Home » SFDA FAST TRACK
Category Archives: SFDA FAST TRACK
EFAVIRENZ – Huahai Pharma China-Approved to Produce AIDS Treatment

Efavirenz
DMP 266
- Sustiva (USA, Bristol-Myers Squibb)
- Stocrin (EU, MSD)
- Aspen Efavirenz (Sub-Saharan Africa, Aspen Pharmacare)
- E.F (McNeil & Argus)
- Efavir (Cipla)
- Efcure (Emcure Pharmaceuticals)
- Efferven (Ranbaxy Laboratories)
- Estiva (Hetero)
- Evirenz (Alkem Laboratories)
- Viranz (Aurobindo Pharma)
Zhejiang Huahai Pharma received CFDA approval to produce efavirenz, an oral non-nucleoside reverse transcriptase inhibitor (NNRTI) used to control the symptoms of AIDS. Huahai is the first China drugmaker approved to make the drug. Huahai produced efavirenz API for Merck, which marketed the drug under the name Stocrin
read at
http://www.sinocast.com/readbeatarticle.do?id=99634
Efavirenz (EFV), sold under the brand names Sustiva among others, is a non-nucleoside reverse transcriptase inhibitor (NNRTI). It is used as part of highly active antiretroviral therapy (HAART) for the treatment of a human immunodeficiency virus (HIV) type 1. For HIV infection that has not previously been treated, the United States Department of Health and Human Services Panel on Antiretroviral Guidelines currently recommends the use of efavirenz in combination with tenofovir/emtricitabine (Truvada) as one of the preferred NNRTI-based regimens in adults and adolescents.[1] Efavirenz is also used in combination with other antiretroviral agents as part of an expanded postexposure prophylaxis regimen to reduce the risk of HIV infection in people exposed to a significant risk (e.g. needlestick injuries, certain types of unprotected sex etc.).
It is usually taken on an empty stomach at bedtime to reduce neurological and psychiatric adverse effects.
Efavirenz was combined with the HIV medications tenofovir and emtricitabine, all of which are reverse transcriptase inhibitors. This combination of three medications under the brand name Atripla, provides HAART in a single tablet taken once a day.
Efavirenz was discovered at Merck Research Laboratories. It is on the WHO Model List of Essential Medicines, the most important medication needed in a basic health system.[2] As of 2015 the cost for a typical month of medication in the United States is more than 200 USD.[3]
Efavirenz (EFV, brand names Sustiva, Stocrin, Efavir etc.) is a non-nucleoside reverse transcriptase inhibitor (NNRTI) and is used as part of highly active antiretroviral therapy(HAART) for the treatment of a human immunodeficiency virus (HIV) type 1.
For HIV infection that has not previously been treated, the United States Department of Health and Human Services Panel on Antiretroviral Guidelines currently recommends the use of efavirenz in combination with tenofovir/emtricitabine (Truvada) as one of the preferred NNRTI-based regimens in adults and adolescents.
Efavirenz is also used in combination with other antiretroviral agents as part of an expanded postexposure prophylaxis regimen to reduce the risk of HIV infection in people exposed to a significant risk (e.g. needlestick injuries, certain types of unprotected sex etc.).
The usual adult dose is 600 mg once a day. It is usually taken on an empty stomach at bedtime to reduce neurological and psychiatric adverse effects.
Efavirenz was combined with the popular HIV medication Truvada, which consists oftenofovir and emtricitabine, all of which are reverse transcriptase inhibitors. This combination of three medications approved by the U.S. Food and Drug Administration(FDA) in July 2006 under the brand name Atripla, provides HAART in a single tablet taken once a day. It results in a simplified drug regimen for many patients.

doi:10.1016/0040-4039(95)01955-H
Merck synthesis of Efavirenz
History
Efavirenz was approved by the FDA on September 21, 1998, making it the 14th approved antiretroviral drug.
-
Efavirenz is a non-nucleoside reverse trancriptase inhibitor being studied clinically for use in the treatment of HIV infections and AIDS.
- Efavirenz chemically known as (-) 6-Chloro-4-cyclopropylethynyl-4-trifluoromethyl- 1 , 4- dihydro-2H-3, 1-benzoxa zin-2-one, is a highly potent non-nucleoside reverse transcriptase inhibitor (NNRTI).A number of compounds are effective in the treatment of the human immunodeficiency virus (HIV) which is the retrovirus that causes progressive destruction of the human immune system. Effective treatment through inhibition of HIV reverse transcriptase is known for non- nucleoside based inhibitors. Benzoxazinones have been found to be useful non-nucleoside based inhibitors of HIV reverse transcriptase.(-) β-chloro^-cyclopropylethynyM-trifluoromethyl-l ,4-dihydro-2H-3,l -ben zoxazin-2-one (Efavirenz) is efficacious against HIV reverse transcriptase resistance. Due to the importance of (-)6-chloro-4-cyclopropylethynyl-4-trifluoromethyl-l,4-dihydro-2H-3,l-ben zoxazin-2- one, economical and efficient synthetic processes for its production needs to be developed.The product patent US5519021. discloses the preparation of Efavirenz, in Example-6, column-29, involving cyclisation of racemic mixture of 2-(2-amino-5-chlorophenyl)-4- cyclopropyl-l,l,l-trifluoro-3-butyn-2-ol using l ,l ‘-carbonyldiimidazole as carbonyl delivering agent to give racemic Efavirenz. Further, resolution of the racemic Efavirenz is carried out using (-) camphanic acid chloride to yield optically pure Efavirenz. However, research article published in the Drugs of the future, 1998, 23(2), 133-141 discloses process for manufacture of optically pure Efavirenz. The process involves cyclisation of racemic 2-(2-amino-5-chlorophenyl)-4-cyclopropyl-l, 1, l-trifluoro-3-butyn-2- ol using 1, 1-carbonyldiimidazole as carbonyl delivering agent to give racemic Efavirenz and further resolution by (-) camphanic acid chloride.Similarly research article published in Synthesis 2000, No. 4, 479-495 discloses stereoselective synthesis of Efavirenz (95%yield, 99.5%ee), as shown below
Even though many prior art processes report method for the preparation of Efavirenz, each process has some limitations with respect to yield, purity, plant feasibility etc. Hence in view of the commercial importance of Efavirenz there remains need for an improved process.
- US 6 028 237 discloses a process for the manufacture of optically pure Efavirenz.
-
The synthesis of efavirenz and structurally similar reverse transcriptase inhibitors are disclosed in US Patents 5,519,021, 5,663,169, 5,665,720 and the corresponding PCT International Patent Application WO 95/20389, which published on August 3, 1995. Additionally, the asymmetric synthesis of an enantiomeric benzoxazinone by a highly enantioselective acetylide addition and cyclization sequence has been described by Thompson, et al., Tetrahedron Letters 1995, 36, 8937-8940, as well as the PCT publication, WO 96/37457, which published on November 28, 1996.
-
Additionally, several applications have been filed which disclose various aspects of the synthesis of(-)-6-chloro-4-cyclopropylethynyl-4-trifluoromethyl-1,4-dihydro-2H-3,1-benzoxazin-2-one including: 1) a process for making the chiral alcohol, U.S.S.N. 60/035,462, filed 14 January 1997; 2) the chiral additive, U.S.S.N. 60/034,926, filed 10 January 1997; 3) the cyclization reaction, U.S.S.N. 60/037,059, filed 12 February 1997; and the anti-solvent crystallization procedure, U.S.S.N. 60/037,385 filed 5 February 1997 and U.S.S.N. 60/042,807 filed 8 April 1997.




Syntheses of EFV API; different routes of manufacturingAPI, active pharmaceutical ingredient; EFV efavirenz. BELOW
Related substances and degradants (partial listing) in EFVAPI, active pharmaceutical ingredient; CPA, cyclopropylacetylene; EFV, efavirenz
Syntheses of EFV API; different routes of manufacturingAPI, active pharmaceutical ingredient; EFV efavirenz.
Chemical properties
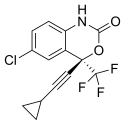
Efavirenz is chemically described as (S)-6-chloro-(cyclopropylethynyl)-1,4-dihydro-4-(trifluoromethyl)-2H-3,1-benzoxazin-2-one. Its empirical formula is C14H9ClF3NO2. Efavirenz is a white to slightly pink crystalline powder with a molecular mass of 315.68 g/mol. It is practically insoluble in water (<10 µg/mL).
History
Efavirenz was approved by the FDA on September 21, 1998, making it the 14th approved antiretroviral drug.
Society and culture
Pricing information
A one-month supply of 600 mg tablets cost approximately $550 in April 2008.[16] Merck provides efavirenz in certain developing countries at cost, currently about $0.65 per day.[17] Some emerging countries have opted to purchase Indian generics[18] such as Efavir by Cipla Ltd.[19] In Thailand, one month supply of efavirenz + truvada, as of June 2012, costs THB 2900 ($90), there’s also a social program for poorer patients who can’t afford even this price. In South Africa, a license has been granted to generics giant Aspen Pharmacare to manufacture, and distribute to Sub-Saharan Africa, a cost-effective antiretroviral drug.[20]
PATENT
http://www.google.com/patents/WO1999061026A1?cl=en
EXAMPLE 1
Cl
1a
To a solution of trifluoroethanol and (IR, 2S)-N-pyrrolidinyl norephedrine in THF (9 L) under nitrogen is added a solution of diethylzinc in hexane at 0 °C slowly enough to keep the temperature below 30 °C. The mixture is stirred at room temperature for 0.5 ~ 1 h. In another dry flask a solution of chloromagnesium cyclopropyl acetylide is prepared as follows: To neat cyclopropyl acetylene at 0 °C is added a solution of rc-butylmagnesium chloride slowly enough to keep the internal temperature < 30 °C. The solution is stirred at 0 °C for ~ 40 min and transfered to the zinc reagent via cannula with 0.36 L of THF as a wash. The mixture is cooled to -10 °C and ketoaniline la is added. The mixture is stirred at -2 to -8 °C for 35 h, warmed to room temperature, stirred for 3 h, and quenched with 30% potassium carbonate over 1.5 h. The mixture is stirred for 4 h and the solid is removed by filtration and washed with THF (2 cake volume). The wet solid still contains -18 wt% of pyrrolidinyl norephedrine and is saved for further study. The filtrate and wash are combined and treated with 30% citric acid. The two layers are separated. The organic layer is washed with water (1.5 L). The combined aqueous layers are extracted with 2.5 L of toluene and saved for norephedrine recovery. The toluene extract is combined with the organic solution and is concentrated to ~ 2.5 L. Toluene is continuously feeded and distilled till THF is not detectable by GC. The final volume is controlled at 3.9 L. Heptane (5.2 L) is added over 1 h. The slurry is cooled to 0 °C, aged for 1 h, and filtered. The solid is washed with heptane (2 cake volume) and dried to give 1.234 Kg (95.2% yield) of amino alcohol 3 as a white crystalline. The material is 99.8 A% pure and 99.3% ee.
EXAMPLE 2
To a three necked round bottom flask, equipped with a mechanical stirrer, nitrogen line, and thermocouple, was charged the solid amino alcohol 3, MTBE (500 L), and aqueous KHCO3 (45 g in 654 mL H2O). Solid 4-nitrophenyl chloroformate was added, in 4 batches, at 25°C. During the addition the solution pH was monitored. The pH was maintained between 8.5 and 4 during the reaction and ended up at 8.0. The mixture was stirred at 20-25°C for two hours. Aqueous KOH (2N) was added over 20 minutes, until the pH of the aqueous layer reached 11.0.
The layers were separated and 500 mL brine was added to the MTBE layer. 0.1 N Acetic acid was added until the pH was 6-7. The layers were separated and the organic phase was washed with brine (500 mL). At this point the mixture was solvent switched to EtOH/IPA and crystallized as recited in Examples 5 and 6.
EXAMPLE 3
To a three necked round bottom flask, equipped with a mechanical stirrer, nitrogen line, and thermocouple, was charged the solid amino alcohol 3a, toulene (500 mL), and aqueous KHCO3 (86.5 g in 500 L H2O). Phosgene solution in toulene was added at 25°C, and the mixture was stirred at 20-25°C for two hours.
The layers were separated and the organic phase was washed with brine (500 mL). At this point the mixture was solvent switched to EtOH/IPA and crystallized as recited in Examples 5 and 6.
EXAMPLE 4
To a three necked round bottom flask, equipped with a mechanical stirrer, nitrogen line, and thermocouple, was charged the solid amino alcohol 3a, MTBE (500 mL), and aqueous KHCO3 (86.5 g in 500 mL H2O). Phosgene gas was slowly passed into the solution at 25°C, until the reaction was complete.
The layers were separated and the organic phase was washed with brine (500 mL). At this point the mixture was solvent switched to EtOH/IPA and crystallized as recited in Examples 5 and 6.
EXAMPLE 5
Crystallization of efavirenz from 30% 2-Propanol in Water using a ratio of 15 ml solvent per gram efavirenz Using Controlled Anti-Solvent Addition on a 400 g Scale.
400 g. of efavirenz starting material is dissolved in 1.8 L of 2- propanol. The solution is filtered to remove extraneous matter. 1.95 L of deionized (DI) water is added to the solution over 30 to 60 minutes. 10 g. to 20 g. of efavirenz seed (Form II wetcake) is added to the solution. The seed bed is aged for 1 hour. The use of Intermig agitators is preferred to mix the slurry. If required (by the presence of extremely long crystals or a thick slurry), the slurry is wet-milled for 15 – 60 seconds. 2.25 L of DI water is added to the slurry over 4 to 6 hours. If required (by the presence of extremely long crystals or a thick slurry), the slurry is wet- milled for 15 – 60 seconds during the addition. The slurry is aged for 2 to 16 hours until the product concentration in the supernatant remains constant. The slurry is filtered to isolate a crystalline wet cake. The wet cake is washed with 1 to 2 bed volumes of 30 % 2-propanol in water and then twice with 1 bed volume of DI water each. The washed wet cake is dried under vacuum at 50°C.
EXAMPLE 6
Crystallization of efavirenz from 30% 2-Propanol in Water using a ratio of 15 ml solvent per gram efavirenz Using a Semi-Continuous Process on a 400 g Scale.
400 g. of efavirenz starting material is dissolved in 1.8 L of 2- propanol. A heel slurry is produced by mixing 20 g. of Form II efavirenz in 0.3 L of 30 % (v/v) 2-propanol in water or retaining part of a slurry froma previous crystallization in the crystallizer. The dissolved batch and 4.2 L of DI water are simultaneously charged to the heel slurry at constant rates over 6 hours to maintain a constant solvent composition in the crystallizer. Use of Intermig agitators during the crystallization is preferred. During this addition the slurry is wet-milled when the crystal lengths become excessively long or the slurry becomes too thick. The slurry is aged for 2 to 16 hours until the product concentration in the supernatant remains constant. The slurry is filtered to isolate a crystalline wet cake. The wet cake is washed with 1 to 2 bed volumes of 30 % 2-propanol in water and then twice with 1 bed volume of DI water each. The washed wet cake is dried under vacuum at 50°C.
EXAMPLE 7 Preparation of Amino Alcohol 3 and ee Upgrading— Through Process
1a
A solution of diethyl zinc in hexane was added to a solution of trifluoroethanol (429.5 g, 4.29’mol) and (IR, 2S)-N-pyrrolidinyl norephedrine (1.35 kg, 6.58 mol) in THF (9 L), under nitrogen, at 0 °C. The resulting mixture was stirred at room temperature for approx. 30 min. In another dry flask a solution of chloromagnesium- cyclopropylacetylide was prepared as follows. To a solution of n- butylmagnesium chloride in THF (2 M, 2.68 L, 5.37 mol) was added neat cyclopropylacetylene at 0 °C keeping the temperature < 25 °C. The solution was stirred at 0 °C for 1 ~ 2 h. The solution of chloromagnesiumcyclopropylacetylide was then warmed to room temperature and was transferred into the zinc reagent via cannula over 5 min followed by vessel rinse with 0.36 L of THF. The resulting mixture was aged at ~ 30 °C for 0.5 h and was then cooled to 20 °C. The ketoaniline 1 (1.00 kg, 4.47 mol) was added in one portion as a solid, and the resulting mixture was stirred at 20-28 °C for 3 h.
The reaction was quenched with 30% aq. potassium carbonate (1.2 L) and aged for 1 h. The solid waste was filtered and the cake was washed with THF (3 cake volumes). The filtrate and wash were combined and solvent switched to IP Ac.
The IPAc solution of product 3 and pyrrolidinyl norephedrine was washed with citric acid (3.5 L) and with water (1.5 L). The combined aqueous layers were extracted with IPAc (2 L) and saved for norephedrine recovery. To the combined organic layers was added
12N HC1 (405 mL, 4.88 mol), to form a thin slurry of the amino alcohol-
HC1 salt. The mixture was aged for 30 min at 25 °C and was then dried azeotropically. The slurry was aged at 25 °C for 30 min and filtered. The cake was washed with 2.5 L of IPAc and dried at 25 °C under vacuum/nitrogen for 24 h to give 1.76 kg of the wet HC1 salt.
The salt was dissolved in a mixture of MTBE (6 L) and aq Na2Cθ3 (1.18 kg in 6.25 L water). The layers were separated and the organic layer was washed with 1.25 L of water. The organic layer was then solvent switched into toluene.
Heptane (5 L) was added over 1 h at 25 °C. The slurry was cooled to 0 °C, aged for 1 h, and filtered. The solid was washed with heptane (2 cake volumes) and was dried to give 1.166 kg (90% overall yield) of amino alcohol 3 as a white crystalline solid. Norephedrine recovery
The aqueous solution was basified to pH13 using 50% aq NaOH, and extracted with heptane (2 L). The heptane solution was washed with water (1 L) and concentrated to remove residual IPAc and water. The final volume was adjusted to about 3 L. The heptane solution was cooled to -20 °C, aged for 2 h, and filtered. The solid was washed with cold heptane (1 cake volume) and dried to give 1.269 kg solid (94% recovery)




CLIPS
http://www.mdpi.com/1420-3049/21/2/221/htm



| WO2007013047A2 * | Jul 31, 2006 | Feb 1, 2007 | Ranbaxy Lab Ltd | Water-dispersible anti-retroviral pharmaceutical compositions |
| WO2007013047A3 * | Jul 31, 2006 | May 31, 2007 | Ranbaxy Lab Ltd | Water-dispersible anti-retroviral pharmaceutical compositions |
| WO2007052289A2 * | Jul 24, 2006 | May 10, 2007 | Rubicon Res Pvt Ltd | Novel dispersible tablet composition |
| WO2007052289A3 * | Jul 24, 2006 | Dec 27, 2007 | Rubicon Res Pvt Ltd | Novel dispersible tablet composition |
| WO2011131943A2 | Apr 20, 2011 | Oct 27, 2011 | Cipla Limited | Pharmaceutical compositions |
| WO2012048884A1 | Oct 14, 2011 | Apr 19, 2012 | Lonza Ltd | Process for the synthesis of cyclic carbamates |
| WO2012048886A1 | Oct 14, 2011 | Apr 19, 2012 | Lonza Ltd | Process for the synthesis of cyclic carbamates |
| WO2015059466A1 | Oct 22, 2014 | Apr 30, 2015 | Cipla Limited | Pharmaceutical compositions comprising efavirenz |
| EP1448170A2 * | Nov 26, 2002 | Aug 25, 2004 | Bristol-Myers Squibb Company | Efavirenz tablet formulation having unique biopharmaceutical characteristics |
| EP2441759A1 | Oct 14, 2010 | Apr 18, 2012 | Lonza Ltd. | Process for the synthesis of cyclic carbamates |
| EP2447255A1 | Oct 14, 2010 | May 2, 2012 | Lonza Ltd. | Process for the synthesis of cyclic carbamates |
| US6238695 | Apr 6, 1999 | May 29, 2001 | Dupont Pharmaceuticals Company | Formulation of fast-dissolving efavirenz capsules or tablets using super-disintegrants |
| US6555133 | Apr 2, 2001 | Apr 29, 2003 | Bristol-Myers Squibb Company | Formulation of fast-dissolving efavirenz capsules or tablets using super-disintegrants |
| US8871271 | Jul 29, 2013 | Oct 28, 2014 | Gilead Sciences, Inc. | Method and composition for pharmaceutical product |
| US8957204 | Oct 14, 2011 | Feb 17, 2015 | Lonza Ltd. | Process for the synthesis of cyclic carbamates |
| US8969550 | Oct 14, 2011 | Mar 3, 2015 | Lonza Ltd. | Process for the synthesis of cyclic carbamates |
| US9018192 | Oct 10, 2013 | Apr 28, 2015 | Bristol-Myers Squibb & Gilead Sciences, Inc. | Unitary pharmaceutical dosage form |
| US9198862 | Jul 24, 2006 | Dec 1, 2015 | Rubicon Research Private Limited | Dispersible tablet composition |
| WO1995020389A1 * | Jan 24, 1995 | Aug 3, 1995 | Merck & Co Inc | Benzoxazinones as inhibitors of hiv reverse transcriptase |
| WO1996037457A1 * | May 21, 1996 | Nov 28, 1996 | Merck & Co Inc | Asymmetric synthesis of (-) 6-chloro-4-cyclopropyl-ethynyl-4-trifluoromethyl-1,4-dihydro-2h-3,1-benzoxazin-2-one |
| WO1998052570A1 * | May 14, 1998 | Nov 26, 1998 | David Walter Barry | Antiviral combinations containing the carbocyclic nucleoside 1592u89 |
References
- 1 “Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents”. Retrieved 10 May 2013.
- 2
- “WHO Model List of EssentialMedicines” (PDF). World Health Organization. October 2013. Retrieved 22 April 2014.
- 3
- Hamilton, Richart (2015). Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. p. 62. ISBN 9781284057560.
- 4
- Cespedes, MS; Aberg, JA (2006). “Neuropsychiatric complications of antiretroviral therapy.”. Drug safety : an international journal of medical toxicology and drug experience 29 (10): 865–74. doi:10.2165/00002018-200629100-00004. PMID 16970510.
- 5
- “www.accessdata.fda.gov” (PDF).
- 6
- DHHS panel. Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents (October 10, 2006). (Available for download from AIDSInfo)
- 7
- Ford, N.; Mofenson, L.; Kranzer, K.; Medu, L.; Frigati, L.; Mills, E. J.; Calmy, A. (2010). “Safety of efavirenz in first-trimester of pregnancy: A systematic review and meta-analysis of outcomes from observational cohorts”. AIDS 24 (10): 1461–1470. doi:10.1097/QAD.0b013e32833a2a14. PMID 20479637.
- 8
- Rossi, S; Yaksh, T; Bentley, H; Van Den Brande, G; Grant, I; Ellis, R (2006). “Characterization of interference with 6 commercial delta9-tetrahydrocannabinol immunoassays by efavirenz (glucuronide) in urine”. Clinical Chemistry 52 (5): 896–7. doi:10.1373/clinchem.2006.067058. PMID 16638958.
- 9
- Röder, CS; Heinrich, T; Gehrig, AK; Mikus, G (2007). “Misleading results of screening for illicit drugs during efavirenz treatment”. AIDS (London, England) 21 (10): 1390–1. doi:10.1097/QAD.0b013e32814e6b3e. PMID 17545727.
- 10
- Ren J, Bird LE, Chamberlain PP; et al. (2002). “Structure of HIV-2 reverse transcriptase at 2.35-A resolution and the mechanism of resistance to non-nucleoside inhibitors”. Proc Natl Acad Sci USA 99 (22): 14410–15. doi:10.1073/pnas.222366699. PMC 137897. PMID 12386343.
- 11
- Sustiva (efavirenz) capsules and tablets. Product information (April 2005)
- 12
- Simen AA, Ma J, Svetnik V, Mayleben D, Maynard J, Roth A, Mixson L, Mogg R, Shera D, George L, Mast TC, Beals C, Stoch A, Struyk A, Shire N, Fraser I (2014). “Efavirenz modulation of sleep spindles and sleep spectral profile”. J Sleep Res 24: 66–73. doi:10.1111/jsr.12196. PMID 25113527.
- 13
- Gatch MB, Kozlenkov A, Huang RQ, Yang W, Nguyen JD, González-Maeso J, Rice KC, France CP, Dillon GH, Forster MJ, Schetz JA (2013). “The HIV antiretroviral drug efavirenz has LSD-like properties”. Neuropsychopharmacology 38 (12): 2373–84. doi:10.1038/npp.2013.135. PMC 3799056. PMID 23702798.
- 14
- Dabaghzadeh F, Ghaeli P, Khalili H, Alimadadi A, Jafari S, Akhondzadeh S, Khazaeipour Z (2013). “Cyproheptadine for prevention of neuropsychiatric adverse effects of efavirenz: a randomized clinical trial”. AIDS Patient Care STDS 27 (3): 146–54. doi:10.1089/apc.2012.0410. PMID 23442031.
- 15
- Dabaghzadeh F, Khalili H, Ghaeli P, Dashti-Khavidaki S (2012). “Potential benefits of cyproheptadine in HIV-positive patients under treatment with antiretroviral drugs including efavirenz”. Expert Opin Pharmacother 13 (18): 2613–24. doi:10.1517/14656566.2012.742887. PMID 23140169.
- 16
- Price listed on http://drugstore.com website, 4/20/2008
- 17
- “Merck & Co., Inc., Again Reduces Price of Stocrin (efavirenz) for Patients in Least Developed Countries and Countries Hardest Hit by Epidemic – Drugs.com MedNews”.
- 18
- IndiaDaily – A new trend in emerging nations – Brazil opts for Indian generic drug ignoring US pharmaceutical giant Merck’s patent on AIDS drug Efavirenz
- 19
- http://www.cipla.com
- 20
- Patrick Lumumba Osewe; Yvonne Korkoi Nkrumah; Emmanuel K. Sackey (15 June 2008). Improving Access to HIV/AIDS Medicines in Africa: Trade-Related Aspects of Intellectual Property Rights (TRIPS) Flexibilities Utilization. World Bank Publications. pp. 35–39. ISBN 978-0-8213-7544-0. Retrieved 30 June 2012.
- 21
- http://www.sustiva.com/
- 22
- http://www.medsafe.govt.nz/consumers/cmi/s/stocrin.pdf
- 23
- Drugsupdate.com generic brands list: http://www.drugsupdate.com/brand/generic/Efavirenz/87
- 24
- http://mcneilargusindia.com/
- 25
- http://www.alkemlabs.com/
- 26
- “Regast® (efavirenz) film-coated tablets.”. http://www.pharmasyntez.com (in Russian). Pharmasyntez, 2011. Retrieved 28 June 2015. External link in
|website=(help) - 27
- IOL: Thugs get high on stolen Aids drugs IOL News May 12, 2007
- 28
- Getting high on HIV drugs in S Africa. BBC News, 8 December 2008.
- 29
- ‘No Turning Back’: Teens Abuse HIV Drugs. ABC News, April 6, 2009.
- 30
- Getting High On HIV Medication Vice 7.04.2014.
- 31
- Gatch, M. B.; Kozlenkov, A.; Huang, R. Q.; Yang, W.; Nguyen, J. D.; González-Maeso, J.; Rice, K. C.; France, C. P.; Dillon, G. H.; Forster, M. J.; Schetz, J. A. (2013). “The HIV Antiretroviral Drug Efavirenz has LSD-Like Properties”. Neuropsychopharmacology 38 (12): 2373–84. doi:10.1038/npp.2013.135. PMC 3799056. PMID 23702798.
- Sütterlin, S.; Vögele, C.; Gauggel, S. (2010). “Neuropsychiatric complications of Efavirenz therapy: suggestions for a new research paradigm”. The Journal of Neuropsychiatry and Clinical Neurosciences 22 (4): 361–369. doi:10.1176/jnp.2010.22.4.361.
External links

|
|
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
(4S)-6-chloro-4-(2-cyclopropylethynyl)-4-(trifluoromethyl)-2,4-dihydro-1H-3,1-benzoxazin-2-one
|
|
| Clinical data | |
| Trade names | Sustiva, Stocrin, others |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a699004 |
| Pregnancy category |
|
| Routes of administration |
By mouth (capsules, tablets) |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 40–45% (under fasting conditions) |
| Protein binding | 99.5–99.75% |
| Metabolism | Hepatic (CYP2A6 and CYP2B6-mediated) |
| Onset of action | 3–5 hours |
| Biological half-life | 40–55 hours |
| Excretion | Urine (14–34%) and feces (16–61%) |
| Identifiers | |
| CAS Number | 154598-52-4 |
| ATC code | J05AG03 (WHO) |
| PubChem | CID 64139 |
| DrugBank | DB00625 |
| ChemSpider | 57715 |
| UNII | JE6H2O27P8 |
| KEGG | D00896 |
| ChEBI | CHEBI:119486 |
| ChEMBL | CHEMBL223228 |
| NIAID ChemDB | 032934 |
| PDB ligand ID | EFZ (PDBe, RCSB PDB) |
| Chemical data | |
| Formula | C14H9ClF3NO2 |
| Molar mass | 315.675 g/mol |

TLC388 (Lipotecan®) Taiwan Liposome Company Hepatic cancer drug candidate gets fast track approval status from SFDA
TLC388 (Lipotecan®) structure can be figured out from a link below of a poster
http://www.tlcbio.com/files/news/2011111701580783.pdf
IT IS A CAMPOTHECIN ANALOGUE
The str can be concluded from above picture from a poster by TLC BIO
TLC388 (Lipotecan) is a potent Topoisomerase-1 inhibitor and it can disrupt both Sonic Hedgehog and HIF1-α pathways to overcome cancer drug resistance and inhibit angiogenesis induced by tumor hypoxia. This phase I first-in-human study of Lipotecan examined the MTD, safety, anti-tumor activity and pharmacokinetic profiles of TLC388 in patients with advanced incurable solid tumors.
Methods: Lipotecan was administered intravenously on day 1, 8 and 15 of a 28-day cycle. Patients underwent safety assessments regularly and tumor assessments every other cycle. Pharmacokinetic samples were drawn on days 1, 8 and 15 of cycles 1 and 2 for all treated patients.
http://mct.aacrjournals.org/cgi/content/meeting_abstract/10/11_MeetingAbstracts/A89
http://clinicaltrials.gov/show/NCT00747474
MAR19 2013
China SFDA has granted fast track approval status to Taiwan Liposome company hepatic cancer drug Lipotecan, shortening the review period. The drug will enter Phase 2 clinical trials in China in the second half of this year. Lipotecan has been granted orphan drug status by US FDA and EU EMEA for the treatment of hepatocellular carcinoma (HCC)
Nexavar is the standard of care in first line advanced liver cancer patients. Lipotecan as a second-line treatment allows patients who have failed prior treatment with Nexavar to maintain a six month course of the disease without progressing

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....






















