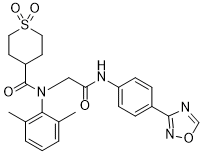
Amenamevir アメナメビル
M-5220
CAS 841301-32-4
Chemical Formula: C24H26N4O5S
Molecular Weight: 482.555
N-(2-((4-(1,2,4-oxadiazol-3-yl)phenyl)amino)-2-oxoethyl)-N-(2,6-dimethylphenyl)tetrahydro-2H-thiopyran-4-carboxamide 1,1-dioxide
PMDA

| 2017/7/3 | PMDA | APPROVED | JAPAN | Amenamevir | Amenalief
BRAND |
Maruho
COMPANY |

Amenamevir, also known as ASP2151, is a herpes virus helicase-primase inhibitor. ASP2151 had significantly better anti-HSV activity against herpes simplex keratitis than valacyclovir and acyclovir after systemic or topical use.
| アメナメビル Amenamevir  C24H26N4O5S : 482.55 [841301-32-4] |
Amenamevir is an oral helicase-primase inhibitor launched in 2017 in Japan for the treatment of herpes zoster (shingles). The product is being marketed by Maruho.
Amenamevir had been in phase III clinical trials for herpes simplex virus;
In August 2012, Astellas Pharma granted Maruho development and commercialization rights in Japan.
US 20050032855
WO 2006082822
WO 2006082820
WO 2006082821
WO 2009123169
WO 2010047295
JP 2006241144
Patent
The publicly known crystal (following and alpha type crystal) of the compound A of disclosure to the aforementioned Patent document 2 is obtained by re-crystallizing from an ethanol water mixed solvent, and has the melting point of about 220 to 222 degree C. The present invention relates to multi-form crystals other than the alpha form crystal concerned, and relates to beta, gamma, delta, and epsilon type crystal specifically. In a surprising thing, each of these multi-form crystals is crystals stable to a degree usable as a medicinal manufacture field object, and has a preferable property in the surface of solubility, absorbency, stability, and/or a handling property
PATENT
US20050032855, EP1844776A1.

REFERENCES
1: Ohtsu Y, Otsuka S, Nakamura T, Noguchi K. Regulated bioanalysis of conformers – A case study with ASP2151 in dog plasma and urine. J Chromatogr B Analyt Technol Biomed Life Sci. 2015 Aug 1;997:56-63. doi: 10.1016/j.jchromb.2015.05.028. Epub 2015 May 28. PubMed PMID: 26093120.
2: James SH, Larson KB, Acosta EP, Prichard MN. Helicase-primase as a target of new therapies for herpes simplex virus infections. Clin Pharmacol Ther. 2015 Jan;97(1):66-78. doi: 10.1002/cpt.3. Epub 2014 Nov 18. Review. PubMed PMID: 25670384.
3: Muylaert I, Zhao Z, Elias P. UL52 primase interactions in the herpes simplex virus 1 helicase-primase are affected by antiviral compounds and mutations causing drug resistance. J Biol Chem. 2014 Nov 21;289(47):32583-92. doi: 10.1074/jbc.M114.609453. Epub 2014 Oct 2. PubMed PMID: 25278021; PubMed Central PMCID: PMC4239612.
4: Biswas S, Sukla S, Field HJ. Helicase-primase inhibitors for herpes simplex virus: looking to the future of non-nucleoside inhibitors for treating herpes virus infections. Future Med Chem. 2014 Jan;6(1):45-55. doi: 10.4155/fmc.13.192. Review. PubMed PMID: 24358947.
5: Andrei G, Snoeck R. Advances in the treatment of varicella-zoster virus infections. Adv Pharmacol. 2013;67:107-68. doi: 10.1016/B978-0-12-405880-4.00004-4. Review. PubMed PMID: 23886000.
6: Sasaki S, Miyazaki D, Haruki T, Yamamoto Y, Kandori M, Yakura K, Suzuki H, Inoue Y. Efficacy of herpes virus helicase-primase inhibitor, ASP2151, for treating herpes simplex keratitis in mouse model. Br J Ophthalmol. 2013 Apr;97(4):498-503. doi: 10.1136/bjophthalmol-2012-302062. Epub 2013 Jan 29. PubMed PMID: 23361434.
7: Katsumata K, Chono K, Kato K, Ohtsu Y, Takakura S, Kontani T, Suzuki H. Pharmacokinetics and pharmacodynamics of ASP2151, a helicase-primase inhibitor, in a murine model of herpes simplex virus infection. Antimicrob Agents Chemother. 2013 Mar;57(3):1339-46. doi: 10.1128/AAC.01803-12. Epub 2012 Dec 28. PubMed PMID: 23274658; PubMed Central PMCID: PMC3591930.
8: Chono K, Katsumata K, Suzuki H, Shiraki K. Synergistic activity of amenamevir (ASP2151) with nucleoside analogs against herpes simplex virus types 1 and 2 and varicella-zoster virus. Antiviral Res. 2013 Feb;97(2):154-60. doi: 10.1016/j.antiviral.2012.12.006. Epub 2012 Dec 20. PubMed PMID: 23261844.
9: Chono K, Katsumata K, Kontani T, Shiraki K, Suzuki H. Characterization of virus strains resistant to the herpes virus helicase-primase inhibitor ASP2151 (Amenamevir). Biochem Pharmacol. 2012 Aug 15;84(4):459-67. doi: 10.1016/j.bcp.2012.05.020. Epub 2012 Jun 9. PubMed PMID: 22687623.
10: Katsumata K, Weinberg A, Chono K, Takakura S, Kontani T, Suzuki H. Susceptibility of herpes simplex virus isolated from genital herpes lesions to ASP2151, a novel helicase-primase inhibitor. Antimicrob Agents Chemother. 2012 Jul;56(7):3587-91. doi: 10.1128/AAC.00133-12. Epub 2012 Apr 23. PubMed PMID: 22526302; PubMed Central PMCID: PMC3393391.
11: Tyring S, Wald A, Zadeikis N, Dhadda S, Takenouchi K, Rorig R. ASP2151 for the treatment of genital herpes: a randomized, double-blind, placebo- and valacyclovir-controlled, dose-finding study. J Infect Dis. 2012 Apr 1;205(7):1100-10. doi: 10.1093/infdis/jis019. Epub 2012 Feb 20. PubMed PMID: 22351940.
12: Himaki T, Masui Y, Chono K, Daikoku T, Takemoto M, Haixia B, Okuda T, Suzuki H, Shiraki K. Efficacy of ASP2151, a helicase-primase inhibitor, against thymidine kinase-deficient herpes simplex virus type 2 infection in vitro and in vivo. Antiviral Res. 2012 Feb;93(2):301-4. doi: 10.1016/j.antiviral.2011.11.015. Epub 2011 Dec 4. PubMed PMID: 22155691.
13: Katsumata K, Chono K, Sudo K, Shimizu Y, Kontani T, Suzuki H. Effect of ASP2151, a herpesvirus helicase-primase inhibitor, in a guinea pig model of genital herpes. Molecules. 2011 Aug 25;16(9):7210-23. doi: 10.3390/molecules16097210. PubMed PMID: 21869749.
14: Andrei G, Snoeck R. Emerging drugs for varicella-zoster virus infections. Expert Opin Emerg Drugs. 2011 Sep;16(3):507-35. doi: 10.1517/14728214.2011.591786. Epub 2011 Jun 24. Review. PubMed PMID: 21699441.
15: Chono K, Katsumata K, Kontani T, Kobayashi M, Sudo K, Yokota T, Konno K, Shimizu Y, Suzuki H. ASP2151, a novel helicase-primase inhibitor, possesses antiviral activity against varicella-zoster virus and herpes simplex virus types 1 and 2. J Antimicrob Chemother. 2010 Aug;65(8):1733-41. doi: 10.1093/jac/dkq198. Epub 2010 Jun 9. PubMed PMID: 20534624
///////////Amenamevir, アメナメビル, japan 2017, ASP2151, ASP 2151, M-5220, MARUHO, Amenalief
O=C(C(CC1)CCS1(=O)=O)N(C2=C(C)C=CC=C2C)CC(NC3=CC=C(C4=NOC=N4)C=C3)=O

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....


[…] via Amenamevir アメナメビル — New Drug Approvals […]
LikeLike