Eslicarbazepine acetate, 236395-14-5 cas no
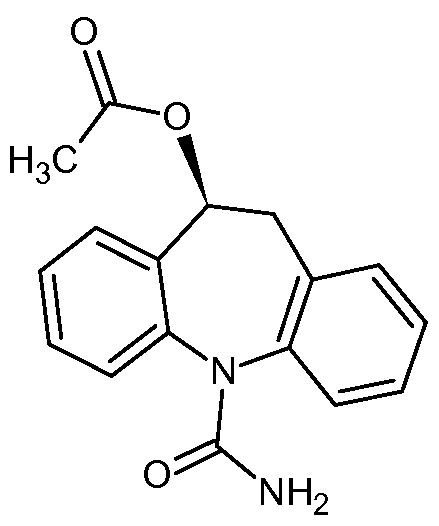
(S)-10-Acetoxy- 10,11-dihydro- 5H-dibenz[b,f]azepine- 5-carboxamide
Sunovion Pharmaceuticals Inc. A US-based subsidiary of Japanese drugmaker Dainippon Sumitomo Pharma Announces FDA Approval of Aptiom® (eslicarbazepine acetate) as Once-Daily Adjunctive Treatment of Partial-Onset Seizures
MONDAY Nov. 11, 2013 — Aptiom (eslicarbazepine acetate) has been approved by the U.S. Food and Drug Administration as an add-on drug to help treat adults with partial epileptic seizures.
Epilepsy, caused by abnormal activity in the brain’s nerve cells, is diagnosed in some 200,000 people annually in the United States, the agency said in a news release. So-called “partial” seizures are the most common type of seizure among people with epilepsy, triggering possible symptoms including repetitive movement of limbs, unusual behavior and convulsions.http://www.drugs.com/news/aptiom-approved-seizures-48845.html
The FDA has determined that APTIOM will not be classified as a controlled substance. Sunovion expects APTIOM to be available in U.S. pharmacies in the second quarter (April – June) of 2014
APTIOM, a voltage-gated sodium channel inhibitor, is a prescription medicine approved for use as adjunctive treatment of partial-onset seizures. Treatment with APTIOM should be initiated at 400 mg once daily. After one week, dosage may be increased to the recommended maintenance dosage of 800 mg once daily. Some patients may benefit from the maximum recommended maintenance dosage of 1,200 mg once daily, although this dosage is associated with an increase in adverse reactions. The maximum dose of 1,200 mg daily should only be initiated after the patient has tolerated 800 mg daily for at least a week. For some patients, treatment may be initiated at 800 mg once daily if the need for additional seizure reduction outweighs an increased risk of adverse reactions during initiation.
The initial research and development of eslicarbazepine acetate was performed by BIAL, a privately held Portuguese research-based pharmaceutical company. Subsequently, Sunovion acquired the rights under an exclusive license to further develop and commercialize eslicarbazepine acetate in the U.S. and Canadian markets from BIAL. In February 2009, Eisai Europe Limited, a European subsidiary of Eisai Co., Ltd. (Eisai), entered into a license and co-promotion agreement with BIAL, which gave the rights to Eisai to sell eslicarbazepine acetate under the trade name Zebinix® in Europe. Zebinix was approved by the European Commission on April 21, 2009 as adjunctive therapy in adult patients with partial-onset seizures with or without secondary generalization and is currently marketed in Europe under the agreement.
Eslicarbazepine acetate (BIA 2-093) is an antiepileptic drug. It is a prodrug which is activated to eslicarbazepine (S–licarbazepine), an active metabolite of oxcarbazepine.[1]
It is being developed by Bial[2] and will be marketed as Zebinix or Exalief by Eisai Co. in Europe and as Stedesa by Sepracor[3] in America.
The European Medicines Agency (EMA) has recommended granting marketing authorization in 2009 for adjunctive therapy for partial-onset seizures, with or without secondary generalisation, in adults with epilepsy.[1] The U.S. Food and Drug Administration (FDA) announced on 2 June 2009 that the drug has been accepted for filing.[3]
Eslicarbazepine acetate is a prodrug for S(+)-licarbazepine, the major active metabolite of oxcarbazepine.[4] Its mechanism of action is therefore identical to that of oxcarbazepine. [5] There may, however, be pharmacokinetic differences. Eslicarbazepine acetate may not produce as high peak levels of (S)-(+)-licarbazepine immediately after dosing as does oxcarbazepine which could theoretically improve tolerability.
Like oxcarbazepine, eslicarbazepine may be used to treat bipolar disorder and trigeminal neuralgia.
The first European patent to protect this drug is EP 0751129 . The priority of this European patent is the Portuguese patent application PT 101732 .
- Dulsat, C., Mealy, N., Castaner, R., Bolos, J. (2009). “Eslicarbazepine acetate”. Drugs of the Future 34 (3): 189. doi:10.1358/dof.2009.034.03.1352675.
- Community register of medicinal products for human use: Exalief
- Medical News Today: Sepracor’s STEDESA (Eslicarbazepine Acetate) New Drug Application Formally Accepted For Review By The FDA
- Rogawski, MA (Jun 2006). “Diverse Mechanisms of Antiepileptic Drugs in the Development Pipeline”. Epilepsy Res 69 (3): 273–294. doi:10.1016/j.eplepsyres.2006.02.004. PMC 1562526. PMID 16621450.
- Rogawski MA, Löscher W (July 2004). “The neurobiology of antiepileptic drugs”. Nature Reviews Neuroscience 5 (7): 553–64. doi:10.1038/nrn1430. PMID 15208697.
- https://newdrugapprovals.wordpress.com/2013/03/11/sunovion-announces-fda-acceptance-for-review-of-new-drug-application-resubmission-for-stedesa-eslicarbazepine-acetate/
Eslicarbazepine acetate of Formula A, chemically known as (10S)-5-carbamoyl- 10,1 l-dihydro-5H-dibenzo[Z?,/]azepin- 10-yl acetate is indicated as adjunctive therapy in adults with partial-onset seizures with or without secondary generalisation.
Formula A
lO-oxo-10,1 l-dihydro-5H-dibenzo[Z?/]azepine-5-carboxamide of Formula 1, commonly known as oxcarbazepine, is an antiepileptic drug marketed under the trade name Trileptal®and is indicated for use as monotherapy or adjunctive therapy in the treatment of partial seizures in adults and as monotherapy in the treatment of partial seizures in children aged 4 years and above with epilepsy, and as adjunctive therapy in children aged 2 years and above with epilepsy. Oxcarbazepine is an intermediate for the preparation of eslicarbazepine.
Formula 1
Several processes are known in the literature for making and purifying eslicarbazepine acetate, for example, U.S. Patent No 5,753,646; and PCT Publications WO 2006/005951 ; WO 2007/1 17166; and WO 2010/1 13179.
U.S. Patent No. 5,753,646 provides a process for the preparation of eslicarbazepine acetate which involves adding dropwise a solution of acetyl chloride in dichloromethane to a suspension of (-)- 10-hydroxy-10,l l-dihydro-5H-dibenz/b,f/azepine-5-carboxamide in dichloromethane and pyridine at a temperature of less than 10°C under stirring. The residue obtained after workup was crystallized from a mixture of dichloromethane and ethyl acetate to give the eslicarbazepine acetate as white crystals.
U.S Publication No. 2009/0203902 provides preparation of eslicarbazepine acetate which involves the acylation of (S)-(+)-10,l l-dihydro-10-hydroxy-5H-dibenz/b,f/azepine- 5-carboxamide with acetic anhydride in presence of 4-(N,N-dimethylamino)pyridine and pyridine in dichloromethane at reflux temperature. The resulting solid obtained after work-up was slurried with isopropanol at reflux to obtain a solution. The solution was cooled to 1°C to 5°C and eslicarbazepine acetate was isolated from the reaction mass by filtration followed by washing with isopropanol.
PCT Publication No. WO 2010/1 13179 provides various purification methods of eslicarbazepine acetate which involve the use of acetonitrile/methyl tertiary butyl ether, tetrahydrofuran/n-hexane, tetrahydrofuran/methyl tertiary butyl methyl ether;
tetrahydrofuran, methyl ethyl ketone/n-hexane.
Several processes are known in the literature for making oxcarbazepine, for example, U.S. Patent Nos. 4,452,738 and 7,459,553; PCT Publication Nos. WO
2010/000196; WO 2008/012837; WO 2007/141798; WO 2006/075925; WO 2005/122671 ; WO 2005/1 18550; WO 2005/096709; WO 2005/092862; WO
2005/066133; WO 02/096881 ; WO 00/55138; and WO 96/21649.
PCT Publication No. WO 02/096881 provides a process for the preparation of oxcarbazepine which involves oxidation of 10,1 1 -dihydro- 10-hydroxy-5H- dibenz/b,f/azepine-5-carboxamide with peroxyacetic acid in presence of potassium dichromate adsorbed on silica gel at room temperature.
Japanese Patent Publication No. JP 2004- 175761 provides a process for the preparation of oxcarbazepine which involves oxidation of 10, 1 1 -dihydro- 10-hydroxy-5H- dibenzo[b,f]azepine-5-carboxamide with dimethyl sulfoxide and an activator such as sulfur trioxide-pyridine complex.
Chinese Publication No. CN 101302198 provides a process for the preparation of oxcarbazepine which involves oxidation of 10-hydroxy- 10, l 1 -dihydro-5H- dibenzo[Z?/]azepine-5-carbonitrile with TEMPO and sodium hypochlorite to provide 10- oxo- 10,1 l-dihydro-5H-dibenzo[Z?/]azepine-5-carbonitrile which was further hydrolysed with sulfuric acid to obtain oxcarbazepine.
-
Eslicarbazepine acetate, (S)-(-)-10-acetoxy-10,11-dihydro-5H-dibenz/b,f/azepine-5-carboxamide (“BIA 2-093”), is a new drug currently being developed which is useful for the treatment of various conditions, such as, for example, epilepsy and affective brain disorders, as well as pain conditions and nervous function alterations in degenerative and post-ischemic diseases. Although chemically related to carbamazepine and oxcarbazepine, eslicarbazepine acetate is believed to avoid the production of certain toxic metabolites (such as, for example, epoxides) and to avoid the unnecessary production of enantiomers or diastereoisomers of metabolites and conjugates, without losing pharmacological activity. See Benes et al., “Anticonvulsant and Sodium Channel-Blocking Properties of Novel 10,11-Dihydro-5H-dibenz[b,f]azepine-5-carboxamide Derivatives,” J. Med. Chem., 42, 2582-2587 (1999).
-
Like carbamazepine and oxcarbazepine, eslicarbazepine acetate is believed to be a voltage-gated sodium channel (VGSC) blocker that competitively interacts with site 2 of the inactivated state of the sodium channel. The affinity for this state of the channel is similar to that of carbamazepine, while the affinity for the resting state of the channel is about 3-fold lower than that of carbamazepine. This profile may suggest an enhanced inhibitory selectivity of eslicarbazepine acetate for rapidly firing neurons over those displaying normal activity. See Bonifacio et al., “Interaction of the Novel Anticonvulsant, BIA 2-093, with Voltage-Gated Sodium Channels: Comparison with Carbamazepine,” Epilepsia, 42, 600-608(2001).
-
Evaluation of the metabolic profile of eslicarbazepine acetate, following chiral analysis, in liver microsomes from rats, dogs, monkeys and humans was found to give the S(+) enantiomer of licarbazepine, (S)-(+)-10,11-dihydro-10-hydroxy-5H dibenz/b,f/azepine-5-carboxamide (also known as “eslicarbazepine”), and not the R(-) form of licarbazepine, (R)-(-)-10,11-dihydro-10-hydroxy-5H dibenz/b,f/azepine-5-carboxamide (also known as “R-licarbazepine”).
-
Studies in humans have shown that, after oral administration, eslicarbazepine acetate appears to be rapidly and extensively metabolized to the active metabolite eslicarbazepine and, in a minor extent, to R-licarbazepine. See Silveira et al., “BIA 2-093 Pharmacokinetics in Healthy Elderly Subjects,” Epilepsia, 45 (suppl. 3), 157 (2004). For example, the plasma concentrations of the parent drug (eslicarbazepine acetate) have been systematically found below the limit of quantification (LOQ) of the assay (10 ng/mL). See Almeida I; Almeida, L. & Soares-da-Silva, P., “Safety, Tolerability and Pharmacokinetic Profile of BIA 2-093, a Novel Putative Antiepileptic Agent, during First Administration to Humans,” Drugs R&D, 4, 269-284 (2003) (herein referred to as “Almeida II“). When a non-chiral method is used, the assay does not distinguish between eslicarbazepine and the R-enantiomer, and the mixture was reported as “BIA 2-005” or “racemic licarbazepine.”
-
The inventors performed entry-into-man studies in healthy subjects, the results of which they described in the Almeida I and Almeida II articles, both of which are hereby incorporated by reference. In these studies, the healthy subjects received a single oral dose of eslicarbazepine acetate wherein the dose ranged from 20 mg to 1200 mg (see Almeida II), and multiple daily-doses of eslicarbazepine acetate ranging from 200 mg twice-daily to 1200 mg once-daily (see Almeida I). Further studies (not yet published) by the inventors have investigated higher doses of eslicarbazepine acetate, including, for example, doses ranging up to 2400 mg once-daily. The studies showed that BIA 2-005 maximum observed plasma concentration (Cmax) was attained at about 1 hour to about 4 hours post-dose (tmax), the extent of systemic exposure to BIA 2-005 was approximately dose-proportional, and steady-state of BIA 2-005 plasma concentrations was attained at about 4 to 5 days. The mean renal clearance of BIA 2-005 from plasma was about 20-30 mL/min, and the total amount of BIA 2-005 recovered in the urine was approximately 20% and 40% within 12 hours and 24 hours post-dose, respectively.
-
The studies also showed that the apparent terminal half-life of BIA 2-005 ranged from about 8 hours to about 17 hours. See, e.g., Almeida II.
-
U.S. Patent No. 6,296,873 discloses a sustained release delivery system for carbamazepine, which has a half-life ranging from 25 hours to 85 hours. To avoid adverse effects, U.S. Patent No. 6,296,873 teaches that the carbamazepine should be administered in tablet form up to two or more times daily to slowly release the compound to maintain concentration levels between 4-12 µg/mL. Such a delivery system requires a form that is capable of delivering the compound over an extended period of time, such as a tablet form.

http://www.sciencedirect.com/science/article/pii/S0040403913005030

ESLICARBAZEPINE ACETATE
Physiochemical Pr operties:
| Molecular weight | : | 296.32 |
| Category | : | Anti-epileptic |
| Molecular formula | : | C17H16N2O5 |
| Chemical Name | : | (S)-(-)-10-acetoxy-10,11-dihydro-5H-dibenz [b, f] |
| azepine-5-carboxamide. | ||
| Description | : | White to off-White, odourless, non-hygroscopic, |
| crystalline powder. | ||
| Solubility | : | Freely soluble in dichloromethane, sparingly soluble |
| in acetone, acetonitrile, methanol, tetrahydrofuran and | ||
| slightly soluble in ethanol and 2-propanol, insoluble in | ||
| water | ||
| Melting Point | : | 184-187°C |
| Storage | : | Can be easily stored at temperatures up to 30°C |
HPLC, NMR

NMR NUMBERING

http://www.sciencedirect.com/science/article/pii/S0731708511006753

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....




Reblogged this on Med.Chem in Nepal.
LikeLike