Inidascamine
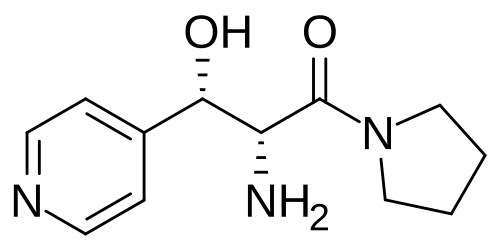
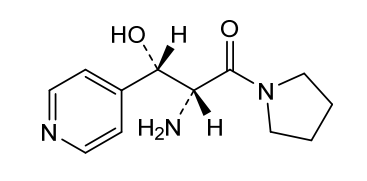
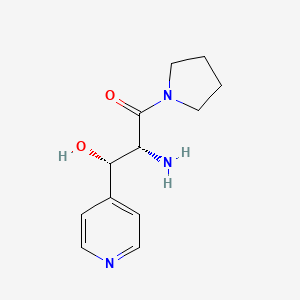
Inidascamine
CAS 903884-71-9
MF C12H17N3O2 MW235.28 g/mol
(-)-(2R,3S)-2-amino-3-hydroxy-3-(pyridin-4-yl)-1-(pyrrolidin-1-yl)propan-1-one
(2R,3S)-2-amino-3-hydroxy-3-pyridin-4-yl-1-pyrrolidin-1-ylpropan-1-one
(2R,3S)-2-amino-3-hydroxy-3-(pyridin-4-yl)-1-(pyrrolidin-1-yl)propan-1-one
schizophrenia, 3LW01V88B7, RL 007
Inidascamine (developmental code name RL-007 or FSV7-007) is an experimental, orally administered drug primarily studied for treating Cognitive Impairment Associated with Schizophrenia (CIAS). Developed jointly by Recognify Life Sciences and atai Life Sciences, the molecule targets the underlying neural mechanisms that restrict verbal learning, memory retention, and mental processing speed in schizophrenia patients
Mechanism of Action
The compound is designed to alter the brain’s complex excitatory and inhibitory balance to produce pro-cognitive effects. It accomplishes this by interacting with three major neurotransmitter systems simultaneously:
- Cholinergic system: Modulates acetylcholine pathways vital for attention and memory.
- Glutamatergic system: Interacts with NMDA/glutamate receptors to influence synaptic plasticity.
- GABAergic system: Targets \(GABA_{B}\) receptors to stabilize neural transmission.
Clinical Status & Current Data
- Phase 2b Trial Results: In July 2025, data from a Phase 2b clinical trial showed that while inidascamine produced numerical improvements in memory and processing speed compared to a placebo, it failed to achieve statistical significance on its primary efficacy endpoint.
- Safety Profile: The drug demonstrated excellent tolerability. It lacked common antipsychotic side effects like heavy sedation, rapid weight gain, or involuntary body movements.
- Commercial Backing: Following the trial shortfall, atai Life Sciences officially deprioritized the asset to shift its primary funding toward its wholly owned pipeline of psychedelic therapies.
Inidascamine (INNTooltip International Nonproprietary Name; developmental code names RL-007, FSV7-007) is an experimental drug which is under development for the treatment of cognitive impairment associated with schizophrenia (CIAS).[1][3][4][5][6][2] It is taken orally.[1][2] The drug is said to act on the cholinergic, NMDA, and GABAB receptor systems.[1][5][2] Inidascamine is being developed by Recognify Life Sciences and atai Life Sciences.[1][3] It was discovered via screening of compounds for effects on synaptic plasticity and cognition.[2] The drug shows structural similarities to phenethylamines and amphetamines.[7]
- A Study to Evaluate RL-007 in the Treatment of Cognitive Impairment Associated With Schizophrenia (CIAS)CTID: NCT05686239Phase: Phase 2Status: CompletedDate: 2025-07-30
- Safety, Biomarker Study of RL-007 in Subjects With SchizophreniaCTID: NCT04822883Phase: Phase 2Status: CompletedDate: 2022-04-27
PAT
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2006081273&_cid=P11-MQ616P-01336-1
OL-threo-2- Amino-3-hvdroxy-3 -(pyridin-4-yl)- 1 -(pyrrolidin- 1 -yDpropan- 1 -one dihydrochloride Compound 22.
Compound 22 was prepared following method E with trans-(4,5-άihydτo-5-(pyridin-4-yl)oxazol-4-yl)(pyrrolidin-l-yl)methanone Compound 19 (0.750 g, 3.07 mmol), hydrochloric acid 37 % (1.0 mL) and methanol (10 mL). After 3.0 h at 50 °C and work-up DL-tAreø-2-amino-3-hydroxy-3-(pyridin-4-yl)-l-(pyrrolidin-l-yl)propan-l-one dihydrochloride Compound 22 was obtained as a white solid (0.935 g, 99 % yield).
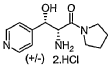
Compound 22
MW: 308.28; Yield: 99 %; White Solid; Mp (°C): 117.0.
1H-NMR (CD3OD3 δ): 1.75-2.03 (m, 4H, 2xCH2), 2.93-3.08 (m, 1H, CHN), 3.32-3.75 (m, 3H, 2xCH2), 4.54 (d, 1H, J= 5.9 Hz, CH1N), 5.40 (d, 1H, J = 5.9 Hz, CH-O), 8.21 (d, 2H, J= 5.8 Hz, ArH), 8.94 (d, 2H, J= 5.8 Hz, ArH).
MS-ESI m/z (% rel. int.): 236.1 ([MH]+, 17), 219 (25), 148 (100).
HPLC: Method A, detection UV 254 nm, Compound 22 RT = 0.8 min, peak area 96.3 %.
PAT
- 3-aryl-3-hydroxy-2-amino-propionic acid amides, 3-heteroaryl-3-hydroxy-2-amino-propionic acid amides and related compounds having analgesic and / or immunostimulatory activityPublication Number: ES-2565236-T3Priority Date: 2005-01-26Grant Date: 2016-04-01
- 3-ARYL-3-HYDROXY-2-AMINO-PROPIONIC ACID, 3-HETEROARYL-3-HYDROXY-2-AMINOPROPIONIC ACID AMIDES AND RELATED COMPOUNDS HAVING ANALGESIC AND/OR IMMUNOSTIMULATING ACTIVITYPublication Number: BR-122018068138-B1Priority Date: 2005-01-26
- 1-aryl-1-hydroxy-2,3-diamino-propyl amines, 1-heteroaryl-1-hydroxy-2,3-diamino-propyl amines and related compounds having analgesic and/or immuno stimulant activityPublication Number: US-2011288094-A1Priority Date: 2005-01-26
- 1-aryl-1-hydroxy-2,3-diamino-propyl amines, 1-heteroaryl-1-hydroxy-2,3-diamino-propyl amines and related compounds having analgesic and/or immuno stimulant activityPublication Number: US-9828349-B2Priority Date: 2005-01-26Grant Date: 2017-11-28
- Compounds having analgesic and/or immunostimulant activityPublication Number: US-2012157497-A1Priority Date: 2005-01-26
- 3-aryl-3-hydroxy-2-amino-propionic acid amides, 3-heteroaryl-3-hydroxy-2-amino-propionic acid amides and related compounds having analgesic and/or immunostimulant activityPublication Number: AU-2012241156-A1Priority Date: 2005-01-26
- 3-Aryl-3-hydroxy-2-amino-propionic acid amide, 3-heteroaryl-3-hydroxy-2-amino-propionic acid amide and related compounds having analgesic activity and / or immunostimulatory activityPublication Number: JP-2012162547-APriority Date: 2005-01-26
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

 | |
| Clinical data | |
|---|---|
| Other names | RL-007; RL007; FSV7-007 |
| Routes of administration | Oral[1][2] |
| Identifiers | |
| IUPAC name | |
| CAS Number | 903884-71-9 |
| PubChem CID | 11535990 |
| ChemSpider | 9710771 |
| UNII | 3LW01V88B7 |
| ChEMBL | ChEMBL5095258 |
| CompTox Dashboard (EPA) | DTXSID90238113 |
| Chemical and physical data | |
| Formula | C12H17N3O2 |
| Molar mass | 235.287 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
References
- “RL 007”. AdisInsight. 10 June 2024. Retrieved 26 February 2025.
- Donello JE, Walker GA, Schweighoffer F, and Pando MP. Abstract Number: 149. RL-007, a novel oral neuromodulator, enhances synaptic plasticity and cognition in non-clinical models. American College of Neuropsychopharmacology (ACNP) annual meeting. December 5, 2023. https://ir.atai.life/static-files/06c60339-e93c-42fc-9323-c0e38e83f86e
- “Delving into the Latest Updates on RL-007 with Synapse”. Synapse. 23 January 2025. Retrieved 26 February 2025.
- Brady LS, Lisanby SH, Gordon JA (2023). “New directions in psychiatric drug development: promising therapeutics in the pipeline”. Expert Opinion on Drug Discovery. 18 (8): 835–850. doi:10.1080/17460441.2023.2224555. PMID 37352473.
- Vita A, Barlati S, Cavallaro R, Mucci A, Riva MA, Rocca P, et al. (2024). “Definition, assessment and treatment of cognitive impairment associated with schizophrenia: expert opinion and practical recommendations”. Frontiers in Psychiatry. 15 1451832. doi:10.3389/fpsyt.2024.1451832. PMC 11450451. PMID 39371908.
- Ye N, Wang Q, Li Y, Zhen X (March 2025). “Current emerging therapeutic targets and clinical investigational agents for schizophrenia: Challenges and opportunities”. Medicinal Research Reviews. 45 (2): 755–787. doi:10.1002/med.22086. PMID 39300769.
- “2-Amino-3-hydroxy-3-(pyridin-4-yl)-1-(pyrrolidin-1-yl)propan-1-one, (2R,3S)-“. PubChem. Retrieved 26 February 2025.
//////////inidascamine, ANAX LABS, schizophrenia, 3LW01V88B7, RL 007
Imofinostat
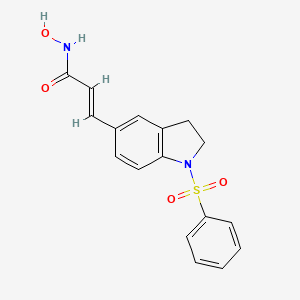
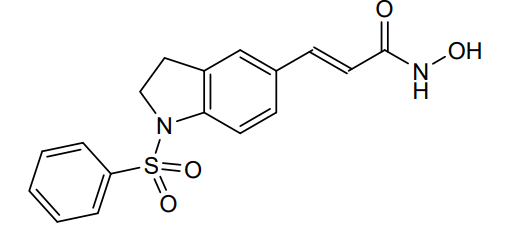
Imofinostat
CAS 1338320-94-7
MF C17H16N2O4S MW 344.4 g/mol
- 3-(1-(Benzenesulfonyl)-2,3-dihydro-1H-indol-5-yl)-N-hydroxyacrylamide
- (E)-3-[1-(benzenesulfonyl)-2,3-dihydroindol-5-yl]-N-hydroxyprop-2-enamide
(2E)-3-[1-(benzenesulfonyl)-2,3-dihydro-1H-indol-5-yl]-N-hydroxyprop2-enamide
histone deacetylase inhibitor, antineoplastic, ABT-301, MPT0E028, ABT 301, MPT0E 028, T65L58FI65
Imofinostat (also known as ABT-301 or MPT0E028) is an orally bioavailable, small-molecule histone deacetylase (HDAC) inhibitor primarily being developed as an innovative precision oncology treatment. Developed by companies like AnBogen Therapeutics and Formosa Pharmaceuticals, it is designed to reactivate tumor suppressor genes that cancer cells have silenced, thereby triggering cancer cell death (apoptosis) and stopping tumor growth.
Mechanism of Action
Imofinostat works through a distinct multi-modality approach to fight cancer cells:
- HDAC Inhibition: It acts as a potent inhibitor of human pan-histone deacetylase enzymes, showing preferential selectivity for Class I HDACs (especially HDAC3). This blocks the deacetylation of histone proteins, causing chromatin to remodel and forcing cancer cells to express tumor-suppressor genes.
- Akt Pathway Targeting: Independent of its epigenetic effects, it can directly target and reduce the activation (phosphorylation) of the Akt protein kinase, a major pathway that cancer cells use to survive and multiply.
- Microenvironment Modulation: Preclinical data shows it alters the tumor microenvironment by converting “cold tumors” (invisible to the immune system) into “hot tumors” by promoting the infiltration of CD8+ cytotoxic T cells.
Current Clinical Status & Indications
Imofinostat is actively moving through clinical trial pipelines, focusing heavily on combination therapies to overcome treatment resistance:
- Colorectal Cancer (CRC): It is currently being evaluated in a global Phase 1/2 clinical trial (NCT07244705). It is combined with the immune checkpoint inhibitor tislelizumab (Tevimbra®) and the anti-angiogenic drug bevacizumab to treat advanced, metastatic colorectal cancer.
- Pancreatic Cancer: Recent data presented at the 2026 American Association for Cancer Research (AACR) Annual Meeting demonstrates that imofinostat disrupts the HDAC3-NRF2 pathway. This action breaks down chemotherapy resistance in highly aggressive KRAS-mutant pancreatic ductal adenocarcinoma, making tumors much more sensitive to treatments like gemcitabine.
- Other Solid Tumors: Phase 1 monotherapy trials have confirmed that the drug possesses a highly competitive safety profile across a broad variety of advanced solid tumors.
Imofinostat is an orally bioavailable N-hydroxyacrylamide-derived inhibitor of both human pan-histone deacetylase (HDAC) enzymes and the serine/threonine protein kinase Akt (protein kinase B), with potential antineoplastic activity. Upon administration, imofinostat selectively binds to and inhibits HDACs, which inhibits deacetylation of histone proteins and leads to the accumulation of highly acetylated histones. This may result in both an induction of chromatin remodeling, and the selective transcription of tumor suppressor genes. This prevents cell division and induces both cell cycle arrest and apoptosis, which may inhibit the proliferation of susceptible tumor cells. In addition, imofinostat inhibits the phosphorylation and activation of Akt, which prevents the activation of downstream signaling pathways, independent of its HDAC inhibitory activity. HDACs, upregulated in many tumor cell types, are a family of enzymes that deacetylate histone proteins. Akt, overexpressed in many tumor cell types, plays a key role in tumor cell proliferation and survival.
Dose-Seeking Study of MPT0E028 in Subjects With Advanced Solid Malignancies Without Standard Treatment
CTID: NCT02350868
Phase: Phase 1
Status: Completed
Date: 2019-04-11
PAT
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2011126821&_cid=P11-MQ4LAI-84972-1
COMD 12
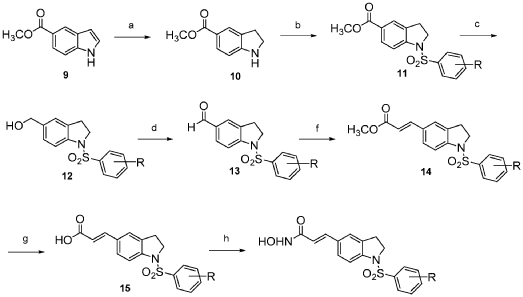
Compound 12 was synthesized via the route as shown in Scheme 3 above (reagents and conditions: (a) NaBH3CN, AcOH; (b) Benzenesulfonyl chloride, 4-methoxybenzenesulfonyl chloride, 3,4-dimethoxybenzenesulfonyl chloride, 4-fluorobenzenesulfonyl chloride, or 4-nitrobenzenesulfonyl chloride, pyridine; (c) L1AIH4, THF; (d) PDC, MS, CH2C12; f) Ph3P = CH-COOCH3, CH2C12; (g) 1M LiOH(aq), dioxane; (h) (i) NH2OTHP, PyBOP, NEt3, DMF; (ii) TFA, MeOH; (i) Fe, NH4C1, Isopropanol, H20).
2,3-Dihydro-lH-indole-5-carboxylic acid methyl ester (10): sodium cyanoborohydride (0.16 g, 2.57 mmol) was added to a solution of methyl indole-5-carboxylate (9) (0.30 g, 1.71 mmol) in AcOH (2 mL) at 0 °C. The reaction mixture was warmed to room temperature and stirred for 2 h before it was quenched with water at 0 °C. Concentrated NaOH was added to reach pH=10. The aqueous layer was extracted with CH2CI2 (15 mL x 3). The combined organic layer was dried over anhydrous MgS04 and concentrated under reduced pressure to give a yellow residue, which was purified by silica gel chromatography (EtOAc: n-hexane = 1 : 2) to afford 10 (0.28 g). 1H NMR (500MHz, CDC13): δ 3.06 (t, J= 8.5 Hz, 2H), 3.65 (t, J= 8.5 Hz, 2H), 3.84 (s, 3H), 6.53-6.55 (m, 1H), 7.75-7.76 (m, 2H).
l-Benzenesulfonyl-2,3-dihydro-lH-indole-5-carboxylic acid methyl ester (11): To a solution of 10 (0.28 g, 1.58 mmol) in pyridine (2 mL), benzenesulfonyl chloride (0.40 ml, 3.16 mmol) was added. The reaction mixture was refluxed overnight. The mixture was then purified by silica gel chromatography (EtOAc: n-hexane = 1 : 3) to afford 11 (0.40 g). 1H NMR (500MHz, CDCI3): δ 2.99 (t, J= 8.6 Hz, 2H), 3.87 (s, 3H), 3.97 (t, J= 8.6 Hz, 2H), 7.45-7.48 (m, 2H), 7.56-7.59 (m, 1H), 7.66 (d, J= 8.5 Hz, 1H), 7.75 (s, 1H), 7.82 (d, J= 7.7 Hz, 2H), 7.90 (d, J= 7.9 Hz, 1H).
(l-Benzenesulfonyl-2,3-dihydro-lH-indol-5-yl)-methanol (12): LAH (0.10 g, 2.52 mmol) was added to a solution of 11 (0.40 g, 1.26 mmol) in THF (10 mL) at 0 °C. The reaction mixture was warmed to room temperature and stirred for 2 h before it was quenched with water and then extracted with CH2CI2 (15 mL x 3). The combined organic layer was dried over anhydrous MgS04 and concentrated under reduced pressure. The reaction mixture was purified by silica gel chromatography (EtOAc: n-hexane = 1 : 1) to afford 12 (0.24 g). 1H NMR (500MHz, CDC13): δ 2.83 (t, J= 8.4 Hz, 2H), 3.92 (t, J= 8.5 Hz, 2H), 4.49 (s, 2H), 7.09 (s, 1H), 7.16 (d, J= 8.2 Hz, 1H), 7.46-7.49 (m, 2H), 7.53 (d, J= 8.2 Hz, 1H), 7.60 (t, J= 7.5 Hz, 1H), 7.76 (d, J= 7.7 Hz, 2H).
l-Benzenesulfonyl-2,3-dihydro-lH-indole-5-carbaldehyde (13): molecular sieves (0.63g) were added to a solution of 12 (0.24 g, 0.83 mmol) in CH2C12 (10 mL), PDC (0.63 g, 1.66 mmol). The mixture was stirred at room temperature overnight before it was filtered through celite. The organic layer was concentrated under reduced pressure then purified by silica gel chromatography (EtOAc: n-hexane = 1 : 2) to afford 13 (0.19 g). 1H NMR (500MHz, CDC13): δ 3.05 (t, J= 8.6 Hz, 2H), 4.01 (t, J= 8.7 Hz, 2H), 7.46-7,49 (m, 2H), 7.58-7.62 (m, 2H), 7.71 (d, J= 8.3 Hz, 1H), 7.75 (d, J= 8.3 Hz, 1H), 7.84 (d, J= 7.8 Hz, 2H), 9.85 (s, 1H).
3-(l-Benzenesulfonyl-2,3-dihydro-lH-indol-5-yl)-acrylic acid methyl ester (14): Methyl (triphenylphosphoranylidene) acetate (0.27 g, 0.79 mmol) was added to a solution of 13 (0.19g,
0.66 mmol) in CH2CI2 (10 mL). The mixture was stirred at room temperature for 3h before it was
quenched with water and then extracted with CH2CI2 (15 mL x 3). The combined organic layer was dried over anhydrous MgS04 and concentrated under reduced pressure to give a yellow residue, which was then purified by silica gel chromatography (EtOAc: n-hexane = 1 : 3) to afford 14
(0.20 g).
3-(l-Benzenesulfonyl-2,3-dihydro-lH-indol-5-yl)-acrylic acid (15): 1M LiOH aqueous solution (1.16 ml, 1.16 mmol) was added to a solution of 14 (0.20g, 0.58 mmol) in dioxane
(15 mL). The reaction mixture was stirred at 40 °C overnight before it was concentrated under reduced pressure. The residue was dissolved in water and concentrated HCl was added up to acidic pH to give the precipitation, which was dried by vacuum to afford 15 (0.16 g). 1H NMR (500MHz, CD3OD): δ 2.92 (t, J= 8.5 Hz, 2H), 3.96 (t, J= 8.5 Hz, 2H), 6.33 (d, J= 15.9 Hz, 1H), 7.38 (s, 1H), 7.41 (d, J= 8.5 Hz, 1H), 7.50-7.53 (m, 2H), 7.55 (d, J= 16.1 Hz, 1H), 7.58-7.64 (m, 2H), 7.82 (d, J = 7.6 Hz, 2H).
3-(l-Benzenesulfonyl-2,3-dihydro-lH-indol-5-yl)-N-hydroxy-acrylamide
(Compound 12): NH2OTHP (0.05 g, 0.44 mmol) was added to a solution of 15 (0.12 g, 0.37 mmol), PyBOP (0.20 g, 0.39 mmol), triethylamine (0.12 ml, 0.88 mmol) in DMF (1.5 mL). The reaction mixture was stirred at room temperature for 1 h before it was quenched with water, followed by extraction with EtOAc (15 mL x 3). The combined organic layer was dried over anhydrous MgS04 and concentrated under reduced pressure. The residue was purified by silica gel chromatography (CH2C12: CH3OH = 30 : 1 : l%NH3(aq)) to give a white solid, which was treated with TFA (1.13 ml, 15.21 mmol) in the presence of CH3OH (25 mL) and stirred overnight at room temperature. The reaction mixture was concentrated under reduced pressure to give a white residue, which was recrystallized by CH3OH to afford Compound 12 (0.12 g). 1H NMR (500MHz,
CD3OD): δ 2.91 (t, J= 8.5 Hz, 2H), 3.96 (t, J= 8.4 Hz, 2H), 6.32 (d, J= 15.8 Hz, 1H), 7.32 (s, 1H), 7.37-7.39 (m, 1H), 7.46 (d, J= 15.7 Hz, 1H), 7.50-7.53 (m, 2H), 7.58-7.64 (m, 2H), 7.82 (d, J= 7.8 Hz, 2H). MS (EI) mlz: 170 (100%), 344 (M+, 3.21%). HRMS (EI) for Ci7Hi6N204S (M+): calcd, 344.0831; found, 344.0829.
PAT
US20150368195
https://patentscope.wipo.int/search/en/detail.jsf?docId=US154007904&_cid=P11-MQ4M0P-01888-1
PAT
- Indolyl or indolinyl hydroxamate compoundsPublication Number: US-8846748-B2Priority Date: 2010-03-29Grant Date: 2014-09-30
- Indolyl or indolinyl hydroxamate compoundsPublication Number: US-9598364-B2Priority Date: 2010-03-29Grant Date: 2017-03-21
- Indolyl or indolinyl hydroxamate compoundsPublication Number: WO-2011126821-A2Priority Date: 2010-03-29
- Indolyl or indolinyl hydroxamate compoundsPublication Number: EP-2552887-A2Priority Date: 2010-03-29
- Indolyl or indolinyl hydroxamate compoundsPublication Number: US-2011245315-A1Priority Date: 2010-03-29
- Indolyl or indolinyl hydroxamate compoundsPublication Number: US-2014364477-A1Priority Date: 2010-03-29
- Indolyl or indolinyl hydroxamate compoundsPublication Number: EP-2552887-B1Priority Date: 2010-03-29Grant Date: 2018-10-24
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
//////////imofinostat, anax labs, histone deacetylase inhibitor, antineoplastic, ABT-301, MPT0E028, ABT 301, MPT0E 028, T65L58FI65
Ifupinostat
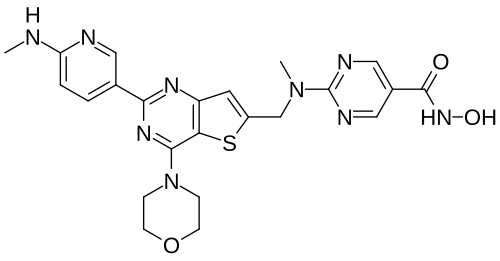
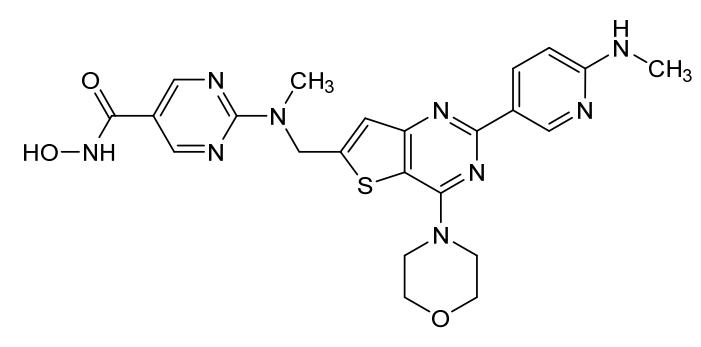
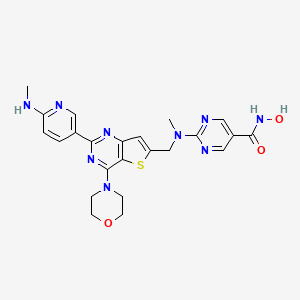
Ifupinostat
CAS 1235449-52-1
MF C23H25N9O3S MW507.6 g/mol
N-hydroxy-2-[methyl-[[2-[6-(methylamino)-3-pyridinyl]-4-morpholin-4-ylthieno[3,2-d]pyrimidin-6-yl]methyl]amino]pyrimidine-5-carboxamide
5-Pyrimidinecarboxamide, N-hydroxy-2-(methyl((2-(6-(methylamino)-3-pyridinyl)-4-(4-morpholinyl)thieno(3,2-d)pyrimidin-6-yl)methyl)amino)-
DQ7TD3X4ZJ, BEBT908 FREE BASE,
Ifupinostat (brand name Betlin; formerly known as BEBT-908) is a first-in-class, dual-action cancer medication used to treat specific types of blood cancer. It is developed by the biopharmaceutical company BeBetter Med.
Approved Clinical Use
The drug is conditionally approved in China as a monotherapy for adults with relapsed or refractory diffuse large B-cell lymphoma (r/r DLBCL). It is specifically indicated for patients who have already undergone at least two prior lines of systemic therapy.
Mechanism of Action
Unlike traditional cancer drugs that target a single pathway, ifupinostat is designed to simultaneously disrupt two major cellular mechanisms that drive tumor growth:
- PI3Kα Inhibition: It blocks phosphoinositide 3-kinase alpha (PI3Kα), shutting down the downstream PI3K/AKT/mTOR survival pathway within cancer cells.
- HDAC Inhibition: It blocks histone deacetylase (HDAC) enzymes, leading to epigenetic modifications (such as increased histone-3 acetylation) that trigger cancer cell death.
By hitting both targets at once, the drug suppresses tumor cell proliferation, downregulates the cancer-driving c-Myc protein, and induces cell death via ferroptosis (an iron-dependent form of programmed cell death).
Clinical Research and Future Outlook
- Combinations: Beyond its use as a single agent, ifupinostat is being evaluated in combination with the monoclonal antibody rituximab as a potential second-line treatment for r/r DLBCL. Early phase 1b clinical data presented at ASCO showed a promising 76.2% objective response rate (ORR).
- Brain Penetration: Lab studies indicate that the molecule successfully crosses the blood-brain barrier (BBB), showing therapeutic potential for central nervous system lymphomas.
- Ongoing Verification: Because its initial regulatory green light was given on a conditional basis, a confirmatory randomized phase 3 trial is currently underway to achieve full approval
Ifupinostat is an inhibitor of both phosphoinositide 3-kinase (PI3K) and histone deacetylase (HDAC) enzymes, with potential antineoplastic activity. Upon administration, ifupinostat binds to and inhibits the activity and mediated signaling of both PI3K and HDAC. In addition, ifupinostat may also inhibit other signaling pathways. This may prevent growth of PI3K and/or HDAC-expressing tumor cells.
Ifupinostat (trade name Betlin) is a drug used for the treatment of cancer. It is approved in China for adults with relapsed or refractory diffuse large B-cell lymphoma who have received at least two lines of systemic therapy.[1] It is being developed by BeBetter Med.[2]
Ifupinostat acts as both a phosphoinositide 3-kinase α (PI3Kα) inhibitor and a histone deacetylase (HDAC) inhibitor.[1][3][4]
SYN
- Hydroxamic acid hybrids: Histone deacetylase inhibitors with anticancer therapeutic potencyPublication Name: European Journal of Medicinal ChemistryPublication Date: 2023-12-15PMID: 37875056DOI: 10.1016/j.ejmech.2023.115879
- A Dual PI3K/HDAC Inhibitor Induces Immunogenic Ferroptosis to Potentiate Cancer Immune Checkpoint TherapyPublication Name: Cancer ResearchPublication Date: 2021-12-15PMID: 34711611DOI: 10.1158/0008-5472.can-21-1547
SYN
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2024086894&_cid=P21-MQ35ZX-16907-1
SYN
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2024086894&_cid=P21-MQ35ZX-16907-1
PAT
- Phosphoinositide 3-Kinase Inhibitors with a Zinc Binding MoietyPublication Number: US-2025230169-A1Priority Date: 2009-01-08
- Phosphoinositide 3-kinase inhibitors with a zinc binding moietyPublication Number: US-2023227467-A1Priority Date: 2009-01-08
- Phosphoinositide 3-kinase inhibitors with a zinc binding moietyPublication Number: US-11261195-B2Priority Date: 2009-01-08Grant Date: 2022-03-01
- PRMT5 inhibitors and uses thereofPublication Number: US-12448388-B2Grant Date: 2025-10-21
- KRAS G12D modulating compoundsPublication Number: US-12448400-B2Grant Date: 2025-10-21
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
 | |
| Clinical data | |
|---|---|
| Trade names | Betlin; 贝特琳 |
| Other names | BEBT-908 |
| Legal status | |
| Legal status | Rx in China |
| Identifiers | |
| IUPAC name | |
| CAS Number | 1235449-52-1 |
| PubChem CID | 59474330 |
| ChemSpider | 45743497 |
| UNII | DQ7TD3X4ZJ |
| ChEMBL | ChEMBL5618885 |
| Chemical and physical data | |
| Formula | C23H25N9O3S |
| Molar mass | 507.57 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
References
- Fung S (December 2025). “Ifupinostat: First Approval”. Drugs. 85 (12): 1629–1633. doi:10.1007/s40265-025-02248-z. PMID 41028651.
- “Ifupinostat – BeBetter Med”. AdisInsight. Springer Nature Switzerland AG.
- Wang N, Mo Z, Pan L, Zhou M, Ye X, Liu X, et al. (November 2023). “Dual PI3K/HDAC Inhibitor BEBT-908 Exhibits Potent Efficacy as Monotherapy for Primary Central Nervous System Lymphoma”. Targeted Oncology. 18 (6): 941–952. doi:10.1007/s11523-023-01006-z. PMID 37855991.
- Luzietti L, Pires GS, Ryan A, Regidor C, Hiller M, Sarti D, et al. (2025). “Design, synthesis, and biological evaluation of novel triazine-based dual HDAC/PI3K inhibitors for breast cancer therapy”. ChemRxiv. doi:10.26434/chemrxiv-2025-tzwbz.
/////////////ifupinostat, anax labs, Q7TD3X4ZJ, BEBT908 FREE BASE, BEBT 908
Gozanertinib
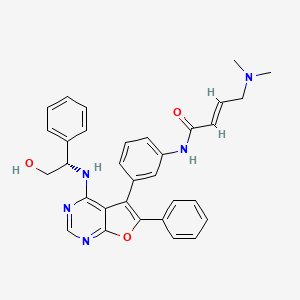
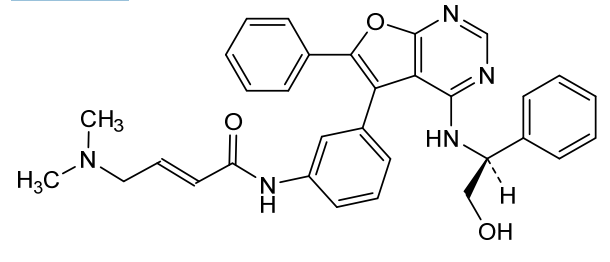
Gozanertinib
CAS 1226549-49-0
MF C32H31N5O3 MW533.6 g/mol
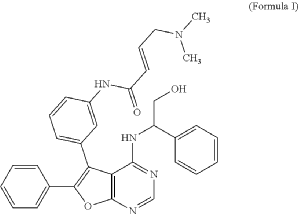
(E)-4-(dimethylamino)-N-[3-[4-[[(1S)-2-hydroxy-1-phenylethyl]amino]-6-phenylfuro[2,3-d]pyrimidin-5-yl]phenyl]but-2-enamide
(2E)-4-(dimethylamino)-N-[3-(4-{[(1S)-2-hydroxy-1-phenylethyl]amino}-6-phenylfuro[2,3-d]pyrimidin-5-yl)phenyl]but-2-
enamide
epidermal growth factor receptor tyrosine kinase inhibitor, antineoplastic, DBPR 112, ABT 101, 6G0COS33K4
Gozanertinib (also known as DBPR112 or ABT-101) is an orally bioavailable, advanced small-molecule dual kinase inhibitor designed to treat advanced non-small cell lung cancer (NSCLC). It targets alterations in the epidermal growth factor receptor (EGFR) and human epidermal growth factor receptor 2 (HER2) families.
Mechanism of Action
Gozanertinib is a furanopyrimidine-based tyrosine kinase inhibitor. It functions by entering the ATP-binding pocket of the receptor and forming an irreversible covalent bond with a specific cysteine residue (Cys797). By permanently blocking these receptors, it halts downstream oncogenic signaling pathways—specifically the RAS/RAF/MEK/ERK and PI3K/AKT cascades—thereby inducing cancer cell death and suppressing tumor expansion.
Target Profile and Key Mutations
Unlike earlier generations of tyrosine kinase inhibitors that only target standard configurations, gozanertinib is optimized to combat specific treatment-resistant mutations:
- EGFR Mutations: It effectively targets wild-type EGFR as well as the dual L858R/T790M resistance mutations.
- Exon 20 Insertions: A standout feature of gozanertinib is its preclinical potency against EGFR and HER2 exon 20 insertion (Ex20ins) mutations. According to chemical development findings published in the Journal of Medicinal Chemistry, it demonstrated ten times better potency against these specific insertions than the widely used third-generation inhibitor, osimertinib.
Development and Status
The drug was initially discovered through scaffold optimization by the National Health Research Institutes (NHRI) and is being co-developed with Anbogen Therapeutics. The International Nonproprietary Name (INN) “gozanertinib” was formally proposed for the compound in early 2025. Preclinical evaluations indicated favorable oral bioavailability and strong anti-tumor efficacy compared to older inhibitors like afatinib, advancing the compound into early-phase clinical trials
Gozanertinib is an orally bioavailable dual kinase inhibitor of epidermal growth factor receptor (EGFR; ErbB1) and human epidermal growth factor receptor 2 (HER2; EGFR2; ErbB2), including EGFR L858R, EGFR T790M and HER2 exon 20 insertion (Ex20ins) mutations, with potential antineoplastic activity. Upon oral administration, gozanertinib targets, binds to and inhibits the activity of EGFR or HER2 insertions or mutations. This prevents EGFR/HER2-mediated signaling, which may induce cell death and inhibit tumor growth in EGFR/HER2-overexpressing tumor cells. The ErbB receptor tyrosine kinase family is involved in key cellular functions, including cell growth and survival. EGFR and HER2 alterations constitutively upregulate kinase activity.
- Phase 1b/2 Study to Evaluate ABT-101 in Solid Tumor and NSCLC PatientsCTID: NCT05532696Phase: Phase 1/Phase 2Status: RecruitingDate: 2024-06-24
- A Study of DBPR112 in Patients With Head and Neck Cancer and EGFR Mutated Lung CancerCTID: NCT03246854Phase: Phase 1Status: TerminatedDate: 2020-12-17
PAT
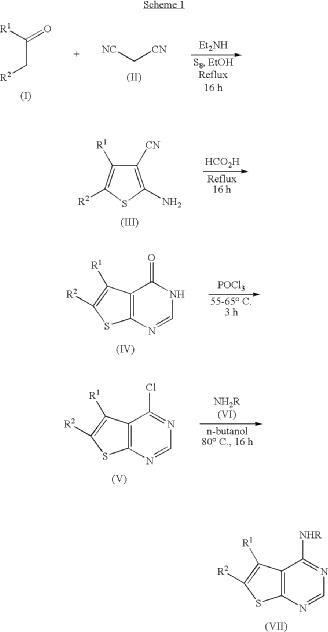
PAT
https://patentscope.wipo.int/search/en/detail.jsf?docId=US43249513&_cid=P11-MQ1QG3-86325-1
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Development of Furanopyrimidine-Based Orally Active Third-Generation EGFR Inhibitors for the Treatment of Non-Small Cell Lung CancerPublication Name: Journal of Medicinal ChemistryPublication Date: 2023-02-07PMCID: PMC9969398PMID: 36749735DOI: 10.1021/acs.jmedchem.2c01434
- Discovery of a Furanopyrimidine-Based Epidermal Growth Factor Receptor Inhibitor (DBPR112) as a Clinical Candidate for the Treatment of Non-Small Cell Lung CancerPublication Name: Journal of Medicinal ChemistryPublication Date: 2019-09-27PMID: 31560541DOI: 10.1021/acs.jmedchem.9b00722
PAT
- Active cancer immunotherapy by immune modulation via globo series antigensPublication Number: EP-4248214-A1Priority Date: 2020-11-19
- Fused Bicyclic and Tricyclic Pyrimidine Compounds as Tyrosine Kinase InhibitorsPublication Number: US-2010120805-A1Priority Date: 2008-11-10
- Fused bicyclic and tricyclic pyrimidine compounds as tyrosine kinase inhibitorsPublication Number: US-8507502-B2Priority Date: 2008-11-10Grant Date: 2013-08-13
- Fused bicyclic and tricyclic pyrimidine compounds as tyrosine kinase inhibitorsPublication Number: WO-2010054285-A2Priority Date: 2008-11-10
- Fused bicyclic and polycyclic pyrimidine compounds as tyrosine kinase inhibitorsPublication Number: CN-102264745-APriority Date: 2008-11-10
- Active cancer immunotherapy through immune modulation via GLOBO series antigensPublication Number: CN-116847875-APriority Date: 2020-11-19
- Active cancer immunotherapy by immune modulation via globo series antigensPublication Number: CA-3200572-A1Priority Date: 2020-11-19
- Active cancer immunotherapy by immune modulation via globo series antigensPublication Number: WO-2022109601-A1Priority Date: 2020-11-19
- Active cancer immunotherapy by immune modulation via globo series antigensPublication Number: IL-302947-APriority Date: 2020-11-19
- Active cancer immunotherapy by immunomodulation through GLOBO family antigensPublication Number: KR-20230110529-APriority Date: 2020-11-19
- Crystalline forms of (s, e)-4-(dimethylamino)-n-(3-(4-(2-hydroxy-1-phenylethylamino)-6-phenylfuro[2,3-d]pyrimidin-5-yl)phenyl)but-2-enamide free basePublication Number: TW-I809967-BPriority Date: 2021-07-06Grant Date: 2023-07-21
- Crystalline forms of (S, E)-4-(dimethylamino)-N-(3-(4-(2-hydroxy-1-phenylethylamino)-6-phenylfuro[2,3-d]pyrimidin-5-yl)phenyl)but-2- enamide free basePublication Number: US-12240858-B2Priority Date: 2021-07-06Grant Date: 2025-03-04
- Active cancer immunotherapy by immune modulation via globo series antigensPublication Number: TW-202237177-APriority Date: 2020-11-19
- Active cancer immunotherapy by immune modulation via globo series antigensPublication Number: US-2024139301-A1Priority Date: 2020-11-19
- Active cancer immunotherapy by immune modulation via globo series antigensPublication Number: AU-2021382807-A1Priority Date: 2020-11-19
- Crystalline forms of (s, e)-4-(dimethylamino)-n-(3-(4-(2-hydroxy-1-phenylethylamino)-6-phenylfuro[2,3-d]pyrimidin-5-yl)phenyl)but-2-enamide free basePublication Number: US-2023021909-A1Priority Date: 2021-07-06
- Crystalline forms of (s, e)-4-(dimethylamino)-n-(3-(4-(2-hydroxy-1-phenylethylamino)-6-phenylfuro[2,3-d]pyrimidin-5-yl)phenyl)but-2-enamide free basePublication Number: WO-2023283269-A1Priority Date: 2021-07-06
- Crystalline forms of (s, e)-4-(dimethylamino)-n-(3-(4-(2-hydroxy-1-phenylethylamino)-6-phenylfuro[2,3-d]pyrimidin-5-yl)phenyl)but-2-enamide free basePublication Number: US-2024368175-A1Priority Date: 2021-07-06
- Crystalline forms of (s, e)-4-(dimethylamino)-n-(3-(4-(2-hydroxy-1-phenylethylamino)-6-phenylfuro[2,3-d]pyrimidin-5-yl)phenyl)but-2-enamide free basePublication Number: TW-202309041-APriority Date: 2021-07-06
- Crystalline forms of (s, e)-4-(dimethylamino)-n- (3-(4-(2-hydroxy-1-phenylethylamino)-6-phenylfuro[2,3-d]pyrimidin-5-yl)phenyl)but-2-enamide free basePublication Number: EP-4330259-A1Priority Date: 2021-07-06
//////gozanertinib, ANAX LABS, epidermal growth factor receptor tyrosine kinase inhibitor, antineoplastic, DBPR 112, ABT 101, 6G0COS33K4
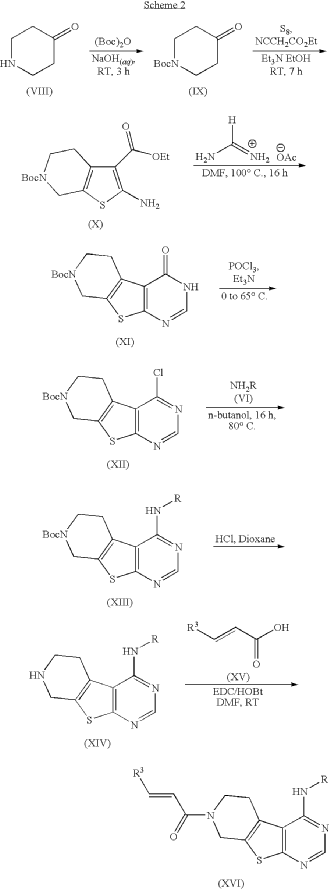
Gintemetostat
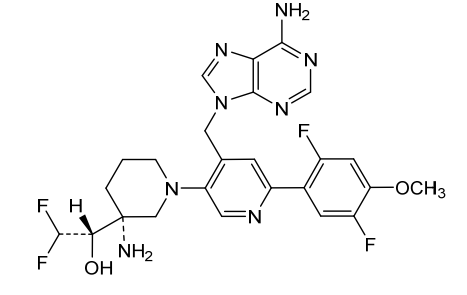
Gintemetostat
(1S)-1-[(3R)-3-amino-4′-[(6-amino-9H-purin-9-yl)methyl]-6′-(2,5-difluoro-4-methoxyphenyl)-3,4,5,6-tetrahydro-2H-[1,3′-bipyridin]-3-yl]-2,2-difluoroethan1-ol
antineoplastic, KTX 1001, NSD2 inhibitor 161, A48CGJ5UQM
CAS 2604513-16-6
MF C25H26F4N8O2 MW 546.5 g/mol
(S)-1-((R)-3-Amino-1-(4-((6-amino-9H-purin-9-yl)methyl)-6-(2,5-difluoro-4-methoxyphenyl)pyridin-3-yl)piperidin-3-yl)-2,2-difluoroethan-1-ol
Gintemetostat (also known as KTX-1001) is a first-in-class, orally administered small molecule being developed to treat relapsed and refractory multiple myeloma. It works as a selective inhibitor of NSD2 (also known as MMSET), targeting the epigenetic drivers of high-risk cancers.
How it Works
- Mechanism: Gintemetostat selectively binds to the catalytic SET domain of the NSD2 enzyme.
- Effect: By blocking this enzyme, it downregulates oncogenic signaling, decreases cancer cell growth, and can enhance T-cell activation against the tumor.
Target Patient Population
- High-Risk Myeloma: The drug focuses heavily on patients harboring the t(4;14) translocation, a genetic alteration found in 10-15% of patients that often causes aggressive relapses.
- Refractory Cases: It has shown notable single-agent activity in heavily pretreated patients who have exhausted standard-of-care, triple-class refractory treatment options.
Current Clinical Status
- Phase 1 Trial: Early data from phase 1 trials (such as NCT05651932) showed the drug has manageable safety profiles and offers clinical benefit (ranging from stable disease to very good partial response) in patients with aggressive, hard-to-treat multiple myeloma.
- Future Developments: Researchers are expanding studies to pair gintemetostat with other standard myeloma treatments, such as proteasome inhibitors and CELMoDs, to create stronger synergistic anti-cancer effects.
Gintemetostat is an orally available small molecule inhibitor of the histone-lysine N-methyltransferase nuclear receptor-binding SET domain protein 2 (NSD2; MMSET; WHSC1), with potential antineoplastic activity. Upon oral administration, gintemetostat selectively targets and binds to NSD2, and inhibits its catalytic activity and the mono- and di-methylation of histone H3 lysine 36 (H3K36). This modulates the expression of genes involved in cellular processes including cellular proliferation, which may lead to decreased growth of cancer cells. NSD2, a member of the NSD family of histone lysine methyltransferase enzymes that catalyzes the mono- and di-methylation of H3K36, is overexpressed and dysregulated in many types of cancers.
SYN
Discovery of a Highly Potent and Selective Inhibitor Targeting Protein Lysine Methyltransferase NSD2
Publication Name: Journal of Medicinal Chemistry
Publication Date: 2024-09-04
PMID: 39230932
DOI: 10.1021/acs.jmedchem.4c00639
SYN
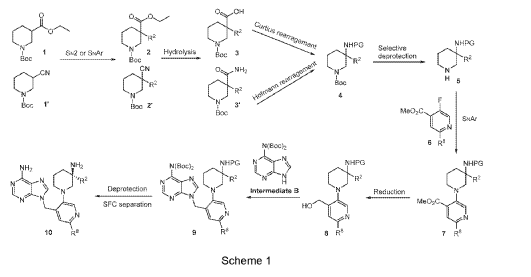
PAT
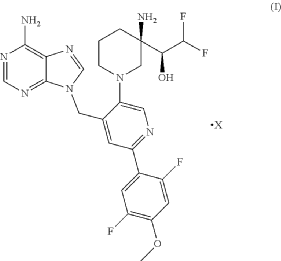
PAT
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2021028854&_cid=P12-MQ0AZT-13511-1
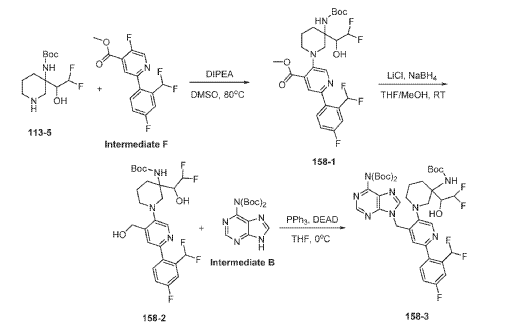
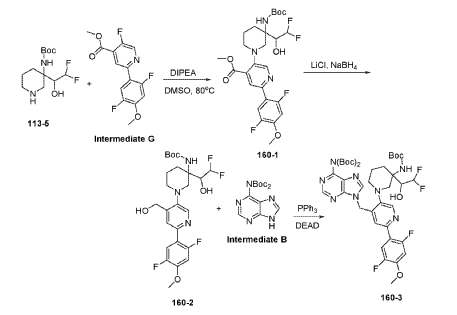
Example 160 and Example 161: (R)-1-((R)-3-amino-1-(4-((6-amino-9H-purin-9-yl)methyl)-6- (2,5-difluoro-4-methoxyphenyl)pyridin-3-yl)piperidin-3-yl)-2,2-difluoroethan-1-ol and (S)-1-((R)-3- amino-1-(4-((6-amino-9H-purin-9-yl)methyl)-6-(2,5-difluoro-4-methoxyphenyl)pyridin-3- yl)piperidin-3-yl)-2,2-difluoroethan-1-ol
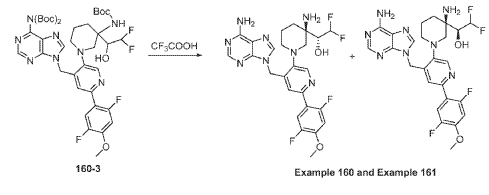
To a solution of tert-butyl (tert-butoxycarbonyl)(9-((5-(3-((tert-butoxycarbonyl)amino)-3-(2,2- difluoro-1-hydroxyethyl)piperidin-1-yl)-2-(2,5-difluoro-4-methoxyphenyl)pyridin-4-yl)methyl)-9H- purin-6-yl)carbamate (Intermediate 160-3) (200 mg, 0.237 mmol) in DCM (18 mL), was added TFA (36 mL), and the reaction mixrture was stirred at rt for 30 min under N2 atmosphere. The reaction mixture was concentrated in vacuo to give the crude product. The crude product was purifed by Pre-HPLC and SFC to afford (R)-1-((R)-3-amino-1-(4-((6-amino-9H-purin-9- yl)methyl)-6-(2,5-difluoro-4-methoxyphenyl)pyridin-3-yl)piperidin-3-yl)-2,2-difluoroethan-1-ol (Example 160) and (S)-1-((R)-3-amino-1-(4-((6-amino-9H-purin-9-yl)methyl)-6-(2,5-difluoro-4- methoxyphenyl)pyridin-3-yl)piperidin-3-yl)-2,2-difluoroethan-1-ol (Example 161).
Example 160: 1H NMR (400 MHz, CD3OD) d ppm 8.48 (s, 1H), 8.20 (d, J = 1.6 Hz, 2H), 7.58 (dd, J = 12.2, 7.3 Hz, 1H), 7.11 (d, J = 1.3 Hz, 1H), 6.90 (dd, J = 12.6, 7.1 Hz, 1H), 6.06 (td, J = 55.1, 3.9 Hz, 1H), 5.67 (s, 2H), 3.87 (s, 3H), 3.75 – 3.58 (m, 1H), 3.25 – 2.75 (m, 4H), 2.26 – 1.60 (m, 4H). LC-MS: [M+H]+ = 547.2, 548.2.
Example 161: 1H NMR (400MHz, CD3OD) d = 8.51 – 8.44 (m, 1H), 8.24 – 8.16 (m, 2H), 7.62 – 7.48 (m, 1H), 7.03 (s, 1H), 6.93 – 6.79 (m, 1H), 6.25 – 5.86 (m, 1H), 5.71 – 5.59 (m, 2H), 4.00 (m, 1H), 3.88 – 3.80 (m, 3H), 3.28 – 2.87 (m, 4H), 1.99 – 1.56 (m, 4H). LC-MS: [M+H]+ =547.4.
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Piperidinyl-methyl-purineamines as nsd2 inhibitors and anti-cancer agentsPublication Number: EP-4559915-A1Priority Date: 2019-08-14
- Piperidinyl-methyl-purineamines as nsd2 inhibitors and anti-cancer agentsPublication Number: EP-4013755-B1Priority Date: 2019-08-14Grant Date: 2025-01-08
- Piperidinyl-methyl-purinamines as NSD2 inhibitors and anticancer agentsPublication Number: CN-114585622-APriority Date: 2019-08-14
- Piperidinyl-methyl-purineamines as NSD2 inhibitors and anti-cancer agentsPublication Number: US-12312353-B2Priority Date: 2019-08-14Grant Date: 2025-05-27
- Piperidinyl-methyl-purineamines as nsd2 inhibitors and anti-cancer agentsPublication Number: WO-2021026803-A1Priority Date: 2019-08-14
- Piperidinyl-methyl-purineamines as nsd2 inhibitors and anti-cancer agentsPublication Number: EP-4013755-A1Priority Date: 2019-08-14
- Piperidinyl-methyl-purineamines as nsd2 inhibitors and anti-cancer agentsPublication Number: WO-2021028854-A1Priority Date: 2019-08-14
- Piperidinyl-methyl-purineamine D-tartrate, crystalline forms, and use thereof in the treatment of medical diseases and conditionsPublication Number: CN-119744262-APriority Date: 2022-05-18
- Piperidinyl-methyl-purine amine d-tartaric acid salts, crystalline forms, and their use in treating medical diseases and conditionsPublication Number: EP-4526305-A1Priority Date: 2022-05-18
- Piperidinyl-methyl-purineamines as nsd2 inhibitors and anti-cancer agentsPublication Number: US-2023002388-A1Priority Date: 2019-08-14
- Piperidinyl-methyl-purinamines as NSD2 inhibitors and anticancer agentsPublication Number: CN-114585622-BPriority Date: 2019-08-14Grant Date: 2024-08-09
- Piperidinyl-methyl-purineamines as NSD2 inhibitors and anti-cancer agentsPublication Number: US-11420970-B1
- Piperidinyl-methyl-purine amine d-tartaric acid salts, crystalline forms, and their use in treating medical diseases and conditionsPublication Number: AU-2023273656-A1Priority Date: 2022-05-18
- Piperidinyl-methyl-purine amine salts, crystalline forms, and their use in treating medical diseases and conditionsPublication Number: WO-2023225144-A1Priority Date: 2022-05-18
- Piperidinyl-methyl-purine amine fumaric acid salts, crystalline forms, and their use in treating medical diseases and conditionsPublication Number: WO-2023225150-A1Priority Date: 2022-05-18
- Piperidinyl-methyl-purine amine salts, crystalline forms, and their use in treating medical diseases and conditionsPublication Number: US-2025326752-A1Priority Date: 2022-05-18
- Piperidinyl-methyl-purinic amine D-tartrate salt, crystalline form and their use in the treatment of medical diseases and conditionsPublication Number: KR-20250012083-APriority Date: 2022-05-18
- Pharmaceutical compositions containing a piperidinyl-methyl-purine amine and their use in treating diseases and conditionsPublication Number: US-2024207192-A1Priority Date: 2022-12-12
- Pharmaceutical compositions containing a piperidinyl-methyl-purine amine and their use in treating diseases and conditionsPublication Number: WO-2024129670-A1Priority Date: 2022-12-12
- Piperidinyl-methyl-purine amine d-tartaric acid salts, crystalline forms, and their use in treating medical diseases and conditionsPublication Number: US-2024002385-A1Priority Date: 2022-05-18
- Piperidinyl-methyl-purine amine salts, crystalline forms, and their use in treating medical diseases and conditionsPublication Number: WO-2023225154-A1Priority Date: 2022-05-18
- Piperidinyl-methyl-purine amine d-tartaric acid salts, crystalline forms, and their use in treating medical diseases and conditionsPublication Number: WO-2023225141-A1Priority Date: 2022-05-18
////////////gintemetostat, ANAX LABS, antineoplastic, KTX 1001, NSD2 inhibitor 161, A48CGJ5UQM
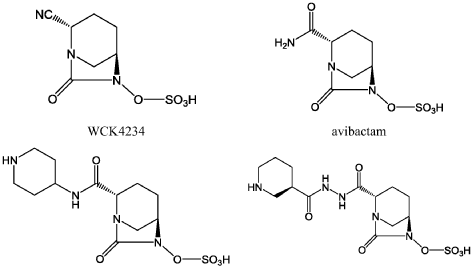
Zidebactam
Zidebactam
FDA 2026, APPROVALS 2026
To treat complicated urinary tract infections, including pyelonephritis, caused by designated susceptible microorganisms
CAS 1436861-97-0, UNII: YPM97423DB, Wockhardt Biopharm, WCK-5107, WCK5107
Molecular Formula, C13-H21-N5-O7-S
Molecular Weight, 391.4029
Disclosed in PCT International Patent Application No. PCT/IB2012/054290D
- 01 Aug 2015 Phase-I clinical trials in Bacterial infections (In volunteers, Combination therapy) in USA (IV) (NCT02532140)
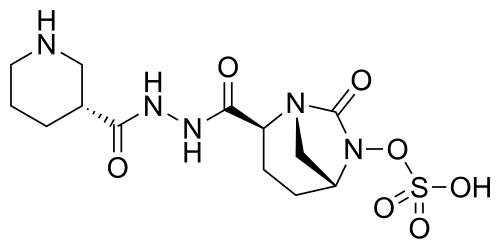
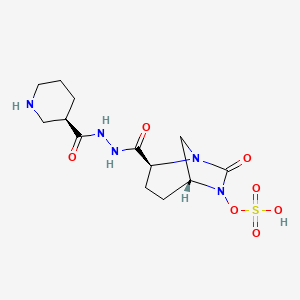
trans- sulphuric acid mono-[2-(N’-[(R)-piperidin-3-carbonyl]-hydrazinocarbonyl)-7-oxo-l,6-diaza-bicyclo[3.2.1]oct-6-yl] ester
(2S, 5R)-sulphuric acid mono-[2-(N’-[(R)-piperidin-3-carbonyl]-hydrazinocarbonyl)-7-oxo-l,6-diaza-bicyclo[3.2.1]oct-6-yl] ester
(1R,2S,5R)-l,6-Diazabicyclo [3.2.1] octane-2-carboxylic acid, 7-oxo-6-(sulfooxy)-, 2-[2-[(3R)-3-piperidinylcarbonyl]hydrazide]
trans- sulphuric acid mono-[2-(N’-[(R)-piperidin-3-carbonyl]-hydrazinocarbonyl)-7-oxo-l,6-diaza-bicyclo[3.2.1]oct-6-yl] ester
(2S, 5R)-sulphuric acid mono-[2-(N’-[(R)-piperidin-3-carbonyl]-hydrazinocarbonyl)-7-oxo-l,6-diaza-bicyclo[3.2.1]oct-6-yl] ester
(lR,2S,5R)-l,6-Diazabicyclo [3.2.1] octane-2-carboxylic acid, 7-oxo-6-(sulfooxy)-, 2-[2-[(3R)-3 -piperidinylcarbonyl] hydrazide]
1,6-Diazabicyclo(3.2.1)octane-2-carboxylic acid, 7-oxo-6-(sulfooxy)-, 2-(2-((3R)-3-piperidinylcarbonyl)hydrazide), (1R,2S,5R)-
Zidebactam potassium
cas is 1706777-49-2
Zidebactam (WCK-5107) is an antibiotic adjuvant drug which acts as a beta-lactamase inhibitor, preventing the breakdown of other antibiotic drugs.[1]
PATENT
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2019016393&_cid=P20-MPYVFE-00532-1
PATENT
http://www.google.com/patents/WO2013030733A1?cl=en

Scheme-1

Example-2
trans-sulfuric acid mono-r2-(N,-r(R)-piperidin-3-carbonyll-hvdrazinocarbonyl)-7-oxo-l,6- diaza-bicyclo Γ3.2.11 oct-6-νΠ ester
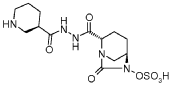
Step-1: Preparation of trans-3-[N’-(6-benzyloxy-7-oxo-l,6-diaza-bicyclo[3.2.1]octane-2-carbonyl)-hydrazinocarbonyl]-(R)-piperidin-l-carboxylic acid tert-butyl ester:
By using the procedure described in Step-1 of Example- 1 above, and by using trans-6-benzyloxy-7-oxo-l,6-diaza-bicyclo[3.2.1]octane-2-carboxylic acid (25 gm, 0.084 mol), N,N-dimethyl formamide (625 ml), EDC hydrochloride (24 gm, 0.126 mol), HOBt (16.96 gm, 0.126 mol), (R)-N-tert-butoxycarbonyl-piperidin-3-carboxylic acid hydrazide (21.40 gm , 0.088 mol) to provide the title compound in 17.0 gm quantity, 41% yield as a white solid.
Analysis: MS (ES+) CzsHasNsOe = 502.1 (M+l);
I^NMR (CDCI3) = 8.40 (br s, IH), 7.34-7.44 (m, 5H), 5.05 (d, IH), 4.90 (d, IH), 4.00 (br d, IH), 3.82 (br s, IH), 3.30 (br s, IH), 3.16-3.21 (m, IH), 3.06 (br d, IH), 2.42 (br s, IH), 2.29-2.34 (m, IH), 1.18-2.02 (m, 4H), 1.60-1.75 (m, 4H), 1.45-1.55 (m, 2H),1.44 (s, 9H).
Step-2: Preparation of trans-3-[N’-(6-hydroxy-7-oxo-l,6-diaza-bicyclo[3.2.1]octane-2-carbonyl)-hydrazinocarbonyl]-(R)-piperidin-l-carboxylic acid tert-butyl ester:
By using the procedure described in Step-2 of Example- 1 above, and by using trans-3-[N ‘ -(6-benzyloxy-7-oxo- 1 ,6-diaza-bicyclo [3.2.1 ]octane-2-carbonyl)-hydrazinocarbonyl] -(R)-piperidin-l-carboxylic acid tert-butyl ester (16.5 gm , 0.033 mol), methanol (170 ml) and 10% palladium on carbon (3.5 gm) to provide the title compound in 13.5 gm quantity as a pale pink solid and it was used for the next reaction immediately.
Analysis: MS (ES+) CiglfeNsOe = 411.1 (M+l);
Step-3: Preparation of tetrabutylammonium salt of trans-3-[N’-(6-sulfooxy-7-oxo-l,6-diaza-bicyclo [3.2.1] octane-2-carbonyl)-hydrazinocarbonyl] -(R)-piperidin- 1 -carboxylic acid tert-butyl ester:
By using the procedure described in Step-3 of Example- 1 above, and by using trans-3-[N’-(6-hydroxy-7-oxo-l,6-diaza-bicyclo[3.2.1]octane-2-carbonyl)-hydrazinocarbonyl]-(R)-piperidin-1 -carboxylic acid tert-butyl ester (13.5 gm , 0.033 mol), pyridine (70 ml) and pyridine sulfur trioxide complex (26.11 gm, 0.164 mol), 0.5 N aqueous potassium dihydrogen
phosphate solution (400 ml) and tetrabutylammonium sulphate (9.74 gm, 0.033 mol) to provide the title compound in 25 gm quantity as a yellowish solid, in quantitative yield.
Analysis: MS (ES-)
as a salt = 490.0 (M-l) as a free sulfonic acid;
Step-4: trans-sulfuric acid mono-[2-(N’-[(R)-piperidin-3-carbonyl]-hydrazinocarbonyl)-7-oxo-l,6-diaza-bicyclo[3.2.1]oct-6-yl]ester:
By using the procedure described in Step-4 of Example- 1 above, and by using tetrabutylammonium salt of trans-3-[N’-(6-sulfooxy-7-oxo-l,6-diaza-bicyclo[3.2.1]octane-2-carbonyl)-hydrazinocarbonyl]-(R)-piperidin-l-carboxylic acid tert-butyl ester (24 gm , 0.032 mmol), dichloromethane (60 ml) and trifluoroacetic acid (60 ml) to provide the title compound in 10 gm quantity as a white solid, in 79% yield.
Analysis: MS (ES-)= C13H21N5O7S = 390.2 (M-l) as a free sulfonic acid;
HXNMR (DMSO-d6) = 9.97 (d, 2H), 8.32 (br s, 2H), 4.00 (br s, IH), 3.81 (d, IH), 3.10-3.22 (m, 3H), 2.97-3.02 (m, 2H), 2.86-2.91 (m, IH), 2.65-2.66 (m, IH), 1.97-2.03 (m, IH), 1.57-1.88 (m, 7H).
-32.6°, (c 0.5, water).
PATENT
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2015110885
PATENT
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2014135931
| Clinical data | |
|---|---|
| License data | US DailyMed: Zidebactam |
| Legal status | |
| Legal status | Investigational |
| Identifiers | |
| IUPAC name | |
| CAS Number | 1436861-97-0 |
| PubChem CID | 77846445 |
| DrugBank | DB13090 |
| ChemSpider | 44209501 |
| UNII | YPM97423DB |
| ChEMBL | ChEMBL4533605 |
| Chemical and physical data | |
| Formula | C13H21N5O7S |
| Molar mass | 391.40 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
References
- Karvouniaris M, Almyroudi MP, Abdul-Aziz MH, Blot S, Paramythiotou E, Tsigou E, et al. (April 2023). “Novel Antimicrobial Agents for Gram-Negative Pathogens”. Antibiotics. 12 (4). Basel, Switzerland: 761. doi:10.3390/antibiotics12040761. PMC 10135111. PMID 37107124.
ERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
///////ZIDEBACTAM, ANAX LABS, FDA 2026, APPROVALS 2026, Cypsedo, WCK-5107, WCK 5107, YPM97423DB
see………http://apisynthesisint.blogspot.in/2015/11/wck-5107-in-phase-1-from-wockhardt.html
SEE BACTAM SERIES…………..http://apisynthesisint.blogspot.in/p/bactam-series.html
C1C[C@H](CNC1)C(=O)NNC(=O)[C@@H]2CC[C@@H]3C[N@]2C(=O)N3OS(=O)(=O)O
or
O=C(NNC(=O)[C@@H]2CC[C@@H]1CN2C(=O)N1OS(=O)(=O)O)[C@@H]3CCCNC3
C1CC(CNC1)C(=O)NNC(=O)C2CCC3CN2C(=O)N3OS(=O)(=O)[O-].[Na+]
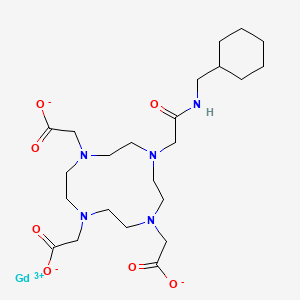
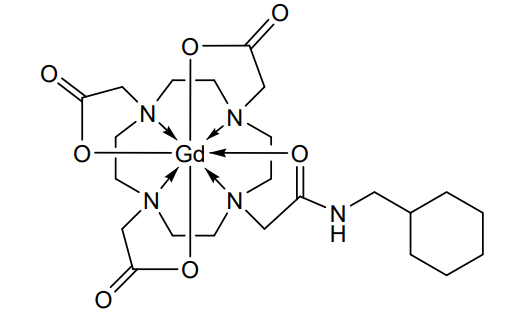
Gadosircoclamide
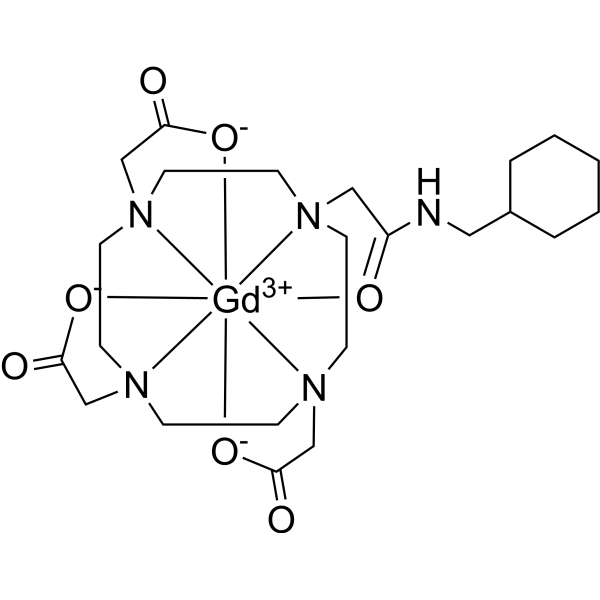
Gadosircoclamide
CAS 1801159-68-1
MF C23H38GdN5O7. MW653.8 g/mol
2-[4,7-bis(carboxylatomethyl)-10-[2-(cyclohexylmethylamino)-2-oxoethyl]-1,4,7,10-tetrazacyclododec-1-yl]acetate;gadolinium(3+)
- [10-[2-[(Cyclohexylmethyl)amino]-2-(oxo-kappaO)ethyl]-1,4,7,10-tetraazacyclododecane-1,4,7-triacetato(3-)-kappaN1,kappaN4,kappaN7,kappaN10,kappaO1,kappaO4,kappaO7]gadolinium
- [2,2′,2”-(10-{2-[(cyclohexylmethyl)amino]-2-oxo-kappaOethyl}-1,4,7,10-tetraazacyclododecane-1,4,7-triyl-kappa4N1,N4,N7,N10)tri(acetato-kappaO)]gadolinium
radiodiagnostic agent, 7V6P6PCM4U
Gadosircoclamide (CAS # 1801159-68-1) is a specialized gadolinium-based coordination complex used primarily as a magnetic resonance imaging (MRI) contrast agent. It is designed to enhance image contrast, help visualize lesions, and accurately track abnormalities during diagnostic scans.
SYN
R=H
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2015105352&_cid=P11-MPXG49-92326-1
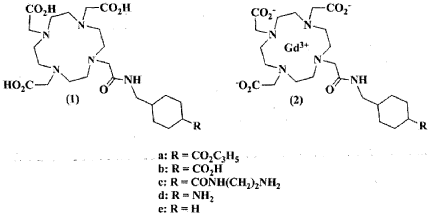
4) Synthesis of le.
DG3A-(f BuO)3 (3.0 g, 5.8 mmol) was added to a solution of 2-chlorocyclohexylmethylacetamid (1.2 g, 6.4 mmol) in acetanitrile (30 mL) prepared according to the conventional literature method (Cho, SD; Song, SY; Kim, . H.; Zhao, BX; Ahn, C; J oo, WH; Yoon, YJ; Falck, JR; Shin, DS B / (or. Chem. St. 2004, 25, 415) . The solution was stirred at room temperature for 24 hours. Solid impurities were removed by filtration, and the filtrate was evaporated under vacuum to obtain an oil phase residue. Subsequently, column chromatography on a silica phase (gradient elution: CH₂C1₂ to 10 % MeOH -CH₂Cl₂ , R f = 0.4 ( MeOH/ CH₂Cl₂ = A 1:9 mixture was performed and evaporated under reduced pressure to obtain a yellowish-white solid. As described in the preparation of the above Id, deprotection with TFA was performed to obtain a yellowish-white solid as a product. Yield: 2.4 g (82%). 1H R ( O): δ = 3.74/3.57 (m, 8H, -NCH₂CO₂- ) , 3.30 (m, 10H , overlapped -NCH₂CH₂N- ( 8H) & -CONHCH₂- ( 2H )), 3.10 (m, 8H, -NCH₂CH₂N- ) , 1.98/1.44/1.27 (in, 4H, -CH₂- , cyclohexyl ) , 1.88 (m, 1H, -NHCH₂CH- ) . Anal . Calculated for C₂₂H₃₅N₅₀ 0 7 · 3CF 3 C00H ■ 3H 2 0 : C, 38.14; H, 5.49; N, 7.94. Found: C, 37.83; H, 5.76; N, 8.44. MALDI-T0F MS (m/z): Calcd for C22H39N5O7,: 485.28, Found: 486.42 ([MH] + ), 508.44 ([MNa] + ).
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
[1].
Kim T, et al., Gadolinium complex comprising do3a-tranexamic acid conjugate. WO2015105352
////////////gadosircoclamide. anax labs, radiodiagnostic agent, 7V6P6PCM4U
Cipepofol
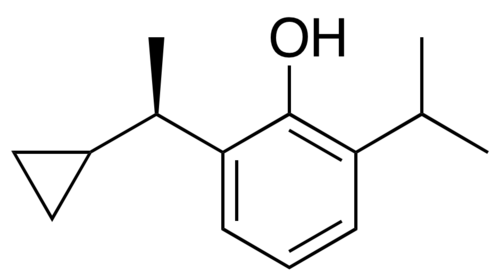
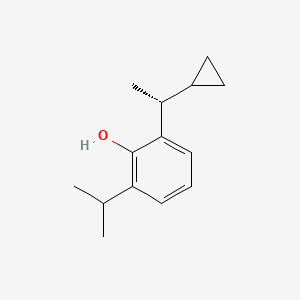
Cipepofol
CAS1637741-58-2
MW 204.31 g/mol MF C14H20O
2-[(1R)-1-cyclopropylethyl]-6-propan-2-ylphenol
FDA 2026, APPROVALS 2026, Cypsedo, HSK 3486, CS-0064163, GTPL 10812, HSK-3486, HY-116152, M3WGS532VY
- OriginatorSichuan Haisco Pharmaceutical
- ClassCyclopropanes; General anaesthetics; Phenols; Small molecules
- Mechanism of ActionGABA A receptor agonists
- RegisteredAnaesthesia; Sedation
- 10 Apr 2026Sichuan Haisco Pharmaceutical plans a phase III trial for Anesthesia (In Children, In adolescents) (IV) in May 2026 (NCT07510945)
- 28 Aug 2024No recent reports of development identified for preclinical development in Sedation in USA (IV, Infusion)
- 01 Aug 2024Zhongda Hospital plans a clinical trial for Sedation (IV) in August 2024 (NCT06538883)
To induce general anesthesia in adults undergoing surgery
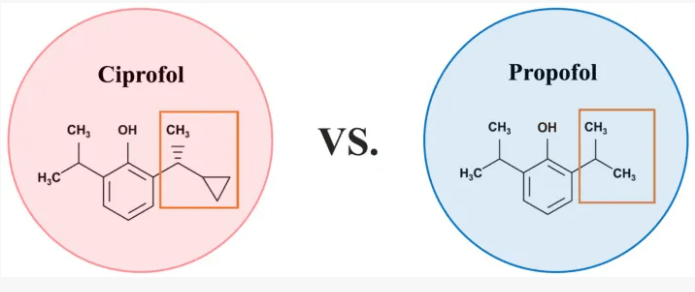
Cipepofol (also known as ciprofol or HSK3486) is a novel, short-acting intravenous anesthetic and sedative. As a structural analog of propofol, it targets \(GABA_{A}\) receptors but is 4 to 6 times more potent. It offers faster recovery, improved cardiovascular stability, and significantly less injection pain than propofol.
Key Clinical Advantages
- Superior Efficacy: Requires a lower dose to achieve the same sedative depth as propofol.
- Better Safety Profile: Associated with a lower incidence of injection pain, reduced respiratory depression, and better hemodynamic (blood pressure) stability.
- Fast Acting: Characterized by rapid onset and quick recovery times, making it ideal for procedures like gastrointestinal endoscopy, bronchoscopy, and general anesthesia induction.
Recent Developments
- FDA Approval: Cipepofol (sold under the brand name CYPSEDO) officially received U.S. FDA marketing approval, becoming the first China-originated innovative intravenous anesthetic to enter the global market.
- Ongoing Trials: Clinical trials and post-marketing studies are actively evaluating its safety in specific populations, such as elderly patients and children.
Cipepofol (INNTooltip International Nonproprietary Name, USANTooltip United States Adopted Name), also known as ciprofol or by its developmental code name HSK3486, is a general anesthetic related to propofol which is used for anesthesia and sedation.[1][2][3][4] The drug is used by intravenous infusion.[1] A short-acting and highly selective γ-aminobutyric acid positive allosteric modulator,[5] ciprofol is 4 to 6 times more potent than other phenol derivatives such as propofol or fospropofol.[6]
In May 2026, cipepofol was approved by the US FDA.[7] Manufactured by Haisco Pharmaceutical Group of Chengdu, Sichuan, China, ciprofol underwentphase I and II trials in Australia and China.[8][9][10] In these early studies, ciprofol was comparable in efficacy to propofol and was associated with fewer adverse events.[4][6][11][12][13][14][15][16][17][18]
Physical properties
Ciprofol is an optically active 2,6-disubstituted alkylphenol with a cyclopropylethyl group incorporated at the second carbon atom. This cyclopropyl group increases the steric effects and introduces stereoselective effects over its anesthetic properties. These properties appear to increase the anesthetic potency of ciprofol, when compared with propofol.[9]
Medical use
Ciprofol is used for the intravenous induction of general anesthesia.[3][4] Studies published in 2022 and 2023 found it was efficacious as a general anesthetic in patients undergoing gynecological surgery[6][11] and kidney transplantation,[19] as well as for endoscopic procedures such as bronchoscopy,[15][20] esophagogastroduodenoscopy and colonoscopy.[21][22]
Ciprofol has also been used for sedation of critically ill patients undergoing mechanical ventilation in the intensive care unit,[23] as well as for the treatment of agitation and delirium in that patient population.[24] When combined with mild therapeutic hypothermia, ciprofol may also be useful as a cerebral protective agent in the setting of cerebral ischemia-reperfusion injury.[25]
Experimental use
In experimental models of isoproterenol-induced myocardial infarction (using mice as subjects), ciprofol appears to protect the heart against oxidative damage, inflammation and apoptosis of cardiac muscle cells.[26]
SYN
https://patentscope.wipo.int/search/en/detail.jsf?docId=US428011434&_cid=P12-MPW0XO-91017-1
PAT
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2014180305&_cid=P12-MPW0R4-87054-1
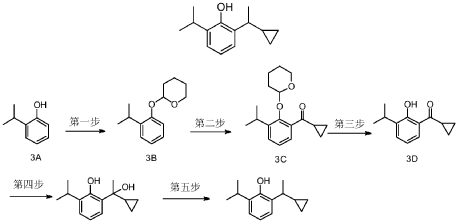
Example 16
[-cyclopropylethyl] -6 -isopropylphenol (compound 16)
2- [(lR)-l-cyclopropylethyl]-6-isopropyl -phenol
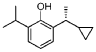
Preparation methods of Examples 16-17:
2-(1-Cyclopropylethyl-6-isopropylphenol (compound 3) 600 mg was used for resolution. Preparation conditions: (Instrument: Agilent 1260/CH-Y-J0404; Column: CHIRALPAK OJ-H (4.6 mm < 250 mm, 5 μm) No.: OJ-H-27; Mobile phase: A: isopropanol, B: n-hexane; Flow rate: 1.0 mL/min; Back pressure: 100 bar; Column temperature: 35°C; Wavelength: 210 nm; Period: 10 min)
Two optical isomers were obtained after separation: peak 1 (retention time: 10.72 min, 280 mg, pale yellow liquid, ee%=99%) and peak 2 (retention time: 13.58 min, 280 mg, pale yellow liquid, ee%=99%).
峰 1 : MS m/z(ESI): 203.1(Ml).
toMR (400 MHz,CDCl3 ) : δ 7.14(dd, 1H), δ 7.08(dd, 1H), 6.91 (t, 1H), 4.93 (s, 1H), 3.22-3.14(m, 1H), 2.55-2.48 (m, 1H), 1.33 (d, 6H), 1.28 (d, 3H), 1.10-1.05 (m, 1H), 0.60-0.58 (m, 1H), 0.49-0.46 (m, 1H), 0.25-0.18 (m, 2H).
峰 2: MS m/z(ESI): 203.1(Ml).
iHNMR (400 MHz,CDCl3) : 57.14(dd, 1H), δ 7.08(dd, 1H), 6.93 (t, 1H), 4.93 (s 1H), 3.22-3.15(m, 1H), 2.55-2.48 (m, 1H), 1.32 (d, 6H), 1.28 (d, 3H), 1.10-1.04 (m, 1H), 0.60-0.58 (m, 1H), 0.49-0.46 (m, 1H), 0.25-0.18 (m, 2H).
PAT
- Phenol derivative compound, methods of preparing said compound, pharmaceutical composition comprising said compound and use thereofPublication Number: BR-112015028212-B1Priority Date: 2013-05-09
- Phenol derivative and its preparation method and application in medicinePublication Number: CN-104507899-APriority Date: 2013-05-09
- Phenol derivative and preparation method and use in medicine thereofPublication Number: US-9517988-B2Priority Date: 2013-05-09Grant Date: 2016-12-13
- Phenol derivative and preparation method and use in medicine thereofPublication Number: EP-2995604-B1Priority Date: 2013-05-09Grant Date: 2019-07-10
- Phenol derivative, its production method and medicinal applicationPublication Number: JP-6431155-B2Priority Date: 2013-05-09Grant Date: 2018-11-28
- Phenol derivative, preparation method and medical application thereofPublication Number: CN-104507899-BPriority Date: 2013-05-09Grant Date: 2016-11-30
- Phenol derivative and method of preparation and medical use thereofPublication Number: ES-2746987-T3Priority Date: 2013-05-09Grant Date: 2020-03-09
- Phenol derivative and preparation method and use in medicine thereofPublication Number: US-2016060197-A1Priority Date: 2013-05-09
- Isopropyl phenol derivative and preparation method thereofPublication Number: WO-2016026459-A1Priority Date: 2014-08-22
- Phenol derivative and preparation method and use in medicine thereofPublication Number: AU-2014264103-B2Priority Date: 2013-05-09Grant Date: 2018-03-22
- Phenol derivative and preparation method and use in medicine thereofPublication Number: AU-2014264103-A1Priority Date: 2013-05-09
- Phenol derivative and preparation method and use in medicine thereofPublication Number: AU-2014264103-C1Priority Date: 2013-05-09Grant Date: 2018-08-02
- Phenol derivative and preparation method and use in medicine thereofPublication Number: EP-2995604-A1Priority Date: 2013-05-09
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
References
- “Sichuan Haisco Pharmaceutical”. AdisInsight. 28 August 2024. Retrieved 1 October 2025.
- “Ciprofol (Cipepofol): A γ-Aminobutyric Acid Receptor Agonist for Induction of Anesthesia”. Chemistry and Pharmacology of Drug Discovery. Wiley. 2024. pp. 251–274. doi:10.1002/9781394225156.ch12. ISBN 978-1-394-22512-5. Retrieved 1 October 2025.
- Wang X, Wang X, Liu J, Zuo YX, Zhu QM, Wei XC, et al. (March 2022). “Effects of ciprofol for the induction of general anesthesia in patients scheduled for elective surgery compared to propofol: a phase 3, multicenter, randomized, double-blind, comparative study”. European Review for Medical and Pharmacological Sciences. 26 (5): 1607–1617. PMID 35302207.
- Zeng Y, Wang DX, Lin ZM, Liu J, Wei XC, Deng J, et al. (February 2022). “Efficacy and safety of HSK3486 for the induction and maintenance of general anesthesia in elective surgical patients: a multicenter, randomized, open-label, propofol-controlled phase 2 clinical trial”. European Review for Medical and Pharmacological Sciences. 26 (4): 1114–1124. PMID 35253166.
- Liao J, Li M, Huang C, Yu Y, Chen Y, Gan J, et al. (2022). “Pharmacodynamics and Pharmacokinetics of HSK3486, a Novel 2,6-Disubstituted Phenol Derivative as a General Anesthetic”. Frontiers in Pharmacology. 13 830791. doi:10.3389/fphar.2022.830791. PMC 8851058. PMID 35185584.
- Chen BZ, Yin XY, Jiang LH, Liu JH, Shi YY, Yuan BY (August 2022). “The efficacy and safety of ciprofol use for the induction of general anesthesia in patients undergoing gynecological surgery: a prospective randomized controlled study”. BMC Anesthesiology. 22 (1) 245. doi:10.1186/s12871-022-01782-7. PMC 9347095. PMID 35922771.
- “Novel Drug Approvals for 2026”. U.S. Food and Drug Administration. 29 May 2026. Retrieved 31 May 2026.
- Lu M, Liu J, Wu X, Zhang Z (2023). “Ciprofol: A Novel Alternative to Propofol in Clinical Intravenous Anesthesia?”. BioMed Research International. 2023 7443226. doi:10.1155/2023/7443226. PMC 9879693. PMID 36714027.
- Qin L, Ren L, Wan S, Liu G, Luo X, Liu Z, et al. (May 2017). “Design, Synthesis, and Evaluation of Novel 2,6-Disubstituted Phenol Derivatives as General Anesthetics”. Journal of Medicinal Chemistry. 60 (9): 3606–3617. doi:10.1021/acs.jmedchem.7b00254. PMID 28430430.
- Nair A, Seelam S (2022). “Ciprofol- a game changing intravenous anesthetic or another experimental drug!”. Saudi Journal of Anaesthesia. 16 (2): 258–259. doi:10.4103/sja.sja_898_21. PMC 9009555. PMID 35431734.
- Man Y, Xiao H, Zhu T, Ji F (March 2023). “Study on the effectiveness and safety of ciprofol in anesthesia in gynecological day surgery: a randomized double-blind controlled study”. BMC Anesthesiology. 23 (1) 92. doi:10.1186/s12871-023-02051-x. PMC 10039513. PMID 36964501.
- Chen X, Guo P, Yang L, Liu Z, Yu D (2022). “Comparison and Clinical Value of Ciprofol and Propofol in Intraoperative Adverse Reactions, Operation, Resuscitation, and Satisfaction of Patients under Painless Gastroenteroscopy Anesthesia”. Contrast Media & Molecular Imaging. 2022 9541060. doi:10.1155/2022/9541060. PMC 9314164. PMID 35935320.
- Zhong J, Zhang J, Fan Y, Zhu M, Zhao X, Zuo Z, et al. (May 2023). “Efficacy and safety of Ciprofol for procedural sedation and anesthesia in non-operating room settings”. Journal of Clinical Anesthesia. 85 111047. doi:10.1016/j.jclinane.2022.111047. PMID 36599219. S2CID 255468218.
- Liang P, Dai M, Wang X, Wang D, Yang M, Lin X, et al. (June 2023). “Efficacy and safety of ciprofol vs. propofol for the induction and maintenance of general anaesthesia: A multicentre, single-blind, randomised, parallel-group, phase 3 clinical trial”. European Journal of Anaesthesiology. 40 (6): 399–406. doi:10.1097/EJA.0000000000001799. PMC 10155686. PMID 36647565.
- Luo Z, Tu H, Zhang X, Wang X, Ouyang W, Wei X, et al. (March 2022). “Efficacy and Safety of HSK3486 for Anesthesia/Sedation in Patients Undergoing Fiberoptic Bronchoscopy: A Multicenter, Double-Blind, Propofol-Controlled, Randomized, Phase 3 Study”. CNS Drugs. 36 (3): 301–313. doi:10.1007/s40263-021-00890-1. PMC 8927014. PMID 35157236.
- Hu C, Ou X, Teng Y, Shu S, Wang Y, Zhu X, et al. (November 2021). “Sedation Effects Produced by a Ciprofol Initial Infusion or Bolus Dose Followed by Continuous Maintenance Infusion in Healthy Subjects: A Phase 1 Trial”. Advances in Therapy. 38 (11): 5484–5500. doi:10.1007/s12325-021-01914-4. PMC 8523013. PMID 34559359.
- Teng Y, Ou M, Wang X, Zhang W, Liu X, Liang Y, et al. (September 2021). “Efficacy and safety of ciprofol for the sedation/anesthesia in patients undergoing colonoscopy: Phase IIa and IIb multi-center clinical trials”. European Journal of Pharmaceutical Sciences. 164 105904. doi:10.1016/j.ejps.2021.105904. PMID 34116176.
- Zhu Q, Luo Z, Wang X, Wang D, Li J, Wei X, et al. (April 2023). “Efficacy and safety of ciprofol versus propofol for the induction of anesthesia in adult patients: a multicenter phase 2a clinical trial”. International Journal of Clinical Pharmacy. 45 (2): 473–482. doi:10.1007/s11096-022-01529-x. PMC 10147789. PMID 36680620.
- Qin K, Qin WY, Ming SP, Ma XF, Du XK (July 2022). “Effect of ciprofol on induction and maintenance of general anesthesia in patients undergoing kidney transplantation”. European Review for Medical and Pharmacological Sciences. 26 (14): 5063–5071. PMID 35916802.
- Wu B, Zhu W, Wang Q, Ren C, Wang L, Xie G (2022). “Efficacy and safety of ciprofol-remifentanil versus propofol-remifentanil during fiberoptic bronchoscopy: A prospective, randomized, double-blind, non-inferiority trial”. Frontiers in Pharmacology. 13 1091579. doi:10.3389/fphar.2022.1091579. PMC 9812563. PMID 36618929.
- Li J, Wang X, Liu J, Wang X, Li X, Wang Y, et al. (August 2022). “Comparison of ciprofol (HSK3486) versus propofol for the induction of deep sedation during gastroscopy and colonoscopy procedures: A multi-centre, non-inferiority, randomized, controlled phase 3 clinical trial”. Basic & Clinical Pharmacology & Toxicology. 131 (2): 138–148. doi:10.1111/bcpt.13761. PMC 9543620. PMID 35653554.
- Long YQ, Feng CD, Ding YY, Feng XM, Liu H, Ji FH, et al. (2022). “Esketamine as an Adjuvant to Ciprofol or Propofol Sedation for Same-Day Bidirectional Endoscopy: Protocol for a Randomized, Double-Blind, Controlled Trial With Factorial Design”. Frontiers in Pharmacology. 13 821691. doi:10.3389/fphar.2022.821691. PMC 8975265. PMID 35370640.
- Liu Y, Yu X, Zhu D, Zeng J, Lin Q, Zang B, et al. (May 2022). “Safety and efficacy of ciprofol vs. propofol for sedation in intensive care unit patients with mechanical ventilation: a multi-center, open label, randomized, phase 2 trial”. Chinese Medical Journal. 135 (9): 1043–1051. doi:10.1097/CM9.0000000000001912. PMC 9276409. PMID 34924506.
- Liu GL, Wu GZ, Ge D, Zhou HJ, Cui S, Gao K, et al. (2023). “Efficacy and safety of ciprofol for agitation and delirium in the ICU: A multicenter, single-blind, 3-arm parallel randomized controlled trial study protocol”. Frontiers in Medicine. 9 1024762. doi:10.3389/fmed.2022.1024762. PMC 9868613. PMID 36698817.
- Wang YC, Wu MJ, Zhou SL, Li ZH (January 2023). “Protective effects of combined treatment with ciprofol and mild therapeutic hypothermia during cerebral ischemia-reperfusion injury”. World Journal of Clinical Cases. 11 (3): 487–492. doi:10.12998/wjcc.v11.i3.487. PMC 9923870. PMID 36793629.
- Yang Y, Xia Z, Xu C, Zhai C, Yu X, Li S (2022). “Ciprofol attenuates the isoproterenol-induced oxidative damage, inflammatory response and cardiomyocyte apoptosis”. Frontiers in Pharmacology. 13 1037151. doi:10.3389/fphar.2022.1037151. PMC 9723392. PMID 36483733.
- Vittori A, Di Fabio C, Cascella M, Marinangeli F, Francia E, Mascilini I, et al. (January 2026). “Advantages of Ciprofol with Special Consideration of Pediatric Anesthesia”. Children (Basel, Switzerland). 13 (2). doi:10.3390/children13020188. PMC 12939459. PMID 41749542.
- Liu SB, Yao X, Tao J, Yang JJ, Zhao YY, Liu DW, et al. (March 2023). “Population total and unbound pharmacokinetics and pharmacodynamics of ciprofol and M4 in subjects with various renal functions”. British Journal of Clinical Pharmacology. 89 (3): 1139–1151. doi:10.1111/bcp.15561. PMID 36217805. S2CID 252818288.
- Hu Y, Li X, Liu J, Chen H, Zheng W, Zhang H, et al. (December 2022). “Safety, pharmacokinetics and pharmacodynamics of a novel γ-aminobutyric acid (GABA) receptor potentiator, HSK3486, in Chinese patients with hepatic impairment”. Annals of Medicine. 54 (1): 2769–2780. doi:10.1080/07853890.2022.2129433. PMC 9559057. PMID 36217101.
- Li X, Yang D, Li Q, Wang H, Wang M, Yan P, et al. (2021). “Safety, Pharmacokinetics, and Pharmacodynamics of a Single Bolus of the γ-aminobutyric Acid (GABA) Receptor Potentiator HSK3486 in Healthy Chinese Elderly and Non-elderly”. Frontiers in Pharmacology. 12 735700. doi:10.3389/fphar.2021.735700. PMC 8430033. PMID 34512361.
- Ding YY, Long YQ, Yang HT, Zhuang K, Ji FH, Peng K (December 2022). “Efficacy and safety of ciprofol for general anaesthesia induction in elderly patients undergoing major noncardiac surgery: A randomised controlled pilot trial”. European Journal of Anaesthesiology. 39 (12): 960–963. doi:10.1097/EJA.0000000000001759. PMID 36214498. S2CID 252779399.
- Duan G, Lan H, Shan W, Wu Y, Xu Q, Dong X, et al. (April 2023). “Clinical effect of different doses of ciprofol for induction of general anesthesia in elderly patients: A randomized, controlled trial”. Pharmacology Research & Perspectives. 11 (2) e01066. doi:10.1002/prp2.1066. PMC 9944862. PMID 36811327. S2CID 257098376.
- Yang Y, Xia Z, Xu C, Zhai C, Yu X, Li S (2022). “Ciprofol attenuates the isoproterenol-induced oxidative damage, inflammatory response and cardiomyocyte apoptosis”. Frontiers in Pharmacology. 13 1037151: 1037151. doi:10.3389/fphar.2022.1037151. PMC 9723392. PMID 36483733.
- Bian Y, Zhang H, Ma S, Jiao Y, Yan P, Liu X, et al. (January 2021). “Mass balance, pharmacokinetics and pharmacodynamics of intravenous HSK3486, a novel anaesthetic, administered to healthy subjects”. British Journal of Clinical Pharmacology. 87 (1): 93–105. doi:10.1111/bcp.14363. PMID 32415708. S2CID 218658207.
Further reading
- Bajwa SJ, Vinayagam S, Shinde S, Dalal S, Vennel J, Nanda S (January 2023). “Recent advancements in total intravenous anaesthesia and anaesthetic pharmacology”. Indian Journal of Anaesthesia. 67 (1): 56–62. doi:10.4103/ija.ija_1022_22. PMC 10034929. PMID 36970470.
- Skiljic S, Budrovac D, Cicvaric A, Neskovic N, Kvolik S (February 2023). “Advances in Analgosedation and Periprocedural Care for Gastrointestinal Endoscopy”. Life. 13 (2): 473. Bibcode:2023Life…13..473S. doi:10.3390/life13020473. PMC 9962362. PMID 36836830.
- Wei A, Yang L, Ma S, Jin G, Yang M, Zhou J (November 2022). “A case report of ciprofol overdose during anesthesia/analgesia and literature review: clinical presentation, blood pressure, and management”. The Journal of International Medical Research. 50 (11) 3000605221132466. doi:10.1177/03000605221132466. PMC 9659933. PMID 36366740.
| Clinical data | |
|---|---|
| Other names | Ciprofol; CS-0064163; CS0064163; GTPL10812; GTPL-10812; HSK-3486; HSK3486; HY-116152; HY116152; (R)-2-(1-Cyclopropylethyl)-6-isopropylphenol |
| Routes of administration | Intravenous infusion[1] |
| Drug class | GABAA receptor positive allosteric modulator |
| Pharmacokinetic data | |
| Metabolism | Liver glucuronidation |
| Excretion | Kidney |
| Identifiers | |
| IUPAC name | |
| CAS Number | 1637741-58-2 |
| PubChem CID | 86301664 |
| DrugBank | DB16295 |
| ChemSpider | 76794458 |
| UNII | M3WGS532VY |
| KEGG | D12449 |
| ChEMBL | ChEMBL4094894 |
| Chemical and physical data | |
| Formula | C14H20O |
| Molar mass | 204.313 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
////////cipepofol, FDA 2026, APPROVALS 2026, Cypsedo, HSK 3486, CS-0064163, GTPL 10812, HSK-3486, HY-116152, M3WGS532VY, ANAESTHETIC
Fudapirine
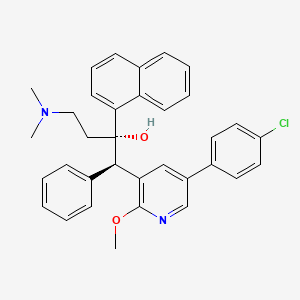
Fudapirine
CAS 1859978-72-5
MFC34H33ClN2O2 MW537.1 g/mol
- (alphaS,betaR)-5-(4-Chlorophenyl)-alpha-[2-(dimethylamino)ethyl]-2-methoxy-alpha-1-naphthalenyl-beta-phenyl-3-pyridineethanol
- 3-Pyridineethanol, 5-(4-chlorophenyl)-alpha-[2-(dimethylamino)ethyl]-2-methoxy-alpha-1-naphthalenyl-beta-phenyl-, (alphaS,betaR)-
(1R,2S)-1-[5-(4-chlorophenyl)-2-methoxy-3-pyridinyl]-4-(dimethylamino)-2-naphthalen-1-yl-1-phenylbutan-2-ol
(1R,2S)-1-[5-(4-chlorophenyl)-2-methoxypyridin-3-yl]-4-(dimethylamino)-2-(naphthalen-1-yl)-1-phenylbutan-2-ol
antibacterial, Sudapyridine, WX-081, WX 081, 7X86XPE5TG,
- A Phase III Study of Oral Sudapyridine (WX-081) Tablets in Rifampicin-Resistant Pulmonary Tuberculosis PatientsCTID: NCT05824871Phase: Phase 3Status: RecruitingDate: 2025-06-29
- Drug-Drug Interaction and Food Effect of Sudapyridine(WX-081) With Itraconazole and Rifampin in Healthy Chinese AdultsCTID: NCT06701136Phase: Phase 1Status: Not yet recruitingDate: 2025-02-12
- Sudapyridine (WX-081) in Healthy VolunteersCTID: NCT06117514Phase: Phase 1Status: CompletedDate: 2023-11-07
- Evaluation of Early Bactericidal Activity and Safety in Pulmonary Tuberculosis With WX-081CTID: NCT04608955Phase: Phase 2Status: CompletedDate: 2023-09-11
Fudapirine (also known as sudapyridine or WX-081) is a novel, next-generation antimycobacterial drug primarily being developed to treat tuberculosis (TB). It belongs to a chemical class called diarylquinolines, making it a close analogue of bedaquiline, an already established drug used for drug-resistant tuberculosis.Key Facts About FudapirinePrimary Function: It displays powerful anti-mycobacterial activity against Mycobacterium tuberculosis strains.
Mechanism: As a diarylquinoline, it selectively inhibits bacterial ATP synthase, effectively cutting off the energy supply that the tuberculosis bacteria need to survive and replicate.Development Stage: According to pharmacology databases like the IUPHAR/BPS Guide to PHARMACOLOGY, the drug has advanced into Phase III clinical evaluation.Official Naming: While initially designated as WX-081, the World Health Organization (WHO) assigned it the International Nonproprietary Name (INN) fudapirine in early 2025.Other Applications: Beyond standard TB, researchers are investigating its therapeutic potential against non-tuberculous mycobacterial (NTM) infections.
PAT
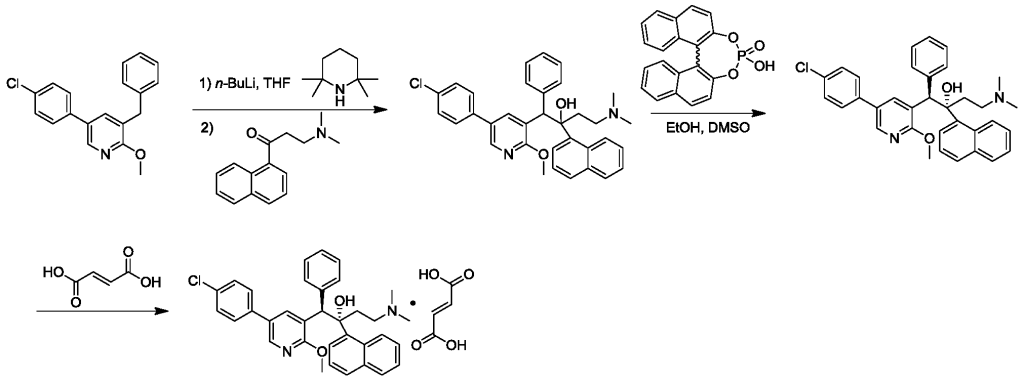
Step 5:
Synthesis of
1-(5-(4-chlorophenyl)-2-methoxypyridin-3-yl)-4-(dimethylamino)-2-(naphth-1-yl)-1-phenylbut-2-ol
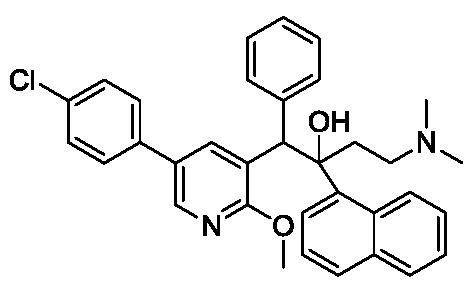
Titration of n-butyllithium : Under nitrogen protection, 1.00 g
of diphenylacetic acid (Alfa, 4.71 mmol) was added to 10 mL of tetrahydrofuran to form a colorless and transparent solution. A hexane solution of
n-butyllithium was slowly added dropwise to this solution using a syringe. The solution was observed to turn yellow locally during the addition, but the yellow color quickly disappeared. The endpoint was reached when a yellow solution formed after one drop and did not fade within half a minute. The volumes of
n-butyllithium were recorded (1.927 mL and 1.985 mL, with an average volume of 1.95 mL). Therefore, the concentration of
the n-butyllithium hexane solution used was 2.42 mol/L.
[0171]TMP (2.74 kg, 19.3 mol) was dissolved in anhydrous tetrahydrofuran (12 L). The reaction temperature was cooled to -65°C using a dry ice-acetone bath, and then
n-butyllithium (8 L, 19.3 mol, 2.42 mol/L n-hexane solution) was added dropwise. The temperature was controlled between -20°C and -78°C. The reaction system was observed to gradually change color from light yellow to red to deep red, eventually forming a yellow suspension. Stirring was continued at this temperature for 30 minutes. Then, the reaction temperature was lowered to -75°C to -80°C, and over 4–6 hours, a solution of
3-benzyl-5-(4-chlorophenyl)-2-methoxypyridine (4.08 kg, 12.9 mol) in anhydrous tetrahydrofuran (6 L) was slowly added dropwise. The temperature was maintained between -65°C and -78°C, and a mild exothermic reaction with a deep red color was observed. After the initial addition was complete, a solution of 3-(dimethylamino)-1-(naphth-1-yl)propyl-1-one (3.26 kg, 12.9 mol, 90% purity) in anhydrous tetrahydrofuran (2.0 L) was slowly added dropwise over 2–4 hours. The system exhibited significant exothermic activity, and the flow rate was controlled to maintain the temperature at -65°C to -78°C. After the addition was complete, the temperature was maintained at -65°C to -78°C, and stirring was continued for another half hour. HPLC analysis showed that the content of
3-benzyl-5-(4-chlorophenyl)-2-methoxypyridine was less than 10%. The reaction solution was slowly added to a saturated
ammonium chloride solution (40 L) for quenching, and the mixture was separated. The aqueous phase was extracted with ethyl acetate (30 L). The combined organic phases were washed and separated with saturated brine (30 L). The organic phases were concentrated under reduced pressure at 40–50 °C to obtain a yellow oily crude product (13.5 kg). The crude product was stirred in a mixed solvent of ethyl acetate/n-heptane (4 L, 1/4) at 5–15 °C for 16 hours to precipitate a white solid. The product was filtered, and the filter cake was slurried with ethanol (4 L × 2). After filtration, the filter cake was vacuum dried to constant weight (50 °C, 24–48 hours) to obtain the target compound 1-(5-(4-chlorophenyl)-2-methoxypyridin-3-yl)-4-(dimethylamino)-2-(naphth-1-yl)-1-phenylbut-2-ol (1.83 kg, yield 23.23%), a white solid. HPLC identification showed that isomer A accounted for 88.3% and isomer B accounted for 4.8%.
1 H NMR (400MHz, CDCl 3 )δ: 8.85(d,J=2.3Hz,1H),8.64(d,J=8.7Hz,1H),8.32(d,J=2.4Hz,1H),7. 98-7.86(m,2H),7.72-7.61(m,2H),7.57(d,J=8.4Hz,2H),7.54-7.43(m,3 H),7.33(t,J=7.8Hz,1H),7.20-7.17(m,2H),6.95-6.87(m,3H),5.85(s,1 H),4.17(s,3H),2.60-2.51(m,1H),2.19-2.04(m,2H),2.01-1.97(m,7H).
[0172]Step 6: Synthesis of (1R,2S)-1-(5-(4-chlorophenyl)-2-methoxypyridin-3-yl)-4-(dimethylamino)-2-(naphth-1-yl)-1-phenylbut-2-ol compound I-2
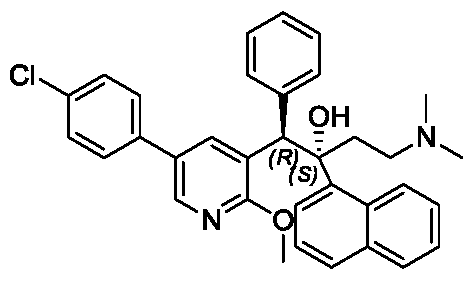
[0175]Two parallel batches were prepared: R-(-)-binaphthol phosphate (519.3 g, 1.49 mol) was suspended in DMSO (1.0 L) and heated to 50 °C with stirring until dissolved and clear. 1-(5-(4-chlorophenyl)-2-methoxypyridin-3-yl)-4-(dimethylamino)-2-(naphthaleneethanol-1-yl)-1-phenylbutyl-2-ol (910 g, 1.49 mol, isomer A content 88.3%) was added to an ethanol (24 L) solution, and the DMSO (1.0 L) solution of R-(-)-binaphthol phosphate prepared above was added dropwise over 1–2 hours with stirring (196 rpm). Undissolved particulate compounds began to dissolve, but a more viscous emulsion formed. After the addition was complete, the reaction mixture was stirred at 15–35 °C for 16 hours. The reaction solution was heated to reflux in an oil bath and refluxed for 1 hour. Heating was then stopped, and the reaction solution was cooled to 15–35°C and stirred for 16 hours. The reaction solution was filtered (two batches were combined for processing). Due to the high viscosity of the solids, filtration was slow. The filter cake was slurried three times with ethanol (20 liters). The combined organic phases were concentrated to constant weight to obtain a yellow oily substance (5 kg). Water (10 liters) and ethyl acetate (5 liters) were added to this crude product. The pH of the system was adjusted to 11 with a 10% cold
sodium hydroxide aqueous solution, and stirring was continued for 1 hour. Then, the mixture was separated. A large amount of solid precipitated from the system. The solid obtained by filtration was isomer A (350 g, purity 97%, white solid). The filtrate was concentrated under reduced pressure at 50°C to constant weight, and ethanol (1.0 L) was added. The mixture was stirred at 15-35°C for 16 hours, filtered, and the filter cake was washed three times with ethanol (400 mL) to obtain a white solid. The solid was dried under vacuum to constant weight (50°C, 24-48 hours) to obtain compound I-2 (400 g, purity 95%, ee value greater than 99.5%, yield 24%), a white solid.
1 H NMR (400MHz, CDCl
3 )δ: 8.85(d,J=2.3Hz,1H),8.64(d,J=8.7Hz,1H),8.32(d,J=2.4Hz,1H),7. 98-7.86(m,2H),7.72-7.61(m,2H),7.57(d,J=8.4Hz,2H),7.54-7.43(m,3 H),7.33(t,J=7.8Hz,1H),7.20-7.17(m,2H),6.95-6.87(m,3H),5.85(s,1 H),4.17(s,3H),2.60-2.51(m,1H),2.19-2.04(m,2H),2.01-1.97(m,7H).
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- In vitro and Intracellular Antibacterial Activity of Sudapyridine (WX-081) Against TuberculosisPublication Name: Infection and Drug ResistancePublication Date: 2023-01PMCID: PMC9840375PMID: 36647451DOI: 10.2147/idr.s390187
- Discovery and preclinical profile of sudapyridine (WX-081), a novel anti-tuberculosis agentPublication Name: Bioorganic & Medicinal Chemistry LettersPublication Date: 2022-09-01PMID: 35636648DOI: 10.1016/j.bmcl.2022.128824
- Sudapyridine (WX-081), a Novel Compound against Mycobacterium tuberculosisPublication Name: Microbiology SpectrumPublication Date: 2022-02-23PMCID: PMC8849072PMID: 35170994DOI: 10.1128/spectrum.02477-21
PAT
- Pharmaceutical composition for treating mycobacterium tuberculosis infection and application thereofPublication Number: CN-117243956-APriority Date: 2023-09-08
- Pharmaceutical composition containing clarithromycin and Shu Da pyridinePublication Number: CN-116763807-APriority Date: 2023-08-08
- Pharmaceutical composition containing clarithromycin and Shu Da pyridinePublication Number: CN-116763807-BPriority Date: 2023-08-08Grant Date: 2025-04-11
- A kind of preparation method of pyridine derivative compound and its intermediate and crystal formPublication Number: CN-108473428-BPriority Date: 2016-01-13Grant Date: 2021-07-23
/////////fudapirine, anax labs, antibacterial, Sudapyridine, WX-081, WX 081, 7X86XPE5TG,
Fosrugocrixan
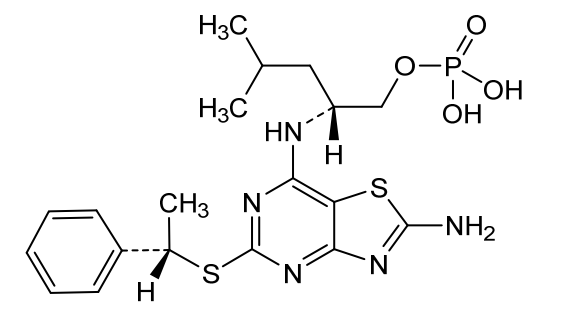
Fosrugocrixan
CAS 2408145-38-8
MF C19H26N5O4PS2, MW483.5 g/mol
[(2R)-2-[[2-amino-5-[(1S)-1-phenylethyl]sulfanyl-[1,3]thiazolo[4,5-d]pyrimidin-7-yl]amino]-4-methylpentyl] dihydrogen phosphate
- (2R)-2-[(2-amino-5-{[(1S)-1-phenylethyl]sulfanyl}[1,3]thiazolo[4,5-d]pyrimidin-7-yl)amino]-4-methylpentyl dihydrogen phosphate
- 1-Pentanol, 2-[[2-amino-5-[[(1S)-1-phenylethyl]thio]thiazolo[4,5-d]pyrimidin-7-yl]amino]-4-methyl-, 1-(dihydrogen phosphate), (2R)-
(2R)-2-[(2-amino-5-{[(1S)-1-phenylethyl]sulfanyl}[1,3]thiazolo[4,5-d]pyrimidin-7-yl)amino]-4-methylpentyl dihydrogen phosphate
CX3C chemokine receptor 1 (CX3CR1) antagonist, antiinflammatory, 4ZXD25SC4S, KAND-145, KAND 145
- OriginatorKancera
- DeveloperNovakand Pharma
- ClassAnti-inflammatories; Antineoplastics; Small molecules
- Mechanism of ActionChemokine CXCL13 inhibitors
- Phase IOvarian cancer
- PreclinicalChronic lymphocytic leukaemia
- No development reportedInflammation
- 22 Sep 2025Kancera is now called Novakand Pharma
- 28 Apr 2025No recent reports of development identified for preclinical development in Ovarian-cancer in Sweden (IV)
- 03 May 2024Efficacy and adverse event data from a phase I trials in healthy volunteers released by Kancera
Fosrugocrixan (also known by its developmental code KAND145) is a novel, small-molecule drug candidate acting as a selective antagonist for CX3C chemokine receptor 1 (CX3CR1), commonly known as the fractalkine receptor.
Key Characteristics and Mechanism
- Drug Class: It represents a first-in-class small molecule immune modulator.
- Phosphate Prodrug: Fosrugocrixan is designed as a soluble phosphate prodrug. Once inside the body (in vivo), it converts into its active drug form, rugocrixan (formerly KAND567).
- Mechanism of Action: By blocking the CX3CR1 fractalkine pathway, it controls and prevents the trafficking of disease-promoting immune cells. This blockage provides potent anti-inflammatory activity.
Clinical Development and Targets
The drug is being actively developed by Novakand Pharma (a company formerly known as Kancera). Its primary therapeutic targets span several conditions driven by runaway inflammation and immune responses:
- Cardiovascular Diseases: Specifically targeted to manage conditions where hyper-inflammation damages tissue (such as post-myocardial infarction or heart conditions).
- Autoimmune & Inflammatory Diseases: Evaluated for broad anti-inflammatory potential.
- Oncology: Investigated for its ability to regulate the tumor microenvironment.
SYN
WO 2020008064
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2020008064&_cid=P21-MPQB4Y-95682-1
SYN
Karlström et al. J. Med. Chem., 2013, 56, 3177-3190
https://pubs.acs.org/doi/10.1021/jm3012273
PAT
(2R)-2-[(2-Amino-5-{[(1S)-1-phenylethyl]sulfanyl}[1,3]thiazolo[4,5-d]pyrimidin-7-yl)amino]-4-methylpentyl dihydrogen phosphate (B), are known to act as antagonists of the fractalkine receptor (CX3CR1) (Karlström et al. J. Med. Chem., 2013, 56, 3177-3190; WO 2020/008064)
PAT
https://patentscope.wipo.int/search/en/detail.jsf?docId=US336567291&_cid=P21-MPQAYT-90868-1
Example 1
Preparation of (2R)-2-[(2-Amino-5-{[(1S)-1-phenylethyl]sulfanyl}[1,3]thiazolo[4,5-d]pyrimidin-7-yl)amino]-4-methylpentyl dihydrogen phosphate
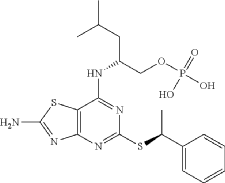
Phosphorus oxychloride (337 mg, 2.2 mmol) was dissolved in THE (0.75 mL) and water (25 mg, 1.4 mmol) was added. The mixture was cooled in an ice-bath and pyridine (111 mg, 113 μL, 1.4 mmol) was added followed by (2R)-2-[(2-amino-5-{[(1S)-1-phenylethyl]sulfanyl}-[1,3] thiazolo[4,5-d]pyrimidin-7-yl)amino]-4-methylpentan-1-ol hydrochloride (110 mg, 0.25 mmol) (Karlstr6m S., et al., J. Med. Chem., 2013, 56, 3177-3190; WO 2006/107258). The reaction mixture was stirred at ice-bath temperature for 1 h. To a mixture of phosphorus oxychloride (337 mg, 2.2 mmol) and water (25 mg, 1.4 mmol) in THE was added, at ice-bath temperature pyridine (111 mg, 113 μL, 1.4 mmol). Half of this mixture was added to the reaction mixture described above. The reaction mixture was stirred at ice-bath temperature for another 1 h. Water (3 mL) was added and the reaction mixture was stirred for 15 min at ice-bath temperature and 20 min at room temperature. DCM (3 mL) was added and the phases were separated. The aqueous phase was extracted with another portion of DCM (3 mL) and the organic phases were combined. At this point the product started to precipitate as a pale-yellow gum in the organic phase. MeOH was added and the now homogeneous solution was transferred to a round-bottomed flask and was evaporated to yield 120 mg of crude product, which according to HPLC was ca. 93% pure. The crude material was dissolved in a MeOH/water mixture and the pH was adjusted to about 6-7 with 1 M NaOH. The material was purified by preparative HPLC (basic method). The pure fractions were pooled, evaporated, and dried in vacuum. The product was assumed to be the diammonium salt after purification. 1H NMR (600 MHz, CD 3OD) δ H ppm 7.43-7.47 (m, 2H) 7.30-7.35 (m, 2H) 7.20-7.24 (m, 1H) 5.08 (q, J=7.03 Hz, 1H) 4.59-4.68 (m, 1H) 3.92 (ddd, J=10.12, 5.67, 4.30 Hz, 1H) 3.88 (dt, J=10.12, 4.94 Hz, 1H) 1.74 (d, J=7.03 Hz, 3H) 1.71-1.79 (m, 1H) 1.68 (ddd, J=13.87, 9.54, 5.67 Hz, 1H) 1.57 (ddd, J=13.87, 8.54, 5.33 Hz, 1H) 0.98 (d, J=6.71 Hz, 3H) 0.96 (d, J=6.56 Hz, 3H). MS (ESI +) m/z 484 [M+H] +.
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Phosphate and phosphonate derivatives of 7-amino-5-thio-thiazolo[4,5-d]pyrimidines and their use in treating conditions associated with elevated levels of cx3cr1 and/or cx3cl1Publication Number: EP-3818065-B1Priority Date: 2018-07-06Grant Date: 2023-03-15
- Phosphate and phosphonate derivatives of 7-amino-5-thio-thiazolo[4,5-d]pyrimidine and their use in treating conditions associated with elevated levels of CX3CR1 and/or CX3CL1Publication Number: KR-102736870-B1Priority Date: 2018-07-06Grant Date: 2024-12-03
- Phosphate and phosphonate derivatives of 7-amino-5-thio-thiazolo[4,5-d]pyrimidines and their use in treating conditions associated with elevated levels of cx3cr1 and/or cx3cl1Publication Number: US-2021340167-A1Priority Date: 2018-07-06
- Phosphate and phosphonate derivatives of 7-amino-5-thio-thiazolo[4,5-D]pyrimidine and their use in the treatment of conditions associated with elevated levels of cx3cr1 and/or cx3cl1 – Patent Application 20070123333Publication Number: JP-7506653-B2Priority Date: 2018-07-06Grant Date: 2024-06-26
- Phosphate and phosphonate derivatives of 7-amino-5-thio-thiazolo[4,5-d]pyrimidines and their use in treating conditions associated with elevated levels of CX3CR1 and/or CX3CL1Publication Number: US-11339183-B2Priority Date: 2018-07-06Grant Date: 2022-05-24
- Phosphate and phosphonate derivatives of 7-amino-5-thio-thiazolo-[4,5-D]-pyrimidines and their uses in the treatment of conditions associated with high levels of CX3CR1 and/or CX3CL1Publication Number: IL-279818-B1Priority Date: 2018-07-06
- PHOSPHATE AND PHOSPHONATE DERIVATIVES OF 7-AMINO-5-THIO-THIAZOLO[4,5-D]PYRIMIDINE AND THEIR USE IN THE TREATMENT OF CONDITIONS ASSOCIATED WITH INCREASED LEVELS OF CX3CR1 AND/OR CX3CL1Publication Number: HR-P20230532-T1Priority Date: 2018-07-06
- Phosphate and phosphonate derivatives of 7-amino-5-thio-thiazolo[4,5-d]pyrimidines and their use in treating conditions associated with elevated levels of CX3CR1 and/or CX3CL1Publication Number: US-12060380-B2Priority Date: 2018-07-06Grant Date: 2024-08-13
- 7-Amino-5-thio-thiazolo [4,5-D] Pyrimidine phosphate and phosphonate derivatives and their use in therapeutic conditions associated with elevated levels of CX3CR1 and / or CX3CL1Publication Number: JP-2021530474-APriority Date: 2018-07-06
- Phosphate and phosphonate derivatives of 7-amino-5-thio-thiazolo-[4,5-D]-pyrimidines and their uses in the treatment of conditions associated with high levels of CX3CR1 and/or CX3CL1Publication Number: IL-279818-B2Priority Date: 2018-07-06
- Phosphate and phosphonate derivatives of 7-amino-5-thio-thiazolo[4,5-d]pyrimidines and their use in treating conditions associated with elevated levels of cx3cr1 and/or cx3cl1Publication Number: US-2021292349-A1Priority Date: 2018-07-06
- Phosphate and phosphonate derivatives of 7-amino-5-thio-thiazolo [4,5-D ] pyrimidine modulators of CX3CR1 receptor and medical uses thereofPublication Number: CN-112867725-BPriority Date: 2018-07-06Grant Date: 2024-09-27
- New usePublication Number: US-2025195548-A1Priority Date: 2023-12-19
- Fractalkine receptor antagonists for use in the prevention of thrombus formation and/or growthPublication Number: EP-4593833-A1Priority Date: 2023-12-19
- Fractalkine receptor antagonists for use in the prevention of thrombus formation and/or growthPublication Number: WO-2025133022-A1Priority Date: 2023-12-19
- New usePublication Number: US-2025195549-A1Priority Date: 2023-12-19
- New treatments of viral infectionsPublication Number: WO-2021224494-A1Priority Date: 2020-05-08
////////fosrugocrixan, anax labs, CX3C chemokine receptor 1 (CX3CR1) antagonist, antiinflammatory, 4ZXD25SC4S, KAND-145, KAND 145

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO



