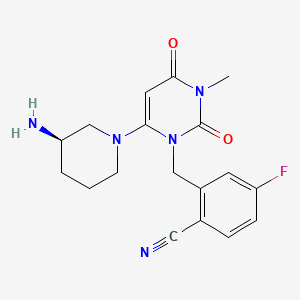
Trelagliptin

Trelagliptin
865759-25-7; UNII-Q836OWG55H
| Molecular Formula: | C18H20FN5O2 |
|---|---|
| Molecular Weight: | 357.382103 g/mol |
2-[[6-[(3R)-3-aminopiperidin-1-yl]-3-methyl-2,4-dioxopyrimidin-1-yl]methyl]-4-fluorobenzonitrile
(R) -2 – ((6 (3-amino-piperidin-1-yl) -3-methyl-2,4-dioxo-3,4-dihydropyrimidine -1 (2H) – yl) methyl) synthesis of 4-fluoro-benzonitrile
(R)-2-((6-(3-amino-3-methylpiperidin-l-yl)-3-methyl-2,4-dioxo-3,4-dihydropyrimidin-l(2H)-yl)methyl)-4-fluorobenzonitrile
A dipeptidyl peptidase-4 (DPP-4) inhibitor used to treat type 2 diabetes.
Research Code SYR-472
CAS No. 865759-25-7 (Trelagliptin)
1029877-94-8 (Trelagliptin Succinate)

Dipeptidyl Peptidase IV (IUBMB Enzyme Nomenclature EC.3.4.14.5) is a type π membrane protein that has been referred to in the literature by a wide a variety of names including DPP4, DP4, DAP-IV, FAPβ, adenosine deaminase complexing protein 2, adenosine deaminase binding protein (AD Abp), dipeptidyl aminopeptidase IV; Xaa-Pro-dipeptidyl-aminopeptidase; Gly-Pro naphthylamidase; postproline dipeptidyl aminopeptidase IV; lymphocyte antigen CD26; glycoprotein GPI lO; dipeptidyl peptidase IV; glycylproline aminopeptidase; glycylproline aminopeptidase; X-prolyl dipeptidyl aminopeptidase; pep X; leukocyte antigen CD26; glycylprolyl dipeptidylaminopeptidase; dipeptidyl-peptide hydrolase; glycylprolyl aminopeptidase; dipeptidyl-aminopeptidase IV; DPP ΓV/CD26; amino acyl-prolyl dipeptidyl aminopeptidase; T cell triggering molecule TρlO3; X-PDAP. Dipeptidyl Peptidase IV is referred to herein as “DPP-IV.” [0003] DPP-W is a non-classical serine aminodipeptidase that removes Xaa-Pro dipeptides from the amino terminus (N-terminus) of polypeptides and proteins. DPP-IV dependent slow release of dipeptides of the type X-GIy or X-Ser has also been reported for some naturally occurring peptides.
DPP-IV is constitutively expressed on epithelial and endothelial cells of a variety of different tissues (intestine, liver, lung, kidney and placenta), and is also found in body fluids. DPP-IV is also expressed on circulating T-lymphocytes and has been shown to be synonymous with the cell-surface antigen, CD-26. DPP-IV has been implicated in a number of disease states, some of which are discussed below.
[0005] DPP-IV is responsible for the metabolic cleavage of certain endogenous peptides (GLP-I (7-36), glucagon) in vivo and has demonstrated proteolytic activity against a variety of other peptides (GHRH, NPY, GLP-2, VIP) in vitro.
GLP-I (7-36) is a 29 amino-acid peptide derived by post-translational processing of proglucagon in the small intestine. GLP-I (7-36) has multiple actions in vivo including the stimulation of insulin secretion, inhibition of glucagon secretion, the promotion of satiety, and the slowing of gastric emptying. Based on its physiological profile, the actions of GLP-I (7-36) are believed to be beneficial in the prevention and treatment of type II diabetes and potentially obesity. For example, exogenous administration of GLP-I (7-36) (continuous infusion) in diabetic patients has been found to be efficacious in this patient population. Unfortunately, GLP-I (7-36) is degraded rapidly in vivo and has been shown to have a short half -life in vivo (t1/2=1.5 minutes).
Based on a study of genetically bred DPP-IV knock out mice and on in vivo I in vitro studies with selective DPP-IV inhibitors, DPP-IV has been shown to be the primary degrading enzyme of GLP-I (7-36) in vivo. GLP-I (7-36) is degraded by DPP-IV efficiently to GLP-I (9-36), which has been speculated to act as a physiological antagonist to GLP-I (7-36). Inhibiting DPP-TV in vivo is therefore believed to be useful for potentiating endogenous levels of GLP-I (7-36) and attenuating the formation of its antagonist GLP-I (9-36). Thus, DPP-IV inhibitors are believed to be useful agents for the prevention, delay of progression, and/or treatment of conditions mediated by DPP-IV, in particular diabetes and more particularly, type 2 diabetes mellitus, diabetic dislipidemia, conditions of impaired glucose tolerance (IGT), conditions of impaired fasting plasma glucose (WG), metabolic acidosis, ketosis, appetite regulation and obesity.
DPP-IV expression is increased in T-cells upon mitogenic or antigenic stimulation (Mattem, T., et al., Scand. J. Immunol, 1991, 33, 737). It has been reported that inhibitors of DPP-IV and antibodies to DPP-IV suppress the proliferation of mitogen-stimulated and antigen-stimulated T-cells in a dose-dependant manner (Schon, E., et al., Biol. Chem., 1991, 372, 305). Various other functions of T-lymphocytes such as cytokine production, IL-2 mediated cell proliferation and B-cell helper activity have been shown to be dependent on DPP-IV activity (Schon, E., et al., Scand. J. Immunol, 1989, 29, 127). DPP-IV inhibitors, based on boroProline, (Flentke, G. R., et al., Proc. Nat. Acad. Set USA, 1991, 88, 1556) although unstable, were effective at inhibiting antigen-induced lymphocyte proliferation and IL-2 production in murine CD4+ T-helper cells. Such boronic acid inhibitors have been shown to have an effect in vivo in mice causing suppression of antibody production induced by immune challenge (Kubota, T. et al, Clin. Exp. Immun., 1992, 89, 192). The role of DPP-IV in regulating T lymphocyte activation may also be attributed, in part, to its cell-surface association with the transmembrane phosphatase, CD45. DPP-IV inhibitors or non-active site ligands may possibly disrupt the CD45-DPP-TV association. CD45 is known to be an integral component of the T-cell signaling apparatus. It has been reported that DPP-IV is essential for the penetration and infectivity of HTV-I and HTV-2 viruses in CD4+ T-cells (Wakselman, M., Nguyen, C, Mazaleyrat, J.-P., Callebaut, C, Krust, B., Hovanessian, A. G., Inhibition of HIV-I infection of CD 26+ but not CD 26-cells by a potent cyclopeptidic inhibitor of the DPP-IV activity of CD 26. Abstract P.44 of the 24.sup.th European Peptide Symposium 1996). Additionally, DPP-IV has been shown to associate with the enzyme adenosine deaminase (ADA) on the surface of T-cells (Kameoka, J., et al., Science, 193, 26 466). ADA deficiency causes severe combined immunodeficiency disease (SCID) in humans. This ADA-CD26 interaction may provide clues to the pathophysiology of SCID. It follows that inhibitors of DPP-TV may be useful immunosuppressants (or cytokine release suppressant drugs) for the treatment of among other things: organ transplant rejection; autoimmune diseases such as inflammatory bowel disease, multiple sclerosis and rheumatoid arthritis; and the treatment of AIDS.
It has been shown that lung endothelial cell DPP-IV is an adhesion molecule for lung-metastatic rat breast and prostate carcinoma cells (Johnson, R. C, et al., J. Cell. Biol, 1993, 121, 1423). DPP-IV is known to bind to fibronectin and some metastatic tumor cells are known to carry large amounts of fibronectin on their surface. Potent DPP-IV inhibitors may be useful as drugs to prevent metastases of, for example, breast and prostrate tumors to the lungs.
High levels of DPP-PV expression have also been found in human skin fibroblast cells from patients with psoriasis, rheumatoid arthritis (RA) and lichen planus (Raynaud, F., et al., J. Cell. Physiol, 1992, 151, 378). Therefore, DPP-TV inhibitors may be useful as agents to treat dermatological diseases such as psoriasis and lichen planus. [0011] High DPP-TV activity has been found in tissue homogenates from patients with benign prostate hypertrophy and in prostatosomes. These are prostate derived organelles important for the enhancement of sperm forward motility (Vanhoof, G., et al., EMr. /.
Clin. Chem. Clin. Biochem., 1992, 30, 333). DPP-IV inhibitors may also act to suppress sperm motility and therefore act as a male contraceptive agent. Conversely, DPP-IV inhibitors have been implicated as novel for treatment of infertility, and particularly human female infertility due to Polycystic ovary syndrome (PCOS, Stein-Leventhal syndrome) which is a condition characterized by thickening of the ovarian capsule and . formation of multiple follicular cysts. It results in infertility and amenorrhea.
DPP-IV is thought to play a role in the cleavage of various cytokines
(stimulating hematopoietic cells), growth factors and neuropeptides.
[0013] Stimulated hematopoietic cells are useful for the treatment of disorders that are characterized by a reduced number of hematopoietic cells or their precursors in vivo. Such conditions occur frequently in patients who are immunosuppressed, for example, as a consequence of chemotherapy and/or radiation therapy for cancer. It was discovered that inhibitors of dipeptidyl peptidase type PV are useful for stimulating the growth and differentiation of hematopoietic cells in the absence of exogenously added cytokines or other growth factors or stromal cells. This discovery contradicts the dogma in the field of hematopoietic cell stimulation, which provides that the addition of cytokines or cells that produce cytokines (stromal cells) is an essential element for maintaining and stimulating the growth and differentiation of hematopoietic cells in culture. (See, e.g., PCT Intl. Application No. PCT/US93/017173 published as WO 94/03055).
[0014] DPP-IV in human plasma has been shown to cleave N-terminal Tyr-Ala from growth hormone-releasing factor and cause inactivation of this hormone. Therefore, inhibitors of DPP-IV may be useful in the treatment of short stature due to growth hormone deficiency (Dwarfism) and for promoting GH-dependent tissue growth or re-growth.
DPP-IV can also cleave neuropeptides and has been shown to modulate the activity of neuroactive peptides substance P, neuropeptide Y and CLIP (Mentlein, R., Dahms, P., Grandt, D., Kruger, R., Proteolytic processing of neuropeptide Y and peptide YY by dipeptidyl peptidase IV, Regul. Pept., 49, 133, 1993; Wetzel, W., Wagner, T., Vogel, D., Demuth, H.-U., Balschun, D., Effects of the CLIP fragment ACTH 20-24 on the duration of REM sleep episodes, Neuropeptides, 31, 41, 1997). Thus DPP-IV inhibitors may also be useful agents for the regulation or normalization of neurological disorders.
Several compounds have been shown to inhibit DPP-IV. Nonetheless, a need still exists for new DPP-IV inhibitors that have advantageous potency, stability, selectivity, toxicity and/or pharmacodynamics properties. In this regard, synthetic methods are provided that can be used to make a novel class of DPP-IV inhibitors.
Trelagliptin (Zafatek) is a pharmaceutical drug used for the treatment of type 2 diabetes (diabetes mellitus).[1]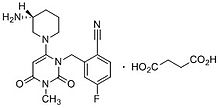
Indications for Medical Use
It is a highly selective dipeptidyl peptidase (DPP-4) inhibitor that is typically used as an add on treatment when the first line treatment of metformin is not achieving the expected glycemic goals; though it has been approved for use as a first line treatment when metformin cannot be used.[1]
Biochemistry
DPP-4 inhibitors activate T-cells and are more commonly known as T-cell activation antigens (specifically CD26).[1][2] Chemically, it is a fluorinated derivative of alogliptin.
Development
Formulated as the salt trelagliptin succinate, it was approved for use in Japan in March 2015.[3] Takeda, the company that developed trelagliptin, chose to not get approval for the drug in the USA and EU.[1] The licensing rights that Takeda purchased from Furiex Pharmaceuticals for DPP-4 inhibitors included a clause specific to development of this drug in the USA and EU.[1] The clause required that all services done for phase II and phase III clinical studies in the USA and EU be purchased through Furiex.[1] Takeda chose to cease development of this drug in the USA and EU because of the high costs quoted by Furiex for these services.[1] Gliptins have been on the market since 2006 and there are 8 gliptins currently registered as drugs (worldwide).[4] Gliptins are an emerging market and are thus being developed at an increasing rate; there are currently two gliptins in advanced stages of development that are expected to be on the market in the coming year.[4]
Gliptins are thought to have cardiovascular protective abilities though the extent of these effects is still being studied.[4] They are also being studied for the ability that this class of drugs has at promoting B-cell survival.[4]
Administration and Dosing
Similar drugs in the same class as trelagliptin are administered once daily while trelagliptin is administered once weekly.[1][5] Alogliptin (Nesina) is the other major DPP-4 inhibitor on the market. It is also owned by Takeda and is administered once daily. A dosing of once per week is advantageous as a reduction in the frequency of required dosing is known to increase patient compliance.[1][2]
Zafatek is administered in the form trelagliptin succinate in a 1:1 mixture of trelagliptin and succinic acid.[6] The drug is marketed with the IUPAC name Succinic acid – 2-({6-[(3R)-3-amino-1-piperidinyl]-3-methyl-2,4-dioxo-3,4-dihydro-1(2H)-pyrimidinyl}methyl)-4-fluorobenzonitrile (1:1), has a molecular mass of 475.470143 grams/mol, and has the molecular formula | C=22 | H=26 | F=1 | N=5 | O=6 .[6][7]
SYNTHESIS …………….
PAPER

The discovery of two classes of heterocyclic dipeptidyl peptidase IV (DPP-4) inhibitors, pyrimidinones and pyrimidinediones, is described. After a single oral dose, these potent, selective, and noncovalent inhibitors provide sustained reduction of plasma DPP-4 activity and lowering of blood glucose in animal models of diabetes. Compounds 13a, 27b, and 27j were selected for development.
2-[6-(3-Aminopiperidin-1-yl)-3-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-ylmethyl]-4-fluorobenzonitrile, TFA salt (27j)
PATENT
WO 2007035629
http://www.google.com/patents/WO2007035629A3?cl=en
(R)-2-((6-(3-amino-3-methylpiperidin-l-yl)-3-methyl-2,4-dioxo-3,4-dihydropyrimidin-l(2H)-yl)methyl)-4-fluorobenzonitrile (30). 2-(6-Chloro-3-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-l-ylmethyl)-4-fluoro-benzonitrile (300 mg, 1.0 mmol), (R)-3-amino-3-methyl-piperidine dihydrochloride (266 mg, 1.4 mmol) and sodium bicarbonate (500 mg, 5.4 mmol) were stirred in a sealed tube in EtOH (3 mL) at 1000C for 2 hrs. The final compound was obtained as TFA salt after HPLC purification. 1H-NMR (400 MHz, CD3OD): δ. 7.78-7.83 (m, IH), 7.14-7.26 (m, 2H), 5.47 (s, IH), 5.12-5.36 (ABq, 2H, J = 105.2, 15.6 Hz), 3.21 (s, IH), 2.72-3.15 (m, 4H), 1.75-1.95 (m, 4H), 1.39 (s, 3H). MS (ES) [m+H] calc’d for C19H22FN5O2, 372.41; found, 372.41.
Compound 34

4-Fluoro-2-methylbenzonitrile (31). A mixture of 2-bromo-5-fluorotoluene (3.5 g, 18.5 mmol) and CuCN (2 g, 22 mmol) in DMF (100 mL) was refluxed for 24 hours. The reaction was diluted with water and extracted with hexane. The organics were dried over MgSO4 and the solvent removed to give product 31 (yield 60%). 1H-NMR (400 MHz, CDCl3): δ 7.60 (dd, J=5.6, 8.8 Hz, IH), 6.93-7.06 (m, 2H), 2.55 (s, 3H).
2-Bromomethyl-4-fluorobenzonitrile (32). A mixture of 4-fluoro-2-methylbenzonitrile (2 g, 14.8 mmol), NBS (2.64 g, 15 mmol) and AIBN (100 mg) in CCl4 was refluxed under nitrogen for 2 hours. The reaction was cooled to room temperature. The solid was removed by filtration. The organic solution was concentrated to give crude product as an oil, which was used in the next step without further purification. 1H-NMR (400 MHz, CDCl3): δ 7.68 (dd, J= 5.2, 8.4 Hz, IH), 7.28 (dd, J= 2.4, 8.8 Hz, IH), 7.12 (m, IH), 4.6 (s, 2H).
Alternatively, 32 was made as follows. 4-Fluoro-2-methylbenzonitrile (1 kg) in DCE (2 L) was treated with AJJBN (122 g) and heated to 750C. A suspension of DBH (353 g) in DCE (500 mL) was added at 750C portionwise over 20 minutes. This operation was repeated 5 more times over 2.5 hours. The mixture was then stirred for one additional hour and optionally monitored for completion by, for example, measuring the amount of residual benzonitrile using HPLC. Additional AJ-BN (e.g., 12.5 g) was optionally added to move the reaction toward completion. Heating was stopped and the mixture was allowed to cool overnight. N,N-diisopropylethylamine (1.3 L) was added (at <10°C over 1.5 hours) and then diethyl phosphite (1.9 L) was added (at <20°C over 30 min). The mixture was then stirred for 30 minutes or until completion. The mixture was then washed with 1% sodium metabisulfite solution (5 L) and purified with water (5 L). The organic phase was concentrated under vacuum to afford 32 as a dark brown oil (3328 g), which was used without further purification (purity was 97% (AUC)).
2-(6-Chloro-3-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-l-ylmethyl)-4-fluoro-benzonitrile (33). A mixture of crude 3-methyl-6-chlorouracil (0.6 g, 3.8 mmol), 2-bromomethyl-4-fluorobenzonitrile (0.86 g, 4 mmol) and K2CO3 (0.5 g, 4 mmol) in DMSO (10 mL) was stirred at 6O0C for 2 hours. The reaction was diluted with water and extracted with EtOAc. The organics were dried over MgSO4 and the solvent removed. The residue was purified by column chromatography. 0.66 g of the product was obtained (yield: 60%). 1H-NMR (400 MHz, CDCl3): δ 7.73 (dd, 1=1.2, 8.4Hz, IH), 7.26 (d, J-4.0Hz, IH), 7.11-7.17 (m, IH), 6.94 (dd, J=2.0, 9.0 Hz, IH), 6.034 (s, 2H), 3.39 (s, 3H). MS (ES) [m+H] calc’d for C13H9ClFN3O2, 293.68; found 293.68.
Alternatively, 33 was made as follows. To a solution of 6-chloro-3-methyluracil (750 g) and W,iV-diisopropylethylarnine (998 mL) in NMP (3 L) was added (at <30°C over 25 min) a solution of 32 (2963 g crude material containing 1300 g of 32 in 3 L of toluene). The mixture was then heated at 6O0C for 2 hours or until completion (as determined, for example, by HPLC). Heating was then stopped and the mixture was allowed to cool overnight. Purified water (3.8 L) was added, and the resultant slurry was stirred at ambient temperature for 1 hour and at <5°C for one hour. The mixture was then filtered under vacuum and the wet cake was washed with IPA (2 X 2.25 L). The material was then dried in a vacuum oven at 40±5°C for 16 or more hours to afford 33 as a tan solid (>85% yield; purity was >99% (AUC)).
2-[6-(3-Amino-piperidin-l-yl)-3-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-l-ylmethyl]-4-fluoro-benzonitrile (34). 2-(6-Chloro-3-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-l-ylmethyl)-4-fluoro-benzonitrile (300 mg, 1.0 mmol), (R)-3-amino-piperidine dihydrochloride (266 mg, 1.5 mmol) and sodium bicarbonate (500 mg, 5.4 mmol) were stirred in a sealed tube in EtOH (3 mL) at 1000C for 2 hrs. The final compound was obtained as TFA salt after HPLC purification. 1H-NMR (400 MHz, CD3OD): δ. 7.77-7.84 (m, IH), 7.16-7.27 (m, 2H), 5.46 (s, IH), 5.17-5.34 (ABq, 2H, J = 35.2, 15.6 Hz), 3.33-3.47 (m, 2H), 3.22 (s, 3H), 2.98-3.08 (m, IH), 2.67-2.92 (m, 2H), 2.07-2.17 (m, IH), 1.82-1.92 (m, IH), 1.51-1.79 (m, 2H). MS (ES) [m+H] calc’d for C18H20FN5O2, 357.38; found, 357.38.
Alternatively, the free base of 34 was prepared as follows. A mixture of 33 (1212 g), IPA (10.8 L), (R)-3-amino-piperidine dihydrochloride (785 g), purified water (78 mL) and potassium carbonate (2.5 kg, powder, 325 mesh) was heated at 6O0C until completion (e.g., for >20 hours) as determined, for example, by HPLC. Acetonitrile (3.6 L) was then added at 6O0C and the mixture was allowed to cool to <25°C. The resultant slurry was filtered under vacuum and the filter cake was washed with acetonitrile (2 X 3.6 L). The filtrate was concentrated at 450C under vacuum (for >3 hours) to afford 2.6 kg of the free base of 34.
The HCl salt of 34 was prepared from the TFA salt as follows. The TFA salt (34) was suspended in DCM, and then washed with saturated Na2CO3. The organic layer was dried and removed in vacuo. The residue was dissolved in acetonitrile and HCl in dioxane (1.5 eq.) was added at 00C. The HCl salt was obtained after removing the solvent. 1H-NMR (400 MHz, CD3OD): δ. 7.77-7.84 (m, IH), 7.12-7.26 (m, 2H), 5.47 (s, IH), 5.21-5.32 (ABq, 2H, J = 32.0, 16.0 Hz), 3.35-3.5 (m, 2H), 3.22 (s, 3H), 3.01-3.1 (m, IH), 2.69-2.93 (m, 2H), 2.07-2.17 (m, IH), 1.83-1.93 (m, IH), 1.55-1.80 (m, 2H). MS (ES) [m+H] calc’d for C18H20FN5O2, 357.38; found, 357.38.
Alternatively, the HCl salt was prepared from the free base as follows. To a solution of free base in CH2Cl2 (12 L) was added (at <35°C over 18 minutes) 2 M hydrochloric acid (3.1 L). The slurry was stirred for 1 hour and then filtered. The wet cake was washed with CH2Cl2 (3.6 L) and then THF (4.8 L). The wet cake was then slurried in THF (4.8 L) for one hour and then filtered. The filter cake was again washed with THF (4.8 L). The material was then dried in a vacuum oven at 5O0C (with a nitrogen bleed) until a constant weight (e.g., >26 hours) to afford 34 as the HCl salt as a white solid (1423 g, >85% yield).
The succinate salt of 34 was prepared from the HCl salt as follows. To a mixture of the HCl salt of 34 (1414 g), CH2Cl2 (7 L) and purifed water (14 L) was added 50% NaOH solution (212 mL) until the pH of the mixture was >12. The biphasic mixture was stirred for 30 min and the organic layer was separated. The aqueous layer was extracted with CH2Cl2 (5.7 L) and the combined organic layers were washed with purified water (6 L). The organic layer was then passed through an in-line filter and concentrated under vacuum at 3O0C over three hours to afford the free base as an off-white solid. The free base was slurried in prefiltered THF (15 L) and prefiltered IPA (5.5 L). The mixture was then heated at 6O0C until complete dissolution of the free base was observed. A prefiltered solution of succinic acid (446 g) in THF (7 L) was added (over 23 min) while maintaining the mixture temperature at >57°C. After stirring at 6O0C for 15 min, the heat was turned off, the material was allowed to cool, and the slurry was stirred for 12 hours at 25±5°C. The material was filtered under vacuum and the wet cake was washed with prefiltered IPA (2 X 4.2 L). The material was then dried in a vacuum oven at 70±5°C (with a nitrogen bleed) for >80 hours to afford the succinate salt of 34 as a white solid (1546 g, >90% yield).
The product was also converted to a variety of corresponding acid addition salts. Specifically, the benzonitrile product (approximately 10 mg) in a solution of MeOH (1 mL) was treated with various acids (1.05 equivalents). The solutions were allowed to stand for three days open to the air. If a precipitate formed, the mixture was filtered and the salt dried. If no solid formed, the mixture was concentrated in vacuo and the residue isolated. In this way, salts of 34 were prepared from the following acids: benzoic, p-toluenesulfonic, succinic, R-(-)-Mandelic and benzenesulfonic. The succinate was found to be crystalline as determined by x-ray powder diffraction analysis.
In addition, the methanesulfonate salt was prepared as follows. A 10.5 g aliquot of the benzonitrile product was mixed with 400 mL of isopropylacetate. The slurry was heated to 75°C and filtered through #3 Whatman filter paper. The solution was heated back to 750C and a IM solution of methanesulfonic acid (30.84 mL) was added slowly over 10 minutes while stirring. The suspension was cooled to room temperature at a rate of about 20°C/hr. After 1 hr at room temperature, the solid was filtered and dried in an oven overnight to obtain the methanesulfonate salt.
PATENT
US 2008227798
http://www.google.com/patents/US20080227798
- EXAMPLES
- Example 1Preparation of 2-[6-(3-amino-piperidin-1-yl)-3-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-ylmethyl]-4-fluoro-benzonitrile succinate (Compound I)
A. Preparation of 4-fluoro-2-methylbenzonitrile (Compound B)
-
Compound B was prepared by refluxing a mixture of 2-bromo-5-fluoro-toluene (Compound A) (3.5 g, 18.5 mmol) and CuCN (2 g, 22 mmol) in DMF (100 mL) for 24 hours. The reaction was diluted with water and extracted with hexane. The organics were dried over MgSO4 and the solvent removed to give product B (yield 60%). 1H-NMR (400 MHz, CDCl3): δ 7.60 (dd, J=5.6, 8.8 Hz, 1H), 6.93-7.06 (m, 2H), 2.55 (s, 3H).
B. Preparation of 2-bromomethyl-4-fluorobenzonitrile (Compound C)
-
Compound C was prepared by refluxing a mixture of 4-fluoro-2-methylbenzonitrile (Compound B) (2 g, 14.8 mmol), N-bromosuccinimide (NBS) (2.64 g, 15 mmol) and azo-bis-isobutyronitrile (AIBN) (100 mg) in CCl4 under nitrogen for 2 hours. The reaction was cooled to room temperature. The solid was removed by filtration. The organic solution was concentrated to give the crude product the form of an oil, which was used in the next step without further purification. 1H-NMR (400 MHz, CDCl3): δ 7.68 (dd, J=5.2, 8.4 Hz, 1H), 7.28 (dd, J=2.4, 8.8 Hz, 1H), 7.12 (m, 1H), 4.6 (s, 2H).
C. Preparation of 2-(6-chloro-3-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-ylmethyl)-4-fluoro-benzonitrile (Compound D)
-
Compound E was prepared by stirring a mixture of crude 3-methyl-6-chlorouracil D (0.6 g, 3.8 mmol), 2-bromomethyl-4-fluorobenzonitrile (0.86 g, 4 mmol) and K2CO3 (0.5 g, 4 mmol) in DMSO (10 mL) at 60° C. for 2 hours. The reaction was diluted with water and extracted with EtOAc. The organics were dried over MgSO4 and the solvent removed. The residue was purified by column chromatography. 0.66 g of the product was obtained (yield: 60%). 1H-NMR (400 MHz, CDCl3): δ 7.73 (dd, J=7.2, 8.4 Hz, 1H), 7.26 (d, J=4.0 Hz, 1H), 7.11-7.17 (m, 1H), 6.94 (dd, J=2.0, 9.0 Hz, 1H), 6.034 (s, 2H), 3.39 (s, 3H). MS (ES) [m+H] calc’d for C13H9ClFN3O2, 293.68; found 293.68.
D. Preparation of 2-(6-chloro-3-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-ylmethyl)-4-fluoro-benzonitrile (Compound F)
-
Compound F was prepared by mixing and stirring 2-(6-chloro-3-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-ylmethyl)-4-fluoro-benzonitrile (Compound E) (300 mg, 1.0 mmol), (R)-3-amino-piperidine dihydrochloride (266 mg, 1.5 mmol) and sodium bicarbonate (500 mg, 5.4 mmol) in a sealed tube in EtOH (3 mL) at 100° C. for 2 hrs. The final compound was obtained as trifluoroacetate (TFA) salt after HPLC purification. 1H-NMR (400 MHz, CD3OD): δ. 7.77-7.84 (m, 1H), 7.16-7.27 (m, 2H), 5.46 (s, 1H), 5.17-5.34 (ABq, 2H, J=35.2, 15.6 Hz), 3.33-3.47 (m, 2H), 3.22 (s, 3H), 2.98-3.08 (m, 1H), 2.67-2.92 (m, 2H), 2.07-2.17 (m, 1H), 1.82-1.92 (m, 1H), 1.51-1.79 (m, 2H). MS (ES) [m+H] calc’d for C18H20FN5O2, 357.38; found, 357.38.
E. Preparation of Compound I: the succinic acid salt of 2-(6-Chloro-3-methyl-2,4-dioxo-3,4-dihydro-2H-pyrimidin-1-ylmethyl)-4-fluoro-benzonitrile
-
The TFA salt prepared in the above step (Example 1, Step D) was suspended in DCM, and then washed with saturated Na2CO3. The organic layer was dried and removed in vacuo. The benzonitrile product (approximately 10 mg) was dissolved in MeOH (1 mL) and to which succinic acid in THF (1.05 equivalents) was added. The solutions were allowed to stand for three days open to the air. If a precipitate formed, the solid was collected by filtration. If no solid formed, the mixture was concentrated in vacuo, and the succinate salt was obtained after removing the solvent. 1H-NMR (400 MHz, CD3OD): δ. 7.77-7.84 (m, 1H), 7.12-7.26 (m, 2H), 5.47 (s, 1H), 5.21-5.32 (ABq, 2H, J=32.0, 16.0 Hz), 3.35-3.5 (m, 2H), 3.22 (s, 3H), 3.01-3.1 (m, 1H), 2.69-2.93 (m, 2H), 2.07-2.17 (m, 1H), 1.83-1.93 (m, 1H), 1.55-1.80 (m, 2H). MS (ES) [m+H] calc’d for C18H20FN5O2, 357.38; found, 357.38.
-
Compound I such prepared was found to be crystalline as determined by x-ray powder diffraction analysis (FIG. 1). The crystal material was designated Form A.
| TABLE A | |||
| Approximate Solubilities of Compound I | |||
| Solubility | |||
| Solvent | (mg/mL)a | ||
| Acetone | 2 | ||
| Acetonitrile (ACN) | <1 | ||
| Dichloromethane (DCM) | <1 | ||
| Dimethyl Formamide (DMF) | 68 | ||
| 1,4-Dioxane | <1 | ||
| Ethanol (EtOH) | 2 | ||
| Ethyl Acetate (EtOAc) | <1 | ||
| di-Ethyl ether | <1 | ||
| Hexanes | <1 | ||
| 2-Propanol (IPA) | <1 | ||
| Methanol (MeOH) | 20 | ||
| Tetrahydrofuran (THF) | <1 | ||
| Toluene | <1 | ||
| Trifluoroethanol (TFE) | >200 | ||
| Water (H2O) | 51 | ||
| ACN:H2O (85:15) | 101 | ||
| EtOH:H2O (95:5) | 5 | ||
| IPA:H2O (88:12) | 11 | ||
| aApproximate solubilities are calculated based on the total solvent used to give a solution; actual solubilities may be greater because of the volume of the solvent portions utilized or a slow rate of dissolution. Solubilities are reported to the nearest mg/mL. | |||
PATENT
Reference Example 2
in the following formula 2, 2 – ((6 – ((3R) -3- amino-piperidin-1-yl) -3-methyl-2,4-dioxo-3,4-dihydropyrimidine -1 (2H ) – yl) shown in the following example of a production process of a methyl) -4-fluoro-benzonitrile succinate (4b).
[Formula 2]
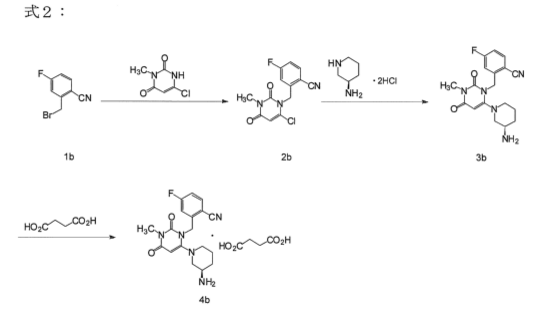
[In the formula 2, 2 – ((6-chloro-3-methyl-2,4-dioxo-3,4-dihydropyrimidine -1 (2H) – yl) methyl) -4-fluorobenzonitrile (2b) manufacturing process]
ethyl acetate (3.5 vol), 2- (bromomethyl) -4-fluorobenzonitrile (1b) (1 equiv, 1wt.), 6- chloro-3-methyl uracil (1.05 eq, 0.79wt), N- methylpyrrolidone (NMP;.. 3.5 times the amount), diisopropylethylamine (Hunig’s base, 2.1 eq, 1.27wt) was heated to an internal temperature of 60 ~ 70 ℃ a.
The mixture was stirred until 2-4 hours or the completion of the reaction at 60 ~ 70 ℃.
Then cooling the solution to 40 ~ 50 ℃, after stirring at least 30 minutes, 40 ~ 50 ℃ isopropanol (1.5 times) while maintaining, water (3.5 times the amount) was added, then at least one hour stirring did. The solution was cooled to 20 ~ 30 ℃, was then stirred for at least 1 hour. The solution was cooled to 0 ~ 10 ℃, was then stirred for at least 1 hour. The resulting slurry was filtered, washed with 0 ~ 10 ℃ in cold isopropanol (4.0 vol), and vacuum dried at 45 ~ 55 ℃, to give the above compound (2b).
[In the formula 2, 2 – ((6 – ((3R) -3- amino-piperidin-1-yl) -3-methyl-2,4-dioxo-3,4-dihydropyrimidine -1 (2H) – yl) methyl) -4-manufacturing process of the fluorobenzonitrile (3b)]
the above compound (2b) (1 eq, 1wt.), (R) -3- aminopiperidine dihydrochloride (1.1 eq, 0.65wt .), potassium carbonate (2.5 equivalents, 1.18wt.), isopropanol (5.0 vol), water (1.5 times) until the completion of the reaction with 65 ~ 75 ℃ (eg, 3 to 7 hours ) was allowed to react. Potassium carbonate in 65 ~ 75 ℃ (7.05 eq, 3.32wt.), Water (5.5 vol) was added, and after stirring for about 30 minutes, the phases were separated at 50 ℃ ~ 70 ℃. The organic solvent was concentrated under reduced pressure to approximately 5 times. And water (5 vol) was added to the solution and concentrated under reduced pressure to approximately 5 times. The solution was stirred for about 40 minutes at 55 ℃ ~ 75 ℃. The solution was cooled to 20 ℃ ~ 30 ℃, was then stirred for at least 1 hour. The solution was cooled to 0 ~ 10 ℃, subsequently stirred for at least 1 hour, the resulting slurry was filtered, washed with 0 ~ 10 ℃ in cold water (2.0 times the amount), 45 ~ 55 ℃ was vacuum dried to give the above compound (3b).
[In the above formula 2, the compound production step of succinate (4b) of (3b)]
Compound (3b), tetrahydrofuran (6.0 vol), isopropanol (3.0 vol), water (0. a 6-fold amount) was heated to 55 ~ 65 ℃. Tetrahydrofuran solution of succinic acid (20 ℃ ~ 30 ℃) was added and the solution was stirred for about 15 minutes and maintained at 55 ~ 65 ℃.
The solution was cooled to 20 ~ 30 ℃, the mixture was stirred for at least 1 hour. The solution was cooled to 0 ~ 10 ℃, was then stirred for at least 1 hour. After the resulting slurry filtered and washed with isopropanol (6.0 vol). The resulting wet crystals were dried at 65 ~ 75 ℃, was obtained succinate of the compound (3b) and (4b) as a white crystalline solid.
PATENT
http://www.google.com/patents/CN103030631A?cl=en
2 – ({6 -! [(3R) -3- amino-piperidin-1-yl] -3-methyl-dihydro-pyrimidin _3,4_ _2,4_ dioxo-1 (2 1) – yl} methyl) benzonitrile is an effective DPP-1V inhibitors class of drugs in recent years in Japan, the structural formula
As shown below.
Chinese Patent Application CN1926128 discloses a process for preparing 2_ ({6_ [(3R) -3- amino-piperidin-1-yl] -3-methyl-2,4-dioxo-3,4- dihydropyrimidine-1 (2 1!) – yl} methyl) benzonitrile method, as shown in Scheme I:
Scheme I
In the above reaction scheme, 6-chloro-uracil and 2-bromomethyl-benzene cyanide in a mixed solvent of DMF-DMSO, in the presence of NaH and LiBr alkylation reaction to give compound 2 in a yield of 54%. Compound 2 is further alkylation reaction of compound yield 3 is 72%. The total yield of the compound 4 prepared in 20% yield is low, and the preparation of compound 4 obtained purity is not high, but also the need for further purification, such as recrystallization, column chromatography and other means in order to obtain high-purity suitable Pharmaceutically acceptable 2 – ({6 – [(3R) -3- amino-piperidin-1-yl] -3-methyl-2,4-dioxo-3,4-dihydro-pyrimidin _1 (2! 1) – yl} methyl) benzonitrile compound. Preparation still find more suitable for industrial production, a higher yield of the 2- ({6- [(3R) -3- amino-piperidin-1-yl] -3-methyl-2,4-dioxo -3, (2Η) 4- dihydropyrimidine-1 – yl} methyl) benzonitrile or a salt or the like.
PATENT
Example 15
(R) -2 – ((6 (3-amino-piperidin-1-yl) -3-methyl-2,4-dioxo-3,4-dihydropyrimidine -1 (2H) – yl) methyl) synthesis of 4-fluoro-benzonitrile
100mL four-necked flask of water and isopropanol 1/1 (v / v) mixture 60mL was added, pyridine 21.4μL [d = 0.98, mw.79.10, 0.26mmol], (R) -1- (3- (2 – cyano-5-fluoro-benzyl) -1-methyl-2,6-dioxo-1,2,3,6-tetra-hydro-4-yl) piperidin-3-carboxamide 2.00g [mw.385.39, 5.19mmol] of It was added to the order. Then, iodobenzene diacetate 1.84g [mw.322.10, 5.71mmol] was added, and the mixture was stirred for 3 h at 20 ℃. After volatile components were distilled off under reduced pressure by an evaporator, and the aqueous solution was washed twice with ethyl acetate 20mL. After cooling to near 0 ℃, potassium carbonate 16g added stepwise at 15 ℃ or less, was extracted by the addition of toluene 6mL and isopropanol 6mL. After separation, the organic layer was washed with saturated brine 10mL, adding toluene 6mL after concentration under reduced pressure by an evaporator, and further subjected to vacuum concentration. It was suspended by the addition of toluene 6mL to concentrate, by the addition of n-heptane 6mL, after 1 hour and aged at 0 ℃, reduced pressure filtration, to obtain the desired compound after drying under reduced pressure at 50 ℃. White crystalline powder, 1.6g, 86% yield.
1 H-NMR (500 MHz, CDCl 3 ) delta (ppm) 1.23 (D, J = 11.03 Hz, 1H) 1.30 (BRS, 2H) 1.56-1.67 (M, 1H) 1.72-1.83 (M, 1H) 1.95 (dd , J = 12.77 Hz, 3.94 Hz, 1H) 2.41 (m, 1H) 2.61 (m, 1H) 2.87-2.98 (m, 2H) 2.99-3.05 (m, 1H) 3.32 (s, 3H) 5.23-5.32 (m , 2H) 5.39 (s, 1H) 6.86 (dd, J = 8.99 Hz, 2.36 Hz, 1H) 7.09 (td, J = 8.04 Hz, 2.52 Hz, 1H) 7.69 (dd, J = 8.51 Hz, 5.36 Hz, 1H ).
13 C NMR (126 MHz, CDCl 3 ) ppm 28.0, 33.4, 46.1, 51.9, 59.7, 90.8, 114.6,114.7, 115.6, 115.8, 116.4, 135.4, 135.5, 144.6, 152.7, 159.5, 162.9.
Reference Example 4
(R) -2 – ((6 (3-amino-piperidin-1-yl) -3-methyl-2,4-dioxo-3,4-dihydropyrimidine -1 (2H) – yl) methyl) synthesis of 4-fluoro-benzonitrile succinate

50mL eggplant-shaped flask (R) -2 – ((6- (3- amino-1-yl) -3-methyl-2,4-dioxo-3,4-dihydro-pyrimidine -1 (2H) – yl) methyl) -4-fluorobenzonitrile 1.0g [mw.357.38, 2.8mmol], it was added tetrahydrofuran 4.5mL and water 2 drops. After heated and dissolved at 65 ℃, was dropped to the solution was dissolved at the same temperature 0.331g succinic acid [mw.118.09, 2.8mmol] with tetrahydrofuran 4mL and isopropanol 2.5mL. Aged for 16 hours at room temperature after stirring for 30 min at 65 ℃, and stirred for a further 2 hours at 0 ℃. The crystallization product was collected by terrorism to vacuum filtration. To obtain the desired compound after drying under reduced pressure at 45 ℃. White crystalline powder, 1.2g, 93% yield.
1 H-NMR (500 MHz, DMSO) delta (ppm) 1.35 (D, J = 8.83 Hz, 1H) 1.42-1.57 (M, 1H) 1.66-1.97 (M, 2H) 2.54-2.77 (M, 2H) 2.91 ( d, J = 11.35 Hz, 1H) 3.00-3.07 (m, 1H) 3.08 (m, 1H) 3.09 (s, 3H) 3.14 (m, 1H) 5.12 (d, J = 16.08 Hz, 1H) 5.20 (d, J = 16.39 Hz, 1H) 5.38 (s, 1H) 7.17 (dd, J = 9.62 Hz, 2.36 Hz, 1H) 7.35 (td, J = 8.51 Hz, 2.52 Hz, 1H) 7.95 (dd, J = 8.67 Hz, 5.52 Hz, 1H).
13 C NMR (126 MHz, DMSO) delta ppm 27.9, 31.6, 46.3, 47.0, 51.7, 55.8, 90.3, 106.9, 115.7, 117.1, 136.45, 136.53, 145.8, 152.3, 159.7, 162.7, 164.1 , 166.1, 175.2.
PATENT
http://www.google.com/patents/CN102964196A?cl=en
PATENT
New Patent, Trelagliptin, SUN PHARMA
SUN PHARMACEUTICAL INDUSTRIES LIMITED [IN/IN]; Sun House, Plot No. 201 B/1 Western Express Highway Goregaon (E) Mumbai, Maharashtra 400 063 (IN)
BARMAN, Dhiren, Chandra; (IN).
NATH, Asok; (IN).
PRASAD, Mohan; (IN)
The present invention provides a process for the preparation of 4-fluoro-2- methylbenzonitrile of Formula (II), and its use for the preparation of trelagliptin or its salts. The present invention provides an efficient, simple, and commercially friendly process for the preparation of 4-fluoro-2-methylbenzonitrile, which is used as an intermediate for the preparation of trelagliptin or its salts. The present invention avoids the use of toxic and hazardous reagents, high boiling solvents, and bromo intermediates such as 2-bromo-5-fluorotoluene, which is lachrymatory in nature and thus difficult to handle at a commercial scale.
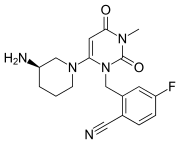
Trelagliptin is a dipeptidyl peptidase IV (DPP-IV) inhibitor, chemically designated as 2- [[6-[(3i?)-3 -aminopiperidin- 1 -yl] -3 -methyl -2,4-dioxopyrimidin- 1 -yljmethyl] -4-fluorobenzonitrile, represented by Formula I.

Formula I
Trelagliptin is administered as a succinate salt of Formula la, chemically designated as 2-[[6-[(3i?)-3-aminopiperidin-l-yl]-3-methyl-2,4-dioxopyrimidin-l-yl]methyl]-4-fluorobenzonitrile butanedioic acid (1 : 1).

Formula la
U.S. Patent Nos. 7,795,428, 8,288,539, and 8,222,411 provide a process for the preparation of 4-fluoro-2-methylbenzonitrile by reacting 2-bromo-5-fluorotoluene with copper (I) cyanide in N,N-dimethylformamide.
Chinese Patent No. CN 102964196 provides a process for the preparation of 4-fluoro-2-methylbenzonitrile by reacting 4-fluoro-2-methylbenzyl alcohol with cuprous iodide in the presence of 2,2′-bipyridine and 2,2,6,6-tetramethylpiperidine oxide (TEMPO) in an anhydrous ethanol.
Copper (I) cyanide is toxic to humans, and therefore its use in the manufacture of a drug substance is not advisable. In addition, 2-bromo-5-fluorotoluene is converted to 4-fluoro-2-methylbenzonitrile by refluxing in N,N-dimethylformamide at 152°C to 155°C for 24 hours. This leads to some charring, resulting in a tedious work-up process and low yield. Furthermore, the use of reagents like cuprous iodide, 2,2′-bipyridine, and 2,2,6,6-tetramethylpiperidine oxide (TEMPO) is hazardous and/or environmentally-unfriendly, and therefore their use in the manufacture of a drug substance is not desirable.
The present invention provides an efficient, simple, and commercially friendly process for the preparation of 4-fluoro-2-methylbenzonitrile, which is used as an intermediate for the preparation of trelagliptin or its salts. The present invention avoids the use of toxic and hazardous reagents, high boiling solvents, and bromo intermediates such as 2-bromo-5-fluorotoluene, which is lachrymatory in nature and thus difficult to handle at a commercial scale.
EXAMPLES
Example 1 : Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methylbenzaldehyde (1.38 g) was added to ethanol (10 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (2.76 g) and pyridine (1 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 3 hours. The solvent was recovered up to maximum extent from the reaction mixture under reduced pressure to afford the title compound. Yield: 3.1 g
Example 2: Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methylbenzaldehyde (5 g) was added to ethanol (37 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (10 g) and N,N-diisopropylethylamine (3.6 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 2 hours. The solvent was recovered up to maximum extent from the reaction mixture under reduced pressure to afford the title compound. Yield: 3.1 g
Example 3 : Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methylbenzaldehyde (10 g) was added to ethanol (40 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (20 g) and N,N-diisopropylethylamine (7.5 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 4 hours. The solvent was recovered from the reaction mixture under reduced pressure to afford the title compound. Yield: 11.0 g
Example 4: Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methylbenzaldehyde (50 g) was added to ethanol (500 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (70 g) and N,N-diisopropylethylamine (36 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 6 hours. The solvent was recovered from the reaction mixture under reduced pressure to afford the title compound. Yield: 51.0 g
Example 5 : Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methylbenzaldehyde (20 g) was added to ethanol (200 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (20 g) and N,N-diisopropylethylamine (18 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 4 hours. The solvent was recovered from the reaction mixture under reduced pressure to obtain a residue. Deionized water (60 mL) was charged into the residue, and then the slurry was stirred at 0°C to 5°C for 1 hour. The solid obtained was filtered, then washed with deionized water (2 x 20 mL). The wet solid was dried in an air oven at 40°C to 45 °C for 4 hours to 5 hours. The crude product obtained was recrystallized in ethanol (50 mL) to afford the pure title compound. Yield: 21.0 g
Example 6: Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methyl benzaldehyde (50 g) was added to ethanol (500 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (50 g) and N,N-diisopropylethylamine (46.4 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 4 hours. The solvent was recovered from the reaction mixture under reduced pressure to obtain a residue. Deionized water (150 mL) was charged to the residue, and then the slurry was stirred at 0°C to 5°C for 1 hour. The solid obtained was filtered, then washed with deionized water (2 x 50 mL). The wet solid was dried in an air oven at 40°C to 45 °C for 4 hours to 5 hours. The crude product obtained was recrystallized in ethanol (200 mL) to afford the pure title compound. Yield: 53.5 g
Example 7: Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methylbenzaldoxime (3.1 g) and phosphorous pentoxide (1 g) were added to toluene (30 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C for 24 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25°C to 30°C. Deionized water (30 mL) was added to the mixture and then the layers were separated. The organic layer was concentrated under reduced pressure to afford the title compound. Yield: 1.1 g
Example 8: Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methylbenzaldoxime (3 g) and phosphorous pentoxide (2 g) were added to toluene (30 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C for 24 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25°C to 30°C. Deionized water (30 mL) was added to the mixture and then the layers were separated. The organic layer was concentrated under reduced pressure to afford the title compound. Yield: 1.0 g
Example 9: Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methylbenzaldoxime (5 g) and concentrated sulphuric acid (2 mL) were added to toluene (100 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C for 5 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25°C to 30°C. Deionized water (50 mL) was added to the mixture and then the layers were separated. The organic layer was concentrated under reduced pressure to afford the title compound. Yield: 3.24 g
Example 10: Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methylbenzaldoxime (25 g) and concentrated sulphuric acid (35 g) were added to toluene (500 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C for 6 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25°C to 30°C. Deionized water (250 mL) was added to the mixture and then the layers were separated. The organic layer was concentrated under reduced pressure to afford the title compound. Yield: 20.5 g
Example 11 : Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methyl benzaldoxime (5 g) and sodium bisulphate monohydrate (3.1 g) were added to toluene (50 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C for 12 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25°C to 30°C, then filtered, and then washed with toluene (10 mL). The filtrate was concentrated under reduced pressure to afford the title compound. Yield: 3.0 g
Example 12: Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methyl benzaldoxime (50 g) and sodium bisulphate monohydrate (31.6 g) were added to toluene (500 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C using a Dean-Stark apparatus for 12 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25 °C to 30°C, then filtered, and then washed with toluene (100 mL). The filtrate was concentrated under reduced pressure to afford a crude product. The crude product obtained was recrystallized in a mixture of toluene (200 mL) and hexane (500 mL) to afford the title compound.
Yield: 38.0 g

Sun Pharma managing director Dilip Shanghvi.
References
- 1 McKeage, Kate (2015-06-27). “Trelagliptin: First Global Approval”. Drugs 75 (10): 1161–1164. doi:10.1007/s40265-015-0431-9. ISSN 0012-6667.
- 2 Inagaki, Nobuya; Onouchi, Hitoshi; Sano, Hiroki; Funao, Nobuo; Kuroda, Shingo; Kaku, Kohei. “SYR-472, a novel once-weekly dipeptidyl peptidase-4 (DPP-4) inhibitor, in type 2 diabetes mellitus: a phase 2, randomised, double-blind, placebo-controlled trial”. The Lancet Diabetes & Endocrinology 2 (2): 125–132. doi:10.1016/s2213-8587(13)70149-9.
- 3″New Drug Application Approval of Zafatek® Tablets for the treatment of Type 2 Diabetes in Japan | Takeda Pharmaceutical Company Limited”. http://www.takeda.com. Retrieved 2015-11-13.
- 4 Cahn, Avivit; Raz, Itamar (2013-06-01). “Emerging gliptins for type 2 diabetes”. Expert Opinion on Emerging Drugs 18 (2): 245–258. doi:10.1517/14728214.2013.807796. ISSN 1472-8214. PMID 23725569.
- 5Inagaki, Nobuya; Onouchi, Hitoshi; Maezawa, Hideaki; Kuroda, Shingo; Kaku, Kohei. “Once-weekly trelagliptin versus daily alogliptin in Japanese patients with type 2 diabetes: a randomised, double-blind, phase 3, non-inferiority study”. The Lancet Diabetes & Endocrinology 3 (3): 191–197. doi:10.1016/s2213-8587(14)70251-7.
- 6″TRELAGLIPTIN SUCCINATE | C22H26FN5O6 – PubChem”. pubchem.ncbi.nlm.nih.gov. Retrieved 2015-11-13.
- 7″Trelagliptin succinate | C22H26FN5O6 | ChemSpider”. http://www.chemspider.com. Retrieved 2015-11-13.
http://www.cbijournal.com/paper-archive/may-june-2014-vol-3/Review-Paper-1.pdf
| Patent | Submitted | Granted |
|---|---|---|
| TABLET [US2012129878] | 2010-07-27 | 2012-05-24 |
| AROMATIC RING COMPOUND [US2015045378] | 2013-02-12 | 2015-02-12 |
| Patent | Submitted | Granted |
|---|---|---|
| Combination therapy for the treatment of diabetes and related conditions [US2011263617] | 2011-10-27 | |
| Treatment for diabetes in patients with insufficient glycemic control despite therapy with an oral or non-oral antidiabetic drug [US2011275561] | 2011-11-10 | |
| Treatment for diabetes in patients with inadequate glycemic control despite metformin therapy comprising a DPP-IV inhibitor [US2011301182] | 2011-12-08 | |
| COATED PREPARATION [US2010166853] | 2008-07-10 | 2010-07-01 |
| Solid preparation comprising 2-[[6-[(3R)-3-amino-1-piperidinyl]-3,4-dihydro-3-methyl-2,4-dioxo-1(2H)-pyrimidinyl]methyl]-4-fluorobenzonitrile [US7994183] | 2008-03-12 | 2011-08-09 |
| Diabetes therapy [US2012165251] | 2011-06-23 | 2012-06-28 |
| MEDICAL USE OF A DPP-4 INHIBITOR [US2014371243] | 2014-06-13 | 2014-12-18 |
| TREATMENT OF GENOTYPED DIABETIC PATIENTS WITH DPP-IV INHIBITORS SUCH AS LINAGLIPTIN [US2013196898] | 2010-11-26 | 2013-08-01 |
| ANTIDIABETIC MEDICATIONS COMPRISING A DPP-4 INHIBITOR (LINAGLIPTIN) OPTIONALLY IN COMBINATION WITH OTHER ANTIDIABETICS [US2012094894] | 2010-02-12 | 2012-04-19 |
| DPP-IV INHIBITORS FOR TREATMENT OF DIABETES IN PEDIATRIC PATIENTS [US2012122776] | 2010-01-29 | 2012-05-17 |
| Patent | Submitted | Granted |
|---|---|---|
| LAMINATED TABLET AND MANUFACTURING METHOD THEREFOR [US2014023708] | 2012-03-02 | 2014-01-23 |
| Combination therapy for the treatment of diabetes and related conditions [US2013310398] | 2013-07-24 | 2013-11-21 |
| USE OF KERATINOCYTES AS A BIOLOGICALLY ACTIVE SUBSTANCE IN THE TREATMENT OF WOUNDS, SUCH AS DIABETIC WOUNDS, OPTIONALLY IN COMBINATION WITH A DPP-4 INHIBITOR [US2013315975] | 2013-05-23 | 2013-11-28 |
| USE OF A DPP-4 INHIBITOR IN AUTOIMMUNE DIABETES, PARTICULARLY LADA [US2013317046] | 2013-05-21 | 2013-11-28 |
| USE OF A DPP-4 INHIBITOR FOR MODIFYING FOOD INTAKE AND REGULATING FOOD PREFERENCE [US2013324463] | 2013-05-21 | 2013-12-05 |
| COMBINATION THERAPY [US2013281373] | 2011-05-05 | 2013-10-24 |
| USE OF A DPP-4 INHIBITOR IN PODOCYTES RELATED DISORDERS AND/OR NEPHROTIC SYNDROME [US2013303462] | 2013-05-13 | 2013-11-14 |
| USE OF A DPP-4 INHIBITOR IN SIRS AND/OR SEPSIS [US2013303554] | 2013-05-13 | 2013-11-14 |
| Combination of a GPR119 Agonist and the DPP-IV Inhibitor Linagliptin for Use in the Treatment of Diabetes and Related Conditions [US2013109703] | 2011-03-18 | 2013-05-02 |
| Treatment for diabetes in patients inappropriate for metformin therapy [US2011263493] | 2011-10-27 |
| Patent | Submitted | Granted |
|---|---|---|
| DIPEPTIDYL PEPTIDASE INHIBITORS [US7781584] | 2008-07-03 | 2010-08-24 |
| POLYMORPHS OF SUCCINATE SALT OF 2-[6-(3-AMINO-PIPERIDIN-1-YL)-3-METHYL-2,4-DIOXO-3,4-DIHYDRO-2H-PYRIMIDIN-1-YLMETHY]-4-FLUOR-BENZONITRILE AND METHODS OF USE THEREFOR [US2008227798] | 2008-09-18 | |
| GPR119 receptor agonists in methods of increasing bone mass and of treating osteoporosis and other conditions characterized by low bone mass, and combination therapy relating thereto [US7816364] | 2009-10-29 | 2010-10-19 |
| DIPEPTIDYL PEPTIDASE INHIBITORS [US8222411] | 2009-11-05 | 2012-07-17 |
| ADMINISTRATION OF DIPEPTIDYL PEPTIDASE INHIBITORS [US2008287476] | 2008-11-20 | |
| POLYMORPHS OF SUCCINATE SALT OF 2-[6-(3-AMINO-PIPERIDIN-1-YL)-3-METHYL-2,4-DIOXO-3,4-DIHYDRO-2H-PYRIMIDIN-1-YLMETHY]-4-FLUOR-BENZONITRILE AND METHODS OF USE THEREFOR [US8084605] | 2008-11-13 | 2011-12-27 |
| WEEKLY ADMINISTRATION OF DIPEPTIDYL PEPTIDASE INHIBITORS [US8093236] | 2008-11-06 | 2012-01-10 |
| Therapeutic Agent for Diabetes [US2009042863] | 2009-02-12 | |
| ADMINISTRATION OF DIPEPTIDYL PEPTIDASE INHIBITORS [US2007060530] | 2007-03-15 | |
| DIPEPTIDYL PEPTIDASE INHIBITORS [US7795428] | 2008-01-03 | 2010-09-14 |
| Patent | Submitted | Granted |
|---|---|---|
| Dipeptidyl peptidase inhibitors [US7807689] | 2005-11-24 | 2010-10-05 |
| DIPEPTIDYL PEPTIDASE INHIBITORS [US2008108807] | 2008-05-08 | |
| DIPEPTIDYL PEPTIDASE INHIBITORS [US2008108808] | 2008-05-08 | |
| FUSED CYCLIC COMPOUNDS [US7732626] | 2010-01-07 | 2010-06-08 |
| DIPEPTIDYL PEPTIDASE INHIBITORS [US7906523] | 2008-08-07 | 2011-03-15 |
| DIPEPTIDYL PEPTIDASE INHIBITORS [US8188275] | 2008-07-24 | 2012-05-29 |
| DIPEPTIDYL PEPTIDASE INHIBITORS [US8173663] | 2009-01-08 | 2012-05-08 |
| ADMINISTRATION OF DIPEPTIDYL PEPTIDASE INHIBITORS [US2011077402] | 2011-03-31 | |
| DPP-IV INHIBITORS FOR USE IN THE TREATMENT OF NAFLD [US2011092510] | 2011-04-21 | |
| PURIN DERIVATIVES FOR USE IN THE TREATMENT OF FAB-RELATED DISEASES [US2011190322] | 2011-08-04 |
| Patent | Submitted | Granted |
|---|---|---|
| Administration of Dipeptidyl Peptidase Inhibitors [US2011192748] | 2011-08-11 | |
| PHARMACEUTICAL COMPOSITION COMPRISING A GLUCOPYRANOSYL-SUBSTITUTED BENZENE DERIVATE [US2011195917] | 2011-08-11 | |
| DPP-IV INHIBITOR COMBINED WITH A FURTHER ANTIDIABETIC AGENT, TABLETS COMPRISING SUCH FORMULATIONS, THEIR USE AND PROCESS FOR THEIR PREPARATION [US2011206766] | 2011-08-25 | |
| COMBINATION OF A CERTAIN DPP-4 INHIBITOR AND VOGLIBOSE [US2014343014] | 2014-05-16 | 2014-11-20 |
| CARDIO- AND RENOPROTECTIVE ANTIDIABETIC THERAPY [US2014274889] | 2014-03-14 | 2014-09-18 |
| TREATMENT FOR DIABETES IN PATIENTS INAPPROPRIATE FOR METFORMIN THERAPY [US2014274890] | 2014-06-03 | 2014-09-18 |
| Fused ring compound and use thereof [US2010190747] | 2010-07-29 | |
| FUSED RING COMPOUND AND USE THEREOF [US2010197683] | 2010-08-05 | |
| Fused cyclic compounds [US8088821] | 2010-08-05 | 2012-01-03 |
| GPR119 Receptor Agonists in Methods of Increasing Bone Mass and of Treating Osteoporosis and Other Conditions Characterized by Low Bone Mass, and Combination Therapy Relating Thereto [US8101626] | 2010-07-29 | 2012-01-24 |
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
Succinic acid – 2-({6-[(3R)-3-amino-1-piperidinyl]-3-methyl-2,4-dioxo-3,4-dihydro-1(2H)-pyrimidinyl}methyl)-4-fluorobenzonitrile (1:1)
|
|
| Clinical data | |
| Trade names | Zafatek |
| Chemical data | |
| Formula | C22H26FN5O6 |
| Molar mass | 475.470143 g/mol |
/////////Trelagliptin, PMDA, JAPAN 2015
Cn1c(=O)cc(n(c1=O)Cc2cc(ccc2C#N)F)N3CCC[C@H](C3)N
WO 2016025720, New Patent, by Assia Chemicals and Teva on Ibrutinib


WO 2016025720, New Patent, by Assia Chemicals and Teva on Ibrutinib
ASSIA CHEMICAL INDUSTRIES LTD. [IL/IL]; 2 Denmark Street 49517 Petach Tikva (IL)
TEVA PHARMACEUTICALS USA, INC. [US/US]; 1090 Horsham Road P.O. Box 1090 North Wales, PA 19454 (US)
COHEN, Meital; (IL).
COHEN, Yuval; (IL).
MITTELMAN, Ariel; (IL).
MOHA-LERMAN, Elana, Ben; (IL).
TZANANI, Idit; (IL).
LEVENFELD, Leonid; (IL)
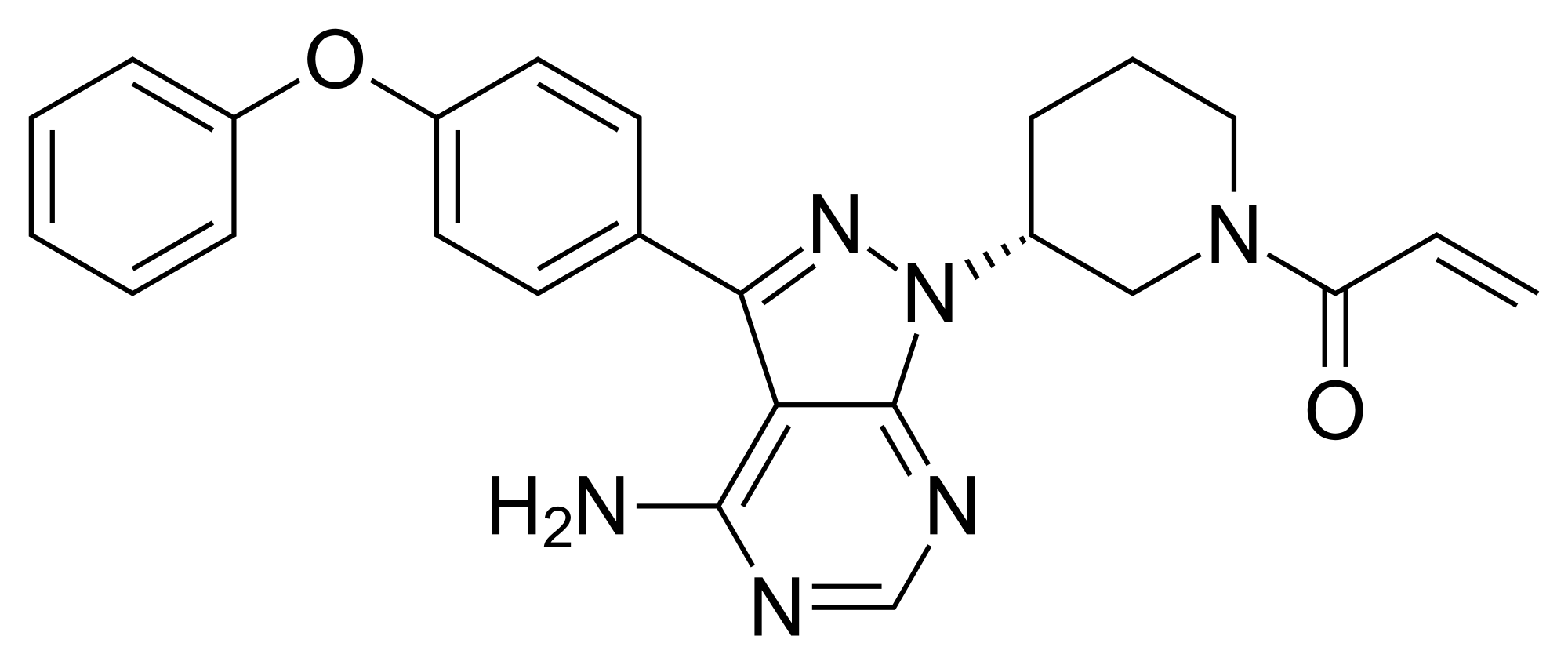
The present invention encompasses solid state forms of Ibrutinib, including forms G, J and K, and pharmaceutical compositions thereof.
Ibrutinib, l-{(3R)-3- [4-amino-3-(4-phenoxyphenyl)-lH-pyrazolo [3,4-d] pyrimidin-l-yl] piperidin-l-yl] prop-2-en-l-one, having the following formula,

is a kinase inhibitor indicated for the treatment of patients with B-cell lymphoma.
Ibrutinib is described in US 7,514,444 and in US 8,008,309. Solid state forms, including forms A-F and amorphous form of Ibrutinib, are described in WO 2013/184572.
Polymorphism, the occurrence of different crystalline forms, is a property of some molecules and molecular complexes. A single molecule may give rise to a variety of polymorphs having distinct crystal structures and physical properties like melting point, thermal behaviors (e.g. measured by thermogravimetric analysis – “TGA”, or differential scanning calorimetry – “DSC”), X-ray diffraction pattern, infrared absorption fingerprint, and solid state (13C-) NMR spectrum. One or more of these techniques may be used to distinguish different polymorphic forms of a compound.
Different salts and solid state forms (including solvated forms) of an active pharmaceutical ingredient may possess different properties. Such variations in the properties of different salts and solid state forms and solvates may provide a basis for improving formulation, for example, by facilitating better processing or handling characteristics, changing the dissolution profile in a favorable direction, or improving stability (polymorph as well as chemical stability) and shelf-life. These variations in the properties of different salts and solid state forms may also offer improvements to the final dosage form, for instance, if they serve to improve bioavailability. Different salts and solid state forms and solvates of an active pharmaceutical ingredient may also give rise to a variety of polymorphs or crystalline forms, which may in turn provide additional opportunities to assess variations in the properties and characteristics of a solid active pharmaceutical ingredient.
Discovering new solid state forms and solvates of a pharmaceutical product may yield materials having desirable processing properties, such as ease of handling, ease of processing, storage stability, and ease of purification, or may serve as desirable intermediate crystal forms that facilitate purification or conversion to other polymorphic forms. New solid state forms of a pharmaceutically useful compound can also provide an opportunity to improve the performance characteristics of a pharmaceutical product. It enlarges the repertoire of materials that a formulation scientist has available for formulation optimization, for example by providing a product with different properties, e.g., a different crystal habit, higher crystallinity or polymorphic stability which may offer better processing or handling characteristics, improved dissolution profile, or improved shelf-life (chemical/physical stability). For at least these reasons, there is a need for additional solid state forms (including solvated forms) of ibrutinib.
Example 1: Preparation of Crystalline Form G of Ibrutinib
[0057] Ibrutinib (0.3 gr, amorphous form) was dissolved in acetic acid (1.2 ml) and the obtained solution was stirred at room temperature overnight followed by the addition of water (2.4 ml). A gum was obtained which was turned into cloudy solution upon stirring. The obtained cloudy solution was stirred for 9 days at room temperature and the obtained precipitate was collected by suction filtration. The obtained solid was dried in an oven at 40°C under vacuum for 16h to obtain form G of Ibrutinib (0.12g), as confirmed by XRPD.
Example 2: Preparation of Crystalline Form J of Ibrutinib
Ibrutinib (5.2 g) was dissolved in Anisole (15 ml), the solution was stirred at room temperature until precipitation was occurred. The slurry was stirred over night at room temperature and the precipitate was collected by suction filtration. The cake was dried in a vacuum oven at 50°C overnight. The obtained product was analyzed by XRPD and found to be form J.
Example 3: Preparation of Crystalline Form J of Ibrutinib
Ibrutinib (10.5 g) was dissolved in Anisole (21 ml) and MTBE (32 ml), the solution was stirred at room temperature until precipitation was occurred . The slurry was heated to reflux and was gradually cooled to room temperature. After 3 hours the precipitate was collected by suction filtration. The obtained product was analyzed by XRPD and found to be form J.
Example 4: Preparation of Crystalline Form G of Ibrutinib
A I L reactor was charged with Ibrutinib (100 g), acetonitrile (417.5 ml_), water (417.5 ml_) and acetic acid (27.15 g). The mixture was heated to 90°C until dissolution; the solution was gradually cooled to 0°C, then heated to 25°C and stirred over 48 hours at 25°C. The obtained slurry was filtered and washed with water (100 ml_). The product was dried overnight in a vacuum oven at 40°C to obtain Ibrutinib form G (72.9 g), as confirmed by XRPD.
Example 5: Preparation of Crystalline Form G of Ibrutinib
A 250 mL round flask was charged with isopropanol (10 ml_) and water (120 ml_), and a solution of Ibrutinib (10 g) in Acetic acid (40 mL) was added dropwise. The mixture was stirred at 25°C for 48 hours. The obtained slurry was filtered and the wet product was slurried in water (50 mL) for 5 min and filtered again. The obtained product was dried under vacuum at room temp in the presence of a N2 atmosphere and found to be form G, as confirmed by XRPD.
Example 6: Preparation of Crystalline Form K of Ibrutinib
Ibrutinib (10 g) was dissolved in toluene (50 mL) and dimethylformamide (DMA) (30 mL) at room temperature, the solution was heated to 50 °C and water (30 mL) was added. The phases were separated and methyl tert-butyl ether (MTBE) (30 mL) was added to the organic phase. The solution was cooled in an ice bath and seeded with amorphous Ibrutinib. After further stirring at the same temperature the obtained slurry was filtered under vacuum. The obtained solid was analyzed by XRPD and found to be Form K (Figure 5).

//////////////WO 2016025720, WO-2016025720, New Patent, Assia Chemicals, Teva, Ibrutinib
WO 2016024224, New Patent, Trelagliptin, SUN PHARMA


WO 2016024224, New Patent, Trelagliptin, SUN PHARMA
SUN PHARMACEUTICAL INDUSTRIES LIMITED [IN/IN]; Sun House, Plot No. 201 B/1 Western Express Highway Goregaon (E) Mumbai, Maharashtra 400 063 (IN)
BARMAN, Dhiren, Chandra; (IN).
NATH, Asok; (IN).
PRASAD, Mohan; (IN)
The present invention provides a process for the preparation of 4-fluoro-2- methylbenzonitrile of Formula (II), and its use for the preparation of trelagliptin or its salts. The present invention provides an efficient, simple, and commercially friendly process for the preparation of 4-fluoro-2-methylbenzonitrile, which is used as an intermediate for the preparation of trelagliptin or its salts. The present invention avoids the use of toxic and hazardous reagents, high boiling solvents, and bromo intermediates such as 2-bromo-5-fluorotoluene, which is lachrymatory in nature and thus difficult to handle at a commercial scale.
Trelagliptin is a dipeptidyl peptidase IV (DPP-IV) inhibitor, chemically designated as 2- [[6-[(3i?)-3 -aminopiperidin- 1 -yl] -3 -methyl -2,4-dioxopyrimidin- 1 -yljmethyl] -4-fluorobenzonitrile, represented by Formula I.

Formula I
Trelagliptin is administered as a succinate salt of Formula la, chemically designated as 2-[[6-[(3i?)-3-aminopiperidin-l-yl]-3-methyl-2,4-dioxopyrimidin-l-yl]methyl]-4-fluorobenzonitrile butanedioic acid (1 : 1).

Formula la
U.S. Patent Nos. 7,795,428, 8,288,539, and 8,222,411 provide a process for the preparation of 4-fluoro-2-methylbenzonitrile by reacting 2-bromo-5-fluorotoluene with copper (I) cyanide in N,N-dimethylformamide.
Chinese Patent No. CN 102964196 provides a process for the preparation of 4-fluoro-2-methylbenzonitrile by reacting 4-fluoro-2-methylbenzyl alcohol with cuprous iodide in the presence of 2,2′-bipyridine and 2,2,6,6-tetramethylpiperidine oxide (TEMPO) in an anhydrous ethanol.
Copper (I) cyanide is toxic to humans, and therefore its use in the manufacture of a drug substance is not advisable. In addition, 2-bromo-5-fluorotoluene is converted to 4-fluoro-2-methylbenzonitrile by refluxing in N,N-dimethylformamide at 152°C to 155°C for 24 hours. This leads to some charring, resulting in a tedious work-up process and low yield. Furthermore, the use of reagents like cuprous iodide, 2,2′-bipyridine, and 2,2,6,6-tetramethylpiperidine oxide (TEMPO) is hazardous and/or environmentally-unfriendly, and therefore their use in the manufacture of a drug substance is not desirable.
The present invention provides an efficient, simple, and commercially friendly process for the preparation of 4-fluoro-2-methylbenzonitrile, which is used as an intermediate for the preparation of trelagliptin or its salts. The present invention avoids the use of toxic and hazardous reagents, high boiling solvents, and bromo intermediates such as 2-bromo-5-fluorotoluene, which is lachrymatory in nature and thus difficult to handle at a commercial scale.
EXAMPLES
Example 1 : Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methylbenzaldehyde (1.38 g) was added to ethanol (10 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (2.76 g) and pyridine (1 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 3 hours. The solvent was recovered up to maximum extent from the reaction mixture under reduced pressure to afford the title compound. Yield: 3.1 g
Example 2: Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methylbenzaldehyde (5 g) was added to ethanol (37 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (10 g) and N,N-diisopropylethylamine (3.6 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 2 hours. The solvent was recovered up to maximum extent from the reaction mixture under reduced pressure to afford the title compound. Yield: 3.1 g
Example 3 : Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methylbenzaldehyde (10 g) was added to ethanol (40 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (20 g) and N,N-diisopropylethylamine (7.5 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 4 hours. The solvent was recovered from the reaction mixture under reduced pressure to afford the title compound. Yield: 11.0 g
Example 4: Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methylbenzaldehyde (50 g) was added to ethanol (500 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (70 g) and N,N-diisopropylethylamine (36 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 6 hours. The solvent was recovered from the reaction mixture under reduced pressure to afford the title compound. Yield: 51.0 g
Example 5 : Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methylbenzaldehyde (20 g) was added to ethanol (200 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (20 g) and N,N-diisopropylethylamine (18 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 4 hours. The solvent was recovered from the reaction mixture under reduced pressure to obtain a residue. Deionized water (60 mL) was charged into the residue, and then the slurry was stirred at 0°C to 5°C for 1 hour. The solid obtained was filtered, then washed with deionized water (2 x 20 mL). The wet solid was dried in an air oven at 40°C to 45 °C for 4 hours to 5 hours. The crude product obtained was recrystallized in ethanol (50 mL) to afford the pure title compound. Yield: 21.0 g
Example 6: Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methyl benzaldehyde (50 g) was added to ethanol (500 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (50 g) and N,N-diisopropylethylamine (46.4 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 4 hours. The solvent was recovered from the reaction mixture under reduced pressure to obtain a residue. Deionized water (150 mL) was charged to the residue, and then the slurry was stirred at 0°C to 5°C for 1 hour. The solid obtained was filtered, then washed with deionized water (2 x 50 mL). The wet solid was dried in an air oven at 40°C to 45 °C for 4 hours to 5 hours. The crude product obtained was recrystallized in ethanol (200 mL) to afford the pure title compound. Yield: 53.5 g
Example 7: Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methylbenzaldoxime (3.1 g) and phosphorous pentoxide (1 g) were added to toluene (30 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C for 24 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25°C to 30°C. Deionized water (30 mL) was added to the mixture and then the layers were separated. The organic layer was concentrated under reduced pressure to afford the title compound. Yield: 1.1 g
Example 8: Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methylbenzaldoxime (3 g) and phosphorous pentoxide (2 g) were added to toluene (30 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C for 24 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25°C to 30°C. Deionized water (30 mL) was added to the mixture and then the layers were separated. The organic layer was concentrated under reduced pressure to afford the title compound. Yield: 1.0 g
Example 9: Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methylbenzaldoxime (5 g) and concentrated sulphuric acid (2 mL) were added to toluene (100 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C for 5 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25°C to 30°C. Deionized water (50 mL) was added to the mixture and then the layers were separated. The organic layer was concentrated under reduced pressure to afford the title compound. Yield: 3.24 g
Example 10: Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methylbenzaldoxime (25 g) and concentrated sulphuric acid (35 g) were added to toluene (500 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C for 6 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25°C to 30°C. Deionized water (250 mL) was added to the mixture and then the layers were separated. The organic layer was concentrated under reduced pressure to afford the title compound. Yield: 20.5 g
Example 11 : Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methyl benzaldoxime (5 g) and sodium bisulphate monohydrate (3.1 g) were added to toluene (50 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C for 12 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25°C to 30°C, then filtered, and then washed with toluene (10 mL). The filtrate was concentrated under reduced pressure to afford the title compound. Yield: 3.0 g
Example 12: Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methyl benzaldoxime (50 g) and sodium bisulphate monohydrate (31.6 g) were added to toluene (500 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C using a Dean-Stark apparatus for 12 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25 °C to 30°C, then filtered, and then washed with toluene (100 mL). The filtrate was concentrated under reduced pressure to afford a crude product. The crude product obtained was recrystallized in a mixture of toluene (200 mL) and hexane (500 mL) to afford the title compound.
Yield: 38.0 g

Sun Pharma managing director Dilip Shanghvi.
/////////////WO 2016024224, New Patent, Trelagliptin, SUN PHARMA
Fidaxomicin, フィダキソマイシン

Fidaxomicin (C52H74Cl2O18, Mr = 1058.0 g/mol)
Launched – 2011 MERCK, Clostridium difficile-associated diarrhea
CUBIST ….INNOVATOR
OPT-80
PAR-101
Also tiacumicin B or lipiarmycin A3,
A bacterial RNA polymerase inhibitor as macrocyclic antibiotic used to treat clostridium difficile-associated diarrhea (CDAD).

SYNTHESIS

REFERENCES
US 4918174
WO 2006085838
J ANTIBIOTICS 1987, 40, PG 567-574 AND 575-588
Idaxomicin(trade names Dificid, Dificlir, and previously OPT-80 and PAR-101) is the first in a new class of narrow spectrum macrocyclic antibiotic drugs.[2] It is a fermentation product obtained from the actinomycete Dactylosporangium aurantiacum subspecies hamdenesis.[3][4] Fidaxomicin is non-systemic, meaning it is minimally absorbed into the bloodstream, it is bactericidal, and it has demonstrated selective eradication of pathogenic Clostridium difficile with minimal disruption to the multiple species of bacteria that make up the normal, healthy intestinal flora. The maintenance of normal physiological conditions in the colon can reduce the probability of Clostridium difficile infection recurrence.[5] [6]
Fidaxomicin is an antibiotic approved and launched in 2011 in the U.S. for the treatment of Clostridium difficile-associated diarrhea (CDAD) in adults 18 years of age and older. In September 2011, the product received a positive opinion in the E.U. and final approval was assigned in December 2011.
First E.U. launch took place in the U.K. in June 2012. Optimer Pharmaceuticals, now part of Cubist (now, Merck & Co.), is conducting phase III clinical trials for the prevention of Clostridium difficile-associated diarrhea in patients undergoing hematopoietic stem cell transplant
In 2014 Astellas initiated in Europe a phase III clinical study for the treatment of Clostridium difficile infection in pediatric patients. Preclinical studies are ongoing for potential use in the prevention of methicillin-resistant Staphylococcus (MRS) infection.


The compound is a novel macrocyclic antibiotic that is produced by fermentation. Its narrow-spectrum activity is highly selective for C. difficile, thus preserving gut microbial ecology, an important consideration for the treatment of CDAD.
It is marketed by Cubist Pharmaceuticals after acquisition of its originating company Optimer Pharmaceuticals. The target use is for treatment of Clostridium difficile infection.
In May 2005, Par Pharmaceutical and Optimer entered into a joint development and collaboration agreement for fidaxomicin. However, rights to the compound were returned to Optimer in 2007. The compound was granted fast track status by the FDA in 2003. In 2010, orphan drug designation was assigned to fidaxomicin in the U.S. by Optimer Pharmaceuticals for the treatment of pediatric Clostridium difficile infection (CDI). In 2011, the compound was licensed by Optimer Pharmaceuticals to Astellas Pharma in Europe and certain countries in the Middle East, Africa, the Commonwealth of Independent States (CIS) and Japan for the treatment of CDAD. In 2011, fidaxomicin was licensed to Cubist by Optimer Pharmaceuticals for comarketing in the U.S. for the treatment of CDAD. In July 2012, the product was licensed by Optimer Pharmaceuticals to Specialised Therapeutics Australia in AU and NZ for the treatment of Clostridium difficile-associated infection. OBI Pharma holds exclusive commercial rights in Taiwan, where the compound was approved for the treatment of CDAD in September 2012, and in December 2012, the product was licensed to AstraZeneca in South America with commercialization rights also for the treatment of CDAD. In October 2013, Optimer Pharmaceuticals was acquired by Cubist.
Fidaxomicin is available in a 200 mg tablet that is administered every 12 hours for a recommended duration of 10 days. Total duration of therapy should be determined by the patient’s clinical status. It is currently one of the most expensive antibiotics approved for use. A standard course costs upwards of £1350.[7]
Fidaxomicin (also known as OPT-80 and PAR-101 ) is a novel antibiotic agent and the first representative of a new class of antibacterials called macrocycles. Fidaxomicin is a member of the tiacumicin family, which are complexes of 18-membered macrocyclic antibiotics naturally produced by a strain of Dactylosporangium aurantiacum isolated from a soil sample collected in Connecticut, USA.
The major component of the tiacumicin complex is tiacumicin B. Optically pure R-tiacumicin B is the most active component of Fidaxomicin. The chiral center at C(19) of tiacumicinB affects biological activity, and R-tiacumicin B has an R-hydroxyl group attached at this position. The isomer displayed significantly higher activity than other tiacumicin B-related compounds and longer post-antibiotic activity.
Tiacumicins are a family of structurally related compounds that contain the 18-membered macrolide ring shown below.
At present, several distinct Tiacumicins have been identified and six of these
(Tiacumicin A-F) are defined by their particular pattern of substituents R1, R2, and R3 (US Patent No. 4,918,174; J. Antibiotics, 1987, 575-588).
The Lipiarmycins are a family of natural products closely related to the Tiacumicins. Two members of the Lipiarmycin family (A3 and B3) are identical to Tiacumicins B and C respectively (J. Antibiotics, 1988, 308-315; J. Chem. Soc. Perkin Trans 1, 1987, 1353-1359).
The Tiacumicins and the Lipiarmycins have been characterized by numerous physical methods. The reported chemical structures of these compounds are based on spectroscopy (UV-vis, IR and !H and 13C NMR), mass spectrometry and elemental analysis (See for example: J. Antibiotics, 1987, 575-588; J. Antibiotics, 1983, 1312-
1322).
Tiacumicins are produced by bacteria, including Dactylosporangium aurantiacum subspecies hamdenensis, which may be obtained from the ARS Patent Collection of the Northern Regional Research Center, United States Department ofAgriculture, 1815 North University Street, Peoria, IL 61604, accession number NRRL
18085. The characteristics of strain AB 718C-41 are given in J. Antibiotics, 1987,567-574 and US Patent No. 4,918,174.
Lipiarmycins are produced by bacteria including Actinoplanes deccanensis (US Patent No. 3,978,211). Taxonomical studies of type strain A/10655, which has been deposited in the ATCC under the number 21983, are discussed in J. Antibiotics,1975, 247-25.
Tiacumicins, specifically Tiacumicin B, show activity against a variety of bacterial pathogens and in particular against Clostridium difficile, a Gram-positive bacterium (Antimicrob. Agents Chemother. 1991, 1108-1111). Clostridium difficile is an anaerobic spore-forming bacterium that causes an infection of the bowel.
As per WIPO publication number 2006085838, Fidaxomicin is an isomeric mixture of the configurationally distinct stereoisomers of tiacumicin B, composed of 70 to 100% of R-tiacumicin B and small quantities of related compounds, such as S-tiacumicin B and lipiarmycin A4. Fidaxomicin was produced by fermentation of the D aurantiacum subspecies hamdenensis (strain 718C-41 ). It has a narrow spectrum antibacterial profile mainly directed against Clostridium difficile and exerts a moderate activity against some other gram-positive species.
Fidaxomicin is bactericidal and acts via inhibition of RNA synthesis by bacterial RNA polymerase at a distinct site from that of rifamycins. The drug product is poorly absorbed and exerts its activity in the gastrointestinal (Gl) tract, which is an advantage when used in the applied indication, treatment of C. difficile infection (CDI) (also known as C. difficile-associated disease or diarrhoea [CDAD]). Fidaxomicin is available as DIFICID oral tablet in US market.
Its CAS chemical name is Oxacyclooctadeca-3,5,9, 13, 15-pentaen-2-one, 3-[[[6-deoxy-4-0-(3,5dichloro-2-ethyl-4,6-dihydroxybenzoyl)-2-0-methyl-P-D-manno pyranosyl]oxy]methyl]-12[[6-deoxy-5-C-methyl-4-0-(2-methyl-1 -oxopropyl)- -D-lyxo-hexo pyranosyl]oxy]-1 1 -ethyl-8-hydroxy-18-[(1 R)-1 -hydroxyethyl] -9,13,15-trimethyl-, (3E.5E, 8S.9E.1 1 S.12R.13E, 15E.18S)-.
Structural formula (I) describes the absolute stereochemistry of fidaxomicin as determined by x-ray.

(I)
WIPO publication number 2004014295 discloses a process for preparation of Tiacumicins that comprises fermentation of Dactylosporangium aurantiacum NRRL18085 in suitable culture medium. It also provides process for isolation of tiacumicin from fermentation broth using techniques selected from the group consisting of: sieving and removing undesired material by eluting with at least one solvent or a solvent mixture; extraction with at least one solvent or a solvent mixture; Crystallization; chromatographic separation; High-Performance Liquid Chromatography (HPLC); MPLC; trituration; and extraction with saturated brine with at least one solvent or a solvent mixture. The product was isolated from /so-propyl alcohol (IPA) having a melting point of 166-169 °C.
U.S. Patent No. 7378508 B2 discloses polymorphic forms A and B of fidaxomicin, solid dosage forms of the two forms and composition thereof. As per the ‘508 patent form A is obtained from methanol water mixture and Form B is obtained from ethyl acetate.
J. Antibiotics, vol. 40(5), 575-588 (1987) discloses purification of Tiacumicins using suitable solvents wherein tiacumicin B exhibited a melting point of 143-145 °C.
PCT application WO2013170142A1 describes three crystalline forms of Fidaxomicn namely, Form-Z, Form-Z1 and Form-C. IN2650/CHE/2013 describes 6 crystalline polymorphic forms of Fidaxomicin namely, Forms I, Form la, Form II, Form Ha, Form III and Form Ilia).
Mechanism
Fidaxomicin binds to and prevents movement of the “switch regions” of bacterial RNAP polymerase. Switch motion is important for opening and closing of the DNA:RNA clamp, a process that occurs throughout RNA transcription but especially during opening of double standed DNA during transcription initiation.[8] It has minimal systemic absorption and a narrow spectrum of activity; it is active against Gram positive bacteria especially clostridia. The minimal inhibitory concentration (MIC) range for C. difficile (ATCC 700057) is 0.03–0.25 μg/mL.[3]
Clinical trials
Good results were reported by the company in 2009 from a North American phase III trial comparing it with oral vancomycin for the treatment of Clostridium difficile infection (CDI)[9][10] The study met its primary endpoint of clinical cure, showing that fidaxomicin was non-inferior to oral vancomycin (92.1% vs. 89.8%). In addition, the study met its secondary endpoint of recurrence: 13.3% of the subjects had a recurrence with fidaxomicin vs. 24.0% with oral vancomycin. The study also met its exploratory endpoint of global cure (77.7% for fidaxomicin vs. 67.1% for vancomycin).[11] Clinical cure was defined as patients requiring no further CDI therapy two days after completion of study medication. Global cure was defined as patients who were cured at the end of therapy and did not have a recurrence in the next four weeks.[12]
Fidaxomicin was shown to be as good as the current standard-of-care, vancomycin, for treating CDI in a Phase III trial published in February 2011.[13] The authors also reported significantly fewer recurrences of infection, a frequent problem with C. difficile, and similar drug side effects.
Approvals and indications
For the treatment of Clostridium difficile-associated diarrhea (CDAD), the drug won an FDA advisory panel’s unanimous approval on April 5, 2011[14] and full FDA approval on May 27, 2011.[15]
PAPER
Enantioselective synthesis of putative lipiarmycin aglycon related to fidaxomicin/tiacumicin B
Angew Chem Int Ed 2015, 54(6): 1929
Dr. William Erb, Dr. Jean-Marie Grassot, Dr. David Linder, Dr. Luc Neuville and Prof. Dr. Jieping Zhu
Article first published online: 24 NOV 2014 | DOI: 10.1002/anie.201409475
Chain gang: In the synthesis of the title compound, the ene-diene ring-closing metathesis was used for the formation of the 18-membered macrolactone and the stereogenic centers of the molecule were installed by Brown’s alkoxyallylboration, allylation, and an Evans aldol reaction, while iterative Horner–Wadsworth–Emmons reactions were used for chain elongation.
http://onlinelibrary.wiley.com/doi/10.1002/anie.201409475/full
PAPER
Total synthesis of the glycosylated macrolide antibiotic fidaxomicin
Org Lett 2015, 17(14): 3514
http://pubs.acs.org/doi/abs/10.1021/acs.orglett.5b01602
http://pubs.acs.org/doi/suppl/10.1021/acs.orglett.5b01602/suppl_file/ol5b01602_si_001.pdf

The first enantioselective total synthesis of fidaxomicin, also known as tiacumicin B or lipiarmycin A3, is reported. This novel glycosylated macrolide antibiotic is used in the clinic for the treatment of Clostridium difficile infections. Key features of the synthesis involve a rapid and high-yielding access to the noviose, rhamnose, and orsellinic acid precursors; the first example of a β-selective noviosylation; an effective Suzuki coupling of highly functionalized substrates; and a ring-closing metathesis reaction of a noviosylated dienoate precursor. Careful selection of protecting groups allowed for a complete deprotection yielding totally synthetic fidaxomicin.

The identity of the synthetic compound to an authentic sample of fidaxomicin (1) was confirmed by coinjection on RP-HPLC and an equimolar mixed NMR-sample with an authentic sample. Rƒ = 0.44 (MeOH/CH2Cl2 1/10).
HRMS ESI calcd. for [C52H74Cl2NaO18] + [M+Na]+ : 1079.4144; found:1079.4151.
1H NMR (600 MHz, Methanol-d4 , containing HCOO- ) δ 7.23 (d, J = 11.5 Hz, 1H), 6.60 (dd, J = 14.9, 11.8 Hz 1H), 5.95 (ddd, J = 14.7, 9.5, 4.8 Hz, 1H), 5.83 (s, 1H), 5.57 (ap t, J = 8.2 Hz, 1H), 5.14 (ap d, J = 10.7, 1H), 5.13 (dd, J = 9.7 Hz, 1H), 5.02 (d, J = 10.2 Hz, 1H), 4.74-4.70 (m, 1H), 4.71 (s, 1H), 4.64 (s, 1H), 4.61 (d, J = 11.6 Hz, 1H), 4.44 (d, J = 11.6 Hz, 1H), 4.22 (ap s, 1H), 4.02 (p, J = 6.3 Hz, 1H), 3.92 (dd, J = 3.2, 1.2 Hz, 1H), 3.75 (ddd, J = 13.9, 10.2, 3.3 Hz, 1H) 3.71 (d, J = 9.7 Hz 1H), 3.58-3.52 (m, 2H) 3.54 (s, 3H), 3.15-3.06 (m, 1H), 3.04-2.95 (m, 1H), 2.76-2.66 (m, 3H), 2.60 (hept, J= 7.0 Hz, 1H), 2.49 (ddd, J = 14.9, 9.5, 4.4 Hz, 1H), 2.43 (ddd, J = 13.8, 8.8, 4.5 Hz, 1H), 2.05-1.98 (m, 1H), 1.82 (d, J = 1.3 Hz, 3H), 1.76 (ap s, 3H), 1.66 (ap s, 3H), 1.32-1.27 (m, 4H), 1.22-1.15 (m, 12H), 1.15 (s, 3H), 1.13 (s, 3H), 0.88 (t, J = 7.4 Hz, 3H).
RP-HPLC tR = 14.87 min (A: H2O+0.1% HCOOH; Solvent B: MeCN+0.1% HCOOH; 1 mL/min; T = 20°C; B[%] (tR [min])= 10 (0 to 3); 100 (15).
PATENT
WO 2004014295
http://www.google.co.in/patents/WO2004014295A2?cl=en
The term “Tiacumicin B” refers to molecule having the structure shown below:
Example 1
Dactylosporangium aurantiacum subsp. hamdenensis AB 718C-41 NRRL 18085 (-20 °C stock), was maintained on 1 mL of Medium No. 104 (Table 1). After standard sterilization conditions (30 min., 121 °C, 1.05 kg/cm2) the seed flask (250 mL) containing Medium No. 104 (50 mL) was inoculated with AB 718C-41 NRRL 18085 on a shaker (set @ 250 rpm) at 30 °C for 72 hr. Five percent vegetative inoculum from the first passage seed flask was then transferred aseptically to a fermentation flask containing the same ingredients as in Table 1.
Table 1: Ingredients of Medium No. 104
Fermentation flasks were incubated on a rotary shaker at 30 °C for 3 to 12 days. Samples of the whole culture fermentation broth were filtered. The filter cake was washed with MeOH and solvents were removed under reduced pressure. The residue was re-constituted in methanol to the same volume of the original fermentation broth. Analysis was performed using a Waters BREEZE HPLC system coupling with Waters 2487 2-channel UV/Vis detector. Tiacumincins were assayed on a 50 x 4.6 μm I.D., 5 μm YMC ODS-A column (YMC catalog # CCA AS05- 0546WT) with a mobile phase consisting of 45% acetonitrile in water containing 0.1% phosphoric acid at a flow rate of 1.5 mL/minute. Tiacumicins were detected at 266 nm. An HPLC chromatogram of a crude product (Tiacumicin B retention time @ 12.6 minutes) is shown in Fig. 1. In this example the crude yield of Tiacumicin B was about 250 mg/L after 7 days. After purification by HPLC, the yield of Tiacumicin B was about 100 mg/L.
Example 2
After standard sterilization conditions (30 min, 121 °C, 1.05 kg/cm2) the seed flask (250 mL) containing Medium No. 104 (50 mL) was inoculated with AB 718C- 41 NRRL 18085 and incubated on a shaker (set @ 250 rpm) at 30° C for 72 hr. Five percent vegetative inoculum from the first passage seed flask was transferred aseptically to a seed flask containing the same ingredients as in Table 1 and was incubated on a rotary shaker at 30 °C for 72 hr. Five percent inoculum from the second passage seed flasks was then used to inoculate with AB 718C-41 NRRL 18085 in a 5-liter fermenter containing Medium No. 104 (2.5 L). Excessive foam formation was controlled by the addition of an antifoaming agent (Sigma A-6426). This product is a mixture of non-silicone organic defoamers in a polyol dispersion.
Glucose consumption was monitored as a growth parameter and its level was controlled by the addition of the feeding medium. Feeding medium and conditions in Example 2 were as follows:
Feeding medium:
Fermenter Medium: No. 104
Fermenter Volume: 5 liters
Sterilization: 40 minutes, 121° C, 1.05 kg/cm2
Incubation Temperature: 30 °C.
Aeration rate: 0.5-1.5 volumes of air per culture volume and minute
Fermenter Agitation: 300-500 rpm
The fermentation was carried out for 8 days and the XAD-16 resin was separated from the culture broth by sieving. After washing with water the XAD-16 resin was eluted with methanol (5-10 x volume of XAD-16). Methanol was evaporated and the oily residue was extracted three times with ethyl acetate. The extracts were combined and concentrated under reduced pressure to an oily residue. The oily residue was dried and washed with hexane to give the crude product as a pale brown powder and its HPLC chromatogram (Tiacumincin B rete tion time @ 11.8 minutes) is shown in Figure 2. This was purified by silica gel column (mixture of ethyl acetate and hexane as eluent) and the resultant material was further purified by RP-HPLC (reverse phase HPLC) to give Tiacumicin B as a white solid. The purity was determined to be >95% by HPLC chromatography and the chromatogram (Tiacumincin B retention time @ 12.0 minutes) is shown in Figure 3. Analysis of the isolated Tiacumincin B gave identical !H and 13C NMR data to those reported in J. Antibiotics, 1987, 575-588, and these are summarized below. Tiacumicin B: mp 129-140 °C (white powder from RP-HPLC); mp 166-169 °C (white needles from isopropanol); [α]D 20-6.9 (c 2.0, MeOH); MS m/z (ESI) 1079.7(M + Na)+; H NMR (400 MHz, CD3OD) δ 7.21 (d, IH), 6.59 (dd, IH), 5.95 (ddd, IH), 5.83 (br s, IH), 5.57 (t, IH), 5.13 (br d, IH), 5.09 (t, IH), 5.02 (d, IH), 4.71 (m, IH), 4.71 (br s, IH), 4.64 (br s, IH), 4.61 (d, IH), 4.42 (d, IH), 4.23 (m, IH), 4.02 (pentet, IH), 3.92 (dd, IH), 3.73 (m, 2H), 3.70 (d, IH), 3.56 (s, 3H), 3.52-3.56 (m, 2H), 2.92 (m, 2H), 2.64-2.76 (m, 3H), 2.59 (heptet, IH), 2.49 (ddd, IH), 2.42 (ddd, IH), 2.01 (dq, IH), 1.81 (s, 3H), 1.76 (s, 3H), 1.65 (s, 3H), 1.35 (d, 3H), 1.29 (m, IH), 1.20 (t, 3H), 1.19 (d, 3 H), 1.17 (d, 3H), 1.16 (d, 3H), 1.14 (s, 3H), 1.12 (s, 3H), 0.87 (t, 3H); 13C NMR (100 MHz, CD3OD) δ 178.4, 169.7, 169.1, 154.6, 153.9, 146.2, 143.7, 141.9, 137.1, 137.0, 136.4, 134.6, 128.5, 126.9, 125.6, 124.6, 114.8, 112.8, 108.8, 102.3, 97.2, 94.3, 82.5, 78.6, 76.9, 75.9, 74.5, 73.5, 73.2, 72.8, 71.6, 70.5, 68.3, 63.9, 62.2, 42.5, 37.3, 35.4, 28.7, 28.3, 26.9, 26.4, 20.3, 19.6, 19.2, 18.7, 18.2, 17.6, 15.5, 14.6, 14.0, 11.4.
PATENT
http://www.google.com/patents/US7378508
macrolide of Formula I:
Structure of R-Tiacumicin B
The structure of the R-Tiacumicin B (the major most active component) is shown below in Formula I. The X-ray crystal structure of the R-Tiacumicin B was obtained as a colorless, parallelepiped-shaped crystal (0.08×0.14×0.22 mm) grown in aqueous methanol. This x-ray structure confirms the structure shown below. The official chemical name is 3-[[[6-Deoxy-4-O-(3,5-dichloro-2-ethyl-4,6-dihydroxybenzoyl)-2-O-methyl-β-D-mannopyranosyl]oxy]-methyl]-12(R)-[[6-deoxy-5-C-methyl-4-O-(2-methyl-1-oxopropyl)-β-D-lyxo-hexopyranosyl]oxy]-11(S)-ethyl-8(S)-hydroxy-18(S)-(1(R)-hydroxyethyl)-9,13,15-trimethyloxacyclooctadeca-3,5,9,13,15-pentaene-2-one.
7.2.1 Analytical Data of R-Tiacumicin B
The analytical data of R-Tiacumicin B (which is almost entirely (i.e., >90%) R-Tiacumicin).
mp 166-169° C. (white needle from isopropanol);
[α]D 20-6.9 (c 2.0, MeOH);
MS m/z (ESI) 1079.7(M+Na)+;
1H NMR (400 MHz, CD3OD) δ 7.21 (d, 1H), 6.59 (dd, 1H), 5.95 (ddd, 1H), 5.83 (br s, 1H), 5.57 (t, 1H), 5.13 (br d, 1H), 5.09 (t, 1H), 5.02 (d, 1H), 4.71 (m, 1H), 4.71 (br s, 1H), 4.64 (br s, 1H), 4.61 (d, 1H), 4.42 (d, 1H), 4.23 (m, 1H), 4.02 (pentet, 1H), 3.92 (dd, 1H), 3.73 (m, 2H), 3.70 (d, 1H), 3.56 (s, 3H), 3.52-3.56 (m, 2H), 2.92 (m, 2H), 2.64-2.76 (m, 3H), 2.59 (heptet, 1H), 2.49 (ddd, 1H), 2.42 (ddd, 1H), 2.01 (dq, 1H), 1.81 (s, 3H), 1.76 (s, 3H), 1.65 (s, 3H), 1.35 (d, 3H), 1.29 (m, 1H), 1.20 (t, 3H), 1.19 (d, 3H), 1.17 (d, 3H), 1.16 (d, 3 H), 1.14 (s, 3H), 1.12 (s, 3H), 0.87 (t, 3H);
13C NMR (100 MHz, CD3OD) δ 178.4, 169.7, 169.1, 154.6, 153.9, 146.2, 143.7, 141.9, 137.1, 137.0, 136.4, 134.6, 128.5, 126.9, 125.6, 124.6, 114.8, 112.8, 108.8, 102.3, 97.2, 94.3, 82.5, 78.6, 76.9, 75.9, 74.5, 73.5, 73.2, 72.8, 71.6, 70.5, 68.3, 63.9, 62.2, 42.5, 37.3, 35.4, 28.7, 28.3, 26.9, 26.4, 20.3, 19.6, 19.2, 18.7, 18.2, 17.6, 15.5, 14.6, 14.0, 11.4.
EXAMPLES
Example 1; General procedure for the preparation of crude Fidaxomycin
Fidaxomycin was prepared by:
i) culturing a microorganism in a nutrient medium to accumulate Fidaxomycin in the nutrient medium;
ii) isolating crude Fidaxomycin from the nutrient medium by methods known from the art;
iii) purifying Fidaxomycin by reversed phase chromatography using a mixture of acetonitrile, water and acetic acid as eluent; and iv) isolating the purified Fidaxomycin from the fractions.
Actionplanes deccanenesis was used during the cultivation. The nutrient medium comprises the following combination based on weight: from about 0% to about 5% Sucrose; from about 0% to about 3% Starch; from about 0.1% to about 1.0 % Soy peptone; from about 2% to about 5% Cotton seed meal; from about 0.01% to about 0.1% Potassium-dihydrogen Phosphate; from about 0.05% to about 0.5% Dipotassium-hydrogen Phosphate; from about 0.05% to about 0.5% Antifoam agent; from about 0% to about 2% Amberlite XAD-16N resin. The preferred temperature of the cultivation is from 28 to 32°C, and the pH is between 6.0 and 8.0. During the cultivation C-source is continuously fed.
The Fidaxomycin fermentation production can also be done by the following procedure:
The Fidaxomycin fermentation production can include a step of inoculation followed by fermentation as follows:
Inoculation: Actinoplanes deccanenesis strain is inoculated into the seed medium. The inoculation parameters are adjusted and maintained until the inoculum transferred to the main fermentation. The inoculum medium comprises: from about 0 to about 5% glucose, from about 0 to about 1% yeast extract, from about 0 to about 1% soy peptone, from about 0 to about 0.5% CaCo3, from about 0 to about 0.2% MgS0 -7H20, from about 0 to about 0.2% K2HP04, from about 0 to about 0.2% KC1, from about 0 to about 0.3% Polypropylene glycol. The pH is adjusted by adding Hydrochloric acid and/or Sodium/potassium hydroxide.
Inoculation parameters :
Inoculation time: 40-48 ± 24 hours.
At the end of the inoculation, the inoculum (or a part of it) is transferred into the sterile fermentation medium at a ratio of 8-15 ± 5 %.
Fermentation: the fermentation medium comprises: from about 0 to aboutl0% Sucrose/Hydrolyzed Starch, from about 0 to about 1% Soy peptone, from about 0 to about 5% Cotton seed meal, from about 0 to about 0.3% K2HP04, from about 0 to about 0.2% KH2P04, from about 0 to aboutl% KC1, from about 0 to about 0.5% Polypropylene glycol (PPG). The pH is adjusted by adding Hydrochloric acid and/or Sodium/potassium hydroxide.
The sterile fermentation medium is seeded with the inoculum.
Feeding:
C-source is fed during the fermentation, For C-source feeding sucrose or hydrolyzed-starch can be applied. Total amount of fed C-source is 0 – 15% related to the initial volume.
Fermentation parameters :
In case of foaming, sterile antifoaming agent should be added.
Fermentation time: 168-192 ± 24 hours.
The inoculation/fermentation medium may also include from about 0% to about 2% Amberlite XAD-16N resin.
Upon completion of fermentation, the Fidaxomycin is extracted from the fermented broth with an organic solvent such as, for example, ethyl acetate, isobutyl acetate or isobutanol. The organic phase is concentrated and the Fidaxomycin is precipitated by addition of an antisolvent such as, for example, n-hexane. Optionally the precipitate can be suspended in a second antisolvent. After filtration and drying, crude Fidaxomycin is obtained.
DIFICID (fidaxomicin) is a macrolide antibacterial drug for oral administration. Its CAS chemical name is Oxacyclooctadeca-3,5,9,13,15-pentaen-2-one, 3-[[[6-deoxy-4-O-(3,5-dichloro-2-ethyl-4,6-dihydroxybenzoyl)-2-Omethyl- β-D- mannopyranosyl]oxy]methyl]-12-[[6-deoxy-5-C-methyl-4-O-(2-methyl-1-oxopropyl)-β-D-lyxohexopyranosyl] oxy]-11-ethyl-8 -hydroxy-18-[(1R)-1-hydroxyethyl]-9,13,15-trimethyl-,(3E,5E,8S,9E,11S,12R,13E,15E,18S)-. The structural formula of fidaxomicin is shown in Figure 1.
Figure 1: Structural Formula of Fidaxomicin



Patent
WO 2016024243, New patent, Dr Reddy’s Laboratories Ltd, Fidaxomicin
WO2016024243, FIDAXOMICIN POLYMORPHS AND PROCESSES FOR THEIR PREPARATION
DR. REDDY’S LABORATORIES LIMITED [IN/IN]; 8-2-337, Road No. 3, Banjara Hills, Telangana State, India Hyderabad 500034 (IN)
CHENNURU, Ramanaiah; (IN).
PEDDY, Vishweshwar; (IN).
RAMAKRISHNAN, Srividya; (IN)
Aspects of the present application relate to crystalline forms of Fidaxomicin IV, V & VI and processes for their preparation. Further aspects relate to pharmaceutical compositions comprising these polymorphic forms of fidaxomicin
The occurrence of different crystal forms, i.e., polymorphism, is a property of some compounds. A single molecule may give rise to a variety of polymorphs having distinct crystal structures and physico-chemical properties.
Polymorphs are different solid materials having the same molecular structure but different molecular arrangement in the crystal lattice, yet having distinct physico-chemical properties when compared to other polymorphs of the same molecular structure. The discovery of new polymorphs and solvates of a pharmaceutical active compound provides an opportunity to improve the performance of a drug product in terms of its bioavailability or release profile in vivo, or it may have improved stability or advantageous handling properties. Polymorphism is an unpredictable property of any given compound. This subject has been reviewed in recent articles, including A. Goho, “Tricky Business,” Science News, August 21 , 2004. In general, one cannot predict whether there will be more than one form for a compound, how many forms will eventually be discovered, or how to prepare any previously unidentified form.
There remains a need for additional polymorphic forms of fidaxomicin and for processes to prepare polymorphic forms in an environmentally-friendly, cost-effective, and industrially applicable manner.

G.V. Prasad, chairman, Dr Reddy’s Laboratories
EXAMPLES
Example 1 : Preparation of fidaxomicin Form IV:
Fidaxomicin (0.5 g) and a mixture of 1 ,4-Dioxane (10 mL), THF (10 ml) and water (20mL) were charged in Easy max reactor (Mettler Toledo). The reactor was set to temperature cycle with following parameters:
Starting temperature: 25 °C;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 25 °C over a period of 2 hours;
Temperature maintained at 25 °C for 6 hours.
After completion of temperature cycling process, the slurry was filtered under suction, followed by drying in air tray dryer (ATD) at 40°C to a constant weight to produce crystalline fidaxomicin form-IV.
Example 2: Preparation of fidaxomicin Form V:
Fidaxomicin (1 g) and a mixture of propylene glycol (10 mL) and water (20mL) were charged in Easy max reactor (Mettler Toledo). The reactor was set to temperature cycle with following parameters:
Starting temperature is 25 °C;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 25 °C over a period of 2 hours;
Temperature maintained at 25 °C for 6 hours.
After completion of temperature cycling process, the slurry was filtered under suction, followed by drying in air tray dryer (ATD) at 40°C to a constant weight to produce crystalline fidaxomicin form-V.
Example 3: Preparation of fidaxomicin Form VI:
Fidaxomicin (0.5 mg) and MIBK (10 mL) were charged in Easy max reactor (Mettler Toledo) and the mixture was heated to 80°C. n-heptane (20 mL) was added to the solution at the same temperature. The mixture was stirred for 1 hour. The reaction mass was then cooled to 25°C. Solid formed was filtered at 25°C and dried at 40°C in air tray dryer (ATD) to a constant weight to produce crystalline fidaxomicin form VI.
Example 4: Preparation of fidaxomicin Form V:
Fidaxomicin (500 mg) and a mixture of R-propylene glycol (5 mL) and water (15 mL) were charged in Easy max reactor (Mettler Toledo). The reactor was set to temperature cycle with following parameters:
Starting temperature is 25 °C;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 25 °C over a period of 2 hours;
Temperature maintained at 25 °C for 2 hours.
After completion of temperature cycling process, the slurry was filtered and dried at 25°C to produce crystalline fidaxomicin form-V.
Example 5: Preparation of fidaxomicin Form V:
Fidaxomicin (1 g) and a mixture of S-propylene glycol (3 ml_) and water (30 mL) were charged in Easy max reactor (Mettler Toledo). The reactor was set to temperature cycle with following parameters:
Starting temperature is 25 °C;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 25 °C over a period of 2 hours;
Temperature maintained at 25 °C for 2 hours.
After completion of temperature cycling process, the slurry was filtered and dried at 25°C to produce crystalline fidaxomicin form-V.
Example 6: Preparation of fidaxomicin Form V:
Fidaxomicin (40 g) and a mixture of propylene glycol (400 mL) and water (1600 mL) were charged in Chem glass reactor. The reactor was set to temperature cycle with following parameters:
Starting temperature is 25 °C;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 25 °C over a period of 2 hours;
Temperature maintained at 25 °C for 6 hours.
After completion of temperature cycling process, the slurry was filtered under suction, followed by drying in air tray dryer (ATD) at 40°C to a constant weight to produce crystalline fidaxomicin form-V.
The 10-member board at pharmaceutical major Dr Reddy’s thrives on diversity. Liberally sprinkled with gray hairs, who are never quite impressed with powerpoint presentations, “they want information to be pre-loaded so that the following discussions (at the board level) are fruitful,” says Satish Reddy, Chairman, Dr Reddy’s. That said, the company has now equipped its board members with a customized application (that runs on their tablets) to manage board agenda and related processes.
see at

Dr. Reddy’s Laboratories Managing Director and Chief Operating Officer Satish Reddy addressing
References
- 1 “DIFICID” (PDF). TGA eBusiness Services. Specialised Therapeutics Australia Pty Ltd. 23 April 2013. Retrieved 31 March 2014.
- 2 Revill, P.; Serradell, N.; Bolós, J. (2006). “Tiacumicin B”. Drugs of the Future 31 (6): 494. doi:10.1358/dof.2006.031.06.1000709.
- 3″Dificid, Full Prescribing Information” (PDF). Optimer Pharmaceuticals. 2013.
- 4 “Fidaxomicin”. Drugs in R&D 10: 37. 2012. doi:10.2165/11537730-000000000-00000.
- 5Louie, T. J.; Emery, J.; Krulicki, W.; Byrne, B.; Mah, M. (2008). “OPT-80 Eliminates Clostridium difficile and is Sparing of Bacteroides Species during Treatment of C. Difficile Infection”. Antimicrobial Agents and Chemotherapy 53 (1): 261–3. doi:10.1128/AAC.01443-07. PMC 2612159. PMID 18955523.
- 6Johnson, Stuart (2009). “Recurrent Clostridium difficile infection: A review of risk factors, treatments, and outcomes”. Journal of Infection 58 (6): 403–10. doi:10.1016/j.jinf.2009.03.010. PMID 19394704.
- 7http://www.medicinescomplete.com/mc/bnf/current/PHP18388-dificlir.htm#PHP18388-dificlir
- 8Srivastava, Aashish; Talaue, Meliza; Liu, Shuang; Degen, David; Ebright, Richard Y; Sineva, Elena; Chakraborty, Anirban; Druzhinin, Sergey Y; Chatterjee, Sujoy; Mukhopadhyay, Jayanta; Ebright, Yon W; Zozula, Alex; Shen, Juan; Sengupta, Sonali; Niedfeldt, Rui Rong; Xin, Cai; Kaneko, Takushi; Irschik, Herbert; Jansen, Rolf; Donadio, Stefano; Connell, Nancy; Ebright, Richard H (2011). “New target for inhibition of bacterial RNA polymerase: ‘switch region'”. Current Opinion in Microbiology 14 (5): 532–43. doi:10.1016/j.mib.2011.07.030. PMC 3196380. PMID 21862392.
- 9″Optimer’s North American phase 3 Fidaxomicin study results presented at the 49th ICAAC” (Press release). Optimer Pharmaceuticals. September 16, 2009. Retrieved May 7, 2013.
- 10″Optimer Pharmaceuticals Presents Results From Fidaxomicin Phase 3 Study for the Treatment” (Press release). Optimer Pharmaceuticals. May 17, 2009. Retrieved May 7, 2013.
- 11Golan Y, Mullane KM, Miller MA (September 12–15, 2009). Low recurrence rate among patients with C. difficile infection treated with fidaxomicin. 49th interscience conference on antimicrobial agents and chemotherapy. San Francisco.
- 12Gorbach S, Weiss K, Sears P; et al. (September 12–15, 2009). Safety of fidaxomicin versus vancomycin in treatment of Clostridium difficile infection. 49th interscience conference on antimicrobial agents and chemotherapy. San Francisco.
- 13Louie, Thomas J.; Miller, Mark A.; Mullane, Kathleen M.; Weiss, Karl; Lentnek, Arnold; Golan, Yoav; Gorbach, Sherwood; Sears, Pamela; Shue, Youe-Kong; Opt-80-003 Clinical Study, Group (2011). “Fidaxomicin versus vancomycin for Clostridium difficile infection”. New England Journal of Medicine 364 (5): 422–31. doi:10.1056/NEJMoa0910812. PMID 21288078.
- 14Peterson, Molly (Apr 5, 2011). “Optimer wins FDA panel’s backing for antibiotic fidaxomicin”. Bloomberg.
- 15Nordqvist, Christian (27 May 2011). “Dificid (fidaxomicin) approved for Clostridium difficile-associated diarrhea”. Medical News Today.
ARNONE A ET AL: “STRUCTURE ELUCIDATION OF THE MACROCYCLIC ANTIBIOTIC LIPIARMYCIN“, JOURNAL OF THE CHEMICAL SOCIETY, PERKIN TRANSACTIONS 1, CHEMICAL SOCIETY, LETCHWORTH; GB, 1 January 1987 (1987-01-01), pages 1353-1359, XP000578201, ISSN: 0300-922X, DOI: 10.1039/P19870001353
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
3-(((6-Deoxy-4-O-(3,5-dichloro-2-ethyl-4,6-dihydroxybenzoyl)-2-O-methyl-β-D-mannopyranosyl)oxy)-methyl)-12(R)-[(6-deoxy-5-C-methyl-4-O-(2-methyl-1-oxopropyl)-β-D-lyxo-hexopyranosyl)oxy]-11(S)-ethyl-8(S)-hydroxy-18(S)-(1(R)-hydroxyethyl)-9,13,15-trimethyloxacyclooctadeca-3,5,9,13,15-pentaene-2-one
|
|
| Clinical data | |
| Trade names | Dificid, Dificlir |
| Licence data | US FDA:link |
| Pregnancy category |
|
| Legal status | |
| Routes of administration |
Oral |
| Pharmacokinetic data | |
| Bioavailability | Minimal systemic absorption[1] |
| Biological half-life | 11.7 ± 4.80 hours[1] |
| Excretion | Urine (<1%), faeces (92%)[1] |
| Identifiers | |
| CAS Number | 873857-62-6 |
| ATC code | A07AA12 |
| PubChem | CID 11528171 |
| ChemSpider | 8209640 |
| UNII | Z5N076G8YQ |
| KEGG | D09394 |
| ChEBI | CHEBI:68590 |
| ChEMBL | CHEMBL1255800 |
| Synonyms | Clostomicin B1, lipiarmicin, lipiarmycin, lipiarmycin A3, OPT 80, PAR 01, PAR 101, tiacumicin B |
| Chemical data | |
| Formula | C52H74Cl2O18 |
| Molar mass | 1058.04 g/mol |
| US4918174 | 26 Sep 1986 | 17 Apr 1990 | Abbott Laboratories | Tiacumicin compounds |
| WO2009025439A1 * | 6 May 2008 | 26 Feb 2009 | Genotech Co Ltd | Method of extraction and yield-up of tricyclo compounds by adding a solid adsorbent resin as their carrier in fermentation medium |
| WO2014023616A1 * | 30 Jul 2013 | 13 Feb 2014 | Olon Spa | Procedure for the production of tiacumicin b |
| WO2014111254A1 | 14 Jan 2014 | 24 Jul 2014 | Astellas Pharma Europe Ltd | Composition of tiacumicin compounds |
| WO2015091851A1 | 18 Dec 2014 | 25 Jun 2015 | Xellia Pharmaceuticals Aps | Process for the preparation of tiacumicin |
| WO2015169451A1 | 11 May 2015 | 12 Nov 2015 | Astellas Pharma Europe Ltd | Treatment regimen tiacumicin compound |
| CN101128114B | 31 Jan 2005 | 28 Mar 2012 | 浩鼎生技公司 | 18-membered macrocycles and analogs thereof |
| CN102614207B * | 31 Jan 2005 | 13 Jan 2016 | 默克夏普&多梅有限公司 | 18元环大环化合物及其类似物 |
| EP1848273A1 * | 31 Jan 2005 | 31 Oct 2007 | Optimer Pharmaceuticals, Inc. | 18-membered macrocycles and analogs thereof |
| EP2070530A1 | 13 May 2005 | 17 Jun 2009 | Optimer Pharmaceuticals, Inc. | Treatment of diseases associated with the use of antibiotics |
| EP2125850A1 † | 22 Jan 2008 | 2 Dec 2009 | Optimer Pharmaceuticals, Inc. | Macrocyclic polymorphs, compositions comprising such polymorphs, and methods of use and manufacture thereof |
| EP2305244A1 | 13 May 2005 | 6 Apr 2011 | Optimer Pharmaceuticals, Inc. | Treatment of diseases associated with the use of antibiotics |
| EP2305245A1 | 13 May 2005 | 6 Apr 2011 | Optimer Pharmaceuticals, Inc. | Treatment of diseases associated with the use of antibiotics |
| EP2468761A1 | 22 Jan 2008 | 27 Jun 2012 | Optimer Pharmaceuticals, Inc. | Macrocyclic polymorphs, compositions comprising such polymorphs, and methods of use and manufacture thereof |
| US7378508 | 31 Jul 2007 | 27 May 2008 | Optimer Pharmaceuticals, Inc. | Polymorphic crystalline forms of tiacumicin B |
| US7863249 | 11 Apr 2008 | 4 Jan 2011 | Optimer Pharmaceuticals, Inc. | Macrolide polymorphs, compositions comprising such polymorphs, and methods of use and manufacture thereof |
| US7906489 | 31 Jul 2007 | 15 Mar 2011 | Optimer Pharmaceuticals, Inc. | 18-membered macrocycles and analogs thereof |
| US8044030 | 28 Nov 2008 | 25 Oct 2011 | Optimer Pharmaceuticals, Inc. | Antibiotic macrocycle compounds and methods of manufacture and use thereof |
| US8586551 | 31 Aug 2009 | 19 Nov 2013 | Optimer Pharmaceuticals, Inc. | 18-membered macrocycles and analogs thereof |
| US8859510 | 22 Jan 2008 | 14 Oct 2014 | Optimer Pharmaceuticals, Inc. | Macrocyclic polymorphs, compositions comprising such polymorphs, and methods of use and manufacture thereof |
| US8883986 | 4 Mar 2009 | 11 Nov 2014 | Optimer Pharmaceuticals, Inc. | Macrolide polymorphs, compositions comprising such polymorphs, and methods of use and manufacture thereof |
| US8916527 | 15 Mar 2013 | 23 Dec 2014 | Optimer Pharmaceuticals, Inc. | Antibiotic macrocycle compounds and methods of manufacture and use thereof |
| US20110166090 * | 7 Jul 2011 | Youe-Kong Shue | 18-Membered Macrocycles and Analogs Thereof | |
| US20140107054 * | 21 Dec 2012 | 17 Apr 2014 | Optimer Pharmaceuticals, Inc. | Method of treating clostridium difficile-associated diarrhea |
| US3978211 * | Oct 31, 1974 | Aug 31, 1976 | Gruppo Lepetit S.P.A. | Lipiarmycin and its preparation |
| US4918174 | Sep 26, 1986 | Apr 17, 1990 | Abbott Laboratories | Tiacumicin compounds |
| US5583115 | May 9, 1995 | Dec 10, 1996 | Abbott Laboratories | Dialkyltiacumicin compounds |
| US5767096 | Jul 12, 1996 | Jun 16, 1998 | Abbott Laboratories | Bromotiacumicin compounds |
| US20060257981 * | Jul 15, 2003 | Nov 16, 2006 | Optimer Pharmaceuticals, Inc. | Tiacumicin production |
| US20070173462 * | May 13, 2005 | Jul 26, 2007 | Optimer Pharmaceuticals, Inc. | Treatment of diseases associated with the use of antibiotics |
| WO2004014295A2 | Jul 15, 2003 | Feb 19, 2004 | Optimer Pharmaceuticals Inc | Tiacumicin production |
| WO2005112990A2 | May 13, 2005 | Dec 1, 2005 | Optimer Pharmaceuticals Inc | Treatment of diseases associated with the use of antibiotics |
| WO2006085838A1 * | Jan 31, 2005 | Aug 17, 2006 | Optimer Pharmaceuticals Inc | 18-membered macrocycles and analogs thereof |
| DE2455230A1 * | Nov 21, 1974 | May 28, 1975 | Lepetit Spa | Lipiarmycin, verfahren zu seiner herstellung, mikroorganismus zur durchfuehrung des verfahrens und arzneimittel |
| EP2125850A1 | Jan 22, 2008 | Dec 2, 2009 | Optimer Pharmaceuticals, Inc. | Macrocyclic polymorphs, compositions comprising such polymorphs, and methods of use and manufacture thereof |
| US7378508 | Jul 31, 2007 | May 27, 2008 | Optimer Pharmaceuticals, Inc. | Polymorphic crystalline forms of tiacumicin B |
| Braga et al., “Making crystals from crystals: a green route tocrystal engineering and polymorphism” Chemical Communications (2005) pp. 3635-3645. | ||
| 2 | * | Chemical Abstracts registry entry 56645-60-4, Tiacumicin B, Copyright 2007, American Chemical Society, p. 1-2. |
| 3 | * | Dean, J., Analytical Chemistry Handbook, Published bt McGraw-Hill, Inc., pp. 10.23-10.26. |
| 4 | J.E. Hochlowski et al., Tiacumicins, A Novel Complex of 18-Membered Macrolides, J. Antibiotics, vol. XL, No. 5, pp. 575-588 (May 1987). | |
| 5 | * | Jain et al., “Polymorphism in Pharmacy” Indian Drugs (1986) vol. 23, No. 6, pp. 315-329. |
| 6 | * | Pharmaceutical Dosage Forms: Tablets, vol. 2, Published by Marcel Dekker, Inc., ed. by Lieberman, Lachman, and Schwartz, pp. 462-472. |
| 7 | * | Polymorphism in Pharmaceutical Solids, published 1999 by Marcel Dekker Inc, ed. by Harry G. Brittain, pp. 1-2. |
| 8 | Robert N. Swanson et al., In Vitro and In Vivo Evaluation of Tiacumicins B and C against Clostridium difficile, Antimicrob. Agents Chemother., Jun. 1991, pp. 1108-1111. | |
| 9 | * | The Condensed Chemical Dictionary, Tenth Edition, published 1981 by the Van Nostrand Reinhold Company, revised by Gessner G. Hawley, p. 35 and 835. |

///////////Fidaxomicin, OPT-80, PAR-101, japan 2018
CC[C@H]1/C=C(/[C@H](C/C=C/C=C(/C(=O)O[C@@H](C/C=C(/C=C(/[C@@H]1O[C@H]2[C@H]([C@H]([C@@H](C(O2)(C)C)OC(=O)C(C)C)O)O)\C)\C)[C@@H](C)O)\CO[C@H]3[C@H]([C@H]([C@@H]([C@H](O3)C)OC(=O)C4=C(C(=C(C(=C4O)Cl)O)Cl)CC)O)OC)O)\C
Fidaxomicin
-
- Synonyms:OPT-80; PAR-101; Tiacumicin B
- ATC:A07AA12
- Use:macrocyclic, antibotic, RNA polymerase inhibitor
- Chemical name:3-(((6-Deoxy-4-O-(3,5-dichloro-2-ethyl-4,6-dihydroxybenzoyl)-2-O-methyl-β-D-mannopyranosyl)oxy)-methyl)-12(R)-[(6-deoxy-5-C-methyl-4-O-(2-methyl-1-oxopropyl)-β-D-lyxo-hexopyranosyl)oxy]-11(S)-ethyl-8(S)-hydroxy-18(S)-(1(R)-hydroxyethyl)-9,13,15-trimethyloxacyclooctadeca-3,5,9,13,15-pentaene-2-one
- Formula:C52H74Cl2O8
- MW:1058.0 g/mol
- CAS-RN:873857-62-6
Substance Classes
Enzymes
Synthesis Path
Trade Names
| Country | Trade Name | Vendor | Annotation |
|---|---|---|---|
| USA | Dificid | Optimer Pharmceuticals, 2011 |
Formulations
- tabl. 200 mg
References
-
- WO 2004 014295 (Optimer Pharmaceuticals; 19.2.2004; USA-prior. 29.7.2002).
- US 7 507 564 (Optimer Pharmaceuticals; 24.3.2009; USA-prior. 29.7.2002).
- US 7 378 508 (Optimer Pharmaceuticals; 27.5.2008; USA-prior. 22.7.2007).
- US 3 978 211 (Gruppo Lepetit; 31.10.1974; GB-prior. 22.11.1973).
- US 4 918 174 (Abbott Laboratories; 17.10.1990; USA-prior. 26.9.1986).
- EP 923 594 (Abbott Laboratories; 2.10.2002; USA-prior. 12.7.1996).
WO 2016024243, New patent, Dr Reddy’s Laboratories Ltd, Fidaxomicin


WO 2016024243, New patent, Dr Reddy’s Laboratories Ltd, Fidaxomicin
WO2016024243, FIDAXOMICIN POLYMORPHS AND PROCESSES FOR THEIR PREPARATION
DR. REDDY’S LABORATORIES LIMITED [IN/IN]; 8-2-337, Road No. 3, Banjara Hills, Telangana State, India Hyderabad 500034 (IN)
CHENNURU, Ramanaiah; (IN).
PEDDY, Vishweshwar; (IN).
RAMAKRISHNAN, Srividya; (IN)
Aspects of the present application relate to crystalline forms of Fidaxomicin IV, V & VI and processes for their preparation. Further aspects relate to pharmaceutical compositions comprising these polymorphic forms of fidaxomicin

Fidaxomicin (also known as OPT-80 and PAR-101 ) is a novel antibiotic agent and the first representative of a new class of antibacterials called macrocycles. Fidaxomicin is a member of the tiacumicin family, which are complexes of 18-membered macrocyclic antibiotics naturally produced by a strain of Dactylosporangium aurantiacum isolated from a soil sample collected in Connecticut, USA. The major component of the tiacumicin complex is tiacumicin B. Optically pure R-tiacumicin B is the most active component of Fidaxomicin. The chiral center at C(19) of tiacumicinB affects biological activity, and R-tiacumicin B has an R-hydroxyl group attached at this position. The isomer displayed significantly higher activity than other tiacumicin B-related compounds and longer post-antibiotic activity.
As per WIPO publication number 2006085838, Fidaxomicin is an isomeric mixture of the configurationally distinct stereoisomers of tiacumicin B, composed of 70 to 100% of R-tiacumicin B and small quantities of related compounds, such as S-tiacumicin B and lipiarmycin A4. Fidaxomicin was produced by fermentation of the D aurantiacum subspecies hamdenensis (strain 718C-41 ). It has a narrow spectrum antibacterial profile mainly directed against Clostridium difficile and exerts a moderate activity against some other gram-positive species. Fidaxomicin is bactericidal and acts via inhibition of RNA synthesis by bacterial RNA polymerase at a distinct site from that of rifamycins. The drug product is poorly absorbed and exerts its activity in the gastrointestinal (Gl) tract, which is an advantage when used in the applied indication, treatment of C. difficile infection (CDI) (also known as C. difficile-associated disease or diarrhoea [CDAD]). Fidaxomicin is available as DIFICID oral tablet in US market. Its CAS chemical name is Oxacyclooctadeca-3,5,9, 13, 15-pentaen-2-one, 3-[[[6-deoxy-4-0-(3,5dichloro-2-ethyl-4,6-dihydroxybenzoyl)-2-0-methyl-P-D-manno pyranosyl]oxy]methyl]-12[[6-deoxy-5-C-methyl-4-0-(2-methyl-1 -oxopropyl)- -D-lyxo-hexo pyranosyl]oxy]-1 1 -ethyl-8-hydroxy-18-[(1 R)-1 -hydroxyethyl] -9,13,15-trimethyl-, (3E.5E, 8S.9E.1 1 S.12R.13E, 15E.18S)-. Structural formula (I) describes the absolute stereochemistry of fidaxomicin as determined by x-ray.

(I)
WIPO publication number 2004014295 discloses a process for preparation of Tiacumicins that comprises fermentation of Dactylosporangium aurantiacum NRRL18085 in suitable culture medium. It also provides process for isolation of tiacumicin from fermentation broth using techniques selected from the group consisting of: sieving and removing undesired material by eluting with at least one solvent or a solvent mixture; extraction with at least one solvent or a solvent mixture; Crystallization; chromatographic separation; High-Performance Liquid Chromatography (HPLC); MPLC; trituration; and extraction with saturated brine with at least one solvent or a solvent mixture. The product was isolated from /so-propyl alcohol (IPA) having a melting point of 166-169 °C.
U.S. Patent No. 7378508 B2 discloses polymorphic forms A and B of fidaxomicin, solid dosage forms of the two forms and composition thereof. As per the ‘508 patent form A is obtained from methanol water mixture and Form B is obtained from ethyl acetate.
J. Antibiotics, vol. 40(5), 575-588 (1987) discloses purification of Tiacumicins using suitable solvents wherein tiacumicin B exhibited a melting point of 143-145 °C.
PCT application WO2013170142A1 describes three crystalline forms of Fidaxomicn namely, Form-Z, Form-Z1 and Form-C. IN2650/CHE/2013 describes 6 crystalline polymorphic forms of Fidaxomicin namely, Forms I, Form la, Form II, Form Ha, Form III and Form Ilia).
The occurrence of different crystal forms, i.e., polymorphism, is a property of some compounds. A single molecule may give rise to a variety of polymorphs having distinct crystal structures and physico-chemical properties.
Polymorphs are different solid materials having the same molecular structure but different molecular arrangement in the crystal lattice, yet having distinct physico-chemical properties when compared to other polymorphs of the same molecular structure. The discovery of new polymorphs and solvates of a pharmaceutical active compound provides an opportunity to improve the performance of a drug product in terms of its bioavailability or release profile in vivo, or it may have improved stability or advantageous handling properties. Polymorphism is an unpredictable property of any given compound. This subject has been reviewed in recent articles, including A. Goho, “Tricky Business,” Science News, August 21 , 2004. In general, one cannot predict whether there will be more than one form for a compound, how many forms will eventually be discovered, or how to prepare any previously unidentified form.
There remains a need for additional polymorphic forms of fidaxomicin and for processes to prepare polymorphic forms in an environmentally-friendly, cost-effective, and industrially applicable manner.

G.V. Prasad, chairman, Dr Reddy’s Laboratories
EXAMPLES
Example 1 : Preparation of fidaxomicin Form IV:
Fidaxomicin (0.5 g) and a mixture of 1 ,4-Dioxane (10 mL), THF (10 ml) and water (20mL) were charged in Easy max reactor (Mettler Toledo). The reactor was set to temperature cycle with following parameters:
Starting temperature: 25 °C;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 25 °C over a period of 2 hours;
Temperature maintained at 25 °C for 6 hours.
After completion of temperature cycling process, the slurry was filtered under suction, followed by drying in air tray dryer (ATD) at 40°C to a constant weight to produce crystalline fidaxomicin form-IV.
Example 2: Preparation of fidaxomicin Form V:
Fidaxomicin (1 g) and a mixture of propylene glycol (10 mL) and water (20mL) were charged in Easy max reactor (Mettler Toledo). The reactor was set to temperature cycle with following parameters:
Starting temperature is 25 °C;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 25 °C over a period of 2 hours;
Temperature maintained at 25 °C for 6 hours.
After completion of temperature cycling process, the slurry was filtered under suction, followed by drying in air tray dryer (ATD) at 40°C to a constant weight to produce crystalline fidaxomicin form-V.
Example 3: Preparation of fidaxomicin Form VI:
Fidaxomicin (0.5 mg) and MIBK (10 mL) were charged in Easy max reactor (Mettler Toledo) and the mixture was heated to 80°C. n-heptane (20 mL) was added to the solution at the same temperature. The mixture was stirred for 1 hour. The reaction mass was then cooled to 25°C. Solid formed was filtered at 25°C and dried at 40°C in air tray dryer (ATD) to a constant weight to produce crystalline fidaxomicin form VI.
Example 4: Preparation of fidaxomicin Form V:
Fidaxomicin (500 mg) and a mixture of R-propylene glycol (5 mL) and water (15 mL) were charged in Easy max reactor (Mettler Toledo). The reactor was set to temperature cycle with following parameters:
Starting temperature is 25 °C;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 25 °C over a period of 2 hours;
Temperature maintained at 25 °C for 2 hours.
After completion of temperature cycling process, the slurry was filtered and dried at 25°C to produce crystalline fidaxomicin form-V.
Example 5: Preparation of fidaxomicin Form V:
Fidaxomicin (1 g) and a mixture of S-propylene glycol (3 ml_) and water (30 mL) were charged in Easy max reactor (Mettler Toledo). The reactor was set to temperature cycle with following parameters:
Starting temperature is 25 °C;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 25 °C over a period of 2 hours;
Temperature maintained at 25 °C for 2 hours.
After completion of temperature cycling process, the slurry was filtered and dried at 25°C to produce crystalline fidaxomicin form-V.
Example 6: Preparation of fidaxomicin Form V:
Fidaxomicin (40 g) and a mixture of propylene glycol (400 mL) and water (1600 mL) were charged in Chem glass reactor. The reactor was set to temperature cycle with following parameters:
Starting temperature is 25 °C;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 25 °C over a period of 2 hours;
Temperature maintained at 25 °C for 6 hours.
After completion of temperature cycling process, the slurry was filtered under suction, followed by drying in air tray dryer (ATD) at 40°C to a constant weight to produce crystalline fidaxomicin form-V.

The 10-member board at pharmaceutical major Dr Reddy’s thrives on diversity. Liberally sprinkled with gray hairs, who are never quite impressed with powerpoint presentations, “they want information to be pre-loaded so that the following discussions (at the board level) are fruitful,” says Satish Reddy, Chairman, Dr Reddy’s. That said, the company has now equipped its board members with a customized application (that runs on their tablets) to manage board agenda and related processes.
see at

Dr. Reddy’s Laboratories Managing Director and Chief Operating Officer Satish Reddy addressing
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
3-(((6-Deoxy-4-O-(3,5-dichloro-2-ethyl-4,6-dihydroxybenzoyl)-2-O-methyl-β-D-mannopyranosyl)oxy)-methyl)-12(R)-[(6-deoxy-5-C-methyl-4-O-(2-methyl-1-oxopropyl)-β-D-lyxo-hexopyranosyl)oxy]-11(S)-ethyl-8(S)-hydroxy-18(S)-(1(R)-hydroxyethyl)-9,13,15-trimethyloxacyclooctadeca-3,5,9,13,15-pentaene-2-one
|
|
| Clinical data | |
| Trade names | Dificid, Dificlir |
| Licence data | US FDA:link |
| Pregnancy category |
|
| Legal status | |
| Routes of administration |
Oral |
| Pharmacokinetic data | |
| Bioavailability | Minimal systemic absorption[1] |
| Biological half-life | 11.7 ± 4.80 hours[1] |
| Excretion | Urine (<1%), faeces (92%)[1] |
| Identifiers | |
| CAS Number | 873857-62-6 |
| ATC code | A07AA12 |
| PubChem | CID 11528171 |
| ChemSpider | 8209640 |
| UNII | Z5N076G8YQ |
| KEGG | D09394 |
| ChEBI | CHEBI:68590 |
| ChEMBL | CHEMBL1255800 |
| Synonyms | Clostomicin B1, lipiarmicin, lipiarmycin, lipiarmycin A3, OPT 80, PAR 01, PAR 101, tiacumicin B |
| Chemical data | |
| Formula | C52H74Cl2O18 |
| Molar mass | 1058.04 g/mol |
///////////WO-2016024243,WO 2016024243, New patent, Dr Reddy’s Laboratories Ltd, Fidaxomicin
CC[C@H]1/C=C(/[C@H](C/C=C/C=C(/C(=O)O[C@@H](C/C=C(/C=C(/[C@@H]1O[C@H]2[C@H]([C@H]([C@@H](C(O2)(C)C)OC(=O)C(C)C)O)O)\C)\C)[C@@H](C)O)\CO[C@H]3[C@H]([C@H]([C@@H]([C@H](O3)C)OC(=O)C4=C(C(=C(C(=C4O)Cl)O)Cl)CC)O)OC)O)\C
WO 2016024284, New Patent, MIRABEGRON, Wanbury Ltd
WO 2016024284, New Patent, MIRABEGRON, Wanbury Ltd

WANBURY LTD. [IN/IN]; BSEL tech park, B wing, 10th floor, sector 30A opp. Vashi Railway Station, Vashi Navi Mumbai 400703 Maharashtra (IN)
DR. NITIN SHARADCHANDRA PRADHAN; (IN).
DR. NILESH SUDHIR PATIL; (IN).
DR. RAJESH RAMCHANDRA WALAVALKAR; (IN).
MR. NILESH SUBHASH KULKARNI; (IN).
MR. SANTOSH NAMDEV RAWOOL; (IN).
MR. PURUSHOTTAM EKANATH AWATE; (IN)

LEFT , DR K CHANDRAN, DIRECTOR WANBURY
MR ASOK SHINKAR
The present invention relates to a novel process for preparation of Mirabegron of Formula (I) using intermediates of Formula (II), (IIIa), (Illb) and (IV).
The present invention relates to a process for preparation of Mirabegron of Formula
(I).

Formula (I)
The present invention further relates to the preparation of Mirabegron of Formula (I) by using compounds of Formula (II), (Ilia), (Illb) and (IV)

Formula (II)

Formula (IlIa) Formula (Illb)

Formula (IV)
Furthermore, the present invention relates to process for preparation of compound of Formula (II), (Ilia), (Illb) and (IV).
Background of the invention:
Mirabegron is chemically known as 2-amino-N-[4-[2-[[(2R)-2-hydroxy-2-phenylethyl]amino]ethyl]phenyl]-4-thiazoleactamide and is marketed under trade name Myrbetiq.
Mirabegron is a drug used for treatment of overactive bladder. It was first disclosed in US 6,346,532, wherein (R)-Styrene oxide is reacted with 4-nitrophenyl ethyl amine hydrochloride to obtain (R)-l- phenyl-2-[[2-(4-nitrophenyl)ethyl]amino]ethanol, the later is then protected with BOC anhydride and subjected to reduction in the presence of Pd/C to yield N-[2-(4-Aminophenyl)ethyl]-N-[(2R)-2-hydroxy-2-phenylethyljcarbamic acid tert-butyl ester. Thus formed compound was then coupled with (2-amino-l,3-thiazol-4yl) acetic acid to obtain BOC protected Mirabegron which is de-protected to give Mirabegron hydrochloride.
The synthetic route proposed in US 6,346,532 is presented in Scheme-I.
Scheme-I

The major draw-backs of the presented synthetic scheme are as follows:
1. Less atomic efficiency
2. Low yield and extensive impurities formations
3. Use of expensive and sensitive protecting agents
4. Column chromatographic techniques for purifications of intermediates.
One more synthetic route for the preparation of Mirabegron have been proposed US 6,346,532, however it is not exemplified.
US 7,342,117 disclose a process for preparation of Mirabegron. The process involves the step of condensation of 4-nitrophenyl ethylamine and (R)- mandelic acid in presence of tri ethylamine, hydroxybentriazole and l-(3-dimethylaminopropyl)-3-ethyl carbodiimide in N,N-dimethylformamide to obtain compound of Formula (A). The second step involves conversion of compound of Formula (A) to compound of Formula (B) in presence of l,3-dimethyl-2-imidazolidone and borontetrahydro fluoride in tetrahydrofuran. In third step, compound of Formula (B) is subjected to reduction using 10% palladium-carbon in methanol to afford (R)-2-[[2′-(4-aminophenyl)-ethyl amino] -1-phenylethanol (Formula IV), which was further condensed with 2-aminothiazol-4-yl acetic acid in presence of l-(3-dimethylaminopropyl)-3 -ethyl carbodiimide and hydrochloric acid in water to obtain Mirabegron of Formula (I). The schematic representation is as Scheme-II

Another patent application CN103193730, discloses a novel process for preparation of Mirabegron wherein the amino group of 2-aminothiazole-5-acetic acid is protected with a protecting group and is condensed with 4-amino phenyl ethanol to obtain an intermediate (A); which on further oxidation yields intermediate (B). The intermediate B is subjected to reductive amination with (R)-2-amino-l -phenyl ethanol and deprotection, simultaneously to yield Mirabegron. The schematic representation is as Scheme-Ill.

Formula (I)
Scheme-Ill
Other references wherein process for preparation of Mirabegron are disclosed CN103387500 and CN103232352.
Most of the prior art reported for preparation of Mirabegron uses expensive and sensitive protecting agents thereby making process less feasible on industrial scale. Furthermore, the yield and purity of Mirabegron obtained by the processes known in art is not satisfactory. It is well known fact that pharmaceutical products like Mirabegron should have high purity due to the therapeutic advantages and also due to the stringent requirements of regulatory agencies. The purity requirements can be fulfilled either by avoiding the formation of by-products during the process or by purifying the end product of the process. The inventors of present invention have skillfully developed the process to provide Mirabegron with unachieved level of purity. Furthermore, the process of present invention is simple, industrially viable, and economic and avoids unfavorable reaction conditions.

According to present invention, the process for preparation of compound of Formula (IV), is depicted in Scheme IV

The present invention further relates to a process for preparation of Mirabegron of Formula (I)
The schematic reaction scheme of Mirabegron according to present invention is depicted in Scheme-V.
 Wherein R is -OH or -CI
Wherein R is -OH or -CI
The detail of the invention provided in the following examples is given by the way of illustration only and should not be construed to limit the scope of the present invention.
EXAMPLES
Example 1: Preparation of [2-(formylamino)-l,3-thiazol-4-yl]acetyl chloride; Formula (V); wherein R is -CI
20g of [2-(formylamino)-l,3-thiazol-4-yl]acetic acid was added to 250 ml of methylene dichloride and the mixture was cooled to -10°C followed by lot wise addition of 25g of phosphorous pentachloride. The mixture stirred while maintaining temperature of -10°C for 2-3 hours. After confirming completion of reaction, the product was filtered out, washed with methylene dichloride and dried to obtain 24g (Yield: 92%) of compound of Formula (V); wherein R is -CI
Example 2: Preparation of 4-nitrophenyl-[2-(formylamino)-l,3-thiazol-4-yl]acetate; Formula (IlIa)
2g of p-nitrophenol was added to 40ml of methylene chloride and 4.963g of potassium carbonate, the mixture was cooled to 10-15°C followed by lot wise addition of 3.95g of compound of Formula (V) of example 1. After confirming completion of reaction, 5.87g (Yield: 99%) of compound of Formula (Ilia) was isolated. The obtained compound has been identified by;
HNMR(D20 Exchange)
8.614 (S,lH),7.359(d,2H),8.119(d,2H),6.561(S,lH),3.765(S,2H).
Example 3: Preparation of (2-amino-l,3-thiazol-4-yl)acetyl chloride; Formula (VI); wherein R is -CI
5g of (2-amino-l,3-thiazol-4-yl)acetic acid was added to 50 ml of methylene dichloride with few drops of dimethylformamide and 6g of oxalyl chloride at temperature ranging from 0-5°C. the mixture was maintained at 0-5°C for 4-5 hours and after completion of reaction, solid mass was filtered out, washed with methylene dichloride and dried to afford 5g (Yield: 89%) of compound of Formula (VI); wherein R is -CI
Example 4: Preparation of 4-nitrophenyl-(2-amino-l,3-thiazol-4-yl)acetate; Formula (Illb)
2g of p-nitrophenol was added to 40ml of methylene chloride and 4.96g of potassium carbonate, and the mixture was cooled to 10-15 °C followed by lot wise addition of 3.95g of compound of Formula (VI) prepared in example 3. After confirming completion of reaction, 6.18g (Yield: 99%) of 4-nitrophenyl-(2-amino-l,3-thiazol-4-yl)acetate of Formula (Illb) was isolated.
The obtained compound has been identified by
HNMR ( D2O Exchange)
7.359(d,2H),8.1 19(d,2H),6.425(S,lH).3.775(S,2H).
Example 5: In-situ preparation of (lR)-2-[[2-(4-aminophenyl)ethyl]amino]-l-phenylethanol or its hydrochloride salt, of Formula (IV)
Step I – Preparation of (2R)-2-hydroxy-N-[2-(4-nitrophenyl)ethyl]-2-phenylethanamide of Formula (IX)
(R)-2-hydroxy-2-phenylacetic acid (75g), triethylamine (50g), hydroxybenzotriazole (HOBt) (33.3g) and l-(3-dimethylaminopropyl)-3-ethyl carbodiimide hydrochloride (EDC.HC1) (50g) were added to a mixture of 2-(4-nitrophenyl)ethylamine hydrochloride (100g) in Ν,Ν-dimethylformamide (375ml) at 25-30°C. The mixture was stirred for 30 minutes followed by addition of another lot of HOBt (33.3g) and EDC.HC1 (50g) in reaction mixture. The reaction mixture was maintained at 25-30°C for 15 hours under stirring. After completion of reaction, water (1850ml) was added to the reaction mixture and stirred. Subsequently, ethyl acetate (1500ml) was added to the reaction mixture at 25-30°C and stirred. The organic phase was separated from aqueous phase, and was washed sequentially with 1M HC1 solution, 20%aqueous potassium carbonate solution and water. The organic solvent was distilled out under reduced pressure to obtain residue comprising of (2R)-2-hydroxy-N-[2-(4-nitrophenyl)ethyl] -2 -phenyl ethanamide of Formula (IX)
Step II – Preparation of (2R)-2-hydroxy-N-[2-(4-aminophenyl)ethyl]-2-phenylethanamide of Formula (X)
The residue from step I, methanol (740ml) and Raney Nickel (14.8g) were charged into an autoclave vessel, 10 kg/cm2 hydrogen gas pressure was applied to the reaction mixture at 25-30°C and the mixture was maintained under stiring 6 hours. Reaction mixture filtered through hyflo bed. Distilled off the solvent completely from the filtrate under reduced pressure to obtain residue comprising (2R)-2-hydroxy-N-[2-(4-aminophenyl)ethyl]-2-phenylethanamide of Formula (X)
Step III – Preparation of (lR)-2-[[2-(4-aminophenyl)ethyl]amino]-l-phenylethanol dihydrochloride salt, of Formula (IV)
The residue of step II was added in tetrahydrofuran (665ml) and the mixture was cooled to -5 to 0°C. To this cooled mixture was then successively added sodium borohydride (56.26g) and BF3-diethyl ether (466g), and the mixture was stirred for 15 minutes. The temperature of reaction mixture was gradually increased to 50-55°C and was maintained under stirring for 5 hours. After completion of reaction, the reaction mixture was cooled to 0-5°C and 50% sodium hydroxide solution was added till pH is basic. The temperature of reaction mixture is then raised to 25-30°C followed by addition of ethyl acetate (500ml). The organic layer was separated and subjected to distillation to afford a residue. To the residue was added isopropyl alcohol (665ml) and mixture was refluxed for 30 minutes. The mixture was then allowed to cool to 40-45°C, isopropyl alcohol hydrochloride (200ml) was added till pH acidic and mixture was stirred for 2 hours to afford precipitate. The precipitate was filtered out and washed with isopropyl alcohol. The wet cake thus obtained was added to 20% aqueous sodium hydroxide solution (till pH basic) followed by addition of dichloromethane (500ml). The organic layer was separated from aqueous layer and was subjected to distillation under reduced pressure to obtain residue. The residue was taken in toluene (500ml), heated to 55-60°C for 30 minutes and cooled to 10-15°C. The precipitate obtained was filtered, washed with toluene and to the wet cake afforded was added isopropyl alcohol (665ml). The mixture was refluxed for 30 minutes and then cooled to 50-55°C. At 50-55°C slowly isopropyl alcohol hydrochloride (200ml) till pH acidic was added and mixture was stirred for 2 hours to obtain precipitate. The precipitate was filtered out, washed with isopropyl alcohol and dried to get (lR)-2-[[2-(4-aminophenyl)ethyl]amino]-l-phenylethanol dihydrochloride salt, of Formula (IV)
Yield-70%
HPLC Purity: 98%
Example 6: Alternate method for preparation of (2R)-2-hydroxy-N-[2-(4-nitrophenyl)ethyl]-2-phenylethanamide of Formula (IX)
Step I – A mixture of (R)-2-hydroxy-2-phenylacetic acid (lOg), dichloromethane (50ml) and triethylamine (24ml) was cooled to 0-5°C and slowly para-toluene sulfonyl chloride (12.53g) was added to it. The temperature of reaction mixture was raised to 25-30°C and maintained for 12 hours. After completion of reaction, water (100ml) was added to the reaction mixture and the mixture was stirred for 15 minutes. The organic phase was separated and distills out completely under reduced pressure to obtain [(R)-2-hydroxy -2-phenyl acetic tosyl ester].
Yield-56%
Step II – 2-(4-nitrophenyl)ethylamine hydrochloride (6g) was added to dichloromethane (50ml) and stirred for 30 minutes at 25-30°C. The mixture was
then cooled to 0-5 °C and triethylamine (13ml) was added. To say cooled mixture was then slowly added a mixture of (R)-2-hydroxy -2-phenyl acetic tosyl ester (lOg) and dichloromethane (50ml). The temperature of reaction mixture was then raised to reflux temperature and maintained for 5 hours. After completion of reaction, water (50ml) was added to the reaction mixture and the mixture was stirred for 15 minutes. The organic phase was separated and distill out completely under reduced pressure to obtain (R)-2-hydroxy-N-[2-(4-nitrophenyl) ethyl]-2-phenylacetamide
Yield-70%, Purity-96%
Example 7: Preparation of compound of Formula (II) from compound of Formula (V); wherein R is -OH
1.58g of [2-(formylamino)-l,3-thiazol-4-yl]acetic acid of Formula (V) was added solution of (1R )-2-{[2-(4-aminophenyl)ethyl]amino}-l-phenylethanol of Formula (IV) in water (2g of Formula (IV) in 50ml water) followed by addition of 0.66g concentrated hydrochloric acid and 3.27g of l-ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride. The mixture was stirred at 25-30°C for 0.5 hours. After completion of reaction, pH was adjusted to 8-9 using aqueous saturated solution of sodium carbonate. The solid precipitated out was filtered, washed with water and dried to obtain 2.1g of compound of Formula (II). (Yield: 72%) The obtained compound has been identified by HNMR
2.502(m,4H),2.599(m,2H),3.685(S,2H),4.9(S, NH protons),7.01(m, 10H, aromatic), 8.54(S,1H), 10.0(S, -OH proton),
HNMR(D20 Exchange) 2.502(m,4H),2.60(m,2H),4.57(m,lH),7.0(m, 10H, aromatic), 8.43(S,1H)
Example 8: Preparation of compound of Formula (II) from compound of Formula (V); wherein R is -CI
lOg of ( 1R)-2-{[2-(4-aminophenyl)ethyl]amino}-l-phenylethanol of Formula (IV) (prepared by methods known in prior art/ as given in example 5), was added to 150ml of acetonitrile with 16.17g of potassium carbonate and the mixture was cooled to 10-15°C. 18.8g of Formula (V) of example 1 was added to above mixture at 10-15°C in lot wise. After completion of reaction, the reaction mixture was concentrated under vacuum and 90ml of water was added for isolation. The product was then filtered out, washed with water and dried to obtain 72g (Yield: 70%) of compound of Formula (II).
Example 9: Preparation of compound of Formula (II) from compound of Formula (IlIa)
5.87g of compound of Formula (IlIa) was added to 40 ml of methylene dichloride with 2.36 g of potassium carbonate and 3.67g of ( 1))-2-{[2-(4-aminophenyl)ethyl]amino}-l-phenylethanol (Formula-IV ; prepared by methods known in prior art/ as given in example 5) . The mixture was stirred at 25-30°C for 1 hour. After completion of reaction, the reaction mixture was concentrated followed by addition of 60 ml of water to isolate lg of compound of Formula (II).
Example 10: Insitu preparation of compound of Formula (II) without isolation of compound of Formula (IlIa)
2g of p-nitrophenol was added to 40 ml of methylene chloride with 4.963g of potassium carbonate, and the mixture was cooled to 10-15°C followed by lot wise addition of 3.95g of [2-(formylamino)-l,3-thiazol-4-yl]acetyl chloride of Formula (V) of example 1. After confirming complete formation of compound of Formula (Ilia), 2.36g of potassium carbonate and 3.67g of (1R)-2-{[2-(4-aminophenyl)ethyl]amino}-1 -phenyl ethanol of Formula (IV) (prepared by methods known in prior art/ as given in example 5) was added insitu, and the mixture was stirred at 25-30°C for 1 hour. After completion of reaction, the reaction mixture was concentrated followed by addition of 60 ml of water to isolate lg of compound of Formula (II).
Example 11: Preparation of Mirabegron from compound of Formula (II)
To 2g of compound of Formula (II) was added 30ml of 10% sodium hydroxide and the mixture was stirred at 55-60°C for 3 hours. After completion of reaction, the mixture was cooled to 25-30°C and the solid obtained was filtered, washed with water and dried to yield 1.3g of Mirabegron. (Yield: 70%)
Example 12: Preparation of Mirabegron from compound of Formula (Illb)
6.18g of 4-nitrophenyl-(2-amino-l,3-thiazol-4-yl)acetate was added to 40ml of methylene dichloride with 2.36g of potassium carbonate and 3.65g of (1R)-2-{ [2-(4-aminophenyl)ethyl]amino}-l-phenylethanol of Formula (IV) (prepared by methods known in prior art/ as given in example 5), and the mixture was stirred at 25-30°C for 1 hour. After completion of reaction, solid was filtered out, washed with methylene dichlrode and dried to yield lg of Mirabegron of Formula (I).
Example 13: Insitu preparation of Mirabegron without isolation of compound of Formula (Illb)
To 40ml of methylene chloride was added 2g of p-nitrophenol and 4.96g of potassium carbonate, and the mixture was cooled to 10-15°C followed by lot wise addition of 3.95g of compound of Formula (VI) prepared in example 3. After confirming complete formation of compound of Formula (Illb), 2.36g of potassium carbonate and 3.65g of (1R)-2-{[2-(4-aminophenyl)ethyl]amino}-l-phenylethanol of Formula (IV) (prepared by methods known in prior art/ as given in example 5) was added insitu, and the mixture was stirred at 25-30°C for 1 hour. After completion of reaction, After completion of reaction, solid was filtered out, washed with methylene dichlrode and dried to yield lg of Mirabegron of Formula (I).
Example 14: Preparation of Mirabegron from compound of Formula (VI); wherein R is -CI
To 20ml of acetone was added 2g of (l/?)-2-{[2-(4-aminophenyl)ethyl]amino}-l-phenylethanol of Formula (IV) and 2.15g of potassium carbonate, and the mixture was cooled to 10-15°C followed by addition of (2-amino-l,3-thiazol-4-yl)acetyl chloride of Formula (VI). After completion of reaction, acetone was concentrated under vacuum and 90ml of water was added for for isolation. The product was then filtered out, washed with water and dried to obtain 2g (Yield: 70%) of Mirabegron.



/////WO-2016024284, WO 2016024284, New Patent, MIRABEGRON, Wanbury Ltd
WO 2016024289, NILOTINIB, New Patent by SUN PHARMA
NILOTINIB

WO 2016024289, NILOTINIB, New Patent by SUN
SUN PHARMACEUTICAL INDUSTRIES LTD [IN/IN]; 17/B, Mahal Industrial Estate, Off Mahakali Caves Road, Andheri (east), Mumbai 400093 (IN)
THENNATI, Rajamannar; (IN).
KILARU, Srinivasu; (IN).
VALANCE SURENDRAKUMAR, Macwan; (IN).
SHRIPRAKASH DHAR, Dwivedi; (IN)
The present invention provides novel salts of nilotinib and polymorphs thereof. The acid addition salts of nilotinib with benzenesulfonic acid, butanedisulfonic acid, 1-5- naphthalenedisulfonic acid, naphthalene-1-sulfonic acid and 1-hydroxynaphthoic acid; hydrates and anhydrates thereof.
Nilotinib, 4-methyl-N-[3-(4-methyl-lH-imidazol-l-yl)-5-(trifluoromethyl)phenyl]-3-[[4-(3-pyridinyl)-2-pyrimidinyl] amino] -benzamide, having the following formula

is marketed under the name Tasigna® in US and Europe. Tasigna contains nilotinib monohydrate monohydrochloride salt and is available as capsules for the treatment of adult patients with newly diagnosed Philadelphia chromosome positive chronic myeloid leukemia (Ph+ CML) in chronic phase. Tasigna is also indicated for the treatment of chronic phase and accelerated phase Philadelphia chromosome positive chronic myelogenous leukemia (Ph+ CML) in adult patients resistant or intolerant to prior therapy that included imatinib.
Nilotinib is considered a low solubility/low permeability (class IV) compound in the Biopharmaceutics Classification System (BCS). Therefore, dissolution of nilotinib can potentially be rate limiting step for in-vivo absorption. It is soluble in acidic media; being practically insoluble in buffer solutions of pH 4.5 and higher.
WIPO publication 2014059518A1 discloses crystalline forms of nilotinib hydrochloride and methods of the preparation of various crystalline solvates of nilotinib hydrochloride including benzyl alcohol, acetic acid and propylene glycol.
WIPO publication 2011033307A1 discloses nilotinib dihydrochloride and its hydrates and method for their preparation.
WIPO publication 2011163222A1 discloses the preparation of nilotinib salts and crystalline forms thereof. The salts of nilotinib disclosed are hydrochloride, fumarate, 2-chloromandelate, succinate, adipate, L-tartrate, glutarate, p-toluenesulfonate, camphorsulfonate, glutamate, palmitate, quinate, citrate, maleate, acetate, L-malate, L-aspartate, formate, hydrobromide, oxalate and malonate.
WIPO publication number 2011086541A1 discloses a nilotinib monohydrochloride monohydrate salt and methods for preparing.
WIPO publication number 2010054056A2 describes several crystalline forms of nilotinib hydrochloride.
WIPO publication number 2007/015871A1 discloses the preparation of nilotinib salts and crystalline forms thereof. The salts are mixtures of nilotinib and one acid wherein the acids are selected from the group consisting of hydrochloric acid, phosphoric acid, sulfuric acid, sulfonic acid, methane sulfonic acid, ethane sulfonic acid, benzene sulfonic acid, p-toluene sul- fonic acid, citric acid, fumaric acid, gentisic acid, malonic acid, maleic acid, and tartaric acid.
WIPO publication number 2007015870A2 discloses several nilotinib salts including amorphous and crystalline forms of nilotinib free base, nilotinib HC1 and nilotinib sulfate along with their hydrate and solvates.
EXAMPLES:
Example 1: Preparation of nilotinib benzenesulfonate crystalline Form I
Nilotinib base (1 g) was suspended in water (20 ml). A solution of benzenesulfonic acid (0.4 g) in water (3ml) was added and the content was heated at 60 °C for 2-3 h. The mixture was cooled to 25-30 °C, filtered, washed with water (3 x 5 ml) and dried under vacuum for 2 h at 50-55 °C.
1H NMR (500 MHz, DMSO-d6) δ 2.40 (s,3H), 2.42 (s,3H), 7.35-7.37 (m,3H), 7.51-7.66 (m,5H),7.83 (d,lH), 7.96 (s,lH),8.08 (s,lH),8.30 (s,lH) 8.39 (s,lH),8.54 (d,lH), 8.61 (d,lH), 8.64 (s,lH), 8.75 (d,lH), 9.25 (s,lH), 9.34 (d,lH), 9.61 (s,lH), 10.84 (s,lH).
The salt provides an XRPD pattern substantially same as set forth in FIG. 1.
Example 2: Preparation of nilotinib butanedisulfonate (2: 1) crystalline Form II
Nilotinib base (100 g) was dissolved in 20 % water in THF solution (2000 ml) at 60-65 °C and insoluble matter was filtered. The filtrate was concentrated under vacuum below 60 °C. Filtered water (1000 ml) was added to the reaction mixture and it was heated at 50-55 °C, followed by addition of 1,4-butanedisulfonic acid -60% aqueous solution (28.6 ml) at same temperature. The content was stirred at 50-55 °C for 2-3h. Reaction mixture as cooled to 25-30 °C and product was filtered, washed with water (200 ml x 2) and dried in air oven at 50-55 °C (yield: 115 g).

Sun Pharma managing director Dilip Shanghvi.
Purity (by HPLC):99.76%
1H NMR (400 MHz,DMSO-d6) δ 1.63-1.66(m,2H), 2.40(d,3H),2.42(s,3H),2.43-2.47(m,2H), 7.51-7.62(m,3H),7.85(dd,lH),7.96(s,lH),8.08(s,lH),8.34(s,lH),8.38(d,lH),8.52-8.55(m,lH), 8.60-8.62 (m,2H), 8.75(d,lH), 9.25(S,1H),9.34(S,1H),9.59(S,1H),10.86(S,1H)
Water content: 7.95 %.
The salt has a XRPD pattern substantially same as set forth in FIG. 2.
Example 3: Preparation of nilotinib butanedisulfonate (2: 1) crystalline Form II
Nilotinib base (300 g) was suspended in methanol (3000 ml) and aqueous hydrochloric acid was added to get pH less than 2. Reaction contents were heated at reflux and was filtered and washed with methanol (100 ml). 5% (w/w) NaOH (1200 ml) solution was added at 40-45 °C within 15 min, reaction mixture was stirred for 2h. Product was filtered, washed with water
(300 ml x 3) and dried for lh. Wet material was suspended in water (3000 ml), heated at 50- 55 °C followed by addition of 1,4-butanedisulfonic acid -60% aqueous solution. The reaction mixture was stirred at 50-55°C for 2hrs. Product was filtered at room temperature, washed with water (500 ml x 2) and dried in air oven at 50-55 °C (yield: 293 g).
Purity (by HPLC): 99.88 %
1H NMR (400 MHz,DMSO-d6+TFA-dl) δ 1.75-1.78(m,2H), 2.36(d,3H),2.38(s,3H),2.69- 2.72(m,2H),7.45(d,lH),7.68(d,lH),7.83(s,lH),7.88(dd,lH),7.97(s,lH),8.16-8.19(m,lH), 8.35
(s,2H), 8.63(d,lH),8.68(d,lH),9.04(d,lH),9.21(d,lH),9.53(br s,lH),9.69(d,lH)10.80 (s,lH)
Water content: 6.44 %
Example 4: Preparation of nilotinib butanedisulfonate (2: 1) crystalline Form III
Nilotinib butanedisulfonate (210g) was dissolved in acetic acid water mixture (50:50) (2520 ml) at 75-80 °C and was filtered to remove insoluble matter and washed with acetic acid water mixture (50:50) (210 ml). Water (3150ml) was added to the filtrate and stirred first at room temperature and then at 0-5 °C. Product was filtered and washed with water. Material was dried in air oven at 70-75 °C. Dried material was leached with methanol (3438 ml) at reflux temperature, filtered and dried in air oven 70-75°C (yield: 152.6 g)
Purity (by HPLC): 99.89 %
1H NMR (400 MHz,DMSO-d6+TFA-dl) δ 1.73-1.77(m,2H), 2.40(s,6H),2.67-2.70(m,2H), 7.50 (d,lH), 7.70(d,lH), 7.88-7.92(m,2H), 8.07(s,lH),8.23 (dd,lH), 8.34(s,2H), 8.67 (d,lH), 8.72 (d,lH), 9.09(d,lH), 9.23 (s,lH), 9.54(d,lH), 9.74(d,lH), 10.86(s,lH).
Water content: 0.61 %
The salt provides an XRPD pattern substantially same as set forth in FIG. 3.
Example 5: Preparation of crystalline form of nilotinib butanedisulfonate (2: 1)
Crystalline Nilotinib butanedisulfonate (1 g) of Example 2 was suspended in methanol (20 ml) and was stirred at reflux for 60 min. The mixture was cooled to room temperature. Solid was filtered, washed with methanol (2 ml x 3) and dried in air oven at 70-75°C (yield: 0.8 g)
Example 6: Preparation of nilotinib butanedisulfonate (1: 1) crystalline Form IV
Nilotinib base (20 g) was suspended in methanol (800 ml) and 1,4-butanedisulfonic acid -60
% aqueous solution (6 ml) was added at 50-55 °C, and was filtered to remove insoluble matter. Filtrate was stirred at room temperature for 2-3 h. Product formed was filtered, washed with methanol (20 ml x 2) and dried the product in air oven at 70-75 °C (yield: 18.4 g).
Purity (by HPLC):99.86 %
1H NMR (400 MHz,DMSO-d6) δ 1.64-1.68(m,4H), 2.47-2.5 l(m,4H), 2.41(s,3H), 2.42(d,3H), 7.52(d,lH), 7.83-7.89(m,2H), 7.99(s,lH), 8.15(s,lH), 8.36 (d,lH), 8.39(s,lH), 8.65-8.66(m,2H), 8.79(d,lH), 8.89(br s,lH), 9.36(s,lH), 9.41(br s,lH), 9.74(d,lH), 10.91(s,lH).
The salt has XRPD pattern substantially same as set forth in FIG. 4.
Example 7: Preparation of nilotinib 1,5-napthalenedisulfonic acid salt (2: 1) crystalline Form V
Nilotinib base (1 g) was suspended in water (20 ml). A solution of 1,5-napthalenedisulfonic acid (0.4 g; 0.6 eq.) in water (5ml) was added and the content was heated at 50-55 °C for lh. The mixture was cooled to 25-30 °C, filtered and washed with water (10 ml). The product was dried in air oven at 50-55°C (yield: 1.2 g).
1H NMR (400 MHz,DMSO-d6) δ 2.39 (s,3H), 2.42 (s,3H), 7.45-7.61 (m,4H),7.84 (d,lH), 7.97(s,2H),8.08 (m,lH),8.31 (s,lH) 8.38 (s,lH),8.55 (d,lH), 8.63 (s,2H), 8.75 (s,lH), 8.92 (d,lH), 9.26 (s, 1H), 9.34 (s,lH),9.62 (s,lH), 10.85 (s,lH).
The salt has a XRPD pattern substantially same as set forth in FIG. 5.
Example 8: Preparation of nilotinib 1,5-napthalenedisulfonic acid salt (1: 1) crystalline Form VI
Nilotinib base (1 g) was suspended in water (20 ml). A solution of 1,5-napthalenedisulfonic acid (0.8 g; 1.2eq) in water (5 ml) was added and the content was heated at 50-55 °C for 1 h. The mixture was cooled to 25-30 °C, filtered, washed with water (10 ml) and dried in air oven at 50-55 °C (yield: 1.4g).
1H NMR(400 MHz,DMSO-d6) δ 2.40 (s,3H),2.41 (s,3H), 7.43-7.52 (m,3H),7.61 (d,lH), 7.85-7.99(m,5H),8.11 (s,lH),8.34 (s,2H), 8.64-8.67 (m,2H), 8.89-8.92 (m,4H),9.40(d,2H), 9.72 (s,lH), 10.87 (s,lH).
The salt has a XRPD pattern substantially same as set forth in FIG. 6.
Example 9: Preparation of nilotinib napthalene-1- sulfonic acid salt crystalline Form VII Nilotinib base (1 g) was suspended in water (10 ml) and heated to 50-55 °C. A solution of napthelene-1 -sulfonic acid and methanol (10 ml) was added to it and heated at 70-75 °C for 30 min. The mixture was cooled to 25-30 °C and stirred for 10 min. The product was filtered, washed with water (2 x 2 ml) and dried under vacuum for 1-2 h at 50-55 °C.
1H NMR (400 MHz,DMSO-d6) δ 2.41 (s,3H),2.42 (s,3H), 7.46-7.58 (m,5H), 7.70-8.00 (m,7H)8.11(s,lH)8.31(s,lH),8.37(s,lH),8.63-8.66 (m,3H), 8.81-8.89 (m,2H), 9.31 (s,lH), 9.37 (d,lH), 9.71 (d,lH), 10.86 (s,lH)
The salt has a XRPD pattern substantially same as set forth in FIG. 7.
Example 10: Preparation of nilotinib l-hydroxy-2-napthoic acid salt crystalline Form VIII Nilotinib base (1 g) was suspended in water (20 ml) and heated to 50-55 °C. l-Hydroxy-2-napthoic acid was added to it and the content was heated at 50-55 °C for 1 h. Methanol (5 ml) was added to the mixture and stirred for 30 min. The content was filtered, washed with water (2 x 2 ml) and dried under vacuum for 1 h at 50-55 °C.
1H NMR (400 MHz, DMSO-d6) δ 2.25 (s,3H), 2.41 (s,3H), 7.40-7.92 (m,l lH), 8.23-8.73 (m,8H), 9.24 (s,lH), 9.34(s,lH), 10.70 (s,lH).
The salt has a XRPD pattern substantially same as set forth in FIG. 8.
 |
|
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)- 5-(trifluoromethyl)phenyl]-3- [(4-pyridin-3-ylpyrimidin-2-yl) amino]benzamide
|
|
| Clinical data | |
| Trade names | Tasigna |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a608002 |
| Licence data | EMA:Link, US FDA:link |
| Pregnancy category |
|
| Legal status | |
| Routes of administration |
Oral |
| Pharmacokinetic data | |
| Bioavailability | 30%[1] |
| Protein binding | 98%[1] |
| Metabolism | Hepatic (mostly CYP3A4-mediated)[1] |
| Biological half-life | 15-17 hours[1] |
| Excretion | Faeces (93%)[1] |
| Identifiers | |
| CAS Number | 641571-10-0(base) |
| ATC code | L01XE08 |
| PubChem | CID 644241 |
| IUPHAR/BPS | 5697 |
| DrugBank | DB04868 |
| ChemSpider | 559260 |
| UNII | F41401512X |
| KEGG | D08953 |
| ChEBI | CHEBI:52172 |
| ChEMBL | CHEMBL255863 |
| PDB ligand ID | NIL (PDBe, RCSB PDB) |
| Chemical data | |
| Formula | C28H22F3N7O |
| Molar mass | 529.5245 g/mol |
//////////////WO 2016024289, WO-2016024289, NILOTINIB, New Patent, SUN
Cc1ccc(cc1Nc2nccc(n2)c3cccnc3)C(=O)Nc4cc(cc(c4)n5cc(nc5)C)C(F)(F)F
Fosfluconazole
Fosfluconazole
Fosfluconazole; 194798-83-9; UNII-3JIJ299EWH; 3JIJ299EWH; NCGC00182029-01;
2-(2,4-difluorophenyl)-1,3-di(1h-1,2,4-triazol-1-yl)propan-2-yl dihydrogen phosphate;
2,4-difluoro-α,α-bis(1H-1,2,4-triazol-1-ylmethyl) benzyl alcohol, dihydrogen phosphate
| Molecular Formula: | C13H13F2N6O4P |
|---|---|
| Molecular Weight: | 386.250688 g/mol |
Research Code:UK-292663, UK 292663, F-FLCZ, F FLCZ
Trade Name:Prodif® PFIZER
MOA:Azole antifungal
Indication:Cryptococcus neoformans; Candidiasis
Status:Approved, Japan PMDA OCT 16 2003
Company:Pfizer (Originator)
Candidiasis,Cryptococcus neoformans, Injection, Solution, Eq. 100 mg/200 mg/400 mg fluconazole per vial
Fosfluconazole (INN) is a water-soluble phosphate prodrug of fluconazole – a triazole antifungal drug used in the treatment and prevention of superficial and systemic fungal infections. The phosphate ester bond is hydrolysed by the action of a phosphatase – an enzyme that removes a phosphate group from its substrate by hydrolysing phosphoric acid monoesters into a phosphate ion and a molecule with a free hydroxyl group (see dephosphorylation).
Fosfluconazole was approved by Pharmaceuticals and Medicals Devices Agency of Japan (PMDA) on Oct 16, 2003. It was developed and marketed as Prodif® by Pfizer in Japan.
Fosfluconazole is a water-soluble phosphate prodrug of fluconazole – a triazole antifungal drug. It is indicated for the treatment of candida and cryptococcus infections.
Prodif® is available as solution for intravenous use, containing 100, 200 or 400 mg of free Fosfluconazole per vial. The recommended dose is 50 to 100 mg administered intravenously once daily for candidiasis. Another dose is 50 to 200 mg fluconazole once daily for cryptococcosis.


Reference:1. WO9728169A1 / US6977302B2.
2. Org. Process Res. Dev.2002, 6, 109-112.
http://pubs.acs.org/doi/pdf/10.1021/op010064%2B
2-(2,4-Difluorophenyl)-1,3-bis(1H-1,2,4-triazole-1-yl)- 2-propyl dihydrogen phosphate (2). A slurry of dibenzyl 2-(2,4-difluorophenyl)-1,3-bis(1H-1,2,4-triazole-1-yl)-2-propyl phosphate (30.1 kg, 53.13 mol), 5% palladium-on-carbon catalyst (50% wet, type 5R39, 1.5 kg), and sodium hydroxide (4.36 kg, 108.9 mol) in low-endotoxin water (75.7 L) was hydrogenated at ambient temperature and 414 kPa (60 psi) for 12 h. The slurry was filtered, and the catalyst was washed with low-endotoxin water (9.8 L). After separating the toluene by-product, the aqueous phase was slurried with carbon (3.1 kg) for 30 min. After the carbon was removed by filtration, the aqueous phase was acidified to pH 1.45 by that addition of sulfuric acid (6.69 kg) in low-endotoxin water (25 L) over 2 h. The resulting slurry was granulated at ambient temperature for 1 h and then filtered. The product was sequentially washed with filtered low-endotoxin water (103 L) and filtered acetone (103 L). The product was dried under vacuum at 50 °C for 12 h to give the title compound (18.1 kg, 88%) a white powder: mp 223-224 °C.
1H NMR (DMSO) δ 5.07 (2H, d), 5.24 (2H, d), 6.77-6.83 (1H, m), 7.00-7.18 (2H, m), 7.75 (2H, s), 8.53 (2H, s).
Found: C, 40.28; H, 3.39; N, 21.63;
[MH]+ 387.0786. C13H13F2N6O4P requires: C, 40.43; H, 3.39; N, 21.78; [MH]+ 387.0782.
US6977302
https://www.google.com/patents/US6977302
EXAMPLE 1 1-(2,4-Difluorophenyl)-1,3-bis(1H-1,2,4-triazol-1-yl)-2-propyl dihydrogen phosphate
(a) Dibenzyl 2-(2,4-difluorophenyl)-1,3-bis(1H-1,2,4-triazol-1-yl)-2-propyl phosphate
Method A
A solution of 2-(2,4-difluorophenyl)-1,3-bis(1H-1,2,4-triazol-1-yl)propan-2-ol (also known as fluconazole, 10.0 g, 32.6 mmol), 1H-tetrazole (6.85 g, 97.8 mmol), dibenzyl diisopropyl phosphoramidite (22.55 g, 65.2 mmol) in methylene chloride (100 ml) was stirred at room temperature under a nitrogen atmosphere for 2 hours. The mixture was then cooled to 0° C., and a solution of 3-chloroperoxybenzoic acid (13.5 g, 50-55% w/w, 39.1 mmol) in methylene chloride (50 ml) was added maintaining the temperature at 0° C. The resulting mixture was allowed to warm to room temperature for 1 hour before washing with aqueous sodium metabisulphite and sodium bicarbonate. After drying (MgSO4) the solvent was removed and replaced with methyl isobutyl ketone (37 ml) and tert-butyl methyl ether (74 ml). After granulating at −10° C. for 1 hour the product was filtered and washed with ice cold methyl isobutyl ketone and tert-butyl methyl ether (1:3, 15 ml) and dried at 50° C. under vacuum for 18 hours to give the subtitle compound (16.05 g, 87%), m.p. 93° C.
Found: C, 57.12; H, 4.46; N, 14.85. C27H25F2N6O4P requires C, 57.24; H, 4.46; N, 14.84%. m/z 567 (MH+) 1H NMR (300 MHz, CDCl3) δ=4.90 (d, 2H), 4.95 (d, 2H), 5.05 (d, 2H), 5.19 (d, 2H), 6.58-6.73 (m, 2H), 6.88-6.95 (m, 1H), 7.20-7.30 (m, 4H) 7.32-7.38 (m; 6H), 7.80 (s, 2H), 8.36 (s, 2H).
Method B
To stirred ethyl acetate (1530 ml) was added 2-(2,4-difluorophenyl)-1,3-bis(1H-1,2,4-triazol-1-yl)propan-2-ol (also known as fluconazole, 306 g, 1.00 mol) and pyridine (237.3 g, 3.00 mol) before cooling to 0° C. Phosphorus trichloride (137.4 g, 1.00 mol) was added dropwise to the reaction mixture maintaining the temperature between 0-5° C. before allowing the reaction mixture to warm to 15° C. over 30 minutes. Benzyl alcohol (216 g, 2.00 mol) was then added over 30 minutes at 15-20° C. After a further 30 minutes hydrogen peroxide (27.5% w/w in water, 373 g) was added maintaining the temperature at 15-20° C. After 30 minutes the aqueous phase was removed and the organic phase washed with aqueous sodium metabisulphite, dilute hydrochloric acid and water. The solvent was removed at reduced pressure and replaced with methyl isobutyl ketone (850 ml) and tert-butyl methyl ether (1132 ml). After granulating at 20° C. for 1 hour and at 0° C. for 1 hour, the product was filtered and washed with ice cold tert-butyl methyl ether (2×220 ml) and dried at 50° C. under vacuum for 18 hours to give the subtitle compound (358 g, 63%). The melting point and spectroscopic data was identical to that stated in method A.
(b) 2-(2,4-Difluorophenyl)-1,3-bis(1H-1,2,4-triazol-1-yl)-2-propyl dihydrogen phosphate
A slurry of the compound of step (a) (9.80 g, 17.3 mmol), 5% palladium on carbon catalyst (50% wet, 1.0 g) and sodium hydroxide (1.38 g, 34.6 mmol) in water (26 ml) was hydrogenated at room temperature and 414 kPa (60 p.s.i.) for 20 hours. The solution was filtered through a pad of celite (trade mark) and washed with water (5 ml). The toluene was separated and the aqueous phase cooled to 0° C. whereupon sulphuric acid (1.70 g, 17.3 mmol) was added. The resulting slurry was granulated at 0° C. for 1 hour and then filtered, washed with water (2×5 ml) and dried under vacuum at 50° C. to give the title compound (5.80 g, 87%). m.p. 223-224° C.
Found: C, 40.28; H, 3.39; N, 21.63. C13H13F2N6O4P requires C, 40.43; H, 3.39; N, 21.76%. 1H NMR (300 MHz, DMSO) δ=5.07 (d, 2H) 5.24 (d, 2H), 6.77-6.83 (m, 1H), 7.00-7.18 (m, 2H), 7.75 (s, 2H), 8.53 (s, 2H).
EXAMPLE 2 2-(2,4-Difluorophenyl)-1,3-bis(1H-1,2,4-triazol-1-yl)-2-propyl disodium phosphate
A solution of the compound of Example 1(a) (10.0 g, 17.7 mmol) and sodium acetate (2.90 g, 35.3 mmol) in ethanol (160 ml) and water (20 ml) was hydrogenated over Pearlman’s catalyst (1.00 g) at room temperature and at 345 kPa (50 p.s.i.) for 16 hours. The solution was filtered through a pad of celite (trade mark) and the solvents removed at reduced pressure to leave a thick syrup. This was dissolved in ethanol (100 ml) with the aid of sonication and warmed to reflux. The resulting solution was allowed to cool slowly and granulate for 1 hour at room temperature. The product was filtered, washed with ethanol (10 ml) and dried under vacuum at 50° C. to give the title compound (4.48 g, 59%). m.p. 160-162° C.
1H NMR (300 MHz, D2O) δ=5.01 (d, 2H), 5.40 (d, 2H), 6.60 (m, 1H), 6.79 (m, 1H), 7.11 (m, 1H), 7.63 (s, 2H), 8.68 (s, 2H).
Route 2

Reference:1. CN103864844A.
http://www.google.com/patents/CN103864844A?cl=en
TRANSLATED BY MACHINE…….TEXT MAY VARY
forskolin fluconazole (fosf Iuconazole, Formula I) is fluconazole (Formula IV) of monophosphate prodrugs, fluconazole in the tertiary alcohol into a phosphate ester, not only did not introduce a chiral center, also increased water solubility, because a long time to overcome the low water solubility of fluconazole resulting larger infusion volume defects. After intravenous administration in the role of phosphatases in vivo hydrolysis into fluconazole, pharmacological effect. Blessing from the Central Institute of the United States Secretary of fluconazole Fai end developed, launched in Japan in 2004 I May 15, for the treatment of candidiasis and cryptococcal infections caused deep as true bacteremia, respiratory fungal disease, fungal peritoneum
Inflammation, gastrointestinal fungal disease, fungal urinary tract infections, fungal meningitis.
Synthesis gas itraconazole on forskolin in W09728169, Organic Process Research & Development (200 2), 6 (2), 109-112, CN1789270, Art of Drug Synthesis (2007), 71-82, etc. have been reported in the literature . Which Organic Process Research & Development (2002) described in detail in the first blessing Secretary fluconazole and improved synthetic route for the route problems to adapt to industrial mass production of synthetic routes.
Document Organic Process Research & Development (2002), 6,109-112 discloses the following two synthetic routes.
Route One:
Route two:
The final step is a route to the removal of benzyl group in a methanol solvent by palladium on carbon catalyzed hydrogenation reaction yield was 65%. Since forskolin fluconazole final product insoluble in methanol, and therefore there is a route following shortcomings: a catalyst poisoning, the final product is easy to form methanol solvate, removing the catalyst in the loss of product, the final product are difficult to separate, low yield not suitable for industrial production.
Two routes still using palladium on carbon hydrogenation debenzylation, except that the solvent was changed to sodium hydroxide solution, the product of soluble and stable in aqueous sodium hydroxide solution, after filtering off the catalyst, forskolin fluoro itraconazole by acidification of sodium sulfate can be easily obtained blessing Secretary of fluconazole, the reaction yield of 85-90%.
In the prior art, the removal of benzyl preparation blessing Secretary of fluconazole, the use of a pressure hydrogenation, relatively harsh reaction conditions; and blessing Secretary of fluconazole in water and slightly soluble in methanol, for blessing Secretary fluconazole further refined and purified more difficult. The present invention aims to provide a new and suitable for industrial production methods blessing Secretary fluconazole.
Example 1
2- (2,4-gas-phenyl) -1,3-bis (1H-1, 2,4- two P sat 1-yl) -2-propyl-di-benzyl-pity Cool ( Preparation blessing Secretary fluconazole dibenzyl ester)
Step The method according to CN1210540A in Example 1 A or Method B of (a), was prepared to give the title compound, having 1H-NMR shown in Figure 1 (SOi) MHz, DMS0-D6) spectrum.
Example 2
2_ (2,4_ two gas-phenyl) -1, double 3_ (1H-1, 2,4_ two 1-yl) propyl pity _2_ di press (forskolin gas
Itraconazole ammonium salt) Preparation
Formula III blessing Secretary fluconazole two benzyl ester (566g, lmol), 120g of dry Pd / C (containing 5% palladium) and ammonium formate (315g, 5mol) in methanol (6L), and stirred under reflux for 5h , TLC monitoring completion of the reaction was filtered, 50 ° C the solvent was distilled off under reduced pressure, ethanol was added (566ml), stirred for beating, and filtered to give a solid forskolin fluconazole salt 415g, yield 98.8%.
] lH-Mffi (500MHz, DMS0-D6) δ: 4.87-4.90, 5.58-5.61,6.56-6.60, 6.94-7.03,7.52-7.61,8.96, having 1H-NMR shown in Figure 2 (500MHz, DMS0 -D6) spectrum.
Example 3
2- (2,4-gas-phenyl) -1,3-bis (1H-1, 2,4- two 1-yl) -2-propyl-pity acid dioxide Cool (forskolin
Fluconazole) Preparation of
[0052] Formula II forskolin fluconazole salt (420g, Imol), in water (IL) while stirring, filtered, 2mol / L sulfuric acid aqueous solution (500ml), 5 ° C under stirring for lh, filtered, cold water ( 200ml) wash, 50 ° C under dry blessed Division fluconazole 379g, yield 98%.
1H-Mffi (SOOMHz) DMSO-De) δ:. 5.09-5.12,5.25-5.28,6.80-6.84,7.05-7.16,7.77,8.55,10.32 [0054] Example 4
2_ (2,4_ two gas-phenyl) -1, double 3_ (1Η-1, 2,4_ two 1-yl) propyl pity _2_ di press (forskolin gas itraconazole salt) Preparation
Under nitrogen, forskolin fluconazole dibenzyl ester (566g, lmol), 84g of dry Pd / C (5% containing button) and ammonium formate (189g, 3mol) in anhydrous methanol (5L) in the mixture was stirred at reflux for 5h, TLC monitoring completion of the reaction was filtered, 50 ° C the solvent was distilled off under reduced pressure, ethanol was added (300ml), stirred for beating, and filtered to give a solid forskolin fluconazole salt 410g, yield 97.5%.
Example 5
2_ (2,4_ two gas-phenyl) -1, double 3_ (1H-1, 2,4_ two 1-yl) propyl pity _2_ di press (forskolin gas itraconazole salt) Preparation
Under nitrogen, forskolin fluconazole dibenzyl ester (566g, lmol), 30g of dry Pd / C (containing 10% palladium) and ammonium formate (315g, 5mol) in anhydrous methanol (5L) in the mixture was stirred at reflux for 5h, TLC monitoring completion of the reaction was filtered, 50 ° C the solvent was distilled off under reduced pressure, ethanol was added (300ml), stirred for beating, and filtered to give a solid forskolin fluconazole salt 405g, yield 96.4%.
Example 6
2_ (2,4_ two gas-phenyl) -1, double 3_ (1H-1, 2,4_ two 1-yl) propyl pity _2_ di press (forskolin gas itraconazole salt) Preparation
Under nitrogen, forskolin fluconazole dibenzyl ester (566g, lmol), 30g of dry Pd / C (containing 10% palladium) and ammonium formate (315g, 5mol) in ethanol (12L) and stirred was refluxed for 5h, TLC monitoring completion of the reaction, was filtered, 50 ° C the solvent was distilled off under reduced pressure, ethanol was added (300ml), stirred for beating, and filtered to give a solid forskolin fluconazole salt 395g, 94% yield.
Example 7
2_ (2,4_ two gas-phenyl) -1, double 3_ (1H-1, 2,4_ two 1-yl) propyl pity _2_ di press (forskolin gas itraconazole salt) Preparation
forskolin fluconazole dibenzyl ester (566g, lmol), 170g of dry Pd / C (containing 5% of palladium) and ammonium formate (315g, 5mol) in ethanol (16L) was stirred under reflux for 5h, TLC monitoring completion of the reaction was filtered, 50 ° C the solvent was distilled off under reduced pressure, ethanol was added (300ml), stirred for beating, and filtered to give a solid forskolin fluconazole salt 398g, yield 94.7%.
Example 8
2_ (2,4_ two gas-phenyl) -1, double 3_ (1H-1, 2,4_ two 1-yl) propyl pity _2_ di press (forskolin gas itraconazole salt) Preparation
Under nitrogen, forskolin fluconazole dibenzyl ester (566g, lmol), 120g of dry Pd / C (containing 5% palladium) and ammonium formate (315g, 5mol) in isopropanol (12L) in the mixture was stirred at reflux for 5h, TLC monitoring completion of the reaction was filtered, 50 ° C the solvent was distilled off under reduced pressure, ethanol was added (300ml), stirred for beating, and filtered to give a solid forskolin fluconazole salt 402g, a yield of 95.7%.
Example 9
2_ (2,4_ two gas-phenyl) -1, double 3_ (1H-1, 2,4_ two 1-yl) propyl pity _2_ di press (forskolin gas itraconazole salt) Preparation
[0071] under nitrogen blessing Secretary fluconazole dibenzyl ester (566g, lmol), 60g of dry Pd / C (containing 5% palladium) and ammonium formate (504g, 8mol) in methanol (8L) in, 50 ° C under stirring reaction 40h, TLC monitoring completion of the reaction, was filtered, 50 ° C the solvent was distilled off under reduced pressure, ethanol was added ^ OOml), stirred for beating, and filtered to give a solid forskolin fluconazole salt 398g, yield 94.8%.
Example 10
2_ (2,4_ two gas-phenyl) -1, double 3_ (1H-1, 2,4_ two 1-yl) propyl pity _2_ di press (forskolin gas itraconazole salt) Preparation
Under nitrogen, forskolin fluconazole dibenzyl ester (5668,111101), 8 (^ dry? (1 / (:( containing palladium 5%) and ammonium formate (315g, 5mol) for n-propyl alcohol (12L) in, 60 ° C the reaction was stirred 20h, TLC monitoring completion of the reaction was filtered, 50 ° C the solvent was distilled off under reduced pressure, ethanol was added (300ml), stirred for beating, and filtered to give a solid forskolin fluconazole salt 398g 95% yield.
Example 11
2- (2,4-gas-phenyl) -1,3-bis (1H-1, 2,4- sit two P-1-yl) -2-propyl-pity acid dioxide Cool (forskolin fluconazole) Preparation of [0077] under nitrogen blessing Secretary fluconazole dibenzyl ester 566 g (Imol) adding 56g of dry Pd / C (containing 5% palladium), methanol 6L, 315 g of ammonium formate, stirring boil under reflux for 5h, TLC after completion of the reaction was filtered, 50 ° C the solvent was distilled off under reduced pressure, addition of IL of water and dissolved with stirring, filtered, 2mol / L sulfuric acid 500mL, 5 ° C with stirring to precipitate lh, filtered, 200mL cold water, 50 ° C drying 365 g, 95% yield.
Example 12 forskolin fluconazole salt and HPLC detection methods blessing Secretary fluconazole:
High performance liquid chromatography (Chinese Pharmacopoeia 2010 edition two Appendix VD): octadecylsilane bonded silica as a filler, Column: Thermo BDS C18 (4.6 X 150mm, 3.5 μ m); methanol as mobile phase A, phosphate buffer (take potassium dihydrogen phosphate 0.68g, set 1000ml water, triethylamine 6ml, adjusted to pH 5.0 with phosphoric acid) as the mobile phase B, a flow rate of 1.0ml / min; column temperature 35 ° C; detection wavelength was 210nm, linear gradient.
After the examination, according to the peak area calculation, purity prepared in Example 2-11 was the implementation of the target product of 99.5%.
| Patent | Submitted | Granted |
|---|---|---|
| Nanoparticulate Anidulafungin Compositions and Methods for Making the Same [US2009238867] | 2009-09-24 | |
| IMIDAZOPYRIDINE SUBSTITUTED TROPANE DERIVATIVES WITH CCR5 RECEPTOR ANTAGONIST ACTIVITY FOR THE TREATMENT OF HIV AND INFLAMMATION [US7790740] | 2008-02-21 | 2010-09-07 |
| Pharmaceutical formulations of cyclodextrins and antifungal azole compounds [US2007082870] | 2007-04-12 | |
| TRIAZOLE DERIVATIVES USEFUL IN THERAPY [EP0880533] | 1998-12-02 | 2002-06-12 |
| Triazole derivatives useful in therapy [US6790957] | 2003-07-31 | 2004-09-14 |
| Process for controlling the hydrate mix of a compound [US7323572] | 2004-01-15 | 2008-01-29 |
| TOPICAL TERBINAFINE FORMULATIONS AND METHODS OF ADMINISTERING SAME FOR THE TREATMENT OF FUNGAL INFECTIONS [US7820720] | 2010-04-29 | 2010-10-26 |
| PHARMACEUTICAL COMPOSITION COMPRISING PHENYLAMIDINE DERIVATIVE AND METHOD OF USING THE PHARMACEUTICAL COMPOSITION IN COMBINATION WITH ANTIFUNGAL AGENT [US8173157] | 2010-04-22 | 2012-05-08 |
| COMPOSITIONS COMPRISING POLYUNSATURATED FATTY ACID MONOGLYCERIDES OR DERIVATIVES THEREOF AND USES THEREOF [US8222295] | 2009-11-26 | 2012-07-17 |
| MASKED CARBOXYLATE NEOPENTYL SULFONYL ESTER CYCLIZATION RELEASE PRODRUGS OF ACAMPROSATE, COMPOSITIONS THEREOF, AND METHODS OF USE [US2009069419] | 2009-03-12 |
| Patent | Submitted | Granted |
|---|---|---|
| Triazole derivatives useful in therapy [US2005130940] | 2005-06-16 | |
| Chemical compounds [US7309790] | 2005-06-16 | 2007-12-18 |
| Combination of voriconazole and an antifungal CYP2C19 inhibitor [US2005182074] | 2005-08-18 | |
| Inhibitors of fungal invasion [US2004106663] | 2004-06-03 | |
| Triazole derivatives useful in therapy [US6977302] | 2004-11-25 | 2005-12-20 |
| Pharmaceuticals [US7691877] | 2007-08-23 | 2010-04-06 |
| SIMPLE PANTOIC ACID ESTER NEOPENTYL SULFONYL ESTER CYCLIZATION RELEASE PRODRUGS OF ACAMPROSATE, COMPOSITIONS THEREOF, AND METHODS OF USE [US7994218] | 2009-03-26 | 2011-08-09 |
| COMPLEX PANTOIC ACID ESTER NEOPENTYL SULFONYL ESTER CYCLIZATION RELEASE PRODRUGS OF ACAMPROSATE, COMPOSITIONS THEREOF, AND METHODS OF USE [US8168617] | 2009-03-19 | 2012-05-01 |
| Purine derivatives [US7642350] | 2006-11-23 | 2010-01-05 |
| IMIDAZOPYRIDINONES [US2009221631] | 2009-09-03 |
IMPURITIES
1
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
{[2-(2,4-Difluorophenyl)-1,3-bis(1H-1,2,4-triazol-1-yl)propan-2-yl]oxy}phosphonic acid
|
|
| Clinical data | |
| AHFS/Drugs.com | International Drug Names |
| Legal status |
|
| Routes of administration |
IV |
| Identifiers | |
| CAS Number | 194798-83-9 |
| ATC code | None |
| PubChem | CID 214356 |
| ChemSpider | 185843 |
| UNII | 3JIJ299EWH |
| ChEMBL | CHEMBL1908301 |
| Chemical data | |
| Formula | C13H13F2N6O4P |
| Molar mass | 386.25 g/mol |
| CN1210540A * | Jan 27, 1997 | Mar 10, 1999 | 辉瑞研究开发公司 | Triazole derivatives useful in therapy |
| CN1789270A * | Dec 16, 2005 | Jun 21, 2006 | 西安新安医药科技有限公司 | Mycotic ingection-resisting fosfluconazole hydrate and preparation method thereof |
| CN101890028A * | Feb 22, 2007 | Nov 24, 2010 | 卫材R&D管理有限公司 | Stabilized pharmaceutical composition |
| CN102439018A * | Mar 3, 2010 | May 2, 2012 | 塞普斯制药有限公司 | Fosfluconazole derivatives, synthesis, and use in long acting formulations |
| US20040007689 * | Jun 23, 2003 | Jan 15, 2004 | Pfizer Inc. | Process for controlling the hydrate mix of a compound |
| Reference | ||||
|---|---|---|---|---|
| 1 | * | ARTHUR BENTLEY等: “The Discovery and Process Development of a Commercial Route to the Water Soluble Prodrug, Fosfluconazole“, 《ORGANIC PROCESS RESEARCH & DEVELOPMENT》, vol. 6, no. 2, 18 December 2001 (2001-12-18), XP002491526, DOI: doi:10.1021/op010064+ | ||
| 2 | * | 国大亮 等: “福司氟康唑“, 《齐鲁药事》, vol. 24, no. 1, 30 January 2005 (2005-01-30), pages 60 | ||
| 3 | * | 村上尚道: “fosfluconazole“, 《NEW DRUGS OF THE WORLD:2003》, vol. 33, no. 10, 15 September 2004 (2004-09-15), pages 56 | ||
//////UK-292663, UK 292663, F-FLCZ, F FLCZ, Fosfluconazole, 194798-83-9, UNII-3JIJ299EWH, 3JIJ299EWH, NCGC00182029-01
Fc1ccc(c(F)c1)C(OP(=O)(O)O)(Cn2ncnc2)Cn3ncnc3
Bromuconazole
Bromuconazole
116255-48-2; HSDB 7419
| Molecular Formula: | C13H12BrCl2N3O |
|---|---|
| Molecular Weight: | 377.06388 g/mol |
1-[[4-bromo-2-(2,4-dichlorophenyl)oxolan-2-yl]methyl]-1,2,4-triazole


| Patent | Submitted | Granted |
|---|---|---|
| Phthalamide derivatives [US7132455] | 2006-02-16 | 2006-11-07 |
| Crystal modification II of 2-[2-(1-chloro-cyclopropyl)-3-(2-chlorophenyl)-2-hydroxy-propyl]-2,4-dihydro-3H-1,2,4-triazole-3-thione [US7176226] | 2006-05-18 | 2007-02-13 |
| Anthranilamide insecticides [US7211270] | 2006-03-09 | 2007-05-01 |
| 2-Phenyl-2-substituted-1,3-diketones [US7227043] | 2006-03-16 | 2007-06-05 |
| Biphenyl derivatives and their use as fungicides [US7241721] | 2006-05-11 | 2007-07-10 |
| Cyano anthranilamide insecticides [US7247647] | 2006-05-25 | 2007-07-24 |
| 3-Phenyl substituted 3-substituted-4ketolactams and ketolactones [US7329634] | 2006-05-04 | 2008-02-12 |
| Substituted isoxazoles as fungicides [US7338967] | 2006-04-06 | 2008-03-04 |
| Insecticidal anthranilamides [US7338978] | 2006-04-13 | 2008-03-04 |
| Pyrazolyl carboxanilides for controlling unwanted microorganisms [US7358214] | 2006-04-27 | 2008-04-15 |
//////////////////
C1=CC(=C(C=C1Cl)Cl)C2(CC(CO2)Br)C[N]3C=NC=N3
| Bromuconazole | |
|---|---|
 |
|
| Identification | |
| No CAS | |
| SMILES | |
| InChI | |
| Apparence | cristaux incolores ou poudre sans odeur1. |
| Propriétés chimiques | |
| Formule brute | C13H12BrCl2N3O [Isomères] |
| Masse molaire2 | 377,064 ± 0,017 g/mol C 41,41 %, H 3,21 %, Br 21,19 %, Cl 18,8 %, N 11,14 %, O 4,24 %, |
| Propriétés physiques | |
| T° fusion | 84 °C1 |
| Solubilité | dans l’eau : 0,5 g·l-11 |
| Pression de vapeur saturante | à 25 °C : négligeable1 |
Saperconazole

Saperconazole
The most common systemic fungal infections in humans are blastomycosis, candidosis, aspergillosis, histoplasmosis, coccidioidomycosis, paracoccidioidomycosis, and cryptococcosis.
Fungal diseases are often confined to typical anatomic sites, and many involve a primary focus in the lung, with more characteristic manifestations of specific fungal infections appearing once the infection spreads from a primary site. For example, blastomycosis primarily involves the lungs, and occasionally spreads to the skin. Similarly, the primary form of coccidioidomycosis occurs as an acute, benign, self-limiting respiratory disease, which can then progress to a chronic, often-fatal infection of the skin, lymph glands, liver, and spleen. Other infectious diseases such as paracoccidioidomycosis and candidiasis present in different manners, and depending on the etiology, may exhibit several forms involving internal organs, the lymph nodes, skin, and mucous membranes. Diagnosis of specific fungal diseases can be made by isolation of the causative fungus from various specimens, such as sputum, urine, blood, or the bone marrow, or with certain fungus types, through evidence of tissue invasion.
Many patients suffering from severe systemic fungal infections are hardly, or not at all, able to receive medication via oral administration, as such patients are often in a coma or suffering from severe gastroparesis. As a result, the use of insoluble or sparingly soluble antifungals such as itraconazole free base, which are difficult to administer intravenously to treat such patients, is significantly impeded.
Local or superficial fungal infections are caused by dermatophytes or fungi that involve the outer layers of the skin, nails, or hair. Such infections may present as a mild inflammation, and can cause alternating remissions and eruptions of a gradually extending, scaling, raised lesion. Yeast infections, such as candidiasis and oral candidiasis (thrush), are usually localized to the skin and mucous membranes, with the symptoms varying depending on the site of infection. In many instances, such infections appear as erythematous, often itchy, exudative patches in the groin, axillas, umbilicus, between toes, and on finger-webs. Oral thrush involves an inflamed tongue or buccal mucosa, typically accompanied by white patches of exudate. Chronic mucocutaneous candidiasis is manifested in the form of red, pustular, crusted, thickened lesions on the forehead or nose.Itraconazole or (±)-£is-4-[4-[4-[4-[[2-(2,4-dichlorophenyl)-2-(lH-l-2,4-triazol-l- ylmethyl)- 1 ,3-dioxolan-4-yl]methoxy]phenyl]- 1 -ρiperazinyl]phenyl]-2,4-dihydro-2-( 1 – methyl-propyl)-3H-l,2,4-triazol-3-one, is a broadspectrum antifungal compound developed for oral, parenteral and topical use and is disclosed in US-4,267,179.
The development of effϊcaceous pharmaceutical compositions of itraconazole and saperconazole is hampered considerably by the fact that said compounds are only very sparingly soluble in water. The solubility of both compounds can be increased by complexation with cyclodextrins or derivatives thereof as described in WO 85/02767 and US-4,764,604.
Unexpectedly, it has now been found that each of the individual stereoisomers of itraconazole and saperconazole have greater water solubility than the diastereomeric mixtures of said compounds, in particular when complexed with cyclodextrin or its derivatives. As a result, pharmaceutical compositions having good bioavailability, yet comprising less cyclodextrin as a complexing agent, can be prepared. The present invention is concemced with the stereoisomeric forms of itraconazole (X = CI) and saperconazole (X = F), which may be represented by the formula
cis-©,and the pharmaceutically acceptable acid addition salt forms thereof. The three asterisks indicate the three chiral centers, and ‘cis’ means that the (lH-l,2,4-triazol-l-ylmethyl) moiety and the substituted phenoxy moiety are located at the same side of the plane defined by the 1,3-dioxolane ring.
The four possible stereoisomeric cis forms can be described using various rules of nomenclature. The following tables show the correlation among the C. A. stereochemical descriptor, the absolute configuration at each of the chiral centers and the specific optical
20 rotation [α]jj in 1% methanol (itraconazole; table I) (saperconazole; table H).
Table I
itraconazole
Table π
saperconazole
Itraconazole is a broad-spectrum antifungal agent developed for oral, parenteral and topical use, and is disclosed in U.S. Patent No. 4,267,179. Itraconazole is a synthetic triazole derivative that disrupts the synthesis of ergosterol, the primary sterol of fungal cell membranes. This disruption appears to result in increased permeability and leakage of intracellular content, and at high concentration, cellular internal organelles involute, peroxisomes increase, and necrosis occurs.
As set forth in the USP Dictionary of Drug Names and USAN, itraconazole is defined as 4-[4-[4-[4- [[2-(2,4-dichlorophenyl)-2-(lH-l,2,4-triazol-l-ylmethyl)-l,3-dioxolan-4-yl] methoxy]phenyl]-l-piperazinyl]phenyl]- 2,4-dihydro-2-(l-methylpropyl)-3H-l,2,4-triazol-3-one, or alternatively, as (±)-l-5ec-butyl-4-[/7-[4-[/7-[[(2R*,4S*)-2-(2,4-dichlorophenyl)-2-(lH-l,2,4-triazol-l-ylmethyl)-l,3-dioxolan-4-yl]methoxy]phenyl]-l-piperazinyl]phenyl]-Δ2-l,2,4-triazolin-5-one. There are three asymmetric carbons in itraconazole: one in the sec-butyl side chain on the triazolone and two in the dioxolane ring. As a result, eight possible stereoisomers of itraconazole exist: (R,R,R), (S,S,S), (R,R,S), (S,S,R), (R,S,S), (R,S,R), (S,R,S), and (S,R,R).
(±)Cz‘s-Itraconazole comprises a mixture of only those isomers that describe a “cis” relationship in the dioxolane ring, i.e., the (1Η-1, 2, 4-triazol-l-ylmethyl) moiety and the substituted phenoxy moiety are located on the same side of a plane defined by the 1, 3-dioxolane ring. By convention, the first represented chiral center is at the C-2 position of the dioxolane ring, the second is at the C-4 position of the dioxolane ring, and the third is in the sec-butyl group. Hence, (±)c.s-itraconazole is a mixture of (R,S,S), (R,S,R), (S,R,S) and (S,R,R) isomers.
The four possible stereoisomeric cis forms of itraconazole, and
diastereomeric pairs thereof, are described in more detail in U.S. Patent Nos. 5,474,997 and 5,998,413. In general, the individual stereoisomeric forms of c s-itraconazole have antifungal properties, and contribute to the overall activity of (±)cw-itraconazole.
(±)Ciy-Itraconazole free base is only very sparingly soluble in water, and thus it is extremely difficult to prepare effective pharmaceutical compositions containing the same. A number of means have been used to increase the solubility of itraconazole free base, including complexing or co-formulation with cyclodextrins or derivatives thereof, as described in U.S. Patent No. 4,764,604, U.S. Patent No.5,998,413, and U.S. Patent No. 5,707,975, and coating beads with a film comprising a hydrophilic polymer and itraconazole, as described in U.S. Patent No. 5,633,015.
[0014] Another approach to increase solubility of itraconazole focuses on preparation of the stereoisomers of c s-itraconazole, and in particular (2R, 4S) itraconazole, which may comprise a mixture of two diastereomers ((R,S,S) and
(R,S,R)), as described in U.S. Patent Nos. 5,414,997 and 5,998,413.
Commercially available itraconazole (SPORANOX® brand (±)cis-itraconazole, Janssen Pharmaceutica Products, L.P., Titusville, NJ, U.S.A.) is a free base and a racemic mixture of the cis isomer in the dioxolane ring and is represented by structural formula (I):

(i)
SPORANOX has been approved for use as an antifungal agent for treating immunocompromised and non-immunocompromised patients having: blastomycosis (pulmonary and extrapulmonary); histoplasmosis, including chronic cavitary pulmonary disease and disseminated non-meningeal histoplasmosis; and aspergillosis. In addition, in non-immunocompromised patients, it has been approved for treatment of onychomycosis. See generally, Physician ‘s Desk Reference, 56th ed. (2002). The compound has also been investigated for use in coccidioidomycosis, cryptococcosis, dermatophyte, and candidiasis infections.
Adverse effects associated with the administration of (±)cts-itraconazole free base include nausea, vomiting, anorexia, headache, dizziness, hepatotoxicity, and inhibition of drug metabolism in the liver, leading to numerous, clinically significant, adverse drug interactions. See, Physician ‘s Desk Reference, 56th ed. (2002); Honig et al., J. Clin. Pharmacol. 33:1201-1206 (1993) (terfenadine interaction); Gascon and Dayer, Eur. J. Clin. Pharmacol., 41_:573-578 (1991) (midazolam interaction); and Neuvonen et al, Clin. Pharmacol. Therap., 60:54-61 (1996) (lovastatin interaction). Reactions associated with hypersensitivity, such as urticaria and serum liver enzymes elevation, are also associated with the administration of the drug. A more serious, though less common, adverse effect is hepatotoxicity. See, e.g., Lavrijsen et al., Lancet, 340:251-252 (1992).
In addition, as discussed herein, c/s-itraconazole free base is only very sparingly soluble in water. Thus, due to its relative non-polarity and insolubility, itraconazole free base suffers from two other drawbacks: it cannot be readily formulated in parenteral solution, and it does not effectively penetrate the blood-brain barrier. The latter problem is exacerbated by drug interactions, such as one observed between itraconazole free base and valproate, as described in Villa et al. , Rev. Inst. Med. Trop., Sao Paulo, pp. 231-234 (Jul-Aug 2000), which is incorporated by reference herein in its entirety. In another case of CNS fungal infection, extremely high doses of itraconazole free base were used to treat residual aspergillus infection, as reported by Imai et al., Intern. Med, 38(10):829-832 (1999), which is incorporated by reference herein in its entirety. As a result, numerous therapeutic indications that require rapid achievement of effective blood levels or access to the CNS are difficult to treat or beyond treatment with itraconazole free base.
Furthermore, the emergence of antifungal resistance (e.g., in Aspergillus fumigatus isolates as described by Dannaoui et al., J. Antimicrob. Chemother., 47:333-340 (2001), which is incorporated by reference herein in its entirety) presents an added challenge to the efficacy of itraconazole free base. For those strains of fungi that show resistance, high and relatively constant levels of itraconazole free base must be produced in the target organs of infected patients.
Over the years, a number of formulation routes have been used in order to enhance the adsorption and bioavailability of itraconazole. For example, the currently marketed SPORANOX® solid dosage capsule form of itraconazole free base utilizes sugar-based beads coated with a hydrophilic polymer and an amorphous film of itraconazole. See Physicians Desk Reference, 56th ed., pp.1800- 1804 (2002); and U.S. Patent No. 5,633,015. This dosage form requires up to two capsules three times daily depending on the condition being treated.
Even with the various formulation routes, the dosage amounts and dose frequency for itraconazole can be burdensome to patients. In addition, administration of existing dosage forms of itraconazole have shown significant variability in bioavailability and adsorption, which likely results from food effects. See, Physician ‘s
Desk Reference, 56th ed., pp. 1800-1804 (2002). Thus, it would be desirable to increase bioavailability and adsorption and decrease the per-dose pill count and decrease dosing frequency (e.g., twice a day to once a day) associated with administration of itraconazole in order to provide an improvement over current therapy, particularly with regard to patient compliance, convenience, ease of ingestion, especially with regard to immunocompromized polypharmacy patients (e.g., AIDS or cancer patients).
Posaconazole and Saperconazole Chemistry and Uses
Other related conazoles have also been discovered and used as antifungals. Two of these conazoles that are closely structurally related to itraconazole are posaconazole and saperconazole. Posaconazole (CAS Registry Number: 171228-49-2; CAS Name: 2,5-Anhydro-l ,3,4-trideoxy-2-C-(2,4-difluorophenyl)-4-[[4-[4-[4-[l -[(1 S,2S)- 1 -ethyl-2-hydroxypropyl]- 1 ,5-dihydro-5-oxo-4H- 1 ,2,4-triazol-4-yl]phenyl]- 1 -piperazinyl]phenoxy]methyl]- 1 -( 1 H- 1 ,2,4-triazol- 1 -yl)-D-t/Veo-pentitol; Additional Name: (3R-c s)-4-[4-[4-[4-[5-(2,4-difluorophenyl)-5-(l,2,4-triazol-l-ylmethyl)tetrahydrofuran-3-ylmethoxy]phenyl]piperazin- 1 -yl]phenyl]-2-[l (S)-ethyl-2(S)-hydroxypropyl]-3,4-dihydro-2H-l,2,4-triazol-3-one) is represented by structural formula (II):

(II)
Saperconazole (CAS Registry Number: 110588-57-3; CAS Name: 4-[4-[4-[4-[[2-(2,4-Difluorophenyl)-2-(lH-l,2,4-triazol-l-ylmethyl)-l,3-dioxolan-4-yl]methoxy]phenyl]- 1 -piperazinyl]phenyl]-2,4-dihydro-2-(l -methylpropyl)-3H- 1 ,2,4-triazol-3-one; Additional Name: (±)-l-sec-butyl-4-[ -[4-| -[[(2R* 4S*)-2-(2,4- difluorophenyl)-2-( 1 H- 1 ,2,4-triazol- 1 -ylmethyl)- 1 ,3 -dioxolan-4-yl]methoxy]phenyl]- 1 -piperazinyl]phenyl]-Δ2-l,2,4-triazolin-5-one) is represented by structural formula (III):

(III)
Consequently, there is a need for soluble forms of conazoles including cis itraconazole, posaconazole and saperconazole that can be readily formulated for use in various modes of administration, including parenteral and oral administration.
A. Preparation of intermediates: Example 1a) utilizing water separator, by 200 parts of glycerin, 90 parts of 1- (2,4-difluorophenyl) -2- (1H-1,2,4- three mixture of 1-yl) ethanone, 600 parts of methanesulfonic acid, 190 parts of benzene was stirred first at reflux for 3 hours, then stirred at room temperature overnight. The reaction mixture was added dropwise a solution of sodium bicarbonate. The product was extracted with chloroform, the extract was washed with water, dried, filtered and evaporated. With 4-methyl-2-pentanone and the residue triturated product was filtered off and dried, yielding 80 parts (67.2%) (cis + trans) -2- (2,4-difluorophenyl) -2- (1H-1,2,4- triazol-1-ylmethyl) -1,3-dioxolane-4-methanol (intermediate 1).
b) by 69 parts of 3,5-dinitrobenzoyl chloride, 80 parts of (cis + trans) -2- (2,4-difluorophenyl) -2- (1H-1,2,4- triazol-1-ylmethyl) -1,3-dioxolane-4-methanol, 400 parts of pyridine and 520 parts of dichloromethane was stirred at room temperature for 3 hours. The reaction mixture was evaporated, and the residue was dissolved in water. The product was extracted with chloroform. The extract was dried, filtered and evaporated. The residue was subjected to silica gel column chromatography, eluting with chloroform / methanol (99:1v / v). Pure fractions were collected, the eluent was evaporated, to give 90 parts (70.4%) of cis -2- (2,4-difluorophenyl) -2- (1H-1,2,4- triazol-1 ylmethyl) -1,3-dioxolane-4-methanol 3,5-dinitrobenzoate (residue) (intermediate 2).
c) by 90 parts of (cis) -2- (2,4-difluorophenyl) -2- (1H-1,2,4- triazol-1-ylmethyl) -1,3-dioxo- dioxolan-4-methanol 3,5-dinitrobenzoate, 16 parts of 50% sodium hydroxide solution, 800 parts of 1,4-dioxane, 400 parts of water and the mixture was stirred at room temperature overnight. The reaction mixture was poured into water and the product was extracted with dichloromethane, extracts washed with water, dried, filtered and evaporated. With 4-methyl-2-pentanone and the residue triturated product was filtered off and dried, yielding 30 parts (56.0%) of cis -2- (2,4-difluorophenyl) -2- (1H-1, 2,4-triazol-1-ylmethyl) -1,3-dioxolane-4-methanol (residue) (intermediate 3).
d) by 11.4 parts of methanesulfonyl chloride, 25 parts of cis -2- (2,4-difluorophenyl) -2- (1H-1,2,4- triazol-1-ylmethyl) -1, mixture of 1,3-dioxolane-4-methanol, 300 parts of pyridine, 390 parts of dichloromethane was stirred at room temperature for 3 hours. The reaction mixture was evaporated, and the residue was dissolved in chloroform. The organic phase was dried, filtered and evaporated. The residue was triturated with dipropyl ether. The product was filtered off and dried, yielding 29.4 parts (93.2%) of cis -2- (2,4-difluorophenyl) -2- (1H-1,2,4- triazol-1-ylmethyl) – 1,3-dioxolane-4-methanol methanesulfonate (residue) of intermediate 4).
In a similar manner there were also prepared: cis-2- (2,4-difluorophenyl) -2- (1H- imidazol-1-ylmethyl) -1,3-dioxolane-4-methanol mesylate ethanedioate (1/1) (interm. 5).
Example 2a) over 2 hours, dissolved in 100 parts of pyridine 121.2 parts of 2-naphthalenesulfonyl chloride was added dropwise to a stirred, was dissolved in 1300 parts of dichloromethane, and 122.0 parts of (cis + trans ) -2- (2,4-difluorophenyl) -2- (1H-1,2,4- triazol-1-ylmethyl) -1,3-dioxolane-4-methanol and 1.0 parts of N, N- dimethyl-4-pyridin-amine solution. Upon completion, stirring was continued at room temperature overnight. The reaction mixture was washed twice with water, and evaporated under reduced pressure. The residue was purified by silica gel column chromatography, eluting with chloroform. Pure fractions were collected, the eluent was evaporated. The residue was crystallized from 4-methyl-2-pentanone. The product was filtered off and dried, yielding 102.3 parts (51.0%) of cis – [[2- (2,4-difluorophenyl) -2- (1H-1,2,4- triazol-1-yl-methyl ) -1,3-dioxolan-4-yl] methyl] -2-naphthalene sulfonate; mp139.5 ℃ (intermediate 6).
Example 3a) at 70 ℃, under nitrogen atmosphere, by 9.0 parts of 4- [4- (4-nitrophenyl) -1-piperazinyl] phenol, 13.6 parts of cis-2- [2,4- difluorophenyl) -2- (1H-1,2,4- triazol-1-ylmethyl) -1,3-dioxolane-4-methanol methanesulfonate ester, 6.0 parts of potassium hydroxide and 90 parts of a mixture of DMF was stirred overnight. After cooling, the reaction mixture was diluted with water. The precipitated product was filtered off and purified by silica gel column chromatography, the chloroform / ethyl acetate / hexane / methanol (500:300:200:0.5v / v / v / v) mixture as eluent. Pure fractions were collected, the eluent was evaporated. The residue was crystallized 4-methyl-2-pentanone. The product was filtered off and dried, yielding 6.69 parts (38.5%) of cis -1- [4 – [[2- (2,4-difluorophenyl) -2- (1H-1,2,4- triazol – 1- ylmethyl) -1,3-dioxolan-4-yl] methoxy) phenyl] -4- (4-nitrophenyl) piperazine; mp169.8 ℃ (Intermediate 7) .
b) at atmospheric pressure, 50 ℃, with 2 parts of 5% palladium – on-charcoal catalyst by 38.3 parts of cis -1- [4 – [[2- (2,4-difluorophenyl) -2- (1H -1,2,4-triazol-1-ylmethyl) -1,3-dioxolan-4-yl] methoxy] phenyl] -4- (4-nitrophenyl) piperazine, 2 parts of a solution of thiophene (4% solution in methanol) and 600 parts of 2-methoxy-ethanol mixture. After absorption of the calculated amount of hydrogen finished, hot filtered to remove the catalyst, and the filtrate was saturated with water. After cooling, the precipitated product was filtered off, washed with water and 2-propanol and crystallized from 1,4-dioxane. The product was filtered off and dried, yielding 22.7 parts (62.6%) of cis-4- [4- [4 – [[2- (2,4-difluorophenyl) -2- (1H-1,2,4- triazol-1-ylmethyl) -1,3-dioxolan-4-yl] methoxy] phenyl] -1-piperazinyl] aniline; mp193.0 ℃ (interm. 8).
Example 4a) by 10 parts of 2,4-dihydro-4- [4- [4- [4-methoxyphenyl) -1-piperazinyl] phenyl] -3H-1,2,4- triazol-3-one (U.S. Patent No. 4,267,179 in the implementation of the method in Example ⅩⅦ obtained), 1.5 parts of sodium hydride (50% dispersion), 300 parts of the mixture consisting of dimethyl sulfoxide, at 60 ℃ under a nitrogen atmosphere begging, stirring, until no bubble up. Was then added 5.24 parts of 2-bromopropane, and at 60 ℃, stirring was continued for 1 hour. Further added 1.5 parts of sodium hydride (50% dispersion) and stirring was continued until no more bubble up. Then 5.24 parts of 2-bromopropane was added, and the whole was stirred for 1 hour at 60 ℃. The reaction mixture was cooled, poured into water and the product was extracted with chloroform. The extract was washed with water, dried, filtered and evaporated. The residue was purified by silica gel column chromatography, eluting with chloroform / methanol (99:1v / v). Pure fractions were collected, the eluent was evaporated, the residue was crystallized in 1-butanol, yielding 5.2 parts (47% (2,4-dihydro-4- [4- [4- (4-methoxyphenyl ) -1-piperazinyl] phenyl] -2- (1-methylethyl) -3H-1,2,4- triazol-3-one; mp209.5 ℃ (intermediate 9).
b) by 4.7 parts of 2,4-dihydro-4- [4- [4- (4-methoxyphenyl) -1-piperazinyl] phenyl] -2- (1-methylethyl) -3H-1,2,4- triazol-3-one, a mixture of 75 parts of 48% aqueous hydrobromic acid was stirred at reflux for 3 hours. The reaction mixture was evaporated, and the residue was dissolved in a mixture of methanol and water. With sodium bicarbonate solution, and the whole was, and the product was extracted with chloroform. The extract was dried, filtered and evaporated. The residue was triturated with 2-propanol, yielding 3.9 parts (86%) of 2,4-dihydro-4- [4- [4- (4-hydroxyphenyl) -1-piperazinyl] phenyl] -2 – (1-methylethyl) -3H-1,2,4- triazol-3-one, mp208.4 ℃ (intermediate 10).
PATENT
| EP0006711A1 * | 13 Jun 1979 | 9 Jan 1980 | Janssen Pharmaceutica N.V. | Heterocyclic derivatives of (4-phenylpiperazin-1-yl-aryloxymethyl-1,3-dioxolan-2-yl)-methyl-1H-imidazoles and 1H-1,2,4-triazoles, processes for preparing them and compositions containing them |
| EP0118138A1 * | 24 Jan 1984 | 12 Sep 1984 | Janssen Pharmaceutica N.V. | ((4-(4-(4-Phenyl-1-piperazinyl)phenoxymethyl)-1,3-dioxolan-2-yl)methyl)-1H-imidazoles and 1H-1,2,4-triazoles |
| DE2804096A1 * | 31 Jan 1978 | 3 Aug 1978 | Janssen Pharmaceutica Nv | 1-(1,3-dioxolan-2-ylmethyl)-1h-imidazole und -1h-1,2,4-triazole und deren salze, verfahren zu ihrer herstellung und ihre verwendung bei der bekaempfung pathogener pilze und bakterien |
| Patent | Submitted | Granted |
|---|---|---|
| ORDERED MESOPOROUS SILICA MATERIAL [US2011081416] | 2010-10-15 | 2011-04-07 |
| BENZOYL PEROXIDE COMPOSITION FOR TREATING SKIN [US2011082216] | 2009-10-02 | 2011-04-07 |
| METHODS RELATED TO TIM 3, A TH1-SPECIFIC CELL SURFACE MOLECULE, FOR ACTIVATING ANTIGEN PRESENTING CELLS [US2015044229] | 2014-08-20 | 2015-02-12 |
| METHODS RELATED TO TIM 3, A TH1-SPECIFIC CELL SURFACE MOLECULE, FOR ACTIVATING ANTIGEN PRESENTING CELLS [US2015044230] | 2014-08-20 | 2015-02-12 |
| COSMETIC METHOD FOR INCREASING COLLAGEN EXPRESSION IN SKIN COMPRISING TOPICALLY APPLYING AN EXTRACT OF QUASSIA AMARA [US2015056310] | 2014-08-20 | 2015-02-26 |
| Flexible bone composite [US8771721] | 2013-03-15 | 2014-07-08 |
| Topical formulation [US8513304] | 2012-11-19 | 2013-08-20 |
| Prolonged release bioadhesive therapeutic systems [US8518442] | 2010-07-02 | 2013-08-27 |
| Preparation method for solid dispersions [US8216495] | 2009-03-25 | 2012-07-10 |
| Flexible bone composite [US8221782] | 2011-08-12 | 2012-07-17 |
| Patent | Submitted | Granted |
|---|---|---|
| Crystalline forms of conazoles and methods of making and using the same [US7446107] | 2005-03-31 | 2008-11-04 |
| CIS-itraconazole crystalline forms and related processes, pharmaceutical compositions and methods [US7078526] | 2004-01-29 | 2006-07-18 |
| Novel Saperconazole Crystalline Forms and Related Processes, Pharmaceutical Compositions and Methods [US2007293674] | 2007-12-20 | |
| NOVEL CRYSTALLINE FORMS OF CONAZOLES AND METHODS OF MAKING AND USING THE SAME [US2009088443] | 2009-04-02 | |
| CONTROLLED RELEASE VEHICLES HAVING DESIRED VOID VOLUME ARCHITECTURES [US2014328884] | 2012-12-17 | 2014-11-06 |
| MOLECULES WITH POTENT DHFR BINDING AFFINITY AND ANTIBACTERIAL ACTIVITY [US2014329840] | 2014-05-05 | 2014-11-06 |
| FUNCTIONALLY-MODIFIED OLIGONUCLEOTIDES AND SUBUNITS THEREOF [US2014330006] | 2012-11-15 | 2014-11-06 |
| ASPARTYL-TRNA SYNTHETASE-FC CONJUGATES [US2014335087] | 2012-12-27 | 2014-11-13 |
| GASTRORETENTIVE CONTROLLED RELEASE VEHICLES THAT INCLUDE ETHYLENE COPOLYMERS, ETHYL CELLULOSES, AND/OR THERMOPLASTIC POLYURETHANES [US2014348936] | 2012-12-17 | 2014-11-27 |
| HISTIDYL-TRNA SYNTHETASE-FC CONJUGATES [US2014349369] | 2014-03-14 | 2014-11-27 |
| ASPARTYL-TRNA SYNTHETASES [US2014302075] | 2012-12-06 | 2014-10-09 |
| Rhinosinusitis Prevention and Therapy with Proinflammatory Cytokine Inhibitors [US2014311482] | 2014-01-24 | 2014-10-23 |
| POLYSACCHARIDE ESTER MICROSPHERES AND METHODS AND ARTICLES RELATING THERETO [US2014315720] | 2014-04-04 | 2014-10-23 |
| MODIFIED GREEN TEA POLYPHENOL FORMULATIONS [US2014256616] | 2014-05-19 | 2014-09-11 |
| PLANT-BASED COMPOSITIONS AND USES THEREOF [US2014260466] | 2013-03-15 | 2014-09-18 |
| PLANT-BASED COMPOSITIONS AND USES THEREOF [US2014271928] | 2014-03-14 | 2014-09-18 |
| LIGHT AND ULTRASONIC TRANSDUCER DEVICE [US2014276247] | 2014-03-14 | 2014-09-18 |
| LIGHT AND/OR ULTRASONIC TRANSDUCER DEVICE WITH SENSOR FEEDBACK FOR DOSE CONTROL [US2014276248] | 2014-03-14 | 2014-09-18 |
| PHOTOPROTECTIVE COMPOSITION CONTAINING AN UNMODIFIED GELLING STARCH AND POLYAMIDE PARTICLES [US2014287005] | 2014-03-18 | 2014-09-25 |
| STABILIZED CHEMICAL DEHYDRATION OF BIOLOGICAL MATERIAL [US2014227686] | 2014-04-16 | 2014-08-14 |
| METHODS RELATED TO TIM 3, A TH1-SPECIFIC CELL SURFACE MOLECULE, FOR ACTIVATING ANTIGEN PRESENTING CELLS [US2014242094] | 2014-02-20 | 2014-08-28 |
| NOVEL ENCOCHLEATION METHODS, COCHLEATES AND METHODS OF USE [US2014242153] | 2014-01-30 | 2014-08-28 |
| METHODS OF REDUCING THE PROLIFERATION AND VIABILITY OF MICROBIAL AGENTS [US2010197621] | 2010-08-05 | |
| METHODS OF ADMINISTERING TOPICAL ANTIFUNGAL FORMULATIONS FOR THE TREATMENT OF FUNGAL INFECTIONS [US2010086504] | 2010-04-08 | |
| COMPOSITIONS AND METHODS FOR INCREASING ERYTHROPOIETIN (EPO) PRODUCTION [US2014024699] | 2011-12-09 | 2014-01-23 |
| PROLONGED RELEASE BIOADHESIVE THERAPEUTIC SYSTEMS [US2013310335] | 2013-07-26 | 2013-11-21 |
| Pharmaceutical Composition [US2013315988] | 2011-05-23 | 2013-11-28 |
| Topical Foam Composition [US2013315998] | 2013-08-05 | 2013-11-28 |
| ANTIFUNGAL NAIL COAT AND METHOD OF USE [US2013323189] | 2013-08-09 | 2013-12-05 |
| TOPICAL FORMULATIONS, SYSTEMS AND METHODS [US2013337031] | 2013-03-08 | 2013-12-19 |

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....