

Gallium 68 PSMA-11
FDA APPROVED, 12/1/2020, Gallium 68 PSMA-11
For detection and localization of prostate cancer
Press Release
Drug Trials Snapshot

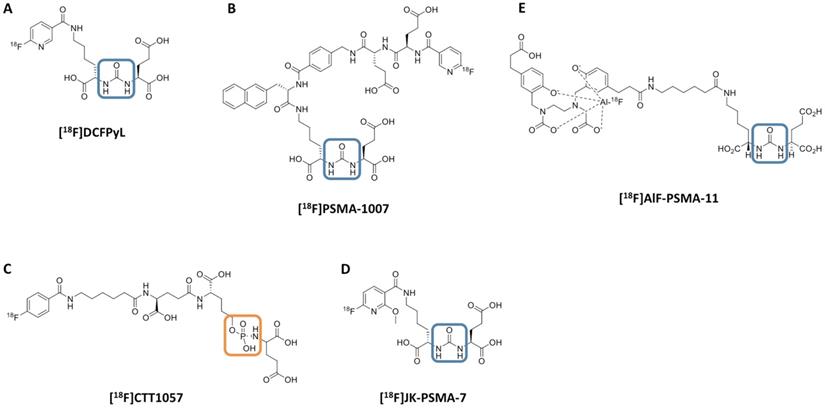
Chemical structure of 18F-labeled radiotracers. [18F]DCFPyL (A), [18F]PSMA-1007 (B), [18F]CTT1057 (C), (D) [18F]JK-PSMA-7 and (E) [18F]AIF-PSMA-11. The urea backbone of (A), (B), (D) and (E) is marked in blue, while the phosphoramidate of [18F]CTT1057 in (C) is highlighted in orange. Modified from Behr et al. [32], © by the Society of Nuclear Medicine and Molecular Imaging, Inc.
PSMA-11, also known as HBED-CC-PSMA or Psma-hbed-CC, is used to make gallium Ga 68-labeled PSMA-11, which has potential use as a tracer for PSMA-expressing tumors during positron emission tomography (PET). Upon intravenous administration of gallium Ga 68-labeled PSMA-11, the Glu-urea-Lys(Ahx) moiety targets and binds to PSMA-expressing tumor cells. Upon internalization, PSMA-expressing tumor cells can be detected during PET imaging. PSMA, a tumor-associated antigen and type II transmembrane protein, is expressed on the membrane of prostatic epithelial cells and overexpressed on prostate tumor cells

Name: PSMA-11
CAS#: 1366302-52-4
Chemical Formula: C44H62N6O17
Exact Mass: 946.4171
(3S,7S)-22-(3-(((2-((5-(2-Carboxyethyl)-2-hydroxybenzyl)(carboxymethyl)amino)ethyl)(carboxymethyl)amino)methyl)-4-hydroxyphenyl)-5,13,20-trioxo-4,6,12,19-tetraazadocosane-1,3,7-tricarboxylic acid
The Food and Drug Administration (FDA) has approved Gallium 68 PSMA-11 (Ga 68 PSMA-11), the first drug for positron emission tomography (PET) imaging of prostate-specific membrane antigen (PSMA) positive lesions in men with prostate cancer.
Ga 68 PSMA-11, a radioactive diagnostic agent, is indicated for patients with suspected prostate cancer metastasis who are potentially curable by surgery or radiation therapy. It is also indicated for patients with suspected prostate cancer recurrence based on elevated serum prostate-specific antigen (PSA) levels.
The approval was based on efficacy and safety data from 2 prospective clinical trials (Trial 1 and 2) with a total of 960 men with prostate cancer who each received 1 injection of Ga 68 PSMA-11. Trial 1 included 325 patients with biopsy-proven prostate cancer who underwent PET/CT or PET/MRI scans performed with Ga 68 PSMA-11. Results from the study showed that positive readings in the pelvic lymph nodes on Ga 68 PSMA-11 PET were associated with a clinically important rate of metastatic cancer confirmed by surgical pathology in those who proceeded to surgery.
In Trial 2, 635 patients with rising serum PSA levels after prostate surgery or radiotherapy received a single Ga 68 PSMA-11 PET/CT scan or PET/MR scan. Findings demonstrated that 74% of patients had at least 1 positive lesion detected by Ga 68 PSMA-11 PET, and local recurrence or metastasis of prostate cancer was confirmed in 91% of cases.
 This is the first drug approved for PET imaging of prostate-specific membrane antigen positive lesions in men with prostate cancer.
This is the first drug approved for PET imaging of prostate-specific membrane antigen positive lesions in men with prostate cancer.
REF
REFERENCES
1: Meißner S, Janssen JC, Prasad V, Brenner W, Diederichs G, Hamm B, Hofheinz F, Makowski MR. Potential of asphericity as a novel diagnostic parameter in the evaluation of patients with (68)Ga-PSMA-HBED-CC PET-positive prostate cancer lesions. EJNMMI Res. 2017 Oct 23;7(1):85. doi: 10.1186/s13550-017-0333-9. PubMed PMID: 29058157; PubMed Central PMCID: PMC5651532.
2: Verburg FA, Pfister D, Drude NI, Mottaghy FM, Behrendt F. PSA levels, PSA doubling time, Gleason score and prior therapy cannot predict measured uptake of [(68)Ga]PSMA-HBED-CC lesion uptake in recurrent/metastatic prostate cancer. Nuklearmedizin. 2017 Oct 18;56(6). doi: 10.3413/Nukmed-0917-17-07. [Epub ahead of print] PubMed PMID: 29044297.
3: Amor-Coarasa A, Kelly JM, Gruca M, Nikolopoulou A, Vallabhajosula S, Babich JW. Continuation of comprehensive quality control of the itG (68)Ge/(68)Ga generator and production of (68)Ga-DOTATOC and (68)Ga-PSMA-HBED-CC for clinical research studies. Nucl Med Biol. 2017 Oct;53:37-39. doi: 10.1016/j.nucmedbio.2017.07.006. Epub 2017 Jul 14. PubMed PMID: 28803001.
4: Janssen JC, Woythal N, Meißner S, Prasad V, Brenner W, Diederichs G, Hamm B, Makowski MR. [(68)Ga]PSMA-HBED-CC Uptake in Osteolytic, Osteoblastic, and Bone Marrow Metastases of Prostate Cancer Patients. Mol Imaging Biol. 2017 Dec;19(6):933-943. doi: 10.1007/s11307-017-1101-y. PubMed PMID: 28707038.
5: Damle NA, Tripathi M, Chakraborty PS, Sahoo MK, Bal C, Aggarwal S, Arora G, Kumar P, Kumar R, Gupta R. Unusual Uptake of Prostate Specific Tracer (68)Ga-PSMA-HBED-CC in a Benign Thyroid Nodule. Nucl Med Mol Imaging. 2016 Dec;50(4):344-347. Epub 2016 Mar 22. PubMed PMID: 27994690; PubMed Central PMCID: PMC5135692.
6: Behrendt F, Krohn T, Mottaghy F, Verburg FA. [(68)Ga]PSMA-HBED-CC PET/CT to differentiate between diffuse bone metastases of prostate cancer and osteopoikilosis. Nuklearmedizin. 2016 Dec 6;55(6):N64-N65. PubMed PMID: 27922151.
7: Krohn T, Birmes A, Winz OH, Drude NI, Mottaghy FM, Behrendt FF, Verburg FA. The reconstruction algorithm used for [(68)Ga]PSMA-HBED-CC PET/CT reconstruction significantly influences the number of detected lymph node metastases and coeliac ganglia. Eur J Nucl Med Mol Imaging. 2017 Apr;44(4):662-669. doi: 10.1007/s00259-016-3571-6. Epub 2016 Nov 29. PubMed PMID: 27900518.
8: Berliner C, Tienken M, Frenzel T, Kobayashi Y, Helberg A, Kirchner U, Klutmann S, Beyersdorff D, Budäus L, Wester HJ, Mester J, Bannas P. Detection rate of PET/CT in patients with biochemical relapse of prostate cancer using [(68)Ga]PSMA I&T and comparison with published data of [(68)Ga]PSMA HBED-CC. Eur J Nucl Med Mol Imaging. 2017 Apr;44(4):670-677. doi: 10.1007/s00259-016-3572-5. Epub 2016 Nov 28. PubMed PMID: 27896369.
9: Sathekge M, Lengana T, Modiselle M, Vorster M, Zeevaart J, Maes A, Ebenhan T, Van de Wiele C. (68)Ga-PSMA-HBED-CC PET imaging in breast carcinoma patients. Eur J Nucl Med Mol Imaging. 2017 Apr;44(4):689-694. doi: 10.1007/s00259-016-3563-6. Epub 2016 Nov 8. PubMed PMID: 27822700; PubMed Central PMCID: PMC5323468.
10: Rauscher I, Maurer T, Beer AJ, Graner FP, Haller B, Weirich G, Doherty A, Gschwend JE, Schwaiger M, Eiber M. Value of 68Ga-PSMA HBED-CC PET for the Assessment of Lymph Node Metastases in Prostate Cancer Patients with Biochemical Recurrence: Comparison with Histopathology After Salvage Lymphadenectomy. J Nucl Med. 2016 Nov;57(11):1713-1719. Epub 2016 Jun 3. PubMed PMID: 27261524.
11: Verburg FA, Behrendt FF, Mottaghy FM, Pfister D, Steib F, Knuechel R. Strong [(68)Ga]PSMA-HBED-CC accumulation in non-cancerous prostate tissue surrounding a PSMA-negative prostate carcinoma recurrence. Nuklearmedizin. 2016 Sep 26;55(5):N44-5. PubMed PMID: 27668299.
12: Kanthan GL, Izard MA, Emmett L, Hsiao E, Schembri GP. Schwannoma Showing Avid Uptake on 68Ga-PSMA-HBED-CC PET/CT. Clin Nucl Med. 2016 Sep;41(9):703-4. doi: 10.1097/RLU.0000000000001281. PubMed PMID: 27405039.
13: Noto B, Vrachimis A, Schäfers M, Stegger L, Rahbar K. Subacute Stroke Mimicking Cerebral Metastasis in 68Ga-PSMA-HBED-CC PET/CT. Clin Nucl Med. 2016 Oct;41(10):e449-51. doi: 10.1097/RLU.0000000000001291. PubMed PMID: 27355852.
14: Pfob CH, Ziegler S, Graner FP, Köhner M, Schachoff S, Blechert B, Wester HJ, Scheidhauer K, Schwaiger M, Maurer T, Eiber M. Biodistribution and radiation dosimetry of (68)Ga-PSMA HBED CC-a PSMA specific probe for PET imaging of prostate cancer. Eur J Nucl Med Mol Imaging. 2016 Oct;43(11):1962-70. doi: 10.1007/s00259-016-3424-3. Epub 2016 May 20. PubMed PMID: 27207281.
15: Amor-Coarasa A, Schoendorf M, Meckel M, Vallabhajosula S, Babich JW. Comprehensive Quality Control of the ITG 68Ge/68Ga Generator and Synthesis of 68Ga-DOTATOC and 68Ga-PSMA-HBED-CC for Clinical Imaging. J Nucl Med. 2016 Sep;57(9):1402-5. doi: 10.2967/jnumed.115.171249. Epub 2016 Apr 21. PubMed PMID: 27103024.
16: Prasad V, Steffen IG, Diederichs G, Makowski MR, Wust P, Brenner W. Biodistribution of [(68)Ga]PSMA-HBED-CC in Patients with Prostate Cancer: Characterization of Uptake in Normal Organs and Tumour Lesions. Mol Imaging Biol. 2016 Jun;18(3):428-36. doi: 10.1007/s11307-016-0945-x. PubMed PMID: 27038316.
17: Pfister D, Porres D, Heidenreich A, Heidegger I, Knuechel R, Steib F, Behrendt FF, Verburg FA. Detection of recurrent prostate cancer lesions before salvage lymphadenectomy is more accurate with (68)Ga-PSMA-HBED-CC than with (18)F-Fluoroethylcholine PET/CT. Eur J Nucl Med Mol Imaging. 2016 Jul;43(8):1410-7. doi: 10.1007/s00259-016-3366-9. Epub 2016 Mar 19. PubMed PMID: 26993315.
18: Kanthan GL, Coyle L, Kneebone A, Schembri GP, Hsiao E. Follicular Lymphoma Showing Avid Uptake on 68Ga PSMA-HBED-CC PET/CT. Clin Nucl Med. 2016 Jun;41(6):500-1. doi: 10.1097/RLU.0000000000001169. PubMed PMID: 26914565.
19: Kanthan GL, Hsiao E, Kneebone A, Eade T, Schembri GP. Desmoid Tumor Showing Intense Uptake on 68Ga PSMA-HBED-CC PET/CT. Clin Nucl Med. 2016 Jun;41(6):508-9. doi: 10.1097/RLU.0000000000001192. PubMed PMID: 26909712.
20: Eiber M, Weirich G, Holzapfel K, Souvatzoglou M, Haller B, Rauscher I, Beer AJ, Wester HJ, Gschwend J, Schwaiger M, Maurer T. Simultaneous (68)Ga-PSMA HBED-CC PET/MRI Improves the Localization of Primary Prostate Cancer. Eur Urol. 2016 Nov;70(5):829-836. doi: 10.1016/j.eururo.2015.12.053. Epub 2016 Jan 18. PubMed PMID: 26795686.
//////////Gallium 68 PSMA-11, FDA 2020, 2020 APPROVALS, RADIO ACTIVE

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
