Valbenazine
- Molecular FormulaC24H38N2O4
- Average mass418.569 Da
- Chemistry Review(s) (PDF)
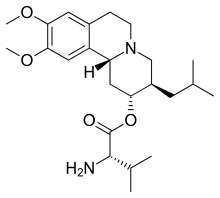
(2R,3R,11bR)-9,10-Dimethoxy-3-(2-methylpropyl)-1,3,4,6,7,11b-hexahydro-2H-benzo(a)quinolizin-2-yl L-valinate bis(4-methylbenzenesulfonate)
and
Valbenazine dihydrochloride
1639208-51-7
April 11, 2017
Release
The U.S. Food and Drug Administration today approved Ingrezza (valbenazine) capsules to treat adults with tardive dyskinesia. This is the first drug approved by the FDA for this condition.
Tardive dyskinesia is a neurological disorder characterized by repetitive involuntary movements, usually of the jaw, lips and tongue, such as grimacing, sticking out the tongue and smacking the lips. Some affected people also experience involuntary movement of the extremities or difficulty breathing.
“Tardive dyskinesia can be disabling and can further stigmatize patients with mental illness,” said Mitchell Mathis, M.D., director of the Division of Psychiatry Products in the FDA’s Center for Drug Evaluation and Research. “Approving the first drug for the treatment of tardive dyskinesia is an important advance for patients suffering with this condition.”
Tardive dyskinesia is a serious side effect sometimes seen in patients who have been treated with antipsychotic medications, especially the older medications, for long periods to treat chronic conditions, such as schizophrenia and bipolar disorder. Tardive dyskinesia can also occur in patients taking antipsychotic medications for depression and certain medications for gastrointestinal disorders and other conditions. It is unclear why some people who take these medications develop tardive dyskinesia yet others do not.
The efficacy of Ingrezza was shown in a clinical trial of 234 participants that compared Ingrezza to placebo. After six weeks, participants who received Ingrezza had improvement in the severity of abnormal involuntary movements compared to those who received placebo.
Ingrezza may cause serious side effects including sleepiness and heart rhythm problems (QT prolongation). Its use should be avoided in patients with congenital long QT syndrome or with abnormal heartbeats associated with a prolonged QT interval. Those taking Ingrezza should not drive or operate heavy machinery or do other dangerous activities until it is known how the drug affects them.
The FDA granted this application Fast Track, Priority Review and Breakthrough Therapy designations.
The FDA granted approval of Ingrezza to Neurocrine Biosciences, Inc.
Valbenazine (INN,[1]:114 proposed trade name Ingrezza) is the first drug approved by the FDA[2] for use in the treatment of tardive dyskinesia.[3][4] Clinical trials are underway to evaluate its efficacy in the treatment of Tourette’s syndrome.[5][6] It acts as a vesicular monoamine transporter 2 (VMAT2) inhibitor.[7]
Pharmacology
Mechanism of action
Valbenazine is known to cause reversible reduction of dopamine release by selectively inhibiting pre-synaptic human vesicular monoamine transporter type 2 (VMAT2). In vitro, valbenazine shows great selectivity for VMAT2 and little to no affinity for VMAT1 or other monoamine receptors.[8] Although the exact cause of tardive dyskinsia is unknown, it is hypothesized that it may result from neuroleptic-induced dopamine hypersensitivity.[9] By selectively reducing the ability of VMAT2 to load dopamine into synaptic vesicles,[10] the drug reduces overall levels of available dopamine in the synaptic cleft, ideally alleviating the symptoms associated with dopamine hypersensitivity. The importance of valbenazine selectivity inhibiting VMAT2 over other monoamine transporters is that VMAT2 is mainly involved with the transport of dopamine, and to a much lesser extent other monoamines such as norepinephrine, serotonin, and histamine. This selectivity is likely to reduce the likelihood of “off-target” adverse effects which may result from the upstream inhibition of these other monoamines.[11]
Society and culture
Commercial aspects
Valbenazine is produced by Neurocrine Biosciences, a company based in San Diego. In addition to the late-stage clinical trials studying valbenazine, Neurocrine Biosciences (partnered with AbbVie Inc.) also has another product, elagolix (a hormone antagonist), undergoing clinical trials.[12] Following the initiation of these trials, on 5 May 2016 Neurocrine reported revenues of $15 million for the first quarter of 2016.[13] The company now focuses on filing the valbenazine new drug application as they prepare for the commercial launch of the drug for the treatment of tardive dyskinesia.Neurocrine’s expenses have risen steadily since May 2015, primarily due to the pre-commercialization activities for valbenazine. [14]
Intellectual property
While Neurocrine Biosciences does not currently hold a final patent for valbenazine or elagolix, they do hold a patent for the VMAT2 inhibitor [9,10-dimethoxy-3-(2-methylpropyl)-1H,2H,3H,4H,6H,7H,11bH-pyrido-[2,1-a]isoquinolin-2-yl]methanol and related compounds, which includes valbenazine.[15]
References
- “International Nonproprietary Names for Pharmaceutical Substances (INN). Recommended International Nonproprietary Names: List 71” (PDF). World Health Organization. Retrieved 18 November 2016.
- Newswire, MultiVu – PR. “Neurocrine Announces FDA Approval of INGREZZA TM (valbenazine) Capsules as the First and Only Approved Treatment for Adults with Tardive Dyskinesia (TD)”. Multivu. Retrieved 2017-04-11.
- Ben Adams (Aug 30, 2016). “Neurocrine submits valbenazine NDA early, set for 2017 approval”. fiercebiotech.com.
- “Safety and Tolerability Study of NBI-98854 for the Treatment of Tardive Dyskinesia – Full Text View – ClinicalTrials.gov”. clinicaltrials.gov. Retrieved 2016-11-13.
- Jump up^ “Tourette Syndrome Clinical Trials | Neurocrine Biosciences”. http://www.neurocrine.com. Retrieved 2016-11-13.
- Jump up^ “Safety and Efficacy Study of NBI-98854 in Adults With Tourette Syndrome – Full Text View – ClinicalTrials.gov”. clinicaltrials.gov. Retrieved 2016-11-13.
- Jump up^ O’Brien, C. F.; Jimenez, R; Hauser, R. A.; Factor, S. A.; Burke, J; Mandri, D; Castro-Gayol, J. C. (2015). “NBI-98854, a selective monoamine transport inhibitor for the treatment of tardive dyskinesia: A randomized, double-blind, placebo-controlled study”. Movement Disorders. 30 (12): 1681–7. doi:10.1002/mds.26330. PMC 5049616
 . PMID 26346941.
. PMID 26346941. - Jump up^ “NBI-98854 – VMAT2 Inhibitor | Tics in Children Treatment | Neurocrine Biosciences”. http://www.neurocrine.com. Retrieved 2016-11-13.
- Jump up^ “tardive-dyskinesia”. http://www.priory.com. Retrieved 2016-11-13.
- Jump up^ Purves, Dale, et al. Neuroscience. Sinauer Associates. 087893646
- “NBIX: NDA for Valbenazine in Tardive Dyskinesia to be Filed in 2016…”. Retrieved 2016-11-13.
- “Endocrine & Movement Disorder R&D | About | Neurocrine Biosciences”. http://www.neurocrine.com. Retrieved 2016-11-14.
- “NBIX: NDA for Valbenazine in Tardive Dyskinesia to be Filed in 2016…”. Retrieved 2016-11-20.
- “Press Release | Neurocrine Biosciences, Inc.”. phoenix.corporate-ir.net. Retrieved 2016-11-20.
- “[9,10-dimethoxy-3-(2-methylpropyl)-1h,2h,3h,4h,6h,7h,11bh-pyrido-[2,1-a]isoquinolin-2-yl]methanol And Compounds, Compositions And Methods Relating Thereto”. Retrieved 2016-11-20.
  |
|
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| Synonyms | NBI-98854 |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C24H38N2O4 |
| Molar mass | 418.58 g·mol−1 |
| 3D model (Jmol) | |

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....

[…] VALBENAZINE […]
LikeLike