Home » Antibiotics
Category Archives: Antibiotics
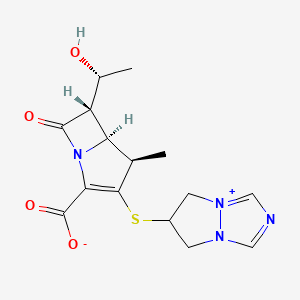
Sulopenem



Sulopenem
- 120788-07-0
- CP-70429
- 349.5 g/mol, C12H15NO5S3
- XX514BJ1XW
- PF-03709270
- PF03709270
(5R,6S)-6-[(1R)-1-hydroxyethyl]-7-oxo-3-[(1R,3S)-1-oxothiolan-3-yl]sulfanyl-4-thia-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid
- (5R,6S)-6-((1R)-1-HYDROXYETHYL)-7-OXO-3-(((1R,3S)-1-OXOTETRAHYDRO-1H-1.LAMBA.(SUP 4)-THIOPHEN-3-YL)SULFANYL)-4-THIA-1-AZABICYCLO(3.2.0)HEPT-2-ENE-2-CARBOXYLIC ACID
- (5R,6S)-6-((1R)-1-Hydroxyethyl)-7-oxo-3-(((3S)-tetrahydro-3-thienyl)thio)-4-thia-1-azabicyclo(3.2.0)hept-2-ene-2-carboxylic acid, (R)-S-oxide
- 4-THIA-1-AZABICYCLO(3.2.0)HEPT-2-ENE-2-CARBOXYLIC ACID, 6-((1R)-1-HYDROXYETHYL)-7-OXO-3-(((1R,3S)-TETRAHYDRO-1-OXIDO-3-THIENYL)THIO)-, (5R,6S)-
- 4-THIA-1-AZABICYCLO(3.2.0)HEPT-2-ENE-2-CARBOXYLIC ACID, 6-(1-HYDROXYETHYL)-7-OXO-3-((TETRAHYDRO-3-THIENYL)THIO)-, S-OXIDE, (5R-(3(1R*,3S*),5.ALPHA.,6.ALPHA.(R*)))-
FDA APPROVED sulopenem etzadroxil, probenecid, 10/25/2024, To treat uncomplicated urinary tract infections (uUTI)
Drug Trial Snapshot
Sulopenem (CP-70,429) is a thiopenem antibiotic derivative from the penem family, which unlike most related drugs is orally active. It was developed in Japan in the 1990s, and has been approved to treat uncomplicated urinary tract infections in combination with probenecid (Brand name: Orlynvah). It has reached Phase III clinical trials on several occasions and continues to be the subject of ongoing research into potential applications, especially in the treatment of multiple drug resistant urinary tract infections.[1][2][3][4][5]
In October 2024, the US Food and Drug Administration approved sulopenem etzadroxil with probenecid combination for the treatment of urinary tract infections caused by Escherichia coli, Klebsiella pneumoniae, or Proteus mirabilis in adult women with limited alternative oral antibiotic options. The combination was developed by Iterum Therapeutics under the trade name ORLYNVAH™.[6]


JP 1995278137; US 5013729; WO 8808845, J Org Chem 1992,57(16),4352-61
1) The reaction of L-aspartic acid (I) with NaNO2, NaBr and H2SO4 gives 2(S)-bromosuccinic acid (II), which is reduced with methyl sulfide borane complex in THF, yielding 2(S)-bromobutane-1,4-diol (III). The cyclization of (III) with Cs2CO3 in methylene chloride affords (R)-(2-hydroxyethyl)oxirane (IV), which is acylated with methanesulfonyl chloride to the corresponding mesylate (V). The cyclization of (V) with Na2S in acetonitrile/water gives 3(R)-hydroxythiolane (VI), which is acylated with p-toluenesulfonyl chloride, affording the corresponding tosylate (VII). The controlled oxidation of (VII) with potassium peroxymonosulfate (oxone) gives 3(R)-(p-toluenesulfonyloxy)thiolane-1(R)-oxide (VIII), which by reaction with potassium thioacetate in acetone is converted to 3(S)-(acetylthio)thiolane 1(R)-oxide (IX). The reaction of (IX) with NaOEt and CS2 in ethanol yields the trithiocarbonate (X), which is condensed with the chloroazetidinone (XI), yielding the trithiocarbonate ester (XII). The condensation of (XII) with 2-chloroallyloxalyl fluoride (XIII) by means of diisopropylethylamine in methylene chloride affords the substituted oxalamic ester (XIV), which is cyclized by means of triethyl phosphite in refluxing chloroform to the fully protected penem derivative (XV). The reaction of (XV) with tetrabutylammonium fluoride (TBAF) in THF eliminates the protecting tert-butyldimethylsilyl group, yielding the chloroallyl ester (XVI), which is treated with triphenylphosphine and sodium 2-ethylhexanoate in dichloromethane to obtain the corresponding sodium salt (XVII). Finally, this compound is treated with HCl in cool water.

US 4921972
2) The intermediate 3(R)-(p-toluenesulfonyloxy)thiolane (VII) can be obtained by two other synthetic pathways: a) The racemic 2-hydroxy-4-(methylsulfanyl)butyric acid ethyl ester (XVIII) is submitted to optical resolution with Pseudomonas fluorescens lipase in toluene/water, yielding the corresponding 2(R)-hydroxy ester (XIX), which is reduced with NaBH4 in THF/water to afford 4-(methylsulfanyl)butane-1,2(R)-diol (XX). The acylation of (XX) with p-toluenesulfonyl chloride and pyridine yields the ditosylate (XXI), which is cyclized in refluxing benzene to give 1(R)-methyl-3(R)-(p-toluenesulfonyloxy)thiolanium p-toluenesulfonate (XXII). Finally, this compound is treated with trifluoroacetic acid in pyridine to afford the thiolane (VII), already described. b) The reduction of 4-chloro-3(R)-hydroxybutyric acid methyl ester (XXIII) with lithium borohydride in THF gives 4-chlorobutane-1,3(R)-diol (XXIV), which is tosylated as before, yielding the bis(tosyloxy) derivative (XXV). Finally, this compound is cyclized with Na2S in hot acetonitrile/water to afford the thiolane (VII), already described.

https://pubsapp.acs.org/cen/coverstory/88/8836cover.html
References
- ^ Minamimura M, Taniyama Y, Inoue E, Mitsuhashi S (July 1993). “In vitro antibacterial activity and beta-lactamase stability of CP-70,429 a new penem antibiotic”. Antimicrobial Agents and Chemotherapy. 37 (7): 1547–1551. doi:10.1128/AAC.37.7.1547. PMC 188011. PMID 8363389.
- ^ Hamilton-Miller JM (November 2003). “Chemical and microbiologic aspects of penems, a distinct class of beta-lactams: focus on faropenem”. Pharmacotherapy. 23 (11): 1497–1507. doi:10.1592/phco.23.14.1497.31937. PMID 14620395. S2CID 43705118.
- ^ Ednie LM, Appelbaum PC (May 2009). “Antianaerobic activity of sulopenem compared to six other agents”. Antimicrobial Agents and Chemotherapy. 53 (5): 2163–2170. doi:10.1128/AAC.01557-08. PMC 2681565. PMID 19223615.
- ^ Bader MS, Loeb M, Leto D, Brooks AA (April 2020). “Treatment of urinary tract infections in the era of antimicrobial resistance and new antimicrobial agents”. Postgraduate Medicine. 132 (3): 234–250. doi:10.1080/00325481.2019.1680052. PMID 31608743. S2CID 204545734.
- ^ Veeraraghavan B, Bakthavatchalam YD, Sahni RD (December 2021). “Oral Antibiotics in Clinical Development for Community-Acquired Urinary Tract Infections”. Infectious Diseases and Therapy. 10 (4): 1815–1835. doi:10.1007/s40121-021-00509-4. PMC 8572892. PMID 34357517.
- ^ “Iterum Therapeutics Receives U.S. FDA Approval of ORLYNVAH™ (Oral Sulopenem) for the Treatment of Uncomplicated Urinary Tract Infections”. Iterum Therapeutics plc. 2024-10-25. Retrieved 2024-10-25.
| Clinical data | |
|---|---|
| ATC code | None |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 120788-07-0 |
| PubChem CID | 9950244 |
| DrugBank | DB15284 |
| ChemSpider | 8125855 |
| UNII | XX514BJ1XW |
| KEGG | D05969 |
| CompTox Dashboard (EPA) | DTXSID20869656 |
| Chemical and physical data | |
| Formula | C12H15NO5S3 |
| Molar mass | 349.43 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI | |
FDA Approved Drug Products: Orlynvah (sulopenem etzadroxil and probenecid) tablets for oral use (October 2024) [Link]
FDA News Release: FDA approves new treatment for uncomplicated urinary tract infections in adult women who have limited or no alternative oral antibiotic treatment options [Link]
//////Sulopenem, Orlynvah, FDA 2024, APPROVALS 2024, CP-70,429, 120788-07-0, CP-70429, XX514BJ1XW, PF-03709270, PF03709270
#Sulopenem, #Orlynvah, #FDA 2024, #APPROVALS 2024, #CP-70,429, #120788-07-0, #CP-70429, #XX514BJ1XW, #PF-03709270, #PF03709270
CEFOPERAZONE
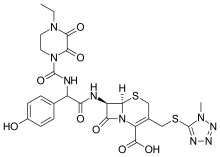
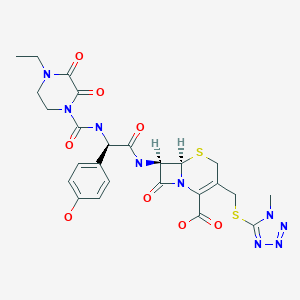
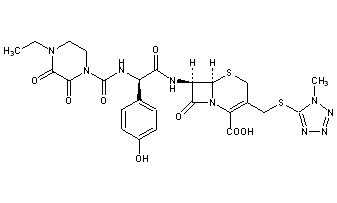
Cefoperazone
- Molecular FormulaC25H27N9O8S2
- Average mass645.667 Da
Product Ingredients
| INGREDIENT | UNII | CAS | INCHI KEY |
|---|---|---|---|
| Cefoperazone sodium | 5FQG9774WD | 62893-20-3 | NCFTXMQPRQZFMZ-WERGMSTESA-M |
(6R,7R)-7-{[(2R)-2-{[(4-ethyl-2,3-dioxopiperazin-1-yl)carbonyl]amino}-2-(4-hydroxyphenyl)acetyl]amino}-3-{[(1-methyl-1H-tetrazol-5-yl)thio]methyl}-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid
(6R,7R)-7-[(2R)-2-[(4-ethyl-2,3-dioxopiperazine-1-carbonyl)amino]-2-(4-hydroxyphenyl)acetamido]-3-{[(1-methyl-1H-1,2,3,4-tetrazol-5-yl)sulfanyl]methyl}-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid
263-749-4[EINECS], 4742
5-Thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid, 7-[[(2R)-2-[[(4-ethyl-2,3-dioxo-1-piperazinyl)carbonyl]amino]-2-(4-hydroxyphenyl)acetyl]amino]-3-[[(1-methyl-1H-tetrazol-5-yl)thio]methyl]-8-oxo- , (6R,7R)- [ACD/Index Name]
5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid, 7-[[(2R)-2-[[(4-ethyl-2,3-dioxo-1-piperazinyl)carbonyl]amino]-2-(4-hydroxyphenyl)acetyl]amino]-3-[[(1-methyl-1H-tetrazol-5-yl)thio]methyl]-8-oxo-, (6R,7R)-
62893-19-0[RN]
7-[D-(-)-a-(4-Ethyl-2,3-dioxo-1-piperazinecarboxamido)-a-(4-hydroxyphenyl)acetamido]-3-[[(1-methyl-1H-tetrazol-5-yl)thio]methyl]-3-cephem-4-carboxylic Acid
7U75I1278D
Experimental Properties
| PROPERTY | VALUE | SOURCE |
|---|---|---|
| melting point (°C) | 188-190 | Saikawa, I., Takano, S., Yoshida, C., Takashima, 0..Momonoi, K., Kuroda, S., Komatsu, M., Yasuda, T.and Kodama, Y.; British Patent 1,508,071; April 19,1978; assigned to Toyama Chemical Co., Ltd. and U.S. Patent 4,110,327; August 29,1978; also assigned to Toyama Chemical Co., Ltd. |
| logP | -0.74 | HANSCH,C ET AL. (1995) |
Cefoperazone
CAS Registry Number: 62893-19-0
CAS Name: (6R,7R)-7-[[(2R)-[[(4-Ethyl-2,3-dioxo-1-piperazinyl)carbonyl]amino](4-hydroxyphenyl)acetyl]amino]-3-[[(1-methyl-1H-tetrazol-5-yl)thio]methyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acidAdditional Names: 7-[D-(-)-a-(4-ethyl-2,3-dioxo-1-piperazinecarboxamido)-a-(4-hydroxyphenyl)acetamido]-3-[[(1-methyl-1H-tetrazol-5-yl)thio]methyl]-3-cephem-4-carboxylic acid
Molecular Formula: C25H27N9O8S2
Molecular Weight: 645.67
Percent Composition: C 46.50%, H 4.21%, N 19.52%, O 19.82%, S 9.93%
Literature References: Broad spectrum third generation cephalosporin antibiotic. Prepn: I. Saikawa et al.,BE837682; eidem,US4410522 (1976, 1983 both to Toyama); eidem,Yakugaku Zasshi99, 929 (1979). Stability in aq soln: eidem,ibid. 1207. In vitro activity: M. V. Borobio et al.,Antimicrob. Agents Chemother.17, 129 (1980). Kinetics in rats: J. Fabre et al.,Schweiz. Med. Wochenschr.110, 264 (1980); in humans: A. F. Allaz, ibid.109, 1999 (1979). Review of pharmacology and therapeutic efficacy: R. N. Brogden et al.,Drugs22, 423-460 (1981). Symposium on clinical studies: ibid. Suppl. 1, 1-124.
Properties: Crystals from acetonitrile/water, mp 169-171° (hydrated). Stable at pH 4.0-7.0; slightly unstable in acid; highly unstable in alkaline soln.
Melting point: mp 169-171° (hydrated)
Derivative Type: Sodium salt
CAS Registry Number: 62893-20-3
Manufacturers’ Codes: CP-52640-2; T-1551
Trademarks: Bioperazone (Biopharma); Cefazone (Firma); Cefobid (Pfizer); Cefobine (Pfizer); Cefobis (Pfizer); Cefogram (Metapharma); Cefoneg (Tosi); Cefosint (Proter); Dardum (Lisapharma); Farecef (Lafare); Kefazon (Esseti); Novobiocyl (Francia); Pathozone (Pfizer); Peracef (Pfizer); Perocef (Pulitzer); Tomabef (Aandersen)
Molecular Formula: C25H26N9NaO8S2
Molecular Weight: 667.65
Percent Composition: C 44.97%, H 3.93%, N 18.88%, Na 3.44%, O 19.17%, S 9.61%
Therap-Cat: Antibacterial., Therap-Cat-Vet: Antibacterial.
Keywords: Antibacterial (Antibiotics); ?Lactams; Cephalosporins.
Cefoperazone is a third-generation cephalosporin antibiotic, marketed by Pfizer under the name Cefobid. It is one of few cephalosporin antibiotics effective in treating Pseudomonas bacterial infections which are otherwise resistant to these antibiotics.
It was patented in 1974 and approved for medical use in 1981.[1] Cefoperazone/sulbactam (Sulperazon) is a co-formulation with sulbactam.
Cefoperazone is a broad-spectrum cephalosporin antibiotic used for the treatment of bacterial infections in various locations, including the respiratory tract, abdomen, skin, and female genital tracts.
Cefoperazone is a semisynthetic broad-spectrum cephalosporin proposed to be effective against Pseudomonas infections. It is a third-generation antiobiotic agent and it is used in the treatment of various bacterial infections caused by susceptible organisms in the body, including respiratory tract infections, peritonitis, skin infections, endometritis, and bacterial septicemia. While its clinical use has been discontinued in the U.S., cefoperazone is available in several European countries most commonly under the product name, Sulperazon.

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////////////////////////////
SYN

English: I. Saikawa, S. Takano, Y. Shuntaro, C. Yoshida, 0.
Takashima, K. Momonoi, S. Kuroda, M. Komatsu, T. Yasuda, and Y. Kodama, German Offen., DE 2,600,880 (1977); Chem.
Abstr., 87_, 184533b (1977).
SYN
Following is one of the synthesis routes:
alpha-(4-Ethyl-2,3-dioxo-1-piperazinocarbonylamino)-p-hydroxyphenylacetic acid (I) is condensed with 7-amino-3-[(1-methyl-1H-tetrazol-5-yl)thiomethyl]-3-cephem-4-carboxylic acid (II) in the presence of ethyl chlorocarbonate and N,O-bis(trimethylsilyl)acetamide in acetonitrile to produce Cefoperazone sodium.

SYN
Antibiotics
R.S. Vardanyan, V.J. Hruby, in Synthesis of Essential Drugs, 2006
Cefoperazone
Cefoperazone, (6R,7R)-7-[(R)-2-(4-ethyl-2,3-dioxo-1-piperazincarboxamido)-2-(p-hydroxyphenyl)acetamido]-3-[[(1-methyl-1 H-tetrazol-5-yl)thio]methyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-en-2-carboxylic acid (32.1.2.84), is synthesized by acylating 7-amino-3-(1-methyl-1,2,3,4-tetrazol-5-yl)-thiomethyl-3-cefem-4-carboxylic acid (32.1.2.24) with a mixed anhydride synthesized from ethyl chloroformate and α-(4-ethylpiperazin-2, 3-dion-1-carbonylamino)-4-hydroxyphenylacetic acid (32.1.2.83), which in turn is synthesized from 4-ethylpiperazin-2,3-dion-1-carboxylic acid (32.1.1.29) and the sodium salt of 4-hydroxyphenylglycine [163–168].


Cefoperazone also has a broad spectrum of antimicrobial action, including most clinically significant microorganisms: Gram-positive, Gram-negative, aerobic, and anaerobic. It is stable with respect to most beta-lactamases of Gram-positive and Gram-negative bacteria.
Cefoperazone is used for bacterial infections of the lower respiratory tract, urinary and sexual tracts, bones, joints, skin, soft tissues, abdominal, and gynecological infections. Synonyms of this drug are cefazon, cefobid, cefobis, and many others.
Spectrum of bacterial susceptibility
Cefoperazone has a broad spectrum of activity and has been used to target bacteria responsible for causing infections of the respiratory and urinary tract, skin, and the female genital tract. The following represents MIC susceptibility data for a few medically significant microorganisms.
- Haemophilus influenzae: 0.12 – 0.25 µg/ml
- Staphylococcus aureus: 0.125 – 32 µg/ml
- Streptococcus pneumoniae: ≤0.007 – 1 µg/ml[2]
Adverse effects
Cefoperazone contains an N-methylthiotetrazole (NMTT or 1-MTT) side chain. As the antibiotic is broken down in the body, it releases free NMTT, which can cause hypoprothrombinemia (likely due to inhibition of the enzyme vitamin K epoxide reductase) and a reaction with ethanol similar to that produced by disulfiram (Antabuse), due to inhibition of aldehyde dehydrogenase.[3]
Mechanism of action
Cefoperazone exerts its bactericidal effect by inhibiting the bacterial cell wall synthesis, and sulbactam acts as a beta-lactamase inhibitor, to increase the antibacterial activity of cefoperazone against beta-lactamase-producing organisms.
References
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 494. ISBN 9783527607495.
- ^ “Cefoperazone (Cefobid) – The Antimicrobial Index Knowledgebase – TOKU-E”. antibiotics.toku-e.com.
- ^ Stork CM (2006). “Antibiotics, antifungals, and antivirals”. In Nelson LH, Flomenbaum N, Goldfrank LR, Hoffman RL, Howland MD, Lewin NA (eds.). Goldfrank’s toxicologic emergencies. New York: McGraw-Hill. p. 847. ISBN 0-07-143763-0. Retrieved 2009-07-03.
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| MedlinePlus | a601206 |
| ATC code | J01DD12 (WHO) QJ51DD12 (WHO) |
| Pharmacokinetic data | |
| Excretion | Hepatic |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 62893-19-0 |
| PubChem CID | 44185 |
| DrugBank | DB01329 |
| ChemSpider | 40206 |
| UNII | 7U75I1278D |
| KEGG | D07645 |
| ChEMBL | ChEMBL507674 |
| CompTox Dashboard (EPA) | DTXSID2022759 |
| ECHA InfoCard | 100.057.936 |
| Chemical and physical data | |
| Formula | C25H27N9O8S2 |
| Molar mass | 645.67 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI | |
| (verify) |
//////////cefoperazone, Antibacterial, Antibiotics, Lactams, Cephalosporins, CP-52640-2, T-1551, CP 52640-2, T 1551
[H][C@]12SCC(CSC3=NN=NN3C)=C(N1C(=O)[C@@]2([H])NC(=O)[C@H](NC(=O)N1CCN(CC)C(=O)C1=O)C1=CC=C(O)C=C1)C(O)=O

NEWDRUG APPROVALS
one time
$10.00
MEROPENEM
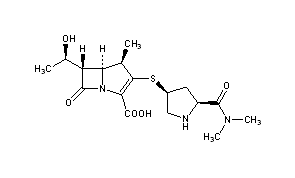
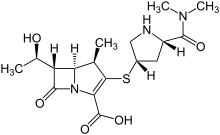
Meropenem
CAS number96036-03-2
IUPAC Name(4R,5S,6S)-3-{[(3S,5S)-5-(dimethylcarbamoyl)pyrrolidin-3-yl]sulfanyl}-6-[(1R)-1-hydroxyethyl]-4-methyl-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid
WeightAverage: 383.463
Monoisotopic: 383.151491615
Chemical FormulaC17H25N3O5S
- Antibiotic SM 7338
- ICI 194660
- SM 7338
CAS Registry Number: 96036-03-2
CAS Name: (4R,5S,6S)-3-[[(3S,5S)-5-[(Dimethylamino)carbonyl]-3-pyrrolidinyl]thio]-6-[(1R)-1-hydroxyethyl]-4-methyl-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid
Additional Names: (1R,5S,6S)-2-[(3S,5S)-5-(dimethylaminocarbonyl)pyrrolidin-3-ylthio]-6-[(R)-1-hydroxyethyl]-1-methylcarbapen-2-em-3-carboxylic acid
Molecular Formula: C17H25N3O5S
Molecular Weight: 383.46
Percent Composition: C 53.25%, H 6.57%, N 10.96%, O 20.86%, S 8.36%
Literature References: Carbapenem antibiotic. Prepn: M. Sunagawa et al.,EP126587; M. Sunagawa, US4943569 (1984, 1990 both to Sumitomo).
Structure-activity study: M. Sunagawa et al.,J. Antibiot.43, 519 (1990).Crystal structure: K. Yanagi et al.,Acta Crystallogr.C48, 1737 (1992).HPLC determn in serum and bronchial secretions: M. Ehrlich et al., J. Chromatogr. B751, 357 (2001). Pharmacokinetics: R. Wise et al.,Antimicrob. Agents Chemother.34, 1515 (1990).Series of articles on antimicrobial activity, metabolism: J. Antimicrob. Chemother.24, Suppl. A, 1-320 (1989); and clinical performance: ibid.36, Suppl. A, 1-223 (1995).Review of clinical experience in intensive care: M. Hurst, H. M. Lamb, Drugs59, 653-680 (2000).
Derivative Type: Trihydrate
CAS Registry Number: 119478-56-7
Manufacturers’ Codes: ICI-194660; SM-7338
Trademarks: Meronem (AstraZeneca); Meropen (Sumitomo); Merrem (AstraZeneca)
Properties: White to pale yellow crystalline powder. Sparingly sol in water; very slightly sol in hydrated ethanol. Practically insol in acetone, ether.
Therap-Cat: Antibacterial.
Keywords: Antibacterial (Antibiotics); ?Lactams; Carbapenems.
Product Ingredients
| INGREDIENT | UNII | CAS | INCHI KEY |
|---|---|---|---|
| Meropenem sodium | Not Available | 211238-34-5 | UBQRNADYCUXRBD-NACOAMSHSA-N |
| Meropenem trihydrate | FV9J3JU8B1 | 119478-56-7 | CTUAQTBUVLKNDJ-OBZXMJSBSA-N |
International/Other BrandsAronem (ACI) / Aropen (Aristopharma) / Carbanem (Sanofi-Aventis) / Erope (Lincoln) / Fulspec (Acme) / I-penam (Incepta) / Merenz (Admac) / Merofit (FHC) / Meronem (AstraZeneca) / Meronis (Neiss) / Meropen (Swiss Parenterals) / Merotec (Zuventus) / Merrem I.V. (AstraZeneca) / Monan (AstraZeneca) / Ropenem (Drug International) / Zeropenem (Sanofi-Aventis)
Synthesis Reference
Yoon Seok Song, Sung Woo Park, Yeon Jung Yoon, Hee Kyoon Yoon, Seong Cheol Moon, Byung Goo Lee, Soo Jin Choi, Sun Ah Jun, “METHOD FOR PREPARING MEROPENEM USING ZINC POWDER.” U.S. Patent US20120065392, issued March 15, 2012.
SYN
Carbapenem antibiotic. Prepn: M. Sunagawa et al., EP 126587; M. Sunagawa, US 4943569 (1984, 1990 both to Sumitomo). Structure-activity study: M. Sunagawa et al., J. Antibiot. 43, 519 (1990).

SYN
https://patents.google.com/patent/WO2012062035A1/enCarbapenem, a type of β-lactam antibiotic, is known for its broad spectrum of antibacterial activity and strong antibacterial activity, such as meropenem (Me r0 p e nem), imine South (Imipenem) and Biabenem, etc., play an important role in the cure of severe infections.

Meropenem Imipenem For the synthetic methods of the Peinan type, the previous studies have mainly synthesized the corresponding Peinan side chain compound and the parent nucleus MAP, respectively, and then condensed and removed the protecting group to obtain the Peinan product. Such as US patentsUSP4933333, starting from 4-acetoxyazetidinone (4AA), obtained a matrix MAP after several steps of reaction. The mother nucleus is then condensed and deprotected from the side chain to obtain meropenem. However, this method is cumbersome, the synthesis step is long, and the total yield is low, and the noble metal catalyst is inevitably used in the synthesis of the compound (9).

MAP (10) Meropenem The Chinese invention patent document CN200810142137.5 has introduced a method for synthesizing meropenem.

(XII) (I)(TBD S = Si (CH 3 ) 2 C (CH 3) 3; PNB = p-N0 2 -C 6 H 4 CH 2; PNZ = 2 -C 6 H 4 CH 2 OCO N0 p-) This method of Scheme Short, easy to operate, easy to get raw materials, but there are some areas for improvement.

Example 11) (3R, 4S)-3-[(R)-l-(tert-butyldimethylsilyloxy)ethyl]-4-[(2,S, 4’R)- 1- (allyl Synthesis of oxycarbonylxiaodimethylaminocarbonylpyrrolidinothio]-2-azetidinone (II) In a 500 ml reaction flask, add 22.6 g (0.075 mol) of (3S,4S)-3-[( R) l-(tert-Butyldimethylsilyloxy)ethyl]-4-[(R)-1-carbonylethyl]-2-azetidinone (IV), 17.1 g (0.083 mol) Dicyclohexylcarbodiimide (DCC) in 100 ml of acetone and 0.76 g of 4-dimethylaminopyridine (DMAP), 20.3 g (0.078 mol) of (2S, 4R)-2-dimethylamine was added dropwise with stirring. A solution of carbonyl-4-mercapto (i-propoxycarbonyl)pyrrolidine (V) in 125 ml of acetone was reacted at room temperature for 14 hours. Filtration, collecting the filtrate, concentrating, adding 200 ml of toluene thereto, using 200 ml of a 5 % acetic acid solution, 200 ml of a saturated sodium hydrogencarbonate solution and 150 ml of saturation Washed with brine, dried over anhydrous magnesium sulfate and evaporated to dryness <mjjjjjjjjjjjjjjjjjjjjjjjjjjjjjjjjjjjjj 4-[(2,8, 4, ) small (propoxycarbonyl dimethyl dimethylaminocarbonyl)pyrrolidinyl]-2-azetidinone (II), directly without further treatment Invest in the next step.1H-NMR (400 MHz, CDC 13): </ RTI> <RTIgt; m), 2.816-2.849 (lH, s), 2.935-2.953 (3H, m), 3.027-079 (3H, d), 3.378-3.401 (lH, m), 3.792-3.796 (1H, d), 3.807- 3.953 (lH, m), 4.042-4.160 (3H, m), 4.492-4.570 (2H, m), 4.670-4.739 (lH, m), 5.164-5.295 (1H, m), 5.807-5.921 (lH, m ), 6.214(1H, s). Example 22) (31,48)-3-[(1 )-1-(tert-butyldimethylsilyloxy)ethyl]-4-[(2,8,4,1 )- 1- (allyl Synthesis of oxycarbonyl-1-dimethylaminocarbonylpyrrolidinothio]-1-(zincpropoxyl)-2-azetidinone (III) In a 1000 ml reaction flask, add 34.8 g (0.064) Mol) (3R, 4S)-3-[(R)-l-(tert-butyldimethylsilyloxy)ethyl]-4-[(2,S, 4,R)-1-(allyl Oxycarbonyl-1-pyrimidinylcarbonyl)pyrrolidinylthio]-2-azetidinone (11), 15.0 ml of triethylamine and 350 ml of toluene, control temperature below -10 °C, add 18.9 g (0.128 mol) p-nitrobenzyl chloroacetate (VI), heated to 0 ° C (-20 ° 5 ° C can be) reaction l ~ 3h. Then slowly add 250 ml of ice water and stir for 10 min. The layers were static and the organic phase was washed three times with saturated sodium bicarbonate solution, 200 ml each time. Dry over anhydrous magnesium sulfate, filtered, and evaporated to dryness to give white crystals, 4,7g (0.0622mol, yield 97.3%) (3R, 4S)-3-[(R) small (tert-butyldimethylsilyloxy)ethyl ]-4-[(2,S, 4,R)-1-(allyloxycarbonyldimethyldimethylaminocarbonyl)pyrrolidinylsulfur]sodium (sweetoxypropanoyl)-2-azetidinone (III), the product was directly put into the next step without further purification.Mp: 33-34 °C1H-NMR (300 MHz, CDC 13):0.819(9H, s), 1.167(3H, d), 1.188(4H, d), 1.693(5H, s), 1.850-1.926(1H, m), 2.631-2.700(1H, m), 2.941-2.960( 3H,d), 3.029-3.080(3H,d), 3.357-3.433(lH, m), 3.506-3.545(2H, m), 3.918-3.968(1H, m), 4.054-4.123 (2H, m), 4.270-4.291(lH, m), 4.391(lH,s), 4.518-4.568(2H, m), 4.588-4.779(3H, m), 5.178-5.416(3H, m), 5.861-5.982(2H,m ). Example 33) (5R,6S,8R,2’S, 4,S)-[(R)-1-(tert-butyldimethylsilyloxy)ethyl]-3-[4-(1-allyloxycarbonyl) -1- dimethylaminocarbonylpyrrolidinothio]-6-(1-allyloxycarbonylethoxy)-1-azabicyclo[3.2.0]-hept-2-en-7-one- Synthesis of 2-carboxylate In a 500 ml reaction flask, 40; 7 g (0.0622 mol) of (3R, 4S)-3-[(R)-l-(tert-butyldimethylsilyloxy) was added. Ethyl]-4-[(2,S,4,R)-1-(indolyloxycarbonyl-1-dimethylaminocarbonyl)pyrrolidinylsulfate]small (sweetoxypropanoyl)-2-nitrogen Heterocyclic butanone (III) and 150 ml of toluene, 22 ml of trimethyl phosphite (furrowing lg of hydroquinone) were added under nitrogen. After reacting at 60 ° C for 16 hours, the solvent was evaporated under reduced pressure. It was recrystallized by adding 300 ml of ethyl acetate, and the solid was collected, and vacuum-dried at 40 ° C to obtain 32.8 g (0.0528 mol, yield: 85.0%) (5R, 6S, 8R, 2’S, 4,S)-[(R)- 1-(tert-Butyldimethylsilyloxy)ethyl]-3-[4-(1-allyloxycarbonyl-1-dimethylaminocarbonyl)pyrrolidinyl] -6-(1-ene Propoxycarbonyl ethoxy) small azabicyclo[3.2.0]-hept-2-en-7-one-2-carboxylate (oxime).1H-NMR (300 MHz, CDC 13):0.82(9H, s), 1.24(6H, d), 1.26(3H, s), 1.36(3H, s), 1.94(1H, m), 2.69(1 H, m), 2.97-3.11(6H, m ), 3.15-3.74(4H, m), 4.35(2H,m), 4.37-4.67(5H, m), 5.24-5.28(4H, m), 5.84(1H, m). Example 44) (5R, 6S, 8R, 2, S, 4’S)-[(R)小(hydroxy)ethyl]-3-[4-(1-allyloxycarbonylsuccinylcarbonyl)pyrrolidinyl Synthesis of thio]-6-(1-allyloxycarbonylethoxy)-1-azabicyclo[3.2.0]-hept-2-en-7-one-2-carboxylate (Vffl) at room temperature , in a 2000ml reaction flask, add 32.8g (0.0528mol) (5R,6S,8R,2’S,4,S)-[(R)-1-(tert-butyldimethylsilyloxy)ethyl] 3-[4-(1-allyloxycarbonyl-1-dimethylaminocarbonyl)pyrrolidinyl]-6-(1-indolyloxycarbonylethoxy)-1-azabicyclo[3.2.0 -Hept-2-ene-7-one-2-carboxylate (W), 27.4 ml of acetic acid, 41.3 g of fluorohydrogenamine and 1000 ml of dichloromethane, stirred at room temperature for 48 h. After completion of the reaction, 500 ml of a saturated aqueous solution of sodium hydrogencarbonate was added to the reaction mixture, and the mixture was stirred for 10 minutes, and the methylene chloride layer was separated and dried over anhydrous magnesium sulfate to give a white solid (26.2 g (0.0517 mol, yield 98.0). %) (5R, 6S, 8R, 2’S, 4’S)-[(R)小(hydroxy)ethyl]-3-[4-(1-allyloxycarbonylsuccinylcarbonyl)pyr Rhodium thio] -6-(l-allyloxycarbonylethoxy)-1-azabicyclo[3. 2. 0]-hept-2-en-7-one-2-carboxylate (ring The product was directly charged to the next step without further purification.1H-NMR (300 MHz, CDC 13):1.26(3H, s), 1.36(3H, s), 1.94(1H, m), 2.67(1H, m), 2.97-3.11(6H, m), 3.2-3.7(4H, m) ; 4.25(2H, m), 4.47-4.87 (5H, m), 5.15-5.50 (4H, m), 5.94 (2H, m). Example 55) (5R,6S,8R,2,S,4,S)-3-[4-dimethylaminocarbonyl)pyrrolidinyl]-6-(l-hydroxyethyl)-1-aza Synthesis of bicyclo[3.2.0]-hept-2-en-7-one-2-carboxylate (I) To the reaction flask, 26.2 g (0.0517 mol) (5R, 6S, 8R, 2’S, 4’S) was added. – [(R)-l-(hydroxy)ethyl]-3-[4-(1-allyloxycarbonyl-1-dimethylaminocarbonyl)pyrrolidinyl] -6-(1-allyloxy Carbonyl ethoxy)-1-azabicyclo[3. 2. 0]-hept-2-en-7-one-2-carboxylate (VDI), 21.3 g (0.152 mol) dimethylcyclohexane The ketone and 550 ml of ethyl acetate were heated to 30 ° C, and a solution of 1.0 g (0.865 mmol) of tetratriphenylphosphine palladium in 150 ml of dichloromethane was added dropwise thereto, and the mixture was reacted at room temperature for 3 h under nitrogen atmosphere. After adding 300 ml of water to the reaction mixture, the aqueous layer was separated, the aqueous layer was washed with ethyl acetate, and then, 500 ml of tetrahydrofuran was added dropwise with stirring in an ice bath, and the crystals were stirred, and the crystals were collected and dried in vacuo to give pale yellow crystals of 13.4 g (0.0352 md, Yield 68.1%) (5R,6S,8R,2,S,4,S)-3-[4-(2-dimethylaminocarbonyl)pyrrolidinylthio]-6-(1-hydroxyethyl) 1-Azabicyclo[3.2.0]-hept-2-en-7-one-2-carboxylic acid trihydrate (I)-Meropectin.IR max KBr cm- 1 : 1755, 1627, 1393, 1252, 1130NMR (D20, 300Hz): 1.25 (3H, d), 1.81-1.96 (1H, m), 2.96 (3H, s), 3.03 (3H, s), 3.14-3.20 (3H, m), 3.31-3.41 (2H, m), 3.62- 3.72 (1H, m), 3.90-4.00 (1H, m), 4.14-4.26 (2H, m), 4.63 (1H, t). Example 6 6) (5R,6S,8R,2’S,4’S)-3-[4-(2-Dimethylaminocarbonyl)pyrrolidinylthio]-6-(l-hydroxyethyl)-1-azabicyclo[ Synthesis of 3.2.0]-hept-2-en-7-one-2-carboxylate (I)21.3 g (0.152 mol) of dimethylcyclohexanedione in Example 5 was replaced with 45.1 g (0.155 mol) of tributyltin hydride, and 0.125 g (0.108 mmol) of tetrakistriphenylphosphine palladium was added dropwise, and the other amount was added. And the same method, the obtained 16.2g (0.0426mol, 82.5%) (5R,6S,8R,2’S,4’S)-3-[4-(2-dimethylaminocarbonyl)pyrrolidinyl Sulfur]-6-(l-hydroxyethyl)-1-azabicyclo[3.2.0]-hept-2-en-7-one-2-carboxylic acid trihydrate (1) ~ meropenem. Example 7 7) (5R,6S,8R,2,S,4,S)-3-[4-(2-dimethylaminocarbonyl)pyrrolidinyl]-6-(1-hydroxyethyl)-1- Synthesis of azabicyclo[3.2.0]-hept-2-en-7-one-2-carboxylate (I) To the reaction flask, 26.2 g (0.0517 mol) of (5R, 6S, 8R, 2, S, 4’S)-[(R)-l-(hydroxy)ethyl]-3-[4-(1-allyl was added) Oxycarbonyl-1-ylaminocarbonylcarbonylpyrrolidinothio]-6-(1-allyloxycarbonylethoxy)azaabicyclo[3. 2.]-hept-2-ene-7- Ketone-2-carboxylate 01), 6.0 g (0.0387 mol) of N, N-dimethylbarbituric acid and 500 ml of dichloromethane, and 6.0 g (5.2 mmol) of tetratriphenylphosphine was added dropwise thereto. A solution of palladium in 100 ml of dichloromethane was reacted at room temperature for 5 h under nitrogen. After adding 300 ml of water to the reaction mixture, the aqueous layer was separated, and the aqueous layer was washed with ethyl acetate. THF was evaporated and evaporated, and the crystals were evaporated, and crystals were collected, and the crystals were dried in vacuo to give 15.7 g (0.0413 mol, yield: 80.1%). 5R, 6S, 8R, 2,S,4,S) – 3-[4-(2-Dimethylaminocarbonyl)pyrrolidinylthio]-6-(1-hydroxyethyl)-1-azabicyclo [3. 2. 0] -Hept-2-ene-7-keto-2-carboxylic acid trihydrate (I)-Meropectin.
ClaimsHide Dependent
Rights requesta synthetic method of meropenem, characterized in that the specific reaction route of the synthetic method

The reaction steps are as follows:1) The compound of the formula (IV) and the compound of the formula (V) are dissolved in an organic solvent and then subjected to a condensation reaction to obtain a compound of the formula (Π), the reaction time is 2 to 24 hours, and the reaction temperature is 0 to 40 ° C. ;2) The compound of the formula (Π) and the compound of the formula (VI) are dissolved in toluene, ethyl acetate or tetrahydrofuran and reacted with a base to form a compound of the formula (III), and the reaction time is ! ~ 3 hours, the reaction temperature is -20~5 °C;3) The compound of the formula (III) is dissolved in cyclohexanyl, n-glyoxime, n-octyl, toluene or xylene, and a Wittig ring-closing reaction is carried out under the action of an organophosphorus reagent to obtain a compound of the formula (VD), the organophosphorus reagent Is triphenylphosphine, tri-n-butylphosphine, triethyl phosphite or trimethyl phosphite;4) The compound of the formula (VII) is dissolved in methanol, tetrahydrofuran, acetone, n-pentane, n-hexane, diethyl ether, acetonitrile, dichloromethane, chloroform or ethyl acetate to hydrolyze the silyl ether bond under the action of an acid to obtain a formula (W). a compound; the acid is dilute hydrochloric acid, hydrofluoric acid, tetrabutylammonium fluoride, benzyltributylammonium fluoride, hydrofluoric hinge or vinegar The acid, the molar ratio of the acid to the compound of the formula is 5 to 15: 1; the temperature of the hydrolysis reaction is 0 to 40 ° C, and the reaction time is 8 to 24 hours;5) a compound of the formula
dissolved in one or more of methanol, ethanol, tert-butanol, isobutanol, isopropanol, tetrahydrofuran, dioxanthene, acetone, dichloromethane, chloroform and water After the solvent is formed, the allylic group is hydrogenated by a palladium catalyst to obtain the target product (1). The molar ratio of the palladium catalyst to the compound of the formula 1) is 0.0001 to 0.5:1; the reaction temperature is 0 to 40 ° C. , the reaction time is 2~24h.2. A method for synthesizing meropenem according to claim 1, wherein the molar ratio of the compound of the formula (IV) to the compound of the formula (V) is 1.05 to 1.0: 1, the condensing agent and The molar ratio of the compound of the formula (IV) is 1.50 to 1.05:1.The method for synthesizing meropenem according to claim 1 or 2, wherein the condensing agent is a carbodiimide reagent or hydrazine, Ν’-carbonyldiimidazole; and the organic solvent is acetone. , acetonitrile, toluene, tetrahydrofuran, chloroform or dimethylformamide.The method for synthesizing meropenem according to claim 1, wherein the molar ratio of the compound of the formula (VI) to the compound of the formula (VI) is from 1.5 to 2.5:1, the base and the The molar ratio of the compound of the formula (VI) is from 1.2 to 2:1.The method for synthesizing meropenem according to claim 1, wherein the molar ratio of the organophosphorus reagent to the compound of formula (III) in step 3) is 2-8: 1; The reaction temperature is 25 to 100 £ ^, and the reaction time is 10 to 24 hours.The method for synthesizing meropenem according to claim 3, wherein the carbodiimide reagent is dicyclohexylcarbodiimide, diisopropylcarbodiimide or 1-( 3-dimethylaminopropyl)-3-ethylcarbodiimide.7. A method for synthesizing meropenem according to claim 1, wherein the base in step 2) is an inorganic base or an organic base; when it is an inorganic base, it is sodium hydroxide, sodium carbonate or Sodium bicarbonate; when it is an organic base, it is pyridine, triethylamine, diisopropylethylamine or 2,6-lutidine.The method for synthesizing meropenem according to claim 1, wherein the palladium catalyst is palladium acetate, palladium chloride, palladium nitrate, bistriphenylphosphine palladium chloride or tetrakistriphenylphosphine. palladium.9. A method for synthesizing meropenem according to claim 1, wherein the protecting group acceptor in step 5) is morpholine, dimethylcyclohexanedione, tributyltin hydride, N, N-dimethylbarbituric acid, -ethylhexanoic acid or hexanoic acid.
SYN

Reference: Nadenik, Peter; Storm, Ole; Kremminger, Peter. Meropenem intermediate in crystalline form. WO 2005118586. (Assignee Sandoz AG, Switz)
SYN 2

Reference: Nishino, Keita; Koga, Teruyoshi. Improved process for producing carbapenem compound. WO 2007111328. (Assignee Kaneka Corporation, Japan)
SYN 3

Reference: Manca, Antonio; Monguzzi, Riccardo Ambrogio. Process for synthesizing carbapenem using Raney nickel. EP 2141167. (Assignee ACS Dobfar S.p.A., Italy)
SYN 4

Reference: Tseng, Wei-Hong; Chang, Wen-Hsin; Chang, Chia-Mao; Yeh, Chia-Wei; Kuo, Yuan-Liang. Improved process for the preparation of carbapenem using carbapenem intermediates and recovery of carbapenem. EP 2388261. (Assignee Savior Lifetec Corp., Taiwan)
STR5

Reference: Gnanaprakasam, Andrew; Ganapathy, Veeramani; Syed Ibrahim, Shahul Hameed; Karthikeyan, Murugesan; Sivasamy, Thangavel; Michael, Sekar Jeyaraj; Arulmoli, Thangavel; Das, Gautam Kumar. Preparation of meropenem trihydrate. WO 2012160576. (Assignee Sequent Anti Biotics Private Limited, India)
SYN 6

Reference: Gnanprakasam, Andrew; Ganapathy, Veeramani; Syed Ibrahim, Shahul Hameed; Karthikeyan, Murugesan; Sivasamy, Thangavel; Sekar, Jeyaraj; Arulmoli, Thangavel. Preparation of meropenem trihydrate. IN 2011CH01780. (Assignee Sequent Scientific Limited, India)
SYN7

Reference: Senthikumar, Udayampalayam Palanisamy; Sureshkumar, Kanagaraj; Babu, Kommoju Nagesh; Sudhan, Henry Syril; Kamaraj, Ponraj Pravin; Suresh, Thangaiyan. An improved process for the preparation of carbapenem antibiotic. WO 2013150550. (Assignee Orchid Chemicals & Pharmaceuticals Limited, India)
SYN 8

Reference: Ong, Winston Zapanta; Nowak, Pawel Wojciech; Kim, Jinsoo; Enlow, Elizabeth M.; Bourassa, James; Cu, Yen; Popov, Alexey; Chen, Hongming. Meropenem derivatives and uses thereof. WO 2014144285. (Assignee Kala Pharmaceuticals, Inc., USA)
SYN9

Reference: Cookson, James; McNair, Robert John; Satoskar, Deepak Vasant. Preparation of a carbapenem antibiotic by hydrogenation in the presence of a heterogeneous catalyst. WO 2015145161. (Assignee Johnson Matthey Public Limited Company, UK)
SYN 10

Reference: Gruenewald, Elena; Weidlich, Stephan; Jantke, Ralf. Process for the deprotection of a carbapenem by heterogeneous catalytic hydrogenation with hydrogen in the presence of an organic amine. WO 2018010974. (Assignee Evonik Degussa GmbH, Germany)
SYN 11


Some improvements in total synthesis of meropenem; Hu, Lai-Xing; Liu, Jun; Jin, Jie; Zhongguo Yiyao Gongye Zazhi; Volume 31; Issue 7; Pages 290-292; Journal; 2000
synhttps://www.researchgate.net/figure/Synthesis-of-MRPD-starting-from-meropenem_fig9_283306781

//////////////////////////

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////////////////////////////
Meropenem is an ultra-broad spectrum injectable antibiotic used to treat a wide variety of infections, including meningitis and pneumonia. It is a beta-lactam and belongs to the subgroup of carbapenem, similar to imipenem and ertapenem. Meropenem was originally developed by Sumitomo Pharmaceuticals. It is marketed outside Japan by AstraZeneca with the brand names Merrem and Meronem. Other brand names include Zwipen (India, Marketed by Nucleus) Mepem (Taiwan) Meropen (Japan, Korea) and Neopenem (NEOMED India) . It gained FDA approval in July 1996. It penetrates well into many tissues and body fluids including the cerebrospinal fluid, bile, heart valves, lung, and peritoneal fluid.
Meropenem, sold under the brandname Merrem among others, is an intravenous β-lactam antibiotic used to treat a variety of bacterial infections.[1] Some of these include meningitis, intra-abdominal infection, pneumonia, sepsis, and anthrax.[1]
Common side effects include nausea, diarrhea, constipation, headache, rash, and pain at the site of injection.[1] Serious side effects include Clostridium difficile infection, seizures, and allergic reactions including anaphylaxis.[1] Those who are allergic to other β-lactam antibiotics are more likely to be allergic to meropenem as well.[1] Use in pregnancy appears to be safe.[1] It is in the carbapenem family of medications.[1] Meropenem usually results in bacterial death through blocking their ability to make a cell wall.[1] It is more resistant to breakdown by β-lactamase producing bacteria.[1]
Meropenem was patented in 1983.[2] It was approved for medical use in the United States in 1996.[1] It is on the World Health Organization’s List of Essential Medicines.[3] The World Health Organization classifies meropenem as critically important for human medicine.[4]
Medical uses
The spectrum of action includes many Gram-positive and Gram-negative bacteria (including Pseudomonas) and anaerobic bacteria. The overall spectrum is similar to that of imipenem, although meropenem is more active against Enterobacteriaceae and less active against Gram-positive bacteria. It works against extended-spectrum β-lactamases, but may be more susceptible to metallo-β-lactamases.[5] Meropenem is frequently given in the treatment of febrile neutropenia. This condition frequently occurs in patients with hematological malignancies and cancer patients receiving anticancer drugs that suppress bone marrow formation. It is approved for complicated skin and skin structure infections, complicated intra-abdominal infections and bacterial meningitis.
In 2017 the FDA granted approval for the combination of meropenem and vaborbactam to treat adults with complicated urinary tract infections.[6]
Administration
Meropenem is administered intravenously as a white crystalline powder to be dissolved in 5% monobasic potassium phosphate solution. Dosing must be adjusted for altered kidney function and for haemofiltration.[7]
As with other ß-lactams antibiotics, the effectiveness of treatment depends on the amount of time during the dosing interval that the meropenem concentration is above the minimum inhibitory concentration for the bacteria causing the infection.[8] For ß-lactams, including meropenem, prolonged intravenous administration is associated with lower mortality than bolus intravenous infusion in persons with whose infections are severe, or caused by bacteria that are less sensitive to meropenem, such as Pseudomonas aeruginosa.[8][9]
Side effects
The most common adverse effects are diarrhea (4.8%), nausea and vomiting (3.6%), injection-site inflammation (2.4%), headache (2.3%), rash (1.9%) and thrombophlebitis (0.9%).[10] Many of these adverse effects were observed in severely ill individuals already taking many medications including vancomycin.[11][12] Meropenem has a reduced potential for seizures in comparison with imipenem. Several cases of severe hypokalemia have been reported.[13][14] Meropenem, like other carbapenems, is a potent inducer of multidrug resistance in bacteria.
Pharmacology
Mechanism of action
Meropenem is bactericidal except against Listeria monocytogenes, where it is bacteriostatic. It inhibits bacterial cell wall synthesis like other β-lactam antibiotics. In contrast to other beta-lactams, it is highly resistant to degradation by β-lactamases or cephalosporinases. In general, resistance arises due to mutations in penicillin-binding proteins, production of metallo-β-lactamases, or resistance to diffusion across the bacterial outer membrane.[10] Unlike imipenem, it is stable to dehydropeptidase-1, so can be given without cilastatin.
In 2016, a synthetic peptide-conjugated PMO (PPMO) was found to inhibit the expression of New Delhi metallo-beta-lactamase, an enzyme that many drug-resistant bacteria use to destroy carbapenems.[15][16]
Society and culture

Meropenem vial
Trade names
| Country | Name | Maker |
|---|---|---|
| India | Inzapenum | Dream India |
| Aurobindo Pharma | ||
| Penmer | Biocon | |
| Meronir | Nirlife | |
| Merowin | Strides Acrolab | |
| Aktimer | Aktimas Biopharmaceuticals | |
| Neopenem | Neomed | |
| Mexopen | Samarth life sciences | |
| Meropenia | SYZA Health Sciences LLP | |
| Ivpenem | Medicorp Pharmaceuticals | |
| Merofit | ||
| Lykapiper | Lyka Labs | |
| Winmero | Parabolic Drugs | |
| Bangladesh | ||
| Meroject | Eskayef Pharmaceuticals Ltd. | |
| Merocon | Beacon Pharmaceuticals | |
| Indonesia | Merofen | Kalbe |
| Brazil | Zylpen | Aspen Pharma |
| Japan, Korea | Meropen | |
| Australia | Merem | |
| Taiwan | Mepem | |
| Germany | Meronem | |
| Nigeria | Zironem | Lyn-Edge Pharmaceuticals |
| US | Meronem | AstraZeneca |
| … | Merosan | Sanbe Farma |
| Merobat | Interbat | |
| Zwipen | ||
| Carbonem | ||
| Ronem | Opsonin Pharma, BD | |
| Neopenem | ||
| Merocon | Continental | |
| Carnem | Laderly Biotech | |
| Penro | Bosch | |
| Meroza | German Remedies | |
| Merotrol | Lupin) | |
| Meromer | Orchid Chemicals | |
| Mepenox | BioChimico | |
| Meromax | Eurofarma | |
| Ropen | Macter | |
| mirage | adwic | |
| Meropex | Apex Pharma Ltd. | |
| Merostarkyl | Hefny Pharma Group[17] |
References
- ^ Jump up to:a b c d e f g h i j “Meropenem”. The American Society of Health-System Pharmacists. Retrieved 8 December 2017.
- ^ Fischer, Janos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 497. ISBN 9783527607495.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ World Health Organization (2019). Critically important antimicrobials for human medicine (6th revision ed.). Geneva: World Health Organization. hdl:10665/312266. ISBN 9789241515528.
- ^ AHFS Drug Information (2006 ed.). American Society of Health-System Pharmacists. 2006.
- ^ Commissioner, Office of the (24 March 2020). “Press Announcements – FDA approves new antibacterial drug”. http://www.fda.gov.
- ^ Bilgrami, I; Roberts, JA; Wallis, SC; Thomas, J; Davis, J; Fowler, S; Goldrick, PB; Lipman, J (July 2010). “Meropenem dosing in critically ill patients with sepsis receiving high-volume continuous venovenous hemofiltration” (PDF). Antimicrobial Agents and Chemotherapy. 54 (7): 2974–8. doi:10.1128/AAC.01582-09. PMC 2897321. PMID 20479205.
- ^ Jump up to:a b Yu Z, Pang X, Wu X, Shan C, Jiang S (2018). “Clinical outcomes of prolonged infusion (extended infusion or continuous infusion) versus intermittent bolus of meropenem in severe infection: A meta-analysis”. PLOS ONE. 13 (7): e0201667. Bibcode:2018PLoSO..1301667Y. doi:10.1371/journal.pone.0201667. PMC 6066326. PMID 30059536.
- ^ Vardakas KZ, Voulgaris GL, Maliaros A, Samonis G, Falagas ME (January 2018). “Prolonged versus short-term intravenous infusion of antipseudomonal β-lactams for patients with sepsis: a systematic review and meta-analysis of randomised trials”. Lancet Infect Dis. 18 (1): 108–120. doi:10.1016/S1473-3099(17)30615-1. PMID 29102324.
- ^ Jump up to:a b Mosby’s Drug Consult 2006 (16 ed.). Mosby, Inc. 2006.
- ^ Erden, M; Gulcan, E; Bilen, A; Bilen, Y; Uyanik, A; Keles, M (7 March 2013). “Pancytopenýa and Sepsýs due to Meropenem: A Case Report” (PDF). Tropical Journal of Pharmaceutical Research. 12 (1). doi:10.4314/tjpr.v12i1.21.
- ^ “Meropenem side effects – from FDA reports”. eHealthMe.
- ^ Margolin, L (2004). “Impaired rehabilitation secondary to muscle weakness induced by meropenem”. Clinical Drug Investigation. 24(1): 61–2. doi:10.2165/00044011-200424010-00008. PMID 17516692. S2CID 44484294.
- ^ Bharti, R; Gombar, S; Khanna, AK (2010). “Meropenem in critical care – uncovering the truths behind weaning failure”. Journal of Anaesthesiology Clinical Pharmacology. 26 (1): 99–101.
- ^ “New molecule knocks out superbugs’ immunity to antibiotics”. newatlas.com. 20 January 2017. Retrieved 2017-01-25.
- ^ K., Sully, Erin; L., Geller, Bruce; Lixin, Li; M., Moody, Christina; M., Bailey, Stacey; L., Moore, Amy; Michael, Wong; Patrice, Nordmann; M., Daly, Seth (2016). “Peptide-conjugated phosphorodiamidate morpholino oligomer (PPMO) restores carbapenem susceptibility to NDM-1-positive pathogens in vitro and in vivo”. Journal of Antimicrobial Chemotherapy. 72 (3): 782–790. doi:10.1093/jac/dkw476. PMC 5890718. PMID 27999041.
- ^ “Hefny Pharma Group”. hefnypharmagroup.info. Retrieved 2018-05-22.
External links
- “Meropenem”. Drug Information Portal. U.S. National Library of Medicine.
| Clinical data | |
|---|---|
| Trade names | Merrem, others |
| AHFS/Drugs.com | Monograph |
| Pregnancy category | AU: B2 |
| Routes of administration | Intravenous |
| ATC code | J01DH02 (WHO) |
| Legal status | |
| Legal status | AU: S4 (Prescription only)UK: POM (Prescription only)US: ℞-only |
| Pharmacokinetic data | |
| Bioavailability | 100% |
| Protein binding | Approximately 2% |
| Elimination half-life | 1 hour |
| Excretion | Renal |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 119478-56-7 |
| PubChem CID | 441130 |
| DrugBank | DB00760 |
| ChemSpider | 389924 |
| UNII | FV9J3JU8B1 |
| KEGG | D02222 |
| ChEBI | CHEBI:43968 |
| ChEMBL | ChEMBL127 |
| PDB ligand | MEM (PDBe, RCSB PDB) |
| CompTox Dashboard (EPA) | DTXSID7045526 |
| ECHA InfoCard | 100.169.299 |
| Chemical and physical data | |
| Formula | C17H25N3O5S |
| Molar mass | 383.46 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI | |
| (verify) |
Patent
Publication numberPriority datePublication dateAssigneeTitleUS4888344A *1986-07-301989-12-19Sumitomo Pharmaceuticals Company, LimitedCarbapenem compound in crystalline form, and its production and useCN101348486A *2008-08-292009-01-21深圳市海滨制药有限公司Preparation of meropenemCN101962383A *2010-11-122011-02-02上海巴迪生物医药科技有限公司Synthesis method of meropenemFamily To Family CitationsJPS6475488A *1987-09-171989-03-22Sumitomo PharmaProduction of beta-lactam compound* Cited by examiner, † Cited by third party
Publication numberPriority datePublication dateAssigneeTitleFamily To Family CitationsCN101962383A *2010-11-122011-02-02上海巴迪生物医药科技有限公司Synthesis method of meropenemCN102250096B *2011-09-052016-04-06江西华邦药业有限公司A kind of preparation method of meropenemCN104072523B *2014-07-142017-10-24上海上药新亚药业有限公司The preparation method of BiapenemCN108191869A *2018-01-222018-06-22重庆天地药业有限责任公司The purification process of Meropenem
PublicationPublication DateTitleEP0007973B11984-02-01Process for the preparation of thienamycin and intermediatesUS4631150A1986-12-23Process for the preparation of penemsWO2012062035A12012-05-18Synthesis method for meropenemWO2010022590A12010-03-04Method for preparation of meropenemUS4443373A1984-04-17Process for the production of antibiotic penemsWO2008035153A22008-03-27Process for the preparation of beta-lactam antibioticEP0167154B11990-01-03Process for preparing 4-acetoxy-3-hydroxyethylazetizin-2-one derivativesKR101059339B12011-08-24Method for preparing carbapenem compound for oral administrationKR100886347B12009-03-03Process for stereoselective preparation of 4-BMA using a chiral auxiliaryUS4841043A1989-06-20Stereoselective synthesis of 1-β-alkyl carbapenem antibiotic intermediatesUS4772683A1988-09-20High percentage beta-yield synthesis of carbapenem intermediatesJP2000344774A2000-12-12Production of carbapenem compoundAU745980B22002-04-11Titanium catalyzed preparation of carbapenem intermediatesUS5700930A1997-12-234-substituted azetidinones as precursors to 2-substituted-3-carboxy carbapenem antibiotics and a method of producing themJP2002338572A2002-11-27Method for producing carbapenemsJP3684339B22005-08-17Method for producing carbapenem compoundsEP0066301B11986-01-22Intermediates for the preparation of thienamycin and process for preparing the sameWO2001053305A12001-07-26Processes for the preparation of carbapenem derivativesAU737502B22001-08-23Preparation of beta-methyl carbapenem intermediatesJP3213734B22001-10-02New β-lactam compoundsJP2004107289A2004-04-08Method for producing vinyl sulfide compoundJPH085853B21996-01-24Lactam compound and its manufacturing methodJPH0827168A1996-01-30Carbapenem intermediate fieldEP0204440A11986-12-10Azetidine derivatives productionWO1994021638A11994-09-29Process for the preparation of condensed carbapeneme derivatives
ApplicationPriority dateFiling dateTitleCN 2010105416652010-11-122010-11-12Synthesis method of meropenemCN201010541665.52010-11-12
Nmrhttps://www.researchgate.net/figure/1HNMR-spectra-of-meropenem-hydrolysis-catalyzed-by-NDM-1-Ecoli-cells-Only-1H-signals-of_fig3_272515470


NMRNMR spectra monitoring meropenem hydrolysis catalyzed by NDM-1. a¹H NMR spectrum of hydrolyzed meropenem recorded before and 6 or 20 min after NDM-1 addition to the reaction system. b Part of a ROESY spectrum of the hydrolysis product. Diagonal and cross peaks are shown in blue and red, respectively. Proton signal assignments are labeled beside the peaks. The chemical shifts of H2, H1, H5, and H10 are highlighted by dashed linesSEEhttps://www.mdpi.com/1420-3049/23/11/2738/htm
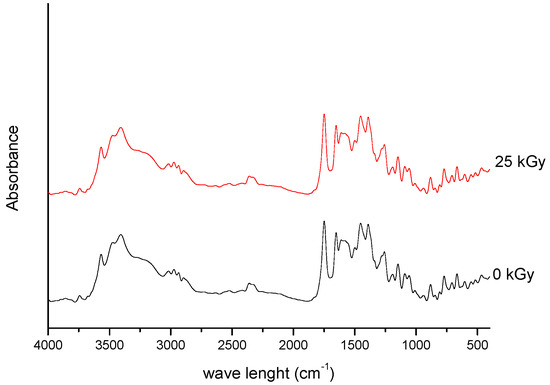
Figure 1. FT-IR spectra of unirradiated and irradiated (25 kGy) meropenem.
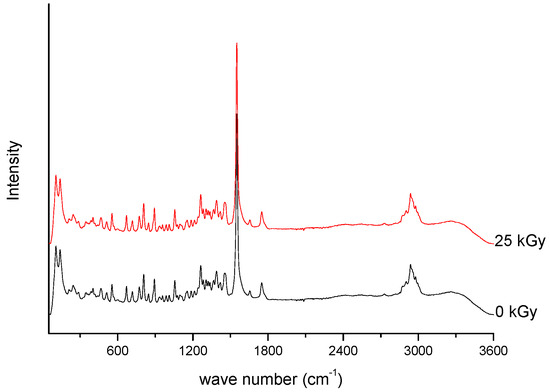
Figure 2. Raman spectra of unirradiated and irradiated (A-25 kGy) meropenem.
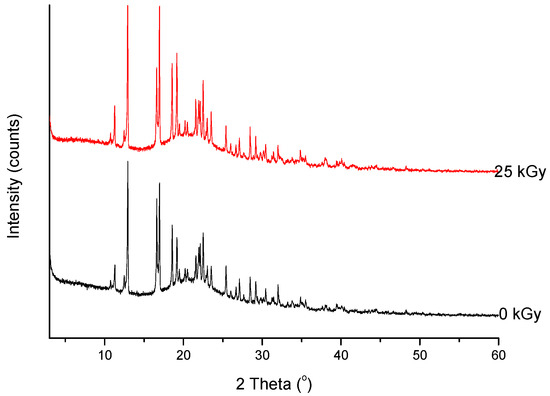
Figure 6. XRPD diffractograms of unirradiated and irradiated (25 kGy) meropenem.
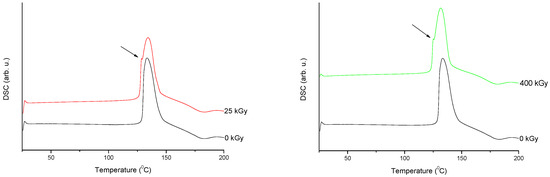
Figure 7. Differential scanning calorimetry (DSC) curves of non-irradiated and irradiated (A-25 kGy, B-400 kGy) meropenem. The arrows indicate the changes in the DSC spectrum after irradiation.
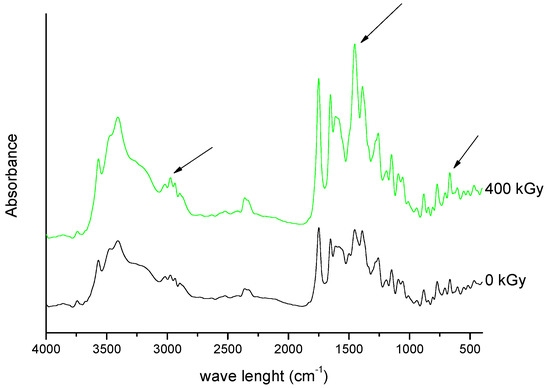
Figure 9. FT-IR spectra of unirradiated and irradiated (400 kGy) meropenem. The arrows indicate the changes in the FT-IR spectrum after irradiation.
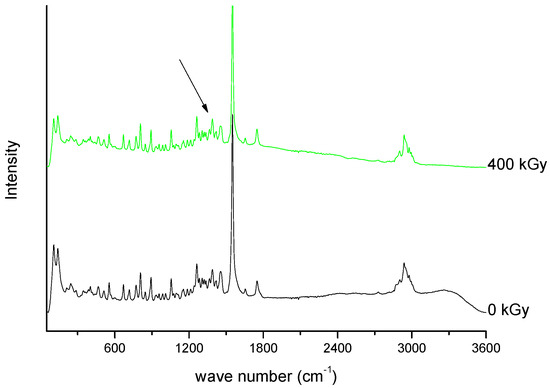
Figure 10. Raman spectra of unirradiated and irradiated (400 kGy) meropenem. The arrow indicates the change in the Raman spectrum after irradiation.
//////////////Meropenem, Merrem, intravenous β-lactam antibiotic, bacterial infections, meningitis, intra-abdominal infection, pneumonia, sepsis, anthrax, Antibiotic SM 7338, ICI 194660, SM 7338, ANTIBACTERIALS
[H][C@]1([C@@H](C)O)C(=O)N2C(C(O)=O)=C(S[C@@H]3CN[C@@H](C3)C(=O)N(C)C)[C@H](C)[C@]12[H]

NEW DRUG APPROVALS
ONE TIME
$10.00
BIAPENEM

Biapenem
RPX7009
- Molecular FormulaC15H18N4O4S
- Average mass350.393 Da
Biapenern
CL 186-815LJ
C10,627LJ
C10627LJC 10627
omegacin
YR5U3L9ZH1
(4R,5S,6S)-3-((6,7-dihydro-5H-pyrazolo[1,2-a][1,2,4]triazol-4-ium-6-yl)thio)-6-((R)-1-hydroxyethyl)-4-methyl-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylate
[4R-[4a,5b,6b(R*)]]-6-[[2-Carboxy-6-(1-hydroxyethyl)-4-methyl-7-oxo-1-azabicyclo[3.2.0]hept-2-en-3-yl]thio]-6,7-dihydro-5 H-pyrazolo[1,2-a][1,2,4]triazol-4-ium inner salt
120410-24-4[RN]
5H-Pyrazolo[1,2-a][1,2,4]triazol-4-ium, 6-[[(4R,5S,6S)-2-carboxy-6-[(1R)-1-hydroxyethyl]-4-methyl-7-oxo-1-azabicyclo[3.2.0]hept-2-en-3-yl]thio]-6,7-dihydro-, inner salt [ACD/Index Name]
6-[[(4R,5S,6S)-2-Carboxy-6-[(1R)-1-hydroxyethyl]-4-methyl-7-oxo-1-azabicyclo[3.2.0]hept-2-en-3-yl]thio]-6,7-dihydro-5H-pyrazolo[1,2-a][1,2,4]triazol-4-ium inner salt
7074
(4R,5S,6S)-3-(6,7-Dihydro-5H-pyrazolo[1,2-a][1,2,4]triazol-4-ium-6-ylsulfanyl)-6-[(1R)-1-hydroxyethyl]-4-methyl-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylate
TL8000539UNII:YR5U3L9ZH1UNII-YR5U3L9ZH1биапенем
بيابينام比
阿培南
INDIA CDSCO APPROVED 25 SEPT 2021, BDR PHARMA, 
https://www.cdsco.gov.in/opencms/resources/UploadCDSCOWeb/2018/UploadCTApprovals/BDR.pdfhttps://medicaldialogues.in/news/industry/pharma/bdr-pharma-gets-dcgi-nod-for-generic-antibiotic-drug-biapenem-82384

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter a
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
Biapenem (INN) is a carbapenem antibiotic. It has in vitro activity against anaerobes.[1] 1-β-methyl-carbapenem antibiotic. Approved in Japan in 2001.
PATENT
EP 168707
EP 289801
JP 02088578
ZA 9100014
EP 533149
CN 1995040
IN 2006DE01555
CN 101121716
IN 2008CH00177
CN 101805359
CN 101851206
CN 101935321
CN 111875622
WO 2018074916
WO 2016059622
US 20150328323
WO 2015151081
WO 2015155753
WO 2015151078
US 20150284416
WO 2015151080
US 20150038726
WO 2014104488
IN 2013MU00181
WO 2014111957
CN 103570750
WO 2014097221
IN 2012CH01371
WO 2013150550
PAPERS
Journal of Organic Chemistry (1992), 57(15), 4243-9.
Heterocycles (1993), 36(8), 1729-34.
Journal of Antibiotics (1993), 46(12), 1866-82.
e-EROS Encyclopedia of Reagents for Organic Synthesis (2008), 1-3.
Bioorganic & medicinal chemistry letters (2009), 19(17), 5162-5.
IP.com Journal (2014), 14(12A), 1-3
IP.com Journal (2014), 14(10A), 1-2.
Bioorganic & medicinal chemistry (2013), 21(18), 5841-50.

NEW DRUG APPROVALS
one time
$10.00
PATENT
https://patents.google.com/patent/WO2014097221A1/esBiapenem is chemically known as 6-[[2(4R,5S,6S)-carboxy-6-[(lR)- hydroxy ethyl] -4-methyl-7-oxo- 1 -azabicyclo [3.2.0]hept-2-en-3 -yljthio] 6,7-dihydro-5H- pyrazolo[l,2-a][l,2,4]triazol-4-ium inner salt, and is represented by Formula 1. It is indicated for the treatment of bacterial infection and sepsis.

Formula 1U.S. Patent No. 4,866,171, in Example 6, discloses the purification of biapenem using chromatography and/or lyophilization techniques. This patent also describes a process for the conversion of amorphous biapenem into a crystalline form by dissolving the amorphous biapenem in water while heating, followed by cooling, then washing the obtained crystals with a 50% aqueous ethanol solution.U.S. Patent No. 5,241,073 describes a process for the purification of biapenem involving column chromatography and crystallization with ethanol.U.S. Patent No. 5,286,856 describes a process for the crystallization of biapenem from an aqueous solution, comprising maintaining the temperature of the aqueous solution from eutectic temperature (-10°C to -2°C) to a temperature lower than 0°C, followed by lyophilization.The Journal of Organic Chemistry, 63(23):8145-8149 (1998) describes the purification of biapenem involving resin chromatography.The present invention provides an alternate process for the purification of biapenem that avoids making use of tedious techniques like chromatography and lyophilization. At the same time, it results in a high yield and high purity of the final product. Advantageously, the crystalline biapenem of this invention can be directly isolated from the reaction mixture. Further, the process of the present invention involves fewer steps, is easily scalable, and industrially advantageous.EXAMPLESExample 1 : Purification of BiapenemBiapenem (12 g) was added into water (300 mL) at 65°C, stirred for 5 minutes, and cooled to 30°C within 10 minutes. Enoantichromos carbon (0.6 g) was added to the reaction mixture and stirred for 10 minutes to 15 minutes at 25°C to 30°C. The reaction mixture was filtered through a hyflo bed and washed with water (36 mL). The filtrate obtained was passed through a 0.45 micron filter, and its pH was adjusted to 5.5 using 5% aqueous sodium hydroxide solution at 10°C to 15°C. Acetone (336 mL) was added to the reaction mixture at 5°C to 10°C. The resultant slurry was stirred for 3 hours at 5°C to 10°C, filtered, and the obtained solid was washed with acetone (60 mL). The solid was dried under reduced pressure (720 mmHg) at 30°C to 35°C to obtain the title product as white crystals.Yield: 84%HPLC Purity: 99.87% Example 2: Purification of BiapenemBiapenem (18 g) was added into water (450 mL) at 65°C, stirred for 5 minutes, and cooled to 30°C within 10 minutes. Enoantichromos carbon (0.9 g) was added to the reaction mixture and stirred for 30 minutes at 25°C to 30°C. The reaction mixture was filtered through a hyflo bed and washed with water (54 mL). The filtrate obtained was passed through a 0.45 micron filter and its pH was adjusted to 4.9 using 5% aqueous sodium hydroxide solution at 10°C to 15°C. Acetone (504 mL) was added to the reaction mixture at 10°C to 15°C. The resultant slurry was stirred for 3 hours at 5°C to 10°C, filtered, and the obtained solid was washed with acetone (90 mL). The solid was dried under reduced pressure (720 mmHg) at 35°C to 40°C to obtain the title product as white crystals.Yield: 81.77%HPLC Purity: 99.80%
PATENThttps://patentscope.wipo.int/search/en/detail.jsf?docId=WO2013150550
The present invention relates to an improved process for the preparation of carbapenem antibiotic; more particularly relates to the preparation of Ertapenem monosodium salt of formula (I) having purity greater than 98.5% and having pharmaceutically acceptable level of residual solvent and palladium content.
The US patents namely US 5,478,820 and US 5,856,321 disclose various processes for preparing Ertapenem and its sodium salt. Example 12 of US 5,478,820 discloses a process in which the Ertapenem was isolated using column purification followed by freeze-drying technique. According to Example-4 of this patent disodium salt of Ertapenem was prepared by dissolving crude product in water using NaHCO3, followed by purification using column chromatography and subsequent lyophilization.
US 6,504,027 provides a process for preparing Ertapenem in crystalline form which comprises deprotecting and extracting a polar organic solution containing a crude mono-protected Ertapenem of formula
wherein P represents protecting group and X represents charge balancing group like sodium
with C4.10 alcohol in the presence of ion-pairing reagent followed by adjusting the pH of the aqueous layer to 5.5 and crystallizing using methanol and 1-propanol to produce a crystalline compound; this patent process involves operations like
multiple extractions which is cumbersome in plant and said operation affects the overall yield.
US 7,145,002 provides a process for producing Ertapenem or its sodium salt and/or its solvate in crystalline form. This patent states (refer para 3, lines 31-41) that contact of Ertapenem sodium with water and alcoholic solvents results in the formation of crystalline solvates. The processes reported in examples- 1 & 2 provide crystalline Ertapenem monosodium which is isolated from a mixture of methanol, 1-propanol and water followed by washing with aqueous isopropyl alcohol which results in the formation of crystalline solvate of Ertapenem sodium. Applicant found the Ertapenem monosodium obtained according to this process contain higher amount of residual solvent and palladium content.
US 7,022,841 provide a process for reducing the levels of organic solvents in Ertapenem to pharmaceutically acceptable levels. This patent discloses (Refer para 1, lines 52-60) that Ertapenem sodium obtained from water/alcohol mixture according to US 7, 145,002 becomes amorphous when water content of the solid is reduced and further the organic solvent present in the solid is not readily removed. In view of this drawback, this patent provides a process wherein the water content of Ertapenem sodium is maintained between 13-25% during the washing and drying process. This patent further discloses that (Refer para 9, lines 6-14) the washing of Ertapenem sodium can be carried out using anhydrous solvents which results in the formation of amorphous solid, which is then dried using hydrated nitrogen by increasing the water content of the solid. Due to the hygroscopic and unstable nature of Ertapenem sodium when in contact with water, the above processes result in more degradation of Ertapenem. The patent further discloses in example 5 that the degradation of Ertapenem sodium is more when it takes more time for drying.
Further this patent requires repetitive washing and control of moisture content to get the desired results.
For isolation of Ertapenem sodium from the reaction mass, all the above discussed prior art patents utilize methanol and 1-propanol as crystallization solvent. The filtration of Ertapenem sodium formed by using these solvents or their mixture takes longer time duration and subsequent drying for the removal of residual solvent also takes several hours due to occlusion of solvent into Ertapenem sodium. During these operations the Ertapenem sodium degrades an results in the formation of many impurities such as several dimers, methanolysis impurity etc., and hence the reported processes is not suitable to manufacture Ertapenem sodium on commercial scale with purity greater than 98.5% and with pharmaceutically acceptable level of residual solvent content.
Methanolysis impurity Dimer-I
Dimer-II
Further the applicant found that Ertapenem monosodium isolated by following the process reported in prior art was having palladium content above the pharmaceutically acceptable level. Hence the process reported in prior art is not suitable on manufacturing scale where maintaining stringent technological condition is cumbersome and involves higher operating cost.
Thus all the reported processes suffer in terms of one or more of the following facts:
■ Filtration time of Ertapenem sodium takes several hours.
■ Drying time takes several hours due to occlusion of solvent and nature of the solid.
■ Stringent technological condition is required for maintenance of moisture content during washing & drying operation.
■ Palladium content is found to be higher (greater than 25 ppm) which is not acceptable for pharmaceutical products.
■ The isolated Ertapenem sodium is having higher amount of residual solvents.
■ The purity is reduced over to several hours of filtration & drying.
With our continued research for developing a process for the preparation of Ertapenem monosodium of formula (I) to overcome the above mentioned drawbacks, we surprisingly found that when esters of organic acid were used as solvents in place of 1-propanol, the solid obtained was easily filterable with less cycle time. Further the washing with hydrocarbon solvents containing 0-75% alcoholic solvent followed by drying results in Ertapenem having residual solvent content well below the pharmaceutically acceptable levels. The use of thiourea, thiosemicarbazide or their N-substituted derivatives in the presence of organic solvents during isolation brings down the palladium content to pharmaceutically acceptable level.
The Ertapenem or its sodium salt can be prepared according the processes provi
(I)
P’ and P” represent carboxylic protecting groups and X is H or Na
Scheme-1
The present invention is illustrated with the following examples, which should not be construed to limit the scope of the invention.
Example- I
Preparation of Ertapenem monosodium of formula (I)
Step-I:
To a stirred solution of p-nitrobenzyl (4R,5S,6S)-3-(diphenyloxy)phosphoryloxy-6-[(lR)-l-hydroxyethyl]-4-methyl-7-oxo-l-azabicyclo[3,2,0]hept-2-ene-2-carboxylate (compound II) (100 g) and (2S,4S)-2-[[(3-carboxyphenyl) amino]carbonyl]-4-mercapto-l-(4-nitrobenzyl)pyrrolidinecarboxylate (compound III) (75 g) in N,N-dimethylformamide was added Ν,Ν-diisopropylethylamine at -30 to -40° C and stirred. The reaction mass, after completion of the reaction, was quenched with a mixture of phosphate buffer solution-ethyl acetate and the pH was adjusted to 5 – 6 with phosphoric acid. The organic layer was separated, washed with water and subjected to carbon treatment. To the organic layer containing the compound of formula (IV) (wherein P’ and P” refers to p-nitrobenzyl), a solution of sodium 2-ethylhexanoate (42 g in 500 mL methanol) was added and taken to next step as such. (If required the compound of formula (IV) is isolated either as sodium salt or as free acid by following the process reported in prior art and taken further)
Step-II:
To the Step-I organic layer containing the compound of formula (IV) (wherein P’ and P” refers to p-nitrobenzyl & X is Na), 3-(N-morpholino)propanesulfonic acid solution was added and subjected to hydrogenation using palladium on carbon at 8- 10° C with 9-10 kg hydrogen pressure. The reaction mass, after completion of reaction, was filtered to remove palladium on carbon. To the filtrate, thiourea (5 g) and tetrahydrofuran were added and stirred. The aqueous layer was separated and treated with carbon and neutral alumina at 10-15° C while degassing and filtered. The filtrate was added to methanol at -20° C and the pH was adjusted to 5 – 6 using aqueous acetic acid. To the mass, ethyl acetate was added and stirred. The solid obtained was filtered, washed with a mixture of cyclohexane: ethanol (200 ml) and dried under vacuum. Yield: 46 g; Purity by HPLC: 98.93%; Palladium content: 1.8 ppm by ICP MS
The HPLC purity of Ertapenem monosodium was checked using the following parameters
Column : Zorbax Eclipse plus C8, (50 mm x 4.6 mm), 1.8μ).
Mobile phase : Ammoniam acetate buffer: Acetonitile: water
Detector : UV at 250 nm
Flow rate : 0.5 mL/min
Run time : 45 min.
Example- II
Preparation of Ertapenem monosodium of formula (I)
To the Step-I organic layer as provided in Example-I, 3-(N-morpholino)propanesulfonic acid solution was added and subjected to hydrogenation using palladium on carbon at 8-10° C with 9-10 kg hydrogen pressure. The reaction mass, after completion of reaction, was filtered and the filtrate was treated with thiourea and 2-methyltetrahydrofuran and the layers separated. The aqueous layer was treated with carbon & neutral alumina at 10-15° C and filtered. The filtrate was mixed with methanol at -20° C and the pH was adjusted to 5 – 6 using aqueous acetic acid. To the mass, ethyl acetate was added and stirred. The solid obtained was filtered, washed with cyclohexane (200 ml) and
dried under vacuum. Yield: 44 g; Purity by HPLC: 98.84%; Palladium content: 0.93 ppm by ICP MS
The term ICP MS method refers to the inductively coupled plasma mass spectrometry. The following parameter was used to determine the content of palladium.
The carbapenem was digested in a closed vessel system in presence of reagents Nitric acid, Hydrogen peroxide and Hydrochloric acid by using Microwave reaction system with microwave radiation power 1200 Watts. The digested sample was introduced into inductively coupled plasma mass spectrometer by help of Peltier cooled spray chamber. The sample aerosol is getting atomized then ionized in the argon plasma. The ionized Palladium was estimated by using Quadrupole mass detector. The sample was quantified against NIST traceable reference standards at mass number ! 05.
Example- III
Preparation of Ertapenem monosodium of formula (I)
To the Step-I organic layer as provided in Example-I, 3-(N-morpholino)propanesulfonic acid solution was added and hydrogenated at 9-10 kg pressure using palladium on carbon at 8-10° C. The reaction mass, after completion of reaction, was filtered and the filtrate was treated with thiourea and tetrahydrofuran and the layers separated. The aqueous layer was separated and treated with carbon, neutral alumina at 10-15° C and filtered. The filtrate was mixed with methanol at -20° C and the pH was adjusted to 5 – 6 using aqueous acetic acid. To the mass, ethyl acetate was added and stirred. The solid obtained was filtered, washed with a mixture of toluene: ethanol (200 ml) and dried under vacuum. Yield: 42 g; Purity by HPLC: 99.03%
Example- IV
Preparation of Ertapenem monosodium of formula (I)
To the Step-I organic layer as provided in Example-I, 3-(N-morpholino)propanesulfonic acid solution was added and hydrogenated using palladium on carbon at 8-10° C. The reaction mass, after completion of reaction was filtered and the filtrate was treated with thiosemicarbazide and tetrahydrofuran and the layers separated. The aqueous layer was treated with carbon, neutral alumina at 10-15° C and filtered. The filtrate was mixed with methanol at -20° C followed by the addition of ethyl acetate and stirred. The solid obtained was filtered, washed with a mixture of cyclohexane: ethanol (200 ml) and dried under vacuum. Yield: 41 g; Purity by HPLC: 99.13%; Palladium content: 1.71 ppm by ICP MS
Example- V
Preparation of Ertapenem monosodium of formula (I)
To the Step-I organic layer as provided in Example-I, 3-(N-morpholino)propanesulfonic acid solution was added and subjected to hydrogenation using palladium on carbon at 8-10° C with 9-10 kg hydrogen pressure. The reaction mass, after completion of reaction, was filtered and the filtrate was treated with thiourea and 2-methyltetrahydrofuran and the layers separated. The aqueous layer was treated with carbon, neutral alumina at 10-15° C and filtered. The filtrate was mixed with methanol at -20° C and the pH was adjusted to 5 – 6 using aqueous acetic acid. To the mass, a mixture of ethyl acetate containing 10% methyl acetate was added and stirred. The solid obtained was
filtered, washed with cyclohexane:ethanol and dried under vacuum. Yield: 40.5 g; Purity by HPLC: 98.77%; Palladium content: 1.43 ppm by ICP MS
Example-VI
(V ) (V I )
The diprotected Meropenem of formula (V) (where P and P’ were p-nitrobenzyl) was dissolved in tetrahydrofuran and 3-(N-morpholino)propanesulfonic acid buffer and hydrogenated using palladium on carbon at 9-10 kg hydrogen pressure. The mass was filtered and the filtrate was washed with ethyl acetate. The aqueous layer was treated with thiourea and 2-methyltetrahydrofuran. The aqueous layer was separated, treated with carbon and degassed. The carbon was filtered off and acetone was added to the filtrate to crystallize Meropenem trihydrate of formula (VI). The product was filtered and washed with aq. acetone and dried under vacuum to get Meropenem trihydrate. Purity: 99.8%; Pd content: 0.08 ppm
Reference example-I:
Preparation of Ertapenem monosodium of formula (I)
To Step-I organic layer as provided in Example-I, 3-(N-morpholino)propanesulfonic acid solution was added and hydrogenated at 9-10 kg pressure using palladium on carbon at 8-10° C. The reaction mass, after completion of reaction, was filtered. The filtrate was treated with thiourea and tetrahydrofuran and the layers separated. The aqueous layer was treated with carbon and neutral alumina at 10-15° C and filtered. The filtrate was mixed with methanol at -20° C and the pH was adjusted to 5.5-5.7 using aqueous acetic acid. To the mass ethyl acetate was added and stirred. The solid obtained was filtered, washed with ethanol (5 * 100 ml) and dried under vacuum. Yield: 31 g; Purity by HPLC: 96.76%
Reference example-II:
Preparation of Ertapenem monosodium of formula (I)
To the Step-I reaction mass , as provided in Example-I, 3-(N-morpholino)propanesulfonic acid solution was added and hydrogenated at 9-10 kg pressure using palladium on carbon at 8-10° C. The reaction mass, after completion of reaction was filtered and the layers separated. The aqueous layer was treated with carbon and neutral alumina at 10-15° C and filtered. The filtrate was mixed with methanol at -20° C and the pH was adjusted to 5.5-5.7 using aqueous acetic acid. To the mass, ethyl acetate was added and stirred. The solid obtained was filtered, washed with a mixture of cyclohexane: ethanol and dried under vacuum. Yield: 43 g; Purity by HPLC: 98.6%; Palladium content: 35.8 ppm by ICP MS.
Reference example-HI:
Preparation of Ertapenem monosodium of formula (I)
To the Step-I reaction mass as provided in Example-I, 3-(N-morpholino)propanesulfonic acid solution was added and hydrogenated at 9-10 kg pressure using palladium on carbon at 8-10° C. The reaction mass, after completion of reaction, was filtered and the layers separated. The aqueous layer was treated with carbon, neutral alumina at 10-15° C and filtered. The filtrate was mixed with 1-propanol at -5° C and the pH was adjusted to 5.5-5.7 using aqueous acetic acid. To the mass methanol and 1-propanol were added and stirred. The solid obtained was filtered, washed with ethanol and dried under nitrogen atmosphere in vacuum. Yield: 25 g; Purity by HPLC: 97 %.: palladium content: 38.2 ppm
The following tables illustrate the advantages of the present invention over prior art process:
Table-I: Comparison of present process with prior art process
The crystallization and washing method disclosed in US 7,022,841 was followed.
The above table indicates that the use of ethyl acetate as crystallization solvent results with improved yield and high purity with less filtration and drying time thereby increasing the productivity significantly on manufacturing scale. Further the use of thiourea or thiosemicarbazide as reagents in the present process results in the pharmaceutically acceptable level of palladium content.
Table-II: Comparison of solvents for washing Ertapenem monosodium
The above table indicates that the use of hydrocarbon solvents containing 0-75% of alcoholic solvent helps in washing to remove the residual solvent content in shorter duration and with single run wash. On the other hands the use of ethanol alone results in Ertapenem monosodium having less yield and purity requiring repetitive washing.
Table-IH: Effect of different reagent in reduction of palladium content
Reagent : thiourea, thiosemicarbazide or its N-substituted derivatives
Advantages of the process of the present invention:
> The use of ester of an organic acid for the crystallization of Ertapenem sodium results in fast filtration and reduced cycle time, thereby increasing the productivity.
> Washing of Ertapenem sodium with hydrocarbon solvent optionally containing alcohol results in improved physical nature of Ertapenem sodium resulting in reduced washing and drying time thereby avoid the degradation of Ertapenem and providing Ertapenem sodium with purity greater than 98.5% by HPLC.
Use of thiourea, thiosemicarbazide or their N-substituted derivatives in the process results in Ertapenem sodium having pharmaceutically acceptable level of palladium content.
PATENT
https://patents.google.com/patent/WO2002057266A1/enEXAMPLE

PNB = p-nitrobenzyl

Ia’A hydrogenator is charged with 63 g of 10% Pd on carbon catalyst (dry weight) in 1.8 L of water. The vessel is placed under hydrogen then vented and placed under nitrogen. Sodium hydroxide (68 g, 50%) is charged adjusting the pH to about 7.5 with carbon dioxide.The enol phosphate (170 g) and the thiol (86 g) are dissolved in 1.3‘L of N- ethylpyrrolidinone (NEP). The mixture is cooled to below -40°C and 1,1,3,3- tetramethylguanidine (109 g) is added. After 3 hours, the reaction mixture is quenched into the hydrogenator at below 15°C adjusting the pH to about 8 with carbon dioxide. The vessel is placed under hydrogen. When the reaction is complete, the hydrogen is vented and the reaction mixture is treated with activated carbon and filtered. The filtrate is extracted with iso-amyl alcohol containing diphenylphosphoric acid (240 g) and 50% NaOH (44 g). The resulting aqueous solution is further extracted with iso-amyl alcohol to give an aqueous solution containing at least 90 mg/mL of the product. Both extractions are performed using two CINC centrifugal separators set in series for countercurrent extraction. The pH is adjusted to 5.5 with acetic acid. The product is crystallized by adding equal volumes of methanol and 1- propanol at below -5°C and isolated by filtration. The solid is washed with a mixture of 2-propanol and water (85: 15 v/v) then dried to yield a compound of formula la’.While certain preferred embodiments of the invention have been described herein in detail, numerous alternative embodiments are contemplated as falling within the scope of the appended claims. Consequently the invention is not to be limited thereby.
Patent Citations
Publication numberPriority datePublication dateAssigneeTitleUS4866171A1987-04-111989-09-12Lederle (Japan), Ltd.(1R,5S,6S)-2-[(6,7-dihydro-5H-pyrazolo[1,2-a][1,2,4]triazolium-6-yl)]thio-6-[R-1-hydroxyethyl]-1-methyl-carbapenum-3-carboxylateUS5241073A1990-10-121993-08-31Lederle (Japan)Process for preparing (1R,5S,6S)-2-[(6,7-dihydro-5H-pyrazolo [1,2-a][1,2,4]triazolium-6-yl)]thio-6-[(R)-1-hydroxyethyl]-1-methyl-carbapenem-3-carboxylate and starting materials thereofUS5286856A1991-09-201994-02-15Takeda Chemical Industries, Ltd.Production of crystalline penemWO2002057266A1 *2001-01-162002-07-25Merck & Co., Inc.Improved process for carbapenem synthesisWO2009047604A1 *2007-10-082009-04-16Orchid Chemicals & Pharmaceuticals LimitedProcess for the preparation of carbapenem antibioticCN102268025A *2011-07-152011-12-07海南美兰史克制药有限公司一种比阿培南化合物及其制法
References
- ^ Aldridge KE, Morice N, Schiro DD (April 1994). “In vitro activity of biapenem (L-627), a new carbapenem, against anaerobes”. Antimicrob. Agents Chemother. 38 (4): 889–93. doi:10.1128/aac.38.4.889. PMC 284564. PMID 8031067.
External links
- (in Japanese) Omegacin
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | IV |
| ATC code | J01DH05 (WHO) |
| Legal status | |
| Legal status | In general: ℞ (Prescription only) |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 120410-24-4 |
| PubChem CID | 71339 |
| ChemSpider | 64442 |
| UNII | YR5U3L9ZH1 |
| ChEBI | CHEBI:3089 |
| ChEMBL | ChEMBL285347 |
| CompTox Dashboard (EPA) | DTXSID5046435 |
| Chemical and physical data | |
| Formula | C15H18N4O4S |
| Molar mass | 350.39 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI | |
| (verify) |
ClinicalTrials.gov
| CTID | Title | Phase | Status | Date |
|---|---|---|---|---|
| NCT04552444 | Clinical Efficacy of Combination Therapy Based on High-dose Biapenem in CRKP Infections | Recruiting | 2020-09-17 | |
| NCT01772836 | Safety Study of Intravenous Biapenem (RPX2003) and RPX7009 Given Alone and in Combination | Phase 1 | Completed | 2013-07-11 |
| NCT01702649 | Safety, Tolerability, Pharmacokinetics of Intravenous RPX2003 (Biapenem) in Healthy Adult Subjects | Phase 1 | Completed | 2012-12-03 |
NIPH Clinical Trials Search of Japan
| CTID | Title | Phase | Status | Date |
|---|---|---|---|---|
| UMIN000017219 | Feasibility and efficacy of the de-escalation therapy by Biapenem for postoperative bacterial pneumonia. | None | Recruiting | 2015-04-22 |
| UMIN000003964 | Clinical evaluation of Biapenem 0.3g, three times daily dosing in eldery patients with pneumonia (moderate and severe infection) | Not applicable | Complete: follow-up complete | 2010-07-29 |
/////////BIAPENEM, TL8000539, UNII:YR5U3L9ZH1, UNII-YR5U3L9ZH1, биапенем, بيابينام ,比阿培南 , Biapenern, CL 186-815, CL 186815, L 627, LJC 10627, Omegacin, Antibacterial, Antibiotics, Lactams, Carbapenems, ind 2021, india 2021, approvals 2021
CC1C2C(C(=O)N2C(=C1SC3CN4C=NC=[N+]4C3)C(=O)[O-])C(C)O
https://clinicaltrials.gov/search/intervention=Biapenem

updated
Biapenem is chemically known as 6-[[2(4R,5S,6S)-carboxy-6-[(lR)-hydroxy ethyl] -4-methyl-7-oxo- 1 -azabicyclo [3.2.0]hept-2-en-3-yljthio] 6,7-dihydro-5H-pyrazolo[1,2-a][1,2,4]triazol-4-ium inner salt, and is represented by Formula 1. It is indicated for the treatment of bacterial infection and sepsis.
Formula 1
U.S. Patent No. 4,866,171, in Example 6, discloses the purification of biapenem using chromatography and/or lyophilization techniques. This patent also describes a process for the conversion of amorphous biapenem into a crystalline form by dissolving the amorphous biapenem in water while heating, followed by cooling, then washing the obtained crystals with a 50% aqueous ethanol solution.
U.S. Patent No. 5,241,073 describes a process for the purification of biapenem involving column chromatography and crystallization with ethanol.
U.S. Patent No. 5,286,856 describes a process for the crystallization of biapenem from an aqueous solution, comprising maintaining the temperature of the aqueous solution from eutectic temperature (-10°C to -2°C) to a temperature lower than 0°C, followed by lyophilization.
The Journal of Organic Chemistry, 63(23):8145-8149 (1998) describes the purification of biapenem involving resin chromatography.
The present invention provides an alternate process for the purification of biapenem that avoids making use of tedious techniques like chromatography and lyophilization. At the same time, it results in a high yield and high purity of the final product. Advantageously, the crystalline biapenem of this invention can be directly isolated from the reaction mixture. Further, the process of the present invention involves fewer steps, is easily scalable, and industrially advantageous.
EXAMPLES
Example 1 : Purification of Biapenem
Biapenem (12 g) was added into water (300 mL) at 65°C, stirred for 5 minutes, and cooled to 30°C within 10 minutes. Enoantichromos carbon (0.6 g) was added to the reaction mixture and stirred for 10 minutes to 15 minutes at 25°C to 30°C. The reaction mixture was filtered through a hyflo bed and washed with water (36 mL). The filtrate obtained was passed through a 0.45 micron filter, and its pH was adjusted to 5.5 using 5% aqueous sodium hydroxide solution at 10°C to 15°C. Acetone (336 mL) was added to the reaction mixture at 5°C to 10°C. The resultant slurry was stirred for 3 hours at 5°C to 10°C, filtered, and the obtained solid was washed with acetone (60 mL). The solid was dried under reduced pressure (720 mmHg) at 30°C to 35°C to obtain the title product as white crystals.
Yield: 84%
HPLC Purity: 99.87%
Example 2: Purification of Biapenem
Biapenem (18 g) was added into water (450 mL) at 65°C, stirred for 5 minutes, and cooled to 30°C within 10 minutes. Enoantichromos carbon (0.9 g) was added to the reaction mixture and stirred for 30 minutes at 25°C to 30°C. The reaction mixture was filtered through a hyflo bed and washed with water (54 mL). The filtrate obtained was passed through a 0.45 micron filter and its pH was adjusted to 4.9 using 5% aqueous sodium hydroxide solution at 10°C to 15°C. Acetone (504 mL) was added to the reaction mixture at 10°C to 15°C. The resultant slurry was stirred for 3 hours at 5°C to 10°C, filtered, and the obtained solid was washed with acetone (90 mL). The solid was dried under reduced pressure (720 mmHg) at 35°C to 40°C to obtain the title product as white crystals.
Yield: 81.77%
HPLC Purity: 99.80%
PATENT
Background of the Invention Biapenem is a synthetic broad-spectrum carbapenem antibiotic which suppresses bacterial growth by inhibiting the enzymes responsible for bacterial cell wall synthesis, and shows broad-spectrum antibacterial activity both against gram-positive bacteria and gram-negative bacteria. Biapenem is chemically known as (4R,5S,6S)-3-(6,7-dihydro-5H-pyrazolo[l,2-a][ 1,2,4] triazol-8-ium-6-ylsulfanyl)-6-( 1 -hydroxyethyl)-4-methyl-7-oxo-1 -azabicyclo [3.2.0]hept-2-ene-2-carboxylate and marketed in Japan as OMEGACIN®.Various methods are reported in the prior art for the preparation of Biapenem of formula (I) which includes the condensation of compound of formula (II) with compound of formula (III) and subsequent deprotection of the protecting group as shown in scheme-1. wherein R1 is hydrogen or hydroxy protecting group such as tert-butyl dimethyl silyl and the like, R2 is hydrogen or carboxyl protecting group such as p-nitrobenzyl, p-methoxy benzyl, allyl and the like, A is an activating group such as P(0)(OR)2, SO2R and the like wherein R is selected from substituted or unsubstituted C1-6 alkyl, aralkyl or aryl to form the compound of formula (II). The X” in compound of formula (III) is halogen selected from Br or CI.Biapenem was first disclosed in US 4,866,171 and the said patent also discloses a process for the preparation of the same. US 5,241,073 disclosed the method for the preparation of compound of formula (III) followed by condensation with compound of general formula (II) using base such as N-ethyldiisopropylamine and subsequent deprotection yields Biapenem which was isolated by column chromatography followed by crystallization from ethanol.EP 0289801 discloses a process for the preparation of crystalline Biapenem wherein Biapenem was dissolved in water and lyophilized to get amorphous compound. The amorphous compound was dissolved in water at 40° C followed by cooling to get crystalline product. This patent further provides the PXRD values of the crystalline Biapenem. The Biapenem obtained according to the process provided in this patent takes longer time for reconstitution and hence not suitable.US 5,286,856 and US 5,424,069 provide a process for the crystallization of Biapenem which utilizes freeze-drying technique and vial lyophillisation method respectively. These patents disclose (refer para 1, lines 10-33 of US’ 856) that the process provided in EP 0289801 results with Biapenem crystals which take relatively longer time for dissolution during use. To overcome the above issues, these patents utilize the freeze-drying and vial lyophillisation methods. The said methods involve freezing of the solution containing Biapenem followed by raising the temperature and repeating the cooling and heating process followed by lyophillisation to get the crystalline product. Lyophillisation and related process are capital intensive techniques and uneconomical in commercial scale operations.All the above said prior arts utilize either the lyophillisation technique or preparing the amorphous material and crystallizing it from water to get crystalline Biapenem.Biapenem is available as powder for injection which needs to be reconstituted with water or saline solution before injection. The process of preparing a solution having an appropriate concentration of an active ingredient for the administration is called “reconstitution”. The reconstitution time (RCT) plays a critical role in injectable powders. Short reconstitution time is preferable for both a member of medical center and patients. If the reconstitution time is too long, it will increase the preparation time thus making it difficult to administrate it to many patients at the same, which will eventually lower the competitiveness of the drug. The problem before the applicants is to find economic and robust process for the preparation of Biapenem with high purity and yield which should dissolve in water in less than 25 seconds (reconstitution time). With our continued intensive and diligent research for developing a process for the preparation of Biapenem having high purity and yield with reconstitution time of less than 25 seconds, we have identified an improved process which is commercially viable and eliminates the issues associated with reconstitution time. The process of this invention is simple and obviates the use of freeze crystallization. Further the present invention fulfils the need for a process for the manufacture of Biapenem which is convenient to operate in commercial scale
Objectives of the inventionThe main objective of the present invention is to provide a simple and commercially viable, industrially scalable process for the crystallization of Biapenem of formula (I) with high purity and good yield.Yet another objective of the present invention is to provide a simple and commercially suitable process for the preparation of Biapenem of formula (I) with reconstitution time less than 25 seconds. The reconstitution time is calculated by the time taken to dissolve 300 mg of Biapenem in 100 ml of water or saline solution.Summary of the inventionAccordingly the primary aspect of the present invention is to provide an improved process for the preparation of Biapenem of formula (I) the said process comprises;(i) obtaining a solution of Biapenem in water containing co-solvent; and(ii) adding anti-solvent in to the solution of step (i) or vice-versa to crystallize Biapenem followed by filtration. Detailed Description In an embodiment of the present invention, the co-solvent used in step (i) is selected from alcoholic solvents consisting of methanol, ethanol, isopropyl alcohol, n-propanol, n-butanol and iso-butanol or mixtures thereof; preferably methanol, ethanol and isopropyl alcohol; more preferably methanol.In another embodiment of the present invention the anti-solvent used in step (ii) is selected from acetone, methyl ethyl ketone, methyl isobutyl ketone, ethyl acetate, methyl acetate, butyl acetate, tetrahydrofuran or mixtures thereof; preferably acetone. In yet another embodiment of the present invention, the solution of Biapenem in step (i) can be obtained by (a) dissolving Biapenem in water followed by addition of co-solvent (b) dissolving Biapenem in water containing the co-solvent (c) the aqueous solution containing Biapenem can be obtained directly from the reaction mass followed by addition of co-solvent (d) the aqueous solution of Biapenem containing co-solvent can be obtained directly from the reaction mass. The said solutions, if necessary can be subjected to sterile filtration before the addition of anti-solvent. Thus the present invention provided a process for the preparation of sterile Biapenem having reconstitution time less than 25 seconds, more preferably less than 15 seconds.The prior art lyophillisation process for the preparation of Biapenem requires capital investment and high operating cost due to the involvement of repetitive heating and cooling process which is tedious technology in commercial scale operations. The reported prior art process for the crystallization of Biapenem of formula (I) from water results in the formation of crystalline powder which takes longer time for dissolution in water or saline solution (reconstitution time). Surprisingly, applicant found that the use of co-solvents during the crystallization of Biapenem results with Biapenem having reconstitution time of less than 25 seconds. This constitutes the novelty of the present invention.In this present invention the Biapenem of formula (I) is obtained as crystalline solid with purity above 99.0 % by HPLC with good stability and further can be easily filled in vials.
The following examples are provided by way of illustration only and should not be construed to limit the scope of the invention.
Crystallization of (4R,5S,6S)-3-(6,7-dihvdro-5H-pyrazolo[l,2-al 11,2,41 triazol-8-ium-6-vlsulfanvl)-6-(l-hydroxvethvl)-4-methvl-7-oxo-l-azabicyclo [3.2.01hept-2-ene-2-carboxvlate [Biapenem of formula (1)1:Example -1:To water (4 lit), Biapenem (100 g) was added at 40° C and dissolved to get a clear solution. Activated carbon and EDTA were added to the clear solution and filtered through hi-flow bed, washed with water followed by filtration through micron filters in sterile area. To the filtrate, methanol (600 mL) was added followed by acetone under stirring. To the reaction mass, Biapenem seed material was added and stirred. The crystallized product was filtered, washed with aqueous acetone and dried under vacuum to get crystalline Biapenem.Yield: 85 g Purity by HPLC: 99.5% Reconstitution time (RCT): < 15 seconds
Example -2:To water (4 lit), Biapenem (100 g) was added at 40° C and dissolved to get a clear solution. To the filtrate, isopropyl alcohol (500 ml) was added followed by acetone under stirring. The mass was cooled and stirred. The crystallized product was filtered, washed with aqueous acetone and dried under vacuum to get crystalline Biapenem.Yield: 83 g Purity by HPLC: 99.6% Reconstitution time: < 15 seconds
Example -3;To water (4 lit), Biapenem (100 g) was added at 40° C and dissolved to get a clear solution. The solution was filtered through micron filters. To the filtrate, ethanol (600 ml) was added followed by acetone and stirred. The crystallized product was filtered, washed with aqueous acetone and dried under vacuum to get crystalline Biapenem.Yield: 84 g Purity by HPLC: 99.5% Reconstitution time : < 15 seconds
Example -4:To water (4 lit), Biapenem (100 g) was added at 40° C and dissolved to get a clear solution. The solution was filtered through hi-flow bed, washed with water followed by filtration through micron filters. To the filtrate, methanol (450 ml) was added followed by acetone and stirred. The crystallized product was filtered, washed with aqueous acetone and dried under vacuum to get crystalline Biapenem. Yield: 87 g Purity by HPLC: 99.4% Reconstitution time (RCT): < 15 seconds
Reference example -1:Preparation of Biapenem (Non-Sterile)Step-I: Preparation of p-Nitrobenzyl (4R,5S,6S)-3-(6,7-dihydro-5H-pvrazolofl,2-al[l,2,41triazol-8-ium-6-vlsulfanvn-6-(l-hvdroxvethyl)-4-methvI-7-oxo-l-azabicvclo[3.2.01hept-2-ene-2-carboxylate [Compound of formula (IV)1To a mixture of acetonitrile and DMF, P-Nitrobenzyl (4R,5S,6S)-3-(dipheny loxy)phosphory loxy-6- [(1R)-1 -hydroxyethy 1] -4-methy 1-7-oxo-1 -azabicyclo[3,2,0]hept-2-ene-2-carboxylate (compound of formula II) and 6,7-dihydro-6-mercapto-5H-pyrazolo[l,2-a] [1,2,4] triazole chloride (compound of formula III) were added and cooled to 0-5° C. To this mixture, N-ethyldiisopropyl amine was added and stirred till the completion of the reaction, followed by the addition of dichloromethane to crystallize the p-Nitrobenzyl (4R,5S,6S)-3-(6,7-dihydro-5H-pyrazolo[l,2-a][l,2,4]triazol-8-ium-6-ylsulfanyl)-6-(l -hydroxyethyl)-4-methyl-7-oxo-1 -azabicyclo[3.2.0] hept-2-ene-2-carboxylate which was filtered and dried under nitrogen.
Step-II: Preparation of BiapenemTo a solution of MOPS buffer and THF, p-Nitrobenzyl (4R,5S,6S)-3-(6,7-dihydro-5H-pyrazolo[l,2-a][l,2,4]triazol-8-ium-6-ylsulfanyl)-6-(l-hydroxy ethyl)-4-methyl-7-oxo-l-azabicyclo[3.2.0]hept-2-ene-2-carboxylate (Compound of formula-IV) was added at pH 7-8 and cooled to 5-10° C. The mixture was hydrogenated using palladium on carbon as catalyst. The catalyst was filtered and the filtrate was treated with activated carbon and filtered. The filtrate was extracted with dichloromethane and the layers separated. The aqueous layer was degassed. To the aqueous layer, acetone was added to crystallize Biapenem at 20-25° C. The product was filtered, washed with aqueous acetone and dried under vacuum to get Biapenem (Non-Sterile).
Reference example -2: Crystallization of Biapenem
Example -1 was repeated without the addition of methanol.Yield: 84 g Purity by HPLC: 99.5%Reconstitution time : > 90 secondsThe reconstitution time is calculated by the time taken to dissolve 300 mg of Biapenem in 100 ml of water or saline solution.Table-1: Comparative Data:The comparative data provided in the table-1 clearly indicates that the addition of co-solvent during crystallization provides Biapenem with reconstitution time less than 25 seconds.
CLARITHROMYCIN

Clarithromycin
Synonyms:A-56268, TE-031, 6-O-methylerythromycin, ATC:J01FA09Use:macrolide antibioticChemical name:6-O-methylerythromycinFormula:C38H69NO13
- MW:747.96 g/mol
- CAS-RN:81103-11-9
- 81103-11-9
klacid XL / Klaricid XL / Macladin / Naxy / Veclam / Zeclar
(3R,4S,5S,6R,7R,9R,11R,12R,13S,14R)-6-{[(2S,3R,4S,6R)-4-(dimethylamino)-3-hydroxy-6-methyloxan-2-yl]oxy}-14-ethyl-12,13-dihydroxy-4-{[(2R,4R,5S,6S)-5-hydroxy-4-methoxy-4,6-dimethyloxan-2-yl]oxy}-7-methoxy-3,5,7,9,11,13-hexamethyl-1-oxacyclotetradecane-2,10-dione
Synthesis Reference
Jih-Hua Liu, David A. Riley, “Preparation of crystal form II of clarithromycin.” U.S. Patent US5844105, issued May, 1997. US5844105
NEW DRUG APPROVALS
ONE TIME
$10.00
Product Ingredients
| INGREDIENT | UNII | CAS | INCHI KEY |
|---|---|---|---|
| Clarithromycin citrate | 16K08R7NG0 | 848130-51-8 | MDRWXDRMSKEMRE-AZFLODHXSA-N |
ClarithromycinCAS Registry Number: 81103-11-9CAS Name: 6-O-MethylerythromycinManufacturers’ Codes: A-56268; TE-031Trademarks: Biaxin (Abbott); Clarosip (Grñenthal); Clathromycin (Taisho); Cyllind (Abbott); Klacid (Abbott); Klaricid (Abbott); Macladin (Guidotti); Naxy (Sanofi Winthrop); Veclam (Zambon); Zeclar (Abbott)Molecular Formula: C38H69NO13Molecular Weight: 747.95Percent Composition: C 61.02%, H 9.30%, N 1.87%, O 27.81%Literature References: Semisynthetic macrolide antibiotic; derivative of erythromycin, q.v. Prepn: Y. Watanabe et al.,EP41355; eidem,US4331803 (1981, 1982 both to Taisho); and in vitro antibacterial activity: S. Morimoto et al.,J. Antibiot.37, 187 (1984). In vitro and in vivo antibacterial activity: P. B. Fernandes et al.,Antimicrob. Agents Chemother.30, 865 (1986). Comparative antibacterial spectrum in vitro: C. Benson et al.,Eur. J. Clin. Microbiol.6, 173 (1987); H. M. Wexler, S. M. Finegold, ibid. 492. HPLC determn in biological fluids: D. Croteau et al.,J. Chromatogr.419, 205 (1987); in plasma: H. Amini, A. Ahmadiani, J. Chromatogr. B817, 193 (2005). Acute toxicity study: S. Abe et al.,Chemotherapy (Tokyo)36, Suppl. 3, 274 (1988). Symposium on pharmacology and comparative clinical studies: J. Antimicrob. Chemother.27, Suppl. A, 1-124 (1991). Comprehensive description: I. I. Salem, Anal. Profiles Drug Subs. Excip.24, 45-85, (1996).Properties: Colorless needles from chloroform + diisopropyl ether (1:2), mp 217-220° (dec). Also reported as crystals from ethanol, mp 222-225° (Morimoto). uv max (CHCl3): 288 nm (e 27.9). uv max (CHCl3): 240, 288 nm; (methanol): 211, 288 nm. [a]D24 -90.4° (c = 1 in CHCl3). Stable at acidic pH. LD50 in male, female mice, male, female rats (mg/kg): 2740, 2700, 3470, 2700 orally, 1030, 850, 669, 753 i.p., >5000 all s.c. (Abe).Melting point: mp 217-220° (dec); mp 222-225° (Morimoto)Optical Rotation: [a]D24 -90.4° (c = 1 in CHCl3)Absorption maximum: uv max (CHCl3): 288 nm (e 27.9). uv max (CHCl3): 240, 288 nmToxicity data: LD50 in male, female mice, male, female rats (mg/kg): 2740, 2700, 3470, 2700 orally, 1030, 850, 669, 753 i.p., >5000 all s.c. (Abe)Therap-Cat: Antibacterial.Keywords: Antibacterial (Antibiotics); Macrolides.
Clarithromycin, a semisynthetic macrolide antibiotic derived from erythromycin, inhibits bacterial protein synthesis by binding to the bacterial 50S ribosomal subunit. Binding inhibits peptidyl transferase activity and interferes with amino acid translocation during the translation and protein assembly process. Clarithromycin may be bacteriostatic or bactericidal depending on the organism and drug concentration.
Clarithromycin, sold under the brand name Biaxin among others, is an antibiotic used to treat various bacterial infections.[2] This includes strep throat, pneumonia, skin infections, H. pylori infection, and Lyme disease, among others.[2] Clarithromycin can be taken by mouth as a pill or liquid.[2]
Common side effects include nausea, vomiting, headaches, and diarrhea.[2] Severe allergic reactions are rare.[2] Liver problems have been reported.[2] It may cause harm if taken during pregnancy.[2] It is in the macrolide class and works by decreasing protein production of some bacteria.[2]
Clarithromycin was developed in 1980 and approved for medical use in 1990.[3][4] It is on the World Health Organization’s List of Essential Medicines, the safest and most effective medicines needed in a health system.[5] Clarithromycin is available as a generic medication.[2] It is made from erythromycin and is chemically known as 6-O-methylerythromycin.[6]
Medical uses
Clarithromycin is primarily used to treat a number of bacterial infections including pneumonia, Helicobacter pylori, and as an alternative to penicillin in strep throat.[2] Other uses include cat scratch disease and other infections due to bartonella, cryptosporidiosis, as a second line agent in Lyme disease and toxoplasmosis.[2] It may also be used to prevent bacterial endocarditis in those who cannot take penicillin.[2] It is effective against upper and lower respiratory tract infections, skin and soft tissue infections and helicobacter pylori infections associated with duodenal ulcers.
Spectrum of bacterial susceptibility
Staphylococcus aureusAerobic Gram-positive bacteria
Aerobic Gram-negative bacteria
- Haemophilus parainfluenzae
- Haemophilus influenzae
- Moraxella catarrhalis
Helicobacter
Mycobacteria
Mycobacterium avium complex consisting of:
Other bacteria
Safety and effectiveness of clarithromycin in treating clinical infections due to the following bacteria have not been established in adequate and well-controlled clinical trials:[7]
Aerobic Gram-positive bacteria
- Streptococcus agalactiae
- Streptococcus (Groups C, F, G)
- Viridans group streptococci
Aerobic Gram-negative bacteria
Anaerobic Gram-positive bacteria
- Clostridium perfringens
- Peptococcus Niger
- Cutibacterium acnes
Anaerobic Gram-negative bacteria
- Prevotella melaninogenica (formerly Bacteroides melaninogenicus)
Contraindications
- Clarithromycin should not be taken by people who are allergic to other macrolides or inactive ingredients in the tablets, including microcrystalline cellulose, croscarmelose sodium, magnesium stearate, and povidone
- Clarithromycin should not be used by people with a history of cholestatic jaundice and/or liver dysfunction associated with prior clarithromycin use.[7]
- Clarithromycin should not be used in the setting of hypokalaemia (low blood potassium)
- Use of clarithromycin with the following medications: cisapride, pimozide, astemizole, terfenadine, ergotamine, ticagrelor, ranolazine or dihydroergotamine is not recommended.[7]
- It should not be used with colchicine in people with kidney or liver impairment.[7]
- Concomitant use with cholesterol medications such as lovastatin or simvastatin.[7]
- Hypersensitivity to clarithromycin or any component of the product, erythromycin, or any macrolide antibiotics.[7]
- QT prolongation or ventricular cardiac arrhythmias, including torsade de pointes.[7]
Side effects
The most common side effects are gastrointestinal: diarrhea (3%), nausea (3%), abdominal pain (3%), and vomiting (6%). It also can cause headaches, insomnia, and abnormal liver function tests. Allergic reactions include rashes and anaphylaxis. Less common side effects (<1%) include extreme irritability, hallucinations (auditory and visual), dizziness/motion sickness, and alteration in senses of smell and taste, including a metallic taste. Dry mouth, panic attacks, and nightmares have also been reported, albeit less frequently.[8]
Cardiac
In February 2018, the FDA issued a Safety Communication warning with respect to an increased risk for heart problems or death with the use of clarithromycin, and has recommended that alternative antibiotics be considered in those with heart disease.[9]
Clarithromycin can lead to a prolonged QT interval. In patients with long QT syndrome, cardiac disease, or patients taking other QT-prolonging medications, this can increase risk for life-threatening arrhythmias.[10]
In one trial, the use of short-term clarithromycin treatment was correlated with an increased incidence of deaths classified as sudden cardiac deaths in stable coronary heart disease patients not using statins.[11] Some case reports suspect it of causing liver disease.[12]
Liver and kidney
Clarithromycin has been known to cause jaundice, cirrhosis, and kidney problems, including kidney failure.[citation needed]
Central nervous system
Common adverse effects of clarithromycin in the central nervous system include dizziness, headaches. Rarely, it can cause ototoxicity, delirium and mania.
Infection
A risk of oral candidiasis and vaginal candidiasis, due to the elimination of the yeast’s natural bacterial competitors by the antibiotic, has also been noted.
Pregnancy and breastfeeding
Clarithromycin should not be used in pregnant women except in situations where no alternative therapy is appropriate.[7] Clarithromycin can cause potential hazard to the fetus hence should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.[7] For lactating mothers it is not known whether clarithromycin is excreted in human milk.[7]
Interactions
Clarithromycin inhibits a liver enzyme, CYP3A4, involved in the metabolism of many other commonly prescribed drugs. Taking clarithromycin with other medications that are metabolized by CYP3A4 may lead to unexpected increases or decreases in drug levels.
A few of the common interactions are listed below.
Colchicine
Clarithromycin has been observed to have a dangerous interaction with colchicine as the result of inhibition of CYP3A4 metabolism and P-glycoprotein transport. Combining these two drugs may lead to fatal colchicine toxicity, particularly in people with chronic kidney disease.[7]
Statins
Taking clarithromycin concurrently with certain statins (a class of drugs used to reduce blood serum cholesterol levels) increases the risk of side effects, such as muscle aches and muscle break down (rhabdomyolysis).[13]
Calcium channel blockers
Concurrent therapy with calcium channel blocker may increase risk of low blood pressure, kidney failure, and death, compared to pairing calcium channel blockers with azithromycin, a drug similar to clarithromycin but without CYP3A4 inhibition.[14] Administration of clarithromycin in combination with verapamil have been observed to cause low blood pressure, low heart rate, and lactic acidosis.[7]
Carbamazepine
Clarithromycin may double the level of carbamazepine in the body by reducing its clearance, which may lead to toxic symptoms of carbamazepine, such as double vision, loss of voluntary body movement, nausea, as well as hyponatremia.[15]
HIV medications
Depending on the combination of medications, clarithromycin therapy could be contraindicated, require changing doses of some medications, or be acceptable without dose adjustments.[16] For example, clarithromycin may lead to decreased zidovudine concentrations.[17]
Mechanism of action
Clarithromycin prevents bacteria from multiplying by acting as a protein synthesis inhibitor. It binds to 23S rRNA, a component of the 50S subunit of the bacterial ribosome, thus inhibiting the translation of peptides.[citation needed]
Pharmacokinetics
MetabolismUnlike erythromycin, clarithromycin is acid-stable, so can be taken orally without having to be protected from gastric acids. It is readily absorbed, and diffuses into most tissues and phagocytes. Due to the high concentration in phagocytes, clarithromycin is actively transported to the site of infection. During active phagocytosis, large concentrations of clarithromycin are released; its concentration in the tissues can be over 10 times higher than in plasma. Highest concentrations are found in liver, lung tissue, and stool.
Clarithromycin has a fairly rapid first-pass metabolism in the liver. Its major metabolites include an inactive metabolite, N-desmethylclarithromycin, and an active metabolite, 14-(R)-hydroxyclarithromycin. Compared to clarithromycin, 14-(R)-hydroxyclarithromycin is less potent against mycobacterial tuberculosis and the Mycobacterium avium complex. Clarithromycin (20%-40%) and its active metabolite (10%-15%) are excreted in urine. Of all the drugs in its class, clarithromycin has the best bioavailability at 50%, which makes it amenable to oral administration. Its elimination half-life is about 3 to 4 hours with 250 mg administered every 12 h, but increased to 5 to 7 h with 500 mg administered every 8 to 12 h. With any of these dosing regimens, the steady-state concentration of this metabolite is generally attained within 3 to 4 days.[18]
History
Clarithromycin was invented by researchers at the Japanese drug company Taisho Pharmaceutical in 1980.[3] The product emerged through efforts to develop a version of the antibiotic erythromycin that did not experience acid instability in the digestive tract, causing side effects, such as nausea and stomachache. Taisho filed for patent protection for the drug around 1980 and subsequently introduced a branded version of its drug, called Clarith, to the Japanese market in 1991. In 1985, Taisho partnered with the American company Abbott Laboratories for the international rights, and Abbott also gained FDA approval for Biaxin in October 1991. The drug went generic in Europe in 2004 and in the US in mid-2005.
Society and culture

A pack of Clarithromycin tablets manufactured by Taisho Pharmaceutical
Available forms
Clarithromycin is available as a generic medication.[2] In the United States, clarithromycin is available as immediate release tablets, extended release tablets, and granules for oral suspension.[2]
Brand names
Clarithromycin is available under several brand names in many different countries, for example Biaxin, Crixan, Claritron, Clarihexal, Clacid, Claritt, Clacee, Clarac, Clariwin, Claripen, Clarem, Claridar, Cloff, Fromilid, Infex, Kalixocin, Karicin, Klaricid, Klaridex, Klacid, Klaram, Klabax, MegaKlar, Monoclar, Resclar, Rithmo, Truclar, Vikrol and Zeclar.
Manufacturers
In the UK the drug product is manufactured in generic form by a number of manufacturers including Somex Pharma, Ranbaxy, Aptil and Sandoz.
SYN
CN 109705180

SYN
Indian Pat. Appl., 2014DE00731, 31 Aug 2016

SYN
Heterocycles, 31(12), 2121-4; 1990

SYN
https://patents.google.com/patent/WO2006064299A1/enErythromycin A is known to be a useful macrolide antibiotic having a strong activity against Gram-positive bacteria, this compound has an undesirable property that it loses rapidly the antibacterial activity by the acid in stomach when administered orally, where- upon its blood concentration remains at a low level. 6-0-Alkyl derivatives of Erythromycin- A are well known as an useful antibacterial agents. 6-O-Methyl-Erythromycin-A (Clarithromycin) and a pharmaceutically acceptable salt is a potent macrolide antibiotic as reported in US Patent No. 4,331 ,803. Clarithromycin is stable in acidic medium and also remarkable in vivo activity and has a strong antibacterial property against Gram-positive bacteria compared to Erythromycin- A. This compound shows excellent effect for the treatment of infections by oral administration.A number of synthetic processes have been reported for preparing 6-O-alkyl erythromycin. US Patent No. 4,331 ,803 discloses a method for the preparation of Clarithromycin by methylating 6-OH group of 2′-O-3′-N-benzyloxycarbonyl erythromycinFormula (III)

21,3′-O-Protected ErythromycinMethylation of 6-OH group of the 2′,3′-benzyloxycarbonyl erythromycin was carried out using methyl iodide in the presence of a suitable base in a solvent. Clarithromycin was obtained from the compound after removing benzyloxycarbonyl group by hydrogenolysis and then subjecting to the reductive methylation in the presence of excess amount of farmaldehyde. Clarithromycin can also be synthesized by the methylation of 6-OH position of Erythromycin-A-9-OximeFormula (II)

Erythromycin-9-OximeSynthesis of Clarithromycin using 9-oxime or its derivatives are well reported in US Patent Nos. 5,274,085; 4,680,386; 4,668,776; 4,670,549 and 4,672,109. In case of Erythromycin-9-Oxime derivatives, the oxime is protected before methylation step with 2- alkenyl group (US Patent Nos. 4,670,549; 4,668,776) or benzyl group (US Patent Nos. 4,680,386 and 4,670,549). However, it has been reported (Ref. Journal of Antibiotics 46, No. 6, Page No. 647, year 1993) that when the Erythromycin-A-9-Oxime is protected by trimethylsilyl group, which is very unstable under basic condition pose potential impurities formation during methylation. There are some methods reported in US Patent Nos., e.g. , 4,680,386; 4,670,549 and US Patent No. 4,311,803 for the synthesis of Clarithromycin by using chlorobenzyloxycarbonyl group for protection at 2′ and 3′ function of of Erythromycin-A-9-Oxime derivatives.For the protection of 2′-OH group (US Patent No. 4,311 ,803) requires large amounts of benzyl chloroformate which poses problems in handling because of its severe irritating and toxic properties. This protection step also leads to the formation of 3′ -N- demethylation, which requires an additional re-methylation step. The de-protection of chlorobenzyloxy carbonyl group leads to the formation of undesired side products. In earlier reported processes, e.g. , US Patent No. 4,990,602; EP 0,272,110 Al where the methylation has been done on Erythromycin-A-9-Oxime derivatives by the protection of 2′ and 4″ hydroxyl groups using DMSO and THF as a solvent at 0° to 50C or at room temperature, smooth methylation takes place with less side product formation. However, by using the above methylation processes the formation of 6, 11-O-dimethyl erythromycin- A (Compound- A) is always more than 1.0 % in Clarithromycin. Hence, there is a need for an efficient methylation process for the production of Clarithromycin with lesser amount of 6,11-O-dimethyl erythromycin-A than reported previously.




EXAMPLE 1Erythromycin-A-9-OximeTo a solution of 201 Ltr water in 561 Kg isopropyl alcohol is added 282 Kg (4057 mol) of hydroxyl amine hydrochloride under stirring and the reaction mixture is brought to 10 to 200C. Caustic flakes (162 Kg, 4050 mol) is added slowly to the reaction mixture by keeping temperature between 10° to 200C. After 15 minutes of completion of addition, pH of reaction mixture is adjusted to 6.5 to 7.0 by the slow addition of glacial acetic acid (96 Ltr, 100.8 Kg, 1678.6 mole). To the stirred reaction mass is added 300 Kg (408.8 mole) of Erythromycin-A base and reaction mixture is stirred at 55° C for 28 hours. After completion of the reaction, mixture is brought to ambient temperature and to it a mixture of ammonia solution (270 Kg) and water (600 Ltr) is added within 1 hour followed by 3000 Ltr of fresh water in next two hours and stirred the reaction mass for further 1 hour. White solid product obtained is centrifuged, wet cake is washed with water and dried at 6O0C for 12 hours to give 270 Kg of erythromycin-A Oxime. Melting point = 156° to 158°C.EXAMPLE 22′,4″-O-Bis(trimethylsilyl)-erythro?nycin-A-9[O-(l-methoxy-l-methyl ethyl)oximeTo a solution of 80 Kg (106.8 mole) of Erythromycin-A-9-Oxime in 400 Ltr of dichloromethane is added 38.50 Kg (534 mole) of 2-methoxy propene at 100C temperature 19.25 Kg (166.6 mole) of pyridine hydrochloride is added under stirring and the reaction mixture is stirred at 8 to 12° C for 6 hours then to it is added 19.30 Kg (119.5 mole) of HMDS and stirring is continued for 12 to 15 hours at 15° to 18°C temperature. After completion of reaction, 400 Ltr of saturated aqueous sodium carbonate solution is added and the mixture is stirred thoroughly at room temperature. Aqueous layer is further extracted with fresh DCM (100 Ltr). Both DCM extracts are mixed together and washed with water (200 Ltr) followed by brine solution (200 Ltr). The solvent is evaporated under reduced pressure. To the obtained crude solid mass is charged isopropyl alcohol (240 Ltr) and distilled out 80 Ltr of isopropyl alcohol. To the reaction mixture 160 Ltr of water is charged and stirring continued at room temperature for 1 hour. Solid crystalline product obtained is centrifuged and dried at 60° to 650C for 8 hours under vacuum to give 85 Kg of title compound. Melting point = 125° to 126°C. HPLC Purity = More than 90 % .EXAMPLE 3Clarithromycin-9- OximeTo a solution of 80 Kg (82.98 mole) of 2′,4″-O-bis(trimethylsilyl)-erythromycin-A- 9-[O-(l-methoxy methyl ethyl)Oxime] in 1200 Ltr of a mixture of dimethyl sulfoxide and diethylether (1 : 1) are added methyl iodide (20.62 Kg, 145.2 mole) and 6.48 Kg (98.35 mole) of 85 % potassium hydroxide powder and the reaction mixture is stirred for 90 minutes at room temperature. To the reaction mass is added 53 Kg of 40 % dimethylamine solution and stirring is continued for 1 hour diethylether layer is separated and DMSO layer is further extracted with fresh diethylether (200 Ltr). Combined ether layer is washed with water and concentrated in vacuum. To the obtained semi solid mass 330 Ltr of isopropyl alcohol is charged and then distilled out 165 Ltr of isopropyl alcohol. To the obtained slurry 165 Ltr of water and 21.71 Kg formic acid (99%) are added and the mixture is stirred at room temperature for 30 minutes. 622 Ltr of water is added to the reaction mixture and pH is adjusted between 10.5 and 11.5 with 25 % aqueous sodium hydroxide solution. Solid compound obtained is centrifuged and wet cake is kept as such for further reaction on the basis of moisture content. Wet weight = 95 Kg, Moisture Content = 33 %, Dried weight = 62 KgEXAMPLE 46-O-Methyl erythromycin- A (Clarithromycin)62 Kg of 6-O-Methyl erythromycin-9-Oxime is charged into a mixture of 434 Ltr of isopropyl alcohol and water (1: 1) and to it is added 38.80 Kg of sodium metabisulphite (203 mole) and then the mixture is heated to reflux for 6 to 8 hours. To the reaction mixture is charged water (620 Ltr) at ambient temperature and then the mixture is adjusted to pH about 10.5 to 11.5 by adding 25% aqueous sodium hydroxide solution and stirred for further 1 hour. White solid crude product is centrifuged, washed with water (300 Ltr), dried at 65° to 750C for 8 hours to give 40 Kg of crude Clarithromycin which on re- crystallization with chloroform isopropyl alcohol mixture provided 20 Kg of Clarithromycin (Form II).
SYNEP 0041355; US 4331803J Antibiot 1984,37(2),187-189
| EP 0147062 |
The methylation of 2′-O,N-bis(benzyloxycarbonyl)-N-demethylerythromycin A (I) with methyl iodide and KOH or NaHI in DMSO-dimethoxyethane gives the 6-O-methyl derivative (II), which is deprotected by hydrogenation with H2 over Pd/C in ethanol acetic acid affording 6-O-methyl-N-demethylerythromycin A (III). Finally, this compound is methylated with formaldehyde under reductive conditions (H2-Pd/C) in ethanol/acetic acid.
CLIP


2 Clarithromycin. Initial attempts of making clarithromycin (2) from erythromycin (1) by methylation of 8 gave approximately equal amounts of 2 and 10 by methylation at O-6 and O-11, respectively (Scheme 2, route A).[28–30] This allowed 2 to be obtained in approximately 39% yield, but it contained a small impurity of di-O-methylated 9. To improve the yields and obtain 2 in pure form, other alternatives were explored. During methylation of analogues of 8 it was observed that the conformation of the macrocyclic core plays an important role for the regioselectivity of the O-methylation.[31] As oximes are readilyhydrolysed and may have different conformations than ketone 8, oximes 11 and 13 were subjected to methylation. Interestingly, methylation of 13, but not of 11, proved to be highly selective for O-6 and provided 14 in 86% yield (Scheme2 route B); an observation which supports that 13 populates different conformations compared to 8 and 11 under the methylation conditions.[31] Compound 14 was then hydrogenated with Pd/C to deprotect the two benzyloxycarbonyl groups and the 2-chlorobenzyl group. The N-methylamine was then methylated by reductive amination and the oxime was deprotected by hydrolysis to provide clarithromycin (2). This procedure was further modified for process-scale synthesis so that clarithromycin (2) could be obtained in 70% yield starting from oxime 11 without the isolation of any intermediate.[32][28] M. Shigeo, T. Yoko, W. Yoshiaki, O. Sadafumi, J. Antibiot. 1984, 37, 187 – 189. [29] Y. Watanabe, T. Adachi, T. Asaka, M. Kashimura, S. Morimoto, Heterocycles 1990, 31, 2121 – 2124. [30] E. H. Flynn, H. W. Murphy, R. E. McMahon, J. Am. Chem. Soc. 1955, 77, 3104 – 3106. [31] Y. Watanabe, S. Morimoto, T. Adachi, M. Kashimura, T. Asaka, J. Antibiot. 1993, 46, 647 – 660.32] R. A. Dominguez, M. D. C. C. Rodriguez, L. . D. Tejo, R. N. Rib, J. S. Cebrin, J. I. B. Bilbao, 2003, US6642364B2.
References
- ^ https://www.ema.europa.eu/documents/psusa/clarithromycin-list-nationally-authorised-medicinal-products-psusa/00000788/202004_en.pdf
- ^ Jump up to:a b c d e f g h i j k l m n “Clarithromycin”. The American Society of Health-System Pharmacists. Archivedfrom the original on September 3, 2015. Retrieved September 4, 2015.
- ^ Jump up to:a b Greenwood D (2008). Antimicrobial drugs : chronicle of a twentieth century medical triumph (1 ed.). Oxford: Oxford University Press. p. 239. ISBN 9780199534845. Archived from the original on 2016-03-05.
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 498. ISBN 9783527607495.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ Kirst HA (2012). Macrolide Antibiotics (2 ed.). Basel: Birkhäuser Basel. p. 53. ISBN 9783034881050. Archived from the original on 2016-03-05.
- ^ Jump up to:a b c d e f g h i j k l “BIAXIN® Filmtab® (clarithromycin tablets, USP) BIAXIN® XL Filmtab® (clarithromycin extended-release tablets) BIAXIN® Granules (clarithromycin for oral suspension, USP)” (PDF). November 2, 2015. Archived (PDF) from the original on August 24, 2015. Retrieved November 2, 2015.
- ^ “Clarithromycin Side Effects in Detail – Drugs.com”. Drugs.com. Archived from the original on 2017-08-19. Retrieved 2017-08-18.
- ^ “Safety Alerts for Human Medical Products – Clarithromycin (Biaxin): Drug Safety Communication – Potential Increased Risk of Heart Problems or Death in Patients With Heart Disease”. FDA. Retrieved 24 February 2018.
- ^ Yamaguchi S, Kaneko Y, Yamagishi T, et al. [Clarithromycin-induced torsades de pointes]. Nippon Naika Gakkai Zasshi. 2003;92(1):143–5.
- ^ Winkel P, Hilden J, Fischer Hansen J, Hildebrandt P, Kastrup J, Kolmos HJ, et al. (2011). “Excess sudden cardiac deaths after short-term clarithromycin administration in the CLARICOR trial: why is this so, and why are statins protective?”. Cardiology. 118 (1): 63–7. doi:10.1159/000324533. PMID 21447948. S2CID 11873791.
- ^ Tietz A, Heim MH, Eriksson U, Marsch S, Terracciano L, Krähenbühl S (January 2003). “Fulminant liver failure associated with clarithromycin”. The Annals of Pharmacotherapy. 37 (1): 57–60. doi:10.1345/1542-6270(2003)037<0057:flfawc>2.0.co;2. PMID 12503933.
- ^ Patel AM, Shariff S, Bailey DG, Juurlink DN, Gandhi S, Mamdani M, et al. (June 2013). “Statin toxicity from macrolide antibiotic coprescription: a population-based cohort study”. Annals of Internal Medicine. 158 (12): 869–76. doi:10.7326/0003-4819-158-12-201306180-00004. PMID 23778904. S2CID 21222679.
- ^ Gandhi S, Fleet JL, Bailey DG, McArthur E, Wald R, Rehman F, Garg AX (December 2013). “Calcium-channel blocker-clarithromycin drug interactions and acute kidney injury”. JAMA. 310 (23): 2544–53. doi:10.1001/jama.2013.282426. PMID 24346990.
- ^ Gélisse P, Hillaire-Buys D, Halaili E, Jean-Pastor MJ, Vespignan H, Coubes P, Crespel A (November 2007). “[Carbamazepine and clarithromycin: a clinically relevant drug interaction]”. Revue Neurologique. 163 (11): 1096–9. doi:10.1016/s0035-3787(07)74183-8. PMID 18033049.
- ^ Sekar VJ, Spinosa-Guzman S, De Paepe E, De Pauw M, Vangeneugden T, Lefebvre E, Hoetelmans RM (January 2008). “Darunavir/ritonavir pharmacokinetics following coadministration with clarithromycin in healthy volunteers”. Journal of Clinical Pharmacology. 48 (1): 60–5. doi:10.1177/0091270007309706. PMID 18094220. S2CID 38368595.
- ^ Polis MA, Piscitelli SC, Vogel S, Witebsky FG, Conville PS, Petty B, et al. (August 1997). “Clarithromycin lowers plasma zidovudine levels in persons with human immunodeficiency virus infection”. Antimicrobial Agents and Chemotherapy. 41 (8): 1709–14. doi:10.1128/AAC.41.8.1709. PMC 163990. PMID 9257746.
- ^ Ferrero JL, Bopp BA, Marsh KC, Quigley SC, Johnson MJ, Anderson DJ, et al. (1990). “Metabolism and disposition of clarithromycin in man”. Drug Metabolism and Disposition. 18 (4): 441–6. PMID 1976065.
- ReferencesAllevi, P. et al.: Bioorg. Med. Chem. (BMECEP) 7, 12, 2749 (1999)Watanabe, Y. et al.: Heterocycles (HTCYAM) 31, 12, 2121 (1990).EP 158 467 (Taisho Pharmaceutical Co.; 22.3.1985; J-prior. 6.4.1984).EP 272 110 (Taisho Pharmaceutical Co.; 16.12.1987; J-prior. 17.12.1986).US 2 001 037 015 (Teva Pharm.; 15.12.2000; USA-prior. 29.2.2000).KR 2 000 043 839 (Hanmi Pharm.; ROK-prior. 29.12.1998).EP 1 150 990 (Hanmi Pharm.; 7.11.2001; ROK-prior. 29.12.1998)EP 41 355 (Taisho Pharmaceutical Co.; 27.5.1981; J-prior. 4.6.1980).Preparation of O,N-dicarbobenzoxy-N-demethylerythromycin:Flynn, E. H. et al.: J. Am. Chem. Soc. (JACSAT) 77, 3104 (1955).Process for preparation of erythromycin A oxime:US 5 808 017 (Abbott; 15.9.1998; USA-prior. 10.4.1996).Alternative synthesis of clarithromycin:Liao, G.; Zhang, G.; He, T.: Zhongguo Kangshengsu Zazhi (ZKZAEY) 27, 3, 148 (2002) (in Chinese).EP 1 134 229 (Hanmi Pharmac. Co.; 19.9.2001; ROK-prior. 15.3.2000).Crystal form 0 of clarithromycin:The Merck Index, 13th Ed., 2362, p. 408.US 5 945 405 (Abbott; 31.8.1999; USA-prior. 17.1.1997).
External links
- “Clarithromycin”. Drug Information Portal. U.S. National Library of Medicine.
- US patent 4331803, Watanabe Y, Morimoto S, Omura S, “Novel erythromycin compounds”, issued 1981-05-19, assigned to Taisho Pharmaceutical
- “FDA review finds additional data supports the potential for increased long-term risks with antibiotic clarithromycin (Biaxin) in patients with heart disease” (PDF). FDA Drug Safety Communication.
| Clinical data | |
|---|---|
| Trade names | Biaxin, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a692005 |
| License data | EU EMA: by INNUS DailyMed: Clarithromycin |
| Pregnancy category | AU: B3 |
| Routes of administration | By mouth, intravenous |
| Drug class | Macrolides |
| ATC code | J01FA09 (WHO) |
| Legal status | |
| Legal status | AU: S4 (Prescription only)US: ℞-onlyEU: Rx-only [1]In general: ℞ (Prescription only) |
| Pharmacokinetic data | |
| Bioavailability | 50% |
| Protein binding | low binding |
| Metabolism | hepatic |
| Elimination half-life | 3–4 h |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 81103-11-9 |
| PubChem CID | 84029 |
| DrugBank | DB01211 |
| ChemSpider | 10342604 |
| UNII | H1250JIK0A |
| KEGG | D00276 |
| ChEMBL | ChEMBL1741 |
| CompTox Dashboard (EPA) | DTXSID3022829 |
| ECHA InfoCard | 100.119.644 |
| Chemical and physical data | |
| Formula | C38H69NO13 |
| Molar mass | 747.964 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| hideSMILESCC[C@@H]1[C@@]([C@@H]([C@H](C(=O)[C@@H](C[C@@]([C@@H]([C@H]([C@@H]([C@H](C(=O)O1)C)O[C@H]2C[C@@]([C@H]([C@@H](O2)C)O)(C)OC)C)O[C@H]3[C@@H]([C@H](C[C@H](O3)C)N(C)C)O)(C)OC)C)C)O)(C)O | |
| hideInChIInChI=1S/C38H69NO13/c1-15-26-38(10,45)31(42)21(4)28(40)19(2)17-37(9,47-14)33(52-35-29(41)25(39(11)12)16-20(3)48-35)22(5)30(23(6)34(44)50-26)51-27-18-36(8,46-13)32(43)24(7)49-27/h19-27,29-33,35,41-43,45H,15-18H2,1-14H3/t19-,20-,21+,22+,23-,24+,25+,26-,27+,29-,30+,31-,32+,33-,35+,36-,37-,38-/m1/s1 Key:AGOYDEPGAOXOCK-KCBOHYOISA-N | |
| (what is this?) (verify) |
////////////////////CLARITHROMYCIN, Antibacterial, Antibiotics, Macrolides, A-56268, TE-031,
#CLARITHROMYCIN, #Antibacterial, #Antibiotics, #Macrolides, #A-56268, #TE-031,
ERYTHROMYCIN

Erythromycin
NSC-55929
UNII63937KV33D
CAS number114-07-8
- Molecular FormulaC37H67NO13
- Average mass733.927 Da
- эритромицин [Russian] [INN]إيريثروميسين [Arabic] [INN]红霉素 [Chinese] [INN]
IUPAC Name(3R,4S,5S,6R,7R,9R,11R,12R,13S,14R)-6-{[(2S,3R,4S,6R)-4-(dimethylamino)-3-hydroxy-6-methyloxan-2-yl]oxy}-14-ethyl-7,12,13-trihydroxy-4-{[(2R,4R,5S,6S)-5-hydroxy-4-methoxy-4,6-dimethyloxan-2-yl]oxy}-3,5,7,9,11,13-hexamethyl-1-oxacyclotetradecane-2,10-dione
Synthesis ReferenceTakehiro Amano, Masami Goi, Kazuto Sekiuchi, Tomomichi Yoshida, Masahiro Hasegawa, “Process for preparing erythromycin A oxime or a salt thereof.” U.S. Patent US5274085, issued October, 1966.
US5274085ErythromycinCAS Registry Number: 114-07-8Additional Names: E-Base; E-Mycin; Erythromycin ATrademarks: Aknemycin (Hermal); Aknin (Lichtenstein); Emgel (GSK); Ery-Derm (Abbott); Erymax (Merz); Ery-Tab (Abbott); Erythromid (Abbott); ERYC (Warner-Chilcott); Erycen (APS); Erycin (Nycomed); Erycinum (Cytochemia); Ermysin (Orion); Gallimycin (Bimeda); Ilotycin (Lilly); Inderm (Dermapharm); PCE (Abbott); Retcin (DDSA); Staticin (Westwood); Stiemycin (Stiefel)Molecular Formula: C37H67NO13Molecular Weight: 733.93Percent Composition: C 60.55%, H 9.20%, N 1.91%, O 28.34%Literature References: Antibiotic substance produced by a strain of Streptomyces erythreus (Waksman) Waksman & Henrici, found in a soil sample from the Philippine Archipelago. Isoln: McGuire et al.,Antibiot. Chemother.2, 281 (1952); Bunch, McGuire, US2653899 (1953 to Lilly); Clark, Jr., US2823203 (1958 to Abbott). Properties: Flynn et al.,J. Am. Chem. Soc.76, 3121 (1954). Solubility data: Weiss et al.,Antibiot. Chemother.7, 374 (1957). Structure: Wiley et al.,J. Am. Chem. Soc.79, 6062 (1957). Configuration: Hofheinz, Grisebach, Ber.96, 2867 (1963); Harris et al.,Tetrahedron Lett.1965, 679. There are three erythromycins produced during fermentation, designated A, B, and C; A is the major and most important component. Erythromycins A and B contain the same sugar moieties, desosamine, q.v., and cladinose (3-O-methylmycarose). They differ in position 12 of the aglycone, erythronolide, A having an hydroxyl substituent. Component C contains desosamine and the same aglycone present in A but differs by the presence of mycarose, q.v., instead of cladinose. Structure of B: P. F. Wiley et al.,J. Am. Chem. Soc.79, 6070 (1957); of C: eidem,ibid. 6074. Synthesis of the aglycone, erythronolide B: E. J. Corey et al.,ibid.100, 4618, 4620 (1978); of erythronolide A: eidem,ibid.101, 7131 (1979). Asymmetric total synthesis of erythromycin A: R. B. Woodward et al.,ibid.103, 3215 (1981). NMR spectrum of A: D. J. Ager, C. K. Sood, Magn. Reson. Chem.25, 948 (1987). HPLC determn in plasma: W. Xiao et al., J. Chromatogr. B817, 153 (2005). Biosynthesis: Martin, Goldstein, Prog. Antimicrob. Anticancer Chemother., Proc. 6th Int. Congr. Chemother.II, 1112 (1970); Martin et al.,Tetrahedron31, 1985 (1975). Cloning and expression of clustered biosynthetic genes: R. Stanzak et al.,Biotechnology4, 229 (1986). Reviews: T. J. Perun in Drug Action and Drug Resistance in Bacteria1, S. Mitsuhashi, Ed. (University Park Press, Baltimore, 1977) pp 123-152; Oleinick in Antibioticsvol. 3, J. W. Corcoran, F. E. Hahn, Eds. (Springer-Verlag, New York, 1975) pp 396-419; Infection10, Suppl. 2, S61-S118 (1982). Comprehensive description: W. L. Koch, Anal. Profiles Drug Subs.8, 159-177 (1979).Properties: Hydrated crystals from water, mp 135-140°, resolidifies with second mp 190-193°. Melting point taken after drying at 56° and 8 mm. [a]D25 -78° (c = 1.99 in ethanol). uv max (pH 6.3): 280 nm (e 50). pKa1 8.8. Basic reaction. Readily forms salts with acids. Soly in water: ~2 mg/ml. Freely sol in alcohols, acetone, chloroform, acetonitrile, ethyl acetate. Moderately sol in ether, ethylene dichloride, amyl acetate.Melting point: mp 135-140°, resolidifies with second mp 190-193°pKa: pKa1 8.8Optical Rotation: [a]D25 -78° (c = 1.99 in ethanol)Absorption maximum: uv max (pH 6.3): 280 nm (e 50) Derivative Type: EthylsuccinateCAS Registry Number: 41342-53-4Trademarks: Anamycin (Chephasaar); Arpimycin (Rosemont); E.E.S. (Abbott); Eritrocina (Abbott); Eryliquid (Linden); Eryped (Abbott); Erythroped (Abbott); Esinol (Toyama); Monomycin (Grñenthal); Paediathrocin (Abbott); Pediamycin (Abbott); Refkas (Maruko)Molecular Formula: C43H75NO16Molecular Weight: 862.05Percent Composition: C 59.91%, H 8.77%, N 1.62%, O 29.70%Literature References: Prepn: GB830846; R. K. Clark, US2967129 (1960, 1961 both to Abbott).Properties: Hydrated crystals from acetone + water, mp 109-110°. [a]D -42.5°.Melting point: mp 109-110°Optical Rotation: [a]D -42.5° Therap-Cat: Antibacterial.Therap-Cat-Vet: Antibacterial.Keywords: Antibacterial (Antibiotics); Macrolides.
NEW DRUG APPROVALS
one time
$10.00
Product Ingredients
| INGREDIENT | UNII | CAS | INCHI KEY |
|---|---|---|---|
| Erythromycin estolate | XRJ2P631HP | 3521-62-8 | AWMFUEJKWXESNL-JZBHMOKNSA-N |
| Erythromycin ethylsuccinate | 1014KSJ86F | 1264-62-6 | NSYZCCDSJNWWJL-YXOIYICCSA-N |
| Erythromycin gluceptate | 2AY21R0U64 | 23067-13-2 | ZXBDZLHAHGPXIG-VTXLJDRKSA-N |
| Erythromycin lactobionate | 33H58I7GLQ | 3847-29-8 | NNRXCKZMQLFUPL-WBMZRJHASA-N |
| Erythromycin phosphate | I8T8KU14X7 | 4501-00-2 | VUEMAFLGEMYXIH-YZPBMOCRSA-N |
| Erythromycin stearate | LXW024X05M | 643-22-1 | YAVZHCFFUATPRK-YZPBMOCRSA-N |
| Erythromycin sulfate | KVW9N83AME | 7184-72-7 | XTSSJGRRFMNXGO-YZPBMOCRSA-N |
| Erythromycin thiocyanate | Y7A95YRI88 | 7704-67-8 | WVRRTEYLDPNZHR-YZPBMOCRSA-N |
Erythromycin is an antibiotic which belongs to the group of macrolide antibiotics. The pharmaceutically distributed product consists of three components: Erythromycin A, B, and C where Erythromycin A represents the main component. Naturally this antibiotic is synthesized by the grampositive bacteria Streptomyces erythreus (Saccharopolyspora erythrea).
In 1949 Erythromycin was found for the first time in a soil sample in the Philippine region Iloilo. A research team, led by J. M. McGuire, was able to isolate Erythromycin which was part of the soil sample. Under the brand name Ilosone the product was launched commercially in 1952. They named the brand after the region where the antibiotic was found. Analogically the first product name was Ilotycin. Furthermore, in 1953 the U.S. patent was granted. Since 1957 the structure of Erythromycin is known and in 1965 the X-ray structure analysis gave awareness of the absolute configuration. In 1981, almost 30 years after the detection of Erythromycin, Robert B. Woodward, the Nobel prize laureate of chemistry in 1965, and his coworkers posthumously reported the first synthesis of Erythromycin A
The structural characteristic of macrolides, to which Erythromycin affiliates, is a macrocyclic lactone ring of fourteen, fifteen or sixteen members. In case of Erythromycin the lactone ring consists of 14-members. Substituents on the mainchain are cladinose on C-3 and desosamine on C-5. Erythromycin is not a single compound but represents an alloy of structural very similar components. The main constituents are Erythromycin A, B and C. As shown in Table 1 and Figure 1 they only differ in two rests on the lactone ring or on the cladinose each case. In addition to the variants already mentioned, further variants, like Erythromycin D and E are known. They are pre- and post-stages in the biosynthesis and often do not have antibiotic effects

Chemical and Pharmacokinetic Properties Formula: C37H67NO13 CAS-Number: 114-07-8 Molar Mass: 733.93g/mol Half Hife 1.5 hours pkA: 8,6 – 8,8 Melting Point: 411K (hydrat) 463-466K (anhydrous)
Erythromycin is an antibiotic used for the treatment of a number of bacterial infections.[1] This includes respiratory tract infections, skin infections, chlamydia infections, pelvic inflammatory disease, and syphilis.[1] It may also be used during pregnancy to prevent Group B streptococcal infection in the newborn,[1] as well as to improve delayed stomach emptying.[3] It can be given intravenously and by mouth.[1] An eye ointment is routinely recommended after delivery to prevent eye infections in the newborn.[4]
Common side effects include abdominal cramps, vomiting, and diarrhea.[1] More serious side effects may include Clostridium difficile colitis, liver problems, prolonged QT, and allergic reactions.[1] It is generally safe in those who are allergic to penicillin.[1] Erythromycin also appears to be safe to use during pregnancy.[2] While generally regarded as safe during breastfeeding, its use by the mother during the first two weeks of life may increase the risk of pyloric stenosis in the baby.[5][6] This risk also applies if taken directly by the baby during this age.[7] It is in the macrolide family of antibiotics and works by decreasing bacterial protein production.[1]
Erythromycin was first isolated in 1952 from the bacteria Saccharopolyspora erythraea.[1][8] It is on the World Health Organization’s List of Essential Medicines, the safest and most effective medicines needed in a health system.[9] The World Health Organization classifies it as critically important for human medicine.[10] It is available as a generic medication.[5] In 2017, it was the 215th most commonly prescribed medication in the United States, with more than two million prescriptions.[11][12]
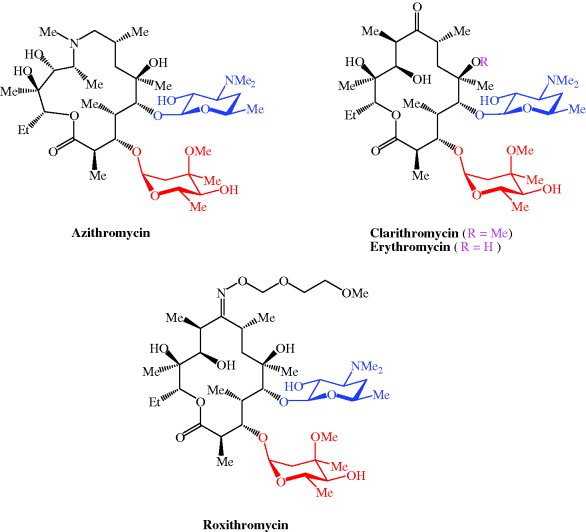
Table 4.2.1 Therapeutic indications for the macrolide antibiotics.
Medical uses
Erythromycin can be used to treat bacteria responsible for causing infections of the skin and upper respiratory tract, including Streptococcus, Staphylococcus, Haemophilus and Corynebacterium genera. The following represents MIC susceptibility data for a few medically significant bacteria:[13]
- Haemophilus influenzae: 0.015 to 256 μg/ml
- Staphylococcus aureus: 0.023 to 1024 μg/ml
- Streptococcus pyogenes: 0.004 to 256 μg/ml
- Corynebacterium minutissimum: 0.015 to 64 μg/ml
It may be useful in treating gastroparesis due to this promotility effect. It has been shown to improve feeding intolerances in those who are critically ill.[14] Intravenous erythromycin may also be used in endoscopy to help clear stomach contents.
Available forms

Enteric-coated erythromycin capsule from Abbott Labs
Erythromycin is available in enteric-coated tablets, slow-release capsules, oral suspensions, ophthalmic solutions, ointments, gels, enteric-coated capsules, non enteric-coated tablets, non enteric-coated capsules, and injections. The following erythromycin combinations are available for oral dosage:[15]
- erythromycin base (capsules, tablets)
- erythromycin estolate (capsules, oral suspension, tablets), contraindicated during pregnancy[16]
- erythromycin ethylsuccinate (oral suspension, tablets)
- erythromycin stearate (oral suspension, tablets)
For injection, the available combinations are:[15]
- erythromycin gluceptate
- erythromycin lactobionate
For ophthalmic use:
- erythromycin base (ointment)
Adverse effects
Gastrointestinal disturbances, such as diarrhea, nausea, abdominal pain, and vomiting, are very common because erythromycin is a motilin agonist.[17] Because of this, erythromycin tends not to be prescribed as a first-line drug.
More serious side effects include arrhythmia with prolonged QT intervals, including torsades de pointes, and reversible deafness. Allergic reactions range from urticaria to anaphylaxis. Cholestasis, Stevens–Johnson syndrome, and toxic epidermal necrolysis are some other rare side effects that may occur.
Studies have shown evidence both for and against the association of pyloric stenosis and exposure to erythromycin prenatally and postnatally.[18] Exposure to erythromycin (especially long courses at antimicrobial doses, and also through breastfeeding) has been linked to an increased probability of pyloric stenosis in young infants.[19][20] Erythromycin used for feeding intolerance in young infants has not been associated with hypertrophic pyloric stenosis.[19]
Erythromycin estolate has been associated with reversible hepatotoxicity in pregnant women in the form of elevated serum glutamic-oxaloacetic transaminase and is not recommended during pregnancy. Some evidence suggests similar hepatotoxicity in other populations.[21]
It can also affect the central nervous system, causing psychotic reactions, nightmares, and night sweats.[22]
Interactions
Erythromycin is metabolized by enzymes of the cytochrome P450 system, in particular, by isozymes of the CYP3A superfamily.[23] The activity of the CYP3A enzymes can be induced or inhibited by certain drugs (e.g., dexamethasone), which can cause it to affect the metabolism of many different drugs, including erythromycin. If other CYP3A substrates — drugs that are broken down by CYP3A — such as simvastatin (Zocor), lovastatin (Mevacor), or atorvastatin (Lipitor)—are taken concomitantly with erythromycin, levels of the substrates increase, often causing adverse effects. A noted drug interaction involves erythromycin and simvastatin, resulting in increased simvastatin levels and the potential for rhabdomyolysis. Another group of CYP3A4 substrates are drugs used for migraine such as ergotamine and dihydroergotamine; their adverse effects may be more pronounced if erythromycin is associated.[22] Earlier case reports on sudden death prompted a study on a large cohort that confirmed a link between erythromycin, ventricular tachycardia, and sudden cardiac death in patients also taking drugs that prolong the metabolism of erythromycin (like verapamil or diltiazem) by interfering with CYP3A4.[24] Hence, erythromycin should not be administered to people using these drugs, or drugs that also prolong the QT interval. Other examples include terfenadine (Seldane, Seldane-D), astemizole (Hismanal), cisapride (Propulsid, withdrawn in many countries for prolonging the QT time) and pimozide (Orap). Theophylline, which is used mostly in asthma, is also contraindicated.
Erythromycin and doxycycline can have a synergistic effect when combined and kill bacteria (E. coli) with a higher potency than the sum of the two drugs together. This synergistic relationship is only temporary. After approximately 72 hours, the relationship shifts to become antagonistic, whereby a 50/50 combination of the two drugs kills less bacteria than if the two drugs were administered separately.[25]
It may alter the effectiveness of combined oral contraceptive pills because of its effect on the gut flora. A review found that when erythromycin was given with certain oral contraceptives, there was an increase in the maximum serum concentrations and AUC of estradiol and dienogest.[26][27]
Erythromycin is an inhibitor of the cytochrome P450 system, which means it can have a rapid effect on levels of other drugs metabolised by this system, e.g., warfarin.
Pharmacology
Mechanism of action
Erythromycin displays bacteriostatic activity or inhibits growth of bacteria, especially at higher concentrations.[28] By binding to the 50s subunit of the bacterial rRNA complex, protein synthesis and subsequent structure and function processes critical for life or replication are inhibited.[28] Erythromycin interferes with aminoacyl translocation, preventing the transfer of the tRNA bound at the A site of the rRNA complex to the P site of the rRNA complex. Without this translocation, the A site remains occupied, thus the addition of an incoming tRNA and its attached amino acid to the nascent polypeptide chain is inhibited. This interferes with the production of functionally useful proteins, which is the basis of this antimicrobial action.
Erythromycin increases gut motility by binding to Motillin, thus it is a Motillin receptor agonist in addition to its antimicrobial properties.
Pharmacokinetics
Erythromycin is easily inactivated by gastric acid; therefore, all orally administered formulations are given as either enteric-coated or more-stable salts or esters, such as erythromycin ethylsuccinate. Erythromycin is very rapidly absorbed, and diffuses into most tissues and phagocytes. Due to the high concentration in phagocytes, erythromycin is actively transported to the site of infection, where, during active phagocytosis, large concentrations of erythromycin are released.
Metabolism
Most of erythromycin is metabolised by demethylation in the liver by the hepatic enzyme CYP3A4. Its main elimination route is in the bile with little renal excretion, 2%-15% unchanged drug. Erythromycin’s elimination half-life ranges between 1.5 and 2.0 hours and is between 5 and 6 hours in patients with end-stage renal disease. Erythromycin levels peak in the serum 4 hours after dosing; ethylsuccinate peaks 0.5-2.5 hours after dosing, but can be delayed if digested with food.[29]
Erythromycin crosses the placenta and enters breast milk. The American Association of Pediatrics determined erythromycin is safe to take while breastfeeding.[30] Absorption in pregnant patients has been shown to be variable, frequently resulting in levels lower than in nonpregnant patients.[29]
Chemistry
Composition
Standard-grade erythromycin is primarily composed of four related compounds known as erythromycins A, B, C, and D. Each of these compounds can be present in varying amounts and can differ by lot. Erythromycin A has been found to have the most antibacterial activity, followed by erythromycin B. Erythromycins C and D are about half as active as erythromycin A.[13][31] Some of these related compounds have been purified and can be studied and researched individually.
Synthesis
Over the three decades after the discovery of erythromycin A and its activity as an antimicrobial, many attempts were made to synthesize it in the laboratory. The presence of 10 stereogenic carbons and several points of distinct substitution has made the total synthesis of erythromycin A a formidable task.[32] Complete syntheses of erythromycins’ related structures and precursors such as 6-deoxyerythronolide B have been accomplished, giving way to possible syntheses of different erythromycins and other macrolide antimicrobials.[33] Woodward successfully completed the synthesis of erythromycin A.[34][35][36]
Erythromycin related compounds
History
In 1949 Abelardo B. Aguilar, a Filipino scientist, sent some soil samples to his employer Eli Lilly. Eli Lilly’s research team, led by J. M. McGuire, managed to isolate erythromycin from the metabolic products of a strain of Streptomyces erythreus (designation changed to Saccharopolyspora erythraea) found in the samples.[37]
Lilly filed for patent protection on the compound which was granted in 1953.[38] The product was launched commercially in 1952 under the brand name Ilosone (after the Philippine region of Iloilo where it was originally collected). Erythromycin was formerly also called Ilotycin.
The antibiotic clarithromycin was invented by scientists at the Japanese drug company Taisho Pharmaceutical in the 1970s as a result of their efforts to overcome the acid instability of erythromycin.
Scientists at Chugai Pharmaceuticals discovered an erythromycin-derived motilin agonist called mitemcinal that is believed to have strong prokinetic properties (similar to erythromycin) but lacking antibiotic properties. Erythromycin is commonly used off-label for gastric motility indications such as gastroparesis. If mitemcinal can be shown to be an effective prokinetic agent, it would represent a significant advance in the gastrointestinal field, as treatment with this drug would not carry the risk of unintentional selection for antibiotic-resistant bacteria.
Society and culture
Cost
It is available as a generic medication.[5]
In the United States in 2014 the price increased to seven dollars per tablet.[39]
The price of Erythromycin rose three times between 2010 and 2015, from 24 cents per tablet in 2010 to $8.96 in 2015.[40] In 2017, a Kaiser Health News study found that the per-unit cost of dozens of generics doubled or even tripled from 2015 to 2016, increasing spending by the Medicaid program. Due to price increases by drug manufacturers, Medicaid paid on average $2,685,330 more for Erythromycin in 2016 compared to 2015 (not including rebates).[41] By 2018, generic drug prices had climbed another 5% on average.[42]
Brand names
Brand names include Robimycin, E-Mycin, E.E.S. Granules, E.E.S.-200, E.E.S.-400, E.E.S.-400 Filmtab, Erymax, Ery-Tab, Eryc, Ranbaxy, Erypar, EryPed, Eryped 200, Eryped 400, Erythrocin Stearate Filmtab, Erythrocot, E-Base, Erythroped, Ilosone, MY-E, Pediamycin, Zineryt, Abboticin, Abboticin-ES, Erycin, PCE Dispertab, Stiemycine, Acnasol, and Tiloryth.
See also
Erythromycin/tretinoin, a combination of tretinoin and the antibiotic erythromycin
SYN
https://basicmedicalkey.com/macrolide-antibiotics/embed/#?secret=VMg8PBg4K9
Synthesis
The total synthesis of the erythromycins (Figure 4.2.2) poses a supreme challenge and has attracted the attention of some of the world’s most eminent synthetic chemists, leading to many elegant examples of the total synthesis of complex natural products. The total synthesis of the erythronolide A aglycone (lacking the sugar units) was first reported by E. J. Corey (Nobel Prize in Chemistry in 1990) in a series of articles in the late 1970s (Scheme 4.2.2) (Corey et al., 1979 and references cited therein), and the total synthesis of erythromycin (known then as erythromycin A) by R. B. Woodward (Nobel Prize in Chemistry in 1965) in a series of articles in 1981, after his death (Scheme 4.2.3) (Woodward et al., 1981 and references cited therein). The Woodward synthesis is particularly elegant, as the dithiadecalin intermediate supplies both the C3-C8 and C9-C13 fragments (Scheme 4.2.3).
Figure 4.2.2 Erythromycins A and B and their aglycones, erythronolides A and B
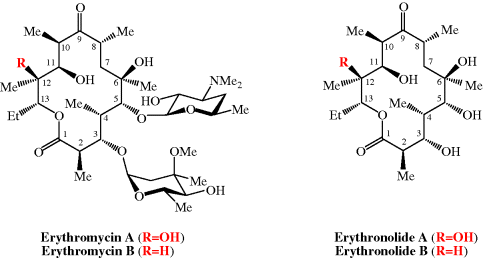
Scheme 4.2.2 Corey’s total synthesis of erythronolide A (38 steps from the cyclohexadiene fragment; 0.04% overall yield)
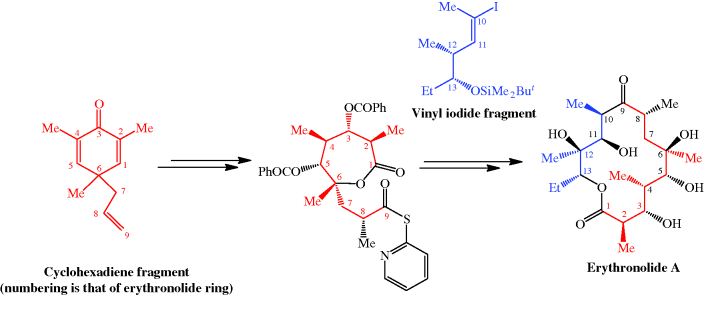
Scheme 4.2.3 Woodward’s total synthesis of erythromycin (56 steps from 4-thianone; 0.01% overall yield)
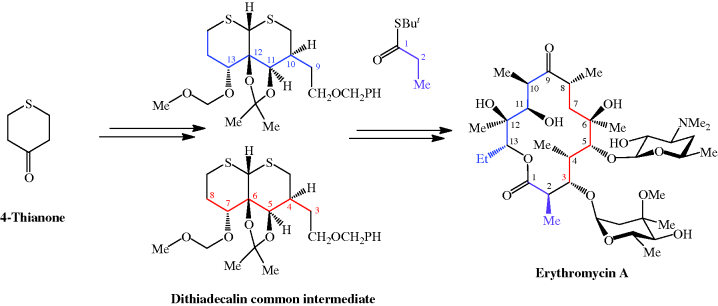
Once again, erythromycin is such a complex antibiotic that its commercial production by total synthesis will never be feasible, and it is obtained from the submerged culture of free or immobilised Saccharopolyspora erythraea (El-Enshasy et al., 2008).
We have now seen a number of examples of how very complex semi-synthetic antibiotics can be prepared through the combination of fermentation (to give the complex natural product) and chemical modification, so you will no doubt already have spotted that both clarithromycin and roxithromycin are semi-synthetic macrolide antibiotics. Clarithromycin can be obtained in a five-step synthetic procedure, from erythromycin oxime (Brunet et al., 2007), while roxithromycin can also be prepared from this oxime (Massey et al., 1970) in a single step (Scheme 4.2.4) (Gouin d’Ambrieres et al., 1982). What is not so obvious is that azithromycin is also a semi-synthetic macrolide, having originally been produced by PLIVA Pharmaceuticals from erythromycin oxime via a sequence of reactions which included the well-known Beckmann rearrangement (Djoki et al., 1986). For more on the synthesis of the erythromycins, see Paterson and Mansuri (1985).
et al., 1986). For more on the synthesis of the erythromycins, see Paterson and Mansuri (1985).
Scheme 4.2.4 Preparation of the semi-synthetic macrolide antibiotic roxithromycin
CLIP

Erythromycin. Erythromycin (1) was discovered in 1952 during the investigation of soil samples from Iloilo, Philippines for antibiotic activity[18, 19] and its molecular structure was assigned in 1957.[20] The microorganism that produced erythromycin was isolated and characterised as Streptomyces erythreus, strain NRRL 2338.[18, 19] Over the years, strain improvements and genetic engineering has allowed the yield of erythromycin to be increased so that 8–10 g L1 can now be produced from a tryptic soy broth.[21–25] Erythromycin forms anhydro-erythromycin 6 and 6:9, 9:12 spiroketal 7 under the acidic conditions in the stomach (Scheme 1), which results in the loss of its antibacterial activity and induction of abdominal pain.[26, 27] Generation of by-products 6 and 7 occurs through an acid-catalysed intramolecular reaction of the C-6 hydroxyl group with the C-9 keto moiety. To avoid this by-product formation several different semi-synthetic derivatives of erythromycin have been prepared in which either of these two functionalities are modified. They led to the discovery of clarithromycin (2) by O-6 methylation of erythromycin (Figure 3). Removal of the C-9 ketone by the formation of an oxime followed by Beckmann rearrangement and reduction led to azithromycin (3), which belongs to a new class of macrolides called “azalides”. Alternatively, conversion of the C-9 ketone to an amine, followed by reaction with an aldehyde, gave dirithromycin (4). Yet another approach involved the transformation of clarithromycin to the conformationally restricted telithromycin
SYN
Chemical Synthesis
Erythromycin, (3R,4S,5S,6R,7R,9R,11R,12R,13S,14R)-4-[(2,6-dideoxy-3-Cmethyl-3-O-methyl-α-L-ribo-hexopyranosyl)-oxy]-14-ethyl-7,12,13-trihydroxy- 3,5,7,9,11,13-hexamethyl-6-[[3,4,6-trideoxy-3-(dimethylamino)-β-D-xylo-hexopyranosyl]oxy ]oxacyclotetradecan-2,10-dione (32.2.1), is more specifically called erythromycin A. It was first isolated in 1952 from the culture liquid of microorganisms of the type Streptomyces erytherus. Minor amounts of erythromycin B and C were also found in the culture fluid. Erythromycin B differs from A in that a hydrogen atom is located at position 12 in the place of a hydroxyl group, while erythromycin C differs from A in that the residue of a different carbohydrate, micarose (2-6-di-deoxy-3-C-methyl-L-ribohexose), is bound to the macrocycle in position 3 in the place of cladinose (4-methoxy-2,4-dimethyl-tetrahydropyran-3,6-diol).
Erythromycin A is produced only microbiologically using active strains of microorganisms of the type Saccharopolospora erythraea.
SYN

Erythromycin synthesis by modular polyketide synthases. The three genes EryAI-III encode three proteins of PKS: DEBS1 (the loading module, modules 1, 2) DEBS2 (modules 3, 4), DEBS3 (modules 5, 6, TE domain). Thus, PKS consists of the loading module, six extension modules, and TE domain. Each module includes from three to six domains: AT-acyl transferase, ACP-acyl carrier protein, KS-ketosynthase, KR-ketoreductase, DH-dehydratase, ERenoyl reductase.
CLIP
The chemical synthesis of Erythromycin poses a huge challenge. The molecule contains ten stereogenic centers of which five are arranged consecutively. R. B. Woodward and his research team first succeeded in synthesizing Erythromycin A. The reaction sequence, however, is so complicated that the yield was only about 0,02 % and, thus, the synthesis is not utilizable comercially. This is the reason for the preferred use of the biosynthesis of Erythromycin via fermentation of Streptomyces erythreus. Other scientists and research teams dealt with the synthesis of Erythromycin as well and developed very similar approaches. Most methods for the Erythromycin synthesis are based on the construction of the aglycon from secoic acid via glycosylation. Indeed the process is also possible inversely: first, a glycosylation, then a lactonization occurs. The yield, however, is considerably less. While earlier scientist mainly dealt with the production of the different secoic acids, the lactonization process is the major problem today because there is no fully developed method for it yet. A lot of side reactions such as dimerization and polymerization appear, because a 14 membered ring is hard to enclose. Even if the chemical synthesis of Erythromycin has no importance for the comercial fabrication of the antibiotic, it is still important for the development and fabrication of its derivatives.
References
- ^ Jump up to:a b c d e f g h i j k “Erythromycin”. The American Society of Health-System Pharmacists. Archived from the original on 2015-09-06. Retrieved Aug 1, 2015.
- ^ Jump up to:a b “Prescribing medicines in pregnancy database”. Australian Government. August 23, 2015. Archived from the original on April 8, 2014.
- ^ Camilleri M, Parkman HP, Shafi MA, Abell TL, Gerson L (January 2013). “Clinical guideline: management of gastroparesis”. The American Journal of Gastroenterology. 108 (1): 18–37, quiz 38. doi:10.1038/ajg.2012.373. PMC 3722580. PMID 23147521.
- ^ Matejcek A, Goldman RD (November 2013). “Treatment and prevention of ophthalmia neonatorum”. Canadian Family Physician. 59 (11): 1187–90. PMC 3828094. PMID 24235191.
- ^ Jump up to:a b c Hamilton RJ (2013). Tarascon pocket pharmacopoeia(2013 delux lab-coat ed., 14th ed.). [Sudbury, Mass.]: Jones & Bartlett Learning. p. 72. ISBN 9781449673611.
- ^ Kong YL, Tey HL (June 2013). “Treatment of acne vulgaris during pregnancy and lactation”. Drugs. 73 (8): 779–87. doi:10.1007/s40265-013-0060-0. PMID 23657872. S2CID 45531743.
- ^ Maheshwai N (March 2007). “Are young infants treated with erythromycin at risk for developing hypertrophic pyloric stenosis?”. Archives of Disease in Childhood. 92 (3): 271–3. doi:10.1136/adc.2006.110007. PMC 2083424. PMID 17337692. Archived from the original on 7 November 2012.
- ^ Vedas JC (2000). Biosynthesis : polyketides and vitamins. Berlin [u.a.]: Springer. p. 52. ISBN 9783540669692.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ World Health Organization (2019). Critically important antimicrobials for human medicine (6th revision ed.). Geneva: World Health Organization. hdl:10665/312266. ISBN 9789241515528. License: CC BY-NC-SA 3.0 IGO.
- ^ “The Top 300 of 2020”. ClinCalc. Retrieved 11 April 2020.
- ^ “Erythromycin – Drug Usage Statistics”. ClinCalc. Retrieved 11 April 2020.
- ^ Jump up to:a b “Erythromycin Susceptibility and Minimum Inhibitory Concentration (MIC) Data” (PDF). TOKU-E.
- ^ Lewis K, Alqahtani Z, Mcintyre L, Almenawer S, Alshamsi F, Rhodes A, et al. (August 2016). “The efficacy and safety of prokinetic agents in critically ill patients receiving enteral nutrition: a systematic review and meta-analysis of randomized trials”. Critical Care. 20 (1): 259. doi:10.1186/s13054-016-1441-z. PMC 4986344. PMID 27527069.
- ^ Jump up to:a b “Erythromycin Oral, Parenteral Advanced Patient Information”. Drugs.com. Archived from the original on 2009-11-30.
- ^ Sexually Transmitted Diseases Treatment Guidelines 2006Archived 2010-02-11 at the Wayback Machine Centers for Disease Control and Prevention. MMWR 2006;55
- ^ Weber FH, Richards RD, McCallum RW (April 1993). “Erythromycin: a motilin agonist and gastrointestinal prokinetic agent”. The American Journal of Gastroenterology. 88 (4): 485–90. PMID 8470625.
- ^ “Pregnancy and lactation”. Archived from the original on 2014-04-20.
- ^ Jump up to:a b Maheshwai N (March 2007). “Are young infants treated with erythromycin at risk for developing hypertrophic pyloric stenosis?”. Archives of Disease in Childhood. 92 (3): 271–3. doi:10.1136/adc.2006.110007. PMC 2083424. PMID 17337692.
- ^ Lund M, Pasternak B, Davidsen RB, Feenstra B, Krogh C, Diaz LJ, et al. (March 2014). “Use of macrolides in mother and child and risk of infantile hypertrophic pyloric stenosis: nationwide cohort study”. BMJ. 348: g1908. doi:10.1136/bmj.g1908. PMC 3949411. PMID 24618148.
- ^ McCormack WM, George H, Donner A, Kodgis LF, Alpert S, Lowe EW, Kass EH (November 1977). “Hepatotoxicity of erythromycin estolate during pregnancy”. Antimicrobial Agents and Chemotherapy. 12 (5): 630–5. doi:10.1128/AAC.12.5.630. PMC 429989. PMID 21610.
- ^ Jump up to:a b “Erythromycine”. Belgisch Centrum voor Farmacotherapeutische Informatie. Archived from the original on 2015-10-06.
- ^ Hunt CM, Watkins PB, Saenger P, Stave GM, Barlascini N, Watlington CO, et al. (January 1992). “Heterogeneity of CYP3A isoforms metabolizing erythromycin and cortisol” (PDF). Clinical Pharmacology and Therapeutics. 51 (1): 18–23. doi:10.1038/clpt.1992.3. hdl:2027.42/109905. PMID 1732074. S2CID 28056649.
- ^ Ray WA, Murray KT, Meredith S, Narasimhulu SS, Hall K, Stein CM (September 2004). “Oral erythromycin and the risk of sudden death from cardiac causes”. The New England Journal of Medicine. 351 (11): 1089–96. doi:10.1056/NEJMoa040582. PMID 15356306.
- ^ Pena-Miller R, Laehnemann D, Jansen G, Fuentes-Hernandez A, Rosenstiel P, Schulenburg H, Beardmore R (April 23, 2013). “When the most potent combination of antibiotics selects for the greatest bacterial load: the smile-frown transition”. PLOS Biology. 11 (4): e1001540. doi:10.1371/journal.pbio.1001540. PMC 3635860. PMID 23630452.
- ^ Blode H, Zeun S, Parke S, Zimmermann T, Rohde B, Mellinger U, Kunz M (October 2012). “Evaluation of the effects of rifampicin, ketoconazole and erythromycin on the steady-state pharmacokinetics of the components of a novel oral contraceptive containing estradiol valerate and dienogest in healthy postmenopausal women”. Contraception. 86 (4): 337–44. doi:10.1016/j.contraception.2012.01.010. PMID 22445438.
- ^ Simmons KB, Haddad LB, Nanda K, Curtis KM (January 2018). “Drug interactions between non-rifamycin antibiotics and hormonal contraception: a systematic review”. American Journal of Obstetrics and Gynecology. 218 (1): 88–97.e14. doi:10.1016/j.ajog.2017.07.003. PMID 28694152. S2CID 36567820.
- ^ Jump up to:a b Katzung PHARMACOLOGY, 9e Section VIII. Chemotherapeutic Drugs Chapter 44. Chloramphenicol, Tetracyclines, Macrolides, Clindamycin, & Streptogramins
- ^ Jump up to:a b “unknown”. Archived from the original on 2014-04-19. Cite uses generic title (help)
- ^ American Academy of Pediatrics Committee on Drugs (September 2001). “Transfer of drugs and other chemicals into human milk” (PDF). Pediatrics. 108 (3): 776–89. doi:10.1542/peds.108.3.776. PMID 11533352. Archived from the original (PDF) on 2016-03-05.
- ^ Kibwage IO, Hoogmartens J, Roets E, Vanderhaeghe H, Verbist L, Dubost M, et al. (November 1985). “Antibacterial activities of erythromycins A, B, C, and D and some of their derivatives”. Antimicrobial Agents and Chemotherapy. 28 (5): 630–3. doi:10.1128/aac.28.5.630. PMC 176346. PMID 4091529.
- ^ Pal S (2006). “A journey across the sequential development of macrolides and ketolides related to erythromycin”. Tetrahedron. 62(14): 3171–3200. doi:10.1016/j.tet.2005.11.064.
- ^ Evans DA, Kim AS (1997). “Synthesis of 6-Deoxyerythronolide B. Implementation of a General Strategy for the Synthesis of Macrolide Antibiotics”. Tetrahedron Lett. 38: 53–56. doi:10.1016/S0040-4039(96)02258-7.
- ^ Woodward RB, Logusch E, Nambiar KP, Sakan K, Ward DE, Au-Yeung, Balaram P, Browne LJ, Card PJ, et al. (1981). “Asymmetric Total Synthesis of Erythromycin. 1. Synthesis of an Erythronolide A Seco Acid Derivative via Asymmetric Induction”. J. Am. Chem. Soc. 103 (11): 3210–3213. doi:10.1021/ja00401a049.
- ^ Woodward RB, Logusch E, Nambiar KP, Sakan K, Ward DE, Au-Yeung, Balaram P, Browne LJ, Card PJ, et al. (1981). “Asymmetric Total Synthesis of Erythromycin. 2. Synthesis of an Erythronolide A Lactone System”. J. Am. Chem. Soc. 103 (11): 3213–3215. doi:10.1021/ja00401a050.
- ^ Woodward RB, Logusch E, Nambiar KP, Sakan K, Ward DE, Au-Yeung, Balaram P, Browne LJ, Card PJ, et al. (1981). “Asymmetric Total Synthesis of Erythromycin. 3. Total Synthesis of Erythromycin”. J. Am. Chem. Soc. 103 (11): 3215–3217. doi:10.1021/ja00401a051.
- ^ Hibionada, F. Remembering the battle of Dr. Abelardo Aguilar: Cure for millions, deprived of millions. The News Today. Retrieved 22 September 2015
- ^ U.S. Patent 2,653,899
- ^ Stahl, Stephanie (September 26, 2014) Health: Generic Drugs Prices Increasing Archived 2016-04-09 at the Wayback Machine CBS Philadelphia. Retrieved March 24, 2016.
- ^ “Some Generic Drugs See Huge Price Increases”. http://www.medscape.com. Retrieved 29 June 2018.
- ^ “Climbing cost of decades-old drugs threatens to break Medicaid bank”. Philly.com. Retrieved 29 June 2018.
- ^ “Are Drugs Really Getting More Expensive? Yes”. The GoodRx Prescription Savings Blog. 27 February 2018. Retrieved 29 June2018.
External links
- “Erythromycin”. Drug Information Portal. U.S. National Library of Medicine.
- U.S. Patent 2,653,899
| Clinical data | |
|---|---|
| Trade names | Eryc, Erythrocin, others[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682381 |
| License data | US DailyMed: Erythromycin |
| Pregnancy category | AU: A[2] |
| Routes of administration | By mouth, intravenous (IV), intramuscular (IM), topical, eye drops |
| Drug class | Macrolide antibiotic |
| ATC code | D10AF02 (WHO) J01FA01 (WHO) S01AA17 (WHO) QJ51FA01 (WHO) |
| Legal status | |
| Legal status | AU: S4 (Prescription only)UK: POM (Prescription only)US: ℞-only |
| Pharmacokinetic data | |
| Bioavailability | Depends on the ester type between 30% – 65% |
| Protein binding | 90% |
| Metabolism | liver (under 5% excreted unchanged) |
| Elimination half-life | 1.5 hours |
| Excretion | bile |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 114-07-8 |
| PubChem CID | 12560 |
| IUPHAR/BPS | 1456 |
| DrugBank | DB00199 |
| ChemSpider | 12041 |
| UNII | 63937KV33D |
| KEGG | D00140 |
| ChEBI | CHEBI:42355 |
| ChEMBL | ChEMBL532 |
| PDB ligand | ERY (PDBe, RCSB PDB) |
| CompTox Dashboard (EPA) | DTXSID4022991 |
| ECHA InfoCard | 100.003.673 |
| Chemical and physical data | |
| Formula | C37H67NO13 |
| Molar mass | 733.937 g·mol−1 |
| hideSMILESCC[C@@H]1[C@@]([C@@H]([C@H](C(=O)[C@@H](C[C@@]([C@@H]([C@H]([C@@H]([C@H](C(=O)O1)C)O[C@H]2C[C@@]([C@H]([C@@H](O2)C)O)(C)OC)C)O[C@H]3[C@@H]([C@H](C[C@H](O3)C)N(C)C)O)(C)O)C)C)O)(C)O | |
| hideInChIInChI=1S/C37H67NO13/c1-14-25-37(10,45)30(41)20(4)27(39)18(2)16-35(8,44)32(51-34-28(40)24(38(11)12)15-19(3)47-34)21(5)29(22(6)33(43)49-25)50-26-17-36(9,46-13)31(42)23(7)48-26/h18-26,28-32,34,40-42,44-45H,14-17H2,1-13H3/t18-,19-,20+,21+,22-,23+,24+,25-,26+,28-,29+,30-,31+,32-,34+,35-,36-,37-/m1/s1 Key:ULGZDMOVFRHVEP-RWJQBGPGSA-N | |
| (verify) |
//////////erythromycin, NSC-55929, NSC 55929, эритромицин , إيريثروميسين , 红霉素 , ANTIBACTERIAL, MACROLIDES, ANTIBIOTICS
#erythromycin, #NSC-55929, #NSC 55929, #эритромицин , #إيريثروميسين , #红霉素 , #ANTIBACTERIAL, #MACROLIDES, #ANTIBIOTICS

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....











