Home » Posts tagged 'Antineoplastic'
Tag Archives: Antineoplastic
Imofinostat
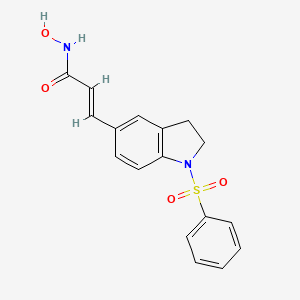
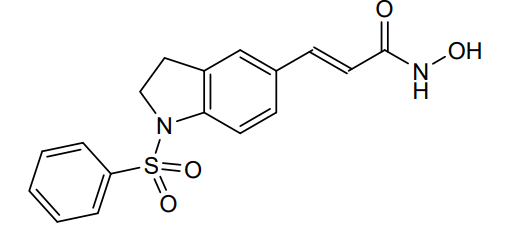
Imofinostat
CAS 1338320-94-7
MF C17H16N2O4S MW 344.4 g/mol
- 3-(1-(Benzenesulfonyl)-2,3-dihydro-1H-indol-5-yl)-N-hydroxyacrylamide
- (E)-3-[1-(benzenesulfonyl)-2,3-dihydroindol-5-yl]-N-hydroxyprop-2-enamide
(2E)-3-[1-(benzenesulfonyl)-2,3-dihydro-1H-indol-5-yl]-N-hydroxyprop2-enamide
histone deacetylase inhibitor, antineoplastic, ABT-301, MPT0E028, ABT 301, MPT0E 028, T65L58FI65
Imofinostat (also known as ABT-301 or MPT0E028) is an orally bioavailable, small-molecule histone deacetylase (HDAC) inhibitor primarily being developed as an innovative precision oncology treatment. Developed by companies like AnBogen Therapeutics and Formosa Pharmaceuticals, it is designed to reactivate tumor suppressor genes that cancer cells have silenced, thereby triggering cancer cell death (apoptosis) and stopping tumor growth.
Mechanism of Action
Imofinostat works through a distinct multi-modality approach to fight cancer cells:
- HDAC Inhibition: It acts as a potent inhibitor of human pan-histone deacetylase enzymes, showing preferential selectivity for Class I HDACs (especially HDAC3). This blocks the deacetylation of histone proteins, causing chromatin to remodel and forcing cancer cells to express tumor-suppressor genes.
- Akt Pathway Targeting: Independent of its epigenetic effects, it can directly target and reduce the activation (phosphorylation) of the Akt protein kinase, a major pathway that cancer cells use to survive and multiply.
- Microenvironment Modulation: Preclinical data shows it alters the tumor microenvironment by converting “cold tumors” (invisible to the immune system) into “hot tumors” by promoting the infiltration of CD8+ cytotoxic T cells.
Current Clinical Status & Indications
Imofinostat is actively moving through clinical trial pipelines, focusing heavily on combination therapies to overcome treatment resistance:
- Colorectal Cancer (CRC): It is currently being evaluated in a global Phase 1/2 clinical trial (NCT07244705). It is combined with the immune checkpoint inhibitor tislelizumab (Tevimbra®) and the anti-angiogenic drug bevacizumab to treat advanced, metastatic colorectal cancer.
- Pancreatic Cancer: Recent data presented at the 2026 American Association for Cancer Research (AACR) Annual Meeting demonstrates that imofinostat disrupts the HDAC3-NRF2 pathway. This action breaks down chemotherapy resistance in highly aggressive KRAS-mutant pancreatic ductal adenocarcinoma, making tumors much more sensitive to treatments like gemcitabine.
- Other Solid Tumors: Phase 1 monotherapy trials have confirmed that the drug possesses a highly competitive safety profile across a broad variety of advanced solid tumors.
Imofinostat is an orally bioavailable N-hydroxyacrylamide-derived inhibitor of both human pan-histone deacetylase (HDAC) enzymes and the serine/threonine protein kinase Akt (protein kinase B), with potential antineoplastic activity. Upon administration, imofinostat selectively binds to and inhibits HDACs, which inhibits deacetylation of histone proteins and leads to the accumulation of highly acetylated histones. This may result in both an induction of chromatin remodeling, and the selective transcription of tumor suppressor genes. This prevents cell division and induces both cell cycle arrest and apoptosis, which may inhibit the proliferation of susceptible tumor cells. In addition, imofinostat inhibits the phosphorylation and activation of Akt, which prevents the activation of downstream signaling pathways, independent of its HDAC inhibitory activity. HDACs, upregulated in many tumor cell types, are a family of enzymes that deacetylate histone proteins. Akt, overexpressed in many tumor cell types, plays a key role in tumor cell proliferation and survival.
Dose-Seeking Study of MPT0E028 in Subjects With Advanced Solid Malignancies Without Standard Treatment
CTID: NCT02350868
Phase: Phase 1
Status: Completed
Date: 2019-04-11
PAT
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2011126821&_cid=P11-MQ4LAI-84972-1
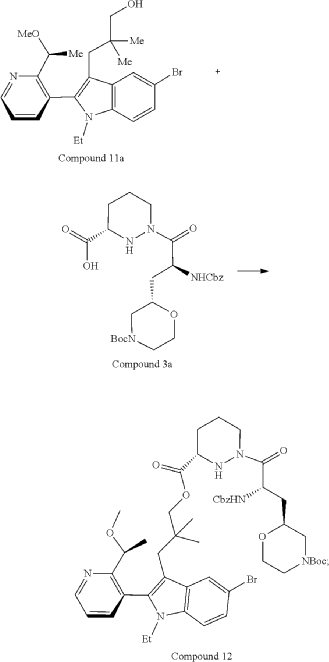
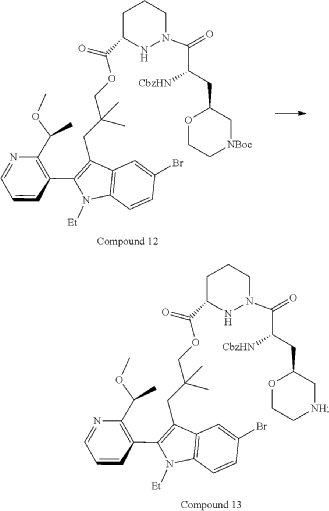
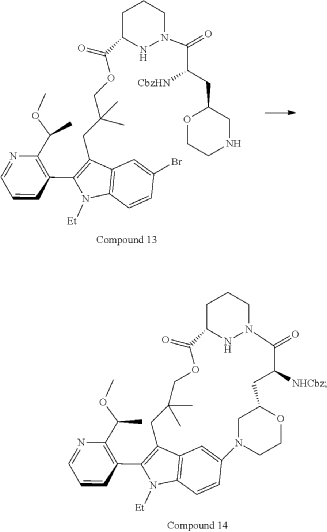
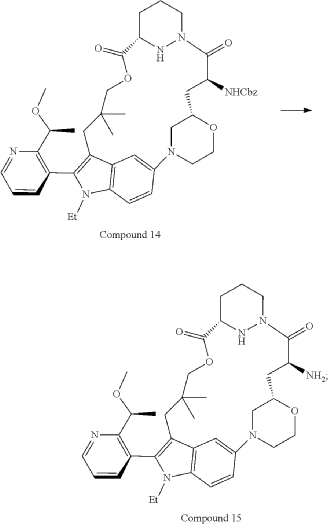
COMD 12
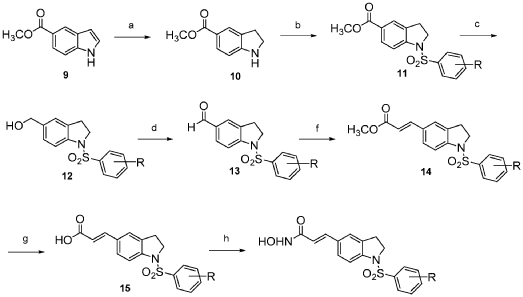
Compound 12 was synthesized via the route as shown in Scheme 3 above (reagents and conditions: (a) NaBH3CN, AcOH; (b) Benzenesulfonyl chloride, 4-methoxybenzenesulfonyl chloride, 3,4-dimethoxybenzenesulfonyl chloride, 4-fluorobenzenesulfonyl chloride, or 4-nitrobenzenesulfonyl chloride, pyridine; (c) L1AIH4, THF; (d) PDC, MS, CH2C12; f) Ph3P = CH-COOCH3, CH2C12; (g) 1M LiOH(aq), dioxane; (h) (i) NH2OTHP, PyBOP, NEt3, DMF; (ii) TFA, MeOH; (i) Fe, NH4C1, Isopropanol, H20).
2,3-Dihydro-lH-indole-5-carboxylic acid methyl ester (10): sodium cyanoborohydride (0.16 g, 2.57 mmol) was added to a solution of methyl indole-5-carboxylate (9) (0.30 g, 1.71 mmol) in AcOH (2 mL) at 0 °C. The reaction mixture was warmed to room temperature and stirred for 2 h before it was quenched with water at 0 °C. Concentrated NaOH was added to reach pH=10. The aqueous layer was extracted with CH2CI2 (15 mL x 3). The combined organic layer was dried over anhydrous MgS04 and concentrated under reduced pressure to give a yellow residue, which was purified by silica gel chromatography (EtOAc: n-hexane = 1 : 2) to afford 10 (0.28 g). 1H NMR (500MHz, CDC13): δ 3.06 (t, J= 8.5 Hz, 2H), 3.65 (t, J= 8.5 Hz, 2H), 3.84 (s, 3H), 6.53-6.55 (m, 1H), 7.75-7.76 (m, 2H).
l-Benzenesulfonyl-2,3-dihydro-lH-indole-5-carboxylic acid methyl ester (11): To a solution of 10 (0.28 g, 1.58 mmol) in pyridine (2 mL), benzenesulfonyl chloride (0.40 ml, 3.16 mmol) was added. The reaction mixture was refluxed overnight. The mixture was then purified by silica gel chromatography (EtOAc: n-hexane = 1 : 3) to afford 11 (0.40 g). 1H NMR (500MHz, CDCI3): δ 2.99 (t, J= 8.6 Hz, 2H), 3.87 (s, 3H), 3.97 (t, J= 8.6 Hz, 2H), 7.45-7.48 (m, 2H), 7.56-7.59 (m, 1H), 7.66 (d, J= 8.5 Hz, 1H), 7.75 (s, 1H), 7.82 (d, J= 7.7 Hz, 2H), 7.90 (d, J= 7.9 Hz, 1H).
(l-Benzenesulfonyl-2,3-dihydro-lH-indol-5-yl)-methanol (12): LAH (0.10 g, 2.52 mmol) was added to a solution of 11 (0.40 g, 1.26 mmol) in THF (10 mL) at 0 °C. The reaction mixture was warmed to room temperature and stirred for 2 h before it was quenched with water and then extracted with CH2CI2 (15 mL x 3). The combined organic layer was dried over anhydrous MgS04 and concentrated under reduced pressure. The reaction mixture was purified by silica gel chromatography (EtOAc: n-hexane = 1 : 1) to afford 12 (0.24 g). 1H NMR (500MHz, CDC13): δ 2.83 (t, J= 8.4 Hz, 2H), 3.92 (t, J= 8.5 Hz, 2H), 4.49 (s, 2H), 7.09 (s, 1H), 7.16 (d, J= 8.2 Hz, 1H), 7.46-7.49 (m, 2H), 7.53 (d, J= 8.2 Hz, 1H), 7.60 (t, J= 7.5 Hz, 1H), 7.76 (d, J= 7.7 Hz, 2H).
l-Benzenesulfonyl-2,3-dihydro-lH-indole-5-carbaldehyde (13): molecular sieves (0.63g) were added to a solution of 12 (0.24 g, 0.83 mmol) in CH2C12 (10 mL), PDC (0.63 g, 1.66 mmol). The mixture was stirred at room temperature overnight before it was filtered through celite. The organic layer was concentrated under reduced pressure then purified by silica gel chromatography (EtOAc: n-hexane = 1 : 2) to afford 13 (0.19 g). 1H NMR (500MHz, CDC13): δ 3.05 (t, J= 8.6 Hz, 2H), 4.01 (t, J= 8.7 Hz, 2H), 7.46-7,49 (m, 2H), 7.58-7.62 (m, 2H), 7.71 (d, J= 8.3 Hz, 1H), 7.75 (d, J= 8.3 Hz, 1H), 7.84 (d, J= 7.8 Hz, 2H), 9.85 (s, 1H).
3-(l-Benzenesulfonyl-2,3-dihydro-lH-indol-5-yl)-acrylic acid methyl ester (14): Methyl (triphenylphosphoranylidene) acetate (0.27 g, 0.79 mmol) was added to a solution of 13 (0.19g,
0.66 mmol) in CH2CI2 (10 mL). The mixture was stirred at room temperature for 3h before it was
quenched with water and then extracted with CH2CI2 (15 mL x 3). The combined organic layer was dried over anhydrous MgS04 and concentrated under reduced pressure to give a yellow residue, which was then purified by silica gel chromatography (EtOAc: n-hexane = 1 : 3) to afford 14
(0.20 g).
3-(l-Benzenesulfonyl-2,3-dihydro-lH-indol-5-yl)-acrylic acid (15): 1M LiOH aqueous solution (1.16 ml, 1.16 mmol) was added to a solution of 14 (0.20g, 0.58 mmol) in dioxane
(15 mL). The reaction mixture was stirred at 40 °C overnight before it was concentrated under reduced pressure. The residue was dissolved in water and concentrated HCl was added up to acidic pH to give the precipitation, which was dried by vacuum to afford 15 (0.16 g). 1H NMR (500MHz, CD3OD): δ 2.92 (t, J= 8.5 Hz, 2H), 3.96 (t, J= 8.5 Hz, 2H), 6.33 (d, J= 15.9 Hz, 1H), 7.38 (s, 1H), 7.41 (d, J= 8.5 Hz, 1H), 7.50-7.53 (m, 2H), 7.55 (d, J= 16.1 Hz, 1H), 7.58-7.64 (m, 2H), 7.82 (d, J = 7.6 Hz, 2H).
3-(l-Benzenesulfonyl-2,3-dihydro-lH-indol-5-yl)-N-hydroxy-acrylamide
(Compound 12): NH2OTHP (0.05 g, 0.44 mmol) was added to a solution of 15 (0.12 g, 0.37 mmol), PyBOP (0.20 g, 0.39 mmol), triethylamine (0.12 ml, 0.88 mmol) in DMF (1.5 mL). The reaction mixture was stirred at room temperature for 1 h before it was quenched with water, followed by extraction with EtOAc (15 mL x 3). The combined organic layer was dried over anhydrous MgS04 and concentrated under reduced pressure. The residue was purified by silica gel chromatography (CH2C12: CH3OH = 30 : 1 : l%NH3(aq)) to give a white solid, which was treated with TFA (1.13 ml, 15.21 mmol) in the presence of CH3OH (25 mL) and stirred overnight at room temperature. The reaction mixture was concentrated under reduced pressure to give a white residue, which was recrystallized by CH3OH to afford Compound 12 (0.12 g). 1H NMR (500MHz,
CD3OD): δ 2.91 (t, J= 8.5 Hz, 2H), 3.96 (t, J= 8.4 Hz, 2H), 6.32 (d, J= 15.8 Hz, 1H), 7.32 (s, 1H), 7.37-7.39 (m, 1H), 7.46 (d, J= 15.7 Hz, 1H), 7.50-7.53 (m, 2H), 7.58-7.64 (m, 2H), 7.82 (d, J= 7.8 Hz, 2H). MS (EI) mlz: 170 (100%), 344 (M+, 3.21%). HRMS (EI) for Ci7Hi6N204S (M+): calcd, 344.0831; found, 344.0829.
PAT
US20150368195
https://patentscope.wipo.int/search/en/detail.jsf?docId=US154007904&_cid=P11-MQ4M0P-01888-1
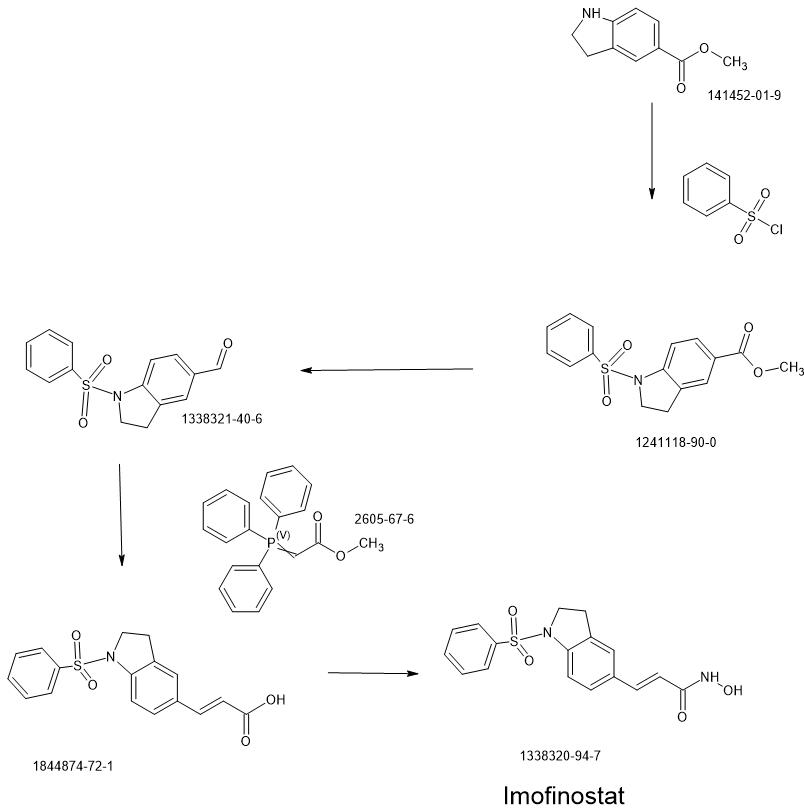
PAT
- Indolyl or indolinyl hydroxamate compoundsPublication Number: US-8846748-B2Priority Date: 2010-03-29Grant Date: 2014-09-30
- Indolyl or indolinyl hydroxamate compoundsPublication Number: US-9598364-B2Priority Date: 2010-03-29Grant Date: 2017-03-21
- Indolyl or indolinyl hydroxamate compoundsPublication Number: WO-2011126821-A2Priority Date: 2010-03-29
- Indolyl or indolinyl hydroxamate compoundsPublication Number: EP-2552887-A2Priority Date: 2010-03-29
- Indolyl or indolinyl hydroxamate compoundsPublication Number: US-2011245315-A1Priority Date: 2010-03-29
- Indolyl or indolinyl hydroxamate compoundsPublication Number: US-2014364477-A1Priority Date: 2010-03-29
- Indolyl or indolinyl hydroxamate compoundsPublication Number: EP-2552887-B1Priority Date: 2010-03-29Grant Date: 2018-10-24
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
//////////imofinostat, anax labs, histone deacetylase inhibitor, antineoplastic, ABT-301, MPT0E028, ABT 301, MPT0E 028, T65L58FI65
Gozanertinib
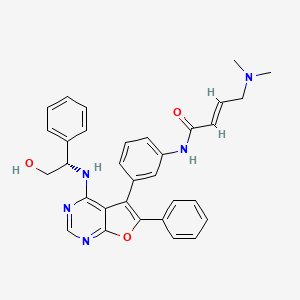
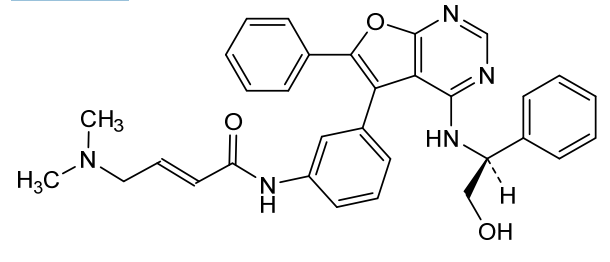
Gozanertinib
CAS 1226549-49-0
MF C32H31N5O3 MW533.6 g/mol
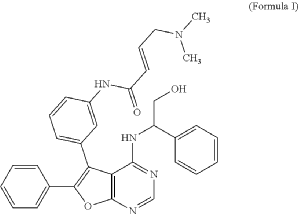
(E)-4-(dimethylamino)-N-[3-[4-[[(1S)-2-hydroxy-1-phenylethyl]amino]-6-phenylfuro[2,3-d]pyrimidin-5-yl]phenyl]but-2-enamide
(2E)-4-(dimethylamino)-N-[3-(4-{[(1S)-2-hydroxy-1-phenylethyl]amino}-6-phenylfuro[2,3-d]pyrimidin-5-yl)phenyl]but-2-
enamide
epidermal growth factor receptor tyrosine kinase inhibitor, antineoplastic, DBPR 112, ABT 101, 6G0COS33K4
Gozanertinib (also known as DBPR112 or ABT-101) is an orally bioavailable, advanced small-molecule dual kinase inhibitor designed to treat advanced non-small cell lung cancer (NSCLC). It targets alterations in the epidermal growth factor receptor (EGFR) and human epidermal growth factor receptor 2 (HER2) families.
Mechanism of Action
Gozanertinib is a furanopyrimidine-based tyrosine kinase inhibitor. It functions by entering the ATP-binding pocket of the receptor and forming an irreversible covalent bond with a specific cysteine residue (Cys797). By permanently blocking these receptors, it halts downstream oncogenic signaling pathways—specifically the RAS/RAF/MEK/ERK and PI3K/AKT cascades—thereby inducing cancer cell death and suppressing tumor expansion.
Target Profile and Key Mutations
Unlike earlier generations of tyrosine kinase inhibitors that only target standard configurations, gozanertinib is optimized to combat specific treatment-resistant mutations:
- EGFR Mutations: It effectively targets wild-type EGFR as well as the dual L858R/T790M resistance mutations.
- Exon 20 Insertions: A standout feature of gozanertinib is its preclinical potency against EGFR and HER2 exon 20 insertion (Ex20ins) mutations. According to chemical development findings published in the Journal of Medicinal Chemistry, it demonstrated ten times better potency against these specific insertions than the widely used third-generation inhibitor, osimertinib.
Development and Status
The drug was initially discovered through scaffold optimization by the National Health Research Institutes (NHRI) and is being co-developed with Anbogen Therapeutics. The International Nonproprietary Name (INN) “gozanertinib” was formally proposed for the compound in early 2025. Preclinical evaluations indicated favorable oral bioavailability and strong anti-tumor efficacy compared to older inhibitors like afatinib, advancing the compound into early-phase clinical trials
Gozanertinib is an orally bioavailable dual kinase inhibitor of epidermal growth factor receptor (EGFR; ErbB1) and human epidermal growth factor receptor 2 (HER2; EGFR2; ErbB2), including EGFR L858R, EGFR T790M and HER2 exon 20 insertion (Ex20ins) mutations, with potential antineoplastic activity. Upon oral administration, gozanertinib targets, binds to and inhibits the activity of EGFR or HER2 insertions or mutations. This prevents EGFR/HER2-mediated signaling, which may induce cell death and inhibit tumor growth in EGFR/HER2-overexpressing tumor cells. The ErbB receptor tyrosine kinase family is involved in key cellular functions, including cell growth and survival. EGFR and HER2 alterations constitutively upregulate kinase activity.
- Phase 1b/2 Study to Evaluate ABT-101 in Solid Tumor and NSCLC PatientsCTID: NCT05532696Phase: Phase 1/Phase 2Status: RecruitingDate: 2024-06-24
- A Study of DBPR112 in Patients With Head and Neck Cancer and EGFR Mutated Lung CancerCTID: NCT03246854Phase: Phase 1Status: TerminatedDate: 2020-12-17
PAT
PAT
https://patentscope.wipo.int/search/en/detail.jsf?docId=US43249513&_cid=P11-MQ1QG3-86325-1
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Development of Furanopyrimidine-Based Orally Active Third-Generation EGFR Inhibitors for the Treatment of Non-Small Cell Lung CancerPublication Name: Journal of Medicinal ChemistryPublication Date: 2023-02-07PMCID: PMC9969398PMID: 36749735DOI: 10.1021/acs.jmedchem.2c01434
- Discovery of a Furanopyrimidine-Based Epidermal Growth Factor Receptor Inhibitor (DBPR112) as a Clinical Candidate for the Treatment of Non-Small Cell Lung CancerPublication Name: Journal of Medicinal ChemistryPublication Date: 2019-09-27PMID: 31560541DOI: 10.1021/acs.jmedchem.9b00722
PAT
- Active cancer immunotherapy by immune modulation via globo series antigensPublication Number: EP-4248214-A1Priority Date: 2020-11-19
- Fused Bicyclic and Tricyclic Pyrimidine Compounds as Tyrosine Kinase InhibitorsPublication Number: US-2010120805-A1Priority Date: 2008-11-10
- Fused bicyclic and tricyclic pyrimidine compounds as tyrosine kinase inhibitorsPublication Number: US-8507502-B2Priority Date: 2008-11-10Grant Date: 2013-08-13
- Fused bicyclic and tricyclic pyrimidine compounds as tyrosine kinase inhibitorsPublication Number: WO-2010054285-A2Priority Date: 2008-11-10
- Fused bicyclic and polycyclic pyrimidine compounds as tyrosine kinase inhibitorsPublication Number: CN-102264745-APriority Date: 2008-11-10
- Active cancer immunotherapy through immune modulation via GLOBO series antigensPublication Number: CN-116847875-APriority Date: 2020-11-19
- Active cancer immunotherapy by immune modulation via globo series antigensPublication Number: CA-3200572-A1Priority Date: 2020-11-19
- Active cancer immunotherapy by immune modulation via globo series antigensPublication Number: WO-2022109601-A1Priority Date: 2020-11-19
- Active cancer immunotherapy by immune modulation via globo series antigensPublication Number: IL-302947-APriority Date: 2020-11-19
- Active cancer immunotherapy by immunomodulation through GLOBO family antigensPublication Number: KR-20230110529-APriority Date: 2020-11-19
- Crystalline forms of (s, e)-4-(dimethylamino)-n-(3-(4-(2-hydroxy-1-phenylethylamino)-6-phenylfuro[2,3-d]pyrimidin-5-yl)phenyl)but-2-enamide free basePublication Number: TW-I809967-BPriority Date: 2021-07-06Grant Date: 2023-07-21
- Crystalline forms of (S, E)-4-(dimethylamino)-N-(3-(4-(2-hydroxy-1-phenylethylamino)-6-phenylfuro[2,3-d]pyrimidin-5-yl)phenyl)but-2- enamide free basePublication Number: US-12240858-B2Priority Date: 2021-07-06Grant Date: 2025-03-04
- Active cancer immunotherapy by immune modulation via globo series antigensPublication Number: TW-202237177-APriority Date: 2020-11-19
- Active cancer immunotherapy by immune modulation via globo series antigensPublication Number: US-2024139301-A1Priority Date: 2020-11-19
- Active cancer immunotherapy by immune modulation via globo series antigensPublication Number: AU-2021382807-A1Priority Date: 2020-11-19
- Crystalline forms of (s, e)-4-(dimethylamino)-n-(3-(4-(2-hydroxy-1-phenylethylamino)-6-phenylfuro[2,3-d]pyrimidin-5-yl)phenyl)but-2-enamide free basePublication Number: US-2023021909-A1Priority Date: 2021-07-06
- Crystalline forms of (s, e)-4-(dimethylamino)-n-(3-(4-(2-hydroxy-1-phenylethylamino)-6-phenylfuro[2,3-d]pyrimidin-5-yl)phenyl)but-2-enamide free basePublication Number: WO-2023283269-A1Priority Date: 2021-07-06
- Crystalline forms of (s, e)-4-(dimethylamino)-n-(3-(4-(2-hydroxy-1-phenylethylamino)-6-phenylfuro[2,3-d]pyrimidin-5-yl)phenyl)but-2-enamide free basePublication Number: US-2024368175-A1Priority Date: 2021-07-06
- Crystalline forms of (s, e)-4-(dimethylamino)-n-(3-(4-(2-hydroxy-1-phenylethylamino)-6-phenylfuro[2,3-d]pyrimidin-5-yl)phenyl)but-2-enamide free basePublication Number: TW-202309041-APriority Date: 2021-07-06
- Crystalline forms of (s, e)-4-(dimethylamino)-n- (3-(4-(2-hydroxy-1-phenylethylamino)-6-phenylfuro[2,3-d]pyrimidin-5-yl)phenyl)but-2-enamide free basePublication Number: EP-4330259-A1Priority Date: 2021-07-06
//////gozanertinib, ANAX LABS, epidermal growth factor receptor tyrosine kinase inhibitor, antineoplastic, DBPR 112, ABT 101, 6G0COS33K4
Gintemetostat
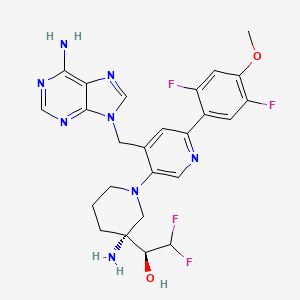
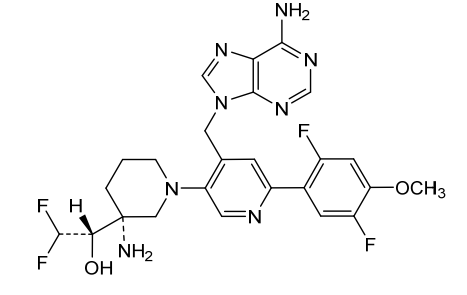
Gintemetostat
(1S)-1-[(3R)-3-amino-4′-[(6-amino-9H-purin-9-yl)methyl]-6′-(2,5-difluoro-4-methoxyphenyl)-3,4,5,6-tetrahydro-2H-[1,3′-bipyridin]-3-yl]-2,2-difluoroethan1-ol
antineoplastic, KTX 1001, NSD2 inhibitor 161, A48CGJ5UQM
CAS 2604513-16-6
MF C25H26F4N8O2 MW 546.5 g/mol
(S)-1-((R)-3-Amino-1-(4-((6-amino-9H-purin-9-yl)methyl)-6-(2,5-difluoro-4-methoxyphenyl)pyridin-3-yl)piperidin-3-yl)-2,2-difluoroethan-1-ol
Gintemetostat (also known as KTX-1001) is a first-in-class, orally administered small molecule being developed to treat relapsed and refractory multiple myeloma. It works as a selective inhibitor of NSD2 (also known as MMSET), targeting the epigenetic drivers of high-risk cancers.
How it Works
- Mechanism: Gintemetostat selectively binds to the catalytic SET domain of the NSD2 enzyme.
- Effect: By blocking this enzyme, it downregulates oncogenic signaling, decreases cancer cell growth, and can enhance T-cell activation against the tumor.
Target Patient Population
- High-Risk Myeloma: The drug focuses heavily on patients harboring the t(4;14) translocation, a genetic alteration found in 10-15% of patients that often causes aggressive relapses.
- Refractory Cases: It has shown notable single-agent activity in heavily pretreated patients who have exhausted standard-of-care, triple-class refractory treatment options.
Current Clinical Status
- Phase 1 Trial: Early data from phase 1 trials (such as NCT05651932) showed the drug has manageable safety profiles and offers clinical benefit (ranging from stable disease to very good partial response) in patients with aggressive, hard-to-treat multiple myeloma.
- Future Developments: Researchers are expanding studies to pair gintemetostat with other standard myeloma treatments, such as proteasome inhibitors and CELMoDs, to create stronger synergistic anti-cancer effects.
Gintemetostat is an orally available small molecule inhibitor of the histone-lysine N-methyltransferase nuclear receptor-binding SET domain protein 2 (NSD2; MMSET; WHSC1), with potential antineoplastic activity. Upon oral administration, gintemetostat selectively targets and binds to NSD2, and inhibits its catalytic activity and the mono- and di-methylation of histone H3 lysine 36 (H3K36). This modulates the expression of genes involved in cellular processes including cellular proliferation, which may lead to decreased growth of cancer cells. NSD2, a member of the NSD family of histone lysine methyltransferase enzymes that catalyzes the mono- and di-methylation of H3K36, is overexpressed and dysregulated in many types of cancers.
SYN
Discovery of a Highly Potent and Selective Inhibitor Targeting Protein Lysine Methyltransferase NSD2
Publication Name: Journal of Medicinal Chemistry
Publication Date: 2024-09-04
PMID: 39230932
DOI: 10.1021/acs.jmedchem.4c00639
SYN
PAT
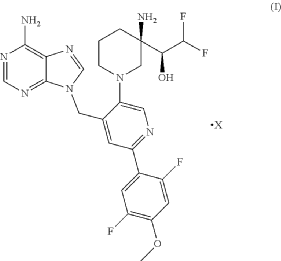
PAT
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2021028854&_cid=P12-MQ0AZT-13511-1
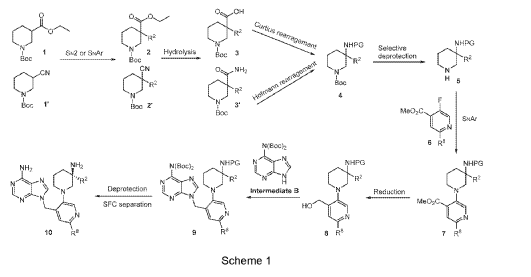
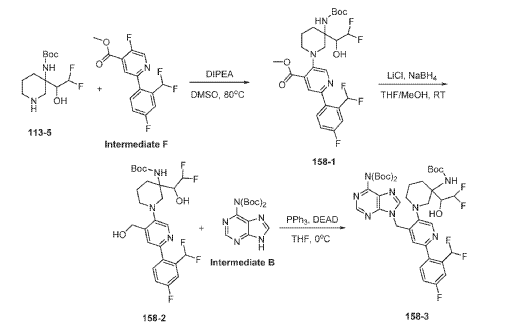
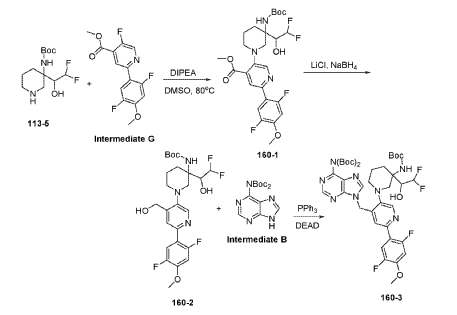
Example 160 and Example 161: (R)-1-((R)-3-amino-1-(4-((6-amino-9H-purin-9-yl)methyl)-6- (2,5-difluoro-4-methoxyphenyl)pyridin-3-yl)piperidin-3-yl)-2,2-difluoroethan-1-ol and (S)-1-((R)-3- amino-1-(4-((6-amino-9H-purin-9-yl)methyl)-6-(2,5-difluoro-4-methoxyphenyl)pyridin-3- yl)piperidin-3-yl)-2,2-difluoroethan-1-ol
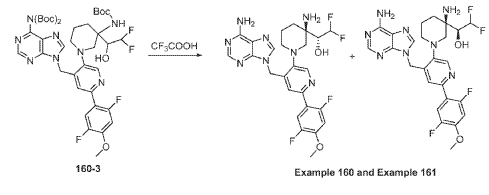
To a solution of tert-butyl (tert-butoxycarbonyl)(9-((5-(3-((tert-butoxycarbonyl)amino)-3-(2,2- difluoro-1-hydroxyethyl)piperidin-1-yl)-2-(2,5-difluoro-4-methoxyphenyl)pyridin-4-yl)methyl)-9H- purin-6-yl)carbamate (Intermediate 160-3) (200 mg, 0.237 mmol) in DCM (18 mL), was added TFA (36 mL), and the reaction mixrture was stirred at rt for 30 min under N2 atmosphere. The reaction mixture was concentrated in vacuo to give the crude product. The crude product was purifed by Pre-HPLC and SFC to afford (R)-1-((R)-3-amino-1-(4-((6-amino-9H-purin-9- yl)methyl)-6-(2,5-difluoro-4-methoxyphenyl)pyridin-3-yl)piperidin-3-yl)-2,2-difluoroethan-1-ol (Example 160) and (S)-1-((R)-3-amino-1-(4-((6-amino-9H-purin-9-yl)methyl)-6-(2,5-difluoro-4- methoxyphenyl)pyridin-3-yl)piperidin-3-yl)-2,2-difluoroethan-1-ol (Example 161).
Example 160: 1H NMR (400 MHz, CD3OD) d ppm 8.48 (s, 1H), 8.20 (d, J = 1.6 Hz, 2H), 7.58 (dd, J = 12.2, 7.3 Hz, 1H), 7.11 (d, J = 1.3 Hz, 1H), 6.90 (dd, J = 12.6, 7.1 Hz, 1H), 6.06 (td, J = 55.1, 3.9 Hz, 1H), 5.67 (s, 2H), 3.87 (s, 3H), 3.75 – 3.58 (m, 1H), 3.25 – 2.75 (m, 4H), 2.26 – 1.60 (m, 4H). LC-MS: [M+H]+ = 547.2, 548.2.
Example 161: 1H NMR (400MHz, CD3OD) d = 8.51 – 8.44 (m, 1H), 8.24 – 8.16 (m, 2H), 7.62 – 7.48 (m, 1H), 7.03 (s, 1H), 6.93 – 6.79 (m, 1H), 6.25 – 5.86 (m, 1H), 5.71 – 5.59 (m, 2H), 4.00 (m, 1H), 3.88 – 3.80 (m, 3H), 3.28 – 2.87 (m, 4H), 1.99 – 1.56 (m, 4H). LC-MS: [M+H]+ =547.4.
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Piperidinyl-methyl-purineamines as nsd2 inhibitors and anti-cancer agentsPublication Number: EP-4559915-A1Priority Date: 2019-08-14
- Piperidinyl-methyl-purineamines as nsd2 inhibitors and anti-cancer agentsPublication Number: EP-4013755-B1Priority Date: 2019-08-14Grant Date: 2025-01-08
- Piperidinyl-methyl-purinamines as NSD2 inhibitors and anticancer agentsPublication Number: CN-114585622-APriority Date: 2019-08-14
- Piperidinyl-methyl-purineamines as NSD2 inhibitors and anti-cancer agentsPublication Number: US-12312353-B2Priority Date: 2019-08-14Grant Date: 2025-05-27
- Piperidinyl-methyl-purineamines as nsd2 inhibitors and anti-cancer agentsPublication Number: WO-2021026803-A1Priority Date: 2019-08-14
- Piperidinyl-methyl-purineamines as nsd2 inhibitors and anti-cancer agentsPublication Number: EP-4013755-A1Priority Date: 2019-08-14
- Piperidinyl-methyl-purineamines as nsd2 inhibitors and anti-cancer agentsPublication Number: WO-2021028854-A1Priority Date: 2019-08-14
- Piperidinyl-methyl-purineamine D-tartrate, crystalline forms, and use thereof in the treatment of medical diseases and conditionsPublication Number: CN-119744262-APriority Date: 2022-05-18
- Piperidinyl-methyl-purine amine d-tartaric acid salts, crystalline forms, and their use in treating medical diseases and conditionsPublication Number: EP-4526305-A1Priority Date: 2022-05-18
- Piperidinyl-methyl-purineamines as nsd2 inhibitors and anti-cancer agentsPublication Number: US-2023002388-A1Priority Date: 2019-08-14
- Piperidinyl-methyl-purinamines as NSD2 inhibitors and anticancer agentsPublication Number: CN-114585622-BPriority Date: 2019-08-14Grant Date: 2024-08-09
- Piperidinyl-methyl-purineamines as NSD2 inhibitors and anti-cancer agentsPublication Number: US-11420970-B1
- Piperidinyl-methyl-purine amine d-tartaric acid salts, crystalline forms, and their use in treating medical diseases and conditionsPublication Number: AU-2023273656-A1Priority Date: 2022-05-18
- Piperidinyl-methyl-purine amine salts, crystalline forms, and their use in treating medical diseases and conditionsPublication Number: WO-2023225144-A1Priority Date: 2022-05-18
- Piperidinyl-methyl-purine amine fumaric acid salts, crystalline forms, and their use in treating medical diseases and conditionsPublication Number: WO-2023225150-A1Priority Date: 2022-05-18
- Piperidinyl-methyl-purine amine salts, crystalline forms, and their use in treating medical diseases and conditionsPublication Number: US-2025326752-A1Priority Date: 2022-05-18
- Piperidinyl-methyl-purinic amine D-tartrate salt, crystalline form and their use in the treatment of medical diseases and conditionsPublication Number: KR-20250012083-APriority Date: 2022-05-18
- Pharmaceutical compositions containing a piperidinyl-methyl-purine amine and their use in treating diseases and conditionsPublication Number: US-2024207192-A1Priority Date: 2022-12-12
- Pharmaceutical compositions containing a piperidinyl-methyl-purine amine and their use in treating diseases and conditionsPublication Number: WO-2024129670-A1Priority Date: 2022-12-12
- Piperidinyl-methyl-purine amine d-tartaric acid salts, crystalline forms, and their use in treating medical diseases and conditionsPublication Number: US-2024002385-A1Priority Date: 2022-05-18
- Piperidinyl-methyl-purine amine salts, crystalline forms, and their use in treating medical diseases and conditionsPublication Number: WO-2023225154-A1Priority Date: 2022-05-18
- Piperidinyl-methyl-purine amine d-tartaric acid salts, crystalline forms, and their use in treating medical diseases and conditionsPublication Number: WO-2023225141-A1Priority Date: 2022-05-18
////////////gintemetostat, ANAX LABS, antineoplastic, KTX 1001, NSD2 inhibitor 161, A48CGJ5UQM
Epaldeudomide
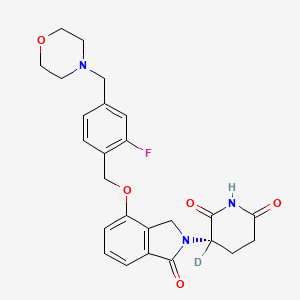
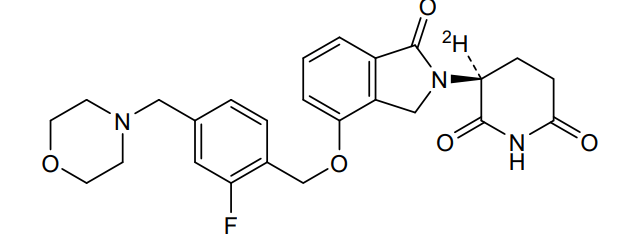
Epaldeudomide
CAS 1918159-31-5
MF C25H252HFN3O5, MW 468.5 g/mol
(3S)-3-deuterio-3-[7-[[2-fluoro-4-(morpholin-4-ylmethyl)phenyl]methoxy]-3-oxo-1H-isoindol-2-yl]piperidine-2,6-dione
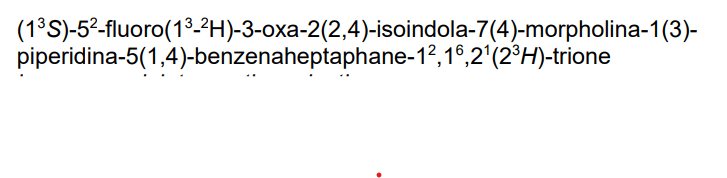
KPG-818, KPG 818, ANTINEOPLASTIC, KV0TBL8MUS
Epaldeudomide (also known as KPG-818) is an investigational, next-generation immunomodulatory drug and “molecular glue” developed by Kangpu Biopharmaceuticals. Designed as a targeted therapy, it works by binding to the CRL4-CRBN E3 ubiquitin ligase complex to degrade specific proteins, showing promise in treating blood cancers, solid tumors, and autoimmune diseases.
Mechanism of Action
- Molecular Glue: It is a small molecule that acts as a modulator of the cereblon (CRBN) E3 ligase.
- Protein Degradation: It targets and induces the rapid degradation of two Ikaros zinc-finger transcription factors: IKZF3 (Aiolos) and IKZF1 (Ikaros).
- Immunomodulation: By degrading these targets, epaldeudomide triggers broad-spectrum immune responses, reduces tumor proliferation, and suppresses inflammation (such as the production of TNF-\(\alpha \)).
Therapeutic Pipeline and Research
Epaldeudomide is currently undergoing clinical evaluation to assess its safety, tolerability, and efficacy.
- Hematology/Oncology: It is being studied for the treatment of hematologic malignancies (such as multiple myeloma and lymphomas). It demonstrates potent anti-tumor and anti-angiogenic activity without several severe side effects typically associated with earlier immunomodulatory drugs.
- Autoimmune and Inflammatory Disorders: Because of its broad anti-inflammatory effects and ability to inhibit TNF-\(\alpha \), it is being explored for use against autoimmune conditions and inflammatory arthritis.
- OriginatorKangpu Biopharmaceuticals
- ClassAnti-inflammatories; Antineoplastics; Small molecules
- Mechanism of ActionCRBN protein modulators; Ubiquitin protein ligase complex modulators
- Phase IIInflammatory bowel diseases
- Phase I/IISystemic lupus erythematosus
- Phase IHaematological malignancies
- PreclinicalBehcet’s syndrome; Crohn’s disease; Multiple myeloma
- 06 Dec 2025Efficacy, pharmacokinetics and adverse events data from a phase I trial in Haematological malignancies presented at 67th American Society of Hematology Annual Meeting and Exposition (ASH-Hem-2025)
- 26 Nov 2025Epaldeudomide is still in phase I trials for Haematological malignancies (Second-line therapy or greater) in USA (PO, Capsule) (NCT04283097)
- 18 Nov 2025Efficacy and adverse events data from a phase I trial in Haematological malignancies released by Kangpu Biopharmaceuticals
SYN
US-10017492-B2
US-20170313676-A1
SYN
EP-3643709-A1
EP-3643709-B1
https://patentscope.wipo.int/search/en/detail.jsf?docId=EP293972088&_cid=P21-MPEVL7-37300-1
Example 37: Compound A382
[0196] 3-(4-((2-fluoro-5-(3-morpholinopropoxy)benzyl)amino)-1-oxoisoindolin-2-yl)piperidine-2,6-dione, A382.
[0197] 1H NMR (DMSO- d 6, 300 MHz): δ 11.00 (s, 1H), 7.32 (t, J = 7.8 Hz, 1H), 7.22 (t, J = 7.8 Hz, 1H), 7.05-7.13 (m, 2H), 6.93 (d, J = 7.5 Hz, 1H), 6.64 (d, J = 7.8 Hz, 1H), 6.28 (t, J = 6.3 Hz, 1H), 5.07-5.13 (m, 1H), 4.38 (d, J= 5.7 Hz, 2H), 4.28 (d, J= 17.4 Hz, 1H), 4.16 (d, J= 17.4 Hz, 1H), 3.54 (t, J= 4.5 Hz, 4H), 3.42 (s, 2H), 2.85-2.97 (m, 1H), 2.57-2.63 (m, 1H), 2.26-2.38 (m, 5H), 2.00-2.09 (m, 1H). LCMS: 467.2 ([M+1] +).
Example 69: Compound A406
[0296] ( S)-3-deuterium-3-(4-((2-fluoro-4-(morpholinomethyl)benzyl)oxy)-1-oxoisoindolin-2-yl)piperidine-2,6-dione, A406.
[0297] 1H NMR (DMSO- d 6, 300 MHz): δ 10.98 (s, 1H),7.47-7.55 (m, 2H), 7.31-7.38 (m, 2H), 7.16-7.20 (m, 2H), 5.24 (s, 2H), 5.06-5.12 (m, 0.04H), 4.35 (d, J = 18.0 Hz, 1H), 4.19 (d, J = 18.0 Hz, 1H), 3.55 (br, 4H), 3.47 (s, 2H), 2.82-2.94 (m, 1H), 2.48-2.57 (m, 1H), 2.33-2.42 (m, 5H), 1.91-1.96 (m, 1H). LCMS: 469.2 ([M+1] +).
ADVT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Isoindoline derivative, intermediate, preparation method, pharmaceutical composition and use thereofPublication Number: US-10017492-B2Priority Date: 2014-10-30Grant Date: 2018-07-10
- Isoindoline derivative, intermediate, preparation method, pharmaceutical composition and use thereofPublication Number: EP-3643709-A1Priority Date: 2014-10-30
- Isoindoline derivative, intermediate, preparation method, pharmaceutical composition and use thereofPublication Number: US-2017313676-A1Priority Date: 2014-10-30
- Isoindoline derivative, intermediate, preparation method, pharmaceutical composition and use thereofPublication Number: EP-3643709-B1Priority Date: 2014-10-30Grant Date: 2021-10-20
/////////epaldeudomide, ANAX LABS, KPG-818, KPG 818, ANTINEOPLASTIC, KV0TBL8MUS
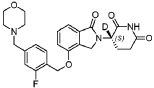
Enozertinib
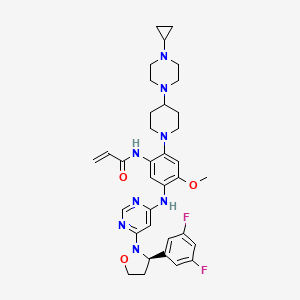
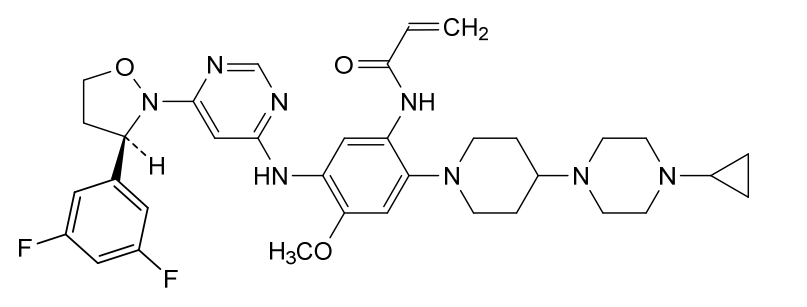
Enozertinib
CAS 2489185-38-6
MF C35H42F2N8O3 MW660.8
N-[2-[4-(4-cyclopropylpiperazin-1-yl)piperidin-1-yl]-5-[[6-[(3R)-3-(3,5-difluorophenyl)-1,2-oxazolidin-2-yl]pyrimidin-4-yl]amino]-4-methoxyphenyl]prop-2-enamide
- N-(2-(4-(4-cyclopropylpiperazine-1-yl)piperidine-1-yl)-5-((6-((R)-3-(3,5-difluorophenyl)isoxazolidine-2-yl)pyrimidine-4-yl)amino)-4-methoxyphenyl)acrylamide
- N-[2-[4-(4-Cyclopropyl-1-piperazinyl)-1-piperidinyl]-5-[[6-[(3R)-3-(3,5-difluorophenyl)-2-isoxazolidinyl]-4-pyrimidinyl]amino]-4-methoxyphenyl]-2-propenamide
- N-[2-[4-(4-cyclopropylpiperazin-1-yl)piperidin-1-yl]-5-[[6-[(3R)-3-(3,5-difluorophenyl)-1,2-oxazolidin-2-yl]pyrimidin-4-yl]amino]-4-methoxyphenyl]prop-2-enamide
epidermal growth factor receptor tyrosine kinase inhibitor, antineoplastic, ORIC-114, ORIC 114, DU24UP8R94
Enozertinib (formerly ORIC-114) is an investigational, orally bioavailable, and brain-penetrant dual EGFR/HER2 inhibitor developed by ORIC Pharmaceuticals. It targets cancers with exon 20 insertion and atypical EGFR mutations. Its core profile highlights its ability to cross the blood-brain barrier.
How it Works
Enozertinib acts as an irreversible, mutant-selective covalent inhibitor. By blocking overactive EGFR and HER2 signaling, it induces cell death and inhibits tumor growth. Because it penetrates the central nervous system (CNS), it is uniquely suited to treat both primary brain tumors and brain metastases—a common complication in non-small cell lung cancer (NSCLC).
Enozertinib is an orally bioavailable, central nervous system (CNS) penetrating, mutant-selective covalent inhibitor of epidermal growth factor receptor (EGFR; ErbB1) and human epidermal growth factor receptor 2 (HER2; EGFR2; ErbB2) alterations, including exon 20 insertion (Ex20ins) mutations, with potential antineoplastic activity. Upon oral administration, enozertinib selectively targets, irreversibly binds to and inhibits the activity of EGFR or HER2 insertions or mutations. This prevents EGFR/HER2-mediated signaling. This may induce cell death and inhibit tumor growth in EGFR/HER2-overexpressing tumor cells. Enozertinib is able to penetrate the blood-brain-barrier (BBB) and may therefore exert its activity against EGFR Ex20ins-driven CNS primary tumors and CNS metastases. The ErbB receptor tyrosine kinase family is involved in key cellular functions, including cell growth and survival. EGFR and HER2 alterations constitutively upregulate kinase activity.
SYN
https://drughunter.com/molecule/enozertinib-oric-114
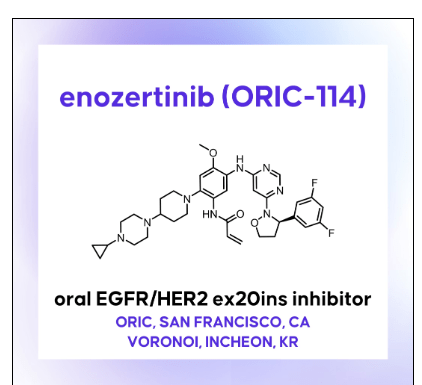
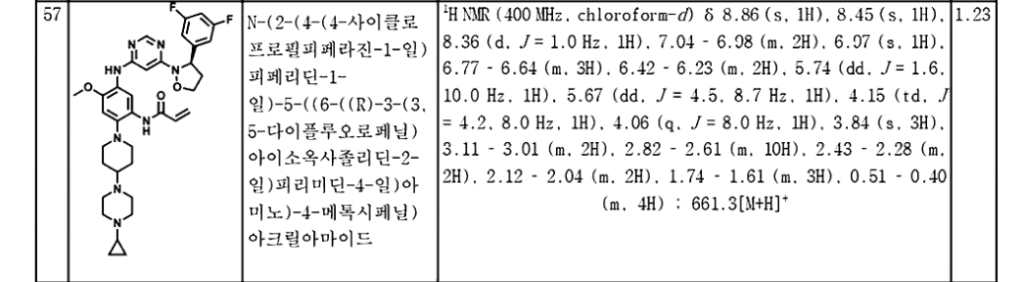
SYN

PAT
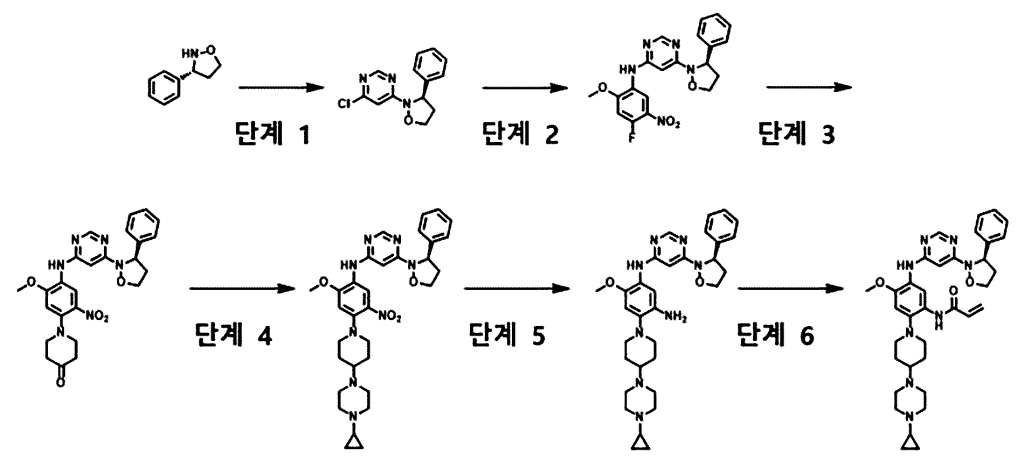
SIMILAR SYNTHESIS
ADVT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Egfr inhibitor for treating cancers comprising atypical egfr mutationsPublication Number: WO-2024233313-A1Priority Date: 2023-05-05
- Fumarate, tartrate, malate, and citrate salts of an egfr inhibitorPublication Number: WO-2024096624-A1Priority Date: 2022-11-03
- Malonate and glycolate salts of an egfr inhibitorPublication Number: WO-2024097848-A1Priority Date: 2022-11-03
- Malonate and glycolate salts of an egfr inhibitorPublication Number: EP-4611902-A1Priority Date: 2022-11-03
- Fumarate, tartrate, malate, and citrate salts of an egfr inhibitorPublication Number: EP-4612147-A1Priority Date: 2022-11-03
- Fumarate, tartrate, malate and citrate salts of EGFR inhibitorsPublication Number: CN-120035590-APriority Date: 2022-11-03
- Heteroaryl derivative, method for producing same, and pharmaceutical composition comprising same as effective componentPublication Number: US-11466000-B2Priority Date: 2019-03-19Grant Date: 2022-10-11
- Heteroaryl derivatives, their preparation methods, and pharmaceutical compositions containing them as active ingredientsPublication Number: CN-115838369-APriority Date: 2019-03-19
- Heteroaryl derivatives, methods for their preparation, and pharmaceutical compositions containing them as active ingredientsPublication Number: CN-114605400-APriority Date: 2019-03-19
- HETEROARYL DERIVATIVE, METHOD FOR PRODUCING THE SAME, AND PHARMACEUTICAL COMPOSITION INCLUDING THE SAME AS AN EFFECTIVE COMPONENTPublication Number: BR-112021018704-B1Priority Date: 2019-03-19
- Heteroaryl derivative, method for producing same, and pharmaceutical composition comprising same as effective componentPublication Number: US-2022289733-A1Priority Date: 2019-03-19
- Heteroaryl derivatives, methods for producing heteroaryl derivatives, and pharmaceutical compositions containing heteroaryl derivatives as active ingredientsPublication Number: JP-7394298-B2Priority Date: 2019-03-19Grant Date: 2023-12-08
- Heteroaryl derivatives, methods for their preparation, and pharmaceutical compositions containing them as active ingredientsPublication Number: CN-113993866-APriority Date: 2019-03-19
//////////enozertinib, anax labs, epidermal growth factor receptor tyrosine kinase inhibitor, antineoplastic, ORIC-114, ORIC 114, DU24UP8R94
Emupertinib
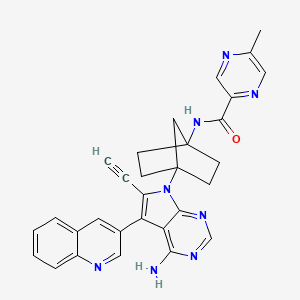
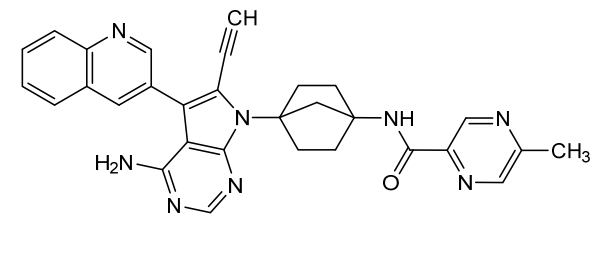
Emupertinib
CAS 2472802-77-8
MFC30H26N8O MW514.6 g/mol
2-Pyrazinecarboxamide, N-[4-[4-amino-6-ethynyl-5-(3-quinolinyl)-7H-pyrrolo[2,3-d]pyrimidin-7-yl]bicyclo[2.2.1]hept-1-yl]-5-methyl-
N-{4-[4-amino-6-ethynyl-5-(quinolin-3-yl)-7Hpyrrolo[2,3-d]pyrimidin-7-yl]bicyclo[2.2.1]heptan-1-yl}-
5-methylpyrazine-2-carboxamide
epidermal growth factor receptor tyrosine kinase, inhibitor, antineoplastic, TAS3351, TAS 3351, CU9YW8A5TP
Emupertinib is a potent, small-molecule epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor. It possesses selective antineoplastic potential for targeting specific mutant profiles of cancer cells. The compound was originally developed by Taiho Pharmaceutical Co., Ltd. under the developmental code TAS3351
Development Profile
The International Nonproprietary Name (INN) for this therapeutic chemical structure was formally proposed under the World Health Organisation (WHO) proposed INN list 132 in early 2025. Global research pipelines list the compound’s structural classification profile within non-small cell lung cancer (NSCLC) primary discovery programs. The drug currently remains a specialized compound designated for global laboratory research use only, rather than standard human prescription or veterinary clinical treatments
SYN
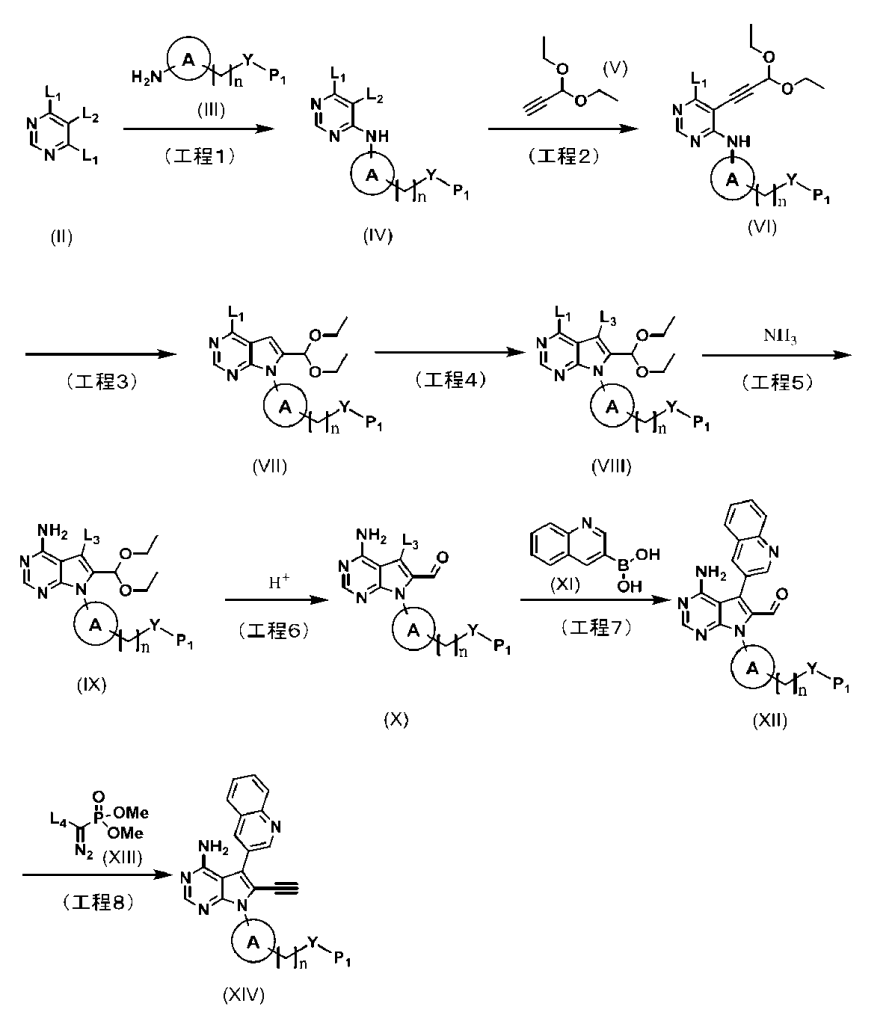
[0184][Example 37]
N-(4-(4-amino-6-ethynyl-5-(quinoline-3-yl)-7H-pyrrolo[2,3-d]pyrimidine-7-yl)bicyclo[2.2.1]heptan-1-yl)
-5-methylpyrazine-2-carboxamide The title compound was obtained by following the same method as in Example 29 (step 6), except that 5-methylpyrazine-2-carboxylic acid was used instead of 5-(fluoromethyl)-2-methylpyrazole-3-carboxylic acid used in Example 29.
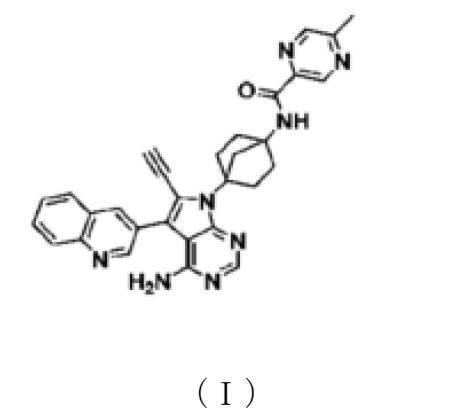
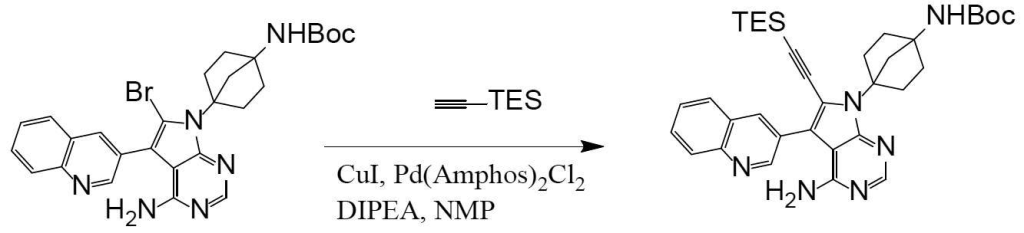
(Step 4)
Synthesis of N-(4-(4-amino-6-ethynyl-5-(quinoline-3-yl)-7H-pyrrolo[2,3-d]pyrimidine-7-yl)bicyclo[2.2.1]heptan-1-yl)-5-methylpyrazine-2-carboxamide (compound (1))
[Chemical Formula 7]
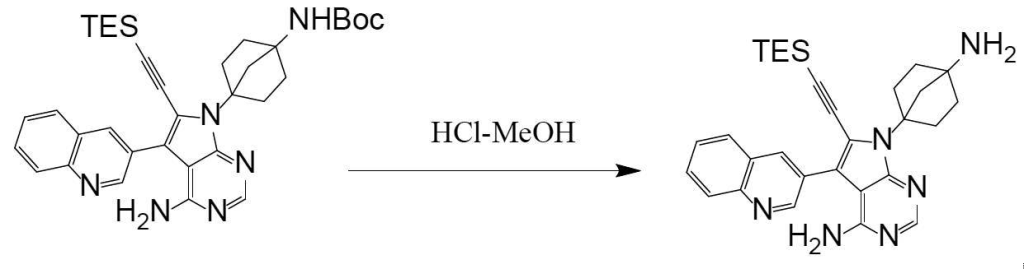
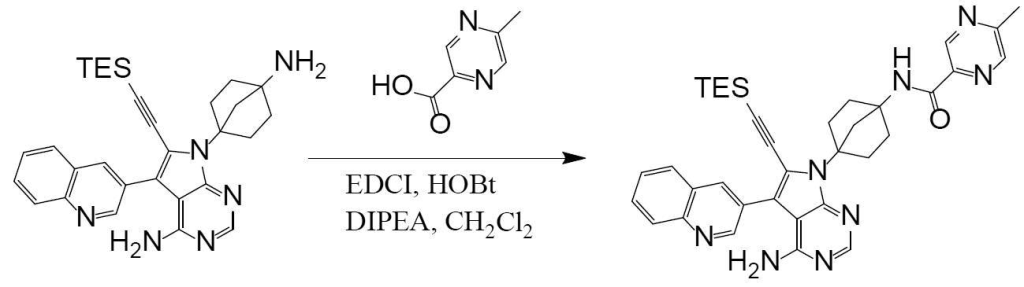
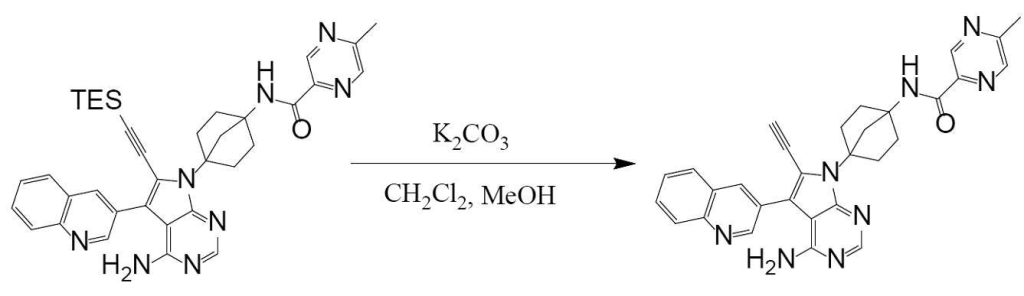
It can be obtained by deprotecting the acetylene protecting group TES of N-(4-(4-amino-5-(quinoline-3-yl)-6-((triethylsilyl)ethynyl)-7H-pyrrolo[2,3-d]pyrimidine-7-yl)bicyclo[2.2.1]heptan-1-yl)-5-methylpyrazine-2-carboxamide obtained in Step 3 under basic conditions.
The reagents used to create basic conditions are not particularly limited as long as the reaction proceeds, but examples of inorganic bases include metal hydroxides (sodium hydroxide, calcium hydroxide, etc.), metal hydrides (lithium hydride, sodium hydride, etc.), and metal carbonates (sodium carbonate, potassium carbonate, cesium carbonate, calcium carbonate, lithium carbonate, magnesium carbonate, sodium bicarbonate, etc.). Examples of organic bases include metal alkoxides (sodium methoxide, potassium tert-butoxide, etc.), metal amides (sodium amide, lithium diisopropylamide, etc.), alkyl metal compounds (n-butyllithium, trimethylaluminum, etc.), alkylamines (triethylamine, tetramethylethylenediamine, piperidine, 1,4-diazabicyclo[2.2.2]octane, etc.), heterocyclic amines (diazabicycloundecene, pyridine, imidazole, etc.), and quaternary ammonium fluorides (tetra-n-butylammonium fluoride). Preferably, the reagent used to create basic conditions is a reagent that does not contain fluoride ions, more preferably a metal carbonate, and even more preferably potassium carbonate. These can be used alone or in combination to adjust the pH to the desired level.
The amount of reagent used is not particularly limited as long as the reaction proceeds, but for example, 0.1 to 50 moles can be used per mole of the starting compound (the compound represented by formula (II)). Preferably, 0.1 to 10 moles, and more preferably 0.1 to 2 moles.
ADVT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
Substituted pyrrolo[2,3-d]pyrimidines as EGFR inhibitors
Publication Number: US-11786534-B2
Priority Date: 2019-02-15
Grant Date: 2023-10-17
- Crystal of 7h-pyrrolo[2,3-d]pyrimidine-4-amine derivativePublication Number: EP-4512808-A1Priority Date: 2022-04-22
- 7H-pyrrolo[2,3-d]pyrimidin-4-amine derivativePublication Number: KR-102645237-B1Priority Date: 2019-02-15Grant Date: 2024-03-07
- 7h-pyrrolo[2,3-d]pyrimidine-4-amine derivativePublication Number: WO-2020166680-A1Priority Date: 2019-02-15
- 7h-pyrrolo[2,3-d]pyrimidine-4-amine derivativePublication Number: US-2022160719-A1Priority Date: 2019-02-15
- 7H-Pyrrolo[2,3-d]pyrimidin-4-amine derivativesPublication Number: CN-113453764-BPriority Date: 2019-02-15Grant Date: 2024-04-16
- Brain-migrating tumor treatment agent containing, as active ingredient, n-(4-(4-amino-6-ethynyl-5-(quinolin-3-yl)-7h-pyrrolo[2,3-d]pyrimidin-7-yl) bicyclo[2.2.1]heptan-1-yl)-5-methylpyrazine-2-carboxamide or salt thereofPublication Number: WO-2025127108-A1Priority Date: 2023-12-13
- Crystals of 7h-pyrrolo[2,3-d]pyrimidin-4-amine derivativesPublication Number: WO-2025072720-A1Priority Date: 2023-09-29
- Crystal of 7h-pyrrolo[2,3-d]pyrimidine-4-amine derivativePublication Number: WO-2023204303-A1Priority Date: 2022-04-22
- Method for producing 7h-pyrrolo[2,3-d]pyrimidine-4-amine derivativePublication Number: WO-2023204304-A1Priority Date: 2022-04-22
- Method for producing 7h-pyrrolo[2,3-d]pyrimidine-4-amine derivativePublication Number: US-2025270214-A1Priority Date: 2022-04-22
///////emupertinib, anax labs, epidermal growth factor receptor tyrosine kinase, inhibitor, antineoplastic, TAS3351, TAS 3351, CU9YW8A5TP
Elisrasib
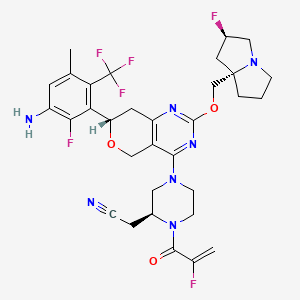
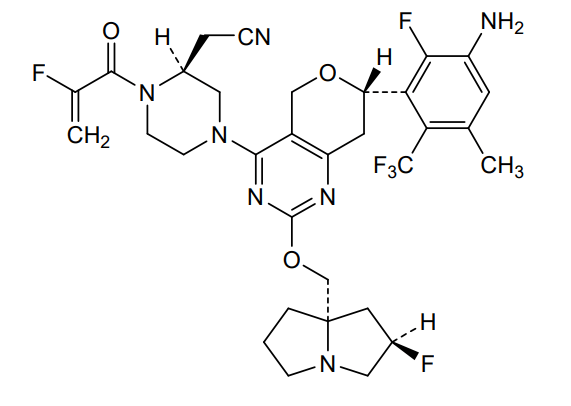
Elisrasib
CAS2914919-85-8
MFC32H35F6N7O3. MW 679.7 g/mol
2-[(2S)-4-[(7S)-7-[3-amino-2-fluoro-5-methyl-6-(trifluoromethyl)phenyl]-2-[[(2R,8S)-2-fluoro-1,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]-7,8-dihydro-5H-pyrano[4,3-d]pyrimidin-4-yl]-1-(2-fluoroprop-2-enoyl)piperazin-2-yl]acetonitrile
[(2S)-4-[(7S)-7-[3-amino-2-fluoro-5-methyl-6-(trifluoromethyl)phenyl]-2-{[(2R,7aS)-2-fluorotetrahydro-1Hpyrrolizin-7a(5H)-yl]methoxy}-7,8-dihydro-5H-pyrano[4,3-d]pyrimidin-4-yl]-1-(2-fluoroprop-2-enoyl)piperazin-2-yl]acetonitrile
Kirsten rat sarcoma viral oncogene homolog inhibitor, antineoplastic, D3S 001, PFW9YLB86H
Elisrasib (D3S-001) is a next-generation, orally available KRAS G12C inhibitor developed by D3 Bio that demonstrates high potency, sustained target engagement, and strong clinical activity in advanced solid tumors, including those resistant to first-generation inhibitors. As of April 2026, clinical trials show it has a 52% objective response rate (ORR) in G12C inhibitor-naive patients and a 30% ORR in refractory populations.
Key Aspects of Elisrasib (D3S-001):
- Mechanism of Action: It is a highly potent, covalent inhibitor that selectively binds the GDP-bound (inactive) form of the KRAS G12C mutant, effectively halting tumor cell proliferation and metastasis.
- Superior Efficacy: Preliminary data suggests elisrasib may be more potent than earlier inhibitors like sotorasib and adagrasib, providing higher target occupancy at lower doses.
- Clinical Performance (AACR 2026 Data):
- Naive Patients: 52% ORR, with a median duration of response (mDOR) of 16.5 months and median progression-free survival (mPFS) of 12.2 months at the 600 mg dose.
- Refractory Patients: 32% ORR, with a mDOR of 15.6 months and mPFS of 8.1 months.
- Targeted Cancers: Clinical trials are focused on KRAS G12C-mutant tumors, specifically non-small cell lung cancer (NSCLC), colorectal cancer (CRC), and other solid tumors.
- Safety Profile: The drug has shown good tolerability and a safe profile in early studies.
Elisrasib is in Phase 1/2 development and was highlighted for its promising results in treating patients with KRAS G12C-mutant tumors
Elisrasib is an orally bioavailable inhibitor of the oncogenic KRAS substitution mutation G12C, with potential antineoplastic activity. Upon oral administration, elisrasib selectively targets the KRAS G12C mutant and inhibits KRAS G12C-mediated signaling. This may halt proliferation and metastasis in susceptible tumor cells. KRAS, a member of the RAS family of oncogenes, serves an important role in cell signaling, division and differentiation. Mutations of KRAS may induce constitutive signal transduction leading to tumor cell proliferation, invasion, and metastasis.
- A Phase 1 Study to Assess Food Effect on the Pharmacokinetics of D3S-001 in Healthy Adult ParticipantsCTID: NCT07093398Phase: Phase 1Status: CompletedDate: 2026-03-25
- A Phase 1/2 Study of D3S-002 as Monotherapy or Combination Therapy in Adult Subjects With Advanced Solid Tumors With MAPK Pathway MutationsCTID: NCT05886920Phase: Phase 1/Phase 2Status: Active, not recruitingDate: 2026-03-23
- A Study of D3S-001 Monotherapy or Combination Therapy in Subjects With Advanced Solid Tumors With a KRAS p.G12C MutationCTID: NCT05410145Phase: Phase 1/Phase 2Status: RecruitingDate: 2026-03-12
PAT
SYN
Example 17
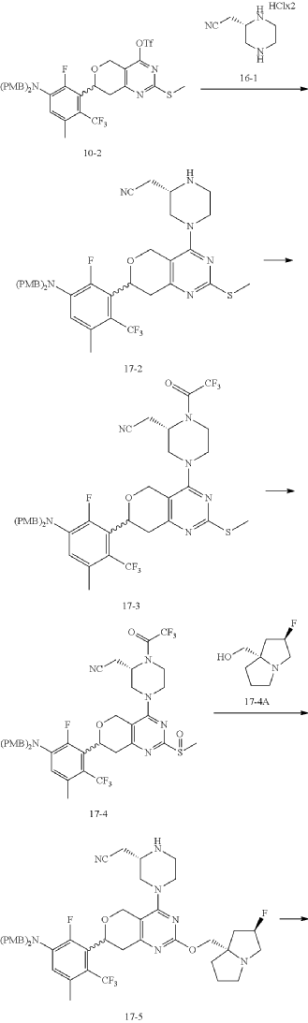
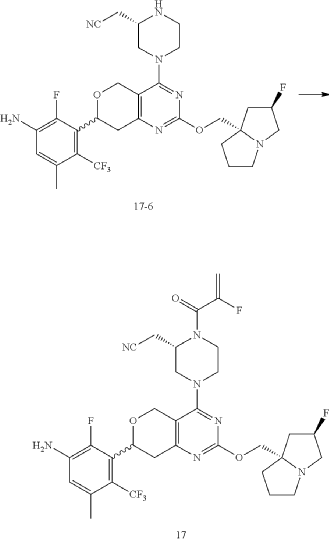
Step 6: Synthesis of Compound 17
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Pyrimidoheterocyclic compounds and application thereofPublication Number: EP-4105211-A1Priority Date: 2020-03-12
- Pyrimidoheterocyclic compounds and application thereofPublication Number: US-2023151004-A1Priority Date: 2020-03-12
//////////elisrasib, anax labs, Kirsten rat sarcoma viral oncogene homolog inhibitor, antineoplastic, D3S 001, PFW9YLB86H
Elironrasib
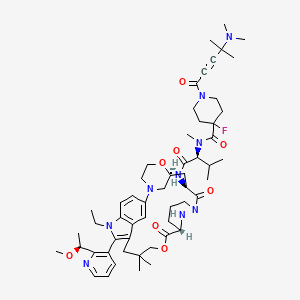
Elironrasib
CAS 2641998-63-0
MFC55H78FN9O8 MW 1012.3 g/mol
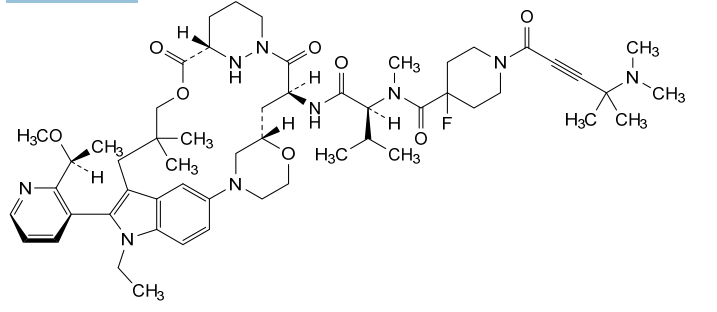
1-[4-(dimethylamino)-4-methylpent-2-ynoyl]-N-[(2S)-1-[[(6S,8S,14S)-22-ethyl-21-[2-[(1S)-1-methoxyethyl]-3-pyridinyl]-18,18-dimethyl-9,15-dioxo-5,16-dioxa-2,10,22,28-tetrazapentacyclo[18.5.2.12,6.110,14.023,27]nonacosa-1(26),20,23(27),24-tetraen-8-yl]amino]-3-methyl-1-oxobutan-2-yl]-4-fluoro-N-methylpiperidine-4-carboxamide
- 1-[4-(dimethylamino)-4-methylpent-2-ynoyl]-N-[(2S)-1-[[(6S,8S,14S)-22-ethyl-21-[2-[(1S)-1-methoxyethyl]pyridin-3-yl]-18,18-dimethyl-9,15-dioxo-5,16-dioxa-2,10,22,28-tetrazapentacyclo[18.5.2.12,6.110,14.023,27]nonacosa-1(26),20,23(27),24-tetraen-8-yl]amino]-3-methyl-1-oxobutan-2-yl]-4-fluoro-N-methylpiperidine-4-carboxamide
- 3-Pyridazinecarboxylic acid, N1-[N-[[1-[4-(dimethylamino)-4-methyl-1-oxo-2-pentyn-1-yl]-4-fluoro-4-piperidinyl]carbonyl]-N-methyl-L-valyl-3-[4-[(2R)-1-ethyl-3-(3-hydroxy-2,2-dimethylpropyl)-2-[2-[(1S)-1-methoxyethyl]-3-pyridinyl]-1H-indol-5-yl]-2-morpholinyl]-L-alanyl]hexahydro-, (3–>2)-lactone, (3S)-
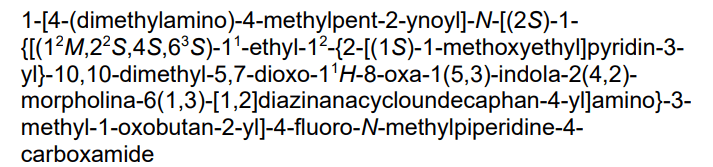
Kirsten rat sarcoma viral oncogene homolog inhibitor, antineoplastic, RMC-6291, RMC 6291, 942KVV5CJP
Elironrasib (RMC-6291) is an investigational, orally bioavailable, RAS(ON) G12C-selective inhibitor developed by Revolution Medicines that targets the active GTP-bound form of KRAS G12C. In Phase 1 trials, it showed significant promise in treating advanced KRAS G12C-mutated solid tumors, including non-small cell lung cancer (NSCLC). [1, 2, 3]
Key Clinical Trial Results (as of Oct 2025):
- Response Rate: 42% objective response rate (ORR).
- Disease Control: 79% disease control rate (DCR).
- Durability: Median duration of response was 11.2 months.
- Survival: Median progression-free survival was 6.2 months.
- Overcoming Resistance: Demonstrated efficacy in patients who had previously progressed on first-generation KRAS G12C(OFF) inhibitors.
Mechanism of Action:
Elironrasib acts as a covalent tri-complex inhibitor (TCI). It forms a complex with the intracellular chaperone protein cyclophilin A (CypA) and the active KRAS G12C(GTP) protein, effectively shutting down oncogenic signaling.
Development Status:
- Designation: It has received FDA breakthrough therapy designation for KRAS G12C-mutant NSCLC.
- Trials: Currently in Phase 1 clinical trials (e.g., NCT05462717) to evaluate safety, tolerability, and efficacy, both as a monotherapy and in combination.
- Target Population: Patients with KRAS G12C-addicted solid tumors.
Discovery of Elironrasib (RMC-6291), a Potent and Orally Bioavailable, RAS(ON) G12C-Selective, Covalent Tricomplex Inhibitor for the Treatment of Patients with RAS G12C-Addicted Cancers – PubMed27 Mar 2025 — This information does not constitute medical advice or diagnosis. Elirronrasib (RMC-6291) is a potent, orally bioavailable,
- Revolution Medicines to Present Updated Elironrasib Safety and Efficacy Data in Patients with KRAS G12C Non-Small Cell Lung Cancer Following Treatment with a KRAS(OFF) G12C Inhibitor22 Oct 2025 — This information does not constitute medical advice or diagnosis. Elirronrasib is a RAS(ON) G12C-selective inhibitor being develop…
Revolution Medicines
- Elironrasib May Overcome Resistance to Prior KRAS G12C Inhibition in Non-small Cell Lung Cancer
- OriginatorREVOLUTION Medicines
- ClassAntineoplastics; Morpholines; Piperidines; Pyridazines; Small molecules
- Mechanism of ActionKRAS protein inhibitors
- Phase I/IISolid tumours
- Clinical Phase UnknownNon-small cell lung cancer
- 30 Jan 2026Phase-I/II clinical trials in Solid tumours (Combination therapy, Late-stage disease, Metastatic disease) in USA (PO) (NCT07397338)
- 31 Oct 2025Elironrasib is still in phase I trial in Solid tumours (Late-stage disease, Metastatic disease, Monotherapy) in Australia, Italy, South Korea, Malaysia, Singapore, Spain, Czech Republic, Thailand and USA (PO, Tablet) (NCT05462717)
- 28 Oct 2025No recent reports of development identified for phase-I development in Solid-tumours(Late-stage disease, Metastatic disease, Monotherapy) in Australia, Italy, South Korea, Malaysia, Singapore, Spain, Czech Republic, Thailand (PO, Tablet)
Elironrasib is an orally bioavailable, covalent inhibitor of the active, guanosine triphosphate (GTP)-bound form of the oncogenic KRAS substitution mutation G12C, KRAS G12C(ON), with potential antineoplastic activity. Upon oral administration, elironrasib forms a tri-complex with the intracellular chaperone protein and immunophilin cyclophilin A (CypA) and KRAS G12C(ON). This tri-complex inhibits KRAS G12C(ON)-mediated signaling, which may inhibit tumor cell proliferation. KRAS, a member of the RAS family of oncogenes, serves an important role in cell signaling, division and differentiation. Mutations of KRAS may induce constitutive signal transduction leading to tumor cell growth, proliferation, invasion, and metastasis.
- Study of Elironrasib and Daraxonrasib as Monotherapies and Combination Therapy in Participants With Advanced KRAS G12C Mutant Solid TumorsCTID: NCT06128551Phase: Phase 1/Phase 2Status: RecruitingDate: 2026-04-23
- Dose Escalation and Dose Expansion Study of RMC-6291 Monotherapy in Subjects With Advanced KRASG12C Mutant Solid TumorsCTID: NCT05462717Phase: Phase 1Status: Active, not recruitingDate: 2026-04-08
- Study of RAS(ON) Inhibitors in Combination With Ivonescimab in Patients With Solid TumorsCTID: NCT07397338Phase: Phase 1/Phase 2Status: RecruitingDate: 2026-03-30
- Study of RAS(ON) Inhibitors in Patients With Advanced RAS-mutated NSCLCCTID: NCT06162221Phase: Phase 1/Phase 2Status: RecruitingDate: 2026-03-09
SYN
https://pubs.acs.org/doi/10.1021/acs.jmedchem.4c02313


SYN
PAT’
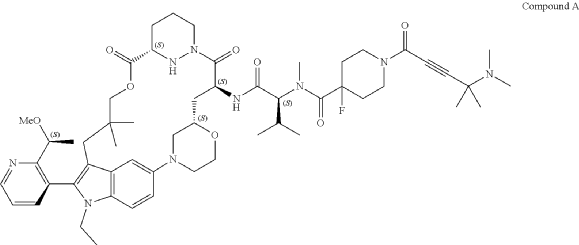
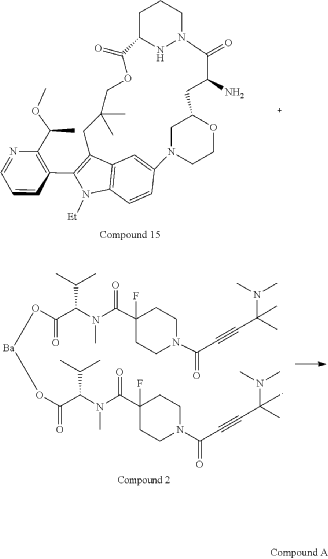
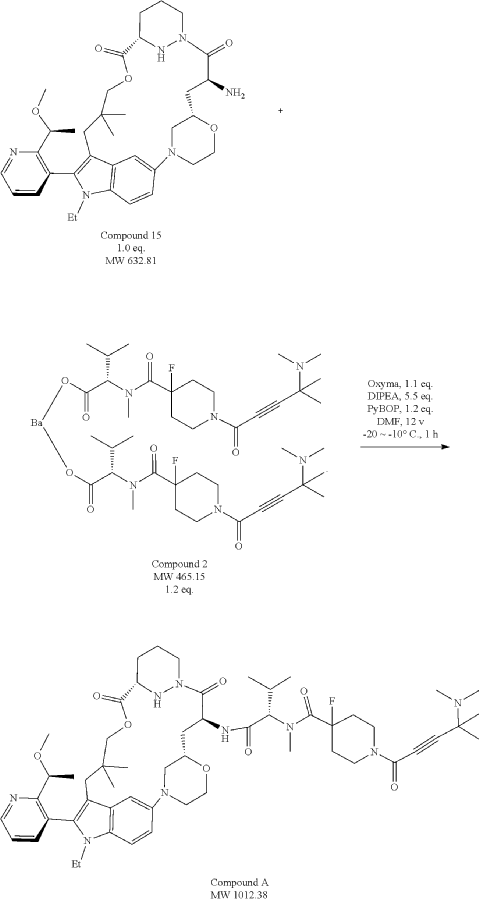
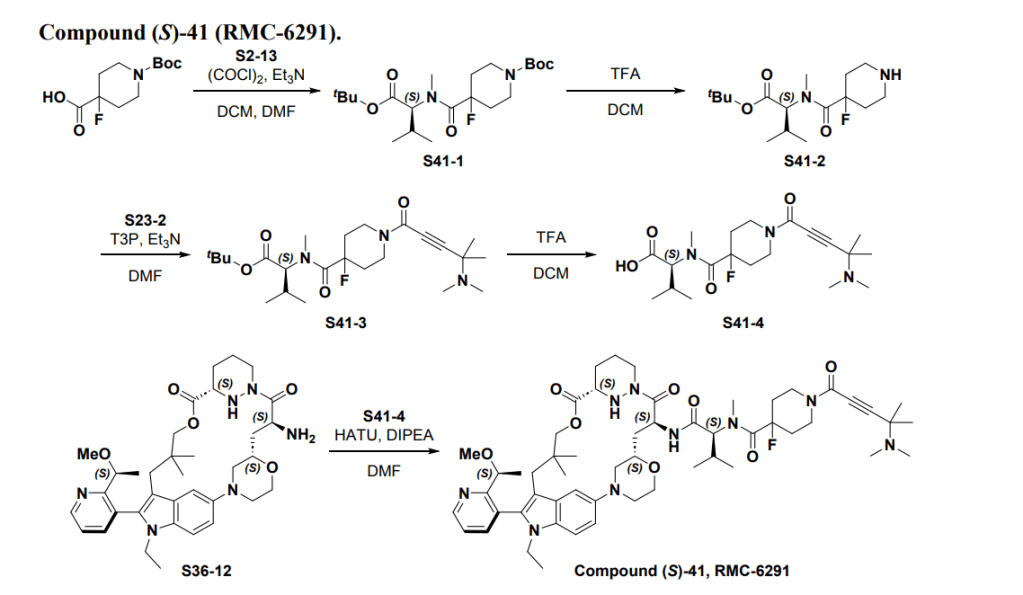
Part 5—Synthesis of Compound A—(12M)-1-(4-(dimethylamino)-4-methylpent-2 ynoyl)-N-((2S)-1-(((22S,63S,4S)-11-ethyl-12-(2-((S)-1-methoxyethyl)pyridin-3-yl)-10,10-dimethyl-5,7-dioxo-61,62,63,64,65,66-hexahydro-11H-8-oxa-2(4,2)-morpholina-1(5,3)-indola-6(1,3) pyridazinacycloundecaphane-4-yl)amino)-3-methyl-1-oxobutan-2 yl)-4-fluoro-N-methylpiperidine-4-carboxamide
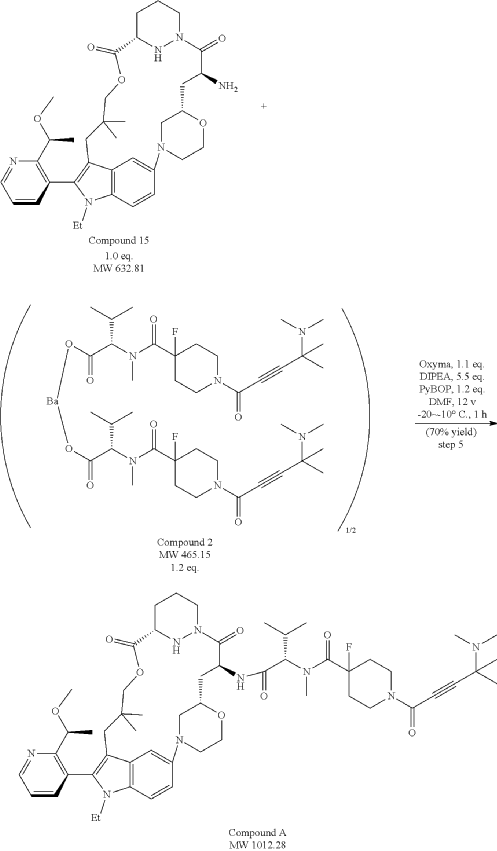
| To a 50 L glass reactor was charged Compound 15 (1.91 kg, 1.0 eq) and DMF (13.9 kg). The mixture was agitated at 20-30° C. until all of the solids were dissolved. Compound 2 (1.70 kg, 1.2 eq) and DMF (3.8 kg) were charged. The mixture was agitated at 20-30° C. until all of the solids were dissolved. DIPEA (2.20 kg, 5.50 eq) was charged at 20-30° C. and the mixture was cooled to −20-−10° C. under agitation. Ethyl cyanoglyoxylate-2-oxime (Oxyma) (0.48 kg, 1.1 eq) was charged to the reactor and the reaction mixture was agitated at −20 to −10° C. for 30 min. PyBOP was charged as a DMF solution (1.89 kg dissolved in 3.62 kg DMF, 1.2 eq) to the reactor at −20 to −10° C. in </=1 h. The reaction mixture was agitated at −20 to −10° C. for 1-3 h. Reaction monitoring by HPLC showed the reaction was complete. |
| The crude product was then further purified by recrystallization with a mixture of EtOAc and n-Heptane to give purified Compound A as a white solid. |



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma
References
- Macrocyclic heterocycles and uses thereofPublication Number: US-2023339952-A1Priority Date: 2022-04-20
- Macrocyclic heterocycles and uses thereofPublication Number: WO-2023205701-A1Priority Date: 2022-04-20
- Methods for delaying, preventing, and treating acquired resistance to ras inhibitorsPublication Number: US-2023233569-A1Priority Date: 2020-06-18
- Ras inhibitorsPublication Number: US-2021130303-A1Priority Date: 2019-11-04
- Ras inhibitorsPublication Number: US-2023234929-A1Priority Date: 2019-11-04
- Ras inhibitors
- Publication Number: US-11566007-B2
- Priority Date: 2019-11-04
- Grant Date: 2023-01-31
////////elironrasib, ANAX LABS, Kirsten rat sarcoma viral oncogene homolog inhibitor, antineoplastic, RMC-6291, RMC 6291, 942KVV5CJP
Dirozalkib
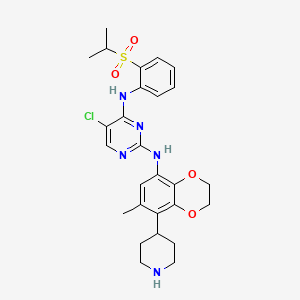
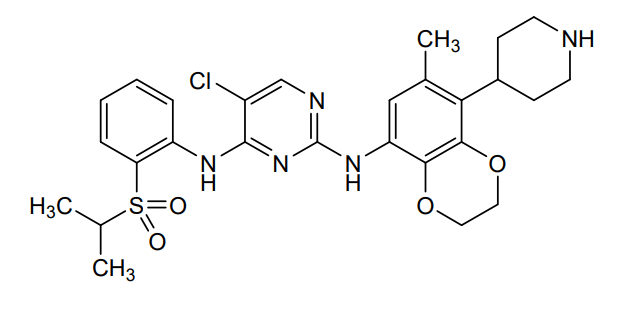
Dirozalkib
CAS 1893419-37-8
MF C27H32ClN5O4S MW558.1 g/mol
5-chloro-2-N-(6-methyl-5-piperidin-4-yl-2,3-dihydro-1,4-benzodioxin-8-yl)-4-N-(2-propan-2-ylsulfonylphenyl)pyrimidine-2,4-diamine

anaplastic lymphoma kinase (ALK) inhibitor, antineoplastic, XZP-3621, XZP 3621, Xuanzhu Biopharmaceutical, 2FH56C28YT
Dirozalkib (XZP-3621) is a novel, potent, and highly selective ALK/ROS1 tyrosine kinase inhibitor developed by Xuanzhu Biopharmaceutical to treat advanced ALK-positive non-small cell lung cancer (NSCLC). It demonstrated high efficacy (47.4% ORR, up to 89.3% in naive patients) in clinical trials and is designed to overcome resistance to earlier inhibitors.
Key Aspects of Dirozalkib
- Indication: Treatment of adult patients with ALK-positive locally advanced or metastatic non-small cell lung cancer (NSCLC).
- Mechanism: Acts as a dual-target ALK/ROS1 tyrosine kinase inhibitor (TKI), effective against ALK fusion-positive cells and various resistance mutations.
- Clinical Efficacy (Phase I/II): In studies, the drug showed significant antitumor activity with an Objective Response Rate (ORR) of 47.4% and an 89.3% ORR in ALK inhibitor-naive patients at 500 mg/day.
- Safety Profile: No dose-limiting toxicities occurred; the maximum tolerated dose was 600 mg/day, with a recommended dose of 500 mg/day. Common adverse events included diarrhea.
- Status: As of early 2026, the NDA (New Drug Application) for Dexitinib (Dirozalkib) was accepted by China’s NMPA, with potential for further market expansion.
- OriginatorXuanzhu Biopharmaceutical
- Class2 ring heterocyclic compounds; Amines; Aniline compounds; Antineoplastics; Chlorinated hydrocarbons; Piperidines; Pyrimidines; Small molecules; Sulfones
- Mechanism of ActionAnaplastic lymphoma kinase inhibitors
- RegisteredNon-small cell lung cancer
- 26 Aug 2025Chemical structure information added.
- 22 Aug 2025Registered for Non-small cell lung cancer (Late-stage disease) in China (PO) – First global approval
- 22 Aug 2025Efficacy and adverse events data from a phase III trial in Non-small cell lung cancer released by Xuanzhu Biopharmaceutical
- A Phase I Study of XZP-3621 in Chinese Patients With ALK or ROS1 Rearrangement Non-small Cell Lung CancerCTID: NCT05055232Phase: Phase 1Status: CompletedDate: 2025-07-24
- Food Effect and Mass Balance Study of XZP-3621 TabletsCTID: NCT05034120Phase: Phase 1Status: CompletedDate: 2025-05-25
- A Study of XZP-3621 in Chinese Patients With ALK Positive NSCLCCTID: NCT05482087Phase: Phase 2Status: Unknown statusDate: 2022-08-01
- A Study to Evaluate and Compare the Efficacy and Safety of XZP-3621 Versus CrizotinibCTID: NCT05204628Phase: Phase 3Status: Unknown statusDate: 2022-01-24
PAT
PAT
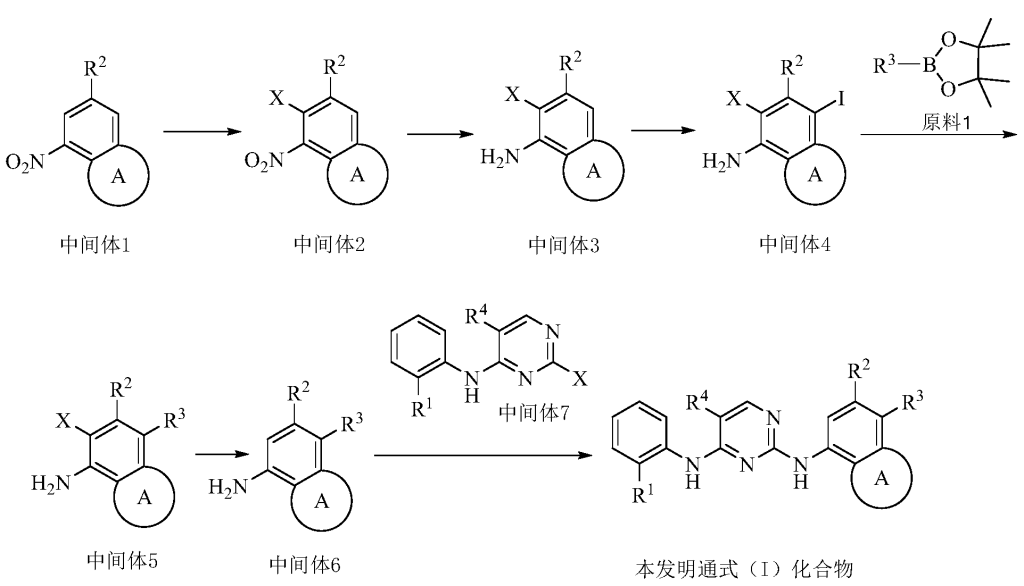
Example 3 Preparation of 2-((5-chloro-2-((7-methyl-8-(piperidin-4-yl)-2,3-dihydrobenzo[b][1,4]dioxin- 5-yl)amino)pyrimidin-4-yl)amino)-N,N-dimethylbenzenesulfonamide (compound 3)
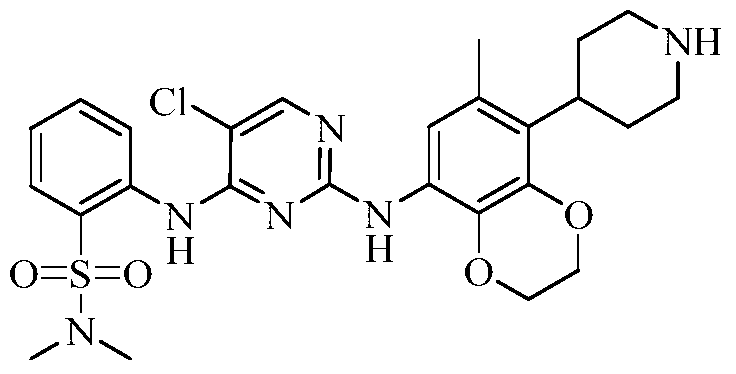
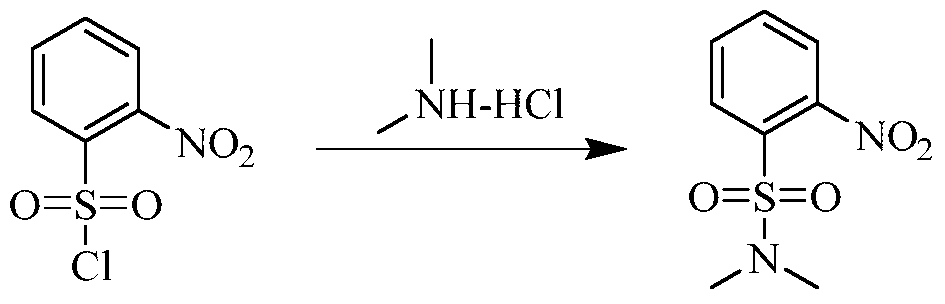
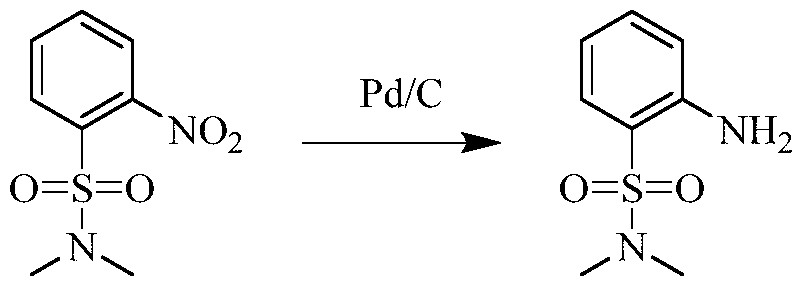
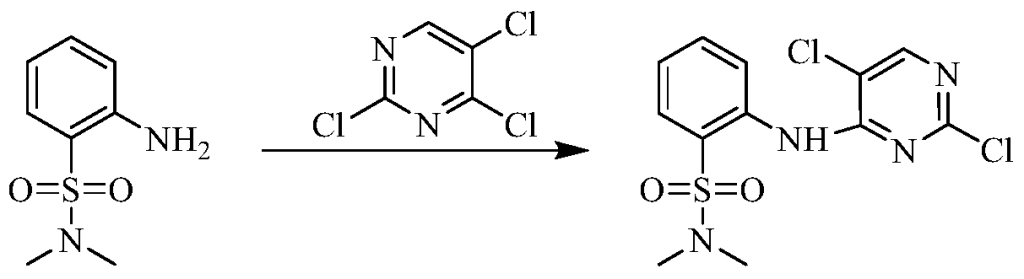
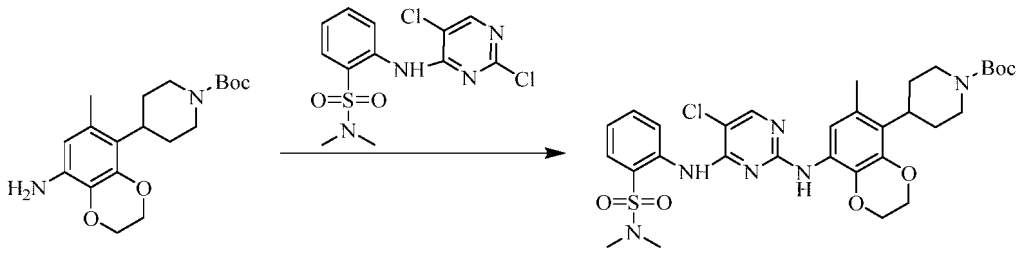
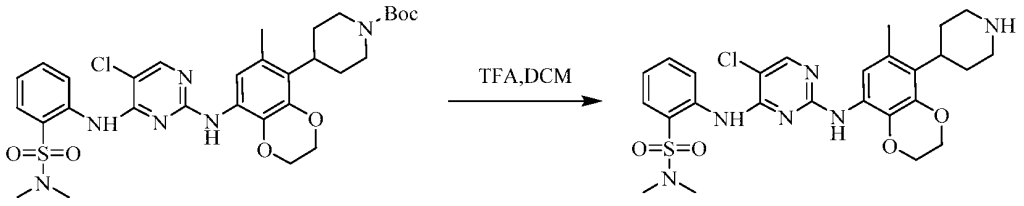
(5) Preparation of 2-((5-chloro-2-((7-methyl-8-(piperidin-4-yl)-2,3-dihydrobenzo[b][1,4]dioxin-5-yl)amino)pyrimidin-4-yl)amino)-N,N-dimethylbenzenesulfonamide
75 mg (0.114 mmol) of tert-butyl 4-(8-((5-chloro-4-((2-(N,N-dimethylaminosulfonyl)phenyl)amino)pyrimidin-2-yl)amino)-6-methyl-2,3-dihydrobenzo[b][1,4]dioxin-5-yl)piperidine-1-carboxylic acid ester was dissolved in dichloromethane (10 mL), and trifluoroacetic acid (1 mL) was added. The mixture was stirred at room temperature for 12 hours. The starting material disappeared as detected by TLC. Water (20 mL) was added, and the mixture was separated. The aqueous phase was extracted twice with dichloromethane (20 mL × 2). The organic phases were combined, dried over anhydrous sodium sulfate, and the solvent was removed by rotary evaporation. The crude product was purified by silica gel column chromatography (methanol:dichloromethane = 1:50) to obtain the final product (30 mg, yield 47.2%).
[0415]Molecular formula:
C26H31ClN6O4S Molecular weight: 559.08 LC-MS (m / z): 280.2 [ M /2+H ] +
[0416]
1H-NMR(400MHz,MeOD)δ:8.44(d,1H,J=1.2),8.11(s,1H),7.86(d,1H,J=1.2),7.56-7.60(m,1H),7.28-7.35(m,2H),4.26(s,4H),3.45-3.48(m,2H),3.06-3.15(m,3H),2.56-2.74(m,8H),2.17(s,3H),1.76-1.80(m,2H).



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Polycyclic inhibitor of anaplastic lymphoma kinasePublication Number: US-10011592-B2Priority Date: 2014-09-29Grant Date: 2018-07-03
- Polycyclic inhibitor of anaplastic lymphoma kinasePublication Number: US-2018086745-A9Priority Date: 2014-09-29
- Polycyclic anaplastic lymphoma kinase inhibitorPublication Number: EP-3202765-A1Priority Date: 2014-09-29
- Polycyclic inhibitor of anaplastic lymphoma kinasePublication Number: KR-20170055555-APriority Date: 2014-09-29
- Crystal form of polycyclic anaplastic lymphoma kinase inhibitorPublication Number: US-2023348443-A1Priority Date: 2020-01-17
- Crystal form of polycyclic anaplastic lymphoma kinase inhibitorPublication Number: US-12441717-B2Priority Date: 2020-01-17Grant Date: 2025-10-14
- Polycyclic inhibitor of anaplastic lymphoma kinasePublication Number: US-2017240534-A1Priority Date: 2014-09-29
- Polycyclic inhibitor of anaplastic lymphoma kinasePublication Number: KR-101909404-B1Priority Date: 2014-09-29Grant Date: 2018-10-17
- Polycyclic anaplastic lymphoma kinase inhibitorPublication Number: WO-2016050171-A1Priority Date: 2014-09-29
- Pharmaceutical composition of anaplastic lymphoma kinase inhibitor and preparation method thereforPublication Number: WO-2025140560-A1Priority Date: 2023-12-28
- CRYSTALLINE FORM OF A POLYCYCLIC ANAPLASIC LYMPHOMA KINASE INHIBITORPublication Number: EP-4092021-A4Priority Date: 2020-01-17
- Polycyclic anaplastic lymphoma kinase inhibitor crystalline formPublication Number: CN-113135905-BPriority Date: 2020-01-17Grant Date: 2023-11-21
- Crystal form of polycyclic anaplastic lymphoma kinase inhibitorPublication Number: WO-2021143819-A1Priority Date: 2020-01-17
- Crystal form of polycyclic anaplastic lymphoma kinase inhibitorPublication Number: EP-4092021-A1Priority Date: 2020-01-17
/////////dirozalkib, anax labs, anaplastic lymphoma kinase (ALK) inhibitor, antineoplastic, XZP-3621, XZP 3621, Xuanzhu Biopharmaceutical, 2FH56C28YT
Dencatistat
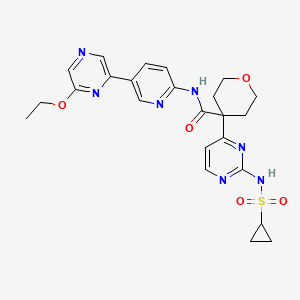
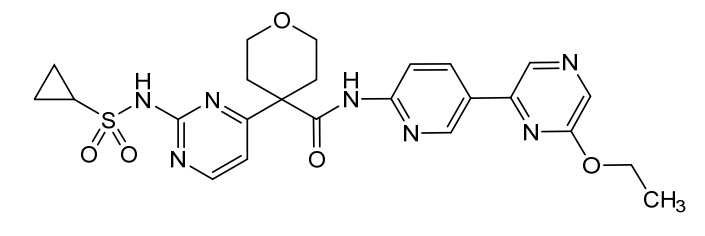
Dencatistat
CAS 2377000-84-3
MFC24H27N7O5S MW 525.6 g/mol
4-[2-(cyclopropylsulfonylamino)pyrimidin-4-yl]-N-[5-(6-ethoxypyrazin-2-yl)-2-pyridinyl]oxane-4-carboxamide
4-[2-(cyclopropanesulfonamido)pyrimidin-4-yl]-N-[5-(6-ethoxypyrazin-2-yl)pyridin-2-yl]oxane-4-carboxamide
CTP synthase 1 inhibitor, antineoplastic, STP 938, CTPS1-IN-2, QG9C9SZZ3T
Dencatistat (formerly known as STP938) is a first-in-class, orally bioavailable cancer drug designed to target specific blood cancers and solid tumours
Dencatistat is an orally bioavailable, small molecule inhibitor of cytidine triphosphate synthase 1 (CTPS1), with potential antineoplastic activity. Upon oral administration, dencatistat targets, binds to and inhibits the activity of CTPS1, thereby decreasing the production of cytidine triphosphate (CTP), an essential building block of deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). This may disrupt DNA and RNA synthesis and trigger apoptosis. CTPS1, an enzyme that catalyzes the rate-limiting step in pyrimidine synthesis, plays an important and nonredundant role in B-cell and T-cell proliferation. CTPS1 is required for rapid cell division in certain types of cancers that arise from blood cells.
Mechanism of Action
It works by inhibiting CTPS1 (Cytidine Triphosphate Synthase 1), a key enzyme that cancer cells “addicted” to for DNA synthesis.
- Targeted approach: It aims to kill cancer cells while leaving healthy cells unharmed by exploiting a “synthetic lethal” dependency in certain tumours.
- Precision medicine: It is particularly being tested in patients whose tumours lack CTPS2, a backup enzyme, which makes them highly vulnerable to dencatistat.
🏥 Clinical Status
Developed by Step Pharma, the drug is currently in several clinical trials:
- Lymphoma: Phase 1/2 trials for relapsed or refractory T-cell and B-cell lymphomas.
- Solid Tumours: Phase 1 studies for patients with solid tumours, specifically ovarian and endometrial cancers.
- Essential Thrombocythaemia: A Phase 1b trial for this blood disorder was initiated in 2025.
- Orphan Drug Status: Received FDA Orphan Drug Designation for T-cell lymphoma in May 2025.
- OriginatorStep Pharma
- ClassAnti-inflammatories; Antineoplastics; Antirheumatics; Antithrombotics; Small molecules
- Mechanism of ActionCTPS1 protein inhibitors
- Orphan Drug StatusYes – T-cell lymphoma
- Phase I/IIB-cell lymphoma; T-cell lymphoma
- Phase ISolid tumours; Thrombocytosis
- PreclinicalGraft-versus-host disease; Inflammation
- No development reportedRheumatoid arthritis
- 23 Feb 2026Step Pharma plans phase II trials for Gynaecological cancer
- 10 Feb 2026Preclinical development in Inflammation is till ongoing in France (PO) (Step Pharma pipeline, February 2026)
- 15 Oct 2025Adverse event data from a phase I/II trial in T-cell lymphoma/B-cell lymphoma released by Step Pharma
SYN
US20250177394, Compound CTPS1-IA
PAT
PAT
A. Preparation of Active Ingredient
20 Example A1 – Preparation of crude 4-(2-(cyclopropanesulfonamido)pyrimidin-4-yl)-N-(5- (6-ethoxypyrazin-2-yl)pyridin-2-yl)tetrahydro-2H-pyran-4-carboxamide
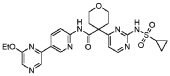
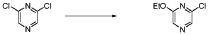
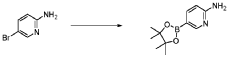
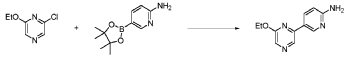
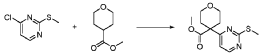
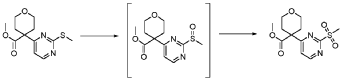
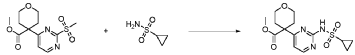
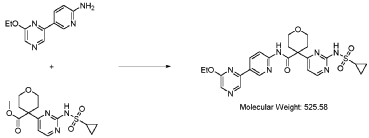
Step 4 – Preparation of crude 4-(2-(cyclopropanesulfonamido)pyrimidin-4-yl)-N-(5-(6- ethoxypyrazin-2-yl)pyridin-2-yl)tetrahydro-2H-pyran-4-carboxamide
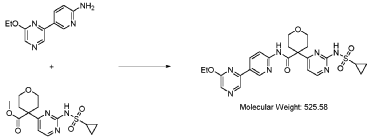
4-(2-(cyclopropanesulfonamido)pyrimidin-4-yl)tetrahydro-2H-pyran-4-carboxylate (1.76 kg, 5.15 mol, 1.00 equiv.) and 5-(6-ethoxypyrazin-2-yl)pyridin-2-amine (1.22 kg, 5.65 mol, 1.10 equiv.) were suspended in a mixture of THF (27.1 L, 15.5 rel. vol.) and DMSO (2.63 L, 1.50 rel. vol.) and stirred until the solids were evenly dispersed. The mixture was concentrated by
STP-P3718PCT
102
distillation at atmospheric pressure and approximately 70 oC to a volume of 15 L. The temperature was adjusted to 20 ± 5 oC, potassium tert-butoxide (6.92 kg 20 wt% solution in THF, 12.3 mol, 2.40 equiv.) was added over 1 h and the reaction mixture stirred at 20 ± 5 oC for 70 minutes until completion. THF (880 mL, 0.500 rel vol.) was charged, followed by acetic acid (780 5 mL, 820 g, 13.6 mol, 2.64 equiv.) over 10 minutes, followed by methanol (4.40 L, 2.50 rel. vol.), followed by water (13.2 L, 7.50 rel. vol.) over 35 minutes. The mixture was stirred at 20 ± 5 oC for 15 minutes and then 16 h at 0 ± 5 oC. The resulting suspension was filtered and washed with water (2 × 8.80 L, 2 × 5.00 rel. vol.), followed by methanol (4.40 L, 2.50 rel. vol.) The filter cake was dried at 35 oC under a flow of nitrogen for 20 h to afford crude 4-(2-10 (cyclopropanesulfonamido)pyrimidin-4-yl)-N-(5-(6-ethoxypyrazin-2-yl)pyridin-2-yl)tetrahydro- 2H-pyran-4-carboxamide (“CTPS1-IA”).
PAT
- Aminopyrimidine derivatives as CTPS1 inhibitorsPublication Number: JP-7428692-B2Priority Date: 2018-03-23Grant Date: 2024-02-06
- Aminopyrimidine derivatives as ctps1 inhibitorsPublication Number: EP-3768674-A1Priority Date: 2018-03-23
- Aminopyrimidine derivative as a CTPS1 inhibitorPublication Number: JP-2021518436-APriority Date: 2018-03-23
- Aminopyrimidine derivatives as ctps1 inhibitorsPublication Number: EP-3768674-B1Priority Date: 2018-03-23Grant Date: 2024-01-03
- Aminopyrimidine derivatives as CTPS1 inhibitorsPublication Number: CN-111868051-APriority Date: 2018-03-23
- Aminopyrimidine derivatives as CTPS1 inhibitorsPublication Number: ES-2974445-T3Priority Date: 2018-03-23Grant Date: 2024-06-27
- CompoundsPublication Number: US-2021024507-A1Priority Date: 2018-03-23
- CompoundsPublication Number: US-2021387965-A1Priority Date: 2018-10-23
- CompoundsPublication Number: US-2023192673-A1Priority Date: 2018-06-04
- Aminopyrimidine derivatives as CTPS1 inhibitorsPublication Number: CN-111868051-BPriority Date: 2018-03-23Grant Date: 2024-04-09
- Aminopyrimidine derivatives as ctps1 inhibitorsPublication Number: WO-2019180244-A1Priority Date: 2018-03-23
- Aminopyrimidine derivatives as ctps1 inhibitorsPublication Number: WO-2019179652-A1Priority Date: 2018-03-23



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
////////dencatistat, anax lab, CTP synthase 1 inhibitor, antineoplastic, STP 938, CTPS1-IN-2, QG9C9SZZ3T

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










