Cobimetinib
934660-93-2 cas ………….(S )enantiomer desired
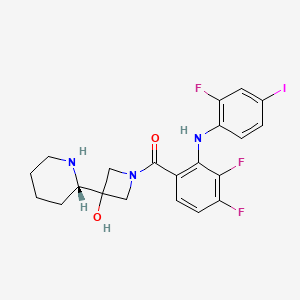
[3,4-Difluoro-2-(2-fluoro-4-iodoanilino)phenyl]{3-hydroxy-3-[(2S)-piperidin-2-yl]azetidin-1-yl} methanone
l-({3,4-difluoro-2-[(2-fluoro-4-iodophenyl)amino]phenyl}carbonyl)-3-[(2S)-piperidin-2- yl]azetidin-3-ol
1-[3,4-Difluoro-2-(2-fluoro-4-iodophenylamino)phenyl]-1-[3-hydroxy-3-[2(S)-piperidinyl]azetidin-1-yl]methanone
.gif) Cobimetinib (racemate)
Cobimetinib (racemate).gif) Cobimetinib (R-enantiomer)
Cobimetinib (R-enantiomer)CAS No: 934660-94-3
cobimetinib fumarate [USAN]
Molecular Formula: C46H46F6I2N6O8
Average mass: 1178.692261 Da
(2E)-2-Butendisäure –{3,4-difluor-2-[(2-fluor-4-iodphenyl)amino]phenyl}{3-hydroxy-3-[(2S)-2-piperidinyl]-1-azetidinyl}methanon (1:2) [German] [ACD/IUPAC Name]
{3,4-Difluoro-2-[(2-fluoro-4-iodophenyl)amino]phenyl}{3-hydroxy-3-[(2S)-2-piperidinyl]-1-azetidinyl}methanone (2E)-2-butenedioate (2:1) [ACD/IUPAC Name]
1369665-02-0 [RN]
Acide (2E)-2-butènedioïque – {3,4-difluoro-2-[(2-fluoro-4-iodophényl)amino]phényl}{3-hydroxy-3-[(2S)-2-pipéridinyl]-1-azétidinyl}méthanone (1:2) [French][ACD/IUPAC Name]
Bis({3,4-difluoro-2-[(2-fluoro-4-iodophenyl)amino]phenyl}{3-hydroxy-3-[(2S)-piperidin-2-yl]azetidin-1-yl}methanone) (2E)-but-2-enedioate
Methanone, [3,4-difluoro-2-[(2-fluoro-4-iodophenyl)amino]phenyl][3-hydroxy-3-[(2S)-2-piperidinyl]-1-azetidinyl]-, (2E)-2-butenedioate (2:1) (salt
Click to access cobimetinib-fumarate.pdf
Cobimetinib (GDC-0973, XL-518) is a MEK inhibitor being developed by Exelixis and Roche. It is being studied in combination withvemurafenib to treat several cancers, including melanoma.
Cobimetinib is an inhibitor of MEK kinase, which is an enzyme that the mitogen-activated protein kinase (MAPK). This compound was originated by Exelixis and is being developed by Genentech. Currently, Cobimetinb is in phase III trials at Genentech for the treatment of metastatic melanoma and in phase I clinical trials for the treatment of solid tumors. Cobimetinib has received an orphan drug designation in the U.S. for treatment of stage IIb, IIc, III, and IV melanoma with BRAFV600E mutation.
GDC-0973 (XL-518; GDC 0973) is a selective inhibitor of MEK GDC-0973 is also known as mitogen activated protein kinase kinase (MAPKK), is a key component of the RAS / RAF / MEK / ERK pathway, which. is frequently activated in human tumors.
Inappropriate activation of the MEK / ERK pathway promotes cell growth in the absence of exogenous growth factors.
The ERK/MAP kinase cascade is a key mechanism subject to dysregulation in cancer and is constitutively activated or highly upregulated in many tumor types. Mutations associated with upstream pathway components RAS and Raf occur frequently and contribute to the oncogenic phenotype through activation of MEK and then ERK. Inhibitors of MEK have been shown to effectively block upregulated ERK/MAPK signaling in a range of cancer cell lines and have further demonstrated early evidence of efficacy in the clinic for the treatment of cancer. Guided by structural insight, a strategy aimed at the identification of an optimal diphenylamine-based MEK inhibitor with an improved metabolism and safety profile versus PD-0325901 led to the discovery of development candidate 1-({3,4-difluoro-2-[(2-fluoro-4-iodophenyl)amino]phenyl}carbonyl)-3-[(2S)-piperidin-2-yl]azetidin-3-ol (XL518, GDC-0973) (1). XL518 exhibits robust in vitro and in vivo potency and efficacy in preclinical models with sustained duration of action and is currently in early stage clinical trials.
Process for the preparation of MEK inhibitors, relates cobimetinib. Genentech and its parent company Roche, under license from Exelixis, are developing cobimetinib, for the treatment of solid tumors, including melanoma, which is in phase 3 trials as of April 2014. The drug was originally disclosed in WO2007044515. For a previous filing on MEK inhibitors, see WO2008124085.
patent
http://www.google.com/patents/WO2007044515A1?cl=en
EXAMPLE 22(a) and 22(b) l-({3,4-difluoro-2-[(2-fluoro-4-iodophenyϊ)amino]phenyl}carbonyI)-3-[(2R)-piperidin-2- yl]azetidin-3-ol
l-({3,4-difluoro-2-[(2-fluoro-4-iodophenyl)amino]phenyl}carbonyl)-3-[(2S)-piperidin-2- yl]azetidin-3-ol
[00334] To a solution of 1 , 1 -dimethylethyl 2-(3 -hydroxy- 1 –
{[(phenylmethyl)oxy]carbonyl}azetidin-3-yl)piperidine-l-carboxylate (368 mg, 0.94 mmol), prepared using procedures similar to those described in Reference 5, in dichloromethane (5 mL) was added DMAP (115 mg, 0.94 mmol) and the resulting solution was cooled to O0C. (i?)-(-)-α-Methoxy-α-trifluoromethylphenylacetyl chloride (105 μL, 0.56 mmol) was added to the solution by syringe and the mixture was allowed to warm to room temperature then stirred an additional 12 hours. The solution was then partitioned with saturated aqueous soldium bicarbonate and the organic phase dried over anhydrous magnesium sulfate then filtered and concentrated to an oily residue.
Silica gel flash chromatography using hexanes:ethyl acetate 3:1 as eluent afforded the less polar 1,1 -dimethyl ethyl (2R)-2-(l- {[(phenylmethyl)oxy]carbonyl}-3-{[(2i?)-3,3,3-trifluoro-2-(methyloxy)-2- phenylpropanoyl]oxy}azetidin-3-yl)piperidine-l-carboxylate (27.5 mg, 5% yield), the more polar 1 , 1 -dimethylethyl (2S)-2-(l -{ [(phenylmethyl)oxy]carbonyl} -3-{ [(2i?)-3,3,3-trifluoro-2- (methyloxy)-2-phenylpropanoyl]oxy}azetidin-3-yl)piperidine-l-carboxylate (105 mg, 19% yield) and starting material (253 mg, 69% recovery).
[00335] The starting material thus recovered was taken into dichloromethane (3 mL) followed by addition of DMAP (115 mg, 0.94 mmol) and (i?)-(-)-α-methoxy-α- trifluoromethylphenylacetyl chloride (105 μL, 0.56 mmol) and the mixture was allowed to stir at room temperature over 12 hours. Proceeding as before afforded combined 1,1- dimethylethyl (2R)-2-(l-{[(phenylmethyl)oxy]carbonyl}-3-{[(2i?)-3,3,3-trifluoro- 2-(methyloxy)-2-phenylpropanoyl]oxy}azetidin-3-yl)piperidine-l-carboxylate (46.6 mg, 8% yield), the more polar 1,1 -dimethylethyl (25)-2-(l-{[(phenylmethyl)oxy]carbonyl}-3-{[(2i?)- 3,3,3-trifluoro-2-(methyloxy)-2-phenylpropanoyl]oxy}azetidin-3-yl)piperidine-l-carboxylate (228 mg, 41% yield) and starting material (100.8 mg, 27% recovery). [00336] The starting material thus recovered was taken into tetrahydrofuran: dichloromethane (1 :1, 2 mL) followed by addition of DMAP (47 mg, 0.39 mmol) and (R)-(-)-α-methoxy-α-trifluoromethylphenylacetyl chloride (80 μL, 0.43 mmol) and the mixture was heated to 60 0C over 12 hours.
Proceeding as before afforded combined less polar 1,1-dimethylethyl (2i?)-2-(l-{[(phenylmethyl)oxy]carbonyl}-3-{[(2i?)-3,3,3- trifluoro-2-(methyloxy)-2-phenylpropanoyl] oxy } azetidin-3 -yl)piperidine- 1 -carboxylate ( 144 mg, 26 % yield). The chiral ester derivatives thus obtained were again subject to silica gel flash chromatography using hexanes:ethyl acetate 3:1 as eluent to give the pure less polar 1,1-dimethylethyl (2i?)-2-(l-{[(phenylmethyl)oxy]carbonyl}-3-{[(2i?)-3,3,3-trifluoro-2-
(methyloxy)-2-phenylpropanoyl]oxy}azetidin-3-yl)piperidine-l-carboxylate (122.8 mg, 22% yield) and the more polar 1,1-dimethylethyl (2£)-2-(l-{[(phenylmethyl)oxy]carbonyl}-3- { [(2R)-3 ,3 ,3 -trifluoro-2-(methyloxy)-2-phenylpropanoyl] oxy } azetidin-3-yl)piperidine- 1 – carboxylate {111.6 mg, 32% yield) both as colorless amorphous residues. [00337] l,l-Dimethylethyl (2i?)-2-(l-{[(phenylmethyl)oxy]carbonyl}-3-{[(2i?)-3,3,3- trifluoro-2-(methyloxy)-2-phenylρropanoyl]oxy}azetidin-3-yl)piperidine-l-carboxylate (122.8 mg, 0.21 mmol) was taken into methanol (4 mL) followed by addition of IM aqueous sodium hydroxide (1 mL) and the resulting solution was stirred for one hour at room temperature.
The solution was then partitioned with ethyl acetate and IN aqueous hydrochloric acid. The organic layer was washed with brine, dried over anhydrous magnesium sulfate then filtered and concentrated. The residue was purified by silica gel flash chromatography using hexanes:ethyl acetate 2:1 to give 1,1-dimethylethyl (2i?)-2-(3- hydroxy- 1 – { [(phenylmethyl)oxy] carbonyl } azetidin-3 -yl)piperidine- 1 -carboxylate (60.8 mg, 81% yield) a colorless amorphous solid. 1,1-dimethylethyl (2ιS)-2-(3-hydroxy-l- {[(phenylmethyl)oxy]carbonyl}azetidin-3-yl)piperidine-l-carboxylate (87.4 mg, 75% yield) was prepared analogously.
[00338] 1 , 1 -Dimethylethyl (2i?)-2-(3-hydroxy- 1 – { [(phenylmethyl)oxy] carbonyl } azetidin- 3 -yl)piperidine-l -carboxylate (60.8 mg, 0.16 mmol) and 10% Pd/C (30 mg) were taken into methanol (2 mL) and the mixture hydrogenated at ambient pressure for one hour. The suspension was then filtered through a celite pad and concentrated then dried in vacuo to a colorless solid. The solid amine was taken into THF (1 mL) followed by addition of DIPEA (42 μL, 0.24 mmol) and 3,4-difluoro-2-[(2-fluoro-4-iodophenyl)amino]benzoyl fluoride (63 mg, 0.16 mmol), prepared using procedures similar to those described in Reference 1, and the mixture stirred at room temperature for 30 minutes.
The reaction mixture was partitioned with ethyl acetate and 1 N aqueous hydrochloric acid and the organic layer washed with brine, dried over anhydrous magnesium sulfate then filtered and concentrated. Purification of the residue by silica gel flash chromatography using hexanes: ethyl acetate 3:2 as eluent afforded 1,1-dimethylethyl (2i?)-2-[l-({3,4-difluoro-2-[(2-fluoro-4- iodophenyl)amino]phenyl}carbonyl)-3-hydroxyazetidin-3-yl]piperidine-l -carboxylate (74.9 mg, 74% yield) as an amorphous solid. 1,1 -Dimethylethyl (2i?)-2-[l-({3,4-difluoro-2-[(2- fluoro~4-iodophenyl)amino]phenyl}carbonyl)-3-hydroxyazetidin-3-yl]piperidine-l- carboxylate 1R NMR (400 MHz, CDCl3): 8.53 (br s, 0.5H), 8.40 (br s, 0.5H), 7.41-7.38 (dd, IH), 7.34-7.31(dt, IH), 7.17-7.14 (m, IH), 6.86-6.79 (m, IH), 6.63-6.587 (m, IH), 4.24-3.90 (m, 4H), 3.37-3.23 (m, IH), 2.90-2.80 (m, IH), 1.85-1.54 (m, 7H), 1.43 (s, 9H); MS (EI) for C26H29F3IN3O4: 576 (M-C4H9 4).
[00339] 1 , 1 -dimethylethyl (2R)-2-[l -({3,4-difluoro-2-[(2-fluoro-4- iodopheny^aminojphenylJcarbonyO-S-hydroxyazetidin-S-yljpiperidine-l-carboxylate (74.9 mg, 0.12 mmol) was taken into methanol (1 mL) followed by addition of 4 N HCl in dioxane (1 mL) and the solution was stirred at room temperature for one hour. The solution was then concentrated and the residue partitioned with chloroform and saturated aqueous sodium bicarbonate.
The organic layer was washed with brine, dried over anhydrous sodium’ sulfate then filtered and concentrated. Purification of the residue by silica gel flash chromatography using ethyl acetate then concentrated aqueous ammonia in chloroform and methanol (0.1 :10:1) as eluents afforded l-({3,4-difluoro-2-[(2-fluoro-4- iodophenyl)amino]phenyl}carbonyl)-3-[(2i?)-piperidin-2-yl]azetidin-3-ol (57.3 mg) as a colorless amorphous solid.
The free base was taken into methanol (1 mL) then brought to about pH 1 by addition of 4 N HCl in dioxane and the solution concentrated. The residue was triturated with ethyl ether to afford a suspension. The solid was collected by filtration to afford l-({3,4-difluoro-2-[(2-fluoro-4-iodophenyl)amino]phenyl}carbonyl)-3-[(2i?)- piperidin-2-yl]azetidin-3-ol hydrochloride salt (49 mg, 72% yield) as a colorless solid. 1H NMR (400 MHz, CDCl3): 8.43-8.39 (d, IH), 7.41-7.38 (dd, IH), 7.33-7.31(dt, IH), 7.14- 7.10 (m, IH), 6.84-6.80 (m, IH), 6.63-6.57 (m, IH), 4.12-3.99 (m, 4H), 3.10-3.08 (d, IH), 2.72-2.69 (d, IH), 2.64-2.62 (m, IH), 1.61-1.58 (m, 2H), 1.36-1.16 (m, 4H); MS (EI) for C21H2IF3IN3O2: 532 (MH+).

………………….
http://www.google.com/patents/WO2014027056A1?cl=en
[3,4-difluoro-2-[(2-fluoro-4- iodophenyl)amino]phenyl][3-hydroxy-3-[(2S)-2-piperidinyl]-l-azetidinyl]methanone.
GDC-0973 has the chemical structure:
[00156] Compound II may be prepared following the methods described in
US2009/0156576 (the contents of which are hereby incorporated by reference). Compound II has the following CAS Registry Number: 934660-93-2 .
http://www.google.com/patents/US20090156576
Example 22(a) and 22(b) 1-({3,4-difluoro-2-[(2-fluoro-4-iodophenyl)amino]phenyl}carbonyl)-3-[(2R)-piperidin-2-yl]azetidin-3-ol
and 1-({3,4-difluoro-2-[(2-fluoro-4-iodophenyl)amino]phenyl}carbonyl)-3-[(2S)-piperidin-2-yl]azetidin-3-ol
To a solution of 1,1-dimethylethyl 2-(3-hydroxy-1-{[(phenylmethyl)oxy]carbonyl}azetidin-3-yl)piperidine-1-carboxylate (368 mg, 0.94 mmol), prepared using procedures similar to those described in Reference 5, in dichloromethane (5 mL) was added DMAP (115 mg, 0.94 mmol) and the resulting solution was cooled to 0° C. (R)-(−)-α-Methoxy-α-trifluoromethylphenylacetyl chloride (105 μL, 0.56 mmol) was added to the solution by syringe and the mixture was allowed to warm to room temperature then stirred an additional 12 hours. The solution was then partitioned with saturated aqueous sodium bicarbonate and the organic phase dried over anhydrous magnesium sulfate then filtered and concentrated to an oily residue. Silica gel flash chromatography using hexanes:ethyl acetate 3:1 as eluent afforded the less polar 1,1-dimethylethyl (2R)-2-(1-{[(phenylmethyl)oxy]carbonyl}-3-{[(2R)-3,3,3-trifluoro-2-(methyloxy)-2-phenylpropanoyl]oxy}azetidin-3-yl)piperidine-1-carboxylate (27.5 mg, 5% yield), the more polar 1,1-dimethylethyl (2S)-2-(1-{[(phenylmethyl)oxy]carbonyl}-3-{[(2R)-3,3,3-trifluoro-2-(methyloxy)-2-phenylpropanoyl]oxy}azetidin-3-yl)piperidine-1-carboxylate (105 mg, 19% yield) and starting material (253 mg, 69% recovery).
The starting material thus recovered was taken into dichloromethane (3 mL) followed by addition of DMAP (115 mg, 0.94 mmol) and (R)-(−)-α-methoxy-α-trifluoromethylphenylacetyl chloride (105 μL, 0.56 mmol) and the mixture was allowed to stir at room temperature over 12 hours. Proceeding as before afforded combined 1,1-dimethylethyl (2R)-2-(1-{[(phenylmethyl)oxy]carbonyl}-3-{[(2R)-3,3,3-trifluoro-2-(methyloxy)-2-phenylpropanoyl]oxy}azetidin-3-yl)piperidine-1-carboxylate (46.6 mg, 8% yield), the more polar 1,1-dimethylethyl (2S)-2-(1-{[(phenylmethyl)oxy]carbonyl}-3-{[(2R)-3,3,3-trifluoro-2-(methyloxy)-2-phenylpropanoyl]oxy}azetidin-3-yl)piperidine-1-carboxylate (228 mg, 41% yield) and starting material (100.8 mg, 27% recovery).
The starting material thus recovered was taken into tetrahydrofuran:dichloromethane (1:1, 2 mL) followed by addition of DMAP (47 mg, 0.39 mmol) and (R)-(−)-α-methoxy-α-trifluoromethylphenylacetyl chloride (80 μL, 0.43 mmol) and the mixture was heated to 60° C. over 12 hours. Proceeding as before afforded combined less polar 1,1-dimethylethyl (2R)-2-(1-{[(phenylmethyl)oxy]carbonyl}-3-{[(2R)-3,3,3-trifluoro-2-(methyloxy)-2-phenylpropanoyl]oxy}azetidin-3-yl)piperidine-1-carboxylate (144 mg, 26% yield). The chiral ester derivatives thus obtained were again subject to silica gel flash chromatography using hexanes:ethyl acetate 3:1 as eluent to give the pure less polar 1,1-dimethylethyl (2R)-2-(1-{[(phenylmethyl)oxy]carbonyl}-3-{[(2R)-3,3,3-trifluoro-2-(methyloxy)-2-phenylpropanoyl]oxy}azetidin-3-yl)piperidine-1-carboxylate (122.8 mg, 22% yield) and the more polar 1,1-dimethylethyl (2S)-2-(1-{[(phenylmethyl)oxy]carbonyl}-3-{[(2R)-3,3,3-trifluoro-2-(methyloxy)-2-phenylpropanoyl]oxy}azetidin-3-yl)piperidine-1-carboxylate (177.6 mg, 32% yield) both as colorless amorphous residues.
1,1-Dimethylethyl (2R)-2-(1-{[(phenylmethyl)oxy]carbonyl}-3-{[(2R)-3,3,3-trifluoro-2-(methyloxy)-2-phenylpropanoyl]oxy}azetidin-3-yl)piperidine-1-carboxylate (122.8 mg, 0.21 mmol) was taken into methanol (4 mL) followed by addition of 1M aqueous sodium hydroxide (1 mL) and the resulting solution was stirred for one hour at room temperature. The solution was then partitioned with ethyl acetate and 1N aqueous hydrochloric acid. The organic layer was washed with brine, dried over anhydrous magnesium sulfate then filtered and concentrated. The residue was purified by silica gel flash chromatography using hexanes:ethyl acetate 2:1 to give 1,1-dimethylethyl (2R)-2-(3-hydroxy-1-{[(phenylmethyl)oxy]carbonyl}azetidin-3-yl)piperidine-1-carboxylate (60.8 mg, 81% yield) a colorless amorphous solid. 1,1-dimethylethyl (2S)-2-(3-hydroxy-1-{[(phenylmethyl)oxy]carbonyl}azetidin-3-yl)piperidine-1-carboxylate (87.4 mg, 75% yield) was prepared analogously.
1,1-Dimethylethyl (2R)-2-(3-hydroxy-1-{[(phenylmethyl)oxy]carbonyl}azetidin-3-yl)piperidine-1-carboxylate (60.8 mg, 0.16 mmol) and 10% Pd/C (30 mg) were taken into methanol (2 mL) and the mixture hydrogenated at ambient pressure for one hour. The suspension was then filtered through a celite pad and concentrated then dried in vacuo to a colorless solid. The solid amine was taken into THF (1 mL) followed by addition of DIPEA (42 μL, 0.24 mmol) and 3,4-difluoro-2-[(2-fluoro-4-iodophenyl)amino]benzoyl fluoride (63 mg, 0.16 mmol), prepared using procedures similar to those described in Reference 1, and the mixture stirred at room temperature for 30 minutes. The reaction mixture was partitioned with ethyl acetate and 1 N aqueous hydrochloric acid and the organic layer washed with brine, dried over anhydrous magnesium sulfate then filtered and concentrated. Purification of the residue by silica gel flash chromatography using hexanes:ethyl acetate 3:2 as eluent afforded 1,1-dimethylethyl (2R)-2-[1-({3,4-difluoro-2-[(2-fluoro-4-iodophenyl)amino]phenyl}carbonyl)-3-hydroxyazetidin-3-yl]piperidine-1-carboxylate (74.9 mg, 74% yield) as an amorphous solid. 1,1-Dimethylethyl (2R)-2-[1-({3,4-difluoro-2-[(2-fluoro-4-iodophenyl)amino]phenyl}carbonyl)-3-hydroxyazetidin-3-yl]piperidine-1-carboxylate 1H NMR (400 MHz, CDCl3): 8.53 (br s, 0.5H), 8.40 (br s, 0.5H), 7.41-7.38 (dd, 1H), 7.34-7.31 (dt, 1H), 7.17-7.14 (m, 1H), 6.86-6.79 (m, 1H), 6.63-6.587 (m, 1H), 4.24-3.90 (m, 4H), 3.37-3.23 (m, 1H), 2.90-2.80 (m, 1H), 1.85-1.54 (m, 7H), 1.43 (s, 9H); MS (EI) for C26H29F3IN3O4: 576 M-C4H9 +).
1,1-dimethylethyl (2R)-2-[1-({3,4-difluoro-2-[(2-fluoro-4-iodophenyl)amino]phenyl}carbonyl)-3-hydroxyazetidin-3-yl]piperidine-1-carboxylate (74.9 mg, 0.12 mmol) was taken into methanol (1 mL) followed by addition of 4 N HCl in dioxane (1 mL) and the solution was stirred at room temperature for one hour. The solution was then concentrated and the residue partitioned with chloroform and saturated aqueous sodium bicarbonate. The organic layer was washed with brine, dried over anhydrous sodium sulfate then filtered and concentrated. Purification of the residue by silica gel flash chromatography using ethyl acetate then concentrated aqueous ammonia in chloroform and methanol (0.1:10:1) as eluents afforded 1-({3,4-difluoro-2-[(2-fluoro-4-iodophenyl)amino]phenyl}carbonyl)-3-[(2R)-piperidin-2-yl]azetidin-3-ol (57.3 mg) as a colorless amorphous solid. The free base was taken into methanol (1 mL) then brought to about pH 1 by addition of 4 N HCl in dioxane and the solution concentrated. The residue was triturated with ethyl ether to afford a suspension. The solid was collected by filtration to afford 1-({3,4-difluoro-2-[(2-fluoro-4-iodophenyl)amino]phenyl}carbonyl)-3-[(2R)-piperidin-2-yl]azetidin-3-ol hydrochloride salt (49 mg, 72% yield) as a colorless solid. 1H NMR (400 MHz, CDCl3): 8.43-8.39 (d, 1H), 7.41-7.38 (dd, 1H), 7.33-7.31 (dt, 1H), 7.14-7.10 (m, 1H), 6.84-6.80 (m, 1H), 6.63-6.57 (m, 1H), 4.12-3.99 (m, 4H), 3.10-3.08 (d, 1H), 2.72-2.69 (d, 1H), 2.64-2.62 (m, 1H), 1.61-1.58 (m, 2H), 1.36-1.16 (m, 4H); MS (EI) for C21H21F3IN3O2: 532 (MH+).
………………………………..
Novel carboxamide-based allosteric MEK inhibitors: Discovery and optimization efforts toward XL518 (GDC-0973)
ACS Med Chem Lett 2012, 3(5): 416
http://pubs.acs.org/doi/abs/10.1021/ml300049d

http://pubs.acs.org/doi/abs/10.1021/ml300049d
|
8-17-2011
|
Methods of using MEK inhibitors
|
|
|
3-30-2011
|
Azetidines as MEK Inhibitors for the Treatment of Proliferative Diseases
|
|
|
9-29-2010
|
Azetidines as MEK Inhibitors for the Treatment of Proliferative Diseases
|
|
|
3-26-2010
|
Methods of Using PI3K and MEK Modulators
|

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....