Lonafarnib
- Molecular FormulaC27H31Br2ClN4O2
- Average mass638.822 Da
193275-84-2[RN]
1-Piperidinecarboxamide, 4-[2-[4-[(11R)-3,10-dibromo-8-chloro-6,11-dihydro-5H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-yl]-1-piperidinyl]-2-oxoethyl]-
4-[2-[4-[(11R)-3,10-Dibromo-8-chloro-6,11-dihydro-5Hbenzo[5,6]cyclohepta[1,2-b]pyridin-11-yl]-1-piperidinyl]-2-oxoethyl]-1-piperidinecarboxamide
8191(+)-4[2-[4-(8-Chloro-3,10-dibromo-6,11-dihydro-5H-benzo[5,6] cyclohepta[1,2-b]-pyridin-11(R)-yl-1-piperidinyl]-2-oxo-ethyl]-1-piperidinecarboxamide
(R)-4-(2-(4-(3,10-dibromo-8-chloro-6,11-dihydro-5H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-yl)piperidin-1-yl)-2-oxoethyl)piperidine-1-carboxamide
4-{2-[4-(3,10-dibromo-8-chloro-6,11-dihydro-5H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-yl)piperidin-1-yl]-2-oxoethyl}piperidine-1-carboxamide
D04768
lonafarnibum TM5989100 UNII:IOW153004F
FDA APPROVED 11/20/2020, Zokinvy
To treat rare conditions related to premature aging
Press Release
Drug Trials Snapshotлонафарниб [Russian] [INN]لونافارنيب [Arabic] [INN]氯那法尼 [Chinese] [INN]
Lonafarnib, sold under the brand name Zokinvy, is a medication used to reduce the risk of death due to Hutchinson-Gilford progeria syndrome and for the treatment of certain processing-deficient progeroid laminopathies in people one year of age and older.[1][2]
The most common side effects included nausea vomiting, diarrhea, infection, decreased appetite and fatigue.[1]
Medical uses
Lonafarnib is indicated to be used to reduce the risk of death due to Hutchinson-Gilford progeria syndrome and for the treatment of certain processing-deficient progeroid laminopathies in people one year of age and older.[1][2]
Contraindications
Lonafarnib is contraindicated for co-administration with strong or moderate CYP3A inhibitors and inducers, as well as midazolam and certain cholesterol-lowering medications.[1]
History
Lonafarnib, a farnesyltransferase inhibitor, is an oral medication that helps prevent the buildup of defective progerin or progerin-like protein.[1] The effectiveness of lonafarnib for the treatment of Hutchinson-Gilford progeria syndrome was demonstrated in 62 patients from two single-arm trials (Trial 1/NCT00425607 and Trial 2/NCT00916747) that were compared to matched, untreated patients from a separate natural history study.[1][2] Compared to untreated patients, the lifespan of Hutchinson-Gilford progeria syndrome patients treated with lonafarnib increased by an average of three months through the first three years of treatment and by an average of 2.5 years through the maximum follow-up time of 11 years.[1] Lonafarnib’s approval for the treatment of certain processing-deficient progeroid laminopathies that are very rare took into account similarities in the underlying genetic mechanism of disease and other available data.[1] The participants were from 34 countries around the world, including the United States.[2]
The U.S. Food and Drug Administration (FDA) granted the application for lonafarnib priority review, orphan drug, and breakthrough therapy designations.[1] In addition, the manufacturer received a rare pediatric disease priority review voucher.[1] The FDA granted the approval of Zokinvy to Eiger BioPharmaceuticals, Inc.[1]

Research
Lonafarnib is a farnesyltransferase inhibitor (FTI) that has been investigated in a human clinical trial as a treatment for progeria, which is an extremely rare genetic disorder in which symptoms resembling aspects of aging are manifested at a very early age.[3][4]
Lonafarnib is a synthetic tricyclic halogenated carboxamide with antineoplastic properties.[5] As such, it is used primarily for cancer treatment. For those with progeria, research has shown that the drug reduces the prevalence of stroke and transient ischemic attack, and the prevalence and frequency of headaches while taking the medication.[6] A phase II clinical trial was completed in 2012, which showed that a cocktail of drugs that included lonafarnib and two other drugs met clinical efficacy endpoints that improved the height and diminished the rigidity of the bones of progeria patients.
SYN
| EP 1019392; EP 1380581; JP 1999501671; WO 9723478 |
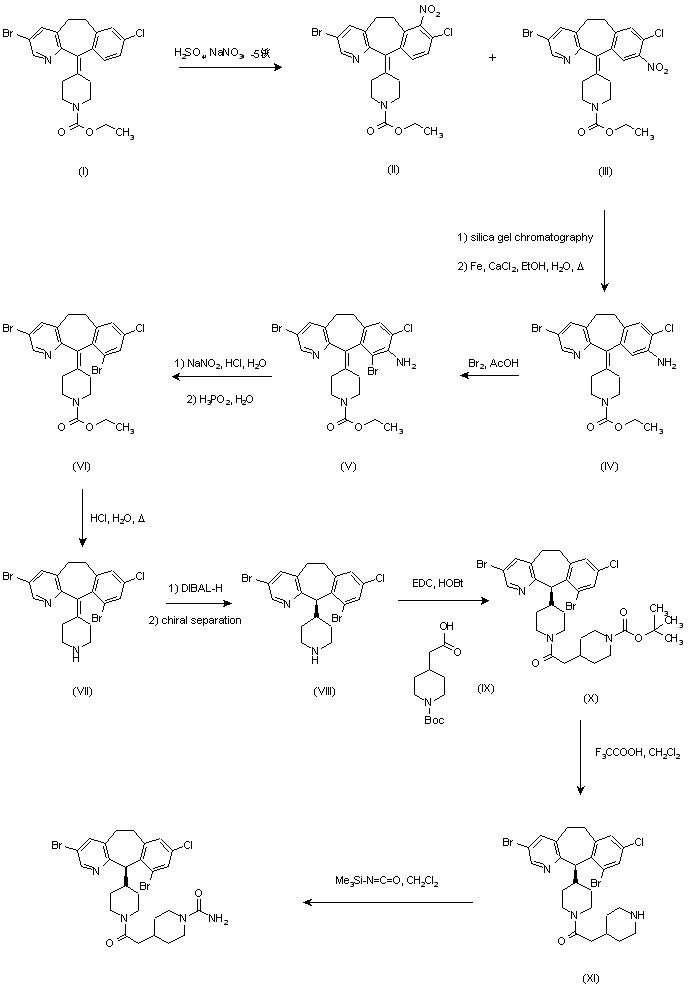
Introduction of a bromine atom at the 10-position of the benzocycloheptapyridine (I) was achieved by the following sequence. Nitration of (I) using NaNO3-H2SO4 afforded a mixture of nitro compounds (II) and (III), from which the major 9-nitro isomer (III) was separated by silica gel chromatography. Reduction of the nitro group of (III) with iron filings and CaCl2 in refluxing aqueous ethanol gave amine (IV), which was brominated at position 10 with Br2 in AcOH. The brominated aniline (VI) was then deaminated by diazotization, followed by reduction of the resulting diazonium salt with hypophosphorous acid to give trihalo compound (VI). Hydrolysis of carbamate group of (VI) in boiling concentrated HCl afforded piperidine (VII). Subsequent reduction of the C-11 double bond of (VII) was carried out using DIBAL-H in refluxing toluene to afford the corresponding racemic piperidine. Separation of enantiomers was achieved by HPLC on a ChiralPak AD column or by chemical resolution using N-acetyl-L-phenylalanine as the resolving agent. The appropriate R-(+) enantiomer (VIII) was coupled with N-Boc-piperidylacetic acid (IX) in the presence of EDC and HOBt to yield protected amide (X). Hydrolysis of the Boc protecting group was performed with trifluoroacetic acid, and the resulting piperidine (XI) was finally treated with trimethylsilyl isocyanate to give the desired carboxamide (3-5).
SYN2
EP 1091954; JP 2002519419; WO 0001689
J Org Chem 2000,65(18),5451
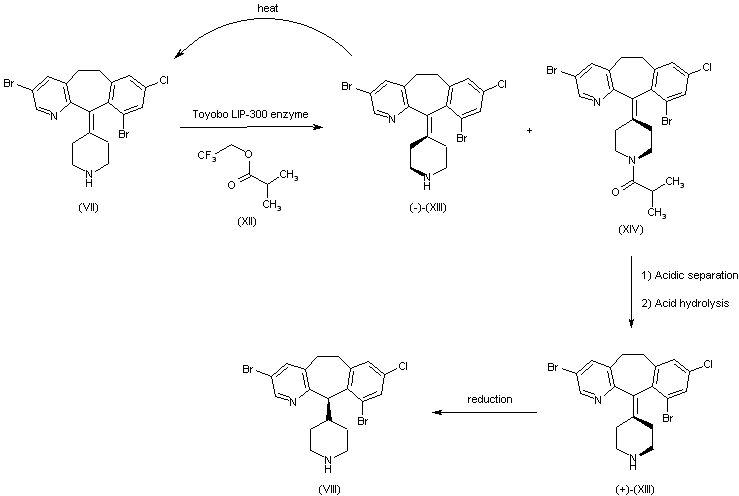
The starting product is the benzocyclohetapyridine (VII), already reported as intermediate (VII) in the synthesis of 25468001a. Compound (VII) is resolved into its atropaisomers by digestion with Toyobo LIP-300 enzyme in the presence of trifluroethyl isobutyrate (XII) to give a mixture of unreacted (-)-(XIII) and acylated compound (+)-(XIV) that are separated by acid extraction. The undesired atropaisomer (-)-(XIII) can be recovered by thermal razemization in diethyleneglycol dibutyl ether at 210 C and new enzymatic separation. The acid hydrolysis of the separated amide (+)-(XIV) produces the desired atropaisomer (+)-(XIII), which is reduced to the (R)-(+)-(VIII), intermediate already reported with no. (VIII) in the synthesis of 25468001a. (6,7)
SYN 3
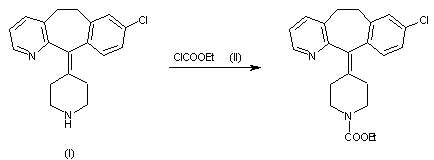
1) By carboxylation of 8-chloro-6,11-dihydro-11-(4-piperidylidene)-5H-benzo[5,6]cyctohepta[1,2-b]pyridine (I) with ethyl chloroformate (II) in refluxing benzene.
SYN 4
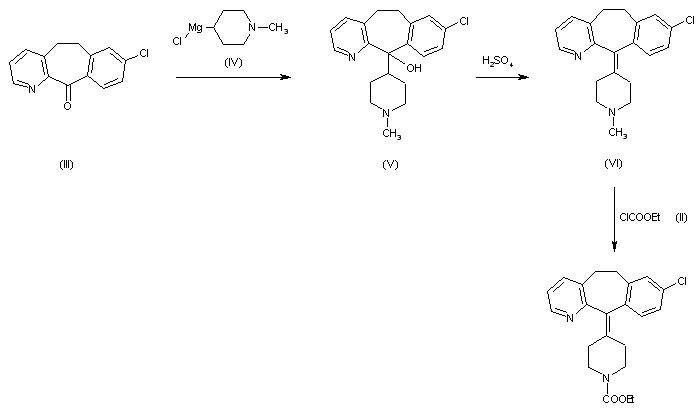
2) By reaction of 8-chloro-6,11-dihydro-5H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-one (III) with the Grignard reagent (IV) to give the tertiary carbinol (V), which is dehydrated with 85% H2SO4 affording 8-chloro-11-piperidinylidene derivative (VI). Finally, cornpound (VI) is treated with ethyl chloroformate (II) in toluene.
SYN 5
J Med Chem 1997,40(26),4290
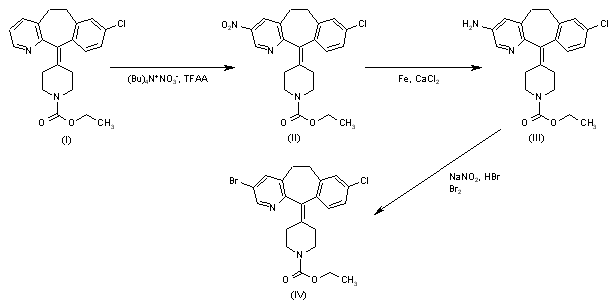
The nitration of loratadine (I) (1) by means of tetrabutylammonium nitrate and trifluoroacetic anhydride (TFAA) in dichloromethane gives the 3-nitro derivative (II), which is reduced with iron filings and CaCl2 in refluxing ethanol/water to yield the 3-amino derivative (III). Treatment of compound (III) with NaNO2, HBr and Br2 provides 4-(3-bromo-8-chloro-5,6-dihydro-1H-benzo[5,6]-cyclohepta[1,2-b]pyridin-11-ylidene)piperidine-1-carboxylic acid ethyl ester (IV) (see scheme 25468001a, intermediate (I).(2)
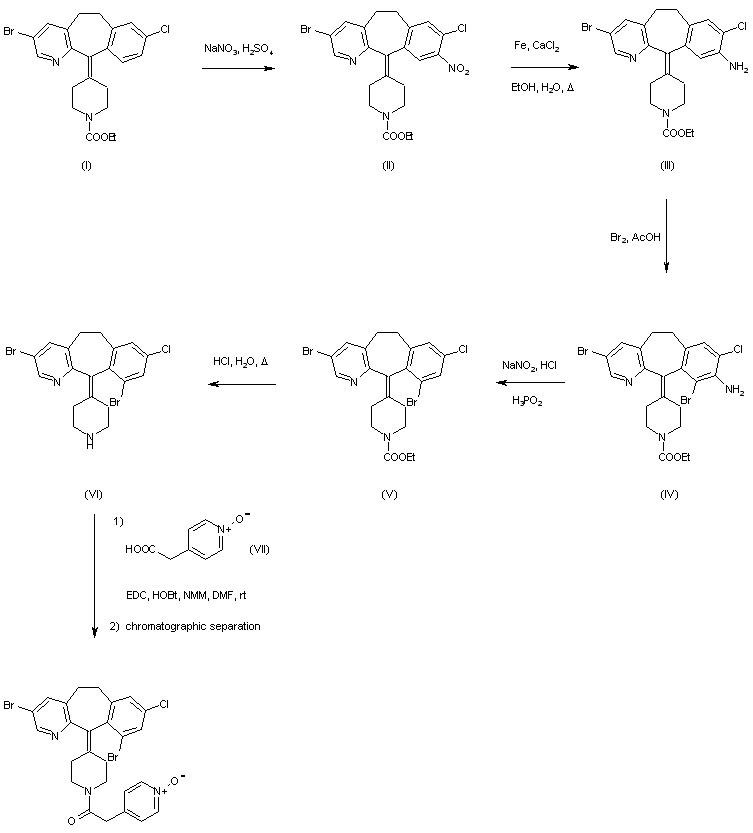
Benzocycloheptapyridine (I) was nitrated with NaNO3 and H2SO4 to afford (II) as the major isomer. Reduction of (III) with iron and CaCl2 gave amine (III), which was brominated to provide (IV). Removal of the amino group of (IV) was accomplished by diazotization, followed by reduction with hypophosphorous acid to give (V). Then, hydrolysis of the carbamate group of (V) in refluxing hydrochloric acid furnished piperidine (VI). Subsequent coupling of (VI) with pyridineacetic acid N-oxide (VII) using EDC and HOBt yielded the corresponding amide. Finally, separation of the target (+)-atropoisomer was achieved by chiral chromatography.
References
- ^ Jump up to:a b c d e f g h i j k “FDA Approves First Treatment for Hutchinson-Gilford Progeria Syndrome and Some Progeroid Laminopathies”. U.S. Food and Drug Administration (FDA) (Press release). 20 November 2020. Retrieved 20 November 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Jump up to:a b c d “Drug Trials Snapshots: Zokinvy”. U.S. Food and Drug Administration. 20 November 2020. Retrieved 11 December 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Liu G, Marrinan CH, Taylor SA, Black S, Basso AD, Kirschmeier P, et al. (September 2007). “Enhancement of the antitumor activity of tamoxifen and anastrozole by the farnesyltransferase inhibitor lonafarnib (SCH66336)”. Anti-Cancer Drugs. 18 (8): 923–31. doi:10.1097/CAD.0b013e3280c1416e (inactive 2020-09-10). PMID 17667598.
- ^ “The FTI Drug Lonafarnib”, Progeria Research Foundation. Accessed October 3, 2017.
- ^ “Lonafarnib”. NCI Drug Dictionary. National Cancer Institute. 2011-02-02.
- ^ Ullrich NJ, Kieran MW, Miller DT, Gordon LB, Cho YJ, Silvera VM, et al. (July 2013). “Neurologic features of Hutchinson-Gilford progeria syndrome after lonafarnib treatment”. Neurology. 81 (5): 427–30. doi:10.1212/WNL.0b013e31829d85c0. PMC 3776537. PMID 23897869.
External links
- “Lonafarnib”. Drug Information Portal. U.S. National Library of Medicine.
- “Experimental Drug Is First To Help Kids With Premature-Aging Disease”, NPR, September 24, 2012
- Clinical trial number NCT00425607 for “Phase II Trial of Lonafarnib (a Farnesyltransferase Inhibitor) for Progeria” at ClinicalTrials.gov
- Clinical trial number NCT00916747 for “Study of Zoledronic Acid, Pravastatin, and Lonafarnib for Patients With Progeria” at ClinicalTrials.gov
| Clinical data | |
|---|---|
| Trade names | Zokinvy |
| Other names | SCH 66336 |
| License data | US DailyMed: Lonafarnib |
| ATC code | None |
| Legal status | |
| Legal status | US: ℞-only |
| Identifiers | |
| IUPAC name[show] | |
| CAS Number | 193275-84-2 |
| PubChem CID | 148195 |
| IUPHAR/BPS | 8024 |
| DrugBank | DB06448 |
| ChemSpider | 130645 |
| UNII | IOW153004F |
| KEGG | D04768 |
| ChEBI | CHEBI:47097 |
| ChEMBL | ChEMBL298734 |
| PDB ligand | 336 (PDBe, RCSB PDB) |
| CompTox Dashboard (EPA) | DTXSID90172927 |
| ECHA InfoCard | 100.204.509 |
| Chemical and physical data | |
| Formula | C27H31Br2ClN4O2 |
| Molar mass | 638.83 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES[hide]C=12CCC=3C=C(C=C(C3[C@H](C1N=CC(=C2)Br)C4CCN(CC4)C(=O)CC5CCN(CC5)C(N)=O)Br)Cl | |
| InChI[hide]InChI=1S/C27H31Br2ClN4O2/c28-20-12-19-2-1-18-13-21(30)14-22(29)24(18)25(26(19)32-15-20)17-5-9-33(10-6-17)23(35)11-16-3-7-34(8-4-16)27(31)36/h12-17,25H,1-11H2,(H2,31,36)/t25-/m1/s1 Key:DHMTURDWPRKSOA-RUZDIDTESA-N |
/////////lonafarnib, Zokinvy, FDA 2020, 2020 APPROVALS, лонафарниб , لونافارنيب , 氯那法尼 , D 04768, lonafarnibum, TM 5989100

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....