Cadexomer Iodine
Cadex, Declat, Decrat, Dextrinomer iodine, Iodoflex, Iodosorb, NI-009
CAS 94820-09-4
Listed in 1984 (Perstorp, Finland). For the treatment of exudative and infectious wounds, such as venous ulcers. This product is in contact with wound exudate to form a non-adhesive protective layer and release antibacterial iodine
Product of reaction of dextrin with epichlorohydrin coupled with ion-exchange groups and iodine
Cadexomer iodine is an iodophor that is produced by the reaction of dextrin with epichlorhydrin coupled with ion-exchange groups and iodine. It is a water-soluble modified starch polymer containing 0.9% iodine, calculated on a weight-weight basis, within a helical matrix.[1]
The Central Drugs Standard Control Organization (CDSCO) is the Central Drug Authority for discharging functions assigned to the Central Government under the Drugs and Cosmetics Act. One of the major functions of CDSCO is approval of new drugs in the country. During the month of March 2018, CDSCO has approved the following drugs classifying them as New Drug Approvals
Cadexomer Iodine Bulk & Powder 100 % w/w (contain 0.9 % w/v Iodine) or Cadexomer Iodine Ointment 500 mg (contains 0.9% w/v iodine)
For the treatment of chronic exuding wounds such as leg ulcers, pressure ulcers and diabetes ulcers infected traumatic and surgical wounds.
Cadexomer iodine is an iodophor that is produced by the reaction of dextrin with epichlorhydrin coupled with ion-exchange groups and iodine. It is a water-soluble modified starch polymer containing 0.9% iodine, calculated on a weight-weight basis, within a helical matrix.
In India, M/s Virchow Biotech Private Limited presented their proposal for grant of license to manufacture and market this product in India. The firm presented the Phase III Clinical trial report titled ‘Safety and efficacy of Dexadine (Cadexomer Iodine) in the treatment of chronic wounds’ before the CDSCO’s Subject Expert Committee on Antibiotics & Antivirals. After detailed deliberation, the committee recommended the manufacturing and marketing of the products (Cadexomer Iodine Ointment & Cadexomer Iodine Powder), as topical preparations for the treatment of chronic exuding wounds
History
Cadexomer iodine was developed in the early 1980s in Sweden by Perstorp AB, and given the name Iodosorb. The product was shown to be effective in the treatment of venous ulcers,.[2][3] More recently, it has been shown in studies in animals and humans that, unlike the iodophor povidone-iodine, Iodosorb causes an acceleration of the healing process in chronic human wounds. This is due to an increase in epidermal regeneration and epithelialization in both partial-thickness and full-thickness wounds.[4] In this way cadexomer iodine acts as a cicatrizant.
Properties
When formulated as a topical wound dressing Iodosorb adsorbs exudate and particulate matter from the surface of granulating wounds and, as the dressing becomes moist, iodine is released. The product thus has the dual effect of cleansing the wound and exerting a bactericidal action.
Uses
In addition to other manufacturers, Smith & Nephew distributes cadexomer iodine as Iodosorb and Iodoflex in many countries of the world for the treatment and healing of various types of wounds. The dosage forms are a paste dressing, an ointment and a gel, all of which contain 0.9% iodine.
PATENT
WO2001070242
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2001070242
PATENT
WO 2008117300
https://patents.google.com/patent/WO2008117300A2/und
Improved Process for the Preparation Of
Cadexomer Iodine
The present invention describes an improved method for the preparation of cadexomer iodine. Cadexomer iodine is a hydrophilic modified starch polymer containing 0.9%w/w iodine within the helical matrix. It is used for its absorbent and antiseptic properties in the management of chronic wounds such as venous leg ulcers, pressure sores, etc. It is applied as a powder or as an ointment over the wound.
Background of the invention
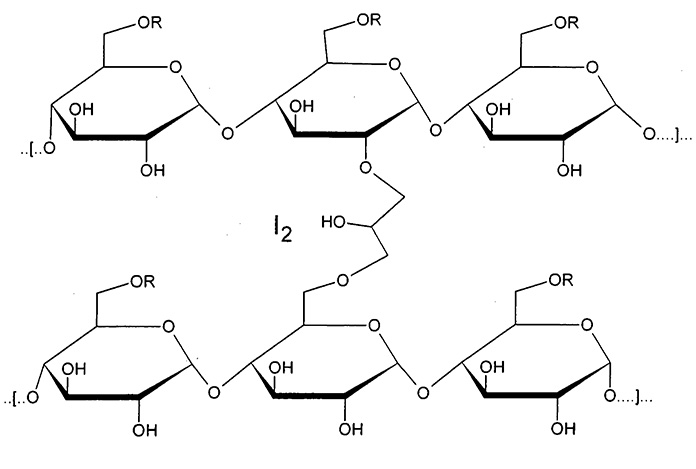
Cadexomer iodine is an iodophor that releases iodine. It contains 0.9%w/w iodine in hydrophilic modified starch carrier. It is used for its absorbent and antiseptic properties, in the management of venous leg ulcers and pressure sores, burn wounds etc. It is applied as a powder of microbeads or ointment containing iodine 0.9%w/w. When applied to the wound it absorbs fluids, removing exudates, pus and debris. As they swell, iodine is released which kills bacteria. When the color of the gel changes it indicates that the dressing should be changed. It is structurally represented as shown figure 1 , and chemically is known as2-hydroxy methylene cross-linked (1-4) α-D-glucan wther containing iodine.

R=H, CH2COOH
Figure: ! Structural representation of cadexomer iodine
The method of preparation of cadexomer iodine and it applications in clinical use is described in the US patent 4,010,259(1977). The process basically consists of two steps. The step one involves preparation of water insoluble, gel forming, and water swell able organic hydrophilic carrier. The next stage involves complexation of iodine with the above organic polymeric carrier.
The carrier is prepared by a polymerization /cross-linking reaction of a polyhydroxylic organic substance by means of a bifunctional organic cross-linking agent of the type Y-R-Z, wherein Y and Z each represent epoxy groups or halogen atoms and R is an organic residue. In this polymerization/cross linking reaction each of the functional groups Y and Z react with a hydroxy group of the polyhydroxylic organic material to form ether bonds. The linking has to proceed to the extent that the formed polymer becomes insoluble in water, but is capable of absorbing water.
The polyhydroxylic starting material used is dextrin or carboxy methyl dextrin and the cross linking agent used for the polymerization reaction is a bifunctional glycerol derivative such as epichlorohydrin, which is capable of forming ether bridges. The reaction between polyhdyroxy starting material and cross-linking agent epichlorohydrin is carried out by emulsion/suspension of polymerization reaction. This type reaction requires specially designed reactors with efficient stirring and an agent to disperse/ stabilize the reaction mass.
The reaction conditions mentioned in the patent uses toluene/water emulsion system, and which is stabilized/dispersed using toluene solution of a mixture of mono and di-esters of ortho phosphoric acid. This process has the following disadvantages:
Disadvantages of the prior art process
1. During cross-linking, the reaction mixture gets dried-up and sticks to the reaction vessel.
2. Efficient stirring is not possible due to formation of lumps.
3. Particles size of the cross-linked carrier is not uniform.
4. Iodine incorporation to carrier is not efficient; hence large excess has to be used.
5. The color of the product obtained by this process is dark brown, whereas product is expected to be golden yellow in color.
6. Results are not reproducible and batch-to-batch variations observed.
7. The stabilizer solution referred in the patent (US 4,010,259) is a solution of a mixture of mono and di-esters of ortho phosphoric acid, which is not available commercially..
Essentially similar procedures are described in Fr, Demande 2,320,1 12 (1977),
Australian 506,419 (1980), Finn 59,014(1981 ), Dan Dk 150,781 ( 1989). However the chemical nature and details of composition of stabilizer solution are not disclosed in these patents also.
An improved method for the preparation of cadexomer iodine is now developed free of these problems and which can easily scaled up to manufacturing level.
ADVANTAGES OF PRESENT INVENTION
1. The particle size of cadexomer iodine by the present process is fine and uniform, which is highly suitable for powder and ointment formulations.
2. Iodine incorporation to the cross-linked dextrin is efficient and consistent and swelling is appropriate
3. The color of cadexomer iodine obtained is golden yellow which is consistent and as per the expected color of the product.
4. The process is simple and economical and can be carried out in regular reactor with out any extra investment on the specialized equipment
5. Present process uses the dispersing agents, which are available commercially.
The details of the invention are described in examples given below which are provided to illustrate the invention only and therefore should not be construed to limit the scope of the present invention.
Example 1
Commercial dextrin (5Og) is dissolved in sodium hydroxide (50ml of 3.1N) containing sodium borohydride (0.75g), to this dispersing agent; sorbitan monooleate (Span 80, 3.75g) dissolved in toluene (125ml) is added. Then of epichlorohydrin (10 g) is added and reaction mixture is heated at 700C for 5h. After completion of 5h, water (600ml) is added to the reaction mixture, and then neutralized to a pH of 6.5 with hydrochloric acid (2N). The product is filtered washed with acetone (500ml). The product is again washed with water (1000ml) and finally with acetone (300ml). The wet product is treated with a solution of iodine (7.8g) in acetone (196ml) and stirred at 250C for 20 hours, then at O0C for 2 hours. The product is filtered in a sintered funnel under nitrogen atmosphere, washed with chilled acetone (150ml) and dried at 250C for 24h in a vacuum
desiccator.
Yield: 33g
Iodine content: .0.91 % w/w
Swelling capacity: 5.0ml/g
Example 2
Commercial dextrin (1Og) is dissolved in sodium hydroxide (10ml of 3.1N) containing sodium borohydride (0.15g); to this dispersing agent; cetrimide (0.25g) dissolved in toluene (25ml) is added. Then of epichlorohydrin (2.Og) is added and reaction mixture is heated at 700C for 5h. After completion of 5h, water (150 ml) is added, and then the reaction mixture was neutralized to a pH of 6.5 with hydrochloric acid (2N). The separated product was filtered and washed with acetone (100ml). Again the product washed with water (200ml) and finally with acetone (60ml). The wet product (carrier) is treated with a solution of iodine ( 1 ,6g) in acetone (40 ml) and stirred at 250C for 20 hours, then at O0C for 2 hours. The product is filtered in a sintered funnel under nitrogen atmosphere, washed with chilled acetone (40ml) and dried at 250C for 24h in a vacuum desiccator.
Yield: 4.2g
Iodine content: 0.91% w/w
Swelling capacity: 6.0ml/g
Example 3
Commercial dextrin (1Og) is dissolved in sodium hydroxide (10ml of 3.1N) containing sodium borohydride (0.15g), to this dispersing agent; glyceryl monostearate (0.25g) dissolved in toluene (25ml) is added. Then of epichlorohydrin (2.Og) is added and reaction mixture is heated at 700C for 5h. After the completion of 5h, water (150 ml) is added, and then the reaction mixture is neutralized to a pH of 6.5 with hydrochloric acid (2N). The separated product is filtered and washed with acetone (100ml). Again the product is washed with water (200ml) and finally with acetone (60ml). The wet product (carrier) is treated with a solution of iodine (1.6g) in acetone (40 ml) and stirred at 250C for 20 hours, then at O0C for 2 hours. The product is filtered in a sintered funnel under nitrogen atmosphere, washed with chilled acetone (40ml) and dried at 250C for 24h in a vacuum desiccator.
Yield: 3.3g
Iodine content: 0.9% w/w
Swelling capacity: 6.2ml/g
Example 4
Commercial carboxymethyl dextrin (20g) was dissolved in sodium hydroxide (20ml of 3.1N) containing sodium borohydride (0.3g), to this dispersing agent; glyceryl monostearate (1.Og) dissolved in toluene (75ml) is added. Then of epichlorohydrin (6.Og) is added and reaction mixture is heated at 700C for 5h After completion of 5h, water (280 ml) is added, then the reaction mixture is neutralized to a pH of 6.5 with hydrochloric acid (2N). The separated product is filtered and washed with acetone (250ml). Again the product is washed with water (500ml) and finally with acetone (150ml). The wet product (carrier) is treated with a solution of iodine (3.Ig) in acetone (60 ml) and stirred at 250C for 20 hours, then at O0C for 2 hours. The product is filtered in a sintered funnel under nitrogen atmosphere, washed with chilled acetone (60ml) and dried at 250C for 24h in a vacuum desiccator.
Yield: 16gms
Iodine content: 0.92 % w/w.
Swelling capacity: 5.8 ml per gram.
References
- Jump up^ Merck Index, 14th Edition, p262 Merck & Co. Inc.
- Jump up^ Skog, E. et al. (1983). A randomized trial comparing cadexomer iodine and standard treatment in the out-patient management of chronic venous ulcers. British Journal of Dermatology 109, 77. PMID 6344906
- Jump up^ Ormiston, M.C., Seymour, M.T., Venn, G.E., Cohen, R.I. and Fox, J.A. (1985). Controlled trial of Iodosorb in chronic venous ulcers. British Medical Journal (Clinical Research Edition) 291, 308-310. PMID 3962169
- Jump up^ Drosou Anna, Falabella Anna, and Kirsner Robert S. (2003) Antiseptics on Wounds: An area of controversy. Wounds 159(5) 149-166. http://cme.medscape.com/viewarticle/456300_2Retrieved 02/03/2009
Tang, M.B.; Tan, E.S.
Hailey-Hailey disease: Effective treatment with topical cadexomer iodine
J Derm Treat 2011, 22(5): 304
Early diagnosis and early corticosteroid administration improves healing of peristomal pyoderma gangrenosum in inflammatory bowel disease
Dis Colon Rectum 2009, 52(2): 311
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| ATC code | |
| Identifiers | |
| CAS Number | |
| ChemSpider |
|
//////////////Cadexomer Iodine, ind 2018, Cadex, Declat, Decrat, Dextrinomer iodine, Iodoflex, Iodosorb, NI-009,

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
