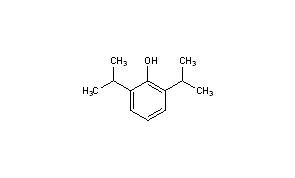
PropofolCAS Registry Number: 2078-54-8
CAS Name: 2,6-Bis(1-methylethyl)phenolAdditional Names: 2,6-diisopropylphenol; disoprofol
Manufacturers’ Codes: ICI-35868
Trademarks: Ansiven (Abbott); Diprivan (AstraZeneca); Disoprivan (AstraZeneca); Rapinovet (Schering-Plough Vet.)Molecular Formula: C12H18OMolecular Weight: 178.27Percent Composition: C 80.85%, H 10.18%, O 8.97%
Literature References: Prepn: A. J. Kolka et al.,J. Org. Chem.21, 712 (1956); 22, 642 (1957); G. G. Ecke, A. J. Kolka, US2831898 (1958 to Ethyl Corp.); T. J. Kealy, D. D. Coffman, J. Org. Chem.26, 987 (1961); B. E. Firth, T. J. Rosen, US4447657 (1984 to Universal Oil Products). Chromatographic study: J. K. Carlton, W. C. Bradbury, J. Am. Chem. Soc.78, 1069 (1956). Animal studies: J. B. Glen, Br. J. Anaesth.52, 731 (1980).Pharmacokinetics: H. K. Adam et al.,ibid. 743; idem,ibid.55, 97 (1983). Determn in blood: eidem,J. Chromatogr.223, 232 (1981). Comparative studies vs other injectable anesthetics: B. Kay, D. K. Stephenson, Anaesthesia35, 1182 (1980); D. V. Rutter et al.,ibid. 1188. Use in i.v. anesthesia: E. Major et al.,ibid.37, 541 (1982). Cardiovascular effects: D. Al-Khudhairi et al.,ibid. 1007. Pharmacology of emulsion formulation: J. B. Glen, S. C. Hunter, Br. J. Anaesth.56, 617 (1984). Series of articles on pharmacology and clinical experience: Postgrad. Med. J.61, Suppl. 3, 1-169 (1985).
Properties: bp30 136°. bp17 126°. mp 19°. nD20 1.5134. nD25 1.5111. d20 0.955.Melting point: mp 19°Boiling point: bp30 136°; bp17 126°Index of refraction:nD20 1.5134; nD25 1.5111Density: d20 0.955Therap-Cat: Anesthetic (intravenous).Therap-Cat-Vet: Intravenous anesthetic (dogs and cats).Keywords: Anesthetic (Intravenous).SYN

Prepn: A. J. Kolka et al., J. Org. Chem. 21, 712 (1956); 22, 642 (1957); G. G. Ecke, A. J. Kolka, US 2831898 (1958 to Ethyl Corp.); T. J. Kealy, D. D. Coffman, J. Org. Chem. 26, 987 (1961); B. E. Firth, T. J. Rosen, US 4447657 (1984 to Universal Oil Products).SYN

SYNhttps://pubs.acs.org/doi/pdf/10.1021/op400300t

A commercially viable manufacturing process for propofol (1) is described. The process avoids acid–base neutralization events during isolation of intermediate, 2,6-di-isopropylbenzoic acid (3) and crude propofol, and thus simplifies the synthesis on industrial scale to a considerable extent. Syntheses of five impurities/related substances (USP and EP) are also described.


SYN

SYN
Propofol is used during surgeries for sedation and an injectable grade with purity > 99.90% is desired by the medical community. An embodiment of the present invention provides an economically feasible, industrial process for the manufacture of high purity injectable grade Propofol. An embodiment of the present invention relates to a process and novel strategy for purification of 2,6-diisopropylphenol (Propofol) and similar products.
[0003] Propofol is a sterile injectable drug that appears in the USP, EP and IP Monographs. Drug product is manufactured by using high purity drug substance 2,6-di-isopropylphenol commonly known as Propofol.
[0004] Propofol is used to put patients to sleep and keep them asleep during general anesthesia for surgery or other medical procedures. It is used in adults as well as children 2 months and older. Propofol is frequently used as a sedative, and has a rapid onset of action and a short recovery period. Propofol slows the activity of brain and nervous system. Propofol is also used to sedate a patient who is under critical care and needs a mechanical ventilator (breathing machine). Propofol is a hypnotic alkylphenol derivative. When formulated for intravenous induction of sedation and hypnosis during anaesthesia, Propofol facilitates inhibitory neurotransmission mediated by gamma- Aminobutyric acid (GABA). Propofol is associated with minimal respiratory depression and has a short half-life with a duration of action of 2 to 10 minutes.
[0005] Propofol is commonly used parenteral anesthetic agent in the United States, extensively used for minor and outpatient surgical procedures because of its rapid onset and reversal of action, and in intensive care units (ICUs) for maintaining coma. Propofol has been associated with rare instances of idiosyncratic acute liver injury; in addition, prolonged high dose Propofol therapy can cause the “Propofol infusion syndrome” which is marked by brady arrhythmias, metabolic acidosis, rhabdomyolysis, hyperlipidemia and an enlarged or fatty liver.
[0006] Friedel-Craft’s alkylation of phenol using propylene gas in the presence of Lewis acid (LA) catalysts is a commonly used method for the synthesis of Propofol and is well documented in a number of publications and patents [Ecke, G. G., Kolka, A. J. US 2,831,898 A, 1958. Firth, B. E., Rosen, T. J. US 4,447,657, 1984. Akio, T., Yoshiaki, I., Hidekichi, H., Kiyoji, K., Takashi, K., Masanobu, M. EP 0169359A1, 1986. Ecke, G. G., Kolka, A. J. US 3,271,314, 1966. Napolitano, J. P. US 3,367,981 A, 1968. Goddard L. E. US 3,766,276, 1973. Firth, B. E. US 4,275,248, 1981, etc.]
[0007] A number of patents and published literature describe the manufacture of Propofol. US. Pat. No. 4,275,248; W0200034218; EP169359; US. Pat. No. 3,367,981; US. Pat. No.
3,271,314; US. Pat. No. 3,766,276; US. Pat. No. 2,831,898; US.Pat.No.2,207,753; GB1318100; U.S. Pat. No. 4,391,998; US. Pat. No. 4,774, 368; US. Pat. No. 5,589,598; US. Pat. No. 6,362,234; etc. EP 0511947, discloses purification of Propofol that is obtained by alkylation of phenol and purified by crystallization at -10 to -20°C (melting point of Propofol is 18°C). This patent also describes purification using non-polar solvents such as Petroleum ether or Hexane, where solvent residue is removed by distillation or evaporation and finally Propofol is obtained using fractional distillation under high vacuum.
[0008] Continuous separation of a mixture of Propofol with phenolic impurities and methanol is described in an U.S. Pat. No. 5,264,085. U.S. Pat. No. 5,705,039 describes the purification of impure 2,6-diisopropylphenol first using continuous distillation and then distilling pure Propofol under high vacuum.
[0009] Patent CN103360219A describes purification wherein 2,6-diisopropyl phenol is reacted with benzoyl chloride to generate ‘benzoic acid-2, 6-diisopropyl benzene ester’, which is then purified to yield Propofol. The patent discloses that an adsorbent is added at the rectifying stage, so that impurities with similar chemical structures and boiling points are effectively removed; the content of a single impurity in the product is not higher than 0.01%; the total impurity is not higher than 0.05%.
[0010] CN105601477A describes purification of Propofol wherein crude Propofol is purified with three-stage distillation method; the crude Propofol enters feeding tank protected by nitrogen and is charged into first-stage film distillation system through pump; then the product is fed to second-stage molecular distillation system and low boiling point impurities are removed; finally, the processed product is charged into third-stage molecular distiller through a pump, high-boiling-point impurities are separated, and the colourless or yellowish high-purity Propofol is obtained.
[0011] In another prior art disclosure, after completion of the reaction, the final product is isolated and purified by high-vacuum distillation. Alkylation of phenol using propylene gas at high pressure and high temperature is reported. Several impurities like 2,4-diisopropyl and 2,4,6-triisopropyl phenol are the major side products along with the corresponding Isopropyl ether. All these impurities need to be controlled at a limit of NMT 0.05% or less in the final API for it to be pharmaceutically acceptable. In another prior art disclosure, isopropanol was used as the propylating agent instead of direct propylene gas. In this method propylene is generated in situ using IPA and strong acid like sulfuric acid and catalysts like Aluminoslicate [See Baltalksne, A. E.; Zitsmanis, A. H. SU 443019, 1974. Jain, K. P., Edaki, D. U., Minhas H. S., Minhas G. S. WO/2011/ 161687 Al, 2011. Wu, M. US 4,391,998, 1983]
[0012] Another method is to use of protected phenol, where 4-chloro or 4-carboxylic acid substituted phenol is used as starting material along with Isopropanol in sulfuric acid, followed by removal of the 4-substituent to give Propofol [Baltalksne, A. E.; Zitsmanis, A. H. SU 443019, 1974. Jain, K. P., Edaki, D. U., Minhas H. S., Minhas G. S. WO/2011/ 161687 Al, 2011. Wu, M. US 4,391,998, 1983. Tsutsumi, S.; Yoshizawa, T.; Koyama, K. Nippon Kagaku Zasshi 1956, 77, 737-738. Paiocchi, M. US 5,589,598, 1996. Nieminen, K., Essen, P. US 5,175,376, 1992. Keller, S., Schlegel, J. WO/2012/152665 Al, 2012.] The final purification is carried out by high- vacuum distillation to get highly pure Propofol. Since the para position is blocked, related impurities such as 2,4-isopropyl and 2,4,6-triisopropyl derivatives are avoided. In this approach, intermediate is purified before converting to crude Propofol using either de-chlorination by hydrogenation or de-carboxylation before vacuum distillation for final purification.
[0013] It is reported in the literature that 4-hydroxybenzoic acid is used as starting material for alkylation with isopropyl alcohol in sulfuric acid. In that method 2,6-diisopropyl-4-hydroxy benzoic acid gets formed, which is extracted in toluene either in presence of an acid or the impurities are extracted in toluene under alkaline condition. The decarboxylation is carried out using solvents like monoethylene glycol or ethoxyethanol at high temperature. At the end of decarboxylation, crude Propofol is isolated by extracting into toluene. The advantage is Propofol does not form sodium salt under the conditions, but all other acidic impurities form sodium salt and thus do not get extracted in toluene. The toluene containing Crude Propofol is distilled to recover toluene and then vacuum distilled to obtain pure Propofol. [Chen, T; Chen, X.; Bois-Choussy, M.; Zhu, J. J. Am. Chem. Soc. 2006, 128, 87-89. Lau, S.; Keay, B. Can. J. Chem. 2001, 79, 1541-1545]
[0014] In summary, strategies disclosed in prior art for the production of 2,6-diisopropylphenol (Propofol) predominantly involve synthesis starting from phenol or by using protected 4-position of phenol like, 4-hydroxybenzoic acid, 4-chlorophenol (references: Baltalksne, A. E.; Zitsmanis, A. H. SU 443019, 1974. Jain, K. P., Edaki, D. U., Minhas H. S., Minhas G. S. WO/2011/ 161687 Al, 2011. Wu, M. US 4,391,998, 1983. Tsutsumi, S.; Yoshizawa, T.; Koyama, K. Nippon Kagaku Zasshi 1956, 77, 737-738. Paiocchi, M. US 5,589,598, 1996. Nieminen, K., Essen, P. US 5,175,376, 1992. Keller, S., Schlegel, J. WO/2012/152665 Al, 2012). Processes described in the literature generally propose purification of crude 2,6-diisopropylphenol by ‘high vacuum distillation or molecular distillation’.
[0015] The phenols are susceptible to oxidation, formation of polymeric and color impurities. There are processes where repeated vacuum distillation has been carried out to obtain desired purity of product. Sometimes, to reduce the oxidized and colored impurities, reduction of impurities by catalytic hydrogenation is also used.
[0016] Propofol that does not meet Pharmaceutical grade may be manufactured by several processes generally known to persons of skill in the art and described in prior art, but purification of Propofol to consistently achieve high purity required for the injectable drug substance using an economical and industrial process remains a challenge.
Example 1:
[0033] Commercially available concentrated sulfuric acid (30 Kg) was diluted with water (2.26 Kg) at low temperature (0-15°C). Methyl 4-hydroxybenzoate (5 Kg 32.79 mol.) was added to this diluted sulfuric acid at 5 to 10 °C with stirring. After complete addition, isopropyl alcohol (5.9 Kg 98.16 mol.) was gradually added to the reaction content, controlling the temperature at 0-15 °C. The reaction mixture was then heated at 60-70°C and continued to complete di-isopropylation and ester hydrolysis to yield methyl-4-hydroxybenzoate. The conversion was checked on TLC or by HPLC for the complete conversion of methyl-4 hydroxybenzoate to 3, 5 -Diisopropyl 4-hydroxybenzoic acid.
[0034] The reaction contents were cooled at room temperature and carefully charged into a stirred, precooled mixture of water (50 L) and Toluene (40 L) at (0 to 5°C). The mixture was stirred and maintained below 15°C for about 30 to 60 minutes.
[0035] The content was then heated at 25 to 30°C, stirred for 30 min., allowed to settle into two layers. The water layer was extracted again with toluene and discarded. The toluene layers, containing the product 3, 5-Diisopropyl 4-hydroxybenzoic acid, were combined and extracted with about 25 L of 10 % NaOH. The aqueous layer containing the sodium salt of 3, 5 -Di-isopropyl 4-hydroxybenzoic acid was acidified with concentrated HC1 (about 9 Kg) to precipitate 3, 5-Diisopropyl 4-hydroxybenzoic acid, filtered, and washed with water (about 50 L) to yield 3, 5 -diisopropyl 4-hydroxybenzoic acid (about 45-60 %)
[0036] To the mixture of 3, 5-diisopropyl 4-hydroxybenzoic acid (3 Kg, 13.5 mol.) in ethylene glycol (5.0 Kg, 80.55 mol.) was added sodium hydroxide (1.25 Kg, 31.25 mol.) for decarboxylation. The reaction mixture was heated at 145 ± 5°C till completion of
decarboxylation by monitoring using TLC or HPLC (or solubility in bicarbonate of precipitated product). After complete decarboxylation, the reaction mixture was cooled at 40 to 45 °C, under nitrogen environment and diluted with water (about 15 L) and allowed to settle. The oily product layer was separated and washed with water (6L) to isolate crude Propofol (i.e., 2,6-diisopropyl phenol) and stored under nitrogen. The isolated volatile Crude Propofol (along with carry over ppm ethylene glycol and NaOH) was then subjected to steam distillation purification process as described below.
[0037] The Crude Propofol is purified by using one of the steam distillation processes as described below.
[0038] The Crude Propofol layer is added to purified water in a reactor (preferably glass lined reactor), and slowly heated to boiling to co-distil Pure Propofol along with water under normal atmospheric pressure and the high volatile initial fraction is isolated first. The biphasic layers of main distillate, are separated and the liquid layer of Propofol is treated with dehydrating agent to absorb dissolved moisture in Pure Propofol under nitrogen or argon. The transparent Pure Propofol liquid layer is then filtered through ultrafme Micron filter (0.45 and 0.2 micron) under nitrogen (or argon) pressure and isolated in controlled environment to obtain pharmaceutical injectable grade Pure Propofol of more than 99.90% purity.
[0039] The Crude Propofol liquid layer is charged into a reactor with steam distillation arrangement, like steam purging dip tube, column, heat exchanger and receivers. Pure steam is purged in the reactor at controlled pressure to co-distil Pure Propofol with water. The layers are allowed settle and water layer is kept aside for recirculation. The transparent Pure Propofol transparent liquid layer is then filtered through ultrafme Micron filter (0.45 and 0.2 micron) under nitrogen (or argon) pressure and isolated in controlled environment to obtain pharmaceutical injectable grade Pure Propofol of more than 99.90% purity.
[0040] The Crude Propofol layer is added to purified water in a reactor (preferably glass lined or Hastelloy reactor) and slowly heated at boiling to co-distil Pure Propofol along with water under mild vacuum. The biphasic layers are separated and the liquid layer of Propofol is treated with dehydrating agent to absorb dissolved moisture in Pure Propofol under nitrogen (or argon). The transparent Pure Propofol liquid layer is then filtered through ultrafme Micron filter (0.45 and 0.2 micron) under nitrogen (or argon) pressure and isolated in controlled environment to obtain pharmaceutical injectable grade Pure Propofol of more than 99.90% purity.
[0041] The Crude Propofol layer is added to reactor containing purified water and 0.1 to 1% antioxidant and 0.1 to 0.5% sodium hydroxide and slowly heated to boiling to co-distil Pure Propofol along with water. The biphasic layers are separated and the liquid layer of Propofol is treated or passed through column packed with dehydrating agent to absorb dissolved moisture in Pure Propofol. The transparent Pure Propofol liquid layer is then filtered through ultrafme Micron filter (0.45 and 0.2 micron) under nitrogen (or argon) pressure and isolated in controlled environment to obtain pharmaceutical injectable grade Pure Propofol of more than 99.90% purity.
[0042] The crude Propofol liquid layer is treated with preferably neutral or basic activated carbon (about 2-5%) and filtered under nitrogen. The filtered liquid is collected, under nitrogen, in distillation reactor containing purified water is slowly heated to boiling to co-distil Pure Propofol along with water under normal pressure or mild vacuum. The co-distilled biphasic layers are separated and the liquid layer of Propofol, is treated under nitrogen, with or passed through column packed with dehydrating agent to absorb dissolved moisture trapped in Pure Propofol. The transparent Pure Propofol liquid layer is then filtered through ultrafme Micron filter (0.45 and 0.2 micron) under nitrogen (or argon) pressure and isolated in controlled environment to obtain pharmaceutical injectable grade Pure Propofol of more than 99.90% purity.
Example No. 2:
[0043] Friedel-Crafts reaction was performed as described in Example 1. Decarboxylation was performed by using KOH instead of NaOH by following the same procedure as described in Example 1.
Example No. 3:
[0044] Decarboxylation was performed as per operations described in Example 1. After complete decarboxylation, the reaction mixture was cooled at 40 to 45°C, under nitrogen environment and diluted with water (about 15 L) The biphasic mixture subjected to steam distillation by any of the purification methods described in Example 1.
Example No. 4:
[0045] Friedel-Crafts reaction was performed as described in Example 1. The reaction contents were cooled at room temperature and carefully charged at 0 to 5°C into a sodium hydroxide solution to basic pH at stirred. The aqueous alkaline solution was extracted twice with toluene or hexane. The aqueous layer was acidified with HC1 to precipitate 3, 5-diisopropyl-4-hydroxybenzoic acid. The wet product was washed with water, dried and decarboxylated using sodium hydroxide in ethylene glycol as solvent at 145±5°C. The reaction contents were cooled to room temperature, diluted with water and acidified and then Crude Propofol was extracted twice in toluene. The toluene layer was washed with water, bicarbonate and with water then distilled to obtain crude oily layer of Propofol (>99% pure). This Crude Propofol was then purified by using purification steam distillation by any of the purification methods described in Example 1.
Example 5:
[0046] Continuous steam distillation of crude Propofol by purging pure steam. Continuous steam distillation of Crude Propofol was carried out using a feed pump for feeding liquid Crude Propofol (prepared by one of the processes described in this application or other literature) to the steam distillation system connected to a pure steam generator. Steam at 1-10 kg pressure was purged in the steam distillation system at controlled rate and the co-distilled Pure Propofol with water was cooled using heat exchanger and continuous separator. The residue was discharged from bottom valve at defined time intervals. The water layer was recycled to steam generator and Pure Propofol was dehydrated, filtered and collected in controlled environment as described in Example 1.
Propofol, marketed as Diprivan, among other names, is a short-acting medication that results in a decreased level of consciousness and a lack of memory for events.[4] Its uses include the starting and maintenance of general anesthesia, sedation for mechanically ventilated adults, and procedural sedation.[4] It is also used for status epilepticus if other medications have not worked.[4] It is given by injection into a vein, and the maximum effect takes about two minutes to occur and typically lasts five to ten minutes.[4] Propofol is also used for medical assistance in dying in Canada.[5]
Common side effects of propofol include an irregular heart rate, low blood pressure, a burning sensation at the site of injection and the cessation of breathing.[4] Other serious side effects may include seizures, infections due to improper use, addiction, and propofol infusion syndrome with long-term use.[4] The medication appears to be safe for use during pregnancy but has not been well studied for use in this case.[4] It is not recommended for use during a cesarean section.[4] It is not a pain medication, so opioids such as morphine may also be used,[6] however whether or not they are always needed is not clear.[7] Propofol is believed to work at least partly via a receptor for GABA.[4]
Propofol was discovered in 1977 and approved for use in the United States in 1989.[4][8] It is on the World Health Organization’s List of Essential Medicines[9] and is available as a generic medication.[4] It has been referred to as milk of amnesia (a play on “milk of magnesia“), because of the milk-like appearance of the intravenous preparation, and because of its tendency to suppress memory recall.[10][11] Propofol is also used in veterinary medicine for anesthesia.[12][13]
Medical uses
Anesthesia
To induce general anesthesia, propofol is the drug used almost always, having largely replaced sodium thiopental.[14][6] It can also be administered as part of an anesthesia maintenance technique called total intravenous anesthesia, using either manually programmed infusion pumps or computer-controlled infusion pumps in a process called target controlled infusion (TCI). Propofol is also used to sedate individuals who are receiving mechanical ventilation but not undergoing surgery, such as patients in the intensive care unit.[15][16] In critically ill patients, propofol is superior to lorazepam both in effectiveness and overall cost.[17] Propofol is relatively inexpensive compared to medications of similar use due to shorter ICU stay length.[17] One of the reasons propofol is thought to be more effective (although it has a longer half-life than lorazepam) is because studies have found that benzodiazepines like midazolam and lorazepam tend to accumulate in critically ill patients, prolonging sedation.[17] Propofol has also been suggested as a sleep aid in critically ill adults in the ICU, however, the effectiveness of this medicine at replicating the mental and physical aspects of sleep for people in the ICU are not clear.[16]
Propofol is often used instead of sodium thiopental for starting anesthesia because recovery from propofol is more rapid and “clear”.[citation needed]
Propofol can be run through a peripheral IV or central line. Propofol is frequently paired with fentanyl (for pain relief) in intubated and sedated people.[18] Both are compatible in IV form.[18]
Procedural sedation
Propofol is also used for procedural sedation. Its use in these settings results in a faster recovery compared to midazolam.[19] It can also be combined with opioids or benzodiazepines.[20][21][22] Because of its rapid induction and recovery time, propofol is also widely used for sedation of infants and children undergoing MRI.[23] It is also often used in combination with ketamine with minimal side effects.[24]
COVID-19
In March 2021, the U.S. Food and Drug Administration (FDA) issued an emergency use authorization (EUA) for Propofol‐Lipuro 1% to maintain sedation via continuous infusion in people greater than age sixteen with suspected or confirmed COVID‑19 who require mechanical ventilation in an intensive care unit ICU setting.[25][26][27][28] In the circumstances of this public health emergency, it would not be feasible to require healthcare providers to seek to limit Fresenius Propoven 2% Emulsion or Propofol-Lipuro 1% only to be used for patients with suspected or confirmed COVID‑19; therefore, this authorization does not limit use to such patients.[28]
Other uses
Executions
The US state of Missouri added propofol to its execution protocol in April 2012. However, Governor Jay Nixon halted the first execution by the administration of a lethal dose of propofol in October 2013 following threats from the European Union to limit the drug’s export if it were used for that purpose.[29][30] The United Kingdom had already banned the export of medicines or veterinary medicines containing propofol to the United States.[31]
Recreational use
Recreational use of the drug via self-administration has been reported[32][33] but is relatively rare due to its potency and the level of monitoring required for safe use.[citation needed] Critically, a steep dose-response curve makes recreational use of propofol very dangerous, and deaths from self-administration continue to be reported.[34][35] The short-term effects sought via recreational use include mild euphoria, hallucinations, and disinhibition.[36][37]
Recreational use of the drug has been described among medical staff, such as anesthetists who have access to the drug.[38][39] It is reportedly more common among anesthetists on rotations with short rest periods, as usage generally produces a well-rested feeling.[40] Long-term use has been reported to result in addiction.[38][41]
Attention to the risks of off-label use of propofol increased in August 2009 due to the Los Angeles County coroner’s conclusion that music icon Michael Jackson died from a mixture of propofol and the benzodiazepine drugs lorazepam, midazolam, and diazepam on June 25, 2009.[42][43][44][45] According to a July 22, 2009 search warrant affidavit unsealed by the district court of Harris County, Texas, Jackson’s physician, Conrad Murray, administered 25 milligrams of propofol diluted with lidocaine shortly before Jackson’s death.[43][44][46] Even so, as of 2016, propofol was not on a US Drug Enforcement Administration schedule.[40][47]
Side effects
One of propofol’s most common side effects is pain on injection, especially in smaller veins. This pain arises from activation of the pain receptor, TRPA1,[48] found on sensory nerves and can be mitigated by pretreatment with lidocaine.[49] Less pain is experienced when infused at a slower rate in a large vein (antecubital fossa). Patients show considerable variability in their response to propofol, at times showing profound sedation with small doses.
Additional side effects include low blood pressure related to vasodilation, transient apnea following induction doses, and cerebrovascular effects. Propofol has more pronounced hemodynamic effects relative to many intravenous anesthetic agents.[50] Reports of blood pressure drops of 30% or more are thought to be at least partially due to inhibition of sympathetic nerve activity.[51] This effect is related to the dose and rate of propofol administration. It may also be potentiated by opioid analgesics.[52] Propofol can also cause decreased systemic vascular resistance, myocardial blood flow, and oxygen consumption, possibly through direct vasodilation.[53] There are also reports that it may cause green discolouration of the urine.[54]
Although propofol is heavily used in the adult ICU setting, the side effects associated with propofol seem to be of greater concern in children. In the 1990s, multiple reported deaths of children in ICUs associated with propofol sedation prompted the FDA to issue a warning.[55]
As a respiratory depressant, propofol frequently produces apnea. The persistence of apnea can depend on factors such as premedication, dose administered, and rate of administration, and may sometimes persist for longer than 60 seconds.[56] Possibly as the result of depression of the central inspiratory drive, propofol may produce significant decreases in respiratory rate, minute volume, tidal volume, mean inspiratory flow rate, and functional residual capacity.[50]
Diminishing cerebral blood flow, cerebral metabolic oxygen consumption, and intracranial pressure are also characteristics of propofol administration.[57] In addition, propofol may decrease intraocular pressure by as much as 50% in patients with normal intraocular pressure.[58]
A more serious but rare side effect is dystonia.[59] Mild myoclonic movements are common, as with other intravenous hypnotic agents. Propofol appears to be safe for use in porphyria, and has not been known to trigger malignant hyperpyrexia.[citation needed]
Propofol is also reported to induce priapism in some individuals,[60][61] and has been observed to suppress REM sleep stage and to worsen the poor sleep quality in some patients.[62]
As with any other general anesthetic agent, propofol should be administered only where appropriately trained staff and facilities for monitoring are available, as well as proper airway management, a supply of supplemental oxygen, artificial ventilation, and cardiovascular resuscitation.[63]
Because of its lipid base, some hospital facilities require the IV tubing (of continuous propofol infusions) to be changed after 12 hours. This is a preventive measure against microbial growth and infection.[64]
Propofol infusion syndrome
Main article: Propofol infusion syndrome
A rare, but serious, side effect is propofol infusion syndrome. This potentially lethal metabolic derangement has been reported in critically ill patients after a prolonged infusion of high-dose propofol, sometimes in combination with catecholamines and/or corticosteroids.[65]
Interactions
The respiratory effects of propofol are increased if given with other respiratory depressants, including benzodiazepines.[66]
Pharmacology
Pharmacodynamics
Propofol has been proposed to have several mechanisms of action,[67][68][69] both through potentiation of GABAA receptor activity and therefore acting as a GABAA receptor positive allosteric modulator, thereby slowing the channel-closing time. At high doses, propofol may be able to activate GABAA receptors in the absence of GABA, behaving as a GABAA receptor agonist as well.[70][71][72] Propofol analogs have been shown to also act as sodium channel blockers.[73][74] Some research has also suggested that the endocannabinoid system may contribute significantly to propofol’s anesthetic action and to its unique properties.[75] EEG research upon those undergoing general anesthesia with propofol finds that it causes a prominent reduction in the brain’s information integration capacity.[76]
Pharmacokinetics
A 20 ml ampoule of 1% propofol emulsion, as sold in Australia by Sandoz
Propofol is highly protein-bound in vivo and is metabolised by conjugation in the liver.[77] The half-life of elimination of propofol has been estimated to be between 2 and 24 hours. However, its duration of clinical effect is much shorter, because propofol is rapidly distributed into peripheral tissues. When used for IV sedation, a single dose of propofol typically wears off within minutes. Propofol is versatile; the drug can be given for short or prolonged sedation, as well as for general anesthesia. Its use is not associated with nausea as is often seen with opioid medications. These characteristics of rapid onset and recovery along with its amnestic effects[78] have led to its widespread use for sedation and anesthesia.
History
John B. Glen, a veterinarian and researcher at Imperial Chemical Industries (ICI) spent 13 years developing propofol, an effort which led to the awarding to him of the prestigious 2018 Lasker Award for clinical research. Propofol was originally developed as ICI 35868. It was chosen for development after extensive evaluation and structure–activity relationship studies of the anesthetic potencies and pharmacokinetic profiles of a series of ortho-alkylated phenols.[79]
First identified as a drug candidate in 1973, clinical trials followed in 1977, using a form solubilised in cremophor EL.[80] However, due to anaphylactic reactions to cremophor, this formulation was withdrawn from the market and subsequently reformulated as an emulsion of a soya oil/propofol mixture in water. The emulsified formulation was relaunched in 1986 by ICI (now AstraZeneca) under the brand name Diprivan. The currently available preparation is 1% propofol, 10% soybean oil, and 1.2% purified egg phospholipid as an emulsifier, with 2.25% glycerol as a tonicity-adjusting agent, and sodium hydroxide to adjust the pH. Diprivan contains EDTA, a common chelation agent, that also acts alone (bacteriostatically against some bacteria) and synergistically with some other antimicrobial agents. Newer generic formulations contain sodium metabisulfite or benzyl alcohol as antimicrobial agents. Propofol emulsion is a highly opaque white fluid due to the scattering of light from the tiny (about 150-nm) oil droplets it contains: Tyndall Effect.
Developments
A water-soluble prodrug form, fospropofol, has been developed and tested with positive results. Fospropofol is rapidly broken down by the enzyme alkaline phosphatase to form propofol. Marketed as Lusedra, this formulation may not produce the pain at injection site that often occurs with the conventional form of the drug. The U.S. Food and Drug Administration (FDA) approved the product in 2008.[81] However fospropofol is a Schedule IV controlled substance with the DEA ACSCN of 2138 in the United States unlike propofol.[82]
By incorporation of an azobenzene unit, a photoswitchable version of propofol (AP2) was developed in 2012, that allows for optical control of GABAA receptors with light.[83] In 2013, a propofol binding site on mammalian GABAA receptors has been identified by photolabeling using a diazirine derivative.[84] Additionally, it was shown that the hyaluronan polymer present in the synovia can be protected from free-radical depolymerization by propofol.[85]

NEW DRUG APPROVALS
ONE TIME
$10.00
References
- ^ “Propofol”. Drugs.com. Retrieved 2 January 2019.
- ^ Ruffle JK (November 2014). “Molecular neurobiology of addiction: what’s all the (Δ)FosB about?”. Am J Drug Alcohol Abuse. 40 (6): 428–437. doi:10.3109/00952990.2014.933840. PMID 25083822. S2CID 19157711.
Propofol is a general anesthetic, however its abuse for recreational purpose has been documented (120). Using control drugs implicated in both ΔFosB induction and addiction (ethanol and nicotine), similar ΔFosB expression was apparent when propofol was given to rats. Moreover, this cascade was shown to act via the dopamine D1 receptor in the NAc, suggesting that propofol has abuse potential (119)
- ^ “Diprivan- propofol injection, emulsion”. DailyMed. Retrieved 17 April 2021.
- ^ Jump up to:a b c d e f g h i j k l m n “Propofol”. The American Society of Health-System Pharmacists. Archived from the original on 9 October 2016. Retrieved 21 January 2017.
- ^ Divisions of Family Practice Medical Assistance in Dying (MAiD): Protocols and Procedures Handbook.
- ^ Jump up to:a b Miner, JR; Burton, JH (August 2007). “Clinical practice advisory: Emergency department procedural sedation with propofol”. Annals of Emergency Medicine. 50 (2): 182–7. doi:10.1016/j.annemergmed.2006.12.017. PMID 17321006.
- ^ Wakai, A; Blackburn, C; McCabe, A; Reece, E; O’Connor, G; Glasheen, J; Staunton, P; Cronin, J; Sampson, C; McCoy, SC; O’Sullivan, R; Cummins, F (29 July 2015). “The use of propofol for procedural sedation in emergency departments”. The Cochrane Database of Systematic Reviews. 7 (7): CD007399. doi:10.1002/14651858.CD007399.pub2. PMC 6517206. PMID 26222247.
- ^ Miller’s Anesthesia (8 ed.). Elsevier Health Sciences. 2014. p. 920. ISBN 9780323280112.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ Euliano TY, JS (2004). “A brief pharmacology related to anesthesia”. Essential anesthesia: from science to practice. Cambridge, UK: Cambridge University Press. p. 173. ISBN 978-0-521-53600-4. Retrieved 2 June 2009.
- ^ MD, David M. Novick (2017). A Gastroenterologist’s Guide to Gut Health: Everything You Need to Know About Colonoscopy, Digestive Diseases, and Healthy Eating. Rowman & Littlefield. p. 15. ISBN 9781442271999.
- ^ “Anesthesia Medications”. Veterinary Dentistry for the Small Animal Technician. Hoboken: Wiley. 2013. ISBN 9781118694800.
- ^ “PropoFlo (propofol) for Animal Use”. Drugs.com. Retrieved 13 February 2019.
- ^ “Discovery and development of propofol, a widely used anesthetic”. The Lasker Foundation. Retrieved 8 September2020.
Propofol is used today to initiate anesthesia in nearly 100% of general anesthesia cases worldwide.
- ^ Barr, Juliana (1995). “Propofol”. International Anesthesiology Clinics. 33 (1): 131–154. doi:10.1097/00004311-199500000-00008. ISSN 0020-5907.
- ^ Jump up to:a b Lewis, Sharon R.; Schofield-Robinson, Oliver J.; Alderson, Phil; Smith, Andrew F. (8 January 2018). “Propofol for the promotion of sleep in adults in the intensive care unit”. The Cochrane Database of Systematic Reviews. 1: CD012454. doi:10.1002/14651858.CD012454.pub2. ISSN 1469-493X. PMC 6353271. PMID 29308828.
- ^ Jump up to:a b c Cox, CE; Reed, SD; Govert, JA; Rodgers, JE; Campbell-Bright, S; Kress, JP; Carson, SS (March 2008). “Economic evaluation of propofol and lorazepam for critically ill patients undergoing mechanical ventilation”. Crit Care Med. 36 (3): 706–14. doi:10.1097/CCM.0B013E3181544248. PMC 2763279. PMID 18176312.
- ^ Jump up to:a b Isert, Peter R.; Lee, Doris; Naidoo, Daya; Carasso, Melanie L.; Kennedy, Ross A. (June 1996). “Compatibility of propofol, fentanyl, and vecuronium mixtures designed for potential use in anesthesia and patient transport”. Journal of Clinical Anesthesia. 8 (4): 329–336. doi:10.1016/0952-8180(96)00043-8. PMID 8695138.
- ^ McQuaid, KR.; Laine, L. (May 2008). “A systematic review and meta-analysis of randomized, controlled trials of moderate sedation for routine endoscopic procedures”. Gastrointest Endosc. 67 (6): 910–23. doi:10.1016/j.gie.2007.12.046. PMID 18440381.
- ^ Canadian National Forumulary 2010
- ^ Appleton & Lange Nursing Drug Guide, 1999
- ^ Numorphan® (oxymorphone) package insert (English), Endo 2009
- ^ Machata, AM; Willschke, H; Kabon, B; Kettner, SC; Marhofer, P (August 2008). “Propofol-based sedation regimen for infants and children undergoing ambulatory magnetic resonance imaging”. British Journal of Anaesthesia. 101 (2): 239–43. doi:10.1093/bja/aen153. PMID 18534971.
- ^ Yan, JW; McLeod, SL; Iansavitchene, A (20 August 2015). “Ketamine-Propofol Versus Propofol Alone for Procedural Sedation in the Emergency Department: A Systematic Review and Meta-analysis”. Academic Emergency Medicine. 22 (9): 1003–13. doi:10.1111/acem.12737. PMID 26292077.
- ^ https://www.bbraunusa.com/content/dam/b-braun/us/website/company/covid-files/210319_Propofol_EUA_Submission_to_FDA_hcp.pdf
- ^ https://www.fda.gov/media/146680/download
- ^ https://www.fda.gov/media/146681/download
- ^ Jump up to:a b “Emergency Use Authorization”. U.S. Food and Drug Administration (FDA). Retrieved 17 April 2021.
- ^ Death Row Improvises, Lacking Lethal Mix Archived 8 July 2017 at the Wayback Machine, By RICK LYMAN, New York Times, 18 August 2013
- ^ After EU threats, Missouri halts execution by Propofol injectionArchived 12 October 2013 at the Wayback Machine Al Jazeera America 12 October 2013
- ^ Article 4A of Export Control Order 2008 – provisions supplementing “the torture Regulation”
- ^ Riezzo I, Centini F, Neri M, Rossi G, Spanoudaki E, Turillazzi E, Fineschi V (2009). “Brugada-like EKG pattern and myocardial effects in a chronic propofol abuser”. Clin Toxicol. 47 (4): 358–63. doi:10.1080/15563650902887842. PMID 19514884. S2CID 22531823.
- ^ Belluck, Pam (6 August 2009). “With High-Profile Death, Focus on High-Risk Drug”. New York Times. Archived from the original on 11 November 2011. Retrieved 7 August 2009.
- ^ Iwersen-Bergmann S, Rösner P, Kühnau HC, Junge M, Schmoldt A (2001). “Death after excessive propofol abuse”. International Journal of Legal Medicine. 114 (4–5): 248–51. CiteSeerX 10.1.1.528.7395. doi:10.1007/s004149900129. PMID 11355404. S2CID 25963187.
- ^ Kranioti EF, Mavroforou A, Mylonakis P, Michalodimitrakis M (22 March 2007). “Lethal self-administration of propofol (Diprivan): A case report and review of the literature”. Forensic Science International. 167 (1): 56–8. doi:10.1016/j.forsciint.2005.12.027. PMID 16431058.
- ^ In Sweetman SC (Ed.). Martindale: The Complete Drug Reference 2005. 34th Edn London pp. 1305–7
- ^ Baudoin Z. General anesthetics and anesthetic gases. In Dukes MNG and Aronson JK (Eds.). Meyler’s Side Effects of Drugs 2000. 14th Edn Amsterdam pp. 330
- ^ Jump up to:a b Roussin A, Montastruc JL, Lapeyre-Mestre M (21 October 2007). “Pharmacological and clinical evidences on the potential for abuse and dependence of propofol: a review of the literature”. Fundamental and Clinical Pharmacology. 21 (5): 459–66. doi:10.1111/j.1472-8206.2007.00497.x. PMID 17868199. S2CID 22477291.
- ^ C.F. Ward, 2008, Propofol: Dancing with a “White Rabbit”Archived 8 September 2017 at the Wayback Machine, CSA Bulletin, pp. 61–63, accessed 24 November 2014.
- ^ Jump up to:a b Charatan F (2009). “Concerns mount over recreational use of propofol among US healthcare professionals”. BMJ. 339: b3673. doi:10.1136/bmj.b3673. PMID 19737827. S2CID 9877560.
- ^ Bonnet U, Harkener J, Scherbaum N (June 2008). “A case report of propofol dependence in a physician”. J Psychoactive Drugs. 40(2): 215–7. doi:10.1080/02791072.2008.10400634. PMID 18720673. S2CID 15779389.
- ^ Moore, Solomon (28 August 2009). “Jackson’s Death Ruled a Homicide”. The New York Times. Archived from the original on 14 November 2013.
- ^ Jump up to:a b Surdin, Ashley (25 August 2009). “Coroner Attributes Michael Jackson’s Death to Propofol”. The Washington Post. Archivedfrom the original on 9 November 2012. Retrieved 22 May 2010.
- ^ Jump up to:a b Itzkoff, Dave (24 August 2009). “Coroner’s Findings in Jackson Death Revealed”. The New York Times. Archived from the original on 11 June 2010. Retrieved 22 May 2010.
- ^ “Jackson’s Death: How Dangerous Is Propofol?”. Time. 25 August 2009. Archived from the original on 25 July 2010. Retrieved 22 May 2010.
- ^ “Michael Jackson search warrant”. Scribd. Archived from the original on 5 March 2016. Retrieved 12 August 2015.
- ^ DEA may limit drug eyed in Jackson case. Associated Press.15 July 2009.
- ^ Matta, J. A.; Cornett, P. M.; Miyares, R. L.; Abe, K.; Sahibzada, N.; Ahern, G. P. (2008). “General anesthetics activate a nociceptive ion channel to enhance pain and inflammation”. Proceedings of the National Academy of Sciences. 105 (25): 8784–8789. doi:10.1073/pnas.0711038105. PMC 2438393. PMID 18574153.
- ^ “Propofol Drug Information, Professional”. m drugs.com. Archived from the original on 23 January 2007. Retrieved 2 January 2007.
- ^ Jump up to:a b Sebel, PS; Lowden, JD (1989). “Propofol: a new intravenous anesthetic”. Anesthesiology. 71 (2): 260–77. doi:10.1097/00000542-198908000-00015. PMID 2667401. S2CID 34331379.
- ^ Robinson, B; Ebert, T; O’Brien, T; et al. (1997). “Mechanisms whereby propofol mediates peripheral vasodilation in humans (1997)”. Anesthesiology. 86 (1): 64–72. doi:10.1097/00000542-199701000-00010. PMID 9009941. S2CID 31288656.
- ^ “New awakening in anaesthesia—at a price”. Lancet. 329 (8548): 1469–70. 1987. doi:10.1016/s0140-6736(87)92214-8. S2CID 28545161.
- ^ Larijani, G; Gratz, I; Afshar, M; et al. (1989). “Clinical pharmacology of propofol: an intravenous anesthetic agent [published erratum appears in DICP 1990 Jan; 24: 102]”. DICP. 23(10): 743–9. doi:10.1177/106002808902301001. PMID 2683416. S2CID 43010280.
- ^ Jung SL, Hyun SJ, Byeong JP (2013). “Green discoloration of urine after propofol infusion”. Korean Journal of Anesthesiology. 65 (2): 177–9. doi:10.4097/kjae.2013.65.2.177. PMC 3766788. PMID 24024005.
- ^ Parke, T. J.; Stevens, J. E.; Rice, A. S.; Greenaway, C. L.; Bray, R. J.; Smith, P. J.; Waldmann, C. S.; Verghese, C. (12 September 1992). “Metabolic acidosis and fatal myocardial failure after propofol infusion in children: five case reports”. BMJ. 305 (6854): 613–616. doi:10.1136/bmj.305.6854.613. ISSN 0959-8138. PMC 1883365. PMID 1393073.
- ^ Langley, M; Heel, R (1988). “Propofol. A review of its pharmacodynamic and pharmacokinetic properties and use as an intravenous anaesthetic”. Drugs. 35 (4): 334–72. doi:10.2165/00003495-198835040-00002. PMID 3292208.
- ^ Bailey, J; Mora, C; Shafer, S (1996). “Pharmacokinetics of propofol in adult patients undergoing coronary revascularization”. Anesthesiology. 84 (6): 1288–97. doi:10.1097/00000542-199606000-00003. PMID 8669668. S2CID 26019589.
- ^ Reilly, C; Nimmo, W (1987). “New intravenous anaesthetics and neuromuscular blocking drugs. A review of their properties and clinical use”. Drugs. 34 (1): 115–9. doi:10.2165/00003495-198734010-00004. PMID 3308413. S2CID 46973781.
- ^ Schramm, BM; Orser, BA (2002). “Dystonic reaction to propofol attenuated by benztropine (Cogentin)”. Anesth Analg. 94 (5): 1237–40. doi:10.1097/00000539-200205000-00034. PMID 11973196.
- ^ Vesta, Kimi; Shaunta’ Martina; Ellen Kozlowski (25 April 2009). “Propofol-Induced Priapism, a Case Confirmed with Rechallenge”. The Annals of Pharmacotherapy. 40 (5): 980–982. doi:10.1345/aph.1G555. PMID 16638914. S2CID 36563320.
- ^ Fuentes, Ennio; Silvia Garcia; Manuel Garrido; Cristina Lorenzo; Jose Iglesias; Juan Sola (July 2009). “Successful treatment of propofol-induced priapism with distal glans to corporal cavernosal shunt”. Urology. 74 (1): 113–115. doi:10.1016/j.urology.2008.12.066. PMID 19371930.
- ^ Eumorfia Kondili; Christina Alexopoulou; Nectaria Xirouchaki; Dimitris Georgopoulos (2012). “Effects of propofol on sleep quality in mechanically ventilated critically ill patients: a physiological study”. Intensive Care Medicine. 38 (10): 1640–1646. doi:10.1007/s00134-012-2623-z. PMID 22752356. S2CID 21206446.
- ^ “AstraZeneca – United States Home Page” (PDF). .astrazeneca-us.com. Archived from the original (PDF) on 4 October 2011. Retrieved 8 June 2013.
- ^ Kim, MD, FACEP, Tae Eung; Shankel, MD, Tamara; Reibling, PhD, MA, Ellen T.; Paik, MSN, RN, Jacqueline; Wright, PhD, RN, Dolores; Buckman, PhD, RN, Michelle; Wild, MS, RN, Kathi; Ngo, MS, Ehren; Hayatshahi, PharmD, Alireza (1 January 2017). “Healthcare students interprofessional critical event/disaster response course”. American Journal of Disaster Medicine. 12 (1): 11–26. doi:10.5055/ajdm.2017.0254. ISSN 1932-149X. PMID 28822211.
- ^ Vasile B, Rasulo F, Candiani A, Latronico N (2003). “The pathophysiology of propofol infusion syndrome: a simple name for a complex syndrome”. Intensive Care Medicine. 29 (9): 1417–25. doi:10.1007/s00134-003-1905-x. PMID 12904852. S2CID 23932736.
- ^ Doheny, Kathleen; Louise Chang; Hector Vila Jr (24 August 2009). “Propofol Linked to Michael Jackson’s Death”. WebMD. Archived from the original on 28 August 2009. Retrieved 26 August 2009.
- ^ Trapani G, Altomare C, Liso G, Sanna E, Biggio G (February 2000). “Propofol in anesthesia. Mechanism of action, structure-activity relationships, and drug delivery”. Curr. Med. Chem. 7 (2): 249–71. doi:10.2174/0929867003375335. PMID 10637364.
- ^ Kotani, Y; Shimazawa, M; Yoshimura, S; Iwama, T; Hara, H (Summer 2008). “The experimental and clinical pharmacology of propofol, an anesthetic agent with neuroprotective properties”. CNS Neuroscience and Therapeutics. 14 (2): 95–106. doi:10.1111/j.1527-3458.2008.00043.x. PMC 6494023. PMID 18482023.
- ^ Vanlersberghe, C; Camu, F (2008). Propofol. Handbook of Experimental Pharmacology. 182. pp. 227–52. doi:10.1007/978-3-540-74806-9_11. ISBN 978-3-540-72813-9. PMID 18175094.
- ^ Trapani, G; Latrofa, A; Franco, M; Altomare, C; Sanna, E; Usala, M; Biggio, G; Liso, G (1998). “Propofol analogues. Synthesis, relationships between structure and affinity at GABAA receptor in rat brain, and differential electrophysiological profile at recombinant human GABAA receptors”. Journal of Medicinal Chemistry. 41 (11): 1846–54. doi:10.1021/jm970681h. PMID 9599235.
- ^ Krasowski MD, Jenkins A, Flood P, Kung AY, Hopfinger AJ, Harrison NL (April 2001). “General anesthetic potencies of a series of propofol analogs correlate with potency for potentiation of gamma-aminobutyric acid (GABA) current at the GABA(A) receptor but not with lipid solubility”. J. Pharmacol. Exp. Ther. 297 (1): 338–51. PMID 11259561.
- ^ Krasowski, MD; Hong, X; Hopfinger, AJ; Harrison, NL (2002). “4D-QSAR analysis of a set of propofol analogues: mapping binding sites for an anesthetic phenol on the GABA(A) receptor”. Journal of Medicinal Chemistry. 45 (15): 3210–21. doi:10.1021/jm010461a. PMC 2864546. PMID 12109905.
- ^ Haeseler G, Leuwer M (March 2003). “High-affinity block of voltage-operated rat IIA neuronal sodium channels by 2,6 di-tert-butylphenol, a propofol analogue”. Eur J Anaesthesiol. 20 (3): 220–4. doi:10.1017/s0265021503000371. PMID 12650493. S2CID 25072723.
- ^ Haeseler, G; Karst, M; Foadi, N; Gudehus, S; Roeder, A; Hecker, H; Dengler, R; Leuwer, M (September 2008). “High-affinity blockade of voltage-operated skeletal muscle and neuronal sodium channels by halogenated propofol analogues”. British Journal of Pharmacology. 155 (2): 265–75. doi:10.1038/bjp.2008.255. PMC 2538694. PMID 18574460.
- ^ Fowler CJ (February 2004). “Possible involvement of the endocannabinoid system in the actions of three clinically used drugs”. Trends Pharmacol. Sci. 25 (2): 59–61. doi:10.1016/j.tips.2003.12.001. PMID 15106622.
- ^ Lee, U; Mashour, GA; Kim, S; Noh, GJ; Choi, BM (2009). “Propofol induction reduces the capacity for neural information integration: implications for the mechanism of consciousness and general anesthesia”. Conscious. Cogn. 18 (1): 56–64. doi:10.1016/j.concog.2008.10.005. PMID 19054696. S2CID 14699319.
- ^ Favetta P, Degoute CS, Perdrix JP, Dufresne C, Boulieu R, Guitton J (2002). “Propofol metabolites in man following propofol induction and maintenance”. British Journal of Anaesthesia. 88(5): 653–8. doi:10.1093/bja/88.5.653. PMID 12067002.
- ^ Veselis RA, Reinsel RA, Feshchenko VA, Wroński M (October 1997). “The comparative amnestic effects of midazolam, propofol, thiopental, and fentanyl at equisedative concentrations”. Anesthesiology. 87 (4): 749–64. doi:10.1097/00000542-199710000-00007. PMID 9357875. S2CID 30185553.
- ^ James, R; Glen, JB (December 1980). “Synthesis, biological evaluation, and preliminary structure-activity considerations of a series of alkylphenols as intravenous anesthetic agents”. Journal of Medicinal Chemistry. 23 (12): 1350–1357. doi:10.1021/jm00186a013. ISSN 0022-2623. PMID 7452689.
- ^ Foundation, Lasker. “Discovery and development of propofol, a widely used anesthetic”. The Lasker Foundation. Retrieved 25 July 2020.
- ^ “Drugs@FDA: FDA Approved Drug Products”. U.S. Food and Drug Administration (FDA). Archived from the original on 13 August 2014. Retrieved 8 June 2013.
- ^ “Archived copy” (PDF). Archived (PDF) from the original on 17 April 2014. Retrieved 15 June 2014. pp. 3. accessed 23. January 2016
- ^ Stein M, et al. (September 2012). “Azo-Propofols: Photochromic Potentiators of GABAA Receptors”. Angewandte Chemie International Edition. 51 (42): 15000–4. doi:10.1002/anie.201205475. PMC 3606271. PMID 22968919.
- ^ Yip G, Z.-W Chen, Edge C J, Smith E H, Dickinson R, Hohenester, E, Townsend R R, Fuchs K, Sieghart W, Evers A S, Franks N P (September 2013). “A propofol binding site on mammalian GABAAreceptors identified by photolabeling”. Nature Chemical Biology. 9 (11): 715–720. doi:10.1038/nchembio.1340. PMC 3951778. PMID 24056400.
- ^ Kvam C, Granese D, Flaibani A, Pollesello P, Paoletti S (1993). “Hyaluronan can be protected from free-radical depolymerization by 2, 6-diisopropylphenol, a novel radical scavenger”. Biochem. Biophys. Res. Commun. 193 (3): 927–33. doi:10.1006/bbrc.1993.1714. PMID 8391811.
External links
| Wikimedia Commons has media related to Propofol. |
- “Propofol”. Drug Information Portal. U.S. National Library of Medicine.
- GB patent 1472793, John B Glen & Roger James, “Pharmaceutical Compositions”, published 1977-05-04, assigned to Imperial Chemical Industries Ltd
| Clinical data | |
|---|---|
| Trade names | Diprivan, others[1] |
| AHFS/Drugs.com | Monograph |
| License data | US DailyMed: Propofol |
| Pregnancy category | AU: C |
| Dependence liability | Physical: very low (seizures) Psychological: no data |
| Addiction liability | Moderate[2] |
| Routes of administration | Intravenous |
| ATC code | N01AX10 (WHO) |
| Legal status | |
| Legal status | AU: S4 (Prescription only)CA: ℞-onlyUK: POM (Prescription only)US: ℞-only [3]In general: ℞ (Prescription only) |
| Pharmacokinetic data | |
| Bioavailability | NA |
| Protein binding | 95–99% |
| Metabolism | Liver glucuronidation |
| Onset of action | 15–30 seconds[4] |
| Elimination half-life | 1.5–31 hours[4] |
| Duration of action | ~5–10 minutes[4] |
| Excretion | Liver |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 2078-54-8 |
| PubChem CID | 4943 |
| IUPHAR/BPS | 5464 |
| DrugBank | DB00818 |
| ChemSpider | 4774 |
| UNII | YI7VU623SF |
| KEGG | D00549 |
| ChEBI | CHEBI:44915 |
| ChEMBL | ChEMBL526 |
| CompTox Dashboard (EPA) | DTXSID6023523 |
| ECHA InfoCard | 100.016.551 |
| Chemical and physical data | |
| Formula | C12H18O |
| Molar mass | 178.275 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI | |
| (verify) |
/////////////PROPOFOL

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
