Home » Promising clips
Category Archives: Promising clips
National award to Anthony Melvin Crasto for contribution to Pharma society from Times Network for Excellence in HEALTHCARE) | 5th July, 2018 | Taj Lands End, Mumbai, India
DR ANTHONY MEVIN CRASTO Conferred prestigious individual national award at function for contribution to Pharma society from Times Network, National Awards for Marketing Excellence ( For Excellence in HEALTHCARE) | 5th July, 2018 | Taj Lands End, Mumbai India
////////////National award, contribution to Pharma society, Times Network, Excellence in HEALTHCARE, 5th July, 2018, Taj Lands End, Mumbai, India, ANTHONY CRASTO
#hotpersoninawheelchair
#worlddrugtracker
Kiran Mazumdar Shaw Conferred with ‘The Global Leadership in Engineering 2016’ Award by USC


http://societyofwomenengineers.swe.org/awards/individual-awards/4153-global-leadership

The Global Leadership Award honors a person with at least fifteen (15) years professional experience who has worked in and led an internationally based engineering, scientific or technology-based business or organization, and in doing so, serves as a role model to women engineers and technologists worldwide. A maximum of three (3) awards may be presented annually.
“This award is a recognition of Biocon’s significant role in harnessing the potential of Biotechnology to provide affordable access to highly complex bio-pharmaceuticals like Insulins and monoclonal antibodies for the benefit of patients the world over.” – Kiran Mazumdar-Shaw

//////Kiran Mazumdar Shaw, ‘The Global Leadership in Engineering 2016’ , Award by USC
Indian Generics 2016
The generic APIs market is expected to continue to rise faster than the branded/innovative APIs, by 7.7%/year to reach $30.3 billion in 2016. Asia-Pacific is expected to show the fastest growth rates (10.8%/year). The 24 fastest growing markets will include 11 in Asia-Pacific, seven in Eastern Europe and CIS, four in Africa-Middle East and two in Latin America (Figure ).

Figure – Top growth markets for generic APIs to 2016
By 2016, China will account for 27.7% of the global generic API merchant market, while the US will have fallen to 23.8%; the mature markets as a whole will see their share fall from 41.8% in 2012 to 36.9%. India will be the third largest, with a 7.2% share.
सुकून उतना ही देना प्रभू, जितने से जिंदगी चल जाये।औकात बस इतनी देना,कि औरों का भला हो जाये।………..P.S. : The views expressed are my personal and in no-way suggest the views of the professional body or the company that I represent.
सुकून उतना ही देना प्रभू, जितने से जिंदगी चल जाये। औकात बस इतनी देना, कि औरों का भला हो जाये।
 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..FOR BLOG HOME CLICK HERE
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..FOR BLOG HOME CLICK HERE
Join me on Facebook FACEBOOK
 amcrasto@gmail.com
amcrasto@gmail.com
 LIONEL MY SON
LIONEL MY SON
जिंदगी चल जाये।
औकात बस इतनी देना,
कि औरों का भला हो जाये।
Practical Process Research and development; Development..Optimizing the Reaction by Minimizing Impurities


2000, Pages 165–184
Chapter 8 – Optimizing the Reaction by Minimizing Impurities
- Process Solutions L.L.C., Nicasio, California
The goals of process optimization change with the successful development of a project from early process research through scale-up into dedicated manufacturing. This general order of optimization may differ according to the nature of the process being considered; for instance, a process generating an inordinate amount of waste may be optimized to decrease waste before scaling up to the pilot plant. The initial goal of all process research and development is to maximize the amount of product generated under the reaction conditions. This is done by driving the reaction to completion, that is, by consuming any starting material that is charged in limiting amounts and by generating product with a minimal amount of by-products. Once the in-process yield has been optimized, the maximum yield of isolated product is expected. Rapid optimization is possible by judiciously changing solvents, reagents, catalysts, and ligands; investigations in this area allow the chemist considerable room for creativity and simplifying a process. Such changes may generate different impurities in the isolated intermediates, and it may be necessary to examine the tolerance of subsequent processes for the new impurities.
-
Chapter 8 – Optimizing the Reaction by Minimizing Impurities
-

Neal G. Anderson, PhD
For the past 17 years Neal has consulted to the pharmaceutical and fine chemical industries on chemical process research and development. He has presented short courses internationally on process R&D for “small molecules” to over 1400 participants from more than 160 companies. Keys to efficient scale-up are anticipating and then avoiding diffiulcties. Prior to consulting he worked at Bristol-Myers Squibb for 17 years. During that time, he had extensive hands-on experience with chemical process R&D in the lab, pilot plant, and manufacturing sites, including 12 manufacturing start-ups and process development for four major drugs. He is the author of Practical Process Research & Development(Academic Press; 2nd edition 2012).
Education & Training
- 1977-1979, post-doctoral studies, McNeil Laboratories
- 1977, Ph.D., Medicinal Chemistry, University of Michigan
- 1972, B.Sc., Honors Biology, University of Illinois
I consult to the pharmaceutical and fine chemical industries on developing and trouble-shooting processes to efficiently prepare drug substances and intermediates on large scale. Anticipating and avoiding problems are key for effective and efficient scale-up. For 17 years I have been consulting and presenting short courses internationally on process chemistry R & D for “small molecules” (over 1400 participants from more than 160 companies). Prior to consulting I worked at Bristol-Myers Squibb for 17 years. During that time I had extensive hands-on experience with chemical process development in the lab, pilot plant, and manufacturing sites, including 12 manufacturing start-ups and process development for four major drugs and many new drug candidates. I wrote Practical Process Research & Development (Academic Press, 2000; 2nd edition 2012).
Practical Process Research & Development describes the development of chemical processes for the pharmaceutical and fine chemicals industries. It provides a comprehensive, step-by-step approach to process R & D, and it is designed for those who want insights into generating rugged, practical, cost-effective processes. Guidelines for industrial process R & D are rarely taught in academia, although this book has been used as a textbook. It is primarily used by those in industry.
The second edition updates the first edition and includes topics not covered in the first edition , such as genotoxins, biocatalysis, green solvents, predicting effective solvent combinations, and process validation. Almost 85% of the references cited were published after the first edition was published, and virtually all examples in the Figures are new. Trevor Laird kindly wrote a forward for this edition.
, such as genotoxins, biocatalysis, green solvents, predicting effective solvent combinations, and process validation. Almost 85% of the references cited were published after the first edition was published, and virtually all examples in the Figures are new. Trevor Laird kindly wrote a forward for this edition.
The second edition has been translated into Japanese and graced with a handsome cover. Noriaki Murase was the translation supervisor, and the translators were Shohei Imachi, Koreaki Imura, Dai Tatsuta, Taro Tsukude, Toyoharu Numata, Yujiro Furuya, Akira Manaka, and Noriaki Murase. Sayaka Nukatsuka was the editor. I am very grateful to these people for their hard work to translate my book.
I am grateful to Barry Sharpless and Jerry Moniot for writing forwards to the first edition I am also grateful to the following people for their translations of the first edition of my book. Noriaki Murase, Yoshinori Murata, Toyoharu Numata, Mio Sakai, and Tatsuo Ueki translated Practical Process Research & Development into Japanese. Kwang-Hyun Ahn, Yeung-Ho Park, and Sung-Kwan Hwang translated Practical Process Research & Development into Korean. Zhinong Gao and Wenhao Hu translated Practical Process Research & Development into Chinese.
 free look
free look- Evaluate the existing synthesis and identify steps, or sequences in the route that may pose a problem for large scale synthesis
- Propose alternatives to any problematic steps or sequences and then implement these alternatives bases upon laboratory experimentation using Ph.D. level chemists with process research expertise
- Ensure the synthesis is suitable for the immediate needs of the project, which maybe for only a few kilograms of API
- Ensure the synthesis is suitable for long term, large scale manufacturing
- Optimize reagent charges, operating temperatures, concentrations, work-up conditions and volumes, and solvent use in general
- Identify which steps can be combined to result in a “through process” and implement the through process
- Optimize purification schemes by identifying key crystalline intermediates and remove chromatographies from the synthesis
- Optimize recrystallization parameters to ensure consistently high purity with similar impurity profiles from batch to batch, with low mother liquor losses
- Institute appropriate analytical controls for in-process assays, end of reaction specifications, and acceptable intermediate or API purity
- The process research team works closely with the analytical team to integrate the chemistry and analytical controls into the process at an early stage of the development cycle. The process research is then documented into a JACS style development report that outlines the chemistry and synthetic approaches that were tried as part of the synthetic development effort. This development report also includes a detailed experimental with supporting analytical data for the successful chemistry that results from our effort.The experimental that is part of these development reports is much more detailed than any journal publication. When coupled with our analytical and cGMP capabilities, the process research we provide is an essential groundwork for any compound that is just advancing from nomination at the discovery phase into clinical trial development. The process we develop provides the foundation of the ultimate manufacturing process, and should not need any changes (at a later date), to the synthetic strategy or bond forming steps used to prepare the API.

Critical Assessment of Pharmaceutical Processes, A Rationale for Changing the Synthetic Route
Critical Assessment of Pharmaceutical Processes A Rationale for Changing the Synthetic Route
A Rationale for Changing the Synthetic Route
Table of Contents
- 1. Introduction
- 2. Criteria for Process Assessment
-
- 2.1. Safety Issues2.1.1. Potential Safety Issues and Their Significance
- 2.1.2. Prediction and Assessment of Safety Issues
- 2.1.3. Options To Manage Safety Issues
- 2.1.4. Designing a Safer New Route
-
- 2.2. Environmental Issues
- 2.2.1. Potential Environmental Issues and Their Significance
- 2.2.2. Prediction and Assessment of Environmental Issues
- 2.2.3. Options To Manage Environmental Issues
- 2.2.4. Designing a New “Greener” Route
-
- 2.3. Legal Issues
- 2.3.1. Potential Legal Issues and Their Significance
- 2.3.2. Prediction and Assessment of Legal Issues Associated with Regulated Substances
- 2.3.3. Prediction and Assessment of Legal Issues Associated with Patent Infringement
- 2.3.4. Options To Manage Patent Issues
- 2.3.5. Designing a New Route with Freedom To Operate
-
- 2.4. Economic Issues
- 2.4.1. Potential Economic Issues and Their Significance
- 2.4.2. Prediction and Assessment of Economic Issues
- 2.4.3. Options To Manage Economic Issues
- 2.4.4. Designing a Cost-Effective New Route
-
- 2.5. Control Issues
- 2.5.1. Potential Control Issues and Their Significance
- 2.5.2. Prediction and Assessment of Control Issues
- 2.5.3. Options To Manage Control Issues
- 2.5.4. Designing a New Route with Adequate Control Measures
-
- 2.6. Throughput Issues
- 2.6.1. Potential Throughput Issues and Their Significance
- 2.6.2. Prediction and Assessment of Throughput Issues
- 2.6.3. Options To Manage Throughput Issues
- 2.6.4. Designing a New Route with High Throughput
- 3. Interrelationships between Process Issues
- 4. Conclusions
- 5. Acknowledgments
- 6. References
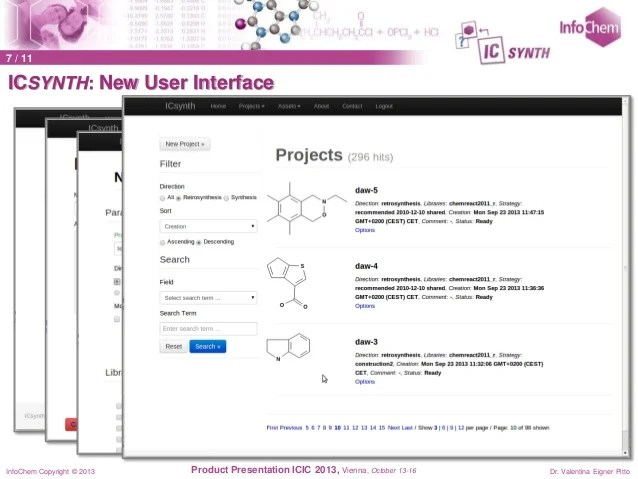
Route Design in the 21st Century: The ICSYNTH Software Tool as an Idea Generator for Synthesis Prediction

The new computer-aided synthesis design tool ICSYNTH has been evaluated by comparing its performance in predicting new ideas for route design to that of historical brainstorm results on a series of commercial pharmaceutical targets, as well as literature data. Examples of its output as an idea generator are described, and the conclusion is that it adds appreciable value to the performance of the professional drug research and development chemist team.



 InfoChem’s powerful synthesis planning tool now in Version 2.0. Read more …
InfoChem’s powerful synthesis planning tool now in Version 2.0. Read more …
 InfoChem will be represented at the forthcoming ACS Meeting in San Diego. You will find Dr. Josef Eiblmaier, Dr. Valentina Eigner Pitto, and Dr. Peter Loew …
InfoChem will be represented at the forthcoming ACS Meeting in San Diego. You will find Dr. Josef Eiblmaier, Dr. Valentina Eigner Pitto, and Dr. Peter Loew …
After inputting the target, users can select different synthetic strategies depending on requirements. ICSYNTH then automatically generates a multistep interactive synthesis tree – each node on the tree representing a precursor. The advantages are that the suggested reactions are based on, and linked to, published reactions (or their analogs) and the precursor availability is automatically checked in commercial catalogs. Users can modify the synthesis tree or select precursors for further analysis.
In addition to retro synthesis design, ICSYNTH has a forward reaction prediction module that offers reactivity mapping for the target molecule. Version 2.0 of ICSYNTH was launched in April 2014. The completely re-designed user interface (based on JavaScript) and major improvements in the algorithm responsible of the precursor search are the main enhancements of Version 2.0. In addition the forward reaction prediction algorithm has been optimized. Click here to see a complete version history.





INFOCHEM GESELLSCHAFT FÜR CHEMISCHE INFORMATION MBH
Landsberger Straße 408/V
D-81241 München
Germany
| Phone: | +49 (0)89 58 30 02 |
|---|---|
| Fax: | +49 (0)89 580 38 39 |
| Email: | info@infochem.de |



 Historische Bilder der Landsberger Straße – An der Trambahnhaltestelle Holzapfelstraße endet – Münchner Straßen – München – Süddeutsche.de
Historische Bilder der Landsberger Straße – An der Trambahnhaltestelle Holzapfelstraße endet – Münchner Straßen – München – Süddeutsche.de
Characterization of the “hygroscopic” properties of active pharmaceutical ingredients
Characterization of the “hygroscopic” properties of active pharmaceutical ingredients.
Source
SSCI, Inc., West Lafayette, IN, USA. ann.newman@aptuit.com
http://www.ncbi.nlm.nih.gov/pubmed/17630643
Abstract
The amount of water vapor taken up by an active pharmaceutical ingredient (API) as a function of relative humidity is routinely evaluated to characterize and monitor its “hygroscopicity” throughout the drug development process. In this minireview we address the necessity of going beyond the measurement of water vapor sorption isotherms to establish the various mechanisms by which solids interact with water and the important role played by the crystalline or amorphous form of the solid. Practical approaches for choosing experimental conditions under which water vapor sorption should be measured, including the pre-treatment of samples and the time allowed to reach an equilibrium state are presented. With the assistance of a flowchart, we provide a basis for the systematic examination of samples to establish the likely mechanisms of sorption and the indicators pointing toward future problems with physical and chemical instabilities. Finally, we present strategies for managing materials that might be susceptible to the detrimental effects of water vapor sorption.
(Copyright) 2008 Wiley-Liss, Inc.
OPRD PAPER-Streamlined Process for the Conversion of Artemisinin to Artemether

Correction to A Streamlined Process for the Conversion of Artemisinin to Artemether
 The structure for β-artemether is shown above, with the correct stereochemistry shown at the anomeric (8a) position. … Assignments are correct for the α- and β-anomers of artemether and dihydroartemisinin as discussed in the text; only the structure drawings are in error. …
The structure for β-artemether is shown above, with the correct stereochemistry shown at the anomeric (8a) position. … Assignments are correct for the α- and β-anomers of artemether and dihydroartemisinin as discussed in the text; only the structure drawings are in error. …The total synthesis of artemisinin from the Isopulegol ((-)-Isopulegol) began [JACS, 1983, 624].Contrast extracted from plants, is not an economical total synthesis method, but activity was found in the total synthesis of analogues are better practical significance of a thing. In this type of terpene total synthesis of natural products stereochemical conformation analysis is also very interesting. Hu menthol with MOMCl protected hydroxy, and get a double borohydride alcohol 1. Hydroboration Addition of anti-Markovnikov rule, which is replaced by hydrogen atoms added to the side of Quito, and the boron atoms added to the less substituted side. As the front side of the double bond MOM large steric hindrance, from the double rear borane adduct, resulting product1 . Compound 1 with a benzyl group protecting the primary alcohol, HCl removal of MOM protecting, PCC oxidation of the secondary alcohol to the ketone 3 . 3 with the hydrogen generating pull enolates LDA 4 , because of steric hindrance than hydrogen methyl, the nucleophilic reaction occurs in the torus , the form compound 5 . Ketone 5 and lithium reagent 6 an addition reaction, if one equivalent of lithium reagent, the resulting product was a 1:1 8 and 9 , if the 10-fold excess of lithium reagent, the resulting product was 8:1 8 and 9 . Lithium reagent 6 as a nucleophile large volume, its addition of cyclohexanone from the equatorial position to attack (such as an intermediate state 7 as shown), so that the generated key in an upright position hydroxyl group. Equivalent of lithium reagent no stereoselectivity of the reaction, but when a large excess of lithium, when chiral ketone 5 lithium reagent of the racemic 6 kinetic resolution becomes possible. Intermediate state 7 in, R configuration of the lithium reagent to Ketones speed is faster than its enantiomer S configuration lithium reagent. So generate eight faster than 9 , and finally get 8 and 9 of the ratio of 8:1. Lithium reagent 6, TMS air resistance maximum (A-value = 2.5 kcal / mol), OMe second air resistance (A-value = 0.75 kcal / mol), so that when the attack is downward TMS, OMe and H is determined by the relative position of cyclohexanone 2,6 substituent to the size and conformation of the decision, and should also be considered in the attack Burgi-Dunitz angle, so that the stereochemistry of the product unpredictable. Compound 8after removal of the benzyl protecting the primary alcohol with excess oxidized to carboxyl groups PCC automatically generate a macrolide 10 . 10 of the vinyl silane with m -CPBA and TFA into one11 , and then generate the enol methyl desilication TBAF ethers 12 , 12 and singlet oxygen reacts13 directly after treatment with acid artemisinin.


Promising clips-Ferulic Acid: a Natural Antioxidant Against Oxidative Stress Induced by Oligomeric A-beta on Sea Urchin Embryo.

Source
Istituto di Biomedicina ed Immunologia Molecolare (IBIM)-CNR, via Ugo La Malfa 153, 90146, Palermo, Italy.
Abstract
Alzheimer’s disease (AD) is a progressive, neurodegenerative disorder, characterized by loss of memory and impairment of multiple cognitive functions. Amyloid beta peptide (Aβ) is the main component of amyloid plaques observed in the brain of individuals affected by AD. Oxidative stress and mitochondrial dysfunction, induced by Aβ, are among the earliest events in AD, triggering neuronal degeneration and cell death. Use of natural molecules with antioxidant properties could be a suitable strategy for inhibiting the cell death cascade. Here, by employing the sea urchin Paracentrotus lividus as a model system, and Aβ oligomers, we tested the effectiveness of ferulic acid (FA), a natural antioxidant, as a putative AD neuroprotective compound. By microscopic inspection we observed that FA is able to reverse morphological defects induced by Aβ oligomers in P. lividus embryos. In addition, FA is able to neutralize reactive oxygen species (ROS), recover mitochondrial membrane potential, and block apoptotic pathways. Moreover, this model system has allowed us to obtain information about down- or up-regulation of some key molecules-Foxo3a, ERK, and p53-involved in the antioxidant mechanism
Ferulic acid is a hydroxycinnamic acid, a type of organic compound. It is an abundant phenolic phytochemical found in plant cell wall components such as arabinoxylans as covalent side chains. It is related to trans-cinnamic acid. As a component of lignin, ferulic acid is a precursor in the manufacture of other aromatic compounds. The etymology is from the genus Ferula, referring to the giant fennel (Ferula communis).






























 http://infochem.de/
http://infochem.de/


