| FDA Orange Book Patents: 1 of 1 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 9549999 |
| Expiration | Mar 10, 2030 |
| Applicant | GE HEALTHCARE |
| Drug Application |
|
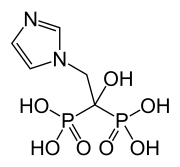
Efonidipine, エホニジピン
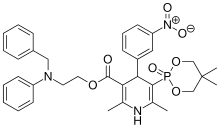
Efonidipine
- Molecular FormulaC34H38N3O7P
- Average mass631.655 Da
- エホニジピン
- CAS 111011-63-3; FREE FORM
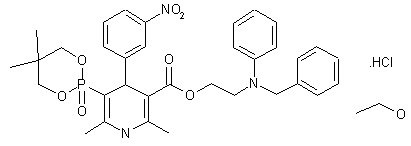
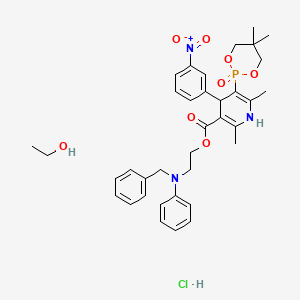
| Molecular Formula: | C36H45ClN3O8P |
|---|---|
| Molecular Weight: | 714.193 g/mol |
LD50:> 5 g/kg (R, p.o.)
- Synonyms:NZ-105
- ATC:C08CA
Efonidipine Hydrochloride Ethanolate Bulk & Tablets 10 mg/20mg/40mg, 

Efonidipine (INN) is a dihydropyridine calcium channel blocker marketed by Shionogi & Co. of Japan. It was launched in 1995, under the brand name Landel (ランデル). The drug blocks both T-type and L-type calcium channels.[1] Drug Controller General of India (DCGI) granted approval to M/s. Zuventus pharma Ltd for marketing efonidipine under brand name Efnocar in India .[2]
Structure Activity Relationship
Efonidipine is a dual Calcium Channel Blocker (L & T-type). It has a unique chemical structure. The phosphonate moiety (Figure 1) at the C5 position of the dihydropyridine ring is considered to be important for the characteristic pharmacological profile of the drug. (figure-1)
Mechanism of action
Efonidipine, a new generation dihydropyridine (DHP) calcium channel blocker, inhibits both L-type and T-type calcium channels.[1]
Pharmacodynamics
- Efonidipine exhibits antihypertensive effect through vasodilatation by blocking L-type and T-type calcium channels.[1]
- Efonidipine has a negative chronotropic effect. Working on sino atrial node cells by inhibiting T-type calcium channel activation, Efonidipine prolongs the late phase-4 depolarization of the sino atrial node action potential and suppresses an elevated HR. The negative chronotropic effect of Efonidipine decreases heart rate, myocardial oxygen demand and increases coronary blood flow.[3]
- Efonidipine increases coronary blood flow by blocking L & T-type calcium channels and attenuates myocardial ischaemia.[4]
- By reducing synthesis and secretion of aldosterone, Efonidipine prevents hypertrophy and remodeling of cardiac myocytes.[5]
- Efonidipine increases glomerular filtration rate without increasing intra-glomerular pressure and filtration fraction. This prevents hypertension induced renal damage.[6]
- Efonidipine prevents Rho-kinase and NFB induced renal parenchymal fibrosis and provides long term renal protection.[7][8]
- Efonidipine suppresses renin secretion from the juxta glomerular apparatus in the kidneys.[9]
- Efonidipine enhances sodium excretion from the kidneys by suppressing aldosterone synthesis and secretion from the adrenal glands. Aldosterone induced renal parenchymal fibrosis is suppressed by Efonidipine.[5]
- Efonidipine prevents NFB induced hypertrophy and inflammation in the renal vasculature and protects the kidneys.[7]
- Efonidipine protects against endothelial dysfunction due to its anti-oxidant activity and by restoring NO bioavailability.[10][11]
- Efonidipine has anti-atherogenic activity and protects the blood vessels from atherosclerosis.[12]
- Efonidipine lowers blood pressure in cerebral resistance vessels and prevents hypertension induced brain damage.[4]
Pharmacokinetics
Absorption
Peak plasma concentration is achieved in about 1.5 to 3.67 hours after administration. Half life is approximately 4 hours. The pharmacokinetic parameters of Efonidipine are depicted in Table-1.
Table 1: PK Parameters in Adult Healthy Male Subjects
| Variable | Efonidipine | |
| Mean | Range | |
| Cmax(ng/ml) | 36.25 | 9.66-66.91 |
| Tmax (hour) | 2.59 | 1.50-3.67 |
| T1/2 (hour) | 4.18 | 2.15-6.85 |
*Data on file
Long Duration of Action
Efonidipine has a slow onset and a long duration of action. This unique characteristic of Efonidipine is because of the following reasons:[13]
- High lipophilicity of Efonidipine allows it to enter the phospholipid rich cell membrane and access the dihydropyridine binding site of the Ca2+ channels.
- Tight binding to the dihydropyridine receptors.
- The dissociation constant of Efonidipine from dihydropyridine receptors is very low (0.0042/min/nM), signifying very slow dissociation from the receptors. This explains the long duration of action of Efonidipine.
Metabolism
Efonidipine is primarily metabolized in the liver. The important metabolites are N-dephenylated Efonidipine (DPH), deaminated Efonidipine (AL) and N-debenzylated Efonidipine (DBZ). DBZ and DPH exhibit activity as calcium antagonists. The vasodilating properties of DBZ and DPH were about two-thirds and one-third respectively than that of the parent compound. Results suggest that the majority of the pharmacological effect after oral dosing of Efonidipine hydrochloride in man is due to unchanged compound and its metabolites make a small contribution to the pharmacological effect.[14]
Elimination
Biliary route is the main pathway of excretion. No significant amount of unchanged drug was excreted in urine. In the urine collected for 24 h after an oral dosing, 1.1 % of the dose was excreted as deaminated Efonidipine, and 0.5% as a pyridine analogue of deaminated Efonidipine.
Indications
- Essential hypertension and renal parenchymal hypertension
- Angina
Dosage and Administration
- Essential hypertension and renal parenchymal hypertension: 20-40 mg orally once daily. A dose of up to 80mg/day is seen to be safe and effective in clinical trials.[15][16]
- Angina: 40 mg/day.
Contraindications
- Contraindicated in patients hypersensitive to Efonidipine or any of the excipients
- It is also contraindicated in pregnancy and lactation.
Precautions
- Should be administered with caution in patients with hepatic impairment
- Dose adjustment may be required in elderly as hypotension can occur
- Efonidipine may worsen clinical condition in patients with sinus bradycardia, sinus arrest or sinus node dysfunction
- As dizziness can occur due to hypotensive action, one should be careful while operating machines, with aerial work platforms and driving of a motor vehicle
- Drug should not be stopped abruptly. Discontinuation should be gradual and under supervision of a qualified physician
Drug Interactions
- Other anti-hypertensive agents: Efonidipine enhances the antihypertensive action additively and may produce hypotension and shock. Blood pressure should be monitored regularly to adjust dose of concomitant drugs.
- Cimetidine: Cimetidine inhibits CYP450 enzymes involved in metabolism of CCBs. Blood concentration of calcium channel antagonists increase leading to higher incidence of side effects (hot flushes).
- Grape fruit juice: Grapefruit juice suppresses enzymes metabolizing calcium channel antagonists (cytochrome P450) and reduces the clearance. Thus, there is a possibility that blood concentration of the drug may increase and the anti-hypertensive effect is enhanced.
- Tacrolimus: Efonidipine inhibits metabolic enzymes involved in Tacrolimus metabolism and reduces its clearance. So, increase in blood concentration of Tacrolimus can occur.
Adverse Drug Reactions
The common side effects are hot flushes, facial flushing and headache. In addition, elevation in serum total cholesterol, ALT (SGPT), AST (SGOT) and BUN may occur. Frequent urination, pedal edema, increased triglycerides occurs in less than 0.1%.[17]
Lesser incidence of pedal edema (< 0.1%)
One common adverse effect of the L-type Ca2+ channel blockers like Amlodipine is vasodilatory Pedal edema. Combined L-/T-type Ca2+ channel blockers, such as Efonidipine, display antihypertensive efficacy similar to their predecessors (Amlodipine) with much less propensity of pedal edema formation. Efonidipine equalizes the hydrostatic pressure across the capillary bed through equal arteriolar and venular dilatation, thus reducing vasodilatory edema. These incremental microcirculatory benefits of efonidipine over the conventional L-type Ca2+ channel blockers (Amlodipine) are likely attributed to their additional T-type Ca2+ channel blocking properties and the increased presence of T-type Ca2+channels in the microvasculature (e.g. arterioles, capillaries, venules etc).[18]
Among the CCBs, Efonidipine (<0.1%)[17] has lowest incidence of pedal edema compared to amlodipine ( 5-16%)[19], cilnidipine (5%)[20], benidipine (5%)[21] and azelnidipine (15.5%).[22]
Use in Special Population
Administration to Elderly
The drug should be started at low dose (20 mg/day) in elderly. Patient should be carefully observed for development of hypo-tension. Dose may be halved if there is intolerance to the 20 mg/day dosage regimen.
Pregnancy and Lactation
The drug should not be administered to pregnant women and women suspected of being pregnant. Administration to lactating women should be avoided unless benefit significantly surpasses the risk to the child. Mothers on Efonidipine treatment should avoid breast feeding.
Pediatric Use
Safety of Efonidipine in low birth weight infants, newborns, infants and children has not been established.
Efonidipine-The Best in Class
Efonidipine is unique among clinically available CCBs. Its antihypertensive efficacy is superior or at par with other CCBs. But, in terms of pleiotropic effects leading to enhanced cerebral, cardiac and renal protection, Efonidipine scores over the other CCBs.
Advantages over Amlodipine
1. Better renoprotection by:
- Dual channel blockade [1]
- Prevention of Rho-kinase and NFkB induced tubulointerstitial fibrosis[23][24]
- Reduction of synthesis and secretion of aldosterone from the adrenal cortex[25]
2. Preferred in angina with hypertension due to negative chronotropic action[26]
3. Better control of reflex tachycardia[3]
4. Reduces cardiac remodelling, arterial stiffness and prevents atherogenesis[27]
5. More useful in patients with diabetes & nephropathy[28]
6. Better protection against cardiac hypertrophy by significant reduction in LVMI[29]
7. Less adverse effects compared to Amlodipine[30]
8. Reduces endothelial dysfunction and oxidative stress(anti-oxidant property)[10]
Advantages over Cilnidipine
1. Strong negative chronotropic effect (less tachycardia) compared to Cilnidipine[3]
2. Significant improvement in exercise tolerance.[31]Better choice in hypertensive patients with angina.
3. Better BP control by marked urinary Na+ excretion[32]
4. Better renoprotection by:
- a. Suppression of plasma renin release[33]
- b. Prevention of Rho-kinase and NFkB induced tubulointerstitial fibrosis[34][35]
- c. Reduction of synthesis and secretion of aldosterone from the adrenal cortex[5]
5. Better choice in diabetic hypertensives[36]
6. Prevents cardiac remodelling by suppression of aldosterone secretion[5]
7. Superior anti-oxidant activity[10]
8. Less adverse effects compared to Cilnidipine[30]
Advantages over Benidipine
L & T-type CCBs have invoked a lot of interest in the management of hypertension because of their unique pharmacological profile. Several novel agents have been developed including Azelnidipine, Barnidipine, Benidipine, Efonidipine, Manidipine and Nilvadipine. Among all the agents, Efonidipine has emerged as the best among its peers. The advantages of Efonidipine over Benidipine are summarized below.
1. More selective blockade of T-type calcium channels [37][38]
2. More balanced renal arteriolar dilatation than benidipine[37][38]
3. Superior anti-proteinuric effect [15]
4. Greater reduction of serum aldosterone [39]
5. Renoprotection by reducing plasma renin unlike Benidipine [39]
6. Greater negative chronotropic effect
7. Efonidipine has anti-platelet activity[12]
8. Efonidipine reduces Insulin Resistance [40]
9. Significantly lower incidence of pedal edema & constipation compared to Benidipine
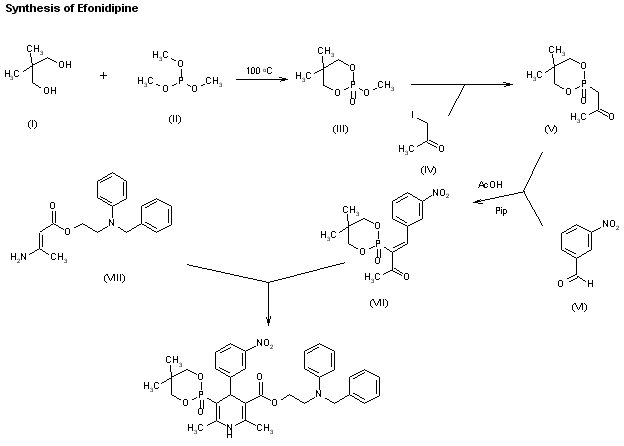
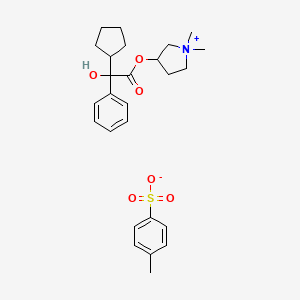
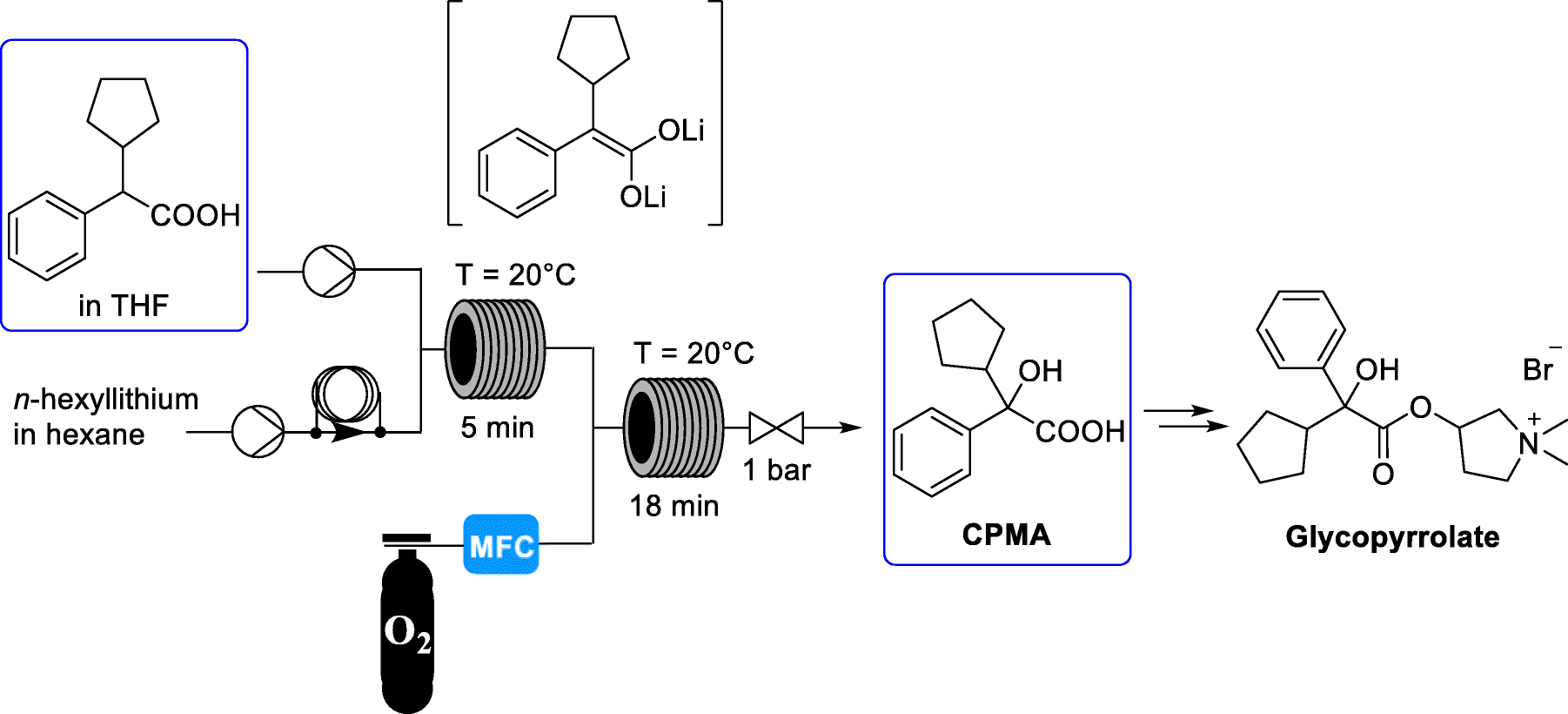
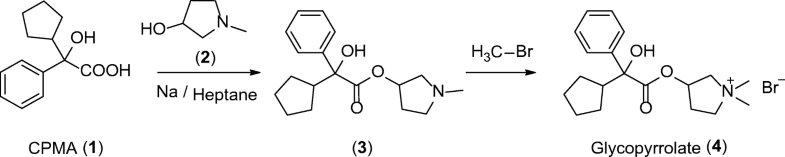
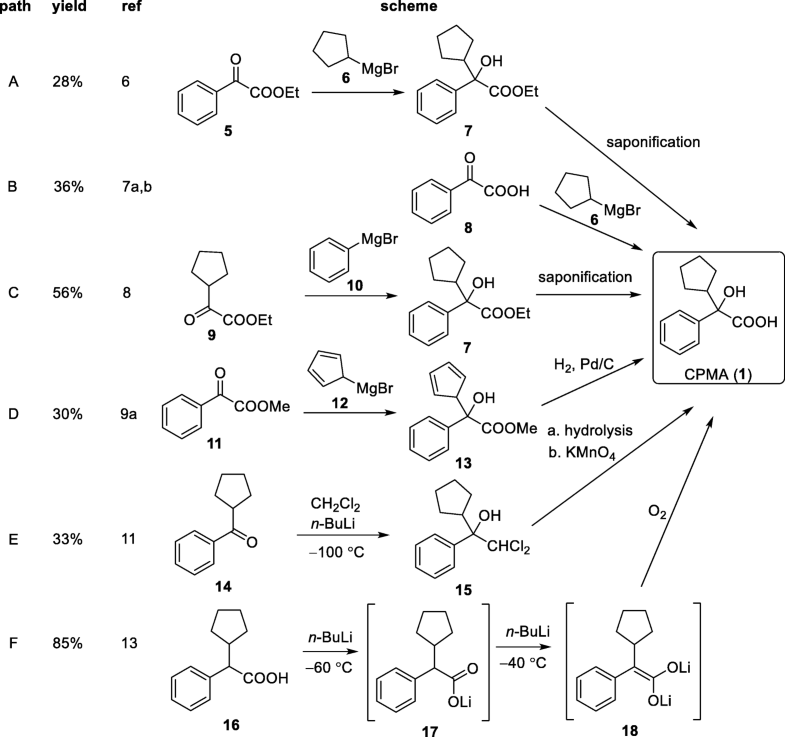
A new synthesis of efonidipine has been described: The cyclization of 2,2-dimethylbutane-1,4-diol (I) with triethyl phosphite (II) by heating at 100 C gives 2-methoxy-5,5-dimethyl-1,3,2-dioxaphosphorinan (III), which, by treatment with iodoacetone (IV) in refluxing ether, yields 2-acetonyl-5,5-dimethyl-1,3,2-dioxaphosphorinan-2-one (V). The condensation of (V) with 3-nitrobenzaldehyde (VI) by means of piperidine in acetic acid affords 3-(5,5-dimethyl-2-oxo-1,3,2-dioxaphosphorinan-2-yl)-4-(3-nitrophenyl)-3-buten-2-one (VII), which is finally cyclized with 3-amino-2-propenoic acid 2-(N-benzyl-N-phenylamino)ethyl ester (VIII) in refluxing toluene.ReferencesChem Pharm Bull 1992,40(9),2362
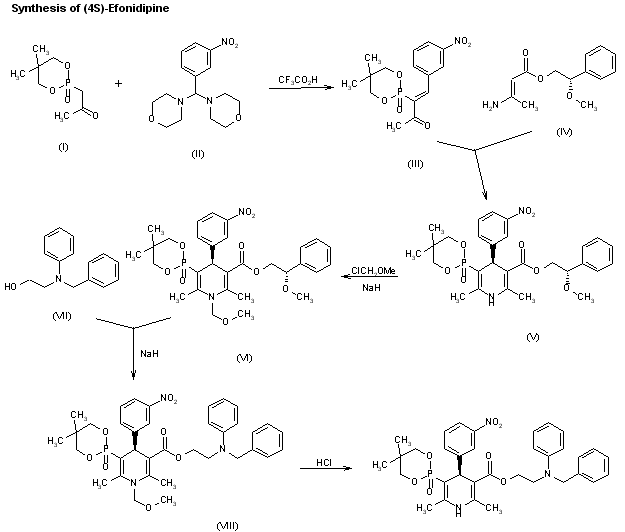
A new synthesis for (4S)-efonidipine has been described: The reaction of 5,5-dimethyl-2-(2-oxopropyl)-1,3,2-dioxaphosphorinan-2-one (I) with dimorpholino(3-nitrophenyl)methane (II) by means of trifluoroacetic acid in hot toluene gives 5,5-dimethyl-2-[1-acetyl-2-(3-nitrophenyl)vinyl]-1,3,2-dioxaphosphorina n-2-one (III), which is cyclized with 3-aminocrotonic acid 2(S)-methoxy-2-phenylethyl ester (IV) in refluxing toluene; the recrystallization of the resulting product affords 5-(5,5-dimethyl-2-oxo-1,3,2-dioxaphosphorinan-2-yl)-2,6-dimethyl-4(S)-(3-nitrophenyl)-1,4-dihydropyridine-3-carboxylic acid 2(S)-methoxy-2-phenylethyl ester (V). The protection of the NH group of (V) with chloromethyl methyl ether and NaH in THF yields the N-methoxymethyl derivative (VI), which is transesterified with 2-(N-benzyl-N-methylamino)ethanol (VII) and NaH in DMSO, giving the protected final product (VIII). Finally, this compound is deprotected with HCl in ethanol.
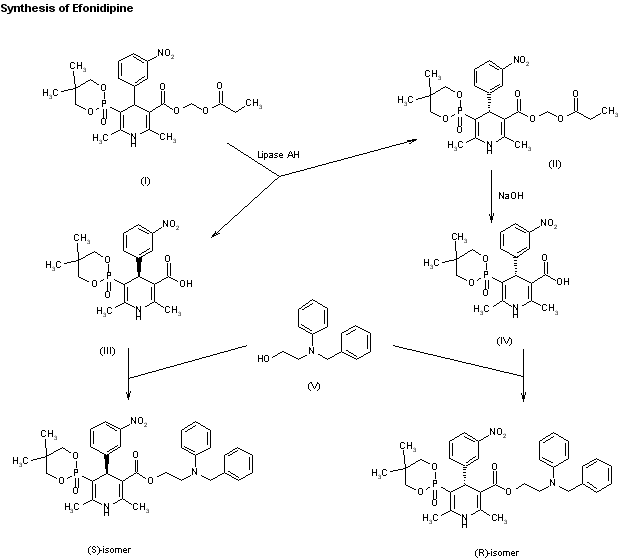
An enantioselective synthesis of efonidipine has been described: The enantioselective hydrolysis of 5-(5,5-dimethyl-2-oxo-1,3,2-dioxaphosphorinan-2-yl)-2,6-dimethyl-4-(3-n itrophenyl)-1,4-dihydropyridine-3-carboxylic acid propionyloxymethyl ester (I) with lipase AH in 2,5-dimethyltetrahydrofuran saturated with water gives the corresponding free acid of the (S)-isomer (III), while the propionyloxymethyl ester of the (R)-isomer (II) remains undisturbed. After chromatographic separation, the (R)-ester (II) is hydrolyzed with NaOH in methanol to the (R)-acid (IV). Finally, both enantiomerically pure acids (III) and (IV) are separately esterified with 2-(N-benzyl-N-phenylamino)ethanol in the usual way
CLIP
PAPER
Synthesis of 1,4-dihydropyridine-5-phosphonates and their calcium antagonistic and antihypertensive activities: Novel calcium-antagonist 2-[benzyl(phenyl)amino]ethyl 5-(5,5-dimethyl-2-oxo-1,3,2-dioxaphosphorinan-2-yl)-1,4-dihydro-2, 6-dimethyl-4-(3-nitrophenyl)-3-pyridinecarboxylate hydrochloride ethanol (NZ-105) and its crystal structure
Chem Pharm Bull 1992, 40(9): 2362
PATENT
IN 201501586
http://ipindiaservices.gov.in/PatentSearch/PatentSearch/ViewPDF
- ^ Jump up to:a b c d Tanaka H, Shigenobu K (2002). “Efonidipine hydrochloride: a dual blocker of L- and T-type Ca2+ channels”. Cardiovasc. Drug Rev. 20 (1): 81–92. PMID 12070536.
- Jump up^http://www.cdsco.nic.in/writereaddata/Minutes%20of%2034th%20SEC%20Cardiovascular%20&%20Renal%2008_11_2016.pdf
- ^ Jump up to:a b c Masumiya H, Shijuku T, Tanaka H, Shigenobu K. Inhibition of myo¬cardial L- and T-type Ca2+ currents by efonidipine: possible mecha¬nism for its chronotropic effect. Eur J Pharmacol. 1998; 349: 351-7.
- ^ Jump up to:a b Masuda Y, Tanaka S. Efonidipine Hydrochloride: A New Calcium Antagonist. Cardiovascular Drug Reviews. 1994; 12 ( 2): 123-135.
- ^ Jump up to:a b c d Ikeda K, Isaka T, Fujioka K, Manome Y, Tojo K. Suppression of Aldosterone Synthesis and Secretion by Ca2+ Channel Antagonists. International Journal of Endocrinology. 2012.
- Jump up^ Hayashi K, et al. T-Type Ca Channel Blockade as a Determinant of Kidney Protection. Keio J Med. 2010; 59(3): 84-95
- ^ Jump up to:a b Hayashi M, Yamaji Y, Nakazato Y, Saruta T. The effects of calcium channel blockers on nuclear factor kappa B activation in the mesangial cells. Hypertens Res. 2000;23:521–525.
- Jump up^ Sugano N, Sugano N, Wakino S, Tatematsu S, Homma K, Yoshioka K, Hasegawa K, Utsunomiya Y, Tokudome G, Hosoya T, Saruta T, Hayashi K. Role of T-type Ca2 channels (TCCs) as a determinant of Rho-kinase activation and epithelial-mesenchymal transition (EMT) in renal injury. J Hypertens. 2006;24(suppl 6):128
- Jump up^ Baylis C, Qiu C, Engels K. Comparison of L-type and mixed L- and T-type calcium channel blockers on kidney injury caused by deoxycorticosterone-salt hypertension in rats. Am J Kidney Dis. 2001;38: 1292–1297.
- ^ Jump up to:a b c Sasaki H, Saiki A, Endo K, Ban N, Yamaguchi T, Kawana H, Nagayama D, Ohhira M, Oyama T, Miyashita Y, Shirai K. Protective effects of efonidipine, a T- and L-type calcium channel blocker, on renal function and arterial stiffness in type 2 diabetic patients with hypertension and nephropathy. J Atheroscler Thromb. 2009 Oct; 16(5): 568-75.
- Jump up^ Oshima T, Ozono R, Yano Y, Higashi Y, Teragawa H, Miho N, Ishida T, Ishida M, Yoshizumi M, Kambe M. Beneficial effect of T-type calcium channel blockers on endothelial function in patients with essential hypertension. Hypertens Res. 2005 Nov;28(11):889-94.
- ^ Jump up to:a b Nomura S, Kanazawa S, Fukuhara S. Effects of efonidipine on platelet and monocyte activation markers in hypertensive patients with and without type 2 diabetes mellitus. J Hum Hypertens. 2002 Aug;16(8):539-47.
- Jump up^ Yamashita T, Masuda Y, et al. NZ-105, a New 1,4-Dihydropyridine Derivative: Correlation between Dihydropyridine Receptor Binding and Inhibition of Calcium Uptake in Rabbit Aorta. Japan J Pharmacol. 1991; 57: 337-348.
- Jump up^ Nakabeppu H, et al.Metabolism of Efonidipine in Man.Xenobiotica.1995 Aug;229-239.
- ^ Jump up to:a b Hayashi K, Kumagai H, Saruta T. Effects of efonidipine and ACE inhibitors on proteinuria in human hypertension with renal impairment. Am J Hypertens. 2003;16:116 –122
- Jump up^ Oh IY, Seo MK, Lee HY, Kim SG, Kim KS, Kim WH, Hyon MS, Han KR, Lim SJ, Kim CH. Beneficial Effect of Efonidipine, an L- and T-Type Dual Calcium Channel Blocker, on Heart Rate and Blood Pressure in Patients With Mild-to-Moderate Essential Hypertension. Korean Circ J. 2010 Oct;40(10):514-9.
- ^ Jump up to:a b http://www.kegg.jp/medicus-bin/japic_med?japic_code=00044638. Missing or empty
|title=(help) - Jump up^ Ge W, Ren J. Combined L-/T-type calcium channel blockers: ready for prime time. Hypertension. 2009 Apr;53(4):592-4. doi: 10.1161/HYPERTENSIONAHA.108.127548. Epub 2009 Feb 23. PubMed PMID 19237678.
- Jump up^ Osterloh IH. An update on the safety of amlodipine. J Cardiovasc Pharmacol.1991;17 Suppl 1:S65-8.
- Jump up^ http://www.kegg.jp/medicus-bin/japic_med?japic_code=00062065. Missing or empty
|title=(help) - Jump up^ http://www.kegg.jp/medicus-bin/japic_med?japic_code=00005939. Missing or empty
|title=(help) - Jump up^ Takihata M, Nakamura A, Kondo Y, Kawasaki S, Kimura M, Terauchi Y. Comparison of Azelnidipine and Trichlormethiazide in Japanese Type 2 Diabetic Patients with Hypertension: The COAT Randomized Controlled Trial. PLoS One. 2015 May 4;10(5):e0125519.
- Jump up^ Song I, KimD, Choi S, Sun M, Kim Y, Shin HS. Role of the α1g T-type calcium channel in spontaneous absence seizures in mutant mice. J Neurosci. 2004; 24: 5249–5257.
- Jump up^ Lory P, Bidaud I, Chemin J. T-Type calcium channels in differentiation and proliferation. Cell Calcium. 2006; 40: 135–146.
- Jump up^ Ikeda K, Isaka T, Fujioka K, Manome Y, Tojo K. Suppression of Aldosterone Synthesis and Secretion by Ca2+ Channel Antagonists. International Journal of Endocrinology. 2012.
- Jump up^ Oh IY, Seo MK, Lee HY, Kim SG, Kim KS, Kim WH, Hyon MS, Han KR, Lim SJ, Kim CH. Beneficial Effect of Efonidipine, an L- and T-Type Dual Calcium Channel Blocker, on Heart Rate and Blood Pressure in Patients With Mild-to-Moderate Essential Hypertension. Korean Circ J. 2010 Oct;40(10):514-9.
- Jump up^ Catena C, Colussi G, Marzano L, Sechi LA. Aldosterone and the heart: from basic research to clinical evidence. Horm Metab Res. 2012;44:181– 187.
- Jump up^ Sasaki H, Saiki A, Endo K, Ban N, Yamaguchi T, Kawana H, Nagayama D, Ohhira M, Oyama T, Miyashita Y, Shirai K. Protective effects of efonidipine, a T- and L-type calcium channel blocker, on renal function and arterial stiffness in type 2 diabetic patients with hypertension and nephropathy. J Atheroscler Thromb. 2009 Oct; 16(5): 568-75.
- Jump up^ Saito T, Fujii K, Takizawa T, Toyosaki T, Kuwabara Y, Kobayashi S, Ichikawa H, Karaki A, Yamazaki Y, Iwata J, Yamada K, Tomiya H, Takeda K, Inagaki Y. Effects of the new calcium antagonist efonidipine hydrochloride on resting and exercise hemodynamics in patients with stable effort angina. Arzneimittelforschung. 1996 Sep;46(9):861-7.
- ^ Jump up to:a b Saruta T. Current status of calcium antagonists in Japan. Am J Cardiol. 1998;82:32R-34R.
- Jump up^ Okayama S, Imagawa K, Naya N, Iwama H, Somekawa S, Kawata H, Horii M, Nakajima T, Uemura S, Saito Y. Blocking T-type Ca2+ channels with efonidipine decreased plasma aldosterone concentration in healthy volunteers. Hypertens Res. 2006 Jul;29(7):493-7.
- Jump up^ Honda M, Hayashi K, Matsuda H, Kubota E, Tokuyama H, Okubo K, Ozawa Y, Saruta T. Divergent natriuretic action of calcium channel antagonists in mongrel dogs: renal haemodynamics as a determinant of natriuresis. Clinical Science. 2001; 101: 421–427
- Jump up^ Wagner C, Kramer KB, Hinder M, Kieninger M, Kurtz A. T-type and L-type calcium channel blockers exert opposite effects on renin secretion and renin gene expression in conscious rats. Br J Pharmacol. 1998;124: 579 –585.
- Jump up^ Song I, KimD, Choi S, Sun M, Kim Y, Shin HS. Role of the α1g T-type calcium channel in spontaneous absence seizures in mutant mice. J Neurosci. 2004; 24: 5249–5257.
- Jump up^ Lory P, Bidaud I, Chemin J. T-Type calcium channels in differentiation and proliferation. Cell Calcium. 2006; 40: 135–146.
- Jump up^ Ando K, Ueshima K, Tanaka S, Kosugi S, Sato T, Matsuoka H, Nakao K, Fujita T. Comparison of the antialbuminuric effects of L-/N-type and L-type calcium channel blockers in hypertensive patients with diabetes and microalbuminuria: the study of assessment for kidney function by urinary microalbumin in randomized (SAKURA) trial. Int J Med Sci. 2013 Jul 30;10(9):1209-16.
- ^ Jump up to:a b Hayashi K, Wakino S, Sugano N, Ozawa Y, Homma K, Saruta T. Ca2+ Channel Subtypes and Pharmacology in the Kidney. Circ Res. 2007;100:342-353.
- ^ Jump up to:a b Hayashi K, Ozawa Y, Fujiwara K, Wakino S, Kumagai H, Saruta T. Role of actions of calcium antagonists on efferent arterioles with special references to glomerular hypertension. Am J Nephrol. 2003 Jul-Aug;23(4):229-44.
- ^ Jump up to:a b Tani S, Takahashi A, Nagao K, Hirayama A. Effects of the T/L-type calcium channel blocker benidipine on albuminuria and plasma aldosterone concentration. A pilot study involving switching from L-type calcium channel blockers to benidipine. Int Heart J. 2014;55(6):519-25
- Jump up^ Li M. Role of T-Type Ca2+ Channels in Basal Insulin Release. T-type Calcium Channels in Basic and Clinical Science. Springer Vienna. 2015; 137-150.
- (in Japanese) Landel ランデル (PDF) Shionogi & Co. April 2005.
 |
|
| Clinical data | |
|---|---|
| Trade names | Landel (ランデル) |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration |
Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C34H38N3O7P |
| Molar mass | 631.65 g/mol |
| 3D model (JSmol) | |

(R)-base
- Formula:C34H38N3O7P
- MW:631.67 g/mol
- CAS-RN:128194-13-8
(S)-base
- Formula:C34H38N3O7P
- MW:631.67 g/mol
- CAS-RN:128194-12-7
///////////Efonidipine, エホニジピン, IND 2017, Landel , NZ 105, Efonidipine Hydrochloride Ethanolate
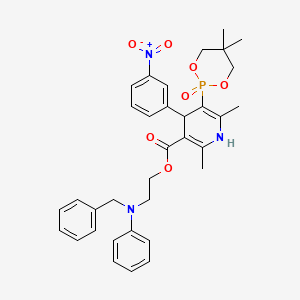
CC1=C(C(C(=C(N1)C)P2(=O)OCC(CO2)(C)C)C3=CC(=CC=C3)[N+](=O)[O-])C(=O)OCCN(CC4=CC=CC=C4)C5=CC=CC=C5
A call to (green) arms: a rallying cry for green chemistry and engineering for CO2 capture, utilisation and storage
A call to (green) arms: a rallying cry for green chemistry and engineering for CO2 capture, utilisation and storage
Abstract
Chemists, engineers, scientists, lend us your ears… Carbon capture, utilisation, and storage (CCUS) is among the largest challenges on the horizon and we need your help. In this perspective, we focus on identifying the critical research needs to make CCUS a reality, with an emphasis on how the principles of green chemistry (GC) and green engineering can be used to help address this challenge. We identify areas where GC principles can readily improve the energy or atom efficiency of processes or reduce the environmental impact. Conversely, we also identify dilemmas where the…
View original post 115 more words
Technetium (99mTc) tetrofosmin, テトロホスミンテクネチウム (99mTc)

Technetium (99mTc) tetrofosmin, 99mTc-Tetrofosmin
テトロホスミンテクネチウム (99mTc)
| Formula | C36H80O10P4Tc |
|---|---|
| Molar mass | 895.813 g/mol |
| CAS Number |
|
|---|
UNII42FOP1YX93
2-[bis(2-ethoxyethyl)phosphanyl]ethyl-bis(2-ethoxyethyl)phosphane;technetium-98;dihydrate
Technetium Tc 99m tetrofosmin; Technetium Tc-99m tetrofosmin; TECHNETIUM TC-99M TETROFOSMIN KIT; Tc-99m tetrofosmin; Technetium-99 tetrofosmin; Technetium (99mTc) tetrofosmin
Technetium Tc-99m Tetrofosmin is a radiopharmaceutical consisting of tetrofosmin, composed of two bidentate diphosphine ligands chelating the metastable radioisotope technetium Tc-99 (99mTc), with potential imaging activity upon SPECT (single photon emission computed tomography). Upon administration, technetium Tc 99m tetrofosmin is preferentially taken up by, and accumulates in, myocardial cells. Upon imaging, myocardial cells can be visualized and changes in ischemia and/or perfusion can be detected.
Technetium Tc-99m tetrofosmin is a drug used in nuclear myocardial perfusion imaging. The radioisotope, technetium-99m, is chelated by two 1,2-bis[di-(2-ethoxyethyl)phosphino]ethane ligands which belong to the group of diphosphines and which are referred to as tetrofosmin. It is a lipophilic technetium phosphine dioxo cation that was formulated into a freeze-dried kit which yields an injection.[A31592] Technetium Tc-99m tetrofosmin was developed by GE Healthcare and FDA approved on February 9, 1996.
Technetium Tc-99m tetrofosmin is a drug used in nuclear myocardial perfusion imaging. The radioisotope, technetium-99m, is chelated by two 1,2-bis[di-(2-ethoxyethyl)phosphino]ethane ligands which belong to the group of diphosphines and which are referred to as tetrofosmin. It is a lipophilic technetium phosphine dioxo cation that was formulated into a freeze-dried kit which yields an injection.[1] Technetium Tc-99m tetrofosmin was developed by GE Healthcare and FDA approved on February 9, 1996.
Technetium (99mTc) tetrofosmin is a drug used in nuclear medicine cardiac imaging. It is sold under the brand name Myoview (GE Healthcare). The radioisotope, technetium-99m, is chelated by two 1,2-bis[di-(2-ethoxyethyl)phosphino]ethane ligands which belong to the group of diphosphines and which are referred to as tetrofosmin.[1][2]
Tc-99m tetrofosmin is rapidly taken up by myocardial tissue and reaches its maximum level in approximately 5 minutes. About 66% of the total injected dose is excreted within 48 hours after injection (40% urine, 26% feces). Tc-99m tetrofosmin is indicated for use in scintigraphic imaging of the myocardium under stress and rest conditions. It is used to determine areas of reversible ischemia and infarcted tissue in the heart. It is also indicated to detect changes in perfusion induced by pharmacologic stress (adenosine, lexiscan, dobutamine or persantine) in patients with coronary artery disease. Its third indication is to assess left ventricular function (ejection fraction) in patients thought to have heart disease. No contraindications are known for use of Tc-99m tetrofosmin, but care should be taken to constantly monitor the cardiac function in patients with known or suspected coronary artery disease. Patients should be encouraged to void their bladders as soon as the images are gathered, and as often as possible after the tests to decrease their radiation doses, since the majority of elimination is renal. The recommended dose of Tc-99m tetrofosmin is between 5 and 33 millicuries (185-1221 megabecquerels). For a two-dose stress/rest dosing, the typical dose is normally a 10 mCi dose, followed one to four hours later by a dose of 30 mCi. Imaging normally begins 15 minutes following injection.[3]

Amersham (formerly Nycomed Amersham , now GE Healthcare ) has developed and launched 99mTc-tetrofosmin (Myoview) as an injectable nuclear imaging agent for ischemic heart disease in several major territories and for use in detecting breast tumors
Technetium (99mTc) tetrofosmin is a drug used in nuclear medicine cardiac imaging. It is sold under the brand name Myoview (GE Healthcare). The radioisotope, technetium-99m, is chelated by two 1, 2-bis-[bis-(2-ethoxyethyl)phosphino] ethane ligands, which belong to the group of diphosphines and which are referred to as tetrofosmin and has the structural Formula 1 :
Formula 1
99mTc -based radiopharmaceuticals are commonly used in diagnostic nuclear medicine, especially for in vivo imaging (e.g. via immunoscintigraphy or radiolabeling). Usually cold kits are manufactured in advance in accordance with strict requirements of Good Manufacturing Practice (GMP) Guidelines, containing the chemical ingredients (e.g. 99mTc -coordinating ligands, preservatives) in lyophilized form. The radioactive isotope 99mTc (ti/2 = 6h) is added to those kits shortly before application to the patient via intravenous or subcutaneous injection.
Tc-99m tetrofosmin is rapidly taken up by myocardial tissue and reaches its maximum level in approximately 5 minutes. About 66% of the total injected dose is excreted within 48 hours after injection (40% urine, 26% feces). Tc-99m tetrofosmin is indicated for use in scintigraphic imaging of the myocardium under stress and rest conditions. It is used to determine areas of reversible ischemia and infarcted tissue in the heart. It is also indicated to detect changes in perfusion induced by pharmacologic stress (adenosine, lexiscan, dobutamine or persantine) in patients with coronary artery disease. Its third indication is to assess left ventricular function (ejection fraction) in patients thought to have heart disease. No contraindications are known for use of Tc-99m tetrofosmin, but care should be taken to constantly monitor the cardiac function in patients with known or suspected coronary artery disease. Patients should be encouraged to void their bladders as soon as the images are gathered, and as often as possible after the tests to decrease their radiation doses, since the majority of elimination is renal. The recommended dose of Tc-99m tetrofosmin is between 5 and 33 millicuries (185-1221 megabecquerels). For a two-dose stress/rest dosing, the typical dose is normally a 10 mCi dose, followed one to four hours later by a dose of 30 mCi. Imaging normally begins 15 minutes following injection.
99mTc -Tetrofosmin is also described to be useful for tumor diagnostics, in particular of breast cancer and parathyroid gland cancer, and for multidrug resistance (MDR) research.
US5045302 discloses 99mTc-coordinating diphosphine ligands (L), wherein one preferred example thereof is the ether functionalized diphosphine ligand l,2-bis[bis(2-ethoxy- ethyl)phosphino]ethane according to Formula 1, called tetrofosmin (“P53”), that forms a dimeric cationic technetium (V) dioxo phosphine complex, [TCO2L2] with 99mTc, useful as myocardial imaging agent. Example 1 of said patent described the process for preparing tetrofosmin by reacting ethyl vinyl ether, bis(diphosphino)ethane in the presence of a-azo-isobutyronitrile (AIBN) in a fischer pressure-bottle equipped with a teflon stirring bar followed by removal of volatile materials and non-distillable material obtained, as per below mentioned Scheme 1.
Scheme 1
Formula 2 Formula 3 Formula 1
CN 1184225 C discloses tetrofosmin salts containing chloride or bromide or aryl sulfonates as negatively charged counter ions, which can be used for the preparation of a 99mTc- Tetrofosmin radiopharmaceutical composition. According to this patent tetrofosmin hydrochloride is a viscous liquid. Own experiments of the inventors of the present invention revealed that the halide salts of tetrofosmin are hygroscopic oils, which are complicated to handle, e.g. when weighed. The oily and hygrospcopic
properties of tetrofosmin hydrochloride hampers its use in pharmaceutical preparations. Attempts to synthesize the subsalicylate salt of tetrofosmin failed because the starting material sulfosalicylic acid was not soluble in ether in the concentration specified in the patent (3.4 g in 15 ml).
WO2006/064175A1 discloses tetrofosmin was converted to tetrofosmin subsalicylate by reaction with 2.3 to 2.5 molar equivalents of 5-sulfosalicyclic acid at room temperature in ethanol, followed by recrystallisation from ethanol/ether.
WO2015/114002A1 relates to tetrafluoroborate salt of tetrafosmin and its process for the preparation thereof. Further this application also discloses one-vial and two vial kit formulation with tetrafluoroborate salt of tetrafosmin.
The article Proceedings of the International Symposium, 7th, Dresden, Germany, June 18-22, 2000 by Amersham Pharmacia Biotech UK Limited titled “The synthesis of [14C]tetrofosmin, a compound vital to the development of Myoview, Synthesis and Applications of Isotopically Labelled Compounds” disclosed a process for the preparation of tetrofosmin as per below mentioned Scheme 2:
Scheme 2
Formula 1A Formula 7
The starting material was bis(2- ethoxyethyl)benzylphosphine of Formula 4 . This was prepared from benzyl phosphonate, PhCH2P(0)(OEt)2 by reduction with lithium aluminium hydride to give the intermediate benzylphosphine, PhCH2PH2, followed by a photolysis reaction in the presence of ethyl vinyl ether to give compound of Formula 4. The compound of Formula 4 in acetonitrile was treated with dibromo[U-14C]ethane to give compound of Formula 6, further it was treated with excess of 30% aqueous sodium hydroxide in ethanol. The mixture was stirred at room temperature for 24 hours. The solvent was removed and the residue was treated with excess concentrated hydrochloric acid at 0°C. Aqueous work up gave compound of Formula 7. Then compound of Formula 7 in dry benzene was treated with hexachlorodisilane and hydrolysed with excess 30% aqueous sodium hydroxide at 0°C. Aqueous work up followed by flash column chromatography on silica gave [bisphosphinoethane- 1,2-14C]tetrofosmin of formula 1A.
The article Polyhedron (1995), 14(8), 1057-65, titled “Synthesis and characterization of Group 10 metal complexes with a new trifunctional ether phosphine. The X-ray crystal structures of bis[bis(2-ethoxyethyl)benzylphosphine]dichloronickel(II) and bis[bis(2-ethoxyethyl)benzylphosphine]chlorophenylnickel(II)” disclosed the process for the preparation of bis(2-ethoxyethyl)benzylphosphine as per below mentioned Scheme 3:
Scheme 3
Formula 8 Formula 9 Formula 4
The compound bis(2-ethoxyethyl)benzylphosphine of Formula 4 was prepared by first reduction of diethylbenzylphosphonate of Formula 8 using lithium aluminium hydride to obtain benzyl phosphine of Formula 9 followed by radical catalysed coupling reaction with ethyl vinyl ether carried out by using UV photolysis.
Tetrofosmin is extremely sensitive to atmospheric oxygen, which makes synthesis of the substance, as well as manufacturing and handling of the kit complicated as the substance has constantly to be handled in an oxygen free atmosphere.
High purity and stability under dry and controlled conditions are pivotal requirements for chemical compounds used as active ingredients in pharmaceuticals.
The processes disclosed in prior art for the preparation of compound of Formula 4 involves that coupling reaction of benzyl phosphine of Formula 9 with ethyl vinyl ether carried out by using photolytic conditions. Such technology is expensive as it requires separate instruments including isolated facility (to avoid the UV radiation exposure etc.), also it is not suitable for commercial scale production.
PATENT
WO-2018162964
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2018162964&tab=PCTDESCRIPTION&maxRec=1000
Example 1
Preparation of benzyl phosphine:
A mixture of lithium aluminium hydride (25 g) in methyl tertiary butyl ether (MTBE) (800 ml) was cooled to 0 to 5°C and added a solution of diethylbenzylphosphonate in methyl tertiary butyl ether (100 g in 200ml). The temperature of reaction mixture was raised to 25 to 30 °C and stirred for 14 to 16 hour. After completion of the reaction, the reaction mixture was cooled to 0 to 5°C and 6N hydrochloric acid was added slowly. Further raised the temperature of reaction mixture to 25 to 30 °C and stirred for 30-45 minutes. The layers were separated, the aqueous layer was extracted with MTBE (250ml) and the combined organic layer was washed with deoxygenated water. The organic layer was dried over sodium sulfate and concentrated to obtain the title compound as non-distillable liquid.
Example 2
Preparation of benzylbis(2-ethoxyethyl)phosphane:
To a mixture of benzyl phosphine (obtained from example 1) and vinyl ethyl ether (250 ml) in pressure RB flask was added a-azo-isobutyronitrile (AIBN) (1.5g). The resulting reaction mixture was maintained at 80 to 90°C for 14 to 16 hours. The mixture was cooled to 20 to 30°C and AIBN (0.5g) added, then continued to heat the reaction mixture at 80 to 90°C for 6 to 7 hours. After completion of the reaction, the reaction mixture was allowed to cool to room temperature and distilled under vacuum to obtain title compound as an oil (107 g).
Example 3
Preparation of Ethane- 1,2-diylbis (benzylbis(2-ethoxyethyl) phosphonium) bromide:
To a mixture of benzylbis(2-ethoxyethyl)phosphane 107.g) in acetonitrile (100ml) in pressure bottle was added 1, 2-dibromoethane (30.5 g). The reaction mixture was maintained at 80 to 90°C for 20 to 25 hours. After completion of the reaction, the reaction mass was cooled to room temperature and stirred for 45 to 60 minutes to obtain the solid. To the solid obtained was added methyl tertiary butyl ether (MTBE) (500ml) and stirred at room temperature for 2 to 3 hour. The reaction mass was filtered, washed with MTBE and suck dried. Further the filtered solid was heated in acetone (400ml) at 50 to 55°C for 2 to 3 hour. Then cooled the reaction mixture to room temperature, stirred, filtered and washed with acetone to obtain the title compound as white solid. (85g)
Example 4
Preparation of Ethane- 1, 2-diylbis (bis (2-ethoxy ethyl) phosphine oxide):
To a mixture of Ethane- 1,2-diylbis (benzylbis(2-ethoxyethyl) phosphonium) bromide (80g) in ethanol (480 ml) was added an aq. solution of sodium hydroxide ( 48g in 160 ml water) at room temperature. The reaction mass was maintained at 25 to 35°C for 10 to 12 hour. After completion of the reaction, the reaction mass was cone, under vacuum to obtained the residue. The residue was dissolved in deoxygenated water (400 ml) and washed with MTBE (400 ml x 2). The layers were separated, the aqueous layer was cooled to 10 to 20°C and 6N hydrochloric acid (200 ml) was added slowly. Then extracted the aqueous layer with dichloromethane (2000 ml), washed the organic layer with deoxygenated water (160 ml), dried the organic layer using sodium sulfate, filtered, and distilled under vacuum to obtain the residue. Further MTBE (160 ml x 2) was added to the residue and continued distillation under vacuum, degassed to obtain the solid. To the obtained solid, MTBE (400 ml) was added and heated at 45 to 50°C for 1-2 hour, further slowly cooled the reaction mass to 25 to 30°C, filtered the solid product. Again MTBE (400 ml) was added to the solid product and heated at 45 to 50°C for 1-2 hour, further slowly cooled the reaction mass to 25 to 30°C, filtered, washed with MTBE and dried under vacuum to obtain the title compound as white solid (32g).
Example 5
Preparation of tetrofosmin free base:
To a mixture of ethane- 1, 2-diylbis (bis (2-ethoxyethyl) phosphine oxide (18g) in toluene (180ml) in pressure RB flask argon/nitrogen gas was purged for 5 minute and hexachlorodisilane (30g) was added. The reaction mixture was heated to 80 to 90°C, stirred for 10 to 12 hour, further slowly cooled to -5 to 0°C and slowly added 30% aqueous sodium hydroxide solution (45g sodium hydroxide in 150 ml deoxygenated water) the temperature of reaction mixture was raised to 25 to 30°C and stirred for 1 to 2 hour. The layers were separated and the aq. layer was extracted with Toluene (180 ml). The combined organic layer was washed with deoxygenated water (180 ml). Further dried the organic layer using sodium sulfate, distilled under vacuum to obtain the residue of tetrofosmin free base (15.5g).
Example 6
Preparation of tetrofosmin disulfosalicylate salt:
To the residue of tetrofosmin free base (15.5g) was added an aq. solution of 5-sulfosalicylic acid dihydrate (21.6g in 75ml deoxygenated water) and stirred at 25 to 30°C for 25 to 30 minutes. Further heated the reaction mass to 55 to 60°C, stirred for 15 to 30 minute, slowly cooled the reaction mass to 10 to 15°C and stirred for 1-2 hour. Filtered, washed with chilled deoxygenated water, and dried under vacuum to obtain the title compound as white solid. (30g).
Example 7
Preparation of Form J of tetrofosmin disulfosalicylate salt:
An aq. solution of 5-sulfosalicylic acid dihydrate (21.6g in 75ml deoxygenated water) was added slowly into tetrofosmin free base (15.5g) and stirred at room temperature for 30 to 40 minutes. The temperature of reaction mixture was further raised to 50 to 60°C, stirred for 20 to 30 minute, cooled the reaction mass to 10 to 15°C and stirred for 1-2 hour. Filtered, washed with chilled deoxygenated water, and dried under vacuum to obtain the title compound.
PATENT
EP337654 ,
PATENT
US9549999
FDA Orange Book Patents
References
- Jump up^ Kelly JD, Alan M. Forster AM, Higley B, et al. (February 1993). “Technetium-99m-Tetrofosmin as a new radiopharmaceutical for myocardial perfusion imaging”. Journal of Nuclear Medicine. 34 (2): 222–227. PMID 8429340.
- Jump up^ Elhendy A, Schinkel AF, et al. (December 2005). “Risk stratification of patients with angina pectoris by stress 99mTc-tetrofosmin myocardial perfusion imaging”. Journal of Nuclear Medicine. 46 (12): 2003–2008. PMID 16330563.
- Jump up^ Myoview package insert. Arlington Heights, IL: GE Healthcare, 2006, Aug.
 |
|
| Clinical data | |
|---|---|
| Routes of administration |
Intravenous |
| ATC code | |
| Pharmacokinetic data | |
| Bioavailability | N/A |
| Identifiers | |
| CAS Number |
|
| Chemical and physical data | |
| Formula | C36H80O10P4Tc |
| Molar mass | 895.813 g/mol |
External links
Myoview Prescribing Information Page
//////////99mTc-Tetrofosmin, Technetium (99mTc) tetrofosmin, テトロホスミンテクネチウム (99mTc)
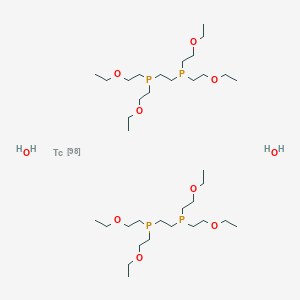
CCOCCP(CCOCC)CCP(CCOCC)CCOCC.CCOCCP(CCOCC)CCP(CCOCC)CCOCC.O.O.[Tc]
Tasimelteon, タシメルテオン

Tasimelteon
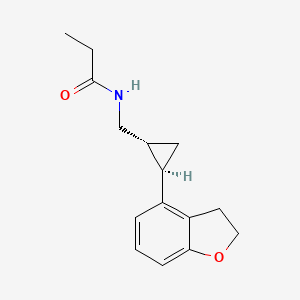
N-([(1R,2R)-2-(2,3-Dihydro-1-benzofuran-4-yl)cyclopropyl]methyl)propanamide,
609799-22-6 cas, BMS-214778; VEC-162, ATC:N05CH03
- Use:Treatment of sleep disorder; Melatonin receptor agonist
- (1R,2R)-N-[2-(2,3-dihydrobenzofuran-4-yl)cyclopropylmethyl]propanamide
- Formula:C15H19NO2, MW:245.3 g/mol
-
Hetlioz Vanda Pharmaceuticals, 2014
Approved fda 2014
EMA
Tasimelteon is a white to off-white crystalline powder, it is non hygroscopic, soluble in water across relevant pH values and freely soluble in alcohols, cyclohexane, and acetonitrile. Conducted in vivo studies demonstrate that tasimelteon is highly permeable substance. Photostability testing and testing on stress conditions demonstrated that the active substance degrades in light.
Tasimelteon exhibits stereoisomerism due to the presence of two chiral centres. Active substance is manufactured as a single, trans-1R,2R isomer. Enantiomeric purity is controlled routinely during manufacture of active substance intermediates by chiral HPLC/specific optical rotation and additionally controlled in the active substance. Stability data indicates tasimelteon is isomerically stable.
Polymorphism has been observed in polymorphic screening studies for tasimelteon and two forms have been identified. The thermodynamically more stable form has been chosen for development and the manufacturing process consistently yields active substance of single, desired polymorphic form. It was demonstrated that milling of the active substance does not affect polymorphic form. Polymorphism is additionally controlled in active substance release and shelf-life specifications using X-ray powder diffraction analysis.
Tasimelteon is synthesized in nine main steps using linear synthesis and using commercially available well-defined starting materials with acceptable specifications. Three intermediates are isolated for control of active substance quality including stereochemical control. The active substance is isolated by slow recrystallisation or precipitation of tasimelteon from an ethanol/water mixture which ensures the formation of desired polymorphic form. Up to two additional, optional recrystallisations may be performed for unmilled tasimelteon to ensure that milled tasimelteon active substance is of high purity. Seed crystals complying with active substance specifications can be used optionally. Active substance is jet milled (micronised) to reduce and control particle size, which is critical in finished product performance with regards to content uniformity and dissolution…….http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Public_assessment_report/human/003870/WC500190309.pdf
launched in 2014 in the U.S. by Vanda Pharmaceuticals for the treatment of non-24-hour sleep-wake disorder in totally blind subjects. In 2015, the European Committee for Medicinal Products of the European Medicines Agency granted approval for the same indication. In 2010 and 2011, orphan drug designations were assigned for the treatment of non-24 hour sleep/wake disorder in blind individuals without light perception in the U.S. and the E.U., respectively.
Tasimelteon (trade name Hetlioz) is a drug approved by the U.S. Food and Drug Administration (FDA)[2] in January 2014 for the treatment of non-24-hour sleep–wake disorder (also called Non-24, N24 and N24HSWD).[3] In June 2014, the European Medicines Agency accepted an EU filing application for tasimelteon[4] and in July 2015, the drug was approved in Europe for the treatment of non-24-hour sleep-wake rhythm disorder in totally blind adults,[5] but not in the rarer case of non-24 in sighted people.
Tasimelteon is a selective agonist for the melatonin receptors MT1 and MT2, similar to other members of the melatonin receptor agonistclass of which ramelteon (2005) and agomelatine (2009) were the first approved.[6] As a treatment for N24HSWD, as with melatonin or other melatonin derivatives, the patient may experience improved sleep timing while taking the drug. Reversion to baseline sleep performance occurs within a month of discontinuation.[7]

Development
Tasimelteon (previously known as BMS-214,778) was developed for the treatment of insomnia and other sleep disorders. A phase II trial on circadian rhythm sleep disorders was concluded in March 2005.[8] A phase III insomnia trial was conducted in 2006.[9] A second phase III trial on insomnia, this time concerning primary insomnia, was completed in June 2008.[10] In 2010, the FDA granted orphan drug status to tasimelteon, then regarded as an investigational medication, for use in totally blind adults with N24HSWD.[11] (Through mechanisms such as easing the approval process and extending exclusivity periods, orphan drug status encourages development of drugs for rare conditions that otherwise might lack sufficient commercial incentive.)
On completion of Phase III trials, interpretations of the clinical trials by the research team concluded that the drug may have therapeutic potential for transient insomnia in circadian rhythm sleep disorders.[12] A year-long (2011–2012) study at Harvard tested the use of tasimelteon in blind subjects with non-24-hour sleep-wake disorder. The drug has not been tested in children nor in any non-blind people.
FDA approval
In May 2013 Vanda Pharmaceuticals submitted a New Drug Application to the Food and Drug Administration for tasimelteon for the treatment of non-24-hour sleep–wake disorder in totally blind people. It was approved by the FDA on January 31, 2014 under the brand name Hetlioz.[3] In the opinion of Public Citizen, an advocacy group, the FDA erroneously allowed it to be labelled without stating that it is only approved for use by totally blind people.[13] However, FDA updated its press release on Oct. 2, 2014 to clarify the approved use of Hetlioz, which includes both sighted and blind individuals. The update did not change the drug labeling (prescribing information).[14]
Toxicity
Experiments with rodents revealed fertility impairments, an increase in certain cancers, and serious adverse events during pregnancy at dosages in excess of what is considered the “human dose”.[15][16]
As expected, advisors to the US Food and Drug Administration have recommended approval of Vanda Pharmaceuticals’ tasimelteon, to be sold as Hetlioz, for the treatment of non-24-hour disorder in the totally blind.http://www.pharmatimes.com/Article/13-11-14/FDA_panel_backs_Vanda_body_clock_drug_for_blind.aspx
The master body clock controls the timing of many aspects of physiology, behavior and metabolism that show daily rhythms, including the sleep-wake cycles, body temperature, alertness and performance, metabolic rhythms and certain hormones which exhibit circadian variation. Outputs from the
suprachiasmatic nucleus (SCN) control many endocrine rhythms including those of melatonin secretion by the pineal gland as well as the control of Cortisol secretion via effects on the hypothalamus, the pituitary and the adrenal glands. This master body clock, located in the SCN, spontaneously generates rhythms of approximately 24.5 hours. These non-24-hour rhythms are synchronized each day to the 24-hour day-night cycle by light, the primary environmental time cue which is detected by specialized cells in the retina and transmitted to the SCN via the retino-hypothalamic tract. Inability to detect this light signal, as occurs in most totally blind individuals, leads to the inability of the master body clock to be reset daily and maintain entrainment to a 24-hour day.
Non-24-Hour Disorder, Non-24, also referred to as Non-24-Hour Sleep-Wake Disorder, (N24HSWD) or Non-24-Hour Disorder, is an orphan indication affecting approximately 65,000 to 95,000 people in the U.S. and 140,000 in Europe. Non- 24 occurs when individuals, primarily blind with no light perception, are unable to synchronize their endogenous circadian pacemaker to the 24-hour light/dark cycle. Without light as a synchronizer, and because the period of the internal clock is typically a little longer than 24 hours, individuals with Non-24 experience their circadian drive to initiate sleep drifting later and later each day. Individuals with Non-24 have abnormal night sleep patterns, accompanied by difficulty staying awake during the day. Non-24 leads to significant impairment, with chronic effects impacting the social and occupational functioning of these individuals.
In addition to problems sleeping at the desired time, individuals with Non-24 experience excessive daytime sleepiness that often results in daytime napping.
The severity of nighttime sleep complaints and/or daytime sleepiness complaints varies depending on where in the cycle the individual’s body clock is with respect to their social, work, or sleep schedule. The “free running” of the clock results in approximately a 1-4 month repeating cycle, the circadian cycle, where the circadian drive to initiate sleep continually shifts a little each day (about 15 minutes on average) until the cycle repeats itself. Initially, when the circadian cycle becomes desynchronous with the 24h day-night cycle, individuals with Non-24 have difficulty initiating sleep. As time progresses, the internal circadian rhythms of these individuals becomes 180 degrees out of synchrony with the 24h day-night cycle, which gradually makes sleeping at night virtually impossible, and leads to extreme sleepiness during daytime hours.
Eventually, the individual’s sleep-wake cycle becomes aligned with the night, and “free-running” individuals are able to sleep well during a conventional or socially acceptable time. However, the alignment between the internal circadian rhythm and the 24-hour day-night cycle is only temporary.
In addition to cyclical nighttime sleep and daytime sleepiness problems, this condition can cause deleterious daily shifts in body temperature and hormone secretion, may cause metabolic disruption and is sometimes associated with depressive symptoms and mood disorders.
It is estimated that 50-75% of totally blind people in the United States (approximately 65,000 to 95,000) have Non-24. This condition can also affect sighted people. However, cases are rarely reported in this population, and the true rate of Non-24 in the general population is not known.
The ultimate treatment goal for individuals with Non-24 is to entrain or synchronize their circadian rhythms into an appropriate phase relationship with the 24-hour day so that they will have increased sleepiness during the night and increased wakefulness during the daytime. Tasimelteon
Tasimelteon is a circadian regulator which binds specifically to two high affinity melatonin receptors, Mella (MT1R) and Mellb (MT2R). These receptors are found in high density in the suprachiasmatic nucleus of the brain (SCN), which is responsible for synchronizing our sleep/wake cycle. Tasimelteon has been shown to improve sleep parameters in prior clinical studies, which simulated a desynchronization of the circadian clock. Tasimelteon has so far been studied in hundreds of individuals and has shown a good tolerability profile.
Tasimelteon has the chemical name: tr ns-N-[[2-(2,3-dihydrobenzofuran- 4-yl)cycloprop-lyl] methyl] propanamide, has the structure of Formula I:
Formula I
and is disclosed in US 5856529 and in US 20090105333, both of which are incorporated herein by reference as though fully set forth.
Tasimelteon is a white to off-white powder with a melting point of about 78°C (DSC) and is very soluble or freely soluble in 95% ethanol, methanol, acetonitrile, ethyl acetate, isopropanol, polyethylene glycols (PEG-300 and PEG- 400), and only slightly soluble in water. The native pH of a saturated solution of tasimelteon in water is 8.5 and its aqueous solubility is practically unaffected by pH. Tasimelteon has 2-4 times greater affinity for MT2R relative to MTIR. It’s affinity (¾) for MTIR is 0.3 to 0.4 and for MT2R, 0.1 to 0.2. Tasimelteon is useful in the practice of this invention because it is a melatonin agonist that has been demonstrated, among other activities, to entrain patients suffering from Non-24.
Metabolites of tasimelteon include, for example, those described in “Preclinical Pharmacokinetics and Metabolism of BMS-214778, a Novel
Melatonin Receptor Agonist” by Vachharajani et al., J. Pharmaceutical Sci., 92(4):760-772, which is hereby incorporated herein by reference. The active metabolites of tasimelteon can also be used in the method of this invention, as can pharmaceutically acceptable salts of tasimelteon or of its active metabolites. For example, in addition to metabolites of Formula II and III, above, metabolites of tasimelteon also include the monohydroxylated analogs M13 of Formula IV, M12 of Formula V, and M14 of Formula VI.
Formula IV
Formula V
MO
Formula VI
Thus, it is apparent that this invention contemplates entrainment of patients suffering free running circadian rhythm to a 24 hour circadian rhythm by administration of a circadian rhythm regulator (i.e., circadian rhythm modifier) capable of phase advancing and/or entraining circadian rhythms, such as a melatonin agonist like tasimelteon or an active metabolite oftasimelteon or a pharmaceutically acceptable salt thereof. Other MT1R and MT2R agonists, i.e., melatonin agonists, can have similar effects on the master body clock. So, for example, this invention further contemplates the use of melatonin agonists such as but not limited to melatonin, N-[l-(2,3-dihydrobenzofuran-4- yl)pyrrolidin-3-yl]-N-ethylurea and structurally related compounds as disclosed in US 6,211,225, LY-156735 ((R)-N-(2-(6-chloro-5-methoxy-lH-indol- 3yl) propyl) acetamide) (disclosed in U.S. Patent No. 4,997,845), agomelatine (N- [2-(7-methoxy-l-naphthyl)ethyl]acetamide) (disclosed in U.S. Patent No.
5,225,442), ramelteon ((S)-N-[2-(l,6,7,8-tetrahydro-2H-indeno- [5,4-b] furan-8- yl)ethyl]propionamide), 2-phenylmelatonin, 8-M-PDOT, 2-iodomelatonin, and 6- chloromelatonin.
Additional melatonin agonists include, without limitation, those listed in U.S. Patent Application Publication No. 20050164987, which is incorporated herein by reference, specifically: TAK-375 (see Kato, K. et al. Int. J.
Neuropsychopharmacol. 2000, 3 (Suppl. 1): Abst P.03.130; see also abstracts P.03.125 and P.03.127), CGP 52608 (l-(3-allyl-4-oxothiazolidine-2-ylidene)-4- met- hylthiosemicarbazone) (See Missbach et al., J. Biol. Chem. 1996, 271, 13515-22), GR196429 (N-[2-[2,3,7,8-tetrahydro-lH-fur-o(2,3-g)indol-l- yl] ethyl] acetamide) (see Beresford et al., J. Pharmacol. Exp. Ther. 1998, 285, 1239-1245), S20242 (N-[2-(7-methoxy napth-l-yl) ethyl] propionamide) (see Depres-Brummer et al., Eur. J. Pharmacol. 1998, 347, 57-66), S-23478 (see Neuropharmacology July 2000), S24268 (see Naunyn Schmiedebergs Arch. June 2003), S25150 (see Naunyn Schmiedebergs Arch. June 2003), GW-290569, luzindole (2-benzyl-N-acetyltryptamine) (see U.S. Patent No. 5,093,352), GR135531 (5-methoxycarbonylamino-N-acetyltrypt- amine) (see U.S. Patent Application Publication No. 20010047016), Melatonin Research Compound A, Melatonin Agonist A (see IMSWorld R&D Focus August 2002), Melatonin
Analogue B (see Pharmaprojects August 1998), Melatonin Agonist C (see Chem. Pharm. Bull. (Tokyo) January 2002), Melatonin Agonist D (see J. Pineal Research November 2000), Melatonin Agonist E (see Chem. Pharm. Bull. (Tokyo) Febrary 2002), Melatonin Agonist F (see Reprod. Nutr. Dev. May 1999), Melatonin Agonist G (see J. Med. Chem. October 1993), Melatonin Agonist H (see Famaco March 2000), Melatonin Agonist I (see J. Med. Chem. March 2000), Melatonin Analog J (see Bioorg. Med. Chem. Lett. March 2003), Melatonin Analog K (see MedAd News September 2001), Melatonin Analog L, AH-001 (2-acetamido-8- methoxytetralin) (see U.S. Patent No. 5,151,446), GG-012 (4-methoxy-2- (methylene propylamide)indan) (see Drijfhout et al., Eur. J. Pharmacol. 1999, 382, 157-66), Enol-3-IPA, ML-23 (N-2,4-dinitrophenyl-5-methoxy-tryptamine ) (see U.S. Patent No. 4,880,826), SL-18.1616, IP-100-9 (US 5580878), Sleep Inducing Peptide A, AH-017 (see U.S. Patent No. 5,151,446), AH-002 (8-methoxy- 2-propionamido-tetralin) (see U.S. Patent No. 5,151,446), and IP-101.
Metabolites, prodrugs, stereoisomers, polymorphs, hydrates, solvates, and salts of the above compounds that are directly or indirectly active can, of course, also be used in the practice of this invention.
Melatonin agonists with a MT1R and MT2R binding profile similar to that of tasimelteon, which has 2 to 4 time greater specificity for MT2R, are preferred.
Tasimelteon can be synthesized by procedures known in the art. The preparation of a 4-vinyl-2,3-dihydrobenzofuran cyclopropyl intermediate can be carried out as described in US7754902, which is incorporated herein by reference as though fully set forth.
Pro-drugs, e.g., esters, and pharmaceutically acceptable salts can be prepared by exercise of routine skill in the art.
In patients suffering a Non-24, the melatonin and Cortisol circadian rhythms and the natural day/night cycle become desynchronized. For example, in patients suffering from a free-running circadian rhythm, melatonin and Cortisol acrophases occur more than 24 hours, e.g., >24.1 hours, prior to each previous day’s melatonin and Cortisol acrophase, respectively, resulting in desynchronization for days, weeks, or even months, depending upon the length of a patient’s circadian rhythm, before the melatonin, Cortisol, and day /night cycles are again temporarily synchronized.
Chronic misalignment of Cortisol has been associated with metabolic, cardiac, cognitive, neurologic, neoplastic, and hormonal disorders. Such disorders include, e.g., obesity, depression, neurological impairments.

INTRODUCTION
Tasimelteon has the chemical name: trans-N-[[2-(2,3-dihydrobenzofuran-4-yl)cycloprop-1yl]methyl]propanamide, has the structure of Formula I:
and is disclosed in U.S. Pat. No. 5,856,529 and in US 20090105333, both of which are incorporated herein by reference as though fully set forth.
Tasimelteon is a white to off-white powder with a melting point of about 78° C. (DSC) and is very soluble or freely soluble in 95% ethanol, methanol, acetonitrile, ethyl acetate, isopropanol, polyethylene glycols (PEG-300 and PEG-400), and only slightly soluble in water. The native pH of a saturated solution of tasimelteon in water is 8.5 and its aqueous solubility is practically unaffected by pH. Tasimelteon has 2-4 times greater affinity for MT2R relative to MT1R. It’s affinity (Ki) for MT1R is 0.3 to 0.4 and for MT2R, 0.1 to 0.2. Tasimelteon is useful in the practice of this invention because it is a melatonin agonist that has been demonstrated, among other activities, to entrain patients suffering from Non-24.
SYNTHESIS
(1R-trans)-N-[[2 – (2,3-dihydro-4 benzofuranyl) cyclopropyl] methyl] propanamide PATENT: BRISTOL-MYERS SQUIBB PRIORITY DATE: 1996 HYPNOTIC


PREPARATION OF XV
XXIV D-camphorsulfonic acid IS REACTED WITH THIONYL CHLORIDE TO GIVE
…………XXV (1S, 4R) -7,7-dimethyl-2-oxo-bicyclo [2.2.1] heptane-1-methanesulfonyl chloride
TREATED WITH
XXVI ammonium hydroxide
TO GIVE
XXVII (1S, 4R) -7,7-dimethyl-2-oxo-bicyclo [2.2.1] heptane-1-methanesulfonamide
TREATED WITH AMBERLYST15
….XXVIII (3aS, 6R) -4,5,6,7-tetrahydro-8 ,8-dimethyl-3H-3a ,6-methano-2 ,1-benzisothiazole-2 ,2-dioxide
TREATED WITH LAH, ie double bond is reduced to get
…..XV (3aS, 6R, 7aR)-hexahydro-8 ,8-dimethyl-3H-3a ,6-methano-2 ,1-benzisothiazole-2 ,2-dioxide

Intermediate
I 3-hydroxybenzoic acid methyl ester
II 3-bromo-1-propene
III 3 – (2-propenyloxy) benzoic acid methyl ester
IV 3-hydroxy-2-(2-propenyl) benzoic acid methyl ester
V 2,3-dihydro-4-hydroxy-2-benzofurancarboxylic acid methyl ester
VI benzofuran-4-carboxylic acid methyl ester
VII benzofuran-4-carboxylic acid
VIII 2,3-dihydro-4-benzofurancarboxylic acid
IX 2,3-dihydro-4-benzofuranmethanol
X 2,3-dihydro-4-benzofurancarboxaldehyde
XI Propanedioic acid
XII (E) -3 – (2,3-dihydro-4-benzofuranyl) propenoic acid
XIII thionyl chloride
XIV (E) -3 – (2,3-dihydro-4-benzofuranyl) propenoyl chloride
XV (3aS, 6R, 7aR)-hexahydro-8 ,8-dimethyl-3H-3a ,6-methano-2 ,1-benzisothiazole-2 ,2-dioxide
XVI (3aS,6R,7aR)-1-[(E)-3-(2,3-dihydro-4-benzofuranyl)-1-oxo-2-propenyl]hexahydro-8,8-dimethyl-3H-3a,6-methano-2,1-benzisothiazole-2,2-dioxide
XVII (3aS,6R,7aR)-1-[[(1R,2R)-2-(2,3-dihydro-4-benzofuranyl)cyclopropyl]carbonyl]hexahydro-8,8-dimethyl-3H-3a,6-methano-2,1-benzisothiazole-2,2-dioxide
XVIII [R-(R *, R *)] -2 – (2,3-dihydro-4-benzofuranyl) cyclopropanemethanol
XIX [R-(R *, R *)] -2 – (2,3-dihydro-4-benzofuranyl) cyclopropanecarboxaldehyde
XX hydroxylamine hydrochloride
XXI [R-(R *, R *)] -2 – (2,3-dihydro-4-benzofuranyl) cyclopropanecarbaldehyde oxime
XXII [R-(R *, R *)] -2 – (2,3-dihydro-4-benzofuranyl) cyclopropanemethanamine
XXIII propanoyl chloride
XXIV D-camphorsulfonic acid
XXV (1S, 4R) -7,7-dimethyl-2-oxo-bicyclo [2.2.1] heptane-1-methanesulfonyl chloride
XXVI ammonium hydroxide
XXVII (1S, 4R) -7,7-dimethyl-2-oxo-bicyclo [2.2.1] heptane-1-methanesulfonamide
XXVIII (3aS, 6R) -4,5,6,7-tetrahydro-8 ,8-dimethyl-3H-3a ,6-methano-2 ,1-benzisothiazole-2 ,2-dioxide
Bibliography
– Patents: Benzofuran and dihydrobenzofuran melatonergic agents: US5856529 (1999)
Priority: US19960032689P, 10 Dec. 1996 (Bristol-Myers Squibb Company, U.S.)
– Preparation III (quinazolines): US2004044015 (2004) Priority: EP20000402845, 13 Oct. 2000
– Preparation of VII (aminoalkylindols): Structure-Activity Relationships of Novel Cannabinoid Mimetics Eissenstat et al, J.. Med. Chem. 1995, 38, 3094-3105
– Preparation XXVIII: Towson et al. Organic Syntheses, Coll. Vol. 8, p.104 (1993) Vol. 69, p.158 (1990)
– Preparation XV: Weismiller et al. Organic Syntheses, Coll. Vol. 8, p.110 (1993) Vol. 69, p.154 (1990).
– G. Birznieks et al. Melatonin agonist VEC-162 Improves sleep onset and maintenance in a model of transient insomnia. Sleep 2007, 30, 0773 Abstract.
-. Rajaratnam SM et al, The melatonin agonist VEC-162 Phase time immediately advances the human circadian system, Sleep 2006, 29, 0159 Abstract.
-. AK Singh et al, Evolution of a manufacturing route for a highly potent drug candidate, 229th ACS Natl Meet, March 13-17, 2005, San Diego, Abstract MEDI 576.
– Vachharajani NN et al, Preclinical pharmacokinetics and metabolism of BMS-214778, a novel melatonin receptor agonist, J Pharm Sci. 2003 Apr; 92 (4) :760-72.
. – JW Scott et al, Catalytic Asymmetric Synthesis of a melotonin antagonist; synthesis and process optimization. 223rd ACS Natl Meet, April 7-11, Orlando, 2002, Abstract ORGN 186.
SYNTHESIS CONSTRUCTION AS IN PATENT
GENERAL SCHEMES
Reaction Scheme 1
The syntheses of the 4-aryl-propenoic acid derivatives, 2 and 3, are shown in Reaction Scheme 1. The starting aldehydes, 1 , can be prepared by methods well known to those skilled in the art. Condensation of malonic acid with the aldehydes, 1, in solvents such as pyridine with catalysts such as piperidine or pyrrolidine, gives the 4-aryl- propenoic acid, 2. Subsequent conversion of the acid to the acid chloride using reagents such as thionyl chloride, phosphoryl chloride, or the like, followed by reaction with N,0-dimethyl hydroxylamine gives the amide intermediate 3 in good yields. Alternatively, aldehyde 1 can be converted directly to amide 3 using reagents such as diethyl (N-methoxy- N-methyl-carbamoylmethyl)phosphonate with a strong base such as sodium hydride.
Reaction Scheme 2
The conversion of the amide intermediate 3 to the racemic, trans- cyclopropane carboxaldehyde intermediate, 4, is shown in Reaction Scheme 2. Intermediate 3 was allowed to react with cyclopropanating reagents such as trimethylsulfoxonium iodide and sodium hydride in solvents such as DMF, THF, or the like. Subsequent reduction using reagents such as LAH in solvents such as THF, ethyl ether, or the like, gives the racemic, trans-cyclopropane carboxaldehyde intermediates, 4.
Reaction Scheme 3
Racemic cyclopropane intermediate 5 (R = halogen) can be prepared from intermediate 2 as shown in Reaction Scheme 3. Intermediate 2 was converted to the corresponding allylic alcohol by treatment with reducing agents such as sodium borohydride plus iodine in solvents such as THF. Subsequent acylation using reagents such as acetic anhydride in pyridine or acetyl chloride gave the allylic acetate which was allowed to react with cyclopropanating reagents such as sodium chloro-difluoroacetate in diglyme to provide the racemic, trans- cyclopropane acetate intermediates, 5. Reaction Scheme 4
The conversion of the acid 2 to the chiral cyclopropane carboxaldehyde intermediate, (-)-(trans)-4, is shown in Reaction Scheme 4. Intermediate 2 is condensed with (-)-2,10-camphorsultam under standard conditions, and then cyclopropanated in the presence of catalysts such as palladium acetate using diazomethane generated from reagents such as 1-methyl-3-nitro-1-nitrosoguanidine. Subsequent reduction using reagents such as LAH in solvents such as THF, followed by oxidation of the alcohol intermediates using reagents such as DMSO/oxalyl chloride, or PCC, gives the cyclopropane carboxaldehyde intermediate, (-)-(trans)-4, in good yields. The enantiomer, (+)-(trans)-4, can also be obtained employing a similar procedure using (+)-2,10- camphorsultam in place of (-)-2,10-camphorsultam.
When it is desired to prepare compounds of Formula I wherein m = 2, the alcohol intermediate may be activated in the conventional manner such as with mesyl chloride and treated with sodium cyanide followed by reduction of the nitrile group with a reducing agent such as LAH to produce the amine intermediate 6.
Reaction Scheme 5
Reaction Scheme 5 shows the conversion of intermediates 4 and 5 to the amine intermediate, 7, and the subsequent conversion of 6. or 7 to compounds of Formula I. The carboxaldehyde intermediate, 4, is condensed with hydroxylamine and then reduced with reagents such as LAH to give the amine intermediate, 7. The acetate intermediate 5 is hydrolyzed with potassium hydroxide to the alcohol, converted to the mesylate with methane sulfonyl chloride and triethyl amine in CH2CI2and then converted to the azide by treatment with sodium azide in solvents such as DMF. Subsequent reduction of the azide group with a reducing agent such as LAH produced the amine intermediate 7. Further reaction of 6 or 7 with acylating reagents gives compounds of Formula I. Suitable acylating agents include carboxylic acid halides, anhydrides, acyl imidazoles, alkyl isocyanates, alkyl isothiocyanates, and carboxylic acids in the presence of condensing agents, such as carbonyl imidazole, carbodiimides, and the like. Reaction Scheme 6
Reaction Scheme 6 shows the alkylation of secondary amides of Formula I (R2 = H) to give tertiary amides of Formula I (R2 = alkyl). The secondary amide is reacted with a base such as sodium hydride, potassium tert-butoxide, or the like, and then reacted with an alkylating reagent such as alkyl halides, alkyl sulfonate esters, or the like to produce tertiary amides of Formula I.
Reaction Scheme 7
Reaction Scheme 7 shows the halogenation of compounds of Formula I. The carboxamides, i (Q1 = Q2 = H), are reacted with excess amounts of halogenating agents such as iodine, N-bromosuccinimide, or the like to give the dihalo-compounds of Formula I (Q1 = Q2 = halogen). Alternatively, a stoichiometric amount of these halogenating agents can be used to give the monohalo-compounds of Formula I (Q1 = H, Q2 = halogen; or Q1 = halogen, Q2 = H). In both cases, additives such as lead IV tetraacetate can be used to facilitate the reaction. Biological Activity of the Compounds
The compounds of the invention are melatonergic agents. They have been found to bind human melatonergic receptors expressed in a stable cell line with good affinity. Further, the compounds are agonists as determined by their ability, like melatonin, to block the forskolin- stimulated accumulation of cAMP in certain cells. Due to these properties, the compounds and compositions of the invention should be useful as sedatives, chronobiotic agents, anxiolytics, antipsychotics, analgesics, and the like. Specifically, these agents should find use in the treatment of stress, sleep disorders, seasonal depression, appetite regulation, shifts in circadian cycles, melancholia, benign prostatic hyperplasia and related conditions
EXPERIMENTAL PROCEDURES
SEE ORIGINAL PATENT FOR CORECTIONS
Preparation 1
Benzofuran-4-carboxaldehyde
Step 1 : N-Methoxy-N-methyl-benzofuran-4-carboxamide
A mixture of benzofuran-4-carboxylic acid [Eissenstat, et al.. J. Medicinal Chemistry, 38 (16) 3094-3105 (1995)] (2.8 g, 17.4 mmol) and thionyl chloride (25 mL) was heated to reflux for 2 h and then concentrated in vacuo. The solid residue was dissolved in ethyl acetate (50 mL) and a solution of N,O-dimethylhydroxylamine hydrochloride (2.8 g) in saturated NaHC03(60 mL) was added with stirring. After stirring for 1.5 h, the ethyl acetate layer was separated. The aqueous layer was extracted with ethyl acetate. The ethyl acetate extracts were combined, washed with saturated NaHCO3 and concentrated in vacuo to give an oil (3.2 g, 95.4%).
Step 2: Benzofuran-4-carboxaldehyde
A solution of N-methoxy-N-methyl-benzofuran-4-carboxamide (3.2 g, 16.6 mmol) in THF (100 mL) was cooled to -45°C and then LAH (0.7 g, 18.7 mmol) was added. The mixture was stirred for 15 min, allowed to warm to -5°C, and then recooled to -45°C. Saturated KHS04 (25 mL) was added with vigorous stirring, and the mixture was allowed to warm to room temperature. The precipitate was filtered and washed with acetone. The filtrate was concentrated in vacuo to give an oil (2.3 g, 94%). Preparation 2
2,3-Dihydrobenzofuran-4-carboxaldehyde
Step 1 : 2,3-Dihydrobenzofuran-4-carboxylic acid
Benzofuran-4-carboxylic acid (10.0 g, 61 .7 mmol) was hydrogenated (60 psi) in acetic acid (100 mL) over 10% Pd/C (2 g) for 12 hr. The mixture was filtered and the filtrate was diluted with water (500 mL) to give 2,3- dihydrobenzofuran-4-carboxylic acid as a white powder (8.4 g, 83%). A sample was recrystallized from isopropanol to give fine white needles (mp: 185.5-187.5°C).
Step 2: (2,3-Dihydrobenzofuran-4-yl)methanol
A solution of 2,3-dihydrobenzofuran-4-carboxylic acid (10 g, 61 mmol) in THF (100 mL) was stirred as LAH (4.64 g, 122 mmol) was slowly added. The mixture was heated to reflux for 30 min. The mixture was cooled and quenched cautiously with ethyl acetate and then with 1 N HCI (150 mL). The mixture was then made acidic with 12 N HCI until all the inorganic precipitate dissolved. The organic layer was separated, and the inorganic layer was extracted twice with ethyl acetate. The organic layers were combined, washed twice with brine, and then concentrated in vacuo. This oil was Kϋgelrohr distilled to a clear oil that crystallized upon cooling (8.53 g, 87.6%).
Step 3: 2.3-Dihydrobenzofuran-4-carboxaldehyde
DMSO (8.10 mL, 1 14 mmol) was added at -78°C to a stirred solution of oxalyl chloride in CH2CI2 (40 mL of a 2M solution). A solution of (2,3- dihydrobenzofuran-4-yl)methanol (8.53 g, 56.9 mmol) in CH2CI2 (35 mL) was added dropwise, and the solution stirred at -78°C for 30 min. Triethyl amine (33 mL, 228 mmol) was added cautiously to quench the reaction. The resulting suspension was stirred at room temperature for 30 min and diluted with CH2CI2 (100 mL). The organic layer was washed three times with water, and twice with brine, and then concentrated in vacuo to an oil (8.42 g, 100%) that was used without purification.
Preparation 16
(±)-(trans)-2-(2,3-Dihyd robenzofuran-4-yl)cyclopropane- carboxaldehyde
Step 1 : (±Htrans)-N-Methoxy-N-methyl-2-(2.3-dihydrobenzofuran-4- yhcyclopropanecarboxamide
Trimethylsulfoxonium iodide (9.9 g, 45 mmol) was added in small portions to a suspension of sodium hydride (1 .8 g, 45 mmol) in DMF (120 mL). After the foaming had subsided (10 min), a solution of (trans)- N-methoxy-N-methyl-3-(2,3-dihydrobenzofuran-4-yl)propenamide (3.5 g, 15 mmol) in DMF (60 mL) was added dropwise, with the temperature maintained between 35-40°C. The mixture was stirred for 3 h at room temperature. Saturated NH4CI (50 mL) was added dropwise and the mixture was extracted three times with ethyl acetate. The organic extracts were combined, washed with H2O and brine, dried over K2CO3, and concentrated in vacuo to give a white wax (3.7 g, 100%).
Step 2: (±)-(trans)- 2-(2.3-Dihydrobenzofuran-4-yl)cyclopropane- carboxaldehyde
A solution of (±)-(trans)-N-methoxy-N-methyl-2-(2,3-dihydrobenzofuran- 4-yl)cyclopropanecarboxamide (3.7 g, 15 mmol) in THF (10 mL) was added dropwise to a rapidly stirred suspension of LAH (683 mg, 18 mmol) in THF (50 mL) at -45°C, maintaining the temperature below -40°C throughout. The cooling bath was removed, the reaction was allowed to warm to 5°C, and then the reaction was immediately recooled to -45°C. Potassium hydrogen sulfate (3.4 g, 25.5 mmol) in H20 (50 mL) was cautiously added dropwise, the temperature maintained below – 30°C throughout. The cooling bath was removed and the suspension was stirred at room temperature for 30 min. The mixture was filtered through Celite and the filter cake was washed with ether. The combined filtrates were then washed with cold 1 N HCI, 1 N NaOH, and brine. The filtrates were dried over MgSO4, and concentrated in vacuo to give a clear oil (2.6 g, 99%).
Preparation 18
(-)-(trans)-2-(2.3-Dihydrobenzofuran-4-yl)cyclopropane-carboxaldehyde
Step 1 : (-Htrans)-N-[3-(2.3-Dihvdrobenzofuran-4-yl)-propenoyll-2.10- camphorsultam
To a solution of (-)-2,10-camphorsultam (8.15 g, 37.9 mmol) in 50 mL toluene at 0°C was added sodium hydride (1.67 g, 41.7 mmol). After stirring for 0.33 h at 0°C and 0.5 h at 20°C and recooling to 0°C, a solution of 3-(2,3-dihydrobenzofuran-4-yl)-2-propenoyl chloride
(37.9 mmol), prepared in situ from the corresponding acid and thionyl chloride (75 mL), in toluene (50 mL), was added dropwise. After stirring for 18 h at 20°C, the mixture was diluted with ethyl acetate and washed with water, 1 N HCI, and 1 N NaOH. The organic solution was dried and concentrated in vacuo to give 15.8 g of crude product. Recrystallization form ethanol-methanol (600 mL, 1 :1) gave the product (13.5 g, 92%, mp 199.5-200°C).
Step 2: (-)-N-[[(trans)-2-(2,3-Dihydrobenzofuran-4-yl)-cyclopropylj- carbonylj-2, 10-camphorsultam
1 -Methyl-3-nitro-1 -nitrosoguanidine (23.88g 163 mmol) was added in portions to a mixture of 10 N sodium hydroxide (60 mL) and ether (200 mL) at 0°C. The mixture was shaken vigorously for 0.25 h and the ether layer carefully decanted into a solution of (-)-N-[3-(2,3-dihydrobenzofuran-4-yl)-2-propenoyl]-2,10-camphorsultam (9.67 g, 25 mmol) and palladium acetate (35 mg) in methylene chloride (200 mL). After stirring for 18 h, acetic acid (5 mL) was added to the reaction and the mixture stirred for 0.5 h. The mixture was washed with 1 N HCI, 1 N NaOH and brine. The solution was dried, concentrated in vacuo and the residue crystallized twice from ethanol to give the product (6.67 g, 66.5%, mp 157-159°C).
Step 3: (-)-(trans)-2-(2,3-Dihydrobenzofuran-4-yl)cyclopropane- methanol
A solution of (-)-N-[(trans)-2-(2,3-dihydrobenzofuran-4-yl)cyclo-propanecarbonylj-2,10-camphorsultam (4.3 g, 10.7 mmol) in THF (50 mL) was added dropwise to a mixture of LAH (0.81 g, 21.4 mmol) in THF (50 mL) at -45°C. The mixture was stirred for 2 hr while it warmed to 10°C. The mixture was recooled to -40°C and hydrolyzed by the addition of saturated KHS0 (20 mL). The mixture was stirred at room temperature for 30 minutes and filtered. The precipitate was washed twice with acetone. The combined filtrate and acetone washes were concentrated in vacuo. The gummy residue was dissolved in ether, washed with 1 N NaOH and 1 N HCI, and then dried in vacuo to give the product (2.0 g, 98.4%).
Step 4: (-)-(trans)-2-(2.3-Dihydrobenzofuran-4-yl)cyclopropane- carboxaldehyde DMSO (1.6 g, 21 mmol) was added to oxalyl chloride in CH2CI2(7.4 mL of 2 M solution, 14.8 mmole) at -78°C. The (-)-(trans)-2-(2,3-dihydrobenzofuran-4-yl)-cyclopropylmethanol (2.0 g, 10.5 mmol) in CH2CI2(15 mL) was added. The mixture was stirred for 20 min and then triethylamine (4.24 g, 42 mmol) was added. The mixture was warmed to room temperature and stirred for 30 min. The mixture was diluted with CH2CI2 and washed with water, 1 N HCI, and then 1 N NaOH. The organic layer was dried and concentrated iι> vacuo to give the aldehyde product (1.98 g, 100%).
Preparation 24
(-)-(trans)-2-(2.3-Dihydrobenzofuran-4-yl)cyclopropane-methanamine A mixture of (-)-(trans)-2-(2,3-dihydrobenzofuran-4-yl)cyclopropane-carboxaldehyde (1.98 g, 10.5 mmol), hydroxylamine hydrochloride (2.29 g, 33 mmol), and 30% NaOH (3.5 mL, 35 mmol), in 5:1
ethanol/water (50 mL) was heated on a steam bath for 2 h. The solution was concentrated in vacuo. and the residue mixed with water. The mixture was extracted with CH2CI2. The organic extracts were dried and concentrated in vacuo to give a solid which NMR analysis showed to be a mixture of the cis and trans oximes. This material was dissolved in THF (20 mL) and added to solution of alane in THF [prepared from LAH (1.14 g, 30 mmol) and H2S04 (1.47 g, 15 mmol) at 0°Cj. The reaction was stirred for 18 h, and quenched successively with water (1.15 mL), 15% NaOH (1.15 mL), and then water (3.45 mL). The mixture was filtered and the filtrate was concentrated in vacuo. The residue was mixed with ether and washed with water and then 1 N HCI. The acid washes were made basic and extracted with CH2CI . The extracts were dried and concentrated in vacuo to give the amine product (1.4 g, 70.5%). The amine was converted to the fumarate salt in ethanol (mp: 197-198°C).
Anal. Calc’d for C12H15NO • C4H404: C, 62.94; H, 6.27; N, 4.59.
Found: C, 62.87; H, 6.31 ; N, 4.52.
FINAL PRODUCT TASIMELTEON
Example 2
(-)-(trans)-N-[[2-(2,3-Dihydrobenzofuran-4-yl)cycloprop-1-yl]methyl]propanamide
This compound was prepared similar to the above procedure using propionyl chloride and (-)-(trans)-2-(2,3-dihydrobenzofuran-4-yl)- cyclopropanemethanamine to give an oil that solidified upon standing to an off-white solid (61 %, mp: 71-72°C). IR (NaCI Film): 3298, 1645, 1548, 1459, 1235 cm“1.
Mo5 : -17.3°
Anal. Calc’d for C15H19N02: C, 73.44; H, 7.87; N, 5.71 . Found: C, 73.28; H, 7.68; N, 5.58
SYNTHESIS
Synthesis Path
SYN

Tasimelteon (Hetlioz)Tasimelteon, which is marketed by Vanda Pharmaceuticals as Hetlioz and developed in partnership with Bristol-Myers Squibb,is a drug that was approved by the US FDA in January 2014 for the treatment of non-24-hour sleep–wake disorder (also called Non-24, N24 and N24HSWD).234 Tasimelteon is a melatonin MT1
and MT2 receptor agonist; because it exhibits a greater affinity to the MT2 receptor than MT1, is also known as Dual Melatonin
Receptor Agonist.234 Two randomized controlled trials (phases II
and III) demonstrated that tasimelteon improved sleep latency
and maintenance of sleep with a shift in circadian rhythms, and
therefore has the potential to treat patients with transient insomnia
associated with circadian rhythm sleep disorders.235 Preclinical
studies showed that the drug has similar phase-shifting properties
to melatonin, but with less vasoconstrictive effects.236 The most
likely scale preparation of the drug, much of which has been published
in the chemical literature, is described below in Scheme 44.
Activation of commercial bis-ethanol 250 with 2.5 equivalents
of the Vilsmeier salt 251 followed by treatment with base resulted
an intramolecular cyclization reaction with the proximal phenol
and concomitant elimination of the remaining imidate to deliver
the vinylated dihydrobenzofuran 252 in 76% yield.237 Interestingly,
this reaction could be performed on multi-kilogram scale, required
no chromatographic purification, and generated environmentallyfriendly
DMF and HCl as byproducts.237 Sharpless asymmetric
dihydroxylation of olefin 252 delivered diol 253 in 86% yield and
impressive enantioselectivity (>99% ee). This diol was then activated
with trimethylsilyl chloride and then treated with base to generate epoxide 254.238 Next, a modified Horner–Wadsworth–
Emmons reaction involving triethylphosphonoacetate (TEPA, 255)
was employed to convert epoxide 254 to cyclopropane 256.239
The reaction presumably proceeds through removal of the acidic
TEPA proton followed by nucleophilic attack at the terminal epoxide
carbon. The resulting alkoxide undergoes an intramolecular
phosphoryl transfer reaction resulting in an enolate, which then attacked the newly formed phosphonate ester in an SN2 fashion
resulting in the trans-cyclopropane ester, which was ultimately
saponified and re-acidified to furnish cyclopropane acid 256.239
Conversion of this acid to the corresponding primary amide preceded
carbonyl reduction with sodium borohydride. The resulting
amine was acylated with propionyl chloride to furnish tasimelteon
(XXXI) as the final product in 86% yield across the four-step
sequence.
PATENTS
| US2010261786 | 10-15-2010 | PREDICTION OF SLEEP PARAMETER AND RESPONSE TO SLEEP-INDUCING COMPOUND BASED ON PER3 VNTR GENOTYPE |
| US2009209638 | 8-21-2009 | TREATMENT FOR DEPRESSIVE DISORDERS |
| US6060506 | 5-10-2000 | Benzopyran derivatives as melatonergic agents |
| US5981571 | 11-10-1999 | Benzodioxa alkylene ethers as melatonergic agents |
| WO9825606 | 6-19-1998 | BENZODIOXOLE, BENZOFURAN, DIHYDROBENZOFURAN, AND BENZODIOXANE MELATONERGIC AGENTS |
| WO2007137244A1 * | May 22, 2007 | Nov 29, 2007 | Gunther Birznieks | Melatonin agonist treatment |
| US4880826 | Jun 25, 1987 | Nov 14, 1989 | Nava Zisapel | Melatonin antagonist |
| US4997845 | May 10, 1990 | Mar 5, 1991 | Eli Lilly And Company | β-alkylmelatonins as ovulation inhibitors |
| US5093352 | May 16, 1990 | Mar 3, 1992 | Whitby Research, Inc. | Antidepressant agents |
| US5151446 | Mar 28, 1991 | Sep 29, 1992 | Northwestern University | Substituted 2-amidotetralins as melatonin agonists and antagonists |
| US5225442 | Jan 3, 1992 | Jul 6, 1993 | Adir Et Compagnie | Compounds having a naphthalene structure |
| US5580878 | Jun 7, 1995 | Dec 3, 1996 | Interneuron Pharmaceuticals, Inc. | Substituted tryptamines phenalkylamines and related compounds |
| US5856529 | Dec 9, 1997 | Jan 5, 1999 | Bristol-Myers Squibb Company | Benzofuran and dihydrobenzofuran melatonergic agents |
| US6211225 | Jun 6, 2000 | Apr 3, 2001 | Bristol-Meyers Squibb | Heterocyclic aminopyrrolidine derivatives as melatonergic agents |
| US7754902 | May 18, 2006 | Jul 13, 2010 | Vanda Pharmaceuticals, Inc. | Ruthenium(II) catalysts for use in stereoselective cyclopropanations |
| US20010047016 | Apr 12, 2001 | Nov 29, 2001 | Gregory Oxenkrug | Method for treating depression |
| US20050164987 | Dec 22, 2004 | Jul 28, 2005 | Barberich Timothy J. | Melatonin combination therapy for improving sleep quality |
| US20090105333 | May 22, 2007 | Apr 23, 2009 | Gunther Birznieks | Melatonin agonist treatment |
extra info
 |
- Department of Chemistry, Drexel University, Philadelphia, PA 19104.
- Shriner, R. L.; Shotton, J. A.; Sutherland, H. J. Am. Chem. Soc.1938, 60, 2794.
- Oppolzer, W.; Chapuis, C.; Bernardinelli, G. Helv. Chim. Acta1984, 67, 1397.
- Vandewalle, M.; Van der Eycken, J.; Oppolzer, W.; Vullioud, C. Tetrahedron1986, 42, 4035.
- Davis, F. A.; Towson, J. C.; Weismiller, M. C.; Lal, G.; Carroll,, P. J. J. Am. Chem. Soc.1988, 110, 8477.
- Oppolzer, W. Tetrahedron1987, 43, 1969.
- Oppolzer, W.; Mills, R. J.; Pachinger, W.; Stevenson, T. Helv. Chim. Acta1986, 69, 1542; Oppolzer, W.; Schneider, P. Helv. Chim. Acta1986, 69, 1817; Oppolzer, W.; Mills, R. J.; Réglier, M. Tetrahedron Lett.1986, 27, 183; Oppolzer, W.; Poli. G.Tetrahedron Lett.1986, 27, 4717; Oppolzer, W.; Poli, G.; Starkemann, C.; Bernardinelli, G. Tetrahedron Lett.1988, 29, 3559.
- Oppolzer, W.; Barras, J-P. Helv. Chim. Acta1987, 70, 1666.
- Curran, D. P.; Kim, B. H.; Daugherty, J.; Heffner, T. A. Tetrahedron Lett.1988, 29, 3555.
- Differding, E.; Lang, R. W. Tetrahedron Lett.1988, 29, 6087.
 |
This preparation is referenced from:
- Org. Syn. Coll. Vol. 8, 110
- Org. Syn. Coll. Vol. 9, 212
-
References and Notes
- Department of Chemistry, Drexel University, Philadelphia, PA 19104.
- Reychler, M. A. Bull. Soc. Chim. III1889, 19, 120.
- Armstrong, H. E.; Lowry, T. M. J. Chem. Soc., Trans.1902, 81, 1441.
- Dauphin, G.; Kergomard, A.; Scarset, A. Bull. Soc. Chim. Fr.1976, 862.
- Davis, F. A.; Jenkins, Jr., R. H.; Awad, S. B.; Stringer, O. D.; Watson, W. H.; Galloy, J. J. Am. Chem. Soc.1982, 104, 5412.
- Vandewalle, M.; Van der Eycken, J.; Oppolzer, W.; Vullioud, C. Tetrahedron, 1986, 42, 4035.
- Davis, F. A.; Towson, J. C.; Weismiller, M. C.; Lal, S.; Carroll, P. J. J. Am. Chem. Soc.1988, 110, 8477.
- Davis, F. A.; Weismiller, M. C.; Lal, G. S.; Chen, B. C.; Przeslawski, R. M. Tetrahedron Lett., 1989, 30, 1613.
- Oppolzer, W. Tetrahedron1987, 43, 1969.
- Glahsl, G.; Herrmann, R. J. Chem. Soc., Perkin Trans. I1988, 1753.
- Differding, E.; Lang, R. W. Tetrahedron Lett.1988, 29, 6087.
- For recent reviews on the chemistry of N-sulfonyloxaziridines, see: (a) Davis, F. A.; Jenkins, Jr., R. H. in “Asymmetric Synthesis,” Morrison, J. D., Ed.; Academic Press: Orlando, FL, 1984, Vol. 4, Chapter 4;
- Davis, F. A.; Haque, S. M. in “Advances in Oxygenated Processes,” Baumstark, A. L., Ed.; JAI Press: London, Vol. 2;
- Davis, F. A.; Sheppard, A. C. Tetrahedron1989, 45, 5703.
- Davis, F. A.; McCauley, Jr., J. P.; Chattopadhyay, S.; Harakal, M. E.; Towson, J. C.; Watson, W. H.; Tavanaiepour, I. J. Am. Chem. Soc.1987, 109, 3370.
- Davis, F. A.; Stringer, O. D.; McCauley, Jr., J. M. Tetrahedron1985, 41, 4747.
- Davis, F. A.; Chattopadhyay, S. Tetrahedron Lett.1986, 27, 5079.
- Davis, F. A.; Harakal, M. E.; Awad, S. B. J. Am. Chem. Soc.1983, 105, 3123.
- Davis, F. A.; Wei, J.; Sheppard, A. C.; Gubernick S. Tetrahedron Lett.1987, 28, 5115.
- Davis, F. A.; Lal, G. S.; Wei, J. Tetrahedron Lett.1988, 29, 4269.
- Davis, F. A.; Haque, M. S.; Ulatowski, T. G.; Towson, J. C. J. Org. Chem.1986, 51, 2402.
- Davis, F. A.; Haque, M. S. J. Org. Chem.1986, 51, 4083; Davis, F. A.; Haque, M. S.; Przeslawski, R. M. J. Org. Chem.1989, 54, 2021.
- Davis, F. A.; Ulatowski, T. G.; Haque, M. S. J. Org. Chem.1987, 52, 5288.
- Davis, F. A.; Sheppard, A. C., Lal, G. S. Tetrahedron Lett.1989, 30, 779.
- Davis, F. A.; Sheppard, A. C.; Chen, B. C.; Haque, M. S. J. Am. Chem. Soc.1990, 112, 6679.
a US 5 856 529 (Bristol-Myers Squibb; 5.1.1999; appl. 9.12.1997; USA-prior. 10.12.1996).
-
- b US 7 754 902 (Vanda Pharms.; 13.7.2010; appl. 18.5.2006).
-
treatment of circadian rhythm disorders:
- US 8 785 492 (Vanda Pharms.; 22.7.2014; appl. 25.1.2013; USA-prior. 26.1.2012).
-
synthesis cis-isomer:
- US 6 214 869 (Bristol-Myers Squibb; 10.4.2001; appl. 25.5.1999; USA-prior. 5.6.1998).
Patents
- USUS5856529 A
- USUS8785492 B2
- US5856529
- US8785492
- US9060995
- US9549913
- US9539234
- US9730910
- USRE46604
- US9855241
References
- Jump up^ “Tasimelteon Advisory Committee Meeting Briefing Materials”(PDF). Vanda Pharmaceuticals Inc. November 2013.
- Jump up^ “FDA transcript approval minutes” (PDF). FDA. November 14, 2013.
- ^ Jump up to:a b Food and Drug Administration (January 31, 2014). “FDA approves Hetlioz: first treatment for non-24 hour sleep-wake disorder”. FDA.
- Jump up^ “tasimelteon (Hetlioz) UKMi New Drugs Online Database”. Retrieved August 6, 2014.
- Jump up^ “HETLIOZ® Receives European Commission Approval for the Treatment of Non-24-Hour Sleep-Wake Disorder in the Totally Blind”. MarketWatch. PR Newswire. 7 July 2015. Retrieved 8 July 2015.
- Jump up^ Vachharajani, Nimish N.; Yeleswaram, Krishnaswamy; Boulton, David W. (April 2003). “Preclinical pharmacokinetics and metabolism of BMS-214778, a novel melatonin receptor agonist”. Journal of Pharmaceutical Sciences. 92 (4): 760–72. doi:10.1002/jps.10348. PMID 12661062.
- Jump up^ Sack, R. L.; Brandes, R. W.; Kendall, A. R.; Lewy, A. J. (2000). “Entrainment of Free-Running Circadian Rhythms by Melatonin in Blind People”. New England Journal of Medicine. 343 (15): 1070–7. doi:10.1056/NEJM200010123431503. PMID 11027741.
- Jump up^ “Safety and Efficacy of VEC-162 on Circadian Rhythm in Healthy Adult Volunteers”. ClinicalTrials.gov. |accessdate=May 15, 2014
- Jump up^ “VEC-162 Study in Healthy Adult Volunteers in a Model of Insomnia”. ClinicalTrials.gov. Retrieved May 15, 2014.
- Jump up^ “VEC-162 Study in Adult Patients With Primary Insomnia”. ClinicalTrials.gov. Retrieved May 15, 2014.
- Jump up^ Lynne Lamberg. “Improving Sleep and Alertness in the Blind (Part 5)”. Matilda Ziegler Magazine for the Blind. Retrieved May 15, 2014.
- Jump up^ Shantha MW Rajaratnam; Mihael H Polymeropoulos; Dennis M Fisher; Thomas Roth; Christin Scott; Gunther Birznieks; Elizabeth B Klerman (2009-02-07). “Melatonin agonist tasimelteon (VEC-162) for transient insomnia after sleep-time shift: two randomised controlled multicentre trials”. The Lancet. 373 (9662): 482–491. doi:10.1016/S0140-6736(08)61812-7. PMID 19054552. Retrieved 2010-02-23.
- Jump up^ Carome, Michael (1 July 2015). “Outrage of the Month: FDA Makes Major Blunder After Approving Drug for Rare Sleep Disorder”. Huffington Post. Retrieved 8 July 2015.
- Jump up^ Food and Drug Administration (January 31, 2014). “FDA NEWS RELEASE: FDA approves Hetlioz: first treatment for non-24 hour sleep–wake disorder in blind individuals”. FDA.
- Jump up^ “Side Effects Drug Center: Hetlioz Clinical Pharmacology”. RxList. February 10, 2014.
- Jump up^ “Side Effects Drug Center: Hetlioz Warnings and Precautions”. RxList. February 10, 2014.
In animal studies, administration of tasimelteon during pregnancy resulted in developmental toxicity (embryofetal mortality, neurobehavioral impairment, and decreased growth and development in offspring) at doses greater than those used clinically.
 |
|
 |
|
| Clinical data | |
|---|---|
| Trade names | Hetlioz |
| License data | |
| Pregnancy category |
|
| Routes of administration |
Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | not determined in humans[1] |
| Protein binding | 89–90% |
| Metabolism | extensive hepatic, primarily CYP1A2 and CYP3A4-mediated |
| Elimination half-life | 0.9–1.7 h / 0.8–5.9 h (terminal) |
| Excretion | 80% in urine, 4% in feces |
| Identifiers | |
| CAS Number | |
| PubChemCID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| ECHA InfoCard | 100.114.889 |
| Chemical and physical data | |
| Formula | C15H19NO2 |
| Molar mass | 245.32 g/mol |
| 3D model (JSmol) | |

DR ANTHONY MELVIN CRASTO Ph.D
GLENMARK SCIENTIST , NAVIMUMBAI, INDIA
Chemical and physical properties
Tasimelteon has two stereogenic centers. Besides the medically used trans-1 R , 2 R isomer (in the picture above left), there are thus three further stereoisomers that do not arise in the synthesis.
Tasimelteon is a white to off-white crystalline non-hygroscopic substance, soluble in water at physiologically relevant pH levels and readily soluble in alcohols, cyclohexane and acetonitrile. The compound occurs in two crystal forms. It is an anhydrate melting at 74 ° C and a hemihydrate . [4] The hemihydrate is from about 35 ° C the water of hydration and converts thereby in the anhydrate form to. [4] The anhydrate crystallizes in a monoclinic lattice with the space group P 2 1 , and the hemihydrate crystallizes in a tetragonal lattice with the space group P 4 3 21 2. [4]
4 Kaihang Liu, Zhou Xinbo, Zhejing Xu, Bai Hongzhen, Jianrong Zhu Jianming Gu, Guping Tang, Liu Xingang, Hu Xiurong: anhydrate and hemihydrate of Tasimelteon: Synthesis, structure, and pharmacokinetic study in J. Pharm. Biomed. Anal. 151 (2018) 235-243, doi : 10.1016 / j.jpba.2017.12.035 .
Glycopyrrolate Tosylate

Glycopyrrolate Tosylate
| Molecular Formula: | C26H35NO6S |
|---|---|
| Molecular Weight: | 489.627 g/mol |
(1,1-dimethylpyrrolidin-1-ium-3-yl) 2-cyclopentyl-2-hydroxy-2-phenylacetate;4-methylbenzenesulfonate
CAS 873295-46-6 , C19 H28 N O3 . C7 H7 O3 S, Pyrrolidinium, 3-[(2-cyclopentyl-2-hydroxy-2-phenylacetyl)oxy]-1,1-dimethyl-, 4-methylbenzenesulfonate (1:1)
Glycopyrronium tosylate monohydrate
Molecular Formula, C19-H28-N-O3.C7-H8-O3-S.H2-O, Molecular Weight, 508.6522
https://chem.nlm.nih.gov/chemidplus/structure/1624259-25-1?maxscale=30&width=300&height=300
CAS 1624259-25-1, C19 H28 N O3 . C7 H7 O3 S . H2 O, Pyrrolidinium, 3-[(2-cyclopentyl-2-hydroxy-2-phenylacetyl)oxy]-1,1-dimethyl-, 4-methylbenzenesulfonate, hydrate (1:1:1)
Dermira (Originator)
DRM-04
DRM-04B
- DRM-04 tosylate monohydrate
- DRM04
- DRM04 tosylate
- Glycopyrronium tosylate
- Glycopyrronium tosylate monohydrate
- Glycopyrronium tosylate [USAN]
- UNII-1PVF6JLU7B
- UNII-X2N5209428
In 2018, the product was approved in the U.S. for the treatment of primary axillary hyperhidrosis in adult and pediatric patients 9 years of age and older.
In 2016, Maruho signed an exclusive license agreement with Dermina for product development and marketing in Japan for the treatment of axillary hyperhidrosis.
PATENT
https://patents.google.com/patent/US8558008B2/en
PATENT
https://patents.google.com/patent/US20130211101A1/en
-
Glycopyrrolate is a quaternary ammonium cation of the muscarinic anticholinergic group. Glycopyrrolate, typically as a bromide salt, has been used in the treatment of a variety of conditions including diarrhea (U.S. Pat. Nos. 6,214,792 and 5,919,760), urinary incontinence (U.S. Pat. Nos. 6,204,285 and 6,063,808), and anxiety (U.S. Pat. No. 5,525,347). Additionally, U.S. Pat. No. 5,976,499 discloses a method for diagnosing cystic fibrosis in a patient by, in part, stimulating sweat production through the injection of a glycopyrrolate solution into a patient. Glycopyrrolate has also been used for the treatment of hyperhidrosis in US 20100276329.
- [0002]
Glycopyrrolate has previously been made available as a bromide salt or an acetate salt. The bromide salt of glycopyrrolate is sold as Rubinol®. The term “glycopyrrolate” as used in the label for Rubinol® refers to the bromide salt which is more formally referred to as glycopyrronium bromide.
-
- Example 6 Glycopyrrolate Tosylate
- [0124]
In a dark room, silver tosylate (3.5 g) was dissolved in water (˜100 mL) by sonication. The solution was heated to approximately 40° C. and additional water was added (˜15 mL). An equimolar amount of glycopyrrolate bromide (5 g) (mixture of R,S and S,R diastereomers) was added and immediately resulted in a yellow precipitate. The slurry was stirred at approximately 40° C. overnight, and then slowly cooled while stirring to ambient temperature. At ambient temperature, the solids were vacuum filtered and the wet cake was washed three times with approximately 10 mL of water. The mother liquor was collected and filtered two times through a 0.2 μm nylon filter with glass microfiber (GMF). A clear solution was observed after filtration and was lyophilized at approximately −50° C. After 6 days, a mixture of white, needle-like and slightly sticky, glassy solids was observed. Toluene (˜20 mL) was added, and the slurry was briefly sonicated and then stirred at ambient temperature. Additional toluene (˜80 mL) was added for easier stirring, and the mixture was allowed to stand at ambient conditions for 1 day. Solids of glycopyrrolate tosylate were collected by vacuum filtration and vacuum drying at ambient temperature for 1 day.
- [0124]
Example 7 Preparation of Glycopyrrolate Tosylate
-
- [0125]
A slurry of equimolar amounts of glycopyrrolate acetate and p-toluenesulfonic acid was prepared in isopropanol (1 mL). The mixture was stirred at ambient temperature. Additional isopropanol (0.5 mL) was added to improve stirring, and the mixture was stirred overnight. Solids of glycopyrrolate tosylate were isolated by vacuum filtration and analyzed.
- [0125]
Example 8 Preparation of Glycopyrrolate Tosylate Form D
-
- [0126]
Glycopyrrolate tosylate (1.0569 g) made from Example 6 was dissolved in 4 mL ACN/H2O (50/50 vol/vol) by sonication. The solution was filtered through 0.2 μm nylon filter into a clean vial. The solvent was allowed to partially evaporate from an open vial under ambient conditions. Further evaporation was subsequently performed under nitrogen gas flow. A gel resulted which was vacuum dried at 40° C. for 1 day. Toluene (5 mL) was added and the mixture was sonicated for approximately 10 minutes causing white solids to precipitate. The mixture was stirred at ambient temperature for 1 day. The solids were isolated by vacuum filtration and the wet cake was washed with approximately 10 mL of toluene. The solids were vacuum dried at ambient temperature for 1 day. After vacuum drying the solids were placed in a vial which remained uncapped and placed inside a relative humidity chamber (˜97%). The chamber was placed inside an oven at 41° C. After 6 days, the solids were analyzed by XRPD showing Form D.
- [0126]
Example 9 Single Crystal Preparation of Form D
-
- [0127]
Glycopyrrolate tosylate (54.9 mg) made from Example 6 was dissolved in EtOAc/DMF (87/13 vol/vol) at approximately 55° C. at 24 mg/ml. The solution was hot filtered through a 0.2 μm nylon filter into a pre-warmed vial. The vial containing the solution was first placed in a dry ice/acetone bath and then in a freezer (approximately −25 to −10° C.). After 3 days, the solution was re-heated to approximately 50° C. and additional EtOAc was added for 96/4 EtOAc/DMF (vol/vol) at 7 mg/ml. The solution was quickly removed from elevated temperature and placed in the freezer. Solids were isolated by decanting the solvent and drying the solids under ambient conditions.
- [0128]
Single Crystal Data Collection
- [0129]
A colorless chunk of C26H37NO7S [C7H7O3S, C19H28NO3, H2O] having approximate dimensions of 0.23×0.20×0.18 mm, was mounted on a fiber in random orientation. Preliminary examination and data collection were performed with Cu Kα radiation (λ=1.54184 Å) on a Rigaku Rapid II diffractometer equipped with confocal optics. Refinements were performed using SHELX97.
- [0127]
Example 10 Preparation of Dehydrated Form D
-
- [0130]
A mixture of glycopyrrolate tosylate solids, including Form C and Form D, and a trace amount of silver tosylate was kept over P2O5 at ambient temperature for 18 days. The resulting solids were composed of a mixture of dehydrated Form D with a trace of silver tosylate as shown by XRPD analysis.
- [0130]
Example 11 Preparation of Form C Glycopyrrolate Tosylate
-
- [0131]
Glycopyrrolate tosylate Form D, containing trace amounts of Form C and silver tosylate, was heated on an Anton Paar TTK 450 stage and XRPD patterns were collected in situ in the range 3.5-26° (2θ). All heating steps were at approximately 10° C./min. The stage was heated in incremental steps of 20° C. from 25 to 125° C. At each step, an XRPD pattern was collected over approximately 4 minutes. The stage was then heated to 135° C. and an XRPD pattern was collected over approximately 16 minutes and after heating further to 145° C., a pattern was collected in approximately 31 minutes. The sample was subsequently cooled to 25° C. at approximately 24° C./min, upon which a final XRPD pattern was collected over approximately 16 min. The XRPD pattern of this final pattern was indexed as Form C.
- [0131]
Example 12 Preparation of Form C Glycopyrrolate Tosylate
-
- [0132]
Glycopyrrolate tosylate Form D from Example 6 was heated to an approximate temperature in the range 143-149° C. under a continuous nitrogen purge for approximately 3.3 hours. The vial containing the solids was capped, placed on a lab bench and allowed to cool down to room temperature. At room temperature, the vial was placed in a jar containing P2O5. The sample was prepared for XRPD analysis under nitrogen which confirmed production of Form C.
- [0132]
Example 13 Preparation of Form C Glycopyrrolate Tosylate
-
- [0133]
Glycopyrrolate tosylate (59.5 mg) from Example 6 was dissolved in acetone at approximately 50° C. at 27 mg/ml. The solution was hot filtered through a 0.2 μm nylon filter into a pre-warmed vial. The vial was capped and left on the hot plate which was subsequently turned off to allow the sample to cool slowly to ambient temperature. At ambient temperature the solution was stirred causing white solids to precipitate. The solids were isolated by vacuum filtration and the wet cake was washed with approximately 2 ml of acetone. XRPD analysis resulted in Form C.
- [0133]
Example 14 Amorphous Glycopyrrolate Tosylate
- [0134]
Glycopyrrolate tosylate from Example 6 was melted and cooled repeatedly until the majority of the solids had the appearance of a glass by microscopy. XRPD analysis indicated that the “glassy” sample was observed to be amorphous. A 2.2% weight loss was observed by TGA from 25 to 250° C. of the amorphous glycopyrrolate tosylate. The onset of the glass transition temperature was measured at 11.6° C.
In a dark room, silver tosylate (3.5 g) was dissolved in water (~ 100 mL) by sonication. The solution was heated to approximately 40°C. and additional water was added (-15 mL). An equimolar amount of glycopyrrolate bromide (5 g) (mixture of R,S and S,R diastereomers) was added and imme diately resulted in a yellow precipitate. The slurry was stirred at approximately 40°C. overnight, and then slowly cooled while stirring to ambient temperature. At ambient tempera ture, the solids were vacuum filtered and the wet cake was washed three times with approximately 10 mL of water. The mother liquor was collected and filtered two times through a 0.2 pm nylon filter with glass microfiber (GMF). A clear solution was observed after filtration and was lyophilized at approximately -50°C. After 6 days, a mixture of white, needle-like and slightly sticky, glassy solids was observed. Toluene (-20 mL) was added, and the slurry was briefly sonicated and then stirred at ambient temperature. Additional toluene (-80 mL) was added for easier stirring, and the mix ture was allowed to stand at ambient conditions for 1 day. Solids of glycopyrrolate tosylate were collected by vacuum filtration and vacuum drying at ambient temperature for 1 day. Glycopyrrolate Tosylate.
///////////Glycopyrrolate Tosylate, DRM-04 , DRM-04B , FDA 2018, Qbrexza
CC1=CC=C(C=C1)S(=O)(=O)[O-].C[N+]1(CCC(C1)OC(=O)C(C2CCCC2)(C3=CC=CC=C3)O)C
Diazoxide choline
Diazoxide choline,
RN: 1098065-76-9
UNII: 2U8NRZ7P8L
Diazoxide choline; UNII-2U8NRZ7P8L; 2U8NRZ7P8L; YLLWQNAEYILHLV-UHFFFAOYSA-N
| Molecular Formula: | C13H20ClN3O3S |
|---|---|
| Molecular Weight: | 333.831 g/mol |
Ethanaminium, 2-hydroxy-N,N,N-trimethyl-, compd. with 7-chloro-3-methyl-2H-1,2,4-benzothiadiazine dioxide (1:1)
7-chloro-3-methyl-1$l^{6},2,4-benzothiadiazin-2-ide 1,1-dioxide;2-hydroxyethyl(trimethyl)azanium
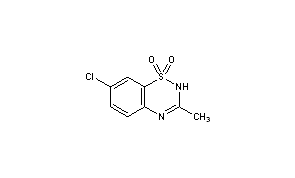 Diazoxide
Diazoxide
CAS: 364-98-7 FREE FORM
2H-1,2,4-Benzothiadiazine, 7-chloro-3-methyl-, 1,1-dioxide
- 4H-1,2,4-Benzothiadiazine, 7-chloro-3-methyl-, 1,1-dioxide (7CI)
- 3-Methyl-7-chloro-1,2,4-benzothiadiazine 1,1-dioxide
- 7-Chloro-3-methyl-2H-1,2,4-benzothiadiazine 1,1-dioxide
- Diazoxide
- Dizoxide
- Eudemine injection
- Hyperstat
- Hypertonalum
- Mutabase
- NSC 64198
- NSC 76130
- Proglicem
- Proglycem
- SRG 95213
- Sch 6783
Diazoxide (INN; brand name Proglycem[1]) is a potassium channel activator, which causes local relaxation in smooth muscle by increasing membrane permeability to potassium ions. This switches off voltage-gated calcium ion channels, preventing calcium flux across the sarcolemma and activation of the contractile apparatus.
In the United States, this agent is only available in the oral form and is typically given in hospital settings.[2]
Medical uses
Diazoxide is used as a vasodilator in the treatment of acute hypertension or malignant hypertension.[3]
Diazoxide also inhibits the secretion of insulin by opening ATP-sensitive potassium channel of beta cells of the pancreas, thus it is used to counter hypoglycemia in disease states such as insulinoma (a tumor producing insulin)[4] or congenital hyperinsulinism.
Diazoxide acts as a positive allosteric modulator of the AMPA and kainate receptors, suggesting potential application as a cognitive enhancer.[5]
Side effects
The Food and Drug Administration published a Safety Announcement in July 2015 highlighting the potential for development of pulmonary hypertension in newborns and infants treated with this drug.[2]Diazoxide interferes with insulin release through its action on potassium channels.[6] Diazoxide is one of the most potent openers of the K+ ATP channels present on the insulin producing beta cells of the pancreas. Opening these channels leads to hyperpolarization of cell membrane, a decrease in calcium influx, and a subsequently reduced release of insulin.[7] This mechanism of action is the mirror opposite of that of sulfonylureas, a class of medications used to increase insulin release in Type 2 Diabetics. Therefore, this medicine is not given to non-insulin dependent diabetic patients.
SYN
Medicinal Chemistry Research, 12(9), 457-470; 2004

PATENT
WO 2009006483
https://patents.google.com/patent/WO2009006483A1/enIt
PATENT
US 20120238554
PATENT
WO 2013130411
able 16. Characterization of Forms A and B of Diazoxide Choline Salt In
Screening Study
Experiment Form A Form B
*Maj or peaks (2-Θ):
Form A (9.8, 10.5, 14.9, 17.8, 17.9, 18.5, 19.5, 22.1, 22.6, 26.2, 29.6, 31.2);
Form B (8.9, 10.3, 12.0, 18.3, 20.6, 24.1, 24.5, 26.3, 27.1, 28.9).
** Unique FTIR (ATR) absorbances (cm 1):
Form A (2926, 2654, 1592, 1449, 1248);
Form B (3256, 2174, 2890, 1605, 1463, 1235).
6.1.5.1. Solubility Screen in organic solvents.
[00725] Diazoxide choline, prepared in MEK using choline hydroxide as 50 wt % solution in water (see above) displayed some solubility in the following solvents:
acetonitrile, acetone, ethanol, IPA, MEK, DMF, and methanol. These solvents were chosen due to differences in functionality, polarity, and boiling points and their ability to dissolve diazoxide. Other solvents which showed poor ability to dissolve salts were used as antisolvents and in slurry experiments where some solubility was observed: dioxane, MTBE, EtOAc, IP Ac, THF, water, cyclohexane, heptane, CH2C12, and toluene.
[00726] Solvents for crystallizations during screening were chosen based on the solubility screen summarized in Table 17. Crystallizations of diazoxide choline from all conditions afforded a total of two forms, A and B. Forms A and B were found to be anhydrous polymorphs of diazoxide choline. Form B was observed to be generated from most solvents used. It was difficult to isolate pure Form A on large scales (>50 mg) as conditions observed to produce Form A on a smaller scale (approximately 50 mg or less) were found to result in Form B or mixtures of both forms on larger scales. Based on room-temperature slurry experiments, anhydrous Form B was found to be the most thermodynamically stable form in this study. Form A readily converted to Form B in all slurry solvents utilized.
Table 17. Solubility Screen for Diazoxide Choline Salt
Solvent Cmpd Solvent Cone. Temp. Soluble
(mg) (mL) (mg/niL) (°C)
CH2CI2 1.3 5.00 0.26 55 Partially
Toluene 1.4 5.00 0.28 55 No
.1.5.2. Single-Solvent Crystallizations
[00727] Fast cooling procedure: Diazoxide (approximately 20 mg) was weighed out into vials and enough solvent (starting with 0.25 mL) was added until the material completely dissolved at elevated temperature. After hot filtration the vials were placed in a refrigerator (4 °C) for 16 hours. After the cooling-process the samples were observed for precipitates which were isolated by filtration. Vials not demonstrating precipitates were evaporated down to dryness using a gentle stream of nitrogen. All solids were dried in vacuo at ambient temperature and 30 in. Hg.
[00728] Slow cooling procedure: Diazoxide (approximately 30 mg of choline salt) was weighed out into vials and enough solvent was added until the material went into solution at elevated temperature. After hot filtration the vials were then slowly cooled to room temperature at the rate of 20 °C/h and stirred at room temperature for 1-2 hours. All solids were dried in vacuo at ambient temperature and 30 in. Hg.
[00729] Based on the initial solubility study, seven solvents were selected for the fast-cooling crystallization: acetonitrile, acetone, ethanol, IPA, MEK, DMF, and methanol. Table 18 shows a list of the solvents that were used and the amount of solvent needed to dissolve the material. After the cooling-process precipitates were noticed in samples # 2, 3, 5, and 6, the solids were isolated by filtration. The other samples (# 1, 4, and 7) were evaporated down to dryness using a gentle stream of nitrogen. The diazoxide choline salts were found to be consistent with Form A by XRPD analysis for all solids with the exception of sample #2 (consistent with the freeform) and sample #5 (consistent with Form B with preferred orientation observed).
Table 18. Single- Solvent Crystallization of Diazoxide Choline Salt Using Fast- Cooling Procedure
[00730] In accordance with the data obtained from fast-cooling experiments, four solvents which showed precipitation of solids were chosen for the slow-cooling experiments: MeOH, EtOH, MeCN, and IPA (Table 19). All obtained analyzable solids of the choline salt were found to be consistent with Form B by XRPD with the exception of Entry #1 which was consistent with diazoxide freeform and Entry #2 which was not analyzable. Mother liquor of Entry #2 was concentrated to dryness and the residual solids were analyzed by XRPD and found to be Form B material. As a result of obtaining freeform material from the single- solvent crystallizations in methanol, three more alcohols were tested for the single- solvent crystallizations using fast- and slow-cooling procedures. Tables 20 and 21 provide a list of the solvents that were used and the amount of solvent needed to dissolve the material. XRPD patterns of the fast-cooling procedure showed freeform of diazoxide from isobutanol, Form B from isoamyl alcohol, and Form A from tert-amyl alcohol compared to the slow-cooling procedure, which afforded Form B material from all three solvents.
Table 19. Single-Solvent Crystallization of Diazoxide Choline Salt Using Slow- Cooling Procedure
Table 20. Single- Solvent Crystallization of Diazoxide Choline Salt Using Fast- Cooling Procedure
Table 21. Single-Solvent Crystallization of Diazoxide Choline Salt Using Slow- Cooling Procedure
[00731] The results of the choline salt single- solvent fast- and slow-cooling crystallizations (see Tables 19 to 21) indicated that Form A was more likely to be isolated with fast-cooling profiles and Form B with slow-cooling profiles.
6.1.5.3. Binary Solvent Crystallizations
[00732] Binary- solvent crystallizations of the choline salt were performed using four primary solvents (MeOH, EtOH, IPA, and MeCN) and nine cosolvents (MTBE, EtOAc, IPAc, THF, c-hexane, heptane, toluene, CH2CI2, and dioxane) with a fast-cooling profile (supra). XRPD patterns showed that Form B was obtained from mixtures of MeOH with MTBE, EtOAc, IPAc, toluene, and dioxane. As shown in Table 22, Form A was obtained from mixtures of MeOH with THF and with CH2CI2 after evaporating the solvent to dryness. The mixtures of MeOH with cyclohexane and heptane provided the freeform of diazoxide. All solids obtained from fast-cooling procedures with EtOH, IPA, and MeCN as primary solvents provided Form B material.
Table 22. Binary-Solvent Crystallizations of Choline Salt of Diazoxide Using Fast- Cooling Procedure and MeOH as a Primary Solvent
* Solids were dissolved at 62 °C.
** Freeform of diazoxide.
[00733] Binary- solvent recrystallizations of the choline salt with the slow-cooling procedure were performed using two primary solvents (IPA and MeCN) and nine cosolvents (MTBE, EtOAc, IPAc, THF, c-hexane, heptane, toluene, CH2C12, and dioxane). All solids obtained from a slow-cooling procedure with IPA and MeCN as primary solvents provided Form B material based on XRPD analysis. The results of
binary- solvent crystallizations indicated that Form B was the most thermodynamic ally stable form of diazoxide choline.
6.1.5.4. Binary Solvent Crystallizations Using Water as a Cosolvent
[00734] In an attempt to investigate the formation of hydrates of the choline salt, experiments was performed using fast- and slow-cooling procedures and water as a cosolvent.
[00735] The fast cooling procedure (supra) was used with the exception of using different primary solvents which were miscible with water: acetone, acetonitrile, DMF, IPA, i-BuOH, i-AmOH, and t-AmOH. Water was utilized in these crystallizations as a cosolvent. All solids obtained from the fast-cooling procedure with water as the cosolvent provided diazoxide freeform material by XRPD analysis.
[00736] To compare the results obtained from the fast-cooling procedure a set of experiments was performed using a slow-cooling procedure and water as a cosolvent. All obtained solids were analyzed by XRPD and afforded patterns consistent with diazoxide freeform. Without wishing to be bound by theory, these results suggest that the conditions used for crystallization caused dissociation of the choline salt. A small amount of a second crop was obtained in each sample, but only two samples were analyzable by XRPD and indicated that the samples were freeform material. All mother liquors were evaporated to dryness and the residual solids were also analyzed by XRPD to afford patterns consistent with Form B of the choline salt.
6.1.5.5. Metastable Zone Width Estimation
[00737] Form B: To produce a robust process, an understanding of the solubility profiles of the various solid forms under consideration is required. From a practical standpoint, this involves the measurement of the metastable zone width (MSZW) of pure forms, whereby the saturation and supersaturation curves of the different forms are generated over a well defined concentration and temperature range. This knowledge can then be used to design a crystallization protocol that should ideally favor a selective crystal growth of the desired form.
[00738] Form B of diazoxide choline salt showed moderate solubility in a solvent mixture made of MeCN/MeOH/MtBE (10: 1: 12, volume ratios). The wide width of the metastable zone as shown in Table 23 gives many seeding options. During the MSZW measurement, aliquots from the crystallizing material were withdrawn and analyzed by XRPD to ensure that no form conversion occurred during the experiment. Indeed, the material remained unchanged during the test.
Table 23. Meta-Stable Zone Width For Form B Diazoxide Choline Salt in
MeCN/MeOH/MtBE (10:1:12) (v/v).
[00739] Form A: The metastable zone width for Form could not be estimated because this polymorphic form converted during the experiment to Form B.
6.1.5.6. Crystallization of Form A of Diazoxide Choline Salt
[00740] The choline salt of diazoxide (160.3 mg) was dissolved in 1 mL of IPA at 55 °C which was then passed through a Millipore 0.45 μΜ filter into a clean vial. This vial was placed in freezer a -20 °C overnight. Solids were not noticed and the flask was scratched with a micro- spatula. The vial was placed back in the freezer and nucleation was noticed after ten minutes. The solids were collected by vacuum filtration and washed with 1 mL of MtBE. The solids were dried in vacuo at 40 °C and 30 in. Hg to afford 70 mg (43.6% recovery) of Form A as determined by XRPD.
6.1.5.7. 500-mg Scale Crystallization of Form B of Diazoxide Choline Salt
[00741] The choline salt of diazoxide (524.3 mg) was dissolved in 3 mL of IPA at 78 °C and this solution was then cooled to 55 °C for the addition of MtBE. The MtBE (4 mL) was added until nucleation was observed. After nucleation the batch was allowed to cool to room temperature at a rate of 20 °C /h. The solids were collected by vacuum filtration and washed with 1 mL of MtBE. The solids were dried in vacuo at 40 °C and 30 in. of Hg to afford 426.7 mg (81.3% recovery) of Form B as determined by XRPD.
6.1.5.8. 2-g Scale Crystallization of Form B of Diazoxide Choline Salt
[00742] The choline salt of diazoxide (2.0015 g) was dissolved in 5.5 mL of IPA at 78 °C to afford a clear solution. This solution was passed through a Millipore Millex FH 0.45 μΜ filter. This solution was then cooled to 55 °C. MtBE was added in 1 mL portions, with a two minute interval between portions. Nucleation was noted after the second addition of MtBE. This suspension was allowed to cool to room temperature at a rate of 20 °C /h and stirred at this temperature for 16 hours. The solids were collected by vacuum filtration and washed with 1 mL of MtBE. The solids were dried in vacuo at 40 °C and 30 in. of Hg to afford 1.6091 g (80.4% recovery) of Form B as determined by XRPD.
6.1.5.9. Detection of Form Impurities
[00743] Mixtures of diazoxide choline Forms A and B were prepared by adding a minor amount of Form A to Form B. Samples were lightly ground by hands with a mortar and pestle for approximately one minute. Samples were then analyzed by XRPD analysis. XRPD analysis was found to be suitable for detecting 5% of Form A in Form B.
References
- Jump up^ Diazoxide, drugs.com
- ^ Jump up to:a b “FDA Drug Safety Communication: FDA warns about a serious lung condition in infants and newborns treated with Proglycem (diazoxide)” (Press release). Food and Drug Administration. July 16, 2015. Retrieved 2015-07-19.
- Jump up^ van Hamersvelt HW, Kloke HJ, de Jong DJ, Koene RA, Huysmans FT (August 1996). “Oedema formation with the vasodilators nifedipine and diazoxide: direct local effect or sodium retention?”. Journal of Hypertension. 14 (8): 1041–5. doi:10.1097/00004872-199608000-00016. PMID 8884561.

- Jump up^ Huang Q, Bu S, Yu Y, et al. (January 2007). “Diazoxide prevents diabetes through inhibiting pancreatic beta-cells from apoptosis via Bcl-2/Bax rate and p38-beta mitogen-activated protein kinase”. Endocrinology. 148 (1): 81–91. doi:10.1210/en.2006-0738. PMID 17053028.

- Jump up^ Randle, John C.R.; Biton, Catherine; Lepagnol, Jean M. (15 November 1993). “Allosteric potentiation by diazoxide of AMPA receptor currents and synaptic potentials”. European Journal of Pharmacology. 247 (3): 257–65. doi:10.1016/0922-4106(93)90193-D. PMID 8307099.

- Jump up^ Panten, Uwe; Burgfeld, Johanna; Goerke, Frank; Rennicke, Michael; Schwanstecher, Mathias; Wallasch, Andreas; Zünkler, Bernd J.; Lenzen, Sigurd (1989-04-15). “Control of insulin secretion by sulfonylureas, meglitinide and diazoxide in relation to their binding to the sulfonylurea receptor in pancreatic islets”. Biochemical Pharmacology. 38 (8): 1217–1229. doi:10.1016/0006-2952(89)90327-4.
- Jump up^ Doyle, Máire E.; Egan, Josephine M. (2003-03-01). “Pharmacological Agents That Directly Modulate Insulin Secretion”. Pharmacological Reviews. 55 (1): 105–131. doi:10.1124/pr.55.1.7. ISSN 1521-0081. PMID 12615955.
 |
|
| Clinical data | |
|---|---|
| Trade names | Proglycem |
| AHFS/Drugs.com | Monograph |
| Pregnancy category |
|
| Routes of administration |
Oral, intravenous |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 90% |
| Metabolism | Hepatic oxidation and sulfate conjugation |
| Elimination half-life | 21-45 hours |
| Excretion | Renal |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| ECHA InfoCard | 100.006.063 |
| Chemical and physical data | |
| Formula | C8H7ClN2O2S |
| Molar mass | 230.672 g/mol |
| 3D model (JSmol) | |
//////////////Diazoxide choline
CC1=NC2=C(C=C(C=C2)Cl)S(=O)(=O)[N-]1.C[N+](C)(C)CCO
CC1=NC2=C(C=C(C=C2)Cl)S(=O)(=O)[N-]1.C[N+](C)(C)CCO
Arterolane Maleate

Arteolane Maleate
C26H40N2O8
Molecular Weight: 508.612
CAS 959520-73-1
664338-39-0 (free base) 959520-73-1 (maleate) 959520-79-7 (acetate) 664338-40-3 (tosylate) 959520-82-2 (tartrate) 959520-83-3 (citrate)
N-(2-amino-2-methylpropyl)-2-((1R,3R,4”S,5R,5’s,7R)-dispiro[adamantane-2,3′-[1,2,4]trioxolane-5′,1”-cyclohexan]-4”-yl)acetamide maleate
Dispiro[cyclohexane-1,3′-[1,2,4]trioxolane-5′,2”-tricyclo[3.3.1.13,7]decane]-4-acetamide, N-(2-amino-2-methylpropyl)-, cis-, (2Z)-2-butenedioate (1:1)
APPROVED 4.11.2017 CDSCO
Arteolane Maleate and Piperaquine phosphate Dispersible tablets (37.5 mg +187.5 mg

cas 664338-39-0
Arterolane
Arterolane, also known as OZ277 or RBx 11160, is a substance that was tested for antimalarial activity[1] by Ranbaxy Laboratories.[2] It was discovered by US and European scientists who were coordinated by the Medicines for Malaria Venture (MMV).[3] Its molecular structure is uncommon for pharmacological compounds in that it has both a ozonide (trioxolane) group and an adamantanesubstituent.[4]
Initial results were disappointing, and in 2007 MMV withdrew support, after having invested $20M in the research;[5] Ranbaxy said at the time that it intended to continue developing the drug combination on its own.[2] Ranbaxy started a Phase II clinical trial of arterolane, in combination with piperaquine in 2009 that published in 2015.[6][7]
In 2012, Ranbaxy obtained approval to market the arterolane/piperaquine combination drug in India, under the brand name Synriam,[5]and in 2014 received approval to market it in Nigeria, Uganda, Senegal, Cameroon, Guinea, Kenya and Ivory Coast; it had already received approval in Uganda.[8]

Ranbaxy launched India’s first new drug, SynriamTM, treating Plasmodium falciparummalaria in adults. The drug provides quick relief from most malaria-related symptoms, including fever, and has a high cure rate of over 95 %.
Just one tablet per day is required, for three days, instead of two to four tablets, twice daily, for three or more days with other medicines. The drug is independent of dietary restrictions for fatty foods or milk.
Ranbaxy developed Synriam as a fixed-dose combination of arterolane maleate and piperaquine phosphate, where arterolane is the new chemical entity (NCE) that was developed as an alternative to artemisinin. It is the first recently developed antimalarial not based on artemisinin, one of the most effective treatments for malaria, which has shown problems with resistance in recent years. Arterolane was discovered by a collaborative drug discovery project funded by the Medicines for Malaria Venture. Since SynriamTM has a synthetic source, unlike artemisinin-based drugs, production can be scaled up whenever required and a consistent supply can be maintained at a low cost.

The new drug, has been approved by the Drug Controller General of India (DCGI) for marketing in India and conforms to the recommendations of the World Health Organization (WHO) for using combination therapy in malaria. Ranbaxy is also working to make it available in African, Asian and South American markets where Malaria is rampant. SynriamTM trials are ongoing for Plasmodium vivax malaria and a paediatric formulation.
Derek Lowe of the famous In the Pipeline blog had written about arterolane in 2009. At the time it was in Phase III trial, which I assumed were the trials that Ranbaxy was conducting. But it turned out that arterolane was developed by a collaboration between researchers in the US, the UK, Switzerland and Australia who were funded by the World Health Organization and Medicines for Malaria Venture (a Swiss non-profit). They published this work in Nature in 2004 and further SAR (Structure Activity Relationship) studies in J Med Chem in 2010. So Ranbaxy did not develop the drug from scratch? But the press release quotes Arun Sawhney, CEO and Managing Director of Ranbaxy which misleads people to think so: “It is indeed gratifying to see that Ranbaxy’s scientists have been able to gift our great nation its first new drug, to treat malaria, a disease endemic to our part of the world. This is a historic day for science and technology in India as well as for the pharmaceutical industry in the country. Today, India joins the elite and exclusive club of nations of the world that have demonstrated the capability of developing a new drug”. So Ranbaxy mixes a known active compound (piperaquine) with a new compound that someone else found to be active (arterolane) and claims that they developed a new drug? In an interview in LiveMint, Sawhney says, “Ranbaxy spent around $30 million on Synriam and the contribution from DST [India’s Department of Science & Technology] was Rs.5 crore. The drug went through several phases of development since the project began in 2003. We did not look at this as a commercial development. Instead, this is a CSR [Corporate Social Responsibility] venture for us.” That’s a give away because developing a new drug from scratch has to cost more than $30 million + Rs.50 million.
- Ranbaxy Laboratories Limited (Ranbaxy), India
 now taken over by sun
now taken over by sun
SynriamTM
|
|
Description SynriamTM is a fixed dose combination of two antimalarial active ingredients arterolane maleate and piperaquine phosphate.
Arterolane maleate is a synthetic trioxolane compound. The chemical name of arterolane maleate is cis-adamantane-2-spiro-3’-8’-[[[(2’-amino-2’ methylpropyl) amino] carbonyl] methyl] 1’,2’,4’-trioxaspiro [4.5] decane hydrogen maleate. The molecular formula is C26H40N2O8 and molecular weight is 508.61. The structural formula is as follows:

| MALARIA |
| Malaria is one of the most prevalent and deadly parasitic diseases in the world. Up to 289 million cases of malaria may have occurred in 2010, causing between 660,000 and 1.25 million deaths, mainly in Africa and mostly of children younger than 5 years. |
| (WHO: http://www.who.int/malaria/publications/world_malaria_report_2012/en/index.html; Fidock, D. A. Eliminating Malaria. Science 2013, 340, 1531-1533.) |
|

Malaria, the most common parasitic disease of humans, remains a major health and economic burden in most tropical countries. Large areas of Central and South America, Hispaniola (Haiti and the Dominican Republic), Africa, the Middle East, the Indian subcontinent, Southeast Asia, and Oceania are considered as malaria-risk areas. It leads to a heavy toll of illness and death, especially amongst children and pregnant women.
According to the World Health Organization, it is estimated that the disease infects about 400 million people each year, and around two to three million people die from malaria every year. There are four kinds of malaria parasites that infect human: Plasmodium falciparum, Plasmodium vivax, Plasmodium ovale and Plasmodium malariae.
Malaria spreads from one person to another by the bite of mosquito, Anopheles gambiae, which serves as vector. When a mosquito sucks the blood of human, sporozoites are transfused into the human body together with saliva of the mosquito. The sporozoites enter into the hepatocytes, reproduce asexually and finally enter into the blood stream. The parasites continue to multiply inside the red blood cells, until they burst and release large number of merozoites. This process continues, destroying a significant number of blood cells and causing the characteristic paroxysm (“chills and fever”) associated with the disease. In the red blood cells, some of the merozoites become male or female gametocytes. These gametocytes are ingested by the mosquito when it feeds on blood. The gametocytes fuse in the vector’s gut; sporozoites are produced and are migrated to the vector’s salivary glands.
The clinical symptoms of malaria are generally associated with the bursting of red blood cells causing an intense fever associated with chills that can leave the infected individual exhausted and bedridden. More severe symptoms associated with repeat infections and/or infection by Plasmodium falciparum include anaemia, severe headaches, convulsions, delirium and, in some instances, death.
Quinine, an antimalarial compound that is extracted from the bark of cinchona tree, is one of the oldest and most effective drugs in existence. Chloroquine and mefloquine are the synthetic analogs of quinine developed in 1940’s, which due to their effectiveness, ease of manufacture, and general lack of side effects, became the drugs of choice. The downside to quinine and its derivatives is that they are short-acting and have bitter taste. Further, they fail to prevent disease relapses and are also associated with side effects commonly known as “Chinchonism syndrome” characterized by nausea, vomiting, dizziness, vertigo and deafness. However, in recent years, with the emergence of drug- resistant strains of parasite and insecticide-resistant strains of vector, the treatment and/or control of malaria is becoming difficult with these conventional drugs.
Malarial treatment further progressed with the discovery of Artemisinin
(qinghaosu), a naturally occurring endoperoxide sesquiterpene lactone isolated from the plant Artemisia annua (Meshnick et al., Microbiol. Rev. 1996, 60, p. 301-315; Vroman et al., Curr. Pharm. Design, 1999, 5, p. 101-138; Dhingra et al., 2000, 66, p. 279-300), and a number of its precursors, metabolites and semi-synthetic derivatives which have shown to possess antimalarial properties. The antimalarial action of artemisinin is due to its reaction with iron in free heme molecules of the malaria parasite, with the generation of free radicals leading to cellular destruction. This initiated a substantial effort to elucidate its molecular mechanism of action (Jefford, dv. Drug Res. 1997, 29, p. 271-325; Cumming et al., Adv. Pharmacol. 1997, 37, p. 254-297) and to identify novel antimalarial peroxides (Dong and Vennerstrom, Expert Opin. Ther. Patents 2001, 1 1, p. 1753-1760).
Although the clinically useful artemisinin derivatives are rapid acting and potent antimalarial drugs, they have several disadvantages including recrudescence,
neurotoxicity, (Wesche et al., Antimicrob. Agents. Chemother. 1994, 38, p. 1813-1819) and metabolic instability (White, Trans. R. Soc. Trop. Med. Hyg., 1994, 88, p. 41-43). A fair number of these compounds are quite active in vitro, but most suffer from low oral activity (White, Trans. R. Soc. Trop. Med. Hyg., 1994, 88, p. 41-43 and van Agtmael et al., Trends Pharmacol. Sci., 1999, 20, p. 199-205). Further all these artemisinin derivatives are conventionally obtained from plant source and are therefore expensive. As the cultivation of the plant material is dependent on many factors including the weather conditions, the supply source thus becomes finite and there are chances of varying yield and potency. This leads to quality inconsistencies and supply constraints. As malaria is more prevalent in developing countries, a switch to cheaper and effective medicine is highly desirable.
Thus there exists a need in the art to identify new peroxide antimalarial agents, especially those which are not dependent on plant source and can be easily synthesized, are devoid of neurotoxicity, and which possess improved solubility, stability and pharmacokinetic properties.
Following that, many synthetic antimalarial 1 ,2,4-trioxanes (Jefford, Adv. Drug Res. 1997, 29, p. 271-325; Cumming et al., Adv. Pharmacol. 1997, 37, p. 254-297), 1,2,4,5-tetraoxanes (Vennerstrom et al., J. Med. Chem., 2000, 43, p. 2753-2758), and other endoperoxides have been prepared. Various patents/applications disclose means and method for treating malaria using Spiro or dispiro 1,2,4-trioxolanes for example, U.S.
Patent Application No. 2004/0186168 and U.S. Patent Nos. 6,486, 199 and 6,825,230. The present invention relates to solid dosage forms of the various spiro or dispiro 1 ,2,4- trioxolanes antimalarial compounds disclosed in these patents/applications and are incorporated herein by reference.
Active compounds representing various Spiro and dispiro 1 ,2,4-trioxolane derivatives possess excellent potency, efficacy against Plasmodium parasites, and a lower degree of neurotoxicity, in addition to their structural simplicity and ease of synthesis. Furthermore, these compounds have half-lives which are believed to permit short-term treatment regimens comparing favorably to other artemisinin-like drugs. In general, the therapeutic dose of trioxolane derivative may range between about 0.1-1000 mg/kg/day, in particular between about 1-100 mg/kg/day. The foregoing dose may be administered as a single dose or may be divided into multiple doses. For malaria prevention, a typical dosing schedule could be, for example, 2.0-1000 mg/kg weekly beginning 1-2 weeks prior to malaria exposure, continued up to 1-2 weeks post-exposure.
Monotherapy with artemisinin (natural or synthetic) class of drugs might cure the patients within 3 days, however perceiving the potential threat of the malarial parasite developing resistance towards otherwise very potent artemisinin class of drugs, WHO had strictly called for an immediate halt to the provision of single-drug artemisinin malaria pills. Combination therapy in case of malaria retards the development of resistance, improve efficacy by lowering recrudescence rate, provides synergistic effect, and increase exposure of the parasite to the drugs.
Artemsinin based combinations are available in the market for a long time.
Artemether-lumafentrine (Co-artem®) was the first fixed dose antimalarial combination containing an artemisinin derivative and has been known since 1999. This combination has passed extensive safety and efficacy trials and has been approved by more than 70 regulatory agencies. Co-artem® is recommended by WHO as the first line treatment for uncomplicated malaria.
Other artemisinin based combinations include artesunate and amodiaquine (Coarsucam®), and dihydroartemisin and piperaquine (Eurartesim®). Unfortunately, all the available artemisinin based combinations have complicated dosage regimens making it difficult and inconvenient for a patient to comply completely with the total prescribed duration. For example, the dosage regimen of Co-artem® for an adult having body weight of more than 35 kg includes 6 doses over three days. The first dose comprises four tablets initially, the second dose comprises four tablets after eight hours, the third to sixth doses comprise four tablets twice for another two days; making it a total of 24 tablets. The dosage regimen of Coarsucam® for an adult having body weight of more than 36 kg or age above 14 years includes three doses over three days; each dose comprises two tablets; making it a total of six tablets. The dosage regimen of Eurartesim® for an adult having body weight between 36 kg – 75 kg includes 3 doses over three days, each dose comprises of three tablets, making it a total of nine tablets.
It is evident that the available artemisinin-based combinations have a high pill burden on patients as they need to consume too many tablets. As noted above, this may increase the possibility of missing a few doses, and, consequently, could result in reduced efficacy due to non-compliance and may even lead to development of resistance for the drug. Therefore, there is an urgent and unmet need for anti-malarial combinations with a simplified daily dosing regimen that reduces the pill burden and would increase patient compliance.
Apart from simplifying the regimen, there are certain limitations for formulators developing formulations with trioxolones, the first being their susceptibility to degradation in presence of moisture that results in reduced shelf lives. Another is their bitter taste, which can result in poor compliance of the regimen or selection of another, possibly less effective, therapeutic agent.
……………………..
http://www.google.st/patents/US6906205

……………………
http://www.google.st/patents/WO2013008218A1?cl=en
structural Formula II.
Formula II
Active compound includes one or more of the various spiro and dispiro trioxolane derivatives disclosed in U.S. Application No. 2004/0186168 and U.S. Patent Nos.
6,486,199 and 6,825,230, which are incorporated herein by reference. These trioxolanes are relatively sterically hindered on at least one side of the trioxolane heterocycle which provides better in vivo activity, especially with respect to oral administration. Particularly, spiro and dispiro 1,2,4-trioxolanes derivatives possess excellent potency and efficacy against Plasmodium parasites, and a lower degree of neurotoxicity.
The term “Active compound I” herein means cis-adamantane-2-spiro-3′-8′-[[[(2′- amino-2′-methylpropyl)amino]carbonyl]-methyl]- 1 ‘,2′,4’-trioxaspiro[4.5]decane hydrogen maleate. The Active compound I may be present in an amount of from about 5% to about 25%, w/w based on the total dosage form.

………………
http://www.google.st/patents/WO2007138435A2?cl=en
A synthetic procedure for preparing compounds of Formula I, salts of the free base c«-adamantane-2-spiro-3′-8′-[[[(2′-amino-2′-methyl propyl) amino] carbonyl] methyl]- 1 ‘, 2′, 4’-trioxaspiro [4.5] decane has been disclosed in U.S. 6,906,205.

The process for the preparation of compounds of Formula I wherein a compound of Formula II (wherein R is lower alkyl) is reacted with a compound of Formula III (wherein R is lower alkyl) to obtain compound of Formula IV;
Formula Formula IV
followed by hydrolysis of the compounds of Formula IV to give a compound of Formula V;
Formula V followed by the reaction of the compound of Formula V with an activating agent, for example, methyl chloroformate, ethyl chloroformate, propyl chloro formate, n-butyl chloro formate, isobutyl chloroformate or pivaloyl chloride leads to the formation of mixed anhydride, which is reacted in situ reaction with 1 ,2-diamino-2-methyl propane to give a compound of Formula VI; and
Formula Vl reacting the compound of Formula VI with an acid of Formula HX (wherein X can be the same as defined earlier) to give compounds of Formula I.
Example 1 : Preparation of O-methyl-2-adamantanone oxime
To a solution of 2-adamantanone (50 g, 0.3328 mol, 1 equiv.) in methanol (0.25 lit), sodium hydroxide solution (15 g, 0.3761mol, 1.13 equiv, in 50 mL water) was added followed by methoxylamine hydrochloride (37.5 g x 81.59% Purity= 30.596 g, 0.366 mol, 1.1 equiv) at room temperature under stirring. The reaction mixture was stirred at room temperature for 1 to 2 h. The reaction was monitored by HPLC. The reaction mixture was concentrated at 40- 45°C under vacuum to get a thick residue. Water (250 mL) was added at room temperature and the reaction mixture was stirred for half an hour. The white solid was filtered, washed with water (50 mL), and dried at 40 to 45°C under reduced pressure. O-methyl 2- adamantanone oxime (57 g, 95 % yield) was obtained as a white solid.
(M++l) 180, 1HNMR (400 MHz, CDCl3 ): δ 1.98 – 1.79 (m, 12H), 2.53 (s, IH), 3.46 ( s, IH), 3.81 (s, 3H).
Example 2: Preparation of 4-(methoxycarbonvmethvPcvclohexanone A high pressure autoclave was charged with a mixture of methyl (4- hydroxyphenyl)acetate (50 g, 0.30 mol), palladium ( 5g) (10 %) on carbon (50 % wet) and O- xylene (250 mL). The reaction mixture was stirred under 110 to 115 psi of hydrogen pressure for 7 to 8 h at 1400C. The reaction was monitored by HPLC. The reaction mixture was then cooled to room temperature, and the catalyst was filtered off. Filtrate was concentrated under reduced pressure to get 4-(methoxycarbonylmethyl)cyclohexanone as light yellow to colorless oily liquid (48.7 g, 97.4 %).
(M++!) 171, ‘ HNMR (400 MHz, CDCl 3): δ 1.48 – 1.51 ( m, 2H), 2.1 1-2.07 (m, 2H), 2.4- 2.23 (m, 7H), 3.7 (s, 3H).
Example 3: Preparation of methyl (Is, 4s)-dispiro [cyclohexane-l, 3′-f 1,2,4] trioxolane-5′, 2″-tricvclor3.3.1.13–71decan1-4-ylacetate
A solution of O-methyl-2-adamantanone oxime (example 1) (11.06 g, 61.7 mmol, 1.5 equiv.) and 4-(methoxycarbonymethyl)cyclohexanone (example 2) (7.0 g, 41.1 mmol, 1 equiv.) in cyclohexane ( 200ml) and dichloromethane (40 mL) was treated with ozone (ozone was produced with an OREC ozone generator [0.6 L/min. O2, 60 V] passed through an empty gas washing bottle that was cooled to -780C). The solvent was removed after the reaction was complete. After removal of solvents, the crude product was purified by crystallization from 80% aqueous ethanol (200 mL) to afford the title compound as a colorless solid. Yield: 10.83 g, 78%, mp: 96-980C; 1HNMR (500 Hz3CDCl3): δ 1.20-1.33 (m, 2H), 1.61-2.09 (m, 5 21H), 2.22 (d, J = 6.8Hz, 2H), 3.67(s,3H).
Example 4: Preparation of (Is, 4s)-dispiro [cyclohexane-1, 3′-[l,2,4] trioxolane-5′, 2″- tricvclo [3.3.1.13‘7] decanl-4-ylacetic acid
Sodium hydroxide (3.86 g, 96.57 mmol, 3 equiv.) in water (80 mL) was added to a solution of methyl (\s, 4s)-dispiro [cyclohexane-1, 3′-[l,2,4] trioxolane-5′, 2″-tricyclo
10 [3.3.1.I3‘7] decan]-4-ylacetate (example 3) (10.83 g, 32.19 mmol, 1 equiv.) in 95% ethanol (150 mL). The mixture was stirred at 500C for about 4 h, cooled to O0C, and treated with IM hydrochloric acid (129ml, 4 equiv). The precipitate was collected by filtration, washed with 50 % aqueous ethanol (150 mL) and dried in vacuum at 40 0C to give the title compound as colorless solid. Yield: 9.952 g, 96%, mp: 146-1480C ( 95% ethanol), 1HNMR (500 Hz,
15 CDCl3): δ 1.19-1.41 (m,2H), 1.60-2.05 (m,21H), 2.27 (d, J=6.8 Hz,2H).
Example 5: Preparation of c?s-adamantane-2-spiro-3′-8′-[[[(2′-amino-2′-methyl propyl) amino] carbonyl] methyl]-! ‘, T , 4’-trioxaspiro [4.5] decane
Method A:
(Is, 4s)-dispiro[cyclohexane- 1 ,3 ‘-[ 1 ,2,4]trioxolane-5 ‘,2 ‘ ‘-tricyclo[3.3.1.13‘7]decan]-4-
.0 ylacetic acid (example 4) (5 g ,15.5mmol, 1 equiv) was mixed with triethylamine (2.5 g , 24.8 mmol, 1.6 equiv) in 100ml of dichloromethane. The reaction mixture was cooled to – 1O0C to 00C. Ethyl chloro formate (1.68 g, 17 mmol, 1.0 equiv) in 15 mL dichloromethane was charged to the above reaction mixture at – 100C to 00C. The reaction mixture was stirred at the same temperature for 10 to 30 minutes. The resulting mixed anhydride reaction mixture
15 was added dropwise to a previously prepared solution of l,2-diamino-2-methylpropane (1.64 g, 18.6 mmol, 1.2 equiv), in 100 mL dichloromethane at -100C to O0C. The temperature of reaction mixture was raised to room temperature. The reaction mixture was stirred at the same temperature till the reaction was complete. Reaction monitoring was done by thin layer chromatography using 5 to 10% methanol in dichloromethane. The reaction was complete
>0 within 2 h. Nitrogen atmosphere was maintained throughout the reaction. Water (50 mL) was charged, organic layer was separated and washed with 10% sodium bicarbonate solution (50 mL) and water (50 mL) at room temperature. The organic layer was dried over sodium sulphate and the solvent was removed at 25 to 4O0C under reduced pressure. Hexane (50ml) was added to obtain residue under stirring at room temperature. The mixture was filtered and washed with 5 mL of chilled hexane. The solid was dried under reduced pressure at room 5 temperature.
Yield: 5.2 g (85.4 %), (M++l) 393, 1HNMR (400 MHz, DMSO-J6 ): δ 0.929 ( s, 6H), 1.105 – 1.079 (m, 2H), 1.887-1.641 (m, 21H), 2.030-2.017 (d, 2H), 2.928 (d, 2H).
Method B:
(Is, 4s)-dispiro [cyclohexane-1, 3′-[l,2,4] trioxolane-5′, 2″-tricyclo [3.3.1.I3‘7]
10 decan]-4-ylacetic acid (example 4) (10 g, 31mmol, 1 equiv) was treated with isobutyl chloroformate (4.5 g, 33mmol, 1.1 equiv) in presence of organic base like triethyl amine (5 g, 49.6mmol, 1.6 equiv) at 00C to 7°C in 250ml of dichloromethane. The solution was stirred at O0C to 7°C for aboutlO to 30 minutes. To the above reaction mixture, previously prepared solution of l,2-diamino-2-methylpropane (3.27 g, 37 mmol, 1.2 equiv), in 50 mL of
15 dichloromethane was added at O0C to 7°C in one lot. The temperature of reaction mixture was raised to room temperature. The reaction mixture was stirred at the room temperature till reaction was over. Reaction monitoring was done by thin layer chromatography using 5 to 10% methanol in dichloromethane. Reaction was complete within 2 h. The reaction nitrogen atmosphere was maintained throughout the reaction. Water (250 mL) was charged, organic
20 layer was separated and washed with 10% sodium bicarbonate solution (200 mL) and water (100 mL) at room temperature and the solvent was removed at 25 to 4O0C under reduced pressure. Hexane (100ml) was added to the residue, under stirring, at room temperature. The mixture was filtered and washed with chilled hexane (10 mL). The resultant solid was dried under reduced pressure at room temperature. Yield: 10.63 g (87%), (M++l) 393, 1HNMR
>5 (400 MHz, DMSO-J6 ) :δ 0.928 ( s, 6H), 1.102 – 1.074 (m, 2H), 1.859-1.616 (m, 21H), 2.031- 2.013 (d, 2H), 2.94-2.925 (d, 2H). Method C:
(\s, 4s)-dispiro[cyclohexane-l,3′-[l,2,4]trioxolane-5′,2″-tricyclo[3.3.1.13>7]decan]-4- ylacetic acid (example 4) (5 g, 15.5mmol, 1 equiv) was treated with pivaloyl chloride (1.87 g, 15.5 mmol, 1 equiv) and triethylamine (2.5gm, 24.8mmol, 1.6 equiv) at -15°C to -100C in dichloromethane (125 mL). The solution was stirred at -150C to -100C for aboutlO to 30 minutes. It resulted in the formation of mixed anydride. To the above reaction mixture, previously prepared solution of 1 ,2-diamino-2-methylpropane (1.64 g, 18.6 mmol, 1.2 equiv) in 25 mL dichloromethane was added at -15°C to -100C. The temperature of reaction mixture was raised to room temperature. The reaction mixture was stirred at the room temperature till reaction was over. Reaction monitoring was done by thin layer chromatography using 5 to 10% methanol in dichloromethane. The reaction was complete within 2 h. Nitrogen atmosphere was maintained throughout the reaction. Water (125 mL) was charged, organic layer was separated and washed with 50 mL of 10% sodium bicarbonate solution and 125 mL of water, respectively at room temperature. Finally solvent was removed at 25 to 4O0C under reduced pressure. 50 mL of 5% Ethyl acetate – hexane solvent mixture was added to the residue under stirring at room temperature. The mixture was filtered and washed with 5 mL of chilled hexane. Solid was dried under reduced pressure at room temperature. Yield: 5.03 g (83 %), (M++l) 393, 1JINMR (400 MHz, OMSO-d6 ):δ 0.93 ( s, 6H), 1.113 – 1.069 (m, 2H), 1.861-1.644 (m, 21H), 2.033-2.015 (d, 2H), 2.948-2.933 (d, 2H).
Example 6: Preparation of c/s-adamantane-2-spiro-3′ -8 ‘-πT(2′-amino-2’ -methyl propyl) amino! carbonyl] methyli-l ‘, 2\ 4′-U-JoXaSpJrQ [4.51 decane maleate To a solution of c/s-adamantane-2-spiro-3′-8′-[[[(2′-amino-2’-methyl propyl) amino] carbonyl] methyl]-! ‘, 2′, 4’-trioxaspiro [4.5] decane (example 5) (60 g, 0.153 moles) in ethanol (150 mL) was added a solution of maleic acid (17.3 g, 0.15 moles, 0.98 equiv. in ethanol 90 mL) and the reaction mixture was stirred for about 1 h. To this clear solution, n- heptane (720 mL) was added at room temperature in 1 h and the reaction mixture was stirred for 3 h. It was then cooled to 0 to 100C and filtered. The cake was washed with n-heptane (60 mL) and dried under vacuum at 40-450C.
Yield: 67 g, 77.4%, mp: 1490C (decomp), (M++l) 393.5, 1HNMR (300 MHz, DMSO-^ ): δ 1.05-1.11 (2H,m), 1.18 (6H,s), 1.64-1.89 (21H,m), 2.07(2H,d), 3.21 (2H,d), 6.06 (2H,d), 7.797 (2H, bs), 8.07 (IH, t).



References
- Dong, Yuxiang; Wittlin, Sergio; Sriraghavan, Kamaraj; Chollet, Jacques; Charman, Susan A.; Charman, William N.; Scheurer, Christian; Urwyler, Heinrich et al. (2010). “The Structure−Activity Relationship of the Antimalarial Ozonide Arterolane (OZ277)”. Journal of Medicinal Chemistry 53 (1): 481–91. doi:10.1021/jm901473s. PMID 19924861.
- Blow to Ranbaxy drug research plans at LiveMint.com, Sep 21 2007
- Vennerstrom, Jonathan L.; Arbe-Barnes, Sarah; Brun, Reto; Charman, Susan A.; Chiu, Francis C. K.; Chollet, Jacques; Dong, Yuxiang; Dorn, Arnulf et al. (2004). “Identification of an antimalarial synthetic trioxolane drug development candidate”. Nature 430 (7002): 900–4.doi:10.1038/nature02779. PMID 15318224.
- In the Pipeline: “Ozonides As Drugs: What Will They Think Of Next?”, by Derek Lowe, November 23, 2009, at Corante.com
- Indian company starts Phase III trials of synthetic artemisinin, May 4 2009, at the WorldWide Antimalarial Resistance Network
- http://www.nature.com/nature/journal/v430/n7002/full/nature02779.html
|
5-27-2011
|
PROCESS FOR THE PREPARATION OF DISPIRO 1,2,4-TRIOXOLANE ANTIMALARIALS (OZ277)
|
|
|
2-13-2009
|
STABLE DOSAGE FORMS OF SPIRO AND DISPIRO 1,2,4-TRIOXOLANE ANTIMALARIALS
|
|
|
6-15-2005
|
Spiro and dispiro 1,2,4-trioxolane antimalarials
|
|
|
11-31-2004
|
Spiro and dispiro 1,2,4-trixolane antimalarials
|
ANTIMALARIALS



http://www.rsc.org/chemistryworld/2013/03/new-antimalarial-drug-class-resistance-elq-300-quinolone

http://www.nature.com/nrd/journal/v9/n11/full/nrd3301.html
Structure of NITD609; the 1R,3Sconfiguration is fundamental for its antimalarial activity
References
- Jump up^ Dong, Yuxiang; Wittlin, Sergio; Sriraghavan, Kamaraj; Chollet, Jacques; Charman, Susan A.; Charman, William N.; Scheurer, Christian; Urwyler, Heinrich; et al. (2010). “The Structure−Activity Relationship of the Antimalarial Ozonide Arterolane (OZ277)”. Journal of Medicinal Chemistry. 53 (1): 481–91. doi:10.1021/jm901473s. PMID 19924861.
- ^ Jump up to:a b Blow to Ranbaxy drug research plans at LiveMint.com, Sep 21 2007
- Jump up^ Vennerstrom, Jonathan L.; Arbe-Barnes, Sarah; Brun, Reto; Charman, Susan A.; Chiu, Francis C. K.; Chollet, Jacques; Dong, Yuxiang; Dorn, Arnulf; et al. (2004). “Identification of an antimalarial synthetic trioxolane drug development candidate”. Nature. 430 (7002): 900–4. doi:10.1038/nature02779. PMID 15318224.
- Jump up^ In the Pipeline: “Ozonides As Drugs: What Will They Think Of Next?”, by Derek Lowe; published November 23, 2009; retrieved November 17, 2015; at Sciencemag.org
- ^ Jump up to:a b Akshat Rathi for Chemistry World. 3 May 2012 Ranbaxy launches new anti-malarial Synriam
- Jump up^ India Clinical trials registry CTRI/2009/091/000531
- Jump up^ Toure OA et al. Efficacy and safety of fixed dose combination of arterolane maleate and piperaquine phosphate dispersible tablets in paediatric patients with acute uncomplicated Plasmodium falciparum malaria: a phase II, multicentric, open-label study. Malar J. 2015 Nov 25;14(1):469. Clinical Trial Registry India: CTRI/2009/091/000531. PMID 26608469 PMC4660726
- Jump up^ Staff, Business Standard. December 16, 2014 Ranbaxy receives approval for malaria drug Synriam from 7 African countries
REFERENCES
1: Valecha N, Savargaonkar D, Srivastava B, Rao BH, Tripathi SK, Gogtay N, Kochar SK, Kumar NB, Rajadhyaksha GC, Lakhani JD, Solanki BB, Jalali RK, Arora S, Roy A, Saha N, Iyer SS, Sharma P, Anvikar AR. Comparison of the safety and efficacy of fixed-dose combination of arterolane maleate and piperaquine phosphate with chloroquine in acute, uncomplicated Plasmodium vivax malaria: a phase III, multicentric, open-label study. Malar J. 2016 Jan 27;15:42. doi: 10.1186/s12936-016-1084-1. PubMed PMID: 26818020; PubMed Central PMCID: PMC4728808.
2: Saha N, Moehrle JJ, Zutshi A, Sharma P, Kaur P, Iyer SS. Safety, tolerability and pharmacokinetic profile of single and multiple oral doses of arterolane (RBx11160) maleate in healthy subjects. J Clin Pharmacol. 2014 Apr;54(4):386-93. doi: 10.1002/jcph.232. PubMed PMID: 24242999.
3: Toure OA, Rulisa S, Anvikar AR, Rao BS, Mishra P, Jalali RK, Arora S, Roy A, Saha N, Iyer SS, Sharma P, Valecha N. Efficacy and safety of fixed dose combination of arterolane maleate and piperaquine phosphate dispersible tablets in paediatric patients with acute uncomplicated Plasmodium falciparum malaria: a phase II, multicentric, open-label study. Malar J. 2015 Nov 25;14:469. doi: 10.1186/s12936-015-0982-y. PubMed PMID: 26608469; PubMed Central PMCID: PMC4660726.
4: Toure OA, Valecha N, Tshefu AK, Thompson R, Krudsood S, Gaye O, Rao BH, Sagara I, Bose TK, Mohanty S, Rao BS, Anvikar AR, Mwapasa V, Noedl H, Arora S, Roy A, Iyer SS, Sharma P, Saha N, Jalali RK; AM–PQP Study Team.. A Phase 3, Double-Blind, Randomized Study of Arterolane Maleate-Piperaquine Phosphate vs Artemether-Lumefantrine for Falciparum Malaria in Adolescent and Adult Patients in Asia and Africa. Clin Infect Dis. 2016 Apr 15;62(8):964-71. doi: 10.1093/cid/ciw029. PubMed PMID: 26908796; PubMed Central PMCID: PMC4803108.
5: Fontaine SD, Spangler B, Gut J, Lauterwasser EM, Rosenthal PJ, Renslo AR. Drug delivery to the malaria parasite using an arterolane-like scaffold. ChemMedChem. 2015 Jan;10(1):47-51. doi: 10.1002/cmdc.201402362. PubMed PMID: 25314098; PubMed Central PMCID: PMC4420023.
6: Gupta A, Singh Y, Srinivas KS, Jain G, Sreekumar VB, Semwal VP. Development and validation of a headspace gas chromatographic method for the determination of residual solvents in arterolane (RBx11160) maleate bulk drug. J Pharm Bioallied Sci. 2010 Jan;2(1):32-7. doi: 10.4103/0975-7406.62706. PubMed PMID: 21814428; PubMed Central PMCID: PMC3146089.
7: Gautam A, Ahmed T, Sharma P, Varshney B, Kothari M, Saha N, Roy A, Moehrle JJ, Paliwal J. Pharmacokinetics and pharmacodynamics of arterolane maleate following multiple oral doses in adult patients with P. falciparum malaria. J Clin Pharmacol. 2011 Nov;51(11):1519-28. doi: 10.1177/0091270010385578. PubMed PMID: 21148048.
8: Patil C, Katare S, Baig M, Doifode S. Fixed dose combination of arterolane and piperaquine: a newer prospect in antimalarial therapy. Ann Med Health Sci Res. 2014 Jul;4(4):466-71. doi: 10.4103/2141-9248.139270. Review. PubMed PMID: 25221689; PubMed Central PMCID: PMC4160665.
9: Valecha N, Looareesuwan S, Martensson A, Abdulla SM, Krudsood S, Tangpukdee N, Mohanty S, Mishra SK, Tyagi PK, Sharma SK, Moehrle J, Gautam A, Roy A, Paliwal JK, Kothari M, Saha N, Dash AP, Björkman A. Arterolane, a new synthetic trioxolane for treatment of uncomplicated Plasmodium falciparum malaria: a phase II, multicenter, randomized, dose-finding clinical trial. Clin Infect Dis. 2010 Sep 15;51(6):684-91. doi: 10.1086/655831. PubMed PMID: 20687837.
10: Lanteri CA, Chaorattanakawee S, Lon C, Saunders DL, Rutvisuttinunt W, Yingyuen K, Bathurst I, Ding XC, Tyner SD. Ex vivo activity of endoperoxide antimalarials, including artemisone and arterolane, against multidrug-resistant Plasmodium falciparum isolates from Cambodia. Antimicrob Agents Chemother. 2014 Oct;58(10):5831-40. doi: 10.1128/AAC.02462-14. PubMed PMID: 25049252; PubMed Central PMCID: PMC4187925.
11: Valecha N, Krudsood S, Tangpukdee N, Mohanty S, Sharma SK, Tyagi PK, Anvikar A, Mohanty R, Rao BS, Jha AC, Shahi B, Singh JP, Roy A, Kaur P, Kothari M, Mehta S, Gautam A, Paliwal JK, Arora S, Saha N. Arterolane maleate plus piperaquine phosphate for treatment of uncomplicated Plasmodium falciparum malaria: a comparative, multicenter, randomized clinical trial. Clin Infect Dis. 2012 Sep;55(5):663-71. doi: 10.1093/cid/cis475. PubMed PMID: 22586253.
12: Dong Y, Wittlin S, Sriraghavan K, Chollet J, Charman SA, Charman WN, Scheurer C, Urwyler H, Santo Tomas J, Snyder C, Creek DJ, Morizzi J, Koltun M, Matile H, Wang X, Padmanilayam M, Tang Y, Dorn A, Brun R, Vennerstrom JL. The structure-activity relationship of the antimalarial ozonide arterolane (OZ277). J Med Chem. 2010 Jan 14;53(1):481-91. doi: 10.1021/jm901473s. PubMed PMID: 19924861.
13: Jourdan J, Matile H, Reift E, Biehlmaier O, Dong Y, Wang X, Mäser P, Vennerstrom JL, Wittlin S. Monoclonal Antibodies That Recognize the Alkylation Signature of Antimalarial Ozonides OZ277 (Arterolane) and OZ439 (Artefenomel). ACS Infect Dis. 2016 Jan 8;2(1):54-61. PubMed PMID: 26819968; PubMed Central PMCID: PMC4718528.
14: Fügi MA, Wittlin S, Dong Y, Vennerstrom JL. Probing the antimalarial mechanism of artemisinin and OZ277 (arterolane) with nonperoxidic isosteres and nitroxyl radicals. Antimicrob Agents Chemother. 2010 Mar;54(3):1042-6. doi: 10.1128/AAC.01305-09. PubMed PMID: 20028825; PubMed Central PMCID: PMC2825978.
15: Mossallam SF, Amer EI, El-Faham MH. Efficacy of Synriam™, a new antimalarial combination of OZ277 and piperaquine, against different developmental stages of Schistosoma mansoni. Acta Trop. 2015 Mar;143:36-46. doi: 10.1016/j.actatropica.2014.12.005. PubMed PMID: 25530543.
16: Longo M, Zanoncelli S, Brughera M, Colombo P, Wittlin S, Vennerstrom JL, Moehrle J, Craft JC. Comparative embryotoxicity of different antimalarial peroxides: in vitro study using the rat whole embryo culture model (WEC). Reprod Toxicol. 2010 Dec;30(4):583-90. doi: 10.1016/j.reprotox.2010.07.011. PubMed PMID: 20708075.
17: Abiodun OO, Brun R, Wittlin S. In vitro interaction of artemisinin derivatives or the fully synthetic peroxidic anti-malarial OZ277 with thapsigargin in Plasmodium falciparum strains. Malar J. 2013 Jan 31;12:43. doi: 10.1186/1475-2875-12-43. PubMed PMID: 23368889; PubMed Central PMCID: PMC3566918.
18: White NJ. Can new treatment developments combat resistance in malaria? Expert Opin Pharmacother. 2016 Jul;17(10):1303-7. doi: 10.1080/14656566.2016.1187134. PubMed PMID: 27191998.
19: Wang X, Dong Y, Wittlin S, Charman SA, Chiu FC, Chollet J, Katneni K, Mannila J, Morizzi J, Ryan E, Scheurer C, Steuten J, Santo Tomas J, Snyder C, Vennerstrom JL. Comparative antimalarial activities and ADME profiles of ozonides (1,2,4-trioxolanes) OZ277, OZ439, and their 1,2-dioxolane, 1,2,4-trioxane, and 1,2,4,5-tetraoxane isosteres. J Med Chem. 2013 Mar 28;56(6):2547-55. doi: 10.1021/jm400004u. PubMed PMID: 23489135.
20: Marfurt J, Chalfein F, Prayoga P, Wabiser F, Wirjanata G, Sebayang B, Piera KA, Wittlin S, Haynes RK, Möhrle JJ, Anstey NM, Kenangalem E, Price RN. Comparative ex vivo activity of novel endoperoxides in multidrug-resistant plasmodium falciparum and P. vivax. Antimicrob Agents Chemother. 2012 Oct;56(10):5258-63. doi: 10.1128/AAC.00283-12. PubMed PMID: 22850522; PubMed Central PMCID: PMC3457353.
 |
|
| Clinical data | |
|---|---|
| Routes of administration |
Oral |
| ATC code |
|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C22H36N2O4 |
| Molar mass | 392.531 g/mol |
| 3D model (JSmol) | |
////////////Arteolane Maleate, OZ-277, RBx-11160, OZ 277, RBx 11160, OZ277, RBx11160, IND 2017
O=C(NCC(C)(N)C)C[C@H](CC1)CC[C@@]21OC3(OO2)[C@H]4C[C@H]5C[C@@H]3C[C@@H](C4)C5.O=C(O)/C=C\C(O)=O
Bepotastine Besilate, ベポタスチンベシル酸塩
Bepotastine Besilate
ベポタスチンベシル酸塩
- Molecular FormulaC27H31ClN2O6S
- Average mass547.063 Da
HL-151
-
- Use:antiallergic, antihistaminic
| 10 mg Tablets | For the treatment of allergic rhinitis | 27.03.2017 CDSCO
APPROVED |
USFDA
NDA 22-288 Bepotastine Besilate 1.5% Ophthalmic Solution ISTA Pharmaceuticals, Inc.
https://www.accessdata.fda.gov/drugsatfda_docs/nda/2009/02228s000_ChemR.pdf
Drug Substance Bepotastine besilate is manufactured by Ube Industries and the information for the NDA is submitted through DMF #19966. Bepotastine besilate is a white crystalline powder with no odor and bitter taste. It is very soluble in but sparingly soluble in . It is stable when exposed to light, and optically active. The S-isomer is the active drug and is controlled as an impurity through synthesis. The distribution coefficient in 1-octanol is higher than in aqueous buffer in the pH 5-9 range. There are 10 potential impurities but only one impurity is above 0.1%. Two potential genotoxic impurities are controlled below . Residual is controlled below Bepotastine besilate is stable under long term storage conditions for (25ºC/60% RH) over 5 years
Bepotastine besilate was originally developed as an oral tablet dosage form and got approval in Japan in 2000 for allergic rhinitis. It is a non-sedating anti-allergic drug. The proposed NDA is an ophthalmic solution indicated for allergic conjunctivitis. Bepotastine besilate ophthalmic solution 1.5% is a sterile solution. It is an aqueous solution to be administered as drops at or near physiological pH range of tears. The formulation contains sodium chloride, monobasic sodium phosphate as dihydrate, benzalkonium chloride, sodium hydroxide and purified water; typically these components are used for , preservative action, pH adjustment,
INTRO
Bepotastine is a non-sedating, selective antagonist of the histamine 1 (H1) receptor. Bepotastine was approved in Japan for use in the treatment of allergic rhinitis and uriticaria/puritus in July 2000 and January 2002, respectively, and is marketed by Tanabe Seiyaku Co., Ltd. under the brand name Talion. It is available in oral and opthalmic dosage forms in Japan. The opthalmic solution is FDA approved since Sept 8, 2009 and is under the brand name Bepreve.
Tae Hee Ha, Chang Hee Park, Won Jeoung Kim, Soohwa Cho, Han Kyong Kim, Kwee Hyun Suh, “PROCESS FOR PREPARING BEPOTASTINE AND INTERMEDIATES USED THEREIN.” U.S. Patent US20100168433, issued July 01, 2010., US20100168433
BEPREVE® (bepotastine besilate ophthalmic solution) 1.5% is a sterile, topically administered drug for ophthalmic use. Each mL of BEPREVE contains 15 mg bepotastine besilate.
Bepotastine besilate is designated chemically as (+) -4-[[(S)-p-chloro-alpha -2pyridylbenzyl] oxy]-1-piperidine butyric acid monobenzenesulfonate. The chemical structure for bepotastine besilate is:
 |
Bepotastine besilate is a white or pale yellowish crystalline powder. The molecular weight of bepotastine besilate is 547.06 daltons. BEPREVE ophthalmic solution is supplied as a sterile, aqueous 1.5% solution, with a pH of 6.8.
The osmolality of BEPREVE (bepotastine besilate ophthalmic solution) 1.5% is approximately 290 mOsm/kg.
ベポタスチンベシル酸塩 JP17
Bepotastine Besilate

C21H25ClN2O3▪C6H6O3S : 547.07
[190786-44-8]
Bepotastine (Talion, Bepreve) is a 2nd generation antihistamine.[1] It was approved in Japan for use in the treatment of allergic rhinitisand urticaria/pruritus in July 2000 and January 2002, respectively. It is currently marketed in the United States under the brand-name Bepreve, by ISTA Pharmaceuticals.
Bepotastine besilate is a second-generation antihistamine that was launched in a tablet formulation under a collaboration between Tanabe Seiyaku and Ube in 2000 and in 2002 for the treatment of allergic rhinitis including sneeze, mucus discharge and solidified mucus, and for the treatment of urticaria, respectively. An orally disintegrating tablet was made available in Japan in 2006, while a dry syrup formulation for the treatment of allergic rhinitis was studied in clinical trials at Tanabe Seiyaku for the treatment of allergic rhinitis
Originally developed at Ube, bepotastine besilate was later licensed to Tanabe Seiyaku as part of a collaboration agreement. In 2010, rights were licensed to Dong-A and Mitsubishi Tanabe Pharma in Korea for the treatment of eye disorders.
Pharmacology
Bepotastine is available as an ophthalmic solution and oral tablet. It is a direct H1-receptor antagonist that inhibits the release of histamine from mast cells.[2] The ophthalmic formulation has shown minimal systemic absorption, between 1 and 1.5% in healthy adults.[3] Common side effects are eye irritation, headache, unpleasant taste, and nasopharyngitis.[3] The main route of elimination is urinary excretion, 75-90% excreted unchanged.[3]
Marketing history
It is marketed in Japan by Tanabe Seiyaku under the brand name Talion. Talion was co-developed by Tanabe Seiyaku and Ube Industries, the latter of which discovered bepotastine. In 2001, Tanabe Seiyaku granted Senju, now owned by Allergan, exclusive worldwide rights, with the exception of certain Asian countries, to develop, manufacture and market bepotastine for ophthalmic use. Senju, in turn, has granted the United States rights for the ophthalmic preparation to ISTA Pharmaceuticals.
Sales and patents
In 2011, ISTA pharmaceuticals experienced a 2.4% increase in net revenues from 2010, which was driven by the sales of Bepreve. Their net revenue for 2011 was $160.3 million.[4] ISTA Pharmaceuticals was acquired by Bausch & Lomb in March 2012 for $500 million.[5] Bausch & Lomb hold the patent for bepotastine besilate (https://www.accessdata.fda.gov/scripts/cder/ob/docs/temptn.cfm. On November 26, 2014, Bausch & Lomb sue Micro Labs USA for patent infringement.[6] Bausch & Lomb was recently bought out by Valeant Pharmaceuticals in May 2013 for $8.57 billion, Valeant’s largest acquisition to date, causing the company’s stock to rise 25% when the deal was announced.[7]
Clinical trials
A Phase III clinical trial was carried out in 2010 to evaluate the effectiveness of bepotastine besilate ophthalmic solutions 1.0% and 1.5%.[8] These solutions were compared to a placebo and evaluated for their ability to reduce ocular itchiness. The study was carried out with 130 individuals and evaluated after 15 minutes, 8 hours, or 16 hours. There was a reduction in itchiness at all-time points for both ophthalmic solutions. The study concluded that bepotastine besilate ophthalmic formulations reduced ocular itchiness for at least 8 hours after dosing compared to placebo. Phase I and II trials were carried out in Japan.
Studies have been performed in animals and bepotastine besilate was not found to be teratogenic in rats during fetal development, even at 3,300 times more that typical use in humans.[3] Evidence of infertility was seen in rats at 33,000 times the typical ocular does in humans.[3] The safety and efficacy has not been established in patients under 2 years of age and has been extrapolated from adults for patients under 10 years of age.[3]
SYN
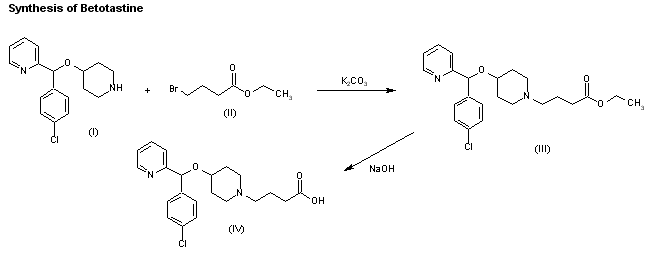
EP 0335586; JP 1989242574; JP 1990025465; JP 1993294929; US 4929618
The reaction of 4-[1-(4-chlorophenyl)-1-(2-pyridyl)methoxy]piperidine (I) with ethyl 4-bromobutyrate (II) by means of K2CO3 in refluxing acetone gives the corresponding condensation product (III), which is then hydrolyzed with NaOH in ethanol/water yielding compound (IV).
SYN 2
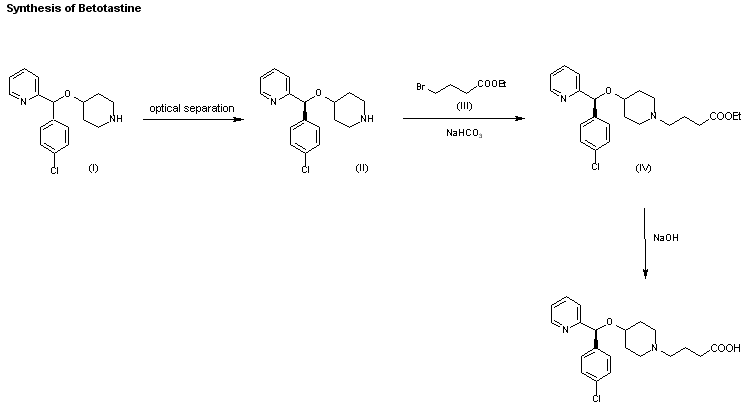
JP 1998237070; JP 2000198784; WO 9829409
A new synthesis of betotastine has been developed: The racemic 4-[1-(4-chlorophenyl)-1-(2-pyridyl)methoxy]piperidine (I) is submitted to optical resolution with N-acyl amino acids such as N-acetyl-L-phenylalanine (preferred), N-acetyl-L-leucine, N-(benzyloxycarbonyl)-L-phenylalanine, N-(benzyloxycarbonyl)-L-valine, N-(benzyloxycarbonyl)-L-threonine, N-(benzyloxycarbonyl)-L-serine or with (2R,3R)-3-(5-chloro-2-nitrophenylsulfanyl)-2-hydroxy-3-(4-methoxyphenyl)propionic acid (preferred) or (2R,3R)-2-hydroxy-3-(4-methoxyphenyl)-3-(2-nitrophenylsulfanyl)propionic acid as chiral intermediates, yielding the (S)-isomer (II). The condensation of (II) with ethyl 4-bromobutyrate (III) by means of a base such as Na2CO3, NaHCO3, K2CO3 or KHCO3 gives the expected 4-(1-piperidinyl)butyric acid ester (IV), which is finally hydrolyzed with NaOH or KOH in aqueous ethanol or methanol.
SYN 3

A new synthesis of betotastine has been developed: The racemic 4-[1-(4-chlorophenyl)-1-(2-pyridyl)methoxy]piperidine (I) is submitted to optical resolution with N-acyl amino acids such as N-acetyl-L-phenylalanine (preferred), N-acetyl-L-leucine, N-(benzyloxycarbonyl)-L-phenylalanine, N-(benzyloxycarbonyl)-L-valine, N-(benzyloxycarbonyl)-L-threonine, N-(benzyloxycarbonyl)-L-serine or with (2R,3R)-3-(5-chloro-2-nitrophenylsulfanyl)-2-hydroxy-3-(4-methoxyphenyl)propionic acid (preferred) or (2R,3R)-2-hydroxy-3-(4-methoxyphenyl)-3-(2-nitrophenylsulfanyl)propionic acid as chiral intermediates, yielding the (S)-isomer (II). The condensation of (II) with ethyl 4-bromobutyrate (III) by means of a base such as Na2CO3, NaHCO3, K2CO3 or KHCO3 gives the expected 4-(1-piperidinyl)butyric acid ester (IV), which is finally hydrolyzed with NaOH or KOH in aqueous ethanol or methanol.
CLIP
A Novel Synthetic Method for Bepotastine, a Histamine H1 Receptor …
file:///C:/Users/91200291/Downloads/B130241_549.pdf


Scheme 1. Synthesis of bepotastine l-menthyl ester N-benzyloxycarbonyl-L-aspartic acid complex (3), bepotastine besilate (4) and bepotastine calcium (5). Reagents and conditions; i) 4-bromobutanoic acid l-menthyl ester, K2CO3, acetone, reflux, 7 h, 95-99%; ii) N-benzyloxycarbonyl-L-aspartic acid (NCbzLAA), ethyl acetate, rt, 12 h, 71-73%; iii) Ethyl acetate/H2O, NaHCO3, 97-99%; iv) EtOH:H2O = 1:1, NaOH, rt, 12 h, 3.0 N-HCl Neutralization, 92- 95%; v) AcOH, reflux, 12 h, racemization 97-100%; vi) Bezensulfonic acid, acetonitrile, rt, 12 h, 64-67%; vii) NaOH, H2O, CaCl2, rt, 12 h, 86-89%.
Synthesis of (S)-Bepotastine Besilate (4). Bepotastine (50 g, 0.13 mol) was dissolved in 500 mL of acetonitrile, and benzenesulfonic acid monohydrate (20 g, 0.11 mol) was added to the reaction mixture. Bepotastine besilate (0.5 g, 1.28 mmol) was seeded in the reaction mixture and stirred at rt for 12 h. The solid precipitate was filtered and dried. The product was obtained 38 g (yield: 64%, optical purity: 99.5% ee) as a pale white crystalline powder. Melting point: 161- 163 o C. Water: 0.2% (Karl-Fischer water determination). MS: m/z 389.1 [M+H]; 1 H-NMR (300 MHz, DMSO-d6) δ 9.2 (br s, 1H), 8.5 (d, J = 4.1 Hz, 1H), 7.8 (t, J = 7.7 Hz, 1H), 7.6 (m, 3H), 7.4 (m, 4H), 7.3 (m, 4H), 5.7 (s, 1H), 3.7 (br s, 2H), 3.3 (br s, 3H), 3.1 (br s, 2H), 2.3 (t, J = 14.1 Hz, 2H), 2.2 (m, 1H), 2.0 (m, 1H), 1.8 (m, 3H), 1.7 (m, 1H); IR (KBr, cm−1 ): 3422, 2996, 2909, 2735, 2690, 2628, 1719, 1592, 1572, 1488, 1470, 1436, 1411, 1320, 1274, 1221, 1160, 1123, 1066, 1031, 1014, 996, 849, 830, 771, 759, 727, 693, 612, 564
Synthesis
FDA Orange Book Patents
| FDA Orange Book Patents: 1 of 3 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 8784789 |
| Expiration | Sep 5, 2024 |
| Applicant | BAUSCH AND LOMB INC |
| Drug Application | N022288 (Prescription Drug: BEPREVE. Ingredients: BEPOTASTINE BESILATE) |
| FDA Orange Book Patents: 2 of 3 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 6780877 |
| Expiration | Sep 19, 2019 |
| Applicant | BAUSCH AND LOMB INC |
| Drug Application | N022288 (Prescription Drug: BEPREVE. Ingredients: BEPOTASTINE BESILATE) |
| FDA Orange Book Patents: 3 of 3 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 8877168 |
| Expiration | Jul 30, 2023 |
| Applicant | BAUSCH AND LOMB INC |
| Drug Application | N022288 (Prescription Drug: BEPREVE. Ingredients: BEPOTASTINE BESILATE) |
 |
|
| Clinical data | |
|---|---|
| Trade names | Bepreve |
| AHFS/Drugs.com | International Drug Names |
| MedlinePlus | a610012 |
| Pregnancy category |
|
| Routes of administration |
Oral, topical (eye drops) |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | High (oral) Minimal (topical) |
| Protein binding | ~55% |
| Excretion | Renal (75–90%) |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C21H25ClN2O3 |
| Molar mass | 388.88 g/mol |
| 3D model (JSmol) | |
References
-
- EP 335 586 (Ube Ind.; appl. 22.3.1989; J-prior. 25.3.1988).
- EP 485 984 (Ube Ind.; appl. 13.11.1991; J-prior. 15.11.1990).
- WO 9 829 409 (Ube Ind.; appl. 25.12.1997; J-prior. 26.12.1996).
-
racemization :
- JP 10 237 069 (Ube Ind.; appl. 21.2.1997).
References
- Jump up^ H. Takahashi; A. Ishida-Yamamoto; H. Iizuka (September 2004). “Effects of bepotastine, cetirizine, fexofenadine, and olopatadine on histamine-induced wheal-and flare-response, sedation, and psychomotor performance”. Clinical and Experimental Dermatology. 29: 526–532. doi:10.1111/j.1365-2230.2004.01618.x.
- Jump up^ “Bepotastine Monograph”. LexiComp.
- ^ Jump up to:a b c d e f “Bepreve prescribing Information” (PDF).
- Jump up^ [phx.corporate-ir.net/External.File?item…t=1 “2011 Net Revenues Increase to $160.3 Million On an Adjusted Cash Net Income Basis, ISTA Posts Second Year of Profitability Company Reaffirms 2012 Financial Guidance”] Check
|url=value (help). - Jump up^ “Bausch & Lomb to Buy ISTA Pharmaceuticals for $500 Million”. DealBook. Retrieved 2015-12-05.
- Jump up^ “Bausch & Lomb Inc. et al. v. Micro Labs USA, Inc. et al.”
- Jump up^ “Valenant pharmaceuticals eyes China with Bausch deal”.
- Jump up^ Macejko, Thomas T.; Bergmann, Mark T.; Williams, Jon I.; Gow, James A.; Gomes, Paul J.; McNamara, Timothy R.; Abelson, Mark B. (2010-07-01). “Multicenter Clinical Evaluation of Bepotastine Besilate Ophthalmic Solutions 1.0% and 1.5% to Treat Allergic Conjunctivitis”. American Journal of Ophthalmology. 150 (1): 122–127.e5. doi:10.1016/j.ajo.2010.02.007.
////////////Bepotastine Besilate, ベポタスチンベシル酸塩 ,Talion , tau284, TAU-284DS, TAU-284, DA-5206
HL-151 , SNJ-1773
C1CN(CCC1OC(C2=CC=C(C=C2)Cl)C3=CC=CC=N3)CCCC(=O)O.C1=CC=C(C=C1)S(=O)(=O)O

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....