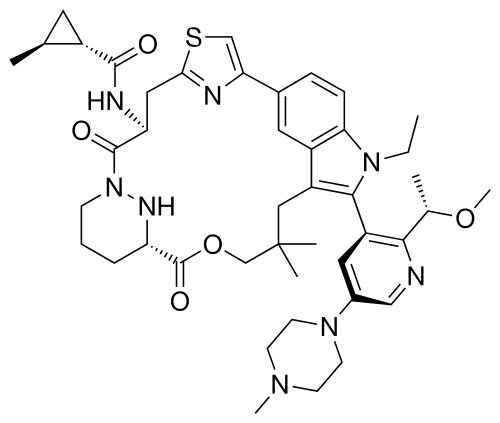
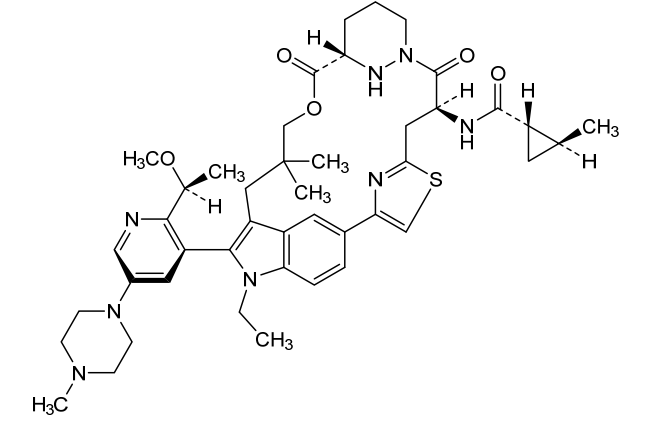
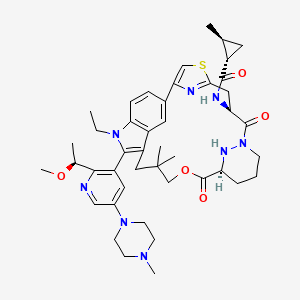
Daraxonrasib
CAS 2765081-21-6
MFC44H58N8O5S MW811.0 g/mol
trans-(1S,2S)-N-[(7S,13S)-21-ethyl-20-[2-[(1S)-1-methoxyethyl]-5-(4-methylpiperazin-1-yl)-3-pyridinyl]-17,17-dimethyl-8,14-dioxo-15-oxa-4-thia-9,21,27,28-tetrazapentacyclo[17.5.2.12,5.19,13.022,26]octacosa-1(25),2,5(28),19,22(26),23-hexaen-7-yl]-2-methylcyclopropane-1-carboxamide
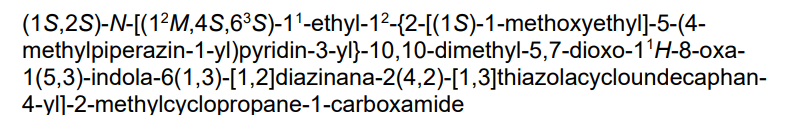
Kirsten rat sarcoma viral oncogene homolog inhibitor, antineoplastic, RMC-6236, RMC 6236, B6T47Y2UAP, RAS-IN-2,
Daraxonrasib (formerly RMC-6236) is an investigational, orally administered “molecular glue” RAS inhibitor developed by Revolution Medicines for treating advanced solid tumors with RAS mutations, particularly metastatic pancreatic cancer. April 2026 Phase 3 trials showed it significantly improves survival, demonstrating high potential as a first-line treatment.
Key Clinical Findings and Updates (as of April 2026):
- Mechanism: It acts as a RAS(ON) inhibitor, targeting mutated and wild-type RAS proteins (
) to disrupt cancer signaling.
- Breakthrough Results: Data from the RASolute 302 trial showed a substantial survival benefit in patients with previously treated metastatic pancreatic ductal adenocarcinoma (PDAC).
- High Response Rates: In trials, daraxonrasib combined with chemotherapy showed a 58% confirmed objective response rate (ORR) and 84% progression-free survival (PFS) at 6 months in untreated RAS-mutant metastatic pancreatic cancer.
- Safety Profile: Generally well-tolerated, with side effects including rash, diarrhea, stomatitis, and nausea.
- Recognition: Named the “2025 Molecule of the Year” by Drug Hunter for its, novel mechanism and clinical potential.
Daraxonrasib is currently being studied in the Phase 3 RASolute 303 trial for first-line treatment of pancreatic cancer.
Daraxonrasib (RMC-6236) is a RAS inhibitor drug. It is undergoing testing by Revolution Medicines to treat advanced solid tumors with RAS mutations, especially metastatic pancreatic ductal adenocarcinoma (PDAC) containing KRAS G12X mutations.[1] It received a breakthrough therapy designation from the U.S. Food and Drug Administration.[2]
Daraxonrasib is orally active and multi-selective RAS inhibitor. It uses a tri-complex mechanism to target the active, GTP-bound form of RAS proteins, including mutant and wild-type forms. Unlike conventional RAS inhibitors, it first binds to the chaperone-like protein cyclophilin A to form a complex, which then attaches to active RAS. This interaction blocks downstream effector binding and inhibits oncogenic signaling.[3]
In 2026, Daraxonrasib clinical trial completed a phase 3 clinical trial (RASolute 302) to assess efficacy compared to standard-of-care chemotherapy.[4] The trial met all primary and key secondary endpoints, including progression-free survival (PFS). The company reported median survival of 13.2 months with daraxonrasib vs. 6.7 months with standard chemotherapy. The hazard ratio for death was 0.40 (a 60% reduction in risk of death; p < 0.0001). Daraxonrasib was generally well tolerated with a manageable safety profile and no new safety signals.[5]
PAT
PAT
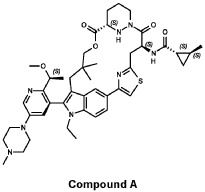
PATENT ATTORNEY DOCKET: 51432-038WO2 Part 4 – Purification of Compound A – (1S,2S)-N-[(7S,13S)-21-ethyl-20-{2-[(1S)-1- methoxyethyl]-5-(4-
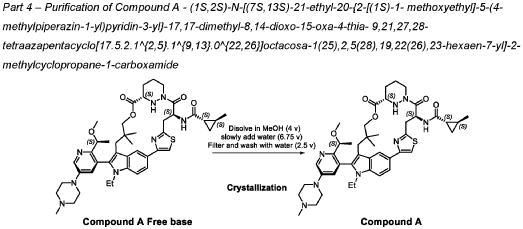
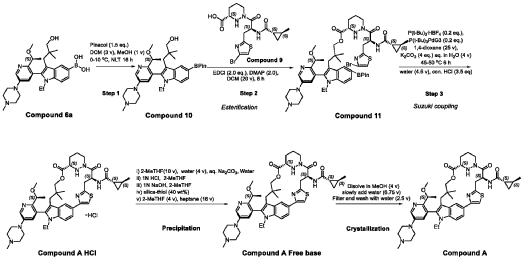
1.0equiv) at 25°C. The resulting suspension was stirred until solids were completely dissolved. The resulting methanol solution was filtered through microporous filter and transferred to another reactor. Then the reactor temperature was maintained at 25°C and slowly water (2.41kg, 1.0 V) water was added over a period of 30 minutes. The resulting cloudy solution was stirred for another 30 minutes at 25°C. Then a solution of methanol and water (3.42kg, 1:2, v/v) slowly over 1 hour. The resulting suspension was stirred for 2 hours at 25°C. Again, to the suspension additional water (2.48kg) slowly added over 1 hour. The final, suspension was stirred for additional 1 hour. Water (9.29kg, 3.75 V) was added to the suspension slowly over 2 hours and the mixture was stirred for at least for 16 hours at 25°C. The resulting suspension was filtered and washed with mixed solvent water: MeOH (3:2, v/v) twice (2x 2.2 kg), followed by water (4.91kg) washing. The wet cake was dried under reduced pressure and controlled humidity (temperature: 25 ± 5 ˚C, vacuum ≥ -0.085 MPa, humidity: 10%~20%) for 37 hours to afford Compound A as a white solid (2.68 kg, 99.4% a/a purity, 93.0% w/w assay, KF: 6.7%, 3.07 mol, 92% yield, Table 27).
PAT
- Synthesis of ras inhibitorsPublication Number: WO-2024216017-A2Priority Date: 2023-04-14
- Macrocycle compounds useful as kras inhibitorsPublication Number: WO-2024008834-A1Priority Date: 2022-07-08
- Ras inhibitorsPublication Number: US-11690915-B2Priority Date: 2020-09-15Grant Date: 2023-07-04
- Ras inhibitorsPublication Number: US-2023226186-A1Priority Date: 2020-09-15



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
References
- Cregg J, Edwards AV, Chang S, Lee BJ, Knox JE, Tomlinson AC, et al. (March 2025). “Discovery of Daraxonrasib (RMC-6236), a Potent and Orally Bioavailable RAS(ON) Multi-selective, Noncovalent Tri-complex Inhibitor for the Treatment of Patients with Multiple RAS-Addicted Cancers”. Journal of Medicinal Chemistry. 68 (6): 6064–6083. doi:10.1021/acs.jmedchem.4c02314. PMID 40056080.
- Sava J (July 1, 2025). “Daraxonrasib Earns FDA Breakthrough Status in Pancreatic Cancer”. Targeted Oncology. Retrieved October 12, 2025.
- Jiang J, Jiang L, Maldonato BJ, Wang Y, Holderfield M, Aronchik I, et al. (June 2024). “Translational and Therapeutic Evaluation of RAS-GTP Inhibition by RMC-6236 in RAS-Driven Cancers”. Cancer Discovery. 14 (6): 994–1017. doi:10.1158/2159-8290.CD-24-0027. PMC 11149917. PMID 38593348.
- Clinical trial number NCT05379985 at ClinicalTrials.gov
- Mast J (2026-04-13). “Revolution Medicines touts ‘unprecedented’ data for pancreatic cancer pill”. STAT. Retrieved 2026-04-13.
| Clinical data | |
|---|---|
| Other names | RMC-6236 |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2765081-21-6 |
| PubChem CID | 164726578 |
| IUPHAR/BPS | 13368 |
| ChemSpider | 115275938 |
| UNII | B6T47Y2UAP |
| KEGG | D13265 |
| ChEBI | CHEBI:746946 |
| Chemical and physical data | |
| Formula | C44H58N8O5S |
| Molar mass | 811.06 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
//////////daraxonrasib, anax labs, Kirsten rat sarcoma viral oncogene homolog inhibitor, antineoplastic, RMC-6236, RMC 6236, B6T47Y2UAP, RAS-IN-2,

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
