

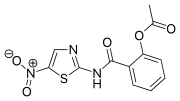
Nitazoxanide
| Formula |
C12H9N3O5S
|
|---|---|
| Exact mass |
307.0263
|
| Mol weight |
307.282
|
Nitazoxanide is a broad-spectrum antiparasitic and broad-spectrum antiviral drug that is used in medicine for the treatment of various helminthic, protozoal, and viral infections.[4][5][6] It is indicated for the treatment of infection by Cryptosporidium parvum and Giardia lamblia in immunocompetent individuals and has been repurposed for the treatment of influenza.[1][6] Nitazoxanide has also been shown to have in vitro antiparasitic activity and clinical treatment efficacy for infections caused by other protozoa and helminths;[4][7] emerging evidence suggests that it possesses efficacy in treating a number of viral infections as well.[6]
Chemically, nitazoxanide is the prototype member of the thiazolides, a class of drugs which are synthetic nitrothiazolyl-salicylamide derivatives with antiparasitic and antiviral activity.[4][6][8] Tizoxanide, an active metabolite of nitazoxanide in humans, is also an antiparasitic drug of the thiazolide class.[4][9]
Uses
Nitazoxanide is an effective first-line treatment for infection by Blastocystis species[10][11] and is indicated for the treatment of infection by Cryptosporidium parvum or Giardia lamblia in immunocompetent adults and children.[1] It is also an effective treatment option for infections caused by other protozoa and helminths (e.g., Entamoeba histolytica,[12] Hymenolepis nana,[13] Ascaris lumbricoides,[14] and Cyclospora cayetanensis[15]).[7]
As of September 2015, it is in phase 3 clinical trials for the treatment influenza due to its inhibitory effect on a broad range of influenza virus subtypes and efficacy against influenza viruses that are resistant to neuraminidase inhibitors like oseltamivir.[6][16] Nitazoxanide is also being researched as a potential treatment for chronic hepatitis B, chronic hepatitis C, rotavirus and norovirus gastroenteritis.[6]
Chronic hepatitis B
Nitazoxanide alone has shown preliminary evidence of efficacy in the treatment of chronic hepatitis B over a one-year course of therapy.[17] Nitazoxanide 500 mg twice daily resulted in a decrease in serum HBV DNA in all of 4 HBeAg-positive patients, with undetectable HBV DNA in 2 of 4 patients, loss of HBeAg in 3 patients, and loss of HBsAg in one patient. Seven of 8 HBeAg-negative patients treated with nitazoxanide 500 mg twice daily had undetectable HBV DNA and 2 had loss of HBsAg. Additionally, nitazoxanide monotherapy in one case and nitazoxanide plus adefovir in another case resulted in undetectable HBV DNA, loss of HBeAg and loss of HBsAg.[18] These preliminary studies showed a higher rate of HBsAg loss than any currently licensed therapy for chronic hepatitis B. The similar mechanism of action of interferon and nitazoxanide suggest that stand-alone nitazoxanide therapy or nitazoxanide in concert with nucleos(t)ide analogs have the potential to increase loss of HBsAg, which is the ultimate end-point of therapy. A formal phase Ⅱ study is being planned for 2009.[19]
Chronic hepatitis C
Romark initially decided to focus on the possibility of treating chronic hepatitis C with nitazoxanide.[20] The drug garnered interest from the hepatology community after three phase II clinical trials involving the treatment of hepatitis C with nitazoxanide produced positive results for treatment efficacy and similar tolerability to placebo without any signs of toxicity.[20] A meta-analysis from 2014 concluded that the previous held trials were of low-quality and with held with a risk of bias. The authors concluded that more randomized trials with low risk of bias are needed to give any determine if Nitazoxanide can be used as an effective treatment for chronic hepatitis C patients.[21]
Clinical trials
Nitazoxanide has gone through Phase II clinical trials for the treatment of hepatitis C, in combination with peginterferon alfa-2a and ribavirin.[22][23]Romark Laboratories has announced encouraging results from international Phase I and II clinical trials evaluating a controlled release version of nitazoxanide in the treatment of chronic hepatitis C virus infection. The company used 675 mg and 1,350 mg twice daily doses of controlled release nitazoxanide showed favorable safety and tolerability throughout the course of the study, with mild to moderate adverse events. Primarily GI-related adverse events were reported.
A randomised double-blind placebo-controlled study published in 2006, with a group of 38 young children (Lancet, vol 368, page 124-129)[24] concluded that a 3-day course of nitazoxanide significantly reduced the duration of rotavirus disease in hospitalized pediatric patients. Dose given was “7.5 mg/kg twice daily” and the time of resolution was “31 hours for those given nitazoxanide compared with 75 hours for those in the placebo group.” Rotavirus is the most common infectious agent associated with diarrhea in the pediatric age group worldwide.
Teran et al.. conducted a study at the Pediatric Center Albina Patinö, a reference hospital in the city of Cochabamba, Bolivia, from August 2007 to February 2008. The study compared nitazoxanide and probiotics in the treatment of acute rotavirus diarrhea. They found Small differences in favor of nitazoxanide in comparison with probiotics and concluded that nitazoxanide is an important treatment option for rotavirus diarrhea.[17]
Lateef et al.. conducted a study in India that evaluated the effectiveness of nitazoxanide in the treatment of beef tapeworm (Taenia saginata) infection. They concluded that nitazoxanide is a safe, effective, inexpensive, and well-tolerated drug for the treatment of niclosamide- and praziquantel-resistant beef tapeworm (Taenia saginata) infection.[18]
A retrospective review of charts of patients treated with nitazoxanide for trichomoniasis by Michael Dan and Jack D. Sobel demonstrated negative result. They reported three case studies; two of which with metronidazole-resistant infections. In Case 3, they reported the patient to be cured with high divided dose tinidazole therapy. They used a high dosage of the drug (total dose, 14–56 g) than the recommended standard dosage (total dose, 3 g) and observed a significant adverse reaction (poorly tolerated nausea) only with the very high dose (total dose, 56 g). While confirming the safety of the drug, they showed nitazoxanide is ineffective for the treatment of trichomoniasis.[25]
Contraindications
Nitazoxanide is contraindicated only in individuals who have experienced a hypersensitivity reaction to nitazoxanide or the inactive ingredients of a nitazoxanide formulation.[1]
Adverse effects
The side effects of nitazoxanide do not significantly differ from a placebo treatment for giardiasis;[1] these symptoms include stomach pain, headache, upset stomach, vomiting, discolored urine, excessive urinating, skin rash, itching, fever, flu syndrome, and others.[1][26] Nitazoxanide does not appear to cause any significant adverse effects when taken by healthy adults.[1][2]
Overdose
Information on nitazoxanide overdose is limited. Oral doses of 4 grams in healthy adults do not appear to cause any significant adverse effects.[1][2] In various animals, the oral LD50 is higher than 10 g/kg.[1]
Interactions
Due to the exceptionally high plasma protein binding (>99.9%) of nitazoxanide’s metabolite, tizoxanide, the concurrent use of nitazoxanide with other highly plasma protein-bound drugs with narrow therapeutic indices (e.g., warfarin) increases the risk of drug toxicity.[1] In vitro evidence suggests that nitazoxanide does not affect the CYP450 system.[1]
Pharmacology
Pharmacodynamics
The anti-protozoal activity of nitazoxanide is believed to be due to interference with the pyruvate:ferredoxin oxidoreductase (PFOR) enzyme-dependent electron transfer reaction which is essential to anaerobic energy metabolism.[1][8] PFOR inhibition may also contribute to its activity against anaerobic bacteria.[27]
It has also been shown to have activity against influenza A virus in vitro.[28] The mechanism appears to be by selectively blocking the maturation of the viral hemagglutinin at a stage preceding resistance to endoglycosidase H digestion. This impairs hemagglutinin intracellular trafficking and insertion of the protein into the host plasma membrane.
Nitazoxanide modulates a variety of other pathways in vitro, including glutathione-S-transferase and glutamate-gated chloride ion channels in nematodes, respiration and other pathways in bacteria and cancer cells, and viral and host transcriptional factors.[27]
Pharmacokinetics
Following oral administration, nitazoxanide is rapidly hydrolyzed to the pharmacologically active metabolite, tizoxanide, which is 99% protein bound.[1][9] Tizoxanide is then glucuronide conjugated into the active metabolite, tizoxanide glucuronide.[1] Peak plasma concentrations of the metabolites tizoxanide and tizoxanide glucuronide are observed 1–4 hours after oral administration of nitazoxanide, whereas nitazoxanide itself is not detected in blood plasma.[1]
Roughly 2⁄3 of an oral dose of nitazoxanide is excreted as its metabolites in feces, while the remainder of the dose excreted in urine.[1] Tizoxanide is excreted in the urine, bile and feces.[1] Tizoxanide glucuronide is excreted in urine and bile.[1]
Chemistry
History
Nitazoxanide is the prototype member of the thiazolides, which is a drug class of structurally-related broad-spectrum antiparasitic compounds.[4] Nitazoxanide is a light yellow crystalline powder. It is poorly soluble in ethanol and practically insoluble in water.
Nitazoxanide was originally discovered in the 1980s by Jean-François Rossignol at the Pasteur Institute. Initial studies demonstrated activity versus tapeworms. In vitro studies demonstrated much broader activity. Dr. Rossignol co-founded Romark Laboratories, with the goal of bringing nitazoxanide to market as an anti-parasitic drug. Initial studies in the USA were conducted in collaboration with Unimed Pharmaceuticals, Inc. (Marietta, GA) and focused on development of the drug for treatment of cryptosporidiosis in AIDS. Controlled trials began shortly after the advent of effective anti-retroviral therapies. The trials were abandoned due to poor enrollment and the FDA rejected an application based on uncontrolled studies.
Subsequently, Romark launched a series of controlled trials. A placebo-controlled study of nitazoxanide in cryptosporidiosis demonstrated significant clinical improvement in adults and children with mild illness. Among malnourished children in Zambia with chronic cryptosporidiosis, a three-day course of therapy led to clinical and parasitologic improvement and improved survival. In Zambia and in a study conducted in Mexico, nitazoxanide was not successful in the treatment of cryptosporidiosis in advanced infection with human immunodeficiency virus at the doses used. However, it was effective in patients with higher CD4 counts. In treatment of giardiasis, nitazoxanide was superior to placebo and comparable to metronidazole. Nitazoxanide was successful in the treatment of metronidazole-resistant giardiasis. Studies have suggested efficacy in the treatment of cyclosporiasis, isosporiasis, and amebiasis.[29] Recent studies have also found it to be effective against beef tapeworm(Taenia saginata).[30]
Research
Nitazoxanide is also under investigation for the treatment of COVID-19.[31]
Pharmaceutical products
Dosage forms
Nitazoxanide is currently available in two oral dosage forms: a tablet (500 mg) and an oral suspension (100 mg per 5 ml when reconstituted).[1]
An extended release tablet (675 mg) has been used in clinical trials for chronic hepatitis C; however, this form is not currently marketed and available for prescription.[20]
Brand names
Nitazoxanide is sold under the brand names Adonid, Alinia, Allpar, Annita, Celectan, Colufase, Daxon, Dexidex, Diatazox, Kidonax, Mitafar, Nanazoxid, Parazoxanide, Netazox, Niazid, Nitamax, Nitax, Nitaxide, Nitaz, Nizonide, NT-TOX, Pacovanton, Paramix, Toza, and Zox.
SYN


https://www.sciencedirect.com/science/article/pii/S0960894X11002848
CLIP


CLIP
PATENT

https://patents.google.com/patent/CN105175352A/zh
References
- ^ Jump up to:a b c d e f g h i j k l m n o p q r s t u v w “Nitazoxanide Prescribing Information” (PDF). Romark Pharmaceuticals. August 2013. pp. 1–5. Archived from the original (PDF) on 16 January 2016. Retrieved 3 January 2016.
- ^ Jump up to:a b c d e Stockis A, Allemon AM, De Bruyn S, Gengler C (May 2002). “Nitazoxanide pharmacokinetics and tolerability in man using single ascending oral doses”. Int J Clin Pharmacol Ther. 40 (5): 213–220. doi:10.5414/cpp40213. PMID 12051573.
- ^ “Nitazoxanide”. PubChem Compound. National Center for Biotechnology Information. Retrieved 3 January 2016.
- ^ Jump up to:a b c d e Di Santo N, Ehrisman J (2013). “Research perspective: potential role of nitazoxanide in ovarian cancer treatment. Old drug, new purpose?”. Cancers (Basel). 5 (3): 1163–1176. doi:10.3390/cancers5031163. PMC 3795384. PMID 24202339.
Nitazoxanide [NTZ: 2-acetyloxy-N-(5-nitro-2-thiazolyl)benzamide] is a thiazolide antiparasitic agent with excellent activity against a wide variety of protozoa and helminths. … Nitazoxanide (NTZ) is a main compound of a class of broad-spectrum anti-parasitic compounds named thiazolides. It is composed of a nitrothiazole-ring and a salicylic acid moiety which are linked together by an amide bond … NTZ is generally well tolerated, and no significant adverse events have been noted in human trials [13]. … In vitro, NTZ and tizoxanide function against a wide range of organisms, including the protozoal species Blastocystis hominis, C. parvum, Entamoeba histolytica, G. lamblia and Trichomonas vaginalis [13]
- ^ White CA (2004). “Nitazoxanide: a new broad spectrum antiparasitic agent”. Expert Rev Anti Infect Ther. 2 (1): 43–9. doi:10.1586/14787210.2.1.43. PMID 15482170.
- ^ Jump up to:a b Anderson, V. R.; Curran, M. P. (2007). “Nitazoxanide: A review of its use in the treatment of gastrointestinal infections”. Drugs. 67(13): 1947–1967. doi:10.2165/00003495-200767130-00015. PMID 17722965.
Nitazoxanide is effective in the treatment of protozoal and helminthic infections … Nitazoxanide is a first-line choice for the treatment of illness caused by C. parvum or G. lamblia infection in immunocompetent adults and children, and is an option to be considered in the treatment of illnesses caused by other protozoa and/or helminths.
- ^ Jump up to:a b Sisson G1, Goodwin A, Raudonikiene A, Hughes NJ, Mukhopadhyay AK, Berg DE, Hoffman PS. (July 2002). “Enzymes associated with reductive activation and action of nitazoxanide, nitrofurans, and metronidazole in Helicobacter pylori”. Antimicrob. Agents Chemother. 46 (7): 2116–23. doi:10.1128/aac.46.7.2116-2123.2002. PMC 127316. PMID 12069963.
Nitazoxanide (NTZ) is a redox-active nitrothiazolyl-salicylamide
- ^ Jump up to:a b Korba BE, Montero AB, Farrar K, et al. (January 2008). “Nitazoxanide, tizoxanide and other thiazolides are potent inhibitors of hepatitis B virus and hepatitis C virus replication”. Antiviral Res. 77 (1): 56–63. doi:10.1016/j.antiviral.2007.08.005. PMID 17888524.
- ^ “Blastocystis: Resources for Health Professionals”. United States Centers for Disease Control and Prevention. 2017-05-02. Retrieved 4 January 2016.
- ^ Roberts T, Stark D, Harkness J, Ellis J (May 2014). “Update on the pathogenic potential and treatment options for Blastocystis sp”. Gut Pathog. 6: 17. doi:10.1186/1757-4749-6-17. PMC 4039988. PMID 24883113.
Blastocystis is one of the most common intestinal protists of humans. … A recent study showed that 100% of people from low socio-economic villages in Senegal were infected with Blastocystis sp. suggesting that transmission was increased due to poor hygiene sanitation, close contact with domestic animals and livestock, and water supply directly from well and river [10]. …
Table 2: Summary of treatments and efficacy for Blastocystis infection - ^ Muñoz P, Valerio M, Eworo A, Bouza E (2011). “Parasitic infections in solid-organ transplant recipients”. Curr Opin Organ Transplant. 16 (6): 565–575. doi:10.1097/MOT.0b013e32834cdbb0. PMID 22027588. Retrieved 7 January 2016.
Nitazoxanide: intestinal amoebiasis: 500 mg po bid x 3 days
- ^ “Hymenolepiasis: Resources for Health Professionals”. United States Centers for Disease Control and Prevention. 2017-05-02. Retrieved 4 January 2016.
- ^ Hagel I, Giusti T (October 2010). “Ascaris lumbricoides: an overview of therapeutic targets”. Infectious Disorders – Drug Targets. 10 (5): 349–67. doi:10.2174/187152610793180876. PMID 20701574.
new anthelmintic alternatives such as tribendimidine and Nitazoxanide have proved to be safe and effective against A. lumbricoides and other soil-transmitted helminthiases in human trials.
- ^ Shoff WH (5 October 2015). Chandrasekar PH, Talavera F, King JW (eds.). “Cyclospora Medication”. Medscape. WebMD. Retrieved 11 January 2016.
Nitazoxanide, a 5-nitrothiazole derivative with broad-spectrum activity against helminths and protozoans, has been shown to be effective against C cayetanensis, with an efficacy 87% by the third dose (first, 71%; second 75%). Three percent of patients had minor side effects.
- ^ Li TC, Chan MC, Lee N (September 2015). “Clinical Implications of Antiviral Resistance in Influenza”. Viruses. 7 (9): 4929–4944. doi:10.3390/v7092850. PMC 4584294. PMID 26389935.
Oral nitazoxanide is an available, approved antiparasitic agent (e.g., against cryptosporidium, giardia) with established safety profiles. Recently, it has been shown (together with its active metabolite tizoxanide) to possess anti-influenza activity by blocking haemagglutinin maturation/trafficking, and acting as an interferon-inducer [97]. … A large, multicenter, Phase 3 randomized-controlled trial comparing nitazoxanide, oseltamivir, and their combination in uncomplicated influenza is currently underway (NCT01610245).
Figure 1: Molecular targets and potential antiviral treatments against influenza virus infection - ^ Jump up to:a b Teran, C. G.; Teran-Escalera, C. N.; Villarroel, P. (2009). “Nitazoxanide vs. Probiotics for the treatment of acute rotavirus diarrhea in children: A randomized, single-blind, controlled trial in Bolivian children”. International Journal of Infectious Diseases. 13(4): 518–523. doi:10.1016/j.ijid.2008.09.014. PMID 19070525.
- ^ Jump up to:a b Lateef, M.; Zargar, S. A.; Khan, A. R.; Nazir, M.; Shoukat, A. (2008). “Successful treatment of niclosamide- and praziquantel-resistant beef tapeworm infection with nitazoxanide”. International Journal of Infectious Diseases. 12 (1): 80–82. doi:10.1016/j.ijid.2007.04.017. PMID 17962058.
- ^ World Journal of Gastroenterology 2009 April 21, Emmet B Keeffe MD, Professor, Jean-François Rossignol The Romark Institute for Medical Research, Tampa
- ^ Jump up to:a b c Keeffe, E. B.; Rossignol, J. F. (2009). “Treatment of chronic viral hepatitis with nitazoxanide and second generation thiazolides”. World Journal of Gastroenterology. 15 (15): 1805–1808. doi:10.3748/wjg.15.1805. PMC 2670405. PMID 19370775.
- ^ Nikolova, Kristiana; Gluud, Christian; Grevstad, Berit; Jakobsen, Janus C (2014). “Nitazoxanide for chronic hepatitis C”. Cochrane Database of Systematic Reviews (4): CD009182. doi:10.1002/14651858.CD009182.pub2. ISSN 1465-1858. PMID 24706397.
- ^ “Romark Initiates Clinical Trial Of Alinia For Chronic Hepatitis C In The United States” (Press release). Medical News Today. August 16, 2007. Retrieved 2007-10-11.
- ^ Franciscus, Alan (October 2, 2007). “Hepatitis C Treatments in Current Clinical Development”. HCV Advocate. Archived from the original on September 6, 2003. Retrieved 2007-10-11.
- ^ Rossignol, Jean-François; Abu-Zekry, Mona; Hussein, Abeer; Santoro, M Gabriella (2006). “Effect of nitazoxanide for treatment of severe rotavirus diarrhoea: randomised double-blind placebo-controlled trial”. The Lancet. 368 (9530): 124–9. CiteSeerX 10.1.1.458.1597. doi:10.1016/S0140-6736(06)68852-1. PMID 16829296.
- ^ Dan, M.; Sobel, J. D. (2007). “Failure of Nitazoxanide to Cure Trichomoniasis in Three Women”. Sexually Transmitted Diseases. 34 (10): 813–4. doi:10.1097/NMD.0b013e31802f5d9a. PMID 17551415.
- ^ “Nitazoxanide”. MedlinePlus. Retrieved 9 April 2014.
- ^ Jump up to:a b Shakya, A; Bhat, HR; Ghosh, SK (2018). “Update on Nitazoxanide: A Multifunctional Chemotherapeutic Agent”. Current Drug Discovery Technologies. 15 (3): 201–213. doi:10.2174/1570163814666170727130003. PMID 28748751.
- ^ Rossignol, J. F.; La Frazia, S.; Chiappa, L.; Ciucci, A.; Santoro, M. G. (2009). “Thiazolides, a New Class of Anti-influenza Molecules Targeting Viral Hemagglutinin at the Post-translational Level”. Journal of Biological Chemistry. 284 (43): 29798–29808. doi:10.1074/jbc.M109.029470. PMC 2785610. PMID 19638339.
- ^ White Jr, AC (2003). “Nitazoxanide: An important advance in anti-parasitic therapy”. Am. J. Trop. Med. Hyg. 68 (4): 382–383. doi:10.4269/ajtmh.2003.68.382. PMID 12875283.
- ^ Lateef, M.; Zargar, S. A.; Khan, A. R.; Nazir, M.; Shoukat, A. (2008). “Successful treatment of niclosamide- and praziquantel-resistant beef tapeworm infection with nitazoxanide”. International Journal of Infectious Diseases. 12 (1): 80–2. doi:10.1016/j.ijid.2007.04.017. PMID 17962058.
- ^ Cynthia Liu, Qiongqiong Zhou, Yingzhu Li, Linda V. Garner, Steve P. Watkins, Linda J. Carter, Jeffrey Smoot, Anne C. Gregg, Angela D. Daniels, Susan Jervey, Dana Albaiu. Research and Development on Therapeutic Agents and Vaccines for COVID-19 and Related Human Coronavirus Diseases. ACS Central Science 2020; doi:10.1021/acscentsci.0c00272
External links
- “Nitazoxanide”. MedlinePlus Drug Information. U.S. National Library of Medicine. 28 July 2010. Retrieved 2010-08-19.
- “Parasitic infections”. Am J Transplant. 4 (Suppl 10): 142–55. 2004. doi:10.1111/j.1600-6135.2004.00677.x. PMID 15504227.
 |
|
| Clinical data | |
|---|---|
| Trade names | Alinia, Nizonide, and others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a603017 |
| License data |
|
| Pregnancy category |
|
| Routes of administration |
Oral |
| Drug class | Antiprotozoal Broad-spectrum antiparasitic Broad-spectrum antiviral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | Nitazoxanide: ? Tizoxanide: over 99%[1][2] |
| Metabolism | Rapidly hydrolyzed to tizoxanide[1] |
| Metabolites | tizoxanide[1][2] tizoxanide glucuronide[1][2] |
| Elimination half-life | 3.5 hours[3] |
| Excretion | Renal, biliary, and fecal[1] |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| NIAID ChemDB | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.054.465 |
| Chemical and physical data | |
| Formula | C12H9N3O5S |
| Molar mass | 307.283 g/mol g·mol−1 |
| 3D model (JSmol) | |
//////////////nitazoxanide, corona virus, covid 19

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
