Home » Uncategorized
Category Archives: Uncategorized
Egalognastat
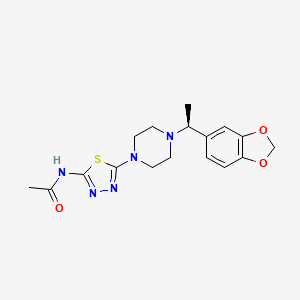
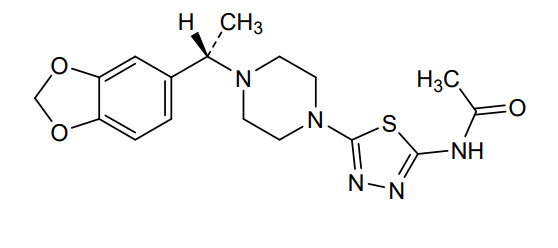
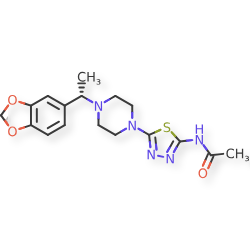
Egalognastat
CAS 1884154-02-2
MF C17H21N5O3S MW375.45
(S)-N-(5-(4-(1-(BENZO(D)(1,3)DIOXOL-5-YL)ETHYL)PIPERAZIN-1-YL)-1,3,4-THIA-DIAZOL-2-YL)ACETAMIDE
ACETAMIDE, N-(5-(4-((1S)-1-(1,3-BENZODIOXOL-5-YL)ETHYL)-1-PIPERAZINYL)-1,3,4-THIADIAZOL-2-YL)-
N-(5-{4-[(1S)-1-(2H-1,3-benzodioxol-5-yl)ethyl]piperazin-1-yl}-1,3,4-thiadiazol-2-yl)acetamide
O-GlcNAcase enzyme inhibitor, ASN90, ASN 90, E9QIS63WUM, ASN 120290
Egalognastat (ASN90) is a potent, selective, and brain-penetrant O-GlcNAcase (OGA) inhibitor (\(IC_{50} = 10.2 \text{ nM}\)). It acts as a disease-modifying agent in pre-clinical studies for neurodegenerative conditions by enhancing protein O-GlcNAcylation, which regulates tau and \(\alpha \)-synuclein pathology. Egalognastat is under investigation for diseases like Alzheimer’s and Parkinson’s.
Key Details on Egalognastat (ASN90):
- Mechanism: As a substrate-competitive OGA inhibitor, it binds to the OGA enzyme and reduces the removal of O-GlcNAc from proteins.
- Disease Targets: It is primarily studied for tauopathies and \(\alpha \)-synucleinopathies.
- Efficacy: Preclinical data shows it raises O-GlcNAcylation of brain proteins and has shown therapeutic potential in models of neurodegeneration.
- Distinction: Unlike earlier sugar-based inhibitors (like Thiamet G), Egalognastat is chemically distinct.
- Status: It is primarily used for research and preclinical development.
- Related Research: Recent studies (2025) have analyzed the potential synaptotoxic effects of OGA inhibitors, including Egalognastat (ASN90) and Ceperognastat, indicating that while they are effective for removing misfolded proteins, they may interfere with synaptic plasticity.
SYN
Publication Name: Journal of Medicinal Chemistry
Publication Date: 2024-08-07
PMID: 39109492
DOI: 10.1021/acs.jmedchem.4c01132
SYN
PAT
PAT
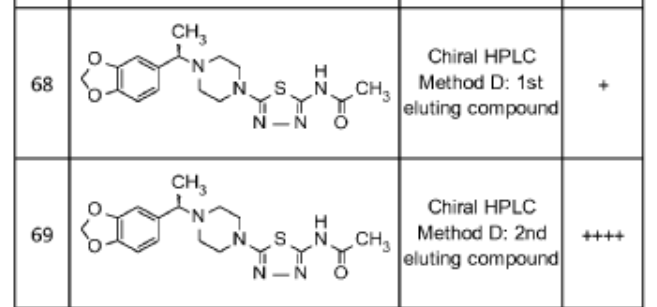

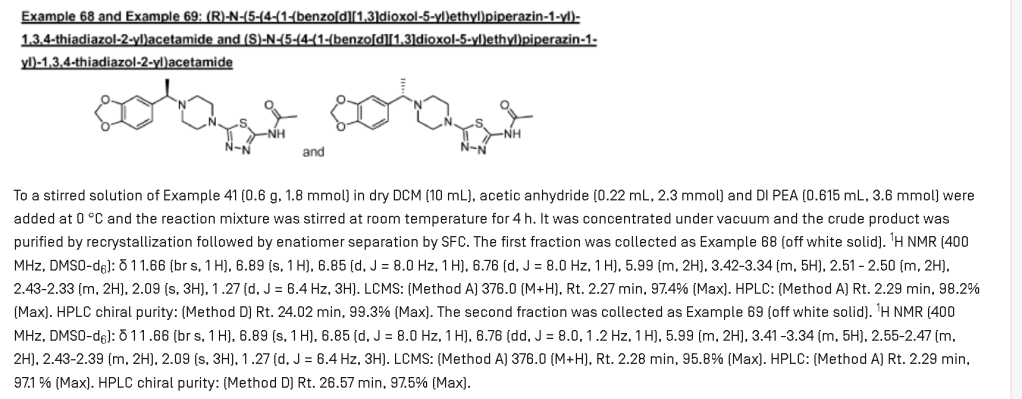
PAT
- Glycosidase inhibitorsPublication Number: US-12187741-B2Priority Date: 2014-08-28Grant Date: 2025-01-07
- Glucosidase inhibitorsPublication Number: ES-2893289-T3Priority Date: 2014-08-28Grant Date: 2022-02-08
- Glycosidase inhibitorsPublication Number: EP-3186243-B1Priority Date: 2014-08-28Grant Date: 2021-07-21
- Glycosidase inhibitorsPublication Number: US-10336775-B2Priority Date: 2014-08-28Grant Date: 2019-07-02
- Glycosidase inhibitorsPublication Number: US-11046712-B2Priority Date: 2014-08-28Grant Date: 2021-06-29
- Glycosidase inhibitorsPublication Number: EP-3868752-A1Priority Date: 2014-08-28
- Glycosidase InhibitorsPublication Number: CN-107108601-APriority Date: 2014-08-28
- Glycosidase inhibitorsPublication Number: US-2019367533-A1Priority Date: 2014-08-28
- Glycosidase inhibitorsPublication Number: WO-2016030443-A1Priority Date: 2014-08-28
- Glycosidase inhibitorsPublication Number: EP-3186243-A1Priority Date: 2014-08-28
- Glycosidase inhibitorPublication Number: JP-2019206554-APriority Date: 2014-08-28
- Glycosidase inhibitorsPublication Number: US-2017298082-A1Priority Date: 2014-08-28



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
////////egalognastat, anax labs, O-GlcNAcase enzyme inhibitor, ASN90, ASN 90, E9QIS63WUM, ASN 120290
Dirozalkib
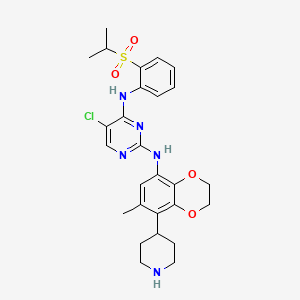
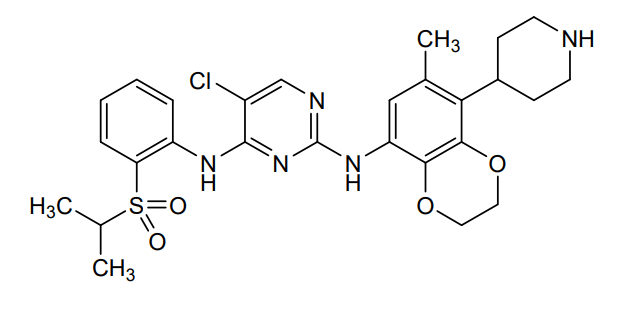
Dirozalkib
CAS 1893419-37-8
MF C27H32ClN5O4S MW558.1 g/mol
5-chloro-2-N-(6-methyl-5-piperidin-4-yl-2,3-dihydro-1,4-benzodioxin-8-yl)-4-N-(2-propan-2-ylsulfonylphenyl)pyrimidine-2,4-diamine

anaplastic lymphoma kinase (ALK) inhibitor, antineoplastic, XZP-3621, XZP 3621, Xuanzhu Biopharmaceutical, 2FH56C28YT
Dirozalkib (XZP-3621) is a novel, potent, and highly selective ALK/ROS1 tyrosine kinase inhibitor developed by Xuanzhu Biopharmaceutical to treat advanced ALK-positive non-small cell lung cancer (NSCLC). It demonstrated high efficacy (47.4% ORR, up to 89.3% in naive patients) in clinical trials and is designed to overcome resistance to earlier inhibitors.
Key Aspects of Dirozalkib
- Indication: Treatment of adult patients with ALK-positive locally advanced or metastatic non-small cell lung cancer (NSCLC).
- Mechanism: Acts as a dual-target ALK/ROS1 tyrosine kinase inhibitor (TKI), effective against ALK fusion-positive cells and various resistance mutations.
- Clinical Efficacy (Phase I/II): In studies, the drug showed significant antitumor activity with an Objective Response Rate (ORR) of 47.4% and an 89.3% ORR in ALK inhibitor-naive patients at 500 mg/day.
- Safety Profile: No dose-limiting toxicities occurred; the maximum tolerated dose was 600 mg/day, with a recommended dose of 500 mg/day. Common adverse events included diarrhea.
- Status: As of early 2026, the NDA (New Drug Application) for Dexitinib (Dirozalkib) was accepted by China’s NMPA, with potential for further market expansion.
- OriginatorXuanzhu Biopharmaceutical
- Class2 ring heterocyclic compounds; Amines; Aniline compounds; Antineoplastics; Chlorinated hydrocarbons; Piperidines; Pyrimidines; Small molecules; Sulfones
- Mechanism of ActionAnaplastic lymphoma kinase inhibitors
- RegisteredNon-small cell lung cancer
- 26 Aug 2025Chemical structure information added.
- 22 Aug 2025Registered for Non-small cell lung cancer (Late-stage disease) in China (PO) – First global approval
- 22 Aug 2025Efficacy and adverse events data from a phase III trial in Non-small cell lung cancer released by Xuanzhu Biopharmaceutical
- A Phase I Study of XZP-3621 in Chinese Patients With ALK or ROS1 Rearrangement Non-small Cell Lung CancerCTID: NCT05055232Phase: Phase 1Status: CompletedDate: 2025-07-24
- Food Effect and Mass Balance Study of XZP-3621 TabletsCTID: NCT05034120Phase: Phase 1Status: CompletedDate: 2025-05-25
- A Study of XZP-3621 in Chinese Patients With ALK Positive NSCLCCTID: NCT05482087Phase: Phase 2Status: Unknown statusDate: 2022-08-01
- A Study to Evaluate and Compare the Efficacy and Safety of XZP-3621 Versus CrizotinibCTID: NCT05204628Phase: Phase 3Status: Unknown statusDate: 2022-01-24
PAT
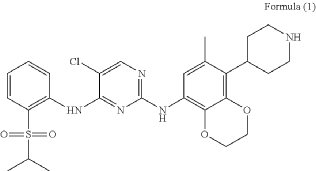
PAT
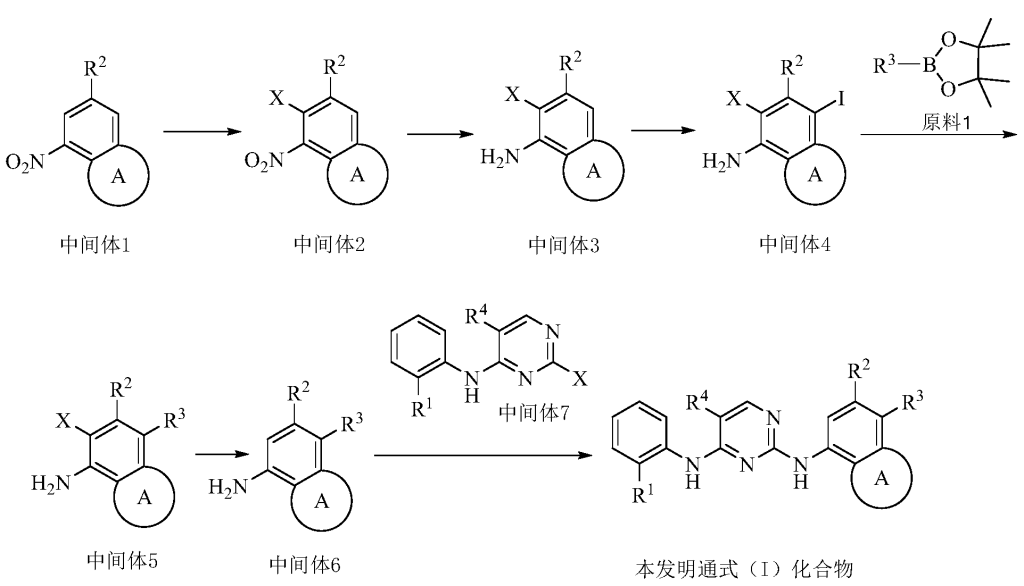
Example 3 Preparation of 2-((5-chloro-2-((7-methyl-8-(piperidin-4-yl)-2,3-dihydrobenzo[b][1,4]dioxin- 5-yl)amino)pyrimidin-4-yl)amino)-N,N-dimethylbenzenesulfonamide (compound 3)
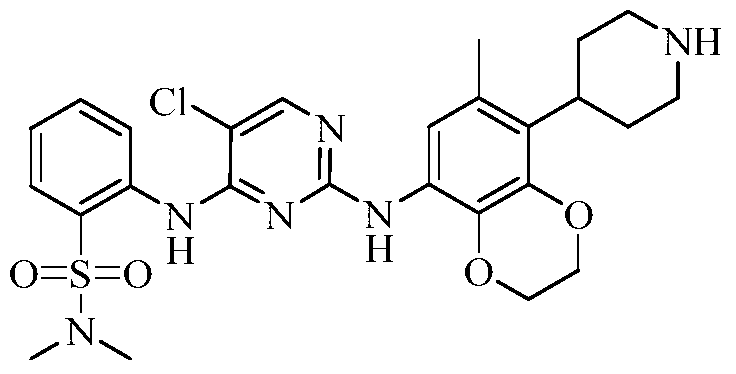
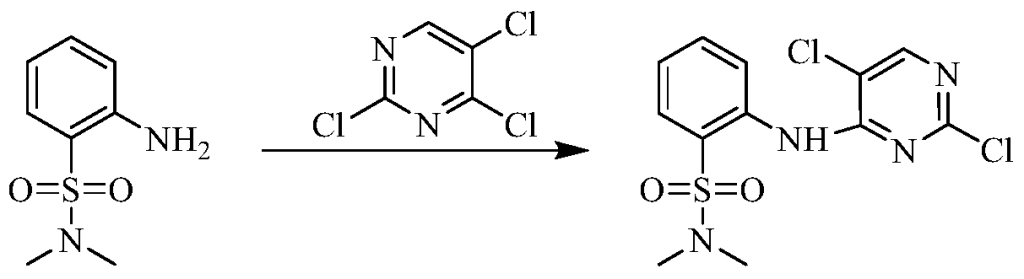
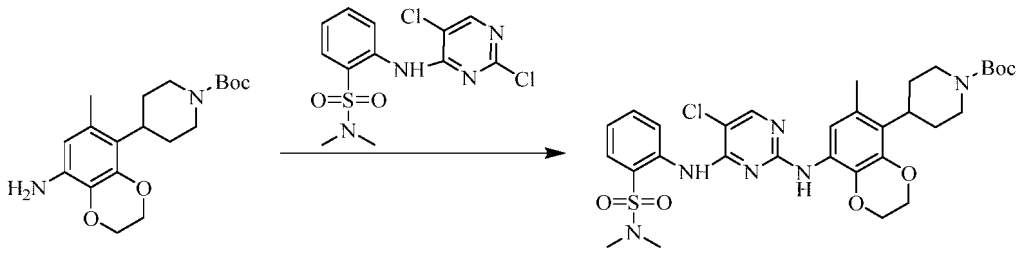
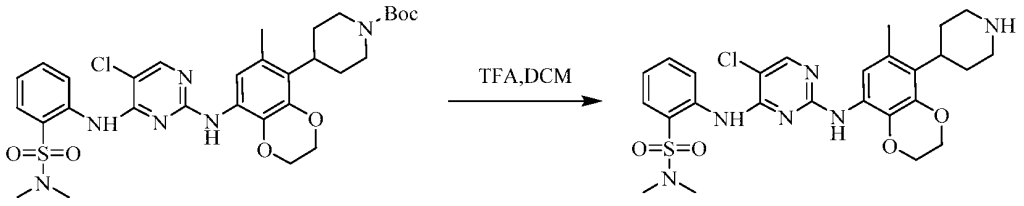
(5) Preparation of 2-((5-chloro-2-((7-methyl-8-(piperidin-4-yl)-2,3-dihydrobenzo[b][1,4]dioxin-5-yl)amino)pyrimidin-4-yl)amino)-N,N-dimethylbenzenesulfonamide
75 mg (0.114 mmol) of tert-butyl 4-(8-((5-chloro-4-((2-(N,N-dimethylaminosulfonyl)phenyl)amino)pyrimidin-2-yl)amino)-6-methyl-2,3-dihydrobenzo[b][1,4]dioxin-5-yl)piperidine-1-carboxylic acid ester was dissolved in dichloromethane (10 mL), and trifluoroacetic acid (1 mL) was added. The mixture was stirred at room temperature for 12 hours. The starting material disappeared as detected by TLC. Water (20 mL) was added, and the mixture was separated. The aqueous phase was extracted twice with dichloromethane (20 mL × 2). The organic phases were combined, dried over anhydrous sodium sulfate, and the solvent was removed by rotary evaporation. The crude product was purified by silica gel column chromatography (methanol:dichloromethane = 1:50) to obtain the final product (30 mg, yield 47.2%).
[0415]Molecular formula:
C26H31ClN6O4S Molecular weight: 559.08 LC-MS (m / z): 280.2 [ M /2+H ] +
[0416]
1H-NMR(400MHz,MeOD)δ:8.44(d,1H,J=1.2),8.11(s,1H),7.86(d,1H,J=1.2),7.56-7.60(m,1H),7.28-7.35(m,2H),4.26(s,4H),3.45-3.48(m,2H),3.06-3.15(m,3H),2.56-2.74(m,8H),2.17(s,3H),1.76-1.80(m,2H).



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Polycyclic inhibitor of anaplastic lymphoma kinasePublication Number: US-10011592-B2Priority Date: 2014-09-29Grant Date: 2018-07-03
- Polycyclic inhibitor of anaplastic lymphoma kinasePublication Number: US-2018086745-A9Priority Date: 2014-09-29
- Polycyclic anaplastic lymphoma kinase inhibitorPublication Number: EP-3202765-A1Priority Date: 2014-09-29
- Polycyclic inhibitor of anaplastic lymphoma kinasePublication Number: KR-20170055555-APriority Date: 2014-09-29
- Crystal form of polycyclic anaplastic lymphoma kinase inhibitorPublication Number: US-2023348443-A1Priority Date: 2020-01-17
- Crystal form of polycyclic anaplastic lymphoma kinase inhibitorPublication Number: US-12441717-B2Priority Date: 2020-01-17Grant Date: 2025-10-14
- Polycyclic inhibitor of anaplastic lymphoma kinasePublication Number: US-2017240534-A1Priority Date: 2014-09-29
- Polycyclic inhibitor of anaplastic lymphoma kinasePublication Number: KR-101909404-B1Priority Date: 2014-09-29Grant Date: 2018-10-17
- Polycyclic anaplastic lymphoma kinase inhibitorPublication Number: WO-2016050171-A1Priority Date: 2014-09-29
- Pharmaceutical composition of anaplastic lymphoma kinase inhibitor and preparation method thereforPublication Number: WO-2025140560-A1Priority Date: 2023-12-28
- CRYSTALLINE FORM OF A POLYCYCLIC ANAPLASIC LYMPHOMA KINASE INHIBITORPublication Number: EP-4092021-A4Priority Date: 2020-01-17
- Polycyclic anaplastic lymphoma kinase inhibitor crystalline formPublication Number: CN-113135905-BPriority Date: 2020-01-17Grant Date: 2023-11-21
- Crystal form of polycyclic anaplastic lymphoma kinase inhibitorPublication Number: WO-2021143819-A1Priority Date: 2020-01-17
- Crystal form of polycyclic anaplastic lymphoma kinase inhibitorPublication Number: EP-4092021-A1Priority Date: 2020-01-17
/////////dirozalkib, anax labs, anaplastic lymphoma kinase (ALK) inhibitor, antineoplastic, XZP-3621, XZP 3621, Xuanzhu Biopharmaceutical, 2FH56C28YT
Deulumateperone
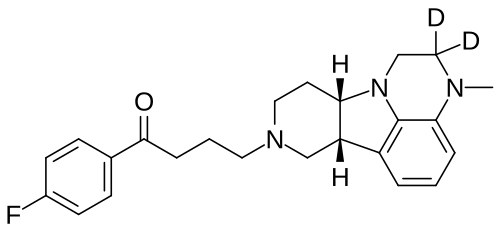
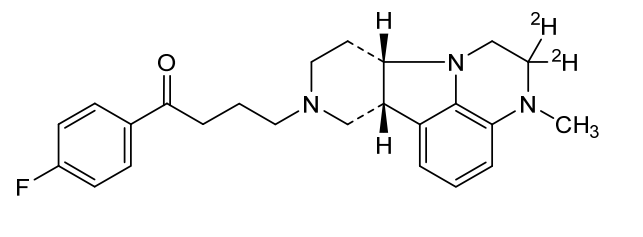
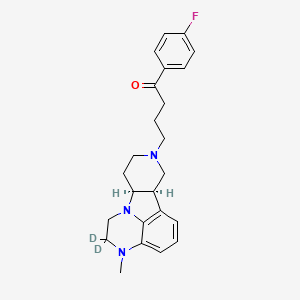
Deulumateperone
CAS 2102683-75-8
MF C24H262H2FN3O MW 395.5 g/mol
4-[(10R,15S)-3,3-dideuterio-4-methyl-1,4,12-triazatetracyclo[7.6.1.05,16.010,15]hexadeca-5,7,9(16)-trien-12-yl]-1-(4-fluorophenyl)butan-1-one
- 1-(4-Fluorophenyl)-4-[(6bR,10aS)-2,3,6b,9,10,10a-hexahydro-2-d-3-methyl-1H-pyrido[3′,4′:4,5]pyrrolo[1,2,3-de]quinoxalin-8(7H)-yl-2-d]-1-butanone
- 1-Butanone, 1-(4-fluorophenyl)-4-[(6bR,10aS)-2,3,6b,9,10,10a-hexahydro-2-d-3-methyl-1H-pyrido[3′,4′:4,5]pyrrolo[1,2,3-de]quinoxalin-8(7H)-yl-2-d]-
antipsychotic, ITI-1284, ITI 1284, NBA7J58PPP,
Deulumateperone (INNTooltip International Nonproprietary Name; developmental code name ITI-1284) is an experimental antipsychotic of the pyridopyrroloquinoxaline and butyrophenone families as well as a deuterated analogue of lumateperone which is under development as a sublingually administered orally disintegrating tablet (ODT) for the treatment of psychotic disorders, agitation, and generalized anxiety disorder.[2][3][4][1] No recent development has been reported for treatment of depressive disorders, behavioral disorders, and dementia.[2] It is being developed by Intra-Cellular Therapies.[2][3][1] As of January 2025, it has reached phase 2 clinical trials.[2][3]
SYN
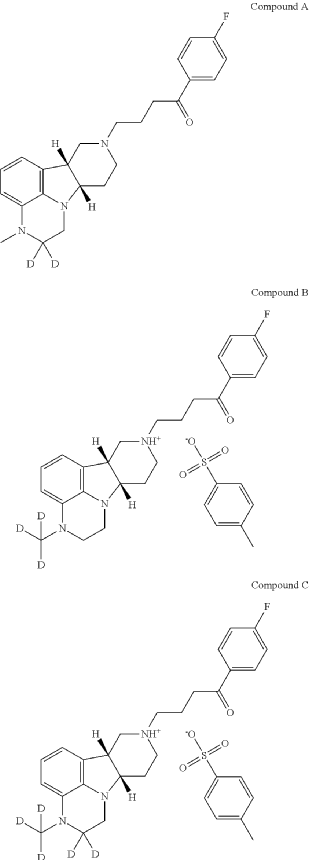
PAT
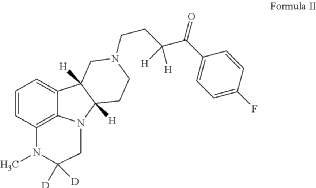
Example 2
2,2-D2-1-(4-fluorophenyl)-4-((6bR,10aS)-3-methyl-2,3,6b,7,10,10a-hexahydro-1H-pyrido[3′,4′: 4,5]pyrrolo[1,2,3-de]quinoxalin-8(9H)-yl)butan-1-one
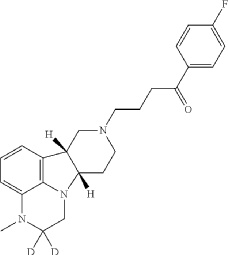
| To a suspension of (6bR, 10aS)-3-Methyl-2-oxo-2,3,6b,9,10,10a-hexahydro-1H,7H-pyrido[3′,4′: 4,5]pyrrolo[1,2,3-de]quinoxaline-8-carboxylic acid ethyl ester (945 mg, 3 mmol) in THF (5 mL) is slowly added BD 3-THF (1.0 M in THF, 10 mL, 10 mmol) at room temperature. After completion of the addition, the reaction mixture is stirred at room temperature overnight and then carefully quenched with D 2O (2.0 mL). The solvent is removed under vacuum and the residue is suspended in HCl (12 N, 9 mL). After stirred at 95° C. for 20 h, the reaction mixture is cooled to room temperature and then adjusted to pH of 12 with 50% NaOH. The mixture is concentrated to dryness to give 2,2-d 2-(6bR, 10aS)-3-Methyl-2,3,6b,7,8,9,10,10a-octahydro-1H-pyrido[3′,4′: 4,5]pyrrolo[1, 2,3-de]quinoxaline as a brown solid, which is used directly for next step without further purification. MS (ESI) m/z 232.2 [M+H] +. |
PAT
- Novel compositions and methodsPublication Number: US-2021315891-A1Priority Date: 2018-08-29
- Transmucosal and subcutaneous compositionsPublication Number: US-10716786-B2Priority Date: 2017-03-24Grant Date: 2020-07-21
- Novel compositions and methodsPublication Number: US-2018271862-A1Priority Date: 2017-03-24
- Transmucosal methods for treating psychiatric and neurological conditionsPublication Number: US-11052083-B2Priority Date: 2017-03-24Grant Date: 2021-07-06
- Novel compositions and methodsPublication Number: US-2020375988-A1Priority Date: 2017-03-24
- Novel compositions and methodsPublication Number: US-2021361648-A1Priority Date: 2017-03-24
- Organic compoundsPublication Number: US-2019231780-A1Priority Date: 2016-03-25
- Organic compoundsPublication Number: US-10688097-B2Priority Date: 2016-03-25Grant Date: 2020-06-23
- Organic compoundsPublication Number: US-11096944-B2Priority Date: 2016-03-25Grant Date: 2021-08-24
- Organic compoundsPublication Number: US-2020352949-A1Priority Date: 2016-03-25
- Organic compounds
- Publication Number: US-2022008423-A1
- Priority Date: 2016-03-25



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
References
- Oguma T, Jino K (2024). “Clinical Pipelines for Alzheimer’s Disease Psychosis and Agitation”. Chemical & Pharmaceutical Bulletin. 72 (7) c23-00416: 610–617. doi:10.1248/cpb.c23-00416. PMID 38945937.
- “Intra-Cellular Therapies”. AdisInsight. 29 January 2025. Retrieved 26 February 2025.
- “Delving into the Latest Updates on ITI-1284 with Synapse”. Synapse. 20 February 2025. Retrieved 26 February 2025.
- “Proposed INN: List 132 International Nonproprietary Names for Pharmaceutical Substances (INN)” (PDF). WHO Drug Information. 38 (4): 1073. 2024.
| Clinical data | |
|---|---|
| Other names | Lumateperone deuterated; Deuterated lumateperone; ITI-1284; ITI1284; ITI-1284-ODT-SL |
| Routes of administration | Sublingual (orally disintegrating tablet)[1] |
| Drug class | Atypical antipsychotic |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2102683-75-8 |
| PubChem CID | 140916642 |
| UNII | NBA7J58PPP |
| KEGG | D13268 |
| Chemical and physical data | |
| Formula | C24H26D2FN3O |
| Molar mass | 395.518 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
////////deulumateperone, anax labs, antipsychotic, ITI-1284, ITI 1284, NBA7J58PPP,
Vepdegestrant
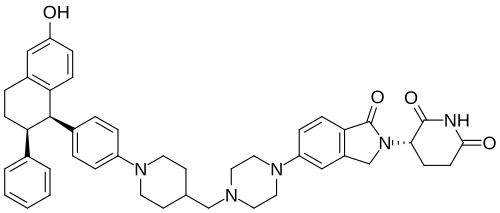
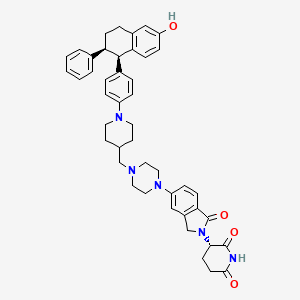
Vepdegestrant
CAS 2229711-08-2
MW 723.9 g/mol, C45H49N5O4
- (S)-3-(5-(4-((1-(4-((1R,2S)-6-Hydroxy-2-phenyl-1,2,3,4-tetrahydronaphthalen-1-yl)phenyl)piperidin-4-yl)methyl)piperazin-1-yl)-1-oxoisoindolin-2-yl)piperidine-2,6-dione
- (3S)-3-[5-[4-[[1-[4-[(1R,2S)-6-hydroxy-2-phenyl-tetralin-1-yl]phenyl]-4-piperidyl]methyl]piperazin-1-yl]-1-oxo-isoindolin-2-yl]piperidine-2,6-dione
- (2(1)R,2(2)S,8(3)S)-2-hydroxy-2(1),2(2),2(3),2-tetrahydro-7(5,2)-isoindola-6(1,4)-piperazina-4(1,4),8(3)-dipiperidina-2(2,1)-naphthalena-1(1),3(1,4)-dibenzenaoctaphane-7(1),8(2),8(7(3)H)-trione
- 2,6-Piperidinedione, 3-(1,3-dihydro-1-oxo-5-(4-((1-(4-((1R,2S)-1,2,3,4-tetrahydro-6-hydroxy-2-phenyl-1-naphthalenyl)phenyl)-4-piperidinyl)methyl)-1-piperazinyl)-2H-isoindol-2-yl)-, (3S)-
(3S)-3-[6-[4-[[1-[4-[(1R,2S)-6-hydroxy-2-phenyl-1,2,3,4-tetrahydronaphthalen-1-yl]phenyl]piperidin-4-yl]methyl]piperazin-1-yl]-3-oxo-1H-isoindol-2-yl]piperidine-2,6-dione
5/1/2026, FDA 2026, APROVALS 2026, Veppanu, ARV 471, WC1U3R1YMI, PF 07850327
To treat estrogen receptor-positive, human epidermal growth factor receptor 2-negative, ESR1-mutated advanced or metastatic breast cancer with disease progression following at least one line of endocrine therapy
On May 1, 2026, the FDA approved vepdegestrant (Veppanu), a first-in-class oral PROTAC estrogen receptor (ER) degrader developed by Arvinas and Pfizer, for adults with ER-positive, HER2-negative, ESR1-mutated advanced or metastatic breast cancer who have progressed on endocrine therapy. It demonstrated significant progression-free survival (PFS) improvements compared to fulvestrant.
Key Details About Vepdegestrant (Veppanu):
- Mechanism of Action: As an oral PROTAC (Proteolysis-Targeting Chimera), vepdegestrant targets the estrogen receptor for degradation, designed to be more effective than traditional endocrine therapies, particularly in ESR1-mutated tumors.
- Approved Indication: For treating adults with ER+/HER2-, ESR1-mutated advanced/metastatic breast cancer (detected by Guardant360 CDx) after at least one line of endocrine therapy.
- Dosage: The recommended dose is 200 mg taken orally once daily with food.
- Clinical Efficacy (VERITAC-2): In trials, vepdegestrant showed a significantly longer PFS compared to intramuscular fulvestrant.
- Side Effects & Risks: Common side effects include decreased white blood cell counts, increased liver function tests, muscle/bone pain, fatigue, and nausea. Warnings include embryo-fetal toxicity and QTc interval prolongation (heart rhythm issues).
- Companion Diagnostic: Guardant360 CDx was approved alongside the drug to identify patients with ESR1 mutations
Vepdegestrant (developmental code name ARV-471) is an investigational oral proteolysis-targeting chimera (PROTAC) compound that targets the estrogen receptor for protein degradation. It is being developed for the treatment of estrogen receptor-positive, HER2-negative (ER+/HER2-) breast cancer by Arvinas and Pfizer.[1][2][3]
Mechanism of action
Vepdegestrant is designed as a PROTAC that recruits the ubiquitin-proteasome system to target the estrogen receptor for degradation.[4] The compound contains both an E3 ubiquitin ligase-binding moiety and an estrogen receptor-binding domain, intended to bring these proteins into proximity to trigger ubiquitination and subsequent proteasomal degradation of the ER protein.[5] In laboratory studies, vepdegestrant demonstrated ER degradation in ER-positive breast cancer cell lines with reported DC50 values of approximately 1-2 nM.[6]
Vepdegestrant is an orally available hetero-bifunctional molecule and selective estrogen receptor (ER) alpha-targeted protein degrader, using the proteolysis targeting chimera (PROTAC) technology, with potential antineoplastic activity. Vepdegestrant is composed of an ER alpha ligand attached to an E3 ligase recognition moiety. Upon oral administration,vepdegestrant targets and binds to the ER ligand binding domain on ER alpha. E3 ligase is recruited to the ER by the E3 ligase recognition moiety and ER alpha is tagged by ubiquitin. This causes ubiquitination and degradation of ER alpha by the proteasome. This decreases ER alpha protein levels, decreases the expression of ER alpha-target genes and halts ER-mediated signaling. This results in an inhibition of proliferation in ER alpha-overexpressing tumor cells. In addition, the degradation of the ER alpha protein releases the ARV-471 and can bind to additional ER alpha target proteins. ER alpha is overexpressed in a variety of cancers and plays a key role in cancer cell proliferation.
SYN
https://chemistry-europe.onlinelibrary.wiley.com/doi/10.1002/slct.202405939
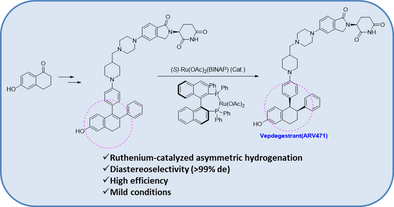
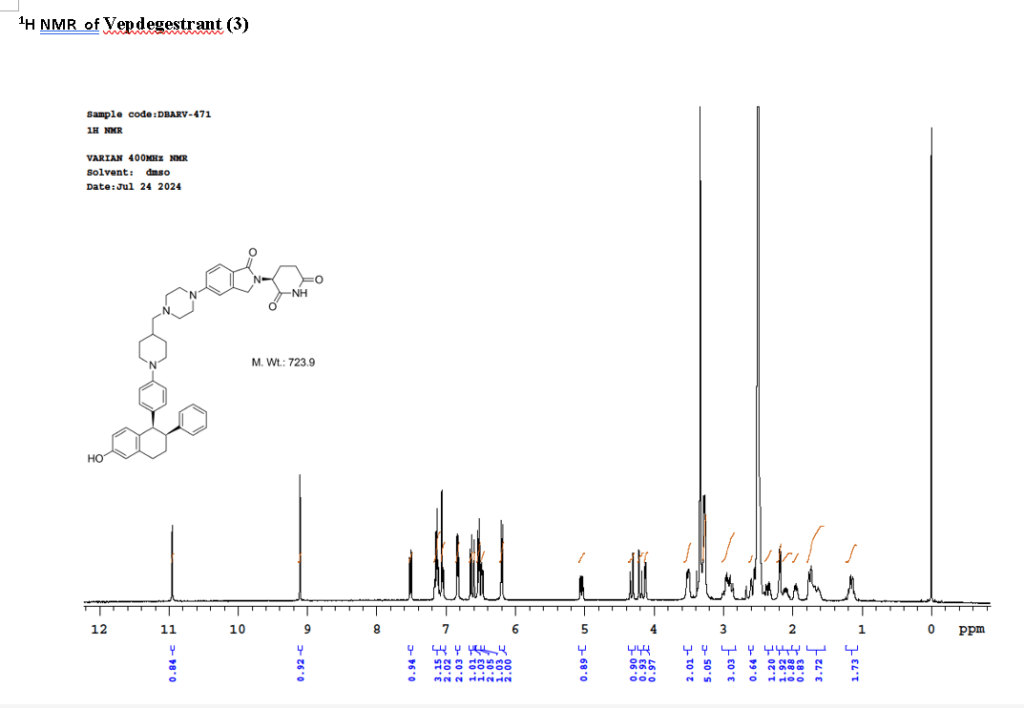
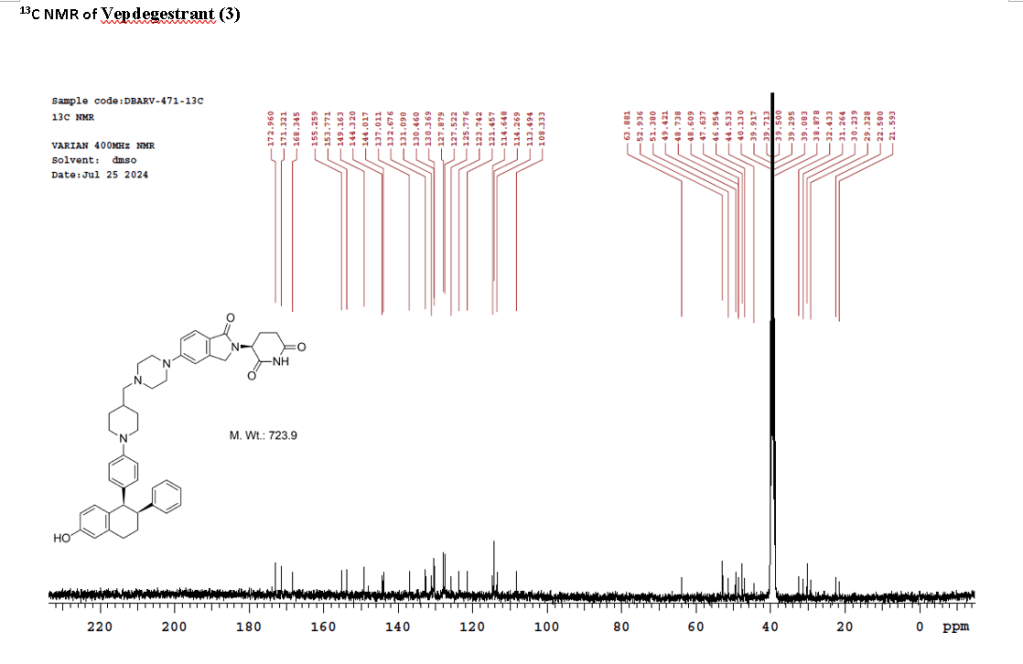
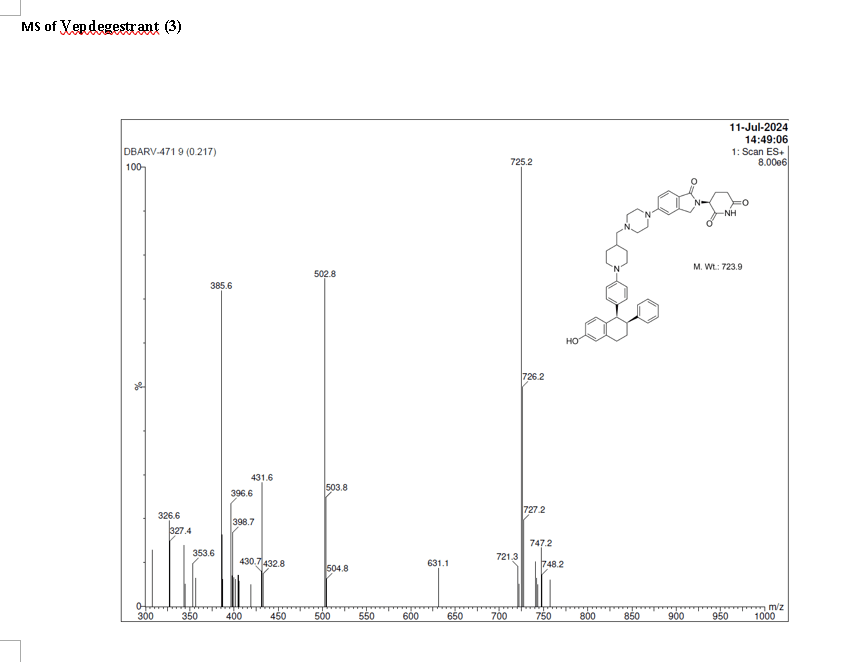
PAT
Step 11: Preparation of 3-[5-[4-[[1-[4-[(1R, 2S)-6-hydroxy-2-phenyl-tetralin-1-yl]phenyl]-4-piperidyl]methyl]piperazin-1-yl]-1-oxo-isoindolin-2-yl]piperidine-2,6-dione (Compound (I-b))
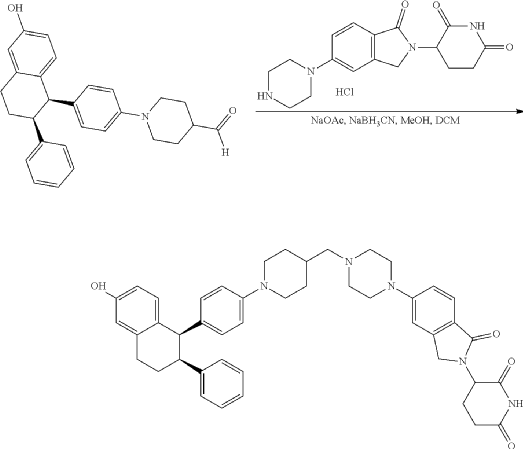
To a solution of 3-(1-oxo-5-piperazin-1-yl-isoindolin-2-yl)piperidine-2,6-dione hydrochloride (319 mg, 0.87 mmol, prepared in Step 17 described for Exemplary Compound 62) in methanol (4 mL) and dichloromethane (4 mL) was added sodium acetate (120 mg, 1.46 mmol, 2 eq). The mixture was stirred at 20° C. for 0.5 h, then to the mixture was added 1-[4-[(1R,2S)-6-hydroxy-2-phenyl-tetralin-1-yl]phenyl]piperidine-4-carbaldehyde (300 mg, 0.73 mmol, 1 eq) and sodium cyanoborohydride (137 mg, 2.19 mmol, 3 eq). The mixture was stirred at 20° C. for 12 h. LC-MS showed the starting material was consumed completely and one main peak with desired MW was detected. The reaction mixture was concentrated under reduced pressure. The residue was purified by prep-HPLC (Phenomenex luna C 18 column, 250×50 mm, 10 um; mobile phase: [water (0.05% HCl)-acetonitrile]; B %: acetonitrile 10%-40% in 30 min). The desired compound 3-[5-[4-[[1-[4-[(1R, 2S)-6-hydroxy-2-phenyl-tetralin-1-yl]phenyl]-4-piperidyl]methyl]piperazin-1-yl]-1-oxo-isoindolin-2-yl]piperidine-2,6-dione (288.4 mg, 0.37 mmol, 51% yield) was obtained as a white solid of hydrochloride salt. LC-MS (ESI) m/z: 724.4 [M+1] +; 1H NMR (400 MHz, DMSO-d 6) δ 10.97 (s, 1H), 10.83 (s, 0.9H, HCl), 7.60 (d, J=8.5 Hz, 1H), 7.40 (br s, 2H), 7.22-7.11 (m, 5H), 6.83 (d, J=6.0 Hz, 2H), 6.69-6.63 (m, 2H), 6.58-6.47 (m, 3H), 5.07 (dd, J=5.2, 13.2 Hz, 1H), 4.41-4.30 (m, 2H), 4.28-4.21 (m, 1H), 4.00 (d, J=12.7 Hz, 2H), 3.61 (d, J=11.0 Hz, 2H), 3.54-3.36 (m, 6H), 3.16 (br s, 4H), 3.06-2.84 (m, 3H), 2.76-2.53 (m, 1H), 2.43-2.33 (m, 1H), 2.27 (br s, 1H), 2.16-2.04 (m, 3H), 2.02-1.69 (m, 5H).
Synthesis of (3S)-3-[5-[4-[[1-[4-[(1R, 2S)-6-hydroxy-2-phenyl-tetralin-1-yl]phenyl]-4-piperidyl]methyl]piperazin-1-yl]-1-oxo-isoindolin-2-yl]piperidine-2,6-dione (Compound (I-c))
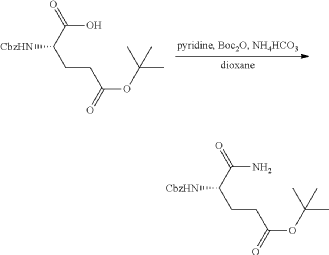
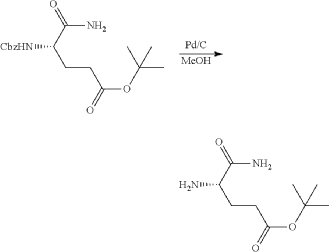
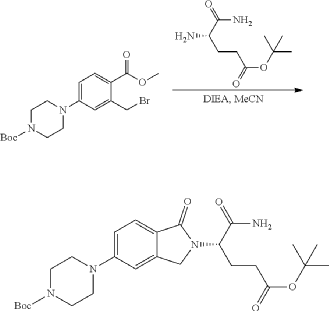
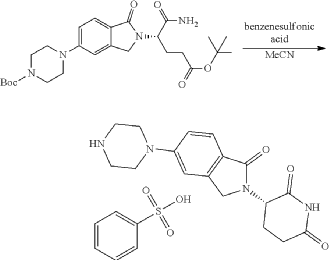
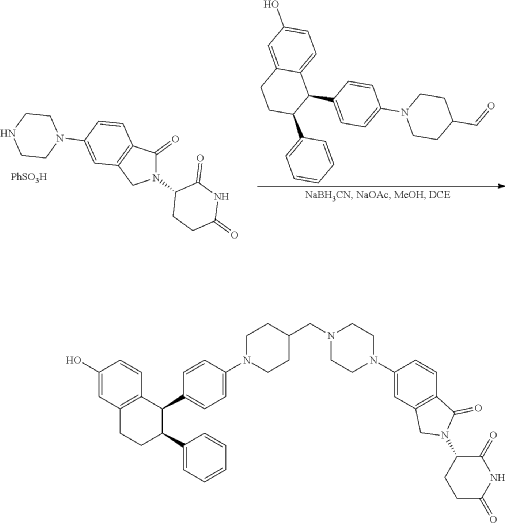
PAT
- Tetralin and tetrahydroisoquinoline derivatives as estrogen receptor degradersPublication Number: CN-118834201-APriority Date: 2016-12-01
- Tetrahydronaphthalene and tetrahydroisoquinoline derivatives as estrogen receptor degradersPublication Number: EP-3689868-B1Priority Date: 2016-12-01Grant Date: 2023-09-27
- Tetrahydronaphthalene and tetrahydroisoquinoline derivatives as estrogen receptor degradersPublication Number: US-10647698-B2Priority Date: 2016-12-01Grant Date: 2020-05-12
- Tetrahydronaphthalene and tetrahydroisoquinoline derivatives as estrogen receptor degradersPublication Number: US-2025320195-A1Priority Date: 2016-12-01
- Tetrahydronaphthalene and tetrahydroisoquinoline derivatives as estrogen receptor degradersPublication Number: US-10899742-B1Priority Date: 2016-12-01Grant Date: 2021-01-26
- Tetrahydronaphthalene and tetrahydroisoquinoline derivatives as estrogen receptor degradersPublication Number: US-11104666-B2Priority Date: 2016-12-01Grant Date: 2021-08-31
- Tetrahydronaphthalene and tetrahydroisoquinoline derivatives as estrogen receptor degradersPublication Number: US-12172981-B2Priority Date: 2016-12-01Grant Date: 2024-12-24



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
References
- Iwata, H.; Naito, Y.; Hattori, M.; Yoshimura, A.; Yonemori, K.; Aizawa, M.; et al. (November 2023). “58P Safety and pharmacokinetics (PK) of vepdegestrant in Japanese patients with estrogen receptor (ER)+/human epidermal growth factor receptor 2 (HER2)- advanced breast cancer: Results from a Japanese phase I study”. Annals of Oncology. 34: S1488–S1489. doi:10.1016/j.annonc.2023.10.193. S2CID 265657144.
- Iwata, H.; Hamilton, E.P.; Ma, C.X.; De Laurentiis, M.; Hurvitz, S.A.; Wander, S.A.; et al. (November 2023). “73TiP Global phase III studies evaluating vepdegestrant in estrogen receptor (ER)+/human epidermal growth factor receptor 2 (HER2)- advanced breast cancer: VERITAC-2 and VERITAC-3”. Annals of Oncology. 34: S1493. doi:10.1016/j.annonc.2023.10.207. S2CID 265654990.
- “Arvinas, Pfizer reworking partnership on ‘Protac’ cancer drug | BioPharma Dive”. http://www.biopharmadive.com. Retrieved 17 September 2025.
- “Estrogen Receptor”. Arvinas. Retrieved 17 September 2025.
- Sakamoto, Kathryn M.; Kim, Kwon B.; Kumagai, Ayumu; Mercurio, Frank; Crews, Craig M.; Deshaies, Raymond J. (18 January 2022). “PROTAC targeted protein degraders: the past is prologue”. Nature Reviews Drug Discovery. 21 (3): 181–200. doi:10.1038/s41573-021-00371-6. PMC 8765495. PMID 35046570.
- “Vepdegestrant (ARV-471) PROTAC ER Degrader”. MedChemExpress. Retrieved 17 September 2025.
- Hamilton, Erika P.; Ma, Cynthia; De Laurentiis, Michelino; Iwata, Hiroji; Hurvitz, Sara A.; Wander, Seth A.; et al. (2024). “VERITAC-2: a Phase III study of vepdegestrant, a PROTAC ER degrader, versus fulvestrant in ER+/HER2- advanced breast cancer”. Future Oncology (London, England). 20 (32): 2447–2455. doi:10.1080/14796694.2024.2377530. ISSN 1744-8301. PMC 11524203. PMID 39072356.
- “A Study to Compare the Efficacy and Safety of Vepdegestrant (ARV-471) Versus Fulvestrant in Participants With Estrogen Receptor-positive, HER2-negative Advanced Breast Cancer (VERITAC-2)”. ClinicalTrials.gov. 30 June 2025. Retrieved 17 September 2025.
- “Arvinas and Pfizer Announce Positive Topline Results from Phase 3 VERITAC-2 Clinical Trial”. Arvinas. Retrieved 17 September 2025.
- “VERITAC-2 Trial Shows Vepdegestrant Significantly Improves Survival in ESR1-Mutant Breast Cancer”. Applied Clinical Trials Online. 24 March 2025. Retrieved 17 September 2025.
- “Arvinas Announces Results from the VERITAC-2 Trial Selected as Late-Breaking Oral Presentation at the 2025 ASCO Annual Meeting”. Arvinas. 23 April 2025. Retrieved 17 September 2025.
- Gough, Sheryl M.; Flanagan, John J.; Teh, Jimmy (15 August 2024). “Oral Estrogen Receptor PROTAC Vepdegestrant (ARV-471) Is Highly Efficacious as Monotherapy and in Combination with CDK4/6 or PI3K/mTOR Pathway Inhibitors in Preclinical ER+ Breast Cancer Models”. Clinical Cancer Research. 30 (16): 3549–3562. doi:10.1158/1078-0432.CCR-23-3465. PMC 11325148. PMID 38819400.
- “FDA Grants Fast Track Status to Vepdegestrant for ER+/HER2– Metastatic Breast Cancer”. Oncology Live. 6 February 2024. Retrieved 17 September 2025.
- “Vepdegestrant Gains FDA Fast Track Designation in ER+/HER2- Breast Cancer”. Targeted Oncology. 6 February 2024. Retrieved 17 September 2025.
- “Arvinas Announces Submission of New Drug Application to U.S. FDA for Vepdegestrant for Patients with ESR1-Mutated ER+/HER2- Advanced or Metastatic Breast Cancer” (Press release). Arvinas. 24 June 2025. Retrieved 17 September 2025.
External links
| Clinical data | |
|---|---|
| Pronunciation | /ˌvɛpdəˈdʒɛstrənt/ VEP-də-JES-trənt |
| Other names | ARV-471 |
| Legal status | |
| Legal status | Investigational |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2229711-68-4 |
| PubChem CID | 134562533 |
| ChemSpider | 114935295 |
| UNII | WC1U3R1YMI |
| ChEMBL | ChEMBL5095210 |
| Chemical and physical data | |
| Formula | C45H49N5O4 |
| Molar mass | 723.918 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
- Targeting the Estrogen Receptor for the Treatment of Breast Cancer: Recent Advances and ChallengesPublication Name: Journal of Medicinal ChemistryPublication Date: 2023-06-28PMID: 37377342DOI: 10.1021/acs.jmedchem.3c00136
- Emerging targeted protein degradation tools for innovative drug discovery: From classical PROTACs to the novel and beyondPublication Name: European Journal of Medicinal ChemistryPublication Date: 2022-03-05PMID: 35092900DOI: 10.1016/j.ejmech.2022.114142
- Structural and Physicochemical Features of Oral PROTACsPublication Name: Journal of Medicinal ChemistryPublication Date: 2024-07-30PMID: 39078401DOI: 10.1021/acs.jmedchem.4c01017
- Discovery of the cereblon-recruiting tubulin PROTACs effective in overcoming Taxol resistance in vitro and in vivoPublication Name: European Journal of Medicinal ChemistryPublication Date: 2024-02-05PMID: 38171146DOI: 10.1016/j.ejmech.2023.116067
- Current advances and development strategies of orally bioavailable PROTACsPublication Name: European Journal of Medicinal ChemistryPublication Date: 2023-12-05PMID: 37708797DOI: 10.1016/j.ejmech.2023.115793
- Discovery of ERD-3111 as a Potent and Orally Efficacious Estrogen Receptor PROTAC Degrader with Strong Antitumor ActivityPublication Name: Journal of Medicinal ChemistryPublication Date: 2023-08-30PMID: 37647546DOI: 10.1021/acs.jmedchem.3c01186
- Expanding Chemical Probe Space: Quality Criteria for Covalent and Degrader ProbesPublication Name: Journal of Medicinal ChemistryPublication Date: 2023-07-05PMCID: PMC10388296PMID: 37403870DOI: 10.1021/acs.jmedchem.3c00550
////////////vepdegestrant, anax lab, approvals 2026, fda 2026, Veppanu, FDA 2026, APROVALS 2026, Veppanu, ARV 471, WC1U3R1YMI, PF 07850327
Dencatistat
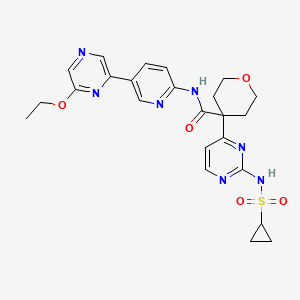
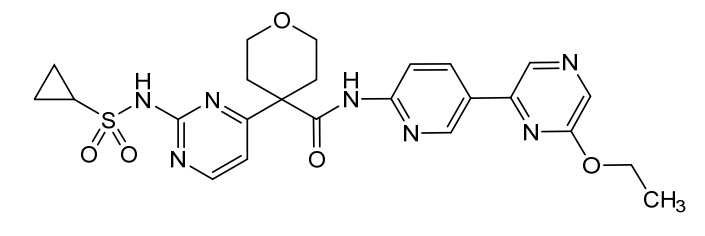
Dencatistat
CAS 2377000-84-3
MFC24H27N7O5S MW 525.6 g/mol
4-[2-(cyclopropylsulfonylamino)pyrimidin-4-yl]-N-[5-(6-ethoxypyrazin-2-yl)-2-pyridinyl]oxane-4-carboxamide
4-[2-(cyclopropanesulfonamido)pyrimidin-4-yl]-N-[5-(6-ethoxypyrazin-2-yl)pyridin-2-yl]oxane-4-carboxamide
CTP synthase 1 inhibitor, antineoplastic, STP 938, CTPS1-IN-2, QG9C9SZZ3T
Dencatistat (formerly known as STP938) is a first-in-class, orally bioavailable cancer drug designed to target specific blood cancers and solid tumours
Dencatistat is an orally bioavailable, small molecule inhibitor of cytidine triphosphate synthase 1 (CTPS1), with potential antineoplastic activity. Upon oral administration, dencatistat targets, binds to and inhibits the activity of CTPS1, thereby decreasing the production of cytidine triphosphate (CTP), an essential building block of deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). This may disrupt DNA and RNA synthesis and trigger apoptosis. CTPS1, an enzyme that catalyzes the rate-limiting step in pyrimidine synthesis, plays an important and nonredundant role in B-cell and T-cell proliferation. CTPS1 is required for rapid cell division in certain types of cancers that arise from blood cells.
Mechanism of Action
It works by inhibiting CTPS1 (Cytidine Triphosphate Synthase 1), a key enzyme that cancer cells “addicted” to for DNA synthesis.
- Targeted approach: It aims to kill cancer cells while leaving healthy cells unharmed by exploiting a “synthetic lethal” dependency in certain tumours.
- Precision medicine: It is particularly being tested in patients whose tumours lack CTPS2, a backup enzyme, which makes them highly vulnerable to dencatistat.
🏥 Clinical Status
Developed by Step Pharma, the drug is currently in several clinical trials:
- Lymphoma: Phase 1/2 trials for relapsed or refractory T-cell and B-cell lymphomas.
- Solid Tumours: Phase 1 studies for patients with solid tumours, specifically ovarian and endometrial cancers.
- Essential Thrombocythaemia: A Phase 1b trial for this blood disorder was initiated in 2025.
- Orphan Drug Status: Received FDA Orphan Drug Designation for T-cell lymphoma in May 2025.
- OriginatorStep Pharma
- ClassAnti-inflammatories; Antineoplastics; Antirheumatics; Antithrombotics; Small molecules
- Mechanism of ActionCTPS1 protein inhibitors
- Orphan Drug StatusYes – T-cell lymphoma
- Phase I/IIB-cell lymphoma; T-cell lymphoma
- Phase ISolid tumours; Thrombocytosis
- PreclinicalGraft-versus-host disease; Inflammation
- No development reportedRheumatoid arthritis
- 23 Feb 2026Step Pharma plans phase II trials for Gynaecological cancer
- 10 Feb 2026Preclinical development in Inflammation is till ongoing in France (PO) (Step Pharma pipeline, February 2026)
- 15 Oct 2025Adverse event data from a phase I/II trial in T-cell lymphoma/B-cell lymphoma released by Step Pharma
SYN
US20250177394, Compound CTPS1-IA
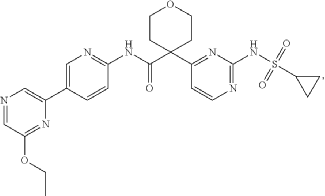
PAT
PAT
A. Preparation of Active Ingredient
20 Example A1 – Preparation of crude 4-(2-(cyclopropanesulfonamido)pyrimidin-4-yl)-N-(5- (6-ethoxypyrazin-2-yl)pyridin-2-yl)tetrahydro-2H-pyran-4-carboxamide
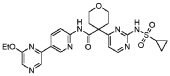
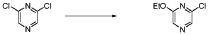
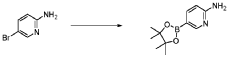
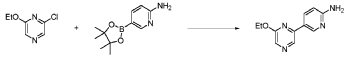
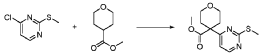
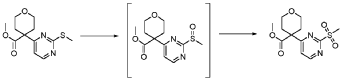
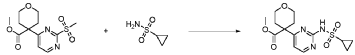
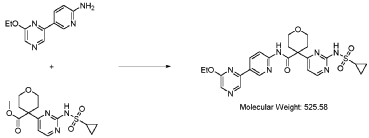
Step 4 – Preparation of crude 4-(2-(cyclopropanesulfonamido)pyrimidin-4-yl)-N-(5-(6- ethoxypyrazin-2-yl)pyridin-2-yl)tetrahydro-2H-pyran-4-carboxamide
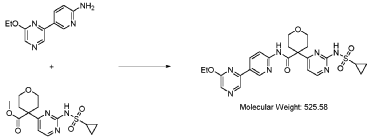
4-(2-(cyclopropanesulfonamido)pyrimidin-4-yl)tetrahydro-2H-pyran-4-carboxylate (1.76 kg, 5.15 mol, 1.00 equiv.) and 5-(6-ethoxypyrazin-2-yl)pyridin-2-amine (1.22 kg, 5.65 mol, 1.10 equiv.) were suspended in a mixture of THF (27.1 L, 15.5 rel. vol.) and DMSO (2.63 L, 1.50 rel. vol.) and stirred until the solids were evenly dispersed. The mixture was concentrated by
STP-P3718PCT
102
distillation at atmospheric pressure and approximately 70 oC to a volume of 15 L. The temperature was adjusted to 20 ± 5 oC, potassium tert-butoxide (6.92 kg 20 wt% solution in THF, 12.3 mol, 2.40 equiv.) was added over 1 h and the reaction mixture stirred at 20 ± 5 oC for 70 minutes until completion. THF (880 mL, 0.500 rel vol.) was charged, followed by acetic acid (780 5 mL, 820 g, 13.6 mol, 2.64 equiv.) over 10 minutes, followed by methanol (4.40 L, 2.50 rel. vol.), followed by water (13.2 L, 7.50 rel. vol.) over 35 minutes. The mixture was stirred at 20 ± 5 oC for 15 minutes and then 16 h at 0 ± 5 oC. The resulting suspension was filtered and washed with water (2 × 8.80 L, 2 × 5.00 rel. vol.), followed by methanol (4.40 L, 2.50 rel. vol.) The filter cake was dried at 35 oC under a flow of nitrogen for 20 h to afford crude 4-(2-10 (cyclopropanesulfonamido)pyrimidin-4-yl)-N-(5-(6-ethoxypyrazin-2-yl)pyridin-2-yl)tetrahydro- 2H-pyran-4-carboxamide (“CTPS1-IA”).
PAT
- Aminopyrimidine derivatives as CTPS1 inhibitorsPublication Number: JP-7428692-B2Priority Date: 2018-03-23Grant Date: 2024-02-06
- Aminopyrimidine derivatives as ctps1 inhibitorsPublication Number: EP-3768674-A1Priority Date: 2018-03-23
- Aminopyrimidine derivative as a CTPS1 inhibitorPublication Number: JP-2021518436-APriority Date: 2018-03-23
- Aminopyrimidine derivatives as ctps1 inhibitorsPublication Number: EP-3768674-B1Priority Date: 2018-03-23Grant Date: 2024-01-03
- Aminopyrimidine derivatives as CTPS1 inhibitorsPublication Number: CN-111868051-APriority Date: 2018-03-23
- Aminopyrimidine derivatives as CTPS1 inhibitorsPublication Number: ES-2974445-T3Priority Date: 2018-03-23Grant Date: 2024-06-27
- CompoundsPublication Number: US-2021024507-A1Priority Date: 2018-03-23
- CompoundsPublication Number: US-2021387965-A1Priority Date: 2018-10-23
- CompoundsPublication Number: US-2023192673-A1Priority Date: 2018-06-04
- Aminopyrimidine derivatives as CTPS1 inhibitorsPublication Number: CN-111868051-BPriority Date: 2018-03-23Grant Date: 2024-04-09
- Aminopyrimidine derivatives as ctps1 inhibitorsPublication Number: WO-2019180244-A1Priority Date: 2018-03-23
- Aminopyrimidine derivatives as ctps1 inhibitorsPublication Number: WO-2019179652-A1Priority Date: 2018-03-23



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
////////dencatistat, anax lab, CTP synthase 1 inhibitor, antineoplastic, STP 938, CTPS1-IN-2, QG9C9SZZ3T
Delocamten
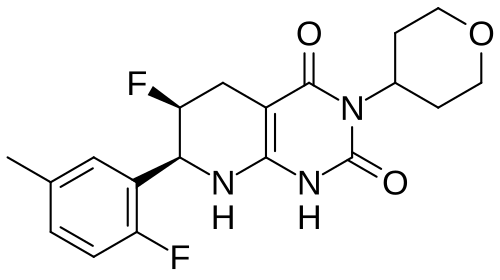
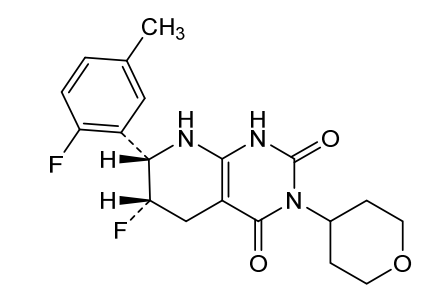
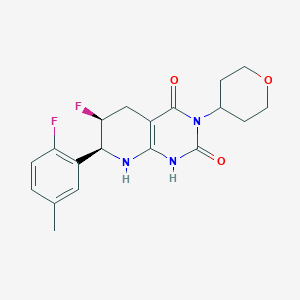
Delocamten
CAS 2417411-02-8
MFC19H21F2N3O3 MW377.4 g/mol
(6S,7S)-6-fluoro-7-(2-fluoro-5-methylphenyl)-3-(oxan-4-yl)-5,6,7,8-tetrahydro-1H-pyrido[2,3-d]pyrimidine-2,4-dione
(6S,7S)-6-fluoro-7-(2-fluoro-5-methylphenyl)-3-(oxan-4-yl)-5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2,4(1H,3H)-dione
cardiac myosin inhibitor, MYK-224; BMS-986435, MYK 224, BMS 986435, IE5886BN8T
Delocamten (development code MYK-224) is a small-molecule cardiac myosin inhibitor developed by Bristol Myers Squibb for hypertrophic cardiomyopathy.[1][2][3]
Delocamten is a small molecule drug. Delocamten is under investigation in clinical trial NCT06122779 (Study to Evaluate Safety, Tolerability and Drug Levels of BMS-986435/MYK-224 in Participants With Heart Failure With Preserved Ejection Fraction (HFpEF)). Delocamten has a monoisotopic molecular weight of 377.16 Da.
SYN
Example 1-3: Preparation of (6S,7S)-6-fluoro-7-(2-fluoro-5-methylphenyl)-3-(tetrahydro-2H-pyran-4-yl)-5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2, 4 (1H, 3H)-dione (3)
Step 7. Synthesis of (6S,7S)-6-fluoro-7-(2-fluoro-5-methylphenyl)-3-(tetrahydro-2H-pyran-4-yl)-5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2, 4 (1H, 3H)-dione (3). A mixture of crude 3G (1.0 g, 2.53 mmol) in CH 3CN (15 mL) was put into a microwave reactor with stirring at 120° C. for 30 min. Subsequently, the mixture was concentrated and the residue was purified by preparative HPLC (column: C18 silica gel; mobile phase: CH3CN:H 2O=20:80 (v v) increasing to CH3CN:H 2O=80:20 (v v) within 40 min; detector: UV 254 nm) to give compound 3 (302 mg, 32%), as a white solid, which was identified as Form 1 polymorph (see Example 2). LC-MS (ES, m/z): 378 [M+H] +; 1H NMR (300 MHz, d-DMSO): δ 10.20 (s, 1H), 7.38-7.05 (m, 3H), 6.45 (s, 1H), 5.11-4.81 (m, 3H), 3.89 (dd, J=10.8, 3.9 Hz, 2H), 3.34-3.27 (m, 3H), 2.76-2.48 (m, 4H), 2.28 (s, 3H), 1.39-1.36 (m, 2H); 19F NMR (376 MHz, d 6-DMSO): δ −123.51 (t, J=86.5 Hz), −191.57 (d, J=129.34 Hz).
PAT
Example 1-3: Preparation of (6S,7S)-6-fluoro-7-(2-fluoro-5-methylphenyl)-3- (tetrahydro-2H-pyran-4-yl)-5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2, 4 (1H, 3H)-dione (3).
Scheme 3
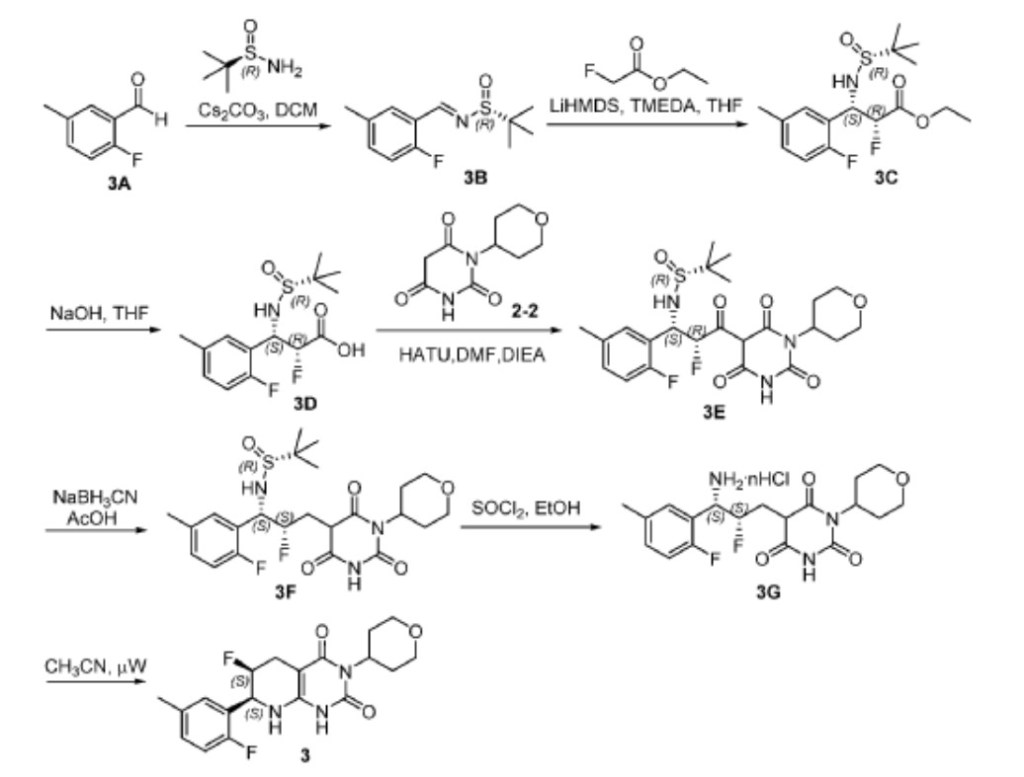
[0165] Step 1. Synthesis of (R,E)-N-(2-fluoro-5-methylbenzylidene)-2-methylpropane-2-sulfinamide (3B). The reaction mixture was filtered and the filtrate was diluted with ether (150 mL). Subsequently, the resulting suspension was filtered. The filtrate was concentrated and the residue was dried in vacuo to give 3B (8.7 g, 97%) as a yellow oil. LC-MS (ES, m/z): 242 [M+H] + ; 1 H NMR (400 MHz, d 6 -DMSO): d 8.87 (s, 1H), 7.76 (m, 1H), 7.29 (m, 1H), 7.03 (m, 1H), 2.37 (d, J = 1.0 Hz, 3H), 1.27 (s, 9H).
[0166] Step 2. Synthesis of ethyl (2R,3S)-3-(((R)-tert-butylsulfinyl)amino)-2-fluoro-3-(2-fluoro-5-methylphenyl)propanoate (3C). To a solution of 3B (4 g, 16.6 mmol), ethyl 2- fluoroacetate (2.6 g, 24.6 mmol), and TMEDA (4.8 mL) in anhydrous THF (40 mL) was added LiHMDS (1 M in THF, 24.6 mL, 24.6 mmol) dropwise at -78 o C over 30 min under an atmosphere of Ar. After stirring at -78 o C for 1 h, the reaction was quenched by adding 1 N aq.
HCl (50 mL), while maintaining the inner temperature of the mixture at < -20 o C. Subsequently, the mixture was concentrated to remove most of the organic solvent and then extracted with EtOAc (100 mL x 3). The combined organic extracts were washed with brine (100 mL) and dried over anhydrous Na2SO4. The solvent was removed and the residue was dried in vacuo to give crude 3C (6.0 g) as a yellow oil, which was used for the next step without further purification. LC-MS (ES, m/z): 348 [M+H] + .
[0167] Step 3. Synthesis of (2R,3S)-3-(((R)-tert-butylsulfinyl)amino)-2-fluoro-3-(2-fluoro-5-methylphenyl)propanoic acid (3D). To a solution of 3C (6.0 g, 17.3 mmol) in THF (40 mL) was added 1N aq. NaOH (34.6 mL, 34.6 mmol) at rt. After stirring at rt for 1 h, the reaction mixture was added ice water (50 mL). The resulting mixture was extracted with EtOAc (100 mL x 2). The aqueous layer was adjusted to pH 5 with sat. aq. citric acid, followed by extraction with EtOAc (100 mL x 3). Subsequently, the combined organic extracts were washed with brine (100 mL) and dried over anhydrous Na 2 SO 4 . The solvent was removed and the residue was purified by preparative HPLC (Column: LC-MS (ES, m/z): 320 [M+H] + ; 1 H NMR (400 MHz, d 6 -DMSO): d 13.57 (br, 1H), 7.55 (dd, J = 7.5, 2.2 Hz, 1H), 7.23– 6.94 (m, 2H), 6.04 (d, J = 10.8 Hz, 1H), 5.37– 4.86 (m, 2H), 2.29 (s, 3H), 1.12 (s, 9H).
[0168] Step 4. Synthesis of (R)-N-((1S,2R)-2-fluoro-1-(2-fluoro-5-methylphenyl)-3-oxo-3- (2,4,6-trioxo-1-(tetrahydro-2H-pyran-4-yl)hexahydropyrimidin-5-yl)propyl)-2-methylpropane-2-sulfinamide (3E). A solution of 3D (700 mg, 2.19 mmol), 2-2 (698 mg, 3.29 mmol), and HATU (1.25 g, 3.29 mmol) in DMF (10 mL) was added DIEA (849 mg, 6.57 mmol) at 0 o C under an atmosphere of Ar. aq. sodium bicarbonate (30 mL) and the resulting solution was extracted with ethyl acetate (50 mL x3). The combined organic extracts were washed with brine (50 mL x 2) and dried over anhydrous Na 2 SO 4 . The solvent was removed and the residue was dried in vacuo to give crude 3E (1.3 g) as a white solid, which was used for the next step without further purification. LC-MS (ES, m/z): 514 [M+H] + ; 1 H NMR (400 MHz, d 6 -DMSO): d 12.16 (br, 1H), 7.66– 7.45 (m, 1H), 7.23– 6.98 (m, 2H), 6.37 (m, 1H), 6.13 (d, J = 10.7 Hz, 1H), 5.22 (m, 1H), 4.79 (m, 1H), 3.94 (m, 2H), 3.35 (t, J = 11.7 Hz, 2H), 2.52– 2.39 (m, 2H), 2.29 (s, 3H), 1.49 (d, J = 12.2 Hz, 2H), 1.04 (s, 9H).
[0169] Step 5. Synthesis of (R)-N-((1S,2S)-2-fluoro-1-(2-fluoro-5-methylphenyl)-3-(2,4,6- trioxo-1-(tetrahydro-2H-pyran-4-yl)hexahydropyrimidin-5-yl)propyl)-2-methylpropane-2-sulfinamide (3F). A solution of crude 3E (1.3 g, 2.53 mmol) in AcOH (10 mL) was added NaBH3CN (398 mg, 6.33 mmol) at 0 o C under an atmosphere of Ar. After stirring at rt for 1 h, the reaction mixture was added ice water (20 mL) and the resulting solution was extracted with EtOAc (50 mL x 3). Next, the combined organic extracts were washed with brine (50 mL) and
dried over anhydrous Na2SO4. The solvent was removed and the residue was dried in vacuo to give crude 3F (1.3 g) as a white solid, which was used for the next step without further purification. LC-MS (ES, m/z): 500 [M+H] + ; 1 H NMR (400 MHz, d 6 -DMSO): d 11.31 (d, J = 28.1 Hz, 1H), 7.41 (d, J = 7.4 Hz, 1H), 7.27– 6.84 (m, 2H), 6.11– 5.78 (m, 2H), 5.08– 4.43 (m, 3H), 3.87 (m, 3H), 2.29 (s, 6H), 1.99 (s, 1H), 1.53– 1.28 (m, 2H), 1.10 (d, J = 2.1 Hz, 10H).
[0170] Step 6. Synthesis of 5-((2S,3S)-3-amino-2-fluoro-3-(2-fluoro-5-methylphenyl)propyl)-1-(tetrahydro-2H-pyran-4-yl)pyrimidine-2, 4, 6 (1H, 3H, 5H)-trione (3G). A solution of crude 3F (1.3 g, 2.60 mmol) in ethanol (10 mL) was added thionyl chloride (334 mg) at 0 o C. After stirring at rt for 1 h, the reaction mixture was concentrated and the residue was dried in vacuo to give crude 3G (1.0 g) as a white solid, which was used for the next step without further purification. LC-MS (ES, m/z): 396 [M+H] + .
[0171] Step 7. Synthesis of (6S,7S)-6-fluoro-7-(2-fluoro-5-methylphenyl)-3-(tetrahydro-2H-pyran-4-yl)-5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2, 4 (1H, 3H)-dione (3). A mixture of crude 3G (1.0 g, 2.53 mmol) in CH 3 CN (15 mL) was put into a microwave reactor with stirring at 120 o C for 30 min. Subsequently, the mixture was concentrated and the residue was purified by preparative HPLC (column: C18 silica gel; mobile phase: CH3CN:H2O = 20:80 (v/v) increasing to CH3CN:H2O = 80:20 (v/v) within 40 min; detector: UV 254 nm) to give compound 3 (302 mg, 32%), as a white solid, which was identified as Form 1 polymorph (see Example 2). LC-MS (ES, m/z): 378 [M+H] + ; 1 H NMR (300 MHz, d 6 -DMSO): d 10.20 (s, 1H), 7.38– 7.05 (m, 3H), 6.45 (s,1H), 5.11– 4.81 (m, 3H), 3.89 (dd, J = 10.8, 3.9 Hz, 2H), 3.34– 3.27 (m, 3H), 2.76–2.48 (m, 4H), 2.28 (s, 3H), 1.39–1.36 (m, 2H); 19 F NMR (376 MHz, d 6 -DMSO): d -123.51 (t, J = 86.5 Hz), -191.57 (d, J = 129.34 Hz).
PAT
- Tetrahydropyran (thp)-substituted bicyclic-pyrimidinedione compoundsPublication Number: EP-4464321-A2Priority Date: 2018-10-29
- Tetrahydropyran-substituted bicyclic pyrimidinedione compounds (THP)Publication Number: ES-2986923-T3Priority Date: 2018-10-29Grant Date: 2024-11-13
- Substituted 5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2,4-diones for treating cardiac diseasesPublication Number: US-2024025894-A1Priority Date: 2018-10-29
- Substituted 5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2,4-diones for treating cardiac diseasesPublication Number: US-12344607-B2Priority Date: 2018-10-29Grant Date: 2025-07-01
- Substituted 5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2,4-diones for treating cardiac diseasesPublication Number: US-2025282779-A1Priority Date: 2018-10-29
- Tetrahydropyran (THP) Substituted Bicyclic Pyrimidinedione CompoundsPublication Number: CN-113056465-APriority Date: 2018-10-29
- Tetrahydropyrane (THP) -substituted bicyclic pyrimidinedione compoundsPublication Number: CN-119977963-APriority Date: 2018-10-29
- Tetrahydropyran (thp)-substituted bicyclic-pyrimidinedione compoundsPublication Number: EP-3873904-B1Priority Date: 2018-10-29Grant Date: 2024-07-10
- Substituted 5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2,4-diones for treating cardiac diseasesPublication Number: US-11034693-B2Priority Date: 2018-10-29Grant Date: 2021-06-15
- Substituted 5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2,4-diones for treating cardiac diseasesPublication Number: US-2022106314-A1Priority Date: 2018-10-29
- Tetrahydropyran (THP)-Substituted Bicyclic-Pyrimidinedione CompoundsPublication Number: JP-2024063091-APriority Date: 2018-10-29
- Tetrahydropyran (thp)-substituted bicyclic-pyrimidinedione compoundsPublication Number: TW-202426449-APriority Date: 2018-10-29
- Tetrahydropyrane (THP) -substituted bicyclic pyrimidinedione compoundsPublication Number: CN-113056465-BPriority Date: 2018-10-29Grant Date: 2025-01-28
- Tetrahydropyran (thp)-substituted bicyclic-pyrimidinedione compoundsPublication Number: US-2020165247-A1Priority Date: 2018-10-29
- BICYCLIC PYRIMIDINODIONA COMPOUNDS REPLACED WITH TETRAHYDROPYRAN, POLYMORPHIC FORM OF THE SAME AND THE USE OF THE SAME FOR THE TREATMENT OF HCMPublication Number: AR-116880-A1Priority Date: 2018-10-29
- Substituted 5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2,4-diones for treating cardiac diseasesPublication Number: US-2022340569-A1Priority Date: 2018-10-29
- Tetrahydropyran (thp)-substituted bicyclic-pyrimidinedione compoundsPublication Number: EP-3873904-A1Priority Date: 2018-10-29
- CRYSTALLINE FORMS OF (6S,7S)-6-FLUORO-7-(2-FLUORO-5-METHYLPHENYL)- 3-(TETRAHYDRO-2H-PYRAN-4-YL)-5,6,7,8-TETRAHYDROPYRIDO[2,3- d]PYRIMIDINE-2,4(1H,3H)-DIONEPublication Number: WO-2024026058-A8Priority Date: 2022-07-29
- Crystalline forms of (6s,7s)-6-fluoro-7-(2-fluoro-5-methylphenyl)- 3-(tetrahydro-2h-pyran-4-yl)-5,6,7,8-tetrahydropyrido[2,3- d]pyrimidine-2,4(1h,3h)-dionePublication Number: EP-4561697-A1Priority Date: 2022-07-29
- Methods of Administering Myosin InhibitorsPublication Number: US-2023338378-A1Priority Date: 2022-04-26
- Methods of treatment with myosin modulatorPublication Number: US-2023158027-A1Priority Date: 2019-11-10
- tetrahydropyran-substituted bicyclic pyrimidinedione compounds (thp)Publication Number: BR-112021008077-A2Priority Date: 2018-10-29
- CRYSTALLINE FORMS OF (6S,7S)-6-FLUORO-7-(2-FLUORO-5-METHYLPHENYL)-3-(TETRAHYDRO-2H-PYRAN-4-YL)-5,6,7,8-TETRAHYDROPYRIDO[2,3-d]PYRIMIDINE-2,4(1H,3H)-DIONEPublication Number: US-2025034129-A1Priority Date: 2023-07-28
- CRYSTALLINE FORMS OF (6S,7S)-6-FLUORO-7-(2-FLUORO-5-METHYLPHENYL)-3-(TETRAHYDRO-2H-PYRANO-4-IL)-5,6,7,8-TETRAHYDROPYRIDE[2 ,3-D]PYRIMIDINE-2,4(1H,3H)-DIONEPublication Number: AR-130058-A1Priority Date: 2022-07-29
- Crystalline forms of (6s,7s)-6-fluoro-7-(2-fluoro-5-methylphenyl)- 3-(tetrahydro-2h-pyran-4-yl)-5,6,7,8-tetrahydropyrido[2,3- d]pyrimidine-2,4(lh,3h)-dionePublication Number: WO-2024026058-A1Priority Date: 2022-07-29
- Crystalline form of (6S,7S)-6-fluoro-7-(2-fluoro-5-methylphenyl)-3-(tetrahydro-2H-pyran-4-yl)-5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2,4(1H,3H)-dionePublication Number: CN-119855816-APriority Date: 2022-07-29
- Crystalline forms of (6s,7s)-6-fluoro-7-(2-fluoro-5-methylphenyl)-3-(tetrahydro-2h-pyran-4-yl)-5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2,4(1h,3h)-dionePublication Number: TW-202412788-APriority Date: 2022-07-29



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
References
- Lehman, Sarah J.; Crocini, Claudia; Leinwand, Leslie A. (June 2022). “Targeting the sarcomere in inherited cardiomyopathies”. Nature Reviews Cardiology. 19 (6): 353–363. doi:10.1038/s41569-022-00682-0. ISSN 1759-5010. PMC 9119933. PMID 35304599.
- Sebastian, Sneha Annie; Padda, Inderbir; Lehr, Eric J.; Johal, Gurpreet (September 2023). “Aficamten: A Breakthrough Therapy for Symptomatic Obstructive Hypertrophic Cardiomyopathy”. American Journal of Cardiovascular Drugs. 23 (5): 519–532. doi:10.1007/s40256-023-00599-0. PMID 37526885. S2CID 260348901.
- Packard, Elizabeth; de Feria, Alejandro; Peshin, Supriya; Reza, Nosheen; Owens, Anjali Tiku (December 2022). “Contemporary Therapies and Future Directions in the Management of Hypertrophic Cardiomyopathy”. Cardiology and Therapy. 11 (4): 491–507. doi:10.1007/s40119-022-00283-5. PMC 9652179. PMID 36243823.
////////delocamten, ANAX LAB, cardiac myosin inhibitor, MYK-224; BMS-986435, MYK 224, BMS 986435, IE5886BN8T
Darlifarnib
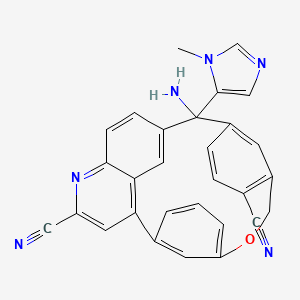
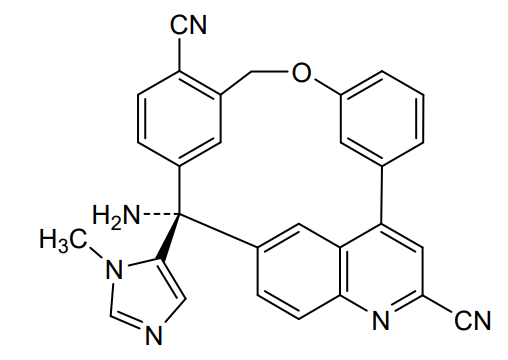
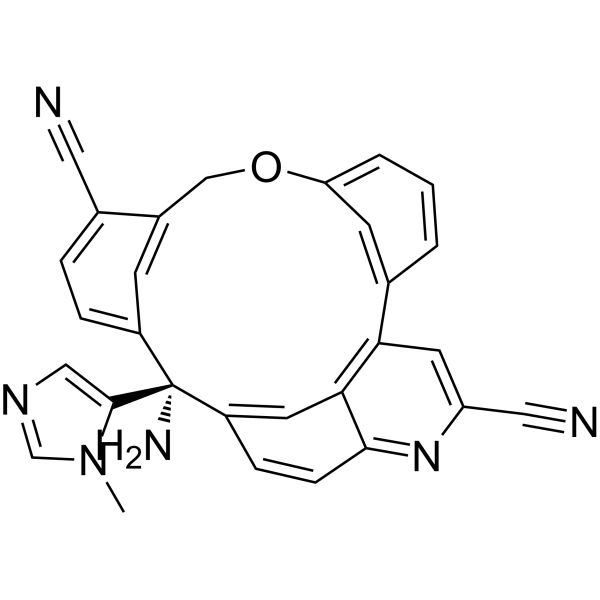
Darlifarnib
CAS 2939824-30-1
MF C29H20N6O MW 468.51
14-amino-14-(3-methylimidazol-4-yl)-7-oxa-19-azapentacyclo[13.6.2.12,6.19,13.018,22]pentacosa-1(22),2(25),3,5,9,11,13(24),15(23),16,18,20-undecaene-10,20-dicarbonitrile
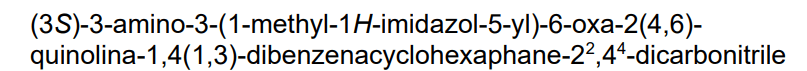
farnesyl transferase inhibitor, antineoplastic, KO-2806, KO 2806, T206317
Darlifarnib (KO-2806) is an investigational, orally active next-generation farnesyl transferase inhibitor (FTI) being developed by Kura Oncology to treat solid tumors, such as clear cell renal cell carcinoma (ccRCC). It inhibits the enzyme farnesyl transferase, blocking KRAS and mTORC1 signaling to induce tumor regression. It is often combined with other agents to overcome resistance.
Key Details About Darlifarnib
- Mechanism of Action: As a FTI, darlifarnib binds to and inhibits farnesyl transferase, which prevents the activation of RAS oncogenes and inhibits downstream mTORC1 signaling, leading to tumor cell death.
- Target Indications: Preclinical and early clinical data show potential in treating KRAS-mutant cancers, including non-small cell lung cancer (NSCLC), colorectal cancer (CRC), and clear cell renal cell carcinoma (ccRCC).
- Combination Therapy: Data from the Phase 1 FIT-001 trial (presented in April 2026) showed that combining darlifarnib with the TKI cabozantinib demonstrated robust activity in patients with pretreated, advanced ccRCC.
- Overcoming Resistance: Darlifarnib is designed to re-sensitize tumors that have become resistant to prior therapies, such as RAS inhibitors and tyrosine kinase inhibitors (TKIs).
- Status: It is an investigational drug and not yet FDA-approved.
- OriginatorKura Oncology
- ClassAntineoplastics; Small molecules
- Mechanism of ActionFarnesyltranstransferase inhibitors
- Phase IAdenocarcinoma; Colorectal cancer; Non-small cell lung cancer; Renal cell carcinoma; Solid tumours
- 12 Jan 2026Kura Oncology plans the one or more expansion cohorts of KO 2806 and cabozantinib in patients with advanced renal cell carcinoma in the first half of 2026
- 22 Oct 2025Pharmacodynamics data from a preclinical trial in Cancer presented at the AACR-NCI-EORTC International Conference on Molecular Targets and Cancer Therapeutics 2025 (AACR-NCI-EORTC-2025)
- 18 Oct 2025Adverse events and efficacy data from a phase I trial in Non-small cell lung cancer, Renal cell carcinoma, Adenocarcinoma released by Kura Oncology
PAT
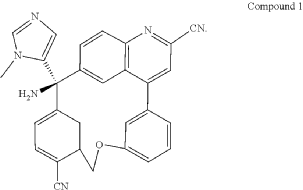
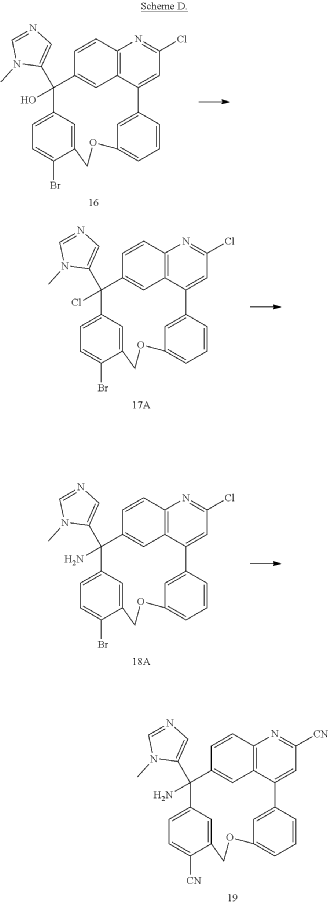
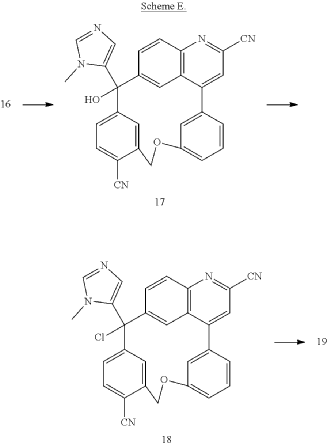
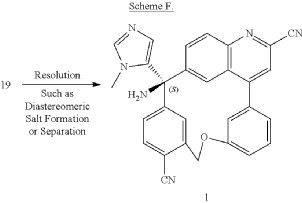
PAT
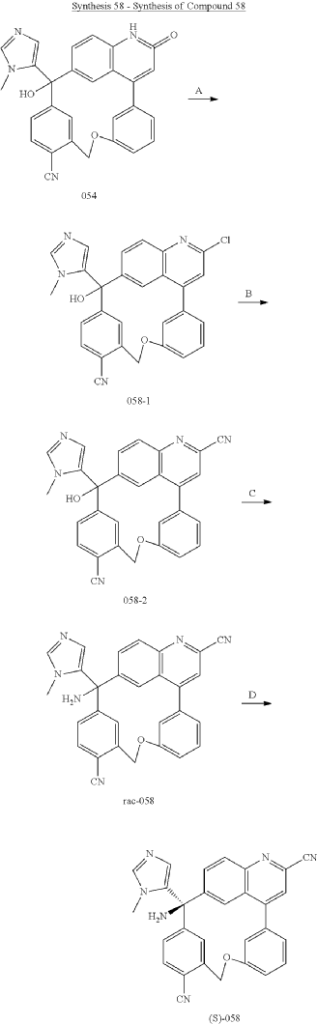
Step A: Preparation of (058-1)
Step B: Preparation of (058-2)
Step C: Preparation of (rac)-3-amino-3-(1-methyl-1H-imidazol-5-yl)-6-oxa-2(4,6)-quinolina-1,4(1,3)-dibenzenacyclohexaphane-22,44-dicarbonitrile (rac-058)
Step D: Preparation of (S)-3-amino-3-(1-methyl-1H-imidazol-5-yl)-6-oxa-2(4,6)-quinolina-1,4(1,3)-dibenzenacyclohexaphane-22,44-dicarbonitrile ((S)-058)
PAT
- Macrocyclic compounds and compositions, and methods of preparing and using the samePublication Number: US-2023322711-A1Priority Date: 2021-11-30
- Macrocyclic compounds and compositions, and methods of preparing and using the samePublication Number: US-12018011-B2Priority Date: 2021-11-30Grant Date: 2024-06-25



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
/////////////darlifarnib, ANAX LAB, farnesyl transferase inhibitor, antineoplastic, KO-2806, KO 2806, T206317
Daraxonrasib
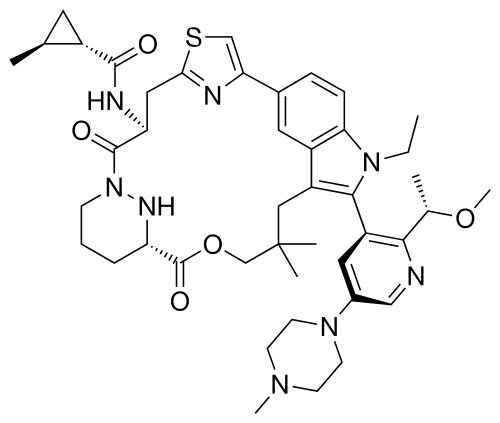
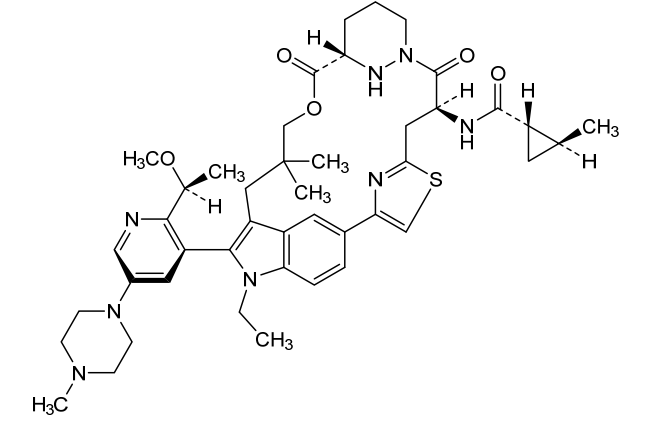
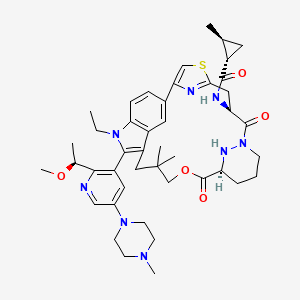
Daraxonrasib
CAS 2765081-21-6
MFC44H58N8O5S MW811.0 g/mol
trans-(1S,2S)-N-[(7S,13S)-21-ethyl-20-[2-[(1S)-1-methoxyethyl]-5-(4-methylpiperazin-1-yl)-3-pyridinyl]-17,17-dimethyl-8,14-dioxo-15-oxa-4-thia-9,21,27,28-tetrazapentacyclo[17.5.2.12,5.19,13.022,26]octacosa-1(25),2,5(28),19,22(26),23-hexaen-7-yl]-2-methylcyclopropane-1-carboxamide
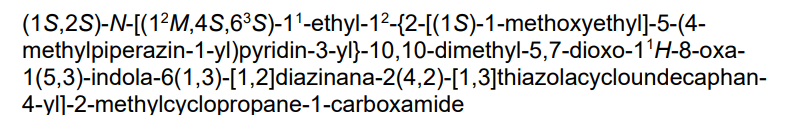
Kirsten rat sarcoma viral oncogene homolog inhibitor, antineoplastic, RMC-6236, RMC 6236, B6T47Y2UAP, RAS-IN-2,
Daraxonrasib (formerly RMC-6236) is an investigational, orally administered “molecular glue” RAS inhibitor developed by Revolution Medicines for treating advanced solid tumors with RAS mutations, particularly metastatic pancreatic cancer. April 2026 Phase 3 trials showed it significantly improves survival, demonstrating high potential as a first-line treatment.
Key Clinical Findings and Updates (as of April 2026):
- Mechanism: It acts as a RAS(ON) inhibitor, targeting mutated and wild-type RAS proteins (
) to disrupt cancer signaling.
- Breakthrough Results: Data from the RASolute 302 trial showed a substantial survival benefit in patients with previously treated metastatic pancreatic ductal adenocarcinoma (PDAC).
- High Response Rates: In trials, daraxonrasib combined with chemotherapy showed a 58% confirmed objective response rate (ORR) and 84% progression-free survival (PFS) at 6 months in untreated RAS-mutant metastatic pancreatic cancer.
- Safety Profile: Generally well-tolerated, with side effects including rash, diarrhea, stomatitis, and nausea.
- Recognition: Named the “2025 Molecule of the Year” by Drug Hunter for its, novel mechanism and clinical potential.
Daraxonrasib is currently being studied in the Phase 3 RASolute 303 trial for first-line treatment of pancreatic cancer.
Daraxonrasib (RMC-6236) is a RAS inhibitor drug. It is undergoing testing by Revolution Medicines to treat advanced solid tumors with RAS mutations, especially metastatic pancreatic ductal adenocarcinoma (PDAC) containing KRAS G12X mutations.[1] It received a breakthrough therapy designation from the U.S. Food and Drug Administration.[2]
Daraxonrasib is orally active and multi-selective RAS inhibitor. It uses a tri-complex mechanism to target the active, GTP-bound form of RAS proteins, including mutant and wild-type forms. Unlike conventional RAS inhibitors, it first binds to the chaperone-like protein cyclophilin A to form a complex, which then attaches to active RAS. This interaction blocks downstream effector binding and inhibits oncogenic signaling.[3]
In 2026, Daraxonrasib clinical trial completed a phase 3 clinical trial (RASolute 302) to assess efficacy compared to standard-of-care chemotherapy.[4] The trial met all primary and key secondary endpoints, including progression-free survival (PFS). The company reported median survival of 13.2 months with daraxonrasib vs. 6.7 months with standard chemotherapy. The hazard ratio for death was 0.40 (a 60% reduction in risk of death; p < 0.0001). Daraxonrasib was generally well tolerated with a manageable safety profile and no new safety signals.[5]
PAT
PAT
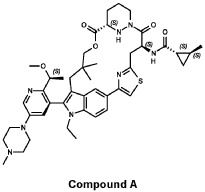
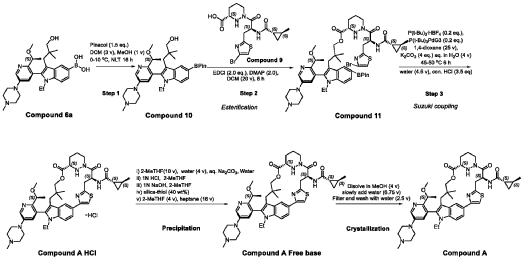
PATENT ATTORNEY DOCKET: 51432-038WO2 Part 4 – Purification of Compound A – (1S,2S)-N-[(7S,13S)-21-ethyl-20-{2-[(1S)-1- methoxyethyl]-5-(4-
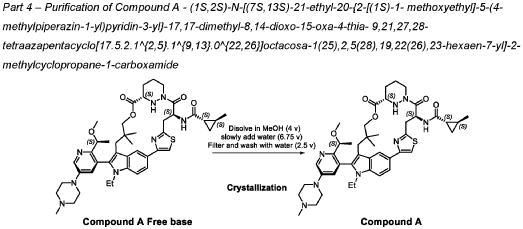
1.0equiv) at 25°C. The resulting suspension was stirred until solids were completely dissolved. The resulting methanol solution was filtered through microporous filter and transferred to another reactor. Then the reactor temperature was maintained at 25°C and slowly water (2.41kg, 1.0 V) water was added over a period of 30 minutes. The resulting cloudy solution was stirred for another 30 minutes at 25°C. Then a solution of methanol and water (3.42kg, 1:2, v/v) slowly over 1 hour. The resulting suspension was stirred for 2 hours at 25°C. Again, to the suspension additional water (2.48kg) slowly added over 1 hour. The final, suspension was stirred for additional 1 hour. Water (9.29kg, 3.75 V) was added to the suspension slowly over 2 hours and the mixture was stirred for at least for 16 hours at 25°C. The resulting suspension was filtered and washed with mixed solvent water: MeOH (3:2, v/v) twice (2x 2.2 kg), followed by water (4.91kg) washing. The wet cake was dried under reduced pressure and controlled humidity (temperature: 25 ± 5 ˚C, vacuum ≥ -0.085 MPa, humidity: 10%~20%) for 37 hours to afford Compound A as a white solid (2.68 kg, 99.4% a/a purity, 93.0% w/w assay, KF: 6.7%, 3.07 mol, 92% yield, Table 27).
PAT
- Synthesis of ras inhibitorsPublication Number: WO-2024216017-A2Priority Date: 2023-04-14
- Macrocycle compounds useful as kras inhibitorsPublication Number: WO-2024008834-A1Priority Date: 2022-07-08
- Ras inhibitorsPublication Number: US-11690915-B2Priority Date: 2020-09-15Grant Date: 2023-07-04
- Ras inhibitorsPublication Number: US-2023226186-A1Priority Date: 2020-09-15



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
References
- Cregg J, Edwards AV, Chang S, Lee BJ, Knox JE, Tomlinson AC, et al. (March 2025). “Discovery of Daraxonrasib (RMC-6236), a Potent and Orally Bioavailable RAS(ON) Multi-selective, Noncovalent Tri-complex Inhibitor for the Treatment of Patients with Multiple RAS-Addicted Cancers”. Journal of Medicinal Chemistry. 68 (6): 6064–6083. doi:10.1021/acs.jmedchem.4c02314. PMID 40056080.
- Sava J (July 1, 2025). “Daraxonrasib Earns FDA Breakthrough Status in Pancreatic Cancer”. Targeted Oncology. Retrieved October 12, 2025.
- Jiang J, Jiang L, Maldonato BJ, Wang Y, Holderfield M, Aronchik I, et al. (June 2024). “Translational and Therapeutic Evaluation of RAS-GTP Inhibition by RMC-6236 in RAS-Driven Cancers”. Cancer Discovery. 14 (6): 994–1017. doi:10.1158/2159-8290.CD-24-0027. PMC 11149917. PMID 38593348.
- Clinical trial number NCT05379985 at ClinicalTrials.gov
- Mast J (2026-04-13). “Revolution Medicines touts ‘unprecedented’ data for pancreatic cancer pill”. STAT. Retrieved 2026-04-13.
| Clinical data | |
|---|---|
| Other names | RMC-6236 |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2765081-21-6 |
| PubChem CID | 164726578 |
| IUPHAR/BPS | 13368 |
| ChemSpider | 115275938 |
| UNII | B6T47Y2UAP |
| KEGG | D13265 |
| ChEBI | CHEBI:746946 |
| Chemical and physical data | |
| Formula | C44H58N8O5S |
| Molar mass | 811.06 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
//////////daraxonrasib, anax labs, Kirsten rat sarcoma viral oncogene homolog inhibitor, antineoplastic, RMC-6236, RMC 6236, B6T47Y2UAP, RAS-IN-2,
Danifexor
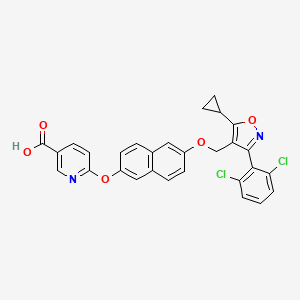
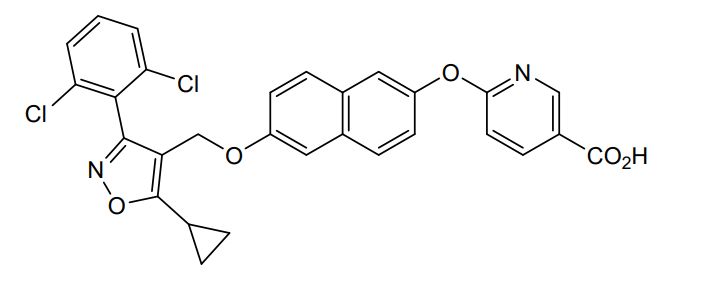
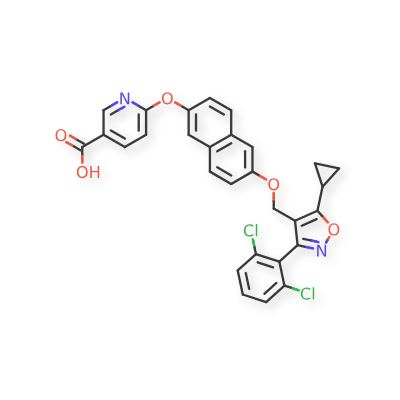
Danifexor
CAS 2648738-68-3
MF C29H20Cl2N2O5 MW547.386
6-[6-[[5-cyclopropyl-3-(2,6-dichlorophenyl)-1,2-oxazol-4-yl]methoxy]naphthalen-2-yl]oxypyridine-3-carboxylic acid
| 3-Pyridinecarboxylic acid, 6-[[6-[[5-cyclopropyl-3-(2,6-dichlorophenyl)-4-isoxazolyl]methoxy]-2-naphthalenyl]oxy]- |

farnesoid X receptor agonist, TUU8G1CX9O, HEC 96719, ASC42
Danifexor is an investigational drug that acts as a potent and selective agonist for the farnesoid X receptor (FXR). It was primarily being developed for the treatment of liver diseases such as Primary Biliary Cholangitis (PBC). ProbeChem +1
However, recent reports from April 2024 indicate that development for Danifexor has been discontinued because it was deemed non-competitive against other emerging therapies for PBC.
Key Properties and Identifiers
Danifexor is a non-steroidal molecule with specific chemical markers used in laboratory research:
- Target: Farnesoid X receptor (FXR).
Therapeutic Context
The drug was designed to target the FXR pathway, which regulates bile acid, lipid, and glucose metabolism.
- Primary Goal: Treatment of Primary Biliary Cholangitis (PBC), a chronic liver disease.
- Mechanism: As an agonist, it binds to and activates FXR to help reduce the toxic buildup of bile acids in the liver.
SYN
Example 1
Preparation of 6-((6-((5-cyclopropyl-3-(2,6-dichlorophenyl)isoxazol-4-yl) methoxy)naphthalen-2-yl)oxy)nicotinic acid (Compound 1)
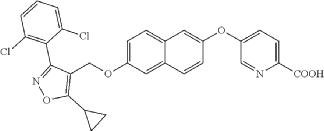
(a) Referring to the following reaction equation (Route A), Compound 1A-1 (1.0 g, 2.88 mmol, 1 eq.), Compound 1A-2 (0.46 g, 2.88 mmol, 1 eq.) and cesium carbonate (1.88 g, 5.76 mmol, 2 eq.) were dissolved in DMF (10 ml). The reaction was carried out at 65° C. for 2 h. After cooling, 10 ml water and 10 ml EA (ethyl acetate) were added for extraction, and the organic phase was washed with water and concentrated to dryness to give Compound 1A, 6-((5-cyclopropyl-3-(2,6-dichlorophenyl)isoxazol-4-yl)methoxy)naphthalen-2-ol, 0.8 g, yield: 65.0%. LCMS (ESI): calculated for C 23H 17C 12NO 3; [M+H] +: 426.1, found: 426.1.
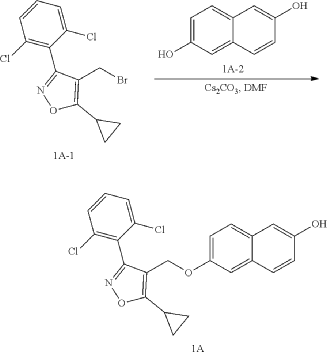
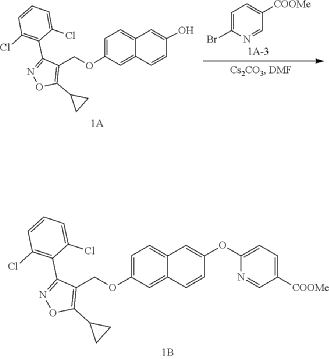
b) Referring to the following reaction equation, Compound 1A (0.2 g, 0.47 mmol, 1 eq.), 6-bromonicotinic acid methyl ester (0.1 g, 0.47 mmol, 1 eq.) and cesium carbonate (0.306 g, 0.94 mmol, 2 eq.) were dissolved in DMF (10 ml). The reaction was carried out at 65° C. for 2 h. After cooling, 10 ml water and 10 ml EA were added for extraction, and the organic phase was washed with water and concentrated to dryness to give Compound 1B, methyl 6((6((5-cyclopropyl-3-(2,6-dichlorophenyl)isoxazol-4-yl)methoxy)naphthalene-2-yl)oxy)nicotinate, 0.21 g, yield: 80.0%. LCMS (ESI): calculated for C 30H 22C 12N 2O 5; [M+H] +: 561.1, found: 561.1.
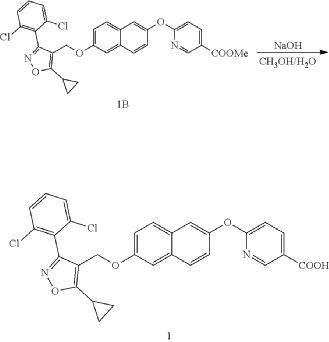
PAT
PAT
- Use and pharmaceutical composition of phenylisoxazolyl methylene-naphthalene-ether derivativesPublication Number: WO-2021109713-A1Priority Date: 2019-12-03
- Compounds for modulating activity of fxr and uses thereofPublication Number: WO-2021109712-A1Priority Date: 2019-12-03
- Uses and pharmaceutical compositions of phenylisoxazolylmethylene-naphthalene-ether derivativesPublication Number: CN-112891348-APriority Date: 2019-12-03
- Compounds that modulate FXR activity and their applicationsPublication Number: CN-112898289-APriority Date: 2019-12-03
- Compounds that modulate FXR activity and their applicationsPublication Number: CN-112898289-BPriority Date: 2019-12-03Grant Date: 2022-11-04
- Uses and pharmaceutical compositions of phenylisoxazolyl methylene-naphthalene-ether derivativesPublication Number: KR-20220101697-APriority Date: 2019-12-03
- Compounds for modulating activity of fxr and uses thereofPublication Number: EP-4073070-A1Priority Date: 2019-12-03
- Use and pharmaceutical composition of phenylisoxazolyl methylene-naphthalene-ether derivativesPublication Number: EP-4073071-A1Priority Date: 2019-12-03
- Compounds for modulating activity of fxr and uses thereofPublication Number: WO-2021108974-A1Priority Date: 2019-12-03



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
////////danifexor, ANAX LAB, farnesoid X receptor agonist, TUU8G1CX9O, HEC 96719, ASC42
Dabogratinib
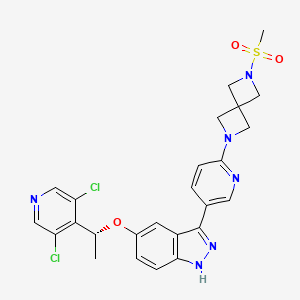
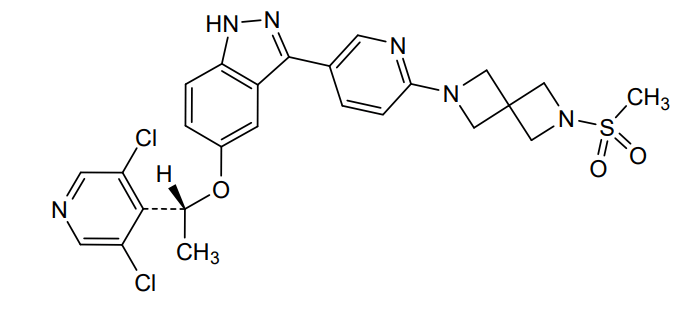
Dabogratinib
CAS 2800223-30-5
MF C25H24Cl2N6O3S, 559.5 g/mol
5-[(1R)-1-(3,5-dichloro-4-pyridinyl)ethoxy]-3-[6-(2-methylsulfonyl-2,6-diazaspiro[3.3]heptan-6-yl)-3-pyridinyl]-1H-indazole
(R)-5-(1-(3,5-Dichloropyridin-4-yl)ethoxy)-3-(6-(6-(methylsulfonyl)-2,6-diazaspiro[3.3]heptan-2-yl)pyridin-3-yl)-1H-indazole
[6-(5-{5-[(1R)-1-(3,5-dichloropyridin-4-yl)ethoxy]-1H-indazol-3-yl}pyridin-2-yl)-2,6-diazaspiro[3.3]heptan-2-yl](methyl)-λ6sulfanedioneTYRA-300
fibroblast growth factor receptor inhibitor, antineoplastic, TYRA-300, TYRA 300, A1AV2, FH245S2JZJ
Dabogratinib (TYRA-300) is an orally active, highly selective inhibitor of fibroblast growth factor receptor 3 (FGFR3), designed to treat cancers with FGFR3 alterations and genetic diseases like achondroplasia. It shows potent tumor growth inhibition in preclinical studies and early phase I/II (SURF301) clinical activity against advanced bladder cancer and metastatic urothelial carcinoma.
Key Aspects of Dabogratinib (TYRA-300)
- Mechanism: It acts as a selective inhibitor of FGFR3 with a high selectivity over other isoforms (FGFR1/2/4), which helps minimize toxicity.
- Target Indications: It is being developed for FGFR3-mutant cancers, including non-muscle invasive bladder cancer (NMIBC) and metastatic urothelial carcinoma, as well as pediatric achondroplasia.
- Preclinical Performance: Studies showed that it reduces tumor growth and drives tumor regression, especially in xenograft models with FGFR3-activating mutations (e.g., S249C).
- Clinical Trials:
- SURF301 (Phase I/II): Ongoing study, Tyra Biosciences reported early efficacy in patients with advanced metastatic urothelial carcinoma (mUC) harboring FGFR3 mutations/fusions.
- SURF302 (Phase II): Evaluating the drug in patients with FGFR3-altered, low-grade, intermediate-risk non–muscle invasive bladder cancer (NMIBC).
- BEACH301 (Phase II): Studying the drug in children with achondroplasia, as it is designed to increase long-bone growth.
- Properties: It is an orally bioavailable molecule with an IC50 of
for FGFR3.
Dabogratinib is an orally bioavailable, selective inhibitor of human fibroblast growth factor receptor 3 (FGFR3), with potential antineoplastic activity. Upon oral administration, dabogratinib specifically targets and binds to certain FGFR3 activating gene alterations, and specifically the gatekeeper mutants V555L/M. This blocks FGFR3-mediated signaling and leads to an inhibition of tumor cell proliferation in FGFR3-overexpressing cells. FGFR3, a receptor tyrosine kinase, is involved in angiogenesis and in the proliferation, differentiation, and survival of tumor cells. FGFR3 expression is associated with poor prognosis. It is overexpressed by certain tumor cell types.
- Efficacy and Safety of TYRA-300 in Participants With FGFR3 Altered Low Grade, Intermediate Risk Non-Muscle Invasive Bladder CancerCTID: NCT06995677Phase: Phase 2Status: RecruitingDate: 2026-04-09
- A Study of TYRA-300 in Children With Achondroplasia: BEACH301CTID: NCT06842355Phase: Phase 2Status: RecruitingDate: 2026-03-06
- Safety and Preliminary Anti-Tumor Activity of TYRA-300 in Advanced Urothelial Carcinoma and Other Solid Tumors With FGFR3 Gene AlterationsCTID: NCT05544552Phase: Phase 1/Phase 2Status: Active, not recruitingDate: 2026-01-12
PAT
Example 46. 5-[(1R)-1-(3,5-dichloro-4-pyridyl)ethoxy]-3-[6-(2-methylsulfonyl-2,6-diazaspiro[3.3]heptan-6-yl)-3-pyridyl]-1H-indazole
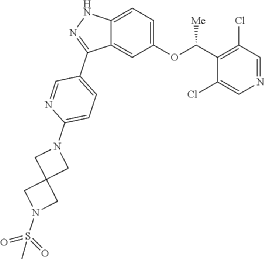
(5-[(1R)-1-(3,5-dichloro-4-pyridyl)ethoxy]-3-[6-(2-methylsulfonyl-2,6-diazaspiro[3.3]heptan-6-yl)-3-pyridyl]-1H-indazole. Triethylamine (20.5 uL, 0.148 mmol, 1.2 equiv) and methylsulfonyl chloride (9.5 uL, 0.123 mmol, 1.0 equiv) were sequentially added at room temperature to a solution of example 45 (59.0 mg, 0.123 mmol, 1 equiv) in anhydrous THE (3 mL). After stirring for 2 hours, the reaction mixture was concentrated under reduced pressure and diluted with saturated brine (30 mL) and dichloromethane (30 mL). The layers were separated. The organic layer was dried over sodium sulfate, filtered and concentrated under reduced pressure on to Celite (1 g). The product was purified on an Interchim automated chromatography system (RediSep Rf Gold HP C18, 15.5 g cartridge), eluting with a gradient of 0 to 100% acetonitrile in water. The fractions containing product were collected and lyophilized to give a white solid (45.0 mg, 65% yield). Analysis: LCMS: m/z=559.2 (M+H); 1H NMR (400 MHz, DMSO-d6) δ 13.02 (br s, 1H), 8.59 (s, 2H), 8.52 (dd, J=0.6, 2.2 Hz, 1H), 7.87 (dd, J=2.4, 8.6 Hz, 1H), 7.46 (d, J=8.9 Hz, 1H), 7.16 (d, J=2.1 Hz, 1H), 7.09 (dd, J=2.3, 9.0 Hz, 1H), 6.54 (dd, J=0.4, 8.6 Hz, 1H), 6.10 (q, J=6.6 Hz, 1H), 4.17 (s, 4H), 4.12 (s, 4H), 3.03 (s, 3H), 1.76 (d, J=6.6 Hz, 3H).
PAT
- Indazole compoundsPublication Number: TW-202241906-APriority Date: 2020-12-30
- Indazole compounds as kinase inhibitorsPublication Number: EP-4271673-A1Priority Date: 2020-12-30
- Indazole compounds as kinase inhibitorsPublication Number: WO-2022147246-A1Priority Date: 2020-12-30
- Indazole compounds as kinase inhibitorsPublication Number: US-12264149-B2Priority Date: 2020-12-30Grant Date: 2025-04-01
- Polymorphic compounds and uses thereofPublication Number: EP-4547670-A1Priority Date: 2022-06-29
- Indazole compounds as kinase inhibitorsPublication Number: US-12071428-B2Priority Date: 2020-12-30Grant Date: 2024-08-27
- Indazole Compounds as Kinase InhibitorsPublication Number: KR-20230152654-APriority Date: 2020-12-30
- Indazole compounds as kinase inhibitorsPublication Number: US-2024109865-A1Priority Date: 2020-12-30
- Indazole compounds as kinase inhibitorsPublication Number: US-2024208941-A1Priority Date: 2020-12-30
- Tyra-300 (5-[(1r)-1-(3,5-dichloro-4-pyridyl)ethoxy]-3-[6-(2-methylsulfonyl-2,6-diazaspiro[3.3]heptan-6-yl)-3-pyridyl]-1h-indazole ) in combination with a pd-1 or pd-l1 antagonist for use in the treatment of cancerPublication Number: WO-2025064744-A1Priority Date: 2023-09-22
- Fgfr inhibitors and methods of use thereofPublication Number: WO-2025061029-A1Priority Date: 2023-09-18
- Polymorphic compounds and uses thereofPublication Number: AU-2023300357-A1Priority Date: 2022-06-29
- Polymorphic compounds and uses thereofPublication Number: WO-2024006883-A1Priority Date: 2022-06-29
- Polymorphic compounds and uses thereofPublication Number: TW-202408493-APriority Date: 2022-06-29
ADVT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
Publication Name: Journal of Medicinal Chemistry
Publication Date: 2024-09-11
PMID: 39258897
DOI: 10.1021/acs.jmedchem.4c01531
////////dabogratinib, anax lab, fibroblast growth factor receptor inhibitor, antineoplastic, TYRA-300, TYRA 300, A1AV2, FH245S2JZJ

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










