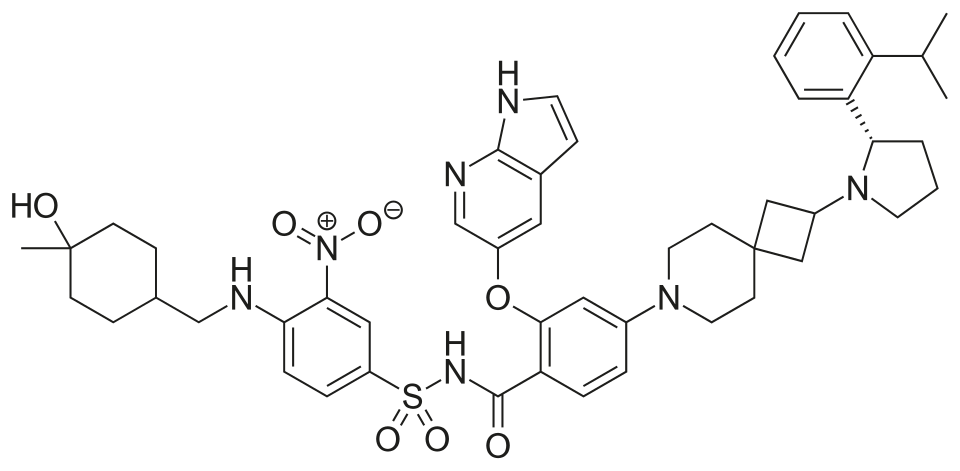
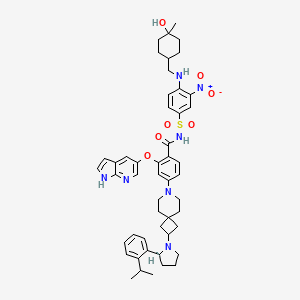
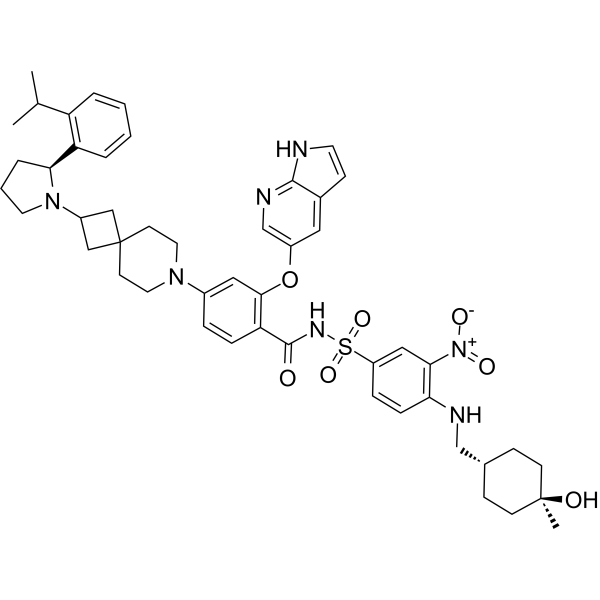
Sonrotoclax
CAS 2383086-06-2
MW 890.1 g/mol, MFC49H59N7O7S
FDA APPROVED 5/13/2026, Beqalzi, APPROVALS 2026, BGB-11417, BGB 11417, 30R67U9KYS
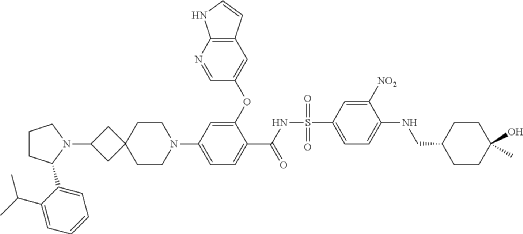
N-[4-[(4-hydroxy-4-methylcyclohexyl)methylamino]-3-nitrophenyl]sulfonyl-4-[2-[(2S)-2-(2-propan-2-ylphenyl)pyrrolidin-1-yl]-7-azaspiro[3.5]nonan-7-yl]-2-(1H-pyrrolo[2,3-b]pyridin-5-yloxy)benzamide
- 2-((1H-Pyrrolo[2,3-b]pyridin-5-yl)oxy)-N-((4-((((1r,4r)-4-hydroxy-4-methylcyclohexyl)methyl)amino)-3-nitrophenyl)sulfonyl)-4-(2-((S)-2-(2-isopropylphenyl)pyrrolidin-1-yl)-7-azaspiro[3.5]nonan-7-yl)benzamide
- 2-((1H-Pyrrolo[2,3-b]pyridin-5-yl)oxy)-N-((4-(((trans-4-hydroxy-4-methylcyclohexyl)methyl)amino)-3-nitrophenyl)sulfonyl)-4-(2-((S)-2-(2-isopropylphenyl)pyrrolidin-1-yl)-7-azaspiro[3.5]nonan-7-yl)benzamide
To treat adults with relapsed or refractory mantle cell lymphoma after at least two lines of systemic therapy, including a Bruton’s tyrosine kinase inhibitor
Sonrotoclax is a potent, orally active Bcl2 inhibitor. Sonrotoclax has effective cell killing effect against a variety of lymphoma and leukemia cell lines.
Regulatory Status & Primary Indication
On May 13, 2026, the U.S. Food and Drug Administration (FDA) granted accelerated approval to sonrotoclax for treating adult patients with relapsed or refractory mantle cell lymphoma (MCL). [1]
- Eligibility Requirement: Patients must have undergone at least two prior lines of systemic therapy, which must include a Bruton’s tyrosine kinase (BTK) inhibitor.
- Clinical Performance: In the supporting Phase 1/2 BGB-11417-201 trial, sonrotoclax demonstrated an overall response rate (ORR) of 52% and a median time to response of 1.9 months
Sonrotoclax is an orally bioavailable inhibitor of the anti-apoptotic protein B-cell lymphoma 2 (Bcl-2), with potential pro-apoptotic and antineoplastic activities. Upon oral administration, sonrotoclax specifically binds to and inhibits the activity of the pro-survival protein Bcl-2. This restores apoptotic processes and inhibits cell proliferation in Bcl-2-overexpressing tumor cells. Bcl-2, a protein that belongs to the Bcl-2 family, is overexpressed in various tumor cell types and plays an important role in the negative regulation of apoptosis. Its tumor expression is associated with increased drug resistance and cancer cell survival.
Sonrotoclax is an investigational new drug that is being evaluated for the treatment of hematologic malignancies, particularly chronic lymphocytic leukemia (CLL) and small lymphocytic lymphoma (SLL).[1] It is a potent and selective BCL2 inhibitor that can overcome resistance associated with BCL2 mutations, such as the G101V variant, which limits the effectiveness of first-generation inhibitors like venetoclax.[2]
SYN
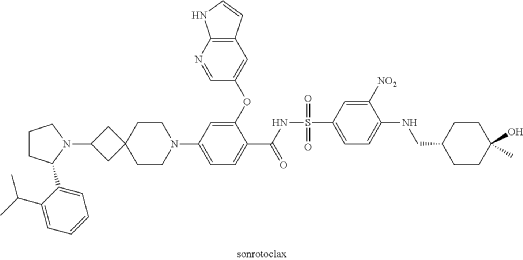
2-((1H-pyrrolo[2,3-b]pyridin-5-yl)oxy)-N-((4-((((1r,4r)-4-hydroxy-4-methylcyclohexyl)methyl)amino)-3-nitrophenyl)sulfonyl)-4-(2-((S)-2-(2-isopropylphenyl)pyrrolidin-1-yl)-7-azaspiro[3.5]nonan-7-yl)benzamide (hereinafter sonrotoclax).
SYN
2-((1H-pyrrolo[2,3-b]pyridin-5-yl)oxy)-N-((4-((((1r,4r)-4-hydroxy-4-methylcyclohexyl)methyl)amino)-3-nitrophenyl)sulfonyl)-4-(2-((S)-2-(2-isopropylphenyl)pyrrolidin-1-yl)-7-azaspiro[3.5]nonan-7-yl)benzamide
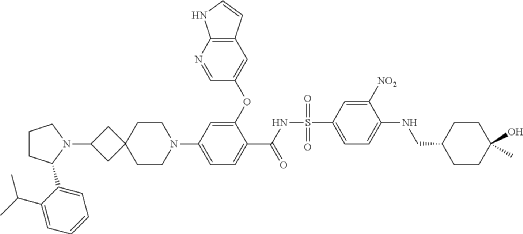
Step 9: 2-((1H-pyrrolo[2,3-b]pyridin-5-yl)oxy)-N-((4-((((1r,4r)-4-hydroxy-4-methylcyclohexyl)methyl)amino)-3-nitrophenyl)sulfonyl)-4-(2-((S)-2-(2-isopropylphenyl)pyrrolidin-1-yl)-7-azaspiro[3.5]nonan-7-yl)benzamide
SYN
- US11420968
- https://patentscope.wipo.int/search/en/detail.jsf?docId=US335022833&_cid=P10-MP4VD4-31735-1
Example F43: 2-((1H-pyrrolo[2,3-b]pyridin-5-yl)oxy)-N-((4-((((1r,4r)-4-hydroxy-4-methylcyclohexyl)methyl)amino)-3-nitrophenyl)sulfonyl)-4-(2-((S)-2-(2-isopropylphenyl)pyrrolidin-1-yl)-7-azaspiro[3.5]nonan-7-yl)benzamide
PAT
- Ketal Protected Intermediate for Sonrotoclax and Preparation Method ThereofPublication Number: US-2025320207-A1Priority Date: 2022-12-27
- Methods of treating multiple myeloma using bcl-2 inhibitorPublication Number: US-2025161279-A1Priority Date: 2022-07-21
- Methods of treating myeloid malignancies using bcl-2 inhibitorPublication Number: WO-2023218410-A1Priority Date: 2022-05-12
- Methods of treating myeloid malignancies using bcl-2 inhibitorPublication Number: US-2025057821-A1Priority Date: 2022-05-12
- Methods of treating myeloid malignancies using bcl-2 inhibitorPublication Number: EP-4522169-A1Priority Date: 2022-05-12
- Methods of cancer treatment using bcl-2 inhibitorPublication Number: WO-2021110102-A1Priority Date: 2019-12-02
- Bcl-2 inhibitorsPublication Number: US-2022402915-A1Priority Date: 2018-04-29
- Bcl-2 inhibitorsPublication Number: US-11420968-B2Priority Date: 2018-04-29Grant Date: 2022-08-23
- Bcl-2 inhibitorsPublication Number: US-2021269433-A1Priority Date: 2018-04-29
- Bcl-2 InhibitorsPublication Number: US-2024376104-A1Priority Date: 2018-04-29
- Bcl-2 inhibitorsPublication Number: EP-3788042-B1Priority Date: 2018-04-29Grant Date: 2025-02-12
- Bcl-2 inhibitorsPublication Number: EP-4545515-A1Priority Date: 2018-04-29
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
References
- “Sonrotoclax – BeiGene”. AdisInsight. Springer Nature Switzerland AG.
- Tomkins O, D’Sa S (2024). “Review of BCL2 inhibitors for the treatment of Waldenström’s macroglobulinaemia and non-IgM lymphoplasmacytic lymphoma”. Frontiers in Oncology. 14 1490202. doi:10.3389/fonc.2024.1490202. PMC 11570586. PMID 39558954.
| Clinical data | |
|---|---|
| Pronunciation | /sɒnˈroʊtəklæks/ son-ROH-tə-klaks |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2383086-06-2 |
| PubChem CID | 149553242 |
| ChemSpider | 129309008 |
| UNII | 30R67U9KYS |
| KEGG | D12883 |
| ChEMBL | ChEMBL5314951 |
| Chemical and physical data | |
| Formula | C49H59N7O7S |
| Molar mass | 890.11 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
/////////sonrotoclax, anax labs, FDA 2026, APPROVALS 2026, Beqalzi, BGB-11417, BGB 11417, 30R67U9KYS, accelerated approval

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
