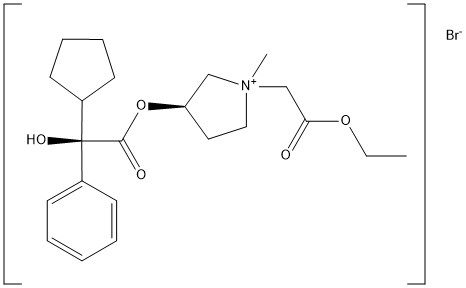
Sofpironium bromide
ソフピロニウム臭化物
BBI 4000
[(3R)-1-(2-ethoxy-2-oxoethyl)-1-methylpyrrolidin-1-ium-3-yl] (2R)-2-cyclopentyl-2-hydroxy-2-phenylacetate;bromide
| Formula |
C22H32NO5. Br
|
|---|---|
| CAS |
1628106-94-4
BASE 1628251-49-9
|
| Mol weight |
470.3972
|
PMDA APPROVED JAPAN 2020/9/25, Ecclock
Anhidrotic
Sofpironium Bromide
1-ambo-(3R)-3-{[(R)-(Cyclopentyl)hydroxy(phenyl)acetyl]oxy}-1-(2-ethoxy-2-oxoethyl)-1-methylpyrrolidinium bromide
C22H32BrNO5 : 470.4
[1628106-94-4]
SYN
PATENT
WO 2018026869
https://patents.google.com/patent/WO2018026869A1/en
Certain glycopyrronium salts and related compounds, as well as processes for making and methods of using these glycopyrronium salts and related compounds, are known. See, for example, US Patent No. 8,558,008, which issued to assignee Dermira, Inc. See also, for example, US Patent No. 2,956,062, which issued to assignee Robins Co Inc. A H. See also, for example, International Patent Application Publication Nos. WO 98/00132 Al and WO 2009/00109A1, both of which list applicant Sepracor, Inc., as well as US Patent Nos. 6,063,808 and 6,204,285, both of which issued to assignee Sepracor, Inc. Certain methods of treating hyperhidrosis using glycopyrronium salts and related compounds are known. See, for example GB 1,080,960. Certain forms of applying glycopyrrolate compounds to a subject are known. See, for example US Patent Nos. 6,433,003 and 8,618,160, both of which issued to assignee Rose U; also US Patent Nos. 7,060,289; 8,252,316; and 8,679,524, which issued to PurePharm, Inc.
[0004] One glycopyrronium salt which is useful in certain medical applications is the following compound:
[0005] As illustrated above, the absolute configuration at the three asymmetric chiral positions is 2R3’R1’RS. This means that the carbon indicated with the number, 2, has the stereochemical R configuration. The carbon indicated with the number, 3′, also has the stereochemical R configuration. The quatemary ammonium nitrogen atom, indicated with a positive charge, may have either the R or the S stereochemical configuration. As drawn, the compound above is a mixture of two diastereoisomers.
[0006] Certain processes for making glycopyrronium salts are known. However, these processes are not as safe, efficient, stereospecific, or stereoselective as the new processes disclosed herein, for example with respect to large-scale manufacturing processes. Certain publications show that higher anticholinergic activity is attributed to the 2R3’R configuration. However, to date, processes for making the 2R3’R isomers, as well as the 2R3’R1’R isomers are low yielding, involve too many reaction steps to be economically feasible, use toxic materials, and/or are not sufficiently stereospecific or stereoselective with respect to the products formed.
EXAMPLE 2
[0179] The below synthetic description refers to the numbered compounds illustrated in FIG. 2. Numbers which refer to these compounds in FIG. 2 are bolded and underlined in this Example.
[0180] Synthesis of R(-)-Cyclopentylmandelic acid (4)
[0181] R(-)-cyclopentylmandelic acid (compound 4) can be synthesized starting with
R(-)-mandelic acid (compound 1) according to Example 1.
[0182] Step 1 : Making Compound 2.
[0183] R(-)-mandelic acid (1) was suspended in hexane and mixed with pivaldehyde and a catalytic amount of trifluoromethanesulfonic acid at room temperature to form a mixture. The mixture was warmed to 36 °C and then allowed to react for about 5 hours. The mixture was then cooled to room temperature and treated with 8% aqueous sodium bicarbonate. The aqueous layer was removed and the organic layer dried over anhydrous sodium sulfate. After filtration and removal of the solvent under vacuum, the crude product was recrystallized to give (5R)-2-(tert-butyl)-5-phenyl-l,3-dioxolan-4-one (compound 2) in 88% yield (per S-enantiomer yield).
[0184] Step 2: Making Compound 3.
[0185] Compound 2 was reacted with lithium hexamethyl disilazide (LiHMDS) in hexane at -78 °C under stirring for one hour. Next, cyclopentyl bromide was added to the reaction mixture including compound 2 and LiHMDS . The reaction was kept cool for about four (4) hours and then slowly warmed to room temperature and allowed to react for at least twelve (12) more hours. The resulting mixture was then treated with 10% aqueous ammonium chloride. The aqueous layer was discarded and the organic layer dried over anhydrous sodium sulfate. The solvent was removed under vacuum and the residue recrystallized from hexane to give pure product (5R)-2-(tert-butyl)-5-cyclopentyl-5-phenyl- l,3-dioxolan-4-one (3) in 63% yield (per S-enantiomer yield).
[0186] Step 3: Making Compound 4.
[0187] R(-)-cyclopentylmandelic acid (compound 4) was prepared by providing compound 3 in aqueous methanolic potassium hydroxide at 65 °C for four hours. After cooling this mixture to room temperature and removing the methanol under vacuum, the aqueous solution was acidified with aqueous hydrochloric acid. The aqueous solution was then extracted twice with ethyl acetate and the organic phase dried with anhydrous sodium sulfate. After removing the solvent and performing a recrystallization, pure R(-)- cyclopentylmandelic acid (compound 4) was obtained in 62% yield (based on S-enantiomer yield).
[0188] Next, a racemic mixture of l -methyl-3-pyrridinol (20) was provided:
[0189] Synthesis of 2R3 ‘R-glycopyrrolate base (8)
[0190] Step 4: Making Compound 8.
[0191] Enantiomerically pure R(-)-cyclopentylmandelic acid (4) was coupled to racemic l-methyl-3-pyrridinol (20) using 1, 1 -carbonyldiimideazole (CDI) activated esterification to make an enantiomerically pure mixture of the following erythro- and threo- glycopyrrolate bases (compounds 8 and 21, respectively):
[0192] The 2R3’R-glycopyrrolate base (compound 8) was then resolved using the 5- nitroisophthalate salt procedure in Finnish Patent 49713, to provide enantiomerically pure 2R3 Έ. {erythro) as well as pure 2R3 ‘S {threo). In this example, the 2R3 ‘S {threo) was discarded. The 2R3 Έ. {erythro) was separated as stereomerically pure compound 8.
[0193] Step 6: Making Compound 9.
[0194] The glycopyrrolate base, compound 8, was treated in dry acetonitrile with methyl bromoacetate at room temperature under stirring for three (3) hours. The crude product was dissolved in a small volume of methylene chloride and poured into dry ethyl ether to obtain a precipitate. This procedure was repeated three times to provide (3R)-3-((R)- 2-cyclopentyl-2-hydroxy-2-phenylacetoxy)-l -(2-ethoxy-2-oxoethyl)-l-methylpyrrolidin-l – ium bromide, also known as 3′(R)-[R-Cyclopentylphenylhydroxyacetoy]- -ethyl- l ‘methoxycarbonylpyrrolidinium bromide (compound 9) in 89% yield. Compound 9 included the following stereoisomers:
E


Synthesis of 9a, 9b, 13a, and 13b.
| Publication Number | Title | Priority Date | Grant Date |
|---|---|---|---|
| US-2019161443-A1 | Processes for making, and methods of using, glycopyrronium compounds | 2016-08-02 |
ClinicalTrials.gov
| CTID | Title | Phase | Status | Date |
|---|---|---|---|---|
| NCT02058264 | A Safety, Tolerability and Preliminary Efficacy Study of BBI-4000 in Subjects With Axillary Hyperhidrosis | Phase 1 | Completed | 2014-09-11 |
NIPH Clinical Trials Search of Japan
| CTID | Title | Phase | Status | Date |
|---|---|---|---|---|
| JapicCTI-184249 | A repeatedly applied study of BBI-4000 in patients with primary hyperhidrosis | complete | 2018-12-13 | |
| JapicCTI-184003 | A long term safety study of BBI-4000 gel in patients with primary axillary hyperhidrosis | complete | 2018-06-15 | |
| JapicCTI-183948 | A confirmatory study of BBI-4000 gel in patients with primary axillary hyperhidrosis | complete | 2018-05-07 | |
| UMIN000020546 | A skin irritation study of BBI-4000 in healthy adult males (phase 1) | Complete: follow-up complete | 2016-01-18 |
////////////Sofpironium bromide, Ecclock, 2020 APPROVALS, JAPAN 2020, Anhidrotic, ソフピロニウム臭化物 , BBI 4000
CCOC(=O)C[N+]1(CCC(C1)OC(=O)C(C2CCCC2)(C3=CC=CC=C3)O)C.[Br-]

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
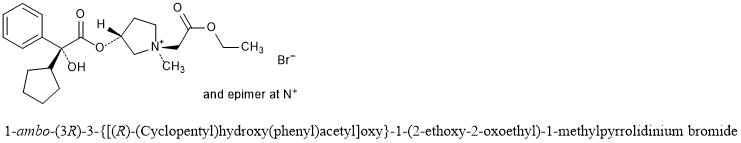





Wow I just loved your article on the beginners guide to starting a blog. I want to start one and I am now pretty clear after reading this post. This was very helpful for me and I am definitely going to mention you in my blog as soon as I start it. Thanks you so much
Neuro Psychiatric PCD Pharma Companies
Neuropsychiatry Products Franchise Company
Franchise Neuropsychiatric Company
Neuropsychiatric Pharma Franchise
Neuropsychiatry PCD Pharma Franchise
Neuropsychiatry Products Franchise Company
Neuro Pharma Companies in India
LikeLike