
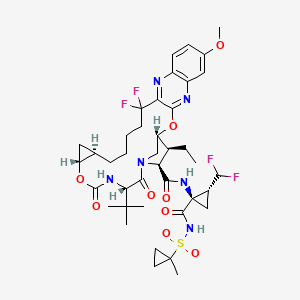
Voxilaprevir
- Molecular FormulaC40H52F4N6O9S
- Average mass868.934 Da
8H-7,10-Methanocyclopropa[18,19][1,10,3,6]dioxadiazacyclononadecino[11,12-b]quinoxaline-8-carboxamide, N-[(1R,2R)-2-(difluoromethyl)-1-[[[(1-methylcyclopropyl)sulfonyl]amino]carbonyl]cyclopropyl]-5-(1 ,1-dimethylethyl)-9-ethyl-18,18-difluoro-1,1a,3,4,5,6,9,10,18,19,20,21,22,22a-tetradecahydro-14-methoxy-3,6-dioxo-, (1aR,5S,8S,9S,10R,22aR)-
Voxilaprevir is a hepatitis C virus (HCV) nonstructural (NS) protein 3/4A protease inhibitor that is used in combination with sofosbuvirand velpatasvir. The combination has the trade name Vosevi and has received a positive opinion from the European Committee for Medicinal Products for Human Use in June 2017.[1]
In July 18, 2017, Vosevi was approved by Food and drug administration.[2]
The hepatitis C virus (HCV), a member of the hepacivirus genera within the Flaviviridae family, is the leading cause of chronic liver disease worldwide (Boyer, N. et al. J Hepatol. 2000, 32, 98-1 12). Consequently, a significant focus of current antiviral research is directed toward the development of improved methods for the treatment of chronic HCV infections in humans (Ciesek, S., von Hahn T., and Manns, MP., Clin. Liver Dis., 201 1 , 15, 597-609; Soriano, V. et al, J. Antimicrob. Chemother., 201 1 , 66, 1573-1686; Brody, H., Nature Outlook, 201 1 , 474, S1 -S7; Gordon, C. P., et al., J. Med. Chem. 2005, 48, 1 -20;
Maradpour, D., et al., Nat. Rev. Micro. 2007, 5, 453-463).
Virologic cures of patients with chronic HCV infection are difficult to achieve because of the prodigious amount of daily virus production in chronically infected patients and the high spontaneous mutability of HCV (Neumann, et al., Science 1998, 282, 103-7; Fukimoto, et al., Hepatology, 1996, 24, 1351 -4;
Domingo, et al., Gene 1985, 40, 1 -8; Martell, et al., J. Virol. 1992, 66, 3225-9). HCV treatment is further complicated by the fact that HCV is genetically diverse and expressed as several different genotypes and numerous subtypes. For example, HCV is currently classified into six major genotypes (designated 1 -6), many subtypes (designated a, b, c, and so on), and about 100 different strains (numbered 1 , 2, 3, and so on).
HCV is distributed worldwide with genotypes 1 , 2, and 3 predominate within the United States, Europe, Australia, and East Asia (Japan, Taiwan, Thailand, and China). Genotype 4 is largely found in the Middle East, Egypt and central Africa while genotype 5 and 6 are found predominantly in South Africa and South East Asia respectively (Simmonds, P. et al. J Virol. 84: 4597-4610, 2010).
The combination of ribavirin, a nucleoside analog, and interferon-alpha (a) (IFN), is utilized for the treatment of multiple genotypes of chronic HCV infections in humans. However, the variable clinical response observed within patients and the toxicity of this regimen have limited its usefulness. Addition of a HCV protease inhibitor (telaprevir or boceprevir) to the ribavirin and IFN regimen improves 12-week post-treatment virological response (SVR12) rates
substantially. However, the regimen is currently only approved for genotype 1 patients and toxicity and other side effects remain.
The use of directing acting antivirals to treat multiple genotypes of HCV infection has proven challenging due to the variable activity of antivirals against the different genotypes. HCV protease inhibitors frequently have compromised in vitro activity against HCV genotypes 2 and 3 compared to genotype 1 (See, e.g., Table 1 of Summa, V. et al., Antimicrobial Agents and Chemotherapy, 2012, 56, 4161 -4167; Gottwein, J. et al, Gastroenterology, 201 1 , 141 , 1067-1079).
Correspondingly, clinical efficacy has also proven highly variable across HCV genotypes. For example, therapies that are highly effective against HCV genotype 1 and 2 may have limited or no clinical efficacy against genotype 3.
(Moreno, C. et al., Poster 895, 61 st AASLD Meeting, Boston, MA, USA, Oct. 29 – Nov. 2, 2010; Graham, F., et al, Gastroenterology, 201 1 , 141 , 881 -889; Foster, G.R. et al., EASL 45th Annual Meeting, April 14-18, 2010, Vienna, Austria.) In some cases, antiviral agents have good clinical efficacy against genotype 1 , but lower and more variable against genotypes 2 and 3. (Reiser, M. et al.,
Hepatology, 2005, 41 ,832-835.) To overcome the reduced efficacy in genotype 3 patients, substantially higher doses of antiviral agents may be required to achieve substantial viral load reductions (Fraser, IP et al., Abstract #48, HEP DART 201 1 , Koloa, HI, December 201 1 .)
Antiviral agents that are less susceptible to viral resistance are also needed. For example, resistance mutations at positions 155 and 168 in the HCV protease frequently cause a substantial decrease in antiviral efficacy of HCV protease inhibitors (Mani, N. Ann Forum Collab HIV Res., 2012, 14, 1 -8;
Romano, KP et al, PNAS, 2010, 107, 20986-20991 ; Lenz O, Antimicrobial agents and chemotherapy, 2010, 54,1878-1887.)
In view of the limitations of current HCV therapy, there is a need to develop more effective anti-HCV therapies. It would also be useful to provide therapies that are effective against multiple HCV genotypes and subtypes.


Kyla Ramey (Bjornson)
Senior CTM Associate at Gilead Sciences
……………………………………………………………………………….


PATENT
WO 2014008285
https://www.google.com/patents/WO2014008285A1?cl=en
RELATIVE SIMILAR EXAMPLE WITHOUT DIFLUORO GROUPS, BUT NOT SAME COMPD
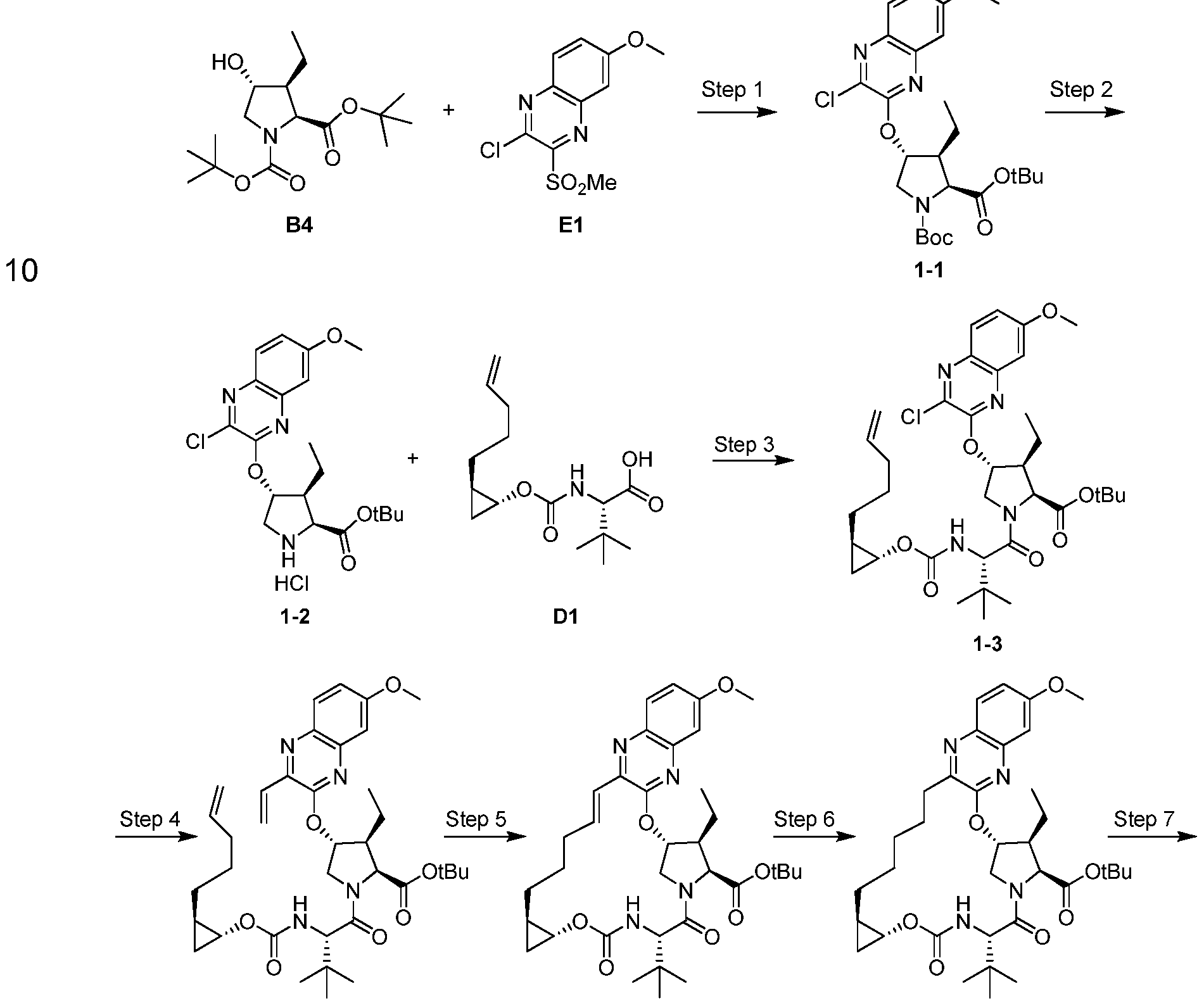
Example 1. Preparation of (1 aR,5S,8S,9S,10R,22aR)-5-tert-butyl-N- [(1 R,2R)-2-(difluoromethyl)-1 -{[(1 – methylcyclopropyl)sulfonyl]carbamoyl}cyclopropyl]-9-ethyl-14-methoxy-3,6-dioxo- 1 ,1 a,3,4,5,6,9,10,18,19,20,21 ,22,22a-tetradecahydro-8H-7,10- methanocyclopropa[18,19][1 ,10,3,6]dioxadiazacyclononadecino[1 1 ,12- b]quinoxaline-8-carboxamide.
Step 1 . Preparation of 1-1 : A mixture containing Intermediate B4 (2.03 g, 6.44 mmol), Intermediate E1 (1 .6 g, 5.85 mmol), and cesium carbonate (3.15 g, 9.66 mmol) in MeCN (40 mL) was stirred vigorously at rt under an atmosphere of Ar for 16 h. The reaction was then filtered through a pad of Celite and the filtrate concentrated in vacuo. The crude material was purified by silica gel
chromatography to provide 1-1 as a white solid (2.5 g). LCMS-ESI+ (m/z): [M- Boc+2H]+ calcd for C2oH27CIN3O4: 408.9; found: 408.6.
Step 2. Preparation of 1-2: To a solution 1 -1 (2.5 g, 4.92 mmol) in dioxane
(10 mL) was added hydrochloric acid in dioxane (4 M, 25 mL, 98.4 mmol) and the reaction stirred at rt for 5 h. The crude reaction was concentrated in vacuo to give 1-2 as a white solid (2.49 g) that was used in subsequently without further purification. LCMS-ESI+ (m/z): [M]+ calcd for C2oH26CIN3O4: 407.9; found: 407.9.
Step 3. Preparation of 1-3: To a DMF (35 mL) solution of 1-2 (2.49 g, 5.61 mmol), Intermediate D1 (1 .75 mg, 6.17 mmol) and DIPEA (3.9 mL, 22.44 mmol) was added COMU (3.12 g, 7.29 mmol) and the reaction was stirred at rt for 3 h. The reaction was quenched with 5% aqueous citric acid solution and extracted with EtOAc, washed subsequently with brine, dried over anhydrous MgSO , filtered and concentrated to produce 1 -3 as an orange foam (2.31 g) that was used without further purification. LCMS-ESI+ (m/z): [M]+ calcd for C35H49CIN4O7: 673.3; found: 673.7.
Step 4. Preparation of 1-4: To a solution of 1-3 (2.31 g, 3.43 mmol), TEA (0.72 mL, 5.15 mmol) and potassium vinyltrifluoroborate (0.69 mg, 5.15 mmol) in EtOH (35 mL) was added PdCI2(dppf) (0.25 g, 0.34 mmol, Frontier Scientific). The reaction was sparged with Argon for 15 min and heated to 80 °C for 2 h. The reaction was adsorbed directly onto silica gel and purified using silica gel chromatography to give 1 -4 as a yellow oil (1 .95 g). LCMS-ESI+ (m/z): [M+H]+ calcd for C37H53N4O7: 665.4; found: 665.3.
Step 5. Preparation of 1 -5: To a solution of 1 -4 (1 .95 g, 2.93 mmol) in
DCE (585 ml_) was added Zhan 1 B catalyst (0.215 g, 0.29 mmol, Strem) and the reaction was sparged with Ar for 15 min. The reaction was heated to 80 °C for 1 .5 h, allowed to cool to rt and concentrated. The crude product was purified by silica gel chromatography to produce 1 -5 as a yellow oil (1 .47 g; LCMS-ESI+ (m/z): [M+H]+ calcd for C35H49N4O7: 637.4; found: 637.3).
Step 6. Preparation of 1 -6: A solution of 1 -5 (0.97 g, 1 .52 mmol) in EtOH (15 ml_) was treated with Pd/C (10 wt % Pd, 0.162 g). The atmosphere was replaced with hydrogen and stirred at rt for 2 h. The reaction was filtered through Celite, the pad washed with EtOAc and concentrated to give 1 -6 as a brown foamy solid (0.803 g) that was used subsequently without further purification. LCMS-ESr (m/z): [M+H]+ calcd for C35H5i N4O7: 639.4; found: 639.3.
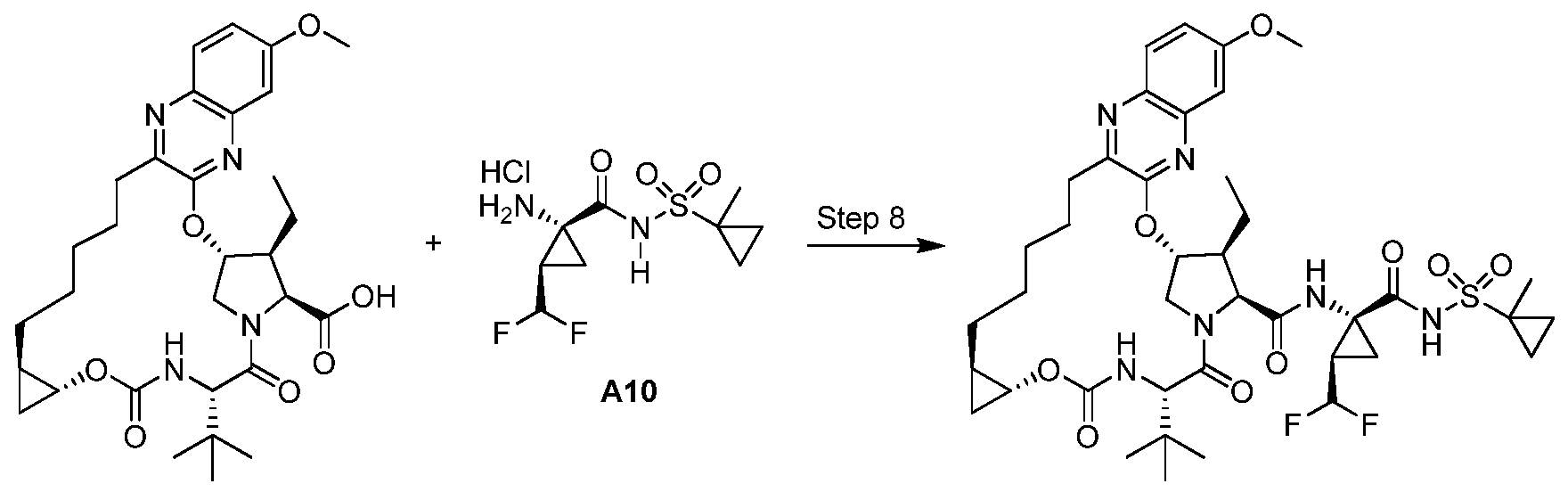
Step 7. Preparation of 1 -7: To a solution of 1 -6 (0.803 g, 1 .26 mmol) in DCM (10 ml_) was added TFA (5 ml_) and stirred at rt for 3 h. An additional 2 ml_ TFA was added and the reaction stirred for another 1 .5 h. The reaction was concentrated to a brown oil that was taken up in EtOAc (35 ml_). The organic solution was washed with water. After separation of the layers, sat. aqueous NaHCO3 was added with stirring until the aqueous layer reached a pH ~ 7-8. The layers were separated again and the aqueous extracted with EtOAc twice. The combined organics were washed with 1 M aqueous citric acid, brine, dried over anhydrous MgSO4, filtered and concentrated to produce 1 -6 as a brown foamy solid (0.719 g) that was used subsequently without further purification. LCMS-ESr (m/z): [M+H]+ calcd for C3i H43N4O7: 583.3; found: 583.4 .
Step 8. Preparation of Example 1 : To a solution of 1 -7 (0.200 g, 0.343 mmol), Intermediate A10 (0.157 g, 0.515 mmol), DMAP (0.063 g, 0.51 mmol) and DIPEA (0.3 ml_, 1 .72 mmol) in DMF (3 ml_) was added HATU (0.235 g, 0.617 mmol) and the reaction was stirred at rt o/n. The reaction was diluted with MeCN and purified directly by reverse phase HPLC (Gemini, 30-100% MeCN/H2O + 0.1 % TFA) and lyophilized to give Example 1 (1 18.6 mg) as a solid TFA salt. Analytic HPLC RetTime: 8.63 min. LCMS-ESI+ (m/z): [M+H]+ calcd for
C40H55F2N6O9S: 833.4; found: 833.5. 1H NMR (400 MHz, CD3OD) δ 9.19 (s, 1 H); 7.80 (d, J = 8.8 Hz, 1 H); 7.23 (dd, J = 8.8, 2.4 Hz, 1 H); 7.15 (d, J = 2.4 Hz, 1 H); 5.89 (d, J = 3.6 Hz, 1 H); 5.83 (td, JH-F = 55.6 Hz, J = 6.4 Hz, 1 H); 4.56 (d, J = 7.2 Hz, 1 H); 4.40 (s, 1 H) 4.38 (ap d, J = 7.2 Hz, 1 H); 4.16 (dd, J = 12, 4 Hz, 1 H); 3.93 (s, 3H); 3.75 (dt, J = 7.2, 4 Hz, 1 H); 3.00-2.91 (m, 1 H); 2.81 (td, J = 12, 4.4 Hz, 1 H); 2.63-2.54 (m, 1 H); 2.01 (br s, 2H); 1 .88-1 .64 (m, 3H); 1 .66-1 .33 (m, 1 1 H) 1 .52 (s, 3H); 1 .24 (t, J = 7.2 Hz, 3H); 1 .10 (s, 9H); 1 .02-0.96 (m, 2H); 0.96- 0.88 (m, 2H); 0.78-0.68 (m, 1 H); 0.55-0.46 (m, 1 H).
PATENT
US 20150175625
PATENT
US 20150175626
The hepatitis C virus (HCV), a member of the hepacivirus genera within the Flaviviridae family, is the leading cause of chronic liver disease worldwide (Boyer, N. et al. J Hepatol. 2000, 32, 98-112). Consequently, a significant focus of current antiviral research is directed toward the development of improved methods for the treatment of chronic HCV infections in humans (Ciesek, S., von Hahn T., and Manns, MP., Clin. Liver Dis., 2011, 15, 597-609; Soriano, V. et al, J. Antimicrob. Chemother., 2011, 66, 1573-1686; Brody, H., Nature Outlook, 2011, 474, S1-S7; Gordon, C. P., et al, J. Med. Chem. 2005, 48, 1-20; Maradpour, D., et al, Nat. Rev. Micro. 2007, 5, 453-463).
Virologic cures of patients with chronic HCV infection are difficult to achieve because of the prodigious amount of daily virus production in chronically infected patients and the high spontaneous mutability of HCV (Neumann, et al, Science 1998, 282, 103-7; Fukimoto, et al, Hepatology, 1996, 24, 1351-4; Domingo, et al, Gene 1985, 40, 1-8; Martell, et al, J. Virol. 1992, 66, 3225-9). HCV treatment is further complicated by the fact that HCV is genetically diverse and expressed as several different genotypes and numerous subtypes. For example, HCV is currently classified into six major genotypes (designated 1-6), many subtypes (designated a, b, c, and so on), and about 100 different strains (numbered 1, 2, 3, and so on).
HCV is distributed worldwide with genotypes 1, 2, and 3 predominate within the United States, Europe, Australia, and East Asia (Japan, Taiwan, Thailand, and China). Genotype 4 is largely found in the Middle East, Egypt and central Africa while genotype 5 and 6 are found predominantly in South Africa and South East Asia respectively (Simmonds, P. et al. J Virol. 84: [0006] There remains a need to develop effective treatments for HCV infections. Suitable compounds for the treatment of HCV infections are disclosed in U.S. Publication No. 2014-0017198, titled “Inhibitors of Hepatitis C Virus” filed on July 2, 2013 including the compound of formula I:

Example 1. Synthesis of (laR,5S,8S,9S,10R,22aR)-5-teri-butyl- V-[(lR,2R)-2-(difluoromethyl)- 1-{ [(1-methylcyclopr opyl)sulfonyl] carbamoyl} cyclopropyl] -9-ethyl- 18,18- difluoro-14-methoxy-3,6-dioxo-l,la,3,4,5,6,9,10,18,19,20,21,22,22a-tetradecahydro-8H-7,10-methanocyclopropa[18,19] [1,10,3,6] dioxadiazacyclononadecino[ll,12-6]quinoxaline-8- carboxamide (I) by route I
[0195] Compound of formula I was synthesized via route I as shown below:

Synthesis of intermediates for compound of formula I SEE PATENT
US 20150175626

References
- “Summary of opinion: Vosevi” (PDF). European Medicines Agency. 22 June 2017.
- FDA approves Vosevi for Hepatitis C
 |
|
| Clinical data | |
|---|---|
| Trade names | Vosevi (combination with sofosbuvir and velpatasvir) |
| Identifiers | |
| CAS Number | |
| PubChemCID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C40H52F4N6O9S |
| Molar mass | 868.94 g·mol−1 |
FDA approves Vosevi for Hepatitis C
The U.S. Food and Drug Administration today approved Vosevi to treat adults with chronic hepatitis C virus (HCV) genotypes 1-6 without cirrhosis (liver disease) or with mild cirrhosis. Vosevi is a fixed-dose, combination tablet containing two previously approved drugs – sofosbuvir and velpatasvir – and a new drug, voxilaprevir. Vosevi is the first treatment approved for patients who have been previously treated with the direct-acting antiviral drug sofosbuvir or other drugs for HCV that inhibit a protein called NS5A.
“Direct-acting antiviral drugs prevent the virus from multiplying and often cure HCV. Vosevi provides a treatment option for some patients who were not successfully treated with other HCV drugs in the past,” said Edward Cox, M.D., director of the Office of Antimicrobial Products in the FDA’s Center for Drug Evaluation and Research.
Hepatitis C is a viral disease that causes inflammation of the liver that can lead to diminished liver function or liver failure. According to the Centers for Disease Control and Prevention, an estimated 2.7 to 3.9 million people in the United States have chronic HCV. Some patients who suffer from chronic HCV infection over many years may have jaundice (yellowish eyes or skin) and develop complications, such as bleeding, fluid accumulation in the abdomen, infections, liver cancer and death.
There are at least six distinct HCV genotypes, or strains, which are genetically distinct groups of the virus. Knowing the strain of the virus can help inform treatment recommendations. Approximately 75 percent of Americans with HCV have genotype 1; 20-25 percent have genotypes 2 or 3; and a small number of patients are infected with genotypes 4, 5 or 6.
The safety and efficacy of Vosevi was evaluated in two Phase 3 clinical trials that enrolled approximately 750 adults without cirrhosis or with mild cirrhosis.
The first trial compared 12 weeks of Vosevi treatment with placebo in adults with genotype 1 who had previously failed treatment with an NS5A inhibitor drug. Patients with genotypes 2, 3, 4, 5 or 6 all received Vosevi.
The second trial compared 12 weeks of Vosevi with the previously approved drugs sofosbuvir and velpatasvir in adults with genotypes 1, 2 or 3 who had previously failed treatment with sofosbuvir but not an NS5A inhibitor drug.
Results of both trials demonstrated that 96-97 percent of patients who received Vosevi had no virus detected in the blood 12 weeks after finishing treatment, suggesting that patients’ infection had been cured.
Treatment recommendations for Vosevi are different depending on viral genotype and prior treatment history.
The most common adverse reactions in patients taking Vosevi were headache, fatigue, diarrhea and nausea.
Vosevi is contraindicated in patients taking the drug rifampin.
Hepatitis B virus (HBV) reactivation has been reported in HCV/HBV coinfected adult patients who were undergoing or had completed treatment with HCV direct-acting antivirals, and who were not receiving HBV antiviral therapy. HBV reactivation in patients treated with direct-acting antiviral medicines can result in serious liver problems or death in some patients. Health care professionals should screen all patients for evidence of current or prior HBV infection before starting treatment with Vosevi.
The FDA granted this application Priority Review and Breakthrough Therapydesignations.
The FDA granted approval of Vosevi to Gilead Sciences Inc
//////////Voxilaprevir, فوكسيلابريفير , 伏西瑞韦 , Воксилапревир , fda 2017, GS 9857, gilead, 1535212-07-7

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....



[…] VOXILAPREVIR […]
LikeLike