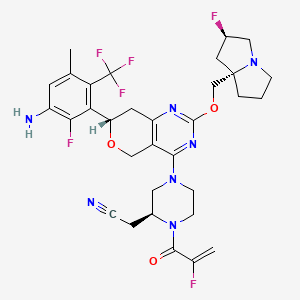
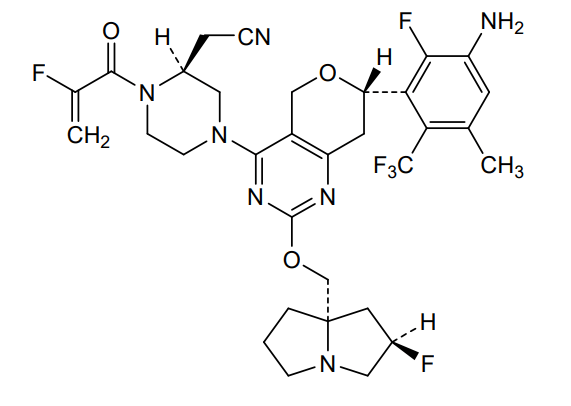
Elisrasib
CAS2914919-85-8
MFC32H35F6N7O3. MW 679.7 g/mol
2-[(2S)-4-[(7S)-7-[3-amino-2-fluoro-5-methyl-6-(trifluoromethyl)phenyl]-2-[[(2R,8S)-2-fluoro-1,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]-7,8-dihydro-5H-pyrano[4,3-d]pyrimidin-4-yl]-1-(2-fluoroprop-2-enoyl)piperazin-2-yl]acetonitrile
[(2S)-4-[(7S)-7-[3-amino-2-fluoro-5-methyl-6-(trifluoromethyl)phenyl]-2-{[(2R,7aS)-2-fluorotetrahydro-1Hpyrrolizin-7a(5H)-yl]methoxy}-7,8-dihydro-5H-pyrano[4,3-d]pyrimidin-4-yl]-1-(2-fluoroprop-2-enoyl)piperazin-2-yl]acetonitrile
Kirsten rat sarcoma viral oncogene homolog inhibitor, antineoplastic, D3S 001, PFW9YLB86H
Elisrasib (D3S-001) is a next-generation, orally available KRAS G12C inhibitor developed by D3 Bio that demonstrates high potency, sustained target engagement, and strong clinical activity in advanced solid tumors, including those resistant to first-generation inhibitors. As of April 2026, clinical trials show it has a 52% objective response rate (ORR) in G12C inhibitor-naive patients and a 30% ORR in refractory populations.
Key Aspects of Elisrasib (D3S-001):
- Mechanism of Action: It is a highly potent, covalent inhibitor that selectively binds the GDP-bound (inactive) form of the KRAS G12C mutant, effectively halting tumor cell proliferation and metastasis.
- Superior Efficacy: Preliminary data suggests elisrasib may be more potent than earlier inhibitors like sotorasib and adagrasib, providing higher target occupancy at lower doses.
- Clinical Performance (AACR 2026 Data):
- Naive Patients: 52% ORR, with a median duration of response (mDOR) of 16.5 months and median progression-free survival (mPFS) of 12.2 months at the 600 mg dose.
- Refractory Patients: 32% ORR, with a mDOR of 15.6 months and mPFS of 8.1 months.
- Targeted Cancers: Clinical trials are focused on KRAS G12C-mutant tumors, specifically non-small cell lung cancer (NSCLC), colorectal cancer (CRC), and other solid tumors.
- Safety Profile: The drug has shown good tolerability and a safe profile in early studies.
Elisrasib is in Phase 1/2 development and was highlighted for its promising results in treating patients with KRAS G12C-mutant tumors
Elisrasib is an orally bioavailable inhibitor of the oncogenic KRAS substitution mutation G12C, with potential antineoplastic activity. Upon oral administration, elisrasib selectively targets the KRAS G12C mutant and inhibits KRAS G12C-mediated signaling. This may halt proliferation and metastasis in susceptible tumor cells. KRAS, a member of the RAS family of oncogenes, serves an important role in cell signaling, division and differentiation. Mutations of KRAS may induce constitutive signal transduction leading to tumor cell proliferation, invasion, and metastasis.
- A Phase 1 Study to Assess Food Effect on the Pharmacokinetics of D3S-001 in Healthy Adult ParticipantsCTID: NCT07093398Phase: Phase 1Status: CompletedDate: 2026-03-25
- A Phase 1/2 Study of D3S-002 as Monotherapy or Combination Therapy in Adult Subjects With Advanced Solid Tumors With MAPK Pathway MutationsCTID: NCT05886920Phase: Phase 1/Phase 2Status: Active, not recruitingDate: 2026-03-23
- A Study of D3S-001 Monotherapy or Combination Therapy in Subjects With Advanced Solid Tumors With a KRAS p.G12C MutationCTID: NCT05410145Phase: Phase 1/Phase 2Status: RecruitingDate: 2026-03-12
PAT
SYN
Example 17
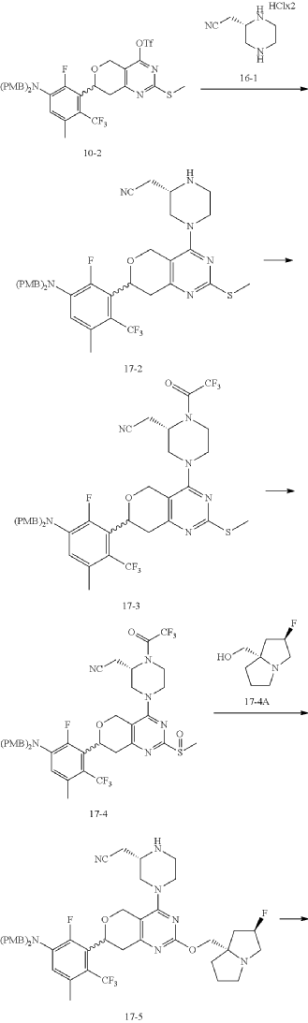
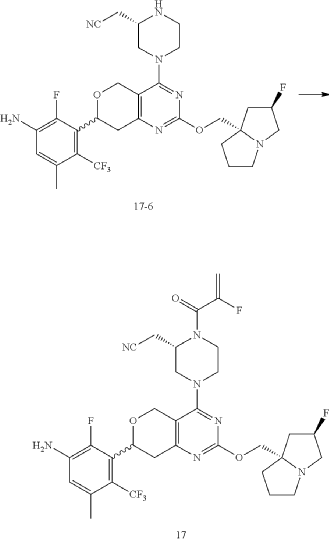
Step 6: Synthesis of Compound 17



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Pyrimidoheterocyclic compounds and application thereofPublication Number: EP-4105211-A1Priority Date: 2020-03-12
- Pyrimidoheterocyclic compounds and application thereofPublication Number: US-2023151004-A1Priority Date: 2020-03-12
//////////elisrasib, anax labs, Kirsten rat sarcoma viral oncogene homolog inhibitor, antineoplastic, D3S 001, PFW9YLB86H

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
