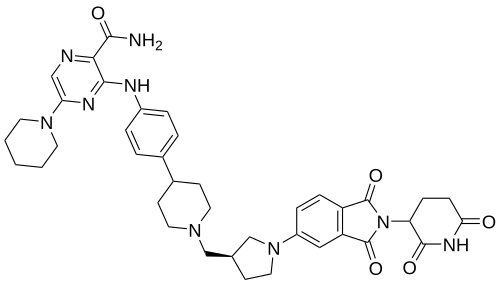
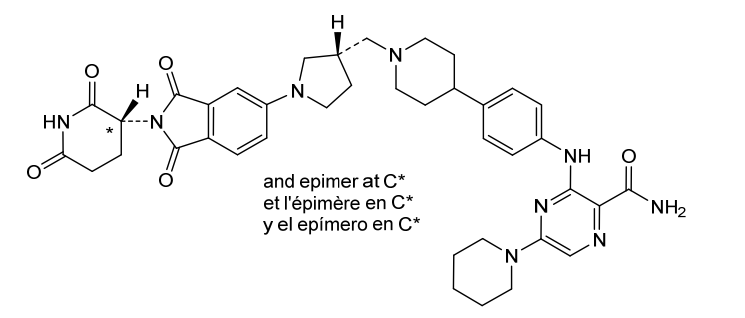
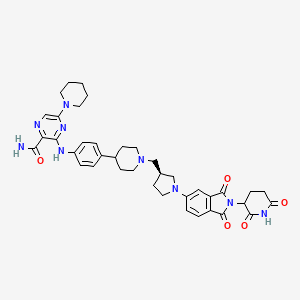
Zelebrudomide
CAS 2416131-46-7
MF C39H45N9O5 MW 719.8 g/mol
3-[4-[1-[[(3S)-1-[2-(2,6-dioxopiperidin-3-yl)-1,3-dioxoisoindol-5-yl]pyrrolidin-3-yl]methyl]piperidin-4-yl]anilino]-5-piperidin-1-ylpyrazine-2-carboxamide
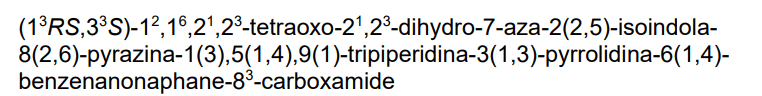
protein degrader, antineoplastic, NX 2127, LSC67HA8DE, NX-2127, BTK Degrader NX-2127
Zelebrudomide (NX-2127) is an investigational new drug that is being evaluated by Nurix Therapeutics for the treatment of relapsed or refractory B-cell malignancies such as chronic lymphocytic leukemia (CLL), mantle cell lymphoma (MCL), diffuse large B-cell lymphoma (DLBCL), and Waldenström macroglobulinemia (WM). It is an orally bioavailable proteolysis targeting chimera (PROTAC) designed to degrade Bruton’s tyrosine kinase (BTK) along with the immunomodulatory proteins Ikaros (IKZF1) and Aiolos (IKZF3).[1]
- OriginatorNurix
- ClassAntineoplastics; Small molecules
- Mechanism of ActionAgammaglobulinaemia tyrosine kinase degraders; IKZF1 protein degraders; IKZF3 protein degraders
Phase IChronic lymphocytic leukaemia; Diffuse large B cell lymphoma; Follicular lymphoma; Lymphoma; Mantle-cell lymphoma; Marginal zone B-cell lymphoma; Waldenstrom’s macroglobulinaemia
- 09 Dec 2024Pharmacodynamics data from a preclinical studies in Chronic lymphocytic leukaemia released by Nurix Therapeutics
- 11 Jul 2024NX 2127 is still in phase I development in Chronic-lymphocytic-leukaemia (Late-stage disease, Second-line therapy or greater) in USA (PO) (NCT04830137)
- 11 Jul 2024NX 2127 is still in phase I development in Diffuse large B cell lymphoma(Late-stage disease, Second-line therapy or greater) in USA (PO) (NCT04830137)
Zelebrudomide, (S)- is the S-enantiomer of zelebrudomide, an orally bioavailable chimeric targeting molecule (CTM) and targeted degrader of Bruton’s tyrosine kinase (BTK), with potential immunomodulatory drug (IMiD) and antineoplastic activities. Zelebrudomide is comprised of a cereblon (CRBN)-binding moiety conjugated, via a linker, to a BTK-binding moiety. Upon administration, zelebrudomide targets and binds to BTK with its BTK-targeting moiety. Upon binding, the CRBN-binding moiety recruits CRBN, a component of the CRL4-CRBN E3 ubiquitin ligase complex. This catalyzes ubiquitination and proteasome-mediated degradation of BTK, and prevents the activation of the B-cell antigen receptor (BCR) signaling pathway. This prevents both B-cell activation and BTK-mediated activation of downstream survival pathways. This leads to an inhibition of the growth of malignant B-cells that overexpress BTK. In addition, zelebrudomide catalyzes the degradation of CRBN neosubstrates Aiolos (IKZF3) and Ikaros (IKZF1), two transcription factors regulating T-cell function. This modulates the activity of the immune system and increases the activation of T-lymphocytes, thereby increasing T-cell-mediated anti-tumor effects. BTK, a member of the src-related BTK/Tec family of cytoplasmic tyrosine kinases, is overexpressed in B-cell malignancies; it plays an important role in B-lymphocyte development, activation, signaling, proliferation and survival. CRBN, the substrate recognition component of the CRL4-CRBN E3 ubiquitin ligase complex, plays a key role in the ubiquitination of certain proteins. Compared to BTK inhibitors, zelebrudomide may overcome tumor resistance associated with BTK inhibitor-induced resistance mutations.
A Study of NX-2127 in Adults With Relapsed/Refractory B-cell Malignancies
CTID: NCT04830137
Phase: Phase 1
Status: Recruiting
Date: 2025-03-13
REF
- Discovery and Preclinical Pharmacology of NX-2127, an Orally Bioavailable Degrader of Bruton’s Tyrosine Kinase with Immunomodulatory Activity for the Treatment of Patients with B Cell MalignanciesPublication Name: Journal of Medicinal ChemistryPublication Date: 2024-02-01PMID: 38300987DOI: 10.1021/acs.jmedchem.3c01007
- The IUPHAR Guide to Immunopharmacology: connecting immunology and pharmacologyPublication Name: ImmunologyPublication Date: 2020-03-02PMCID: PMC7160657PMID: 32020584DOI: 10.1111/imm.13175
- Effect of osmotic pressure, ionic strength and dibutyryl cyclic adenosine monophosphate on the adhesion of hen erythrocytesPublication Name: Blut Zeitschrift für die gesamte BlutforschungPublication Date: 1976-07PMID: 10025DOI: 10.1007/bf01005212
SYN
compound 28 Journal of Medicinal ChemistryPublication Date: 2024-02-01PMID: 38300987DOI: 10.1021/acs.jmedchem.3c01007
SYN
WO2021219070A1
PAT
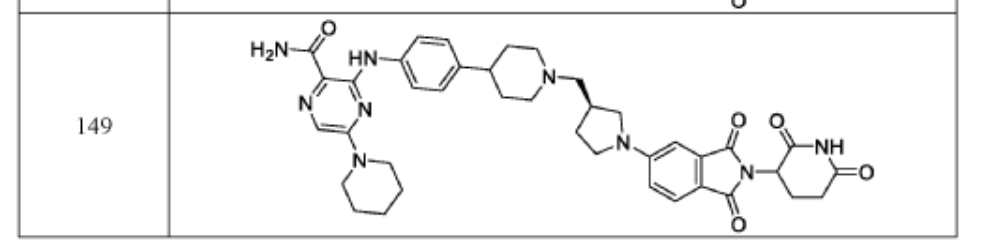
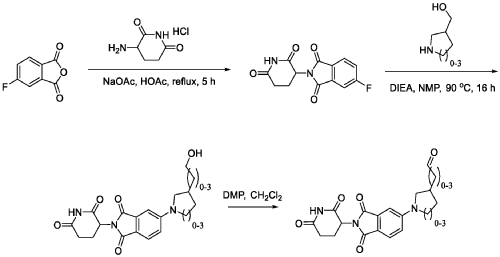
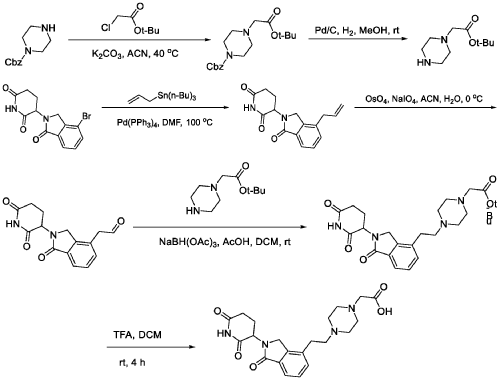
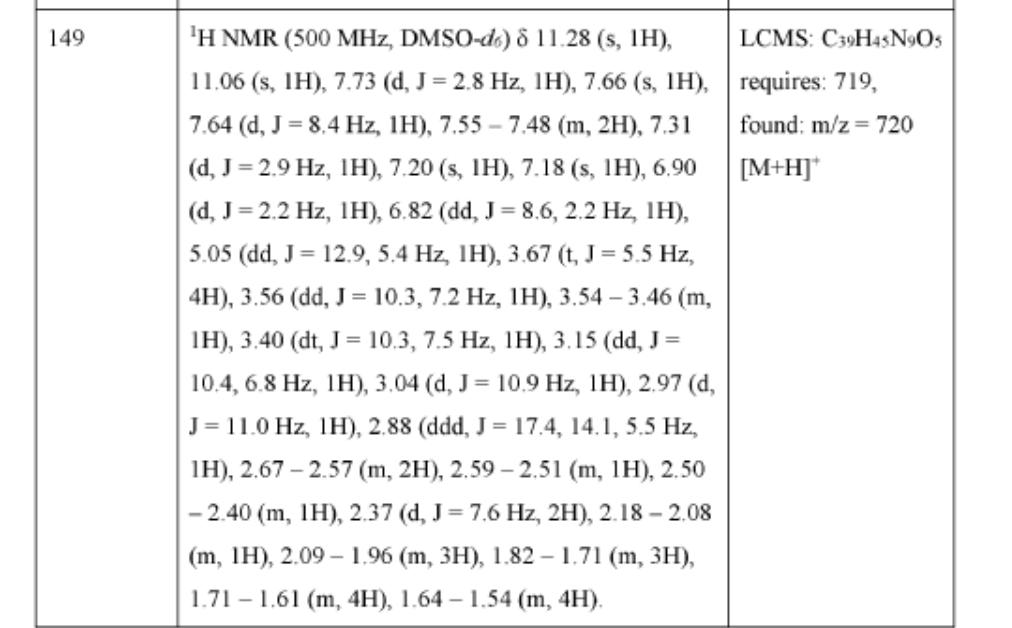
PAT
- Bifunctional compounds for degrading btk via ubiquitin proteosome pathwayPublication Number: US-2023227471-A1Priority Date: 2019-12-04
- Bifunctional compounds for degrading btk via ubiquitin proteosome pathwayPublication Number: WO-2021113557-A1Priority Date: 2019-12-04
- Bifunctional compounds for degrading btk via ubiquitin proteosome pathwayPublication Number: US-2023029378-A1Priority Date: 2018-10-15
- Bifunctional compounds for degrading btk via ubiquitin proteosome pathwayPublication Number: WO-2020081450-A1Priority Date: 2018-10-15
- Bifunctional compounds for degrading BTK via ubiquitin proteosome pathwayPublication Number: US-11479556-B1Priority Date: 2018-10-15Grant Date: 2022-10-25
- Btk reducing molecules for treatment of cancers and immune system disordersPublication Number: WO-2023235691-A1Priority Date: 2022-05-31
- Piperidinylpyrazine-carboxamide compounds for treating and preventing cancer and for degrading btkPublication Number: US-2023149416-A1Priority Date: 2021-10-26
- Piperidinylpyrazine-carboxamide compounds for treating and preventing cancer and for degrading btkPublication Number: WO-2023076303-A1Priority Date: 2021-10-26
- Bifunctional compounds for degrading BTK via ubiquitin proteosome pathwayPublication Number: US-11820781-B2Priority Date: 2019-12-04Grant Date: 2023-11-21
- Bifunctional compounds for degrading btk via ubiquitin proteosome pathwayPublication Number: US-2021198280-A1Priority Date: 2019-12-04
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
References
- Salvaris RT, Brennan J, Lewis KL (February 2025). “BTK Is the Target That Keeps on Giving: A Review of BTK-Degrader Drug Development, Clinical Data, and Future Directions in CLL”. Cancers. 17 (3): 557. doi:10.3390/cancers17030557. PMC 11817010. PMID 39941922.
| Clinical data | |
|---|---|
| Other names | NX-2127 |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2416131-46-7 |
| PubChem CID | 146559796 |
| ChemSpider | 128922006 |
| UNII | LSC67HA8DE |
| Chemical and physical data | |
| Formula | C39H45N9O5 |
| Molar mass | 719.847 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
//////////zelebrudomide, anax lab, protein degrader, antineoplastic, NX 2127, LSC67HA8DE, NX-2127, BTK Degrader NX-2127

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
