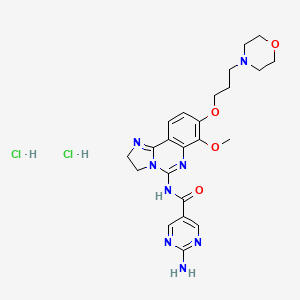
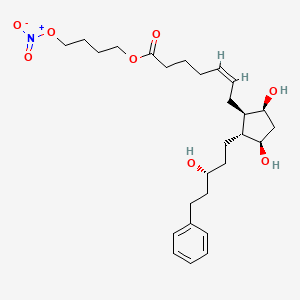
In the literature (Journal of Medicinal Chemistry, 2005,48,5504) in order cytidine as a raw material, first selectively protected 3 ‘, 5′-hydroxyl group, and then oxidizing the 2′-hydroxyl to a carbonyl group, and the reaction of methyllithium get the 2’-hydroxyl compound, and then removing the protective group, use benzoyl protected 3 ‘, 5’-hydroxyl group, and then reacted with DAST fluorinated compound, followed by hydrolysis and aminolysis reaction products, such as the following Reaction Scheme. The method of route length, the need to use expensive silicon ether protecting group molecule relatively poor economy; conducting methylation time will generate a non-methyl enantiomer beta bits.
Home » Posts tagged 'Sofosbuvir'
Tag Archives: Sofosbuvir
SOFOSBUVIR, NEW PATENT, WO 2018032356, Pharmaresources (Shanghai) Co Ltd

SOFOSBUVIR, NEW PATENT, WO 2018032356, Pharmaresources (Shanghai) Co Ltd
WO-2018032356, Pharmaresources (Shanghai) Co Ltd
CHEN, Ping; (CN).
PENG, Shaoping; (CN).
LI, Yinqiang; (CN).
LI, Dafeng; (CN).
DONG, Xuejun; (CN)
Process for the preparation of lactone derivatives and their intermediates are important precursors for the synthesis of anti-hepatitis C virus agents, including sofosbuvir . Represents a first filing from Pharmaresources (Shanghai) Co Ltd and the inventors on this API. Gilead Sciences , following its acquisition of Pharmasset , has developed and launched sofosbuvir, a pure chiral isomer of PSI-7851, a next-generation HCV uracil nucleotide analog polymerase inhibitor prodrug for once-daily oral use.
///////////////////
Novel Drug Approvals for 2017, A Review/Compilation
CDSCO
 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO, Novel Drug Approvals for 2017, A Review Compilation (USFDA, EMA, PMDA, CDSCO).
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO, Novel Drug Approvals for 2017, A Review Compilation (USFDA, EMA, PMDA, CDSCO).
Any errors in this compilation, email amcrasto@gmail.com, Call +919323115463
Some gaps will be filled up soon keep watching……………..
INDEX, NAME (click on the title, it contains link)
SECTION A; USFDA Approvals
6 BENRALIZUMAB
17 DURVALUMAB
24 GUSELKUMAB
36 OZENOXACIN
40 SARILUMAB
41 SECNIDAZOLE
INDEX, FORMULATION NAME
USFDA
•Aliqopa (COPANLISIB) to treat adults with relapsed follicular lymphoma — a slow-growing type of nonHodgkin lymphoma (a cancer of the lymph system) — who have received at least two prior systemic therapies;
• ALUNBRIG, BRIGATINIB, To treat patients with anaplastic lymphoma kinase (ALK)-positive metastatic non-small cell lung cancer (NSCLC) who have progressed on or are intolerant to crizotinib
• Austedo, Deutetrabenazine For the treatment of chorea associated with Huntington’s disease
• Bavencio (avelumab) for the treatment of patients 12 years and older with a rare and aggressive form of cancer called metastatic Merkel cell carcinoma, including those who have not received prior chemotherapy;
•BAXDELLA, Delafloxacin, BACTERIAL INFECTIONS
• Benznidazole to treat children ages 2 to 12 years with Chagas disease, a parasitic infection that can cause serious heart illness after years of infection, and can also affect swallowing and digestion. This is the first treatment approved in the United States for this rare disease;
• Besponsa (inotuzumab ozogamicin) for the treatment of adults with a type of cancer of the blood called relapsed or refractory B-cell precursor acute lymphoblastic leukemia;
•BEVYXXA, BETRIXABAN, For the prophylaxis of venous thromboembolism (VTE) in adult patients hospitalized for an acute medical illness
• BRINEURA, CERLIPONASE ALFA, To treat a specific form of Batten disease
• Calquence (ACALABRUTINIB) to treat adults with mantle cell lymphoma who have received at least one prior therapy. Mantle cell lymphoma is a particularly aggressive cancer;
• DUPIXENT, (DUPILUMAB) To treat adults with moderate-to-severe eczema (atopic dermatitis)
• Emflaza (deflazacort) to treat patients age 5 years and older with Duchenne muscular dystrophy, a rare genetic disorder that causes progressive muscle deterioration and weakness;
• FASENRA, BENRALIZUMAB, For add-on maintenance treatment of patients with severe asthma aged 12 years and older, and with an eosinophilic phenotype
• Giapreza (angiotensin II), for the treatment of hypotension in adults with distributive or vasodilatory shock (dangerously low blood pressure despite adequate heart function) whose blood pressure remains low despite receiving fluids and treatment with drugs called vasopressors;
• HEMLIBRA EMICIZUMAB To prevent or reduce the frequency of bleeding episodes in adult and pediatric patients with hemophilia A who have developed antibodies called Factor VIII (FVIII) inhibitors.
• Idhifa (enasidenib) for the treatment of adults with relapsed or refractory acute myeloid leukemia, a form of blood cancer, who have a specific genetic mutation;
• IMFINZI, DURVALUMAB To treat patients with locally advanced or metastatic urothelial carcinoma
• Ingrezza (valbenazine) to treat adults with tardive dyskinesia, a side effect of some antipsychotic medications whereby patients can experience uncontrollable stiff, jerky movements of their face and body, and other uncontrolled movements such as eye-blinking, sticking out the tongue, and arm-waving;
• KEVZARA SARILUMAB, RHEUMATOID ARTHRITIS
• KISQALI, RIBOCICLIB, To treat postmenopausal women with a type of advanced breast cancer
• Macrilen macimorelin acetate, For the diagnosis of adult growth hormone deficiency
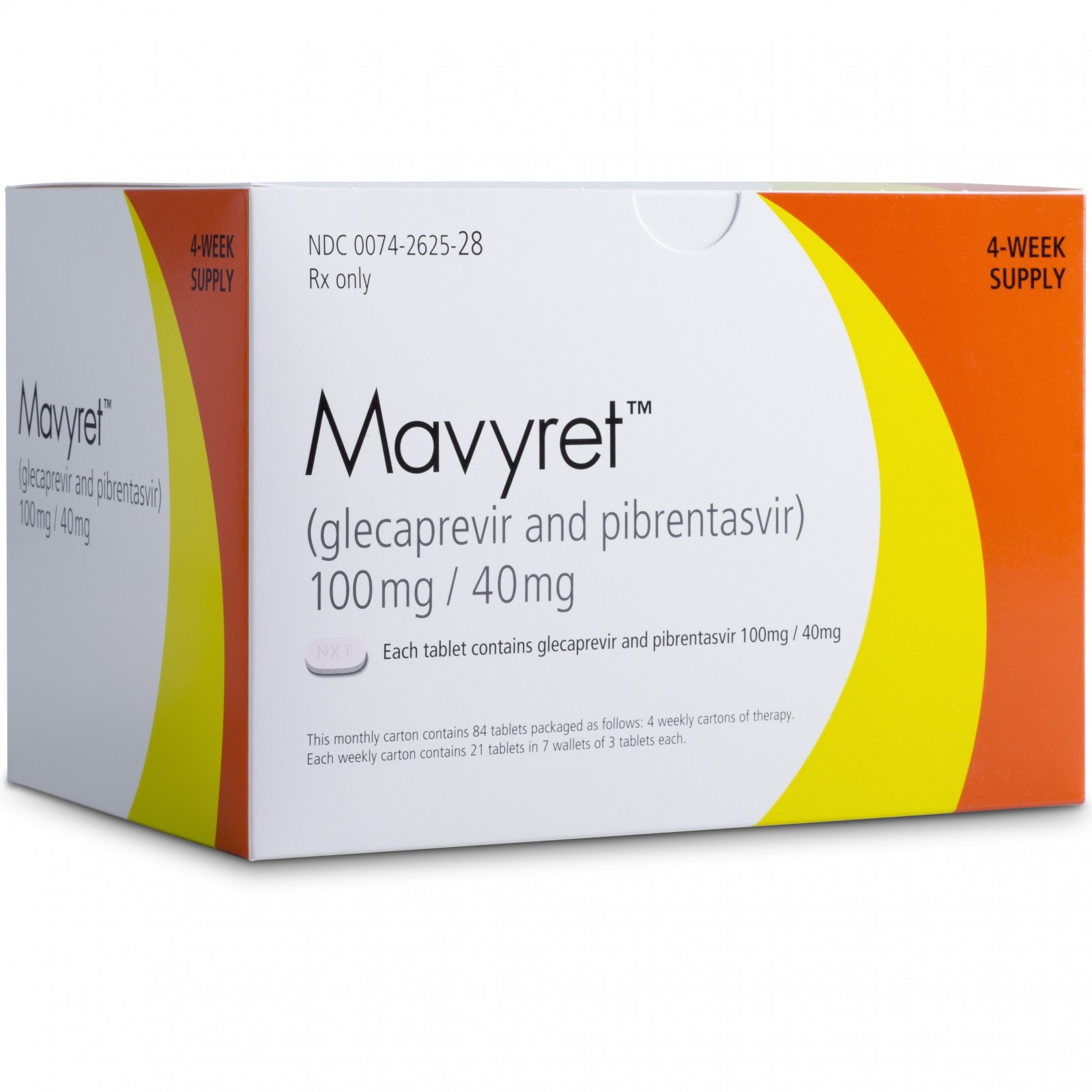
• Mavyret (glecaprevir and pibrentasvir) to treat adults with chronic hepatitis C virus genotypes 1-6 without cirrhosis (liver disease) or with mild cirrhosis, including patients with moderate to severe kidney disease, as well as those who are on hemodialysis;
• Mepsevii (vestronidase alfa-vjbk) to treat patients with Sly syndrome or mucopolysaccharidosis type 7 – a rare genetic disorder where an enzyme deficiency results in skeletal abnormalities, developmental delay, enlarged liver and spleen, and narrowed airways, which can lead to respiratory infections;
• Nerlynx (neratinib) for the extended adjuvant treatment — a form of therapy administered after an initial treatment to further lower the risk of the cancer coming back — of early-stage, human epidermal growth factor receptor 2 (HER2)-positive breast cancer;
• OCREVUS, OCRELIZUMAB, To treat patients with relapsing and primary progressive forms of multiple sclerosis
• OZEMPIC SEMAGLUTIDE To improve glycemic control in adults with type 2 diabetes mellitus
•PARSABIV, ETELCALCETIDE, To treat secondary hyperparathyroidism in adult patients with chronic kidney disease undergoing dialysis
• Prevymis (letermovir) for prevention of an infection called cytomegalovirus (CMV) in patients who are receiving a bone marrow transplant. CMV disease can cause serious health issues in these patients;
• Radicava (edaravone) to treat patients with amyotrophic lateral sclerosis, commonly referred to as Lou Gehrig’s disease, a rare disease that attacks and kills the nerve cells that control voluntary muscles;
• RHOPRESSA, NETARSUDIL, To treat glaucoma or ocular hypertension
• Rydapt (midostaurin) to treat adults newly diagnosed with a form of blood cancer known as acute myeloid leukemia who have a specific genetic mutation called FLT3, in combination with chemotherapy;
• Siliq (brodalumab) to treat adults with moderate-to-severe plaque psoriasis, a chronic disorder in which the body’s immune system sends out faulty signals that speed growth of skin cells that then accumulate, causing red, flaky patches that can appear anywhere on the body;
•SOLOSEC, SECNIDAZOLE To treat bacterial vaginosis
• STEGLATRO ERTUGLIFLOZIN To improve glycemic control in adults with type 2 diabetes mellitus
• Symproic (Naldemedine) for the treatment of opioid-induced constipation in adults with chronic noncancer pain; • Tremfya (guselkumab) for the treatment of adults with moderate-to-severe plaque psoriasis;
• Trulance (plecanatide) to treat adults with chronic idiopathic constipation, which is a persistent condition of constipation due to unknown origin;
• TYMLOS, Abaloparatide, To treat osteoporosis in postmenopausal women at high risk of fracture or those who have failed other therapies
• Vabomere (vaborbactam and meropenem) for treatment of adults with complicated urinary tract infections, including pyelonephritis (kidney infection) caused by bacteria;
• Verzenio (abemaciclib) to treat adults who have hormone receptor (HR)-positive, HER2-negative advanced or metastatic breast cancer that has progressed after taking therapy that alters a patient’s hormones (endocrine therapy);
• Vosevi (sofosbuvir/velpatasvir/voxilaprevir) to treat adults with chronic hepatitis C virus genotypes 1-6 without cirrhosis (liver disease) or with mild cirrhosis;
• Xadago (safinamide) as an add-on treatment for patients with Parkinson’s disease who are currently taking levodopa/carbidopa and experiencing “off” episodes;
• XERMELO, TELOTRISTAT ETHYL combined with somatostatin analog (SSA) therapy to treat adults with carcinoid syndrome diarrhea that SSA therapy alone has inadequately controlled, and;
• XEPI OZENOXACIN TO TREAT IMPETIGO
•XERMELO, TELOTRISTAT ETHYL, To treat carcinoid syndrome diarrhea
• Zejula (niraparib) for the maintenance treatment (intended to delay cancer growth) of adults with recurrent epithelial ovarian, fallopian tube or primary peritoneal cancer, whose tumors have completely or partially shrunk (complete or partial response, respectively) in response to platinum-based chemotherapy
USFDA
| No. | Drug Name |
Active Ingredient | Approval Date | FDA-approved use on approval date |
|---|---|---|---|---|
| 46. | Giapreza | angiotensin II | 12/21/2017 | To increase blood pressure in adults with septic or other distributive shock |
| 45. | Macrilen | macimorelin acetate | 12/20/2017 | For the diagnosis of adult growth hormone deficiency |
| 44. | Steglatro | ertugliflozin | 12/19/2017 | To improve glycemic control in adults with type 2 diabetes mellitus |
| 43. | Rhopressa | netarsudil | 12/18/2017 | To treat glaucoma or ocular hypertension |
| 42. | Xepi | ozenoxacin | 12/11/2017 | To treat impetigo Drug Trials Snapshot |
| 41. | Ozempic | semaglutide | 12/5/2017 | To improve glycemic control in adults with type 2 diabetes mellitus |
| 40. | Hemlibra | emicizumab | 11/16/2017 | To prevent or reduce the frequency of bleeding episodes in adult and pediatric patients with hemophilia A who have developed antibodies called Factor VIII (FVIII) inhibitors. |
| 39. | Mepsevii | vestronidase alfa-vjbk | 11/15/2017 | To treat pediatric and adult patients with an inherited metabolic condition called mucopolysaccharidosis type VII (MPS VII), also known as Sly syndrome. |
| 38. | Fasenra | benralizumab | 11/14/2017 | For add-on maintenance treatment of patients with severe asthma aged 12 years and older, and with an eosinophilic phenotype Drug Trials Snapshot |
| 37. | Prevymis | letermovir | 11/8/2017 | To prevent infection after bone marrow transplant Drug Trials Snapshot |
| 36. | Vyzulta | latanoprostene bunod ophthalmic solution | 11/2/2017 | To treat intraocular pressure in patients with open-angle glaucoma or ocular hypertension. Drug Trials Snapshot |
| 35. | Calquence | acalabrutinib | 10/31/2017 | To treat adults with mantle cell lymphoma Press Release Drug Trials Snapshot |
| 34. | Verzenio | abemaciclib | 9/28/2017 | To treat certain advanced or metastatic breast cancers Press Release Drug Trials Snapshot |
| 33. | Solosec | secnidazole | 9/15/2017 | To treat bacterial vaginosis Drug Trials Snapshot |
| 32. | Aliqopa | copanlisib | 9/14/2017 | To treat adults with relapsed follicular lymphoma Press Release Drug Trials Snapshot |
| 31. | benznidazole | benznidazole | 8/29/2017 | To treat children ages 2 to 12 years old with Chagas disease Press Release Drug Trials Snapshot |
| 30. | Vabomere | meropenem and vaborbactam | 8/29/2017 | To treat adults with complicated urinary tract infections Press Release Drug Trials Snapshot |
| 29. | Besponsa | inotuzumab ozogamicin | 8/17/2017 | To treat adults with relapsed or refractory acute lymphoblastic leukemia Press Release Drug Trials Snapshot |
| 28. | Mavyret | glecaprevir and pibrentasvir | 8/3/2017 | To treat adults with chronic hepatitis C virus Press Release Drug Trials Snapshot |
| 27. | Idhifa | enasidenib | 8/1/2017 | To treat relapsed or refractory acute myeloid leukemia Press Release Drug Trials Snapshot |
| 26. | Vosevi | sofosbuvir, velpatasvir and voxilaprevir | 7/18/2017 | To treat adults with chronic hepatitis C virus Press Release Drug Trials Snapshot |
| 25. | Nerlynx | neratinib maleate | 7/17/2017 | To reduce the risk of breast cancer returning Press Release Drug Trials Snapshot |
| 24. | Tremfya | guselkumab | 7/13/2017 | For the treatment of adult patients with moderate-to-severe plaque psoriasis Drug Trials Snapshot |
| 23. | Bevyxxa | betrixaban | 6/23/2017 | For the prophylaxis of venous thromboembolism (VTE) in adult patients hospitalized for an acute medical illness Drug Trials Snapshot |
| 22. | Baxdela | delafloxacin | 6/19/2017 | To treat patients with acute bacterial skin infections Drug Trials Snapshot |
| 21. | Kevzara | sarilumab | 5/22/2017 | To treat adult rheumatoid arthritis Drug Trials Snapshot |
| 20. | Radicava | edaravone | 5/5/2017 | To treat patients with amyotrophic lateral sclerosis (ALS) Press Release Drug Trials Snapshot |
| 19. | Imfinzi | durvalumab | 5/1/2017 | To treat patients with locally advanced or metastatic urothelial carcinoma Web Post Drug Trials Snapshot |
| 18. | Tymlos | abaloparatide | 4/28/2017 | To treat osteoporosis in postmenopausal women at high risk of fracture or those who have failed other therapies Drug Trials Snapshot |
| 17. | Rydapt | midostaurin | 4/28/2017 | To treat acute myeloid leukemia Press Release Chemistry Review(s) (PDF) Drug Trials Snapshot |
| 16. | Alunbrig | brigatinib | 4/28/2017 | To treat patients with anaplastic lymphoma kinase (ALK)-positive metastatic non-small cell lung cancer (NSCLC) who have progressed on or are intolerant to crizotinib Drug Trials Snapshot |
| 15. | Brineura | cerliponase alfa | 4/27/2017 | To treat a specific form of Batten disease Press Release Drug Trials Snapshot |
| 14. | Ingrezza | valbenazine | 4/11/2017 | To treat adults with tardive dyskinesia Press Release Chemistry Review(s) (PDF)Drug Trials Snapshot |
| 13. | Austedo | deutetrabenazine | 4/3/2017 | For the treatment of chorea associated with Huntington’s disease Drug Trials Snapshot, Chemistry Review(s) (PDF) |
| 12. | Ocrevus | ocrelizumab | 3/28/2017 | To treat patients with relapsing and primary progressive forms of multiple sclerosis Press Release Drug Trials Snapshot |
| 11. | Dupixent | dupilumab | 3/28/2017 | To treat adults with moderate-to-severe eczema (atopic dermatitis) Press Release Drug Trials Snapshot |
| 10. | Zejula | niraparib | 3/27/2017 | For the maintenance treatment for recurrent epithelial ovarian, fallopian tube or primary peritoneal cancers Press Release Drug Trials Snapshot |
| 9. | Symproic | naldemedine | 3/23/2017 |
For the treatment of opioid-induced constipation |
| 8. | Bavencio | avelumab | 3/23/2017 | To treat metastatic Merkel cell carcinoma Press Release Drug Trials Snapshot |
| 7. | Xadago | safinamide | 3/21/2017 | To treat Parkinson’s disease Press Release Drug Trials SnapshotChemistry Review(s) (PDF) |
| 6. | Kisqali | ribociclib | 3/13/2017 | To treat postmenopausal women with a type of advanced breast cancer Drug Trials Snapshot |
| 5. | Xermelo | telotristat ethyl | 2/28/2017 | To treat carcinoid syndrome diarrhea Press Release Drug Trials Snapshot |
| 4. | Siliq | brodalumab | 2/15/2017 | To treat adults with moderate-to-severe plaque psoriasis Press Release Drug Trials Snapshot |
| 3. | Emflaza | deflazacort | 2/9/2017 | To treat patients age 5 years and older with Duchenne muscular dystrophy (DMD) Press Release Drug Trials Snapshot |
| 2. | Parsabiv | etelcalcetide | 2/7/2017 | To treat secondary hyperparathyroidism in adult patients with chronic kidney disease undergoing dialysis Drug Trials Snapshot |
| 1. | Trulance | plecanatide | 1/19/2017 | To treat Chronic Idiopathic Constipation (CIC) in adult patients. Press Release Drug Trials Snapshot |
* This information is currently accurate. In rare instances, it may be necessary for FDA to change a drug’s new molecular entity (NME) designation or the status of its application as a novel new biologics license application (BLA). For instance, new information may become available which could lead to a reconsideration of the original designation or status. If changes must be made to a drug’s designation or the status of an application as a novel BLA, the Agency intends to communicate the nature of, and the reason for, any revisions as appropriate.
| USFDA 2017 | ||||||
| 2017/12/21 | Angiotensin II | Giapreza | La Jolla Pharmaceutical | |||
| 2017/12/20 | Ertugliflozin | Steglatro | Merck Sharp Dohme | |||
| 2017/12/20 | Macimorelin acetate | Macrilen | Aeterna Zentaris GmbH | |||
| 2017/12/18 | Netarsudil mesylate | Rhopressa | Aerie Pharmaceuticals | |||
| 2017/12/11 | Ozenoxacin | Xepi | Ferrer Internacional S.A. | |||
| 2017/12/5 | Semaglutide | Ozempic | Novo Nordisk Inc | |||
| 2017/11/16 | Emicizumab | Hemlibra | Genentech | BLA | ||
| 2017/11/15 | Vestronidase alfa | Mepsevii | Ultragenyx Pharmaceutical | BLA | ||
| 2017/11/14 | Benralizumab | Fasenra | AstraZeneca AB | BLA | ||
| 2017/11/8 | Letermovir | Prevymis | Merck Sharp Dohme | |||
| 2017/11/2 | Latanoprostene bunod | Vyzulta | Bausch & Lomb Incorporated | |||
| 2017/10/31 | Acalabrutinib | Calquence | AstraZeneca Pharmaceuticals LP | |||
| 2017/9/28 | Abemaciclib | Verzenio | Eli Lilly | |||
| 2017/9/15 | Secnidazole | Solosec | Symbiomix Therapeutics | |||
| 2017/9/14 | Copanlisib | Aliqopa | Bayer Healthcare Pharmaceuticals | |||
| 2017/8/29 | Benznidazole | Chemo Research | ||||
| 2017/8/29 | Meropenem – Vaborbactam | Vabomere | Rempex Pharmaceuticals | |||
| 2017/8/17 | Inotuzumab ozogamicin | Besponsa | Wyeth Pharmaceuticals | BLA | ||
| 2017/8/3 | Glecaprevir – Pibrentasvir | Mavyret | AbbVie | |||
| 2017/8/1 | Enasidenib | Idhifa | Celgene Corporation | |||
| 2017/7/18 | Sofosbuvir – Velpatasvir – Voxilaprevir | Vosevi | Gilead Sciences | |||
| 2017/7/17 | Neratinib maleate | Nerlynx | Puma Biotechnology | |||
| 2017/7/13 | Guselkumab | Tremfya | Janssen Biotech | BLA | ||
| 2017/6/23 | Betrixaban | Bevyxxa | Portola Pharmaceuticals | |||
| 2017/6/19 | Delafloxacin meglumine | Baxdela | Melinta Therapeutics | |||
| 2017/5/22 | Sarilumab | Kevzara | Sanofi Synthelabo | BLA | ||
| 2017/5/5 | Edaravone | Radicava | Mitsubishi Tanabe Pharma America | |||
| 2017/5/1 | Durvalumab | Imfinzi | AstraZeneca UK | BLA | ||
| 2017/4/28 | Abaloparatide | Tymlos | Radius Health | |||
| 2017/4/28 | Midostaurin | Rydapt | Novartis Pharmaceuticals | |||
| 2017/4/28 | Brigatinib | Alunbrig | Ariad Pharmaceuticals | |||
| 2017/4/27 | Cerliponase alfa | Brineura | BioMarin Pharmaceutical | BLA | ||
| 2017/4/11 | Valbenazine | Ingrezza | Neurocrine Biosciences | |||
| 2017/4/3 | Deutetrabenazine | Austedo | Teva Pharmaceuticals | |||
| 2017/3/28 | Ocrelizumab | Ocrevus | Genentech | BLA | ||
| 2017/3/28 | Dupilumab | Dupixent | Regeneron Pharmaceuticals | BLA | ||
| 2017/3/27 | Niraparib | Zejula | Tesaro | |||
| 2017/3/23 | Naldemedine tosylate | Symproic | Shionogi | |||
| 2017/3/23 | Avelumab | Bavencio | EMD Serono | BLA | ||
| 2017/3/23 | Safinamide mesylate | Xadago | Newron Pharmaceuticals | |||
| 2017/3/21 | Ribociclib | Kisqali | Novartis Pharmaceuticals | |||
| 2017/2/28 | Telotristat ethyl | Xermelo | Lexicon Pharmaceuticals | |||
| 2017/2/15 | Brodalumab | Siliq | Valeant Pharmaceuticals | BLA | ||
| 2017/2/9 | Deflazacort | Emflaza | Marathon Pharmaceuticals | |||
| 2017/2/8 | Etelcalcetide hydrochloride | Parsavib | KAI Pharmaceuticals | |||
| 2017/1/19 | Plecanatide | Trulance | Synergy Pharmaceuticals |
1 Abaloparatide
RADIUS

FDA 4/28/2017
To treat osteoporosis in postmenopausal women at high risk of fracture or those who have failed other therapies
Drug Trials Snapshot

2 Abemaciclib
ELI LILLY
| Verzenio | abemaciclib | FDA 9/28/2017 | To treat certain advanced or metastatic breast cancers Press Release Drug Trials Snapshot |
LINK https://newdrugapprovals.org/2015/10/19/abemaciclib-bemaciclib/
3 Acalabrutinib
| Calquence | FDA APPROVED
10/31/2017 |
To treat adults with mantle cell lymphoma Press Release Drug Trials Snapshot |

-Facebook.png)
4 Angiotensin II
LA JOLLA
| Giapreza | angiotensin II | 12/21/2017 | To increase blood pressure in adults with septic or other distributive shock Press Release Drug Trials Snapshot |


5 AVELUMAB
MERCK

| Bavencio | FDA 3/23/2017 | To treat metastatic Merkel cell carcinoma Press Release Drug Trials Snapshot |
6 BENRALIZUMAB
ASTRA ZENECA
Fasenra benralizumab
FDA 11/14/2017
For add-on maintenance treatment of patients with severe asthma aged 12 years and older, and with an eosinophilic phenotype
Drug Trials Snapshot


7 Benznidazole
CHEMO RESEARCH



| benznidazole | FDA
8/29/2017 |
To treat children ages 2 to 12 years old with Chagas disease Press Release Drug Trials Snapshot |
8 BETRIXABAN
PORTOLA PHARMA

| Bevyxxa | FDA
6/23/2017 |
For the prophylaxis of venous thromboembolism (VTE) in adult patients hospitalized for an acute medical illness Drug Trials Snapshot
|


9 BRIGATINIB
TAKEDA


| Alunbrig | FDA
4/28/2017 |
To treat patients with anaplastic lymphoma kinase (ALK)-positive metastatic non-small cell lung cancer (NSCLC) who have progressed on or are intolerant to crizotinib Drug Trials Snapshot |
10 BRODALUMAB
VALEANT PHARMA
| Siliq | FDA
2/15/2017 |
To treat adults with moderate-to-severe plaque psoriasis Press Release Drug Trials Snapshot |

LINK ,,,,https://newdrugapprovals.org/2017/02/16/fda-approves-new-psoriasis-drug-siliq-brodalumab/
11 CERLIPONASE ALFA

| Brineura | FDA 4/27/2017 | To treat a specific form of Batten disease Press Release Drug Trials Snapshot |
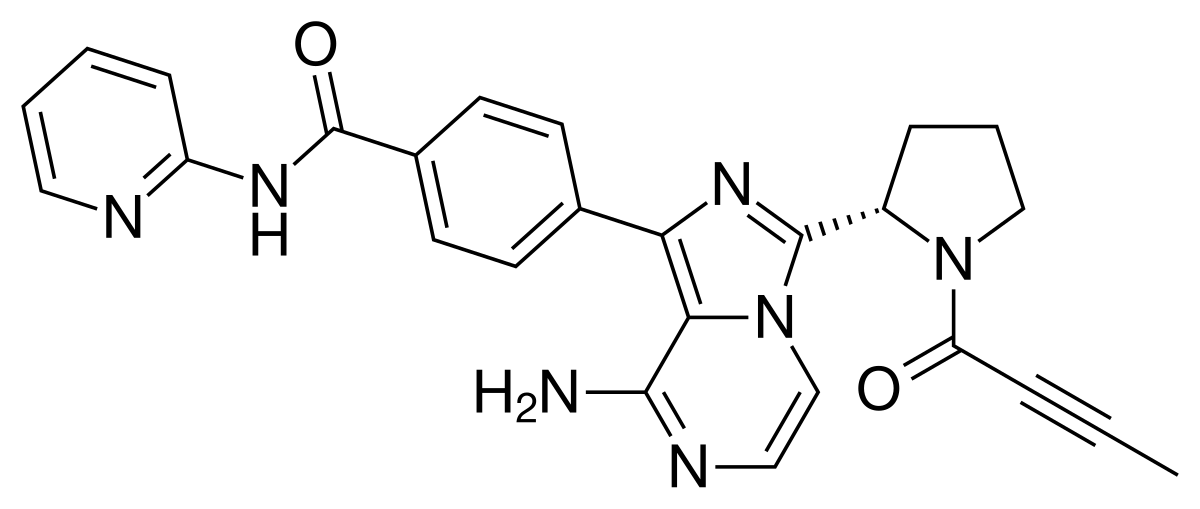
12 Copanlisib
| Aliqopa | FDA APPROVED
9/14/2017 |
To treat adults with relapsed follicular lymphoma Press Release Drug Trials Snapshot |

LINK…..https://newdrugapprovals.org/2017/11/20/copanlisib/
13 DEFLAZACORT
MARATHON PHARMA

| Emflaza | FDA 2/9/2017 | To treat patients age 5 years and older with Duchenne muscular dystrophy (DMD) Press Release Drug Trials Snapshot |
LINK……https://newdrugapprovals.org/2017/02/17/deflazacort/
14 DELAFLOXACIN
| Baxdela | FDA APPROVED
6/19/2017 |
To treat patients with acute bacterial skin infections |


LINK……..https://newdrugapprovals.org/2018/01/25/delafloxacin/
15 Deutetrabenazine
TEVA
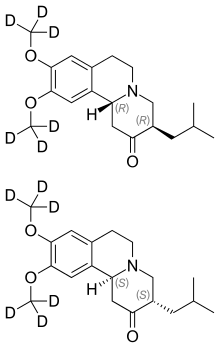

LINK……………https://newdrugapprovals.org/2015/08/15/sd-809-deutetrabenazine-nda-submitted-by-teva/
| Austedo | FDA 4/3/2017 | For the treatment of chorea associated with Huntington’s disease Drug Trials Snapshot Chemistry Review(s) (PDF) |
16 DUPILUMAB
SANOFI/REGENERON


| Dupixent | FDA | 3/28/2017 | To treat adults with moderate-to-severe eczema (atopic dermatitis) Press Release Drug Trials Snapshot |
LINK…….https://newdrugapprovals.org/2017/03/29/fda-approves-new-eczema-drug-dupixent-dupilumab/
17 DURVALUMAB
ASTRA ZENECA
durvalumab FDA 5/1/2017To treat patients with locally advanced or metastatic urothelial carcinoma
Web Post
Drug Trials Snapshot
18 EDAVARONE

MITSUBISHI TANABE
| Radicava | FDA 5/5/2017 | To treat patients with amyotrophic lateral sclerosis (ALS) Press Release Drug Trials Snapshot |


19 EMICIZUMAB
ROCHE

| Hemlibra | emicizumab | FDA 11/16/2017 | To prevent or reduce the frequency of bleeding episodes in adult and pediatric patients with hemophilia A who have developed antibodies called Factor VIII (FVIII) inhibitors. Press Release Drug Trials Snapshot |

20 Enasidenib


| Idhifa | FDA
8/1/2017 |
To treat relapsed or refractory acute myeloid leukemia Press Release Drug Trials Snapshot |
21 Ertugliflozin
MERCK
| Steglatro | ertugliflozin | FDA
12/19/2017 |
To improve glycemic control in adults with type 2 diabetes mellitus Drug Trials Snapshot |
LINK https://newdrugapprovals.org/2014/02/10/ertugliflozin/

22 ETELCALCETIDE
Amgen
| Parsabiv | FDA APPROVED
2/7/2017 |
To treat secondary hyperparathyroidism in adult patients with chronic kidney disease undergoing dialysis Drug Trials SnapshotSYNTHESIS LINK……..https://cen.acs.org/articles/96/i4/the-year-in-new-drugs-2018.html |
SYNTHESIS LINK……..https://cen.acs.org/articles/96/i4/the-year-in-new-drugs-2018.html
23 GLECAPREVIR
ABBVIE

| Mavyret | glecaprevir and pibrentasvir | FDA 8/3/2017 | To treat adults with chronic hepatitis C virus Press Release Drug Trials Snapshot |
LINK https://newdrugapprovals.org/2016/10/05/glecaprevir-abt-493/
24 GUSELKUMAB
JOHNSON AND JOHNSON
guselkumab
FDA 7/13/2017
For the treatment of adult patients with moderate-to-severe plaque psoriasis
Drug Trials Snapshot


25 Inotuzumab ozogamicin
PFIZER



| Besponsa | FDA
8/17/2017 |
To treat adults with relapsed or refractory acute lymphoblastic leukemia Press Release Drug Trials Snapshot |
26 LATANOPROSTENE
VALEANT
latanoprostene bunod ophthalmic solution
FDA 11/2/2017
To treat intraocular pressure in patients with open-angle glaucoma or ocular hypertension.
Drug Trials Snapshot
27 LETERMOVIR
MERCK
| Prevymis | FDA 11/8/2017 | To prevent infection after bone marrow transplant Drug Trials Snapshot |
LINK https://newdrugapprovals.org/2016/05/16/letermovir-aic-246/
28 Macimorelin acetate
AETERNA ZENTARIS
| Macrilen | macimorelin acetate | FDA
12/20/2017 |
For the diagnosis of adult growth hormone deficiency Drug Trials Snapshot |
29 MEROPENEM

30 MIDOSTAURIN
NOVARTIS
- Chemistry Review(s) (PDF)

| Rydapt | FDA
4/28/2017 |
To treat acute myeloid leukemia Press Release Drug Trials Snapshot |
31 Naldemedine
FDA 3/23/2017, Symproic, For the treatment of opioid-induced constipation


32 NERATINIB MALEATE
PUMA BIOTECH


| Nerlynx | FDA | 7/17/2017 | To reduce the risk of breast cancer returning Press Release Drug Trials Snapshot |
33 NETARSUDIL
| Rhopressa | FDA APPROVED
12/18/2017 |
To treat glaucoma or ocular hypertension |


LINK……https://newdrugapprovals.org/2018/01/29/netarsudil/
34 NIRAPARIB
TESARO
| Zejula | FDA | 3/27/2017 | For the maintenance treatment for recurrent epithelial ovarian, fallopian tube or primary peritoneal cancers Press Release Drug Trials Snapshot |

![]()

LINK…https://newdrugapprovals.org/2016/12/22/niraparib-mk-4827/
35 OCRELIZUMAB
ROCHE
| Ocrevus | FDA | 3/28/2017 | To treat patients with relapsing and primary progressive forms of multiple sclerosis Press Release Drug Trials Snapshot |


36 OZENOXACIN
MEDIMETRIX

| Xepi | ozenoxacin | FDA
12/11/2017 |
To treat impetigo Drug Trials Snapshot |
37 Pibrentasvir
ABBVIE

| Mavyret | glecaprevir and pibrentasvir | FDA 8/3/2017 | To treat adults with chronic hepatitis C virus Press Release Drug Trials Snapshot |
LINK https://newdrugapprovals.org/2016/06/08/abt-530-pibrentasvir/
38 PLECANATIDE
Plecanatide 普卡那肽 ليكاناتيد плеканатид
SYNERGY PHARMA


| Trulance | FDA APPROVED
1/19/2017 |
To treat Chronic Idiopathic Constipation (CIC) in adult patients. Press Release Drug Trials Snapshot |
39 RIBOCICLIB
NOVARTIS
Structure..link for correct structure
| Kisqali | FDA 3/13/2017 | To treat postmenopausal women with a type of advanced breast cancer Drug Trials Snapshot |

LINK https://newdrugapprovals.org/2015/10/19/ribociclib/
40 SARILUMAB
SANOFI /REGENERON

| Kevzara | sarilumab | FDA 5/22/2017 | To treat adult rheumatoid arthritis Drug Trials Snapshot |
LINK https://newdrugapprovals.org/2013/11/25/late-stage-success-for-sanofiregeneron-ra-drug-sarilumab/


41 SECNIDAZOLE
SYMBIOMIX

| Solosec | FDA | 9/15/2017 | To treat bacterial vaginosis Drug Trials Snapshot |

42 SAFINAMIDE
NEWRON PHARMA



- Chemistry Review(s) (PDF) for correct structure
| Xadago | FDA 3/21/2017 | To treat Parkinson’s disease Press Release Drug Trials Snapshot |
43 Semaglutide
NOVO NORDISK
| Ozempic | semaglutide | FDA
12/5/2017 |
To improve glycemic control in adults with type 2 diabetes mellitus Drug Trials Snapshot |
LINK https://newdrugapprovals.org/2013/07/22/a-survey-of-promising-late-stage-diabetes-drugs/

44 SOFOSBUVIR
45 TELOTRISTAT ETHYL
LEXICON
| Xermelo | FDA
2/28/2017 |
To treat carcinoid syndrome diarrhea Press Release Drug Trials Snapshot |


46 VABORBACTAM
THE MEDICINES CO

| Vabomere | meropenem and vaborbactam | FDA
8/29/2017 |
To treat adults with complicated urinary tract infections Press Release Drug Trials Snapshot |

47 VALBENAZINE
NEUROCRINE
- Chemistry Review(s) (PDF)
| Ingrezza | FDA
4/11/2017 |
To treat adults with tardive dyskinesia Press Release Drug Trials Snapshot |
48 Vestronidase alfa-vjbk
ULTRAGENYX
| Mepsevii | vestronidase alfa-vjbk | FDA 11/15/2017 | To treat pediatric and adult patients with an inherited metabolic condition called mucopolysaccharidosis type VII (MPS VII), also known as Sly syndrome. Press Release Drug Trials Snapshot |

49 VELPATASVIR
50 VOXILAPREVIR
GILEAD
| Vosevi | sofosbuvir, velpatasvir and voxilaprevir | FDA 7/18/2017 | To treat adults with chronic hepatitis C virus Press Release Drug Trials Snapshot |
SECTION B; EMA approvals
European Medicines Agency’s – Human medicines: Highlights of 2017
Advances in medicines authorizations are essential for public health as they have the potential to improve treatment of diseases. In 2017, EMA recommended 92 medicines for marketing authorization. Of these, 35 had a new active substance, which has never been authorized in the European Union (EU) before. Many of these medicines represent a significant improvement in their therapeutic areas; they include medicines for children, for rare diseases and advanced therapies42. Amongst the 35 new active substances (NAS) that EMA recommended, 11 were new drugs and biologics to treat cancer, 05 to treat neurological disorders, 04 for infectious diseases, 04 for immunology/rheumatology, 03 for endocrinology, 02 each for Uro-nephrology, haematology, and dermatology, 01 for Pneumonology, and 01 for hepatology/gastroenterology class of drugs.
EUROPE
| 2017/11/16 | Niraparib | Zejula | Tesaro UK Limited | O | NME | ||
| 2017/11/10 | Adalimumab | Cyltezo | Boehringer Ingelheim International GmbH | B | |||
| 2017/11/10 | Miglustat | Miglustat Gen.Orph | Gen.Orph | G | |||
| 2017/11/10 | Ritonavir | Ritonavir Mylan | MYLAN S.A.S | G | |||
| 2017/11/10 | Padeliporfin | Tookad | STEBA Biotech S.A | ||||
| 2017/11/10 | Guselkumab | Tremfya | Janssen-Cilag International N.V. | BLA | |||
| 2017/9/27 | Dupilumab | Dupixent | sanofi-aventis groupe | BLA | |||
| 2017/9/21 | Darunavir / Cobicistat / Emtricitabine / Tenofovir alafenamide | Symtuza | Janssen-Cilag International N.V. | ||||
| 2017/9/21 | Atezolizumab | Tecentriq | Roche Registration Limited | BLA | |||
| 2017/9/18 | Avelumab | Bavencio | Merck Serono Europe Limited | O | BLA | ||
| 2017/9/18 | Entecavir | Entecavir Mylan | Mylan S.A.S | G | |||
| 2017/9/18 | Lacosamide | Lacosamide Accord | Accord Healthcare Ltd | G | |||
| 2017/9/18 | Midostaurin | Rydapt | Novartis Europharm Ltd | O | NME | ||
| 2017/9/18 | Telotristat ethyl | Xermelo | Ipsen Pharma | O | NME | ||
| 2017/9/5 | Trientine | Cuprior | GMP-Orphan SA | ||||
| 2017/9/5 | Efavirenz / Emtricitabine / Tenofovir disoproxil | Efavirenz/Emtricitabine/Tenofovir disoproxil Mylan | Mylan S.A.S | G | |||
| 2017/8/24 | Tivozanib hydrochloride monohydrate | Fotivda | EUSA Pharma (UK) Limited | NME | |||
| 2017/8/24 | Adalimumab | Imraldi | Samsung Bioepis UK Limited (SBUK) | B | |||
| 2017/8/24 | Nitisinone | Nitisinone MDK (previously Nitisinone MendeliKABS) | MendeliKABS Europe Ltd | G | |||
| 2017/8/22 | Ribociclib | Kisqali | Novartis Europharm Ltd | NME | |||
| 2017/8/22 | Cladribine | Mavenclad | Merck Serono Europe Limited | ||||
| 2017/7/26 | Glecaprevir / Pibrentasvir | Maviret | AbbVie Limited | NME | |||
| 2017/7/26 | Sofosbuvir / Velpatasvir / Voxilaprevi | Vosevi | Gilead Sciences International Ltd | NME | |||
| 2017/7/19 | Insulin lispro | Insulin lispro Sanofi | sanofi-aventis groupe | B | |||
| 2017/7/19 | Patiromer sorbitex calcium | Veltassa | Vifor Fresenius Medical Care Renal Pharma France | NME | |||
| 2017/7/17 | Efavirenz / Emtricitabine / Tenofovir disoproxil | Efavirenz/Emtricitabine/Tenofovir disoproxil Zentiva | Zentiva k.s. | G | |||
| 2017/7/17 | Brodalumab | Kyntheum | LEO Pharma A/S | BLA | |||
| 2017/7/17 | beclometasone / formoterol / glycopyrronium bromide | Trimbow | Chiesi Farmaceutici S.p.A. | ||||
| 2017/7/13 | Rituximab | Blitzima | Celltrion Healthcare Hungary Kft. | B | |||
| 2017/7/13 | Cariprazine | Reagila | Gedeon Richter | ||||
| 2017/7/10 | Spheroids of human autologous matrix-associated chondrocytes | Spherox | CO.DON AG | ||||
| 2017/7/6 | Cenegermin | Oxervate | Dompe farmaceutici s.p.a. | O | BLA | ||
| 2017/6/29 | Inotuzumab ozogamicin | Besponsa | Pfizer Limited | O | BLA | ||
| 2017/6/23 | Etanercept | Erelzi | Sandoz GmbH | ||||
| 2017/6/23 | Sarilumab | Kevzara | Sanofi-Aventis Groupe | NME | |||
| 2017/6/23 | Dimethyl fumarate | Skilarence | Almirall S.A | ||||
| 2017/6/23 | Carglumic acid | Ucedane | Lucane Pharma | G | |||
| 2017/6/15 | Rituximab | Rixathon, Riximyo B | Sandoz GmbH | ||||
| 2017/6/2 | Pentosan polysulfate sodium | Elmiron | bene-Arzneimittel GmbH | ||||
| 2017/6/2 | Nonacog beta pegol | Refixia | Novo Nordisk A/S | BLA | |||
| 2017/5/30 | Cerliponase alfa | Brineura | BioMarin International Limited | O E | BLA | ||
| 2017/5/30 | Nusinersen | Spinraza | Biogen Idec Ltd | O | NME | ||
| 2017/5/24 | Meningococcal group b vaccine (recombinant, adsorbed) | Trumenba | Pfizer Limited | ||||
| 2017/5/22 | Ivabradine | Ivabradine Accord | Accord Healthcare Ltd | G | |||
| 2017/5/8 | Dinutuximab beta | Dinutuximab beta Apeiron | Apeiron Biologics AG | O E | |||
| 2017/4/28 | Emtricitabine – tenofovir disoproxil mixt | Emtricitabine/Tenofovir disoproxil Krka d.d. | KRKA, d.d., Novo mesto | G | |||
| 2017/4/24 | Parathyroid hormone | Natpar | Shire Pharmaceuticals Ireland Ltd | O C | BLA | ||
| 2017/4/20 | Edoxaban | Roteas | Daiichi Sankyo Europe GmbH | ||||
| 2017/3/22 | Tofacitinib citrate | Xeljanz | Pfizer Limited | NME | |||
| 2017/3/20 | Umeclidinium | Rolufta | GlaxoSmithKline Trading Services Limited | ||||
| 2017/3/3 | Chlormethine | Ledaga | Actelion Registration Ltd. | O | |||
| 2017/2/27 | Pregabalin | Pregabalin Zentiva | Zentiva k.s. | G | |||
| 2017/2/17 | Rituximab | Truxima | Celltrion Healthcare Hungary Kft. | B | |||
| 2017/2/13 | Etanercept | Lifmior | Pfizer Limited | ||||
| 2017/2/13 | Baricitinib | Olumiant | Eli Lilly Nederland B.V. | NME | |||
| 2017/1/19 | Mercaptamine | Cystadrops | Orphan Europe S.A.R.L. | O | |||
| 2017/1/18 | Bezlotoxumab | Zinplava | Merck Sharp & Dohme Limited | NME | |||
| 2017/1/11 | Teriparatide | Movymia | STADA Arzneimittel AG | B | |||
| 2017/1/11 | Insulin glargine / lixisenatide | Suliqua | Sanofi-Aventis Groupe | ||||
| 2017/1/9 | Insulin aspart | Fiasp | Novo Nordisk A/S | ||||
| 2017/1/9 | Tadalafil | Tadalafil | Mylan S.A.S | G | |||
| 2017/1/9 | Tenofovir alafenamide | Vemlidy | Gilead Sciences International Ltd | ||||
| 2017/1/4 | Lonoctocog alfa | Afstyla | CSL Behring GmbH | BLA | |||
| 2017/1/4 | Darunavir | Darunavir Mylan | Mylan S.A.S. | G | |||
| 2017/1/4 | Insulin glargine | Lusduna | Merck Sharp & Dohme Limited | B | |||
| 2017/1/4 | Teriparatide | Terrosa | Gedeon Richter Plc. | B |
SECTION B; EMA Approvals
Combined drugs USFDA+EMA +PMDA list are listed below. trying to simplify search
1 Abaloparatide USFDA
2 Abemaciclib USFDA
3 ACALABRUTINIB USFDA
3A ALOFISEL EMA
3B AMENAMEVIR JAPAN
4 ANGIOTENSIN II USFDA
4A Atezolizumab EMA
5 AVELUMAB USFDA+EMA
6 BENRALIZUMAB USFDA+EMA
6A BARICITINIB JAPAN
7 BENZNIDAZOLE USFDA
8 BETRIXABAN USFDA
9 BRIGATINIB USFDA
10 BRODALUMAB USFDA+EMA
10A BUROSUMAB EMA
10B CARIPRAZINE HYDROCHLORIDE EMA
11 CERLIPONASE ALPA USFDA+EMA
12 COPANLISIB USFDA
13 DEFLAZACORT USFDA
14 Delafloxacin USFDA
15 Deutetrabenazine USFDA
16DUPILUMAB USFDA+EMA
17 DURVALUMAB USFDA
18 EDAVARONE USFDA
19 EMICIZUMAB USFDA
20 Enasidenib USFDA
21 ERTUGLIFLOZIN USFDA
22 ETELCALCETIDE USFDA
22A FORODESINE JAPAN
22B FLUCICLOVINE EMA
23 GLECAPREVIR USFDA+EMA
24 GUSELKUMAB USFDA+EMA
25 INOTUZUMAB OZOGAMICIN USFDA+EMA
26 LATANOPROSTENE USFDA
27 LETERMOVIR USFDA+EMA
27A Utetium lu 177 dotatate EMA
28 MACIMORELIN ACETATE USFDA
29 MEROPENEM USFDA
30 MIDOSTAURIN USFDA+EMA
31 NALDEMEDINE USFDA
32 NERATINIB USFDA
33 NETARSUDIL USFDA
34A NONACOG EMA
34B NUCINERSEN EMA +Japan
35 Ocrelizumab USFDA+EMA
35A OXERVATE EMA
36 OZENOXACIN USFDA
36A PATIROMER EMA
36B PADELIPORFIN EMA
36C PEMAFIBRATE JAPAN
37 PIBRENTASVIR USFDA+EMA
38 PLECANATIDE USFDA
39A ROLAPITANT EMA
39BRURLOCTOCOG EMA
40 SARILUMAB USFDA+EMA
41 SECNIDAZOLE USFDA
42 SAFINAMIDE USFDA
43 SEMAGLUTIDE USFDA+EMA
43A SODIUM ZIRCONIUM CYCLOCYLICATE EMA
44 SOFOSBUVIR USFDA+EMA
44A SPHEROX EMA
45 TELOTRISTAT ETHYL USFDA+EMA
45A TIVOZANIB EMA
45B TOFACITINIB EMA
45C TRUMENBA EMA
46 VABORBACTAM USFDA
47 VALBENAZINE USFDA
48 VESTRONIDASE ALFA-VJBK USFDA
49 VELPATASVIR USFDA+EMA
50 VOXILAPREVIR USFDA+EMA
Drugs EMA list missed out in usfda list
3A ALOFISEL
link………https://newdrugapprovals.org/2018/03/02/alofisel-darvadstrocel-cx-601/
4A Atezolizumab
WILL BE UPDATED
10A BUROSUMAB
WILL BE UPDATED
10B CARIPRAZINE HYDROCHLORIDE
WILL BE UPDATED
22B FLUCICLOVINE

SEE EMA
| Axumin : EPAR – Summary for the public | EN = English | 06/07/2017 |
27A Lutetium lu 177 dotatate
WILL BE UPDATED
34A NONACOG
WILL BE UPDATED
34B NUCINERSEN
EMA AND JAPAN 2017 APPROVED


35A OXERVATE
WILL BE UPDATED
36A PATIROMER
WILL BE UPDATED
36B PADELIPORFIN

| NAME | Tookad |
|---|---|
| AGENCY PRODUCT NUMBER | EMEA/H/C/004182 |
| ACTIVE SUBSTANCE | padeliporfin di-potassium |
| INTERNATIONAL NON-PROPRIETARY NAME(INN) OR COMMON NAME | padeliporfin |
| THERAPEUTIC AREA | Prostatic Neoplasms |
| ANATOMICAL THERAPEUTIC CHEMICAL (ATC) CODE | L01XD07 |
| ADDITIONAL MONITORING | This medicine is under additional monitoring. This means that it is being monitored even more intensively than other medicines. For more information, see medicines under additional monitoring. |
| MARKETING-AUTHORISATION HOLDER | STEBA Biotech S.A |
|---|---|
| REVISION | 0 |
| DATE OF ISSUE OF MARKETING AUTHORISATION VALID THROUGHOUT THE EUROPEAN UNION | 10/11/2017 |
Contact address:
STEBA Biotech S.A
7 place du theatre
L-2613 Luxembourg
Luxembourg
38A PRALATREXATE

Japan approved 2017
| 2017/7/3 | PMDA | JAPAN | Pralatrexate | Difolta | Mundipharma | NME |
39A ROLAPITANT
WILL BE UPDATED
39B RURLOCTOCOG
WILL BE UPDATED
43A SODIUM ZIRCONIUM
WILL BE UPDATED
44A SPHEROX
WILL BE UPDATED
45A TIVOZANIB

Pharmacotherapeutic group
Antineoplastic agents
Therapeutic indication
Fotivda is indicated for the first line treatment of adult patients with advanced renal cell carcinoma (RCC) and for adult patients who are VEGFR and mTOR pathway inhibitor-naïve following disease progression after one prior treatment with cytokine therapy for advanced RCC.
Treatment of advanced renal cell carcinoma
Fotivda : EPAR -Product Information

Tivozanib is synthesized in three main steps using well defined starting materials with acceptable
specifications.
Adequate in-process controls are applied during the synthesis. The specifications and control methods for
intermediate products, starting materials and reagents have been presented. The critical process
parameters are duly justified, methodology is presented and control is adequate.
The characterisation of the active substance and its impurities are in accordance with the EU guideline on
chemistry of new active substances. Potential and actual impurities were well discussed with regards to
their origin and characterised.
The active substance is packaged in a low-density polyethylene (LDPE) bag which complies with the EC
directive 2002/72/EC and EC 10/2011 as amended.
Product details
| Name | Fotivda |
|---|---|
| Agency product number | EMEA/H/C/004131 |
| Active substance | tivozanib |
| International non-proprietary name(INN) or common name | tivozanib hydrochloride monohydrate |
| Therapeutic area | Carcinoma, Renal Cell |
| Anatomical therapeutic chemical (ATC) code | L01XE |
Publication details
| Marketing-authorisation holder | EUSA Pharma (UK) Limited |
|---|---|
| Revision | 0 |
| Date of issue of marketing authorisation valid throughout the European Union | 24/08/2017 |
Contact address:
EUSA Pharma (UK) Limited
Breakspear Park, Breakspear Way
Hemel Hempstead, HP2 4TZ
United Kingdom
45B TOFACITINIB
WILL BE UPDATED
45C TRUMENBA
WILL BE UPDATED
SECTION C JAPANFORODOS
SECTION C New Drugs JAPAN
https://www.pmda.go.jp/english/review-services/reviews/approved-information/drugs/0002.html
JAPAN 2017
| 2017/9/27 | Avelumab (genetical recombination) | Bavencio | Merck Serono | BLA |
| 2017/9/27 | Glecaprevir – pibrentasvir mixt | Maviret | Abbvie | NME |
| 2017/9/27 | Daratumumab (genetical recombination) | Darzalex | Janssen Pharmaceutical | BLA |
| 2017/9/27 | Belimumab (genetical recombination) | Benlysta | GlaxoSmithKline | BLA |
| 2017/9/27 | Bezlotoxumab (genetical recombination) | Zinplava | MDS | BLA |
| 2017/9/27 | Palbociclib | Ibrance | Pfizer | NME |
| 2017/9/27 | Lonoctocog alfa (genetical recombination) | Afstyla | CSL Behring | BLA |
| 2017/9/27 | Rupatadine fumarate | Rupafin | Teikoku seiyaku | NME |
| 2017/9/27 | Sarilumab (genetical receombination) | Kevzara | Sanofi | BLA |
| 2017/9/27 | Flutemetamol (18F) | Vizamyl | Nihon Medi-Physics | NME |
| 2017/7/3 | Nusinersen sodium | Spinraza | Biogen Japan | |
| 2017/7/3 | Romidepsin | Istodax | Celgene | NME |
| 2017/7/3 | Pralatrexate | Difolta | Mundipharma | NME |
| 2017/7/3 | Amenamevir | Amenalief | Maruho | NME |
| 2017/7/3 | Baricitinib | Olumiant | Lilly | NME |
| 2017/7/3 | Pemafibrate | Parmodia | Kowa | NME |
| 2017/3/30 | Human prothrombin complex, freeze-dried concentrated | Kcentra | CSL Behring | |
| 2017/3/30 | Ixazomib citrate | Ninlaro | Takeda | NME |
| 2017/3/30 | Forodesine hydrochloride | Mundesine | Mundipharma | |
| 2017/3/30 | Aflibercept beta (genetical recombination) | Zaltrap | Sanofi | |
| 2017/3/30 | Hydromorphone hydrochloride | Narusus, Narurapid | DaiichiSankyo-pp | |
| 2017/3/30 | Naldemedine tosylate | Symproic | Shionogi | NME |
| 2017/3/30 | Guanfacine hydrochloride | Intuniv | Shionogi |
3B AMENAMEVIR

Originally developed by Astellas, the drug was licensed to Maruho. Amenamevir treats herpes zoster by inhibiting the activity of the helicase-primer enzyme during viral DNA replication and blocking the virus’s proliferation.
Amenalief® is an oral film-coated tablet containing 200 mg of amenamevir per tablet. Recommended dose of 1 day, 400mg each time, after meals.
22A FORODESINE HYDROCHLORIDE

LINK https://newdrugapprovals.org/2018/03/06/forodesine-hydrochloride/
6A BARICITINIB JAPAN

Originally developed by Incyte, Baricitinib was later licensed to and for sale by Lilly under the trade name Olumiant®. Baricitinib is an irreversible inhibitor of Janus kinase 1 (JAK1) and Janus kinase 2 (JAK2). Olumiant® is approved for the treatment of mild to moderate rheumatoid arthritis in adult patients who are not responsive or intolerant to other anti-arthritic drugs. This product can be used alone or in combination with methotrexate.
Olumiant® is a film-coated tablet containing 2 mg or 4 mg per tablet. Recommended oral dose is 4mg daily, with meals or fasting food, you can take any time period.
2017/7/3PMDA Baricitinib Olumiant Lilly
36C PEMAFIBRATE

LINK https://newdrugapprovals.org/2016/04/24/pemafibrate/
SECTION D
CDSCO INDIA


http://www.cdsco.nic.in/forms/list.aspx?lid=2034&Id=11 http://www.cdsco.nic.in/forms/list.aspx?lid=2034&Id=11






KEEP WATCHING UNDER CONSTRUCTION AND WILL BE PASTED SOON………………………………………..
KEEP WATCHING UNDER CONSTRUCTION AND WILL BE PASTED SOON………………………………………..
KEEP WATCHING UNDER CONSTRUCTION AND WILL BE PASTED SOON………………………………………..
KEEP WATCHING UNDER CONSTRUCTION AND WILL BE PASTED SOON………………………………………..
REFERENCES
2 http://www.ema.europa.eu/docs/en_GB/document_library/Report/2018/01/WC500242079.pdf
“NEW DRUG APPROVALS” CATERS TO EDUCATION GLOBALLY, No commercial exploits are done or advertisements added by me. This is a compilation for educational purposes only. P.S. : The views expressed are my personal and in no-way suggest the views of the professional body or the company that I represent
 amcrasto@gmail.com
amcrasto@gmail.com
I , Dr A.M.Crasto is writing this blog to share the knowledge/views, after reading Scientific Journals/Articles/News Articles/Wikipedia. My views/comments are based on the results /conclusions by the authors(researchers). I do mention either the link or reference of the article(s) in my blog and hope those interested can read for details. I am briefly summarising the remarks or conclusions of the authors (researchers). If one believe that their intellectual property right /copyright is infringed by any content on this blog, please contact or leave message at below email address amcrasto@gmail.com. It will be removed ASAP
////////EMA APPROVALS, USFDA Approvals, ACALABRUTINIB, AVELUMAB, BETRIXABAN, BRODALUMAB, COPANLISIB, DEFLAZACORT, Delafloxacin, Deutetrabenazine, DUPILUMAB, ETELCALCETIDE, Naldemedine, NETARSUDIL, NIRAPARIB, Ocrelizumab, PLECANATIDE, RIBOCICLIB, SAFINAMIDE, TELOTRISTAT ETHYL, VALBENAZINE, CERLIPONASE, BRIGATINIB, MIDOSTAURIN, Abaloparatide, BENZNIDAZOLE, NERATINIB, inotuzumab ozogamicin, Enasidenib, LETERMOVIR, GLECAPREVIR, PIBRENTASVIR, VOXILAPREVIR, SOFOSBUVIR, EDAVARONE, abemaciclib, ANGIOTENSIN II, VESTRONIDASE, macimorelin acetate, ERTUGLIFLOZIN, SEMAGLUTIDE, EMICIZUMAB, eu 2017, fda 2017, BENRALIZUMAB, DURVALUMAB, GUSELKUMAB, LATANOPROSTENE, OZENOXACIN, SARILUMAB, SECNIDAZOLE, BENRALIZUMAB, TIVOZANIB, SARILUMAB, FLUCICLOVINE,
FDA approves Vosevi for Hepatitis C
The U.S. Food and Drug Administration today approved Vosevi to treat adults with chronic hepatitis C virus (HCV) genotypes 1-6 without cirrhosis (liver disease) or with mild cirrhosis. Vosevi is a fixed-dose, combination tablet containing two previously approved drugs – sofosbuvir and velpatasvir – and a new drug, voxilaprevir. Vosevi is the first treatment approved for patients who have been previously treated with the direct-acting antiviral drug sofosbuvir or other drugs for HCV that inhibit a protein called NS5A.
“Direct-acting antiviral drugs prevent the virus from multiplying and often cure HCV. Vosevi provides a treatment option for some patients who were not successfully treated with other HCV drugs in the past,” said Edward Cox, M.D., director of the Office of Antimicrobial Products in the FDA’s Center for Drug Evaluation and Research.
Hepatitis C is a viral disease that causes inflammation of the liver that can lead to diminished liver function or liver failure. According to the Centers for Disease Control and Prevention, an estimated 2.7 to 3.9 million people in the United States have chronic HCV. Some patients who suffer from chronic HCV infection over many years may have jaundice (yellowish eyes or skin) and develop complications, such as bleeding, fluid accumulation in the abdomen, infections, liver cancer and death.
There are at least six distinct HCV genotypes, or strains, which are genetically distinct groups of the virus. Knowing the strain of the virus can help inform treatment recommendations. Approximately 75 percent of Americans with HCV have genotype 1; 20-25 percent have genotypes 2 or 3; and a small number of patients are infected with genotypes 4, 5 or 6.
The safety and efficacy of Vosevi was evaluated in two Phase 3 clinical trials that enrolled approximately 750 adults without cirrhosis or with mild cirrhosis.
The first trial compared 12 weeks of Vosevi treatment with placebo in adults with genotype 1 who had previously failed treatment with an NS5A inhibitor drug. Patients with genotypes 2, 3, 4, 5 or 6 all received Vosevi.
The second trial compared 12 weeks of Vosevi with the previously approved drugs sofosbuvir and velpatasvir in adults with genotypes 1, 2 or 3 who had previously failed treatment with sofosbuvir but not an NS5A inhibitor drug.
Results of both trials demonstrated that 96-97 percent of patients who received Vosevi had no virus detected in the blood 12 weeks after finishing treatment, suggesting that patients’ infection had been cured.
Treatment recommendations for Vosevi are different depending on viral genotype and prior treatment history.
The most common adverse reactions in patients taking Vosevi were headache, fatigue, diarrhea and nausea.
Vosevi is contraindicated in patients taking the drug rifampin.
Hepatitis B virus (HBV) reactivation has been reported in HCV/HBV coinfected adult patients who were undergoing or had completed treatment with HCV direct-acting antivirals, and who were not receiving HBV antiviral therapy. HBV reactivation in patients treated with direct-acting antiviral medicines can result in serious liver problems or death in some patients. Health care professionals should screen all patients for evidence of current or prior HBV infection before starting treatment with Vosevi.
The FDA granted this application Priority Review and Breakthrough Therapydesignations.
The FDA granted approval of Vosevi to Gilead Sciences Inc
//////////////Vosevi, Gilead Sciences Inc, Priority Review, Breakthrough Therapy designations, fda 2017, sofosbuvir, velpatasvir , voxilaprevir, Hepatitis B
LUPIN LIMITED, WO 2016181313, NEW PATENT, SOFOSBUVIR

WO2016181313, A PROCESS FOR THE PREPARATION OF SOFOSBUVIR INTERMEDIATES & ITS POLYMORPH
| LUPIN LIMITED [IN/IN]; Kalpataru Inspire 3rd Floor, Off Western Express Highway Santacruz (East) Mumbai 400 055 (IN) |
SINGH, Girij, Pal; (IN).
SRIVASTAVA, Dhananjai; (IN).
MEHARE, Kishor, Gulabrao; (IN).
MALIK, Vineet; (IN).
DEOKAR, Sharad, Chandrabhan; (IN).
DANGE, Abhijeet, Avinash; (IN)


SUCCESS QUOTIENT: Lupin chairman DB Gupta (sitting) with managing director Kamal K Sharma (centre), directors Vinita Gupta (right) and Nilesh Gupta.
The present invention provides a novel process for preparation N-[(2,3,4,5,6- Pentafluorophenoxy)phenoxyphosphinyl]-L-alanine 1-methylethyl ester (formula 2) and resolving the formula 2 in the presence base to form N-[(S)-(2,3,4,5,6- Pentafluorophenoxy)phenoxyphosphinyl]-L-alanine 1-methylethyl ester (formula 2′).
Sofosbuvir is chemically named as (S)-isopropyl 2-((S)-(((2R,3R,4R,5R)-5-(2,4- dioxo3,4-dihydropyrimidin-l(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran- 2yl)methoxy)-(phenoxy)phosphorylamino)propanoate and is represented by the following chemical structure:

Formula 1
PCT publications WO2011123645 and WO2010135569 describes process for preparation of compound of formula 2′ by reacting isopropyl (chloro(phenoxy)phosphoryl)-L-alaninate and pentaflurophenol in the presence of base.

Formula 2′
Example-1:
Preparation of sodium 2,3,4,5,6-pentaflurophenolate using sodium hydride
10.2g of sodium hydride was dissolved in 100 ml anhydrous THF. This solution was slowly added to a solution of pentafluorophenol (50g) in THF (100ml), Reaction mass was stirred for 60-120 min at 25-30°C. Reaction mass was distilled under reduced pressure, obtained solid was dried under vacuum at 45-50°C (yield=55g, confirmed by IR)
Example-2:
Preparation of sodium 2,3,4,5,6-pentaflurophenolate using sodium methoxide
2,3,4,5, 6-pentafluorophenol (lOg) was dissolved in methanol (100ml), solution was cooled to 5-10°C. To this was added a solution of sodium methoxide in methanol. The reaction mass was stirred for 60-120 min at 25-30°C. Reaction mass was distilled under reduced pressure, obtained residue was striped with toluene. Obtained solid was dried under vacuum at 45-50°C (yield=l lg)
Example 3:
Preparation of sodium 2,3,4,5,6-pentaflurophenolate using sodium hydroxide
2,3,4,5, 6-pentafluorophenol (lOOg) dissolved in methanol (—ml), solution was cooled to 5-10°C. To this was added a solution of sodium hydroxide (— g) in methanol. The reaction mass was stirred for 60-120 min at 25-30°C. Reaction mass was distilled under reduced pressure, obtained residue was striped with dichloromethane. Obtained solid was dried under vacuum at 45-50°C (yield=— g)
Example 4:
Preparation of (2S)-isopropyl-2-((chloro(phenoxy)posphoryl)amino)propanoate:
phenyl phosphodichloridate (30.6g) was dissolved in dichloromethane , to this was added a solution of 1-alanine isopropyl ester free base (19.16g) in dichloromethane at-60°C under nitrogen. Solution of triethylamine (20.7ml) was added to above reaction mass. Reaction mass was stirredat -60°C for 30 min and then temperature was raised to 25 °C. Reaction mass was stirred at 20-25 °C for 60 min & filtered and washed with dichloromethane. Clear filtrate was distilled under reduced pressure obtained residue was stirred with diisopropyl ether & filtered. Clear filtrate was distilled under reduced pressure to get (2S)-isopropyl-2-((chloro(phenoxy)posphoryl)amino)propanoate compound.
Example 5:
Preparation of isopropyl ((perfluorophenoxy)(phenoxy)phosphoryl)-L-alaninate (formula 2):

(Formula 2)
Obtained (2S)-isopropyl-2-((chloro(phenoxy)phosphoryl)amino)propanoate (1.2 mol eq.) was dissolved in dichloromethane and cooled to 0-5°C under nitrogen atmosphere. To this was added solution of sodium 2,3,4,5,6-pentaflurophemolate (1 mol eq.) in tetrahydrofuran . Temperature of reaction mass was raised to 25°C and reaction mass was stirred for 3 hrs. After completion of reaction, reaction mass was distilled under reduced pressure & obtained residue was dissolved I ethyl acetate. Ethyl acetate layer was washed with water, dried over sodium sulfate & distilled off under reduced pressure. Diisopropyl ether was added to obtained residue and stirred for 60 min at 25 °C, obtained mass was filtered & washed with diisopropyl ether. Solid product was dried under vacuum at 40-45 °C .(yield=20g, enantiomer purity=93.45%)
Example 6:
Preparation of (S)-isopropyl 2-(((S)- (perfluorophenoxy)phenoxy)phosphoyl)amino)propanoate (Formula 2′):

Formula 2′
(2S)-isopropyl-2-((chloro(phenoxy)phosphoryl)amino)propanoate (1.2 mol eq.) was dissolved in tetrahydrofuran (3.5 volumes). The reaction mass was cooled to -10°C. Solution of sodium salt of pentafluorophenol (1 mol eq.) in tetrahydrofuran (3.5 volumes) was added dropwise to the reaction mass at -10°C. After completion of the reaction solvent was distilled off. Ethyl acetate and water were added to the reaction mass. Reaction mass was stirred, ethyl acetate layer was separated and washed with sodium bicarbonate solution and brine. Ethyl acetate layer was concentrated under reduced pressure. Reaction mass was stripped with n-hepatane to get crude product. Crude product was dissolved in Methyl tert-butyl ether and n-heptane (1 : 1 ratio). The pH of reaction mass was adjusted to pH 8 by using triethylamine. Reaction mass was stirred overnight. Solid mass was filtered and washed with a mixture of methyl tertiary-butyl ether: n-heptane (1 : 1). The obtained product was dissolved in ethyl-acetate and washed with water and 20% brine solution. Ethyl acetate layer was separated; solvent was distilled off under reduced pressure. Reaction mass was stripped with diisopropyl ether. Di-isopropyl ether was added to the reaction mass. Reaction mass was stirred at 45-50°C. Reaction mass was cooled to 5-10°C and stirred. The titled compound was isolated by filtration and washed with di-isopropyl ether. The titled compound was dried under reduced pressure at 40°C. Yield 66.81%.

Vinita Gupta, CEO, Lupin Pharmaceuticals Inc

Desh Bhandu Gupta- Founder and chairman of Lupin Limited
////////////LUPIN LIMITED, WO 2016181313, NEW PATENT, SOFOSBUVIR
LUPIN, SOFOSBUVIR, NEW PATENT, WO 2016016865


(WO2016016865) A PROCESS FOR THE PREPARATION OF NUCLEOSIDE PHOSPHORAMIDATE
LUPIN LIMITED [IN/IN]; 159 CST Road, Kalina, Santacruz (East), State of Maharashtra, Mumbai 400 098 (IN)
ROY, Bhairab, Nath; (IN).
SINGH, Girij, Pal; (IN).
SHRIVASTAVA, Dhananjai; (IN).
MEHARE, Kishor, Gulabrao; (IN).
MALIK, Vineet; (IN).
DEOKAR, Sharad, Chandrabhan; (IN).
DANGE, Abhijeet, Avinash; (IN)
The present invention pertains to process for preparing nucleoside phosphoramidates and their intermediates. Phosphoramidates are inhibitors of RNA-dependent RNA viral replication and are useful as inhibitors of HCV NS5B polymerase, as inhibitors of HCV replication and for treatment of hepatitis C infection in mammals. One of the recently approved phosphoramidate by USFDA is Sofosbuvir [1190307-88-0]. Sofosbuvir is a component of the first all-oral, interferon-free regimen approved for treating chronic hepatitis C. The present invention provides novel intermediate, its process for preparation and use for the preparation of Sofosbuvir. The present invention also gives one pot process for preparation of Sofosbuvir.
Hepatitis C virus (HCV) infection is a major health problem that leads to chronic liver disease, such as cirrhosis and hepatocellular carcinoma, in a substantial number of infected individuals. There are limited treatment options for individuals infected with hepatitis C virus. The current approved therapeutic option is the use of immunotherapy with recombinant interferon- [alpha] alone or in combination with the nucleoside analog ribavirin.
US 7964580 (‘580) is directed towards novel nucleoside phosphoramidate prodrug for the treatment of hepatitis C virus infection.
US’580 patent claims Sofosbuvir and rocess for preparation of Sofosbuvir of Formula 1.

Formula 1
Process for preparation of Sofosbuvir as per US ‘580 patent involve reaction of compound of Formula 4″ with a nucleoside 5’

Compound 4″ nucleoside 5′
Wherein X’ is a leaving group, such as CI, Br, I, tosylate, mesylate, trifluoroacetate, trifluroslfonate, pentafluorophenoxide, p-nitro-phenoxide.
Objects of the invention
The object of the present invention is to provide a novel intermediate of Formula 2

Formula 2
wherein X’ is a leaving group selected from 1-hydroxybenzotriazole, 5-(Difluoromethoxy)-lH-benzimidazole-2-thiol, 2-Mercapto-5-methoxybenzimidazole, cyanuric acid, 2-oxazolidinone, 2-Hydroxy Pyridine. The above leaving group can be optionally substituted with n-alkyl, branched alkyl, substituted alkyl; cycloalkyl; halogen; nitro; or aryl, which includes, but not limited to, phenyl or naphthyl, where phenyl or naphthyl are further optionally substituted with at least one of Ci-C6 alkyl, C2-C6 alkenyl, C2-C6 alkynyl, Ci-C6 alkoxy, F, CI, Br, I, nitro, cyano, Ci-C6 haloalkyl, -N(Rr)2, Ci-C6 acylamino, -NHS02Ci-C6 alkyl, -S02N(Rr)2, COR1″, and -S02Ci-C6 alkyl; (Rr is independently hydrogen or alkyl, which includes, but is not limited to, Ci-C2o alkyl, Ci-Cio alkyl, or Ci-C6 alkyl, R1” is -OR1 or -N(Rr)2).
Another object of the present invention is to provide a process to prepare the intermediate of Formula 2.
Another object of the present invention is use of the intermediate of Formula 2 in the preparation of Sofosbuvir of Formula 1.

Formula 1

Example 1:
Process for the preparation of S-oxazolidinone derivative of Formula 2

Step-1 Preparation of phosphorochloridate solution:
Dichloromethane (DCM 400ml) was charged in round bottom flask flushed with nitrogen. Phenyl phosphodichloridate (18.30ml) was added in one portion in the flask. The flask was cooled to -60°-70°C with a dry ice-acetone bath. Solution of L-alanine isopropyl ester hydrochloride (20.6gm)) in DCM (50ml) was added to the reaction flask. To this was added a solution of triethylamine (11.20ml) in MDC (100 ml) was added over a course of 60 minutes, while maintaining internal temperature below -70 °C throughout the addition. After completion of reaction, temperature of reaction mass was raised to room temperature.
100ml THF was charged in another round bottom flask flushed with nitrogen followed by the addition of S-4-phenyloxazolidnone (lOgm). Triethyl-amine (11.2ml) & LiCl (2.85gm) were added to the above flask. The reaction mass was stirred for 15-30 min at room temperature and was cooled to 0-5 °C. Phosphorochloridate solution from step-1 was added drop- wise to the reaction flask in 15-45 min maintaining reaction temperature at 0-5 °C. The reaction mass was stirred for 30-60min at 0°-5°C. The reaction progress was monitored on thin layer chromatography. After completion of the reaction, the reaction temperature was raised to room temperature. Agitation was resumed for an additional 30min. The reaction mass was filtered and concentrated under reduced pressure. To this was added diisopropyl ether (400ml) and aqueous saturated ammonium chloride solution and reaction mass was stirred for 10-15 minutes. Organic layer was separated and was washed with water (100ml) & dried over sodium sulfate and concentrated under vacuum. Cyclohexane (50ml) was charged to the obtained oily mass and reaction mass was stirred till solid precipitated out. Solid was filtered and washed with cyclohexane and dried under vacuum (8.80gm MP 56.5°-56.6°C). The obtained product was characterized by mass, NMR & IR. 1H NMR (DMSO-d6) δ 1.142 -1.18
(m, 9H), 3.85-3.92 (m, 1H), 4.72-4.89(m, 2H), 5.31-5.32(d, 1H), 6.25-6.3 (m, 1H), 6.95-7.31 (m, 10H); MS, m/e 433 (M+l) +
Example 2: Process for the preparation of 2-hydroxy pyridine derivatives of formula 2:

Anhydrous dichloromethane (DCM) 700ml was charged in round bottom flask flushed with nitrogen. The flask was cooled to -60° to -70°C in a dry ice acetone bath. Phenyl phosphodichloridate (76.04 gm) was added in one portion in the flask at -65°C. Solution of L-alanine isopropyl ester hydrochloride (60.56 gm) in DCM (50 ml) was added to the reaction mass. Solution of triethylamine (72.44gm) in DCM (50ml) was added to the reaction mass over a course of 60 minutes, while maintaining internal temperature below -70°C throughout the addition. The resulting white slurry was agitated for additional 60 minutes. Then the temperature of reaction mass was raised to room temperature. Reaction mass was stirred for 60 min & TLC was checked. Reaction mass was filtered and rinsed with anhydrous dichloromethane (2 XI 00 mL). The filtrate was concentrate under vacuum to 20 V and reaction mass was filtered, washed with DCM (15ml). The filtrate was transferred to RBF. The reaction mass was cooled to 0°-10°C. A solution of 2-hydroxy-3-nitro-5- (trifluoromethyl) pyridine (15.gm) in DCM (100ml) & triethyl amine (21.89gm) was added to the reaction mass. Temperature of reaction mass was raised to 20-30°C. Reaction mass was stirred overnight. Reaction was monitored using TLC. After completion, the reaction mass was filtered and washed with DCM (30ml). Filtrate was washed with water (150 ml x 2). Organic layer was concentrated under vacuum and degased. Diisopropyl ether (200ml) was charged to reaction mass and reaction mass was stirred for 15 minutes , filtered and washed with methyl ter-butyl ether (MTBE 30ml). Filtrate was concentrated under vacuum and dried. (8.68gm, MP-125.5°-131.5°C). Obtained compound was characterized by Mass, NMR & IR. 1H NMR (DMSO-d6) δ 1.07 -1.27 (m, 9H), 4.04-4. l l(m, 1H), 4.73-4.79(m, 1H), 6.76-7.43 (m, 5H), 9.00-9.02 (d, 2H); MS, m/e 478 (M+l) +; FTIR, 1203, 1409, 1580, 1732, 3217.
Other 2-hydroxy pyridine derivatives of Formula 2 were prepared by following the process disclosed in example 2-
2-Hydroxy-5-fluoropyridine derivative of Formula 2;-1H NMR (DMSO-d6) δ 1.09 -1.23 (m, 9H), 3.02-3.06 (m, lH), 3.85-4.01 (m,lH), 4.79-4.87(m, 1H), 6.4-6.52 (m,lH), 7.10-7.89 (m,6H); MS, m/e 383 (M+l) +,
2-Hydroxy-5-nitropyridine derivative of Formula 2:- 1H NMR (DMSO-d6) δ 1.06 -1.22 (m, 9H),4.0-4.02 (m,lH), 4.7-4.8(m,lH), 6.5-6.6 (m,lH),7.12-7.42 (m,6H),8.66-8.68 (d, lH),9.07-9.13(d,lH); MS, m/e 410 (M+l) +
2-Hydroxy-3, 5-dinitropyridine derivative of Formula 2:- 1H NMR (DMSO-d6) δ 1.11 -1.24 (m, 9H), 3.04-3.09(m,lH), 4.8-4.86(m,lH), 7.09-7.39 (m,5H),8.97-9.06 (d,2H)
Example 3: Process for the preparation of Sofosbuvir by coupling of isopropyl(((3-nitro-5-(trifluromethyl)pyridin-2-yl)oxy)phenoxy)phosphoryl-L-alaninate with 1-((2R,3R,4R,5R)-3-fluoro-4-hydroxy-5-(hydroxymethyl)-3-methyltetrahydrofuran-2-yl)pyrimidine-2,4(lH,3H)-dione :

To a solution of l-((2R,3R,4R,5R)-3-fluoro-4-hydroxy-5-(hydroxymethyl)-3-methyltetrahydrofuran-2-yl)pyrimidine-2,4(lH,3H)-dione (0.2gm) in THF (4 ml), tert- butylmagnesium chloride (0.80ml, 1.7 M solution in THF) was added dropwise at room temperature and reaction mass was stirred for 30 minutes. A solution of pyridine derivative from example 2 (0.36gm) in THF (4ml) was added dropwise to the reaction mass at room temperature. Completion of reaction was monitored using TLC. After completion of reaction, reaction mass was quenched by using saturated ammonium chloride solution (10ml). Reaction mass was extracted with ethyl acetate (50ml). Organic layer was separated, dried over magnesium sulfate and concentrated under vacuum. The resulting residue was purified by column chromatography on silica gel & obtained solid product was characterized. MS, m/e 530.2 (M+l) +.
/////////LUPIN, SOFOSBUVIR, NEW PATENT, WO 2016016865
SOFOSBUVIR HPLC
SEE http://www.google.co.in/patents/US20100298257
Example 28
-
Chemical Purity Determination by HPLC
-
Various HPLC conditions can be used to determine the chemical purity of the compounds disclosed herein. One such example is disclosed above in relation to the thermodynamic aqueous solubility studies. Another example is disclosed below.
-
HPLC Conditions:
-
- LC: Waters Alliance 2695 Separations Module, Waters 2996 PDA detector and Waters Empower 2 Software (Version 6.00)
- Column: Phenomenex Luna C18(2); 4.6×50 mm; 3 μm
- Flow rate: 1.2 mL/min
- Injection Volume: 10 μL
- Mobile phase: Solvent A: 95% Water with 5% Methanol and 10 mM Ammonium Acetate; pH˜5.3
- Gradient Solvent B: MeOH with 10 mM Ammonium Acetate hold at 0% B 3 min
- 0-47% B 3-4 min
- hold at 47% B 4-10 min
- 47%-74% B 10-11 min
- hold at 74% B 11-13.5 min
- return to 0% B 13.5-13.6 min
- hold at 0% B 13.6-15.5 min
-
Under these conditions, the purity of 4, RP-4, and SP-4 was determined to be ˜99.6, ˜99%, and ˜99.5%, respectively. It is noted that higher purities can be realized by optimizing the methods disclosed above.
-
Inspection of the XRPD diffractograms shows that the two crystalline single diastereoisomers gave clearly different XRPD patterns. Additionally, there was a clear difference in the melting point of the two crystalline diastereoisomers, with RP-4 having a considerably higher onset than SP-4 (136° C. vs. 94° C.).
-
Example 29Additional Separation Methods
-
The following SFC separation (conditions listed below) yielded adequate separation of a mixture of the diastereomers, RP-4 and SP-4.
-
Preparative Method: Analytical Method: Chiralpak AS-H (2 × 25 cm) SN# 07-8656 Chiralpak AS-H (25 × 0.46 cm) 20% methanol/CO2 (100 bar) 20% methanol/CO2 (100 bar) 50 ml/min, 220 nm. 3 ml/min, 220 nm. Conc.: 260 mg/30 ml methanol, inj vol.: 1.5 ml -
The following SFC separation (conditions listed below) yielded adequate separation of a mixture of the diastereomers, RP-4 and SP-4.
-
Preparative Method: Analytical Method: Chiralpak IA(2 × 15 cm) 802091 Chiralpak IA(15 × 0.46 cm) 30% isopropanol(0.1% DEA)/CO2, 40% methanol(DEA)/CO2, 100 bar 100 bar 60 mL/min, 220 nm. 3 mL/min, 220 nm. inj vol.: 2 mL, 20 mg/mL methanol -
TABLE 16 Summary of results from the batch characterization of RP-4, 4, and SP-4. Analysis RP-4 4 SP-4 Proton NMR Single diastereoisomer 1:1 Mixture of Single diastereoisomer diastereoisomers XRPD Crystalline – different Amorphous Crystalline – different DSC from SP-4 Endotherm; 59° C. from RP-4 Endotherm; melt – 136° C. Endotherm; melt – 94° C. TGA No wt loss, No wt loss, decomposition No wt loss, decomposition >240° C. >240° C. decomposition >240° C. IR See above See above See above Aq Solubility 1.58 6.11 5.65 (mg · ml−1) HPLC Purity 96.9% 99.6% 99.5% 40° C./75% RH No form change Deliquescence inside 1.5 h Deliquescence inside 4.5 h 25° C./53% RH — Deliquescence No form change GVS Non-hygroscopic up to 90% — Non-hygroscopic up to 60% RH RH
- Example 27Thermodynamic Aqueous Solubility
-
Aqueous solubility was determined by suspending a sufficient amount of compound in water to give a maximum final concentration of ≧10 mg.ml−1 of the parent free-form of the compound. The suspension was equilibrated at 25° C. for 24 hours then the pH was measured. The suspension was then filtered through a glass fiber C filter into a 96 well plate. The filtrate was then diluted by a factor of 101. Quantitation was by HPLC with reference to a standard solution of approximately 0.1 mg.ml−1 in DMSO. Different volumes of the standard, diluted and undiluted sample solutions were injected. The solubility was calculated using the peak areas determined by integration of the peak found at the same retention time as the principal peak in the standard injection.
-
TABLE 14 HPLC Method Parameters for Solubility Measurements Type of method: Reverse phase with gradient elution Column: Phenomenex Luna, C18 (2) 5 μm 50 × 4.6 mm Column Temperature 25 (° C.): Standard Injections (μl): 1, 2, 3, 5, 7, 10 Test Injections (μl): 1, 2, 3, 10, 20, 50 Detection: 260, 80 Wavelength, Bandwidth (nm): Flow Rate (ml · min−1): 2 Phase A: 0.1% TFA in water Phase B: 0.085% TFA in acetonitrile Time (min) % Phase A % Phase B Timetable: 0.0 95 5 1.0 80 20 2.3 5 95 3.3 5 95 3.5 95 5 4.4 95 5 -
[0306]Analysis was performed under the above-noted conditions on an Agilent HP1100 series system equipped with a diode array detector and using ChemStation software vB.02.01-SR1.
-
TABLE 15 Aqueous solubility result for RP-4, 4, and SP-4. pH of Unfiltered Sample ID mixture Solubility/mg · ml−1 Comments RP-4 7.12 1.58 Suspension 4 7.03 6.11 Residual solid SP-4 6.88 5.65 Residual solid
FIG 1
Chemical structures of RBV, BOC, TVR, and VRT-127394. Shown are the chemical structures of the anti-HCV drugs RBV {1-[(2R,3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]-1,2,4-triazole-3-carboxamide)} (A), BOC {(1R,2S,5S)-N-(4-amino-1-cyclobutyl-3,4-dioxobutan-2-yl)-3-[(2S)-2(tertbutylcarbamoylamino)-3,3-dimethylbutanoyl]-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxamide} (B), TVR {(3S,3aS,6aR)-2-[(2S)-2-[[(2S)-2-cyclohexyl-2-(pyrazine-2-carbonylamino)acetyl]amino]-3,3-dimethylbutanoyl]-N-[(3S)-1-(cyclopropylamino)-1, 2-dioxohexan-3-yl]-3,3a,4,5,6,6a-hexahydro-1H-cyclopenta[c]pyrrole-3-carboxamide} (C), and VRT-127394 (R diastereoisomer of TVR) (D).
Blank plasma samples used for matrix effect (ME) assessment and for the preparation of calibration and control samples were obtained from citrated blood (1,850 × g, 10 min, +4°C, Beckman J6B centrifuge) collected from Vaquez disease patients on the occasion of their regular phlebotomy.
The blank plasma used for the preparation of the calibration and quality control (QC) samples was acidified with 10% FA (50 μl of 10% FA added to 950 μl of plasma). The acidification of plasma aims at preventing the conversion of TVR to its epimer VRT-127394 that occurs in vivo and in vitro. (Tibotec-Janssen, personal communication).
Equipment.The LC system used consisted of Rheos Allegro quaternary pumps equipped with an online degasser and an HTS PAL autosampler (CTC Analytics AG, Zwingen, Switzerland) controlled by Janeiro-CNS 1.1 software (Flux Instruments AG, Thermo Fischer Scientific Inc., Waltham, MA). Separations were done on a Hypercarb 3-μm column (2.1 mm ID by 100 mm; Thermo Fischer Scientific) placed in a column oven thermostat regulated at +80°C (HotDog 5090; ProLab GmbH, Reinach, Switzerland). The chromatographic system was coupled to a triple-stage quadrupole quantum mass spectrometer (Thermo Fischer Scientific) equipped with an electrospray ionization (ESI) Ion Max interface and operated with the Xcalibur software package (version 2.0; Thermo Fischer Scientific).
READ
http://www.us.edu.pl/uniwersytet/jednostki/wydzialy/chemia/acta/ac14/zrodla/14_AC14.pdf
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2291777/
UPDATE………….DEC2015

SOFOSBUVIR
NEW PATENT WO2015188782,
(WO2015188782) METHOD FOR PREPARING SOFOSBUVIR
CHIA TAI TIANQING PHARMACEUTICAL GROUP CO., LTD [CN/CN]; No. 8 Julong North Rd., Xinpu District Lianyungang, Jiangsu 222006 (CN)
Sofosbuvir synthesis routes currently used include the following two methods:
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2015188782&redirectedID=true


![]()
//////
////
Gilead’s HCV drug Sovaldi gets Europe OK
Gilead Sciences’ closely-watched hepatitis C drug Sovaldi has been given the green light in Europe.
The European Commission has granted marketing authorisation for Sovaldi (sofosbuvir) 400mg tablets
which, as part of HCV combination therapy with peg-interferon and ribavirin, offers cure rates of around 90% in previously-untreated adults. However, most significant is that the once-daily nucleotide analogue polymerase inhibitor is the first all-oral treatment option for up to 24 weeks for patients unsuitable for interferon.
Read more at: http://www.pharmatimes.com/Article/14-01-20/Gilead_s_HCV_drug_Sovaldi_gets_Europe_OK.aspx#ixzz2qwHI3iJi
SYNTHESIS
-
sofosbuvir » All About Drugs
http://www.allfordrugs.com/tag/sofosbuvir/ALL ABOUT DRUGS BY DR ANTHONY MELVIN CRASTO, WORLD DRUG TRACKER HELPING … US Approves Breakthrough Hepatitis C Drug,Sofosbuvir.
US Approves Breakthrough Hepatitis C Drug, Sofosbuvir » All About Drugs
SOFOSBUVIR
DO NOT FORGET TO CLICK
US Approves Breakthrough Hepatitis C Drug, Sofosbuvir » All About Drugs
AND ALSO
DO NOT FORGET TO CLICK
SEE………………….http://orgspectroscopyint.blogspot.in/2015/02/sofosbuvir-visited.html
READ ABOUT SYNTHESIS BY CLICKING ABOVE LINK
Sofosbuvir
Sovaldi
M.Wt: 529.45
Formula: C22H29FN3O9P
Isopropyl (2S)-2-[[[(2R,3R,4R,5R)-5-(2,4-dioxopyrimidin-1-yl)-4-fluoro-3-hydroxy-4-methyl-tetrahydrofuran-2-yl]methoxy-phenoxy-phosphoryl]amino]propanoate
A prodrug of 2′-deoxy-2′-alpha-F-2′-beta-C-methyluridine 5′-monophosphate.
GS-7977, PSI-7977
- GS 7977
- GS-7977
- PSI 7977
- PSI-7977
- Sofosbuvir
- Sovaldi
- UNII-WJ6CA3ZU8B
CAS Registry Number :1190307 -88-0
http://www.ama-assn.org/resources/doc/usan/sofosbuvir.pdf
Indications: Chronic hepatitis C (HCV GT1, GT2, GT3, GT4)
Mechanism: nucleoside NS5B polymerase inhibitor
approved Time: December 6, 2013
,U.S. Patent Number: 7964580,8415322,8334270,7429572;, patent validity: March 26, 2029 (U.S. Patent No.: 7,964,580 and 8,334,270), April 3, 2025 (U.S. Patent No.: 7,429,572 and 8,415,322)
US patent number 7964580, US patent number 8415322, US patent number 8334270,US patent number 7429572 Patent Expiration Date: March 26, 2029 for US patent number 7964580 and 8334270 (2028 in EU); April 3, 2025 for US patent number 7429572 and 8415322
Sales value (estimated): $ 1.9 billion (2014), 6600000000 USD (2016)
Drug Companies: Gilead Sciences, Inc. (Gilead Sciences)
WASHINGTON, Dec. 6, 2013 (AP) — Federal health officials have approved a highly anticipated hepatitis C drug from Gilead Sciences Inc. that is expected to offer a faster, more palatable cure to millions of people infected with the liver-destroying virus.
The Food and Drug Administration said Friday it approved the pill Sovaldi in combination with older drugs to treat the main forms of hepatitis C that affect U.S. patients.
Current treatments for hepatitis C can take up to a year of therapy and involve weekly injections of a drug that causes flu-like side effects. That approach only cures about three out of four patients. Sovaldi is a daily pill that in clinical trials cured roughly 90 percent of patients in just 12 weeks, when combined with the older drug cocktail.http://www.pharmalive.com/us-approves-breakthrough-hepatitis-c-drug
- The end of October 2013 saw a nod from the FDA given to Gilead’s New Drug Application for Sofosbuvir, a much needed treatment for hepatitis C.
- As a nucleotide analogue, Sofosbuvir is designed as a once daily treatment.
- There are roughly 170 million cases of hepatitis C around the world.
- A report in the Journal of the American Medical Association on August 28, 2013 revealed that the Sofosbuvir and Ribavirin combination treatment effectively cured many patients with the Hepatitis C Virus.
- The Sofosbuvir and Ribavirin drug combination was void of interferon-based treatments, which many patients are resistant too.
- More than 3 million Americans have chronic Hepatitis C Virus, and 22 percent of these patients are African American.
Sofosbuvir (brand names Sovaldi and Virunon) is a drug used for hepatitis C virus (HCV) infection, with a high cure rate.[1][2] It inhibits the RNA polymerase that the hepatitis C virus uses to replicate its RNA. It was discovered at Pharmasset and developed by Gilead Sciences.[3]
Sofosbuvir is a component of the first all-oral, interferon-free regimen approved for treating chronic Hepatitis C.[4]
In 2013, the FDA approved sofosbuvir in combination with ribavirin (RBV) for oral dual therapy of HCV genotypes 2 and 3, and for triple therapy with injected pegylated interferon (pegIFN) and RBV for treatment-naive patients with HCV genotypes 1 and 4.[4] Sofosbuvir treatment regimens last 12 weeks for genotypes 1, 2 and 4, compared to 24 weeks for treatment of genotype 3. The label furhter states that sofosbuvir in combination with ribavirin may be considered for patients infected with genotype 1 who are interferon-ineligible.[5] Sofosbuvir will cost $84,000 for 12 weeks of treatment and $168,000 for the 24 weeks, which some patient advocates have criticized as unaffordable.
Interferon-free therapy for treatment of hepatitis C eliminates the substantial side-effects associated with use of interferon. Up to half of hepatitis C patients cannot tolerate the use of interferon.[6]
Sofosbuvir is a prodrug that is metabolized to the active antiviral agent 2′-deoxy-2′-α-fluoro-β-C-methyluridine-5′-triphosphate.[7] Sofosbuvir is anucleotide analog inhibitor of the hepatitis C virus (HCV) polymerase.[8] The HCV polymerase or NS5B protein is a RNA-dependent RNA polymerase critical for the viral cycle.
The New Drug Application for Sofosbuvir was submitted on April 8, 2013 and received the FDA’s Breakthrough Therapy Designation, which grants priority review status to drug candidates that may offer major treatment advantages over existing options.[9]
On 6th December 2013, the U.S. Food and Drug Administration approved sofosbuvir for the treatment of chronic hepatitis C.[10]
Sofosbuvir is being studied in combination with pegylated interferon and ribavirin, with ribavirin alone, and with other direct-acting antiviral agents.[11][12] It has shown clinical efficacy when used either with pegylated interferon/ribavirin or in interferon-free combinations. In particular, combinations of sofosbuvir with NS5A inhibitors, such as daclatasvir or GS-5885, have shown sustained virological response rates of up to 100% in people infected with HCV.[13]
Data from the ELECTRON trial showed that a dual interferon-free regimen of sofosbuvir plus ribavirin produced a 24-week post-treatment sustained virological response (SVR24) rate of 100% for previously untreated patients with HCV genotypes 2 or 3.[14][15]
Data presented at the 20th Conference on Retroviruses and Opportunistic Infections in March 2013 showed that a triple regimen of sofosbuvir, ledipasvir, and ribavirin produced a 12-week post-treatment sustained virological response (SVR12) rate of 100% for both treatment-naive patients and prior non-responders with HCV genotype 1.[16] Gilead has developed a sofosbuvir + ledipasvir coformulation that is being tested with and without ribavirin.
Sofosbuvir will cost $84,000 for 12 weeks of treatment used for genotype 1 and 2, and $168,000 for the 24 weeks used for genotype 3.[17] This represents a substantial pricing increase from previous treatments consisting of interferon and ribavirin, which cost between $15,000 and $20,000.[18] The price is also significantly higher than that of Johnson & Johnson‘s recently approved drug simeprevir (Olysio), which costs $50,000 and also treats chronic hepatitis C.[18] The high cost of the drug has resulted in a push back from insurance companies and the like, includingExpress Scripts, which has threatened to substitute lower priced competitors, even if those therapies come with a more unfriendly dosing schedule.[18] Other treatments that have recently entered the market have not matched the efficacy of sofosbuvir, however, allowing Gilead to set a higher price until additional competition enters the market.[18] Patient advocates such as Doctors Without Borders have complained about the price, which is particularly difficult for underdeveloped countries to afford.[19]
sofosbuvir
- News: United States to approve potent oral drugs for hepatitis C, Sara Reardon, Nature, 30 October 2013
- Sofia MJ, Bao D, Chang W, Du J, Nagarathnam D, Rachakonda S, Reddy PG, Ross BS, Wang P, Zhang HR, Bansal S, Espiritu C, Keilman M, Lam AM, Steuer HM, Niu C, Otto MJ, Furman PA (October 2010). “Discovery of a β-d-2′-deoxy-2′-α-fluoro-2′-β-C-methyluridine nucleotide prodrug (PSI-7977) for the treatment of hepatitis C virus”. J. Med. Chem. 53 (19): 7202–18.doi:10.1021/jm100863x. PMID 20845908.
- “PSI-7977”. Gilead Sciences.
- Tucker M (December 6, 2013). “FDA Approves ‘Game Changer’ Hepatitis C Drug Sofosbuvir”. Medscape.
- “U.S. Food and Drug Administration Approves Gilead’s Sovaldi™ (Sofosbuvir) for the Treatment of Chronic Hepatitis C – See more at: http://www.gilead.com/news/press-releases/2013/12/us-food-and-drug-administration-approves-gileads-sovaldi-sofosbuvir-for-the-treatment-of-chronic-hepatitis-c#sthash.T9uTbSWK.dpuf”. Gilead. December 6, 2013.
- “Sofosbuvir is safer than interferon for hepatitis C patients, say scientists”. News Medical. April 25, 2013.
- Murakami E, Tolstykh T, Bao H, Niu C, Steuer HM, Bao D, Chang W, Espiritu C, Bansal S, Lam AM, Otto MJ, Sofia MJ, Furman PA (November 2010). “Mechanism of activation of PSI-7851 and its diastereoisomer PSI-7977”. J. Biol. Chem. 285 (45): 34337–47.doi:10.1074/jbc.M110.161802. PMC 2966047. PMID 20801890.
- Alejandro Soza (November 11, 2012). “Sofosbuvir”. Hepaton.
- “FDA Advisory Committee Supports Approval of Gilead’s Sofosbuvir for Chronic Hepatitis C Infection”. Drugs.com. October 25, 2013.
- “FDA approves Sovaldi for chronic hepatitis C”. FDA New Release. U.S. Food and Drug Administration. 2013-12-06.
- Murphy T (November 21, 2011). “Gilead Sciences to buy Pharmasset for $11 billion”.Bloomberg Businessweek.
- Asselah T (January 2014). “Sofosbuvir for the treatment of hepatitis C virus”. Expert Opin Pharmacother 15 (1): 121–30. doi:10.1517/14656566.2014.857656. PMID 24289735.
- “AASLD 2012: Sofosbuvir and daclatasvir dual regimen cures most people with HCV genotypes 1, 2, or 3”. News. European Liver Patients Association. 2012-11-21.
- AASLD: PSI-7977 plus Ribavirin Can Cure Hepatitis C in 12 Weeks without Interferon. Highleyman, L. HIVandHepatitis.com. 8 November 2011.
- Gane EJ, Stedman CA, Hyland RH, Ding X, Svarovskaia E, Symonds WT, Hindes RG, Berrey MM (January 2013). “Nucleotide polymerase inhibitor sofosbuvir plus ribavirin for hepatitis C”.N. Engl. J. Med. 368 (1): 34–44. doi:10.1056/NEJMoa1208953. PMID 23281974.
- CROI 2013: Sofosbuvir + Ledipasvir + Ribavirin Combo for HCV Produces 100% Sustained Response. Highleyman, L. HIVandHepatitis.com. 4 March 2013.
- Campbell T (December 11, 2013). “Gilead’s Sofosbuvir Gets New Name, Price, Headaches”. The Motley Fool.
- Cohen, J. (2013). “Advocates Protest the Cost of a Hepatitis C Cure”. Science 342 (6164): 1302–1303. doi:10.1126/science.342.6164.1302. PMID 24337268. edit
The chemical structure
GS-7977, (S)-isopropyl 2-(((S)-(((2R,3R,4R,5R)-5-(2,4-dioxo-3,4- dihydropyrimidin^l(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2- yl)methoxy)(phenoxy)phosphoryl)amino)propanoate, available from Gilead Sciences, Inc., is described and claimed in U.S. Patent No. 7,964,580. (See also US 2010/0016251, US 2010/0298257, US 201 1/0251 152 and US 2012/0107278.) GS-7977 has the structure:
GS-7977 can be crystalline or amorphous. Examples of preparing crystalline and amorphous forms of GS-7977 are disclosed in US 2010/0298257 (US 12/783,680) and US 201 1/0251 152 (US 13/076,552),

Commerically available isopropylidine protected D-glyceraldehyde was reacted with (carbethoxyethylidene)triphenylmethylphosphorane gave the chiral pentenoate ester YP-1. Permanganate dihydroxylation of YP-1 in acetone gave the D-isomer diol YP-2. The cyclic sulfate YP-3 was obtained by first making the cyclic sulfite with thionyl chloride and then oxidizing to cyclic sulfate with sodium hypochlorite. Fluorination of YP-3 with triethylamine-trihydrofluoride(TEA-3HF) in the presence of triethylamine, followed by the hydrolysis of sulfate ester in the presence of concentrated HCl provided diol YP-4 which was benzoylated to give ribonolactone YP-5. Reduction of YP-5 with Red-Al followed by chlorination with sulfuryl chloride in the presence of catalytic amount of tetrabutylammonium bromide yielded YP-6. The conversion of YP-6 to benzoyl protected 2′-deoxyl-2′-alpha-F-2′-Beta-C-methylcytidine (YP-7) was achieved by using O-trimethyl silyl-N4-benzoylcytosine and stannic chloride. Preparation of the uridine nucleoside YP-8 was accomplished by first heating benzoyl cytidine YP-7 in acetic acid then treating with methoanolic ammonia to provide YP-8 in 78% yield.
The phosphoramidating reagent YP-9 was obtained by first reacting phenyldichlorophosphate with L-Alanine isopropyl ester hydrochloride and then with pentafluorophenol. Isolation of single Sp diastereomer YP-9 was achieved via crystallization-induced dynamic resolution in the presence of 20% MTBE/hexane at room temperature.
The uridine nucleoside YP-8 was treated with tert-butylmagnesium chloride in dry THF, followed by pentafluorophenyl Sp diastereomer YP-9 to furnish the Isopropyl (2S)-2-[[[(2R,3R,4R,5R)-5-(2,4-dioxopyrimidin-1-yl)-4-fluoro-3-hydroxy-4-methyl-tetrahydrofuran-2-yl]methoxy-phenoxy-phosphoryl]amino]propanoate (Sovaldi, sofosbuvir, GS-7977, PSI-7977)。
…………
US 7429572
US 8415322
US 7964580
US 8334270B
WO 2006012440
WO 2011123668
……………………………………………
In US 20050009737 published Jan. 13, 2005, J. Clark discloses fluoro-nucleoside derivatives that inhibit Hepatitis C Virus (HCV) NS5B polymerase. In particular, 4-amino-1-((2R,3R,4R,5R)-3-fluoro-4-hydroxy-5-hydroxymethyl-3-methyl-tetrahydro-faran-2-yl)-1H-pyrimidin-2-one (18) was a particularly potent inhibitor of HCV polymerase as well as the polymerase of other Flaviviridae.
In WO2006/012440 published Feb. 2, 2006, P. Wang et al disclose processes for the preparation of 18. Introduction of the cytosine is carried out utilizing the Vorbruggen protocol. In US 20060122146 published Jun. 8, 2006, B.-K. Chun et al. disclose and improved procedures for the preparation of the 2-methyl-2-fluoro-lactone 10. In the latter disclosure the nucleobase is glycosylated by reacting with ribofuranosyl acetate which is prepared by reduction of 10 with LiAlH(O-tert-Bu)3 followed by acetylaton of the intermediate lactol which was treated with an O-trimethylsilyl N4-benzoylcytosine in the presence of SnCl4 to afford the O,O,N-tribenzoylated nucleoside.
……………………………………………………………….
http://www.google.nl/patents/US20080139802
The present process as described in SCHEME A and the following examples contain numerous improvements which have resulted in higher yields of the desired nucleoside. The asymmetric hydroxylation of 22 was discovered to be best carried out with sodium permanganate in the presence of ethylene glycol, sodium bicarbonate in acetone which afforded the diol in 60-64% on pilot plant scale. The sodium permanganate procedure avoids introduction of osmium into the process stream. Further more the stereospecific hydroxylation can be accomplished without using an expensive chiral ligand. The requisite olefin is prepared from (1S,2S)-1,2-bis-((R)-2,2-dimethyl-[1,3]dioxolan-4-yl)-ethane-1,2-diol (20) (C. R. Schmid and J. D. Bryant, Org. Syn. 1995 72:6-13) by oxidative cleavage of the diol and treating the resulting aldehyde with 2-(triphenyl-λ5-phosphanylidene)-propionic acid ethyl ester to afford 22.
(i) NaIO4, NaHCO3, DCM; (ii) MeC(═PPh3)CO2Et; (iii) acetone-NaMnO4 (aq), ethylene glycol, NaHCO3, −10 to 0° C.; aq. NaHSO3 (quench); (iv) i-PrOAc, MeCN, TEA, SOCl2; (v) i-PrOAc, MeCN, NaOCl; (vi) TEA-3HF, TEA; (vii) HCl (aq)-BaCl2-aq; (viii) (PhCO)2O, DMAP, MeCN, (ix) RED-AL/TFE (1:1), DCM; (x) SO2Cl2-TBAB, DCM; (xi) 32, SnCl4-PhCl; (xii) MeOH-MeONa
EXAMPLE 3 (2S,3R)-3-[(4R)-2,2-dimethyl-[1,3]dioxolan-4-yl]-2,3-dihydroxy-2-methyl-propionic acid ethyl ester (24)
A suspension of 22 (10 kg, CAS Reg. No. 81997-76-4), ethylene glycol (11.6 kg), solid NaHCO3 (11.8 kg) and acetone (150 L) is cooled to ca.-15° C. A solution of 36% aqueous NaMnO4 (19.5 kg) is charged slowly (over 4 h) to the suspension maintaining reaction temperature at or below −10° C. After stirring for 0.5 h at −10° C., an aliquot of the reaction mixture (ca. 5 mL) is quenched with 25% aqueous sodium bisulfite (ca. 15 mL). A portion of resulting slurry is filtered and submitted for GC analysis to check the progress of the reaction. When the reaction is complete, the reaction mixture is quenched by slow addition (over 40 min) of cooled (ca. 0° C.) 25% aqueous NaHSO3 (60 L). The temperature of the reaction mixture is allowed to reach 4° C. during the quench. CELITE® (ca. 2.5 kg) is then slurried in acetone (8 kg) and added to the dark brown reaction mixture. The resulting slurry is aged at RT to obtain light tan slurry. The slurry is filtered, and the filter cake is washed with acetone (3×39 kg). The combined filtrate is concentrated by vacuum distillation (vacuum approximately 24 inches of Hg; max pot temperature is 32° C.) to remove the acetone. The aqueous concentrate is extracted with EtOAc (3×27 kg), and the combined organic extracts were washed with water (25 L). The organic phase is then concentrated by atmospheric distillation and EtOAc is replaced with toluene. The volume of the batch is adjusted to ca. 20 L. Heptane (62 kg) is added and the batch cooled to ca. 27° C. to initiate crystallization. The batch is then cooled to −10° C. After aging overnight at −10° C., the product is filtered, washed with 10% toluene in heptane and dried at 50° C. under vacuum to afford 6.91 kg (59.5%) of 24 (CARN 81997-76-4) as a white crystalline solid.
EXAMPLE 4 (3R,4R,5R)-3-Fluoro-4-hydroxy-5-hydroxymethyl-3-methyl-dihydro-furan-2-one (10)
steps 1 & 2—A dry, clean vessel was charged with 24 (6.0 kg), isopropyl acetate (28.0 kg), MeCN (3.8 kg) and TEA (5.4 kg). The mixture was cooled to 5-10° C., and thionyl chloride (3.2 kg) was added slowly while cooling the solution to maintain the temperature below 20° C. The mixture was stirred until no starting material was left (GC analysis). The reaction was typically complete within 30 min after addition is complete. To the mixture was added water (9 kg) and after stirring, the mixture was allowed to settle. The aqueous phase was discarded and the organic phase was washed with a mixture of water (8 kg) and saturated NaHCO3 (4 kg) solution. To the remaining organic phase containing 36 was added MeCN (2.5 kg) and solid NaHCO3 (3.1 kg). The resulting slurry was cooled to ca. 10° C. Bleach (NaOCl solution, 6.89 wt % aqueous solution, 52.4 kg, 2 eq.) was added slowly while cooling to maintain temperature below 25° C. The mixture was aged with stirring over 90-120 min at 20-25° C., until the reaction was complete (GC analysis). After completion of the reaction, the mixture was cooled to ca. 10° C. and then quenched with aqueous Na2SO3 solution (15.1% w/w, 21 kg) while cooling to maintain temperature below 20° C. The quenched reaction mixture was filtered through a cartridge filter to remove inorganic solids. The filtrate was allowed to settle, and phases are separated and the aqueous phase is discarded. The organic layer was washed first with a mixture of water (11 kg) and saturated NaHCO3 solution (4.7 kg), then with of saturated NaHCO3 solution (5.1 kg). DIPEA (220 mL) was added to the organic phase and the resulting solution was filtered through CELITE® (bag filter) into a clean drum. The reactor was rinsed with isopropyl acetate (7 kg) and the rinse is transferred to the drum. The organic phase was then concentrated under vacuum (25-28 inches of Hg) while maintaining reactor jacket temperature at 45-50° C. to afford 26 as an oil (˜10 L). Additional DIPEA (280 mL) was added and the vacuum distillation was continued (jacket temperature 50-55° C.) until no more distillate was collected. (batch volume ca. 7 L).
step 3—To the concentrated oil from step 2 containing 26 was added TEA (2.34 kg) and TEA-trihydrofluoride (1.63 kg). The mixture was heated to 85° C. for 2 h. The batch was sampled to monitor the progress of the reaction by GC. After the reaction was complete conc. HCl (2.35 kg) was added to the mixture and the resulting mixture heated to ca. 90° C. (small amount of distillate collected). The reaction mixture was stirred at ca. 90° C. for 30 min and then saturated aqueous BaCl2solution (18.8 kg) was added. The resulting suspension was stirred at about 90° C. for 4 h. The resulting mixture was then azeotropically dried under a vacuum (9-10 inches of Hg) by adding slowly n-propanol (119 kg) while distilling off the azeotropic mixture (internal batch temperature ca. 85-90° C.). To the residual suspension was added toluene (33 kg) and vacuum distillation was continued to distill off residual n-propanol (and traces of water) to a minimum volume to afford 28.
step 4—To the residue from step 3 containing 28 was added MeCN (35 kg) and ca. 15 L was distilled out under atmospheric pressure. The reaction mixture was cooled to ca. 10° C. and then benzoyl chloride (8.27 kg) and DMAP (0.14 kg) are added. TEA (5.84 kg) was added slowly to the reaction mixture while cooling to maintain temperature below 40° C. The batch was aged at ca. 20° C. and the progress of the benzoylation is monitored by HPLC. After completion of the reaction, EtOAc (30 kg) was added to the mixture and the resulting suspension is stirred for about 30 min. The reaction mixture was filtered through a CELITE® pad (using a nutsche filter) to remove inorganic salts. The solid cake was washed with EtOAc (38 kg). The combined filtrate and washes were washed successively with water (38 kg), saturated NaHCO3 solution (40 kg) and saturated brine (44 kg). The organic phase was polish-filtered (through a cartridge filter) and concentrated under modest vacuum to minimum volume. IPA (77 kg) was added to the concentrate and ca. 25 L of distillate was collected under modest vacuum allowing the internal batch temperature to reach ca. 75° C. at the end of the distillation. The remaining solution was then cooled to ca. 5° C. over 5 h and optionally aged overnight. The precipitate was filtered and washed with of cold (ca. 5° C.) IPA (24 kg). The product was dried under vacuum at 60-70° C. to afford 6.63 kg (70.7% theory of 10 which was 98.2% pure by HPLC.
EXAMPLE 1 Benzoic acid 3-benzoyloxy-5-(4-benzoylamino-2-oxo-2H-pyrimidin-1-yl)-4-fluoro-4-methyl-tetrahydro-furan-2-ylmethyl ester (14)
Trifluoroethanol (4.08 kg) is added slowly to a cold solution (−15° C.) of RED-AL® solution (12.53 kg) and toluene (21.3 kg) while maintaining the reaction temperature at or below −10° C. After warming up to RT (ca. 20° C.), the modified RED-AL reagent mixture (30.1 kg out of the 37.6 kg prepared) is added slowly to a pre-cooled solution (−15° C.) of fluorolactone dibenzoate 10 (10 kg) in DCM (94.7 kg) while maintaining reaction temperature at or below −10° C. After reduction of the lactone (monitored by in-process HPLC), a catalytic amount of tetrabutylammonium bromide (90 g) is added to the reaction mixture. Sulfiiryl chloride (11.86 kg) is then added while maintaining reaction temperature at or below 0° C. The reaction mixture is then heated to 40° C. until formation of the chloride is complete (ca. 4 h) or warmed to RT (20-25° C.) and stirred over night (ca. 16 h). The reaction mixture is cooled to about 0° C., and water (100 L) is added cautiously while maintaining reaction temperature at or below 15° C. The reaction mixture is then stirred at RT for ca. 1 h to ensure hydrolytic decomposition of excess sulfuryl chloride and the phases are separated. The organic layer is washed with a dilute solution of citric acid (prepared by dissolving 15.5 kg of citric acid in 85 L of water) and then with dilute KOH solution (prepared by dissolving 15 kg of 50% KOH in 100 L of water). The organic phase is then concentrated and solvents are replaced with chlorobenzene (2×150 kg) via atmospheric replacement distillation. The resulting solution containing 30 is dried azeotropically.
A suspension of N-benzoyl cytosine (8.85 kg), ammonium sulfate (0.07 kg) and hexamethyldisilazane (6.6 kg) in chlorobenzene (52.4 kg) is heated to reflux (ca. 135° C.) and stirred (ca. 1 h) until the mixture becomes a clear solution. The reaction mixture is then concentrated in vacuo to obtain 32 as a syrupy liquid. The anhydrous solution of 30 in chlorobenzene (as prepared) and stannic chloride (28.2 kg) is added to this concentrate. The reaction mixture is maintained at about 70° C. until the desired coupling reaction is complete (ca. 10 h) as determined by in-process HPLC. Upon completion, the reaction mixture is cooled to RT and diluted with DCM (121 kg). This solution is added to a suspension of solid NaHCO3 (47 kg) and CELITE® (9.4 kg) in DCM (100.6 kg). The resulting slurry is cooled to 10-15° C., and water (8.4 kg) is added slowly to quench the reaction mixture. The resulting suspension is very slowly (caution: gas evolution) heated to reflux (ca. 45° C.) and maintained for about 30 min. The slurry is then cooled to ca. 15° C. and filtered. The filter cake is repeatedly reslurried in DCM (4×100 L) and filtered. The combined filtrate is concentrated under atmospheric pressure (the distillate collected in the process is used for reslurrying the filter cake) until the batch temperature rises to about 90° C. and then allowed to cool slowly to about −5° C. The resulting slurry is aged for at least 2 h at −5° C. The precipitated product is filtered and washed with IPA (30 kg+20 kg), and oven-dried in vacuo at about 70° C. to afford 8.8 kg (57.3%) of 1-(2-deoxy-2-fluoro-2-methyl-3-5-O-dibenzoyl-β-D-ribofuranosyl)-N-4-benzoylcytosine (14, CAS Reg No. 817204-32-3) which was 99.3% pure.
EXAMPLE 2 4-Amino-1-(3-fluoro-4-hydroxy-5-hydroxymethyl-3-methyl-tetrahydro-furan-2-yl)-1H-pyrimidin-2-one (18)
A slurry of 14 (14.7 kg) in MeOH (92.6 kg) is treated with catalytic amounts of methanolic sodium methoxide (0.275 kg). The reaction mixture is heated to ca. 50° C. and aged (ca. 1 h) until the hydrolysis is complete. The reaction mixture is quenched by addition of isobutyric acid (0.115 kg). The resulting solution is concentrated under moderate vacuum and then residual solvents are replaced with IPA (80 kg). The batch is distilled to a volume of ca. 50 L. The resulting slurry is heated to ca. 80° C. and then cooled slowly to ca. 5° C. and aged (ca. 2 h). The precipitated product is isolated by filtration, washed with IPA (16.8 kg) and dried in an oven at 70° C. in vacuo to afford 6.26 kg (88.9%) of 18 which assayed at 99.43% pure.
………………………………………………………………………
https://www.google.com/patents/US8334270
EXAMPLE 4 Preparation of 2′-deoxy-2′-fluoro-2′-C-methyluridine

2′-Deoxy-2′-fluoro-2′-C-methylcytidine (1.0 g, 1 eq) (Clark, J., et al., J. Med. Chem., 2005, 48, 5504-5508) was dissolved in 10 ml of anhydrous pyridine and concentrated to dryness in vacuo. The resulting syrup was dissolved in 20 ml of anhydrous pyridine under nitrogen and cooled to 0° C. with stirring. The brown solution was treated with benzoyl chloride (1.63 g, 3 eq) dropwise over 10 min. The ice bath was removed and stirring continued for 1.5 h whereby thin-layer chromatography (TLC) showed no remaining starting material. The mixture was quenched by addition of water (0.5 ml) and concentrated to dryness. The residue was dissolved in 50 mL of dichloromethane (DCM) and washed with saturated NaHCO3 aqueous solution and H2O. The organic phase was dried over NaSO4 and filtered, concentrated to dryness to give N4,3′,5′-tribenzoyl-2′-Deoxy-2′-fluoro-2′-C-methylcytidine (2.0 g, Yield: 91%).
N4,3′,5′-tribenzoyl-2′-Deoxy-2′-fluoro-2′-C-methylcytidine (2.0 g, 1 eq) was refluxed in 80% aqueous AcOH overnight. After cooling and standing at room temperature (15° C.), most of the product precipitated and then was filtered through a sintered funnel. White precipitate was washed with water and co-evaporated with toluene to give a white solid. The filtrate was concentrated and co-evaporated with toluene to give additional product which was washed with water to give a white solid. Combining the two batches of white solid gave 1.50 g of 3′,5′-dibenzoyl-2′-Deoxy-2′-fluoro-2′-C-methyluridine (Yield: 91%).
To a solution of 3′,5′-dibenzoyl-2′-Deoxy-2′-fluoro-2′-C-methyluridine (1.5 g, 1 eq) in MeOH (10 mL) was added a solution of saturated ammonia in MeOH (20 mL). The reaction mixture was stirred at 0° C. for 30 min, and then warmed to room temperature slowly. After the reaction mixture was stirred for another 18 hours, the reaction mixture was evaporated under reduced pressure to give the residue, which was purified by column chromatography to afford pure compound 2′-deoxy-2′-fluoro-2′-C-methyluridine (500 mg, Yield: 60%).
Example numbers 13-54 and 56-66 are prepared using similar procedures described for examples 5-8. The example number, compound identification, and NMR/MS details are shown below:
| entry 25 |
 |
| entry 251H NMR (DMSO-d6) δ 1.13-1.28 (m, 12H), 3.74-3.81 (m, 2H), 3.95-4.08 (m, 1H), 4.20-4.45 (m, 2H), 4.83-4.87 (m, 1H), 5.52-5.58 (m, 1H),5.84-6.15 (m, 3H), 7.17-7.23 (m, 3H), 7.35-7.39 (m, 2H), 7.54-7.57(m, 1H), 11.50 (s. 1H); MS, m/e 530.2 (M + 1)+ | ||||||
…………………………………..
Synthesis of diastereomerically pure nucleotide phosphoramidates.
Ross BS, Reddy PG, Zhang HR, Rachakonda S, Sofia MJ.
J Org Chem. 2011 Oct 21;76(20):8311-9. doi: 10.1021/jo201492m. Epub 2011 Sep 26.
The HCV NS5B nucleoside and non-nucleoside inhibitors.
Membreno FE, Lawitz EJ.
Clin Liver Dis. 2011 Aug;15(3):611-26. doi: 10.1016/j.cld.2011.05.003. Review.
Sofia MJ, Bao D, Chang W, Du J, Nagarathnam D, Rachakonda S, Reddy PG, Ross BS, Wang P, Zhang HR, Bansal S, Espiritu C, Keilman M, Lam AM, Steuer HM, Niu C, Otto MJ, Furman PA.
J Med Chem. 2010 Oct 14;53(19):7202-18. doi: 10.1021/jm100863x.
Mechanism of activation of PSI-7851 and its diastereoisomer PSI-7977.
Murakami E, Tolstykh T, Bao H, Niu C, Steuer HM, Bao D, Chang W, Espiritu C, Bansal S, Lam AM, Otto MJ, Sofia MJ, Furman PA.
J Biol Chem. 2010 Nov 5;285(45):34337-47. doi: 10.1074/jbc.M110.161802. Epub 2010 Aug 26.
Michael J. Sofia,Donghui Bao, Wonsuk Chang, Jinfa Du, Dhanapalan Nagarathnam, Suguna Rachakonda, P. Ganapati Reddy, Bruce S. Ross, Peiyuan Wang, Hai-Ren Zhang, Shalini Bansal, Christine Espiritu, Meg Keilman, Angela M. Lam, Holly M. Micolochick Steuer, Congrong Niu, Michael J. Otto, and Phillip A. Furman; Discovery of a β-D-2’-Deoxy-2’-a-fluoro-2’-β-C-methyluridine Nucleotide Prodrug (PSI-7977) for the Treatment of Hepatitis C Virus; J. Med. Chem. 2010, 53, 7202–7218; Pharmasset, Inc.
Bruce S. Ross, P. Ganapati Reddy , Hai-Ren Zhang , Suguna Rachakonda , and Michael J. Sofia; Synthesis of Diastereomerically Pure Nucleotide Phosphoramidates; J. Org. Chem., 2011, 76 (20), pp 8311–8319; Pharmasset, Inc.
Peiyuan Wang, Byoung-Kwon Chun, Suguna Rachakonda, Jinfa Du, Noshena Khan, Junxing Shi, Wojciech Stec, Darryl Cleary, Bruce S. Ross and Michael J. Sofia; An Efficient and Diastereoselective Synthesis of PSI-6130: A Clinically Efficacious Inhibitor of HCV NS5B Polymerase; J. Org. Chem., 2009, 74 (17), pp 6819–6824;Pharmasset, Inc.
Jeremy L. Clark, Laurent Hollecker, J. Christian Mason, Lieven J. Stuyver, Phillip M. Tharnish, Stefania Lostia, Tamara R. McBrayer, Raymond F. Schinazi, Kyoichi A. Watanabe, Michael J. Otto, Phillip A. Furman, Wojciech J. Stec, Steven E. Patterson, and Krzysztof W. Pankiewicz; Design, Synthesis, and Antiviral Activity of 2‘-Deoxy-2‘-fluoro-2‘-C-methylcytidine, a Potent Inhibitor of Hepatitis C Virus Replication; J. Med. Chem., 2005, 48 (17), pp 5504–5508; Pharmasset, Inc
- Harrison C. Patent battle lines drawn as sofosbuvir gains approval. , Nat Rev Drug Discov , Volume 13 , Issue 1 , 2013 Dec 31
- Traynor K. Sofosbuvir approved for chronic hepatitis C infection. , Am J Health Syst Pharm , Volume 71 , Issue 2 , 2014 Jan 15
- Benhamou Y. HCV F1/F2 patients: treat now or continue to wait. , Liver Int , Volume 34 Suppl 1 , 2014 Feb
- Asselah T. HCV direct-acting antiviral agents: the best interferon-free combinations. , Liver Int , Volume 34 Suppl 1 , 2014 Feb
- Marcellin P. Second-wave IFN-based triple therapy for HCV genotype 1 infection: simeprevir, faldaprevir and sofosbuvir. , Liver Int , Volume 34 Suppl 1 , 2014 Feb
- Corouge M. Treatment of hepatitis C virus genotype 3-infection. , Liver Int , Volume 34 Suppl 1 , 2014 Feb
- Osinusi A. IFNL4-ΔG Genotype is Associated with Slower Viral Clearance in Hepatitis C, Genotype-1 Patients Treated with Sofosbuvir and Ribavirin. , J Infect Dis , 2013 Dec 23
- Alavian SM. Sofosbuvir has come out of the magic box. , Hepat Mon , Volume 13 , Issue 12 , 2013 Dec 16
- Ferguson MC. Sofosbuvir with ribavirin is safe and effective in hepatitis C genotype 1 with unfavourable pretreatment characteristics. , Evid Based Med , 2013 Dec 12
- Hunt S. Minimal Impact of Sofosbuvir and Ribavirin on Health Related Quality of Life in Chronic Hepatitis C (CH-C). , J Hepatol , 2013 Dec 10
SOVALDI is the brand name for sofosbuvir, a nucleotide analog inhibitor of HCV NS5B polymerase.
The IUPAC name for sofosbuvir is (S)-Isopropyl 2-((S)-(((2R,3R,4R,5R)-5-(2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)-4-fluoro-3-hydroxy-4-methyltetrahydrofuran-2-yl)methoxy)-(phenoxy)phosphorylamino)propanoate. It has a molecular formula of C22H29FN3O9P and a molecular weight of 529.45. It has the following structural formula:
 |
Sofosbuvir is a white to off-white crystalline solid with a solubility of ≥ 2 mg/mL across the pH range of 2-7.7 at 37 oC and is slightly soluble in water.
SOVALDI tablets are for oral administration. Each tablet contains 400 mg of sofosbuvir. The tablets include the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, mannitol, and microcrystalline cellulose. The tablets are film-coated with a coating material containing the following inactive ingredients: polyethylene glycol, polyvinyl alcohol, talc, titanium dioxide, and yellow iron oxide.
Want to know everything on vir series
click
http://drugsynthesisint.blogspot.in/p/vir-series-hep-c-virus-22.html
AND
http://medcheminternational.blogspot.in/p/vir-series-hep-c-virus.html
updated

J. Med. Chem. 2005, 48, 5504.
WO2008045419A1
CN201180017181
(WO2015139602) Sofosbuvir New Patent

SOFOSBUVIR
NEW PATENT WO2015188782,
(WO2015188782) METHOD FOR PREPARING SOFOSBUVIR
CHIA TAI TIANQING PHARMACEUTICAL GROUP CO., LTD [CN/CN]; No. 8 Julong North Rd., Xinpu District Lianyungang, Jiangsu 222006 (CN)
Sofosbuvir synthesis routes currently used include the following two methods:
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2015188782&redirectedID=true


![]()
//////


Positive Review For Gilead’s Hep C Drug Sofosbuvir
Sofosbuvir
Isopropyl (2S)-2-[[[(2R,3R,4R,5R)-5-(2,4-dioxopyrimidin-1-yl)-4-fluoro-3-hydroxy-4-methyl-tetrahydrofuran-2-yl]methoxy-phenoxy-phosphoryl]amino]propanoate
http://www.ama-assn.org/resources/doc/usan/sofosbuvir.pdf –for cas no
The U.S. Food and Drug Administration (FDA) has issued a positive review for a highly anticipated hepatitis C drug from Gilead Sciences, saying the pill cures more patients in less time than currently available treatments.
The agency posted its review of Gilead’s sofosbuvir online ahead of a meeting Friday where government experts will vote on whether to recommend the drug’s approval.
old article cut paste
Jun. 7, 2013– Gilead Sciences, Inc. today announced that the U.S. Food and Drug Administration (FDA) has granted priority review to the company’s New Drug Application (NDA) for sofosbuvir, a once-daily oral nucleotide analogue inhibitor for the treatment of chronic hepatitis C virus (HCV) infection. The FDA grants priority review status to drug candidates that may offer major advances in treatment over existing options. Gilead filed the NDA for sofosbuvir on April 8, 2013, and FDA has set a target review date under the Prescription Drug User Fee Act (PDUFA) of December 8, 2013.

The data submitted in this NDA support the use of sofosbuvir and ribavirin (RBV) as an all-oral therapy for patients with genotype 2 and 3 HCV infection, and for sofosbuvir in combination with RBV and pegylated interferon (peg-IFN) for treatment-naïve patients with genotype 1, 4, 5 and 6 HCV infection.
Sofosbuvir is an investigational product and its safety and efficacy have not yet been established.
About Gilead Sciences
Gilead Sciences is a biopharmaceutical company that discovers, develops and commercializes innovative therapeutics in areas of unmet medical need. The company’s mission is to advance the care of patients suffering from life-threatening diseases worldwide. Headquartered in Foster City, California, Gilead has operations in North America, Europe and Asia Pacific.
Sofosbuvir (formerly PSI-7977 or GS-7977) is an experimental drug candidate for the treatment of hepatitis C.[1] It was discovered at Pharmasset and then acquired for development by Gilead Sciences. It is currently in Phase III clinical trials.[2]
Sofosbuvir is a prodrug that is metabolized to the active antiviral agent 2′-deoxy-2′-α-fluoro-β-C-methyluridine-5′-monophosphate.[3]
Sofosbuvir is a nucleotide analogue inhibitor of the hepatitis C virus (HCV) polymerase.[4] The HCV polymerase or NS5B protein is a RNA-dependent RNA polymerase critical for the viral cycle.
Sofosbuvir is being studied in combination with pegylated interferon and ribavirin, with ribavirin alone, and with other direct-acting antiviral agents.[5] It has shown excellent clinical efficacy when used either with pegylated interferon/ribavirin or in interferon-free combinations. In particular, combinations of sofosbuvir with NS5A inhibitors, such as daclatasvir or GS-5885, have shown sustained virological response rates of up to 100% in people infected with HCV.[6]
Data from the ELECTRON trial showed that a dual interferon-free regimen of sofosbuvir plus ribavirin produced a 24-week post-treatment sustained virological response (SVR24) rate of 100% for previously untreated patients with HCV genotypes 2 or 3.[7][8]
Data presented at the 20th Conference on Retroviruses and Opportunistic Infections in March 2013 showed that a triple regimen of sofosbuvir, ledipasvir (formerly GS-5885), and ribavirin produced a 12-week post-treatment sustained virological response (SVR12) rate of 100% for both treatment-naive patients and prior non-responders with HCV genotype 1.[9]Gilead has developed a sofosbuvir + ledipasvir coformulation that is being tested with and without ribavirin.

- Sofia, M. J.; Bao, D.; Chang, W.; Du, J.; Nagarathnam, D.; Rachakonda, S.; Reddy, P. G.; Ross, B. S. et al. (2010). “Discovery of a β-d-2′-Deoxy-2′-α-fluoro-2′-β-C-methyluridine Nucleotide Prodrug (PSI-7977) for the Treatment of Hepatitis C Virus”. Journal of Medicinal Chemistry 53 (19): 7202–7218. doi:10.1021/jm100863x. PMID 20845908. edit
- “PSI-7977″. Gilead Sciences.
- Murakami, E.; Tolstykh, T.; Bao, H.; Niu, C.; Steuer, H. M. M.; Bao, D.; Chang, W.; Espiritu, C. et al. (2010). “Mechanism of Activation of PSI-7851 and Its Diastereoisomer PSI-7977″. Journal of Biological Chemistry 285 (45): 34337–34347.doi:10.1074/jbc.M110.161802. PMC 2966047. PMID 20801890. edit
- Alejandro Soza (November 11, 2012). “Sofosbuvir”. Hepaton.
- Tom Murphy (November 21, 2011). “Gilead Sciences to buy Pharmasset for $11 billion”. Bloomberg Businessweek.
- http://www.gilead.com/pr_1757156
- AASLD: PSI-7977 plus Ribavirin Can Cure Hepatitis C in 12 Weeks without Interferon. Highleyman, L. HIVandHepatitis.com. 8 November 2011.
- Nucleotide Polymerase Inhibitor Sofosbuvir plus Ribavirin for Hepatitis C. Gane, E et al. New England Journal of Medicine 368:3444. January 3, 2013.
- CROI 2013: Sofosbuvir + Ledipasvir + Ribavirin Combo for HCV Produces 100% Sustained Response. Highleyman, L. HIVandHepatitis.com. 4 March 2013.
Gilead Announces U.S. FDA Priority Review Designation for Sofosbuvir for the Treatment of Hepatitis C
Sofosbuvir
Isopropyl (2S)-2-[[[(2R,3R,4R,5R)-5-(2,4-dioxopyrimidin-1-yl)-4-fluoro-3-hydroxy-4-methyl-tetrahydrofuran-2-yl]methoxy-phenoxy-phosphoryl]amino]propanoate
http://www.ama-assn.org/resources/doc/usan/sofosbuvir.pdf –for cas no
Jun. 7, 2013– Gilead Sciences, Inc. today announced that the U.S. Food and Drug Administration (FDA) has granted priority review to the company’s New Drug Application (NDA) for sofosbuvir, a once-daily oral nucleotide analogue inhibitor for the treatment of chronic hepatitis C virus (HCV) infection. The FDA grants priority review status to drug candidates that may offer major advances in treatment over existing options. Gilead filed the NDA for sofosbuvir on April 8, 2013, and FDA has set a target review date under the Prescription Drug User Fee Act (PDUFA) of December 8, 2013.

The data submitted in this NDA support the use of sofosbuvir and ribavirin (RBV) as an all-oral therapy for patients with genotype 2 and 3 HCV infection, and for sofosbuvir in combination with RBV and pegylated interferon (peg-IFN) for treatment-naïve patients with genotype 1, 4, 5 and 6 HCV infection.
Sofosbuvir is an investigational product and its safety and efficacy have not yet been established.
About Gilead Sciences
Gilead Sciences is a biopharmaceutical company that discovers, develops and commercializes innovative therapeutics in areas of unmet medical need. The company’s mission is to advance the care of patients suffering from life-threatening diseases worldwide. Headquartered in Foster City, California, Gilead has operations in North America, Europe and Asia Pacific.
Sofosbuvir (formerly PSI-7977 or GS-7977) is an experimental drug candidate for the treatment of hepatitis C.[1] It was discovered at Pharmasset and then acquired for development by Gilead Sciences. It is currently in Phase III clinical trials.[2]
Sofosbuvir is a prodrug that is metabolized to the active antiviral agent 2′-deoxy-2′-α-fluoro-β-C-methyluridine-5′-monophosphate.[3]
Sofosbuvir is a nucleotide analogue inhibitor of the hepatitis C virus (HCV) polymerase.[4] The HCV polymerase or NS5B protein is a RNA-dependent RNA polymerase critical for the viral cycle.
Sofosbuvir is being studied in combination with pegylated interferon and ribavirin, with ribavirin alone, and with other direct-acting antiviral agents.[5] It has shown excellent clinical efficacy when used either with pegylated interferon/ribavirin or in interferon-free combinations. In particular, combinations of sofosbuvir with NS5A inhibitors, such as daclatasvir or GS-5885, have shown sustained virological response rates of up to 100% in people infected with HCV.[6]
Data from the ELECTRON trial showed that a dual interferon-free regimen of sofosbuvir plus ribavirin produced a 24-week post-treatment sustained virological response (SVR24) rate of 100% for previously untreated patients with HCV genotypes 2 or 3.[7][8]
Data presented at the 20th Conference on Retroviruses and Opportunistic Infections in March 2013 showed that a triple regimen of sofosbuvir, ledipasvir (formerly GS-5885), and ribavirin produced a 12-week post-treatment sustained virological response (SVR12) rate of 100% for both treatment-naive patients and prior non-responders with HCV genotype 1.[9]Gilead has developed a sofosbuvir + ledipasvir coformulation that is being tested with and without ribavirin.

- Sofia, M. J.; Bao, D.; Chang, W.; Du, J.; Nagarathnam, D.; Rachakonda, S.; Reddy, P. G.; Ross, B. S. et al. (2010). “Discovery of a β-d-2′-Deoxy-2′-α-fluoro-2′-β-C-methyluridine Nucleotide Prodrug (PSI-7977) for the Treatment of Hepatitis C Virus”. Journal of Medicinal Chemistry 53 (19): 7202–7218. doi:10.1021/jm100863x. PMID 20845908. edit
- “PSI-7977″. Gilead Sciences.
- Murakami, E.; Tolstykh, T.; Bao, H.; Niu, C.; Steuer, H. M. M.; Bao, D.; Chang, W.; Espiritu, C. et al. (2010). “Mechanism of Activation of PSI-7851 and Its Diastereoisomer PSI-7977″. Journal of Biological Chemistry 285 (45): 34337–34347.doi:10.1074/jbc.M110.161802. PMC 2966047. PMID 20801890. edit
- Alejandro Soza (November 11, 2012). “Sofosbuvir”. Hepaton.
- Tom Murphy (November 21, 2011). “Gilead Sciences to buy Pharmasset for $11 billion”. Bloomberg Businessweek.
- http://www.gilead.com/pr_1757156
- AASLD: PSI-7977 plus Ribavirin Can Cure Hepatitis C in 12 Weeks without Interferon. Highleyman, L. HIVandHepatitis.com. 8 November 2011.
- Nucleotide Polymerase Inhibitor Sofosbuvir plus Ribavirin for Hepatitis C. Gane, E et al. New England Journal of Medicine 368:3444. January 3, 2013.
- CROI 2013: Sofosbuvir + Ledipasvir + Ribavirin Combo for HCV Produces 100% Sustained Response. Highleyman, L. HIVandHepatitis.com. 4 March 2013.