Catadegbrutinib
CAS 2736508-60-2
MF C47H54N12O4 MW851.0 g/mol
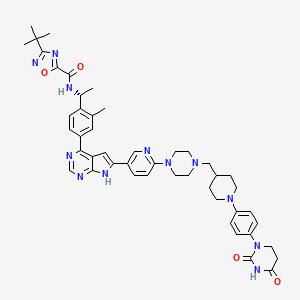
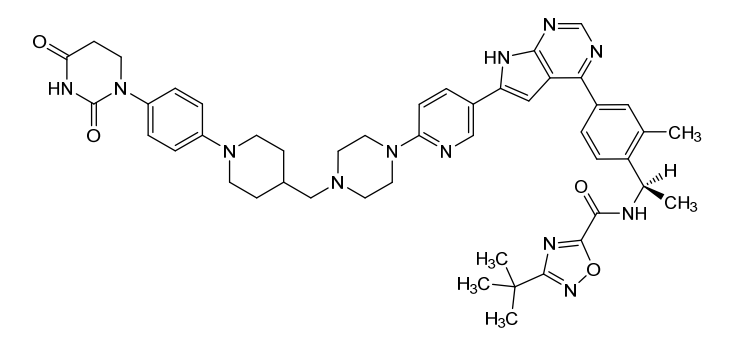
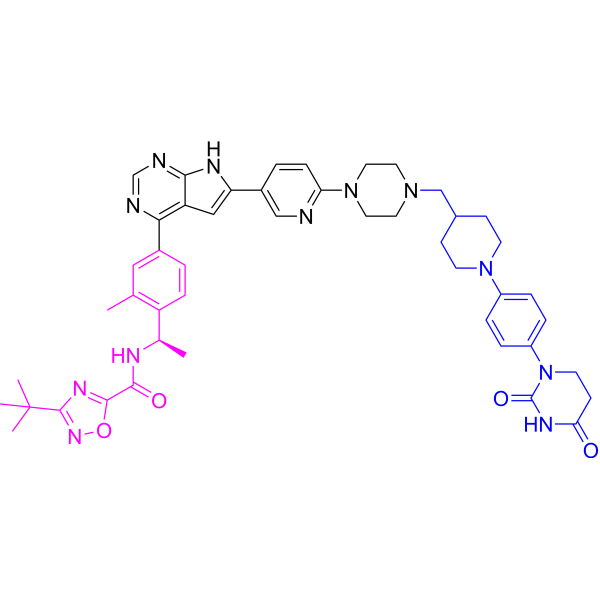
3-tert-butyl-N-[(1R)-1-[4-[6-[6-[4-[[1-[4-(2,4-dioxo-1,3-diazinan-1-yl)phenyl]piperidin-4-yl]methyl]piperazin-1-yl]-3-pyridinyl]-7H-pyrrolo[2,3-d]pyrimidin-4-yl]-2-methylphenyl]ethyl]-1,2,4-oxadiazole-5-carboxamide
3-tert-butyl-N-{(1R)-1-[13-methyl-82,84-dioxo-27H-2(4,6)-pyrrolo[2,3-d]pyrimidina-8(1)-[1,3]diazinana-4(1,4)-piperazina3(5,2)-pyridina-6(4,1)-piperidina-1(1),7(1,4)-dibenzenaoctaphan-14-yl]ethyl}-1,2,4-oxadiazole-5-carboxamide
Bruton tyrosine kinase degrader, antineoplastic, BGB-16673, BGB 16673, PF6GPZ4DYT, BTK-IN-29, Tacabrutideg
Catadegbrutinib (BGB-16673) is an orally active, potent Bruton’s tyrosine kinase (BTK) degrader, or chimeric degradation activator compound (CDAC). It works by targeting BTK for proteasomal degradation, showing high efficacy against wild-type and mutated forms (including C481S) in B-cell malignancies. It is under investigation for cancers such as CLL, SLL, and MCL.
Key Details About Catadegbrutinib
- Mechanism of Action: As a PROTAC-class molecule, it binds to BTK and recruits E3 ubiquitin ligase, causing polyubiquitination and degradation of the protein.
- Target Potency: It shows strong degradation activity, with a
of
(concentration required for 50% degradation) and a
binding
of
.
- Clinical Potential: Developed for B-cell malignancies (chronic lymphocytic leukemia, mantle cell lymphoma) that have developed resistance to covalent and non-covalent BTK inhibitors.
- Synonyms/Codes: BGB-16673, BGB-116673, BTK-IN-29, and recently listed in WHO proposed INN as tacabrutideg.
- Status: Used primarily in research for treating B-cell malignancies, lymphomas, and potentially autoimmune diseases.
Catadegbrutinib is designed to overcome resistance mechanism challenges seen with existing BTK inhibitors.
SYN
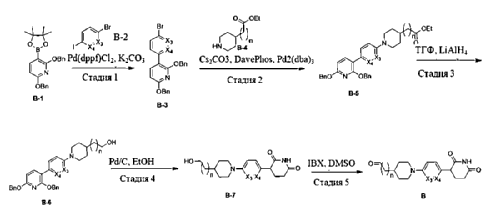
Example 14: (R) -3- (tert-butyl) -N- (1- (4- (6- (6- (4- ( (1- (4- (2, 4-dioxotetrahydropyrimidin-1 (2H) -yl) phenyl) piperidin-4-yl) methyl) piperazin-1-yl) pyridin-3-yl) -7H-pyrrolo [2, 3-d] pyrimidin-4-yl) -2-methylphenyl) ethyl) -1, 2, 4-oxadiazole-5-carboxamide
[0357]
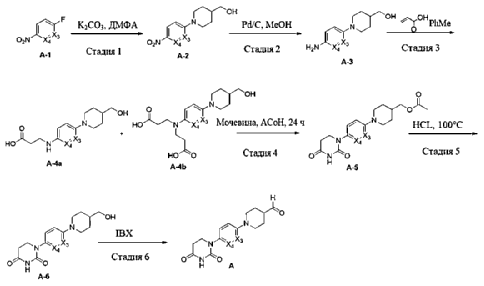
Step 1: tert-butyl 4- (5- (4-chloro-7H-pyrrolo [2, 3-d] pyrimidin-6-yl) pyridin-2-yl) piperazine-1- carboxylate
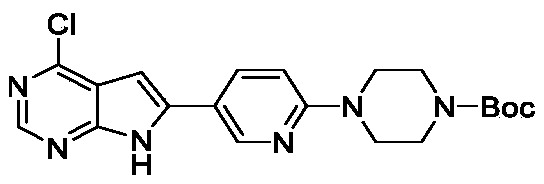
A mixture of 4-chloro-6-iodo-7H-pyrrolo [2, 3-d] pyrimidine (3 g, 10.73 mmol) , tert-butyl 4- (5- (4, 4, 5, 5-tetramethyl-1, 3, 2-dioxaborolan-2-yl) pyridin-2-yl) piperazine-1-carboxylate (4.18 g, 10.73 mmol) , Na 2CO 3(1.25 g, 11.80 mmol) and Pd (dppf) Cl 2(0.39 g, 0.537 mmol) in dioxane (120 mL) and H 2O (20 mL) was stirred in a sealed tube at 85 ℃ overnight. After cooling, the reaction mixture was filtered and the solid was washed with 20 mL of MeOH and dried under vacuum to afford the product (4.05 g, 91%) . [M+H] += 415.0.
[0360]
Step 2: tert-butyl (R) -4- (5- (4- (4- (1- (3- (tert-butyl) -1, 2, 4-oxadiazole-5-carboxamido) ethyl) -3- methylphenyl) -7H-pyrrolo [2, 3-d] pyrimidin-6-yl) pyridin-2-yl) piperazine-1-carboxylate
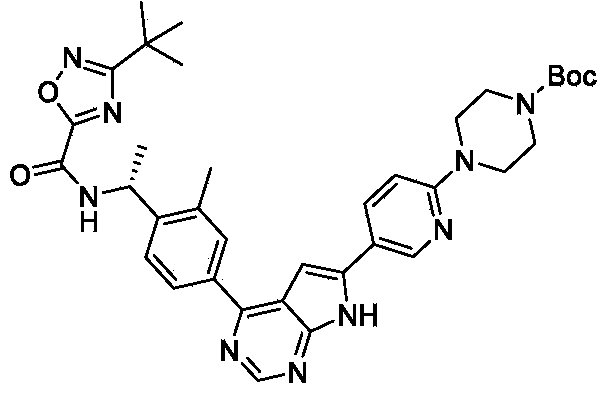
A mixture of tert-butyl 4- (5- (4-chloro-7H-pyrrolo [2, 3-d] pyrimidin-6-yl) pyridin-2-yl) piperazine-1-carboxylate (0.9 g, 2.17 mmol) , (R) -3- (tert-butyl) -N- (1- (2-methyl-4- (4, 4, 5, 5-tetramethyl-1, 3, 2-dioxaborolan-2-yl) phenyl) ethyl) -1, 2, 4-oxadiazole-5-carboxamide (0.94 g, 2.28 mmol) , Na 2CO 3(0.46 g, 4.34 mmol) and Pd (dppf) Cl 2(79.3 mg, 0.108mmol) in dioxane (60 mL) and H 2O (10 mL) was stirred in a sealed tube at 100 ℃ overnight. After cooling, the reaction mixture was filtered and the solid was washed with 5 mL of MeOH and dried under vacuum to afford the product (1.02 g, 70.6%) . [M+H] += 666.0.
[0363]
Step 3: (R) -3- (tert-butyl) -N- (1- (2-methyl-4- (6- (6- (piperazin-1-yl) pyridin-3-yl) -7H- pyrrolo [2, 3-d] pyrimidin-4-yl) phenyl) ethyl) -1, 2, 4-oxadiazole-5-carboxamide, hydrogen chloride salt
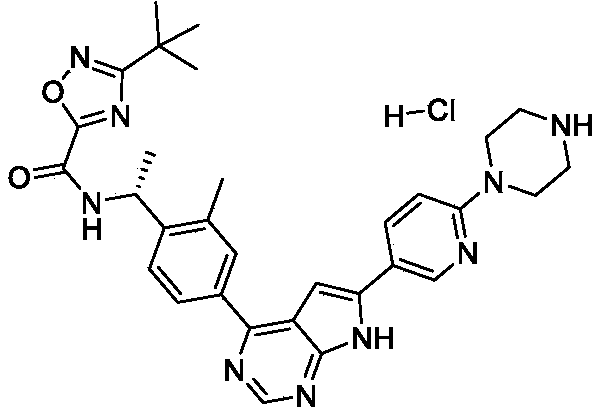
To a solution of tert-butyl (R) -4- (5- (4- (4- (1- (3- (tert-butyl) -1, 2, 4-oxadiazole-5-carboxamido) ethyl) -3-methylphenyl) -7H-pyrrolo [2, 3-d] pyrimidin-6-yl) pyridin-2-yl) piperazine-1-carboxylate (1.02 g, 1.53 mmol) in DCM (50 mL) in a round bottom flask was added HCl in dioxane (4 N, 35 mL) at 0 ℃. The mixture was stirred for 2 h at 20 ℃. The precipitate was collected with filtration and dried in vacuum to afford the product (0.92 g, 100%) . 1H NMR (400 MHz, DMSO) δ H13.53 (s, 1H) , 10.06 (d, J = 7.5 Hz, 1H) , 9.33 (s, 2H) , 9.00 (s, 1H) , 8.93 (s, 1H) , 8.35 (d, J = 8.7 Hz, 1H) , 8.05 (d, J = 8.1 Hz, 1H) , 7.99 (s, 1H) , 7.75 (d, J = 8.0 Hz, 1H) , 7.55 (s, 1H) , 7.12 (d, J = 8.9 Hz, 1H) , 5.50-5.28 (m, 1H) , 3.89 (s, 4H) , 3.20 (s, 4H) , 2.57 (s, 3H) , 1.56 (d, J = 6.9 Hz, 3H) , 1.38 (s, 9H) . [M+H] += 566.3.
[0366]
Step 4: (R) -3- (tert-butyl) -N- (1- (4- (6- (6- (4- ( (1- (4- (2, 4-dioxotetrahydropyrimidin-1 (2H) – yl) phenyl) piperidin-4-yl) methyl) piperazin-1-yl) pyridin-3-yl) -7H-pyrrolo [2, 3-d] pyrimidin-4-yl) -2- methylphenyl) ethyl) -1, 2, 4-oxadiazole-5-carboxamide
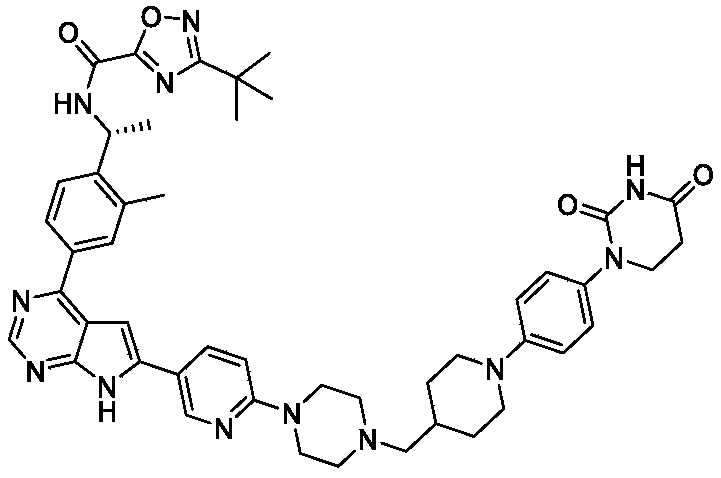
A mixture of (R) -3- (tert-butyl) -N- (1- (2-methyl-4- (6- (6- (piperazin-1-yl) pyridin-3-yl) -7H-pyrrolo [2, 3-d] pyrimidin-4-yl) phenyl) ethyl) -1, 2, 4-oxadiazole-5-carboxamide, hydrogen chloride salt (0.06 g, 0.1 mmol) , 1- (4- (2, 4-dioxotetrahydropyrimidin-1 (2H) -yl) phenyl) piperidine-4-carbaldehyde (0.033 g, 0.11 mmol) and NaOAc (8.2 mg, 0.1 mmol) in DCM/EtOH (30 mL/10 mL) was stirred in a round bottom flask for 1 h at 20 ℃. Then NaBH 3CN (12.6 mg, 0.2 mmol) was added. The mixture was stirred overnight at 20 ℃. The mixture was concentrated to dryness and purified with silica gel column chromatography (MeOH in DCM from 0%to 12%gradient elution) to give the product (0.049 g, 57.8%) . 1H NMR (400 MHz, DMSO) δ H12.60 (s, 1H) , 10.27 (s, 1H) , 9.97 (d, J =6.1 Hz, 1H) , 8.79 (d, J = 18.7 Hz, 2H) , 8.18 (d, J = 7.8 Hz, 1H) , 8.09 (d, J = 7.0 Hz, 1H) , 8.04 (s, 1H) , 7.67 (d, J = 7.7 Hz, 1H) , 7.30 (s, 1H) , 7.13 (d, J = 6.9 Hz, 2H) , 6.97-6.92 (m, 3H) , 5.41-5.34 (m, 1H) , 3.71-3.68 (m, 4H) , 3.64-3.56 (m, 4H) , 2.70-2.64 (m, 4H) , 2.53 (s, 3H) , 2.47-2.43 (m, 4H) , 2.25-2.19 (m, 2H) , 1.84-1.81 (m, 2H) , 1.75-1.70 (m, 1H) , 1.56 (t, J = 9.1 Hz, 3H) , 1.37 (s, 9H) , 1.28-1.18 (m, 2H) .
PAT
PAT
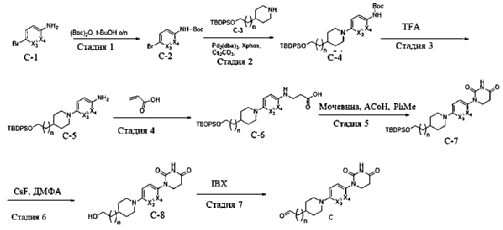
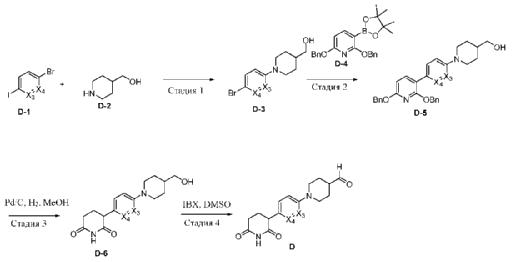
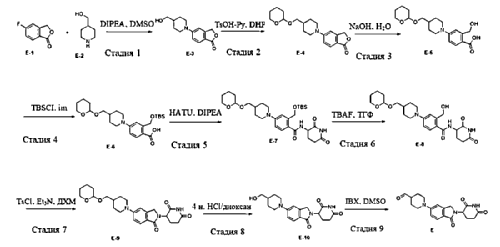
- Degradation of bruton’s tyrosine kinase (btk) by conjugation of btk inhibitors with e3 ligase ligand and methods of usePublication Number: WO-2021219070-A1Priority Date: 2020-04-30
- Degradation of Bruton’s tyrosine kinase (BTK) by conjugation of BTK inhibitors to E3 ligase ligands and methods of use thereofPublication Number: CN-115485278-APriority Date: 2020-04-30
- Degradation of bruton’s tyrosine kinase (btk) by conjugation of btk inidbitors with e3 ligase ligand and methods of usePublication Number: US-2023167118-A1Priority Date: 2020-04-30



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- [1]. Wang H, et al. BGB-16673, a selective BTK degrader, exhibits deeper inhibition of cancer cell signaling pathways and better efficacy in MCL models. Blood, 2024, 144: 5833.[2]. Wu Y, et al. Translational modeling to predict human pharmacokinetics and pharmacodynamics of a Bruton’s tyrosine kinase-targeted protein degrader BGB-16673. Br J Pharmacol. 2024 Dec;181(24):4973-4987. [Content Brief][3]. Hexiang Wang, et al. Degradation of bruton’s tyrosine kinase (btk) by conjugation of btk inhibitors with e3 ligase ligand and methods of use. WO2021219070A1. 2021-11-04.
/////////catadegbrutinib, Bruton tyrosine kinase degrader, antineoplastic, BGB-16673, BGB 16673, PF6GPZ4DYT, BTK-IN-29, Tacabrutideg

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
