Home » Posts tagged 'preclinical'
Tag Archives: preclinical
JNJ-A07



JNJ-A07
S + FORM
CAS 2135640-93-4 ROT (+)S
Butanoic acid, 4-[3-[[1-(4-chlorophenyl)-2-[2,3-dihydro-6-(trifluoromethoxy)-1H-indol-1-yl]-2-oxoethyl]amino]-5-methoxyphenoxy]-, (+)-
(+)-4-[3-[[1-(4-Chlorophenyl)-2-[2,3-dihydro-6-(trifluoromethoxy)-1H-indol-1-yl]-2-oxoethyl]amino]-5-methoxyphenoxy]butanoic acid
(+)-4-[3-([(1S)-1-(4-Chlorophenyl)-2-oxo-2-[6-(trifluoromethoxy)-2,3-dihydro-1H-indol-1-yl]ethyl]amino)-5-methoxyphenoxy]butanoic acidMolecular FormulaC28 H26 Cl F3 N2 O6Molecular Weight578.964
REF
Kaptein, S.J.F., Goethals, O., Kiemel, D. et al. A pan-serotype dengue virus inhibitor targeting the NS3–NS4B interaction. Nature (2021). https://doi.org/10.1038/s41586-021-03990-6

JNJ-018
CAS 2135640-91-2 +/-, R,S
CAS 2135640-92-3 ROT (-)R
Butanoic acid, 4-[3-[[1-(4-chlorophenyl)-2-[2,3-dihydro-6-(trifluoromethoxy)-1H-indol-1-yl]-2-oxoethyl]amino]-5-methoxyphenoxy]-, (-)-
(-)-4-[3-[[1-(4-Chlorophenyl)-2-[2,3-dihydro-6-(trifluoromethoxy)-1H-indol-1-yl]-2-oxoethyl]amino]-5-methoxyphenoxy]butanoic acid
- Janssen (Originator)
- Katholieke Universiteit Leuven (Originator)
- NS4B Protease (Dengue Virus) Inhibitors
- Serine Protease NS3/Non-Structural Protein NS4B Protease (Dengue Virus) Interaction Inhibitors
A pan-serotype dengue virus inhibitor targeting the NS3–NS4B interaction
https://www.nature.com/articles/s41586-021-03990-6
https://www.nature.com/articles/s41586-021-03990-6#citeas
Abstract
Dengue virus causes approximately 96 million symptomatic infections annually, manifesting as dengue fever or occasionally as severe dengue1,2. There are no antiviral agents available to prevent or treat dengue. Here, we describe a highly potent dengue virus inhibitor (JNJ-A07) that exerts nanomolar to picomolar activity against a panel of 21 clinical isolates that represent the natural genetic diversity of known genotypes and serotypes. The molecule has a high barrier to resistance and prevents the formation of the viral replication complex by blocking the interaction between two viral proteins (NS3 and NS4B), thus revealing a previously undescribed mechanism of antiviral action. JNJ-A07 has a favourable pharmacokinetic profile that results in outstanding efficacy against dengue virus infection in mouse infection models. Delaying start of treatment until peak viraemia results in a rapid and significant reduction in viral load. An analogue is currently in further development.


2-(4-Chlorophenyl)-1-(6-(trifluoromethoxy)indolin-1-yl)-ethanone (1)
127 A mixture of 6-(trifluoromethoxy)indoline ([CAS 959235-95-1], 2 g, 9.84 mmol), 2-(4-chlorophenyl)acetic acid 128 ([CAS 1878-66-6], 1.85 g, 10.8 mmol), HATU (5.6 g, 14.8 mmol) and diisopropylethylamine (4.9 mL, 29.5
129 mmol) in DMF (40 mL) was stirred at room temperature for 12 h. Water was added and the precipitate was
130 filtered off. The residue was taken up with EtOAc. The organic solution was washed with a 10 % aqueous
131 solution of K2CO3, brine, dried over MgSO4, filtered, and the solvent was evaporated under reduced pressure. 132 The residue was purified by chromatography on silica gel (15-40 pm, 80 g, heptane/EtOAc gradient 90/10 to 133 60/40). The pure fractions were combined and the solvent was concentrated under reduced pressure to give 2-(4-
134 chlorophenyl)-1-(6-(trifluoromethoxy)indolin-1-yl)-ethanone 1 (3 g, yield: 86 %).
135 1 H NMR (400 MHz, DMSO-d6) d ppm 7.99 (s, 1 H), 7.37 – 7.41 (m, 2 H), 7.29 – 7.34 (m, 3 H), 6.97 (dd, J = 8.1, 1.3 Hz, 1 H), 4.25 (t, J = 8.6 Hz, 2 H), 3.88 (s, 2 H), 3.18 (t, J = 8.5 Hz, 2 H); 13
136 C NMR (101 MHz, 137 CHLOROFORM-d) δ ppm 168.91, 148.65, 148.63, 144.05, 133.16, 132.26, 130.63, 129.54, 128.93, 124.87, 120.50 (q, J=257.2 Hz), 116.38, 110.83, 77.26, 48.86, 42.52, 27.59; LC-MS: [M+H]+
138 728; purity 99 % (method LCMS2); Melting Point: 116-131 °C (DSC peak: 120.2 °C); HRMS (ESI+) m/z: [M]+ 139 calcd for C17H13ClF3NO2,
140 356.0660; found, 356.0657
141 2-Bromo-2-(4-chlorophenyl)-1- (6-(trifluoromethoxy)indolin-1-yl)ethanone (2)
142 At -78 °C, under nitrogen flow, LiHMDS (1.5 M in THF, 11.2 mL, 16.9 mmol) was added dropwise to a mixture 143 of 1 (3 g, 8.43 mmol) in THF (50 mL). The mixture was stirred for 15 min at -78 °C and a solution of N
144 bromosuccinimide (1.65 g, 9.3 mmol) in THF (30 mL) was added dropwise. After stirring for 2 h at -78 °C, the 145 reaction was quenched with a saturated aqueous solution of NH4Cl. The mixture was extracted with EtOAc. The 146 organic layer was separated, dried over MgSO4, filtered, and the solvent was evaporated under reduced pressure
147 to give 2-bromo-2-(4-chlorophenyl)-1- (6-(trifluoromethoxy)indolin-1-yl)ethanone 2 (3.6 g, yield: 98 %) as an 148 oil. The compound was used without further purification in the next step.
149 1 H NMR (400 MHz, CHLOROFORM-d) δ ppm 8.19 (s), 7.52 – 7.57 (m), 7.34 – 7.39 (m), 7.17 (d, J=8.2 Hz), 6.92 (dd, J=8.2, 1.1 Hz), 5.56 (s), 4.37 (td, J=10.1, 6.5 Hz), 4.09 (td, J=10.1, 6.7 Hz), 3.12 – 3.31 (m); 13
150 C NMR
151 (101 MHz, CHLOROFORM-d) δ ppm 164.90 (s), 148.68 (d, J=2.2 Hz), 143.75 (s), 135.46 (s), 133.99 (s), 152 130.52 (s), 129.79 (s), 129.10 (s), 125.01 (s), 117.20 (s), 120.47 (q, J=257.2 Hz), 111.36 (s), 48.88 (s), 46.61 (s), 27.65 (s); LC-MS: [M+H]+ 436; purity 100 % (method LCMS2); HRMS (ESI+) m/z: [M]+ 153 calcd for
154 C17H13O2NBrClF3, 433.9765; found, 433.9764
155 tert-Butyl 4-(3-amino-5-methoxyphenoxy)butanoate (3) 156 To a mechanically stirred solution of tert-butyl 4-bromobutanoate ([CAS 110661- 5 91-1], 42.3 g, 0.19 mol) in
157 DMF (600 mL) was added in portions a solid mixture of 3-amino-5-methoxyphenol ([CAS 162155-27-3], 26.4 158 g, 0.19 mol) and Cs2CO3 (123.6 g, 0.379 mol). The reaction mixture was stirred at 60 °C for 65 h, and allowed to
159 reach room temperature. The mixture was poured out into water (2.5 L). The product was extracted with Et2O (2 160 x). The combined organic layers were washed with brine, dried over MgSO4, and filtered. The solvent was
161 evaporated under reduced pressure, and then co-evaporated with toluene. The residue was purified by normal 162 phase HPLC (Stationary phase: silica gel 60A 25-40 pm (Merck), Mobile phase: gradient EtOAc/heptane 20/80 163 to 60/40), yielding tert-butyl 4-(3-amino-5-methoxyphenoxy)butanoate 3 as an oil (27 g, yield: 50 %).
164 1 H NMR (400 MHz, CHLOROFORM-d) δ ppm 5.89 – 5.92 (m), 5.86 (d, J=2.2 Hz), 3.92 (t, J=6.2 Hz), 3.73 (s), 3.66 (br s), 2.40 (t, J=7.4 Hz), 1.98 – 2.08 (m), 1.45 (s); 13
165 C NMR (101 MHz, CHLOROFORM-d) δ ppm 172.61 166 (s), 161.69 (s), 161.02 (s), 148.35 (s), 94.33 (s), 93.89 (s), 91.52 (s), 80.35 (s), 66.74 (s), 55.17 (s), 32.07 (s), 28.13 (s), 24.78 (s); LC-MS: [M+H]+ 282; purity 94 % (method LCMS2); HRMS (ESI+) m/z: [M]+
167 calcd for 168 C15H24O4N, 282.1700; found, 282.1695
169 tert-Butyl 4-(3-((1-(4-chlorophenyl)-2-oxo-2-(6-(trifluoromethoxy)indolin-1-yl)ethyl)amino)-5-
170 methoxyphenoxy)butanoate (4)
171 A mixture of 2 (3.6 g, 8.3 mmol), 3 (2.3 g, 8.3 mmol) and diisopropylethylamine (1.7 ml, 9.94 mmol) in CH3CN 172 (80 mL) was stirred at 70 °C for 4 h. The mixture was concentrated under reduced pressure, diluted with EtOAc,
173 and washed with 1 N aqueous HCl and water. The organic phase was separated, dried over MgSO4, filtered, and 174 the solvent was evaporated under reduced pressure. The compound was purified by flash chromatography on 175 silica gel (15-40 pm, 120 g, heptane/EtOAc 80/20). The pure fractions were combined and evaporated to dryness
176 to give, after crystallization from diisopropyl ether, tert-butyl 4-(3-((1-(4-chlorophenyl)-2-oxo-2-(6-
177 (trifluoromethoxy)indolin-1-yl)ethyl)amino)-5-methoxyphenoxy)butanoate 4 (2.6 g, yield: 49 %).
178 1 H NMR (400 MHz, DMSO-d6) d ppm 8.03 (s, 1 H), 7.55 (d, J = 8.6 Hz, 2 H), 7.43 (d, J = 8.6 Hz, 2 H), 7.33 (d, 179 J = 8.1 Hz, 1 H), 7.01 (dd, J = 8.1, 1.5 Hz, 1 H), 6.44 (d, J = 8.8 Hz, 1 H), 5.94 (d, J = 2.0 Hz, 2 H), 5.75 (t, J = 180 2.0 Hz, 1 H), 5.55 (d, J = 8.8 Hz, 1 H), 4.51 (td, J = 10.3, 6.5 Hz, 1 H), 4.04 (td, J = 10.3, 7.3 Hz, 1 H), 3.84 (t, J 6 181 = 6.3 Hz, 2 H), 3.62 (s, 3 H), 3.09 – 3.23 (m, 2 H), 2.31 (t, J = 7.3 Hz, 2 H), 1.86 (quin, J = 6.8 Hz, 2 H), 1.39 (s, 9 H); 13
182 C NMR (101 MHz, CHLOROFORM-d) δ ppm 172.57, 168.84, 161.66, 161.02, 148.65, 148.63, 147.68,
183 143.79, 135.66, 134.48, 129.58, 129.42, 129.38, 124.99, 116.92, 120.50 (q, J=257.2 Hz), 111.13, 93.02, 92.72, 91.06, 80.38, 77.25, 66.79, 59.74, 55.17, 48.31, 32.09, 28.15, 27.64, 24.77; LC-MS: [M+H]+
184 635; purity: 98 % (method LCMS3); Melting Point: 109-125 °C (DSC peak: 116.1 °C); HRMS (ESI+) m/z: [M]+ 185 calcd for 186 C32H34ClF3N2O6, 635.2130; found, 635.2127 187 (+)-4-(3-((1-(4-Chlorophenyl)-2-oxo2-(6-(trifluoromethoxy)indolin-1-yl)ethyl)amino)-5-
188 methoxyphenoxy)butanoic acid (JNJ-A07) 189 A solution of 4 (2.4 g, 3.8 mmol) in 4 M HCl in dioxane (24 mL) was stirred at 5 °C for 3 h and at room 190 temperature for 3 h. The precipitate was filtered off and dried to afford 4-(3-((1-(4-chlorophenyl)-2-oxo2-(6- 191 (trifluoromethoxy)indolin-1-yl)ethyl)amino)-5-methoxyphenoxy)butanoic acid as an HCl salt (racemic JNJ192 A07, 2 g, 0.8 eq. HCl, 0.07 eq. H2O). This salt was neutralized prior to chiral separation by dissolving it in 193 EtOAc and treating this solution with 1 N aqueous NaOH and evaporation of the organic layer under reduced 194 pressure.
The enantiomers were separated via preparative chiral SFC (Stationary phase: Chiralcel® OD-H 5 pm 195 250 x 30 mm, Mobile phase: 50 % CO2, 50 % iPrOH (+ 0.3 % iPrNH2)) and further purified via preparative 196 achiral SFC (Stationary phase: Cyano® 6 pm 150 x 21.2 mm, Mobile phase: 80 % CO2, 20 % MeOH (+ 0.3 % 197 iPrNH2)). The product fractions were combined and evaporated under reduced pressure. Each enantiomer was 198 taken up with EtOAc and washed with 1 N aqueous HCl. The organic layers were separated, dried over MgSO4, 199 filtered, and the solvent was evaporated under reduced pressure. The first eluted enantiomer was solidified from 200 diethyl ether/diisopropyl ether to give the epimer of JNJ-A07 (616 mg, yield: 28 %).
The second eluted
201 enantiomer was solidified from diethyl ether/diisopropyl ether to give JNJ-A07 (715 mg, yield: 32 %).
202 203 Epimer of JNJ-A07:
204 1 H NMR (500 MHz, DMSO-d6) δ ppm 12.12 (br s, 1 H), 8.04 (br s, 1 H), 7.55 (br d, J = 8.2 Hz, 2 H), 7.44 (br d, 205 J = 8.5 Hz, 2 H), 7.34 (br d, J = 7.9 Hz, 1 H), 7.01 (br d, J = 7.6 Hz, 1 H), 6.45 (br s, 1 H), 5.95 (br d, J = 10.1 206 Hz, 2 H), 5.76 (s, 1 H), 5.57 (br s, 1 H), 4.47 – 4.57 (m, 1 H), 3.99 – 4.11 (m, 1 H), 3.85 (br t, J = 6.3 Hz, 2 H), 3.62 (s, 3 H), 3.08 – 3.27 (m, 2 H), 2.34 (br t, J = 7.3 Hz, 2 H), 1.87 (quin, J = 6.7 Hz, 2 H); 13
207 C NMR (101 MHz, 208 DMSO-d6) δ ppm 174.56, 169.79, 161.46, 160.71, 149.08, 147.64, 144.48, 137.28, 132.91, 131.95, 130.56, 209 128.89, 126.34, 120.58 (d, J=256.0 Hz), 116.69, 109.52, 93.08, 92.80, 90.23, 66.65, 58.69, 55.20, 48.65, 30.57, 27.48, 24.72; LC-MS: [M+H]+ 579; purity: 100 % (method LCMS1);
Chiral SFC: [M+H]+ 210 579; chiral purity 100 % (method SFC1); [a]D20 211 : -48.5° (589 nm, c 0.27 w/v %, DMF, 20 °C); Melting Point: 62-80 °C (DSC peak: 70.6 °C); HRMS (ESI+) m/z: [M]+ 212 calcd for C28H27O6N2ClF3, 579.1504; found, 579.1501 213 214
JNJ-A07: 215 1 H NMR (500 MHz, DMSO-d6) δ ppm 12.12 (brs, 1 H), 8.04 (br s, 1 H), 7.55 (br d, J = 8.2 Hz, 2 H), 7.44 (br d, 216 J = 8.2 Hz, 2 H), 7.34 (br d, J = 7.9 Hz, 1 H), 7.01 (br d, J = 7.9 Hz, 1 H), 6.45 (br s, 1 H), 5.95 (br d, J = 10.1 217 Hz, 2 H), 5.76 (br s, 1 H), 5.57 (s, 1 H), 4.46 – 4.59 (m, 1 H), 3.99 – 4.10 (m, 1 H), 3.85 (br t, J = 6.1 Hz, 2 H), 3.62 (s, 3 H), 3.09 – 3.27 (m, 2 H), 2.34 (br t, J = 7.3 Hz, 2 H), 1.87 (br t, J = 6.8 Hz, 2 H); 13
218 C NMR (101 MHz,
219 DMSO-d6) δ ppm 174.53 (C28), 169.79 (C10), 161.47 (C20), 160.72 (C22), 149.08 (C18), 147.65 (C6), 144.48 220 (C8), 137.29 (C12), 132.92 (C15), 131.95 (C3), 130.56 (C13, C17), 128.89 (C14, C16), 126.34 (C4), 120.58 (q, 221 J = 255.1 Hz, C9), 116.67 (C5), 109.51 (C7), 93.11 (C23), 92.81 (C21), 90.26 (C19), 66.66 (C25), 58.70 (C11), 55.21 (C24), 48.67 (C1), 30.57 (C27), 27.49 (C2), 24.72 (C26); LC/MS: [M+H]+
222 579; purity 100 % (method LCMS1); Chiral SFC: [M+H]+ 579; chiral purity 100 % (method SFC1); [a]D20
223 : +42.9° (589 nm, c 0.28 w/v %, 224 DMF, 20 °C); Melting point: 62-78 °C (DSC peak: 71.3 °C) ; HRMS (ESI+) m/z calcd for C28H27O6N2ClF3 [M]+ 225 , 579.1504, found 579.1500; Elemental analysis requires C, 58.09 %; H, 4.53 %; N, 4.84 % found C, 226 58.60 %; H, 4.59 %; N, 4.80 %
CLIP
https://www.bioworld.com/articles/512333-potent-selective-pan-serotype-dengue-inhibitor-developed
Blocking the interaction between two dengue virus (DENV) nonstructural proteins, NS3 and NS4B, with a newly developed small-molecule inhibitor resulted in potent antiviral activity in mouse models, according to an international collaborative study led by scientists at the University of Leuven (KU Leuven), CD3 the Centre for Drug Design and Discovery in Leuven, and Janssen Pharmaceutica in Beerse, Belgium.
This protein interaction represents a promising new target for the development of pan-serotype DENV inhibitors with a high barrier to resistance, with the potency of the inhibition warranting further development of these compounds, the authors reported in the October 6, 2021, edition of Nature.
“This is the first study to show that blocking the NS3/NS4B interaction has potent antiviral activity in mice warranting the further development of such inhibitors,” said study co-leader Johan Neyts, professor of virology at KU Leuven.
Dengue is currently among the leading threats to global public health, with an estimated 96 million individuals developing dengue disease, which is probably an underestimation.
In addition, the incidence of dengue has increased approximately 30-fold over the past 50 years. DENV is now endemic in the subtropical regions of 128 countries, with an estimated 4 billion people at risk of infection, predicted to increase to 6 billion by 2080.
This dengue upsurge is driven by various factors, most notably rapid urbanization and the spread of the Aedes mosquito vectors due to climate change.
The DENV has four serotypes that are further classified into genotypes, which are increasingly co-circulating in endemic regions. Antibodies to infection with one serotype can lead to a more severe second infection with a different serotype increases the risk of potentially life-threatening severe dengue.
The DENV vaccine Dengvaxia (Sanofi-Pasteur), which has been approved in several countries for individuals aged at least 9 years, is only recommended for those with previous DENV exposure.
Moreover, there are currently no available antiviral agents for dengue prevention or treatment, while development of pan-serotype DENV inhibitors has proven challenging.
“The major developmental challenge has been to obtain ultrapotent antivirals that also have equipotent activity against the four DENV serotypes,” Neyts told BioWorld Science.
Such drugs should lower viral loads during an ongoing infection, thereby reducing dengue-associated morbidity and mortality, as well as transmission.
In their new Nature study, researchers co-led by Neyts, Patrick Chaltin, managing director of CD3 the Centre for Drug Design and Discovery, and Marnix Van Loock, R&D Lead Emerging Pathogens, Janssen Global Public Health at Janssen Pharmaceutica, identified potential new DENV inhibitors using large-scale cell-based anti-DENV-2 screening.
“We screened tens of thousands of molecules and interesting hits were further optimized to eventually obtain JNJ-A07 and other ultrapotent and selective analogues, with roughly 2,000 analogues being synthesized and tested,” said Neyts.
Notably, the promising small molecule JNJ-A07 was demonstrated to have nanomolar to picomolar activity against a panel of 21 clinical isolates representing the natural genetic diversity of known DENV genotypes and serotypes.
The molecule was then shown to have a high barrier to resistance “by months of culturing the dengue virus in suboptimal concentrations of the inhibitor,” Neyts said.
JNJ-A07 was then shown to prevent formation of the viral replication complex by blocking the interaction between the nonstructural proteins NS3 and NS4B, thereby revealing a previously undescribed mechanism of antiviral action.
JNJ-A07 was further demonstrated to have a favorable pharmacokinetic (PK) profile resulting in outstanding efficacy against DENV infection in mouse models.
“JNJ-A07’s favorable PK profile resulted from optimization of the ADME [absorption, distribution metabolism and excretion] properties of the analogues within this chemical series,” Janssen’s Van Loock told BioWorld Science.
“This enabled us to administer the compound [twice daily] in mice and assess its efficacy, which resulted in a significantly reduced viral load and protected against mortality in a mouse lethal challenge model.”
However, “additional research will be required in preclinical models, to understand how these findings reflect those in humans, as currently no translational models are available to assess the potential effect in humans,” noted Van Loock.
Delaying treatment commencement until peak viremia had developed was shown to result in a rapid and significant reduction in viral load in the mouse models of infection.
This is an important finding, as “one wants an antiviral effect that is independent of how much [viral] replication is ongoing,” Van Loock said.
“In these mice, the reduction in viral load was also very pronounced if the treatment was initiated on the day of peak viral load, when the effect was quantified 24 hours later.”
On safety, said Neyts, as JNJ-A07 and its analogues “target specific viral proteins that have no homologues in eukaryotic cells, we expect a considerable safety window, with these agents being very well tolerated.” The safety and potency of DENV inhibition established in this study justifies the further development of these novel antivirals, with an analogue being currently in further development.
Further development will include “using our know-how to also develop drugs against the other member of the flavivirus family to which DENV belongs, including Japanese encephalitis, Zika, yellow fever, West Nile virus, et cetera,” said Neyts.
Meanwhile, “Janssen has moved the compound into clinical development and continues to work closely in this regard with teams at KU Leuven and elsewhere,” said Van Loock.
“We will be sharing information about progress of the compound’s clinical development during the American Society of Tropical Medicine and Hygiene meeting this November.”

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////////////////////////////PatentWO2017167951https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2017167951
- IN201827040889
- US2020299235
- US2019112266
- US10689340
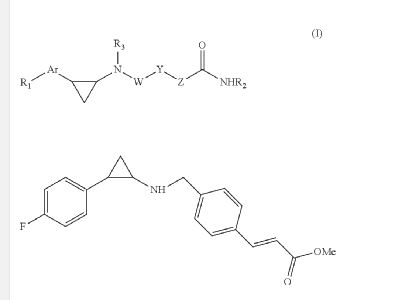
Due to the presence of said chiral carbon atom, a “compound of formula (I)” can be the (R)-enantiomer, the (S)-enantiomer, the racemic form, or any possible combination of the two individual enantiomers in any ratio. When the absolute (R)-or (S)-configuration of an enantiomer is not known, this enantiomer can also be identified by indicating whether the enantiomer is dextrorotatory (+)- or levorotatory (-)- after measuring the specific optical rotation of said particular enantiomer.
In an aspect the present invention relates to a first group of compound of formula (I) wherein the compounds of formula (I) have the (+) specific rotation.
In a further aspect the present invention relates to a second ground of compounds of formula (I) wherein the compounds of formula (I) have the (-) specific rotation.
Example 4: synthesis of 4-(3-((1 -(4-chlorophenyl)-2-oxo-2-(6-(trifluoromethoxy)-indolin-1 -yl)ethyl)amino)-5-methoxyphenoxy)butanoic acid (Compound 4) and chiral separation into Enantiomers 4A and 4B.
Synthesis of intermediate 4a:
A mixture of 6-(trifluoromethoxy)indoline [CAS 959235-95-1] (2 g, 9.84 mmol), 2-(4-chlorophenyl)acetic acid [CAS 1878-66-6] (1 .85 g, 10.8 mmol), HATU (5.6 g, 14.8 mmol) and diisopropylethylamine (4.9 ml_, 29.5 mmol) in DMF (40 ml_) was stirred at room temperature for 12 h. Water was added and the precipitate was filtered off. The residue was taken up with EtOAc. The organic solution was washed with a 10% aqueous solution of K2CO3, brine, dried over MgSO4, filtered and the solvent was evaporated under reduced pressure. The residue was purified by chromatography on silica gel (15-40 μιτι, 80 g, heptane/EtOAc gradient 90/10 to 60/40). The pure fractions were combined and the solvent was concentrated under reduced pressure to give 2-(4-chlorophenyl)-1 -(6-(trifluoromethoxy)indolin-1 -yl)-ethanone 4a (3 g).
Synthesis of intermediate 4b:
At -78°C, under N2 flow, LiHMDS 1 .5 M in THF (1 1 .2 ml_, 16.9 mmol) was added dropwise to a mixture of 2-(4-chlorophenyl)-1 -(6-(trifluoromethoxy)indolin-1 -yl)-ethanone 4a (3 g, 8.43 mmol) in THF (50 ml_). The mixture was stirred for 15 min at -78°C and a solution of /V-bromosuccinimide (1 .65 g, 9.3 mmol) in THF (30 ml_) was added dropwise. After stirring for 2 h at -78°C, the reaction was quenched with a saturated solution of NH CI. The mixture was extracted with EtOAc. The organic layer was separated, dried over MgSO4, filtered and the solvent was evaporated under reduced pressure to give 2-bromo-2-(4-chlorophenyl)-1 -(6-(trifluoromethoxy)indolin-1 -yl)ethanone 4b (3.6 g). The compound was used as such in the next step.
Synthesis of intermediate 4c:
A mixture of 2-bromo-2-(4-chlorophenyl)-1 -(6-(trifluoromethoxy)indolin-1 -yl)-ethanone 4b (3.6 g, 8.3 mmol), terf-butyl 4-(3-amino-5-methoxyphenoxy)-butanoate 1a (2.3 g, 8.3 mmol) and diisopropylethylamine (1 .7 mL, 9.94 mmol) in CH3CN (80 mL) was stirred at 70°C for 4 h. The mixture was concentrated under reduced pressure, diluted with EtOAc, and washed with 1 N HCI and water. The organic phase was separated, dried over MgSO4, filtered and the solvent was evaporated under reduced pressure. The compound was purified by flash chromatography on silica gel (15-40 μιτι, 120 g, heptane/EtOAc 80/20). The pure fractions were combined and evaporated to dryness to give, after crystallization from diisopropyl ether, te/t-butyl 4-(3-((1 -(4-chlorophenyl)-2-oxo-2-(6-(trifluoro-methoxy)indolin-1 -yl)ethyl)amino)-5-methoxyphenoxy)butanoate 4c (2.6 g).
Synthesis of Compound 4 and chiral separation into Enantiomers 4A and 4B: A solution of terf-butyl 4-(3-((1 -(4-chlorophenyl)-2-oxo-2-(6-(trifluoromethoxy)-indolin-1 -yl)ethyl)amino)-5-methoxyphenoxy)butanoate 4c (2.4 g, 3.8 mmol) in 4M HCI in dioxane (24 mL) was stirred at 5°C for 3 h and at room temperature for 3h. The precipitate was filtered off and dried to afford 4-(3-((1 -(4-chlorophenyl)-2-oxo-2-(6-(trifluoromethoxy)indolin-1 -yl)ethyl)amino)-5-methoxyphenoxy)butanoic acid as an HCI salt (Compound 4, 2 g, 0.8 equiv. HCI, 0.07 equiv. H2O). Compound 4 (2 g, HCI salt) was neutralized prior to chiral separation by treatment of a solution of Compound 4 (HCI salt) in ethylacetate with 1 N NaOH and evaporation of the organic layer under reduced pressure. The enantiomers were separated via Preparative Chiral SFC (Stationary phase: Chiralcel® OD-H 5 μηη 250 x 30 mm, Mobile phase: 50% CO2, 50% iPrOH (+ 0.3% iPrNH2)) and further purified via Preparative achiral SFC (Stationary phase: Cyano® 6 μιτι 150×21 .2mm, Mobile phase: 80% CO2, 20% MeOH (+ 0.3% iPrNH2)). The product fractions were combined and evaporated under reduced pressure. The two enantiomers were taken up with EtOAc and washed with 1 N HCI. The organic layers were separated, dried over MgSO4, filtered and the solvent was evaporated under reduced pressure. The first eluted enantiomer was solidified from ether/diisopropyl ether to give Enantiomer 4A (616 mg). The second eluted enantiomer was solidified from ether/diisopropyl ether to give Enantiomer 4B (715 mg).
It is also possible to separate the enantiomers starting from the HCI salt of the racemate using the same conditions for chiral separation.
Compound 4:
1H NMR (500 MHz, DMSO-c/6) δ ppm 1 .87 (quin, J=6.9 Hz, 2 H) 2.34 (t, J=7.3 Hz, 2 H) 3.07 – 3.28 (m, 2 H) 3.62 (s, 3 H) 3.85 (t, J=6.5 Hz, 2 H) 4.04 (td, J=10.5, 7.1 Hz, 1 H) 4.52 (td, J=10.3, 6.5 Hz, 1 H) 5.57 (s, 1 H) 5.76 (t, J=2.2 Hz, 1 H) 5.90 – 6.00 (m, 2 H) 7.01 (dd, J=8.2, 1 .6 Hz, 1 H) 7.33 (d, J=8.2 Hz, 1 H) 7.41 – 7.48 (m, 2 H) 7.55 (d, J=8.5 Hz, 2 H) 8.03 (s, 1 H)
LC/MS (method LC-B): Rt 2.70 min, MH+ 579
Melting point: 150°C
Enantiomer 4A:
1H NMR (500 MHz, DMSO-c/6) δ ppm 1 .87 (quin, J=6.7 Hz, 2 H) 2.34 (br t, J=7.3 Hz, 2 H) 3.08 – 3.27 (m, 2 H) 3.62 (s, 3 H) 3.85 (br t, J=6.3 Hz, 2 H) 3.99 -4.1 1 (m, 1 H) 4.47 – 4.57 (m, 1 H) 5.57 (br s, 1 H) 5.76 (s, 1 H) 5.95 (br d, J=10.1 Hz, 2 H) 6.45 (br s, 1 H) 7.01 (br d, J=7.6 Hz, 1 H) 7.34 (br d, J=7.9 Hz, 1 H) 7.44 (br d, J=8.5 Hz, 2 H) 7.55 (br d, J=8.2 Hz, 2 H) 8.04 (br s, 1 H) 12.12 (br s, 1 H) LC/MS (method LC-A): Rt 2.95 min, MH+ 579
[a]D20: -48.5° (c 0.27, DMF)
Chiral SFC (method SFC-A): Rt 1 .13 min, MH+ 579, chiral purity 100%.
Enantiomer 4B:
1H NMR (500 MHz, DMSO-c/6) δ ppm 1 .87 (br t, J=6.8 Hz, 2 H) 2.34 (br t, J=7.3 Hz, 2 H) 3.09 – 3.27 (m, 2 H) 3.62 (s, 3 H) 3.85 (br t, J=6.1 Hz, 2 H) 3.99 -4.10 (m, 1 H) 4.46 – 4.59 (m, 1 H) 5.57 (s, 1 H) 5.76 (br s, 1 H) 5.95 (br d, J=10.1 Hz, 2 H) 6.45 (br s, 1 H) 7.01 (br d, J=7.9 Hz, 1 H) 7.34 (br d, J=7.9 Hz, 1 H) 7.44 (br d, J=8.2 Hz, 2 H) 7.55 (br d, J=8.2 Hz, 2 H) 8.04 (br s, 1 H) 12.12 (br s, 1 H) LC/MS (method LC-A): Rt 2.94 min, MH+ 579
[a]D20: +42.9° (c 0.28, DMF)
Chiral SFC (method SFC-A): Rt 2.13 min, MH+ 579, chiral purity 100%.
Patent
WO2021094563
The compounds of formula I according to the present invention may be synthesized according to methods described in the art, as disclosed in WO 2016/180696. The compounds of formula II according to the present invention may be prepared according to methods described in the art, as disclosed in WO2017/167951.
Compound (b) of the present invention was tested in AG129 mouse viremia model. The synthesis of compound (b) is described in WO 2017/167951, under Example 4.
compound (b)
PATENT
WO 2018215316
The compounds of formula (I) of the present invention all have at least one asymmetric carbon atom as indicated in the figure below by the carbon atom labelled with * :
Ref
https://doi.org/10.1038/s41586-021-03990-6
////////////////////JNJ-A07, DENGUE, VIRUS, PRECLINICAL

NEW DRUG APPROVALS
ONE TIME
$10.00
JBI-802 BY JUBILANT
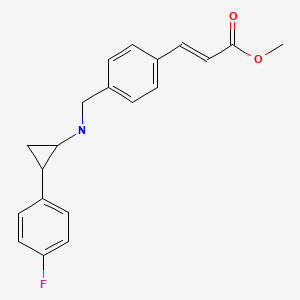
EXAMPLE
O=C(OC)/C=C/c1ccc(CNC2CC2c2ccc(F)cc2)cc1
EXAMPLE ONLY NOT CONFIRMED
JBI-802
- Myeloid Leukemia Therapy
- Solid Tumors Therapy
Epigenetic Modifier Modulators
- Histone Deacetylase 6 (HDAC6) Inhibitors
- Lysine-Specific Histone Demethylase 1A (KDM1A; LSD1) Inhibitors
Jubilant Therapeutics Announces Successful Completion of Pre-IND Meeting with FDA for its Novel Dual LSD1 and HDAC6 Inhibitor JB1-802
https://markets.businessinsider.com/news/stocks/jubilant-therapeutics-announces-successful-completion-of-pre-ind-meeting-with-fda-for-its-novel-dual-lsd1-and-hdac6-inhibitor-jb1-802-1030834551
PRESS RELEASE PR Newswire
Sep. 30, 2021, 10:23 AM
BEDMINSTER, NJ, Sept. 30, 2021 /PRNewswire/ — Jubilant Therapeutics Inc., a biopharmaceutical company advancing small molecule precision therapeutics to address unmet medical needs in oncology and autoimmune diseases, today announced the successful completion of a pre-IND (Investigational New Drug) meeting with the U.S. Food and Drug Administration (FDA) regarding the development plan, clinical study design and dosing strategy for the Phase I/II trial of JB1-802, a dual inhibitor of LSD1 and HDAC6, for the treatment of small cell lung cancer, treatment-induced neuro-endocrine prostate cancer and other mutation-defined neuroendocrine tumors.
 A pre-IND meeting provides the drug development sponsor an opportunity for an open communication with the FDA to discuss the IND development plan and to obtain the agency’s guidance regarding planned clinical evaluation of the sponsor’s new drug candidate. After reviewing the preclinical data provided, plans for additional data generation and the Phase I/II clinical trial protocol, the FDA addressed Jubilant Therapeutics’ questions, provided guidance and aligned with the sponsor on the proposed development plan for JBI-802.
A pre-IND meeting provides the drug development sponsor an opportunity for an open communication with the FDA to discuss the IND development plan and to obtain the agency’s guidance regarding planned clinical evaluation of the sponsor’s new drug candidate. After reviewing the preclinical data provided, plans for additional data generation and the Phase I/II clinical trial protocol, the FDA addressed Jubilant Therapeutics’ questions, provided guidance and aligned with the sponsor on the proposed development plan for JBI-802.
“We appreciate the FDA’s guidance as we endeavor to find an innovative new treatment for high unmet-need tumors with devastatingly low survival rates,” said Hari S Bhartia, Chairman, Jubilant Therapeutics Inc.
“We are pleased with the outcome of the pre-IND meeting with the FDA and plan to submit the IND application by the end of 2021,” said Syed Kazmi, Chief Executive Officer, Jubilant Therapeutics Inc.
About Jubilant TherapeuticsJubilant Therapeutics Inc. is a patient-centric biopharmaceutical company advancing potent and selective small molecule modulators to address unmet medical needs in oncology and autoimmune diseases. Its advanced discovery engine integrates structure-based design and computational algorithms to discover and develop novel, precision therapeutics against both first-in-class and validated but intractable targets in genetically defined patient populations. The Company plans to file an IND later this year for the first in class dual inhibitor of LSD1/HDAC6, followed by two additional INDs in 2022 with novel modulators of PRMT5 and PAD4 in oncology and inflammatory indications. Jubilant Therapeutics is headquartered in Bedminster NJ and guided by globally renowned key opinion leaders and scientific advisory board members. For more information, please visit www.jubilanttx.com or follow us on Twitter @JubilantTx and LinkedIn.
View original content:https://www.prnewswire.com/news-releases/jubilant-therapeutics-announces-successful-completion-of-pre-ind-meeting-with-fda-for-its-novel-dual-lsd1-and-hdac6-inhibitor-jb1-802-301388983.html
SOURCE Jubilant Therapeutics Inc.

Mohd Zainuddin
Director at Jubilant Therapeutics Inc
PATENT
IN 201641016129
PATENT
US20200308110 – CYCLOPROPYL-AMIDE COMPOUNDS AS DUAL LSD1/HDAC INHIBITORS
https://patentscope.wipo.int/search/en/detail.jsf?docId=US306969204&tab=NATIONALBIBLIO&_cid=P21-KUANET-85789-2ApplicantsJubilant Epicore LLC
Inventors
Sridharan RAJAGOPAL
Mahanandeesha S. HALLUR
Purushottam DEWANG
Kannan MURUGAN
Durga Prasanna KUMAR C.H.
Pravin IYER
Chandrika MULAKALA
Dhanalakshmi SIVANANDHAN
Sreekala NAIR
Mohd ZAINUDDIN
Subramanyam Janardhan TANTRY
Chandru GAJENDRAN
Sriram RAJAGOPAL
Priority Data201641016129 09.05.2016 IN

Sridharan Rajagopal
Vice President-Head of Medicinal Chemistry at Jubilant Therapeutics Inc

Dhanalakshmi Sivanandhan
Vice President at Jubilant Therapeutics Inc

Mahanandeesha Hallur
Associate Director at Jubilant Biosys

Sreekala Nair

Chandrika Mulakala

Pravin Iyer

Purushottam (M.) Dewang
ERRORS CALL ME , +919321316780
AND TO ADD TOO
EXAMPLE
C20 H20 F N O22-Propenoic acid, 3-[4-[[[2-(4-fluorophenyl)cyclopropyl]amino]methyl]phenyl]-, methyl ester, (2E)-Molecular Weight, 325.38
Patent
WO2017195216
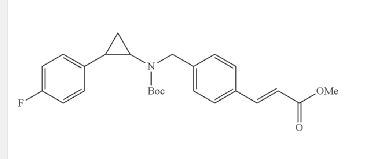
I-3methyl (E)-3-(4-(((tert-butoxycarbonyl)(2-(4-((4-fluorobenzyl)oxy)phenyl) cyclopropyl)amino)methyl)phenyl)acrylate

The compound was synthesized using amine B6 and (E)-3-(4-Formyl-phenyl)-acrylic acid methyl esterfoUowing the procedure for the synthesis of 1-2. LC-MS m/z calcd for C32H34FN05, 531.2; found 532.2 [M+H]+.

| Publication Number | Title | Priority Date | Grant Date |
|---|---|---|---|
| EP-3455204-A1 | Cyclopropyl-amide compounds as dual lsd1/hdac inhibitors | 2016-05-09 | |
| WO-2017195216-A1 | Cyclopropyl-amide compounds as dual lsd1/hdac inhibitors | 2016-05-09 | |
| US-2020308110-A1 | Cyclopropyl-amide compounds as dual lsd1/hdac inhibitors | 2016-05-09 |

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com
Step 2: (E)-3-[4-({tert-Butoxycarbonyl-[2-(4-fluoro-phenyl)-cyclopropyl]-amino}-methyl)-phenyl]-acrylic acid methyl ester (I-2)
| To a stirred solution of (E)-3-(4-{[2-(4-fluoro-phenyl)-cyclopropylamino]-methyl}-phenyl)-acrylic acid methyl ester (XLVI, 0.25 g, 0.76 mmol) in tetrahydrofuran and water mixture (6 mL, 1:1) was added sodium bicarbonate (0.087 g, 2.3 mmol) and Boc anhydride (0.22 mL, 0.92 mmol) at room temperature and the resulting mixture was stirred at that temperature for 2 h. The progress of the reaction was monitored by TLC. The reaction mixture was diluted with ethylacetate and the organic portion was washed with water and brine solution, dried over sodium sulphate and concentrated under reduced pressure to get the crude product which was purified by column chromatography using ethylacetate-hexane gradient to afford the titled product as sticky oil (I-2, 0.19 g, 58%). LC-MS m/z calcd for C 25H 28FNO 4, 425.2; found 326.3 [M-Boc+1] +. |
| The following compounds were synthesized using procedure for the synthesize of I-2 |
REFJBI-802, novel dual inhibitor of LSD1-HDAC6 for treatment of cancerSivanandhan, D.; Rajagopal, S.; Nair, S.; et al.Annu Meet Am Assoc Cancer Res (AACR) · 2020-06-22 / 2020-06-24 · Virtual, N/A · Abst 1756Synthesis and optimization of a novel series of LSD1-HDAC dual inhibitors led to the discovery of JBI-802 as the lead compound, with IC50 of 0.05 mcM against LSD1 and isoform selective HDAC6/8 activity, with IC50 of 0.011 and 0.098 mcM for HDAC6 and HDAC8, respectively. The candidate also showed excellent selectivity against other HDACs, with approximately 77-fold selectivity for HDAC6. In vitro, JBI-802 showed strong antiproliferative activity on selected cell lines, including acute myeloid leukemia, chronic lymphocytic leukemia, lymphoma and certain solid tumors, such as small cell lung cancer and sarcoma. In vivo, JBI-802 demonstrated strong efficacy in erythroleukemia xenograft model, leading to prolonged survival of mice bearing HEL92.1.7 tumors. The candidate showed excellent dose-response and superior efficacy compared to single agents in this model, with ED50 of approximately 6.25 mg/kg twice-daily by oral administration. When evaluated in CT-26 syngeneic model, JBI-802 showed promising activity as single agent and in the combination of JBI-802 plus anti-programmed cell death protein 1 (PD-1) monoclonal antibody (MAb), with approximately 80% tumor growth inhibition observed for the combination. Exploratory toxicology studies showed that JBI-802 was well tolerated at efficacious doses. Further preclinical IND-enabling studies are currently underway for this molecule, which is to be developed as a clinical candidate for the treatment of acute myeloid leukemia and other tumor types.
REFNovel dual inhibitor of LSD1-HDAC6/8 for treatment of cancerDhanalakshmi, S.; Rajagopal, S.; Sadhu, N.; et al.62nd Annu Meet Am Soc Hematol · 2020-12-05 / 2020-12-08 · Virtual, N/A · Abst 3378 Blood 2020, 136(Suppl. 1)
REFJubilant Therapeutics Presents Preclinical Data at the American Association for Cancer Research, Reveals Unique Dual-Action Anti-Cancer Mechanism Underscoring First-in-Class Pipeline Asset in Hematological Tumors
Jubilant Therapeutics Press Release 2020, June 22
////////////////JB1-802, JUBILANT, CANCER, PRECLINICAL
EXTRAS…………
PATENTWO2021062327 – FUSED PYRIMIDINE COMPOUNDS, COMPOSITIONS AND MEDICINAL APPLICATIONS THEREOFhttps://patentscope.wipo.int/search/en/detail.jsf?docId=WO2021062327&_cid=P21-KUAMRR-83330-1PCT/US2020/052953
Priority Data
| 201941039277 | 27.09.2019 | IN |
Inventors
- VENKATESHAPPA, Chandregowda
- SIVANANDHAN, Dhanalakshmi
- RAJAGOPAL, Sridharan
- ROTH, Bruce
- PANDEY, Anjali
- SAXTON, Tracy
- HALLUR, Gurulingappa
- MADHYASTHA, Naveena
- SADHU M, Naveen
Lung cancer accounts for the greatest number of cancer deaths, and approximately 85% of lung cancer cases are non-small cell lung cancer (NSCLC). The development of targeted therapies for lung cancer has primarily focused on tumors displaying specific oncogenic drivers, namely mutations in epidermal growth factor receptor (EGFR) and anaplastic lymphoma kinase (ALK). Three generations of tyrosine kinase inhibitors (TKIs) have been developed for cancers with the most frequently observed EGFR mutations, however, other oncogenic drivers in the EGFR family of receptor tyrosine kinases have received less research and development focus and several oncogenic drivers, including insertions in the exon 20 gene of EGFR, have no currently approved therapeutics to treat their cancers.
[0003] The mutation, amplification and/or overexpression of human epidermal growth factor receptor 2 (HER2), another member of the human epidermal growth factor receptor family of receptor tyrosine kinases, has been implicated in the oncogenesis of several cancers, including lung, breast, ovarian, and gastric cancers. Although targeted therapies such as trastuzumab and lapatinib have shown clinical efficacy especially in breast tumors, their utility in lung cancer has been limited. It is likely that this variation is due to tissue-specific factors, including the low potency of kinase inhibitors like lapatinib for the mutagenic alterations in HER2 that are observed in the lung cancer patient population, including insertions in the exon 20 gene of HER2.
[0004] Given that many patients with mutations in EGFR and HER2 do not derive clinical benefit from currently available therapies against these targets, there remains a significant unmet need for the development of novel therapies for the treatment of cancers associated with EGFR and HER2 mutations.
Compound 49: (E)-N-(3-(3-benzyl-7-((1-methyl-1H-pyrazol-4-yl)amino)-2-oxo-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)-4-(dimethylamino)but-2-enamide
Step 1: Synthesis of (E)-4-(dimethylamino)but-2-enoyl chloride
[0280] To a stirred mixture of acetonitrile (2 mL) and DMF (2 drop) under N2 atmosphere was added N,N-dimethylamino crotonic acid hydrochloride (0.1 g, 0.77 mmol). After 10 min, this solution was cooled to 0-5 °C. Oxalyl chloride (0.122 g, 0.968 mmol) was added and the reaction mixture was maintained at 0-5 °C for 30 min. It was allowed to warm to RT and stirring was continued for 2 h. It was then heated to 40 °C for 5 min and again brought to RT and stirred for 10 min. Formation of product was confirmed by TLC and the reaction mass was used as such to the next step without any workup.
Step-2: Synthesis of (E)-N-(3-(3-benzyl-7-((1-methyl-1H-pyrazol-4-yl)amino)-2-oxo-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)-4-(dimethylamino)but-2-enamide (Compound 49)
[0281] 1-(3-Aminophenyl)-3-benzyl-7-((1-methyl-1H-pyrazol-4-yl)amino)-3,4-dihydropyrimido[4,5-d]pyrimidin-2(1H)-one (0.11g, 0.7 mmol) in DMP (2 mL) was cooled to -15 °C and then (E)-4-(dimethylamino)but-2-enoylchloride was added. The reaction mixture was stirred for 1 h at -15 °C to RT. After the completion of reaction, the reaction mass was quenched with ice water, sodium bicarbonate solution and extracted with DCM (100 mL x 2). The combined organic layer was washed with cold water (3 x 50 mL), brine solution (10 mL), dried over anhydrous sodium sulfate and evaporated under reduced pressure to obtain crude product. The crude product was purified by prep HPLC to get pure product (E)-N-(3-(3-benzyl-7-((1-methyl-1H-pyrazol-4-yl)amino)-2-oxo-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)-4-(dimethylamino)but-2-enamide (Compound 49, 0.022 g, 16 % yield) as white solid.1H NMR (400 MHz, DMSO-d6): δ 10.21 (s, 1H), 9.32 (s, 1H), 8.06 (s, 1H), 7.76 (bs, 1H) 7.65 (s, 1H), 7.48 (bs, 1H), 7.39-7.29 (m, 5H), 7.03 (d, J = 7.2 Hz, 2H), 6.74-6.68 (m, 1H), 6.62 (s, 1H), 6.25 (d, J = 15.2 Hz, 1H), 4.62 (s, 2H), 4.37 (s, 2H), 3.47 (s, 3H), 3.03 (d, J = 5.6 Hz, 2H), 2.15 (s, 6H); LCMS Calcd for [M+H] + 538.2, found 538.5
Compound 50: (E)-N-(3-(3-benzyl-7-((1-methyl-1H-pyrazol-4-yl)amino)-2-oxo-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)-3-chloroacrylamide
Step-1: Synthesis of (Z)-3-chloroacrylic acid
[0282] To a stirred solution propiolic acid (2 g, 28.5 mmol) in DMF (15 mL) under N2 atmosphere was added thionyl chloride (4.07 g, 34.2 moles) slowly and the reaction mixture was maintained at 25 °C for 1 h. The reaction was monitored by TLC, after the completion of reaction, the residue was poured into ice and the resulting aqueous solution was extracted with ether (3 x100 mL). The organic layer was washed with brine (20 mL), dried over anhydrous sodium sulfate and evaporated under reduced pressure to obtain crude product. The crude product was purified to get pure product (Z)-3-chloroacrylic acid (1.9 g, 62.9 % yield). LCMS Calcd for [M-H] +, 104.98, found 105.1
Step-2: Synthesis of (Z)-3-chloroacryloyl chloride
[0283] To a stirred solution of acetonitrile (3 mL) and DMF (3 drop) under N2 atmosphere was added of (Z)-3-chloroacrylic acid (0.2 g, 1.87 mmol). After 10 min this solution was cooled 0-5 °C. Oxalyl chloride (0.122 g, 0.968 mmol) was added and the reaction mixture was maintained at 0-5 °C for 30 min. It was allowed to warm to RT and stirring was continued for 2 h to get (Z)-3-chloroacryloyl chloride. Formation of product was confirmed by TLC and the reaction mass was used as such to the next step without any workup.
Step-3: Synthesis of (E)-3-((3-(3-benzyl-7-((1-methyl-1H-pyrazol-4-yl)amino)-2-oxo-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)amino)acryloyl chloride (Compound 50)
[0284] A solution of 1-(3-Aminophenyl)-3-benzyl-7-((1-methyl-1H-pyrazol-4-yl)amino)-3,4-dihydropyrimido[4,5-d]pyrimidin-2(1H)-one (0.11 g, 0.7 mmol) in DMP (2 mL) was cooled to -15 °C and then (Z)-3-chloroacryloyl chloride was added. The reaction mixture was stirred for 1 h at -15 °C to RT. The reaction was monitored by TLC. After the completion of reaction, reaction mass was quenched with ice water and sodium bicarbonate solution. The aqueous layer was e 0.028 g, 22% yield) as a white solid.1H NMR (400 MHz, DMSO-d6): δ 10.35 (s, 1H), 9.32 (s, 1H), 8.06 (s, 1H), 7.74 (s, 1H), 7.59 (s, 1H), 7.51 (s, 1H), 7.41-7.35 (m, 5H), 7.30-7.29 (m, 1H), 7.08-7.02 (m, 2H), 6.62-6.58 (m, 2H), 4.62 (s, 2H), 4.37 (s, 2H), 3.47 (s, 3H); LCMS Calcd for [M+H] + 515.1, LCMS found 515.2
Compound 51: (E)-N-(3-(7-((3-chloro-1-methyl-1H-pyrazol-4-yl)amino)-3-phenyl-2-thioxo-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)-4-(dimethylamino)but-2-enamide
Step-1: Synthesis of 2,4-dichloro-5-(chloromethyl)pyrimidine
[0285] Title compound was prepared in a similar manner to general procedure I.5-(hydroxymethyl)pyrimidine-2,4-diol (15 g, 106 mmol) gave 2,4-dichloro-5-(chloromethyl)pyrimidine (11.50 g, 55% yield) as a white solid.1H NMR (400 MHz, CDCl3): δ 8.66 (s, 1H), 4.65 (s, 2H).
Step-2: Synthesis of 2,4-dichloro-5-(iodomethyl)pyrimidine
[0286] Title compound was prepared in a similar manner to general procedure J.2,4-dichloro-5-(chloromethyl)pyrimidine (11.50 g, 58.20 mmol) on treatment with NaI (10.50 g, 69.0 mmol) in acetone (100 mL) resulted in 2,4-dichloro-5-(iodomethyl)pyrimidine (15.20 g, 91% yield). The solid was immediately taken up in toluene and stored under refrigeration.1H NMR (400 MHz, CDCl3): δ 8.60 (s, 1H), 4.39 (s, 2H).
Step-3: Synthesis of N-((2,4-dichloropyrimidin-5-yl)methyl)aniline
[0287] A solution of iodo compound (18, 7.0 g, 24.20 mmol) in toluene (50 mL) was cooled to 0 °C and aniline (2.20 g, 24.20 mmol) was added. The reaction mixture was stirred for 30 min at 0 °C. Then a solution of sodium hydroxide (1.30 g, 32.50 mmol) in water (5 ml) was added and reaction mixture was stirred for 16 h at RT. The reaction was monitored by TLC. After completion of the reaction, water (25 mL) was added and extracted with ethyl acetate (2 x 100 mL). The organic layer was washed with brine solution, dried over anhydrous sodium sulfate and evaporated under reduced pressure to obtain the crude residue. The crude compound was purified by silica gel column chromatography to afford the title compound as a white solid (10 g, 81% yield). LCMS Calcd for [M+H] + 254.11, found 254.09
Step-4: Synthesis of tert-butyl (3-((2-chloro-5-((phenylamino)methyl)pyrimidin-4-yl)amino)phenyl)carbamate
[0288] To a stirred solution of N-((2,4-dichloropyrimidin-5-yl)methyl)aniline (4.0 g, 15.08 mmol) in IPA (30 mL), tert-butyl (3-aminophenyl)carbamate (4.90 g, 23.0 mmol) and DIPEA (8.20 mL, 47 mmol) were added. The reaction mixture was heated at 100 °C for 16 h in a sealed tube. Solvent was then evaporated and the crude thus obtained was purified by flash column chromatography to afford the title compound as off white solid (2.50 g, 37% yield). LCMS Calcd for [M+H] + 425.92, found 426.35
Step-5: Synthesis of tert-butyl (3-(7-chloro-3-phenyl-2-thioxo-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)carbamate
[0289] To a solution of tert-butyl (3-((2-chloro-5-((phenylamino)methyl)pyrimidin-4-yl)amino)phenyl)carbamate (1.50 g, 3.50 mmol) in THF (35 mL) was added DIPEA (2.40 mL, 14.10 mmol) and thiophosgene (0.27 g, 3.50 mmol) at 0 °C. The reaction mixture was stirred at RT for 24 h with TLC monitoring. After completion of the reaction, sodium bicarbonate solution was added. The reaction mixture was partitioned between DCM (2 x 100 mL) and water (50 mL). The organic layer was washed with brine (10 mL), dried over anhydrous sodium sulfate and evaporated under reduced pressure to obtain crude product. The crude product was purified by silica gel column chromatography to afford the title compound as a yellow solid (1.36 g, 82% yield). LCMS Calcd for [M+H] + 467.97, found 468.27
Step-6: Synthesis of tert-butyl (3-(7-((3-chloro-1-methyl-1H-pyrazol-4-yl)amino)-3-phenyl-2-thioxo-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)carbamate
[0290] To a solution of tert-butyl (3-(7-chloro-3-phenyl-2-thioxo-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)carbamate (1.30 g, 2.78 mmol) in IPA (15 mL) was added 3-
chloro-1-methyl-1H-pyrazol-4-amine (0.44 g, 3.34 mmol) and TFA (1 mL). The reaction mixture was heated for 16 h at 110 °C. Reaction was monitored by TLC. After the completion of reaction, the reaction mixture was concentrated, water (10 mL) and saturated sodium bicarbonate (20 mL) solution were added to the residue and extracted with DCM (3 x 200 mL). The combined organic layer was washed with brine solution, dried over anhydrous sodium sulfate and evaporated under reduced pressure to obtain the title compound (1.30 g) that was used as such for the next step without further purification. LCMS Calcd for [M+H] + 563.08, found 562.90
Step-7: Synthesis of 1-(3-aminophenyl)-7-((3-chloro-1-methyl-1H-pyrazol-4-yl)amino)-3-phenyl-3,4-dihydropyrimido[4,5-d]pyrimidine-2(1H)-thione
[0291] To an ice-cold solution of tert-butyl (3-(7-((3-chloro-1-methyl-1H-pyrazol-4-yl)amino)-3-phenyl-2-thioxo-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)carbamate (1.30 g, 2.30 mmol) in DCM (20 mL) and MeOH (10 mL) was added 4N HCl in dioxane (5 mL). The reaction mixture was stirred for 16 h at RT. The reaction was monitored by TLC. After completion of the reaction, the solvent was evaporated followed by addition of water (10 mL) and saturated sodium bicarbonate (20 mL) solution and extraction with DCM (3 x 200 mL). The combined organic layer was washed with brine solution, dried over anhydrous sodium sulfate and evaporated under reduced pressure to obtain crude product. The crude product was purified by silica gel column chromatography to afford the title compound as a brown solid (0.20 g). LCMS Calcd for [M+H] + 462.96, found 463.0. Purity: 68%
Step-8: Synthesis of (E)-N-(3-(7-((3-chloro-1-methyl-1H-pyrazol-4-yl)amino)-3-phenyl-2-thioxo-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)-4-(dimethylamino)but-2-enamide (Compound 51)
[0292] To an ice-cold solution of 1-(3-aminophenyl)-7-((3-chloro-1-methyl-1H-pyrazol-4-yl)amino)-3-phenyl-3,4-dihydropyrimido[4,5-d]pyrimidine-2(1H)-thione (0.18 g, 0.39 mmol) and trans-N,N-dimethylaminocrotonic acid hydrochloride (0.077 g, 0.47 mmol) in dichloromethane (10 mL) was added triethyl amine (1.2 mmol) followed by drop wise addition of propylphosphonic anhydride (T3P) (0.26 g, 0.97 mmol). The mixture was stirred at RT for 6 h. Completion of the reaction was monitored by TLC. The reaction mixture was portioned between 5% methanol in dichloromethane and saturated bicarbonate solution. The organic phase was dried over anhydrous sodium sulfate, filtered and concentrated. The crude obtained was purified by silica gel chromatography to afford the title compound as off white solid (Compound 51, 0.010 g, 5% yield).1H NMR (400 MHz, DMSO-d6): δ 10.36 (bs, 1H), 8.97 (bs, 1H), 8.25 (s, 1H), 7.72 (bs, 2H), 7.48-7.42 (m, 5H), 7.36-7.32 (m, 1H), 7.03 (d, J = 7.6 Hz, 1H), 6.76-6.60 (m, 2H), 6.30 (d, J = 14.8 Hz, 1H), 4.95 (s, 2H), 3.50 (s, 3H), 3.12 (bs, 2H), 2.21 (s, 6H); LCMS Calcd for [M+H] + 574.10, found 574.41
Scheme 28: Preparation of (E)-N-(3-(3-benzyl-7-((1-methyl-1H-pyrazol-3-yl)amino)-2-oxo-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)-4-(dimethylamino)but-2-enamide (Compound 52):
Step 1: Preparation of ethyl 4-((3-((tert-butoxycarbonyl) amino) phenyl) amino)-2-(methylthio) pyrimidine-5-carboxylate (106):
[0293] Title compound (106) was prepared as off-white solid (142 g; Yield: 74%) in a manner substantially similar to procedure mentioned in General procedure O.1H-NMR (400 MHz, CDCl3): ^ 10.36 (s, 1H), 8.77 (d, 1H), 7.89 (s, 1H), 7.35 (d, J = 8.0 Hz, 1H), 7.25-7.22 (m, 1H), 7.03 (d, J = 8.0 Hz, 1H), 6.51 (s, 1H), 4.35 (q, J = 7.2 Hz, 2H), 2.54 (s, 3H), 1.51 (s, 9H), 1.42-1.38 (m, 3H). LCMS: [M+H]+ 405.21, 89.28%.
Step 2: Preparation of tert-butyl (3-((5-(hydroxymethyl)-2-(methylthio)pyrimidin-4-yl)amino)phenyl)carbamate (107):
[0294] Title compound was prepared in a manner substantially similar to procedure mentioned in General procedure P. The crude was triturated with dichloromethane afforded 107 as off white solid (40.0 g; Yield: 31%).1H-NMR (400 MHz, CDCl3): ^ 8.09 (s, 1H), 7.86 (m, 2H),
7.36 (d, J = 8.0 Hz, 1H), 7.25-7.15 (m, 1H), 6.95 (d, J = 8.0 Hz, 1H), 6.55 (s, 1H), 4.59 (s, 2H), 2.50 (s, 3H), 1.51 (s, 9H). LCMS: [M+H]+ 363.05, 91.24%.
Step 3: Preparation of tert-butyl (3-((5-formyl-2-(methylthio)pyrimidin-4-yl)amino)phenyl)carbamate (108):
[0295] Title compound (108) was prepared as a pale yellow solid (31.0 g; Yield: 78%) in a manner substantially similar to procedure mentioned in General procedure Q.1H-NMR (400 MHz, CDCl3): ^ 10.59 (s, 1H), 9.75 (s, 1H), 8.42 (s, 1H), 7.97 (s, 1H), 7.35 (d, J = 8.0 Hz, 1H), 7.04 (d, J = 8.0 Hz, 1H), 6.59 (s, 1H), 3.48 (s, 1H), 2.58 (s, 3H), 1.52 (s, 9H). LCMS: [M+H]+ 361.30, 97.51%.
Step 4: Preparation of tert-butyl (E)-(3-((5-((benzylimino)methyl)-2(methylthio)pyrimidin-4-yl)amino)phenyl)carbamate (110):
[0296] Title compound (110) was prepared as a yellow solid (28 g; Yield: 72%) in a manner substantially similar to procedure mentioned in General procedure R.1H-NMR (400 MHz, CDCl3): ^ 12.15 (s, 1H), 8.31 (s, 1H), 8.16 (s, 1H), 7.91 (s, 1H), 7.41 (m, 4H), 7.35-7.33 (m, 1H), 7.32-7.29 (m, 1H), 7.26-7.22 (m, 1H), 7.03 (d, J = 8.0 Hz, 1H), 6.46 (s, 1H), 4.84 (s, 2H), 2.59 (s, 3H), 1.52 (s, 9H). LCMS: [M+H]+ 450.38; 99.66%.
Step 5: Preparation of tert-butyl (3-((5-((benzylamino)methyl)-2-(methylthio)pyrimidin-4-yl)amino)phenyl)carbamate (111):
[0297] Title compound (111) was prepared as a pale yellow solid (40 g; Yield: 80%) in a manner substantially similar to procedure mentioned in General procedure S. LCMS: [M+H]+ 452.44; 83.57%
Step 6: Preparation of tert-butyl (3-(3-benzyl-7-(methylthio)-2-oxo-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)carbamate (112):
[0298] Title compound was prepared in a manner substantially similar to procedure mentioned in General procedure T. The crude was triturated with diethyl ether afforded 112 as off white solid (12 g; Yield: 28%).1H-NMR (400 MHz, CDCl3): ^ 8.03 (s, 1H), 7.50 (s, 1H), 7.37 (m, 6H), 7.26 (m, 1H), 6.96 (m, 1H), 6.59 (s, 1H), 4.69 (s, 2H), 4.34 (s, 2H), 2.16 (s, 3H), 1.50 (s, 9H). LCMS: [M+H]+ 478.16; 95.62%.
Step 7: Preparation of tert-butyl (3-(3-benzyl-7-(methylsulfonyl)-2-oxo-3,4-dihydropyrimido [4,5-d]pyrimidin-1(2H)-yl)phenyl)carbamate (113):
[0299] Title compound was prepared in a manner substantially similar to procedure mentioned in General procedure U. The crude was triturated with diethyl ether afforded 113 as an off white solid (8.0 g; Yield: 76%).1H-NMR (400 MHz, CDCl3): ^ 8.39 (s, 1H), 7.63 (s, 1H), 7.40 (m, 6H), 7.17 (d, J = 8.0 Hz, 1H), 6.95 (d, J = 8.0 Hz, 1H), 6.61 (s, 1H), 4.71 (s, 2H), 4.48 (s, 2H), 2.97 (s, 3H), 1.49 (s, 9H). LCMS: [M+H]+ 510.31, 93.69%.
Step 8: Preparation of tert-butyl (3-(3-benzyl-7-((1-methyl-1H-pyrazol-3-yl)amino)-2-oxo-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)carbamate (114):
[0300] Title compound was prepared in a manner substantially similar to General procedure V, tert-butyl (3-(3-benzyl-7-(methylsulfonyl)-2-oxo-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)carbamate (113) and 1-methyl-1H-pyrazol-3-amine (41) gave (tert-butyl (3-(3-benzyl-7-((1-methyl-1H-pyrazol-3-yl)amino)-2-oxo-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)carbamate (114) as a brown solid (Yield: 77%), which was used directly for the next step without any further purification. MS: [M+H]+ 527.46.
Step 9: Preparation of 1-(3-aminophenyl)-3-benzyl-7-((1-methyl-1H-pyrazol-3-yl)amino)-3,4-dihydropyrimido[4,5-d]pyrimidin-2(1H)-one (115):
[0301] Title compound was prepared in a manner substantially similar to General procedure W, tert-butyl (3-(3-benzyl-7-((1-methyl-1H-pyrazol-3-yl)amino)-2-oxo-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)carbamate (114) gave 1-(3-aminophenyl)-3-benzyl-7-((1-methyl-1H-pyrazol-3-yl)amino)-3,4-dihydropyrimido[4,5-d]pyrimidin-2(1H)-one (115) as a brown solid (Yield: 93%), which was used directly for the next step. MS: [M+H]+ 427.44.
Step 10: Preparation of (E)-N-(3-(3-benzyl-7-((1-methyl-1H-pyrazol-3-yl)amino)-2-oxo-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)-4-(dimethylamino)but-2-enamide (Compound 52):
[0302] Title compound was prepared in a manner substantially similar General procedure X, 1-(3-aminophenyl)-3-benzyl-7-((1-methyl-1H-pyrazol-3-yl)amino)-3,4-dihydropyrimido[4,5-d]pyrimidin-2(1H)-one (115) and trans-N,N-dimethylaminocrotonic acid hydrochloride gave (E)-N-(3-(3-benzyl-7-((1-methyl-1H-pyrazol-3-yl)amino)-2-oxo-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)-4-(dimethylamino)but-2-enamide Compound 52, as a white solid (48 mg; Yield: 13%), after prep-HPLC purification.1H-NMR (400 MHz, CDCl3): δ 10.17 (s, 1H), 9.51 (s, 1H), 8.08 (s, 1H), 7.72 (d, J = 8.4 Hz, 1H), 7.60 (s, 1H), 7.43-7.35 (m, 5H), 7.33-7.29 (m, 1H), 7.10 (s, 1H), 7.01 (d, J = 8.8 Hz, 1H), 6.75-6.69 (m, 1H), 6.27 (d, J = 15.3 Hz, 1H), 5.51 (s, 1H), 4.62 (s, 2H), 4.39 (s, 2H), 3.59 (s, 3H), 3.06 (d, J = 4.8 Hz, 2H), 2.17 (s, 6H). MS: [M+H]+ 538.32.
Scheme 30: Alternative Preparation of (E)-N-(3-(7-((3-chloro-1-methyl-1H-pyrazol-4- yl)amino)-2-oxo-3-phenyl-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)-4- (dimethylamino)but-2-enamide (Compound 35):
Step 1: Preparation of 5-(hydroxymethyl)pyrimidine-2,4(1H,3H)-dione (119):
[0308] An ice-cold solution of pyrimidine-2,4(1H,3H)-dione (118) (10 g, 89.21 mmol) and paraformaldehyde (9.63 g, 107.05 mmol) in aqueous potassium hydroxide (132 mL, 0.5 M,
66.74 mmol) was heated at 55 °C for 14 hours. After completion of starting material (TLC), the reaction mixture was cooled to 0 °C and the pH was adjusted to 6 with 12N hydrochloric acid, the resulting white precipitate was filtered through sintered funnel and washed with diethyl ether afforded 119 as a white solid (6.3 g, Yield: 50%) which was used directly for the next step.1H-NMR (400 MHz, DMSO-d6): ^ 10.98 (bs, 1H), 10.64 (bs, 1H), 7.24 (s, 1H), 4.78 (m, 1H), 4.12 (d, J = 12.8 Hz, 2H). LCMS: [M+H]+ 143.04 (99.92% purity).
Step 2: Preparation of 2,4-dichloro-5-(chloromethyl)pyrimidine (120):
[0309] To an ice-cold solution of 5-(hydroxymethyl)pyrimidine-2,4(1H,3H)-dione (119) (10 g, 70.36 mmol) in toluene (25 mL) was added phosphoryl chloride (14 mL, 140.72 mmol) then N,N-diisopropylethylamine (37 mL, 211 mmol). The reaction mixture was heated at 120 °C for 16 hours. After the complete disappearance of starting material on TLC, the reaction mixture was quenched slowly with sodium bicarbonate solution and extracted with ethyl acetate (3 x 200 mL). The combined organic layer was washed with brine, dried over anhydrous sodium sulfate, filtered and evaporated under reduced pressure afforded 120 as a brown solid (12 g, Yield: 86%) which was used directly for the next step.1H NMR (400 MHz, CDCl3): ^ 8.66 (s, 1H), 4.64 (s, 2H). MS: [M+H]+ 197.0
Step 3: Preparation of 2,4-dichloro-5-(iodomethyl)pyrimidine (121):
[0310] To a solution of 2,4-dichloro-5-(chloromethyl)pyrimidine (120) (8.0 g, 40.51 mmol in acetone (40 mL) was added sodium iodide (9.71 g, 64.82 mmol). The reaction mixture was stirred at room temperature for 30 min and heated to reflux for 2 hours. After completion of reaction (TLC monitoring), the reaction mixture cooled to room temperature. The resulting white precipitate was filtered through sintered funnel and washed with acetone. The filtrate was concentrated under reduced pressure afforded 121 as a brown solid (10 g, Yield: 85%) which was used directly for the next step.1H-NMR (400 MHz, CDCl3): ^ 8.60 (s, 1H), 4.39 (s, 2H). Step 4: Preparation of N-((2,4-dichloropyrimidin-5-yl)methyl)aniline (122):
[0311] To an ice-cold solution of 2, 4-dichloro-5-(iodomethyl)pyrimidine (121) (5.0 g, 17.30 mmol) in acetone (50 mL) was added potassium carbonate (5.26 g, 38.06 mmol) and aniline (1.93 g, 20.76 mmol). The resulting reaction mixture was stirred at room temperature for 16 hours. After completion the reaction (as per TLC monitoring), the resulting white precipitate was filtered through sintered funnel and washed with acetone. The filtrate was concentrated under reduced pressure and crude was purified by column chromatography on silica gel (100-200 mesh) using 15% ethyl acetate-hexane as an eluent afforded 122 as a brown solid (2.5 g, Yield: 57%).1H-NMR (400 MHz, CDCl3): ^ 8.61 (s, 1H), 7.07 (t, J = 7.6 Hz, 2H), 6.58 (m, 3H), 6.30 (bs, 1H), 4.33 (m, 2H). LCMS: [M+H]+ 254.03 (99.01% purity).
Step 5: Preparation of tert-butyl (3-(7-chloro-2-oxo-3-phenyl-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)carbamate (123):
[0312] To an ice-cold solution of N-((2,4-dichloropyrimidin-5-yl)methyl)aniline (122) (500 mg, 1.96 mmol), in isopropanol (5 mL) was added N,N-diisopropylethylamine (1.47 mL, 8.42 mmol) and tert-butyl (3-aminophenyl)carbamate (105) (409 mg, 1.96 mmol). The resulting reaction mixture was heated at 100 °C for 16 hours in a sealed tube. After completion of reaction (TLC monitoring), the solvent was then evaporated under reduced pressure and resulting crude was purified by column chromatography on silica gel (100-200 mesh) using 30% ethyl acetate-hexane as an eluent afforded 123 as a brown solid (500 mg, Yield: 60%).1H-NMR (400 MHz, DMSO-d6): δ 9.41 (s, 1H), 8.96 (s, 1H), 8.10 (s, 1H), 7.73 (s, 1H), 7.25 (m, 2H), 7.12 (m, 3H), 6.61 (m, 3H), 6.14 (t, J = 7.2 Hz, 1H), 4.26 (m, 2H) and 1.53 (s, 9H). LCMS: [M+H]+ 426.14 (93% purity).
Step 6: Preparation of tert-butyl (3-(7-chloro-2-oxo-3-phenyl-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)carbamate (124):
[0313] To an ice-cold solution of tert-butyl (3-(7-chloro-2-oxo-3-phenyl-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)carbamate (123) (500 mg, 1.17 mmol) in tetrahydrofuran (6 mL) was added N,N-diisopropylethylamine (0.81 ml, 4.68 mmol) and triphosgene (139 mg, 0.46 mmol). The reaction mixture was stirred at room temperature for 3 hours. After completion of the reaction (TLC monitoring), aqueous triethylamine solution was added and extracted with dichloromethane (3 times). The combined organic layer was washed with brine and dried over sodium sulfate and evaporated under reduced pressure to obtain the crude residue. The crude was purified by column chromatography on silica gel (100-200 mesh) using 30% ethyl acetate-hexane as an eluent afforded 124 as a brown solid (450 mg, Yield: 85%).1H-NMR (400 MHz, DMSO-d6): δ 9.54 (s, 1H), 8.43 (s, 1H), 7.58 (s, 1H), 7.44 (m, 4H), 7.29 (t, J = 7.2 Hz, 3H), 6.94 (s, 1H), 5.0 (s, 2H) and 1.47 (s, 9H). LCMS: [M+H]+ 452.27 (99% purity).
Step 7: Preparation of tert-butyl (3-(7-((3-chloro-1-methyl-1H-pyrazol-4-yl)amino)-2-oxo-3-phenyl-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)carbamate (125):
[0314] Title compound was prepared in a manner substantially similar to procedure mentioned in General procedure V, (tert-butyl(3-(7-chloro-2-oxo-3-phenyl-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)carbamate (124) and 3-chloro-1-methyl-1H-pyrazol-4-amine (44) gave tert-butyl (3-(7-((3-chloro-1-methyl-1H-pyrazol-4-yl)amino)-2-oxo-3-phenyl-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)carbamate (125) as a brown solid in 70% yield, which was used directly for the next step. MS: [M+H]+ 547.17.
Step 8: Preparation of 1-(3-aminophenyl)-7-((3-chloro-1-methyl-1H-pyrazol-4-yl)amino)-3-phenyl-3,4-dihydropyrimido[4,5-d]pyrimidin-2(1H)-one (126):
[0315] Title compound was prepared in a manner substantially similar to procedure mentioned in General procedure W, tert-butyl (3-(7-((3-chloro-1-methyl-1H-pyrazol-4-yl)amino)-2-oxo-3-phenyl-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)carbamate (125) gave 1-(3-aminophenyl)-7-((3-chloro-1-methyl-1H-pyrazol-4-yl)amino)-3-phenyl-3,4-dihydropyrimido[4,5-d]pyrimidin-2(1H)-one (126) as a brown solid (800 mg, Yield: 82%) which was used directly for the next step. MS: [M+H]+ 447.08.
Step 9: Preparation of (E)-N-(3-(7-((3-chloro-1-methyl-1H-pyrazol-4-yl)amino)-2-oxo-3-phenyl-3,4-dihydropyrimido[4,5-d]pyrimidin-1(2H)-yl)phenyl)-4-(dimethylamino)but-2-enamide (Compound 35):
[0316] Title compound was prepared in a manner substantially similar to procedure mentioned in General procedure X, 1-(3-aminophenyl)-7-((3-chloro-1-methyl-1H-pyrazol-4-yl)amino)-3-phenyl-3,4-dihydropyrimido[4,5-d]pyrimidin-2(1H)-one (126) and trans-N,N-dimethylaminocrotonic acid hydrochloride gave the titled compound, which was purified by prep-HPLC purification to afforded the title compound Compound 35 as a white solid (285 mg, Yield: 23%).1H-NMR (400 MHz, DMSO-d6): δ 10.27 (bs, 1H), 8.86 (s, 1H), 8.21 (s, 1H), 7.73 (s, 2H), 7.51-7.40 (m, 5H), 7.30-7.25 (m, 1H), 7.09 (d, J = 7.6 Hz, 1H), 6.76-6.70 (m, 2H), 6.29 (d, J = 15.4 Hz, 1H), 4.88 (s, 2H), 3.50 (s, 3H), 3.05 (d, J = 4.8 Hz, 2H) and 2.16 (s, 6H). MS:
[M+H]+ 558.16.

NEW DRUG APPROVALS
ONE TIME
$10.00
GST-HG-121
GST-HG-121
mw 431.4
C23 H29 N07
Fujian Cosunter Pharmaceutical Co Ltd
Preclinical for the treatment of hepatitis B virus infection
This compound was originally claimed in WO2018214875 , and may provide the structure of GST-HG-121 , an HBsAg inhibitor which is being investigated by Fujian Cosunter for the treatment of hepatitis B virus infection; in June 2019, an IND application was planned in the US and clinical trials of the combination therapies were expected in 2020. Fujian Cosunter is also investigating GST-HG-131 , another HBsAg secretion inhibitor, although this appears to be being developed only as a part of drug combination.
WO2017013046A1
PATENT
WO2018214875
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2018214875&_cid=P21-KB0QYA-12917-1
PATENT
WO-2020103924
Novel crystalline forms of 11-oxo-7,11-dihydro-6H-benzo[f]pyrido[1,2-d][1,4]azepine, a hepatitis B surface antigen and HBV replication inhibitor, useful for treating HBV infection.
Step H: Compound 9 (15.80 g, 35.95 mmol) was dissolved in dichloromethane (150.00 mL), and trifluoroacetic acid (43.91 mL, 593.12 mmol) was added. The reaction solution was stirred at 10 degrees Celsius for 3 hours. The reaction solution was concentrated under reduced pressure and spin-dried, sodium bicarbonate aqueous solution (100.00 mL) was added, and dichloromethane (100.00 mL) was extracted. The organic phase was dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to obtain compound 10.
Step J: Compound 12 (875.00 mg, 1.90 mmol) was dissolved in toluene (20.00 mL) and ethylene glycol dimethyl ether (20.00 mL), and tetrachlorobenzoquinone (1.40 g, 5.69 mmol) was added. The reaction solution was stirred at 120 degrees Celsius for 12 hours. The reaction solution was cooled to room temperature, and a saturated aqueous sodium carbonate solution (50.00 ml) and ethyl acetate (60.00 ml) were added. The mixed solution was stirred at 10-15 degrees Celsius for 20 minutes, and the liquid was separated to obtain an organic phase. Add 2.00 mol/L aqueous hydrochloric acid solution (60.00 mL) to the organic phase, stir at 10-15 degrees Celsius for 20 minutes, and separate the liquid. Wash the organic phase with 2 mol/L aqueous hydrochloric acid solution (60.00 mL×2), separate the liquid, and separate the water phase A 2 mol/L aqueous sodium hydroxide solution (200.00 ml) and dichloromethane (200.00 ml) were added. The layers were separated, and the organic phase was dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to obtain compound 13.
Step K: Compound 13 (600.00 mg, 1.31 mmol) was dissolved in methanol (6.00 mL), and 4.00 mol/L aqueous sodium hydroxide solution (2.00 mL, 6.39 equiv) was added. The reaction solution was stirred at 15 degrees Celsius for 0.25 hours. The reaction solution was adjusted to pH=3-4 with a 1.00 mol/L hydrochloric acid aqueous solution, and then extracted with dichloromethane (50.00 mL×3). The organic phases were combined, washed with saturated brine (50.00 mL), and dried over anhydrous sodium sulfate , Filtered and concentrated under reduced pressure to obtain the compound of formula (I). ee value (enantiomeric excess): 100%.
////////////GST-HG-121, Fujian Cosunter, Preclinical , hepatitis B, virus infection
O=C(O)C1=CN2C(=CC1=O)c3cc(OC)c(OCCCOC)cc3OC[C@H]2C(C)(C)C
O=C(O)C1=CN2C(=CC1=O)c3cc(OC)c(OCCCOC)cc3OC[C@H]2C(C)(C)C
ADX-103
ADX-103
CAS 916056-81-0
Preclinical, Antiinflammatory Ophthalmic Agents, Diabetic Retinopathy,
Agents for Ophthalmic Drugs
MF C16 H16 N2 O2
5-Amino-α,α-dimethyl-2-phenyl-6-benzoxazolemethanol
Aldeyra Therapeutics Inc
ADX-103 , an aldehyde trap being investigated by Aldeyra for the treatment of dry eye syndrome; in May 2018, preclinical data were presented at 2018 ARVO Meeting in Honolulu, HI. Aldeyra, in collaboration with an undisclosed company, is also investigating an anti-inflammatory agent for treating ocular inflammation.
PATENT
WO-2020033344
Novel crystalline forms of a specific benzoxazole and it’s salts, process for their preparation, and compositions comprising them are claimed, useful for treating dry eye, inflammation and diabetes, through action as an aldehyde scavenger.
It has now been found that compounds of the present invention, and compositions thereof, are useful for treating, preventing, and/or reducing a risk of a disease, disorder, or condition in which aldehyde toxicity is implicated in the pathogenesis. In general, salt forms or freebase forms, and pharmaceutically acceptable compositions thereof, are useful for treating or lessening the severity of a variety of diseases or disorders as described in detail herein. Such compounds are represented by the chemical structure below, denoted as compound A:
or a pharmaceutically acceptable salt thereof.
[0008] Compounds of the present invention, and pharmaceutically acceptable compositions thereof, are useful for treating a variety of diseases, disorders or conditions, associated with toxic aldehydes. Such diseases, disorders, or conditions include those described herein.
[0009] Compounds provided by this invention are also useful for the study of certain aldehydes in biology and pathological phenomena.
Scheme 1 – Synthesis of Compound A
Step 1: Synthesis of Compound A2
[00549] A 30L jacketed vessel equipped with mechanical agitation, baffle and nitrogen bleed was charged with methanol (10L). Compound A1 (2.0kg) was added, followed by further methanol to rinse (9L). The reaction mixture was warmed to Tjacket=40°C. Once temperature had stabilized, sulfuric acid (220 mL, 0.4eq.) was slowly added. Once addition was complete, agitation was maintained for 30 mins then the vessel was heated to Tjmt=62°C. Reaction progress was
monitored by LC-MS analysis of reaction mixture. The reaction does not go to completion but is deemed complete when no change is apparent in ratio of starting material : product.
[00550] The vessel contents were cooled to Tjmt=24°C and stirred 60 minutes before filtration under vacuum. The filter cake was air dried for 2 hours and the contents then dissolved in ethyl acetate (18L) which was then washed sequentially with saturated sodium bicarbonate (8L), water (8L) and brine (8L) before drying over sodium sulfate, filtration and evaporation in vacuo. Compound A2 (1.5kg, 68.1%) was obtained as a bright orange powder.
Step 2: Synthesis of Compound A3
[00551] A 30L jacketed vessel equipped with mechanical agitation, baffle and nitrogen bleed was charged with /V,/V-dimethylformamide (16L). Compound A2 (1.5kg) was added and the brown reaction mixture set to cool to Tint<20oC. Once temperature had stabilized, A-bromosucci ni mi de (l.5kg, 1.1 eq.) was added portion wise, maintaining Tint<27°C. Once addition was complete, the reaction was allowed to stir until starting material content was <1% AUC (250nm) by LCMS analysis.
[00552] A secondary jacketed vessel equipped with mechanical agitation, baffle and nitrogen bleed was charged with ethyl acetate (16L) and deionized water (22L). The reaction mixture was vacuum transferred into this vessel and held at high agitation for not less than 30 minutes. The aqueous layer was discharged and the organic layer washed with saturated sodium chloride (2 x 8L) then dried over sodium sulfate before evaporation in vacuo to Compound A3 as a deep brown oil (2.lkg, 100.8%), suitable for use in following step without purification.
Step 3: Synthesis of Compound A4
[00553] A 30L jacketed vessel equipped with mechanical agitation, baffle and nitrogen bleed was charged with dichloromethane (9L). Compound A3 (2.lkg) was added and the reaction mixture cooled to Tmt<l°C. A solution of Di-/er/-butyl dicarbonate (3.6kg, 2.2 eq.) in dichloromethane (0.5L) was added followed by a solution of A, A-di methyl ami nopyri di ne (92g, 0.1 eq.) in dichloromethane (0.5L). The resultant clear brown solution was stirred for 30 minutes whereupon pyridine (1.3L, 1.7 eq.) was dropwise added, maintaining Tint<5°C. Upon complete addition internal temperature was ramped from Tint=l°C to Tint=20°C over 18 hours.
[00554] The reaction mixture was sequentially washed with saturated sodium chloride (3 x 4.5L), 10 % w/v aqueous citric acid (2 x 4L), saturated sodium bicarbonate (4L), aqueous hydrochloric acid (1M, 4L), saturated sodium bicarbonate (4L) and saturated sodium chloride (4L) then dried over sodium sulfate and evaporated in vacuo with one azeotropic distillation with toluene (2L) to a very dark, heavy tar (3.4kg).
[00555] The isolated tar was mixed with absolute ethanol (3.1L) for 2 days whereupon it was filtered providing light cream colored, granular solids and a black mother liquor. The solids were washed with ice-cold ethanol (3 x 1L) and dried to constant mass. Compound A4 was obtained as off- white granules (1.7 kg, 50.2%).
Step 4: Synthesis of Compound AS
[00556] A 30L jacketed vessel equipped with mechanical agitation, baffle and nitrogen bleed was charged with reagent alcohol (6.1 L) and Compound A4 (0.8kg), Tmt<20°C. Iron powder (0.5kg, 5.0 eq.) was added and the suspension stirred vigorously for 30 minutes. Acetic acid (glacial, 1.6L, 15.7 eq.) was added, maintaining Tint<30C.
[00557] Once LCMS confirmed complete consumption of starting material, ethyl acetate (10.2L) and water (10.2L) were added. Sodium bicarbonate (2.3kg, 15.9 eq.) was added portion wise and the layers separated once gas evolution had ceased. The aqueous layer was washed with ethyl acetate until LCMS indicated no further product was being extracted (8 x 2L) and the combined organic layers were sequentially washed with deionized water (6L) then saturated sodium chloride (6L) before drying over magnesium sulfate and evaporation in vacuo. Compound A5 was obtained as a light orange solid (0.7kg, 91.5%).
Step 5: Synthesis of Compound A6
[00558] A 30L jacketed vessel equipped with mechanical agitation, baffle and nitrogen bleed was charged with dichloromethane (9L), Compound A5 (0.7kg), and the reaction mixture cooled to Tint 20°C. Benzoyl chloride (0.3L, 1.5 eq.) was added and the reaction stirred 15 minutes. N,N-dimethylaminopyridine (7g, 0.04 eq.) in dichloromethane (0.1L) was added and the reaction stirred 15 minutes. Pyridine (0.5L, 2.5 eq.) was dropwise added, maintaining Tint<20°C. Upon complete addition the reaction was stirred until LCMS indicated consumption of starting material.
[00559] The reaction mixture was washed with deionized water (11L) and the organic layer extracted sequentially with aqueous hydrochloric acid (1M, 3 x 5L), saturated aqueous sodium bicarbonate (11 L), saturated sodium chloride (11 L), dried over magnesium sulfate and evaporated in vacuo. Compound A6 was obtained as a cream colored solid, suitable for use without further purification (0.9kg, 100.7%).
Step 6: Synthesis of Compound A 7
[00560] A 30L jacketed vessel equipped with mechanical agitation, baffle and nitrogen bleed was charged with l,2-dimethoxy ethane (16L) and temperature set to Tint=2l°C. Compound A6 (0.9kg) was added and stirred to dissolution. Copper iodide (0.3kg, 1.0 eq.) was added and the mixture stirred 15 minutes. l, lO-phenanthroline (0.3kg, 1.2 eq.) was added and the mixture stirred 15 minutes. Cesium carbonate (l .5kg, 3.0 eq.) was added and the reaction was stirred for 15 minutes. The reaction temperature was ramped to Tint=80-85oC and maintained for 23 hours whereupon it was cooled to Tmt=20°C.
[00561] The reaction mixture was filtered through a celite pad, washing sequentially with deionized water (8L) and ethyl acetate (8L). The organic layer was extracted sequentially with deionized water (2 x 5L), saturated sodium chloride (4L), dried over sodium sulfate and evaporated in vacuo. Compound A7 was obtained as a brown solid, suitable for use without further purification (0.8kg, 104.1%).
Step 7: Synthesis of Compound A8
[00562] A 12L 3 -neck round bottom flask with nitrogen bleed and mechanical stirring was charged with a solution of Compound A7 (0.8kg) in dichloromethane (3.6L) and cooled to Tmt<5°C in an ice bath. Hydrochloric acid in dioxane (4M, 1 2L, 3.1 eq.) was added dropwise with vigorous stirring, maintaining Tmt<25°C. Once addition was complete, the reaction mixture was allowed to stir for 18 hours at Tint=20-25oC.
[00563] The reaction mixture was filtered and the filter cake washed with dichloromethane (2 x 1L) and dried to constant mass. The hydrochloride salt of Compound A8 was isolated as an off-white solid (0.5kg, 88.7%).
Step 8: Synthesis of Compound A
[00564] A 12L 3 -neck round bottom flask with nitrogen bleed and mechanical stirring was charged with a solution of Compound A8 (0.5kg) in tetrahydrofuran (4.8L) and cooled to Tint<-30°C in a dry-ice / acetone bath. Methylmagnesium bromide (3.4M in 2-methyltetrahydrofuran, 2.4L, 5.0eq.) was added slowly, maintaining Tmt<-lO°C. Once addition was complete, the reaction was allowed to warm to room temperature overnight.
[00565] Saturated aqueous ammonium chloride (2L) and ethyl acetate (2L) were added and the reaction mixture stirred for 30 minutes. The aqueous layer was extracted with further ethyl acetate (2 x 2L) and the combined organic layers washed with saturated sodium chloride (2L), dried over sodium sulfate and evaporated in vacuo to a dark heavy oil. The heavy oil was purified by column chromatography on silica gel, eluting with ethyl acetate : heptane 1 : 19 to 1 : 1. Pure Compound A was obtained after evaporation and drying as a brown powder (99.8 g, 23.0%).
Example 1 – Preparation of Free Base Forms A, B and C of Compound A
Compound A
Primary Polymorph Screen
[00566] Based on solubility screen results, a primary polymorph screen using an initial set of 24 solvents, as shown in Table 18, was performed as follows: A) To 24 x 20 mL vials, approximately 50 mg of the received ADX-103 was added; B) The solids were then slurried in 2 mL of the solvents and left placed in an incubator/shaker to temperature cycle between ambient and 40 °C in 4 hour cycles; C) After 72 hours temperature cycling, the mother liquors were removed from the vials and split evenly between 4 x 2 mL vials. The vials were then split between evaporation, crash cooling to 2 °C and -18 °C and anti-solvent addition; and D) Any solids
recovered were analysed by XRPD, any new patterns identified were also analysed by TG/DTA and PLM.
Table 18. Solvents Selected for Initial Primary Polymorph Screen
PATENT
WO2018039197 , as compound I-8.
PATENT
WO 2006127945
WO 2011072141
WO 2014116593
US 20150344447
WO 2020028820
////////////ADX-103, Preclinical, Antiinflammatory, Ophthalmic Agents, Diabetic Retinopathy, Aldeyra Therapeutics Inc,
CC(C)(O)c1cc2oc(nc2cc1N)c3ccccc3
SK1-I , BML 258


SK1-I , BML 258
Sphingosine kinase 1 (SphK1) inhibitor; antiproliferative
- (1E)-1,2,4-Trideoxy-4-(methylamino)-1-(4-pentylphenyl)-D-erythro-pent-1-enitol
- (E,2R,3S)-2-(Methylamino)-5-(4-pentylphenyl)pent-4-ene-1,3-diol
- D-erythro-Pent-1-enitol, 1,2,4-trideoxy-4-(methylamino)-1-(4-pentylphenyl)-, (1E)-
| Name: | (2R,3S,4E)-N-methyl-5-(4′-pentylphenyl)-2-aminopent-4-ene-1,3-diol . HCl |
| Formula: | C17H27NO2 . HCl |
| MW: | 313.9 |
| CAS: | 1072443-89-0
|
- Originator Enzo Biochem; Virginia Commonwealth University
- Developer Enzo Biochem
- Class Antineoplastics; Small molecules
- Mechanism of Action Sphingosine kinase inhibitors
- Preclinical Autoimmune hepatitis; Haematological malignancies; Liver cancer; Solid tumours
- 07 May 2019 Preclinical trials in Liver cancer in USA (unspecified route)
- 03 Dec 2018 SK1 I is available for licensing as of 03 Dec 2018. http://www.enzo.com/
- 03 Dec 2018 Enzo Biochem has patent pending for SK1 I worldwide
SK1 I, a small molecule that specifically inhibits sphingosine kinase 1, is being developed by Enzo Biochem for the treatment of cancer and autoimmune diseases. Preclinical development is underway for the treatment of solid tumours, liver cancer, haematological malignancies and autoimmune hepatitis in the US.
As at December 2018, Enzo Biochem seeks partners for the development of SK1
SK1-I is a sphingosine analog and a sphingosine competitive inhibitor specific for sphingosine kinase 1 (SK1), with ki~10µM and excellent water solubility. It is not to be confused with SKI-I, 5-naphthalen-2-yl-2H-pyrazole-3-carboxylic acid (2-hydroxy-naphthalen-1-ylmethylene)-hydrazide, CAS 306301-68-8, a noncompetitive inhibitor of both SK1 and SK2 with poor water solubility (K.J. French, et al., 2006; N.J. Pyne and S. Pyne, 2010). SK1-I does not inhibit SK2, PKCα, PKCδ, PKA, AKT1, ERK1, EGFR, CDK2, IKKβ or CamK2β. Not only does it decrease sphingosine-1-phosphate levels, it also causes an accumulation of its proapoptotic precursor ceremide. Inhibits tumor cell growth in vitro and in vivo.
PATENTS
US 20100035959
WO 2010127093
US 20100278741
WO 2011025545
Patent
US-10364211
This patent was granted in July 30, 2019 and set to expire on October 24, 2038. Claims methods for synthesizing the compound (2R,3S,4E)-N-methyl-5-(4′-pentylphenyl)-2-aminopent-4-ene-1,3-diol (also known as SK1-I and BML-258 (as HCl salt)) and its intermediates.
(2R,3S,4E)-N-methyl-5-(4′-pentylphenyl)-2-aminopent-4-ene-1,3-diol, also known as SK1-I and BML-258 (as HCl salt), is a pharmaceutical inhibitor of sphingosine kinase 1 initially described in Paugh et al., Blood. 2008 Aug. 15; 112(4): 1382-1391. An existing method for synthesizing SK1-I is disclosed in U.S. Pat. No. 8,314,151.
|
and |
|
|
N-Boc-(D)-Serine Methyl Ester
Protection of N-Boc-(D)-Serine Methyl Ester
(R)—Garner Aldehyde
Addition of 4-Pentylphenyl Acetylene to the Above Aldehyde
Deprotection of the Above Oxazolidine
Reduction of the Above Alcohol
Deprotection to SK1-I (BML-258)
PATENT
WO2018237379 ,
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2018237379
claiming sphingosine pathway modulating compounds for the treatment of cancers, assigned to Enzo Biochem Inc , naming different team
Sphingosine- 1 -phosphate (SIP) was discovered to be a bioactive signaling molecule over 20 years ago. Studies have since identified two related kinases, sphingosine kinase 1 and 2 (a/k/a sphingosine kinase “type I” and “type II” respectively, and SphKl and SphK2 respectively), which catalyze the phosphorylation of sphingosine to SIP. Extracellular SIP can bind to and activate each of five S IP-specific, G protein-coupled receptors (designated S IPR1-5) to regulate cellular and physiological processes in an autocrine or paracrine manner. Selective inhibitors of each of sphingosine kinase 1 and 2, as well as both nonselective and selective agonists of SlPRs, have been developed and are known in the art.
Product Literature References
General Literature References
/////////SK1-I , SK1I , SK1 I , BML 258, Enzo Biochem, Virginia Commonwealth, Preclinical, solid tumours, liver cancer, haematological malignancies, autoimmune hepatitis,
CCCCCC1=CC=C(/C=C/[C@H](O)[C@H](NC)CO)C=C1.Cl
SYN 01

SYN-01, SYN-510
Synthena AG
Preclinical
Synthena , presumed to be under license from University of Bern , is investigating (presumably SYN-01 ), a lead from the tricyclo(tc)-DNA based antisense oligonucleotides (AON) developed using its proprietary tricyclo-DNA technology platform, for the treatment of Duchenne muscular dystrophy. In January 2017, the drug was listed as being in preclinical development.
Patent
WO-2019142135
Process for preparing tricyclo-deoxyribonucleic acid (tc-DNA) which may be used as building blocks for tc-DNA containing antisense oligonucleotide-based therapies.
Antisense technology is an effective means for reducing the expression of specific gene products and can therefore be useful in therapeutic, diagnostic, and research applications.
Generally, the principle behind antisense technology is that an antisense oligomeric compound (a sequence of nucleotides or analogues thereof) hybridizes to a target nucleic acid and modulates gene expression activities or function, such as transcription and/or translation.
[003] Antisense oligomeric compounds may be prepared from chemically-modified antisense oligonucleotides, which may include a variety of different structural variations depending upon the therapeutic strategy. For example, tricyclo-deoxyribonucleic acids (tc-DNA) are conformationally constrained DNA analogs.
[004] There is a need in the field for processes that allow for the bulk preparation of tc-DNA nucleoside precursors that may be used as building blocks for tc-DNA containing antisense oligonucleotide-based therapies.
Example 4 – Cvclopropanation of Compound 17 with Carbenoid Prepared from CH2I2 and Et2Zn in the Absence of Additives
[00127] According to the following scheme, compound 17 was converted to tc-DNA Nucleoside Precursor 18 using the cyclopropanation conditions set forth in Examples 4 to 7 :
[00128] 1.07 g purified a-anomer (3.736 mmol) 17 was dissolved in 37 ml of dry CH2C12 and cooled to 0 °C (ice). Subsequently, 22.3 ml (22.3 mmol, 6 eq.) Et2Zn 1.0 M in hexane (Aldrich) were added dropwise and stirred under Ar for 30 min at 0 °C. Then, 3.02 ml (37.2 mmol, 10 eq.) of CH2I2 were added dropwise over 15 min at the same temperature and stirred for further 2 h at 0 °C. Afterwards the cooling bath was removed and the mixture was stirred for additional 21 h at ambient temperature. TLC showed substantial amount of unreacted a- 17. It was diluted by addition of EtOAc and quenched with 50 mL of sat. aqueous NH4Cl. Extractive work-up provided 1.79 g of crude which was purified by chromatography on silica-gel giving 0.43 g (39%) of 18 and 0.49 g of mixture of compound 17 and 18 (approximately 20:80).
PATENT
WO2018193428
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2018193428
claiming a composition comprising an oligomeric compound having tricyclo-deoxyribonucleic acid (tc-DNA) nucleosides and a lipid moiety.
EXAMPLE 1
Inventive compositions for the treatment of Duchenne muscular dystrophy
Evaluation of efficacy
[00464] Adult mdx mice were treated weekly over 4 weeks with intravenous injections of different 13-mer AONs targeting the donor splice site of exon 23 of the dystrophin pre-mRNA (M23D: +2-11), namely with either SY-0308, SY-0210 and the inventive SY-0299, SY-0343, SY-0442 and SY-0455. SY-0308 (also named “tcDNA-PO M23D” interchangeably herein) corresponds to p-CCTCGGCTTACCT-OH of SEQ ID NO: l, with all nucleotides being tc-DNAs and all internucleosidic linkage groups being phosphorodiester linkage groups, and p being a phosphate moiety at the 5′ end. SY-0210 (also named “tcDNA-PS M23D” interchangeably herein) corresponds to p-CCTCGGCTTACCT-OH of SEQ ID NO: 1, with all nucleotides being tc-DNAs and all internucleosidic linkage groups being phosphorothioate linkage groups, and p being a phosphate moiety at the 5′ end. The inventive composition SY-0343 is herein interchangeably referred to as “Palm-2PS-tcDNA-PO M23D” which is depicted in the following:
[00465] The inventive composition SY-0442 is herein interchangeably referred to as “Palm-lPS-tcDNA-PO M23D” which is depicted in the following:
[00466] The inventive composition SY-0299 is herein interchangeably referred to as “Palm-2PO-tcDNA-PO M23D” which is depicted in the following:
//////////////////SYN-01, SYN 01, SYN01, preclinical , Duchenne muscular dystrophy, University of Bern,
CS 3001
CS-3001
BB 7, VX 033
- Molecular Weight, 478.37
C17 H18 Br F2 N3 O2 S2
CStone Pharmaceuticals Co Ltd, JUNE 2018 IND FILED CHINA
URAT1 inhibitor – useful for treating hyperuricemia and gout.
The compound was originally claimed in WO2017202291 , covering thiophene derivative URAT1 inhibitors, useful for treating hyperuricemia and gouty arthritis, assigned to Medshine Discovery Inc , but naming the inventors.and has been reported in some instances to be a URAT1 modulator. In June 2018, an IND application was filed in

WO-2019101058
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2019101058&tab=FULLTEXT&maxRec=1000
Novel crystalline forms of URAT1 inhibitor (designated as Forms A and B) are claimed. The compounds are disclosed to be useful for treating hyperuricemia and gouty arthritis.
Novel crystalline forms of a URAT1 inhibitor, designated as Forms A and B, and their preparation.
For example, remove 2.0 mL of phosphoric acid into 2000 mL of water, sonicate for 10 minutes, mix, and let cool to room temperature as mobile phase A.
////////////CS-3001, BB 7, VX 033, CHINA, PRECLINICAL, CStone Pharmaceuticals, URAT1 inhibitor, hyperuricemia, gout
O=C(O)C(C)(C)Sc4nnc(Br)n4c2sc(c1CC(F)(F)CCc12)C3CC3
TL 487
TL-487
- Molecular Weight, 507.58, MF C30 H29 N5 O3
Teligene Inc(2E)-N-[3-Cyano-7-ethoxy-4-[(4-phenoxyphenyl)amino]-6-quinolinyl]-4-(dimethylamino)-2-butenamide
(E)-N-(3-cyano-7-ethoxy-4-((4-phenoxyphenyl)amino)quinolin-6-yl)-4-(dimethylamino)but-2-enamide
Maleate in anhydrous or monohydrate CAS, 2326561-36-6, AND 2326561-38-8 form are BTK and HER-2 kinase inhibitor useful for treating cancer
Useful for treating breast cancer, ovary cancer and colon cancer. are BTK and HER-2 kinase inhibitor useful for treating cancer.
Anticancer protein kinase inhibitor
The compound was originally claimed in WO2013152135 , and may provide the structure of TL-487 , a small molecule inhibitor to HERs, being investigated by Teligene for the treatment of breast cancer; in July 2016, the company intended to develop the product as a class 1.1 chemical drug in China.
PATENT
US 20150057312
PATENT
WO2013152135

PATENT
WO-2019096327
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2019096327&redirectedID=true
Novel crystalline maleate salt of (E)-N-(3-cyano-7-ethoxy-4-((4-phenoxyphenyl)amino)quinolin-6-yl)-4-(dimethylamino)but-2-enamide (first disclosed in WO2013152135) and its hydrates (monohydrate) and anhydrates, process for its preparation, composition comprising it and its use for treating cancers such as breast cancer, ovary cancer, colon cancer, prostate cancer, kidney cancer, bladder cancer, stomach cancer, lung cancer, mantle cell lymphoma and multiple myeloma are claimed. The compound is disclosed to be an irreversible inhibitor to BTK and Her-2 (also known as Erb-2 or neu).
///////////////TL-487, PRECLINICAL, CHINA, breast cancer, ovary cancer, olon cancer, BTK, HER-2 kinase inhibitor,
CN(C)C\C=C\C(=O)Nc3cc4c(Nc2ccc(Oc1ccccc1)cc2)c(cnc4cc3OCC)C#N
HM04 or H0900
HM04 or H0900
Cas 1808913-24-7
(R)-3-(1-(2,3-dichloro-4-(pyrazin-2-yl)phenyl)-2,2,2-trifluoroethyl)-1-methyl-1-(1-methylpiperidin-4-yl) urea
The compound was disclosed in WO2015134839 . Helsinn under license from Novo Nordisk , is investigating ghrelin antagonists for treating obesity, Prader-Willi syndrome and other metabolic disorders; in May 2015, the program was listed as being in preclinical development
Helps reducing ghrelin signaling activity and treating disorder associated with an increase in ghrelin level (eg food abuse, alcohol addiction, and Prader-Willi syndrome).
Ghrelin, a growth hormone-releasing peptide produced by ghrelinergic cells in the gastrointestinal tract, is understood to function as a neuropeptide that regulates energy metabolism by stimulating appetite. The modulation, for example inhibition, of ghrelin signaling, through the ghrelin/growth hormone secretagogue receptor (GHS-Rla), is an attractive target for pharmacological treatment of disorders associated with high ghrelin level. Potential disorders for treatment using ghrelin modulators include food abuse (such as binge eating, obesity, hyperphagia (or uncontrollable appetite), post-dieting body weight rebound (including post-dieting hyperphagia), alcohol addiction, and genetic diseases associated with increased ghrelin level (e.g., Prader-Willi syndrome (PWS)).
PATENT
US 20150252021

PATENT
WO2015134839
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2015134839
Example 1
nthesis of Intermediate lk
Intermediate k
Step 1:
To a solution of la (100 g, 0.62 mol) in DMF (1.2 L) was added N-bromosuccinimide (110 g, 0.62 mol) at 0 °C. The mixture was stirred at room temperature for 4 h, then water (800 mL) was added and the resulting mixture was extracted with EtOAc (3 x 500 mL). The combined organic layers were dried over anhydrous Na2S04 and concentrated under reduced pressure. The residue was triturated with petroleum ether to provide lb (133.7 g, 89% yield) as a brown solid. !H-NMR (CDC13, 300 MHz): δ= 7.30 (d, 1 H), 6.59 (d, 1 H), 4.22 (br, 2 H). LC-MS: 241 [M+l]+.
Step 2:
To a solution of lb (133.7 g, 0.55 mol) in dry CH2C12 (1.5 L) was added acetic anhydride (110 g, 0.62 mol) dropwise over a period of 20 minutes at room temperature. The mixture was stirred at room temperature overnight, then diluted with CH2C12 (300 mL) and washed with water (150 mL) and brine (200 mL). The organic layer was separated, dried over anhydrous Na2SC>4 and concentrated under reduced pressure. The residue was triturated with petroleum ether (300 mL) to provide compound lc (143.0 g, 91% yield) as a white solid. ¾-NMR (CDC13, 400 MHz): δ= 8.26 (d, 1 H), 7.63 (br, 1 H), 7.54 (d, 1 H), 2.26 (s, 3 H). LC-MS: 280 [M-l]“.
Step 3:
A mixture of compound lc (50.0 g, 0.18 mol), butyl vinyl ether (Id, 89.0 g, 0.89 mol), bis(l,3-diphenylphosphino)propane (DPPP, 22.0 g, 0.053 mol), TEA (100 mL, 0.71 mol) and Pd(OAc)2 (6.4 g, 0.027 mol) in DMSO (1.2 L) was heated at 130 °C under N2 overnight. After the reaction was completed, the mixture was cooled to 0 °C and 2N HC1 (480 mL) was added dropwise over a period of 30 minutes. Then, the mixture was extracted with EtOAc (3 x 100 mL). The combined organic layers were dried over anhydrous a2S04 and concentrated under reduced pressure. The residue was purified by column chromatography (silica, EtOAc: PE=1 : 10) to provide le (19.5 g, 45% yield) as a yellow solid. 1H-NMR (CDC13, 400 MHz): 3= 8.46 (d, 1 H), 7.82 (br, 1 H), 7.51 (d, 1 H), 2.63 (s, 3 H), 2.29 (s, 3 H). LC-MS: 244 [M-l]“.
Step 4:
To a solution of le (21.9 g, 89.4 mmol) in MeOH (350 mL) was added 2N NaOH solution (350 mL) at room temperature. The mixture was heated at 50 °C overnight, then cooled and concentrated under reduced pressure. The resulting solid was triturated with water (100 mL) for 30 min and filtered to provide If (18.0 g, 98% yield) as a brown solid. ¾-NMR (CDC13, 400 MHz): 3= 7.48 (d, 1 H), 6.68 (d, 1 H), 4.56 (br, 2 H), 2.62 (s, 3 H). LC-MS: 202[M-1]\
Step 5:
To a mixture of compound If (18.0 g, 89.2 mmol) and ice (360 g) in cone. HC1 (180 mL) was added a solution of NaN02 (9.2 g, 133.7 mmol) in water (20 mL) dropwise over a period of 30 minutes, and the resulting mixture stirred in an ice bath for 30 min. A solution of KI (74.0 g, 446 mmol) in water (360 mL) was added dropwise over 45 min at 0 °C. The mixture was stirred for 30 min and then extracted with EtOAc (3 x 100 mL). The combined organic layers were dried over anhydrous Na2SC>4 and concentrated under reduced pressure. The residue was purified by column chromatography (silica, EtOAc: PE=1 :40) to provide lg (23.9 g, 86% yield) as a yellow solid. 1H-NMR (CDC13, 400 MHz): 3= 7.6 (d, 1 H), 7.06 (d, 1 H), 2.62 (s, 3 H).
Step 6:
To a solution of lg (23.9 g, 76.1 mmol) in MeOH (100 mL)/THF (100 mL) was slowly added NaB¾ (2.9 g, 76.1 mmol) at 0 °C. The mixture was stirred at room temperature for 5 min, and then quenched with water (100 mL). The mixture was extracted with EtOAc (3 x 100 mL). The combined organic layers were dried over anhydrous Na2SC>4 and concentrated under reduced pressure. The residue was purified by column chromatography (silica, EtOAc: PE=1 : 10) to provide lh (22.4 g, 93% yield) as a white solid. 1H-NMR (CDC13, 400 MHz): 3= 7.81 (d, 1 H), 7.26 (d, 1 H), 5.23 (q, 1 H), 2.17 (br, 1 H), 1.47 (d, 3 H).
Step 7:
To a mixture of lh (22.4 g, 70.9 mmol), phthalimide (12.5 g, 85.0 mmol) and PPh3 (22.3 g, 85.0 mmol) in dry THF (450 mL) was added DIAD (21.5 g, 106.3 mmol) at room temperature under N2 protection. The mixture was stirred at room temperature overnight and then concentrated under reduced pressure. The residue was purified by column chromatography (silica, EtOAc: PE=1 : 15) to provide li (18.5 g, 58% yield) as a white solid. 1H-NMR (CDC13, 400 MHz): 3= 7.78-7.84 (m, 3 H), 7.70-7.73 (m, 2 H), 7.41-7.43 (d, 1 H), 5.76-5.81 (q, 1 H), 1.84 (d, 3 H).
Step 8:
A solution of li (7.2 g, 16.2 mmol) and hydrazine hydrate (98%, 4.0 g, 80.9 mmol) in MeOH (150 mL) was heated under reflux for 2 h, then cooled and concentrated under reduced pressure. The residue was diluted with water (100 mL) and extracted with CH2C12 (3 x 100 mL). The combined organic layers were dried over anhydrous Na2SC>4 and concentrated under reduced pressure to give lj (3.8 g, 75% yield) as a white solid. 1H-NMR (CDC13, 400 MHz): 3= 7.81 (d, 1 H), 7.25 (d, 1 H), 4.55 (q, 1 H), 1.36-1.38 (d, 3 H). LC-MS: 316 [M+l]+.
Step 9:
To a solution of lj (41. Og, 0.13 mol) in methyl tert-butyl ether (750 mL) was added slowly a solution of D-mandelic acid (7.8 g, 0.052 mol) in methyl tert-butyl ether (1 10 mL) at 45°C. The mixture was stirred at this temperature for 30 min then cooled and filtered. White solid obtained was partitioned between 5% NaOH solution (300 mL) and methyl tert-butyl ether (300 mL). The bi -phases were separated and the aqueous phase was extracted with methyl tert-butyl ether (300 mL). The combined organic layer was concentrated to provide Intermediate lk (12 g, 58.5% yield) as a white solid (ee%=98.0%, Chiralpak AD-H, 5 μπι, 4.6*250mm, mobile phase: Hex: EtOH : DEA=80 : 20 : 0.2), retention time = 6.408 min).
Example 2
Synthesis of Compoun
A suspension of N-methyl-4-piperidone 2a (13.3 g, 58.6 mmol), NH2Me (30% in MeOH, 100 mL) and Pd/C (0.66 g) in MeOH (200 mL) was heated at 60 °C under H2 atmosphere (50 psi) overnight, then cooled and filtered. The filtrate was concentrated under reduced pressure and the residue was dissolved in HC1 in dioxane (3N, 100 mL) and stirred for 30 min. The precipitate was filtered and washed with EtOAc (50 mL) to provide 2b (7.7g, 54% yield) as white powder. 1H-NMR (DMSO, 400 MHz): δ= 9.50 (br, 2 H), 3.48 (d, 2 H), 3.15-3.16 (m, 1 H), 2.96-3.01 (m, 2 H), 2.70 (s, 3 H), 2.51 (s, 3 H), 2.22-2.28 (m, 2 H), 1.94-2.02 (m, 2 H), LC-MS: 129 [M+l]+ .
Example 20
Synthesis of H0900
Step 1:
To a mixture of 16d (32 g, 120 mmol) in dry CH2CI2 (800 mL) was added Dess-Martin peroxide reagent (76 g, 180 mmol) portion- wise at 0 °C. The mixture was stirred at room temperature for 1 h, then diluted with DCM (800 mL), washed with aqueous NaHC03 solution (300 mL) and brine (300 mL). The organic phase was separated, dried over anhydrous Na2S04 and
concentrated under reduced pressure to afford crude 18a (31.4 g) which was used directly in the next step without further purification.
Step 2:
To a solution of 18a (12 g, 40 mmol) and 3b (22.2 g, 60 mmol) in DME (560 mL) were added Pd(PPh3)4 (9.25 g, 8 mmol) and Cul (1.52 g, 8 mmol) at room temperature. The mixture was stirred at 90 °C overnight, then concentrated under reduced pressure. The residue was purified with silica gel column chromatography (silica, EA : PE = 1 :5) to provide 18b (8.0 g, 79.3%) as a white solid. LC-MS: 253 [M+l]+.
Step 3:
To a solution of 18b (7 g, 27.7 mmol) and (¾)-tert-butylsulfinamide (7.27 g, 30.56 mmol) in dry THF (200 mL) was added Ti(i-OPr)4 (15.7 g, 55.4 mmol) dropwise at room temperature. The mixture was stirred at 80 °C overnight, and then cooled. Ethyl acetate (40 mL) was added, the resulting mixture was filtered and the filtrate was concentrated under reduced pressure. The residue was purified with silica gel column chromatography (silica, EA:PE =1 :5) to provide 18c (6.8 g, 69%) as a yellow solid. 1H-NMR (CDC13, 400 MHz): 3= 9.10 (s, 1H), 8.97 (s, 1H), 8.72 (s, 1H), 8.64 (d, 1H),8.12 (d, 1H), 7.59 (d, 1H), 1.30 (s, 9H).LC-MS: 356 [M+l]+.
Step 4:
To a stirred solution of 18c (6.8 g, 19 mmol) and Tetrabutylammonium difluorotnphenylsilicate (15.8 g, 29 mmol) in dry THF (250 mL) was added a solution of TMSCF3 (11 g, 77 mmol) in anhydrous THF (50 mL) at -65 °C. The mixture was then stirred at -65 °C for 2 h, and at that point aqueous NH4CI solution (250 mL) was added. The mixture was diluted with ethyl acetate (250 mL), washed with brine (250 mL), dried over anhydrous Na2SC>4 and concentrated under reduced pressure. The residue was purified with silica gel column chromatography (silica, EA : PE=1 :2) to provide 18d (4.3 g, 52%) as a yellow solid. LC-MS: 426 [M+l]+.
Step 5:
To a stirred solution of 18d (4.3 g, 10.1 mmol) in MeOH (40 mL) was added a solution of HCl/MeOH (4N, 40 mL) at room temperature. The mixture was stirred for 1 h, then concentrated under reduced pressure. The residue was triturated with ethyl acetate (40 mL) to afford crude 18e (4.3g) which was directly in the next step without further purification. LC-MS: 322 [M+l]+.
Step 6:
To a solution of 18e (2.7 g, 7.1 mmol), 2b (3.4 g, 21.3 mmol) and TEA (80 mL) in DCM (220 mL) was added thiphosgene (3.15 g, 10.6 mmol) in DCM (40 mL) dropwise at 0 °C. The solution was warmed to ambient temperature and stirred for 1 h, then diluted with DCM ( 100 mL) and washed with aqueous Na2C03 solution (100 mL) and brine (100 mL). The organic layer was separated, dried over anhydrous Na2SC>4 and concentrated. The residue was purified with silica gel column chromatography (silica, DCM : CH3OH=10 : 1) to provide crude H0900 (2.13 g, ee%=92.5%) which was further purified through chiral separation to afford H0900 (1.6 g, 49% yield) as a white solid. (ee%=98.5%, Chiralpak IC 5um, 4.6*250mm, Phase: Hex: EtOH:
DEA=90: 10:0.2), retention tine =12.829 min. 1H-NMR (CDC13, 400 MHz): δ= 8.86 (d, 1H), 8.63 (dd, 1H), 8.55 (d, 1H), 7.47 (d, 1H), 7.40 (d, 1H), 6.28 (m, 1H), 5.18 (d, 1H), 4.12 (m, 1H), 2.88 (t, 2H), 2.77 (s, 3H), 2.22 (s, 3H), 2.05 (m, 2H), 2.48 (m, 2H), 1.52 (m, 2H), 1.73-1.49 (m, 4H). LC-MS: 476 [M+l]+.
PATENT
WO-2019118298
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2019118298&tab=PCTDESCRIPTION&maxRec=1000
Novel crystalline fumarate salt forms of (R)-3-(1-(2,3-dichloro-4-(pyrazin-2-yl)phenyl)-2,2,2-trifluoroethyl)-1-methyl-1-(1-methylpiperidin-4-yl) urea (also referred to as HM04 or H0900; designated as Forms 1-4), process for their preparation and compositions comprising them are claimed.
PWS occurs in approximately 1 in 10,000 births and is associated with deletion or lack of expression of region 15ql 1.2 of the paternal chromosome 15.
Characteristics of PWS include short stature, low muscle tone, and hyperphagia. Growth hormone replacement is frequently used to treat growth deficiencies and hypotonia. However, treatment for the insatiable appetite is lacking and PWS children can mature into adults suffering from obesity and type 2 diabetes. Levels of ghrelin are generally elevated in PWS; however, the relationship with ghrelin signaling and food intake in PWS remains unclear. See Purtell L., et ah, In adults with Prader-Willi syndrome, elevated ghrelin levels are more consistent with hyperphagia than high PYY and GLP-l levels. Neuropeptides. 201 l;45(4):30l-7; Cummings D.E., et ah, Elevated plasma ghrelin levels in Prader Willi syndrome. Nature Medicine . 2002;8(7):643-4; DelParigi A., et ah, High circulating ghrelin: a potential cause for hyperphagia and obesity in Prader-Willi syndrome. The Journal of Clinical Endocrinology and Metabolism. 2002;87(l2):546l-4.
[005] Accordingly, it is desirable to find treatments that effectively inhibit GHSRla, that are tolerable to the patient, and that do not interfere with other functions of the growth hormones. GHSRla modulators, including inhibitors such as (R)-3-(l-(2,3-dichloro-4-(pyrazin-2-yl)phenyl)-2, 2, 2-trifluoroethyl)-l -methyl- l-(l-m ethylpiperidin-4-yl) urea (HM04, H0900) depicted below, are reported in LT.S. Patent No. 9,546,157.
Step 1 : Synthesis of compound 2A
[00106] 2,2,6,6-tetramethylpiperidine (7.20 kg, 51.1 mol, 3.0 eq.,
KF=0.30%) was added into a 100 L reactor equipped with a temperature probe and overhead stirrer and mixed at RT under nitrogen protection. THF (50 L) was added into the reactor and stirred. The vessel was purged with nitrogen three times and cooled to 0 °C. n-BuLi (20.4 L, 3.0 eq.; 2.5 M hexane solution) was added to the mixture dropwise while keeping the temperature at about 0 °C to about 5 °C for over one hour. The color of the solution turned yellow. The mixture was stirred at about 0 °C to about 5 °C for 30 minutes. The mixture was cooled to about -78 °C to about -70 °C to form Solution A.
[00107] Compound 1 (3.25 kg, 17.0 mol. 1.0 eq., KF=0.03%) was dissolved in 15 L of THF to form Solution B.
[00108] Solution B was added to solution A dropwise at a temperature of about -70 °C to about -78 °C over one hour and then stirred for 30 minutes to form solution C. Tri-isopropyl borate ((i-PrO)3B) (3.52 kg, 18.7 mol., 1.1 eq.) was added dropwise into solution C over 10 minutes. The reaction mixture was stirred at a
temperature of about -70 °C to about -78 °C for one hour. HC1 (40 L, 3M, 7.0 eq.) was added over 30 minutes to quench the reaction. A 10 degree rise in temperature was noted.
[00109] The resulting aqueous layer was separated and extracted with EtOAc (40 L). The aqueous layer was separated and extracted twice again with EtOAc (35 L, 30 L). The organic layers were combined resulting in about 160 L of liquid. The combined organic layer was washed twice with 50 L of a 1M aqueous HC1 solution saturated with NaCl. The organic layer was concentrated to about 5 L in a 50 L rotavapor at a temperature of about 50 °C to about 55 °C under 30-40 mmHg for about 8 hours.
[00110] The residual EtOAc was swapped with DME for 3 times (10 L x 3). The organic layer was concentrated in the 50 L rotavapor at a temperature of about 50 °C to about 55 °C under 30-40 mmHg for about 6 hours. Each time about 5 L of residual remained. DME (20 L) was added to the residual to obtain a deep brown solution of 14.2% compound 2A (3.55 kg in 25 kg of solution; 88.8% yield; 97.4% purity (AETC by HPLC, retention time = 1.6 minutes); 0.24% residual ethyl acetate). 1H-NMR (400 MHz, DMSO): 5=8.55 (s, 2H), 7.36 (d, 1H), 7.69 (d, 1H). A second batch of compound 2A was prepared by the same method to produce 3.29 kg (95.4% purity, 82.3% yield, 0.11% residual ethyl acetate).
[00111] Step 2: Synthesis of Compound 3A
C! , N
M
K2CO3 (I .O equiv)
2A OH
DME/H20 3:1 (20 vol), 50 e C 3A N
[00112] Compound 2 A (2.91 kg in 20.5 kg solution) was added into a 100
L reactor at room temperature under nitrogen. DME (45 mL), 2-chloropyrazin (1.42 kg,
12.4 mol., 1.0 eq.), and Pd(dppf)Cl2 (10% w/w, 291 g) were added sequentially, and each
mixed at room temperature under nitrogen. Nitrogen was bubbled into the mixture for 20
minutes and the resulting mixture was purged and filled with nitrogen (3 times). The
mixture was heated to 48-52°C over 60 minutes. K2CO3 (2.57 kg, 18.6 mol, 1.5 eq.) was
added to 22 L of water in another reactor at room temperature and then added dropwise to
the compound 2 A mixture over 10 minutes. The mixture was stirred at 48-52°C for 16
hours and then cooled to room temperature. This procedure was repeated twice and all
three batches were combined.
[00113] An aqueous solution of K2CO3 (1.0 kg) was dissolved in 22 L of
water and added to the combined mixture to adjust the pH to 9. TBME (50 L) was added
into the mixture and filtered (PET filter, 3-5 pm, 205g/m2) to remove about 50 g of
sticky, brown solid material (catalyst analog). The aqueous layer was twice separated and
extracted with TBME (40 L, 40L).
[00114] The aqueous layer was combined with the aqueous layer of a
fourth batch prepared according to the above method. The pH of the combined aqueous layers was adjusted to pH<3 with HC1 (2N, 48 L). The solid precipitated out slowly as
the mixture was stirred at room temperature for 1 hour. The mixture was filtered (PET
filter, 3-5 pm, 205g/m2) over 30 minutes to obtain 20 kg of wet product. ACN (40 L) was
added into a 100 L reactor equipped with an overhead stirrer at room temperature. The 20
kg of wet product was added into the reactor and the reaction mixture heated to reflux
and stirred at reflux for 4 hours. The reaction mixture was cooled to room temperature
over 3 hours (around 15 °C/hour) and filtered to obtain 8.5 kg of wet solid. The wet solid
was dried under vacuum (20-30 mmHg) at 50-55 °C for 15 hours to obtain compound 3 A
as a pale white solid (6.1 kg; 97.4% purity (AUC by HPLC, retention time = 3.7
minutes); 83.8% yield). 1H-NMR (400 MHz, DMSO): 5=7.67 (d, 1H), 7.82 (d, 1H), 8.75
(d, 1H), 8.82 (t, 1H), 8.98 (d, 1H), 13.89 (bs, 1H).
[00115] Step 3: Synthesis of compound 6A
3A 6A N
N
[00116] Compound 3 A (6.1 kg, 22.7 mol, 1.0 eq.) was added into a 100 L
reactor equipped with a temperature probe, overhead stirrer, and condenser. Methanol
(92 L) was added into the reactor at room temperature. The mixture was cooled to
0-10 °C and added with SOCk (5.4 kg, 45.3 mol, 2.0 eq.) dropwise at 0-10 °C over 30
minutes. The reaction mixture was heated to reflux (65 °C) and stirred at reflux for 15
hours. A suspension was formed. Most of the solvent and SOCk was removed under
vacuum distillation until about 30 L remained. The mixture was concentrated under
vacuum (30-40 mmHg) at 50-55 °C for about 6 hours. Water (10 L) was added to the residual at -5 to 15 °C. The pH was adjusted to 8-9 with an aqueous solution of K2CO3 (200 g, dissolved in 2L of water) at -5 to 15 °C. The resulting aqueous layer was extracted twice with isopropyl acetate (25 L, 25 L). The combination of organic layers (about 50 kg) was washed with 20 L of NaHCCb aqueous layer. The organic layer was separated and washed with 10 L of of an aqueous solution of NaHCCb. All the aqueous layers were combined (55.8 kg). The organic layer was filtered through a silica pad (30 cm) and the pad washed with extra isopropyl acetate until the compound 6 A was filtered from the silica gel (about 3 hours). The organic layer was concentrated to about 5 L. THF (10 L) was added to the residual and concentrated to about 5 L (3 times) under vacuum (30-40 mmHg) at 50-55 °C for about 3 hours. Another 10 L of THF was added to the residual concentrate, giving a concentrated solution of compound 6A (15.8 kg; 32.83%,
5.19 kg compound 6A in solution; 97.9% purity (AUC by HPLC, retention time = 8.5 min); 80.8% yield). 1H-NMR (400 MHz, DMSO): 5=3.98 (s, 3H), 7.54 (d, 1H), 7.78 (d, 1H), 8.63 (d, 1H), 8.72 (t, 1H), 8.94 (d, 1H).
[00117] Step 4: Synthesis of compound 6B
[00118] THF (26 L) was added into a 100 L reactor equipped with a temperature probe and overhead stirrer under nitrogen. DIBAL-H (26 kg, 46 mol, 5.0 eq.) was added and the system purged and filled with nitrogen three times. The mixture was cooled to -78 to -70 °C to form solution A. A room temperature solution of compound 6A (2.6 kg, 9.2 mol, 1.0 eq.) in 52 L of THF was added dropwise at -78 to -70 °C over 30 minutes under nitrogen. The mixture was warmed to -30 °C over about 5-6 hours. The reaction mixture was stirred at -40 to -30 °C for 30 minutes. The mixture was slowly added to 42 L of 2N HCL over 1 hour reaching a maximum temperature of 35 °C. The mixture was extracted with 26 L of isopropyl acetate. The organic layer was separated and washed with 30 L of brine. This procedure was repeated and both batches of organic layer were combined and concentrated from about 100 L to about 5-10 L under vacuum.
A solid slowly formed during concentration. The mixture was cooled to 5-15 °C and stirred for 1 hour. The mixture was filtered (30-50 pm) over 30 minutes. The solid was dried under vacuum at 50 °C for 6 hours to obtain compound 6B as a brown solid (2.1 kg; 97.5% purity (AUC by HPLC, retention time = 8.6 min); 45.7% yield). 1H-NMR (400 MHz, DMSO): d = 4.65 (d, 2H), 5.68 (t, 1H), 7.62 (d, 1H), 7.68 (d, 1H), 8.72 (d, 1H),
8.80 (t, 1H), 8.94 (d, 1H).
[00119] Step 5: Synthesis of compound 7
[00120] DMSO (10 L) was added to a 50 L flask equipped with a temperature probe and overhead stirrer under nitrogen at room temperature. Compound 6B (2.05 kg, 8.04 mol, 1.0 eq.) was added under nitrogen at room temperature. Et3N (8 L) was added under nitrogen at RT and the mixture was then cooled to 15-20 °C.
SCb. pyridine (5.1 kg, 32.08 mol, 4.0 eq.) was dissolved into 10 L of DMSO at 5-15 °C in a separate flask and added to the mixture dropwise over 3.5 hours at about 20 °C. The reaction mixture was transferred to 70 L of ice-water. The suspension mixture was stirred at 0-10 °C for 1 hour and filtered (PET, 3-5 pm, 205 g/m2) by centrifuge over 1.5 hours to obtain compound 7 as a brown solid. The solid was dissolved in 35 L of DCM at room temperature. The resulting DCM layer was washed with 5 L of brine. The organic layer was separated and concentrated under vacuum at 40-45 °C to dryness to obtain compound 7 as a brown solid (2.33 kg; 96.3% purity (AEiC by HPLC, retention time = 9.2 minutes); 93.5% yield). 1H-NMR (400 MHz, DMSO): d = 7.67 (d, 1H), 7.99 (d, 1H), 8.67 (d, 1H), 8.75 (s, 1H), 8.99 (d, 1H), 10.56 (s, 1H).
[00121] Step 6: Synthesis of compound 8
[00122] THF (23 L) was added to a 50 L flask equipped with a temperature probe and overhead stirrer under nitrogen at room temperature. Compound 7 (2.3 kg, 9.1 mol, 1.0 eq.) and (S)-2-methylpropane-2-sulfmamide (1.21 kg, 10 mol, 1.1 eq.) were added sequentially to the flask under nitrogen. Ti(OEt)4 (6.22 kg, 27.3 mol, 3.0 eq.) was added dropwise to the flask over 1 hour at 30-35 °C under nitrogen. The system was purged with nitrogen three times and then the mixture was stirred at room temperature for 2 hours. Isopropyl acetate (40 L) was added to the reaction mixture. The entire reaction mixture was then charged to 20 L of brine while stirring slowly at RT. A lot of solid was formed and no heat release was observed. The solid (about 18 kg) was filtered using centrifuge, and then the solid was slurried with 20 L of isopropyl acetate again for 20 minutes, and filtered again, resulting is slightly less solid (17.3 kg). The filtrates were then combined and washed with 20 L of brine. The organic layer was separated and concentrated in a rotavapor under vacuum (30-40 mmHg) at 40-50 °C for about 4 hours to remove the solvents and obtain a brown oil (compound 8). The oil was dissolved in DMF to obtain a black solution (7.36 kg; 40.1%; 3.0 kg compound 8 in solution; 92.1% purity (AUC by HPLC, retention time = 9.7 minutes); >100% yield). 1H-NMR (400 MHz, CDCb): d = 1.30 (s, 9H), 7.59 (d, 1H), 8.11 (d, 1H), 8.64 (s, 1H), 8.73 (m, 1H), 8.97 (s, 1H), 9.10 (s, 1H).
[00123] Step 7: Synthesis of compound 11
O
S
10 s C
8
11 N
[00124] DMF (26 L, 10 v/w) was added to a 50 L flask equipped with a temperature probe and overhead stirrer under nitrogen at 15 °C. Compound 8 (7.3 kg of
DMF solution, containing 2.9 kg, 8.1 mol, 1.0 eq.) and TBAA (2.44 kg, 8.1 mol, 1.0 eq.) were added sequentially to the flask under nitrogen. The mixture was cooled to 0-10 °C.
TMSCF3 (2.88 kg, 20.3 mol, 2.5 eq.) was then added to the flask over 60 min at 0-10 °C.
The reaction mixture was stirred at 0-5 °C under nitrogen protection for 3 hours.
Isopropyl acetate (60 L) was added to the mixture, followed by the addition of 45 L of
NaHCCb under stirring at 5-25 °C. The organic layer was separated, washed three times with NaHC03 (30 L x 3), and concentrated from 60 kg to 2.5 kg of brown oil. The oil product was dissolved in 20 L of TBME and filtered through a pad of silica gel (about 40 cm high, 30 cm diameter) over 2 hours to obtain 2.14 kg of compound 1 1 in TBME solution. The solution was concentrated at 45-50 °C to dryness to obtain compound 1 1 as a black oil (1.85 kg; 85.2% purity (AETC by HPLC, retention time = 9.1 minutes, 9.6 minutes for diastereoisomer); 53.6% yield). 1H-NMR (400 MHz, CDCh): d = 1.33 (s, 9H), 3.82-3.85 (d, 1H), 5.61-5.66 (m, 1H), 7.53-7.60 (m, 2H), 8.63-8.64 (d, 1H), 8.71-8.72 (m, 1H), 8.95 (s, 1H).
[00125] Step 8: Synthesis of compound 12 (free base)
[00126] Compound 1 1 (1.8 kg, 4.23 mol, 1.0 eq., crude) was added to a 50 L reactor equipped with a temperature probe and overhead stirrer under nitrogen at 25 °C. Anhydrous MeOH (18 L) was added to dissolve compound 1 1. Then MeOH/HCl (18 L, 1 N) was added dropwise at 25-30 °C over 10 minutes and the mixture was stirred at 25-30 °C for 1 hour. Water (15 L) was added to the reaction and the mixture concentrated in a rotavapor under vacuum (30-40 mmHg) at 45-50 °C for about 4 hours to remove the solvent. The pH of the mixture was adjusted to 10 with 5 L of K2CO3 solution. 20 L of EtOAc was then added to the mixture and the organic layer was separated and the aqueous layer extracted twice with EtOAc (15 L x 2). The organic layers were combined and washed with 10 L of brine. The combined organic layers contained 996 g of
compound 12 in 40 kg of EtOAc solution (84% purity (AUC by HPLC, retention time =
2.8 minutes). The organic layers were concentrated in a rotavapor under vacuum (30-40 mmHg) at 45-50 °C for about 3 hours to a 7.5 kg volume of compound 12 in EtOAc solution (83% purity (AETC by HPLC, retention time = 2.7 minutes).
[00127] In a separate 50 L reactor equipped with a temperature probe and overhead stirrer, D-CSA was added (930 g, 4.0 mol, 1.0 eq. to 1.26 kg compound 12) and stirred at room temperature under nitrogen. EtOAc (10 L) and then the EtOAc solution of compound 12 (1.26 kg, 3.9 mol, 1.0 eq.) were each sequentially added to the reactor. The mixture was stirred at room temperature for 1 hour and slowly became a suspension. The mixture was filtered by centrifuge and washed with EtOAc to produce 2.3 kg of compound 12 as an off-white solid (96.0% purity).
[00128] The solid product, 20 L of EtOAc, and 10 L of 10% aqueous K2CO3 were added sequentially to a 50 L flask and stirred at room temperature until no solid remained (pH = 9-10). The organic layer was separated and the aqueous layer extracted twice with EtOAc (10 L x 2). The organic layers were combined (about 32 kg) and washed with 10 L of brine. The organic layer contained 716 g of compound 12 in
31.8 kg of solution.
[00129] The organic layer was concentrated under vacuum at 45-50 °C to about 8 L. Activated carbon (200 g) was added to the organic layer and the mixture stirred at 60-70 °C for 1 hour, cooled to room temperature, and filtered using a Buchner funnel and filter paper (pore size: 30-50 pm) over 30 minutes to remove the activated carbon. The mixture was concentrated in a rotavapor under vacuum (30-40 mmHg) at 45-50 °C for about 3 hours to yield 710 g of compound 12 as a yellow solid (99.4% purity). [00130] D-CSA (410 g, 1.77 mol, 1.0 eq. to 680 g compound 12), 3.4 L iPrOH, and 68 mL of water were added sequentially to a 10 L reactor equipped with a temperature probe and overhead stirrer and stirred at room temperature under nitrogen. The mixture was heated to reflux (84 °C) to form solution A after 1 hour. Compound 12 (680 g) was dissolved in 3.4 L of iPrOH and added into solution A for one partition. A clear solution was formed and the temperature decreased to 65 °C. The mixture was stirred at 65 °C for about 15 minutes after which a solid appeared. The mixture was cooled to 10 °C over 2 hours, stirred at 10 °C for an additional 30 minutes, and filtered through a Buchner funnel and filter paper (pore size: 30-50 pm) over 30 minutes to collect the 1.1 kg of white solid.
[00131] EtOAc (10 L), 1.1 kg of white solid product, and 5 L of 10% K2CO3 were added sequentially to a 20 L flask and mixed for 5 minutes. The solid dissolved (pH = 9-10). The EtOAc layer was separated and the aqueous layer extracted twice with EtOAc (5 L each). The organic layers were combined (about 20 L), washed with 5 L of brine, and concentrated in a rotavapor under vacuum (30-40 mmHg) at 45-55 °C for about 3 hours to remove most of the solution and until the residue weight reached 1 kg. Heptanes (1 L) was added to the mixture and stirred at room temperature for 30 minutes. The mixture was filtered using a Buchner funnel and filter paper (pore size: 30-50 pm) over 30 minutes to obtain 419 g of compound 12 base as a white solid (99.7% purity). The filtrate was concentrated to 135 g of compound 12 as a yellow solid (98.7% purity). 1H-NMR (400 MHz, CDCh): d = 1.85 (bs, 2H), 5.17 (m, 1H), 7.56 (d, 1H), 7.68 (d, 1H), 8.62 (d, 1H), 8.70-8.71 (m, 1H), 8.93 (s, 1H). Combined, the products resulted in a 40.7% yield of compound 12.
[00132] Step 9: Synthesis of compound 10
10A 10
[00133] Pd/C (40 g, 5% w/w) was added into a 10 L autoclave reactor at room temperature under nitrogen. THF (2 L), 2 L of methylamine (27%-30% alcoholic solution, 2.1 eq.), and 800 g of compound 10A (7 mol, 1.0 eq.) were sequentially added into the reactor. The system was purged with hydrogen three times. The mixture was stirred at hydrogen pressure (50 psi) at 70-75 °C overnight and was then filtered using a Biichner funnel and filter paper (pore size: 30-50 pm) over 10 minutes to remove the Pd/C. The filtrate was concentrated in a rotavapor under vacuum (30-40 mmHg) at 45-50 °C for about 3 hours to obtain 933 g of yellow oil. The mixture was distilled without a column at atmospheric pressure and the 140-170 °C portion was collected to obtain 763 g of compound 10 as a colorless oil (98.6% purity (AUC by HPLC, retention time = 4.8 minutes); 84.2% yield; 8000 ppm residual ethanol). A portion of the oil (563 g) was distilled using a 3 cm column at atmospheric pressure and the 140-170 °C portion was collected to obtain 510 g of compound 10 (75.8% yield; 134 ppm residual ethanol). 1H-NMR (400 MHz, CDCb): d = 0.82 (bs, 1H), 1.10-1.12 (q, 2H), 1.66 (d, 2H), 1.73-1.81 (t, 2H), 2.05 (s, 3H), 2.08-2.19 (m, 1H), 2.22 (s, 3H), 2.60 (d, 2H).
[00134] Step 10: Synthesis of HM04 fumarate salt
[00135] DCM (1L), 200 g CDI (1.23 mol, 2.0 eq.), and 35 g DABCO (0.31 mol, 0.5 eq.) were sequentially added into a 3 L reactor equipped with a temperature probe and overhead stirrer, and stirred at room temperature under nitrogen. The mixture was cooled to -10 to -5 °C. Compound 12 (200 g) was dissolved in 1 L of DCM and added into the mixture dropwise over 1 hour, followed by stirring for 16 hours at -10 to -5 °C. Compound 10 (159 g, 1.24 mol, 2.0 eq.) was added at -10 to 0 °C over 10 minutes. The mixture was then warmed to 0 to 5 °C and held for 2 hours. The mixture was concentrated under vacuum at 40-45 °C to about 1 L. HC1 (1 L of 1 N) was added to the residual and concentrated in a rotavapor under vacuum (30-40 mmHg) at 45-50 °C for about 2 hours to remove the DCM. Another 3 L of 1N HC1 was added to the residual and extracted three times with TBME (4 L, 2 L, 2 L). The aqueous layer was slowly adjusted to pH = 9-10 with 20% aqueous K2CO3 (about 1.5 L) and extracted with DCM (2 L x 3). The organic layers were combined (about 4 L) and washed three times with 0.25 N KH2PO4 (1.2 L x 3). The organic layer was washed with 2 L of brine to bring the pH to neutral and concentrated in a rotavapor under vacuum (30-40 mmHg) at 45-50 °C for about 2 hours to 450 g (335 mL). MTBE (1.5 L) was added to the residual and distilled until 500 mL of liquid was collected. This step was repeated four times with the addition of 500 mL of TBME and collection of 500 mL of distillate, with the exception that 330 mL of liquid was collected at the final distillation. About 1 to 1.2 L of residual remained in the flask. The residual was slowly cooled to room temperature and stirred at room temperature overnight. The mixture was filtered, washed twice with TBME (400 mL x 2), and dried to obtain 192 g of HM04 free base a light yellow solid (99.3% purity (AUC by HPLC, retention time = 11.0 minutes). The product on the wall was dissolved in DCM and concentrated under vacuum to obtain 22 g of HM04 free base as a brown sticky oil (97.6% purity). The filtrate was concentrated under vacuum to obtain 22.5 g of yellow solid (94.0% purity).
[00136] HM04 free base (187 g, 0.39 mol, 1.0 eq., 99.3% purity) and 1.9 L of ACN were sequentially added to a 3 L flask equipped with a temperature probe and overhead stirrer and stirred at 15 °C under nitrogen to obtain a light-yellow suspension. Fumaric acid (45.6 g, 0.39 mol, 1.0 eq.) was added to the flask and generated a white suspension after 1 minute. The reaction suspension was stirred overnight at room temperature, filtered (15-20 pm, ash<0.l5), washed twice with ACN (50 mL x 2), and dried under vacuum at 50 °C for 6 hours to obtain 207 g of HM04 fumarate salt as a light yellow solid (99.4% purity (AUC by HPLC, retention time = 11.1 minutes); 57.8% yield; 3100 ppm residual ACN). The filtrate was concentrated under vacuum to obtain 20.1 g of HM04 fumarate salt as a light yellow solid (97.3% purity).
[00137] A portion of the product (117 g) was further dried in a vacuum oven (20-40 mmHg) to lower the residual acetonitrile content. After drying at 60 °C for 6 hours, 15 hours, and 72 hours; and at 65 °C for 18 hours, the residual acetonitrile content was measured as 3100 ppm, 2570 ppm, 1300 ppm, and 256 ppm, respectively. After the drying process, 98 g of HM04 fumarate salt was isolated (99.4% purity (AUC by HPLC, retention time = 11.0 minutes); 1H-NMR (400 MHz, DMSO): d = 1.49-1.58 (m, 2H),
1.81-1.92 (m, 2H), 2.44-2.53 (m, 5H), 2.78 (s, 3H), 3.12 (m, 2H), 4.06-4.13 (m, 1H), 6.36-6.41 (m, 1H), 6.55 (s, 2H), 7.47 (d, 1H), 7.73 (d, 1H), 8.11 (d, 1H), 8.75 (d, 1H),
8.81-8.82 (m, 1H), 8.99 (d, 1H). The yield of 98g of HM04 fumarate salt isolated after drying the partial batch was extrapolated over the whole batch to calculate an
approximate yield of 48% for step 10.
[00138] XRPD analysis of HM04 fumarate salt products obtained after drying at 60 °C for 6 hours, 15 hours, and 72 hours; and at 65 °C for 18 hours was performed (see Figures 6-9, respectively). The XRPD profile showed that the HM04 fumarate salt product was consistent with Form 1.
Example 6. Streamlined Synthesis of HM04 Fumarate Salt Form 1
[00139] The overall yield of HM04 fumarate salt produced using Step 10 of Example 5 was calculated as approximately 48%. In order to increase the overall yield, a streamlined synthesis was investigated that eliminated the step of isolating HM04 free base. In particular, step 10 of the method of Example 5 shown in Figure 5 was changed. An overview of the streamlined synthesis beginning after step 9 of Example 5 is shown in Figure 10.
[00140] Streamlined HM04 Fumarate Salt Trial 1 : PCM (121.4 g). CPI (20.0 g, 123 mmol, 2 eq.) and DABCO (3.5 g, 31 mmol) were sequentially added into an inertized 1 L reactor. The mixture was cooled to -10 °C. Separately, a solution of DCM (132.5 g) and compound 12 (20.0 g, 62.1 mmol) were charged into a vessel and stirred until a solution was obtained. This solution was dropped into the 1 L reactor over 33 minutes by keeping the internal temperature at -10 to -5 °C. At the end of the addition, the vessel was rinsed with DCM (7.0 g), which was then added to the reaction mixture.
After stirring overnight (19 hours) and positive IPC, compound 10 (15.9 g, 124 mmol, 2 eq.) was added over 15 minutes and the vessel rinsed with DCM (9.0 g). After heating at 0 °C, 1 hour of stirring, positive IPC, and a further 1.5 hours of stirring, the mixture was heated at room temperature and charged with water (200.1 g). The aqueous layer was separated and the organic layer extracted twice with 1 N HC1 (201, 200 g). The combined aqueous layers containing the product were washed with TBME (148 g). After removal of the organic layer, the aqueous layer was charged with DCM (265.0 g) and 50% K2CO3 solution (about 240 ml) until reaching pH 9.61.
[00141] Meanwhile, a solution of KH2PO4 (8.2 g) in water (240 g) was prepared. The organic layer containing the product was charged with the KH2PO4 solution until reaching pH 7.12 (142.2 g). After separation of the aqueous layer, the organic layer was washed with water (200 g). After separation of the aqueous layer, the organic layer was evaporated at 50 °C. ACN (314.4 g) was added and the solvent distilled again at 70-75 °C under vacuum. ACN (235.8 g) was added and the solvent distilled again under vacuum. ACN (141.5 g) was added, the resulting solution polish filtered and the filter washed with ACN (16 g). After heating at 60 °C, fumaric acid (7.2 g, 62 mmol) was added to the solution, causing a white precipitate. After cooling to 20 °C over 1 hour, the suspension was filtered and washed twice with TBME (2 x 30 g). After drying on the filter with nitrogen flow, 70.7 g of wet raw product was obtained. This was slurried with TBME (177.0 g) for 1 hour, filtered, and washed with TBME (70 g). After drying on the filter under nitrogen flow, 33.0 g of wet product was obtained. Heating at 50 °C under vacuum afforded the dry product as a white powder of HM04 fumarate salt (21.1 g;
| Patent ID | Title | Submitted Date | Granted Date |
|---|---|---|---|
| US9926337 | SUBSTITUTED ASYMMETRIC UREAS AND MEDICAL USES THEREOF | 2016-12-02 | |
| US9546157 | p-Substituted Asymmetric Ureas and Medical Uses Thereof | 2015-03-06 | 2015-09-10 |
////////////HM04, H0900, Helsinn, Novo Nordisk, PRECLINICAL, obesity, Prader-Willi syndrome, ghrelin
CN(C1CCN(C)CC1)C(=O)N[C@H](c3ccc(c2cnccn2)c(Cl)c3Cl)C(F)(F)F
GFH 018
GFH-018
CAS 2169299-67-4
GenFleet Therapeutics
Advanced solid tumor; Cancer
TGF-beta Receptor Type-1 (TGFBR1; ALK5; SKR4; TbetaR-I) Inhibitors
Signal Transduction Modulators
GFH-018 , a TGFBR1 inhibitor, being investigated by GenFleet as an oral tablet formulation, for the treatment of cancer, including advanced solid tumors and hepatocellular carcinoma, in March 2019, the company was developing GFH-018 as a class 1 chemical drug in China, with a clinical trial expected to begin in the second half of 2019.


PATENT
WO2017215506
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2017215506
PATENT
WO-2019114792
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2019114792&tab=FULLTEXT&maxRec=1000
Novel crystalline and salt (hydrochloride, sulfate and mesylate) forms of a TGF-βRI inhibitor, designated as Forms A and B, processes for their preparation and compositions comprising them are claimed for treating cancers. The compound was originally claimed in WO2017215506 , assigned to Medshine Discovery Inc alone.
Step C: 1-9 (4.50 g, 15.20 mmol), 1-6 (4.43 g, 18.24 mmol), sodium carbonate (4.83 g, 45.60 mmol), [1,1′-bis (diphenyl) Phosphine) ferrocene] palladium dichloride (556.07 mg, 759.96 μmol), 2-biscyclohexylphosphine-2′, 6′-dimethoxybiphenyl (311.98 mg, 759.96 μmol) and [2-( 2-Aminophenyl)phenyl]-chloro-palladium-cyclohexyl-[2-(2,6-dimethoxyphenyl)phenyl]phosphine (547.64 mg, 759.96 μmol) was added to the dioxane (100 ml) and water (20 ml) in a mixed solvent. It was replaced with nitrogen three times and then heated to 90 to 100 ° C and stirred for 2 hours. The reaction mixture was poured into water (200 ml) and evaporated and evaporated. The combined organic layers were washed with EtOAc EtOAc m. The residue was purified on a silica gel column (eluent: methylene chloride/methanol, v/v=30/1) to afford crude crude product in petroleum ether/ethyl acetate (v/v=5/1) After stirring for 12 hours, the solid was collected by filtration, and the solid was concentrated and dried under reduced pressure to give 1-10. . 1 H NMR (400 MHz, CDCl3 . 3 ) [delta] 8.49 (S, IH), 7.82-7.74 (m, 2H), 7.59-7.46 (m, 4H), 6.99 (dd, J = 2.6,6.1Hz, IH), 4.39 (d, J = 6.3 Hz, 2H), 2.90 – 2.70 (m, 4H), 2.20 (s, 3H).
192 mg of the compound of formula (I) was weighed into a glass bottle. 10 ml of a tetrahydrofuran:acetic acid (v/v=9/1) mixed solvent was added, and after ultrasonic assisted for 30 minutes, the sample was dissolved into a clear solution. Stir on a magnetic stirrer (40 ° C). After 1.05 equivalents of p-toluenesulfonic acid monohydrate was slowly added, the sample was stirred overnight. After naturally cooling to room temperature, the supernatant was discarded by centrifugation, stirred for 10 hours by adding 10 ml of tetrahydrofuran, and the supernatant was discarded by centrifugation, and the same procedure was repeated twice more. The obtained solid was dried in a vacuum oven at 40 ° C for 1 hour, and after milling, it was further dried in a vacuum oven at 30 ° C for 16 hours to obtain a crystal form B of the compound of the formula (II).
.///////////////////GFH-018, GFH 018, GenFleet Therapeutics, Advanced solid tumor, Cancer, PRECLINICAL
NC(=O)/C=C/c4n5ncnc5ccc4c2c3CCCn3nc2c1cccc(C)n1

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










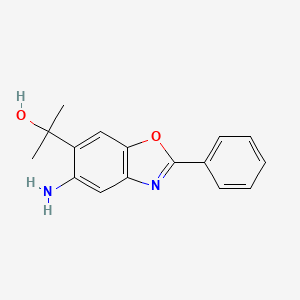





![3-[(1R)-1-(2,3-Dichloro-4-pyrazin-2-ylphenyl)-2,2,2-trifluoroethyl]-1-methyl-1-(1-methylpiperidin-4-yl)urea.png](https://pubchem.ncbi.nlm.nih.gov/image/imgsrv.fcgi?cid=91884613&t=l)
![(E)-3-[6-[2-(6-Methylpyridin-2-yl)-5,6-dihydro-4H-pyrrolo[1,2-b]pyrazol-3-yl]-[1,2,4]triazolo[1,5-a]pyridin-5-yl]prop-2-enamide.png](https://pubchem.ncbi.nlm.nih.gov/image/imgsrv.fcgi?cid=132246793&t=l)
