MIRDAMETINIB


MIRDAMETINIB
391210-10-9
Chemical Formula: C16H14F3IN2O4
Molecular Weight: 482.19
PD0325901; PD 0325901; PD-325901; mirdametinib
FDA APPROVED 2/11/2025, Gomekli, To treat neurofibromatosis type 1 who have symptomatic plexiform neurofibromas not amenable to complete resection
IUPAC/Chemical Name: (R)-N-(2,3-dihydroxypropoxy)-3,4-difluoro-2-((2-fluoro-4-iodophenyl)amino)benzamide
SpringWorks Therapeutics (a spin out of Pfizer ) is developing mirdametinib, a second-generation, non-ATP competitive, allosteric MEK1 and MEK2 inhibitor derived from CI-1040, for treating type 1 neurofibromatosis (NF1) and advanced solid tumors. In June 2021, a phase I/II trial was initiated in patients with low grade glioma.
- OriginatorPfizer
- DeveloperAstraZeneca; BeiGene; BIOENSIS; Pfizer; SpringWorks Therapeutics; St. Jude Childrens Research Hospital; University of Oxford
- ClassAniline compounds; Anti-inflammatories; Antineoplastics; Benzamides; Immunotherapies; Small molecules
- Mechanism of ActionMAP kinase kinase 1 inhibitors; MAP kinase kinase 2 inhibitors
- Orphan Drug StatusYes – Neurofibromatosis 1
- Phase IINeurofibromatosis 1
- Phase I/IIGlioma
- Phase ISolid tumours
- PreclinicalChronic obstructive pulmonary disease
- No development reportedCervical cancer
- DiscontinuedBreast cancer; Cancer; Colorectal cancer; Malignant melanoma; Non-small cell lung cancer
- 22 Jul 2021SpringWorks Therapeutics receives patent allowance for mirdametinib from the US Patent and Trademark Office for the treatment of Neurofibromatosis type 1-associated plexiform neurofibromas
- 16 Jun 2021SpringWorks Therapeutics and St. Jude Children’s Research Hospital agree to develop mirdametinib in USA for glioma
- 15 Jun 2021Efficacy and safety data from the phase IIb RENEU trial for Neurofibromatosis type 1-associated plexiform neurofibromas released by SpringWorks Therapeutics
Mirdametinib, sold under the brand name Gomekli, is a medication used for the treatment of people with neurofibromatosis type 1.[1] Mirdametinib is a kinase inhibitor.[1][2] It is taken by mouth.[1]
The most common adverse reactions in adults include rash, diarrhea, nausea, musculoskeletal pain, vomiting, and fatigue.[3] The most common grade 3 or 4 laboratory abnormalities include increased creatine phosphokinase.[3] The most common adverse reactions in children include rash, diarrhea, musculoskeletal pain, abdominal pain, vomiting, headache, paronychia, left ventricular dysfunction, and nausea.[3] The most common grade 3 or 4 laboratory abnormalities include decreased neutrophil count and increased creatine phosphokinase.[3]
Mirdametinib was approved for medical use in the United States in February 2025.[1][3]
SCHEME
SIDE CHAIN
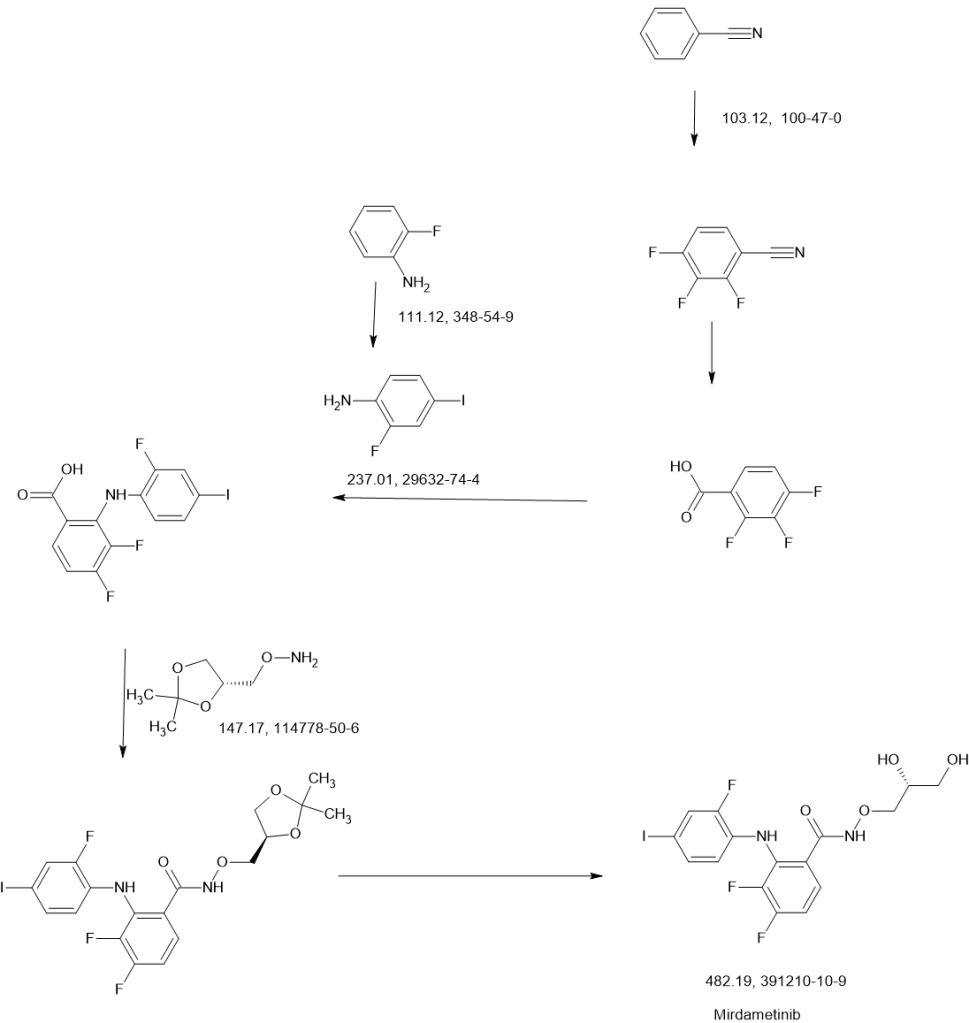
MAIN

Medical uses
Mirdametinib is indicated for the treatment of people with neurofibromatosis type 1 who have symptomatic plexiform neurofibromas not amenable to complete resection.[1]
Adverse effects
The most common adverse reactions in adults include rash, diarrhea, nausea, musculoskeletal pain, vomiting, and fatigue.[3] The most common grade 3 or 4 laboratory abnormalities include increased creatine phosphokinase.[3] The most common adverse reactions in children include rash, diarrhea, musculoskeletal pain, abdominal pain, vomiting, headache, paronychia, left ventricular dysfunction, and nausea.[3] The most common grade 3 or 4 laboratory abnormalities include decreased neutrophil count and increased creatine phosphokinase.[3]
Mirdametinib can cause left ventricular dysfunction and ocular toxicity including retinal vein occlusion, retinal pigment epithelial detachment, and blurred vision.[3]
History
The efficacy of mirdametinib was evaluated in ReNeu (NCT03962543), a multicenter, single-arm trial in 114 participants aged two years of age and older (58 adults, 56 pediatric participants) with symptomatic, inoperable NF1-associated plexiform neurofibromas causing significant morbidity.[3] An inoperable plexiform neurofibromas was defined as a plexiform neurofibromas that could not be completely surgically removed without risk for substantial morbidity due to encasement or close proximity to vital structures, invasiveness, or high vascularity.[3]
The US Food and Drug Administration (FDA) granted the application for mirdametinib priority review, fast track, and orphan drug designations along with a priority review voucher.[3]
Society and culture
Legal status
Mirdametinib was approved for medical use in the United States in February 2025.[3][4][5]
PATENT
US-11066358
On July 20, 2021, SpringWorks Therapeutics announced that the United States Patent and Trademark Office (USPTO) has issued US11066358 , directed to mirdametinib , the Company’s product candidate in development for several oncology indications, including as a monotherapy for patients with neurofibromatosis type 1-associated plexiform neurofibromas (NF1-PN) and was assigned to Warner-Lambert Company (a subsidiary of Pfizer ).This patent was granted on July 20, 2021, and expires on Feb 17, 2041. Novel crystalline forms of mirdametinib and compositions comprising them are claimed.
| N—((R)-2,3-dihydroxypropoxy)-3,4-difluoro-2-(2-fluoro-4-iodo-phenylamino)-benzamide (“mirdametinib”, or “PD-0325901”) is a small molecule drug which has been designed to inhibit mitogen-activated protein kinase kinase 1 (“MEK1”) and mitogen-activated protein kinase kinase 2 (“MEK2”). MEK1 and MEK2 are proteins that play key roles in the mitogen-activated protein kinase (“MAPK”) signaling pathway. The MAPK pathway is critical for cell survival and proliferation, and overactivation of this pathway has been shown to lead to tumor development and growth. Mirdametinib is a highly potent and specific allosteric non-ATP-competitive inhibitor of MEK1 and MEK2. By virtue of its mechanism of action, mirdametinib leads to significantly inhibited phosphorylation of the extracellular regulated MAP kinases ERK1 and ERK2, thereby leading to impaired growth of tumor cells both in vitro and in vivo. In addition, evidence indicates that inflammatory cytokine-induced increases in MEK/ERK activity contribute to the inflammation, pain, and tissue destruction associated with rheumatoid arthritis and other inflammatory diseases. |
Example 1: Production of Essentially Pure Form IV
Lab Scale Production of Essentially Pure Form IV
| All reactions were performed in toluene other than otherwise stated. Triflic anhydride gave the best yield. |
[TABLE-US-00002]TABLE 1 Coupling Agents for Step 1Entry No.Coupling AgentYieldNotes 1Mesyl Chloridedid not react 2Benzyl chloride27Had to heat 70° C. for 166 hr34-fluorobenzensulfonylchloride27Ran 93 hrs. at 70° C.44-chlorobenzensulfonylchloride35Complete after 68 hrs. 50° C.5Tosyl Chloride36Had to heat to 70° C. for 164 hrs6Benzyl chloride52study solvent effects: DMF, DMSO, NMP – all similar DMSO fastest all complete after 110 hrs., heated to 70° C. after 66 hrs.7Triflic anhydride91Cooled to −74° C. |
| [TABLE-US-00004]TABLE 3 Yields for base deprotection ReagentYield* Methyl hydrazine85-95% Anhydrous NH3 (sparged)78-90% Anhydrous NH3 (50 psi)80-92% Aqueous NH390-97% *from PD-0333760 |
Step 2: Fluoride Displacement
Pilot Plant Preparation of Essentially Pure Form IV
Step 1: Preparation of “Side Chain”, PD-0337792
Step 2: Preparation of PD-0315209
Step 3: Preparation of PD-0325901
Polymorph Transformation
| 21.4 kg PD-0315209, 9.7 kg CDI (1.05 equiv.), 91 kg solution of 9.7% PD-0337792 in Toluene (1.1 equiv.) were used and resulted in 12.74 kg of PD-0325901 (assay 99.4%, 100% Form IV, Yield 48%). |
PATENT
WO2006134469 , claiming methods of preparing MEK inhibitor, assigned to Warner-Lambert Co .
https://patents.google.com/patent/WO2006134469A1/enThe compound Λ/-[(R)-2,3-dihydroxy-propoxy]-3,4-difluoro-2-(2-fluoro-4-iodo-phenylamino)- benzamide represented by formula 1

i is a highly specific non-ATP-competitive inhibitor of MEK1 and MEK2. The compound of formula ± (Compound I) is also known as the compound PD 0325901. Compound I is disclosed in WO 02/06213; WO 04/045617; WO 2005/040098; EP 1262176; U.S. Patent Application Pub. No. 2003/0055095 A1 ; U.S. Patent Application Pub. No. 2004/0054172 A1; U.S. Patent Application Pub. No. 2004/0147478 A1 ; and U.S. Patent Application No. 10/969,681, the disclosures of which are incorporated herein by reference in their entireties.Numerous mitogen-activated protein kinase (MAPK) signaling cascades are involved in controlling cellular processes including proliferation, differentiation, apoptosis, and stress responses. Each MAPK module consists of 3 cytoplasmic kinases: a mitogen-activated protein kinase (MAPK), a mitogen-activated protein kinase kinase (MAPKK), and a mitogen-activated protein kinase kinase kinase (MAPKKK). MEK occupies a strategic downstream position in this intracellular signaling cascade catalyzing the phosphorylation of its MAP kinase substrates, ERK1 and ERK2. Anderson et al. “Requirement for integration of signals from two distinct phosphorylation pathways for activation of MAP kinase.” Nature 1990, v.343, pp. 651-653. In the ERK pathway, MAPKK corresponds with MEK (MAP kinase ERK Kinase) and the MAPK corresponds with ERK (Extracellular Regulated Kinase). No substrates for MEK have been identified other than ERK1 and ERK2. Seger et al. “Purification and characterization of mitogen-activated protein kinase activator(s) from epidermal growth factor-stimulated A431 cells.” J. Biol. Chem., 1992, v. 267, pp. 14373-14381. This tight selectivity in addition to the unique ability to act as a dual-specificity kinase is consistent with MEK’s central role in integration of signals into the MAPK pathway. The RAF-MEK-ERK pathway mediates proliferative and anti-apoptotic signaling from growth factors and oncogenic factors such as Ras and Raf mutant phenotypes that promote tumor growth, progression, and metastasis. By virtue of its central role in mediating the transmission of growth- promoting signals from multiple growth factor receptors, the Ras-MAP kinase cascade provides molecular targets with potentially broad therapeutic applications.One method of synthesizing Compound I is disclosed in the above-referenced WO 02/06213 andU.S. Patent Application Pub. No. 2004/0054172 A1. This method begins with the reaction of 2-fluoro-4- iodo-phenylamine and 2,3,4-trifluoro-benzoic acid in the presence of an organic base, such as lithium diisopropylamide, to form 3,4-difluoro-2-(2-fluoro-4-iodo-phenylamino)-benzoic acid, which is then reacted with (R)-0-(2,2-dimethyl-[1,3]dioxolan-4-ylmethyl)-hydroxylamine in the presence of a peptide coupling agent (e.g., diphenylphosphinic chloride) and a tertiary amine base (e.g., diisopropylethylamine). The resulting product is hydrolyzed under standard acidic hydrolysis conditions (e.g., p-TsOH in MeOH) to provide Compound 1. (R)-O-(2,2-dimethyl-[1,3]dioxolan-4-ylmethyl)-hydroxylamine is prepared by reaction of [(4S)-2,2-dimethyl-1,3-dioxolan-4-yl]methanol with N-hydroxyphthalimide in the presence of Ph3P and diethyl azodicarboxylate.Another method of synthesizing Compound I, which is disclosed in the above-referenced U.S.Patent Application No. 10/969,681, comprises reaction of 3,4-difluoro-2-(2-fluoro-4-iodo-phenylamino)- benzoic acid with (R)-O-(2,2-dimethyl-[1,3]dioxolan-4-ylmethyl)-hydroxylamine in the presence of N1N1– carbonyldiimidazole. The resulting product is hydrolyzed with aqueous acid and crystallized to provide polymorphic form IV of Compound I.Although the described methods are effective synthetic routes for small-scale synthesis of Compound I, there remains a need in the art for new synthetic routes that are safe, efficient and cost effective when carried out on a commercial scale.The present invention provides a new synthetic route including Steps I through Step III to the MEK inhibitor Λ/-[(R)-2,3-dihydroxy-propoxy]-3,4-difluoro-2-(2-fluoro-4-iodo-phenylamino)-benzamide (Compound I).Step I: Preparation of 0-{r(4RV2.2-dimethyl-1.3-dioxolan-4-ynmethyl}hydroxylanπine (6) The method of the present invention comprises a novel Step I of preparing of 0-{[(4R)-2,2- dimethyl-1 ,3-dioxolan-4-yl]methyl}hydroxylamine (6) from [(4S)-2,2-dimethyl-1 ,3-dioxoIan-4-yl]methanol (1) through the formation of [(4R)-2,2-dimethyl-1 ,3-dioxolan-4-yl]methyl trifluoromethanesulfonate (3) and its coupling with N-hydroxyphthalimide (4) to afford 2-{[(4R)-2,2-dimethyl-1 ,3-dioxolan-4-yl]methoxy}-1 H- isoindole-1 ,3(2H)-dione (5), which is subsequently de-protected to give 6 as shown in Scheme 1.Scheme 1



The reaction of compound (1) with trifluoromethanesulfonic anhydride (2) is carried out in the presence of a non-nucleophilic base, such as, for example, a tertiary organic amine, in an aprotic solvent at a temperature of from -5O0C to 50C, preferably, at a temperature less than -150C, to form triflate (3). A preferred tertiary organic amine is triethylamine, and a preferred solvent is toluene. Treatment of triflate (3) with N-hydroxyphthalimide (4) furnishes phthalimide (5), which can be isolated if desired. However, in order to minimize processing time and increase overall yield, 0-{[(4R)- 2,2-dimethyl-1,3-dioxolan-4-yl]methyl}hydroxylamine (6) can be prepared in a one-pot process with no phthalimide (S) isolation. Cleavage of the phthalimide function could be achieved by methods known in the art, for example, by hydrazinolysis. However, the use of less hazardous aqueous or anhydrous ammonia instead of methyl hydrazine (CH3NHNH2) is preferred.Step II: Preparation of 3.4-difluoro-2-(2-fluoro-4-iodophenylamino)-benzoic acid (9) As shown in Scheme 2, Step Il of the method of the present invention provides 3,4-difluoro-2-(2- fluoro-4-iodophenylamino)-benzoic acid (9).Scheme 2

Preparation of compound (9) can be carried out by reacting compound (7), wherein X is halogen, or O-SC^R^ or 0-P(3O)(OR^, wherein R^ is alkyl or aryl, with compound (8) optionally in a solvent, and in the presence of from about 1 mol equivalent to about 10 mol equivalents of at least one base, wherein the base is selected from: a Group I metal cation hydride or a Group 2 metal cation hydride, including lithium hydride, sodium hydride, potassium hydride, and calcium hydride, a Group I metal cation dialkylamide or a Group 2 metal cation dialkylamide, including lithium diisopropylamide, a Group I metal cation amide or a Group 2 metal cation amide, including lithium amide, sodium amide, potassium amide, a Group I metal cation alkoxide or a Group 2 metal cation alkoxide, including sodium ethoxide, potassium terf-butoxide, and magnesium ethoxide, and a Group I metal cation hexamethyldisilazide, including lithium hexamethyldisilazide; for a time, and at a temperature, sufficient to yield compound (9).Preferably, preparation of compound (9) is carried out by reacting compound (7), wherein X is halogen, more preferably, X is fluorine, in an aprotic solvent with compound (8) in the presence of from about 3 mol equivalents to about 5 mol equivalents of a Group I metal cation amide at a temperature of from 2O C to 55°C, more preferably, at a temperature from 45°C to 55°C. A catalytic amount of Group I metal cation dialkylamide can be added if necessary. A preferred Group I metal cation amide is lithium amide, a preferred Group I metal cation dialkylamide is lithium diisopropylamide, and a preferred solvent is tetrahydrofuran. Preferably, the reaction is performed by adding a small amount of compound (7) and compound (8) to lithium amide in tetrahydrofuran followed by slow continuous addition of the remaining portion. This procedure minimizes the risk of reactor over-pressurization due to gas side product (ammonia) generation.Step III: Preparation of N-((RV2.3-dihydroxypropoxy)-3.4-difluoro-2-(2-fluoro-4-iodo-phenylamino)- benzamide (Compound I)Compound I can be obtained by coupling 0-{[(4R)-2,2-dimethyl-1,3-dioxolan-4- yl]methyl}hydroxylamine (6) with 3,4-difluoro-2-(2-fluoro-4-iodophenylamino)-benzoic acid (9) using a carboxylic acid activating reagent such as, for example, COCI2, S(O)C^, S(O)2Cl2, P(O)Cl3, triphenylphosphine/diethylazodicarboxylate, diphenylphosphinic chloride, N, N’-dicyclohexylcarbodiimide, (benzotriazol-1 -yloxy)tripyrolidinophosphonium hexafluorophosphate, (benzotriazol-1 – yloxy)tris(dimethylamino)phosphonium hexafluorophosphate, N-ethyl-N’-(3- dimethylaminopropyl)carbodiimide hydrochloride, or 1,1′-carbonyldiimidazole (CDI).A preferred carboxylic acid activating reagent is 1,1′-carbonyldimidazole (CDI) shown in Scheme 3. Preparation of the desirable polymorphic Form IV of Compound I using CDI is described in the above- referenced U.S. Patent Application No. 10/969,681.Scheme 3

10

10 11 Compound IIn according to the present invention, the method was modified to include the advantageous procedure for product purification and isolation, which procedure is performed in single-phase systems such as, for example, toluene/acetonitrile for the first isolation/crystallization and ethanol/toluene for the second recrystallization. Water addition, implemented in the previous procedure, was omitted to avoid the two-phase crystallization from the immiscible water-toluene system that caused inconsistent product purity. The one-phase procedure of the present invention provides consistent control and removal of un- reacted starting material and side products. Alternatively, Compound I can be obtained by coupling 0-{[(4R)-2,2-dimethyl-1,3-dioxolan-4- yl]methyl}hydroxylamine (6) with 3,4-difluoro-2-(2-fluoro-4-iodophenylamino)-benzoic acid (9) using thionyl chloride (SOCI2) as shown in Scheme 4.Scheme 4


Compound IExamplesThe reagents and conditions of the reactions described herein are merely illustrative of the wide variety of starting materials, their amounts and conditions which may be suitably employed in the present invention as would be appreciated by those skilled in the art, and are not intended to be limiting in any way.HPLC (Conditions A): 10 μL injection volume onto Agilent Zorbax RX-C18 150 mm x 4.6 mm x 3.5 μm column at 30°C column temperature, 1.0 mL/min flow rate and detection at 246 nm. Mobile phase A (v/v): 25 mM Acetate Buffer, pH 6.0; Mobile phase B (v/v): Acetonitrile, and Linear Gradient Table:

Sample Preparation: Dilute 100 μL reaction mixture to 10 mL with acetonitrile. Mix in a vial 200 μL of this sample solution with 300 μL carbonate buffer pH 10.0 and 300 μL solution of 2-mercaptopyridine in acetonitrile (18 mM), heat the vial for 10 minutes at 500C and dilute to 1:1 ratio in mobile phase A.GC (Conditions B): 1 μL injection onto an RTX-5 column (30 m x 0.25 mm x 0.25 μm) with initial oven temperature of 120°C for 2 min. to final temperature of 250°C in 15°C/minute ramping and a final time of 2.33 min; Flow rate: 1 mL/min.HPLC (Conditions C): 5 μL injection onto Phenomenex Luna C18(2) 150 mm x 4.6 mm x 3μm column ; flow rate : 1.0 mL/min; detection at 225 nm; mobile phase A: 95/5 v/v Water/Acetonitrile with 0.1% Trifluoroacetic acid (TFA), mobile phase B: 5/95 v/v Water/Acetonitriie with 0.1% TFA; Linear Gradient Table:

Sample preparation: Dilute 1 ml_ reaction mixture to 100 mL with acetonitrile and dilute 1 mL of this solution to 10 mL with 50:50 Water/Acetonitrile.HPLC (Conditions D): 5 μL injection onto Waters SymmetryShield RP 18, 150 mm x 4.6 mm x 3.5 μm column; flow rate: 1.0 mL/min; detection at 235 nm; mobile phase A: 25 mM Acetate Buffer adjusted to pH 5.5, mobile phase B: Acetonitrile; Linear Gradient Table:

Sample preparation: Dilute 40 μL of reaction mixture in 20 mL acetonitrile.HPLC (Conditions E): 10 μL sample injection onto YMC ODS-AQ 5 μm, 250 mm x 4.6 mm column; flow rate: 1.0 ml_/min; detection at 280 nm; temperature 30°C; mobile phase : 75/25 v/v Acetonitrile/Water with 0.1% Formic acid.Sample preparation: Quench reaction mixture sample with dipropylamine and stir for about 5 minutes before further dilution with mobile phase.DSC measurement was performed using a Mettler-Toledo DSC 822, temperature range 25° to 150°C with 5°C/min heating rate in a 40 μL aluminum pan. Experimental Conditions for Powder X-Rav Diffraction (XRD):A Rigaku Miniflex+ X-ray diffractometer was used for the acquisition of the powder XRD patterns. The instrument operates using the Cu Ka1 emission with a nickel filter at 1.50451 units. The major instrumental parameters are set or fixed at:X-ray: Cu / 30 kV (fixed) / 15 mA (fixed)Divergence Slit: Variable Scattering Slit: 4.2° (fixed) Receiving Slit: 0.3 mm (fixed) Scan Mode: FT Preset Time: 2.0 s Scan Width: 0.050° Scan Axis: 2Theta/Theta Scan Range: 3.000° to 40.000°Jade Software Version: 5.0.36(SP1) 01/05/01 (Materials Data, Inc.) Rigaku Software: Rigaku Standard Measurement for Windows 3.1 Version 3.6(1994-1995) Example 1. Preparation of 0-ffl4R)-2.2-dimethyl-1.3-dioxolan-4-vπmethyl}hvdroxylamine (6)A solution containing [(4S)-2,2-dimethyl-1,3-dioxolan-4-yl]methanol (1) (13.54 ml_, 0.109 mol) (DAISO Co., Ltd., CAS# 22323-82-6) and triethylamine (18.2 ml_, 0.131 mol) in 115 mL toluene was cooled to -15 C, then trifluoromethanesulfonic anhydride (2) (18.34 mL, 30.75 g, 0.109 mol) (Aldrich, Catalog # 17,617-6 ) was added drop wise while maintaining the temperature at less than -15°C. The mixture was then stirred for 2 hours, and transferred to a separate flask containing a mixture (slurry) of N- hydroxyphthalimide (4) (18.99 g, 0.116 mol) (Aldrich, Catalog # H5.370-4) and 18.2 mL (0.13 mol) triethylamine in 95 mL toluene. The resulting mixture was warmed to 20-25°C and stirred for at least 5 hours or until reaction completion (determined by HPLC (Conditions A)). Water (93 mL) was then added to quench the reaction mixture, the phases were separated, and the bottom aqueous layer was discarded. The water quench was repeated two more times resulting in a pale yellow organic layer. The organic layer was heated to 35 C and treated with 36.7 mL ammonium hydroxide solution (contains about 28-29% wt/wt ammonia). The mixture was stirred for at least 12 hours or until the reaction was deemed complete as determined by GC (Conditions B). The water was then removed under reduced pressure by co- distilling it with toluene to about half of the original volume at temperatures around 35-45 C. Toluene (170 mL) was added to the concentrated solution and the distillation was repeated. A sample was drawn for water content determination by Karl Fisher method (using EM Science Aquastar AQV-2000 Titrator with a sample injected to a pot containing methanol and salicylic acid). The distillation was repeated ifl water content was more than 0.1%. The concentrated solution was filtered to remove the white solid side product, and the filtrate was stored as 112mL (98 g) product solution containing 9.7% w/w compound 6 in toluene. This solution was ready for use in the final coupling step (Example 3). Overall chemical yield was 59%. A small sample was evaporated to yield a sample for NMR identification.1H NMR (400 MHz, CDCI3): δ 5.5 (bs, 2H), 4.35 (m, 1H), 4.07 (dd, 1H), 3.77 (m, 2H), 3.69 (dd, 1H), 1.44 (s, 3H), 1.37 (s, 3H).Example 2. Preparation of 3.4-difluoro-2-(2-fluoro-4-iodophenylamino)-benzoic acid (9)A solution of 2-fluoro-4-iodoaniline (8) (16.4 g, 0.069 mol) (Aldrich, Catalog # 30,660-6) and 2,3,4- trifluorobenzoic acid (7) (11.98 g, 0.068 mol) (Aldrich, Cat # 33,382-4) in 38 mL tetrahydrofuran (THF) was prepared and a portion (about 5%) of this solution was added to a stirring slurry of lithium amide (5 g, 0.22 mol) in 40 mL THF at 50-55 C. After about 15-30 min. an exotherm followed by gas release and color change are observed. The remaining portion of the (8) and (7) solution was added slowly over 1-2 hr while maintaining temperatures within 45-55°C. The mixture was stirred until the reaction was deemed complete (by HPLC (Conditions C). The final mixture was then cooled to 20-25°C and transferred to another reactor containing 6 N hydrochloric acid (47 mL) followed by 25 mL acetonitrile, stirred, and the bottom aqueous phase was discarded after treatment with 40 mL 50% sodium hydroxide solution. The organic phase was concentrated under reduced pressure and 57 mL acetone was added. The mixture was heated to 50°C, stirred, and added with 25 mL warm (40-50°C) water and cooled to 25-30°C to allow crystallization to occur (within 1-4 hours). Once the crystallization occurred, the mixture was further cooled to 0 to -5°C and stirred for about 2 hours. The solid product was filtered and the wet cake was dried in vacuum oven at about 55°C. Overall chemical yield was 21.4 g, 80%. 1H NMR (400 MHz, (CD3)2SO): δ 13.74 (bs, 1H), 9.15 (m, 1 H), 7.80 (dd, 1H), 7.62 (d, 1H), 7.41 (d, 1H), 7.10 (q, 1H), 6.81 (m, 1H).Example 2B. Preparation of 3.4-difluoro-2-(2-fluoro-4-iodophenylamino)-benzoic acid (9) by the solid addition of lithium amide methodTo a stirring solution of 2,3,4-trifluorobenzoic acid (13) (5.0 g, 28.4 mmol) and 2-fluoro-4- iodoaniline (14) (6.73 g, 28.4 mmol) in MeCN (100 mL), under N2 atmosphere was added lithium amide (2.61 g, 113.6 mmol) in small portions. The reaction mixture was heated to reflux for 45 minutes, cooled to ambient temperature and quenched with 1 N HCI and then water. The yellowish white precipitate was filtered, washed with water. The solid was triturated in CH2CI2 (30 mL) for 1h, filtered and dried in a vacuum oven at 45°C for 14 hours to give 8.Og (72%) of compound (9) as an off-white solid, mp 201.5-203 °C.Example 3. Preparation of N-((R)-2.3-dihvdroxypropoxy)-3.4-difluoro-2-(2-fluoro-4-iodo-phenylamino)- benzamide (Compound \)3,4-Difluoro-2-(2-fluoro-4-iodophenylamino)-benzoic acid (9) (20 g, 0.051 mol) in 100 mL acetonitrile was treated with 1,1′-carbonyldiimidazole (CDI) (8.66 g, 0.053 mol) (Aldrich, Cat # 11,553-3) and stirred for about 2 hours at 20-25°C until the reaction was deemed complete by HPLC (Conditions D). 94 mL (84.9 g) of 9.7% w/w solution of O-{[(4R)-2,2-dimethyl-1,3-dioxolan-4-yl]methyl}hydroxylamine (6) in toluene was then added and stirred for about 4 hours or until the reaction was deemed complete by HPLC (Conditions D). To this mixture was added 66 mL of 5.6 % hydrochloric acid solution, and after stirring, the bottom aqueous phase was discarded. Again 66 mL of 5.6 % hydrochloric acid solution was added to the organic phase and stirred at 20-25°C for 12-18 hours or until the reaction was deemed complete by HPLC (Conditions D). The bottom layer was then discarded and the remaining organic layer was concentrated under reduced pressure to remove about 10-20% solvent, and the volume was adjusted to about 9-11 mL/g with toluene (80 mL). Crude product was then crystallized at 10-15°C. The slurry was allowed to stir for about 2 hours and the crude solid product was filtered, and dried. The dried crude product was recharged to the reactor and dissolved into 150 mL of 5% v/v ethanol/toluene mixture at 55- 67°C. The solution was then clarified at this temperature through filter (line filter) to remove any remaining particulate matter. The solution was then cooled slowly to 5°C to crystallize and stirred for at least 2 h, filtered and dried. The dried solid product was redissolved in EtOH (60 mL) at 35°C, and product was precipitated out by adding water (300 mL) at 35°C followed by cooling to 200C. The slurry was stirred for at least 2 hours to transform the crystals to the desired polymorphic Form IV as determined by DSC and Powder X-ray Diffraction pattern (PXRD). The slurry was filtered and dried under vacuum oven at 70- 90°C to yield the final N-((R)-2,3-dihydroxypropoxy)-3,4-difluoro-2-(2-fluoro-4-iodo-phenylamino)- benzamide (Compound I) product. Overall chemical yield was 13 g, 53%. Melting point (DSC): 112+1° C. Appearance: White to off-white crystals.Shown in Figure 1, PXRD conforms to polymorphic crystal Form IV disclosed in the above mentioned U.S. Patent Application No. 10/969,681 1H NMR (400 MHz, (CD3)2SO): δ 11.89 (bs, 1H), 8.71 (bs, 1H), 7.57 (d, 1H), 7.37 (m, 2H), 7.20 (q, 1H), 6.67 (m, 1H), 4.84 (bs, 1H), 4.60 (m, 1H), 3.87 (m, 1 H), 3.7 (m, 2H), 3.34 (m, 2H).Example 4. Preparation of N-((R)-2.3-dihydroxypropoxyV3.4-difluoro-2-(2-fluoro-4-iodo-phenylanrιinoV benzamide (Compound \)To a stirring solution of 3,4-difluoro-2-(2-fluoro-4-iodophenylamino)-benzoic acid (9) (120 g, 0.30 mol) in a mixture of 1 mL N,N-dimethylformamide and 1000 mL toluene was added thionyl chloride (55 g, 0.462 mol). The mixture was heated to 50-65 C and stirred for 2 hours or until reaction completion as determined by HPLC (Conditions E). The final reaction mixture was then cooled and concentrated under reduced pressure to a slurry keeping the temperature below 35°C. Toluene (600 mL) was added to dissolve the slurry and vacuum distillation was repeated. Additional toluene (600 mL) was added to the slurry dissolving all solids and the solution was then cooled to 5° -10°C. The solution was then treated with O-{[(4R)-2,2-dimethyl-1,3-dioxolan-4-yl]methyl}hydroxylamine (6) (63 g, 0.43 mol) solution in 207 mL toluene followed by potassium carbonate (65 g) and water (200 mL), stirred for at least 2 hours at 20- 25°C. The stirring was stopped to allow phase separation and the bottom phase was discarded. The remaining organic layer was treated with hydrochloric acid solution (7.4%, 240 mL) until pH was less than 1 and stirred for 2 hours. The final reaction mixture was slightly concentrated under vacuum collecting about 100 mL distillate and the resulting organic solution was cooled to 5°C to crystallize the product and filtered. The filter cake was washed with toluene (1000 mL) followed by water (100 mL) and the wet cake (crude product Compound I) was charged back to the flask. Toluene (100 mL), ethanol (100 mL) and water (100 mL) are then added, stirred at 30-35°C for about 15 min, and the bottom aqueous phase was discarded. Water (200 mL) was then added to the organic solution and the mixture was stirred at about 3O C to allow for crystallization. The stirring was continued for 2 hours after product crystallized, then it was further cooled to about 0°C and stirred for at least 2 hours. The slurry was filtered and wet cake was dried under reduced pressure at 55-85°C to yield the final product N-((R)-2,3-dihydroxypropoxy)-3,4- difluoro-2-(2-fluoro-4-iodo-phenylamino)-benzamide (Compound I) product. Overall chemical yield was 86 g, 58%.
PATENT
WO2002/006213 describes crystalline Forms I and II. U.S. Pat. No. 7,060,856 (“the ‘856 patent”)
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2002006213
| Clinical data | |
|---|---|
| Trade names | Gomekli |
| Other names | PD-0325901 |
| AHFS/Drugs.com | Gomekli |
| License data | US DailyMed: Mirdametinib |
| Routes of administration | By mouth |
| Drug class | Antineoplastic |
| ATC code | L01EE05 (WHO) |
| Legal status | |
| Legal status | US: ℞-only[1] |
| Identifiers | |
| CAS Number | 391210-10-9 |
| PubChem CID | 9826528 |
| IUPHAR/BPS | 7935 |
| DrugBank | DB07101 |
| ChemSpider | 10814340 |
| UNII | 86K0J5AK6M |
| KEGG | D11675 |
| ChEBI | CHEBI:9826528 |
| ChEMBL | ChEMBL507361 |
| PDB ligand | 4BM (PDBe, RCSB PDB) |
| Chemical and physical data | |
| Formula | C16H14F3IN2O4 |
| Molar mass | 482.198 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI | |
References
- ^ Jump up to:a b c d e f “Gomekli- mirdametinib capsule; Gomekli- mirdametinib tablet, for suspension”. DailyMed. 27 February 2025. Retrieved 2 April 2025.
- ^ Armstrong AE, Belzberg AJ, Crawford JR, Hirbe AC, Wang ZJ (June 2023). “Treatment decisions and the use of MEK inhibitors for children with neurofibromatosis type 1-related plexiform neurofibromas”. BMC Cancer. 23 (1): 553. doi:10.1186/s12885-023-10996-y. PMC 10273716. PMID 37328781.
- ^ Jump up to:a b c d e f g h i j k l m n “FDA approves mirdametinib for adult and pediatric patients with neurofibromatosis type 1 who have symptomatic plexiform neurofibromas not amenable to complete resection”. U.S. Food and Drug Administration (FDA). 11 February 2025. Archived from the original on 13 February 2025. Retrieved 16 February 2025.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ “UPDATE: SpringWorks Therapeutics Announces FDA Approval of Gomekli (mirdametinib) for the Treatment of Adult and Pediatric Patients with NF1-PN” (Press release). SpringWorks Therapeutics. 12 February 2025. Archived from the original on 13 February 2025. Retrieved 16 February 2025 – via GlobeNewswire News Room.
- ^ “Novel Drug Approvals for 2025”. U.S. Food and Drug Administration (FDA). 14 February 2025. Retrieved 16 February 2025.
External links
- “Mirdametinib (Code C52195)”. NCI Thesaurus.
- Clinical trial number NCT03962543 for “MEK Inhibitor Mirdametinib (PD-0325901) in Patients With Neurofibromatosis Type 1 Associated Plexiform Neurofibromas (ReNeu)” at ClinicalTrials.gov
- Moertel CL, Hirbe AC, Shuhaiber HH, Bielamowicz K, Sidhu A, Viskochil D, Weber MD, Lokku A, Smith LM, Foreman NK, Hajjar FM, McNall-Knapp RY, Weintraub L, Antony R, Franson AT, Meade J, Schiff D, Walbert T, Ambady P, Bota DA, Campen CJ, Kaur G, Klesse LJ, Maraka S, Moots PL, Nevel K, Bornhorst M, Aguilar-Bonilla A, Chagnon S, Dalvi N, Gupta P, Khatib Z, Metrock LK, Nghiemphu PL, Roberts RD, Robison NJ, Sadighi Z, Stapleton S, Babovic-Vuksanovic D, Gershon TR: ReNeu: A Pivotal, Phase IIb Trial of Mirdametinib in Adults and Children With Symptomatic Neurofibromatosis Type 1-Associated Plexiform Neurofibroma. J Clin Oncol. 2025 Feb 20;43(6):716-729. doi: 10.1200/JCO.24.01034. Epub 2024 Nov 8. [Article]
- Weiss BD, Wolters PL, Plotkin SR, Widemann BC, Tonsgard JH, Blakeley J, Allen JC, Schorry E, Korf B, Robison NJ, Goldman S, Vinks AA, Emoto C, Fukuda T, Robinson CT, Cutter G, Edwards L, Dombi E, Ratner N, Packer R, Fisher MJ: NF106: A Neurofibromatosis Clinical Trials Consortium Phase II Trial of the MEK Inhibitor Mirdametinib (PD-0325901) in Adolescents and Adults With NF1-Related Plexiform Neurofibromas. J Clin Oncol. 2021 Mar 1;39(7):797-806. doi: 10.1200/JCO.20.02220. Epub 2021 Jan 28. [Article]
- Ioannou M, Lalwani K, Ayanlaja AA, Chinnasamy V, Pratilas CA, Schreck KC: MEK Inhibition Enhances the Antitumor Effect of Radiotherapy in NF1-Deficient Glioblastoma. Mol Cancer Ther. 2024 Sep 4;23(9):1261-1272. doi: 10.1158/1535-7163.MCT-23-0510. [Article]
- FDA Approved Drug Products: GOMEKLI (mirdametinib) capsules and tablets for oral and oral suspension use (Feb 2024) [Link]
- FDA News: FDA approves mirdametinib for adult and pediatric patients with neurofibromatosis type 1 who have symptomatic plexiform neurofibromas not amenable to complete resection [Link]
////////MIRDAMETINIB, Orphan Drug Status, Neurofibromatosis 1, PHASE 2, PD0325901, PD 0325901, PD-325901, FDA 2025, GOMEKLI, APPROVALS 2025
O=C(NOC[C@H](O)CO)C1=CC=C(F)C(F)=C1NC2=CC=C(I)C=C2F
OTESECONAZOLE
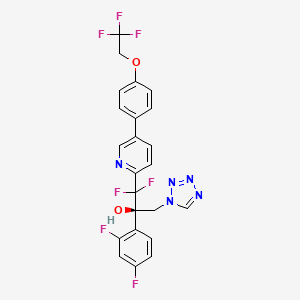
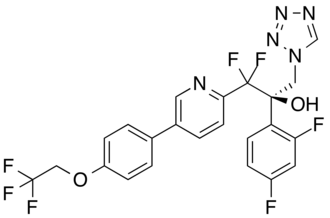
OTESECONAZOLE
VT 1161
オテセコナゾール;
(2R)-2-(2,4-difluorophenyl)-1,1-difluoro-3-(tetrazol-1-yl)-1-[5-[4-(2,2,2-trifluoroethoxy)phenyl]pyridin-2-yl]propan-2-ol
| C23H16F7N5O2 527.4 | |
| Synonyms | VT 1161 Oteseconazole CAS1340593-59-0 |
|---|
Other Names
- (αR)-α-(2,4-Difluorophenyl)-β,β-difluoro-α-(1H-tetrazol-1-ylmethyl)-5-[4-(2,2,2-trifluoroethoxy)phenyl]-2-pyridineethanol
- (2R)-2-(2,4-difluorophenyl)-1,1-difluoro-3-(1H-1,2,3,4-tetrazol-1-yl)- 1-{5-[4-(2,2,2-trifluoroethoxy)phenyl]pyridin-2-yl}propan-2-ol
UPDATE MAY 2022… FDA APPROVED 2022/4/26, Vivjoa
Oteseconazole, sold under the brand name Vivjoa, is a medication used for the treatment of vaginal yeast infections.[1]
It was approved for medical use in the United States in April 2022.[2][3] It was developed by Mycovia Pharmaceuticals.[3]
Names
Oteseconazole is the international nonproprietary name (INN).[4]
Oteseconazole is an azole antifungal used to prevent recurrent vulvovaginal candidiasis in females who are not of reproductive potential.
Oteseconazole, also known as VT-1161, is a tetrazole antifungal agent potentially for the treatment of candidal vaginal infection. VT-1161 Protects Immunosuppressed Mice from Rhizopus arrhizus var. arrhizus Infection. VT-1161 dosed once daily or once weekly exhibits potent efficacy in treatment of dermatophytosis in a guinea pig model.
Oteseconazole has been used in trials studying the treatment of Tinea Pedis, Onychomycosis, Candidiasis, Vulvovaginal, and Recurrent Vulvovaginal Candidiasis.
Mycovia Pharmaceuticals is developing oteseconazole, the lead from a program of metalloenzyme Cyp51 (lanosterol demethylase) inhibitors, developed using the company’s Metallophile technology, for treating fungal infections including onychomycosis and recurrent vulvovaginal candidiasis (RVVC). In July 2021, oteseconazole was reported to be in phase 3 clinical development. Licensee Jiangsu Hengrui Medicine is developing otesaconazole, as an oral capsule formulation, for treating fungal conditions, including RVVC, onychomycosis and invasive fungal infections, in Greater China and planned for a phase 3 trial in April 2021 for treating VVC.
- OriginatorViamet Pharmaceuticals
- DeveloperMycovia Pharmaceuticals; Viamet Pharmaceuticals
- ClassAntifungals; Foot disorder therapies; Pyridines; Small molecules; Tetrazoles
- Mechanism of Action14-alpha demethylase inhibitors
- PreregistrationVulvovaginal candidiasis
- Phase IIOnychomycosis
- No development reportedTinea pedis
- 01 Jun 2021Preregistration for Vulvovaginal candidiasis (In adolescents, In adults, In children, Recurrent) in USA (PO)
- 01 Jun 2021Mycovia intends to launch otesaconazole (Recurrent) for Vulvovaginal candidiasis in the US in early 2022
- 06 Jan 2021Interim efficacy and adverse events data from a phase III ultraVIOLET trial in Vulvovaginal candidiasis released by Mycovia Pharmaceuticals
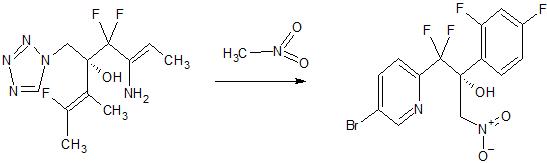
Synthesis Reference
Hoekstra, WJ., et al. (2020). Antifungal compound process (U.S. Patent No. US 10,745,378 B2). U.S. Patent and Trademark Office. https://patentimages.storage.googleapis.com/f4/62/19/5ba525b1caad0e/US10745378.pdf
PATENT
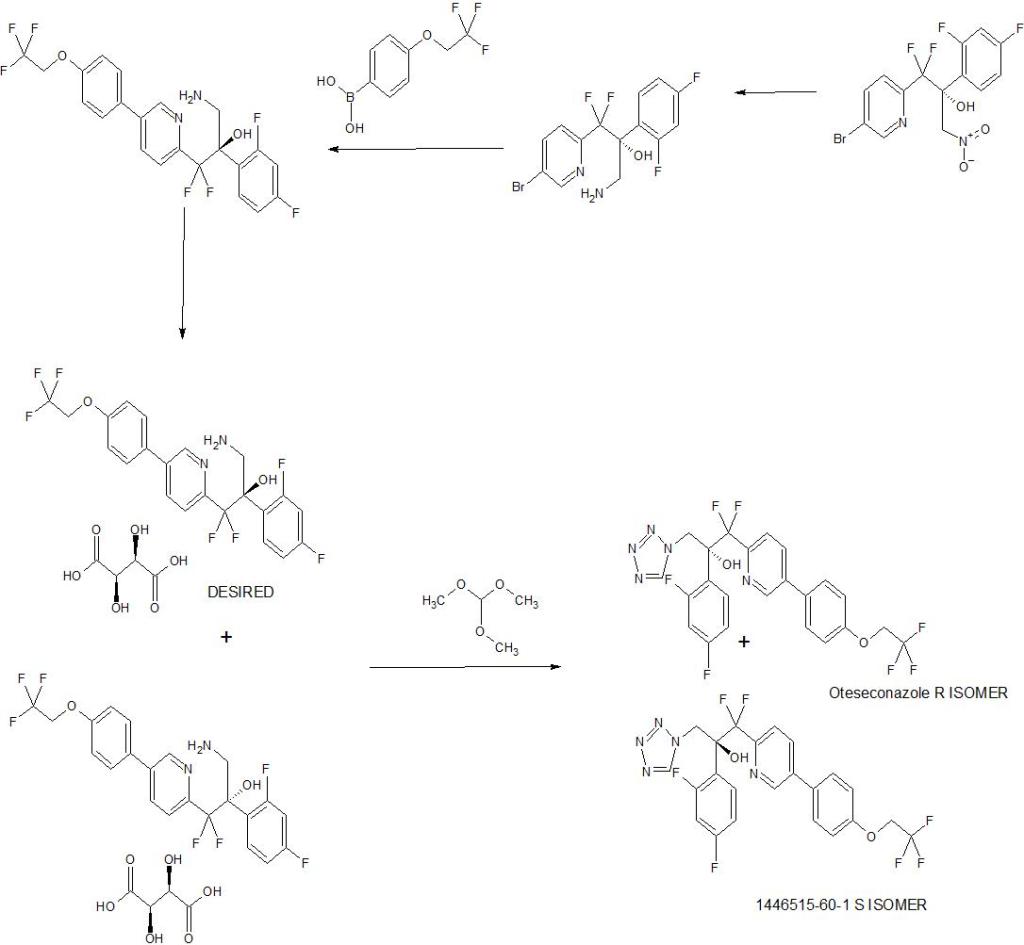
WO 2017049080
WO 2016149486
US 20150024938
WO 2015143172
WO 2015143184
WO 2015143180
WO 2015143142
WO 2013110002
WO 2013109998
WO 2011133875
PATENT
WO 2017049080,
Syn
J. Med. Chem. 2024, 67, 4376−4418
Oteseconazole was approved by the USFDA in April 2022 for the treatment of recurrent vulvovaginal candidiasis in women with a history of vulvovaginal candidiasis and who are not of reproductive
potential. Additional studies for other invasive and opportunistic infections and for onychomycosis are underway.40, The design and discovery of oteseconazole is published by a group from Viamet Pharmaceuticals, now part of Mycovia Pharmaceuticals. It details the racemic synthesis of the drug on
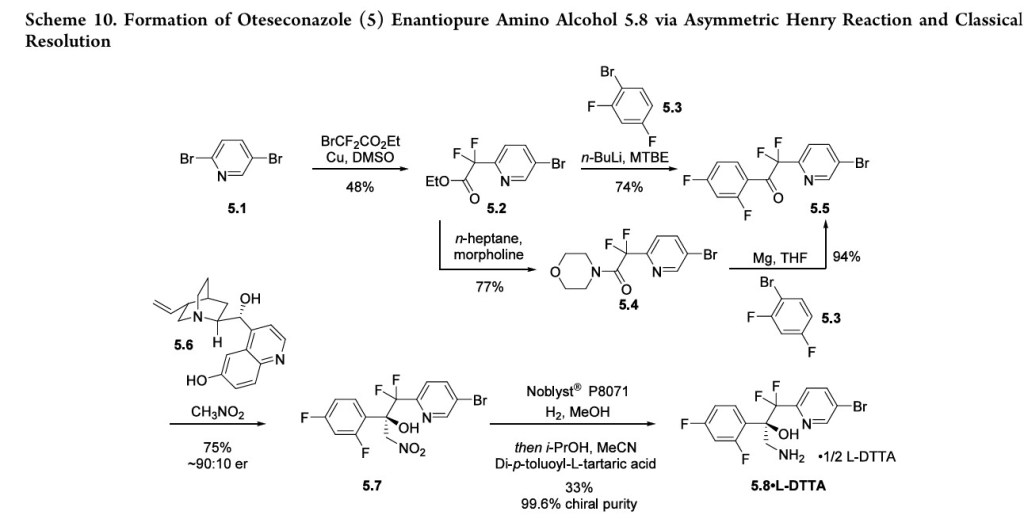
<1 g scale in which the metal-binding tetrazole is installed by treatment of ester 5.2 (Scheme 10) with diazomethane and tetrazole.42
A more scale-friendly asymmetric route that avoided the use of diazomethane was subsequently disclosed in patents and is detailed in Scheme 10 and Scheme 11.43
First, a mixture of ethyl bromodifluoroacetate, stoichiometric copper
powder, and 2,5-dibromopyridine (5.1) in DMSO provided ester 5.2 as an oil that was purified via distillation (Scheme10). Conversion to the aryl ketone 5.5 was achieved via direct addition of lithiated 5.3 or via a two-step process by first conversion to morpholine amide 5.4 followed by addition of
the Grignard generated from aryl bromide 5.3. The resulting ketone 5.5 was a liquid that was carried into the next step without purification.
The key step in the synthesis of 5 is an asymmetric Henry reaction using cinchona alkaloid catalyst 5.6. Addition of nitromethane to ketone 5.5 furnished alcohol 5.7 in 75% yield and ∼90:10 ratio of enantiomers. Next, reduction of the nitro group to the primary amine was accomplished using Pt
catalyzed hydrogenation. The chiral purity of the resulting amine was upgraded by classical resolution using di-p-toluoyl L-tartaric acid to provide 5.8·L-DTTA in 33% yield and >99% chiral purity.Conversion of amino alcohol 5.8 to oteseconazole (5) required two steps: cross coupling to introduce the aryltrifluoroethyl ether fragment and tetrazole formation. These steps were performed in either sequence in the patent. The route shown in Scheme 11 represents the largest scale demonstrated (>100 g input of 5.8). While the use of azide containing reagents presents significant safety risks, no information was provided on safe operation of the tetrazole forming step in the laboratory or on plant scale. Some of the
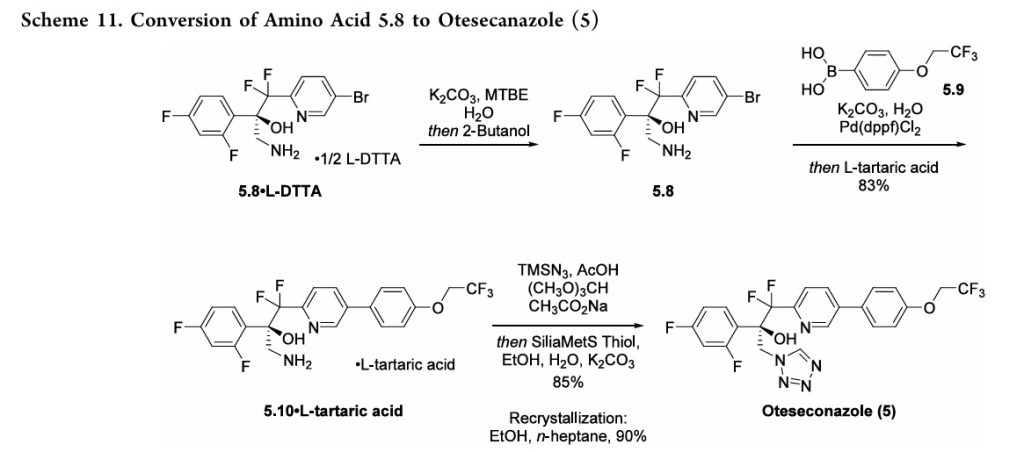
procedures for tetrazole formation described in the patent would likely require modification for safe scale-up.
To complete the synthesis of oteseconazole, resolved amino alcohol 5.8 first underwent a salt break followed by Suzuki coupling using boronic acid 5.9 to provide biaryl product 5.10 as the L-tartrate salt (Scheme 11). Conversion of 5.10 to 5 was accomplished using TMSN3 in acetic acid with sodium acetate and trimethoxy orthoformate. Treatment of the resulting solution with a Pd scavenger preceded crystallization of the product from EtOH and water after pH adjustment with potassium carbonate. The product was isolated in 85% yield as a hydrated form. Another patent described conversion of the oteseconazolehydrate totheanhydrous form byrecrystallizationfrom EtOHandn-heptanetofurnish5 in90%yield.45
(40) Hoy, S. M. Oteseconazole: First approval. Drugs 2022, 82,1017−1023.
(41) Sobel, J. D.; Nyirjesy, P. Oteseconazole: an advance in
treatment of recurrent vulvovaginal candidiasis. Future Microbiol 2021,
16, 1453−1461.
(42) Hoekstra, W. J.; Garvey, E. P.; Moore, W. R.; Rafferty, S. W.;
Yates, C. M.; Schotzinger, R. J. Design and optimization of highly
selective fungal CYP51 inhibitors. Bioorg. Med. Chem. Lett. 2014, 24,
3455−3458.
(43) Wirth, D. D.; Yates, C. M.; Hoekstra, W. J.; Bindl, M. F.;
Hartmann, E. Process for enantioselective preparation of tetrazolyl
pyridinyl diaryl propanols as antifungal drugs and their precursors.
WO 2017049080, 2017.
(44) González-Bobes, F.; Kopp, N.; Li, L.; Deerberg, J.; Sharma, P.;
Leung, S.; Davies, M.; Bush, J.; Hamm, J.; Hrytsak, M. Scale-up of
Azide Chemistry: A Case Study. Org. Process Res. Dev. 2012, 16,
2051−2057.
(45) Hoekstra, W. J.; Wirth, D. D.; Ehiwe, T.; Bonnaud, T.
Antifungal compounds and processes for making. WO 2016149486,
2016.
.
PATENT
WO-2021143811
Novel crystalline polymorphic form of VT-1161 (also known as oteseconazole) phosphate disodium salt, useful as a prodrug of oteseconazole, for treating systemic fungal infection (eg Candida albicans infection) or onychomycosis.The function of metalloenzymes is highly dependent on the presence of metal ions in the active site of the enzyme. It is recognized that reagents that bind to and inactivate metal ions at the active site greatly reduce the activity of the enzyme. Nature uses this same strategy to reduce the activity of certain metalloenzymes during periods when enzyme activity is not needed. For example, the protein TIMP (tissue inhibitor of metalloproteinases) binds to zinc ions in the active sites of various matrix metalloproteinases, thereby inhibiting enzyme activity. The pharmaceutical industry has used the same strategy in the design of therapeutic agents. For example, the azole antifungal agents fluconazole and voriconazole contain 1-(1,2,4-triazole) group, which exists in the active site of the target enzyme lanosterol demethylase The heme iron binds, thereby inactivating the enzyme. Another example includes zinc-bound hydroxamic acid groups, which have been introduced into most of the published inhibitors of matrix metalloproteinases and histone deacetylases. Another example is the zinc-binding carboxylic acid group, which has been introduced into most of the published angiotensin converting enzyme inhibitors.
VT-1161, the compound 2-(2,4-difluorophenyl)-1,1-difluoro-3-(1H-tetrazol-1-yl)-1-(5-(4-(2, 2,2-Trifluoroethoxy)phenyl)pyridin-2-yl)propan-2-ol, is an antifungal drug developed by VIAMET, currently in the clinical research stage, its structure is as follows Shown:
This compound mainly acts on the CYP51 target of fungal cells. Compared with the previous triazole antifungal drugs, it has the advantages of wider antibacterial spectrum, low toxicity, high safety and good selectivity. However, this compound is not suitable for Liquid preparations (including or excluding the parenteral delivery carrier) are used to treat patients in need thereof.
2-(2,4-Difluorophenyl)-1,1-difluoro-3-(1H-tetrazol-1-yl)-1-(5-(4-(2,2,2-trifluoro Ethoxy)phenyl)pyridin-2-yl)propan-2-yl dihydrogen phosphate is a prodrug of VT-1161.
On the other hand, nearly half of the drug molecules are in the form of salts, and salt formation can improve certain undesirable physicochemical or biological properties of the drug. Relative to 2-(2,4-difluorophenyl)-1,1-difluoro-3-(1H-tetrazol-1-yl)-1-(5-(4-(2,2,2- Trifluoroethoxy)phenyl)pyridin-2-yl)propan-2-yl dihydrogen phosphate, it is of great significance to develop salts with more excellent properties in terms of physical and chemical properties or pharmaceutical properties.To this end, the present disclosure provides a new pharmaceutically acceptable salt form of a metalloenzyme inhibitor.Example 1:[0161](R)-2-(2,4-Difluorophenyl)-1,1-difluoro-3-(1H-tetrazol-1-yl)-1-(5-(4-(2,2, 2-Trifluoroethoxy)phenyl)pyridin-2-yl)propan-2-yl phosphate disodium salt (Compound 1)[0162]
[0163](R)-2-(2,4-Difluorophenyl)-1,1-difluoro-3-(1H-tetrazol-1-yl)-1-(5-(4-(2,2 ,2-Trifluoroethoxy)phenyl)pyridin-2-yl)propan-2-yl phosphate (compound 1a, prepared according to the method of patent WO2013110002, 0.28g, 0.46mmol, 1.0eq) and ethanol (5mL ) Add to the reaction flask and stir evenly. A solution of NaOH (36.90 mg, 2.0 eq) dissolved in water (1 mL) was added dropwise into the above reaction flask, stirring was continued for 2 h, and concentrated to obtain compound 1, 300 mg of white solid.[0164]After X-ray powder diffraction detection, the XRPD spectrum has no sharp diffraction peaks, as shown in FIG. 10.[0165]Ms:608.10[M-2Na+3H] + .[0166]Ion chromatography detected that the sodium ion content was 6.23%.[0167]Example 2: (R)-((2-(2,4-Difluorophenyl)-1,1-difluoro-3-(1H-tetrazol-1-yl)-1-(5-(4 -(2,2,2-Trifluoroethoxy)phenyl)pyridin-2-yl)prop-2-yl)oxy)methyl phosphate disodium salt (compound 2)
[0169]Under ice-cooling, NaH (58mg, 0.87mmol) was added to the reaction flask, 1.5mL of N,N-dimethylformamide and 0.6mL of tetrahydrofuran were added, followed by iodine (38mg, 0.15mmol), and then Compound 2-(2,4-difluorophenyl)-1,1-difluoro-3-(1H-tetrazol-1-yl)-1-(5-(4-(2,2,2-tri Fluoroethoxy)phenyl)pyridin-2-yl)propan-2-ol (2b, prepared according to the method of patent WO2013110002, 158mg, 0.3mmol) tetrahydrofuran (1ml) solution was added to the reaction solution, stirred and reacted for 1-4h , And then add compound 2a (519mg, 2.01mmol) in tetrahydrofuran (1ml) solvent to the reaction, stir until the reaction is complete, 10% aqueous ammonium chloride solution to quench the reaction, extract, concentrate and drain, the crude product 2c is directly used for the next One-step reaction, Ms: 750.0[M+H] + .[0170]
[0171]Under ice-bath cooling, add trifluoroacetic acid (0.5mL) to the crude product 2c (300mg) in dichloromethane (2mL) solution, stir until the reaction is complete, and after concentration, the target compound 2d, 82mg, Ms was separated by high performance liquid phase separation. :638.0[M+H] + .[0172]
Add compound 2d (0.29g, 0.46mmol, 1.0eq) and ethanol (5mL) obtained in the previous step into the reaction flask, stir, and add NaOH (36.90mg, 2.0eq) water (1ml) solution dropwise to the aforementioned reaction solution , Stirred for 2-5 h, and concentrated to obtain 2,313 mg of the target compound.
Ms:638.10[M-2Na+3H] + .
PATENT
WO2011133875
https://patents.google.com/patent/WO2011133875A2/en
Product pat, WO2011133875 , protection in the EU states and the US April 2031.
PATENT
WO2015143184 ,
https://patents.google.com/patent/WO2015143184A1/en
Mycovia, claiming a process for preparing antifungal compounds, particularly oteseconazole.EXAMPLE 11

2-(2,4-Difluorophenyl)-l,l-difluoro-3-(lH-tetrazol-l-yl)-l-(5-(4-(2,2,2- trifluoroethoxy)phenyl)pyridin-2-yl)propan-2-ol (11)Compound 11 was prepared using the conditions employed for 1: 0.33 g as a solid. The precursor l-bromo-4-(2,2,2-trifluoroethoxy)benzene was prepared as described below in one step.1H NMR (500 MHz, CDC13): δ 8.76 (s, 1 H), 8.70 (s, 1 H), 7.95 (d, / = 8.0 Hz, 1 H), 7.70 (s, 1 H), 7.64 (d, / = 8.5 Hz, 1 H), 7.54 (d, / = 8.5 Hz, 2 H), 7.42- 7.37 (m, 1 H), 7.08 (d, / = 8.5 Hz, 2 H), 6.79- 6.75 (m, 1 H), 6.69- 6.66 (m, 1 H), 5.58 (d, / = 14.0 Hz, 1 H), 5.14 (d, / = 14.0 Hz, 1 H), 4.44 – 4.39 (m, 2 H). HPLC: 99.1%. MS (ESI): m/z 528 [M++l].Chiral preparative HPLC Specifications for (+)-ll:Column: Chiralpak IA, 250 x 4.6mm, 5uMobile Phase: A) w-Hexane, B) IPAIsocratic: A: B (65:35)Flow Rte: l.OO mL/minOptical rotation [a]D: + 24° (C = 0.1 % in MeOH). 1 -Bromo-4-( 2,2,2-trifluoroethoxy )benzeneTo a stirred solution of trifluoroethyl tosylate (1.5 g, 5.8 mmol) in DMF (20 mL) was added K2CO3 (4 g, 29.4 mmol) followed by addition of p-bromo phenol (1.1 g, 6.46 mmol) at RT under inert atmosphere. The reaction mixture was stirred at 120 °C for 6 h. The volatiles were evaporated under reduced pressure; the residue was diluted with water (5 mL) and extracted with ethyl acetate (3 x 30 mL). The organic layer was washed with water, brine and dried over anhydrous Na2S04, filtered and concentrated in vacuo. The crude compound was purified by silica gel column chromatography eluting with 5% EtOAc/hexane to afford the desired product (0.8 g, 3.13 mmol, 53.3%) as semi solid. 1H NMR (200 MHz, CDC13): δ 7.44 – 7.38 (m, 2 H), 6.86-6.80 (m, 2 H), 4.38- 4.25 (m, 2 H).ExamplesThe present invention will now be demonstrated using specific examples that are not to be construed as limiting.General Experimental ProceduresDefinitions of variables in the structures in schemes herein are commensurate with those of corresponding positions in the formulae delineated herein.Synthesis of 1 or la

A process to prepare enantiopure compound 1 or la is disclosed. Syntheses of lor la may be accomplished using the example syntheses that are shown below (Schemes 1-4). The preparation of precursor ketone 3-Br is performed starting with reaction of 2,5-dibromo- pyridine with ethyl 2-bromo-difluoroacetate to produce ester 2-Br. This ester can be reacted with morpholine to furnish morpholine amide 2b-Br, followed by arylation to provide ketone 3-Br. Alternatively, ketone 3-Br can be afforded directly from ester 2-Br as shown in Scheme 1. Scheme 1. Synthesis of ketone 3-Br r

Ketone 3 may be prepared in an analogous fashion as described in Scheme 1 starting from corresponding substituted 2-bromo-pyridines, which can be prepared according to synthetic transformations known in the art and contained in the references cited herein (Scheme 2).Scheme 2. Synthesis of ketone 3

R-i = halo, -0(C=0)-alkyl, -0(C=0)-substituted alkyl, -0(C=0)-aryl, -0(C=0)-substituted aryl, -0(C=0)-0-alkyl, – 0(C=0)-0-substituted alkyl, -0(C=0)-0-aryl, -0(C=0)-0-substituted aryl, -0(S02)-alkyl, -0(S02)-substituted alkyl, – 0(S02)-aryl, or -0(S02)-substituted aryl.Alternatively, compound 1 can be prepared according to Scheme 3 utilizing diols 2-6b (or 2- 6d, the enantiomer of 2-6b, or mixtures thereof) or 2-6a (or 2-6c, the enantiomer of 2-6a, or mixtures thereof). Olefins 2-5a and 2-5 can be prepared by reacting ketones 3 and 1-4 under Wittig olefination conditions (e.g., Ph3PCH3Br and BuLi). Also, as indicated in Scheme 5, any of pyridine compounds, 3, 2-5a, 2-6b, 2-7b, 4*, 4b, or 6 can be converted to the corresponding 4-CF3CH2O-PI1 analogs (e.g., 1-4, 2-5, 2-6a, 2-7a, 5*, 1-6*, or 1 or the corresponding enantiomers, or mixtures thereof) by cross-coupling with 4,4,5, 5-tetramethyl-2- (4-(2,2,2-trifluoroethoxy)phenyl)-l,3,2-dioxaborolane (or the corresponding alkyl boronates or boronic acid or the like), in a suitable solvent system (e.g., an organic-aqueous solvent mixture), in the presence of a transition metal catalyst (e.g., (dppf)PdCl2), and in the presence of a base (e.g., KHCO3, K2C03, Cs2C03, or Na2C03, or the like). Olefins 2-5a and 2-5 can be transformed to the corresponding chiral diols, 2-6b (or 2-6d, the enantiomer of 2-6b, or mixtures thereof) or 2-6a (or 2-6c, the enantiomer of 2-6a, or mixtures thereof), through exposure to Sharpless asymmetric dihydroxylation conditions: 1) commercially available AD- mix alpha or AD-mix beta with or without additional osmium oxidant and methanesulfonamide, 2) combination of a catalytic osmium oxidant (e.g., Os04 or K20sC>2(OH)4), a stoichiometric iron oxidant (e.g., K3Fe(CN)6), a base (e.g., KHCO3, K2CO3, Cs2C03, or Na2C03, or the like), and a chiral ligand (e.g., (DHQ)2PHAL, (DHQD)2PHAL, (DHQD)2AQN, (DHQ)2AQN, (DHQD)2PYR, or (DHQ)2PYR; preferably (DHQ)2PHAL, (DHQD)2PHAL, (DHQD)2AQN, and (DHQD)2PYR), or 3) option 2) with methanesulfonamide. The primary alcohol of the resultant chiral diols, 2-6b (or 2-6d, the enantiomer of 2-6b, or mixtures thereof) or 2-6a (or 2-6c, the enantiomer of 2-6a, or mixtures thereof), can then be activated to afford compounds 2-7b (or 2-7d, the enantiomer of 2-7b, or mixtures thereof) or 2-7a (or 2-7c, the enantiomer of 2-7a, or mixtures thereof). For example, the mesylates can be prepared by exposing chiral diols, 2-6b (or 2-6d, the enantiomer of 2-6b, or mixtures thereof) or 2-6a (or 2-6c, the enantiomer of 2-6a, or mixtures thereof), to methanesulfonyl chloride and a base. Epoxide formation can be affected by the base-mediated (e.g., KHCO3, K2CO3, CS2CO3, or Na2CC>3, or the like) ring closure of compounds 2-7b (or 2- 7d, the enantiomer of 2-7b, or mixtures thereof) or 2-7a (or 2-7c, the enantiomer of 2-7a, or mixtures thereof) to provide epoxides 4* (or 4c*, the enantiomer of 4*, or mixtures thereof) and 5* (or 5-b*, the enantiomer of 5*, or mixtures thereof). The epoxides can then be converted into amino-alcohols 4b (or 4c, the enantiomer of 4b, or mixtures thereof) and 1-6* (or 1-7*, the enantiomer of 1-6*, or mixtures thereof) through ammonia-mediated epoxide opening using ammonia in a suitable solvent (e.g., MeOH, EtOH, or water). Subsequent treatment with TMS-azide in the presence of trimethylorthoformate and sodium acetate in acetic acid would yield compounds 6 (or 6a, the enantiomer of 6, or mixtures thereof) or 1 (or la, the enantiomer of 1, or mixtures thereof) (US 4,426,531).Scheme 3. Synthesis of 1 via Asymmetric Dihydroxylation Method


Y is -OS02-alkyl, -OS02-substituted alkyl, -OS02-aryl, -OS02- substituted aryl, -0(C=0)-alkyl, -0(C=0)-substituted alkyl, – 0(C=0)-aryl, -0(C=0)-substituted aryl, or halogen

R-i = halo, -0(C=0)-alkyl, -0(C=0)-substituted alkyl, -0(C=0)-aryl, -0(C=0)-substituted aryl, -0(C=0)-0-alkyl, -0(C=0)-0-substituted alkyl, -0(C=0)-0-aryl, -0(C=0)-0-substituted aryl, -0(S02)-alkyl, -0(S02)-substituted alkyl, -0(S02)-aryl, or -0(S02)-substituted aryl.Compound 1 (or la, the enantiomer of 1, or mixtures thereof) prepared by any of the methods presented herein can be converted to a sulfonic salt of formula IX (or IXa, the enantiomer of IX, or mixtures thereof), as shown in Scheme 4. This can be accomplished by a) combining compound 1 (or la, the enantiomer of 1, or mixtures thereof), a crystallization solvent or crystallization solvent mixture (e.g., EtOAc, i‘PrOAc, EtOH, MeOH, or acetonitrile, or oZ-S-OHcombinations thereof), and a sulfonic acid o (e.g., Z = Ph, p-tolyl, Me, or Et), b) diluting the mixture with an appropriate crystallization co-solvent or crystallization co-solvent mixture (e.g., pentane, methyl i-butylether, hexane, heptane, or toluene, or combinations thereof), and c) filtering the mixture to obtain a sulfonic acid salt of formula IX (or IXa, the enantiomer of IX, or mixtures thereof). cheme 4. Synthesis of a Sulfonic Acid Salt of Compound 1 or la

The following describes the HPLC method used in assessing HPLC purity of the examples and intermediates presented below:Column: Waters XBridge Shield RP18, 4.6 x 150 mm, 3.5 μιηMobile Phase: A = 0.05% TFA/H20, B = 0.05% TFA/ACNAutosampler flush: 1 : 1 ACN/H20Diluent: 1:1 ACN/H20Flow Rate: 1.0 ml/minTemperature: 45 °CDetector: UV 275 nmPump Parameters:

EXAMPLE 1Preparation of ethyl 2-(5-bromopyridin-2-yl)-2,2-difluoroacetate (2-Br)

2-Br Dialkylated impurity In a clean multi-neck round bottom flask, copper powder (274.7 g, 2.05 eq) was suspended in dimethyl sulfoxide (3.5 L, 7 vol) at 20 – 35 °C. Ethyl bromodifluoroacetate (449 g, 1.05 eq) was slowly added to the reaction mixture at 20 – 25 °C and stirred for 1 – 2 h. 2, 5- dibromopyridine (500 g, 1 eq) was added to the reaction mixture and the temperature was increased to 35 – 40 °C. The reaction mixture was maintained at this temperature for 18 – 24 h and the reaction progress was monitored by GC.After the completion of the reaction, ethyl acetate (7 L, 14 vol) was added to the reaction mixture and stirring was continued for 60 – 90 min at 20 – 35 °C. The reaction mixture was filtered through a Celite bed (100 g; 0.2 times w/w Celite and 1L; 2 vol ethyl acetate). The reactor was washed with ethyl acetate (6 L, 12 vol) and the washings were filtered through a Celite bed. The Celite bed was finally washed with ethyl acetate (1 L, 2 vol) and all the filtered mother liquors were combined. The pooled ethyl acetate solution was cooled to 8 – 10 °C, washed with the buffer solution (5 L, 10 vol) below 15 °C (Note: The addition of buffer solution was exothermic in nature. Controlled addition of buffer was required to maintain the reaction mixture temperature below 15 °C). The ethyl acetate layer was washed again with the buffer solution until (7.5 L; 3 x 5 vol) the aqueous layer remained colorless. The organic layer was washed with a 1: 1 solution of 10 % w/w aqueous sodium chloride and the buffer solution (2.5 L; 5 vol). The organic layer was then transferred into a dry reactor and the ethyl acetate was distilled under reduced pressure to get crude 2-Br.The crude 2-Br was purified by high vacuum fractional distillation and the distilled fractions having 2-Br purity greater than 93 % (with the dialkylated not more than 2 % and starting material less than 0.5 %) were pooled together to afford 2-Br.Yield after distillation: 47.7 % with > 93 % purity by GC (pale yellow liquid). Another 10 % yield was obtained by re-distillation of impure fractions resulting in overall yield of ~ 55 – 60 %.*H NMR: δ values with respect to TMS (DMSO-d6; 400 MHz): 8.85 (1H, d, 1.6 Hz), 8.34 (1H, dd, J = 2.0 Hz, 6.8 Hz), 7.83 (1H, d, J = 6.8 Hz), 4.33 (2H, q, J = 6.0 Hz), 1.22 (3H, t, J = 6.0 Hz). 13C NMR: 162.22 (i, -C=0), 150.40 (Ar-C-), 149.35 (t, Ar-C), 140.52 (Ar-C), 123.01 (Ar-C), 122.07 (Ar-C), 111.80 (t, -CF2), 63.23 (-OCH2-), 13.45 (-CH2CH3).EXAMPLE 2
Preparation of2-( 5-bromopyridin-2-yl )-l -(2,4-difluorophenyl )-2, 2-difluoroethanone ( 3-Br ) A. One-step Method

l-Bromo-2,4-difluorobenzene (268.7 g; 1.3 eq) was dissolved in methyl tert butyl ether (MTBE, 3.78 L, 12.6 vol) at 20 – 35 °C and the reaction mixture was cooled to -70 to -65 °C using acetone/dry ice bath. n-Butyl lithium (689 rriL, 1.3 eq; 2.5 M) was then added to the reaction mixture maintaining the reaction temperature below -65 °C (Note: Controlled addition of the n-Butyl Lithium to the reaction mixture was needed to maintain the reaction mixture temperature below – 65 °C). After maintaining the reaction mixture at this temperature for 30 – 45 min, 2-Br (300 g, 1 eq) dissolved in MTBE (900 rriL, 3 vol) was added to the reaction mixture below – 65 °C. The reaction mixture was continued to stir at this temperature for 60 – 90 min and the reaction progress was monitored by GC.The reaction was quenched by slow addition of 20 % w/w ammonium chloride solution (750 mL, 2.5 vol) below -65 °C. The reaction mixture was gradually warmed to 20 – 35 °C and an additional amount of 20 % w/w ammonium chloride solution (750 mL, 2.5 vol) was added. The aqueous layer was separated, the organic layer was washed with a 10 % w/w sodium bicarbonate solution (600 mL, 2 vol) followed by a 5 % sodium chloride wash (600 mL, 2 vol). The organic layer was dried over sodium sulfate (60 g; 0.2 times w/w), filtered and the sodium sulfate was washed with MTBE (300 mL, 1 vol). The organic layer along with washings was distilled below 45 °C under reduced pressure until no more solvent was collected in the receiver. The distillation temperature was increased to 55 – 60 °C, maintained under vacuum for 3 – 4 h and cooled to 20 – 35 °C to afford 275 g (73.6 % yield, 72.71 % purity by HPLC) of 3-Br as a pale yellow liquid.*H NMR: δ values with respect to TMS (DMSO-d6; 400 MHz):8.63 (1H, d, 1.6 Hz, Ar-H), 8.07 – 8.01 (2H, m, 2 x Ar-H), 7.72 (1H, d, J = 6.8 Hz, Ar-H), 7.07 – 6.82 (1H, m, Ar-H), 6.81 – 6.80 (1H, m, Ar-H). 13C NMR: 185.60 (t, -C=0), 166.42 (dd, Ar-C-), 162.24 (dd, Ar-C),150.80 (Ar-C), 150.35 (Ar-C), 140.02 (Ar-C), 133.82 (Ar-C), 123.06 (Ar-C), 1122.33 (Ar-C), 118.44 (Ar-C), 114.07 (-CF2-), 122.07 (Ar-C), 105.09 (Ar-C).
B. Two-step Method via 2b-Br

2-Br (147.0 g) was dissolved in n-heptane (1.21 L) and transferred to a 5-L reactor equipped with overhead stirrer, thermocouple, condenser and addition funnel. Morpholine (202 ml) was added. The solution was heated to 60 °C and stirred overnight. The reaction was complete by HPLC analysis (0.2% 2-Br; 94.7% 2b-Br). The reaction was cooled to room temperature and 1.21 L of MTBE was added. The solution was cooled to ~4 °C and quenched by slow addition of 30% citric acid (563 ml) to maintain the internal temperature <15 °C. After stirring for one hour the layers were allowed to settle and were separated (Aq. pH=5). The organic layer was washed with 30% citric acid (322 ml) and 9% NaHC03 (322 ml, aq. pH 7+ after separation). The organic layer was concentrated on the rotary evaporator (Note 1) to 454 g (some precipitation started immediately and increased during concentration). After stirring at room temperature the suspension was filtered and the product cake was washed with n-heptane (200 ml). The solid was dried in a vacuum oven at room temperature to provide 129.2 g (77%) dense powder. The purity was 96.5% by HPLC analysis.To a 1-L flask equipped with overhead stirring, thermocouple, condenser and addition funnel was added magnesium turnings (14.65 g), THF (580 ml) and l-bromo-2,4-difluorobenzene (30.2 g, 0.39 equiv). The mixture was stirred until the reaction initiated and self-heating brought the reaction temperature to 44 °C. The temperature was controlled with a cooling bath as the remaining l-bromo-2,4-difluorobenzene (86.1 g, 1.11 equiv) was added over about 30 min. at an internal temperature of 35-40 °C. The reaction was stirred for 2 hours while gradually cooling to room temperature. The dark yellow solution was further cooled to 12 °C.During the Grignard formation, a jacketed 2-L flask equipped with overhead stirring, thermocouple, and addition funnel was charged with morpholine amide 2b-Br (129.0 g) and THF (645 ml). The mixture was stirred at room temperature until the solid dissolved, and then the solution was cooled to -8.7 °C. The Grignard solution was added via addition funnel over about 30 min. at a temperature of -5 to 0 °C. The reaction was stirred at 0 °C for 1 hour and endpointed by HPLC analysis. The reaction mixture was cooled to -5 °C and quenched by slow addition of 2N HC1 over 1 hour at <10 °C. The mixture was stirred for 0.5 h then the layers were allowed to settle and were separated. The aqueous layer was extracted with MTBE (280 ml). The combined organic layers were washed with 9% NaHCC>3 (263 g) and 20% NaCl (258 ml). The organic layer was concentrated on the rotary evaporator with THF rinses to transfer all the solution to the distillation flask. Additional THF (100 ml) and toluene (3 x 100 ml) were added and distilled to remove residual water from the product. After drying under vacuum, the residue was 159.8 g of a dark brown waxy solid (>theory). The purity was approximately 93% by HPLC analysis.EXAMPLE 3Preparation of 3-amino-l-(5-bromopyridin-2-yl)-2-(2,4-difluorophenyl)-l,l-difluoropropan- -ol (±ib-Br)

4-Br (200g, 1 eq) was added into methanolic ammonia (8.0 L; 40 vol; ammonia content: 15 – 20 % w/v) in an autoclave at 10 – 20 °C. The reaction mixture was gradually heated to 60 – 65 °C and at 3 – 4 kg/cm2 under sealed conditions for 10 – 12 h. The reaction progress was monitored by GC. After completion of the reaction, the reaction mixture was cooled to 20 – 30 °C and released the pressure gradually. The solvent was distilled under reduced pressure below 50 °C and the crude obtained was azeotroped with methanol (2 x 600 mL, 6 vol) followed by with isopropanol (600 mL, 2 vol) to afford 203 g (96.98 % yield, purity by HPLC: 94.04 %) of +4b-Br. EXAMPLE 4Preparation of3-amino-l-(5-bromopyridin-2-yl)-2-(2,4-difluorophenyl)-l,l-difluoropropan- -ol (4b-Br or 2c-Br)

Amino alcohol ±4b-Br (150 g, 1 eq) was dissolved in an isopropanol /acetonitrile mixture (1.5L, 8:2 ratio, 10 vol) and Di-p-toluoyl-L-tartaric acid (L-DPTTA) (84.05 g, 0.55 eq) was added into the reactor at 20 – 30 °C. The reaction mixture was heated to 45 – 50 °C for 1 – 1.5 h (Note: The reaction mixture becomes clear and then became heterogeneous). The reaction mixture was gradually cooled to 20 – 30 °C and stirred for 16 – 18 h. The progress of the resolution was monitored by chiral HPLC analysis.After the completion of the resolution, the reaction mixture was gradually cooled to 20 – 35 °C. The reaction mixture was filtered and the filtered solid was washed with a mixture of acetonitrile and isopropanol (8:2 mixture, 300 mL, 2 vol) and dried to afford 75 g of the L- DPTTA salt (95.37 % ee). The L-DPTTA salt obtained was chirally enriched by suspending the salt in isopropanol /acetonitrile (8:2 mixture; 750 mL, 5 vol) at 45 – 50 °C for 24 – 48 h. The chiral enhancement was monitored by chiral HPLC; the solution was gradually cooled to 20 – 25 °C, filtered and washed with an isoporpanol /acetonitrile mixture (8:2 mixture; 1 vol). The purification process was repeated and after filtration, the salt resulted in chiral purity greater than 96 % ee. The filtered compound was dried under reduced pressure at 35 – 40 °C to afford 62 g of the enantio-enriched L-DPPTA salt with 97.12% ee as an off-white solid. The enantio-enriched L-DPTTA salt (50 g, 1 eq) was dissolved in methanol (150 mL, 3 vol) at 20 – 30 °C and a potassium carbonate solution (18.05 g K2CO3 in 150 mL water) was slowly added at 20 – 30 °C under stirring. The reaction mixture was maintained at this temperature for 2 – 3 h (pH of the solution at was maintained at 9). Water (600 mL, 12 vol) was added into the reaction mixture through an additional funnel and the reaction mixture was stirred for 2 – 3 h at 20 – 30 °C. The solids were filtered; washed with water (150 mL, 3 vol) and dried under vacuum at 40 – 45 °C to afford 26.5 g of amino alcohol 4b-Br or 4c-Br with 99.54 % chemical purity, 99.28 % ee as an off-white solid. (Water content of the chiral amino alcohol is below 0.10 % w/w).1H NMR: δ values with respect to TMS (DMSO-d6; 400 MHz):8.68 (1H, d, J = 2.0 Hz, Ar- H), 8.16 (1H, dd, J = 8.0 Hz, 2.0 Hz, Ar-H), 7.49 – 7.43 (1H, m, Ar-H), 7.40 (1H, d, J = 8 Hz, Ar-H), 7.16 – 7.11 (1H, m, Ar-H), 7.11 – 6.99 (1H, m, Ar-H), 3.39 – 3.36 (1H, m, -OCHAHB– ), 3.25 – 3.22 (1H, m, -OCHAHB-).13C NMR: 163.87 -158.52 (dd, 2 x Ar-C-), 150.88 (Ar-C), 149.16 (Ar-C), 139.21 (Ar-C), 132.39 (Ar-C), 124.49 (Ar-C), 122.17 (Ar-C), 121.87 (d, Ar- C), 119.91 (t, -CF2-), 110.68 (Ar-C), 103.97 (i, Ar-C), 77.41 (i,-C-OH), 44.17 (-CH2-NH2).EXAMPLE 5
Preparation of l-(5-bromopyridin-2-yl)-2-(2,4-difluorophenyl)-l,l-difluoro-3-(lH-tetrazol-l- yl)propan-2-ol (l-6*-Br or l-7*-Br)

4b-Br or 4c-Br (20.0 g, 1 eq.) was added to acetic acid (50 mL, 2.5 vol) at 25 – 35 °C followed by the addition of anhydrous sodium acetate (4.32 g, 1 eq), trimethyl orthoformate (15.08 g, 2.7 eq). The reaction mixture was stirred for 15 – 20 min at this temperature and trimethylsilyl azide (12.74 g, 2.1 eq) was added to the reaction mixture (Chilled water was circulated through the condenser to minimize the loss of trimethylsilyl azide from the reaction mixture by evaporation). The reaction mixture was then heated to 70 – 75 °C and maintained at this temperature for 2 -3 h. The reaction progress was monitored by HPLC. Once the reaction was complete, the reaction mixture was cooled to 25 – 35 °C and water (200 mL, 10 vol) was added. The reaction mixture was extracted with ethyl acetate (400 mL, 20 vol) and the aqueous layer was back extracted with ethyl acetate (100 mL, 5 vol). The combined organic layers were washed with 10 % potassium carbonate solution (3 x 200 mL; 3 x 10 vol) followed by a 10 % NaCl wash (1 x 200 mL, 10 vol). The organic layer was distilled under reduced pressure below 45 °C. The crude obtained was azeotroped with heptanes (3 x 200 mL) to get 21.5g (94 % yield, 99.26 5 purity) of tetrazole 1-6* or 1-7* compound as pale brown solid (low melting solid).1H NMR: δ values with respect to TMS (DMSO-d6; 400 MHz NMR instrument): 9.13 (1H, Ar-H), 8.74 (1H, Ar-H), 8.22 – 8.20 (1H, m, Ar-H), 7.44 (1H, d, J = 7.2 Hz, Ar-H), 7.29 (1H„Ar-H), 7.23 – 7.17 (1H, m, Ar-H), 6.92 – 6.88 (1H, Ar-H), 5.61 (1H, d, J = 1 1.2 Hz, – OCHAHB-), 5.08 (1H, d, J = 5.6 Hz, -OCHAHB-).13C NMR: 163.67 -161.59 (dd, Ar-C-), 160.60 – 158.50 (dd, Ar-C-), 149.65 (Ar-C), 144.99 (Ar-C), 139.75 (Ar-C), 131.65 (Ar-C), 124.26 (Ar-C), 122.32 (d, Ar-C), 119.16 (t, -CF2-), 118.70 (d, Ar-C), 1 11.05 (d, Ar-C) 104.29 (t, Ar-C), 76.79 (i,-C-OH), 59.72 (Ar-C), 50.23 (-OCH2N-). EXAMPLE 6Preparation of 2-(2,4-difluorophenyl)-l , 1 -difluoro-3-( 1 H-tetrazol-1 -yl)-l -(5-(4-(2,2,2- trifluoroethoxy)phenyl)pyridin-2-yl)propan-2-ol (1 or la)A. Preparation of 1 or la via l-6*-Br or l-7*-Br

Synthesis of 4,4,5, 5-tetramethyl-2-(4-(2,2,2-trifluoroethoxy)phenyl)-l,3,2-dioxaborolane Potassium carbonate (59.7 g, 2.2 eq.) was added to a slurry of DMF (190 mL, 3.8 Vol.), 4- Bromo phenol (37.4g, 1.1 eq.) and 2,2,2-trifluroethyl tosylate (50.0 g, 1.0 eq.) at 20 – 35 °C under an inert atmosphere. The reaction mixture was heated to 115 – 120 °C and maintained at this temperature for 15 – 18 h. The reaction progress was monitored by GC. The reaction mixture was then cooled to 20 – 35 °C, toluene (200 mL, 4.0 vol.) and water (365 mL, 7. 3 vol.) were added at the same temperature, stirred for 10 – 15 minutes and separated the layers. The aqueous layer was extracted with toluene (200 mL, 4.0 vol.). The organic layers were combined and washed with a 2M sodium hydroxide solution (175 mL, 3.5 vol.) followed by a 20 % sodium chloride solution (175 mL, 3.5 vol.). The organic layer was then dried over anhydrous sodium sulfate and filtered. The toluene layer was transferred into clean reactor, spurged with argon gas for not less than 1 h. Bis(Pinacolato) diborane (47 g, 1.1 eq.), potassium acetate (49.6 g, 3.0 eq.) and 1,4-dioxane (430 mL, 10 vol.) were added at 20 -35 °C, and spurged the reaction mixture with argon gas for at least 1 h. Pd(dppf)Cl2 (6.88 g, 0.05eq) was added to the reaction mixture and continued the argon spurging for 10 – 15 minutes. The reaction mixture temperature was increased to 70 – 75 °C, maintained the temperature under argon atmosphere for 15 – 35 h and monitored the reaction progress by GC. The reaction mixture was cooled to 20 – 35 °C, filtered the reaction mixture through a Celite pad, and washed with ethyl acetate (86 mL, 2 vol.). The filtrate was washed with water (430 mL, 10 vol.). The aqueous layer was extracted with ethyl acetate (258 mL, 6 vol.) and washed the combined organic layers with a 10 % sodium chloride solution (215 mL, 5 vol.). The organic layer was dried over anhydrous sodium sulfate (43g, 1 time w/w), filtered and concentrated under reduced pressure below 45 °C to afford crude 4,4,5, 5-tetramethyl-2-(4-(2,2,2- trifluoroethoxy)phenyl)-l,3,2-dioxaborolane (65 g; 71 % yield with the purity of 85.18 % by GC). The crude 4,4,5,5-tetramethyl-2-(4-(2,2,2-trifluoroethoxy)phenyl)-l,3,2-dioxaborolane (65 g, 1 eq.) was dissolved in 10 % ethyl acetate – n-Heptane (455 mL, 7 vol.) and stirred for 30 – 50 minutes at 20 – 35 °C. The solution was filtered through a Celite bed and washed with 10 % ethyl acetate in n-Heptane (195 mL, 3 vol.). The filtrate and washings were pooled together, concentrated under vacuum below 45 °C to afford 4,4,5, 5-tetramethyl-2-(4-(2,2,2- trifluoroethoxy)phenyl)-l,3,2-dioxaborolane as a thick syrup (45.5 g; 70 % recovery). This was then dissolved in 3 % ethyl acetate-n-heptane (4 vol.) and adsorbed on 100 – 200 M silica gel (2 times), eluted through silica (4 times) using 3 % ethyl acetate – n- heptane. The product rich fractions were pooled together and concentrated under vacuum. The column purified fractions (> 85 % pure) were transferred into a round bottom flask equipped with a distillation set-up. The compound was distilled under high vacuum below 180 °C and collected into multiple fractions. The purity of fractions was analyzed by GC (should be > 98 % with single max impurity < 1.0 %). The less pure fractions (> 85 % and < 98 % pure fraction) were pooled together and the distillation was repeated to get 19g (32% yield) of 4,4,5, 5-tetramethyl-2-(4- (2,2,2-trifluoroethoxy)phenyl)-l,3,2-dioxaborolane as a pale yellow liquid.*H NMR: δ values with respect to TMS (DMSO-d6; 400 MHz):7.64 (2H, d, 6.8 Hz), 7.06 (2H, d, J = 6.4 Hz), 4.79 (2H, q, J = 6.8 Hz), 1.28 (12H, s).13C NMR: 159.46 (Ar-C-O-), 136.24 (2 x Ar-C-), 127.77 – 120.9 (q, -CF3), 122.0 (Ar-C-B), 114.22 (2 x Ar-C-), 64.75 (q, J = 27.5 Hz).Synthesis of 2-(2.4-difluorophenyl)-l.l-difluoro-3-(lH-tetrazol-l-yl)-l-(5-(4-(2.2.2- trifluoroethoxy)phenyl)pyridin-2-yl)propan-2-ol (1 or la)l-6*-Br or l-7*-Br (14 g, 0.03 mol, 1 eq) was added to tetrahydrofuran (168 mL, 12 vol) at 25 – 35 °C and the resulting solution was heated to 40 – 45 °C. The reaction mixture was maintained at this temperature for 20 – 30 min under argon bubbling. Sodium carbonate (8.59 g, 0.08 mol, 2.5 eq) and water (21 mL, 1.5 vol) were added into the reaction mixture and the bubbling of argon was continued for another 20 – 30 min. 4,4,5, 5-tetramethyl-2-(4-(2,2,2- trifluoroethoxy)phenyl)-l,3,2-dioxaborolane (10.76 g, 1.1 eq) dissolved in tetrahydrofuran (42 mL, 3 vol) was added into the reaction mixture and argon bubbling was continued for 20 – 30 min. Pd(dppf)Cl2 (2.65 g, 0.1 eq) was added to the reaction mixture under argon bubbling and stirred for 20 – 30 min (Reaction mixture turned into dark red color). The reaction mixture was heated to 65 – 70 °C and maintained at this temperature for 3 – 4 h. The reaction progress was monitored by HPLC. The reaction mixture was cooled to 40 – 45 °C and the solvent was distilled under reduced pressure. Toluene (350 mL, 25 vol.) was added to the reaction mixture and stirred for 10 – 15 min followed by the addition of water (140 mL, 10 vol). The reaction mixture was filtered through Hyflo (42 g, 3 times), the layers were separated and the organic layer was washed with water (70 mL, 5 vol) and a 20 % w/w sodium chloride solution (140 mL, 10 vol). The organic layer was treated with charcoal (5.6 g, 0.4 times, neutral chalrcoal), filtered through Hyflo. (lS)-lO-Camphor sulfonic acid (7.2 g, 1 eq.) was added to the toluene layer and the resulting mixture was heated to 70 – 75 °C for 2 – 3 h. The reaction mixture was gradually cooled to 25 – 35 °C and stirred for 1 – 2 h. The solids were filtered, washed with toluene (2 x 5 vol.) and then dried under vacuum below 45 °C to afford 18.0 g of an off white solid. The solids (13.5 g, 1 eq.) were suspended in toluene (135 mL, 10 vol) and neutralized by adding 1M NaOH solution (1.48 vol, 1.1 eq) at 25 – 35 °C and stirred for 20 – 30 min. Water (67.5 mL, 5 vol) was added to the reaction mixture and stirred for 10 – 15 min, and then the layers were separated. The organic layer was washed with water (67.5 mL, 5 vol) to remove the traces of CSA. The toluene was removed under reduced pressure below 45 °C to afford crude 1 or la. Traces of toluene were removed by azeotroping with ethanol (3 x 10 vol), after which light brown solid of crude 1 or la (7.5 g, 80% yield) was obtained.The crude 1 or la (5 g) was dissolved in ethanol (90 mL, 18 vol.) at 20 – 35 °C, and heated to 40 – 45 °C. Water (14 vol) was added to the solution at 40 – 45 °C, the solution was maintained at this temperature for 30 – 45 min and then gradually cooled to 20 – 35 °C. The resulting suspension was continued to stir for 16 – 18 h at 20 – 35 °C, an additional amount of water (4 vol.) was added and the stirring continued for 3 – 4 h. The solids were filtered to afford 4.0 g (80% recovery) of 1 or la (HPLC purity >98%) as an off-white solid.1H NMR: δ values with respect to TMS (DMSO-d6; 400 MHz):9.15 (1H, s, Ar-H), 8.93 (1H, d, J = 0.8 Hz, Ar-H), .8.22 – 8.20 (1H, m, Ar-H), 7.80 (2H, d, J = 6.8 Hz, Ar-H), 7.52 (1H, d, J = 6.8 Hz, Ar-H), 7.29 (1H, d,J = 3.2Hz, Ar-H), 7.27 – 7.21 (1H, m, Ar-H), 7.23 – 7.21 (2H, d, J = 6.8 Hz, Ar-H), 7.19 (1H, d, J = 6.8 Hz, Ar-H), 6.93 – 6.89 (1H, m, Ar-H), 5.68 (1H, / = 12 Hz, -CHAHB), 5.12 (2H, d, J = 11.6 Hz, -CHAHB), 4.85 (2H, q, J = 1.6 Hz).13C NMR: 163.93 – 158.33 (m, 2 x Ar-C), 157.56 (Ar-C), 149.32 (i, Ar-C), 146.40 (Ar-C), 145.02 (Ar-C), 136.20 (Ar-C), 134.26 (2 x Ar-C), 131.88 – 131.74 (m, AR-C), 129.72 (Ar-C), 128.47 (2 x Ar-C), 123.97 (q, -CF2-), 122.41 (Ar-C), 119.30 (-CF3), 118.99 (Ar-C), 115.65 (2 x Ar-C), 110.99 (d, Ar-C), 104.22 (i, Ar-C), 77.41 – 76.80 (m, Ar-C), 64.72 (q, -OCH2-CF3), 50.54 (-CH2-N-).B. Preparation of 1 or la via 4b-Br or 4c-Br


Synthesis of 3-amino-2-(2.4-difluorophenyl)-l.l-difluoro-l-(5-(4-(2.2.2- trifluoroethoxy)phenyl)pyridin-2-yl)propan-2-ol (8a or 8b)Potassium carbonate (30.4 g) and water (53.3 g) were charged to a 1-L flask equipped with overhead stirring, thermocouple, and nitrogen/vacuum inlet valve, and stirred until dissolved. The boronic acid (19.37 g), a solution of 4b-Br or 4c-Br in 2-butanol (103.5 g, 27.8 g theoretical 4b-Br or 4c-Br)) and 2-BuOH (147.1 g) were added and stirred to form a clear mixture. The flask was evacuated and refilled with nitrogen 3 times. Pd(d f)2Cl2 (0.30 g) was added and stirred to form a light orange solution. The flask was evacuated and refilled with nitrogen 4 times. The mixture was heated to 85 °C and stirred overnight and endpointed by HPLC analysis. The reaction mixture was cooled to 60 °C and the layers were allowed to settle. The aqueous layer was separated. The organic layer was washed with 5% NaCl solution (5 x 100 ml) at 30-40 °C. The organic layer was filtered and transferred to a clean flask with rinses of 2-BuOH. The combined solution was 309.7 g, water content 13.6 wt% by KF analysis. The solution was diluted with 2-BuOH (189 g) and water (10 g). Theoretically the solution contained 34.8 g product, 522 ml (15 volumes) of 2-BuOH, and 52.2 ml (1.5 volumes) of water. L-Tartaric acid (13.25 g) was added and the mixture was heated to a target temperature of 70-75 °C. During the heat-up, a thick suspension formed. After about 15 minutes at 70-72 °C the suspension became fluid and easily stirred. The suspension was cooled at a rate of 10 °C/hour to 25 °C then stirred at 25 °C for about 10 hours. The product was collected on a vacuum filter and washed with 10:1 (v/v) 2-BuOH/water (50 ml) and 2- butanol (40 ml). The salt was dried in a vacuum oven at 60 °C with a nitrogen purge for 2 days. The yield was 40.08 g of 8a or 8b as a fluffy, grayish-white solid. The water content was 0.13 wt% by KF analysis. The yield was 87.3% with an HPLC purity of 99.48%. Synthesis of 2-(2,4-difluorophenyl)-l,l-difluoro-3-(lH-tetrazol-l-yl)-l-(5-(4-(2,2,2- trifluoroethoxy)phenyl)pyridin-2-yl)propan-2-ol (1 or la)To a 350 ml pressure bottle were charged acetic acid (73 ml), 8a or 8b (34.8 g), sodium acetate (4.58 g) and trimethylorthoformate (16.0 g). The mixture was stirred for 18 min. at room temperature until a uniform suspension was obtained. Azidotrimethylsilane (8.88 g) was added and the bottle was sealed. The bottle was immersed in an oil bath and magnetically stirred. The oil bath was at 52 °C initially, and was warmed to 62-64 °C over about ½ hour. The suspension was stirred at 62-64 °C overnight. After 20.5 hours the suspension was cooled to room temperature and sampled. The reaction was complete by HPLC analysis. The reaction was combined with three other reactions that used the same raw material lots and general procedure (total of 3.0 g additional starting material). The combined reactions were diluted with ethyl acetate (370 ml) and water (368 ml) and stirred for about ½ hour at room temperature. The layers were settled and separated. The organic layer was washed with 10% K2C03 solution (370 ml/ 397 g) and 20% NaCl solution (370 ml/ 424 g). The organic layer (319 g) was concentrated, diluted with ethanol (202 g) and filtered, rinsed with ethanol (83 g). The combined filtrate was concentrated to 74 g of amber solution.The crude 1 or la solution in ethanol (74 g solution, containing theoretically 31.9 g 1 or la) was transferred to a 2-L flask equipped with overhead stirring, thermocouple, and addition funnel. Ethanol (335 g) was added including that used to complete the transfer of the 1 or la solution. The solution was heated to nominally 50 °C and water (392 g) was added over 12 minutes. The resulting hazy solution was seeded with 1 or la crystals and stirred at 50 °C. After about ½ hour the mixture was allowed to cool to 40 °C over about ½ hour during which time crystallization started. Some darker colored chunky solid separated out from the main suspension. The pH of the crystallizing mixture was adjusted from 4.5 to 6 using 41% KOH (1.7 g). After about 1 hour a good suspension had formed. Additional water (191 g) was added slowly over ½ hour. The suspension was heated to 50 °C and cooled at 5 °C/min to room temperature. After stirring overnight the suspension was cooled in a water bath to 16 °C and filtered after 1 hour. The wet cake was washed with 55:45 (v/v) water/ethanol (2 x 50 ml) and air-dried on the vacuum filter funnel overnight. Further drying at 40 °C in a vacuum oven with a nitrogen bleed resulted in no additional weight loss. The yield was 30.2 g of off-white fine powder plus some darker granular material. By in-process HPLC analysis there was no difference in the chemical purity of the darker and lighter materials. The purity was 99.4%. The water content was 2.16 wt% by KF analysis. The residual ethanol was 1.7 wt% estimated by ‘Ft NMR analysis. The corrected yield was 29.0 g, 91.0% overall yield for tetrazole formation and crystallization. The melting point was 65 °C by DSC analysis.
FDA Approves Mycovia Pharmaceuticals’ VIVJOA™ (oteseconazole), the First and Only FDA-Approved Medication for Recurrent Vulvovaginal Candidiasis (Chronic Yeast Infection)
– Approval of VIVJOA™ marks a significant therapeutic advancement for reducing the incidence of RVVC, a condition with substantial unmet need, in permanently infertile and postmenopausal women
– VIVJOA™ is the first FDA approval in Mycovia’s pipeline of novel treatments for fungal infections
– U.S. commercial launch of VIVJOA™ expected in Q2
April 28, 2022 07:55 AM Eastern Daylight Time
DURHAM, N.C.–(BUSINESS WIRE)–The U.S. Food and Drug Administration (FDA) approved VIVJOA™ (oteseconazole capsules), an azole antifungal indicated to reduce the incidence of recurrent vulvovaginal candidiasis (RVVC) in females with a history of RVVC who are NOT of reproductive potential. VIVJOA is the first and only FDA-approved medication for this condition and provides sustained efficacy demonstrated by significant long-term reduction of RVVC recurrence through 50 weeks versus comparators. VIVJOA is the first FDA-approved product for Mycovia Pharmaceuticals, Inc. (Mycovia), an emerging biopharmaceutical company dedicated to recognizing and empowering those living with unmet medical needs by developing novel therapies.
“We believe the market need for VIVJOA is strong, and we are eager to execute our commercial plans”Tweet this
RVVC, also known as chronic yeast infection, is defined by the Centers for Disease Control and Prevention (CDC) as three or more symptomatic acute episodes of yeast infection in 12 months. RVVC is a distinct condition from vulvovaginal candidiasis (VVC), and until now, there have been no FDA-approved medications specifically indicated for it. Nearly 75% of all adult women will have at least one yeast infection in their lifetime, with approximately half experiencing a recurrence. Of those women, up to 9% develop RVVC.
“After nearly two decades of living with chronic yeast infection and feeling like there was no hope from the itchiness, irritation and constant dread of when the next yeast infection would return, I was overjoyed to even be a part of this clinical trial,” said Leslie Ivey, RVVC patient and clinical trial participant. “It is gratifying to see RVVC finally get the attention it deserves.”
Symptoms of RVVC include vaginal itching, burning, irritation and inflammation. Some women may experience abnormal vaginal discharge and painful sexual intercourse or urination, causing variable but often severe discomfort and pain.
VIVJOA’s FDA approval is based upon the positive results from three Phase 3 trials of oteseconazole – two global, pivotal VIOLET studies and one U.S.-focused ultraVIOLET study, including 875 patients at 232 sites across 11 countries. In the two global VIOLET studies, 93.3% and 96.1% of women with RVVC who received VIVJOA did not have a recurrence for the 48-week maintenance period compared to 57.2% and 60.6% of patients who received placebo (p <0.001). In the ultraVIOLET study, 89.7% of women with RVVC who received VIVJOA cleared their initial yeast infection and did not have a recurrence for the 50-week maintenance period compared to 57.1% of those who received fluconazole followed by placebo (p <0.001). The most common side effects reported in Phase 3 clinical studies were headache (7.4%) and nausea (3.6%). VIVJOA is contraindicated in those with a hypersensitivity to oteseconazole, and based on data from rat studies, also in females who are of reproductive potential, pregnant, or lactating. Please see additional Important Safety Information below.
Patrick Jordan, CEO of Mycovia Pharmaceuticals and Partner at NovaQuest Capital Management, stated, “We celebrate this important milestone for Mycovia, as VIVJOA is the first antifungal in our pipeline to obtain FDA approval and achieves our goal to fulfill a previously unmet medical need among women suffering from RVVC. We are honored to lead this advancement in women’s health.”
“We believe the market need for VIVJOA is strong, and we are eager to execute our commercial plans,” Jordan continued. “As we enter a new chapter of our history as a commercial biopharmaceutical company, we will continue driving our mission forward to develop novel therapies for overlooked conditions.”
Oteseconazole is designed to inhibit fungal CYP51, which is required for fungal cell wall integrity, and this selective interaction is also toxic to fungi, resulting in the inhibition of fungal growth. Due to its chemical structure, oteseconazole has a lower affinity for human CYP enzymes as compared to fungal CYP enzymes. The FDA granted oteseconazole Qualified Infectious Disease Product and Fast Track designations.
“A medicine with VIVJOA’s sustained efficacy combined with the clinical safety profile has been long needed, as until now, physicians and their patients have had no FDA-approved medications for RVVC,” stated Stephen Brand, Ph.D., Chief Development Officer of Mycovia. “We are excited to be the first to offer a medication designed specifically for RVVC, a challenging and chronic condition that is expected to increase in prevalence over the next decade.”
Mycovia is planning its commercial launch of VIVJOA™ in the second quarter of 2022.
About Recurrent Vulvovaginal Candidiasis
RVVC is a debilitating, chronic infectious condition that affects 138 million women worldwide each year. RVVC, also known as chronic yeast infection, is a distinct condition from vulvovaginal candidiasis (VVC) and defined as three or more symptomatic acute episodes of yeast infection in 12 months. Primary symptoms include vaginal itching, burning, irritation and inflammation. Some women may experience abnormal vaginal discharge and painful sexual intercourse or urination, causing variable but often severe discomfort and pain.
About VIVJOA™
VIVJOA™ (oteseconazole) is an azole antifungal indicated to reduce the incidence of recurrent vulvovaginal candidiasis (RVVC) in females with a history of RVVC who are NOT of reproductive potential. VIVJOA is the first and only FDA-approved medication that provides sustained efficacy demonstrated by significant long-term reduction of RVVC recurrence through 50 weeks versus comparators. Oteseconazole is designed to inhibit fungal CYP51, which is required for fungal cell wall integrity, and this selective interaction is also toxic to fungi, resulting in the inhibition of fungal growth. Due to its chemical structure, oteseconazole has a lower affinity for human CYP enzymes as compared to fungal CYP enzymes. The FDA approved VIVJOA based upon the positive results from three Phase 3 clinical trials of oteseconazole – two global, pivotal VIOLET studies and one U.S.-focused ultraVIOLET study, including 875 patients at 232 sites across 11 countries.
References
- ^ Jump up to:a b https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/215888s000lbl.pdf
- ^ “Vivjoa: FDA-Approved Drugs”. U.S. Food and Drug Administration (FDA). Retrieved 27 April 2022.
- ^ Jump up to:a b “FDA Approves Mycovia Pharmaceuticals’ VIVJOA (oteseconazole), the First and Only FDA-Approved Medication for Recurrent Vulvovaginal Candidiasis (Chronic Yeast Infection)” (Press release). Mycovia Pharmaceuticals. 28 April 2022. Retrieved 28 April 2022 – via Business Wire.
- ^ World Health Organization (2016). “International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 76”. WHO Drug Information. 30 (3). hdl:10665/331020.
Further reading
- Sobel JD, Nyirjesy P (December 2021). “Oteseconazole: an advance in treatment of recurrent vulvovaginal candidiasis”. Future Microbiology. 16: 1453–1461. doi:10.2217/fmb-2021-0173. PMID 34783586.
External links
- “Oteseconazole”. Drug Information Portal. U.S. National Library of Medicine.
- Clinical trial number NCT03562156 for “A Study of Oral Oteseconazole for the Treatment of Patients With Recurrent Vaginal Candidiasis (Yeast Infection) (VIOLET)” at ClinicalTrials.gov
- Clinical trial number NCT03561701 for “A Study of Oral Oteseconazole (VT-1161) for the Treatment of Patients With Recurrent Vaginal Candidiasis (Yeast Infection) (VIOLET)” at ClinicalTrials.gov
- Clinical trial number NCT03840616 for “Study of Oral Oteseconazole (VT-1161) for Acute Yeast Infections in Patients With Recurrent Yeast Infections (ultraVIOLET)” at ClinicalTrials.gov
| Clinical data | |
|---|---|
| Trade names | Vivjoa |
| Other names | VT-1161 |
| License data | US DailyMed: Oteseconazole |
| Routes of administration | By mouth |
| Drug class | Antifungal |
| ATC code | J02AC06 (WHO) |
| Legal status | |
| Legal status | US: ℞-only [1] |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 1340593-59-0 |
| PubChem CID | 77050711 |
| DrugBank | DB13055 |
| ChemSpider | 52083215 |
| UNII | VHH774W97N |
| KEGG | D11785 |
| ChEBI | CHEBI:188153 |
| ChEMBL | ChEMBL3311228 |
| ECHA InfoCard | 100.277.989 |
| Chemical and physical data | |
| Formula | C23H16F7N5O2 |
| Molar mass | 527.403 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI |
/////////OTESECONAZOLE, vt 1161, fungal infection, Candida albicans infection, onychomycosis, PHASE 3,



AS ON JUNE2025 4.45 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
C1=CC(=CC=C1C2=CN=C(C=C2)C(C(CN3C=NN=N3)(C4=C(C=C(C=C4)F)F)O)(F)F)OCC(F)(F)F
NEW DRUG APPROVALS
ONE TIME
$10.00
Nangibotide


Nangibotide
LQEEDAGEYGCM-amide
CAS 2014384-91-7
- Molecular FormulaC54H82N14O22S2
- Average mass1343.439 Da
- 2014384‐91‐7
- L-Leucyl-L-glutaminyl-L-α-glutamyl-L-α-glutamyl-L-α-aspartyl-L-alanylglycyl-L-α-glutamyl-L-tyrosylglycyl-L-cysteinyl-L-methioninamide
- LR 12 peptide
- LQEEDAGEYG CM
L-Leucyl-L-glutaminyl-L-glutaminyl-L-α-glutamyl-L-α-aspartyl-L-alanylglycyl-L-α-glutamyl-L-tyrosylglycyl-L-cysteinyl-L-methionine
L-Methionine, L-leucyl-L-glutaminyl-L-glutaminyl-L-α-glutamyl-L-α-aspartyl-L-alanylglycyl-L-α-glutamyl-L-tyrosylglycyl-L-cysteinyl-нангиботидمانغيبوتيد南吉博肽
| Sequence (one letter code) | LQEEDAGEYGCM-amide |
|---|---|
| Sequence (three letter code) | H-Leu-Gln-Glu-Glu-Asp-Ala-Gly-Glu-Tyr-Gly-Cys-Met-NH2 |
- OriginatorInotrem
- ClassAnti-infectives; Anti-inflammatories; Anti-ischaemics; Antivirals; Peptides
- Mechanism of ActionTREML1 protein inhibitors
- Phase II/IIICOVID 2019 infections
- Phase IISeptic shock
- Phase IMyocardial infarction
- 12 Jul 2021Inotrem has patents pending for nangibotide use in severe forms of COVID-19
- 12 Jul 2021Inotrem receives funding from French government by Bpifrance for nangibotide development in COVID-2019 infections
- 12 Jul 2021Inotrem receives authorization from both the French and Belgian authorities to proceed with clinical development of nangibotide up to registration in COVID-2019 infections
Nangibotide, also referred as LR12, is an antagonist of triggering receptor expressed on myeloid cells (TREM)-1, and was derived from residues 94 to 105 of TREM-like transcript-1 (TLT-1).
TREM-1 plays a crucial role in the onset of sepsis by amplifying the host immune response. TLT-1– and TLT-1–derived peptides therefore exhibit anti-inflammatory properties by dampening TREM-1 signalling. LR12 blocks TREM-1 by binding to the TREM-1 ligand and provides protective effects during sepsis such as inhibiting hyper-responsiveness, organ damage, and death, without causing deleterious effects. The protective effects of modulating TREM-1 signalling are also evident in other models of inflammation such as: pancreatitis; haemorrhagic shock; inflammatory bowel diseases and inflammatory arthritis
Inotrem is developing the peptide nangibotide, a triggering receptor expressed on myeloid cells 1 inhibitor, for treating sepsis and septic shock. In July 2021, this drug was reported to be in phase 3 clinical development.
Nangibotide is an inhibitor of TREM-1, a receptor found on certain white blood cells. Activation of TREM-1 stimulates inflammation. Nangibotide is therefore being investigated as a treatment for the overwhelming inflammation typically seen in severe sepsis.
Mode of action
TREM-1 is a receptor found on neutrophils, macrophages and monocytes, key elements of the immune system. Activation of TREM-1 results in expression of NF-κB, which promotes systemic inflammation. Nangibotide inhibits TREM-1, thereby preventing the inflammatory activation. Absence of TREM-1 results in vastly reduced inflammation without impairing the ability to fight infection.[2]
Animal models
LR17, a mouse equivalent of nangibotide, improves survival in mouse models of severe sepsis.[3] In a pig model of sepsis, LR12 – another animal equivalent of nangibotide – resulted in significantly improved haemodynamics and less organ failure.[4] In monkeys, LR12 also reduced the inflammatory and hypotensive effects of sepsis.[5]
Human studies
Nangibotide has demonstrated safety in Phase 1 (healthy volunteers)[6] and Phase 2 (sick patients with septic shock)[7] studies. The ASTONISH trial will examine clinical efficacy in 450 patients with septic shock.[8]
Inotrem Receives Approval to Expand Nangibotide Clinical Trial in Critically Ill COVID-19 Patients and Receives Additional Public Funding of €45 Million
- Inotrem’s phase 2/3 clinical trial “ESSENTIAL” will enroll up to 730 patients in Europe to demonstrate the safety and efficacy of nangibotide to treat critically ill COVID-19 patients with respiratory failure.
- Recent preclinical studies have strengthened the body of evidence for targeting the TREM-1 pathway which is activated in a subset of patients suffering from severe COVID-19.
July 12, 2021 03:00 AM Eastern Daylight Time
PARIS–(BUSINESS WIRE)–Inotrem S.A., a biotechnology company specializing in the development of immunotherapies targeting the TREM-1 pathway, announces that it has obtained authorization to pursue the clinical development of nangibotide up to registration in COVID-19 patients from both the French and Belgian competent authorities.
As part of this program, Inotrem receives additional 45 million euros in public funding under the “Capacity Building” Call for Expression of Interest, operated on behalf of the French government by Bpifrance, the French national investment bank, as part of the Programme d’investissements d’avenir (PIA) and the France Recovery Plan, bringing French state support for the project to a total of 52,5 million euros. This public funding will support Inotrem’s clinical program including the phase 2/3 study “ESSENTIAL” which aims to demonstrate the efficacy and safety of nangibotide in treating patients in respiratory distress with severe forms of COVID-19.
The primary endpoint is evaluation of the impact of nangibotide on the progression of disease in patients receiving ventilatory support due to COVID-19 as well as on the severity of the respiratory failure, duration of mechanical ventilation, length of stay in intensive care and mortality. In “ESSENTIAL”, a Phase 2/3 clinical program, up to 730 patients will be enrolled initially in France and Belgium and, possibly in other European countries. Pre-defined interim analyses will be conducted by an independent Data Monitoring Board to test futility and to allow for the study design to be adapted as necessary. “ESSNTIAL” is the continuation of a 60 patients phase 2a evaluating the safety and efficacy of nangibotide in patients suffering from severe COVID-19. In July 2020, the CoviTREM-1 consortium, which includes the Nancy and Limoges university hospitals and Inotrem, obtained public funding of 7,5 million euros under the “PSPC-COVID” call for projects, operated on behalf of the French government by Bpifrance
New pre-clinical studies with nangibotide have demonstrated that the administration of nangibotide in murine models infected with SARS-CoV-2 was associated with a decrease in inflammatory mediators and an improvement of clinical signs, in particular respiratory function, and survival. Inotrem also confirmed in 3 different and independent cohorts that sTREM-1, a marker of the activation of the TREM-1 biological pathway, is associated with both severity and mortality in critically ill COVID-19 patients.
Leveraging the results of these preclinical studies and the implications for the role of the TREM-1 pathway in COVID-19, Inotrem has filed additional patents to cover nangibotide use in severe forms of COVID-19 as well as the use of sTREM-1 as a biomarker and companion diagnostic. This significantly strengthens Inotrem’s already broad patent estate.
Jean-Jacques Garaud, Executive Vice-President, Head of Scientific and Medical Affairs and Inotrem’s co-founder said :“We are eager to pursue the development of nangibotide in these severe forms of COVID-19. Nangibotide is a TREM-1 inhibitor which has already demonstrated a trend towards efficacy in septic shock patients and has the potential to modulate the dysregulated immune response in critically ill COVID-19 patients. With this large clinical study, we can demonstrate efficacy for nangibotide in a further indication with the goals of reducing the duration of hospitalization and mortality.”
Sven Zimmerman, CEO of Inotrem, also declared: “The size of the financial support awarded to us as part of the French government’s initiative against COVID-19 is a testimony to the relevance of targeting the TREM-1 pathway with nangibotide in these severely ill patients. We are delighted by the confidence placed in our technology and our team. Everyone at Inotrem is fully committed to deliver on this ambitious program alongside nangibotide’s ongoing Phase 2b trial in septic shock patients.”
About Inotrem
Inotrem S.A. is a biotechnology company specialized in immunotherapy for acute and chronic inflammatory syndromes. The company has developed a new concept of immunomodulation that targets the TREM-1 pathway to control unbalanced inflammatory responses. Through its proprietary technology platform, Inotrem has developed the first-in-class TREM-1 inhibitor, LR12 (nangibotide), with potential applications in a number of therapeutic indications such as septic shock and myocardial infarction. In parallel, Inotrem has also launched another program to develop a new therapeutic modality targeting chronic inflammatory diseases. The company was founded in 2013 by Dr. Jean-Jacques Garaud, a former head of research and early development at the Roche Group, Prof. Sébastien Gibot and Dr. Marc Derive. Inotrem is supported by leading European and North American investors.
About TREM-1 pathway
TREM-1 pathway is an amplification loop of the immune response that triggers an exuberant and hyperactivated immune state which is known to play a crucial role in the pathophysiology of septic shock and acute myocardial infarction.
About Nangibotide
Nangibotide is the formulation of the active ingredient LR12, which is a 12 amino-acid peptide prepared by chemical synthesis. LR12 is a specific TREM-1 inhibitor, acting as a decoy receptor and interfering in the binding of TREM-1 and its ligand. In preclinical septic shock models, nangibotide was able to restore appropriate inflammatory response, vascular function, and improved animals’ survival post septic shock.
About ESSENTIAL study:
The Efficacy and Safety Study Exploring Nangibotide Treatment in COVID-19 pAtients with ventiLatory support, is a randomized, double-blind, placebo-controlled confirmatory study with adaptive features that will be performed in Europe. This is a pivotal study and it is expected that based on its results, nangibotide could be registered in this indication. The first part of the study (i.e.: 60 patients) has been already finalized and assessed by an independent data monitoring committee with excellent safety results. The study will recruit up to 730 patients in up to 40 sites. Several interim and futility analyses are foreseen as part of the adaptive design of the study.
About Bpifrance
Bpifrance is the French national investment bank: it finances businesses – at every stage of their development – through loans, guarantees, equity investments and export insurances. Bpifrance also provides extra-financial services (training, consultancy.). to help entrepreneurs meet their challenges (innovation, export…).
PATENT
WO-2021144388
Process for preparing nangibotide by solid phase synthesis, useful for treating acute inflammatory disorders such as septic shock. Also claims novel peptide fragments, useful in the synthesis of nangibotide.
Example 1
Preparation of nangibotide by full SPPS (Reference)
Step 1 : Loading of the first amino acid onto the Rink Amide Resin
2 g of MBHA resin (1.0-1.3 mmol/g) was swelled using 16 mL of DMF for 30 min. 2 eq Fmoc-Met-OH (2.4 mmol, 2.67 g), 2 eq DIC (2.4 mmol, 1.136 mL) and 2 eq OxymaPure (2.4 mmol, 1.023 g) were dissolved in 8 mL of DMF at 0.3 M cone, and added to the resin after 5 min. All the coupling steps were conducted in this way unless described differently. The loading step was carried out for 1.5 hour. After the loading, the resin was filtered and washed 3 times with 12 mL of DMF. The Fmoc deprotection step was carried out by addition of 12 mL of 20% piperidine solution in DMF for two 10 min cycles. This step was performed analogously for all the amino acid residues. The loading, calculated by UV absorption for the peptidyl resin, was 0.8 mmol/g.
Step 2: peptide elongation
For the coupling of all the amino acids involved in the synthesis of nangibotide, 3 eq of each amino acid were activated by 3 eq of DIC and OxymaPure dissolved in DMF at 0.3 M cone. At the end of the peptide elongation, a final Fmoc deprotection, as already described, was performed before moving to the cleavage step.
Step 3: Cleavage and precipitation of crude nangibotide
The cleavage of nangibotide off the resin was carried out using a solution of 16 mL of TFA/DODT/TIPS/water in 90/4/3/3 ratio cooled at 0°C. The peptidyl resin was added portionwise in 30 min keeping the internal temperature under 25°C. The cleavage was run for 3.5 hours, then the resin was filtered and washed by 10 mL of TFA for 10 min.
DIPE was used for the precipitation of the peptide, adding 12 volumes (300 mL) dropwise to the peptide TFA solution, keeping the temperature under 20°C. The suspension with nangibotide was filtered on a gooch funnel, the peptide washed again with 100 mL of DIPE and then dried under vacuum overnight. Molar yield 40%. Purity 61%.
Example 2
Preparation of nangibotide by three-fragment condensation
In the approach using three fragments, only the cysteine residue was coupled to the methionine on rink amide resin to prepare fragment 11-12, whereas protected peptide fragments 1-7 and 8-10 were synthesized using 2-CTC resin.
Step 1: Synthesis of fragment 11-12
2 g of MBHA resin (1.0-1.3 mmol/g) was swelled using 16 mL of DMF for 30 min 2 eq of Fmoc-Met-OH (2.4 mmol, 2.67 g), 2 eq DIC (2.4 mmol, 1.136 mL) and 2 eq OxymaPure (2.4 mmol, 1.023 g) were dissolved in 8 mL of DMF at 0.3 M cone, and added to the resin. The loading step was carried out for 1 and half hour. After the loading, the resin was filtered and washed 3 times with 12 mL of DMF. The Fmoc deprotection step was carried out by
addition of 12 mL of a 20% piperidine solution in DMF for two 10 min cycles. Same procedure was repeated for the coupling of Fmoc-Cys(Trt)-OH to obtain resin-attached Fmoc-deprotected fragment 11-12. The loading, calculated by UV absorption for the peptidyl resin relative to the first amino acid inserted, was 0.8 mmol/g.
Step 2: Synthesis of fragments 1-7 and 8-10
For the synthesis of both fragments the loading of 2-chloro trityl chloride resin was performed on 5 g (1.6 mmol/g) using 0.8 eq Fmoc-Gly-OH (6.40 mmol, 1.90 g) dissolved in 30 mL of DCM and addition of 3 eq DIPEA (24 mmol, 4.19 mL). The loading step was carried out for 1 hour, then the resin was washed by 30 mL DCM for three times and eventual Cl-groups were capped by two different capping solutions: first by 30 mL of methanol/DIPEA/DCM (1:2:7) and then by 30 mL AC2O/DIPEA/DCM in the same ratio. After the treatment with these solutions for 15 min and subsequent washing with DCM, the resin was washed three times with DMF, before deprotection of Fmoc and evaluation of the resin loading. Generally, this protocol gave a resin loaded with 1.1 mmol/g Fmoc-Gly-OH. The Fmoc deprotection and coupling step protocols were equally performed with all the amino acids in the respective sequences: Fmoc-Tyr(tBu)-OH and Fmoc-Glu(tBu)-OH for fragment 8-10, and Fmoc-Ala-OH, Fmoc-Asp(OtBu)-OH, Fmoc-Glu(OtBu)-OH twice, Fmoc-Gln(Trt)-OH and Fmoc-Leu-OH for fragment 1-7.
For each coupling, 3 eq amino acid were activated by 3 eq DIC and 3 eq OxymaPure dissolved in DMF at 0.3 M cone.
Fragment Fmoc-Glu(tBu)-Tyr(tBu)-Gly-OH (8-10) was obtained by cleavage off the resin using 6 volumes (30 mL) of a TFA 1.5 % solution in DCM, 5 times for 2 min. The final TFA solution was neutralized by 1.2 eq pyridine (15.89 mmol, 1.3 mL) diluted in 30 mL methanol. The final solution was concentrated to 50 mL under vacuum then washed by water and brine. The organic layer was dried by anhydrous sodium sulphate, filtered and further concentrated before crystallization of the tripeptide with 5 volumes of petroleum ether at 0°C. The peptide was filtered, washed by petroleum ether and dried overnight in a vacuum oven at 37°C. Molar yield 65%. Purity 90%.
Fragment Fmoc-Leu-Gln(Trt)-Glu(OtBu)-Glu(OtBu)-Asp(OtBu)-Ala-Gly-OH (1-7) was obtained by cleavage off the resin using 6 volumes (30 mL) of a TFA 1.5 % solution in DCM, 5 times for 2 min. The final TFA solution was neutralized by 1.2 eq pyridine (15.89 mmol, 1.3 mL) diluted in 30 mL methanol. The DCM was evaporated and replaced by methanol, adding and evaporating 30 mL methanol a couple of times till one third of the volume. The peptide fragment was precipitated by adding 5 volumes (150 mL) water to the methanol solution at 0°C and filtered after stirring for 30 min. The full protected heptapeptide was washed by water and dried overnight in a vacuum oven at 37°C. Molar yield 85%. Purity 89%.
Step 3: Synthesis of fragment 8-12 (Fragment condensation 1)
The fragment condensation between Fmoc-Glu(tBu)-Tyr(tBu)-Gly-OH (8-10) and H-Cys(Trt)-Met-MBHA resin (11-12) was carried out activating 2 eq (1.6 mmol, 1.12 g) of fragment 8-10 dissolved in 6 mL of DMF at 40°C by using 2 eq OxymaPure (1.6 mmol, 0.22 g) and 2 eq DIC (1.6 mmol, 0.25 mL) for 10 min. The activated ester of tripeptide 8-10 was added to the resin-attached fragment 11-12 and stirred for 3 hours at 40°C. After filtration, the resin was washed three times by 15 mL DMF and then capped by 12 mL of AC2O 10% in DMF for 15 min. The resin was washed three timed by 12 mL DMF before deprotection of Fmoc to finally obtain resin-attached Fmoc-protected fragment 8-12. Molar yield 91%. Purity 89%.
Step 4: Synthesis of nanaibotide (Fragment condensation 2)
The fragment condensation between fragment 1-7 and H-Glu(OtBu)-Tyr(tBu)-Gly-Cys(Trt)-Met-MBHA resin (8-12) was carried out activating 1.5 eq (2.25 mmol, 2.64 g) of fragment 1-7 dissolved in 25 mL DMF at 40°C by using 2 eq OxymaPure (2.25 mmol, 0.32 g) and 2 eq DIC (2.25 mmol, 0.35 mL) for 15 min. The activated ester of fragment 1-7 was added to the resin-attached fragment 8-12 and stirred for 3.5 hours at 40°C. After filtration, the resin was washed three times by 12 mL DMF before deprotection of Fmoc with the standard procedure described above. After Fmoc deprotection, the resin was washed again by DMF and DCM and then dried at vacuum pump.
Step 5: Cleavage and precipitation of crude nanaibotide
The cleavage of nangibotide off the resin was carried out using a solution of 16 mL of TFA/DODT/TIPS/water in 90/4/3/3 ratio cooled at 0°C. The peptidyl resin was added portionwise in 30 min keeping the internal temperature under 25°C. The cleavage was run for 3.5 hours, then the resin filtered and washed by 10 mL of TFA for 10 min.
DIPE was used to precipitate the peptide, adding 12 volumes (300 mL) dropwise to the peptide TFA solution, keeping the temperature under 20°C. The suspension with nangibotide was filtered on a gooch funnel, the peptide washed again with 100 mL of DIPE and then dried at vacuum pump overnight. Molar yield 61%. Purity 73%.
Example 3
Preparation of nangibotide by two-fragment condensation
In the approach using two fragments, the SPPS elongation onto MBHA resin, as described in Example 2, step 1, was continued until Glu8 was attached to provide fragment 8-12, then fragment 1-7, synthesized on 2-CTC resin as described in example 2, step 2, was coupled to the resin-attached fragment 8-12 as described in example 2, step 4.
Step 1: Synthesis of fragment 8-12
2 g of MBHA resin (1.0-1.3 mmol/g) was swelled using 16 mL of DMF for 30 min 2 eq of Fmoc-Met-OH (2.4 mmol, 2.67 g), 2 eq DIC (2.4 mmol, 1.136 mL) and 2 eq OxymaPure (2.4 mmol, 1.023 g) were dissolved in 8 mL of DMF at 0.3 M cone, and added to the resin. The loading step was carried out for 1 and half hour. After the loading, the resin was filtered and washed 3 times with 12 mL of DMF. The Fmoc deprotection step was carried out by addition of 12 mL of a 20% piperidine solution in DMF for two 10 min cycles. Same procedure was repeated for the coupling of Fmoc-Cys(Trt)-OH; Fmoc-Glu(OtBu)-OH; Fmoc-Tyr(tBu)-OH; Fmoc-Gly-OH to obtain fragment 8-12. The loading, calculated by UV absorption for the peptidyl resin relative to the first amino acid inserted, was 0.8 mmol/g. Molar yield 88%. Purity 83%.
Step 2: Synthesis of nanaibotide (Fragment condensation 2)
The final fragment condensation was performed as described in example 2, step 4.
Step 3: Cleavage and precipitation of crude nanaibotide
The cleavage of nangibotide off the resin was carried out as described in example 2, step 5. Molar yield 60%. Purity 70%.
PAPER
Methods in enzymology (2000), 312, 293-304
Journal of the American College of Cardiology (2016), 68(25), 2776-2793
PATENT
https://patents.google.com/patent/WO2011124685A1/en
Product pat, WO2011124685 ,protection in the EU states and the US April 2031
References
- ^ Cuvier V, Lorch U, Witte S, Olivier A, Gibot S, Delor I, Garaud JJ, Derive M, Salcedo-Magguilli M (2018). “A first-in-man safety and pharmacokinetics study of nangibotide, a new modulator of innate immune response through TREM-1 receptor inhibition”. Br J Clin Pharmacol. 84 (10): 2270–2279. doi:10.1111/bcp.13668. PMC 6138490. PMID 29885068.
- ^ Weber B, Schuster S, Zysset D, Rihs S, Dickgreber N, Schürch C, Riether C, Siegrist M, Schneider C, Pawelski H, Gurzeler U, Ziltener P, Genitsch V, Tacchini-Cottier F, Ochsenbein A, Hofstetter W, Kopf M, Kaufmann T, Oxenius A, Reith W, Saurer L, Mueller C (2014). “TREM-1 deficiency can attenuate disease severity without affecting pathogen clearance”. PLOS Pathog. 10 (1): e1003900. doi:10.1371/journal.ppat.1003900. PMC 3894224. PMID 24453980.
- ^ Derive M, Bouazza Y, Sennoun N, Marchionni S, Quigley L, Washington V, Massin F, Max JP, Ford J, Alauzet C, Levy B, McVicar DW, Gibot S (1 June 2012). “Soluble TREM-like transcript-1 regulates leukocyte activation and controls microbial sepsis”. Journal of Immunology. 188 (11): 5585–5592. doi:10.4049/jimmunol.1102674. PMC 6382278. PMID 22551551.
- ^ Derive M, Boufenzer A, Bouazza Y, Groubatch F, Alauzet C, Barraud D, Lozniewski A, Leroy P, Tran N, Gibot S (Feb 2013). “Effects of a TREM-like transcript 1-derived peptide during hypodynamic septic shock in pigs”. Shock. 39 (2): 176–182. doi:10.1097/SHK.0b013e31827bcdfb. PMID 23324887. S2CID 23583753.
- ^ Derive M, Boufenzer A, Gibot S (April 2014). “Attenuation of responses to endotoxin by the triggering receptor expressed on myeloid cells-1 inhibitor LR12 in nonhuman primate”. Anaesthesiology. 120 (4): 935–942. doi:10.1097/ALN.0000000000000078. PMID 24270127. S2CID 10347527.
- ^ Cuvier V, Lorch U, Witte S, Olivier A, Gibot S, Delor I, Garaud JJ, Derive M, Salcedo-Magguilli M (2018). “A first-in-man safety and pharmacokinetics study of nangibotide, a new modulator of innate immune response through TREM-1 receptor inhibition”. Br J Clin Pharmacol. 84 (10): 2270–2279. doi:10.1111/bcp.13668. PMC 6138490. PMID 29885068.
- ^ François B, Wittebole X, Ferrer R, Mira JP, Dugernier T, Gibot S, Derive M, Olivier A, Cuvier V, Witte S, Pickkers P, Vandenhende F, Garaud JJ, Sánchez M, Salcedo-Magguilli M, Laterre PF (July 2020). “Nangibotide in patients with septic shock: a Phase 2a randomized controlled clinical trial”. Intensive Care Medicine. 46 (7): 1425–1437. doi:10.1007/s00134-020-06109-z. PMID 32468087. S2CID 218912723.
- ^ “Efficacy, Safety and Tolerability of Nangibotide in Patients With Septic Shock (ASTONISH)”. ClinicalTrials.gov. US National Library of Medicine. Retrieved 13 July 2020.
Derive et al (2013) Effects of a TREM-Like Transcript 1–Derived Peptide During Hypodynamic Septic Shock in Pigs. Shock39(2) 176 PMID: 23324887
Derive et al (2014) Attenuation of Responses to Endotoxin by the Triggering Receptor Expressed on Myeloid Cells-1 Inhibitor LR12 in Nonhuman Primate. Anesthesiology120(4) 935 PMID: 24270127
Derive et al (2012) Soluble Trem-like Transcript-1 Regulates Leukocyte Activation and Controls Microbial Sepsis. J. Immunol.188(11) 5585 PMID: 22551551
| Clinical data | |
|---|---|
| Routes of administration | Intravenous; intraperitoneal |
| Physiological data | |
| Receptors | TREM-1 |
| Metabolism | Enzymatic in bloodstream |
| Pharmacokinetic data | |
| Metabolism | Enzymatic in bloodstream |
| Elimination half-life | 3 minutes |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 2014384‐91‐7 |
| ChemSpider | 64835227 |
| UNII | 59HD7BLX9H |
| ChEMBL | ChEMBL4297793 |
| Chemical and physical data | |
| Formula | C54H82N14O22S2 |
| Molar mass | 1343.439 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI |
//////////////Nangibotide, phase 3, нангиботид , مانغيبوتيد , 南吉博肽 , INOTREM, SEPTIC SHOCK, PEPTIDE
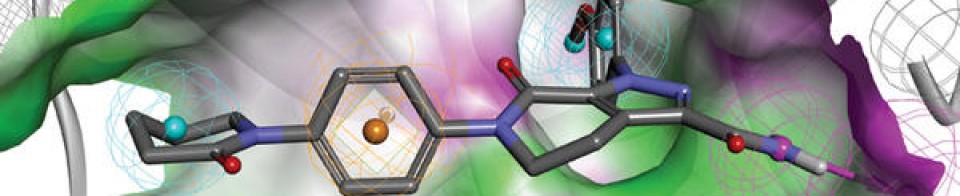
NEW DRUG APPROVALS
one time
$10.00
Melarsoprol

Melarsoprol
MelarsoprolCAS Registry Number: 494-79-1
CAS Name: 2-[4-[(4,6-Diamino-1,3,5-triazin-2-yl)amino]phenyl]-1,3,2-dithiarsolane-4-methanolAdditional Names:p-[(4,6-diamino-s-triazin-2-yl)amino]dithiobenzenearsonous acid 3-hydroxypropylene ester; 2-p-(4,6-diamino-s-triazin-2-ylamino)phenyl-4-hydroxymethyl-1,3,2-dithiarsoline; 2-(4-melamin-2-ylphenyl)-4-hydroxymethyl-1,3-dithia-2-arsolaneTrademarks: Mel B; Arsobal (Specia)
Molecular Formula: C12H15AsN6OS2Molecular Weight: 398.34
Percent Composition: C 36.18%, H 3.80%, As 18.81%, N 21.10%, O 4.02%, S 16.10%Literature References: Prepn: Friedheim, US2659723 (1953); US2772303 (1956).Properties: Practically insol in water, cold ethanol, methanol. Sol in propylene glycol.
Therap-Cat: Antiprotozoal (Trypanosoma).Keywords: Antiprotozoal (Trypanosoma).
Melarsoprol is a medication used for the treatment of sleeping sickness (African trypanosomiasis).[1] It is specifically used for second-stage disease caused by Trypanosoma brucei rhodesiense when the central nervous system is involved.[1] For Trypanosoma brucei gambiense, eflornithine or fexinidazole is usually preferred.[1] It is effective in about 95% of people.[3] It is given by injection into a vein.[2]
Melarsoprol has a high number of side effects.[4] Common side effects include brain dysfunction, numbness, rashes, and kidney and liver problems.[2] About 1-5% of people die during treatment.[3] In those with glucose-6-phosphate dehydrogenase (G6PD) deficiency, red blood cell breakdown may occur.[2] It has not been studied in pregnancy.[2] It works by blocking pyruvate kinase, an enzyme required for aerobic metabolism by the parasite.[2]
Melarsoprol has been used medically since 1949.[1] It is on the World Health Organization’s List of Essential Medicines, the safest and most effective medicines needed in a health system.[5] In regions of the world where the disease is common, melarsoprol is provided for free by the World Health Organization.[4] It is not commercially available in Canada or the United States.[2] In the United States, it may be obtained from the Centers for Disease Control and Prevention, while in Canada it is available from Health Canada.[1][2]
Medical uses
People diagnosed with trypanosome-caused disease should be treated with an anti-trypanosomal. Treatment is based on stage, 1 or 2, and parasite,T. b. rhodesiense or T. b. gambiense. In stage 1 disease, trypanosomes are present only in the peripheral circulation. In stage 2 disease, trypanosomes have crossed the blood-brain barrier and are present in the central nervous system.[6]
The following are considerable treatment options:[6]
Melarsoprol is a treatment used during the second stage of the disease. So far, it is the only treatment available for late-stage T. b. rhodesiense.[7]
Due to high toxicity, melarsoprol is reserved only for the most dangerous cases. Other agents associated with lower toxicity levels are used during stage 1 of the disease.[8] The approval of the nifurtimox-eflornithine combination therapy (NECT) in 2009 for the treatment of T. b. gambiense limited the use of melarsoprol to the treatment of second-stage T. b. rhodesiense.[9]
Failure rates of 27% in certain African countries have been reported.[10] This was caused by both drug resistance and additional mechanisms that have not yet been elucidated. Resistance is likely due to transport problems associated with the P2 transporter, an adenine-adenosine transporter. Resistance can occur with point mutations within this transporter.[11] Resistance has been present since the 1970s.[12]
Mechanism of action
Melarsoprol is a prodrug, which is metabolized to melarsen oxide (Mel Ox) as its active form. Mel Ox is an phenylarsonous acid derivative that irreversibly binds to sulfhydryl groups on pyruvate kinase, which disrupts energy production in the parasite. The inability to distinguish between host and parasite PK renders this drug highly toxic with many side effects.
Mel Ox also reacts with trypanothione (a spermidine-glutathione adduct that replaces glutathione in trypanosomes). It forms a melarsen oxide-trypanothione adduct (Mel T) that competitively inhibits trypanothione reductase, effectively killing the protist.[11]
Pharmacokinetics
The half-life of melarsoprol is less than one hour, but bioassays indicate a 35-hour half-life. This is commonly associated with pharmacologic agents that have active metabolites. One such metabolite, Mel Ox, reaches maximum plasma levels about 15 minutes after melarsoprol injection. Melarsoprol clearance is 21.5 ml/min/kg and the Mel Ox half-life is approximately 3.9 hours.[13]
Dosage
Two arsenic-containing stereoisomers exist in a 3:1 molar ratio. Since melarsoprol is insoluble in water, dosage occurs via a 3.6% propylene glycol intravenous injection.[11] To avoid the risk of injection site reactions, melarsoprol must be given slowly.[citation needed]
Melarsoprol used for the treatment of African trypanosomiasis with CNS involvement is given under a complicated dosing schedule. The dosing schedule for children and adults is 2–3.6 mg/kg/day intravenously for three days, then repeated every seven days for a total of three series.[6] To monitor for relapse, follow-up is recommended every six months for at least two years.[3]
Side effects
Although melarsoprol cures about 96% of people with late stage disease, its toxicity limits its use.[7] About 1-5% of people die during treatment.[3] As a toxic organic compound of arsenic, melarsoprol is a dangerous treatment that is typically only administered by injection under the supervision of a licensed physician. Notable side effects are similar to arsenic poisoning. Among clinicians, it is colloquially referred to as “arsenic in antifreeze”.[14] Severe and life-threatening adverse reactions are associated with melarsoprol. It is known to cause a range of side effects including convulsions, fever, loss of consciousness, rashes, bloody stools, nausea and vomiting. In approximately 5-10% of cases, it causes encephalopathy. Of those, about 50% die due to encephalopathy-related adverse reactions.[6] Additional potentially serious side effects of melarsoprol include damage to the heart, presence of albumin in the urine that could be associated with kidney damage, and an increase in blood pressure.[3]
Cautions
Numerous warnings must be examined before melarsoprol treatment can be initiated. Prior to initiation, the following must be noted: glucose-6-phosphate dehydrogenase deficiency, kidney or liver disease, cardiac problems (high blood pressure, irregular beating of the heart or arrhythmias, any damage to the heart muscles and potential signs of heart failure), preexisting nervous system disorders, and any signs of leprosy.
Routine laboratory testing is needed before and after melarsoprol initiation. Laboratory parameters for both therapeutic effects and toxic effects need to be evaluated.
Blood analysis is used to detect the presence of trypanosomes. An evaluation of the cerebrospinal fluid via a lumbar puncture is also used to determine an individual’s white blood count and level of protein. These are diagnostic criteria such that the presence of trypanosomes, an elevated white blood count greater than five per microliter, or a protein content greater than 40 mg are considered abnormal and initiation should be considered. Continuous cerebrospinal fluid evaluation should be repeated every six months for at least three years in individuals that have undergone melarsoprol treatment.
To assess potential concerns related to toxicity, the following should be completed: a complete blood count, an assessment of electrolyte levels, liver and kidney function tests, and a urinalysis to detect the appearance, concentration and content of the urine.
Melarsoprol should be given using glass syringes (if they can be reliably sterilised). The propylene glycol it contains is capable of dissolving plastic.[15]
Pregnancy and breastfeeding
Currently, melarsoprol is not recommended for use in pregnant women. The World Health Organization suggests that treatment be deferred until immediately after delivery since the effects of the medication on the developing fetus have not yet been established.[3]
Lactation guidelines associated with melarsoprol have not yet been established.
Society and culture
Melarsoprol is produced by Sanofi-Aventis and under an agreement with the WHO, they donate melarsoprol to countries where the disease is common.[medical citation needed]
Melarsoprol was used to treat a patient with African trypanosomiasis on season 1 episode 7 “Fidelity” of the medical drama House MD.[16]
PAPER
Journal of Organometallic Chemistry (2006), 691(5), 1081-1084.

https://www.sciencedirect.com/science/article/abs/pii/S0022328X05009344
Graphical abstract
(2-Phenyl-[1,3,2]dithiarsolan-4-yl)-methanol derivatives were tested on K562 and U937 human leukemia cell lines. Their systemic toxicity was estimated by the corresponding LD50 on mice. The cytotoxic activity of each derivative was significantly better than that of arsenic trioxide and the therapeutic index (T.I. = LD50/IC50) was improved.

References
- ^ Jump up to:a b c d e f “Our Formulary Infectious Diseases Laboratories CDC”. http://www.cdc.gov. 22 September 2016. Archived from the original on 16 December 2016. Retrieved 7 December 2016.
- ^ Jump up to:a b c d e f g h “Melarsoprol Drug Information, Professional”. http://www.drugs.com. 20 December 1994. Archived from the original on 30 December 2016. Retrieved 7 December 2016.
- ^ Jump up to:a b c d e f “WHO Model Prescribing Information: Drugs Used in Parasitic Diseases – Second Edition: Protozoa: African trypanosomiasis: Melarsoprol”. WHO. 1995. Archived from the original on 2016-11-10. Retrieved 2016-11-09.
- ^ Jump up to:a b “Trypanosomiasis, human African (sleeping sickness)”. World Health Organization. February 2016. Archived from the original on 4 December 2016. Retrieved 7 December 2016.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ Jump up to:a b c d CDC (2013). “Disease Control and Prevention: Parasites – African Trypanosomiasis”. Archived from the original on 2017-06-19.
- ^ Jump up to:a b “Trypanosoma brucei gambiense and Trypanosoma brucei rhodesiense (African Trypanosomiasis) – Infectious Disease and Antimicrobial Agents”. http://www.antimicrobe.org. Archived from the original on 2016-11-28. Retrieved 2016-11-17.
- ^ Bisser S, N’Siesi FX, Lejon V, et al. (2007). “Equivalence trial of melarsoprol and nifurtimox monotherapy and combination therapy for the treatment of second-stage Trypanosoma brucei rhodesiense sleeping sickness”. J. Infect. Dis. 195 (3): 322–9. doi:10.1086/510534. PMID 17205469.
- ^ Farrar J (2014). “Manson’s Tropical Diseases: Expert Consult-Online”. 23: 616.
- ^ Kioy, D.; Jannin, J.; Mattock, N. (March 2004). “Human African trypanosomiasis”. Nature Reviews Microbiology. 2 (3): 186–187. doi:10.1038/nrmicro848. PMID 15751187. S2CID 36525641.
- ^ Jump up to:a b c Brunton L (2011). “Goodman & Gillman’s The Pharmacological Basis of Therapeutics”. McGraw Hill Medical: 1427–28.
- ^ Brun, Reto; Schumacher, Reto; Schmid, Cecile; Kunz, Christina; Burri, Christian (November 2001). “The phenomenon of treatment failures in Human African Trypanosomiasis”. Tropical Medicine and International Health. 6 (11): 906–914. doi:10.1046/j.1365-3156.2001.00775.x. PMID 11703845. S2CID 21542129.
- ^ Keiser J.; Ericsson O; Burri C (2000). “Investigations of the metabolites of the trypanocidal drug melarsoprol”. Clinical Pharmacology. 67 (5): 478–88. doi:10.1067/mcp.2000.105990. PMID 10824626. S2CID 24326873.
- ^ Hollingham R (2005). “Curing diseases modern medicine has left behind”. New Scientist. 2005 (2482): 40–41. Archived from the original on 2015-05-11.
- ^ “MELARSOPROL injectable – Essential drugs”. medicalguidelines.msf.org. Retrieved 6 December 2019.
- ^ Holtz, Andrew (2006). The Medical Science of House, M.D.Penguin. p. 272. ISBN 1440628734. Retrieved 25 March 2020.
External links
- “Melarsoprol”. Drug Information Portal. U.S. National Library of Medicine.
| Clinical data | |
|---|---|
| Trade names | Arsobal[1] |
| Other names | Mel B, Melarsen Oxide-BAL[2] |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Routes of administration |
IV |
| ATC code | P01CD01 (WHO) QP51AD04 (WHO) |
| Pharmacokinetic data | |
| Elimination half-life | 35 hours |
| Excretion | Kidney |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 494-79-1 |
| PubChem CID | 10311 |
| ChemSpider | 9889 |
| UNII | ZF3786Q2E8 |
| KEGG | D00832 |
| ChEMBL | ChEMBL166 |
| CompTox Dashboard (EPA) | DTXSID90862033 |
| ECHA InfoCard | 100.007.086 |
| Chemical and physical data | |
| Formula | C12H15AsN6OS2 |
| Molar mass | 398.33 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI | |
| (what is this?) (verify) |
/////////Melarsoprol
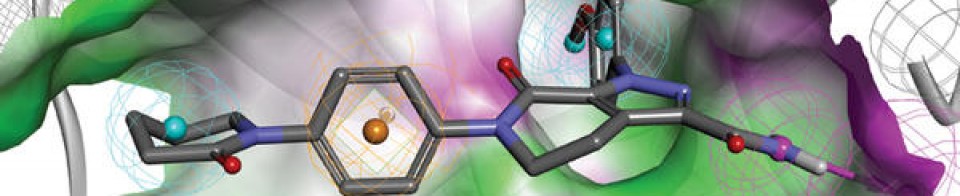
NEW DRUG APPROVALS
ONE TIME
$10.00
Rilzabrutinib
![(R)-2-(3-(4-Amino-3-(2-fluoro-4-phenoxyphenyl)-1H-pyrazolo[3,4-d]-pyrimidin-1-yl)piperidine-1-carbonyl)-4-methyl-4-(4-(oxetan-3-yl)piperazin-1-yl)pent-2-enenitrile.png](https://pubchem.ncbi.nlm.nih.gov/image/imgsrv.fcgi?cid=118325989&t=l)

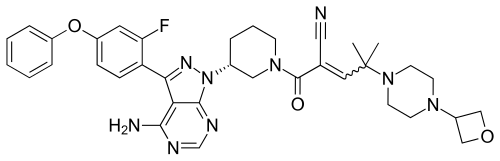
PRN 1008, Rilzabrutinib
CAS 1575591-66-0
| リルザブルチニブ; |
C36H40FN9O3,
| MW 665.7597 |
2-[(3R)-3-[4-amino-3-(2-fluoro-4-phenoxyphenyl)pyrazolo[3,4-d]pyrimidin-1-yl]piperidine-1-carbonyl]-4-methyl-4-[4-(oxetan-3-yl)piperazin-1-yl]pent-2-enenitrile
Anti-inflammatory disease, Autoimmune disease treatment
Fda 2025, approvals 2025 8/29/2025, Wayrilz, To treat persistent or chronic immune thrombocytopenia that has not sufficiently responded to immunoglobulins, anti-D therapy, or corticosteroids
- OriginatorPrincipia Biopharma
- Class2 ring heterocyclic compounds; Amines; Anti-inflammatories; Fluorobenzenes; Nitriles; Phenyl ethers; Piperazines; Piperidines; Pyrazoles; Pyrimidines; Skin disorder therapies; Small molecules
- Mechanism of ActionAgammaglobulinaemia tyrosine kinase inhibitors
- Orphan Drug StatusYes – Idiopathic thrombocytopenic purpura; Pemphigus vulgaris
- Phase IIIIdiopathic thrombocytopenic purpura; Pemphigus vulgaris
- Phase IIAutoimmune disorders
- 02 Jun 2021Efficacy data from a phase IIa trial in Ankylosing spondylitis presented at the 22nd Annual Congress of the European League Against Rheumatism (EULAR-2021)
- 07 Apr 2021Sanofi initiates enrollment in a phase I pharmacokinetics trial in healthy volunteers in Australia (PO, Tablet, Capsule) (NCT04748926)
- 31 Mar 2021Sanofi announces intention to seek regulatory approval for Idiopathic thrombocytopenic purpura in 2023 (Sanofi pipeline, May 2021)
Rilzabrutinib, sold under the brand name Wayrilz, is an anti-cancer medication used for the treatment of immune thrombocytopenia.[1] Rilzabrutinib is a tyrosine kinase inhibitor.[1] It is taken by mouth.[1]
Rilzabrutinib may increase the risk of serious infections (including bacterial, viral, or fungal).[2] The most common side effects include diarrhea, nausea, headache, abdominal pain, and COVID-19.[2]
Rilzabrutinib was approved for medical use in the United States in August 2025.[2]
CLIP
Sanofi to acquire BTK inhibitor firm Principia for $3.7 billion
Principia is testing its small-molecule compounds in multiple sclerosis and immune system diseases
Sanofi will pay $3.7 billion to acquire Principia Biopharma, a San Francisco-based biotech firm developing small molecules that inhibit Bruton tyrosine kinase (BTK). The price represents about a 75% premium over Principia’s stock market value in early July, before reports surfaced that Sanofi was interested in buying the firm.
BTK is a protein important for both normal B cell development and the proliferation of lymphomas, which are B cell cancers. AbbVie, AstraZeneca, and BeiGene all market BTK inhibitors for treating specific kinds of lymphomas. Sales of AbbVie’s inhibitor, Imbruvica, approached $4.7 billion in 2019.
Other drug firms have been eager to get in on the action as well. In January, Merck & Co. spent $2.7 billion to acquire ArQule, whose experimental noncovalent BTK inhibitor is designed to overcome resistance that some cancers develop after treatment with current covalent BTK inhibitors. Eli Lilly and Company’s $8 billion acquisition of Loxo Oncology in 2019 also included a noncovalent BTK inhibitor.
BTK is also linked to inflammation, and Principia focuses on developing BTK inhibitors for immune system diseases and multiple sclerosis. Its compound rilzabrutinib is currently in clinical trials for pemphigus and immune thrombocytopenia. In 2017, Sanofi struck a deal to develop Principia’s brain-penetrant BTK inhibitor, SAR442168, for multiple sclerosis.
Sanofi announced in April of this year that the inhibitor reduced formation of new lesions—the scarred nervous tissue that gives multiple sclerosis its name—by 85% in a Phase II clinical trial. A Phase III trial of the compound began in June.
Upon announcing its deal to acquire Principia, Sanofi said that both rilzabrutinib and SAR442168 have the potential to become a “pipeline in a product,” indicating they can be used for many immune-related and neurological diseases, respectively.
The anti-inflammatory effects of BTK inhibitors have raised interest in the drugs as treatments for people hospitalized with COVID-19. Notably, the US National Cancer Institute conducted a small study suggesting acalabrutinib may help reduce the respiratory distress and inflammation in people with COVID-19. Based on that preliminary study, AstraZeneca—which markets acalabrutinib as Calquence—is conducting a 60-person randomized trial of the drug for COVID-19.
Sanofi has not indicated interest in investigating Principia’s BTK inhibitors as COVID-19 treatments.Chemical & Engineering NewsISSN 0009-2347
PATENT
WO 2021127231https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2021127231&tab=PCTDESCRIPTION&_cid=P20-KRA0I9-18818-1
SOLID FORMS OF 2-[3-[4-AMTNO-3-(2-FT,TTORO-4-PHENOXY- PHEN¥L)PYRAZOLO[3,4 D]PYRIMIDIN l~YL]PIPERIDINE~l~CARBON¥L] 4~
METHYL-4-[4-(OXETAN-3-YL)PIPERAZIN-l-YLjPENT-2-ENENITRILE
[11 This application claims the benefit of priority to U.S. Provisional Application
No 62/951,958, filed December 20, 2019, and U.S Provisional Application No. 63/122,309, filed December 7, 2020, the contents of each of which are incorporated by reference herein in their entirety.
[2] Disclosed herein are solid forms of 2-[3-[4~amino-3~(2~fluoro-4-phenoxy-plienyl)pyrazolo[3,4-d]pyrimidin-l-yl]piperidine-l Carbonyl]~4-nietliyl-4~[4-(oxetaii~3-yl)piperazin-!~yi]pent-2~enenitriie (Compound (I)), methods of using the same, and processes for making Compound (I), including its solid forms. The solid forms of Compound (I) may be inhibitors of Bruton’s tyrosine kinase (BTK) comprising low residual solvent content.
[3| The enzyme BTK is a member of the Tec family non-receptor tyrosine kinases.
BTK is expressed in most hematopoietic cells, including B cells, mast cells, and macrophages BTK plays a role in the development and activation of B cells. BTK activity has been implicated in the pathogenesis of several disorders and conditions, such as B cell-related hematological cancers (e.g., non-Hodgkin lymphoma and B cell chronic lymphocytic leukemia) and autoimmune diseases (e.g., rheumatoid arthritis, Sjogren’s syndrome, pemphigus, IBD, lupus, and asthma).
[4] Compound (I), pharmaceutically acceptable salts thereof, and solid forms of any of the foregoing may inhibit BTK and be useful in the treatment of disorders and conditions mediated by BTK activity. Compound (I) is disclosed in Example 31 of WO 2014/039899 and has the following structure:
where *C is a stereochemical center. An alternative procedure for producing Compound (!) is described in Example 1 of WO 2015/127310.
[5] Compound (I) obtained by the procedures described in WO 2014/039899 and WO 2015/127310 comprises residual solvent levels well above the limits described in the International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use (“ICH”) guidelines. In general, manufacturing processes producing residual solvent levels near or above the ICH limits are not desirable for preparing active pharmaceutical ingredients (APIs).
Example 1: Spray Drying Process A
[311] A solution of Compound (I) in dichloromethane (prepared according to Example 31 on pages 86-87 of WO 2014/039899) was washed with pH 3 phosphate buffer to remove basic impurities that are more soluble than Compound (I) in the aqueous layer. The dichloromethane solution was then washed with pH 7 buffer and solvent exchanged into isopropyl acetate. The isopropyl acetate solution was then washed with pH 3 phosphate buffer, bringing Compound (I) into the aqueous layer and removing non-basic impurities. The pH of the aqueous layer was adjusted to pH 9 with 10% sodium hydroxide, and the aqueous layer was extracted with isopropyl acetate. Upon concentration under vacuum, Compound (I) was precipitated from heptane at 0 °C, filtered and dried to give a white amorphous solid as a mixture of the (E) and (Z) isomers, as wet Compound (I). Wet Compound (I) was dissolved in methanol and spray dried at dryer inlet temperature of 125 °C to 155 °C and dryer outlet temperature of 48 to 58 °C to obtain the stable amorphous Compound (I) free base with levels of isopropyl acetate and heptane below 0.5% and 0.05%, respectively.
Example 2: Spray Drying Process B
intermediate A
Compound (!)
[241] A jacketed reactor with overhead stirrer, condenser, nitrogen line, temperature probe, and recirculating fluid chiller/heater was charged with Intermediate A (20.2 kg) and Intermediate B (13.6 kg, 1.5 equiv). DCM (361.3 kg, 14.5 vol) was charged to the reactor. The mixture was agitated, and the batch cooled to 0 °C to 5 °C. The reactor was charged with pyrrolidine (18.3 kg, 6 equiv) and then charged with TMSC1 (18.6 kg, 4 eq). Stirring was continued at 0 °C to 5 °C for 0.5 to 1 hour
[242] At 0 °C to 5 °C, acetic acid (2.0 equiv) was charged to the reactor followed by water (5 equiv). Stirring was continued at 0 °C to 5 °C for 1 to 1.5 hours. Water (10 equiv) was charged to the reactor, and the solution was adjusted to 20 °C to 25 °C. The internal temperature was adjusted to 20 °C to 25 °C and the biphasic mixture was stirred for 15 to 20 mins. Stirring was stopped and phases allowed to separate for at least 0.5 h. The lower aqueous layer was removed.
[243] Water (7 vol) was charged to the reactor. The pH was adjusted to 2.8-3.3 with a 10 wt. % solution of citric acid. Stirring was continued at 0 to 5 °C for 1 to 1.5 hours. Stirring was stopped and phases allowed to separate for at least 0.5 h. The lower aqueous layer was removed.
[244] A jacketed reactor with overhead stirrer, condenser, nitrogen line, temperature probe, and recirculating fluid chiller/heater was charged with an approximately 9% solution of NaHCCri (1 vol) and the organic layer. The internal temperature was adjusted to 20 °C to 25 °C, and the biphasic mixture was stirred for 15 to 20 mins. Stirring was stopped and phases allowed to separate for at least 0.5 h. The lower aqueous layer was removed. The aqueous layer was measured to have a pH greater than 7.
[245] A jacketed reactor with overhead stirrer, condenser, nitrogen line, temperature probe and recirculating fluid chiller/heater was charged with the organic layer. The organic phase ¾s distilled under vacuum at less than 25 °C to 4 total volumes. IP AC (15 vol) was charged to the reactor. The organic phase was distilled under vacuum at less than 25 °C to 10 total volumes. Water (15 vol) followed by pH 2.3 phosphate buffer were charged to the reactor at an internal temperature of 20 °C to 25 °C. The pH adjusted to 3 Stirring was stopped and phases allowed to separate for at least 0.5 h. The organic phase was removed.
[246] The following steps were repeated twice: IP AC (5 vol) was charged to the reactor containing the aqueous layer. Stirring was continued for 0.25 to 0.5 hours. Stirring was stopped and phases allowed to separate for at least 0.5 h. The organic phase was removed. [247] IP AC (15 vol) was charged to the reactor containing the aqueous layer. A pH 10 phosphate buffer was charged to the reactor and the pH adjusted to 10 with 14% NaOH solution. Stirring was continued for 1.5 to 2 hours. Stirring was stopped and phases allowed to separate for at. least 0.5 h. The aqueous layer was discarded. The organic layer was dried over brine.
[248] The organic solution was distilled under vacuum at less than 25 °C to 5 total volumes.
[249] A jacketed reactor with overhead stirrer, condenser, nitrogen line, temperature probe and recirculating fluid chiller/heater was charged with n-heptane (20 vol). The internal temperature was adjusted to 0 to 5 °C, and the IP AC solution was added.
[250] The suspension was filtered. The filter cake was washed with n-heptane and the tray was dried at 35 °C. Compound (I) (24.6 kg) was isolated in 86% yield.
[251] Compound (1) was dissolved in methanol (6 kg) and spray dried to remove residual IP AC and n-heptane.
Example 3: Precipitation Process A
[252] A solution of Compound (I) in dichloromethane (prepared according to Example 31 on pages 86-87 of WO 2014/039899) was quenched with acetic acid and water, followed by washing with pH 3 aqueous solution to remove basic impurities that are more soluble than Compound (1) in the aqueous layer. Washing was repeated as needed to reduce impurities. Methanesulfonic acid was added to the dichloromethane solution, and the dichloromethane solution was concentrated by distillation under reduced pressure, followed by addition of 1% NaCi aqueous solution and isopropyl acetate before adjustment of pH to approximately 3 with potassium hydroxide. The isopropyl acetate layer was removed and discarded. The aqueous layer containing Compound (I) was washed with isopropyl acetate to remove hydrophobic impurities. Washing was repeated as needed to reduce related substance impurities. Residual isopropyl acetate was removed by distillation under reduced pressure. The aqueous solution containing Compound (I) was cooled to 0 to 5°C before adjusting the pH to approximately 9 with potassium hydroxide. The free base of Compound (I) was allowed to precipitate and maturate at 20 °C for 20 hours. The mixture temperature was then adjusted to 20 °C to 25 °C, and the hydrate impurity was verified to be less than 0.3% (< 0.3%). The cake of the free base of Compound (I) was filtered and washed as needed to reduce conductivity. The cake was then allowed to dry on the filter under vacuum and nitrogen swept to reduce water content by Karl-Fischer (KF < 50%) before transferring to the oven for drying. The wet cake of the free base of Compound (1) was dried under vacuum at 25 °C until water content by Karl -Fischer was less than 1.5% (KF < 1.5%), and then dehmiped by milling to yield a uniform white amorphous solid as a mixture of the (E) and (Z) isomers, with no detectible levels of isopropyl acetate or heptane.
Example 4: Precipitation Process 3B
[253] A solution of Compound (I) in dichloromethane (prepared according to Example 31 on pages 86-87 of WO 2014/039899) was quenched with acetic acid and water, followed by washing with pH 3 aqueous solution to remove basic impurities that are more soluble than Compound (I) in the aqueous layer. The washing was repeated as needed to reduce residual solvents and impurities. The dichloromethane solution was then washed with saturated sodium bicarbonate (pH > 7). Dichloromethane was removed by distillation under reduced pressure, followed by addition of water and isopropyl acetate. The pH of the aqueous layer was adjusted to pH to 2.8 – 3.3 with 2 M aqueous sulfuric acid (H2SQ4) at 0 – 5 °C, and the mixture rvas stirred and settled. After phase separation removal of the organic layer, the aqueous layer was washed with isopropyl acetate three times and the residual isopropyl acetate in aqueous layer was distilled out under vacuum at a temperature below 25 °C and the solution was basitied with 5% aqueous KOFI to pH 9 – 10 to a slurry . The resulting suspension was stirred and warmed up to 20 °C to 25 °C and aged for 20 h. The product was filtered and washed with water and dried to give white solid in 86% yield.
Example 5: Precipitation Process C
[254] A solution of Compound (I) in dichloromethane (prepared according to Example 31 on pages 86-87 of WO 2014/039899) was quenched with acetic acid and water, followed by washing to remove basic impurities that are more soluble than Compound (I) in the aqueous layer. Washing was repeated as needed to reduce impurities. Methanesulfonic acid was added to the d chloromethane solution, and the dichloromethane solution was concentrated under reduced pressure to obtain a thin oil. The concentrated oil was cooled to approximately 5°C before washing with an aqueous solution of sodium chloride. The organic phase was discarded. Washing of the aqueous layer was repeated as needed with dichloromethane to remove low level impurities. The pH of the aqueous solution was adjusted to approximately 3 with an aqueous solution of potassium hydroxide. Residual dichloromethane was removed
under reduced pressure. The level of residual acetic acid was determined by, for example, titration. The aqueous solution containing Compound (I) was cooled to a temperature between 0°C and 5°C. Acetic acid was present at 0 wt % to 8 wt. %. Acetic acid level was 0 wt % if the aqueous acid solution was washed with aqueous sodium bicarbonate or another aqueous inorganic base. Optionally, additional acetic acid was added to achieve a 0 wt.% to 8 wt. % acetic acid level. An aqueous solution of potassium hydroxide was constantly charged to the aqueous solution to obtain a pH to approximately 9.5. The free base of Compound (I) was allowed to precipitate and maturate at approximately 20 °C for least 3 hours. The cake (wet solid) of the free base of Compound (I) was filtered and washed with water. The wet cake was then dried under reduced vacuum with slight heat. Alternatively, instead of washing the wet cake with water, the wet cake was reslurried with water at approximately 15 °C for at least 1 hour before filtering. The free base of Compound (I) in the fomi of a wet cake was dried under vacuum with slight heat at 25°C.
[255] FIGs. 12-15 are example SEM images showing the variable morphologies of particles of Compound (I) during the filtration step to isolate Compound (I) based on the amount acetic acid added during the initial step in the precipitation of Compound (Ϊ) (FIG. 12: at 0 wt. % acetic acid; FIG 13: at 3 wt. % acetic acid; FIG. 14: at 5 wt. % acetic acid; FIG 15: at 8 wt. % acetic acid). Filtration speed depended on the morphology and was the fastest for 0 wt. % acetic acid. At 1 wt. % acetic acid, the filtration speed diminished considerably, improving at 2 wt. % to 3 wt. % acetic acid. Morphologies with more open holes (such as, e.g., more porous particles) resulted in improved filtration speeds, whereas more compact particles resulted in decreased filtration speed.
Example 6: Conversion of a Crystalline Form of Compound (Ϊ) to an Amorphous Form
[256] 9.8 grams of a crystalline form of Compound (I) were dissolved in approximately 20 mL of dichloromethane and approximately 120 ml. of brine solution. Then, approximately 1 equivalent of methanesulfonic acid was added. The pH w¾s approximately 2. The layers were separated. The aqueous layer was concentrated at a temperature between 0°C and 5°C to remove residual dichloromethane before slowly adding aqueous KOI I solution (approximately 5%) to adjust the pH to a value between 9 and 10. During aqueous KOH addition, an amorphous form of Compound (I) precipitated out. The slurry was slowly warmed to room temperature and then was stirred for approximately 24 hours before filtering and rinsing the wet cake with water. The wet cake was dried under vacuum with slight heat at approximately 30°C to provide 7 grams of a white to an off-white solid (87% yield and 98 4% purity). XRPD showed that the product was an amorphous solid form of Compound (I).
Example 7: Micronization of Compound (I) Particles Obtained by Precipitation Processes
[257] A fluid jet mill equipment was used during lab scale jet milling trials. The fluid jet mill equipment includes a flat cylindrical chamber with 1.5” diameter, fitted with four symmetric jet nozzles winch are tangentially positioned in the inner wall. Prior to feeding material to the fluid jet mill in each trial, the material was sieved in a 355 iim screen to remove any agglomerates and avoid blocking of the nozzles during the feed of material to the micronization chamber. The material to be processed was drawn into the grinding chamber through a vacuum created by the venturi (P vent ~ 0 5 – 1 0 bar above P grind). The feed flow rate of solids (F_feed) was controlled by a manual valve and an infinite screw volumetric feeder. Compressed nitrogen was used to inject the feed material; compressed nitrogen was also used for the jet nozzles in the walls of the milling chamber. Compressed fluid issuing from the nozzles expands from P grind and imparts very’ high rotational speeds in the chamber. Accordingly, material is accelerated by rotating and expanding gases and subjected to centrifugal forces. Particles move outward and are impacted by high velocity jets, directing the particles radially inward at very high speeds. Rapidly moving particles impact the slower moving path of particles circulating near the periphery of the chamber. Attrition takes place due to the violent impacts of particles against each other. Particles with reduced size resulting from this sequence of impacts are entrained in the circulating stream of gas and swept against the action of centrifugal force toward the outlet at the center. Larger particles in the gas stream are subjected to a centrifugal force and returned to the grinding zone. Fine particles are carried by the exhaust gas to the outlet and pass from the grinding chamber into a collector.
[258] The feeder has continuous feed rate control; however, to more precisely control the feed rate, the full scale of feed rates was arbitrary divided in 10 positions. To calibrate F feed, the feeder was disconnected from milling chamber and 10 g of Compound (I) powder was fed through the feeder operating at various feed rate positions. The mass of powder flowing through the feeder over 6 minutes was marked. The resulting feed rate was directly proportional to feeder position. After processing each of the four trials, the jet mill was stopped, micronized product removed from the container, and the milling chamber checked for any powder accumulation.
Variables/Parameters
F_feed Feed flow rate of solids [kg/h]
P grind Grinding pressure inside the
drying chamber [bar]
P vent Feed pressure in the venturi [bar]
Example 8: Residual Solvent Levels
[251] Retention of process solvents (/.<?., res dual solvents) depends on van der Waal s’ forces that are unique to and an inherent property of each molecule. Additionally, solvent retention depends how the API solid is formed, isolated, washed, and dried (i.e., during the manufacturing process). Because residual solvents may pose safety risks, pharmaceutical processes should be designed to minimize residual solvent levels (e.g , to result in residual solvent levels below the limits established in the ICH guidelines).
[252] Residual solvent analysis was performed using gas chromatography-mass spectrometry. The residual solvent levels in solid forms of Compound (I) prepared by spray drying processes described herein and precipitation processes described herein are provided in Table 2. The residual solvent levels in crude Compound (I) listed in Table 2 are comparable to the residual solvent levels in crude Compound (I) prepared according to the procedures detailed in Example 31 of WO 2014/039899 and Example 1 of WO 2015/127310.
Table 2: Residual solvent levels in solid forms of Compound (I)
PATENT
WO 2015127310
https://patents.google.com/patent/WO2015127310A1/enExample 1Synthesis of 2-[(3R)-3-[4-amino-3-(2-fluoro-4-phenoxy-phenyl)pyrazolo[3,4-d]pyrimidin-l- yl]-piperidine-l-carbonyl]-4-m iperazin-l-yl]pent-2-enenitrile

Step 1To a solution of 3-[(3R)-3-[4-amino-3-(2-fluoro-4-phenoxy-phenyl)pyrazolo[3,4- d]pyrimidin-l -yl]-l-piperidyl]-3-oxo-propanenitrile (15 g, 3.12mmol), 2-methyl-2-[4- (oxetan-3-yl)piperazin-l-yl]propanal (794.25mg, 3.74mmol) in DCM (40mL), pyrrolidine (1.54mL,18.71mmol) at 0-5 °C was added, which is followed by TMS-Cl (1.58mL,12.47mmol). The reaction mixture was stirred at 0-5 °C for 3 h and was quenched with 1 M potassium phosphate buffer (pH 3). Layers were separated and the organic layer was washed once more with 1 M potassium phosphate buffer (pH 3). The organic layer was extracted withl M potassium Phosphate buffer at pH 1.5. Layers were separated. The aqueous phase contained the desired product while the impurities stayed in the organic phase. The aqueous phase was neutralized with 1 M potassium phosphate (pH 7) and was extracted with isopropylacetate (10 volumes). Upon concentration 2-[(3R)-3-[4-amino-3-(2-fluoro-4- phenoxy-phenyl)pyrazolo[3,4-d]pyrimidin-l-yl]piperidine-l-carbonyl]-4-methyl-4-[4-(oxetan-3-yl)piperazin-l-yl]pent-2-enenitrile was obtained as a foam having >99% HPLC purity. MS (pos. ion) m/z: 666 (M+l ).The foam containing high levels of residual solvent was dissolved in 2 M HC1 and the resulting solution was placed under vacuum to remove residual organic solvents. pH of the solution was then adjusted to ~ 7 and the resulting paste was filtered and dried in vacuum without heat. This resulted in isolation of 2-[(3R)-3-[4-amino-3-(2-fluoro-4-phenoxy- phenyl)pyrazolo[3,4-d]pyrimidin-l-yl]piperidine-l-carbonyl]-4-methyl-4-[4-(oxetan-3- yl)piperazin- l-yl]pent-2-enenitrile containing residual water up to 10%. Drying under vacuum without heat reduces the water level but lead to generation of impurities.Step 1AAlternatively, the isopropylacetate solution of 2-[(3R)-3-[4-amino-3-(2-fluoro-4- phenoxy-phenyl)pyrazolo[3,4-d]pyrimidin- 1 -yl]piperidine- 1 -carbonyl]-4-methyl-4-[4- (oxetan-3-yl)piperazin-l -yl]pent-2-enenitrile can be concentrated to 4 vol and added to heptane (20 volume) at 0 °C. The resulting suspension was stirred at 0 °C overnight and the product was filtered, washed twice with heptane and dried at 45 °C for 2 days under vacuum to give 2-[(3R)-3-[4-amino-3-(2-fluoro-4-phenoxy-phenyl)pyrazolo[3,4-d]pyrimidin-l – yl]piperidine-l-carbonyl]-4-methyl-4-[4-(oxetan-3-yl)piperazin-l-yl]pent-2-enenitrile in 85 – 90 % yield as a free flowing solid. However, the solids obtained by this method contained high residual solvents (3.9 wt% isopropylacetate and 1.7 wt% heptane). In addition, the free base form was not very stable as degradation products were observed during the drying process at less than 45 °C.Salt formationExample 2Preparation of 2-[(3R)-3-[4-amino-3-(2-fluoro-4-phenoxy-phenyl)-pyrazolo[3,4-d]pyrimidin- l-yl]-piperidine-l-carbonyl]-4-methyl-4-[4-(oxetan-3-yl)-piperazin-l-yl]pent-2-enenitrile hemisulfate and sulfate saltHemisulfate: To the solution of 2-[(3R)-3-[4-amino-3-(2-fluoro-4-phenoxy-phenyl)-pyrazolo[3,4- d]pyrimidin-l-yl]-piperidine -carbonyl]-4-methyl-4-[4-(oxetan-3-yl)-piperazin-l-yl]pent-2- enenitrile (4.2 g) in EtOAc (60 mL, 15 vol) was added sulfuric acid (0.31 g, 0.17 mL, 0.5 eq) in EtOAc (20 mL, 5 vol) at ambient temperature. The suspension was stirred at ambient temperature for ~ 2 hr and then 40 °C for 4 hr and then at ambient temperature for at least 1 hr. After filtration and drying at ambient temperature under vacuum, 1.5 g of white powder was obtained. Solubility of the hemi-sulfate at ambient temperature was > 100 mg/mL in water.Sulfate saltTo the solution of 2-[(3R)-3-[4-amino-3-(2-fluoro-4-phenoxy-phenyl)-pyrazolo[3,4- d]pyrimidin-l-yl]-piperidine-l-carbonyl]-4-methyl-4-[4-(oxetan-3-yl)-piperazin-l-yl]pent-2- enenitrile (810 mg) in EtOAc (8 mL, 10 vol) was added sulfuric acid (0.06 mL, 1.0 equiv.) in EtOAc (2.5 mL, 5 vol) at ambient temperature. The resulting suspension was stirred at 40 °C for 2 hr and then cooled to ambient temperature for at least 1 hr. After filtration, solids were dried by suction under Argon for 1 h to give a white powder (0.68 g) in 69% yield.
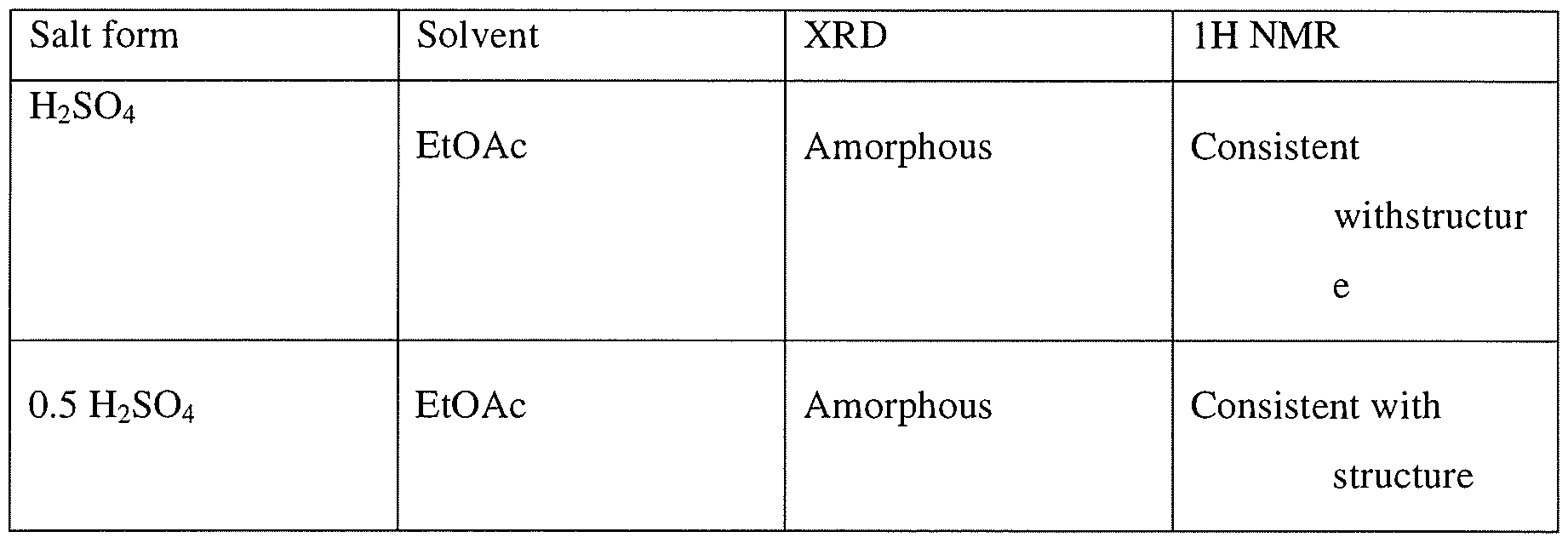
Example 3Preparation of 2-[(3R)-3-[4-amino-3-(2-fluoro-4-phenoxy-phenyl)-pyrazolo[3,4- d]pyrimidin- 1 -yl]-piperidine- 1 -carbonyl] -4-methyl-4-[4-(oxetan-3-yl)-piperazin- 1 -yl]pent-2- enenitrile hydrochlorideTo a solution of 2-[(3R)-3-[4-amino-3-(2-fluoro-4-phenoxy-phenyl)pyrazolo[3,4- d]pyrimidin- 1 -yl]piperidine- 1 -carbonyl]-4-methyl-4-[4-(oxetan-3-yl)piperazin- 1 -yl]pent-2- enenitrile (100 mg, 0.15 mmol) in CH2CI2 (1ml) at ambient temperature was added 2 equivalent of HC1 (0.3 mmol, 0.15 ml of 2M HC1 in 1 : 1 dioaxane:CH2Cl2). The resulting homogeneous solution was stirred at ambient temperature for 1 h and was added dropwise to 15 volumes of ethylacetate (as compared to CH2C12) resulting in formation of a white solid. The mixtures was aged at ambient temperature for lh and placed at 2-8 C for 19 h. Upon filtration and washing of the filter cake with ethylacetate and drying a white solid was obtained. Analysis by XRPD indicated formation of an amorphous solid. Both Ή-NMR and IC analysis indicated formation of the salt. IC indicated formation mono-HCl salt.

Example 4General procedure for preparation of 2-[(3R)-3-[4-amino-3-(2-fluoro-4-phenoxy- phenyl)pyrazolo[3,4-d]pyrimidin-l-yl]-piperidine-l-carbonyl]-4-methyl-4-[4-(oxetan-3-yl)- piperazin-l-yl]pent-2-enenitrile mono- and di-mesylate saltsTo a solution of 2-[(3R)-3-[4-amino-3-(2-fluoro-4-phenoxy-phenyl)pyrazolo[3,4- d]pyrimidin-l-yl]piperidine-l-carbonyl]-4-methyl-4-[4-(oxetan-3-yl)piperazin-l-yl]pent-2- enenitrile (100 mg, 0.15 mmol) in CH2C12 (1 ml) at ambient temperature was added either 1 equivalent of methanesulfonic acid (0.15 mmol, 0.2 ml of 74 mg/ml solution in CH2C12) or 2 equivalent of methanesulfonic acid (0.3 mmol, 0.4 ml of 74 mg/ml solution in CH2C12). The resulting homogeneous solution was stirred at ambient temperature for 1 h and was added dropwise to 10 volumes of antisolvents (ethylacetate, methyl tert-butylether (MTBE), or cyclohexane) (10 ml as compared to CH2C12) resulting in formation of a white solid. The mixture was aged at ambient temperature for lh and placed at 2-8 °C for 19 h. Upon filtration and washing of the filter cake with the antisolvent and drying, a white solid was obtained. Analysis by XRPD indicated formation of an amorphous solid. Both Ή-NMR and IC analysis indicated formation of the salt as well as counterion ratio.Alternatively 2-[(3R)-3-[4-amino-3-(2-fluoro-4-phenoxy-phenyl)pyrazolo[3,4-d]- pyrimidin- 1 -yl]piperidine- 1 -carbonyl]-4-methyl-4-[4-(oxetan-3-yl)piperazin- 1 -yl]pent-2- enenitrile can be dissolved in 4 volumes of isopropylacetate and added to 2 equivalent of methanesulfonic acid in 6 volumes of isopropylacetate at 0 °C to generate the dimesylate salt.

1. Theoretical mesylate content, monomesylate=12.6% and dimesylate=22.4%, NO- not determinedExample 5 General procedure for the preparation of carboxylate salt Approximately 20 mg of the compound (I) was dissolved in minimum amount of the allocated solvent system. These were then mixed with the appropriate number of equivalents of counterion dissolved or slurried in the allocated solvent.If compound (I) was insoluble in the selected solvent, slurry of the sample was used after adding 300 μί.If the acid was insoluble in the selected solvent, slurry of the acid was used after adding 300 xL.If the acid was a liquid, the acid was added to the dissolved/slurried compound (I) from a stock solution in the allocated solvent.The suspensions/ precipitates resulting from the mixtures of compound (I) were temperature cycled between ambient (ca. 22°C) and 40°C in 4 hour cycles for ca. 48 hrs (the cooling/heating rate after each 4 hour period was ca. 1 °C/min). The mixtures were visually checked and any solids present were isolated and allowed to dry at ambient conditions prior to analysis. Where no solid was present, samples were allowed to evaporate at ambient. Samples which produced amorphous material, after the treatment outlined above, were re- dissolved and precipitated using anti-solvent (ter/-butylmethylether) addition methods at ambient conditions (ca. 22°C). i.e. the selected anti-solvent was added to each solution, until no further precipitation could be observed visually or until no more anti-solvent could be added. The solvents used in this preparation were acetonitrile, acetone, isopropyl acetate, THF and MTBE. The acid used were oxalic acid, L-aspartic acid, maleic acid, malonic acid, L-tartaric acid, and fumaric acid.Example 6General procedure for preparation of 2-[(3R)-3-[4-amino-3-(2-fluoro-4-phenoxy- phenyl)pyrazolo[3,4-d]pyrimidin-l-yl]-piperidine-l-carbonyl]-4-methyl-4-[4-(oxetan-3-yl)- piperazin-l-yl]pent-2-enenitrile hemicitrate saltTo a solution 2-[(3R)-3-[4-amino-3-(2-fluoro-4-phenoxy-phenyl)pyrazolo[3,4-d]- pyrimidin- 1 -yl]piperidine- 1 -carbonyl]-4-methyl-4-[4-(oxetan-3-yl)piperazin- 1 -yl]pent-2- enenitrile (5 g, 7.5 mmol) in ethanol (50 ml) was added citric acid (720.5 mg, 3.76 mmol) dissolved in 2 ml of water. Mixture was stirred at ambient temperature for 15 min, additional 0.5 ml of water was added and the mixture was stirred for 1 h, concentrated in vacuo to a gum. Ethanol was added and the mixture was concentrated. This process was repeated twice more and then CH2CI2 was added to the mixture. Upon concentration a white solid was obtained which was tumble dried under reduced pressure at 40 C for 4 h, then in a vacuum oven for 19h to give 5.4 g of a solid. Analysis by XRD indicated formation of an amorphous solid
PATENT
WO2014039899, Example 31
Rilzabrutinib (PRN1008) is an oral, reversible covalent inhibitor of Bruton’s tyrosine kinase (BTK) [1].
https://patents.google.com/patent/WO2014039899A1/enExample 31Synthesis of (R)-2-(3-(4-amino-3-(2-fluoro-4-phenoxyphenyl)- 1 H-pyrazolo[3,4-d]pyrimidin- 1 -yl)piperidine- 1 -carbonyl)-4-methyl-4-(4-(oxetan-3-yl)piperazin- 1 -yl)pent-2-enenitrile

Step 1A solution of 2-bromo-2-methyl-propanal (696.6 mg, 4.61 mmol) in DCM (10 mL) was cooled with an ice bath and l -(oxetan-3-yl)piperazine (328 mg, 2.31 mmol), diluted with 5-10 mL of DCM, was slowly added via addition funnel over a 15 min period. Next, Hunig’s base (0.4 mL, 2.31 mmol) was added and then the cooling bath was removed. The reaction mixture was stirred at room temperature overnight and the DCM layer was washed three times with 0.5N HC1. The combined aqueous layer was neutralized with NaOH to pH 10-11 and extracted with DCM. The combined organic layer was washed with brine and dried over Na?S04. Filtration and removal of solvent afforded 2-methyl-2-[4-(oxetan-3-yl)piperazin-l- yl]propanal as a light yellow liquid, which was used directly in the next step without further purification.Step 2To a cooled (0 °C) solution of 3-[(3R)-3-[4-amino-3-(2-fluoro-4-phenoxy-phenyl)- pyrazolo[3,4-d]pyrimidin-l-yl]-l-piperidyl]-3-oxo-propanenitrile (80 mg, 0.17 mmol), was added 2-methyl-2-[4-(oxetan-3-yl)piperazin-l-yl]propanal (-108 mg, 0.51 mmol) in DCM (10 mL) followed by pyrrolidine (0.08 mL, 1.02 mmol) and TMS-C1 (0.09 raL, 0.68 mmol.) The ice bath was removed, and the reaction stirred 1 hour. Most of the solvent was removed and the residues were purified by chromatography, using 95:5 CH2Cl2:MeOH to obtain 79 mg of (R)-2-(3-(4-amino-3-(2-fluoro-4-phenoxyphenyl)-lH-pyrazolo[3,4-d]-pyrimidin-l- yl)piperidine- 1 -carbonyl)-4-methyl-4-(4-(oxetan-3-yl)piperazin- 1 -yl)pent-2-enenitrile as a white solid. MS (pos. ion) m/z: 666 (M+l).
PAPER
https://www.sciencedirect.com/science/article/abs/pii/S0223523421001781?dgcid=rss_sd_all
Therapy based on Bruton’s tyrosine kinase (BTK) inhibitors one of the major treatment options currently recommended for lymphoma patients. The first generation of BTK inhibitor, Ibrutinib, achieved remarkable progress in the treatment of B-cell malignancies, but still has problems with drug-resistance or off-target induced serious side effects. Therefore, numerous new BTK inhibitors were developed to address this unmet medical need. In parallel, the effect of BTK inhibitors against immune-related diseases has been evaluated in clinical trials. This review summarizes recent progress in the research and development of BTK inhibitors, with a focus on structural characteristics and structure-activity relationships. The structure-refinement process of representative pharmacophores as well as their effects on binding affinity, biological activity and pharmacokinetics profiles were analyzed. The advantages and disadvantages of reversible/irreversible BTK inhibitors and their potential implications were discussed to provide a reference for the rational design and development of novel potent BTK inhibitors.

Research
Rilzabrutinib is an oral, reversible covalent inhibitor of Bruton’s tyrosine kinase, that may increase platelet counts in people with immune thrombocytopenia by means of dual mechanisms of action: decreased macrophage (Fcγ receptor)–mediated platelet destruction and reduced production of pathogenic autoantibodies.[5]
References
- https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/219685s000lbl.pdf
- “FDA Approves Drug to Treat Adults with Persistent or Chronic Immune Thrombocytopenia”. U.S. Food and Drug Administration. 2 September 2025. Retrieved 5 September 2025.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - “Press Release: Sanofi’s Wayrilz approved in US as first BTK inhibitor for immune thrombocytopenia” (Press release). Sanofi. 29 August 2025. Retrieved 5 September 2025 – via GlobeNewswire.
- World Health Organization (2020). “International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 83”. WHO Drug Information. 34 (1). hdl:10665/339768.
- Kuter DJ, Efraim M, Mayer J, Trněný M, McDonald V, Bird R, et al. (April 2022). “Rilzabrutinib, an Oral BTK Inhibitor, in Immune Thrombocytopenia”. The New England Journal of Medicine. 386 (15): 1421–1431. doi:10.1056/NEJMoa2110297. PMID 35417637.
External links
- “Rilzabrutinib ( Code – C174769 )”. EVS Explore.
- Clinical trial number NCT04562766 for “Study to Evaluate Rilzabrutinib in Adults and Adolescents With Persistent or Chronic Immune Thrombocytopenia (ITP) (LUNA 3)” at ClinicalTrials.gov
| Clinical data | |
|---|---|
| Trade names | Wayrilz |
| Other names | PRN-1008 |
| AHFS/Drugs.com | Wayrilz |
| License data | US DailyMed: Rilzabrutinib |
| Routes of administration | By mouth |
| Drug class | Antineoplastic |
| ATC code | None |
| Legal status | |
| Legal status | US: ℞-only[1] |
| Identifiers | |
| IUPAC name | |
| CAS Number | 1575591-66-0 |
| PubChem CID | 73388818 |
| DrugBank | DB17709 |
| ChemSpider | 58893525 |
| UNII | NWN58M4F5T |
| KEGG | D11873 |
| ChEMBL | ChEMBL3702854 |
| Chemical and physical data | |
| Formula | C36H40FN9O3 |
| Molar mass | 665.774 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
///////////////PRN-1008, PRN 1008, Rilzabrutinib, リルザブルチニブ, Fda 2025, approvals 2025 8/29/2025, Wayrilz,
N#CC(=CC(N(C1COC1)C)(C)C)C(=O)N1CCCC1Cn1nc(c2c1ncnc2N)c1ccc(cc1F)Oc1ccccc1
PAT
PAT



AS ON JUNE2025 4.45 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
PRN 473, SAR 444727


SAR-444727
1414354-91-8C30 H30 F N7 O2 Molecular Weight539.601-Piperidinepropanenitrile, 3-[4-amino-3-(2-fluoro-4-phenoxyphenyl)-1H-pyrazolo[3,4-d]pyrimidin-1-yl]-α-(2,2-dimethylpropylidene)-β-oxo-, (3R)-
(3R)-3-[4-Amino-3-(2-fluoro-4-phenoxyphenyl)-1H-pyrazolo[3,4-d]pyrimidin-1-yl]-α-(2,2-dimethylpropylidene)-β-oxo-1-piperidinepropanenitrile
2-(3-(4-amino~3-(2-fiuoro~4-phenoxyphenyl)-1H-pyrazolo[3,4-d]pyrimidin-1-yl)piperidine-1-carbonyl)-4,4-dimethylpent-2-enenitrile
- OriginatorPrincipia Biopharma
- ClassSmall molecules
- Mechanism of ActionAgammaglobulinaemia tyrosine kinase inhibitors
- Phase IAutoimmune disorders
- DiscontinuedArthritis
- 28 Sep 2020Principia Biopharma has been acquired by Sanofi
- 22 Jun 2020Principia Biopharma plans a pharmacokinetic phase I trial (In volunteers) for Hypersensitivity (for Immunoglobulin E-mediated allergies) in Australia (Topical) (ACTRN12620000693921)
- 10 Mar 2020Phase-I clinical trials in Autoimmune disorders (In volunteers) in Australia (Topical)
- US 8957080
- US 8673925
- WO 2014022569
- WO 2013191965
- WO 2012158764
Useful for treating pemphigus vulgaris, immune thrombocytopenia, inflammatory bowel disease, Sjogren’s syndrome, multiple sclerosis, chronic lymphocytic leukemia and ankylosing spondylitis. Principia Biopharma is developing a topical formulation PRN-473 (presumed to be SAR-444727), a reversible covalent bruton’s (BTK) tyrosine kinase inhibitor, developed based on Principia’s reversible, tailored covalency platform, for treating immune-mediated diseases [phase I, July 2021]. Principia Biopharma was also investigating BTK inhibitors , developed based on Principia’s reversible, tailored covalency platform, for treating hematologic malignancies [no development reported since July 2019]. At the time of publication, Zhu was also affiliated with Nurix Therapeutics , while By and Phiasivongsa were based at Rain Therapeutics and Kronos Bio , respectively.
PATENT
WO-2021142131
Novel crystalline polymorphic forms (I to V) of PRN-473 and their preparation method.
CRYSTALLINE FORMS OF 2- [3- [4- AMINO-3-(2- FLUORO-4-PHENOXY- PHENYL)-1H-PYRAZOLO[3,4-D]PYRIMIDIN-1-YL]PIPERIDINE-1-CARBONYL]- 4,4-DIMETHYLPENT-2-ENENITRILE
This application claims the benefit of priority to U.S. Provisional Application No. 62/958,389, filed January 8, 2020, the contents of which are incorporated by reference herein in their entirety.
Disclosed herein are crystalline forms of 2-(3-(4-amino~3-(2-fiuoro~4-phenoxyphenyl)-1H-pyrazolo[3,4-d]pyrimidin-1-yl)piperidine-1-carbonyl)-4,4-dimethylpent-2-enenitrile (Compound (I)), methods of using the same, and processes for making Compound (I), including its various crystalline forms. The crystalline forms of Compound (I) are inhibitors of Bruton’s tyrosine kinase (BTK). The enzyme BTK is a member of the Tec family non-receptor tyrosine kinases.
BTK is expressed in most hematopoietic cells, including B cells, mast cells, and macrophages. BTK plays a role in the development and activation of B cells and has been implicated in multiple signaling pathways across a wide range of immune-mediated diseases. BTK activity has been implicated in the pathogenesis of several disorders and conditions, such as B cel1-related hematological cancers (e.g,, non-Hodgkin lymphoma and B cell chronic lymphocytic leukemia) and autoimmune diseases (e.g, rheumatoid arthritis,
Sjogren’s syndrome, pemphigus, IBD, lupus, and asthma).
Compound (I) and various solid forms thereof may inhibit BTK and be useful in the treatment of disorders and conditions mediated by BTK activity. Compound (I) is disclosed as, e.g., Compound 125A in Table 1 of WO 2012/158764 and has the following structure:

Example 1: Preparation of Crystalline Form (I) of Compound (I)
Methyl isobutyl ketone (MIBK; 6 mL) was added to amorphous (R)-2-(3-(4-amino-3- (2-fluoro-4-phenoxyphenyJ)-1H-pyrazolo[3,4-d]pyrimidin-1-yl)piperidine-1-carbonyl)-4,4- dimethylpent-2-enenitrile (1,0 g) and stirred to fonn a solution. After approximately five minutes of agitation, a precipitate began to form. Additional MIBK (10 mL) was charged, and the slurry was stirred. After approximately ten days, the solid was filtered and rinsed with MIBK (10 mL). The solid was dried under vacuum with heating to afford approximately 0.5 g of crystalline Form (I) of Compound (I) as a white solid.
PATENT
WO2012158764 , claiming BTK tyrosine kinase inhibitors, useful for treating cancer.
https://patents.google.com/patent/WO2012158764A1/en
WO 2012/158764 125A

PATENT
US20210205313
PATENT
US20210205312 ,
for concurrently published filings, claiming a gel composition comprising PRN-473 and use of another BTK tyrosine kinase inhibitor ie PRN1008 , respectively.
PATENT
WO2016100914 , claiming use of a BTK inhibitor ie PRN-473, alone or in combination with corticosteroid therapy, for treating pemphigus vulgaris.
PATENT
WO 2014022569
https://patents.google.com/patent/WO2014022569A1/en
//////// PRN-473, PRN 473, SAR 444727, PHASE 1
CC(C)(C)C=C(C#N)C(=O)N1CCC[C@H](C1)n1nc(c2c(N)ncnc21)c1ccc(Oc2ccccc2)cc1F
NEW DRUG APPROVALS
ONE TIME
$10.00
DEUCRAVACITINIB
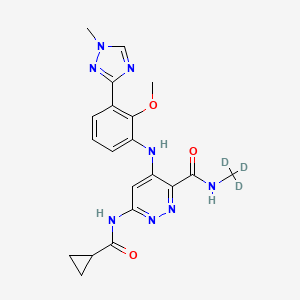
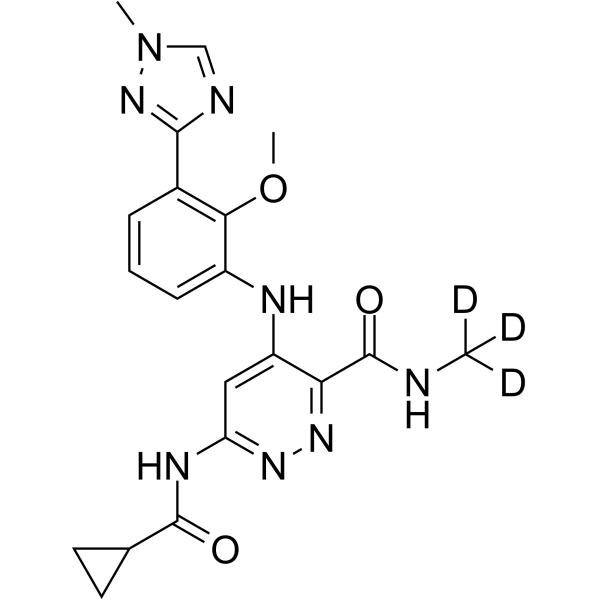
DEUCRAVACITINIB
BMS-986165
CAS 1609392-27-9, C20H22N8O3, 425.46
6-(cyclopropanecarbonylamino)-4-[2-methoxy-3-(1-methyl-1,2,4-triazol-3-yl)anilino]-N-(trideuteriomethyl)pyridazine-3-carboxamide
- OriginatorBristol-Myers Squibb
- ClassAmides; Aniline compounds; Anti-inflammatories; Antipsoriatics; Antirheumatics; Cyclopropanes; Ethers; Hepatoprotectants; Organic deuterium compounds; Pyridazines; Skin disorder therapies; Small molecules; Triazoles
- Mechanism of ActionTYK2 kinase inhibitors
- Phase IIIPlaque psoriasis
- Phase IICrohn’s disease; Lupus nephritis; Psoriatic arthritis; Systemic lupus erythematosus; Ulcerative colitis
- Phase IAutoimmune disorders
- No development reportedInflammatory bowel diseases; Psoriasis
- 02 Jul 2021Bristol-Myers Squibb plans a phase I pharmacokinetics trial (In volunteers) in USA (PO, Tablet) in July 2021 (NCT04949269)
- 14 Jun 2021Bristol-Myers Squibb plans a phase III trial for Psoriatic arthritis (Treatment-naïve) in USA, Brazil, Colombia, Czech republic, Hungary, Italy, Mexico, Romania, Spain and Taiwan in July 2021 (NCT04908202) (EudraCT2020-005097-10)
- 02 Jun 2021Interim efficacy and adverse events data from the phase III POETYK-PSO-1 trial in Psoriatic psoriasis presented at the 22nd Annual Congress of the European League Against Rheumatism (EULAR-2021)
BMS , presumed to be in collaboration with Jinan University and Chinese Academy of Sciences , is developing deucravacitinib, a TYK2 inhibitor, for treating autoimmune diseases, primarily psoriasis. In July 2021, deucravacitinib was reported to be in phase 3 clinical development.
Deucravacitinib (BMS-986165) is a highly selective, orally bioavailable allosteric TYK2 inhibitor for the treatment of autoimmune diseases, which selectively binds to TYK2 pseudokinase (JH2) domain (IC50=1.0 nM) and blocks receptor-mediated Tyk2 activation by stabilizing the regulatory JH2 domain. Deucravacitinib inhibits IL-12/23 and type I IFN pathways.
PAPER
https://pubs.acs.org/doi/abs/10.1021/acs.jmedchem.9b00444

Small molecule JAK inhibitors have emerged as a major therapeutic advancement in treating autoimmune diseases. The discovery of isoform selective JAK inhibitors that traditionally target the catalytically active site of this kinase family has been a formidable challenge. Our strategy to achieve high selectivity for TYK2 relies on targeting the TYK2 pseudokinase (JH2) domain. Herein we report the late stage optimization efforts including a structure-guided design and water displacement strategy that led to the discovery of BMS-986165 (11) as a high affinity JH2 ligand and potent allosteric inhibitor of TYK2. In addition to unprecedented JAK isoform and kinome selectivity, 11 shows excellent pharmacokinetic properties with minimal profiling liabilities and is efficacious in several murine models of autoimmune disease. On the basis of these findings, 11 appears differentiated from all other reported JAK inhibitors and has been advanced as the first pseudokinase-directed therapeutic in clinical development as an oral treatment for autoimmune diseases.
Bristol Myers Squibb Presents Positive Data from Two Pivotal Phase 3 Psoriasis Studies Demonstrating Superiority of Deucravacitinib Compared to Placebo and Otezla® (apremilast)
Significantly more patients treated with deucravacitinib achieved PASI 75 and sPGA 0/1 compared to patients treated with placebo and Otezla at Week 16, with an increased benefit versus Otezla at Week 24 and maintained through Week 52
Deucravacitinib was well tolerated with a low rate of discontinuation due to adverse events
Deucravacitinib is a first-in-class, oral, selective tyrosine kinase 2 (TYK2) inhibitor with a unique mechanism of action
Results presented as late-breaking research at the 2021 American Academy of Dermatology Virtual Meeting Experience
PRINCETON, N.J.–(BUSINESS WIRE)– Bristol Myers Squibb (NYSE:BMY) today announced positive results from two pivotal Phase 3 trials evaluating deucravacitinib, an oral, selective tyrosine kinase 2 (TYK2) inhibitor, for the treatment of patients with moderate to severe plaque psoriasis. The POETYK PSO-1 and POETYK PSO-2 trials, which evaluated deucravacitinib 6 mg once daily, met both co-primary endpoints versus placebo, with significantly more patients achieving Psoriasis Area and Severity Index (PASI) 75 response and a static Physician’s Global Assessment score of clear or almost clear (sPGA 0/1) after 16 weeks of treatment with deucravacitinib. Deucravacitinib was well tolerated with a low rate of discontinuation due to adverse events (AEs).
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20210423005134/en(Graphic: Business Wire)
Deucravacitinib demonstrated superior skin clearance compared with Otezla® (apremilast) for key secondary endpoints in both studies, as measured by PASI 75 and sPGA 0/1 responses at Week 16 and Week 24. Findings include:
PASI 75 Response in POETYK PSO-1 and POETYK PSO-2:
- At Week 16, 58.7% and 53.6% of patients receiving deucravacitinib achieved PASI 75 response, respectively, versus 12.7% and 9.4% receiving placebo and 35.1% and 40.2% receiving Otezla.
- At Week 24, 69.0% and 59.3% of patients receiving deucravacitinib achieved PASI 75 response, respectively, versus 38.1% and 37.8% receiving Otezla.
- Among patients who achieved PASI 75 response at Week 24 with deucravacitinib and continued treatment with deucravacitinib, 82.5% and 81.4%, respectively, maintained PASI 75 response at Week 52.
sPGA 0/1 Response in POETYK PSO-1 and POETYK PSO-2:
- At Week 16, 53.6% and 50.3% of patients receiving deucravacitinib achieved sPGA 0/1 response, respectively, versus 7.2% and 8.6% receiving placebo and 32.1% and 34.3% receiving Otezla.
- At Week 24, 58.4% and 50.4% of patients receiving deucravacitinib achieved sPGA 0/1 response, respectively, versus 31.0% and 29.5% receiving Otezla.
“In both pivotal studies, deucravacitinib was superior to Otezla across multiple endpoints, including measures of durability and maintenance of response, suggesting that deucravacitinib has the potential to become a new oral standard of care for patients who require systemic therapy and need a better oral option for their moderate to severe plaque psoriasis,” said April Armstrong, M.D., M.P.H., Associate Dean and Professor of Dermatology at the University of Southern California. “As many patients with moderate to severe plaque psoriasis remain undertreated or even untreated, it is also highly encouraging to see that deucravacitinib improved patient symptoms and outcomes to a greater extent than Otezla.”
Superiority of Deucravacitinib Versus Placebo and Otezla
Deucravacitinib demonstrated a robust efficacy profile, including superiority to placebo for the co-primary endpoints and to Otezla for key secondary endpoints. In addition to PASI 75 and sPGA 0/1 measures, deucravacitinib was superior to Otezla across both studies in multiple other secondary endpoints, demonstrating significant and clinically meaningful efficacy improvements in symptom burden and quality of life measures.
| POETYK PSO-1 and POETYK PSO-2 Results at Week 16 and Week 24 | ||||||
| Endpoint | POETYK PSO-1 (n=666) | POETYK PSO-2 (n=1,020) | ||||
| Deucravacitinib6 mg(n=332) | Otezla30 mg(n=168) | Placebo(n=166) | Deucravacitinib6 mg(n=511) | Otezla30 mg(n=254) | Placebo(n=255) | |
| PASI 75*a | ||||||
| Week 16 | 58.7%*† | 35.1% | 12.7% | 53.6%*‡ | 40.2% | 9.4% |
| Week 24 | 69.0%† | 38.1% | – | 59.3%† | 37.8% | – |
| sPGA 0/1*b | ||||||
| Week 16 | 53.6%*† | 32.1% | 7.2% | 50.3%*† | 34.3% | 8.6% |
| Week 24 | 58.4%† | 31.0% | – | 50.4%† | 29.5% | – |
| (Scalp) ss-PGA 0/1c | ||||||
| Week 16 | 70.8%*† | 39.1% | 17.4% | 60.3%*† | 37.3% | 17.3% |
| Week 24 | 71.8%† | 42.7% | – | 59.7%‡ | 41.6% | – |
| PSSD-Symptoms CFBd | ||||||
| Week 16 | -26.7*† | -17.8 | -3.6 | -28.3*† | -21.1 | -4.7 |
| Week 24 | -31.9† | -20.7 | – | -29.1† | -21.4 | – |
| DLQI 0/1e | ||||||
| Week 16 | 40.7%*† | 28.6% | 10.6% | 38.0%*† | 23.1% | 9.8% |
| Week 24 | 47.8%‡ | 24.2% | – | 41.8%† | 21.5% | – |
| *Co-primary endpoints for POETYK PSO-1 and POETYK PSO-2 were PASI 75 and sPGA 0/1 for deucravacitinib vs placebo at Week 16. |
| a. PASI 75 is defined as at least a 75% improvement from baseline in Psoriasis Area and Severity Index (PASI) scores. *p<0.0001 vs placebo. †p<0.0001 vs Otezla. ‡p=0.0003 vs Otezla. |
| b. sPGA 0/1 is defined as a static Physician’s Global Assessment (sPGA) score of clear or almost clear. *p<0.0001 vs placebo. †p<0.0001 vs Otezla. |
| c. ss-PGA 0/1 is defined as a scalp-specific Physician’s Global Assessment (ss-PGA) score of clear or almost clear in those with ss-PGA of at least 3 (moderate) at baseline. POETYK PSO-1: *p<0.0001 vs placebo. †p<0.0001 vs Otezla. POETYK PSO-2: *p<0.0001 vs placebo. †p<0.0001 vs Otezla. ‡p=0.0002 vs Otezla. |
| d. Change from baseline (CFB) in Psoriasis Symptoms and Signs Diary (PSSD) captures improvement in symptoms of itch, pain, stinging, burning and skin tightness in patient eDiaries. *p<0.0001 vs placebo. †p<0.0001 vs Otezla. |
| e. Dermatology Life Quality Index (DLQI) 0/1 scores reflect no effect at all on patient’s life in patients with a baseline DLQI score of ≥2. POETYK PSO-1: *p<0.0001 vs placebo. †p=0.0106 vs Otezla. ‡p<0.0001 vs Otezla. POETYK PSO-2: *p<0.0001 vs placebo. †p<0.0001 vs Otezla. |
Safety and Tolerability
Deucravacitinib was well-tolerated and had a similar safety profile in both trials. At Week 16, 2.9% of 419 patients on placebo, 1.8% of 842 patients on deucravacitinib and 1.2% of 422 patients on Otezla experienced serious adverse events (SAEs) across both studies. The most common AEs (≥5%) with deucravacitinib treatment at Week 16 were nasopharyngitis and upper respiratory tract infection with low rates of headache, diarrhea and nausea. At Week 16, 3.8% of patients on placebo, 2.4% of patients on deucravacitinib and 5.2% of patients on Otezla experienced AEs leading to discontinuation. Across POETYK PSO-1 and POETYK PSO-2 over 52 weeks, SAEs when adjusted for exposure (exposure adjusted incidence per 100 patient-years [EAIR]) were 5.7 with placebo, 5.7 with deucravacitinib and 4.0 with Otezla. In the same timeframe across both studies, EAIRs for AEs leading to discontinuation were 9.4 with placebo, 4.4 with deucravacitinib and 11.6 with Otezla. No new safety signals were observed during Weeks 16‒52.
Across both Phase 3 trials, rates of malignancy, major adverse cardiovascular events (MACE), venous thromboembolism (VTE) and serious infections were low and generally consistent across active treatment groups. No clinically meaningful changes were observed in multiple laboratory parameters (including anemia, blood cells, lipids and liver enzymes) over 52 weeks.
“The findings from both studies affirm that deucravacitinib – a first-in-class, oral, selective TYK2 inhibitor with a unique mechanism of action that inhibits the IL-12, IL-23 and Type 1 IFN pathways –may become an oral treatment of choice for people living with psoriasis. We believe deucravacitinib has significant potential across a broad range of immune-mediated diseases, and we are committed to further advancing our expansive clinical program with this agent,” said Mary Beth Harler, M.D., head of Immunology and Fibrosis Development, Bristol Myers Squibb. “We are in discussions with health authorities with the goal of bringing this new therapy to appropriate patients as soon as possible. At Bristol Myers Squibb, we are committed to building an immunology portfolio that addresses pressing unmet needs that exist for those impacted by serious dermatologic conditions and other immune-mediated diseases, to ultimately deliver the promise of living a better life.”
These results are available as a late-breaking research presentation (Session S033 – Late-Breaking Research Abstracts) as part of the 2021 American Academy of Dermatology (AAD) Virtual Meeting Experience (VMX). Full results of both studies will be submitted to a medical journal for peer review. In November 2020 and February 2021, respectively, Bristol Myers Squibb announced positive topline results from POETYK PSO-1 and POETYK PSO-2.
Visit www.bms.com/media/medical-meetings/bms-at-aad-vmx.html for more information on Bristol Myers Squibb’s scientific approach and resources on psoriasis and immune-mediated diseases.
About Deucravacitinib
Deucravacitinib (pronounced doo-krav-a-sih-ti-nib) is a first-in-class, oral, selective tyrosine kinase 2 (TYK2) inhibitor with a unique mechanism of action. Deucravacitinib is the first and only TYK2 inhibitor in clinical studies across multiple immune-mediated diseases. Bristol Myers Squibb scientists designed deucravacitinib to selectively target TYK2, thereby inhibiting signaling of interleukin (IL)-12, IL-23 and Type 1 interferon (IFN), key cytokines involved in psoriasis pathogenesis. Deucravacitinib achieves a high degree of selectivity by uniquely binding to the regulatory, rather than the active, domain of TYK2, which is structurally distinct from the regulatory domains of Janus kinase (JAK) 1, 2 and 3. At therapeutic doses, deucravacitinib does not inhibit JAK1, JAK2 or JAK3. Due to the innovative design of deucravacitinib, Bristol Myers Squibb earned recognition with the 2019 Thomas Alva Edison Patent Award for the science underpinning the clinical development of deucravacitinib.
Deucravacitinib is being studied in multiple immune-mediated diseases, including psoriasis, psoriatic arthritis, lupus and inflammatory bowel disease. In addition to POETYK PSO-1 and POETYK PSO-2, Bristol Myers Squibb is evaluating deucravacitinib in three other Phase 3 studies in psoriasis: POETYK PSO-3 (NCT04167462); POETYK PSO-4 (NCT03924427); POETYK PSO-LTE (NCT04036435). Deucravacitinib is not approved for any use in any country.
About the Phase 3 POETYK PSO-1 and POETYK PSO-2 Studies
PrOgram to Evaluate the efficacy and safety of deucravacitinib, a selective TYK2 inhibitor (POETYK) PSO-1 (NCT03624127) and POETYK PSO-2 (NCT03611751) are global Phase 3 studies designed to evaluate the safety and efficacy of deucravacitinib compared to placebo and Otezla® (apremilast) in patients with moderate to severe plaque psoriasis. Both POETYK PSO-1, which enrolled 666 patients, and POETYK PSO-2, which enrolled 1,020 patients, were multi-center, randomized, double-blind trials that evaluated deucravacitinib (6 mg once daily) compared with placebo and Otezla (30 mg twice daily). POETYK PSO-2 included a randomized withdrawal and retreatment period after Week 24.
The co-primary endpoints of both POETYK PSO-1 and POETYK PSO-2 were the percentage of patients who achieved Psoriasis Area and Severity Index (PASI) 75 response and those who achieved static Physician’s Global Assessment (sPGA) score of 0 or 1 at Week 16 versus placebo. Key secondary endpoints of the trials included the percentage of patients who achieved PASI 75 and sPGA 0/1 compared to Otezla at Week 16 and other measures.
About Psoriasis
Psoriasis is a widely prevalent, chronic, systemic immune-mediated disease that substantially impairs patients’ physical health, quality of life and work productivity. Psoriasis is a serious global problem, with at least 100 million people worldwide impacted by some form of the disease, including around 14 million people in Europe and approximately 7.5 million people in the United States. Up to 90 percent of patients with psoriasis have psoriasis vulgaris, or plaque psoriasis, which is characterized by distinct round or oval plaques typically covered by silvery-white scales. Despite the availability of effective systemic therapy, many patients with moderate to severe psoriasis remain undertreated or even untreated and are dissatisfied with current treatments. People with psoriasis report an impact on their emotional well-being, straining both personal and professional relationships and causing a reduced quality of life. Psoriasis is associated with multiple comorbidities that may impact patients’ well-being, including psoriatic arthritis, cardiovascular disease, metabolic syndrome, obesity, diabetes, inflammatory bowel disease and depression.
About Bristol Myers Squibb
Bristol Myers Squibb is a global biopharmaceutical company whose mission is to discover, develop and deliver innovative medicines that help patients prevail over serious diseases. For more information about Bristol Myers Squibb, visit us at BMS.com or follow us on LinkedIn, Twitter, YouTube, Facebook and Instagram.
Celgene and Juno Therapeutics are wholly owned subsidiaries of Bristol-Myers Squibb Company. In certain countries outside the U.S., due to local laws, Celgene and Juno Therapeutics are referred to as, Celgene, a Bristol Myers Squibb company and Juno Therapeutics, a Bristol Myers Squibb company.
Otezla® (apremilast) is a registered trademark of Amgen Inc.
PATENT
WO-2021129467
Novel crystalline polymorphic forms (CSI and CSII) of deucravacitinib (also known as BMS-986165), useful a tyrosine kinase 2 pseudokinase domain (TYK2) inhibitor for treating psoriasis, systemic lupus erythematosus, and Crohn’s disease.Tyrosine kinase 2 (TYK2) is an intracellular signal transduction kinase that can mediate interleukin-23 (IL-23), interleukin-12 (IL-12) and type I interferon (IFN) These cytokines are involved in inflammation and immune response.
BMS-986165 is the first and only new oral selective TYK2 inhibitor, clinically used to treat autoimmune and autoinflammatory diseases (such as psoriasis, psoriatic arthritis, lupus and inflammatory bowel disease, Crowe Graciousness, etc.). The results of a phase III clinical study of the drug announced in November 2020 showed that BMS-986165 has shown positive clinical effects in the treatment of moderate to severe plaque psoriasis. In addition, BMS-986165 also shows good therapeutic effects in the treatment of systemic lupus erythematosus and Crohn’s disease.
The chemical name of BMS-986165 is 6-(cyclopropaneamido)-4-((2-methoxy-3-(1-methyl-1H-1,2,4-triazol-3-yl)benzene (Yl)amino)-N-(methyl-D3)pyridazine-3-carboxamide, the structural formula is shown below, and is hereinafter referred to as “compound I”:
The crystal form is a solid in which the compound molecules are arranged in a three-dimensional order in the microstructure to form a crystal lattice. The phenomenon of drug polymorphism refers to the existence of two or more different crystal forms of the drug. Because of different physical and chemical properties, different crystal forms of the drug may have different dissolution and absorption in the body, which in turn affects the clinical efficacy and safety of the drug to a certain extent. Especially for poorly soluble solid drugs, the crystal form will have a greater impact. Therefore, drug crystal form must be an important content of drug research and also an important content of drug quality control.
WO2018183656A1 discloses compound I crystal form A (hereinafter referred to as “crystal form A”) and a preparation method thereof. The crystalline form A disclosed in WO2018183656A1 is the only known free crystalline form of Compound I. The inventor of the present application repeated the preparation method disclosed in WO2018183656A1 to obtain and characterize the crystal form A. The results show that the crystal form A has poor compressibility and high adhesion. Therefore, there is still a need in the art to develop a compound I crystalline form with good stability, good compressibility, and low adhesion for the development of drugs containing compound I.
The inventor of the present application has paid a lot of creative work and unexpectedly discovered the crystalline form CSI of compound I and the crystalline form CSII of compound I provided by the present invention, which have advantages in physical and chemical properties, preparation processing performance and bioavailability, for example, There are advantages in at least one aspect of melting point, solubility, hygroscopicity, purification, stability, adhesion, compressibility, fluidity, dissolution in vivo and in vitro, and bioavailability, especially good physical and chemical stability and mechanical stability It has good performance, good compressibility, and low adhesion, which solves the problems existing in the prior art, and is of great significance to the development of drugs containing compound I.
PATENT
US9505748 , a family member of WO2014074661 .
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2014074661
Preparation 1
Step l Int1
Step 2 Int2 Step 3 Int3 Step 4 Int4
Example 52
Step 1
[00219] To a solution of 2-methoxy-3-(l-methyl-lH-l ,2,4-triazol-3-yl)aniline (10.26 g, 50.2 mmol) and Int8 (10.5 g, 50.2 mmol) in THF (120 mL) was added lithium bis(trimethylsilyl)amide (LiHMDS, 1M in THF, 151 mL, 151 mmol) in a dropwise manner using a pressure equalized addition funnel. The reaction was run for 10 minutes after the completion of the addition and then quenched with HCl (1M aq., 126 mL, 126 mmol). The reaction was concentrated on a rotary evaporator until the majority of the THF was removed and a precipitate prevailed throughout the vessel. Water (-500 mL) was then added and the slurry sonicated for 5 minutes and stirred for 15 min. The solid was filtered off, rinsing with water and then air dried for 30 minutes. The powder was collected and dissolved in dichloromethane. The organic layer was washed with water and brine and then dried over sodium sulfate, filtered and concentrated to provide the product (12.5 g, 66% yield) (carried on as is). 1H NMR (400MHz, DMSO-d6) δ 11.11 (s, 1H), 9.36 (s, 1H), 8.56 (s, 1H), 7.72 (dd, J=7.8, 1.6 Hz, 1H), 7.60 (dd, J=7.9, 1.5 Hz, 1H), 7.29 (t, J=7.9 Hz, 1H), 7.19 (s, 1H), 3.95 (s, 3H), 3.72 (s, 3H). LC retention time 1.18 [E]. MS(E+) m/z: 377 (MH+).
Step 2
[00220] Intl3 (2.32 g, 6.16 mmol) and cyclopropanecarboxamide (1.048 g, 12.31 mmol) were dissolved in dioxane (62 mL) and Pd2(dba)3 (564 mg, 0.616 mmol), Xantphos (534 mg, 0.924 mmol) and cesium carbonate (4.01 g, 12.3 mmol) were added. The vessel was evacuated three times (backfilling with nitrogen) and then sealed and heated to 130 °C for 140 minutes. The reaction was filtered through CELITE® (eluting with ethyl acetate) and concentrated (on smaller scale this material could then be purified using preparative HPLC). The crude product was adsorbed onto CELITE® using dichloromethane, dried and purified using automated chromatography (100% EtOAc) to provide example 52 (1.22 g, 46% yield). 1H NMR (500MHz, chloroform-d) δ 10.99 (s, 1H), 8.63 (s, 1H), 8.18 (s, 1H), 8.10 (d, J=0.5 Hz, 2H), 7.81 (dd, J=7.9, 1.7 Hz, 1H), 7.51 (dd, J=7.9, 1.4 Hz, 1H), 7.33 – 7.20 (m, 7H), 4.01 (d, J=0.3 Hz, 3H), 3.82 (s, 3H), 1.73 -1.60 (m, 1H), 1.16 – 1.06 (m, 2H), 0.97 – 0.84 (m, 2H). LC retention time 6.84 [N]. MS(E+) m/z: 426 (MH+).
Example 53
[00221] To a homogeneous solution of Example 52 (50 mg, 0.12 mmol) in dichloromethane (3 mL) was added HCI (1M aq., 0.13 mL, 0.13 mmol) resulting in the solution turning yellow. The homogenous solution was concentrated down and then re-concentrated from dichloromethane twice to remove residual water, resulting in a white powder. The powder was suspended in dichloromethane and sonicated for 15 minutes, the powder was then collected via filtration, rinsing with dichloromethane to provide the corresponding HCI salt (38 mg, 70% yield). 1H NMR (500MHz, chloroform-d) δ 12.02 (s, 1H), 8.35 (s, 1H), 8.16 (s, 1H), 8.01 (dd, J=7.9, 1.5 Hz, 1H), 7.57 (br. s., 1H), 7.52 -7.46 (m, 1H), 7.36 (t, J=7.9 Hz, 1H), 4.03 (s, 3H), 3.83 (s, 3H), 2.05 – 1.95 (m, 1H), 1.16 – 1.09 (m, 2H), 1.03 (dd, J=7.4, 3.6 Hz, 2H). LC retention time 0.62 [j]. MS(E+) m/z: 426 (MH+).
[00222] Compare to NMR of parent free base: 1H NMR (500MHz, chloroform-d) δ 10.99 (s, 1H), 8.63 (s, 1H), 8.18 (s, 1H), 8.10 (d, J=0.5 Hz, 2H), 7.81 (dd, J=7.9, 1.7 Hz, 1H), 7.51 (dd, J=7.9, 1.4 Hz, 1H), 7.33 – 7.20 (m, 7H), 4.01 (d, J=0.3 Hz, 3H), 3.82 (s, 3H), 1.73 – 1.60 (m, 1H), 1.16 – 1.06 (m, 2H), 0.97 – 0.84 (m, 2H).
////////////DEUCRAVACITINIB, phase 3, BMS-986165, BMS 986165, psoriasis, systemic lupus erythematosus, Crohn’s disease,
CNC(=O)C1=NN=C(C=C1NC2=CC=CC(=C2OC)C3=NN(C=N3)C)NC(=O)C4CC4

NEW DRUG APPROVALS
one time
$10.00
BELUMOSUDIL

BELUMOSUDIL
MW 452.5
911417-87-3, SLx-2119, KD-025, KD 025, WHO 11343
2-[3-[4-(1H-indazol-5-ylamino)quinazolin-2-yl]phenoxy]-N-propan-2-ylacetamide
2-(3-(4-(lH-indazol-5-ylamino)quinazolin-2-yl)phenoxy)-N-isopropylacetamide
Belumosudil mesylate
KD025 mesylate
2109704-99-4
UPDATE FDA APPROVED 7/16/2021 To treat chronic graft-versus-host disease after failure of at least two prior lines of systemic therapy, Rezurock
New Drug Application (NDA): 214783
Company: KADMON PHARMA LLC
200 MG TABLET
FDA approves belumosudil for chronic graft-versus-host disease
On July 16, 2021, the Food and Drug Administration approved belumosudil (Rezurock, Kadmon Pharmaceuticals, LLC), a kinase inhibitor, for adult and pediatric patients 12 years and older with chronic graft-versus-host disease (chronic GVHD) after failure of at least two prior lines of systemic therapy.
Efficacy was evaluated in KD025-213 (NCT03640481), a randomized, open-label, multicenter dose-ranging trial that included 65 patients with chronic GVHD who were treated with belumosudil 200 mg taken orally once daily.
The main efficacy outcome measure was overall response rate (ORR) through Cycle 7 Day 1 where overall response included complete response (CR) or partial response (PR) according to the 2014 criteria of the NIH Consensus Development Project on Clinical Trials in Chronic Graft-versus-Host Disease. The ORR was 75% (95% CI: 63, 85); 6% of patients achieved a CR, and 69% achieved a PR. The median time to first response was 1.8 months (95% CI: 1.0, 1.9). The median duration of response, calculated from first response to progression, death, or new systemic therapies for chronic GVHD, was 1.9 months (95% CI: 1.2, 2.9). In patients who achieved response, no death or new systemic therapy initiation occurred in 62% (95% CI: 46, 74) of patients for at least 12 months since response.
The most common adverse reactions (≥ 20%), including laboratory abnormalities, were infections, asthenia, nausea, diarrhea, dyspnea, cough, edema, hemorrhage, abdominal pain, musculoskeletal pain, headache, phosphate decreased, gamma glutamyl transferase increased, lymphocytes decreased, and hypertension.
The recommended dosage of belumosudil is 200 mg taken orally once daily with food.
View full prescribing information for Rezurock.
This review was conducted under Project Orbis, an initiative of the FDA Oncology Center of Excellence. Project Orbis provides a framework for concurrent submission and review of oncology drugs among international partners. For this review, FDA collaborated with Australia’s Therapeutic Goods Administration, Health Canada, Switzerland’s Swissmedic, and the United Kingdom’s Medicines and Healthcare products Regulatory Agency.
This review used the Real-Time Oncology Review (RTOR) pilot program, which streamlined data submission prior to the filing of the entire clinical application, and the Assessment Aid, a voluntary submission from the applicant to facilitate the FDA’s assessment. The FDA approved this application 6 weeks ahead of the FDA goal date.
This application was granted priority review and breakthrough therapy designation. A description of FDA expedited programs is in the Guidance for Industry: Expedited Programs for Serious Conditions-Drugs and Biologics.
Belumosudil mesylate is an orally available rho kinase 2 (ROCK 2) inhibitor being developed at Kadmon. In 2020, the drug candidate was submitted for a new drug application (NDA) in the U.S., under a real-time oncology review pilot program, for the treatment of chronic graft-versus-host disease (cGVHD). The compound is also in phase II clinical development for the treatment of idiopathic pulmonary fibrosis and diffuse cutaneous systemic sclerosis. Formerly, the company had also been conducting clinical research for the treatment of psoriasis and non-alcoholic steatohepatitis (NASH); however, no further development has been reported for these indications. Originally developed by Nano Terra, the product was licensed to Kadmon on an exclusive global basis in 2011. In 2019, Kadmon entered into a strategic partnership with BioNova Pharmaceuticals and established a joint venture, BK Pharmaceuticals, to exclusively develop and commercialize KD-025 for the treatment of graft-versus-host disease in China. The compound has been granted breakthrough therapy designation in the U.S. for the treatment of cGVHD and orphan drug designations for cGVHD and systemic sclerosis. In the E.U. belumosudil was also granted orphan drug status in the E.U. for the treatment of cGVHD.
Kadmon , under license from NT Life Sciences , is developing belumosudil as mesylate salt, a ROCK-2 inhibitor, for treating IPF, chronic graft-versus-host disease, hepatic impairment and scleroderma. In July 2021, belumosudil was reported to be in pre-registration phase.
Belumosudil (formerly KD025 and SLx-2119) is an experimental drug being explored for the treatment of chronic graft versus host disease (cGvHD), idiopathic pulmonary fibrosis (IPF), and moderate to severe psoriasis. It is an inhibitor of Rho-associated coiled-coil kinase 2 (ROCK2; ROCK-II).[1] Belumosudil binds to and inhibits the serine/threonine kinase activity of ROCK2. This inhibits ROCK2-mediated signaling pathways which play major roles in pro- and anti-inflammatory immune cell responses. A genomic study in human primary cells demonstrated that the drug also has effects on oxidative phosphorylation, WNT signaling, angiogenesis, and KRAS signaling.[2] Originally developed by Surface Logix, Inc,[1] Belumosudil was later acquired by Kadmon Corporation. As of July 2020 the drug was in completed or ongoing Phase II clinical studies for cGvHD, IPF and psoriasis.[3]
cGvHD is a complication that can follow stem cell or hematopoietic stem cell transplantation where the transplanted cells (graft) attack healthy cells (host). This causes inflammation and fibrosis in multiple tissues. Two cytokines controlled by the ROCK2 signaling pathway, IL-17 and IL-21, have a major role in the cGvHD response. In a 2016 report using both mouse models and a limited human clinical trial ROCK2 inhibition with belumosudil targeted both the immunologic and fibrotic components of cGvHD and reversed the symptoms of the disease.[4] In October 2017 KD025 was granted orphan drug status in the United States for treatment of patients with cGvHD.[5]
IPF is a progressive fibrotic disease where the lining of the lungs become thickened and scarred.[6] Increased ROCK activity has been found in the lungs of humans and animals with IPF. Treatment with belumosudil reduced lung fibrosis in a bleomycin mouse model study.[7] Belumosudil may have a therapeutic benefit in IPF by targeting the fibrotic processes mediated by the ROCK signaling pathway.
Psoriasis is an inflammatory skin condition where patients experiences eruptions and remissions of thickened, erythematous, and scaly patches of skin. Down-regulation of pro-inflammatory responses was observed with KD025 treatment in Phase 2 clinical studies in patients with moderate to severe psoriasis.[8]
“Substance Name:Substance Name: Belumosudil [USAN]”.
PATENT
| WO2012040499 |
https://patents.google.com/patent/WO2012040499A2/en
PATENT
| CN106916145 |
https://patents.google.com/patent/CN106916145A/en

WO 2014055996, WO 2015157556



Patent
WO-2021129589
Novel crystalline polymorphic forms (N1, N2 and N15) of KD-025 (also known as belumosudil ), useful as a Rho A kinase 2 (ROCK-2) inhibitor for treating multiple sclerosis, psoriasis, rheumatoid arthritis, idiopathic pulmonary fibrosis (IPF), atherosclerosis, non-alcoholic fatty liver and systemic sclerosis. Represents the first filing from Sunshine Lake Pharma or its parent HEC Pharm that focuses on belumosudil.KD-025 is a selective ROCK2 (Rho-associated protein kinase 2, Rho-related protein kinase 2) inhibitor. It has multiple clinical indications such as the treatment of multiple sclerosis, psoriasis, rheumatoid arthritis, and Primary pulmonary fibrosis, atherosclerosis, non-alcoholic fatty liver, etc., among which many indications are in clinical phase I, and psoriasis and systemic sclerosis are in clinical phase II.
The structure of KD-025 is shown in the following formula (1).
Example 1 Preparation method of crystal form N1 of KD-025[0222]300mg of KD-025 solid was suspended and stirred in 10mL methanol at room temperature. After 22h, it was filtered, suction filtered and placed in a drying oven at 50°C under vacuum overnight to obtain 262mg of powder. The obtained crystal was detected by XPRD and confirmed to be KD-025 crystal form N1; its X-ray powder diffraction pattern was basically the same as that of Fig. 1, its DSC pattern was basically the same as that of Fig. 2, and the TGA pattern was basically the same as that of Fig. 3.
PATENT
WO2006105081 ,
Belumosudil product pat,
protection in the EU states until March 2026, expires in the US in May 2029 with US154 extension.
Example 82
2-(3-(4-(lH-indazol-5-ylamino)quinazolin-2-yl)phenoxy)-N-isopropylacetamide
[0257] A suspension of 2-(3-(4-(lH-indazol-5-ylamino)qumazolin-2-yl)ρhenoxy)acetic acid (70 mg, 0.14 mmol), PyBOP® (40 mg, 0.077 mmol), DlEA (24 μL, 0.14 mmol) in dry CH2Cl2 : DMF (2 : 0.1 mL) was stirred at RT for 15 minutes. To this solution of activated acid was added propan-2-amine (5.4 mg, 0.091 mmol). After 30 minutes, 1.0 equivalent of DIEA and 0.55 equivalents of PyBOP® were added. After stirring the solution for 15 minutes, 0.65 equivalents of propan-2-aminewere added and the mixture was stirred for an additional 30 minutes. The solvent was removed in vacuo and the crude product was purified using prep HPLC (25-50 90 rnins) to afford 2-(3-(4-(lH-indazol-5-ylamino)quinazolin-2-yl)phenoxy)-N-isopropylacetamide. (40 mg, 0.086 mmol, 61 %).
References
- ^ Jump up to:a b Boerma M, Fu Q, Wang J, Loose DS, Bartolozzi A, Ellis JL, et al. (October 2008). “Comparative gene expression profiling in three primary human cell lines after treatment with a novel inhibitor of Rho kinase or atorvastatin”. Blood Coagulation & Fibrinolysis. 19 (7): 709–18. doi:10.1097/MBC.0b013e32830b2891. PMC 2713681. PMID 18832915.
- ^ Park J, Chun KH (5 May 2020). “Identification of novel functions of the ROCK2-specific inhibitor KD025 by bioinformatics analysis”. Gene. 737: 144474. doi:10.1016/j.gene.2020.144474. PMID 32057928.
- ^ “KD025 – Clinical Trials”. ClinicalTrials.gov. Retrieved 25 July 2020.
- ^ Flynn R, Paz K, Du J, Reichenbach DK, Taylor PA, Panoskaltsis-Mortari A, et al. (April 2016). “Targeted Rho-associated kinase 2 inhibition suppresses murine and human chronic GVHD through a Stat3-dependent mechanism”. Blood. 127 (17): 2144–54. doi:10.1182/blood-2015-10-678706. PMC 4850869. PMID 26983850.
- ^ Shanley M (October 6, 2017). “Therapy to Treat Transplant Complications Gets Orphan Drug Designation”. RareDiseaseReport. Retrieved 25 July 2018.
- ^ “Pulmonary Fibrosis”. The Mayo Clinic. Retrieved July 25, 2018.
- ^ Semedo D (June 5, 2016). “Phase 2 Study of Molecule Inhibitor for Idiopathic Pulmonary Fibrosis Begins”. Lung Disease News. BioNews Services, LLC. Retrieved 25 July 2018.
- ^ Zanin-Zhorov A, Weiss JM, Trzeciak A, Chen W, Zhang J, Nyuydzefe MS, et al. (May 2017). “Cutting Edge: Selective Oral ROCK2 Inhibitor Reduces Clinical Scores in Patients with Psoriasis Vulgaris and Normalizes Skin Pathology via Concurrent Regulation of IL-17 and IL-10”. Journal of Immunology. 198 (10): 3809–3814. doi:10.4049/jimmunol.1602142. PMC 5421306. PMID 28389592.
| Clinical data | |
|---|---|
| Routes of administration |
Oral administration (tablets or capsules) |
| ATC code | None |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 911417-87-3 |
| PubChem CID | 11950170 |
| UNII | 834YJF89WO |
| CompTox Dashboard (EPA) | DTXSID80238425 |
| Chemical and physical data | |
| Formula | C26H24N6O2 |
| Molar mass | 452.518 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI |
////////////BELUMOSUDIL, SLx-2119, KD-025, KD 025, WHO 11343, PHASE 2, cGvHD, IPF, psoriasis, Breakthrough Therapy, Orphan Drug Designation
CC(C)NC(=O)COC1=CC=CC(=C1)C2=NC3=CC=CC=C3C(=N2)NC4=CC5=C(C=C4)NN=C5
NEW DRUG APPROVALS
ONE TIME
$10.00
Asparaginase erwinia chrysanthemi (recombinant)-rywn
Sequence:
1ADKLPNIVIL ATGGTIAGSA ATGTQTTGYK AGALGVDTLI NAVPEVKKLA51NVKGEQFSNM ASENMTGDVV LKLSQRVNEL LARDDVDGVV ITHGTDTVEE101SAYFLHLTVK SDKPVVFVAA MRPATAISAD GPMNLLEAVR VAGDKQSRGR151GVMVVLNDRI GSARYITKTN ASTLDTFKAN EEGYLGVIIG NRIYYQNRID201KLHTTRSVFD VRGLTSLPKV DILYGYQDDP EYLYDAAIQH GVKGIVYAGM251GAGSVSVRGI AGMRKAMEKG VVVIRSTRTG NGIVPPDEEL PGLVSDSLNP301AHARILLMLA LTRTSDPKVI QEYFHTY
>Protein sequence for asparaginase (Erwinia chrysanthemi) monomer ADKLPNIVILATGGTIAGSAATGTQTTGYKAGALGVDTLINAVPEVKKLANVKGEQFSNM ASENMTGDVVLKLSQRVNELLARDDVDGVVITHGTDTVEESAYFLHLTVKSDKPVVFVAA MRPATAISADGPMNLLEAVRVAGDKQSRGRGVMVVLNDRIGSARYITKTNASTLDTFKAN EEGYLGVIIGNRIYYQNRIDKLHTTRSVFDVRGLTSLPKVDILYGYQDDPEYLYDAAIQH GVKGIVYAGMGAGSVSVRGIAGMRKAMEKGVVVIRSTRTGNGIVPPDEELPGLVSDSLNP AHARILLMLALTRTSDPKVIQEYFHTY
References:
- Therapeutic Targets Database: TTD Biologic drug sequences in fasta format [Link]
Asparaginase erwinia chrysanthemi (recombinant)-rywn
JZP458-201
JZP458
CAS Registry Number 1349719-22-7
Protein Chemical FormulaC1546H2510N432O476S9
Protein Average Weight 140000.0 Da
Rylaze, FDA APPROVED 6/30/2021, BLA 761179
L-Asparaginase (ec 3.5.1.1, L-asparagine amidohydrolase) erwinia chrysanthemi tetramer alpha4Asparaginase (Dickeya chrysanthemi subunit)
Other Names
- Asparaginase Erwinia chrysanthemi
- Crisantaspase
- Cristantaspase
- Erwinase
- Erwinaze
- L-Asparagine amidohydrolase (Erwinia chrysanthemi subunit)
Asparaginase erwinia chrysanthemi [USAN]
L-Asparaginase, erwinia chrysanthemi
Asparaginase (erwinia chrysanthemi)
Asparaginase erwinia chrysanthemi
L-Asparaginase (ec 3.5.1.1, L-asparagine amidohydrolase) erwinia chrysanthemi tetramer alpha4
Asparaginase erwinia chrysanthemi (recombinant) [USAN]
Asparaginase erwinia chrysanthemi (recombinant)
A hydrolase enzyme that converts L-asparagine and water to L-aspartate and NH3.
NCI: Asparaginase Erwinia chrysanthemi. An enzyme isolated from the bacterium Erwinia chrysanthemi (E. carotovora). Asparagine is critical to protein synthesis in leukemic cells, which cannot synthesize this amino acid due to the absence of the enzyme asparagine synthase. Asparaginase hydrolyzes L-asparagine to L-aspartic acid and ammonia, thereby depleting leukemic cells of asparagine and blocking protein synthesis and tumor cell proliferation, especially in the G1 phase of the cell cycle. This agent also induces apoptosis in tumor cells. The Erwinia-derived product is often used for those patients who have experienced a hypersensitivity reaction to the E. Coli formulation. (NCI Thesaurus)
- Treatment of Acute Lymphoblastic Leukemia (ALL)
- Antineoplastic Agents
| 10MG/0.5ML | INJECTABLE;INTRAMUSCULAR |
PATENT
WO 2011003633
https://patents.google.com/patent/WO2011003633A1/en
The present invention concerns a conjugate of a protein having substantial L-asparagine aminohydrolase activity and polyethylene glycol, particularly wherein the polyethylene glycol has a molecular weight less than or equal to about 5000 Da, particularly a conjugate wherein the protein is a L-asparaginase from Erwinia, and its use in therapy.Proteins with L-asparagine aminohydrolase activity, commonly known as L- asparaginases, have successfully been used for the treatment of Acute Lymphoblastic Leukemia(ALL) in children for many years. ALL is the most common childhood malignancy (Avramis and Panosyan, Clin. Pharmacokinet. (2005) 44:367-393).[0003] L-asparaginase has also been used to treat Hodgkin’s disease, acute myelocytic leukemia, acute myelomonocytic leukemia, chronic lymphocytic leukemia, lymphosarcoma, reticulosarcoma, and melanosarcoma (Kotzia and Labrou, J. Biotechnol. 127 (2007) 657-669).The anti-tumor activity of L-asparaginase is believed to be due to the inability or reduced ability of certain malignant cells to synthesize L-asparagine (Kotzia and Labrou, J. Biotechnol. 127 (2007) 657-669). These malignant cells rely on an extracellular supply of L-asparagine. However, the L-asparaginase enzyme catalyzes the hydrolysis of L-asparagine to aspartic acid and ammonia, thereby depleting circulating pools of L-asparagine and killing tumor cells which cannot perform protein synthesis without L-asparagine (Kotzia and Labrou, J. Biotechnol. 127 (2007) 657-669).[0004] L-asparaginase from E. coli was the first enzyme drug used in ALL therapy and has been marketed as Elspar® in the USA or as Kidrolase® and L-asparaginase Medac® in Europe. L- asparaginases have also been isolated from other microorganisms, e.g., an L-asparaginase protein from Erwinia chrysanthemi, named crisantaspase, that has been marketed as Erwinase® (Wriston Jr., J.C. (1985) “L-asparaginase” Meth. Enzymol. 113, 608-618; Goward, CR. et al. (1992) “Rapid large scale preparation of recombinant Erwinia chrysanthemi L-asparaginase”, Bioseparation 2, 335-341). L-asparaginases from other species of Erwinia have also been identified, including, for example, Erwinia chrysanthemi 3937 (Genbank Accession#AAS67028), Erwinia chrysanthemi NCPPB 1125 (Genbank Accession #CAA31239), Erwinia carotovora (Genbank Accession #AAP92666), and Erwinia carotovora subsp. Astroseptica (Genbank Accession #AAS67027). These Erwinia chrysanthemi L-asparaginases have about 91-98% amino acid sequence identity with each other, while the Erwinia carotovora L- asparaginases have approximately 75-77% amino acid sequence identity with the Erwinia chrysanthemi L-asparaginases (Kotzia and Labrou, J. Biotechnol. 127 (2007) 657-669).[0005] L-asparaginases of bacterial origin have a high immunogenic and antigenic potential and frequently provoke adverse reactions ranging from mild allergic reaction to anaphylactic shock in sensitized patients (Wang, B. et al. (2003) “Evaluation of immunologic cross reaction of anti- asparaginase antibodies in acute lymphoblastic leukemia (ALL and lymphoma patients),Leukemia 17, 1583-1588). E. coli L-asparaginase is particularly immunogenic, with reports of the presence of anti-asparaginase antibodies to E. coli L-asparaginase following i.v. or i.m. administration reaching as high as 78% in adults and 70% in children (Wang, B. et al. (2003) Leukemia 17, 1583-1588).[0006] L-asparaginases from Escherichia coli and Erwinia chrysanthemi differ in their pharmacokinetic properties and have distinct immunogenic profiles, respectively (Klug Albertsen, B. et al. (2001) “Comparison of intramuscular therapy with Erwinia asparaginase and asparaginase Medac: pharmacokinetics, pharmacodynamics, formation of antibodies and influence on the coagulation system” Brit. J. Haematol. 115, 983-990). Furthermore, it has been shown that antibodies that developed after a treatment with L-asparaginase from E. coli do not cross react with L-Asparaginase from Erwinia (Wang, B. et al., Leukemia 17 (2003) 1583-1588). Thus, L-asparaginase from Erwinia (crisantaspase) has been used as a second line treatment of ALL in patients that react to E. coli L-asparaginase (Duval, M. et al. (2002) “Comparison of Escherichia co/z-asparaginase with £Vwzm‘α-asparaginase in the treatment of childhood lymphoid malignancies: results of a randomized European Organisation for Research and Treatment ofCancer, Children’s Leukemia Group phase 3 trial” Blood 15, 2734-2739; Avramis and Panosyan,Clin. Pharmacokinet. (2005) 44:367-393).[0007] In another attempt to reduce immunogenicity associated with administration of microbial L-asparaginases, an E. coli L-asparaginase has been developed that is modified with methoxy- polyethyleneglycol (mPEG). This method is commonly known as “PEGylation” and has been shown to alter the immunological properties of proteins (Abuchowski, A. et al. (1977) “Alteration of Immunological Properties of Bovine Serum Albumin by Covalent Attachment of Polyethylene Glycol,” J.Biol.Chem. 252 (11), 3578-3581). This so-called mPEG-L- asparaginase, or pegaspargase, marketed as Oncaspar® (Enzon Inc., USA), was first approved in the U.S. for second line treatment of ALL in 1994, and has been approved for first- line therapy of ALL in children and adults since 2006. Oncaspar® has a prolonged in vivo half-life and a reduced immunogenicity/antigenicity.[0008] Oncaspar® is E. coli L-asparaginase that has been modified at multiple lysine residues using 5 kDa mPEG-succinimidyl succinate (SS-PEG) (U.S. Patent No. 4,179,337). SS-PEG is aPEG reagent of the first generation that contains an instable ester linkage that is sensitive to hydro lysis by enzymes or at slightly alkaline pH values (U.S. Patent No. 4,670,417; Makromol. Chem. 1986, 187, 1131-1144). These properties decrease both in vitro and in vivo stability and can impair drug safety.[0009] Furthermore, it has been demonstrated that antibodies developed against L-asparaginase from E. coli will cross react with Oncaspar® (Wang, B. et al. (2003) “Evaluation of immunologic cross-reaction of anti-asparaginase antibodies in acute lymphoblastic leukemia (ALL and lymphoma patients),” Leukemia 17, 1583-1588). Even though these antibodies were not neutralizing, this finding clearly demonstrated the high potential for cross-hypersensitivity or cross-inactivation in vivo. Indeed, in one report 30-41% of children who received pegaspargase had an allergic reaction (Wang, B. et al. (2003) Leukemia 17, 1583-1588).[0010] In addition to outward allergic reactions, the problem of “silent hypersensitivity” was recently reported, whereby patients develop anti-asparaginase antibodies without showing any clinical evidence of a hypersensitivity reaction (Wang, B. et al. (2003) Leukemia 17, 1583-1588). This reaction can result in the formation of neutralizing antibodies to E. coli L-asparaginase and pegaspargase; however, these patients are not switched to Erwinia L-asparaginase because there are not outward signs of hypersensitivity, and therefore they receive a shorter duration of effective treatment (Holcenberg, J., J. Pediatr. Hematol. Oncol. 26 (2004) 273-274).[0011] Erwinia chrysanthemi L-asparaginase treatment is often used in the event of hypersensitivity to E. co/z-derived L-asparaginases. However, it has been observed that as many as 30-50% of patients receiving Erwinia L-asparaginase are antibody-positive (Avramis andPanosyan, Clin. Pharmacokinet. (2005) 44:367-393). Moreover, because Erwinia chrysanthemi L-asparaginase has a significantly shorter elimination half-life than the E. coli L-asparaginases, it must be administered more frequently (Avramis and Panosyan, Clin. Pharmacokinet. (2005) 44:367-393). In a study by Avramis et al., Erwinia asparaginase was associated with inferior pharmacokinetic profiles (Avramis et al., J. Pediatr. Hematol. Oncol. 29 (2007) 239-247). E. coli L-asparaginase and pegaspargase therefore have been the preferred first-line therapies for ALL over Erwinia L-asparaginase.[0012] Numerous biopharmaceuticals have successfully been PEGylated and marketed for many years. In order to couple PEG to a protein, the PEG has to be activated at its OH terminus. The activation group is chosen based on the available reactive group on the protein that will bePEGylated. In the case of proteins, the most important amino acids are lysine, cysteine, glutamic acid, aspartic acid, C-terminal carboxylic acid and the N-terminal amino group. In view of the wide range of reactive groups in a protein nearly the entire peptide chemistry has been applied to activate the PEG moiety. Examples for this activated PEG-reagents are activated carbonates, e.g., p-nitrophenyl carbonate, succinimidyl carbonate; active esters, e.g., succinimidyl ester; and for site specific coupling aldehydes and maleimides have been developed (Harris, M., Adv. Drug – A -DeI. Rev. 54 (2002), 459-476). The availability of various chemical methods for PEG modification shows that each new development of a PEGylated protein will be a case by case study. In addition to the chemistry the molecular weight of the PEG that is attached to the protein has a strong impact on the pharmaceutical properties of the PEGylated protein. In most cases it is expected that, the higher the molecular weight of the PEG, the better the improvement of the pharmaceutical properties (Sherman, M. R., Adv. Drug Del. Rev. 60 (2008), 59-68; Holtsberg, F. W., Journal of Controlled Release 80 (2002), 259-271). For example, Holtsberg et al. found that, when PEG was conjugated to arginine deaminase, another amino acid degrading enzyme isolated from a microbial source, pharmacokinetic and pharmacodynamic function of the enzyme increased as the size of the PEG attachment increased from a molecular weight of 5000Da to 20,000 Da (Holtsberg, F.W., Journal of Controlled Release 80 (2002), 259-271).[0013] However, in many cases, PEGylated biopharmaceuticals show significantly reduced activity compared to the unmodified biopharmaceutical (Fishburn, CS. (2008) Review “The Pharmacology of PEGylation: Balancing PD with PK to Generate Novel Therapeutics” J. Pharm. Sd., 1-17). In the case of L-asparaginase from Erwinia carotovora, it has been observed that PEGylation reduced its in vitro activity to approximately 57% (Kuchumova, A.V. et al. (2007) “Modification of Recombinant asparaginase from Erwinia carotovora with Polyethylene Glycol 5000” Biochemistry (Moscow) Supplement Series B: Biomedical Chemistry, 1, 230-232). The L-asparaginase from Erwinia carotovora has only about 75% homology to the Erwinia chrysanthemi L-asparaginase (crisantaspase). For Oncaspar® it is also known that its in vitro activity is approximately 50% compared to the unmodified E. coli L-asparaginase.[0014] The currently available L-asparaginase preparations do not provide alternative or complementary therapies— particularly therapies to treat ALL— that are characterized by high catalytic activity and significantly improved pharmacological and pharmacokinetic properties, as well as reduced immunogenicity. L-asparaginase protein has at least about 80% homology or identity with the protein comprising the sequence of SEQ ID NO:1, more specifically at least about 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% homology or identity with the protein comprising the sequence of SEQ ID NO:1. SEQ ID NO:1 is as follows:ADKLPNIVILATGGTIAGSAATGTQTTGYKAGALGVDTLINAVPEVKKLANVKGE QFSNMASENMTGDVVLKLSQRVNELLARDDVDGVVITHGTDTVEESAYFLHLTV KSDKPVVFVAAMRPATAISADGPMNLLEAVRVAGDKQSRGRGVMVVLNDRIGSA RYITKTNASTLDTFKANEEGYLGVIIGNRIYYQNRIDKLHTTRSVFDVRGLTSLPKV DILYGYQDDPEYLYDAAIQHGVKGIVYAGMGAGSVSVRGIAGMRKAMEKGVVVIRSTRTGNGIVPPDEELPGLVSDSLNPAHARILLMLALTRTSDPKVIQEYFHTY (SEQ ID NO:1) [0048] The term “comprising the sequence of SEQ ID NO:1” means that the amino-acid sequence of the protein may not be strictly limited to SEQ ID NO:1 but may contain additional amino-acids.ExamplesExample 1 : Preparation of Recombinant Crisantaspase [0100] The recombinant bacterial strain used to manufacture the naked recombinant Erwinia chrysanthemi L-asparaginase protein (also referred to herein as “r-crisantaspase”) was an E. coli BL21 strain with a deleted ansB gene (the gene encoding the endogenous E. coli type II L- asparaginase) to avoid potential contamination of the recombinant Erwinia chrysanthemi L- asparaginase with this enzyme. The deletion of the ansB gene relies on homologous recombination methods and phage transduction performed according to the three following steps:1) a bacterial strain (NMI lOO) expressing a defective lambda phage which supplies functions that protect and recombine electroporated linear DNA substrate in the bacterial cell was transformed with a linear plasmid (kanamycin cassette) containing the kanamycin gene flanked by an FLP recognition target sequence (FRT). Recombination occurs to replace the ansB gene by the kanamycin cassette in the bacterial genome, resulting in a ΛansB strain; 2) phage transduction was used to integrate the integrated kanamycin cassette region from the ΛansB NMI lOO strain to the ansB locus in BL21 strain. This results in an E. coli BL21 strain with a deleted ansB gene and resistant to kanamycin; 3) this strain was transformed with a FLP -helper plasmid to remove the kanamycin gene by homologous recombination at the FRT sequence. The genome of the final strain (BL21 ΛansB strain) was sequenced, confirming full deletion of the endogenous ansB gene.[0101] The E. co/z‘-optimized DNA sequence encoding for the mature Erwinia chrysanthemi L- asparaginase fused with the ENX signal peptide from Bacillus subtilis was inserted into an expression vector. This vector allows expression of recombinant Erwinia chrysanthemi L- asparaginase under the control of hybrid T5/lac promoter induced by the addition of Isopropyl β- D-1-thiogalactopyranoside (IPTG) and confers resistance to kanamycin.[0102] BL21 ΛansB strain was transformed with this expression vector. The transformed cells were used for production of the r-crisantaspase by feed batch glucose fermentation in Reisenberg medium. The induction of the cell was done 16h at 23°C with IPTG as inducer. After cell harvest and lysis by homogenization in 1OmM sodium phosphate buffer pH6 5mM EDTA (Buffer A), the protein solution was clarified by centrifugation twice at 1500Og, followed by 0.45μm and 0.22μm filtration steps. The recombinant Erwinia chrysanthemi L-asparaginase was next purified using a sequence of chromatography and concentration steps. Briefly, the theoretical isoelectric point of the Erwinia chrysanthemi L-asparaginase (7.23) permits the recombinant enzyme to adsorb to cation exchange resins at pH6. Thus, the recombinant enzyme was captured on a Capto S column (cation exchange chromatography) and eluted with salt gradient in Buffer A. Fractions containing the recombinant enzyme were pooled. The pooled solution was next purified on Capto MMC column (cation exchange chromatography) in Buffer A with salt gradient. . The eluted fractions containing Erwinia chrysanthemi L-asparaginase were pooled and concentrated before protein separation on Superdex 200pg size exclusion chromatography as polishing step. Fractions containing recombinant enzymes were pooled, concentrated, and diafiltered against 10OmM sodium phosphate buffer pH8. The purity of the final Erwinia chrysanthemi L-asparaginase preparation was evaluated by SDS-PAGE (Figure 1) and RP-HPLC and was at least 90%. The integrity of the recombinant enzyme was verified byN-terminal sequencing and LC-MS. Enzyme activity was measured at 37°C using Nessler’s reagent. The specific activity of the purified recombinant Erwinia chrysanthemi L-asparaginase was around 600 U/mg. One unit of enzyme activity is defined as the amount of enzyme that liberates lμmol of ammonia from L-asparagine per minute at 37°C. Example 2: Preparation of 10 kDa mPEG-L- Asparaginase Conjugates[0103] A solution of L-asparaginase from Erwinia chrysanthemi was stirred in a 100 mM sodium phosphate buffer at pH 8.0, at a protein concentration between 2.5 and 4 mg/mL, in the presence of 150 mg/mL or 36 mg/mL 10 kDa mPEG-NHS, for 2 hours at 22°C. The resulting crude 10 kDa mPEG-L-asparaginase was purified by size exclusion chromatography using a Superdex 200 pg column on an Akta purifier UPC 100 system. Protein-containing fractions were pooled and concentrated to result in a protein concentration between 2 and 8 mg/mL. Two 10 kDa mPEG-L-asparaginase conjugates were prepared in this way, differing in their degree of PEGylation as determined by TNBS assay with unmodified L-asparaginase as a reference, one corresponding to full PEGylation (100% of accessible amino groups (e.g., lysine residues and/or the N-terminus) residues being conjugated corresponding to PEGylation of 78% of total amino groups (e.g., lysine residues and/or the N-terminus)); the second one corresponding to partial PEGylation (39% of total amino groups (e.g., lysine residues and/or the N-terminus) or about 50% of accessible amino groups (e.g., lysine residues and/or the N-terminus)) . SDS-PAGE analysis of the conjugates is shown in Figure 2. The resulting conjugates appeared as an essentially homogeneous band and contained no detectable unmodified r-crisantaspase.Example 3: Preparation of 5 kDa mPEG-L-Asparaginase Conjugates[0104] A solution of L-asparaginase from Erwinia chrysanthemi was stirred in a 100 mM sodium phosphate buffer at pH 8.0, at a protein concentration of 4 mg/mL, in the presence of 150 mg/mL or 22.5 mg/mL 5 kDa mPEG-NHS, for 2 hours at 22°C. The resulting crude 5 kDa mPEG-L-asparaginase was purified by size exclusion chromatography using a Superdex 200 pg column on an Akta purifier UPC 100 system. Protein-containing fractions were pooled and concentrated to result in a protein concentration between 2 and 8 mg/mL. Two 5 kDa mPEG-L- asparaginase conjugates were prepared in this way, differing in their degree of PEGylation as determined by TNBS assay with unmodified L-asparaginase as a reference, one corresponding to full PEGylation (100% of accessible amino groups (e.g., lysine residues and/or the N-terminus) being conjugated corresponding to PEGylation of 84% of total amino groups (e.g., lysine residues and/or the N-terminus)); the second one corresponding to partial PEGylation (36% of total amino groups (e.g., lysine residues and/or the N-terminus) or about 43% of accessible amino groups (e.g., lysine residues and/or the N-terminus)). SDS-PAGE analysis of the conjugates is shown in Figure 2. The resulting conjugates appeared as an essentially homogeneous band and contained no detectable unmodified r-crisantaspase.Example 4: Preparation of 2 kDa mPEG-L-Asparaginase Conjugates[0105] A solution of L-asparaginase from Erwinia chrysanthemi was stirred in a 100 mM sodium phosphate buffer pH 8.0 at a protein concentration of 4 mg/mL in the presence of150 mg/mL or 22.5 mg/mL 2 kDa mPEG-NHS for 2 hours at 22°C. The resulting crude 2 kDa mPEG-L-asparaginase was purified by size exclusion chromatography using a Superdex 200 pg column on an Akta purifier UPC 100 system. Protein containing fractions were pooled and concentrated to result in a protein concentration between 2 and 8 mg/mL. Two 2 kDa mPEG-L- asparaginase conjugates were prepared in this way, differing in their degree of PEGylation as determined by TNBS assay with unmodified L-asparaginase as reference, one corresponding to maximum PEGylation (100% of accessible amino groups (e.g., lysine residues and/or the N- terminus) being conjugated corresponding to PEGylation of 86% of total amino groups (e.g., lysine residues and/or the N-terminus)); the second one corresponding to partial PEGylation (47% of total amino groups (e.g., lysine residues and/or the N-terminus) or about 55% of accessible amino groups {e.g., lysine residues and/or the N-terminus)). SDS-PAGE analysis of the conjugates is shown in Figure 2. The resulting conjugates appeared as an essentially homogeneous band and contained no detectable unmodified r-crisantaspase.Example 5: Activity of mPEG-r-Crisantaspase Conjugates[0106] L-asparaginase aminohydrolase activity of each conjugate described in the proceeding examples was determined by Nesslerization of ammonia that is liberated from L-asparagine by enzymatic activity. Briefly, 50μL of enzyme solution were mixed with 2OmM of L-asparagine in a 50 mM Sodium borate buffer pH 8.6 and incubated for 10 min at 37°C. The reaction was stopped by addition of 200μL of Nessler reagent. Absorbance of this solution was measured at 450 nm. The activity was calculated from a calibration curve that was obtained from Ammonia sulfate as reference. The results are summarized in Table 2, below:Table 2: Activity of mPEG-r-crisantaspase conjugates

* the numbers “40%” and “100%” indicate an approximate degree of PEGylation of respectively 40-55% and 100% of accessible amino groups (see Examples 2-4, supra).** the ratio mol PEG / mol monomer was extrapolated from data using TNBS assay, that makes the assumption that all amino groups from the protein (e.g., lysine residues and the N-terminus) are accessible.[0107] Residual activity of mPEG-r-crisantaspase conjugates ranged between 483 and 543 Units/mg. This corresponds to 78-87% of L-asparagine aminohydrolase activity of the unmodified enzyme. Example 6: L-Asparagine-Depleting Effect of Unmodified Crisantaspase
PAPER
Biotechnology and Applied Biochemistry (2019), 66(3), 281-289. |
https://iubmb.onlinelibrary.wiley.com/doi/10.1002/bab.1723
Crisantaspase is an asparaginase enzyme produced by Erwinia chrysanthemi and used to treat acute lymphoblastic leukemia (ALL) in case of hypersensitivity to Escherichia coli l-asparaginase (ASNase). The main disadvantages of crisantaspase are the short half-life (10 H) and immunogenicity. In this sense, its PEGylated form (PEG-crisantaspase) could not only reduce immunogenicity but also improve plasma half-life. In this work, we developed a process to obtain a site-specific N-terminal PEGylated crisantaspase (PEG-crisantaspase). Crisantaspase was recombinantly expressed in E. coli BL21(DE3) strain cultivated in a shaker and in a 2-L bioreactor. Volumetric productivity in bioreactor increased 37% compared to shaker conditions (460 and 335 U L−1 H−1, respectively). Crisantaspase was extracted by osmotic shock and purified by cation exchange chromatography, presenting specific activity of 694 U mg−1, 21.7 purification fold, and yield of 69%. Purified crisantaspase was PEGylated with 10 kDa methoxy polyethylene glycol-N-hydroxysuccinimidyl (mPEG-NHS) at different pH values (6.5–9.0). The highest N-terminal pegylation yield (50%) was at pH 7.5 with the lowest poly-PEGylation ratio (7%). PEG-crisantaspase was purified by size exclusion chromatography and presented a KM value three times higher than crisantaspase (150 and 48.5 µM, respectively). Nonetheless, PEG-crisantaspase was found to be more stable at high temperatures and over longer periods of time. In 2 weeks, crisantaspase lost 93% of its specific activity, whereas PEG-crisantaspase was stable for 20 days. Therefore, the novel PEG-crisantaspase enzyme represents a promising biobetter alternative for the treatment of ALL.
ADKLPNIVILATGGTIAGSAATGTQTTGYKAGALGVDTLINAVPEVKKLANVKGEQFSN
MASENMTGDVVLKLSQRVNELLARDDVDGVVITHGTDTVEESAYFLHLTVKSDKPVV
FVAAMRPATAISADGPMNLLEAVRVAGDKQSRGRGVMVVLNDRIGSARYITKTNAST
LDTFKANEEGYLGVIIGNRIYYQNRIDKLHTTRSVFDVRGLTSLPKVDILYGYQDDPEY
LYDAAIQHGVKGIVYAGMGAGSVSVRGIAGMRKAMEKGVVVIRSTRTGNGIVPPDEE
LPGLVSDSLNPAHARILLMLALTRTSDPKVIQEYFHTY
Figure S1 – Amino acid sequence of the enzyme crisantaspase without the signal peptide and with the lysines highlighted in red (Swiss-Prot/TrEMBL accession number: P06608|22-348 AA).
……………………………………………………………………………………………………………………………..
As a component of a chemotherapy regimen to treat acute lymphoblastic leukemia and lymphoblastic lymphoma in patients who are allergic to E. coli-derived asparaginase products
Press ReleaseFor Immediate Release:June 30, 2021
FDA Approves Component of Treatment Regimen for Most Common Childhood Cancer
Alternative Has Been in Global Shortage Since 2016
Today, the U.S. Food and Drug Administration approved Rylaze (asparaginase erwinia chrysanthemi (recombinant)-rywn) as a component of a chemotherapy regimen to treat acute lymphoblastic leukemia and lymphoblastic lymphoma in adult and pediatric patients who are allergic to the E. coli-derived asparaginase products used most commonly for treatment. The only other FDA-approved drug for such patients with allergic reactions has been in global shortage for years.
“It is extremely disconcerting to patients, families and providers when there is a lack of access to critical drugs for treatment of a life-threatening, but often curable cancer, due to supply issues,” said Gregory Reaman, M.D., associate director for pediatric oncology in the FDA’s Oncology Center of Excellence. “Today’s approval may provide a consistently sourced alternative to a pivotal component of potentially curative therapy for children and adults with this type of leukemia.”
Acute lymphoblastic leukemia occurs in approximately 5,700 patients annually, about half of whom are children. It is the most common type of childhood cancer. One component of the chemotherapy regimen is an enzyme called asparaginase that kills cancer cells by depriving them of substances needed to survive. An estimated 20% of patients are allergic to the standard E. coli-derived asparaginase and need an alternative their bodies can tolerate.
Rylaze’s efficacy was evaluated in a study of 102 patients who either had a hypersensitivity to E. coli-derived asparaginases or experienced silent inactivation. The main measurement was whether patients achieved and maintained a certain level of asparaginase activity. The study found that the recommended dosage would provide the target level of asparaginase activity in 94% of patients.
The most common side effects of Rylaze include hypersensitivity reactions, pancreatic toxicity, blood clots, hemorrhage and liver toxicity.
This review was conducted under Project Orbis, an initiative of the FDA Oncology Center of Excellence. Project Orbis provides a framework for concurrent submission and review of oncology drugs among international partners. For this review, FDA collaborated with Health Canada, where the application review is pending.
Rylaze received Fast Track and Orphan Drug designations for this indication. Fast Track is a process designed to facilitate the development and expedite the review of drugs to treat serious conditions and fulfill an unmet medical need. Orphan Drug designation provides incentives to assist and encourage drug development for rare diseases.
The FDA granted approval of Rylaze to Jazz Pharmaceuticals.
REF
DUBLIN, June 30, 2021 /PRNewswire/ — Jazz Pharmaceuticals plc (Nasdaq: JAZZ) today announced the U.S. Food and Drug Administration (FDA) approval of Rylaze™ (asparaginase erwinia chrysanthemi (recombinant)-rywn) for use as a component of a multi-agent chemotherapeutic regimen for the treatment of acute lymphoblastic leukemia (ALL) or lymphoblastic lymphoma (LBL) in pediatric and adult patients one month and older who have developed hypersensitivity to E. coli-derived asparaginase.1 Rylaze is the only recombinant erwinia asparaginase manufactured product that maintains a clinically meaningful level of asparaginase activity throughout the entire duration of treatment, and it was developed by Jazz to address the needs of patients and healthcare providers with an innovative, high-quality erwinia-derived asparaginase with reliable supply.
“We are excited to bring this important new treatment to patients who are in critical need, and we are grateful to FDA for the approval of Rylaze based on its established safety and efficacy profile. We are pleased Rylaze was approved before the trial is complete and are diligently working to advance additional clinical trial data. We are committed to quickly engaging with FDA to evolve the Rylaze product profile with additional dosing options and an IV route of administration,” said Bruce Cozadd, chairman and CEO of Jazz Pharmaceuticals. “Thank you to our collaborators within the Children’s Oncology Group, the clinical trial investigators, patients and their families, and all of the other stakeholders who helped us achieve this significant milestone.”
Rylaze was granted orphan drug designation for the treatment of ALL/LBL by FDA in June 2021. The Biologics Licensing Application (BLA) approval followed review under the Real-Time Oncology Review (RTOR) program, an initiative of FDA’s Oncology Center of Excellence designed for efficient delivery of safe and effective cancer treatments to patients.
The company expects Rylaze will be commercially available in mid-July.
“The accelerated development and approval of Rylaze marks an important step in bringing a meaningful new treatment option for many ALL patients – most of whom are children – who cannot tolerate E. coli-derived asparaginase medicine,” said Dr. Luke Maese, assistant professor at the University of Utah, Primary Children’s Hospital and Huntsman Cancer Institute. “Before the approval of Rylaze, there was a significant need for an effective asparaginase medicine that would allow patients to start and complete their prescribed treatment program with confidence in supply.”
Recent data from a Children’s Oncology Group retrospective analysis of over 8,000 patients found that patients who did not receive a full course of asparaginase treatment due to associated toxicity had significantly lower survival outcomes – regardless of whether those patients were high risk or standard risk, slow early responders.2
About Study JZP458-201
The FDA approval of Rylaze, also known as JZP458, is based on clinical data from an ongoing pivotal Phase 2/3 single-arm, open-label, multicenter, dose confirmation study evaluating pediatric and adult patients with ALL or LBL who have had an allergic reaction to E. coli-derived asparaginases and have not previously received asparaginase erwinia chrysanthemi. The study was designed to assess the safety, tolerability and efficacy of JZP458. The determination of efficacy was measured by serum asparaginase activity (SAA) levels. The Phase 2/3 study is being conducted in two parts. The first part is investigating the intramuscular (IM) route of administration, including a Monday-Wednesday-Friday dosing schedule. The second part remains active to further confirm the dose and schedule for the intravenous (IV) route of administration.
The FDA approval of Rylaze was based on data from the first of three IM cohorts, which demonstrated the achievement and maintenance of nadir serum asparaginase activity (NSAA) greater than or equal to the level of 0.1 U/mL at 48 hours using IM doses of Rylaze 25 mg/m2. The results of modeling and simulations showed that for a dosage of 25 mg/m2 administered intramuscularly every 48 hours, the proportion of patients maintaining NSAA ≥ 0.1 U/mL at 48 hours after a dose of Rylaze was 93.6% (95% CI: 92.6%, 94.6%).1
The most common adverse reactions (incidence >15%) were abnormal liver test, nausea, musculoskeletal pain, fatigue, infection, headache, pyrexia, drug hypersensitivity, febrile neutropenia, decreased appetite, stomatitis, bleeding and hyperglycemia. In patients treated with the Rylaze, a fatal adverse reaction (infection) occurred in one patient and serious adverse reactions occurred in 55% of patients. The most frequent serious adverse reactions (in ≥5% of patients) were febrile neutropenia, dehydration, pyrexia, stomatitis, diarrhea, drug hypersensitivity, infection, nausea and viral infection. Permanent discontinuation due to an adverse reaction occurred in 9% of patients who received Rylaze. Adverse reactions resulting in permanent discontinuation included hypersensitivity (6%) and infection (3%).1
The company will continue to work with FDA and plans to submit additional data from a completed cohort of patients evaluating 25mg/m2 IM given on Monday and Wednesday, and 50 mg/m2 given on Friday in support of a M/W/F dosing schedule. Part 2 of the study is evaluating IV administration and is ongoing. The company also plans to submit these data for presentation at a future medical meeting.
Investor Webcast
The company will host an investor webcast on the Rylaze approval in July. Details will be announced separately.
About Rylaze™ (asparaginase erwinia chrysanthemi (recombinant)-rywn)
Rylaze, also known as JZP458, is approved in the U.S. for use as a component of a multi-agent chemotherapeutic regimen for the treatment of acute lymphoblastic leukemia (ALL) or lymphoblastic lymphoma (LBL) in pediatric and adult patients one month and older who have developed hypersensitivity to E. coli-derived asparaginase. Rylaze has orphan drug designation for the treatment of ALL/LBL in the United States. Rylaze is a recombinant erwinia asparaginase that uses a novel Pseudomonas fluorescens expression platform. JZP458 was granted Fast Track designation by the U.S. Food and Drug Administration (FDA) in October 2019 for the treatment of this patient population. Rylaze was approved as part of the Real-Time Oncology Review program, an initiative of the FDA’s Oncology Center of Excellence designed for efficient delivery of safe and effective cancer treatments to patients.
The full U.S. Prescribing Information for Rylaze is available at: <http://pp.jazzpharma.com/pi/rylaze.en.USPI.pdf>
Important Safety Information
RYLAZE should not be given to people who have had:
- Serious allergic reactions to RYLAZE
- Serious swelling of the pancreas (stomach pain), serious blood clots, or serious bleeding during previous asparaginase treatment
RYLAZE may cause serious side effects, including:
- Allergic reactions (a feeling of tightness in your throat, unusual swelling/redness in your throat and/or tongue, or trouble breathing), some of which may be life-threatening
- Swelling of the pancreas (stomach pain)
- Blood clots (may have a headache or pain in leg, arm, or chest)
- Bleeding
- Liver problems
Contact your doctor immediately if any of these side effects occur.
Some of the most common side effects with RYLAZE include: liver problems, nausea, bone and muscle pain, tiredness, infection, headache, fever, allergic reactions, fever with low white blood cell count, decreased appetite, mouth swelling (sometimes with sores), bleeding, and too much sugar in the blood.
RYLAZE can harm your unborn baby. Inform your doctor if you are pregnant, planning to become pregnant, or nursing. Females of reproductive potential should use effective contraception (other than oral contraceptives) during treatment and for 3 months following the final dose. Do not breastfeed while receiving RYLAZE and for 1 week after the final dose.
Tell your healthcare provider if there are any side effects that are bothersome or that do not go away.
These are not all the possible side effects of RYLAZE. For more information, ask your healthcare provider.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088 (1-800-332-1088).
About ALL
ALL is a cancer of the blood and bone marrow that can progress quickly if not treated.3 Leukemia is the most common cancer in children, and about three out of four of these cases are ALL.4 Although it is one of the most common cancers in children, ALL is among the most curable of the pediatric malignancies due to recent advancements in treatment.5,6 Adults can also develop ALL, and about four of every 10 cases of ALL diagnosed are in adults.7 The American Cancer Society estimates that almost 6,000 new cases of ALL will be diagnosed in the United States in 2021.7 Asparaginase is a core component of multi-agent chemotherapeutic regimens in ALL.8 However, asparaginase treatments derived from E. coli are associated with the potential for development of hypersensitivity reactions.9
About Lymphoblastic Lymphoma
LBL is a rare, fast-growing, aggressive subtype of Non-Hodgkin’s lymphoma, most often seen in teenagers and young adults.8 LBL is a very aggressive lymphoma – also called high-grade lymphoma – which means the lymphoma grows quickly with early spread to different parts of the body.10,11
About Jazz Pharmaceuticals plc
Jazz Pharmaceuticals plc (NASDAQ: JAZZ) is a global biopharmaceutical company whose purpose is to innovate to transform the lives of patients and their families. We are dedicated to developing life-changing medicines for people with serious diseases – often with limited or no therapeutic options. We have a diverse portfolio of marketed medicines and novel product candidates, from early- to late-stage development, in neuroscience and oncology. We actively explore new options for patients including novel compounds, small molecules and biologics, and through cannabinoid science and innovative delivery technologies. Jazz is headquartered in Dublin, Ireland and has employees around the globe, serving patients in nearly 75 countries. For more information, please visit www.jazzpharmaceuticals.com and follow @JazzPharma on Twitter.
About The Children’s Oncology Group (COG)
COG (childrensoncologygroup.org), a member of the NCI National Clinical Trials Network (NCTN), is the world’s largest organization devoted exclusively to childhood and adolescent cancer research. COG unites over 10,000 experts in childhood cancer at more than 200 leading children’s hospitals, universities, and cancer centers across North America, Australia, and New Zealand in the fight against childhood cancer. Today, more than 90% of the 14,000 children and adolescents diagnosed with cancer each year in the United States are cared for at COG member institutions. Research performed by COG institutions over the past 50 years has transformed childhood cancer from a virtually incurable disease to one with a combined 5-year survival rate of 80%. COG’s mission is to improve the cure rate and outcomes for all children with cancer.
Caution Concerning Forward-Looking Statements
This press release contains forward-looking statements, including, but not limited to, statements related to Jazz Pharmaceuticals’ belief in the potential of Rylaze to provide a reliable therapeutic option for adult and pediatric patients to maximize their chance for a cure, plans for a mid-July 2021 launch of Rylaze, the availability of a reliable supply of Rylaze and other statements that are not historical facts. These forward-looking statements are based on Jazz Pharmaceuticals’ current plans, objectives, estimates, expectations and intentions and inherently involve significant risks and uncertainties. Actual results and the timing of events could differ materially from those anticipated in such forward-looking statements as a result of these risks and uncertainties, which include, without limitation, effectively launching and commercializing new products; obtaining and maintaining adequate coverage and reimbursement for the company’s products; delays or problems in the supply or manufacture of the company’s products and other risks and uncertainties affecting the company, including those described from time to time under the caption “Risk Factors” and elsewhere in Jazz Pharmaceuticals’ Securities and Exchange Commission filings and reports (Commission File No. 001-33500), including Jazz Pharmaceuticals’ Annual Report on Form 10-K for the year ended December 31, 2020 and future filings and reports by Jazz Pharmaceuticals. Other risks and uncertainties of which Jazz Pharmaceuticals is not currently aware may also affect Jazz Pharmaceuticals’ forward-looking statements and may cause actual results and the timing of events to differ materially from those anticipated. The forward-looking statements herein are made only as of the date hereof or as of the dates indicated in the forward-looking statements, even if they are subsequently made available by Jazz Pharmaceuticals on its website or otherwise. Jazz Pharmaceuticals undertakes no obligation to update or supplement any forward-looking statements to reflect actual results, new information, future events, changes in its expectations or other circumstances that exist after the date as of which the forward-looking statements were made.
Jazz Media Contact:
Jacqueline Kirby
Vice President, Corporate Affairs
Jazz Pharmaceuticals plc
CorporateAffairsMediaInfo@jazzpharma.com
Ireland, +353 1 697 2141
U.S. +1 215 867 4910
Jazz Investor Contact:
Andrea N. Flynn, Ph.D.
Vice President, Head, Investor Relations
Jazz Pharmaceuticals plc
investorinfo@jazzpharma.com
Ireland, +353 1 634 3211
References
- Rylaze (asparaginase erwinia chrysanthemi (recombinant)-rywn) injection, for intramuscular use Prescribing Information. Palo Alto, CA: Jazz Pharmaceuticals, Inc.
- Gupta S, Wang C, Raetz EA et al. Impact of Asparaginase Discontinuation on Outcome in Childhood Acute Lymphoblastic Leukemia: A Report From the Children’s Oncology Group. J Clin Oncol. 2020 Jun 10;38(17):1897-1905. doi: 10.1200/JCO.19.03024
- National Cancer Institute. Adult Acute Lymphoblastic Leukemia Treatment (PDQ®)–Patient Version. Available at www.cancer.gov/types/leukemia/patient/adult-all-treatment-pdq. Accessed June 29, 2021
- American Cancer Society. Key Statistics for Childhood Leukemia. Available at https://www.cancer.org/cancer/leukemia-in-children/about/key-statistics.html. Accessed June 29, 2021.
- American Cancer Society. Cancer Facts & Figures 2019. www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/cancer-facts-figures-2019.html. Accessed June 29, 2021.
- Pui C, Evans W. A 50-Year Journey to Cure Childhood Acute Lymphoblastic Leukemia. Seminars in Hematology. 2013;50(3), 185-196.
- American Cancer Society. Key Statistics for Acute Lymphocytic Leukemia (ALL). Available at https://cancerstatisticscenter.cancer.org/?_ga=2.8163506.1018157754.1621008457-1989786785.1621008457#!/data-analysis/NewCaseEstimates. Accessed June 29, 2021.
- Salzer W, Bostrom B, Messinger Y et al. 2018. Asparaginase activity levels and monitoring in patients with acute lymphoblastic leukemia. Leukemia & Lymphoma. 59:8, 1797-1806, DOI: 10.1080/10428194.2017.1386305.
- Hijiya N, van der Sluis IM. Asparaginase-associated toxicity in children with acute lymphoblastic leukemia. Leuk Lymphoma. 2016;57(4):748–757. DOI: 10.3109/10428194.2015.1101098.
- Leukemia Foundation. Lymphoblastic Lymphoma. Available at https://www.leukaemia.org.au/disease-information/lymphomas/non-hodgkin-lymphoma/other-non-hodgkin-lymphomas/lymphoblastic-lymphoma/. Accessed June 29, 2021.
- Mayo Clinic. Acute Lymphocytic Leukemia Diagnosis. Available at https://www.mayoclinic.org/diseases-conditions/acute-lymphocytic-leukemia/diagnosis-treatment/drc-20369083. Accessed June 29, 2021.
SOURCE Jazz Pharmaceuticals plc
Related Links
CLIP
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4776285/

/////////////asparaginase erwinia chrysanthemi (recombinant)-rywn, Rylaze, Jazz Pharmaceuticals, JZP458-201, JZP458, FDA 2021, APPROVALS 2021, ORPHAN, Fast Track, Acute Lymphoblastic Leukemia, ALL, Antineoplastic Agents
https://chem.nlm.nih.gov/chemidplus/id/1349719227
https://go.drugbank.com/drugs/DB08886

NEW DRUG APPROVALS
ONE TIME
$10.00
Soberana 02, FINLAY-FR-2

Soberana 02
FINLAY-FR-2
cas 2543416-58-4
A SARS-CoV-2 vaccine comprising a conjugate of the spike protein RBD domain with tetanus toxoid (Finlay Vaccine Institute of Cuba)
Soberana 02, is a conjugate vaccine developed by Instituto Finlay de Vacunas.[517]
Cuba[518]
Iran[517]
517 Zimmer, Carl; Corum, Jonathan; Wee, Sui-Lee. “Coronavirus Vaccine Tracker”. The New York Times. Retrieved 30 June 2021.
518 Sesin, Carmen (14 May 2021). “Cuba begins mass Covid-19 vaccine inoculation before concluding trials”. NBC News. Retrieved 2 July 2021.
Soberana 02, technical name FINLAY-FR-2, is a COVID-19 vaccine produced by the Finlay Institute, a Cuban epidemiological research institute. It is a conjugate vaccine. This candidate followed a previous one called SOBERANA-01 (FINLAY-FR-1).[2] Professor Ihosvany Castellanos Santos said that the antigen is safe because it contains parts instead of the whole live virus, and therefore it does not require extra refrigeration, like other candidates in the world.[3] According to the WHO candidate landscape vaccine document, this vaccine requires two doses, the second one being administered 28 days after the first shot.[4]
The name of the vaccine, Soberana, is a Spanish word that means “sovereign”.[5]

Efficacy
It has shown an efficacy of 62% after only two doses, according to BioCubaFarma, though a pre-print or details of the study have not been released.[6][7][8]
Pharmacology
FINLAY-FR-2 is a conjugate vaccine. It consists of the receptor binding domain of the SARS-CoV-2 spike protein conjugated chemically to tetanus toxoid.[2]
Manufacturing
The spike protein subunit is produced in Chinese hamster ovary cell culture.[2] In a pre-print article scientists from Cuba explain details of the vaccines technology and production.[9][non-primary source needed]
| Production Deliveries Planned Production Potential Production |
Deliveries (0)Effective production (implies deliveries) (1)
Planned production
- Iran
Potential Production
- Ghana
- Argentina
In Cuba
The Cuban government says it is planning to produce 100 million doses of its vaccine to respond to its own demand and that of other countries.[12][13] Cuba has also suggested that, once it’s approved, it will offer the vaccine to tourists visiting the country.[14][15][16]
The production of the first batch of about 100,000 doses will start in April.[17] José Moya, representative of the World Health Organization and the Pan American Health Organization (PAHO) in Cuba, suggested that after the vaccine passes all clinical stages, it could be included as part of PAHO’s Revolving Fund.[18]
The roll-out began with an “Interventional Trial”[19] that consisted of inoculating 150,000 at-risk participants which seems to be defined as health-care workers.[20][21] On April 11, 2021, the Ministry of Public Health of Cuba announced that 75,000 health-care workers were inoculated with their first dose of either of the two Cuba’s Phase III vaccines (the other being Abdala).[22][23]
Outside Cuba
Vietnam, Iran, Venezuela, Argentina,[24][25][26] Pakistan, India, the African Union, Jamaica and Suriname[27] have expressed interest in purchasing the vaccine, although they are waiting on Phase 3 results.[28][29]
Iran has signed an agreement to manufacture the vaccine[30] and Argentina is negotiating one.[24][25][26] Additionally, the Cuban government offered a “transfer of technology” to Ghana and will also supply “active materials” needed to make the vaccine.[31][32][33]
While the price is currently unknown, the commercialization strategy of the vaccine will be a combination of the “impact on health” and the capability of Cuba’s system to financially support “the production of vaccines and drugs for the country”, per the director of the Finlay Institute, Vicente Vérez.[34]
Clinical trials
Phase I
FINLAY-FR-2, which started being developed in October 2020, had 40 volunteers for its Phase I, according to the Cuban Public Registry of Clinical Trials, with an open, sequential and adaptive study to assess safety, reactogenicity and explore immunogenicity of the vaccine.[35]
Phase II
Phase IIa involved 100 Cubans, and phase IIb of the vaccine will have 900 volunteers between 19 and 80 years.[36][37] Vicente Vérez, director general of the Finlay Vaccine Institute, said that the vaccine has shown to give an immune response after 14 days.[38] The second phase has been supervised by Iranian officials from the Pasteur Institute.[5]
Phase III
Phase III commenced at the beginning of March as originally scheduled,[39][15] and “ready to publish” results are expected by June.[40][41][42] The trial volunteers are divided into three groups: some will receive two doses of the vaccine 28 days apart, another group will get two doses plus a third immune booster (Soberana Plus[43][44][45]), and the third a placebo.[39]
Although the trials involve thousands of adult volunteers recruited in Havana,[46] Cuba’s public health officials have said that they will also need to conduct phase III trials abroad because the island doesn’t have an outbreak of sufficient scale to produce meaningful statistics on vaccine protection.[5][14]
On March 13, 2021, the Cuban Biotechnology and Pharmaceutical Industries Business Group (BioCubaFarma) announced on social media that it had sent 100,000 doses of its Soberana 02 coronavirus vaccine candidate to the Pasteur Institute of Iran for clinical testing, “as part of the collaboration with other countries in the development of COVID-19 vaccines.” [47]
On April 26, 2021, it was reported that a Phase III conducted by the Pasteur Institute of Iran was approved to be started in Iran[48][49][50] It was previously reported that the Institute will host Phase 3 but the pre-requisites were “technology transfer and joint production”.[51][5]
Mexico plans to host a phase 3 trial.[52]
Interventional Study
The “Interventional Study” is set both in Havana,[53] Cuba’s capital and Santiago de Cuba, Cuba’s second most populous city [54][55] and in other provinces.[56] On May 6, 2021, the Finlay Institute of Vaccines announced on social media that the following adverse events have been observed: injection site pain (20%), inflammation at the injection site (5%), and general discomfort (5%).[57][58]
Authorizations
| Full authorization Emergency authorization |
See also: List of COVID-19 vaccine authorizations § Soberana 02
References
- ^ “Cuba’s Soberana Plus against Covid-19 is showing good results”. Prensa Latina. Retrieved 10 May 2021.
- ^ Jump up to:a b c Malik JA, Mulla AH, Farooqi T, Pottoo FH, Anwar S, Rengasamy KR (January 2021). “Targets and strategies for vaccine development against SARS-CoV-2”. Biomedicine & Pharmacotherapy. 137: 111254. doi:10.1016/j.biopha.2021.111254. PMC 7843096. PMID 33550049.
- ^ Santos IC (January 2021). “Rapid response to: Covid 19: Hope is being eclipsed by deep frustration”. BMJ. 372: n171. doi:10.1136/bmj.n171.
- ^ “Draft landscape and tracker of COVID-19 candidate vaccines”. http://www.who.int. World Health Organization. Retrieved 2021-02-04.
- ^ Jump up to:a b c d Rasmussen SE, Eqbali A (12 January 2021). “Iran, Cuba, Under U.S. Sanctions, Team Up for Covid-19 Vaccine Trials”. The Wall Street Journal.
- ^ “Cuba’s homegrown Covid vaccine shows promise”. http://www.ft.com. Retrieved 2021-06-20.
- ^ “Cuba encouraged by early efficacy results of homegrown COVID-19 vaccine”. http://www.zawya.com. Retrieved 2021-06-20.
- ^ Acosta, Nelson (2021-06-20). “Cuba encouraged by early results of homegrown COVID-19 vaccine amid worst outbreak”. The Age. Retrieved 2021-06-20.
- ^ Valdes-Balbin, Yury; Santana-Mederos, Darielys; Quintero, Lauren; Fernández, Sonsire; Rodriguez, Laura; Ramirez, Belinda Sanchez; Perez, Rocmira; Acosta, Claudia; Méndez, Yanira; Ricardo, Manuel G.; Hernandez, Tays (2021-02-09). “SARS-CoV-2 RBD-Tetanus toxoid conjugate vaccine induces a strong neutralizing immunity in preclinical studies”. doi:10.1101/2021.02.08.430146.
- ^ Melimopoulos, Elizabeth. “Is Cuba closing in on COVID vaccine sovereignty?”. http://www.aljazeera.com. Retrieved 2021-05-07.
- ^ “Optimism as Cuba set to test its own Covid vaccine”. BBC News. 2021-02-16. Retrieved 2021-05-07.
- ^ “Cuba espera fabricar 100 millones de dosis de su candidato vacunal Soberana 02”. Nodal (in Spanish). 21 January 2021.
- ^ “Vaccino, Cuba pronta a produrre 100 milioni di dosi di ‘Soberana 02′”. Dire (in Italian). 21 January 2021.
- ^ Jump up to:a b Ribeiro G (4 February 2021). “Cuba to offer coronavirus vaccines to tourists”. Brazilian Report.
- ^ Jump up to:a b “Coronavirus: Vacuna cubana Soberana 02 alista fase 3 y ensayos”. Deutsche Welle (in Spanish). 5 February 2021.
- ^ Meredith S (23 February 2021). “‘Sun, sea, sand and Soberana 02’: Cuba open to inoculating tourists with homegrown Covid vaccine”. CNBC.
- ^ “Coronavirus: Vacuna cubana Soberana 02 alista fase 3 y ensayos”. Deutsche Welle (in Spanish). 5 February 2021.
Las expectativas sobre Soberana 02 son tales que el titular del organismo estatal que desarrolló la vacuna, Vicente Vérez, confirmó que mientras se aguarden los resultados de la Fase 3 solo en La Habana, en abril se dará inicio a la producción del primer lote, de alrededor de 100 mil dosis.
- ^ “Cuba anuncia fase 3 de la vacuna Soberana 02”. La Jornada(in Spanish). 7 February 2021.
Una vez que superen las etapas clínicas, la OMS podría contar con el fármaco cubano, afirmó Moya, y “pasar a ser parte del grupo de vacunas que se oferten a través del Fondo Rotatorio”, un mecanismo que desde hace cuatro décadas permite gestionar antígenos e insumos a los países de las Américas.
- ^ “SOBERANA – INTERVENTION | Registro Público Cubano de Ensayos Clínicos”. rpcec.sld.cu. Retrieved 2021-04-11.
- ^ “Cuba says it’s ‘betting it safe’ with its own Covid vaccine”. NBC News. Retrieved 2021-04-11.
- ^ “Cuba begins testing 2nd COVID-19 vaccine on health care workers”. medicalxpress.com. Retrieved 2021-04-11.
- ^ Ministry of Public Health of Cuba (11 April 2021). “[Translated] “The administration of the 1st dose of the Cuban vaccine candidates #Soberana02 and #Abdala to the 75 thousand health workers and Biocubafarma who are part of the intervention study taking place in #LaHabana has concluded.””. Twitter. Retrieved 2021-04-11.
- ^ “Cuban scientists, health workers received first anti-Covid-19 dose”. http://www.plenglish.com/index.php?o=rn&id=66247&SEO=cuban-scientists-health-workers-received-first-anti-covid-19-dose (in Spanish). Retrieved 2021-04-11.
- ^ Jump up to:a b “ILARREGUI (EMBAJADOR EN CUBA): “DURANTE ESTE AÑO PODREMOS TENER VACUNAS CUBANAS EN ARGENTINA””. RadioCut. Retrieved 2021-05-07.
- ^ Jump up to:a b Argentina, Cadena 3. “Argentina comenzó a negociar con Cuba la vacuna Soberana”. Cadena 3 Argentina (in Spanish). Retrieved 2021-05-07.
- ^ Jump up to:a b de 2021, 6 de Mayo. “Sin definiciones sobre cuándo podrían llegar, el Gobierno avanza para conseguir las vacunas Soberana y Abdala de Cuba”. infobae (in Spanish). Retrieved 2021-05-07.
- ^ admin (2021-04-09). “Cuba’s COVID-19 Vaccines Being Sought After by CARICOM Countries”. Caribbean News. Retrieved 2021-05-07.
- ^ Guenot, Marianne (2021-02-15). “Cuba is working on a homegrown COVID-19 vaccine program. It has a history of fighting disease without help from the West”. Business Insider France (in French). Retrieved 2021-05-07.
- ^ Página12 (2021-01-22). “Soberana 02: Cuba prepara cien millones de dosis de la vacuna contra el coronavirus | “No somos una multinacional. Nuestro fin es crear salud”, dijo el director del Instituto Finlay de Vacunas”. PAGINA12. Retrieved 2021-05-07.
- ^ “Cuban coronavirus vaccine to start third clinical trial phase in Iran”. Tehran Times. 2021-04-18. Retrieved 2021-05-07.
- ^ Banini | 0542440286, Awofisoye Richard. “CEO OF FDA DISCUSSES PRODUCTION OF COVID-19 VACCINE WITH CUBAN AMBASSADOR”. http://www.fdaghana.gov.gh. Retrieved 2021-05-05.
- ^ “Cuba To Transfer COVID-19 Vaccine Technology To Ghana”. http://www.gnbcc.net. Retrieved 2021-05-05.
- ^ “Cuban government offers to transfer COVID-19 Soberana 02 vaccine technology to Ghana”. Rio Times Online. 16 February 2021.
- ^ “Coronavirus: Cuba will produce 100 million doses of its Soberana 02 vaccine”. OnCubaNews English. 2021-01-21. Retrieved 2021-05-07.
- ^ “SOBERANA 02 | Registro Público Cubano de Ensayos Clínicos”. Cuban Registry of Clinical Trials (in Spanish). Retrieved 24 January 2021.
- ^ Cuba inicia nova fase de testes com vacina que desenvolve contra covid-19 (in Portuguese), Universo Online, 19 January 2021, Wikidata Q105047566
- ^ “Cuba apuesta por crear primera vacuna de América Latina contra el covid-19”. France 24 (in Spanish). 2021-01-21. Retrieved 24 January 2021.
- ^ “Cuba negotiates with other countries to develop phase 3 of Soberana 02 vaccine”. OnCubaNews English. 2020-12-30. Retrieved 24 January 2021.
- ^ Jump up to:a b “Cuban-developed vaccine enters Phase III trial”. ABS CBN. 5 March 2021.
- ^ Mega, Emiliano Rodríguez (2021-04-29). “Can Cuba beat COVID with its homegrown vaccines?”. Nature. doi:10.1038/d41586-021-01126-4. PMID 33927405.
- ^ “Cuban Vaccine Ready in July. Interview with the Cuban Ambassador to the Czech Republic”. Pressenza. 2021-03-23. Retrieved 2021-04-29.
- ^ Augustin, Ed (2021-05-12). “Cuba deploys unproven homegrown vaccines, hoping to slow an exploding virus outbreak”. The New York Times. ISSN 0362-4331. Retrieved 2021-05-14.
- ^ “L’esempio cubano sui vaccini”. http://www.ilfoglio.it (in Italian). Retrieved 2021-05-07.
- ^ Avances de las vacunas cubanas contra la COVID-19, retrieved 2021-05-07
- ^ Mega, Emiliano Rodríguez (2021-04-29). “Can Cuba beat COVID with its homegrown vaccines?”. Nature. doi:10.1038/d41586-021-01126-4. PMID 33927405.
- ^ Yaffe, Helen. “Cuba’s five COVID-19 vaccines: the full story on Soberana 01/02/Plus, Abdala, and Mambisa”. LSE Latin America and Caribbean blog. Retrieved 2021-03-31.
- ^ “Cuba sends 100,000 doses of the Soberana 02 vaccine candidate to Iran” oncubanews.com. Retrieved 19 March 2021.
- ^ “Iran-Cuba vaccine enters phase three clinical trials”. Tehran Times. 2021-04-26. Retrieved 2021-04-28.
- ^ “Cuban coronavirus vaccine to start third clinical trial phase in Iran”. Tehran Times. 2021-04-18. Retrieved 2021-04-28.
- ^ “América Latina apura una vacuna propia. Cuba, adelante; México avanza. Pero no son los únicos”. http://www.poresto.net (in Spanish). Retrieved 2021-04-28.
- ^ Marsh S (2021-01-09). “Cuba to collaborate with Iran on coronavirus vaccine”. Reuters. Retrieved 2021-01-24.
- ^ “Mexico Hopes to Work With Cuba on Covid Vaccine Phase 3 Trial”. Bloomberg.com. 2021-02-14. Retrieved 2021-05-07.
- ^ Marsh, Sarah (2021-03-24). “Nearly all Havana to receive experimental Cuban COVID-19 vaccines”. Reuters. Retrieved 2021-04-28.
- ^ BioCubaFarma (April 6, 2021). “[Translated] Updating the vaccination process with vaccine candidates #Soberana02 and #Abdala during ongoing clinical trials.#VacunasCubanasCovid19”. Twitter (in Spanish). Retrieved 2021-04-11.
- ^ “Intervention study with Covid-19 vaccine candidate Abdala begins”. Radio Cadena Agramonte. Retrieved 2021-04-28.
- ^ “Cuba administers over 62,000 doses in intervention trials”. http://www.plenglish.com/index.php?o=rn&id=66012&SEO=cuba-administers-over-62000-doses-in-intervention-trials (in Spanish). Retrieved 2021-04-28.
- ^ “[Trnslated] In more than 62 thousand applied doses of #Soberana02 the safety of the vaccine has been demonstrated. Adverse effects have been: 👉 Pain at the injection site (20%). 👉 Redness at the injection site (5%). 👉 Feeling of general malaise (5%)”. Twitter. Retrieved 2021-05-07.
- ^ “[Translated]In more than 62 thousand applied doses of #Soberana02 the safety of the vaccine has been demonstrated. Adverse effects have been: 👉 Pain at the injection site (20%). 👉 Redness at the injection site (5%). 👉 Feeling of general malaise (5%)”. Facebook. Retrieved 2021-05-07.
External links
| Scholia has a profile for SOBERANA 02 (Q105047585). |
| Vaccine description | |
|---|---|
| Target | SARS-CoV-2 |
| Vaccine type | Conjugate |
| Clinical data | |
| Other names | FINLAY-FR-2, SOBERANA PLUS[1] |
| Routes of administration | Intramuscular |
| Legal status | |
| Legal status | Full and Emergency Authorizations: List of Soberana 02 authorizations |
| Part of a series on the |
| COVID-19 pandemic |
|---|
| COVID-19 (disease)SARS-CoV-2 (virus)CasesDeaths |
| showTimeline |
| showLocations |
| showInternational response |
| showMedical response |
| showEconomic impact and recession |
| showImpacts |
| COVID-19 portal |
/////////////////SARS-CoV-2, covid 19, corona virus, vaccine, iran, cuba, Soberana 02, FINLAY-FR-2
Nature (London, United Kingdom) (2021),

NEW DRUG APPROVALS
one time
$10.00

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










![2-[(3R)-3-[4-Amino-3-(2-fluoro-4-phenoxyphenyl)pyrazolo[3,4-d]pyrimidin-1-yl]piperidine-1-carbonyl]-4,4-dimethylpent-2-enenitrile.png](https://pubchem.ncbi.nlm.nih.gov/image/imgsrv.fcgi?cid=123342594&t=l)
