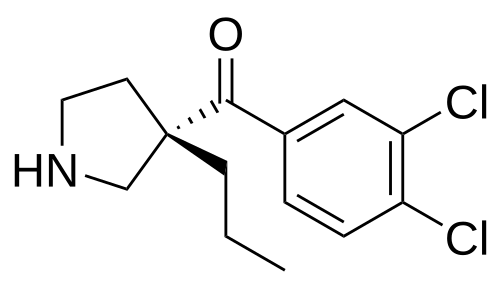
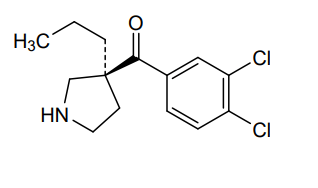
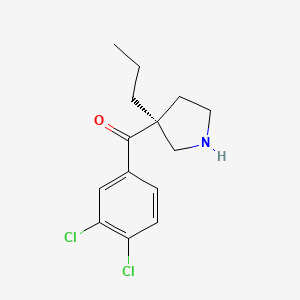
Cendifensine
CAS 1034048-49-1
MF C14H17Cl2NO MW286.2 g/mol
Methanone, (3,4-dichlorophenyl)[(3S)-3-propyl-3-pyrrolidinyl]-
(3,4-dichlorophenyl)[(3S)-3-propylpyrrolidin-3-yl]methanone
monoamine reuptake inhibitor, NOE-115, NOE 115, N4U2JR8GCX,
Cendifensine (INNTooltip International Nonproprietary Name) is a monoamine reuptake inhibitor (MRI)[1] related to the amphetamines and cathinones which has not been marketed at this time.[2][3][4] It was first described by 2013[4] and its INNTooltip International Nonproprietary Name was proposed in 2024.[2] The drug has been patented by Noema Pharma, which is developing a serotonin–norepinephrine–dopamine reuptake inhibitor (SNDRI) known as NOE-115 for the treatment of vasomotor symptoms associated with menopause[5] as well as for binge-eating disorder and depressive disorders.[6][7][8]
- OriginatorRoche
- DeveloperNoema Pharma
- ClassAlkanes; Antidepressants; Behavioural disorder therapies; Chlorobenzenes; Ketones; Pyrrolidines; Small molecules
- Mechanism of ActionAdrenergic uptake inhibitors; Dopamine uptake inhibitors; Serotonin uptake inhibitors
- Phase IIVasomotor symptoms
- No development reportedBinge-eating disorder; Depressive disorders
- 28 Jan 2026No recent reports of development identified for phase-I development in Binge-Eating-Disorder in Switzerland (Parenteral)
- 28 Jan 2026No recent reports of development identified for phase-I development in Depressive disorders in Switzerland (Parenteral)
- 17 Nov 2025Chemical structure information added.
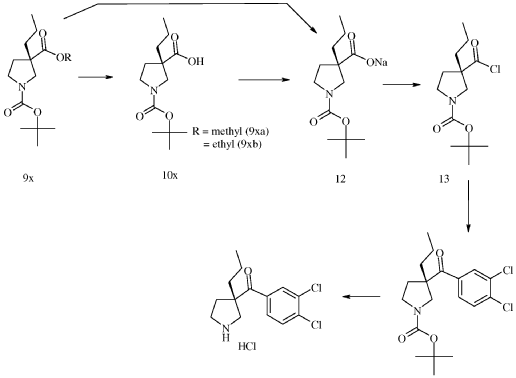
SYN
WO2023161533
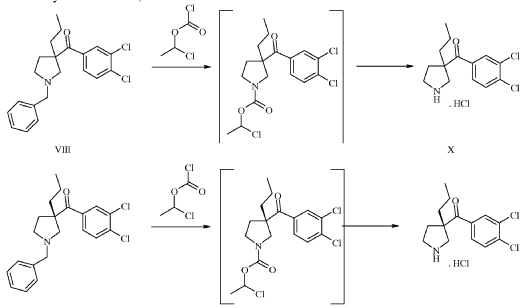
Example 1. Synthesis of (3,4-dichloro-phenyl)-((S)-3-propyl-pyrrolidin-3-yl)-methanone hydrochloride (15) quarterhydrate [See U.S. Patent No. 9,527,810]
0123-(l-benzyl-3-propylpyrrolidin-3-yl)(3,4-dichlorophenyl)methanone (IX-1) (5 g, 13.3 mmol, Eq: 1.00, see U.S. Patent No. 9,527,810 for synthesis) was dissolved in dichloromethane (30 mL). The light yellow solution was cooled to 0-5 °C and N-ethyldiisopropylamine (172 mg, 226 pL, 1.33 mmol, Eq: 0.1) was added. 1-Chloroethyl chloroformate (2.28 g, 1.74 ml, 15.9 mmol, Eq: 1.2) was added dropwise while the temperature was maintained in between 0-5 °C. The reaction was warmed to room temperature over 30 min and was stirred 1 h at room temperature. Methanol (25 mL) was added and the light yellow solution was heated to 40 °C for 40 min. The reaction mixture was concentrated under reduced pressure (40 °C, 600-15 mbar) to give 5.48 g of crude product. Ethyl acetate (30.0 mL) was added and the suspension was heated to 50 °C. A solution of water (239 mg, 239 pL, 13.3 mmol, Eq: 1.0) in ethyl acetate (35 mL) was added over 10 min. The white suspension was stirred for 1 h at 50 °C and cooled to room temperature over 1.5 h. The suspension was filtered, and the filter cake was washed twice with ethyl acetate (10 mL) and dried under reduced pressure (40° C, 15 mbar) to give 4.02 g of (15) as quarterhydrate (93% yield).
SYN
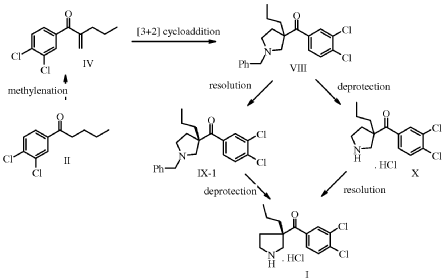
Synthesis of l-(3,4-Dichloro-phenyl)-pentan-l-one (II)
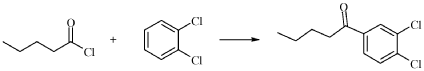
Aluminum chloride (12.4 g, 93.3 mmol, Eq: 1.5) was charged in the reactor followed by 1,2-dichlorobenzene (27.4 g, 21.0 ml, 187 mmol, Eq: 3). The suspension was heated to 80°C in 10 min and pentanoyl chloride (7.5 g, 7.58 ml, 62.2 mmol, Eq: 1.00) was added dropwise over 30 min. The reaction mixture went from a yellow suspension to an orange/brown viscous solution. After 5h reaction at 80°C the deep orange/brown reaction mixture was cooled to 25°C and stirred at 25 °C overnight. The reaction mixture was poured onto a mixture of n-heptane (68.4 g, 100 ml) and water/ice 50:50 (100 g, 100 ml). The organic phase was separated and washed with water (50.0 g, 50 ml) then with NaHC03aq 5% (50 ml) and finally with water (50.0 g, 50 ml) The organic phase was dried azeotropically (60°C/ca 150 mbar) with n-heptane (205 g, 300 ml) to give 28g of crude product as an orange oil (ca 96:4 Product/2,3-dichlorovalerophenone isomer). The crude oil was dissolved in n-heptane (27.4 g, 40 ml) and the solution was cooled to -20°C for 2 h. The suspension was filtered. The filter was washed with cold n-heptane (10.3 g, 15 ml) and dried at 35°C/10 mbar to give 8.8 g of the title product (>98a GC, isomer <1%).
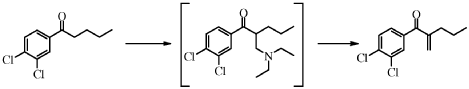
Synthesis of l-(3,4-Dichloro-phenyl)-2-methylene-pentan-l-one (IV)
II ΠΙ-1 IV
Alternative A
l-(3,4-dichlorophenyl)pentan-l-one II (15 g, 63.0 mmol, equivalents: 1.00) and paraformaldehyde (3.58 g, 113 mmol, equivalents: 1.8) were charged in the reactor followed by heptane (30.0 ml). Temperature was set at 25°C. Diethylamine (8.84 g, 12.5 ml, 120 mmol, equivalents: 1.9) was added. Paraformaldehyde partially dissolved over time. Acetic acid (11.4 g, 10.9 ml, 189 mmol, equivalents: 3) was slowly added and the reaction mixture was heated to 60°C. After 17h reaction (< 2 % starting material), deionized water (30.0 ml) was added and the reaction mixture was heated to 80°C. After completion of the reaction (usually < 5h, < 1% intermediate by HPLC), the reaction mixture was cooled to room temperature. The organic phase was separated and washed twice with 20 mL deionized water. The organic phase was
concentrated under reduced pressure and dried azeotropically with heptane to give 15.32 g of the olefin IV as orange oil (96% yield corrected for 96a% purity by HPLC).
Alternative B
l-(3,4-dichlorophenyl)pentan-l-one II (15 g, 63.0 mmol, equivalents: 1.00) and paraformaldehyde (3.58 g, 113 mmol, equivalents 1.8) were charged in the reactor followed by heptane (20.5 g, 30.0 ml). Temperature was set to 25°C. Acetic acid (11.4 g, 10.9 ml, 189 mmol, equivalents: 3) was added followed by diethylamine (8.84 g, 12.5 ml, 120 mmol, equivalents: 1.9). The reaction mixture was heated to 60°C. After 17h30 reaction (< 2% starting material), deionized water (30.0 ml) was added and the reaction mixture was heated to 80°C. After completion of the reaction (usually < 5h; < 1% intermediate by HPLC), the reaction mixture was cooled to room temperature and polish filtered. The aqueous phase was separated and discarded. The organic phase was washed twice with 20 mL deionized water and once with 10 mL 25% aqueous sodium chloride. The organic phase was concentrated under reduced pressure and dried azeotropically with heptane to give 15.53 g of the desired product IV as orange oil (99% yield, corrected for 97.7 %).
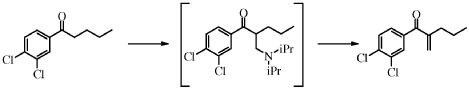
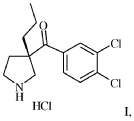
Synthesis of (3,4-Dichloro-phenyl)-(3-(S)-propyl-pyrrolidin-3-yl)-methanonehydrochloride I
Alternative A
(S)-(3,4-dichlorophenyl)(3-propylpyrrolidin-3-yl)methanone (2S,3S)-2,3-dihydroxysuccinate X-TAR (20 g, 45.7 mmol, Equivalents: 1.00) was suspended in methyl iert-butyl ether (150 ml)and treated with 2M aqueous sodium hydroxide (48.0 ml, 96.0 mmol, Equivalents: 2.1). The organic phase was separated and washed twice with water (50 ml). Ethanol (150 ml) was added to the organic extract followed by 37% hydrochloric acid (4.01 ml, 48.0 mmol, Equivalents: 1.05). The solution was concentrated under reduced pressure (300 mbar/60°C) to ca 100 mL and was polish filtered. Ethyl acetate (300 ml) was added and the solution was seeded. The resulting mixture was concentrated under reduced pressure (300 mbar/60°C) to a white suspension (ca 150 g). A solution of water (412 mg, 412 μΐ, 22.9 mmol, Equivalents: 0.5) in ethanol (15 ml) was added at room temperature. The suspension was stirred at room temperature overnight and cooled to 0°C for lh. The suspension was filtered and the filter cake was washed with cold (0°C) ethyl acetate (60 ml). The crystals were dried at 50°C under reduced pressure to give 14.3 g of product I as quarterhydrate (96% yield).
PAT
- HYPEREROYL PYROLIDINYL AND PIPERIDINYL KETONEPublication Number: BR-PI0720742-A2Priority Date: 2006-12-19
- Heteroaryl pyrrolidinyl and piperidinyl ketone derivativesPublication Number: JP-5394252-B2Priority Date: 2006-12-19Grant Date: 2014-01-22
- Heteroaryl pyrrolidinyl and piperidinyl ketone derivativesPublication Number: CA-2671378-CPriority Date: 2006-12-19Grant Date: 2015-10-20
- Heteroaryl pyrrolidinyl and piperidinyl ketone derivativesPublication Number: RU-2479575-C2Priority Date: 2006-12-19Grant Date: 2013-04-20
- Heteroaryl pyrrolidinyl and piperidinyl ketone derivatives and uses thereofPublication Number: US-8513425-B2Priority Date: 2006-12-19Grant Date: 2013-08-20
- HETEROARIL-PYRROLIDINYL AND PIPERIDINYL-CETONE DERIVATIVESPublication Number: PT-2354124-EPriority Date: 2006-12-19
- Heteroaryl pyrrolidinyl and piperidinyl ketone derivativesPublication Number: NZ-577114-APriority Date: 2006-12-19
- Heteroaryl pyrrolidinyl and piperidinyl ketone derivatives and uses thereofPublication Number: US-2012065225-A1Priority Date: 2006-12-19
- derivatives of heteroaryl pyrrolidinyl and piperidinyl ketone, their uses and pharmaceutical compositionPublication Number: BR-PI0720742-B1Priority Date: 2006-12-19
- Heteroaryl pyrrolidinyl and piperidinyl ketone derivativesPublication Number: EP-2354124-B1Priority Date: 2006-12-19Grant Date: 2013-02-13
- Heteroaryl pyrrolidinyl and piperidinyl ketone derivativesPublication Number: EP-2684871-A1Priority Date: 2006-12-19
- Heteroaryl pyrrolidinyl and piperidinyl ketone derivativesPublication Number: EP-2354124-A2Priority Date: 2006-12-19
- Pyrrolidinyl and piperidinyl ketone derivatives and uses thereofPublication Number: US-8084623-B2Priority Date: 2006-12-19Grant Date: 2011-12-27
- Heteroaryl-pyrrolidinyl- and -piperidinyl-ketone derivativesPublication Number: ES-2401129-T3Priority Date: 2006-12-19Grant Date: 2013-04-17



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
References
- WO, Garibaldi G, “Triple uptake inhibitor for the treatment of atypical depression”, published 31 August 2023, assigned to Noema Pharma AG
- “Proposed INN: List 132 International Nonproprietary Names for Pharmaceutical Substances (INN)” (PDF). WHO Drug Information. 38 (4). 2024.
cendifensinum cendifensine (3,4-dichlorophenyl)[(3S)-3-propylpyrrolidin-3-yl]methanone monoamine reuptake inhibitor […] C14H17Cl2NO 1034048-49-1
- “(3,4-Dichlorophenyl)[(3S)-3-propyl-3-pyrrolidinyl]methanone”. Global Substance Registration System (GSRS). National Center for Advancing Translational Sciences (NCATS), U.S. National Institutes of Health. Retrieved 24 February 2025.
- CA, Adam JM, Dvorak CA, Fishlock D, Humphreys ER, Iding H, Pfleger C, Rege PD, Shi X, Vitale J, Wang S, Zajac M, “(3,4-dichloro-phenyl)-((s)-3-propyl-pyrrolidin-3-yl)-methanone hydrochloride and manufacturing processes”, published 23 April 2013, assigned to F Hoffmann La Roche AG
- Young Moss S, Lee A, Simon JA (November 2025). “Advances in Pharmacotherapy for Menopausal Vasomotor Symptoms”. Drugs. 85 (11): 1363–1379. doi:10.1007/s40265-025-02231-8. PMC 12572072. PMID 41028653.
- “NOE 115”. AdisInsight. 21 May 2024. Retrieved 24 February 2025.
- “Delving into the Latest Updates on Noema Pharma AG with Synapse”. Synapse. 23 January 2025. Retrieved 24 February 2025.
- “Noema Pharma”. Noema Pharma. 18 June 2021. Retrieved 24 February 2025.
| Clinical data | |
|---|---|
| Other names | NOE-115 |
| Drug class | Monoamine reuptake inhibitor |
| Identifiers | |
| IUPAC name | |
| CAS Number | 1034048-49-1 |
| PubChem CID | 59744668 |
| ChemSpider | 42806369 |
| UNII | N4U2JR8GCX |
| Chemical and physical data | |
| Formula | C14H17Cl2NO |
| Molar mass | 286.20 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
///////////cendifensine, monoamine reuptake inhibitor, NOE-115, NOE 115, N4U2JR8GCX,

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
