Home » Posts tagged 'benznidazole'
Tag Archives: benznidazole
Novel Drug Approvals for 2017, A Review/Compilation
CDSCO
 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO, Novel Drug Approvals for 2017, A Review Compilation (USFDA, EMA, PMDA, CDSCO).
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO, Novel Drug Approvals for 2017, A Review Compilation (USFDA, EMA, PMDA, CDSCO).
Any errors in this compilation, email amcrasto@gmail.com, Call +919323115463
Some gaps will be filled up soon keep watching……………..
INDEX, NAME (click on the title, it contains link)
SECTION A; USFDA Approvals
6 BENRALIZUMAB
17 DURVALUMAB
24 GUSELKUMAB
36 OZENOXACIN
40 SARILUMAB
41 SECNIDAZOLE
INDEX, FORMULATION NAME
USFDA
•Aliqopa (COPANLISIB) to treat adults with relapsed follicular lymphoma — a slow-growing type of nonHodgkin lymphoma (a cancer of the lymph system) — who have received at least two prior systemic therapies;
• ALUNBRIG, BRIGATINIB, To treat patients with anaplastic lymphoma kinase (ALK)-positive metastatic non-small cell lung cancer (NSCLC) who have progressed on or are intolerant to crizotinib
• Austedo, Deutetrabenazine For the treatment of chorea associated with Huntington’s disease
• Bavencio (avelumab) for the treatment of patients 12 years and older with a rare and aggressive form of cancer called metastatic Merkel cell carcinoma, including those who have not received prior chemotherapy;
•BAXDELLA, Delafloxacin, BACTERIAL INFECTIONS
• Benznidazole to treat children ages 2 to 12 years with Chagas disease, a parasitic infection that can cause serious heart illness after years of infection, and can also affect swallowing and digestion. This is the first treatment approved in the United States for this rare disease;
• Besponsa (inotuzumab ozogamicin) for the treatment of adults with a type of cancer of the blood called relapsed or refractory B-cell precursor acute lymphoblastic leukemia;
•BEVYXXA, BETRIXABAN, For the prophylaxis of venous thromboembolism (VTE) in adult patients hospitalized for an acute medical illness
• BRINEURA, CERLIPONASE ALFA, To treat a specific form of Batten disease
• Calquence (ACALABRUTINIB) to treat adults with mantle cell lymphoma who have received at least one prior therapy. Mantle cell lymphoma is a particularly aggressive cancer;
• DUPIXENT, (DUPILUMAB) To treat adults with moderate-to-severe eczema (atopic dermatitis)
• Emflaza (deflazacort) to treat patients age 5 years and older with Duchenne muscular dystrophy, a rare genetic disorder that causes progressive muscle deterioration and weakness;
• FASENRA, BENRALIZUMAB, For add-on maintenance treatment of patients with severe asthma aged 12 years and older, and with an eosinophilic phenotype
• Giapreza (angiotensin II), for the treatment of hypotension in adults with distributive or vasodilatory shock (dangerously low blood pressure despite adequate heart function) whose blood pressure remains low despite receiving fluids and treatment with drugs called vasopressors;
• HEMLIBRA EMICIZUMAB To prevent or reduce the frequency of bleeding episodes in adult and pediatric patients with hemophilia A who have developed antibodies called Factor VIII (FVIII) inhibitors.
• Idhifa (enasidenib) for the treatment of adults with relapsed or refractory acute myeloid leukemia, a form of blood cancer, who have a specific genetic mutation;
• IMFINZI, DURVALUMAB To treat patients with locally advanced or metastatic urothelial carcinoma
• Ingrezza (valbenazine) to treat adults with tardive dyskinesia, a side effect of some antipsychotic medications whereby patients can experience uncontrollable stiff, jerky movements of their face and body, and other uncontrolled movements such as eye-blinking, sticking out the tongue, and arm-waving;
• KEVZARA SARILUMAB, RHEUMATOID ARTHRITIS
• KISQALI, RIBOCICLIB, To treat postmenopausal women with a type of advanced breast cancer
• Macrilen macimorelin acetate, For the diagnosis of adult growth hormone deficiency
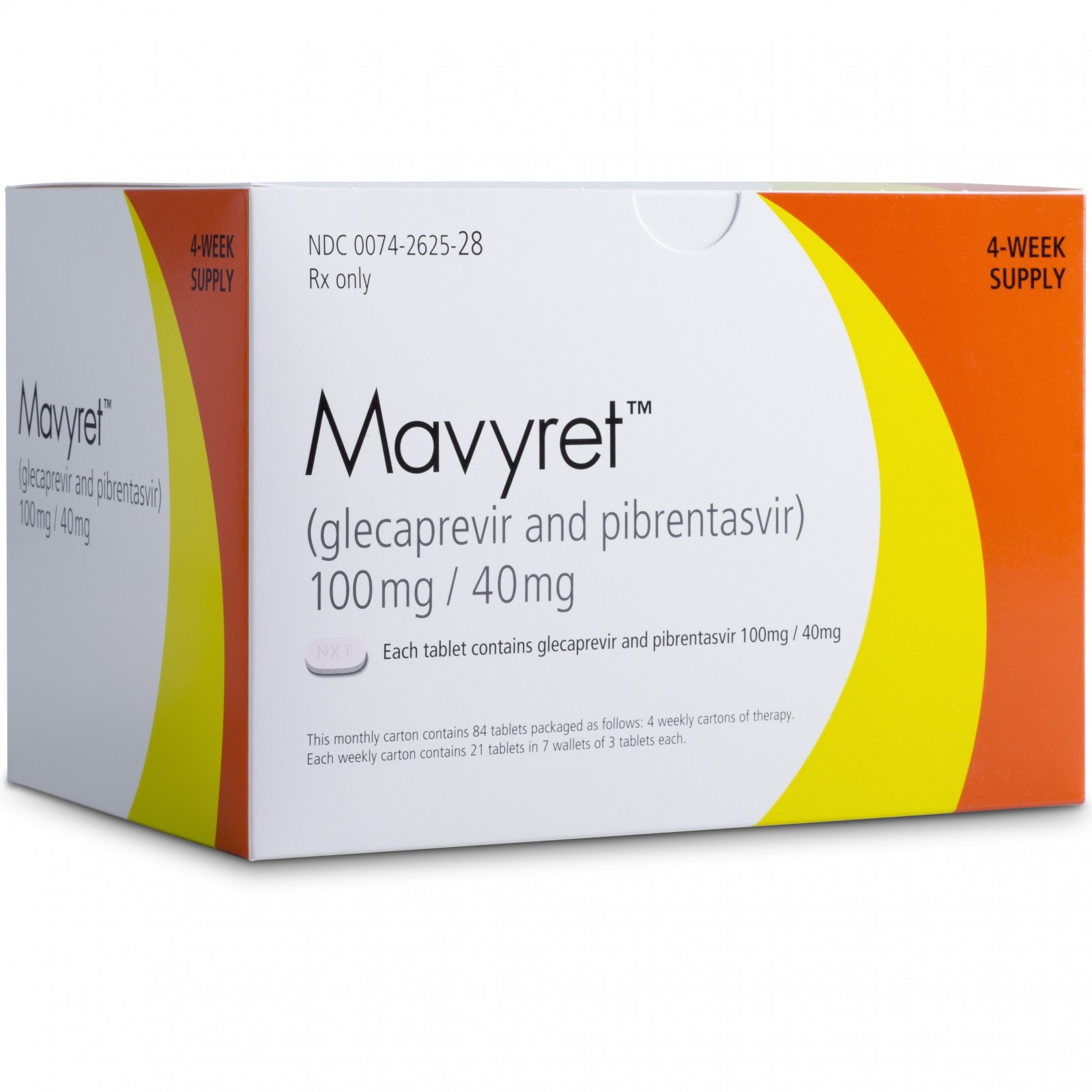
• Mavyret (glecaprevir and pibrentasvir) to treat adults with chronic hepatitis C virus genotypes 1-6 without cirrhosis (liver disease) or with mild cirrhosis, including patients with moderate to severe kidney disease, as well as those who are on hemodialysis;
• Mepsevii (vestronidase alfa-vjbk) to treat patients with Sly syndrome or mucopolysaccharidosis type 7 – a rare genetic disorder where an enzyme deficiency results in skeletal abnormalities, developmental delay, enlarged liver and spleen, and narrowed airways, which can lead to respiratory infections;
• Nerlynx (neratinib) for the extended adjuvant treatment — a form of therapy administered after an initial treatment to further lower the risk of the cancer coming back — of early-stage, human epidermal growth factor receptor 2 (HER2)-positive breast cancer;
• OCREVUS, OCRELIZUMAB, To treat patients with relapsing and primary progressive forms of multiple sclerosis
• OZEMPIC SEMAGLUTIDE To improve glycemic control in adults with type 2 diabetes mellitus
•PARSABIV, ETELCALCETIDE, To treat secondary hyperparathyroidism in adult patients with chronic kidney disease undergoing dialysis
• Prevymis (letermovir) for prevention of an infection called cytomegalovirus (CMV) in patients who are receiving a bone marrow transplant. CMV disease can cause serious health issues in these patients;
• Radicava (edaravone) to treat patients with amyotrophic lateral sclerosis, commonly referred to as Lou Gehrig’s disease, a rare disease that attacks and kills the nerve cells that control voluntary muscles;
• RHOPRESSA, NETARSUDIL, To treat glaucoma or ocular hypertension
• Rydapt (midostaurin) to treat adults newly diagnosed with a form of blood cancer known as acute myeloid leukemia who have a specific genetic mutation called FLT3, in combination with chemotherapy;
• Siliq (brodalumab) to treat adults with moderate-to-severe plaque psoriasis, a chronic disorder in which the body’s immune system sends out faulty signals that speed growth of skin cells that then accumulate, causing red, flaky patches that can appear anywhere on the body;
•SOLOSEC, SECNIDAZOLE To treat bacterial vaginosis
• STEGLATRO ERTUGLIFLOZIN To improve glycemic control in adults with type 2 diabetes mellitus
• Symproic (Naldemedine) for the treatment of opioid-induced constipation in adults with chronic noncancer pain; • Tremfya (guselkumab) for the treatment of adults with moderate-to-severe plaque psoriasis;
• Trulance (plecanatide) to treat adults with chronic idiopathic constipation, which is a persistent condition of constipation due to unknown origin;
• TYMLOS, Abaloparatide, To treat osteoporosis in postmenopausal women at high risk of fracture or those who have failed other therapies
• Vabomere (vaborbactam and meropenem) for treatment of adults with complicated urinary tract infections, including pyelonephritis (kidney infection) caused by bacteria;
• Verzenio (abemaciclib) to treat adults who have hormone receptor (HR)-positive, HER2-negative advanced or metastatic breast cancer that has progressed after taking therapy that alters a patient’s hormones (endocrine therapy);
• Vosevi (sofosbuvir/velpatasvir/voxilaprevir) to treat adults with chronic hepatitis C virus genotypes 1-6 without cirrhosis (liver disease) or with mild cirrhosis;
• Xadago (safinamide) as an add-on treatment for patients with Parkinson’s disease who are currently taking levodopa/carbidopa and experiencing “off” episodes;
• XERMELO, TELOTRISTAT ETHYL combined with somatostatin analog (SSA) therapy to treat adults with carcinoid syndrome diarrhea that SSA therapy alone has inadequately controlled, and;
• XEPI OZENOXACIN TO TREAT IMPETIGO
•XERMELO, TELOTRISTAT ETHYL, To treat carcinoid syndrome diarrhea
• Zejula (niraparib) for the maintenance treatment (intended to delay cancer growth) of adults with recurrent epithelial ovarian, fallopian tube or primary peritoneal cancer, whose tumors have completely or partially shrunk (complete or partial response, respectively) in response to platinum-based chemotherapy
USFDA
| No. | Drug Name |
Active Ingredient | Approval Date | FDA-approved use on approval date |
|---|---|---|---|---|
| 46. | Giapreza | angiotensin II | 12/21/2017 | To increase blood pressure in adults with septic or other distributive shock |
| 45. | Macrilen | macimorelin acetate | 12/20/2017 | For the diagnosis of adult growth hormone deficiency |
| 44. | Steglatro | ertugliflozin | 12/19/2017 | To improve glycemic control in adults with type 2 diabetes mellitus |
| 43. | Rhopressa | netarsudil | 12/18/2017 | To treat glaucoma or ocular hypertension |
| 42. | Xepi | ozenoxacin | 12/11/2017 | To treat impetigo Drug Trials Snapshot |
| 41. | Ozempic | semaglutide | 12/5/2017 | To improve glycemic control in adults with type 2 diabetes mellitus |
| 40. | Hemlibra | emicizumab | 11/16/2017 | To prevent or reduce the frequency of bleeding episodes in adult and pediatric patients with hemophilia A who have developed antibodies called Factor VIII (FVIII) inhibitors. |
| 39. | Mepsevii | vestronidase alfa-vjbk | 11/15/2017 | To treat pediatric and adult patients with an inherited metabolic condition called mucopolysaccharidosis type VII (MPS VII), also known as Sly syndrome. |
| 38. | Fasenra | benralizumab | 11/14/2017 | For add-on maintenance treatment of patients with severe asthma aged 12 years and older, and with an eosinophilic phenotype Drug Trials Snapshot |
| 37. | Prevymis | letermovir | 11/8/2017 | To prevent infection after bone marrow transplant Drug Trials Snapshot |
| 36. | Vyzulta | latanoprostene bunod ophthalmic solution | 11/2/2017 | To treat intraocular pressure in patients with open-angle glaucoma or ocular hypertension. Drug Trials Snapshot |
| 35. | Calquence | acalabrutinib | 10/31/2017 | To treat adults with mantle cell lymphoma Press Release Drug Trials Snapshot |
| 34. | Verzenio | abemaciclib | 9/28/2017 | To treat certain advanced or metastatic breast cancers Press Release Drug Trials Snapshot |
| 33. | Solosec | secnidazole | 9/15/2017 | To treat bacterial vaginosis Drug Trials Snapshot |
| 32. | Aliqopa | copanlisib | 9/14/2017 | To treat adults with relapsed follicular lymphoma Press Release Drug Trials Snapshot |
| 31. | benznidazole | benznidazole | 8/29/2017 | To treat children ages 2 to 12 years old with Chagas disease Press Release Drug Trials Snapshot |
| 30. | Vabomere | meropenem and vaborbactam | 8/29/2017 | To treat adults with complicated urinary tract infections Press Release Drug Trials Snapshot |
| 29. | Besponsa | inotuzumab ozogamicin | 8/17/2017 | To treat adults with relapsed or refractory acute lymphoblastic leukemia Press Release Drug Trials Snapshot |
| 28. | Mavyret | glecaprevir and pibrentasvir | 8/3/2017 | To treat adults with chronic hepatitis C virus Press Release Drug Trials Snapshot |
| 27. | Idhifa | enasidenib | 8/1/2017 | To treat relapsed or refractory acute myeloid leukemia Press Release Drug Trials Snapshot |
| 26. | Vosevi | sofosbuvir, velpatasvir and voxilaprevir | 7/18/2017 | To treat adults with chronic hepatitis C virus Press Release Drug Trials Snapshot |
| 25. | Nerlynx | neratinib maleate | 7/17/2017 | To reduce the risk of breast cancer returning Press Release Drug Trials Snapshot |
| 24. | Tremfya | guselkumab | 7/13/2017 | For the treatment of adult patients with moderate-to-severe plaque psoriasis Drug Trials Snapshot |
| 23. | Bevyxxa | betrixaban | 6/23/2017 | For the prophylaxis of venous thromboembolism (VTE) in adult patients hospitalized for an acute medical illness Drug Trials Snapshot |
| 22. | Baxdela | delafloxacin | 6/19/2017 | To treat patients with acute bacterial skin infections Drug Trials Snapshot |
| 21. | Kevzara | sarilumab | 5/22/2017 | To treat adult rheumatoid arthritis Drug Trials Snapshot |
| 20. | Radicava | edaravone | 5/5/2017 | To treat patients with amyotrophic lateral sclerosis (ALS) Press Release Drug Trials Snapshot |
| 19. | Imfinzi | durvalumab | 5/1/2017 | To treat patients with locally advanced or metastatic urothelial carcinoma Web Post Drug Trials Snapshot |
| 18. | Tymlos | abaloparatide | 4/28/2017 | To treat osteoporosis in postmenopausal women at high risk of fracture or those who have failed other therapies Drug Trials Snapshot |
| 17. | Rydapt | midostaurin | 4/28/2017 | To treat acute myeloid leukemia Press Release Chemistry Review(s) (PDF) Drug Trials Snapshot |
| 16. | Alunbrig | brigatinib | 4/28/2017 | To treat patients with anaplastic lymphoma kinase (ALK)-positive metastatic non-small cell lung cancer (NSCLC) who have progressed on or are intolerant to crizotinib Drug Trials Snapshot |
| 15. | Brineura | cerliponase alfa | 4/27/2017 | To treat a specific form of Batten disease Press Release Drug Trials Snapshot |
| 14. | Ingrezza | valbenazine | 4/11/2017 | To treat adults with tardive dyskinesia Press Release Chemistry Review(s) (PDF)Drug Trials Snapshot |
| 13. | Austedo | deutetrabenazine | 4/3/2017 | For the treatment of chorea associated with Huntington’s disease Drug Trials Snapshot, Chemistry Review(s) (PDF) |
| 12. | Ocrevus | ocrelizumab | 3/28/2017 | To treat patients with relapsing and primary progressive forms of multiple sclerosis Press Release Drug Trials Snapshot |
| 11. | Dupixent | dupilumab | 3/28/2017 | To treat adults with moderate-to-severe eczema (atopic dermatitis) Press Release Drug Trials Snapshot |
| 10. | Zejula | niraparib | 3/27/2017 | For the maintenance treatment for recurrent epithelial ovarian, fallopian tube or primary peritoneal cancers Press Release Drug Trials Snapshot |
| 9. | Symproic | naldemedine | 3/23/2017 |
For the treatment of opioid-induced constipation |
| 8. | Bavencio | avelumab | 3/23/2017 | To treat metastatic Merkel cell carcinoma Press Release Drug Trials Snapshot |
| 7. | Xadago | safinamide | 3/21/2017 | To treat Parkinson’s disease Press Release Drug Trials SnapshotChemistry Review(s) (PDF) |
| 6. | Kisqali | ribociclib | 3/13/2017 | To treat postmenopausal women with a type of advanced breast cancer Drug Trials Snapshot |
| 5. | Xermelo | telotristat ethyl | 2/28/2017 | To treat carcinoid syndrome diarrhea Press Release Drug Trials Snapshot |
| 4. | Siliq | brodalumab | 2/15/2017 | To treat adults with moderate-to-severe plaque psoriasis Press Release Drug Trials Snapshot |
| 3. | Emflaza | deflazacort | 2/9/2017 | To treat patients age 5 years and older with Duchenne muscular dystrophy (DMD) Press Release Drug Trials Snapshot |
| 2. | Parsabiv | etelcalcetide | 2/7/2017 | To treat secondary hyperparathyroidism in adult patients with chronic kidney disease undergoing dialysis Drug Trials Snapshot |
| 1. | Trulance | plecanatide | 1/19/2017 | To treat Chronic Idiopathic Constipation (CIC) in adult patients. Press Release Drug Trials Snapshot |
* This information is currently accurate. In rare instances, it may be necessary for FDA to change a drug’s new molecular entity (NME) designation or the status of its application as a novel new biologics license application (BLA). For instance, new information may become available which could lead to a reconsideration of the original designation or status. If changes must be made to a drug’s designation or the status of an application as a novel BLA, the Agency intends to communicate the nature of, and the reason for, any revisions as appropriate.
| USFDA 2017 | ||||||
| 2017/12/21 | Angiotensin II | Giapreza | La Jolla Pharmaceutical | |||
| 2017/12/20 | Ertugliflozin | Steglatro | Merck Sharp Dohme | |||
| 2017/12/20 | Macimorelin acetate | Macrilen | Aeterna Zentaris GmbH | |||
| 2017/12/18 | Netarsudil mesylate | Rhopressa | Aerie Pharmaceuticals | |||
| 2017/12/11 | Ozenoxacin | Xepi | Ferrer Internacional S.A. | |||
| 2017/12/5 | Semaglutide | Ozempic | Novo Nordisk Inc | |||
| 2017/11/16 | Emicizumab | Hemlibra | Genentech | BLA | ||
| 2017/11/15 | Vestronidase alfa | Mepsevii | Ultragenyx Pharmaceutical | BLA | ||
| 2017/11/14 | Benralizumab | Fasenra | AstraZeneca AB | BLA | ||
| 2017/11/8 | Letermovir | Prevymis | Merck Sharp Dohme | |||
| 2017/11/2 | Latanoprostene bunod | Vyzulta | Bausch & Lomb Incorporated | |||
| 2017/10/31 | Acalabrutinib | Calquence | AstraZeneca Pharmaceuticals LP | |||
| 2017/9/28 | Abemaciclib | Verzenio | Eli Lilly | |||
| 2017/9/15 | Secnidazole | Solosec | Symbiomix Therapeutics | |||
| 2017/9/14 | Copanlisib | Aliqopa | Bayer Healthcare Pharmaceuticals | |||
| 2017/8/29 | Benznidazole | Chemo Research | ||||
| 2017/8/29 | Meropenem – Vaborbactam | Vabomere | Rempex Pharmaceuticals | |||
| 2017/8/17 | Inotuzumab ozogamicin | Besponsa | Wyeth Pharmaceuticals | BLA | ||
| 2017/8/3 | Glecaprevir – Pibrentasvir | Mavyret | AbbVie | |||
| 2017/8/1 | Enasidenib | Idhifa | Celgene Corporation | |||
| 2017/7/18 | Sofosbuvir – Velpatasvir – Voxilaprevir | Vosevi | Gilead Sciences | |||
| 2017/7/17 | Neratinib maleate | Nerlynx | Puma Biotechnology | |||
| 2017/7/13 | Guselkumab | Tremfya | Janssen Biotech | BLA | ||
| 2017/6/23 | Betrixaban | Bevyxxa | Portola Pharmaceuticals | |||
| 2017/6/19 | Delafloxacin meglumine | Baxdela | Melinta Therapeutics | |||
| 2017/5/22 | Sarilumab | Kevzara | Sanofi Synthelabo | BLA | ||
| 2017/5/5 | Edaravone | Radicava | Mitsubishi Tanabe Pharma America | |||
| 2017/5/1 | Durvalumab | Imfinzi | AstraZeneca UK | BLA | ||
| 2017/4/28 | Abaloparatide | Tymlos | Radius Health | |||
| 2017/4/28 | Midostaurin | Rydapt | Novartis Pharmaceuticals | |||
| 2017/4/28 | Brigatinib | Alunbrig | Ariad Pharmaceuticals | |||
| 2017/4/27 | Cerliponase alfa | Brineura | BioMarin Pharmaceutical | BLA | ||
| 2017/4/11 | Valbenazine | Ingrezza | Neurocrine Biosciences | |||
| 2017/4/3 | Deutetrabenazine | Austedo | Teva Pharmaceuticals | |||
| 2017/3/28 | Ocrelizumab | Ocrevus | Genentech | BLA | ||
| 2017/3/28 | Dupilumab | Dupixent | Regeneron Pharmaceuticals | BLA | ||
| 2017/3/27 | Niraparib | Zejula | Tesaro | |||
| 2017/3/23 | Naldemedine tosylate | Symproic | Shionogi | |||
| 2017/3/23 | Avelumab | Bavencio | EMD Serono | BLA | ||
| 2017/3/23 | Safinamide mesylate | Xadago | Newron Pharmaceuticals | |||
| 2017/3/21 | Ribociclib | Kisqali | Novartis Pharmaceuticals | |||
| 2017/2/28 | Telotristat ethyl | Xermelo | Lexicon Pharmaceuticals | |||
| 2017/2/15 | Brodalumab | Siliq | Valeant Pharmaceuticals | BLA | ||
| 2017/2/9 | Deflazacort | Emflaza | Marathon Pharmaceuticals | |||
| 2017/2/8 | Etelcalcetide hydrochloride | Parsavib | KAI Pharmaceuticals | |||
| 2017/1/19 | Plecanatide | Trulance | Synergy Pharmaceuticals |
1 Abaloparatide
RADIUS

FDA 4/28/2017
To treat osteoporosis in postmenopausal women at high risk of fracture or those who have failed other therapies
Drug Trials Snapshot

2 Abemaciclib
ELI LILLY
| Verzenio | abemaciclib | FDA 9/28/2017 | To treat certain advanced or metastatic breast cancers Press Release Drug Trials Snapshot |
LINK https://newdrugapprovals.org/2015/10/19/abemaciclib-bemaciclib/
3 Acalabrutinib
| Calquence | FDA APPROVED
10/31/2017 |
To treat adults with mantle cell lymphoma Press Release Drug Trials Snapshot |
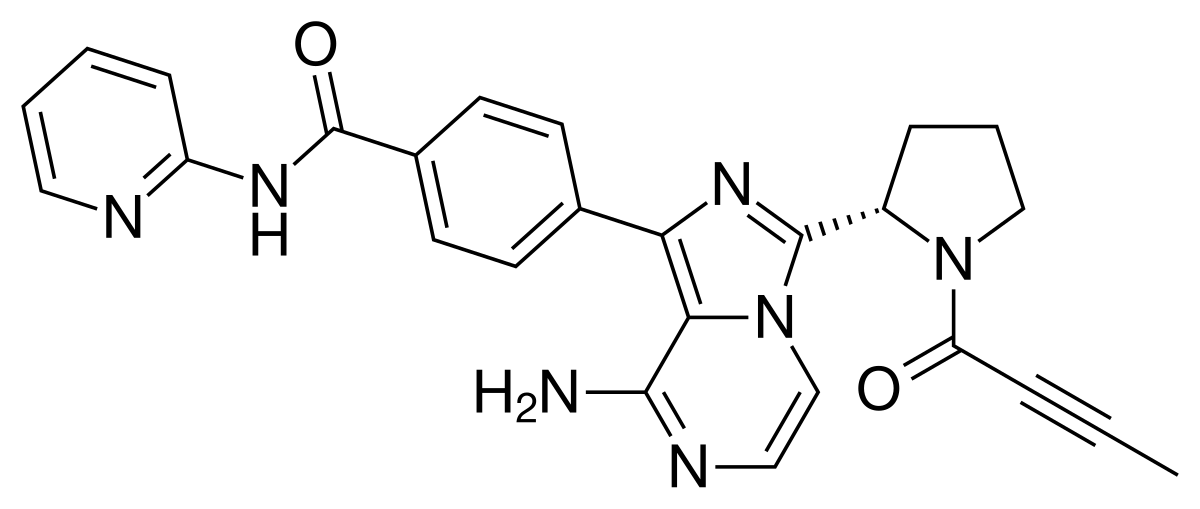

-Facebook.png)
4 Angiotensin II
LA JOLLA
| Giapreza | angiotensin II | 12/21/2017 | To increase blood pressure in adults with septic or other distributive shock Press Release Drug Trials Snapshot |


5 AVELUMAB
MERCK

| Bavencio | FDA 3/23/2017 | To treat metastatic Merkel cell carcinoma Press Release Drug Trials Snapshot |
6 BENRALIZUMAB
ASTRA ZENECA
Fasenra benralizumab
FDA 11/14/2017
For add-on maintenance treatment of patients with severe asthma aged 12 years and older, and with an eosinophilic phenotype
Drug Trials Snapshot


7 Benznidazole
CHEMO RESEARCH



| benznidazole | FDA
8/29/2017 |
To treat children ages 2 to 12 years old with Chagas disease Press Release Drug Trials Snapshot |
8 BETRIXABAN
PORTOLA PHARMA

| Bevyxxa | FDA
6/23/2017 |
For the prophylaxis of venous thromboembolism (VTE) in adult patients hospitalized for an acute medical illness Drug Trials Snapshot
|


9 BRIGATINIB
TAKEDA


| Alunbrig | FDA
4/28/2017 |
To treat patients with anaplastic lymphoma kinase (ALK)-positive metastatic non-small cell lung cancer (NSCLC) who have progressed on or are intolerant to crizotinib Drug Trials Snapshot |
10 BRODALUMAB
VALEANT PHARMA
| Siliq | FDA
2/15/2017 |
To treat adults with moderate-to-severe plaque psoriasis Press Release Drug Trials Snapshot |

LINK ,,,,https://newdrugapprovals.org/2017/02/16/fda-approves-new-psoriasis-drug-siliq-brodalumab/
11 CERLIPONASE ALFA

| Brineura | FDA 4/27/2017 | To treat a specific form of Batten disease Press Release Drug Trials Snapshot |
12 Copanlisib
| Aliqopa | FDA APPROVED
9/14/2017 |
To treat adults with relapsed follicular lymphoma Press Release Drug Trials Snapshot |

LINK…..https://newdrugapprovals.org/2017/11/20/copanlisib/
13 DEFLAZACORT
MARATHON PHARMA

| Emflaza | FDA 2/9/2017 | To treat patients age 5 years and older with Duchenne muscular dystrophy (DMD) Press Release Drug Trials Snapshot |
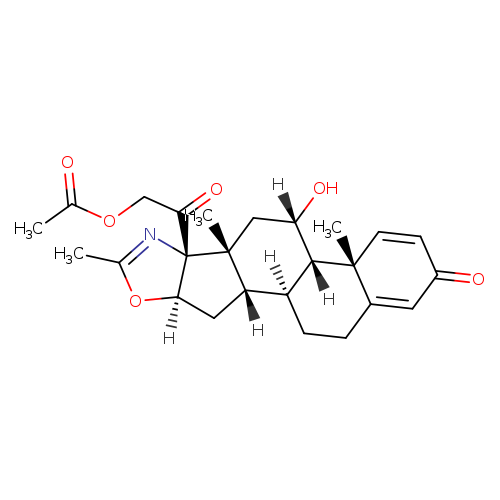
LINK……https://newdrugapprovals.org/2017/02/17/deflazacort/
14 DELAFLOXACIN
| Baxdela | FDA APPROVED
6/19/2017 |
To treat patients with acute bacterial skin infections |


LINK……..https://newdrugapprovals.org/2018/01/25/delafloxacin/
15 Deutetrabenazine
TEVA
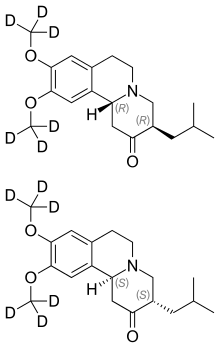

LINK……………https://newdrugapprovals.org/2015/08/15/sd-809-deutetrabenazine-nda-submitted-by-teva/
| Austedo | FDA 4/3/2017 | For the treatment of chorea associated with Huntington’s disease Drug Trials Snapshot Chemistry Review(s) (PDF) |
16 DUPILUMAB
SANOFI/REGENERON


| Dupixent | FDA | 3/28/2017 | To treat adults with moderate-to-severe eczema (atopic dermatitis) Press Release Drug Trials Snapshot |
LINK…….https://newdrugapprovals.org/2017/03/29/fda-approves-new-eczema-drug-dupixent-dupilumab/
17 DURVALUMAB
ASTRA ZENECA
durvalumab FDA 5/1/2017To treat patients with locally advanced or metastatic urothelial carcinoma
Web Post
Drug Trials Snapshot
18 EDAVARONE

MITSUBISHI TANABE
| Radicava | FDA 5/5/2017 | To treat patients with amyotrophic lateral sclerosis (ALS) Press Release Drug Trials Snapshot |


19 EMICIZUMAB
ROCHE

| Hemlibra | emicizumab | FDA 11/16/2017 | To prevent or reduce the frequency of bleeding episodes in adult and pediatric patients with hemophilia A who have developed antibodies called Factor VIII (FVIII) inhibitors. Press Release Drug Trials Snapshot |

20 Enasidenib


| Idhifa | FDA
8/1/2017 |
To treat relapsed or refractory acute myeloid leukemia Press Release Drug Trials Snapshot |
21 Ertugliflozin
MERCK
| Steglatro | ertugliflozin | FDA
12/19/2017 |
To improve glycemic control in adults with type 2 diabetes mellitus Drug Trials Snapshot |
LINK https://newdrugapprovals.org/2014/02/10/ertugliflozin/

22 ETELCALCETIDE
Amgen
| Parsabiv | FDA APPROVED
2/7/2017 |
To treat secondary hyperparathyroidism in adult patients with chronic kidney disease undergoing dialysis Drug Trials SnapshotSYNTHESIS LINK……..https://cen.acs.org/articles/96/i4/the-year-in-new-drugs-2018.html |
SYNTHESIS LINK……..https://cen.acs.org/articles/96/i4/the-year-in-new-drugs-2018.html
23 GLECAPREVIR
ABBVIE

| Mavyret | glecaprevir and pibrentasvir | FDA 8/3/2017 | To treat adults with chronic hepatitis C virus Press Release Drug Trials Snapshot |
LINK https://newdrugapprovals.org/2016/10/05/glecaprevir-abt-493/
24 GUSELKUMAB
JOHNSON AND JOHNSON
guselkumab
FDA 7/13/2017
For the treatment of adult patients with moderate-to-severe plaque psoriasis
Drug Trials Snapshot


25 Inotuzumab ozogamicin
PFIZER



| Besponsa | FDA
8/17/2017 |
To treat adults with relapsed or refractory acute lymphoblastic leukemia Press Release Drug Trials Snapshot |
26 LATANOPROSTENE
VALEANT
latanoprostene bunod ophthalmic solution
FDA 11/2/2017
To treat intraocular pressure in patients with open-angle glaucoma or ocular hypertension.
Drug Trials Snapshot
27 LETERMOVIR
MERCK
| Prevymis | FDA 11/8/2017 | To prevent infection after bone marrow transplant Drug Trials Snapshot |
LINK https://newdrugapprovals.org/2016/05/16/letermovir-aic-246/
28 Macimorelin acetate
AETERNA ZENTARIS
| Macrilen | macimorelin acetate | FDA
12/20/2017 |
For the diagnosis of adult growth hormone deficiency Drug Trials Snapshot |
29 MEROPENEM

30 MIDOSTAURIN
NOVARTIS
- Chemistry Review(s) (PDF)

| Rydapt | FDA
4/28/2017 |
To treat acute myeloid leukemia Press Release Drug Trials Snapshot |
31 Naldemedine
FDA 3/23/2017, Symproic, For the treatment of opioid-induced constipation


32 NERATINIB MALEATE
PUMA BIOTECH
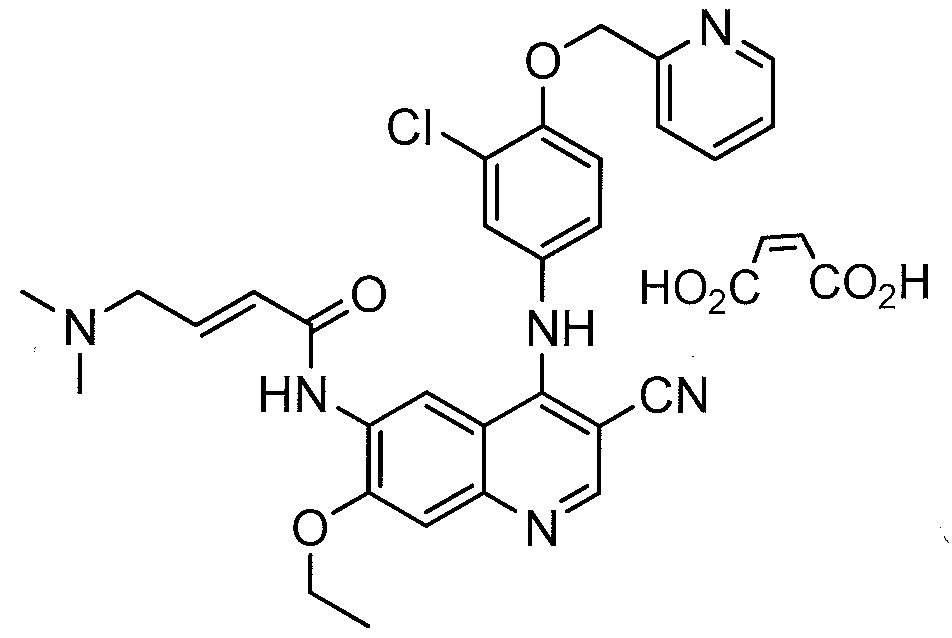


| Nerlynx | FDA | 7/17/2017 | To reduce the risk of breast cancer returning Press Release Drug Trials Snapshot |
33 NETARSUDIL
| Rhopressa | FDA APPROVED
12/18/2017 |
To treat glaucoma or ocular hypertension |


LINK……https://newdrugapprovals.org/2018/01/29/netarsudil/
34 NIRAPARIB
TESARO
| Zejula | FDA | 3/27/2017 | For the maintenance treatment for recurrent epithelial ovarian, fallopian tube or primary peritoneal cancers Press Release Drug Trials Snapshot |

![]()

LINK…https://newdrugapprovals.org/2016/12/22/niraparib-mk-4827/
35 OCRELIZUMAB
ROCHE
| Ocrevus | FDA | 3/28/2017 | To treat patients with relapsing and primary progressive forms of multiple sclerosis Press Release Drug Trials Snapshot |


36 OZENOXACIN
MEDIMETRIX

| Xepi | ozenoxacin | FDA
12/11/2017 |
To treat impetigo Drug Trials Snapshot |
37 Pibrentasvir
ABBVIE

| Mavyret | glecaprevir and pibrentasvir | FDA 8/3/2017 | To treat adults with chronic hepatitis C virus Press Release Drug Trials Snapshot |
LINK https://newdrugapprovals.org/2016/06/08/abt-530-pibrentasvir/
38 PLECANATIDE
Plecanatide 普卡那肽 ليكاناتيد плеканатид
SYNERGY PHARMA


| Trulance | FDA APPROVED
1/19/2017 |
To treat Chronic Idiopathic Constipation (CIC) in adult patients. Press Release Drug Trials Snapshot |
39 RIBOCICLIB
NOVARTIS
Structure..link for correct structure
| Kisqali | FDA 3/13/2017 | To treat postmenopausal women with a type of advanced breast cancer Drug Trials Snapshot |

LINK https://newdrugapprovals.org/2015/10/19/ribociclib/
40 SARILUMAB
SANOFI /REGENERON

| Kevzara | sarilumab | FDA 5/22/2017 | To treat adult rheumatoid arthritis Drug Trials Snapshot |
LINK https://newdrugapprovals.org/2013/11/25/late-stage-success-for-sanofiregeneron-ra-drug-sarilumab/


41 SECNIDAZOLE
SYMBIOMIX

| Solosec | FDA | 9/15/2017 | To treat bacterial vaginosis Drug Trials Snapshot |

42 SAFINAMIDE
NEWRON PHARMA



- Chemistry Review(s) (PDF) for correct structure
| Xadago | FDA 3/21/2017 | To treat Parkinson’s disease Press Release Drug Trials Snapshot |
43 Semaglutide
NOVO NORDISK
| Ozempic | semaglutide | FDA
12/5/2017 |
To improve glycemic control in adults with type 2 diabetes mellitus Drug Trials Snapshot |
LINK https://newdrugapprovals.org/2013/07/22/a-survey-of-promising-late-stage-diabetes-drugs/

44 SOFOSBUVIR
45 TELOTRISTAT ETHYL
LEXICON
| Xermelo | FDA
2/28/2017 |
To treat carcinoid syndrome diarrhea Press Release Drug Trials Snapshot |


46 VABORBACTAM
THE MEDICINES CO

| Vabomere | meropenem and vaborbactam | FDA
8/29/2017 |
To treat adults with complicated urinary tract infections Press Release Drug Trials Snapshot |

47 VALBENAZINE
NEUROCRINE
- Chemistry Review(s) (PDF)
| Ingrezza | FDA
4/11/2017 |
To treat adults with tardive dyskinesia Press Release Drug Trials Snapshot |
48 Vestronidase alfa-vjbk
ULTRAGENYX
| Mepsevii | vestronidase alfa-vjbk | FDA 11/15/2017 | To treat pediatric and adult patients with an inherited metabolic condition called mucopolysaccharidosis type VII (MPS VII), also known as Sly syndrome. Press Release Drug Trials Snapshot |

49 VELPATASVIR
50 VOXILAPREVIR
GILEAD
| Vosevi | sofosbuvir, velpatasvir and voxilaprevir | FDA 7/18/2017 | To treat adults with chronic hepatitis C virus Press Release Drug Trials Snapshot |
SECTION B; EMA approvals
European Medicines Agency’s – Human medicines: Highlights of 2017
Advances in medicines authorizations are essential for public health as they have the potential to improve treatment of diseases. In 2017, EMA recommended 92 medicines for marketing authorization. Of these, 35 had a new active substance, which has never been authorized in the European Union (EU) before. Many of these medicines represent a significant improvement in their therapeutic areas; they include medicines for children, for rare diseases and advanced therapies42. Amongst the 35 new active substances (NAS) that EMA recommended, 11 were new drugs and biologics to treat cancer, 05 to treat neurological disorders, 04 for infectious diseases, 04 for immunology/rheumatology, 03 for endocrinology, 02 each for Uro-nephrology, haematology, and dermatology, 01 for Pneumonology, and 01 for hepatology/gastroenterology class of drugs.
EUROPE
| 2017/11/16 | Niraparib | Zejula | Tesaro UK Limited | O | NME | ||
| 2017/11/10 | Adalimumab | Cyltezo | Boehringer Ingelheim International GmbH | B | |||
| 2017/11/10 | Miglustat | Miglustat Gen.Orph | Gen.Orph | G | |||
| 2017/11/10 | Ritonavir | Ritonavir Mylan | MYLAN S.A.S | G | |||
| 2017/11/10 | Padeliporfin | Tookad | STEBA Biotech S.A | ||||
| 2017/11/10 | Guselkumab | Tremfya | Janssen-Cilag International N.V. | BLA | |||
| 2017/9/27 | Dupilumab | Dupixent | sanofi-aventis groupe | BLA | |||
| 2017/9/21 | Darunavir / Cobicistat / Emtricitabine / Tenofovir alafenamide | Symtuza | Janssen-Cilag International N.V. | ||||
| 2017/9/21 | Atezolizumab | Tecentriq | Roche Registration Limited | BLA | |||
| 2017/9/18 | Avelumab | Bavencio | Merck Serono Europe Limited | O | BLA | ||
| 2017/9/18 | Entecavir | Entecavir Mylan | Mylan S.A.S | G | |||
| 2017/9/18 | Lacosamide | Lacosamide Accord | Accord Healthcare Ltd | G | |||
| 2017/9/18 | Midostaurin | Rydapt | Novartis Europharm Ltd | O | NME | ||
| 2017/9/18 | Telotristat ethyl | Xermelo | Ipsen Pharma | O | NME | ||
| 2017/9/5 | Trientine | Cuprior | GMP-Orphan SA | ||||
| 2017/9/5 | Efavirenz / Emtricitabine / Tenofovir disoproxil | Efavirenz/Emtricitabine/Tenofovir disoproxil Mylan | Mylan S.A.S | G | |||
| 2017/8/24 | Tivozanib hydrochloride monohydrate | Fotivda | EUSA Pharma (UK) Limited | NME | |||
| 2017/8/24 | Adalimumab | Imraldi | Samsung Bioepis UK Limited (SBUK) | B | |||
| 2017/8/24 | Nitisinone | Nitisinone MDK (previously Nitisinone MendeliKABS) | MendeliKABS Europe Ltd | G | |||
| 2017/8/22 | Ribociclib | Kisqali | Novartis Europharm Ltd | NME | |||
| 2017/8/22 | Cladribine | Mavenclad | Merck Serono Europe Limited | ||||
| 2017/7/26 | Glecaprevir / Pibrentasvir | Maviret | AbbVie Limited | NME | |||
| 2017/7/26 | Sofosbuvir / Velpatasvir / Voxilaprevi | Vosevi | Gilead Sciences International Ltd | NME | |||
| 2017/7/19 | Insulin lispro | Insulin lispro Sanofi | sanofi-aventis groupe | B | |||
| 2017/7/19 | Patiromer sorbitex calcium | Veltassa | Vifor Fresenius Medical Care Renal Pharma France | NME | |||
| 2017/7/17 | Efavirenz / Emtricitabine / Tenofovir disoproxil | Efavirenz/Emtricitabine/Tenofovir disoproxil Zentiva | Zentiva k.s. | G | |||
| 2017/7/17 | Brodalumab | Kyntheum | LEO Pharma A/S | BLA | |||
| 2017/7/17 | beclometasone / formoterol / glycopyrronium bromide | Trimbow | Chiesi Farmaceutici S.p.A. | ||||
| 2017/7/13 | Rituximab | Blitzima | Celltrion Healthcare Hungary Kft. | B | |||
| 2017/7/13 | Cariprazine | Reagila | Gedeon Richter | ||||
| 2017/7/10 | Spheroids of human autologous matrix-associated chondrocytes | Spherox | CO.DON AG | ||||
| 2017/7/6 | Cenegermin | Oxervate | Dompe farmaceutici s.p.a. | O | BLA | ||
| 2017/6/29 | Inotuzumab ozogamicin | Besponsa | Pfizer Limited | O | BLA | ||
| 2017/6/23 | Etanercept | Erelzi | Sandoz GmbH | ||||
| 2017/6/23 | Sarilumab | Kevzara | Sanofi-Aventis Groupe | NME | |||
| 2017/6/23 | Dimethyl fumarate | Skilarence | Almirall S.A | ||||
| 2017/6/23 | Carglumic acid | Ucedane | Lucane Pharma | G | |||
| 2017/6/15 | Rituximab | Rixathon, Riximyo B | Sandoz GmbH | ||||
| 2017/6/2 | Pentosan polysulfate sodium | Elmiron | bene-Arzneimittel GmbH | ||||
| 2017/6/2 | Nonacog beta pegol | Refixia | Novo Nordisk A/S | BLA | |||
| 2017/5/30 | Cerliponase alfa | Brineura | BioMarin International Limited | O E | BLA | ||
| 2017/5/30 | Nusinersen | Spinraza | Biogen Idec Ltd | O | NME | ||
| 2017/5/24 | Meningococcal group b vaccine (recombinant, adsorbed) | Trumenba | Pfizer Limited | ||||
| 2017/5/22 | Ivabradine | Ivabradine Accord | Accord Healthcare Ltd | G | |||
| 2017/5/8 | Dinutuximab beta | Dinutuximab beta Apeiron | Apeiron Biologics AG | O E | |||
| 2017/4/28 | Emtricitabine – tenofovir disoproxil mixt | Emtricitabine/Tenofovir disoproxil Krka d.d. | KRKA, d.d., Novo mesto | G | |||
| 2017/4/24 | Parathyroid hormone | Natpar | Shire Pharmaceuticals Ireland Ltd | O C | BLA | ||
| 2017/4/20 | Edoxaban | Roteas | Daiichi Sankyo Europe GmbH | ||||
| 2017/3/22 | Tofacitinib citrate | Xeljanz | Pfizer Limited | NME | |||
| 2017/3/20 | Umeclidinium | Rolufta | GlaxoSmithKline Trading Services Limited | ||||
| 2017/3/3 | Chlormethine | Ledaga | Actelion Registration Ltd. | O | |||
| 2017/2/27 | Pregabalin | Pregabalin Zentiva | Zentiva k.s. | G | |||
| 2017/2/17 | Rituximab | Truxima | Celltrion Healthcare Hungary Kft. | B | |||
| 2017/2/13 | Etanercept | Lifmior | Pfizer Limited | ||||
| 2017/2/13 | Baricitinib | Olumiant | Eli Lilly Nederland B.V. | NME | |||
| 2017/1/19 | Mercaptamine | Cystadrops | Orphan Europe S.A.R.L. | O | |||
| 2017/1/18 | Bezlotoxumab | Zinplava | Merck Sharp & Dohme Limited | NME | |||
| 2017/1/11 | Teriparatide | Movymia | STADA Arzneimittel AG | B | |||
| 2017/1/11 | Insulin glargine / lixisenatide | Suliqua | Sanofi-Aventis Groupe | ||||
| 2017/1/9 | Insulin aspart | Fiasp | Novo Nordisk A/S | ||||
| 2017/1/9 | Tadalafil | Tadalafil | Mylan S.A.S | G | |||
| 2017/1/9 | Tenofovir alafenamide | Vemlidy | Gilead Sciences International Ltd | ||||
| 2017/1/4 | Lonoctocog alfa | Afstyla | CSL Behring GmbH | BLA | |||
| 2017/1/4 | Darunavir | Darunavir Mylan | Mylan S.A.S. | G | |||
| 2017/1/4 | Insulin glargine | Lusduna | Merck Sharp & Dohme Limited | B | |||
| 2017/1/4 | Teriparatide | Terrosa | Gedeon Richter Plc. | B |
SECTION B; EMA Approvals
Combined drugs USFDA+EMA +PMDA list are listed below. trying to simplify search
1 Abaloparatide USFDA
2 Abemaciclib USFDA
3 ACALABRUTINIB USFDA
3A ALOFISEL EMA
3B AMENAMEVIR JAPAN
4 ANGIOTENSIN II USFDA
4A Atezolizumab EMA
5 AVELUMAB USFDA+EMA
6 BENRALIZUMAB USFDA+EMA
6A BARICITINIB JAPAN
7 BENZNIDAZOLE USFDA
8 BETRIXABAN USFDA
9 BRIGATINIB USFDA
10 BRODALUMAB USFDA+EMA
10A BUROSUMAB EMA
10B CARIPRAZINE HYDROCHLORIDE EMA
11 CERLIPONASE ALPA USFDA+EMA
12 COPANLISIB USFDA
13 DEFLAZACORT USFDA
14 Delafloxacin USFDA
15 Deutetrabenazine USFDA
16DUPILUMAB USFDA+EMA
17 DURVALUMAB USFDA
18 EDAVARONE USFDA
19 EMICIZUMAB USFDA
20 Enasidenib USFDA
21 ERTUGLIFLOZIN USFDA
22 ETELCALCETIDE USFDA
22A FORODESINE JAPAN
22B FLUCICLOVINE EMA
23 GLECAPREVIR USFDA+EMA
24 GUSELKUMAB USFDA+EMA
25 INOTUZUMAB OZOGAMICIN USFDA+EMA
26 LATANOPROSTENE USFDA
27 LETERMOVIR USFDA+EMA
27A Utetium lu 177 dotatate EMA
28 MACIMORELIN ACETATE USFDA
29 MEROPENEM USFDA
30 MIDOSTAURIN USFDA+EMA
31 NALDEMEDINE USFDA
32 NERATINIB USFDA
33 NETARSUDIL USFDA
34A NONACOG EMA
34B NUCINERSEN EMA +Japan
35 Ocrelizumab USFDA+EMA
35A OXERVATE EMA
36 OZENOXACIN USFDA
36A PATIROMER EMA
36B PADELIPORFIN EMA
36C PEMAFIBRATE JAPAN
37 PIBRENTASVIR USFDA+EMA
38 PLECANATIDE USFDA
39A ROLAPITANT EMA
39BRURLOCTOCOG EMA
40 SARILUMAB USFDA+EMA
41 SECNIDAZOLE USFDA
42 SAFINAMIDE USFDA
43 SEMAGLUTIDE USFDA+EMA
43A SODIUM ZIRCONIUM CYCLOCYLICATE EMA
44 SOFOSBUVIR USFDA+EMA
44A SPHEROX EMA
45 TELOTRISTAT ETHYL USFDA+EMA
45A TIVOZANIB EMA
45B TOFACITINIB EMA
45C TRUMENBA EMA
46 VABORBACTAM USFDA
47 VALBENAZINE USFDA
48 VESTRONIDASE ALFA-VJBK USFDA
49 VELPATASVIR USFDA+EMA
50 VOXILAPREVIR USFDA+EMA
Drugs EMA list missed out in usfda list
3A ALOFISEL
link………https://newdrugapprovals.org/2018/03/02/alofisel-darvadstrocel-cx-601/
4A Atezolizumab
WILL BE UPDATED
10A BUROSUMAB
WILL BE UPDATED
10B CARIPRAZINE HYDROCHLORIDE
WILL BE UPDATED
22B FLUCICLOVINE

SEE EMA
| Axumin : EPAR – Summary for the public | EN = English | 06/07/2017 |
27A Lutetium lu 177 dotatate
WILL BE UPDATED
34A NONACOG
WILL BE UPDATED
34B NUCINERSEN
EMA AND JAPAN 2017 APPROVED


35A OXERVATE
WILL BE UPDATED
36A PATIROMER
WILL BE UPDATED
36B PADELIPORFIN

| NAME | Tookad |
|---|---|
| AGENCY PRODUCT NUMBER | EMEA/H/C/004182 |
| ACTIVE SUBSTANCE | padeliporfin di-potassium |
| INTERNATIONAL NON-PROPRIETARY NAME(INN) OR COMMON NAME | padeliporfin |
| THERAPEUTIC AREA | Prostatic Neoplasms |
| ANATOMICAL THERAPEUTIC CHEMICAL (ATC) CODE | L01XD07 |
| ADDITIONAL MONITORING | This medicine is under additional monitoring. This means that it is being monitored even more intensively than other medicines. For more information, see medicines under additional monitoring. |
| MARKETING-AUTHORISATION HOLDER | STEBA Biotech S.A |
|---|---|
| REVISION | 0 |
| DATE OF ISSUE OF MARKETING AUTHORISATION VALID THROUGHOUT THE EUROPEAN UNION | 10/11/2017 |
Contact address:
STEBA Biotech S.A
7 place du theatre
L-2613 Luxembourg
Luxembourg
38A PRALATREXATE

Japan approved 2017
| 2017/7/3 | PMDA | JAPAN | Pralatrexate | Difolta | Mundipharma | NME |
39A ROLAPITANT
WILL BE UPDATED
39B RURLOCTOCOG
WILL BE UPDATED
43A SODIUM ZIRCONIUM
WILL BE UPDATED
44A SPHEROX
WILL BE UPDATED
45A TIVOZANIB

Pharmacotherapeutic group
Antineoplastic agents
Therapeutic indication
Fotivda is indicated for the first line treatment of adult patients with advanced renal cell carcinoma (RCC) and for adult patients who are VEGFR and mTOR pathway inhibitor-naïve following disease progression after one prior treatment with cytokine therapy for advanced RCC.
Treatment of advanced renal cell carcinoma
Fotivda : EPAR -Product Information

Tivozanib is synthesized in three main steps using well defined starting materials with acceptable
specifications.
Adequate in-process controls are applied during the synthesis. The specifications and control methods for
intermediate products, starting materials and reagents have been presented. The critical process
parameters are duly justified, methodology is presented and control is adequate.
The characterisation of the active substance and its impurities are in accordance with the EU guideline on
chemistry of new active substances. Potential and actual impurities were well discussed with regards to
their origin and characterised.
The active substance is packaged in a low-density polyethylene (LDPE) bag which complies with the EC
directive 2002/72/EC and EC 10/2011 as amended.
Product details
| Name | Fotivda |
|---|---|
| Agency product number | EMEA/H/C/004131 |
| Active substance | tivozanib |
| International non-proprietary name(INN) or common name | tivozanib hydrochloride monohydrate |
| Therapeutic area | Carcinoma, Renal Cell |
| Anatomical therapeutic chemical (ATC) code | L01XE |
Publication details
| Marketing-authorisation holder | EUSA Pharma (UK) Limited |
|---|---|
| Revision | 0 |
| Date of issue of marketing authorisation valid throughout the European Union | 24/08/2017 |
Contact address:
EUSA Pharma (UK) Limited
Breakspear Park, Breakspear Way
Hemel Hempstead, HP2 4TZ
United Kingdom
45B TOFACITINIB
WILL BE UPDATED
45C TRUMENBA
WILL BE UPDATED
SECTION C JAPANFORODOS
SECTION C New Drugs JAPAN
https://www.pmda.go.jp/english/review-services/reviews/approved-information/drugs/0002.html
JAPAN 2017
| 2017/9/27 | Avelumab (genetical recombination) | Bavencio | Merck Serono | BLA |
| 2017/9/27 | Glecaprevir – pibrentasvir mixt | Maviret | Abbvie | NME |
| 2017/9/27 | Daratumumab (genetical recombination) | Darzalex | Janssen Pharmaceutical | BLA |
| 2017/9/27 | Belimumab (genetical recombination) | Benlysta | GlaxoSmithKline | BLA |
| 2017/9/27 | Bezlotoxumab (genetical recombination) | Zinplava | MDS | BLA |
| 2017/9/27 | Palbociclib | Ibrance | Pfizer | NME |
| 2017/9/27 | Lonoctocog alfa (genetical recombination) | Afstyla | CSL Behring | BLA |
| 2017/9/27 | Rupatadine fumarate | Rupafin | Teikoku seiyaku | NME |
| 2017/9/27 | Sarilumab (genetical receombination) | Kevzara | Sanofi | BLA |
| 2017/9/27 | Flutemetamol (18F) | Vizamyl | Nihon Medi-Physics | NME |
| 2017/7/3 | Nusinersen sodium | Spinraza | Biogen Japan | |
| 2017/7/3 | Romidepsin | Istodax | Celgene | NME |
| 2017/7/3 | Pralatrexate | Difolta | Mundipharma | NME |
| 2017/7/3 | Amenamevir | Amenalief | Maruho | NME |
| 2017/7/3 | Baricitinib | Olumiant | Lilly | NME |
| 2017/7/3 | Pemafibrate | Parmodia | Kowa | NME |
| 2017/3/30 | Human prothrombin complex, freeze-dried concentrated | Kcentra | CSL Behring | |
| 2017/3/30 | Ixazomib citrate | Ninlaro | Takeda | NME |
| 2017/3/30 | Forodesine hydrochloride | Mundesine | Mundipharma | |
| 2017/3/30 | Aflibercept beta (genetical recombination) | Zaltrap | Sanofi | |
| 2017/3/30 | Hydromorphone hydrochloride | Narusus, Narurapid | DaiichiSankyo-pp | |
| 2017/3/30 | Naldemedine tosylate | Symproic | Shionogi | NME |
| 2017/3/30 | Guanfacine hydrochloride | Intuniv | Shionogi |
3B AMENAMEVIR

Originally developed by Astellas, the drug was licensed to Maruho. Amenamevir treats herpes zoster by inhibiting the activity of the helicase-primer enzyme during viral DNA replication and blocking the virus’s proliferation.
Amenalief® is an oral film-coated tablet containing 200 mg of amenamevir per tablet. Recommended dose of 1 day, 400mg each time, after meals.
22A FORODESINE HYDROCHLORIDE

LINK https://newdrugapprovals.org/2018/03/06/forodesine-hydrochloride/
6A BARICITINIB JAPAN

Originally developed by Incyte, Baricitinib was later licensed to and for sale by Lilly under the trade name Olumiant®. Baricitinib is an irreversible inhibitor of Janus kinase 1 (JAK1) and Janus kinase 2 (JAK2). Olumiant® is approved for the treatment of mild to moderate rheumatoid arthritis in adult patients who are not responsive or intolerant to other anti-arthritic drugs. This product can be used alone or in combination with methotrexate.
Olumiant® is a film-coated tablet containing 2 mg or 4 mg per tablet. Recommended oral dose is 4mg daily, with meals or fasting food, you can take any time period.
2017/7/3PMDA Baricitinib Olumiant Lilly
36C PEMAFIBRATE

LINK https://newdrugapprovals.org/2016/04/24/pemafibrate/
SECTION D
CDSCO INDIA


http://www.cdsco.nic.in/forms/list.aspx?lid=2034&Id=11 http://www.cdsco.nic.in/forms/list.aspx?lid=2034&Id=11






KEEP WATCHING UNDER CONSTRUCTION AND WILL BE PASTED SOON………………………………………..
KEEP WATCHING UNDER CONSTRUCTION AND WILL BE PASTED SOON………………………………………..
KEEP WATCHING UNDER CONSTRUCTION AND WILL BE PASTED SOON………………………………………..
KEEP WATCHING UNDER CONSTRUCTION AND WILL BE PASTED SOON………………………………………..
REFERENCES
2 http://www.ema.europa.eu/docs/en_GB/document_library/Report/2018/01/WC500242079.pdf
“NEW DRUG APPROVALS” CATERS TO EDUCATION GLOBALLY, No commercial exploits are done or advertisements added by me. This is a compilation for educational purposes only. P.S. : The views expressed are my personal and in no-way suggest the views of the professional body or the company that I represent
 amcrasto@gmail.com
amcrasto@gmail.com
I , Dr A.M.Crasto is writing this blog to share the knowledge/views, after reading Scientific Journals/Articles/News Articles/Wikipedia. My views/comments are based on the results /conclusions by the authors(researchers). I do mention either the link or reference of the article(s) in my blog and hope those interested can read for details. I am briefly summarising the remarks or conclusions of the authors (researchers). If one believe that their intellectual property right /copyright is infringed by any content on this blog, please contact or leave message at below email address amcrasto@gmail.com. It will be removed ASAP
////////EMA APPROVALS, USFDA Approvals, ACALABRUTINIB, AVELUMAB, BETRIXABAN, BRODALUMAB, COPANLISIB, DEFLAZACORT, Delafloxacin, Deutetrabenazine, DUPILUMAB, ETELCALCETIDE, Naldemedine, NETARSUDIL, NIRAPARIB, Ocrelizumab, PLECANATIDE, RIBOCICLIB, SAFINAMIDE, TELOTRISTAT ETHYL, VALBENAZINE, CERLIPONASE, BRIGATINIB, MIDOSTAURIN, Abaloparatide, BENZNIDAZOLE, NERATINIB, inotuzumab ozogamicin, Enasidenib, LETERMOVIR, GLECAPREVIR, PIBRENTASVIR, VOXILAPREVIR, SOFOSBUVIR, EDAVARONE, abemaciclib, ANGIOTENSIN II, VESTRONIDASE, macimorelin acetate, ERTUGLIFLOZIN, SEMAGLUTIDE, EMICIZUMAB, eu 2017, fda 2017, BENRALIZUMAB, DURVALUMAB, GUSELKUMAB, LATANOPROSTENE, OZENOXACIN, SARILUMAB, SECNIDAZOLE, BENRALIZUMAB, TIVOZANIB, SARILUMAB, FLUCICLOVINE,
FDA approves first U.S. treatment benznidazole for Chagas disease
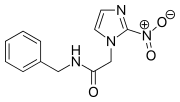
The U.S. Food and Drug Administration today granted accelerated approval to benznidazole for use in children ages 2 to 12 years old with Chagas disease. It is the first treatment approved in the United States for the treatment of Chagas disease.
Chagas disease, or American trypanosomiasis, is a parasitic infection caused by Trypanosoma cruzi and can be transmitted through different routes, including contact with the feces of a certain insect, blood transfusions, or from a mother to her child during pregnancy. After years of infection, the disease can cause serious heart illness, and it also can affect swallowing and digestion. While Chagas disease primarily affects people living in rural parts of Latin America, recent estimates are that there may be approximately 300,000 persons in the United States with Chagas disease.
“The FDA is committed to making available safe and effective therapeutic options to treat tropical diseases,” said Edward Cox, M.D., director of the Office of Antimicrobial Products in the FDA’s Center for Drug Evaluation and Research.
The safety and efficacy of benznidazole were established in two placebo-controlled clinical trials in pediatric patients 6 to 12 years old. In the first trial, approximately 60 percent of children treated with benznidazole had an antibody test change from positive to negative compared with approximately 14 percent of children who received a placebo. Results in the second trial were similar: Approximately 55 percent of children treated with benznidazole had an antibody test change from positive to negative compared with 5 percent who received a placebo. An additional study of the safety and pharmacokinetics (how the body absorbs, distributes and clears the drug) of benznidazole in pediatric patients 2 to 12 years of age provided information for dosing recommendations down to 2 years of age.
The most common adverse reactions in patients taking benznidazole were stomach pain, rash, decreased weight, headache, nausea, vomiting, abnormal white blood cell count, urticaria (hives), pruritus (itching) and decreased appetite. Benznidazole is associated with serious risks including serious skin reactions, nervous system effects and bone marrow depression. Based on findings from animal studies, benznidazole could cause fetal harm when administered to a pregnant woman.
Benznidazole was approved using the Accelerated Approval pathway. The Accelerated Approval pathway allows the FDA to approve drugs for serious conditions where there is unmet medical need and adequate and well-controlled trials establish that the drug has an effect on a surrogate endpoint that is reasonably likely to predict a clinical benefit to patients. Further study is required to verify and describe the anticipated clinical benefit of benznidazole.
The FDA granted benznidazole priority review and orphan product designation. These designations were granted because Chagas disease is a rare disease, and until now, there were no approved drugs for Chagas disease in the United States.
With this approval, benznidazole’s manufacturer, Chemo Research, S. L., is awarded a Tropical Disease Priority Review Voucher in accordance with a provision included in the Food and Drug Administration Amendments Act of 2007 that aims to encourage development of new drugs and biological products for the prevention and treatment of certain tropical diseases.
 |
|
| Clinical data | |
|---|---|
| Trade names | Rochagan, Radanil[1] |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Routes of administration |
by mouth |
| ATC code | |
| Pharmacokinetic data | |
| Bioavailability | High |
| Metabolism | Liver |
| Biological half-life | 12 hours |
| Excretion | Kidney and fecal |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| ECHA InfoCard | 100.153.448 |
| Chemical and physical data | |
| Formula | C12H12N4O3 |
| Molar mass | 260.249 g/mol |
| 3D model (JSmol) | |
| Melting point | 188.5 to 190 °C (371.3 to 374.0 °F) |
Benznidazole is an antiparasitic medication used in the treatment of Chagas disease.[2] While it is highly effective in early disease this decreases in those who have long term infection.[3] It is the first line treatment given its moderate side effects compared to nifurtimox.[1] It is taken by mouth.[2]
Side effects are fairly common. They include rash, numbness, fever, muscle pain, loss of appetite, and trouble sleeping.[4][5] Rare side effects include bone marrow suppression which can lead to low blood cell levels.[1][5] It is not recommended during pregnancy or in people with severe liver or kidney disease.[4][3]Benznidazole is in the nitroimidazole family of medication and works by the production of free radicals.[5][6]
Benznidazole came into medical use in 1971.[2] It is on the World Health Organization’s List of Essential Medicines, the most effective and safe medicines needed in a health system.[7] It is not commercially available in the United States, but can be obtained from the Centers of Disease Control.[2] As of 2012 Laboratório Farmacêutico do Estado de Pernambuco, a government run pharmaceutical company in Brazil was the only producer.[8]
Medical uses
Benznidazole has a significant activity during the acute phase of Chagas disease, with a therapeutical success rate up to 80%. Its curative capabilities during the chronic phase are, however, limited. Some studies have found parasitologic cure (a complete elimination of T. cruzi from the body) in pediatric and young patients during the early stage of the chronic phase, but overall failure rate in chronically infected individuals is typically above 80%.[6]
However, some studies indicate treatment with benznidazole during the chronic phase, even if incapable of producing parasitologic cure, because it reduces electrocardiographic changes and a delays worsening of the clinical condition of the patient.[6]
Benznidazole has proven to be effective in the treatment of reactivated T. cruzi infections caused by immunosuppression, such as in people with AIDS or in those under immunosuppressive therapy related to organ transplants.[6]
Children
Benznidazole can be used in children and infants, with the same 5–7 mg/kg per day weight-based dosing regimen that is used to treat adult infections.[9] Children are found to be at a lower risk of adverse events compared to adults, possibly due to increased hepatic clearance of the drug. The most prevalent adverse effects in children were found to be gastrointestinal, dermatologic, and neurologic in nature. However, the incidence of severe dermatologic and neurologic adverse events is lower in the pediatric population compared to adults.[10]
Pregnant women
Studies in animals have shown that benznidazole can cross the placenta.[11] Due to its potential for teratogenicity, use of benznidazole in pregnancy is not recommended.[9]
Side effects
Side effects tend to be common and occur more frequently with increased age.[12] The most common adverse reactions associated with benznidazole are allergic dermatitis and peripheral neuropathy.[1] It is reported that up to 30% of people will experience dermatitis when starting treatment.[11][13] Benznidazole may cause photosensitization of the skin, resulting in rashes.[1] Rashes usually appear within the first 2 weeks of treatment and resolve over time.[13] In rare instances, skin hypersensitivity can result in exfoliative skin eruptions, edema, and fever.[13] Peripheral neuropathy may occur later on in the treatment course and is dose dependent.[1] It is not permanent, but takes time to resolve.[13]
Other adverse reactions include anorexia, weight loss, nausea, vomiting, insomnia, and dysguesia, and bone marrow suppression.[1] Gastrointestinal symptoms usually occur during the initial stages of treatment and resolves over time.[13] Bone marrow suppression has been linked to the cumulative dose exposure.[13]
Contraindications
Benznidazole should not be used in people with severe liver and/or kidney disease.[12] Pregnant women should not use benznidazole because it can cross the placenta and cause teratogenicity.[11]
Pharmacology
Mechanism of action
Benznidazole is a nitroimidazole antiparasitic with good activity against acute infection with Trypanosoma cruzi, commonly referred to as Chagas disease.[11] Like other nitroimidazoles, benznidazole’s main mechanism of action is to generate radical species which can damage the parasite’s DNA or cellular machinery.[14] The mechanism by which nitroimidazoles do this seems to depend on whether or not oxygen is present.[15] This is particularly relevant in the case of Trypanosoma species, which are considered facultative anaerobes.[16]
Under anaerobic conditions, the nitro group of nitroimidazoles is believed to be reduced by the pyruvate:ferredoxin oxidoreductase complex to create a reactive nitro radical species.[14] The nitro radical can then either engage in other redox reactions directly or spontaneously give rise to a nitrite ion and imidazole radical instead.[15] The initial reduction takes place because nitroimidazoles are better electron acceptors for ferredoxin than the natural substrates.[14] In mammals, the principal mediators of electron transport are NAD+/NADH and NADP+/NADPH, which have a more positive reduction potential and so will not reduce nitroimidazoles to the radical form.[14] This limits the spectrum of activity of nitroimidazoles so that host cells and DNA are not also damaged. This mechanism has been well-established for 5-nitroimidazoles such as metronidazole, but it is unclear if the same mechanism can be expanded to 2-nitroimidazoles (including benznidazole).[15]
In the presence of oxygen, by contrast, any radical nitro compounds produced will be rapidly oxidized by molecular oxygen, yielding the original nitroimidazole compound and a superoxide anion in a process known as “futile cycling“.[14] In these cases, the generation of superoxide is believed to give rise to other reactive oxygen species.[15] The degree of toxicity or mutagenicity produced by these oxygen radicals depends on cells’ ability to detoxify superoxide radicals and other reactive oxygen species.[15] In mammals, these radicals can be converted safely to hydrogen peroxide, meaning benznidazole has very limited direct toxicity to human cells.[15] In Trypanosoma species, however, there is a reduced capacity to detoxify these radicals, which results in damage to the parasite’s cellular machinery.[15]
Pharmacokinetics
Oral benznidazole has a bioavailability of 92%, with a peak concentration time of 3–4 hours after administration.[17] 5% of the parent drug is excreted unchanged in the urine, which implies that clearance of benznidazole is mainly through metabolism by the liver.[18] Its elimination half-life is 10.5-13.6 hours.[17]
Interactions
Benznidazole and other nitroimidazoles have been shown to decrease the rate of clearance of 5-fluorouracil (including 5-fluorouracil produced from its prodrugs capecitabine, doxifluridine, and tegafur).[19]While co-administration of any of these drugs with benznidazole is not contraindicated, monitoring for 5-fluorouracil toxicity is recommended in the event they are used together.[20]
The GLP-1 receptor agonist lixisenatide may slow down the absorption and activity of benznidazole, presumably due to delayed gastric emptying.[21]
Because nitroimidazoles can kill Vibrio cholerae cells, use is not recommended within 14 days of receiving a live cholera vaccine.[22]
Alcohol consumption can cause a disulfiram like reaction with benznidazole.[1]
References
- ^ Jump up to:a b c d e f g h Bern, Caryn; Montgomery, Susan P.; Herwaldt, Barbara L.; Rassi, Anis; Marin-Neto, Jose Antonio; Dantas, Roberto O.; Maguire, James H.; Acquatella, Harry; Morillo, Carlos (2007-11-14). “Evaluation and Treatment of Chagas Disease in the United States: A Systematic Review”. JAMA. 298 (18): 2171–81. ISSN 0098-7484. PMID 18000201. doi:10.1001/jama.298.18.2171.
- ^ Jump up to:a b c d “Our Formulary | Infectious Diseases Laboratories | CDC”. http://www.cdc.gov. 22 September 2016. Retrieved 7 December2016.
- ^ Jump up to:a b “Chagas disease”. World Health Organization. March 2016. Retrieved 7 December 2016.
- ^ Jump up to:a b Prevention, CDC – Centers for Disease Control and. “CDC – Chagas Disease – Resources for Health Professionals – Antiparasitic Treatment”. http://www.cdc.gov. Retrieved 2016-11-05.
- ^ Jump up to:a b c Castro, José A.; de Mecca, Maria Montalto; Bartel, Laura C. (2006-08-01). “Toxic side effects of drugs used to treat Chagas’ disease (American trypanosomiasis)”. Human & Experimental Toxicology. 25 (8): 471–479. ISSN 0960-3271. PMID 16937919. doi:10.1191/0960327106het653oa.
- ^ Jump up to:a b c d Urbina, Julio A. “Nuevas drogas para el tratamiento etiológico de la Enfermedad de Chagas” (in Spanish). Retrieved March 24, 2012.
- Jump up^ “WHO Model List of Essential Medicines (19th List)” (PDF). World Health Organization. April 2015. Retrieved 8 December 2016.
- Jump up^ “Treatment for Chagas: Enter Supplier Number Two | End the Neglect”. endtheneglect.org. 21 March 2012. Retrieved 7 December 2016.
- ^ Jump up to:a b Carlier, Yves; Torrico, Faustino; Sosa-Estani, Sergio; Russomando, Graciela; Luquetti, Alejandro; Freilij, Hector; Vinas, Pedro Albajar (2011-10-25). “Congenital Chagas Disease: Recommendations for Diagnosis, Treatment and Control of Newborns, Siblings and Pregnant Women”. PLOS Negl Trop Dis. 5 (10): e1250. ISSN 1935-2735. PMC 3201907
 . PMID 22039554. doi:10.1371/journal.pntd.0001250.
. PMID 22039554. doi:10.1371/journal.pntd.0001250. - Jump up^ Altcheh, Jaime; Moscatelli, Guillermo; Moroni, Samanta; Garcia-Bournissen, Facundo; Freilij, Hector (2011-01-01). “Adverse Events After the Use of Benznidazole in Infants and Children With Chagas Disease”. Pediatrics. 127 (1): e212–e218. ISSN 0031-4005. PMID 21173000. doi:10.1542/peds.2010-1172.
- ^ Jump up to:a b c d Pérez-Molina, José A.; Pérez-Ayala, Ana; Moreno, Santiago; Fernández-González, M. Carmen; Zamora, Javier; López-Velez, Rogelio (2009-12-01). “Use of benznidazole to treat chronic Chagas’ disease: a systematic review with a meta-analysis”. Journal of Antimicrobial Chemotherapy. 64 (6): 1139–1147. ISSN 0305-7453. PMID 19819909. doi:10.1093/jac/dkp357.
- ^ Jump up to:a b Prevention, CDC – Centers for Disease Control and. “CDC – Chagas Disease – Resources for Health Professionals – Antiparasitic Treatment”. http://www.cdc.gov. Retrieved 2016-11-07.
- ^ Jump up to:a b c d e f Grayson, M. Lindsay; Crowe, Suzanne M.; McCarthy, James S.; Mills, John; Mouton, Johan W.; Norrby, S. Ragnar; Paterson, David L.; Pfaller, Michael A. (2010-10-29). Kucers’ The Use of Antibiotics Sixth Edition: A Clinical Review of Antibacterial, Antifungal and Antiviral Drugs. CRC Press. ISBN 9781444147520.
- ^ Jump up to:a b c d e Edwards, David I (1993). “Nitroimidazole drugs – action and resistance mechanisms. I. Mechanism of action”. Journal of Antimicrobial Chemotherapy. 31: 9–20. doi:10.1093/jac/31.1.9.
- ^ Jump up to:a b c d e f g Eller, Gernot. “Synthetic Nitroimidazoles: Biological Activities and Mutagenicity Relationships”. Scientia Pharmaceutica. 77: 497–520. doi:10.3797/scipharm.0907-14.
- Jump up^ Cheng, Thomas C. (1986). General Parasitology. Orlando, Florida: Academic Press. p. 140. ISBN 0-12-170755-5.
- ^ Jump up to:a b Raaflaub, J; Ziegler, WH (1979). “Single-dose pharmacokinetics of the trypanosomicide benznidazole in man”. Arzneimittelforschung. 29 (10): 1611–1614.
- Jump up^ Workman, P.; White, R. A.; Walton, M. I.; Owen, L. N.; Twentyman, P. R. (1984-09-01). “Preclinical pharmacokinetics of benznidazole.”. British Journal of Cancer. 50 (3): 291–303. ISSN 0007-0920. PMC 1976805
 . PMID 6466543. doi:10.1038/bjc.1984.176.
. PMID 6466543. doi:10.1038/bjc.1984.176. - Jump up^ Product Information: Teysuno oral capsules, tegafur gimeracil oteracil oral capsules. Nordic Group BV (per EMA), Hoofddorp, The Netherlands, 2012.
- Jump up^ Product Information: TINDAMAX(R) oral tablets, tinidazole oral tablets. Mission Pharmacal Company, San Antonio, TX, 2007.
- Jump up^ Product Information: ADLYXIN(TM) subcutaneous injection, lixisenatide subcutaneous injection. sanofi-aventis US LLC (per manufacturer), Bridgewater, NJ, 2016.
- Jump up^ Product Information: VAXCHORA(TM) oral suspension, cholera vaccine live oral suspension. PaxVax Inc (per manufacturer), Redwood City, CA, 2016.
External links
- “Rochagan [Patient Information]” (PDF) (in Portuguese). Hoffmann-La Roche. Retrieved 2006-11-27.
////////////benznidazole, Chemo Research, Tropical Disease Priority Review Voucher, Chagas disease, rare disease, FDA 2017