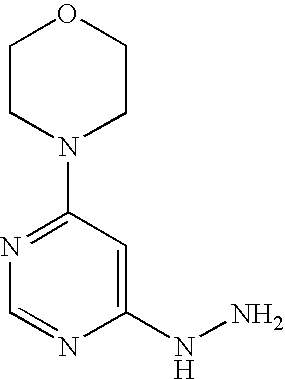
Molidustat
UNII-9JH486CZ13, cas no 1154028-82-6, MW: 314.3076
2-(6-morpholin-4-ylpyrimidin-4-yl)-4-(triazol-1-yl)-1H-pyrazol-3-one
Hypoxia-Inducible Factor Prolyl Hydroxylase Inhibitors
- Originator Bayer Schering Pharma
- Developer Bayer HealthCare Pharmaceuticals
- Class Antianaemics; Morpholines; Pyrazoles; Pyrazolones; Pyrimidines; Small molecules; Triazoles
- Mechanism of Action Hypoxia-inducible factor-proline dioxygenase inhibitors
- Phase III Anaemia
- 24 Jun 2018 Biomarkers information updated
- 23 Jun 2018 Bayer initiates enrolment in the MIYABI HD-M phase III trial for Anaemia in Japan (PO) (NCT03543657)
- 05 Jun 2018 Bayer plans a phase III trial for Anaemia (renal) in Japan in June 2018 (NCT03543657)
For the cardio-renal syndrome, a Phase IIb program with the investigational new drug Molidustat (BAY 85-3934) is under initiation in patients with anemia associated with chronic kidney disease and/or end-stage renal disease. Molidustat is a novel inhibitor of hypoxia-inducible factor (HIF) prolyl hydroxylase (PH) which stimulates erythropoietin (EPO) production and the formation of red blood cells. Phase I data have shown that inhibition of HIF-PH by Molidustat results in an increase in endogenous production of EPO.
About Bayer HealthCare
The Bayer Group is a global enterprise with core competencies in the fields of health care, agriculture and high-tech materials. Bayer HealthCare, a subgroup of Bayer AG with annual sales of EUR 18.6 billion (2012), is one of the world’s leading, innovative companies in the healthcare and medical products industry and is based in Leverkusen, Germany. The company combines the global activities of the Animal Health, Consumer Care, Medical Care and Pharmaceuticals divisions. Bayer HealthCare’s aim is to discover, develop, manufacture and market products that will improve human and animal health worldwide. Bayer HealthCare has a global workforce of 54,900 employees (Dec 31, 2012) and is represented in more than 100 countries. More information at www.healthcare.bayer.com.

Molidusat sodium
RN: 1375799-59-9
UNII: CI0NE7C96T
Molecular Formula, C13-H13-N8-O2.Na, Molecular Weight, 336.2897
Sodium 1-[6-(morpholin-4-yl)pyrimidin-4-yl]-4-(1H-1,2,3-triazol-1-yl)-1H-pyrazol-5-olate
Molidustat sodium is an orally-available hypoxia-inducible factor prolyl hydroxylase inhibitor in phase I clinical trials at Bayer for the treatment of patients suffering from renal anemia due to chronic kidney disease.
Molidustat (INN; Bay 85-3934) is a drug which acts as a HIF prolyl-hydroxylase inhibitor and thereby increases endogenous production of erythropoietin, which stimulates production of hemoglobin and red blood cells. It is in Phase III clinical trials for the treatment of anemia secondary to chronic kidney disease.[1][2] Due to its potential applications in athletic doping, it has also been incorporated into screens for performance-enhancing drugs.[3]
WO 2008067871
WO 2012065967
WO 2013167552
2-Heteroaryl-4-aryl-1,2-dihydropyrazolones having a bactericidal and/or fungicidal action are disclosed in EP 165 448 and EP 212 281. The use of 2-heteroaryl-4-aryl-1,2-dihydropyrazolones as lipoxygenase inhibitors for treatment of respiratory tract, cardiovascular and inflammatory diseases is claimed in EP 183 159. 2,4-Diphenyl-1,2-dihydropyrazolones having a herbicidal activity are described in DE 2 651 008.
The preparation and pharmacological properties of certain 2-pyridyl-1,2-dihydropyrazolones are reported in Helv. Chim. Acta 49 (1), 272-280 (1966). WO 96/12706, WO 00/51989 and WO 03/074550 claim compounds having a dihydropyrazolone partial structure for treatment of various diseases, and hydroxy- or alkoxy-substituted bipyrazoles for treatment of neuropsychiatric diseases are disclosed in WO 2006/101903.
Heteroaryl-substituted pyrazole derivatives for treatment of pain and various CNS diseases are furthermore described in WO 03/051833 and WO 2004/089303. WO 2006/114213 has meanwhile disclosed 2,4-dipyridyl-1,2-dihydropyrazolones as inhibitors of HIF prolyl 4-hydroxylases.
The x-ray crystal structure of the compound 3-methyl-1-(pyridin-2-yl)-4-(1-pyridin-2-yl-3-methyl-1H-pyrazol-5-yl)-2H-3-pyrazolin-5 (114)-one (other name: 5,5′-dimethyl-2,2′-di-pyridin-2-yl-1′,2′-dihydro-2H,3′H-3,4′-bipyrazol-3′-one) is reported inActa Crystallogr., Section E: Structure Reports Oμline E57 (11), o1126-o1127 (2001) [Chem. Abstr. 2001:796190].
The synthesis of certain 3′,5-dimethyl-2-phenyl-1′-(1,3-thiazol-2-yl)-1′H,2H-3,4′-bipyrazol-5′-ol derivatives is described inIndian J. Heterocyclic Chem. 3 (1), 5-8 (1993) [Chem. Abstr. 1994:323362].
The preparation and tautomerism of individual 4-(pyrazol-5-yl)-pyrazolin-5-one derivatives is reported in J. Heterocyclic Chem. 27 (4), 865-870 (1990) [Chem. Abstr. 1991:428557]. A therapeutic use has not hitherto been described for the compounds mentioned in these publications. The compound 2-tert-butyl-1′-[4-(4-chlorophenyl)-1,3-thiazol-2-yl]-3′,5-dimethyl-1′H,2H-3,4′-bipyrazol-5′-ol is listed as a test example in WO 2007/008541.
SYN

WO 2013167552

CLIP
https://onlinelibrary.wiley.com/doi/pdf/10.1002/cmdc.201700783
1-[6-(Morpholin-4-yl)pyrimidin-4-yl]-4-(1H-1,2,3-triazol-1-yl)-1Hpyrazol-5-ol (molidustat, BAY 85-3934, 45): Method A (gram-scale): Ethyl 3-(dimethylamino)-2-(1H-1,2,3-triazol-1-yl)acrylate (73, 1.98 g, 9.43 mmol) and 4-(6-hydrazinopyrimidin-4-yl)morpholine (78, 1.89 g, 9.70 mmol) were introduced into ethyl acetate (25 mL) and TFA (502 mg, 4.4 mmol) was added at RT. The mixture was stirred under reflux for 18 h, then cooled to 0–58C and subsequently stirred for a further 2 h. The solid formed was filtered off, washed with cold ethyl acetate and dried first in air and thereafter under a high vacuum. Yield: 2.13 g (71%);
1H NMR (400 MHz, [D6 ]DMSO): d=8.42 (s, 1H), 8.38 (s, 1H), 8.01 (s, 1H), 7.73 (s, 1H), 7.70 (s, 1H), 3.71–3.65 (m, 4H), 3.57–3.51 ppm (m, 4H);
13C NMR (125 MHz, [D6 ]DMSO): d=44.3, 65.6, 85.6, 102.8, 123.7, 132.9, 135.8, 152.4, 154.1, 154.7, 162.0 ppm;
IR (KBr): n˜ =3441, 3135–3108, 2965–2884, 1636–1345, 1257 cm@1 ;
UV/Vis (acetonitrile/water 1:1): lmax (e)= 249 nm (34928 L (mol cm)@1 );
MS (EI+) m/z: 315 [M+H]+ ;
Anal. calcd for C13H14N8O2 : C 49.7, H 4.5, N 35.7, O 10.2, found: C 49.5, H 4.4, N 35.5, O 12.6.
Method B (kilogram-scale): Inastirred vessel, 4- (6-hydrazinopyrimidin-4-yl)morpholine (78, 42.0 kg, 215.1 mol) and methyl 3-(dimethylamino)-2-(1H-1,2,3-triazol-1-yl)acrylate (83, 44.0 kg, 224.2 mol) were suspended in ethyl acetate (378 kg), admixed with TFA (12.1 kg, 106.1 mol) and heated under reflux (from 788C to 81 8C) at a jacket temperature of 908C for 26 h. The suspension obtained was cooled to 0 8C, stirred at 08C for 1 h and filtered. The filter cake was washed with ethyl acetate (53 kg) and dried under reduced pressure at up to 458C. The filter cake was admixed with a mixture of water (355 kg) and acetic acid (11.7 kg), then suspended and stirred at 50–548C for 1 h. After cooling to 248C, the suspension was filtered. The filter cake was washed first with water (90 kg), then twice with methanol (50 kg each time) and finally dried at 35–458C under reduced pressure. Yield: 57.4 kg (85%)
Synthesis of molidustat sodium (84)
Sodium 1-[6-(morpholin-4-yl)pyrimidin-4-yl]-4-(1 H-1,2,3-triazol1-yl)-1H-pyrazol-5-olate (molidustat sodium, 84): Kilogram scale: In a stirred vessel, compound 45 (55 kg, 175.0 mol) was suspended in a mixture of methanol (200 kg) and water (30 kg), admixed with triethylamine (17.8 kg, 175.9 mmol), heated at 608C, stirred further for about 1 h and filtered hot to separate off undissolved constituents. The filter cake was washed with methanol (15 kg, 608C). Sodium hydroxide solution (18.7 kg, 210.4 mmol, 45% strength) was slowly introduced at 608C and methanol (5 kg) was added. Sodium 1-[6-(morpholin-4-yl)pyrimidin-4-yl]-4-(1H-1,2,3-triazol-1-yl)- 1H-pyrazol-5-olate (84, 0.12 kg) was added as seed crystals and the mixture was stirred at 608C for another 1 h and cooled to 248C over a period of about 2 h. The mixture was stirred for 8 h at this temperature, subsequently cooled to 08C over a period of about 1 h and filtered in portions by means of a centrifuge. The filter cake was washed with a mixture of water (24 kg) and methanol (168 kg) and also methanol (about 23 kg in each case) and dried all together at 40 8C under reduced pressure in a dryer for 8 h. Yield: 57.6 kg (98%);
1H NMR (500 MHz, [D6 ]DMSO): d=8.98 (d, J= 1.4 Hz, 1H), 8.72 (s, 1H), 8.68 (s, 1H), 8.64 (d, J=1.4 Hz, 1H), 7.77 (s, 1H), 4.25–4.00 ppm (m, 8H);
13C NMR (125 MHz, [D6 ]DMSO): d= 48.2, 67.8, 91.5, 107.0, 129.6, 130.9, 138.0, 151.7, 152.0, 157.4, 159.9 ppm;
IR (KBr): n˜ =3153–3006, 2976–2855, 1630–1439, 1241, 1112, 987 cm@1 ;
UV/Vis (acetonitrile/water 1:1): lmax (e)=284 nm (16855 L [mol cm]@1 );
MS (EI+) m/z: 337 [M+Na]+ , 315 [M+H]+ ;
Anal. calcd for C13H13N8O2Na: C 46.4, H 3.9, N 33.3, found: C 46.1, H 4.0, N 33.1.
PATENT
RM 1
Example 3A 3-(Dimethylamino)-2-(1H-1,2,3-triazol-1-yl)acrylic acid ethyl ester
The preparation of the starting compound is carried out analogously to 2A starting from 1.00 g (6.45 mmol) 2-(1H-1,2,3-triazol-1-yl)acetic acid ethyl ester.
Yield: 1.4 g (100% of th.)
1H-NMR (400 MHz, DMSO-d6): δ=8.10 (d, 1H), 7.78 (d, 1H), 7.65 (s, 1H), 4.03 (q, 2H), 3.06 (br. s, 3H), 2.10 (br. s, 3H), 1.12 (t, 3H).
LC-MS (Method 5): Rt=1.40 min; MS (ESIpos): m/z=211 [M+H]+.
…………
RM 2
Example 16A 4-(6-Hydrazinopyrimidin-4-yl)morpholine
Stage a):
4-(6-Chloropyrimidin-4-yl)morpholine
45.0 g (302.1 mmol) 4,6-dichloropyrimidine are initially introduced into 450 ml water. 26.3 g (302.1 mmol) morpholine are added and the mixture is stirred at 90° C. for 16 h. Thereafter, it is cooled to 0° C. and the precipitate formed is filtered off. The precipitate is washed once with 50 ml water and dried in air.
Yield: 51.0 g (85% of th.)
LC-MS (Method 4): Rt=1.09 min; MS (ESIpos): m/z=200 [M+H]+;
1H-NMR (400 MHz, DMSO-d6): δ=8.35 (s, 1H), 6.95 (s, 1H), 3.62 (s, 8H).
Stage b)
4-(6-Hydrazinopyrimidin-4-yl)morpholine
53.0 g (2.7 mmol) 4-(6-chloropyrimidin-4-yl)morpholine are initially introduced into 260 ml ethanol. 132.9 g (2.7 mol) hydrazine hydrate are added and the mixture is stirred under reflux for 16 h. Thereafter, it is cooled to RT and approx. half of the solvent is removed by distillation. The mixture is cooled to 0° C. and the solid formed is filtered off. It is rinsed with cold ethanol and the solid is dried first in air and then in vacuo.
Yield: 35.0 g (68% of th.)
LC-MS (Method 1): Rt=0.17 min; MS (ESIpos): m/z=196 [M+H]+;
1H-NMR (400 MHz, DMSO-d6): δ=7.94 (s, 1H), 7.70 (s, 1H), 5.91 (s, 1H), 4.15 (s, 2H), 3.66-3.60 (m, 4H), 3.45-3.37 (m, 4H).
………..
Example 71
2-(6-Morpholin-4-ylpyrimidin-4-yl)-4-(1H-1,2,3-triazol-1-yl)-1,2-dihydro-3H-pyrazol-3-one
1.9 g (8.8 mmol) of the compound from Example 3A and 1.9 g (9.7 mmol) of the compound from Example 16A are initially introduced into 25 ml ethyl acetate and 504 mg (4.4 mmol) TFA are added at RT. The mixture is stirred under reflux for 16 h, then cooled to 5° C. and subsequently stirred for a further 2 h. The solid formed is filtered off, washed with ethyl acetate and dried first in air and thereafter under a high vacuum. 1.7 g of product are obtained.
The mother liquor is combined with the wash solution and the solvent is removed. According to LC-MS, the residue (2.4 g) still contains the intermediate 3-[2-(6-morpholin-4-ylpyrimidin-4-yl)hydrazino]-2-(1H-1,2,3-triazol-1-yl)prop-2-enoic acid ethyl ester (intermediate stage of the cyclization), which is used directly for the preparation of Example 72 (see there).
Yield: 1.7 g (61% of th.)
LC-MS (Method 9): Rt=0.90 min; MS (ESIpos): m/z=315 [M+H]+;
1H-NMR (400 MHz, DMSO-d6): δ=8.42 (s, 1H), 8.38 (s, 1H), 8.01 (s, 1H), 7.73 (s, 1H), 7.70 (s, 1H), 3.71-3.65 (m, 4H), 3.57-3.51 (m, 4H).
………..
Hydrochloride
Example 72
2-(6-Morpholin-4-ylpyrimidin-4-yl)-4-(1H-1,2,3-triazol-1-yl)-1,2-dihydro-3H-pyrazol-3-one hydrochloride
Batch 1: 7.5 ml of a 4 N solution of hydrogen chloride in dioxane are added to 1.7 g (5.4 mmol) of the compound from Example 71. The mixture is stirred at RT, 5 ml dioxane are added and the mixture is stirred at RT for 16 h. The solid is filtered off and washed with 5 ml dioxane. The mixture is dried under a high vacuum for 16 h, 10 ml methanol are then added and the mixture is stirred at RT for 1 h. The solid is filtered off, washed with 4 ml methanol and dried under a high vacuum. 1.6 g of the title compound are obtained.
Batch 2: A further amount of the title compound is obtained as follows: The residue (2.4 g) obtained from the mother liquor during the synthesis of Example Compound 71, which contains the open-ring intermediate state of the cyclization, 3-[2-(6-morpholin-4-ylpyrimidin-4-yl)hydrazino]-2-(1H-1,2,3-triazol-1-yl)prop-2-enoic acid ethyl ester, is dissolved in 12 ml ethanol and 1.5 ml 30% strength sodium methylate solution in methanol are added at RT, while stirring. The mixture is subsequently stirred at RT for 45 min, then adjusted to pH 5 with 2 N hydrochloric acid and subsequently stirred at RT for a further 16 h. The mixture is cooled to 10° C. and the solid is filtered off and washed with 3.5 ml dioxane. The mixture is dried under a high vacuum for 16 h, 5 ml methanol are then added and the mixture is subsequently stirred at RT for 1 h. The solid is filtered off, washed with 2 ml methanol and dried under a high vacuum to give a further 997 mg of the title compound in this way.
Yield: together 2.6 g (83% of th.)
LC-MS (Method 6): Rt=0.89 min; MS (ESIpos): m/z=315 [M+H]+;
1H-NMR (400 MHz, DMSO-d6): δ=8.54 (s, 1H), 8.39 (s, 1H), 8.28 (s, 1H), 7.88 (s, 1H), 7.42 (s, 1H), 3.71 (s, 8H).
References
- Jump up^ Flamme, I; Oehme, F; Ellinghaus, P; Jeske, M; Keldenich, J; Thuss, U (2014). “Mimicking hypoxia to treat anemia: HIF-stabilizer BAY 85-3934 (Molidustat) stimulates erythropoietin production without hypertensive effects”. PLoS ONE. 9 (11): e111838. Bibcode:2014PLoSO…9k1838F. doi:10.1371/journal.pone.0111838. PMC 4230943. PMID 25392999.
- Jump up^ Gupta, Nupur; Wish, Jay B (2017). “Hypoxia-Inducible Factor Prolyl Hydroxylase Inhibitors: A Potential New Treatment for Anemia in Patients with CKD”. American Journal of Kidney Diseases. 69 (6): 815. doi:10.1053/j.ajkd.2016.12.011. PMID 28242135.
- Jump up^ Dib, Josef; Mongongu, Cynthia; Buisson, Corinne; Molina, Adeline; Schänzer, Wilhelm; Thuss, Uwe; Thevis, Mario (2017). “Mass spectrometric characterization of the hypoxia-inducible factor (HIF) stabilizer drug candidate BAY 85-3934 (molidustat) and its glucuronidated metabolite BAY-348, and their implementation into routine doping controls”. Drug Testing and Analysis. 9 (1): 61–67. doi:10.1002/dta.2011. PMID 27346747.
 |
|
| Clinical data | |
|---|---|
| Synonyms | Bay 85-3934 |
| ATC code |
|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| UNII | |
| Chemical and physical data | |
| Formula | C13H14N8O2 |
| Molar mass | 314.31 g·mol−1 |
| 3D model (JSmol) | |
//////////Molidustat, Bay 85-3934

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....