Home » Posts tagged 'Antineoplastic' (Page 2)
Tag Archives: Antineoplastic
Darlifarnib
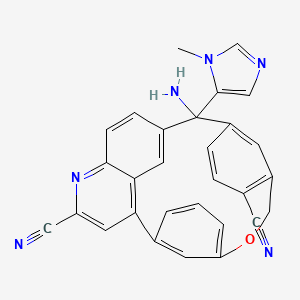
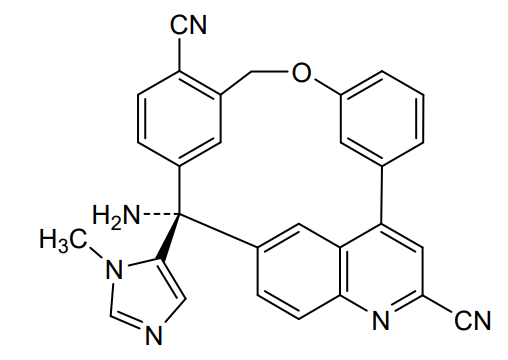
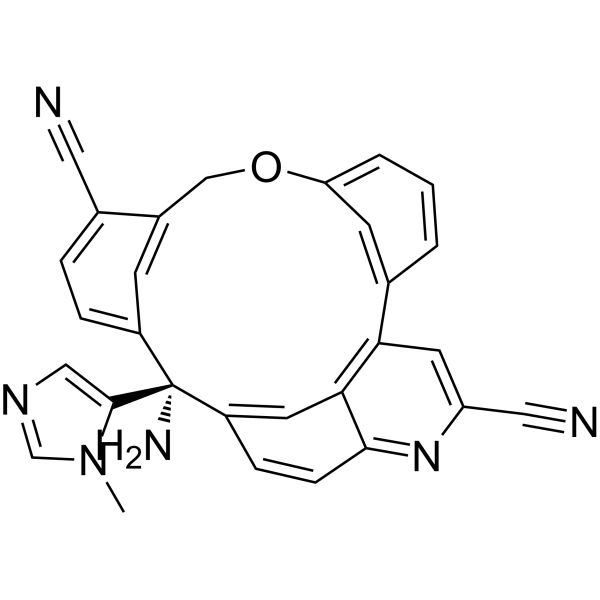
Darlifarnib
CAS 2939824-30-1
MF C29H20N6O MW 468.51
14-amino-14-(3-methylimidazol-4-yl)-7-oxa-19-azapentacyclo[13.6.2.12,6.19,13.018,22]pentacosa-1(22),2(25),3,5,9,11,13(24),15(23),16,18,20-undecaene-10,20-dicarbonitrile
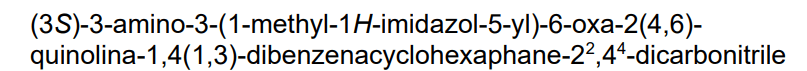
farnesyl transferase inhibitor, antineoplastic, KO-2806, KO 2806, T206317
Darlifarnib (KO-2806) is an investigational, orally active next-generation farnesyl transferase inhibitor (FTI) being developed by Kura Oncology to treat solid tumors, such as clear cell renal cell carcinoma (ccRCC). It inhibits the enzyme farnesyl transferase, blocking KRAS and mTORC1 signaling to induce tumor regression. It is often combined with other agents to overcome resistance.
Key Details About Darlifarnib
- Mechanism of Action: As a FTI, darlifarnib binds to and inhibits farnesyl transferase, which prevents the activation of RAS oncogenes and inhibits downstream mTORC1 signaling, leading to tumor cell death.
- Target Indications: Preclinical and early clinical data show potential in treating KRAS-mutant cancers, including non-small cell lung cancer (NSCLC), colorectal cancer (CRC), and clear cell renal cell carcinoma (ccRCC).
- Combination Therapy: Data from the Phase 1 FIT-001 trial (presented in April 2026) showed that combining darlifarnib with the TKI cabozantinib demonstrated robust activity in patients with pretreated, advanced ccRCC.
- Overcoming Resistance: Darlifarnib is designed to re-sensitize tumors that have become resistant to prior therapies, such as RAS inhibitors and tyrosine kinase inhibitors (TKIs).
- Status: It is an investigational drug and not yet FDA-approved.
- OriginatorKura Oncology
- ClassAntineoplastics; Small molecules
- Mechanism of ActionFarnesyltranstransferase inhibitors
- Phase IAdenocarcinoma; Colorectal cancer; Non-small cell lung cancer; Renal cell carcinoma; Solid tumours
- 12 Jan 2026Kura Oncology plans the one or more expansion cohorts of KO 2806 and cabozantinib in patients with advanced renal cell carcinoma in the first half of 2026
- 22 Oct 2025Pharmacodynamics data from a preclinical trial in Cancer presented at the AACR-NCI-EORTC International Conference on Molecular Targets and Cancer Therapeutics 2025 (AACR-NCI-EORTC-2025)
- 18 Oct 2025Adverse events and efficacy data from a phase I trial in Non-small cell lung cancer, Renal cell carcinoma, Adenocarcinoma released by Kura Oncology
PAT
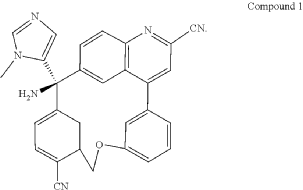
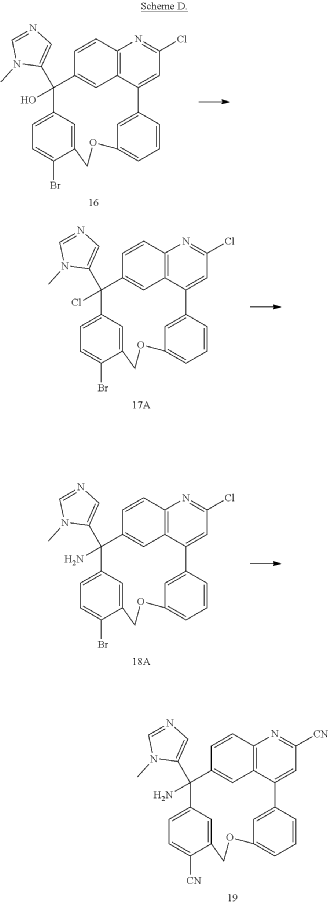
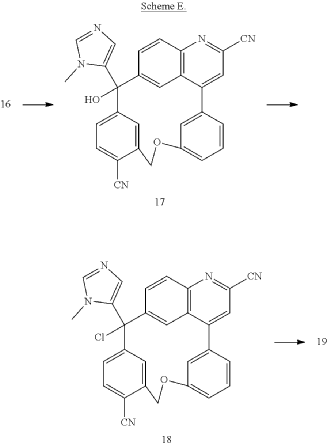
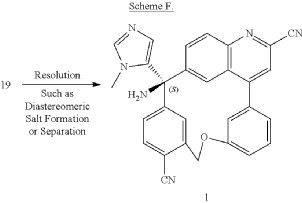
PAT
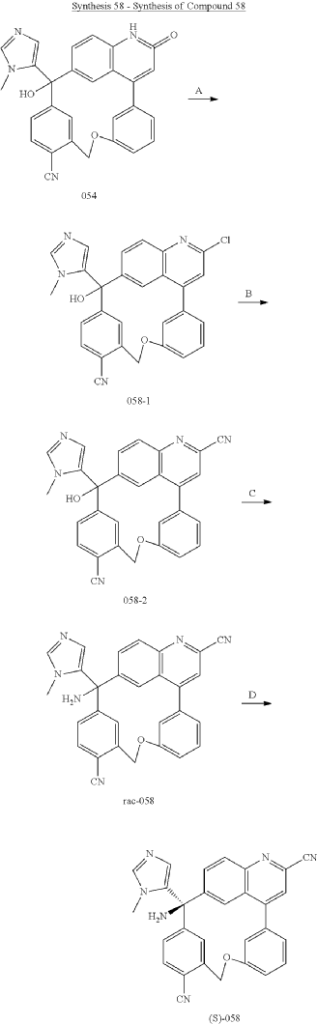
Step A: Preparation of (058-1)
Step B: Preparation of (058-2)
Step C: Preparation of (rac)-3-amino-3-(1-methyl-1H-imidazol-5-yl)-6-oxa-2(4,6)-quinolina-1,4(1,3)-dibenzenacyclohexaphane-22,44-dicarbonitrile (rac-058)
Step D: Preparation of (S)-3-amino-3-(1-methyl-1H-imidazol-5-yl)-6-oxa-2(4,6)-quinolina-1,4(1,3)-dibenzenacyclohexaphane-22,44-dicarbonitrile ((S)-058)
PAT
- Macrocyclic compounds and compositions, and methods of preparing and using the samePublication Number: US-2023322711-A1Priority Date: 2021-11-30
- Macrocyclic compounds and compositions, and methods of preparing and using the samePublication Number: US-12018011-B2Priority Date: 2021-11-30Grant Date: 2024-06-25



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
/////////////darlifarnib, ANAX LAB, farnesyl transferase inhibitor, antineoplastic, KO-2806, KO 2806, T206317
Daraxonrasib
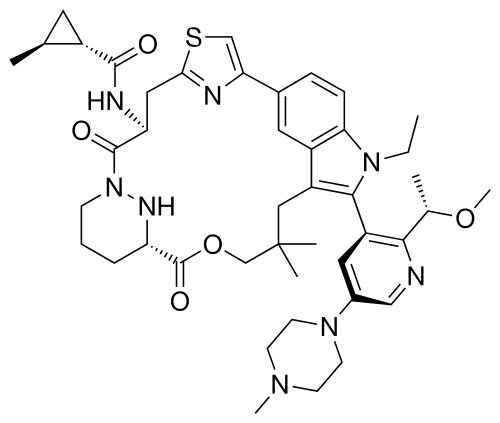
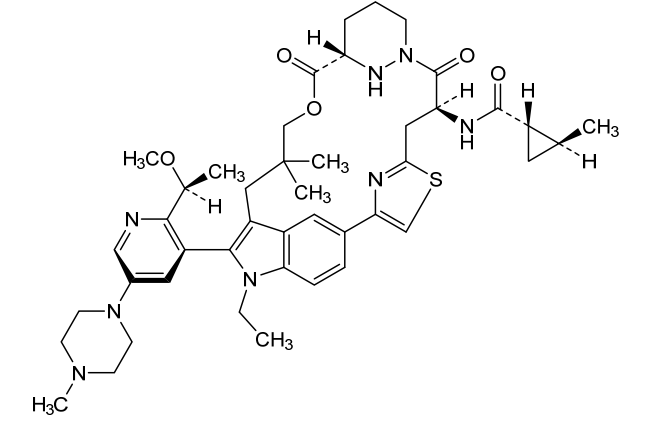
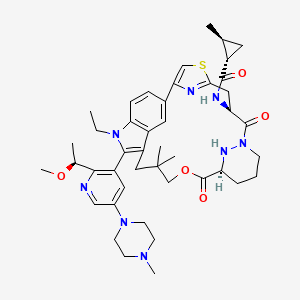
Daraxonrasib
CAS 2765081-21-6
MFC44H58N8O5S MW811.0 g/mol
trans-(1S,2S)-N-[(7S,13S)-21-ethyl-20-[2-[(1S)-1-methoxyethyl]-5-(4-methylpiperazin-1-yl)-3-pyridinyl]-17,17-dimethyl-8,14-dioxo-15-oxa-4-thia-9,21,27,28-tetrazapentacyclo[17.5.2.12,5.19,13.022,26]octacosa-1(25),2,5(28),19,22(26),23-hexaen-7-yl]-2-methylcyclopropane-1-carboxamide
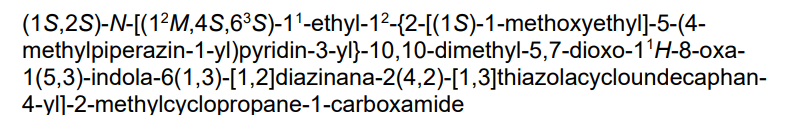
Kirsten rat sarcoma viral oncogene homolog inhibitor, antineoplastic, RMC-6236, RMC 6236, B6T47Y2UAP, RAS-IN-2,
Daraxonrasib (formerly RMC-6236) is an investigational, orally administered “molecular glue” RAS inhibitor developed by Revolution Medicines for treating advanced solid tumors with RAS mutations, particularly metastatic pancreatic cancer. April 2026 Phase 3 trials showed it significantly improves survival, demonstrating high potential as a first-line treatment.
Key Clinical Findings and Updates (as of April 2026):
- Mechanism: It acts as a RAS(ON) inhibitor, targeting mutated and wild-type RAS proteins (
) to disrupt cancer signaling.
- Breakthrough Results: Data from the RASolute 302 trial showed a substantial survival benefit in patients with previously treated metastatic pancreatic ductal adenocarcinoma (PDAC).
- High Response Rates: In trials, daraxonrasib combined with chemotherapy showed a 58% confirmed objective response rate (ORR) and 84% progression-free survival (PFS) at 6 months in untreated RAS-mutant metastatic pancreatic cancer.
- Safety Profile: Generally well-tolerated, with side effects including rash, diarrhea, stomatitis, and nausea.
- Recognition: Named the “2025 Molecule of the Year” by Drug Hunter for its, novel mechanism and clinical potential.
Daraxonrasib is currently being studied in the Phase 3 RASolute 303 trial for first-line treatment of pancreatic cancer.
Daraxonrasib (RMC-6236) is a RAS inhibitor drug. It is undergoing testing by Revolution Medicines to treat advanced solid tumors with RAS mutations, especially metastatic pancreatic ductal adenocarcinoma (PDAC) containing KRAS G12X mutations.[1] It received a breakthrough therapy designation from the U.S. Food and Drug Administration.[2]
Daraxonrasib is orally active and multi-selective RAS inhibitor. It uses a tri-complex mechanism to target the active, GTP-bound form of RAS proteins, including mutant and wild-type forms. Unlike conventional RAS inhibitors, it first binds to the chaperone-like protein cyclophilin A to form a complex, which then attaches to active RAS. This interaction blocks downstream effector binding and inhibits oncogenic signaling.[3]
In 2026, Daraxonrasib clinical trial completed a phase 3 clinical trial (RASolute 302) to assess efficacy compared to standard-of-care chemotherapy.[4] The trial met all primary and key secondary endpoints, including progression-free survival (PFS). The company reported median survival of 13.2 months with daraxonrasib vs. 6.7 months with standard chemotherapy. The hazard ratio for death was 0.40 (a 60% reduction in risk of death; p < 0.0001). Daraxonrasib was generally well tolerated with a manageable safety profile and no new safety signals.[5]
PAT
PAT
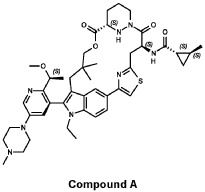
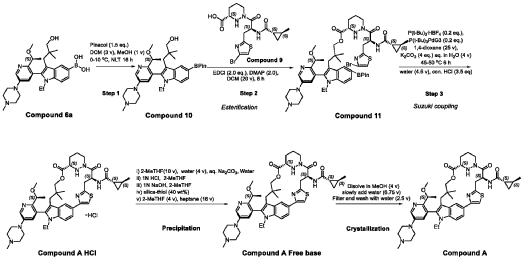
PATENT ATTORNEY DOCKET: 51432-038WO2 Part 4 – Purification of Compound A – (1S,2S)-N-[(7S,13S)-21-ethyl-20-{2-[(1S)-1- methoxyethyl]-5-(4-
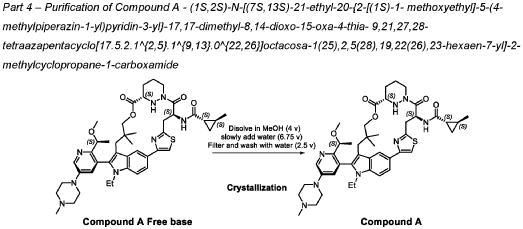
1.0equiv) at 25°C. The resulting suspension was stirred until solids were completely dissolved. The resulting methanol solution was filtered through microporous filter and transferred to another reactor. Then the reactor temperature was maintained at 25°C and slowly water (2.41kg, 1.0 V) water was added over a period of 30 minutes. The resulting cloudy solution was stirred for another 30 minutes at 25°C. Then a solution of methanol and water (3.42kg, 1:2, v/v) slowly over 1 hour. The resulting suspension was stirred for 2 hours at 25°C. Again, to the suspension additional water (2.48kg) slowly added over 1 hour. The final, suspension was stirred for additional 1 hour. Water (9.29kg, 3.75 V) was added to the suspension slowly over 2 hours and the mixture was stirred for at least for 16 hours at 25°C. The resulting suspension was filtered and washed with mixed solvent water: MeOH (3:2, v/v) twice (2x 2.2 kg), followed by water (4.91kg) washing. The wet cake was dried under reduced pressure and controlled humidity (temperature: 25 ± 5 ˚C, vacuum ≥ -0.085 MPa, humidity: 10%~20%) for 37 hours to afford Compound A as a white solid (2.68 kg, 99.4% a/a purity, 93.0% w/w assay, KF: 6.7%, 3.07 mol, 92% yield, Table 27).
PAT
- Synthesis of ras inhibitorsPublication Number: WO-2024216017-A2Priority Date: 2023-04-14
- Macrocycle compounds useful as kras inhibitorsPublication Number: WO-2024008834-A1Priority Date: 2022-07-08
- Ras inhibitorsPublication Number: US-11690915-B2Priority Date: 2020-09-15Grant Date: 2023-07-04
- Ras inhibitorsPublication Number: US-2023226186-A1Priority Date: 2020-09-15



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
References
- Cregg J, Edwards AV, Chang S, Lee BJ, Knox JE, Tomlinson AC, et al. (March 2025). “Discovery of Daraxonrasib (RMC-6236), a Potent and Orally Bioavailable RAS(ON) Multi-selective, Noncovalent Tri-complex Inhibitor for the Treatment of Patients with Multiple RAS-Addicted Cancers”. Journal of Medicinal Chemistry. 68 (6): 6064–6083. doi:10.1021/acs.jmedchem.4c02314. PMID 40056080.
- Sava J (July 1, 2025). “Daraxonrasib Earns FDA Breakthrough Status in Pancreatic Cancer”. Targeted Oncology. Retrieved October 12, 2025.
- Jiang J, Jiang L, Maldonato BJ, Wang Y, Holderfield M, Aronchik I, et al. (June 2024). “Translational and Therapeutic Evaluation of RAS-GTP Inhibition by RMC-6236 in RAS-Driven Cancers”. Cancer Discovery. 14 (6): 994–1017. doi:10.1158/2159-8290.CD-24-0027. PMC 11149917. PMID 38593348.
- Clinical trial number NCT05379985 at ClinicalTrials.gov
- Mast J (2026-04-13). “Revolution Medicines touts ‘unprecedented’ data for pancreatic cancer pill”. STAT. Retrieved 2026-04-13.
| Clinical data | |
|---|---|
| Other names | RMC-6236 |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2765081-21-6 |
| PubChem CID | 164726578 |
| IUPHAR/BPS | 13368 |
| ChemSpider | 115275938 |
| UNII | B6T47Y2UAP |
| KEGG | D13265 |
| ChEBI | CHEBI:746946 |
| Chemical and physical data | |
| Formula | C44H58N8O5S |
| Molar mass | 811.06 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
//////////daraxonrasib, anax labs, Kirsten rat sarcoma viral oncogene homolog inhibitor, antineoplastic, RMC-6236, RMC 6236, B6T47Y2UAP, RAS-IN-2,
Dabogratinib
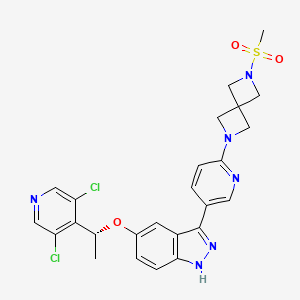
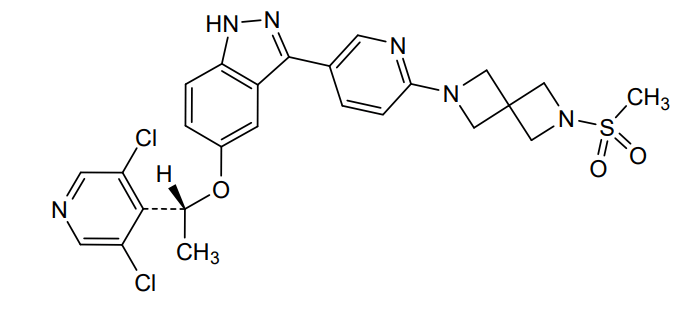
Dabogratinib
CAS 2800223-30-5
MF C25H24Cl2N6O3S, 559.5 g/mol
5-[(1R)-1-(3,5-dichloro-4-pyridinyl)ethoxy]-3-[6-(2-methylsulfonyl-2,6-diazaspiro[3.3]heptan-6-yl)-3-pyridinyl]-1H-indazole
(R)-5-(1-(3,5-Dichloropyridin-4-yl)ethoxy)-3-(6-(6-(methylsulfonyl)-2,6-diazaspiro[3.3]heptan-2-yl)pyridin-3-yl)-1H-indazole
[6-(5-{5-[(1R)-1-(3,5-dichloropyridin-4-yl)ethoxy]-1H-indazol-3-yl}pyridin-2-yl)-2,6-diazaspiro[3.3]heptan-2-yl](methyl)-λ6sulfanedioneTYRA-300
fibroblast growth factor receptor inhibitor, antineoplastic, TYRA-300, TYRA 300, A1AV2, FH245S2JZJ
Dabogratinib (TYRA-300) is an orally active, highly selective inhibitor of fibroblast growth factor receptor 3 (FGFR3), designed to treat cancers with FGFR3 alterations and genetic diseases like achondroplasia. It shows potent tumor growth inhibition in preclinical studies and early phase I/II (SURF301) clinical activity against advanced bladder cancer and metastatic urothelial carcinoma.
Key Aspects of Dabogratinib (TYRA-300)
- Mechanism: It acts as a selective inhibitor of FGFR3 with a high selectivity over other isoforms (FGFR1/2/4), which helps minimize toxicity.
- Target Indications: It is being developed for FGFR3-mutant cancers, including non-muscle invasive bladder cancer (NMIBC) and metastatic urothelial carcinoma, as well as pediatric achondroplasia.
- Preclinical Performance: Studies showed that it reduces tumor growth and drives tumor regression, especially in xenograft models with FGFR3-activating mutations (e.g., S249C).
- Clinical Trials:
- SURF301 (Phase I/II): Ongoing study, Tyra Biosciences reported early efficacy in patients with advanced metastatic urothelial carcinoma (mUC) harboring FGFR3 mutations/fusions.
- SURF302 (Phase II): Evaluating the drug in patients with FGFR3-altered, low-grade, intermediate-risk non–muscle invasive bladder cancer (NMIBC).
- BEACH301 (Phase II): Studying the drug in children with achondroplasia, as it is designed to increase long-bone growth.
- Properties: It is an orally bioavailable molecule with an IC50 of
for FGFR3.
Dabogratinib is an orally bioavailable, selective inhibitor of human fibroblast growth factor receptor 3 (FGFR3), with potential antineoplastic activity. Upon oral administration, dabogratinib specifically targets and binds to certain FGFR3 activating gene alterations, and specifically the gatekeeper mutants V555L/M. This blocks FGFR3-mediated signaling and leads to an inhibition of tumor cell proliferation in FGFR3-overexpressing cells. FGFR3, a receptor tyrosine kinase, is involved in angiogenesis and in the proliferation, differentiation, and survival of tumor cells. FGFR3 expression is associated with poor prognosis. It is overexpressed by certain tumor cell types.
- Efficacy and Safety of TYRA-300 in Participants With FGFR3 Altered Low Grade, Intermediate Risk Non-Muscle Invasive Bladder CancerCTID: NCT06995677Phase: Phase 2Status: RecruitingDate: 2026-04-09
- A Study of TYRA-300 in Children With Achondroplasia: BEACH301CTID: NCT06842355Phase: Phase 2Status: RecruitingDate: 2026-03-06
- Safety and Preliminary Anti-Tumor Activity of TYRA-300 in Advanced Urothelial Carcinoma and Other Solid Tumors With FGFR3 Gene AlterationsCTID: NCT05544552Phase: Phase 1/Phase 2Status: Active, not recruitingDate: 2026-01-12
PAT
Example 46. 5-[(1R)-1-(3,5-dichloro-4-pyridyl)ethoxy]-3-[6-(2-methylsulfonyl-2,6-diazaspiro[3.3]heptan-6-yl)-3-pyridyl]-1H-indazole
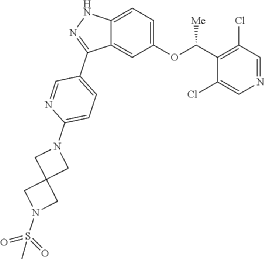
(5-[(1R)-1-(3,5-dichloro-4-pyridyl)ethoxy]-3-[6-(2-methylsulfonyl-2,6-diazaspiro[3.3]heptan-6-yl)-3-pyridyl]-1H-indazole. Triethylamine (20.5 uL, 0.148 mmol, 1.2 equiv) and methylsulfonyl chloride (9.5 uL, 0.123 mmol, 1.0 equiv) were sequentially added at room temperature to a solution of example 45 (59.0 mg, 0.123 mmol, 1 equiv) in anhydrous THE (3 mL). After stirring for 2 hours, the reaction mixture was concentrated under reduced pressure and diluted with saturated brine (30 mL) and dichloromethane (30 mL). The layers were separated. The organic layer was dried over sodium sulfate, filtered and concentrated under reduced pressure on to Celite (1 g). The product was purified on an Interchim automated chromatography system (RediSep Rf Gold HP C18, 15.5 g cartridge), eluting with a gradient of 0 to 100% acetonitrile in water. The fractions containing product were collected and lyophilized to give a white solid (45.0 mg, 65% yield). Analysis: LCMS: m/z=559.2 (M+H); 1H NMR (400 MHz, DMSO-d6) δ 13.02 (br s, 1H), 8.59 (s, 2H), 8.52 (dd, J=0.6, 2.2 Hz, 1H), 7.87 (dd, J=2.4, 8.6 Hz, 1H), 7.46 (d, J=8.9 Hz, 1H), 7.16 (d, J=2.1 Hz, 1H), 7.09 (dd, J=2.3, 9.0 Hz, 1H), 6.54 (dd, J=0.4, 8.6 Hz, 1H), 6.10 (q, J=6.6 Hz, 1H), 4.17 (s, 4H), 4.12 (s, 4H), 3.03 (s, 3H), 1.76 (d, J=6.6 Hz, 3H).
PAT
- Indazole compoundsPublication Number: TW-202241906-APriority Date: 2020-12-30
- Indazole compounds as kinase inhibitorsPublication Number: EP-4271673-A1Priority Date: 2020-12-30
- Indazole compounds as kinase inhibitorsPublication Number: WO-2022147246-A1Priority Date: 2020-12-30
- Indazole compounds as kinase inhibitorsPublication Number: US-12264149-B2Priority Date: 2020-12-30Grant Date: 2025-04-01
- Polymorphic compounds and uses thereofPublication Number: EP-4547670-A1Priority Date: 2022-06-29
- Indazole compounds as kinase inhibitorsPublication Number: US-12071428-B2Priority Date: 2020-12-30Grant Date: 2024-08-27
- Indazole Compounds as Kinase InhibitorsPublication Number: KR-20230152654-APriority Date: 2020-12-30
- Indazole compounds as kinase inhibitorsPublication Number: US-2024109865-A1Priority Date: 2020-12-30
- Indazole compounds as kinase inhibitorsPublication Number: US-2024208941-A1Priority Date: 2020-12-30
- Tyra-300 (5-[(1r)-1-(3,5-dichloro-4-pyridyl)ethoxy]-3-[6-(2-methylsulfonyl-2,6-diazaspiro[3.3]heptan-6-yl)-3-pyridyl]-1h-indazole ) in combination with a pd-1 or pd-l1 antagonist for use in the treatment of cancerPublication Number: WO-2025064744-A1Priority Date: 2023-09-22
- Fgfr inhibitors and methods of use thereofPublication Number: WO-2025061029-A1Priority Date: 2023-09-18
- Polymorphic compounds and uses thereofPublication Number: AU-2023300357-A1Priority Date: 2022-06-29
- Polymorphic compounds and uses thereofPublication Number: WO-2024006883-A1Priority Date: 2022-06-29
- Polymorphic compounds and uses thereofPublication Number: TW-202408493-APriority Date: 2022-06-29
ADVT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
Publication Name: Journal of Medicinal Chemistry
Publication Date: 2024-09-11
PMID: 39258897
DOI: 10.1021/acs.jmedchem.4c01531
////////dabogratinib, anax lab, fibroblast growth factor receptor inhibitor, antineoplastic, TYRA-300, TYRA 300, A1AV2, FH245S2JZJ
Claturafenib
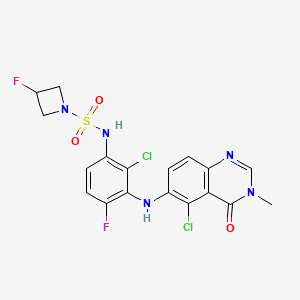
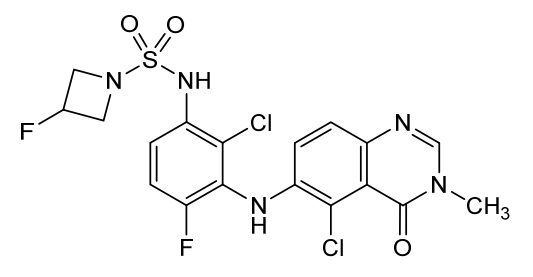
Claturafenib
CAS 2754408-94-9
MF C18H15Cl2F2N5O3S MW490.3 g/mol
N-[2-chloro-3-[(5-chloro-3-methyl-4-oxoquinazolin-6-yl)amino]-4-fluorophenyl]-3-fluoroazetidine-1-sulfonamide
N-{2-chloro-3-[(5-chloro-3-methyl-4-oxo-3,4-dihydroquinazolin-6-yl)amino]-4-fluorophenyl}-3-fluoroazetidine-1-sulfonamide
B-Raf (BRAF) inhibitor, antineoplastic, PF-07799933, PF 07799933, ARRY440, ARRY 440, PC35M52J8T
Claturafenib (development code PF-07799933) is an investigational cancer drug currently being developed by Pfizer. It is a selective, orally active pan-mutant BRAF inhibitor designed to treat advanced solid tumours with specific genetic alterations
Mechanism of Action
Claturafenib belongs to a class of drugs that target the MAPK/ERK signaling pathway, which is often hijacked by cancer cells to promote uncontrolled growth.
- Pan-Mutant Inhibition: Unlike first-generation BRAF inhibitors, claturafenib inhibits multiple classes of BRAF mutations, including Class 1 (V600), Class 2, and Class 3 alterations.
- Brain-Penetrant: It is designed to cross the blood-brain barrier, allowing it to potentially treat brain metastases or primary brain tumours.
- Dimer Disruption: It works by disrupting the formation of BRAF-containing dimers, which are responsible for signaling in many resistant or non-V600 mutant cancers.
- Selectivity: It is highly selective for mutant BRAF, significantly sparing normal (wild-type) cells to reduce off-target side effects.
🏥 Clinical Status
As of April 2026, claturafenib is in Phase 1 clinical trials.
- Target Indications: Advanced solid malignancies, including melanoma, colorectal cancer (CRC), and non-small cell lung cancer (NSCLC).
- Combination Therapy: It is being studied both as a single agent (monotherapy) and in combination with other drugs like binimetinib (a MEK inhibitor) or cetuximab (an EGFR inhibitor).
- Ongoing Study: Clinical trial NCT05355701 is currently evaluating its safety, dosage, and efficacy in patients whose disease has progressed on other treatments.
- A Study to Learn About the Study Medicine Called PF-07799933 in People With Advanced Solid Tumors With BRAF Alterations.CTID: NCT05355701Phase: Phase 1Status: RecruitingDate: 2026-03-27
- A Study to Learn About the Study Medicine Called PF-07799544 as Monotherapy or in Combination in People With Advanced Solid TumorsCTID: NCT05538130Phase: Phase 1Status: RecruitingDate: 2026-03-27
Claturafenib is an orally bioavailable class 1 and 2 inhibitor of the serine/threonine-protein kinase B-raf (BRAF) protein, with potential antineoplastic activity. Upon oral administration, claturafenib selectively binds to and inhibits the activity of class 1 and 2 BRAF alterations. This inhibits the proliferation of tumor cells which express these BRAF alterations. BRAF, a member of the raf family of serine/threonine protein kinases, plays a role in the regulation of mitogen-activated protein kinase (MAPK) and extracellular signal-regulated kinase (ERK) signaling pathways, which may be constitutively activated due to BRAF gene mutations. Mutated forms and fusions of BRAF are associated with a number of neoplastic diseases.
| Property | Value |
|---|---|
| Molecular Formula | |
| Molecular Weight | 490.31 g/mol |
| CAS Number | 2754408-94-9 |
| Other Names | ARRY-440, PF07799933 |
📍 Note: Claturafenib is an investigational compound and has not yet been approved by the FDA or other regulatory agencies for general use
SYN
SYN
Example 126
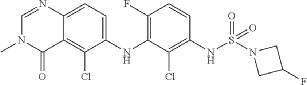
N-(2-chloro-3-((5-chloro-3-methyl-4-oxo-3,4-dihydroquinazolin-6-yl)amino)-4-fluorophenyl)-3-fluoroazetidine-1-sulfonamide
PAT
- 4-oxo-3,4-dihydroquinazolinon compounds for the treatment of braf-associated diseases and disordersPublication Number: CA-3186343-A1Priority Date: 2020-06-09
- Compounds for the treatment of BRAF-associated diseases and disordersPublication Number: US-12303509-B2Priority Date: 2020-06-09Grant Date: 2025-05-20
- 3,4-dihydro-2,7-naphthyridine-1,6(2H,7H)-dione compound as MEK inhibitorPublication Number: CN-117561255-APriority Date: 2021-03-31
- 4-oxo-3,4-dihydroquinazolinone compounds for the treatment of BRAF-related diseases and disordersPublication Number: KR-20230019944-APriority Date: 2020-06-09
- Compounds for the treatment of braf-associated diseases and disordersPublication Number: US-2022288074-A1Priority Date: 2020-06-09
- 4-oxo-3, 4-dihydroquinazolinone compounds for the treatment of BRAF related diseases and disordersPublication Number: CN-116096710-APriority Date: 2020-06-09
- 4-oxo-3,4-dihydroquinazolinon compounds for the treatment of braf-associated diseases and disordersPublication Number: EP-4161907-A1Priority Date: 2020-06-09
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
////////claturafenib, ANAX, B-Raf (BRAF) inhibitor, antineoplastic, PF-07799933, PF 07799933, ARRY440, ARRY 440, PC35M52J8T
Cirtociclib
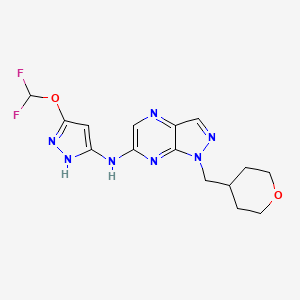
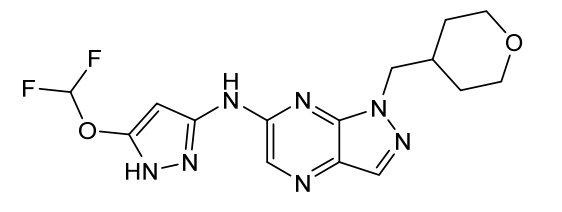
Cirtociclib
CAS 2888704-84-3
MF C15H17F2N7O2 MW365.34 g/mol
N-[3-(difluoromethoxy)-1H-pyrazol-5-yl]-1-(oxan-4-ylmethyl)pyrazolo[3,4-b]pyrazin-6-amine
N-[5-(difluoromethoxy)-1H-pyrazol-3-yl]-1-[(oxan-4-yl)methyl]-1H-pyrazolo[3,4-b]pyrazin-6-amine
cyclin-dependent kinase inhibitor, antineoplastic, BLU-222, BLU 222, BLU 170298, U93X72ED47, CDK2 Inhibitor BLU-222
Cirtociclib (also known as BLU-222) is an investigational drug that acts as a highly selective inhibitor of cyclin-dependent kinase 2 (CDK2). It is being developed by Blueprint Therapeutics for the treatment of advanced solid tumours, particularly those with genetic drivers like CCNE1 amplification, which are common in certain ovarian and breast cancers
Certociclib is a small molecule drug. Certociclib is under investigation in clinical trial NCT05252416 ((VELA) Study of BLU-222 in Advanced Solid Tumors). Certociclib has a monoisotopic molecular weight of 365.14 Da.
Certociclib is an orally bioavailable inhibitor of cyclin-dependent kinase 2 (CDK2), with potential antineoplastic activity. Upon administration, certociclib selectively targets, binds to and inhibits the activity of CDK2. This may lead to cell cycle arrest, the induction of apoptosis, and the inhibition of tumor cell proliferation. CDK2, a serine/threonine kinase that plays an important role in the regulation of cell cycle progression and cellular proliferation, is overexpressed in certain tumor cells.
How It Works
- Targeting CDK2: It binds to CDK2, a protein that regulates the cell cycle.
- Cell Cycle Arrest: By inhibiting CDK2, the drug causes G1 arrest, preventing cancer cells from replicating.
- Selectivity: It is designed to be “best-in-class” for its high selectivity for CDK2 over other kinases like CDK1, CDK4, or CDK6.
Therapeutic Potential
- Ovarian Cancer: Specifically targets high-grade serous ovarian cancer where CCNE1 is amplified.
- Breast Cancer: Shows promise in treating hormone receptor-positive/HER2-negative (HR+/HER2-) breast cancer, especially when the cancer has become resistant to existing CDK4/6 inhibitors.
- Combination Therapy: Researchers are testing it alongside other drugs, such as palbociclib, ribociclib, or chemotherapy agents like carboplatin, to enhance efficacy.
Current Status
- Clinical Trials: It is currently being evaluated in a Phase 1/2 clinical trial known as the VELA study (NCT05252416) for patients with advanced solid tumours.
- Research Status: It is not yet approved for general medical use and is primarily available for research and clinical trial participants.
(VELA) Study of BLU-222 in Advanced Solid Tumors
CTID: NCT05252416
Phase: Phase 1
Status: Terminated
Date: 2025-11-28
🌟 Key Point: Cirtociclib represents a new generation of precision medicine aimed at overcoming resistance to standard cancer therapies by specifically targeting the CDK2 pathway
PAT
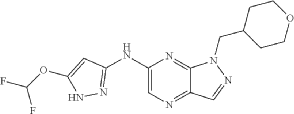
The structure of one CDK2 inhibitor, referred to herein as “a compound of formula (I)” or N-(5-(difluoromethoxy)-lH-pyrazol-3-yl)-l-((tetrahydro-2H-pyran-4-yl)methyl)-lH-pyrazolo[3,4-b]pyrazin-6-amine is shown below:
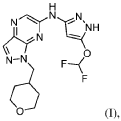
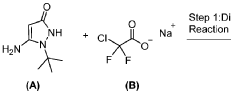
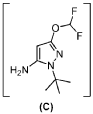
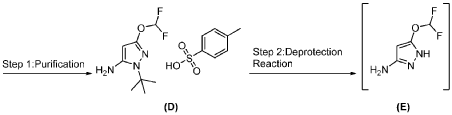
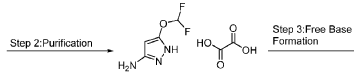
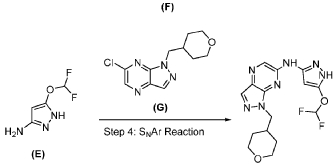
PAT
Example 2
N-(5-(difluoromethoxy)-1H-pyrazol-3-yl)-1-((tetrahydro-2H-pyran-4-yl)methyl)-1H-pyrazolo[3,4-b]pyrazin-6-amine
A mixture of 6-chloro-1-((tetrahydro-2H-pyran-4-yl)methyl)-1H-pyrazolo[3,4-b]pyrazine (Preparation 87, 780 mg, 3.09 mmol), 5-(difluoromethoxy)-1H-pyrazol-3-amine (554 mg, 3.72 mmol), tBuXphos Pd G3 (150 mg, 0.19 mmol) and KOAc (892 mg, 9.08 mmol) in dioxane (15 mL) was stirred at 90° C. for 6 h under N 2. The reaction mixture was evaporated to dryness in vacuo and the residue was purified by prep-HPLC-4 to afford the title compound as a white solid (361.4 mg, 32%). LCMS m/z=366 [M+H] +; 1H NMR (400 MHz, DMSO-d 6) δ: 12.21 (s, 1H), 10.82 (s, 1H), 8.19 (s, 1H), 8.17 (s, 1H), 7.32 (t, 1H), 5.98 (d, 1H), 4.40 (d, 2H), 3.87-3.75 (m, 2H), 3.29-3.16 (m, 2H), 2.24-2.11 (m, 1H), 1.46-1.29 (m, 4H).
PAT
- The cdk2 inhibitor blu-222 for treatment of cancerPublication Number: WO-2024168298-A1Priority Date: 2023-02-10
- Solid forms of a cdk2 inhibitorPublication Number: WO-2024148083-A1Priority Date: 2023-01-04
- Cdk2 inhibitorsPublication Number: US-2023322791-A1Priority Date: 2021-06-28
- Cdk2 inhibitorsPublication Number: US-2023159535-A1Priority Date: 2021-06-28
- CDK2 inhibitorsPublication Number: US-11970498-B2Priority Date: 2021-06-28Grant Date: 2024-04-30
- Cdk2 inhibitorsPublication Number: US-2024383902-A1Priority Date: 2021-06-28
- CDK2 inhibitorsPublication Number: US-11932648-B2Priority Date: 2021-06-28Grant Date: 2024-03-19



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
/////////cirtociclib, cyclin-dependent kinase inhibitor, antineoplastic, BLU-222, BLU 222, BLU 170298, U93X72ED47, CDK2 Inhibitor BLU-222
Catadegbrutinib
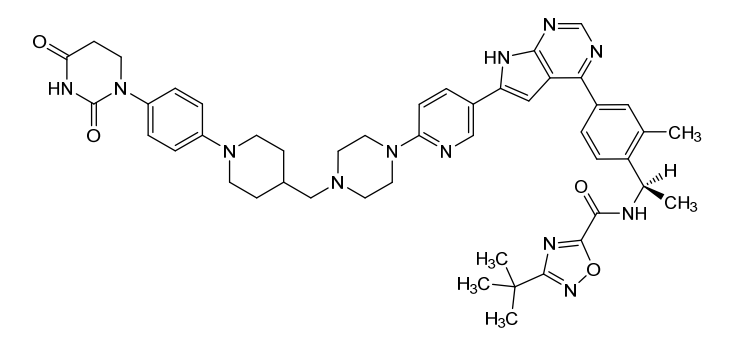
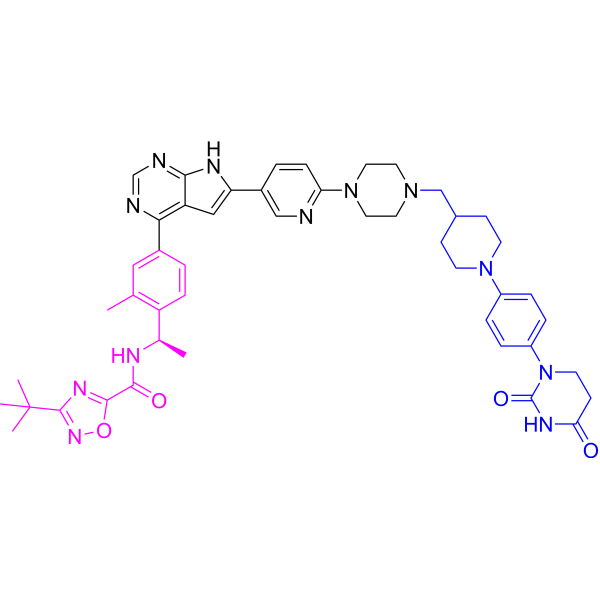
Catadegbrutinib
CAS 2736508-60-2
MF C47H54N12O4 MW851.0 g/mol
3-tert-butyl-N-[(1R)-1-[4-[6-[6-[4-[[1-[4-(2,4-dioxo-1,3-diazinan-1-yl)phenyl]piperidin-4-yl]methyl]piperazin-1-yl]-3-pyridinyl]-7H-pyrrolo[2,3-d]pyrimidin-4-yl]-2-methylphenyl]ethyl]-1,2,4-oxadiazole-5-carboxamide
3-tert-butyl-N-{(1R)-1-[13-methyl-82,84-dioxo-27H-2(4,6)-pyrrolo[2,3-d]pyrimidina-8(1)-[1,3]diazinana-4(1,4)-piperazina3(5,2)-pyridina-6(4,1)-piperidina-1(1),7(1,4)-dibenzenaoctaphan-14-yl]ethyl}-1,2,4-oxadiazole-5-carboxamide
Bruton tyrosine kinase degrader, antineoplastic, BGB-16673, BGB 16673, PF6GPZ4DYT, BTK-IN-29, Tacabrutideg
Catadegbrutinib (BGB-16673) is an orally active, potent Bruton’s tyrosine kinase (BTK) degrader, or chimeric degradation activator compound (CDAC). It works by targeting BTK for proteasomal degradation, showing high efficacy against wild-type and mutated forms (including C481S) in B-cell malignancies. It is under investigation for cancers such as CLL, SLL, and MCL.
Key Details About Catadegbrutinib
- Mechanism of Action: As a PROTAC-class molecule, it binds to BTK and recruits E3 ubiquitin ligase, causing polyubiquitination and degradation of the protein.
- Target Potency: It shows strong degradation activity, with a
of
(concentration required for 50% degradation) and a
binding
of
.
- Clinical Potential: Developed for B-cell malignancies (chronic lymphocytic leukemia, mantle cell lymphoma) that have developed resistance to covalent and non-covalent BTK inhibitors.
- Synonyms/Codes: BGB-16673, BGB-116673, BTK-IN-29, and recently listed in WHO proposed INN as tacabrutideg.
- Status: Used primarily in research for treating B-cell malignancies, lymphomas, and potentially autoimmune diseases.
Catadegbrutinib is designed to overcome resistance mechanism challenges seen with existing BTK inhibitors.
SYN
Example 14: (R) -3- (tert-butyl) -N- (1- (4- (6- (6- (4- ( (1- (4- (2, 4-dioxotetrahydropyrimidin-1 (2H) -yl) phenyl) piperidin-4-yl) methyl) piperazin-1-yl) pyridin-3-yl) -7H-pyrrolo [2, 3-d] pyrimidin-4-yl) -2-methylphenyl) ethyl) -1, 2, 4-oxadiazole-5-carboxamide
[0357]
Step 1: tert-butyl 4- (5- (4-chloro-7H-pyrrolo [2, 3-d] pyrimidin-6-yl) pyridin-2-yl) piperazine-1- carboxylate
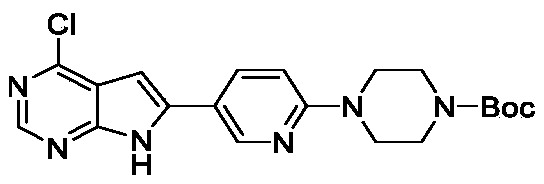
A mixture of 4-chloro-6-iodo-7H-pyrrolo [2, 3-d] pyrimidine (3 g, 10.73 mmol) , tert-butyl 4- (5- (4, 4, 5, 5-tetramethyl-1, 3, 2-dioxaborolan-2-yl) pyridin-2-yl) piperazine-1-carboxylate (4.18 g, 10.73 mmol) , Na 2CO 3(1.25 g, 11.80 mmol) and Pd (dppf) Cl 2(0.39 g, 0.537 mmol) in dioxane (120 mL) and H 2O (20 mL) was stirred in a sealed tube at 85 ℃ overnight. After cooling, the reaction mixture was filtered and the solid was washed with 20 mL of MeOH and dried under vacuum to afford the product (4.05 g, 91%) . [M+H] += 415.0.
[0360]
Step 2: tert-butyl (R) -4- (5- (4- (4- (1- (3- (tert-butyl) -1, 2, 4-oxadiazole-5-carboxamido) ethyl) -3- methylphenyl) -7H-pyrrolo [2, 3-d] pyrimidin-6-yl) pyridin-2-yl) piperazine-1-carboxylate
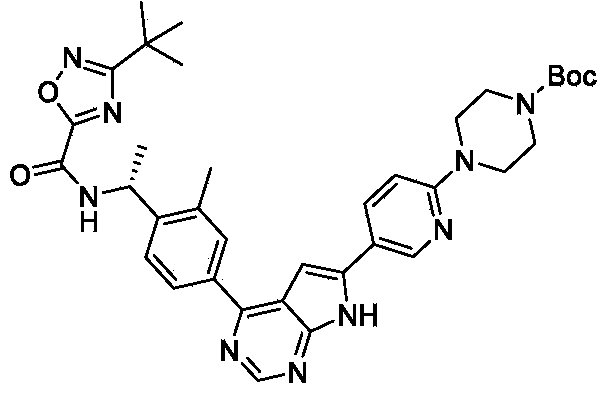
A mixture of tert-butyl 4- (5- (4-chloro-7H-pyrrolo [2, 3-d] pyrimidin-6-yl) pyridin-2-yl) piperazine-1-carboxylate (0.9 g, 2.17 mmol) , (R) -3- (tert-butyl) -N- (1- (2-methyl-4- (4, 4, 5, 5-tetramethyl-1, 3, 2-dioxaborolan-2-yl) phenyl) ethyl) -1, 2, 4-oxadiazole-5-carboxamide (0.94 g, 2.28 mmol) , Na 2CO 3(0.46 g, 4.34 mmol) and Pd (dppf) Cl 2(79.3 mg, 0.108mmol) in dioxane (60 mL) and H 2O (10 mL) was stirred in a sealed tube at 100 ℃ overnight. After cooling, the reaction mixture was filtered and the solid was washed with 5 mL of MeOH and dried under vacuum to afford the product (1.02 g, 70.6%) . [M+H] += 666.0.
[0363]
Step 3: (R) -3- (tert-butyl) -N- (1- (2-methyl-4- (6- (6- (piperazin-1-yl) pyridin-3-yl) -7H- pyrrolo [2, 3-d] pyrimidin-4-yl) phenyl) ethyl) -1, 2, 4-oxadiazole-5-carboxamide, hydrogen chloride salt
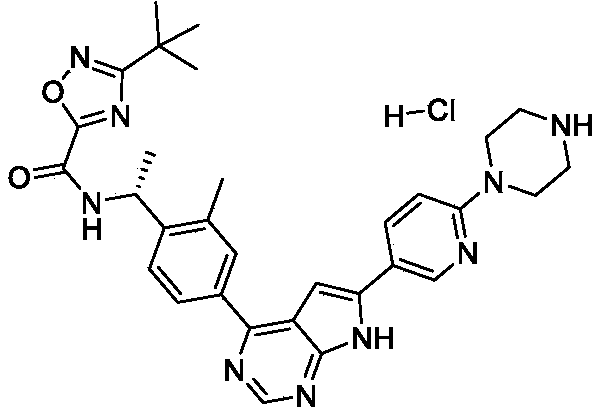
To a solution of tert-butyl (R) -4- (5- (4- (4- (1- (3- (tert-butyl) -1, 2, 4-oxadiazole-5-carboxamido) ethyl) -3-methylphenyl) -7H-pyrrolo [2, 3-d] pyrimidin-6-yl) pyridin-2-yl) piperazine-1-carboxylate (1.02 g, 1.53 mmol) in DCM (50 mL) in a round bottom flask was added HCl in dioxane (4 N, 35 mL) at 0 ℃. The mixture was stirred for 2 h at 20 ℃. The precipitate was collected with filtration and dried in vacuum to afford the product (0.92 g, 100%) . 1H NMR (400 MHz, DMSO) δ H13.53 (s, 1H) , 10.06 (d, J = 7.5 Hz, 1H) , 9.33 (s, 2H) , 9.00 (s, 1H) , 8.93 (s, 1H) , 8.35 (d, J = 8.7 Hz, 1H) , 8.05 (d, J = 8.1 Hz, 1H) , 7.99 (s, 1H) , 7.75 (d, J = 8.0 Hz, 1H) , 7.55 (s, 1H) , 7.12 (d, J = 8.9 Hz, 1H) , 5.50-5.28 (m, 1H) , 3.89 (s, 4H) , 3.20 (s, 4H) , 2.57 (s, 3H) , 1.56 (d, J = 6.9 Hz, 3H) , 1.38 (s, 9H) . [M+H] += 566.3.
[0366]
Step 4: (R) -3- (tert-butyl) -N- (1- (4- (6- (6- (4- ( (1- (4- (2, 4-dioxotetrahydropyrimidin-1 (2H) – yl) phenyl) piperidin-4-yl) methyl) piperazin-1-yl) pyridin-3-yl) -7H-pyrrolo [2, 3-d] pyrimidin-4-yl) -2- methylphenyl) ethyl) -1, 2, 4-oxadiazole-5-carboxamide
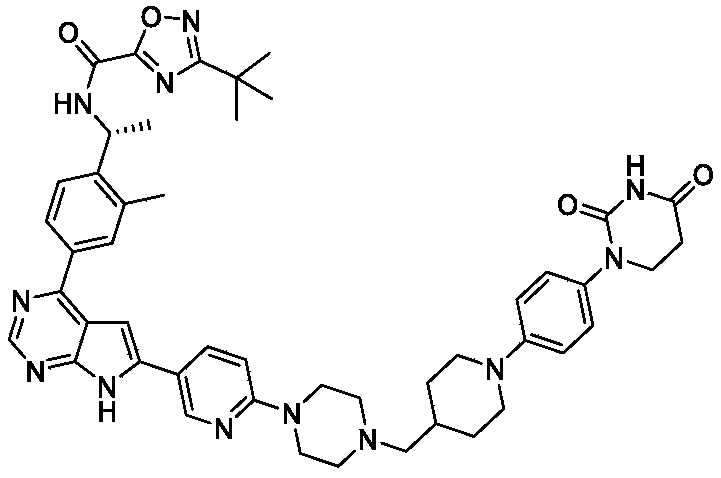
A mixture of (R) -3- (tert-butyl) -N- (1- (2-methyl-4- (6- (6- (piperazin-1-yl) pyridin-3-yl) -7H-pyrrolo [2, 3-d] pyrimidin-4-yl) phenyl) ethyl) -1, 2, 4-oxadiazole-5-carboxamide, hydrogen chloride salt (0.06 g, 0.1 mmol) , 1- (4- (2, 4-dioxotetrahydropyrimidin-1 (2H) -yl) phenyl) piperidine-4-carbaldehyde (0.033 g, 0.11 mmol) and NaOAc (8.2 mg, 0.1 mmol) in DCM/EtOH (30 mL/10 mL) was stirred in a round bottom flask for 1 h at 20 ℃. Then NaBH 3CN (12.6 mg, 0.2 mmol) was added. The mixture was stirred overnight at 20 ℃. The mixture was concentrated to dryness and purified with silica gel column chromatography (MeOH in DCM from 0%to 12%gradient elution) to give the product (0.049 g, 57.8%) . 1H NMR (400 MHz, DMSO) δ H12.60 (s, 1H) , 10.27 (s, 1H) , 9.97 (d, J =6.1 Hz, 1H) , 8.79 (d, J = 18.7 Hz, 2H) , 8.18 (d, J = 7.8 Hz, 1H) , 8.09 (d, J = 7.0 Hz, 1H) , 8.04 (s, 1H) , 7.67 (d, J = 7.7 Hz, 1H) , 7.30 (s, 1H) , 7.13 (d, J = 6.9 Hz, 2H) , 6.97-6.92 (m, 3H) , 5.41-5.34 (m, 1H) , 3.71-3.68 (m, 4H) , 3.64-3.56 (m, 4H) , 2.70-2.64 (m, 4H) , 2.53 (s, 3H) , 2.47-2.43 (m, 4H) , 2.25-2.19 (m, 2H) , 1.84-1.81 (m, 2H) , 1.75-1.70 (m, 1H) , 1.56 (t, J = 9.1 Hz, 3H) , 1.37 (s, 9H) , 1.28-1.18 (m, 2H) .
PAT
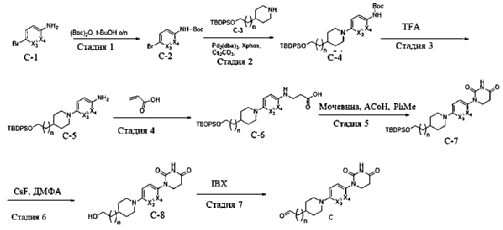
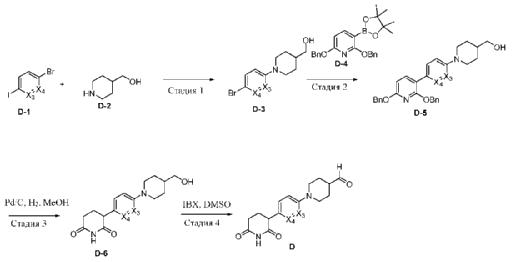
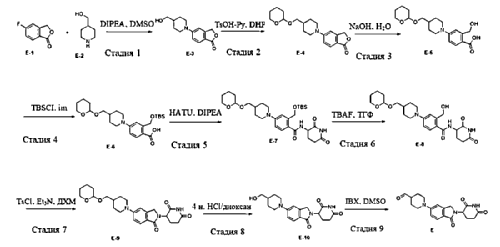
PAT
- Degradation of bruton’s tyrosine kinase (btk) by conjugation of btk inhibitors with e3 ligase ligand and methods of usePublication Number: WO-2021219070-A1Priority Date: 2020-04-30
- Degradation of Bruton’s tyrosine kinase (BTK) by conjugation of BTK inhibitors to E3 ligase ligands and methods of use thereofPublication Number: CN-115485278-APriority Date: 2020-04-30
- Degradation of bruton’s tyrosine kinase (btk) by conjugation of btk inidbitors with e3 ligase ligand and methods of usePublication Number: US-2023167118-A1Priority Date: 2020-04-30



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- [1]. Wang H, et al. BGB-16673, a selective BTK degrader, exhibits deeper inhibition of cancer cell signaling pathways and better efficacy in MCL models. Blood, 2024, 144: 5833.[2]. Wu Y, et al. Translational modeling to predict human pharmacokinetics and pharmacodynamics of a Bruton’s tyrosine kinase-targeted protein degrader BGB-16673. Br J Pharmacol. 2024 Dec;181(24):4973-4987. [Content Brief][3]. Hexiang Wang, et al. Degradation of bruton’s tyrosine kinase (btk) by conjugation of btk inhibitors with e3 ligase ligand and methods of use. WO2021219070A1. 2021-11-04.
/////////catadegbrutinib, Bruton tyrosine kinase degrader, antineoplastic, BGB-16673, BGB 16673, PF6GPZ4DYT, BTK-IN-29, Tacabrutideg
Birelentinib
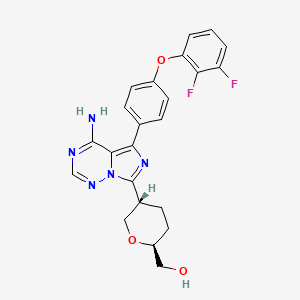
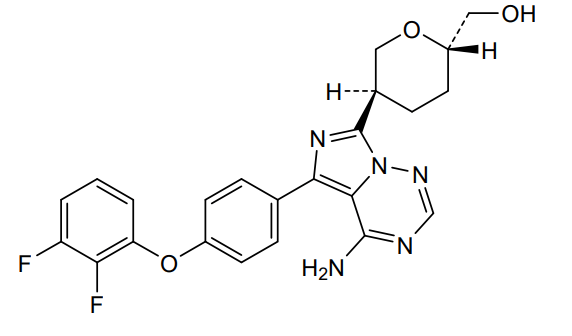
Birelentinib
CAS 2662512-15-2
MF C23H21F2N5O3 MW453.4 g/mol
[(2S,5S)-5-[4-amino-5-[4-(2,3-difluorophenoxy)phenyl]imidazo[5,1-f][1,2,4]triazin-7-yl]oxan-2-yl]methanol
[(2S,5S)-5-{4-amino-5-[4-(2,3-difluorophenoxy)phenyl]imidazo[5,1-f][1,2,4]triazin-7-yl}oxan-2-yl]methanol
tyrosine kinase inhibitor, antineoplastic, DZD8586, DZD 8586, Fast Track designation, BTK-IN-30, Z2F599L9GD
Birelentinib (also known as DZD8586) is a first-in-class, non-covalent dual inhibitor of LYN (lymphocyte-specific protein tyrosine kinase) and BTK (Bruton’s tyrosine kinase).
It is currently being developed by Dizal Pharmaceutical as an oral therapy for various B-cell malignancies.
Clinical Status and FDA Designations
As of late 2025, birelentinib has received significant attention for its potential in treating resistant blood cancers:
- Fast Track Designation: In August 2025, the U.S. FDA granted Fast Track designation to birelentinib for adult patients with relapsed or refractory (R/R) chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL).
- Target Population: It is specifically intended for those who have failed at least two prior therapies, including a covalent BTK inhibitor and a BCL-2 inhibitor.
- Key Trials: It is being evaluated in multiple studies, including the Phase 3 Tai-Shan6 trial comparing it against standard treatments like bendamustine and rituximab.
Unique Therapeutic Properties
Birelentinib is designed to overcome common drug resistance mechanisms found in existing treatments:
- Overcoming Resistance: It targets both BTK-dependent pathways (including the common C481X mutation) and BTK-independent B-cell receptor (BCR) signaling pathways.
- Blood-Brain Barrier (BBB) Penetration: A notable feature is its ability to fully penetrate the blood-brain barrier, which may offer therapeutic benefits for patients with central nervous system (CNS) involvement.
- Efficacy: Early Phase 1/2 data presented at the ASH Annual Meeting and EHA Congress in 2025 showed an Objective Response Rate (ORR) of 84.2% in heavily pretreated patients
Birelentinib is an orally bioavailable non-covalent dual inhibitor of tyrosine-protein kinases Lyn (LYN) and BTK (Bruton’s tyrosine kinase; Bruton agammaglobulinemia tyrosine kinase), with potential antineoplastic activity. Upon oral administration, birelentinib targets and inhibits both LYN and BTK, thereby blocking both BTK-dependent and BTK-independent B-cell antigen receptor (BCR) signaling pathways. This prevents the proliferation of malignant B-cells in which the BCR signaling pathway is overactivated. Birelentinib is able to cross the blood-brain barrier (BBB) and thus potentially useful in the treatment of central nervous system (CNS) metastases
SYN’
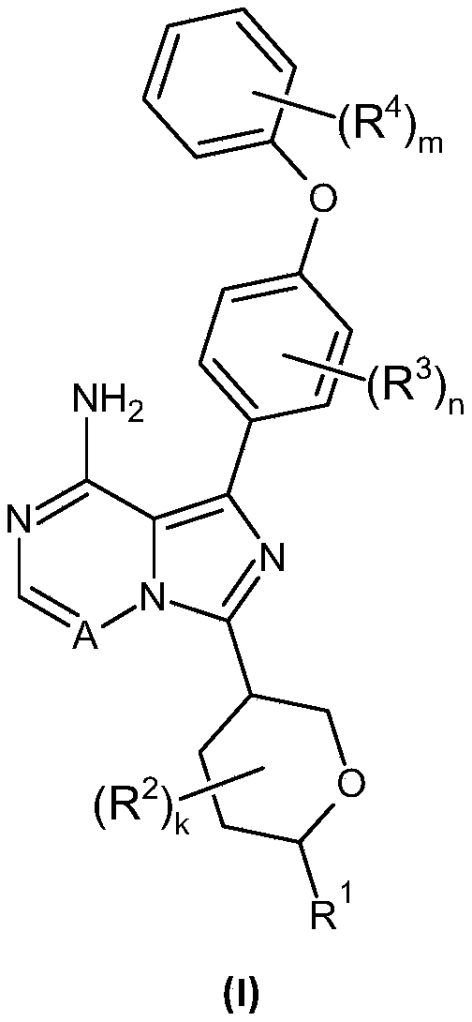
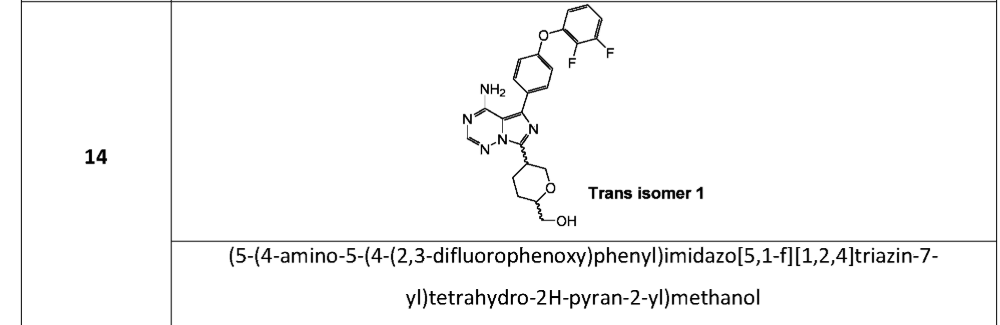
SYN



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
Publication Number: WO-2021136219-A1
Priority Date: 2020-01-02
/////////birelentinib, tyrosine kinase inhibitor, antineoplastic, DZD8586, DZD 8586, Fast Track designation, BTK-IN-30, Z2F599L9GD
Balomenib
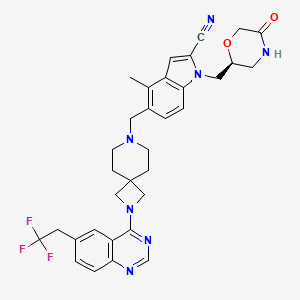
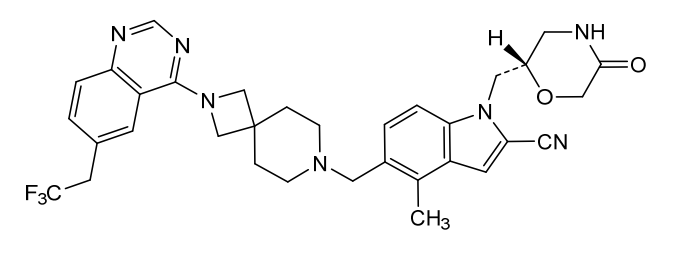
Balomenib
CAS 2939850-17-4
MF C33H34F3N7O2 MW617.7 g/mol
4-methyl-1-[[(2S)-5-oxomorpholin-2-yl]methyl]-5-[[2-[6-(2,2,2-trifluoroethyl)quinazolin-4-yl]-2,7-diazaspiro[3.5]nonan-7-yl]methyl]indole-2-carbonitrile
- 1H-Indole-2-carbonitrile, 4-methyl-1-[[(2S)-5-oxo-2-morpholinyl]methyl]-5-[[2-[6-(2,2,2-trifluoroethyl)-4-quinazolinyl]-2,7-diazaspiro[3.5]non-7-yl]methyl]-
- 4-methyl-1-[(2S)-5- oxomorpholin-2- yl]methyl]-5- [[2-[6-(2,2,2- trifluoroethyl) quinazolin-4-yl]-2,7- diazaspiro[3.5]nonan- 7-yl]methyl]indole-2- carbonitrile
4-methyl-1-{[(2S)-5-oxomorpholin-2-yl]methyl}-5-({2-[6-(2,2,2-trifluoroethyl)quinazolin-4-yl]-2,7-diazaspiro[3.5]nonan-7-
yl}methyl)-1H-indole-2-carbonitrile
menin inhibitor, antineoplastic, ZE63-0302, 3BEG4BWN8E
Balomenib (also known as ZE63-0302) is an oral, small-molecule menin inhibitor currently in clinical development for metabolic and oncological conditions. It works by disrupting the protein-protein interaction between menin and KMT2A (formerly MLL), a mechanism that plays a critical role in both pancreatic beta-cell function and certain types of leukemia.
Key Therapeutic Areas
- Type 2 Diabetes (T2D): Balomenib is being investigated as a potentially disease-modifying treatment to improve pancreatic beta-cell function and survival. As of late 2025, it has advanced into Phase 1b clinical trials specifically for adults with T2D to evaluate its effects on fasting glucose, insulin dynamics, and HbA1c.
- Oncology (AML): It is also a candidate for treating acute myeloid leukemia (AML) with KMT2A rearrangements or NPM1 mutations. Preclinical data suggests it may be more effective against resistance mutations than earlier menin inhibitors.
Development and Safety
- Corporate Development: The drug was originally developed by Eilean Therapeutics. It is now the lead program for Clywedog Therapeutics, which is merging with Barinthus Biotherapeutics to focus on metabolic diseases.
- Safety Profile: Early trial results indicate a favorable safety profile. Notably, it was designed to minimize QTc prolongation (heart rhythm issues), a side effect common in other menin inhibitors.
- Сlinical Study Aiming to Evaluate the Safety, Tolerability, Pharmacokinetics and Pharmacodynamics of Single and Multiple Ascending Doses of ZE63-0302 in Healthy VolunteersCTID: NCT06780124Phase: Phase 1Status: CompletedDate: 2026-01-22
- Study to Assess Safety, Tolerability, PK, and PD of Multiple Doses of ZE63-0302 Administrated Orally in T2DM Patients.CTID: NCT07234864Phase: Phase 1Status: RecruitingDate: 2026-01-22
SYN
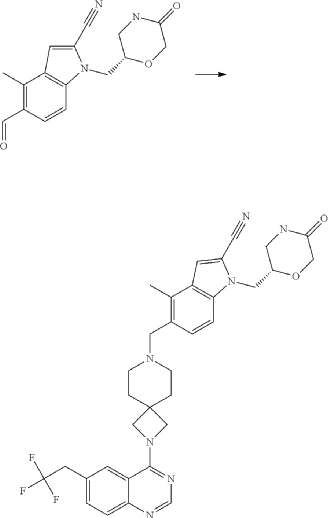
Example 46. 4-Methyl-1-{[(2S)-5-oxomorpholin-2-yl]methyl}-5-({2-[6-(2,2,2-trifluoroethyl)quinazolin-4-yl]-2,7-diazaspiro[3.5]non-7-yl}methyl)-1H-indole-2-carbonitrile (Compound 102)
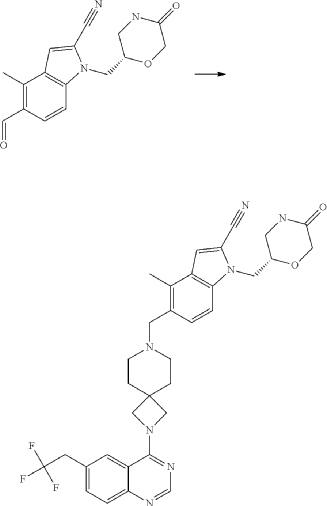
Compound was prepared using procedure described in the Example 45 and 5-formyl-4-methyl-1-{[(2S)-5-oxomorpholin-2-yl]methyl}-1H-indole-2-carbonitrile P177 instead of 5-formyl-4-methyl-1-{[(2R)-5-oxomorpholin-2-yl]methyl}-1H-indole-2-carbonitrile P176. Compound 102 was obtained with yield 49%. 1H NMR (400 MHz, DMSO-d 6), δ: 8.46 (s, 1H), 7.99 (m, 2H), 7.73 (m, 2H), 7.52 (m, 1H), 7.46 (d, J=5.6 Hz, 1H), 7.31 (d, J=4.8 Hz, 1H), 4.54 (m, 1H), 4.20 (m, 2H), 4.05 (m, 1H), 3.90 (m, 4H), 3.52 (m, 2H), 3.35 (m, 1H), 3.17 (m, 1H), 2.39 (m, 2H), 1.79 (m, 4H). LCMS (ESI) [MH] +: 618.
PAT
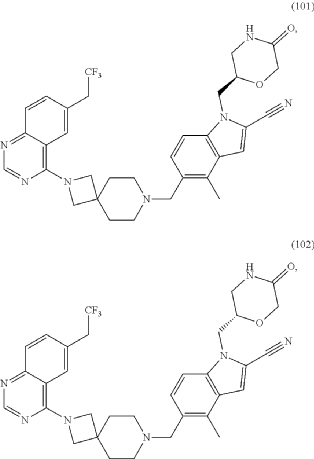
Example 46. 4-Methyl-1-{[(2S)-5-oxomorpholin-2-yl]methyl}-5-({2-[6-(2,2,2-trifluoroethyl)quinazolin-4-yl]-2,7-diazaspiro[3.5]non-7-yl}methyl)-1H-indole-2-carbonitrile (Compound 102)



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Inhibitors of menin-mll interactionPublication Number: WO-2023107696-A2Priority Date: 2021-12-09
- Inhibitors of menin-mll interactionPublication Number: US-2025163061-A1Priority Date: 2021-12-09
- Inhibitors of menin-mll interactionPublication Number: EP-4444300-A2Priority Date: 2021-12-09
///////////balomenib, menin inhibitor, antineoplastic, ZE63-0302, 3BEG4BWN8E
Atebimetinib
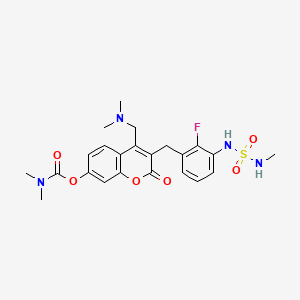
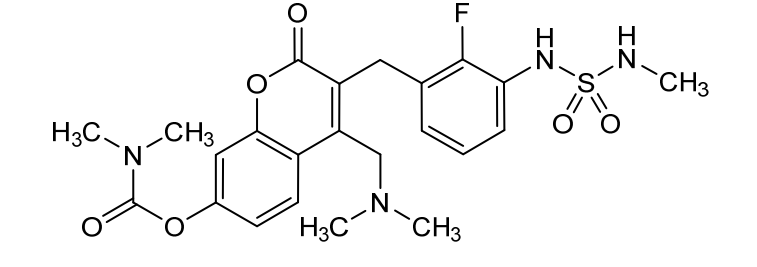
Atebimetinib
CAS 2669009-92-9
MF C23H27FN4O6S MW506.5 g/mol
[4-[(dimethylamino)methyl]-3-[[2-fluoro-3-(methylsulfamoylamino)phenyl]methyl]-2-oxochromen-7-yl] N,N-dimethylcarbamate
4-[(dimethylamino)methyl]-3-({2-fluoro-3-[(methylsulfamoyl)amino]phenyl}methyl)-2-oxo-2H-1-benzopyran-7-yl
dimethylcarbamate
MEK tyrosine kinase inhibitor, antineoplastic, IMM-104, IMM 104, Fast Track designation, TEL9243A3N
Atebimetinib (IMM-104) is an investigational oral, deep cyclic inhibitor (DCI) that targets the MAP kinase (MAPK) pathway in solid tumors, particularly RAS-mutant pancreatic cancer. Designed for rapid, pulsatile inhibition to minimize resistance and side effects, it is currently in Phase 2a trials, having shown promising, durable tumor shrinkage and high 1-year survival rates.
Key Aspects of Atebimetinib:
- Mechanism of Action: As a DCI, it works differently from standard inhibitors by targeting MAPK with a short half-life, allowing for rapid “pulsing” that suppresses tumor growth while permitting healthy cells to recover, thus improving tolerability.
- Targeted Cancers: Primarily aimed at RAS-mutant advanced or metastatic solid tumors, including pancreatic ductal adenocarcinoma (PDAC).
- Clinical Trial Results: In a Phase 2a study (NCT05585320), the combination of atebimetinib with modified chemotherapy showed a 64% overall survival (OS) rate at 12 months for first-line pancreatic cancer patients.
- Fast Track Designation: In 2024, the FDA granted fast track designation for atebimetinib to treat patients with pancreatic adenocarcinoma who have progressed after one line of therapy.
- Advantage over Traditional Inhibitors: It is designed to avoid typical MAP kinase inhibitor adverse events like pyrexia (fever) while overcoming the rapid resistance often seen in other therapies.
Atebimetinib is being developed by Immuneering Corporation.
Development Status
- FDA Designations: In 2024, the FDA granted atebimetinib Fast Track designation for patients with pancreatic adenocarcinoma (PDAC) who have progressed after one line of treatment.
- Future Plans: A global registrational Phase 3 trial, named MAPKeeper 301, is planned to begin dosing patients in mid-2026.
Clinical Trial Results (Phase 2a)
Recent data from the Phase 2a trial (as of early 2026) showed significant survival benefits when combined with modified chemotherapy (gemcitabine and nab-paclitaxel) for first-line pancreatic cancer:
- Overall Survival (OS): Reported at 94% at 6 months, 86% at 9 months, and 64% at 12 months. This is roughly double the 1-year survival rate typically seen with standard chemotherapy alone (~35%).
- Progression-Free Survival (PFS): Median PFS reached 8.5 months.
- Disease Control Rate: Approximately 81% of patients achieved disease control.
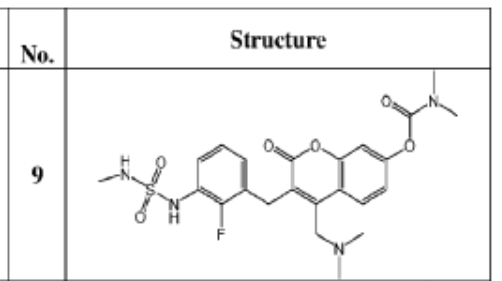
SYN
WO2023076991 COMBINATION THERAPY FOR TREATING ABNORMAL CELL GROWTH
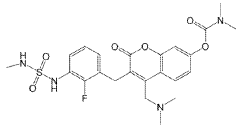
SYN
WO2025010293 MEK IMMUNE ONCOLOGY INHIBITOR PHARMACEUTICAL COMPOSITIONS
EXAMPLE 1A
Synthesis of Compound A
[0198] Compound A was prepared in 1 step:
[0199] 4-(bromomethyl)-3-(2-fluoro-3-((N-methylsulfamoyl)amino)benzyl)-2-oxo-2H-chromen-7-yl dimethylcarbamate (22.22 g, 34.79 mmol) was suspended in methanol. Dimethylamine 2M was added and the formed reaction mixture was stirred until full conversion was observed. After full conversion the reaction was concentrated under reduced pressure. IM HC1 was added to the residue and the water layer was extracted with CH2CI2. The water layer was made basic with solid Na CCE. The basic water layer was extracted with CH2CI2. The organic layer from the basic extraction was washed with brine, dried over Na2SO4, filtered and concentrated under reduced pressure to obtain the title compound (13.23 g, 25.7 mmol, yield: 74%) as a light yellow amorphous solid.
[0200] Yield: Compound A was isolated as a light yellow solid (74% over 1 step). Analysis: LCMS (Method T): tR = 1.53 min; m/z calculated for [M+H]+ = 507.2, found = 507.2; 1H NMR (400 MHz, DMSO) d 9.38 (s, 1H), 8.08 (d, J = 8.8 Hz, 1H), 7.28 (td, J = 8.0, 1.6 Hz, 1H), 7.25 – 7.18 (m, 2H), 7.15 (dd, J = 8.8, 2.4 Hz, 1H), 7.00 (t, J = 7.9 Hz, 1H), 6.90 – 6.77 (m, 1H), 4.04 (s, 2H), 3.64 (s, 2H), 3.06 (s, 3H), 2.93 (s, 3H), 2.52 (d, J = 4.9 Hz, 3H), 2.19 (s, 6H).
ADVT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Mek immune oncology inhibitor pharmaceutical compositionsPublication Number: WO-2025010293-A2Priority Date: 2023-07-03
- Inhibiting mitogen-activated protein (map)/erk kinase (mek)1 and mek2 and related methods of treatmentPublication Number: WO-2024220440-A1Priority Date: 2023-04-17
- Methods of treating cancer with a ras mutationPublication Number: WO-2024186693-A1Priority Date: 2023-03-03
- Combination therapy for treating abnormal cell growthPublication Number: WO-2023235356-A1Priority Date: 2022-06-03
- Combination therapy for treating abnormal cell growthPublication Number: WO-2023147297-A2Priority Date: 2022-01-25
- Methods of treating abnormal cell growthPublication Number: WO-2023081676-A1Priority Date: 2021-11-02
- Combination therapy for treating abnormal cell growthPublication Number: WO-2023076991-A1Priority Date: 2021-10-28
- Mek inhibitors and therapeutic uses thereofPublication Number: US-2023119327-A1Priority Date: 2020-01-10
//////atebimetinib, FLAX LAB, antineoplastic, IMM-104, IMM 104, Fast Track designation, TEL9243A3N
Anvumetostat
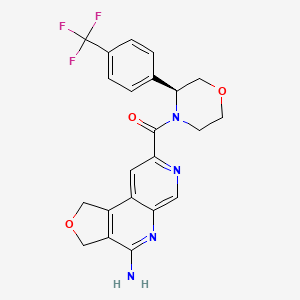
Anvumetostat
CAS 2790567-82-5
MF C22H19F3N4O3 MW444.4 g/mol
(4-amino-1,3-dihydrofuro[3,4-c][1,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl]methanone
(4-amino-1,3-dihydrofuro[3,4-c][1,7]naphthyridin-8-yl){(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl}methanone
antineoplastic, AMG 193, QAT649EJ5E, PRMT5-IN-27,
Anvumetostat (also known as AMG 193) is an orally available, small-molecule inhibitor of protein arginine methyltransferase 5 (PRMT5), primarily being developed for the treatment of advanced solid tumours with MTAP-null (methylthioadenosine phosphorylase-deficient) mutations.
Mechanism of Action
- Targeting PRMT5: It is a potent and selective MTA-cooperative inhibitor of PRMT5.
- Synthetic Lethality: In cells where the MTAP gene is deleted (a common occurrence in various cancers), a metabolite called MTA (methylthioadenosine) accumulates. Anvumetostat selectively binds to the PRMT5-MTA complex, inhibiting its methyltransferase activity.
- Cellular Impact: By blocking PRMT5, the drug reduces the methylation of arginine residues in histones (H2A, H3, and H4), which can lead to decreased growth or death of cancer cells.
Clinical Development
Anvumetostat was initially developed by Amgen, Inc. and is currently in clinical trials. Institute (.gov) +1
- Current Status: As of early 2026, it is in Phase 2 of global research and development.
- Study Focus: Trials are evaluating its efficacy both as a monotherapy and in combination with other treatments for adult patients with metastatic or locally advanced MTAP-null cancers.
Key Identifiers
- Alternate Names: AMG 193, AMG-193.
- Chemical Class: Orally bioavailable small molecule.
- Genetic Biomarker: Specifically targets cancers with MTAP-null status
Anvumetostat is an orally available small molecule inhibitor of protein arginine methyltransferase 5 (PRMT5), with potential antiproliferative and antineoplastic activities. Upon oral administration, anvumetostat selectively binds to PRMT5 and inhibits its function. By inhibiting its methyltransferase activity, levels of both monomethylated and dimethylated arginine residues in histones H2A, H3 and H4 are decreased. This modulates the expression of genes involved in several cellular processes, including cellular proliferation. This may increase the expression of antiproliferative genes and/or decrease the expression of genes that promote cell proliferation, which may lead to decreased growth of rapidly proliferating cells, including cancer cells. PRMT5, a type II methyltransferase that catalyzes the formation of both omega-N monomethylarginine (MMA) and symmetric dimethylarginine (sDMA) on histones and a variety of other protein substrates involved in signal transduction and cellular transcription, is overexpressed in several neoplasms. Elevated levels are associated with decreased patient survival. Methylthioadenosine phosphorylase (MTAP) is deleted in certain cancer cells leading to an accumulation of methylthioadenosine (MTA). As MTA inhibits PRMT5, MTAP-null cancer cells are specifically sensitive to PRMT5 inhibitors.
SYN
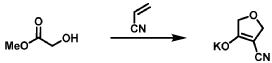
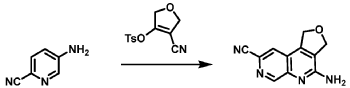
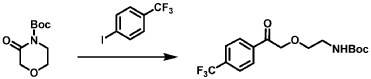
[0163] Examples 481 and 482: (4-amino-l,3-dihydrofuro[3,4-c][l,7]naphthyridin-8-yl)(3-(4- (trifluoromethyl)phenyl)morpholino)methanone
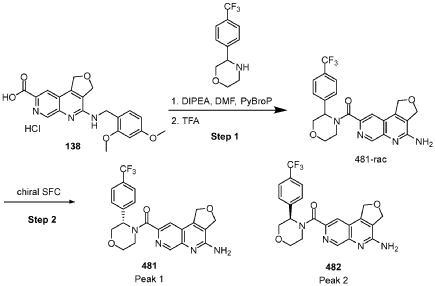
[0164] Step 1: To a solution of 3-(4-(trifluoromethyl)phenyl)morpholine (0.100 g, 0.432 mmol, Enamine), 4-((2,4-dimethoxybenzyl)amino)-l,3-dihydrofuro[3,4-c][l,7]naphthyridine-8-carboxylic acid hydrochloride (138) (0.271 g, 0.649 mmol) and l,l’-dimethyltriethylamine (0.559 g, 0.755 mL, 4.32 mmol, Sigma- Aldrich Corporation) in DMF (4 mL) was added bromotripyrrolidinophosphonium hexafluorophosphate (0.202 g, 0.432 mmol, Sigma-Aldrich Corporation) and the resulting mixture was heated at 50 °C for 30 min. The reaction was brought to rt, diluted with water, sat.NaHCCh and extracted with EtOAc (3x). The combined organics were dried over Na2SO4, filtered and concentrated. The residue was then chromatographed on silica gel using 0-50% 3:1 EtOAc/EtOH in heptane to afford (4-((2,4-dimethoxybenzyl)amino)- 1 ,3 -dihy drofuro [3 ,4-c] [ 1 ,7]naphthyridin-8-y 1) (3 – (4 -(trifluoromethyl)phenyl)morpholino)methanone (0.160 g, 0.269 mmol, 62.2% yield) as a light yellow solid, m/z (ESI): 595 (M+H)+.
[0165] To a solution of (4-((2,4-dimethoxybenzyl)amino)-l,3-dihydrofuro[3,4-c] [l,7]naphthyridin-8-yl)(3-(4-(trifluoromethyl)phenyl)morpholino)methanone (0.160 g, 0.269 mmol, 62.2 % yield) in DCM (2 mL) was added TFA (14.80 g, 10 mL, 130 mmol, Aldrich) and the resulting mixture was heated at 50 °C for 1 h. The reaction was concentrated, washed with 10% Na2CO3 and extracted with DCM. The combined organics were concentrated and chromatographed on silica gel using 0-50% 3:1 EtOAc/EtOH in heptane to afford (4-amino-l,3-dihydrofuro[3,4-c][l,7]naphthyridin-8-yl)(3-(4-(trifluoromethyl)phenyl)morpholino)methanone as the TFA salt (0.078 g, 0.140 mmol, 32.3% yield) as an off-white solid, m/z (ESI): 445 (M+H)+.
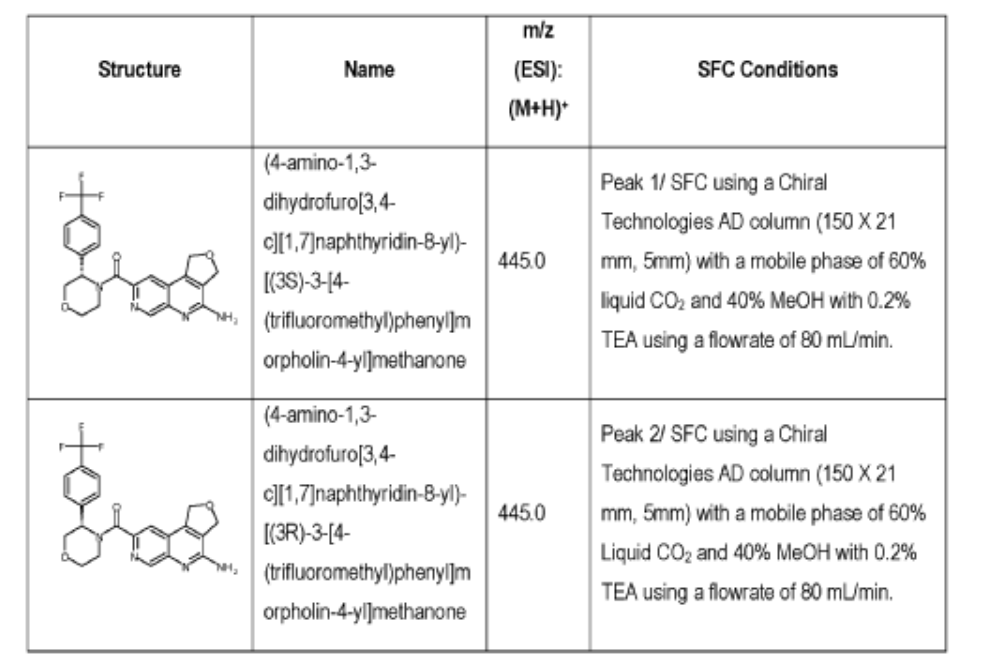
[0166] Step 2: (S)-(4-amino-l,3-dihydrofuro[3,4-c][l,7]naphthyridin-8-yl)(3-(4- (trifluoromethy l)phenyl)morpholino)methanone and (R)-(4-amino- 1 ,3 -dihy drofuro [3,4-c][l,7]naphthyridin-8-yl)(3-(4-(trifluoromethyl)phenyl)morpholino)methanone
(4-amino-l,3-dihydrofuro[3,4-c][l,7]naphthyridin-8-yl)(3-(4-(trifluoromethyl)phenyl)morpholino)methanone 2,2,2-trifluoroacetate were separated via preparative SFC using a Chiral Technologies AD column (150 x 21 mm, 5mm) with a mobile phase of 60% Liquid CO2 and 40% MeOH with 0.2% TEA using a flowrate of 80 mL/min to generate peak 1, (S)-(4-amino-l,3-dihydrofuro[3,4-c][l,7]naphthyridin-8-yl)(3-(4-(trifluoromethyl)phenyl)morpholino)methanone with an ee of >99%, and peak 2, (R)-(4-amino-l,3-dihydrofuro[3,4-c][l,7]naphthyridin-8-yl)(3-(4-(trifluoromethyl)phenyl)morpholino)methanone with an ee of 99.28%. Peak assignment determined by
SFC with AD column with 60% Liquid CO2 and 40% MeOH with 0.2% TEA and absolute
stereochemistry was arbitrarily assigned.
Peak 1: (S)-(4-amino-l,3-dihydrofuro[3,4-c][l,7]naphthyridin-8-yl)(3-(4-(trifluoromethyl)phenyl)morpholino)methanone (481) as a white solid . m/z (ESI): 445 (M+H)+. NMR 1H (400 MHz, DMSO-d6) 5 ppm 8.67 – 9.03 (m, 1 H), 7.85 (s, 1 H), 7.77 (br s, 4 H), 7.07 (br s, 2 H), 5.75 (s, 1 H), 5.37 (br s, 2 H), 5.04 (br s, 2 H), 4.46 – 4.61 (m, 1 H), 3.89 (br dd, J=12.2, 3.3 Hz, 4 H), 3.58 (br d, ./=5,8 Hz, 1 H). 19F NMR (377 MHz, DMSO-d6 ) 5 ppm -60.90 (br s, 3 F).
Peak 2: (R)-(4-amino- 1 ,3 -dihy drofuro [3 ,4-c] [ 1 ,7]naphthyridin-8-yl)(3 -(4-(trifluoromethyl)phenyl)morpholino)methanone (482) as a white solid, m/z (ESI): 445 (M+H)+. NMR 1H (400 MHz, DMSO-d6) 5 ppm 8.88 (br s, 1 H), 7.85 (s, 1 H), 7.77 (br d, J=1.7 Hz, 4 H), 7.07 (br s, 2 H),
5.69 – 5.78 (m, 1 H), 5.37 (br s, 2 H), 5.04 (br s, 2 H), 4.45 – 4.61 (m, 1 H), 3.89 (br dd, J=12.4, 3.3 Hz, 4 H), 3.51 – 3.64 (m, 1 H). 19F NMR (DMSO-d6, 377 MHz) 5 -60.90 (s, 3 F).
SYN
Example 4. Synthesis of Compound I – (4-amino-1 ,3-di hydrofuro[3,4-c][1 ,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl]methanone
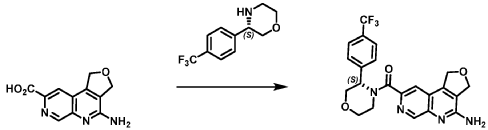
Reaction Scale 1
[0137] 4-Amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridine-8-carboxylic acid (1.0 kg, 4.3 mol, 1.0 equiv), (3S)-3-[4-(trifluoromethyl)phenyl]morpholine (1.2 kg, 5.2 mmol, 1.2- equiv), and DMF, (6.6 kg, 7.0 V) were charged to a clean, dry reactor. To the mixture was added triethylamine (1.1 Kg, 13.8 mol, 2.6 equiv). The mixture was cooled to 10 ± 5 °C and O-(benzotriazol-1-yl)-N,N,N’,N’-tetramethyluronium tetrafluoroborate (TBTU) (1.67 kg, 5.2 mol, 1.2 equiv) was added slowly. Next, an additional amount of DMF (0.94 Kg, 1 V) was added. The reaction mixture was warmed to 25 ± 5 °C and stirred over 18 hours. Water (1 .0 kg, 1 V) was charged followed by MeCN (1 .6 kg, 2 V) and the reaction mass was warmed to 45 °C. Next, water (7.0 Kg, 7 V) was added over 30 min. A seed lot of 4-amino-1 ,3-dihydrofuro[3,4-c][1,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl]methanone (10 g, 22 mmol, 0.01 equiv), was charged and the mixture was stirred at 45 °C for over 2 hours before being cooled to 20 °C over 10 hours. Water (12.0 kg, 12 V) was added over 2 hours at 20 °C and further stirred for over 4 hours before being filtered. The reactor was rinsed with a mixture of 10% DMF in water (9.83 kg, 10 V) and the resulting rinse mixture was used to wash the cake. The reactor was rinsed with a mixture of water (10.0k kg, 10 V) and the resulting rinse mixture was used to wash the cake. This rinsing and washing protocol was repeated once more with water (10.0k kg, 10V). The cake was dried under vacuum with a stream of nitrogen to afford (4-amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridin-8-yl)-[(3S)-3-[4-
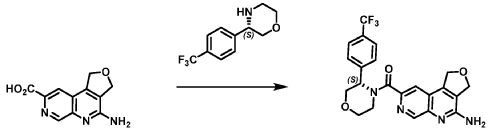
(trifluoromethyl)phenyl]morpholin-4-yl]methanone. LCMS: 445.20 1H NMR (400 MHz, DMS0-d6 at 130 °C): 8.87 (s, 1 H), 7.80 (s, 1 H), 7.73 (d, 0=8.7 Hz, 2H), 7.71 (d, 0=8.7 Hz, 2H), 6.58 (br s, 2H), 5.72 (br s, 1 H), 5.38 (m, 2H), 5.09 (t, 0=3.5 Hz, 2H), 4.44 (br d, 0=12.3 Hz, 1 H), 4.08 (br d, 0=13.4 Hz, 1 H), 3.96 (dd, 0=12.3, 3.7 Hz, 1 H), 3.86 (br dd, 0=11 .4, 3.0 Hz, 1 H), 3.66 (td, 0=11 .4, 3.0 Hz, 1 H), 3.28 (m, 1 H).
Reaction Scale 2
[0138] 4-Amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridine-8-carboxylic acid (85.0 g, 352.2 mmol, 1.0 equiv), (3S)-3-[4-(trifluoromethyl)phenyl]morpholine (99.6 g, 422.6 mmol, 1.2- equiv), and DMF, (674 mL, 8.7 mol, 7.9 V) were charged to a clean, dry 5 L reactor. To the mixture was added 1 -methylimidazole (75.2 g, 916.2 mmol, 2.6 equiv). The mixture was cooled to 0 °C and N,N,N’,N’-tetramethylchloroformamidinium hexafluorophosphate (TCFH) (118.6 g, 422.6 mmol, 1.2 equiv) was added slowly. Next, an additional amount of DMF (170 mL, 2 V) was added at 0 °C. The reaction mixture was warmed to 25 °C and stirred overnight. Next, the reaction mass was warmed to 45 °C and 2-methyltetrahydrofuran, (169.2 mL, 2 V) was added followed by slow addition of water (850 mL, 10 V) over 30 min by addition funnel. A seed lot of 4-amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl]methanone (1.6 g, 3.5 mmol, 0.1 equiv), was charged as a slurry in a 1 :1 v/v of DMF and water (31 .3 mL) and the mixture was stirred at 45 °C for approximately 12 hrs. Water (510 mL, 6 V) was added over 1 h 10 min by addition funnel and the mixture was further stirred at 45°C for 30 min before being filtered. The reactor was rinsed with water (340 mL, 4 V) and the resulting rinse mixture was used to wash the cake. This rinsing and washing protocol was repeated twice more. The cake was dried under vacuum with a stream of nitrogen to afford (4-amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl]methanone. LCMS: 445.20 1H NMR (400 MHz, DMSO-d6 at 130 °C): 8.87 (s, 1 H), 7.80 (s, 1 H), 7.73 (d, J=8.7 Hz, 2H), 7.71 (d, J=8.7 Hz, 2H), 6.58 (br s, 2H), 5.72 (br s, 1 H), 5.38 (m, 2H), 5.09 (t, J=3.5 Hz, 2H), 4.44 (br d, J=12.3 Hz, 1 H), 4.08 (br d, J=13.4 Hz, 1 H), 3.96 (dd, J=12.3, 3.7 Hz, 1 H), 3.86 (br dd, J=11.4, 3.0 Hz, 1 H), 3.66 (td, J=11.4, 3.0 Hz, 1 H), 3.28 (m, 1 H).
Reaction Scale 3:
[0139] 4-Amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridine-8-carboxylic acid (Compound A’) (20.0 g, 86.5 mmol, 1.0 equiv) was added to dimethylsulfoxide (400 mL) at 20 °C. To the mixture was added 1 ,T-carbonyldiimidazole (15.4 g, 95.2 mmol, 1.1 equiv) and the mixture was heated to 60 °C for 1 hour. A solution of (S)-3-(4-(trifluoromethyl)phenyl)morpholin-4-ium chloride (25.5 g, 95.2 mmol, 1.1 equiv) and dimethylsulfoxide (40 mL) was added, and the mixture was heated to 80 °C for 11 hours. The reaction mixture was cooled to 35 °C, then water (265 mL) was added, then the batch was cooled to 20 °C. The reaction was filtered, washed with 40% water:DMSO (80 mL), then washed with water (100 mL). The cake was dried under vacuum with a stream of nitrogen to afford (4-amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl]methanone (Compound I). LCMS: 445.20 1H NMR (400 MHz, DMSO-d6 at 130 °C): 8.87 (s, 1 H), 7.80 (s, 1 H), 7.73 (d, J=8.7 Hz, 2H), 7.71 (d, J=8.7 Hz, 2H), 6.58 (br s, 2H), 5.72 (br s, 1 H), 5.38 (m, 2H), 5.09 (t, >3.5 Hz, 2H), 4.44 (br d, >12.3 Hz, 1H), 4.08 (br d, >13.4 Hz, 1 H), 3.96 (dd, >12.3, 3.7 Hz, 1 H), 3.86 (br dd, >11 .4, 3.0 Hz, 1 H), 3.66 (td, >11 .4, 3.0 Hz, 1 H), 3.28 (m, 1 H).
Recrystallization of Compound I
[0140] A clean, dry 5 L reactor was charged with (4-amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl]methanone (279.7 g, 0.6 mol, 1.0 equiv) followed by acetone (6.2 L,
22 V). The mixture was stirred at 40 °C for 15 minutes before cooling to 25 °C. The reactor was discharged into a flask and the reactor was rinsed with acetone and the process stream was polish-filtered back into the reactor.
The reactor jacket was set to 65 °C and the reaction volume was reduced to approximately 6 V by distillation at atmospheric pressure, crystallization was observed. The reaction temperature was set to cool to 20 °C over two hours. Heptane (2.8 L, 10 V) was added over two hours. The slurry was filtered and the cake was washed twice with a 4:1 Heptane/acetone mix (750 mL, 3 V each) and dried under vacuum with a nitrogen purge to afford (4-amino-1,3-dihydrofuro[3,4-c][1,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl] methanone.


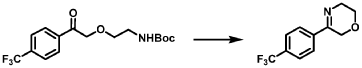

ADVT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Process for synthesizing naphthyridine derivatives and intermediates thereofPublication Number: EP-4396170-A1Priority Date: 2021-08-30
- PRMTS inhibitorsPublication Number: CN-116888120-APriority Date: 2020-12-16
- Prmts inhibitorsPublication Number: WO-2022132914-A1Priority Date: 2020-12-16
- Prmts inhibitorsPublication Number: EP-4263545-A1Priority Date: 2020-12-16
- Mta-cooperative prmt5 inhibitors for use in the treatment of cancerPublication Number: EP-4572760-A1Priority Date: 2022-08-15
- Cancer treatments using mta-cooperative prmt5 inhibitorsPublication Number: WO-2023196545-A1Priority Date: 2022-04-08
- Process for the synthesis of naphthyridine derivatives and intermediates thereofPublication Number: CN-117897379-APriority Date: 2021-08-30
- Process for synthesizing naphthyridine derivatives and intermediates thereofPublication Number: WO-2023034786-A1Priority Date: 2021-08-30
- Process for Synthesizing Naphthyridine Derivatives and Intermediates ThereofPublication Number: US-2024360147-A1Priority Date: 2021-08-30
- Prmt5 inhibitor for use in cancer therapyPublication Number: WO-2024170488-A1Priority Date: 2023-02-13
- Cancer treatments using a prmt5 inhibitor and a mat2a inhibitorPublication Number: WO-2024118897-A1Priority Date: 2022-11-30
- Cancer treatments using a prmt5 inhibitor and a mat2a inhibitorPublication Number: EP-4626435-A1Priority Date: 2022-11-30
- MTA synergizes with PRMT5 inhibitors for cancer treatmentPublication Number: CN-119730853-APriority Date: 2022-08-15
- Mta-cooperative prmt5 inhibitors for use in the treatment of cancerPublication Number: WO-2024038004-A1Priority Date: 2022-08-15
////////anvumetostat, ANAX LAB, antineoplastic, AMG 193, QAT649EJ5E, PRMT5-IN-27,

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










