48° C. to ca. 3 vol. Determine the residual toluene content of the mixture by 1H-NMR analysis. Sample for determination of residual water content by KF analysis, AKX reagent. Charge TBME (8.7 vol, 6.4 wt) to the mixture at 40 to 50° C. Cool the solution to 0 to 5° C., targeting 2° C. Charge concentrated hydrochloric acid (0.54 vol, 0.46 wt) maintaining the temperature <15° C. Caution: Exothermic. Line rinse with TBME (1.0 vol, 0.7 wt). If necessary, adjust the temperature to 0 to 10° C. and stir the mixture at 0 to 10° C. for a further 2 to 3 hours. Filter the mixture and wash the filter-cake with TBME (2×4.4 vol, 2×3.3 wt) at 0 to 10° C. Dry the solid at up to 40° C. until the TBME content is <0.5% w/w TBME by 1H-NMR analysis. 4 to 8 hours. - [0122]
Yields and Profiles: The yield for the step 3 Demonstration batch is summarized in Table 1C below. Input: 856.8 g corr. Ketone, 44.2% w/w assay (NMR, using TCNB as internal standard in CDCl3), (1.00 eq, 1.00 wt corr. for w/w assay) for input calculation. FIG. 2 and Table 1D shows an exemplary HPLC chromatogram of a crude preparation of fenfluramine hydrochloride (210 nm UV absorbance). - TABLE 1C Table for isolated yields for step 3 Demonstration batch Corr. % area Reference Corr. Corr. Yield % w/w (HPLC, number Input Output (% th) (1H-NMR)* QC) Comments Batch A1 856.8 g 836.31 g 85.3 44.2 99.15 Fenfluramine free base (in situ intermediate) Batch A2 880.7 84.0 based 99.5 100.00 Fenfluramine•HCl on ketone crude (step 3 an bisulfite d 4.1) adduct (77.6 based on purified ketone)
- TABLE 1D Purity of crude fenfluramine hydrochloride by HPLC (see FIG. 2) Processed Channel Descr. DAD AU Ch 1 Sample 210, Bw 4 Peak Results USP USP USP Name RT RelRT Area Height Tailing Resolution Plate Count EP s/n % Area 1 NorFenfluramine 7.46 2 2-Fenfluramine 7.68 3 Fenfluramine 8.67 1.000 3789064 778178 1.7 70796 2549.8 99.15 4 4-Fenfluramine 8.95 5 11 34 1.308 6073 1449 1.2 23.5 215529 3 8 0.16 6 ZX008 acid 12.93 7 Fenfluramine alcohol 14.16 1.633 15266 2972 1.3 24.8 215040 8.7 0.40 8 ZX008 ketone 14.83 9 Fenfluramine acetamide 15.55 10 TOLUENE 15 75 11 15.92 1.836 4110 1122 2.7 0.11 12 16.60 1.915 6861 1630 1.5 451209 4.3 0.18 Sum 3821374 100.00
- [0123]
Step 4.2: Crystallization of Fenfluramine Hydrochloride - [0124]
Procedure: Charge Fenfluramine.HCl (crude) (1.00 wt, 1.0 eq.) and TBME (10.0 vol, 7.4 wt) to the vessel and commence stirring. Heat the suspension to reflux (50 to 58° C.). Charge ethanol (5.0 vol, 3.9 wt) maintaining the temperature at 50 to 58° C. Addition time 20 minutes. Stir at 50 to 58° C. for 5 to 10 minutes and check for dissolution. Stir the solution at 50 to 58° C. for 5 to 10 minutes, targeting 54 to 58° C. Clarify the reaction mixture through a 0.1 μm in-line filter at 54 to 58° C., followed by a line rinse with TBME (1 vol, 0.7 wt). Cool the solution to 48 to 50° C. Charge Fenfluramine HCl, code FP0188 (0.01 wt). Check for crystallization. Allow the suspension to cool to 15 to 20° C., target 17° C. over 5 to 5.5 hours at an approximately constant rate. Stir the mixture at 15 to 20° C., target 17° C. for 2 to 3 hours. Filter the mixture and wash the filter-cake with clarified TBME (2×3.0 vol, 2×2.2 wt) at 5 to 15° C. Dry the solid at up to 40° C. until the TBME content is <0.5% w/w TBME and the ethanol content is <0.5% w/w EtOH by 1H-NMR analysis. 4 to 8 hours. Determine the w/w assay of the isolated Fenfluramine.HCl by 1H-NMR analysis. - [0125]
Yields and Profiles: The yield for the stage 4 Demonstration batch is summarized in Table 1E below. Input: 750.0 g uncorr. Fenfluramine HCl crude (1.00 eq, 1.00 wt uncorr.) for input calculation. FIG. 3 shows an exemplary HPLC chromatogram of a crystallized fenfluramine hydrochloride sample (210 nm UV absorbance). - TABLE 1E Table for isolated yields for stage 4 Demonstration batch Uncorr. Uncorr. Uncorr. Yield HPLC (% area, Input Output (% th) QC) Comments 750.0 g 608.0 81.1 100.00* Fenfluramine•HCl
5. In-Process Controls
- [0126]
Table 2 summarizes the in-process controls (IPCs) by IPC number as cited in the narrative procedures above used for Process 1. - TABLE 2 In-Process Controls Performed during Process 1 Critical IPC Synthesis Process No. Step Sample Description Method Acceptance Criteria 1 1 Reaction Reaction HPLC NMT 3.0% Ketone (1) Mixture Completion 2 1 Extraction Purity HPLC Report percent Aqueous Fenfluramine Free Base and Phase Fenfluramine Alcohol 3 1 Extraction Purity HPLC Report percent Organic Fenfluramine Free Base and Phase Fenfluramine Alcohol 4 1 Extraction Purity HPLC Report percent Organic Fenfluramine Free Base and Phase Fenfluramine Alcohol 5 1 Extraction Purity HPLC NLT 98.0% Fenfluramine Aqueous HCl Phase LT 1.0% Fenfluramine Alcohol 6 1 Extraction Purity HPLC Report percent result of Aqueous Fenfluramine HCl Phase Fenfluramine Alcohol 7 2 Reaction Purity 1H-NMR Residual Solvents by 1H- Mixture NMR Ethanol NMT 0.50% w/w Ethyl Acetate NMT 0.50% w/w Methanol NMT 0.50% w/w Toluene NMT 0.50% w/w MTBE NMT 0.50% w/w
6. Starting Materials
- [0127]
This section provides information and specification controls for the starting materials used to produce clinical supplies of fenfluramine per the routes shown herein. - TABLE 3 Starting Materials via the Route 1 Chemical Name Code [CAS. No.] Name Structure Source Step 1-(3- (Trifluoromethyl)- phenylacetone [21906-39-8] Ketone (1)Fluorochem 1 Ethyl Amine Ethyl EtNH2 Alfa Aesar 1 (70% in water) Amine [75-04-7]
- TABLE 4 Starting Materials via Route 2 Chemical Name Code [CAS. No.] Name Structure Source Step 3-(Trifluoromethyl)- phenylacetic acid [351-35-9] Acid (1a)To be determined 1 Acetic Anhydride [108-24-7] Acetic AnhydrideVarious 1 Ethyl Amine Ethyl EtNH2 Various 3 (70% in water) Amine [75-04-7]
- [0128]
Table 5 provides a list of the intermediates for the Route 2 synthesis. Both routes share the same intermediate Fenfluramine Free Base (1). Fenfluramine Free Base (1) was treated as an isolated intermediate in the Route 1 process however the Route 2 process uses fixed equipment where both Ketone (2) and Fenfluramine Free Base 1, both non-isolatable oils, are telescoped as a solution and controlled as in-situ intermediates. The Bisulfate Complex (3) is isolated as a solid thus is amenable to treatment as an isolated intermediate and released as such. Crude Fenfluramine HCl can be isolated as an intermediate before recrystallization. - [0129]
A Specification and Testing Strategy for Intermediates is used. Additional tests and acceptance criteria are be added based upon review of data from the primary stability batches and process validation critical parameter studies. Analytical reference standards are used in full characterization of each intermediate. HPLC methods to determine assay and impurities are the same as the drug substance release method and are validated for Accuracy, Precision: Repeatability, Intermediate Precision, Selectivity/Specificity, Detection limit, Quantitation limit, Linearity, Range, and Robustness. - TABLE 5 In-Situ and Isolated Intermediates Chemical Name [CAS No] Code Name Step No. Control Structure Bisulfate Complex of Ketone 1 Bisulfate Complex (3) Step 1 Isolated (Solid)1-(3- (Trifluoromethyl)- phenylacetone [21906-39-8] Ketone (2) Step 2 In-Situ (oil)Fenfluramine Free Base [458-24-2] Fenfluramine Free Base (1) Step 3 In-Situ (oil)Fenfluramine HCl [404-82-0] Crude Fenfluramine HCl Step 4 Isolated (Solid)
7. Characterization
- [0130]
Physiochemical Characteristics of Drug Substance. - [0131]
Fenfluramine HCl is developed as a single polymorph Form 1. A polymorphism and pre-formulation study has been conducted. Under a wide range of solvents and conditions crystalline material is produced of the same polymorph Form 1 based on a well-defined XRPD pattern and a consistent reproducible endotherm by DSC analysis. A summary of the chemophysical properties of Fenfluramine HCl from this study is provided below. Tabulated data includes example diffractograms, DSCs, and micrographs. - [0132]
The input Fenfluramine HCl (from precipitative isolation) was characterized to provide reference data and also to determine if the salt was of the same form as that identified from previous salt formations. The XRPD pattern of the salt reveals a crystalline solid that visually matches the reflection patterns obtained from formal crystallization of Fenfluramine HCl and has been arbitrarily termed Form 1. Comparison of the μATR-FTIR data for the salt from various batches gave profiles that had a 99.95% match. - [0133]
Thermal data analysis matched previous data obtained with only one major endotherm on the DSC thermograph peaking at 172.3° C. that matches the beginning of potential decomposition shown in a TGA thermograph. This also matches the reported melting point quoted for the reference standard. - [0134]
Isolation of the amorphous form has been shown to be difficult, with attempts using three common methods (rapid solvent evaporation, anti-solvent precipitation and lyophilization) all yielding highly crystalline solids that very closely share the same XRPD pattern of the input Form 1. - [0135]
Stability analysis of the salt after one week at 40° C./0% RH, three weeks at 40° C./75% RH, and under photostability conditions revealed that the input Form 1 has been maintained with no new impurities observed at 0.1% threshold. - [0136]
Results from DSC heat cycling analysis of Fenfluramine HCl are comparable to results generated when the material was held at 170° C. No crystallization event was noted and the amorphous was not generated but rather Form 1 was returned. - [0137]
Holding Fenfluramine HCl at approximately 170° C. for several hours causes a melt and evaporation event to take place with recombination and cooling to provide a white solid. Analysis of the white solid by XRPD, DSC and 1H NMR indicates no change in chemical or physical form, purity, or dissociation. - [0138]
Forced degradation studies carried out have proven Fenfluramine HCl to be stable under a range of conditions. Thermal modulation of Fenfluramine HCl repeatedly yielded the input Form 1.
8. Impurities
- [0139]
Impurities in a drug substance can be organic impurities (process impurities or drug substance-related degradants), inorganic impurities (salt residues or metals) and residual solvents; some of these impurities must be evaluated as to whether or not they are genotoxic agents. These impurities are taken into consideration and controlled in Fenfluramine HCl preparation by using either compendia or validated analytical methods per the specifications or by separate “for information only” testing. The following sections address the actual and potential impurities in Fenfluramine HCl. - [0140]
Actual Impurities and the Qualification of Synthesis Batch - [0141]
No impurities reported in cGMP drug substance batches intended for use in humans have exceeded the ICHQ3A qualification thresholds of 0.15% (Table 8). All impurities >0.1% are identified and handled as described in ICH Q3A unless they are genotoxic impurities. - [0142]
Process Impurities - [0143]
Table 6 lists the known potential impurities arising from the route of synthesis. All of these impurities are controlled to below ICHQ3A qualification threshold of 0.15% by either process changes and/or control of starting material input purities. - TABLE 6 Fenfluramine HCl Known Potential Process Impurities (Route 1) Observed Observed in in Development cGMP Name PLC Batches Batches [Cas. No.] Source (RRT) ≧0.10%1) ≧0.10%1) Ketone (2) Starting RRT No No [351-35-9] Material or 0.89 Intermediate Fenfluramine By-product RRT Yes No Alcohol 1.60 [621-45-4] Norfenfluramine By-product RRT Yes Yes [1886-26-6] 1.67 2-Fenfluramine Starting RRT No No [172953-70-7] Material 0.89 (isomer) 4-Fenfluramine Starting RRT Yes Yes [1683-15-4] Material 1.02 (isomer) N-(3- By-product RRT Yes Yes (trifluoromethyl)- 0.53-0.57 benzyl)ethanamine [90754-95-3] 1)ICH Q3A Identification threshold. The Reporting threshold (LOQ) for the HPLC method is 0.05%.
- [0144]
Degradation Impurities - [0145]
No change in impurity profile is observed upon long-term storage based on forced degradation studies under the ICH Q1A(R2) conditions of heat (solid, solution), acid, base, oxidizing, and ICH Q1B photostability conditions (solid, solution). Fenfluramine HCL is stable for 7 days as a solid at 150° C. (99.90 parent area %), as a solution in water-acetonitrile at 70° C. (99.73 parent area %), as a solution in acid, base, or photosensitizing conditions at ambient. Only oxidizing conditions (peroxide conditions) produced degradation of Fenfluramine HCl to 94.42% after 1 day producing several new related substances at −1% each consistent by LC-MS with +16 oxidation by-products - [0146]
Organic Volatiles/Residual Solvents - [0147]
Table 11 in the Batch Analysis section summarizes the solvents used in the process and the resulting amounts found in drug substance. All solvents used in the GMP steps are controlled at ICH Q3A limits using a suitably qualified Head-Space (HS) GC method. - [0148]
Inorganic Impurities - [0149]
Heavy Metals conform to either USP <231> or ICP method USP <233> as well as ICH Q3D. - [0150]
Genotoxic Impurities - [0151]
The ICH guidelines Q3A and Q3B are not sufficient to provide guidance on impurities that are DNA-reactive. The European Medicines Agency (EMA) guideline (2006) “Guideline on the Limits of Genotoxic Impurities” (EMA 2006) and the ICH Guideline M7 (2014) “Assessment and Control of DNA Reactive (Mutagenic) Impurities in Pharmaceuticals to Limit Potential Carcinogenic Risk” (ICH Guideline M7) are taken into consideration in controlling for potential genotoxic impurities. The diazonium route to prepare ketone (2) described in FIG. 5 has a disadvantage due to the potential formation of genotoxic intermediates shown as boxed compounds (e.g., N-hydroxyaryl, N-nitrosamine and Nitro compound). Muller et al. (Regulatory Toxicology and Pharmacology 44 (2006) 198-211) list potential functional alert groups that can be genotoxic. Safety guidances and regulations indicate that analysis of a process and identification of potential genotoxic agents, and control of such impurities at sub 10 parts per million levels is critical for safety. Often removal of such impurities and/or demonstrating their absence is costly and time consuming and sometimes difficult to achieve technically. For these reasons, selecting synthetic routes that circumvent the potential for such toxic intermediates is important. Because of the potential problems with the diazo route discussed above, as well as potential safety issues using diazo (shock-sensitive) intermediates, as well as the lower purity profiles with this route, this route is less preferred than the preferred route to ketone (2) starting from Nitrile (5). This route produces no potential genotoxic agents and leads to high purity Ketone (2) after isolation by distillation or via the bisulfite salt adduct—hydrolysis sequence. - [0152]
Additionally, attempts to remove isomer by-products present in commercial supplies of Aniline were unsuccessful whereas crystallization the Acid (4) resulting from hydrolysis of the nitrile (5) provides crystalline Acid (4) which can be purified to remove isomers early in synthesis. Removing impurities and/or isomers early in a synthesis is preferred if it is known such impurities track to final product, as the need to crystallize a final product at the end of a synthesis is more costly in losses and impacts cost of goods more greatly than removing early in synthesis before raw materials are invested along the process. - TABLE 7 Potential Impurities in Fenfluramine Synthesis Synthesis Route No. Compound Route 1 Route 2 CAS. No. 1No Starting Material [351-35-9] 2Starting Material Intermediate [21906-39-8] 4No Intermediate Not Available 5Potential Impurity Potential Impurity [621-45-4] 6Potential Impurity Potential Impurity [1886-26-6] 7Potential Impurity Potential Impurity [172953-70-7] 8Potential Impurity Potential Impurity [1683-15-4] 9Potential Impurity Potential Impurity [90754-95-3]
- TABLE 8 Batch Analyses of Fenfluramine HCl Drug Substance Test Batch 1 Batch 2 Batch 3 Batch 4 Appearance* White solid White solid White solid White solid Identification: FTIR* a) a Conforms Conforms Identification: 1H-NMR Conforms Conforms Conforms Conforms Identification: 13C-NMR Conforms Conforms Conforms Conforms Identification: MS Conforms Conforms Conforms Conforms Purity (HPLC area %) 99.57 99.77 b) b Assay (w/w %)* 99.49 100.37 100.79 100.13 Anhydrous Basis (HPLC) Impurities 2-Fenfluramine ND ND ND ND (HPLC 4-Fenfluramine) 0.16 0.15 0.11 0.12 area %) Fenfluramine Alcohol ND ND ND ND 1-((3-trifluoromethyl)phenyl)acetone ND ND ND ND Acetamide 0.27 ND ND ND N-(3-(trifluoromethyl)- ND 0.08 0.07 0.13 benzyl)ethanamine (RRT 0.53-0.57) Total 0.43 0.23 0.19 0.25 Residual Solvents Methanol ND ND ND ND (GC): ppm Ethanol ND ND ND ND MTBE 597 844 472 800 Ethyl Acetate 115 164 79 150 Toluene 4 7 ND ND Residue on Ignition (w/w %) 0.01 0.02 0.04 ND Heavy Metals (as Pb) <10 ppm <10 ppm b b Heavy Metals ICP (ppm) As a a <0.1 <0.1 Cd a a 0.1 0.1 Hg a a <0.1 <0.1 Pb a a 0.2 <0.4 Co a a <0.1 0.1 Mo a a <0.1 <0.1 Se a a <0.1 <0.1 V a a <0.1 <0.1 Water Determination* 0.21 0.08 0.02 0.03 (Karl Fischer) Chloride content by titration 13.19 13.09 12.92 12.93 XRPD* Form 1 Form 1 Form 1 Form 1 Differential Scanning Onset 169.42° C. 169.23° C. 169.85° C. 168.70° C. Calorimetry (DSC)* Peak 172.82° C. 171.55° C. 172.22° C. 171.97° C. Particle Size % Volume mean (D) a 11 11 19 Malvern (μm) D10 a 1 1 1 D50 a 5 7 9 D90 a 17 26 32 Microbial Total aerobic a a LT 100 CFU/g LT 100 CFU/g Limits Tests* microbial Count USP <61> Total yeast and a a LT 100 CFU/g LT 100 CFU/g molds count USP <62> Absence of a a Absent Absent Pathogens a)These tests were added to the specifications recently thus only recent lots have been tested using this test. b)These tests have been dropped from the specifications thus only historical lots have been tested using this test.
Example 3Method for Hydrolysis of Nitrile (5) to Acid
- [0153]
- TABLE 9 Step Operation 1. Charge 3-(trifluoromethyl)phenyl acetonitrile (1.0 eq., 1.00 wt) and purified water (5.0 vol) to a vessel and commence stirring. 2. Dissolve sodium hydroxide (1.1 wt, 5.0 eq.) in purified water (4.0 vol) at up to 40° C. in a suitable make-up vessel. Caution very exothermic. 3. Charge the aqueous sodium hydroxide solution to the mixture from step 1 at up to 40° C. followed by a line rinse with purified water, code RM0120 (1.0 vol) at up to 40° C. 4. Heat the mixture to 75 to 85° C., target 80° C. over 1 to 2 hours. 5. Heat the mixture at 80° C. until ≦0.1% area nitrile by HPLC analysis, expected 4 to 6 hours. 6. Cool the mixture to 18 to 23° C. 7. Adjust the pH of the mixture to pH ≦2 by charging 6M HCl (expected 7.0 vol) to the mixture at 18 to 23° C. Caution exothermic. 8. Stir the mixture for 15 to 30 minutes at 18 to 23° C. 9. Filter and wash the filter-cake with purified water (2 × 5.0 vol) at 18 to 23° C. 10. Slurry wash the filter-cake with n-heptane, code RM (2 × vol) at 0 to 5° C. 11. Dry the isolated solid at up to 45° C. until the water content is ≦.0.2% w/w by KF analysis according to MET/AN/0163, AKX-reagent. 12. Crystallization of crude stage 1 acid (1.00 wt for input calculation) 13. Charge the crude stage 1 acid (1.00 wt), ethyl acetate (0.75 vol) and n-heptane (10.5 vol) to a vessel and commence stirring. 14. Heat the mixture to 50° C. to achieve dissolution. 15. Cool the mixture to 5° C. and age at 5° C. for at least 30 mins. 16. Filter and wash the filter-cake with n-heptane (2 × 5.0 vol). 17. Dry the isolated solid at up to 45° C. until the residual solvent content by 1H-NMR analysis is ≦.0% w/w EtOAc and ≦.0% w/w n-heptane. Expected yield: 60 to 90% th uncorr. 68 to 103% w/w Expected purity: 93.00 to 99% area by HPLC
Example 4Evaluation of Minor Components Formed During Dakin-West Reaction in Preparation of Ketone (2)
- [0154]
The impurities formed during the Dakin-West chemistry and their subsequent removal using the distillation or via isolation of the product ketone as the bisulfite salt are described. The two major impurities found are shown below. - [0155]
Table 10 shows a table of analytical data for crude Ketone (2) isolated from Dakin-West reaction before and after bisulfite purification. In entry 1 crude Ketone isolated directly from the Dakin-West step (pre-bisulfite treatment) is 61.66% purity (e.g. about 62%) and contains 1.98% (e.g., about 2%) and 4.64% (e.g., about 5%) respectively of impurities having RRTs 1.20 and 1.34, which are proposed to be the acetate and dimer impurities (e.g., depicted above), respectively. In entry 2 which is post bisulfite treatment these are other impurities are removed leading to an overall purity of 95.55% (e.g., about 96%). Other entries shown in Table 10 provide other examples of this impurity enhancement by bisulfite treatment of crude Dakin-West ketone. The last two entries use pure Fluorchem ketone as input to the salt formation step and re-isolation of ketone thus illustrating that the salt formation and re-isolation does not produce any impurities itself. Additionally use of bicarbonate extraction procedure during reaction workup provides an improvement in purity of the resulting composition as it serves to remove any unreacted acid. Crude Ketone (2) made by the Diazo route showed similar improvements in purity when treated with bisulfite and isolated. - TABLE 10 Analytical purity data for crude Ketone (2) isolated from Dakin-West reaction before and after bisulfite purification. RRT is relative retention time (min) in chromatogram. RRT 0.93 1.00 1.009 1.06 Entry 0.85 Aniline 0.95 0.99 Ketone 1.004 Nitrile 1.02 Acid 1.10 1.15 1.34 1.38 1 1.38 1.76 0.04 0.49 61.66 nd 0.29 0.29 0.26 1.98 0.66 4.64 0.14 2 0.82 nd nd nd 95.53 0.31 0.14 nd 0.23 0.01 0.10 0.43 0.27 3 nd nd nd nd 77.82 nd nd nd nd 3.12 0.01 7.76 6.16 4 nd nd nd nd 98.82 nd 0.63 nd nd nd 0.02 0.30 0.22 5 0.08 nd nd 0.05 72.02 nd 0.02 nd nd 7.11 0.04 3.58 10.33 6 nd nd nd nd 99.49 nd 0.02 nd nd nd 0.02 0.11 0.24 7 0.15 0.23 nd nd 98.35 nd nd nd nd nd nd nd 0.24 8 nd nd nd nd 99.84 nd nd nd nd nd nd nd nd Entry 1 (Crude ketone from Route 1); Entry 2 (Ketone Route 1 post bisulfite release); Entry 3 (Crude ketone using crude acid); Entry 4 (Ketone using crude acid Post bisulfite); Entry 5 (Crude ketone using cryst. acid); Entry 6 (Crude ketone using cryst. acid post bisulfite); Entry 7 (Crude ketone using cryst. acid); Entry 8 (Fluorochem ketone); Entry 9 (Fluorochem ketone post bisulfite).
Example 5
- [0156]
- Additional Method for Preparation of 1-(3-trifluoromethyl)phenyl-propan-2-one
- [0157]
35 mL of water and 45 g of 37% (w/w) aqueous hydrochloric acid are put in a flask equipped with stirrer and dropping funnel. 24.25 Grams (0.151 moles) of m-trifluoromethylaniline are added after having cooled to 10 degree C. with an ice bath and then, at 5 degree C., an aqueous solution containing 12.43 g (0.180 moles) of sodium nitrite in 150 ml of water is slowly added. The reaction mixture is stirred for 30 minutes and then is poured during 30 minutes into a mixture made by 90 ml of water, 1.35 g (0.014 moles) of cuprous chloride, 2.30 g (0.013 moles) of cupric chloride dihydrate, 50 ml of acetone, 40.8 g (0.300 moles) of sodium acetate trihydrate and 23 g (0.230 moles) of isopropenyl acetate while keeping the reaction temperature at 30 degree C. After further 30 minutes of stirring, the reaction mixture is brought to 20 degree C., 50 ml of methylene chloride are added and the two layers are separated. - The aqueous layer is discarded while the organic layer is concentrated under vacuum until an oil is obtained which is treated with 35 g of sodium metabisulfite, 70 ml of water and 150 ml of heptane under stirring at room temperature for 12 hours. The suspension is filtered, the bisulfite complex is washed on the filter with 50 ml of heptane and then suspended in a two-phase mixture made by 100 ml of methylene chloride and 150 ml of a 10% (w/v) aqueous solution of sodium hydroxide. The layers are separated after one hour of stirring at room temperature, the aqueous phase is discarded while the organic layer is washed with water and evaporated under vacuum to give pure ketone.
Lurbinectedin

Lurbinectedin
(1’R,6R,6aR,7R,13S,14S,16R)-5-(Acetyloxy)-2′,3′,4′,6,6a,7,9′-decahydro-8,14-dihydroxy-6′,9-dimethoxy-4,10,23-trimethyl-spiro(6,16-(epithiopropaneoxymethano)-7.13-imino-12H-1,3-dioxolo[7,8]soquino[3,2-b][3]benzazocine-20,1′-[1H]pyrido[3,4-b]indol]-19-one
| Molecular Weight | 784.87 |
| Formula | C41H44N4O10S |
| CAS No. | 497871-47-3 (Lurbinectedin); |
| Chemical Name | Spiro[6,16-(epithiopropanoxymethano)-7,13-imino-12H-1,3-dioxolo[7,8]isoquino[3,2-b][3]benzazocine-20,1′-[1H]pyrido[3,4-b]indol]-19-one, 5-(acetyloxy)-2′,3′,4′,6,6a,7,9′,13,14,16-decahydro-8,14-dihydroxy-6′,9-dimethoxy-4,10,23-trimethyl-, (1’R,6R,6aR,7R,13S,14S,16R)- (9CI) |
fda approved , 6/15/2020 , ZEPZELCA, Pharma Mar S.A.
To treat metastatic small cell lung cancer
Drug Trials Snapshot
Research Code:PM-01183; PM-1183
MOA:RNA polymerase inhibitor
Indication:Ovarian cancer; Breast cancer; Non small cell lung cancer (NSCLC)лурбинектединلوربينيكتيدين芦比替定(1R,1’R,2’R,3’R,11’S,12’S,14’R)-5′,12′-Dihydroxy-6,6′-dimethoxy-7′,21′,30′-trimethyl-27′-oxo-2,3,4,9-tetrahydrospiro[β-carboline-1,26′-[17,19,28]trioxa[24]thia[13,30]diazaheptacyclo[12.9.6.13,11. 02,13.04,9.015,23.016,20]triaconta[4,6,8,15,20,22]hexaen]-22′-yl acetate [ACD/IUPAC Name]2CN60TN6ZS497871-47-3[RN]9397
Lurbinectedin is in phase III clinical development for the treatment of platinum refractory/resistant ovarian cancer.
Phase II clinical trials are also ongoing for several oncology indications: non-small cell lung cancer, breast cancer, small cell lung cancer, head and neck carcinoma, neuroendocrine tumors, biliary tract carcinoma, endometrial carcinoma, germ cell tumors and Ewing’s family of tumors.
Lurbinectedin, sold under the brand name Zepzelca, is a medication for the treatment of adults with metastatic small cell lung cancer (SCLC) with disease progression on or after platinum-based chemotherapy.[1][2][3]
The most common side effects include leukopenia, lymphopenia, fatigue, anemia, neutropenia, increased creatinine, increased alanine aminotransferase, increased glucose, thrombocytopenia, nausea, decreased appetite, musculoskeletal pain, decreased albumin, constipation, dyspnea, decreased sodium, increased aspartate aminotransferase, vomiting, cough, decreased magnesium and diarrhea.[1][2][3]
Lurbinectedin is a synthetic tetrahydropyrrolo [4, 3, 2-de]quinolin-8(1H)-one alkaloid analogue with potential antineoplastic activity.[4] Lurbinectedin covalently binds to residues lying in the minor groove of DNA, which may result in delayed progression through S phase, cell cycle arrest in the G2/M phase and cell death.[4]
Lurbinectedin was approved for medical use in the United States in June 2020.[5][1][2][3][6]
Structure
Lurbinectedin is structurally similar to trabectedin, although the tetrahydroisoquinoline present in trabectedin is replaced with a tetrahydro β-carboline which enables lurbinectedin to exhibit increased antitumor activity compared with trabectedin.[7]
Biosynthesis
Lurbinectedin a marine agent isolated from the sea squirt species Ecteinascidia turbinata. Synthetic production is necessary because very small amounts can be obtained from sea organisms. For example, one ton (1000 kg) of sea squirts are required to produce one gram of trabectedin, which is analogue of lurbinectedin. Complex synthesis of lurbinectedin starts from small, common starting materials that require twenty-six individual steps to produce the drug with overall yield of 1.6%.[8][9]
Mechanism of action
According to PharmaMar,[10] lurbinectedin inhibits the active transcription of the encoding genes. This has two consequences. On one hand, it promotes tumor cell death, and on the other it normalizes tumor microenvironment. Active transcription is the process by which there are specific signal where information contained in the DNA sequence is transferred to an RNA molecule. This activity depends on the activity of an enzyme called RNA polymerase II. Lurbinectedin inhibits transcription through a very precise mechanism. Firstly, lurbinectedin binds to specific DNA sequences. It is at these precise spots that slides down the DNA to produce RNA polymerase II that is blocked and degraded by lurbinectedin. Lurbinectedin also has important role in tumor microenvironment. The tumor cells act upon macrophages to avoid them from behaving like an activator of the immune system. Literally, macrophages work in any tumor’s favor. Macrophages can contribute to tumor growth and progression by promoting tumor cell proliferation and invasion, fostering tumor angiogenesis and suppressing antitumor immune cells.[11][12] Attracted to oxygen-starved (hypoxic) and necrotic tumor cells they promote chronic inflammation. So, not only that macrophages inhibit immune system avoiding the destruction of tumor cells, but they also create tumor tissue that allows tumor growth. However, macrophages associated with tumors are cells that are addicted to the transcription process. Lurbinectedin acts specifically on the macrophages associated with tumors in two ways: firstly, by inhibiting the transcription of macrophages that leads to cell death and secondly, inhibiting the production of tumor growth factors. In this way, lurbinectedin normalizes the tumor microenvironment.
History
Lurbinectedin was approved for medical use in the United States in June 2020.[5][1][2][3][6]
Efficacy was demonstrated in the PM1183-B-005-14 trial (Study B-005; NCT02454972), a multicenter open-label, multi-cohort study enrolling 105 participants with metastatic SCLC who had disease progression on or after platinum-based chemotherapy.[3][6] Participants received lurbinectedin 3.2 mg/m2 by intravenous infusion every 21 days until disease progression or unacceptable toxicity.[3] The trial was conducted at 26 sites in the United States, Great Britain, Belgium, France, Italy, Spain and Czech Republic.[6]
The U.S. Food and Drug Administration (FDA) granted the application for lurbinectedin priority review and orphan drug designations and granted the approval of Zepzelca to Pharma Mar S.A.[3][13]
Research
Clinical Trials
Lurbinectedin can be used as monotherapy in the treatment of SCLC. Lurbinectedin monotherapy demonstrated the following clinical results in relapsed extensive stage SCLC:
- For sensitive disease (chemotherapy-free interval of ≥ 90 days) overall response rate (ORR) was 46.6% with 79.3% disease control rate and median overall survival (OS) being increased to 15.2 months.[14]
- For resistant disease (chemotherapy-free interval of < 90 days) overall response rate (ORR) was 21.3% with 46.8% disease control rate and 5.1 months median overall survival (OS).[14]
Lurbinectedin is also being investigated in combination with doxorubicin as second-line therapy in a randomized Phase III trial.[medical citation needed] While overall survival in this trial is not yet known, response rates at second line were
- 91.7% in sensitive disease with median progression-free survival of 5.8 months, and
- 33.3% in resistant disease with median progression-free of 3.5 months.[15]
Lurbinectedin is available in the U.S. under Expanded Access Program (EAP).[15][16]
SYN
SYN
WO2011/147828
Ecteinascidins is a group of naturally occurring marine compounds and analogs thereof, which are well identified and structurally characterized, and are disclosed to have antibacterial and cytotoxic properties. See for example, European Patent 309.477; WO 03/66638; WO 03/08423; WO 01 /771 15; WO 03/014127; R. Sakai et al., 1992, Proc. Natl. Acad. Sci. USA 89, pages 1 1456- 1 1460; R. Menchaca et al., 2003, J. Org. Chem. 68(23), pages 8859-8866; and I. Manzanares et al., 2001 , Curr. Med. Chem. Anti-Cancer Agents, 1 , pages 257-276; and references therein. Examples of ecteinascidins are provided by ET-743, ET-729, ET-745, ET-759A, ET-759B, ET-759C, ET-770, ET-815, ET-731 , ET-745B, ET-722, ET-736, ET-738, ET-808, ET-752, ET-594, ET-552, ET-637, ET-652, ET-583, ET-597, ET-596, ET-639, ET-641 , and derivatives thereof, such as acetylated forms, formylated forms, methylated forms, and oxide forms.
The structural characterizations of such ecteinascidins are not given again explicitly herein because from the detailed description provided in such references and citations any person of ordinary skill in this technology is capable of obtaining such information directly from the sources cited here and related sources.
At least one of the ecteinascidin compounds, ecteinascidin 743 (ET-743), has been extensively studied, and it will be referred to
specifically herein to illustrate features of this invention. ET-743 is being employed as an anticancer medicament, under the international nonproprietary name (INN) trabectedin, for the treatment of patients with advanced and metastatic soft tissue sarcoma (STS), after failure of anthracyclines and ifosfamide, or who are unsuited to receive such agents, and for the treatment of relapsed platinum- sensitive ovarian cancer in combination with pegylated liposomal doxorubicin.
ET-743 has a complex tris(tetrahydroisoquinoline) structure of formula
It was originally prepared by isolation from extracts of the marine tunicate Ecteinascidia turbinata. The yield was low, and alternative preparative processes had been sought.
The first synthetic process for producing ecteinascidin compounds was described in US Patent 5,721 ,362. This process employed sesamol as starting material and yielded ET-743 after a long and complicated sequence of 38 examples each describing one or more steps in the synthetic sequence.
An improvement in the preparation of one intermediate used in such process was disclosed in US Patent 6,815,544. Even with this improvement, the total synthesis was not suitable for manufacturing ET-743 at an industrial scale.
A hemisynthetic process for producing ecteinascidin compounds was described in EP 1.185.536. This process employs cyanosafracin B as starting material to provide ET-743. Cyanosafracin B is a pentacyclic antibiotic obtained by fermentation from the bacteria Pseudomonas fluorescens.
Cyanosafracin B
An improvement in such hemisynthetic process was disclosed in
EP 1.287.004.
To date four additional synthetic process (2 total and 2 formal synthesis) have been disclosed in patent applications JP 2003221395, WO 2007/045686, and WO 2007/087220 and in J. Org. Chem. 2008, 73, pages 9594-9600.
WO 2007/045686 also relates to the synthesis of Ecteinascidins-583 and 597 using intermediate compounds of formula:
Total synthesis strategies for the synthesis of the pentacyclic core -743 are overviewed in Figure I.
X = OH or CI
R = Protecting Group
WO2007087220 JOC 2008, 73, 9594-9600
EXAMPLE 3: SYNTHESIS OF COMPOUND 17.
Scheme X above provides an example of the synthesis of compound 17 from intermediate 10.
Compounds 16 and 17 are obtainable from intermediate 15 using the same procedures than those previously described in WO03/014127.
SYN

Reference:
1. WO2003014127A1.
https://patents.google.com/patent/WO2003014127A1/en
The ecteinascidins are exceedingly potent antitumour agents isolated from the marine tunicate Ecteinascidia turbinata. Several ecteinascidins have been reported previously in the patent and scientific literature. See, for example:
U.S. Patent No 5.256.663, which describes pharmaceutical compositions comprising matter extracted from the tropical marine invertebrate, Ecteinascidia turbinata, and designated therein as ecteinascidins, and the use of such compositions as antibacterial, antiviral, and/ or antitumour agents in mammals.
U.S. Patent No 5.089.273, which describes novel compositions of matter extracted from the tropical marine invertebrate, Ecteinascidia turbinata, and designated therein as ecteinascidins 729, 743, 745, 759A, 759B and 770. These compounds are useful as antibacterial and/or antitumour agents in mammals.
U.S. Patent No 5.149.804 which describes Ecteinascidins 722 and 736 (Et’s 722 and 736) isolated from the Caribbean tunicate Ecteinascidia turbinata and their structures. Et’s 722 and 736 protect mice in vivo at very low concentrations against P388 lymphoma, B 16 melanoma, and Lewis lung carcinoma.
U.S. Patent No 5.478.932, which describes ecteinascidins isolated from the Caribbean tunicate Ecteinascidia turbinata, which provide in vivo protection against P388 lymphoma, B 16 melanoma, M5076 ovarian sarcoma, Lewis lung carcinoma, and the LX- 1 human lung and MX- 1 human mammary carcinoma xenografts.
U.S. Patent No 5.654.426, which describes several ecteinascidins isolated from the Caribbean tunicate Ecteinascidia turbinata, which provide in vivo protection against P388 lymphoma, B 16 melanoma, M5076 ovarian sarcoma, Lewis lung carcinoma, and the LX-1 human lung and MX- 1 human mammary carcinoma xenografts.
U.S. Patent No 5.721.362 which describes a synthetic process for the formation of ecteinascidin compounds and related structures.
U.S. Patent No 6.124.292 which describes a series of new ecteinascidin- like compounds.
WO 0177115, WO 0187894 and WO 0187895, which describe new synthetic compounds of the ecteinascidin series, their synthesis and biological properties.
See also: Corey, E.J., J. Am. Chem. Soc, 1996, 118 pp. 9202-9203; Rinehart, et al., Journal of Natural Products, 1990, “Bioactive Compounds from Aquatic and Terrestrial Sources”, vol. 53, pp. 771- 792; Rinehart et al., Pure and Appl. Chem., 1990, “Biologically active natural products”, vol 62, pp. 1277- 1280; Rinehart, et al., J. Org. Chem., 1990, “Ecteinascidins 729, 743, 745, 759A, 759B, and 770: potent Antitumour Agents from the Caribbean Tunicate Ecteinascidia tuminata”, vol. 55, pp. 4512-4515; Wright et al., J. Org. Chem., 1990, “Antitumour Tetrahydroisoquinoline Alkaloids from the Colonial ascidian Ecteinascidia turbinata”, vol. 55, pp. 4508-4512; Sakai et al., Proc. Natl. Acad. Sci. USA 1992, “Additional anitumor ecteinascidins from a Caribbean tunicate: Crystal structures and activities in vivo”, vol. 89, 1 1456- 1 1460; Science 1994, “Chemical Prospectors Scour the Seas for Promising Drugs”, vol. 266, pp.1324; Koenig, K.E., “Asymmetric Synthesis”, ed. Morrison, Academic Press, Inc., Orlando, FL, vol. 5, 1985, p. 71; Barton, et al., J. Chem Soc. Perkin Trans., 1 , 1982, “Synthesis and Properties of a Series of Sterically Hindered Guanidine bases”, pp. 2085; Fukuyama et al., J. Am. Chem. Soc, 1982, “Stereocontrolled Total Synthesis of (+)-Saframycin B”, vol. 104, pp. 4957; Fukuyama et al., J. Am. Chem. Soc, 1990, “Total Synthesis of (+) – Saframycin A”, vol. 112, p. 3712; Saito, et al., J. Org. Chem., 1989, “Synthesis of Saframycins. Preparation of a Key tricyclic Lactam Intermediate to Saframycin A”, vol. 54, 5391; Still, et al., J Org. Chem., 1978, “Rapid Chromatographic Technique for Preparative Separations with Moderate Resolution”, vol. 43, p. 2923; Kofron, W.G.; Baclawski, L.M., J. Org. Chem., 1976, vol. 41, 1879; Guan et al., J. Biomolec Struc & Dynam., vol. 10, pp. 793-817 (1993); Shamma et al., “Carbon- 13 NMR Shift Assignments of Amines and Alkaloids”, p. 206 (1979); Lown et al., Biochemistry, 21, 419-428 (1982); Zmijewski et al., Chem. Biol. Interactions, 52, 361-375 (1985); Ito, CRC Crit. Rev. Anal. Chem., 17, 65- 143 (1986); Rinehart et al., “Topics in Pharmaceutical Sciences 1989”, pp. 613-626, D. D. Breimer, D. J. A. Cromwelin, K. K. Midha, Eds., Amsterdam Medical Press B. V., Noordwijk, The Netherlands (1989); Rinehart et al., “Biological Mass Spectrometry”, 233-258 eds. Burlingame et al., Elsevier Amsterdam (1990); Guan et al., Jour. Biomolec. Struct. & Dynam., vol. 10 pp. 793-817 (1993); Nakagawa et al., J. Amer. Chem. Soc, 11 1 : 2721-2722 (1989);; Lichter et al., “Food and Drugs from the Sea Proceedings” (1972), Marine Technology Society, Washington, D.C. 1973, 117- 127; Sakai et al., J. Amer. Chem. Soc, 1996, 1 18, 9017; Garcϊa-Rocha et al., Brit. J. Cancer, 1996, 73: 875-883; and pommier et al., Biochemistry, 1996, 35: 13303- 13309;
In 2000, a hemisynthetic process for the formation of ecteinascidin compounds and related structures such as phthalascidin starting from natural bis(tetrahydroisoquinoline) alkaloids such as the saframycin and safracin antibiotics available from different culture broths was reported; See Manzanares et al., Org. Lett., 2000, “Synthesis of Ecteinascidin ET-743 and Phthalascidin Pt-650 from Cyanosafracin B”, Vol. 2, No 16, pp. 2545-2548; and International Patent Application WO 00 69862.
Ecteinascidin 736 was first discovered by Rinehart and features a tetrahydro-β-carboline unit in place of the tetrahydroisoquinoline unit more usually found in the ecteinascidin compounds isolated from natural sources; See for example Sakai et al., Proc. Natl. Acad. Sci. USA 1992, “Additional antitumor ecteinascidins from a Caribbean tunicate: Crystal structures and activities in vivo”, vol. 89, 11456-11460.
Et-736
WO 9209607 claims ecteinascidin 736, as well as ecteinascidin 722 with hydrogen in place of methyl on the nitrogen common to rings C and D of ecteinascidin 736 and O-methylecteinascidin 736 with methoxy in place of hydroxy on ring C of ecteinascidin 736.
Despite the positive results obtained in clinical applications in chemotherapy, the search in the field of ecteinascidin compounds is still open to the identification of new compounds with optimal features of cytotoxicity and selectivity toward the tumour and with a reduced systemic toxicity and improved pharmacokinetic properties.
PATENT
WO2001087894A1.
PATENT
US 20130066067
https://patents.google.com/patent/US20130066067A1/en
- Ecteinascidins is a group of naturally occurring marine compounds and analogs thereof, which are well identified and structurally characterized, and are disclosed to have antibacterial and cytotoxic properties. See for example, European Patent 309.477; WO 03/66638; WO 03/08423; WO 01/77115; WO 03/014127; R. Sakai et al., 1992, Proc. Natl. Acad. Sci. USA 89, pages 11456-11460; R. Menchaca et al., 2003, J. Org. Chem. 68(23), pages 8859-8866; and I. Manzanares et al., 2001, Curr. Med. Chem. Anti–Cancer Agents, 1, pages 257-276; and references therein. Examples of ecteinascidins are provided by ET-743, ET-729, ET-745, ET-759A, ET-759B, ET-759C, ET-770, ET-815, ET-731, ET-745B, ET-722, ET-736, ET-738, ET-808, ET-752, ET-594, ET-552, ET-637, ET-652, ET-583, ET-597, ET-596, ET-639, ET-641, and derivatives thereof, such as acetylated forms, formylated forms, methylated forms, and oxide forms.
- [0003]
The structural characterizations of such ecteinascidins are not given again explicitly herein because from the detailed description provided in such references and citations any person of ordinary skill in this technology is capable of obtaining such information directly from the sources cited here and related sources. - [0004]
At least one of the ecteinascidin compounds, ecteinascidin 743 (ET-743), has been extensively studied, and it will be referred to specifically herein to illustrate features of this invention. ET-743 is being employed as an anticancer medicament, under the international nonproprietary name (INN) trabectedin, for the treatment of patients with advanced and metastatic soft tissue sarcoma (STS), after failure of anthracyclines and ifosfamide, or who are unsuited to receive such agents, and for the treatment of relapsed platinum-sensitive ovarian cancer in combination with pegylated liposomal doxorubicin. - [0005]
ET-743 has a complex tris(tetrahydroisoquinoline) structure of formula - [0006]
It was originally prepared by isolation from extracts of the marine tunicate Ecteinascidia turbinata. The yield was low, and alternative preparative processes had been sought. - [0007]
The first synthetic process for producing ecteinascidin compounds was described in U.S. Pat. No. 5,721,362. This process employed sesamol as starting material and yielded ET-743 after a long and complicated sequence of 38 examples each describing one or more steps in the synthetic sequence. - [0008]
An improvement in the preparation of one intermediate used in such process was disclosed in U.S. Pat. No. 6,815,544. Even with this improvement, the total synthesis was not suitable for manufacturing ET-743 at an industrial scale. - [0009]
A hemisynthetic process for producing ecteinascidin compounds was described in EP 1.185.536. This process employs cyanosafracin B as starting material to provide ET-743. Cyanosafracin B is a pentacyclic antibiotic obtained by fermentation from the bacteria Pseudomonas fluorescens. - [0010]
An improvement in such hemisynthetic process was disclosed in EP 1.287.004. - [0011]
To date four additional synthetic process (2 total and 2 formal synthesis) have been disclosed in patent applications JP 2003221395, WO 2007/045686, and WO 2007/087220 and in J. Org. Chem. 2008, 73, pages 9594-9600. - [0012]
WO 2007/045686 also relates to the synthesis of Ecteinascidins-583 and 597 using intermediate compounds of formula: - [0013]
Total synthesis strategies for the synthesis of the pentacyclic core of ET-743 are overviewed in FIG. 1.
PAPER
Angewandte Chemie, International Edition (2019), 58(12), 3972-3975.
https://onlinelibrary.wiley.com/doi/abs/10.1002/anie.201900035
An efficient and scalable approach is described for the total synthesis of the marine natural product Et‐743 and its derivative lubinectedin, which are valuable antitumor compounds. The method delivers 1.6 % overall yield in 26 total steps from Cbz‐protected (S)‐tyrosine. It features the use of a common advanced intermediate to create the right and left parts of these compounds, and a light‐mediated remote C−H bond activation to assemble a benzo[1,3]dioxole‐containing intermediate.

Synthesis of lactone SI-5. A mixture of 19 (98.0 mg, 0.16 mmol, 1.0 equiv), 2-(5-methoxy-1H-indol-3-yl) ethanamine hydrochloride salt (357.8 mg, 1.58 mmol, 10.0 equiv) and NaOAc (144 mg, 1.74 mmol, 11.0 equiv) in anhydrous EtOH (5.0 mL) was stirred at 60 oC for 5 h. The cooled mixture was extracted with ethyl acetate, and the organic layer was dried over sodium sulfate and concentrated. The residue was purified by flash column chromatography (eluting with DCM/MeOH = 20:1) to afford compound SI-5 (109 mg, 87%). [α]𝐷 20 = -27.7 (c = 1.0, CHCl3). 1H NMR (400 MHz, CDCl3) δ 7.61 (s, 1H), 7.13 (d, J = 8.8 Hz, 1H), 6.82 (d, J = 2.2 Hz, 1H), 6.75 (dd, J = 8.8, 2.4 Hz, 1H), 6.66 (s, 1H), 6.22 (d, J = 1.0 Hz, 1H), 6.02 (d, J = 1.0 Hz, 1H), 5.78 (s, 1H), 5.08 (d, J = 11.7 Hz, 1H), 4.55 (s, 1H), 4.32 (s, 1H), 4.27 (d, J = 3.8 Hz, 1H), 4.23–4.15 (m, 2H), 3.81 (s, 3H), 3.79 (s, 3H), 3.47–3.39 (m, 2H), 3.20–3.10 (m, 1H), 3.06 (d, J = 18.1 Hz, 1H), 2.93 (dd, J = 18.2, 9.1 Hz, 1H), 2.86–2.76 (m, 1H), 2.62 (dt, J = 14.9, 4.8 Hz, 1H), 2.56–2.47 (m, 2H), 2.37 (s, 3H), 2.30–2.27 (m, 1H), 2.26 (s, 3H), 2.22 (s, 3H), 2.06 (s, 3H); 13C NMR (100 MHz, CDCl3) δ 171.6, 168.8, 154.0, 148.2, 145.8, 143.1, 141.3, 140.5, 131.4, 130.8, 130.7, 129.4, 127.3, 120.9, 120.8, 118.4, 118.4, 113.9, 113.8, 112.2, 111.8, 110.2, 102.2, 100.5, 62.6, 61.4, 60.7, 60.5, 59.6, 59.6, 55.9, 54.9, 54.8, 42.1, 41.6, 39.9, 39.5, 29.5, 24.0, 20.8, 16.0, 9.9; HRMS (ESI) m/z calcd. for C42H43N5O9S [M + H]+ 794.2860, found 794.2858

Lurbinectedin: To a solution of SI-5 (80 mg, 0.1 mmol, 1.0 equiv) in acetonitrile and water (3:2, v/v, 10 mL) was added silver nitrate (514 mg, 3 mmol, 30.0 equiv). The suspension was stirred at 25 oC for 24 h before a mixture of saturated brine (5.0 mL) and saturated sodium hydrogen carbonate (5 mL) were added. The resultant mixture was stirred at 25 oC for 15 min before it was filtered through celite and extracted with ethyl acetate (3 × 20 mL). The combined organic layers were dried over sodium sulfate and concentrated, and the residue was purified by flash column chromatography (eluting with DCM/MeOH = 20:1) to afford Lurbinectedin (71 mg, 89%). [α]𝐷 20 = -45.0 (c = 1.0, CHCl3) 1H NMR (400 MHz, CDCl3) δ 7.61 (s, 1H), 7.13 (d, J = 8.8 Hz, 1H), 6.82 (d, J = 2.2 Hz, 1H), 6.74 (dd, J = 8.8, 2.4 Hz, 1H), 6.67 (s, 1H), 6.19 (d, J = 1.1 Hz, 1H), 5.99 (d, J = 1.1 Hz, 1H), 5.77 (br s, 1H), 5.20 (d, J = 11.3 Hz, 1H), 4.82 (s, 1H), 4.53–4.40 (m, 2H), 4.18–4.08 (m, 2H), 3.81 (s, 3H), 3.79 (s, 3H), 3.49 (d, J = 4.2 Hz, 1H), 3.24–3.13 (m, 2H), 3.01 (d, J = 17.9 Hz, 1H), 2.88–2.79 (m, 2H), 2.63 (dt, J = 15.0, 4.9 Hz, 1H), 2.56–2.47 (m, 2H), 2.37 (s, 3H), 2.32–2.27 (m, 1H), 2.26 (s, 3H), 2.19 (s, 3H), 2.05 (s, 3H); 13C NMR (100 MHz, CDCl3) δ 171.4, 168.8, 153.8, 147.9, 145.5, 142.9, 141.1, 140.7, 131.8, 131.3, 130.7, 129.1, 127.3, 121.4, 121.0, 118.2, 115.6, 112.9, 111.9, 111.7, 110.0, 101.8, 100.4, 82.0, 62.4, 61.9, 60.4, 57.8, 57.5, 56.0, 55.8, 55.0, 42.2, 41.3, 39.8, 39.3, 29.3, 23.6, 20.6, 15.9, 9.7; HRMS (ESI) m/z calcd. for C41H44N4O10S [M – OH]+ 767.2745, found 767.2742.
References
- ^ Jump up to:a b c d e “Zepzelca- lurbinectedin injection, powder, lyophilized, for solution”. DailyMed. 15 June 2020. Retrieved 24 September 2020.
- ^ Jump up to:a b c d “Jazz Pharmaceuticals Announces U.S. FDA Accelerated Approval of Zepzelca (lurbinectedin) for the Treatment of Metastatic Small Cell Lung Cancer” (Press release). Jazz Pharmaceuticals. 15 June 2020. Retrieved 15 June 2020 – via PR Newswire.
- ^ Jump up to:a b c d e f g “FDA grants accelerated approval to lurbinectedin for metastatic small”. U.S. Food and Drug Administration (FDA). 15 June 2020. Retrieved 16 June 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Jump up to:a b “Lurbinectedin”. National Cancer Institute. Retrieved 15 June 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Jump up to:a b “Zepzelca: FDA-Approved Drugs”. U.S. Food and Drug Administration (FDA). Retrieved 15 June 2020.
- ^ Jump up to:a b c d “Drug Trials Snapshots: Zepzelca”. U.S. Food and Drug Administration (FDA). 15 June 2020. Retrieved 28 June 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Takahashi, Ryoko; Mabuchi, Seiji; Kawano, Mahiru; Sasano, Tomoyuki; Matsumoto, Yuri; Kuroda, Hiromasa; Kozasa, Katsumi; Hashimoto, Kae; Sawada, Kenjiro; Kimura, Tadashi (17 March 2016). “Preclinical Investigations of PM01183 (Lurbinectedin) as a Single Agent or in Combination with Other Anticancer Agents for Clear Cell Carcinoma of the Ovary”. PLOS ONE. 11 (3): e0151050. Bibcode:2016PLoSO..1151050T. doi:10.1371/journal.pone.0151050. PMC 4795692. PMID 26986199.
- ^ Total synthesis of marine antitumor agents trabectedin and lurbinectedin | https://www.sciencedaily.com/releases/2019/02/190219111659.htm
- ^ A Scalable Total Synthesis of the Antitumor Agents Et‐743 and Lurbinectedin | https://onlinelibrary.wiley.com/doi/full/10.1002/anie.201900035
- ^ PharmaMar presentation of Lurbinectedin’s Mechanism of Action Lurbinectedin Mechanisim of Action | https://www.youtube.com/watch?v=8daELhxAXcQ
- ^ Qian BZ, Pollard JW (April 2010). “Macrophage diversity enhances tumor progression and metastasis”. Cell. 141 (1): 39–51. doi:10.1016/j.cell.2010.03.014. PMC 4994190. PMID 20371344.
- ^ Engblom C, Pfirschke C, Pittet MJ (July 2016). “The role of myeloid cells in cancer therapies”. Nature Reviews. Cancer. 16 (7): 447–62. doi:10.1038/nrc.2016.54. PMID 27339708. S2CID 21924175.
- ^ “Lurbinectedin Orphan Drug Designation and Approval”. U.S. Food and Drug Administration (FDA). 1 August 2018. Retrieved 16 June 2020.
- ^ Jump up to:a b Paz-Ares, Luis G.; Trigo Perez, Jose Manuel; Besse, Benjamin; Moreno, Victor; Lopez, Rafael; Sala, Maria Angeles; Ponce Aix, Santiago; Fernandez, Cristian Marcelo; Siguero, Mariano; Kahatt, Carmen Maria; Zeaiter, Ali Hassan; Zaman, Khalil; Boni, Valentina; Arrondeau, Jennifer; Martinez Aguillo, Maite; Delord, Jean-Pierre; Awada, Ahmad; Kristeleit, Rebecca Sophie; Olmedo Garcia, Maria Eugenia; Subbiah, Vivek (20 May 2019). “Efficacy and safety profile of lurbinectedin in second-line SCLC patients: Results from a phase II single-agent trial”. Journal of Clinical Oncology. 37 (15_suppl): 8506. doi:10.1200/JCO.2019.37.15_suppl.8506.
- ^ Jump up to:a b Calvo, E.; Moreno, V.; Flynn, M.; Holgado, E.; Olmedo, M.E.; Lopez Criado, M.P.; Kahatt, C.; Lopez-Vilariño, J.A.; Siguero, M.; Fernandez-Teruel, C.; Cullell-Young, M.; Soto Matos-Pita, A.; Forster, M. (October 2017). “Antitumor activity of lurbinectedin (PM01183) and doxorubicin in relapsed small-cell lung cancer: results from a phase I study”. Annals of Oncology. 28 (10): 2559–2566. doi:10.1093/annonc/mdx357. PMC 5834091. PMID 28961837. Lay summary.
- ^ Farago, Anna F; Drapkin, Benjamin J; Lopez-Vilarino de Ramos, Jose Antonio; Galmarini, Carlos M; Núñez, Rafael; Kahatt, Carmen; Paz-Ares, Luis (January 2019). “ATLANTIS: a Phase III study of lurbinectedin/doxorubicin versus topotecan or cyclophosphamide/doxorubicin/vincristine in patients with small-cell lung cancer who have failed one prior platinum-containing line”. Future Oncology. 15 (3): 231–239. doi:10.2217/fon-2018-0597. PMC 6331752. PMID 30362375.
External links
- “Lurbinectedin”. Drug Information Portal. U.S. National Library of Medicine.
- “Lurbinectedin”. NCI Dictionary of Cancer Terms. National Cancer Institute.
- Clinical trial number NCT02454972 for “Clinical Trial of Lurbinectedin (PM01183) in Selected Advanced Solid Tumors” at ClinicalTrials.gov
FDA grants accelerated approval to lurbinectedin for metastatic small cell lung cancer
On June 15, 2020, the Food and Drug Administration granted accelerated approval to lurbinectedin(ZEPZELCA, Pharma Mar S.A.) for adult patients with metastatic small cell lung cancer (SCLC) with disease progression on or after platinum-based chemotherapy.
Efficacy was demonstrated in the PM1183-B-005-14 trial (Study B-005; NCT02454972), a multicenter open-label, multi-cohort study enrolling 105 patients with metastatic SCLC who had disease progression on or after platinum-based chemotherapy. Patients received lurbinectedin 3.2 mg/m2 by intravenous infusion every 21 days until disease progression or unacceptable toxicity.
The main efficacy outcome measures were confirmed overall response rate (ORR) determined by investigator assessment using RECIST 1.1 and response duration. Among the 105 patients, the ORR was 35% (95% CI: 26%, 45%), with a median response duration of 5.3 months (95% CI: 4.1, 6.4). The ORR as per independent review committee was 30% (95% CI: 22%, 40%) with a median response duration of 5.1 months (95% CI: 4.9, 6.4).
The most common adverse reactions (≥20%), including laboratory abnormalities, were myelosuppression, fatigue, increased creatinine, increased alanine aminotransferase, increased glucose, nausea, decreased appetite, musculoskeletal pain, decreased albumin, constipation, dyspnea, decreased sodium, increased aspartate aminotransferase, vomiting, cough, decreased magnesium and diarrhea.
The recommended lurbinectedin dose is 3.2 mg/m2 every 21 days.
View full prescribing information for ZEPZELCA.
This indication is approved under accelerated approval based on overall response rate and duration of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trials.
This review was conducted under Project Orbis, an initiative of the FDA Oncology Center of Excellence. Project Orbis provides a framework for concurrent submission and review of oncology drugs among international partners. For this application, a modified Project Orbis was undertaken because of the timing of submission to other regulatory agencies. FDA is collaborating with the Australian Therapeutic Goods Administration (TGA). FDA approved this application 2 months ahead of the goal date. The review is ongoing for the Australian TGA.
FDA granted lurbinectedin orphan drug designation for the treatment of SCLC and priority review to this application. A description of FDA expedited programs is in the Guidance for Industry: Expedited Programs for Serious Conditions-Drugs and Biologics.
REFERENCES
1: Calvo E, Moreno V, Flynn M, Holgado E, Olmedo ME, Lopez Criado MP, Kahatt C, Lopez-Vilariño JA, Siguero M, Fernandez-Teruel C, Cullell-Young M, Soto Matos-Pita A, Forster M. Antitumor activity of lurbinectedin (PM01183) and doxorubicin in relapsed small-cell lung cancer: results from a phase I study. Ann Oncol. 2017 Oct 1;28(10):2559-2566. doi: 10.1093/annonc/mdx357. PubMed PMID: 28961837.
2: Erba E, Romano M, Gobbi M, Zucchetti M, Ferrari M, Matteo C, Panini N, Colmegna B, Caratti G, Porcu L, Fruscio R, Perlangeli MV, Mezzanzanica D, Lorusso D, Raspagliesi F, D’Incalci M. Ascites interferes with the activity of lurbinectedin and trabectedin: Potential role of their binding to alpha 1-acid glycoprotein. Biochem Pharmacol. 2017 Nov 15;144:52-62. doi: 10.1016/j.bcp.2017.08.001. Epub 2017 Aug 4. PubMed PMID: 28782526.
3: Belgiovine C, Bello E, Liguori M, Craparotta I, Mannarino L, Paracchini L, Beltrame L, Marchini S, Galmarini CM, Mantovani A, Frapolli R, Allavena P, D’Incalci M. Lurbinectedin reduces tumour-associated macrophages and the inflammatory tumour microenvironment in preclinical models. Br J Cancer. 2017 Aug 22;117(5):628-638. doi: 10.1038/bjc.2017.205. Epub 2017 Jul 6. PubMed PMID: 28683469; PubMed Central PMCID: PMC5572168.
4: Jimeno A, Sharma MR, Szyldergemajn S, Gore L, Geary D, Diamond JR, Fernandez Teruel C, Soto Matos-Pita A, Iglesias JL, Cullell-Young M, Ratain MJ. Phase I study of lurbinectedin, a synthetic tetrahydroisoquinoline that inhibits activated transcription, induces DNA single- and double-strand breaks, on a weekly × 2 every-3-week schedule. Invest New Drugs. 2017 Aug;35(4):471-477. doi: 10.1007/s10637-017-0427-2. Epub 2017 Jan 20. PubMed PMID: 28105566.
5: Paz-Ares L, Forster M, Boni V, Szyldergemajn S, Corral J, Turnbull S, Cubillo A, Teruel CF, Calderero IL, Siguero M, Bohan P, Calvo E. Phase I clinical and pharmacokinetic study of PM01183 (a tetrahydroisoquinoline, Lurbinectedin) in combination with gemcitabine in patients with advanced solid tumors. Invest New Drugs. 2017 Apr;35(2):198-206. doi: 10.1007/s10637-016-0410-3. Epub 2016 Nov 21. PubMed PMID: 27873130.
6: Harlow ML, Maloney N, Roland J, Guillen Navarro MJ, Easton MK, Kitchen-Goosen SM, Boguslawski EA, Madaj ZB, Johnson BK, Bowman MJ, D’Incalci M, Winn ME, Turner L, Hostetter G, Galmarini CM, Aviles PM, Grohar PJ. Lurbinectedin Inactivates the Ewing Sarcoma Oncoprotein EWS-FLI1 by Redistributing It within the Nucleus. Cancer Res. 2016 Nov 15;76(22):6657-6668. doi: 10.1158/0008-5472.CAN-16-0568. Epub 2016 Oct 3. PubMed PMID: 27697767; PubMed Central PMCID: PMC5567825.
7: Céspedes MV, Guillén MJ, López-Casas PP, Sarno F, Gallardo A, Álamo P, Cuevas C, Hidalgo M, Galmarini CM, Allavena P, Avilés P, Mangues R. Lurbinectedin induces depletion of tumor-associated macrophages, an essential component of its in vivo synergism with gemcitabine, in pancreatic adenocarcinoma mouse models. Dis Model Mech. 2016 Dec 1;9(12):1461-1471. Epub 2016 Oct 20. PubMed PMID: 27780828; PubMed Central PMCID: PMC5200894.
8: Santamaría Nuñez G, Robles CM, Giraudon C, Martínez-Leal JF, Compe E, Coin F, Aviles P, Galmarini CM, Egly JM. Lurbinectedin Specifically Triggers the Degradation of Phosphorylated RNA Polymerase II and the Formation of DNA Breaks in Cancer Cells. Mol Cancer Ther. 2016 Oct;15(10):2399-2412. Epub 2016 Sep 14. PubMed PMID: 27630271.
9: Metaxas Y, Cathomas R, Mark M, von Moos R. Combination of cisplatin and lurbinectedin as palliative chemotherapy in progressive malignant pleural mesothelioma: Report of two cases. Lung Cancer. 2016 Dec;102:136-138. doi: 10.1016/j.lungcan.2016.07.012. Epub 2016 Jul 14. PubMed PMID: 27440191.
10: Lima M, Bouzid H, Soares DG, Selle F, Morel C, Galmarini CM, Henriques JA, Larsen AK, Escargueil AE. Dual inhibition of ATR and ATM potentiates the activity of trabectedin and lurbinectedin by perturbing the DNA damage response and homologous recombination repair. Oncotarget. 2016 May 3;7(18):25885-901. doi: 10.18632/oncotarget.8292. PubMed PMID: 27029031; PubMed Central PMCID: PMC5041952.
11: Takahashi R, Mabuchi S, Kawano M, Sasano T, Matsumoto Y, Kuroda H, Kozasa K, Hashimoto K, Sawada K, Kimura T. Preclinical Investigations of PM01183 (Lurbinectedin) as a Single Agent or in Combination with Other Anticancer Agents for Clear Cell Carcinoma of the Ovary. PLoS One. 2016 Mar 17;11(3):e0151050. doi: 10.1371/journal.pone.0151050. eCollection 2016. PubMed PMID: 26986199; PubMed Central PMCID: PMC4795692.
12: Pernice T, Bishop AG, Guillen MJ, Cuevas C, Aviles P. Development of a liquid chromatography/tandem mass spectrometry assay for the quantification of PM01183 (lurbinectedin), a novel antineoplastic agent, in mouse, rat, dog, Cynomolgus monkey and mini-pig plasma. J Pharm Biomed Anal. 2016 May 10;123:37-41. doi: 10.1016/j.jpba.2016.01.043. Epub 2016 Jan 21. PubMed PMID: 26871278.
13: Elez ME, Tabernero J, Geary D, Macarulla T, Kang SP, Kahatt C, Pita AS, Teruel CF, Siguero M, Cullell-Young M, Szyldergemajn S, Ratain MJ. First-in-human phase I study of Lurbinectedin (PM01183) in patients with advanced solid tumors. Clin Cancer Res. 2014 Apr 15;20(8):2205-14. doi: 10.1158/1078-0432.CCR-13-1880. Epub 2014 Feb 21. PubMed PMID: 24563480.
14: Romano M, Frapolli R, Zangarini M, Bello E, Porcu L, Galmarini CM, García-Fernández LF, Cuevas C, Allavena P, Erba E, D’Incalci M. Comparison of in vitro and in vivo biological effects of trabectedin, lurbinectedin (PM01183) and Zalypsis® (PM00104). Int J Cancer. 2013 Nov;133(9):2024-33. doi: 10.1002/ijc.28213. Epub 2013 May 25. PubMed PMID: 23588839.
15: Vidal A, Muñoz C, Guillén MJ, Moretó J, Puertas S, Martínez-Iniesta M, Figueras A, Padullés L, García-Rodriguez FJ, Berdiel-Acer M, Pujana MA, Salazar R, Gil-Martin M, Martí L, Ponce J, Molleví DG, Capella G, Condom E, Viñals F, Huertas D, Cuevas C, Esteller M, Avilés P, Villanueva A. Lurbinectedin (PM01183), a new DNA minor groove binder, inhibits growth of orthotopic primary graft of cisplatin-resistant epithelial ovarian cancer. Clin Cancer Res. 2012 Oct 1;18(19):5399-411. doi: 10.1158/1078-0432.CCR-12-1513. Epub 2012 Aug 15. PubMed PMID: 22896654.
| Clinical data | |
|---|---|
| Pronunciation | LOOR-bih-NEK-teh-din |
| Trade names | Zepzelca |
| Other names | PM-01183 |
| AHFS/Drugs.com | Professional Drug Facts |
| MedlinePlus | a620049 |
| License data | US DailyMed: Lurbinectedin |
| Pregnancy category | US: N (Not classified yet) |
| Routes of administration | Intravenous |
| Drug class | Antineoplastic agent |
| ATC code | None |
| Legal status | |
| Legal status | US: ℞-only [1] |
| Identifiers | |
| IUPAC name[show] | |
| CAS Number | 497871-47-3 |
| PubChem CID | 57327016 |
| DrugBank | 12674 |
| ChemSpider | 32701856 |
| UNII | 2CN60TN6ZS |
| KEGG | D11644 |
| ChEMBL | ChEMBL4297516 |
| CompTox Dashboard (EPA) | DTXSID30198065 |
| Chemical and physical data | |
| Formula | C41H44N4O10S |
| Molar mass | 784.88 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES[hide]CC1=CC2=C([C@@H]3[C@@H]4[C@H]5C6=C(C(=C7C(=C6[C@@H](N4[C@H]([C@H](C2)N3C)O)COC(=O)[C@@]8(CS5)C9=C(CCN8)C2=C(N9)C=CC(=C2)OC)OCO7)C)OC(=O)C)C(=C1OC)O | |
| InChI[hide]InChI=1S/C41H44N4O10S/c1-17-11-20-12-25-39(48)45-26-14-52-40(49)41(38-22(9-10-42-41)23-13-21(50-5)7-8-24(23)43-38)15-56-37(31(45)30(44(25)4)27(20)32(47)33(17)51-6)29-28(26)36-35(53-16-54-36)18(2)34(29)55-19(3)46/h7-8,11,13,25-26,30-31,37,39,42-43,47-48H,9-10,12,14-16H2,1-6H3/t25-,26-,30+,31+,37+,39-,41+/m0/s1Key:YDDMIZRDDREKEP-HWTBNCOESA-N |
//////////lurbinectedin, FDA 2020, 2020 APPROVALS, ORPHAN, priority review , ZEPZELCA, Pharma Mar, PM-1183, PM 1183, PM 01183, лурбинектедин , لوربينيكتيدين , 芦比替定
Cc1cc2c(c(c1OC)O)[C@@H]3[C@@H]4[C@H]5c6c(c7c(c(c6OC(=O)C)C)OCO7)[C@@H](N4[C@H]([C@H](C2)N3C)O)COC(=O)[C@@]8(CS5)c9c(c1cc(ccc1[nH]9)OC)CCN8
Naxitamab

(Heavy chain)
QVQLVESGPG VVQPGRSLRI SCAVSGFSVT NYGVHWVRQP PGKGLEWLGV IWAGGITNYN
SAFMSRLTIS KDNSKNTVYL QMNSLRAEDT AMYYCASRGG HYGYALDYWG QGTLVTVSSA
STKGPSVFPL APSSKSTSGG TAALGCLVKD YFPEPVTVSW NSGALTSGVH TFPAVLQSSG
LYSLSSVVTV PSSSLGTQTY ICNVNHKPSN TKVDKRVEPK SCDKTHTCPP CPAPELLGGP
SVFLFPPKPK DTLMISRTPE VTCVVVDVSH EDPEVKFNWY VDGVEVHNAK TKPREEQYNS
TYRVVSVLTV LHQDWLNGKE YKCKVSNKAL PAPIEKTISK AKGQPREPQV YTLPPSRDEL
TKNQVSLTCL VKGFYPSDIA VEWESNGQPE NNYKTTPPVL DSDGSFFLYS KLTVDKSRWQ
QGNVFSCSVM HEALHNHYTQ KSLSLSPGK
(Light chain)
EIVMTQTPAT LSVSAGERVT ITCKASQSVS NDVTWYQQKP GQAPRLLIYS ASNRYSGVPA
RFSGSGYGTE FTFTISSVQS EDFAVYFCQQ DYSSFGQGTK LEIKRTVAAP SVFIFPPSDE
QLKSGTASVV CLLNNFYPRE AKVQWKVDNA LQSGNSQESV TEQDSKDSTY SLSSTLTLSK
ADYEKHKVYA CEVTHQGLSS PVTKSFNRGE C
(Disulfide bridge: H22-H95, H146-H202, H222-L211, H228-H’228, H231-H’231, H263-H323, H369-H427, H’22-H’95, H’146-H’202, H’222-L’211, H’263-H’323, H’369-H’427, L23-L88, L131-L191, L’23-L’88, L’131-L’191)
Naxitamab
ナキシタマブ;
Antineoplastic, Anti-GD2 antibody
| Formula | C6414H9910N1718O1996S44 |
|---|---|
| CAS | 1879925-92-4 |
| Mol weight | 144434.4882 |
FDA APPROVED 2020/11/25, Danyelza
FDA grants accelerated approval to naxitamab for high-risk neuroblastoma in bone or bone marrow
On November 25, 2020, the Food and Drug Administration granted accelerated approval to naxitamab (DANYELZA, Y-mAbs Therapeutics, Inc.) in combination with granulocyte-macrophage colony-stimulating factor (GM-CSF) for pediatric patients one year of age and older and adult patients with relapsed or refractory high-risk neuroblastoma in the bone or bone marrow demonstrating a partial response, minor response, or stable disease to prior therapy.
Efficacy was evaluated in patients with relapsed or refractory neuroblastoma in the bone or bone marrow enrolled in two single-arm, open-label trials: Study 201 (NCT 03363373) and Study 12-230 (NCT 01757626). Patients with progressive disease following their most recent therapy were excluded. Patients received 3 mg/kg naxitamab administered as an intravenous infusion on days 1, 3, and 5 of each 4-week cycle in combination with GM-CSF subcutaneously at 250 µg/m2/day on days -4 to 0 and at 500 µg/m2/day on days 1 to 5. At the investigator’s discretion, patients were permitted to receive pre-planned radiation to the primary disease site in Study 201 and radiation therapy to non-target bony lesions or soft tissue disease in Study 12-230.
The main efficacy outcome measures were confirmed overall response rate (ORR) per the revised International Neuroblastoma Response Criteria (INRC) and duration of response (DOR). Among 22 patients treated in the multicenter Study 201, the ORR was 45% (95% CI: 24%, 68%) and 30% of responders had a DOR greater or equal to 6 months. Among 38 patients treated in the single-center Study 12-230, the ORR was 34% (95% CI: 20%, 51%) with 23% of patients having a DOR greater or equal to 6 months. For both trials, responses were observed in either the bone, bone marrow or both.
The prescribing information contains a Boxed Warning stating that naxitamab can cause serious infusion-related reactions and neurotoxicity, including severe neuropathic pain, transverse myelitis and reversible posterior leukoencephalopathy syndrome (RPLS). To mitigate these risks, patients should receive premedication prior to each naxitamab infusion and be closely monitored during and for at least two hours following completion of each infusion.
The most common adverse reactions (incidence ≥25% in either trial) in patients receiving naxitamab were infusion-related reactions, pain, tachycardia, vomiting, cough, nausea, diarrhea, decreased appetite, hypertension, fatigue, erythema multiforme, peripheral neuropathy, urticaria, pyrexia, headache, injection site reaction, edema, anxiety, localized edema, and irritability. The most common Grade 3 or 4 laboratory abnormalities (≥5% in either trial) were decreased lymphocytes, decreased neutrophils, decreased hemoglobin, decreased platelet count, decreased potassium, increased alanine aminotransferase, decreased glucose, decreased calcium, decreased albumin, decreased sodium and decreased phosphate.
The recommended naxitamab dose is 3 mg/kg/day (up to 150 mg/day) on days 1, 3, and 5 of each treatment cycle, administered after dilution as an intravenous infusion in combination with GM-CSF, subcutaneously at 250 µg/m2/day on days -4 to 0 and at 500 µg/m2/day on days 1 to 5. Treatment cycles are repeated every 4 to 8 weeks.
View full prescribing information for DANYELZA. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/761171lbl.pdf
This review used the Real-Time Oncology Review (RTOR) pilot program and the Assessment Aid, a voluntary submission from the applicant to facilitate the FDA’s assessment.
This application was granted accelerated approval based on overall response rate and duration of response. Continued approval may be contingent upon verification and description of clinical benefit in confirmatory trials.
This application was granted priority review, breakthrough therapy, and orphan drug designation. A priority review voucher was issued for this rare pediatric disease product application. A description of FDA expedited programs is in the Guidance for Industry: Expedited Programs for Serious Conditions-Drugs and Biologics.
////////////Naxitamab, priority review, breakthrough therapy, orphan drug, FDA 2020, 2020 APPROVALS, Danyelza, MONOCLONAL ANTIBODY, PEPTIDE, ナキシタマブ,
Ansuvimab-zykl

Ansuvimab-zykl
FDA APPROVED, 12/21/2020, EBANGA
To treat ebola
https://www.fda.gov/drugs/drug-safety-and-availability/fda-approves-treatment-ebola-virus
The U.S. Food and Drug Administration approved Ebanga (Ansuvimab-zykl), a human monoclonal antibody, for the treatment for Zaire ebolavirus (Ebolavirus) infection in adults and children. Ebanga blocks binding of the virus to the cell receptor, preventing its entry into the cell.
Zaire ebolavirus is one of four Ebolavirus species that can cause a potentially fatal human disease. It is transmitted through blood, body fluids, and tissues of infected people or wild animals, and through surfaces and materials, such as bedding and clothing, contaminated with these fluids. Individuals who care for people with the disease, including health care workers who do not use correct infection control precautions, are at the highest risk for infection.
During an Ebola outbreak in the Democratic Republic of the Congo (DRC) in 2018-2019, Ebanga was evaluated in a clinical trial (the PALM trial). The PALM trial was led by the U.S. National Institutes of Health and the DRC’s Institut National de Recherche Biomédicale with contributions from several other international organizations and agencies.
In the PALM trial, the safety and efficacy of Ebanga was evaluated in a multi-center, open-label, randomized controlled trial. 174 participants (120 adults and 54 pediatric patients) with confirmed Ebolavirus infection received Ebanga intravenously as a single 50 mg/kg infusion and 168 participants (135 adults and 33 pediatric patients) received an investigational control. The primary efficacy endpoint was 28-day mortality. The primary analysis population was all patients who were randomized and concurrently eligible to receive either Ebanga or the investigational control during the same time period of the trial. Of the 174 patients who received Ebanga, 35.1% died after 28 days, compared to 49.4% of the 168 patients who received a control.
The most common symptoms experienced while receiving Ebanga include: fever, tachycardia (fast heart rate), diarrhea, vomiting, hypotension (low blood pressure), tachypnea (fast breathing) and chills; however, these are also common symptoms of Ebolavirus infection. Hypersensitivity, including infusion-related events, can occur in patients taking Ebanga, and treatment should be discontinued in the event of a hypersensitivity reaction.
Patients who receive Ebanga should avoid the concurrent administration of a live virus vaccine against Ebolavirus. There is the potential for Ebanga to inhibit replication of a live vaccine virus and possibly reduce the efficacy of this vaccine.
Ebanga was granted an Orphan Drug designation, which provides incentives to assist and encourage drug development for rare diseases. Additionally, the agency granted Ebanga a Breakthrough Therapy designation.
FDA granted the approval to Ridgeback Biotherapeutics, LP.
Ansuvimab, sold under the brand name Ebanga, is a monoclonal antibody medication for the treatment of Zaire ebolavirus (Ebolavirus) infection.[1][2]
The most common symptoms include fever, tachycardia (fast heart rate), diarrhea, vomiting, hypotension (low blood pressure), tachypnea (fast breathing) and chills; however, these are also common symptoms of Ebolavirus infection.[1]
Ansuvimab was approved for medical use in the United States in December 2020.[1][2]
Chemistry
The drug is composed of a single monoclonal antibody (mAb) and was initially isolated from immortalized B-cells that were obtained from a survivor of the 1995 outbreak of Ebola virus disease in Kikwit, Democratic Republic of Congo.[3] In work supported by the United States National Institutes of Health and the Defense Advanced Projects Agency, the heavy and light chain sequences of ansuvimab mAb was cloned into CHO cell lines and initial production runs were produced by Cook Phamica d.b.a. Catalent under contract of Medimmune.[4][5]
Mechanism of action
Neutralization
Ansuvimab is a monoclonal antibody therapy that is infused intravenously into patients with Ebola virus disease. Ansuvimab is a neutralizing antibody,[3] meaning it binds to a protein on the surface of Ebola virus that is required to infect cells. Specifically, ansuvimab neutralizes infection by binding to a region of the Ebola virus envelope glycoprotein that, in the absence of ansuvimab, would interact with virus’s cell receptor protein, Niemann-Pick C1 (NPC1).[6][7][8] This “competition” by ansuvimab prevents Ebola virus from binding to NPC1 and “neutralizes” the virus’s ability to infect the targeted cell.[6]
Effector function
Antibodies have antigen-binding fragment (Fab) regions and constant fragment (Fc) regions. The Neutralization of virus infection occurs when the Fab regions of antibodies binds to virus antigen(s) in a manner that blocks infection. Antibodies are also able to “kill” virus particles directly and/or kill infected cells using antibody-mediated “effector functions” such as opsonization, complement-dependent cytotoxicity, antibody-dependent cell-mediated cytotoxicity and antibody-dependent phagocytosis. These effector functions are contained in the Fc region of antibodies, but is also dependent on binding of the Fab region to antigen. Effector functions also require the use of complement proteins in serum or Fc-receptor on cell membranes. Ansuvimab has been found to be capable of killing cells by antibody-dependent cell-mediated cytotoxicity.[3] Other functional killing tests have not been performed.
History
Ansuvimab is a monoclonal antibody that is being evaluated as a treatment for Ebola virus disease.[9] Its discovery was led by the laboratory of Nancy Sullivan at the United States National Institute of Health Vaccine Research Center and J. J. Muyembe-Tamfum from the Institut National pour la Recherche Biomedicale (INRB) in the Democratic Republic of Congo, working in collaboration with the Institute of Biomedical Research and the United States Army Medical Research Institute of Infectious Diseases.[3][10] Ansuvimab was isolated from the blood of a survivor of the 1995 outbreak of Ebola virus disease in Kikwit, Democratic Republic of Congo roughly ten years later.[3]
In 2018, a Phase 1 clinical trial of ansuvimab was conducted by Martin Gaudinski within the Vaccine Research Center Clinical Trials Program that is led by Julie E. Ledgerwood.[5][4][11] Ansuvimab is also being evaluated during the 2018 North Kivu Ebola outbreak.[12]
Ansuvimab has also shown success with lowering the mortality rate from ~70% to about 34%. In August 2019, Congolese health authorities, the World Health Organization, and the U.S. National Institutes of Health promoted the use of ansuvimab, alongside REGN-EB3, a similar Regeneron-produced monoclonal antibody treatment, over other treatments yielding higher mortality rates, after ending clinical trials during the outbreak.[13][14]
Discovery
A 2016 paper describes the efforts of how ansuvimab was originally developed as part of research efforts lead by Dr. Nancy Sullivan at the United States National Institute of Health Vaccine Research Center and Dr. J. J. Muyembe-Tamfum from the Institut National de Recherche Biomedicale (INRB) in the Democratic Republic of Congo.[3][10] This collaborative effort also involved researchers from Institute of Biomedical Research and the United States Army Medical Research Institute of Infectious Diseases.[3][10] A survivor from the 1995 outbreak of Ebola virus disease in Kikwit, Democratic Republic of Congo donated blood to the project that began roughly ten years after they had recovered.[3] Memory B cells isolated from the survivor’s blood were immortalized, cultured and screened for their ability to produce monoclonal antibodies that reacted with the glycoprotein of Ebola virus. Ansuvimab was identified from one of these cultures and the antibody heavy and light chain gene sequences were sequenced from the cells.[3] These sequences were then cloned into recombinant DNA plasmids and purified antibody protein for initial studies was produced in cells derived from HEK 293 cells.[3]
Ansuvimab and mAb100 combination
In an experiment described in the 2016 paper, rhesus macaques were infected with Ebola virus and treated with a combination of ansuvimab and another antibody isolated from the same subject, mAb100. Three doses of the combination were given once a day starting 1 day after the animals were infected. The control animal died and the treated animals all survived.[3]
Ansuvimab monotherapy
In a second experiment described in the 2016 paper, rhesus macaques were infected with Ebola virus and only treated with ansuvimab. Three doses of ansuvimab were given once a day starting 1 day or 5 days after the animals were infected. The control animals died and the treated animals all survived.[3] Unpublished data referred to in a publication of the 2018 Phase I clinical trial results of ansuvimab, reported that a single infusion of ansuvimab provided full protection of rhesus macaques and was the basis of the dosing used for human studies.[5][4]
Development
Ansuvimab was developed by the Vaccine Research Center with support of the United States National Institutes of Health and the Defense Advanced Projects Agency. The heavy and light chain sequences of ansuvimab mAb were cloned into CHO cell lines to enable large-scale production of antibody product for use in humans.[4][5]
Human safety testing
In early 2018,[9] a Phase 1 clinical trial of ansuvimab’s safety, tolerability and pharmacokinetics was conducted by Dr. Martin Gaudinski within the Vaccine Research Center Clinical Trials Program that is led by Dr. Julie E. Ledgerwood.[5][4][11] The study was performed in the United States at the NIH Clinical Center and tested single dose infusions of ansuvimab infused over 30 minutes. The study showed that ansuvimab was safe, had minimal side effects and had a half-life of 24 days.[5][4]
Ridgeback Biotherapeutics
A license for ansuvimab was obtained by Ridgeback Biotherapeutics in 2018, from the National Institutes of Health–National Institute of Allergy and Infectious Diseases.[15] Ansuvimab was given orphan drug status in May 2019 and March 2020.[16][17][18]
Experimental use in the Democratic Republic of Congo
During the 2018 Équateur province Ebola outbreak, ansuvimab was requested by the Democratic Republic of Congo (DRC) Ministry of Public Health. Ansuvimab was approved for compassionate use by the World Health Organization MEURI ethical protocol and at DRC ethics board. Ansuvimab was sent along with other therapeutic agents to the outbreak sites.[19][20][11] However, the outbreak came to a conclusion before any therapeutic agents were given to patients.[11]
Approximately one month following the conclusion of the Équateur province outbreak, a distinct outbreak was noted in Kivu in the DRC (2018–20 Kivu Ebola outbreak). Once again, ansuvimab received approval for compassionate use by WHO MEURI and DRC ethic boards and has been given to many patients under these protocols.[11] In November 2018, the Pamoja Tulinde Maisha (PALM [together save lives]) open-label randomized clinical control trial was begun at multiple treatment units testing ansuvimab, REGN-EB3 and remdesivir to ZMapp. Despite the difficulty of running a clinical trial in a conflict zone, investigators have enrolled 681 patients towards their goal of 725. An interim analysis by the Data Safety and Monitoring Board (DSMB) of the first 499 patient found that ansuvimab and REGN-EB3 were superior to the comparator ZMapp. Overall mortality of patients in the ZMapp and remdesivir groups were 49% and 53% compared to 34% and 29% for ansuvimab and REGN-EB3. When looking at patients who arrived early after disease symptoms appeared, survival was 89% for ansuvimab and 94% for REGN-EB3. While the study was not powered to determine whether there is any difference between REGN-EB3 and ansuvimab, the survival difference between those two therapies and ZMapp was significant. This led to the DSMB halting the study and PALM investigators dropping the remdesivir and ZMapp arms from the clinical trial. All patients in the outbreak who elect to participate in the trial will now be given either ansuvimab or REGN-EB3.[21][22][13][12]
In October 2020, the U.S. Food and Drug Administration (FDA) approved atoltivimab/maftivimab/odesivimab (Inmazeb, formerly REGN-EB3) with an indication for the treatment of infection caused by Zaire ebolavirus.[23]
FDA approves ansuvimab-zykl for Ebola virus infection
DECEMBER 21, 2020 BY JANICE REICHERThttps://www.antibodysociety.org/antibody-therapeutic/fda-approves-ansuvimab-zykl-for-ebola-virus-infection/embed/#?secret=zWW0Sr0BdW
On December 21, 2020, the US Food and Drug Administration approved Ebanga (ansuvimab-zykl) for the treatment for Zaire ebolavirus (Ebolavirus) infection in adults and children. Ebanga had been granted US Orphan Drug designation and Breakthrough Therapy designations. Ansuvimab is a human IgG1 monoclonal antibody that binds and neutralizes the virus.
The safety and efficacy of Ebanga were evaluated in the multi-center, open-label, randomized controlled PALM trial. In this study, 174 participants (120 adults and 54 pediatric patients) with confirmed Ebolavirus infection received Ebanga intravenously as a single 50 mg/kg infusion and 168 participants (135 adults and 33 pediatric patients) received an investigational control. The primary efficacy endpoint was 28-day mortality. Of the 174 patients who received Ebanga, 35.1% died after 28 days, compared to 49.4% of the 168 patients who received a control.
Ebanga is the 12th antibody therapeutic to be granted a first approval in the US or EU during 2020.
The Antibody Society maintains a comprehensive table of approved monoclonal antibody therapeutics and those in regulatory review in the EU or US. The table, which is located in the Web Resources section of the Society’s website, can be downloaded in Excel format.
References
- ^ Jump up to:a b c d “FDA Approves Treatment for Ebola Virus”. U.S. Food and Drug Administration. 21 December 2020. Retrieved 23 December 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Jump up to:a b “Ridgeback Biotherapeutics LP Announces the Approval of Ebanga for Ebola” (Press release). Ridgeback Biotherapeutics LP. 22 December 2020. Retrieved 23 December 2020– via Business Wire.
- ^ Jump up to:a b c d e f g h i j k l Corti D, Misasi J, Mulangu S, Stanley DA, Kanekiyo M, Wollen S, et al. (March 2016). “Protective monotherapy against lethal Ebola virus infection by a potently neutralizing antibody”. Science. 351 (6279): 1339–42. Bibcode:2016Sci…351.1339C. doi:10.1126/science.aad5224. PMID 26917593.
- ^ Jump up to:a b c d e f Clinical trial number NCT03478891 for “Safety and Pharmacokinetics of a Human Monoclonal Antibody, VRC-EBOMAB092-00-AB (MAb114), Administered Intravenously to Healthy Adults” at ClinicalTrials.gov
- ^ Jump up to:a b c d e f Gaudinski MR, Coates EE, Novik L, Widge A, Houser KV, Burch E, et al. (March 2019). “Safety, tolerability, pharmacokinetics, and immunogenicity of the therapeutic monoclonal antibody ansuvimab targeting Ebola virus glycoprotein (VRC 608): an open-label phase 1 study”. Lancet. 393 (10174): 889–898. doi:10.1016/S0140-6736(19)30036-4. PMC 6436835. PMID 30686586.
- ^ Jump up to:a b Misasi J, Gilman MS, Kanekiyo M, Gui M, Cagigi A, Mulangu S, et al. (March 2016). “Structural and molecular basis for Ebola virus neutralization by protective human antibodies”. Science. 351 (6279): 1343–6. Bibcode:2016Sci…351.1343M. doi:10.1126/science.aad6117. PMC 5241105. PMID 26917592.
- ^ Côté M, Misasi J, Ren T, Bruchez A, Lee K, Filone CM, et al. (August 2011). “Small molecule inhibitors reveal Niemann-Pick C1 is essential for Ebola virus infection”. Nature. 477 (7364): 344–8. Bibcode:2011Natur.477..344C. doi:10.1038/nature10380. PMC 3230319. PMID 21866101.
- ^ Carette JE, Raaben M, Wong AC, Herbert AS, Obernosterer G, Mulherkar N, et al. (August 2011). “Ebola virus entry requires the cholesterol transporter Niemann-Pick C1”. Nature. 477 (7364): 340–3. Bibcode:2011Natur.477..340C. doi:10.1038/nature10348. PMC 3175325. PMID 21866103.
- ^ Jump up to:a b “NIH begins testing Ebola treatment in early-stage trial”. National Institutes of Health (NIH). 2018-05-23. Retrieved 2018-10-15.
- ^ Jump up to:a b c Hayden EC (2016-02-26). “Ebola survivor’s blood holds promise of new treatment”. Nature. doi:10.1038/nature.2016.19440. ISSN 1476-4687.
- ^ Jump up to:a b c d e “NIH VideoCast – CC Grand Rounds: Response to an Outbreak: Ebola Virus Monoclonal Antibody (mAb114) Rapid Clinical Development”. videocast.nih.gov. Retrieved 2019-08-09.
- ^ Jump up to:a b Kingsley-Hall A. “Congo’s experimental mAb114 Ebola treatment appears successful: authorities | Central Africa”. http://www.theafricareport.com. Retrieved 2018-10-15.
- ^ Jump up to:a b McNeil DG (12 August 2019). “A Cure for Ebola? Two New Treatments Prove Highly Effective in Congo”. The New York Times. Retrieved 13 August 2019.
- ^ Molteni M (12 August 2019). “Ebola is Now Curable. Here’s How The New Treatments Work”. Wired. Retrieved 13 August 2019.
- ^ “Ridgeback Biotherapeutics LP announces licensing of mAb114, an experimental Ebola treatment, from the National Institute of Allergy and Infectious Diseases” (Press release). Ridgeback Biotherapeutics LP. Retrieved 2019-08-17 – via PR Newswire.
- ^ “Ansuvimab Orphan Drug Designations and Approvals”. accessdata.fda.gov. 8 May 2019. Retrieved 24 December 2020.
- ^ “Ansuvimab Orphan Drug Designations and Approvals”. accessdata.fda.gov. 30 March 2020. Retrieved 24 December 2020.
- ^ “Ridgeback Biotherapeutics LP Announces Orphan Drug Designation for mAb114”(Press release). Ridgeback Biotherapeutics LP. Retrieved 2019-08-17 – via PR Newswire.
- ^ Check Hayden, Erika (May 2018). “Experimental drugs poised for use in Ebola outbreak”. Nature. 557 (7706): 475–476. Bibcode:2018Natur.557..475C. doi:10.1038/d41586-018-05205-x. ISSN 0028-0836. PMID 29789732.
- ^ WHO: Consultation on Monitored Emergency Use of Unregistered and Investigational Interventions for Ebola virus Disease. https://www.who.int/emergencies/ebola/MEURI-Ebola.pdf
- ^ Mole B (2019-08-13). “Two Ebola drugs boost survival rates, according to early trial data”. Ars Technica. Retrieved 2019-08-17.
- ^ “Independent monitoring board recommends early termination of Ebola therapeutics trial in DRC because of favorable results with two of four candidates”. National Institutes of Health (NIH). 2019-08-12. Retrieved 2019-08-17.
- ^ “FDA Approves First Treatment for Ebola Virus”. U.S. Food and Drug Administration(FDA) (Press release). 14 October 2020. Retrieved 14 October 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
External links
- “Ansuvimab”. Drug Information Portal. U.S. National Library of Medicine.
| Monoclonal antibody | |
|---|---|
| Type | Whole antibody |
| Source | Human |
| Target | Zaire ebolavirus |
| Clinical data | |
| Trade names | Ebanga |
| Other names | Ansuvimab-zykl, mAb114 |
| License data | US DailyMed: Ansuvimab |
| Routes of administration | Intravenous |
| Drug class | Monoclonal antibody |
| ATC code | None |
| Legal status | |
| Legal status | US: ℞-only [1] |
| Identifiers | |
| CAS Number | 2375952-29-5 |
| DrugBank | DB16385 |
| UNII | TG8IQ19NG2 |
| KEGG | D11875 |
| Chemical and physical data | |
| Formula | C6368H9924N1724O1994S44 |
| Molar mass | 143950.15 g·mol−1 |
//////////Ansuvimab-zykl , EBANGA, FDA 2020, 2020 APPROVALS, MONOCLONAL ANTIBODY, Orphan Drug designation, , Breakthrough Therapy designation , Ridgeback Biotherapeutics,
Tirbanibulin
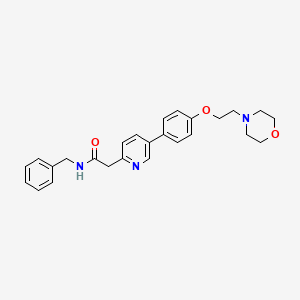
Tirbanibulin
CAS 897016-82-9, 1038395-65-1 DI HCL
1080645-95-9 MESYLATE
N-benzyl-2-[5-[4-(2-morpholin-4-ylethoxy)phenyl]pyridin-2-yl]acetamide
| Molecular Weight | 431.53 |
|---|---|
| Formula | C₂₆H₂₉N₃O₃ |
FDA APPROVED 12/14/2020, Klisyri
To treat actinic Keratosis of the face or scalp
Tirbanibulin (KX2-391) is an inhibitor of Src that targets the peptide substrate site of Src, with GI50 of 9-60 nM in cancer cell lines.
- Originator Kinex Pharmaceuticals
- Developer Almirall S.A.; Athenex; Hanmi Pharmaceutical; Kinex Pharmaceuticals; PharmaEssentia Corporation
- ClassAcetamides; Amides; Antineoplastics; Antipsoriatics; Morpholines; Phenyl ethers; Pyridines; Skin disorder therapies; Small molecules
- Mechanism of ActionAngiogenesis inhibitors; Src-Family kinase inhibitors; Tubulin polymerisation inhibitors
- PreregistrationActinic keratosis
- Phase IIPsoriasis
- Phase I/IISolid tumours
- Phase IPhotodamage
- PreclinicalSkin cancer
- 09 Mar 2020FDA assigns PDUFA action date of 30/12/2020 for tirbanibulin for Actinic keratosis
- 09 Mar 2020US FDA accepts NDA for tirbanibulin for Actinic keratosis for review
- 02 Mar 2020European Medicines Agency accepts Marketing Authorization Application for tirbanibulin for Actinic keratosis for review
KX-01 is a dual inhibitor of Src kinase and tubulin polymerization. KX01 promotes the induction of p53, G2/M arrest of proliferating cell populations and subsequent apoptosis via the stimulation of Caspase-3 and PARP cleavage. The drug was developed by Kinex Pharmaceuticals and reached phase II of clinical trials for the treatment of Castration-Resistant Prostate Cancer and Actinic Keratosis. KX-01 demonstrated good in vitro pofile against different cancer cell lines with IC50 in nanomolar range.
Tirbanibulin (Mesylate) (KX2-391 (Mesylate)) is an inhibitor of Src that targets the peptide substrate site of Src, with GI50 of 9-60 nM in cancer cell lines.
Tirbanibulin (KX2-391) is a Src inhibitor that is directed to the Src substrate pocket. Tirbanibulin (KX2-391) shows steep dose-response curves against Huh7 (GI50=9 nM), PLC/PRF/5 (GI50=13 nM), Hep3B (GI50=26 nM), and HepG2 (GI50=60 nM), four hepatic cell cancer (HCC) cell lines[1]. Tirbanibulin (KX2-391) is found to inhibit certain leukemia cells that are resistant to current commercially available drugs, such as those derived from chronic leukemia cells with the T3151 mutation. Tirbanibulin (KX2-391) is evaluated in engineered Src driven cell growth assays inNIH3T3/c-Src527F and SYF/c-Src527F cells and exhibits GI50 with 23 nM and 39 nM, respectively[2].
Orally administered Tirbanibulin (KX2-391) is shown to inhibit primary tumor growth and to suppress metastasis, in pre-clinical animal models of cancer[2].
[1]. Lau GM, et al. Expression of Src and FAK in hepatocellular carcinoma and the effect of Src inhibitors on hepatocellular carcinoma in vitro. Dig Dis Sci, 2009, 54(7), 1465-1474. [2]. Fallah-Tafti A, et al. Thiazolyl N-benzyl-substituted acetamide derivatives: synthesis, Src kinase inhibitory and anticancer activities. Eur J Med Chem, 2011, 46(10), 4853-4858.

Approval allows Almirall to move forward with the topical ointment for individuals with AK on the face or scalp.

The US Food and Drug Administration (FDA) has approved tirbanibulin (Klisyri) as a topical treatment for actinic keratosis (AK).
The approval, awarded to Almirall, S.A., will allow the novel, topical first-in-class microtubule inhibitor for treatment of the disease on the face or scalp, representing a significant breakthrough in treatment of AK because of its short treatment protocol of once daily application for 5 days.
Actinic keratosis represents the second most common diagnosis in dermatology in the US, with a reported prevalence between 11-25%.
“Early diagnosis and treatment of actinic keratosis (AK) is critical, since those who already have an AK are likely to develop more actinic keratoses (plural) in the future,” said Deborah S. Sarnoff, MD, President of the Skin Cancer Foundation, said in a statement. “Patients with AK are at higher risk for skin cancer, since AKs can progress into squamous cell carcinoma (SCC), a common and sometimes invasive form of skin cancer.”
The approval is based on recent data from a large phase 3 clinical study, as well as 2 randomized, double-blind, vehicle-controlled phase 3 studies evaluating the efficacy and safety of tirbanibulin ointment 1% in adults with AK on the face or scalp.
“These studies enrolled a total of 702 patients across 62 sites in the United States, providing robust data,” Andrew Blauvelt, MD, MBA, President of Oregon Medical Research Center, and one of the lead investigators of the studies, said in a statement. “Tirbanibulin achieved a significantly higher number of patients with complete (100%) clearance of AK lesions in the treated area compared to vehicle (44% vs. 5% in study 1 and 54% vs. 13% in study 2), as well as reaching the secondary endpoint of partial (≥75%) clearance of lesions.”
PATENT
WO 2006071960
US 20070015752
US 20080287436
WO 2008082637
WO 2008002676
US 20090318450
https://patents.google.com/patent/US20090318450A1/en
- .
- [0374]A 1 L single-necked round-bottomed flask was charged with 7 (61.4 g, 0.172 mol), benzyl amine (55.6 g, 0.519 mol, 3 eq), and anhydrous anisole (300 g) and then stirred at reflux until reaction was essentially complete (23 h, 165° C. oil bath temperature; internal temperature was 147° C.) and then allowed to cool to near room temperature. A portion (1 mL) of the reaction mixture was diluted with toluene (1 mL) resulting in the complete crystallization of that portion. This seed was then added to the reaction mixture and allowed to stand until the whole reaction mixture had crystallized to a single block. Toluene (150 mL) was added and the mixture swirled to break up the solid. Heptane/toluene (1:1, 100 mL) was added and the solid mixture broken up further. Finally, heptane (50 mL, then 25 mL) was added and the mixture broken up even further, allowing to stand an additional 30 min before filtering the solid. Filtration of the solid, washing with 2:1 toluene/heptane (300 mL), 1:2 toluene/heptane (300 mL), and then heptane (2×300 mL), and then drying (air, then high vac) gave 60.16 g (yield of 81%) of title product as a white solid (>98.9% AUC). Another 2.5 g of less pure (97.4%) material was obtained from the mother liquors.
- [0375]1H NMR (CDCl3) δ 2.60 (t, 4 H), 2.83 (t, 2 H), 3.74 (t, 4 H), 3.82 (s, 2 H), 4.18 (t, 2 H), 4.49 (d, 2 H), 7.01 (d, 2 H), 7.2-7.35 (m, 6 H), 7.49 (d, 2 H), 7.64 (br t, 1 H), 7.81 (dd, 1 H), 8.69 (fine d, 1 H). MS (from LC/MS): m/z 432.5 [M+1].
- [0376]To a stirred suspension of KX2-391 (free base, 60.00 g) in absolute EtOH (600 mL) was added 170 mL of 2.5 M HCl (in ethanol), 25 mL EtOH being added to wash down the sides of the flask. The resulting homogeneous solution was stirred at room temperature (20 min) and then evaporated to near dryness (to frothing). After chasing with EtOH (2×150 mL), the residue was taken up again in EtOH (150 mL) and then was followed by the slow addition of heptane until the mixture appeared saturated (33 mL required for cloudiness to remain). After sitting overnight, two layers had formed. After adding additional heptane (250 mL) crystallization still could not be induced and so the reaction mixture was concentrated to a volume of ˜200 mL at which time the mixture was homogeneous. This thick homogeneous solution was added dropwise to very rapidly stirred (mechanical) EtOAc (2 L). After the addition was complete, a 25 mL EtOH rinse of the original flask and addition funnel was added to the rapidly stirred mixture. The rapid stirring was continued for another ˜1 h and then the mixture was filtered and the solid (partly gummy) was washed with EtOAc (300 mL) and then heptane. As soon as the heptane wash began, the solid got much gummier. The fritted Buchner funnel and its contents were covered (paper towel/rubber band) and immediately placed in the vacuum oven. After overnight vacuum at ˜45° C., the vacuum was released under nitrogen, and the Buchner funnel containing the product (foamy solid) was immediately placed in a zip-lock back and then, under nitrogen (glove bag), transferred to a bottle and the foamy solid broken up (spatula) to a powder. A second night under high vacuum (˜45° C.) resulted in only 1.3 g of additional weight loss. Constant weight was essentially attained with the third night of high vacuum (˜45° C.) where only 0.2 g of weight was lost. The final weight of material was 68.05 g (yield of 97%), containing 0.29 eq (4.8% w/w) of EtOAc, 0.035 eq (0.3% w/w) EtOH, and 0.03 eq (0.6% w/w) heptane. The purity was 99.6%.
- [0377]1H NMR (DMSO-d6) δ 3.1-3.3 (m, 2 H), 3.45-3.65 (m, 4 H), 3.8-4.0 (m, 4 H), 4.11 (s, 2 H), 4.32 (d, 2 H), 4.57 (t, 2 H), 7.19 (d, 2 H), 7.2-7.4 (m, 5 H), 7.88 (d, 2 H), 7.93 (d, 1 H), 8.68 (dd, 1 H), 8.99 (br t, 1 H), 9.10 (fine d, 1 H), 11.8 (br s, 1 H). MS (from LC/MS): m/z 432.5 [M+1 of free base].
- [0378]Elemental analysis (for C26H29N3O3.2 HCl.0.035 EtOH.0.29.EtOAc.0.03 heptane.0.8 H2O):
- [0379]Calculated (%): C, 60.03; H, 6.54; N, 7.65; Cl, 12.91
- [0380]Observed (%):C, 59.85/59.97; H, 6.54/6.47; N, 7.67/7.67; Cl, 13.10/13.24
- [0381]Calculated FW: 534.63 (does not take into account the 0.8 H2O which probably arose during handling of this very hygroscopic powder, since 1H NMR shows no evidence for H2O).
- [0382]The ethyl chloride level in this material was measured and found to be 98 ppm. The sample was also analyzed and found to contain 5,800 ppm of heptane.
- [0383]Analysis of another portion of this sample yielded the following results: 99.6% AUC, 1640 ppm ethanol, 41,480 ppm ethyl acetate, 5600 ppm heptane, no anisole detected, and 120 ppm ethyl chloride.
- [0384]A procedure for recrystallizing the salt was also developed using the above dried salt. This procedure would work just was well on the highly pure crude salt (containing residual EtOH) obtained from concentrating the HCl salt-forming reaction mixture:
- [0385]The salt (575 mg) was dissolved in twice the mass of absolute EtOH (1.157 g) and then heated under nitrogen. To this hot solution (stirred) was added 1.6 g of 25% EtOH (in EtOAc) followed by the addition of EtOAc (0.25 mL) resulting in a cloudiness that remained. The cloudy hot solution was allowed to cool to room temperature during which time crystallization occurred. After crystallization was complete (2 h), the crystalline solid was filtered, washed with anhydrous EtOAc (˜40 mL), and vacuum dried to give 424 mg of the dihydrochloride salt of KX2-391 as a free-flowing solid (tiny beads, 99.8% AUC) containing only 0.05 eq (0.45% w/w) of EtOH and 0.015 eq (0.26% w/w) of EtOAc. Slightly better recovery (460 mg from 586 mg) was attained using isopropanol/EtOAc but the level of solvent entrapment was higher [0.085 eq (1.0% w/w) of isopropanol and 0.023 eq (0.4% w/w) of EtOAc].
PATENT
WO 2009051848
https://patents.google.com/patent/WO2009051848A1/en
].

Preparation of 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2-yl)-N-benzylacetamide (Compound (I) free base).
[ 000242 ] A l L single-necked round-bottomed flask was charged with 7 (61.4 g, 0.172 mol), benzyl amine (55.6 g, 0.519 mol, 3 eq), and anhydrous anisole (300 g) and then stirred at reflux until reaction was essentially complete (23 h, 165 0C oil bath temperature; internal temperature was 147 0C) and then allowed to cool to near room temperature. A portion (1 mL) of the reaction mixture was diluted with toluene (1 mL) resulting in the complete crystallization of that portion. This seed was then added to the reaction mixture and allowed to stand until the whole reaction mixture had crystallized to a single block. Toluene (150 mL) was added and the mixture swirled to break up the solid. Heptane/toluene (1:1, 100 mL) was added and the solid mixture broken up further. Finally, heptane (50 mL, then 25 mL) was added and the mixture broken up even further, allowing to stand an additional 30 min before filtering the solid. Filtration of the solid, washing with 2:1 toluene/heptane (300 mL), 1:2 toluene/heptane (300 mL), and then heptane (2 x 300 mL), and then drying (air, then high vac) gave 60.16 g (yield of 81%) of title product as a white solid (>98.9% AUC). Another 2.5 g of less pure (97.4%) material was obtained from the mother liquors.
[000243 ] 1H NMR (CDCl3) δ 2.60 (t, 4 H), 2.83 (t, 2 H), 3.74 (t, 4 H), 3.82 (s, 2 H), 4.18 (t, 2 H), 4.49 (d, 2 H), 7.01 (d, 2 H), 7.2-7.35 (m, 6 H), 7.49 (d, 2 H), 7.64 (br t, 1 H), 7.81 (dd, 1 H), 8.69 (fine d, 1 H). MS (from LC/MS): m/z 432.5 [M + I].

Preparation of 4-(2-(4-(6-(2-(benzylamino)-2-oxoethyl)pyridinium-3-yl)phenoxy)ethyl)- morpholin-4-ium chloride (Compound (I), diHCI salt).
[000244 ] To a stirred suspension of compound (I) (free base, 60.00 g) in absolute EtOH (600 mL) was added 170 mL of 2.5 M HCl (in ethanol), 25 mL EtOH being added to wash down the sides of the flask. The resulting homogeneous solution was stirred at room temperature (20 min) and then evaporated to near dryness (to frothing). After chasing with EtOH (2 x 150 mL), the residue was taken up again in EtOH (150 mL) and then was followed by the slow addition of heptane until the mixture appeared saturated (33 mL required for cloudiness to remain). After sitting overnight, two layers had formed. After adding additional heptane (250 mL) crystallization still could not be induced and so the reaction mixture was concentrated to a volume of -200 mL at which time the mixture was homogeneous. This thick homogeneous solution was added dropwise to very rapidly stirred (mechanical) EtOAc (2 L). After the addition was complete, a 25 mL EtOH rinse of the original flask and addition funnel was added to the rapidly stirred mixture. The rapid stirring was continued for another ~1 h and then the mixture was filtered and the solid (partly gummy) was washed with EtOAc (300 mL) and then heptane. As soon as the heptane wash began, the solid got much gummier. The fritted Buchner funnel and its contents were covered (paper towel/rubber band) and immediately placed in the vacuum oven. After overnight vacuum at -45 0C, the vacuum was released under nitrogen, and the Buchner funnel containing the product (foamy solid) was immediately placed in a zip-lock back and then, under nitrogen (glove bag), transferred to a bottle and the foamy solid broken up (spatula) to a powder. A second night under high vacuum (-45 0C) resulted in only 1.3 g of additional weight loss. Constant weight was essentially attained with the third night of high vacuum (-45 0C) where only 0.2 g of weight was lost. The final weight of material was 68.05 g (yield of 97%), containing 0.29 eq (4.8% w/w) of EtOAc, 0.035 eq (0.3% w/w) EtOH, and 0.03 eq (0.6% w/w) heptane. The purity was 99.6%.
[000245] 1H NMR (DMSO-Cl6) δ 3.1-3.3 (m, 2 H), 3.45-3.65 (m, 4 H), 3.8-4.0 (m, 4 H), 4.11 (s, 2 H), 4.32 (d, 2 H), 4.57 (t, 2 H), 7.19 (d, 2 H), 7.2-7.4 (m, 5 H), 7.88 (d, 2 H), 7.93 (d, 1 H), 8.68 (dd, 1 H), 8.99 (br t, 1 H), 9.10 (fine d, 1 H), 11.8 (br s, 1 H). MS (from LC/MS): m/z 432.5 [M + 1 of free base].
[000246] Elemental analysis (for C26H29N3O3 • 2 HCl • 0.035 EtOH • 0.29 EtOAc • 0.03 heptane • 0.8 H2O): a. Calculated (%): C, 60.03; H, 6.54; N, 7.65; Cl, 12.91 b. Observed (%):C, 59.85/59.97; H, 6.54/6.47; N, 7.67/7.67; Cl, 13.10/13.24
[ 000247] Calculated FW: 534.63 (does not take into account the 0.8 H2O which probably arose during handling of this very hygroscopic powder, since 1H NMR shows no evidence for H2O).
[ 000248] The ethyl chloride level in this material was measured and found to be 98 ppm. The sample was also analyzed and found to contain 5,800 ppm of heptane.
[000249] Analysis of another portion of this sample yielded the following results: 99.6% AUC, 1640 ppm ethanol, 41,480 ppm ethyl acetate, 5600 ppm heptane, no anisole detected, and 120 ppm ethyl chloride.
[000250] A procedure for recrystallizing the salt was also developed using the above dried salt. This procedure would work just was well on the highly pure crude salt (containing residual EtOH) obtained from concentrating the HCl salt-forming reaction mixture:
[000251] The salt (575 mg) was dissolved in twice the mass of absolute EtOH (1.157 g) and then heated under nitrogen. To this hot solution (stirred) was added 1.6 g of 25% EtOH (in EtOAc) followed by the addition of EtOAc (0.25 mL) resulting in a cloudiness that remained. The cloudy hot solution was allowed to cool to room temperature during which time crystallization occurred. After crystallization was complete (2 h), the crystalline solid was filtered, washed with anhydrous EtOAc (~40 mL), and vacuum dried to give 424 mg of the dihydrochloride salt of compound (I) as a free-flowing solid (tiny beads, 99.8% AUC) containing only 0.05 eq (0.45% w/w) of EtOH and 0.015 eq (0.26% w/w) of EtOAc. Slightly better recovery (460 mg from 586 mg) was attained using isopropanol/EtOAc but the level of solvent entrapment was higher [0.085 eq (1.0% w/w) of isopropanol and 0.023 eq (0.4% w/w) ofEtOAc].
Example 3: Large Scale Synthesis of Compound (I) di-HCl
[000252 ] Reagents and solvents were used as received from commercial suppliers. Progress of the reactions was monitored by HPLC, GC/MS, or 1H NMR. Thin-layer chromatography (TLC) was performed using Analtech silica gel plates and visualized by UV light (254 nm). High pressure liquid chromatography (HPLC) was performed on an Agilent 1100 Series instruments. Proton and carbon nuclear magnetic resonance spectra were obtained using a Bruker AV 300 at 300 MHz for proton and 75 MHz for carbon. The solvent peak was used as the reference peak for proton and carbon spectra. Preparation of 4-(2-(4-Bromophenoxy)ethyl)morpholine (2)
[000253 ] A 50 L jacketed reactor equipped with a reflux condenser and temperature probe was charged with 4-(3-chloropropyl)morpholine (2.44 kg, 0.54 mol), 4-bromophenol (2.27 kg, 0.54 mol, 1.0 equiv.), powdered potassium carbonate (6.331 kg, 1.88 mol, 3.50 equiv.), and DMF (12.2 L) and stirred. The reaction mixture was then heated to 60-65 0C and stirred overnight. After 17.5 h, the reaction mixture was cooled to 20-25 °C. The reaction mixture was charged to a different reactor equipped with bottom valve for the work-up. While maintaining a temperature between 20-30 0C, DI water (48.7 L) was charged to the reactor. The phases were separated. The aqueous layer was extracted with MTBE (3 x 24.4 L). To the combined organics, DI water (18.3 L) and then 6M sodium hydroxide (18.2 L) were added. The mixture was stirred for 2-5 minutes and the phases were separated. The organic phase was washed with water (24.4 L) and brine (24.4 L), dried over magnesium sulfate, filtered, and concentrated to give 337Og of a yellow oil (89% crude yield, 99.4% AUC by HPLC).
Preparation of 6-fluoropyridin-3-ylboronic acid (4)
[000254] A 72 L reactor equipped with reflux condenser, and temperature probe. To the reactor 5-bromo-2-fluoropyridine (1.17 L, 0.568 mol), toluene (18.2 L), and triisopropyl borate (3.13 L, 0.68 mol, 1.2 equiv.) were charged and stirred. Tetrahydrofuran (4.4 L) was added to the reactor and the reaction mixture was cooled to between —35 to -50 0C. While maintaining a temperature between -35 to —45 0C, n-butyl lithium (2.5 M solution of hexanes, 5.44 L, 0.68 mol, 1.2 equiv.) was cautiously added to the reactor. After 5 h, the reaction was deemed complete and the reaction mixture was warmed to between -15 to -20 0C. To the reaction was added 2M HCl (11.80L) to the reactor while maintaining a temperature between -15 0C and 0 0C. The reaction mixture was stirred at 18 to 23 0C for (16 h) and the phases were separated. The organics were then extracted with 6 M sodium hydroxide (6.0 L). The acidic anbasic aqueous phases were mixed in the reactor and 6 M HCl (2.5 L) was added until pH 7.5 was achieved. Sodium chloride (6.0 kg) was then added to the aqueous phase. The aqueous phase was then extracted with THF (3 * 20 L). The combined organics were dried with magnesium sulfate and concentrated to give 1300 g of a tan solid (81% crude yield).
Preparation of 4-(2-(4-(6-fluoropyridin-3-yl)phenoxy)ethyl)morpholine (5) [000255] A 72 L reactor equipped with reflux condenser, sparging tube, bubbler, and temperature probe was charged with 6-fluoropyridin-3-ylboric acid (2.84 kg, 1.24 equiv.), 4- (2-(4-bromophenoxy)ethyl)morpholine (4.27 kg, 1.0 equiv.), and DME (27 L). Agitation was started and sodium carbonate (4.74 kg, 3.0 equiv.) as a solution in DI water (17.1 L) was then charged to the reaction mixture. Argon was bubbled through the reaction mixture for 50 minutes. Under an argon atmosphere, tetrakis(triphenylphosphine)palladium (750 g, 0.04 equiv.) was added to the reaction mixture as a slurry in DME (1.0 L). The reaction mixture was heated to 75 – 85 0C and stirred overnight (17 h). The reaction mixture was cooled to between 18 – 22°C. DI water (26.681kg) and MTBE (26.681 L) were charged to the reactor and stirred for 5 minutes. The phases were separated and the aqueous phase was extracted with MTBE (2 x 26.7 L). The combined organics were extracted with 2M HCl (1 x 15.0 L, 3 x 21.8 L). The aqueous phase was then charged back to the reactor and ethyl acetate was added (26.7 L). The pH was adjusted to 6.2 using 6 M sodium hydroxide (26.7 L) while maintaining a temperature between 15 – 25 0C. The phases were separated and the aqueous phase was extracted with ethyl acetate (2 x 26.7 L). The combined organics were dried with magnesium sulfate and concentrated to give 4555 g of a residue (101% crude yield, 67.1% AUC by HPLC).
Purification of 4-(2-(4-(6-fluoropyridin-3-yl)phenoxy)ethyl)morpholine (5)
[000256] The crude product (575 g) was purified by silica gel chromatography by eluting with methanol/ethyl acetate/heptane (30% ethyl acetate/heptane, 50% ethyl acetate/heptane, 75% ethyl acetate/heptane, 100% ethyl acetate, and 5% methanol/ethyl acetate). Concentration of the pure fractions by TLC (10% methanol/dichloromethane, Rf = 0.3) provided 420 g of a light brown solid (73% recovery, >99.9% AUC by HPLC).
Preparation of 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2-yl)acetonitrile (6)
[ 000257] A 1 M solution of NaHMDS (2.0 L, 5.0 equiv.) in THF was charged to a 5-L flask and cooled to -20 to -15 0C. While maintaining a temperature below -10 0C, fluoride (119.7g, 1.0 equiv.) in THF (500 mL) was charged to the flask over 20 minutes. Acetonitrile (82.5 mL, 4.0 equiv.) in THF (170 mL) was added to the flask over 20 minutes, while maintaining a temperature below —100C. The reaction mixture was then stirred for 1 h. To the reaction was added brine (1.5 L, 12.6 vol.) at a rate as to maintain a temperature below 10 0C. The solution was then warmed to room temperature and the layers were allowed to separate. The mixture was filtered over Celite and washed with THF (I x 200 mL, 1 x 100 mL). The aqueous phase was extracted with toluene (750 mL). The combined organics were dried with magnesium sulfate, filtered, washed with toluene (2 * 25OmL), and concentrated to dryness. Toluene (IL) was added and the solution was concentrated to dryness again to give 169.8 g of an oil. MTBE (1190 mL, 7 vol.) was added to the oil at 50 0C and stirred for 15 minutes. Heptane (850 mL, 5vol.) was added over ten minutes at 50 0C. The mixture was then cooled to room temperature over 1.5 h and stirred for 2 h. The slurry was filtered, washed with 1 :4 MBTE/heptane (2 x 100 mL), and dried in an oven overnight at 45 0C to give 102.3 g of an off-white solid (80% yield, 98.8% AUC by HPLC).
Preparation of methyl 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2-yl)acetate (7)
[000258] Nitrile 6 (101 g) and methanol (1.01 L, 10 vol.) were charged to a 3-L flask equipped with stir bar and thermocouple. Concentrated H2SO4 (175 mL, 10.0 equiv.) was added drop wise to the solution over 15 minutes while maintaining a temperature below 60 0C. Followed by 30% fuming sulfuric acid (124 mL) was added drop wise to the solution while maintaining a temperature below 60 0C. The solution was then heated to reflux with a heating mantle and stirred overnight. When the reaction was deemed complete, it was cooled to 20 0C. In a second flask (22 L), saturated sodium bicarbonate (10.7 L) and dichloromethane (1.1 L) were charged and cooled to 15 0C. While maintaining a temperature below 20 0C, the reaction mixture was added to the sodium bicarbonate/dichloromethane mixture. The quench was stirred for 15 minutes and the phases were separated. The aqueous phase was extracted with dichloromethane (I x 55OmL, 1 x 30OmL). The combined organics were dried with magnesium sulfate and concentrated to dryness to give 105 g of an orange solid (94% crude yield, 97.7% AUC by HPLC).
Preparation of 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2-yl)-N-benzylacetamide (Compound (I))
[ 000259] Ester 7 (103 g), anisole (513 mL, 5 vol.), and benzylamine (94 mL, 3.0 equiv.) were charged to a 3 L flask equipped with thermocouple and overhead stirrer. The reaction mixture was then heated to 142 0C and stirred for two days. The reaction mixture was cooled to 45-50 0C and stirred for 2 hours. To the mixture was added n-heptane (1.5 L) dropwise over an hour. The solution was cooled to room temperature over three hours and then stirred overnight. The resulting slurry was filtered, washed with 4: 1 Anisole/n-heptane (200 mL) and n-heptane (3 x 100 mL). Drying in the oven overnight, the resulting product was 112. Ig of a tan solid (90% yield, 99.6% AUC by HPLC). The use of a single isomer of heptane was essential to adequately quantitate the residual solvent.
Preparation of 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2-yl)-N-benzylacetamide dihydrochloride salt (Compound (I) 2HC1)
[000260 ] EtOH (1.0 L) was charged to a 2-L flask and acetyl chloride (62.5 raL, 3.0 equiv.) was added slowly to the flask and stirred for 40 minutes. The resulting solution was added to compound (I) (100 g) over 30 minutes while maintaining a temperature of 30 0C. The solution was concentrated to a mass of 270 g. The concentrated solution was added to ethyl acetate (2 L) over 20 minutes with rapid stirring. The mixture was stirred overnight and then filtered under nitrogen to give two distinct solid products, tan solids (73.5 g) and darker solids (42.2 g). The solids were dry blended to give a combined yield of 99%. The HPLC analysis indicated 99.0% purity (AUC).
Analysis indicated that ethanol was present at 2530 ppm, ethyl acetate at 48,110 ppm, ethyl chloride at 170 ppm, and no heptane and anisole were detected. Palladium content was assayed three times and measured to be 29 ppm, 2 ppm, and less than 1 ppm.
Crystallization Study of Compound (I) 2HCl
Preparation of 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2-yl)-N-benzylacetamide (Compound (I))
[000268] To a 22-L reactor was charged compound 7 (650 g, 1.82 mol), anisole (3.25
L, 5 vol, anhydrous) and benzylamine (600 mL, 0.92 vol, 3 equiv). The batch (approximately 18 °C) was heated to 142 ± 5 °C over 1 hour 44 minutes, with dissolution occurring at 30 0C. The batch was maintained at 142 ± 5 0C for 69 hours 30 minutes at which point HPLC analysis indicated that compound 7 was 0.9% by conversion (specification <1.7% by conversion). The batch was cooled to 45-50 0C over 5 hours 12 minutes (to aid cooling the nitrogen flow was increased once the batch was approximately 72 0C). At that temperature range, the batch was poorly stirring and on mixing, the batch temperature increased to 52 0C. It was >50 °C for <15 minutes. The batch was aged for 2 hours 2 minutes once initially <50 0C, then n-heptane (9.75 L, 15 vol, 99%) was added to the batch over 1 hour 56 minutes, maintaining the batch temperature at 45-50 °C. The heating was then discontinued and the batch cooled to 25 0C over 10 hours 32 minutes and then to approximately 20 °C over 20 minutes. The total time the batch was maintained <25 0C was 4 hours 50 minutes (2 hours 47 minutes at approximately 20 0C). The batch was filtered under suction via a 24-inch polypropylene filter funnel (fitted with a PTFE cloth) and the reactor rinsed with anisole/n- heptane (1.3 L, 4: 1) and the rinse transferred to the cake. The cake was then washed successively with two portions of /i-heptane (1.3 L, 0.65 L). The total filtration time was 39 minutes. The batch (net wet weight 1004 g of KX2391) was transferred to three glass trays and placed into a vacuum oven set at 50 0C and dried to constant weight over 96 hours 26 minutes.
Preparation of 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2-yl)-N-benzylacetamide mesylate (Compound (I)-MSA)
[000269] Compound (I) (520 g, 1.21 mol) was transferred to reactor 1 using acetone (41.6 vol, 80 vol, ACS) to facilitate the transfer. The batch was heated to 50 ± 5 0C over 33 minutes with dissolution occurring at 30 0C . The batch was clarified into a second reactor via a transfer pump fitted with an inline filter (Pall P/N 12077, 10 micron) and reheated from 46 0C to 50 ± 5 0C. Methanesulfonic acid (121.4 g, 1.05 equiv, 99% extra pure) was added to the pale yellow batch over 12 minutes and the heating then discontinued. After fourteen minutes, white solids were observed, which increased in number to give after 59 minutes a white suspension. The batch was in the range of 25 ± 5 0C after 7 hours 51 minutes and aged for a further 19 hours 21 minutes (10 hours 30 minutes at <27 0C). The batch was filtered under suction via a 24-inch polypropylene filter (PTFE cloth) and the reactor rinsed with acetone (2.0 L, clarified, ACS) and the rinse transferred to the cake. The cake was covered with a stainless steel cover and sucked dry under a flow of nitrogen. The total filtration time was 21 minutes. The batch (net wet weight 764 g) was transferred to three glass drying trays and dried in a vacuum oven to constant weight at 25 ± 5 °C over 21 hours 54 minutes (565 g, 89% of theory). A sample was removed for analysis and the batch maintained in vacuo at 25 ± 5 °C. The batch was then transferred to two 80-oz amber glass bottles (Teflon lined polypropylene closure), blanketed with argon and stored at -10 to -20 °C.
PATENT
WO 2010135429
https://patents.google.com/patent/WO2010135429A2/en
Preparation of KX2-391 and its salts
[00045] The synthesis of 4-(2-(4-(6-fluoropyridin-3-yl)phenoxy)ethyl)morpholine is shown in the scheme below:

[00046] 4-(2-(4-(6-fluoropyridin-3-yl)phenoxy)ethyl)morpholine (5) was synthesized in 3 steps. Intermediate 2 was synthesized using an ether coupling reaction e.g., using Williamson ether synthesis. Ether formation between 4-(2-chloroethyl)morpholine (1) and A- bromophenol was carried out in the presence of potassium carbonate and DMF to afford 4-(2- (4-bromophenoxy)ethyl)morpholine (2). Rigorously dry conditions were not essential for this reaction and a basic wash with sodium hydroxide was used to remove any remaining A- bromophenol. In another aspect of the invention, intermediate 2 is synthesized using any ether formation reaction. Intermediate 2 is synthesized starting from compound 1 containing any leaving group. For example, the skilled chemist would start with compounds of the
general formula

wherein the leaving group “LG” includes but is not limited to halogen, tosylate, mesylate, trifluate, etc.
[00047] Compound 5 was formed using a Suzuki reaction. Formation of the aryl borate, 6-fluoropyridin-3-yl-3-boronic acid (4), was carried out by forming the aryl anion using n-BuLi followed by in situ quenching with triisopropylborate (Li, et ah, J. Org. Chem. 2002, 67, 5394-5397). The resulting 6-fluoropyridin-3-yl-3-boronic acid (4) was coupled to 4-(2-(4-bromophenoxy)ethyl)morpholine (2) in a solution of DME and aqueous sodium carbonate using tetrakis(triphenylphosphine)palladium to afford 4-(2-(4-(6-fluoropyridin-3- yl)phenoxy)ethyl)morpholine (5), which was purified using silica gel chromatography. The skilled chemist would know that other transition metal coupling reaction are used to prepare compound 5.
[00048] The synthesis of 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2-yl)-JV- benzylacetamide dihydro chloride is shown below:

[00049] 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2-yl)-N-benzylacetamide dihydrochloride (KX2-391 HCl) was synthesized in four linear steps. The fluoride of 4-(2-(4- (6-fluoropyridin-3-yl)phenoxy)ethyl)morpholine (5) was displaced by the anion of acetonitrile formed using commercially available NaHMDS. Acetonitrile was added slowly to a cooled mixture of compound 5 and base to form 2-(5-(4-(2- morpholinoethoxy)phenyl)pyridin-2-yl)acetonitrile (6). In another aspect of the invention, intermediate 5 may have a leaving group other than fluorine. Thus, compounds of the general formula:

would be pursued where LG includes other leaving groups known to the skilled chemist.
[00050] Acid catalyzed methanolysis of 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-
2-yl)acetonitrile (6) was carried out using a mixture of concentrated sulfuric and fuming sulfuric acid. The use of fuming sulfuric acid removed residual water from the reaction mixture and reduced the amount of carboxylic acid by-product formed. The reaction mixture was quenched by adding the reaction mixture to a solution of saturated sodium bicarbonate and dichloromethane while maintaining the temperature below 20 ºC. Any carboxylic acid contaminant was readily removed with aqueous work-up. In another aspect of the invention, other acid catalyzed conditions are used by the skilled artisan for alcoho lysis of the nitrile of compound 6 to produce compound 7.
[00051] The resulting methyl 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2- yl)acetate (7) and benzyl amine were coupled in anisole at high temperature to afford 2-(5-(4- (2-morpholinoethoxy)phenyl)pyridin-2-yl)-N-benzylacetamide (KX2-391). An HCl solution formed by adding acetyl chloride to absolute ethanol was added to KX2-391 to form the bis- HCl salt, 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2-yl)-N-benzylacetamide dihydrochloride, (KX2-di-HCl).
[00052] The synthesis of the mesylate salt of KX2-391 (KX2-391 -MSA) is depicted in the scheme below:

[00053] 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2-yl)-N-benzylacetamide mesylate (KX2-391 MSA) was synthesized in four linear steps starting from compound 5.
The first 3 steps were carried out similar to the procedure discussed above for KX2-391 2HCl to afford methyl 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2-yl)acetate (KX2-391). KX2-
391 was converted to the methanesulfonate salt by treatment with methanesulfonic acid
(MSA) in acetone at 50 ºC to afford 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2-yl)-JV- benzylacetamide mesylate (KX2-391 MSA).
EXAMPLES Example 1: Small Scale Synthesis of KX2-391

[000343] The preliminary synthesis described below was illustrated in
US20060160800A1. This procedure is useful for small scale reactions, for example, reactions that produce up to 50 g of product.
[000344] For the following synthesis, unless otherwise noted, reagents and solvents were used as received from commercial suppliers. Proton and carbon nuclear magnetic resonance spectra were obtained on a Bruker AC 300 or a Bruker AV 300 spectrometer at 300 MHz for proton and 75 MHz for carbon. Spectra are given in ppm (δ) and coupling constants, J, are reported in Hertz. Tetramethylsilane was used as an internal standard for proton spectra and the solvent peak was used as the reference peak for carbon spectra. Mass spectra and LC-MS mass data were obtained on a Perkin Elmer Sciex 100 atmospheric pressure ionization (APCI) mass spectrometer. LC-MS analyses were obtained using a Luna C8(2) Column (100 x 4.6 mm, Phenomenex) with UV detection at 254 nm using a standard solvent gradient program (Method B). Thin-layer chromatography (TLC) was performed using Analtech silica gel plates and visualized by ultraviolet (UV) light, iodine, or 20 wt % phosphomolybdic acid in ethanol. HPLC analyses were obtained using a Prevail Cl 8 column (53 x 7 mm, Alltech) with UV detection at 254 nm using a standard solvent gradient program (Method A or B). Method A:
A = Water with 0.1 v/v Trifluoroacetic Acid
B = Acetonitrile with 0.1 v/v Trifluoroacetic Acid

Method B:
A = Water with 0.02 v/v Trifluoroacetic Acid
B = Acetonitrile with 0.02 v/v Trifluoroacetic Acid

Synthesis of Η-benzyl-2- (5-bromopyridin-2-yl)acetamide :

[000345] A flask was charged with 5-(5-bromopyridin-2(lH)-ylidene)-2,2-dimethyl- l,3-dioxane-4,6-dione (1.039 g, 3.46 mmol), benzylamine (0.50 mL, 4.58 mmol), and toluene (20 mL). The reaction was brought to reflux under nitrogen for 18 hours, then cooled and placed in a freezer until cold. The product was collected by filtration and washed with hexanes to yield a mass of bright white crystals (1.018 g, 96%).
Synthesis of 4- (2- (4- (4, 4, 5, 5-tetramethylfl, 3, 2] dioxaborolan-2-yl)- phenoxy) ethyl)morpholine :

[000346] To a stirring solution of 4-(4,4,5,5-tetramethyl[l,3,2]dioxaborolan-2-yl)- phenol (2.55 g, 11.58 mmol), 2-morpholin-4-ylethanol (1.60 mL, 1.73 g, 13.2 mmol) and triphenyl phosphine (3.64 g, 13.9 mmol) in methylene chloride (60 mL) at 0 ºC was added dropwise DIAD (2.82 g, 13.9 mmol). The reaction was allowed to warm to room temperature and stir overnight. After 18 hours, additional portions of triphenyl phosphine (1.51 g, 5.8 mmol), 2-morpholin-4-ylethanol (0.70 mL, 5.8 mmol), and DIAD (1.17 g, 5.8 mmol) were added. After stirring an additional 2 hours at room temperature the reaction was concentrated and the residue purified by flash chromatography (5% to 25% EtOAc in CHCI3) to provide the product as a white solid (2.855 g, 74%).
Synthesis of 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2-yl)-N-benzylacetamide KX2-391

[000347] A lO rnL reaction tube with a septum closure and stir bar was charged with N- benzyl-2-(5-bromopyridin-2-yl)acetamide (123 mg, 0.403 mmol), 4-(2-(4-(4,4,5,5- tetramethyl[l,3,2]dioxaborolan-2-yl)-phenoxy)ethyl)morpholine (171 mg, 0.513 mmol), and FibreCat 1007 (30 mg, 0.015 mmol). Ethanol (3 mL) was added, followed by aqueous potassium carbonate solution (0.60 mL, 1.0 M, 0.60 mmol). The tube was sealed and heated under microwave conditions at 150 ºC for 10 minutes. The reaction was cooled and concentrated to remove the majority of the ethanol, and then taken up in 10 mL of ethyl acetate and washed successively with water and saturated sodium chloride solution. The organic layer was dried with MgSO4, filtered and concentrated to a white solid. This white solid was triturated with ethyl ether to give KX2-391 as a white solid (137 mg, 79%): mp 135-137 ºC; 1H NMR (300 MHz,CDCl3) δ 8.70 (d, IH, J=2.0 Hz), 7.81 (dd, IH, J=2.4 Hz, J=8.0Hz), 7.65 (br s, IH), 7.49 (d, 2H, J=8.8 Hz), 7.37-7.20 (m, 6H), 7.01 (d, 2H, J=8.8 Hz), 4.49 (d, 2H, J=5.8 Hz), 4.16 (t, 2H, J=5.7 Hz, 3.82 (s, 2H), 3.78-3.72 (m, 4H), 2.84 (t, 2H, J=5.7 Hz), 2.62-2.58 (m, 4H); HPLC (Method B) 98.0% (AUC), tR = 1.834 min.; APCI MS m/z 432 [M+H]+.
Example 2: Intermediate Scale Synthesis of KX2-391 di-hydrochloride
[000348] The synthesis outlined in this example can be used on intermediate-scale reactions. The preparation of batches of at least 50 g of the dihydrochloride salt of KX2-391 is shown in Scheme 1. The linear synthesis consisted of 6 steps, a seventh step being the preparation of one of the reagents, 6-fluoropyridin-3-ylboronic acid (which is also available commercially). The overall yield of the sequence was 35% with an average yield of 83%, with the lowest yielding step being that of 68%. Of the seven steps only one required chromatography. The procedure listed below was performed on a 70 g scale.
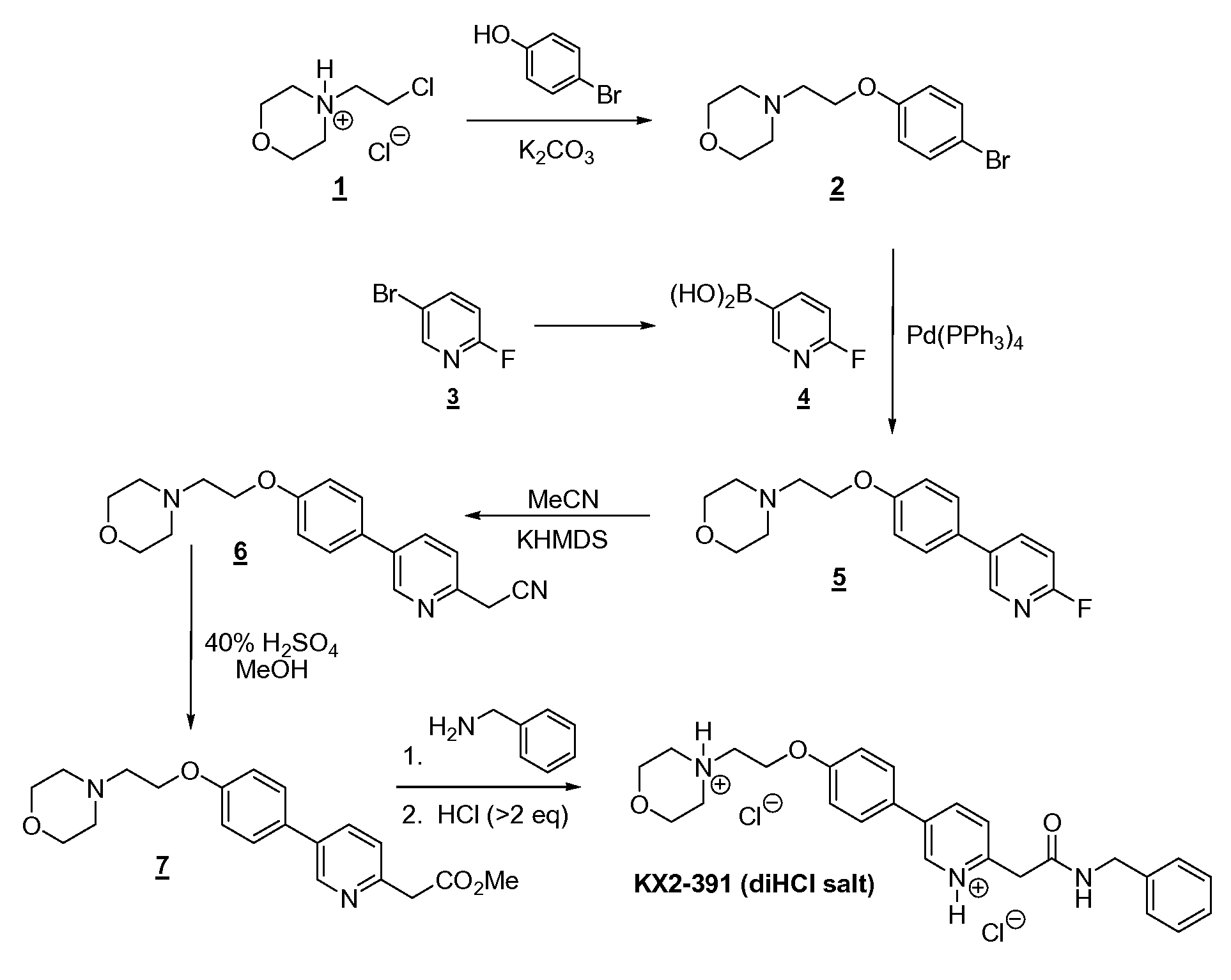
[000349] The first step is a Williamson ether synthesis between 4-bromophenol (131 g) and N-chloroethylmorpholine (1 as the HCl salt; 141 g) using K2CO3 powder (3 to 3.5 equivalents) as the base and having acetonitrile as the solvent. The ingredients were mixed and stirred at reflux overnight with high conversion (96.3-99.1%). After dilution with dichloromethane and heptane, the reaction mixture was filtered and evaporated to give the desired product 2 in essentially a quantitative yield (216 g). Note that with similar substrates (e.g., 4-bromo-3-fluorophenol), conversions (even with extensive heating) were not always so high (e.g., 59.9-98.3%). Both the alkyl chloride and the K2CO3 are preferably purchased from Aldrich. If continued heating does not drive reaction to completion, unreacted bromophenol can readily be removed by dissolving the crude reaction mixture in 4 parts toluene and washing out the phenol with 4 parts 15% aqueous NaOH. [000350] One of the reagents required for the second step (Suzuki coupling) was 6- fluoropyridin-3-ylboronic acid (4). Although available commercially, this reagent was readily prepared by lithium-bromide exchange of 5-bromo-2-fluoropyridine (3, 102 g) with n- butyllithium (1.2 eq) at low temperatures (<-60 ºC) in TBME followed by the addition of triisopropylborate (1.65 eq). Both stages of the reaction are brief, with an overall reaction time (including addition times) of ~3 h. Quenching is achieved with aqueous 24% NaOH, which also extracts the product leaving impurities in the organic layer. Once the aqueous layer is removed, it is then neutralized with HCl and extracted with EtOAc. After drying the organics and diluting with some heptane, concentration leads to precipitation/ crystallization of the product. Filtration gave the boronic acid 4 in relatively high purity (96.4% AUC) and good yield (69 g, 79-90%; see note on estimation of yield in the experimental section), which can be used without further purification.
[000351] The second reaction step in the linear sequence (a Suzuki coupling) is a simple reaction to set up; all the reagents [2 (111 g), aqueous Na2CO3, DME, and Pd(PPh3)4 (0.04 eq)] were charged to the reaction flask and the mixture heated at reflux; note that the reaction mixture was degassed to remove oxygen. Once the reaction is complete (within 7 h), the work-up involved decanting (or siphoning off) of reaction solution from the organic salts on the side of the flask (there was no visible aqueous layer), the flask was rinsed, and dried, and the solvent was removed from the combined organics. Crystallization of crude 5 from isopropanol/heptane provided material of improved purity compared to the crude, but still required chromatography (ratio of silica gel to crude was -8.5:1) to obtain material of adequate purity (>98%); the yield was 68% (79.5 g). Use of clean 5 prevented the need for chromatography in the next step, acetonitrile displacement of the fluorine atom. [000352] The replacement of fluoride with acetonitrile was also a simple reaction, and a simple room temperature crystallization of the crude product provided clean 6 in high yield and purity. The reaction involved initial formation of the “enolate” from acetonitrile (6.5 eq) using potassium hexamethyldisilane KHMDS (8 eq)/THF at -10 ºC followed immediately by the addition of fluoride 5 (79 g). The reaction was quick and after one hour quenching was achieved with saturated brine. After drying and evaporation of solvent of the organics, the resulting crude mixture consisted of only two components, the desired product and a much less polar product from apparent self-condensation of acetonitrile. The crude mixture was swirled in isopropanol/heptane and allowed to sit overnight, which resulted in complete crystallization of the product, which was filtered off and washed to provide high purity 6 (99.3% AUC) in good yield (64 g, 76%).
[000353] Methanolysis of 6 (64 g) was accomplished by heating in 40% H2SO4 (in
MeOH) until the reaction was complete (25 h). The reaction was then cooled, stirred with MgSO4 to convert traces of hydro lyzed product (ArCH2-CO2Me) back to product, and then added to cooled, aqueous K2CO3, with simultaneous extraction into dichloromethane. Drying and evaporation of most of the DCM followed by addition of 5% EtOAc (in heptane) and further concentration resulted in the crystallization of the product. Filtration of the solid and washing gave high purity (98.9% AUC) 7 in good yield (82%), additional high purity product (4 g) being obtained from the mother liquors for a total yield of 61.7 g (87%). [000354] The amidation step also involved charging of the reaction vessel with the ingredients (7 (61 g), benzyl amine (3 eq), and high boiling anisole) and then heating at reflux until the reaction was complete. Cooling of the reaction mixture resulted in complete crystallization of the target compound with high purity (98.9%) and good yield (81%). [000355] The final step was the formation of the dihydro chloric salt of the target compound. In order to ensure complete protonation at both basic sites, the reaction was conducted in absolute ethanol, which freely dissolved the dihydrochloride salt. After evaporation to near dryness, the reaction mixture was “chased” with ethanol twice to remove excess hydrogen chloride. The resulting viscous oil was dissolved in ethanol (2 parts) and then added, with rapid stirring, to a large volume (20 parts) EtOAc (ethyl acetate). Filtration, washing with ethyl acetate (no heptane) and vacuum drying provided the dihydrochloride salt of KX2-391 as a creamy-white powder. A total of 68 g (yield of 97%) was obtained of the final salt in high purity (99.6% AUC), which contained traces of EtOAc (4.8% w/w), EtOH (0.3% w/w), and heptane (0.6% w/w; from a final wash with heptane prior to vacuum drying). This salt was also crystallized (instead of the precipitation method described above) from hot EtOH/EtOAc to afford crystalline beads that had much lower entrapped solvent levels (only 0.26% w/w of EtOAc and 0.45% w/w of EtOH) and was free-flowing.

Preparation of 4-(2-(4-bromophenoxy)ethyl)morpholine (2):
[000356] A 5 L three-necked round-bottomed flask, equipped with mechanical stirrer, thermometer with adapter, condenser, and nitrogen inlet (on top of condenser), was charged with 1 (140.7 g, 0.756 mol), 4-bromophenol (130.6 g, 0.755 mol), anhydrous K2CO3 powder (367.6 g, 2.66 mol, 3.5 eq), and acetonitrile (1.3 L). The mixture was vigorously stirred (blade touching bottom of flask) at 80 ºC (overnight), followed by dilution with DCM (500 mL) and heptane (200 mL) and filtration through Celite. Evaporation to dryness (rotovap, then high vac) gave 2 as a light yellow oil (216.00 g, yield of 100%, 96.3% AUC, contains 3.7% unreacted bromophenol). This material was used successfully without further purification.
[000357] 1H NMR (CDCl3) δ 2.57 (t, 4 H), 2.79 (t, 2 H), 3.73 (t, 4 H), 4.08 (t, 2 H), 6.78
(d, 2 H), 7.37 (d, 2 H). MS (from LC/MS): m/z 287.1 [M + I].
[000358] That the bromophenol can be readily removed was demonstrated on a 2 g sample by first dissolving the sample in toluene (8 g) and washing with 8 g of 15% aqueous NaOH; liquid chromatography showed no trace of unreacted bromophenol in the recovered product (1.97 g; 98.5% recovery).

Preparation of 6-fluoropyridin-3-ylboronic acid (4):
[000359] To stirred and cooled (dry ice-acetone bath) anhydrous [TBME] (620 mL; in a
3 L three-necked round-bottomed flask equipped with mechanical stirrer, temperature probe with adapter, and nitrogen inlet) was added (via syringe) 2 M BuLi (352 mL, 0.704 mol, 1.2 eq). To this rapidly stirred and cooled (< -75 ºC) mixture was added a solution of 3 (102.2 g, 0.581 mol) in anhydrous TBME (100 mL) over a period of 13 min during which time the internal temperature rose to -62 ºC. The reaction was stirred for another 45 min (the temperature was maintained between -62 ºC and -80 ºC), followed by the rapid and sequential addition of four portions of triisopropylborate (total of 180 g, 0.957 mol, 1.65 eq). At the end of the addition the internal temperature had risen to -33 ºC. After stirring an additional 45 min over the cold bath (internal temperature lowered from -33 ºC to -65 ºC), the cold bath was removed and the stirred mixture on its own rose to -22 ºC over a period of 50 min. After warming (via water bath) to 6 ºC over a period of 15 min, the stirred reaction mixture was placed in an ice-water bath and then quenched under nitrogen with a cooled solution of NaOH (160 g) in water (500 mL). Once the addition was complete, the internal temperature was 20 ºC. This mixture was stirred at room temperature for 1.5 h. The aqueous layer was removed, neutralized to pH 7 with -350 mL concentrated HCl, and then extracted with EtOAc (3 x 1 L). Because the pH was now 8-9, the aqueous layer was adjusted to pH 7 using ~15 mL concentrated HCl and extracted further (2 x 1 L) with ethyl acetate. The combined EtOAc extracts were dried (Na2SO4), filtered, and concentrated to a volume of -150 mL. With swirling of the concentrate, heptane was added in portions (total volume of 300 mL) resulting in the precipitation/crystallization of the product. Filtration, washing of the solid with heptane (100 mL, 300 mL, then another 300 mL), and air drying gave the title product as an off-white solid (68.6 g, yield of 79-90%*; LC purity of 96.4%, NMR showed an estimated 5.5% w/w of heptane), which was used successfully without further purification. LC/MS showed it to be a mixture of the two following entities, the intensity of the higher molecular weight entity being major (*Note: yield of reaction is 79% if the boronic acid is assumed to be the only constituent and is 90% if it is assumed that the cyclic borate is the only constituent):

1H NMR (CDCl3) δ 7.14 (dd, 1 H), 8.27 (ddd, 1 H), 8.39 (br s, 2 H, 2 OH), 8.54 (fine d, 1 H). MS (from LC/MS): m/z 143.0 [M + 1; for boronic acid] and 370.0 [M + 1; for cyclic borate above].

[000360] A 2 L three-necked round-bottomed flask equipped with mechanical stirrer, thermometer and adapter, condenser, and nitrogen inlet (at top of condenser) was charged with 2 (110.7 g, 0.387 mol), 4 (71.05 g, 0.477 mol, 1.23 eq) and DME (700 mL). The resulting stirred solution was degassed by passing a rapid stream of nitrogen through the stirred solution over a period of 5 min followed by the addition of a degassed solution of Na2CO3 (121.06 g, 1.142 mol, 3 eq) in H2O (250 mL) and also solid Pd(PPh3)4 (19.8 g, 0.044 eq). Immediately after the last addition, the head space above the reaction mixture was purged with nitrogen and the mixture then stirred at 80-85 ºC (internal temperature) for 7 h, followed by cooling to room temperature. Because of the lack of an aqueous layer, the supernatant was decanted, leaving behind the inorganic salts (with adsorbed water). The reaction flask with the inorganic salts was washed with 50% dichloromethane/ethyl acetate (2 x 250 mL), the washes being added to the decanted supernatant. These combined organics were dried (Na2SO4), filtered, and evaporated to dryness to a dark brown oil (148 g). To this oil was added 15O g of 50% heptane/isopropyl alcohol (IPA) and after swirling and cooling (via ice water bath), crystallization began. Additional heptane (50 g) was added and the resulting solid was filtered, washed, and air dried to give 48 g of a light brown solid. After evaporating the filtrate to dryness, the resulting mixture was swirled in 100 mL of 50% heptane/IPA followed by the addition of more heptane (-100 mL), stoppering and placing in the freezer for crystallization. The resulting solid was filtered, washed with heptane, and air dried to give 61 g of a gummy solid. Evaporation of the resulting filtrate gave an oil (34 g) which contained significant less polar impurities including Ph3P=O and so it was partitioned between 2 N HCl (240 mL) and EtOAc (220 mL). The bottom aqueous layer was removed and then stirred with EtOAc while neutralizing with K2CO3 to a pH of 7-8. The EtOAc layer was dried, filtered, and evaporated to dryness (22 g). The 48 g, 61 g, and 22 g portions were chromato graphed over silica gel (1.1 Kg) packed in DCM. Elution with DCM (400 mL), 50% DCM/EtOAc (5 L), and then 50% DCM/EtOAc (8 L) containing increasing amounts of MeOH/Et3N (beginning with 1.5% MeOH/1% Et3N and ending with 5% MeOH/3% Et3N) gave 77.68 g of a viscous oil (purity 98.0%) which immediately crystallized upon swirling in heptane (300 mL). Filtration, washing with heptane and air drying gave 75.55 g (98.7% AUC) of solid 5. Additional pure 5 (total of 3.9 g, 98.6-99.3% AUC) was obtained from earlier chromatographic fractions containing Ph3P=O by cleaning them up as done for the above 34 g sample, followed by evaporative crystallization. The total yield of 5 was 79.5 g (68%). 1H NMR (CDCl3) δ 2.59 (t, 4 H), 2.84 (t, 2 H), 3.75 (t, 4 H), 4.16 (t, 2 H), 6.97 (dd, 1 H), 7.01 (d, 2 H), 7.46 (d, 2 H), 7.92 (ddd, 1 H), 8.37 (fine d, 1 H). MS (from LC/MS): m/z 303.2 [M + I].

Preparation of 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2-yl)acetonitrile (6):
[000361] A 3 L three-necked round-bottomed flask was equipped with mechanical stirrer, thermometer and adapter, additional funnel, and nitrogen inlet (on top of addition funnel, positive pressure through a bubbler). With a rapid stream of nitrogen going through the bubbler, the stopper was removed and the flask was charged with KHMDS (415.8 g, 2.08 mol) and then anhydrous THF (1 L). To the stirred and cooled (ice/methanol bath, internal temperature of solution was -8 ºC) KHMDS/THF solution was added dropwise a solution of MeCN (70 g) in THF (110 mL) over a period of 22 min followed immediately by the relatively rapid (4 min) addition of a solution of 5 (79.06 g, 0.262 mol) in THF (400 mL), after which time the internal temperature of the reaction mixture had reached 10 ºC. With continued cooling (1 h) the internal temperature was -6 ºC and by TLC the reaction appeared complete. After an additional 30 min (internal temperature of -3 ºC), the reaction mixture was quenched with saturated brine (1 L) and diluted with EtOAc (500 mL). After removing the aqueous layer, the organic solution was dried (Na2SO4), filtered, and evaporated to dryness (to an oil) followed by completely dissolving in IPA (150 mL), diluting with heptane (300 mL), adding seed crystals (prepared by dissolving -100 mg of crude oil in IPA (-150 mg) and diluting with heptane (-2.5 mL)), and allowing to stand overnight. After stirring to break up the crystalline solid, the solid was filtered, washed with 250 mL 2:1 heptane/IP A and then multiple washes with heptane and air dried to give 64.38 g (yield of 76%) of title product 6 as a crystalline tan solid (LC purity of 99.3%). Another 5.88 g of less pure material was obtained from the filtrate.
[000362] 1H NMR (CDCl3) δ 2.59 (t, 4 H), 2.84 (t, 2 H), 3.74 (t, 4 H), 3.97 (s, 2 H),
4.17 (t, 2 H), 7.02 (d, 2 H), 7.46 (d, 1 H), 7.51 (d, 2 H), 7.87 (dd, 1 H), 8.77 (fine d, 1 H). MS (from LC/MS): m/z 324 A [M + I].

Preparation of methyl 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2-yl)acetate (7): [000363] A 2 L single-necked round-bottomed flask was charged with 6 (64.00 g, 0.198 mol) and MeOH (360 g) followed by the slow, careful, and dropwise addition OfH2SO4 (240 g) and the resulting homogeneous solution stirred at reflux (115 ºC oil bath) until the reaction was complete (25 h with 0.8% unreacted starting material) with 3.5% ArCH2CO2H. After brief cooling, MgSO4 (75 g) was added and the mixture swirled and allowed to stand an additional 45 min (composition now 96.3% product, 0.8% unreacted starting material, and 2.5% ArCH2CO2H). The reaction mixture was then added slowly to a rapidly stirred and cooled (ice-water bath) mixture of DCM (2 L) and a solution OfK2CO3 (450 g) in H2O (600 mL). The resulting emulsion was allowed to stand overnight. The clear portions of organic solution were siphoned off and the remainder portions were treated iteratively with water and DCM, the clear organics being combined with the original portion that was siphoned off. The combined organics were dried (Na2SO4), filtered, and concentrated to a volume of ~1.2 L followed by the addition of 300 mL of 5% EtOAc (in heptane) and then heptane (300 mL) and the mixture concentrated (rotovap with heat) again to remove the DCM. At this point 15 mL EtOAc was added and the hot mixture swirled until crystallization had begun, swirling continued until crystallization was near complete, and then allowed to stand and cool to room temperature for complete crystallization. The solid was then filtered, washed with 300 mL 5% EtOAc (in heptane) and heptane (100 mL) and then fully air dried to give 57.74 g (yield of 82%) of 7 as a light yellow solid (98.9% AUC). Another 3.94 g of clean product (97.9% AUC) was obtained from the filtrate (total yield of 87%).
[000364] 1H NMR (CDCl3) δ 2.60 (t, 4 H), 2.84 (t, 2 H), 3.74 (overlapping t and s, 6 H),
3.89 (s, 2 H), 4.17 (t, 2 H), 7.01 (d, 2 H), 7.34 (d, 1 H), 7.49 (d, 2 H), 7.80 (dd, 1 H), 8.74 (fine d, 1 H). MS (from LC/MS): m/z 357.4 [M + I].

Preparation of 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2-yl)-N-benzylacetamide (KX2-391 free base).
[000365] A l L single-necked round-bottomed flask was charged with 7 (61.4 g, 0.172 mol), benzyl amine (55.6 g, 0.519 mol, 3 eq), and anhydrous anisole (300 g) and then stirred at reflux until reaction was essentially complete (23 h, 165 ºC oil bath temperature; internal temperature was 147 ºC) and then allowed to cool to near room temperature. A portion (1 mL) of the reaction mixture was diluted with toluene (1 mL) resulting in the complete crystallization of that portion. This seed was then added to the reaction mixture and allowed to stand until the whole reaction mixture had crystallized to a single block. Toluene (150 mL) was added and the mixture swirled to break up the solid. Heptane/toluene (1 :1, 100 mL) was added and the solid mixture broken up further. Finally, heptane (50 mL, then 25 mL) was added and the mixture broken up even further, allowing to stand an additional 30 min before filtering the solid. Filtration of the solid, washing with 2:1 toluene/heptane (300 mL), 1 :2 toluene/heptane (300 mL), and then heptane (2 x 300 mL), and then drying (air, then high vac) gave 60.16 g (yield of 81%) of title product as a white solid (≥98.9% AUC). Another 2.5 g of less pure (97.4%) material was obtained from the mother liquors. 1H NMR (CDCl3) δ 2.60 (t, 4 H), 2.83 (t, 2 H), 3.74 (t, 4 H), 3.82 (s, 2 H), 4.18 (t, 2 H), 4.49 (d, 2 H), 7.01 (d, 2 H), 7.2-7.35 (m, 6 H), 7.49 (d, 2 H), 7.64 (br t, 1 H), 7.81 (dd, 1 H), 8.69 (fine d, 1 H). MS (from LC/MS): m/z 432.5 [M + I].
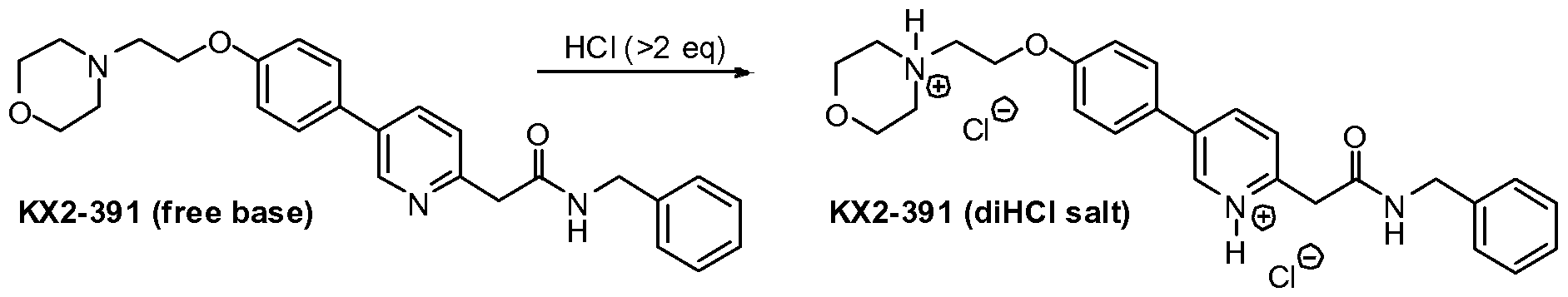
Preparation of 4-(2-(4-(6-(2-(benzylamino)-2-oxoethyl)pyridinium-3-yl)phenoxy)ethyl)- morpholin-4-ium chloride (KX2-391, diHCl salt).
[000366] To a stirred suspension of KX2-391 (free base, 60.00 g) in absolute EtOH (600 niL) was added 170 niL of 2.5 M HCl (in ethanol), 25 niL EtOH being added to wash down the sides of the flask. The resulting homogeneous solution was stirred at room temperature (20 min) and then evaporated to near dryness (to frothing). After chasing with EtOH (2 x 150 mL), the residue was taken up again in EtOH (150 mL) and then was followed by the slow addition of heptane until the mixture appeared saturated (33 mL required for cloudiness to remain). After sitting overnight, two layers had formed. After adding additional heptane (250 mL) crystallization still could not be induced and so the reaction mixture was concentrated to a volume of -200 mL at which time the mixture was homogeneous. This thick homogeneous solution was added dropwise to very rapidly stirred (mechanical) EtOAc (2 L). After the addition was complete, a 25 mL EtOH rinse of the original flask and addition funnel was added to the rapidly stirred mixture. The rapid stirring was continued for another ~1 h and then the mixture was filtered and the solid (partly gummy) was washed with EtOAc (300 mL) and then heptane. As soon as the heptane wash began, the solid got much gummier. The fritted Buchner funnel and its contents were covered (paper towel/rubber band) and immediately placed in the vacuum oven. After overnight vacuum at ~45 ºC, the vacuum was released under nitrogen, and the Buchner funnel containing the product (foamy solid) was immediately placed in a zip-lock back and then, under nitrogen (glove bag), transferred to a bottle and the foamy solid broken up (spatula) to a powder. A second night under high vacuum (-45 ºC) resulted in only 1.3 g of additional weight loss. Constant weight was essentially attained with the third night of high vacuum (~45 ºC) where only 0.2 g of weight was lost. The final weight of material was 68.05 g (yield of 97%), containing 0.29 eq (4.8% w/w) of EtOAc, 0.035 eq (0.3% w/w) EtOH, and 0.03 eq (0.6% w/w) heptane. The purity was 99.6%.
1H NMR (DMSO-d6) δ 3.1-3.3 (m, 2 H), 3.45-3.65 (m, 4 H), 3.8-4.0 (m, 4 H), 4.11 (s, 2 H), 4.32 (d, 2 H), 4.57 (t, 2 H), 7.19 (d, 2 H), 7.2-7.4 (m, 5 H), 7.88 (d, 2 H), 7.93 (d, 1 H), 8.68 (dd, 1 H), 8.99 (br t, 1 H), 9.10 (fine d, 1 H), 11.8 (br s, 1 H). MS (from LC/MS): m/z 432.5 [M + 1 of free base].
Elemental analysis (for C26H29N3O3 • 2 HCl • 0.035 EtOH • 0.29 EtOAc • 0.03 heptane • 0.8 H2O): Calculated (%): C, 60.03; H, 6.54; N, 7.65; Cl, 12.91 Observed (%):C, 59.85/59.97; H, 6.54/6.47; N, 7.67/7.67; Cl, 13.10/13.24 Calculated FW: 534.63 (does not take into account the 0.8 H2O which probably arose during handling of this very hygroscopic powder, since 1H NMR shows no evidence for H2O). [000367] The ethyl chloride level in this material was measured and found to be 98 ppm. The sample was also analyzed and found to contain 5,800 ppm of heptane. [000368] Analysis of another portion of this sample yielded the following results: 99.6% AUC, 1640 ppm ethanol, 41,480 ppm ethyl acetate, 5600 ppm heptane, no anisole detected, and 120 ppm ethyl chloride.
[000369] A procedure for recrystallizing the salt was also developed using the above dried salt. This procedure would work just was well on the highly pure crude salt (containing residual EtOH) obtained from concentrating the HCl salt-forming reaction mixture: [000370] The salt (575 mg) was dissolved in twice the mass of absolute EtOH (1.157 g) and then heated under nitrogen. To this hot solution (stirred) was added 1.6 g of 25% EtOH (in EtOAc) followed by the addition of EtOAc (0.25 mL) resulting in a cloudiness that remained. The cloudy hot solution was allowed to cool to room temperature during which time crystallization occurred. After crystallization was complete (2 h), the crystalline solid was filtered, washed with anhydrous EtOAc (~40 mL), and vacuum dried to give 424 mg of the dihydrochloride salt of KX2-391 as a free-flowing solid (tiny beads, 99.8% AUC) containing only 0.05 eq (0.45% w/w) of EtOH and 0.015 eq (0.26% w/w) of EtOAc. Slightly better recovery (460 mg from 586 mg) was attained using isopropanol/EtOAc but the level of solvent entrapment was higher [0.085 eq (1.0% w/w) of isopropanol and 0.023 eq (0.4% w/w) OfEtOAc].
Example 3: Large Scale Synthesis of KX2-391 di-HCl
[000371] Reagents and solvents were used as received from commercial suppliers.
Progress of the reactions was monitored by HPLC, GC/MS, or 1H NMR. Thin-layer chromatography (TLC) was performed using Analtech silica gel plates and visualized by UV light (254 nm). High pressure liquid chromatography (HPLC) was performed on an Agilent 1100 Series instruments. Proton and carbon nuclear magnetic resonance spectra were obtained using a Bruker AV 300 at 300 MHz for proton and 75 MHz for carbon. The solvent peak was used as the reference peak for proton and carbon spectra.
Preparation of 4-(2-(4-Bromophenoxy)ethyl)morpholine (2) [000372] A 50 L jacketed reactor equipped with a reflux condenser and temperature probe was charged with 4-(3-chloropropyl)morpholine (2.44 kg, 0.54 mol), 4-bromophenol (2.27 kg, 0.54 mol, 1.0 equiv.), powdered potassium carbonate (6.331 kg, 1.88 mol, 3.50 equiv.), and DMF (12.2 L) and stirred. The reaction mixture was then heated to 60-65 ºC and stirred overnight. After 17.5 h, the reaction mixture was cooled to 20-25 ºC. The reaction mixture was charged to a different reactor equipped with bottom valve for the work-up. While maintaining a temperature between 20-30 ºC, DI water (48.7 L) was charged to the reactor. The phases were separated. The aqueous layer was extracted with MTBE (3 x 24.4 L). To the combined organics, DI water (18.3 L) and then 6M sodium hydroxide (18.2 L) were added. The mixture was stirred for 2-5 minutes and the phases were separated. The organic phase was washed with water (24.4 L) and brine (24.4 L), dried over magnesium sulfate, filtered, and concentrated to give 337Og of a yellow oil (89% crude yield, 99.4% AUC by HPLC).
Preparation of 6-fluoropyridin-3-ylboronic acid (4)
[000373] A 72 L reactor equipped with reflux condenser, and temperature probe. To the reactor 5-bromo-2-fluoropyridine (1.17 L, 0.568 mol), toluene (18.2 L), and triisopropyl borate (3.13 L, 0.68 mol, 1.2 equiv.) were charged and stirred. Tetrahydrofuran (4.4 L) was added to the reactor and the reaction mixture was cooled to between -35 to -50 ºC. While maintaining a temperature between -35 to -45 ºC, n-butyl lithium (2.5 M solution of hexanes, 5.44 L, 0.68 mol, 1.2 equiv.) was cautiously added to the reactor. After 5 h, the reaction was deemed complete and the reaction mixture was warmed to between -15 to -20 ºC. To the reaction was added 2M HCl (11.80L) to the reactor while maintaining a temperature between -15 ºC and 0 ºC. The reaction mixture was stirred at 18 to 23 ºC for (16 h) and the phases were separated. The organics were then extracted with 6 M sodium hydroxide (6.0 L). The acidic anbasic aqueous phases were mixed in the reactor and 6 M HCl (2.5 L) was added until pH 7.5 was achieved. Sodium chloride (6.0 kg) was then added to the aqueous phase. The aqueous phase was then extracted with THF (3 x 20 L). The combined organics were dried with magnesium sulfate and concentrated to give 1300 g of a tan solid (81% crude yield).
Preparation of 4-(2-(4-(6-fluoropyridin-3-yl)phenoxy)ethyl)morpholine (5)
[000374] A 72 L reactor equipped with reflux condenser, sparging tube, bubbler, and temperature probe was charged with 6-fluoropyridin-3-ylboric acid (2.84 kg, 1.24 equiv.), A- (2-(4-bromophenoxy)ethyl)morpholine (4.27 kg, 1.0 equiv.), and DME (27 L). Agitation was started and sodium carbonate (4.74 kg, 3.0 equiv.) as a solution in DI water (17.1 L) was then charged to the reaction mixture. Argon was bubbled through the reaction mixture for 50 minutes. Under an argon atmosphere, tetrakis(triphenylphosphine)palladium (750 g, 0.04 equiv.) was added to the reaction mixture as a slurry in DME (1.0 L). The reaction mixture was heated to 75 – 85 ºC and stirred overnight (17 h). The reaction mixture was cooled to between 18 – 22ºC. DI water (26.681kg) and MTBE (26.681 L) were charged to the reactor and stirred for 5 minutes. The phases were separated and the aqueous phase was extracted with MTBE (2 x 26.7 L). The combined organics were extracted with 2M HCl (1 x 15.0 L, 3 x 21.8 L). The aqueous phase was then charged back to the reactor and ethyl acetate was added (26.7 L). The pH was adjusted to 6.2 using 6 M sodium hydroxide (26.7 L) while maintaining a temperature between 15 – 25 ºC. The phases were separated and the aqueous phase was extracted with ethyl acetate (2 x 26.7 L). The combined organics were dried with magnesium sulfate and concentrated to give 4555 g of a residue (101% crude yield, 67.1% AUC by HPLC).
Purification of 4-(2-(4-(6-fluoropyridin-3-yl)phenoxy)ethyl)morpholine (5)
[000375] The crude product (575 g) was purified by silica gel chromatography by eluting with methanol/ethyl acetate/heptane (30% ethyl acetate/heptane, 50% ethyl acetate/heptane, 75% ethyl acetate/heptane, 100% ethyl acetate, and 5% methanol/ethyl acetate). Concentration of the pure fractions by TLC (10% methanol/dichloromethane, Rf = 0.3) provided 420 g of a light brown solid (73% recovery, >99.9% AUC by HPLC).
Preparation of 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2-yl)acetonitrile (6)
[000376] A 1 M solution of NaHMDS (2.0 L, 5.0 equiv.) in THF was charged to a 5-L flask and cooled to -20 to -15 ºC. While maintaining a temperature below -10 ºC, fluoride (119.7g, 1.0 equiv.) in THF (500 mL) was charged to the flask over 20 minutes. Acetonitrile (82.5 mL, 4.0 equiv.) in THF (170 mL) was added to the flask over 20 minutes, while maintaining a temperature below -10 ºC. The reaction mixture was then stirred for 1 h. To the reaction was added brine (1.5 L, 12.6 vol.) at a rate as to maintain a temperature below 10 ºC. The solution was then warmed to room temperature and the layers were allowed to separate. The mixture was filtered over Celite and washed with THF (1 x 200 mL, 1 x 100 mL). The aqueous phase was extracted with toluene (750 mL). The combined organics were dried with magnesium sulfate, filtered, washed with toluene (2 x 25OmL), and concentrated to dryness. Toluene (IL) was added and the solution was concentrated to dryness again to give 169.8 g of an oil. MTBE (1190 niL, 7 vol.) was added to the oil at 50 ºC and stirred for 15 minutes. Heptane (850 rnL, 5vol.) was added over ten minutes at 50 ºC. The mixture was then cooled to room temperature over 1.5 h and stirred for 2 h. The slurry was filtered, washed with 1 :4 MBTE/heptane (2 x 100 mL), and dried in an oven overnight at 45 ºC to give 102.3 g of an off-white solid (80% yield, 98.8% AUC by HPLC).
Preparation of methyl 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2-yl)acetate (7)
[000377] Nitrile 6 (101 g) and methanol (1.01 L, 10 vol.) were charged to a 3-L flask equipped with stir bar and thermocouple. Concentrated H2SO4 (175 mL, 10.0 equiv.) was added drop wise to the solution over 15 minutes while maintaining a temperature below 60 ºC. Followed by 30% fuming sulfuric acid (124 mL) was added drop wise to the solution while maintaining a temperature below 60 ºC. The solution was then heated to reflux with a heating mantle and stirred overnight. When the reaction was deemed complete, it was cooled to 20 ºC. In a second flask (22 L), saturated sodium bicarbonate (10.7 L) and dichloromethane (1.1 L) were charged and cooled to 15 ºC. While maintaining a temperature below 20 ºC, the reaction mixture was added to the sodium bicarbonate/dichloromethane mixture. The quench was stirred for 15 minutes and the phases were separated. The aqueous phase was extracted with dichloromethane (I x 55OmL, 1 x 30OmL). The combined organics were dried with magnesium sulfate and concentrated to dryness to give 105 g of an orange solid (94% crude yield, 97.7% AUC by HPLC).
Preparation of 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2-yl)-N-benzylacetamide (KX2-391)
[000378] Ester 7 (103 g), anisole (513 mL, 5 vol.), and benzylamine (94 mL, 3.0 equiv.) were charged to a 3 L flask equipped with thermocouple and overhead stirrer. The reaction mixture was then heated to 142 ºC and stirred for two days. The reaction mixture was cooled to 45-50 ºC and stirred for 2 hours. To the mixture was added n-heptane (1.5 L) dropwise over an hour. The solution was cooled to room temperature over three hours and then stirred overnight. The resulting slurry was filtered, washed with 4:1 Anisole/n-heptane (200 mL) and n-heptane (3 χ100 mL). Drying in the oven overnight, the resulting product was 112. Ig of a tan solid (90% yield, 99.6% AUC by HPLC). The use of a single isomer of heptane was essential to adequately quantitate the residual solvent. See Figure 5 for 1H NMR of KX2- 391. Preparation of 2-(5-(4-(2-morpholinoethoxy)phenyl)pyridin-2-yl)-N-benzylacetamide dihydrochloride salt (KX2-391 2HC1)
[000379] EtOH (1.0 L) was charged to a 2-L flask and acetyl chloride (62.5 niL, 3.0 equiv.) was added slowly to the flask and stirred for 40 minutes. The resulting solution was added to KX2-391 (100 g) over 30 minutes while maintaining a temperature of 30 ºC. The solution was concentrated to a mass of 270 g. The concentrated solution was added to ethyl acetate (2 L) over 20 minutes with rapid stirring. The mixture was stirred overnight and then filtered under nitrogen to give two distinct solid products, tan solids (73.5 g) and darker solids (42.2 g). The solids were dry blended to give a combined yield of 99%. The HPLC analysis indicated 99.0% purity (AUC). Analysis indicated that ethanol was present at 2530 ppm, ethyl acetate at 48,110 ppm, ethyl chloride at 170 ppm, and no heptane and anisole were detected. Palladium content was assayed three times and measured to be 29 ppm, 2 ppm, and less than 1 ppm.
PATENT
CN 106810490
| 2-(5-(4-(2-morpholinylethoxy)phenyl)pyridine-2-yl)-N-benzyl-acetamide, development code KX -01, KX2-391, have the structure shown in formula I. |
| |
| Patent CN10118473B and US7300931B disclose compound KX2-391, and disclose its application in the treatment of cell proliferative disorders. KX2-391 and its pharmaceutically acceptable salts are effective Src tyrosine kinase inhibitors, which can effectively treat diseases and disorders regulated by Src kinase. KX2-391 has a GI50 of 9-60 nM in cancer cell lines and is currently in clinical phase II. |
| KX2-391 has polymorphism. Polymorphism refers to the phenomenon that the same compound can form two or more molecular spatial arrangements by controlling its different production conditions to produce different solid crystals. Different crystal forms of the same compound have the same chemical composition. , But the microscopic crystal structure is different, which leads to differences in their appearance, physical and chemical properties and biological activity. The phenomenon of polymorphism directly affects the processing performance of the drug formulation, and affects the stability, solubility, and bioavailability of the drug, and further affects the quality, safety, effectiveness and application of the drug. Therefore, in drug research and development, the polymorphism of drugs should be fully considered. At present, KX2-391 is still in the research and development stage, and a comprehensive study of its solid form is of great significance to the research and development of KX2-391 and the approval of the market. |
| Example 1 |
| 2-(5-(4-(2-morpholinylethoxy)phenyl)pyridin-2-yl)-N-benzyl-acetamide (KX2-391) crystal form (i.e. having formula (I) The structure of the crystalline diaryl compound, the subsequent examples are referred to as the preparation of KX2-391 crystal form B) |
| Put KX2-391 (5.0g) in a 500ml round bottom flask, add 150ml methanol to dissolve KX2-391 completely, and place it at 50°C and stir. 300ml of purified water was gradually added dropwise. After the addition, the resulting slurry was stirred at room temperature for 1 hour to crystallize, filtered with suction, and dried under vacuum at 50°C. The resulting solid was KX2-391 crystal form B. The purity detected by HPLC is ≥99.83%. |
| Example 2 Preparation of KX2-391 crystal form B |
| Put KX2-391 (5.0g) in a 100ml round bottom flask, add 25ml of DMSO to dissolve KX2-391 completely, and stir at room temperature. Gradually add 50ml of purified water dropwise. After the dropwise addition, the resulting slurry was stirred at 0°C for 1h to crystallize, filtered with suction, and dried under vacuum at 50°C. The resulting solid was KX2-391 crystal form B. HPLC detection purity ≥99.81%. |
| Example 3 Preparation of KX2-391 crystal form B |
| Put KX2-391 (5.0g) in a 250ml round bottom flask, add 15ml of dichloromethane to dissolve KX2-391 completely, and stir at 30°C. Gradually add 100ml of n-heptane dropwise. After the dropwise addition, the resulting slurry was stirred at room temperature for 0.5h to crystallize, filtered with suction, and dried under vacuum at 50°C. The resulting solid was KX2-391 crystal form B. HPLC detection purity ≥99.80%. |
| Example 4 Preparation of KX2-391 crystal form B |
| Put KX2-391 (2.0g) in a 500ml round bottom flask, add 100ml of acetone to completely dissolve KX2-391, and stir at room temperature. Gradually add 150 ml of n-hexane, and after the addition is complete, the resulting slurry is stirred at 0°C for 1 h to crystallize, filtered with suction, and dried in vacuum at 50°C. The obtained solid is KX2-391 crystal form B. HPLC detection purity ≥99.79%. |
| Example 5 Preparation of KX2-391 crystal form B |
| Put KX2-391 (2.0g) in a 250ml round bottom flask, add 50ml of THF to dissolve KX2-391 completely, and place it at 40°C and stir. Gradually add 100 ml of methyl tert-butyl ether dropwise. After the dropwise addition, the resulting slurry was stirred at room temperature for 2 hours to crystallize, filtered with suction, and dried under vacuum at 50°C. The resulting solid was KX2-391 crystal form B. HPLC detection purity ≥99.81%. |
| Example 6 Detection of KX2-391 crystal form B |
| The KX2-391 crystal form B prepared in Example 1 was tested by XRPD method. The equipment used is RIGAKU TTR III X-ray powder diffractometer, measurement conditions and methods: Cu (target), 40KV-30mA (working voltage and current), 2θ=2~50 degrees (scanning range), 4.0deg /min. (scanning speed), the obtained spectrum is shown in Figure 1. It can be seen from Figure 1 that the XRPD spectrum of KX2-391 crystal form B provided in Example 1 is 2.10, 3.68, 4.16, 6.24, 8.33, There are peaks at 12.53, 16.26, 16.75, 18.33, 19.05, 19.85, 21.00, 21.50, 21.92, 22.50, 23.16, 25.08, 25.35, 25.70, 27.49, 29.67, 33.97, and 38.43. |
| The invention also adopts the DSC-TGA method to detect the crystal form B of KX2-391 provided by the invention. The equipment used is METTLER TOLEDO’s TGA-DSC, testing environment conditions 22℃, relative humidity RH68%, temperature range 0-400℃, heating rate 12℃/min, protective gas N 2 , The resulting maps are shown in Figure 2 and Figure 3. It can be seen from Figure 2 that the DSC spectrum of KX2-391 crystal form B provided in Example 1 has endothermic peaks at 126.9°C and 137.4°C. It can be seen from Figure 3 that the TGA pattern of KX2-391 crystal form B provided in Example 1 has no significant weight loss before 200°C. |
PAPER
Journal of Medicinal Chemistry (2018), 61(11), 4704-4719.
[1]. Lau GM, et al. Expression of Src and FAK in hepatocellul
https://pubs.acs.org/doi/10.1021/acs.jmedchem.8b00164
Abstract

The discovery of potent, peptide site directed, tyrosine kinase inhibitors has remained an elusive goal. Herein we describe the discovery of two such clinical candidates that inhibit the tyrosine kinase Src. Compound 1 is a phase 3 clinical trial candidate that is likely to provide a first in class topical treatment for actinic keratosis (AK) with good efficacy and dramatically less toxicity compared to existing standard therapy. Compound 2 is a phase 1 clinical trial candidate that is likely to provide a first in class treatment of malignant glioblastoma and induces 30% long-term complete tumor remission in animal models. The discovery strategy for these compounds iteratively utilized molecular modeling, along with the synthesis and testing of increasingly elaborated proof of concept compounds, until the final clinical candidates were arrived at. This was followed with mechanism of action (MOA) studies that revealed tubulin polymerization inhibition as the second MOA.

[1]. Lau GM, et al. Expression of Src and FAK in hepatocellular carcinoma and the effect of Src inhibitors on hepatocellular carcinoma in vitro. Dig Dis Sci, 2009, 54(7), 1465-1474.
//////////Tirbanibulin, Klisyri, FDA 2020, 2020 APPROVALS, KX2 391, KX 2391, KX-01, actinic Keratosis
O=C(CC1=NC=C(C2=CC=C(OCCN3CCOCC3)C=C2)C=C1)NCC4=CC=CC=C4
Lonafarnib

Lonafarnib
- Molecular FormulaC27H31Br2ClN4O2
- Average mass638.822 Da
193275-84-2[RN]
1-Piperidinecarboxamide, 4-[2-[4-[(11R)-3,10-dibromo-8-chloro-6,11-dihydro-5H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-yl]-1-piperidinyl]-2-oxoethyl]-
4-[2-[4-[(11R)-3,10-Dibromo-8-chloro-6,11-dihydro-5Hbenzo[5,6]cyclohepta[1,2-b]pyridin-11-yl]-1-piperidinyl]-2-oxoethyl]-1-piperidinecarboxamide
8191(+)-4[2-[4-(8-Chloro-3,10-dibromo-6,11-dihydro-5H-benzo[5,6] cyclohepta[1,2-b]-pyridin-11(R)-yl-1-piperidinyl]-2-oxo-ethyl]-1-piperidinecarboxamide
(R)-4-(2-(4-(3,10-dibromo-8-chloro-6,11-dihydro-5H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-yl)piperidin-1-yl)-2-oxoethyl)piperidine-1-carboxamide
4-{2-[4-(3,10-dibromo-8-chloro-6,11-dihydro-5H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-yl)piperidin-1-yl]-2-oxoethyl}piperidine-1-carboxamide
D04768
lonafarnibum TM5989100 UNII:IOW153004F
FDA APPROVED 11/20/2020, Zokinvy
To treat rare conditions related to premature aging
Press Release
Drug Trials Snapshotлонафарниб [Russian] [INN]لونافارنيب [Arabic] [INN]氯那法尼 [Chinese] [INN]
Lonafarnib, sold under the brand name Zokinvy, is a medication used to reduce the risk of death due to Hutchinson-Gilford progeria syndrome and for the treatment of certain processing-deficient progeroid laminopathies in people one year of age and older.[1][2]
The most common side effects included nausea vomiting, diarrhea, infection, decreased appetite and fatigue.[1]
Medical uses
Lonafarnib is indicated to be used to reduce the risk of death due to Hutchinson-Gilford progeria syndrome and for the treatment of certain processing-deficient progeroid laminopathies in people one year of age and older.[1][2]
Contraindications
Lonafarnib is contraindicated for co-administration with strong or moderate CYP3A inhibitors and inducers, as well as midazolam and certain cholesterol-lowering medications.[1]
History
Lonafarnib, a farnesyltransferase inhibitor, is an oral medication that helps prevent the buildup of defective progerin or progerin-like protein.[1] The effectiveness of lonafarnib for the treatment of Hutchinson-Gilford progeria syndrome was demonstrated in 62 patients from two single-arm trials (Trial 1/NCT00425607 and Trial 2/NCT00916747) that were compared to matched, untreated patients from a separate natural history study.[1][2] Compared to untreated patients, the lifespan of Hutchinson-Gilford progeria syndrome patients treated with lonafarnib increased by an average of three months through the first three years of treatment and by an average of 2.5 years through the maximum follow-up time of 11 years.[1] Lonafarnib’s approval for the treatment of certain processing-deficient progeroid laminopathies that are very rare took into account similarities in the underlying genetic mechanism of disease and other available data.[1] The participants were from 34 countries around the world, including the United States.[2]
The U.S. Food and Drug Administration (FDA) granted the application for lonafarnib priority review, orphan drug, and breakthrough therapy designations.[1] In addition, the manufacturer received a rare pediatric disease priority review voucher.[1] The FDA granted the approval of Zokinvy to Eiger BioPharmaceuticals, Inc.[1]

Research
Lonafarnib is a farnesyltransferase inhibitor (FTI) that has been investigated in a human clinical trial as a treatment for progeria, which is an extremely rare genetic disorder in which symptoms resembling aspects of aging are manifested at a very early age.[3][4]
Lonafarnib is a synthetic tricyclic halogenated carboxamide with antineoplastic properties.[5] As such, it is used primarily for cancer treatment. For those with progeria, research has shown that the drug reduces the prevalence of stroke and transient ischemic attack, and the prevalence and frequency of headaches while taking the medication.[6] A phase II clinical trial was completed in 2012, which showed that a cocktail of drugs that included lonafarnib and two other drugs met clinical efficacy endpoints that improved the height and diminished the rigidity of the bones of progeria patients.
SYN
| EP 1019392; EP 1380581; JP 1999501671; WO 9723478 |
Introduction of a bromine atom at the 10-position of the benzocycloheptapyridine (I) was achieved by the following sequence. Nitration of (I) using NaNO3-H2SO4 afforded a mixture of nitro compounds (II) and (III), from which the major 9-nitro isomer (III) was separated by silica gel chromatography. Reduction of the nitro group of (III) with iron filings and CaCl2 in refluxing aqueous ethanol gave amine (IV), which was brominated at position 10 with Br2 in AcOH. The brominated aniline (VI) was then deaminated by diazotization, followed by reduction of the resulting diazonium salt with hypophosphorous acid to give trihalo compound (VI). Hydrolysis of carbamate group of (VI) in boiling concentrated HCl afforded piperidine (VII). Subsequent reduction of the C-11 double bond of (VII) was carried out using DIBAL-H in refluxing toluene to afford the corresponding racemic piperidine. Separation of enantiomers was achieved by HPLC on a ChiralPak AD column or by chemical resolution using N-acetyl-L-phenylalanine as the resolving agent. The appropriate R-(+) enantiomer (VIII) was coupled with N-Boc-piperidylacetic acid (IX) in the presence of EDC and HOBt to yield protected amide (X). Hydrolysis of the Boc protecting group was performed with trifluoroacetic acid, and the resulting piperidine (XI) was finally treated with trimethylsilyl isocyanate to give the desired carboxamide (3-5).
SYN2
EP 1091954; JP 2002519419; WO 0001689
J Org Chem 2000,65(18),5451
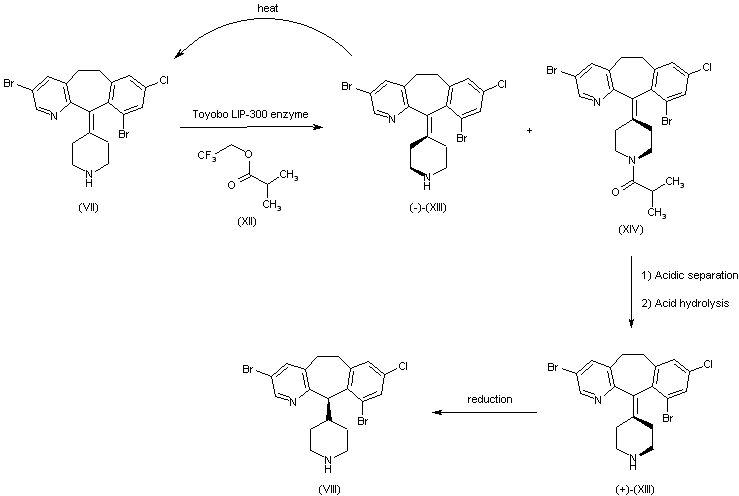
The starting product is the benzocyclohetapyridine (VII), already reported as intermediate (VII) in the synthesis of 25468001a. Compound (VII) is resolved into its atropaisomers by digestion with Toyobo LIP-300 enzyme in the presence of trifluroethyl isobutyrate (XII) to give a mixture of unreacted (-)-(XIII) and acylated compound (+)-(XIV) that are separated by acid extraction. The undesired atropaisomer (-)-(XIII) can be recovered by thermal razemization in diethyleneglycol dibutyl ether at 210 C and new enzymatic separation. The acid hydrolysis of the separated amide (+)-(XIV) produces the desired atropaisomer (+)-(XIII), which is reduced to the (R)-(+)-(VIII), intermediate already reported with no. (VIII) in the synthesis of 25468001a. (6,7)
SYN 3
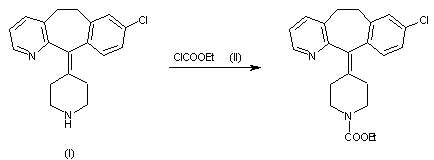
1) By carboxylation of 8-chloro-6,11-dihydro-11-(4-piperidylidene)-5H-benzo[5,6]cyctohepta[1,2-b]pyridine (I) with ethyl chloroformate (II) in refluxing benzene.
SYN 4
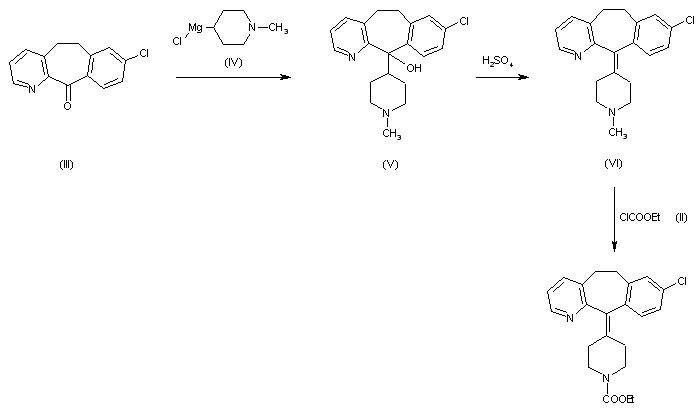
2) By reaction of 8-chloro-6,11-dihydro-5H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-one (III) with the Grignard reagent (IV) to give the tertiary carbinol (V), which is dehydrated with 85% H2SO4 affording 8-chloro-11-piperidinylidene derivative (VI). Finally, cornpound (VI) is treated with ethyl chloroformate (II) in toluene.
SYN 5
J Med Chem 1997,40(26),4290
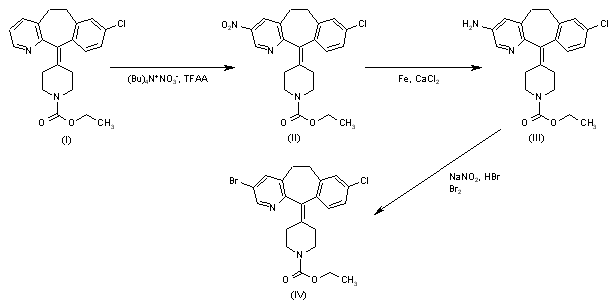
The nitration of loratadine (I) (1) by means of tetrabutylammonium nitrate and trifluoroacetic anhydride (TFAA) in dichloromethane gives the 3-nitro derivative (II), which is reduced with iron filings and CaCl2 in refluxing ethanol/water to yield the 3-amino derivative (III). Treatment of compound (III) with NaNO2, HBr and Br2 provides 4-(3-bromo-8-chloro-5,6-dihydro-1H-benzo[5,6]-cyclohepta[1,2-b]pyridin-11-ylidene)piperidine-1-carboxylic acid ethyl ester (IV) (see scheme 25468001a, intermediate (I).(2)
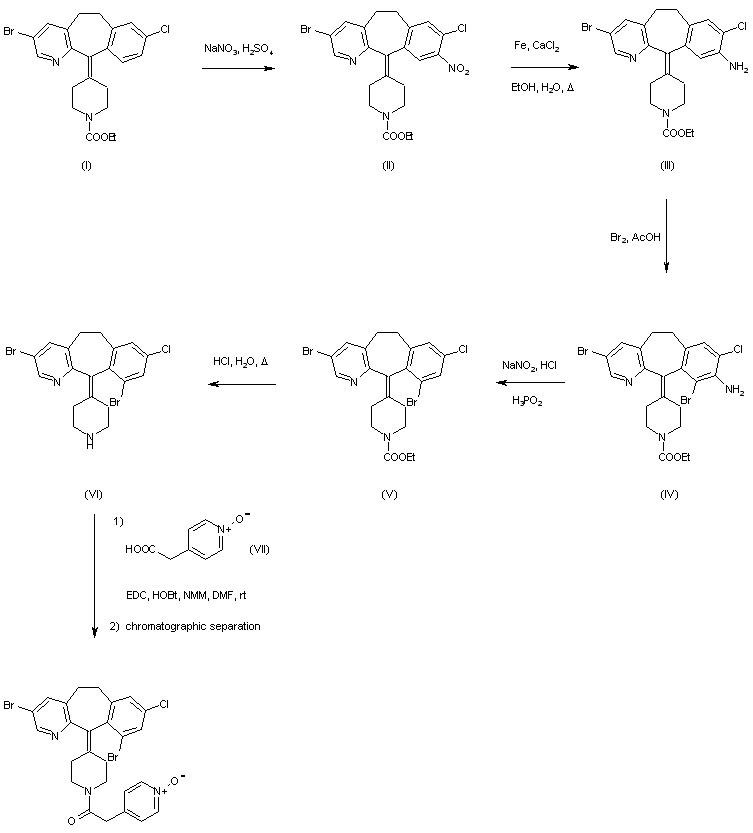
Benzocycloheptapyridine (I) was nitrated with NaNO3 and H2SO4 to afford (II) as the major isomer. Reduction of (III) with iron and CaCl2 gave amine (III), which was brominated to provide (IV). Removal of the amino group of (IV) was accomplished by diazotization, followed by reduction with hypophosphorous acid to give (V). Then, hydrolysis of the carbamate group of (V) in refluxing hydrochloric acid furnished piperidine (VI). Subsequent coupling of (VI) with pyridineacetic acid N-oxide (VII) using EDC and HOBt yielded the corresponding amide. Finally, separation of the target (+)-atropoisomer was achieved by chiral chromatography.
References
- ^ Jump up to:a b c d e f g h i j k “FDA Approves First Treatment for Hutchinson-Gilford Progeria Syndrome and Some Progeroid Laminopathies”. U.S. Food and Drug Administration (FDA) (Press release). 20 November 2020. Retrieved 20 November 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Jump up to:a b c d “Drug Trials Snapshots: Zokinvy”. U.S. Food and Drug Administration. 20 November 2020. Retrieved 11 December 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Liu G, Marrinan CH, Taylor SA, Black S, Basso AD, Kirschmeier P, et al. (September 2007). “Enhancement of the antitumor activity of tamoxifen and anastrozole by the farnesyltransferase inhibitor lonafarnib (SCH66336)”. Anti-Cancer Drugs. 18 (8): 923–31. doi:10.1097/CAD.0b013e3280c1416e (inactive 2020-09-10). PMID 17667598.
- ^ “The FTI Drug Lonafarnib”, Progeria Research Foundation. Accessed October 3, 2017.
- ^ “Lonafarnib”. NCI Drug Dictionary. National Cancer Institute. 2011-02-02.
- ^ Ullrich NJ, Kieran MW, Miller DT, Gordon LB, Cho YJ, Silvera VM, et al. (July 2013). “Neurologic features of Hutchinson-Gilford progeria syndrome after lonafarnib treatment”. Neurology. 81 (5): 427–30. doi:10.1212/WNL.0b013e31829d85c0. PMC 3776537. PMID 23897869.
External links
- “Lonafarnib”. Drug Information Portal. U.S. National Library of Medicine.
- “Experimental Drug Is First To Help Kids With Premature-Aging Disease”, NPR, September 24, 2012
- Clinical trial number NCT00425607 for “Phase II Trial of Lonafarnib (a Farnesyltransferase Inhibitor) for Progeria” at ClinicalTrials.gov
- Clinical trial number NCT00916747 for “Study of Zoledronic Acid, Pravastatin, and Lonafarnib for Patients With Progeria” at ClinicalTrials.gov
| Clinical data | |
|---|---|
| Trade names | Zokinvy |
| Other names | SCH 66336 |
| License data | US DailyMed: Lonafarnib |
| ATC code | None |
| Legal status | |
| Legal status | US: ℞-only |
| Identifiers | |
| IUPAC name[show] | |
| CAS Number | 193275-84-2 |
| PubChem CID | 148195 |
| IUPHAR/BPS | 8024 |
| DrugBank | DB06448 |
| ChemSpider | 130645 |
| UNII | IOW153004F |
| KEGG | D04768 |
| ChEBI | CHEBI:47097 |
| ChEMBL | ChEMBL298734 |
| PDB ligand | 336 (PDBe, RCSB PDB) |
| CompTox Dashboard (EPA) | DTXSID90172927 |
| ECHA InfoCard | 100.204.509 |
| Chemical and physical data | |
| Formula | C27H31Br2ClN4O2 |
| Molar mass | 638.83 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES[hide]C=12CCC=3C=C(C=C(C3[C@H](C1N=CC(=C2)Br)C4CCN(CC4)C(=O)CC5CCN(CC5)C(N)=O)Br)Cl | |
| InChI[hide]InChI=1S/C27H31Br2ClN4O2/c28-20-12-19-2-1-18-13-21(30)14-22(29)24(18)25(26(19)32-15-20)17-5-9-33(10-6-17)23(35)11-16-3-7-34(8-4-16)27(31)36/h12-17,25H,1-11H2,(H2,31,36)/t25-/m1/s1 Key:DHMTURDWPRKSOA-RUZDIDTESA-N |
/////////lonafarnib, Zokinvy, FDA 2020, 2020 APPROVALS, лонафарниб , لونافارنيب , 氯那法尼 , D 04768, lonafarnibum, TM 5989100
Gallium 68 PSMA-11


Gallium 68 PSMA-11
FDA APPROVED, 12/1/2020, Gallium 68 PSMA-11
For detection and localization of prostate cancer
Press Release
Drug Trials Snapshot

Chemical structure of 18F-labeled radiotracers. [18F]DCFPyL (A), [18F]PSMA-1007 (B), [18F]CTT1057 (C), (D) [18F]JK-PSMA-7 and (E) [18F]AIF-PSMA-11. The urea backbone of (A), (B), (D) and (E) is marked in blue, while the phosphoramidate of [18F]CTT1057 in (C) is highlighted in orange. Modified from Behr et al. [32], © by the Society of Nuclear Medicine and Molecular Imaging, Inc.
PSMA-11, also known as HBED-CC-PSMA or Psma-hbed-CC, is used to make gallium Ga 68-labeled PSMA-11, which has potential use as a tracer for PSMA-expressing tumors during positron emission tomography (PET). Upon intravenous administration of gallium Ga 68-labeled PSMA-11, the Glu-urea-Lys(Ahx) moiety targets and binds to PSMA-expressing tumor cells. Upon internalization, PSMA-expressing tumor cells can be detected during PET imaging. PSMA, a tumor-associated antigen and type II transmembrane protein, is expressed on the membrane of prostatic epithelial cells and overexpressed on prostate tumor cells

Name: PSMA-11
CAS#: 1366302-52-4
Chemical Formula: C44H62N6O17
Exact Mass: 946.4171
(3S,7S)-22-(3-(((2-((5-(2-Carboxyethyl)-2-hydroxybenzyl)(carboxymethyl)amino)ethyl)(carboxymethyl)amino)methyl)-4-hydroxyphenyl)-5,13,20-trioxo-4,6,12,19-tetraazadocosane-1,3,7-tricarboxylic acid
The Food and Drug Administration (FDA) has approved Gallium 68 PSMA-11 (Ga 68 PSMA-11), the first drug for positron emission tomography (PET) imaging of prostate-specific membrane antigen (PSMA) positive lesions in men with prostate cancer.
Ga 68 PSMA-11, a radioactive diagnostic agent, is indicated for patients with suspected prostate cancer metastasis who are potentially curable by surgery or radiation therapy. It is also indicated for patients with suspected prostate cancer recurrence based on elevated serum prostate-specific antigen (PSA) levels.
The approval was based on efficacy and safety data from 2 prospective clinical trials (Trial 1 and 2) with a total of 960 men with prostate cancer who each received 1 injection of Ga 68 PSMA-11. Trial 1 included 325 patients with biopsy-proven prostate cancer who underwent PET/CT or PET/MRI scans performed with Ga 68 PSMA-11. Results from the study showed that positive readings in the pelvic lymph nodes on Ga 68 PSMA-11 PET were associated with a clinically important rate of metastatic cancer confirmed by surgical pathology in those who proceeded to surgery.
In Trial 2, 635 patients with rising serum PSA levels after prostate surgery or radiotherapy received a single Ga 68 PSMA-11 PET/CT scan or PET/MR scan. Findings demonstrated that 74% of patients had at least 1 positive lesion detected by Ga 68 PSMA-11 PET, and local recurrence or metastasis of prostate cancer was confirmed in 91% of cases.
 This is the first drug approved for PET imaging of prostate-specific membrane antigen positive lesions in men with prostate cancer.
This is the first drug approved for PET imaging of prostate-specific membrane antigen positive lesions in men with prostate cancer.
REF
REFERENCES
1: Meißner S, Janssen JC, Prasad V, Brenner W, Diederichs G, Hamm B, Hofheinz F, Makowski MR. Potential of asphericity as a novel diagnostic parameter in the evaluation of patients with (68)Ga-PSMA-HBED-CC PET-positive prostate cancer lesions. EJNMMI Res. 2017 Oct 23;7(1):85. doi: 10.1186/s13550-017-0333-9. PubMed PMID: 29058157; PubMed Central PMCID: PMC5651532.
2: Verburg FA, Pfister D, Drude NI, Mottaghy FM, Behrendt F. PSA levels, PSA doubling time, Gleason score and prior therapy cannot predict measured uptake of [(68)Ga]PSMA-HBED-CC lesion uptake in recurrent/metastatic prostate cancer. Nuklearmedizin. 2017 Oct 18;56(6). doi: 10.3413/Nukmed-0917-17-07. [Epub ahead of print] PubMed PMID: 29044297.
3: Amor-Coarasa A, Kelly JM, Gruca M, Nikolopoulou A, Vallabhajosula S, Babich JW. Continuation of comprehensive quality control of the itG (68)Ge/(68)Ga generator and production of (68)Ga-DOTATOC and (68)Ga-PSMA-HBED-CC for clinical research studies. Nucl Med Biol. 2017 Oct;53:37-39. doi: 10.1016/j.nucmedbio.2017.07.006. Epub 2017 Jul 14. PubMed PMID: 28803001.
4: Janssen JC, Woythal N, Meißner S, Prasad V, Brenner W, Diederichs G, Hamm B, Makowski MR. [(68)Ga]PSMA-HBED-CC Uptake in Osteolytic, Osteoblastic, and Bone Marrow Metastases of Prostate Cancer Patients. Mol Imaging Biol. 2017 Dec;19(6):933-943. doi: 10.1007/s11307-017-1101-y. PubMed PMID: 28707038.
5: Damle NA, Tripathi M, Chakraborty PS, Sahoo MK, Bal C, Aggarwal S, Arora G, Kumar P, Kumar R, Gupta R. Unusual Uptake of Prostate Specific Tracer (68)Ga-PSMA-HBED-CC in a Benign Thyroid Nodule. Nucl Med Mol Imaging. 2016 Dec;50(4):344-347. Epub 2016 Mar 22. PubMed PMID: 27994690; PubMed Central PMCID: PMC5135692.
6: Behrendt F, Krohn T, Mottaghy F, Verburg FA. [(68)Ga]PSMA-HBED-CC PET/CT to differentiate between diffuse bone metastases of prostate cancer and osteopoikilosis. Nuklearmedizin. 2016 Dec 6;55(6):N64-N65. PubMed PMID: 27922151.
7: Krohn T, Birmes A, Winz OH, Drude NI, Mottaghy FM, Behrendt FF, Verburg FA. The reconstruction algorithm used for [(68)Ga]PSMA-HBED-CC PET/CT reconstruction significantly influences the number of detected lymph node metastases and coeliac ganglia. Eur J Nucl Med Mol Imaging. 2017 Apr;44(4):662-669. doi: 10.1007/s00259-016-3571-6. Epub 2016 Nov 29. PubMed PMID: 27900518.
8: Berliner C, Tienken M, Frenzel T, Kobayashi Y, Helberg A, Kirchner U, Klutmann S, Beyersdorff D, Budäus L, Wester HJ, Mester J, Bannas P. Detection rate of PET/CT in patients with biochemical relapse of prostate cancer using [(68)Ga]PSMA I&T and comparison with published data of [(68)Ga]PSMA HBED-CC. Eur J Nucl Med Mol Imaging. 2017 Apr;44(4):670-677. doi: 10.1007/s00259-016-3572-5. Epub 2016 Nov 28. PubMed PMID: 27896369.
9: Sathekge M, Lengana T, Modiselle M, Vorster M, Zeevaart J, Maes A, Ebenhan T, Van de Wiele C. (68)Ga-PSMA-HBED-CC PET imaging in breast carcinoma patients. Eur J Nucl Med Mol Imaging. 2017 Apr;44(4):689-694. doi: 10.1007/s00259-016-3563-6. Epub 2016 Nov 8. PubMed PMID: 27822700; PubMed Central PMCID: PMC5323468.
10: Rauscher I, Maurer T, Beer AJ, Graner FP, Haller B, Weirich G, Doherty A, Gschwend JE, Schwaiger M, Eiber M. Value of 68Ga-PSMA HBED-CC PET for the Assessment of Lymph Node Metastases in Prostate Cancer Patients with Biochemical Recurrence: Comparison with Histopathology After Salvage Lymphadenectomy. J Nucl Med. 2016 Nov;57(11):1713-1719. Epub 2016 Jun 3. PubMed PMID: 27261524.
11: Verburg FA, Behrendt FF, Mottaghy FM, Pfister D, Steib F, Knuechel R. Strong [(68)Ga]PSMA-HBED-CC accumulation in non-cancerous prostate tissue surrounding a PSMA-negative prostate carcinoma recurrence. Nuklearmedizin. 2016 Sep 26;55(5):N44-5. PubMed PMID: 27668299.
12: Kanthan GL, Izard MA, Emmett L, Hsiao E, Schembri GP. Schwannoma Showing Avid Uptake on 68Ga-PSMA-HBED-CC PET/CT. Clin Nucl Med. 2016 Sep;41(9):703-4. doi: 10.1097/RLU.0000000000001281. PubMed PMID: 27405039.
13: Noto B, Vrachimis A, Schäfers M, Stegger L, Rahbar K. Subacute Stroke Mimicking Cerebral Metastasis in 68Ga-PSMA-HBED-CC PET/CT. Clin Nucl Med. 2016 Oct;41(10):e449-51. doi: 10.1097/RLU.0000000000001291. PubMed PMID: 27355852.
14: Pfob CH, Ziegler S, Graner FP, Köhner M, Schachoff S, Blechert B, Wester HJ, Scheidhauer K, Schwaiger M, Maurer T, Eiber M. Biodistribution and radiation dosimetry of (68)Ga-PSMA HBED CC-a PSMA specific probe for PET imaging of prostate cancer. Eur J Nucl Med Mol Imaging. 2016 Oct;43(11):1962-70. doi: 10.1007/s00259-016-3424-3. Epub 2016 May 20. PubMed PMID: 27207281.
15: Amor-Coarasa A, Schoendorf M, Meckel M, Vallabhajosula S, Babich JW. Comprehensive Quality Control of the ITG 68Ge/68Ga Generator and Synthesis of 68Ga-DOTATOC and 68Ga-PSMA-HBED-CC for Clinical Imaging. J Nucl Med. 2016 Sep;57(9):1402-5. doi: 10.2967/jnumed.115.171249. Epub 2016 Apr 21. PubMed PMID: 27103024.
16: Prasad V, Steffen IG, Diederichs G, Makowski MR, Wust P, Brenner W. Biodistribution of [(68)Ga]PSMA-HBED-CC in Patients with Prostate Cancer: Characterization of Uptake in Normal Organs and Tumour Lesions. Mol Imaging Biol. 2016 Jun;18(3):428-36. doi: 10.1007/s11307-016-0945-x. PubMed PMID: 27038316.
17: Pfister D, Porres D, Heidenreich A, Heidegger I, Knuechel R, Steib F, Behrendt FF, Verburg FA. Detection of recurrent prostate cancer lesions before salvage lymphadenectomy is more accurate with (68)Ga-PSMA-HBED-CC than with (18)F-Fluoroethylcholine PET/CT. Eur J Nucl Med Mol Imaging. 2016 Jul;43(8):1410-7. doi: 10.1007/s00259-016-3366-9. Epub 2016 Mar 19. PubMed PMID: 26993315.
18: Kanthan GL, Coyle L, Kneebone A, Schembri GP, Hsiao E. Follicular Lymphoma Showing Avid Uptake on 68Ga PSMA-HBED-CC PET/CT. Clin Nucl Med. 2016 Jun;41(6):500-1. doi: 10.1097/RLU.0000000000001169. PubMed PMID: 26914565.
19: Kanthan GL, Hsiao E, Kneebone A, Eade T, Schembri GP. Desmoid Tumor Showing Intense Uptake on 68Ga PSMA-HBED-CC PET/CT. Clin Nucl Med. 2016 Jun;41(6):508-9. doi: 10.1097/RLU.0000000000001192. PubMed PMID: 26909712.
20: Eiber M, Weirich G, Holzapfel K, Souvatzoglou M, Haller B, Rauscher I, Beer AJ, Wester HJ, Gschwend J, Schwaiger M, Maurer T. Simultaneous (68)Ga-PSMA HBED-CC PET/MRI Improves the Localization of Primary Prostate Cancer. Eur Urol. 2016 Nov;70(5):829-836. doi: 10.1016/j.eururo.2015.12.053. Epub 2016 Jan 18. PubMed PMID: 26795686.
//////////Gallium 68 PSMA-11, FDA 2020, 2020 APPROVALS, RADIO ACTIVE

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....