Home » Antivirals
Category Archives: Antivirals
Suricapavir
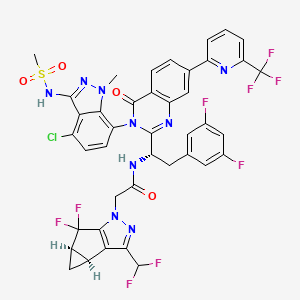
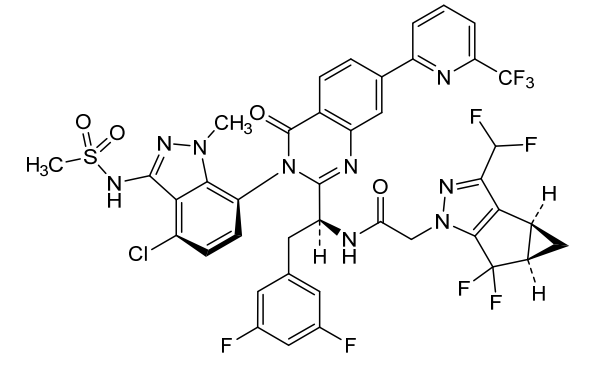
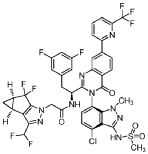
Suricapavir
CAS 2417270-21-2
MF C41H29ClF9N9O4S MF950.2 g/mol
N-[(1S)-1-[3-[4-chloro-3-(methanesulfonamido)-1-methylindazol-7-yl]-4-oxo-7-[6-(trifluoromethyl)-2-pyridinyl]quinazolin-2-yl]-2-(3,5-difluorophenyl)ethyl]-2-[(2S,4R)-9-(difluoromethyl)-5,5-difluoro-7,8-diazatricyclo[4.3.0.02,4]nona-1(6),8-dien-7-yl]acetamide
N-[(1S)-1-[3-[4-chloro-3-(methanesulfonamido)-1-methyl-indazol-7-yl]-4-oxo-7-[6-(trifluoromethyl)-2-pyridyl]quinazolin-2-yl]-2-(3,5-difluorophenyl)ethyl]-2-[(2S,4R)-9-(difluoromethyl)-5,5-difluoro-7,8-diazatricyclo[4.3.0.02,4]nona-1(6),8-dien-7-yl]acetamide
N-[(1S)-1-{(3P)-3-[4-chloro-3-(methanesulfonamido)-1-methyl-1Hindazol-7-yl]-4-oxo-7-[6-(trifluoromethyl)pyridin-2-yl]-3,4-
dihydroquinazolin-2-yl}-2-(3,5-difluorophenyl)ethyl]-2-[(3bS,4aR)-3-
(difluoromethyl)-5,5-difluoro-3b,4,4a,5-tetrahydro-1Hcyclopropa[3,4]cyclopenta[1,2-c]pyrazol-1-yl]acetamide
inhibitor of viral replication, antiviral, ZZ799EX5KN
tructurally resembles:
- Lenacapavir-type macroheterocyclic capsid inhibitors (Gilead class)
PAT
Preparation of Example 59: N-((S)-1-((3P)-3-(4-chloro-1-methyl-3-(methylsulfonamido)-1H-indazol-7-yl)-4-oxo-7-(6-(trifluoromethyl)pyridin-2-yl)-3,4-dihydroquinazolin-2-yl)-2- (3,5-difluorophenyl)ethyl)-2-((3bS,4aR)-3-(difluoromethyl)-5,5-difluoro-3b,4,4a,5- tetrahydro-1H-cyclopropa[3,4]cyclopenta[1,2-c]pyrazol-1-yl)acetamide.
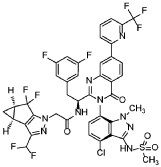
The title compound was prepared according to General Procedure D using 2-chloro-6-(trifluoromethyl)pyridine as the coupling partner. The experiment afforded the title compound, N-((S)-1-((3P)-3-(4-chloro-1-methyl-3-(methylsulfonamido)-1H-indazol-7-yl)-4-oxo-7-(6-(trifluoromethyl)pyridin-2-yl)-3,4-dihydroquinazolin-2-yl)-2-(3,5-difluorophenyl)ethyl)-2-((3bS,4aR)-3-(difluoromethyl)-5,5-difluoro-3b,4,4a,5-tetrahydro-1H-cyclopropa[3,4]cyclopenta[1,2-c]pyrazol-1-yl)acetamide. The sample was analyzed using LCMS Method F: retention time = 1.51 min.; observed ion = 948.4 (M-H).1H NMR (METHANOL-d4, 500 MHz) Shift 8.66 (s, 1H), 8.4-8.4 (m, 3H), 8.22 (t, 1H, J=7.9 Hz), 7.88 (d, 1H, J=7.7 Hz), 7.28 (br d, 1H, J=8.0 Hz), 7.20 (d, 1H, J=7.7 Hz), 6.7-6.8 (m, 1H), 6.61 (dd, 2H, J=2.2, 8.2 Hz), 6.67 (br t, 2H, J=54.7 Hz), 4.5-4.6 (m, 2H), 3.61 (s, 3H), 3.4-3.5 (m, 1H), 3.2-3.2 (m, 3H), 3.1-3.2 (m, 1H), 2.41 (br dd, 2H, J=3.7, 7.3 Hz), 1.34 (br d, 1H, J=5.4 Hz), 0.99 (br dd, 1H, J=1.9, 3.7 Hz)
PAT
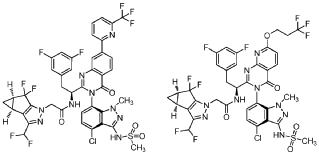
WO 2020/084492 and WO 2020/254985 disclose certain Capsid Inhibitor compounds including the two compounds shown below which will be referred to in this application as the compounds of Formula la and Formula lb.
PAT
- Inhibitors of human immunodeficiency virus replicationPublication Number: WO-2023062559-A1Priority Date: 2021-10-13
- Inhibitors of human immunodeficiency virus replicationPublication Number: US-2023149408-A1Priority Date: 2020-04-15
- Inhibitors of human immunodeficiency virus replicationPublication Number: WO-2021209900-A1Priority Date: 2020-04-15
- Pharmaceutical compositions comprising cabotegravirPublication Number: US-2023045509-A1Priority Date: 2019-12-09
- Inhibitors of human immunodeficiency virus replicationPublication Number: US-11541055-B2Priority Date: 2018-10-24Grant Date: 2023-01-03
- Inhibitors of human immunodeficiency virus replicationPublication Number: US-2020360384-A1Priority Date: 2018-10-24
- Inhibitors of human immunodeficiency virus replicationPublication Number: EP-3870577-B1Priority Date: 2018-10-24Grant Date: 2025-03-19
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

///////////////suricapavir, ANAX, inhibitor of viral replication, antiviral, ZZ799EX5KN
Pixavir marboxil
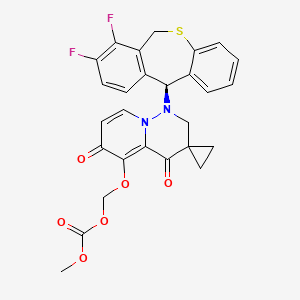
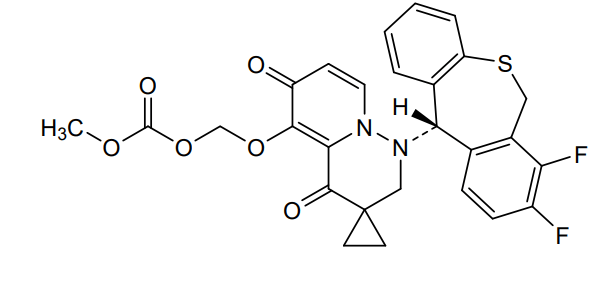
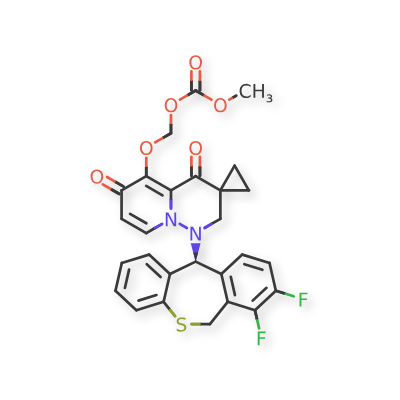
Pixavir marboxil
CAS 2365473-17-0
MF C27H22F2N2O6S MW540.535
(1-((11S)-7,8-DIFLUORO-6,11-DIHYDROBENZO(C)(1)BENZOTHIEPIN-11-YL)-4,6-DIOXOSPIRO(2H-PYRIDO(1,2-B)PYRIDAZINE-3,1′-CYCLOPROPANE)-5-YL)OXYMETHYL METHYL CARBONATE
({1′-[(11S)-7,8-difluoro-6,11-dihydrodibenzo[b,e]thiepin-11-yl]-4′,6′-dioxo1′,2′,4′,6′-tetrahydrospiro[cyclopropane-1,3′-pyrido[1,2-b]pyridazin]-5′-yl}oxy)methyl methyl carbonate
antiviral, TG 1000, Yi Li Kang, SV42843XSX, Cap-dependent endonuclease-IN-1, Influenza virus infections, TaiGen Biotechnology
- OriginatorTaiGen Biotechnology
- Class3-ring heterocyclic compounds; Antivirals; Benzene derivatives; Carbonates; Cyclopropanes; Dibenzothiepins; Esters; Ethers; Fluorobenzenes; Organic sulfur compounds; Pyridazines; Pyridones; Small molecules; Spiro compounds
- Mechanism of ActionEndonuclease inhibitors; Virus replication inhibitors
- MarketedInfluenza virus infections
- 27 Feb 2026Launched for Influenza virus infections (In adults, In adolescents) in China (PO), prior to February 2026 (TaiGen Biotechnology pipeline, February 2026)
- 26 Jan 2026Pixavir marboxil licensed to Boryung Biopharma for commercialization in South Korea
- 16 Dec 2025Chemical structure information added.
Pixavir marboxil (also known as TG-1000) is an investigational antiviral drug designed to treat and inhibit influenza virus infections. It belongs to a class of compounds known as cap-dependent endonuclease (CEN) inhibitors, which target a key viral enzyme necessary for influenza virus replication.
Mechanism of Action
- Blocks viral replication: Pixavir marboxil works by inhibiting the influenza virus’s cap-dependent endonuclease, a part of the viral RNA polymerase complex the virus needs to “snatch” capped RNA fragments from host cell mRNA. Without this process, the virus cannot efficiently produce its own viral proteins or replicate.
What Viruses It Targets
Pixavir marboxil has shown activity against:
- Influenza A viruses
- Influenza B viruses
- Certain drug-resistant influenza strains
This broad spectrum makes it useful for seasonal flu and potentially strains less responsive to older antiviral drugs
Clinical Development & Approval Status
Phase Trials & Results
- Completed Phase III: Clinical trials in adults and adolescents (age ≥12) showed that a single dose shortened time to symptom relief compared to placebo (e.g., median ~60.9 h vs ~87.9 h).
- Symptom relief benefits: The data indicated statistically significant improvement in flu symptoms and faster viral inactivation in treated patients versus placebo.
- Pediatric Formulation: China’s health authority approved pediatric Phase III studies for Pixavir (children <12), indicating further development for younger patients.
Regulatory Filings
- NDA (New Drug Application): Pixavir marboxil has been submitted for approval to the National Medical Products Administration (NMPA) in mainland China based on Phase III results.
- Generic Name Approved: The drug has been officially recognized with the generic name “Pixavir marboxil,” moving it closer to commercialization.
Pixavir marboxil is a small molecule drug. The usage of the INN stem ‘-xavir’ in the name indicates that Pixavir marboxil is a influenza CAP-dependent endonuclease inhibitor. Pixavir marboxil has a monoisotopic molecular weight of 540.12 Da.
SYN
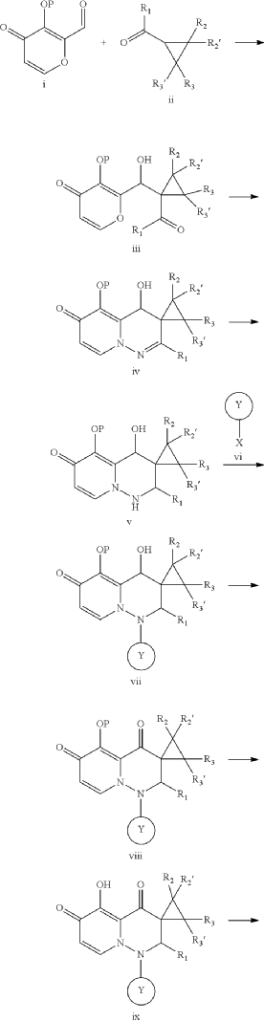
PAT
- Cap-dependent endonuclease inhibitorsPublication Number: US-10596171-B2Patent Family: AU-2019209426-A1; AU-2019209426-B2; BR-112020014810-A2; CA-3078391-A1; CA-3078391-C; CL-2020001919-A1; CN-110300753-A; CN-110300753-B; CO-2020006411-A2; EA-202090658-A1; EP-3743424-A1; EP-3743424-A4; IL-274199-A; IL-274199-B; JO-P20200159-A1; JP-2021512146-A; JP-6994121-B2; KR-102432975-B1; KR-20200086385-A; MX-2020007722-A; MY-197875-A; NZ-763248-A; PE-20211240-A1; PH-12020550921-A1; SG-11202003014V-A; TW-201938166-A; TW-I714951-B; US-10596171-B2; US-2019224198-A1; WO-2019144089-A1; ZA-202002037-BPriority Date: 2018-01-22Grant Date: 2020-03-24Inventor(s): LIN CHU-CHUNG; CHEN HUNG-CHUAN; CHIANG CHIAYN; YEN CHI-FENG; HSU MING-CHUAssignee(s): TAIGEN BIOTECHNOLOGY CO LTD; HSU MING CHUClassification: A61K31/5025; A61P31/16Abstract: Provided is a compound of Formula (I) below, or a pharmaceutically acceptable salt, metabolite, or prodrug thereof: n nwherein: A 1 is CR 4 or N; A 2 is CR 5 R 6 or NR 7 ; A 3 is CR 5 ′R 6 ′ or NR 7 ′; each of R 1 , R 2 , R 2 ′, R 3 , R 3 ′, R 4 , R 5 , R 5 ′, R 6 , R 6 ′, R 7 , and R 7 ′, independently, is hydrogen, deuterium, halogen, cyano, hydroxyl, carboxyl, amino, formyl, nitro, C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, C 1-6 alkoxy, C 2-6 alkenyloxy, C 1-6 alkylcarbonyl, C 1-6 alkyloxycarbonyl, C 1-6 alkylamine, C 3-20 carbocyclyl, or C 3-20 heterocyclyl; or R 5 and R 6 , R 5 ′ and R 6 ′, or R 5 and R 5 ′, together with the adjacent atom to which they are each attached, form C 3-10 carbocyclyl or C 3-10 heterocyclyl. Further provided are a method of using the above-described compound, or the pharmaceutically acceptable salt, metabolite, or prodrug thereof for treating influenza and a pharmaceutical composition containing same.Linked Compounds: 274Linked Substances: 411
- Cap-dependent endonuclease inhibitorsPublication Number: US-2019224198-A1Priority Date: 2018-01-22Linked Compounds: 275Linked Substances: 411



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

////////////pixavir marboxil, antiviral, TG 1000, Yi Li Kang, SV42843XSX, Cap-dependent endonuclease-IN-1, Influenza virus infections, TaiGen Biotechnology
Dezecapavir




Dezecapavir
CAS 2570323-59-8
MF C37H29ClF9N9O5S MW918.19
1H-Cyclopropa[3,4]cyclopenta[1,2-c]pyrazole-1-acetamide, N-[(1S)-1-[(3S)-3-[4-chloro-1-methyl-3-[(methylsulfonyl)amino]-1H-indazol-7-yl]-3,4-dihydro-4-oxo-7-(3,3,3-trifluoropropoxy)pyrido[2,3-d]pyrimidin-2-yl]-2-(3,5-difluorophenyl)ethyl]-3-(difluoromethyl)-5,5-difluoro-3b,4,4a,5-tetrahydro-, (3bS,4aR)-
N-[(1S)-1-[(3P)-3-[4-chloro-3-(methanesulfonamido)-1-methyl-1H-indazol-7-yl]-4-oxo-7-(3,3,3-
trifluoropropoxy)-3,4-dihydropyrido[2,3-d]pyrimidin-2-yl]-2-(3,5-difluorophenyl)ethyl]-2-[(3bS,4aR)-3-
(difluoromethyl)-5,5-difluoro-3b,4,4a,5-tetrahydro-1Hcyclopropa[3,4]cyclopenta[1,2-c]pyrazol-1-
yl]acetamide
inhibitor of viral replication, antiviral, SEK9LN2LSM, VH 4011499, VH4011499, VH-4011499, VH 499, GSK2838232, GSK 2838232
Dezecapavir is a potent experimental antiviral compound, specifically a novel HIV-1 capsid inhibitor, developed to block HIV replication by targeting the virus’s capsid protein, showing high effectiveness in lab settings (low nM range EC50) and representing a new class of drugs for HIV treatment, potentially for long-acting injectable therapies. It’s a complex molecule with a unique structure designed to disrupt the HIV capsid assembly, halting the virus’s life cycle early on.
Key Characteristics:
- Mechanism: Inhibits HIV-1 capsid assembly, a crucial step in the viral lifecycle.
- Potency: Very effective in cell cultures, with a low nanomolar EC50 (effective concentration).
- Class: Belongs to a new class of antivirals, distinct from integrase or reverse transcriptase inhibitors, offering a novel approach to HIV treatment.
- Development: Under investigation, often mentioned as a potential candidate for long-acting injectable (LAI) treatments due to its potency.
What it does:
Dezecapavir binds to the HIV capsid, preventing the virus from uncoating and maturing, thereby stopping new infections from forming.
Significance:
It represents a promising new option for HIV treatment, especially in the context of growing resistance to existing drugs, and could be part of future long-acting regimens.
SYN
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2020254985&_cid=P20-MKHOOT-76990-1

Preparation of Example 1: N-((S)-l-((3P)-3-(4-chloro-l-methyl-3-(methylsulfonamido)-lH- indazol-7-yl)-4-oxo-7-(3,3,3-trifluoropropoxy)-3,4-dihydropyrido[2,3-d]pyrimidin-2-yl)-2-(3,5- difluorophenyl)ethyl)-2-((3bSr4aR)-3-(difluoromethyl)-5r5-difluoro-3br4r4ar5-tetrahydro-lH- cyciopropa[3, 4]cydopenta[ l,2-c]pyrazoi-l-yi)acetamide

A solution of diisopropyl (E)-diazene-l,2-dicarboxylate (“DIAD”, 0.125 ml, 0.637 mmol) in THF (0.2 mL) was added dropwise to a mixture of N-(l-((3P)-3-(4-chloro-3-(N-(4-methoxybenzyl)methylsulfonamido)-l-methyl-lH-indazol-7-yl)-7-hydroxy-4-oxo-3,4-dihydropyrido[2,3-d]pyrimidin-2-yl)-2-(3,5-difluorophenyl)ethyl)-2-((3bS,4aR)-3-(difluoromethyl)-5,5-difluoro-3b,4,4a,5-tetrahydro-lH-cyclopropa[3,4]cyclopenta[l,2-c]pyrazol-l-yl)acetamide (0.2 g, 0.212 mmol)), 3,3,3-trifluoropropan-l-ol (0.073 g, 0.637 mmol) and triphenylphosphine (0.178 g, 0.679 mmol) in Tetrahydrofuran (2.1 mL) at rt. The reaction mixture was stirred for 18 h at rt and then was concentrated in vacuo. The residue was purified on silica gel (24 g RediSep Gold column) using a gradient of 0-60 % ethyl acetate in hexanes over 15 CV, and then holding at 60 % ethyl acetate in hexanes for 5 CV. Fractions containing the pure product were pooled and then concentrated to give a yellow solid. This solid was taken up in DCM (1 mL):TFA (0.5 mL); the solution was cooled to 0 °C; and to the solution was added triflic acid (0.057 mL, 0.637 mmol). The mixture was stirred for 1 h and then concentrated in vacuo. The residue was taken up in ethyl acetate; washed with 1 N
NaOH; washed with 0.5M citric acid; dried over Na2SC>4; filtered; and then was concentrated in vacuo. The residue was subjected to silica gel chromatography (24 g RediSep Gold column) using 0-60 % ethyl acetate in hexanes over 20 CV, then at 60 % ethyl acetate for 10 CV. Fractions containing the pure product were pooled and then concentrated in vacuoto give N-(l-((6P)-3-(4-chloro-l-methyl-3-(methylsulfonamido)-lH-indazol-7-yl)-4-oxo-7-(3,3,3-trifluoropropoxy)-3,4-dihydropyrido[2,3-d]pyrimidin-2-yl)-2-(3,5-difluorophenyl)ethyl)-2-((3bS,4aR)-3-(difluoromethyl)-5,5-difluoro-3b,4,4a,5-tetrahydro-lH-cyclopropa[3,4]cyclopenta[l,2-c]pyrazol-l-yl)acetamide (0.078 g, 0.081 mmol, 38.0 % yield) as a brown solid. *H NMR (500 MHz, METHANOL-d^ d ppm 8.46 – 8.53 (m, 1 H) 7.28 – 7.34 (m, 1 H) 7.19 – 7.24 (m, 1 H) 7.03 – 7.09 (m, 1 H) 6.53 – 6.81 (m, 4 H) 4.80 (dd, J=5.96, 2.98Hz, 3 H) 4.49 – 4.62 (m, 2 H) 3.58 – 3.62 (m, 3 H) 3.40 – 3.49 (m, 1 H) 3.22 – 3.24 (m, 3 H) 3.06 – 3.14 (m, 1 H) 2.80 – 2.89 (m, 2 H) 2.37 – 2.44 (m, 2 H) 1.32 – 1.37 (m, 1 H) 0.96 – 1.01 (m, 1 H). LCMS Analysis Method: Column = Acquity UPLC BEH C18, 2.1 x 100 mm, 1.7 pm particles; Injection Volume = 5.00 pL; Flowrate = 0.80 mL/min; Solvent A = 95:5
WatenMeCN w/ 0.1% v/v formic acid; Solvent B = 5:95 WatenMeCN w/ 0.1% v/v formic acid;
Elution profile = Start %B: 0, End %B: 100, Gradient Time: 3.5 min. then hold at 100% B for 1 min.; Detection wavelength 1 = 220 nm, wavelength 2 = 254 nm. LCMS retention time = 3.097 min; m/z = 918.05 [M+l]+.



PAT
- Pyrido[2,3-d]pyrimidine derivatives as inhibitors of human immunodeficiency virus replicationPublication Number: US-2021323967-A1Priority Date: 2019-06-19
- Pyrido[2,3-d]pyrimidine derivatives as inhibitors of human immunodeficiency virus replicationPublication Number: US-2025019383-A1Priority Date: 2019-06-19
- Pyrido[2,3-D]pyrimidine derivatives as inhibitors of human immunodeficiency virus replicationPublication Number: KR-20220024608-APriority Date: 2019-06-19
- Pyrido[2,3-d]pyrimidine derivatives as inhibitors of human immunodeficiency virus replicationPublication Number: EP-3986561-A1Priority Date: 2019-06-19
- Pyrido[2,3-d]pyrimidine derivatives as inhibitors of human immunodeficiency virus replicationPublication Number: EP-3986561-B1Priority Date: 2019-06-19Grant Date: 2024-02-14
- Pyrido [2,3-d]pyrimidine derivatives as inhibitors of human immunodeficiency virus replicationPublication Number: US-12129255-B2Priority Date: 2019-06-19Grant Date: 2024-10-29
- Pyrido[2,3-d]pyrimidine derivatives as inhibitors of human immunodeficiency virus replicationPublication Number: WO-2020254985-A1Priority Date: 2019-06-19
- Inhibitors of human immunodeficiency virus replicationPublication Number: EP-4415685-A1Priority Date: 2021-10-13
- Inhibitors of human immunodeficiency virus replicationPublication Number: WO-2023062559-A1Priority Date: 2021-10-13
- Inhibitors of human immunodeficiency virus replicationPublication Number: US-2024423985-A1Priority Date: 2021-10-13
- Inhibitors of human immunodeficiency virus replicationPublication Number: US-2023149408-A1Priority Date: 2020-04-15
- Pharmaceutical compositions comprising cabotegravirPublication Number: US-2023045509-A1Priority Date: 2019-12-09
ADVERTISEMENT
BLUE JET HEALTHCARE LTD, https://bluejethealthcare.com
Looking for a Reliable SNAC Manufacturer? Let’s Talk.
At Blue Jet Healthcare Ltd, we specialize in the scalable, high-purity production of SNAC—a critical excipient powering the next generation of oral peptide therapeutics.
With increasing demand for SNAC across global pharma pipelines, choosing the right manufacturing partner is essential. Quality, timelines, and consistency matter.

Phone No. +91 (22) 22075307 / +91 (22) 22071691
Business Development/ Contract Manufacturing: marketing1@bluejethealthcare.com, madhu.gautam71@gmail.com



AS ON OCT2025 4.511 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
NEXT
ADVERTISEMENT
Advect Process Systems Ltd.
ADVECT PROCESS SYSTEMS CANADA LTD
51 Beechwood Rd., Cambridge, ON Canada N1S 3S1
Call Now +1 306 850 6737, Mail Now, ask@advectprocess.com
ADVERTISEMENT
BLUE JET HEALTHCARE LTD, https://bluejethealthcare.com
Looking for a Reliable SNAC Manufacturer? Let’s Talk.
At Blue Jet Healthcare Ltd, we specialize in the scalable, high-purity production of SNAC—a critical excipient powering the next generation of oral peptide therapeutics.
With increasing demand for SNAC across global pharma pipelines, choosing the right manufacturing partner is essential. Quality, timelines, and consistency matter.

Phone No. +91 (22) 22075307 / +91 (22) 22071691
Business Development/ Contract Manufacturing: marketing1@bluejethealthcare.com, madhu.gautam71@gmail.com
//////////dezecapavir, inhibitor of viral replication, antiviral, SEK9LN2LSM, VH 4011499, VH4011499, VH-4011499, VH 499, GSK2838232, GSK 2838232
Limnetrelvir



Limnetrelvir
CAS 2923500-04-1
MF C27H23F4N5O4 MW 557.50
N-[(3R)-1-[4-cyano-2-(morpholine-4-carbonyl)-6-(trifluoromethyl)phenyl]pyrrolidin-3-yl]-8-fluoro-2-oxo-1H-quinoline-4-carboxamide
N-{(3R)-1-[4-cyano-2-(morpholine-4-carbonyl)-6-
(trifluoromethyl)phenyl]pyrrolidin-3-yl}-8-fluoro-2-oxo1,2-dihydroquinoline-4-carboxamide
antiviral, ABBV-903, ABBV 903, 4TPS988XGG
Limnetrelvir (ABBV-903) is a MPro inhibitor. Limnetrelvir could be used in antiviral research.
SYN

Example 1 – Synthesis of Compound (2) (R)-N-(1-(4-cyano-2-(morpholine-4-carbonyl)-6-(trifluoromethyl)phenyl)pyrrolidin-3-yl)-8-fluoro-2-oxo-1,2-dihydroquinoline-4-carboxamide

Compound 2F – Synthesis of 8-fluoro-2-oxo-1,2-dihydroquinoline-4-carboxylic acid

[00035] A suspension of 7-fluoroindoline-2,3-dione (55 g, 333 mmol), malonic acid (41.6 g, 400 mmol) and sodium acetate (68.3 g, 833 mmol) in acetic acid (500 mL) was heated at 112 °C overnight. The reaction mixture was cooled to room temperature and poured into cold 0.4 M aqueous HCl (2200 mL). The precipitate was collected by filtration and rinsed thoroughly with ice-cold water (~250 mL) followed by methyl tert-butyl ether (~100 mL) and then concentrated twice from acetonitrile with high vacuum. The materials were largely dissolved into 1 M aqueous NaOH (370 mL) and filtered through diatomaceous earth with a 0.1 M aqueous NaOH (50 mL) rinse. Then the filtrate was washed thrice with dichloromethane (3 x 200 mL) which removed the color. After this aqueous layer was filtered again through diatomaceous earth, it was acidified by the dropwise addition of concentrated aqueous HCl (33 mL, ~0.4 moles). The material was collected by filtration. After prolonged drying under heat and vacuum, the material was treated with water (1 L) and the mixture was made acidic by the addition of a small amount of 1 M aqueous HCl. The suspension was heated to 80 °C and then allowed to slowly cool to room temperature. The resulting material was collected by filtration, washed with 0.01 M aqueous HCl (150 mL) and dried under vacuum at 80 °C to provide the title compound (2F).1H NMR (500 MHz, DMSO-d6) δ ppm 14.00 (bs, 1H), 12.07 (bs, 1H), 8.00 (dd, J = 8.2, 1.2 Hz, 1H), 7.49 (ddd, J = 11.0, 8.1, 1.2 Hz, 1H), 7.23 (ddd, J = 8.2, 8.1, 5.2 Hz, 1H), 6.95 (s, 1H); 13C NMR (101 MHz, DMSO-d6, 90 °C) δ ppm 165.75 – 165.73 (m), 160.30, 148.75 (d, J = 246.0 Hz), 140.85 – 140.80 (m), 128.11 (d, J = 13.7 Hz), 123.85, 121.60 – 121.53 (m), 121.53 – 121.43 (m), 117.70 – 117.65 (m), 115.33 (d, J = 17.2 Hz); 19F NMR (376 MHz, DMSO-d6, 90 °C) δ ppm -130.47 (dd, J = 10.9, 5.3 Hz); MS (APCI, M+H+) m/z 208.
Compound 2G – Synthesis of (R)-N-(1-(4-cyano-2-(morpholine-4-carbonyl)-6-(trifluoromethyl)phenyl)pyrrolidin-3-yl)-8-fluoro-2-oxo-1,2-dihydroquinoline-4-carboxamide (2)

00036] To a mixture of Compound 2F (29.84 g, 144 mmol) in anhydrous N,N-dimethylformamide (360 mL) was added DMTMM (4-(4,6-dimethoxy-1,3,5-triazin-2-yl)-4-
methylmorpholinium chloride) (43.17 g, 156 mmol) over twelve minutes at room temperature. After the suspension had been stirred forty minutes, it was added over eight minutes to a suspension of Compound 2E (≤120 mmol) and N-methylmorpholine (16 mL, 146 mmol) in N,N-dimethylformamide (120 mL) with a N,N-dimethylformamide (20 mL) rinse. After forty minutes, the reaction mixture was added to rapidly stirred 0.1 M aqueous K2HPO4 (2.5 L) and extracted four times with 4:1 isopropyl acetate / heptanes then once with isopropyl acetate alone. The product, which had begun to precipitate out from the combined extracts, was separated by decantation and filtration, then washed with dichloromethane. The remaining aqueous phase was extracted twice more with isopropyl acetate and all the organic extracts were combined, then washed with additional 0.1 M aqueous K2HPO4 followed by water, dried (Na2SO4), and filtered. The filtrate was concentrated with the dichloromethane wash of the material collected above. The residue was concentrated, dissolved in acetonitrile / CH2Cl2, filtered, and purified by chromatography on silica (20 to 100% acetonitrile / CH2Cl2). The collected fractions were concentrated to a small volume, and stirred in ethyl acetate overnight.
[00037] The suspension was heated at 70 °C for twenty minutes, then allowed to slowly cool to room temperature. Methyl tert-butyl ether was stirred in, the suspension was cooled to 0 °C, and the purified product was collected by filtration with rinses of 1:1 ethyl acetate / methyl tert-butyl ether followed by methyl tert-butyl ether before being dried under vacuum with heat. The material obtained previously from the early extracts were also stirred in ethyl acetate, heated at 70 °C, then allowed to slowly cool to room temperature. Methyl tert-butyl ether was stirred in, the suspension was cooled to 0 °C, and the purified product was collected by filtration with rinses of 1:1 ethyl acetate / methyl tert-butyl ether rinse followed by methyl tert-butyl ether. The material was dried overnight under vacuum to provide the title compound (2).1H NMR (500 MHz, DMSO-d6) δ ppm 11.96 (s, 1H), 9.03 – 8.84 (m, 1H), 8.21 – 8.18 (m, 1H), 7.98 – 7.93 (m, 1H), 7.56 – 7.50 (m, 1H), 7.49 – 7.43 (m, 1H), 7.21 – 7.15 (m, 1H), 6.66 – 6.59 (m, 1H), 4.51 – 4.40 (m, 1H), 3.73 – 3.55 (m, 6H), 3.54 – 3.22 (m, 6H), 2.29 – 2.18 (m, 1H), 2.07 – 1.95 (m, 1H); 1H NMR (400 MHz, DMSO-d6, 90 °C) δ ppm 11.47 (bs, 1H), 8.77 – 8.47 (m, 1H), 8.09 (d, J = 2.1 Hz, 1H), 7.88 (d, J = 2.1 Hz, 1H), 7.54 (dd, J = 8.1, 1.2 Hz, 1H), 7.39 (ddd, J = 11.0, 8.1, 1.2 Hz, 1H), 7.15 (ddd, J = 8.1, 8.1, 5.1 Hz, 1H), 6.60 (s, 1H), 4.54 – 4.43 (m, 1H), 3.74 – 3.20 (m, 12H), 2.31 – 2.21 (m, 1H), 2.06 – 1.96 (m, 1H); 13C NMR (101 MHz, DMSO-d6, 90 °C) δ
ppm 166.61, 165.97, 161.31, 149.59 (d, J = 246.3 Hz), 148.95, 146.10 – 146.03 (m), 136.00, 135.65, 133.13 (q, J = 6.1 Hz), 128.84 – 128.63 (m), 123.76 (q, J = 273.7 Hz), 122.19, 122.16, 122.12, 121.60, 118.99 – 118.91 (m), 117.75, 116.17 (d, J = 17.3 Hz), 105.57, 66.04, 57.95, 51.06, 50.35, 47.74, 42.35, 31.54; 19F NMR (376 MHz, DMSO-d6) δ ppm -57.54 – -58.10 (m), -130.02 – -130.15 (m); 19F NMR (376 MHz, DMSO-d6, 90 °C) δ ppm -58.37 – -58.97 (m), -130.96 (dd, J = 11.0, 5.1 Hz). MS (APCI, M+H+) m/z 558.






PAT
- Pyrrolidine Main Protease Inhibitors as Antiviral AgentsPublication Number: US-2024158368-A1Priority Date: 2022-10-14
- Pyrrolidine main protease inhibitors as antiviral agentsPublication Number: WO-2024081351-A1Priority Date: 2022-10-14



AS ON OCT2025 4.511 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
/////////limnetrelvir, antiviral, ABBV-903, ABBV 903, 4TPS988XGG
Iscartrelvir



Iscartrelvir
CAS 2921711-74-0
MF 2921711-74-0, 526.4 g/mol
N-{(1S,2R)-2-[4-bromo-2-(methylcarbamoyl)-6-nitroanilino]cyclohexyl}isoquinoline-4-carboxamide
antiviral, WU-04, WU 04, W2LTV65R5E
Iscartrelvir is an investigational new drug developed by the Westlake University for the treatment of COVID-19. It targets the SARS-CoV-2 3CL protease, which is crucial for the replication of the virus responsible for COVID-19.[1][2]
Iscartrelvir is a small molecule drug. The usage of the INN stem ‘-trelvir’ in the name indicates that Iscartrelvir is a antiviral 3CL protease inhibitor. Iscartrelvir has a monoisotopic molecular weight of 525.1 Da.
PAT
WO2022150962A1
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2022150962&_cid=P11-MJKTXT-76321-1

SYN
https://patentscope.wipo.int/search/en/detail.jsf?docId=CN331401594&_cid=P11-MJKTO7-65334-1
PAT
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2024243841&_cid=P11-MJKTO7-65334-1
N-((1S,2R)-2-((4-bromo-2-(methylcarbamoyl)-6-nitrophenyl)amino)cyclohexyl)isoquinoline-4-carboxamide, and its structure is as follows:

Example 1: Preparation of Compound 1
[0189]A free, amorphous compound 1, a yellow solid, was prepared according to the method disclosed in paragraphs [00121]-[00128] of WO2022150962A1, and was used in the following examples. The specific synthetic steps are shown in steps a to d:

The reagents and conditions for steps a to d are further described below: (a) 1-[bis(dimethylamino)methylene]-1H-1,2,3-triazolo[4,5-b]pyridinium 3-oxide hexafluorophosphate (HATU), N,N-diisopropylethylamine (DIPEA), CH₂Cl₂
or
dichloromethane (DCM), 0°C, 2 h; (b) DIPEA, dimethylformamide (DMF), 80°C, 16 h; (c) 3M ethyl hydrochloride (HCl·EA), CH₂Cl₂ , 1
h ; (d ) HATU, DIPEA, DMF, room temperature, 12 h.
[0191]Step a: Synthesis of N-methyl-5-bromo-2-fluoro-3-nitrobenzamide (I-1)
[0192]A solution of 5-bromo-2-fluoro-3-nitrobenzoic acid (0.8 g, 3.80 mmol) in dichloromethane (20 mL) was stirred at 0 °C. Then, HATU (2.0 g, 5.25 mmol), DIPEA (1.88 mL, 11.4 mmol), and methylamine hydrochloride (0.31 g, 4.5 mmol) were added to the reaction mixture. The mixture was stirred at 0 °C for 2 hours until it became clear. The mixture was extracted three times with dichloromethane, and the combined organic layers were washed with a saturated brine solution. The organic phase was then dried over anhydrous Na₂SO₄ and concentrated
under vacuum. Finally, the mixture was purified by chromatography to give compound I-1 (0.8 g, 76% yield) as a yellow solid.
[0193]Step b: Synthesis of tert-butyl 2-((4-bromo-2-(methylcarbamoyl)-6-nitrophenyl)amino)cyclohexyl)carbamate (I-2)
[0194]A solution of compound I-1 (0.8 g, 2.9 mmol) in 15 mL of DMF was stirred at room temperature. Then, tert-butyl ((1S,2R)-2-aminocyclohexyl)carbamate (0.75 g, 3.5 mmol) (the corresponding stereoisomer of this reagent can be used to synthesize the stereoisomer of compound I-2) and DIPEA (1.44 mL, 8.7 mmol) were added to the reaction mixture. The mixture was heated to 80 °C and stirred for 16 hours. The mixture was extracted three times with ethyl acetate, and the combined organic layers were washed with saturated salt solution. The organic phase was then dried over anhydrous Na₂SO₄ and concentrated under vacuum to give compound
I -2 as a yellow solid, requiring no further purification.

Step c: Synthesis of 2-(2-aminocyclohexyl)amino)-5-bromo-N-methyl-3-nitrobenzamide hydrochloride (I-3)
[0196]A solution of compound I-2 (90 mg, 0.19 mmol) (or the corresponding stereoisomer) in anhydrous dichloromethane (6 mL) was stirred at room temperature. Then, HCl (4 mL, 3 M in ethyl acetate) was added. The mixture was stirred at room temperature for 2 hours. The mixture was concentrated under vacuum to give compound I-3 as a yellow solid, requiring no further purification.
[0197]Step d: Synthesis of N-((1S,2R)-2-((4-bromo-2-(methylcarbamoyl)-6-nitrophenyl)amino)cyclohexyl)isoquinoline-4-carboxamide
[0198]At room temperature, a solution of the corresponding isoquinoline-4-carboxylic acid (1 equivalent) and HATU (1.5 equivalent) in anhydrous DMF (6 mL) was stirred. Then, compound I-3 and DIPEA (5.0 equivalent) were added. The mixture was stirred overnight at room temperature. The mixture was extracted three times with ethyl acetate, and the combined organic layers were washed with saturated brine. The organic phase was then dried over anhydrous Na₂SO₄ and
concentrated under vacuum. Finally, the mixture was purified by chromatography to give compound 1 as a free amorphous solid in yellow form.
PAT
- Aromatic ring-containing pyridone amide compoundsPublication Number: CN-119100980-APriority Date: 2023-06-07
- Crystal of viral protease inhibitor and usePublication Number: WO-2024243841-A1Priority Date: 2023-05-31
- Protease inhibitors, their preparation and usePublication Number: CN-113072497-BPriority Date: 2021-01-12Grant Date: 2023-04-14
- Protease inhibitors, their preparation and usePublication Number: CN-113072497-APriority Date: 2021-01-12
- Protease inhibitors, their preparation and usePublication Number: CN-116751164-APriority Date: 2021-01-12



AS ON OCT2025 4.511 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
| Clinical data | |
|---|---|
| Other names | WPV01; WU-04 |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2921711-74-0 |
| PubChem CID | 156774920 |
| ChemSpider | 129307041 |
| UNII | W2LTV65R5E |
| PDB ligand | J7R (PDBe, RCSB PDB) |
| Chemical and physical data | |
| Formula | C24H24BrN5O4 |
| Molar mass | 526.391 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
References
- Yang L, Wang Z (September 2023). “Bench-to-bedside: Innovation of small molecule anti-SARS-CoV-2 drugs in China”. European Journal of Medicinal Chemistry. 257 115503. doi:10.1016/j.ejmech.2023.115503. PMC 10193775. PMID 37229831.
- Hou N, Shuai L, Zhang L, Xie X, Tang K, Zhu Y, et al. (February 2023). “Development of Highly Potent Noncovalent Inhibitors of SARS-CoV-2 3CLpro”. ACS Central Science. 9 (2): 217–227. doi:10.1021/acscentsci.2c01359. PMC 9885526. PMID 36844503.
- Resistance mechanisms of SARS-CoV-2 3CLpro to the non-covalent inhibitor WU-04Publication Name: Cell DiscoveryPublication Date: 2024-04-09PMCID: PMC11003996PMID: 38594245DOI: 10.1038/s41421-024-00673-0
- Identification of Ebselen derivatives as novel SARS-CoV-2 main protease inhibitors: Design, synthesis, biological evaluation, and structure-activity relationships explorationPublication Name: Bioorganic & Medicinal ChemistryPublication Date: 2023-12-15PMID: 37972434DOI: 10.1016/j.bmc.2023.117531
- The molecular mechanism of non-covalent inhibitor WU-04 targeting SARS-CoV-2 3CLpro and computational evaluation of its effectiveness against mainstream coronavirusesPublication Name: Physical chemistry chemical physics : PCCPPublication Date: 2023-09-13PMID: 37655706DOI: 10.1039/d3cp03828a
- Bench-to-bedside: Innovation of small molecule anti-SARS-CoV-2 drugs in ChinaPublication Name: European Journal of Medicinal ChemistryPublication Date: 2023-09-05PMCID: PMC10193775PMID: 37229831DOI: 10.1016/j.ejmech.2023.115503
- Development of Highly Potent Noncovalent Inhibitors of SARS-CoV-2 3CLproPublication Name: ACS Central SciencePublication Date: 2023-01-25PMCID: PMC9885526PMID: 36844503DOI: 10.1021/acscentsci.2c01359
////////iscartrelvir, antiviral, WU-04, WU 04, W2LTV65R5E
Odentegravir



Odentegravir
CAS 2495436-99-0
MF C20H18F3N3O4 MW421.4 g/mol
(7S)-12-hydroxy-1,11-dioxo-N-[(2,4,6-trifluorophenyl)methyl]-1,4,5,6,7,11-hexahydro-3H-2,7-
methanopyrido [1,2-a][1,4]diazonine-10-carboxamide
(7S)-1,4,5,6,7,11-HEXAHYDRO-12-HYDROXY-1,11-DIOXO-N-((2,4,6-TRIFLUOROPHENYL)METHYL)-3H-2,7-METHANOPYRIDO(1,2-A)(1,4)DIAZONINE-10-CARBOXAMIDE
(7S)-12-HYDROXY-1,11-DIOXO-N-((2,4,6-TRIFLUOROPHENYL)METHYL)-1,4,5,6,7,11-HEXAHYDRO-3H-2,7-METHANOPYRIDO(1,2-A)(1,4)DIAZONINE-10-CARBOXAMIDE
3H-2,7-METHANOPYRIDO(1,2-A)(1,4)DIAZONINE-10-CARBOXAMIDE, 1,4,5,6,7,11-HEXAHYDRO-12-HYDROXY-1,11-DIOXO-N-((2,4,6-TRIFLUOROPHENYL)METHYL)-, (7S)-
antiviral, H8B26JZ4A4, orb2664247
Odentegravir is a small molecule drug classified as a
HIV integrase inhibitor, indicated by the “-tegravir” stem in its name. It is a chemical compound with the molecular formula
has been used in research for its antiviral properties.
- Drug Class: HIV integrase inhibitor
- Chemical Formula:
C20H18F3N3O4cap C sub 20 cap H sub 18 cap F sub 3 cap N sub 3 cap O sub 4𝐶20𝐻18𝐹3𝑁3𝑂4
- Molecular Weight:
421.12421.12421.12 Da (monoisotopic)
- Classification: Small molecule drug
SYN
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2020197991&_cid=P12-MHY8KB-06018-1
Example 23: Preparation of racemic-12-hydroxy-1,11-dioxo-N-(2,4,6-trifluorobenzyl)-1,4,5,6,7,11-hexahydro-3H-2,7-methanopyrido[1,2-a][1,4]diazonine-10-carboxamide (26), (7R)-12-hydroxy-1,11-dioxo-N-(2,4,6-trifluorobenzyl)-1,4,5,6,7,11-hexahydro-3H-2,7-
methanopyrido[1,2-a][1,4]diazonine-10-carboxamide (26-1) and (7S)-12-hydroxy-1,11-dioxo-N-(2,4,6-trifluorobenzyl)-1,4,5,6,7,11-hexahydro-3H-2,7-methanopyrido[1,2-a][1,4]diazonine-10-carboxamide (26-2):

Synthesis of 12-Hydroxy-1,11-dioxo-N-(2,4,6-trifluorobenzyl)-1,4,5,6,7,11-hexahydro-3H-2,7-methanopyrido[1,2-a][1,4]diazonine-10-carboxamide (26):
[0335] 12-(Benzyloxy)-1,11-dioxo-1,4,5,6,7,11-hexahydro-3H-2,7-methanopyrido[1,2-a][1,4]diazonine-10-carboxylic acid (57 mg, 0.155 mmol) was dissolved in DCM (2 mL) with (2,4,6-trifluorophenyl)methanamine (27 mg, 0.17 mmol) and triethylamine (60 mg, 0.464 mmol). HATU (60 mg, 0.186 mmol) was added and the mixture was stirred at room
temperature. After overnight reaction, the reaction was concentrated to dryness, purified by silicon gel chromatography to obtain compound 12-(benzyloxy)-1,11-dioxo-N-(2,4,6-trifluorobenzyl)-1,4,5,6,7,11-hexahydro-3H-2,7-methanopyrido[1,2-a][1,4]diazonine-10-carboxamide (26a) MS (m/z) 512.06 [M+H]+.
[0336] Compound 12-(benzyloxy)-1,11-dioxo-N-(2,4,6-trifluorobenzyl)-1,4,5,6,7,11-hexahydro-3H-2,7-methanopyrido[1,2-a][1,4]diazonine-10-carboxamide (26a) (7 mg, 0.014 mmol) was dissloved in Tolune (1 mL), then followed by the addition of TFA (1 mL). The resulting mixture was stirred at rt for overnight. The solvent was removed under vacuo an the residue was purifed by HPLC to obtain the title compound (26). MS (m/z) 422.091 [M+H]+.1H NMR (400 MHz, DMSO-d6) d 10.39 (t, J = 5.8 Hz, 1H), 8.45 (s, 1H), 7.24 – 7.11 (m, 2H), 4.72 (dd, J = 5.9, 2.9 Hz, 1H), 4.54 (dd, J = 6.0, 2.4 Hz, 2H), 4.11 (d, J = 13.3 Hz, 1H), 3.88 – 3.79 (m, 1H), 3.64 (dd, J = 14.7, 1.9 Hz, 1H), 3.05 (dq, J = 9.5, 3.4 Hz, 1H), 2.06 – 1.91 (m, 1H), 1.89 – 1.74 (m, 3H), 1.61 (d, J = 7.7 Hz, 1H), 1.11 (d, J = 12.7 Hz, 1H).
Synthesis of (7S)-12-hydroxy-1,11-dioxo-N-(2,4,6-trifluorobenzyl)-1,4,5,6,7,11-hexahydro-3H-2,7-methanopyrido[1,2-a][1,4]diazonine-10-carboxamide (26-2) and (7R)-12-hydroxy-1,11-dioxo-N-(2,4,6-trifluorobenzyl)-1,4,5,6,7,11-hexahydro-3H-2,7-methanopyrido[1,2-a][1,4]diazonine-10-carboxamide (26-1):
[0337] Racemic 12-(benzyloxy)-1,11-dioxo-N-(2,4,6-trifluorobenzyl)-1,4,5,6,7,11-hexahydro-3H-2,7-methanopyrido[1,2-a][1,4]diazonine-10-carboxamide (26a) was separated by chiral HPLC separation (SFC chromatography on an IB 4.6X100mm 5mic column using MeOH(20) as co-solvent) to obtain compounds (7R)-12-(Benzyloxy)-1,11-dioxo-N-(2,4,6-trifluorobenzyl)-1,4,5,6,7,11-hexahydro-3H-2,7-methanopyrido[1,2-a][1,4]diazonine-10-carboxamide (26a-1) and (7S)-12-(benzyloxy)-1,11-dioxo-N-(2,4,6-trifluorobenzyl)-1,4,5,6,7,11-hexahydro-3H-2,7-methanopyrido[1,2-a][1,4]diazonine-10-carboxamide (26a-2)
[0338] Compound (7S)-12-(benzyloxy)-1,11-dioxo-N-(2,4,6-trifluorobenzyl)-1,4,5,6,7,11-hexahydro-3H-2,7-methanopyrido[1,2-a][1,4]diazonine-10-carboxamide (26a-2) (20 mg, 0.039 mmol) was dissloved in Tolune (1 mL), then followed by the addition of TFA (1 mL). The resulting mixture was stireed at rt for overnight. The solvent was removed under vacuo an the residue was purifed by HPLC to obtain the title compound (26-2). (MS (m/z) 422.123 [M+H]+. 1H NMR (400 MHz, DMSO-d6) d 10.59 (s, 1H), 10.39 (d, J = 5.9 Hz, 1H), 8.45 (s, 1H), 7.18 (t, J = 8.6 Hz, 2H), 4.72 (s, 1H), 4.59 – 4.48 (m, 2H), 4.11 (d, J = 13.2 Hz, 1H), 3.85 (d, J = 14.6 Hz, 1H), 3.69 – 3.59 (m, 1H), 3.05 (ddd, J = 11.3, 6.7, 3.6 Hz, 1H), 1.97 (m, 1H), 1.87 – 1.71 (m, 3H), 1.67 – 1.55 (m, 1H), 1.10 (m, 1H).
[0339] Compound (7R)-12-(benzyloxy)-1,11-dioxo-N-(2,4,6-trifluorobenzyl)-1,4,5,6,7,11-hexahydro-3H-2,7-methanopyrido[1,2-a][1,4]diazonine-10-carboxamide (26a-1) ((20 mg, 0.039 mmol) was dissloved in Tolune (1 mL), then followed by the addition of TFA (1 mL). The resulting mixture was stireed at rt for overnight. The solvent was removed under vacuo an the residue was purifed by HPLC to obtain the title compound (26-1). MS (m/z) 422.116 [M+H]+. 1H NMR (400 MHz, DMSO-d6) d 10.58 (s, 1H), 10.39 (t, J = 5.8 Hz, 1H), 8.45 (s, 1H), 7.18 (dd, J = 9.2, 8.0 Hz, 2H), 4.73 (s, 1H), 4.58 – 4.49 (m, 2H), 4.11 (d, J = 13.3 Hz, 1H), 3.85 (d, J = 14.6 Hz, 1H), 3.65 (d, J = 14.2 Hz, 1H), 3.10 – 3.00 (m, 1H), 1.96 (m, 1H), 1.82 (d, J = 12.2 Hz, 3H), 1.61 (d, J = 7.4 Hz, 1H), 1.18 – 1.05 (m, 1H).
SYN
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2023196875&_cid=P12-MHY8FJ-02517-1
PAT
- Bridged tricyclic carbamoylpyridone compounds and their pharmaceutical usesPublication Number: JP-2025013503-APriority Date: 2019-03-22
- Bridged tricyclic carbamoylpyridone compounds and their pharmaceutical usesPublication Number: KR-102714084-B1Priority Date: 2019-03-22Grant Date: 2024-10-08
- Bridged tricyclic carbamoylpyridone compounds and their pharmaceutical usePublication Number: KR-20240151256-APriority Date: 2019-03-22
- Bridged Tricyclic Carbamoylpyridone Compounds and Their Pharmaceutical UsePublication Number: ES-2927041-T3Priority Date: 2019-03-22Grant Date: 2022-11-03
- Bridged tricyclic carbamoylpyridone compounds and their pharmaceutical usePublication Number: US-11548902-B1Priority Date: 2019-03-22Grant Date: 2023-01-10
- Bridged tricyclic carbamoylpyridone compounds and their pharmaceutical usePublication Number: US-2023027019-A1Priority Date: 2019-03-22
- Bridged tricyclic carbamoylpyridone compounds and their pharmaceutical usePublication Number: AU-2020245350-B2Priority Date: 2019-03-22Grant Date: 2023-04-20
- Bridged tricyclic carbamoylpyridone compounds and their pharmaceutical usePublication Number: US-2023203061-A1Priority Date: 2019-03-22
- Bridged tricyclic carbamoylpyridone compounds and their pharmaceutical usePublication Number: US-11084832-B2Priority Date: 2019-03-22Grant Date: 2021-08-10
- Bridged tricyclic carbamoylpyridone compounds and their pharmaceutical usePublication Number: AU-2020245350-A1Priority Date: 2019-03-22
- Bridged tricyclic carbamoylpyridone compounds and their pharmaceutical usePublication Number: EP-3938047-A1Priority Date: 2019-03-22
- Bridged tricyclic carbamoylpyridone compounds and their pharmaceutical usePublication Number: EP-3938047-B1Priority Date: 2019-03-22Grant Date: 2022-06-22
- Bridged tricyclic carbamoylpyridone compounds and their pharmaceutical usePublication Number: EP-4122537-A1Priority Date: 2019-03-22
- Bridged tricyclic carbamoylpyridone compounds and uses thereofPublication Number: US-2023339971-A1Priority Date: 2022-04-06
- Bridged tricyclic carbamoylpyridone compounds and uses thereofPublication Number: US-2023339972-A1Priority Date: 2022-04-06
- Bridged tricyclic carbamoylpyridone compounds and uses thereofPublication Number: WO-2023196875-A1Priority Date: 2022-04-06
- Bridged tricyclic carbamoylpyridone compounds and their pharmaceutical usePublication Number: US-2020317689-A1Priority Date: 2019-03-22
- Bridged tricyclic carbamoylpyridone compounds and their pharmaceutical usePublication Number: WO-2020197991-A1Priority Date: 2019-03-22



AS ON OCT2025 4.511 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
//////Odentegravir, antiviral, H8B26JZ4A4, orb2664247
Tixagevimab
| (Heavy chain) QMQLVQSGPE VKKPGTSVKV SCKASGFTFM SSAVQWVRQA RGQRLEWIGW IVIGSGNTNY AQKFQERVTI TRDMSTSTAY MELSSLRSED TAVYYCAAPY CSSISCNDGF DIWGQGTMVT VSSASTKGPS VFPLAPSSKS TSGGTAALGC LVKDYFPEPV TVSWNSGALT SGVHTFPAVL QSSGLYSLSS VVTVPSSSLG TQTYICNVNH KPSNTKVDKR VEPKSCDKTH TCPPCPAPEF EGGPSVFLFP PKPKDTLYIT REPEVTCVVV DVSHEDPEVK FNWYVDGVEV HNAKTKPREE QYNSTYRVVS VLTVLHQDWL NGKEYKCKVS NKALPASIEK TISKAKGQPR EPQVYTLPPS REEMTKNQVS LTCLVKGFYP SDIAVEWESN GQPENNYKTT PPVLDSDGSF FLYSKLTVDK SRWQQGNVFS CSVMHEALHN HYTQKSLSLS PGK (Light chain) EIVLTQSPGT LSLSPGERAT LSCRASQSVS SSYLAWYQQK PGQAPRLLIY GASSRATGIP DRFSGSGSGT DFTLTISRLE PEDFAVYYCQ HYGSSRGWTF GQGTKVEIKR TVAAPSVFIF PPSDEQLKSG TASVVCLLNN FYPREAKVQW KVDNALQSGN SQESVTEQDS KDSTYSLSST LTLSKADYEK HKVYACEVTH QGLSSPVTKS FNRGEC (Disulfide bridge: H22-H96, H101-H106, H150-H206, H216-L216, H232-H’232, H235-H’235, H267-H327, H373-H431, H’22-H’96, H’101-H’106, H’150-H’206, H’226-L’216, H’267-H’327, H’373-H’431, L23-L89, L136-L196, L’23-L’89, L’136-L’196) |
Tixagevimab
FDA 2021, 2021/12/8
ANTI VIRAL, CORONA VIRUS, PEPTIDE
Monoclonal antibody
Treatment and prevention of SARS-CoV-2 infection
| Formula | C6488H10034N1746O2038S50 |
|---|---|
| CAS | 2420564-02-7 |
| Mol weight | 146704.817 |
- 2196
- AZD-8895
- AZD8895
- COV2-2196
- Tixagevimab
- Tixagevimab [INN]
- UNII-F0LZ415Z3B
- WHO 11776
- OriginatorVanderbilt University
- DeveloperAstraZeneca; INSERM; National Institute of Allergy and Infectious Diseases
- ClassAntivirals; Monoclonal antibodies
- Mechanism of ActionVirus internalisation inhibitors
- RegisteredCOVID 2019 infections
- 24 Dec 2021Pharmacodynamics data from a preclinical trial in COVID-2019 infections released by AstraZeneca
- 16 Dec 2021Pharmacodynamics data from a preclinical trial in COVID-2019 infections released by AstraZeneca
- 10 Dec 2021Registered for COVID-2019 infections (In the elderly, Prevention, In adults) in USA (IM) – Emergency Use Authorization
Tixagevimab/cilgavimab is a combination of two human monoclonal antibodies, tixagevimab (AZD8895) and cilgavimab (AZD1061) targeted against the surface spike protein of SARS-CoV-2[4][5] used to prevent COVID-19. It is being developed by British-Swedish multinational pharmaceutical and biotechnology company AstraZeneca.[6][7] It is co-packaged and given as two separate consecutive intramuscular injections (one injection per monoclonal antibody, given in immediate succession).[2]
/////////////////////////////////////////////////////////////////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
Development
In 2020, researchers at Vanderbilt University Medical Center discovered particularly potent monoclonal antibodies, isolated from COVID-19 patients infected with a SARS-CoV-2 circulating at that time. Initially designated COV2-2196 and COV2-2130, antibody engineering was used to transfer their SARS-CoV-2 binding specificity to IgG scaffolds that would last longer in the body, and these engineered antibodies were named AZD8895 and AZD1061, respectively (and the combination was called AZD7442).[8]
To evaluate the antibodies’ potential as monoclonal antibody based prophylaxis (prevention), the ‘Provent’ clinical trial enrolled 5,000 high risk but not yet infected individuals and monitored them for 15 months.[9][10] The trial reported that those receiving the cocktail showed a 77% reduction in symptomatic COVID-19 and that there were no severe cases or deaths. AstraZeneca also found that the antibody cocktail “neutralizes recent emergent SARS-CoV-2 viral variants, including the Delta variant“.[7]
In contrast to pre-exposure prophylaxis, the Storm Chaser study of already-exposed people (post-exposure prophylaxis) did not meet its primary endpoint, which was prevention of symptomatic COVID-19 in people already exposed. AZD7442 was administered to 1,000 volunteers who had recently been exposed to COVID.[9]
Regulatory review
In October 2021, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) started a rolling review of tixagevimab/cilgavimab, which is being developed by AstraZeneca AB, for the prevention of COVID-19 in adults.[11]
Also in October 2021, AstraZeneca requested Emergency Use Authorization for tixagevimab/cilgavimab to prevent COVID-19 from the U.S. Food and Drug Administration (FDA).[12][13]
Emergency use authorization
On 14 November 2021, Bahrain granted emergency use authorization.[14]
On 8 December 2021, the U.S. Food and Drug Administration (FDA) granted emergency use authorization of this combination to prevent COVID-19 (before exposure) in people with weakened immunity or who cannot be fully vaccinated due to a history of severe reaction to coronavirus vaccines.[15] The FDA issued an emergency use authorization (EUA) for AstraZeneca’s Evusheld (tixagevimab co-packaged with cilgavimab and administered together) for the pre-exposure prophylaxis (prevention) of COVID-19 in certain people aged 12 years of age and older weighing at least 40 kilograms (88 lb).[2] The product is only authorized for those individuals who are not currently infected with the SARS-CoV-2 virus and who have not recently been exposed to an individual infected with SARS-CoV-2.[2]
References
- ^ “Evusheld- azd7442 kit”. DailyMed. Retrieved 4 January 2022.
- ^ Jump up to:a b c d “Coronavirus (COVID-19) Update: FDA Authorizes New Long-Acting Monoclonal Antibodies for Pre-exposure Prevention of COVID-19 in Certain Individuals”. U.S. Food and Drug Administration (FDA) (Press release). 8 December 2021. Retrieved 9 December 2021.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ O’Shaughnessy, Jacqueline A. (20 December 2021). “Re: Emergency Use Authorization 104” (PDF). Food and Drug Administration. Letter to AstraZeneca Pharmaceuticals LP | Attention: Stacey Cromer Berman, PhD. Archived from the original on 29 December 2021. Retrieved 18 January 2022.
- ^ “IUPHAR/BPS Guide to PHARMACOLOGY”. IUPHAR. 27 December 2021. Retrieved 27 December 2021.
- ^ “IUPHAR/BPS Guide to PHARMACOLOGY”. IUPHAR. 27 December 2021. Retrieved 27 December 2021.
- ^ Ray, Siladitya (21 August 2021). “AstraZeneca’s Covid-19 Antibody Therapy Effective In Preventing Symptoms Among High-Risk Groups, Trial Finds”. Forbes. ISSN 0015-6914. Archived from the original on 21 August 2021. Retrieved 18 January 2022.
- ^ Jump up to:a b Goriainoff, Anthony O. (20 August 2021). “AstraZeneca Says AZD7442 Antibody Phase 3 Trial Met Primary Endpoint in Preventing Covid-19”. MarketWatch. Archived from the original on 21 August 2021. Retrieved 18 January 2022.
- ^ Dong J, Zost SJ, Greaney AJ, Starr TN, Dingens AS, Chen EC, et al. (October 2021). “Genetic and structural basis for SARS-CoV-2 variant neutralization by a two-antibody cocktail”. Nature Microbiology. 6 (10): 1233–1244. doi:10.1038/s41564-021-00972-2. ISSN 2058-5276. PMC 8543371. PMID 34548634.
- ^ Jump up to:a b Haridy, Rich (23 August 2021). “”Game-changing” antibody cocktail prevents COVID-19 in the chronically ill”. New Atlas. Retrieved 23 August 2021.
- ^ “AZD7442 PROVENT Phase III prophylaxis trial met primary endpoint in preventing COVID-19”. AstraZeneca (Press release). 20 August 2021. Retrieved 15 October 2021.
- ^ “EMA starts rolling review of Evusheld (tixagevimab and cilgavimab)”. European Medicines Agency. 14 October 2021. Retrieved 15 October 2021.
- ^ “AZD7442 request for Emergency Use Authorization for COVID-19 prophylaxis filed in US”. AstraZeneca US (Press release). 5 October 2021. Retrieved 15 October 2021.
- ^ “AZD7442 request for Emergency Use Authorization for COVID-19 prophylaxis filed in US”. AstraZeneca (Press release). 5 October 2021. Retrieved 15 October 2021.
- ^ Abd-Alaziz, Moaz; Elhamy, Ahmad (14 November 2021). Macfie, Nick (ed.). “Bahrain authorizes AstraZeneca’s anti-COVID drug for emergency use”. Reuters. Archived from the original on 23 November 2021. Retrieved 18 January 2022.
- ^ Mishra, Manas; Satija, Bhanvi (8 December 2021). Dasgupta, Shounak (ed.). “U.S. FDA authorizes use of AstraZeneca COVID-19 antibody cocktail”. Reuters. Archived from the original on 13 January 2022. Retrieved 18 January 2022.
External links
“Tixagevimab”. Drug Information Portal. U.S. National Library of Medicine.
- “Cilgavimab”. Drug Information Portal. U.S. National Library of Medicine.
- Clinical trial number NCT04625972 for “Phase III Double-blind, Placebo-controlled Study of AZD7442 for Post-exposure Prophylaxis of COVID-19 in Adults (STORM CHASER)” at ClinicalTrials.gov
- Clinical trial number NCT04625725 for “Phase III Double-blind, Placebo-controlled Study of AZD7442 for Pre-exposure Prophylaxis of COVID-19 in Adult. (PROVENT)” at ClinicalTrials.gov
| Tixagevimab (teal, right) and cilgavimab (purple, left) binding the spike protein RBD. From PDB: 7L7E. | |
| Combination of | |
|---|---|
| Tixagevimab | Monoclonal antibody |
| Cilgavimab | Monoclonal antibody |
| Clinical data | |
| Trade names | Evusheld |
| Other names | AZD7442 |
| License data | US DailyMed: Tixagevimab |
| Routes of administration | Intramuscular |
| ATC code | J06BD03 (WHO) |
| Legal status | |
| Legal status | US: ℞-only via emergency use authorization[1][2][3] |
| Identifiers | |
| KEGG | D12262 |
| Clinical data | |
|---|---|
| Drug class | Antiviral |
| ATC code | None |
| Identifiers | |
| CAS Number | 2420564-02-7 |
| DrugBank | DB16394 |
| UNII | F0LZ415Z3B |
| KEGG | D11993 |
| Chemical and physical data | |
| Formula | C6488H10034N1746O2038S50 |
| Molar mass | 146706.82 g·mol−1 |
| Clinical data | |
|---|---|
| Drug class | Antiviral |
| ATC code | None |
| Identifiers | |
| CAS Number | 2420563-99-9 |
| DrugBank | DB16393 |
| UNII | 1KUR4BN70F |
| KEGG | D11994 |
| Chemical and physical data | |
| Formula | C6626H10218N1750O2078S44 |
| Molar mass | 149053.44 g·mol−1 |
/////////////////Tixagevimab, ANTI VIRAL, CORONA VIRUS, PEPTIDE, Monoclonal antibody, SARS-CoV-2 , WHO 11776, 2196, AZD-8895, AZD 8895, COV2-2196, COVID 19
NEW DRUG APPROVALS
ONE TIME
$10.00
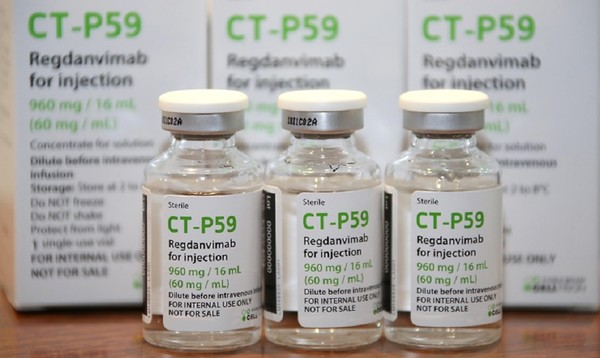
Regdanvimab

| (Heavy chain) QITLKESGPT LVKPTQTLTL TCSFSGFSLS TSGVGVGWIR QPPGKALEWL ALIDWDDNKY HTTSLKTRLT ISKDTSKNQV VLTMTNMDPV DTATYYCARI PGFLRYRNRY YYYGMDVWGQ GTTVTVSSAS TKGPSVFPLA PSSKSTSGGT AALGCLVKDY FPEPVTVSWN SGALTSGVHT FPAVLQSSGL YSLSSVVTVP SSSLGTQTYI CNVNHKPSNT KVDKRVEPKS CDKTHTCPPC PAPELLGGPS VFLFPPKPKD TLMISRTPEV TCVVVDVSHE DPEVKFNWYV DGVEVHNAKT KPREEQYNST YRVVSVLTVL HQDWLNGKEY KCKVSNKALP APIEKTISKA KGQPREPQVY TLPPSRDELT KNQVSLTCLV KGFYPSDIAV EWESNGQPEN NYKTTPPVLD SDGSFFLYSK LTVDKSRWQQ GNVFSCSVMH EALHNHYTQK SLSLSPGK (Light chain) ELVLTQPPSV SAAPGQKVTI SCSGSSSNIG NNYVSWYQQL PGTAPKLLIY DNNKRPSGIP DRFSGSKSGT SATLGITGLQ TGDEADYYCG TWDSSLSAGV FGGGTELTVL GQPKAAPSVT LFPPSSEELQ ANKATLVCLI SDFYPGAVTV AWKADGSPVK AGVETTKPSK QSNNKYAASS YLSLTPEQWK SHRSYSCQVT HEGSTVEKTV APTECS (Disulfide bridge: H22-H97, H155-H211, H231-L215, H237-H’237, H240-H’240, H272-H332, H378-H436, H’22-H’97, H’155-H’211, H’231-L’215, H’272-H’332, H’378-H’436, L22-L89, L138-L197, L’22-L’89, L’138-L’197) |
>Regdanvimab light chain: ELVLTQPPSVSAAPGQKVTISCSGSSSNIGNNYVSWYQQLPGTAPKLLIYDNNKRPSGIP DRFSGSKSGTSATLGITGLQTGDEADYYCGTWDSSLSAGVFGGGTELTVLGQPKAAPSVT LFPPSSEELQANKATLVCLISDFYPGAVTVAWKADGSPVKAGVETTKPSKQSNNKYAASS YLSLTPEQWKSHRSYSCQVTHEGSTVEKTVAPTECS
>Regdanvimab heavy chain: QITLKESGPTLVKPTQTLTLTCSFSGFSLSTSGVGVGWIRQPPGKALEWLALIDWDDNKY HTTSLKTRLTISKDTSKNQVVLTMTNMDPVDTATYYCARIPGFLRYRNRYYYYGMDVWGQ GTTVTVSSASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHT FPAVLQSSGLYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKRVEPKSCDKTHTCPPC PAPELLGGPSVFLFPPKPKDTLMISRTPEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKT KPREEQYNSTYRVVSVLTVLHQDWLNGKEYKCKVSNKALPAPIEKTISKAKGQPREPQVY TLPPSRDELTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDGSFFLYSK LTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK
Regdanvimab
レグダンビマブ;
EMA APPROVED, 2021/11/12, Regkirona
Treatment of adults with coronavirus disease 2019 (COVID-19)
MONOCLONAL ANTIBODY, ANTI VIRAL, PEPTIDE
CAS: 2444308-95-4, CT-P59
Regdanvimab, sold under the brand name Regkirona, is a human monoclonal antibody used for the treatment of COVID-19.[1] The antibody is directed against the spike protein of SARS-CoV-2. It is developed by Celltrion.[2][3] The medicine is given by infusion (drip) into a vein.[1][4]
The most common side effects include infusion-related reactions, including allergic reactions and anaphylaxis.[1]
Regdanvimab was approved for medical use in the European Union in November 2021.[1]
Regdanvimab is a monoclonal antibody targeted against the SARS-CoV-2 spike protein used to treat patients with COVID-19 who are at risk of progressing to severe COVID-19.
Regdanvimab (CT-P59) is a recombinant human IgG1 monoclonal antibody directed at the receptor binding domain (RBD) of the SARS-CoV-2 spike protein.4 It blocks the interaction between viral spike proteins and angiotensin-converting enzyme 2 (ACE2) that allows for viral entry into the cell, thereby inhibiting the virus’ ability to replicate. Trials investigating the use of regdanvimab as a therapeutic candidate for the treatment of COVID-19 began in mid-2020.1,3 It received its first full approval in South Korea in September 2021,3 followed by the EU in November 2021.5

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////////////////////////////
Synthesis Reference
Kim C, Ryu DK, Lee J, Kim YI, Seo JM, Kim YG, Jeong JH, Kim M, Kim JI, Kim P, Bae JS, Shim EY, Lee MS, Kim MS, Noh H, Park GS, Park JS, Son D, An Y, Lee JN, Kwon KS, Lee JY, Lee H, Yang JS, Kim KC, Kim SS, Woo HM, Kim JW, Park MS, Yu KM, Kim SM, Kim EH, Park SJ, Jeong ST, Yu CH, Song Y, Gu SH, Oh H, Koo BS, Hong JJ, Ryu CM, Park WB, Oh MD, Choi YK, Lee SY: A therapeutic neutralizing antibody targeting receptor binding domain of SARS-CoV-2 spike protein. Nat Commun. 2021 Jan 12;12(1):288. doi: 10.1038/s41467-020-20602-5.
Celltrion’s Monoclonal Antibody Treatment regdanvimab, Approved by the European Commission for the Treatment of COVID-19
- The European Commission (EC) granted marketing authorisation for Celltrion’s regdanvimab following positive opinion by the European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP) last week (11/11/2021)
- Celltrion continues to discuss supply agreements with regulatory agencies and contractors in more than 30 countries in Europe, Asia and LATAM to accelerate global access to regdanvimab
- The use of regdanvimab across the Republic of Korea is rapidly increasing to address the ongoing outbreaks
November 14, 2021 08:04 PM Eastern Standard Time
INCHEON, South Korea–(BUSINESS WIRE)–Celltrion Group announced today that the European Commission (EC) has approved Regkirona (regdanvimab, CT-P59), one of the first monoclonal antibody treatments granted marketing authorisation from the European Medicines Agency (EMA). The EC granted marketing authorisation for adults with COVID-19 who do not require supplemental oxygen and who are at increased risk of progressing to severe COVID-19. The decision from the EC follows a positive opinion by the European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP) on November 11th, 2021.1
“Today’s achievement, coupled with CHMP positive opinion for regdanvimab, underscores our ongoing commitment to addressing the world’s greatest health challenges,” said Dr. HoUng Kim, Ph.D., Head of Medical and Marketing Division at Celltrion Healthcare. “Typically, the recommendations from the CHMP are passed on to the EC for rapid legally binding decisions within a month or two, however, given the unprecedented times, we have received the EC approval within a day. As part of our global efforts to accelerate access, we have been communicating with the governments and contractors in 30 countries in Europe, Asia and LATAM. We will continue working with all key stakeholders to ensure COVID-19 patients around the world have access to safe and effective treatments.”
Monoclonal antibodies are proteins designed to attach to a specific target, in this case the spike protein of SARS-CoV-2, which works to block the path the virus uses to enter human cells. The EC approval is based on the global Phase III clinical trial involving more than 1,315 people to evaluate the efficacy and safety of regdanvimab in 13 countries including the U.S., Spain, and Romania. Data showed regdanvimab significantly reduced the risk of COVID-19 related hospitalisation or death by 72% for patients at high-risk of progressing to severe COVID-19.
Emergency use authorisations are currently in place in Indonesia and Brazil, and the monoclonal antibody treatment is fully approved in the Republic of Korea. In the U.S., regdanvimab has not yet been approved by the Food and Drug Administration (FDA), but the company is in discussion with the FDA to submit applications for an Emergency Use Authorisation (EUA).
As of November 12th, 2021, more than 22,587 people have been treated with regdanvimab in 129 hospitals in the Republic of Korea.
Notes to Editors:
About Celltrion Healthcare
Celltrion Healthcare is committed to delivering innovative and affordable medications to promote patients’ access to advanced therapies. Its products are manufactured at state-of-the-art mammalian cell culture facilities, designed and built to comply with the US FDA cGMP and the EU GMP guidelines. Celltrion Healthcare endeavours to offer high-quality cost-effective solutions through an extensive global network that spans more than 110 different countries. For more information please visit: https://www.celltrionhealthcare.com/en-us.
About regdanvimab (CT-P59)
CT-P59 was identified as a potential treatment for COVID-19 through screening of antibody candidates and selecting those that showed the highest potency in neutralising the SARS-CoV-2 virus. In vitro and in vivo pre- clinical studies showed that CT-P59 strongly binds to SARS-CoV-2 RBD and significantly neutralise the wild type and mutant variants of concern. In in vivo models, CT-P59 effectively reduced the viral load of SARS-CoV-2 and inflammation in lung. Results from the global Phase I and Phase II/III clinical trials of CT-P59 demonstrated a promising safety, tolerability, antiviral effect and efficacy profile in patients with mild-to-moderate symptoms of COVID-19.2 Celltrion also has recently commenced the development of a neutralising antibody cocktail with CT-P59 against new emerging variants of SARS-CoV-2.
Medical uses
In the European Union, regdanvimab is indicated for the treatment of adults with COVID-19 who do not require supplemental oxygen and who are at increased risk of progressing to severe COVID-19.[1]
Society and culture
Names
Regdanvimab is the proposed international nonproprietary name (pINN).[5]
Legal status
In March 2021, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) started a rolling review of data on regdanvimab.[6][7] In October 2021, the EMA started evaluating an application for marketing authorization for the monoclonal antibody regdanvimab (Regkirona) to treat adults with COVID-19 who do not require supplemental oxygen therapy and who are at increased risk of progressing to severe COVID 19.[8] The applicant is Celltrion Healthcare Hungary Kft.[8] The European Medicines Agency (EMA) concluded that regdanvimab can be used for the treatment of confirmed COVID-19 in adults who do not require supplemental oxygen therapy and who are at high risk of progressing to severe COVID-19.[4]
In November 2021, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) recommended granting a marketing authorization in the European Union for regdanvimab (Regkirona) for the treatment of COVID-19.[9][10] The company that applied for authorization of Regkirona is Celltrion Healthcare Hungary Kft.[10] Regdanvimab was approved for medical use in the European Union in November 2021.[1]
| Monoclonal antibody | |
|---|---|
| Type | Whole antibody |
| Source | Human |
| Target | Spike protein of SARS-CoV-2 |
| Clinical data | |
| Trade names | Regkirona |
| Other names | CT-P59 |
| License data | EU EMA: by INN |
| Routes of administration | Intravenous infusion |
| ATC code | None |
| Legal status | |
| Legal status | EU: Rx-only [1] |
| Identifiers | |
| CAS Number | 2444308-95-4 |
| DrugBank | DB16405 |
| UNII | I0BGE6P6I6 |
| KEGG | D12241 |
- Tuccori M, Ferraro S, Convertino I, Cappello E, Valdiserra G, Blandizzi C, Maggi F, Focosi D: Anti-SARS-CoV-2 neutralizing monoclonal antibodies: clinical pipeline. MAbs. 2020 Jan-Dec;12(1):1854149. doi: 10.1080/19420862.2020.1854149. [Article]
- Kim C, Ryu DK, Lee J, Kim YI, Seo JM, Kim YG, Jeong JH, Kim M, Kim JI, Kim P, Bae JS, Shim EY, Lee MS, Kim MS, Noh H, Park GS, Park JS, Son D, An Y, Lee JN, Kwon KS, Lee JY, Lee H, Yang JS, Kim KC, Kim SS, Woo HM, Kim JW, Park MS, Yu KM, Kim SM, Kim EH, Park SJ, Jeong ST, Yu CH, Song Y, Gu SH, Oh H, Koo BS, Hong JJ, Ryu CM, Park WB, Oh MD, Choi YK, Lee SY: A therapeutic neutralizing antibody targeting receptor binding domain of SARS-CoV-2 spike protein. Nat Commun. 2021 Jan 12;12(1):288. doi: 10.1038/s41467-020-20602-5. [Article]
- Syed YY: Regdanvimab: First Approval. Drugs. 2021 Nov 1. pii: 10.1007/s40265-021-01626-7. doi: 10.1007/s40265-021-01626-7. [Article]
- EMA Summary of Product Characteristics: Regkirona (regdanvimab) concentrate for solution for intravenous infusion [Link]
- EMA COVID-19 News: EMA recommends authorisation of two monoclonal antibody medicines [Link]
- EMA CHMP Assessment Report: Celltrion use of regdanvimab for the treatment of COVID-19 [Link]
- Protein Data Bank: Crystal Structure of COVID-19 virus spike receptor-binding domain complexed with a neutralizing antibody CT-P59 [Link]
References
- ^ Jump up to:a b c d e f g “Regkirona EPAR”. European Medicines Agency. Retrieved 12 November 2021. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ “Celltrion Develops Tailored Neutralising Antibody Cocktail Treatment with CT-P59 to Tackle COVID-19 Variant Spread Using Its Antibody Development Platform” (Press release). Celltrion. 11 February 2021. Retrieved 4 March 2021 – via Business Wire.
- ^ “Celltrion Group announces positive top-line efficacy and safety data from global Phase II/III clinical trial of COVID-19 treatment candidate CT-P59” (Press release). Celltrion. 13 January 2021. Retrieved 4 March 2021 – via Business Wire.
- ^ Jump up to:a b “EMA issues advice on use of regdanvimab for treating COVID-19”. European Medicines Agency. 26 March 2021. Retrieved 15 October 2021.
- ^ World Health Organization (2020). “International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 124 – COVID-19 (special edition)” (PDF). WHO Drug Information. 34 (3): 660–1.
- ^ “EMA starts rolling review of Celltrion antibody regdanvimab for COVID-19” (Press release). European Medicines Agency (EMA). 24 February 2021. Retrieved 4 March 2021.
- ^ “EMA review of regdanvimab for COVID-19 to support national decisions on early use” (Press release). European Medicines Agency (EMA). 2 March 2021. Retrieved 4 March 2021.
- ^ Jump up to:a b “EMA receives application for marketing authorisation Regkirona (regdanvimab) treating patients with COVID-19”. European Medicines Agency. 4 October 2021. Retrieved 15 October 2021.
- ^ “Regkirona: Pending EC decision”. European Medicines Agency. 11 November 2021. Retrieved 11 November 2021.
- ^ Jump up to:a b “COVID-19: EMA recommends authorisation of two monoclonal antibody medicines”. European Medicines Agency (EMA) (Press release). 11 November 2021. Retrieved 11 November 2021.
Further reading
- Kim C, Ryu DK, Lee J, Kim YI, Seo JM, Kim YG, et al. (January 2021). “A therapeutic neutralizing antibody targeting receptor binding domain of SARS-CoV-2 spike protein”. Nature Communications. 12 (1): 288. doi:10.1038/s41467-020-20602-5. PMC 7803729. PMID 33436577.
External links
- “Regdanvimab”. Drug Information Portal. U.S. National Library of Medicine.
///////////Regdanvimab, Regkirona, MONOCLONAL ANTIBODY, ANTI VIRAL, EU 2021, APPROVALS 2021, EMA 2021, COVID 19, CORONAVIRUS, PEPTIDE, レグダンビマブ , CT-P59, CT P59

NEW DRUG APPROVALS
ONE TIME
$10.00
Ropeginterferon alfa-2b
PCDLPQTHSL GSRRTLMLLA QMRRISLFSC LKDRHDFGFP QEEFGNQFQK AETIPVLHEM
IQQIFNLFST KDSSAAWDET LLDKFYTELY QQLNDLEACV IQGVGVTETP LMKEDSILAV
RKYFQRITLY LKEKKYSPCA WEVVRAEIMR SFSLSTNLQE SLRSKE
(Disulfide bridge: 2-99, 30-139)
Ropeginterferon alfa-2b
- AOP2014
CAS 1335098-50-4
UNII981TME683S
FDA APPROVED, 2021/11/12, BESREMI
PEPTIDE, Antineoplastic, Antiviral
Polycythemia vera (PV) is the most common Philadelphia chromosome-negative myeloproliferative neoplasm (MPN), characterized by increased hematocrit and platelet/leukocyte counts, an increased risk for hemorrhage and thromboembolic events, and a long-term propensity for myelofibrosis and leukemia.1,2 Interferon alfa-2b has been used for decades to treat PV but requires frequent dosing and is not tolerated by all patients.2 Ropeginterferon alfa-2b is a next-generation mono-pegylated type I interferon produced from proline-IFN-α-2b in Escherichia coli that has high tolerability and a long half-life.4,6 Ropeginterferon alfa-2b has shown efficacy in PV in in vitro and in vivo models and clinical trials.3,4
Ropeginterferon alfa-2b was approved by the FDA on November 12, 2021, and is currently marketed under the trademark BESREMi by PharmaEssentia Corporation.6
Ropeginterferon alfa-2b, sold under the brand name Besremi, is a medication used to treat polycythemia vera.[1][2][3][4] It is an interferon.[1][3] It is given by injection.[1][3]
The most common side effects include low levels of white blood cells and platelets (blood components that help the blood to clot), muscle and joint pain, tiredness, flu-like symptoms and increased blood levels of gamma-glutamyl transferase (a sign of liver problems).[3] Ropeginterferon alfa-2b can cause liver enzyme elevations, low levels of white blood cells, low levels of platelets, joint pain, fatigue, itching, upper airway infection, muscle pain and flu-like illness.[2] Side effects may also include urinary tract infection, depression and transient ischemic attacks (stroke-like attacks).[2]
It was approved for medical use in the European Union in February 2019,[3] and in the United States in November 2021.[2][5] Ropeginterferon alfa-2b is the first medication approved by the U.S. Food and Drug Administration (FDA) to treat polycythemia vera that people can take regardless of their treatment history, and the first interferon therapy specifically approved for polycythemia vera.[2]
https://www.fda.gov/news-events/press-announcements/fda-approves-treatment-rare-blood-disease#:~:text=FDA%20NEWS%20RELEASE-,FDA%20Approves%20Treatment%20for%20Rare%20Blood%20Disease,FDA%2DApproved%20Option%20Patients%20Can%20Take%20Regardless%20of%20Previous%20Therapies,-ShareFor Immediate Release:November 12, 2021
Today, the U.S. Food and Drug Administration approved Besremi (ropeginterferon alfa-2b-njft) injection to treat adults with polycythemia vera, a blood disease that causes the overproduction of red blood cells. The excess cells thicken the blood, slowing blood flow and increasing the chance of blood clots.
“Over 7,000 rare diseases affect more than 30 million people in the United States. Polycythemia vera affects approximately 6,200 Americans each year,” said Ann Farrell, M.D., director of the Division of Non-Malignant Hematology in the FDA’s Center for Drug Evaluation and Research. “This action highlights the FDA’s commitment to helping make new treatments available to patients with rare diseases.”
Besremi is the first FDA-approved medication for polycythemia vera that patients can take regardless of their treatment history, and the first interferon therapy specifically approved for polycythemia vera.
Treatment for polycythemia vera includes phlebotomies (a procedure that removes excess blood cells though a needle in a vein) as well as medicines to reduce the number of blood cells; Besremi is one of these medicines. Besremi is believed to work by attaching to certain receptors in the body, setting off a chain reaction that makes the bone marrow reduce blood cell production. Besremi is a long-acting drug that patients take by injection under the skin once every two weeks. If Besremi can reduce excess blood cells and maintain normal levels for at least one year, then dosing frequency may be reduced to once every four weeks.
The effectiveness and safety of Besremi were evaluated in a multicenter, single-arm trial that lasted 7.5 years. In this trial, 51 adults with polycythemia vera received Besremi for an average of about five years. Besremi’s effectiveness was assessed by looking at how many patients achieved complete hematological response, which meant that patients had a red blood cell volume of less than 45% without a recent phlebotomy, normal white cell counts and platelet counts, a normal spleen size, and no blood clots. Overall, 61% of patients had a complete hematological response.
Besremi can cause liver enzyme elevations, low levels of white blood cells, low levels of platelets, joint pain, fatigue, itching, upper airway infection, muscle pain and flu-like illness. Side effects may also include urinary tract infection, depression and transient ischemic attacks (stroke-like attacks).
Interferon alfa products like Besremi may cause or worsen neuropsychiatric, autoimmune, ischemic (not enough blood flow to a part of the body) and infectious diseases, which could lead to life-threatening or fatal complications. Patients who must not take Besremi include those who are allergic to the drug, those with a severe psychiatric disorder or a history of a severe psychiatric disorder, immunosuppressed transplant recipients, certain patients with autoimmune disease or a history of autoimmune disease, and patients with liver disease.
People who could be pregnant should be tested for pregnancy before using Besremi due to the risk of fetal harm.
Besremi received orphan drug designation for this indication. Orphan drug designation provides incentives to assist and encourage drug development for rare diseases.
The FDA granted the approval of Besremi to PharmaEssentia Corporation.
Medical uses
In the European Union, ropeginterferon alfa-2b is indicated as monotherapy in adults for the treatment of polycythemia vera without symptomatic splenomegaly.[3] In the United States it is indicated for the treatment of polycythemia vera.[1][2][5]
History
The effectiveness and safety of ropeginterferon alfa-2b were evaluated in a multicenter, single-arm trial that lasted 7.5 years.[2] In this trial, 51 adults with polycythemia vera received ropeginterferon alfa-2b for an average of about five years.[2] The effectiveness of ropeginterferon alfa-2b was assessed by looking at how many participants achieved complete hematological response, which meant that participants had a red blood cell volume of less than 45% without a recent phlebotomy, normal white cell counts and platelet counts, a normal spleen size, and no blood clots.[2] Overall, 61% of participants had a complete hematological response.[2] The U.S. Food and Drug Administration (FDA) granted the application for Ropeginterferon_alfa-2b orphan drug designation and granted the approval of Besremi to PharmaEssentia Corporation[2]
REF
- Bartalucci N, Guglielmelli P, Vannucchi AM: Polycythemia vera: the current status of preclinical models and therapeutic targets. Expert Opin Ther Targets. 2020 Jul;24(7):615-628. doi: 10.1080/14728222.2020.1762176. Epub 2020 May 18. [Article]
- How J, Hobbs G: Use of Interferon Alfa in the Treatment of Myeloproliferative Neoplasms: Perspectives and Review of the Literature. Cancers (Basel). 2020 Jul 18;12(7). pii: cancers12071954. doi: 10.3390/cancers12071954. [Article]
- Verger E, Soret-Dulphy J, Maslah N, Roy L, Rey J, Ghrieb Z, Kralovics R, Gisslinger H, Grohmann-Izay B, Klade C, Chomienne C, Giraudier S, Cassinat B, Kiladjian JJ: Ropeginterferon alpha-2b targets JAK2V617F-positive polycythemia vera cells in vitro and in vivo. Blood Cancer J. 2018 Oct 4;8(10):94. doi: 10.1038/s41408-018-0133-0. [Article]
- Gisslinger H, Zagrijtschuk O, Buxhofer-Ausch V, Thaler J, Schloegl E, Gastl GA, Wolf D, Kralovics R, Gisslinger B, Strecker K, Egle A, Melchardt T, Burgstaller S, Willenbacher E, Schalling M, Them NC, Kadlecova P, Klade C, Greil R: Ropeginterferon alfa-2b, a novel IFNalpha-2b, induces high response rates with low toxicity in patients with polycythemia vera. Blood. 2015 Oct 8;126(15):1762-9. doi: 10.1182/blood-2015-04-637280. Epub 2015 Aug 10. [Article]
- EMA Approved Products: Besremi (ropeginterferon alfa-2b ) solution for injection [Link]
- FDA Approved Drug Products: BESREMi (ropeginterferon alfa-2b-njft) injection [Link]

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////////////////////////////
References
- ^ Jump up to:a b c d e https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/761166s000lbl.pdf
- ^ Jump up to:a b c d e f g h i j k l “FDA Approves Treatment for Rare Blood Disease”. U.S. Food and Drug Administration (FDA) (Press release). 12 November 2021. Retrieved 12 November 2021.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Jump up to:a b c d e f g “Besremi EPAR”. European Medicines Agency (EMA). Retrieved 14 November 2021. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ Wagner SM, Melchardt T, Greil R (March 2020). “Ropeginterferon alfa-2b for the treatment of patients with polycythemia vera”. Drugs of Today. Barcelona, Spain. 56 (3): 195–202. doi:10.1358/dot.2020.56.3.3107706. PMID 32282866. S2CID 215758794.
- ^ Jump up to:a b “U.S. FDA Approves Besremi (ropeginterferon alfa-2b-njft) as the Only Interferon for Adults With Polycythemia Vera” (Press release). PharmaEssentia. 12 November 2021. Retrieved 14 November 2021 – via Business Wire.
External links
- “Ropeginterferon alfa-2b”. Drug Information Portal. U.S. National Library of Medicine.
- Clinical trial number NCT01193699 for “Safety Study of Pegylated Interferon Alpha 2b to Treat Polycythemia Vera (PEGINVERA)” at ClinicalTrials.gov
- Clinical trial number NCT02218047 for “AOP2014 vs. BAT in Patients With Polycythemia Vera Who Previously Participated in the PROUD-PV Study. (CONTI-PV)” at ClinicalTrials.gov
| Clinical data | |
|---|---|
| Trade names | Besremi |
| Other names | AOP2014, ropeginterferon alfa-2b-njft |
| License data | EU EMA: by INNUS DailyMed: Ropeginterferon_alfa |
| Pregnancy category | Contraindicated |
| Routes of administration | Subcutaneous |
| Drug class | Interferon |
| ATC code | L03AB15 (WHO) |
| Legal status | |
| Legal status | US: ℞-only [1][2]EU: Rx-only [3] |
| Identifiers | |
| CAS Number | 1335098-50-4 |
| DrugBank | DB15119 |
| UNII | 981TME683S |
| KEGG | D11027 |
/////////Ropeginterferon alfa-2b, FDA 2021, APPROVALS 2021, BESREMI, PEPTIDE, Antineoplastic, Antiviral, AOP 2014, PharmaEssentia

NEW DRUG APPROVALS
ONE TIME
$10.00
RITONAVIR
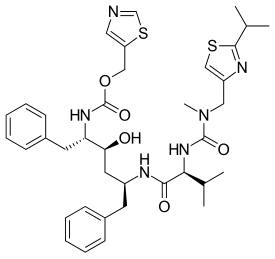
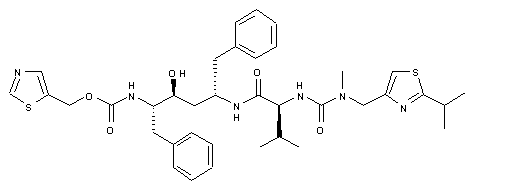
RITONAVIR
- Molecular FormulaC37H48N6O5S2
- Average mass720.944 Da
1,3-Thiazol-5-ylmethyl-[(1S,2S,4S)-1-benzyl-2-hydroxy-4-({(2S)-3-methyl-2-[(methyl{[2-(1-methylethyl)-1,3-thiazol-4-yl]methyl}carbamoyl)amino]butanoyl}amino)-5-phenylpentyl]carbamat
155213-67-5[RN]
7449Abbott 84538
UNII-O3J8G9O825
ритонавир
ريتونافير
利托那韦
(1E,2S)-N-[(2S,4S,5S)-4-Hydroxy-5-{(E)-[hydroxy(1,3-thiazol-5-ylmethoxy)methylene]amino}-1,6-diphenyl-2-hexanyl]-2-[(E)-(hydroxy{[(2-isopropyl-1,3-thiazol-4-yl)methyl](methyl)amino}methylene)amino]-3- methylbutanimidic acid
(2S,3S,5S)-5-[N-[N-[[N-methyl-N-[(2-isopropyl-4-thiazolyl)methyl]amino]carbonyl]valinyl]amino]-2-[N-[(5-thiazolyl)methoxycarbonyl]amino]-1,6-diphenyl-3-hydroxyhexane
- A-84538
- Abbott 84538
- ABBOTT-84538
- ABT 538
- ABT-538
- DRG-0244
- NSC-693184
- TMC 114r
Ritonavir
CAS Registry Number: 155213-67-5
CAS Name: (5S,8S,10S,11S)-10-Hydroxy-2-methyl-5-(1-methylethyl)-1-[2-(1-methylethyl)-4-thiazolyl]-3,6-dioxo-8,11-bis(phenylmethyl)-2,4,7,12-tetraazatridecan-13-oic acid 5-thiazolylmethyl ester
Additional Names: (2S,3S,5S)-5-[N-[N-[[N-methyl-N-[(2-isopropyl-4-thiazolyl)methyl]amino]carbonyl]valinyl]amino]-2-[N-[(5-thiazolyl)methoxycarbonyl]amino]-1,6-diphenyl-3-hydroxyhexane
Manufacturers’ Codes: A-84538; Abbott 84538; ABT-538
Trademarks: Norvir (Abbott)
Molecular Formula: C37H48N6O5S2
Molecular Weight: 720.94
Percent Composition: C 61.64%, H 6.71%, N 11.66%, O 11.10%, S 8.90%
Literature References: Peptidomimetic HIV-1 protease inhibitor. Prepn: D. J. Kempf et al.,WO9414436; eidem,US5541206 (1994, 1996 both to Abbott). Antiretroviral spectrum, pharmacokinetics: idemet al.,Proc. Natl. Acad. Sci. USA92, 2484 (1995). Structural model for drug resistance: M. Markowitz et al.,J. Virol.69, 701 (1995). HPLC determn in biological fluids: R. M. W. Hoetelmans et al.,J. Chromatogr. B705, 119 (1998). Review of clinical experience: A. P. Lea, D. Faulds, Drugs52, 541-546 (1996). Clinical trial with nucleoside analogs in HIV-infected children: S. A. Nachman et al.,J. Am. Med. Assoc.283, 492 (2000). Clinical trial with lopinavir, q.v., in HIV infection: S. Walmsley et al.,N. Engl. J. Med.346, 2039 (2002).
Therap-Cat: Antiviral.
Keywords: Antiviral; Peptidomimetics; HIV Protease Inhibitor.Company:Abbvie (Originator)Sales:$389 Million (Y2012); 
$419 Million (Y2011);ATC Code:J05AE03
| Approval Date | Approval Type | Trade Name | Indication | Dosage Form | Strength | Company | Review Classification |
|---|---|---|---|---|---|---|---|
| 2010-02-10 | New dosage form | Norvir | HIV infection | Tablet | 100 mg | Abbvie | |
| 1999-06-29 | Additional approval | Norvir | HIV infection | Capsule | 100 mg | Abbvie | |
| 1996-03-01 | New dosage form | Norvir | HIV infection | Capsule | 100 mg | Abbvie | Priority |
| 1996-03-01 | First approval | Norvir | HIV infection | Solution | 80 mg/mL | Abbvie |
| Approval Date | Approval Type | Trade Name | Indication | Dosage Form | Strength | Company | Review Classification |
|---|---|---|---|---|---|---|---|
| 1996-08-26 | First approval | Norvir | HIV infection | Capsule | 100 mg | Abbvie | |
| 1996-08-26 | First approval | Norvir | HIV infection | Tablet, Film coated | 100 mg | Abbvie | |
| 1996-08-26 | First approval | Norvir | HIV infection | Solution | 80 mg/mL | Abbvie |
| Approval Date | Approval Type | Trade Name | Indication | Dosage Form | Strength | Company | Review Classification |
|---|---|---|---|---|---|---|---|
| 2011-02-28 | New dosage form | Norvir | HIV infection | Tablet | 100 mg | Abbvie | |
| 1998-09-25 | First approval | Norvir | HIV infection | Solution | 80 mg/mL | Abbvie |
| Approval Date | Approval Type | Trade Name | Indication | Dosage Form | Strength | Company | Review Classification |
|---|---|---|---|---|---|---|---|
| 2013-10-15 | Marketing approval | HIV infection | Tablet | 100 mg | Abbvie | ||
| 2010-07-29 | Marketing approval | 爱治威/Norvir | HIV infection | Capsule | 100 mg | Abbott | |
| 2010-07-13 | Marketing approval | 迈可欣 | HIV infection | Solution | 75 ml:6 g | 美吉斯制药(厦门) | 3.1类 |
Ritonavir was first approved by the U.S. Food and Drug Administration (FDA) on March 1, 1996, then approved by European Medicine Agency (EMA) on August 26, 1996, and approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on September 25, 1998. It was developed and marketed as Norvir® by Abbvie.
Ritonavir is an HIV protease inhibitor. It is indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection.
Norvir® is available as solution for oral use, containing 80 mg of free Ritonavir per mL. The recommended dose is 600 mg twice-day with meals for adult patients.
Ritonavir is an HIV protease inhibitor used in combination with other antivirals in the treatment of HIV infection.
Ritonavir (RTV), sold under the brand name Norvir, is an antiretroviral medication used along with other medications to treat HIV/AIDS.[2] This combination treatment is known as highly active antiretroviral therapy (HAART).[2] Often a low dose is used with other protease inhibitors.[2] It may also be used in combination with other medications for hepatitis C.[3] It is taken by mouth.[2] The capsules of the medication do not work the same as the tablets.[2]
Common side effects include nausea, vomiting, loss of appetite, diarrhea, and numbness of the hands and feet.[2] Serious side effects include liver problems, pancreatitis, allergic reactions, and arrythmias.[2] Serious interactions may occur with a number of other medications including amiodarone and simvastatin.[2] At low doses it is considered to be acceptable for use during pregnancy.[4] Ritonavir is of the protease inhibitor class.[2] Typically, however, it is used to inhibit the enzyme that metabolizes other protease inhibitors.[5] This inhibition allows lower doses of these latter medication to be used.[5]
Ritonavir was patented in 1989 and came into medical use in 1996.[6][7] It is on the World Health Organization’s List of Essential Medicines.[8] Ritonavir capsules were approved as a generic medication in the United States in 2020.[9]

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////////////////////////////
In the first step shown, an aldehyde derived from phenylalanine is treated with zinc dust in the presence of vanadium(III) chloride. This results in a pinacol coupling reaction which dimerises the material to provide an intermediate which is converted to its epoxide and then reduced to (2S,3S,5S)-2,5-diamino-1,6-diphenylhexan-3-ol. Importantly, this retains the absolute stereochemistry of the amino acid precursor. The diamine is then treated sequentially with two thiazole derivatives, each linked by an amide bond, to provide ritonavir.[23][24]
REF 23, 24 BELOW
- WO patent 1994014436, Kempf, Dale J.; Norbeck, Daniel W.; Sham, Hing Leung; Zhao, Chen; Sowin, Thomas J.; Reno, Daniel S.; Haight, Anthony R. and Cooper, Arthur J., “Retroviral protease inhibiting compounds”, published 1994-07-07, assigned to Abbott Laboratories
- ^ Vardanyan, Ruben; Hruby, Victor (2016). “34: Antiviral Drugs”. Synthesis of Best-Seller Drugs. pp. 698–701. doi:10.1016/B978-0-12-411492-0.00034-1. ISBN 9780124114920. S2CID 75449475.
SYN
EP 0674513; EP 0727419; JP 1996505844; JP 1997118679; JP 1998087639; WO 9414436 |
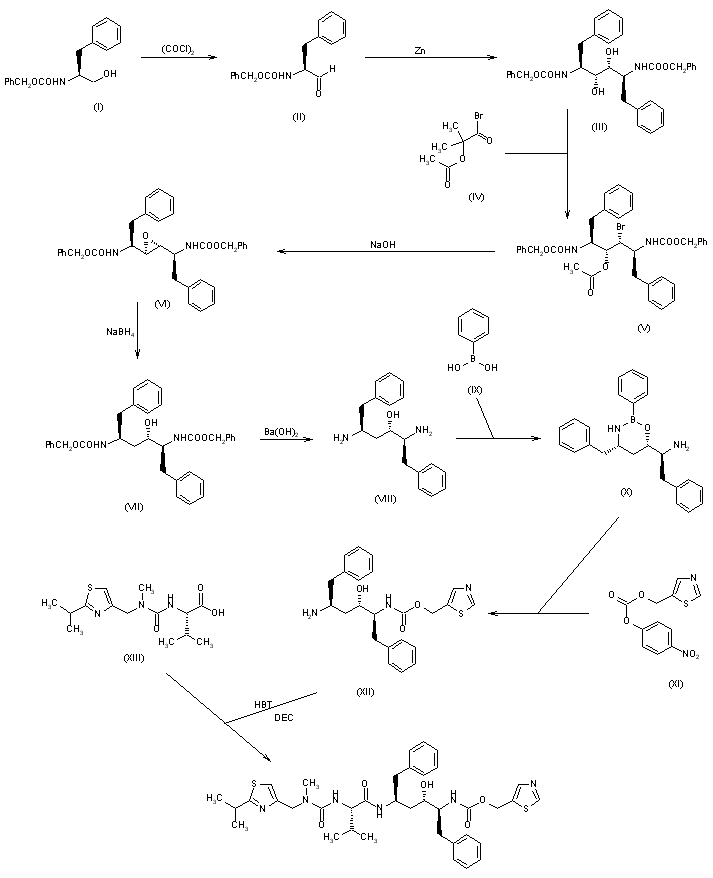
The oxidation of N-(benzyloxycarbonyl)-L-phenylalaninol (I) with oxalyl chloride in DMSO gives the corresponding aldehyde (II), which by dimerization with Zn dust in dichloromethane yields (2S,3R,4R,5S)-2,5-bis(benzyloxycarbonylamino)-1,6-diphenylhexane-3,4-diol (III) [along with the (2S,3S,4S,5S)-isomer]. The reaction of (III) with alpha-acetoxyisobutyryl bromide (IV) in hexane/dichloromethane affords (2S,3R,4R,5S)-2,5-bis(benzyloxycarbonylamino)-4-bromo-1,6-diphenylhexan-3-ol acetate ester (V), which is converted into the corresponding epoxide (VI) with NaOH in dioxane/water. The reduction of the epoxide (VI) with NaBH4 in THF affords (2S,3S,5S)-2,5-bis(benzyloxycarbonylamino)-1,6-diphenylhexan-3-ol (VII), which is deprotected with Ba(OH)2 in refluxing dioxane/water yielding the corresponding diamine (VIII). The cyclization of (VIII) with phenylboronic acid (IX) in refluxing toluene gives (4S,6S)-6-[1(S)-amino-2-phenylethyl]-4-benzyl-2-phenyl-1,3,2-oxaazaborinane (X), which is condensed with (5-thiazolylmethyl)(4-nitrophenyl)carbonate (XI) in THF to afford (2S,3S,5S)-5-amino-1,6-diphenyl-2-(5-thiazolylmethoxycarbonylamino) hexan-3-ol (XII). Finally, this compound is condensed with N-[N-(2-isopropylthiazol-4-ylmethyl)-N-methylaminocarbonyl]-L-valine (XIII) by means of 1-hydroxybenzotriazole (HBT) and N-[3-(dimethylamino)propyl]-N’-ethylcarbodiimide (DEC) in THF.
SYN
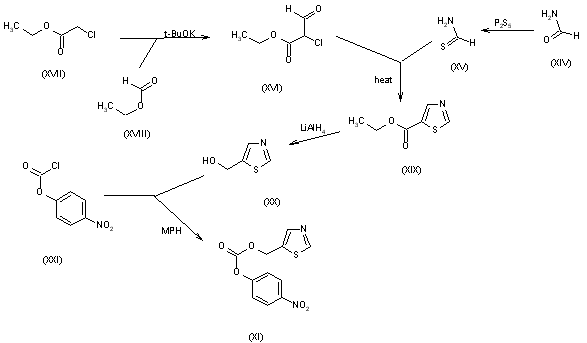
The thiazolyl carbonate (XI) has been synthesized as follows: The reaction of formamide (XIV) with P2S5 in ethyl ether gives thioformamide (XV), which is cyclized with 2-chloro-3-oxopropionic acid ethyl ester (XVI) [obtained by condensation of ethyl chloroacetate (XVII) with ethyl formate (XVIII) by means of t-BuOK in THF] yielding thiazol-5-carboxylic acid ethyl ester (XIX). The reduction of (XIX) with LiAlH4 in THF affords 5-thiazolylmethanol (XX), which is then esterified with 4-nitrophenyl chloroformate (XXI) by means of 4-methylmorpholine (MPH) in dichloromethane to give the desired product (XI).
SYN
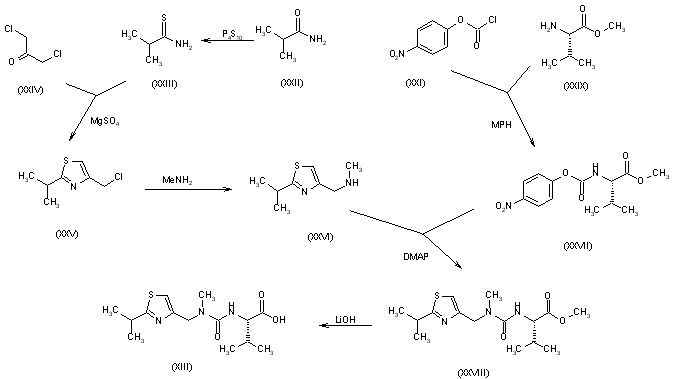
The N-substituted valine (XIII) has been synthesized as follows: The reaction of isobutyramide (XXII) with phosphorus pentasulfide (P4S10) in ethyl ether gives the corresponding thioamide (XXIII), which is cyclized with 1,3-dichloroacetone (XXIV) by means of MgSO4 in refluxing acetone yielding 4-(chloromethyl)-2-isopropylthiazole (XXV). The reaction of (XXV) with methylamine in water affords N-(2-isopropylthiazol-4-ylmethyl)-N-methylamine (XXVI), which is condensed with N-(4-nitrophenoxycarbonyl)-L-valine methyl ester (XXVII) by means of 4-(dimethylamino)pyridine (DMAP) and triethylamine in refluxing THF to give the thiazol-substituted L-valine ester (XXVIII). Finally, this compound is converted into the corresponding free acid with LiOH in dioxane/water. The N-(4-nitrophenoxycarbonyl)-L-valine methyl ester (XXVII) has been synthesized by reaction of chloroformate (XXI) with L-valine methyl ester (XXIX) by means of 4-methylmorpholine (MPH) in dichloromethane.
SYN
Org Process Res Dev 1999,3(2),94 |
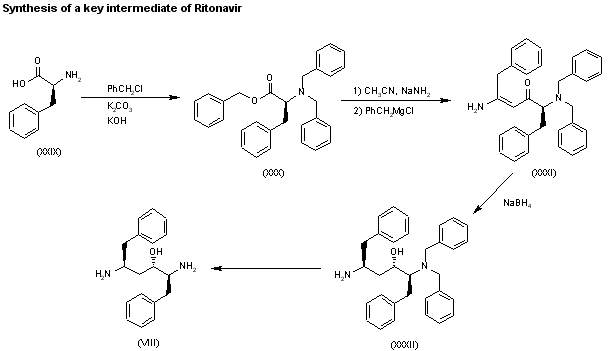
A new method for the preparation of a key diaminoalcohol intermediate in the synthesis of ritonavir has been described: The reaction of L-phenylalanine (XXIX) with benzyl chloride by means of K2CO3 and KOH in hot water gives N,N-dibenzyl-L-phenylalanine benzyl ester (XXX), which is condensed first with acetonitrile by means of NaNH2 and then with benzylmagnesium chloride, both reactions in methyl tert-butyl ether, yielding 5-amino-2-(dibenzylamino)-1,6-diphenylhex-4-en-3-one (XXXI). The reduction of (XXXI) with NaBH4 with an accurate control of the solvent and acidity, results in an enhanced enantioselectivity towards the desired (S,S,S)-enantiomer of 5-amino-2-(dibenzylamino)-1,6-diphenyl-3-hexanol (XXXII). Finally, this compound is deprotected by hydrogenation with ammonium formate over Pd/C in methanol/water to provide the target chiral diaminoalcohol (VIII).
SYN
Chem Commun (London) 2001,(2),203 |
he reaction of N-(tert-butoxycarbonyl)-L-phenylalanine methyl ester (I) with trimethylphosphonate lithium salt (II) in THF gives the dimethyl L-phenylalanylmethylphosphonate (III), which is condensed with 2-phenylacetaldehyde (IV) by means of Na2CO3 in ethanol yielding the diphenylhexenone (V). The reduction of the carbonyl group of (V) with NaBH4 in methanol affords a diastereomeric mixture of alcohols (VI) and (VII) from which the desired major isomer (VII) is separated by column chromatography.. The epoxidation of the double bond of (VII) with MCPBA in dichloromethane gives the epoxide (VIII) (1), which is cleaved with Red-Al in THF providing the diol (IX). The reaction of (IX) with Ms-Cl and DIEA yields the intermediate dimesylate (X) which, without isolation, affords oxazolidinone (XI). The reaction of (XI) with sodium azide and 18-C-6 in DMSO gives the azide (XII), which is treated with NaH and Boc2O in THF in order to cleave the oxazolidine ring and furnish the protected aminoalcohol (XIII). Finally the azido group of (XIII) is hydrogenate with H2 over Pd/C in methanol to afford the monoprotected diaminoalcohol (XIV) the target compound.
SYN
https://www.scielo.br/j/mioc/a/FT4WXwhbCPCkm7W6XdX4GHm/?lang=en
Ritonavir (2) – The first disclosure of ritonavir (2) is presented in a patent from 1994, assigned to Abbott Laboratories. Even though the patent consists of a range of Markush structures, among which is found (2), its synthesis is not presented.249 The synthetic pathway to obtain (2) was disclosed for the first time in a second-generation patent in 1995, by the same company.250,251 Some drawbacks related to the synthesis presented by Abbott include the employment of expensive condensing agents, as 1-Ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDC) and poor reaction yields on the first steps of the synthetic pathway, making this strategy too expensive and not suitable for scale-up production. Considering the above deficiencies, a recent Chinese patent concerning the synthesis of ritonavir (2) has been released.
The synthetic pathway described in the patent starts with a nucleophilic addition-elimination reaction between the starting material (2-isopropylthiazol-4-yl)- nitro-methylamine (71) and N-[(2,2,2-trichloroethoxy)carbonyl]-L-valine (72), generating intermediate (73). The intermediate is mixed with p-toluenesulfonyl chloride and triethylamine to activate the acid function, followed by the addition of reagent (74) in a one-pot procedure. The condensation allows the formation of intermediate (75), which is submitted to acidic conditions for a Boc deprotection, followed by another nucleophilic addition-elimination reaction with reagent (76), thus obtaining the final product ritonavir (2) (Fig. 16).252 When comparing both patents, it is possible to highlight important advantages present in the latest one, including the use of cheap and easily available reagents, for instance, the use of p-toluenesulfonyl chloride as an amide condensing agent, the synthetic pathway has a high yield (79% overall), low cost and it is ease to scale-up the production.

SYN
US5543551

Reference:1. US5541206A / EP0402646B1.
Reference:1. Org. Process Res. Dev. 1999, 3, 94-100.
https://pubs.acs.org/doi/abs/10.1021/op9802071
Patent
2. WO2006090264A1.
Reference:1. Tetrahedron Lett. 2011, 52, 6968-6970.
SYN
Synthesis ReferenceUS5484801
SYN
Method of synthesis
i. Valine condensed with bis-trichloromethyl carbonate to give an intermediate compound 4-isopropyloxazolidine-2,5-dione.
ii. The intermediate compound is reacted with compound (1) to give compound (2).
ii. (2) reacts with bis-trichloromethyl carbonate followed by reaction with (2-isopropylthiazol-4-yl)-N-methylmethanamine to give compound (3).
iv. Primary amine group of (3) is subjected to deprotection by the removal of two benzyl groups to give compound (4).
v. (4) reacts with (5) to give Ritonavir in good yield.
CLICK ON BELOW IMAGE
CLIP
SYN
US5543551

SYN
Ruben Vardanyan, Victor Hruby, in Synthesis of Best-Seller Drugs, 2016
Lopinavir–Kaletra
The first report of a new protease inhibitor candidate lopinavir (34.1.21) seems to be Abbott’s patent [134].
The core of lopinavir is identical to that of ritonavir. The 5-thiazolyl end group in ritonavir was replaced by the phenoxyacetyl group, and the 2-isopropylthiazolyl group in ritonavir was replaced by a modified valine in which the amino terminus had a six-membered cyclic urea attached.
Synthetic strategy employed for the synthesis of multikilogram quantities of lopinavir is very similar to that implemented for ritonavir (34.1.22), but using the protected version (34.1.79) of the “core” diamino alcohol (34.1.55), which was sequentially acylated with the acid chlorides of (S)-3-methyl-2-(2-oxotetrahydropyrimidin-1(2H)-yl)butanoic acid (34.1.89) and 2-(2,6-dimethylphenoxy)acetic acid (34.1.94) [135,136].
The bulk synthesis of protected diamino alcohol (34.1.79) was proposed [137,138] by a method closely related to the method [122,123] for ritonavir. For that purpose L-phenylalanine (34.1.74) was sequentially trialkylated with benzyl chloride using a K2CO3/water system at reflux to produce a tribenzylated product (34.1.75). A solution with generated acetonitrile anion in THF was added to the benzylated product (34.1.75) at less than −40°C to yield the cyanomethylketone (34.1.76), which was exposed to Grignard reagent–benzyl magnesium chloride to produce an enaminone (34.1.77). No racemization was observed in these two steps. The addition of the obtained enaminone (34.1.77) in THF/PrOH to a solution of NaBH4 and MsOH in THF at 5°C produced an intermediate aminoketone (34.1.78). No further reduction of the keto group occurs under these conditions. Reduction of the keto group could proceed by addition of a preformed solution of sodium tris(trifluoroacetoxy)borohydride in tetrahydrofuran [NaBH3(OTFA)], which produces a mixture of amino alcohols composed of 93% of the desired (2S)-5-amino-2-(dibenzylamino)-1,6-diphenylhexan-3-ol (34.1.79) along with 7% of the three undesired diastereomers. The crude mixture was debenzylated (Pd-C, HCONH4), and the product was purified by precipitation from iPrOH/HCl (aq) to produce (34.1.55) in greater than 99% purity and in high yield (Scheme 34.8.).

An efficient synthesis of each of the side chain moieties and their coupling with the “core” diamino alcohol derivatives was developed as follows: (S)-3-methyl-2-(2-oxotetrahydropyrimidin-1(2H)-yl)butanoic acid (34.1.85) was prepared starting from L-valine (34.1.80), which was first converted to N-phenoxycarbonyl-L-valine (34.1.82) with phenylchloroformate (34.1.81). Accurate pH monitoring (pH 9.5 to 10.2) was necessary and LiOH was found to be a superior base. Control of pH was essential as the valine dimer and its derivatives were formed as reaction byproducts outside of this pH margin. LiCl was added to provide a lower freezing point to the aqueous solution and neutral Al2O3 was added to prevent gumming and emulsion formation during the course of the reaction.
Treatment of N-phenoxycarbonyl-L-valine (34.1.82) with 3-chloropropylamine hydrochloride (34.1.3) and solid NaOH in THF produced the unisolated salt of chloropropylurea (34.1.84), which was then treated with t-BuOK, effecting cyclization to produce the desired acid (34.1.85) in 75 to 85% yield and in greater than 99% enantiomeric excess. Acylation of (34.1.79) with synthesized acid (34.1.85) was initially achieved by well-known peptide coupling methods. Optimization of this transformation allowed the discovery of a more cost-effective method for implementing acyl chloride (34.1.86), which was easily prepared using thionyl chloride in THF at room temperature.
The reaction of dibenzylamino alcohols (34.1.79) with acyl chloride (34.1.86) in the presence of 3.0 equivalents of imidazole in EtOAc and DMF produced acylated intermediate (34.1.87) as a mixture of diastereomers, which, without any further purification, was subjected to debenzylation with Pd/C and HCO2NH4 in MeOH at 50°C, which proceeded without significant complications to produce (34.1.88).
Exposure of crude (34.1.88) to L-pyroglutamic acid (34.1.89) in dioxane at 50°C followed by cooling, allowed for the isolation of (S)-N-((2S,4S,5S)-5-amino-4-hydroxy-1,6-diphenylhexan-2-yl)-3-methyl-2-(2-oxotetrahydropyrimidin-1(2H)-yl)butanamide (34.1.90) pyroglutamic salt as virtually a single diastereomer in high yield (Scheme 34.9.).

Acyl chloride (34.1.92) was prepared by the reaction of 2-(2,6-dimethylphenoxy)acetic acid (34.1.91) with thionyl chloride in EtOAc, at room temperature adding a single drop of DMF, and warming the slurry to 50°C, which produced a clear solution of (34.1.92) that was used in the subsequent acylation of amine (34.1.90). Reaction of pyroglutamate salt (34.1.90) with acyl chloride (34.1.95) in ethyl acetate under Schotten-Baumann reaction conditions (use of a two-phase solvent system) in the presence of a water solution of NaHCO3 for liberation of free amine, produced the desired lopinavir (34.1.21) in high yield and purity (Scheme 34.10.).

Lopinavir is a novel and strong protease inhibitor developed from ritonavir with high specificity for HIV-1 protease [139-141]. It is indicated, in combination with other antiretroviral agents, for the treatment of HIV-1 infection. Numerous clinical trials have shown that lopinavir/ritonavir (Kaletra) is highly effective as a component of highly active antiretroviral therapy [142,143].
Ritonavir is indicated in combination with other antiretroviral agents for the treatment of HIV-1-infected patients (adults and children of two years of age and older).[1][2]
PATENT
https://patents.google.com/patent/EP1133485B1/en
- Ritonavir (CAS Number [155213-67-5]), the structural formula of which is given below,is an inhibitor of human HIV protease which was described for the first time by Abbott in International Patent Application WO 94/14436, together with the process for the preparation thereof.
- [0002]
In the synthesis scheme given in WO 94/14436, Ritonavir is manufactured starting from valine and from compounds 1, 4 and 7, the structural formulae of which are also given below. - [0003]
The synthesis process in question was subsequently optimised in its various parts by Abbot, who then described and claimed the individual improvements in the patent documents listed below: US 5,354,866, US 5,541,206, US 5,491,253, WO98/54122, WO 98/00393 and WO 99/11636. - [0004]
The process for the synthesis of Ritonavir carried out on the basis of the above-mentioned patent documents requires, however, a particularly large number of intermediate stages; it is also unacceptable from the point of view of so-called “low environmental impact chemical synthesis” (B.M.Trost Angew. Chem. Int. Ed. Engl. (1995) 34, 259-281) owing to the increased use of activating groups and protective groups which necessitate not inconsiderable additional work in disposing of the by-products of the process, with a consequent increase in the overall manufacturing costs. - [0005]
The object of the present invention is therefore to find a process for the synthesis of Ritonavir which requires a smaller number of intermediate stages and satisfies the requirements of low environmental impact chemical synthesis, thus limiting “waste of material”. - [0006]
A process has now been found which, by using as starting materials the same compounds as those used in WO 94/14436, leads to the formation of Ritonavir in only five stages and with a minimum use of carbon atoms that are not incorporated in the final molecule.
EXAMPLESStage 1
- [0014]
ReagentMolecular weightAmountMmolEq.Amine 1464.6515 g32.2821Val-NCA 2142.136.9 g48.5471.5Triethylamine101.194.5 ml32.2831 (d = 0.726) Dichloromethane 108 ml Sol. 0.3 M - [0015]
Compound Val-NCA 2 was added to a solution of amine 1 (note 1, 2) in dichloromethane (60 ml) at -15°C under nitrogen followed by a solution of triethylamine in dichloromethane (48 ml) (note 3). The reaction mixture was maintained under agitation at from -15° to -13°C for two hours (note 4). The solution was used directly for the next stage without further purification.
- Note 1. Amine 1 was prepared using the procedure described in A.R. Haight et al. Org. Proc. Res. Develop. (1999) 3, 94-100.
- Note 2. Amine 1 was a mixture of stereoisomers with a ratio of 80 : 3.3 : 2.1 : 1.9.
- Note 3. This solution was added dropwise over a period of 25 minutes.
- Note 4. HPLC analysis after 2 hours: Amide 3 70.2 % – starting material 4.3 %
Stage 2
- [0016]
ReagentMolecular weightAmountMmolEq.Amine 3 Solution from stage 1563.78 32.2821Triethylamine101.19 (d = 0.726)2.6 ml18.6540.6Bis(trichloromethyl) carbonate (BTC)296.753.5 g11.7940.36dichloromethane 65 ml N-Methyl-4-aminomethyl-2-isopropylthiazole 4170.275.5 g32.2821triethylamine101.196.9 ml49.5041.5 (d = 0.726) Dichloromethane 52 ml - [0017]
The triethylamine was added slowly to the solution of amine 3 resulting from stage 1 and this mixture was in turn added slowly to a solution of BTC in dichloromethane (65 ml) at from -15° to -13°C and the reaction mixture was maintained under agitation at that temperature for 1.5 hours (note 1). A solution of amine 4 and triethylamine in dichloromethane (52 ml) was then added slowly to the reaction mixture (note 2) and maintained under agitation at that temperature for one hour (note 3). The reaction was stopped with water (97 ml) and the two phases were separated. The organic phase was washed with a 10% aqueous citric acid solution, filtered over Celite and evaporated with a yield of 25 g of crude urea 5 which was purified by flash chromatography on silica gel (eluant: toluene – ethyl acetate 6:4) to give 9.4 g of pure compound 5 (total yield of the two stages 38%) (note 4).
- Note 1. HPLC analysis after 1.5 hours of the reaction stopped in tert-butylamine: Amide 3 = 0.41 % – intermediate isocyanate = 61.3 %
- Note 2. The solution of amine 4 was added over a period of approximately 20 minutes.
- Note 3. HPLC analysis after 1 hour: Amide 3 = 7 % – Urea 5 = 54 %
- Note 4. 1H-NMR (CDCl3, 600 MHz) δ 7.31 (m, 4H), 7.28-7.24 (m, 4H), 7.21-7.14 (m, 8H), 7.11 (d, 2H), 7.05 (d, 2H), 6.96 (s, 1H), 6.75 (bd, 1H), 5.96 (bs, 1H), 4.48 (d, 1H), 4.39 ( d, 1H), 4.13 (dd, 1H), 4.09 (m, 1H), 3.93 (bd, 2H), 3.56 (bt, 1H), 3.39 (d, 2H), 3.28 (m, 1H), 3.04 (dd, 1H), 2.97 (s, 3H), 2.89 (m, 1H), 2.74 (q, 1H), 2.61 (m, 2H), 2.25 (m, 1H), 1.53 ( ddd, 1H), 1.38 (d, 6H), 1.28 (dt, 1H), 0.98 (d, 3H), 0.91 (d, 3H).
Stage 3
- [0018]
ReagentMolecular weightAmountMmolEq.Urea 5760.053.5 g4.6051Pd(OH)2/C 20% 5.25 g 30% w/wAcetic acid 31 ml Sol. 0.15 M - [0019]
The Pearlman catalyst (note 1) was added to a solution of urea 5 in acetic acid and the mixture was hydrogenated for 5 hours at from 4 to 4.5 bar and from 78 to 82°C (note 2). The catalyst was filtered and the solvent was removed under reduced pressure. The crude product was dissolved in water (35 ml), the pH was adjusted to a value of 8 with NaOH, and extracted with CH2Cl2 (2×15 ml). The organic phase was washed with water (10 ml) and the solvent was evaporated under reduced pressure with a yield of 1.8 g of amine 6 as a free base which was used for the next stage without further purification (note 3, 4).
- Note 1. Of the various reaction conditions tested, such as HCOONH4/Pd-C in MeOH, H2/Pd-C in methanol, H2Pd-C/CH3SO3H in methanol, H2/Pd(OH)2/C in methanol, etc., the best conditions we have found hitherto are those described in this Example.
- Note 2. HPLC analysis after 5 hours: amine 6 = 52 % – monobenzyl derivative = 9.3%.
- Note 3. HPLC analysis on the free base: amine 6 = 54 % – monobenzyl derivative = 6%.
- Note 4. 1H-NMR (CDCl3, 600 MHz) δ 7.29 (m, 4H), 7.22-7.14 (m, 6H), 7.00 (s, 1H), 6.83 (bd, 1H), 6.15 (bs, 1H), 4.49 (d, 1H), 4.41 ( d, 1H), 4.25 (m, 1H), 4.08 (m, 1H), 3.38 (m, 1H), 3.29 (m, 1H), 2.99 (s, 3H), 2.92 (dd 1H), 2.84 (m, 2H), 2.76 (m, 1H), 2.47 (m, 1H), 2.28 (m, 1H), 1.76 ( dt, 1H), 1.61 (dt, 1H), 1.38 (d, 6H), 0.95 (d, 3H), 0.89 (d, 3H).
Stage 4
- [0020]
ReagentMolecular weightAmountMmolEq.Amine 6579.801.8 g3.101Compound 7.HCl316.701.28 g4.031.3NaHCO384.00370 mg4.401.4Ethyl acetate 31 ml Sol. 0.1M - [0021]
A solution of the chloride of compound 7 in ethyl acetate was treated with an aqueous sodium bicarbonate solution. The phases were separated and the organic phase was added to a solution of amine 6 in ethyl acetate. The reaction mixture was heated at 60°C for 12 hours, then concentrated; ammonia was added and the solution was maintained under agitation for 1 hour. The organic phase was washed with a 10% aqueous potassium carbonate solution (3x5ml) and with a saturated sodium chloride solution (5 ml); the solvent was removed under reduced pressure. The crude product was purified by flash chromatography on silica gel (eluant ethyl acetate) to give 900 mg of pure Ritonavir (total yield of the two stages 27 %) (note 1).
- Note 1. 1H-NMR (DMSO, 600 MHz) δ 9.05(s, 1H), 7.86 (s, 1H), 7.69 (d, 1H), 7.22-7.10 (m,11H), 6.88 (d, 1H), 6.02 (d, 1H), 5.16 (d, 1H), 5.12 (d, 1H), 4.60 (bs, 1H), 4.48 (d, 1H), 4.42 (d, 1H), 4.15 (m, 1H), 3.94 (dd, 1H), 3.83 (m, 1H), 3.59 (bt, 1H), 3.23 (m, 1H), 2.87 (s, 3H), 2.69-2.63 (m, 3H), 2.60 (m, 1H), 1.88 (m, 1H), 1.45 (m, 2H), 1.30 (d, 6H), 0.74 (d, 6H).
Side effects
When administered at the initially tested higher doses effective for anti-HIV therapy, the side effects of ritonavir are those shown below.[10]
- asthenia, malaise
- diarrhea
- nausea and vomiting
- abdominal pain
- dizziness
- insomnia
- sweating
- taste abnormality
- metabolic effects, including
- hypercholesterolemia
- hypertriglyceridemia
- elevated transaminases
- elevated creatine kinase
One of ritonavir’s side effects is hyperglycemia, through inhibition of the GLUT4 insulin-regulated transporter, thus keeping glucose from entering fat and muscle cells.[medical citation needed] This can lead to insulin resistance and cause problems for people with type 2 diabetes.[medical citation needed]
Drug interactions
Ritonavir induces CYP 1A2 and inhibits the major P450 isoforms 3A4 and 2D6.[according to whom?][medical citation needed] Concomitant therapy of ritonavir with a variety of medications may result in serious and sometimes fatal drug interactions.[11] The list of clinically significant interactions of ritonavir includes the following drugs:
Mechanism of action
Ritonavir was originally developed as an inhibitor of HIV protease, one of a family of pseudo-C2-symmetric small molecule inhibitors.
Ritonavir (center) bound to the active site of HIV protease.[medical citation needed]
Ritonavir is now rarely used for its own antiviral activity but remains widely used as a booster of other protease inhibitors. More specifically, ritonavir is used to inhibit a particular enzyme, in intestines, liver, and elsewhere, that normally metabolizes protease inhibitors, cytochrome P450-3A4 (CYP3A4).[16] The drug binds to and inhibits CYP3A4, so a low dose can be used to enhance other protease inhibitors. This discovery drastically reduced the adverse effects and improved the efficacy of protease inhibitors and HAART. However, because of the general role of CYP3A4 in xenobiotic metabolism, dosing with ritonavir also affects the efficacy of numerous other medications, adding to the challenge of prescribing drugs concurrently.[medical citation needed][17][better source needed]
Pharmocodymanics and pharmacokinetics
The capsules of the medication do not have the same bioavailability as the tablets.[2]
History

New HIV infections and deaths, before and after the FDA approval of “highly active antiretroviral therapy”,[18] of which saquinavir and ritonavir were key as the first two protease inhibitors.[medical citation needed] As a result of the new therapies, HIV deaths in the United States fell dramatically within two years.[18]
Ritonavir is manufactured as Norvir by AbbVie, Inc..[citation needed] The US Food and Drug Administration (FDA) approved ritonavir on March 1, 1996,[19] making it the seventh U.S.-approved antiretroviral drug and the second U.S.-approved protease inhibitor (after saquinavir four months earlier).[citation needed] As a result of the introduction of new “highly active antiretroviral thearap[ies]”—of which the protease inhibitors ritonavir and saquinavir were critical[citation needed]—the annual U.S. HIV-associated death rate fell from over 50,000 to about 18,000 over a period of two years.[20][21]
In 2003, Abbott (now AbbVie, Inc.) raised the price of a Norvir course from US$1.71 per day to US$8.57 per day, leading to claims of price gouging by patients’ groups and some members of Congress. Consumer group Essential Inventions petitioned the NIH to override the Norvir patent, but the NIH announced on August 4, 2004, that it lacked the legal right to allow generic production of Norvir.[22]
In 2014, the FDA approved a combination of ombitasvir/paritaprevir/ritonavir for the treatment of hepatitis C virus (HCV) genotype 4,[3] where the presence of ritonavir again capitalizes on its inhibitory interaction with the human drug metabolic enzyme CYP3A4.
Polymorphism and temporary market withdrawal
Ritonavir was originally dispensed as an ordinary capsule that did not require refrigeration. This contained a crystal form of ritonavir that is now called form I.[23] However, like many drugs, crystalline ritonavir can exhibit polymorphism, i.e., the same molecule can crystallize into more than one crystal type, or polymorph, each of which contains the same repeating molecule but in different crystal packings/arrangements. The solubility and hence the bioavailability can vary in the different arrangements, and this was observed for forms I and II of ritonavir.[24]
During development—ritonavir was introduced in 1996—only the crystal form now called form I was found; however, in 1998, a lower free energy,[25] more stable polymorph, form II, was discovered. This more stable crystal form was less soluble, which resulted in significantly lower bioavailability. The compromised oral bioavailability of the drug led to temporary removal of the oral capsule formulation from the market.[24] As a consequence of the fact that even a trace amount of form II can result in the conversion of the more bioavailable form I into form II, the presence of form II threatened the ruin of existing supplies of the oral capsule formulation of ritonavir; and indeed, form II was found in production lines, effectively halting ritonavir production.[23] Abbott (now AbbVie) withdrew the capsules from the market, and prescribing physicians were encouraged to switch to a Norvir suspension.[citation needed]
The company’s research and development teams ultimately solved the problem by replacing the capsule formulation with a refrigerated gelcap.[when?][citation needed] In 2000, Abbott (now AbbVie) received FDA-approval for a tablet formulation of lopinavir/ritonavir (Kaletra) which contained a preparation of ritonavir that did not require refrigeration.[26]
Research
In 2020, the fixed-dose combination of lopinavir/ritonavir was found not to work in severe COVID-19.[27] In the trial the medication was started around thirteen days after the start of symptoms.[27] Virtual screening of the 1930 FDA-approved drugs followed by molecular dynamics analysis predicted ritonavir blocks the binding of the SARS-CoV-2 spike (S) protein to the human angiotensin-converting enzyme-2 (hACE2) receptor, which is critical for the virus entry into human cells.[28]
References
- ^ Jump up to:a b “Norvir EPAR”. European Medicines Agency (EMA). Retrieved 20 August 2020. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ Jump up to:a b c d e f g h i j k “Ritonavir”. The American Society of Health-System Pharmacists. Archived from the original on 2015-10-17. Retrieved Oct 23, 2015.
- ^ Jump up to:a b “FDA approves Viekira Pak to treat hepatitis C”. Food and Drug Administration. December 19, 2014. Archived from the original on October 31, 2015.
- ^ “Ritonavir Pregnancy and Breastfeeding Warnings”. drugs.com. Archived from the original on 7 September 2015. Retrieved 23 October 2015.
- ^ Jump up to:a b British National Formulary 69 (69 ed.). Pharmaceutical Pr. March 31, 2015. p. 426. ISBN 9780857111562.
- ^ Hacker, Miles (2009). Pharmacology principles and practice. Amsterdam: Academic Press/Elsevier. p. 550. ISBN 9780080919225.
- ^ Fischer, Jnos; Ganellin, C. Robin (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 509. ISBN 9783527607495.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ “First Generic Drug Approvals”. U.S. Food and Drug Administration (FDA). Retrieved 13 February 2021.
- ^ “Norvir side effects (Ritonavir) and drug interactions – prescription drugs and medications at RxList”. June 27, 2007. Archived from the original on 2007-06-27.
- ^ “Ritonavir: Drug Information Provided by Lexi-Comp: Merck Manual Professional”. April 30, 2008. Archived from the originalon 2008-04-30.
- ^ Henry, J. A.; Hill, I. R. (1998). “Fatal interaction between ritonavir and MDMA”. Lancet. 352 (9142): 1751–1752. doi:10.1016/s0140-6736(05)79824-x. PMID 9848354. S2CID 45334940.
- ^ Papaseit, E.; Vázquez, A.; Pérez-Mañá, C.; Pujadas, M.; De La Torre, R.; Farré, M.; Nolla, J. (2012). “Surviving life-threatening MDMA (3,4-methylenedioxymethamphetamine, ecstasy) toxicity caused by ritonavir (RTV)”. Intensive Care Medicine. 38 (7): 1239–1240. doi:10.1007/s00134-012-2537-9. PMID 22460853. S2CID 19375709.
- ^ Nieminen, Tuija H.; Hagelberg, Nora M.; Saari, Teijo I.; Neuvonen, Mikko; Neuvonen, Pertti J.; Laine, Kari; Olkkola, Klaus T. (2010). “Oxycodone concentrations are greatly increased by the concomitant use of ritonavir or lopinavir/ritonavir”. European Journal of Clinical Pharmacology. 66 (10): 977–985. doi:10.1007/s00228-010-0879-1. ISSN 0031-6970. PMID 20697700. S2CID 25770818.
- ^ Hsieh, Yi-Ling; Ilevbare, Grace A.; Van Eerdenbrugh, Bernard; Box, Karl J.; Sanchez-Felix, Manuel Vincente; Taylor, Lynne S. (2012-05-12). “pH-Induced Precipitation Behavior of Weakly Basic Compounds: Determination of Extent and Duration of Supersaturation Using Potentiometric Titration and Correlation to Solid State Properties”. Pharmaceutical Research. 29 (10): 2738–2753. doi:10.1007/s11095-012-0759-8. ISSN 0724-8741. PMID 22580905. S2CID 15502736.
- ^ Zeldin RK, Petruschke RA (2004). “Pharmacological and therapeutic properties of ritonavir-boosted protease inhibitor therapy in HIV-infected patients”. Journal of Antimicrobial Chemotherapy. 53 (1): 4–9. doi:10.1093/jac/dkh029. PMID 14657084.
- ^ “Drug Development and Drug Interactions: Table of Substrates, Inhibitors and Inducers”. U.S. Food and Drug Administration(FDA). December 3, 2019.
- ^ Jump up to:a b (PDF)https://web.archive.org/web/20150924044850/http://www.cdc.gov/mmwr/PDF/wk/mm6021.pdf. Archived from the original (PDF)on 2015-09-24. Retrieved 2020-02-17. Missing or empty
|title=(help) - ^ “Ritonavir FDA approval package” (PDF). 1 March 1996.
- ^ “HIV Surveillance—United States, 1981-2008”. Archived from the original on 9 November 2013. Retrieved 8 November 2013.
- ^ The CDC, in its Morbidity and Mortality Weekly Report, ascribes this to “highly active antiretroviral therapy”, without mention of either of these drugs, see the preceding citation. A further citation is needed to make this accurate connection between this drop and the introduction of the protease inhibitors.
- ^ Ceci Connolly (2004-08-05). “NIH Declines to Enter AIDS Drug Price Battle”. The Washington Post. Archived from the original on 2008-08-20. Retrieved 2006-01-16.
- ^ Jump up to:a b Bauer J; et al. (2001). “Ritonavir: An Extraordinary Example of Conformational Polymorphism”. Pharmaceutical Research. 18 (6): 859–866. doi:10.1023/A:1011052932607. PMID 11474792. S2CID 20923508.
- ^ Jump up to:a b S. L. Morissette; S. Soukasene; D. Levinson; M. J. Cima; O. Almarsson (2003). “Elucidation of crystal form diversity of the HIV protease inhibitor ritonavir by high-throughput crystallization”. Proc. Natl. Acad. Sci. USA. 100 (5): 2180–84. doi:10.1073/pnas.0437744100. PMC 151315. PMID 12604798.
- ^ Lüttge, Andreas (February 1, 2006). “Crystal dissolution kinetics and Gibbs free energy”. Journal of Electron Spectroscopy and Related Phenomena. 150 (2): 248–259. doi:10.1016/j.elspec.2005.06.007.
- ^ “Kaletra FAQ”. AbbVie’s Kaletra product information. AbbVie. 2011. Archived from the original on 7 July 2014. Retrieved 5 July2014.
- ^ Jump up to:a b Cao, Bin; Wang, Yeming; Wen, Danning; Liu, Wen; Wang, Jingli; Fan, Guohui; et al. (18 March 2020). “A Trial of Lopinavir–Ritonavir in Adults Hospitalized with Severe Covid-19”. New England Journal of Medicine. 382 (19): 1787–1799. doi:10.1056/NEJMoa2001282. PMC 7121492. PMID 32187464.
- ^ Bagheri, Milad; Niavarani, Ahmadreza (2020-10-08). “Molecular dynamics analysis predicts ritonavir and naloxegol strongly block the SARS-CoV-2 spike protein-hACE2 binding”. Journal of Biomolecular Structure and Dynamics: 1–10. doi:10.1080/07391102.2020.1830854. ISSN 0739-1102. PMID 33030105. S2CID 222217607.
Further reading
- Chemburkar, Sanjay R.; Bauer, John; Deming, Kris; Spiwek, Harry; Patel, Ketan; Morris, John; Henry, Rodger; Spanton, Stephen; et al. (2000). “Dealing with the Impact of Ritonavir Polymorphs on the Late Stages of Bulk Drug Process Development”. Organic Process Research & Development. 4 (5): 413–417. doi:10.1021/op000023y.
External links
- “Ritonavir”. Drug Information Portal. U.S. National Library of Medicine.
| Clinical data | |
|---|---|
| Trade names | Norvir |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a696029 |
| License data | EU EMA: by INNUS DailyMed: Ritonavir |
| Pregnancy category | AU: B3 |
| Routes of administration | By mouth |
| ATC code | J05AE03 (WHO) |
| Legal status | |
| Legal status | AU: S4 (Prescription only)CA: ℞-onlyUK: POM (Prescription only)US: ℞-onlyEU: Rx-only [1] |
| Pharmacokinetic data | |
| Protein binding | 98-99% |
| Metabolism | Liver |
| Elimination half-life | 3-5 hours |
| Excretion | mostly fecal |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 155213-67-5 |
| PubChem CID | 392622 |
| DrugBank | DB00503 |
| ChemSpider | 347980 |
| UNII | O3J8G9O825 |
| KEGG | D00427 |
| ChEBI | CHEBI:45409 |
| ChEMBL | ChEMBL163 |
| NIAID ChemDB | 028478 |
| PDB ligand | RIT (PDBe, RCSB PDB) |
| CompTox Dashboard (EPA) | DTXSID1048627 |
| ECHA InfoCard | 100.125.710 |
| Chemical and physical data | |
| Formula | C37H48N6O5S2 |
| Molar mass | 720.95 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI | |
| (what is this?) (verify) |
/////////////RITONAVIR, Antiviral, Peptidomimetics, HIV Protease Inhibitor, A-84538, Abbott 84538, ABBOTT-84538, ABT 538, ABT-538, DRG-0244, NSC-693184, TMC 114r
CC(C)[C@H](NC(=O)N(C)CC1=CSC(=N1)C(C)C)C(=O)N[C@H](C[C@H](O)[C@H](CC1=CC=CC=C1)NC(=O)OCC1=CN=CS1)CC1=CC=CC=C1

NEW DRUG APPROVALS
ONE TIME
$10.00

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....











