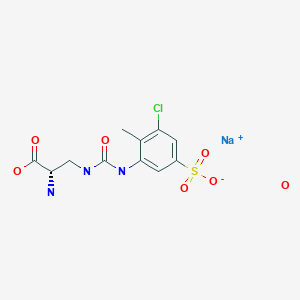
Upacicalcet sodium hydrate, ウパシカルセトナトリウム水和物
CAS 2052969-18-1
1333218-50-0 free
PMDA JAPAN APPROVED 2021/6/23, Upasita
Calcium sensing receptor agonist
(2S)-2-amino-3-[(3-chloro-2-methyl-5-sulfophenyl)carbamoylamino]propanoic acid
| Formula | C11H13ClN3O6S. Na. xH2O |
|---|
- OriginatorAjinomoto Pharma
- DeveloperSanwa Kagaku Kenkyusho
- ClassAmines; Chlorobenzenes; Propionic acids; Small molecules; Sulfonic acids; Toluenes
- Mechanism of ActionCalcium-sensing receptor agonists
- RegisteredSecondary hyperparathyroidism
- 25 Jun 2021Chemical structure information added
- 23 Jun 2021Sanwa Kagaku Kenkyusho and Kissei Pharmaceutical agree to co-promote upacicalcet in Japan for Secondary hyperparathyroidism
- 23 Jun 2021Registered for Secondary hyperparathyroidism in Japan (IV) – First global approval
| Upacicalcet Sodium HydrateMonosodium 3-({[(2S)-2-amino-2-carboxyethyl]carbamoyl}amino)-5-chloro-4-methylbenzenesulfonate hydrateC11H13ClN3NaO6S▪xH2O [2052969-18-1 , anhydride] |
Announcement of Marketing Authorization Approval in Japan and Co-promotion Agreement of UPASITA® IV Injection Syringe for the Treatment of Secondary Hyperparathyroidism in Dialysis Patients
SANWA KAGAKU KENKYUSHO Co., Ltd. (Head Office: Nagoya, President and CEO : Shusaku Isono, Suzuken Group, ; “SANWA KAGAKU”) has received Marketing Authorization approval today for UPASITA® IV Injection Syringes (generic name: Upacicalcet Sodium Hydrate; “UPASITA®”) for the treatment of secondary hyperparathyroidism in patients on hemodialysis.
UPASITA® was created by Ajinomoto Pharmaceuticals Co., Ltd. (currently EA Phama Co., Ltd.) and developed by SANWA KAGAKU for the treatment of secondary hyperparathyroidism under a licensing agreement with EA Pharma. UPASITA® acts on calcium sensing receptor in the parathyroid and suppresses excessive secretions of parathyroid hormones (PTH). UPASITA® is administered by intravenous injection to dialysis patients through dialysis circuit by physicians or medical staffs upon completion of dialysis and such administration is expected to reduce the burden of patients with many oral medications whose drinking water volume is severely restricted.
Regarding provision of medical and drug information, SANWA KAGAKU entered into a co-promotion agreement in Japan with Kissei Pharmaceutical Co., Ltd. (Head Office: Matsumoto, Nagano; Chairman and CEO: Mutsuo Kanzawa ; “Kissei”). SANWA KAGAKU will handle the production, marketing, and distribution of the Product while SANWA KAGAKU and Kissei collaboratively promote it to medical institutions in the field in accordance with the agreement. Through the co-promotion activity in the field, SANWA KAGAKU and Kissei will contribute to the treatment of dialysis patients suffering from secondary hyperparathyroidism.
《Reference》
About secondary hyperparathyroidism (SHPT)
SHTP is one of complications that occur as chronic kidney disease (chronic kidney failure) progresses and is a pathological condition where excessive PTH is secreted by the parathyroid gland. It has been reported that excessive secretion of parathyroid hormone promotes efflux of phosphorus and calcium from the bone into the blood, thereby increasing the risk of developing bone fractures and arteriosclerosis due to calcification of the cardiovascular system and affecting the vital prognosis.
Product Summary of UPASITA® IV Injection Syringe for Dialysis
Brand name:
UPASITA® IV Injection Syringe for Dialysis 25μg
UPASITA® IV Injection Syringe for Dialysis 50μg
UPASITA® IV Injection Syringe for Dialysis 100μg
UPASITA® IV Injection Syringe for Dialysis 150μg
UPASITA® IV Injection Syringe for Dialysis 200μg
UPASITA® IV Injection Syringe for Dialysis 250μg
UPASITA® IV Injection Syringe for Dialysis 300μg
Generic Name (JAN):
Upacicalcet Sodium Hydrate
Date of Marketing Approval:
June 23, 2021
Indications:
Secondary hyperparathyroidism in patients on hemodialysis
Dosage and Administration:
In adults, UPASITA® is usually administered into venous line of the dialysis circuit at the end of dialysis session during rinse back at a dose of 25 μg sodium upacicalcet 3 times a week as a starting dose.
The starting dose can be 50 μg depending on the concentration of serum calcium. Thereafter, the dose may be adjusted in a range from 25 to 300 μg while parathyroid hormone (PTH) and serum calcium level should be carefully monitored in patients.
SYN
WO 2020204117

PATENT
WO 2011108724
WO 2011108690
JP 2013063971
WO 2016194881
JP 6510136
PATENT
WO 2016194881
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2016194881&tab=FULLTEXT(Example 1) Synthesis of
(2S) -2-amino-3-{[(5-chloro-2-hydroxy-3-sulfophenyl) carbamoyl] amino} propanoic acid (Compound 1 )
[Chemical formula 14]CDI 150. 2 g (926.6Mmol, 1.1 eq. vs Boc-DAP-O t Bu) to and stirred at 5 ° C. acetone was added 750mL (3.0L / kg). 250 g (842.6 mmol) of Boc-DAP-OtBu was added in two portions, and the mixture was washed with 125 mL (0.5 L / kg) of acetone. After stirring for 30 minutes, completion of the IC (imidazolylcarbonylation) reaction was confirmed by HPLC. 282.6 g (1263.8 mmol, 1.5 eq.) Of ACHB was added in 3 portions, and the mixture was washed with 125 mL (0.5 L / kg) of acetone. After raising the temperature to 30 ° C. and stirring for 18 hours, the completion of the urea conversion reaction was confirmed by HPLC. After cooling to 5 ° C., 124.5 mL (1432.4 mmol, 1.7 eq.) Of concentrated hydrochloric acid was added, and the mixture was stirred for 1 hour. The precipitated unwanted material was filtered and washed with 1000 mL (4.0 L / kg) of acetone. The filtrate was concentrated to 1018 g (4.1 kg / kg), the temperature was raised to 50 ° C., and 625.0 mL (7187 mmol, 8.5 eq.) Of concentrated hydrochloric acid was added dropwise. After stirring for 30 minutes and confirming the completion of deprotection by HPLC, 750 mL of water was added (3.0 L / kg). This liquid was concentrated under reduced pressure to 1730 g (6.9 kg / kg) to precipitate a solid. After stirring at 20 ° C. for 14 hours, vacuum filtration was performed. The filtered solid was washed with 500 mL (2.0 L / kg) of acetone and then dried under reduced pressure at 60 ° C. for 6 hours to obtain 201.4 g of the target product (64.5%).
1H-NMR (400MHz, DMSO-d6): δ 8.3 (s, 1H), 8.2 (bs, 3H), 8.1 (d, 1H, J = 2.6Hz), 7.3 (t, 1H, J = 6.0Hz), 7.0 (d, 1H, J = 2.6Hz), 4.0-4.1 (m, 1H), 3.6-3.7 (m, 1H), 3.4-3.5 (m, 1H)[0026](Example 2) Synthesis of
(2S) -2-amino-3-{[(3-sulfophenyl) carbamoyl] amino} propanoic acid (Compound 2 )
[Chemicalformula 15] CDI 120.2 g (741.2 mmol, 1. 600 mL (3.0 L / kg) of acetone was added to 1 eq. Vs Boc-DAP-OtBu), and the mixture was stirred at 5 ° C. 200 g (673.9 mmol) of Boc-DAP-OtBu was added in two portions, and the mixture was washed with 100 mL (0.5 L / kg) of acetone. After stirring for 30 minutes, the completion of the IC reaction was confirmed by HPLC. 175.0 g (1010.8 mmol, 1.5 eq.) Of ABS was added in 3 portions and washed with 100 mL (0.5 L / kg) of acetone. After raising the temperature to 30 ° C. and stirring for 18 hours, the completion of the urea conversion reaction was confirmed by HPLC. After cooling to 5 ° C., 99.6 mL (1145.4 mmol, 1.7 eq.) Of concentrated hydrochloric acid was added, and the mixture was stirred for 1 hour. The precipitated unwanted material was filtered and washed with 1400 mL (7.0 L / kg) of acetone. The filtrate was concentrated to 800.1 g (4.0 kg / kg), heated to 50 ° C., and then 500.0 mL (5750.0 mmol, 8.5 eq.) Of concentrated hydrochloric acid was added dropwise. After stirring for 30 minutes and confirming the completion of deprotection by HPLC, 600 mL of water was added (3.0 L / kg). This liquid was concentrated under reduced pressure to 1653.7 g to precipitate a solid. After aging at 20 ° C. for 15 hours, vacuum filtration was performed. The filtered solid was washed with 400 mL (2.0 L / kg) of acetone and then dried under reduced pressure at room temperature for 6 hours to obtain 140.3 g of the desired product (net 132.2 g, 64.7%).
1H-NMR (400MHz, DMSO-d6): δ 8.8 (s, 1H), 8.2 (bs, 3H), 7.7 (s, 1H), 7.3-7.4 (m, 1H), 7.1-7.2 (m, 2H) , 6.3-6.4 (bs, 1H), 4.0-4.1 (bs, 1H), 3.6-3.7 (bs, 1H), 3.5-3.6 (bs, 1H)[0027](Example 3) Synthesis of
(2S) -2-amino-3-{[(3-chloro-2-methyl-5-sulfophenyl) carbamoyl] amino} propanoic acid (Compound 3 )
[Chemical formula 16]CDI 14. To 4 g (88.8 mmol, 1.05 eq. Vs Boc-DAP-OtBu), 75 mL (3.0 L / kg vs DAP-OtBu) of acetone was added and stirred at 5 ° C. After adding 25 g (84.3 mmol) of Boc-DAP-OtBu in two portions and stirring for 30 minutes, the completion of the IC reaction was confirmed by HPLC. 26.1 g (118.0 mmol, 1.4 eq.) Of ACTS was added in 3 portions and washed with 25 mL (1.0 L / kg) of acetone. After the temperature was raised to 30 ° C., the mixture was stirred overnight, and the completion of the urea conversion reaction was confirmed by HPLC. After concentrating under reduced pressure at 10 kPa and 40 ° C. until the solvent was completely removed, 37.5 mL (1.5 L / kg) of water and 22.8 mL (257.6 mmol) of concentrated hydrochloric acid were added to perform deprotection for 2 hours. After confirming the completion of the reaction by HPLC, the mixture was cooled to 5 ° C., 60 mL (2.4 L / kg) of MeCN was added, and the mixture was stirred overnight. Further, when 120 mL (4.8 L / kg) of MeCN was added, stratification occurred, so 10 mL (0.4 L / kg) of water and 2.5 mL (0.1 L / kg) of MeCN were added. The precipitated solid was filtered under reduced pressure, washed with 60 mL of MeCN / water (1/2), and then dried under reduced pressure at 60 ° C. for 14 hours to obtain 20.1 g of the desired product as a white solid (net18.3 g, yield 61). 0.8%).
1H-NMR (400MHz, DMSO-d6): δ 14.70-13.30 (bs, 1H), 8.27 (bs, 3H), 8.15 (s, 1H), 7.98 (d, 1H, J = 1.6Hz), 7.27 (d , 1H, J = 1.6Hz), 6.82 (t, 1H, J = 6.0Hz), 4.04 (bs, 1H), 3.70-3.60 (m, 1H), 3.60-3.50 (m, 1H), 2.22 (s, 3H)[0028](Example 4) Synthesis of
compound 3 using phenylchloroformate as a carbonyl group-introducing reagent
(Step 1)
[Chemicalformula 17] MeCN 375 mL (7.5 L / kg vs ACTS), Py for 50 g (225.6 mmol) of ACTS. 38.1 mL (473.7 mmol, 2.1 eq.) Was added and stirred at 25 ° C. 29.9 mL (236.8 mmol, 1.05 eq.) Of ClCO 2 Ph (phenyl chloroformate) was added dropwise, and after stirring for 30 minutes, completion of the CM (carbamate) reaction was confirmed by HPLC. 68.9 g (232.4 mmol) of Boc-DAP-OtBu was added, 97.5 mL (699.3 mmol, 3.1 eq.) Of TEA was added dropwise, and the mixture was stirred at 25 ° C. for 3 hours. The completion of the urea conversion reaction was confirmed by HPLC. Here, 103.5 g of the total amount of 517.43 g was used to move to the next step (down to ACTS 10 g scale).
30 mL of water was added and concentrated to 77.0 g at 40 ° C. and 5 kPa. After 100 mL (10 L / kg) of AcOEt was added and the liquid separation operation was performed, 30 mL of water was added to the organic layer and the liquid separation operation was performed again. The organic layer was concentrated to 47.6 g at 40 ° C. and 10 kPa, and then 15 mL (1.5 L / kg) of AcOEt and 100 mL (10 L / kg) of THF were added. Again, it was concentrated to 50.7 g and THF was added up to 146 g. When it was concentrated again to 35.5 g and added to AcOEt 30 mL (3 L / kg) and THF 100 mL (10 L / kg), a solid was precipitated. It was cooled to 5 ° C. and aged overnight. The precipitated solid was filtered under reduced pressure, washed with 20 mL (2.0 L / kg) of THF, and then dried under reduced pressure at 40 ° C. for 3 hours overnight at 30 ° C. to obtain 24.9 g of the desired product as a white solid (net). 23.0 g, 83.6%).
1 H-NMR (400MHz, DMSO-d6): δ 8.86 (bs, 1H), 8.09 (s, 1H), 7.88 (s, 1H), 7.25 (d, 1H, J = 1.6Hz), 7.14 (d, 1H, J = 7.6Hz), 6.60 (t, 1H, J = 5.6Hz), 4.00-3.90 (m, 1H), 3.60-3.50 (m, 1H), 3.30-3.20 (m, 1H), 3.15-3.05 (m, 6H), 2.19 (s, 3H), 1.50-1.30 (m, 18H), 1.20-1.10 (m, 9H)
(Step 2)
[Chemical
formula 18] Compound 4 21.64 g (net. 20.0 g, 68 mL of water (3.4 L / kg vs. compound 4) vs. 32.8 mmol) ) Was added, the mixture was stirred at 50 ° C., and 12 mL (135.6 mmol, 4.1 eq.) Of concentrated hydrochloric acid was added dropwise. After stirring for 1 hour, the temperature was raised to 70 ° C. to dissolve the precipitated solid. After confirming the completion of the reaction by HPLC, the mixture was cooled to 50 ° C. and aged for 1 hour, and then cooled to 5 ° C. over 4 hours. The precipitated solid was filtered under reduced pressure, washed with 40 mL (2.0 L / kg) of MeCN / water (2/1), and then dried under reduced pressure at 60 ° C. for 3 hours to obtain 11.2 g of the desired product as a white solid (11.2 g). net 10.5 g, 91.1%).[0029](Example 5)
[Chemicalformula 19] MeCN 10.0 mL (10.0 L / kg vs ACSS), Py 0.75 mL (9.25 mmol, 2.05 eq.) For 1.00 g (4.51 mmol) of ACTS. , And stirred at 8 ° C. After dropping 0.59 mL (4.74 mmol, 1.05 eq.) Of ClCO 2 Ph, raising the temperature to room temperature and stirring for 1 hour, completion of the CM conversion reaction was confirmed by HPLC. 1.33 g (4.51 mmol, 1.0 eq.) Of Boc-DAP-OtBu was added, 1.92 mL (13.76 mmol, 3.05 eq.) Of TEA was added dropwise, and the mixture was stirred at 40 ° C. for 1 hour. After confirming the completion of the urea conversion reaction by HPLC, the mixture was concentrated until the solvent was completely removed. 1.0 mL of water and 2.0 mL of concentrated hydrochloric acid (22.6 mmol, 5.0 eq.) Were added, and the mixture was stirred at 50 ° C. for 4 hours. After confirming the completion of deprotection by HPLC, MeCN 7.5 mL (7.5 L / kg), 1 M HCl aq. After adding 4.5 mL, the mixture was stirred at 5 ° C. overnight. The precipitated solid was filtered under reduced pressure, washed with 3.0 mL (3.0 L / kg) of MeCN, and then dried at 60 ° C. overnight to obtain 1.28 g of the desired product as a white solid (net 1.18 g, 77). .0%).[0030](Example 6)
(Step 1)
3-({[(2S) -2-amino-3-methoxy-3-oxopropyl] carbamoyl} amino) -5-chloro-4-methylbenzene-1-sulfonic acid ( Synthesis of Compound 5 )
[Chemical formula 20] To5 g (22.56 mmol) of ACTS, 37.5 mL (7.5 L / kg vs ACTS) of MeCN and 3.81 mL (47.38 mmol, 2.1 eq.) Of Py were added. The mixture was stirred at 25 ° C. 2.99 mL (23.68 mmol, 1.05 eq.) Of ClCO 2 Ph was added dropwise, and after stirring for 30 minutes, the completion of the CM reaction was confirmed by HPLC. 5.92 g (23.23 mmol, 1.03 eq.) Of Boc-DAP-OMe was added, 9.75 mL (69.93 mmol, 3.1 eq.) Of TEA was added dropwise, and the mixture was stirred at 25 ° C. for 3 hours. 0.4 g (1.58 mmol, 0.07 eq.) Of Boc-DAP-OMe and 0.22 mL (1.58 mmol, 0.07 eq.) Of TEA were added, and the completion of the ureaization reaction was confirmed by HPLC. 7.32 mL (112.8 mmol, 5.0 eq.) Of MsOH was added, the temperature was raised to 50 ° C., and the mixture was stirred for 4 hours. After confirming the completion of deprotection by HPLC, the mixture was cooled to 25 ° C. and 37.5 mL (7.5 L / kg) of MeCN and 7.5 mL (1.5 L / kg) of water were added to precipitate a solid. It was cooled to 5 ° C. and aged for 16 hours. The precipitated solid was filtered under reduced pressure, washed with 20 mL (4.0 L / kg) of water / MeCN (1/2), and then dried under reduced pressure at 40 ° C. for 5 hours to obtain 7.72 g of the target product as a white solid (772 g of the target product). net 7.20 g, 87.3%).
1H-NMR (400MHz, DMSO-d6): δ 8.39 (bs, 3H), 8.16 (d, 1H, J = 1.2Hz), 7.90 (d, 1H, J = 1.6Hz), 7.28 (d, 1H, J = 1.6Hz), 6.78 (t, 1H, J = 5.6Hz), 4.20-4.10 (m, 1H), 3.77 (s, 3H), 3.70-3.60 (m, 1H), 3.55-3.45 (m, 1H) , 2.21 (s, 3H)
HRMS (FAB – ): calcd for m / z 364.0369 (MH), found The m / z 364.0395 (MH)
(step 2)
[Formula 21]
compound 5 10.64 g (net Non 10.0 g, To 27.34 mmol), 18 mL of water (1.8 L / kg vs. compound 5 ) was added and stirred at 8 ° C. 3.42 mL (57.41 mmol, 2.1 eq.) Of a 48% aqueous sodium hydroxide solution was added dropwise, and the mixture was washed with 1.0 mL (1.0 L / kg) of water and then stirred at 8 ° C. for 15 minutes. After confirming the completion of hydrolysis by HPLC, the temperature was raised to 25 ° C. and 48% HBr aq. The pH was adjusted to 5.8 by adding about 3.55 mL. After confirming the precipitation of the target product by dropping 65 mL (6.5 L / kg) of IPA, the mixture was aged for 1 hour. 81 mL (8.1 L / kg) of IPA was added dropwise and aged at 8 ° C. overnight. The precipitated solid was filtered under reduced pressure, washed with 20 mL (2.0 L / kg) of IPA, and then dried under reduced pressure at 40 ° C. for 4 hours to obtain 10.7 g of the desired product as a white solid (net 9.46 g, 92. 6%).
1 H-NMR (400MHz, DMSO-d6): δ8.76 (s, 1H), 7.91 (d, 1H, J = 1.6Hz), 8.00-7.50 (bs, 2H), 7.24 (d, 1H, J = 1.6Hz), 7.20 (t, 1H, J = 5.6Hz), 3.58-3.54 (m, 1H), 3.47-3.43 (m, 1H), 3.42-3.37 (m, 1H), 2.23 (s, 3H)[0031](Example 7)
(Step 1)
[Chemicalformula 22] For 10.0 g (45.1 mmol) of ACTS, 50 mL (5.0 L / kg vs ACTS) of MeCN, 7.46 mL (92.5 mmol, 2.05 eq. ) Was added, and the mixture was stirred at 8 ° C. 5.98 mL (47.4 mmol, 1.05 eq.) Of ClCO 2 Ph was added dropwise, the temperature was raised to 25 ° C., and the mixture was stirred for 1 hour, and then the completion of the CM reaction was confirmed by HPLC. 100 ml of acetone (10.0 L / kg vs ACTS) was added, the mixture was cooled to 8 ° C., and aged for 1 hour. The precipitated solid was filtered under reduced pressure, washed with 30 mL of acetone (3.0 L / kg vs ACTS), and then dried under reduced pressure at 60 ° C. for 2 hours to obtain 17.8 g of the target product (net 14.4 g as a free form). Quant).
1 H-NMR (400MHz, DMSO-d6): δ 9.76 (bs, 1H), 8.93-8.90 (m, 2H), 8.60-8.50 (m, 1H), 8.10-8.00 (m, 2H), 7.60 (s , 1H), 7.50-7.40 (m, 3H), 7.30-7.20 (m, 3H), 2.30 (s, 3H)
(Step 2)
[Chemical 23]
Compound 6 To 5.0 g (11.9 mmol), 50 ml of acetonitrile and 3.53 g (11.9 mmol) of Boc-DAP-OtBu were added, and the mixture was stirred at 8 ° C. 3.5 ml (25 mmol) of triethylamine was added dropwise, and the mixture was stirred overnight at room temperature. The solvent was distilled off under reduced pressure, and 25 ml of ethyl acetate and 5 ml of water were added for extraction. The organic layer was washed with 5 ml of water, the solvent was distilled off, 50 ml of tetrahydrofuran was added, the mixture was cooled to 8 ° C., and aged for 1 hour. The precipitated solid was filtered under reduced pressure, washed with 10 ml of tetrahydrofuran, and dried under reduced pressure at 60 ° C. overnight to obtain 6.3 g of the desired product as a white solid.[0032](Example 8)
[Chemicalformula 24] For 1.08 g (4.89 mmol) of ACTS, 8.1 mL (7.5 L / kg vs ACTS) of MeCN and 827 μL (10.27 mmol, 2.1 eq.) Of Py were added. In addition, it was stirred at room temperature. ClCO 2 Ph 649 μL (5.14 mmol, 1.05 eq.) Was added dropwise, and the mixture was stirred for 30 minutes, and then the completion of the CM conversion reaction was confirmed by HPLC. 1.48 g (5.04 mmol, 1.03 eq.) Of Cbz-DAP-OMe HCl was added, 2.1 mL (15.17 mmol, 3.1 eq.) Of TEA was added dropwise, and the mixture was stirred at room temperature for about 5 hours. After confirming the completion of the urea conversion reaction by HPLC, the mixture was concentrated until the solvent was completely removed. 15.0 mL of 30% HBr / AcOH was added, and the mixture was stirred at room temperature for 70 minutes, and the completion of deprotection was confirmed by HPLC. After concentration to dryness, 10 mL of water and 4 mL of AcOEt were added to carry out an extraction operation, and then the aqueous layer was stirred at room temperature overnight. The precipitated solid was filtered under reduced pressure, washed with 15 mL of water and 10 mL of AcOEt, and then dried at 40 ° C. for 3 hours to obtain 1.45 g of the desired product as a white solid (58.8%).[0033](Example 9) Synthesis of compound 7 ( methyl ester of compound 1 )
using phenyl chloroformate as a carbonyl group introduction reagent [Chemical formula 25] MeCN 73 mL (14.6 L) with respect to 5.00 g (22.4 mmol) of ACHB. / Kg vs ACHB), Py 3.8 mL (47 mmol, 2.1 eq.), Was added and stirred at 40 ° C. After adding 3.0 mL (24 mmol, 1.05 eq.) Of ClCO 2 Ph and stirring for 30 minutes, the completion of the CM conversion reaction was confirmed by HPLC. 5.87 g (23 mmol, 1.0 eq.) Of Boc-DAP-OMe was added, washed with a small amount of MeCN, 9.7 mL (70 mmol, 3.1 eq.) Of TEA was added dropwise, and the mixture was stirred at 40 ° C. for 3 hours. After confirming the completion of the urea conversion reaction by HPLC, the mixture was cooled to room temperature. 7.3 mL (112 mmol, 5.0 eq.) Of MsOH was added, the temperature was raised to 50 ° C., and the mixture was stirred for 7 hours. Further, 1.5 mL (23 mmol, 1.0 eq.) Of MsOH was added, and the reaction was carried out at 50 ° C. overnight. After confirming the completion of deprotection by HPLC, 90 mL of acetone was added to the reaction solution, and the mixture was cooled to room temperature. The precipitated solid was obtained and dried under reduced pressure at 60 ° C. to obtain the desired product. 1 H-NMR (400MHz, DMSO-d6): δ 7.22 (m, 1H), 7.14 (m, 1H), 4.36 (m, 1H), 3.80 (s, 3H), 3.20-3.40 (m, 2H).[0034](Example 10) Synthesis of
compound 5 using 4-chlorophenylchloroformate as a carbonyl group-introducing reagent
[Chemical formula 26] For5.00 g (22.6 mmol) of ACTS, 73 mL (14.6 L / kg vs ACTS) of MeCN, 3.8 mL (47 mmol, 2.1 eq.) Of Py was added and stirred at 40 ° C. After adding 3.25 mL (23.7 mmol, 1.05 eq.) Of 4-chloroformic acid 4-chlorophenylate and stirring at 40 ° C. for 1.5 hours, completion of the CM conversion reaction was confirmed by HPLC. Add 5.92 g (23.2 mol, 1.0 eq.) Of Boc-DAP-OMe, wash with a small amount of MeCN, add 9.7 mL (70 mmol, 3.1 eq.) Of TEA, and stir at 40 ° C. for 2 hours. did. After confirming the completion of the urea conversion reaction by HPLC, the mixture was cooled to room temperature. 7.3 mL (113 mmol, 5.0 eq.) Of MsOH was added, the temperature was raised to 50 ° C., and the mixture was stirred for 3.5 hours. After confirming the completion of deprotection by HPLC, the reaction solution was cooled to room temperature, 7.5 mL of water was added, the mixture was cooled to 8 ° C., and the mixture was stirred overnight. The precipitated solid was filtered, washed with a small amount of MeCN water, and dried at 60 ° C. overnight to obtain 6.94 g of the desired product as a white solid (84.1%).[0035](Example 11) Synthesis of
compound 5 using 4-nitrophenyl chloroformate as a carbonyl group-introducing reagent
[Chemical formula 27]73 mL (14.6 L / kg vs. ACTS) of MeCN with respect to 5.00 g (22.6 mmol) of ACTS. , Py 3.8 mL (47 mmol, 2.1 eq.), And stirred at 40 ° C. 4.77 mL (23.7 mmol, 1.05 eq.) Of 4-nitrophenyl chloroformate was added dropwise, and the mixture was stirred at 40 ° C. for 3.5 hours, and then the completion of the CM reaction was confirmed by HPLC. Add 5.92 g (23.2 mmol, 1.0 eq.) Of Boc-DAP-OMe, wash with a small amount of MeCN, add 9.7 mL (70 mmol, 3.1 eq.) Of TEA, and stir at 40 ° C. for 2 hours. did. After confirming the completion of the urea conversion reaction by HPLC, the mixture was cooled to room temperature. 7.3 mL (113 mmol, 5.0 eq.) Of MsOH was added, the temperature was raised to 50 ° C., and the mixture was stirred for 3.5 hours. After confirming the completion of deprotection by HPLC, the reaction solution was cooled to room temperature, 7.5 mL of water was added, the mixture was cooled to 8 ° C., and the mixture was stirred overnight. The precipitated solid was filtered, washed with a small amount of MeCN water, and dried at 60 ° C. overnight to obtain 5.96 g of the desired product as a white solid (72.2%).[0036](Example 12) Synthesis of
compound 3 using Boc-DAP-OH
[Chemical 28]MeCN 73 mL (14.6 L / kg vs ACTS), Py 3.8 mL, relative to 5.00 g (22.6 mmol) of ACTS. (47 mmol, 2.1 eq.) Was added and stirred at 40 ° C. After adding 3.00 mL (23.8 mmol, 1.05 eq.) Of phenylchloroformate and stirring at 40 ° C. for 0.5 hours, the completion of the CM conversion reaction was confirmed by HPLC (CM conversion reaction product: 4.37 minutes). , ACTS: N.D.). Add 4.75 g (23.2 mmol, 1.0 eq.) Of Boc-DAP-OH, wash with a small amount of MeCN, add 9.7 mL (70 mmol, 3.1 eq.) Of TEA, and stir at 40 ° C. for 2 hours. did. After confirming the completion of the urea-forming reaction by HPLC (urea-forming reaction product: 3.81 minutes, CM-forming reaction product: 0.02 area% vs. urea-forming reaction product), the mixture was cooled to room temperature. By adding 7.3 mL (113 mmol, 5.0 eq.) Of MsOH, raising the temperature to 50 ° C., stirring for 4.5 hours, and further adding 1.5 mL (23 mmol, 1.0 eq.) Of MsOH, stirring for 1 hour. , The formation of the target product was confirmed by HPLC (Compound 3: 2.49 minutes, urea conversion reaction product: 0.50 area vs. compound 3, area of compound 3 with respect to the total area excluding pyridine: 71.0 area).
PATENT
JP 6510136
PATENT
WO 2020204117
Reference Example 1
Synthesis of 3-{[(2S) -2-amino-2-carboxyethyl] carbamoylamino} -5-chloro-4-methylbenzenesulfonate sodium (Compound A1)
(Step 1)
Synthesis of
3 -({[(2S) -2-amino-3-methoxy-3-oxopropyl] carbamoyl} amino) -5-chloro-4-methylbenzene-1-sulfonic acid 3-amino- 37.5 mL (7.5 L / kg vs ACTS) of acetonitrile and 3.81 mL (47.38 mmol, 2.1 eq.) Of pyridine against 5 g (22.56 mmol) of 5-chloro-4-methylbenzenesulfonic acid (ACTS). Was added and stirred at 25 ° C. 2.99 mL (23.68 mmol, 1.05 eq.) Of ClCO 2 Ph was added dropwise, and after stirring for 30 minutes, the completion of the carbamate reaction was confirmed by HPLC. Add 5.92 g (23.23 mmol, 1.03 eq.) Of 3-amino-N- (tert-butoxycarbonyl) -L-alanine methyl ester hydrochloride and 9.75 mL (69.93 mmol, 3.1 eq.) Triethylamine. Was added dropwise, and the mixture was stirred at 25 ° C. for 3 hours. Add 0.4 g (1.58 mmol, 0.07 eq.) Of 3-amino-N- (tert-butoxycarbonyl) -L-alanine methyl ester hydrochloride and 0.22 mL (1.58 mmol, 0.07 eq.) Of triethylamine. Then, the completion of the urea conversion reaction was confirmed by HPLC. 7.32 mL (112.8 mmol, 5.0 eq.) Of methanesulfonic acid was added, the temperature was raised to 50 ° C., and the mixture was stirred for 4 hours. After confirming the completion of deprotection by HPLC, the mixture was cooled to 25 ° C. and 37.5 mL (7.5 L / kg) of acetonitrile and 7.5 mL (1.5 L / kg) of water were added to precipitate a solid. It was cooled to 5 ° C. and aged for 16 hours. The precipitated solid was filtered under reduced pressure, washed with 20 mL (4.0 L / kg) of water / acetonitrile (1/2), and then dried under reduced pressure at 40 ° C. for 5 hours to obtain 7.72 g of the desired product as a white solid (. net 7.20 g, 87.3%).
1 H-NMR (400MHz, DMSO-d6): δ 8.39 (bs, 3H), 8.16 (d, 1H, J = 1.2Hz), 7.90 (d, 1H, J = 1.6Hz), 7.28 (d, 1H, J = 1.6Hz), 6.78 (t, 1H, J = 5.6Hz), 4.20-4.10 (m, 1H), 3.77 (s, 3H), 3.70-3.60 (m, 1H), 3.55-3.45 (m, 1H) ), 2.21 (S, 3H)HRMS (FAB – ): Calcd For M / Z 364.0369 (MH & lt;), Found M / Z 364.0395 (MH & lt;)
(Step 2)
(2)
Compound obtained in step 1 of synthesis of 3-{[(2S) -2-amino-2-carboxyethyl] carbamoylamino} -5-chloro-4-methylbenzenesulfonate . To 64 g (net 10.0 g, 27.34 mmol), 18 mL of water (1.8 L / kg vs. the compound of Step 1) was added, and the mixture was stirred at 8 ° C. 3.42 mL (57.41 mmol, 2.1 eq.) Of a 48% aqueous sodium hydroxide solution was added dropwise, and the mixture was washed with 1.0 mL (1.0 L / kg) of water and then stirred at 8 ° C. for 15 minutes. After confirming the completion of hydrolysis by HPLC, the temperature was raised to 25 ° C. and 48% HBr aq. About 3.55 mL was added to adjust the pH to 5.8. After confirming the precipitation of the desired product by dropping 65 mL (6.5 L / kg) of isopropyl alcohol, the mixture was aged for 1 hour. 81 mL (8.1 L / kg) of isopropyl alcohol was added dropwise and the mixture was aged at 8 ° C. overnight. The precipitated solid was filtered under reduced pressure, washed with 20 mL (2.0 L / kg) of isopropyl alcohol, and then dried under reduced pressure at 40 ° C. for 4 hours to obtain 10.7 g of the desired product as a white solid (net 9.46 g, 92). .6%).
1 H-NMR (400MHz, DMSO-d6): δ8.76 (s, 1H), 7.91 (d, 1H, J = 1.6Hz), 8.00-7.50 (bs, 2H), 7.24 (d, 1H, J = 1.6Hz), 7.20 (t, 1H, J = 5.6Hz), 3.58-3.54 (m, 1H), 3.47-3.43 (m, 1H), 3.42-3.37 (m, 1H), 2.23 (s, 3H)

///////////Upacicalcet sodium hydrate, Upasita, ウパシカルセトナトリウム水和物 , APPROVALS 2021, JAPAN 2021, Upacicalcet
NEW DRUG APPROVALS
ONE TIME
$10.00

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
