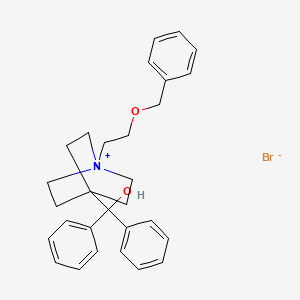
Umeclidinium bromide
GSK-573719A, ウメクリジニウム臭化物
- Molecular FormulaC29H34BrNO2
- Average mass508.490 Da
Umeclidinium bromide (trade name Incruse Ellipta) is a long-acting muscarinic antagonist approved for the maintenance treatment of chronic obstructive pulmonary disease (COPD).[1] It is also approved for this indication in combination with vilanterol (as umeclidinium bromide/vilanterol).[2][3]
In the 2014, the drug was also approved in the E.U. and in the U.S. for the maintenance treatment to relieve symptoms in adult patients with chronic obstructive pulmonary disease (COPD). It was launched in the U.K. in October 2014 and in the U.S. in January 2015. In Japan, the product candidate was approved in 2015 as monotherapy for the maintenance bronchodilator treatment to relieve symptoms in adult patients with chronic obstructive pulmonary disease (COPD) and launched on October in the same year.

Umeclidinium bromide (Ellipta)
Umeclidinium bromide is a long-acting muscarinic acetylcholine antagonist developed by GlaxoSmithKline and approved by the US FDA at the end of 2013 for use in combination with vilanterol, a b2 agonist, for the treatment of chronic obstructive pulmonary disease.269 Due to umeclidinium’s poor oral bioavailability, the drug is administrated by inhalation as dry powder.269
The most likely scale preparation of the drug is described in Scheme .270
Commercially available ethyl isonipecotate (278) was alkylated with 1-bromo-2-chloroethane in the presence of K2CO3 in acetone to give ethyl 1-(2-chloroethyl)piperidine-4-carboxylate (279). This material was then treated with lithium diisopropylamine (LDA) in THF to affect a transannular substitution reaction resulting in the cyclized quinuclidine 280 in 96% yield.270 Excess of phenyllithium was added to ester 280 in THF starting at low temperature then gradually warming to room temperature to give tertiary alcohol 281 in 61% yield. Amine 281 was finally alkylated with benzyl 2-bromoethyl ether (282) in MeCN/CHCl3 at elevated temperatures
to afford umeclidinium bromide (XXXV) in 69% yield.
269. Tal-Singer, R.; Cahn, A.; Mehta, R.; Preece, A.; Crater, G.; Kelleher, D.;Pouliquen, I. J. Eur. J. Pharmacol. 2013, 701, 40.
270. Laine, D. I.; McCleland, B.; Thomas, S.; Neipp, C.; Underwood, B.; Dufour, J.;Widdowson, K. L.; Palovich, M. R.; Blaney, F. E.; Foley, J. J.; Webb, E. F.;Luttmann, M. A.; Burman, M.; Belmonte, K.; Salmon, M. J. Med. Chem. 2009, 52, 2493.
FDA
https://www.accessdata.fda.gov/drugsatfda_docs/nda/2013/203975Orig1s000ChemR.pdf
1-[2-(benzyloxy)ethyl]-4-(hydroxydiphenylmethyl)-1-azoniabicyclo[2.2.2]octane bromide

PATENT
https://patents.google.com/patent/CN105461710A/en
umeclidinium bromide prepared patent US7439393, US RE44874, US 7488827, US 7498440, US7361787 and the like using phenyllithium prepared by reaction of intermediate 4 – [(diphenyl) hydroxymethyl] azabicyclo [2.2.2 ] octane.Specific methods: azabicyclo [2.2.2] octane-nucleophilic addition reaction with 4-carboxylate-fold amount of 2.02-2.5 phenyllithium occurs, the reaction temperature is controlled to -78 ° 0_15 ° C ο lithium Reagents expensive, difficult to store, use of harsh conditions, relatively high cost.
Example 1
Phenyl magnesium chloride: Under nitrogen atmosphere to 55g (2.3mol) of metallic magnesium sandpaper lit with 3 L of tetrahydrofuran was added dropwise 215g (1.91mol) chlorobenzene, micro-thermal reaction proceeds, controlled dropping, the reaction was kept boiling, dropwise for about 1.5 hours, after the dropping was heated slightly under reflux for 30min. Cool reserve.
[0008] Example 2
Phenyl magnesium bromide: The under argon 50.4g (2.lmol) sandpaper lit magnesium metal with 4.2 liters of anhydrous ethyl ether was added a solution of 300g (1.91mol) of bromobenzene, was added an iodine initiator, electrical hair fever reaction proceeds, controlled dropping, the reaction was kept boiling, about 1.5 hours dropwise was added dropwise to a gentle reflux heated 30min. Cool reserve.
[0009] Example 3
Preparation of crude product: azabicyclo [2.2.2] octane-4-carboxylate (135g, 0.736mo 1) was dissolved in 3L of tetrahydrofuran, under nitrogen, was cooled to -5~0 ° C, was added dropwise 300g preparation of benzyl bromide Grignard reagent. After incubation -5~0 ° C stirred for 1 hour (progress of the reaction was monitored by TLC sample). Adding 50ml of water quenching. Liquid separation, the aqueous phase was extracted twice with 500ml of tetrahydrofuran, and the combined organic phases were washed with water, dried and filtered. The solvent was partially removed under reduced pressure, the balance maintaining approximately 1L, the residue was stirred overnight at 20 ° C crystallization.Filtered, washed (petroleum ether 2 X 200 ml), the filter cake was dried at 40 ° C in vacuo to give a yellowish white crystals 121.2 g, yield 54.2%.
[0010] Example 4
Preparation of crude product: azabicyclo [2.2.2] octane-4-carboxylate (18.3g, 0.lOmo 1) was dissolved in 3L of tetrahydrofuran, under nitrogen, was cooled to 0~5 ° C, was added dropwise 0.25 mol phenyl magnesium chloride. After incubation 0~5 ° C stirred for 1 hour (progress of the reaction was monitored by TLC sample) o quenched with 10ml of water was added. Liquid separation, the aqueous phase was extracted twice with 100ml of tetrahydrofuran, and the combined organic phases were washed with water, dried and filtered. The solvent was partially removed under reduced pressure, the balance maintaining approximately 50mL, the residue was stirred overnight at 20 ° C crystallization.Filtered, washed (petroleum ether 2X20 ml), the filter cake was dried at 40 ° C in vacuo to give a yellowish white crystals 14.63 g, yield 48.1%.
[0011] Example 5
Preparation of crude product: azabicyclo [2.2.2] octane-4-carboxylate (18.38,0.1011101) ^ 31 was dissolved in tetrahydrofuran, under nitrogen, was cooled to 5~15 ° C, was added dropwise 0.30 mol of benzene bromide. After incubation 5~15 ° C stirred for 1 hour (progress of the reaction was monitored by TLC sample) o quenched with 10ml of water was added. Liquid separation, the aqueous phase was extracted twice with 100ml of tetrahydrofuran, and the combined organic phases were washed with water, dried and filtered. The solvent was partially removed under reduced pressure, the balance maintaining approximately 50mL, the residue was stirred overnight at 20 ° C crystallization.Filtered, washed (petroleum ether 2 X 20 ml), the filter cake was dried at 40 ° C in vacuo to yield 13.80 g of yellow-white crystals, yield 47.1%.
[0012] Example 6
Umeclidinium bromide purification: 100g crude product was dissolved in 320ml of water to 80 ° C a mixture of 640ml of acetone, add 5g active carbon, and filtered.The filtrate was cooled to 25 ° C, for 1 hour. Within 1 to 2 hours and cooled to 0~5 ° C for 3 hours. The filter cake with chilled 1: 2 acetone – washed twice with water (2x20ml). The filter cake was dried in vacuo at 60 ° C to give white crystalline solid (92 g, yield 92%). Purity (HPLC normalization method) 99.25%.
[0013] Example 7
Umeclidinium bromide purification: 100g crude product was dissolved in 180ml water at 50 ° C a mixture of 360ml of acetone, add 5g active carbon, and filtered.The filtrate was ~ 2 hours to 25 ° C, for 1 hour. Within 1 to 2 hours cooled to 0 ° C and left overnight protection. The filter cake with chilled 1: 2 acetone – washed twice with water (2x20ml). The filter cake was dried at 60 ° C in vacuo to give fine (98.3 g, yield 98.3%). Purity (HPLC normalization method) 97.75%.
PATENT
https://patents.google.com/patent/WO2014027045A1
International Patent Publication Number WO 2005/104745 (Glaxo Group Limited), filed 27th April 2005, discloses muscarinic acetylcholine receptor antagonists. In particular, WO 2005/104745 discloses 4- [hydroxy(diphenyl)methyl]-l-{2-[(phenylmethyl)oxy]ethyl}-l-azoniabicyclo[2.2.2]octane bromide, of formula (I), and a process for the preparation of this compound (Example 84):
4-[Hydroxy(diphenyl)methyl]-l-{2-[(phenylmethyl)oxy]ethyl}-l-azoniabicyclo[2.2.2]octane bromide may also be referred to as umeclidinium bromide.
International Patent Publication Number WO 2011/029896 (Glaxo Group Limited), filed 10th September 2010, discloses an alternative preparation for an early intermediate, ethyl-l-azabicyclo[2.2.2] octane-4-carboxylate, in the multi-step synthesis of umeclidinium bromide.
There exists a need for an alternative process for the preparation of umeclidinium bromide. In particular, a process that offers advantages over those previously disclosed in WO 2005/104745 and WO 2011/029896 is desired. Advantages may include, but are not limited to, improvements in safety, control (i.e of final product form and physical characteristics), yield, operability, handling, scalability, and efficiency.
Summary of the Invention
The present invention provides, in a first aspect, a process for the preparation of umeclidinium bromide, which comprises: a) reacting ((2-bromoethoxy)methyl)benzene, of formula (II)
in a dipolar aprotic solvent with a boiling point greater than about 90°C or an alcohol with a boiling point greater than about 80°C; and optionally
b) re-crystallising the product of step (a).
The present invention is further directed to intermediates used in the preparation of the compound of formula (III), and hence of umeclidinium bromide. The process disclosed herein provides a number of advantages over prior art processes of WO 2005/104745 and WO 2011/029896.

PATENT
EP 3248970
FORM A B AND AMORPHOUS
https://patents.google.com/patent/EP3248970A1/en
The invention relates to novel solid forms of umeclidinium bromide (I), chemically 1-[2-(benzyloxy)ethyl]-4-(hydroxydiphenylmethyl)-1-azabicyclo[2.2.2]octane bromide. In particular, to its novel crystalline forms, identified as form A and form B, as well as to an amorphous form, and to their characterization by means of analytic methods. The invention further relates to methods of their preparation and their use for the preparation of umeclidinium bromide in the API quality.
Umeclidinium bromide is indicated as an inhalation anticholinergic drug with an ultra-long-term effect in cooperating patients with the diagnosis of COPD (chronic obstructive pulmonary disease). COPD is defined as a preventable and treatable disease that is characterized by a persistent obstruction of air flow in the bronchi (bronchial obstruction), which usually progresses and is related to an intensified inflammatory response of the airways to harmful particles or gases. The main goal of the treatment of COPD is an improvement of the current control, i.e. elimination of symptoms, improvement of toleration of physical effort, improvement of the health condition and reduction of future risks, i.e. prevention and treatment of exacerbations, prevention of progression of the disease and mortality reduction
The structure of umeclidinium bromide, 1-[2-(benzyloxy)ethyl]-4-(hydroxydiphenylmethyl)-1-azabicyklo[2.2.2]octane bromide, is first mentioned in the general patent application WO2005009362 of 2003 .
Preparation of umeclidinium bromide is first disclosed in the patent EP 1 740 177B ( WO2005104745 ), where two methods (A and B) are mentioned, differing in the final processing and the product yield (method B included in Scheme 1). There, the last steps of the synthesis are described, the product being described by means of EI-MS, 1H NMR and elementary analysis. There is no information concerning the chemical purity or polymorphic form.
-
- Example 5
Preparation of the amorphous form of umeclidinium bromide
PAPER
Synthetic Communications An International Journal for Rapid Communication of Synthetic Organic Chemistry , Volume 48, 2018 – Issue 9, Convenient new synthesis of umeclidinium bromide
Umeclidinium bromide, a drug used for chronic obstructive pulmonary disease, is synthesized through a new intermediate of phenyl(quinuclidin-4-yl)methanone. This novel method with simple operation flow and cheap reagents, makes it suitable for scale up. The overall four-step process provides umeclidinium bromide in 29% yield and the purity up to 99.83%. The X-ray crystal structure of the drug molecule was first reported.



External links
References
- ^ Jump up to:a b “Incruse Ellipta (umeclidinium inhalation powder) for Oral Inhalation Use. Full Prescribing Information” (PDF). GlaxoSmithKline, Research Triangle Park, NC 27709. Retrieved 22 February 2016.
- Jump up^ Feldman, GJ; Edin, A (2013). “The combination of umeclidinium bromide and vilanterol in the management of chronic obstructive pulmonary disease: Current evidence and future prospects”. Therapeutic advances in respiratory disease. 7 (6): 311–9. doi:10.1177/1753465813499789. PMID 24004659.
- Jump up^ “FDA Approves Umeclidinium and Vilanterol Combo for COPD”. Medscape. December 18, 2013.
 |
|
| Clinical data | |
|---|---|
| Trade names | Incruse Ellipta |
| Synonyms | GSK573719A |
| License data | |
| Pregnancy category |
|
| Routes of administration |
Inhalation (DPI) |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | ~89%[1] |
| Metabolism | Hepatic (CYP2D6) |
| Elimination half-life | 11 hours |
| Excretion | Feces (58%) and urine(22%) |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| KEGG | |
| ChEBI | |
| ECHA InfoCard | 100.166.375 |
| Chemical and physical data | |
| Formula | C29H34BrNO2 |
| Molar mass | 508.49 g/mol |
| 3D model (JSmol) | |
//////////////Umeclidinium bromide, Incruse Ellipta, ウメクリジニウム臭化物 , GSK573719A, UNII-7AN603V4JV, FDA 2014
C1C[N+]2(CCC1(CC2)C(C3=CC=CC=C3)(C4=CC=CC=C4)O)CCOCC5=CC=CC=C5.[Br-]
Synthesis
| FDA Orange Book Patents: 1 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 9750726 |
| Expiration | Nov 29, 2030 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 2 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 6759398 |
| Expiration | Aug 3, 2021 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 3 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 7439393 |
| Expiration | May 21, 2025 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 4 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 7629335 |
| Expiration | Aug 3, 2021 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 5 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 7776895 |
| Expiration | Sep 11, 2022 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 6 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 8161968 |
| Expiration | Feb 5, 2028 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 7 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 8201556 |
| Expiration | Feb 5, 2029 |
| Applicant | GLAXO GRP ENGLAND |
| Drug Application | N205382 (Prescription Drug: INCRUSE ELLIPTA . Ingredients: UMECLIDINIUM BROMIDE) |
| FDA Orange Book Patents: 8 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 6537983 |
| Expiration | Aug 3, 2021 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 9 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 7498440 |
| Expiration | Apr 27, 2025 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 10 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 7488827 |
| Expiration | Dec 18, 2027 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 11 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 8183257 |
| Expiration | Jul 27, 2025 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 12 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 6878698 |
| Expiration | Aug 3, 2021 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 13 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 8511304 |
| Expiration | Jun 14, 2027 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 14 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | RE44874 |
| Expiration | Mar 23, 2023 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|
| FDA Orange Book Patents: 15 of 15 (FDA Orange Book Patent ID) | |
|---|---|
| Patent | 8309572 |
| Expiration | Apr 27, 2025 |
| Applicant | GLAXOSMITHKLINE |
| Drug Application |
|

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....