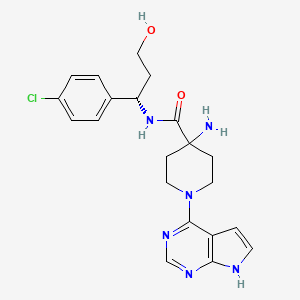
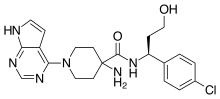
Capivasertib
C21H25ClN6O2
428.915
- 1143532-39-1
AZD 5363
4-amino-N-[(1S)-1-(4-chlorophenyl)-3-hydroxypropyl]-1-(7H-pyrrolo[2,3-d]pyrimidin-4-yl)piperidine-4-carboxamide
(S)-4-AMINO-N-(1-(4-CHLOROPHENYL)-3-HYDROXYPROPYL)-1-(7H-PYRROLO[2,3-D]PYRIMIDIN-4-YL)PIPERIDINE-4-CARBOXAMIDE
FDA APPROVED 11/16/2023, To treat breast cancer that meets certain disease criteria, Truqap
Capivasertib, sold under the brand name Truqap, is an anti-cancer medication used for the treatment of breast cancer.[1][2]
The most common adverse reactions include diarrhea, cutaneous adverse reactions, increased random glucose, decreased lymphocytes, decreased hemoglobin, increased fasting glucose, nausea, fatigue, decreased leukocytes, increased triglycerides, decreased neutrophils, increased creatinine, vomiting, and stomatitis.[3]
In November 2023, capivasertib was approved in the United States for people with hormone receptor-positive, human epidermal growth factor receptor 2-negative breast cancer when used in combination with fulvestrant.[3][4][5]
Capivasertib is a novel pyrrolopyrimidine derivative, and an orally available inhibitor of the serine/threonine protein kinase AKT (protein kinase B) with potential antineoplastic activity. Capivasertib binds to and inhibits all AKT isoforms. Inhibition of AKT prevents the phosphorylation of AKT substrates that mediate cellular processes, such as cell division, apoptosis, and glucose and fatty acid metabolism. A wide range of solid and hematological malignancies show dysregulated PI3K/AKT/mTOR signaling due to mutations in multiple signaling components. By targeting AKT, the key node in the PIK3/AKT signaling network, this agent may be used as monotherapy or combination therapy for a variety of human cancers.
Medical uses
Capivasertib, used in combination with fulvestrant (Faslodex), is indicated for adults with hormone receptor-positive, human epidermal growth factor receptor 2-negative locally advanced or metastatic breast cancer with one or more PIK3CA/AKT1/PTEN-alterations, as detected by an FDA-approved test, following progression on at least one endocrine-based regimen in the metastatic setting or recurrence on or within twelve months of completing adjuvant therapy.[1][3]
History
Efficacy was evaluated in CAPItello-291 (NCT04305496), a randomized, double-blind, placebo-controlled, multicenter trial in 708 participants with locally advanced or metastatic HR-positive, HER2-negative breast cancer, of which 289 participants had tumors with PIK3CA/AKT1/PTEN-alterations.[3] All participants were required to have progression on aromatase inhibitor-based treatment.[3] Participants could have received up to two prior lines of endocrine therapy and up to one line of chemotherapy for locally advanced or metastatic disease.[3]
PATENT
EXAMPLE 9: (S)-4-AMINO-N-(1-(4-CHLOROPHENYL)-3-HYDROXYPROPYL)-1-(7H-PYRROLO[2,3-D]PYRIMIDIN-4-YL)PIPERIDINE-4-CARBOXAMIDE (E9)
EXAMPLE 9 ALTERNATIVE ROUTE 1: (S)-4-AMINO-N-(1-(4-CHLOROPHENYL)-3-HYDROXYPROPYL)-1-(7H-PYRROLO[2,3-D]PYRIMIDIN-4-YL)PIPERIDINE-4-CARBOXAMIDE
EXAMPLE 9 ALTERNATIVE ROUTE 2: (S)-4-AMINO-N-(1-(4-CHLOROPHENYL)-3-HYDROXYPROPYL)-1-(7H-PYRROLO[2,3-D]PYRIMIDIN-4-YL)PIPERIDINE-4-CARBOXAMIDE


AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
//////////
| Clinical data | |
|---|---|
| Trade names | Truqap |
| Other names | AZD-5363 |
| AHFS/Drugs.com | Truqap |
| License data | US DailyMed: Capivasertib |
| Routes of administration | By mouth |
| Drug class | Threonine kinase inhibitor |
| ATC code | L01EX27 (WHO) |
| Legal status | |
| Legal status | US: ℞-only[1] |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 1143532-39-1 |
| PubChem CID | 25227436 |
| DrugBank | DB12218 |
| ChemSpider | 28189073 |
| UNII | WFR23M21IE |
| KEGG | D11371 |
| ChEMBL | ChEMBL2325741 |
| PDB ligand | 0XZ (PDBe, RCSB PDB) |
| CompTox Dashboard (EPA) | DTXSID40150710 |
| ECHA InfoCard | 100.208.066 |
| Chemical and physical data | |
| Formula | C21H25ClN6O2 |
| Molar mass | 428.92 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI | |
References
- ^ Jump up to:a b c “Truqap- capivasertib tablet, film coated”. DailyMed. 16 November 2023. Archived from the original on 20 November 2023. Retrieved 20 November 2023.
- ^ Turner NC, Oliveira M, Howell SJ, Dalenc F, Cortes J, Gomez Moreno HL, et al. (June 2023). “Capivasertib in Hormone Receptor–Positive Advanced Breast Cancer”. New England Journal of Medicine. 388 (22): 2058–2070. doi:10.1056/NEJMoa2214131. PMID 37256976. S2CID 259002400.
- ^ Jump up to:a b c d e f “FDA approves capivasertib with fulvestrant for breast cancer”. U.S. Food and Drug Administration. 16 November 2023. Archived from the original on 17 November 2023. Retrieved 17 November 2023.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ “Oncology (Cancer) / Hematologic Malignancies Approval Notifications”. U.S. Food and Drug Administration. 16 November 2023. Archived from the original on 17 November 2023. Retrieved 17 November 2023.
- ^ “Truqap (capivasertib) plus Faslodex approved in the US for patients with advanced HR-positive breast cancer”. AstraZeneca (Press release). 17 November 2023. Archived from the original on 17 November 2023. Retrieved 17 November 2023.
External links
- Clinical trial number NCT04305496 for “Capivasertib+Fulvestrant vs Placebo+Fulvestrant as Treatment for Locally Advanced (Inoperable) or Metastatic HR+/HER2- Breast Cancer (CAPItello-291)” at ClinicalTrials.gov
///////Capivasertib, Truqap, FDA 2023, APPROVALS 2023, AZD 5363
NC1(CCN(CC1)C1=C2C=CNC2=NC=N1)C(=O)N[C@@H](CCO)C1=CC=C(Cl)C=C1

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
