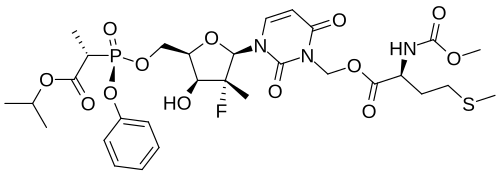
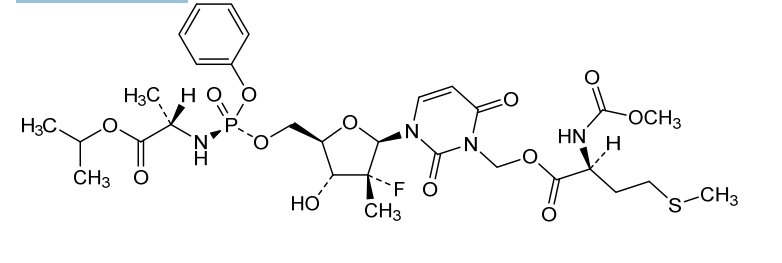
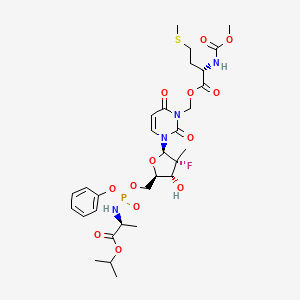
Encofosbuvir, Yiqibuvir
CAS 2232134-77-7
MF C30H42FN4O13PS MW 748.7 g/mol
- L-Alanine, O3-[N-(methoxycarbonyl)-L-methionyl]-[P(S),2’R]-2′-deoxy-2′-fluoro-3-(hydroxymethyl)-2′-methyl-P-phenyl-5′-uridylyl-, 1-methylethyl ester
- O3-[N-(Methoxycarbonyl)-L-methionyl]-[P(S),2’R]-2′-deoxy-2′-fluoro-3-(hydroxymethyl)-2′-methyl-P-phenyl-5′-uridylyl-L-alanine 1-methylethyl ester
[3-[(2R,3R,4R,5R)-3-fluoro-4-hydroxy-3-methyl-5-[[[[(2S)-1-oxo-1-propan-2-yloxypropan-2-yl]amino]-phenoxyphosphoryl]oxymethyl]oxolan-2-yl]-2,6-dioxopyrimidin-1-yl]methyl (2S)-2-(methoxycarbonylamino)-4-methylsulfanylbutanoate

antiviral, HEC 110114; Yiqibuvir, CHINA 2025, APPROVALS 2025, 82E4Q8WQV7
Encofosbuvir is a novel, oral small-molecule direct-acting antiviral (DAA) drug used to treat the hepatitis C virus (HCV). Approved by China’s National Medical Products Administration (NMPA) in March 2025, it serves as a core component of a domestic, pan-genotypic treatment regimen.
🔬 Mechanism of Action
Encofosbuvir works by targeting the machinery the virus needs to replicate itself:
- Target Enzyme: It functions as an HCV NS5B RNA-dependent RNA polymerase inhibitor.
- Viral Suppression: By selectively binding to this polymerase, it blocks the synthesis of viral RNA, effectively halting the replication and spread of the hepatitis C virus in mammals
Clinical Indications and Usage
According to the official regulatory updates from the China NMPA, encofosbuvir is prescribed under specific clinical parameters:
- Combination Therapy: It must be used in combination with netanasvir (specifically netanasvir phosphate capsules).
- Target Genotypes: The regimen is highly effective across multiple viral strains, covering HCV genotypes 1, 2, 3, and 6.
- Patient Profile: It is indicated for adult patients who are treatment-naïve (never treated before) or who have been previously treated with interferon. It is safe for use in patients with or without compensated liver cirrhosis.
The drug is classified as a Class 1 innovative drug, representing a milestone in self-developed, domestic intellectual property:
- Developers: It was jointly developed and brought to market by Sunshine Lake Pharma (a subsidiary of HEC Pharm) and YiChang HEC ChangJiang Pharmaceutical Co., Ltd.
- Dosage Form: It is distributed commercially as 0.3g tablets.
- Therapeutic Context: This medication expands the developer’s innovative hepatitis C pipeline, building upon their previously approved portfolio like emitasvir phosphate
- OriginatorHEC Pharm; Sunshine Lake Pharma
- DeveloperSunshine Lake Pharma
- ClassAntivirals
- Mechanism of ActionHepatitis C virus NS 5 protein inhibitors
- RegisteredHepatitis C
- 27 Mar 2025Registered for Hepatitis C (Combination therapy, Treatment-naive) in China (PO)
- 08 Feb 2025Preregistered for Hepatitis C (Combination therapy, Treatment-experienced) in China (PO)
- 08 Feb 2025Registered for Hepatitis C (Combination therapy, Treatment-experienced) in China (PO)
Encofosbuvir is an antiviral drug used to treat hepatitis C virus (HCV). In China, encofosbuvir is approved for use in combination with netanasvir for the treatment of adult patients with chronic HCV genotypes 1, 2, 3, or 6, who are either treatment-naive or have been previously treated with interferon.[1]
SYN
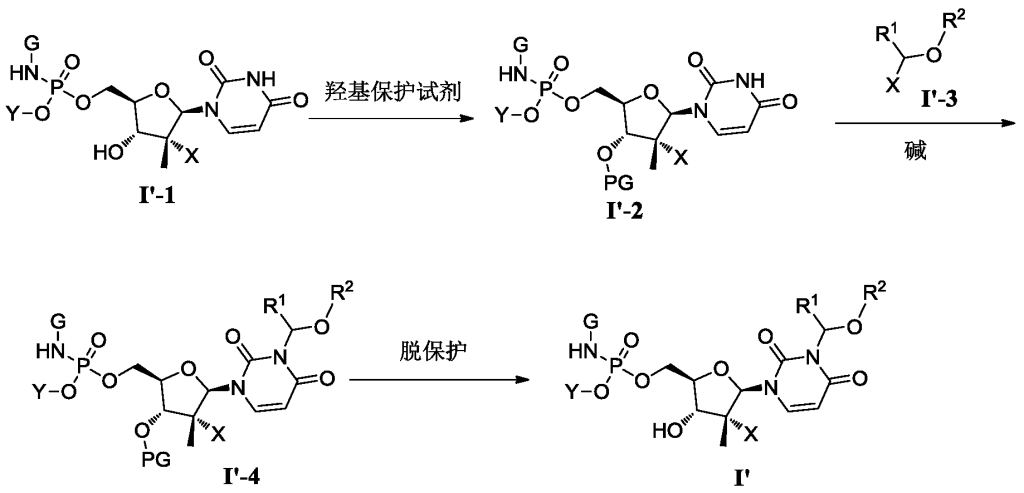
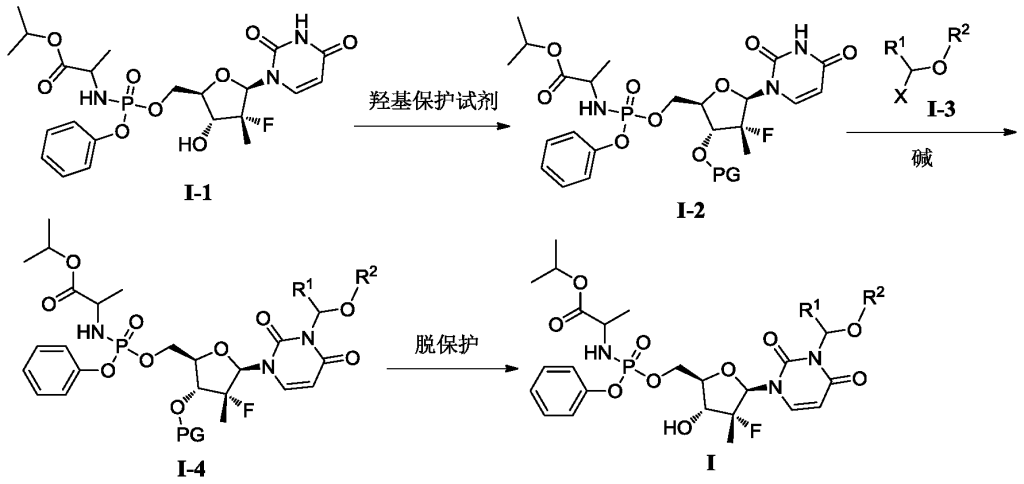
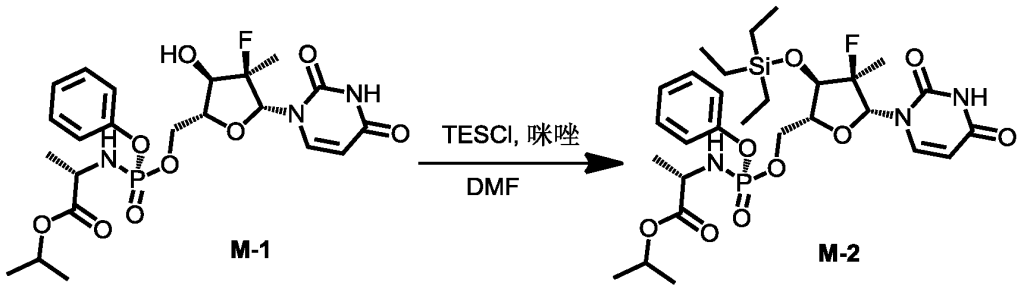
[0515](S)-(3-((2R,3R,4R,5R)-3-fluoro-4-hydroxy-5-((((S)-(((S)-1-isopropoxy-1-oxopropyl-2-yl)amino)(phenoxy)phosphoryl)oxy)methyl)-3-methyltetrahydrofuran-2-yl)-2,6-dioxo-2,3-dihydropyrimidin-1(6H)-yl)methyl-2-((methoxycarbonyl)amino)-4-(methylthio)butyrate
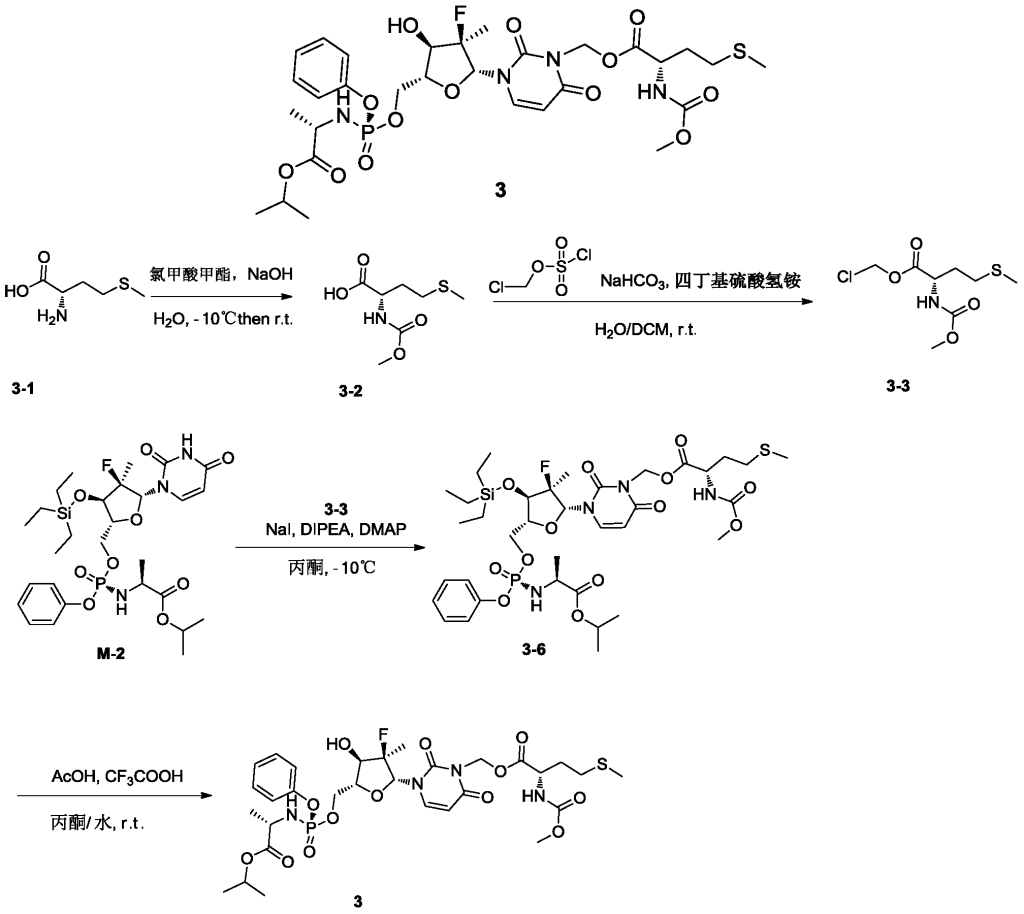
[0531]4) Synthesis of compound 3
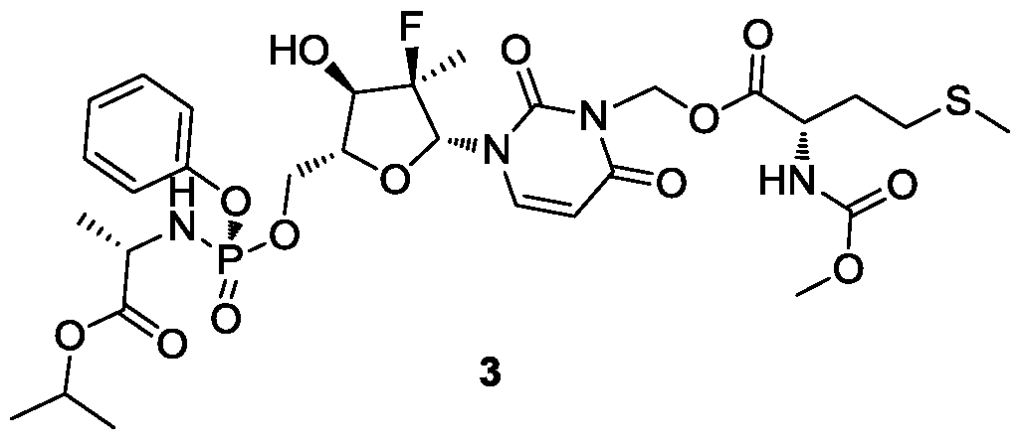
Compound 3-6 (3.63 g, 4.2 mmol, 1 eq) was dissolved in acetone (12 mL), and water (9 mL), trifluoroacetic acid (3 mL), and glacial acetic acid (12 mL) were added sequentially at room temperature, followed by a reaction time of 2 hours. After the reaction was monitored by TLC until complete, 30 mL of dichloromethane was added to the reaction solution, stirred thoroughly, and allowed to stand for phase separation. The organic phase was washed sequentially with water (10 mL × 3), saturated sodium chloride (10 mL), dried over anhydrous sodium sulfate, and the solvent was removed under reduced pressure. The solution was purified by column chromatography using DCM:MeOH = 50:1 as the eluent, yielding 2.8 g of a white foamy solid.
[0534]MS-ESI: m/z 748.8[M+1] +;
[0535]
1H NMR(400MHz,CDCl 3)δ7.49(d,J=8.2Hz,1H),7.34(d,J=7.6Hz,2H),7.24–7.16(m,3H),6.19(d,J=17.3Hz,1H),6.07–5.94(m,2H),5.75(d,J=8.3Hz,1H),5.41(d,J=7.4Hz,1H),5.07–4.95(m,1H),4.58–4.39(m,3H),4.12(d,J=8.6Hz,1H),4.02–3.80(m,4H),3.67(s,3H),2.52(t,J=7.5Hz,2H),2.14–1.90(m,5H),1.37(dd,J=18.4,14.3Hz,6H),1.24(d,J=6.3Hz,6H)。
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
“Netanasvir Phosphate Capsules Approved for Marketing by China NMPA”. National Medical Products Administration. 2025-06-11.
| Clinical data | |
|---|---|
| Trade names | 英强布韦 |
| Legal status | |
| Legal status | Rx in China |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2232134-77-7 |
| PubChem CID | 141522644 |
| UNII | 82E4Q8WQV7 |
| Chemical and physical data | |
| Formula | C30H42FN4O13PS |
| Molar mass | 748.71 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
//////////encofosbuvir, anax labs, antiviral, HEC 110114; Yiqibuvir, CHINA 2025, APPROVALS 2025, 82E4Q8WQV7

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
