
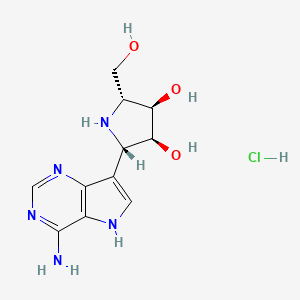
Galidesivir
- Molecular FormulaC11H15N5O3
- Average mass265.268 Da
Galidesivir
249503-25-1
222631-44-9, BCX-4430 (HCL salt form of galidesivir)
Galidesivir (BCX4430, Immucillin-A) is an antiviral drug, an adenosine analog[1] (a type of nucleoside analog).[2] It is developed by BioCryst Pharmaceuticals with funding from NIAID, originally intended as a treatment for hepatitis C, but subsequently developed as a potential treatment for deadly filovirus infections such as Ebola virus disease and Marburg virus disease.
It also shows broad-spectrum antiviral effectiveness against a range of other RNA virus families, including bunyaviruses, arenaviruses, paramyxoviruses, coronaviruses, flaviviruses and phleboviruses.[3] BCX4430 has been demonstrated to protect against both Ebola and Marburg viruses in both rodents and monkeys, even when administered up to 48 hours after infection,[1] and development for use in humans was then being fast-tracked due to concerns about the lack of treatment options for the 2013-2016 Ebola virus epidemic in West Africa.[4]
BCX4430 later showed efficacy against Zika virus in a mouse model, though there are no plans for human trials at this stage.[5]
Galidesivir is one of several antiviral drugs being tested for coronavirus disease 2019.[6]

CLIP
https://www.sciencedirect.com/science/article/pii/S0040402017305926

CLIP

When any new virus emerges, drug and vaccine developers spring into action, searching for products to stop it in its tracks. Drug discovery campaigns launch, vaccine development efforts ramp up, and everyone mobilizes to get it all into the clinic as quickly as possible.
The current pandemic, driven by a coronavirus known as SARS-CoV-2, is no different. Already, a Phase I study of an mRNA-based vaccine developed by Moderna has begun, and major pharma companies and small biotechs are working on other types of vaccines. But even if they work, the most optimistic timelines put a vaccine a year to 18 months away.
The more immediate approach to an outbreak is to scour the medicine cabinet for existing molecules that could be repurposed against a new virus. The most advanced potential treatment is Gilead Sciences’ remdesivir, an antiviral discovered during the 2014 Ebola epidemic. The compound is already being tested in four, Phase III trials—two in China and two in the US—against the respiratory disease COVID-19. Gilead expects the first dataset from those studies to come out in April.
A new paper from CAS explored remdesivir and other possible options the cabinet might contain (ACS Cent. Sci. 2020, DOI: 10.1021/acscentsci.0c00272). CAS, a division of the American Chemical Society, which publishes C&EN, looked at the landscape of patent and journal articles covering small molecules, antibodies, and other therapeutic classes to identify therapies with potential activity against COVID-19.
SARS-CoV-2, belongs to the same family as two coronaviruses responsible for earlier outbreaks, Severe Acute Respiratory Syndrome (SARS) and Middle East Respiratory Syndrome (MERS). Because all three feature structurally similar proteins that allow entry into and replication inside host cells, CAS searched for patent data related to those more well-studied coronaviruses.
C&EN has assembled the relevant small molecules identified by CAS, which can be explored by the stage in the viral life cycle they aim to disrupt.
Patents
| Patent ID | Title | Submitted Date | Granted Date |
|---|---|---|---|
| US7390890 | Inhibitors of nucleoside metabolism | 2007-08-23 | 2008-06-24 |
| US7211653 | Inhibitors of nucleoside metabolism | 2005-02-03 | 2007-05-01 |
| US6803455 | Inhibitors of nucleoside metabolism | 2003-05-22 | 2004-10-12 |
| US6492347 | Inhibitors of nucleoside metabolism | 2002-05-23 | 2002-12-10 |
| US6228847 | Inhibitors of nucleoside metabolism | 2001-05-08 |
| Patent ID | Title | Submitted Date | Granted Date |
|---|---|---|---|
| EP1023308 | INHIBITORS OF NUCLEOSIDE METABOLISM | 2000-08-02 | 2005-09-07 |
| US6066722 | Inhibitors of nucleoside metabolism | 2000-05-23 |
References
- ^ Jump up to:a b Warren TK, Wells J, Panchal RG, Stuthman KS, Garza NL, Van Tongeren SA, et al. (April 2014). “Protection against filovirus diseases by a novel broad-spectrum nucleoside analogue BCX4430” (PDF). Nature. 508 (7496): 402–5. Bibcode:2014Natur.508..402W. doi:10.1038/nature13027. PMID 24590073.
- ^ Kamat SS, Burgos ES, Raushel FM (October 2013). “Potent inhibition of the C-P lyase nucleosidase PhnI by Immucillin-A triphosphate”. Biochemistry. 52 (42): 7366–8. doi:10.1021/bi4013287. PMC 3838859. PMID 24111876.
- ^ Westover JB, et al. Galidesivir limits Rift Valley fever virus infection and disease in Syrian golden hamsters. Antiviral Res. 2018 Aug;156:38-45. Westover, J. B.; Mathis, A.; Taylor, R.; Wandersee, L.; Bailey, K. W.; Sefing, E. J.; Hickerson, B. T.; Jung, K. H.; Sheridan, W. P.; Gowen, B. B. (2018). “Galidesivir limits Rift Valley fever virus infection and disease in Syrian golden hamsters”. Antiviral Research. 156: 38–45. doi:10.1016/j.antiviral.2018.05.013. PMC 6035881. PMID 29864447.
- ^ Rodgers P (8 April 2014). “BioWar Lab Helping To Develop Treatment For Ebola”. Forbes Magazine.
- ^ Julander JG, Siddharthan V, Evans J, Taylor R, Tolbert K, Apuli C, et al. (January 2017). “Efficacy of the broad-spectrum antiviral compound BCX4430 against Zika virus in cell culture and in a mouse model”. Antiviral Research. 137: 14–22. doi:10.1016/j.antiviral.2016.11.003. PMC 5215849. PMID 27838352.
- ^ Praveen Duddu. Coronavirus outbreak: Vaccines/drugs in the pipeline for Covid-19. clinicaltrialsarena.com 19 February 2020.
 |
|
| Legal status | |
|---|---|
| Legal status | |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C11H15N5O3 |
| Molar mass | 265.268 g·mol−1 |
| 3D model (JSmol) | |
//////////////Galidesivir, Immucillin-A, OLF97F86A7, UNII:OLF97F86A7, галидесивир , غاليديسيفير , 加利司韦 , BCX4430, BCX 4430, CORONAVIRUS, COVID 19
nitazoxanide

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....

nice contribution dr Anthony
LikeLike