Home » Posts tagged 'Phase2 drugs'
Tag Archives: Phase2 drugs
Phase 2, Sarepta Therapeutics, Efficacy, Safety, and Tolerability Rollover Study of Eteplirsen in Subjects With Duchenne Muscular Dystrophy
Eteplirsen, also called AVI-4658, is an experimental drug, currently in clinical trials. It is designed for treatment of some mutations which cause Duchenne muscular dystrophy (DMD), a genetic degenerative muscle disease. Eteplirsen is a product of Sarepta Therapeutics Inc.
s excision of exon 51 during pre-mRNA splicing of the dystrophin RNA transcript. Skipping exon 51 changes the downstream reading frame of dystrophin;[1] giving eteplirsen to a healthy person would result in production of dystrophin mRNA which would not code for functional dystrophin protein but, for DMD patients with particular frameshifting mutations, giving eteplirsen can restore the reading frame of the dystrophin mRNA and result in production of functional (though internally-truncated) dystrophin.[2] Eteplirsen is given by intravenous infusion for systemic treatment of DMD.
Clinical studies
Several clinical trials have been conducted to test eteplirsen, one in the UK involving local injection to the foot,[3][4] one in the UK involving systemic injection at low doses[5][6] and one in the USA at higher systemic doses[7] that progressed to a rollover extension study.[8]
References
- “Exon Skipping Quantification by qRT-PCR in Duchenne Muscular Dystrophy Patients Treated with the Antisense Oligomer Eteplirsen”. Hum Gene Ther Methods.. 17 Oct 2012.
- “Morpholinos and Their Peptide Conjugates: Therapeutic Promise and Challenge for Duchenne Muscular Dystrophy.”. Biochim Biophys Acta. 1798 (12): 2296–303.. 17 Feb 2010.
- Gary Roper/Manager Clinical Research Governance Organisation, Imperial College London. “Safety and Efficacy Study of Antisense Oligonucleotides in Duchenne Muscular Dystrophy”. ClinicalTrials.gov. US Government, NIH. Retrieved 30 October 2012.
- Lancet Neurol. 8 (10): 918–28. 25 Aug 2009.
- Professor Francesco Muntoni, University College of London Institute of Child Health. “Dose-Ranging Study of AVI-4658 to Induce Dystrophin Expression in Selected Duchenne Muscular Dystrophy (DMD) Patients”. ClinicalTrials.gov. US Government, NIH. Retrieved 30 October 2012.
- “Exon skipping and dystrophin restoration in patients with Duchenne muscular dystrophy after systemic phosphorodiamidate morpholino oligomer treatment: an open-label, phase 2, dose-escalation study.”. Lancet. 378 (9791): 595–605. 23 Jul 2011.
- Sarepta Therapeutics. “Efficacy Study of AVI-4658 to Induce Dystrophin Expression in Selected Duchenne Muscular Dystrophy Patients”. ClinicalTrials.gov. US Government, NIH. Retrieved 30 October 2012.
- Sarepta Therapeutics. “Efficacy, Safety, and Tolerability Rollover Study of Eteplirsen in Subjects With Duchenne Muscular Dystrophy”. ClinicalTrials.gov. US Government, NIH. Retrieved 30 October 2012.
Phase 2 Drug: Ustekinumab A monoclonal antibody against the p40 subunit of IL-12/23 Other Name: Stelara
| Monoclonal antibody | |
|---|---|
| Type | Whole antibody |
| Source | Human |
| Target | IL-12 and IL-23
|

Ustekinumab, CAS number 815610-63-0, is also known by it’s brand name Stelara, which is marketed by Janssen Biotech, Inc. Developed as a treatment for adults with moderate to severe plaque psoriasis
Rockefeller University, MAR 2013
http://clinicaltrials.gov/ct2/show/NCT01806662
Atopic dermatitis (AD) is a chronic disease associated with intense itching, which affects most aspects of everyday life in the majority of patients. Acute inflammation and extensor/facial involvement is common in infants, whereas chronic inflammation increases in prevalence with age, as do localization to flexures. AD has a complex background characterized by immune activation, increased epidermal thickness in chronic diseased skin, and defective barrier function. In normal, healthy skin, the outer layer of the epidermis, the stratum corneum is made up flattened dead cells called corneocytes held together by a mixture of lipids and proteins. The stratum corneum and, in particular, the lipid layer are vital in providing a natural barrier function that locks water inside the skin and keeps allergens and irritants out. In people with AD, the barrier function is defective, which leads to dry skin. As the skin dries out, it cracks allowing allergens and irritants to penetrate.
Mild AD can be controlled with emollients and topical medications. However, moderate to severe AD is extremely difficult to control and requires systemic treatment that is often unsatisfactory due to impracticality and lack of effectiveness. Only three therapeutic options exist for moderate to severe AD, including: 1) oral steroids 2) cyclosporine A (CsA), that is not widely used in the US as it is not FDA approved for AD and 3) ultraviolet phototherapy. Oral steroids and CsA treatments have major side effects and UV radiation therapy is highly inconvenient for patients. Several biologic medications, such as TNF-alpha inhibitors, are effective, convenient, and relatively safe therapies for psoriasis, but have thus far not shown efficacy in AD. Ustekinumab is a unique biologic medication that may specifically target AD.
The investigators study will determine whether there is a reversal of the skin thickness and the immune pathways involved in the disease during treatment with Ustekinimab and what specific immune cells are involved. The investigators are also interested to understand how the clinical reversal of the disease will correlate with tissue reversal of the disease.
Detailed Description:
In psoriasis, epidermal hyperplasia is driven by underlying immune activation, whether as a direct response to IL-20 family cytokines that induces hyperplasia and inhibits keratinocyte terminal differentiation or as an indirect response to immune-mediated injury to keratinocytes. The epidermal reaction in psoriasis is largely restored to normal with selective immune suppression. Hence, one might hypothesize that similar epidermal responses should occur in the presence of “generalized” cellular immune activation, in diseases with similar inflammatory infiltrate and epidermal hyperplasia, such as AD. In fact, psoriasis and AD share features of dense T-cells and dentritic cell infiltrates, as well as over-expression of IL-22 in skin lesions. These diseases also share similar epidermal hyperplasia in their chronic phases.
Work from the investigators group showed that IL-22 is a key cytokine in the pathogenesis of both AD and psoriasis. The investigators have demonstrated that in psoriasis, ustekinumab suppresses the production of IL-12, IL-23, and IL-22. Additionally, by RT-PCR the investigators demonstrated that the mRNA expression of p40 cytokine and the IL23R is up-regulated in AD as compared to both normal skin and psoriasis. The investigators therefore hypothesize that ustekinumab will suppress IL-22 and possibly also p40 production in AD lesions and reverse both the epidermal growth/differentiation defects and the underlying immune activation, and hence will suppress disease activity. Interestingly, p40 was also found to be significantly up-regulated in non-lesional AD skin as compared with normal skin.
Although AD is thought to be predominately a disease of Th2-type cells, in the chronic stage, there is large Th1 component. To date, the precise mechanism by which sequential activation of Th2 and Th1 cells in AD is achieved remains unknown. IL-12 induces the differentiation and maturation of human Th cells into Th1-type cells. Recent circumstantial evidence suggests that in AD patients IL-12 may facilitate a change from the Th2-type to a Th1 cytokine profile. IL-12 was recently shown to be highly elevated in pediatric AD and its levels were strongly associated with disease severity.
Expression of IL-12 p40 mRNA is significantly enhanced in lesional skin from AD, suggesting that the enhanced local production of IL-12 in dendritic cells and macrophages may be responsible for the increased production of IFN-γ in chronic lesions potentially suggesting that IL-12 may have a pivotal role in promoting inflammation in atopic dermatitis. Topical steroids which constitute a mainstay of therapy in AD are known to strongly down-regulate IL-12 expression, possibly also indicating that targeted anti IL-12 therapy might important role in treating AD.
Recently, the Th1/Th2 paradigm in autoimmunity and allergy has been revisited to include a role for a new population of IL-17-producing Th cells known as Th17. Th17 cells are characterized by the production of inflammatory cytokines such as IL-17A, IL-17F, IL-22, and IL-26. One of the key factors involved in naive Th-cell commitment to a Th17 phenotype is IL-23.
Patients with acute AD were found to have increased Th17 T-cells in peripheral blood by flow cytometry and intracellular cytokine staining 26 as well as by immunohistochemistry (IHC) in lesions. Since IL-23 is the major inducer of Th17 T-cells, as well as “T22” T-cells, neutralization of IL-23 could potentially result in both decreased Th17 signal in acute AD as well as decreased “T22/IL22″ signal. Therefore the investigators postulate that ustekinumab in AD will act both inhibiting the IL-12-dependent Th1 shift in chronic AD stage as well as the pathogenic IL-22/”T22” axis in this disease.
Ustekinumab [1] (INN, experimental name CNTO 1275, proprietary commercial name Stelara,[2] Centocor) is a human monoclonal antibody. It is directed against interleukin 12 and interleukin 23, naturally occurring proteins that regulate the immune system and immune-mediated inflammatory disorders.[3]
In two Phase III trials for moderate to severe psoriasis, the longest >76 weeks, ustekinumab was safe and effective.[4][5]
A third Phase III trial, ACCEPT, compared the efficacy and safety of ustekinumab with etanercept in the treatment of moderate to severe plaque psoriasis.[6] This trial found a significantly higher clinical response with ustekinumab over the 12-week study period compared to high-dose etanercept.[6] It also demonstrated the clinical benefit of ustekinumab among patients who failed to respond to etanercept.[6]
Ustekinumab is approved in Canada, Europe and the United States to treat moderate to severe plaque psoriasis.[7]
As of November 2009, the drug is being investigated for the treatment of psoriatic arthritis.[8][9] It has also been tested in Phase II studies for multiple sclerosis[10] and sarcoidosis, the latter versus golimumab (Simponi).[11]
- Cingoz, Oya (2009). “Ustekinumab”. MAbs 1 (3): 216–221. doi:10.4161/mabs.1.3.8593. PMC 2726595. PMID 20069753.
- ^ European Medicines Agency, 20 November 2008, http://www.emea.europa.eu/pdfs/human/opinion/Stelara_58227008en.pdf
- ^ Reddy M, Davis C, Wong J, Marsters P, Pendley C, Prabhakar U (May 2007). “Modulation of CLA, IL-12R, CD40L, and IL-2Ralpha expression and inhibition of IL-12- and IL-23-induced cytokine secretion by CNTO 1275”. Cell. Immunol. 247 (1): 1–11. doi:10.1016/j.cellimm.2007.06.006. PMID 17761156.
- ^ Leonardi CL, Kimball AB, Papp KA, et al. (May 2008). “Efficacy and safety of ustekinumab, a human interleukin-12/23 monoclonal antibody, in patients with psoriasis: 76-week results from a randomised, double-blind, placebo-controlled trial (PHOENIX 1)”. Lancet 371 (9625): 1665–74. doi:10.1016/S0140-6736(08)60725-4. PMID 18486739.
- ^ Papp KA, Langley RG, Lebwohl M, et al. (May 2008). “Efficacy and safety of ustekinumab, a human interleukin-12/23 monoclonal antibody, in patients with psoriasis: 52-week results from a randomised, double-blind, placebo-controlled trial (PHOENIX 2)”. Lancet 371 (9625): 1675–84. doi:10.1016/S0140-6736(08)60726-6. PMID 18486740.
- ^ a b c Griffiths C, Strober B, van de Kerkhof P et al. (2010). “Comparison of Ustekinumab and Etanercept for Moderate-to-Severe Psoriasis”. N Engl J Med 362 (2): 118–28. doi:10.1056/NEJMoa0810652. PMID 20071701.
- ^ Medarex to Receive Milestone Payment for Approval of STELARA(TM) (Ustekinumab) for the Treatment of Moderate to Severe Plaque Psoriasis
- ^ ClinicalTrials.gov NCT00267956 A Study of the Safety and Efficacy of CNTO 1275 in Patients With Active Psoriatic Arthritis
- ^ ClinicalTrials.gov NCT01009086 A Study of the Safety and Efficacy of Ustekinumab in Patients With Psoriatic Arthritis
- ^ ClinicalTrials.gov NCT00207727 A Safety and Efficacy Study of CNTO1275 in Patients With Multiple Sclerosis
- ^ ClinicalTrials.gov NCT00955279 A Study to Evaluate the Safety and Effectiveness of Ustekinumab or Golimumab Administered Subcutaneously (SC) in Patients With Sarcoidosis
- ^ http://www.empr.com/stelara-approved-for-moderate-to-severe-psoriasis/article/149760/
- ^ a b Centocor 12/19/08 Press Release, http://www.centocor.com/centocor/i/press_releases/FDA_ISSUES_COMPLETE_RESPONSE_LETTER_TO_CENTOCOR_FOR_USTEKINUMAB_BIOLOGIC_LICENSE_APPLICATION_
- ^ Johnson LL. “Study: Drug for serious psoriasis tops competition” The Associated Press. 18 Sept 2008.[dead link]
- ^ Wild, David (November 2011), “Novel IL-12/23 Antagonist Shows Potential in Severe Crohn’s”, Gastroenterology & Endoscopy News 62 (11), retrieved 2011-12-04
- ^ a b c Weber J, Keam SJ (2009). “Ustekinumab”. BioDrugs 23 (1): 53–61. doi:10.2165/00063030-200923010-00006. PMID 19344192.
- ^ Segal BM, Constantinescu CS, Raychaudhuri A, Kim L, Fidelus-Gort R, Kasper LH (September 2008). “Repeated subcutaneous injections of IL12/23 p40 neutralising antibody, ustekinumab, in patients with relapsing-remitting multiple sclerosis: a phase II, double-blind, placebo-controlled, randomised, dose-ranging study”. Lancet Neurol 7 (9): 796–804. doi:10.1016/S1474-4422(08)70173-X. PMID 18703004.
- ^ “Important Safety Information”. STELARA® (ustekinumab). Janssen Biotech.
External links
- Centocor Ortho Biotech official site
- CNTO 1275 research studies registered with U.S. National Institutes of Health:
- ClinicalTrials.gov NCT00207727 Phase II Study on Multiple Sclerosis
- ClinicalTrials.gov NCT00320216 Phase II Study on Psoriasis
- ClinicalTrials.gov NCT00267969 Phase III Study on Psoriasis
- ClinicalTrials.gov NCT00307437 Phase III Study on Psoriasis
- ClinicalTrials.gov NCT00267956 Phase II Study on Psoriatic Arthritis
- Sylvester, Bruce (2006-03-06). “CNTO 1275 Shows Efficacy for Psoriasis: Presented at AAD”. Doctor’s Guide Publishing. Retrieved 2007-01-25.
Phase 2 SMP-986(afacifenacin fumarate) Overactive bladder/ Dainippon Sumitomo Pharma and Nippon Shinyaku conclude a license agreement for SMP-986, a therapeutic agent for urology
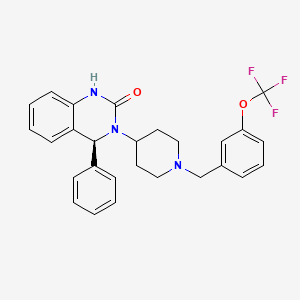
877606-63-8 cas no of afacifenacin
any str error, mail to amcrasto@gmail.com
SMP-986 (afacifenacin fumarate)
(4S)-4-phenyl-3-(1-{[3-(trifluoromethoxy)phenyl]methyl}piperidin- 4-yl)-3,4-dihydroquinazolin-2(1H)-one muscarinic receptor antagonist

- Developed in-house
- SMP-986 possesses the dual pharmacological actions of muscarinic receptor antagonism (non-selective) and inhibition of the bladder afferent pathway through Na+-channel blockade. This compound is being evaluated for its ability to ease urinary urgency and reduce the frequency of both urination and incontinence. The compound has also exhibited the potential to have lower incidence of side effects related to muscarinic receptor antagonism, such as dry mouth.
- Development stage: Phase II in the U.S. and Europe. Phase II in Japan
7 mar 2013
Dainippon Sumitomo Pharma Co., Ltd. (DSP) and Nippon Shinyaku Co., Ltd. Announce they have concluded a license agreement for exclusive rights to develop, manufacture and commercialize SMP-986 in Japan, a new therapeutic agent for overactive bladder created by DSP.
DCVax®-Direct Phase I/II Trial For All Inoperable Solid Tumors Is Expected To Produce Ongoing Results In 2013

BETHESDA, Md., \
March 5, 2013 Northwest Biotherapeutics (NW Bio), a biotechnology company developing DCVax® personalized immune therapies for solid tumor cancers, announced today that its Phase I/II DCVax-Direct clinical trial for all inoperable solid tumor cancers is planned to begin within approximately the next sixty days. As a Phase I/II trial, this trial is not blinded and the results will be seen as the trial proceeds. With an efficacy endpoint of tumor regression (i.e., tumor shrinkage or elimination), this innovative trial is expected to yield meaningful ongoing results by the second half of 2013.
This clinical trial is approved by the FDA for all types of solid tumor cancers (i.e., cancers in any tissues of the body), and is configured to provide rapid results. In the Phase I portion, it will test both safety and a variety of dosing regimes, and will do so in multiple different cancers – avoiding the need to conduct separate Phase I studies in each such cancer, as is usually the case. Then, the trial will go directly into the Phase II portion, testing for efficacy, without the need for another FDA review.
DCVax®
All immune responses start in the same way that involves initially a single cell type, the dendritic cell (DC). This cell functions like the General of an army, in that it directs all ensuing activities of the immune response. The immune system is like an army with many divisions, and multiple soldiers with various types of weapons. When an invader, which could be a virus, bacteria, or a cancer cell, encounters a DC the result is that the DC consumes or eats the invader and chops it into small pieces. In the process, the DC becomes activated and starts traveling to the lymph node. In the lymph nodes the DC elicits a cascade of events eventually involving the entire army that leads to a full-blown immune response. Importantly, the nature of the invader and the nature of the DC activation dictate the type of immune response: the DC is the general of the army who directs all the soldiers to work in synchrony, and who determines which weapons to use to best defeat the enemy. We believe that preparing the DC outside the body, as done for DCVax® products, allows the greatest degree of control and begins the immune response in the natural fashion leading to the most complete attack on the foreign invader.
Different Approaches
We believe that the optimal way to arrive at the most effective immune response is through the control of the DC. Most traditional immunization approaches, including traditional virus, specific antigen or peptide vaccines as well as some that are used for immunotherapy of cancer, try to control the already existing DC in the body, or they try to modulate only one arm of the immune system. The immunogen, i.e. the virus, specific antigen, peptide or the cancer cells used to prepare the vaccine, is in those cases injected into the body in a formulation that aims at targeting and activating local DC. Examples are viral, specific antigen or peptide vaccines formulated with adjuvant, or killed tumor cells alone or modified to produce the DC mobilizing protein GM-CSF. In these instances, it is left to chance as to whether the immunogen arrives at the DC, whether the DC are properly activated, and effectively migrate to lymph nodes to produce an effective immune response. We believe that the failure of several recent clinical trials may be the result of the inability of these other approaches to effectively mount a natural and robust immune response.
Treatments that use only a single division of the immune system may employ only large amounts of T cells, or a single (monoclonal) antibody. DCVax® products are being developed to activate all aspects of the immune response, both cellular and antibody, thus potentially providing a broader and longer lasting immune and clinical response. Northwest Biotherapeutics’ products are deigned as pure, activated DC loaded with the immunogen as would naturally occur, and that are capable of migrating to lymph nodes. The intended result is a very robust, and full immune response consisting of both a specific cellular T cell response and a specific antibody response against the cancer associated antigen. Data obtained in our Phase I and Phase II clinical trials suggest that such response may occur and may translate into a clinical benefit.
Cancer and the Immune System
Cancer cells produce many substances that shut down the immune response, as well as substances that paralyze the DC that are resident in the body. We believe therefore that the optimal time for controlling cancer growth by activating the immune system is at the time when tumor burden is low. Northwest Biotherapeutics targets patients with brain cancer following surgery, radiation and chemotherapy, and hormone independent prostate cancer patients with no detectable tumor growth. This approach aims at inducing powerful immune responses to control progression of the disease.
Manufacturing
Northwest Biotherapeutics has focused on solving many of the challenges that are typically associated with producing personalized products that consist of living cells. The Company’s new automated cell processing system allows high-throughput production of products for a fraction of the historical cost.
- DCVax® products contain pure DC
- The DC in DCVax® are prepared outside the body, which eliminates many uncontrollable variables, and are subjected to a potency test designed to ensure that the DC administered to the patient are capable of eliciting an immune response
- DCVax® is used in patients with low tumor burden
- DCVax® products can be manufactured in a cost-efficient manner
Immune begins Phase II study of ulcerative colitis drug, Bertilimumab
| Monoclonal antibody | |
|---|---|
| Type | Whole antibody |
| Source | Human |
| Target | CCL11 (eotaxin-1) |
phase 2 NCT01671956; C2a/BRT/UC-01
A Randomized, Double-Blind, Placebo-Controlled Study Designed to Evaluate the Safety, Clinical Efficacy, and Pharmacokinetic Profile of Bertilimumab in Subjects With Active Moderate to Severe Ulcerative Colitis
25 February 2013
Immune Pharmaceuticals has started Phase II study of its bertilimumab (iCo-008 or CAT-213) drug, designed for the treatment of moderate-to-severe ulcerative colitis.
Bertilimumab is a human immunoglobulin monoclonal antibody which targets eotaxin-1, a member of the chemokine family of proteins regulating eosinophilic inflammation.
The double-blind, parallel group, randomized, placebo-controlled 90 patients-based study is designed to demonstrate the safety, clinical efficacy, and pharmacokinetic profile of bertilimumab in subjects with active moderate-to-severe ulcerative colitis.
60 patients in the study will be treated with bertilimumab 7mg/kg, while 30 patients with placebo every two weeks at days 0, 14, and 28, according to the company.
In addition, the patients will be evaluated for clinical response after six weeks to determine the decrease if any in the full Mayo Clinic Ulcerative Colitis Score.
Secondary and exploratory end points of the study include clinical remission defined as symptom free, fecal calprotectin, a recognized marker of gastro-intestinal inflammation, histopathology improvement and degree of mucosal injury.
The company, which is expecting to follow-up the patients for up to day 90, said the patient enrollment and clinical results are likely to be completed in 2014.
The company has also announced that bertilimumab will be the lead clinical stage development drug for the combined company following completion of the proposed merger with EpiCept in the second quarter of 2013.
Bertilimumab is a human monoclonal antibody that binds to eotaxin-1, an important regulator of overall eosinophil function.
patent WO00166754
It was discovered by Cambridge Antibody Technology using their phage displaytechnology.[1] Named CAT-213 during early discovery and development by CAT, it was to be used to treat severe allergic disorders.[2]
In January 2007, CAT licensed the drug for treatment of allergy disorders to iCo Therapeutics Inc.[3] iCo Therapeutics Inc. is a Vancouver-based reprofiling company focused on redosing or reformulating drugs with clinical history for new or expanded indications – a so-called ‘search and development company’.[4]
iCo Therapeutics Inc. renamed the drug from CAT-213 to iCo-008 and, at that stage, planned to initiate a Phase II clinical trial in patients with vernal keratoconjunctivitis.[5]
In March 2008, iCo announced iCo-008 had been in 126 patients in Phase I and II clinical trials. The drug substance had been manufactured by Lonza, in its cGMP facilities inSlough, UK. Subsequently iCo moved the drug substance to a fill-finish site for the final stage of manufacturing. iCo reported that the iCo-008 drug product was within specifications and contained a high antibody yield.[6]
In June 2011, IMMUNE Pharmaceuticals[7] (Herzliya, Israel) in-licensed Bertilimumab from iCo for non-ophthalmic indications. [8]IMMUNE is initiating Phase II clinical trials of Bertilimumab in inflammatory bowel disease (ulcerative colitis & Crohn’s disease) in 2012 and 2013.
- http://jpet.aspetjournals.org/cgi/content/abstract/319/3/1395
- Bertilimumab Cambridge Antibody Technology Group. 5. November 2004. pp. 1213–8. PMID 15573873.
- http://www.icotherapeutics.com/site/investor-relations/cambridge_antibody_tech_licenses_monoclonal_antibody_treatment_allergy/
- http://www.icotherapeutics.com/site/corporate_overview/overview/
- http://www.icotherapeutics.com/site/pipeline/ico008/
- http://www.icotherapeutics.com/site/investor-relations/ico_therapeutics_provides_ico_008_phase_ii_clinical_update/
- http://immunepharmaceuticals.com/
- http://immunepharmaceuticals.com/index.php?option=com_content&view=article&id=32&Itemid=20
…………………………………….
DR ANTHONY MELVIN CRASTO Ph.D , Born in Mumbai in 1964 and graduated from Mumbai University, Completed his PhD from ICT ,1991, Mumbai, India in Organic chemistry, The thesis topic was Synthesis of Novel Pyrethroid Analogues,
Currently he is working with GLENMARK- GENERICS LTD, Research centre as Principal Scientist, Process Research (bulk actives) at Mahape, Navi Mumbai, India.
Prior to joining Glenmark, he worked with major multinationals like Hoechst Marion Roussel, now Sanofi Aventis, & Searle India ltd, now Rpg lifesciences, etc. He has worked in Basic research, Neutraceuticals, Natural products, Flavors, Fragrances, Pheromones, Vet Drugs, Drugs, formulation, GMP etc. He has total 25 yrs exp in this field, he is now helping millions, has million hits on google on all organic chemistry websites.
His New Drug Approvals , Green Chemistry International, Eurekamoments in Organic Chemistry , Organic Chemistry by Dr Anthony, WIX BLOG , are some most read chemistry blogs
He has hands on experience in initiation and developing novel routes for drug molecules and implementation them on commercial scale over a 25 year tenure, good knowledge of IPM, GMP, Regulatory aspects, he has several international drug patents published worldwide .
He has good proficiency in Technology Transfer, Spectroscopy , Stereochemistry , Synthesis, Reactions in Org Chem , Polymorphism, Pharmaceuticals , Medicinal chemistry , Organic chemistry literature , Patent related site , Green chemistry , Reagents , R & D , Molecules , Heterocyclic chem , Sourcing etc
He suffered a paralytic stroke in dec 2007 and is bound to a wheelchair, this seems to have injected feul in him to help chemists around the world, he is more active than before and is pushing boundaries, he has one lakh connections on all networking sites, He makes himself available to all, contact him on +91 9323115463, amcrasto@gmail.com

Edison commences EPI-743 Vatiquinone Phase 2 study in cobalamin C deficient patients

19 February 2013
EPI-743 Vatiquinone is a new drug that is based on vitamin E. Tests have shown that it can help improve the function of cells with mitochondrial problems. It may be able to treat people with genetic disorders that affect metabolism and mitochondria
Edison Pharmaceuticals and Bambino Gesu Children’s Hospital have announced the commencement of EPI-743 Phase 2 cobalamin C deficiency syndrome trial.
EPI-743 is an orally bioavailable small molecule and a member of the para-benzoquinone class of drugs.
The trial’s principal investigator, Bambino Gesu Children’s Hospital, division of metabolism Professor Carlo Dionisi-Vici said, “Given the central role of glutathione in cellular redox balance and antioxidant defense systems, we are eager to explore whether a therapeutic that increases glutathione such as EPI-743 will provide clinical benefit.”
Improvement in visual function is the primary endpoint of the placebo-controlled study while secondary outcome measurements assess neurologic and neuromuscular function, glutathione biomarkers, quality of life, in addition to safety parameters.
The investigation is aimed at assessing the efficacy of EPI-743 in disorders of intermediary metabolism that also result in redox disturbances.
EPI-743 is an orally absorbed small molecule that readily crosses into the central nervous system. It works by targeting the enzyme NADPH quinone oxidoreductase 1 (NQO1). Its mode of action is to synchronize energy generation in mitochondria with the need to counter cellular redox stress
Neurim Pharmaceuticals Announces Positive Phase 2 Clinical Trial Results of Piromelatine for the Treatment of Insomnia
Chemical structures of Neu-P11 (a) and Neu-P5 (b).
structure from- http://www.sciencedirect.com/science/article/pii/S0731708512005304#gr1

ZURICH, Feb. 18, 2013 Neurim Pharmaceuticals announced today positive results from a phase II clinical study evaluating the efficacy and safety of Piromelatine (Neu-P11), a novel investigational multimodal sleep medicine developed for the treatment of patients with primary and co-morbid insomnia. The new results are from a recent double-blind, randomized, placebo controlled, parallel group, non-confirmatory, sleep-laboratory study. The study evaluated piromelatine compared to placebo in 120 adult primary insomnia patients ages 18 years and older.
Piromelatine 20/50mg treatment for 4 weeks resulted in statistically significant and clinically meaningful improvements relative to placebo in key polysomnographic (PSG) parameters including Wake After Sleep Onset (WASO) (p=0.02 for both doses) and in particular WASO for the first 6 hours of sleep (WASO-6h) (p=0.0008 and p=0.04 for the 50 mg and 20 mg groups, respectively). Piromelatine 50 mg also improved Sleep Efficiency (SEF) (p=0.02), Total Sleep Time (TST) (p=0.02), Total Time Awake (TTA) (p=0.01) and time in NREM sleep (p=0.028) indicating beneficial effects on sleep maintenance. Subjective improvements relative to placebo in quality of sleep and total sleep time measured by the Pittsburg Sleep Quality Questionnaire (PSQI) were also observed, confirming the PSG findings. Piromelatine enhanced NREM sleep EEG delta power and significantly reduced beta power (p<0.05). The decrease in EEG beta activity, a marker of cortical arousal, is a physiological surrogate marker of the efficacy of Piromelatine in sleep maintenance. Piromelatine was generally safe and well tolerated, had no detrimental effects on next-day psychomotor performance (as assessed by the Digit Symbol Substitution Test (DSST)) for any dose group and no deleterious effects on sleep structure and architecture.
“Piromelatine demonstrates a good potential for the treatment of primary insomnia characterized by sleep maintenance disturbances as well as insomnia with psychiatric or medical co-morbidities,” said Prof. Nava Zisapel, CSO of Neurim. The study results will be presented at the 27th Annual Meeting of the Associated Professional Sleep Societies (APSS) -Sleep 2013 meeting in Baltimore.
PIROMELATINE (NEU-P11)
Piromelatine is a novel compound under development for the treatment of insomnia associated with pain.
Piromelatine is a melatonin agonist, serotonin 5-HT-1A and 5-HT-1D agonist. The compound binds to the MT1, 2 and 3 receptors which govern the body’s sleep-wake cycle and circadian rhythm. The sleep promoting, analgesic, anti-diabetic, antihypertensive, anti-neurodegenerative, anxiolytic and antidepressant effects of Piromelatine have been demonstrated in a series of relevant animal models.
Piromelatine is a multi-facet drug addressing a wide range of potential indications including, but not limited to, insomnia, IBS, neuropathy and fibromyalgia.
Neurim has recently completed a phase-II clinical trial to assess the efficacy and safety of Piromelatine in patients with primary insomnia.
Peregrine Pharmaceuticals Announces Results From Phase II Clinical Trial of Bavituximab in Stage IV Pancreatic Cancer
TUSTIN, CA 02/13/13 — Peregrine Pharmaceuticals announced results from its 70 patient open-label, randomized Phase II clinical trial of bavituximab used in combination with gemcitabine in patients with previously untreated, advanced Stage IV pancreatic cancer. The trial included the enrollment of patients with advanced metastatic disease including significant liver involvement and poor performance status associated with rapid disease progression. Results showed that the combination of bavituximab and gemcitabine resulted in more than a doubling of overall response rates (ORR) and an improvement in overall survival (OS) when compared with gemcitabine alone (control arm). In the trial, patients treated with a combination of bavituximab and gemcitabine had a 28% tumor response rate as compared to 13% in the control arm. Median OS, the primary endpoint of the trial, was 5.6 months for the bavituximab plus gemcitabine arm and 5.2 months for the control arm (hazard ratio = 0.75).
Bavituximab binds to phosphatidylserine which is exposed on the surface of certain atypical animal cells, including tumour cells and cells infected with any of six different families of virus. These viral families contain the viruses hepatitis C, influenza A and B, HIV 1 and 2, measles, respiratory syncytial virus and pichinde virus, which is a model for the deadly Lassa virus.[2] Other cells are not affected since phosphatidylserine normally is only intracellular.[3]

Bavituximab binds to various aminophospholipids and is dependent on interaction with plasma protein beta-2 glycoprotein I to mediate binding.
These target aminophospholipids, usually residing only on the inner leaflet of the plasma membrane of cells, become exposed in virally infected, damaged or malignant cells, and more generally in most cells undergoing the process of apoptosis.
The antibody’s binding to phospholipids alerts the body’s immune system to attack the tumor endothelial cells, thrombosing the tumor’s vascular network and/or attacking free floating virally infected and metastatic cells while potentially minimizing side effects in healthy tissues.
- Statement on a nonproprietary name adopted by the USAN council
- Nature Medicine 14, 1357 – 1362 (2008)
- He, J.; Yin, Y.; Luster, T. A.; Watkins, L.; Thorpe, P. E. (2009). “Antiphosphatidylserine Antibody Combined with Irradiation Damages Tumor Blood Vessels and Induces Tumor Immunity in a Rat Model of Glioblastoma”. Clinical Cancer Research 15 (22): 6871–6880. doi:10.1158/1078-0432.CCR-09-1499. PMID 19887482. edit
- New Progression-Free Survival Data From Peregrine’s Bavituximab in Phase II Refractory Breast Cancer
- Phase II Advanced Breast Cancer Data to Be Presented at ASCO Highlight Promising Tumor Response and Progression-Free Survival Data With Peregrine’s Bavituximab
- Pharma company completes humanization of 3G4 antibody
- He, J.; Luster, T. A.; Thorpe, P. E. (2007). “Radiation-Enhanced Vascular Targeting of Human Lung Cancers in Mice with a Monoclonal Antibody That Binds Anionic Phospholipids”. Clinical Cancer Research 13 (17): 5211–5218. doi:10.1158/1078-0432.CCR-07-0793. PMID 17785577. edit
- Ran; Downes, A.; Thorpe, P. E. (2002). “Increased exposure of anionic phospholipids on the surface of tumor blood vessels”. Cancer Research 62 (21): 6132–6140. PMID 12414638.
<a href=”http://www.bloglovin.com/blog/4758019/?claim=ukqfxgh6tk3″>Follow my blog with Bloglovin</a>
BioAlliance Pharma announces the forthcoming extension of its phase II clinical trial with Validive® in the United States
CLONIDINE
FEB14 2013
BioAlliance Pharma SA (Euronext Paris – BIO), an innovative Company dedicated to the development of orphan oncology products and to supportive care products, announces the extension of its phase II clinical trial with Validive® (clonidine Lauriad™) in the United States in radio/chemotherapy-induced oral mucositis prevention in patients with head and neck cancer.
Further to approval by the United States FDA (Food and Drug Administration), BioAlliance Pharma will extend its clinical trial to the United States, increasing the number of clinical investigation centers involved in this randomized double blind phase II trial.
So far almost 50% of planned patients have been enrolled in about 30 European centers. With the upcoming initiation of several centers in the United States, BioAlliance Pharma expects to finalize trial recruitment in early 2014 with results expected the same year.
“Beyond accelerating recruitment, the extension of the trial to the United States is also a key factor to reinforce our international panel of scientific experts and clinical investigators around Validive®. This will raise awareness and create hands-on experience of the drug of future key prescribers of Validive® in major US centers specialized in oncology and radiotherapy,” stated Judith Greciet, CEO of BioAlliance Pharma.
Severe oral mucositis is a particularly invalidating pathology occurring in more than 60% of patients treated with radio/chemotherapy for head and neck cancer and has currently no validated curative or preventive treatment. It may induce intense oral pain and eating disability requiring enteral or parenteral nutritional support. Thirty per cent of patients need to be hospitalized as a result and symptoms can force patients to stop treatment for an undefined period thus reducing treatment efficacy.
Clonidine is a sympatholytic medication used to treat medical conditions, such as high blood pressure, ADHD, anxiety/panic disorder, and certain pain conditions. It is classified as a centrally acting α2 adrenergic agonist. An alternative hypothesis that has been proposed is that clonidine acts centrally as an imidazoline receptor agonist.
Synthesis
Clonidine, 2-(2,6-dichlorophenylamino)imidazoline, is synthesized from 2,6-dichloroaniline, the reaction of which with ammonium thiocyanate gives N-(2,6- dichlorophenyl)thiourea. Methylation of this product, followed by the subsequent reaction with ethylene diamine gives clonidine. 
- K. Zeile, H. Staehle, K. H. Hauotman, DE 1303141 (1961).
- H. Stahle, K. Zeile, U.S. Patent 3,202,660 (1965).
- K. Zeile, K. H. Hauotman, H. Stahle, U.S. Patent 3,236,857 (1966).
- Boehringer Sohn Ingelheim, BE 653933 (1964).
- Boehringer Sohn Ingelheim, GB 1016514 (1962).
- Boehringer Ingelheim GmbH, GB 1034938 (1964).
US patent 3937717, Stahle, H.; Koppe, H.; Kummer, W.; Stockhaus, K., “2-Phenylamino-imidazolines-(2)”, issued 1976-02-10, assigned to Boehringer Ingelheim GmbH
CLORPRES (clonidine hydrochloride and chlorthalidone) ® is a combination of clonidine hydrochloride (a centrally acting antihypertensive agent) and chlorthalidone (a diuretic).
CLORPRES (clonidine hydrochloride and chlorthalidone) ® is available as tablets for oral administration in three dosage strengths: 0.1 mg/15 mg, 0.2 mg/15 mg and 0.3 mg/15 mg of clonidine hydrochloride/chlorthalidone, respectively.
The inactive ingredients are ammonium chloride, colloidal silicon dioxide, croscarmellose sodium (Type A), magnesium stearate, microcrystalline cellulose, sodium lauryl sulfate, D&C yellow #10.
Clonidine Hydrochloride
Clonidine hydrochloride is an imidazoline derivative and exists as a mesomeric compound. The chemical name is 2-[(2,6-dichlorophenyl)imino]imidazoline monohydrochloride. The following are the structural formula, molecular formula and molecular weight:
 |
Clonidine hydrochloride is an odorless, bitter, white crystalline substance soluble in water and alcohol.
About BioAlliance Pharma
Dedicated to cancer and supportive care treatment with a focus on resistance targeting and orphan products, BioAlliance conceives and develops innovative products, for specialty markets especially in the hospital setting and for orphan or rare diseases.
Created in 1997 and introduced to the Euronext Paris market in 2005, BioAlliance Pharma’s ambition is to become a leading player in these fields by coupling innovation to patient needs. The company’s teams have the key competencies required to identify, develop and register drugs in Europe and the USA.
Cerulean doses first patient in Phase 2 study of CRLX101 drug


Chemical structure of CRLX-101
(source: Svenson S, Wolfgang M, Hwang J, Ryan J, Eliasof S. J Control Release. 2011 Jul 15;153(1):49-55. Epub 2011 Mar 23.).
CRLX101 is a novel approach to cancer chemotherapy that is currently under investigation in human trials, and is an example of ananomedicine.
The agent represents a nanoparticle conjugate that consists of a drug delivery molecule, namely a cyclodextrin-based polymer (CDP) and an anti-cancer compound (camptothecin). It was developed by Dr. Mark E. Davis, professor of Chemical Engineering at theCalifornia Institute of Technology, and associates at Insert Therapeutics, Inc., now Calando Pharmaceuticals, Inc., hence the original name “IT-101”. Its novel delivery mode allows the agent, and thus the toxic anti-cancer component, to be preferentially accumulated in cancer tissue. In turn, toxic side effect are expected to be reduced. The technology was licensed by Calando and Caltech to Cerulean Pharma Inc., in June, 2009.
CRLX101 is a camptothececin-nanoparticle conjugate, which is a novel approach to cancer chemotherapy that is currently under investigation in human trials. CRLX101 represents a nanoparticle conjugate that consists of a drug delivery molecule, namely a cyclodextrin-based polymer (CDP) and an anti-cancer compound (camptothecin). It was developed by Dr. Mark E. Davis, professor of Chemical Engineering at the California Institute of Technology, and associates at Insert Therapeutics, Inc., now Calando Pharmaceuticals, Inc., hence the original name “IT-101”. Its novel delivery mode allows the agent, and thus the toxic anti-cancer component, to be preferentially accumulated in cancer tissue. In turn, toxic side effect are expected to be reduced. The technology was licensed by Calando and Caltech to Cerulean Pharma Inc., in June, 2009. (source: http://en.wikipedia.org/wiki/CRLX101).
Camptothecin (CPT) is a potent broad-spectrum anticancer agent that acts through inhibition of topoisomerase 1. Clinical development of CPT was unsuccessful due to poor drug solubility, insufficient in vivo stability of the active form, and toxicity. In order to address these issues, a polymeric nanoparticle comprised of cyclodextrin-poly(ethylene glycol) copolymer (CDP) conjugated to CPT (CRLX101) has been developed and Phase 2 clinical studies are ongoing. Camptothecin is conjugated to the polymer in its active form at 10-12 wt.% loading. CRLX101 self-assembles in solution into nanoparticles with an apparent solubility increase of >1000-fold as compared to the parent drug camptothecin.
Current developer: Calando Pharmaceuticals, Inc/Cerulean Pharma Inc.
Cerulean Pharma has dosed the first patient in a Phase 2 study of its investigational CRLX101 drug, designed for the treatment of extensive-stage small cell lung cancer (SCLC) patients sensitive to first-line platinum-based chemotherapy.
CRLX101, a tumor-targeted nanopharmaceutical, is a dual inhibitor of topoisomerase 1 and hypoxia-inducible factor-1a and releases camptothecin over an extended period of time.
The randomized study, which is being conducted at the University of Chicago School of Medicine and affiliated institutions, has enrolled 150-patient to compare the efficacy of CRLX101 with topotecan, a second-line therapy for relapsed SCLC.
The trial has co-primary endpoints of progression-free survival (PFS) and three-month PFS rate, claims the company.
During the company’s preclinical and Phase 1/2a clinical trial, CRLX101 has demonstrated significant anti-tumor activity.
Cerulean Pharma chief medical officer Edward Garmey said the company’s clinical experience with CRLX101 shows a benign safety profile.
“The standard of care in SCLC is not well tolerated, so if we can demonstrate an efficacy benefit versus standard of care, CRLX101 would have the added benefit of improved quality of life for these very sick patients,” Garmey added.

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....















