
1-(5-(2-fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl)-N-methylmethanamine fumarate
Vonoprazan Fumarate
(Takecab®) Approved
Vonoprazan Fumarate
CAS#: 1260141-27-2 (fumarate); 881681-00-1 (free base).
Chemical Formula: C21H20FN3O6S
Molecular Weight: 461.46
A potassium-competitive acid blocker (P-CAB) used to treat gastric ulcer, duodenal ulcer and reflux esophagitis.

Research Code TAK-438
CAS No. 881681-00-1
Cas 1260141-27-2(Vonoprazan Fumarate)
1-(5-(2-fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl)-N-methylmethanamine fumarate
| Molecular Weight | 461.46 |
| Formula | C17H16FN3O2S ● C4H4O4 |
| Company | Takeda Pharmaceutical Co. Ltd. |
| Description | Small molecule potassium-competitive acid blocker |
| Molecular Target | H+/K ATPase pump |
Vonoprazan (Takecab(®)) is an orally bioavailable potassium-competitive acid blocker (P-CAB) being developed by Takeda for the treatment and prevention of acid-related diseases. The drug is approved in Japan for the treatment of acid-related diseases, including erosive oesophagitis, gastric ulcer, duodenal ulcer, peptic ulcer, gastro-oesophageal reflux, reflux oesophagitis and Helicobacter pylori eradication. Phase III development is underway for the prevention of recurrence of duodenal and gastric ulcer in patients receiving aspirin or NSAID therapy. Phase I development was conducted in the UK for gastro-oesophageal reflux; however, no further development has been reported. This article summarizes the milestones in the development of vonoprazan leading to this first approval for acid-related diseases.
Vonoprazan Fumarate was approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on December 26, 2014. It was co-developed and marketed as Takecab® by Takeda & Otsuka.
Vonoprazan has a novel mechanism of action called potassium-competitive acid blockers (P-CABs) which competitively inhibits the binding the potassium ions to H+, K+-ATPase (also known as the proton pump) in the final step of gastric acid secretion in gastric parietal cells. Vonoprazan provides a strong and sustained acid section inhibitory effect. It is indicated for the treatment of gastric ulcer, duodenal ulcer and reflux esophagitis.
Cometriq® is available as tablet for oral use, containing 10 or 20 mg of free Vonoprazan, and the recommended dose is 20 mg orally once daily for adluts.
Vonoprazan fumarate (Takecab(®)) is a first-in-class potassium-competitive acid blocker that has been available in the market in Japan since February 2015. Vonoprazan is administered orally at 20 mg once daily for the treatment of gastroduodenal ulcer, at 20 and 10 mg once daily for the treatment and secondary prevention of reflux esophagitis, respectively, at 10 mg once daily for the secondary prevention of low-dose aspirin- or non-steroidal anti-inflammatory drug-induced peptic ulcer, and at 20 mg twice daily in combination with clarithromycin and amoxicillin for the eradication of Helicobacter pylori. It inhibits H(+),K(+)-ATPase activities in a reversible and potassium-competitive manner with a potency of inhibition approximately 350 times higher than the proton pump inhibitor, lansoprazole. Vonoprazan is absorbed rapidly and reaches maximum plasma concentration at 1.5-2.0 h after oral administration. Food has minimal effect on its intestinal absorption. Oral bioavailability in humans remains unknown. The plasma protein binding of vonoprazan is 80 % in healthy subjects. It distributes extensively into tissues with a mean apparent volume of distribution of 1050 L. Being a base with pKa of 9.6 and with acid-resistant properties, vonoprazan is highly concentrated in the acidic canaliculi of the gastric parietal cells and elicited an acid suppression effect for longer than 24 h after the administration of 20 mg. The mean apparent terminal half-life of the drug is approximately 7.7 h in healthy adults. Vonoprazan is metabolized to inactive metabolites mainly by cytochrome P450 (CYP)3A4 and to some extent by CYP2B6, CYP2C19, CYP2D6, and SULT2A1. A mass balance study showed that 59 and 8 % of the orally administered radioactivity was recovered in urine as metabolites and in an unchanged form, respectively, indicating extensive metabolism. Genetic polymorphism of CYP2C19 may influence drug exposure but only to a clinically insignificant extent (15-29 %), according to the population pharmacokinetic study performed in Japanese patients. When vonoprazan was co-administered with clarithromycin, the mean AUC from time 0 to time of the next dose (dosing interval) of vonoprazan and clarithromycin were increased by 1.8 and 1.5 times, respectively, compared with the corresponding control values, indicating mutual metabolic inhibition. The mean area under the curve from time zero to infinity obtained from patients with severe liver and renal dysfunction were elevated by 2.6 and 2.4 times, respectively, compared with healthy subjects, with no significant changes in plasma protein binding. Vonoprazan increases intragastric pH above 4.0 as early as 4 h after an oral dose of 20 mg, and the extensive anti-secretory effect is maintained up to 24 h post-dose. During repeated dosing of 20 mg once daily, the 24-h intragastric pH >4 holding time ratios were 63 and 83 % on days 1 and 7, respectively. Because vonoprazan elicited a more extensive gastric acid suppression than the proton pump inhibitor, lansoprazole, it also gave rise to two to three times greater serum gastrin concentrations as compared with lansoprazole. In pre-approval clinical studies for the treatment of acid-related disorders, mild to moderate adverse drug reactions (mostly constipation or diarrhea) occurred at frequencies of 8-17 %. Neither severe liver toxicity nor neuroendocrine tumor has been reported in patients receiving vonoprazan.
Vonoprazan fumarate is a first-in-class potassium-competitive acid blocker. It was approved in the Japanese market in February, 2015.[1]
Vonoprazan can be used for the treatment of gastroduodenal ulcer, reflux esophagitis, and for some drug-induced peptic ulcers. It can be combined with other antibiotics for the eradication of Helicobacter pylori.[2]
PATENT


Reference:1. WO2006036024A1 / US8048909B2.
2. WO2007026916A1 / US7498337B2.
3. CN104327051A.
1- [5- (2-fluorophenyl) -1- (pyridin-3-ylsulfonyl) -IH- pyrrol-3-yl] -N- methylmethanamine fumarate Takeda single An R & D for the gastric acid secretion inhibitors (codename: TAK-438, generic name: vonoprazan fumarate), the drug belongs to the potassium ion (K +) competitive acid blocker (P-CAB) for a new inhibitors, with a strong, long-lasting inhibition of gastric acid secretion, while the gastric parietal cells in the final stage of gastric acid secretion by inhibiting K + for H +, K + -ATP enzyme (proton pump) binding effect on gastric acid secretion also advance termination action.
Its molecular formula is: C17H16FN3O2S · C4H4O4, MW: 461.46, the chemical structure of formula I as shown.
CN101300229A discloses 1- [5- (2_-fluorophenyl) -1- (pyridin-3-ylsulfonyl) -1Η- pyrrol -3-yl] -N- methylmethanamine fumarate alone, but not related to its crystalline form.
The present invention discloses a I- [5- (2- fluorophenyl) -I- (pyridin-3-ylsulfonyl) -IH- pyrrol-3-yl] -N- methylmethanamine single rich fumarate A method for preparing a crystalline form. 1- [5- (2_-fluorophenyl) -1- (Batch-3-ylsulfonyl) -IH- pyrrol-3-yl] -N- methylmethanamine fumarate single crystalline form A, according to prepared by the following routes:
Example 1
A method of preparing polymorph having pyrrole derivatives maleate described in detail below.
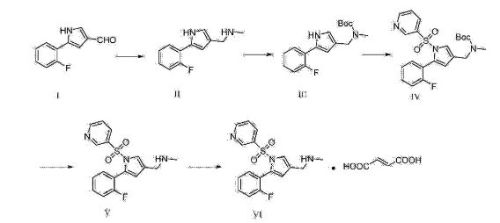
Step I: 5- (2- fluorophenyl) -1- (pyridin-3-ylsulfonyl) -IH- pyrrole-3-carbaldehyde Synthesis of
Compound II (260mg) was dissolved in tetrahydrofuran (50ml) was added 60% NaH, the reaction was stirred for 30 minutes at room temperature. Was added 15-crown–5 (I. 5g), the reaction mixture was stirred at room temperature for 1 hour and then pyridine-3-sulfonyl chloride was added, stirred at room temperature for 2 hours until complete reaction was followed by thin layer chromatography, and then was added to the reaction system 20mL saturated brine with ethyl acetate (IOOmLX2) and the combined organic phase was washed with saturated brine 50ml organic phase, an appropriate amount of anhydrous sodium sulfate, filtered, and the filtrate was concentrated under reduced pressure to give the crude compound IV (200mg) administered directly in the next reaction.
Synthesis 1_ [5- (2-fluorophenyl) -1- (piperidin-3-sulfonyl batch) -IH- pyrrol-3-yl] -N- methylmethanamine of: Step 2
The brown residue obtained in the previous step IV compound (200mg) was dissolved in 30mL methanol was added 27% -33% methyl amine solution, the reaction was stirred for 1.5 hours. Sodium borohydride (68mg), the reaction was stirred for 20 minutes, was added lmol / LHCl to an acidic aqueous solution, and stirred until complete reaction was followed by thin layer chromatography. To the reaction mixture was added saturated sodium bicarbonate solution until weakly basic system was extracted with ethyl acetate (IOOmLX2), the combined organic phases with saturated brine (50mL), dried over anhydrous Na2SO4, filtered and concentrated to give the crude product ( 208mg, yellow oil). Yield: 100%.
Step 3: 1_ [5- (2-fluorophenyl) -1- (pyridin-3-ylsulfonyl) -IH- pyrrol-3-yl] -N- methylmethanamine fumarate single synthesis
Compound V obtained in the previous step was dissolved in 20mL of ethyl acetate, taking the mass fraction of equivalents of fumaric acid was dissolved in 2ml of methanol. Added dropwise with stirring to a solution of compound V in ethyl acetate, stirred for 30 minutes at room temperature. Then warmed to 55-65 degrees reflux one hour, cooled to room temperature and filtered to give an off-white solid was washed with cold ethyl acetate IOml and dried in vacuo to give 170mg of crystalline Compound I, about 20% overall yield. X- ray diffraction spectrum of the crystalline sample is shown in Figure 1. DSC spectrum shown in Figure 2, this polymorph is defined as A crystalline form.
Reference:1. CN105085484A.
http://www.google.com/patents/CN105085484A?cl=en
Fumaric Wonuo La Like (TAK-438, Vonoprazan fumarate) is Takeda Pharmaceuticals and Otsuka Pharmaceutical to launch a new type of oral anti-acid drugs. As a potassium ion (K +) competitive acid blocker (P-CAB), Wonuo La Like gastric acid secretion in the gastric parietal cells play a role in the final step, by inhibiting K + for H +, K + -ATP enzyme (proton chestnut) combine to inhibit gastric acid secretion and early termination. Compared to the current power of the proton chestnut inhibitors (PPIs), due to the absence of praise Wonuo La CYP2C19 metabolism, so the performance in clinical trials showing good effect: the treatment of gastric ulcer / duodenal ulcer, reflux esophagitis eradication of H. pylori and other effects are better than lansoprazole, while having a similar security.
fumarate Wonuo La Like chemical name: I- [5_ (2_ gas) -1- (pyridin _3_ cross-acyl group) -IH- P ratio slightly 3-yl] – N- methylmethanamine fumarate, structured as follows:
Preparation of fumaric Wonuo La Like synthetic route mainly follows:
Takeda patent CN200680040789 original study discloses a 5- (2-fluorophenyl) -lH- pyrrole-3-carbaldehyde as a starting material, the solvent is tetrahydrofuran, sodium hydride doing acid binding agent, crown ethers do a phase transfer catalyst, with 3-pyridine sulfonyl chloride to give the intermediate 5- (2-fluorophenyl) -1- (pyridin-3-ylsulfonyl) -IH- pyrrole-3-carbaldehyde, then to form a Schiff base with methylamine boron sodium hydride reduction to give Wonuo La Like the free base and then fumaric acid salt formation, generate fumaric Wonuo La Chan, the reaction equation is as follows:
Takeda company disclosed in 2010 it 0 01,080,018,114 in improved synthetic route: Intermediate 5- (2-fluorophenyl) -I- (pyridine-3-ylsulfonyl) -IH–3 formaldehyde synthesis, instead of acetonitrile as solvent, DIEA do acid-binding agent, DMAP as catalyst, but side reactions, tedious post-processing operation, the lower the yield, the overall yield of less than 40%.
CN201080018114 improved synthetic route to 5- (2-fluorophenyl) -IH- pyrrole-3-carbonitrile as a starting material of the synthesis route, but this route is converted to the cyano aldehyde used Raney catalytic hydrogenation, industrial scale there is a big security risk, its reaction equation is as follows:
Y. Arikawa et J. Med Chem 2012, 55, 4446-4456 reported the following synthetic route.:
In phenyl pyrrole-3-carbaldehyde and methylamine alcohol imine by metal borohydride reduction, to give further protection to give Boc ((5-phenyl -IH- pyrrol-3-yl) -N -) methyl carbamate; the above product with an arylsulfonyl chloride, and then de-Boc protection to give 1- (5-phenyl-1 aromatic sulfonyl -IH- pyrrol-3-yl) – N- methyl methylamine;
Y. Arikawa et al reported that the above process step is prolonged, the probability g [J reacting a corresponding increase in the above reaction scheme conditional optimization, control side reactions is one of the present invention is to solve the problem. On the other hand the above literature after the synthesis process used in chromatography, is not conducive to fumaric Wonuo La Like industrial production. Therefore, the development of fumaric acid Wonuo La Like New synthesis process, simplify the synthesis operations, reduce costs, improve productivity, it has important implications for fumaric Wonuo La Like this one which attract anti-acid drugs.
PAPER
J. Med Chem 2012, 55, 4446-4456
http://pubs.acs.org/doi/abs/10.1021/jm300318t
Discovery of a Novel Pyrrole Derivative 1-[5-(2-Fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl]-N-methylmethanamine Fumarate (TAK-438) as a Potassium-Competitive Acid Blocker (P-CAB)

In our pursuit of developing a novel and potent potassium-competitive acid blocker (P-CAB), we synthesized pyrrole derivatives focusing on compounds with low log D and high ligand-lipophilicity efficiency (LLE) values. Among the compounds synthesized, the compound 13e exhibited potent H+,K+-ATPase inhibitory activity and potent gastric acid secretion inhibitory action in vivo. Its maximum efficacy was more potent and its duration of action was much longer than those of proton pump inhibitors (PPIs). Therefore, compound 13e (1-[5-(2-fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl]-N-methylmethanamine fumarate, TAK-438) was selected as a drug candidate for the treatment of gastroesophageal reflux disease (GERD), peptic ulcer, and other acid-related diseases.
SYNTHESIS
References
References
1: Arikawa Y, Nishida H, Kurasawa O, Hasuoka A, Hirase K, Inatomi N, Hori Y, Matsukawa J, Imanishi A, Kondo M, Tarui N, Hamada T, Takagi T, Takeuchi T, Kajino M. Discovery of a novel pyrrole derivative 1-[5-(2-fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl]-N-methylmethanamin e fumarate (TAK-438) as a potassium-competitive acid blocker (P-CAB). J Med Chem. 2012 May 10;55(9):4446-56. doi: 10.1021/jm300318t. Epub 2012 Apr 30. PubMed PMID: 22512618.
2: Kondo M, Kawamoto M, Hasuoka A, Kajino M, Inatomi N, Tarui N. High-throughput screening of potassium-competitive acid blockers. J Biomol Screen. 2012 Feb;17(2):177-82. doi: 10.1177/1087057111421004. Epub 2011 Sep 22. PubMed PMID: 21940711.
3: Shin JM, Inatomi N, Munson K, Strugatsky D, Tokhtaeva E, Vagin O, Sachs G. Characterization of a novel potassium-competitive acid blocker of the gastric H,K-ATPase, 1-[5-(2-fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl]-N-methylmethanamin e monofumarate (TAK-438). J Pharmacol Exp Ther. 2011 Nov;339(2):412-20. doi: 10.1124/jpet.111.185314. Epub 2011 Aug 9. PubMed PMID: 21828261; PubMed Central PMCID: PMC3199995.
4: Hori Y, Matsukawa J, Takeuchi T, Nishida H, Kajino M, Inatomi N. A study comparing the antisecretory effect of TAK-438, a novel potassium-competitive acid blocker, with lansoprazole in animals. J Pharmacol Exp Ther. 2011 Jun;337(3):797-804. doi: 10.1124/jpet.111.179556. Epub 2011 Mar 16. PubMed PMID: 21411494.
5: Matsukawa J, Hori Y, Nishida H, Kajino M, Inatomi N. A comparative study on the modes of action of TAK-438, a novel potassium-competitive acid blocker, and lansoprazole in primary cultured rabbit gastric glands. Biochem Pharmacol. 2011 May 1;81(9):1145-51. doi: 10.1016/j.bcp.2011.02.009. Epub 2011 Mar 1. PubMed PMID: 21371447.
6: Hori Y, Imanishi A, Matsukawa J, Tsukimi Y, Nishida H, Arikawa Y, Hirase K, Kajino M, Inatomi N. 1-[5-(2-Fluorophenyl)-1-(pyridin-3-ylsulfonyl)-1H-pyrrol-3-yl]-N-methylmethanamin e monofumarate (TAK-438), a novel and potent potassium-competitive acid blocker for the treatment of acid-related diseases. J Pharmacol Exp Ther. 2010 Oct;335(1):231-8. doi: 10.1124/jpet.110.170274. Epub 2010 Jul 12. PubMed PMID: 20624992.
- 1 “Vonoprazan: first global approval. – PubMed – NCBI”. Ncbi.nlm.nih.gov. 2015-09-28. Retrieved 2016-03-30.
- 2
“The First-in-Class Potassium-Competitive Acid Blocker, Vonoprazan Fumarate: Pharmacokinetic and Pharmacodynamic Considerations. – PubMed – NCBI”. Ncbi.nlm.nih.gov. 2015-09-28. Retrieved 2016-03-30.
/////

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....











