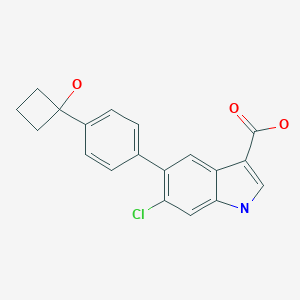
PF-06409577
6-Chloro-5-[4-(1-hydroxycyclobutyl)phenyl]-1H-indole-3-carboxylic acid
CAS Number 1467057-23-3, C19H16ClNO3, 341.79
Biochem/physiol Actions
PF-06409577 is a potent and selective activator of 5′ adenosine monophosphate-activated protein kinase (AMPK).
PF-06409577 potently activates a1β1γ1 AMPK (5′ adenosine monophosphate-activated protein kinase) isoform, and prevents its dephosphorylation. It is similarly potent for β1 containing isoforms, but shows significantly lower potency for β2-containing isoforms of AMPK. Patch-clamp assays show that this compound does not inhibit hERG (human ether-a-go-go gene). It interacts with the allosteric drug and metabolite site (ADaM) of AMPK.
General description
PF-06409577 is a 6-chloro-indole derivative obtained from 5-bromo-6-chloro-indole.
PF-06409577 is a potent and selective activator of 5′ adenosine monophosphate-activated protein kinase (AMPK) for the Potential Treatment of diabetic nephropathy. PF-06409577 has AMPK α1β1γ1 Kd=9.0 nM. AMPK α1β1γ1 EC50 = 7.0 nM; AMPK α1β2γ1 EC50 > 40000 nM. PF-06409577 showed efficacy in a preclinical model of diabetic nephropathy. Upon the basis of its potent and selective AMPK activation, low metabolic turnover in human hepatocytes, clean off-target profile, and favorable preclinical in vivo efficacy results, PF-06409577 was profiled in regulatory toxicology studies and was subsequently advanced to clinical trials to assess human pharmacokinetics and safety/ tolerability.
Diabetes is a major public health concern because of its increasing prevalence and associated health risks. The disease is characterized by high levels of blood glucose resulting from defects in insulin production, insulin action, or both. Two major forms of diabetes are recognized, type I and type II. Type I diabetes develops when the body’s immune system destroys pancreatic beta cells, the only cells in the body that make the hormone insulin that regulates blood glucose. To survive, people with type 1 diabetes must have insulin delivered by injection or a pump. Type II diabetes accounts for about 90 to 95 percent of all diagnosed cases of diabetes. Type II diabetes usually begins as insulin resistance, a disorder in which the cells do not use insulin properly. Key target tissues, including liver, muscle, and adipose tissue, are resistant to the effects of insulin in stimulating glucose and lipid metabolism. As the need for insulin rises, the pancreas gradually loses its ability to produce insulin. Controlling type II diabetes with medication is essential; otherwise it can progress into pancreatic beta-cell failure requiring complete dependence on insulin.
Obesity increases the risk of type II diabetes as well as many other health conditions including coronary heart disease, stroke, and high blood pressure. More than one-third of U.S. adults (over 72 million people) and 17% of U.S. children are obese. During 1980-2008, obesity rates doubled for adults and tripled for children. During the past several decades, obesity rates for all population groups— regardless of age, sex, race, ethnicity, socioeconomic status, education level, or geographic region— have increased markedly.
Research has identified the enzyme 5′ adenosine monophosphate-activated protein kinase (AMPK) as a regulator of cellular and whole-body energy homeostasis. AMPK is activated by cellular stress resulting in downstream events that serve to conserve or generate ATP. AMPK is composed of three distinct subunits, each with multiple isoforms: the alpha subunit (alpha 1 or 2); the beta subunit (beta 1 or 2); and the gamma subunit (gamma 1, 2, or 3); for a total of twelve possible heterotrimeric isoforms.
In the liver, activated AMPK phosphorylates a variety of substrates including 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase (Clarke, P.R. & Hardie, D.G., EMBO J 9, 2439-2446 (1990)) and acetyl-CoA carboxylase (Carling, D. et al. FEBS Letters 223, 217-222 (1987)) which inhibits cholesterol biosynthesis and decreases fatty acid synthesis, respectively. Therefore, activation of AMPK should lead to decreases in the levels of triglycerides and cholesterol. AMPK is also thought to regulate plasma glucose levels by decreasing hepatic gluconeogenesis through downregulation of key gene products following phosphorylation of CRTC2 (Koo S.H. et. AL, Nature 437, 1109-1111 (2005)). In muscle and myocardial tissues, AMPK activates the transport activity of glucose transporter 4 (GLUT4) increasing glucose uptake into cells thereby producing an additional avenue for decreasing plasma glucose (Kurth-Kraczek, E.J. et. al., Diabetes 48, 1667-1671 (1999)). AMPK activation has also been shown to enhance mitochondrial biogenesis improving fatty acid oxidation and decreasing circulating lipids (Merrill, G.M. et. al., Am. J. Physiol. 273, E1107-E1112 (1997)). Direct activation of AMPK using AICAR (5-aminoimidazole-4-carboxamide riboside) has been shown to lead to beneficial effects on several metabolic endpoints including improved glucose disposal, decreased hepatic glucose output and decreases in plasma triglycerides and free fatty acids (Song, X.M. et. al., Diabetologia 45, 56-65 (2002); Bergeron, R. et. al., Diabetes 50, 1076-1082 (2001); Buhl, E.S.et. al., Diabetes 50, 12-17 (2001); Iglesias, M.A. et. al., Diabetes 51, 2886-2894 (2002), Fogarty, S. & Hardie, D.G., Biochim et Biophys Acta 1804, 581-591 (2010)). Because of AMPK’s pluripotent effects on carbohydrate, lipid, and cholesterol metabolism and biosynthesis, agents that activate AMPK are attractive therapeutic targets for treating metabolic syndrome disorders such as diabetes, obesity, and dyslipidemia.
Decreases in renal AMPK activation have been implicated in the etiology of diseases of the kidney, including diabetic nephropathy, acute kidney injury (AKI), and polycystic kidney disease (PKD); activation of AMPK through hormonal (adiponectin) or pharmacological (AICAR) mechanisms has been shown to be protective in rodent models of these diseases. In diabetic nephropathy decreased AMPK activation in podocytes occurs early in the disease and is associated with increased expression of the NADPH-Oxidase protein Nox4 and increased proteinuria. These effects were reduced following administration of the AMPK activators AICAR, metformin, and Adiponectin (Lee, MJ. et.al. American Journal of Physiology – Renal Physiology. 292.
F617-F627 (2007); Sharma, K. et.al. Journal of Clinical Investigation.118. 1645-1656. (2008)). In ischemia/reperfusion models of AKI the AMPK activators metformin and AICAR were shown to dose-dependently reduce subsequent proteinuria, oxidative tissue damage, and kidney macrophage infiltration (Lempiainen, J. et.al. British Journal of Pharmacology 166. 1905-1915 (2012); Seo-Mayer, P.W. et.al. American Journal of Physiology – Renal Physiology, 301, F1346-F1357 (2011)). In two rodent models of PKD the AMPK activator metformin was shown to reduce renal cyst expansion (Takiar, V. et. al. PNAS 108, 2462-2467 (2011)). These studies suggest a broad benefit of AMPK activators in multiple renal diseases.
The compounds of the present invention activate AMPK and are, therefore, useful in treating metabolic disorders such as diabetes, obesity, and dyslipidemia as well as the renal diseases chronic kidney disease, diabetic nephropathy, acute kidney injury and polycystic kidney disease.
PATENT
US 20130267493
WO 2014140704
Example 5
6-Chloro-5-(4-(3-hydroxyoxetan-3-yl)phenyl)-1H-indole-3-carboxylic acid

Step 1
6-chloro-5-(4-(3-hydroxyoxetan-3-yl)phenyl)-1H-indole-3-carbaldehyde
A mixture of 5,5,5′,5′-tetramethyl-[2,2′]bi[[1,3,2]dioxaborinanyl] (149.0 mg, 0.44 mmol), oven dried potassium acetate (173.0 mg, 1.75 mmol) and 3-(4-bromo-phenyl)-oxetan-3-ol (100.0 mg, 0.44 mmol) in 1,4-dioxane (2 mL) was degassed with N2 for 5 minutes, treated with [1,1′-bis(diphenylphosphino)ferrocene]dichloropalladium(II) (33.0 mg, 0.044 mmol) and subjected to microwave irradiation at 110 °C for 1 hour. The cooled reaction mixture was filtered through celite and concentrated in vacuo to give a black oil. To the dark oil was added 5-bromo-6-chloro-1H-indole-3-carbaldehyde (112.0 mg, 0.43 mmol), 2 N aqueous potassium carbonate (0.4 mL, 0.80 mmol), toluene (1.5 mL) and EtOH (0.5 mL). The reaction mixture was degassed with N2 for 10 minutes, treated with [1, 1′-bis(diphenylphosphino)ferrocene] dichloropalladium(II) (25.0 mg, 0.034 mmol), and heated in a pressure tube to 110 °C for 2 hours. The cooled reaction mixture was purified by flash chromatography (33-100% EtOAc/ heptanes) to give a solid. The solid was triturated in MeOH and filtered to afford the title compound (50 mg, 35%) as a yellow solid. MS (ES+) 328.0 (M+H)+. 1H NMR (400 MHz, DMSO-d6) δ 12.23 (s, 1 H), 9.92 (s, 1 H), 8.35 (s, 1 H), 8.02 (s, 1 H), 7.66 (d, J = 9.4 Hz, 2 H), 7.44 (d, J = 8.2 Hz, 2 H), 6.36 (s, 1 H), 4.80 – 4.76 (m, 2 H), 4.75 – 4.71 (m, 2 H).
Step 2
6-Chloro-5-(4-(3-hydroxyoxetan-3-yl)phenyl)-1 H-indole-3-carboxylic acid To the mixture of 6-chloro-5-[4-(3-hydroxy-oxetan-3-yl)-phenyl]-1H-indole-3-carbaldehyde (50.0 mg, 0.15 mmol) in MeCN (2 mL) was added 2-methyl-2-butene (2.0 mL, 13.7 mmol), followed by sodium chlorite (138 mg, 1.53 mmol) and sodium phosphate monobasic hydrate (211.0 mg, 1.53 mmol) in water (1 mL). The reaction mixture was stirred at room temperature for 20 hours, and concentrated in vacuo. The residue was acidified with 1 N aqueous citric acid (1 mL) and extracted with EtOAc. The organic layer was dried over MgSO4 and concentrated in vacuo. The crude material was purified by flash chromatography (34-80% EtOAc/heptanes, with 0.2% formic acid modifier) to afford the title compound (18 mg, 34%) as a brown solid. MS (ES-) 342.3 (M-H)-. 1H NMR (400 MHz, CD3OD) δ 8.02 (s, 1 H), 7.98 (s, 1 H), 7.66 (d, J = 8.20 Hz, 2 H), 7.56 (s, 1 H), 7.47 (d, J = 8.20 Hz, 2 H), 4.87 – 4.80 (m, 4 H).
Paper
Discovery and Preclinical Characterization of 6-Chloro-5-[4-(1-hydroxycyclobutyl)phenyl]-1H-indole-3-carboxylic Acid (PF-06409577), a Direct Activator of Adenosine Monophosphate-activated Protein Kinase (AMPK), for the Potential Treatment of Diabetic Nephropathy. Cameron KO et al Journal of Medicinal Chemistry 59(17), 8068-8081, (2016)
Adenosine monophosphate-activated protein kinase (AMPK) is a protein kinase involved in maintaining energy homeostasis within cells. On the basis of human genetic association data, AMPK activators were pursued for the treatment of diabetic nephropathy. Identification of an indazole amide high throughput screening (HTS) hit followed by truncation to its minimal pharmacophore provided an indazole acid lead compound. Optimization of the core and aryl appendage improved oral absorption and culminated in the identification of indole acid, PF-06409577 (7). Compound 7 was advanced to first-in-human trials for the treatment of diabetic nephropathy.
Discovery and Preclinical Characterization of 6-Chloro-5-[4-(1-hydroxycyclobutyl)phenyl]-1H-indole-3-carboxylic Acid (PF-06409577), a Direct Activator of Adenosine Monophosphate-activated Protein Kinase (AMPK), for the Potential Treatment of Diabetic Nephropathy
ACS Editors’ Choice – This is an open access article published under an ACS AuthorChoice License, which permits copying and redistribution of the article or any adaptations for non-commercial purposes.
6-Chloro-5-[4-(1-hydroxycyclobutyl)phenyl]-1H-indole-3-carboxylic Acid (7)
PAPER
Evolution of the Synthesis of AMPK Activators for the Treatment of Diabetic Nephropathy: From Three Preclinical Candidates to the Investigational New Drug PF-06409577
https://pubs.acs.org/doi/10.1021/acs.oprd.8b00059

Indole acids 1, 2, and 3 are potent 5′-adenosine monophosphate-activated protein kinase (AMPK) activators for the potential treatment of diabetic nephropathy. Compounds 1–3 were scaled to supply material for preclinical studies, and indole 3 was selected for advancement to first-in-human clinical trials and scaled to kilogram quantities. The progression of the synthesis strategy for these AMPK activators is described, as routes were selected for efficient structure–activity relationship generation and then improved for larger scales. The developed sequences employed practical isolations of intermediates and APIs, reproducible cross-coupling, hydrolysis, and other transformations, and enhanced safety and purity profiles and led to the production of 40–50 g of 1and 2 and 2.4 kg of 3. Multiple polymorphs of 3 were observed, and conditions for the reproducible formation of crystalline material suitable for clinical development were identified.
Mp: 192–194 °C. 1H NMR (400 MHz, DMSO-d6): δ 12.12 (s, 1H), 11.94 (br d, J = 2.2 Hz, 1H), 8.08 (d, J = 2.9 Hz, 1H), 7.95 (s, 1H), 7.64 (s, 1H), 7.57 (d, J = 8.3 Hz, 2H), 7.40 (d, J = 8.1 Hz, 2H), 5.52 (s, 1H), 2.48–2.40 (m, 2H), 2.35–2.26 (m, 2H), 2.00–1.89 (m, 1H), 1.74–1.63 (m, 1H). 13C NMR (101 MHz, DMSO-d6): δ 165.6, 146.6, 138.1, 136.0, 133.8, 133.0, 129.2, 125.6, 125.3, 124.6, 122.8, 112.9, 107.6, 75.1, 37.3, 12.8. MS (ES): calcd for C19H17ClNO3 ([M – H]−) 340.1; found 340.3. Anal. Calcd (%): C, 66.77; H, 4.72; N, 4.10. Found: C, 66.59; H, 4.71; N, 3.96.
///////////////////PF-06409577, PHASE 1
O=C(C1=CNC2=C1C=C(C3=CC=C(C4(O)CCC4)C=C3)C(Cl)=C2)O

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....