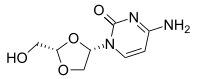
Troxacitabine
CAS 145918-75-8
- Molecular FormulaC8H11N3O4
- Average mass213.191 Da
троксацитабин
تروكساسيتابين
曲沙他滨
2(1H)-Pyrimidinone, 4-amino-1-[(2S,4S)-2-(hydroxymethyl)-1,3-dioxolan-4-yl]-
Hmd-cytosine; NCGC00183848-01; Beta-L-Dioxolane-cytidine; 4-amino-1-[(2S)-2-(hydroxymethyl)-1,3-dioxolan-4-yl]pyrimidin-2-one; 2R(-)-cis-Hmd-cytosine, (-)-ODDC
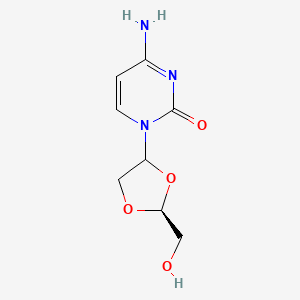
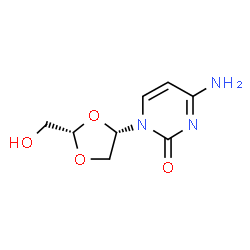
4-amino-1-[(2S)-2-(hydroxymethyl)-1,3-dioxolan-4-yl]pyrimidin-2-one
Troxacitabine (brand name Troxatyl) is a nucleoside analogue with anticancer activity. Its use is being studied in patients with refractory lymphoproliferative diseases.[1]
Troxacitabine (brand name Troxatyl) is a nucleoside analogue with anticancer activity. Its use is being studied in patients with refractory lymphoproliferative diseases.
| Investigated for use/treatment in leukemia (myeloid). |
PATENT
https://www.google.com/patents/WO1992018517A1?cl=en
WO 9218517
SYNTHESIS
WO 2016030335

PATENT
WO 2016030335
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2016030335&recNum=1&maxRec=&office=&prevFilter=&sortOption=&queryString=&tab=PCTDescription
PATENT
WO-2017031994
MACHINE TRANSLATED FROM CHINESE, BEAWARE OF FUNNY NAMES
Qu sand gemcitabine (4-amino -1 – [(2S, 4S) -2- ( hydroxymethyl) -1,3-dioxolan-4-yl] pyrimidin-2-one, Troxacitabine, Troxatyl (TM) ) Is an anti-tumor cytidine analogue developed by Yale University. In a multi-year Phase I / II clinical study in the United States, tacitabine was administered alone or mixed with other chemotherapeutic agents in a variety of dosage regimens, treating more than 825 patients with multiple solid tumors or blood Malignant tumor patients. In particular, tricatadine has the ability to inhibit the growth of hepatitis B virus and anti-hepatoma cells.
Chinese Patent Application No. 201310275643.2 discloses a method for the synthesis of tricatadine, which uses a L-menthol ester of dihydroxyacetic acid as a raw material and undergoes condensation reaction with hydroxyacetaldehyde, and then the hydroxyl group is halogenated to obtain a halide , The halide is coupled with cytosine to obtain the coupling, and the conjugate is reduced to obtain tricatadine. However, the present inventors have found that the method requires a four-step reaction, such as condensation, halogenation, coupling and reduction, which is required to be carried out in different reaction systems. The steps are long and cumbersome, and in particular, the intermediate product is required to be separated and replaced Containers, and not suitable for amplification, it is not suitable for industrial production.
the present invention provides a process for the synthesis of a compound of formula III, wherein the synthesis reaction formula is as follows:

Example 1 Synthesis of tricatadine
The synthetic route is as follows:
Step 1: Preparation of Formula II
18.0 g of methylene chloride was added to the reaction kettle, and the mixture of the formula I was homogeneously added. After the temperature was lowered to 0 ± 3 ° C under the protection of nitrogen, 1.5 g of trimethyl iodosilane was slowly added; (V / v), and R f = 0.5 at the point of disappearance). The reaction was carried out under nitrogen atmosphere for 2.5 ± 0.5 hours until the reaction was complete (sampling TLC test: developing solvent: petroleum ether: ethyl acetate = 4: 1 (v / v) Subsequently, the temperature of the autoclave was kept at 0 ± 3 ° C, and 3.64 g of hexamethyldisilazane and 1.15 g of N 4 -acetyl cytosine were slowly added dropwise . After the completion of the addition, the temperature of the control kettle was 0 ± 3 ℃, and the reaction was carried out under the protection of nitrogen for 3.5 ± 0.5 hours until the reaction was complete (sampling TLC test: developing agent: petroleum ether: ethyl acetate = 4: 1 (v / v), R F = 0.2 points disappear).
Then, the temperature was maintained at 22 ± 3 ° C, and 10 %% (w / w) aqueous sodium thiosulfate solution was slowly added dropwise. After adding 5 g of aqueous sodium thiosulfate solution, 0.5 g of diatomaceous earth was added, hour. Filter, filter cake washed with methylene chloride 3 times, filter cake collection stand-by. The filtrate and the washing liquid were combined into the kettle, the aqueous phase and the organic phase were separated. The organic phase was washed once with 11.3 g of saturated brine. The organic phase was separated and dried overnight with anhydrous sodium sulfate to remove the water. Remove the sodium sulfate solid, the filtrate into the rotary evaporator, steaming temperature does not exceed 45 ℃, until the end of distillation. The residue obtained by steaming was transferred to a reaction vessel, 11.2 g of acetone and 18.5 g of isopropyl acetate were added, and the mixture was heated to reflux (68 ± 3 ° C) and stirred for 1 hour. Within 2.5 ± 0.5 hours, slowly cool down until the kettle temperature is 22 ± 3 ° C. The filter was filtered in a vacuum oven at about 40 ° C and dried overnight under vacuum to give a white solid (formula II).
The diatomaceous earth cake obtained by the above-mentioned filtration was transferred to a reaction vessel, heated to 27 ± 3 ° C, 18.0 g of methylene chloride was added, and the mixture was stirred and stirred for 2 hours. The filtrate was filtered and the filtrate was transferred to a rotary evaporator. The steaming temperature did not exceed 45 ° C until the distillation was completed. The crude solid obtained by steaming (the crude product of formula II) and the white solid used in the previous step were combined and transferred to a reaction kettle, and an isopropyl acetate: acetone = 3: 2 (v / v) mixed solvent was added (1 g of crude (13.3 g of isopropyl acetate + 7.9 g of acetone) was added and heated to reflux (68 ± 3 ° C), and the mixture was stirred for 1 hour. In 2.5 ± 0.5 hours, slow down to the kettle temperature of 22 ± 3 ℃. Quickly filter the filter cake with cold acetone 1.5g once. The filter cake was placed in a vacuum oven at about 40 ° C and dried overnight under vacuum to give the formula II.
Step 2: Preparation and purification of formula III
Take the type II boutique 1g added to the four bottles, add methanol 5.0g, stir the solid dispersed evenly. And 0.045 g of sodium methoxide was weighed, and the mixture was added to 0.135 g of methanol and stirred to dissolve sodium methoxide. The methanol solution of sodium methoxide was added dropwise to a four-necked flask. The incubation was carried out at 22.5 ± 2.5 ° C for 1 hour until the reaction was complete (sampling TLC: developing solvent: dichloromethane: methanol = 4: 1 (v / v) and R f = 0.8).
After completion of the reaction, the pH of the system was adjusted to 6.5 ± 0.5 with ice acetic acid under ice bath. And then adding 200-300 mesh silica gel (available from Qingdao Ocean Chemical Plant) 10 g of sand, filling the column, the column chromatography, which was dichloromethane: methanol = 4: 1 (v / V), collecting the fraction containing tricatropa, and steaming to dryness. The steamed solid was transferred to a three-necked flask, 3.0 g of absolute ethanol was added, and the mixture was uniformly dispersed (suspended) and heated to 78 ± 2 ° C for 0.5 hour. After completion of the reflux, slowly (2.5 ± 0.5 hours) was cooled to room temperature and stirred at room temperature for 12 hours. Continue to cool down to 2.5 ± 2.5 ℃, at this temperature holding 4.5 ± 0.5 hours. Filter, filter cake with 1.0g cold ethanol washing once, thoroughly filter, the filtrate abandoned. The filter cake was transferred to a vacuum oven and dried at 38 ± 2 ° C until constant weight to obtain a purity of the formula III.
The above method can be equal to the proportion of stable amplification, for example, can be directly amplified about 60 to 180 times, that is, I feed 61.7g ~ 185.97g (other reactants equal ratio increase), after amplification of the final product (formula III) HPLC detection purity To 99.3% ~ 99.8%, the yield of 65 ~ 85%, fully meet the tricatitabine medicinal industrial needs.
PATENT
CN 104861067
PATENT
CN 105503838
PAPER
In vitro optimization of non-small cell lung cancer activity with troxacitabine, L-1,3-dioxolane-cytidine, prodrugs
Journal of medicinal chemistry (2007), 50, (9), 2249-53.
J. Med. Chem., 2007, 50 (9), pp 2249–2253
DOI: 10.1021/jm0612923

l-1,3-Dioxolane-cytidine, a potent anticancer agent against leukemia, has limited efficacy against solid tumors, perhaps due to its hydrophilicity. Herein, a library of prodrugs were synthesized to optimize in vitro antitumor activity against non-small cell lung cancer. N4-Substituted fatty acid amide prodrugs of 10−16 carbon chain length demonstrated significantly improved antitumor activity over l-1,3-dioxolane-cytidine. These in vitro results suggest that the in vivo therapeutic efficacy of l-1,3-dioxolane-cytidine against solid tumors may be improved with prodrug strategies.
PAPER
- Kim, Hea O.; Schinazi, Raymond F.; Shanmuganathan, Kirupathevy; Jeong, Lak S.; Beach, J. Warren; Nampalli, Satyanarayana; Cannon, Deborah L.; Chu, Chung K.
- From Journal of Medicinal Chemistry (1993), 36(5), 519-28.
PAPER
- Jin, Haolun; Tse, Allan Tse; Evans, Colleen A.; Mansour, Tarek S.; Beels, Christopher M.; Ravenscroft, Paul; Humber, David C.; Jones, Martin F.; Payne, Jeremy J.; Ramsay, Michael V. J.
- From Tetrahedron: Asymmetry (1993), 4(2), 211-14
PAPER
- Belleau, Bernard R.; Evans, Colleen A.; Tse, H. L. Allan; Jin, Haolun; Dixit, Dilip M.; Mansour, Tarek S.
- From Tetrahedron Letters (1992), 33(46), 6949-52.
PAPER
http://pubs.acs.org/doi/pdf/10.1021/jm00089a007
J. Med. Chem. 1992,35,1987-1995 Asymmetric Synthesis of 1,3-Dioxolane-Pyrimidine Nucleosides and Their Anti-HIV Activity
 References
References
- Jump up^ Vose, Julie M.; Panwalkar, Amit; Belanger, Robert; Coiffier, Bertrand; Baccarani, Michele; Gregory, Stephanie A.; Facon, Thierry; Fanin, Renato; Caballero, Dolores; Ben-Yehuda, Dina; Giles, Francis (2007). “A phase II multicenter study of troxacitabine in relapsed or refractory lymphoproliferative neoplasms or multiple myeloma”. Leukemia & Lymphoma. 48 (1): 39–45. doi:10.1080/10428190600909578.
- Lee CK, Rowinsky EK, Li J, Giles F, Moore MJ, Hidalgo M, Capparelli E, Jolivet J, Baker SD: Population pharmacokinetics of troxacitabine, a novel dioxolane nucleoside analogue. Clin Cancer Res. 2006 Apr 1;12(7 Pt 1):2158-65. [PubMed:16609029 ]
- Quintas-Cardama A, Cortes J: Evaluation of the L-stereoisomeric nucleoside analog troxacitabine for the treatment of acute myeloid leukemia. Expert Opin Investig Drugs. 2007 Apr;16(4):547-57. [PubMed:17371201 ]
- Swords R, Giles F: Troxacitabine in acute leukemia. Hematology. 2007 Jun;12(3):219-27. [PubMed:17558697 ]
- Orsolic N, Giles FJ, Gourdeau H, Golemovic M, Beran M, Cortes J, Freireich EJ, Kantarjian H, Verstovsek S: Troxacitabine and imatinib mesylate combination therapy of chronic myeloid leukaemia: preclinical evaluation. Br J Haematol. 2004 Mar;124(6):727-38. [PubMed:15009060 ]
- Boivin AJ, Gourdeau H, Momparler RL: Action of troxacitabine on cells transduced with human cytidine deaminase cDNA. Cancer Invest. 2004;22(1):25-9. [PubMed:15069761 ]
- Kim TE, Park SY, Hsu CH, Dutschman GE, Cheng YC: Synergistic antitumor activity of troxacitabine and camptothecin in selected human cancer cell lines. Mol Pharmacol. 2004 Aug;66(2):285-92. [PubMed:15266019 ]
| Patent ID |
Patent Title |
Submitted Date |
Granted Date |
| US2013011392 |
METHOD FOR ASSESSING THE ABILITY OF A PATIENT TO RESPOND TO OR BE SAFELY TREATED BY A NUCLEOSIDE ANALOG BASED-CHEMOTHERAPY |
2010-11-19 |
2013-01-10 |
| US7927613 |
Pharmaceutical co-crystal compositions |
2003-09-11 |
2011-04-19 |
| US7790905 |
Pharmaceutical propylene glycol solvate compositions |
2003-12-29 |
2010-09-07 |
//////////////TROXACITABINE, троксацитабин , تروكساسيتابين , 曲沙他滨 , Hmd-cytosineM, NCGC00183848-01, Beta-L-Dioxolane-cytidine, 2R(-)-cis-Hmd-cytosine, (-)-ODDC



 References
References

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....

