| MF | C 1 4 H 1 2 BrNO 2 S |
|---|---|
| MW | 338.21958 g / mol |
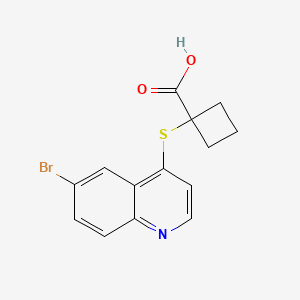
1- (6-bromoquinolin-4-yl) sulfanylcyclobutane-1-carboxylic acid
CAS…….1638327-48-6
Cyclobutanecarboxylic acid, 1-[(6-bromo-4-quinolinyl)thio]-
COMING ………….
MS m / z (ESI): 338.0 [M + l]
1H NMR (400 MHz, DMSO) δ 13.17 (s, 1H), 8.75-8.79 (m, 1H), 8.24 (s, 1H), 7.87-7.98 (m, 2H), 7.21-7.25 (m, 1H), 2.83-2.95 (m, 2H), 2.30-2.41 (m, 2H), 2.16-2.27 (m, 1H), 1.97-2.08 (m, 1H)
WO-2014183555-A1 / 2014-11-20
http://www.google.co.in/patents/WO2014183555A1?cl=en
PROCEDURE
6-bromo-quinoline-4-thiol
A mixture of 6-bromo-4-chloro-quinoline 3a (260 mg, 1.1 mmol, using known methods “Bioorganic &
Medicinal Chemistry Letters, 2012, 22 (4), 1569-1574 “prepared to give) and sodium sulfide (100 mg, 1.3 mmol) was added to 4 mL of N, N- dimethyl formamide, plus complete, heated 80 ° C, the reaction was stirred for 2 hours. To the reaction mixture was added 50 mL of water, 1 M hydrochloric acid was added dropwise to the reaction solution to pH 5-6, extracted with ethyl acetate (50 mL X 3), the combined organic phases, with no over anhydrous sodium sulfate, filtered, and the filtrate concentrated under reduced pressure to give the title product 6-bromo-quinolin-4-thiol 3b (257 mg, yellow oil), it was used directly in the next reaction.
The second step
L – ((6-bromo-quinolin-4-yl) thio) cyclobutyl carboxylate
Under an argon atmosphere, 6-bromo-quinolin-4-thiol 3b (257 mg, 1.1 mmol), 1- bromo-cyclobutyloxy embankment carboxylate (266 mg, 1.3 mmol) and cesium carbonate (371 mg, 1.1 mmol) were sequentially added to 5 mL of N, N- dimethylformamide and heated to 60 ° C, the reaction was stirred for 2 hours. The reaction solution was filtered, the filter cake washed with ethyl acetate (10 mL X 3) and the filtrate was concentrated under reduced pressure to give the title product l – ((6-bromo-quinolin-4-yl) thio) ethyl cyclobutyl 3c ( 300 mg, brown oil). Yield: 77%.
MS m / z (ESI): 368.2 [M + l]
1H MR (400 MHz, CDCl 3 ) [delta] 8.67 (d, = 4.77 Hz, IH), 8.31 (d, = 2.13 Hz, IH), 7.94 (d, = 8.91Hz, IH), 7.78 (dd, = 9.03, 2.13Hz, IH), 7.15 (d, = 4.89Hz, IH), 4.16 (q, = 7.15Hz, 2H), 2.86-3.04 (m, 2H), 2.39-2.51 (m, 2H), 2.25-2.37 ( m, IH), 2.00-2.15 (m, IH), 1.16 (t, = 7.09Hz, 3H)
third step
L – ((6-bromo-quinolin-4-yl) thio) cyclobutyl acid
L – ((6-bromo-quinolin-4-yl) thio) ethyl cyclobutyl 3c (100 mg, 0.27 mmol) and lithium hydroxide monohydrate (23 mg, 0.55 mmol) was dissolved in 6 mL of tetrahydrofuran, ethanol and water (^ = 4: 1: 1) mixed solvent, the reaction was stirred for 3 hours. 1M hydrochloric acid was added dropwise to the reaction solution pH of 5 to 6, liquid separation, the aqueous phase was extracted (10 mL X 3) with dichloromethane, the combined organic phases, the organic phase was washed with a saturated sodium chloride solution (10 mL XI), dried over anhydrous sodium sulfate, filtered, and the filtrate was concentrated under reduced pressure, the resulting A by thin layer chromatography in a developing solvent system, and the residue was purified to give the title product l – ((6-bromo-quinolin-4-yl) thio) cyclobutyl acid 3 (20 mg, white solid), yield: 22%.
MS m / z (ESI): 338.0 [M + l]
1H NMR (400 MHz, DMSO) δ 13.17 (s, 1H), 8.75-8.79 (m, 1H), 8.24 (s, 1H), 7.87-7.98 (m, 2H), 7.21-7.25 (m, 1H), 2.83-2.95 (m, 2H), 2.30-2.41 (m, 2H), 2.16-2.27 (m, 1H), 1.97-2.08 (m, 1H)
L – ((6-bromo-quinolin-4-yl) thio) cyclobutyl acid
First step
6-bromo-quinoline-4-thiol
A mixture of 6-bromo-4-chloro-quinoline 3a (260 mg, 1.1 mmol, a known method of “Bioorganic &
Medicinal Chemistry Letters, 2012, 22 (4), 1569-1574 “prepared to give) and sodium sulfide (100 mg, 1.3 mmol) was added to 4 mL of N, N- dimethyl formamide, plus complete, heated 80 ° C, the reaction was stirred for 2 hours. To the reaction mixture was added 50 mL of water, 1 M hydrochloric acid was added dropwise to the reaction solution to pH 5-6, extracted with ethyl acetate (50 mL X 3), the combined organic phases, with no over anhydrous sodium sulfate, filtered, and the filtrate concentrated under reduced pressure to give the title product 6-bromo-quinolin-4-thiol 3b (257 mg, yellow oil), it was used directly in the next reaction.
The second step
L – ((6-bromo-quinolin-4-yl) thio) ethyl cyclobutyl
Under an argon atmosphere, 6-bromo-quinolin-4-thiol 3b (257 mg, 1.1 mmol), 1- bromo-cyclobutyloxy embankment carboxylate (266 mg, 1.3 mmol) and cesium carbonate (371 mg, 1.1 mmol) were added to 5 mL of N, N- dimethylformamide and heated to 60 ° C, the reaction was stirred for 2 hours. The reaction mixture was filtered, the filter cake washed with ethyl acetate (10 mL X 3) and the filtrate was concentrated under reduced pressure to give the title product l – ((6-bromo-quinolin-4-yl) thio) ethyl cyclobutyl 3c ( 300 mg, brown oil). Yield: 77%.
MS m / z (ESI): 368.2 [M + l]
1H MR (400 MHz, CDC1 3) δ 8.67 (d, = 4.77Hz, IH), 8.31 (d, = 2.13Hz, IH), 7.94 (d, = 8.91Hz, IH), 7.78 (dd, = 9.03, 2.13Hz, IH), 7.15 (d, = 4.89Hz, IH), 4.16 (q, = 7.15Hz, 2H), 2.86-3.04 (m, 2H), 2.39-2.51 (m, 2H), 2.25-2.37 ( m, IH), 2.00-2.15 (m, IH), 1.16 (t, = 7.09Hz, 3H) Step
L – ((6-bromo-quinolin-4-yl) thio) cyclobutyl acid
L – ((6-bromo-quinolin-4-yl) thio) ethyl cyclobutyl 3c (100 mg, 0.27 mmol) and lithium hydroxide monohydrate (23 mg, 0.55 mmol) was dissolved in 6 mL of tetrahydrofuran, ethanol and water (^ = 4: 1: 1) mixed solvent, the reaction was stirred for 3 hours. 1M hydrochloric acid was added dropwise to the reaction solution pH of 5 to 6, liquid separation, the aqueous phase was extracted (10 mL X 3) with dichloromethane, the combined organic phases, the organic phase was washed with a saturated sodium chloride solution (10 mL XI), dried over anhydrous sodium sulfate, filtered, and the filtrate was concentrated under reduced pressure, to the resulting thin layer chromatography using a developing solvent system A and the residue was purified to give the title product l – ((6-bromo-quinolin-4-yl) thio) cyclobutyl acid 3 (20 mg, white solid), yield: 22%. MS m / z (ESI): 338.0 [M + l]
1H NMR (400 MHz, DMSO) δ 13.17 (s, 1H), 8.75-8.79 (m, 1H), 8.24 (s, 1H), 7.87-7.98 (m, 2H), 7.21-7.25 (m, 1H), 2.83-2.95 (m, 2H), 2.30-2.41 (m, 2H), 2.16-2.27 (m, 1H), 1.97-2.08 (m, 1H)
Discovery of potent and orally bioavailable inhibitors of Human Uric Acid Transporter 1 (hURAT1) and binding mode prediction using homology model
- Shanghai Hengrui Pharmaceutical Co. Ltd, 279 Wenjing Rd., Shanghai 200245, China
This Letter describes the Discovery of a series of potent inhibitors of Human Uric Acid Transporter 1 (hURATl). Lead generation via 3D pharmacophore Analysis and Optimization resulted in compound 41 . With an IC 50 of 33.7 nM, 41 Also Demonstrated good Oral Bioavailability in RAT (74.8%) and displayed a consistent PK profile across all species tested (rat, dog and monkey).
http://www.sciencedirect.com/science/article/pii/S0960894X1530353X
Volume 26, Issue 2 , 15 January 2016, Pages 277-282

//////// Shanghai Hengrui, inhibitors of Human Uric Acid Transporter 1 (hURAT1), 1- (6-bromoquinolin-4-yl) sulfanylcyclobutane-1-carboxylic acid
c13cc (ccc3nccc1SC2 (C (= O) O) CCC2) Br

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....