CDSCO
 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO, Novel Drug Approvals for 2017, A Review Compilation (USFDA, EMA, PMDA, CDSCO).
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO, Novel Drug Approvals for 2017, A Review Compilation (USFDA, EMA, PMDA, CDSCO).
Any errors in this compilation, email amcrasto@gmail.com, Call +919323115463
Some gaps will be filled up soon keep watching……………..
INDEX, NAME (click on the title, it contains link)
SECTION A; USFDA Approvals
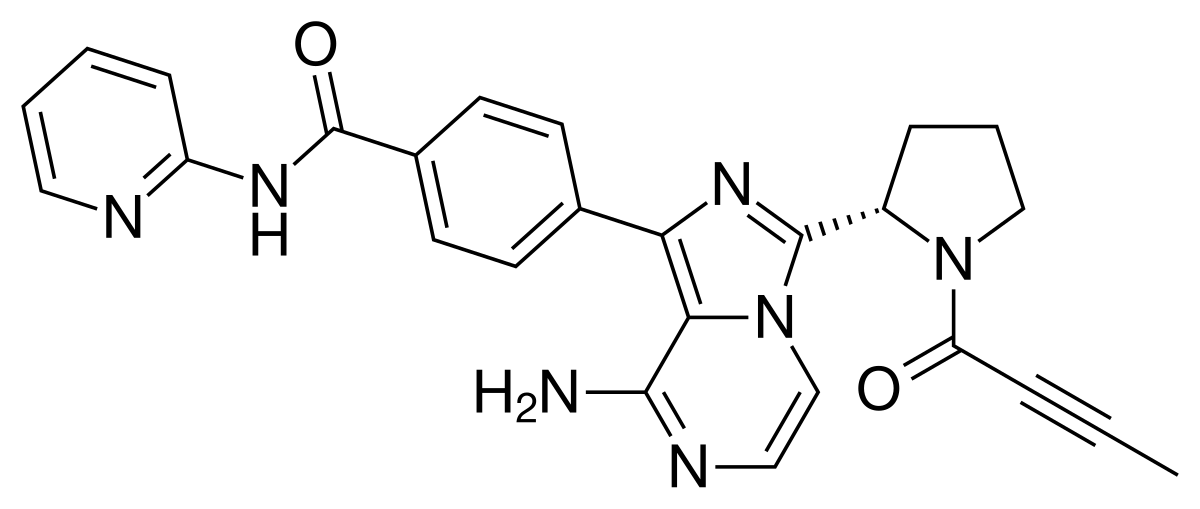
1 Abaloparatide
2 Abemaciclib
3 ACALABRUTINIB
4 ANGIOTENSIN II
5 AVELUMAB
6 BENRALIZUMAB
7 BENZNIDAZOLE
8 BETRIXABAN
9 BRIGATINIB
10 BRODALUMAB
11 CERLIPONASE ALPA
12 COPANLISIB
13 DEFLAZACORT
14 Delafloxacin
15 Deutetrabenazine
16DUPILUMAB
17 DURVALUMAB
18 EDAVARONE
19 EMICIZUMAB
20 Enasidenib
21 ERTUGLIFLOZIN
22 ETELCALCETIDE
23 GLECAPREVIR
24 GUSELKUMAB
25 INOTUZUMAB OZOGAMICIN
26 LATANOPROSTENE
27 LETERMOVIR
28 MACIMORELIN ACETATE
29 MEROPENEM
30 MIDOSTAURIN
31 NALDEMEDINE
32 NERATINIB
33 NETARSUDIL
34 NIRAPARIB
35 Ocrelizumab
36 OZENOXACIN
37 PIBRENTASVIR
38 PLECANATIDE
39 RIBOCICLIB
40 SARILUMAB
41 SECNIDAZOLE
42 SAFINAMIDE
43 SEMAGLUTIDE
44 SOFOSBUVIR
45 TELOTRISTAT ETHYL
46 VABORBACTAM
47 VALBENAZINE
48 VESTRONIDASE ALFA-VJBK
49 VELPATASVIR
50 VOXILAPREVIR
INDEX, FORMULATION NAME
USFDA
•Aliqopa (COPANLISIB) to treat adults with relapsed follicular lymphoma — a slow-growing type of nonHodgkin lymphoma (a cancer of the lymph system) — who have received at least two prior systemic therapies;
• ALUNBRIG, BRIGATINIB, To treat patients with anaplastic lymphoma kinase (ALK)-positive metastatic non-small cell lung cancer (NSCLC) who have progressed on or are intolerant to crizotinib
• Austedo, Deutetrabenazine For the treatment of chorea associated with Huntington’s disease
• Bavencio (avelumab) for the treatment of patients 12 years and older with a rare and aggressive form of cancer called metastatic Merkel cell carcinoma, including those who have not received prior chemotherapy;
•BAXDELLA, Delafloxacin, BACTERIAL INFECTIONS
• Benznidazole to treat children ages 2 to 12 years with Chagas disease, a parasitic infection that can cause serious heart illness after years of infection, and can also affect swallowing and digestion. This is the first treatment approved in the United States for this rare disease;
• Besponsa (inotuzumab ozogamicin) for the treatment of adults with a type of cancer of the blood called relapsed or refractory B-cell precursor acute lymphoblastic leukemia;
•BEVYXXA, BETRIXABAN, For the prophylaxis of venous thromboembolism (VTE) in adult patients hospitalized for an acute medical illness
• BRINEURA, CERLIPONASE ALFA, To treat a specific form of Batten disease
• Calquence (ACALABRUTINIB) to treat adults with mantle cell lymphoma who have received at least one prior therapy. Mantle cell lymphoma is a particularly aggressive cancer;
• DUPIXENT, (DUPILUMAB) To treat adults with moderate-to-severe eczema (atopic dermatitis)
• Emflaza (deflazacort) to treat patients age 5 years and older with Duchenne muscular dystrophy, a rare genetic disorder that causes progressive muscle deterioration and weakness;
• FASENRA, BENRALIZUMAB, For add-on maintenance treatment of patients with severe asthma aged 12 years and older, and with an eosinophilic phenotype
• Giapreza (angiotensin II), for the treatment of hypotension in adults with distributive or vasodilatory shock (dangerously low blood pressure despite adequate heart function) whose blood pressure remains low despite receiving fluids and treatment with drugs called vasopressors;
• HEMLIBRA EMICIZUMAB To prevent or reduce the frequency of bleeding episodes in adult and pediatric patients with hemophilia A who have developed antibodies called Factor VIII (FVIII) inhibitors.
• Idhifa (enasidenib) for the treatment of adults with relapsed or refractory acute myeloid leukemia, a form of blood cancer, who have a specific genetic mutation;
• IMFINZI, DURVALUMAB To treat patients with locally advanced or metastatic urothelial carcinoma
• Ingrezza (valbenazine) to treat adults with tardive dyskinesia, a side effect of some antipsychotic medications whereby patients can experience uncontrollable stiff, jerky movements of their face and body, and other uncontrolled movements such as eye-blinking, sticking out the tongue, and arm-waving;
• KEVZARA SARILUMAB, RHEUMATOID ARTHRITIS
• KISQALI, RIBOCICLIB, To treat postmenopausal women with a type of advanced breast cancer
• Macrilen macimorelin acetate, For the diagnosis of adult growth hormone deficiency
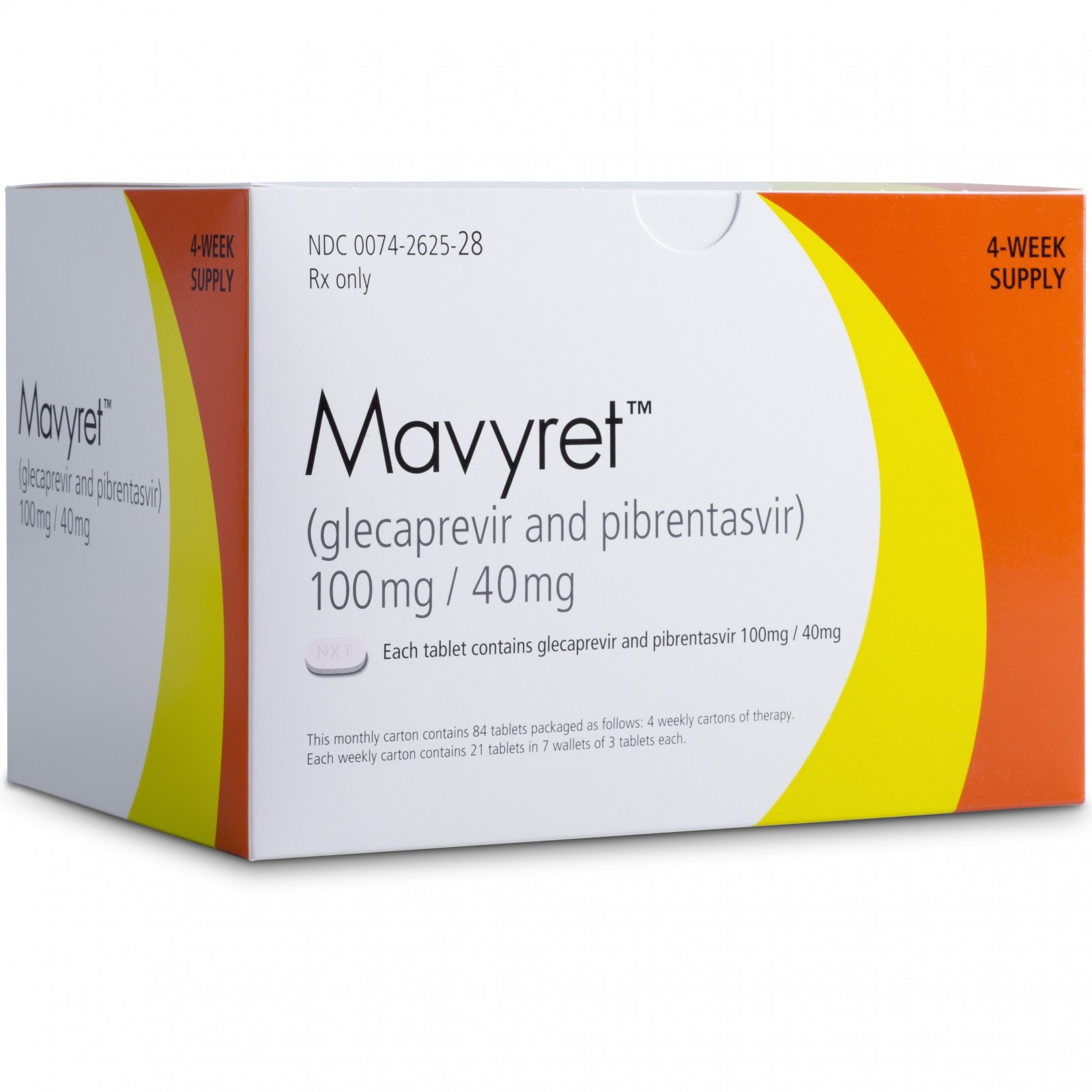
• Mavyret (glecaprevir and pibrentasvir) to treat adults with chronic hepatitis C virus genotypes 1-6 without cirrhosis (liver disease) or with mild cirrhosis, including patients with moderate to severe kidney disease, as well as those who are on hemodialysis;
• Mepsevii (vestronidase alfa-vjbk) to treat patients with Sly syndrome or mucopolysaccharidosis type 7 – a rare genetic disorder where an enzyme deficiency results in skeletal abnormalities, developmental delay, enlarged liver and spleen, and narrowed airways, which can lead to respiratory infections;
• Nerlynx (neratinib) for the extended adjuvant treatment — a form of therapy administered after an initial treatment to further lower the risk of the cancer coming back — of early-stage, human epidermal growth factor receptor 2 (HER2)-positive breast cancer;
• OCREVUS, OCRELIZUMAB, To treat patients with relapsing and primary progressive forms of multiple sclerosis
• OZEMPIC SEMAGLUTIDE To improve glycemic control in adults with type 2 diabetes mellitus
•PARSABIV, ETELCALCETIDE, To treat secondary hyperparathyroidism in adult patients with chronic kidney disease undergoing dialysis
• Prevymis (letermovir) for prevention of an infection called cytomegalovirus (CMV) in patients who are receiving a bone marrow transplant. CMV disease can cause serious health issues in these patients;
• Radicava (edaravone) to treat patients with amyotrophic lateral sclerosis, commonly referred to as Lou Gehrig’s disease, a rare disease that attacks and kills the nerve cells that control voluntary muscles;
• RHOPRESSA, NETARSUDIL, To treat glaucoma or ocular hypertension
• Rydapt (midostaurin) to treat adults newly diagnosed with a form of blood cancer known as acute myeloid leukemia who have a specific genetic mutation called FLT3, in combination with chemotherapy;
• Siliq (brodalumab) to treat adults with moderate-to-severe plaque psoriasis, a chronic disorder in which the body’s immune system sends out faulty signals that speed growth of skin cells that then accumulate, causing red, flaky patches that can appear anywhere on the body;
•SOLOSEC, SECNIDAZOLE To treat bacterial vaginosis
• STEGLATRO ERTUGLIFLOZIN To improve glycemic control in adults with type 2 diabetes mellitus
• Symproic (Naldemedine) for the treatment of opioid-induced constipation in adults with chronic noncancer pain; • Tremfya (guselkumab) for the treatment of adults with moderate-to-severe plaque psoriasis;
• Trulance (plecanatide) to treat adults with chronic idiopathic constipation, which is a persistent condition of constipation due to unknown origin;
• TYMLOS, Abaloparatide, To treat osteoporosis in postmenopausal women at high risk of fracture or those who have failed other therapies
• Vabomere (vaborbactam and meropenem) for treatment of adults with complicated urinary tract infections, including pyelonephritis (kidney infection) caused by bacteria;
• Verzenio (abemaciclib) to treat adults who have hormone receptor (HR)-positive, HER2-negative advanced or metastatic breast cancer that has progressed after taking therapy that alters a patient’s hormones (endocrine therapy);
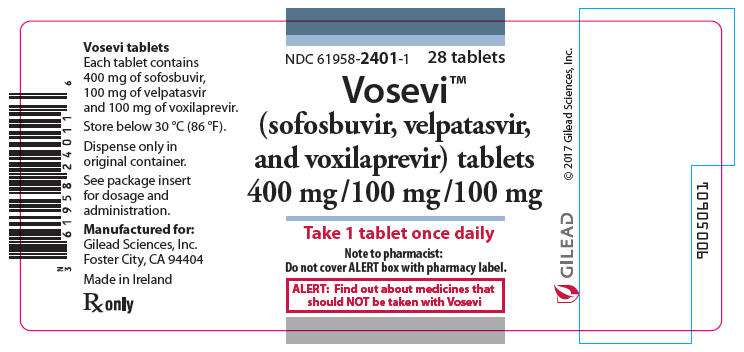
• Vosevi (sofosbuvir/velpatasvir/voxilaprevir) to treat adults with chronic hepatitis C virus genotypes 1-6 without cirrhosis (liver disease) or with mild cirrhosis;
• VYZULTA LATANOPROSTENE To treat intraocular pressure in patients with open-angle glaucoma or ocular hypertension.
• Xadago (safinamide) as an add-on treatment for patients with Parkinson’s disease who are currently taking levodopa/carbidopa and experiencing “off” episodes;
• XERMELO, TELOTRISTAT ETHYL combined with somatostatin analog (SSA) therapy to treat adults with carcinoid syndrome diarrhea that SSA therapy alone has inadequately controlled, and;
• XEPI OZENOXACIN TO TREAT IMPETIGO
•XERMELO, TELOTRISTAT ETHYL, To treat carcinoid syndrome diarrhea
• Zejula (niraparib) for the maintenance treatment (intended to delay cancer growth) of adults with recurrent epithelial ovarian, fallopian tube or primary peritoneal cancer, whose tumors have completely or partially shrunk (complete or partial response, respectively) in response to platinum-based chemotherapy
USFDA
* This information is currently accurate. In rare instances, it may be necessary for FDA to change a drug’s new molecular entity (NME) designation or the status of its application as a novel new biologics license application (BLA). For instance, new information may become available which could lead to a reconsideration of the original designation or status. If changes must be made to a drug’s designation or the status of an application as a novel BLA, the Agency intends to communicate the nature of, and the reason for, any revisions as appropriate.
| USFDA 2017 |
|
| 2017/12/21 |
|
|
Angiotensin II |
Giapreza |
La Jolla Pharmaceutical |
|
| 2017/12/20 |
|
|
Ertugliflozin |
Steglatro |
Merck Sharp Dohme |
|
| 2017/12/20 |
|
|
Macimorelin acetate |
Macrilen |
Aeterna Zentaris GmbH |
|
| 2017/12/18 |
|
|
Netarsudil mesylate |
Rhopressa |
Aerie Pharmaceuticals |
|
| 2017/12/11 |
|
|
Ozenoxacin |
Xepi |
Ferrer Internacional S.A. |
|
| 2017/12/5 |
|
|
Semaglutide |
Ozempic |
Novo Nordisk Inc |
|
| 2017/11/16 |
|
|
Emicizumab |
Hemlibra |
Genentech |
BLA |
| 2017/11/15 |
|
|
Vestronidase alfa |
Mepsevii |
Ultragenyx Pharmaceutical |
BLA |
| 2017/11/14 |
|
|
Benralizumab |
Fasenra |
AstraZeneca AB |
BLA |
| 2017/11/8 |
|
|
Letermovir |
Prevymis |
Merck Sharp Dohme |
|
| 2017/11/2 |
|
|
Latanoprostene bunod |
Vyzulta |
Bausch & Lomb Incorporated |
|
| 2017/10/31 |
|
|
Acalabrutinib |
Calquence |
AstraZeneca Pharmaceuticals LP |
|
| 2017/9/28 |
|
|
Abemaciclib |
Verzenio |
Eli Lilly |
|
| 2017/9/15 |
|
|
Secnidazole |
Solosec |
Symbiomix Therapeutics |
|
| 2017/9/14 |
|
|
Copanlisib |
Aliqopa |
Bayer Healthcare Pharmaceuticals |
|
| 2017/8/29 |
|
|
Benznidazole |
|
Chemo Research |
|
| 2017/8/29 |
|
|
Meropenem – Vaborbactam |
Vabomere |
Rempex Pharmaceuticals |
|
| 2017/8/17 |
|
|
Inotuzumab ozogamicin |
Besponsa |
Wyeth Pharmaceuticals |
BLA |
| 2017/8/3 |
|
|
Glecaprevir – Pibrentasvir |
Mavyret |
AbbVie |
|
| 2017/8/1 |
|
|
Enasidenib |
Idhifa |
Celgene Corporation |
|
| 2017/7/18 |
|
|
Sofosbuvir – Velpatasvir – Voxilaprevir |
Vosevi |
Gilead Sciences |
|
| 2017/7/17 |
|
|
Neratinib maleate |
Nerlynx |
Puma Biotechnology |
|
| 2017/7/13 |
|
|
Guselkumab |
Tremfya |
Janssen Biotech |
BLA |
| 2017/6/23 |
|
|
Betrixaban |
Bevyxxa |
Portola Pharmaceuticals |
|
| 2017/6/19 |
|
|
Delafloxacin meglumine |
Baxdela |
Melinta Therapeutics |
|
| 2017/5/22 |
|
|
Sarilumab |
Kevzara |
Sanofi Synthelabo |
BLA |
| 2017/5/5 |
|
|
Edaravone |
Radicava |
Mitsubishi Tanabe Pharma America |
|
| 2017/5/1 |
|
|
Durvalumab |
Imfinzi |
AstraZeneca UK |
BLA |
| 2017/4/28 |
|
|
Abaloparatide |
Tymlos |
Radius Health |
|
| 2017/4/28 |
|
|
Midostaurin |
Rydapt |
Novartis Pharmaceuticals |
|
| 2017/4/28 |
|
|
Brigatinib |
Alunbrig |
Ariad Pharmaceuticals |
|
| 2017/4/27 |
|
|
Cerliponase alfa |
Brineura |
BioMarin Pharmaceutical |
BLA |
| 2017/4/11 |
|
|
Valbenazine |
Ingrezza |
Neurocrine Biosciences |
|
| 2017/4/3 |
|
|
Deutetrabenazine |
Austedo |
Teva Pharmaceuticals |
|
| 2017/3/28 |
|
|
Ocrelizumab |
Ocrevus |
Genentech |
BLA |
| 2017/3/28 |
|
|
Dupilumab |
Dupixent |
Regeneron Pharmaceuticals |
BLA |
| 2017/3/27 |
|
|
Niraparib |
Zejula |
Tesaro |
|
| 2017/3/23 |
|
|
Naldemedine tosylate |
Symproic |
Shionogi |
|
| 2017/3/23 |
|
|
Avelumab |
Bavencio |
EMD Serono |
BLA |
| 2017/3/23 |
|
|
Safinamide mesylate |
Xadago |
Newron Pharmaceuticals |
|
| 2017/3/21 |
|
|
Ribociclib |
Kisqali |
Novartis Pharmaceuticals |
|
| 2017/2/28 |
|
|
Telotristat ethyl |
Xermelo |
Lexicon Pharmaceuticals |
|
| 2017/2/15 |
|
|
Brodalumab |
Siliq |
Valeant Pharmaceuticals |
BLA |
| 2017/2/9 |
|
|
Deflazacort |
Emflaza |
Marathon Pharmaceuticals |
|
| 2017/2/8 |
|
|
Etelcalcetide hydrochloride |
Parsavib |
KAI Pharmaceuticals |
|
| 2017/1/19 |
|
|
Plecanatide |
Trulance |
Synergy Pharmaceuticals |
RADIUS

Tymlos
FDA 4/28/2017
To treat osteoporosis in postmenopausal women at high risk of fracture or those who have failed other therapies
Drug Trials Snapshot
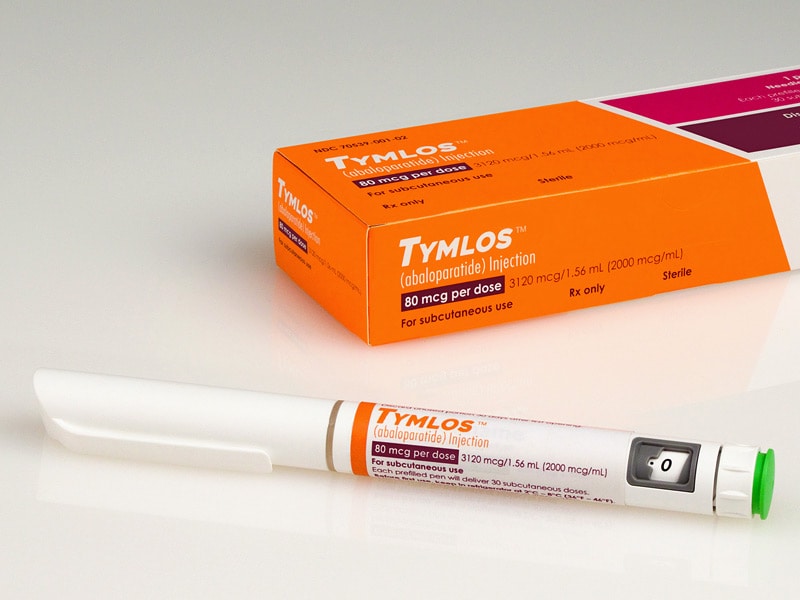

link……..https://newdrugapprovals.org/2018/02/13/abaloparatide-%D0%B0%D0%B1%D0%B0%D0%BB%D0%BE%D0%BF%D0%B0%D1%80%D0%B0%D1%82%D0%B8%D0%B4-%D8%A3%D8%A8%D8%A7%D9%84%D9%88%D8%A8%D8%A7%D8%B1%D8%A7%D8%AA%D9%8A%D8%AF-%E5%B7%B4%E7%BD%97%E6%97%81/
ELI LILLY
LINK https://newdrugapprovals.org/2015/10/19/abemaciclib-bemaciclib/



-Facebook.png)
LINK……….https://newdrugapprovals.org/2018/02/02/acalabrutinib-acp-196-%D0%B0%D0%BA%D0%B0%D0%BB%D0%B0%D0%B1%D1%80%D1%83%D1%82%D0%B8%D0%BD%D0%B8%D0%B1-%D8%A3%D9%83%D8%A7%D9%84%D8%A7%D8%A8%D8%B1%D9%88%D8%AA%D9%8A%D9%86%D9%8A%D8%A8-%E9%98%BF/
LA JOLLA


LINK https://newdrugapprovals.org/2017/12/22/fda-approves-drug-giapreza-angiotensin-ii-to-treat-dangerously-low-blood-pressure/
MERCK

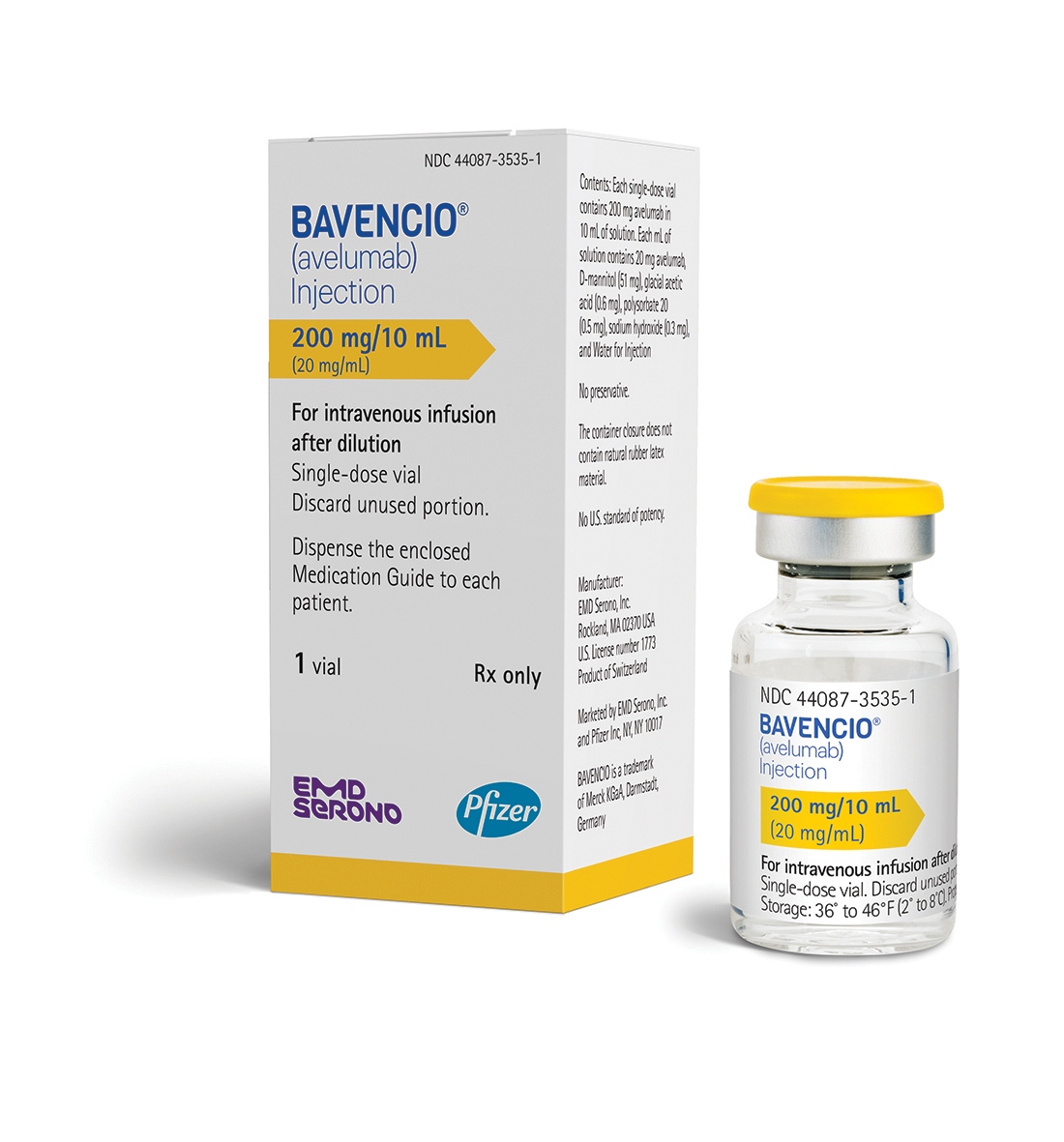
LINK…..https://newdrugapprovals.org/2017/03/24/fda-approves-first-treatment-bavencio-avelumabfor-rare-form-of-skin-cancer/
6 BENRALIZUMAB
ASTRA ZENECA
Fasenra benralizumab
FDA 11/14/2017
For add-on maintenance treatment of patients with severe asthma aged 12 years and older, and with an eosinophilic phenotype
Drug Trials Snapshot


CHEMO RESEARCH



LINK…https://newdrugapprovals.org/2017/08/30/fda-approves-first-u-s-treatment-benznidazole-for-chagas-disease/
PORTOLA PHARMA

| Bevyxxa |
|
FDA
6/23/2017 |
For the prophylaxis of venous thromboembolism (VTE) in adult patients hospitalized for an acute medical illness
Drug Trials Snapshot
|




LINK…….https://newdrugapprovals.org/2013/03/05/phase-3-portola-pharma-betrixaban-long-acting-oral-direct-factor-xa-inhibitor/
TAKEDA


| Alunbrig |
|
FDA
4/28/2017 |
To treat patients with anaplastic lymphoma kinase (ALK)-positive metastatic non-small cell lung cancer (NSCLC) who have progressed on or are intolerant to crizotinib
Drug Trials Snapshot |
LINK..https://newdrugapprovals.org/2017/01/20/brigatinib-%D0%B1%D1%80%D0%B8%D0%B3%D0%B0%D1%82%D0%B8%D0%BD%D0%B8%D0%B1-%D8%A8%D8%B1%D9%8A%D8%BA%D8%A7%D8%AA%D9%8A%D9%86%D9%8A%D8%A8-%E5%B8%83%E6%A0%BC%E6%9B%BF%E5%B0%BC/
VALEANT PHARMA

LINK ,,,,https://newdrugapprovals.org/2017/02/16/fda-approves-new-psoriasis-drug-siliq-brodalumab/



LINK….https://newdrugapprovals.org/2017/04/28/fda-approves-first-treatment-for-a-form-of-batten-disease-brineura-cerliponase-alfa/
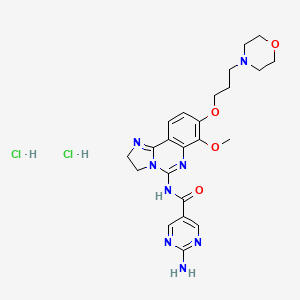

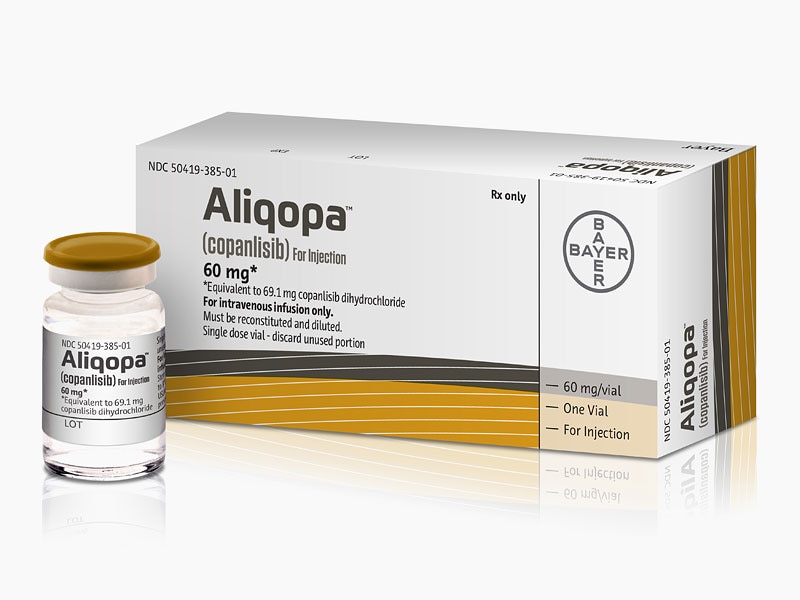
LINK…..https://newdrugapprovals.org/2017/11/20/copanlisib/
MARATHON PHARMA

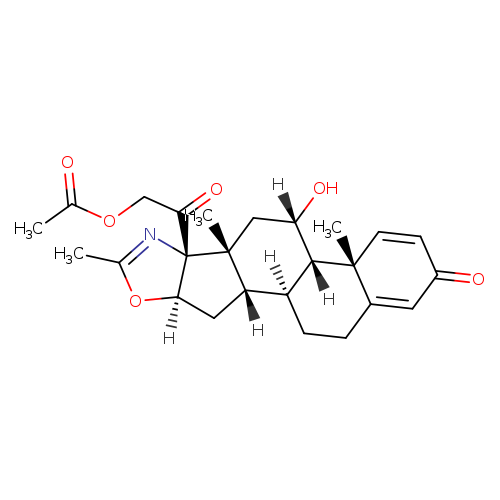
LINK……https://newdrugapprovals.org/2017/02/17/deflazacort/
| Baxdela |
|
FDA APPROVED
6/19/2017 |
To treat patients with acute bacterial skin infections |
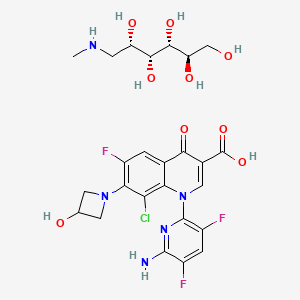


LINK……..https://newdrugapprovals.org/2018/01/25/delafloxacin/
TEVA
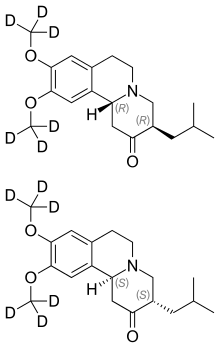


LINK……………https://newdrugapprovals.org/2015/08/15/sd-809-deutetrabenazine-nda-submitted-by-teva/



SANOFI/REGENERON


LINK…….https://newdrugapprovals.org/2017/03/29/fda-approves-new-eczema-drug-dupixent-dupilumab/
17 DURVALUMAB
ASTRA ZENECA

Imfinzi
durvalumab FDA 5/1/2017To treat patients with locally advanced or metastatic urothelial carcinoma
Web Post
Drug Trials Snapshot

MITSUBISHI TANABE


LINK………https://newdrugapprovals.org/2017/05/06/fda-approves-drug-to-treat-als-radicava-edaravone-%D1%8D%D0%B4%D0%B0%D1%80%D0%B0%D0%B2%D0%BE%D0%BD-%D8%A5%D9%8A%D8%AF%D8%A7%D8%B1%D8%A7%D9%81%D9%88%D9%86-%E4%BE%9D%E8%BE%BE%E6%8B%89%E5%A5%89/
ROCHE

LINK https://newdrugapprovals.org/2017/11/17/fda-approves-new-treatment-hemlibra-emicizumab-kxwh-to-prevent-bleeding-in-certain-patients-with-hemophilia-a/

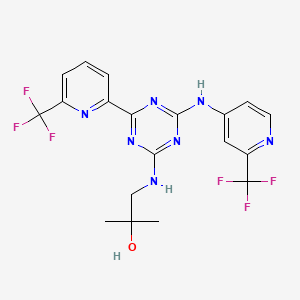



LINK……https://newdrugapprovals.org/2017/08/02/enasidenib-%D1%8D%D0%BD%D0%B0%D1%81%D0%B8%D0%B4%D0%B5%D0%BD%D0%B8%D0%B1-%D8%A5%D9%8A%D9%86%D8%A7%D8%B3%D9%8A%D8%AF%D9%8A%D9%86%D9%8A%D8%A8-%E4%BC%8A%E9%82%A3%E5%B0%BC%E5%B8%83/
MERCK

LINK https://newdrugapprovals.org/2014/02/10/ertugliflozin/

Amgen
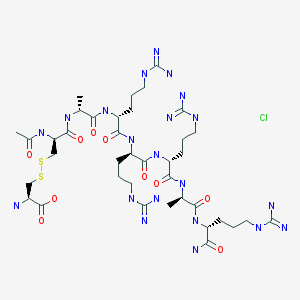


SYNTHESIS LINK……..https://cen.acs.org/articles/96/i4/the-year-in-new-drugs-2018.html
ABBVIE

LINK https://newdrugapprovals.org/2016/10/05/glecaprevir-abt-493/



24 GUSELKUMAB
JOHNSON AND JOHNSON
Tremfya
guselkumab
FDA 7/13/2017
For the treatment of adult patients with moderate-to-severe plaque psoriasis
Drug Trials Snapshot


PFIZER



LINK….https://newdrugapprovals.org/2015/10/23/fda-grants-breakthrough-status-for-pfizers-leukaemia-drug-inotuzumab-ozogamicin/
VALEANT
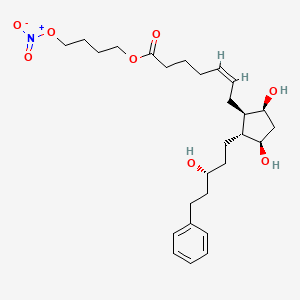
latanoprostene bunod ophthalmic solution
FDA 11/2/2017
To treat intraocular pressure in patients with open-angle glaucoma or ocular hypertension.
Drug Trials Snapshot

LINK https://newdrugapprovals.org/2014/09/27/nicox-stock-leaps-on-positive-ph-iii-glaucoma-drug-data-%E8%8B%B1%E6%96%87%E5%90%8D%E7%A7%B0/
MERCK
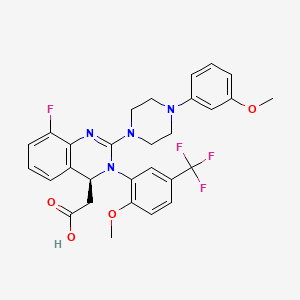
LINK https://newdrugapprovals.org/2016/05/16/letermovir-aic-246/


AETERNA ZENTARIS

LINK https://newdrugapprovals.org/2014/01/07/aeterna-zentaris-submits-new-drug-application-to-fda-for-macimorelin-acetate-aezs-130-for-evaluation-of-aghd-2/


NOVARTIS




LINK…….https://newdrugapprovals.org/2017/04/29/fda-approves-new-combination-treatment-for-acute-myeloid-leukemia-rydapt-midostaurin/
FDA 3/23/2017, Symproic, For the treatment of opioid-induced constipation



LINK……..https://newdrugapprovals.org/2018/01/24/naldemedine-%E3%83%8A%E3%83%AB%E3%83%87%E3%83%A1%E3%82%B8%E3%83%B3%E3%83%88%E3%82%B7%E3%83%AB%E9%85%B8%E5%A1%A9/
PUMA BIOTECH
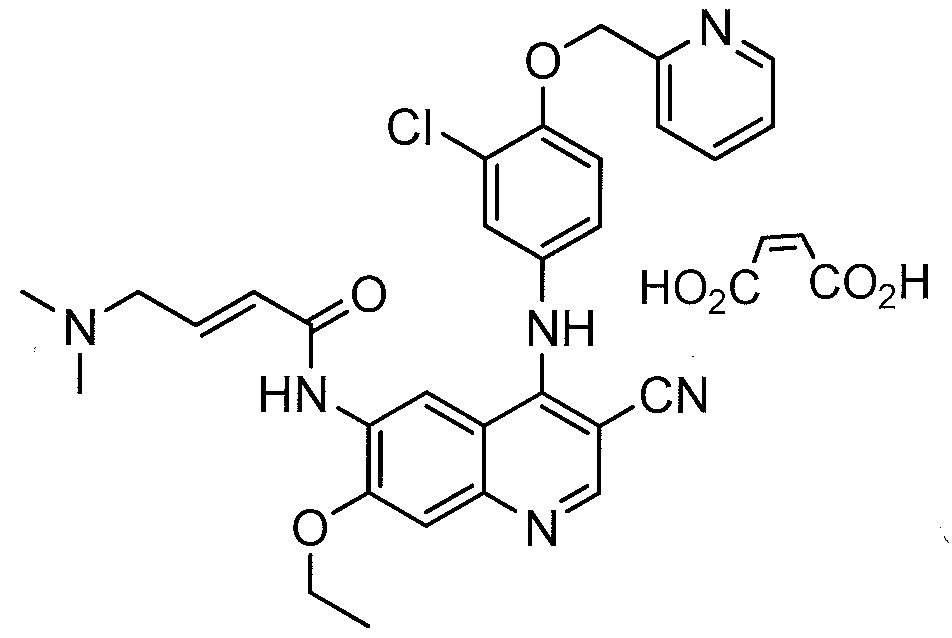



LINK…https://newdrugapprovals.org/2014/04/11/neratinib-hki-272-puma-presents-positive-results-from-phase-ii-trial-of-its-investigational-drug-pb272/
| Rhopressa |
|
FDA APPROVED
12/18/2017 |
To treat glaucoma or ocular hypertension |


LINK……https://newdrugapprovals.org/2018/01/29/netarsudil/
TESARO



LINK…https://newdrugapprovals.org/2016/12/22/niraparib-mk-4827/
ROCHE


LINK..https://newdrugapprovals.org/2017/03/30/fda-approves-new-drug-to-treat-multiple-sclerosis-ocrevus-ocrelizumab/
MEDIMETRIX
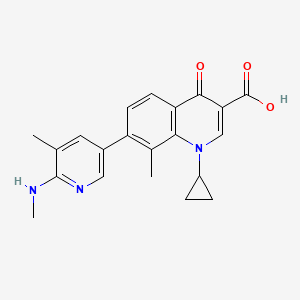
LINK https://newdrugapprovals.org/2014/03/28/ozenoxacin-in-phase-3-topical-formulation-in-the-treatment-of-impetigo/

ABBVIE

LINK https://newdrugapprovals.org/2016/06/08/abt-530-pibrentasvir/

Plecanatide 普卡那肽 ليكاناتيد плеканатид
SYNERGY PHARMA


LINK ….https://newdrugapprovals.org/2016/04/21/plecanatide-%E6%99%AE%E5%8D%A1%E9%82%A3%E8%82%BD-%D9%84%D9%8A%D9%83%D8%A7%D9%86%D8%A7%D8%AA%D9%8A%D8%AF-%D0%BF%D0%BB%D0%B5%D0%BA%D0%B0%D0%BD%D0%B0%D1%82%D0%B8%D0%B4/
NOVARTIS

Structure..link for correct structure

LINK https://newdrugapprovals.org/2015/10/19/ribociclib/
SANOFI /REGENERON

LINK https://newdrugapprovals.org/2013/11/25/late-stage-success-for-sanofiregeneron-ra-drug-sarilumab/


SYMBIOMIX


link….https://newdrugapprovals.org/2017/11/03/secnidazole-%D1%81%D0%B5%D0%BA%D0%BD%D0%B8%D0%B4%D0%B0%D0%B7%D0%BE%D0%BB-%D8%B3%D9%8A%D9%83%D9%86%D9%8A%D8%AF%D8%A7%D8%B2%D9%88%D9%84-%E5%A1%9E%E5%85%8B%E7%A1%9D%E5%94%91/
NEWRON PHARMA




LINK…https://newdrugapprovals.org/2017/03/22/fda-approves-drug-xadago-safinamide-%D1%81%D0%B0%D1%84%D0%B8%D0%BD%D0%B0%D0%BC%D0%B8%D0%B4-%D8%B3%D8%A7%D9%81%D9%8A%D9%86%D8%A7%D9%85%D9%8A%D8%AF-%E6%B2%99%E9%9D%9E%E8%83%BA-to-treat-parkins/
NOVO NORDISK
LINK https://newdrugapprovals.org/2013/07/22/a-survey-of-promising-late-stage-diabetes-drugs/

LINK https://newdrugapprovals.org/2013/12/11/us-approves-breakthrough-hepatitis-c-drug-sofosbuvir-all-about-drugs/
LEXICON


THE MEDICINES CO


LINK https://newdrugapprovals.org/2017/09/05/vaborbactam-%D0%B2%D0%B0%D0%B1%D0%BE%D1%80%D0%B1%D0%B0%D0%BA%D1%82%D0%B0%D0%BC-%D9%81%D8%A7%D8%A8%D9%88%D8%B1%D8%A8%D8%A7%D9%83%D8%AA%D8%A7%D9%85-%E6%B3%95%E7%A1%BC%E5%B7%B4%E5%9D%A6/
NEUROCRINE


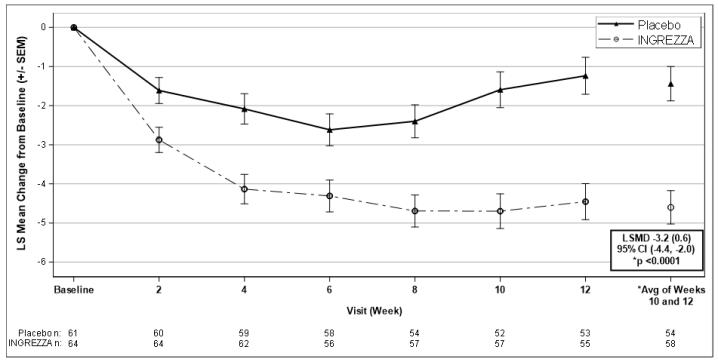
LINK…………..https://newdrugapprovals.org/2017/04/12/fda-approves-first-drug-ingrezza-valbenazine-to-treat-tardive-dyskinesia/
ULTRAGENYX

LINK…https://newdrugapprovals.org/2017/11/16/fda-approves-mepsevii-vestronidase-alfa-vjbk-for-treatment-for-rare-genetic-enzyme-disorder/
LINK https://newdrugapprovals.org/2016/07/30/velpatasvir-gs-5816-gilead-sciences-%D0%B2%D0%B5%D0%BB%D0%BF%D0%B0%D1%82%D0%B0%D1%81%D0%B2%D0%B8%D1%80-%D9%81%D8%A7%D9%84%D8%A8%D8%A7%D8%AA%D8%A7%D8%B3%D9%81%D9%8A%D8%B1-%E7%BB%B4%E5%B8%95/
GILEAD
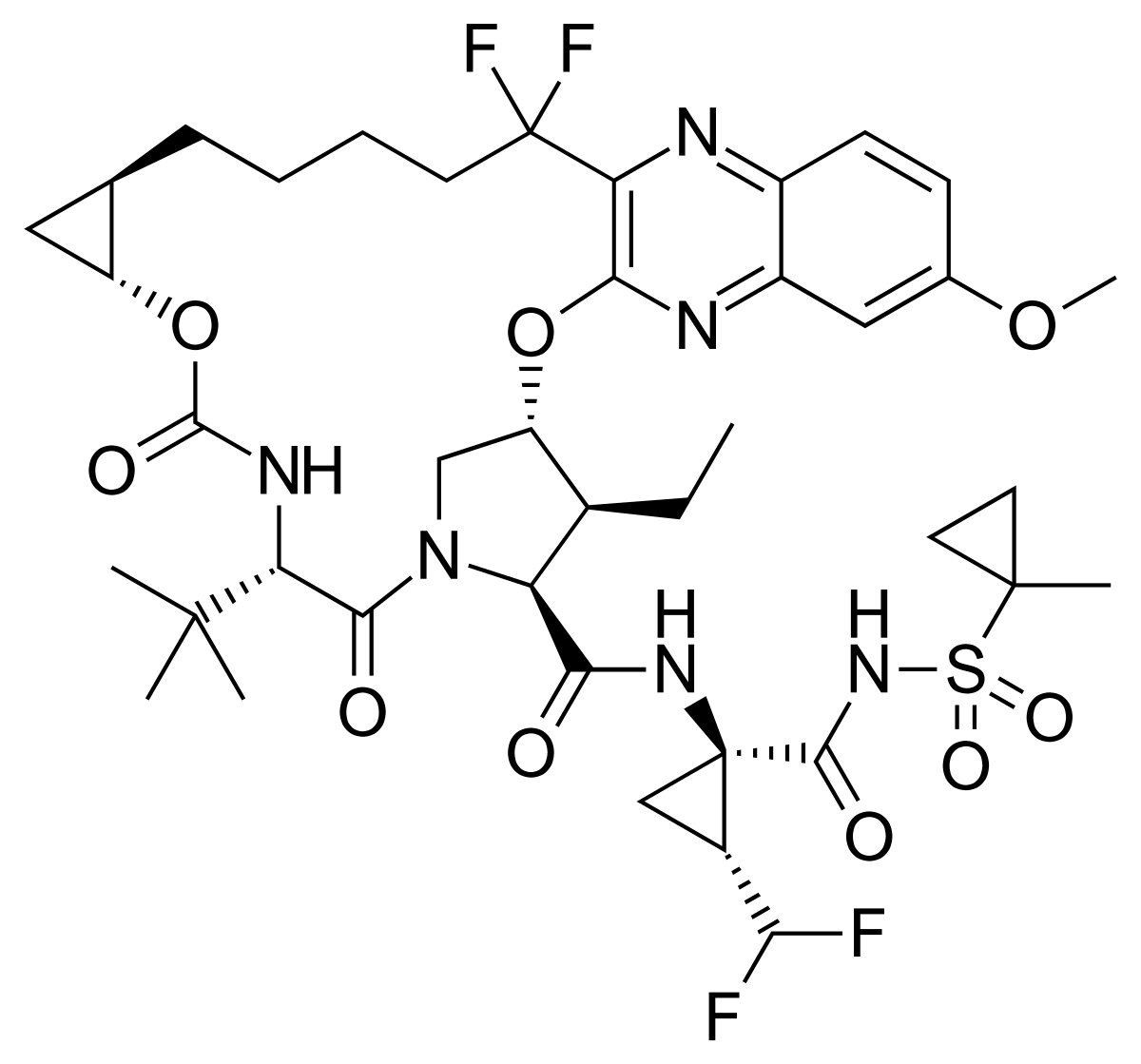
LINK https://newdrugapprovals.org/2017/07/19/voxilaprevir-%D9%81%D9%88%D9%83%D8%B3%D9%8A%D9%84%D8%A7%D8%A8%D8%B1%D9%8A%D9%81%D9%8A%D8%B1-%E4%BC%8F%E8%A5%BF%E7%91%9E%E9%9F%A6-%D0%B2%D0%BE%D0%BA%D1%81%D0%B8%D0%BB%D0%B0%D0%BF%D1%80%D0%B5%D0%B2/
SECTION B; EMA approvals
European Medicines Agency’s – Human medicines: Highlights of 2017
Advances in medicines authorizations are essential for public health as they have the potential to improve treatment of diseases. In 2017, EMA recommended 92 medicines for marketing authorization. Of these, 35 had a new active substance, which has never been authorized in the European Union (EU) before. Many of these medicines represent a significant improvement in their therapeutic areas; they include medicines for children, for rare diseases and advanced therapies42. Amongst the 35 new active substances (NAS) that EMA recommended, 11 were new drugs and biologics to treat cancer, 05 to treat neurological disorders, 04 for infectious diseases, 04 for immunology/rheumatology, 03 for endocrinology, 02 each for Uro-nephrology, haematology, and dermatology, 01 for Pneumonology, and 01 for hepatology/gastroenterology class of drugs.







EUROPE
| 2017/11/16 |
|
|
Niraparib |
Zejula |
Tesaro UK Limited |
O |
NME |
| 2017/11/10 |
|
|
Adalimumab |
Cyltezo |
Boehringer Ingelheim International GmbH |
B |
|
| 2017/11/10 |
|
|
Miglustat |
Miglustat Gen.Orph |
Gen.Orph |
G |
|
| 2017/11/10 |
|
|
Ritonavir |
Ritonavir Mylan |
MYLAN S.A.S |
G |
|
| 2017/11/10 |
|
|
Padeliporfin |
Tookad |
STEBA Biotech S.A |
|
|
| 2017/11/10 |
|
|
Guselkumab |
Tremfya |
Janssen-Cilag International N.V. |
|
BLA |
| 2017/9/27 |
|
|
Dupilumab |
Dupixent |
sanofi-aventis groupe |
|
BLA |
| 2017/9/21 |
|
|
Darunavir / Cobicistat / Emtricitabine / Tenofovir alafenamide |
Symtuza |
Janssen-Cilag International N.V. |
|
|
| 2017/9/21 |
|
|
Atezolizumab |
Tecentriq |
Roche Registration Limited |
|
BLA |
| 2017/9/18 |
|
|
Avelumab |
Bavencio |
Merck Serono Europe Limited |
O |
BLA |
| 2017/9/18 |
|
|
Entecavir |
Entecavir Mylan |
Mylan S.A.S |
G |
|
| 2017/9/18 |
|
|
Lacosamide |
Lacosamide Accord |
Accord Healthcare Ltd |
G |
|
| 2017/9/18 |
|
|
Midostaurin |
Rydapt |
Novartis Europharm Ltd |
O |
NME |
| 2017/9/18 |
|
|
Telotristat ethyl |
Xermelo |
Ipsen Pharma |
O |
NME |
| 2017/9/5 |
|
|
Trientine |
Cuprior |
GMP-Orphan SA |
|
|
| 2017/9/5 |
|
|
Efavirenz / Emtricitabine / Tenofovir disoproxil |
Efavirenz/Emtricitabine/Tenofovir disoproxil Mylan |
Mylan S.A.S |
G |
|
| 2017/8/24 |
|
|
Tivozanib hydrochloride monohydrate |
Fotivda |
EUSA Pharma (UK) Limited |
|
NME |
| 2017/8/24 |
|
|
Adalimumab |
Imraldi |
Samsung Bioepis UK Limited (SBUK) |
B |
|
| 2017/8/24 |
|
|
Nitisinone |
Nitisinone MDK (previously Nitisinone MendeliKABS) |
MendeliKABS Europe Ltd |
G |
|
| 2017/8/22 |
|
|
Ribociclib |
Kisqali |
Novartis Europharm Ltd |
|
NME |
| 2017/8/22 |
|
|
Cladribine |
Mavenclad |
Merck Serono Europe Limited |
|
|
| 2017/7/26 |
|
|
Glecaprevir / Pibrentasvir |
Maviret |
AbbVie Limited |
|
NME |
| 2017/7/26 |
|
|
Sofosbuvir / Velpatasvir / Voxilaprevi |
Vosevi |
Gilead Sciences International Ltd |
|
NME |
| 2017/7/19 |
|
|
Insulin lispro |
Insulin lispro Sanofi |
sanofi-aventis groupe |
B |
|
| 2017/7/19 |
|
|
Patiromer sorbitex calcium |
Veltassa |
Vifor Fresenius Medical Care Renal Pharma France |
|
NME |
| 2017/7/17 |
|
|
Efavirenz / Emtricitabine / Tenofovir disoproxil |
Efavirenz/Emtricitabine/Tenofovir disoproxil Zentiva |
Zentiva k.s. |
G |
|
| 2017/7/17 |
|
|
Brodalumab |
Kyntheum |
LEO Pharma A/S |
|
BLA |
| 2017/7/17 |
|
|
beclometasone / formoterol / glycopyrronium bromide |
Trimbow |
Chiesi Farmaceutici S.p.A. |
|
|
| 2017/7/13 |
|
|
Rituximab |
Blitzima |
Celltrion Healthcare Hungary Kft. |
B |
|
| 2017/7/13 |
|
|
Cariprazine |
Reagila |
Gedeon Richter |
|
|
| 2017/7/10 |
|
|
Spheroids of human autologous matrix-associated chondrocytes |
Spherox |
CO.DON AG |
|
|
| 2017/7/6 |
|
|
Cenegermin |
Oxervate |
Dompe farmaceutici s.p.a. |
O |
BLA |
| 2017/6/29 |
|
|
Inotuzumab ozogamicin |
Besponsa |
Pfizer Limited |
O |
BLA |
| 2017/6/23 |
|
|
Etanercept |
Erelzi |
Sandoz GmbH |
|
|
| 2017/6/23 |
|
|
Sarilumab |
Kevzara |
Sanofi-Aventis Groupe |
|
NME |
| 2017/6/23 |
|
|
Dimethyl fumarate |
Skilarence |
Almirall S.A |
|
|
| 2017/6/23 |
|
|
Carglumic acid |
Ucedane |
Lucane Pharma |
G |
|
| 2017/6/15 |
|
|
Rituximab |
Rixathon, Riximyo B |
Sandoz GmbH |
|
|
| 2017/6/2 |
|
|
Pentosan polysulfate sodium |
Elmiron |
bene-Arzneimittel GmbH |
|
|
| 2017/6/2 |
|
|
Nonacog beta pegol |
Refixia |
Novo Nordisk A/S |
|
BLA |
| 2017/5/30 |
|
|
Cerliponase alfa |
Brineura |
BioMarin International Limited |
O E |
BLA |
| 2017/5/30 |
|
|
Nusinersen |
Spinraza |
Biogen Idec Ltd |
O |
NME |
| 2017/5/24 |
|
|
Meningococcal group b vaccine (recombinant, adsorbed) |
Trumenba |
Pfizer Limited |
|
|
| 2017/5/22 |
|
|
Ivabradine |
Ivabradine Accord |
Accord Healthcare Ltd |
G |
|
| 2017/5/8 |
|
|
Dinutuximab beta |
Dinutuximab beta Apeiron |
Apeiron Biologics AG |
O E |
|
| 2017/4/28 |
|
|
Emtricitabine – tenofovir disoproxil mixt |
Emtricitabine/Tenofovir disoproxil Krka d.d. |
KRKA, d.d., Novo mesto |
G |
|
| 2017/4/24 |
|
|
Parathyroid hormone |
Natpar |
Shire Pharmaceuticals Ireland Ltd |
O C |
BLA |
| 2017/4/20 |
|
|
Edoxaban |
Roteas |
Daiichi Sankyo Europe GmbH |
|
|
| 2017/3/22 |
|
|
Tofacitinib citrate |
Xeljanz |
Pfizer Limited |
|
NME |
| 2017/3/20 |
|
|
Umeclidinium |
Rolufta |
GlaxoSmithKline Trading Services Limited |
|
|
| 2017/3/3 |
|
|
Chlormethine |
Ledaga |
Actelion Registration Ltd. |
O |
|
| 2017/2/27 |
|
|
Pregabalin |
Pregabalin Zentiva |
Zentiva k.s. |
G |
|
| 2017/2/17 |
|
|
Rituximab |
Truxima |
Celltrion Healthcare Hungary Kft. |
B |
|
| 2017/2/13 |
|
|
Etanercept |
Lifmior |
Pfizer Limited |
|
|
| 2017/2/13 |
|
|
Baricitinib |
Olumiant |
Eli Lilly Nederland B.V. |
|
NME |
| 2017/1/19 |
|
|
Mercaptamine |
Cystadrops |
Orphan Europe S.A.R.L. |
O |
|
| 2017/1/18 |
|
|
Bezlotoxumab |
Zinplava |
Merck Sharp & Dohme Limited |
|
NME |
| 2017/1/11 |
|
|
Teriparatide |
Movymia |
STADA Arzneimittel AG |
B |
|
| 2017/1/11 |
|
|
Insulin glargine / lixisenatide |
Suliqua |
Sanofi-Aventis Groupe |
|
|
| 2017/1/9 |
|
|
Insulin aspart |
Fiasp |
Novo Nordisk A/S |
|
|
| 2017/1/9 |
|
|
Tadalafil |
Tadalafil |
Mylan S.A.S |
G |
|
| 2017/1/9 |
|
|
Tenofovir alafenamide |
Vemlidy |
Gilead Sciences International Ltd |
|
|
| 2017/1/4 |
|
|
Lonoctocog alfa |
Afstyla |
CSL Behring GmbH |
|
BLA |
| 2017/1/4 |
|
|
Darunavir |
Darunavir Mylan |
Mylan S.A.S. |
G |
|
| 2017/1/4 |
|
|
Insulin glargine |
Lusduna |
Merck Sharp & Dohme Limited |
B |
|
| 2017/1/4 |
|
|
Teriparatide |
Terrosa |
Gedeon Richter Plc. |
B |
SECTION B; EMA Approvals
Combined drugs USFDA+EMA +PMDA list are listed below. trying to simplify search
1 Abaloparatide USFDA
2 Abemaciclib USFDA
3 ACALABRUTINIB USFDA
3A ALOFISEL EMA
3B AMENAMEVIR JAPAN
4 ANGIOTENSIN II USFDA
4A Atezolizumab EMA
5 AVELUMAB USFDA+EMA
6 BENRALIZUMAB USFDA+EMA
6A BARICITINIB JAPAN
7 BENZNIDAZOLE USFDA
8 BETRIXABAN USFDA
9 BRIGATINIB USFDA
10 BRODALUMAB USFDA+EMA
10A BUROSUMAB EMA
10B CARIPRAZINE HYDROCHLORIDE EMA
11 CERLIPONASE ALPA USFDA+EMA
12 COPANLISIB USFDA
13 DEFLAZACORT USFDA
14 Delafloxacin USFDA
15 Deutetrabenazine USFDA
16DUPILUMAB USFDA+EMA
17 DURVALUMAB USFDA
18 EDAVARONE USFDA
19 EMICIZUMAB USFDA
20 Enasidenib USFDA
21 ERTUGLIFLOZIN USFDA
22 ETELCALCETIDE USFDA
22A FORODESINE JAPAN
22B FLUCICLOVINE EMA
23 GLECAPREVIR USFDA+EMA
24 GUSELKUMAB USFDA+EMA
25 INOTUZUMAB OZOGAMICIN USFDA+EMA
26 LATANOPROSTENE USFDA
27 LETERMOVIR USFDA+EMA
27A Utetium lu 177 dotatate EMA
28 MACIMORELIN ACETATE USFDA
29 MEROPENEM USFDA
30 MIDOSTAURIN USFDA+EMA
31 NALDEMEDINE USFDA
32 NERATINIB USFDA
33 NETARSUDIL USFDA
34 NIRAPARIB USFDA+EMA
34A NONACOG EMA
34B NUCINERSEN EMA +Japan
35 Ocrelizumab USFDA+EMA
35A OXERVATE EMA
36 OZENOXACIN USFDA
36A PATIROMER EMA
36B PADELIPORFIN EMA
36C PEMAFIBRATE JAPAN
37 PIBRENTASVIR USFDA+EMA
38 PLECANATIDE USFDA
38A PRALATREXATE JAPAN
39 RIBOCICLIB USFDA+EMA
39A ROLAPITANT EMA
39BRURLOCTOCOG EMA
40 SARILUMAB USFDA+EMA
41 SECNIDAZOLE USFDA
42 SAFINAMIDE USFDA
43 SEMAGLUTIDE USFDA+EMA
43A SODIUM ZIRCONIUM CYCLOCYLICATE EMA
44 SOFOSBUVIR USFDA+EMA
44A SPHEROX EMA
45 TELOTRISTAT ETHYL USFDA+EMA
45A TIVOZANIB EMA
45B TOFACITINIB EMA
45C TRUMENBA EMA
46 VABORBACTAM USFDA
47 VALBENAZINE USFDA
48 VESTRONIDASE ALFA-VJBK USFDA
49 VELPATASVIR USFDA+EMA
50 VOXILAPREVIR USFDA+EMA
Drugs EMA list missed out in usfda list
link………https://newdrugapprovals.org/2018/03/02/alofisel-darvadstrocel-cx-601/
4A Atezolizumab
WILL BE UPDATED
10A BUROSUMAB
WILL BE UPDATED
10B CARIPRAZINE HYDROCHLORIDE
WILL BE UPDATED


LINK https://newdrugapprovals.org/2016/05/28/fda-approves-new-diagnostic-imaging-agent-fluciclovine-f-18-to-detect-recurrent-prostate-cancer/
SEE EMA
http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/human/medicines/004197/human_med_002100.jsp&mid=WC0b01ac058001d124
| Marketing-authorisation holder |
Blue Earth Diagnostics Ltd |
| Revision |
0 |
| Date of issue of marketing authorisation valid throughout the European Union |
22/05/2017 |
Contact address:
Blue Earth Diagnostics Ltd
215 Euston Road
London NW1 2BE
United Kingdom
27A Lutetium lu 177 dotatate
WILL BE UPDATED
34A NONACOG
WILL BE UPDATED
EMA AND JAPAN 2017 APPROVED


LINK …….https://newdrugapprovals.org/2018/03/14/nusinersen-sodium-%E3%83%8C%E3%82%B7%E3%83%8D%E3%83%AB%E3%82%BB%E3%83%B3%E3%83%8A%E3%83%88%E3%83%AA%E3%82%A6%E3%83%A0/
35A OXERVATE
WILL BE UPDATED
36A PATIROMER
WILL BE UPDATED

| NAME |
Tookad |
| AGENCY PRODUCT NUMBER |
EMEA/H/C/004182 |
| ACTIVE SUBSTANCE |
padeliporfin di-potassium |
| INTERNATIONAL NON-PROPRIETARY NAME(INN) OR COMMON NAME |
padeliporfin |
| THERAPEUTIC AREA |
Prostatic Neoplasms |
| ANATOMICAL THERAPEUTIC CHEMICAL (ATC) CODE |
L01XD07 |
| ADDITIONAL MONITORING |
This medicine is under additional monitoring. This means that it is being monitored even more intensively than other medicines. For more information, see medicines under additional monitoring. |
| MARKETING-AUTHORISATION HOLDER |
STEBA Biotech S.A |
| REVISION |
0 |
| DATE OF ISSUE OF MARKETING AUTHORISATION VALID THROUGHOUT THE EUROPEAN UNION |
10/11/2017 |
Contact address:
STEBA Biotech S.A
7 place du theatre
L-2613 Luxembourg
Luxembourg


Japan approved 2017
| 2017/7/3 |
PMDA |
JAPAN |
|
Pralatrexate |
Difolta |
Mundipharma |
NME |
LINK https://newdrugapprovals.org/2018/03/16/pralatrexate-%E3%83%97%E3%83%A9%E3%83%A9%E3%83%88%E3%83%AC%E3%82%AD%E3%82%B5%E3%83%BC%E3%83%88/
39A ROLAPITANT
WILL BE UPDATED
39B RURLOCTOCOG
WILL BE UPDATED
43A SODIUM ZIRCONIUM
WILL BE UPDATED
44A SPHEROX
WILL BE UPDATED

Pharmacotherapeutic group
Antineoplastic agents
Therapeutic indication
Fotivda is indicated for the first line treatment of adult patients with advanced renal cell carcinoma (RCC) and for adult patients who are VEGFR and mTOR pathway inhibitor-naïve following disease progression after one prior treatment with cytokine therapy for advanced RCC.
Treatment of advanced renal cell carcinoma
Fotivda : EPAR -Product Information
http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/human/medicines/004131/human_med_002146.jsp&mid=WC0b01ac058001d124
http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Public_assessment_report/human/004131/WC500239035.pdf

Tivozanib is synthesized in three main steps using well defined starting materials with acceptable
specifications.
Adequate in-process controls are applied during the synthesis. The specifications and control methods for
intermediate products, starting materials and reagents have been presented. The critical process
parameters are duly justified, methodology is presented and control is adequate.
The characterisation of the active substance and its impurities are in accordance with the EU guideline on
chemistry of new active substances. Potential and actual impurities were well discussed with regards to
their origin and characterised.
The active substance is packaged in a low-density polyethylene (LDPE) bag which complies with the EC
directive 2002/72/EC and EC 10/2011 as amended.
Product details
| Name |
Fotivda |
| Agency product number |
EMEA/H/C/004131 |
| Active substance |
tivozanib |
| International non-proprietary name(INN) or common name |
tivozanib hydrochloride monohydrate |
| Therapeutic area |
Carcinoma, Renal Cell |
| Anatomical therapeutic chemical (ATC) code |
L01XE |
Publication details
| Marketing-authorisation holder |
EUSA Pharma (UK) Limited |
| Revision |
0 |
| Date of issue of marketing authorisation valid throughout the European Union |
24/08/2017 |
Contact address:
EUSA Pharma (UK) Limited
Breakspear Park, Breakspear Way
Hemel Hempstead, HP2 4TZ
United Kingdom
LINK………https://newdrugapprovals.org/2018/02/26/tivozanib-%E3%83%86%E3%82%A3%E3%83%9C%E3%82%B6%E3%83%8B%E3%83%96%E5%A1%A9%E9%85%B8%E5%A1%A9%E6%B0%B4%E5%92%8C%E7%89%A9/
45B TOFACITINIB
WILL BE UPDATED
45C TRUMENBA
WILL BE UPDATED
SECTION C JAPANFORODOS

SECTION C New Drugs JAPAN
https://www.pmda.go.jp/english/review-services/reviews/approved-information/drugs/0002.html



JAPAN 2017
| 2017/9/27 |
Avelumab (genetical recombination) |
Bavencio |
Merck Serono |
BLA |
| 2017/9/27 |
Glecaprevir – pibrentasvir mixt |
Maviret |
Abbvie |
NME |
| 2017/9/27 |
Daratumumab (genetical recombination) |
Darzalex |
Janssen Pharmaceutical |
BLA |
| 2017/9/27 |
Belimumab (genetical recombination) |
Benlysta |
GlaxoSmithKline |
BLA |
| 2017/9/27 |
Bezlotoxumab (genetical recombination) |
Zinplava |
MDS |
BLA |
| 2017/9/27 |
Palbociclib |
Ibrance |
Pfizer |
NME |
| 2017/9/27 |
Lonoctocog alfa (genetical recombination) |
Afstyla |
CSL Behring |
BLA |
| 2017/9/27 |
Rupatadine fumarate |
Rupafin |
Teikoku seiyaku |
NME |
| 2017/9/27 |
Sarilumab (genetical receombination) |
Kevzara |
Sanofi |
BLA |
| 2017/9/27 |
Flutemetamol (18F) |
Vizamyl |
Nihon Medi-Physics |
NME |
| 2017/7/3 |
Nusinersen sodium |
Spinraza |
Biogen Japan |
| 2017/7/3 |
Romidepsin |
Istodax |
Celgene |
NME |
| 2017/7/3 |
Pralatrexate |
Difolta |
Mundipharma |
NME |
| 2017/7/3 |
Amenamevir |
Amenalief |
Maruho |
NME |
| 2017/7/3 |
Baricitinib |
Olumiant |
Lilly |
NME |
| 2017/7/3 |
Pemafibrate |
Parmodia |
Kowa |
NME |
| 2017/3/30 |
Human prothrombin complex, freeze-dried concentrated |
Kcentra |
CSL Behring |
| 2017/3/30 |
Ixazomib citrate |
Ninlaro |
Takeda |
NME |
| 2017/3/30 |
Forodesine hydrochloride |
Mundesine |
Mundipharma |
| 2017/3/30 |
Aflibercept beta (genetical recombination) |
Zaltrap |
Sanofi |
| 2017/3/30 |
Hydromorphone hydrochloride |
Narusus, Narurapid |
DaiichiSankyo-pp |
| 2017/3/30 |
Naldemedine tosylate |
Symproic |
Shionogi |
NME |
| 2017/3/30 |
Guanfacine hydrochloride |
Intuniv |
Shionogi |

Originally developed by Astellas, the drug was licensed to Maruho. Amenamevir treats herpes zoster by inhibiting the activity of the helicase-primer enzyme during viral DNA replication and blocking the virus’s proliferation.
Amenalief® is an oral film-coated tablet containing 200 mg of amenamevir per tablet. Recommended dose of 1 day, 400mg each time, after meals.
LINK https://newdrugapprovals.org/2018/03/12/amenamevir-%E3%82%A2%E3%83%A1%E3%83%8A%E3%83%A1%E3%83%93%E3%83%AB/
CDSCO INDIA


http://www.cdsco.nic.in/forms/list.aspx?lid=2034&Id=11 http://www.cdsco.nic.in/forms/list.aspx?lid=2034&Id=11








KEEP WATCHING UNDER CONSTRUCTION AND WILL BE PASTED SOON………………………………………..
KEEP WATCHING UNDER CONSTRUCTION AND WILL BE PASTED SOON………………………………………..
KEEP WATCHING UNDER CONSTRUCTION AND WILL BE PASTED SOON………………………………………..
KEEP WATCHING UNDER CONSTRUCTION AND WILL BE PASTED SOON………………………………………..
REFERENCES
1 http://www.ema.europa.eu/ema/index.jsp?curl=pages/news_and_events/news/2018/01/news_detail_002886.jsp&mid=WC0b01ac058004d5c1
2 http://www.ema.europa.eu/docs/en_GB/document_library/Report/2018/01/WC500242079.pdf
“NEW DRUG APPROVALS” CATERS TO EDUCATION GLOBALLY, No commercial exploits are done or advertisements added by me. This is a compilation for educational purposes only. P.S. : The views expressed are my personal and in no-way suggest the views of the professional body or the company that I represent
Join me on Linkedin

Join me on Facebook  FACEBOOK
FACEBOOK
Join me on twitter

Join me on Researchgate

 amcrasto@gmail.com
amcrasto@gmail.com
I , Dr A.M.Crasto is writing this blog to share the knowledge/views, after reading Scientific Journals/Articles/News Articles/Wikipedia. My views/comments are based on the results /conclusions by the authors(researchers). I do mention either the link or reference of the article(s) in my blog and hope those interested can read for details. I am briefly summarising the remarks or conclusions of the authors (researchers). If one believe that their intellectual property right /copyright is infringed by any content on this blog, please contact or leave message at below email address amcrasto@gmail.com. It will be removed ASAP
////////EMA APPROVALS, USFDA Approvals, ACALABRUTINIB, AVELUMAB, BETRIXABAN, BRODALUMAB, COPANLISIB, DEFLAZACORT, Delafloxacin, Deutetrabenazine, DUPILUMAB, ETELCALCETIDE, Naldemedine, NETARSUDIL, NIRAPARIB, Ocrelizumab, PLECANATIDE, RIBOCICLIB, SAFINAMIDE, TELOTRISTAT ETHYL, VALBENAZINE, CERLIPONASE, BRIGATINIB, MIDOSTAURIN, Abaloparatide, BENZNIDAZOLE, NERATINIB, inotuzumab ozogamicin, Enasidenib, LETERMOVIR, GLECAPREVIR, PIBRENTASVIR, VOXILAPREVIR, SOFOSBUVIR, EDAVARONE, abemaciclib, ANGIOTENSIN II, VESTRONIDASE, macimorelin acetate, ERTUGLIFLOZIN, SEMAGLUTIDE, EMICIZUMAB, eu 2017, fda 2017, BENRALIZUMAB, DURVALUMAB, GUSELKUMAB, LATANOPROSTENE, OZENOXACIN, SARILUMAB, SECNIDAZOLE, BENRALIZUMAB, TIVOZANIB, SARILUMAB, FLUCICLOVINE,

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
















-Facebook.png)


















































































































































