SYN 01

SYN-01, SYN-510
Synthena AG
Preclinical
Synthena , presumed to be under license from University of Bern , is investigating (presumably SYN-01 ), a lead from the tricyclo(tc)-DNA based antisense oligonucleotides (AON) developed using its proprietary tricyclo-DNA technology platform, for the treatment of Duchenne muscular dystrophy. In January 2017, the drug was listed as being in preclinical development.
Patent
WO-2019142135
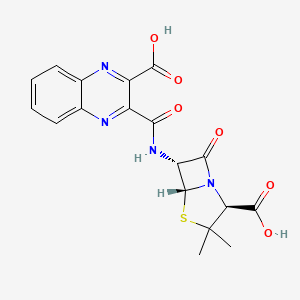
Process for preparing tricyclo-deoxyribonucleic acid (tc-DNA) which may be used as building blocks for tc-DNA containing antisense oligonucleotide-based therapies.
Antisense technology is an effective means for reducing the expression of specific gene products and can therefore be useful in therapeutic, diagnostic, and research applications.
Generally, the principle behind antisense technology is that an antisense oligomeric compound (a sequence of nucleotides or analogues thereof) hybridizes to a target nucleic acid and modulates gene expression activities or function, such as transcription and/or translation.
[003] Antisense oligomeric compounds may be prepared from chemically-modified antisense oligonucleotides, which may include a variety of different structural variations depending upon the therapeutic strategy. For example, tricyclo-deoxyribonucleic acids (tc-DNA) are conformationally constrained DNA analogs.
[004] There is a need in the field for processes that allow for the bulk preparation of tc-DNA nucleoside precursors that may be used as building blocks for tc-DNA containing antisense oligonucleotide-based therapies.
Example 4 – Cvclopropanation of Compound 17 with Carbenoid Prepared from CH2I2 and Et2Zn in the Absence of Additives
[00127] According to the following scheme, compound 17 was converted to tc-DNA Nucleoside Precursor 18 using the cyclopropanation conditions set forth in Examples 4 to 7 :
[00128] 1.07 g purified a-anomer (3.736 mmol) 17 was dissolved in 37 ml of dry CH2C12 and cooled to 0 °C (ice). Subsequently, 22.3 ml (22.3 mmol, 6 eq.) Et2Zn 1.0 M in hexane (Aldrich) were added dropwise and stirred under Ar for 30 min at 0 °C. Then, 3.02 ml (37.2 mmol, 10 eq.) of CH2I2 were added dropwise over 15 min at the same temperature and stirred for further 2 h at 0 °C. Afterwards the cooling bath was removed and the mixture was stirred for additional 21 h at ambient temperature. TLC showed substantial amount of unreacted a- 17. It was diluted by addition of EtOAc and quenched with 50 mL of sat. aqueous NH4Cl. Extractive work-up provided 1.79 g of crude which was purified by chromatography on silica-gel giving 0.43 g (39%) of 18 and 0.49 g of mixture of compound 17 and 18 (approximately 20:80).
PATENT
WO2018193428
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2018193428
claiming a composition comprising an oligomeric compound having tricyclo-deoxyribonucleic acid (tc-DNA) nucleosides and a lipid moiety.
EXAMPLE 1
Inventive compositions for the treatment of Duchenne muscular dystrophy
Evaluation of efficacy
[00464] Adult mdx mice were treated weekly over 4 weeks with intravenous injections of different 13-mer AONs targeting the donor splice site of exon 23 of the dystrophin pre-mRNA (M23D: +2-11), namely with either SY-0308, SY-0210 and the inventive SY-0299, SY-0343, SY-0442 and SY-0455. SY-0308 (also named “tcDNA-PO M23D” interchangeably herein) corresponds to p-CCTCGGCTTACCT-OH of SEQ ID NO: l, with all nucleotides being tc-DNAs and all internucleosidic linkage groups being phosphorodiester linkage groups, and p being a phosphate moiety at the 5′ end. SY-0210 (also named “tcDNA-PS M23D” interchangeably herein) corresponds to p-CCTCGGCTTACCT-OH of SEQ ID NO: 1, with all nucleotides being tc-DNAs and all internucleosidic linkage groups being phosphorothioate linkage groups, and p being a phosphate moiety at the 5′ end. The inventive composition SY-0343 is herein interchangeably referred to as “Palm-2PS-tcDNA-PO M23D” which is depicted in the following:
[00465] The inventive composition SY-0442 is herein interchangeably referred to as “Palm-lPS-tcDNA-PO M23D” which is depicted in the following:
[00466] The inventive composition SY-0299 is herein interchangeably referred to as “Palm-2PO-tcDNA-PO M23D” which is depicted in the following:
//////////////////SYN-01, SYN 01, SYN01, preclinical , Duchenne muscular dystrophy, University of Bern,
LK-01, Apomorphine

LK-01
Leukos Biotech S.L.
APL-130277, H-001, Apokyn
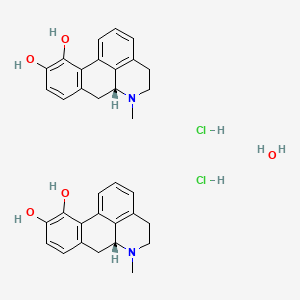
Apomorphine hydrochloride hemihydrate
CAS 41372-20-7
Leukos Biotech (following its spin-off from Jose Carreras Leukaemia Research Institute) is developing LK-01 , a solid form of apomorphine for the sc treatment of acute myeloid leukemia (AML) and the phase II trial results were expected later in 2019.
Apomorphine (brand names Apokyn, Ixense, Spontane, Uprima) is a type of aporphine having activity as a non-selective dopamine agonist which activates both D2-like and, to a much lesser extent, D1-like receptors.[1] It also acts as an antagonist of 5-HT2 and α-adrenergic receptors with high affinity. The compound is historically a morphine decomposition product made by boiling morphine with concentrated acid, hence the –morphine suffix. Contrary to its name, apomorphine does not actually contain morphine or its skeleton, nor does it bind to opioid receptors. The apo– prefix relates to it being a morphine derivative (“[comes] from morphine”).
Historically, apomorphine has been tried for a variety of uses, including as a way to relieve anxiety and craving in alcoholics, an emetic (to induce vomiting), for treating stereotypies (repeated behaviour) in farmyard animals, and more recently in treating erectile dysfunction. Currently, apomorphine is used in the treatment of Parkinson’s disease. It is a potent emetic and should not be administered without an antiemetic such as domperidone. The emetic properties of apomorphine are exploited in veterinary medicine to induce therapeutic emesis in canines that have recently ingested toxic or foreign substances.
Apomorphine was also used as a private treatment of heroin addiction, a purpose for which it was championed by the author William S. Burroughs. Burroughs and others claimed that it was a “metabolic regulator” with a restorative dimension to a damaged or dysfunctional dopaminergic system. There is more than enough anecdotal evidence to suggest that this offers a plausible route to an abstinence-based model; however, no clinical trials have ever tested this hypothesis. A recent study indicates that apomorphine might be a suitable marker for assessing central dopamine system alterations associated with chronic heroin consumption.[2] There is, however, no clinical evidence that apomorphine is an effective and safe treatment regimen for opiate addiction.[3]
Uses
Apomorphine is used in advanced Parkinson’s disease intermittent hypomobility (“off” episodes), where a decreased response to an anti-Parkinson drug such as L-DOPA causes muscle stiffness and loss of muscle control.[4][5] While apomorphine can be used in combination with L-DOPA, the intention is usually to reduce the L-DOPA dosing, as by this stage the patient often has many of dyskinesias caused by L-DOPA and hypermobility periods.[6][7] When an episode sets in, the apomorphine is injected subcutaneously, and signs subside. It is used an average of three times a day.[6] Some people use portable mini-pumps that continuously infuse them with apomorphine, allowing them to stay in the “on” state and using apomorphine as an effective monotherapy.[7][8]
Contraindications
The main and absolute contraindication to using apomorphine is the concurrent use of adrenergic receptor antagonists; combined, they cause a severe drop in blood pressure and fainting.[6][5] Alcohol causes an increased frequency of orthostatic hypotension (a sudden drop in blood pressure when getting up), and can also increase the chances of pneumonia and heart attacks.[6] Dopamine antagonists, by their nature of competing for sites at dopamine receptors, reduce the effectiveness of the agonistic apomorphine.[6][5]
IV administration of apomorphine is highly discouraged, as it can crystallize in the veins and create a blood clot (thrombus) and block a pulmonary artery (pulmonary embolism).[6][5]
Side effects
Nausea and vomiting are common side effects when first beginning therapy with apomorphine;[9] antiemetics such as trimethobenzamide or domperidone, dopamine antagonists,[10] are often used while first starting apomorphine. Around 50% of people grow tolerant enough to apomorphine’s emetic effects that they can discontinue the antiemetic.[5][6]
Other side effects include orthostatic hypotension and resultant fainting, sleepiness, dizziness, runny nose, sweating, paleness, and flushing. More serious side effects include dyskenesias (especially when taking L-DOPA), fluid accumulation in the limbs (edema), suddenly falling asleep, confusion and hallucinations, increased heart rate and heart palpitations, and persistent erections(priaprism).[5][6][11] The priaprism is caused by apomorphine increasing arterial blood supply to the penis. This side effect has been exploited in studies attempting to treat erectile dysfunction.[12]
Pharmacology
Mechanism of action
Apomorphine’s R-enantiomer is an agonist of both D1 and D2 dopamine receptors, with higher activity at D2.[6][10] The members of the D2 subfamily, consisting of D2, D3, and D4receptors, are inhibitory G protein–coupled receptors. The D4 receptor in particular is an important target in the signaling pathway, and is connected to several neurological disorders.[13] Shortage or excess of dopamine can prevent proper function and signaling of these receptors leading to disease states.[14]
Apomorphine improves motor function by activating dopamine receptors in the nigrostriatal pathway, the limbic system, the hypothalamus, and the pituitary gland.[15] It also increases blood flow to the supplementary motor area and to the dorsolateral prefrontal cortex (stimulation of which has been found to reduce the tardive dyskinesia effects of L-DOPA).[16][17]Parkinson’s has also been found to have excess iron at the sites of neurodegeneration; both the R- and S-enantiomers of apomorphine are potent iron chelators and radical scavengers.[10][18]
Apomorphine also reduces the breakdown of dopamine in the brain (though it inhibits its synthesis as well).[19][20] It is a powerful upregulator of certain neural growth factors,[21] in particular NGF and BDNF, epigenetic downregulation of which has been associated with addictive behaviour in rats.[22][23]
Apomorphine causes vomiting by acting on dopamine receptors in the chemoreceptor trigger zone of the medulla; this activates the nearby vomiting center.[15][20][24]
Pharmacokinetics
While apomorphine has lower bioavailability when taken orally, due to not being absorbed well in the GI tract and undergoing heavy first-pass metabolism,[18][8] it has a bioavailability of 100% when given subcutaneously.[6][15] It reaches peak plasma concentration in 10–60 minutes. Ten to twenty minutes after that, it reaches its peak concentration in the cerebrospinal fluid. Its lipophilic structure allows it to cross the blood–brain barrier.[6][15]
Apomorphine possesses affinity for the following receptors (note that a higher Ki indicates a lower affinity):[25][26][27]
| Receptor | Ki (nM) | Action |
|---|---|---|
| D1 | 484 | (partial) agonista |
| D2 | 52 | partial agonist (IA = 79% at D2S; 53% at D2L) |
| D3 | 26 | partial agonist (IA = 82%) |
| D4 | 4.37 | partial agonist (IA = 45%) |
| D5 | 188.9 | (partial) agonista |
| aThough its efficacies at D1 and D5 are unclear, it is known to act as an agonist at these sites.[28] | ||
| Receptor | Ki (nM) | Action |
|---|---|---|
| 5-HT1A | 2,523 | partial agonist |
| 5-HT1B | 2,951 | no action |
| 5-HT1D | 1,230 | no action |
| 5-HT2A | 120 | antagonist |
| 5-HT2B | 132 | antagonist |
| 5-HT2C | 102 | antagonist |
| Receptor | Ki (nM) | Action |
|---|---|---|
| α1A-adrenergic | 1,995 | antagonist |
| α1B-adrenergic | 676 | antagonist |
| α1D-adrenergic | 64.6 | antagonist |
| α2A-adrenergic | 141 | antagonist |
| α2B-adrenergic | 66.1 | antagonist |
| α2C-adrenergic | 36.3 | antagonist |
It has a Ki of over 10,000 nM (and thus negligible affinity) for β-adrenergic, H1, and mACh.[1]
Apomorphine has a high clearance rate (3–5 L/kg/hr) and is mainly metabolized and excreted by the liver.[15] It is likely that while the cytochrome P450 system plays a minor role, most of apomorphine’s metabolism happens via auto-oxidation, O-glucuronidation, O-methylation, N-demethylation, and sulfation.[6][15][20] Only 3–4% of the apomorphine is excreted unchanged and into the urine. The half-life is 30–60 minutes, and the effects of the injection last for up to 90 minutes.[6][7][15]
Toxicity depends on the route of administraion; the LD50s in mice were 300 mg/kg for the oral route, 160 mg/kg for intraperitoneal, and 56 mg/kg intravenous.[29]
Chemistry
Properties
Apomorphine has a catechol structure similar to that of dopamine.[19]
Synthesis
Several techniques exist for the creation of apomorphine from morphine. In the past, morphine had been combined with hydrochloric acid at high temperatures (around 150 °C) to achieve a low yield of apomorphine, ranging anywhere from 0.6% to 46%.[30]
More recent techniques create the apomorphine in a similar fashion, by heating it in the presence of any acid that will promote the essential dehydration rearrangement of morphine-type alkaloids, such as phosphoric acid. The method then deviates by including a water scavenger, which is essential to remove the water produced by the reaction that can react with the product and lead to decreased yield. The scavenger can be any reagent that will irreversibly react with water such as phthalic anhydride or titanium chloride. The temperature required for the reaction varies based upon choice of acid and water scavenger. The yield of this reaction is much higher: at least 55%.[30]
Conversion of Morphine (I) to Apomorphine (II) in the presence of acid following the example of the morphine skeleton dehydration rearrangement, outlined by Bentley.[31]
History
The pharmacological effects of the naturally-occurring analog aporphine in the blue lotus (N. caerulea)[32] were known to the ancient Egyptians and Mayans,[33] with the plant featuring in tomb frescoes and associated with entheogenic rites. It is also observed in Egyptian erotic cartoons, suggesting that they were aware of its erectogenic properties.
The modern medical history of apomorphine begins with its synthesis by Arppe in 1845[34] from morphine and sulfuric acid, although it was named sulphomorphide at first. Matthiesen and Wright (1869) used hydrochloric acid instead of sulfuric acid in the process, naming the resulting compound apomorphine. Initial interest in the compound was as an emetic, tested and confirmed safe by London doctor Samuel Gee,[35] and for the treatment of stereotypies in farmyard animals.[36] Key to the use of apomorphine as a behavioural modifier was the research of Erich Harnack, whose experiments in rabbits (which do not vomit) demonstrated that apomorphine had powerful effects on the activity of rabbits, inducing licking, gnawing and in very high doses convulsions and death.
Treatment of alcoholism
Apomorphine was one of the earliest used pharmacotherapies for alcoholism. The Keeley Cure (1870s to 1900) contained apomorphine, among other ingredients, but the first medical reports of its use for more than pure emesis come from James Tompkins[37] and Charles Douglas.[38][39] Tompkins reported, after injection of 6.5 mg (“one tenth of a grain”):
In four minutes free emesis followed, rigidity gave way to relaxation, excitement to somnolence, and without further medication the patient, who before had been wild and delirious, went off into a quiet sleep.
Douglas saw two purposes for apomorphine:
[it can be used to treat] a paroxysm of dipsomania [an episode of intense alcoholic craving]… in minute doses it is much more rapidly efficient in stilling the dipsomaniac craving than strychnine or atropine… Four or even 3m [minim – roughly 60 microlitres] of the solution usually checks for some hours the incessant demands of the patient… when he awakes from the apomorphine sleep he may still be demanding alcohol, though he is never then so insistent as before. Accordingly it may be necessary to repeat the dose, and even to continue to give it twice or three times a day. Such repeated doses, however, do not require to be so large: 4 or even 3m is usually sufficient.
This use of small, continuous doses (1/30th of a grain, or 2.16 mg by Douglas) of apomorphine to reduce alcoholic craving comes some time before Pavlov‘s discovery and publication of the idea of the “conditioned reflex” in 1903. This method was not limited to Douglas; the Irish doctor Francis Hare, who worked in a sanatorium outside London from 1905 onwards, also used low-dose apomorphine as a treatment, describing it as “the most useful single drug in the therapeutics of inebriety”.[40] He wrote:
In (the) sanatorium it is used in three different sets of circumstances: (1) in maniacal or hysterical drunkenness: (2) during the paroxysm of dipsomania, in order to still the craving for alcohol; and (3) in essential insomnia of a special variety… [after giving apomorphine] the patient’s mental condition is entirely altered. He may be sober: he is free from the time being from any craving from alcohol. The craving may return, however, and then it is necessary to repeat the injection, it may be several times at intervals of a few hours. These succeeding injections should be quite small, 3 to 6 min. being sufficient. Doses of this size are rarely emetic. There is little facial pallor, a sensation as of the commencement of sea-sickness, perhaps a slight malaise with a sudden subsidence of the craving for alcohol, followed by a light and short doze.
He also noted there appeared to be a significant prejudice against the use of apomorphine, both from the associations of its name and doctors being reluctant to give hypodermic injections to alcoholics. In the US, the Harrison Narcotics Tax Act made working with any morphine derivatives extremely hard, despite apomorphine itself not being an opiate.
In the 1950s the neurotransmitter dopamine was discovered in the brain by Kathleen Montagu, and characterised as a neurotransmitter a year later by Arvid Carlsson, for which he would be awarded the Nobel Prize.[41] A. N. Ernst then discovered in 1965 that apomorphine was a powerful stimulant of dopamine receptors.[42] This, along with the use of sublingual apomorphine tablets, led to a renewed interest in the use of apomorphine as a treatment for alcoholism. A series of studies of non-emetic apomorphine in the treatment of alcoholism were published, with mostly positive results.[43][44][45][46][47] However, there was little clinical consequence.
Parkinson’s disease
The use of apomorphine to treat “the shakes” was first suggested by Weil in France in 1884,[48] although seemingly not pursued until 1951.[49] Its clinical use was first reported in 1970 by Cotzias et al.,[50] although its emetic properties and short half-life made oral use impractical. A later study found that combining the drug with the antiemetic domperidoneimproved results significantly.[51] The commercialization of apomorphine for Parkinson’s disease followed its successful use in patients with refractory motor fluctuations using intermittent rescue injections and continuous infusions.[52]
Aversion therapy
Aversion therapy in alcoholism had its roots in Russia in the early 1930s,[53] with early papers by Pavlov, Galant and Sluchevsky and Friken,[54] and would remain a strain in the Soviet treatment of alcoholism well into the 1980s. In the US a particularly notable devotee was Dr Voegtlin,[55] who attempted aversion therapy using apomorphine in the mid to late 1930s. However, he found apomorphine less able to induce negative feelings in his subjects than the stronger and more unpleasant emetic emetine.
In the UK, however, the publication of J Y Dent’s (who later went on to treat Burroughs) 1934 paper “Apomorphine in the treatment of Anxiety States”[56] laid out the main method by which apomorphine would be used to treat alcoholism in Britain. His method in that paper is clearly influenced by the then-novel idea of aversion:
He is given his favourite drink, and his favourite brand of that drink… He takes it stronger than is usual to him… The small dose of apomorphine, one-twentieth of a grain [3.24mg], is now given subcutaneously into his thigh, and he is told that he will be sick in a quarter of an hour. A glass of whisky and water and a bottle of whisky are left by his bedside. At six o’clock (four hours later) he is again visited and the same treatment is again administered… The nurse is told in confidence that if he does not drink, one-fortieth [1.62mg] of a grain of apomorphine should be injected during the night at nine o’clock, one o’clock, and five o’clock, but that if he drinks the injection should be given soon after the drink and may be increased to two hourly intervals. In the morning at about ten he is again given one or two glasses of whisky and water… and again one-twentieth of a grain [3.24mg] of apomorphine is injected… The next day he is allowed to eat what he likes, he may drink as much tea as he likes… He will be strong enough to get up and two days later he leaves the home.
However, even in 1934 he was suspicious of the idea that the treatment was pure conditioned reflex – “though vomiting is one of the ways that apomorphine relives the patient, I do not believe it to be its main therapeutic effect.” – and by 1948 he wrote:[3]
It is now twenty-five years since I began treating cases of anxiety and alcoholism with apomorphine, and I read my first paper before this Society fourteen years ago. Up till then I had thought, and, unfortunately, I said in my paper, that the virtue of the treatment lay in the conditioned reflex of aversion produced in the patient. This statement is not even a half truth… I have been forced to the conclusion that apomorphine has some further action than the production of a vomit.
This led to his development of lower-dose and non-aversive methods, which would inspire a positive trial of his method in Switzerland by Dr Harry Feldmann[57] and later scientific testing in the 1970s, some time after his death. However, the use of apomorphine in aversion therapy had escaped alcoholism, with its use to treat homosexuality leading to the death of a British Army Captain Billy Clegg HIll in 1962,[58] helping to cement its reputation as a dangerous drug used primarily in archaic behavioural therapies.
Opioid addiction
In his Deposition: Testimony Concerning a Sickness in the introduction to later editions of Naked Lunch (first published in 1959), William S. Burroughs wrote that apomorphine treatment was the only effective cure to opioid addiction he has encountered:
The apomorphine cure is qualitatively different from other methods of cure. I have tried them all. Short reduction, slow reduction, cortisone, antihistamines, tranquilizers, sleeping cures, tolserol, reserpine. None of these cures lasted beyond the first opportunity to relapse. I can say that I was never metabolically cured until I took the apomorphine cure… The doctor, John Yerbury Dent, explained to me that apomorphine acts on the back brain to regulate the metabolism and normalize the blood stream in such a way that the enzyme stream of addiction is destroyed over a period of four to five days. Once the back brain is regulated apomorphine can be discontinued and only used in case of relapse.
He goes on to lament the fact that as of his writing, little to no research has been done on apomorphine or variations of the drug to study its effects on curing addiction, and perhaps the possibility of retaining the positive effects while removing the side effect of vomiting.
Despite his claims throughout his life, Burroughs never really cured his addiction and was back to using opiates within years of his apomorphine “cure”.[59] However, he insisted on apomorphine’s effectiveness in several works and interviews.[citation needed]
Society and culture
- Apomorphine has a vital part in Agatha Christie‘s detective story Sad Cypress.
- The 1965 Tuli Kupferberg song “Hallucination Horrors” recommends apomorphine at the end of each verse as a cure for hallucinations brought on by a humorous variety of intoxicants; the song was recorded by The Fugs and appears on the album Virgin Fugs.
Research
There is renewed interest in the use of apomorphine to treat addiction, in both smoking cessation[60] and alcoholism.[61] As the drug is old, out of patent, and safe for use in humans, it is a viable target for repurposing.
Apomorphine has been researched as a possible treatment for erectile dysfunction and female hypoactive sexual desire disorder, though the arousal effects were found not to be reliable enough. One large study found that only 39.4% got erections (compared to baseline 13.1); another found that apomorphine was successful 45–51% of the time, but the placebo also worked 36% of the time.[12][62] Nonetheless, it was under development as a treatment for erectile dysfunction by TAP Pharmaceuticals under the brand name Uprima. In 2000, TAP withdrew its new drug application after an FDA review panel raised questions about the drug’s safety, due to many clinical trial subjects fainting after taking the drug.[63]
Alzheimer’s disease
Apomorphine is reported to be an inhibitor of amyloid beta protein (Aβ) fiber formation, whose presence is a hallmark of Alzheimer’s disease (AD), and a potential therapeutic under the amyloid hypothesis.[64] While it promotes oligomerization of the Aβ40 group of molecules, it inhibits more advanced fibril formation; this is thought to be due to the autoxidation that occurs at the hydroxyl groups. Once this functional group was altered, the inhibitory effect could be seen to decrease, reducing either the indirect or direct interference of the fibril formation.[64]
The protective effects of apomorphine were tested in mouse models with mutations in genes related to AD, such as the amyloid precursor protein gene. Apomorphine was seen to significantly improve memory function through the increased successful completion of the Morris Water Maze. The levels of the aberrant proteins that lead to neuronal disruption were also tested in the brains of mice. Treatment was seen to decrease the intraneuronal levels of the more aggressive Aβ42 molecule when compared to the control mice. This result is consistent with the finding that another protein linked to AD, tau protein, was seen to decrease with apomorphine treatment.[65]
Veterinary use
Apomorphine is used to inducing vomiting in dogs the after ingestion of various toxins or foreign bodies. It can be given subcutaneously, intramuscularly, intravenously, or, when a tablet is crushed, in the conjunctiva of the eye.[66][67] The oral route is ineffective, as apomorphine cannot cross the blood–brain barrier fast enough, and blood levels don’t reach a high enough concentration to stimulate the chemoreceptor trigger zone.[66] It can remove around 40–60% of the contents in the stomach.[68]
One of the reasons apomorphine is a preferred drug is its reversibility:[69] in cases of prolonged vomiting, the apomorphine can be reversed with dopamine antagonists like the phenothiazines (for example, acepromazine). Giving apomorphine after giving acepromazine, however, will no longer stimulate vomiting, because apomorphine’s target receptors are already occupied.[66] An animal who undergoes severe respiratory depression due to apomorphine can be treated with naloxone.[66][67]
Apomorphine does not work in cats, who have too few dopamine receptors.[66]
PATENT
WO-2019141673
Novel crystalline forms of apomorphine or its palmitate or hydrochloride salt useful treating acute myeloid leukemia, Parkinson’s disease, sexual dysfunction and solid tumors. Also claims the process for preparing apomorphine palmitic acid cocrystal salt.
Apomorphine (APO) is a commercial available medical drug with the chemical formula C-17H-17NO2 and structure:
Apomorphine (APO) has been described for treatment of different medical indications – for instance:
– WO2015/197839A1 : leukemia such as acute myeloid leukemia (AML);
– WO2016/103262A2: Parkinson’s disease;
– WO02/39879A2: sexual dysfunction in a patient taking antidepressant medication;
– W02004/082630A2: neurological function of an individual who has a brain injury.
Apomorphine hydrochloride (HCI) is a salt present in commercially available medical products (e.g. APO-Go® PFS or Apokyn®).
A common side effect of administering apomorphine hydrochloride by e.g. subcutaneous injection is e.g. the development of subcutaneous nodules at the injection site, which can become infected, necessitating treatment or surgical involvement.
In relation to this problem – above discussed WO2016/103262A2 describes an alternative solid form of apomorphine, which is e.g. an alcohol solvate crystal of apomorphine free base, wherein the solvate forming solvent is (C-|-C8) alkanol, preferably isopropanol (IPA – i.e. a solid crystalline form of apomorphine-IPA.
Palmitic acid (hexadecanoic acid in IUPAC nomenclature) is a fatty acid found with the chemical formula CH3(CH2)14COOH.
Palmitate is the salt and ester of palmitic acid.
A herein relevant synonyms name may e.g. be palmitoate.
Beside apomorphine hydrochloride, above discussed WO2015/197839A1 and W02004/082630A2 provide a list of other possible suitable pharmaceutically acceptable salts – palmitic acid (or synonyms like palmitate or palmitoate) is not mentioned in the lists of these two WO documents.
As discussed in the review article of Schultheiss et al. (“Pharmaceutical Cocrystals and Their Physicochemical Properties”; Crystal Growth & Design, Vol. 9, No. 6, 2009, p. 2950-2967) – solid-state chemists call upon a variety of different strategies when attempting to alter the chemical and physical solid-state properties of active pharmaceutical ingredients (APIs), namely, the formation of salts, polymorphs, hydrates, solvates, and cocrystals.
Salt formation is one of the primary solid-state approaches used to modify the physical properties of APIs, and it is estimated that over half of the medicines on the market are administered as salts. However, a limitation within this approach is that the API must possess a suitable (basic or acidic)
ionizable site. In comparison, cocrystals (multicomponent assemblies held together by freely reversible, noncovalent interactions) offer a different pathway, where any API regardless of acidic, basic, or ionizable groups, could potentially be cocrystallized.
Above discussed WO02/39879A2 also provides a long list of suitable pharmaceutically acceptable salts and mentions palmitoate (see page 5, line 16).
However, in all herein relevant experimental work of this WO document was used apomorphine hydrochloride and a palmitic acid based salt is simply mentioned in a list – i.e. a palmitic acid based salt is not a preferred salt.
Alternatively expressed, by reading this WO document the skilled person has in practice no motivation to use any other solid form than apomorphine-HCI – one reason for this is that apomor-phine-HCI is used in all herein relevant experimental work of this WO document.
PATENT
WO2018130685
claiming synergistic combination comprising antimetabolite antineoplastic agent (eg cytarabine ) and type 1 serotonin receptor antagonist (5-HTR1) (eg apomorphine ), useful for treating cancer.
SYN
SYN

PAPER
- Small, L. et al.: J. Org. Chem. (JOCEAH) 5, 334 (1940)
References
- ^ Jump up to:a b Millan MJ, Maiofiss L, Cussac D, Audinot V, Boutin JA, Newman-Tancredi A (November 2002). “Differential actions of antiparkinson agents at multiple classes of monoaminergic receptor. I. A multivariate analysis of the binding profiles of 14 drugs at 21 native and cloned human receptor subtypes”. The Journal of Pharmacology and Experimental Therapeutics. 303 (2): 791–804. doi:10.1124/jpet.102.039867. PMID 12388666.
- ^ Guardia J, Casas M, Prat G, Trujols J, Segura L, Sánchez-Turet M (October 2002). “The apomorphine test: a biological marker for heroin dependence disorder?”. Addiction Biology. 7 (4): 421–6. doi:10.1080/1355621021000006206. PMID 14578019.
- ^ Jump up to:a b Dent JY (1949). “Apomorphine Treatment of Addiction”. British Journal of Addiction to Alcohol & Other Drugs. 46 (1): 15–28. doi:10.1111/j.1360-0443.1949.tb04502.x.
- ^ “Apomorphine Uses, Side Effects & Warnings”. Drugs.com. Retrieved 27 February2018.
- ^ Jump up to:a b c d e f Clayton BD, Willihnganz M (2016). Basic Pharmacology for Nurses – E-Book. Elsevier Health Sciences. pp. 210–211. ISBN 978-0-323-37697-6.
- ^ Jump up to:a b c d e f g h i j k l m “Apomorphine Hydrochloride Monograph for Professionals”. Drugs.com. Retrieved 26 February 2018.
- ^ Jump up to:a b c Chaudhuri KR, Clough C (February 1998). “Subcutaneous apomorphine in Parkinson’s disease”. BMJ. 316 (7132): 641. doi:10.1136/bmj.316.7132.641. PMC 1112674. PMID 9522772.
- ^ Jump up to:a b Schapira AH, Olanow CW (2005). Principles of Treatment in Parkinson’s Disease(illustrated ed.). Elsevier Health Sciences. p. 35. ISBN 978-0-7506-5428-9.
- ^ Dressler D (June 2005). “[Apomorphine in the treatment of Parkinson’s Disease]”. Der Nervenarzt (in German). 76 (6): 681–9. doi:10.1007/s00115-004-1830-4. PMID 15592807.
- ^ Jump up to:a b c Youdim MB, Gassen M, Gross A, Mandel S, Grünblatt E (2000). “Iron chelating, antioxidant and cytoprotective properties of dopamine receptor agonist; apomorphine”. In Mizuno Y, Calne D, Horowski R, Poewe W, Riederer P, Youdim M (eds.). Advances in Research on Neurodegeneration. 7 (illustrated ed.). Springer Science & Business Media. pp. 83–96. ISBN 978-3-211-83485-5.
- ^ “Apomorphine”. Medline Plus. US National Library of Medicine. 15 June 2017. Retrieved 26 February 2018.
- ^ Jump up to:a b Porst H, Buvat J (2008). Standard Practice in Sexual Medicine. John Wiley & Sons. p. 77. ISBN 978-1-4051-7872-3.
- ^ Ptácek R, Kuzelová H, Stefano GB (September 2011). “Dopamine D4 receptor gene DRD4 and its association with psychiatric disorders”. Medical Science Monitor. 17 (9): RA215–20. doi:10.12659/MSM.881925. PMC 3560519. PMID 21873960.
- ^ Stacy M, Silver D (2008). “Apomorphine for the acute treatment of “off” episodes in Parkinson’s disease”. Parkinsonism & Related Disorders. 14 (2): 85–92. doi:10.1016/j.parkreldis.2007.07.016. PMID 18083605.
- ^ Jump up to:a b c d e f g U.S. National Library of Medicine. “Apomorphine”. PubChem. Retrieved 26 February 2018.
- ^ Lewitt P, Oertel WH (1999). Parkinsons’s Disease: The Treatment Options. CRC Press. p. 22. ISBN 978-1-85317-379-0.
- ^ Rektorova I, Sedlackova S, Telecka S, Hlubocky A, Rektor I (2008). “Dorsolateral prefrontal cortex: a possible target for modulating dyskinesias in Parkinson’s disease by repetitive transcranial magnetic stimulation”. International Journal of Biomedical Imaging. 2008: 372125. doi:10.1155/2008/372125. PMC 2233877. PMID 18274665.
- ^ Jump up to:a b Galvez-Jimenez N (2013). Scientific Basis for the Treatment of Parkinson’s Disease, Second Edition. CRC Press. p. 195. ISBN 978-0-203-33776-9.
- ^ Jump up to:a b Iversen L (2012). Biogenic Amine Receptors. Springer Science & Business Media. p. 238. ISBN 978-1-4684-8514-1.
- ^ Jump up to:a b c Advances in Pharmacology and Chemotherapy, Volume 15. Silvio Garattini, A. Goldin, F. Hawking, Irwin J. Kopin. Academic Press. 1978. pp. 27, 93, 96. ISBN 978-0-08-058106-4.
- ^ Ohta M, Mizuta I, Ohta K, Nishimura M, Mizuta E, Hayashi K, Kuno S (May 2000). “Apomorphine up-regulates NGF and GDNF synthesis in cultured mouse astrocytes”. Biochemical and Biophysical Research Communications. 272 (1): 18–22. doi:10.1006/bbrc.2000.2732. PMID 10872797.
- ^ McGeary JE, Gurel V, Knopik VS, Spaulding J, McMichael J (October 2011). “Effects of nerve growth factor (NGF), fluoxetine, and amitriptyline on gene expression profiles in rat brain”. Neuropeptides. 45 (5): 317–22. doi:10.1016/j.npep.2011.06.002. PMID 21820738.
- ^ Heberlein A, Muschler M, Frieling H, Behr M, Eberlein C, Wilhelm J, Gröschl M, Kornhuber J, Bleich S, Hillemacher T (May 2013). “Epigenetic down regulation of nerve growth factor during alcohol withdrawal”. Addiction Biology. 18 (3): 508–10. doi:10.1111/j.1369-1600.2010.00307.x. PMID 21392176.
- ^ Riviere JE, Papich MG (2009). Veterinary Pharmacology and Therapeutics. John Wiley & Sons. p. 318. ISBN 978-0-8138-2061-3.
- ^ Roth BL, Driscol J (12 January 2011). “PDSP Ki Database”. Psychoactive Drug Screening Program (PDSP). University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Retrieved 1 July 2014. Note: Values for humans are used. If there is more than one value listed for humans, their average is used.
- ^ Newman-Tancredi A, Cussac D, Audinot V, Nicolas JP, De Ceuninck F, Boutin JA, Millan MJ (November 2002). “Differential actions of antiparkinson agents at multiple classes of monoaminergic receptor. II. Agonist and antagonist properties at subtypes of dopamine D(2)-like receptor and alpha(1)/alpha(2)-adrenoceptor”. The Journal of Pharmacology and Experimental Therapeutics. 303 (2): 805–14. doi:10.1124/jpet.102.039875. PMID 12388667.
- ^ Newman-Tancredi A, Cussac D, Quentric Y, Touzard M, Verrièle L, Carpentier N, Millan MJ (November 2002). “Differential actions of antiparkinson agents at multiple classes of monoaminergic receptor. III. Agonist and antagonist properties at serotonin, 5-HT(1) and 5-HT(2), receptor subtypes”. The Journal of Pharmacology and Experimental Therapeutics. 303 (2): 815–22. doi:10.1124/jpet.102.039883. PMID 12388668.
- ^ Hsieh GC, Hollingsworth PR, Martino B, Chang R, Terranova MA, O’Neill AB, Lynch JJ, Moreland RB, Donnelly-Roberts DL, Kolasa T, Mikusa JP, McVey JM, Marsh KC, Sullivan JP, Brioni JD (January 2004). “Central mechanisms regulating penile erection in conscious rats: the dopaminergic systems related to the proerectile effect of apomorphine”. The Journal of Pharmacology and Experimental Therapeutics. 308 (1): 330–8. doi:10.1124/jpet.103.057455. PMID 14569075.
- ^ Lewis, R.J. Sr. (2004). Sax’s Dangerous Properties of Industrial Materials (11 ed.). Wiley, John & Sons, Incorporated. p. 287. ISBN 978-0471476627.
- ^ Jump up to:a b Narayanasamy Gurusamy. “Process for making apomorphine and apocodeine”.
- ^ Bentley, K.W. (2014-04-24). The Isoquinoline Alkaloids: A Course in Organic Chemistry. Elsevier, 2014. pp. 118–120. ISBN 978-1483152233.
- ^ Poklis JL, Mulder HA, Halquist MS, Wolf CE, Poklis A, Peace MR (July 2017). “The Blue Lotus Flower (Nymphea caerulea) Resin Used in a New Type of Electronic Cigarette, the Re-Buildable Dripping Atomizer”. Journal of Psychoactive Drugs. 49 (3): 175–181. doi:10.1080/02791072.2017.1290304. PMC 5638439. PMID 28266899.
- ^ Bertol E, Fineschi V, Karch SB, Mari F, Riezzo I (February 2004). “Nymphaea cults in ancient Egypt and the New World: a lesson in empirical pharmacology”. Journal of the Royal Society of Medicine. 97 (2): 84–5. doi:10.1177/014107680409700214. PMC 1079300. PMID 14749409.
- ^ Taba P, Lees A, Stern G (2013). “Erich Harnack (1852-1915) and a short history of apomorphine”. European Neurology. 69 (6): 321–4. doi:10.1159/000346762. PMID 23549143.
- ^ Gee S (1869). “On the action of a new organic base, apomorphia”. Transactions of the Clinical Society of London. 2: 166–169.
- ^ Feser J (1873). “Die in neuester Zeit in Anwendung gekommen Arzneimittel: 1. Apomorphinum hydrochloratum”. Z Prakt Veterinairwiss: 302–306.
- ^ Tompkins J (1899). “Apomorphine in Acute Alcoholic Delirium”. Medical Record.
- ^ “Apomorphine as a hypnotic”. The Lancet. 155 (3998): 1083. 1900. doi:10.1016/s0140-6736(01)70565-x.
- ^ Douglas CJ (1899). “The withdrawal of alcohol in delirium tremens”. The New York Medical Journal: 626.
- ^ Hare, Francis (1912). On alcoholism; its clinical aspects and treatment. London: Churchill.
- ^ Benes FM (January 2001). “Carlsson and the discovery of dopamine”. Trends in Pharmacological Sciences. 22 (1): 46–7. doi:10.1016/S0165-6147(00)01607-2. PMID 11165672.
- ^ Ernst AM (May 1965). “Relation between the action of dopamine and apomorphine and their O-methylated derivatives upon the CNS”. Psychopharmacologia. 7 (6): 391–9. doi:10.1007/BF00402361. PMID 5831877.
- ^ Moynihan NH (1965). “The Treatment of Alcoholism in General Practice”. Practitioner: 223–7.
- ^ Carlsson C, Johansson PR, Gullberg B (May 1977). “A double-blind cross-over study: apomorphine/placebo in chronic alcoholics”. International Journal of Clinical Pharmacology and Biopharmacy. 15 (5): 211–3. PMID 326687.
- ^ Halvorsen KA, Martensen-Larsen O (April 1978). “Apomorphine revived: fortified, prolonged, and improved therapeutical effect”. The International Journal of the Addictions. 13 (3): 475–84. doi:10.3109/10826087809045262. PMID 352969.
- ^ Jensen SB, Christoffersen CB, Noerregaard A (December 1977). “Apomorphine in outpatient treatment of alcohol intoxication and abstinence: a double-blind study”. The British Journal of Addiction to Alcohol and Other Drugs. 72 (4): 325–30. doi:10.1111/j.1360-0443.1977.tb00699.x. PMID 341937.
- ^ Schlatter EK, Lal S (June 1972). “Treatment of alcoholism with Dent’s oral apomorphine method”. Quarterly Journal of Studies on Alcohol. 33 (2): 430–6. PMID 5033142.
- ^ Weil E (1884). “De l’apomorphine dans certain troubles nerveux”. Lyon Med (in French). 48: 411–419.
- ^ Schwab RS, Amador LV, Lettvin JY (1951). “Apomorphine in Parkinson’s disease”. Transactions of the American Neurological Association. 56: 251–3. PMID 14913646.
- ^ Cotzias GC, Papavasiliou PS, Fehling C, Kaufman B, Mena I (January 1970). “Similarities between neurologic effects of L-dopa and of apomorphine”. The New England Journal of Medicine. 282 (1): 31–3. doi:10.1056/NEJM197001012820107. PMID 4901383.
- ^ Corsini GU, Del Zompo M, Gessa GL, Mangoni A (May 1979). “Therapeutic efficacy of apomorphine combined with an extracerebral inhibitor of dopamine receptors in Parkinson’s disease”. Lancet. 1 (8123): 954–6. doi:10.1016/S0140-6736(79)91725-2. PMID 87620.
- ^ Stibe CM, Kempster P, Lees AJ & Stern GM (1988). “Subcutaneous apomorphine in parkinsonian on-off oscillations”. Lancet. 331 (8582): 403–406. doi:10.1016/S0140-6736(88)91193-2.
- ^ Ban TA (2008). Conditioning behavior and psychiatry. New Brunswick [N.J.]: AldineTransaction. ISBN 978-0-202-36235-9. OCLC 191318001.
- ^ Raikhel EA (2016). Governing habits : treating alcoholism in the post-Soviet clinic. Ithaca. ISBN 9781501703133. OCLC 965905763.
- ^ Lemere F, Voegtlin WL (June 1950). “An evaluation of the aversion treatment of alcoholism”. Quarterly Journal of Studies on Alcohol. 11 (2): 199–204. PMID 15424345.
- ^ Dent JY (1934-10-01). “Apomorphine in the Treatment of Anxiety States, with Especial Reference to Alcoholism*”. British Journal of Inebriety. 32 (2): 65–88. doi:10.1111/j.1360-0443.1934.tb05016.x. ISSN 1360-0443.
- ^ De Morsier G, Feldmann H (1952). “[Apomorphine therapy of alcoholism; report of 500 cases]”. Schweizer Archiv für Neurologie und Psychiatrie. Archives Suisses de Neurologie et de Psychiatrie. Archivio Svizzero di Neurologia e Psichiatria. 70 (2): 434–40. PMID 13075975.
- ^ “Gay injustice ‘was widespread‘“. 2009-09-12. Retrieved 2018-01-24.
- ^ Birmingham, Jed (2 November 2009). “William Burroughs and the History of Heroin”. RealityStudio.
- ^ Morales-Rosado JA, Cousin MA, Ebbert JO, Klee EW (December 2015). “A Critical Review of Repurposing Apomorphine for Smoking Cessation”. Assay and Drug Development Technologies. 13 (10): 612–22. doi:10.1089/adt.2015.680. PMID 26690764.
- ^ “Apomorphine – A forgotten treatment for alcoholism”. apomorphine.info. Retrieved 2018-01-24.
- ^ IsHak, Waguih William (2017). The Textbook of Clinical Sexual Medicine. Springer. p. 388. ISBN 978-3-319-52539-6.
- ^ “Abbott Withdraws Application for an Impotence Pill”. Bloomberg News via The New York Times. 1 July 2000.
- ^ Jump up to:a b Lashuel HA, Hartley DM, Balakhaneh D, Aggarwal A, Teichberg S, Callaway DJ (November 2002). “New class of inhibitors of amyloid-beta fibril formation. Implications for the mechanism of pathogenesis in Alzheimer’s disease”. The Journal of Biological Chemistry. 277 (45): 42881–90. doi:10.1074/jbc.M206593200. PMID 12167652.
- ^ Himeno E, Ohyagi Y, Ma L, Nakamura N, Miyoshi K, Sakae N, Motomura K, Soejima N, Yamasaki R, Hashimoto T, Tabira T, LaFerla FM, Kira J (February 2011). “Apomorphine treatment in Alzheimer mice promoting amyloid-β degradation” (PDF). Annals of Neurology. 69 (2): 248–56. doi:10.1002/ana.22319. PMID 21387370.
- ^ Jump up to:a b c d e Bill RL (2016). Clinical Pharmacology and Therapeutics for Veterinary Technicians – E-Book. Elsevier Health Sciences. p. 94. ISBN 978-0-323-44402-6.
- ^ Jump up to:a b Khan SN, Hooser SB (2012). Common Toxicologic Issues in Small Animals, an Issue of Veterinary Clinics: Small Animal Practice – E-Book. Elsevier Health Sciences. p. 310. ISBN 978-1-4557-4325-4.
- ^ Plumb, Donald C. (2011). “Apomorphine”. Plumb’s Veterinary Drug Handbook (7th ed.). Stockholm, Wisconsin: Wiley. pp. 77–79. ISBN 978-0-470-95964-0.
- ^ Peterson ME, Talcott PA (2006). Small Animal Toxicology. Elsevier Health Sciences. p. 131. ISBN 978-0-7216-0639-2.
 |
|
 |
|
| Clinical data | |
|---|---|
| Trade names | Apokyn |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a604020 |
| Pregnancy category |
|
| Routes of administration |
SQ |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 100% following injection |
| Protein binding | ~50% |
| Metabolism | Hepatic, phase II |
| Onset of action | 10–20 min |
| Elimination half-life | 40 minutes |
| Duration of action | 60–90 min |
| Excretion | Hepatic |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.000.327 |
| Chemical and physical data | |
| Formula | C17H17NO2 |
| Molar mass | 267.322 g/mol g·mol−1 |
| 3D model (JSmol) | |
Apomorphine
-
- ATC:N04BC07
- Use:emetic, erectile dysfunction
- Chemical name:(R)-5,6,6a,7-tetrahydro-6-methyl-4H-dibenzo[de,g]quinoline-10,11-diol
- Formula:C17H17NO2
- MW:267.33 g/mol
- CAS-RN:58-00-4
- InChI Key:VMWNQDUVQKEIOC-UHFFFAOYSA-N
- InChI:InChI=1S/C17H17NO2/c1-18-8-7-10-3-2-4-12-15(10)13(18)9-11-5-6-14(19)17(20)16(11)12/h2-6,13,19-20H,7-9H2,1H3
- EINECS:200-360-0
- LD50:56 mg/kg (M, i.v.); >100 mg/kg (M, p.o.)
///////////// LK-01, LK 01 , LK01, Apomorphine
CN1CCC2=C3C1CC4=C(C3=CC=C2)C(=C(C=C4)O)O.CN1CCC2=C3C1CC4=C(C3=CC=C2)C(=C(C=C4)O)O.O.Cl.Cl
SY-008


SY-008
CAS 1878218-66-6
FREE FORM 1480443-32-0
SGLT1 inhibitor (type 2 diabetes),
β-D-Glucopyranoside, 4-[[4-[(1E)-4-(2,9-diazaspiro[5.5]undec-2-yl)-1-buten-1-yl]-2-methylphenyl]methyl]-5-(1-methylethyl)-1H-pyrazol-3-yl, acetate (1:1)
acetic acid;(2S,3R,4S,5S,6R)-2-[[4-[[4-[(E)-4-(2,9-diazaspiro[5.5]undecan-2-yl)but-1-enyl]-2-methylphenyl]methyl]-5-propan-2-yl-1H-pyrazol-3-yl]oxy]-6-(hydroxymethyl)oxane-3,4,5-triol
MF H50 N4 O6 . C2 H4 O2
MW 58.8 g/mol,C35H54N4O8
Originator Eli Lilly
- Developer Eli Lilly; Yabao Pharmaceutical Group
- Class Antihyperglycaemics; Small molecules
- Mechanism of Action Sodium-glucose transporter 1 inhibitors
- Phase I Diabetes mellitus
- 28 Aug 2018 No recent reports of development identified for phase-I development in Diabetes-mellitus in Singapore (PO)
- 24 Jun 2018 Biomarkers information updated
- 12 Mar 2018 Phase-I clinical trials in Diabetes mellitus (In volunteers) in China (PO) (NCT03462589)
-
Eli Lilly is developing SY 008, a sodium glucose transporter 1 (SGLT1) inhibitor, for the treatment of diabetes mellitus. The approach of inhibiting SGLT1 could be promising because it acts independently of the beta cell and could be effective in both early and advanced stages of diabetes. Reducing both glucose and insulin may improve the metabolic state and potentially the health of beta cells, without causing weight gain or hypoglycaemia. Clinical development is underway in Singapore and China.
As at August 2018, no recent reports of development had been identified for phase-I development in Diabetes-mellitus in Singapore (PO).
Suzhou Yabao , under license from Eli Lilly , is developing SY-008 , an SGLT1 inhibitor, for the potential oral capsule treatment of type 2 diabetes in China. By April 2019, a phase Ia trial was completed
PATENT
WO 2013169546
The present invention is in the field of treatment of diabetes and other diseases and disorders associated with hyperglycemia. Diabetes is a group of diseases that is characterized by high levels of blood glucose. It affects approximately 25 million people in the United States and is also the 7th leading cause of death in U.S. according to the 201 1 National Diabetes Fact Sheet (U.S. Department of Health and Human Services, Centers for Disease Control and Prevention). Sodium-coupled glucose cotransporters (SGLT’s) are one of the transporters known to be responsible for the absorption of carbohydrates, such as glucose. More specifically, SGLTl is responsible for transport of glucose across the brush border membrane of the small intestine. Inhibition of SGLTl may result in reduced absorption of glucose in the small intestine, thus providing a useful approach to treating diabetes.
U.S. Patent No. 7,655,632 discloses certain pyrazole derivatives with human SGLTl inhibitory activity which are further disclosed as useful for the prevention or treatment of a disease associated with hyperglycemia, such as diabetes. In addition, WO 201 1/039338 discloses certain pyrazole derivatives with SGLT1/SGLT2 inhibitor activity which are further disclosed as being useful for treatment of bone diseases, such as osteoporosis.
There is a need for alternative drugs and treatment for diabetes. The present invention provides certain novel inhibitors of SGLTl which may be suitable for the treatment of diabetes.
Accordingly, the present invention provides a compound of Formula II:
Preparation 1
Synthesis of (4-bromo-2-methyl-phenyl)methanol.
Scheme 1, step A: Add borane-tetrahydrofuran complex (0.2 mol, 200 mL, 1.0 M solution) to a solution of 4-bromo-2-methylbenzoic acid (39 g, 0.18 mol) in
tetrahydrofuran (200 mL). After 18 hours at room temperature, remove the solvent under the reduced pressure to give a solid. Purify by flash chromatography to yield the title compound as a white solid (32.9 g, 0.16 mol). 1H NMR (CDCI3): δ 1.55 (s, 1H), 2.28 (s, 3H), 4.61 (s, 2H), 7.18-7.29 (m, 3H).
Alternative synthesis of (4-bromo-2-methyl-phenyl)methanol.
Borane-dimethyl sulfide complex (2M in THF; 1 16 mL, 0.232 mol) is added slowly to a solution of 4-bromo-2-methylbenzoic acid (24.3 g, 0.1 13 mol) in anhydrous tetrahydrofuran (THF, 146 mL) at 3 °C. After stirring cold for 10 min the cooling bath is removed and the reaction is allowed to warm slowly to ambient temperature. After 1 hour, the solution is cooled to 5°C, and water (100 mL) is added slowly. Ethyl acetate (100 mL) is added and the phases are separated. The organic layer is washed with saturated aqueous NaHC03 solution (200 mL) and dried over Na2S04. Filtration and concentration under reduced pressure gives a residue which is purified by filtration through a short pad of silica eluting with 15% ethyl acetate/iso-hexane to give the title compound (20.7 g, 91.2% yield). MS (m/z): 183/185 (M+l-18).
Preparation 2
Synthesis of 4-bromo- l-2-methyl-benzene.
Scheme 1, step B: Add thionyl chloride (14.31 mL, 0.2 mol,) to a solution of (4-bromo-2-methyl-phenyl)methanol (32.9 g, 0.16 mol) in dichloromethane (200 mL) and
-Cl-
dimethylformamide (0.025 mol, 2.0 mL) at 0°C. After 1 hour at room temperature pour the mixture into ice-water (100 g), extract with dichloromethane (300 mL), wash extract with 5% aq. sodium bicarbonate (30 mL) and brine (200 mL), dry over sodium sulfate, and concentrate under reduced pressure to give the crude title compound as a white solid (35.0 g, 0.16 mol). The material is used for the next step of reaction without further purification. XH NMR (CDC13): δ 2.38 (s, 3H), 4.52 (s, 2H), 7.13-7.35 (m, 3H).
Alternative synthesis of 4-bromo- 1 -chloromethyl-2-methyl-benzene. Methanesulfonyl chloride (6.83 mL, 88.3 mmol) is added slowly to a solution of (4-bromo-2-methyl-phenyl)methanol (16.14 g, 80.27 mmol) and triethylamine (16.78 mL; 120.4 mmol) in dichloromethane (80.7 mL) cooled in ice/water. The mixture is allowed to slowly warm to ambient temperature and is stirred for 16 hours. Further
methanesulfonyl chloride (1.24 mL; 16.1 mmol) is added and the mixture is stirred at ambient temperature for 2 hours. Water (80mL) is added and the phases are separated. The organic layer is washed with hydrochloric acid (IN; 80 mL) then saturated aqueous sodium hydrogen carbonate solution (80 mL), then water (80 mL), and is dried over Na2S04. Filtration and concentration under reduced pressure gives a residue which is purified by flash chromatography (eluting with hexane) to give the title compound (14.2 g; 80.5% yield). XH NMR (300.1 1 MHz, CDC13): δ 7.36-7.30 (m, 2H), 7.18 (d, J= 8.1 Hz, 1H), 4.55 (s, 2H), 2.41 (s, 3H).
Preparation 3
Synthesis of 4-[(4-bromo-2-methyl-phenyl)methyl]-5-isopropyl-lH-pyrazol-3-ol.
Scheme 1, step C: Add sodium hydride (8.29 g, 0.21 mol, 60% dispersion in oil) to a solution of methyl 4-methyl-3-oxovalerate (27.1 mL, 0.19 mol) in tetrahydrofuran at 0°C. After 30 min at room temperature, add a solution of 4-bromo- l-chloromethyl-2-methyl-benzene (35.0 g, 0.16 mol) in tetrahydrofuran (50 mL). Heat the resulting mixture at 70 °C overnight (18 hours). Add 1.0 M HC1 (20 mL) to quench the reaction.
Extract with ethyl acetate (200 mL), wash extract with water (200 rnL) and brine (200 mL), dry over a2S04, filter and concentrate under reduced pressure. Dissolve the resulting residue in toluene (200 mL) and add hydrazine monohydrate (23.3 mL, 0.48 mol). Heat the mixture at 120 °C for 2 hours with a Dean-Stark apparatus to remove water. Cool and remove the solvent under the reduced pressure, dissolve the residue with dichloromethane (50 mL) and methanol (50 mL). Pour this solution slowly to a beaker with water (250 mL). Collect the resulting precipitated product by vacuum filtration. Dry in vacuo in an oven overnight at 40 °C to yield the title compound as a solid (48.0 g, 0.16 mol). MS (m/z): 311.0 (M+l), 309.0 (M-l).
Alternative synthesis of 4-r(4-bromo-2-methyl-phenyl)methyl1-5-isopropyl- !H-pyrazol- 3-oL
A solution of 4-bromo- 1 -chloromethyl-2-methyl-benzene (13.16 g, 59.95 mmoles) in acetonitrile (65.8 mL) is prepared. Potassium carbonate (24.86 g, 179.9 mmol), potassium iodide (1 1.94 g, 71.94 mmol) and methyl 4-methyl-3-oxo valerate (8.96 mL; 62.95 mmol) are added. The resulting mixture is stirred at ambient temperature for 20 hours. Hydrochloric acid (2N) is added to give pH 3. The solution is extracted with ethyl acetate (100 ml), the organic phase is washed with brine (100 ml) and dried over Na2S04. The mixture is filtered and concentrated under reduced pressure. The residue is dissolved in toluene (65.8 mL) and hydrazine monohydrate (13.7 mL, 0.180 mol) is added. The resulting mixture is heated to reflux and water is removed using a Dean and Stark apparatus. After 3 hours the mixture is cooled to 90 °C and additional hydrazine monohydrate (13.7 mL; 0.180 mol) is added and the mixture is heated to reflux for 1 hour. The mixture is cooled and concentrated under reduced pressure. The resulting solid is triturated with water (200 mL), filtered and dried in a vacuum oven over P2O5 at 60°C. The solid is triturated in iso-hexane (200 mL) and filtered to give the title compound (14.3 g; 77.1% yield). MS (m/z): 309/31 1 (M+l).
Preparation 4
Synthesis of 4-(4-bromo-2-methylbenzyl)-5-(propan-2-yl)-lH-pyrazol-3-yl 2,3,4,6-tetra- O-benzoyl-beta-D-glucopyranoside.
Scheme 1, step D: To a 1L flask, add 4-[(4-bromo-2-methyl-phenyl)methyl]-5-isopropyl-lH-pyrazol-3-ol (20 g, 64.7 mmol), alpha-D-glucopyranosyl bromide tetrabenzoate (50 g, 76 mmol), benzyltributylammonium chloride (6 g, 19.4 mmol), dichloromethane (500 mL), potassium carbonate (44.7 g, 323 mmol) and water (100 mL). Stir the reaction mixture overnight at room temperature. Extract with dichloromethane (500mL). Wash extract with water (300 mL) and brine (500 mL). Dry organic phase over sodium sulfate, filter, and concentrate under reduced pressure. Purify the residue by flash chromatography to yield the title compound (37 g, 64 mmol). MS (ml 2): 889.2 (M+l), 887.2 (M-l).
Preparation 5
Synthesis of 4- {4-[( lis)-4-hydroxybut- 1 -en- 1 -yl]-2-methylbenzyl} -5-(propan-2-yl)- 1H- pyrazol-3-yl 2,3,4,6-tetra-O-benzoyl-beta-D-glucopyranoside.
Scheme 1, step E: Add 3-buten-l-ol (0.58 mL, 6.8 mmol) to a solution of 4-(4-bromo-2-methylbenzyl)-5-(propan-2-yl)-lH-pyrazol-3-yl 2,3,4,6-tetra-O-benzoyl-beta-D-glucopyranoside (3 g, 3.4 mmol) in acetonitrile (30 mL) and triethylamine (20 mL). Degas the solution with nitrogen over 10 minutes. Add tri-o-tolylphosphine (205 mg, 0.67 mmol) and palladium acetate (76 mg, 0.34 mmol). Reflux at 90 °C for 2 hours. Cool to room temperature and concentrate to remove the solvent under the reduced pressure. Purify the residue by flash chromatography to yield the title compound (2.1 g, 2.4 mmol). MS (m/z): 878.4 (M+l).
Preparation 6
Synthesis of 4-{4-[(l£)-4-oxybut-l-en-l-yl]-2-methylbenzyl}-5-(propan-2-yl)-lH- pyrazol-3-yl 2,3,4,6-tetra-O-benzoyl-beta-D-glucopyranoside.
Scheme 1, step F: Add 3,3,3-triacetoxy-3-iodophthalide (134 mg, 0.96 mmol) to a solution of 4-{4-[(l£)-4-hydroxybut-l-en-l-yl]-2-methylbenzyl}-5-(propan-2-yl)-lH-pyrazol-3-yl 2,3,4,6-tetra-O-benzoyl-beta-D-glucopyranoside (280 mg, 0.32 mmol) and sodium bicarbonate (133.8 mg, 1.6 mmol) in dichloromethane (20 mL) at 0 °C. After 15 minutes at room temperature, quench the reaction with saturated aqueous sodium thiosulfate (10 mL). Extract with dichloromethane (30 mL). Wash extract with water (30 mL) and brine (40 mL). Dry organic phase over sodium sulfate, filter, and concentrate under reduced pressure. Purify the resulting residue by flash chromatography to yield the title compound (270 mg, 0.31 mmol). MS (m/z): 876.5 (M+l), 874.5 (M-l).
Preparation 7
Synthesis of tert-butyl 2- {(3JE)-4-[3-methyl-4-({5-(propan-2-yl)-3-[(2,3,4,6-tetra-0-benzoyl-beta-D-glucopyranosyl)oxy]-lH-pyrazol-4-yl}methyl)phenyl]but-3-en-l-yl}-2,9- diazaspiro[5.5]undecane-9-carboxylate.
Scheme 1, step G: Add sodium triacetoxyborohydride (98 mg, 0.46 mmol) to a solution of 4- {4-[(lis)-4-oxybut- 1 -en-1 -yl]-2-methylbenzyl} -5-(propan-2-yl)- lH-pyrazol-3-yl 2,3,4,6-tetra-O-benzoyl-beta-D-glucopyranoside (270 mg, 0.31 mmol) and tert-butyl 2,9-diazaspiro[5.5]undecane-9-carboxylate hydrochloride (179 mg, 0.62 mmol) in 1,2-dichloroethane (5 mL). After 30 minutes at room temperature, quench the reaction with saturated aqueous sodium bicarbonate (10 mL). Extract with dichloromethane (30 mL). Wash extract with water (30 mL) and brine (40 mL), dry organic phase over sodium sulfate, filter and concentrate under reduced pressure. Purify the resulting residue by flash chromatography to yield the title compound (275 mg, 0.25 mmol).
MS (m/z): 1115.6 (M+1).
Preparation 8
Synthesis of 4-{4-[(l£)-4-(2,9-diazaspiro[5.5]undec-2-yl)but-l-en-l-yl]-2- methylbenzyl}-5-(propan-2-yl)-lH-pyrazol-3-yl 2,3,4,6-tetra-O-benzoyl-beta-D- glucopyranoside dihydrochloride.
Scheme 1, step H: Add hydrogen chloride (4.0 M solution in 1,4-dioxane, 0.6 mL, 2.4 mmol) to a solution of tert-butyl 2-{(3is)-4-[3-methyl-4-({5-(propan-2-yl)-3-[(2,3,4,6-tetra-0-benzoyl-beta-D-glucopyranosyl)oxy]-lH-pyrazol-4-yl}methyl)phenyl]but-3-en-l-yl}-2,9-diazaspiro[5.5]undecane-9-carboxylate (275 mg, 0.25 mmol) in dichloromethane (5 mL). After overnight (18 hours) at room temperature, concentrate to remove the solvent under reduced pressure to yield the title compound as a solid (258 mg, 0.24 mmol). MS (m/z): 1015.6 (M+l).
Example 1
Synthesis of 4-{4-[(l£)-4-(2,9-diazaspiro[5.5]undec-2-yl)but-l-en-l-yl]-2- methylbenzyl} -5-(propan-2-yl)- lH-pyrazol-3-yl beta-D-glucopyranoside.
Scheme 1, step I: Add sodium hydroxide (0.5 mL, 0.5 mmol, 1.0 M solution) to a solution of 4-{4-[(l£)-4-(2,9-diazaspiro[5.5]undec-2-yl)but-l-en-l-yl]-2-methylbenzyl}-5-(propan-2-yl)-lH-pyrazol-3-yl 2,3,4,6-tetra-O-benzoyl-beta-D-glucopyranoside dihydrochloride (258 mg, 0.24 mmol) in methanol (2 mL). After 2 hours at 40 °C, concentrate to remove the solvent under reduced pressure to give a residue, which is purified by preparative HPLC method: high pH, 25% B for 4 min, 25-40 B % for 4 min @ 85 mL/min using a 30 x 75 mm, 5 um C18XBridge ODB column, solvent A – 1¾0 w NH4HCO3 @ pH 10, solvent B – MeCN to yield the title compound as a solid (46 mg, 0.08 mmol). MS (m/z): 598.8 (M+l), 596.8 (M-l).
Preparation 9
Synthesis of 4-(4-bromo-2-methylbenzyl)-5-(propan-2-yl)-lH-pyrazol-3-yl 2,3,4,6-tetra- O-acetyl-beta-D-glucopyranoside.
Scheme 2, step A: To a 1 L flask, add 4-[(4-bromo-2-methyl-phenyl)methyl]-5-isopropyl-lH-pyrazol-3-ol (24 g, 77.6 mmol), 2,3,4,6-tetra-O-acetyl-alpha-D-glucopyranosyl bromide (50.4 g, 116 mmol), benzyltributylammomum chloride (5 g, 15.5 mmol), dichloromethane (250 mL), potassium carbonate (32 g, 323 mmol) and water (120 mL). Stir the reaction mixture overnight at room temperature. Extract with dichloromethane (450 mL). Wash extract with water (300 mL) and brine (500 mL). Dry organic phase over sodium sulfate, filter, and concentrate under reduced pressure. Purify the resulting residue by flash chromatography to yield the title compound (36.5 g, 57 mmol). MS (m/z): 638.5 (M+l), 636.5 (M-l).
Alternative synthesis of 4-(4-bromo-2-methylbenzyl)-5-(propan-2-yl)-lH-pyrazol-3-yl
2,3,4,6-tetra-O-acetyl-beta-D-glucopyranoside.
Reagents 4-[(4-bromo-2-methyl-phenyl)methyl]-5-isopropyl-lH-pyrazol-3-ol (24.0 g, 77.6 mmol), 2,3,4,6-tetra-O-acetyl-alpha-D-glucopyranosyl bromide (50.4 g, 116 mmol), benzyltributylammonium chloride (4.94 g, 15.52 mmol), potassium carbonate
(32.18 g, 232.9 mmol), dichloromethane (250 mL) and water (120 mL) are combined and the mixture is stirred at ambient temperature for 18 hours. The mixture is partitioned between dichloromethane (250 mL) and water (250 mL). The organic phase is washed with brine (250 mL), dried over Na2S04, filtered, and concentrated under reduced pressure. The resulting residue is purified by flash chromatography (eluting with 10% ethyl acetate in dichloromethane to 70% ethyl acetate in dichloromethane) to give the title compound (36.5 g, 74% yield). MS (m/z): 639/641 (M+l).
Preparation 10
Synthesis of 4- {4-[( lis)-4-hydroxybut- 1 -en- 1 -yl]-2-methylbenzyl} -5-(propan-2-yl)- 1H- pyrazol-3-yl 2,3,4,6-tetra-O-acetyl-beta-D-glucopyranoside.
Scheme 2, step B: Add 3-buten-l-ol (6.1 mL, 70 mmol) to a solution of 4-(4-bromo-2-methylbenzyl)-5-(propan-2-yl)-lH-pyrazol-3-yl 2,3,4,6-tetra-O-acetyl-beta-D-glucopyranoside (15 g, 23.5 mmol) in acetonitrile (200 mL) and triethylamine (50 mL). Degas the solution with nitrogen over 10 minutes. Add tri-o-tolylphosphine (1.43 g, 4.7 mmol) and palladium acetate (526 mg, 2.35 mmol). After refluxing at 90 °C for 2 hours, cool, and concentrate to remove the solvent under the reduced pressure. Purify the resulting residue by flash chromatography to yield the title compound (7.5 g, 11.9 mmol). MS (m/z): 631.2 (M+l), 629.2 (M-l).
Preparation 11
Synthesis of 4-{4-[(l£)-4-oxybut-l-en-l-yl]-2-methylbenzyl}-5-(propan-2-yl)-lH- pyrazol-3-yl 2,3,4,6-tetra-O-acetyl-beta-D-glucopyranoside.
Scheme 2, step C: Add 3,3,3-triacetoxy-3-iodophthalide (2.1g, 4.76 mmol) to a solution of 4-{4-[(l£)-4-hydroxybut-l-en-l-yl]-2-methylbenzyl}-5-(propan-2-yl)-lH-pyrazol-3-yl 2,3,4,6-tetra-O-acetyl-beta-D-glucopyranoside ( 1.5 g, 2.38 mmol) and sodium bicarbonate (2 g, 23.8 mmol) in dichloromethane (50 mL) at 0 °C. After 15 minutes at room temperature, quench the reaction with saturated aqueous sodium thiosulfate (10 mL). Extract with dichloromethane (30 mL), wash extract with water (30 mL) and brine (40 mL). Dry organic phase over sodium sulfate, filter, and concentrate under reduced pressure. Purify the resulting residue by flash chromatography to yield the title compound (0.95 g, 1.51 mmol). MS (m/z): 628.8(M+1), 626.8 (M-l).
Preparation 12
Synthesis of tert-butyl 2-{(3JE)-4-[3-methyl-4-({5-(propan-2-yl)-3-[(2,3,4,6-tetra-0- acetyl-beta-D-glucopyranosyl)oxy]-lH-pyrazol-4-yl}methyl)phenyl]but-3-en-l-yl}-2,9- diazaspiro[5.5]undecane-9-carboxylate.
Scheme 2, Step D: Add sodium triacetoxyborohydride (303 mg, 1.4 mmol) to a solution of 4- {4-[(lis)-4-oxybut- 1 -en-1 -yl]-2-methylbenzyl} -5-(propan-2-yl)- lH-pyrazol-3-yl 2,3,4,6-tetra-O-acetyl-beta-D-glucopyranoside (600 mg, 0.95 mmol) and tert-butyl 2,9-diazaspiro[5.5]undecane-9-carboxylate hydrochloride (333 mg, 1.2 mmol) in 1,2-dichloroethane (30 mL). After 30 minutes at room temperature, quench the reaction with saturated aqueous sodium bicarbonate (15 mL). Extract with dichloromethane (60 mL). Wash extract with water (30 mL) and brine (60 mL). Dry organic phase over sodium sulfate, filter, and concentrate under reduced pressure. Purify the resulting residue by flash chromatography to yield the title compound (500 mg, 0.58 mmol).
MS (m/z): 866.8, 867.8 (M+l), 864.8, 865.8 (M-l).
Preparation 13
Synthesis oftert-butyl 2-{(3E)-4-[3-methyl-4-({5-(propan-2-yl)-3-[(2,3,4,6-tetra-0-acetyl-beta-D-glucopyranosyl)oxy]-lH-pyrazol-4-yl}methyl)phenyl]but-3-en-l-yl}-2,8- diazaspiro[4.5]decane-8-carboxylate.
The title compound is prepared essentially by the method of Preparation 12. S (m/z): 852.8, 853.6 (M+l), 850.8, 851.6 (M-l).
Preparation 14
Synthesis oftert-butyl 9-{(3E)-4-[3-methyl-4-({5-(propan-2-yl)-3-[(2,3,4,6-tetra-0-acetyl-beta-D-glucopyranosyl)oxy]-lH-pyrazol-4-yl}methyl)phenyl]but-3-en-l-yl}-3,9- diazaspiro[5.5]undecane-3-carboxylate.
The title compound is prepared essentially by the method of Preparation 12. S (m/z): 866.8, 867.6 (M+l), 864.8, 865.6 (M-l).
Preparation 15
Synthesis of 4-{4-[(l£)-4-(2,9-diazaspiro[5.5]undec-2-yl)but-l-en-l-yl]-2- methylbenzyl}-5-(propan-2-yl)-lH-pyrazol-3-yl 2,3,4,6-tetra-O-acetyl-beta-D- glucopyranoside dihydrochloride.
Scheme 2, step E: Add hydrogen chloride (4.0 M solution in 1,4-dioxane, 1.5 mL, 5.8 mmol) to a solution of tert-butyl 2-{(3£)-4-[3-methyl-4-({5-(propan-2-yl)-3-[(2,3,4,6-tetra-0-acetyl-beta-D-glucopyranosyl)oxy]- lH-pyrazol-4-yl} methyl)phenyl]but-3 -en- 1 -yl}-2,9-diazaspiro[5.5]undecane-9-carboxylate (500 mg, 0.58 mmol) in dichloromethane (20 mL). After 2 hours at room temperature, concentrate to remove the solvent under reduced pressure to yield the title compound as a solid (480 mg, 0.57 mmol).
MS (m/z): 767.4 (M+l).
Preparation 16
Synthesis of 4-{4-[(lE)-4-(2,8-diazaspiro[4.5]dec-2-yl)but-l-en-l-yl]-2-methylbenzyl}-5- (propan-2-yl)-lH-pyrazol-3-yl 2,3,4,6-tetra-O-acetyl-beta-D-glucopyranoside
dihydrochloride.
The title compound is prepared essentially by the method of Preparation 15. MS (m/z): 752.8, 753.8 (M+1), 750.8 (M-1).
First alternative synthesis of Example 1
First alternative synthesis of 4-{4-[(l£)-4-(2,9-diazaspiro[5.5]undec-2-yl)but-l-en- 2-methylbenzyl}-5-(propan-2-yl)-lH-pyrazol-3-yl beta-D-glucopyranoside.
Scheme 2, step F: Add methanol (5 mL), triethylamine (3 mL), and water (3 mL) to 4-{4-[(l£)-4-(2,9-diazaspiro[5.5]undec-2-yl)but-l-en-l-yl]-2-methylbenzyl}-5-(propan-2-yl)-lH-pyrazol-3-yl 2,3,4,6-tetra-O-acetyl-beta-D-glucopyranoside dihydrochloride (480 mg, 0.24 mmol). After 18 hours (overnight) at room temperature, concentrate to dryness under reduced pressure. Purify the resulting residue by preparative HPLC method: high pH, 25% B for 4 min, 25-40 B % for 4 min @ 85 mL/min using a 30 x 75 mm, 5 urn C18XBridge ODB column, solvent A – H20 w NH4HCO3 @ pH 10, solvent B – MeCN to yield the title compound as a solid (50 mg, 0.08 mmol).
MS (m/z): 598.8 (M+1), 596.8 (M-1). 1H MR (400.31 MHz, CD3OD): δ 7.11 (d, J=1.3
Hz, 1H), 7.04 (dd, J=1.3,8.0 Hz, 1H), 6.87 (d, J= 8.0 Hz, 1H), 6.36 (d, J= 15.8 Hz, 1H), 6.16 (dt, J= 15.8, 6.3 Hz, 1H), 5.02 (m, 1H), 3.81 (d, J= 11.7 Hz, 1H), 3.72 (d, J= 16.8 Hz, 1H), 3.68 (d, J= 16.8 Hz, 1H) , 3.64 (m, 1H), 3.37-3.29 (m, 4H), 2.79 (m, 1H), 2.72 (t, J= 5.8 Hz, 4H), 2.44-2.33 (m, 6H), 2.30 (s, 3H), 2.26 ( broad s, 2H), 1.59 (m, 2H), 1.50 (m, 2H), 1.43 (m, 2H), 1.36 (m, 2H), 1.1 1 (d, J= 7.0 Hz, 3H), 1.10 (d, J= 7.0 Hz, 3H).
Example 2
Synthesis of 4- {4-[(lE)-4-(2,8-diazaspiro[4.5]dec-2-yl)but-l-en-l-yl]-2-methylbi
(propan-2-yl)-lH-pyrazol-3-yl beta-D-glucopyranoside.
O H
The title compound is prepared essentially by the method of the first alternative synthesis of Example 1. MS (m/z): 584.7 (M+l), 582.8 (M-l).
Example 3
Synthesis of 4- {4-[( 1 E)-4-(3 ,9-diazaspiro[5.5]undec-3 -yl)but- 1 -en- 1 -yl]-2- methylbenzyl} -5-(propan-2-yl)- lH-pyrazol-3-yl beta-D-glucopyranoside.
The title compound is prepared essentially by first treating the compound of Prearation 14 with HC1 as discussed in Preparation 15 then treating the resulting hydrochloride salt with triethyl amine as discussed in the first alternative synthesis of Example 1. MS (m/z): 598.8, 599.8 (M+l), 596.8, 597.8 (M-l).
Example 1 Preparation 17
Synthesis of tert-butyl 4-but-3- nyl-4,9-diazaspiro[5.5]undecane-9-carboxylate.
Scheme 3, step A: Cesium carbonate (46.66 g, 143.21 mmol) is added to a suspension of tert-butyl 4,9-diazaspiro[5.5]undecane-9-carboxylate hydrochloride (16.66 g, 57.28 mmoles) in acetonitrile (167 mL). The mixture is stirred for 10 minutes at ambient temperature then 4-bromobutyne (6.45 mL, 68.74 mmol) is added. The reaction is heated to reflux and stirred for 18 hours. The mixture is cooled and concentrated under reduced pressure. The residue is partitioned between water (200 mL) and ethyl acetate (150 mL). The phases are separated and the aqueous layer is extracted with ethyl acetate (100 mL). The combined organic layers are washed with water (200 mL), then brine (150 mL), dried over MgSC^, filtered, and concentrated under reduced pressure to give the title compound (17.2 g, 98% yield). iH MR (300.11 MHz, CDC13): δ 3.43-3.31 (m, 4H),
2.53-2.48 (m, 2H), 2.37-2.29 (m, 4H), 2.20 (s, 2H), 1.94 (t, J= 2.6 Hz, 1H), 1.44 (s, 17H).
Preparation 18
Synthesis of tert-butyl 4-[(£)-4-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2-yl)but-3-enyl]- 4,9-diazaspiro[5.5]undecane-9-carboxylate.
Scheme 3, step B: Triethylamine (5.62 mmoles; 0.783 mL), 4,4,5, 5-tetramethyl-1,3,2-dioxaborolane (8.56 mL, 59.0 mmol) and zirconocene chloride (1.45 g, 5.62 mmoles) are added to tert-butyl 4-but-3-ynyl-4,9-diazaspiro[5.5]undecane-9-carboxylate (17.21 g, 56.16 mmoles). The resulting mixture is heated to 65 °C for 3.5 hours. The mixture is cooled and dissolved in dichloromethane (150 mL). The resulting solution is passed through a ~4cm thick pad of silica gel, eluting with dichloromethane (2 x 200 mL). The filtrate is concentrated under reduced pressure to give the title compound (21.2 g, 87% yield), !H NMR (300.1 1 MHz, CDC13): δ 6.65-6.55 (m, 1H), 5.49-5.43 (m, 1H),
3.42-3.29 (m, 4H), 2.40-2.27 (m, 6H), 2.25-2.08 (m, 2H), 1.70 – 1.13 (m, 29H).
Preparation 19
Synthesis of tert-butyl 2-{(3JE)-4-[3-methyl-4-({5-(propan-2-yl)-3-beta-D- glucopyranosyl)oxy]- lH-pyrazol-4-yl} methyl)phenyl]but-3 -en- 1 -yl} -2,9- diazaspiro[5.5]undecane-9-carboxylate.
Scheme 3, step C: A solution of 4-(4-bromo-2-methylbenzyl)-5-(propan-2-yl)-lH-pyrazol-3-yl 2,3,4,6-tetra-O-acetyl-beta-D-glucopyranoside (20 g, 31.3 mmol), tert-butyl 4-[(£)-4-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2-yl)but-3-enyl]-4,9-diazaspiro[5.5]undecane-9-carboxylate (16.3 g, 37.5 mmol) and potassium carbonate (12.97 g, 93.82 mmol) in tetrahydrofuran (200 mL) and water (40 mL) is degassed for 15 min by bubbling nitrogen gas through it. Pd(OAc)2 (140 mg, 625 μιηοΐ) and 2-dicyclohexylphosphino-2′,4′,6′-tri-i-propyl-l, r-biphenyl (0.596 g, 1.25 mmol) are added and the reaction is heated to reflux for 16 h. The solution is cooled to ambient temperature and methanol (200 mL) is added. After 30 minutes the solvent is removed under reduced pressure. The mixture is partitioned between ethyl acetate (500 mL) and brine (500 ml) adding aqueous MgS04 (1M; 500 ml) to aid the phase separation. The layers are separated and the organic layer is dried over MgS04 and filtered through a 10 cm pad of silica gel, eluting with ethyl acetate (-1.5 L). The filtrate is discarded and the silica pad is flushed with 5% MeOH in THF (2 L). The methanolic filtrate is concentrated under reduced pressure to give the title compound (20. lg, 92%).
MS (m/z): 699 (M+l).
Second alternative Synthesis of Example 1
Second alternative synthesis of 4- {4-[(l£)-4-(2,9-diazaspiro[5.5]undec-2-yl)but-l-en-l- yl]-2-methylbenzyl}-5-(propan-2-yl)-lH-pyrazol-3-yl beta-D-glucopyranoside.
Scheme 3, step D: Trifluoroacetic acid (32.2 mL; 0.426 mol) is added to a solution of tert-butyl 2- {(3JE)-4-[3-methyl-4-({5-(propan-2-yl)-3-beta-D-glucopyranosyl)oxy]-lH-pyrazol-4-yl}methyl)phenyl]but-3-en-l-yl}-2,9-diazaspiro[5.5]undecane-9-carboxylate (14.87 g; 21.28 mmol) in dichloromethane (149 mL) cooled in iced water. The solution is allowed to warm to room temperature. After 30 minutes, the mixture is slowly added to ammonia in MeOH (2M; 300 mL), applying cooling as necessary to maintain a constant temperature. The solution is stirred at room temperature for 15 min. The mixture is concentrated under reduced pressure and the residue is purified using SCX-2 resin. The basic filtrate is concentrated under reduced pressure and the residue is triturated/sonicated in ethyl acetate, filtered and dried. The resulting solid is dissolved in MeOH (200ml) and concentrated in vacuo. This is repeated several times give the title compound (12.22 g, yield 96%). MS (m/z): 599 (M+l). [a]D20 = -12 ° (C=0.2, MeOH).
PATENT
WO 2015069541
https://patents.google.com/patent/WO2015069541A1
4-{4-[(1 E)-4-(2,9-DIAZASPIRO[5.5]UNDEC-2-YL)BUT-1 -EN-1
-YL]-2-METHYLBENZYL}-5-(PROPAN-2-YL)-1 H-PYRAZOL-3-YL
BETA-D- GLUCOPYRANOSIDE ACETATE
The present invention relates to a novel SGLT1 inhibitor which is an acetate salt of a pyrazole compound, to pharmaceutical compositions comprising the compound, to methods of using the compound to treat physiological disorders, and to intermediates and processes useful in the synthesis of the compound.
The present invention is in the field of treatment of diabetes and other diseases and disorders associated with hyperglycemia. Diabetes is a group of diseases that is characterized by high levels of blood glucose. It affects approximately 25 million people in the United States and is also the 7th leading cause of death in U.S. according to the 2011 National Diabetes Fact Sheet (U.S. Department of Health and Human Services, Centers for Disease Control and Prevention). Sodium-coupled glucose cotransporters (SGLT’s) are one of the transporters known to be responsible for the absorption of carbohydrates, such as glucose. More specifically, SGLT1 is responsible for transport of glucose across the brush border membrane of the small intestine. Inhibition of SGLT1 may result in reduced absorption of glucose in the small intestine, thus providing a useful approach to treating diabetes.
U.S. Patent No. 7,655,632 discloses certain pyrazole derivatives with human SGLT1 inhibitory activity which are further disclosed as useful for the prevention or treatment of a disease associated with hyperglycemia, such as diabetes. In addition, WO 2011/039338 discloses certain pyrazole derivatives with SGLT1/SGLT2 inhibitor activity which are further disclosed as being useful for treatment of bone diseases, such as osteoporosis.
There is a need for alternative drugs and treatment for diabetes. The present invention provides an acetate salt of a pyrazole compound, which is an SGLT1 inhibitor, and as such, may be suitable for the treatment of certain disorders, such as diabetes. Accordingly, the present invention provides a compound of Formula I:
or hydrate thereof.
Preparation 1
(4-bromo-2-methyl-phenyl)methanol
Scheme 1, step A: Add borane-tetrahydrofuran complex (0.2 mol, 200 mL, 1.0 M solution) to a solution of 4-bromo-2-methylbenzoic acid (39 g, 0.18 mol) in
tetrahydrofuran (200 mL). After 18 hours at room temperature, remove the solvent under the reduced pressure to give a solid. Purify by flash chromatography to yield the title compound as a white solid (32.9 g, 0.16 mol). !H NMR (CDCI3): δ 1.55 (s, 1H), 2.28 (s, 3H), 4.61 (s, 2H), 7.18-7.29 (m, 3H).
Alternative synthesis of (4-bromo-2-methyl-phenyl)mefhanol.
Borane-dimethyl sulfide complex (2M in THF; 116 mL, 0.232 mol) is added slowly to a solution of 4-bromo-2-methylbenzoic acid (24.3 g, 0.113 mol) in anhydrous tetrahydrofuran (THF, 146 mL) at 3 °C. After stirring cold for 10 min the cooling bath is removed and the reaction is allowed to warm slowly to ambient temperature. After 1 hour, the solution is cooled to 5°C, and water (100 mL) is added slowly. Ethyl acetate (100 mL) is added and the phases are separated. The organic layer is washed with saturated aqueous NaHC03 solution (200 mL) and dried over Na2S04. Filtration and concentration under reduced pressure gives a residue which is purified by filtration through a short pad of silica eluting with 15% ethyl acetate/iso-hexane to give the title compound (20.7 g, 91.2% yield). MS (m/z): 183/185 (M+l-18).
Preparation 2
4-bromo- 1 -chloromethyl -2 -methyl -benzene
Scheme 1, step B: Add thionyl chloride (14.31 mL, 0.2 mol,) to a solution of (4- bromo-2 -methyl -phenyl)methanol (32.9 g, 0.16 mol) in dichloromethane (200 mL) and dimethylformamide (0.025 mol, 2.0 mL) at 0°C. After 1 hour at room temperature pour the mixture into ice-water (100 g), extract with dichloromethane (300 mL), wash extract with 5% aq. sodium bicarbonate (30 mL) and brine (200 mL), dry over sodium sulfate, and concentrate under reduced pressure to give the crude title compound as a white solid (35.0 g, 0.16 mol). The material is used for the next step of reaction without further purification. !H NMR (CDC13): δ 2.38 (s, 3H), 4.52 (s, 2H), 7.13-7.35 (m, 3H).
Alternative synthesis of 4-bromo-l-chloromethyl-2-methyl -benzene. Methanesulfonyl chloride (6.83 mL, 88.3 mmol) is added slowly to a solution of (4-bromo-2-methyl-phenyl)methanol (16.14 g, 80.27 mmol) and triethylamine (16.78 mL; 120.4 mmol) in dichloromethane (80.7 mL) cooled in ice/water. The mixture is allowed to slowly warm to ambient temperature and is stirred for 16 hours. Further
methanesulfonyl chloride (1.24 mL; 16.1 mmol) is added and the mixture is stirred at ambient temperature for 2 hours. Water (80mL) is added and the phases are separated. The organic layer is washed with hydrochloric acid (IN; 80 mL) then saturated aqueous sodium hydrogen carbonate solution (80 mL), then water (80 mL), and is dried over Na2S04. Filtration and concentration under reduced pressure gives a residue which is purified by flash chromatography (eluting with hexane) to give the title compound (14.2 g; 80.5% yield). !H NMR (300.11 MHz, CDC13): δ 7.36-7.30 (m, 2H), 7.18 (d, J= 8.1 Hz, 1H), 4.55 (s, 2H), 2.41 (s, 3H).
Preparation 3
4- [(4-bromo-2-methyl-phenyl)methyl] -5 -isopropyl- lH-pyrazol-3 -ol
Scheme 1, step C: Add sodium hydride (8.29 g, 0.21 mol, 60% dispersion in oil) to a solution of methyl 4-methyl-3-oxovalerate (27.1 mL, 0.19 mol) in tetrahydrofuran at 0°C. After 30 min at room temperature, add a solution of 4-bromo-l-chloromethyl-2- methyl-benzene (35.0 g, 0.16 mol) in tetrahydrofuran (50 mL). Heat the resulting mixture at 70 °C overnight (18 hours). Add 1.0 M HC1 (20 mL) to quench the reaction. Extract with ethyl acetate (200 mL), wash extract with water (200 mL) and brine (200 mL), dry over Na2S04, filter and concentrate under reduced pressure. Dissolve the resulting residue in toluene (200 mL) and add hydrazine monohydrate (23.3 mL, 0.48 mol). Heat the mixture at 120 °C for 2 hours with a Dean-Stark apparatus to remove water. Cool and remove the solvent under the reduced pressure, dissolve the residue with dichloromethane (50 mL) and methanol (50 mL). Pour this solution slowly to a beaker with water (250 mL). Collect the resulting precipitated product by vacuum filtration. Dry in vacuo in an oven overnight at 40 °C to yield the title compound as a solid (48.0 g, 0.16 mol). MS (m/z): 311.0 (M+l), 309.0 (M-l). Alternative synthesis of 4-[(4-bromo-2-methyl-phenyl)methyl] -5 -isopropyl- lH-pyrazol-
3-ol.
A solution of 4-bromo-l-chloromethyl-2-methyl-benzene (13.16 g, 59.95 mmoles) in acetonitrile (65.8 mL) is prepared. Potassium carbonate (24.86 g, 179.9 mmol), potassium iodide (11.94 g, 71.94 mmol) and methyl 4-methyl-3-oxovalerate (8.96 mL; 62.95 mmol) are added. The resulting mixture is stirred at ambient temperature for 20 hours. Hydrochloric acid (2N) is added to give pH 3. The solution is extracted with ethyl acetate (100 ml), the organic phase is washed with brine (100 ml) and dried over Na2S04. The mixture is filtered and concentrated under reduced pressure. The residue is dissolved in toluene (65.8 mL) and hydrazine monohydrate (13.7 mL, 0.180 mol) is added. The resulting mixture is heated to reflux and water is removed using a Dean and Stark apparatus. After 3 hours the mixture is cooled to 90 °C and additional hydrazine monohydrate (13.7 mL; 0.180 mol) is added and the mixture is heated to reflux for 1 hour. The mixture is cooled and concentrated under reduced pressure. The resulting solid is triturated with water (200 mL), filtered and dried in a vacuum oven over P2Os at 60°C. The solid is triturated in iso-hexane (200 mL) and filtered to give the title compound (14.3 g; 77.1% yield). MS (m/z): 309/311 (M+l).
Preparation 4
4-(4-bromo-2-methylbenzyl)-5-(propan-2-yl)-lH-pyrazol-3-yl 2,3,4,6-tetra-O-benzoyl- beta-D-glucopyranoside
Scheme 1, step D: To a 1L flask, add 4-[(4-bromo-2-methyl-phenyl)methyl]-5- isopropyl-lH-pyrazol-3-ol (20 g, 64.7 mmol), alpha-D-glucopyranosyl bromide tetrabenzoate (50 g, 76 mmol), benzyltributylammonium chloride (6 g, 19.4 mmol), dichloromethane (500 mL), potassium carbonate (44.7 g, 323 mmol) and water (100 mL). Stir the reaction mixture overnight at room temperature. Extract with dichloromethane (500mL). Wash extract with water (300 mL) and brine (500 mL). Dry organic phase over sodium sulfate, filter, and concentrate under reduced pressure. Purify the residue by flash chromatography to yield the title compound (37 g, 64 mmol). MS (m/z): 889.2 (M+l), 887.2 (M-l).
Preparation 5
4- {4- [(lis)-4-hydroxybut- 1 -en- 1 -yl] -2-methylbenzyl } -5 -(propan-2-yl)- lH-pyrazol-3-yl
2,3,4,6-tetra-O-benzoyl-beta-D-glucopyranoside
Scheme 1, step E: Add 3-buten-l-ol (0.58 mL, 6.8 mmol) to a solution of 4-(4- bromo-2-methylbenzyl)-5 -(propan-2-yl)- lH-pyrazol-3 -yl 2,3 ,4,6-tetra-O-benzoyl-beta-D- glucopyranoside (3 g, 3.4 mmol) in acetonitrile (30 mL) and triethylamine (20 mL). Degas the solution with nitrogen over 10 minutes. Add tri-o-tolylphosphine (205 mg, 0.67 mmol) and palladium acetate (76 mg, 0.34 mmol). Reflux at 90 °C for 2 hours. Cool to room temperature and concentrate to remove the solvent under the reduced pressure. Purify the residue by flash chromatography to yield the title compound (2.1 g, 2.4 mmol). MS (m/z): 878.4 (M+l).
Preparation 6
4-{4-[(l£)-4-oxybut-l-en-l-yl]-2-methylbenzyl}-5-(propan-2-yl)-lH-pyrazol-3-yl
2,3,4,6-tetra-O-benzoyl-beta-D-glucopyranoside
Scheme 1, step F: Add 3,3,3-triacetoxy-3-iodophthalide (134 mg, 0.96 mmol) to a solution of 4-{4-[(l£)-4-hydroxybut-l-en-l-yl]-2-methylbenzyl}-5-(propan-2-yl)-lH- pyrazol-3-yl 2,3,4,6-tetra-O-benzoyl-beta-D-glucopyranoside (280 mg, 0.32 mmol) and sodium bicarbonate (133.8 mg, 1.6 mmol) in dichloromethane (20 mL) at 0 °C. After 15 minutes at room temperature, quench the reaction with saturated aqueous sodium thiosulfate (10 mL). Extract with dichloromethane (30 mL). Wash extract with water (30 mL) and brine (40 mL). Dry organic phase over sodium sulfate, filter, and concentrate under reduced pressure. Purify the resulting residue by flash chromatography to yield the title compound (270 mg, 0.31 mmol). MS (m/z): 876.5 (M+l), 874.5 (M-l).
Preparation 7
tert-butyl 2-{(3JE)-4-[3-methyl-4-({5-(propan-2-yl)-3-[(2,3,4,6-tetra-0-benzoyl-beta-D- glucopyranosyl)oxy]-lH-pyrazol-4-yl}methyl)phenyl]but-3-en-l-yl} -2,9- diazaspiro[5.5]undecane-9-carboxylate
Scheme 1, step G: Add sodium triacetoxyborohydride (98 mg, 0.46 mmol) to a solution of 4-{4-[(l£)-4-oxybut-l-en-l-yl]-2-methylbenzyl}-5-(propan-2-yl)-lH-pyrazol- 3-yl 2,3,4,6-tetra-O-benzoyl-beta-D-glucopyranoside (270 mg, 0.31 mmol) and tert-butyl 2,9-diazaspiro[5.5]undecane-9-carboxylate hydrochloride (179 mg, 0.62 mmol) in 1,2- dichloroethane (5 mL). After 30 minutes at room temperature, quench the reaction with saturated aqueous sodium bicarbonate (10 mL). Extract with dichloromethane (30 mL). Wash extract with water (30 mL) and brine (40 mL), dry organic phase over sodium sulfate, filter and concentrate under reduced pressure. Purify the resulting residue by flash chromatography to yield the title compound (275 mg, 0.25 mmol).
MS (m/z): 1115.6 (M+l).
Preparation 8
4- {4- [( l£)-4-(2,9-diazaspiro [5.5]undec-2-yl)but- 1 -en- 1 -yl] -2-methylbenzyl} -5-(propan- 2-yl)-lH-pyrazol-3-yl 2,3,4,6-tetra-O-benzoyl-beta-D-glucopyranoside dihydrochloride
Scheme 1, step H: Add hydrogen chloride (4.0 M solution in 1,4-dioxane, 0.6 mL, 2.4 mmol) to a solution of tert-butyl 2-{(3£)-4-[3-methyl-4-({5-(propan-2-yl)-3- [(2,3,4,6-tetra-0-benzoyl-beta-D-glucopyranosyl)oxy]-lH-pyrazol-4- yl}methyl)phenyl]but-3-en-l-yl}-2,9-diazaspiro[5.5]undecane-9-carboxylate (275 mg, 0.25 mmol) in dichloromethane (5 mL). After overnight (18 hours) at room temperature, concentrate to remove the solvent under reduced pressure to yield the title compound as a solid (258 mg, 0.24 mmol). MS (m/z): 1015.6 (M+l).
Preparation 9
4-(4-bromo-2-methylbenzyl)-5-(propan-2-yl)-lH-pyrazol-3-yl 2,3,4,6-tetra-O-acetyl- beta-D-glucopyranoside.
Scheme 2, step A: To a 1 L flask, add 4-[(4-bromo-2-methyl-phenyl)mefhyl]-5- isopropyl-lH-pyrazol-3-ol (24 g, 77.6 mmol), 2,3,4,6-tetra-O-acetyl-alpha-D- glucopyranosyl bromide (50.4 g, 116 mmol), benzyltributylammonium chloride (5 g, 15.5 mmol), dichloromethane (250 mL), potassium carbonate (32 g, 323 mmol) and water (120 mL). Stir the reaction mixture overnight at room temperature. Extract with dichloromethane (450 mL). Wash extract with water (300 mL) and brine (500 mL). Dry organic phase over sodium sulfate, filter, and concentrate under reduced pressure. Purify the resulting residue by flash chromatography to yield the title compound (36.5 g, 57 mmol). MS (m/z): 638.5 (M+l), 636.5 (M-l).
Alternative synthesis of 4-(4-bromo-2-methylbenzyl)-5-(propan-2-yl)-lH-pyrazol-3-yl
2,3,4,6-tetra-O-acetyl-beta-D-glucopyranoside.
Reagents 4-[(4-bromo-2-methyl-phenyl)methyl]-5-isopropyl-lH-pyrazol-3-ol (24.0 g, 77.6 mmol), 2,3,4,6-tetra-O-acetyl-alpha-D-glucopyranosyl bromide (50.4 g, 116 mmol), benzyltributylammonium chloride (4.94 g, 15.52 mmol), potassium carbonate (32.18 g, 232.9 mmol), dichloromethane (250 mL) and water (120 mL) are combined and the mixture is stirred at ambient temperature for 18 hours. The mixture is partitioned between dichloromethane (250 mL) and water (250 mL). The organic phase is washed with brine (250 mL), dried over Na2S04, filtered, and concentrated under reduced pressure. The resulting residue is purified by flash chromatography (eluting with 10% ethyl acetate in dichloromethane to 70% ethyl acetate in dichloromethane) to give the title compound (36.5 g, 74% yield). MS (m/z): 639/641 (M+l). Preparation 10
4- {4- [(lis)-4-hydroxybut- 1 -en- 1 -yl] -2-methylbenzyl } -5 -(propan-2-yl)- lH-pyrazol-3-yl
2,3,4,6-tetra-O-acetyl-beta-D-glucopyranoside
Scheme 2, step B: Add 3-buten-l-ol (6.1 mL, 70 mmol) to a solution of 4-(4- bromo-2-methylbenzyl)-5 -(propan-2-yl)- 1 H-pyrazol-3 -yl 2,3 ,4,6-tetra-O-acetyl-beta-D- glucopyranoside (15 g, 23.5 mmol) in acetonitrile (200 mL) and triethylamine (50 mL). Degas the solution with nitrogen over 10 minutes. Add tri-o-tolylphosphine (1.43 g, 4.7 mmol) and palladium acetate (526 mg, 2.35 mmol). After refluxing at 90 °C for 2 hours, cool, and concentrate to remove the solvent under the reduced pressure. Purify the resulting residue by flash chromatography to yield the title compound (7.5 g, 11.9 mmol) MS (m/z): 631.2 (M+l), 629.2 (M-l).
Preparation 11
4-{4-[(l£)-4-oxybut-l-en-l-yl]-2-methylbenzyl}-5-(propan-2-yl)-lH-pyrazol-3-yl
2,3,4,6-tetra-O-acetyl-beta-D-glucopyranoside
Scheme 2, step C: Add 3,3,3-triacetoxy-3-iodophthalide (2.1g, 4.76 mmol) to a solution of 4-{4-[(l£)-4-hydroxybut-l-en-l-yl]-2-methylbenzyl}-5-(propan-2-yl)-lH- pyrazol-3-yl 2,3,4,6-tetra-O-acetyl-beta-D-glucopyranoside ( 1.5 g, 2.38 mmol) and sodium bicarbonate (2 g, 23.8 mmol) in dichloromethane (50 mL) at 0 °C. After 15 minutes at room temperature, quench the reaction with saturated aqueous sodium thiosulfate (10 mL). Extract with dichloromethane (30 mL), wash extract with water (30 mL) and brine (40 mL). Dry organic phase over sodium sulfate, filter, and concentrate under reduced pressure. Purify the resulting residue by flash chromatography to yield the title compound (0.95 g, 1.51 mmol). MS (m/z): 628.8(M+1), 626.8 (M-l).
Preparation 12a
tert-butyl 2-{(3JE)-4-[3-methyl-4-({5-(propan-2-yl)-3-[(2,3,4,6-tetra-0-acetyl-beta-D- glucopyranosyl)oxy] -lH-pyrazol-4-yl}methyl)phenyl]but-3-en- 1 -yl} -2,9- diazaspiro[5.5]undecane-9-carboxylate
Scheme 2, Step D: Add sodium triacetoxyborohydride (303 mg, 1.4 mmol) to a solution of 4-{4-[(l£)-4-oxybut-l-en-l-yl]-2-methylbenzyl}-5-(propan-2-yl)-lH-pyrazol- 3-yl 2,3,4,6-tetra-O-acetyl-beta-D-glucopyranoside (600 mg, 0.95 mmol) and tert-butyl 2,9-diazaspiro[5.5]undecane-9-carboxylate hydrochloride (333 mg, 1.2 mmol) in 1,2- dichloroethane (30 mL). After 30 minutes at room temperature, quench the reaction with saturated aqueous sodium bicarbonate (15 mL). Extract with dichloromethane (60 mL). Wash extract with water (30 mL) and brine (60 mL). Dry organic phase over sodium sulfate, filter, and concentrate under reduced pressure. Purify the resulting residue by flash chromatography to yield the title compound (500 mg, 0.58 mmol).
MS (m/z): 866.8, 867.8 (M+l), 864.8, 865.8 (M-l).
Preparation 13
4- {4- [( l£)-4-(2,9-diazaspiro [5.5]undec-2-yl)but- 1 -en- 1 -yl] -2-methylbenzyl} -5-(propan- 2-yl)- lH-pyrazol-3-yl 2,3,4,6-tetra-O-acetyl-beta-D-glucopyranoside dihydrochloride
Scheme 2, step E: Add hydrogen chloride (4.0 M solution in 1,4-dioxane, 1.5 mL, 5.8 mmol) to a solution of tert-butyl 2-{(3£)-4-[3-methyl-4-({5-(propan-2-yl)-3-[(2,3,4,6- tetra-0-acetyl-beta-D-glucopyranosyl)oxy] – lH-pyrazol-4-yl} methyl)phenyl]but-3 -en- 1 – yl}-2,9-diazaspiro[5.5]undecane-9-carboxylate (500 mg, 0.58 mmol) in dichloromethane (20 mL). After 2 hours at room temperature, concentrate to remove the solvent under reduced pressure to yield the title compound as a solid (480 mg, 0.57 mmol).
MS (m/z): 767.4 (M+l).
Scheme 3
Preparation 14
tert-butyl 4-but-3-ynyl-4,9-diazas iro[5.5]undecane-9-carboxylate
Scheme 3, step A: Cesium carbonate (46.66 g, 143.21 mmol) is added to a suspension of tert-butyl 4,9-diazaspiro[5.5]undecane-9-carboxylate hydrochloride (16.66 g, 57.28 mmoles) in acetonitrile (167 mL). The mixture is stirred for 10 minutes at ambient temperature then 4-bromobutyne (6.45 mL, 68.74 mmol) is added. The reaction is heated to reflux and stirred for 18 hours. The mixture is cooled and concentrated under reduced pressure. The residue is partitioned between water (200 mL) and ethyl acetate (150 mL). The phases are separated and the aqueous layer is extracted with ethyl acetate (100 mL). The combined organic layers are washed with water (200 mL), then brine (150 mL), dried over MgS04, filtered, and concentrated under reduced pressure to give the title compound (17.2 g, 98% yield). lH NMR (300.11 MHz, CDC13): δ 3.43-3.31 (m, 4H), 2.53-2.48 (m, 2H), 2.37-2.29 (m, 4H), 2.20 (s, 2H), 1.94 (t, J= 2.6 Hz, 1H), 1.44 (s, 17H).
Preparation 15
tert-butyl 4-[(£)-4-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2-yl)but-3-enyl]-4,9- diazaspiro[5.5]undecane-9-carboxylate
Scheme 3, step B: Triethylamine (5.62 mmoles; 0.783 mL), 4,4,5,5-tetramethyl- 1,3,2-dioxaborolane (8.56 mL, 59.0 mmol) and zirconocene chloride (1.45 g, 5.62 mmoles) are added to tert-butyl 4-but-3-ynyl-4,9-diazaspiro[5.5]undecane-9-carboxylate (17.21 g, 56.16 mmoles). The resulting mixture is heated to 65 °C for 3.5 hours. The mixture is cooled and dissolved in dichloromethane (150 mL). The resulting solution is passed through a ~4cm thick pad of silica gel, eluting with dichloromethane (2 x 200 mL). The filtrate is concentrated under reduced pressure to give the title compound (21.2 g, 87% yield). 1H NMR (300.11 MHz, CDCI3): δ 6.65-6.55 (m, 1H), 5.49-5.43 (m, 1H), 3.42-3.29 (m, 4H), 2.40-2.27 (m, 6H), 2.25-2.08 (m, 2H), 1.70 – 1.13 (m, 29H).
Preparation 16
tert-butyl 2-{(3£’)-4-[3-methyl-4-({5-(propan-2-yl)-3-beta-D-glucopyranosyl)oxy]-lH- pyrazol-4-yl} methyl)phenyl]but-3 -en- 1 -yl} -2,9-diazaspiro [5.5]undecane-9-carboxylate
Scheme 3, step C: A solution of 4-(4-bromo-2-methylbenzyl)-5-(propan-2-yl)- lH-pyrazol-3-yl 2,3,4,6-tetra-O-acetyl-beta-D-glucopyranoside (20 g, 31.3 mmol), tert- butyl 4-[(£)-4-(4,4,5 ,5 -tetramethyl- 1 ,3,2-dioxaborolan-2-yl)but-3 -enyl] -4,9- diazaspiro[5.5]undecane-9-carboxylate (16.3 g, 37.5 mmol) and potassium carbonate (12.97 g, 93.82 mmol) in tetrahydrofuran (200 mL) and water (40 mL) is degassed for 15 min by bubbling nitrogen gas through it. Pd(OAc)2 (140 mg, 625 μιηοΐ) and 2- dicyclohexylphosphino-2′,4′,6′-tri-i -propyl- Ι, -biphenyl (0.596 g, 1.25 mmol) are added and the reaction is heated to reflux for 16 h. The solution is cooled to ambient temperature and methanol (200 mL) is added. After 30 minutes the solvent is removed under reduced pressure. The mixture is partitioned between ethyl acetate (500 mL) and brine (500 ml) adding aqueous MgS04 (1M; 500 ml) to aid the phase separation. The layers are separated and the organic layer is dried over MgS04 and filtered through a 10 cm pad of silica gel, eluting with ethyl acetate (-1.5 L). The filtrate is discarded and the silica pad is flushed with 5% MeOH in THF (2 L). The methanolic filtrate is concentrated under reduced pressure to give the title compound (20. lg, 92%).
MS (m/z): 699 (M+l).
Preparation 17
tert-butyl 4- [(E)-4- [4- [(3 -hydroxy-5-isopropyl- 1 H-pyrazol-4-yl)methyl] -3 -methyl- phenyl]but-3-enyl]-4,9-diazaspiro[5.5]undecane-9-carboxylate
Scheme 4, step A: Add tert-butyl 4-[(£)-4-(4,4,5,5-tetramethyl-l,3,2- dioxaborolan-2-yl)but-3-enyl]-4,9-diazaspiro[5.5]undecane-9-carboxylate (35.8 kg, 82.4 mol) in methanol (130 L) to a solution of (4-[(4-bromo-2-methyl-phenyl)methyl]-5- isopropyl-lH-pyrazol-3-ol (23.9 kg, 77.3 mol) in methanol (440 L) at room temperature. Add water (590 L) and tripotassium phosphate (100 kg, 471.7 mol) and place the reaction under nitrogen atmosphere. To the stirring solution, add a suspension of
tris(dibenzylideneacetone) dipalladium (1.42 kg, 1.55 mol) and di-tert- butylmethylphosphonium tetrafluoroborate (775 g, 3.12 mol) in methanol (15 L). The resulting mixture is heated at 75 °C for 2 hours. Cool the mixture and filter over diatomaceous earth. Rinse the the filter cake with methanol (60 L), and concentrate the filtrate under reduced pressure. Add ethyl acetate (300 L), separate the layers, and wash the organic layer with 15% brine (3 x 120 L). Concentrate the organic layer under reduced pressure, add ethyl acetate (300 L), and stir the mixture for 18 to 20 hours. Add heptane (300 L), cool the mixture to 10 °C, and stir the mixture for an additional 18 to 20 hours. Collect the resulting solids by filtration, rinse the cake with ethyl acetate/heptane (2:3, 2 x 90 L), and dry under vacuum at 40°C to give the title compound (29.3 kg, 70.6% yield) as a white solid. lH NMR (400 MHz, CD3OD): δ 7.14 (s, 1H), 7.07 (d, J= 8.0 Hz, 1H), 6.92 (d, J= 7.6 Hz, 1H), 6.39 (d, J= 16.0 Hz, 1H), 6.25-6.12 (m, 1H), 3.63 (s, 2H), 3.45-3.38 (bs, 3H), 3.34 (s, 3 H), 3.33 (s, 3H), 2.85-2.75 (m, 1H), 2.49-2.40 (m, 5 H), 2.33 (s, 3H), 1.68-1.62 (m, 2H), 1.60-1.36 (m, 15H), 1.11 (s, 3H), 1.10 (s, 3H).
Preparation 12b
Alterternative preparation of tert-butyl 2-{(3£)-4-[3-methyl-4-({5-(propan-2-yl)-3- [(2,3,4,6-tetra-0-acetyl-beta-D-glucopyranosyl)oxy]-lH-pyrazol-4-yl}methyl)phenyl]but- 3-en-l-yl}-2,9-diazaspiro[5.5]undecane-9-carboxylate.
Scheme 4, step B: Combine tert-butyl 4-[(E)-4-[4-[(3-hydroxy-5-isopropyl-lH- pyrazol-4-yl)methyl] -3-methyl-phenyl]but-3 -enyl] -4,9-diazaspiro [5.5]undecane-9- carboxylate (17.83 kg, 33.2 moles), acetonitrile (180 L), and benzyltributylammonium chloride (1.52 kg, 4.87 moles) at room temperature. Slowly add potassium carbonate (27.6 kg, 199.7 moles) and stir the mixture for 2 hours. Add 2,3,4,6-tetra-O-acetyl-alpha- D-glucopyranosyl bromide (24.9 kg, 60.55 mol), warm the reaction mixture to 30°C and stir for 18 hours. Concentrate the mixture under reduced pressure and add ethyl acetate (180 L), followed by water (90 L). Separate the layers, wash the organic phase with 15% brine (3 x 90 L), concentrate the mixture, and purify using column chromatography over silica gel (63 kg, ethyl acetate/heptanes as eluent (1 :2→1 :0)) to provide the title compound (19.8 kg, 94% purity, 68.8% yield) as a yellow foam, !H NMR (400 MHz, CDC13): δ 7.13 (s, 1H), 7.03 (d, J= 8.0 Hz, 1H), 6.78 (d, J= 8.0 Hz, 1H), 6.36 (d, J= 16.0,
1H), 6.25-6.13 (m, 1H), 5.64 (d, J= 8.0 Hz, 1H), 5.45-5.25 (m, 2H), 5.13-4.95 (m, 2H), 4.84-4.76 (m, 1H), 4.25-4.13 (m, 2H), 4.10-4.00 (m, 2H), 3.90-3.86 (m, 1H), 3.58-3.50 (m, 2H), 3.40-3.22 (m, 4H), 2.89-2.79 (m, 1H), 2.10-1.90 (m, 18 H), 1.82 (s, 3H), 1.62- 0.82 (m, 22H).
Preparation 18
2-{(3JE)-4-[3-methyl-4-({5-(propan-2-yl)-3-[(2,3,4,6-tetra-0-acetyl-beta-D- glucopyranosyl)oxy]-lH-pyrazol-4-yl}methyl)phenyl]but-3-en-l-yl} -2,9- diazaspiro[5.5]undecane
Scheme 4, step C: Combine tert-butyl 2-{(3JE)-4-[3-methyl-4-({5-(propan-2-yl)- 3-[(2,3,4,6-tetra-0-acetyl-beta-D-glucopyranosyl)oxy]-lH-pyrazol-4- yl}methyl)phenyl]but-3-en-l-yl}-2,9-diazaspiro[5.5]undecane-9-carboxylate (19.6 kg, 22.6 moles) with dichloromethane (120 L) and cool to 0°C. Slowly add trifluoroacetic acid (34.6 L, 51.6 kg, 452 moles) and stir for 9 hours. Quench the reaction with ice water (80 L), and add ammonium hydroxide (85-90 L) to adjust the reaction mixture to pH (8- 9). Add dichloromethane (120 L), warm the reaction mixture to room temperature, and separate the layers. Wash the organic layer with water (75 L), brine, and concentrate under reduced pressure to provide the title compound (16.2 kg, 95.0% purity, 93% yield) as a yellow solid. lH NMR (400 MHz, CDC13): δ 7.08 (s, IH), 6.99 (d, J= 8.0 Hz, IH),
6.76 (d, J= 7.6 Hz, IH), 6.38 (d, J=15.6 Hz, IH), 6.00-5.83 (m, IH), 5.31 (d, J= 7.6 Hz, IH), 5.25-5.13 (m, 4H), 4.32 (dd, J= 12.8, 9.2 Hz, IH), 4.14 (d, J= 11.2 Hz, IH), 3.90 (d, J= 10.0 Hz, IH), 3.75-3.50 (m, 3H), 3.30-3.00 (m, 5 H), 2.85-2.75 (m, IH), 2.70-2.48 (m, 3H), 2.25 (s, IH), 2.13-1.63 (m, 19H), 1.32-1.21 (m, IH), 1.14 (s, 3H), 1.13 (s, 3H), 1.12 (s, 3H), 1.10 (s, 3H).
Example 1
Hydrated crystalline 4- {4-[(l£)-4-(2,9-diazaspiro[5.5]undec-2-yl)but- 1 -en- 1 -yl]-2- methylbenzyl} -5-(propan-2-yl)-lH-pyrazol-3-yl beta-D-glucopyranoside acetate
First alternative preparation of 4-{4-[(l£’)-4-(2.9-diazaspiro[5.5]undec-2-yl)but-l-en-l- yl]-2-methylbenzyl| -5-(propan-2-yl)-lH-pyrazol-3-yl beta-D-glucopyranoside (free base).
Scheme 1, step I: Add sodium hydroxide (0.5 mL, 0.5 mmol, 1.0 M solution) to a solution of 4- {4-[( l£)-4-(2,9-diazaspiro [5.5]undec-2-yl)but- 1 -en- 1 -yl] -2-methylbenzyl} – 5-(propan-2-yl)-lH-pyrazol-3-yl 2,3,4,6-tetra-O-benzoyl-beta-D-glucopyranoside dihydrochloride (258 mg, 0.24 mmol) in methanol (2 mL). After 2 hours at 40°C, concentrate to remove the solvent under reduced pressure to give a residue, which is purified by preparative HPLC method: high pH, 25% B for 4 min, 25-40 B % for 4 min @ 85 mL/min using a 30 x 75 mm, 5 μιη C18XBridge ODB column, solvent A – H.0 with NH4HCO3 @ pH 10, solvent B – MeCN to yield the title compound (free base) as a solid (46 mg, 0.08 mmol). MS (m/z): 598.8 (M+l), 596.8 (M-l).
Second alternative preparation of 4-{4-r(l-£’)-4-(2.9-diazaspiror5.51undec-2-yl)but-l-en- 1 -yl] -2-methylbenzyl I -5 -(propan-2-yl)- lH-pyrazol-3 -yl beta-D-glucopyranoside (free base“).
Scheme 2, step F: Add methanol (5 mL), triethylamine (3 mL), and water (3 mL) to 4- {4-[( lJE)-4-(2,9-diazaspiro [5.5]undec-2-yl)but- 1 -en- 1 -yl] -2-methylbenzyl } -5 – (propan-2-yl)-lH-pyrazol-3-yl 2,3,4,6-tetra-O-acetyl-beta-D-glucopyranoside dihydrochloride (480 mg, 0.24 mmol). After 18 hours (overnight) at room temperature, concentrate to dryness under reduced pressure. Purify the resulting residue by preparative HPLC method: high pH, 25% B for 4 min, 25-40 B % for 4 min @ 85 mL/min using a 30 x 75 mm, 5 μιη C18XBridge ODB column, solvent A – H20 with NH4HCO3 @ pH 10, solvent B – MeCN to yield the title compound (free base) as a solid (50 mg, 0.08 mmol).
MS (m/z): 598.8 (M+l), 596.8 (M-l). 1H NMR (400.31 MHz, CD3OD): δ 7.11 (d, J=1.3
Hz, 1H), 7.04 (dd, J=l .3,8.0 Hz, 1H), 6.87 (d, J= 8.0 Hz, 1H), 6.36 (d, J= 15.8 Hz, 1H), 6.16 (dt, J= 15.8, 6.3 Hz, 1H), 5.02 (m, 1H), 3.81 (d, J= 11.7 Hz, 1H), 3.72 (d, J= 16.8 Hz, 1H), 3.68 (d, J= 16.8 Hz, 1H) , 3.64 (m, 1H), 3.37-3.29 (m, 4H), 2.79 (m, 1H), 2.72 (t, J= 5.8 Hz, 4H), 2.44-2.33 (m, 6H), 2.30 (s, 3H), 2.26 ( broad s, 2H), 1.59 (m, 2H), 1.50 (m, 2H), 1.43 (m, 2H), 1.36 (m, 2H), 1.11 (d, J= 7.0 Hz, 3H), 1.10 (d, J= 7.0 Hz, 3H).
Third alternative preparation of 4-{4-[(l£,)-4-(2,9-diazaspiro[5.51undec-2-yl)but-l-en-l- yll-2-methylbenzyl|-5-(propan-2-yl)-lH-pyrazol-3-yl beta-D-glucopyranoside.
Scheme 3, step D: Trifluoroacetic acid (32.2 mL; 0.426 mol) is added to a solution of tert-butyl 2-{(3JE)-4-[3-methyl-4-({5-(propan-2-yl)-3-beta-D- glucopyranosyl)oxy]-lH-pyrazol-4-yl}methyl)phenyl]but-3-en-l-yl}-2,9- diazaspiro[5.5]undecane-9-carboxylate (14.87 g; 21.28 mmol) in dichloromethane (149 mL) cooled in iced water. The solution is allowed to warm to room temperature. After 30 minutes, the mixture is slowly added to ammonia in MeOH (2M; 300 mL), applying cooling as necessary to maintain a constant temperature. The solution is stirred at room temperature for 15 min. The mixture is concentrated under reduced pressure and the residue is purified using SCX-2 resin. The basic filtrate is concentrated under reduced pressure and the residue is triturated/sonicated in ethyl acetate, filtered and dried. The resulting solid is dissolved in MeOH (200mL) and concentrated in vacuo. This is repeated several times to give the title compound (free base) (12.22 g, yield 96%). MS (m/z): 599 (M+l); [a]D 20 = -12 ° (C=0.2, MeOH).
Preparation of final title compound, hydrated crystalline 4-{4-|YlE)-4-(2.9- diazaspiro [5.5“|undec-2-yl)but- 1 -en- 1 -yl] -2-methylbenzyl I -5-(propan-2-vD- 1 H-pyrazol-3 – yl beta-D-glucopyranoside acetate.
4- {4- [(1 E)-4-(2,9-diazaspiro [5.5]undec-2-yl)but- 1 -en- 1 -yl] -2-methylbenzyl } -5 – (propan-2-yl)-lH-pyrazol-3-yl beta-D-glucopyranoside (902 mg) is placed in a round bottom flask (100 mL) and treated with wet ethyl acetate (18 mL). [Note – wet ethyl acetate is prepared by mixing ethyl acetate (100 mL) and dionized water (100 mL). After mixing, the layers are allowed to separate, and the top wet ethyl acetate layer is removed for use. Acetic acid is a hydrolysis product of ethyl acetate and is present in wet ethyl acetate.] The compound dissolves, although not completely as wet ethyl acetate is added. After several minutes, a white precipitate forms. An additional amount of wet ethyl acetate (2 mL) is added to dissolve remaining compound. The solution is allowed to stir uncovered overnight at room temperature during which time the solvent partially evaporates. The remaining solvent from the product slurry is removed under vacuum, and the resulting solid is dried under a stream of nitrogen to provide the final title compound as a crystalline solid. A small amount of amorphous material is identified in the product by solid-state NMR. This crystalline final title compound may be used as seed crystals to prepare additional crystalline final title compound.
Alternative preparation of final title compound, hvdrated crystalline 4-{4-[(lE)-4-(2.,9- diazaspiro [5.5]undec-2-yl)but- 1 -en- 1 -yl] -2-methylbenzyl I -5-(propan-2-yl)- 1 H-pyrazol-3 – yl beta-D-glucopyranoside acetate.
Under a nitrogen atmosphere combine of 4-{4-[(lE)-4-(2,9-diazaspiro[5.5]undec- 2-yl)but- 1 -en- 1 -yl] -2-methylbenzyl} -5-(propan-2-yl)- 1 H-pyrazol-3-yl 2,3,4,6-tetra-O- acetyl-beta-D-glucopyranoside (2.1 kg, 2.74 mol), methanol (4.4 L), tetrahydrofuran (4.2 L), and water (210 mL). Add potassium carbonate (460 g, 3.33 moles) and stir for four to six hours, then filter the reaction mixture to remove the solids. Concentrate the filtrate under reduced pressure, then add ethanol (9.0 L) followed by acetic acid (237 mL, 4.13 mol) and stir at room temperature for one hour. To the stirring solution add wet ethyl acetate (10 L, containing approx. 3 w/w% water) slowly over five hours, followed by water (500 mL). Stir the suspension for twelve hours and add wet ethyl acetate (4.95 L, containing approx. 3 w/w% water) over a period of eight hours. Stir the suspension for twelve hours and add additional wet ethyl acetate (11.5 L, containing approx. 3 w/w% water) slowly over sixteen hours. Stir the suspension for twelve hours, collect the solids by filtration and rinse the solids with wet ethyl acetate (3.3 L, containing approx. 3 w/w% water). Dry in an oven under reduced pressure below 30°C to give the title compound as an off-white crystalline solid (1.55 kg, 2.35 mol, 96.7% purity, 72.4 w/w% potency, 68.0% yield based on potency). HRMS (m/z): 599.3798 (M+l).
PATENT
CN105705509
https://patentscope.wipo.int/search/en/detail.jsf?docId=CN175101669&tab=PCTDESCRIPTION
The present invention is in the field of treatment of diabetes and other diseases and conditions associated with hyperglycemia. Diabetes is a group of diseases characterized by high blood sugar levels. It affects approximately 25 million people in the United States, and according to the 2011 National Diabetes Bulletin, it is also the seventh leading cause of death in the United States (US Department of Health and Human Resources Services, Centers for Disease Control and Prevention). Sodium-coupled glucose cotransporters (SGLT’s) are one of the transporters known to be responsible for the uptake of carbohydrates such as glucose. More specifically, SGLT1 is responsible for transporting glucose across the brush border membrane of the small intestine. Inhibition of SGLT1 can result in a decrease in glucose absorption in the small intestine, thus providing a useful method of treating diabetes.
Alternative medicines and treatments for diabetes are needed. The present invention provides an acetate salt of a pyrazole compound which is an SGLT1 inhibitor, and thus it is suitable for treating certain conditions such as diabetes.
U.S. Patent No. 7,655,632 discloses certain pyrazole derivatives having human SGLT1 inhibitory activity, which are also disclosed for use in the prevention or treatment of diseases associated with hyperglycemia, such as diabetes. Moreover, WO 2011/039338 discloses certain pyrazole derivatives having SGLT1/SGLT2 inhibitor activity, which are also disclosed for use in the treatment of bone diseases such as osteoporosis.
PATENT
WO-2019141209
Process for preparing pyranoglucose-substituted pyrazole compound, used as a pharmaceutical intermediate in SGLT inhibitor for treating diabetes.

| Patent ID | Title | Submitted Date | Granted Date |
|---|---|---|---|
| US9573970 | 4–5-(PROPAN-2-YL)-1H-PYRAZOL-3-YL BETA-D GLUCOPYRANOSIDE ACETATE | 2014-10-30 | 2016-07-28 |
/////////////SY-008 , SY 008 , SY008, ELI LILY, PHASE 1, GLT1 inhibitor, type 2 diabetes, Yabao Pharmaceutical, CHINA, DIABETES
CC(=O)O.Cc5cc(\C=C\CCN2CCCC1(CCNCC1)C2)ccc5Cc3c(nnc3C(C)C)O[C@@H]4O[C@H](CO)[C@@H](O)[C@H](O)[C@H]4O
Cc5cc(\C=C\CCN2CCCC1(CCNCC1)C2)ccc5Cc3c(nnc3C(C)C)O[C@@H]4O[C@H](CO)[C@@H](O)[C@H](O)[C@H]4
O
Fasoracetam
Fasoracetam
- Molecular FormulaC10H16N2O2
- Average mass196.246 Da
Fasoracetam is a research chemical of the racetam family.[3] It is a putative nootropic that failed to show sufficient efficacy in clinical trials for vascular dementia. It is currently being studied for its potential use for attention deficit hyperactivity disorder.[2][4]
Fasoracetam appears to agonize all three groups of metabotropic glutamate receptors and has improved cognitive function in rodent studies.[5] It is orally bioavailable and is excreted mostly unchanged via the urine.[6]
Fasoracetam was discovered by scientists at the Japanese pharmaceutical company Nippon Shinyaku, which brought it through Phase 3 clinical trials for vascular dementia, and abandoned it due to lack of efficacy.[5][7]
Scientists at Children’s Hospital of Philadelphia led by Hakon Hakonarson have studied fasoracetam’s potential use in attention deficit hyperactivity disorder.[5] Hakonarson started a company called neuroFix Therapeutics to try to bring the drug to market for this use; neuroFix acquired Nippon Shinyaku’s clinical data as part of its efforts.[7][8] neuroFix was acquired by Medgenics in 2015.[8] Medgenics changed its name to Aevi Genomic Medicine in 2016.[9] Clinical trials in adolescents with ADHD who also have mGluR mutations started in 2016.[8]

SYN
Chemistry – A European Journal, 24(27), 7033-7043; 2018
PAPER
Chemistry – A European Journal (2018), 24, (27), 7033-7043
https://onlinelibrary.wiley.com/doi/full/10.1002/chem.201800372
Amidation of unprotected amino acids has been investigated using a variety of ‘classical“ coupling reagents, stoichiometric or catalytic group(IV) metal salts, and boron Lewis acids. The scope of the reaction was explored through the attempted synthesis of amides derived from twenty natural, and several unnatural, amino acids, as well as a wide selection of primary and secondary amines. The study also examines the synthesis of medicinally relevant compounds, and the scalability of this direct amidation approach. Finally, we provide insight into the chemoselectivity observed in these reactions.
Patent
WO-2019143829
Novel crystalline forms of fasoracetam , processes for their preparation and compositions comprising them are claimed.
PATENT
WO2019143824
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2019143824&_cid=P10-JYNTH3-69052-1
Novel crystalline and cocrystal forms of fasoracetam (R-fasoracetam) and a co-former, processes for their preparation and compositions comprising them are claimed. Also claims are novel crystalline forms of fasoracetam and 4-aminobenzoic acid or trimesic acid or R- ibuprofen or phloroglucinol or methyl-3,4-5-trihydroxybenzoate or ethyl gallate or phthalic acid or 6-hydroxy-2-napthoic acid or 4-nitrobenzoic acid or 2-indole-3-acetic acid or urea and their monohydrate and dihydrate (designated as Form B).
PATENT
WO2018195184
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2018195184&_cid=P10-JYNTI8-69210-1
claiming methods for diagnosing and treating ADHD in biomarker positive subjects, assigned to Aevi Genomics Medicine, Inc and The Childrens Hospital of Philadelphia , naming different teams.
PAPER
https://advances.sciencemag.org/content/3/9/e1701028

References
- ^ FDA/NIH Substance registration system. Page accessed March 21, 2016
- ^ Jump up to:a b “Drug Profile Fasoracetam”.
- ^ “5-oxo-D-prolinepiperidinamide monohydrate – Compound Summary”. Retrieved 21 July 2013.
- ^ “Recommended INN List 40” (PDF). WHO Drug Information. 12 (2). 1998.
- ^ Jump up to:a b c Connolly, J; Glessner, J; Kao, C; Elia, J; Hakonarson, H. “ADHD & Pharmacotherapy: Past, Present and Future: A Review of the Changing Landscape of Drug Therapy for Attention Deficit Hyperactivity Disorder”. Ther Innov Regul Sci. 49 (5): 632–642. doi:10.1177/2168479015599811. PMC 4564067. PMID 26366330.
- ^ Malykh, AG; Sadaie, MR (12 February 2010). “Piracetam and piracetam-like drugs: from basic science to novel clinical applications to CNS disorders”. Drugs. 70 (3): 287–312. doi:10.2165/11319230-000000000-00000. PMID 20166767.
- ^ Jump up to:a b Moskowitz, D. H. (2017). Finding the Genetic Cause and Therapy for ADHD, Autism and 22q. BookBaby (self published). ISBN 9781483590981.
- ^ Jump up to:a b c Sharma, B. “Medgenics: NFC-1 Could Be A Key Future Revenue Driver”.
- ^ “Press Release: Medgenics, Inc. Announces Name Change to Aevi Genomic Medicine, Inc”. Aevi via MarketWired. 16 December 2016.
|
|
|||
| Names | |||
|---|---|---|---|
| IUPAC name
(5R)-5-(Piperidine-1-carbonyl)pyrrolidin-2-one
|
|||
| Other names | |||
| Identifiers | |||
|
3D model (JSmol)
|
|||
| ChEMBL | |||
| ChemSpider | |||
| KEGG | |||
|
PubChem CID
|
|||
|
CompTox Dashboard (EPA)
|
|||
| Properties | |||
| C10H16N2O2 | |||
| Molar mass | 196.250 g·mol−1 | ||
| Pharmacology | |||
| Oral | |||
| Legal status |
|
||
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|||
/////////Fasoracetam, attention deficit hyperactivity disorder, NS 105, Phase 3, vascular dementia
C1CCN(CC1)C(=O)[C@H]2CCC(=O)N2
BQ-788
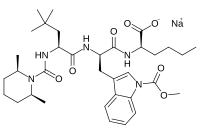
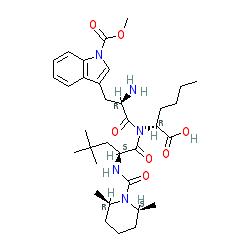
BQ-788
- Molecular FormulaC34H50N5NaO7
- Average mass663.780 Da
SP ROT +3.8 ° Conc: 1.032 g/100mL; methanol; Wavlenght: 589.3 nm, Development of an efficient strategy for the synthesis of the ETB receptor antagonist BQ-788 and some related analogues
Peptides (New York, NY, United States) (2005), 26, (8), 1441-1453., https://doi.org/10.1016/j.peptides.2005.03.022
FOR FREE FORM +19.6 °, Conc: 0.998 g/100mL; : N,N-dimethylformamide; 589.3 nm
BQ-788 is a selective ETB antagonist.[1]
presumed to be under license from Banyu , was investigating BQ-788, a selective endothelin receptor B (ETRB) antagonist, for treating metastatic melanoma. By December 2009, the drug was in validation.
Also claimed is their use as an ETBR antagonist and for treating cancers, such as brain cancer, pancreas cancer, colon cancer, breast cancer, ovary cancer, prostate cancer, glioblastoma, solid tumor, melanoma and squamous cell carcinoma. Represent a first filing from ENB Therapeutics Inc and the inventors on these deuterated forms of BQ-788. Melcure SarL ,

SYN
By Brosseau, Jean-Philippe et alFrom Peptides (New York, NY, United States), 26(8), 1441-1453; 2005
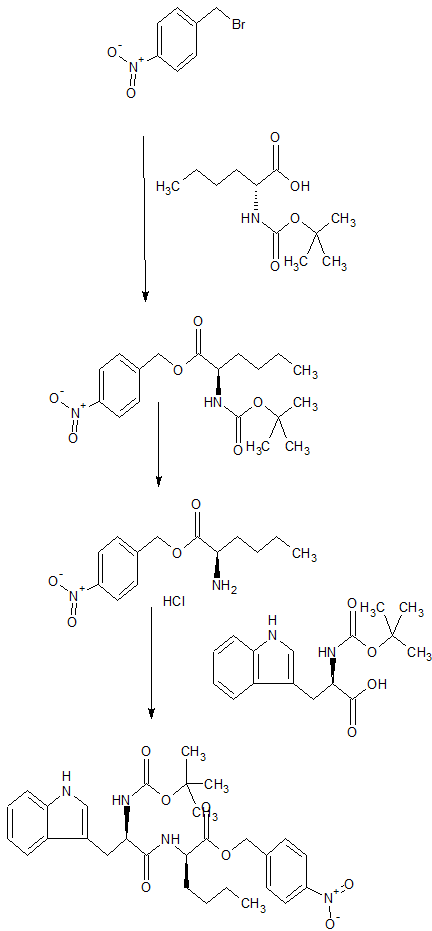
CONTD…………

PAPER
https://pubs.acs.org/doi/pdf/10.1021/jo00130a028

N-(cw-2,6-Dimethylpiperidinocarbonyl)-y-methylleucylD-l-(methoxycarbonyl)tryptophanyl-D-norleucine Sodium Salt (1, BQ-788). To a solution of 15 (3.5 g, 5.5 mmol) in methanol (50 mL) was slowly added 5% aqueous NaHCOs (300 mL) over a period of 30 min. The solution was stirred until clarity was achieved (30 min, 23 °C). The solution was diluted with water (200 mL), and the resulting solution was passed through a C18 (60 mL) cartridge preequilbrated in water. BQ-788 (1) was eluted with methanol (2 x 50 mL), concentrated under reduced pressure, resuspended in water (50 mL), and lyophilized to quantitatively yield compound 1 as a white powder:
HPLC £r = 16.4 (gradient A, > 99%);
NMR (400 MHz, DMSO-d6) ó 0.80 (s, 9H), 0.74-0.85 (m, 3H), 1.00 (d, 3H), 1.02 (d, 3H), 1.10-1.25 (m, 6H), 1.30-1.55 (m, 6H), 1.60-1.75 (m, 2H), 2.92 (dd, 1H), 3.12 (dd, 1H), 3.78 (m, 1H), 3.95 (s, 3H), 4.08 (m, 1H), 4.13 (m, 1H), 4.29 (m, 1H), 4.50 (m, 1H), 5.98 (d, 1H), 7.22 (t, 1H), 7.32 (t, 1H), 7.50 (s, 1H), 7.58 (br d, 1H), 7.65 (d, 1H), 8.05 (d, 1H), 8.15 (br d, 1H) ESMS m/z 640.6 (M).
PATENT
WO-2019140324
Novel deuterated analogs of a substituted heterocyclic compound, particularly BQ-788 , processes for their preparation and compositions and combinations comprising them are claimed.
PAPER
https://www.sciencedirect.com/science/article/abs/pii/S0196978105001415

PAPER
By He, John X.; Cody, Wayne L.; Doherty, Annette M., From Journal of Organic Chemistry (1995), 60(25), 8262-6
Journal of medicinal chemistry (1996), 39(12), 2313-30.
References
 |
|
| Names | |
|---|---|
| Systematic IUPAC name
Sodium N-{[(2R,6S)-2,6-dimethyl-1-piperidinyl]carbonyl}-4-methyl-L-leucyl-N-[(1R)-1-carboxylatopentyl]-1-(methoxycarbonyl)-D-tryptophanamide
|
|
| Identifiers | |
|
3D model (JSmol)
|
|
| ChemSpider | |
|
PubChem CID
|
|
|
CompTox Dashboard (EPA)
|
|
| Properties | |
| C34H50N5NaO7 | |
| Molar mass | 663.792 g·mol−1 |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
///////////BQ-788, BQ 788, BQ788, ETBR antagonist, cancers, brain cancer, pancreas cancer, colon cancer, breast cancer, ovary cancer, prostate cancer, glioblastoma, solid tumor, melanoma, squamous cell carcinoma, PEPTIDE
CCCC[C@H](C(=O)O)NC(=O)[C@@H](Cc1cn(c2c1cccc2)C(=O)OC)NC(=O)[C@H](CC(C)(C)C)NC(=O)N3[C@@H](CCC[C@@H]3C)C
Fluazolepali, 氟唑帕利 , Fluzoparib

Fluazolepali
CAS 2170504-09-1
Fluzoparib; SHR-3162, (HS10160)
- HS 10160
- SHR 3162
An orally available inhibitor of poly(ADP-ribose) polymerase 1 and 2 (PARP-1/2) for treatment of solid tumors (Jiangsu Hengrui Medicine Co. Ltd., Lianyungang, China)
Fluazolepali, developed by Hengrui and Howson, is intended for the treatment of recurrent ovarian cancer, triple-negative breast cancer, advanced gastric cancer and other advanced solid tumors. Currently, the drug has been introduced into China for recurrent ovarian cancer. Clinical stage.
In February 2019, a randomized, double-blind, controlled, multicenter, phase III clinical study (CTR20190294) of flazopril capsule versus placebo for maintenance of recurrent ovarian cancer was initiated in China and was sponsored by Hengrui Medicine.
Jiangsu Hansoh Pharmaceutical , in collaboration with Jiangsu Hengrui Medicine , is developing an oral capsule formulation of fluazolepali (fluzoparib; SHR-3162), a small molecule inhibitor to PARP-1 and PARP-2, for the treatment of solid tumors including epithelial ovarian, fallopian tube or primary peritoneal, breast and gastric cancer.
- Originator Jiangsu Hengrui Medicine Co.
- Class Antineoplastics
- Mechanism of Action Poly(ADP-ribose) polymerase 1 inhibitors; Poly(ADP-ribose) polymerase 2 inhibitors
- Phase II Ovarian cancer
- Phase I Breast cancer; Fallopian tube cancer; Gastric cancer; Peritoneal cancer; Solid tumours
- 09 Jul 2019 Jiangsu HengRui Medicine initiates a phase I trial in Solid tumors in China (NCT04013048) [14C]-Fluzoparib
- 01 Jul 2019 Jiangsu HengRui Medicine plans a phase I drug-drug interaction trial (In volunteers) in China (PO) (NCT04011124)
- 12 Jun 2019 Jiangsu HengRui Medicine completes a phase I trial in Gastric cancer (Combination therapy, Recurrent, Metastatic disease, Second-line therapy or greater, Late-stage disease) in China (PO) (NCT03026881)
Fluzoparib (SHR 3162) is a selective poly [ADP-ribose] polymerase 1 (PARP1) and poly [ADP-ribose] polymerase 2 inhibitor (PARP2), being developed by Jiangsu HengRui Medicine, for the treatment of cancer. PARP enzymes play a vital role in repair of DNA damage and maintaining genomic stability. Fluzoparib inhibits PARP enzymes and induces DNA-double strands breaks, G2/M arrest and apoptosis in homologous recombination repair (HR)-deficient cells. Clinical development for ovarian cancer, breast cancer, fallopian tube cancer, peritoneal cancer, gastric cancer and solid tumours is underway in China and Australia.
An orally available inhibitor of poly (ADP-ribose) polymerase (PARP) types 1 and 2, with potential antineoplastic activity. Upon oral administration, fluzoparib inhibits PARP 1 and 2 activity, which inhibits PARP-mediated repair of damaged DNA via the base excision repair (BER) pathway, enhances the accumulation of DNA strand breaks, promotes genomic instability, and leads to an induction of apoptosis. The PARP family of proteins catalyze post-translational ADP-ribosylation of nuclear proteins, which then transduce signals to recruit other proteins to repair damaged DNA. PARP inhibition may enhance the cytotoxicity of DNA-damaging agents and may reverse tumor cell chemoresistance and radioresistance. Check for active clinical trials using this agent. (NCI Thesaurus)
PATENT
WO-2019137358
Process for preparing heterocyclic compounds (presumed to be fluazolepali ) and its intermediates as PARP inhibitors useful for treating cancer.


PATENT
WO2019109938
claiming synergistic combination comprising PARP inhibitor fluazolepali and apatinib mesylate .
PATENT
WO 2018005818
WO 2018129553
WO 2018129559
WO 2018208968
WO 2018213732
WO 2018191277
WO 2018201096
WO 2018085469
WO 2018085468
WO 2019090227
WO 2019133697
WO 2019067978
WO 2019071123
WO 2019090141
///////////Fluazolepali, Jiangsu Hansoh Pharmaceutical, Jiangsu Hengrui Medicine, fluzoparib, SHR-3162, 氟唑帕利 , Phase II, Ovarian cancer, HS10160, CHINA, HS 10160
WXFL-10203614
WXFL-10203614
CAS 2054932-34-0 R isomer, (S isomer 2054932-33-9 )
C15 H15 N7, 293.33
(7R)-7-[methyl(7H-pyrrolo[2,3-d]pyrimidin-4-yl)amino]-5,6,7,8-tetrahydroimidazo[1,2-a]pyridine-2-carbonitrile
- (7R)-5,6,7,8-Tetrahydro-7-(methyl-7H-pyrrolo[2,3-d]pyrimidin-4-ylamino)imidazo[1,2-a]pyridine-2-carbonitrile
-
Imidazo[1,2-a]pyridine-2-carbonitrile, 5,6,7,8-tetrahydro-7-(methyl-7H-pyrrolo[2,3-d]pyrimidin-4-ylamino)-, (7R)-
Wuxi Fortune Pharmaceutical Co Ltd
Jak1 tyrosine kinase inhibitor
Wuxi Fuxin Pharmaceutical Research and Development , in collaboration with Wuxi Apptec , is investigating a tablet formulation of WXFL-10203614 , a JAK1 tyrosine kinase inhibitor, for the oral treatment of rheumatoid arthritis. In January 2019, a phase I trial was planned.
- Imidazo[1,2-a]pyridine-2-carbonitrile, 5,6,7,8-tetrahydro-7-(methyl-7H-pyrrolo[2,3-d]pyrimidin-4-ylamino)-, (7R)-, 4-methylbenzenesulfonate, hydrate (1:1:1)
- cas 2226936-85-0
- Imidazo[1,2-a]pyridine-2-carbonitrile, 5,6,7,8-tetrahydro-7-(methyl-7H-pyrrolo[2,3-d]pyrimidin-4-ylamino)-, (7R)-, 2,2,2-trifluoroacetate (1:1)
- cas 2226936-87-2
syn

PATENT
WO2018095345 claiming novel crystalline salt forms of similar compound
PATENT
WO 2016192563
PATENT
US-20190218231
Novel crystalline forms of 7h-pyrrolo[2,3-D]pyrimidine compounds (designated as forms A to E) useful as JAK1 and JAK2 inhibitors for treating arthritis, inflammation and autoimmune diseases.
JAK belongs to the family of tyrosine kinases involved in inflammation, autoimmune diseases, proliferative diseases, transplant rejection, impaired cartilage turnover-related diseases, congenital cartilage malformations, and/or diseases associated with excessive secretion of IL6. The present invention also provides a method for preparing the compound or a pharmaceutical composition comprising the compound, and a method for preventing and/or treating inflammation, autoimmune diseases, proliferative diseases, transplant rejection, impaired cartilage turnover-related diseases, congenital cartilage malformations, and/or diseases associated with excessive secretion of IL6 by administrating the compound of the present invention.
Janus kinase (JAK) is a cytoplasmic tyrosine kinase that transduces a cytokine signal from a membrane receptor to an STAT transcription factor. The prior art has described four members of the JAK family: JAK1, JAK2, JAK3 and TYK2. When cytokines bind to their receptors, JAK family members are auto-phosphorylated and/or trans-phosphorylated from each other, followed by STATs phosphorylation, and then are migrated into the cell nucleus to regulate the transcription. JAK-STAT intracellular signal transduction is suitable for interferons, most interleukins, as well as various cytokines and endocrine factors, such as EPO, TPO, GH, OSM, LIF, CNTF, GM-CSF and PRL (Vainchenker W. et al. (2008)).
A combinatorial study of a genetic model and a small molecule JAK inhibitor has revealed the therapeutic potential of several JAKs. It has been confirmed by mouse and human genetics that JAK3 is an immunosuppressive target (O’Shea J. et al. (2004)). A JAK3 inhibitor has been successfully used in clinical development. At first, it was used in organ transplant rejection, and later also used in other immunoinflammatory indications such as rheumatoid arthritis (RA), psoriasis and Crohn’s disease (http://clinicaltrials.gov/). It has been confirmed by human genetics and mouse knockout studies that TYK2 is a potential target for immunoinflammatory diseases (Levy D. and Loomis C. (2007)). JAK1 is a new target in the field of immunoinflammatory diseases. The heterodimerization of JAK1 and other JAKs arouses a transduction of cytokine-driven pro-inflammatory signaling. Thus, it is expected that inhibition of JAK1 and/or other JAKs has a therapeutic benefit for a series of inflammatory diseases and other diseases driven by JAK-mediated signal transduction.
transduction.
Example 1: Preparation of Compound 1
Step 1: 2-chloro-4-nitro-1-oxo-pyridin-1-ium (40.0 g, 229.2 mmol) and (4-methoxyphenyl)methylamine (63 g, 458.4 mmol) were dissolved in EtOH (400 mL), and the resulting solution was stirred at reflux for 5 hours. TLC (PE:EA=2:1) showed that the reaction was complete. The EtOH was concentrated to half of its volume and was cooled in an ice bath for 2-3 hours. The resulting cold mixture was filtered, and the isolated solid was washed with PE (60 mL*3) and ice water (60 mL*3), respectively. Drying in vacuum given an orange solid, N-[(4-methoxyphenyl)methyl]-4-nitro-1-oxo-pyridin-1-ium-2-amine (2) (38.6 g, 140.2 mmol, with a yield of 61.2%). MS (ESI) calcd. For r C 13H 13N 3O 4 [M+H] + 275, found 276.
Step 2: to a solution of N-[(4-methoxyphenyl)methyl]-4-nitro-1-oxo-pyridin-1-ium-2-amine (5.0 g, 18.16 mmol) in CHCI 3 (50 mL) was dropwise added PCI 3 (8.4 g, 60.8 mmol) at 0° C. After the addition, the reaction mixture was heated to 25° C. and stirred vigorously for 16 hours. TLC (PE:EA=1:1) showed that the reaction was complete. The reaction mixture was filtered, and the resulting solid was washed with PE (30 mL*3) to give a yellow solid compound, N-[(4-methoxyphenyl)methyl]-4-nitro-pyridin-2-amine (3) (4.2 g, a crude product) which was directly used in the next step without further purification. MS (ESI) calcd. For C 15H 18N 6 [M+H] +259, found 260.
Step 3: to a solution of N-[(4-methoxyphenyl)methyl]-4-nitro-pyridin-2-amine (4.2 g, 16.2 mmol) in toluene (10 mL) was dropwise added TFA (5.0 mL) at atmospheric temperature. Then, the mixture was stirred at 80° C. for 2 hours. TLC (PE:EA=1:1) showed that the reaction was complete. The mixture was concentrated under reduced pressure to remove the solvent. The residue was diluted with H 2O (50 mL), and its pH was adjusted to be neutral with solid NaHCO 3. The aqueous phase was extracted with EA (50 mLE*3). The combined organic phase was dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure. The resulting residue was purified by column chromatography (silica, petroleum ether/ethyl acetate=1/0-1:1) to obtain an orange solid compound, 4-nitropyridine-2-amine (4) (700 mg, 5.0 mmol, with a yield of 31.1%). MS (ESI) calcd. For C 5H 5N 3O 2 [M+H] + 139, found 140.
Step 4: to a solution of 4-nitropyridine-2-amine (200 mg, 1.4 mmol) in DME (5 mL) was added 3-bromo-2-oxo-propanoate (280 mg, 1.4 mmol) at atmospheric temperature. The resulting mixture was stirred at 25° C. for 1 hour, and then was concentrated under reduced pressure to remove the solvent. The residue was dissolved with EtOH (10 mL); and then was refluxed for 3 hours. TLC showed that the reaction was complete. The reaction solution was cooled to room temperature, and the solvent was concentrated under reduced pressure. The residue was basified with saturated NaHCO 3 aqueous solution (25 mL). The aqueous phase was extracted with DCM (15 mL*3); and the combined organic phase was dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure. The resulting residue was purified by flash column chromatography (EA:PE=10-60%) to obtain a light yellow solid compound, ethyl 7-nitroimidazo[1,2-]pyridin-2-carboxylate (5) (302 mg, with a yield of 88.9%). MS (ESI) calcd. For C 10H 9N 3O 4 [M+H] + 235, found 236.
Step 5: a solution of ethyl 7-nitroimidazo[1,2-a]pyridin-2-carboxylate (150 mg, 637.8 mmol) in ethanol (20 mL) was added HCl (7 mg, 0.2 mmol) and PtO 2 (15 mg, 0.6 mmol) at atmospheric temperature. The reaction system was repeatedly vacuumed and filled with N 2 for three times, then filled with H 2(50 psi), and was stirred at 50° C. for 16 hours. TLC (PE:EA=1:1) showed that the reaction was complete. The reaction mixture was concentrated to half of its volume, and filtered to obtain a white solid compound, ethyl 7-amino-5,6,7,8-tetrahydroimidazo[1,2-a]pyridin-2-carboxylate hydrochloride (6) (120 mg, a crude product). MS (ESI) calcd. For C 10H 15N 3O 2 [M+H] + 209, found 210.
Step 6: ethyl 7-amino-5,6,7,8-tetrahydroimidazo[1,2-a]pyridin-2-carboxylate hydrochloride (100 mg, 0.4 mmol) and 4-chloro-7-(p-toluenesulfonyl)pyrrolo[2,3-d]pyrimidine (137 mg, 0.4 mmol) were dissolved in n-BuOH (5 mL), and DIEA (158 mg, 1.2 mmol) were added to the above solution. The resulting mixture was stirred under reflux for 16 hours. LC-MS showed that the reaction was complete. The reaction mixture was concentrated under reduced pressure, and the resulting residue was diluted with H 2O (10 mL). The aqueous phase was extracted with EA (20 mL*3); and the combined organic phase was dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure. The resulting residue was purified by preparative TLC (PE:EA=0:1) to obtain a light yellow solid compound, ethyl 7-[[7-(p-toluenesulfonyl) pyrrolo[2,3-d]pyrimidin-4-yl] amino]-5,6,7,8-tetrahydroimidazo[1,2-α]pyridin-2-carboxylate (7) (55 mg, 0.11 mmol, with a yield of 28.1%). MS (ESI) calcd. For C 23H 24N 6O 4S [M+H] + 480, found 481.
Step 7: to a solution of ethyl 7-[[7-(p-toluenesulfonyl) pyrrolo[2,3-d]pyrimidin-4-yl]amino]-5,6,7,8-tetrahydroimidazo[1,2-α]pyridin-2-carboxylate (3.0 g, 6.2 mmol) in THF (150 mL) was added NaH (499 mg, 12.5 mmol) in portions under N 2 atmosphere at 0° C. The mixture was stirred at that temperature for 1 hour, and then was dropwise added MeI (7.1 g, 50.2 mmol). After the addition, the mixture was stirred at atmospheric temperature for 1 hour. TLC showed that the reaction was complete. The reaction was quenched by the addition of saturated NH 4Cl (10 mL), and then was diluted by the addition of ice water (50 mL). The aqueous phase was extracted with a mixed solvent of DCM/MeOH (3:1, 50 mL*3). The combined organic phase was dried over sodium sulfate, filtered, and concentrated under reduced pressure. The resulting crude product was purified by flash column chromatography (DCM:MeOH=10:1) to obtain a light yellow solid, ethyl 7-[methyl-[7-(p-toluenesulfonyl)pyrrolo[2,3-d]pyrimidin-4-yl]amino]-5,6,7,8-tetrahydroimidazo[1,2-a]pyridin-2-carboxylate (8) (1.5 g, with a yield of 45%). MS (ESI) calcd. For C 24H 26N 6O 4S [M+H] + 494, found 495.
Step 8: to a solution of 7-[methyl-[7-(p-toluenesulfonyl) pyrrolo[2,3-d]pyrimidin-4-yl]amino]-5,6,7,8-tetrahydroimidazo[1,2-a]pyridin-2-carboxylate (4.0 g, 8.1 mmol) in THF (40 mL) and H 2O (8 mL) was added LiOH.H 2O (509 mg, 12.1 mmol), and the mixture was stirred at 20° C. for 10 hours. TLC showed that the reactants were completely consumed. THF in the reaction mixture was removed under reduced pressure; and the pH of the residue was adjusted to 2-3 with 2M HCl (4 mL) to form a white solid. The solid was filtered out, and was concentrated under reduced pressure to obtain 7-[methyl-[7-(p-toluenesulfonyl)pyrrolo[2,3-d]pyrimidin-4-yl]amino]-5,6,7,8-tetrahydroimidazo[1, 2-a]pyridin-2-carboxylic acid (9) as a white solid (3.6 g, with a yield of 95.4%). MS (ESI) calcd. For C 22H 22N 6O 4S [M+H] + 466, found 467.
Step 9: to a solution of 7-[methyl-[7-(p-toluenesulfonyl)pyrrolo[2,3-d]pyrimidin-4-yl]amino]-5,6,7,8-tetrahydroimidazo[1, 2-a]pyridin-2-carboxylic acid (1.8 g, 3.9 mmol) in DMF (20 mL) was added CDI (751 mg, 4.6 mmol) at 0° C. The reaction solution was heated to 25° C. and stirred for 2 hours, and after that, solid ammonium chloride (2.1 g, 38.6 mmol) was added, and then the reaction was kept overnight at atmospheric temperature. LC-MS showed that the reactants were completely consumed. The reaction mixture was poured into ice water (50 mL), and a white solid was precipitated. The solid was filtered out, washed with water (20 mL), and was dried under reduced pressure in a rotating manner to obtain 7-[methyl-[7-(p-toluenesulfonyl)pyrrolo[2,3-d]pyrimidin-4-yl]amino]-5,6,7,8-tetrahydroimidazo[1,2-a]pyridin-2-carboxamide (10) as a white solid (2.5 g, a crude product) which product was directly used in the next step. MS (ESI) calcd. For C 22H 23N 7O 3S [M+H] + 465, found 466.
Step 10: 7-[methyl-[7-(p-toluenesulfonyl)pyrrolo[2,3-d]pyrimidin-4-yl]amino]-5,6,7,8-tetrahydroimidazo[1, 2-a]pyridin-2-carboxamide (2.5 g, 5.4 mmol) was dissolved in a mixture of THF (20 mL), MeOH (10 mL) and H 2O (6 mL), and NaOH (429.6 mg, 10.7 mmol) was added. The mixture was heated to 60° C. and stirred for 30 minutes. LC-MS showed that the reactants were completely consumed. The reaction mixture was concentrated under reduced pressure to obtain 7-[methyl-[heptahydropyrrolo[2,3-d]pyrimidin-4-yl]amino]-5,6,7,8-tetrahydroimidazo[1,2-α]pyridin-2-carboxamide (11) as a white solid (2.5 g, a crude product) which was directly used in the next step. MS (ESI) calcd. For C 15H 17N 7O [M+H] + 311, found 312.
Step 11: to a solution of 7-[methyl-[heptahydropyrrolo[2,3-d]pyrimidin-4-yl]amino]-5,6,7,8-tetrahydroimidazo[1,2-a]pyridin-2-carboxamide (2.0 g, 6.4 mmol) and triethylamine (3.9 g, 38.5 mmol) in THF (20 mL) was dropwise added TFAA (4.1 g, 19.3 mmol) at 0° C. After the addition, the reaction solution was stirred at atmospheric temperature for 30 minutes. LC-MS showed the starting materials were completely consumed. The reaction mixture was poured into ice water (20 mL), and extracted with DCM/MeOH (5:1, 100 mL*2). The combined organic layer was washed with saturated saline (20 mL), dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure to obtain a residue. The residue was purified by column chromatography (DCM/MeOH=40/1 to 20:1) to obtain 7-[methyl-[7-hydropyrrolo[2,3-d]pyrimidin-4-yl]amino]-5,6,7,8-tetrahydroimidazo[1,2-a]pyridin-2-nitrile (12,378 mg, with a yield of 19.8%). MS (ESI) calcd. For C 15H 15N 7 [M+H] + 293, found 294. 1H NMR (400 MHz, DMSO-d6) 11.44-11.71 (m, 1H), 7.99-8.17 (m, 2H), 7.11-7.20 (m, 1H), 6.63 (dd, J=1.76, 3.26 Hz, 1H), 5.33 (br. s., 1H), 4.21-4.21-4.31 (m, 1H), 4.13 (dt, J=4.14, 12.49 Hz, 1H), 3.27 (s, 3H), 2.91-3.11 (m, 2H), 2.31-2.44 (m, 1H), 2.07 (d, J=11.54 Hz, 1H).
Step 12: racemic 7-[methyl-[7-hydropyrrolo[2,3-d]pyrimidin-4-yl]amino]-5,6,7,8-tetrahydroimidazo[1,2-a]pyridin-2-nitrile (30 mg, 102.3 umol) was separated by a chiral column to obtain the compound 1 (10 mg, with a yield of 32.8%).
Compound 1: retention time 6.407 min; MS (ESI) calcd. For C 15H 15N 7 [293, found 294 M+H]+. Purity 98.8%, e.e. was 98.9%; [α] D 20=+78.4° (c=0.6, DMSO). MS ESI calcd. For C 15H 15N 7 [M+H] + 294, found 294. 1H NMR (400 MHz, DMSO-d6) δ ppm 2.02-2.15 (m, 1H) 2.39 (qd, J=12.42, 5.90 Hz, 1H) 2.92-3.12 (m, 2H) 3.28 (s, 3H) 4.05-4.36 (m, 2H) 5.20-5.45 (m, 1H) 6.64 (dd, J=3.39, 1.88 Hz, 1H) 7.17 (dd, J=3.26, 2.51 Hz, 1H) 8.02-8.17 (m, 2H) 11.69 (br s, 1H).
//////////WXFL-10203614, WXFL 10203614 , WXFL10203614, Wuxi Fuxin, arthritis, inflammation, autoimmune diseases, Wuxi Apptec, JAK1, JAK2 inhibitors
N#Cc1cn2CC[C@H](Cc2n1)N(C)c4ncnc3nccc34
Tanzisertib
Tanzisertib
CAS 899805-25-5
trans-4-((9-((3S)-Tetrahydrofuran-3-yl)-8-((2,4,6-trifluorophenyl)amino)-9H-purin-2-yl)amino)cyclohexanol
4-[[9-[(3S)-oxolan-3-yl]-8-(2,4,6-trifluoroanilino)purin-2-yl]amino]cyclohexan-1-ol
C21-H23-F3-N6-O2, 448.4467
- CC 930
- CC-930
- Tanzisertib
- UNII-M5O06306UO
- A c-Jun amino-terminal kinase inhibitor.UNII, M5O06306UO
Treatment of Idiopathic Pulmonary Fibrosis (IPF)
- Originator Celgene Corporation
- Class Antifibrotics; Small molecules
- Mechanism of ActionJ NK mitogen-activated protein kinase inhibitors
- Orphan Drug Status Yes – Idiopathic pulmonary fibrosis
- Discontinued Discoid lupus erythematosus; Idiopathic pulmonary fibrosis
- 16 Jul 2012 Celgene Corporation terminates a phase II trial in Discoid lupus erythematosus in USA (NCT01466725)
- 23 Feb 2012 Celgene initiates enrolment in a phase II trial for Discoid lupus erythematosus in the USA (NCT01466725)
- 08 Nov 2011The Committee for Orphan Medicinal Products (COMP) recommends orphan drug designation for tanzisertib in European Union for Idiopathic pulmonary fibrosis
Tanzisertib has been granted orphan drug status by the FDA for the treatment of idiopathic pulmonary fibrosis. A positive opinion has been received from the EU Committee for Orphan Medicinal Products (COMP
Tanzisertib has been used in trials studying the treatment of Fibrosis, Discoid Lupus, Pulmonary Fibrosis, Interstitial Lung Disease, and Lung Diseases, Interstitial, among others.
PATENT
https://patents.google.com/patent/US20090048275A1/de


PATENT
WO 2006076595
US 20070060598
WO 2008057252
US 20080021048
US 20140094456
WO 2014055548
PATENT
WO 2015153683
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2015153683
/////////Tanzisertib, CC 930, Idiopathic Pulmonary Fibrosis, Orphan Drug, phase II, CELGENE
c1c(c(c(cc1F)F)Nc2n(c3nc(ncc3n2)N[C@H]4CC[C@@H](CC4)O)[C@@H]5COCC5)F
CC-90009

CC-90009
CC-90009-AML-001
CAS 1860875-51-9
461.8 g/mol, C22H18ClF2N3O4
2-(4-chlorophenyl)-N-((2-(2,6-dioxopiperidin-3-yl)-1-oxoisoindolin-5-yl)methyl)-2,2-difluoroacetamide
- 4-Chloro-N-[[2-(2,6-dioxo-3-piperidinyl)-2,3-dihydro-1-oxo-1H-isoindol-5-yl]methyl]-α,α-difluorobenzeneacetamide
- Benzeneacetamide, 4-chloro-N-[[2-(2,6-dioxo-3-piperidinyl)-2,3-dihydro-1-oxo-1H-isoindol-5-yl]methyl]-α,α-difluoro-
Phase 1 Clinical, Acute myelogenous leukemia, Protein cereblon modulator
Useful for treating chronic lymphocytic leukemia, chronic myelocytic leukemia, acute lymphoblastic leukemia or acute myeloid leukemia.
Celgene is developing CC-90009, a cereblon E3 ligase modulator, for treating AML; in January 2019, data from a phase I trial were expected later that year.
- 0iginator Celgene Corporation
- Class Antineoplastics
- Mechanism of Action CRBN protein modulators; Ubiquitin protein ligase complex modulators
- Phase I Acute myeloid leukaemia
- 28 Mar 2019 No recent reports of development identified for clinical-Phase-Unknown development in Acute-myeloid-leukaemia in USA (IV)
- 01 Sep 2016 Phase-I clinical trials in Acute myeloid leukaemia (Second-line therapy or greater) in Canada (IV) (NCT02848001)
- 04 Aug 2016 Celgene plans a phase I trial for Acute Myeloid Leukaemia in USA and Canada (NCT02848001)
In September 2016, Celgene initiated a phase I dose-finding trial of CC 90009 in patients with relapsed or refractory acute myeloid leukaemia (NCT02848001; CC-90009-AML-001). The open-label study intends to enrol 60 patients in the US and Canada
CC-90009 is a cereblon modulator. CC-90009 specifically binds to CRBN, thereby affecting the activity of the ubiquitin E3 ligase complex. This leads to the ubiquitination of certain substrate proteins and induces the proteasome-mediated degradation of certain transcription factors, including Ikaros (IKZF1) and Aiolos (IKZF3), which are transcriptional repressors in T-cells. This reduces the levels of these transcription factors, and modulates the activity of the immune system, which may include the activation of T-lymphocytes. .
Development Overview
cereblon modulator CC-90009A modulator of cereblon (CRBN), which is part of the cullin 4-RING E3 ubiquitin ligase complex (CRL4-CRBN E3 ubiquitin ligase; CUL4-CRBN E3 ubiquitin ligase), with potential immunomodulating and pro-apoptotic activities. Upon administration, CC-90009 specifically binds to CRBN, thereby affecting the activity of the ubiquitin E3 ligase complex. This leads to the ubiquitination of certain substrate proteins and induces the proteasome-mediated degradation of certain transcription factors, including Ikaros (IKZF1) and Aiolos (IKZF3), which are transcriptional repressors in T-cells. This reduces the levels of these transcription factors, and modulates the activity of the immune system, which may include the activation of T-lymphocytes. In addition, this downregulates the expression of other proteins, including interferon regulatory factor 4 (IRF4) and c-myc, which plays a key role in the proliferation of certain cancer cell types. CRBN, the substrate recognition component of the E3 ubiquitin ligase complex, plays a key role in the ubiquitination of certain proteins. Check for active clinical trials using this agent. (NCI Thesaurus)

WO 2017120446,
PATENT
WO2016007848
US 20170348298
WO 2017120415
WO 2017120446
WO 2017120437
PATENT
WO2017214014
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2017214014&tab=PCTDESCRIPTION
Provided herein are methods of treating, preventing, managing, and/or ameliorating a hematologic malignancy with 2-(4-chlorophenyl)-N-((2-(2,6-dioxopiperidin-3-yl)-l-oxoisoindolin-5-yl)methyl)-2,2-difluoroacetamide or a stereoisomer or a mixture of
stereoisomers, an isotopologue, pharmaceutically acceptable salt, tautomer, solvate, hydrate, co-crystal, clathrate, or polymorph thereof. Further provided is a compound for use in methods of treating, preventing, managing, and/or ameliorating a hematologic malignancy, wherein the compound is 2-(4-chlorophenyl)-N-((2-(2,6-dioxopiperidin-3-yl)-l-oxoisoindolin-5-yl)methyl)-2,2-difluoroacetamide or a stereoisomer or a mixture of stereoisomers, an isotopologue, pharmaceutically acceptable salt, tautomer, solvate, hydrate, co-crystal, clathrate, or polymorph thereof.
The term Compound 1 refers to”2-(4-chlorophenyl)-N-((2-(2,6-dioxopiperidin-3-yl)-l-oxoisoindolin-5-yl)methyl)-2,2-difluoroacetamide” having the structure:
and its stereoisomers or mixture of stereoisomers, isotopologues, pharmaceutically acceptable salts, tautomers, solvates, hydrates, co-crystals, clathrates, or polymorphs thereof. In certain embodiments, Compound 1 refers to 2-(4-chlorophenyl)-N-((2-(2,6-dioxopiperidin-3-yl)-l-oxoisoindolin-5-yl)methyl)-2,2-difluoroacetamide and its tautomers. In certain embodiments, Compound 1 refers to a polymorph of 2-(4-chlorophenyl)-N-((2-(2,6-dioxopiperidin-3-yl)-l-
oxoisoindolin-5-yl)methyl)-2,2-difluoroacetamide. In certain embodiments, Compound 1 refers to polymorph Form C of 2-(4-chlorophenyl)-N-((2-(2,6-dioxopiperidin-3-yl)-l-oxoisoindolin-5-yl)methyl)-2,2-difluoroacetamide. In one embodiment, the stereoisomer is an enantiomer.
PATENT
WO-2019136016
Novel isotopologs of the compound presumed to be CC-90009 , processes for their preparation and compositions comprising them are claimed.



| Patent ID | Title | Submitted Date | Granted Date |
|---|---|---|---|
| US2017199193 | METHODS FOR TREATING CANCER AND THE USE OF BIOMARKERS AS A PREDICTOR OF CLINICAL SENSITIVITY TO THERAPIES | 2017-01-06 | |
| US2018224435 | METHODS FOR MEASURING SMALL MOLECULE AFFINITY TO CEREBLON | 2018-02-02 | |
| US2018353496 | FORMULATIONS OF 2-(4-CHLOROPHENYL)-N-((2-(2,6-DIOXOPIPERIDIN-3-YL)-1-OXOISOINDOLIN-5-YL)METHYL)-2,2-DIFLUOROACETAMIDE | 2018-07-19 | |
| US2017196847 | FORMULATIONS OF 2-(4-CHLOROPHENYL)-N-((2-(2,6-DIOXOPIPERIDIN-3-YL)-1-OXOISOINDOLIN-5-YL)METHYL)-2,2-DIFLUOROACETAMIDE | 2017-01-06 | |
| US2017348298 | TREATMENT OF A HEMATOLOGIC MALIGNANCY WITH 2-(4-CHLOROPHENYL)-N-((2-(2,6-DIOXOPIPERIDIN-3-YL)-1-OXOISOINDOLIN-5-YL)METHYL)-2,2-DIFLUOROACETAMIDE | 2017-06-05 |
| Patent ID | Title | Submitted Date | Granted Date |
|---|---|---|---|
| US2018221361 | ANTIPROLIFERATIVE COMPOUNDS AND METHODS OF USE THEREOF | 2018-04-09 | |
| US9968596 | Antiproliferative compounds and methods of use thereof | 2017-10-02 | 2018-05-15 |
| US2017197934 | SOLID FORMS OF 2-(4-CHLOROPHENYL)-N-((2-(2,6-DIOXOPIPERIDIN-3-YL)-1-OXOISOINDOLIN-5-YL)METHYL)-2,2-DIFLUOROACETAMIDE, AND THEIR PHARMACEUTICAL COMPOSITIONS AND USES | 2017-01-06 | |
| US9499514 | ANTIPROLIFERATIVE COMPOUNDS AND METHODS OF USE THEREOF | 2015-07-09 | 2016-01-14 |
| US9808451 | ANTIPROLIFERATIVE COMPOUNDS AND METHODS OF USE THEREOF | 2016-09-23 |
////////CC-90009 , CC 90009 , CC90009, chronic lymphocytic leukemia, chronic myelocytic leukemia, acute lymphoblastic leukemia, acute myeloid leukemia, phase I, CANCER, CC-90009-AML-001
Clc1ccc(cc1)C(F)(F)C(=O)NCc2ccc3C(=O)N(Cc3c2)C4CCC(=O)NC4=O
Quinacillin
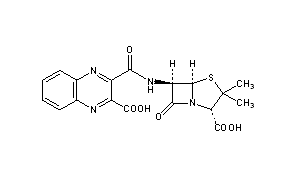
UNII-83NB50X92M
Cas 1596-63-0
83NB50X92M
Quinacilina
MW 416.4 g/mol, MF C18H16N4O6S
(2S,5R,6R)-6-[(3-carboxyquinoxaline-2-carbonyl)amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid
- 4-Thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid, 6-(3-carboxy-2-quinoxalinecarboxamido)-3,3-dimethyl-7-oxo- (7CI,8CI)
- 4-Thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid, 6-[[(3-carboxy-2-quinoxalinyl)carbonyl]amino]-3,3-dimethyl-7-oxo-, [2S-(2α,5α,6β)]-
- (2S,5R,6R)-6-[[(3-Carboxy-2-quinoxalinyl)carbonyl]amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid
- 3-Carboxy-2-quinoxalinylpenicillanic acid
- 3-Carboxy-2-quinoxalinylpenicillin
- 6-(3-Carboxy-2-quinoxalinecarboxamido)-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid
- Penicillin, (3-carboxy-2-quinoxalinyl)-
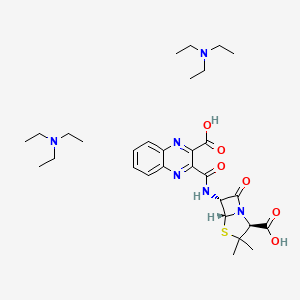

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....












![Acetic acid;(2S,3R,4S,5S,6R)-2-[[4-[[4-[(E)-4-(2,9-diazaspiro[5.5]undecan-2-yl)but-1-enyl]-2-methylphenyl]methyl]-5-propan-2-yl-1H-pyrazol-3-yl]oxy]-6-(hydroxymethyl)oxane-3,4,5-triol.png](https://pubchem.ncbi.nlm.nih.gov/image/imgsrv.fcgi?cid=91758795&t=l)

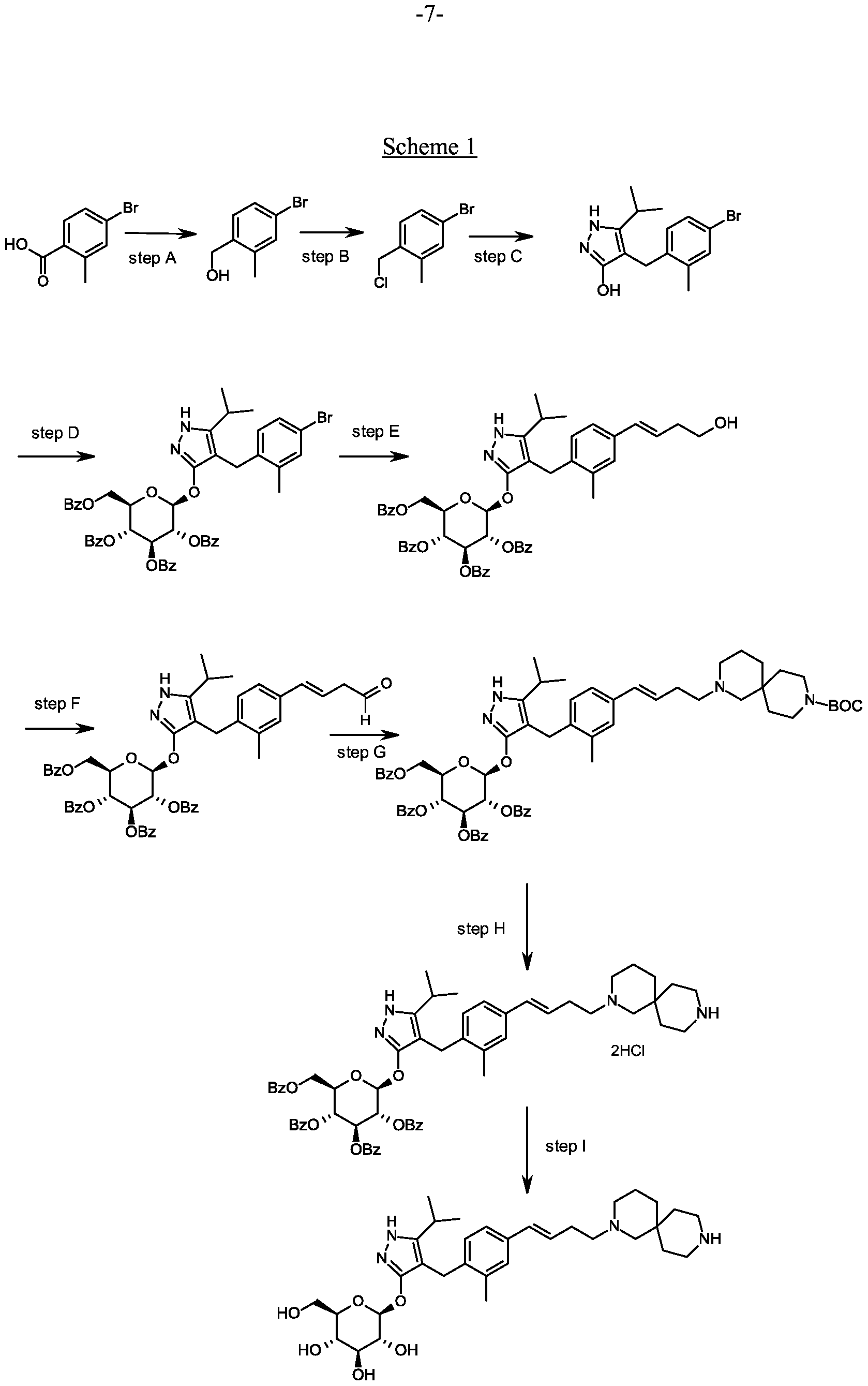



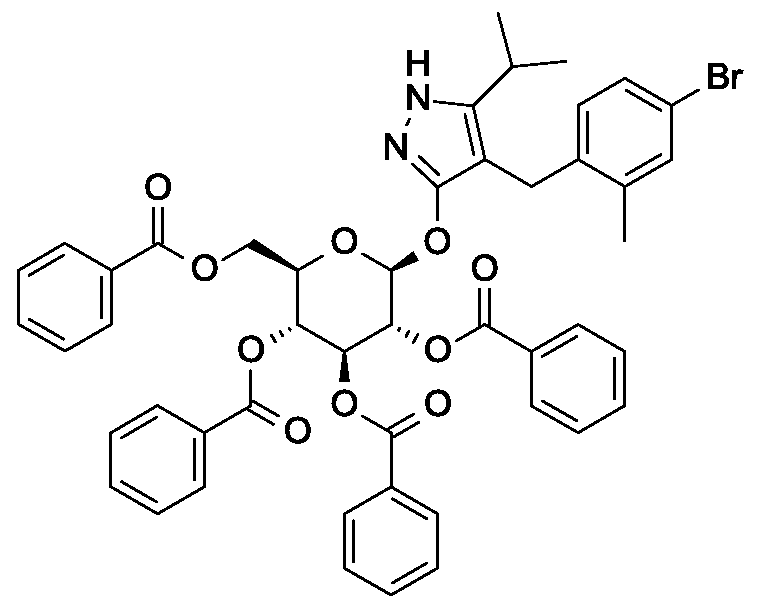
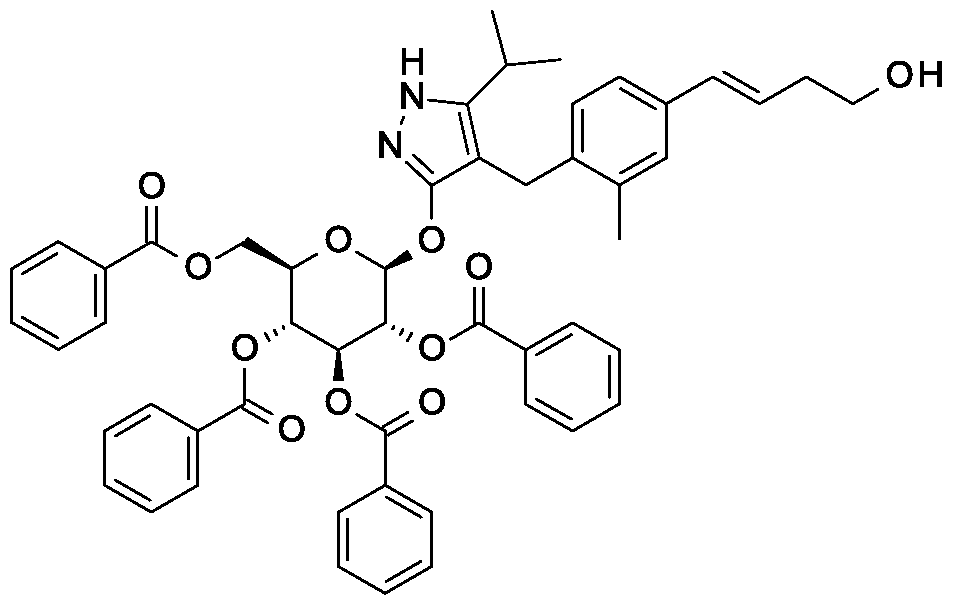
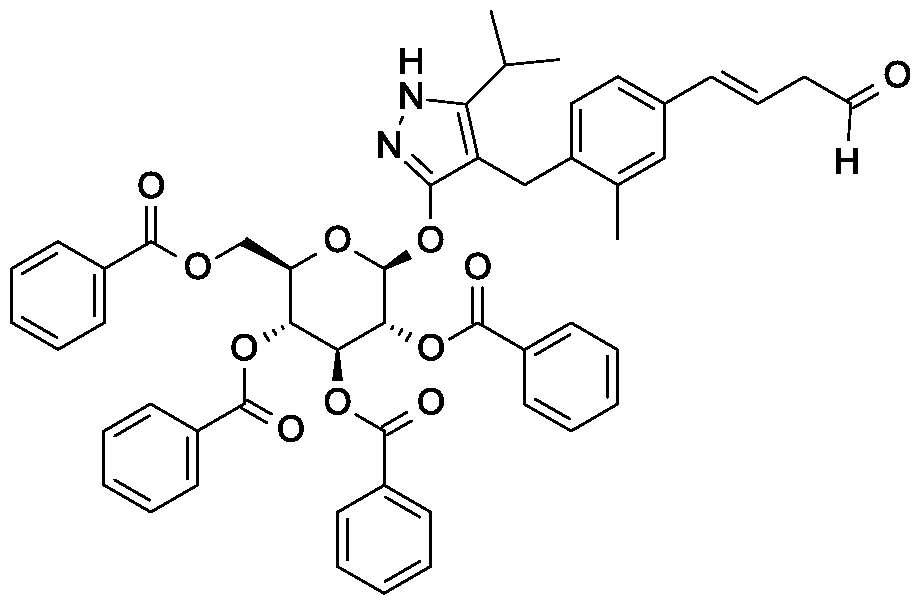


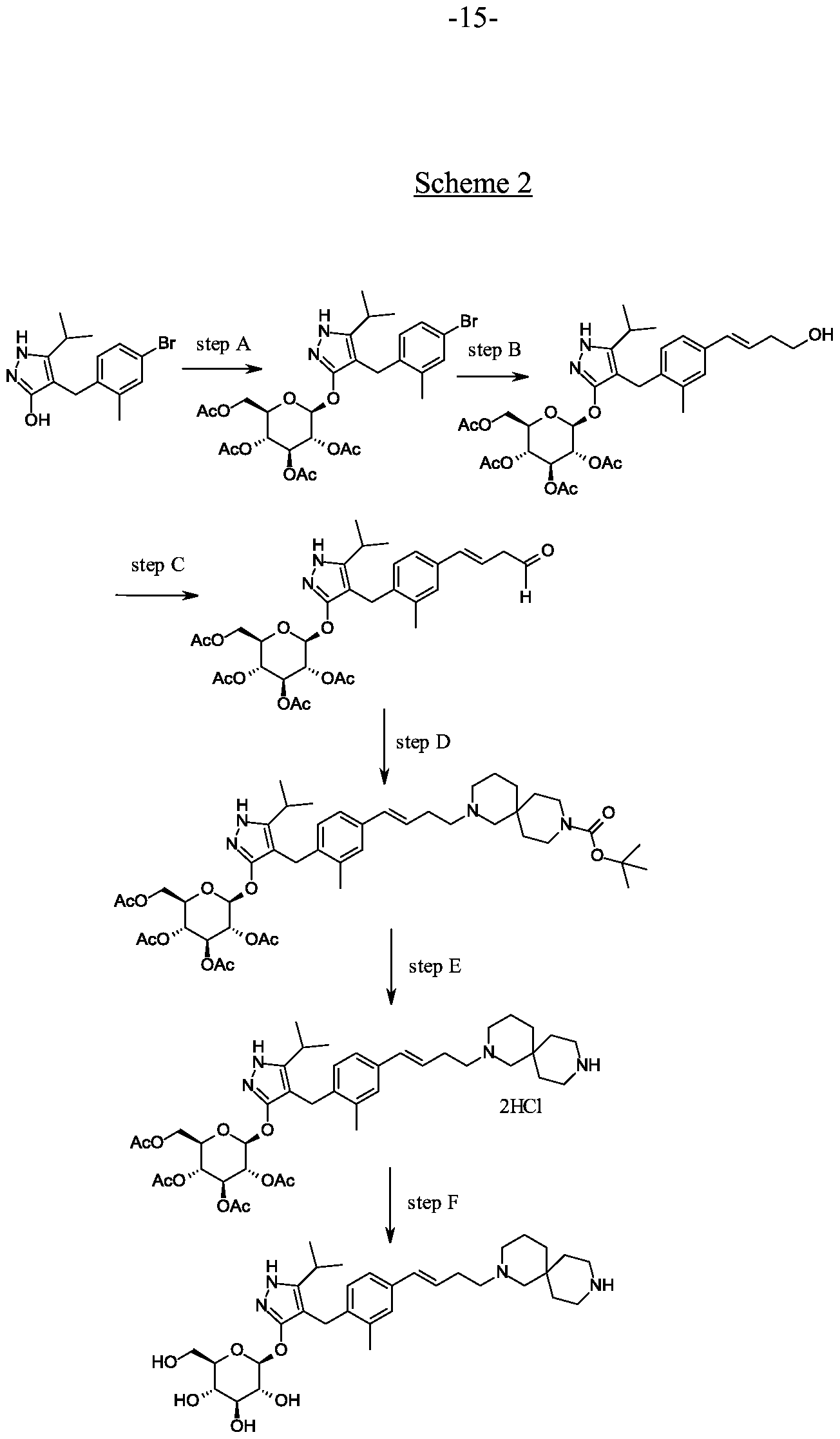
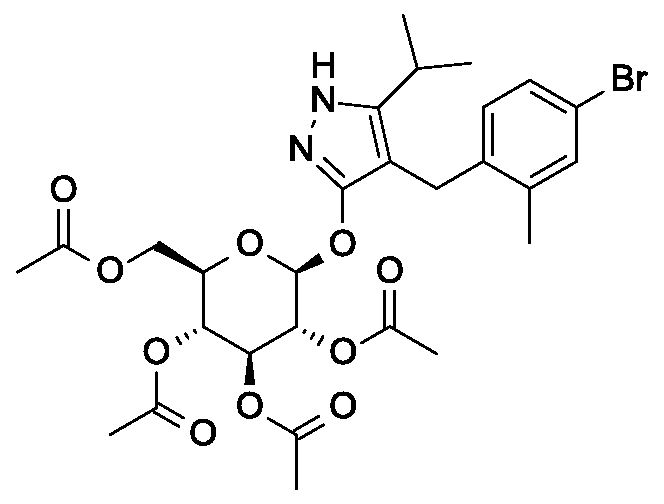
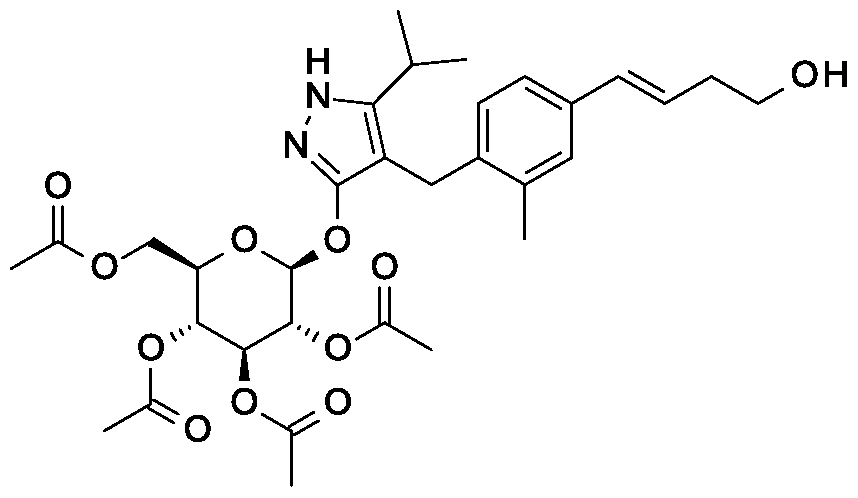

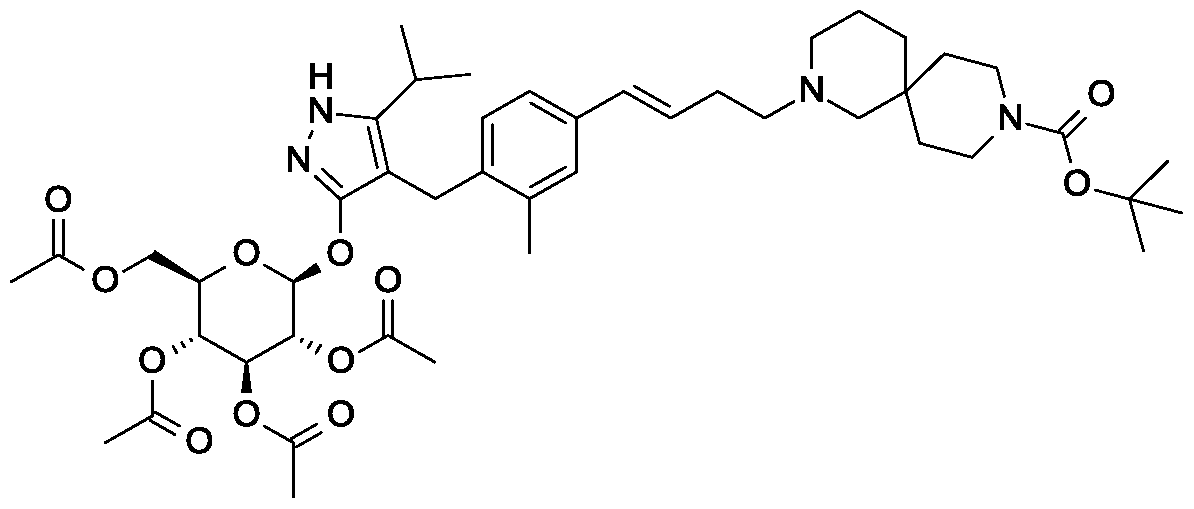

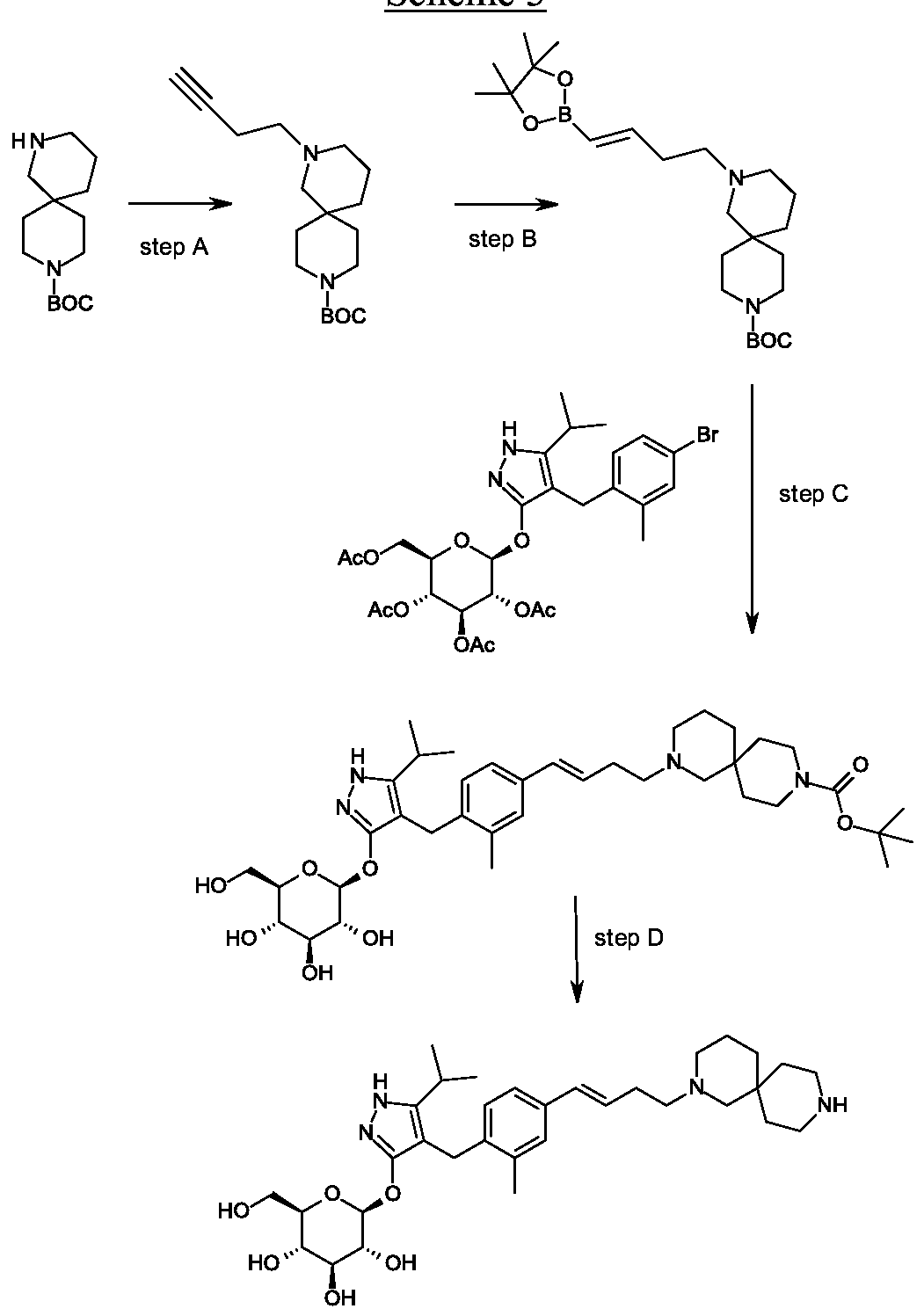

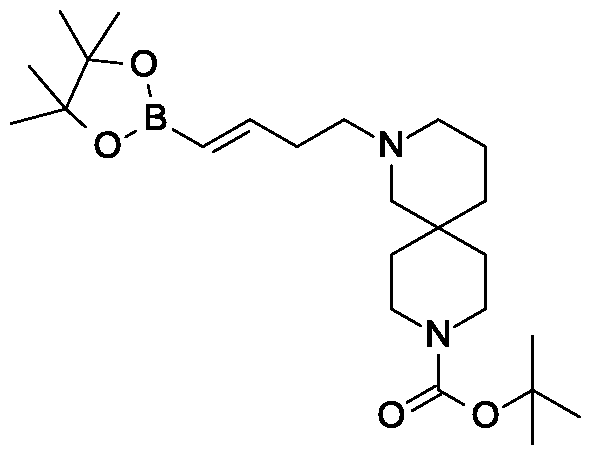


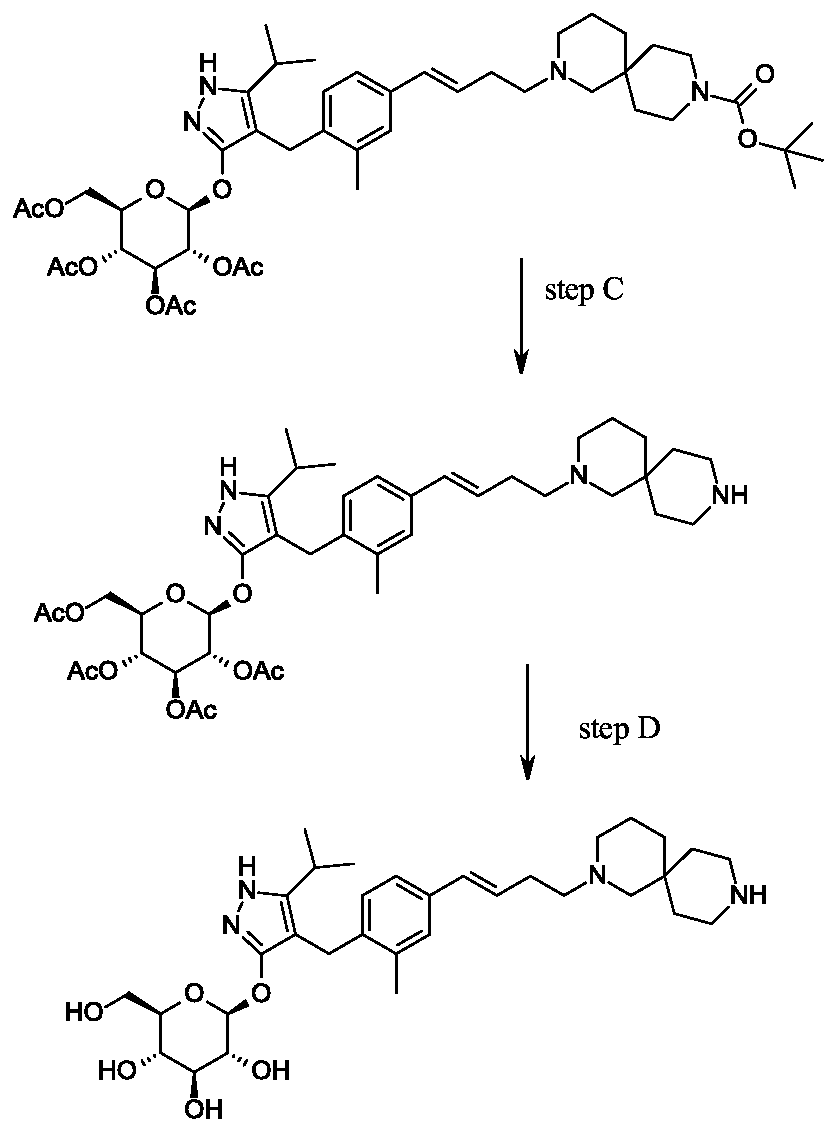



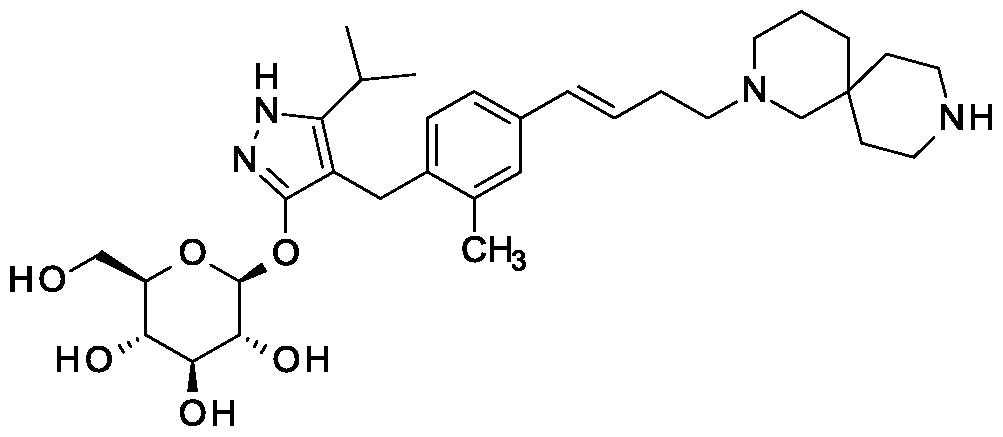





![(7R)-7-[Methyl(7H-pyrrolo[2,3-d]pyrimidin-4-yl)amino]-5,6,7,8-tetrahydroimidazo[1,2-a]pyridine-2-carbonitrile.png](https://pubchem.ncbi.nlm.nih.gov/image/imgsrv.fcgi?cid=134405105&t=l)

![2-(4-Chlorophenyl)-N-[[2-(2,6-dioxopiperidin-3-yl)-1-oxo-3H-isoindol-5-yl]methyl]-2,2-difluoroacetamide.png](https://pubchem.ncbi.nlm.nih.gov/image/imgsrv.fcgi?cid=118647211&t=l)