Home » CLINICAL TRIALS
Category Archives: CLINICAL TRIALS
Tianagliflozin IND filed by Tianjin Institute of Pharmaceutical research
Tianagliflozin,
taigeliejing, 6-deoxydapagliflozin
| Molecular Formula: | C21H25ClO5 |
|---|---|
| Molecular Weight: | 392.8732 g/mol |
IND Filing…Tianjin Institute of Pharmaceutical research
Tianjin Institute Of Pharmaceutical Research,
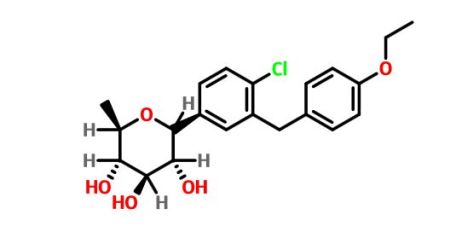
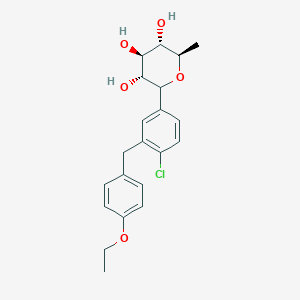
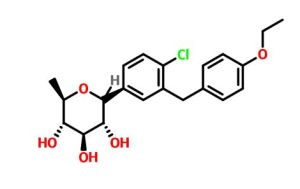
(3R,4S,5S,6R)-2-[4-chloro-3-[(4-ethoxyphenyl)methyl]phenyl]-6-methyloxane-3,4,5-triol
1–[4–Chloro–3–(4–ethoxybenzyl)phenyl]–1,6–dideoxy–β–d–glucopyranose

CAS N. 1461750-27-5
The structures of dapagliflozin and 6-deoxydapagliflozin (1)
,deletion of the 6-OH in the sugar moiety of dapagliflozin led to the discovery of a more potent SGLT2 inhibitor, 6-deoxydapagliflozin (1, ). In an in vitro assay, 1 was a more active SGLT2 inhibitor, with IC 50 = 0.67 nM against human SGLT2 (hSGLT2), as compared with 1.1 nM for dapagliflozin, leading to the identification of 1 as the most active SGLT2 inhibitor discovered so far in this field. Also in an in vivo assay, 1 also introduced more urinary glucose in a rat urinary glucose excretion test (UGE) and exhibited more potent blood glucose inhibitory activity in a rat oral glucose tolerance test (OGTT) than dapagliflozin.
Tianjin Institute Of Pharmaceutical Research,天津药物研究院

SPECTRAL DATA of Tianagliflozin
1 as a white solid (3.65 g, 93 %). R f = 0.35 (EtOAc);
m.p.: 148–149 °C;
1H NMR (400 MHz, DMSO-d 6): δ = 7.35 (d, 1H, J = 8.4 Hz), 7.25 (s, 1H), 7.18 (d, 1H, J = 8.0 Hz), 7.08 (d, 2H, J = 8.4 Hz), 6.81 (d, 2H, J = 8.4 Hz), 4.95 (d, 1H, J = 5.2 Hz, OH), 4.90 (d, 1H, J = 4.4 Hz, OH), 4.79 (d, 1H, J = 5.6 Hz, OH), 3.92–4.01 (m, 5H), 3.24–3.29 (m, 1H), 3.18–3.22 (m, 1H), 3.09–3.15 (m, 1H), 2.89–2.95 (m, 1H), 1.29 (t, 3H, J = 7.0 Hz, CH2 CH 3 ), 1.15 (d, 3H, J = 6.0 Hz, CHCH 3 ) ppm;
13C NMR (100 MHz, DMSO-d 6): δ = 156.85, 139.65, 137.82, 131.83, 131.16, 130.58, 129.52, 128.65, 127.14, 114.26, 80.71, 77.98, 75.77, 75.51, 74.81, 62.84, 37.55, 18.19, 14.62 ppm;
IR (KBr): v¯¯¯ = 3,564 (w), 3,385 (s), 2,981 (s), 2,899 (s), 2,861 (s), 1,613 (m), 1,512 (s), 1,477 (m), 1,247 (s), 1,102 (s), 1,045 (s), 1,012 (s) cm−1;
HR–MS: calcd for C21H29ClNO5 ([M + NH4]+) 410.1729, found 410.1724.
PATENT
CN 103864737
http://www.google.com/patents/CN103864737A?cl=en
PATENT
WO 2014094544
http://www.google.com/patents/WO2014094544A1?cl=en

-27-
1 D1 -6 Optionally, the step (7 ‘) is the step (7’) in place:
LS l- [4 – D (I- Dl- 6)
A.
(DMSO-d 6, 400 MHz), δ 7.35 (d, 1H, J = 8.0 Hz), 7.28 (d, 1H, J ‘. 2.0 Hz), 7.17 (dd, IH, / = 2.0 Hz and 8.4 Hz), 7.05 (d, 2H, J: 8.8 Hz), 6.79 (d, 2H, 8.8 Hz): 4.924,95 (m, 2H), 4,81 (d, IH, 6,0 Hz), 3.93- 3.99 (m, 5H), 3,85 (d, 1H, J = 10,4 Hz), 3,66 (dd, IH, 5,2 Hz and 11,6 Hz), 3.17-3,28 (m, 3H), 3.02-3.08 (m: IH), 1.28 (t, 3H, J = 7,0 Hz), 0,80 (s, 9H), -0.05 (s, 3H), -0.09 (s, 3H) .
PATENT
[0066] The added 100mL dried over anhydrous methanol 0. 5g of sodium metal, nitrogen at room temperature with stirring, until the sodium metal disappeared. Followed by addition of 5. 2g (10mmol) of compound 6, stirring was continued at room temperature for 3 hours. To the reaction system was added 5g strong acid cation exchange resin, stirred at room temperature overnight, the reaction mixture until pH = 7. The resin was removed by suction, and the filtrate evaporated to dryness on a rotary evaporator, the residue was further dried on a vacuum pump to give the product I-D1-6, as a white foamy solid.
PATENT
WO 2014139447
PATENT related
http://www.google.com/patents/WO2013044608A1?cl=en
http://link.springer.com/article/10.1007%2Fs40242-014-4043-9#/page-1
Design of SGLT2 Inhibitors for the Treatment of Type 2 Diabetes: A History Driven by Biology to Chemistry.
Abstract
A brief history of the design of sodium-dependent glucose cotransporter 2 (SGLT2) inhibitors is reviewed. The design of O-glucoside SGLT2 inhibitors by structural modification of phlorizin, a naturally occurring O-glucoside, in the early stage was a process mainly driven by biology with anticipation of improving SGLT2/SGLT1 selectivity and increasing metabolic stability. Discovery of dapagliflozin, a pioneering C-glucoside SGLT2 inhibitor developed by Bristol-Myers Squibb, represents an important milestone in this history. In the second stage, the design of C-glycoside SGLT2 inhibitors by modifications of the aglycone and glucose moiety of dapagliflozin, an original structural template for almost all C-glycoside SGLT2 inhibitors, was mainly driven by synthetic organic chemistry due to the challenge of designing dapagliflozin derivatives that are patentable, biologically active and synthetically accessible. Structure-activity relationships (SAR) of the SGLT2 inhibitors are also discussed.
http://www.ncbi.nlm.nih.gov/pubmed/25557661
Paper

Discovery of 6-Deoxydapagliflozin as a Highly Potent Sodium-dependent Glucose Cotransporter 2 (SGLT2) Inhibitor for the Treatment of Type 2 Diabetes
http://www.ingentaconnect.com/content/ben/mc/2014/00000010/00000003/art00009?crawler=true
CLIP
A facile synthesis of 6-deoxydapagliflozin


The synthetic route to the target compound 1 is shown in Scheme 3. The starting material methyl 2,3,4-tri-O-benzyl-6-deoxy-6-iodo-α–d-glucopyranoside (3) was prepared from commercially available methyl α–d-glucopyranoside (2) according to a known method [5, 6].
Iodide 3 was reductively deiodinated to give 4 in 91 % yield under hydrogenolytic conditions using 10 % Pd/C as catalyst in the presence of Et3N as base in THF/MeOH at room temperature.
when the iodide 3 was treated with Barton–McCombie reagent (n-Bu3SnH/AIBN) [7] in toluene at room temperature no reaction occurred; however, when the reaction was carried out at elevated temperatures, such as reflux, a complex mixture formed with only a trace amount (3 %, entry 1) of the desired product 4.
When the iodide 3 was treated with LiAlH4 in THF at 0 °C to room temperature, another complex mixture was produced with only a trace amount (2 %, entry 2) of 4.
When Pd(OH)2 was used as the hydrogenolysis catalyst instead of 10 % Pd/C, the desired 4 was indeed formed (14 %, entry 4), but most of the starting material was converted to a few more polar byproducts, which were believed to result from the cleavage of at least one of the benzyl groups.
pdf available
Monatshefte für Chemie – Chemical Monthly
December 2013, Volume 144, Issue 12, pp 1903-1910
////////IND Filing, SGLT-2 inhibitor, type 2 diabetes, Tianagliflozin, taigeliejing, 6-deoxydapagliflozin, 1461750-27-5
Clc1c(cc(cc1)C2[C@@H]([C@H]([C@@H]([C@H](O2)C)O)O)O)Cc3ccc(cc3)OCC
Lefucoxib (乐福昔布)
Lefucoxib (乐福昔布)
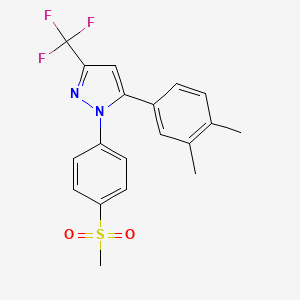
5-(3,4-dimethyl-phenyl)-1-methanesulfonyl-3-trifluoromethol-pyrazole
1 [4- (methylsulfonyl) phenyl] -3-trifluoromethyl-5- (3,4-dimethylphenyl) – pyrazole
CAS 849048-84-6
![]()
| Molecular Formula: | C19H17F3N2O2S |
|---|---|
| Molecular Weight: | 394.41069 g/mol |
IND FILED
Prostaglandin G/H Synthase 2 (PTGS2; COX-2) Inhibitors
A COX-2 inhibitor potentially for the treatment of rheumatoid arthritis.
cyclooxygenase-2 (COX-2) inhibitor
National Center of Biomedical Analysis
![]()
 CHINA FLAG
CHINA FLAG
PATENT
CN 1468854
http://www.google.com/patents/CN1468854A?cl=en
Example 1
1 [4- (methylsulfonyl) phenyl] -3-trifluoromethyl-5- (3,4-dimethylphenyl) – pyrazole (I1)
1- (3,4- two toluene-yl) -4,4,4-trifluoro-methyl – D-1,3-dione (IV1) of sodium metal was weighed 2.3g (0.1mol) was added 50ml of anhydrous toluene to prepare a sodium sand. After cooling, ethanol was added dropwise 12ml, and then heated at 60 ℃, complete reaction of sodium metal. After cooling to room temperature, was added 3,4-dimethylphenyl ethanone 23.8g (0.1mol) and trifluoroacetic ethyl acetate 20ml (0.2mol), reacted at 100 ℃ 5 hours. Toluene was distilled off under reduced pressure, a 10% aqueous hydrochloric acid was added, the pH was adjusted to 2-3, extracted with ethyl acetate, washed with water, dried over anhydrous MgSO4, ethyl acetate was distilled off under reduced pressure. Then under reduced pressure, distillation, collecting fractions 105-107 ℃ / 0.7mmHg, was 14.6g, 60% yield.
1- [4- (methylsulfonyl) phenyl] -3-trifluoromethyl-5- (3,4-dimethylphenyl) – pyrazole (I1) take the above-prepared substituted (IV1) 2.38g (0.01mol ), 15ml of ethanol, then added p-methanesulfonyl phenyl hydrazine salt alkoxide 2.3g (0.01ml). Was refluxed for 15 hours. Place the refrigerator overnight, the crystals were collected by filtration, recrystallized from ethanol, mp 129-31 ℃, to give 3.1 g.
Elemental analysis: C19H17F3N2O2S Calculated: C, 57.86; H, 4.34; N, 7.10 Found: C, 57.97; H, 4.29; N, 7.20MS (m / z): 395 (M + 1)
References
Cheng, Feixiong, Edited by Lee, Philip W, From Handbook of Metabolic Pathways of Xenobiotics (2014), 4, 1655-1656
Bi, X.; Meng, Z.; Chen, H.; Zhu, X.; Dou, G.
In vivo and in vitro metabolism of lefucoxib in rats, J Pharm Biomed Anal. 2008 Sep 10;48(1):134-9. doi: 10.1016/j.jpba.2008.04.024. Epub 2008 Apr 30.
Bi, X.; Meng, Z.; Dou, G. Determination of lefucoxib in rat plasma, urine, and feces by high-performance liquid chromatography with fluorescence detection: Application in pharmacokinetic studies
J Chromatogr B Anal Technol Biomed Life Sci 2007, 850(1-2): 199
Talanta (2011), 85(1), 8-27
Jiefangjun Yaoxue Xuebao (2009), 25(6), 496-498.
Yaowu Fenxi Zazhi (2006), 26(9), 1222-1224.
Zhongguo Yaolixue Yu Dulixue Zazhi (2007), 21(2), 147-151.
| CN101497585B | Jan 31, 2008 | Jan 12, 2011 | 中国科学院理化技术研究所 | Method for photocatalytic synthesis of 1,3,5-trisubstituted-2-pyrazole derivative |

 .
.

//////////c1c(ccc(c1C)C)c2n(nc(c2)C(F)(F)F)c3ccc(cc3)S(=O)(=O)C
CC1=C(C=C(C=C1)C2=CC(=NN2C3=CC=C(C=C3)S(=O)(=O)C)C(F)(F)F)C
How To Apply QbD Principles In Clinical Trials

By Frederic L. “Rick” Sax, M.D., global head for the Center for Integrated Drug Development, Quintiles.
The biopharmaceutical manufacturing industry has used quality by design (QbD) principles for decades. The essence of QbD is designing with the end in mind (in this case, the efficient manufacture of a high-quality drug product). This approach emphasizes that the operative word in QbD is not quality, but design.
read all at
http://www.pharmaceuticalonline.com/doc/how-to-apply-qbd-principles-in-clinical-trials-0001



\
FDA Guidance for Industry: Electronic Source Data in Clinical Investigations
FDA Guidance for Industry: Electronic Source Data in Clinical Investigations
The FDA published its new Guidance for Industry (GfI) – “Electronic Source Data in Clinical Investigations” in September 2013. The Guidance defines the expectations of the FDA concerning electronic source data generated in the context of clinical trials. Find out more about this Guidance.
|
FDA Guidance for Industry: Electronic Source Data in Clinical Investigations |
|
After more than 5 years and two draft versions, the final version of the Guidance for Industry (GfI) – “Electronic Source Data in Clinical Investigations” was published in September 2013. This new FDA Guidance defines the FDA’s expectations for sponsors, CROs, investigators and other persons involved in the capture, review and retention of electronic source data generated in the context of FDA-regulated clinical trials. In an effort to encourage the modernization and increased efficiency of processes in clinical trials, the FDA clearly supports the capture of electronic source data and emphasizes the agency’s intention to support activities aimed at ensuring the reliability, quality, integrity and traceability of this source data, from its electronic source to the electronic submission of the data in the context of an authorization procedure. The Guidance addresses aspects as data capture, data review and record retention. When the computerized systems used in clinical trials are described, the FDA recommends that the description not only focus on the intended use of the system, but also on data protection measures and the flow of data across system components and interfaces. In practice, the pharmaceutical industry needs to meet significant requirements regarding organisation, planning, specification and verification of computerized systems in the field of clinical trials. The FDA also mentions in the Guidance that it does not intend to apply 21 CFR Part 11 to electronic health records (EHR). Author: Source: |
ISPE GAMP R and D and Clinical Systems SIG publish first Concept Paper

| ISPE GAMP R&D and Clinical Systems SIG publish first Concept Paper |
| When changing from paper-based to computerized systems and processes in the field of Good Clinical Practice (GCP), validating these systems is of critical importance, as inspectors are increasingly focussing on this facet of clinical trials. The ISPE GAMP R&D and Clinical Systems SIG has published a Concept Paper on the application of GAMP 5 validation principles to the GCP field. Read more here about the Concept Paper |
|
Monoclonal antibody (mAbs) 2013
2013——-29 monoclonal antibody (mAbs) drugs are in Phase III clinical development.
While around 350 therapeutic mAbs are currently in clinical development globally, only 28 had entered active Phase 2/3 or Phase 3 studies as of January 2013, Additionally one mAb mixture was under evaluation in Phase III.
Historically, mAbs that target antigens relevant to cancer have comprised approximately 50% of the mAb clinical pipeline,
but in 2013 the picture has changed: 66% or 19 of the antibodies to watch in 2013 are for non-cancer indications.

The non-cancer mAbs include alirocumab (Regeneron; Sanofi, hypercholesterinemia);
AMG 145 (Amgen, hypercholesterinemia),
epratuzumab (UCB, SLE),
gantenerumab (Roche; Alzheimer’s disease),
gevokizumab (Xoma/Servier, Non-infectious uveitis),
itolizumab (Biocon, Plaque psoriasis), ixekizumab (Eli Lilly and Co., psoriasis),
lebrikizumab (Roche/Genentech, rheumatoid arthritis),
mepolizumab (GSK, Asthma, COPD etc.),
ocrelizumab (Roche/Genentech, multiple sclerosis),
reslizumab (Teva, Eosinophilic asthma), romosozumab (Amgen, Postmenopausal osteoporosis),
sarilumab (Regeneron; Sanofi, rheumatoid arthritis),
secukinumab (Novartis, rheuma, psoriasis),
sirukumab (Janssen R&D LLC, rheumatoid arthritis),
solanezumab (Eli Lilly and Co., Alzheimer’s disease),
tabalumab (Eli Lilly and Co., rheuma, SLE)
and
vedolizumab (Millenium, Ulcerative colitis; Crohn disease).
The mixture of actoxumab and bezlotoxumab (MK-3415A, Merck & Co.) is being evaluated in two Phase 3 studies as a treatment for Clostridium difficile infection.
The ten cancer mAbs are:
elotuzumab (Bristol-Myers Squibb, Abbott, multiple myeloma),
farletuzumab (Morphotek, ovarian cancer),
inotuzumab ozogamicin (Pfizer; UCB, ALL, NHL),
naptumomab estafenatox (Active Biotech, renal cell carcinoma),
necitumumab (ImClone LLC, NSCL),
nivolumab (Bristol-Myers Squibb, NSCL, renal cell carcinoma),
obinutuzumab (Roche/Genetech, Diffuse large B cell lymphoma, CLL, NHL),
onartuzumab (Roche/Genetech, NSCL cancer; gastric cancer),
racotumomab (CIMAB; Laboratorio Elea S.A.C.I.F. y A, NSCL),
and ramucirumab (ImClone LLC, Gastric; liver, breast, colorectal, NSCL cancers).
Antibody
Sihuan Pharma’s clinical study application for oncology drug Pirotinib accepted by CFDA

The China Food and Drug Administration (CFDA) has accepted Sihuan Pharmaceutical’s application for clinical trial approval for its Pirotinib, a Category 1.1 innovative oncology drug developed by the company’s drug R&D team.

By developing Pirotinib, Sihuan Pharma has demonstrated its capability for the oncology products market. The company holds the largest cardio-cerebral vascular (CCV) drug franchise in China’s prescription market.
The new drug is a second generation (pan-HER) inhibitor intended to treat patients with lung and breast cancer.http://www.pharmaceutical-technology.com/news/newssihuan-pharmas-clinical-study-application-oncology-drug-pirotinib-accepted-cfda?WT.mc_id=DN_News

Access 4,000+ profiles of new drugs in development!
Sign up for a 5-day trial and learn why the Drugs in Clinical Trials Database is a cost-effective way to find detailed information on new drug therapies in hundreds of disease conditions worldwide, monitor drug performance, track competitors and find study opportunities.

India approved 26 drugs without clinical trials
New Delhi: Officials in the Indian health ministry have admitted that about 26 new drug molecules were given approval since 2010 without conducting any proper clinical trials on local population to test their safety and efficacy. Despite strict instructions by the parliamentary standing committee on health, so many new drugs have continued to make their way into the market.
19 August 2013 Officials in the Indian health ministry has accepted that about 26 new drugs were permitted for sale in the country without holding any clinical trials on Indian patients to test their safety and efficacy –
Read more at: http://www.biospectrumasia.com/biospectrum/news/193708/india-approved-26-drugs-clinical-trials#.UhHPwaI3CSo
DR A.M. CRASTO
Pharmathene Will Proceed With Anthrax Vaccine Test
May 31, 2013 — PharmAthene Inc. said Thursday that the Food and Drug Administration will allow it to proceed with new clinical trials of its experimental anthrax vaccine SparVax.
http://www.pharmalive.com/pharmathene-will-proceed-with-anthrax-vaccine-test

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....