Funapide TV 45070, XEN-402, Funapide, (+)-
- Molecular FormulaC22H14F3NO5
- Average mass429.345 Da
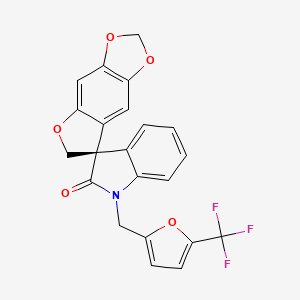
(S)-1′-[(5-Methyl-2-furyl)methyl]spiro[6H-furo[3,2-f][1,3]benzodioxole-7,3′-indoline]-2′-one
Spiro(furo(2,3-F)-1,3-benzodioxole-7(6H),3′-(3H)indol)-2′(1’H)-one, 1′-((5-(trifluoromethyl)-2-furanyl)methyl)-, (3’S)-
(3’S)-1′-((5-(Trifluoromethyl)furan-2-yl)methyl)-2H,6H-spiro(furo(2,3-F)(1,3)benzodioxole-7,3′-indol)-2′(1’H)-one
Phase II clinical trials for Postherpetic neuralgia (PHN)
Treatment of Neuropathic Pain
- Originator Xenon Pharmaceuticals
- Developer Teva Pharmaceutical Industries; Xenon Pharmaceuticals
- Class Benzodioxoles; Fluorobenzenes; Furans; Indoles; Non-opioid analgesics; Small molecules; Spiro compounds
- Mechanism of Action Nav1.7-voltage-gated-sodium-channel-inhibitors; Nav1.8 voltage-gated sodium channel inhibitors
- Orphan Drug Status Yes – Erythromelalgia
Highest Development Phases
- Phase II Erythromelalgia; Postherpetic neuralgia
- No development reported Dental pain; Pain
- Discontinued Musculoskeletal pain
Most Recent Events
- 09 May 2017 Teva Pharmaceutical Industries completes a phase IIb trial for Postherpetic neuralgia in USA (Topical) (NCT02365636)
- 26 Sep 2016 Adverse events data from a phase II trial in Musculoskeletal pain presented at the 16th World Congress on Pain (PAN – 2016)
- 19 Aug 2015 No recent reports of development identified – Phase-I for Pain (In volunteers) in Canada (PO)
MP 100 – 102 DEG CENT EP2538919
S ROT ALPHA 0.99 g/100ml, dimethyl sulfoxide, 14.04, US 20110087027
Funapide (INN) (former developmental code names TV-45070 and XEN402) is a novel analgesic under development by Xenon Pharmaceuticals in partnership with Teva Pharmaceutical Industries for the treatment of a variety of chronic pain conditions, including osteoarthritis, neuropathic pain, postherpetic neuralgia, and erythromelalgia, as well as dental pain.[1][2][3][4] It acts as a small-moleculeNav1.7 and Nav1.8 voltage-gated sodium channel blocker.[1][2][4] Funapide is being evaluated in humans in both oral and topicalformulations, and as of July 2014, has reached phase IIb clinical trials.[1][3]

Sodium channels play a diverse set of roles in maintaining normal and pathological states, including the long recognized role that voltage gated sodium channels play in the generation of abnormal neuronal activity and neuropathic or pathological pain. Damage to peripheral nerves following trauma or disease can result in changes to sodium channel activity and the development of abnormal afferent activity including ectopic discharges from axotomised afferents and spontaneous activity of sensitized intact nociceptors. These changes can produce long-lasting abnormal hypersensitivity to normally innocuous stimuli, or allodynia. Examples of neuropathic pain include, but are not limited to, post-herpetic neuralgia, trigeminal neuralgia, diabetic neuropathy, chronic lower back pain, phantom limb pain, and pain resulting from cancer and chemotherapy, chronic pelvic pain, complex regional pain syndrome and related neuralgias.
There have been some advances in treating neuropathic pain symptoms by using medications, such as gabapentin, and more recently pregabalin, as short-term, first-line treatments. However, pharmacotherapy for neuropathic pain has generally had limited success with little response to commonly used pain reducing drugs, such as NSAIDS and opiates. Consequently, there is still a considerable need to explore novel treatment modalities.
There remain a limited number of potent effective sodium channel blockers with a minimum of adverse events in the clinic. There is also an unmet medical need to treat neuropathic pain and other sodium channel associated pathological states effectively and without adverse side effects. PCT Published Patent Application No. WO 2006/110917, PCT Published Patent Application No. WO 2010/045251 , PCT Published Patent Application No. WO 2010/045197, PCT Published Patent Application No. WO 2011/047174 and PCT Published Patent Application No. WO 2011/002708 discloses certain spiro-oxindole compounds. These compounds are disclosed therein as being useful for the treatment of sodium channel-mediated diseases, preferably diseases related to pain, central nervous conditions such as epilepsy, anxiety, depression and bipolar disease;
cardiovascular conditions such as arrhythmias, atrial fibrillation and ventricular fibrillation; neuromuscular conditions such as restless leg syndrome; neuroprotection against stroke, neural trauma and multiple sclerosis; and channelopathies such as erythromelalgia and familial rectal pain syndrome.
Methods of preparing these compounds and pharmaceutical compositions containing them are also disclosed in PCT Published Patent Application No. WO 2006/110917, PCT Published Patent Application No. WO 2010/045251 , PCT
Published Patent Application No. WO 2010/045197, PCT Published Patent Application No. WO 2011/047174 and PCT Published Patent Application No. WO 2011/002708.

| Inventors | Mikhail Chafeev, Jianmin Fu, Jean-Jacques Cadieux |
| Original Assignee | Xenon Pharmaceuticals Inc. |



Column: Chiralcel® OJ-RH; 20 mm I.D.×250 mm, 5 mic; Lot: OJRH CJ-EH001 (Daicel Chemical Industries, Ltd)
Eluent: Acetonitrile/Water (60/40, v/v, isocratic)
Flow rate: 10 mL/min
Run time: 60 min
Loading: 100 mg of compound of formula (I) in 1 mL of acetonitrileTemperature: Ambient
Under the above chiral HPLC conditions, the (R)-enantiomer of the compound of formula (I), i.e., (R)-1′-{[5-(trifluoromethyl)furan-2-yl]methyl}spiro[furo[2,3-f][1,3]-benzodioxole-7,3′-indol]-2′(1′H)-one, was isolated as the first fraction as a white solid; ee (enantiomeric excess)>99% (analytical OJ-RH, 55% acetonitrile in water); mp 103-105° C.; 1H NMR (300 MHz, DMSO-d6) δ 7.32-6.99 (m, 5H), 6.71 (d, J=3.4 Hz, 1H), 6.67 (s, 1H), 6.05 (s, 1H), 5.89 (d, J=6.2 Hz, 2H), 5.13, 5.02 (ABq, JAB=16.4 Hz, 2H), 4.82, 4.72 (ABq, JAB=9.4 Hz, 2H); 13C NMR (75 MHz, CDCl3) δ 177.2, 155.9, 152.0, 149.0, 142.4, 142.0, 141.3, 132.0, 129.1, 123.9, 120.6, 119.2, 117.0, 112.6, 109.3, 108.9, 103.0, 101.6, 93.5, 80.3, 58.2, 36.9; MS (ES+) m/z 430.2 (M+1), [α]D−17.46° (c 0.99, DMSO).
The (S)-enantiomer of the compound of formula (I), i.e., (S)-1′-{[5-(trifluoromethypfuran-2-yl]methyl}spiro-[furo[2,3-f][1,3]benzodioxole-7,3′-indol]-2′(1′H)-one was isolated as the second fraction as a white solid; ee >99% (analytical OJ-RH, 55% acetonitrile in water); mp 100-102° C.; 1H NMR (300 MHz, DMSO-d6) δ 7.32-6.99 (m, 5H), 6.71 (d, J=3.4 Hz, 1H), 6.67 (s, 1H), 6.05 (s, 1H), 5.89 (d, J=6.3 Hz, 2H), 5.12, 5.02 (ABq, JAB=16.4 Hz, 2H), 4.82, 4.72 (ABq, JAB=9.4 Hz, 2H); 13C NMR (75MHz, CDCl3) δ 177.2, 155.9, 152.0, 149.0, 142.4, 142.0, 141.3, 132.0, 129.1, 123.9, 120.6, 119.2, 117.0, 112.6, 109.3, 108.9, 103.0, 101.6, 93.5, 80.3, 58.2, 36.9; MS (ES+) m/z 430.2 (M+1), [α]D+14.04° (c 0.99, DMSO)
Synthetic Example 3Resolution of Compound of Formula (I) by SMB Chromatography
The compound of formula (I) was resolved into the (S)-enantiomer of the invention and the corresponding (R)-enantiomer by SMB chromatography under the following conditions:
Extract: 147.05 mL/min, Raffinate: 76.13 mL/min Eluent: 183.18 mL/min Feed: 40 mL/min Recycling: 407.88 mL/min Run Time: 0.57 min Temperature: 25° C. Pressure: 46 bar
The feed solution (25 g of compound of formula (I) in 1.0 L of mobile phase (25:75:0.1 (v:v:v) mixture of acetonitrile/methanol/trifluoroacetic acid)) was injected continuously into the SMB system (Novasep Licosep Lab Unit), which was equipped with eight identical columns in 2-2-2-2 configuration containing 110 g (per column, 9.6 cm, 4.8 cm I.D.) of ChiralPAK-AD as stationary phase. The first eluting enantiomer (the (R)-enantiomer of the compound of formula (I)) was contained in the raffinate stream and the second eluting enantiomer (the (S)-enantiomer of the compound of formula (I)) was contained in the extract stream. The characterization data of the (S)-enantiomer and the (R)-enantiomer obtained from the SMB resolution were identical to those obtained above utilizing chiral HPLC.
The compound of formula (I) was resolved into its constituent enantiomers on a Waters preparative LCMS autopurification system. The first-eluting enantiomer from the chiral column was brominated (at a site well-removed from the stereogenic centre) to give the corresponding 5′-bromo derivative, which was subsequently crystallized to generate a single crystal suitable for X-ray crystallography. The crystal structure of this brominated derivative of the first-eluting enantiomer was obtained and its absolute configuration was found to be the same as the (R)-enantiomer of the invention. Hence, the second-eluting enantiomer from the chiral column is the (S)-enantiomer of the invention. Moreover, the material obtained from the extract stream of the SMB resolution had a specific optical rotation of the same sign (positive, i.e. dextrorotatory) as that of the material obtained from the aforementioned LC resolution.
Patent
EXAMPLE 8
Synthesis of (7S)-1 ‘-{[5-(trifluoromethyl)furan-2- yllmethylJspirotfurop.S-flll .Sl enzoclioxole-y.S’-indoll-Zil ‘Wi-one
Compound of formula (ia1 )
To a cooled (0 °C) solution of (3S)-3-(6-hydroxy-1 ,3-benzodioxol-5-yl)-3- (hydroxymethyl)-1-{[5-(trifluoromethyl)furan-2-yl]methyl}-1 ,3-dihydro-2H-indol-2-one prepared according to the procedure described in Example 7 (16.4 mmol) and 2- (diphenylphosphino)pyridine (5.2 g, 20 mmol) in anhydrous tetrahydrofuran (170 mL) was added di-ferf-butylazodicarboxylate (4.5 g, 20 mmol). The mixture was stirred for 2 h at 0 °C, then the reaction was diluted with ethyl acetate (170 mL), washed with 3 N hydrochloric acid (7 x 50 mL) and brine (2 x 100 mL), dried over anhydrous sodium sulfate, filtered and concentrated in vacuo. The residue was dissolved in ethanol (80 mL), decolorizing charcoal (15 g) was added and the mixture was heated at reflux for 1 h. The mixture was filtered while hot through a pad of diatomaceous earth. The filtrate was concentrated in vacuo and the residue triturated in a mixture of diethyl ether/hexanes to afford (7S)-1 ‘-{[5-(trifluoromethyl)furan-2-yl]methyl}spiro- [furo[2,3-/][1 ,3]benzodioxole-7,3’-indol]-2′(1 ‘H)-one (1.30 g) as a colorless solid in 18% yield. The mother liquor from the trituration was concentrated in vacuo, trifluoroacetic acid (20 mL) was added and the mixture stirred for 3 h at ambient temperature. The mixture was diluted with ethyl acetate (100 mL), washed with saturated aqueous ammonium chloride (100 mL), 3 N hydrochloric acid (4 x 60 mL) and brine (2 x 100 mL), dried over anhydrous sodium sulfate, filtered and concentrated in vacuo. The residue was purified by column chromatography, eluting with a gradient of ethyl acetate in hexanes to afford further (7S)-1 ‘-{[5-(trifluoromethyl)furan-2-yl]methyl}spiro- [furo[2,3- ][1 ,3]benzodioxole-7,3’-indol]-2′(1 ‘H)-one (2.6 g) as a colorless solid (37% yield, overall yield 55% over 2 steps): H NMR (300 MHz, CDCI3) £7.29-6.96 (m, 4H), 6.73 (s, 1 H), 6.50 (s, 1 H), 6.38 (s, 1 H), 6.09 (s, 1 H), 5.85 (br s, 2H), 5.06 (d, J = 16.0 Hz, 1 H), 4.93-4.84 (m, 2H), 4.68-4.65 (m, 1 H); MS (ES+) m/z 429.8 (M + 1 ); ee (enantiomeric excess) >99.5% (HPLC, Chiralpak IA, 2.5% acetonitrile in methyl tert- butyl ether).
EXAMPLE 9
Synthesis of 1-(diphenylmethyl)-1 H-indole-2,3-dione
Compound of formula (15a)
A. To a suspension of hexanes-washed sodium hydride (34.0 g, 849 mmol) in anhydrous Λ/,/V-dimethylformamide (400 mL) at 0 °C was added a solution of isatin (99.8 g, 678 mmol) in anhydrous Λ/,/V-dimethylformamide (400 mL) dropwise over 30 minutes. The reaction mixture was stirred for 1 h at 0 °C and a solution of benzhydryl bromide (185 g, 745 mmol) in anhydrous N-dimethylformamide (100 mL) was added dropwise over 5 minutes. The reaction mixture was allowed to warm to ambient temperature, stirred for 16 h and heated at 60 °C for 2 h. The mixture was cooled to 0 °C and water (500 mL) was added. The mixture was poured into water (2 L), causing a precipitate to be deposited. The solid was collected by suction filtration and washed with water (2000 mL) to afford 1-(diphenylmethyl)-1H-indole-2,3- dione (164 g) as an orange solid in 77% yield.
B. Alternatively, to a mixture of isatin (40.0 g, 272 mmol), cesium carbonate (177 g, 543 mmol) and A/./V-dimethylformamide (270 mL) at 80 °C was added dropwise a solution of benzhydryl bromide (149 g, 544 mmol) in N,N- dimethyiformamide (200 mL) over 30 minutes. The reaction mixture was heated at 80 °C for 3 h, allowed to cool to ambient temperature and filtered through a pad of diatomaceous earth. The pad was rinsed with ethyl acetate (1000 mL). The filtrate was washed with saturated aqueous ammonium chloride (4 x 200 mL), 1 N
hydrochloric acid (200 mL) and brine (4 x 200 mL), dried over anhydrous sodium sulfate, filtered and concentrated in vacuo. The residue was triturated with diethyl ether to afford 1 -(diphenylmethyl)-1 H-indole-2,3-dione (59.1 g) as an orange solid in 69% yield. The mother liquor from the trituration was concentrated in vacuo and the residue triturated in diethyl ether to afford a further portion of 1-(diphenylmethyl)-1 H- indole-2,3-dione (8.2 g) in 10% yield: 1H NMR (300 MHz, CDCI3) £7.60 (d, J = 7.4 Hz, 1 H), 7.34-7.24 (m, 1 1 H), 7.05-6.97 (m, 2H), 6.48 (d, J = 8.0 Hz, 1 H); MS (ES+) m/z 313.9 (M + 1 ).
C. Alternatively, a mixture of isatin (500 g, 3.4 mol) and anhydrous N,N- dimethylformamide (3.5 L) was stirred at 15-35 °C for 0.5 h. Cesium carbonate (2.2 kg, 6.8 mol) was added and the mixture stirred at 55-60 °C for 1 h. A solution of benzhydryl bromide (1.26 kg, 5.1 mol) in anhydrous N, A/-dimethylformamide (1.5 L) was added and the resultant mixture stirred at 80-85 °C for 1 h, allowed to cool to ambient temperature and filtered. The filter cake was washed with ethyl acetate (12.5 L). To the combined filtrate and washes was added 1 N hydrochloric acid (5 L). The phases were separated and the aqueous phase was extracted with ethyl acetate (2.5 L). The combined organic extracts were washed with 1 N hydrochloric acid (2 * 2.5 L) and brine (3 χ 2.5 L) and concentrated in vacuo to a volume of approximately 750 mL. Methyl ferf-butyl ether (2 L) was added and the mixture was cooled to 5-15 °C, causing a solid to be deposited. The solid was collected by filtration, washed with methyl ferf- butyl ether (250 mL) and dried in vacuo at 50-55 °C for 16 h to afford 1- (diphenylmethyl)-1 H-indole-2,3-dione (715 g) as an orange solid in 67% yield: 1H NMR (300 MHz, CDCI3) 7.60 (d, J = 7.4 Hz, H), 7.34-7.24 (m, 1 H), 7.05-6.97 (m, 2H), 6.48 (d, J = 8.0 Hz, 1 H); MS (ES+) m/z 313.9 (M + 1 ).
EXAMPLE 10
Synthesis of 1-(diphenylmethyl)-3-hydroxy-3-(6-hydroxy-1 ,3-benzodioxol-5-yl)-1 ,3- dihydro-2H-indol-2-one
Compound of formula (16a1 )
A. To a solution of sesamol (33.1 g, 239 mmol) in anhydrous
tetrahydrofuran (500 mL) at 0 °C was added dropwise a 2 M solution of
isopropylmagnesium chloride in tetrahydrofuran (104 mL, 208 mmol), followed by 1 – (diphenylmethyl)-1H-indole-2,3-dione (50.0 g, 160 mmol) and tetrahydrofuran (100 mL). The reaction mixture was stirred at ambient temperature for 5 h, diluted with ethyl acetate (1500 mL), washed with saturated aqueous ammonium chloride (400 mL) and brine (2 x 400 mL), dried over anhydrous sodium sulfate, filtered and concentrated in vacuo. The residue was triturated with a mixture of diethyl ether and hexanes to afford 1- (diphenylmethyl)-3-hydroxy-3-(6-hydroxy-1 ,3-benzodioxol-5-yl)-1 ,3-dihydro-2H-in
2- one (70.7 g) as a colorless solid in 98% yield: 1H NMR (300 MHz, CDCI3) <59.12 (br s, 1 H), 7.45-7.43 (m, 1 H), 7.30-7.22 (m, 10H), 7.09-7.07 (m, 2H), 6.89 (s, 1 H), 6.56- 6.55 (m, 1 H), 6.47-6.46 (m, 1 H), 6.29-6.28 (m, 1 H), 5.86 (s, 2H), 4.52 (br s, 1 H); MS (ES+) m/z 433.7 (M – 17).
B. Alternatviely, a mixture of sesamol (0.99 kg, 7.2 mol) and anhydrous tetrahydrofuran (18 L) was stirred at 15-35 °C for 0.5 h and cooled to -5-0 °C.
Isopropyl magnesium chloride (2.0 M solution in tetrahydrofuran, 3.1 L, 6.2 mol) was added, followed by 1-(diphenylmethyl)-1 H-indole-2,3-dione (1.50 kg, 4.8 mol) and further anhydrous tetrahydrofuran (3 L). The mixture was stirred at 15-25 °C for 5 h. Ethyl acetate (45 L) and saturated aqueous ammonium chloride (15 L) were added. The mixture was stirred at 15-25 °C for 0.5 h and was allowed to settle for 0.5 h. The phases were separated and the organic phase was washed with brine (2.3 L) and concentrated in vacuo to a volume of approximately 4 L. Methyl ferf-butyl ether (9 L) was added and the mixture concentrated in vacuo to a volume of approximately 4 L. Heptane (6 L) was added and the mixture was stirred at 15-25 °C for 2 h, causing a solid to be deposited. The solid was collected by filtration, washed with methyl tert- butyl ether (0.3 L) and dried in vacuo at 50-55 °C for 7 h to afford 1-(diphenylmethyl)-3- hydroxy-3-(6-hydroxy-1 ,3-benzodioxol-5-yl)-1 ,3-dihydro-2H-indol-2-one (2.12 kg) as an off-white solid in 98% yield: 1H NMR (300 MHz, CDCI3) 9.12 (br s, 1 H), 7.45-7.43 (m, 1 H), 7.30-7.22 (m, 10H), 7.09-7.07 (m, 2H), 6.89 (s, 1 H), 6.56-6.55 (m, 1 H), 6.47-6.46 (m, 1 H), 6.29-6.28 (m, 1 H), 5.86 (s, 2H), 4.52 (br s, 1 H); MS (ES+) m/z 433.7 (M – 17).
EXAMPLE 1 1
Synthesis of 3-[6-(benzyloxy)-1 ,3-benzodioxol-5-yl]-1-(diphenylmethyl)-3-hydroxy-1 ,3- dihydro-2H-indol-2-one
Compound of formula (17a1)
A. A mixture of 1-(diphenylmethyl)-3-hydroxy-3-(6-hydroxy-1 ,3- benzodioxol-5-yl)-1 ,3-dihydro-2H-indol-2-one (30.0 g, 66.5 mmol), benzyl bromide (8.3 mL, 70 mmol), and potassium carbonate (18.4 g, 133 mmol) in anhydrous N,N- dimeihylformamide (100 mL) was stirred at ambient temperature for 16 h. The reaction mixture was filtered and the solid was washed with /V,A/-dimethylformamide (100 mL). The filtrate was poured into water (1000 mL) and the resulting precipitate was collected by suction filtration and washed with water to afford 3-[6-(benzyloxy)-1 ,3-benzodioxol- 5-yl]-1-(diphenylmethyl)-3-hydroxy-1 ,3-dihydro-2H-indol-2-one (32.0 g) as a beige solid in 83% yield: 1H NMR (300 MHz, CDCI3) 7.42-7.28 (m, 9H), 7.22-7.14 (m, 6H), 7.10- 6.93 (m, 3H), 6.89-6.87 (m, 2H), 6.53 (d, J = 7.6 Hz, 1 H), 6.29 (br s, 1 H), 5.88 (s, 1 H), 5.85 (s, 1 H), 4.66 (d, J = 14.2 Hz, 1 H), 4.51 (d, J = 14.1 Hz, 1 H), 3.95 (s, 1 H); MS (ES+) m/z 542.0 (M + 1), 523.9 (M – 17).
B. Alternatively, to a solution of 1-(diphenylmethyl)-3-hydroxy-3-(6- hydroxy-1 ,3-benzodioxol-5-yl)-1 ,3-dihydro-2H-indol-2-one (2.1 kg, 4.6 mol) in anhydrous A/,A/-dimethylformamide (8.4 L) at 20-30 °C was added potassium carbonate (1.3 kg, 9.2 mol), followed by benzyl bromide (0.58 L, 4.8 mol). The mixture was stirred at 20-30 °C for 80 h and filtered. The filter cake was washed with
A/,/V-dimethylformamide (0.4 L) and the filtrate was poured into water (75 L), causing a solid to be deposited. The mixture was stirred at 15-25 °C for 7 h. The solid was collected by filtration, washed with water (2 L) and dried in vacuo at 50-60 °C for 48 h to afford 3-[6-(benzyloxy)-1 ,3-benzodioxol-5-yl]-1-(diphenylmethyl)-3-hydroxy-1 ,3- dihydro-2H-indol-2-one (2.1 1 kg) as an off-white solid in 84% yield; 1H NMR (300
MHz, CDCI3) £7.42-7.28 (m, 9H), 7.22-7.14 (m, 6H), 7.10-6.93 (m, 3H), 6.89-6.87 (m, 2H), 6.53 (d, J = 7.6 Hz, 1 H), 6.29 (br s, 1 H), 5.88 (s, 1 H), 5.85 (s, 1 H), 4.66 (d, J = 14.2 Hz, 1 H), 4.51 (d, J = 14.1 Hz, 1 H), 3.95 (s, 1 H); MS (ES+) m/z 542.0 (M + 1 ).
EXAMPLE 12
Synthesis of 3-[6-(benzyloxy)-1 ,3-benzodioxol-5-yl]-1 -(diphenylmethyl)-l ,3-dihydro-2H- indol-2-one
Compound of formula (18a1 )
A. To a solution of 3-[6-(benzyloxy)-1 ,3-benzodioxol-5-yl]-1- (diphenylmethyl)-3-hydroxy-1 ,3-dihydro-2H-indol-2-one (32.0 g, 57.7 mmol) in dichloromethane (100 mL) was added trifluoroacetic acid (50 mL) followed by triethylsilane (50 mL). The reaction mixture was stirred at ambient temperature for 2 h and concentrated in vacuo. The residue was dissolved in ethyi acetate (250 mL), washed with saturated aqueous ammonium chloride (3 x 100 mL) and brine (3 x 100 mL), dried over anhydrous sodium sulfate, filtered and concentrated in vacuo. The residue was triturated with diethyl ether to afford 3-[6-(benzyloxy)-1 ,3-benzodioxol-5- yl]-1-(diphenylmethyl)-1 ,3-dihydro-2H-indol-2-one (19.0 g) as a colorless solid in 61 % yield: 1H NMR (300 MHz, CDCI3) 7.31 -7.23 (m, 15H), 7.10-6.88 (m, 4H), 6.50-6.45 (m, 3H), 5.86 (s, 2H), 4.97-4.86 (m, 3H); MS (ES+) m/z 525.9 (M + 1).
B. Alternatively, to a solution of 3-[6-(benzyloxy)-1 ,3-benzodioxol-5-yl]-1- (diphenylmethyl)-3-hydroxy-1 ,3-dihydro-2H-indol-2-one (2.0 kg, 3.7 mol) in
dichloromethane (7 L) at 20-30 °C was added trifluoracetic acid (2.5 L), followed by triethylsilane (3.1 L). The mixture was stirred at 15-35 °C for 4 h and concentrated in vacuo to dryness. To the residue was added ethyl acetate (16 L) and the mixture was stirred at 15-35 °C for 0.5 h, washed with saturated aqueous ammonium chloride (3 x 7 L) and brine (3 χ 7 L) and concentrated in vacuo to a volume of approximately 7 L. Methyl ferf-butyl ether (9 L) was added and the mixture concentrated in vacuo to a volume of approximately 9 L and stirred at 10-20 °C for 2.5 h, during which time a solid was deposited. The solid was collected by filtration, washed with methyl te/t-butyl ether (0.4 L) and dried in vacuo at 50-55 °C for 7 h to afford 3-[6-(benzyloxy)-1 ,3- benzodioxol-5-yl]-1-(diphenylmethyl)-1 ,3-dihydro-2H-indol-2-one (1 .26 kg) as an off-white solid in 65% yield: 1H NMR (300 MHz, CDCI3) £7.31 -7.23 (m, 15H), 7.10- 6.88 (m, 4H), 6.50-6.45 (m, 3H), 5.86 (s, 2H), 4.97-4.86 (m, 3H); MS (ES+) m/z 525.9 (M + 1).
EXAMPLE 13
Synthesis of (3S)-3-[6-(benzyloxy)-1 ,3-benzodioxol-5-yl]-3-[(benzyloxy)methyl]-1 –
(diphenylmethyl)-1 ,3-dihydro-2H-indol-2-one
Compound of formula (19a1 )
A. To a nitrogen-degassed mixture of 50% w/w aqueous potassium hydroxide (69.6 mL, 619 mmol), toluene (100 mL), and (9S)-1 -(anthracen-9- ylmethyl)cinchonan-1 -ium-9-ol chloride (0.50 g, 0.95 mmol) cooled in an ice/salt bath to an internal temperature of -18 °C was added a nitrogen-degassed solution of 3-[6- (benzyloxy)-l ,3-benzodioxol-5-yl]-1 -(diphenylmethyl)-l ,3-dihydro-2H-indol-2-one (10.0 g, 19.0 mmol) and benzyl chloromethyl ether (2.9 mL, 21 mmol) in
toluene/tetrahydrofuran (1 :1 v/v, 80 mL) dropwise over 1 h. The reaction mixture was stirred for 3.5 h and diluted with ethyl acetate (80 mL). The organic phase was washed with 1 N hydrochloric acid (3 x 150 mL) and brine (2 x 100 mL), dried over anhydrous sodium sulfate, filtered and concentrated in vacuo to afford (3S)-3-[6-(benzyloxy)-1 ,3- benzodioxol-5-yl]-3-[(benzyloxy)methyl]-1-(diphenylmethyl)-1 ,3-dihydro-2/-/-indol-2-one (12.6 g) as a colorless solid in quantitative yield: 1H NMR (300 MHz, CDCI3) 7.42 (d, 2H), 7.24-6.91 (m, 21 H), 6.69-6.67 (m, 2H), 6.46 (d, J = 7.7 Hz, 1 H), 6.15 (s, 1 H), 5.83- 5.81 (m, 2H), 4.53-4.31 (m, 3H), 4.17-4.09 (m, 3H); MS (ES+) m/z 646.0 (M + 1); ee (enantiomeric excess) 90% (HPLC, Chiralpak IA, 2.5% acetonitrile in methyl tert-butyl ether).
B. Alternatively, a mixture of 50% w/v aqueous potassium hydroxide (4.2 kg), toluene (12 L) and (9S)-1 -(anthracen-9-ylmethyl)cinchonan-1 -ium-9-ol chloride (0.06 kg, 0.1 mol) was degassed with dry nitrogen and cooled to -18 to -22 °C. To this mixture was added a cold (-18 to -22 °C), nitrogen-degassed solution of 3-[6-
(benzyloxy)-l ,3-benzodioxol-5-yl]-1 ~(diphenylmethyl)-1 ,3-dihydro-2H-indol-2-one (1.2 kg, 2.3 mol) and benzyl chloromethyl ether (0.43 kg, 2.8 mol) in toluene (10 L) and tetrahydrofuran (10 L) at -18 to 22 °C over 3 h. The mixture was stirred at -18 to -22 °C for 5 h, allowed to warm to ambient temperature and diluted with ethyl acetate (10 L). The phases were separated and the organic layer was washed with 1 N
hydrochloric acid (3 χ 8 L) and brine (2 χ 12 L) and concentrated in vacuo to dryness to afford (3S)-3-[6-(benzyloxy)-1 ,3-benzodioxol-5-yl]-3-[(benzyloxy)methyl]-1- (diphenylmethyl)-1 ,3-dihydro-2H-indol-2-one (1.5 kg) as a colorless solid in quantitative yield: 1H NMR (300 MHz, CDCI3) £7.42 (d, 2H), 7.24-6.91 (m, 21 H), 6.69-6.67 (m, 2H), 6.46 (d, J = 7.7 Hz, 1 H), 6.15 (s, 1 H), 5.83-5.81 (m, 2H), 4.53-4.31 (m, 3H), 4.17- 4.09 (m, 3H); MS (ES+) m/z 646.0 (M + 1); ee (enantiomeric excess) 90% (HPLC, ChiralPak IA). EXAMPLE 14
Synthesis of (3S)-1-(diphenylmethyl)-3-(6-hydroxy-1 ,3-benzodioxol-5-yl)-3- (hydroxymethyl)-1 ,3-dihydro-2/-/-indol-2-one
Compound of formula (20a1)
A. A mixture of (3S)-3-[6-(benzyloxy)-1 ,3-benzodioxol-5-yl]-3- [(benzyloxy)methyl]-1 -(diphenylmethyl)-1 ,3-dihydro-2/-/-indol-2-one (8.8 g, 14 mmol), 10% w/w palladium on carbon (50% wetted powder, 3.5 g, 1.6 mmol), and acetic acid (3.9 ml_, 68 mmol) in a nitrogen-degassed mixture of ethanol/tetrahydrofuran (1 : 1 v/v, 140 mL) was stirred under hydrogen gas (1 atm) at ambient temperature for 4 h. The reaction mixture was filtered through a pad of diatomaceous earth and the pad was rinsed with ethyl acetate (100 mL). The filtrate was concentrated in vacuo to afford (3S)-1-(diphenylmethyl)-3-(6-hydroxy-1 ,3-benzodioxol-5-yl)-3-(hydroxymethyl)-1 ,3- dihydro-2H-indol-2-one as a colorless solid that was carried forward without further purification: H NMR (300 MHz, CDCI3) 9.81 (br s, 1 H), 7.35-7.24 (m, 1 1 H), 7.15- 7.01 (m, 3H), 6.62 (s, 1 H), 6.54-6.47 (m, 2H), 5.86-5.84 (m, 2H), 4.76 (d, J = 1 1.0 Hz, 1 H), 4.13-4.04 (m, 1 H), 2.02 (s, 1 H); MS (ES+) m/z 465.9 (M + 1); ee (enantiomeric excess) 93% (HPLC, Chiralpak IA, 2.5% acetonitrile in methyl ie t-butyl ether).
B. Alternatively, a glass-lined hydrogenation reactor was charged with (3S)-3-[6-(benzyloxy)-1 ,3-benzodioxol-5-yl]-3-[(benzyloxy)methyl]-1 -(diphenylmethyl)- 1 ,3-dihydro-2H-indol-2-one (0.1 kg, 0.15 mol), tetrahydrofuran (0.8 L), ethanol (0.4 L), acetic acid (0.02 L) and 20% w/w palladium (li) hydroxide on carbon (0.04 kg). The reactor was purged three times with nitrogen. The reactor was then purged three times with hydrogen and was then pressurized to 50-55 lb/in2 with hydrogen. The mixture was stirred at 20-30 °C for 5 h under a 50-55 lb/in2 atmosphere of hydrogen. The reactor was purged and the mixture was filtered. The filtrate was concentrated in vacuo to a volume of approximately 0.2 L and methyl te/t-butyl ether (0.4 L) was added. The mixture was concentrated in vacuo to a volume of approximately 0.2 L and methyl ie/t-butyl ether (0.2 L) was added, followed by heptane (0.25 L). The mixture was stirred at ambient temperature for 2 h, during which time a solid was deposited. The solid was collected by filtration, washed with heptane (0.05 L) and dried in vacuo at a temperature below 50 °C for 8 h to afford (3S)-1 -(diphenylmethyl)-3-(6-hydroxy- 1 ,3-benzodioxol-5-yl)-3-(hydroxymethyl)-1 ,3-dihydro-2H-indol-2-one (0.09 kg) as a colorless solid in 95% yield: 1H NMR (300 MHz, CDCI3) 9.81 (br s, 1 H), 7.35-7.24 (m, 1 1 H), 7.15-7.01 (m, 3H), 6.62 (s, 1 H), 6.54-6.47 (m, 2H), 5.86-5.84 (m, 2H), 4.76 (d, J = 1 1.0 Hz, 1 H), 4.13-4.04 (m, 1 H), 2.02 (s, 1 H); MS (ES+) m/z 465.9 (M + 1); ee (enantiomeric excess) 91% (HPLC, ChiralPak IA).
EXAMPLE 15
Synthesis of (7S)-1′-(diphenylmethyl)spiro[furo[2,3-/][1 ,3]benzodioxole-7,3′-indol]-
2′(1 ‘tf)-one
Compound of formula (21 a1 )
A. To a cooled (0 °C) solution of (3S)-1 -(diphenylmethyl)-3-(6-hydroxy-1 ,3- benzodioxol-5-yl)-3-(hydroxymethyl)-1 ,3-dihydro-2H-indol-2-one prepared according to the procedure described in Example 14 (13.6 mmol) and 2-
(diphenylphosphino)pyridine (4.3 g, 16 mmol) in anhydrous tetrahydrofuran (140 mL) was added di-tert-butylazodicarboxylate (3.8 g, 17 mmol). The reaction mixture was stirred at 0 °C for 3 h, diluted with ethyl acetate (140 mL), washed with 3 N
hydrochloric acid (6 * 50 mL) and brine (2 χ 100 mL), dried over anhydrous sodium sulfate, filtered and concentrated in vacuo. The residue was triturated with a mixture of diethyl ether and hexanes to afford (7S)-1 ‘-(diphenylmethyl)spiro[furo[2,3- ][1 ,3]benzodioxole-7,3’-indol]-2′(1 ‘H)-one (4.55 g) as a colorless solid in a 75% yield over 2 steps: 1H NMR (300 MHz, CDCI3) 7.34-7.24 (m, 10H), 7.15-7.13 (m, 1 H), 7.04 (s, 1 H), 6.99-6.95 (m, 2H), 6.50-6.48 (m, 2H), 6.06 (s, 1 H), 5.85-5.83 (m, 2H), 4.96 (d, J = 8.9 Hz, 1 H), 4.69 (d, J = 8.9 Hz, 1 H); MS (ES+) m/z 447.9 (M + 1); ee
(enantiomeric excess) 93% (HPLC, Chiraipak IA, 2.5% acetonitrile in methyl te/f-butyl ether).
B. Alternativel, to a cooled (0-5 °C) solution of (3S)-1-(diphenylmethyl)-3- (6-hydroxy-1 ,3-benzodioxol-5-yl)-3-(hydroxymethyl)-1 ,3-dihydro-2 -/-indol-2-one (1 .0 kg, 2.1 mol) and 2-(diphenylphosphino)pyridine (0.66 kg, 2.5 mol) in anhydrous tetrahydrofuran (20 L) was added over 2 h a solution of di-terf-butylazodicarboxylate (0.62 kg, 2.7 mmol) in anhydrous tetrahydrofuran (5 L). The mixture was stirred for 4 h at 0-5 °C and was allowed to warm to ambient temperature. The mixture was diluted with ethyl acetate (20 L), washed with 3 N hydrochloric acid (6 * 8 L) and brine (2 x 12 L) and concentrated in vacuo to a volume of approximately 1.5 L. Methyl rert-butyl ether (4 L) was added and the mixture concentrated in vacuo to a volume of
approximately 1.5 L. Methyl terf-butyl ether (2 L) and heptane (2 L) were added and the mixture was stirred at ambient temperature for 2 h, during which time a solid was deposited. The solid was collected by filtration, washed with heptane (0.5 L) and dried in vacuo below 50 °C for 8 h to afford (7S)-1′-(diphenylmethyl)spiro[furo[2,3- f][1 ,3]benzodioxole-7,3′-indol]-2′(1’H)-one (0.76 kg) as a colorless solid in 79% yield: 1H NMR (300 MHz, CDCI3) 7.34-7.24 (m, 10H), 7.15-7.13 (m, 1 H), 7.04 (s, 1 H), 6.99- 6.95 (m, 2H), 6.50-6.48 (m, 2H), 6.06 (s, 1 H), 5.85-5.83 (m, 2H), 4.96 (d, J = 8.9 Hz, 1 H), 4.69 (d, J = 8.9 Hz, 1 H); MS (ES+) m/z 447.9 (M + 1 ); ee (enantiomeric excess) 92% (HPLC, ChiralPak IA).
EXAMPLE 16
Synthesis of (7S)-spiro[furo[2,3-f][1 ,3]benzodioxole-7,3′-indol]-2′(1 ‘H)-one
Compound of formula (22a1)
A. To a solution of (7S)-1′-(diphenylmethyl)spiro[furo[2,3- f][1 ,3]benzodioxole-7,3′-indol]-2′(1’H)-one (4.55 g, 10.2 mmol) in trifluoroacetic acid (80 ml_) was added triethylsilane (7 ml_). The reaction mixture was heated at reflux for 2.5 h, allowed to cool to ambient temperature and concentrated in vacuo. The residue was triturated with a mixture of diethyl ether and hexanes to afford
(7S)-spiro[furo[2,3-/][1 ,3]benzodioxole-7,3,-indol]-2′(1’W)-one (2.30 g) as a colorless solid in 80% yield: 1H NMR (300 MHz, CDCI3) £8.27 (br s, 1 H), 7.31-7.26 (m, 1 H), 7.17-7.15 (m, 1 H), 7.07-7.02 (m, 1 H), 6.96-6.94 (m, 1 H), 6.53-6.52 (m, 1 H), 6.24-6.23 (m, 1 H), 5.88-5.87 (m, 2H), 4.95 (d, J = 8.6 Hz, 1 H), 4.68 (d, J = 8.9 Hz, 1 H); MS (ES+) m/z 281.9 (M + 1 ); ee (enantiomeric excess) 99% (HPLC, Chiralpak IA, 2.5% acetonitrile in methyl fert-butyl ether). B. Alternatively, a mixture of (7S)-1 ‘-(diphenylmethyl)spiro[furo[2,3- /Kl^benzodioxole^-indol^ r^-one (0.70 kg, 1.6 mol), trifluoroacetic acid (12 L) and triethylsilane (1.1 L) was heated at reflux under nitrogen atmosphere for 3 h, allowed to cool to ambient temperature and concentrated in vacuo to dryness. To the residue was added ethyl acetate (0.3 L), methyl fert-butyl ether (1 L) and heptane (3.5 L), causing a solid to be deposited. The solid was collected by filtration, taken up in dichloromethane (3 L), stirred at ambient temperature for 1 h and filtered. The filtrate was concentrated in vacuo to dryness. The residue was taken up in ethyl acetate (0.3 L), methyl ferf-butyl ether (1 L) and heptane (3.5 L), causing a solid to be deposited. The solid was collected by filtration and dried in vacuo below 50 °C for 8 h to afford (7S)-spiro[furo[2,3- ][1 ,3]benzodioxole-7,3’-indol]-2′(1 ‘ -/)-one (0.40 kg) as a colorless solid in 91 % yield: 1H NMR (300 MHz, CDCI3) 8.27 (br s, 1 H), 7.31-7.26 (m, 1 H), 7.17-7.15 (m, 1 H), 7.07-7.02 (m, 1 H), 6.96-6.94 (m, 1 H), 6.53-6.52 (m, 1 H), 6.24-6.23 (m, 1 H), 5.88-5.87 (m, 2H), 4.95 (d, J = 8.6 Hz, 1 H), 4.68 (d, J = 8.9 Hz, 1 H); MS (ES+) m/z 281.9 (M + 1); ee (enantiomeric excess) 98.6% (HPLC, ChiralPak IA).
EXAMPLE 17
Synthesis of of (7S)-1 ‘-{[5-(trifluoromethyl)furan-2- yl]methyl}spiro[furo[2,3- ][1 ,3]benzodioxole-7,3’-indol]-2′(rH)-one
Compound of formula (Ia1)
A. To a mixture of (7S)-6H-spiro[[1 ,3]dioxolo[4,5-f]benzofuran-7,3′-indolin]- 2′-one (1.80 g, 6.41 mmol) and 2-(bromomethyl)-5-(trifluoromethyl)furan (1.47 g, 6.41 mmol) in acetone (200 mL) was added cesium carbonate (3.13 g, 9.61 mmol). The reaction mixture was heated at reflux for 2 h and filtered while hot through a pad of diatomaceous earth. The filtrate was concentrated in vacuo to afford (7S)-1′-{[5- (trifluoromethyOfuran^-yllmethy^spiroIfurop.S- ltl .Slbenzodioxole^.S’-indol^ rH)- one (2.71 g) as a colorless solid in quantitative yield (97% purity by HPLC). The product was crystallized from a mixture of methanol and hexanes to afford (7S)-1 ‘-{[5- (trifluoromethy furan^-yllmethylJspirotfuro^.S- lfl .Slbenzodioxole^.S’-indoll^ rH)- one (1.46 g) as colorless needles in 53% yield. The mother liquor was concentrated in vacuo and subjected to a second crystallization in methanol and hexanes to afford further (7S)-1 ‘-{[5-(trifluoromethyl)furan-2-yl]methyl}spiro[furo[2,3-/][1 ,3]benzodioxole- 7,3’-indol]-2′(1 ‘H)-one (0.469 g) as a colorless solid in 17% yield (total yield 70%): 1H NMR (300 MHz, CDCI3) δ 7.29-6.96 (m, 4H), 6.73 (s, 1 H), 6.50 (s, 1 H), 6.38 (s, 1 H), 6.09 (s, 1 H), 5.85 (br s, 2H), 5.06 (d, J = 16.0 Hz, 1 H), 4.93-4.84 (m, 2H), 4.68-4.65 (m, 1 H); MS (ES+) m/z 429.8 (M + 1); ee (enantiomeric excess) >99.5% (HPLC, Chiralpak IA, 2.5% acetonitrile in methyl tert-butyl ether).
B. Alternatively, to a solution of (7S)-spiro[furoI2,3-f][1 ,3]benzodioxole-7,3′- indol]-2′(1’H)-one (0.40 kg, 1.4 mol) in anhydrous N, W-dimethylformamide (5 L) was added cesium carbonate (1.2 kg, 3.4 mol), followed by 2-(bromomethyl)-5- (trifluromethyl)furan (0.24 L, 1.7 mol). The mixture was heated at 80-85 °C for 3 h, allowed to cool to ambient temperature and filtered through a pad of diatomaceous earth. The pad was washed with ethyl acetate (8 L). The combined filtrate and washes were washed with water (4 L), saturated aqueous ammonium chloride (2 * 4 L) and brine (2 * 4 L) and concentrated in vacuo to dryness. The residue was purified by recrystallization from te/t-butyl methyl ether (0.4 L) and heptane (0.8 L), followed by drying of the resultant solid in vacuo at 40-50 °C for 8 h to afford (7S)-1 ‘-{[5- (trifluoromethyl)furan-2-yl]methyl}spiro[furo[2,3-f][1 ,3]benzodioxole-7,3’-indol]-2′(1 ‘H)- one (0.37 kg) as a colorless solid in 61% yield: 1H NMR (300 MHz, CDCI3) δ 7.29-6.96 (m, 4H), 6.73 (s, 1 H), 6.50 (s, 1 H), 6.38 (s, 1 H), 6.09 (s, 1 H), 5.85 (br s, 2H), 5.06 (d, J = 16.0 Hz,1 H), 4.93-4.84 (m, 2H), 4.68-4.65 (m, 1 H); MS (ES+) m/z 429.8 (M + 1 ); ee (enantiomeric excess) > 99% (HPLC, Chiralpak IA).
TV-45070 is a small-molecule lactam containing a chiral spiro-ether that has been reported as a potential topical therapy for pain associated with the Nav1.7 sodium ion channel encoded by the gene SCN9A. A pilot-scale synthesis is presented that is highlighted by an asymmetric aldol coupling at ambient temperature, used to create a quaternary chiral center. Although only a moderate ee is obtained, the removal of the undesired isomer is achieved through preferential precipitation of a near racemic mixture from the reaction, leaving the enantiopure isomer in solution. Cyclization to form the final API uses an uncommon diphenylphosphine-based leaving group which proved successful on the neopentyl system when other traditional leaving groups failed.
The First Asymmetric Pilot-Scale Synthesis of TV-45070
*E-mail: jasclafan@yahoo.com.
(S)-1′-[(5-Methyl-2-furyl)methyl]spiro[6H-furo[3,2-f][1,3]benzodioxole-7,3′-indoline]-2′-one (1)



References
- ^ Jump up to:a b c Bagal, Sharan K.; Chapman, Mark L.; Marron, Brian E.; Prime, Rebecca; Ian Storer, R.; Swain, Nigel A. (2014). “Recent progress in sodium channel modulators for pain”. Bioorganic & Medicinal Chemistry Letters. 24 (16): 3690–9. ISSN 0960-894X. PMID 25060923. doi:10.1016/j.bmcl.2014.06.038.
- ^ Jump up to:a b Stephen McMahon; Martin Koltzenburg; Irene Tracey; Dennis C. Turk (1 March 2013). Wall & Melzack’s Textbook of Pain: Expert Consult – Online. Elsevier Health Sciences. p. 508. ISBN 0-7020-5374-0.
- ^ Jump up to:a b Xenon Pharma. “TV-45070: A Small Molecule for the Treatment of the Orphan Disease EM and Other Pain Disorders”.
- ^ Jump up to:a b Xenon Pharma (2012). “Teva and Xenon Announce Teva’s World Wide License of Xenon’s Pain Drug XEN402”.
External links
| Cited Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| WO2006110917A2 | 11 Apr 2006 | 19 Oct 2006 | Xenon Pharmaceuticals Inc. | Spiro-oxindole compounds and their uses as therapeutic agents |
| WO2010045197A1 | 13 Oct 2009 | 22 Apr 2010 | Xenon Pharmaceuticals, Inc. | Spiro-oxindole compounds and their use as therapeutic agents |
| WO2010045251A2 | 13 Oct 2009 | 22 Apr 2010 | Xenon Pharmaceuticals, Inc. | Spiro-oxindole compounds and their use as therapeutic agents |
| WO2010104525A1 | 1 Jun 2009 | 16 Sep 2010 | Telcordia Technologies, Inc. | Scalable disruptive-resistant communication method |
| WO2011002708A1 | 28 Jun 2010 | 6 Jan 2011 | Xenon Pharmaceuticals Inc. | Enantiomers of spiro-oxindole compounds and their uses as therapeutic agents |
| WO2011047173A2 | 14 Oct 2010 | 21 Apr 2011 | Xenon Pharmaceuticals Inc. | Pharmaceutical compositions for oral administration |
| WO2011047174A1 | 14 Oct 2010 | 21 Apr 2011 | Xenon Pharmaceuticals Inc. | Synthetic methods for spiro-oxindole compounds |
| WO2011106729A2 | 25 Feb 2011 | 1 Sep 2011 | Xenon Pharmaceuticals Inc. | Pharmaceutical compositions of spiro-oxindole compound for topical administration and their use as therapeutic agents |
| Reference | ||
|---|---|---|
| 1 | * | DEHMLOW E V ET AL: “Monodeazacinchona alkaloid derivatives: synthesis and preliminary applications as phase-transfer catalysts“, EUROPEAN JOURNAL OF ORGANIC CHEMISTRY, WILEY – V C H VERLAG GMBH & CO. KGAA, DE, vol. 13, 1 January 2002 (2002-01-01), pages 2087 – 2093, XP002399953, ISSN: 1434-193X, DOI: 10.1002/1099-0690(200207)2002:13<2087::AID-EJOC2087>3.0.CO;2-Z |
| 2 | E.J. COREY; M.C. NOE, ORG. SYNTH., vol. 80, 2003, pages 38 – 45 | |
| 3 | GARST, J. F.; UNGVARY, F.: “Grignard Reagents”, 2000, JOHN WILEY & SONS, article “Mechanism of Grignard reagent formation“, pages: 185 – 275 | |
| 4 | GREENE, T.W.; P.G.M. WUTS: “Greene’s Protective Groups in Organic Synthesis, 4th Ed.,“, 2006, WILEY | |
| 5 | GREENE, T.W.; WUTS, P.G.M.: “Greene’s Protective Groups in Organic Synthesis, 4th Ed.“, 2006, WILEY | |
| 6 | HUGHES, D.L., ORG. PREP., vol. 28, 1996, pages 127 – 164 | |
| 7 | KUMARA SWAMY, K.C. ET AL.: “Mitsunobu and Related Reactions: Advances and Applications“, CHEM. REV., vol. 109, 2009, pages 2551 – 2651, XP055023394, DOI: doi:10.1021/cr800278z | |
| 8 | MERSMANN, A.: “Crystallization Technology Handbook; 2nd ed.“, 2001, CRC | |
| 9 | SMITH, M.; BAND J. MARCH: “Advanced Organic Chemistry: Reactions, Mechanisms, and Structure, 5th edition“, December 2000, WILEY | |
| 10 | SMITH, M.B.; J. MARCH: “Advanced Organic Chemistry: Reactions, Mechanisms, and Structure, 5th edition“, December 2000, WILEY | |
| 11 | * | TAKASHI OOI ET AL: “Recent Advances in Asymmetric Phase-Transfer Catalysis“, ANGEWANDTE CHEMIE INTERNATIONAL EDITION, vol. 46, no. 23, 4 June 2007 (2007-06-04), pages 4222 – 4266, XP055060024, ISSN: 1433-7851, DOI: 10.1002/anie.200601737 |
| Citing Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| WO2016109795A1 | 31 Dec 2015 | 7 Jul 2016 | Concert Pharmaceuticals, Inc. | Deuterated funapide and difluorofunapide |
| US8742109 | 14 Sep 2012 | 3 Jun 2014 | Xenon Pharmaceuticals Inc. | Synthetic methods for spiro-oxindole compounds |
| US8883840 | 14 Sep 2012 | 11 Nov 2014 | Xenon Pharmaceuticals Inc. | Enantiomers of spiro-oxindole compounds and their uses as therapeutic agents |
| US8916580 | 6 Mar 2013 | 23 Dec 2014 | Xenon Pharmaceuticals Inc. | Spiro-oxindole compounds and their use as therapeutic agents |
| US9260446 | 7 May 2014 | 16 Feb 2016 | Xenon Pharmaceuticals Inc. | Synthetic methods for spiro-oxindole compounds |
| US9458178 | 14 Nov 2014 | 4 Oct 2016 | Xenon Pharmaceuticals Inc. | Spiro-oxindole compounds and their use as therapeutic agents |
| US9480677 | 9 Oct 2014 | 1 Nov 2016 | Xenon Pharmaceuticals Inc. | Enantiomers of spiro-oxindole compounds and their uses as therapeutic agents |
| US9487535 | 11 Mar 2013 | 8 Nov 2016 | Xenon Pharmaceuticals Inc. | Asymmetric syntheses for spiro-oxindole compounds useful as therapeutic agents |
| US9504671 | 25 Feb 2011 | 29 Nov 2016 | Xenon Pharmaceuticals Inc. | Pharmaceutical compositions of spiro-oxindole compound for topical administration and their use as therapeutic agents |
| US9682033 | 5 Feb 2016 | 20 Jun 2017 | Teva Pharmaceuticals International Gmbh | Methods of treating postherpetic neuralgia with a topical formulation of a spiro-oxindole compound |
| US9695185 | 6 Jan 2016 | 4 Jul 2017 | Xenon Pharmaceuticals Inc. | Synthetic methods for spiro-oxindole compounds |
 |
|
| Clinical data | |
|---|---|
| Routes of administration |
By mouth, topical |
| ATC code |
|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C22H14F3NO5 |
| Molar mass | 429.34547 g/mol |
| 3D model (JSmol) | |

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....