- USUS5856529 A
- USUS8785492 B2
- US5856529
- US8785492
- US9060995
- US9549913
- US9539234
- US9730910
- USRE46604
- US9855241
Home » EU 2015
Category Archives: EU 2015
Fosnetupitant

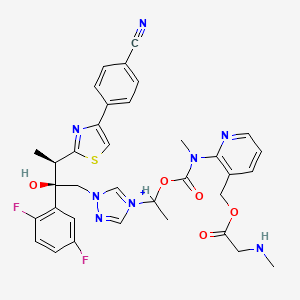
Fosnetupitant
- Molecular FormulaC31H35F6N4O5P
- Average mass688.598 Da
[4-[5-[[2-[3,5-bis(trifluoromethyl)phenyl]-2-methylpropanoyl]-methylamino]-4-(2-methylphenyl)pyridin-2-yl]-1-methylpiperazin-1-ium-1-yl]methyl hydrogen phosphate(4-{5-[{2-[3,5-Bis(trifluoromethyl)phenyl]-2-methylpropanoyl}(methyl)amino]-4-(2-methylphenyl)-2-pyridinyl}-1-methylpiperazin-1-ium-1-yl)methyl hydrogen phosphate07-PNET10146
CAS 1703748-89-3
HCL 1643757-72-5
FDA 2014 AND EMA 2015Фоснетупитант [Russian] [INN]فوسنيتوبيتانت [Arabic] [INN]磷奈匹坦 [Chinese] [INN]
- 07-PNET
In April 2018, the U.S. Food and Drug Administration (FDA) and the Swiss company Helsinn approved the intravenous formulation of AKYNZEO® (NEPA, a fixed antiemetic combination of fosnetupitant, 235mg, and palonosetron, 0.25mg) as an alternative treatment option for patients experiencing chemotherapy-induced nausea and vomiting. Fosnetupitant is the pro-drug form of netupitant. Generally, 25% to 30% of patients with a diagnosis of cancer receive chemotherapy as a treatment modality and 70% to 80% of these patients undergoing chemotherapy treatment may experience nausea and vomiting as major side effects. Considered one of the most distressing side effects of chemotherapy, nausea and vomiting has an immense impact on the quality of life of patients receiving certain antineoplastic therapies.
In April 2018, the U.S. Food and Drug Administration (FDA) and the Swiss company Helsinn approved the intravenous formulation of AKYNZEO® (NEPA, a fixed antiemetic combination of fosnetupitant, 235mg, and palonosetron, 0.25mg) as an alternative treatment option for patients experiencing chemotherapy-induced nausea and vomiting 3. Fosnetupitant is the pro-drug form of netupitant Label.
Generally, 25% to 30% of patients with a diagnosis of cancer receive chemotherapy as a treatment modality and 70% to 80% of these patients undergoing chemotherapy treatment may experience nausea and vomiting as major side effects. Considered one of the most distressing side effects of chemotherapy, nausea and vomiting has an immense impact on the quality of life of patients receiving certain antineoplastic therapies 1.
Fosnetupitant: Fosnetupitant is a selective antagonist of human substance P/neurokinin 1 (NK-1) receptors. Upon intravenous administration, Fosnetupitant is converted by phosphatases to its active form. It competitively binds to and blocks the activity of NK-1 receptors in the central nervous system, by inhibiting binding of substance P (SP) to NK-1 receptors. This prevents delayed emesis, which is associated with SP secretion. AKYNZEO is a combination of palonosetron, a serotonin-3 receptor antagonist, and Fosnetupitant (capsules for oral use) or Fosnetupitant (injections for intravenous use). AKYNZEO for injection is indicated in combination with dexamethasone in adults for the prevention of acute and delayed nausea and vomiting associated with initial and repeat courses of highly emetogenic cancer chemotherapy.
EMA
The chemical name of fosnetupitant chloride hydrochloride is 4-(5-(2-(3,5-bis(trifluoromethyl)phenyl)- N,2-dimethylpropanamido)-4-(o-tolyl)pyridin-2-yl)-1-methyl-1-((phosphonooxy)methyl)piperazin-1- ium chloride hydrochloride is corresponding to the molecular formula C31H37Cl2N4O5P. It has a relative molecular mass of 761.53 g/mol and the following structure:

The chemical structure of fosnetupitant chloride hydrochloride was elucidated by a combination of 1H and 13C NMR spectroscopy, infrared spectroscopy, mass spectrometry and elemental analysis. The active substance is achiral. The solid state properties of the active substance were measured by gravimetric vapour sorption and x-ray powder diffraction (XRPD). It is a white to off-white to yellowish, crystalline, hygroscopic solid. Three polymorphic forms have been identified following extensive screening, requiring isolation from different solvent mixtures. Fosnetupitant chloride hydrochloride is always isolated as form I following the commercial manufacturing process. Since it is dissolved and lyophilised during finished product manufacture, particle size and polymorphic form are not considered critical quality attributes (CQAs) of the active substance and are not included in the specification.
RX
AKYNZEO (300 mg netupitant/0.5 mg palonosetron) capsules are an oral combination product of netupitant, a substance P/neurokinin 1 (NK-1) receptor antagonist, and palonosetron hydrochloride, a serotonin-3 (5-HT3) receptor antagonist. Both netupitant and palonosetron hydrochloride are anti-nausea and anti-emetic agents.
Netupitant is chemically described: 2-[3,5-bis(trifluoromethyl)phenyl]-N, 2 dimethyl-N-[4-(2methylphenyl)-6-(4-methylpiperazin-1-yl)pyridin-3-yl] propanamide. The empirical formula is C30H32F6N4O, with a molecular weight of 578.61. Netupitant exists as a single isomer and has the following structural formula:
 |
Palonosetron hydrochloride is chemically described: (3aS)-2-[(S)-1-Azabicyclo [2.2.2]oct-3-yl]2,3,3a,4,5,6-hexahydro-1-oxo-1H-benz[de]isoquinoline hydrochloride. The empirical formula is C19H24N2O.HCl, with a molecular weight of 332.87. Palonosetron hydrochloride exists as a single isomer and has the following structural formula:
 |
Netupitant is white to off-white crystalline powder. It is freely soluble in toluene and acetone, soluble in isopropanol and ethanol, and very slightly soluble in water.
Palonosetron hydrochloride is a white to off-white crystalline powder. It is freely soluble in water, soluble in propylene glycol, and slightly soluble in ethanol and 2-propanol.
Each AKYNZEO capsule is composed of one white-caramel hard gelatin capsule which contains three tablets each containing 100 mg netupitant and one gelatin capsule containing 0.5 mg palonosetron (equivalent to 0.56 mg palonosetron hydrochloride). The inactive ingredients are butylated hydroxyanisole (BHA), croscarmellose sodium, gelatin, glycerin, magnesium stearate, microcrystalline cellulose, mono-and di-glycerides of capryl/capric acid, polyglyceryl dioleate, povidone K-30, purified water, red iron oxide, silicon dioxide, sodium stearyl fumarate, sorbitol, sucrose fatty acid esters, titanium dioxide and yellow iron oxide. It may contain traces of medium-chain triglycerides, lecithin, and denatured ethanol.
AKYNZEO (235 mg fosnetupitant/0.25 mg palonosetron) for injection is a combination product of fosnetupitant, a prodrug of netupitant, which is a substance P/neurokinin 1 (NK-1) receptor antagonist, and palonosetron hydrochloride, a serotonin-3 (5-HT3) receptor antagonist.
Fosnetupitant chloride hydrochloride is chemically described as 2-(3,5-bistrifluoromethylphenyl)-N-methyl-N-[6-(4-methyl-4-O-methylene-phosphatepiperazinium-1-yl)4-o-tolyl-pyridin-3-yl]-isobutyramide chloride hydrochloride. The empirical formula is C31H36F6N4O5P•Cl•HCl, with a molecular weight of 761.53. Fosnetupitant chloride hydrochloride exists as a single isomer and has the following structural formula:
 |
Fosnetupitant chloride hydrochloride is white to off-white to yellowish solid or powder. Its solubility is pH dependent: at acidic pH (pH 2), its solubility is 1.4 mg/mL; at basic pH (pH 10), its solubility is 11.5 mg/mL.
Palonosetron hydrochloride is described above in this section.
AKYNZEO for injection is available for intravenous infusion, and is supplied as a sterile lyophilized powder in a single-dose vial. Each vial contains 235 mg of fosnetupitant (equivalent to 260 mg fosnetupitant chloride hydrochloride) and 0.25 mg of palonosetron (equivalent to 0.28 mg of palonosetron hydrochloride). The inactive ingredients are edetate disodium (6.4 mg), mannitol (760 mg), sodium hydroxide and/or hydrochloric acid (for pH adjustment).
PATENT
WO 2013082102
https://patents.google.com/patent/WO2013082102A1/un



PATENT
US 20150011510
https://patents.google.com/patent/US20150011510A1/en
Step 1:
- [0160]
13.0 g (82.5 mMol) 6-Chloro-nicotinic acid in 65 ml THF were cooled to 0° C. and 206.3 ml (206.3 mMol) o-tolylmagnesium chloride solution (1M in THF) were added over 45 minutes. The solution obtained was further stirred 3 hours at 0° C. and overnight at room temperature. It was cooled to −60° C. and 103.8 ml (1.8 Mol) acetic acid were added, followed by 35 ml THF and 44.24 g (165 mMol) manganese(III) acetate dihydrate. After 30 minutes at −60° C. and one hour at room temperature, the reaction mixture was filtered and THF removed under reduced pressure. The residue was partitioned between water and dichloromethane and extracted. The crude product was filtered on silica gel (eluent: ethyl acetate/toluene/formic acid 20:75:5) then partitioned between 200 ml aqueous half-saturated sodium carbonate solution and 100 ml dichloromethane. The organic phase was washed with 50 ml aqueous half-saturated sodium carbonate solution. The combined aqueous phases were acidified with 25 ml aqueous HCI 25% and extracted with dichloromethane. The organic extracts were dried (Na2SO4) and concentrated under reduced pressure to yield 10.4 g (51%) of 6-chloro-4-o-tolyl-nicotinic acid as a yellow foam. MS (ISN): 246 (M−H, 100), 202 (M-CO2H, 85), 166 (36).
Step 2:
- [0161]
To a solution of 8.0 g (32.3 mMol) 6-chloro-4-o-tolyl-nicotinic acid in 48.0 ml THF were added 3.1 ml (42.0 mMol) thionylchloride and 143 .mu.l (1.8 mMol) DMF. After 2 hours at 50° C., the reaction mixture was cooled to room temperature and added to a solution of 72.5 ml aqueous ammonium hydroxide 25% and 96 ml water cooled to 0° C. After 30 minutes at 0° C., THF was removed under reduced pressure and the aqueous layer was extracted with ethyl acetate. Removal of the solvent yielded 7.8 g (98%) 6-chloro-4-o-tolyl-nicotinamide as a beige crystalline foam. MS (ISP): 247 (M+H+, 100).
Step 3:
- [0162]
1.0 g (4.05 mMol) 6-Chloro-4-o-tolyl-nicotinamide in 9.0 ml 1-methyl-piperazine was heated to 100° C. for 2 hours. The excess N-methyl-piperazine was removed under high vacuum and the residue was filtered on silica gel (eluent: dichloromethane) to yield 1.2 g (95%) 6-(4-methyl-piperazin-1-yl)-4-o-tolyl-nicotinamide as a light yellow crystalline foam. - [0163]
MS (ISP): 311 (M+H+, 100), 254 (62).
Step 4:
- [0164]
A solution of 0.2 g (0.6 mMol) 6-(4-methyl-piperazin-1-yl)-4-o-tolyl-nicotinamide in 1.0 ml methanol was added to a solution of 103 mg (2.6 mMol) sodium hydroxide in 1.47 ml (3.2 mMol) NaOCl (13%) and heated for 2 hours at 70° C. After removal of methanol, the aqueous layer was extracted with ethyl acetate. The combined organic extracts were dried (Na2SO4), concentrated under reduced pressure and the residue filtered on silica gel (eluent: dichloromethane/methanol 4:1) to yield 100 mg (70%) 6-(4-methyl-piperazin-1-yl)-4-o-tolyl-pyridin-3-ylamine as a brown resin. MS (ISP): 283 (M+H+, 100), 226 (42).
Step 5:
- [0165]
2.15 mil (11.6 mMol) Sodium methoxide in methanol were added over 30 minutes to a suspension of 0.85 g (4.6 mMol) N-bromosuccinimide in 5.0 ml dichloromethane cooled to −5° C. The reaction mixture was stirred 16 hours at −5° C. Still at this temperature, a solution of 1.0 g (3.1 mMol) 6-(4-methyl-piperazin-1-yl)-4-o-tolyl-nicotinamide in 5.0 ml methanol was added over 20 minutes and stirred for 5 hours. 7.1 ml (7.1 mMol) Aqueous HCl 1N and 20 ml dichloromethane were added. The phases were separated and the organic phase was washed with deionized water. The aqueous phases were extracted with dichloromethane, brought to pH=8 with aqueous NaOH 1N and further extracted with dichloromethane. The latter organic extracts were combined, dried (Na2SO4) and concentrated to yield 1.08 g (quant.) [6-(4-methyl-piperazin-1-yl)-4-o-tolyl-pyridin-3-yl]-carbamic acid methyl ester as a grey foam. - [0166]
MS (ISP): 341 (M+H+, 100), 284 (35).
Step 6:
- [0167]
A solution of 0.5 g (1.4 mMol) [6-(4-methyl-piperazin-1-yl)-4-o-tolyl-pyridin-3-yl]-carbamic acid methyl ester in 3.0 ml dichloromethane was added over 10 minutes to a solution of 1.98 ml (6.9 mMol) Red-Al®. (70% in toluene) and 2.5 ml toluene (exothermic, cool with a water bath to avoid temperature to go >50° C.). The reaction mixture was stirred 2 hours at 50° C. in CH2Cl2, extracted with ethyl acetate and cooled to 0° C. 4 ml Aqueous NaOH 1N were carefully (exothermic) added over 15 minutes, followed by 20 ml ethyl acetate. The phases were separated and the aqueous phase was extracted with ethyl acetate. The combined organic extracts were washed with deionized water and brine, dried (Na2SO4) and concentrated under reduced pressure to yield 0.37 g (89%) methyl-[6-(4-methyl-piperazin-1-yl)-4-o-tolyl-pyridin-3-yl]-amine as an orange resin. MS (ISP): 297 (M+H+, 100).
Synthesis of 2-(3,5-bis-Trifluoromethyl-phenyl)-2-methyl-propionyl Chloride
- [0168]
- [0169]
15.0 g (50 mmol) 2-(3,5-bis-trifluoromethyl-phenyl)-2-methyl-propionic acid were dissolved in 127.5 ml dichloromethane in the presence of 0.75 ml DMF. 8.76 ml (2 eq.) Oxalyl chloride were added and after 4.5 hours, the solution was rotary evaporated to dryness. 9 ml Toluene were added and the resulting solution was again rotary evaporated, then dried under high vacuum yielding 16.25 g (quant.) of 2-(3,5-bis-trifluoromethyl-phenyl)-2-methyl-propionyl chloride as a yellow oil of 86% purity according to HPLC analysis. NMR (250 MHz, CDCl3): 7.86 (br s, 1H); 7.77, (br s, 2H, 3Harom); 1.77 (s, 6H, 2 CH3).
Synthesis of 2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethyl-N-(6-(4-methylpiperazin-1-yl)-4-(o-tolyl)pyridin-3-yl)propanamide (Netupitant)
- [0170]
- [0171]
A solution of 20 g (67.5 mmol) methyl-[6-(4-methyl-piperazin-1-yl)-4-o-tolyl-pyridin-3-yl]-amine and 17.5 ml (101 mmol) N-ethyldiisopropylamine in 200 ml dichloromethane was cooled in an ice bath and a solution of 24 g (75 mmol)2-(3,5-bis-trifluoromethyl-phenyl)-2-methyl-propionyl chloride in 50 ml dichloromethane was added dropwise. The reaction mixture was warmed to 35-40° C. for 3 h, cooled to room temperature again and was stirred with 250 ml saturated sodium bicarbonate solution. The organic layer was separated and the aqueous phase was extracted with dichloromethane. The combined organic layers were dried (magnesium sulfate) and evaporated. The residue was purified by flash chromatography to give 31.6 g (81%) of 2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethyl-N-(6-(4-methylpiperazin-1-yl)-4-(o-tolyl)pyridin-3-yl)propanamide as white crystals. - [0172]
M.P. 155-157° C.; MS m/e (%): 579 (M+H+, 100).
Synthesis of 5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-2-(4-methylpiperazin-1-yl)-4-(o-tolyl)pyridine 1-oxide
Step 1:
- [0174]
The solution of 6-chloropyridin-3-amine (115 g, 0.898 mol) and (Boc)2O (215.4 g, 0.988 mol) in 900 mL of dioxane was refluxed overnight. The resulting solution was poured into 1500 mL of water. The resulting solid was collected, washed with water and re-crystallized from EtOAc to afford 160 g tert-butyl (6-chloropyridin-3-yl)carbamate as a white solid (Yield: 78.2%).
Step 2:
- [0175]
To the solution of tert-butyl (6-chloropyridin-3-yl)carbamate (160 g, 0.7 mol) in 1 L of anhydrous THF was added n-BuLi (600 mL, 1.5 mol) at −78° C. under N2 atmosphere. After the addition was finished, the solution was stirred at −78° C. for 30 min, and the solution of I2 (177.68 g, 0.7 mol) in 800 mL of anhydrous THF was added. Then the solution was stirred at −78° C. for 4 hrs. TLC indicated the reaction was over. Water was added for quench, and EtOAc was added to extract twice. The combined organic phases were washed with brine, dried over Na2SO4, filtered and purified by flash chromatography to afford 80 g of tert-butyl (6-chloro-4-iodopyridin-3-yl)carbamate as a yellow solid (32.3%).
Step 3:
- [0176]
To the solution of tert-butyl (6-chloro-4-iodopyridin-3-yl)carbamate (61 g, 0.172 mol) in 300 mL of anhydrous THF was added 60% NaH (7.6 g, 0.189 mol) at 0° C. under N2 atmosphere. After the addition was finished, the solution was stirred for 30 min, and then the solution of MeI (26.92 g, 0.189 mol) in 100 mL of dry THF was added. Then the solution was stirred at 0° C. for 3 hrs. TLC indicated the reaction was over. Water was added for quench, and EtOAc was added to extract twice. The combined organic phases were washed with brine, dried over Na2SO4, filtered and concentrated to afford 63 g of crude tert-butyl (6-chloro-4-iodopyridin-3-yl)(methyl)carbamate used into the following de-protection without the further purification.
Step 4:
- [0177]
To the solution of tert-butyl (6-chloro-4-iodopyridin-3-yl)(methyl)carbamate (62.5 g, 0.172 mol) in 500 mL of anhydrous DCM was added 180 mL of TFA. Then the solution was stirred at room temperature for 4 hrs. Concentrated to remove the solvent, and purified by flash chromatography to afford 45.1 g 6-chloro-4-iodo-N-methylpyridin-3-amine as a yellow solid (Yield: 97.3%).
Step 5:
- [0178]
To the solution of 6-chloro-4-iodo-N-methylpyridin-3-amine (40.3 g, 0.15 mol) and 2-methylbenzene boric acid (24.5 g, 0.18 mol) in 600 mL of anhydrous toluene was added 400 mL of 2 N aq. Na2CO3 solution, Pd(OAc)2 (3.36 g, 15 mmol) and PPh3 (7.87 g, 0.03 mmol). The solution was stirred at 100° C. for 2 hrs. Cooled to room temperature, and diluted with water. EtOAc was added to extract twice. The combined organic phases were washed with water and brine consecutively, dried over Na2SO4, concentrated and purified by flash chromatography to afford 19 g 6-chloro-N-methyl-4-(o-tolyl)pyridin-3-amine as a white solid (Yield: 54.6%).
Step 6:
- [0179]
To the solution of 6-chloro-N-methyl-4-(o-tolyl)pyridin-3-amine (18.87 g, 81.3 mmol) and DMAP (29.8 g, 243.9 mmol) in 200 mL of anhydrous toluene was added the solution of 2-(3,5-bis-trifluoromethyl-phenyl)-2-methyl-propionyl chloride (28.5 g, 89.4 mmol) in toluene under N2 atmosphere. The solution was heated at 120° C. for 23 hrs. Cooled to room temperature, poured into 1 L of 5% aq. NaHCO3 solution, and extracted with EtOAc twice. The combined organic phases were washed by water and brine consecutively, dried over Na2SO4, filtered and purified by flash chromatography to afford 35 g 2-(3,5-bis(trifluoromethyl)phenyl)-N-(6-chloro-4-(o-tolyl)pyridin-3-yl)-N,2-dimethylpropanamide as a white solid (Yield: 83.9%).
Step 7:
- [0180]
To the solution of 2-(3,5-bis(trifluoromethyl)phenyl)-N-(6-chloro-4-(o-tolyl)pyridin-3-yl)-N,2-dimethylpropanamide (5.14 g, 10 mmol) in 60 mL of DCM was added m-CPBA (6.92 g, 40 mmol) at 0° C. under N2 atmosphere. Then the solution was stirred overnight at room temperature. 1 N aq. NaOH solution was added to wash twice for removing the excess m-CPBA and a side product. The organic phase was washed by brine, dried over Na2SO4, filtered and concentrated to afford 5.11 g of crude 5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-2-chloro-4-(o-tolyl)pyridine 1-oxide as a white solid (Yield: 96.4%).
Step 8:
- [0181]
To the solution of crude 5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-2-chloro-4-(o-tolyl)pyridine 1-oxide (5.1 g, 9.62 mmol) in 80 mL of n-BuOH was added N-methylpiperazine (7.41 g, 74.1 mmol) under N2 atmosphere. Then the solution was stirred at 80° C. overnight. Concentrated and purified by flash chromatography to afford 4.98 g 5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-2-(4-methylpiperazin-1-yl)-4-(o-tolyl)pyridine 1-oxide as a white solid (Yield: 87.2%). 1HNMR (CDCl3, 400 MHz) δ 8.15 (s, 1H), 7.93 (s, 1H), 7.78 (s, 2H), 7.38 (m, 2H), 7.28 (m, 1H), 7.17 (m, 1H), 7.07 (s, 1H), 5.50 (s, 3H), 2.72 (d, J=4.4 Hz, 4H), 2.57 (m, 3H), 2.40 (s, 3H), 2.23 (s, 3H), 1.45˜1.20 (m, 6H).
Synthesis of 4-(5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-1-oxido-4-(o-tolyl)pyridin-2-yl)-1-methylpiperazine 1-oxide
- [0182]
- [0183]
To a solution of 5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-2-(4-methylpiperazin-1-yl)-4-(o-tolyl)pyridine 1-oxide (3 g, 5.05 mmol) and NaHCO3 (0.354 g, 12.66 mmol) in 60 mL of MeOH and 15 mL of H2O were added potassium monopersulfate triple salt (1.62 g, 26.25 mmol) at room temperature during 15 min. After stirring for 4 hrs at room temperature under N2 atmosphere, the reaction mixture was concentrated in vacuo and purified by flash chromatography (eluent: MeOH). The product was dissolved into DCM, the formed solid was filtered off, and the solution was concentrated under reduced pressure to afford 1.77 g 4-(5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-1-oxido-4-(o-tolyl)pyridin-2-yl)-1-methylpiperazine 1-oxide as a white solid (Yield: 57.4%). 1HNMR (CDCl3, 400 MHz) δ 8.06 (s, 1H), 7.78 (s, 1H), 7.60 (s, 2H), 7.37˜7.20 (m, 4H), 6.81 (s, 1H), 3.89 (s, 21H), 3.74 (m, 4H), 3.31 (m, 5H), 2.48 (s, 3H), 2.18 (s, 3H), 1.36 (s, 6H).
Synthesis of 1-(5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-4-(o-tolyl)pyridin-2-yl)-4-methylpiperazine 1,4-dioxide
- [0184]
- [0185]
To the solution of 2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethyl-N-(6-(4-methylpiperazin-1-yl)-4-(o-tolyl)pyridin-3-yl)propanamide (11.1 g, 19.2 mmol) in 75 ml of Methanol was added sodium bicarbonate (3.38 g, 40.3 mmol) dissolved in 20 ml of water. Then Oxone (14.75 g, 48.0 mmol) was added to the stirred solution at room temperature in 3-4 portions. The suspension was heated for 4 h at 50° C. After filtration of the salts (washed with 3×8 ml of methanol), the solvent has been evaporated under reduced pressure and substituted by DCM (30 ml). The organic phase was washed with water (5×30 ml), dried over Na2SO4, filtered, concentrated and purified by precipitation in toluene to afford 9.3 g 1-(5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-4-(o-tolyl)pyridin-2-yl)-4-methylpiperazine 1,4-dioxide as a white solid (Yield: 80%). 1H-NMR (CDCl3, 400 MHz, at 333K) δ 8.27 (s, 2H), 7.75 (s, 1H), 7.63 (s, 2H), 7.26˜7.19 (m, 2H), 7.14 (t, 1H, J=7.4 Hz), 7.09 (d, 1H, J=7.4 Hz), 4.93 (t, 2H, J=11.6 Hz), 4.70 (t, 2H, J=11.6 Hz), 4.12 (d, 2H, J=10.7 Hz), 3.84 (s, 3H), 3.50 (d, 2H, J=10.3 Hz), 2.47 (s, 3H), 2.12 (s, 3H), 1.40 (s, 6H).
Synthesis (A) of di-tert-butyl (chloromethyl)phosphate
- [0186]
- [0187]
Di-tert-butyl phosphohite (40.36 mmole) was combined with potassium bicarbonate (24.22 mmole) in 35 ml of water. The solution was stirred in an ice bath and potassium permanganate (28.25 mmole) was added in three equal portions over one hour’s time. The reaction as then allowed to continue at room temperature for an additional half hour. - [0188]
Decolorizing carbon (600 mg) was then incorporated as the reaction was heated to 60° C. for 15 minutes. The reaction was then vacuum filtered to remove solid magnesium dioxide. The solid was washed several times with water. The filtrate was then combined with one gram of decolorizing carbon and heated at 60° C. for an additional twenty minutes. The solution was again filtered to yield a colorless solution, which was then evaporated under vacuum to afford crude Di-tert-butyl phosphate potassium salt. Di-tert-butyl phosphate potassium salt (5 g, 20.14 mmole) was dissolved in methanol (15 g): to this solution at 0° C. a slight excess of concentrated HCl is slowly added with efficient stirring at 0° C. The addition of acid causes the precipitation of potassium chloride. The solid is then filtered and washed with methanol. The compound in the mother liquor is then converted to the ammonium form by adding an equal molar amount of tetramethylammonium hydroxide (3.65 g, 20.14 mmole) while keeping the reaction cooled by a salt/ice bath with efficient stirring. The resulting clear solution is placed under reduced pressure to give the crude product. To the tetramethylammonium di-tert-butyl-phosphate dissolved in refluxing dimethoxyethane is then added 4.3 grams of chloroiodomethane (24.16 mmole) and stirred for 1-2 hours. The reaction is then filtered and the filtrate is placed under reduced pressure to concentrate the solution in DME. The chloromethyl di-tert-butyl phosphate 12-16% in DME is used in the synthesis of 4-(5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-4-(o-tolyl)pyridin-2-yl)-1-methyl-1-((phosphonooxy)methyl)piperazin-1-ium without further purifications (60% yield): 1HNMR (CD3OD, 300 MHz) δ 1.51 (s, 12H), 5.63 (d, 2H, J=14.8). 31P-NMR (CD3OD, 300 MHz) δ −11.3 (s, 1P).
Synthesis (B) of di-tert-butyl (chloromethyl)phosphate
- [0189]
- [0190]
Di-tert-butyl phosphate potassium salt (5 g, 20.14 mmole) is dissolved in methanol (15 g): to this solution at 0° C. a slight excess of concentrated HCl is slowly added with efficient stirring at 0° C. The addition of acid causes the precipitation of potassium chloride. The solid is then filtered and washed with methanol. The compound in the mother liquor is then converted to the ammonium form by adding an equal molar amount of tetrabuthylammonium hydroxide 1 M in methanol (20.14 mmole) while keeping the reaction cooled at 0° C. with efficient stirring. The resulting clear solution is placed under reduced pressure to give the intermediate product. The tetrabuthylammonium di-tert-butyl-phosphate dissolved in acetone is then added dropwise to 53.3 grams of chloroiodomethane (302.1 mmole) and stirred at 40° C. for 1-2 hours. The solvent and excess of chloroiodomethane are distilled off, the reaction mass suspended in TBME and then filtered. The filtrate is washed by a saturated solution of sodium bicarbonate and water and then placed under reduced pressure to substitute the solvent by acetone, i.e., to remove the solvent after which it is replaced with acetone. The chloromethyl di-tert-butyl phosphate 7-20% in acetone is used in the next step without further purifications (70-80% yield): 1H-NMR (CD3OD, 300 MHz) δ 1.51 (s, 12H), 5.63 (d, 2H, J=14.8). 31P-NMR (CD3OD, 300 MHz) δ −11.3 (s, 1P).
Stability studies of 4-(5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-4-(o-tolyl)pyridin-2-yl)-1-methyl-1-((phosphonooxy)methyl)piperazin-1-ium salts
- [0191]
In order to further improve the stability and solubility of 4-(5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-4-(o-tolyl)pyridin-2-yl)-1-methyl-1-((phosphonooxy)methyl)piperazin-1-ium, a variety of its derivative salts were synthesized and tested. Their synthesis employed either a) neutralization of the dried diacid phosphate species and its corresponding base salts or b) a direct acid deprotection starting from the dried di(tert-butyl)-protected phosphate species. Neutralization was performed with L-histidine, magnesium salt, N-methyl-D-glucamine (dimeglumine), and L-lysine. Both procedures were tried in the synthesis of citric derivatives whereas with other acids the direct deprotection reaction was used. The figures below show the most relevant structures. - [0192]
When the parent acid species was not stored in dry condition it was found to undergo over 8% degradation in the first week and over 65% degradation in the first six months. When the dried parent acid species was held at 30° C. in air it underwent 0.05% degradation in the first 7 days and at total of 7.03% degradation in six months. When the dried parent acid species was held under argon at room temperature it underwent up to 0.13% degradation in the first 7 days but then was essentially stable for six months. Results for various derivative salts are shown in Table 1 below. - TABLE 1 Representative Degradation Results for Salts Purity A % Solvents Additives Yield % HPLC Comments MeOH L-Histidine, 2 eq. 26.6% 95.94% Degradation: +0.70% in 6 days (in air) +0.46% in 6 days (in argon) MeOH Mg(OH)2, 2 eq. 48.6% 94.11% Degradation: +0.81% in 6 days (in air) +0.29% in 6 days (in argon) MeOH + Citric acid, 2 eq. N.A. 94.40% From protected species. DCM, 1:1 MeOH 1. HCl dioxane, 4 eq. >90% 94.50% From protected species. 2. Ca(OH)2 MeOH H3PO4, 85%, 2 eq. >90% 98.81% From protected species and retains 0.39% of that species. MeOH HBr, 48%, 4 eq. 84.6% 96.11% From protected species. Product degrades rapidly, MeOH + CH3SO3H N.A. 61.54% From protected species. DCM, Product NOT stable: contains 1:4 32.45% decomposition species. MeOH NaH2PO4, 4 eq. N.A. n.d. Only 1.27 of parent species formed. Poor reaction. MeOH N-methyl-D- N.A. 96.88% Degradation: glucamine +0.87% in 6 days (in air) (Meglumine), 2 eq. +1.52% in 11 days (in argon) MeOH N-methyl-D- >99% 97.42% Degradation: glucamine +0.77% in 6 days (in air) (Meglumine), 1 eq. +0.83% in 7 days (in argon) MeOH+ 1. NaOH, 3 eq 96.5% 97.49% Degradation: DCM, 2. Citric acid, 1 eq. +0.09% in 2 days (in argon) 5:2 +0.59% in 89 days (in argon) MeOH+ 1. NaOH, 3 eq. 93.8% 97.46% Degradation: DCM, 2. Fumaric acid, 1 eq. +1.95% in 14 days (in air) 5:2 +1.80% in 12 days (in argon) MeOH L-lysine, 1 eq. >99% 97.62% Degradation: +0.69% in 14 days (in air) +0.48% in 12 days (in argon)
- [0194]
- [0195]
The solution of chloromethyl di-tert-butyl phosphate in DME (250 g from a 10% solution, 96.64 mmole) was evaporated under reduced pressure until the formation of pale yellow oil, dissolved then at 50° C. with 318 ml of Acetonitrile. 17.2 g (80.54 mmole) of 1,8-bis(dimethylamino)naphtalene and 46.6 g (80.54 mmole) of 2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethyl-N-(6-(4-methylpiperazin-1-yl)-4-(o-tolyl)pyridin-3-yl)propanamide were added and the solution heated at 90° C. for at least 12 h. After the addition of 75 g of isopropylether, the precipitated crude product was cooled at room temperature, filtered and washed with acetonitrile, isopropylether/acetone, 3:1 and isopropylether, and dried under reduced pressure to afford 20-33 g of the 4-(5-{2-[3,5-bis(trifluoromethyl)phenyl]-N,2-dimethylpropanamido}-4-(o-tolyl)pyridin-2-yl)-1-methyl-1-{[(tert-butoxy)phosphoryl]oxymethyl}piperazin-1-ium as white solid (Yield: 30-50%). 1H-NMR (CD3OD, 400 MHz) δ 7.98 (s, 1H), 7.86 (s, 1H), 7.76 (s, 2H), 7.33-7.10 (m, 4H), 6.80 (s, 1H), 5.03 (d, 2H, JPH=8.5 Hz), 4.52 (s, 2H), 4.13 (m, 2H), 3.83 (m, 2H), 3.69 (m, 2H), 3.52 (m. 2H), 3.23 (s, 3H), 2.53 (s, 3H), 2.18 (s, 3H), 1.46 (s, 18H), 1.39 (s, 6H). 31P-NMR (CD3OD, 161 MHz) δ −5.01 (s, 1P). To 20 g (23.89 mmole) of the 4-(5-{2-[3,5-bis(trifluoromethyl)phenyl]-N,2-dimethylpropanamido}-4-(o-tolyl)pyridin-2-yl)-1-methyl-1-{[(tert-butoxy)phosphoryl]oxymethyl}piperazin-1-ium dissolved in 180 g of methanol and 400 g of dichloromethane was added HCl 4M in dioxane (18.8 g, 71.66 mmole) and the solution was heated for 3 h at reflux. After the addition of 200 g of dioxane, DCM and methanol were distilled under reduced pressure until precipitation of the product, which was filtered and washed with isopropylether (100 g), acetone (30 g) and pentane (2×60 g). The product was finally dried under reduced pressure at 55° C. to afford 15-17 g of 4-(5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-4-(o-tolyl)pyridin-2-yl)-1-methyl-1-((phosphonooxy)methyl)piperazin-1-ium chloride hydrochloride as white solid (Yield: 88-93%). 1H-NMR (CD3OD, 400 MHz) δ 7.02 (s, 1H), 7.87 (s, 1H), 7.74 (s, 2H), 7.33-7.40 (m, 2H), 7.27 (m, 1H), 7.21 (s, 1H), 7.16 (d, 1H, J=8.2 Hz), 5.27 (d, 2H, JPH=7.9 Hz), 4.29 (m, 2H), 4.05 (m, 2H), 3.85 (m, 2H), 3.74 (m, 2H), 3.35 (s, 3H), 2.62 (s, 3H), 2.23 (s, 3H), 1.38 (s, 6H). 31P-NMR (CD3OD, 161 MHz) δ −2.81 (t, 1P, JPH=7.9 Hz).
Synthesis (B) of 4-(5-(2-(3,5-bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-4-(o-tolyl)pyridin-2-yl)-1-methyl-1-((phosphonooxy)methyl)piperazin-1-ium chloride hydrochloride
- [0196]
- [0197]
To the solution of chloromethyl di-tert-butyl phosphate in Acetone (22.1 g from a 10% solution, 85.58 mmole), 15.5 g (103.24 mmole) of sodium iodide and 33.0 g (57.00 mmole) of netupitant were added and the solution heated at 50° C. for at 6-16 h. The precipitated salts were filtered off, the acetone distilled under reduced pressure and the crude product dissolved in 43.0 g of methanol and 43.0 g 1,4-dioxane. 12.6 g of HCl 4M in dioxane (113.85 mmole) were added, and then methanol is distilled off at 40° C. under reduced pressure. The solution is cooled at 5° C. and stirred at 5° C. for at least 2 h at 5° C. The product was isolated by filtration, purified by additional slurry in acetone (238 g), and filtered and washed with acetone (47 g) and pentane (2×72 g). - [0198]
The product was finally dried under reduced pressure at 60° C. to afford 22-30 g of white-yellowish solid (Yield: 50-70%) - [0199]
1H-NMR (CD3OD, 400 MHz) δ 7.02 (s, 1H), 7.87 (s, 1H), 7.74 (s, 2H), 7.33-7.40 (m, 2H), 7.27 (m, 1H), 7.21 (s, 1H), 7.16 (d, 1H, J=8.2 Hz), 5.27 (d, 2H, JPH=7.9 Hz), 4.29 (m, 2H), 4.05 (m, 2H), 3.85 (m, 2H), 3.74 (m, 2H), 3.35 (s, 3H), 2.62 (s, 3H), 2.23 (s, 3H), 1.38 (s, 6H). 31P-NMR (CD3OD, 161 MHz) δ −2.81 (t, IP, JPH=7.9 Hz).
PATENT
US 8,426,450
PATENT
US 9,403,772
SYN
https://chemistry-europe.onlinelibrary.wiley.com/doi/10.1002/chem.201901840
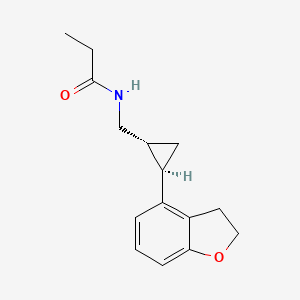
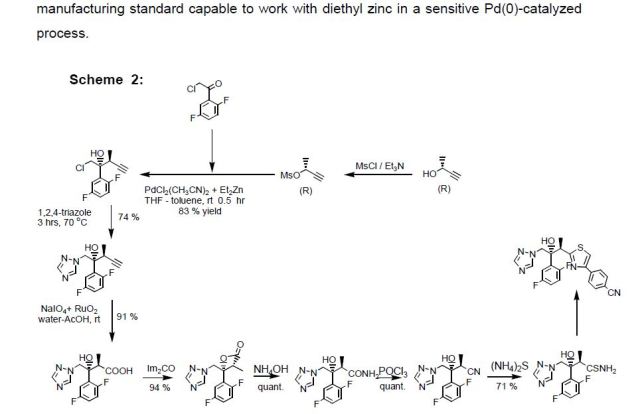
The synthesis of fosnetupitant (195) was developed by the Swiss company Helsinn (Scheme 34).[58] The synthesis started with the reaction of 6-chloronicotinic acid (196) with o-tolylmagnesium chloride followed by manganese(III) acetate to give acid derivative 197. This was converted to amide 198 after reaction with thionyl chloride and ammonium hydroxide. Next, reaction with N-methylpiperazine furnished intermediate 199, which was then transformed into carbamate 200 after reaction with NBS in methanol. Reduction with Red-Al followed by acylation with acyl chloride 202 afforded netupitant (203).
Finally, reaction with di-tert-butyl chloromethyl phosphate followed by the removal of the tert-butyl groups by treatment with HCl in dioxane afforded fosnetupitant (195).
L. Fadini, P. Manini, C. Pietra, C. Giuliano, E. Lovati, R. Cannella, S. Venturini, V. J. Stella, WO 082102 A1, 2013.


SYN
Fosnetupitant chloride HCl

PATENT
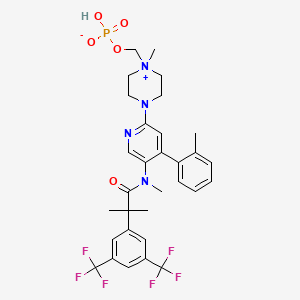
Fosnetupitant is a neurokynin-1 (“NK-1”) antagonist under development by Helsinn Healthcare SA, Lugano/Pazzallo Switzerland, for the treatment of chemotherapy induced nausea and vomiting. The compound is known chemically as 4-(5-(2-(3,5- bis(trifluoromethyl)phenyl)-N,2-dimethylpropanamido)-4-(o-tolyl)pyridin-2-yl)-l-methyl- 1 -((phosphonooxy)methyl)piperazin- 1 -ium, and has the following chemical structure in its acidic/free base form:

[004] The chloride monohydrochloride salt, and a method for its preparation, is described in WO 2013/082102. The chemical structure for this salt is reported as follows:

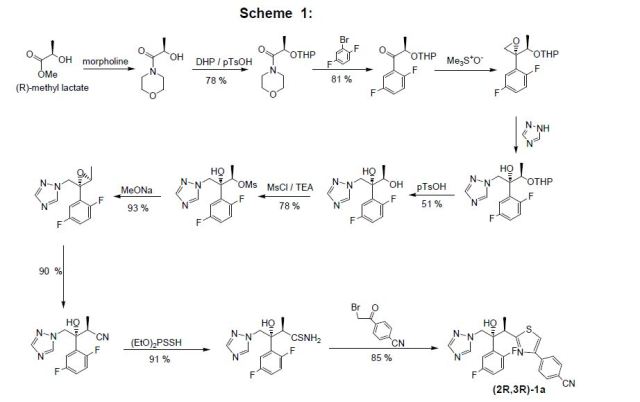
[005] The molecule can be challenging to manufacture, particularly in a highly pure crystalline form in a commercially acceptable yield. Solvents used in the manufacture of the product pose special challenges. Prior art processes have removed these solvents via evaporative techniques, which can degrade the fosnetupitant due to the excessive heat.
EXAMPLES
[089] In all the examples reported, unless otherwise reported, the starting compound was Form I of the chloride hydrochloride salt of 4-(5-(2-(3,5-bis(trifluoromethyl)phenyl)- N,2-dimethylpropanamido)-4-(o-tolyl)pyridin-2-yl)-l-methyl-l – ((phosphonooxy)methyl)piperazin-l-ium, produced substantially according to the methods described in WO 2013/082102.
EXAMPLE 1 : CHARACTERIZATION OF FOSNETUPITANT
1 . Experimental Methods
1.1 Solubility
[090] The solubility of the starting compound was determined in 25 pharmaceutically acceptable solvents (class II and III) of differing polarity. The procedure was as follows:
[091] Approximately 20 mg of material was weighed out into each glass vial.
[092] 5 volume aliquots of each solvent were added separately with stirring (i.e. 1 volume = 20 μΐ; hence, 5 volume = 100 μΐ (5 x 20 μΐ)).
[093] The mixture was stirred at RT for 5- 10 minutes. Visual checks were then made for solubility.
[094] If no solubility was achieved then steps (ii) and (iii) were repeated until either the solubility was achieved or the 50 volume aliquots of that solvent were added.
[095] Solubility was then approximated.
[096] Solubility was finally checked at the elevated temperature (40°C).
1.2 Polymorph Screen (including slurry studies)
[097] Using the information from the solubility study, the compound was slurried in the solvents outlined in Table I and two more mixtures of water/ MeOH (10:90) and water/ Acetone (1 :20) respectively with temperature cycling between 40°C and RT (4 hour periods at each temperature) over 48 hours. After the slurries the resulting solids were isolated and analyzed by Raman and XRPD (where enough material was available) for any change in physical form.
[098] The compound was also dissolved in the listed solvents and two more mixtures of water/organic solvent to yield saturated solutions, and crystallization was induced by: crash cooling (at ca. -1 8°C); evaporation (at RT); and addition of an anti-solvent. Solid materials generated were then isolated and examined by Raman and XRPD (where enough material was available).
1.3 Scale-up of any new polymorphic forms
[099] Any new potential polymorphic forms of the Form I fosnetupitant were then scaled-up to ~500mg level for further characterizations by PLM, SEM, DSC, TGA, GVS (XRPD post GVS) and NMR. Further studies of conversion between each polymorphic form were also performed. From this information, an understanding of the polymorphic space was achieved.
Synthetic Reference
Fadini, Luca; Manini, Peter; Pietra, Claudio; Giuliano, Claudio; Lovati, Emanuela; Cannella, Roberta; Venturini, Alessio; Stella, Valentino. (Assignee: Helsinn Healthcare SA, Switz). Substituted 4 – phenyl – pyridines for the treatment of nk-1 receptor related diseases. WO2013082102 (2013).
//////////Fosnetupitant, 07-PNET, Фоснетупитант , فوسنيتوبيتانت , 磷奈匹坦 , FDA 2014, EMA 2015
Tasimelteon, タシメルテオン

Tasimelteon
N-([(1R,2R)-2-(2,3-Dihydro-1-benzofuran-4-yl)cyclopropyl]methyl)propanamide,
609799-22-6 cas, BMS-214778; VEC-162, ATC:N05CH03
- Use:Treatment of sleep disorder; Melatonin receptor agonist
- (1R,2R)-N-[2-(2,3-dihydrobenzofuran-4-yl)cyclopropylmethyl]propanamide
- Formula:C15H19NO2, MW:245.3 g/mol
-
Hetlioz Vanda Pharmaceuticals, 2014
Approved fda 2014
EMA
Tasimelteon is a white to off-white crystalline powder, it is non hygroscopic, soluble in water across relevant pH values and freely soluble in alcohols, cyclohexane, and acetonitrile. Conducted in vivo studies demonstrate that tasimelteon is highly permeable substance. Photostability testing and testing on stress conditions demonstrated that the active substance degrades in light.
Tasimelteon exhibits stereoisomerism due to the presence of two chiral centres. Active substance is manufactured as a single, trans-1R,2R isomer. Enantiomeric purity is controlled routinely during manufacture of active substance intermediates by chiral HPLC/specific optical rotation and additionally controlled in the active substance. Stability data indicates tasimelteon is isomerically stable.
Polymorphism has been observed in polymorphic screening studies for tasimelteon and two forms have been identified. The thermodynamically more stable form has been chosen for development and the manufacturing process consistently yields active substance of single, desired polymorphic form. It was demonstrated that milling of the active substance does not affect polymorphic form. Polymorphism is additionally controlled in active substance release and shelf-life specifications using X-ray powder diffraction analysis.
Tasimelteon is synthesized in nine main steps using linear synthesis and using commercially available well-defined starting materials with acceptable specifications. Three intermediates are isolated for control of active substance quality including stereochemical control. The active substance is isolated by slow recrystallisation or precipitation of tasimelteon from an ethanol/water mixture which ensures the formation of desired polymorphic form. Up to two additional, optional recrystallisations may be performed for unmilled tasimelteon to ensure that milled tasimelteon active substance is of high purity. Seed crystals complying with active substance specifications can be used optionally. Active substance is jet milled (micronised) to reduce and control particle size, which is critical in finished product performance with regards to content uniformity and dissolution…….http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Public_assessment_report/human/003870/WC500190309.pdf
launched in 2014 in the U.S. by Vanda Pharmaceuticals for the treatment of non-24-hour sleep-wake disorder in totally blind subjects. In 2015, the European Committee for Medicinal Products of the European Medicines Agency granted approval for the same indication. In 2010 and 2011, orphan drug designations were assigned for the treatment of non-24 hour sleep/wake disorder in blind individuals without light perception in the U.S. and the E.U., respectively.
Tasimelteon (trade name Hetlioz) is a drug approved by the U.S. Food and Drug Administration (FDA)[2] in January 2014 for the treatment of non-24-hour sleep–wake disorder (also called Non-24, N24 and N24HSWD).[3] In June 2014, the European Medicines Agency accepted an EU filing application for tasimelteon[4] and in July 2015, the drug was approved in Europe for the treatment of non-24-hour sleep-wake rhythm disorder in totally blind adults,[5] but not in the rarer case of non-24 in sighted people.
Tasimelteon is a selective agonist for the melatonin receptors MT1 and MT2, similar to other members of the melatonin receptor agonistclass of which ramelteon (2005) and agomelatine (2009) were the first approved.[6] As a treatment for N24HSWD, as with melatonin or other melatonin derivatives, the patient may experience improved sleep timing while taking the drug. Reversion to baseline sleep performance occurs within a month of discontinuation.[7]

Development
Tasimelteon (previously known as BMS-214,778) was developed for the treatment of insomnia and other sleep disorders. A phase II trial on circadian rhythm sleep disorders was concluded in March 2005.[8] A phase III insomnia trial was conducted in 2006.[9] A second phase III trial on insomnia, this time concerning primary insomnia, was completed in June 2008.[10] In 2010, the FDA granted orphan drug status to tasimelteon, then regarded as an investigational medication, for use in totally blind adults with N24HSWD.[11] (Through mechanisms such as easing the approval process and extending exclusivity periods, orphan drug status encourages development of drugs for rare conditions that otherwise might lack sufficient commercial incentive.)
On completion of Phase III trials, interpretations of the clinical trials by the research team concluded that the drug may have therapeutic potential for transient insomnia in circadian rhythm sleep disorders.[12] A year-long (2011–2012) study at Harvard tested the use of tasimelteon in blind subjects with non-24-hour sleep-wake disorder. The drug has not been tested in children nor in any non-blind people.
FDA approval
In May 2013 Vanda Pharmaceuticals submitted a New Drug Application to the Food and Drug Administration for tasimelteon for the treatment of non-24-hour sleep–wake disorder in totally blind people. It was approved by the FDA on January 31, 2014 under the brand name Hetlioz.[3] In the opinion of Public Citizen, an advocacy group, the FDA erroneously allowed it to be labelled without stating that it is only approved for use by totally blind people.[13] However, FDA updated its press release on Oct. 2, 2014 to clarify the approved use of Hetlioz, which includes both sighted and blind individuals. The update did not change the drug labeling (prescribing information).[14]
Toxicity
Experiments with rodents revealed fertility impairments, an increase in certain cancers, and serious adverse events during pregnancy at dosages in excess of what is considered the “human dose”.[15][16]
As expected, advisors to the US Food and Drug Administration have recommended approval of Vanda Pharmaceuticals’ tasimelteon, to be sold as Hetlioz, for the treatment of non-24-hour disorder in the totally blind.http://www.pharmatimes.com/Article/13-11-14/FDA_panel_backs_Vanda_body_clock_drug_for_blind.aspx
The master body clock controls the timing of many aspects of physiology, behavior and metabolism that show daily rhythms, including the sleep-wake cycles, body temperature, alertness and performance, metabolic rhythms and certain hormones which exhibit circadian variation. Outputs from the
suprachiasmatic nucleus (SCN) control many endocrine rhythms including those of melatonin secretion by the pineal gland as well as the control of Cortisol secretion via effects on the hypothalamus, the pituitary and the adrenal glands. This master body clock, located in the SCN, spontaneously generates rhythms of approximately 24.5 hours. These non-24-hour rhythms are synchronized each day to the 24-hour day-night cycle by light, the primary environmental time cue which is detected by specialized cells in the retina and transmitted to the SCN via the retino-hypothalamic tract. Inability to detect this light signal, as occurs in most totally blind individuals, leads to the inability of the master body clock to be reset daily and maintain entrainment to a 24-hour day.
Non-24-Hour Disorder, Non-24, also referred to as Non-24-Hour Sleep-Wake Disorder, (N24HSWD) or Non-24-Hour Disorder, is an orphan indication affecting approximately 65,000 to 95,000 people in the U.S. and 140,000 in Europe. Non- 24 occurs when individuals, primarily blind with no light perception, are unable to synchronize their endogenous circadian pacemaker to the 24-hour light/dark cycle. Without light as a synchronizer, and because the period of the internal clock is typically a little longer than 24 hours, individuals with Non-24 experience their circadian drive to initiate sleep drifting later and later each day. Individuals with Non-24 have abnormal night sleep patterns, accompanied by difficulty staying awake during the day. Non-24 leads to significant impairment, with chronic effects impacting the social and occupational functioning of these individuals.
In addition to problems sleeping at the desired time, individuals with Non-24 experience excessive daytime sleepiness that often results in daytime napping.
The severity of nighttime sleep complaints and/or daytime sleepiness complaints varies depending on where in the cycle the individual’s body clock is with respect to their social, work, or sleep schedule. The “free running” of the clock results in approximately a 1-4 month repeating cycle, the circadian cycle, where the circadian drive to initiate sleep continually shifts a little each day (about 15 minutes on average) until the cycle repeats itself. Initially, when the circadian cycle becomes desynchronous with the 24h day-night cycle, individuals with Non-24 have difficulty initiating sleep. As time progresses, the internal circadian rhythms of these individuals becomes 180 degrees out of synchrony with the 24h day-night cycle, which gradually makes sleeping at night virtually impossible, and leads to extreme sleepiness during daytime hours.
Eventually, the individual’s sleep-wake cycle becomes aligned with the night, and “free-running” individuals are able to sleep well during a conventional or socially acceptable time. However, the alignment between the internal circadian rhythm and the 24-hour day-night cycle is only temporary.
In addition to cyclical nighttime sleep and daytime sleepiness problems, this condition can cause deleterious daily shifts in body temperature and hormone secretion, may cause metabolic disruption and is sometimes associated with depressive symptoms and mood disorders.
It is estimated that 50-75% of totally blind people in the United States (approximately 65,000 to 95,000) have Non-24. This condition can also affect sighted people. However, cases are rarely reported in this population, and the true rate of Non-24 in the general population is not known.
The ultimate treatment goal for individuals with Non-24 is to entrain or synchronize their circadian rhythms into an appropriate phase relationship with the 24-hour day so that they will have increased sleepiness during the night and increased wakefulness during the daytime. Tasimelteon
Tasimelteon is a circadian regulator which binds specifically to two high affinity melatonin receptors, Mella (MT1R) and Mellb (MT2R). These receptors are found in high density in the suprachiasmatic nucleus of the brain (SCN), which is responsible for synchronizing our sleep/wake cycle. Tasimelteon has been shown to improve sleep parameters in prior clinical studies, which simulated a desynchronization of the circadian clock. Tasimelteon has so far been studied in hundreds of individuals and has shown a good tolerability profile.
Tasimelteon has the chemical name: tr ns-N-[[2-(2,3-dihydrobenzofuran- 4-yl)cycloprop-lyl] methyl] propanamide, has the structure of Formula I:
Formula I
and is disclosed in US 5856529 and in US 20090105333, both of which are incorporated herein by reference as though fully set forth.
Tasimelteon is a white to off-white powder with a melting point of about 78°C (DSC) and is very soluble or freely soluble in 95% ethanol, methanol, acetonitrile, ethyl acetate, isopropanol, polyethylene glycols (PEG-300 and PEG- 400), and only slightly soluble in water. The native pH of a saturated solution of tasimelteon in water is 8.5 and its aqueous solubility is practically unaffected by pH. Tasimelteon has 2-4 times greater affinity for MT2R relative to MTIR. It’s affinity (¾) for MTIR is 0.3 to 0.4 and for MT2R, 0.1 to 0.2. Tasimelteon is useful in the practice of this invention because it is a melatonin agonist that has been demonstrated, among other activities, to entrain patients suffering from Non-24.
Metabolites of tasimelteon include, for example, those described in “Preclinical Pharmacokinetics and Metabolism of BMS-214778, a Novel
Melatonin Receptor Agonist” by Vachharajani et al., J. Pharmaceutical Sci., 92(4):760-772, which is hereby incorporated herein by reference. The active metabolites of tasimelteon can also be used in the method of this invention, as can pharmaceutically acceptable salts of tasimelteon or of its active metabolites. For example, in addition to metabolites of Formula II and III, above, metabolites of tasimelteon also include the monohydroxylated analogs M13 of Formula IV, M12 of Formula V, and M14 of Formula VI.
Formula IV
Formula V
MO
Formula VI
Thus, it is apparent that this invention contemplates entrainment of patients suffering free running circadian rhythm to a 24 hour circadian rhythm by administration of a circadian rhythm regulator (i.e., circadian rhythm modifier) capable of phase advancing and/or entraining circadian rhythms, such as a melatonin agonist like tasimelteon or an active metabolite oftasimelteon or a pharmaceutically acceptable salt thereof. Other MT1R and MT2R agonists, i.e., melatonin agonists, can have similar effects on the master body clock. So, for example, this invention further contemplates the use of melatonin agonists such as but not limited to melatonin, N-[l-(2,3-dihydrobenzofuran-4- yl)pyrrolidin-3-yl]-N-ethylurea and structurally related compounds as disclosed in US 6,211,225, LY-156735 ((R)-N-(2-(6-chloro-5-methoxy-lH-indol- 3yl) propyl) acetamide) (disclosed in U.S. Patent No. 4,997,845), agomelatine (N- [2-(7-methoxy-l-naphthyl)ethyl]acetamide) (disclosed in U.S. Patent No.
5,225,442), ramelteon ((S)-N-[2-(l,6,7,8-tetrahydro-2H-indeno- [5,4-b] furan-8- yl)ethyl]propionamide), 2-phenylmelatonin, 8-M-PDOT, 2-iodomelatonin, and 6- chloromelatonin.
Additional melatonin agonists include, without limitation, those listed in U.S. Patent Application Publication No. 20050164987, which is incorporated herein by reference, specifically: TAK-375 (see Kato, K. et al. Int. J.
Neuropsychopharmacol. 2000, 3 (Suppl. 1): Abst P.03.130; see also abstracts P.03.125 and P.03.127), CGP 52608 (l-(3-allyl-4-oxothiazolidine-2-ylidene)-4- met- hylthiosemicarbazone) (See Missbach et al., J. Biol. Chem. 1996, 271, 13515-22), GR196429 (N-[2-[2,3,7,8-tetrahydro-lH-fur-o(2,3-g)indol-l- yl] ethyl] acetamide) (see Beresford et al., J. Pharmacol. Exp. Ther. 1998, 285, 1239-1245), S20242 (N-[2-(7-methoxy napth-l-yl) ethyl] propionamide) (see Depres-Brummer et al., Eur. J. Pharmacol. 1998, 347, 57-66), S-23478 (see Neuropharmacology July 2000), S24268 (see Naunyn Schmiedebergs Arch. June 2003), S25150 (see Naunyn Schmiedebergs Arch. June 2003), GW-290569, luzindole (2-benzyl-N-acetyltryptamine) (see U.S. Patent No. 5,093,352), GR135531 (5-methoxycarbonylamino-N-acetyltrypt- amine) (see U.S. Patent Application Publication No. 20010047016), Melatonin Research Compound A, Melatonin Agonist A (see IMSWorld R&D Focus August 2002), Melatonin
Analogue B (see Pharmaprojects August 1998), Melatonin Agonist C (see Chem. Pharm. Bull. (Tokyo) January 2002), Melatonin Agonist D (see J. Pineal Research November 2000), Melatonin Agonist E (see Chem. Pharm. Bull. (Tokyo) Febrary 2002), Melatonin Agonist F (see Reprod. Nutr. Dev. May 1999), Melatonin Agonist G (see J. Med. Chem. October 1993), Melatonin Agonist H (see Famaco March 2000), Melatonin Agonist I (see J. Med. Chem. March 2000), Melatonin Analog J (see Bioorg. Med. Chem. Lett. March 2003), Melatonin Analog K (see MedAd News September 2001), Melatonin Analog L, AH-001 (2-acetamido-8- methoxytetralin) (see U.S. Patent No. 5,151,446), GG-012 (4-methoxy-2- (methylene propylamide)indan) (see Drijfhout et al., Eur. J. Pharmacol. 1999, 382, 157-66), Enol-3-IPA, ML-23 (N-2,4-dinitrophenyl-5-methoxy-tryptamine ) (see U.S. Patent No. 4,880,826), SL-18.1616, IP-100-9 (US 5580878), Sleep Inducing Peptide A, AH-017 (see U.S. Patent No. 5,151,446), AH-002 (8-methoxy- 2-propionamido-tetralin) (see U.S. Patent No. 5,151,446), and IP-101.
Metabolites, prodrugs, stereoisomers, polymorphs, hydrates, solvates, and salts of the above compounds that are directly or indirectly active can, of course, also be used in the practice of this invention.
Melatonin agonists with a MT1R and MT2R binding profile similar to that of tasimelteon, which has 2 to 4 time greater specificity for MT2R, are preferred.
Tasimelteon can be synthesized by procedures known in the art. The preparation of a 4-vinyl-2,3-dihydrobenzofuran cyclopropyl intermediate can be carried out as described in US7754902, which is incorporated herein by reference as though fully set forth.
Pro-drugs, e.g., esters, and pharmaceutically acceptable salts can be prepared by exercise of routine skill in the art.
In patients suffering a Non-24, the melatonin and Cortisol circadian rhythms and the natural day/night cycle become desynchronized. For example, in patients suffering from a free-running circadian rhythm, melatonin and Cortisol acrophases occur more than 24 hours, e.g., >24.1 hours, prior to each previous day’s melatonin and Cortisol acrophase, respectively, resulting in desynchronization for days, weeks, or even months, depending upon the length of a patient’s circadian rhythm, before the melatonin, Cortisol, and day /night cycles are again temporarily synchronized.
Chronic misalignment of Cortisol has been associated with metabolic, cardiac, cognitive, neurologic, neoplastic, and hormonal disorders. Such disorders include, e.g., obesity, depression, neurological impairments.

INTRODUCTION
Tasimelteon has the chemical name: trans-N-[[2-(2,3-dihydrobenzofuran-4-yl)cycloprop-1yl]methyl]propanamide, has the structure of Formula I:
and is disclosed in U.S. Pat. No. 5,856,529 and in US 20090105333, both of which are incorporated herein by reference as though fully set forth.
Tasimelteon is a white to off-white powder with a melting point of about 78° C. (DSC) and is very soluble or freely soluble in 95% ethanol, methanol, acetonitrile, ethyl acetate, isopropanol, polyethylene glycols (PEG-300 and PEG-400), and only slightly soluble in water. The native pH of a saturated solution of tasimelteon in water is 8.5 and its aqueous solubility is practically unaffected by pH. Tasimelteon has 2-4 times greater affinity for MT2R relative to MT1R. It’s affinity (Ki) for MT1R is 0.3 to 0.4 and for MT2R, 0.1 to 0.2. Tasimelteon is useful in the practice of this invention because it is a melatonin agonist that has been demonstrated, among other activities, to entrain patients suffering from Non-24.
SYNTHESIS
(1R-trans)-N-[[2 – (2,3-dihydro-4 benzofuranyl) cyclopropyl] methyl] propanamide PATENT: BRISTOL-MYERS SQUIBB PRIORITY DATE: 1996 HYPNOTIC


PREPARATION OF XV
XXIV D-camphorsulfonic acid IS REACTED WITH THIONYL CHLORIDE TO GIVE
…………XXV (1S, 4R) -7,7-dimethyl-2-oxo-bicyclo [2.2.1] heptane-1-methanesulfonyl chloride
TREATED WITH
XXVI ammonium hydroxide
TO GIVE
XXVII (1S, 4R) -7,7-dimethyl-2-oxo-bicyclo [2.2.1] heptane-1-methanesulfonamide
TREATED WITH AMBERLYST15
….XXVIII (3aS, 6R) -4,5,6,7-tetrahydro-8 ,8-dimethyl-3H-3a ,6-methano-2 ,1-benzisothiazole-2 ,2-dioxide
TREATED WITH LAH, ie double bond is reduced to get
…..XV (3aS, 6R, 7aR)-hexahydro-8 ,8-dimethyl-3H-3a ,6-methano-2 ,1-benzisothiazole-2 ,2-dioxide

Intermediate
I 3-hydroxybenzoic acid methyl ester
II 3-bromo-1-propene
III 3 – (2-propenyloxy) benzoic acid methyl ester
IV 3-hydroxy-2-(2-propenyl) benzoic acid methyl ester
V 2,3-dihydro-4-hydroxy-2-benzofurancarboxylic acid methyl ester
VI benzofuran-4-carboxylic acid methyl ester
VII benzofuran-4-carboxylic acid
VIII 2,3-dihydro-4-benzofurancarboxylic acid
IX 2,3-dihydro-4-benzofuranmethanol
X 2,3-dihydro-4-benzofurancarboxaldehyde
XI Propanedioic acid
XII (E) -3 – (2,3-dihydro-4-benzofuranyl) propenoic acid
XIII thionyl chloride
XIV (E) -3 – (2,3-dihydro-4-benzofuranyl) propenoyl chloride
XV (3aS, 6R, 7aR)-hexahydro-8 ,8-dimethyl-3H-3a ,6-methano-2 ,1-benzisothiazole-2 ,2-dioxide
XVI (3aS,6R,7aR)-1-[(E)-3-(2,3-dihydro-4-benzofuranyl)-1-oxo-2-propenyl]hexahydro-8,8-dimethyl-3H-3a,6-methano-2,1-benzisothiazole-2,2-dioxide
XVII (3aS,6R,7aR)-1-[[(1R,2R)-2-(2,3-dihydro-4-benzofuranyl)cyclopropyl]carbonyl]hexahydro-8,8-dimethyl-3H-3a,6-methano-2,1-benzisothiazole-2,2-dioxide
XVIII [R-(R *, R *)] -2 – (2,3-dihydro-4-benzofuranyl) cyclopropanemethanol
XIX [R-(R *, R *)] -2 – (2,3-dihydro-4-benzofuranyl) cyclopropanecarboxaldehyde
XX hydroxylamine hydrochloride
XXI [R-(R *, R *)] -2 – (2,3-dihydro-4-benzofuranyl) cyclopropanecarbaldehyde oxime
XXII [R-(R *, R *)] -2 – (2,3-dihydro-4-benzofuranyl) cyclopropanemethanamine
XXIII propanoyl chloride
XXIV D-camphorsulfonic acid
XXV (1S, 4R) -7,7-dimethyl-2-oxo-bicyclo [2.2.1] heptane-1-methanesulfonyl chloride
XXVI ammonium hydroxide
XXVII (1S, 4R) -7,7-dimethyl-2-oxo-bicyclo [2.2.1] heptane-1-methanesulfonamide
XXVIII (3aS, 6R) -4,5,6,7-tetrahydro-8 ,8-dimethyl-3H-3a ,6-methano-2 ,1-benzisothiazole-2 ,2-dioxide
Bibliography
– Patents: Benzofuran and dihydrobenzofuran melatonergic agents: US5856529 (1999)
Priority: US19960032689P, 10 Dec. 1996 (Bristol-Myers Squibb Company, U.S.)
– Preparation III (quinazolines): US2004044015 (2004) Priority: EP20000402845, 13 Oct. 2000
– Preparation of VII (aminoalkylindols): Structure-Activity Relationships of Novel Cannabinoid Mimetics Eissenstat et al, J.. Med. Chem. 1995, 38, 3094-3105
– Preparation XXVIII: Towson et al. Organic Syntheses, Coll. Vol. 8, p.104 (1993) Vol. 69, p.158 (1990)
– Preparation XV: Weismiller et al. Organic Syntheses, Coll. Vol. 8, p.110 (1993) Vol. 69, p.154 (1990).
– G. Birznieks et al. Melatonin agonist VEC-162 Improves sleep onset and maintenance in a model of transient insomnia. Sleep 2007, 30, 0773 Abstract.
-. Rajaratnam SM et al, The melatonin agonist VEC-162 Phase time immediately advances the human circadian system, Sleep 2006, 29, 0159 Abstract.
-. AK Singh et al, Evolution of a manufacturing route for a highly potent drug candidate, 229th ACS Natl Meet, March 13-17, 2005, San Diego, Abstract MEDI 576.
– Vachharajani NN et al, Preclinical pharmacokinetics and metabolism of BMS-214778, a novel melatonin receptor agonist, J Pharm Sci. 2003 Apr; 92 (4) :760-72.
. – JW Scott et al, Catalytic Asymmetric Synthesis of a melotonin antagonist; synthesis and process optimization. 223rd ACS Natl Meet, April 7-11, Orlando, 2002, Abstract ORGN 186.
SYNTHESIS CONSTRUCTION AS IN PATENT
GENERAL SCHEMES
Reaction Scheme 1
The syntheses of the 4-aryl-propenoic acid derivatives, 2 and 3, are shown in Reaction Scheme 1. The starting aldehydes, 1 , can be prepared by methods well known to those skilled in the art. Condensation of malonic acid with the aldehydes, 1, in solvents such as pyridine with catalysts such as piperidine or pyrrolidine, gives the 4-aryl- propenoic acid, 2. Subsequent conversion of the acid to the acid chloride using reagents such as thionyl chloride, phosphoryl chloride, or the like, followed by reaction with N,0-dimethyl hydroxylamine gives the amide intermediate 3 in good yields. Alternatively, aldehyde 1 can be converted directly to amide 3 using reagents such as diethyl (N-methoxy- N-methyl-carbamoylmethyl)phosphonate with a strong base such as sodium hydride.
Reaction Scheme 2
The conversion of the amide intermediate 3 to the racemic, trans- cyclopropane carboxaldehyde intermediate, 4, is shown in Reaction Scheme 2. Intermediate 3 was allowed to react with cyclopropanating reagents such as trimethylsulfoxonium iodide and sodium hydride in solvents such as DMF, THF, or the like. Subsequent reduction using reagents such as LAH in solvents such as THF, ethyl ether, or the like, gives the racemic, trans-cyclopropane carboxaldehyde intermediates, 4.
Reaction Scheme 3
Racemic cyclopropane intermediate 5 (R = halogen) can be prepared from intermediate 2 as shown in Reaction Scheme 3. Intermediate 2 was converted to the corresponding allylic alcohol by treatment with reducing agents such as sodium borohydride plus iodine in solvents such as THF. Subsequent acylation using reagents such as acetic anhydride in pyridine or acetyl chloride gave the allylic acetate which was allowed to react with cyclopropanating reagents such as sodium chloro-difluoroacetate in diglyme to provide the racemic, trans- cyclopropane acetate intermediates, 5. Reaction Scheme 4
The conversion of the acid 2 to the chiral cyclopropane carboxaldehyde intermediate, (-)-(trans)-4, is shown in Reaction Scheme 4. Intermediate 2 is condensed with (-)-2,10-camphorsultam under standard conditions, and then cyclopropanated in the presence of catalysts such as palladium acetate using diazomethane generated from reagents such as 1-methyl-3-nitro-1-nitrosoguanidine. Subsequent reduction using reagents such as LAH in solvents such as THF, followed by oxidation of the alcohol intermediates using reagents such as DMSO/oxalyl chloride, or PCC, gives the cyclopropane carboxaldehyde intermediate, (-)-(trans)-4, in good yields. The enantiomer, (+)-(trans)-4, can also be obtained employing a similar procedure using (+)-2,10- camphorsultam in place of (-)-2,10-camphorsultam.
When it is desired to prepare compounds of Formula I wherein m = 2, the alcohol intermediate may be activated in the conventional manner such as with mesyl chloride and treated with sodium cyanide followed by reduction of the nitrile group with a reducing agent such as LAH to produce the amine intermediate 6.
Reaction Scheme 5
Reaction Scheme 5 shows the conversion of intermediates 4 and 5 to the amine intermediate, 7, and the subsequent conversion of 6. or 7 to compounds of Formula I. The carboxaldehyde intermediate, 4, is condensed with hydroxylamine and then reduced with reagents such as LAH to give the amine intermediate, 7. The acetate intermediate 5 is hydrolyzed with potassium hydroxide to the alcohol, converted to the mesylate with methane sulfonyl chloride and triethyl amine in CH2CI2and then converted to the azide by treatment with sodium azide in solvents such as DMF. Subsequent reduction of the azide group with a reducing agent such as LAH produced the amine intermediate 7. Further reaction of 6 or 7 with acylating reagents gives compounds of Formula I. Suitable acylating agents include carboxylic acid halides, anhydrides, acyl imidazoles, alkyl isocyanates, alkyl isothiocyanates, and carboxylic acids in the presence of condensing agents, such as carbonyl imidazole, carbodiimides, and the like. Reaction Scheme 6
Reaction Scheme 6 shows the alkylation of secondary amides of Formula I (R2 = H) to give tertiary amides of Formula I (R2 = alkyl). The secondary amide is reacted with a base such as sodium hydride, potassium tert-butoxide, or the like, and then reacted with an alkylating reagent such as alkyl halides, alkyl sulfonate esters, or the like to produce tertiary amides of Formula I.
Reaction Scheme 7
Reaction Scheme 7 shows the halogenation of compounds of Formula I. The carboxamides, i (Q1 = Q2 = H), are reacted with excess amounts of halogenating agents such as iodine, N-bromosuccinimide, or the like to give the dihalo-compounds of Formula I (Q1 = Q2 = halogen). Alternatively, a stoichiometric amount of these halogenating agents can be used to give the monohalo-compounds of Formula I (Q1 = H, Q2 = halogen; or Q1 = halogen, Q2 = H). In both cases, additives such as lead IV tetraacetate can be used to facilitate the reaction. Biological Activity of the Compounds
The compounds of the invention are melatonergic agents. They have been found to bind human melatonergic receptors expressed in a stable cell line with good affinity. Further, the compounds are agonists as determined by their ability, like melatonin, to block the forskolin- stimulated accumulation of cAMP in certain cells. Due to these properties, the compounds and compositions of the invention should be useful as sedatives, chronobiotic agents, anxiolytics, antipsychotics, analgesics, and the like. Specifically, these agents should find use in the treatment of stress, sleep disorders, seasonal depression, appetite regulation, shifts in circadian cycles, melancholia, benign prostatic hyperplasia and related conditions
EXPERIMENTAL PROCEDURES
SEE ORIGINAL PATENT FOR CORECTIONS
Preparation 1
Benzofuran-4-carboxaldehyde
Step 1 : N-Methoxy-N-methyl-benzofuran-4-carboxamide
A mixture of benzofuran-4-carboxylic acid [Eissenstat, et al.. J. Medicinal Chemistry, 38 (16) 3094-3105 (1995)] (2.8 g, 17.4 mmol) and thionyl chloride (25 mL) was heated to reflux for 2 h and then concentrated in vacuo. The solid residue was dissolved in ethyl acetate (50 mL) and a solution of N,O-dimethylhydroxylamine hydrochloride (2.8 g) in saturated NaHC03(60 mL) was added with stirring. After stirring for 1.5 h, the ethyl acetate layer was separated. The aqueous layer was extracted with ethyl acetate. The ethyl acetate extracts were combined, washed with saturated NaHCO3 and concentrated in vacuo to give an oil (3.2 g, 95.4%).
Step 2: Benzofuran-4-carboxaldehyde
A solution of N-methoxy-N-methyl-benzofuran-4-carboxamide (3.2 g, 16.6 mmol) in THF (100 mL) was cooled to -45°C and then LAH (0.7 g, 18.7 mmol) was added. The mixture was stirred for 15 min, allowed to warm to -5°C, and then recooled to -45°C. Saturated KHS04 (25 mL) was added with vigorous stirring, and the mixture was allowed to warm to room temperature. The precipitate was filtered and washed with acetone. The filtrate was concentrated in vacuo to give an oil (2.3 g, 94%). Preparation 2
2,3-Dihydrobenzofuran-4-carboxaldehyde
Step 1 : 2,3-Dihydrobenzofuran-4-carboxylic acid
Benzofuran-4-carboxylic acid (10.0 g, 61 .7 mmol) was hydrogenated (60 psi) in acetic acid (100 mL) over 10% Pd/C (2 g) for 12 hr. The mixture was filtered and the filtrate was diluted with water (500 mL) to give 2,3- dihydrobenzofuran-4-carboxylic acid as a white powder (8.4 g, 83%). A sample was recrystallized from isopropanol to give fine white needles (mp: 185.5-187.5°C).
Step 2: (2,3-Dihydrobenzofuran-4-yl)methanol
A solution of 2,3-dihydrobenzofuran-4-carboxylic acid (10 g, 61 mmol) in THF (100 mL) was stirred as LAH (4.64 g, 122 mmol) was slowly added. The mixture was heated to reflux for 30 min. The mixture was cooled and quenched cautiously with ethyl acetate and then with 1 N HCI (150 mL). The mixture was then made acidic with 12 N HCI until all the inorganic precipitate dissolved. The organic layer was separated, and the inorganic layer was extracted twice with ethyl acetate. The organic layers were combined, washed twice with brine, and then concentrated in vacuo. This oil was Kϋgelrohr distilled to a clear oil that crystallized upon cooling (8.53 g, 87.6%).
Step 3: 2.3-Dihydrobenzofuran-4-carboxaldehyde
DMSO (8.10 mL, 1 14 mmol) was added at -78°C to a stirred solution of oxalyl chloride in CH2CI2 (40 mL of a 2M solution). A solution of (2,3- dihydrobenzofuran-4-yl)methanol (8.53 g, 56.9 mmol) in CH2CI2 (35 mL) was added dropwise, and the solution stirred at -78°C for 30 min. Triethyl amine (33 mL, 228 mmol) was added cautiously to quench the reaction. The resulting suspension was stirred at room temperature for 30 min and diluted with CH2CI2 (100 mL). The organic layer was washed three times with water, and twice with brine, and then concentrated in vacuo to an oil (8.42 g, 100%) that was used without purification.
Preparation 16
(±)-(trans)-2-(2,3-Dihyd robenzofuran-4-yl)cyclopropane- carboxaldehyde
Step 1 : (±Htrans)-N-Methoxy-N-methyl-2-(2.3-dihydrobenzofuran-4- yhcyclopropanecarboxamide
Trimethylsulfoxonium iodide (9.9 g, 45 mmol) was added in small portions to a suspension of sodium hydride (1 .8 g, 45 mmol) in DMF (120 mL). After the foaming had subsided (10 min), a solution of (trans)- N-methoxy-N-methyl-3-(2,3-dihydrobenzofuran-4-yl)propenamide (3.5 g, 15 mmol) in DMF (60 mL) was added dropwise, with the temperature maintained between 35-40°C. The mixture was stirred for 3 h at room temperature. Saturated NH4CI (50 mL) was added dropwise and the mixture was extracted three times with ethyl acetate. The organic extracts were combined, washed with H2O and brine, dried over K2CO3, and concentrated in vacuo to give a white wax (3.7 g, 100%).
Step 2: (±)-(trans)- 2-(2.3-Dihydrobenzofuran-4-yl)cyclopropane- carboxaldehyde
A solution of (±)-(trans)-N-methoxy-N-methyl-2-(2,3-dihydrobenzofuran- 4-yl)cyclopropanecarboxamide (3.7 g, 15 mmol) in THF (10 mL) was added dropwise to a rapidly stirred suspension of LAH (683 mg, 18 mmol) in THF (50 mL) at -45°C, maintaining the temperature below -40°C throughout. The cooling bath was removed, the reaction was allowed to warm to 5°C, and then the reaction was immediately recooled to -45°C. Potassium hydrogen sulfate (3.4 g, 25.5 mmol) in H20 (50 mL) was cautiously added dropwise, the temperature maintained below – 30°C throughout. The cooling bath was removed and the suspension was stirred at room temperature for 30 min. The mixture was filtered through Celite and the filter cake was washed with ether. The combined filtrates were then washed with cold 1 N HCI, 1 N NaOH, and brine. The filtrates were dried over MgSO4, and concentrated in vacuo to give a clear oil (2.6 g, 99%).
Preparation 18
(-)-(trans)-2-(2.3-Dihydrobenzofuran-4-yl)cyclopropane-carboxaldehyde
Step 1 : (-Htrans)-N-[3-(2.3-Dihvdrobenzofuran-4-yl)-propenoyll-2.10- camphorsultam
To a solution of (-)-2,10-camphorsultam (8.15 g, 37.9 mmol) in 50 mL toluene at 0°C was added sodium hydride (1.67 g, 41.7 mmol). After stirring for 0.33 h at 0°C and 0.5 h at 20°C and recooling to 0°C, a solution of 3-(2,3-dihydrobenzofuran-4-yl)-2-propenoyl chloride
(37.9 mmol), prepared in situ from the corresponding acid and thionyl chloride (75 mL), in toluene (50 mL), was added dropwise. After stirring for 18 h at 20°C, the mixture was diluted with ethyl acetate and washed with water, 1 N HCI, and 1 N NaOH. The organic solution was dried and concentrated in vacuo to give 15.8 g of crude product. Recrystallization form ethanol-methanol (600 mL, 1 :1) gave the product (13.5 g, 92%, mp 199.5-200°C).
Step 2: (-)-N-[[(trans)-2-(2,3-Dihydrobenzofuran-4-yl)-cyclopropylj- carbonylj-2, 10-camphorsultam
1 -Methyl-3-nitro-1 -nitrosoguanidine (23.88g 163 mmol) was added in portions to a mixture of 10 N sodium hydroxide (60 mL) and ether (200 mL) at 0°C. The mixture was shaken vigorously for 0.25 h and the ether layer carefully decanted into a solution of (-)-N-[3-(2,3-dihydrobenzofuran-4-yl)-2-propenoyl]-2,10-camphorsultam (9.67 g, 25 mmol) and palladium acetate (35 mg) in methylene chloride (200 mL). After stirring for 18 h, acetic acid (5 mL) was added to the reaction and the mixture stirred for 0.5 h. The mixture was washed with 1 N HCI, 1 N NaOH and brine. The solution was dried, concentrated in vacuo and the residue crystallized twice from ethanol to give the product (6.67 g, 66.5%, mp 157-159°C).
Step 3: (-)-(trans)-2-(2,3-Dihydrobenzofuran-4-yl)cyclopropane- methanol
A solution of (-)-N-[(trans)-2-(2,3-dihydrobenzofuran-4-yl)cyclo-propanecarbonylj-2,10-camphorsultam (4.3 g, 10.7 mmol) in THF (50 mL) was added dropwise to a mixture of LAH (0.81 g, 21.4 mmol) in THF (50 mL) at -45°C. The mixture was stirred for 2 hr while it warmed to 10°C. The mixture was recooled to -40°C and hydrolyzed by the addition of saturated KHS0 (20 mL). The mixture was stirred at room temperature for 30 minutes and filtered. The precipitate was washed twice with acetone. The combined filtrate and acetone washes were concentrated in vacuo. The gummy residue was dissolved in ether, washed with 1 N NaOH and 1 N HCI, and then dried in vacuo to give the product (2.0 g, 98.4%).
Step 4: (-)-(trans)-2-(2.3-Dihydrobenzofuran-4-yl)cyclopropane- carboxaldehyde DMSO (1.6 g, 21 mmol) was added to oxalyl chloride in CH2CI2(7.4 mL of 2 M solution, 14.8 mmole) at -78°C. The (-)-(trans)-2-(2,3-dihydrobenzofuran-4-yl)-cyclopropylmethanol (2.0 g, 10.5 mmol) in CH2CI2(15 mL) was added. The mixture was stirred for 20 min and then triethylamine (4.24 g, 42 mmol) was added. The mixture was warmed to room temperature and stirred for 30 min. The mixture was diluted with CH2CI2 and washed with water, 1 N HCI, and then 1 N NaOH. The organic layer was dried and concentrated iι> vacuo to give the aldehyde product (1.98 g, 100%).
Preparation 24
(-)-(trans)-2-(2.3-Dihydrobenzofuran-4-yl)cyclopropane-methanamine A mixture of (-)-(trans)-2-(2,3-dihydrobenzofuran-4-yl)cyclopropane-carboxaldehyde (1.98 g, 10.5 mmol), hydroxylamine hydrochloride (2.29 g, 33 mmol), and 30% NaOH (3.5 mL, 35 mmol), in 5:1
ethanol/water (50 mL) was heated on a steam bath for 2 h. The solution was concentrated in vacuo. and the residue mixed with water. The mixture was extracted with CH2CI2. The organic extracts were dried and concentrated in vacuo to give a solid which NMR analysis showed to be a mixture of the cis and trans oximes. This material was dissolved in THF (20 mL) and added to solution of alane in THF [prepared from LAH (1.14 g, 30 mmol) and H2S04 (1.47 g, 15 mmol) at 0°Cj. The reaction was stirred for 18 h, and quenched successively with water (1.15 mL), 15% NaOH (1.15 mL), and then water (3.45 mL). The mixture was filtered and the filtrate was concentrated in vacuo. The residue was mixed with ether and washed with water and then 1 N HCI. The acid washes were made basic and extracted with CH2CI . The extracts were dried and concentrated in vacuo to give the amine product (1.4 g, 70.5%). The amine was converted to the fumarate salt in ethanol (mp: 197-198°C).
Anal. Calc’d for C12H15NO • C4H404: C, 62.94; H, 6.27; N, 4.59.
Found: C, 62.87; H, 6.31 ; N, 4.52.
FINAL PRODUCT TASIMELTEON
Example 2
(-)-(trans)-N-[[2-(2,3-Dihydrobenzofuran-4-yl)cycloprop-1-yl]methyl]propanamide
This compound was prepared similar to the above procedure using propionyl chloride and (-)-(trans)-2-(2,3-dihydrobenzofuran-4-yl)- cyclopropanemethanamine to give an oil that solidified upon standing to an off-white solid (61 %, mp: 71-72°C). IR (NaCI Film): 3298, 1645, 1548, 1459, 1235 cm“1.
Mo5 : -17.3°
Anal. Calc’d for C15H19N02: C, 73.44; H, 7.87; N, 5.71 . Found: C, 73.28; H, 7.68; N, 5.58
SYNTHESIS
Synthesis Path
SYN

Tasimelteon (Hetlioz)Tasimelteon, which is marketed by Vanda Pharmaceuticals as Hetlioz and developed in partnership with Bristol-Myers Squibb,is a drug that was approved by the US FDA in January 2014 for the treatment of non-24-hour sleep–wake disorder (also called Non-24, N24 and N24HSWD).234 Tasimelteon is a melatonin MT1
and MT2 receptor agonist; because it exhibits a greater affinity to the MT2 receptor than MT1, is also known as Dual Melatonin
Receptor Agonist.234 Two randomized controlled trials (phases II
and III) demonstrated that tasimelteon improved sleep latency
and maintenance of sleep with a shift in circadian rhythms, and
therefore has the potential to treat patients with transient insomnia
associated with circadian rhythm sleep disorders.235 Preclinical
studies showed that the drug has similar phase-shifting properties
to melatonin, but with less vasoconstrictive effects.236 The most
likely scale preparation of the drug, much of which has been published
in the chemical literature, is described below in Scheme 44.
Activation of commercial bis-ethanol 250 with 2.5 equivalents
of the Vilsmeier salt 251 followed by treatment with base resulted
an intramolecular cyclization reaction with the proximal phenol
and concomitant elimination of the remaining imidate to deliver
the vinylated dihydrobenzofuran 252 in 76% yield.237 Interestingly,
this reaction could be performed on multi-kilogram scale, required
no chromatographic purification, and generated environmentallyfriendly
DMF and HCl as byproducts.237 Sharpless asymmetric
dihydroxylation of olefin 252 delivered diol 253 in 86% yield and
impressive enantioselectivity (>99% ee). This diol was then activated
with trimethylsilyl chloride and then treated with base to generate epoxide 254.238 Next, a modified Horner–Wadsworth–
Emmons reaction involving triethylphosphonoacetate (TEPA, 255)
was employed to convert epoxide 254 to cyclopropane 256.239
The reaction presumably proceeds through removal of the acidic
TEPA proton followed by nucleophilic attack at the terminal epoxide
carbon. The resulting alkoxide undergoes an intramolecular
phosphoryl transfer reaction resulting in an enolate, which then attacked the newly formed phosphonate ester in an SN2 fashion
resulting in the trans-cyclopropane ester, which was ultimately
saponified and re-acidified to furnish cyclopropane acid 256.239
Conversion of this acid to the corresponding primary amide preceded
carbonyl reduction with sodium borohydride. The resulting
amine was acylated with propionyl chloride to furnish tasimelteon
(XXXI) as the final product in 86% yield across the four-step
sequence.
PATENTS
| US2010261786 | 10-15-2010 | PREDICTION OF SLEEP PARAMETER AND RESPONSE TO SLEEP-INDUCING COMPOUND BASED ON PER3 VNTR GENOTYPE |
| US2009209638 | 8-21-2009 | TREATMENT FOR DEPRESSIVE DISORDERS |
| US6060506 | 5-10-2000 | Benzopyran derivatives as melatonergic agents |
| US5981571 | 11-10-1999 | Benzodioxa alkylene ethers as melatonergic agents |
| WO9825606 | 6-19-1998 | BENZODIOXOLE, BENZOFURAN, DIHYDROBENZOFURAN, AND BENZODIOXANE MELATONERGIC AGENTS |
| WO2007137244A1 * | May 22, 2007 | Nov 29, 2007 | Gunther Birznieks | Melatonin agonist treatment |
| US4880826 | Jun 25, 1987 | Nov 14, 1989 | Nava Zisapel | Melatonin antagonist |
| US4997845 | May 10, 1990 | Mar 5, 1991 | Eli Lilly And Company | β-alkylmelatonins as ovulation inhibitors |
| US5093352 | May 16, 1990 | Mar 3, 1992 | Whitby Research, Inc. | Antidepressant agents |
| US5151446 | Mar 28, 1991 | Sep 29, 1992 | Northwestern University | Substituted 2-amidotetralins as melatonin agonists and antagonists |
| US5225442 | Jan 3, 1992 | Jul 6, 1993 | Adir Et Compagnie | Compounds having a naphthalene structure |
| US5580878 | Jun 7, 1995 | Dec 3, 1996 | Interneuron Pharmaceuticals, Inc. | Substituted tryptamines phenalkylamines and related compounds |
| US5856529 | Dec 9, 1997 | Jan 5, 1999 | Bristol-Myers Squibb Company | Benzofuran and dihydrobenzofuran melatonergic agents |
| US6211225 | Jun 6, 2000 | Apr 3, 2001 | Bristol-Meyers Squibb | Heterocyclic aminopyrrolidine derivatives as melatonergic agents |
| US7754902 | May 18, 2006 | Jul 13, 2010 | Vanda Pharmaceuticals, Inc. | Ruthenium(II) catalysts for use in stereoselective cyclopropanations |
| US20010047016 | Apr 12, 2001 | Nov 29, 2001 | Gregory Oxenkrug | Method for treating depression |
| US20050164987 | Dec 22, 2004 | Jul 28, 2005 | Barberich Timothy J. | Melatonin combination therapy for improving sleep quality |
| US20090105333 | May 22, 2007 | Apr 23, 2009 | Gunther Birznieks | Melatonin agonist treatment |
extra info
 |
- Department of Chemistry, Drexel University, Philadelphia, PA 19104.
- Shriner, R. L.; Shotton, J. A.; Sutherland, H. J. Am. Chem. Soc.1938, 60, 2794.
- Oppolzer, W.; Chapuis, C.; Bernardinelli, G. Helv. Chim. Acta1984, 67, 1397.
- Vandewalle, M.; Van der Eycken, J.; Oppolzer, W.; Vullioud, C. Tetrahedron1986, 42, 4035.
- Davis, F. A.; Towson, J. C.; Weismiller, M. C.; Lal, G.; Carroll,, P. J. J. Am. Chem. Soc.1988, 110, 8477.
- Oppolzer, W. Tetrahedron1987, 43, 1969.
- Oppolzer, W.; Mills, R. J.; Pachinger, W.; Stevenson, T. Helv. Chim. Acta1986, 69, 1542; Oppolzer, W.; Schneider, P. Helv. Chim. Acta1986, 69, 1817; Oppolzer, W.; Mills, R. J.; Réglier, M. Tetrahedron Lett.1986, 27, 183; Oppolzer, W.; Poli. G.Tetrahedron Lett.1986, 27, 4717; Oppolzer, W.; Poli, G.; Starkemann, C.; Bernardinelli, G. Tetrahedron Lett.1988, 29, 3559.
- Oppolzer, W.; Barras, J-P. Helv. Chim. Acta1987, 70, 1666.
- Curran, D. P.; Kim, B. H.; Daugherty, J.; Heffner, T. A. Tetrahedron Lett.1988, 29, 3555.
- Differding, E.; Lang, R. W. Tetrahedron Lett.1988, 29, 6087.
 |
This preparation is referenced from:
- Org. Syn. Coll. Vol. 8, 110
- Org. Syn. Coll. Vol. 9, 212
-
References and Notes
- Department of Chemistry, Drexel University, Philadelphia, PA 19104.
- Reychler, M. A. Bull. Soc. Chim. III1889, 19, 120.
- Armstrong, H. E.; Lowry, T. M. J. Chem. Soc., Trans.1902, 81, 1441.
- Dauphin, G.; Kergomard, A.; Scarset, A. Bull. Soc. Chim. Fr.1976, 862.
- Davis, F. A.; Jenkins, Jr., R. H.; Awad, S. B.; Stringer, O. D.; Watson, W. H.; Galloy, J. J. Am. Chem. Soc.1982, 104, 5412.
- Vandewalle, M.; Van der Eycken, J.; Oppolzer, W.; Vullioud, C. Tetrahedron, 1986, 42, 4035.
- Davis, F. A.; Towson, J. C.; Weismiller, M. C.; Lal, S.; Carroll, P. J. J. Am. Chem. Soc.1988, 110, 8477.
- Davis, F. A.; Weismiller, M. C.; Lal, G. S.; Chen, B. C.; Przeslawski, R. M. Tetrahedron Lett., 1989, 30, 1613.
- Oppolzer, W. Tetrahedron1987, 43, 1969.
- Glahsl, G.; Herrmann, R. J. Chem. Soc., Perkin Trans. I1988, 1753.
- Differding, E.; Lang, R. W. Tetrahedron Lett.1988, 29, 6087.
- For recent reviews on the chemistry of N-sulfonyloxaziridines, see: (a) Davis, F. A.; Jenkins, Jr., R. H. in “Asymmetric Synthesis,” Morrison, J. D., Ed.; Academic Press: Orlando, FL, 1984, Vol. 4, Chapter 4;
- Davis, F. A.; Haque, S. M. in “Advances in Oxygenated Processes,” Baumstark, A. L., Ed.; JAI Press: London, Vol. 2;
- Davis, F. A.; Sheppard, A. C. Tetrahedron1989, 45, 5703.
- Davis, F. A.; McCauley, Jr., J. P.; Chattopadhyay, S.; Harakal, M. E.; Towson, J. C.; Watson, W. H.; Tavanaiepour, I. J. Am. Chem. Soc.1987, 109, 3370.
- Davis, F. A.; Stringer, O. D.; McCauley, Jr., J. M. Tetrahedron1985, 41, 4747.
- Davis, F. A.; Chattopadhyay, S. Tetrahedron Lett.1986, 27, 5079.
- Davis, F. A.; Harakal, M. E.; Awad, S. B. J. Am. Chem. Soc.1983, 105, 3123.
- Davis, F. A.; Wei, J.; Sheppard, A. C.; Gubernick S. Tetrahedron Lett.1987, 28, 5115.
- Davis, F. A.; Lal, G. S.; Wei, J. Tetrahedron Lett.1988, 29, 4269.
- Davis, F. A.; Haque, M. S.; Ulatowski, T. G.; Towson, J. C. J. Org. Chem.1986, 51, 2402.
- Davis, F. A.; Haque, M. S. J. Org. Chem.1986, 51, 4083; Davis, F. A.; Haque, M. S.; Przeslawski, R. M. J. Org. Chem.1989, 54, 2021.
- Davis, F. A.; Ulatowski, T. G.; Haque, M. S. J. Org. Chem.1987, 52, 5288.
- Davis, F. A.; Sheppard, A. C., Lal, G. S. Tetrahedron Lett.1989, 30, 779.
- Davis, F. A.; Sheppard, A. C.; Chen, B. C.; Haque, M. S. J. Am. Chem. Soc.1990, 112, 6679.
a US 5 856 529 (Bristol-Myers Squibb; 5.1.1999; appl. 9.12.1997; USA-prior. 10.12.1996).
-
- b US 7 754 902 (Vanda Pharms.; 13.7.2010; appl. 18.5.2006).
-
treatment of circadian rhythm disorders:
- US 8 785 492 (Vanda Pharms.; 22.7.2014; appl. 25.1.2013; USA-prior. 26.1.2012).
-
synthesis cis-isomer:
- US 6 214 869 (Bristol-Myers Squibb; 10.4.2001; appl. 25.5.1999; USA-prior. 5.6.1998).
Patents
References
- Jump up^ “Tasimelteon Advisory Committee Meeting Briefing Materials”(PDF). Vanda Pharmaceuticals Inc. November 2013.
- Jump up^ “FDA transcript approval minutes” (PDF). FDA. November 14, 2013.
- ^ Jump up to:a b Food and Drug Administration (January 31, 2014). “FDA approves Hetlioz: first treatment for non-24 hour sleep-wake disorder”. FDA.
- Jump up^ “tasimelteon (Hetlioz) UKMi New Drugs Online Database”. Retrieved August 6, 2014.
- Jump up^ “HETLIOZ® Receives European Commission Approval for the Treatment of Non-24-Hour Sleep-Wake Disorder in the Totally Blind”. MarketWatch. PR Newswire. 7 July 2015. Retrieved 8 July 2015.
- Jump up^ Vachharajani, Nimish N.; Yeleswaram, Krishnaswamy; Boulton, David W. (April 2003). “Preclinical pharmacokinetics and metabolism of BMS-214778, a novel melatonin receptor agonist”. Journal of Pharmaceutical Sciences. 92 (4): 760–72. doi:10.1002/jps.10348. PMID 12661062.
- Jump up^ Sack, R. L.; Brandes, R. W.; Kendall, A. R.; Lewy, A. J. (2000). “Entrainment of Free-Running Circadian Rhythms by Melatonin in Blind People”. New England Journal of Medicine. 343 (15): 1070–7. doi:10.1056/NEJM200010123431503. PMID 11027741.
- Jump up^ “Safety and Efficacy of VEC-162 on Circadian Rhythm in Healthy Adult Volunteers”. ClinicalTrials.gov. |accessdate=May 15, 2014
- Jump up^ “VEC-162 Study in Healthy Adult Volunteers in a Model of Insomnia”. ClinicalTrials.gov. Retrieved May 15, 2014.
- Jump up^ “VEC-162 Study in Adult Patients With Primary Insomnia”. ClinicalTrials.gov. Retrieved May 15, 2014.
- Jump up^ Lynne Lamberg. “Improving Sleep and Alertness in the Blind (Part 5)”. Matilda Ziegler Magazine for the Blind. Retrieved May 15, 2014.
- Jump up^ Shantha MW Rajaratnam; Mihael H Polymeropoulos; Dennis M Fisher; Thomas Roth; Christin Scott; Gunther Birznieks; Elizabeth B Klerman (2009-02-07). “Melatonin agonist tasimelteon (VEC-162) for transient insomnia after sleep-time shift: two randomised controlled multicentre trials”. The Lancet. 373 (9662): 482–491. doi:10.1016/S0140-6736(08)61812-7. PMID 19054552. Retrieved 2010-02-23.
- Jump up^ Carome, Michael (1 July 2015). “Outrage of the Month: FDA Makes Major Blunder After Approving Drug for Rare Sleep Disorder”. Huffington Post. Retrieved 8 July 2015.
- Jump up^ Food and Drug Administration (January 31, 2014). “FDA NEWS RELEASE: FDA approves Hetlioz: first treatment for non-24 hour sleep–wake disorder in blind individuals”. FDA.
- Jump up^ “Side Effects Drug Center: Hetlioz Clinical Pharmacology”. RxList. February 10, 2014.
- Jump up^ “Side Effects Drug Center: Hetlioz Warnings and Precautions”. RxList. February 10, 2014.
In animal studies, administration of tasimelteon during pregnancy resulted in developmental toxicity (embryofetal mortality, neurobehavioral impairment, and decreased growth and development in offspring) at doses greater than those used clinically.
 |
|
 |
|
| Clinical data | |
|---|---|
| Trade names | Hetlioz |
| License data | |
| Pregnancy category |
|
| Routes of administration |
Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | not determined in humans[1] |
| Protein binding | 89–90% |
| Metabolism | extensive hepatic, primarily CYP1A2 and CYP3A4-mediated |
| Elimination half-life | 0.9–1.7 h / 0.8–5.9 h (terminal) |
| Excretion | 80% in urine, 4% in feces |
| Identifiers | |
| CAS Number | |
| PubChemCID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| ECHA InfoCard | 100.114.889 |
| Chemical and physical data | |
| Formula | C15H19NO2 |
| Molar mass | 245.32 g/mol |
| 3D model (JSmol) | |

DR ANTHONY MELVIN CRASTO Ph.D
GLENMARK SCIENTIST , NAVIMUMBAI, INDIA
Chemical and physical properties
Tasimelteon has two stereogenic centers. Besides the medically used trans-1 R , 2 R isomer (in the picture above left), there are thus three further stereoisomers that do not arise in the synthesis.
Tasimelteon is a white to off-white crystalline non-hygroscopic substance, soluble in water at physiologically relevant pH levels and readily soluble in alcohols, cyclohexane and acetonitrile. The compound occurs in two crystal forms. It is an anhydrate melting at 74 ° C and a hemihydrate . [4] The hemihydrate is from about 35 ° C the water of hydration and converts thereby in the anhydrate form to. [4] The anhydrate crystallizes in a monoclinic lattice with the space group P 2 1 , and the hemihydrate crystallizes in a tetragonal lattice with the space group P 4 3 21 2. [4]
4 Kaihang Liu, Zhou Xinbo, Zhejing Xu, Bai Hongzhen, Jianrong Zhu Jianming Gu, Guping Tang, Liu Xingang, Hu Xiurong: anhydrate and hemihydrate of Tasimelteon: Synthesis, structure, and pharmacokinetic study in J. Pharm. Biomed. Anal. 151 (2018) 235-243, doi : 10.1016 / j.jpba.2017.12.035 .
Isavuconazonium sulfate, Изавуконазониев сулфат


| MOLECULAR FORMULA: | C35H36F2N8O9S2 |
|---|---|
| MOLECULAR WEIGHT: | 814.837 g/mol |


Syn……https://newdrugapprovals.org/2013/10/02/isavuconazole-basilea-reports-positive-results-from-study/
PRODUCT PATENT
https://patents.google.com/patent/US6300353
InventorTadakatsu HayaseShigeyasu IchiharaYoshiaki IsshikiPingli LiuJun OhwadaToshiya SakaiNobuo ShimmaMasao TsukazakiIsao UmedaToshikazu Yamazaki
Current Assignee Basilea Pharmaceutica International Ltd Original
AssigneeBasilea Pharmaceutica AG Priority date 1998-03-06
https://patents.google.com/patent/WO1999045008A1/en
POLYMORPHS OF BASE
WO 2016055918
https://patents.google.com/patent/WO2016055918A1/en
PATENT
IN 2014MU03189
WOCKHARDT
Isavuconazole, isavuconazonium, Voriconazole, and Ravuconazole are azole derivatives and known as antifungal drugs for treatment of systemic mycoses as reported in US 5,648,372, US 5,792,781, US 6,300,353 and US 6,812,238. The US patent No. 6,300,353 discloses Isavuconazole and its process. It has chemical name [(2R,3R)-3-[4-(4-cyanophenyl)thiazol-2-yl)]-1-(1H-1,2,4-triazol-1-yl)-2-(2,5- difluorophenyl)-butan-2-ol;
The Isavuconazonium iodide hydrochloride and Isavuconazonium sulfate can be prepared according to known methods, e.g. pending Indian Patent Applications IN 2424/MUM/2014 and IN 2588/MUM/2014.
Example-1: Preparation of Amorphous Isavuconazole
4-cyano Phenacyl bromide F F N N N OH N S CN Formula-I Formula-III In a round bottomed flask charged ethanol (250 ml), thioamide compound of formula-II (25.0 gm) and 4-cyano phenacyl bromide (18.4 gm) under stirring. The reaction mixture were heated to 70 0C. After completion of reaction the solvent was removed under vacuum distillation and water (250 ml) and Ethyl acetate (350 ml) were added to reaction mass. The reaction mixture was stirred and its pH was adjusted between 7 to 7.5 by 10 % solution of sodium bicarbonate. The layer aqueous layer was discarded and organic layer was washed with saturated sodium chloride solution (100 ml) and concentrated under vacuum to get residue. The residue was suspended in methyl tert-butyl ether (250 ml) and the reaction mixture was heated to at 40°C to make crystals uniform and finally reaction mass is cooled to room temperature filtered and washed with the methyl tert-butyl ether. The product was isolated dried to get pale yellowish solid product. Yield: 26.5 gm HPLC purity: 92.7%
CLIP
March 6, 2015
Release
The U.S. Food and Drug Administration today approved Cresemba (isavuconazonium sulfate), a new antifungal drug product used to treat adults with invasive aspergillosis and invasive mucormycosis, rare but serious infections.
Aspergillosis is a fungal infection caused by Aspergillus species, and mucormycosis is caused by the Mucorales fungi. These infections occur most often in people with weakened immune systems.
Cresemba belongs to a class of drugs called azole antifungal agents, which target the cell wall of a fungus. Cresemba is available in oral and intravenous formulations.
“Today’s approval provides a new treatment option for patients with serious fungal infections and underscores the importance of having available safe and effective antifungal drugs,” said Edward Cox, M.D., M.P.H, director of the Office of Antimicrobial Products in the FDA’s Center for Drug Evaluation and Research.
Cresemba is the sixth approved antibacterial or antifungal drug product designated as a Qualified Infectious Disease Product (QIDP). This designation is given to antibacterial or antifungal drug products that treat serious or life-threatening infections under the Generating Antibiotic Incentives Now (GAIN) title of the FDA Safety and Innovation Act.
As part of its QIDP designation, Cresemba was given priority review, which provides an expedited review of the drug’s application. The QIDP designation also qualifies Cresemba for an additional five years of marketing exclusivity to be added to certain exclusivity periods already provided by the Food, Drug, and Cosmetic Act. As these types of fungal infections are rare, the FDA also granted Cresemba orphan drug designations for invasive aspergillosis and invasive mucormycosis.
The approval of Cresemba to treat invasive aspergillosis was based on a clinical trial involving 516 participants randomly assigned to receive either Cresemba or voriconazole, another drug approved to treat invasive aspergillosis. Cresemba’s approval to treat invasive mucormycosis was based on a single-arm clinical trial involving 37 participants treated with Cresemba and compared with the natural disease progression associated with untreated mucormycosis. Both studies showed Cresemba was safe and effective in treating these serious fungal infections.
The most common side effects associated with Cresemba include nausea, vomiting, diarrhea, headache, abnormal liver blood tests, low potassium levels in the blood (hypokalemia), constipation, shortness of breath (dyspnea), coughing and tissue swelling (peripheral edema). Cresemba may also cause serious side effects including liver problems, infusion reactions and severe allergic and skin reactions.
Cresemba is marketed by Astellas Pharma US, Inc., based in Northbrook, Illinois.

The active substance is isavuconazonium sulfate, a highly water soluble pro-drug of the active triazole isavuconazole. The chemical name of the active substance isavuconazonium sulfate is 1-{(2R,3R)-3-[4-(4-cyanophenyl)-1,3- thiazol-2-yl]-2-(2,5-difluoro-phenyl)-2-hydroxybutyl}-4-[(1RS)-1-({methyl[3-({[(methylamino)acetyl] oxy}methyl) pyridin-2-yl]carbamoyl}oxy)ethyl]-1H-1,2,4-triazol-4-ium monosulfate (IUPAC), corresponding to the molecular formula C35H35F2N8O5S·HSO4 and has a relative molecular mass of 814.84 g/mol. The relative molecular mass of isavuconazole is 437.47. The active substance has the following structure:
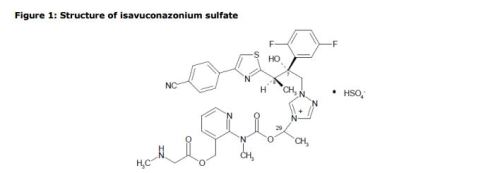
The structure of the active substance has been confirmed by elemental analysis, mass spectrometry, UV, IR, 1H-, 13C- and 19F-NMR spectrometry, and single crystal X-ray analysis, all of which support the chemical structure. It appears as a white, amorphous, hygroscopic powder. It is very soluble in water and over the pH range 1-7. It is also very soluble in methanol and sparingly soluble in ethanol. Two pKa values have been found and calculated to be 2.0 and 7.3. Its logPoct/wat calculated by software is 1.31.
Isavuconazonium sulfate has three chiral centres. The stereochemistry of the active substance is introduced by one of the starting materials which is controlled by appropriate specification. The two centres, C7 and C8 in the isavuconazole moiety and in an intermediate of the active substance, have R configuration. The third chiral centre, C29, is not located on isavuconazole moiety and has both the R and S configurations. The nondefined stereo centre at C29 has been found in all batches produced so far to be racemic. Erosion of stereochemical purity has not been observed in the current process. The active substance is a mixture of two epimers of C29.
An enantiomer of drug substance was identified as C7 (S), C8 (S) and C29 (R/S) structure. The control of the stereochemistry of isavuconazonium sulfate is performed by chiral HPLC on the active substance and its two precursors. Subsequent intermediates are also controlled by relevant specification in the corresponding steps. Two crystal forms have been observed by recrystallisation studies. However the manufacturing process as described yields amorphous form only.
Two different salt forms of isavuconazonuium (chloride and sulfate) were identified during development. The sulfate salt was selected for further development. A polymorph screening study was also performed. None of the investigated salts could be obtained in crystalline Form………http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Public_assessment_report/human/002734/WC500196130.pdf

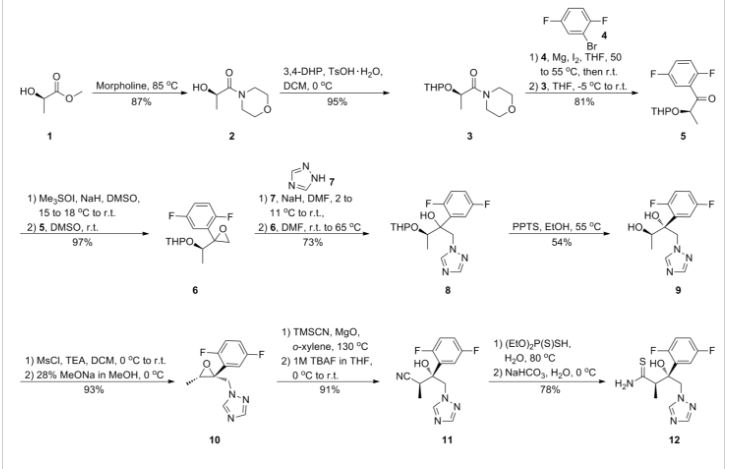
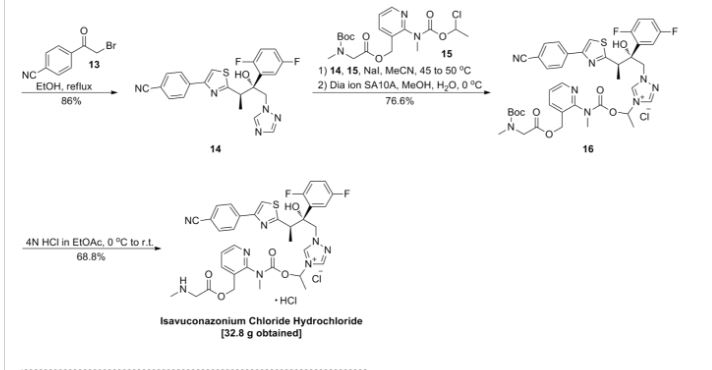
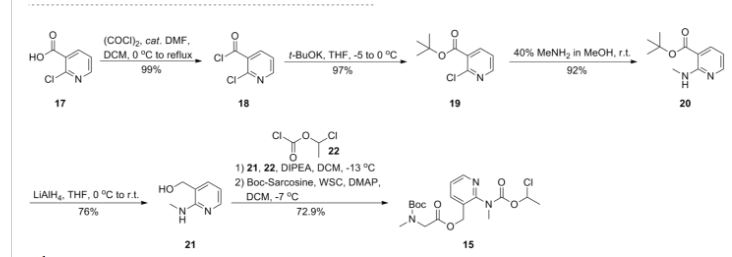
Clip
Isavuconazonium (Cresemba ) is a water-soluble prodrug of the triazole antifungal isavuconazole (BAL4815), a 14-a-demethylase inhibitor, under development byBasilea Pharmaceutica International Ltd and Astellas Pharma Inc. Isavuconazonium, in both its intravenous and oral formulations, was approved for the treatment of invasive aspergillosis and invasive mucormycosis (formerly termed zygomycosis) in the US in March 2015. Isavuconazonium is under regulatory review in the EU for invasive aspergillosis and mucormycosis. It is also under phase III development worldwide for the treatment of invasive candidiasis and candidaemia. This article summarizes the milestones in the development of isavuconazonium leading to the first approval for invasive spergillosis and mucormycosis.
Introduction
The availability of both an intravenous (IV) and an oral formulation of isavuconazonium (Cresemba ), as a result of its water solubility, rapid hydrolysis to the active entity isavuconazole and very high oral bioavailability, provides maximum flexibility to clinicians for treating seriously ill patients with invasive fungal infections [1]. Both the IV and oral formulations have been approved by the US Food and Drug Administration (FDA) to treat adults with invasive aspergillosis and invasive mucormycosis [2]. The recommended dosages of each formulation are identical, consisting of loading doses of 372 mg (equivalent to 200 mg of isavuconazole) every eight hours for six doses, followed by maintenance therapy with 372 mg administered once daily [3]. The Qualified Infectious Disease Product (QIDP) designation of the drug with priority review status by the FDA isavuconazonium in the US provided and a five year extension of market exclusivity from launch. Owing to the rarity of the approved infections,
isavuconazonium was also granted orphan drug designation by the FDA for these indications [2]. It has also been granted orphan drug and QIDP designation in the US for the treatment of invasive candidiasis [4]. In July 2014, Basilea Pharmaceutica International Ltd submitted a Marketing Authorization Application to the European Medicines Agency (EMA) for isavuconazonium in the treatment of invasive aspergillosis and invasive mucormycosis, indications for which the EMA has granted isavuconazonium orphan designation [5, 6]. Isavuconazonium is under phase III development in many countries worldwide for the treatment of invasive candidiasis and candidaemia.
1.1 Company agreements
In 2010, Basilea Pharmaceutica International Ltd (a spinoff from Roche, founded in 2000) entered into a licence agreement with Astellas Pharma Inc in which the latter would co-develop and co-promote isavuconazonium worldwide, including an option for Japan. In return for milestone payments, Astellas Pharma was granted an exclusive right to commercialize isavuconazonium, while Basilea Pharmaceutica retained an option to co-promote the drug in the US, Canada, major European countries and China [7]. The companies amended their agreement in 2014, making Astellas Pharma responsible for all regulatory filings, commercialization and manufacturing of isavuconazonium in the US and Canada. Basilea Pharmaceutica waived its right to co-promote the product in the US and Canada, in order to assume all rights in the rest of the world [8]. However, Astellas Pharma remains as sponsor of the multinational, phase III ACTIVE trial in patients with invasive candidiasis.
2 Scientific Summary
Isavuconazonium (as the sulphate; BAL 8557) is a prodrug that is rapidly hydrolyzed by esterases (mainly butylcholinesterase) in plasma into the active moiety isavuconazole
(BAL 4815) and an inactive cleavage product (BAL 8728).
References
1. Falci DR, Pasqualotto AC. Profile of isavuconazole and its potential in the treatment of severe invasive fungal infections. Infect Drug Resist. 2013;6:163–74.
2. US Food and Drug Administration. FDA approves new antifungal drug Cresemba. 2015. http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm437106.htm. Accessed 12 Mar 2015.
3. US Food and Drug Administration. Cresemba (isavuconazonium sulfate): US prescribing information. 2015. http://www.accessdata.fda.gov/drugsatfda_docs/label/2015/207500Orig1s000lbl.pdf. Accessed 18 Mar 2015.
4. Astellas Pharma US Inc. FDA grants Astellas Qualified Infectious Disease Product designation for isavuconazole for the treatment of invasive candidiasis (media release). 2014. http://newsroom astellas.us/2014-07-16-FDA-Grants-Astellas-Qualified-Infectious-Disease-Product-Designation-for-Isavuconazole-for-the-Treatmentof-Invasive-Candidiasis.
5. European Medicines Agency. Public summary of opinion on orphan designation: isavuconazonium sulfate for the treatment of invasive aspergillosis. 2014. http://www.ema.europa.eu/docs/en_GB/document_library/Orphan_designation/2014/07/WC500169890.pdf. Accessed 18 Mar 2015.
European Medicines Agency. Public summary of opinion on orphan designation: isavuconazonium sulfate for the treatment of mucormycosis. 2014. http://www.ema.europa.eu/docs/en_GB/document_library/Orphan_designation/2014/07/WC500169714.pdf. Accessed 18 Mar 2015.
7. Basilea Pharmaceutica. Basilea announces global partnership with Astellas for its antifungal isavuconazole (media release).2010. http://www.basilea.com/News-and-Media/Basilea-announcesglobal-partnership-with-Astellas-for-its-antifungal-isavuconazole/343.
8. Basilea Pharmaceutica. Basilea swaps its isavuconazole North American co-promote rights for full isavuconazole rights outside of North America (media release). 2014. http://www.basilea.com/News-and-Media/Basilea-swaps-its-isavuconazole-North-Americanco-promote-rights-for-full-isavuconazole-rights-outside-
CLIP
http://www.jpharmsci.org/article/S0022-3549(15)00035-0/pdf
A CLIP
http://www.accessdata.fda.gov/drugsatfda_docs/nda/2015/207500Orig1207501Orig1s000ChemR.pdf
EMA
On 4 July 2014 orphan designation (EU/3/14/1284) was granted by the European Commission to Basilea Medical Ltd, United Kingdom, for isavuconazonium sulfate for the treatment of invasive aspergillosis.
Update: isavuconazonium sulfate (Cresemba) has been authorised in the EU since 15 October 2015. Cresemba is indicated in adults for the treatment of invasive aspergillosis.
Consideration should be given to official guidance on the appropriate use of antifungal agents.
The active substance is isavuconazonium sulfate, a highly water soluble pro-drug of the active triazole isavuconazole. The chemical name of the active substance isavuconazonium sulfate is 1-{(2R,3R)-3-[4-(4-cyanophenyl)-1,3- thiazol-2-yl]-2-(2,5-difluoro-phenyl)-2-hydroxybutyl}-4-[(1RS)-1-({methyl[3-({[(methylamino)acetyl] oxy}methyl) pyridin-2-yl]carbamoyl}oxy)ethyl]-1H-1,2,4-triazol-4-ium monosulfate (IUPAC), corresponding to the molecular formula C35H35F2N8O5S·HSO4 and has a relative molecular mass of 814.84 g/mol. The relative molecular mass of isavuconazole is 437.47. The active substance has the following structure
It appears as a white, amorphous, hygroscopic powder. It is very soluble in water and over the pH range 1-7. It is also very soluble in methanol and sparingly soluble in ethanol. Two pKa values have been found and calculated to be 2.0 and 7.3. Its logPoct/wat calculated by software is 1.31.
Isavuconazonium sulfate has three chiral centres. The stereochemistry of the active substance is introduced by one of the starting materials which is controlled by appropriate specification. The two centres, C7 and C8 in the isavuconazole moiety and in an intermediate of the active substance, have R configuration. The third chiral centre, C29, is not located on isavuconazole moiety and has both the R and S configurations. The nondefined stereo centre at C29 has been found in all batches produced so far to be racemic. Erosion of stereochemical purity has not been observed in the current process. The active substance is a mixture of two epimers of C29. An enantiomer of drug substance was identified as C7 (S), C8 (S) and C29 (R/S) structure. The control of the stereochemistry of isavuconazonium sulfate is performed by chiral HPLC on the active substance and its two precursors.
FDA Orange Book Patents
US 6812238
US 7459561
| FDA ORANGE BOOK PATENTS: 1 OF 2 | |
|---|---|
| Patent | 7459561 |
| Expiration | Oct 31, 2020 |
| Applicant | ASTELLAS |
| Drug Application | N207500 (Prescription Drug: CRESEMBA. Ingredients: ISAVUCONAZONIUM SULFATE) |
FREE FORM
Isavuconazonium; Isavuconazonium ion; Cresemba; BAL-8557; 742049-41-8;
[2-[1-[1-[(2R,3R)-3-[4-(4-cyanophenyl)-1,3-thiazol-2-yl]-2-(2,5-difluorophenyl)-2-hydroxybutyl]-1,2,4-triazol-4-ium-4-yl]ethoxycarbonyl-methylamino]pyridin-3-yl]methyl 2-(methylamino)acetate
| MOLECULAR FORMULA: | C35H35F2N8O5S+ |
|---|---|
| MOLECULAR WEIGHT: | 717.773 g/mol |
Patent IDDatePatent Title
US20102494262010-09-30STABILIZED PHARMACEUTICAL COMPOSITION
US74595612008-12-02N-substituted carbamoyloxyalkyl-azolium derivativesUS71898582007-03-13N-phenyl substituted carbamoyloxyalkyl-azolium derivatives
US71511822006-12-19Intermediates for N-substituted carbamoyloxyalkyl-azolium derivatives
US68122382004-11-02N-substituted carbamoyloxyalkyl-azolium derivatives
REF
http://www.drugbank.ca/drugs/DB06636
////////// BAL 8557, BAL-8557-002, CRESEMBA, ISAVUCONAZONIUM SULFATE, QIDP designation, Cresemba , priority review, FDA 2015, EU 2015, BAL8557-002, BCS CLASS I, orphan designation, invasive aspergillosis, invasive mucormycosis, RO-0098557 , AK-1820, fast track designation, QIDP, 946075-13-4
CC(C1=NC(=CS1)C2=CC=C(C=C2)C#N)C(CN3C=[N+](C=N3)C(C)OC(=O)N(C)C4=C(C=CC=N4)COC(=O)CNC)(C5=C(C=CC(=C5)F)F)O
CC(C1=NC(=CS1)C2=CC=C(C=C2)C#N)C(CN3C=[N+](C=N3)C(C)OC(=O)N(C)C4=C(C=CC=N4)COC(=O)CNC)(C5=C(C=CC(=C5)F)F)O.OS(=O)(=O)[O-]
UPDATE NEW PATENT
WOCKHARDT, WO 2016016766, ISAVUCONAZONIUM SULPHATE, NEW PATENT


(WO2016016766) A PROCESS FOR THE PREPARATION OF ISAVUCONAZONIUM OR ITS SALT THEREOF
WOCKHARDT LIMITED [IN/IN]; D-4, MIDC Area, Chikalthana, Aurangabad 431006 (IN)
KHUNT, Rupesh Chhaganbhai; (IN).
RAFEEQ, Mohammad; (IN).
MERWADE, Arvind Yekanathsa; (IN).
DEO, Keshav; (IN)
The present invention relates to a process for the preparation of stable Isavuconazonium or its salt thereof. In particular of the present invention relates to process for the preparing of isavuconazonium sulfate, Isavuconazonium iodide hydrochloride and Boc-protected isavuconazonium iodide has purity more than 90%. The process is directed to preparation of solid amorphous form of isavuconazonium sulfate, isavuconazonium iodide hydrochloride and Boc-protected isavuconazonium iodide. The present invention process of Isavuconazonium or its salt thereof is industrially feasible, simple and cost effective to manufacture of isavuconazonium sulfate with the higher purity and better yield.
Isavuconazonium sulfate is chemically known l-[[N-methyl-N-3-[(methylamino) acetoxymethyl]pyridin-2-yl] carbamoyloxy]ethyl-l-[(2R,3R)-2-(2,5-difluorophenyl)-2-hydroxy-3-[4-(4-cyanophenyl)thiazol-2-yl]butyl]-lH-[l,2,4]-triazo-4-ium Sulfate and is structurally represented by formula (I):


Formula I
Isavuconazonium sulfate (BAL8557) is indicated for the treatment of antifungal infection. Isavuconazonium sulfate is a prodrug of Isavuconazole (BAL4815), which is chemically known 4-{2-[(lR,2R)-(2,5-Difluorophenyl)-2-hydroxy-l-methyl-3-(lH-l ,2,4-triazol-l-yl)propyl]-l ,3-thiazol-4-yl}benzonitrile compound of Formula II


Formula II
US Ppatent No. 6,812,238 (referred to herein as ‘238); 7,189,858 (referred to herein as ‘858); 7,459,561 (referred to herein as ‘561) describe Isavuconazonium and its process for the preparation thereof.
The US Pat. ‘238 patent describes the process of preparation of Isavuconazonium chloride hydrochloride.
The US Pat. ‘238 described the process for the Isavuconazonium chloride hydrochloride, involves the condensation of Isavuconazole and [N-methyl-N-3((tert-butoxycarbonyl methylamino) acetoxymethyl) pyridine-2-yl]carbamic acid 1 -chloro-ethyl ester. The prior art reported process require almost 15-16 hours, whereas the present invention process requires only 8-10 hours. Inter alia prior art reported process requires too many step to prepare isavuconazonium sulfate, whereas the present invention process requires fewer steps.
Moreover, the US Pat. ‘238 describes the process for the preparation Isavuconazonium hydrochloride, which may be used as the key intermediate for the synthesis of isavuconazonium sulfate, compound of formula I. There are several drawbacks in the said process, which includes the use of anionic resin to prepare Isavuconazonium chloride hydrochloride, consequently it requires multiple time lyophilization, which makes the said prior art process industrially, not feasible.
The inventors of the present invention surprisingly found that Isavuconazonium or a pharmaceutically acceptable salt thereof in yield and purity could be prepared by using substantially pure intermediates in suitable solvent.
Thus, an object of the present invention is to provide simple, cost effective and industrially feasible processes for manufacture of isavuconazonium sulfate. Inventors of the present invention surprisingly found that isavuconazonium sulfate prepared from isavuconazonium iodide hydrochloride, provides enhanced yield as well as purity.
The process of the present invention is depicted in the following scheme:


Formula I
Formula-IA
The present invention is further illustrated by the following example, which does not limit the scope of the invention. Certain modifications and equivalents will be apparent to those skilled in the art and are intended to be included within the scope of the present application.
Examples
Example-1: Synthesis of l-[[N-methyl-N-3-[(t-butoxycarbonylmethylamino) acetoxymethyl]pyridin-2-yl]carbamoyloxy]ethyl-l-[(2R,3R)-2-(2,5-difluorophenyl)-2-hydroxy-3 – [4-(4-cyanophenyl)thiazol-2-yl]butyl] – 1 H-[ 1 ,2,4] -triazo-4-ium iodide
Isavuconazole (20 g) and [N-methyl-N-3((tert-butoxycarbonylmethylamino)acetoxy methyl)pyridine-2-yl]carbamic acid 1 -chloro-ethyl ester (24.7 g) were dissolved in acetonitrile (200ml). The reaction mixture was stirred to add potassium iodide (9.9 g). The reaction mixture was stirred at 47-50°C for 10-13 hour. The reaction mixture was cooled to room temperature. The reaction mass was filtered through celite bed and washed acetonitrile. Residue was concentrated under reduced pressure to give the crude solid product (47.7 g). The crude product was purified by column chromatography to get its pure iodide form (36.5 g).
Yield: 84.5 %
HPLC Purity: 87%
Mass: m/z 817.4 (M- 1)+
Example-2: Synthesis of l-[[N-methyl-N-3-[(methylamino)acetoxymethyl]pyridin-2-yl] carbamoyloxy]ethyl-l-[(2R,3R)-2-(2,5-difluorophenyl)-2-hydroxy-3-[4-(4-cyanophenyl) thiazol-2-yl]butyl]-lH-[l ,2,4]-triazo-4-ium iodide hydrochloride
l-[[N-methyl-N-3-[(t-butoxycarbonylmethylamino)acetoxymethyl]pyridin-2-yl] carbamoyloxy]ethyl-l-[(2R,3R)-2-(2,5-difluorophenyl)-2-hydroxy-3-[4-(4-cyanophenyl) thiazol-2-yl]butyl]-lH-[l ,2,4]-triazo-4-ium iodide (36.5 g) was dissolved in ethyl acetate (600 ml). The reaction mixture was cooled to -5 to 0 °C. The ethyl acetate hydrochloride (150 ml) solution was added to reaction mixture. The reaction mixture was stirred for 4-5 hours at room temperature. The reaction mixture was filtered and obtained solid residue washed with ethyl acetate. The solid dried under vacuum at room temperature for 20-24 hrs to give 32.0 gm solid.
Yield: 93 %
HPLC Purity: 86%
Mass: m/z 717.3 (M-HC1- 1)
Example-3: Preparation of Strong anion exchange resin (Sulfate).
Indion GS-300 was treated with aqueous sulfate anion solution and then washed with DM water. It is directly used for sulfate salt.
Example-4: Synthesis of l-[[N-methyl-N-3-[(methylamino)acetoxymethyl]pyridin-2-yl] carbamoyloxy]ethyl-l-[(2R,3R)-2-(2,5-difluorophenyl)-2-hydroxy-3-[4-(4-cyanophenyl) thiazol-2-yl]butyl]-lH-[l ,2,4]-triazo-4-ium Sulfate
Dissolved 10.0 g l-[[N-methyl-N-3-[(methylamino)acetoxymethyl]pyridin-2-yl] carbamoyloxy]ethyl-l-[(2R,3R)-2-(2,5-difluorophenyl)-2-hydroxy-3-[4-(4-cyanophenyl) thiazol-2-yl]butyl]-lH-[l ,2,4]-triazo-4-ium iodide hydrochloride in 200 ml deminerahzed water and 30 ml methanol. The solution was cooled to about 0 to 5°C. The strong anion exchange resin (sulfate) was added to the cooled solution. The reaction mixture was stirred to about 60-80 minutes. The reaction was filtered and washed with 50ml of demineralized water and methylene chloride. The aqueous layer was lyophilized to obtain
(8.0 g) white solid.
Yield: 93 %
HPLC Purity: > 90%
Mass: m/z 717.4 (M- HS04) +
PATENT
CN 105288648
PATENT
CN 106883226
https://patents.google.com/patent/CN106883226A/en
PATENT
CN 107982221
PAPER
| Title: Introduction of New Drugs Approved by the U.S. FDA in 2015 |
| Author: Ma Shuai; Wenying Ling; Zhou Weicheng; |
| Source: China Pharmaceutical Industry |
| Publisher: Tongfangzhiwang Beijing Technology Co., Ltd. |
| Year of publication: |
| DOI code: 10.16522/j.cnki.cjph.2016.01.022 |
| Registration Time: 2016-02-19 02:04:15 |
///////////////
Efmoroctocog alfa, エフモロクトコグアルファ;

(Heavy chain)
ATRRYYLGAV ELSWDYMQSD LGELPVDARF PPRVPKSFPF NTSVVYKKTL FVEFTDHLFN
IAKPRPPWMG LLGPTIQAEV YDTVVITLKN MASHPVSLHA VGVSYWKASE GAEYDDQTSQ
REKEDDKVFP GGSHTYVWQV LKENGPMASD PLCLTYSYLS HVDLVKDLNS GLIGALLVCR
EGSLAKEKTQ TLHKFILLFA VFDEGKSWHS ETKNSLMQDR DAASARAWPK MHTVNGYVNR
SLPGLIGCHR KSVYWHVIGM GTTPEVHSIF LEGHTFLVRN HRQASLEISP ITFLTAQTLL
MDLGQFLLFC HISSHQHDGM EAYVKVDSCP EEPQLRMKNN EEAEDYDDDL TDSEMDVVRF
DDDNSPSFIQ IRSVAKKHPK TWVHYIAAEE EDWDYAPLVL APDDRSYKSQ YLNNGPQRIG
RKYKKVRFMA YTDETFKTRE AIQHESGILG PLLYGEVGDT LLIIFKNQAS RPYNIYPHGI
TDVRPLYSRR LPKGVKHLKD FPILPGEIFK YKWTVTVEDG PTKSDPRCLT RYYSSFVNME
RDLASGLIGP LLICYKESVD QRGNQIMSDK RNVILFSVFD ENRSWYLTEN IQRFLPNPAG
VQLEDPEFQA SNIMHSINGY VFDSLQLSVC LHEVAYWYIL SIGAQTDFLS VFFSGYTFKH
KMVYEDTLTL FPFSGETVFM SMENPGLWIL GCHNSDFRNR GMTALLKVSS CDKNTGDYYE
DSYEDISAYL LSKNNAIEPR SFSQNPPVLK RHQREITRTT LQSDQEEIDY DDTISVEMKK
EDFDIYDEDE NQSPRSFQKK TRHYFIAAVE RLWDYGMSSS PHVLRNRAQS GSVPQFKKVV
FQEFTDGSFT QPLYRGELNE HLGLLGPYIR AEVEDNIMVT FRNQASRPYS FYSSLISYEE
DQRQGAEPRK NFVKPNETKT YFWKVQHHMA PTKDEFDCKA WAYFSDVDLE KDVHSGLIGP
LLVCHTNTLN PAHGRQVTVQ EFALFFTIFD ETKSWYFTEN MERNCRAPCN IQMEDPTFKE
NYRFHAINGY IMDTLPGLVM AQDQRIRWYL LSMGSNENIH SIHFSGHVFT VRKKEEYKMA
LYNLYPGVFE TVEMLPSKAG IWRVECLIGE HLHAGMSTLF LVYSNKCQTP LGMASGHIRD
FQITASGQYG QWAPKLARLH YSGSINAWST KEPFSWIKVD LLAPMIIHGI KTQGARQKFS
SLYISQFIIM YSLDGKKWQT YRGNSTGTLM VFFGNVDSSG IKHNIFNPPI IARYIRLHPT
HYSIRSTLRM ELMGCDLNSC SMPLGMESKA ISDAQITASS YFTNMFATWS PSKARLHLQG
RSNAWRPQVN NPKEWLQVDF QKTMKVTGVT TQGVKSLLTS MYVKEFLISS SQDGHQWTLF
FQNGKVKVFQ GNQDSFTPVV NSLDPPLLTR YLRIHPQSWV HQIALRMEVL GCEAQDLYDK
THTCPPCPAP ELLGGPSVFL FPPKPKDTLM ISRTPEVTCV VVDVSHEDPE VKFNWYVDGV
EVHNAKTKPR EEQYNSTYRV VSVLTVLHQD WLNGKEYKCK VSNKALPAPI EKTISKAKGQ
PREPQVYTLP PSRDELTKNQ VSLTCLVKGF YPSDIAVEWE SNGQPENNYK TTPPVLDSDG
SFFLYSKLTV DKSRWQQGNV FSCSVMHEAL HNHYTQKSLS LSPG
(Lignt chain)
DKTHTCPPCP APELLGGPSV FLFPPKPKDT LMISRTPEVT CVVVDVSHED PEVKFNWYVD
GVEVHNAKTK PREEQYNSTY RVVSVLTVLH QDWLNGKEYK CKVSNKALPA PIEKTISKAK
GQPREPQVYT LPPSRDELTK NQVSLTCLVK GFYPSDIAVE WESNGQPENN YKTTPPVLDS
DGSFFLYSKL TVDKSRWQQG NVFSCSVMHE ALHNHYTQKS LSLSPG
(disulfide bridges: H153-H179, H248-H329, H528-H554, H630-H711, H938-H964, H1005-H1009, H1127-H1275, H1280-H1432, H1444-L6, H1447-L9, H1479-H1539, H1585-H1643, L41-L101, L147-L205)
Efmoroctocog alfa
Protein chemical formulaC9736H14863N2591O2855S78
Protein average weight220000.0 Da (Apparent, B-domain deleted)
Peptide
CAS: 1270012-79-7
エフモロクトコグアルファ;
| 2015/11/19 ema APPROVED elocta |




Efmoroctocog alfa is a fully recombinant factor VIII-Fc fusion protein (rFVIIIFc) with an extended half-life compared with conventional factor VIII (FVIII) preparations, including recombinant FVIII (rFVIII) products such as Moroctocog alfa[1]. It is an antihemorrhagic agent used in replacement therapy for patients with haemophilia A (congenital factor VIII deficiency). It is suitable for all age groups. Haemophilia A is a rare bleeding disorder associated with a slow clotting process caused by the deficiency of factor VIII. Patients with this disorder are more susceptible to recurrent bleeding episodes and excessive bleeding following minor traumatic injuries or surgical procedures [1]. Prophylactic treatment may dramatically improve the management of severe haemophilia A in the future by reducing joint bleeding and other hemorrhages that cause chronic pain and disability to patients [1, 2]. Prophylaxis has also shown to reduce the formation of neutralizing anti-FVIII antibodies, or inhibitors [2].
Factor VIII is a blood coagulant factor involved in the intrinsic pathway to form fibrin, or a blood clot. Efmoroctocog alfa is a first commercially available rFVIII-Fc fusion protein (rFVIIIFc) where the conjugated molecule of rFVIII to polyethylene glycol is covalently fused to the dimeric Fc domain of human immunoglobulin G1, a long-lived plasma protein [FDA Label]. The B domain of factor VIII is deleted. In animal models of haemophilia, efmoroctocog alfa demonstrated an approximately two-fold longer t½ than commercially available rFVIII products [1].
Other drug products with similar structure and function to Efmoroctocog alfa include Moroctocog alfa, which is produced by recombinant DNA technology and is identical in sequence to endogenously produced Factor VIII, but does not contain the B-domain, which has no known biological function, and Antihemophilic factor human, which is purified endogenous Factor VIII from human pooled blood and contains both A- and B-subunits.
It is commonly marketed as Elocta or Eloctate for intravenous injection. To date, no confirmed inhibitory autoantibodies were seen in previously treated patients included in clinical studies and treatment-emergent adverse events were generally consistent with those expected in the patient populations being studied [1]. The extended half-life of efmoroctocog alfa provides several clinical benefits for patients, including reduced frequency of injections required and improved adherence to prophylaxis [1].
Haemophilia A is an inherited sex-linked disorder of blood coagulation in which affected males (very rarely females) do not produce functional coagulation FVIII in sufficient quantities to achieve satisfactory haemostasis. The incidence of congenital haemophilia A is approximately 1 in 10,000 births. Disease severity is classified according to the level of FVIII activity (% of normal) as mild (>5% to <40%), moderate (1% to 5%) or severe (<1%). This deficiency in FVIII predisposes patients with haemophilia A to recurrent bleeding episodes in joints, muscles or internal organs, either spontaneously or as a result of accidental or surgical trauma. Without adequate treatment these repeated haemarthroses and haematomas lead to long-term sequelae with severe disability. Other less frequent, but more severe bleeding sites, are the central nervous system, the urinary or gastrointestinal tract, eyes and the retro-peritoneum. Patients with haemophilia A are at high risk of developing major and life-threatening bleeds after surgical procedures, even after minor procedures such as tooth extraction. The development of cryoprecipitate and subsequently FVIII concentrates, obtained by fractionation of human plasma, provided replacement FVIII and greatly improved clinical management and life expectancy of patients with haemophilia A. Current treatment approaches focus on either prophylactic or on demand factor replacement therapy with plasma-derived FVIII or recombinant FVIII products. In the short term, prophylaxis can prevent spontaneous bleeding and in the long term, prophylaxis can prevent bleeding into joints that will eventually lead to debilitating arthropathy. Prophylaxis with FVIII concentrates is currently the preferred treatment regimen for patients with severe haemophilia A, especially in very young patients. The majority of patients receiving prophylaxis are treated 3-times weekly or every other day at a dose of 25–40 international units (IU)/kg (or 15–25 IU/kg in an intermediate dose regimen), although an escalating dose regimen is also used. However, on-demand treatment is still the predominant replacement approach in many countries. The most serious complication in the treatment of haemophilia A is the development of neutralising antibodies (inhibitors) against FVIII, rendering the patient resistant to replacement therapy and thereby increasing the risk of unmanageable bleeding, particularly arthropathy, and disability.
ELOCTA (efmoroctocog alfa) is a recombinant human coagulation factor VIII Fc fusion protein (rFVIIIFc) consisting of B-domain deleted FVIII covalently attached to the Fc domain of human immunoglobulin G1 (IgG1) thus aiming at prolongation of plasma half-life. It has been developed as a long-acting version of recombinant FVIII (rFVIII) for the control and prevention of bleeding episodes, routine prophylaxis, and perioperative management (surgical prophylaxis) in individuals with hemophilia A. ELOCTA is formulated as powder for intravenous administration in a single-use vial. Each single-use vial contains nominally 250, 500, 750, 1000, 1500, 2000, or 3000 International Units (IU) of rFVIIIFc for reconstitution with a solvent (Sterile Water for Injections), which is provided in a pre-filled syringe. In 2013, national scientific advice was sought from the United Kingdom Medicines and Healthcare Products Regulatory Agency (MHRA), Swedish Medicinal Products Agency, and German Paul-Ehrlich-Institute. No substantial deviations from the advices provided could be identified. On 2 April 2014, the Paediatric Committee (PDCO) of the European Medicines Agency adopted a favourable opinion on the modification of an agreed paediatric investigation plan (PIP) (P/0077/2014) and a partially completed compliance procedure was finalised on 16-18 July 2014 (EMEA-C1-001114-PIP01-10-MO2). Completed studies, Study 997HA301 and Study 8HA02PED, and the initiation of Study 8HA01EXT are considered compliant with EMA Decision P/0077/2014.
The active substance of ELOCTA, efmoroctocog alfa, is a recombinant human coagulation factor VIII, Fc fusion protein (rFVIIIFc) comprising B-domain deleted (BDD) human FVIII covalently linked to the Fc domain of human immunoglobulin G1(IgG1). It has been developed as a long-acting version of recombinant FVIII (rFVIII). ELOCTA is formulated as a sterile, non-pyrogenic, preservative-free, lyophilized, white to off-white powder to cake for intravenous administration in a single-use vial. Each single-use vial contains nominally 250, 500, 750, 1000, 1500, 2000, or 3000 International Units (IU) of rFVIIIFc for reconstitution with liquid diluent (Sterile Water for Injection), which is provided in a pre-filled syringe. The finished medicinal product consists of a package containing a rFVIIIFc drug product vial, a pre-filled diluent (SWFI) syringe and medical devices (a plunger rod, a vial adapter (used as a transfer device during reconstitution), an infusion set, alcohol swabs, plasters and gauze pad for intravenous administration).
Structure The active substance of Elocta, efmoroctocog alfa, is a recombinant human coagulation factor VIII, Fc fusion protein (rFVIIIFc) comprised of a single molecule of B-domain deleted human Factor VIII (BDD FVIII) fused to the dimeric Fc region of human IgG1 with no intervening linker sequence.
The rFVIIIFc protein has a molecular weight of approximately 220 kDa. rFVIIIFc is synthesized as 2 polypeptide chains, one chain consisting of BDD FVIII fused to the N-terminal of human IgG1 Fc domain the other chain consisting of the same Fc region alone. The two subunits of rFVIIIFc, FVIIIFc single chain and Fc single chain, are associated through disulfide bonds in the hinge region of Fc as well as through extensive noncovalent interactions between the Fc fragments.
Characterisation rFVIIIFc was extensively characterised by physicochemical methods in accordance with guideline ICH Q6B. The structural characterisation and the physicochemical properties confirmed the expected properties for a recombinant FVIIIFc product. In general, the characterization performed was considered appropriate for this complex fusion molecule. The panel of tests was comprehensive and covered most of its structural and functional attributes. The comparability between representative batches from development and commercial manufacture (including process validation batches) as well as with rFVIIIFc reference materials was demonstrated. The biological activity was analysed by the FVIII one stage clotting assay (activated partial thromboplastin time (aPTT)), the FVIII chromogenic assay and the FcRn binding assay. Additional in vitro functional tests were performed comprising the binding to von Willebrand factor and the generation of Factor Xa. Since it is anticipated that the potency of modified products measured by the one stage clotting assay (aPTT) may be dependent on the choice of the aPTT reagent, the ISTH recommends for all new FVIII products to perform a study including assay variations (different aPTT reagents) for FVIII testing when using the coagulation assay. Respective studies were provided by the Applicant in Module 5 (no significant dependence on the aPTT reagent was observed). REF 3
AUSTRALIA REF 4
Submission details Type of submission: New biological entity Decision: Approved Date of decision: 18 June 2014 Active ingredient: Efmoroctocog alfa (rhu2)3
Product name: Eloctate Sponsor’s name and address: Biogen Idec Australia Pty Ltd Suite 1, Level 5 123 Epping Rd North Ryde, NSW 2113 Dose form: Powder for injection and diluent Strengths: 250 international units (IU), 500 IU, 750 IU, 1000 IU, 1500 IU, 2000 IU and 3000 IU Containers: Type I glass vial (powder) and pre-filled syringe (diluent) Pack size: Single Approved therapeutic use: Eloctate is a long-acting antihaemophilic factor (recombinant) indicated in adults and children ( ≥ 12 years) with haemophilia A (congenital factor VIII deficiency) for: · control and prevention of bleeding episodes · routine prophylaxis to prevent or reduce the frequency of bleeding episodes · perioperative management (surgical prophylaxis) Eloctate does not contain von Willebrand factor, and therefore is not indicated in patients with von Willebrand’s disease. Route of administration: Intravenous (IV) infusion Dosage: Refer to the Product Information (PI; Attachment 1) ARTG numbers: 210521 (250 IU), 210519 (500 IU), 210523 (750 IU), 210525 (1000 IU), 210522 (1500 IU), 210524 (2000 IU), 210520 (3000 IU). 2 recombinant human 3 The ingredient name at the time of submission and registration was Efraloctocog alfa, The name was subsequently changed on 20 February 2015 to harmonise to the International Non-proprietary Name (INN) Efmoroctocog alfa. The AusPAR document has been amended by replacing the previous name efraloctocog alfa with approved INN efmoroctocog alfa.
- Frampton JE: Efmoroctocog Alfa: A Review in Haemophilia A. Drugs. 2016 Sep;76(13):1281-1291. doi: 10.1007/s40265-016-0622-z. [PubMed:27487799]
- Tiede A: Half-life extended factor VIII for the treatment of hemophilia A. J Thromb Haemost. 2015 Jun;13 Suppl 1:S176-9. doi: 10.1111/jth.12929. [PubMed:26149020]
- http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Public_assessment_report/human/003964/WC500198644.pdf
- https://www.tga.gov.au/sites/default/files/auspar-efmoroctocog-alfa-rhu-150317.pdf
- http://www.who.int/medicines/publications/druginformation/innlists/RL73_pre.pdf
///////////Efmoroctocog alfa, Peptide, ema 2015
EMA approves AstraZeneca’s lesinurad to treat gout patients

EMA approves AstraZeneca’s lesinurad to treat gout patients
British-Swedish drugmaker AstraZeneca has received approval from European Medicines Agency (EMA) for its lesinurad 200mg tablets to treat gout patients. READ AT…..[LINK]

“The company submitted a MAA based on data from the Clear1, Clear2 and Crystal pivotal Phase III combination therapy studies.”
AstraZeneca’s subsidiary Ardea Biosciences carried out Clear1, Clear2 and Crystal trials.


Novartis obtains European approval for Cosentyx to treat psoriasis

Novartis obtains European approval for Cosentyx to treat psoriasis
Swiss drug-maker Novartis has received approval from the European Commission (EC) for its Cosentyx (secukinumab, formerly known as AIN457) to treat moderate-to-severe plaque psoriasis in adults who are candidates for systemic therapy.SEE
 PSORIAIS
PSORIAIS
secukinumab

Secukinumab is a human monoclonal antibody designed for the treatments of uveitis, rheumatoid arthritis, ankylosing spondylitis, and psoriasis. It targets member A from the cytokine family of interleukin 17.[1][2] At present, Novartis Pharma AG, the drug’s developer, plans to market it under the trade name “Cosentyx.” [3] It is highly specific to the human immunoglobulin G1k (IgG1k) subclass.[2]
In July 2014 secukinumab established superiority to placebo and to etanercept for the treatment of chronic plaque psoriasis in Phase III clinical trials.[4] In October 2014, the FDA Dermatologic and Ophthalmic Drugs Advisory Committee unanimously voted to recommend the drug for FDA approval, although this vote in and of itself does not constitute an approval. However, the FDA typically follows recommendations from these committees.[5] In October 2014, Novartis announced that the drug had achieved a primary clinical endpoint in two phase III clinical trials for ankylosing spondylitis.[6] As of 28 October, the relevant FDA committee had not yet responded to these results. In early November 2014, Novartis also released the results of a Phase 3 study on Psoriatic Arthritis that yielded very promising results.[7]

Although the drug was originally intended to treat rheumatoid arthritis, phase II clinical trials for this condition yielded disappointing results.[8] Similarly, while patients in a phase II clinical trial for [psoriatic arthritis] did show improvement over placebo, the improvement did not meet adequate endpoints and Novartis is considering whether to do more research for this condition.[9] Novartis has said that it is targeting approval and release in early 2015 for plaque psoriasis and ankyloding spondylitis indications.
It is also in a phase II clinical trial for Multiple Sclerosis [10] as it has exhibited efficacy in treating experimental autoimmune encephalomyelitis (EAE), an animal model of MS.
CAS registry numbers
- 875356-43-7 (heavy chain)
- 875356-44-8 (light chain)
References
- “Statement On A Nonproprietary Name Adopted By The USAN Council: Secukinumab”. American Medical Association.
- Hueber, W.; Patel, D. D.; Dryja, T.; Wright, A. M.; Koroleva, I.; Bruin, G.; Antoni, C.; Draelos, Z.; Gold, M. H.; Psoriasis Study, P.; Durez, P. P.; Tak, J. J.; Gomez-Reino, C. S.; Rheumatoid Arthritis Study, R. Y.; Foster, C. M.; Kim, N. S.; Samson, D. S.; Falk, D.; Chu, Q. D.; Callanan, K.; Nguyen, A.; Uveitis Study, F.; Rose, K.; Haider, A.; Di Padova, F. (2010). “Effects of AIN457, a Fully Human Antibody to Interleukin-17A, on Psoriasis, Rheumatoid Arthritis, and Uveitis”. Science Translational Medicine 2 (52): 52ra72.doi:10.1126/scitranslmed.3001107. PMID 20926833.
- http://www.medscape.com/viewarticle/835331
- Langley RG, Elewski BE, Mark Lebwohl M, et al., for the ERASURE and FIXTURE Study Groups (July 24, 2014). “Secukinumab in Plaque Psoriasis — Results of Two Phase 3 Trials”. N Engl J Med 371: 326–338. doi:10.1056/NEJMoa1314258.
- committees.http://www.familypracticenews.com/index.php?id=2934&type=98&tx_ttnews=306073[dead link]
- http://inpublic.globenewswire.com/2014/10/23/Novartis+AIN457+secukinumab+meets+primary+endpoint+in+two+Phase+III+studies+in+ankylosing+spondylitis+a+debilitating+joint+condition+of+the+spine+HUG1864939.html
- http://www.medpagetoday.com/MeetingCoverage/ACR/48743
- http://www.medscape.com/viewarticle/806510_6
- http://www.ncbi.nlm.nih.gov/pubmed/23361084
- http://clinicaltrials.gov/show/NCT01874340
| Monoclonal antibody | |
|---|---|
| Type | Whole antibody |
| Source | Human |
| Target | IL17A |
| Clinical data | |
| Legal status |
|
| Identifiers | |
| CAS number | |
| ATC code | L04AC10 |
| DrugBank | DB09029 |
| Synonyms | AIN457 |
| Chemical data | |
| Formula | C6584H10134N1754O2042S44 |
| Molecular mass | 147.94 kDa |

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....