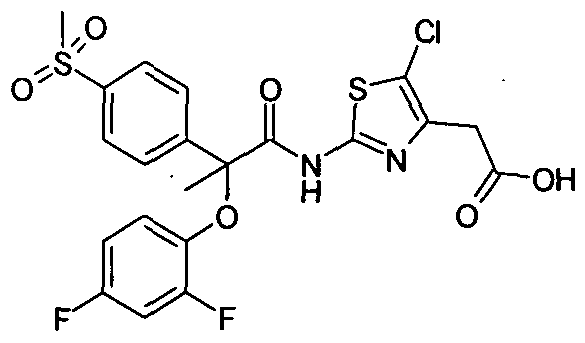
GKM 001……Several probables
Watch out on this post as I get to correct structure………..![]()
![]()
![]()
![]()
![]()
![]()
![]()





Advinus Therapeutics Private L,

A glucokinase activator for treatment of type II diabetes
In October 2012, Takeda and Advinus have entered into an agreement to initiate a three-year discovery collaboration program focused on novel targets for inflammation, CNS, and metabolic diseases.
| Company | Advinus Therapeutics Ltd. |
| Description | Activator of glucokinase (GCK; GK) |
| Molecular Target | Glucokinase (GCK) (GK) |
| Mechanism of Action | Glucokinase activator |
| Therapeutic Modality | Small molecule |
| Latest Stage of Development | Phase I/II |
| Standard Indication | Diabetes |
| Indication Details | Treat Type II diabetes |

Advinus chief executive officer/MD Dr. Rashmi Barbhaiya.

PATENT
https://www.google.co.in/patents/WO2009047798A2?cl=en
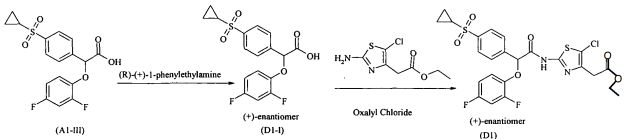
Example Cl : (-)-{5-ChIoro-2-[2-(4-cyclopropanesulfonylphenyI)-2-(2,4- difluorophenoxy)acetylamino]thiazol-4-yl}-acetic acid, ethyl ester
Step I: Preparation of (-)-(4-Cyclopropanesulfonylphenyl)-(2,4- difluorophenoxy)acetic acid (Cl-I):
To a solution of (4-cyclopropanesulfonylphenyl)-(2,4-difluorophenoxy)acetic acid (obtained in example Al -step III) in ethyl acetate was added (S)-(-)-l-phenylethylamine drop wise at -15 °C. After completion of addition the reaction was stirred for 4-6 hours. Solid was filtered and washed with ethyl acetate. The solid was then taken in IN HCl and extracted with ethyl acetate, ethyl acetate layer was washed with brine, dried over anhydrous sodium sulfate. Solvent was removed under reduced pressure to obtain (-)-(4- cyclopropanesulfonylphenyl)-(2,4-difluorophenoxy)acetic acid. Enantiomeric enrichment was done by repeating the diasteriomeric crystallization. [α]23 589 = – 107.1 ° (c = 2%Chloroform) Enantiomeric purity > 99. % (chiral HPLC)
Step II: (-)-{5-Chloro-2-[2-(4-cyclopropanesulfonylphenyl)-2-(2,4- difluorophenoxy)acetyIamino]thiazol-4-yl}-acetic acid ethyl ester : To a solution of (-)-4-cyclopropanesulfonylphenyl)-(2,4-difluorophenoxy)acetic acid (Cl-I) in DCM, was added DMF and cooled to 0 °C, followed by the addition of oxalyl chloride under stirring. Stirring was continued for 1 hour at the same temperature. The resulting mixture was further cooled to -35 °C, and to that, a solution of excess (2- amino-5-chlorothiazol-4-yl)acetic acid ethyl ester in DCM was added drop wise. After completion of reaction, the reaction mixture was poured into IN aqueous HCl under stirring, organic layer was washed with IN HCl, followed by 5% brine, dried over anhydrous sodium sulfate, solvent was removed under reduced pressure to get the crude compound which was purified by preparative TLC to get the title compound. [α]23 589 = – ve (c = 2%Chloroform)
1H NMR(400 MHz, CDCl3): δ 1.06-1.08 (m, 2H), 1.30 (t, J=7.2 Hz, 3H), 1.33-1.38 (m, 2H), 2.42-2.50 (m, IH), 3.73 (d, J=2 Hz, 2H), 4.22 (q, J=7.2 Hz ,2H), 5.75 (s, IH), 6.76- 6.77 (m, IH), 6.83-6.86 (m, IH), 6.90-6.98 (m, IH), 7.73 (d, J=8.4 Hz, 2H), 7.96 (d, J=8.4 Hz, 2H), 9.96 (bs, IH). MS (EI) m/z: 571.1 and 573.1 (M+ 1; for 35Cl and 37Cl respectively).
Examples C2 and C3 were prepared in analogues manner of example (Cl) from the appropriate chiral intermediate:
Example Dl : (+)-{5-Chloro-2-[2-(4-cyclopropanesulfonylphenyl)-2-(2,4- difluorophenoxy)acetylamino]thiazol-4-yl}acetic acid, ethyl ester
Preparation of (+)-(4-Cyclopropanesulfonylphenyl)-(2,4-difluorophenoxy)acetic acid (Dl-I):
To a solution of (4-cyclopropanesulfonylphenyl)-(2,4-difluorophenoxy)acetic acid (obtained in example Al -step III) in ethyl acetate, was added (R) (+)-l- phenylethylamine drop wise at -15 °C. After completion of addition the reaction was stirred for 4-6 hours. Solid was filtered and washed with ethyl acetate. The solid was then taken in IN HCl and extracted with ethyl acetate, ethyl acetate layer was washed with brine, dried over anhydrous sodium sulfate. Solvent was removed under reduced pressure to obtain (+)-(4-Cyclopropanesulfonylphenyl)-(2,4-difluorophenoxy)acetic acid. Enantiomeric enrichment was done by repeating the diasteriomeric crystallization. [α]23 589 = +93.07° (c = 2%Chloroform) Enantiomeric purity > 99. % (by chiral HPLC)
(+)-(4-CyclopropanesuIfonylphenyI)-(2,4-difluorophenoxy)acetic acid ethyl ester (Dl)
The example Dl was prepared using (+)-4-cyclopropanesulfonylphenyl)-(2,4- difluorophenoxy)acetic acid (Dl-I), and following the same reaction condition for amide coupling as described in example Cl, [ot]23 589 = + ve (c = 2%Chloroform)
PATENT
https://www.google.co.in/patents/WO2008104994A2?cl=en
Synthesis Type-P
Example Pl : {5-Chloro-2-[2-(2,4-difluoro-phenoxy)-2-(4-methanesulfonyl-phenyl)- propionylamino]-thiazol-4-yI}-acetic acid
To a solution of {5-Chloro-2-[2-(2,4-difluoro-phenoxy)-2-(4-methanesulfonyl- phenyl)-propionylamino]-thiazol-4-yl}-acetic acid methyl ester (0.03 g, 0.05 mmol) in THF: Ethanol: water ( ImI + 0.3ml + 0.3 ml) was added lithium hydroxide (0.0046 g, 0.11 mmol). The resulting mixture was stirred for 5 hours at room temperature followed by removal of solvent under reduced pressure. The residue was suspended in water (15 ml), extracted with ethyl acetate to remove impurities. The aqueous layer was acidified with IN HCl (0.5 ml) and extracted with ethyl acetate (2×10 ml), This ethyl acetate layer was washed with water (15 ml), brine (20 ml), dried over anhydrous sodium sulfate and solvent was removed under reduced pressure to give solid product {5-Chloro-2-[2-(2,4-difluoro-phenoxy)-2-(4- methanesulfonyl-phenyl)-propionylamino]-thiazol-4-yl} -acetic acid (9 mg). 1H NMR (400 MHz, CDCl3): δ 1.85 (s, 3H) , 3.07 (s, 3H) , 3.72 ( s, 2H), 6.64-6.69 ( m, 2H ) , 6.89-6.91 (m, IH ), 7.84 ( d, J – 8.4 Hz, 2H), 8.00 ( d, J = 8.8 Hz, 2H). MS (EI) mlz: 530.70 (M + 1), mp: 109-111 0C.
Preparation of {5-Chloro-2-[2-(2,4-difluoro-phenoxy)-2-(4-methanesulfonyl-phenyl)- propionylamino)-thiazol-4-yl}-acetic acid methyl ester used in Example Pl:
To a mixture of 2-(2, 4-Difluoro-phenoxy)-2-(4-methanesulfonyl-phenyl)-propionic acid (0.110 g, 0.22 mmol), (2-Amino-5-chloro-thiazol-4-yl)-acetic acid methyl ester (0.071 g, 0.32 mmol), HOBt (0.052g, 0.38 mmol), and EDCI (0.074 g, 0.38 mmol) in methylene dichloride (10 ml) was added N-methylmorpholine (0.039 g, 0.38 mmol). The resulting mixture was stirred at room temperature for overnight followed by dilution with 10 ml methylene dichloride. The reaction mixture was poured onto water (20 ml), and organic layer separated, washed with water (2x 20 ml), brine (20 ml), dried over sodium sulfate and solvent evaporated to get residue which was purified by preparative TLC using 50% ethyl acetate in hexane as mobile. To give desired compound (0.30 g). 1H NMR (400 MHz, CDCl3): δ 1.45 (t, J = 7.2 Hz, 3H), 1.93 (s, 3H), 3.14 (s, 3H), 3.77 (d, J = 2.8 Hz, IH), 4.26 (q, J = 7.2 Hz, IH), 6.69-6.77(m, 2H), 6.96-7.02 (m, IH), 7.89 (d, J = 8.4 Hz, 2H), 8.07 (d, J= 8.4Hz, IH).; MS (EI) m/z: 559 .00 (M + 1).
PATENT
http://www.google.com/patents/WO2012020357A1?cl=en
 4-{2-[2-Cyclopentyloxy-2-(4-cyclopropanesulfonyl-phenyl)-acetylamino]- thiazol-5-yloxy}-benzoic acid, cas 1359151-08-8
4-{2-[2-Cyclopentyloxy-2-(4-cyclopropanesulfonyl-phenyl)-acetylamino]- thiazol-5-yloxy}-benzoic acid, cas 1359151-08-8
Step I: (4-Cyclopropylsulfanyl-phenyl)-oxo-acetic acid ethyl ester:
A1C13 (7.98 g, 48.42 mmole) was suspended in DCM (50 mL) and cooled to 0 C under argon atmosphere. To this suspension was added chlorooxo ethylacetate (4.5 mL, 39.98 mmol) at 0 °C and stirred for 45 min. followed by addition of a solution of cyclopropylsulfanyl-benzene (5 g, 33.28 mmol) in DCM (10 mL) and stirred at 25 °C for 2 hr. Reaction mixture was slowly poured over crushed ice, organic layer was separated and aqueous layer was extracted with DCM (3 X 50 mL), combined organic layer was washed with brine solution, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to obtain (4- cyclopropylsulfanyl-phenyl)-oxo-acetic acid ethyl ester (3.1 g) as an oily product.
*H NMR (400 MHz, CDC13): δ 0.72-0.73 (m, 2H), 1.15-1.17 (m, 2H), 1.40 (t, J = 6.6 Hz, 3H), 2.18-2.21 (m, 1H), 4.41 (q, J = 6.8 Hz, 2H), 7.43 (d, J = 8.0 Hz, 2H), 7.90 (d, J = 8.0 Hz, 2H); MS (EI) m/z: 250.9 (M+l).
Step II: (4-Cyclopropanesulfonyl-phenyl) oxo acetic acid ethyl ester:
(4-Cyclopropylsulfanyl-phenyl)-oxo-acetic acid ethyl ester (3.1 g, 12.53 mmole) in DCM (50 mL) was cooled to 0-5 °C followed by addition of mCPBA (9.8 g , 31.33 mmol) in portion wise at 0 °C. After stirring at 25 °C for 4 hr, the reaction mixture was filtered; filtrate was washed with saturated aq. Na2S203 and satd. aq. sodium bicarbonate solution followed by brine solution, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to give (4-cyclopropanesulfonyl-phenyl) oxo acetic acid ethyl ester (3 g).
*H NMR (400 MHz, CDC13): δ 1.05-1.10 (m, 2H), 1.36-1.39 (m, 2H), 1.40 (t, J = 6.8 Hz, 3H), 2.45-2.50 (m, 1H), 4.42 (q, J = 7.2 Hz, 2H), 8.01 (d, J = 8.4 Hz, 2H), 8.20 (d, J = 8.4 Hz, 2H); MS (EI) m/z: 297.1 (M+NH4).
Step III: p-Toluene sulfonyl hydrazone (4-cyclopropyl sulfonyl) phenyl acetic acid ethyl ester:
A mixture of (4-cyclopropanesulfonyl-phenyl) oxo acetic acid ethyl ester (0.5 g, 1.77 mmole) and p-toluene sulfonyl hydrazide (0.48 g , 2.3 mmol) in toluene (15 mL) was refluxed for 16 hr using a Dean-Stark apparatus. Reaction mixture was concentrated to give the crude product which was purified by column chromatography over silica gel using 20-25% ethyl acetate in hexane as eluent to provide p-toluene sulfonyl hydrazone (4-cyclopropyl sulfonyl) phenyl acetic acid ethyl ester (0.5 g).
MS (EI) m/z 451.0 (M+l).
Step IV: (4-Cyclopropanesulfonyl-phenyl) diazo acetic acid ethyl ester:
To a solution of p-toluene sulfonyl hydrazone (4-cyclopropyl sulfonyl) phenyl acetic acid ethyl ester (0.5 g, 1.23 mmol) in dry DCM (6 mL), was added triethylamine (0.17 mL, 1.35 mmol) and stirred at 25 °C for 1 hr. Reaction mixture was concentrated to provide (4- cyclopropanesulfonyl-phenyl) diazo acetic acid ethyl ester (0.5 g) which was used in next reaction without any purification.
MS (EI) m/z: 295.1 (M+l).
Step V: Cyclopentyloxy-(4-cyclopropanesulfonyl-phenyl)-acetic acid ethyl ester:
(4-Cyclopropanesulfonyl-phenyl) diazo acetic acid ethyl ester (1 g, 3.37 mmol) was dissolved in DCM (16 mL) under argon atmosphere. To this solution, cyclopentanol (0.77 mL, 8.44 mmol) was added followed by rhodium(II)acetate dimer (0.062 g, 0.14 mmol). Mixture was stirred at 25 C for 12 hr. Reaction mixture was diluted with DCM (25 mL), organic layer was washed with water followed by brine solution, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to give a crude product which was purified by column chromatography using 25-35% ethyl acetate in hexane as eluent to provide cyclopentyloxy-(4- cyclopropanesulfonyl-phenyl)-acetic acid ethyl ester (0.35 g).
*H NMR (400 MHz, CDC13): δ 1.02-1.05 (m, 2H), 1.24 (t, J = 6.8 Hz, 3H), 1.35-1.37 (m, 2H), 1.53-1.82 (m, 8H), 2.42-2.50 (m, 1H), 4.02-4.04 (m, 1H), 4.15-4.22 (m, 2H), 5.00 (s, 1H), 7.66 (d, J = 8.0 Hz, 2H), 7.88 (d, J = 8.0 Hz, 2H); MS (EI) m/z: 370.0 (M+18).
Step VI: Cyclopentyloxy-(4-cyclopropanesulfonyl-phenyl)-acetic acid:
To cyclopentyloxy-(4-cyclopropanesulfonyl-phenyl)-acetic acid ethyl ester (0.35 g, 0.99 mmol) was added a solution of lithium hydroxide (0.208 g, 4.97 mmol) in water (4 mL) followed by THF (2 mL) and methanol (1 drop) and stirred for 12 hours at 25 0 C. Organic solvents were evaporated from the reaction mixture and aqueous layer was acidified IN HCl, extracted with ethyl acetate (3 X 10 mL), organic layer was washed with brine solution, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure to provide cyclopentyloxy-(4- cyclopropanesulfonyl-phenyl)-acetic acid (0.210 g).
*H NMR (400 MHz, CDC13): δ 1.02-1.07 (m, 2H), 1.34-1.38 (m, 2H), 1.55-1.62 (m, 2H), 1.69- 1.82 (m, 6H), 2.43-2.47 (m, 1H), 4.08-4.10 (m, 1H), 5.02 (s, 1H), 7.65 (d, J = 8.4 Hz, 2H), 7.91 (d, J = 8.4 Hz, 2H); MS (EI) m/z: 342.0 (M+18)
Example Al: 4-{2-[2-Cyclopentyloxy-2-(4-cyclopropanesulfonyl-phenyl)-acetylamino]-
To a mixture of cyclopentyloxy-(4-cyclopropanesulfonyl-phenyl)-acetic acid (Preparation 1) (0.1 g, 0.30 mmol), 4-(2-Amino-thiazol-5-yloxy)-benzoic acid methyl ester (0.085 g, 0.33 mmol), HOBt (0.045 g, 0.33 mmol), and EDCI (0.063 g, 0.33 mmol) in DCM (5 mL), was added N-methyl morpholine (0.033 g, 0.30 mmol). The resulting mixture was stirred at room temperature overnight followed by dilution with methylene chloride (20 mL). The reaction mixture was poured into water; organic layer was washed with water, brine, dried over sodium sulfate, and the organic solvent evaporated to get a residue which was purified by preparative TLC to provide the title compound (0.145 g).
*H NMR (400 MHz, CDC13): δ 1.03-1.05 (m, 2H), 1.34-1.38 (m, 2H), 1.58- 1.65 (m, 2H), 1.76- 1.81 (m, 6H), 2.42-2.45 (m, 1H), 3.89 (s, 3H), 4.05-4.15 (m, 1H), 5.08 (s, 1H), 7.07 (d, J = 8.8 Hz, 2H), 7.15 (s, 1H), 7.68 (d, J = 8.4 Hz, 2H), 7.92 (d, J = 8.4 Hz, 2H), 7.99 (d, J = 8.8 Hz, 2H), 9.72 (s, 1H); MS (EI) m/z: 556.9 (M + 1).
Example Bl: 4-{2-[2-Cyclopentyloxy-2-(4-cyclopropanesulfonyl-phenyl)-acetylamino]- thiazol-5-yloxy}-benzoic acid:
4-{2-[2-Cyclopentyloxy-2-(4-cyclopropanesulfonyl-phenyl)-acetylamino]-thiazol-5-yloxy}- benzoic acid methyl ester (0.145 g, 0.26 mmol, obtained in example Al) was taken in H20: THF (1 :2, 6 mL) to it was added MeOH (1 drop) followed by LiOH (0.054 g, 1.30 mmol) and stirred for 12 hr. After completion of the reaction, organic solvent was removed under reduced pressure. The aqueous layer was washed with diisopropyl ether then acidified with 1 N HC1 to pH 4. The solid formed was filtered, washed with water, diisopropyl ether & dried under vacuum to get the title_compound (0.12 g).
IH NMR- (400 MHz DMSO-ifc):- δ 1.01-1.05 (m, 2H), 1.09-1.13 (m, 2H), 1.22-1.49 (m, 2H), 1.59-1.73 (m, 6H), 2.82-2.86 (m, IH), 3.99-4.01 (m, IH), 5.31 (s, IH), 7.16 (d, J = 8.4 Hz, 2H), 7.37 (s, IH), 7.74 (d, J = 8.4 Hz, 2H), 7.91 (m, 4H), 12.55 (br. s, IH), 12.90 (br.s, IH); MS (EI) m/z: 542.9 (M+l)
CLIPPINGS
Advinus’ GK-activator Achieves Early POC for Diabetes
November 29 2011
Partnership Dialog Actively Underway
Advinus Therapeutics, a research-based pharmaceutical company founded by globally experienced industry executives and promoted by the TATA Group, announced that it has successfully completed a 14-day POC study in 60 Type II diabetic patients on its lead molecule, GKM-001, a glucokinase activator. The results of the trial show effective glucose lowering across all doses tested without any incidence of hypoglycemia or any other clinically relevant adverse events.
The clinical trials on GKM-001 validate the company’s pre-clinical hypothesis that a liver selective Glucokinase activator would not cause hypoglycemia (very low blood sugar), while showing robust efficacy.
“GKM-001 is differentiated from most other GK molecules that are in development, or have been discontinued, due to its novel liver selective mechanism of action. GKM-001 has a prolonged pharmacological effect and a half-life that should support a once a day dosing as both mono and combination therapy.” said Dr. Rashmi Barbhaiya, MD & CEO, Advinus Therapeutics. He added that Advinus is actively exploring partnership options to expedite further development and global marketing of GKM-001.
GKM-001 belongs to a novel class of molecules for treatment of type II diabetes. It is an activator of Glucokinase (GK), a glucose-sensing enzyme found mainly in the liver and pancreas. Being liver selective, GKM-001 mostly activates GK in the liver and not in pancreas, which is its key differentiation from most competitor molecules that activate GK in pancreas as well. The resulting increase in insulin secretion creates a potential for hypoglycemia-a risk GKM-001 is designed to avoid. Advinus has the composition of matter patent on GKM-001 for all major markets globally. Both the Single Ascending Dose data, in healthy and type II diabetics, and the Multiple Ascending Dose Study in Type II diabetics has shown that the molecule shows effective glucose lowering in a dose dependent manner and has excellent safety and tolerability profile over a 40-fold dose range. The pharmacokinetic properties of the molecule support once a day dosing. GKM-001 has the potential to be “First-in-Class” drug to address this large, growing and yet poorly addressed market.
Advinus also has identified a clinical candidate as a back-up to GKM-001, which is structurally different. In its portfolio, the company has a growing pipeline for COPD, sickle cell disease, inflammatory bowel disease, type 2 diabetes, acute and chronic pain and rheumatoid arthritis in various stages of late discovery and pre-clinical development.
About the Diabetes Market:
The present 300 million diabetics population is estimated to jump to 450 million by 2030 worldwide. A large proportion of these patients are poorly controlled despite multiple therapies. Total sales of diabetic prescription products were $32 billion in 2010.
Advinus Therapeutics team discovers novel molecule for treatment of diabetes
- The first glucokinase modulator discovered and developed in India
- A new concept for the management of diabetes for patients, globally
- 100 per cent ‘made in India’ molecule for the treatment of diabetes
- IND approved by DGCI, Phase I clinical trial shows excellent safety and tolerance profiles with efficacy
Bangalore: Advinus Therapeutics (Advinus), the research-based pharmaceutical company founded by leading global pharmaceutical executives and promoted by the Tata group, today, announced the discovery of a novel molecule for the treatment of type II diabetes — GKM-001.The molecule is an activator of glucokinase; an enzyme that regulates glucose balance and insulin secretion in the body.
GKM-001 is a completely indigenously developed molecule and the initial clinical trials have shown excellent results for both safety and efficacy.
“Considering past failures of other companies on this target, our discovery programme primarily focused on identifying a molecule that would be efficacious without causing hypoglycaemia; a side effect associated with most compounds developed for this target.
“Recently completed Phase I data indicate that Advinus’ GKM–001 is a liver selective molecule that has overcome the biggest clinical challenge of hypoglycaemia. GKM-001 is differentiated from most other GK molecules in development due to this novel mechanism of action,” said Dr Rashmi Barbhaiya, MD and CEO, Advinus Therapeutics.
He further added, “We are very proud that GKM-001 is 100 per cent Indian. Advinus’s discovery team in Pune discovered the molecule and entire preclinical development was carried out at our centre in Bangalore. The Investigational New Drug (IND) application was filed with the DGCI for approval to initiate clinical trials in India within 34 months of initiation of the discovery programme. Subsequent to the approval of the IND, we have completed the Phase I Single Ascending Dose study in India within two months.”
GKM-001 is a novel molecule for the treatment of type II diabetes. It is the first glucokinase modulator discovered and developed in India and has potential to be both first or best in class. The success in discovering GKM-001 is attributed to the science-driven efforts in Advinus laboratories and ‘breaking the conventional mold’ for selection of a drug candidate. Advinus has ‘composition of matter’ patent on the molecule for all major markets globally. Glucokinase as a class of target is considered to be novel as currently there is no product in the market or in late clinical trials. The strategy for early clinical development revolved around assessing safety (particularly hypoglycaemia) and early assessment of therapeutic activity (glucose lowering and other biomarkers) in type II diabetics. The Phase I data, in both healthy and type II diabetics, shows excellent safety and tolerability over a 40-fold dose range and desirable pharmacokinetic properties consistent with ‘once a day’ dosing. The next wave of clinical studies planned continues on this strategy of early testing in type II diabetics.
Right behind the lead candidate GKM-001, Advinus has a rich pipeline of back up compounds on the same target. These include several structurally different compounds with diverse potency, unique pharmacology and tissue selectivity. Having discovered the molecule with early indication of wide safety margins, desired efficacy and pharmacokinetic profiles, the company now seeks to out-licence GKM-001 and its discovery portfolio.
Patent
wo 2008104994
| WO2008104994A2 * | 25 Feb 2008 | 4 Sep 2008 | Advinus Therapeutics Private L | 2,2,2-tri-substituted acetamide derivatives as glucokinase activators, their process and pharmaceutical application |
| WO2008104994A2 * | Feb 25, 2008 | Sep 4, 2008 | Advinus Therapeutics Private L | 2,2,2-tri-substituted acetamide derivatives as glucokinase activators, their process and pharmaceutical application |
| WO2009047798A2 * | Oct 7, 2008 | Apr 16, 2009 | Advinus Therapeutics Private L | Acetamide derivatives as glucokinase activators, their process and medicinal applications |
///////GKM 001, pipeline, Diabetes, Advinus, type II diabetes, glucokinase modulator, Rashmi Barbhaiya
Some pics

Annual day party at Advinus !!!with Rashmi Barbhaiya
Dr. Rashmi Barbhaiya, MD & CEO, Advinus Therapeutics Pvt.
 .
.
with Kaushal Joshi, Vishal Pathade, Ramanareddy Jinugu, Mohammed Kakajiwala, Vishal Baxi and Dilip Reddy.
///////

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....











[…] GKM 001 in pipeline for Diabetes by Advinus […]
LikeLike